Attached files
| file | filename |
|---|---|
| 8-K - PHARMACYCLICS INC | form8k207380_11052012.htm |
| EX-99.9 - PHARMACYCLICS INC | ex999to8k207380_11052012.htm |
| EX-99.4 - PHARMACYCLICS INC | ex994to8k207380_11052012.htm |
| EX-99.8 - PHARMACYCLICS INC | ex998to8k207380_11052012.htm |
| EX-99.6 - PHARMACYCLICS INC | ex996to8k207380_11052012.htm |
| EX-99.1 - PHARMACYCLICS INC | ex991to8k207380_11052012.htm |
| EX-99.2 - PHARMACYCLICS INC | ex992to8k207380_11052012.htm |
| EX-99.5 - PHARMACYCLICS INC | ex995to8k207380_11052012.htm |
| EX-99.7 - PHARMACYCLICS INC | ex997to8k207380_11052012.htm |
| EX-99.3 - PHARMACYCLICS INC | ex993to8k207380_11052012.htm |
| EX-99.18 - PHARMACYCLICS INC | ex9918to8k207380_11052012.htm |
| EX-99.15 - PHARMACYCLICS INC | ex9915to8k207380_11052012.htm |
| EX-99.10 - PHARMACYCLICS INC | ex9910to8k207380_11052012.htm |
| EX-99.16 - PHARMACYCLICS INC | ex9916to8k207380_11052012.htm |
| EX-99.13 - PHARMACYCLICS INC | ex9913to8k207380_11052012.htm |
| EX-99.14 - PHARMACYCLICS INC | ex9914to8k207380_11052012.htm |
| EX-99.17 - PHARMACYCLICS INC | ex9917to8k207380_11052012.htm |
| EX-99.11 - PHARMACYCLICS INC | ex9911to8k207380_11052012.htm |
Exhibit 99.12
Chronic Lymphocytic Leukemia Oral Presentation
Title: In vivo inhibition of BCR activation in high-risk CLL patients on therapy with Bruton’s tyrosine kinase inhibitor Ibrutinib: correlative studies from an ongoing Phase 2 clinical trial
Session: 641. CLL - Biology and Pathophysiology, excluding Therapy: BCR and clone dynamics in mice and men
Date/Time: Sunday, December 9, 2012 Presentation Time: 5:45 PM
Location: Georgia World Congress Center, B211-B212
Presenter: Julia Hoellenriegel
Julia Hoellenriegel1*, Susan O'Brien, MD2, Michael J. Keating, MD1, William G. Wierda, MD, PhD3, Joseph J. Buggy, PhD4* and Jan A. Burger, MD, PhD5
1Department of Leukemia, The University of Texas M.D. Anderson Cancer Center, Houston, TX
2The University of Texas M.D. Anderson Cancer Center, Department of Leukemia, Houston, TX
3Department of Leukemia, The University of Texas MD Anderson Cancer Center, Houston, TX
4Pharmacyclics, Inc., Sunnyvale, CA
5Department of Leukemia, MD Anderson Cancer Center, Houston, TX
ABSTRACT:
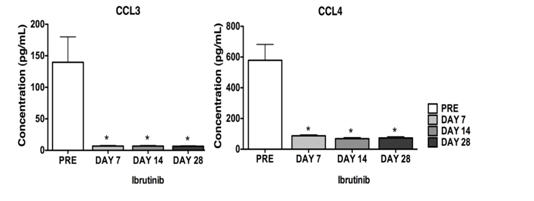
B cell receptor (BCR) signaling is essential for normal B cell development and plays an important role in several B cell malignancies, including chronic lymphocytic leukemia (CLL). BrutonÕs tyrosine kinase (Btk) transmits B cell receptor (BCR) signaling and can be inhibited by ibrutinib, a selective, covalent Btk inhibitor. Because of highly encouraging results with ibrutinib in high-risk CLL patients in the Phase 1/2 trial, we explored the combination of ibrutinib and rituximab in forty high-risk CLL patients, characterized by the presence of 17p deletion or TP53 mutation (treated or untreated), previously treated patients with 11q deletion, or patients with a short remission duration (< 3 years) after first-line chemo-immunotherapy. Here, we present early correlative studies from this trial, focusing on BCR-related CLL responses (chemokine secretion, viability) and CLL cell migration, based on our recent experience with ibrutinib in preclinical CLL models (Ponader S et al., Blood 119:1182-9, 2012). The aim of this study was to determine if preclinical results of Btk inhibition with ibrutinib can be recapitulated in vivo with specimen from the high-risk population enrolled on this trial. Plasma levels of CLL3 and CCL4 (MIP-1α/β), two chemokines secreted by CLL cells in response to BCR activation, were assessed before, at 14, and at 28 days on treatment with ibrutinib. In 28 analyzed patients, we demonstrate robust, significant reductions in CCL3 and CCL4 plasma concentrations after 14 and 28 days of treatment. As shown in Figure 1, plasma CCL3 levels were reduced from 139.6 (± 40.4) pg/mL before treatment to 6.9 (± 0.9) pg/mL or 6.5 (± 0.7) pg/mL after 14 and 28 days of treatment, respectively. We also evaluated in vitro secretion of CCL3 and CCL4 into supernatants of CLL cells isolated from 12 patients before and during ibrutinib therapy in response to stimulation with anti-IgM. Compared to CLL cells from pre-treatment specimen, CLL cells from patients on Ibrutinib therapy showed reduced levels of CLL3 and CCL4 secretion, and additional treatment with Ibrutinib (0.5 μM – 1μM) led to reduced chemokine levels only in pre-treatment samples, indicating complete Btk target inhibition. We next evaluated the effect of ibrutinib on CLL cell viability after anti-IgM stimulation. In pre-treatment samples, ibrutinib abrogated BCR-triggered CLL cell survival. Surprisingly, CLL cells from ibrutinib-treated patients remained anti-IgM responsive in these viability assays. These Btk-independent pro-survival effects could not be inhibited by in vitro treatment with ibrutinib, indicating that some of the anti-IgM-triggered pro-survival signaling can bypass Btk. Next, we analyzed migration of CLL cells towards the chemokines CXCL12 and CXCL13 in transwell chemotaxis assays. Pre-treatment samples displayed significant higher chemotaxis towards CXCL12 and CXCL13 when compared to CLL samples from patients on therapy with ibrutinib. The mean relative migration of such samples toward CXCL12 or CXCL13 was reduced to 28% (± 5%) or 35% (±15%) of respective CLL cells isolated before ibrutinib therapy (100%), n=6. Collectively, our results demonstrate that Ibrutinib blocks BCR-dependent survival and migration responses in high-risk CLL patients in vivo. They also corroborate the validity and robustness of CCL3 and CCL4 as biomarkers for BCR inhibition in CLL patients.
Figure 1
Disclosures: O'Brien: Pharmacyclics: Research support Other. Buggy: Pharmacyclics: Employment, Equity Ownership. Burger: Pharmacyclics: Consultancy, Research Funding.
