Attached files
| file | filename |
|---|---|
| EXCEL - IDEA: XBRL DOCUMENT - Oncternal Therapeutics, Inc. | Financial_Report.xls |
| EX-23.1 - EX-23.1 - Oncternal Therapeutics, Inc. | d305905dex231.htm |
| EX-31.1 - EX-31.1 - Oncternal Therapeutics, Inc. | d305905dex311.htm |
| EX-32.2 - EX-32.2 - Oncternal Therapeutics, Inc. | d305905dex322.htm |
| EX-31.2 - EX-31.2 - Oncternal Therapeutics, Inc. | d305905dex312.htm |
| EX-32.1 - EX-32.1 - Oncternal Therapeutics, Inc. | d305905dex321.htm |
| EX-10.61 - EX-10.61 - Oncternal Therapeutics, Inc. | d305905dex1061.htm |
| EX-10.60 - EX-10.60 - Oncternal Therapeutics, Inc. | d305905dex1060.htm |
Table of Contents
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
| x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2011
OR
| ¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission file number 000-50549
GTx, Inc.
(Exact name of registrant as specified in its charter)
| Delaware | 62-1715807 | |
| (State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer Identification No.) | |
| 175 Toyota Plaza 7th Floor Memphis, Tennessee |
38103 | |
| (Address of principal executive offices) | (Zip Code) | |
(901) 523-9700
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
| Title of Each Class |
Name of Each Exchange on Which Registered | |
| Common Stock, par value $0.001 per share | The NASDAQ Stock Market, LLC |
Securities registered pursuant to Section 12(g) of the Act:
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ¨ No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ¨ No x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No ¨
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes x No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. x
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act:
| Large accelerated filer | ¨ | Accelerated filer | x | |||
| Non-accelerated filer | ¨ (Do not check if a smaller reporting company) | Smaller reporting company | ¨ | |||
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ¨ No x
The aggregate market value of common stock held by non-affiliates of the registrant based on the closing sales price of the registrant’s common stock on June 30, 2011 as reported on The NASDAQ Global Market was $139,617,253.
There were 62,803,673 shares of registrant’s common stock issued and outstanding as of February 29, 2012.
Table of Contents
DOCUMENTS INCORPORATED BY REFERENCE
Certain portions of the registrant’s definitive proxy statement to be filed with the Securities and Exchange Commission pursuant to Regulation 14A, not later than 120 days after the end of the fiscal year covered by this Annual Report on Form 10-K, in connection with the Registrant’s 2012 Annual Meeting of Stockholders are incorporated by reference into Part III of this Annual Report on Form 10-K.
Table of Contents
Table of Contents
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K contains forward-looking statements. The forward-looking statements are contained principally in the sections entitled “Risk Factors,” “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and “Business.” These statements involve known and unknown risks, uncertainties and other factors that may cause our actual results, performance or achievements to be materially different from any future results, performances or achievements expressed or implied by the forward-looking statements. Forward-looking statements include statements about:
| • | the anticipated progress of our research, development and clinical programs, including whether our ongoing clinical trials of enobosarm (also known as OstarineTM or GTx-024) will achieve similar results to clinical trials that we have previously concluded; |
| • | the timing, scope and anticipated initiation, enrollment and completion of our ongoing clinical trials of enobosarm and any other future clinical trials that we may conduct; |
| • | whether and to what extent the U.S. Food and Drug Administration will allow us to further develop Capesaris® (GTx-758) in any indication; |
| • | the timing of regulatory submissions and the timing, scope and anticipated outcome of related regulatory actions; |
| • | our ability to establish and maintain potential new collaborative arrangements for the development and commercialization of our product candidates; |
| • | our ability to obtain and maintain regulatory approvals of our product candidates and any related restrictions, limitations, and/or warnings in the label of an approved product candidate; |
| • | our ability to market, commercialize and achieve market acceptance for our product candidates or products that we may develop; |
| • | our ability to generate additional product candidates for clinical testing; |
| • | our ability to protect our intellectual property and operate our business without infringing upon the intellectual property rights of others; and |
| • | our estimates regarding the sufficiency of our cash resources, expenses, capital requirements and needs for additional financing, and our ability to obtain additional financing. |
In some cases, you can identify forward-looking statements by terms such as “anticipates,” “believes,” “could,” “estimates,” “expects,” “intends,” “may,” “plans,” “potential,” “predicts,” “projects,” “should,” “will,” “would,” and similar expressions intended to identify forward-looking statements. Forward-looking statements reflect our current views with respect to future events, are based on assumptions, and are subject to risks, uncertainties and other important factors. We discuss many of these risks in this Annual Report on Form 10-K in greater detail in the section entitled “Risk Factors” under Part I, Item 1A below. Given these risks, uncertainties and other important factors, you should not place undue reliance on these forward-looking statements. Also, forward-looking statements represent our estimates and assumptions only as of the date of this Annual Report on Form 10-K. You should read this Annual Report on Form 10-K and the documents that we incorporate by reference in and have filed as exhibits to this Annual Report on Form 10-K, completely and with the understanding that our actual future results may be materially different from what we expect.
Except as required by law, we assume no obligation to update any forward-looking statements publicly, or to update the reasons actual results could differ materially from those anticipated in any forward-looking statements, even if new information becomes available in the future.
1
Table of Contents
| ITEM 1. | BUSINESS |
Overview
GTx, Inc., a Delaware corporation incorporated on September 24, 1997 and headquartered in Memphis, Tennessee, is a biopharmaceutical company dedicated to the discovery, development and commercialization of small molecules that selectively target hormone pathways for the treatment of cancer, cancer supportive care, and other serious medical conditions.
We are developing selective androgen receptor modulators, or SARMs, a new class of drugs with the potential to prevent and treat muscle wasting in patients with cancer and other musculoskeletal wasting or muscle loss conditions, including chronic sarcopenia (age related muscle loss). Our lead SARM product candidate, enobosarm (also known as OstarineTM or GTx-024), has to date been evaluated in eight completed clinical trials enrolling approximately 600 subjects, including in a Phase Ib and two Phase II efficacy studies. Enobosarm is the generic name given to the compound by the USAN Council and the World Health Organization and is the first compound to receive the SARM stem in its name from USAN, recognizing enobosarm as the first in this new class of compounds.
We held End of Phase II meetings with the U.S. Food and Drug Administration, or FDA, prior to initiating our Phase III clinical development of enobosarm for the prevention and treatment of muscle wasting in patients with non-small cell lung cancer, or NSCLC. Based upon data from our Phase II clinical trials and with feedback from the FDA, we designed the POWER 1 and POWER 2 (Prevention and treatment Of muscle Wasting in cancER) pivotal Phase III clinical trials evaluating enobosarm for this indication. We also met with representatives of the Medicines and Healthcare Products Regulatory Agency (United Kingdom) and Medical Products Agency (Sweden), who confirmed that the design of the POWER 1 and POWER 2 clinical trials should be sufficient for the European Medicines Agency to support registration in Europe. We are conducting each of these trials in clinical sites in the United States, Europe, and South America. In each of the placebo-controlled, double-blind clinical trials, approximately 300 patients with Stage III or IV non-small cell lung cancer will be randomized to placebo or enobosarm 3 mg at the time they are to begin first line chemotherapy. Enrollment in each of the pivotal Phase III clinical trials is ongoing. The trials are evaluating as co-primary endpoints the effect of enobosarm versus placebo on total lean body mass (muscle) assessed by dual x-ray absorptiometry, or DXA, and on physical function assessed by the Stair Climb Test at three months. Durability of effect is being assessed as a secondary endpoint at five months. We currently expect data from these pivotal Phase III clinical trials in the first quarter of 2013. We intend to continue our pursuit of a strategic partnership or collaboration for the development and commercialization of SARMs, including enobosarm.
On February 21, 2012, we announced that the FDA had placed a clinical hold on our clinical trials evaluating Capesaris® (GTx-758), an oral nonsteroidal selective estrogen receptor alpha agonist, or ER alpha agonist, for primary (first line) androgen deprivation therapy for advanced prostate cancer and secondary (second line) hormonal treatment. A clinical hold is a notification issued by the FDA to the trial sponsor to delay a clinical trial or suspend an ongoing clinical trial. The clinical hold, which was effective as of February 17, 2012, affects our Phase II maintenance dose finding clinical trial to evaluate Capesaris® 1000 mg and 2000 mg once-a-day doses compared to Lupron Depot® (leuprolide acetate for depot suspension) in 164 men with advanced prostate cancer, and our Phase II loading dose finding clinical trial evaluating a 1000 mg twice-a-day dose or 1500 mg twice-a-day dose to medically castrate men with advanced prostate cancer. Additionally, the clinical hold applies to our Phase II clinical trial evaluating Capesaris® 2000 mg once-a-day dose to determine the ability of Capesaris® to reduce serum prostate specific antigen, or PSA, and the duration of this PSA reduction in 25 men with castration resistant prostate cancer, or CRPC, who were currently receiving androgen deprivation therapy, or ADT. The clinical hold followed our reports to the FDA of venous thromboembolic events, or blood clots, in subjects treated with Capesaris® at the doses being studied in the trials (1000 mg and higher). There were two deaths in subjects treated with Capesaris® and two deaths in subjects treated with Lupron Depot®. As a result of the clinical hold, we suspended further enrollment into these three studies and notified clinical sites to discontinue treatment of subjects with Capesaris®. Subject to further input from the FDA, we believe there may be a path forward to develop Capesaris® at lower doses to treat men with metastatic hormone sensitive prostate cancer and men with CRPC. We plan to work with the FDA to determine the appropriate course of action, if any, to evaluate Capesaris® in these patient populations. There can be no assurance that the FDA will allow us to further develop Capesaris® in these or any other patient populations.
2
Table of Contents
We have an extensive preclinical pipeline generated from our own discovery program, including inhibitors of steroid biosynthetic enzymes, anticancer therapies (receptor tyrosine kinase and tubulin inhibitors), estrogen receptor beta agonists, and other novel compounds that are currently in preclinical development for the potential treatment of cancer, metabolic diseases, ophthalmic diseases, psoriasis and/or pain.
We sell FARESTON® (toremifene citrate) 60 mg tablets, approved for the treatment of advanced metastatic breast cancer in postmenopausal women, in the United States. In 2011, we discontinued our toremifene 80 mg and toremifene 20 mg development programs.
Scientific Background on Estrogen and Androgen Hormones and
Selective Hormone Receptor Modulators
Estrogens and androgens are hormones that play critical roles in regulating the reproductive system and contributing to the homeostasis of the muscular, skeletal, cardiovascular, metabolic and central nervous systems.
Testosterone, the predominant androgen, is important for masculine physical characteristics, such as muscle size and strength and bone strength, as well as for mental well-being. Male reproductive health is dependent on testosterone for sexual interest, fertility, erectile function and normal prostate function. Testosterone is converted into a more potent androgen, dihydrotestosterone, or DHT, which also stimulates sebaceous and hair glands and may cause unwanted effects like acne and hair loss. In aging men, there is a gradual decline in testosterone levels, which contributes to a loss of muscle mass and strength, erectile dysfunction, decreased sexual interest, depression and mood changes. Moreover, in men, testosterone is converted to estradiol, the primary estrogen in men and women. Estrogens improve bone quality and reduce the risk of skeletal fractures.
Estrogens and androgens perform their physiologic functions by binding to and activating their respective hormone receptors located in various tissues. Once a hormone binds with its receptor, this activates a series of cellular events that results in the hormone specific tissue effects.
Pharmaceuticals that target estrogen or androgen receptors have been used medically for over 50 years. The drugs that have been used to stimulate androgen receptors are either natural or synthetic hormones, known as steroids. Steroids are generally believed to activate hormone receptors in all tissue types in a non-selective manner resulting in not only beneficial effects but also in unwanted clinical effects. In men, the lack of selectivity of testosterone and its conversion to DHT may result in unwanted side effects, such as the potential stimulation of latent into clinical prostate cancer, worsening of benign prostatic hyperplasia, or BPH, development or worsening of acne and gynecomastia, or loss of hair in men. Hair growth, acne and masculinization are also of concern in women who are exposed to exogenous testosterone. To date, no orally available testosterone products have been approved for use in the United States. Those testosterone products that are available must be administered by intramuscular injections or by transdermal patches or gels that may not be convenient for patients and, in some cases, can result in inconsistent blood levels of testosterone.
There are also classes of small molecules that are not steroids that can bind to the same hormone receptors. These nonsteroidal small molecules may either stimulate or block hormone receptors depending on the type of tissue in which the receptor is found and the interaction of the small molecule with the receptor. A drug that has the ability to either block or stimulate the hormone receptor in this manner is called a selective hormone receptor modulator. A selective hormone receptor modulator may be able to mimic the beneficial, while minimizing the unwanted, effects of natural or synthetic steroid hormones.
A SARM is a small molecule that binds to and selectively modulates androgen receptors, the primary receptor to which testosterone binds. SARMs potentially have beneficial effects in muscle and bone while avoiding testosterone’s unwanted effects in the prostate in men or skin in men and women. Although no SARMs have been commercialized to date, we believe that SARMs, without the harmful side effects of testosterone or other exogenous anabolic steroid therapies, can potentially be developed to treat a range of medical conditions, including: (1) muscle loss conditions of chronic diseases, such as cancer, AIDS, chronic kidney disease, end-stage renal disease, and neurodegenerative disorders; (2) muscle loss of acute conditions such as trauma, burns, and rehabilitation; (3) muscle loss conditions associated with aging such as frailty and chronic sarcopenia; (4) the prevention and/or treatment of osteoporosis; (5) prostate disorders, such as BPH; (6) disorders of the central nervous system, such as low libido, depression and other mood disorders; (7) low testosterone conditions, such as primary and secondary hypogonadism; (8) disorders of male reproductive functions, such as infertility, male contraception and erectile dysfunction; and (9) other conditions, such as anemia and male hair loss.
3
Table of Contents
A selective estrogen receptor alpha agonist is a nonsteroidal compound with the ability to preferentially bind and activate estrogen receptor alpha as compared to estrogen receptor beta. Capesaris®, as a selective estrogen receptor alpha agonist, may have the ability to treat men with metastatic hormone sensitive prostate cancer and CRPC with lower incidences of hot flashes, insulin resistance, bone loss or other side effects related to luteinizing hormone releasing hormone, or LHRH, agonists and antagonists. As data from our recent clinical trials indicated that a greater risk of blood clots are associated with higher dosages of Capesaris®, we plan to discuss with the FDA whether there may be a path forward to develop Capesaris® at lower doses in these patient populations. There can be no assurance that the FDA will allow us to further develop Capesaris® in these or any other patient populations.
Commercial Product
FARESTON®
We sell FARESTON® (toremifene citrate) 60 mg tablets, approved for the treatment of advanced metastatic breast cancer in postmenopausal women in the United States. Toremifene is a selective estrogen receptor modulator, or SERM, owned and manufactured by Orion and is the active pharmaceutical ingredient in FARESTON®. On January 1, 2005, we entered into an amended and restated license and supply agreement with Orion to exclusively license toremifene for all indications in the United States and for all indications in humans except breast cancer outside of the United States.
We sell FARESTON® primarily through wholesale drug distributors. Our top three distributors, McKesson Corporation, Cardinal Health, Inc. and AmerisourceBergen Corporation, accounted for approximately 95% of our product sales generated from the sale of FARESTON® for the year ended December 31, 2011. The loss of any of these three distributors could have a material adverse effect on continued FARESTON® sales. FARESTON® net product sales accounted for 45%, 6%, and 22% of our total revenue for the years ended December 31, 2011, 2010 and 2009, respectively.
Product Candidates
The following table identifies the development phase and status for each of our clinical product candidates:
| Product Candidate/ Proposed Indication |
Program |
Clinical Development Phase |
Status | |||
| Enobosarm (OstarineTM; GTx-024) |
||||||
| Prevention and treatment of muscle wasting in patients with non-small cell lung cancer | SARM | Phase III | Enrolling the POWER 1 and POWER 2 pivotal Phase III clinical trials for the prevention and treatment of muscle wasting in patients with non-small cell lung cancer. | |||
| Capesaris® (GTx-758) | ||||||
| First and second line hormonal treatment of advanced prostate cancer1 |
Selective ER alpha agonist |
Phase II | Clinical development suspended as a result of a clinical hold placed on our three Capesaris clinical trials by the FDA in February 2012 for safety reasons. | |||
| 1 | We believe there may be a path forward to develop Capesaris® at lower doses to treat men with metastatic hormone sensitive prostate cancer and men with CRPC. We plan to work with the FDA to determine the appropriate course of action, if any, to evaluate Capesaris® in these or any other patient populations. |
4
Table of Contents
SARMs
SARMs are a new class of drugs with the potential to prevent and treat muscle wasting in patients with cancer and other musculoskeletal wasting or muscle loss conditions, including chronic sarcopenia (age related muscle loss).
Enobosarm for the Prevention and Treatment of Muscle Wasting in Patients with Cancer
Scientific Overview. Muscle wasting, a cancer related symptom, can begin early in the course of cancer and frequently leads to cancer cachexia, a complex metabolic condition characterized by accelerated loss of skeletal muscle and severe weight loss. Cancer cachexia is usually viewed as an end of life condition in patients with advanced or incurable malignancies. The common clinical symptoms attributed to muscle wasting include decline in physical function and impaired immune function which contribute to increased disability, fatigue, diminished quality of life, and reduced survival.
Although muscle wasting associated with cancer can be partially attributed to poor nutrition, treatment with appetite stimulants and nutritional intervention alone is not effective, likely because they do not address the underlying catabolic processes responsible for muscle wasting. Additionally, patients with severe weight loss, low performance status, and metastatic cancer that is no longer responding to cancer treatment may be less likely to respond to single therapies designed to increase muscle mass and physical function. Because muscle wasting, which often leads to refractory cancer cachexia, has a significant negative impact on the patient and their family, early prevention and treatment of muscle wasting are critical.
Enobosarm is an oral nonsteroidal SARM which means that it is similar to testosterone in activating androgen receptors in muscle, thereby promoting lean body mass (muscle) and improving physical function, while avoiding other effects which have been observed with testosterone such as stimulation of sebaceous glands, the cause of hair growth and acne, or epithelial cells of the prostate, which may exacerbate BPH or stimulate prostate cancer. Enobosarm, as a selective anabolic agent, may play an important role in the prevention and treatment of muscle wasting in cancer patients.
Potential Market. Lung cancer accounts for more deaths than any other cancer in both men and women. Worldwide, there are an estimated 1.5 million new cases and approximately 1.3 million deaths annually. In the United States, there are approximately 225,000 new cases and 160,000 deaths attributed to lung cancer each year. Approximately 85% of all newly diagnosed lung cancers are non-small cell lung cancer. Up to 50% of non-small cell lung cancer patients have severe muscle wasting at diagnosis with the majority developing severe wasting throughout the course of their disease. Lower body functional limitations, such as the inability to walk up or down steps without rest, or the inability to lift 10 pounds, are present in almost 90% of lung cancer survivors.
There are currently no drugs approved by the FDA for the prevention or treatment of muscle wasting in patients with NSCLC. Supplemental nutritional support alone has little or no benefit in counteracting muscle wasting in cancer patients. Although there are two commercially available anabolic steroids that can be prescribed off-label for the treatment of weight loss in cancer patients, chronic use of these drugs may result in liver toxicity or other adverse events and has limited their use. Megace® (megestrol acetate), an appetite stimulant, has also been used off-label for cancer patients, but has not been shown to increase lean body mass or improve physical function despite increasing appetite in some patients. Additionally, there are other companies developing drugs for the treatment of muscle wasting, appetite stimulation and cachexia. These compounds may compete with enobosarm if approved for commercial sale.
Clinical Trials. Enobosarm has been evaluated in eight completed clinical trials enrolling approximately 600 subjects, and is currently being evaluated in two pivotal Phase III clinical trials.
In May 2006, we initiated a Phase II proof of concept, double blind, randomized, dose finding placebo controlled clinical trial in 60 elderly men and 60 postmenopausal women. The trial was designed to evaluate enobosarm treatment in building lean muscle mass, as well as to assess safety in both elderly men and postmenopausal women. In December 2006, we reported the top line results. Without a prescribed diet or exercise regimen, all subjects treated with enobosarm had dose dependent increases in the primary endpoint of total lean body mass. Treatment with enobosarm also resulted in a dose dependent improvement in functional performance, a secondary endpoint, measured by stair climb. Enobosarm had a favorable safety profile, with no serious adverse events reported. Enobosarm also exhibited tissue selectivity with beneficial effects on lean body mass and performance and with no clinically relevant changes in measurements of serum PSA, sebum production, serum LH, or hair growth.
5
Table of Contents
In July 2007, we initiated a Phase IIb randomized, double blind, placebo controlled clinical trial evaluating enobosarm for the treatment of muscle wasting in 159 patients diagnosed with non-small cell lung cancer, colorectal cancer, non-Hodgkin’s lymphoma, chronic lymphocytic leukemia, or breast cancer. In October 2008, we announced top line results of this clinical trial. The study met its primary endpoint of absolute change in total lean body mass compared to placebo and the secondary endpoint of physical function, measured by stair climb, after 16 weeks of treatment. The Phase III clinical trials currently being conducted with enobosarm are based on the data in the subset of subjects in this Phase IIb study with non-small cell lung cancer.
In 2009, enobosarm was evaluated in a 12 week, randomized Phase Ib clinical trial comparing enobosarm to two doses of another SARM (Merck compound MK-3984) and to placebo in 88 postmenopausal women. Total lean body mass was measured at baseline and 12 weeks, and physical performance was evaluated at the same interval by bilateral leg press machine. After 12 weeks of treatment, enobosarm 3 mg significantly increased total lean body mass. Enobosarm treatment also resulted in increased leg muscle strength. Enobosarm treatment did not cause virilization, as there was no change in sebaceous gland volume, rate of sebum excretion, or hair follicle gene expression.
We held End of Phase II meetings with the FDA prior to initiating our Phase III clinical development of enobosarm for the prevention and treatment of muscle wasting in patients with NSCLC. Based upon data from our Phase II clinical trials and with feedback from the FDA, we designed the POWER 1 and POWER 2 (Prevention and treatment Of muscle Wasting in cancER) pivotal Phase III clinical trials evaluating enobosarm for this indication. We also met with representatives of the Medicines and Healthcare Products Regulatory Agency (United Kingdom) and Medical Products Agency (Sweden), who confirmed that the design of the POWER 1 and POWER 2 clinical trials should be sufficient for the European Medicines Agency to support registration in Europe. We have commenced each of these trials, which are being conducted in clinical sites in the United States, Europe, and South America. In each of the placebo-controlled, double-blind clinical trials, approximately 300 patients with Stage III or IV non-small cell lung cancer will be randomized to placebo or enobosarm 3 mg at the time they are to begin first line chemotherapy. Enrollment in each of the pivotal Phase III clinical trials is ongoing. The trials are evaluating as co-primary endpoints the effect of enobosarm versus placebo on total lean body mass (muscle) assessed by DXA and on physical function assessed by the Stair Climb Test at three months. Durability of effect is being assessed as a secondary endpoint at five months. We currently expect data from these pivotal Phase III clinical trials in the first quarter of 2013.
Selective ER Alpha Agonist
Capesaris® for the Treatment of Metastatic Hormone Sensitive Prostate Cancer and Castration Resistant Prostate Cancer
Scientific Overview. ADT is the most common treatment for patients who have advanced prostate cancer. ADT reduces testosterone, a primary growth factor for prostate cancer, to castrate levels. ADT is currently accomplished either surgically by removal of the testes, or chemically by injection with LHRH agonists or antagonists. These LHRH agents work by shutting off LH secretion by the pituitary gland thereby stopping testosterone production by the testes. The reduction in testosterone by ADT also results in very low estrogen levels in men, because estrogen is derived from testosterone in men. Estrogen deficiency side effects associated with LHRH therapies may include bone loss and fractures, adverse lipid changes, hot flashes, gynecomastia, decreased libido, impaired cognitive function, increase in body fat composition, metabolic syndrome, diabetes and cardiovascular disease.
We have been developing Capesaris®, an oral selective ER alpha agonist, for first and second line hormonal treatment of advanced prostate cancer. Capesaris® was designed to achieve medical castration in men with prostate cancer by feedback inhibition of the hypothalamic- pituitary-gonadal axis. Capesaris® causes the liver to secrete a protein, sex hormone binding globulin, which binds to testosterone circulating in the blood and reduces levels of free testosterone, which is the form of testosterone that is responsible for the stimulation of prostate cancer. Because of the mechanism of action of Capesaris®, we designed Phase II clinical studies to demonstrate that castration may be achieved and maintained with lower incidences of concomitant bone loss, insulin resistance, or hot flashes typically associated with LHRH agonists or antagonists.
6
Table of Contents
On February 21, 2012, we announced that the FDA had placed a clinical hold on our clinical trials to evaluate Capesaris® for first line androgen deprivation therapy for advanced prostate cancer and second line hormonal treatment. The clinical hold followed our reports to the FDA of venous thromboembolic events, or blood clots, in subjects treated with Capesaris® at doses studied (1000 mg and higher). There were two deaths in subjects treated with Capesaris® and two deaths in subjects treated with Lupron Depot®. As a result of the clinical hold, we suspended further enrollment into these ongoing studies and notified clinical sites to discontinue treatment of subjects with Capesaris®. Subject to further input from the FDA, we believe there may be a path forward to develop Capesaris® at lower doses to treat men with metastatic hormone sensitive prostate cancer and men with CRPC. There can be no assurances that the FDA will allow us to further develop Capesaris® in these or any other patient populations.
Potential Market. We plan to discuss with the FDA the potential development of Capesaris® as an oral secondary hormonal therapy for CRPC and as first line hormonal therapy for men with hormone sensitive metastatic prostate cancer. We believe there are approximately 80,000 men in the United States with CRPC who have not received chemotherapy. We believe there are approximately 36,000 men diagnosed each year with metastatic hormone sensitive prostate cancer.
There are no drugs currently approved for the treatment of CRPC in patients who have not yet received chemotherapy, although several drugs are in clinical development for this indication. We believe new hormonal therapies in development, if approved, will be used prior to chemotherapy as physicians and patients look for treatment options capable of delaying cancer progression and possibly prolonging survival prior to chemotherapy.
For metastatic hormone sensitive prostate cancer, ADT is the most common treatment. ADT reduces testosterone, a primary growth factor for prostate cancer, to castrate levels. ADT is currently accomplished either surgically by removal of the testes, or chemically by injection with LHRH agonists or antagonists. These LHRH agents work by shutting off LH secretion by the pituitary gland thereby stopping testosterone production by the testes. The reduction in testosterone by ADT also results in very low estradiol levels in men, because estradiol is derived from testosterone in men. Estrogen deficiency side effects associated with LHRH therapies may include bone loss and fractures, adverse lipid changes, hot flashes, gynecomastia, decreased libido, impaired cognitive function, increase in body fat composition, insulin resistance, metabolic syndrome, diabetes and cardiovascular disease. Capesaris® was designed to achieve medical castration with lower incidences of hot flashes, concomitant bone loss, or insulin resistance typically associated with LHRH therapies.
There are no approved androgen deprivation therapies designed to significantly minimize estrogen deficiency side effects, including bone loss, fractures, insulin resistance and hot flashes. For many men on ADT, physicians are currently prescribing certain drugs, some of them on an off-label basis, to help ameliorate some of the specific estrogen deficiency related side effects of ADT. These drugs include off-label use of bisphosphonates for osteoporosis and Megace® (megestrol acetate) and antidepressants for hot flashes.
In the event that the FDA determines we may continue the development of Capesaris®, the ability of Capesaris® to be an effective secondary hormonal therapy for men with CRPC or primary hormonal therapy for men with metastatic hormone sensitive prostate cancer will be dependent on our ability to find an appropriate dose that is both effective and safe for these patient populations.
Clinical Trials. In 2009, we evaluated Capesaris® in healthy male volunteers in two Phase I clinical trials, including a ten day multiple ascending dose study in 61 subjects where Capesaris® demonstrated the ability to increase serum SHBG and to reduce serum total and free testosterone. In September 2010, we announced that in a Phase II, open label, pharmacokinetic and pharmacodynamic clinical trial in young healthy male volunteers, Capesaris® suppressed serum total testosterone to castrate levels (levels of serum total testosterone less than 50ng/dL), increased serum SHBG, and reduced serum free testosterone, the form of testosterone which is available to prostate cancer cells for growth. The percentage of treatment compliant subjects receiving 1500 mg of Capesaris® who achieved medical castration was comparable to rates of castration observed with LHRH agonists or antagonists therapies. In May 2011, we completed a Phase I clinical trial of Capesaris® using a tablet formulation in older healthy male volunteers. In this trial, reductions in testosterone to medical castration levels, increases in SHBG and decreases in free testosterone were observed in Capesaris® doses given orally each day.
We designed two Phase II clinical trials to identify an appropriate dose of Capesaris® to achieve and maintain medical castration (less than 50ng/dL) from day 28 to day 364 in men with advanced prostate cancer. In June 2011, we initiated the Phase II maintenance dose finding clinical trial evaluating Capesaris® 1000 mg and 2000 mg once-a-day doses compared to Lupron Depot® (leuprolide acetate for depot suspension) in 164 men with advanced prostate cancer. We also initiated the Phase II loading dose finding clinical trial evaluating 1000 mg and 1500 mg doses twice-a-day to medically castrate men by day 28 in 104 men with advanced prostate cancer. After day 28, castrate patients were to continue treatment on one of two once-a-day doses of Capesaris®, 2000 mg or 1000 mg, until day
7
Table of Contents
360. We were also conducting a second line hormonal therapy Phase II clinical trial evaluating Capesaris® 2000 mg once-a-day dose in 25 men with CRPC. The objective of this trial was to determine the ability of Capesaris® to reduce serum PSA and the duration of this PSA reduction in men with CRPC who are currently receiving ADT. On February 21, 2012, we announced that the FDA had placed a clinical hold on these three clinical trials, effective February 17, 2012. The clinical hold followed our reports to the FDA of venous thromboembolic events, or blood clots, in subjects treated with Capesaris® at the doses being studied in the trials (1000 mg and higher). There were two deaths in subjects treated with Capesaris® and two deaths in subjects treated with Lupron Depot®. As a result of the clinical hold, we suspended further enrollment into these three studies and notified clinical sites to discontinue treatment of subjects with Capesaris®.
Subject to further input from the FDA, we believe there may be a path forward to develop Capesaris® at lower doses to treat men with metastatic hormone sensitive prostate cancer and men with CRPC. We plan to work with the FDA to determine the appropriate course of action to evaluate Capesaris® in these patient populations. There can be no assurance that the FDA will allow us to further develop Capesaris® in these or any other patient populations.
Drug Discovery and Other Research and Development
We have an extensive preclinical pipeline generated from our own discovery program, including inhibitors of steroid biosynthetic enzymes, anticancer therapies (receptor tyrosine kinase and tubulin inhibitors), estrogen receptor beta agonists, and other novel compounds that are currently in preclinical development for the potential treatment of cancer, metabolic diseases, ophthalmic diseases, psoriasis and/or pain.
We believe that our drug discovery expertise will allow us to sustain our clinical pipeline through the design and development of nonsteroidal small molecule drugs that selectively modulate hormone receptors, inhibit cancer growth, or treat inflammatory conditions. Our in-house medicinal chemists and scientists provide us with significant discovery and development expertise. Using our capabilities in hormone receptor biology, cancer pharmacology and medicinal chemistry, we are able to target many hormone receptors or other cellular targets and generate compounds that are designed to address important unmet medical needs.
We design and synthesize new compounds based on computer, or in silico, models and crystal structures of a molecular target’s binding sites. We continually modify and improve these models to reflect our study of the activity of new compounds in the laboratory, in which we determine the link between chemical structures and biological activity, or structure-activity relationships.
We also have significant medicinal scale-up and high throughput capabilities, which facilitate our rapid synthesis and evaluation of new compounds. Throughout our discovery process, we build diversity into our chemistry structures in order to improve our likelihood of success in developing novel compounds that have the potential to treat multiple indications. Through this approach, we have generated clinical product candidates that target the androgen receptor, such as enobosarm, and the estrogen receptor alpha, such as Capesaris®. We continue to conduct research and development efforts focused on other androgen receptor agonists and antagonists, ER alpha agents, ER beta agents, SARM compounds, other selective hormone receptor modulator compounds and anticancer agents.
Toremifene
In December 2008, we submitted a New Drug Application, or NDA, for toremifene 80 mg to reduce fractures in men with prostate cancer on ADT to the FDA. In October 2009, we received a Complete Response Letter from the FDA regarding our NDA for toremifene 80 mg notifying us that the FDA would not approve our NDA in its present form as a result of certain clinical deficiencies indentified in the Complete Response Letter, which deficiencies could only be addressed by conducting an additional pivotal Phase III clinical trial of toremifene 80 mg. In the second quarter of 2011, we evaluated the business case for toremifene 80 mg and concluded that we would discontinue our toremifene 80 mg development program.
In May 2010, we announced that toremifene 20 mg failed to meet the primary efficacy endpoint in a completed Phase III clinical trial evaluating toremifene 20 mg for the prevention of prostate cancer in high risk men with high grade prostatic intraepithelial neoplasia. During 2010, we determined to discontinue our toremifene 20 mg development program.
8
Table of Contents
Our Strategy
Our objective is to discover, develop and commercialize small molecules that selectively target hormone pathways for the treatment of cancer, cancer supportive care, and other serious medical conditions. Key elements of our strategy to achieve this objective are to:
Pursue Clinical Development and Commercialization of SARMs. Based upon data from our Phase II clinical trials and with feedback from the FDA, we have initiated two pivotal Phase III clinical trials evaluating enobosarm for the prevention and treatment of muscle wasting in patients with non-small cell lung cancer. We also intend to continue our pursuit of a strategic partnership or collaboration for the development and commercialization of SARMs, which includes enobosarm for the prevention and treatment of muscle wasting in patients with non-small cell lung cancer, as well as other indications.
Evaluate Potential Future Development of Capesaris®. Following the clinical hold placed on our three Phase II Capesaris® clinical trials for the first and second line hormonal treatment of advanced prostate cancer, we are evaluating the feasibility of potential future development of Capesaris®. Subject to further input from the FDA, we believe there may be a path forward to develop Capesaris® at lower doses to treat men with metastatic hormone sensitive prostate cancer and men with CRPC. We plan to work with the FDA to determine the appropriate course of action, if any, to evaluate Capesaris® in these patient populations. There can be no assurance that the FDA will allow us to further develop Capesaris® in these or any other patient populations.
Build Upon Our Other Drug Discovery Capabilities to Sustain Our Small Molecule Product Candidate Pipeline. While our clinical development efforts to date have focused on selective hormone receptor modulator technologies, we have the capability to discover and develop additional drug candidates for other important disease targets. We intend to develop new molecules to treat diseases that affect patients that are underserved by available alternatives or for which there are no current alternatives.
Licenses and Collaborative Relationships
In addition to our internally-developed and discovered small molecules, we have established and intend to continue to pursue licenses from and collaborative relationships with pharmaceutical companies and academic institutions to further the development and commercialization of our small molecule product candidates. Our most significant license and collaboration relationships are described below.
Orion Corporation
In March 2000, we entered into a license and supply agreement with Orion to develop and commercialize products containing toremifene. Our rights under the original license agreement were limited to specific disease fields pertaining to prostate cancer. In December 2004, we entered into an agreement with Orion to purchase specified FARESTON® related assets which Orion had re-acquired from another licensee. We also entered into an amended and restated license and supply agreement in January 2005. We paid Orion approximately $5.2 million under these agreements for the assets and related license rights.
Under the amended and restated license and supply agreement, we obtained an exclusive license from Orion to develop and commercialize toremifene-based products for all human indications worldwide, except breast cancer outside of the United States. We are required to pay Orion a portion of certain types of upfront and milestone income that we receive from third-party sublicensees, after we recover our clinical development costs. We are also required to pay Orion a royalty in the low-teens on sales by us, our affiliates and third-party sublicensees of toremifene-based products, including FARESTON®. In addition, we are required to pay up to $1.0 million if we are acquired before receiving marketing approval for the use of toremifene for the treatment or prevention of prostate cancer or related conditions, the development of which we have discontinued.
Our license and supply agreement with Orion requires that Orion will manufacture and supply all of our and our sublicensees’ needs for clinical trial and commercial grade material for toremifene-based products developed and marketed in the United States and abroad, including toremifene globally and FARESTON® in the United States, and that we purchase such products exclusively from Orion, unless and until Orion terminates its supply obligations in accordance with the terms of the agreement. Orion has the right to terminate its supply obligations at its election at any time, in which event we will have the right to enter into a contract manufacturing agreement with another
9
Table of Contents
supplier for toremifene-based products. However, any arrangements we make for an alternative supply would have to be made with a qualified alternative supplier with appropriate FDA approval in order for us to obtain our supply requirements for toremifene. The term of the amended and restated license and supply agreement lasts, on a country-by-country basis, until the later of expiration of our own patents claiming the manufacturing or the methods of use of toremifene for prostate cancer or the end of all marketing or regulatory exclusivity which we may obtain for toremifene-based products. Our method of use patents pertaining to toremifene for prostate cancer expires in 2022. Orion may terminate the amended and restated license and supply agreement, in its entirety or in some cases on a country-by-country basis, as a result of our uncured material breach or our bankruptcy.
University of Tennessee Research Foundation
In July 2007, we and the University of Tennessee Research Foundation, or UTRF, entered into a consolidated, amended and restated license agreement, or the SARM License Agreement, to consolidate and replace our two previously existing SARM license agreements with UTRF and to modify and expand certain rights and obligations of each of the parties under both license agreements. Pursuant to the SARM License Agreement, we were granted exclusive worldwide rights in all existing SARM technologies owned or controlled by UTRF, and certain improvements thereto, and exclusive rights to certain future SARM technology that may be developed by certain scientists at the University of Tennessee or subsequently licensed to UTRF under certain existing inter-institutional agreements with The Ohio State University. Unless terminated earlier, the term of the SARM License Agreement will continue, on a country-by-country basis, for the longer of 20 years or until the expiration of the last valid claim of any licensed patent in the particular country in which a licensed product is being sold. UTRF may terminate the SARM License Agreement for our uncured breach or upon our bankruptcy.
Under the SARM License Agreement, we paid UTRF a one-time, upfront fee of $290,000 as consideration for entering into the SARM License Agreement. We are also obligated to pay UTRF annual license maintenance fees, low single-digit royalties on net sales of products and mid single-digit royalties on sublicense revenues. During the year ended December 31, 2007, we paid UTRF a sublicense royalty of approximately $1.9 million as a result of our previous collaboration with Merck & Co., Inc. We also agreed to pay all expenses to file, prosecute and maintain the patents relating to the licensed SARM technologies, and are obligated to use commercially reasonable efforts to develop and commercialize products based on the licensed SARM technologies. In December 2008, we and UTRF amended the SARM License Agreement, or the SARM License Amendment, to, among other things, clarify the treatment of certain payments that we may receive from our current and future sublicensees for purposes of determining sublicense fees payable to UTRF, including the treatment of payments made to us in exchange for the sale of our securities in connection with sublicensing arrangements. In consideration for the execution of the SARM License Amendment, we paid UTRF $494,000.
Former Collaboration with Ipsen
In March 2011, we and Ipsen mutually agreed to terminate our former collaboration and license agreement, which was entered into in September 2006 and amended in March 2010. Under the former collaboration and license agreement with Ipsen, we granted Ipsen exclusive rights to develop and commercialize toremifene in all indications (except the treatment and prevention of breast cancer outside of the United States), initially in the European Union, Switzerland, Norway, Iceland, Lichtenstein, and the Commonwealth of Independent States, and subsequently expanded to include Australia and certain countries in North Africa, the Middle East and Asia (excluding Japan). In connection with the termination of the collaboration and license agreement, and in exchange for our reacquiring all of Ipsen’s rights under the former collaboration agreement, we agreed to pay Ipsen a low single digit royalty on net sales of toremifene 80 mg in the United States if approved for commercial sale. Following the termination of our collaboration and license agreement with Ipsen, we discontinued our toremifene development programs.
Manufacturing
We do not currently own or operate manufacturing facilities, and we rely, and expect to continue to rely, on third parties for the production of clinical and commercial quantities of our product candidates.
There are no complicated chemistries or unusual equipment required in the manufacturing process for enobosarm and our other SARM compounds. The active ingredient in enobosarm is manufactured using a five-step synthetic process that uses commercially available starting materials for each step. Enobosarm drug product is manufactured using conventional manufacturing technology without the use of novel excipients. We rely on third-party vendors for drug substance and drug product manufacturing.
10
Table of Contents
There are no complicated chemistries or unusual equipment required in the manufacturing process for Capesaris®. The active ingredient in Capesaris® is manufactured using a three-step synthetic process that uses commercially available starting materials for each step. Capesaris® drug product is manufactured using conventional manufacturing technology without the use of novel excipients. However, we continue to evaluate new processing and manufacturing technologies, and may use these new technologies in the manufacturing of Capesaris® drug product formulations in the future. We rely on third-party vendors for the manufacture of Capesaris® drug substance and drug product.
We have agreed to purchase from Orion our commercial requirements of FARESTON® tablets for at a specified price under an amended and restated license and supply agreement. Although the raw materials necessary to manufacture toremifene citrate tablets are readily available, we rely on Orion as our only supplier of FARESTON® tablets. Orion’s manufacturing facility complies with the FDA’s current Good Manufacturing Practice regulations. Orion may terminate its obligation to supply us at its election at any time. Under these circumstances, we would need to make arrangements for FARESTON® tablets to be made by a qualified alternative supplier with the appropriate FDA approval in order for us to obtain our supply requirements for FARESTON® tablets.
Competition
The biotechnology and biopharmaceutical industries are characterized by rapidly advancing technologies, intense competition and a strong emphasis on proprietary products. We face competition from many different sources, including commercial pharmaceutical and biotechnology enterprises, academic institutions, government agencies and private and public research institutions.
Many of our competitors have significantly greater financial resources and expertise in research and development, manufacturing, preclinical testing, conducting clinical trials, obtaining regulatory approvals and marketing approved products than we do. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. Our commercial opportunities will be reduced or eliminated if our competitors develop and commercialize similar products that are safer, more effective, have fewer side effects or are less expensive than any products that we and/or our collaborators may develop.
FARESTON® for the Treatment of Breast Cancer
There are a number of drugs that have been approved by the FDA for the treatment of breast cancer. Tamoxifen, which is marketed by several generic manufacturers, has been approved by the FDA for the treatment of advanced breast cancer and the reduction of breast cancer in women at high risk for developing the disease. Aromatase inhibitors, or AIs, such as anastrozole, letrozole and exemestane, are used to treat breast cancer in postmenopausal women. The AIs are growing at the expense of SERMs due to clinical trials such as the clinical trial entitled “Arimidex and Tamoxifen: Alone or in Combination” which has shown efficacy and tolerability advantages for AIs compared to tamoxifen.
SARMs for the Prevention and Treatment of Muscle Wasting in Patients with Cancer
There are currently no drugs approved by the FDA for the prevention and treatment of muscle wasting in patients with cancer. Although there are two commercially available steroids, nandrolone and oxandrolone, that are sometimes prescribed off-label for the treatment of weight loss in patients with chronic illnesses, including cancer, chronic use of these drugs may result in significant and serious adverse effects. For example, oxandrolone has a black box warning for liver toxicity as well as warnings and precautions related to increased risk for prostate cancer in men and virilization in women. Oxandrolone is indicated as an adjunctive therapy to promote weight gain after weight loss following extensive surgery, chronic infections and severe trauma, and in some patients who without pathophysiologic reasons fail to maintain normal weight and to offset the protein catabolism associated with prolonged administration of corticosteroids.
Testosterone products have been used off-label to treat secondary hypogonadism. Owing to their potentially unwanted effects and inconvenient dosing, we believe that testosterone products have had a limited impact on the market for muscle wasting. There are other SARM product candidates in development that may compete with our SARM product candidates if approved, including SARMs in development from Ligand Pharmaceuticals Inc., GlaxoSmithKline, Galapagos NV, and Bristol-Myers Squibb Company. Pfizer Inc., Eli Lilly & Co., and Amgen
11
Table of Contents
have previously reported myostatin inhibitors in development that may compete with enobosarm if approved for commercial sale. In addition, Cytokinetics, Inc. is developing a troponin activator with a muscle specific mechanism in Phase II studies with a focus on neurological muscle diseases (amyotrophic lateral sclerosis and myasthenia gravis). Moreover, there are other categories of drugs in development, including ghrelin receptor agonists, inflammatory modulators, and growth hormone secretagogues, that may have some muscle activity. A ghrelin receptor agonist is being evaluated by Helsinn Group for the treatment of cancer cachexia in patients with non-small cell lung cancer. Other appetite stimulants such as Megace® (megestrol acetate) and dronabinol are also used off-label for weight loss and loss of appetite in patients with cancer.
Capesaris® for the Treatment of Metastatic Hormone Sensitive Prostate Cancer and Castration Resistant Prostate Cancer
There are currently no drugs approved by the FDA for secondary hormonal therapy for men with CRPC. There are several hormonal therapy product candidates in development to treat these patients. MDV 3100 is being developed by Medivation, Inc. TAK-700 is being developed by Millennium: The Takeda Oncology Company to treat CRPC in men who have not yet received chemotherapy and in men who have failed chemotherapy. Zytiga® (abiraterone acetate) coadministered with prednisone is being marketed by Johnson & Johnson for the treatment of metastatic prostate cancer in men who have failed chemotherapy. Johnson & Johnson is also developing Zytiga® with prednisone for the treatment of men with CRPC who have not yet received chemotherapy.
For hormone sensitive metastatic prostate cancer, ADT is the most common treatment. ADT results in a castrate level of serum total testosterone by reducing LH secretion by the pituitary and is the primary treatment for advanced prostate cancer. Several major pharmaceutical companies market an ADT product and they have been very competitive in defending their business. Leuprolide (Lupron® marketed by Abbott Laboratories) and goserelin (Zoladex® marketed by Astra-Zeneca) are the leading ADT products and both are LHRH agonists. Manufacturers have patented new formulations (Eligard® marketed by Sanofi-Aventis and QLT, Inc.) or extended release formulations which have extended their brand’s life (Lupron® marketed by Abbott Laboratories). Histerlin an implant marketed as Vantas® (Endo Pharmaceuticals) is a one year LHRH agonist implant. Triptorelin marketed as Trelstar® (Watson Pharmaceuticals) and marketed as Decapeptyl® (Ipsen) is also a LHRH agonist. The most recently approved ADT product is Degarelix marketed as Firmagon® (Ferring Pharmaceuticals). Degarelix is a LHRH antagonist that avoids the testosterone flare seen with LHRH agonists. All existing ADT products are injectables and all can result in estrogen deficiency side effects (hot flashes, bone loss and fractures, increase in body fat composition, loss of libido, impaired cognition and adverse lipid changes). It is generally believed that all marketed ADT products have similar efficacy. There are no known late stage non-LHRH agonist and antagonist products in development for ADT.
Product Sales
We sell FARESTON® (toremifene citrate) 60 mg tablets, approved for the treatment of advanced metastatic breast cancer in postmenopausal women, in the United States. Effective June 1, 2011, we no longer utilize a sales force for FARESTON® promotional efforts, which has the potential to result in a decline in sales volume in future periods. In order to commercialize any future products, we will need to develop our sales and marketing capabilities or establish and maintain agreements with third parties to market and sell our product candidates.
Intellectual Property
We will be able to protect our technology from unauthorized use by third parties only to the extent it is covered by valid and enforceable patents or is effectively maintained as trade secrets. Patents and other proprietary rights are an essential element of our business.
For enobosarm and our other SARM compounds, we have an exclusive license from UTRF under its issued patents and pending patent applications in the United States, Canada, Australia, Japan, China and other countries in Asia, before the European Patent Office designating Germany, Great Britain, Spain, France, Italy, and other European Union countries, as well as in certain other countries outside those regions, covering the composition of matter of the active pharmaceutical ingredient for pharmaceutical products, pharmaceutical compositions and methods of synthesizing the active pharmaceutical ingredients. We have also exclusively licensed from UTRF issued and pending patent applications in the United States, Canada, Australia, Japan, China and other countries in Asia, before the European Patent Office designating Germany, Great Britain, Spain, France, Italy and other
12
Table of Contents
European Union countries, as well as in certain other countries outside those regions, related to methods for treating muscle wasting disorders, including cancer cachexia, and for treating sarcopenia and increasing muscle performance, muscle size and muscle strength and increasing the strength of or mass of a bone and for treating bone related disorders, including bone frailty and osteoporosis. The patents we licensed from UTRF and issued in the United States for enobosarm expire in 2024. Issued patents for our other SARM compounds in the United States will expire between 2021 and 2024, depending on the specific SARM compound. The patents we licensed from UTRF and issued outside of the United States for enobosarm expire in 2025, and with respect to other SARM compounds, expire in 2021 and 2023, depending on the specific SARM compound. We have pending patent applications for enobosarm and our other SARM compounds that, if issued, would expire in the United States and in countries outside the United States in 2025 and 2027, depending on the specific SARM compound. We have pending patent applications for SARMs in combination with anti-cancer agents that, if issued, would expire in the United States in 2024 and in countries outside the United States in 2028.
We have our own pending patent applications in the United States, Australia, Canada, before the European Patent office designating Germany, Great Britain, Spain, France, Italy and other European Union countries, Japan, and other jurisdictions internationally covering Capesaris® as the composition of matter of the active pharmaceutical ingredient for products developed with this compound and for pharmaceutical compositions and/or methods of treating advanced prostate cancer and treating bone loss, bone fractures, bone mineral density and osteoporosis in male subjects with prostate cancer having undergone androgen deprivation therapy. Patents issuing from our patent applications covering Capesaris® would, if issued, expire in 2028 or 2031 in the United States and in 2026 or 2031 in countries outside of the United States.
We cannot be certain that any of our patent applications, or those of our licensors, will result in issued patents. In addition, because the patent positions of biopharmaceutical companies are highly uncertain and involve complex legal and factual questions, the patents we own and license, or any further patents we may own or license, may not prevent other companies from developing similar or therapeutically equivalent products. Patents also will not protect our product candidates if competitors devise ways of making or using these product candidates without legally infringing our patents. In recent years, several companies have been extremely aggressive in challenging patents covering pharmaceutical products, and the challenges have often been successful. We cannot be assured that our patents will not be challenged by third parties or that we will be successful in any defense we undertake. Failure to successfully defend a patent challenge could materially and adversely affect our business.
In addition, changes in either patent laws or in interpretations of patent laws in the United States and other countries may materially diminish the value of our intellectual property or narrow the scope of our patent protection. For example, on September 16, 2011, the Leahy-Smith America Invents Act, or the Leahy-Smith Act, was signed into law. The Leahy-Smith Act includes a number of significant changes to United States patent law. These include provisions that affect the way patent applications will be prosecuted and may also affect patent defense and enforcement. The United States Patent Office is currently developing regulations and procedures to govern administration of the Leahy-Smith Act, and many of the substantive changes to patent law associated with the Leahy-Smith Act will not become effective until one year or 18 months after its enactment. Accordingly, it is too early to determine what effect or impact the Leahy-Smith Act will have on the operation of our business and the protection and enforcement of our intellectual property. However, the Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents, all of which could have a material adverse effect on our business and financial condition.
We also rely on trade secrets, technical know-how and continuing innovation to develop and maintain our competitive position. We seek to protect our proprietary information by requiring our employees, consultants, contractors, outside scientific collaborators and other advisors to execute non-disclosure and confidentiality agreements and our employees to execute assignment of invention agreements to us on commencement of their employment. Agreements with our employees also prevent them from bringing any proprietary rights of third parties to us. We also require confidentiality or material transfer agreements from third parties that receive our confidential data or materials.
13
Table of Contents
Government Regulation
New Drug Development and Approval Process
Numerous governmental authorities in the United States and other countries extensively regulate the testing, clinical development, manufacturing and marketing of pharmaceutical products and ongoing research and development activities. In the United States, the FDA rigorously reviews pharmaceutical products under the Federal Food, Drug, and Cosmetic Act and applicable regulations. Non-compliance with FDA regulations can result in administrative and judicial sanctions, including warning letters, clinical holds, fines, recall or seizure of products, injunctions, total or partial suspension of production, refusal of the government to approve marketing applications or allow entry into supply contracts, refusal to permit import or export of products, civil penalties, criminal prosecution and other actions affecting a company and its products. The FDA also has the authority to revoke previously granted marketing authorizations.
To secure FDA approval, an applicant must submit extensive preclinical and clinical data, as well as information about product manufacturing processes and facilities and other supporting information to the FDA for each indication to establish a product candidate’s safety and efficacy. The development and approval process takes many years, requires the expenditure of substantial resources and may be subject to delays or limitations of approval or rejection of an applicant’s new drug application. Even if the FDA approves a product, the approval is subject to post-marketing surveillance, adverse drug experience and other recordkeeping and reporting obligations, and may involve ongoing requirements for post-marketing studies. The FDA also recently obtained authority to place conditions on any approvals that could restrict the commercial applications, advertising, promotion or distribution of these products. Product approvals may be withdrawn if compliance with regulatory standards is not maintained or if problems occur following initial marketing.
Preclinical and Clinical Testing
Preclinical studies involve laboratory evaluation of product characteristics and animal studies to assess the biological activity and safety of the product. In some cases, long-term preclinical studies are conducted while clinical studies are ongoing. The FDA, under its Good Laboratory Practices regulations, regulates preclinical studies. Violations of these regulations can, in some cases, lead to invalidation of the studies, requiring these studies to be replicated. When the preclinical testing is considered adequate by the sponsor to demonstrate the safety and scientific rationale for initial human studies, the results of the preclinical tests, together with manufacturing information and analytical data, are submitted to the FDA as part of an Investigational New Drug application, or IND. The IND becomes effective, if not rejected by the FDA, within 30 days after the FDA receives the IND. The FDA may, either during the 30-day period after filing of an IND or at any future time, impose a clinical hold on proposed or ongoing clinical trials on various grounds, including that the study subjects are or would be exposed to an unreasonable and significant health risk. If the FDA imposes a clinical hold, clinical trials cannot commence or recommence without FDA authorization and then only under terms authorized by the FDA.
Clinical trials involve the administration of the investigational product candidates to humans under the supervision of a qualified principal investigator. Clinical trials must be conducted in accordance with Good Clinical Practices under protocols submitted to the FDA as part of the IND. In addition, each clinical trial must be approved and conducted under the auspices of an Investigational Review Board, or IRB, and with patient informed consent. The IRB typically considers, among other things, ethical factors and the safety of human subjects.
Clinical trials are conducted in three sequential phases, but the phases may overlap. Phase I clinical trials usually involve healthy human subjects. The goal of a Phase I clinical trial is to establish initial data about the safety, tolerability and pharmacokinetic properties of the product candidates in humans. In Phase II clinical trials, controlled studies are conducted on an expanded population of patients with the targeted disease. The primary purpose of these tests is to evaluate the effectiveness of the drug candidate on the patients to determine if there are any side effects or other risks associated with the drug and to determine the optimal dose of the drug from the safety and efficacy profile developed from the clinical study. Phase III trials involve even larger patient populations, often with several hundred or even several thousand patients, depending on the use for which the drug is being studied. Phase III trials are intended to establish the overall risk-benefit ratio of the drug and provide, if appropriate, an adequate basis for product labeling. During all clinical trials, physicians monitor the patients to determine effectiveness and to observe and report any reactions or other safety risks that may result from use of the drug candidate.
14
Table of Contents
Product Formulation and Manufacture
Concurrent with clinical trials and preclinical studies, companies must develop information about the chemistry and physical characteristics of the drug and finalize a process for manufacturing the product. In addition, manufacturers, including contract manufacturers, are required to comply with current applicable FDA Good Manufacturing Practice regulations. The current Good Manufacturing Practice regulations include requirements relating to quality control and quality assurance, as well as the corresponding maintenance of records and documentation. The manufacturing process must be capable of consistently producing quality batches of the product and the manufacturer must develop methods for testing the quality, purity and potency of the final drugs. Additionally, appropriate packaging must be selected and tested and chemistry stability studies must be conducted to demonstrate that the product does not undergo unacceptable deterioration over its shelf-life.
Compliance with current Good Manufacturing Practice regulations also is a condition of new drug application approval. The FDA must approve manufacturing facilities before they can be used in the commercial manufacture of drug products. In addition, manufacturing establishments are subject to pre-approval inspections and unannounced periodic inspections.
New Drug Application Process
After the completion of the clinical trial phases of development, if the sponsor concludes that there is substantial evidence that the drug candidate is safe and effective for its intended use, the sponsor may submit a NDA to the FDA. The application must contain all of the information on the drug candidate gathered to that date, including data from the clinical trials, and be accompanied by a user fee.
Under the Prescription Drug User Fee Act, or PDUFA, submission of a NDA with clinical data requires payment of a fee, with some exceptions. In return, the FDA assigns a goal of six or ten months from filing of the application to return of a first “complete response,” in which the FDA may approve the product or request additional information. There can be no assurance that an application will be approved within the performance goal timeframe established under PDUFA. The FDA initially determines whether a NDA as submitted is acceptable for filing. The FDA may refuse to file an application, in which case the FDA retains one-half of the user fees. If the submission is accepted for filing, the FDA begins an in-depth review of the application. As part of this review, the FDA may refer the application to an appropriate advisory committee, typically a panel of clinicians, for review, evaluation and a recommendation. The FDA is not bound by the recommendation of an advisory committee.
If the FDA evaluations of the NDA and the manufacturing facilities are favorable, the FDA may issue an approval letter authorizing commercial marketing of the drug candidate for specified indications. The FDA could also issue a “complete response” letter at the end of the review period. A “complete response” letter will be issued to let a company know that the review period for a drug is complete and that the application is not yet ready for approval. The letter will describe specific deficiencies and, when possible, will outline recommended actions the applicant might take to get the application ready for approval.
Marketing Approval and Post-Marketing Obligations
If the FDA approves an application, the drug becomes available for physicians to prescribe. Periodic reports must be submitted to the FDA, including descriptions of any adverse reactions reported. The FDA may require post-marketing studies, also known as Phase IV studies, as a condition of approval. In addition to studies required by the FDA after approval, trials and studies are often conducted to explore new indications for the drug. The purpose of these trials and studies and related publications is to develop data to support additional indications for the drug, which must be approved by the FDA, and to increase its acceptance in the medical community. In addition, some post-marketing studies are done at the request of the FDA to develop additional information regarding the safety of a product.
In accordance with authority gained pursuant to the Food and Drug Administration Amendments Act of 2007, or FDAAA, the FDA may impose risk evaluation mitigation strategies, or REMS, on a product if the FDA believes there is a reason to monitor the safety of the drug in the marketplace. REMS are a regulatory tool for the FDA that became effective in March 2008, and the agency applies this tool on a case-by-case assessment as to whether a REMS is needed. Since the effective date, the FDA has not used its REMS enforcement authority for every product approval, but it has exercised this authority on a regular basis, and it is anticipated the agency will continue to do so going forward. REMS could add training requirements for healthcare professionals, safety communications efforts, and limits on channels of distribution, among other things. The sponsor would be required to evaluate and monitor the various REMS activities and adjust them if need be. Whether a REMS would be imposed on a product and any resulting financial impact is uncertain at this time.
15
Table of Contents
Any products manufactured or distributed pursuant to FDA approvals are subject to continuing regulation by the FDA, including record keeping requirements, reporting of adverse experiences with the drug, drug sampling and distribution requirements, notifying the FDA and gaining its approval of certain manufacturing or labeling changes, complying with certain electronic records and signature requirements, and complying with FDA promotion and advertising requirements. Drug manufacturers and their subcontractors are required to register their establishments and are subject to periodic unannounced inspections for compliance with Good Manufacturing Practice requirements. Also, newly discovered or developed safety or effectiveness data may require changes to a product’s approved labeling, including the addition of new warnings and contraindications, or even in some instances revocation or withdrawal of the product’s approval.
Drug Price Competition and Patent Term Restoration Act of 1984
Under the Drug Price Competition and Patent Term Restoration Act of 1984, known as the Hatch-Waxman Act, a portion of a product’s patent term that was lost during clinical development and application review by the FDA may be restored. The Hatch-Waxman Act also provides for a statutory protection, known as exclusivity, against the FDA’s acceptance or approval of certain competitor applications. The Hatch-Waxman Act also provides the legal basis for the approval of abbreviated new drug applications, or ANDAs.
Patent term extension can compensate for time lost during product development and the regulatory review process by returning up to five years of patent life for a patent that covers a new product or its use. This period is generally one-half the time between the effective date of an IND and the submission date of a NDA, plus the time between the submission date of a NDA and the approval of that application. Patent term extensions, however, are subject to a maximum extension of five years, and the patent term extension cannot extend the remaining term of a patent beyond a total of 14 years. The application for patent term extension is subject to approval by the United States Patent and Trademark Office in conjunction with the FDA. It generally takes at least six months to obtain approval of the application for patent term extension.
The Hatch-Waxman Act also provides for a period of statutory protection for new drugs that receive NDA approval from the FDA. If a new drug receives NDA approval as a new chemical entity, meaning that the FDA has not previously approved any other new drug containing the same active entity, then the Hatch-Waxman Act prohibits an ANDA or a NDA submitted pursuant to section 505(b)(2) of the Federal Food, Drug, and Cosmetics Act, where the applicant does not own or have a legal right of reference to all of the data required for approval to be submitted by another company for a generic version of such drug (505(b)(2) NDA), with some exceptions, for a period of five years from the date of approval of the NDA. The statutory protection provided pursuant to the Hatch-Waxman Act will not prevent the filing or approval of a full NDA, as opposed to an ANDA or 505(b)(2) NDA, for any drug, including, for example, a drug with the same active ingredient, dosage form, route of administration, strength and conditions of use. In order to obtain a NDA, however, a competitor would be required to conduct its own clinical trials, and any use of the drug for which marketing approval is sought could not violate another NDA holder’s patent claims.
If NDA approval is received for a new drug containing an active ingredient that was previously approved by the FDA but the NDA is for a drug that includes an innovation over the previously approved drug, for example, a NDA approval for a new indication or formulation of the drug with the same active ingredient, and if such NDA approval was dependent upon the submission to the FDA of new clinical investigations, other than bioavailability studies, then the Hatch-Waxman Act prohibits the FDA from making effective the approval of an ANDA or 505(b)(2) NDA for a generic version of such drug for a period of three years from the date of the NDA approval. This three year exclusivity, however, only covers the innovation associated with the NDA to which it attaches. Thus, the three year exclusivity does not prohibit the FDA, with limited exceptions, from approving ANDAs or 505(b)(2) NDAs for drugs containing the same active ingredient but without the new innovation.
While the Hatch-Waxman Act provides certain patent restoration and exclusivity protections to innovator drug manufacturers, it also permits the FDA to approve ANDAs for generic versions of their drugs assuming the approval would not violate another NDA holder’s patent claims. The ANDA process permits competitor companies to obtain marketing approval for a drug with the same active ingredient for the same uses but does not require the conduct and submission of clinical studies demonstrating safety and effectiveness for that product. Instead of safety and effectiveness data, an ANDA applicant needs only to submit data demonstrating that its product is bioequivalent to the innovator product as well as relevant chemistry, manufacturing and product data. The Hatch-Waxman Act also
16
Table of Contents
instituted a third type of drug application that requires the same information as a NDA, including full reports of clinical and preclinical studies, except that some of the information from the reports required for marketing approval comes from studies which the applicant does not own or have a legal right of reference. This type of application, a 505(b)(2) NDA, permits a manufacturer to obtain marketing approval for a drug without needing to conduct or obtain a right of reference for all of the required studies.
If a competitor submits an ANDA or 505(b)(2) NDA for a compound or use of any compound covered by another NDA holder’s patent claims, the Hatch-Waxman Act requires, in some circumstances, the applicant to notify the patent owner and the holder of the approved NDA of the factual and legal basis of the applicant’s opinion that the patent is not valid or will not be infringed. Upon receipt of this notice, the patent owner and the NDA holder have 45 days to bring a patent infringement suit in federal district court and obtain a 30-month stay against the company seeking to reference the NDA. The NDA holder could still file a patent suit after the 45 days, but if they miss the 45-day deadline, they would not have the benefit of the 30-month stay. Alternatively, after this 45-day period, the applicant may file a declaratory judgment action, seeking a determination that the patent is invalid or will not be infringed. Depending on the circumstances, however, the applicant may not be able to demonstrate a controversy sufficient to confer jurisdiction on the court. The discovery, trial and appeals process in such suits can take several years. If such a suit is commenced, the Hatch-Waxman Act provides a 30-month stay on the approval of the competitor’s ANDA or 505(b)(2) NDA. If the litigation is resolved in favor of the competitor or the challenged patent expires during the 30-month period, unless otherwise extended by court order, the stay is lifted and the FDA may approve the application. Under regulations recently issued by the FDA, and essentially codified under the recent Medicare prescription drug legislation, the patent owner and the NDA holder have the opportunity to trigger only a single 30-month stay per ANDA or 505(b)(2) NDA. Once the applicant of the ANDA or 505(b)(2) NDA has notified the patent owner and the NDA holder of the infringement, the applicant cannot be subjected to another 30-month stay, even if the applicant becomes aware of additional patents that may be infringed by its product.
Pharmaceutical Pricing and Reimbursement
In both domestic and foreign markets, sales of any products for which we receive or have received regulatory approval for commercial sale will depend in part on the availability of reimbursement from third-party payors. Third-party payors include government health administrative authorities, managed care providers, private health insurers and other organizations. These third-party payors are increasingly challenging the price and examining the cost-effectiveness of medical products and services. In addition, significant uncertainty exists as to the reimbursement status of newly approved healthcare product candidates. We may need to conduct expensive pharmacoeconomic studies in order to demonstrate the cost-effectiveness of our products. Our product candidates may not be considered cost-effective. Adequate third-party reimbursement may not be available to enable us to maintain price levels sufficient to realize an appropriate return on our investment in product development.
The marketability of any products for which we receive or have received regulatory approval for commercial sale may suffer if the government and third-party payors fail to provide adequate coverage and reimbursement. In addition, an increasing emphasis on managed care in the United States has and will continue to increase the pressure on pharmaceutical pricing. Currently, our only marketed product, FARESTON® for the treatment of advanced metastatic breast cancer, is eligible for coverage and reimbursement by most third-party payors.
With respect to government payors, FARESTON® is generally covered under Medicaid, Medicare Part D and the Public Health Service (PHS) pharmaceutical pricing program as well as available for purchase by federal agencies. Medicaid is a joint federal and state program that is administered by the states for low income and disabled beneficiaries. Under the Medicaid Drug Rebate Program, we are required to pay a rebate for each unit of product reimbursed by the state Medicaid programs. The amount of the rebate for each product is set by law and may be subject to an additional discount if certain pricing increases more than inflation.
Medicare is a federal program that is administered by the federal government that covers individuals age 65 and over as well as those with certain disabilities. FARESTON® is a drug within the scope of Medicare Part D. Medicare Part D provides coverage to enrolled Medicare patients for self-administered drugs (i.e., drugs that do not need to be injected or otherwise administered by a physician). Medicare Part D is administered by private prescription drug plans approved by the U.S. government and each drug plan establishes its own Medicare Part D formulary for prescription drug coverage and pricing, which the drug plan may modify from time-to-time. The prescription drug plans negotiate pricing with manufacturers and may condition formulary placement on the availability of manufacturer discounts. Effective January 1, 2011, manufacturers, including us, were required to provide a 50% discount on brand name prescription drugs utilized by Medicare Part D beneficiaries when those beneficiaries reach the coverage gap in their drug benefits.
17
Table of Contents
Drug products like FARESTON® are subject to discounted pricing when purchased by federal agencies via the Federal Supply Schedule (FSS). FFS participation is required for our product to be covered and reimbursed by certain federal agencies and for coverage under Medicaid and the PHS pharmaceutical pricing program. FSS pricing is negotiated periodically with the Department of Veterans Affairs. FSS pricing is intended not to exceed the price that we charge our most-favored non-federal customer for our product. In addition, prices for drugs purchased by the Veterans Administration, Department of Defense (including drugs purchased by military personnel and dependents through the TRICARE retail pharmacy program), Coast Guard, and PHS are subject to a cap on pricing (known as the “federal ceiling price”) and may be subject to an additional discount if pricing increases more than inflation.
To maintain coverage of FARESTON® under the Medicaid Drug Rebate Program, we are required to extend discounts to certain purchasers under the PHS pharmaceutical pricing program. Purchasers eligible for discounts include hospitals that serve a disproportionate share of financially needy patients, community health clinics and other entities that receive health services grants from the PHS.
The United States and state governments continue to propose and pass legislation designed to reduce the cost of healthcare. In March 2010, the United States Congress enacted the Patient Protection and Affordable Care Act and the Health Care and Education Reconciliation Act which includes changes to the coverage and reimbursement of drug products under government health care programs. The changes, implemented in 2010 and thereafter, include the following: (1) increasing drug rebates under state Medicaid programs for brand name prescription drugs and extending those rebates to Medicaid managed care; (2) extending discounted rates on drug products available under the PHS pharmaceutical pricing program to additional hospitals and other providers; (3) assessing a fee on manufacturers and importers of brand name prescription drugs reimbursed under certain government programs, including Medicare and Medicaid; and (4) requiring drug manufacturers to provide a 50% discount on brand name prescription drugs sold to Medicare beneficiaries whose prescription drug costs cause the beneficiaries to be subject to the Medicare Part D coverage gap (i.e., the so-called “donut hole”). Adoption of other new legislation at the federal or state level could further limit reimbursement for pharmaceuticals.
Regulations Pertaining to Sales and Marketing
We are subject to various federal and state laws pertaining to health care “fraud and abuse,” including anti-kickback laws and false claims laws. Anti-kickback laws generally prohibit a prescription drug manufacturer from soliciting, offering, receiving, or paying any remuneration to generate business, including the purchase or prescription of a particular drug. Although the specific provisions of these laws vary, their scope is generally broad and there may not be regulations, guidance or court decisions that apply the laws to particular industry practices. There is therefore a possibility that our practices might be challenged under such anti-kickback laws. False claims laws prohibit anyone from knowingly and willingly presenting, or causing to be presented, any claims for payment for reimbursed drugs or services to third party payors (including Medicare and Medicaid) that are false or fraudulent, for items or services not provided as claimed, or for medically unnecessary items or services. Our activities relating to the sale and marketing of our products may be subject to scrutiny under these laws. Violations of fraud and abuse laws may be punishable by criminal or civil sanctions, including fines and civil monetary penalties, and/or exclusion from federal health care programs (including Medicare and Medicaid).
Laws and regulations have been enacted by the federal government and various states to regulate the sales and marketing practices of pharmaceutical manufacturers. The laws and regulations generally limit financial interactions between manufacturers and health care providers and/or require disclosure to the government and public of such interactions. The laws include federal “sunshine” provisions enacted in 2010 as part of the comprehensive federal health care reform legislation. Many of these laws and regulations contain ambiguous requirements. Given the lack of clarity in laws and their implementation, our reporting actions could be subject to the penalty provisions of the pertinent federal and state laws and regulations.
Research and Development
Since our inception in 1997, we have been focused on drug discovery and development programs. Research and development expenses include, but are not limited to, our expenses for personnel associated with our research activities, screening and identification of product candidates, formulation and synthesis activities, manufacturing, preclinical studies, toxicology studies, clinical trials, regulatory and medical affairs activities, quality assurance activities and license fees. Our research and development expenses were $31.9 million for the year ended December 31, 2011, $28.5 million for the year ended December 31, 2010, and $32.3 million for the year ended December 31, 2009.
18
Table of Contents
Employees
As of December 31, 2011, we had 90 employees, 28 of whom were M.D.s and/or Ph.D.s. None of our employees are subject to a collective bargaining agreement. We believe that we have good relations with our employees.
Available Information
We file electronically with the U.S. Securities and Exchange Commission, or SEC, our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Securities Exchange Act of 1934. We make available on our Web site at www.gtxinc.com, free of charge, copies of these reports as soon as reasonably practicable after we electronically file such material with, or furnish it to, the SEC. Further, copies of these reports are located at the SEC’s Public Reference Room at 100 F Street, NE, Washington, D.C. 20549. Information on the operation of the Public Reference Room can be obtained by calling the SEC at 1-800-SEC-0330. The SEC maintains a Web site that contains reports, proxy statements, and other information regarding our filings at www.sec.gov. The information provided on our website is not part of this report, and is therefore not incorporated by reference unless such information is otherwise specifically referenced elsewhere in this report.
Management
The following table sets forth information about our executive officers and other key medical, clinical and regulatory officers as of February 29, 2012.
| Name |
Age | Position(s) | ||
| Executive Officers |
||||
| Mitchell S. Steiner, M.D., F.A.C.S |
51 | Chief Executive Officer and Vice Chairman of the Board of Directors | ||
| Marc S. Hanover |
49 | President and Chief Operating Officer | ||
| James T. Dalton, Ph.D |
49 | Vice President, Chief Scientific Officer | ||
| Ronald A. Morton, Jr., M.D., F.A.C.S |
53 | Vice President, Chief Medical Officer | ||
| Henry P. Doggrell |
63 | Vice President, General Counsel and Secretary | ||
| Mark E. Mosteller |
49 | Vice President, Chief Financial Officer | ||
| Other Key Medical, Clinical and Regulatory Officers |
||||
| K. Gary Barnette, Ph.D |
44 | Vice President, Clinical Research and Development Strategy | ||
| Shontelle Dodson, Pharm. D |
41 | Vice President, Medical Affairs | ||
| Jeffrey G. Hesselberg |
53 | Vice President, Regulatory Affairs | ||
| Domingo Rodriguez, M.D |
50 | Vice President, Clinical Operations | ||
Executive Officers of the Registrant
Mitchell S. Steiner, M.D., F.A.C.S., a co-founder of GTx, has served as our Chief Executive Officer and Vice Chairman of our Board of Directors since our inception in September 1997. From 1995 to 2003, Dr. Steiner held numerous academic appointments, including Chairman and Professor of Urology, Director of Urologic Oncology and Research and the Chair of Excellence in Urologic Oncology at the University of Tennessee. Since 2003, Dr. Steiner has continued to serve on the faculty at the University of Tennessee. Dr. Steiner holds a B.A. in Molecular Biology from Vanderbilt University and an M.D. from the University of Tennessee, and performed his surgery and urologic training at The Johns Hopkins Hospital.
Marc S. Hanover, a co-founder of GTx, has served as our President and Chief Operating Officer since our inception in September 1997. Mr. Hanover also served as a member of our Board of Directors until August 2011. Prior to joining GTx, Mr. Hanover was a founder of Equity Partners International, Inc., a private equity firm in Memphis, Tennessee, and participated as a founder and investor in three healthcare companies. From 1985 to 1997, Mr. Hanover was a Senior Vice President and a member of the Executive Management Committee of National Bank of Commerce in Memphis, Tennessee. Mr. Hanover holds a B.S. in Biology from the University of Memphis and a MBA in Finance from the University of Memphis.
19
Table of Contents
James T. Dalton, Ph.D., was appointed Vice President, Chief Scientific Officer on January 1, 2011, and prior to that he served as Vice President, Preclinical Research and Development since January 2005. Dr. Dalton served as a scientific consultant to GTx from 1999 to 2005. Prior to joining GTx, Dr. Dalton held several academic appointments including Assistant and Associate Professor of Pharmaceutical Sciences in the College of Pharmacy at the University of Tennessee, Memphis (1992-2000) and Professor in the Division of Pharmaceutics, College of Pharmacy at The Ohio State University (2000-2007). SARMs were first discovered in Dr. Dalton’s research laboratories, and he is co-inventor on all SARM patents. Dr. Dalton holds a B.S. in Pharmacy from the University of Cincinnati and a Ph.D. in Pharmaceutics and Pharmaceutical Chemistry from The Ohio State University.
Ronald A. Morton, Jr., M.D., F.A.C.S., was appointed Vice President and Chief Medical Officer in April 2007. He joined GTx from the University of Medicine & Dentistry of New Jersey Robert Wood Johnson Medical School, where he served as Professor of Surgery, Chief of Urology and Director of Urologic Oncology for the Cancer Institute of New Jersey from January 2004 until April 2007. Dr. Morton also held the Conzen Chair for Clinical Research and was the Director of the New Jersey Center for Clinical and Translational Sciences. Prior to joining Robert Wood Johnson Medical School in 2004, Dr. Morton held a dual faculty appointment at the Baylor College of Medicine in the Scott Department of Urology and in the Department of Molecular and Cell Biology (May 1994 to December 2003), was Clinical Director of the Baylor Adult Urology Program (July 2000 to December 2003), Chief of Urology at the Houston Veterans Administration Medical Center (January 1999 to December 2003), and Director of the Baylor Prostate Cancer Center Research Laboratories (July 1996 to December 2003). He received his bachelor and medical degrees from The Johns Hopkins University and completed his urology training and postdoctoral fellowship and was an AFUD Scholar at The Johns Hopkins Brady Urological Institute.
Henry P. Doggrell has served as our General Counsel and Secretary since October 2001 and was appointed Vice President on January 20, 2005. From April 1998 to August 2001, Mr. Doggrell was Senior Vice President, Corporate Affairs at Buckeye Technologies, Inc., a specialty cellulose company, where he was responsible for matters including corporate finance, investor relations, mergers and acquisitions, intellectual property and licensing and strategic development. From 1996 to 1998, Mr. Doggrell served as General Counsel and Secretary of Buckeye Technologies. Prior to joining Buckeye Technologies, Mr. Doggrell was a partner of the Baker, Donelson, Bearman, Caldwell and Berkowitz law firm from 1988 to 1996, where he served as a member of the law firm management committee and Chair of the firm’s Corporate Securities department. Mr. Doggrell holds a B.S. in Commerce from the University of Virginia and a JD from Vanderbilt University.
Mark E. Mosteller has served as our Chief Financial Officer since August 2001 and was appointed Vice President on January 20, 2005. From April 1997 to August 2001, Mr. Mosteller was an Executive Vice President of Union Planters Bank National Association, a subsidiary of Union Planters Corporation and Chief Operating Officer of Union Planters Mortgage. From 1994 to 1997, Mr. Mosteller was the Chief Financial Officer of Boatmen’s National Mortgage, Inc., the mortgage subsidiary of Boatmen’s Bancshares, Inc. From 1984 to 1994, Mr. Mosteller served as a Senior Manager in Audit Services at Ernst & Young LLP. Mr. Mosteller is a Certified Public Accountant and holds a B.S. in Accounting from the University of Tennessee.
Other Key Medical, Clinical and Regulatory Officers of the Registrant
K. Gary Barnette, Ph.D., was appointed Vice President, Clinical Research and Development Strategy in November 2005, and prior to that he served as Vice President, Clinical Research and Development since January 20, 2005. He also served as our Director of Regulatory Affairs from December 2001 to May 2007. From 1998 to 2001, Dr. Barnette was Assistant Director and then Director, Regulatory Affairs at Solvay Pharmaceuticals, Inc. From 1995 to 1998, Dr. Barnette was a Clinical Pharmacology and Biopharmaceutics Reviewer at the FDA, where he reviewed in the Divisions of Reproductive and Urologic Drug Products, Metabolic and Endocrine Drug Products and Gastrointestinal and Coagulation Drug Products. Dr. Barnette holds a B.S. in Biology from Salem College and a Ph.D. in Basic Pharmaceutical Sciences from West Virginia University.
Shontelle Dodson, Pharm.D., was appointed Vice President, Medical Affairs in 2008. Dr. Dodson has over 15 years of pharmaceutical experience, most recently at Pfizer, Inc., where she served as Senior Director and Group Leader, Global Medical, heading up the field-based medical organization. She also served as Director, Team Leader, US Medical, leading the Viagra® and Revatio® medical teams and the US field-based medical effort supporting the company’s urology and respiratory medicines with total product sales exceeding $5 billion. Dr. Dodson holds a Doctor of Pharmacy degree from Mercer University Southern School of Pharmacy and completed a postdoctoral residency at the Department of Veterans Affairs Medical Center in Nashville.
20
Table of Contents
Jeffrey G. Hesselberg was appointed Vice President, Regulatory Affairs in May 2007. He joined GTx from ICOS Corporation, where from 1996 to May 2007 he served as Manager, Associate Director, and then Director of Regulatory Affairs. Most recently, Mr. Hesselberg worked on the successful development, launch and commercialization of Cialis® (tadalafil) for the treatment of erectile dysfunction. From 1984 to 1996, Mr. Hesselberg worked for Immunex Corporation and the Puget Sound Blood Center. Mr. Hesselberg holds a B.S. in Molecular Biology from the University of Wisconsin–Madison and a MBA from the University of Washington.
Domingo Rodriguez, M.D., was appointed Vice President of Clinical Operations in May 2008. Prior to his appointment, Dr. Rodriguez was the Director of Clinical Operations since October 2005. Dr. Rodriguez joined GTx in November 2004 as a Regional Medical Scientist. From 2001 to 2004, Dr. Rodríguez served as a Medical Director, Medical Science Liaison and District Sales Manager for ICOS Corporation. He began his career in 1987 with Bristol Myers Squibb and for almost 14 years served in various roles in medical affairs, sales and sales training and management. Dr. Rodriguez completed medical school in the Dominican Republic.
| ITEM 1A. | RISK FACTORS |
We have identified the following additional risks and uncertainties that may have a material adverse effect on our business, financial condition or results of operations. Investors should carefully consider the risks described below before making an investment decision. Our business faces significant risks and the risks described below may not be the only risks we face. Additional risks not presently known to us or that we currently believe are immaterial may also significantly impair our business operations. If any of these risks occur, our business, results of operations or financial condition could suffer, the market price of our common stock could decline and you could lose all or part of your investment in our common stock.
Risks Related to Our Financial Condition and Need for Additional Financing
We have incurred losses since inception, and we anticipate that we will incur continued losses for the foreseeable future.
As of December 31, 2011, we had an accumulated deficit of $386.2 million. Our net loss for the year ended December 31, 2011 was $33.3 million. We expect to incur significant net losses for the foreseeable future as we continue our clinical development and research and development activities. These losses, among other things, have had and will continue to have an adverse effect on our stockholders’ equity and working capital.
We have discontinued our toremifene 80 mg and toremifene 20 mg development programs, and we do not anticipate that we will receive any return on our investment in either of our toremifene 80 mg or toremifene 20 mg product candidates. The clinical studies we were conducting for our product candidate, Capesaris® (GTx-758), have been placed on hold by the FDA for safety reasons, pending additional information about the drug product the FDA will be requesting from us, and we have suspended further enrollment in these studies and notified clinical sites to discontinue treatment of subjects with Capesaris®. Our only product candidate in late-stage clinical trials is enobosarm (also known as OstarineTM or GTx-024) and there can be no assurance that the FDA will allow us to further develop Capesaris® in any indication. Significant additional clinical development and financial resources will be required to obtain necessary regulatory approvals for each of these product candidates and to develop these product candidates into commercially viable products. Accordingly, we do not expect to obtain FDA or any other regulatory approvals to market any of our product candidates in the near future, and it is possible our product candidates will never gain regulatory approval.
Because of the numerous risks and uncertainties associated with developing and commercializing small molecule drugs, we are unable to predict the extent of any future losses or when we will become profitable, if at all. We have financed our operations and internal growth primarily through public offerings and private placement of our common stock, as well as payments from our former collaborators. Currently, we have no ongoing collaborations for the development and commercialization of our product candidates. FARESTON® is currently our only commercial product and, until such time that we receive regulatory approval to market any of our product candidates, if ever, we expect that FARESTON® will account for all of our product revenue. For the year ended December 31, 2011, we recognized $6.7 million in net revenues from the sale of FARESTON®. If we and/or any potential future collaborators are unable to develop and commercialize any of our product candidates, if
21
Table of Contents
development is further delayed or eliminated, such as the clinical hold that was placed on the development of Capesaris®, or if sales revenue from any product candidate that receives marketing approval is insufficient, we may never become profitable and we will not be successful.
We will need to raise substantial additional capital and may be unable to raise capital when needed, which would force us to further delay, reduce or eliminate our product development programs or commercialization efforts.
We will need to raise substantial additional capital to:
| • | fund our operations and conduct clinical trials; |
| • | continue our research and development; |
| • | seek regulatory approval for our product candidates; and |
| • | commercialize our product candidates, if any such product candidates receive regulatory approval for commercial sale. |
As of the date of this Annual Report on Form 10-K, we estimate that our current cash, cash equivalents, and short-term investments, together with interest income and net product sales of FARESTON®, will be sufficient to meet our projected operating requirements for at least the next twelve months. We have based this estimate on our current business plan and assumptions that may prove to be wrong. We could utilize our available capital resources sooner than we currently expect, and we could need additional funding sooner than currently anticipated. In any event, to complete the development of and seek regulatory approvals for enobosarm and, if development is permitted to continue, Capesaris®, we will need to obtain substantial additional funding. Our future funding requirements will depend on many factors, including:
| • | the scope, rate of progress and cost of our clinical trials and other research and development activities, including additional development activities or studies that may be required by the FDA if we are permitted to continue developing Capesaris®, as well as our ongoing and any future clinical trials of enobosarm; |
| • | the terms and timing of any potential future collaborative, licensing and other arrangements that we may establish; |
| • | the amount and timing of any licensing fees, milestone payments and royalty payments from potential future collaborators, if any; |
| • | future clinical trial results; |
| • | the cost and timing of regulatory filings and/or approvals to commercialize our product candidates and any related restrictions, limitations, and/or warnings in the label of an approved product candidate; |
| • | the cost and timing of establishing medical education, sales, marketing and distribution capabilities; |
| • | the cost of establishing clinical and commercial supplies of our product candidates and any products that we and/or any potential future collaborators may develop; |
| • | the effect of competing technological and market developments; |
| • | the cost of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights, and the cost of defending any other litigation claims; and |
| • | the extent to which we acquire or invest in businesses, products and technologies, although we currently have no commitments or agreements relating to any of these types of transactions. |
We do not currently have any commitments for future external funding and until we can generate a sufficient amount of product revenue, which we may never do, we expect to finance future cash needs through public or private equity offerings, debt financings or collaboration and licensing arrangements, or a combination of the above, as well as through interest income earned on the investment of our cash balances and short-term investments and revenues from the sale of FARESTON®. In June 2011, we announced a workforce reduction of approximately 15% in order to reduce our operating expenses relating to our discontinued toremifene development programs. If we are unable to raise additional funds when needed, we may need to further reduce our expenditures, perhaps significantly, to preserve our cash. Cost-cutting measures that we may take in the future may not be sufficient to enable us to meet our cash requirements, and they may negatively affect our business and growth prospects.
22
Table of Contents
To the extent we raise additional funds by issuing equity securities, our stockholders may experience dilution, and debt financing, if available, may involve restrictive covenants. Any debt financing or additional equity that we raise may contain terms that are not favorable to us or our stockholders. To the extent we raise additional funds through potential future collaboration and licensing arrangements, it may be necessary to relinquish rights to some of our technologies or product candidates, or grant licenses on terms that are not favorable to us. Our ability to raise additional funds and the terms upon which we are able to raise such funds may be adversely impacted by the clinical hold placed by the FDA on our three Phase II clinical trials to evaluate Capesaris® and the related uncertainty regarding the continued development of Capesaris®, as well as uncertainty regarding our financial condition, the outcomes of our ongoing clinical trials of enobosarm and/or current economic conditions, including the effects of disruptions to and volatility in the credit and financial markets in the United States, the European Union and other regions of the world, including those resulting from or associated with rising government debt levels. As a result of these and other factors, we cannot be certain that additional funding will be available on acceptable terms, or at all. If adequate funds are not available when we need them, we may be required to delay, reduce the scope of or eliminate one or more of our research or development programs, including our SARM and selective ER alpha agonist programs, or conduct additional workforce or other expense reductions, any of which could have a material adverse effect on our business.
Risks Related to Development of Product Candidates
We and any potential future collaborators will not be able to commercialize our product candidates if our preclinical studies do not produce successful results or if our clinical trials do not adequately demonstrate safety and efficacy in humans.
Significant additional research and development and financial resources will be required to obtain necessary regulatory approvals for our product candidates and to develop them into commercially viable products. Preclinical and clinical testing is expensive, can take many years to complete and has an uncertain outcome. Success in preclinical testing and early clinical trials does not ensure that later clinical trials will be successful, and interim results of a clinical trial do not necessarily predict final results. Typically, the failure rate for development candidates is high. If a product candidate fails at any stage of development, we will not have the anticipated revenues from that product candidate to fund our operations, and we will not receive any return on our investment in that product candidate. For example, after we had incurred significant development costs, we announced in February 2012 that the FDA has placed our Phase II clinical studies of Capesaris® on clinical hold pending additional information about Capesaris® the FDA will be requesting from us and in May 2010 our toremifene 20 mg product candidate failed to meet its primary efficacy endpoint in our Phase III clinical trial of toremifene 20 mg for the prevention of prostate cancer in high risk men with high grade prostatic intraepithelial neoplasia. Even if the results of a clinical trial are positive, the efficacy and/or safety results from the trial may be insufficient to support the submission of a NDA to the FDA, or if submitted, the approval of the NDA by the FDA. For example, we received a Complete Response Letter in October 2009 from the FDA regarding our NDA for toremifene 80 mg to reduce fractures in men with prostate cancer on ADT, notifying us that the FDA would not approve the NDA. We have since discontinued our toremifene development programs.
Significant delays in clinical testing could materially impact our product development costs. We do not know whether future clinical trials will begin on time, or whether ongoing clinical trials will need to be restructured or will be completed on schedule, if at all. With our Capesaris® Phase II clinical studies currently on clinical hold by the FDA, we do not know when or if and under what circumstances the FDA will permit further clinical development of Capesaris®, and it is possible that we or the FDA could determine that Capesaris® has an unacceptable safety profile at any dosage preventing any further development of Capesaris®. We or any potential future collaborators may experience numerous unforeseen and/or adverse events during, or as a result of, preclinical testing and the clinical trial process that could delay or prevent our or our potential future collaborators’ ability to commercialize our product candidates, including:
| • | regulators or institutional review boards may not authorize us or any potential future collaborators to commence a clinical trial or conduct a clinical trial at a prospective trial site, or we may experience substantial delays in obtaining these authorizations; |
23
Table of Contents
| • | preclinical or clinical trials may produce negative or inconclusive results, which may require us or any potential future collaborators to conduct additional preclinical or clinical testing or to abandon projects that we expect to be promising; |
| • | even if preclinical or clinical trial results are positive, the FDA or foreign regulatory authorities could nonetheless require us to conduct unanticipated additional clinical trials; |
| • | registration or enrollment in clinical trials may be slower than we anticipate, resulting in significant delays or study terminations; |
| • | we or any potential future collaborators may suspend or terminate clinical trials if the participating patients are being exposed to unacceptable health risks; |
| • | regulators or institutional review boards may suspend or terminate clinical research for various reasons, including noncompliance with regulatory requirements; and |
| • | our product candidates may not have the desired effects or may include undesirable side effects, such as the blood clots observed in our Phase II clinical studies of Capesaris®. |
If any of these events were to occur and, as a result, we or any potential future collaborators have significant delays in or termination of clinical trials, our costs could increase and our ability to generate revenue could be impaired, which would materially and adversely impact our business, financial condition and growth prospects.
If we or any potential future collaborators observe serious or other adverse events during the time our product candidates are in development or after our products are approved and on the market, we or any potential future collaborators may be required to perform lengthy additional clinical trials, may be denied regulatory approval of such products, may be forced to change the labeling of such products or may be required to withdraw any such products from the market, any of which would hinder or preclude our ability to generate revenues.
In our Phase II clinical studies of Capesaris®, we reported to the FDA venous thromboembolic events, or blood clots, in subjects treated with Capesaris at the doses being studied in the trial (1000 mg and higher). There were two deaths in subjects treated with Capesaris® and two deaths in subjects treated with Lupron Depot®. The FDA has placed our Phase II clinical studies of Capesaris® on clinical hold pending our providing additional information about Capesaris® the FDA will be requesting from us. As a result of the clinical hold, we suspended further enrollment into these studies and notified clinical sites to discontinue treatment of subjects with Capesaris®. We do not know when or if and under what circumstances the FDA will permit further clinical development of Capesaris®, and it is possible that we or the FDA could determine that Capesaris® has an unacceptable safety profile at any dosage preventing any further development of Capesaris®.
In our Phase II clinical trials for enobosarm for the treatment of muscle wasting in patients with cancer and healthy older males and postmenopausal females, we observed mild elevations of hepatic enzymes, which can lead to liver failure, in a few patients in both the placebo and enobosarm treated groups. Reductions in high-density lipoproteins have also been observed in subjects treated with enobosarm. Lower levels of high-density lipoproteins could lead to increased risk of cardiovascular events.
If the incidence of serious or other adverse events related to our product candidates increases in number or severity, if a regulatory authority believes that these or other events constitute an adverse effect caused by the drug, or if other effects are identified during clinical trials that we or any potential future collaborators may conduct in the future or after any of our product candidates are approved and marketed:
| • | we or any potential future collaborators may be required to conduct additional preclinical or clinical trials, make changes in the labeling of any such approved products, reformulate any such products, or implement changes to or obtain new approvals of our contractors’ manufacturing facilities; |
| • | regulatory authorities may be unwilling to approve our product candidates or may withdraw approval of our products; |
| • | we may experience a significant drop in the sales of the affected products; |
| • | our reputation in the marketplace may suffer; and |
| • | we may become the target of lawsuits, including class action suits. |
Any of these events could prevent approval or harm sales of the affected product candidates or products, or could substantially increase the costs and expenses of commercializing and marketing any such products.
24
Table of Contents
Risks Related to Our Dependence on Third Parties
If we do not establish collaborations for our product candidates or otherwise raise substantial additional capital, we will likely need to alter our development and any commercialization plans.
Our strategy includes selectively partnering or collaborating with leading pharmaceutical and biotechnology companies to assist us in furthering development and potential commercialization of our product candidates. We face significant competition in seeking appropriate collaborators, and collaborations are complex and time consuming to negotiate and document. We may not be successful in entering into new collaborations with third parties on acceptable terms, or at all, including as a result of the collaboration discussions we are pursuing for enobosarm. In addition, we are unable to predict when, if ever, we will enter into any additional collaborative arrangements because of the numerous risks and uncertainties associated with establishing such arrangements. If we are unable to negotiate new collaborations, we may have to curtail the development of a particular product candidate, reduce, delay, or terminate its development or one or more of our other development programs, delay its potential commercialization or reduce the scope of our sales or marketing activities or increase our expenditures and undertake development or commercialization activities at our own expense. If we elect to increase our expenditures to fund development or commercialization activities on our own, we will need to raise substantial additional capital, which may not be available to us on acceptable terms, or at all. If we do not have sufficient funds, we will not be able to bring our product candidates to market and generate product revenues.
Any collaborative arrangements that we establish in the future may not be successful or we may otherwise not realize the anticipated benefits from these collaborations. In addition, any future collaboration arrangements may place the development and commercialization of our product candidates outside our control, may require us to relinquish important rights or may otherwise be on terms unfavorable to us.
We have in the past established and intend to continue to establish collaborations with third parties to develop and commercialize some of our current and future product candidates, and these collaborations may not be successful or we may otherwise not realize the anticipated benefits from these collaborations. For example, in March 2011, we and Ipsen Biopharm Limited, or Ipsen, mutually agreed to terminate our collaboration and, as a result, we will not receive any additional milestone payments from Ipsen on account of our collaboration with Ipsen. As of the date of this report, we have no ongoing collaborations for the development and commercialization of our product candidates. In the future, we may not be able to locate third-party collaborators to develop and market our product candidates, and we may lack the capital and resources necessary to develop our product candidates alone.
Dependence on collaborative arrangements subjects us to a number of risks, including:
| • | we may not be able to control the amount and timing of resources that our potential future collaborators may devote to our product candidates; |
| • | potential future collaborations may experience financial difficulties or changes in business focus; |
| • | we may be required to relinquish important rights such as marketing and distribution rights; |
| • | should a collaborator fail to develop or commercialize one of our compounds or product candidates, we may not receive any future milestone payments and will not receive any royalties for the compound or product candidate; |
| • | business combinations or significant changes in a collaborator’s business strategy may also adversely affect a collaborator’s willingness or ability to complete its obligations under any arrangement; |
| • | under certain circumstances, a collaborator could move forward with a competing product candidate developed either independently or in collaboration with others, including our competitors; and |
| • | collaborative arrangements are often terminated or allowed to expire, which could delay the development and may increase the cost of developing our product candidates. |
25
Table of Contents
If third parties do not manufacture our product candidates in sufficient quantities, in the required timeframe, and at an acceptable cost, clinical development and commercialization of our product candidates would be delayed.
We do not currently own or operate manufacturing facilities, and we rely, and expect to continue to rely, on third parties for the production of clinical and commercial quantities of our product candidates. Our current and anticipated future dependence upon others for the manufacture of our product candidates may adversely affect our future profit margins, if any, and our ability to develop product candidates and commercialize any product candidates on a timely and competitive basis.
We rely on third-party vendors for the manufacture of enobosarm drug substance. If our supply of enobosarm becomes unusable or if the contract manufacturers that we are currently utilizing to meet our supply needs for enobosarm or any future SARM product candidates prove incapable or unwilling to continue to meet our supply needs, we could experience a delay in conducting any additional clinical trials of enobosarm or any future SARM product candidates. In addition, we rely on third-party contractors for the manufacture of Capesaris® drug substance. We may not be able to maintain or renew our existing or any other third-party manufacturing arrangements on acceptable terms, if at all. If our suppliers fail to meet our requirements for Capesaris®, enobosarm or any future product candidates for any reason, we would be required to obtain alternate suppliers. Any inability to obtain alternate suppliers, including an inability to obtain approval from the FDA of an alternate supplier, would delay or prevent the clinical development and commercialization of these product candidates.
Use of third-party manufacturers may increase the risk that we will not have adequate supplies of our product candidates or products.
Reliance on third-party manufacturers entails risks, to which we would not be subject if we manufactured product candidates or products ourselves, including:
| • | reliance on the third party for regulatory compliance and quality assurance; |
| • | the possible breach of the manufacturing agreement by the third party because of factors beyond our control; |
| • | the possible termination or non-renewal of the agreement by the third party, based on its own business priorities, at a time that is costly or inconvenient for us; and |
| • | drug product supplies not meeting the requisite requirements for clinical trial use. |
If we are not able to obtain adequate supplies of our product candidates, it will be more difficult for us to develop our product candidates and compete effectively. Our product candidates and any products that we and/or our potential future collaborators may develop may compete with other product candidates and products for access to manufacturing facilities.
We have agreed to purchase exclusively from Orion our worldwide requirements of toremifene in a finished tablet form at specified prices under a license and supply agreement. Orion may terminate its supply obligations to us at its election at any time. If Orion elects to terminate its obligation to manufacture and supply us with FARESTON® tablets, any arrangements we make for an alternative supply would have to be made with a qualified alternative supplier with appropriate FDA approval in order for us to obtain our supply requirements for toremifene. In addition, although Orion’s composition of matter patents have expired, and as such, we would not be prevented from manufacturing FARESTON® tablets, there is no obligation on the part of Orion to transfer its manufacturing technology to us or to assist us in developing manufacturing capabilities to meet our supply needs. If our supply rights for FARESTON® tablets are terminated by Orion for any reason, a disruption in the supply could impair our ability to continue to commercialize FARESTON®.
Our present or future manufacturing partners may not be able to comply with FDA-mandated current Good Manufacturing Practice regulations, other FDA regulatory requirements or similar regulatory requirements outside the United States. Failure of our third-party manufacturers or us to comply with applicable regulations could result in sanctions being imposed on us, including fines, injunctions, civil penalties, failure of regulatory authorities to grant marketing approval of our product candidates, delays, suspension or withdrawal of approvals, license revocation, seizures or recalls of product candidates or products, operating restrictions and criminal prosecutions, any of which could significantly and adversely affect supplies of our product candidates.
If third parties on whom we rely do not perform as contractually required or expected, we may not be able to obtain regulatory approval for or successfully commercialize our product candidates.
We do not have the ability to independently conduct clinical trials for our product candidates, and we must rely on third parties, such as contract research organizations, medical institutions, clinical investigators and contract laboratories to conduct our clinical trials. In addition, we rely on third parties to assist with our preclinical development of product candidates. If these third parties do not successfully carry out their contractual duties or
26
Table of Contents
regulatory obligations or meet expected deadlines, if the third parties need to be replaced, or if the quality or accuracy of the data they obtain is compromised due to the failure to adhere to our clinical protocols or regulatory requirements or for other reasons, our preclinical development activities or clinical trials may be extended, delayed, suspended or terminated, and we may not be able to obtain regulatory approval for or successfully commercialize our product candidates.
Risks Related to Our Intellectual Property
If we lose our license from the University of Tennessee Research Foundation, or UTRF, we may be unable to continue a substantial part of our business.
We have licensed intellectual property rights and technology from UTRF used in a substantial part of our business. This license agreement, under which we were granted rights to SARM compounds and technologies, may be terminated by UTRF if we are in breach of our obligations under, or fail to perform any terms of, the agreement and fail to cure that breach. If this agreement is terminated, then we may lose our rights to utilize the SARM technology and intellectual property covered by that agreement to market, distribute and sell licensed products, including enobosarm, which may prevent us from continuing a substantial part of our business and may result in a serious adverse effect on our financial condition, results of operations and any prospects for growth.
If some or all of our, or our licensors’, patents expire or are invalidated or are found to be unenforceable, or if some or all of our patent applications do not result in issued patents or result in patents with narrow, overbroad, or unenforceable claims, or claims that are not supported in regard to written description or enablement by the specification, or if we are prevented from asserting that the claims of an issued patent cover a product of a third party, we may be subject to competition from third parties with products in the same class of products as our product candidates or products with the same active pharmaceutical ingredients as our product candidates.
Our commercial success will depend in part on obtaining and maintaining patent and trade secret protection for our product candidates, as well as the methods for treating patients in the product indications using these product candidates. We will be able to protect our product candidates and the methods for treating patients in the product indications using these product candidates from unauthorized use by third parties only to the extent that we or our exclusive licensors own or control such valid and enforceable patents or trade secrets.
Our rights to certain patents and patent applications relating to SARM compounds that we have licensed from UTRF are subject to the terms of UTRF’s inter-institutional agreements with The Ohio State University, or OSU, and our rights to future related improvements in some instances are subject to UTRF’s exercise of exclusive options under its agreements with OSU for such improvements.
Even if our product candidates and the methods for treating patients for prescribed indications using these product candidates are covered by valid and enforceable patents and have claims with sufficient scope, disclosure and support in the specification, the patents will provide protection only for a limited amount of time. Our and our licensors’ ability to obtain patents can be highly uncertain and involve complex and in some cases unsettled legal issues and factual questions. Furthermore, different countries have different procedures for obtaining patents, and patents issued in different countries provide different degrees of protection against the use of a patented invention by others. Therefore, if the issuance to us or our licensors, in a given country, of a patent covering an invention is not followed by the issuance, in other countries, of patents covering the same invention, or if any judicial interpretation of the validity, enforceability, or scope of the claims in, or the written description or enablement in, a patent issued in one country is not similar to the interpretation given to the corresponding patent issued in another country, our ability to protect our intellectual property in those countries may be limited. Changes in either patent laws or in interpretations of patent laws in the United States and other countries may materially diminish the value of our intellectual property or narrow the scope of our patent protection. For example, on September 16, 2011, the Leahy-Smith America Invents Act, or the Leahy-Smith Act, was signed into law. The Leahy-Smith Act includes a number of significant changes to United States patent law. These include provisions that affect the way patent applications will be prosecuted and may also affect patent defense and enforcement. The United States Patent Office is currently developing regulations and procedures to govern administration of the Leahy-Smith Act, and many of the substantive changes to patent law associated with the Leahy-Smith Act will not become effective until one year or 18 months after its enactment. Accordingly, it is too early to determine what effect or impact the Leahy-Smith Act will have on the operation of our business and the protection and enforcement of our intellectual property. However, the Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents, all of which could have a material adverse effect on our business and financial condition.
27
Table of Contents
Even if patents are issued to us or our licensors regarding our product candidates or methods of using them, those patents can be challenged by our competitors who can argue such patents are invalid or unenforceable, lack sufficient written description or enablement, or that the claims of the issued patents should be limited or narrowly construed. Patents also will not protect our product candidates if competitors devise ways of making or using these product candidates without legally infringing our patents. The Federal Food, Drug, and Cosmetic Act and FDA regulations and policies create a regulatory environment that encourages companies to challenge branded drug patents or to create non-infringing versions of a patented product in order to facilitate the approval of abbreviated new drug applications for generic substitutes. These same types of incentives encourage competitors to submit new drug applications that rely on literature and clinical data not prepared for or by the drug sponsor, providing another less burdensome pathway to approval.
We also rely on trade secrets to protect our technology, especially where we do not believe that patent protection is appropriate or obtainable. However, trade secrets are difficult to protect. Our employees, consultants, contractors, outside scientific collaborators and other advisors may unintentionally or willfully disclose our confidential information to competitors, and confidentiality agreements may not provide an adequate remedy in the event of unauthorized disclosure of confidential information. Enforcing a claim that a third party illegally obtained and is using our trade secrets is expensive and time-consuming, and the outcome is unpredictable. Moreover, our competitors may independently develop equivalent knowledge, methods and know-how. Failure to obtain or maintain trade secret protection could adversely affect our competitive business position.
If we infringe intellectual property rights of third parties, it may increase our costs or prevent us from being able to commercialize our product candidates.
There is a risk that we are infringing the proprietary rights of third parties because numerous United States and foreign issued patents and pending patent applications, which are owned by third parties, exist in the fields that are the focus of our drug discovery, development, and manufacture and process synthesis efforts. Others might have been the first to make the inventions covered by each of our or our licensors’ pending patent applications and issued patents and might have been the first to file patent applications for these inventions. In addition, because patent applications can take many years to issue, there may be currently pending applications, unknown to us or our licensors, which may later result in issued patents that cover the production, manufacture, synthesis, commercialization, formulation or use of our product candidates. In addition, the production, manufacture, synthesis, commercialization, formulation or use of our product candidates may infringe existing patents of which we are not aware. Defending ourselves against third-party claims, including litigation in particular, would be costly and time consuming and would divert management’s attention from our business, which could lead to delays in our development or commercialization efforts. If third parties are successful in their claims, we might have to pay substantial damages or take other actions that are adverse to our business.
As a result of intellectual property infringement claims, or to avoid potential claims, we might:
| • | be prohibited from selling or licensing any product that we and/or any potential future collaborators may develop unless the patent holder licenses the patent to us, which the patent holder is not required to do; |
| • | be required to pay substantial royalties or other amounts, or grant a cross license to our patents to another patent holder; or |
| • | be required to redesign the formulation of a product candidate so that it does not infringe, which may not be possible or could require substantial funds and time. |
Risks Related to Regulatory Approval of Our Product Candidates
If we or any potential future collaborators are not able to obtain required regulatory approvals, we or such collaborators will not be able to commercialize our product candidates, and our ability to generate revenue will be materially impaired.
Our product candidates and the activities associated with their development and commercialization are subject to comprehensive regulation by the FDA, other regulatory agencies in the United States and by comparable authorities in other countries. Failure to obtain regulatory approval for a product candidate will prevent us or any potential future collaborator from commercializing the product candidate. We have not received regulatory approval to market any of our product candidates in any jurisdiction, and we do not expect to obtain FDA or any other regulatory approvals to market any of our product candidates in the near future, if at all. The process of obtaining regulatory approvals is expensive, often takes many years, if approval is obtained at all, and can vary substantially based upon the type, complexity and novelty of the product candidates involved.
28
Table of Contents
Changes in the regulatory approval policy during the development period, changes in or the enactment of additional regulations or statutes, or changes in regulatory review for each submitted product application may cause delays in the approval or rejection of an application. For example, the FDA announced in 2008 that, due to staffing and resource limitations, it has given its managers discretion to miss certain timing goals for completing reviews of NDAs set forth under the Prescription Drug User Fee Act, or PDUFA. Although the FDA has since publicly expressed a recommitment to meeting PDUFA deadlines, it remains unclear whether and to what extent the FDA will adhere to PDUFA deadlines in the future. If the FDA were to miss a PDUFA timing goal for one of our product candidates, the development and commercialization of the product candidate could be delayed. In addition, the Food and Drug Administration Amendments Act of 2007, or FDAAA, which was enacted in September 2007, expands the FDA’s authority to regulate drugs throughout the product life cycle, including enhanced authority to require post-approval studies and clinical trials. Other proposals have been made to impose additional requirements on drug approvals, further expand post-approval requirements and restrict sales and promotional activities. This new legislation, and the additional proposals if enacted, may make it more difficult or burdensome for us or our potential future collaborators to obtain approval of our product candidates. Even if the FDA approves a product candidate, the approval may impose significant restrictions on the indicated uses, conditions for use, labeling, advertising, promotion, marketing and/or production of such product, and may impose ongoing requirements for post-approval studies, including additional research and development and clinical trials. The approval may also impose risk evaluation mitigation strategies, or REMS, on a product if the FDA believes there is a reason to monitor the safety of the drug in the market place. REMS may include requirements for additional training for health care professionals, safety communication efforts and limits on channels of distribution, among other things. The sponsor would be required to evaluate and monitor the various REMS activities and adjust them if need be. The FDA also may impose various civil or criminal sanctions for failure to comply with regulatory requirements, including withdrawal of product approval.
Furthermore, the approval procedure and the time required to obtain approval varies among countries and can involve additional testing beyond that required by the FDA. Approval by one regulatory authority does not ensure approval by regulatory authorities in other jurisdictions.
The FDA has substantial discretion in the approval process and may refuse to accept any application or may decide that our data are insufficient for approval and require additional preclinical, clinical or other studies. For example, in October 2009, we received a Complete Response Letter from the FDA regarding our NDA for toremifene 80 mg to reduce fractures in men with prostate cancer on ADT notifying us that the FDA would not approve our NDA as a result of certain clinical deficiencies identified in the Complete Response Letter. We have since determined to discontinue our toremifene 80 mg development program. While we have met with the FDA to discuss the development program and required endpoints to obtain approval of enobosarm, there can be no assurance that the FDA will ultimately determine that data from our current Phase III clinical trials of enobosarm will be sufficient for approval of this product candidate. With our Phase II clinical studies of Capesaris® on clinical hold by the FDA, our ability to conduct additional studies of Capesaris® and the circumstances under which we will be allowed to do so is subject to discretion of the FDA, and it is possible that the FDA will not permit any further development of Capesaris®.
In addition, varying interpretations of the data obtained from preclinical and clinical testing could delay, limit, or prevent regulatory approval of a product candidate. Even if we submit an application to the FDA for marketing approval of a product candidate, it may not result in marketing approval from the FDA.
We do not expect to receive regulatory approval for the commercial sale of any of our product candidates that are in development in the near future, if at all. The inability to obtain FDA approval or approval from comparable authorities in other countries for our product candidates would prevent us or any potential future collaborators from commercializing these product candidates in the United States or other countries. See the section entitled “Business — Government Regulation” under Part 1, Item 1 of this Annual Report on Form 10-K for additional information regarding risks associated with marketing approval, as well as risks related to potential post-approval requirements.
Risks Related to Commercialization
The commercial success of any products that we and/or any potential future collaborators may develop will depend upon the market and the degree of market acceptance among physicians, patients, health care payors and the medical community.
Any products that we and/or any potential future collaborators may develop may not gain market acceptance among physicians, patients, health care payors and the medical community. If these products do not achieve an
29
Table of Contents
adequate level of acceptance, we may not generate material product revenues or receive royalties to the extent we currently anticipate, and we may not become profitable. The degree of market acceptance of our product candidates, if approved for commercial sale, will depend on a number of factors, including:
| • | efficacy and safety results in clinical trials; |
| • | the prevalence and severity of any side effects; |
| • | potential advantages over alternative treatments; |
| • | whether the products we commercialize remain a preferred course of treatment; |
| • | the ability to offer our product candidates for sale at competitive prices; |
| • | relative convenience and ease of administration; |
| • | the strength of marketing and distribution support; and |
| • | sufficient third-party coverage or reimbursement. |
Our only marketed product generating revenue is FARESTON®, which is subject to a number of risks. These risks may cause sales of FARESTON® to decline.
FARESTON® is currently our only marketed product. FARESTON® is indicated for the treatment of advanced metastatic breast cancer in postmenopausal women. FARESTON® competes against tamoxifen, fulvestrant, and several aromatase inhibitors, including anastrozole, letrozole, and exemestane, for hormonal treatment of breast cancer. Sales of pharmaceuticals for breast cancer in the SERM class have declined in recent years as competitors have gained market share, and we believe this trend will continue. Further, the branded competitors have greater resources and generic competitors are preferred by insurers. Although we continue to market and sell FARESTON®, we no longer utilize a sales force for promotional efforts. Additionally, continued sales may be impacted by many other factors, including the boxed warning added to the label of FARESTON® in March 2011 to highlight that FARESTON® has been shown to prolong the QTc interval in a dose- and concentration-related manner and that prolongation of the QTc interval can result in a type of ventricular tachycardia called Torsades de pointes, which may result in syncope, or temporary loss of consciousness, seizure, and/or death. A boxed warning is the strongest type of warning that the FDA can require for a drug product and warns prescribers that the drug carries a significant risk of serious or even life-threatening adverse effects. The occurrence of one or more of the following risks may cause sales of FARESTON® to decline:
| • | the loss of one or more of our three largest wholesale drug distributors, which together accounted for approximately 95% of our product sales of FARESTON® for the year ended December 31, 2011; |
| • | any further restrictions, limitations, and/or warnings added to the FARESTON® label, including any such changes to its boxed warning; |
| • | the continued success of competing products, including aromatase inhibitors; |
| • | the loss of coverage or reimbursement for FARESTON® from Medicare and Medicaid, private health insurers or other third-party payors; |
| • | exposure to product liability claims related to the commercial sale of FARESTON®, which may exceed our product liability insurance; |
| • | the failure of Orion to maintain regulatory filings or comply with applicable FDA requirements with respect to FARESTON®; |
| • | the introduction of generic toremifene products that compete with FARESTON® for the treatment of breast cancer; and |
| • | the loss of Orion, upon which we rely as a single source, as our supplier of FARESTON®. |
If we are unable to establish sales and marketing capabilities or establish and maintain agreements with third parties to market and sell our product candidates, we may be unable to generate product revenue from such candidates.
We have limited experience as a company in the sales, marketing and distribution of pharmaceutical products, and in any event, we will need to establish sales and marketing capabilities or establish and maintain agreements with third parties to market and sell our product candidates. We may be unable to build our own sales and marketing
30
Table of Contents
capabilities, and there are risks involved with entering into arrangements with third parties to perform these services, which could delay the commercialization of any of our product candidates if approved for commercial sale. In addition, to the extent that we enter into arrangements with third parties to perform sales, marketing and distribution services, our product revenues are likely to be lower than if we market and sell any products that we develop ourselves.
If we and/or any potential future collaborators are unable to obtain reimbursement or experience a reduction in reimbursement from third-party payors for products we sell, our revenues and prospects for profitability will suffer.
Sales of products developed by us and/or any potential future collaborators are dependent on the availability and extent of reimbursement from third-party payors. Changes in the reimbursement policies of these third-party payors that reduce reimbursements for FARESTON® and any other products that we and/or any potential future collaborators may develop and sell could negatively impact our future operating and financial results.
The Medicare Prescription Drug, Improvement and Modernization Act of 2003 established comprehensive Medicare coverage and reimbursement of prescription drugs under Medicare Part D. The prescription drug program established by this legislation may have the effect of reducing the prices that we or any potential future collaborators are able to charge for products we and/or any potential future collaborators develop and sell through the program. This legislation may also cause third-party payors other than the federal government, including the states under the Medicaid program, to discontinue coverage for products that we and/or any potential future collaborators may develop or to lower the amount that they pay.
In March 2010, the United States Congress enacted the Patient Protection and Affordable Care Act and the Health Care and Education Reconciliation Act. This health care reform legislation will increase the number of individuals who receive health insurance coverage and will close a gap in drug coverage under Medicare Part D as established in 2003. However, the legislation also implements cost containment measures that could adversely affect our revenues. These measures include increased drug rebates under Medicaid for brand name prescription drugs, such as FARESTON®, and extension of these rebates to Medicaid managed care, each of which have reduced the amount of net reimbursement received for FARESTON® and would reduce the amount of net reimbursement for any other products that we and/or any potential future collaborators may develop and sell. The legislation also extended 340B discounted pricing on outpatient drugs to children’s hospitals, critical access hospitals, and rural health centers, which has reduced the amount of reimbursement received for drugs purchased by these new 340B-covered entities.
Additional provisions of the health care reform legislation may negatively affect our revenues and prospects for profitability in the future. Along with other pharmaceutical manufacturers and importers of brand name prescription drugs, we are assessed a fee based on our proportionate share of sales of brand name prescription drugs to certain government programs, including Medicare and Medicaid, made in the preceding year if such sales exceed a defined threshold. As part of the health care reform legislation’s provisions closing a funding gap that currently exists in the Medicare Part D prescription drug program (commonly known as the “donut hole”), as of January 1, 2011, we are required to provide a 50% discount on brand name prescription drugs, including FARESTON®, sold to beneficiaries who fall within the donut hole.
The health care reform legislation has been subject to judicial challenge. While some courts have upheld the law, other courts have concluded that the individual mandate component of the law is unconstitutional. One of those courts determined that the individual mandate component could not be severed from the law and therefore concluded that the entire law was void. All of the rulings on the merits are being appealed. There is no certainty regarding the final outcome of the litigation or the impact of the outcome on the pricing and potential profitability of any products that we and/or any potential future collaborators may develop.
Economic pressure on state budgets may result in states increasingly seeking to achieve budget savings through mechanisms that limit coverage or payment for drugs. State Medicaid programs are increasingly requesting manufacturers to pay supplemental rebates and requiring prior authorization for use of drugs where supplemental rebates are not provided. Private health insurers and managed care plans are likely to continue challenging the prices charged for medical products and services, and many of these third-party payors may limit reimbursement for newly-approved health care products. In particular, third-party payors may limit the indications for which they will reimburse patients who use any products that we and/or any potential future collaborators may develop or sell. These cost-control initiatives could decrease the price we might establish for products that we or any potential future collaborators may develop or sell, which would result in lower product revenues or royalties payable to us.
31
Table of Contents
Similar cost containment initiatives exist in countries outside of the United States, particularly in the countries of the European Union, where the pricing of prescription pharmaceuticals is subject to governmental control. In these countries, pricing negotiations with governmental authorities can take six to twelve months or longer after the receipt of regulatory marketing approval for a product. To obtain reimbursement or pricing approval in some countries, we or any potential future collaborators may be required to conduct a clinical trial that compares the cost effectiveness of our product candidates or products to other available therapies. The conduct of such a clinical trial could be expensive and result in delays in our or a potential future collaborators’ commercialization efforts. Third-party payors are challenging the prices charged for medical products and services, and many third-party payors limit reimbursement for newly-approved health care products. In particular, third-party payors may limit the indications for which they will reimburse patients who use any products that we and/or any potential future collaborators may develop or sell. Cost-control initiatives could decrease the price we might establish for products that we or any potential future collaborators may develop or sell, which would result in lower product revenues or royalties payable to us.
Another development that could affect the pricing of drugs would be if the Secretary of Health and Human Services allowed drug reimportation into the United States. The Medicare Prescription Drug, Improvement and Modernization Act of 2003 gives discretion to the Secretary of Health and Human Services to allow drug reimportation into the United States under some circumstances from foreign countries, including from countries where the drugs are sold at a lower price than in the United States. If the circumstances were met and the Secretary exercised the discretion to allow for the direct reimportation of drugs, it could decrease the price we or any potential future collaborators receive for any products that we and/or any potential future collaborators may develop, negatively affecting our revenues and prospects for profitability.
Health care reform measures could hinder or prevent our product candidates’ commercial success.
Among policy makers and payors in the United States and elsewhere, there is significant interest in promoting health care reform, as evidenced by the enactment in the United States of the Patient Protection and Affordable Care Act and the Health Care and Education Reconciliation Act in 2010. It is likely that federal and state legislatures within the United States and foreign governments will continue to consider changes to existing health care legislation. These changes adopted by governments may adversely impact our business by lowering the price of health care products in the United States and elsewhere.
We operate in a highly regulated industry and new laws, regulations or judicial decisions, or new interpretations of existing laws, regulations or decisions, related to health care availability, method of delivery or payment for health care products and services, or sales, marketing and pricing practices could negatively impact our business, operations and financial condition.
If product liability lawsuits are brought against us, we may incur substantial liabilities and may be required to limit commercialization of any products that we may develop.
We face an inherent risk of product liability exposure related to our commercial sale of FARESTON® and the testing of our product candidates in human clinical trials and will face an even greater risk if we commercially sell any product that we may develop. If we cannot successfully defend ourselves against claims that our product candidates or products caused injuries, we will incur substantial liabilities. Regardless of merit or eventual outcome, liability claims may result in:
| • | decreased demand for any product candidates or products; |
| • | injury to our reputation; |
| • | withdrawal of clinical trial participants; |
| • | costs to defend the related litigation; |
| • | substantial monetary awards to trial participants or patients; |
| • | loss of revenue; and |
| • | the inability to commercialize any products for which we obtain or hold marketing approvals. |
We have product liability insurance that covers our clinical trials and commercial products up to a $20 million annual aggregate limit. Insurance coverage is increasingly expensive. We may not be able to maintain insurance coverage at a reasonable cost, and we may not be able to obtain insurance coverage that will be adequate to satisfy any liability that may arise.
32
Table of Contents
If our competitors are better able to develop and market products than any products that we and/or any potential future collaborators may develop, our commercial opportunity will be reduced or eliminated.
We face competition from commercial pharmaceutical and biotechnology enterprises, as well as from academic institutions, government agencies and private and public research institutions. Our commercial opportunities will be reduced or eliminated if our competitors develop and commercialize products that are safer, more effective, have fewer side effects or are less expensive than any products that we and/or any potential future collaborators may develop. Competition could result in reduced sales and pricing pressure on our product candidates, if approved, which in turn would reduce our ability to generate meaningful revenue and have a negative impact on our results of operations. In addition, significant delays in the development of our product candidates could allow our competitors to bring products to market before us and impair any ability to commercialize our product candidates.
Various products are currently marketed or used off-label for some of the diseases and conditions that we are targeting in our pipeline, and a number of companies are or may be developing new treatments. These product uses, as well as promotional efforts by competitors and/or clinical trial results of competitive products, could significantly diminish any ability to market and sell any products that we and/or any potential future collaborators may develop.
With respect to our SARM program, there are other SARM product candidates in development that may compete with enobosarm and any future SARM product candidates, if approved for commercial sale, including SARMs in development from Ligand Pharmaceuticals Inc., GlaxoSmithKline, Galapagos NV and Bristol-Myers Squibb Company. Pfizer Inc., Eli Lilly & Co. and Amgen have myostatin inhibitors in development that may compete with enobosarm if approved for commercial sale. In addition, Cytokinetics, Inc. is developing a troponin activator with a muscle specific mechanism in Phase II studies, with a focus on neurological muscle diseases (amyotrophic lateral sclerosis and myasthenia gravis). Moreover, there are other categories of drugs in development, including ghrelin receptor agonists, growth hormone, secretagogues, inflammatory modulators and other agents, that may have some muscle activity. Helsinn Group is developing anamorelin, a ghrelin receptor agonist, in Phase III clinical trials for treatment of cancer cachexia in patients with non-small cell lung cancer. Appetite stimulants such as Megace® (megestrol acetate) and dronabinol are used off-label for the treatment of weight loss and the treatment of loss of appetite in patients with cancer.
We have been developing Capesaris® for first line hormonal treatment of advanced prostate cancer, and there are several products approved to reduce testosterone levels in men with advanced prostate cancer that may compete with Capesaris® if approved for commercial sale, including those marketed by Abbott Laboratories (Lupron Depot®), Sanofi-Aventis (Eligard®), AstraZeneca (Zoladex®), Ferring Pharmaceuticals (Firmagon®), Endo Pharmaceuticals (Vantas®) and Watson Pharmaceuticals (Trelstar®). Additionally, there are various products approved or under clinical development to treat men with advanced prostate cancer who have castration resistant prostate cancer, or CRPC. If the FDA permits us to continue development of Capesaris® to treat men with CRPC, there may be several products to compete with Capesaris® if approved for this indication, such as products being developed by Medivation, Inc., Millennium: The Takeda Oncology Company and Johnson & Johnson.
Many of our competitors have significantly greater financial resources and expertise in research and development, manufacturing, preclinical testing, conducting clinical trials, obtaining regulatory approvals and marketing approved products than we do. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These third parties compete with us in recruiting and retaining qualified scientific and management personnel, establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies and technology licenses complementary to our programs or advantageous to our business.
Risks Related to Employees and Growth
If we fail to attract and keep senior management and key scientific personnel, we may be unable to successfully develop or commercialize our product candidates.
Our success depends on our continued ability to attract, retain and motivate highly qualified management, clinical and scientific personnel and on our ability to develop and maintain important relationships with leading academic institutions, clinicians and scientists. If we are not able to attract and keep senior management and key scientific personnel, particularly Dr. Mitchell S. Steiner, we may not be able to successfully develop or commercialize our product candidates. All of our employees are at-will employees and can terminate their employment at any time. We do not carry “key person” insurance covering members of senior management, other than $22.5 million of insurance covering Dr. Steiner.
33
Table of Contents
In December 2009, we announced a reduction of approximately 26% of our workforce in order to reduce our operating expenses in connection with the receipt of the Complete Response Letter regarding our NDA for toremifene 80 mg. We also announced a reduction of approximately 15% of our workforce in June 2011 in connection with our decision to discontinue the development of toremifene 80 mg and toremifene 20 mg. These and any future workforce reductions may negatively affect our ability to retain or attract talented employees.
We will need to hire additional employees in order to commercialize our product candidates in the future. Any inability to manage future growth could harm our ability to commercialize our product candidates, increase our costs and adversely impact our ability to compete effectively.
In order to commercialize our product candidates in the future, we may need to hire experienced sales and marketing personnel to sell and market those product candidates we decide to commercialize, and we will need to expand the number of our managerial, operational, financial and other employees to support commercialization. Competition exists for qualified personnel in the biotechnology field.
Future growth will impose significant added responsibilities on members of management, including the need to identify, recruit, maintain and integrate additional employees. Our future financial performance and our ability to commercialize our product candidates and to compete effectively will depend, in part, on our ability to manage any future growth effectively.
Risks Related to Our Common Stock
Market volatility may cause our stock price and the value of your investment to decline.
The market prices for securities of biotechnology companies in general have been highly volatile and may continue to be so in the future. The following factors, in addition to other risk factors described in this section, may have a significant impact on the market price of our common stock:
| • | delays in the enrollment or completion of our ongoing clinical trials of enobosarm or adverse results or events reported in any of our enobosarm clinical trials; |
| • | whether, to what extent and under what conditions the FDA will permit us to continue developing Capesaris, if at all, and if development is continued, any further reports of blood clots or other adverse events observed in any potential future studies of Capesaris®; |
| • | our ability to enter into new collaborative arrangements with respect to our product candidates; |
| • | the terms and timing of any future collaborative, licensing or other arrangements that we may establish; |
| • | our ability to raise additional capital to carry through with our clinical development plans and current and future operations and the terms of any related financing arrangements; |
| • | the timing of achievement of, or failure to achieve, our and any potential future collaborators’ clinical, regulatory and other milestones, such as the commencement of clinical development, the completion of a clinical trial or the receipt of regulatory approval; |
| • | announcement of FDA approval or non-approval of our product candidates or delays in or adverse events during the FDA review process; |
| • | actions taken by regulatory agencies with respect to our product candidates or products, our clinical trials or our sales and marketing activities, including regulatory actions requiring or leading to restrictions, limitations and/or warnings in the label of an approved product candidate; |
| • | additional changes to the label for FARESTON®, including its boxed warning, that further restrict how we market and sell FARESTON®; |
| • | the commercial success of any product approved by the FDA or its foreign counterparts; |
| • | introductions or announcements of technological innovations or new products by us, our potential future collaborators, or our competitors, and the timing of these introductions or announcements; |
| • | market conditions for equity investments in general, or the biotechnology or pharmaceutical industries in particular; |
| • | regulatory developments in the United States and foreign countries; |
34
Table of Contents
| • | changes in the structure or reimbursement policies of health care payment systems; |
| • | any intellectual property infringement lawsuit involving us; |
| • | actual or anticipated fluctuations in our results of operations; |
| • | changes in financial estimates or recommendations by securities analysts; |
| • | hedging or arbitrage trading activity that may develop regarding our common stock; |
| • | sales of large blocks of our common stock; |
| • | sales of our common stock by our executive officers, directors and significant stockholders; |
| • | changes in accounting principles; and |
| • | the loss of any of our key scientific or management personnel. |
The stock markets in general, and the markets for biotechnology stocks in particular, have experienced significant volatility that has often been unrelated to the operating performance of particular companies. The financial markets continue to face significant uncertainty, resulting in a decline in investor confidence and concerns about the proper functioning of the securities markets, which decline in general investor confidence has resulted in depressed stock prices for many companies notwithstanding the lack of a fundamental change in their underlying business models or prospects. These broad market fluctuations may adversely affect the trading price of our common stock.
In the past, class action litigation has often been instituted against companies whose securities have experienced periods of volatility in market price. Any such litigation brought against us could result in substantial costs, which would hurt our financial condition and results of operations and divert management’s attention and resources, which could result in delays of our clinical trials or commercialization efforts.
Our executive officers, directors and largest stockholders have the ability to control all matters submitted to stockholders for approval.
As of December 31, 2011, our executive officers, directors and holders of 5% or more of our outstanding common stock beneficially owned approximately 59.4% of our outstanding common stock, and our executive officers and directors alone beneficially owned approximately 38.5% of our outstanding common stock. As a result, these stockholders, acting together, may or will have the ability to control all matters requiring approval by our stockholders, including the election of directors and the approval of mergers or other business combination transactions. The interests of this group of stockholders may not always coincide with our interests or the interests of other stockholders.
Anti-takeover provisions in our charter documents and under Delaware law could make an acquisition of us, which may be beneficial to our stockholders, more difficult and may prevent attempts by our stockholders to replace or remove our current management.
Provisions in our certificate of incorporation and our bylaws may delay or prevent an acquisition of us or a change in our management. In addition, these provisions may frustrate or prevent any attempts by our stockholders to replace or remove our current management by making it more difficult for stockholders to replace members of our Board of Directors. Because our Board of Directors is responsible for appointing the members of our management team, these provisions could in turn affect any attempt by our stockholders to replace current members of our management team. These provisions include:
| • | a classified Board of Directors; |
| • | a prohibition on actions by our stockholders by written consent; |
| • | the ability of our Board of Directors to issue preferred stock without stockholder approval, which could be used to institute a “poison pill” that would work to dilute the stock ownership of a potential hostile acquirer, effectively preventing acquisitions that have not been approved by our Board of Directors; and |
| • | limitations on the removal of directors. |
Moreover, because we are incorporated in Delaware, we are governed by the provisions of Section 203 of the Delaware General Corporation Law, which prohibits a person who owns in excess of 15% of our outstanding voting stock from merging or combining with us for a period of three years after the date of the transaction in which the
35
Table of Contents
person acquired in excess of 15% of our outstanding voting stock, unless the merger or combination is approved in a prescribed manner. Finally, these provisions establish advance notice requirements for nominations for election to our Board of Directors or for proposing matters that can be acted upon at stockholder meetings. These provisions would apply even if the offer may be considered beneficial by some stockholders.
If there are substantial sales of our common stock, the market price of our common stock could drop substantially, even if our business is doing well.
For the 12-month period ended December 31, 2011, the average daily trading volume of our common stock on The NASDAQ Global Market was 246,934 shares. As a result, future sales of a substantial number of shares of our common stock in the public market, or the perception that such sales may occur, could adversely affect the then-prevailing market price of our common stock. As of December 31, 2011, we had 62,790,223 shares of common stock outstanding.
Moreover, J.R. Hyde, III and Oracle Partners, L.P., two of our largest stockholders, and certain of their affiliates, have rights, subject to some conditions, to require us to file registration statements covering the approximately 10.8 million shares of common stock they hold in the aggregate which are subject to registration rights or to include these shares in registration statements that we may file for ourselves or other stockholders. If any of these large stockholders were to sell large blocks of shares in a short period of time, the market price of our common stock could drop substantially.
| ITEM 1B. | UNRESOLVED STAFF COMMENTS |
None.
| ITEM 2. | PROPERTIES |
We sublease approximately 31,000 square feet of laboratory and office space located at 3 North Dunlap Street, Memphis, Tennessee from the University of Tennessee Research Foundation. The sublease expires on December 31, 2012, unless sooner terminated in accordance with the terms of this sublease. We have an option to extend this sublease for an additional two years. We also sublease approximately 31,000 square feet of office space located at 175 Toyota Plaza, Memphis, Tennessee, under an operating lease which expires on April 30, 2015.
| ITEM 3. | LEGAL PROCEEDINGS |
We are not currently involved in any material legal proceedings.
| ITEM 4. | MINE SAFETY DISCLOSURES |
Not applicable.
| ITEM 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES |
Market for Registrant’s Common Equity
Our common stock began trading on The NASDAQ Global Market under the symbol “GTXI” on February 3, 2004. The following table presents, for the periods indicated, the high and low intraday sales prices per share of our common stock as reported on The NASDAQ Global Market.
36
Table of Contents
| 2011 | 2010 | |||||||||||||||
| High | Low | High | Low | |||||||||||||
| First Quarter |
$ | 3.12 | $ | 2.27 | $ | 4.47 | $ | 3.21 | ||||||||
| Second Quarter |
6.86 | 2.60 | 4.30 | 1.90 | ||||||||||||
| Third Quarter |
4.93 | 2.67 | 3.60 | 2.76 | ||||||||||||
| Fourth Quarter |
4.74 | 2.34 | 3.95 | 2.45 | ||||||||||||
On February 29, 2012, the closing price of our common stock as reported on The NASDAQ Global Market was $3.46 per share and there were approximately 77 holders of record of our common stock.
Performance Graph1
The rules of the SEC require that we include in our annual report to stockholders a line-graph presentation comparing cumulative stockholder returns on our common stock with a broad equity market index that includes companies whose equity securities are traded on the NASDAQ and either a published industry or line-of-business standard index or an index of peer companies selected by us. We have elected to use The NASDAQ Composite Index (which tracks the aggregate price performance of equity securities of companies traded on NASDAQ Stock Market) and The NASDAQ Biotechnology Index (consisting of a group of approximately 117 companies in the biotechnology sector, including us) for purposes of the performance comparison that appears below.
The following graph shows the cumulative total stockholder return assuming the investment of $100.00 at the closing prices on December 31, 2006 on The NASDAQ Global Market for: (1) our common stock; (2) The NASDAQ Composite Index and (3) The NASDAQ Biotechnology Index. All values assume reinvestment of the full amounts of all dividends. No dividends have been declared on our common stock. The closing sale price of our common stock on December 31, 2011 as reported on The NASDAQ Global Market was $3.36.
The stockholder return shown on the graph below is not necessarily indicative of future performance, and we do not make or endorse any predictions as to future stockholder returns.
37
Table of Contents
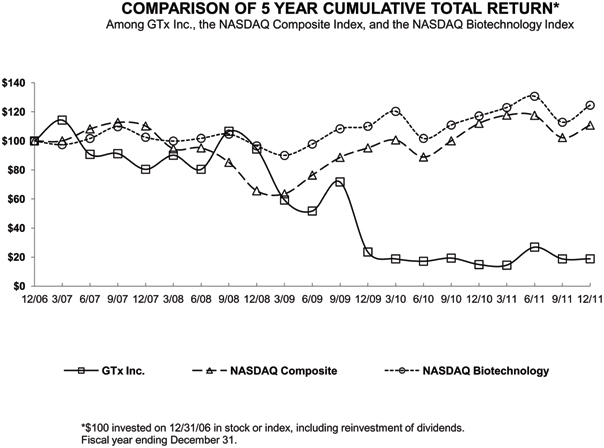
| 1 | The material in this section is not “soliciting material,” is not deemed filed with the SEC and is not to be incorporated by reference in any filing of GTx, Inc. under the Securities Act of 1933 or the Securities Exchange Act of 1934 whether made before or after the date hereof and irrespective of any general incorporation language in such filing. |
Dividend Policy
We have never declared or paid any cash dividends on our capital stock. We currently intend to retain any future earnings to fund the development and expansion of our business, and therefore we do not anticipate paying cash dividends on our common stock in the foreseeable future. Any future determination to pay dividends will be at the discretion of our Board of Directors.
| ITEM 6. | SELECTED FINANCIAL DATA |
You should read the selected financial data below in conjunction with “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and the audited financial statements, notes thereto and other financial information included elsewhere in this Annual Report on Form 10-K. The statements of operations data for the years ended December 31, 2011, 2010 and 2009, and the balance sheet data as of December 31, 2011 and 2010, are derived from our audited financial statements included elsewhere in this Annual Report on Form 10-K. The statements of operations data for the years ended December 31, 2008 and 2007, and the consolidated balance sheet data as of December 31, 2009, 2008 and 2007, are derived from our audited financial statements that are not included in this Annual Report on Form 10-K. Historical results are not indicative of the results to be expected in the future.
38
Table of Contents
| Years Ended December 31, | ||||||||||||||||||||
| 2011 | 2010 | 2009 | 2008 | 2007 | ||||||||||||||||
| (in thousands, except per share data) | ||||||||||||||||||||
| Statement of Operations Data: |
||||||||||||||||||||
| Revenues: |
||||||||||||||||||||
| Product sales, net |
$ | 6,673 | $ | 3,827 | $ | 3,289 | $ | 1,088 | $ | 1,076 | ||||||||||
| Collaboration revenue |
8,066 | 56,786 | 11,441 | 12,440 | 6,050 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Total revenues |
14,739 | 60,613 | 14,730 | 13,528 | 7,126 | |||||||||||||||
| Cost and expenses: |
||||||||||||||||||||
| Cost of product sales |
1,055 | 768 | 1,290 | 649 | 621 | |||||||||||||||
| Research and development expenses |
31,938 | 28,495 | 32,344 | 44,259 | 38,508 | |||||||||||||||
| General and administrative expenses |
15,438 | 17,419 | 27,778 | 23,140 | 13,667 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Total costs and expenses |
48,431 | 46,682 | 61,412 | 68,048 | 52,796 | |||||||||||||||
| (Loss) income from operations |
(33,692 | ) | 13,931 | (46,682 | ) | (54,520 | ) | (45,670 | ) | |||||||||||
| Other income, net |
398 | 1,363 | 188 | 2,740 | 5,311 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| (Loss) income before income taxes |
(33,294 | ) | 15,924 | (46,494 | ) | (51,780 | ) | (40,359 | ) | |||||||||||
| Income tax benefit |
— | — | 238 | — | — | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Net (loss) income |
(33,294 | ) | $ | 15,294 | $ | (46,256 | ) | $ | (51,780 | ) | $ | (40,359 | ) | |||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Net (loss) income per share: |
||||||||||||||||||||
| Basic and diluted |
$ | (0.58 | ) | $ | 0.39 | $ | (1.27 | ) | $ | (1.43 | ) | $ | (1.16 | ) | ||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| As of December 31, | ||||||||||||||||||||
| 2011 | 2010 | 2009 | 2008 | 2007 | ||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| Balance Sheet Data: |
||||||||||||||||||||
| Cash, cash equivalents and short-term investments |
$ | 74,440 | $ | 58,631 | $ | 49,044 | $ | 97,667 | $ | 109,988 | ||||||||||
| Working capital |
70,772 | 55,055 | 34,723 | 79,047 | 132,932 | |||||||||||||||
| Total assets |
78,184 | 64,250 | 57,721 | 108,109 | 159,730 | |||||||||||||||
| Accumulated deficit |
(386,174 | ) | (352,880 | ) | (368,174 | ) | (321,918 | ) | (270,138 | ) | ||||||||||
| Total stockholders’ equity (deficit) |
71,874 | 51,727 | (8,750 | ) | 32,018 | 78,917 | ||||||||||||||
39
Table of Contents
| ITEM 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
The following discussion and analysis should be read in conjunction with our financial statements and related notes included elsewhere in this Annual Report on Form 10-K. This discussion contains forward-looking statements based upon current expectations that involve risks and uncertainties. Our actual results and the timing of selected events could differ materially from those anticipated in these forward-looking statements as a result of several factors, including those set forth under Part I, Item 1A “Risk Factors” and elsewhere in this Annual Report on Form 10-K. See “Special Note Regarding Forward-Looking Statements” in this Annual Report on Form 10-K.
Overview
Business Overview
We are a biopharmaceutical company dedicated to the discovery, development and commercialization of small molecules that selectively target hormone pathways for the treatment of cancer, cancer supportive care, and other serious medical conditions.
Business Highlights
We are developing selective androgen receptor modulators, or SARMs, a new class of drugs with the potential to prevent and treat muscle wasting in patients with cancer and other musculoskeletal wasting or muscle loss conditions, including chronic sarcopenia (age related muscle loss). Our lead SARM product candidate, enobosarm (also known as OstarineTM or GTx-024), has to date been evaluated in eight completed clinical trials enrolling approximately 600 subjects, including in a Phase Ib and two Phase II efficacy studies. Enobosarm is the generic name given to the compound by the USAN Council and the World Health Organization, and is the first compound to receive the SARM stem in its name from USAN, recognizing enobosarm as the first in this new class of compounds.
We held End of Phase II meetings with the U.S. Food and Drug Administration, or FDA, prior to initiating our Phase III clinical development of enobosarm for the prevention and treatment of muscle wasting in patients with non-small cell lung cancer, or NSCLC. Based upon data from our Phase II clinical trials and with feedback from the FDA, we designed the POWER 1 and POWER 2 (Prevention and treatment Of muscle Wasting in cancER) pivotal Phase III clinical trials evaluating enobosarm for this indication. We also met with representatives of the Medicines and Healthcare Products Regulatory Agency (United Kingdom) and Medical Products Agency (Sweden), who confirmed that the design of the POWER 1 and POWER 2 clinical trials should be sufficient for the European Medicines Agency to support registration in Europe. We are conducting these trials in clinical sites in the United States, Europe, and South America. In each of the placebo-controlled, double-blind clinical trials, approximately 300 patients with Stage III or IV non-small cell lung cancer will be randomized to placebo or enobosarm 3 mg at the time they are to begin first line chemotherapy. Enrollment in each of the pivotal Phase III clinical trials is ongoing. The trials are evaluating as co-primary endpoints the effect of enobosarm versus placebo on total lean body mass (muscle) assessed by dual x-ray absorptiometry, or DXA, and on physical function assessed by the Stair Climb Test at three months. Durability of effect is being assessed as a secondary endpoint at five months. We currently expect data from these pivotal Phase III clinical trials in the first quarter of 2013. We intend to continue our pursuit of a strategic partnership or collaboration for the development and commercialization of SARMs, including enobosarm.
On February 21, 2012, we announced that the FDA had placed a clinical hold on our clinical trials to evaluate Capesaris® (GTx-758), an oral nonsteroidal selective estrogen receptor alpha agonist, or ER alpha agonist, for primary (first line) androgen deprivation therapy for advanced prostate cancer and secondary (second line) hormonal treatment. A clinical hold is a notification issued by the FDA to the trial sponsor to delay a clinical trial or suspend an ongoing clinical trial. The clinical hold, which was effective as of February 17, 2012, affects our Phase II maintenance dose finding clinical trial evaluating Capesaris® 1000 mg and 2000 mg once-a-day doses compared to Lupron Depot® (leuprolide acetate for depot suspension) in 164 men with advanced prostate cancer and our Phase II loading dose finding clinical trial evaluating a 1000 mg twice-a-day dose or 1500 mg twice-a-day dose to medically castrate up to 104 men with advanced prostate cancer by day 28. After day 28, castrate patients were to continue treatment on one of two once-a-day doses of Capesaris®, 2000 mg or 1000 mg, until day 360. Additionally, the clinical hold applies to our Phase II clinical trial evaluating Capesaris® 2000 mg once-a-day dose to determine the ability of Capesaris® to reduce serum prostate specific antigen, or PSA, and the duration of this PSA reduction in 25 men with castration resistant prostate cancer, or CRPC, who were currently receiving androgen deprivation therapy, or ADT. The clinical hold followed our reports to the FDA of venous thromboembolic events, or
40
Table of Contents
blood clots, in subjects treated with Capesaris® at the doses being studied in the trials (1000 mg and higher). There were two deaths in subjects treated with Capesaris® and two deaths in subjects treated with Lupron Depot®. As a result of the clinical hold, we suspended further enrollment into these three studies and notified clinical sites to discontinue treatment of subjects with Capesaris®. Subject to further input from the FDA, we believe there may be a path forward to develop Capesaris® at lower doses to treat men with metastatic hormone sensitive prostate cancer and men with CRPC. We plan to work with the FDA to determine the appropriate course of action, if any, to evaluate Capesaris® in these patient populations. There can be no assurance that the FDA will allow us to further develop Capesaris® in these or any other patient populations.
We sell FARESTON® (toremifene citrate) 60 mg tablets, approved for the treatment of advanced metastatic breast cancer in postmenopausal women, in the United States. We previously discontinued our toremifene 80 mg and toremifene 20 mg development programs.
Financial Highlights
Our net loss for the year ended December 31, 2011 was $33.3 million. Our net loss included the recognition of $8.1 million of deferred revenue following the termination of our former license and collaboration agreement with Ipsen Biopharm Limited, or Ipsen, and FARESTON® net product sales of $6.7 million. In June 2011, we announced a reduction of approximately 15% of our workforce in order to reduce our operating expenses relating to our discontinued toremifene development programs. We expect to incur significant net losses in 2012 and for the foreseeable future as we continue our clinical development and research and development activities.
At December 31, 2011, we had cash, cash equivalents and short-term investments of $74.4 million, compared to $58.6 million at December 31, 2010. On June 28, 2011, we completed an underwritten public offering of 11,023,000 shares of our common stock at a price to the public of $4.75 per share. Net cash proceeds from the public offering were approximately $49.0 million, after deducting the underwriting discount and offering expenses.
As of the date of this Annual Report on Form 10-K, we estimate that our current cash, cash equivalents, and short-term investments, together with interest income and net product sales of FARESTON®, will be sufficient to meet our projected operating requirements for at least the next twelve months. We have based this estimate on our current business plan and assumptions that may prove to be wrong. We could utilize our available capital resources sooner than we currently expect, and we could need additional funding sooner than currently anticipated. In any event, to complete the development of and seek regulatory approval for enobosarm and, if development is permitted to continue, Capesaris®, we will need to obtain substantial additional funding.
Research and Development
Since our inception in 1997, we have been focused on drug discovery and development programs. Research and development expenses include, but are not limited to, our expenses for personnel associated with our research activities, screening and identification of product candidates, formulation and synthesis activities, manufacturing, preclinical studies, toxicology studies, clinical trials, regulatory and medical affairs activities, quality assurance activities and license fees.
We expect our research and development expenses for fiscal year 2012 to increase as compared to fiscal year 2011 and to be primarily focused on the following:
| • | the continued clinical development of enobosarm; |
| • | if and to the extent the FDA permits us to continue developing Capesaris®, development activities relating to Capesaris®; and |
| • | the continued preclinical development of other potential product candidates; |
There is a risk that any drug discovery and development program may not produce revenue. Moreover, because of uncertainties inherent in drug discovery and development, including those factors described in Part I, Item 1A “Risk Factors” of this Annual Report on Form 10-K, we may not be able to successfully develop and commercialize any of our product candidates.
41
Table of Contents
Drug development in the United States is a process that includes several steps defined by the FDA. The FDA approval process for a new drug involves completion of preclinical studies and the submission of the results of these studies to the FDA, together with proposed clinical protocols, manufacturing information, analytical data and other information in an Investigational New Drug application which must become effective before human clinical trials may begin. Clinical development typically involves three phases of study: Phase I, II and III. The most significant costs associated with clinical development are the Phase III clinical trials as they tend to be the longest and largest studies conducted during the drug development process. After completion of clinical trials, a New Drug Application, or NDA, may be submitted to the FDA. In responding to a NDA, the FDA may refuse to file the application, or if accepted for filing, the FDA may not grant marketing approval, request additional information or deny the application if it determines that the application does not provide an adequate basis for approval. Even if the FDA grants marketing approval, the FDA may impose restrictions, limitations and/or warnings in the label of an approved product candidate, which may adversely affect the marketability of the product or limit the patients to whom the product is prescribed. In some cases, the FDA may give conditional approval of a NDA for a product candidate on the NDA sponsor’s agreement to conduct additional clinical trials to further assess the product’s safety and effectiveness after NDA approval. Any approval of a NDA by the FDA conditioned on completing additional clinical trials may require the sponsor to discontinue further marketing of the product if data from the clinical trial fails to demonstrate sufficient efficacy and safety in accordance with the agreed-upon protocol for the clinical trial.
The successful development and commercialization of our product candidates is highly uncertain. We cannot reasonably estimate or know the nature, timing and estimated costs of the efforts necessary to complete the development and commercialization of, or the period in which material net cash inflows are expected to commence from, any of our product candidates due to the numerous risks and uncertainties associated with developing and commercializing drugs, including the uncertainty of:
| • | our ability to progress product candidates into preclinical and clinical trials; |
| • | the scope, rate of progress and cost of our clinical trials and other research and development activities, including additional development activities or studies that may be required by the FDA if we are permitted to continue developing Capesaris®, as well as our ongoing and any future clinical trials of enobosarm and any future clinical trials of enobosarm; |
| • | the terms and timing of any potential future collaborative, licensing and other arrangements that we may establish; |
| • | the amount and timing of any licensing fees, milestone payments and royalty payments from potential future collaborators, if any; |
| • | future clinical trials; |
| • | the cost and timing of regulatory filings and/or approvals to commercialize our product candidates and any related restrictions, limitations, and/or warnings in the label of an approved product candidate; |
| • | the cost and timing of establishing medical education, sales, marketing and distribution capabilities; |
| • | the cost of establishing clinical and commercial supplies of our product candidates and any products that we and/or any potential future collaborators may develop; |
| • | the effect of competing technological and market developments; and |
| • | the cost of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights, and the cost of defending any other litigation claims. |
Any failure to complete the development of our product candidates in a timely manner could have a material adverse effect on our operations, financial position and liquidity. A discussion of the risks and uncertainties associated with completing our development and commercialization efforts on schedule, or at all, and some consequences of failing to do so, are set forth under Part I, Item 1A “Risk Factors” of this Annual Report on Form 10-K.
42
Table of Contents
Product Sales
We sell FARESTON® (toremifene citrate) 60 mg tablets, approved for the treatment of advanced metastatic breast cancer in postmenopausal women, in the United States. Effective June 1, 2011, we no longer utilize a sales force for FARESTON® promotional efforts, which has the potential to result in a decline in sales volume in future periods. In order to commercialize any future products, we will need to develop our sales and marketing capabilities or establish and maintain agreements with third parties to market and sell our product candidates.
General and Administrative Expenses
Our general and administrative expenses consist primarily of salaries and other related costs for personnel serving executive, finance, legal, human resources, information technology, investor relations, and marketing functions. General and administrative expenses also include facility costs, insurance costs, professional fees for legal, accounting, public relations, and marketing services, and FARESTON® selling and distribution expenses. We expect our general and administrative expenses for fiscal year 2012 to be less than fiscal year 2011, primarily due to the reduction in our workforce that occurred in June 2011 and decreased FARESTON® promotional efforts.
Critical Accounting Policies and Significant Judgments and Estimates
Our management’s discussion and analysis of our financial condition and results of operations is based on our financial statements, which have been prepared in accordance with accounting principles generally accepted in the United States of America. The preparation of these financial statements requires us to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements as well as the reported revenues and expenses during the reporting periods. On an ongoing basis, we evaluate our estimates and judgments related to revenue recognition, income taxes, intangible assets, long-term service contracts, share-based compensation, and other contingencies. We base our estimates on historical experience and on various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions.
While our significant accounting policies are more fully described in Note 2 to our financial statements appearing at the end of this Annual Report on Form 10-K, we believe that the following accounting policies are most critical to aid you in fully understanding and evaluating our reported financial results.
Revenue Recognition
Our revenues consist of product sales of FARESTON® and in 2011 and prior years, also consisted of revenues derived from our former collaboration and license agreements.
Collaboration revenue consisted of non-refundable upfront payments, license fees, reimbursements for research and development activities, and milestone payments associated with our former collaboration and license agreements and was based on the performance requirements of the specific agreements. We analyzed our agreements with multiple element arrangements to determine whether the deliverables under the agreement, including license and performance obligations such as joint steering committee participation and research and development activities, could have been separated or whether all of the deliverables must have been accounted for as a single unit of accounting. Revenues from milestone payments for which we had no continuing performance obligations were recognized upon achievement of the performance milestone, as defined in the related agreement, provided the milestone was substantive and a culmination of the earnings process had occurred. Performance obligations typically consisted of significant milestones in the development life cycle of the related product candidates and technology, such as initiation of clinical trials, achievement of specified clinical trial endpoints, filing for approval with regulatory agencies and approvals by regulatory agencies.
We recognize revenue from product sales of FARESTON® less deductions for estimated sales discounts and sales returns. We recognize revenue from product sales when persuasive evidence of an arrangement exists, title passes, the price is fixed or determinable, and collectability is reasonably assured. We account for rebates to certain governmental agencies as a reduction of product sales. We allow customers to return product within a specified
43
Table of Contents
time period prior to and subsequent to the product’s labeled expiration date. As a result, we estimate an accrual for product returns, which is recorded as a reduction of product sales. We consider historical product return trend information that we continue to update each period. We estimate the number of months of product on hand and the amount of product which is expected to exceed its expiration date and be returned by the customer by receiving information from our three largest wholesale customers about the levels of FARESTON® inventory held by these customers. These three largest wholesale customers accounted for 95% of our product sales of FARESTON® for the year ended December 31, 2011. Based on this information, and other factors, we estimate an accrual for product returns. At December 31, 2011 and 2010, our accrual for product returns was $1.1 million and $802,000, respectively.
Research and Development Expenses
Research and development expenses include, but are not limited to, our expenses for personnel, supplies, and facilities associated with research activities, screening and identification of product candidates, formulation and synthesis activities, manufacturing, preclinical studies, toxicology studies, clinical trials, regulatory and medical affairs activities, quality assurance activities and license fees. We expense these costs in the period in which they are incurred. We estimate our liabilities for research and development expenses in order to match the recognition of expenses to the period in which the actual services are received. As such, accrued liabilities related to third party research and development activities are recognized based upon our estimate of services received and degree of completion of the services in accordance with the specific third party contract.
Research and development expenses for the year ended December 31, 2011 included an impairment charge of $1.6 million related to the unamortized portion of capitalized license fees paid to Orion Corporation, or Orion, related to our former toremifene 80 mg program. Research and development expenses for the year ended December 31, 2010 included an impairment charge of $1.7 million related to the unamortized portion of capitalized license fees paid to Orion and the University of Tennessee Research Foundation related to our former toremifene 20 mg program.
Share-Based Compensation
We have stock option and equity incentive plans that provide for the purchase of our common stock by certain of our employees and non-employee directors. We recognize compensation expense for our share-based payments based on the fair value of the awards on the grant date and recognize the expense over the period during which an employee or non-employee director is required to provide service in exchange for the award.
The determination of the fair value of share-based payment awards on the date of grant include the expected life of the award, the expected stock price volatility over the expected life of the awards, expected dividend yield, and risk-free interest rate. We estimate the expected life of options by calculating the average of the vesting term and contractual term of the options. We estimate the expected stock price volatility based on the historical volatility of our common stock. The risk-free interest rate is determined using U.S. Treasury rates where the term is consistent with the expected life of the stock options. Expected dividend yield is not considered as we have not made any dividend payments and have no plans of doing so in the foreseeable future. The amount of share-based compensation expense recognized is reduced ratably over the vesting period by an estimate of the percentage of options granted that are expected to be forfeited or canceled before becoming fully vested. This estimate is adjusted periodically based on the extent to which actual forfeitures differ, or are expected to differ, from the previous estimate.
Total share-based compensation expense for the year ended December 31, 2011 was $4.4 million, of which approximately $2.0 million and approximately $2.4 million were recorded in the statements of operations as research and development expenses and general and administrative expenses, respectively. Total share-based compensation expense for the years ended December 31, 2010 and 2009 was $4.9 million and $5.4 million, respectively. Included in share-based compensation expense for all periods presented is share-based compensation expense related to deferred compensation arrangements for our non-employee directors, which was $178,000, $187,000 and $178,000 for the years ended December 31, 2011, 2010 and 2009, respectively. At December 31, 2011, the total compensation cost related to non-vested awards not yet recognized was approximately $6.3 million with a weighted average expense recognition period of 2.78 years.
44
Table of Contents
Income Taxes
We account for deferred taxes by recognition of deferred tax assets and liabilities for the expected future tax consequences of events that have been included in the financial statements or tax returns. Under this method, deferred tax assets and liabilities are determined based on the difference between the financial statement and tax basis of assets and liabilities using enacted tax rates in effect for the year in which the differences are expected to reverse. A valuation allowance is provided when it is more likely than not that some portion or all of the deferred tax assets will not be realized. Accordingly, at December 31, 2011 and 2010, net of the valuation allowance, the net deferred tax assets were reduced to zero.
Intangible Assets
We amortize our purchased intangible assets with finite lives over their estimated economic lives. We review long-lived assets for impairment whenever events or changes in facts and circumstances, both internally and externally, may indicate that an impairment of long-lived assets held for use are present. An impairment loss is recognized when estimated future cash flows is less than the carrying amount. The cash flow estimates are based on management’s best estimates, using appropriate and customary assumptions and projections at the time.
Based upon our decision to discontinue toremifene 80 mg development and after analyzing future cash flows and estimates of fair market value from a market participant perspective, we determined that our toremifene 80 mg intangible asset was impaired and recorded an impairment charge of $1,598 during the first quarter of 2011. The impaired intangible asset consisted of capitalized license fees paid to Orion related to our toremifene 80 mg program. The impairment charge was included in research and development expenses in the statement of operations for the year ended December 31, 2011.
In the second quarter of 2010, we discontinued our toremifene 20 mg development program and, as a result, recorded an impairment charge of $1,687. The impaired intangible assets consisted of capitalized license fees related to our toremifene 20 mg program paid to Orion and the University of Tennessee Research Foundation. The impairment charge was included in research and development expenses in the statement of operations for the year ended December 31, 2010.
Recent Accounting Pronouncements
In May 2011, the Financial Accounting Standards Board issued Accounting Standard Update 2011-04, Amendments to Achieve Common Fair Value Measurement and Disclosure Requirements in U.S. GAAP and IFRS. The new guidance results in a consistent definition of fair value and common requirements for measurement of and disclosure about fair value between U.S. GAAP and International Financial Reporting Standards. While many of the amendments to U.S. GAAP are not expected to have a significant effect on practice, the new guidance changes some fair value measurement principles and disclosure requirements. This new guidance is effective for fiscal years and interim periods beginning after December 15, 2011. We do not expect the adoption of the standard update to have a significant impact on our financial position or results of operations.
Results of Operations
Comparison of Years Ended December 31, 2011 and December 31, 2010
Revenues. Revenues for the year ended December 31, 2011 were $14.7 million as compared to $60.6 million for the same period of 2010. Revenues for the year ended December 31, 2011 included net product sales of FARESTON® and collaboration revenue from Ipsen. During the years ended December 31, 2011 and 2010, FARESTON® net product sales were $6.7 million and $3.8 million, respectively, while cost of products sales were $1.1 million and $768,000, respectively. FARESTON® net product sales for the year ended December 31, 2011 increased from the same period in the prior year due primarily to an increase of 35% in the sales price of FARESTON® and, to a lesser extent, an increase of 25% in sales volume. Collaboration revenue was $8.1 million for the year ended December 31, 2011, which resulted from recognition of all remaining unamortized deferred revenue due to the termination of our license and collaboration agreement with Ipsen in March 2011. Collaboration revenue for the year ended December 31, 2010 was $56.8 million, which consisted of approximately $1.9 million from Ipsen and $54.9 million from Merck resulting from the termination of our license and collaboration agreement with Merck & Co., or Merck, in March 2010.
45
Table of Contents
Research and Development Expenses. Research and development expenses increased 12% to $31.9 million for the year ended December 31, 2011 from $28.5 million for the year ended December 31, 2010. The increase in research and development expenses during the year ended December 31, 2011 compared to the year ended December 31, 2010 was due primarily to the initiation of the two Phase III clinical trials for enobosarm and the initiation of three Phase II clinical trials for Capesaris® in 2011. This was partially offset by a decrease in research and development expenses due to the discontinuance of our toremifene 80 mg and toremifene 20 mg clinical development programs.
The following table identifies the research and development expenses for each of our clinical product candidates, as well as research and development expenses pertaining to our other research and development efforts, for each of the periods presented. “Other research and development” expenses in the table below include the cost of our former toremifene 80 mg and toremifene 20 mg clinical programs, as well as the cost of personnel, supplies and facilities associated with preclinical and discovery research and development activities. Additionally, “Other research and development” includes impairment charges of $1.6 million related to our toremifene 80 mg intangible asset and $1.7 million related to our toremifene 20 mg intangible asset for the year ended December 31, 2011 and 2010, respectively. Research and development spending for past periods is not indicative of spending in future periods.
| Product Candidate/ | Years
Ended December 31, |
Increase/ (Decrease) |
||||||||||||
| Proposed Indication |
Program |
2011 | 2010 | |||||||||||
| (in thousands) | ||||||||||||||
| Enobosarm |
||||||||||||||
| Prevention and treatment of muscle wasting in patients with non-small cell lung cancer |
SARM | $ | 10,474 | $ | 3,207 | $ | 7,267 | |||||||
|
Capesaris® |
||||||||||||||
| First and second line hormonal treatment of advanced prostate cancer1 | Selective ER alpha agonist |
12,052 | 8,647 | 3,405 | ||||||||||
| Other research and development2 |
9,412 | 16,641 | (7,229 | ) | ||||||||||
|
|
|
|
|
|
|
|||||||||
| Total research and development expenses |
$ | 31,938 | $ | 28,495 | $ | 3,443 | ||||||||
|
|
|
|
|
|
|
|||||||||
| 1 | The FDA has placed a clinical hold on our clinical trials for Capesaris®. We believe there may be a path forward to develop Capesaris® at lower doses to treat men with metastatic hormone sensitive prostate cancer and men with CRPC. |
| 2 | “Other research and development” includes research and development expenses of $2.0 million and $4.3 million for the years ended December 31, 2011 and 2010, respectively, for toremifene 80 mg, and $486,000 and $5.1 million for the years ended December 31, 2011 and 2010, respectively, for toremifene 20 mg. |
General and Administrative Expenses. General and administrative expenses decreased 11% to $15.4 million for the year ended December 31, 2011 from $17.4 million for the year ended December 31, 2010. This decrease was primarily due to reduced marketing, insurance, legal and occupancy expenses of $842,000, $360,000, $280,000, and $240,000, respectively.
Other Income, Net. Other income, net decreased to $398,000 for the year ended December 31, 2011 from $1.4 million for the year ended December 31, 2010. For the year ended December 31, 2010, other income, net included income from grants totaling $1.2 million awarded to us by the United States Government under the Qualifying Therapeutic Discovery Project Program, which was established under Patient Protection and Affordable Care Act. We were granted $244,000 for each of five applications submitted for our cancer and cancer supportive care research and development programs.
46
Table of Contents
Comparison of Years Ended December 31, 2010 and December 31, 2009
Revenues. Revenues for the year ended December 31, 2010 were $60.6 million as compared to $14.7 million for the same period of 2009. Revenues for the year ended December 31, 2010 included net product sales of FARESTON® and collaboration revenue from Ipsen and Merck. During the years ended December 31, 2010 and 2009, FARESTON® net product sales were $3.8 million and $3.3 million, respectively, while cost of products sales were $768,000 and $1.3 million, respectively. FARESTON® net product sales for the year ended December 31, 2010 increased from the same period in the prior year due primarily to an increase in the sales price of FARESTON® taken during the second quarter of 2010 and, to a lesser extent, an approximately 16% increase in sales volume. These increases were partially offset by an increase in the provision for product returns due to an increase in sales price and increases in governmental rebates. Cost of product sales decreased from the prior year due to a reduction in the royalty payable to Orion on our net sales of FARESTON® in the third quarter of 2009. Collaboration revenue was $56.8 million for the year ended December 31, 2010, which consisted of approximately $1.9 million from Ipsen and $54.9 million from Merck. As a result of the termination of our license and collaboration agreement with Merck in March 2010, we recognized as collaboration revenue the remaining $49.9 million of unamortized deferred revenue in the first quarter of 2010, as well as the final payment of $5.0 million of cost reimbursement that was received from Merck in December 2010. For the year ended December 31, 2009, collaboration revenue was $11.4 million, of which $5.8 million and $5.6 million was from the amortization of deferred revenue from Ipsen and Merck, respectively.
Research and Development Expenses. Research and development expenses decreased 12% to $28.5 million for the year ended December 31, 2010 from $32.3 million for the year ended December 31, 2009. The decrease in research and development expenses during the year ended December 31, 2010 compared to the year ended December 31, 2009 was due primarily to the completion of the toremifene 20 mg Phase III clinical trial in early 2010, the completion of two Phase I clinical trials for Capesaris® during 2009, and a decrease in discovery research and development activities in 2010. The decrease was partially offset by a Phase II clinical trial for Capesaris ® conducted in 2010 and increased spending on enobosarm development. Additionally, research and development expenses for the year ended December 31, 2010 included an impairment charge of $1.7 million related to our toremifene 20 mg intangible assets.
The following table identifies the research and development expenses for each of our clinical product candidates, as well as research and development expenses pertaining to our other research and development efforts, for each of the periods presented. “Other research and development” expenses in the table below include the cost of our former toremifene 80 mg and toremifene 20 mg clinical programs, as well as the cost of personnel, supplies and facilities associated with preclinical and discovery research and development activities. Research and development spending for past periods is not indicative of spending in future periods.
| Product Candidate/ | Years
Ended December 31, |
Increase/ | ||||||||||||
| Proposed Indication |
Program |
2010 | 2009 | (Decrease) | ||||||||||
| (in thousands) | ||||||||||||||
| Enobosarm |
||||||||||||||
| Prevention and treatment of muscle wasting in patients with cancer | SARM | $ | 3,207 | $ | 679 | $ | 2,528 | |||||||
|
Capesaris® |
||||||||||||||
| First line treatment of advanced prostate cancer1 | Selective ER alpha agonist |
8,647 | 10,074 | (1,427 | ) | |||||||||
| Other research and development2 |
16,641 | 21,591 | (4,950 | ) | ||||||||||
|
|
|
|
|
|
|
|||||||||
| Total research and development expenses |
$ | 28,495 | $ | 32,344 | $ | (3,849 | ) | |||||||
|
|
|
|
|
|
|
|||||||||
| 1 | The FDA has placed a clinical hold on our clinical trials for Capesaris®. We believe there may be a path forward to develop Capesaris® at lower doses to treat men with metastatic hormone sensitive prostate cancer and men with CRPC. |
| 2 | “Other research and development” includes research and development expenses of $4.3 million and $3.4 million for the years ended December 31, 2010 and 2009, respectively, for toremifene 80 mg, and $5.1 million and $5.9 million for the years ended December 31, 2010 and 2009, respectively, for toremifene 20 mg. |
47
Table of Contents
General and Administrative Expenses. General and administrative expenses decreased 37% to $17.4 million for the year ended December 31, 2010 from $27.8 million for the year ended December 31, 2009. This decrease was primarily due to decreased personnel related expenses of $5.8 million resulting from the reduction in our workforce in December 2009, reduced marketing expenses of $2.7 million, and reduced medical education expenses of $693,000, in each case, as a result of our not receiving regulatory approval of our toremifene 80 mg product candidate.
Other Income, Net. Other income, net increased to $1.4 million for the year ended December 31, 2010 from $188,000 for the year ended December 31, 2009. For the year ended December 31, 2010, other income, net included income from grants totaling $1.2 million awarded to us by the United States Government under the Qualifying Therapeutic Discovery Project Program.
Liquidity and Capital Resources
Through December 31, 2011, we have financed our operations primarily through public offerings and private placements of our common stock, as well as payments from our former collaborators. We have incurred significant losses since our inception in 1997 as we have devoted substantially all of our resources to research and development, including our clinical trials. As of December 31, 2011, we had an accumulated deficit of $386.2 million, which resulted primarily from:
| • | our research and development activities associated with: |
| • | the preclinical and clinical development of our SARM compounds, including enobosarm for the prevention and treatment of muscle wasting in patients with cancer; |
| • | the preclinical and clinical development of Capesaris® for first and second line hormonal treatment of advanced prostate cancer, which has been placed on clinical hold by the FDA; |
| • | the development of toremifene 80 mg to reduce fractures and treat other estrogen deficiency side effects of androgen deprivation therapy in men with prostate cancer, including two Phase II clinical trials, a pivotal Phase III clinical trial, and the preparation and submission of a NDA to the FDA; |
| • | the development of toremifene 20 mg for the prevention of prostate cancer in high risk men with high grade prostatic intraepithelial neoplasia, including a Phase IIb clinical trial and a Phase III clinical trial; |
| • | the preclinical development of other product candidates; and |
| • | general and administrative expense. |
We expect to incur significant operating losses in 2012 and for the foreseeable future as we continue our clinical development and research and development activities. We do not expect to obtain FDA or any other regulatory approvals to market any of our product candidates in the near future.
At December 31, 2011, we had cash, cash equivalents and short-term investments of $74.4 million, compared to $58.6 million at December 31, 2010 and $49.0 million at December 31, 2009. As of December 31, 2011, our cash and cash equivalents consisted of bank deposits, certificates of deposit with original maturities of 90 days or less, and money market mutual funds which are required to comply with Rule 2a-7 under the Investment Company Act of 1940. Our short-term investments consisted of investments in certificates of deposit with original maturities greater than three months and less than one year.
In June 2011, we completed an underwritten public offering of 11,023,000 shares of our common stock at a price to the public of $4.75 per share. Net cash proceeds from the public offering were approximately $49.0 million, after deducting the underwriting discount and offering expenses.
48
Table of Contents
In November 2010, we completed an underwritten public offering of 15,285,715 shares of our common stock at a price to the public of $2.80 per share. Net cash proceeds from the public offering were approximately $40.3 million after deducting underwriting discounts and commissions and other offering expenses.
In September 2006, we entered into a collaboration and license agreement with Ipsen, under which Ipsen paid us €21.5 million (approximately $27.1 million) as a license fee and expense reimbursement and paid us €1.5 million in equal installments over a three year period from the date of the agreement. In September of 2009, 2008, and 2007, we received €500,000 (approximately $726,000, $711,000, and $688,000, respectively) from Ipsen for the three annual installment payments. In February 2008, we earned a milestone of €1.0 million (approximately $1.5 million) with the achievement of the primary endpoint in the toremifene 80 mg Phase III clinical trial. As a result of the termination of our collaboration with Ipsen in March of 2011, we will not receive any future milestone payments, royalties or reimbursement of clinical development expenses for the development or sale of toremifene from Ipsen provided for under our former collaboration with Ipsen.
In December 2007, we entered into an exclusive license and collaboration agreement with Merck, or the Merck Collaboration Agreement, which was terminated in March 2010. In connection with entering into this agreement, we received an upfront licensing fee of $40.0 million in January 2008, and Merck purchased approximately $30.0 million of our common stock in December 2007. Merck also paid us $15.0 million in guaranteed cost reimbursements for research and development activities in equal annual installments over a three year period beginning on the first anniversary of the effective date of the agreement. We received the three annual payments of $5 million from Merck in December 2010, 2009, and 2008, respectively. As a result of the termination of our collaboration with Merck, we will not receive any milestone payments or royalties for the development or sale of SARMs from Merck provided for under our former collaboration with Merck. We have no ongoing collaborations for the development and commercialization of our product candidates, and we do not currently have any commitments for future external funding.
Net cash used in operating activities was $33.1 million, $30.5 million and $46.0 million for the years ended December 31, 2011, 2010 and 2009, respectively. The use of cash in all periods resulted primarily from funding our operations. In 2010, this was reduced by $5.0 million received from Merck related to the third and final cost reimbursement payment in conjunction with our exclusive license and collaboration agreement and grants totaling $1.2 million received from the United States government under the Qualifying Therapeutic Discovery Project Program. In 2009, the cash used in operating activities was partially offset by approximately $726,000 related to the third and final annual license fee and expense reimbursement installment payment from Ipsen in conjunction with our collaboration and license agreement, $5.0 million from Merck related to the second annual installment cost reimbursement payment in conjunction with our exclusive license and collaboration agreement, and approximately $2.3 million in distributions from our investment in Bank of America Corporation’s Columbia Strategic Cash Portfolio.
Net cash used in investing activities for the year ended December 31, 2011 was $10.3 million and resulted from the purchase of short-term investments of $15.1 million and the purchase of information technology equipment and research and development equipment of approximately $54,000, offset by the maturities of short-term investments of $4.9 million. Net cash provided by investing activities for the year ended December 31, 2010 was $8.3 million and resulted from the maturities of short-term investments of $16.9 million, offset by the purchase of short-term investments of $8.6 million and the purchase of information technology equipment and research and development equipment of approximately $95,000. Net cash used in investing activities for the year ended December 31, 2009 was $9.4 million and was for the purchase of short-term investments of approximately $11.3 million and the purchase of information technology equipment, research and development equipment, and software totaling approximately $600,000. This was reduced by the maturities of short-term investments of approximately $2.5 million.
Net cash provided by financing activities was $49.0 million, $40.2 million and $131,000 for the years ended December 31, 2011, 2010 and 2009, respectively. Net cash provided by financing activities for the year ended December 31, 2011 and 2010 reflected proceeds from our underwritten public offering of common stock in June 2011 and November 2010, respectively, and proceeds from the exercise of employee stock options. These proceeds were reduced by payments on our capital lease and financed equipment obligations. Net cash provided by financing activities for the year ended December 31, 2009 was provided primarily from proceeds from the exercises of employee stock options.
49
Table of Contents
As of the date of this Annual Report on Form 10-K, we estimate that our current cash, cash equivalents, and short-term investments, together with interest income and net product sales of FARESTON®, will be sufficient to meet our projected operating requirements for at least the next twelve months. We have based this estimate on our current business plan and assumptions that may prove to be wrong. We could utilize our available capital resources sooner than we currently expect, and we could need additional funding sooner than currently anticipated. In any event, to complete the development of and seek regulatory approval for enobosarm and, if development is permitted to continue, Capesaris®, we will need to obtain substantial additional funding.
Our estimate of the period of time through which our financial resources will be adequate to support our projected operating requirements is a forward-looking statement and involves risks and uncertainties, and actual results could vary as a result of a number of factors, including the factors discussed under Part II, Item 1A “Risk Factors” section of this Annual Report on Form 10-K. Because of the numerous risks and uncertainties associated with the development and potential commercialization of our product candidates and other research and development activities, including risks and uncertainties that could impact the rate of progress of our development and commercialization activities, we are unable to estimate with certainty the amounts of increased capital outlays and operating expenditures associated with our anticipated future clinical trials, other research and development activities, and potential commercialization activities. Our future funding requirements will depend on many factors, including:
| • | the scope, rate of progress and cost of our clinical trials and other research and development activities, including additional development activities or studies that may be required by the FDA if we are permitted to continue developing Capesaris®, as well as our ongoing and any future clinical trials of enobosarm; |
| • | the terms and timing of any potential future collaborative, licensing and other arrangements that we may establish; |
| • | the amount and timing of any licensing fees, milestone payments and royalty payments from potential future collaborators, if any; |
| • | future clinical trial results; |
| • | the cost and timing of regulatory filings and/or approvals to commercialize our product candidates and any related restrictions, limitations, and/or warnings in the label of an approved product candidate; |
| • | the cost and timing of establishing medical education, sales, marketing and distribution capabilities; |
| • | the cost of establishing clinical and commercial supplies of our product candidates and any products that we and/or any potential future collaborators may develop; |
| • | the effect of competing technological and market developments; |
| • | the cost of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights, and the cost of defending any other litigation claims; and |
| • | the extent to which we acquire or invest in businesses, products and technologies, although we currently have no commitments or agreements relating to any of these types of transactions. |
We do not currently have any commitments for future external funding and until we can generate a sufficient amount of product revenue, which we may never do, we expect to finance future cash needs through public or private equity offerings, debt financings or collaboration and licensing arrangements, or a combination of the above, as well as through interest income earned on the investment of our cash balances and short-term investments and revenues from the sale of FARESTON®. In June 2011, we announced a workforce reduction of approximately 15% in order to reduce our operating expenses relating to our discontinued toremifene development programs. If we are unable to raise additional funds when needed, we may need to further reduce our expenditures, perhaps significantly, to preserve our cash. Cost-cutting measures that we may take in the future may not be sufficient to enable us to meet our cash requirements, and they may negatively affect our business and growth prospects.
To the extent we raise additional funds by issuing equity securities, our stockholders may experience dilution, and debt financing, if available, may involve restrictive covenants. Any debt financing or additional equity that we raise may contain terms that are not favorable to us or our stockholders. To the extent we raise additional funds through potential future collaboration and licensing arrangements, it may be necessary to relinquish rights to some of our technologies or product candidates, or grant licenses on terms that are not favorable to us. Our ability to raise additional funds and the terms upon which we are able to raise such funds may be adversely impacted by the clinical hold placed by the FDA on our three Phase II clinical trials evaluating Capesaris® and the related uncertainty regarding the continued development of Capesaris®, as well as uncertainty regarding our financial condition, the
50
Table of Contents
outcomes of our ongoing clinical trials of enobosarm and/or current economic conditions, including the effects of disruptions to and volatility in the credit and financial markets in the United States, the European Union and other regions of the world, including those resulting from or associated with rising government debt levels. As a result of these and other factors, we cannot be certain that additional funding will be available on acceptable terms, or at all. If adequate funds are not available when we need them, we may be required to delay, reduce the scope of or eliminate one or more of our research or development programs, including our SARM and selective ER alpha agonist programs, or conduct additional workforce or other expense reductions, any of which could have a material adverse effect on our business.
At December 31, 2011, we had contractual obligations as follows:
| Payment Due by Period (in thousands) |
||||||||||||||||||||
| Total | Less than 1 year |
1-3 years | 4-5 years | More than 5 years |
||||||||||||||||
| Capital lease obligations |
$ | 16 | $ | 7 | $ | 9 | $ | — | $ | — | ||||||||||
| Operating lease obligations |
2,209 | 933 | 1,092 | 184 | — | |||||||||||||||
| Equipment financing obligation |
78 | 78 | — | — | — | |||||||||||||||
| Purchase obligations |
12 | 12 | — | — | — | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Total |
$ | 2,315 | $ | 1,030 | $ | 1,101 | $ | 184 | $ | — | ||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
Our long-term commitments under the operating leases shown above consist of payments relating to a sublease for laboratory and office space at 3 North Dunlap Street, Memphis, Tennessee and a sublease for office space at 175 Toyota Plaza, Memphis, Tennessee. Our sublease agreement for the premises located at 3 North Dunlap Street expires on December 31, 2012, with an option to extend for two additional years. The sublease for the premises at 175 Toyota Plaza expires on April 30, 2015 and includes escalating rental payments. The table above excludes contingent payments under the license agreements to which we are a party because the amount and timing of such payments are unknown or uncertain.
| ITEM 7A. | QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK |
Our exposure to market risk for changes in interest rates relates to our cash equivalents on deposit in highly liquid money market funds and investments in certificates of deposit. The primary objective of our cash investment activities is to preserve principal while at the same time maximizing the income we receive from our invested cash without significantly increasing risk of loss. We do not use derivative financial instruments in our investment portfolio. The effect of a hypothetical decrease of ten percent in the average yield earned on our cash equivalents and short-term investments would have resulted in an immaterial decrease in our interest income for the year ended December 31, 2011.
In addition, we have exposure to fluctuations in certain foreign currencies in countries in which we conduct clinical trials. Most of our foreign expenses incurred are associated with conducting clinical trials for enobosarm at clinical trial sites outside of the United States. As a result, when the U.S. dollar rises and falls against the functional currencies of these other nations, our costs will either increase or decrease by the relative change in the exchange rate. Foreign currency gains and losses were not significant during the year ended December 31, 2011. Further, the effect of an immediate 10% change in foreign currency rates would have no material impact on our financial condition, results of operations or cash flows at December 31, 2011. We have elected not to hedge our exposure to foreign currency fluctuations.
| ITEM 8. | FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA |
Our financial statements and the reports of our independent registered public accounting firm are included in this Annual Report on Form 10-K beginning on page F-1. The index to these reports and our financial statements is included in Part IV, Item 15 below.
51
Table of Contents
| ITEM 9. | CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE |
Not applicable.
| ITEM 9A. | CONTROLS AND PROCEDURES |
Disclosure Controls and Procedures
We maintain disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) of the Securities Exchange Act of 1934, as amended (the “Exchange Act”)) that are designed to ensure that information required to be disclosed in the reports that we file or submit under the Exchange Act is recorded, processed, summarized, and reported within the time periods specified in the SEC’s rules and forms and that such information is accumulated and communicated to our management, including our chief executive officer and chief financial officer, as appropriate, to allow for timely decisions regarding required disclosures.
We have carried out an evaluation, under the supervision and with the participation of our management, including our chief executive officer and chief financial officer, of the effectiveness of the design and operation of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act) as of the end of the period covered by this report. Based on the evaluation of these disclosure controls and procedures, our chief executive officer and chief financial officer have concluded that our disclosure controls and procedures were effective.
Management’s Report on Internal Control Over Financial Reporting
We, as management of GTx, Inc., are responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Securities Exchange Act Rule 13a-15(f). Internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with United States generally accepted accounting principles. Any system of internal control, no matter how well designed, has inherent limitations, including the possibility that a control can be circumvented or overridden and misstatements due to error or fraud may occur and not be detected. Also, because of changes in conditions, internal control effectiveness may vary over time. Accordingly, even an effective system of internal control will provide only reasonable assurance that the objectives of the internal control system are met.
Under the supervision and with the participation of management, including our principal executive officer and principal financial officer, we conducted an evaluation of the effectiveness of our internal control over financial reporting as of December 31, 2011 using the criteria for effective internal control over financial reporting as described in “Internal Control – Integrated Framework,” issued by the Committee of Sponsoring Organizations of the Treadway Commission. Based on this evaluation, we concluded that, as of December 31, 2011, our internal control over financial reporting was effective. The effectiveness of our internal control over financial reporting has been audited by Ernst & Young LLP, independent registered public accounting firm.
Attestation Report of the Independent Registered Public Accounting Firm
Ernst & Young LLP, an independent registered public accounting firm, has issued an audit report on our internal control over financial reporting, which report is included elsewhere herein.
Changes in Internal Control Over Financial Reporting
There were no changes in our internal control over financial reporting during the fourth quarter of 2011 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
| ITEM 9B. | OTHER INFORMATION |
Not applicable.
52
Table of Contents
Certain information required by Part III is omitted from this Annual Report on Form 10-K because we will file our definitive proxy statement for our 2012 Annual Meeting of Stockholders with the U.S. Securities and Exchange Commission pursuant to Regulation 14A (the “2012 Proxy Statement”) not later than 120 days after the end of the fiscal year covered by this Annual Report on Form 10-K, and certain information included in the 2012 Proxy Statement is incorporated herein by reference.
| ITEM 10. | DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE |
(1) The information required by this Item concerning our directors and nominees for director, including information with respect to our audit committee and audit committee financial experts, may be found under the section entitled “Proposal No. 1 – Election of Directors” and “Additional Information About the Board of Directors and Certain Corporate Governance Matters” appearing in the 2012 Proxy Statement. Such information is incorporated herein by reference.
(2) The information required by this Item concerning compliance with Section 16(a) of the Securities Exchange Act of 1934 may be found in the section entitled “Section 16(a) Beneficial Ownership Reporting Compliance” appearing in the 2012 Proxy Statement. Such information is incorporated herein by reference.
(3) The information required by this Item concerning our executive officers is set forth in the section entitled “Management — Executive Officers of Registrant” in Part I, Item 1 of this Form 10-K.
(4) Our Board has adopted a Code of Business Conduct and Ethics applicable to all officers, directors and employees as well as Guidelines on Governance Issues. These documents are available on our website (www.gtxinc.com) under “About GTx” at “Governance.” We will provide a copy of these documents to any person, without charge, upon request, by writing to us at GTx, Inc., Director, Corporate Communications and Financial Analysis, 175 Toyota Plaza, Suite 700, Memphis, Tennessee 38103. We intend to satisfy the disclosure requirement under Item 5.05 of Form 8-K regarding an amendment to, or waiver from, a provision of the Code of Business Conduct and Ethics by posting such information on our website at the address and the location specified above.
| ITEM 11. | EXECUTIVE COMPENSATION |
(1) The information required by this Item concerning director and executive compensation is incorporated herein by reference to the information from the 2012 Proxy Statement under the sections entitled “Compensation Discussion and Analysis,” “Executive Compensation” and “Director Compensation.”
(2) The information required by this Item concerning Compensation Committee interlocks and insider participation is incorporated herein by reference to the information from the 2012 Proxy Statement under the section entitled “Compensation Committee Interlocks and Insider Participation.”
(3) The information required by this Item concerning our Compensation Committee’s review and discussion of our Compensation Discussion and Analysis is incorporated herein by reference to the information from the 2012 Proxy Statement under the section entitled “Compensation Committee Report.”
| ITEM 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS |
(1) The information required by this Item with respect to security ownership of certain beneficial owners and management is incorporated herein by reference to the information from the 2012 Proxy Statement under the section entitled “Security Ownership of Certain Beneficial Owners and Management.”
(2) The information required by this Item with respect to securities authorized for issuance under our equity compensation plans is incorporated herein by reference to the information from the 2012 Proxy Statement under the section entitled “Equity Compensation Plan Information.”
53
Table of Contents
| ITEM 13. | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE |
(1) The information required by this Item concerning related party transactions is incorporated herein by reference to the information from the 2012 Proxy Statement under the section entitled “Certain Relationships and Related Party Transactions.”
(2) The information required by this Item concerning director independence is incorporated herein by reference to the information from the 2012 Proxy Statement under the section entitled “Additional Information About the Board of Directors and Certain Corporate Governance Matters – Director Independence.”
| ITEM 14. | PRINCIPAL ACCOUNTING FEES AND SERVICES |
The information required by this Item is incorporated herein by reference to the information from the 2012 Proxy Statement under the section entitled “Proposal No. 2 – Ratification of Appointment of Independent Registered Public Accounting Firm.”
| ITEM 15. | EXHIBITS AND FINANCIAL STATEMENT SCHEDULES |
(a)(1) Index to Financial Statements
| Page |
Description | |
| F-2 | Management’s Report on Internal Control Over Financial Reporting | |
| F-3 | Reports of Independent Registered Public Accounting Firm | |
| F-5 | Balance Sheets at December 31, 2011 and 2010 | |
| F-6 | Statements of Operations for the Years Ended December 31, 2011, 2010 and 2009 | |
| F-7 | Statements of Stockholders’ Equity (Deficit) for the Years Ended December 31, 2011, 2010 and 2009 | |
| F-8 | Statements of Cash Flows for the Years Ended December 31, 2011, 2010 and 2009 | |
| F-9 | Notes to Financial Statements |
(a)(2) Financial statement schedules are omitted as they are not applicable.
(a)(3) See 15(b) below.
(b) Exhibits
| Number |
Description | |
| 3.1 | Restated Certificate of Incorporation of GTx, Inc. (1) | |
| 3.2 | Certificate of Amendment of Restated Certificate of Incorporation of GTx, Inc.(18) | |
| 3.3 | Amended and Restated Bylaws of GTx, Inc.(2) | |
| 4.1 | Reference is made to Exhibits 3.1, 3.2 and 3.3 | |
| 4.2 | Specimen of Common Stock Certificate(3) | |
| 4.3 | Amended and Restated Registration Rights Agreement between Registrant and Oracle Partners, L.P. dated August 7, 2003(3) | |
| 4.4* | Amended and Restated Registration Rights Agreement between Registrant and J. R. Hyde, III dated August 7, 2003(3) | |
| 4.5 | Consent, Waiver and Amendment between the Registrant and Oracle Partners, L.P., Oracle Investment Management, Inc. and Oracle Institutional Partners, L.P. dated November 29, 2007(4) | |
| 4.6* | Consent, Waiver and Amendment between Registrant and J. R. Hyde, III and Pittco Associates, L.P. dated December 3, 2007(4) | |
| 10.1* | Genotherapeutics, Inc. 1999 Stock Option Plan, as amended through December 10, 2009, and Form of Stock Option Agreement(5) | |
| 10.2* | GTx, Inc. 2000 Stock Option Plan, as amended through December 10, 2009, and Form of Stock Option Agreement(5) | |
54
Table of Contents
| Number |
Description | |
| 10.3* | GTx, Inc. 2001 Stock Option Plan, as amended through November 3, 2009, and Form of Stock Option Agreement(5) | |
| 10.4* | GTx, Inc. 2002 Stock Option Plan, as amended through November 3, 2009, and Form of Stock Option Agreement(5) | |
| 10.5* | GTx, Inc. 2004 Equity Incentive Plan and Form of Stock Option Agreement(3) | |
| 10.6* | GTx, Inc. 2004 Equity Incentive Plan, as amended effective April 30, 2008(6) | |
| 10.8* | Amended and Restated Employment Agreement dated November 10, 2008, between Registrant and Mitchell S. Steiner, M.D.(7) | |
| 10.9* | Amended and Restated Employment Agreement dated November 10, 2008, between Registrant and Marc S. Hanover(7) | |
| 10.10* | Amended and Restated Employment Agreement dated November 10, 2008, between Registrant and Mark E. Mosteller(7) | |
| 10.11* | Amended and Restated Employment Agreement dated November 10, 2008, between Registrant and Henry P. Doggrell(7) | |
| 10.12* | Form of Indemnification Agreement(3) | |
| 10.13 | Lease Agreement, dated March 7, 2001, between The University of Tennessee and TriStar Enterprises, Inc.(3) | |
| 10.14 | Sublease Agreement dated October 1, 2000, as amended, between Registrant and TriStar Enterprises, Inc.(3) | |
| 10.24† | Purchase Agreement dated December 13, 2004, between Registrant and Orion Corporation(10) | |
| 10.25† | Amended and Restated License and Supply Agreement effective January 1, 2005, between Registrant and Orion Corporation(11) | |
| 10.26 | Sublease Agreement dated April 1, 2005, as amended, between Registrant and TriStar Enterprises, Inc.(12) | |
| 10.27* | Amended and Restated Employment Agreement dated November 10, 2008 between Registrant and James T. Dalton(7) | |
| 10.29* | 2011 Compensation Information for Registrant’s Executive Officers (19) | |
| 10.30* | GTx, Inc. 2004 Non-Employee Directors’ Stock Option Plan and Form of Stock Option Agreement(9) | |
| 10.31* | Amended and Restated GTx, Inc. 2004 Non-Employee Directors’ Stock Option Plan, effective April 26, 2006(13) | |
| 10.32† | Amendment dated May 23, 2006 to the Amended and Restated License and Supply Agreement effective January 1, 2005, between Registrant and Orion Corporation(14) | |
| 10.33† | Amendment dated June 30, 2006 to the Amended and Restated License and Supply Agreement effective January 1, 2005, between Registrant and Orion Corporation(15) | |
| 10.34* | Form of Stock Option Agreement under the Amended and Restated GTx, Inc. 2004 Non-Employee Directors’ Stock Option Plan(16) | |
| 10.38* | Amended and Restated Employment Agreement dated November 10, 2008, between Registrant and Ronald A. Morton, Jr., M.D.(7) | |
| 10.40† | Consolidated, Amended, and Restated License Agreement dated July 24, 2007, between Registrant and University of Tennessee Research Foundation(8) | |
| 10.44* | Amended and Restated Employment Agreement, dated November 10, 2008, between Registrant and Gregory A. Deener(7) | |
| 10.46 | Sublease Agreement, dated December 17, 2007 by and between the Registrant and ESS SUSA Holdings, LLC(17) | |
| 10.47 | First Amendment, dated December 29, 2008, to the Consolidated, Amended and Restated License Agreement dated July 24, 2007 between the Registrant and University of Tennessee Research Foundation(7) | |
| 10.49* | Directors’ Deferred Compensation Plan, as amended effective November 4, 2008(7) | |
| 10.50* | Non-Employee Director Compensation Policy of GTx, Inc., effective February 18, 2011(19) | |
| 10.51* | Amended and Restated GTx, Inc. 2004 Non-Employee Directors’ Stock Option Plan, as amended effective November 4, 2008(7) | |
| 10.52* | GTx, Inc. 2004 Equity Incentive Plan, as amended effective November 4, 2008 and Form of Stock Option Agreement(7) | |
| 10.53* | Amended and Restated GTx, Inc. Executive Bonus Compensation Plan, effective November 4, 2008(7) | |
| 10.54 | First Amendment, dated July 21, 2008, to the Sublease and Parking Sublicense Agreements dated December 17, 2007 by and between the Registrant and ESS SUSA Holdings, LLC(7) | |
| 10.55 | Sublease Agreement dated October 1, 2009 between Registrant and University of Tennessee Research Foundation(5) | |
| 10.57 | Second Amendment to Sublease and Parking Sublicense Agreements dated January 1, 2011 by and between the Registrant and ESS SUSA Holdings, LLC(20) | |
55
Table of Contents
| Number |
Description | |
| 10.59 | Memorandum of Understanding Concerning the Lease Agreement between The University of Tennessee Research Foundation and the Registrant as Amended July 20, 2009(21) | |
| 10.60*+ | 2012 Compensation Information for Registrant’s Executive Officers | |
| 10.61*+ | Confidential Separation Agreement and General Release between the Registrant and Gregory A. Deener | |
| 23.1+ | Consent of Independent Registered Public Accounting Firm | |
| 24.1+ | Power of Attorney (included on the signature pages hereto) | |
| 31.1+ | Certification of Chief Executive Officer, as required by Rule 13a-14(a) or Rule 15d-14(a) | |
| 31.2+ | Certification of Chief Financial Officer, as required by Rule 13a-14(a) or Rule 15d-14(a) | |
| 32.1+ | Certification of Chief Executive Officer, as required by Rule 13a-14(b) or Rule 15d-14(b) and Section 1350 of Chapter 63 of Title 18 of the United States Code (18 U.S.C. §1350)(22) | |
| 32.2+ | Certification of Chief Financial Officer, as required by Rule 13a-14(b) or Rule 15d-14(b) and Section 1350 of Chapter 63 of Title 18 of the United States Code (18 U.S.C. §1350)(22) | |
| 101.INS+ | XBRL Instance Document(23) | |
| 101.SCH+ | XBRL Taxonomy Extension Schema Document(23) | |
| 101.CAL+ | XBRL Taxonomy Extension Calculation Linkbase Document(23) | |
| 101.LAB+ | XBRL Taxonomy Extension Labels Linkbase Document(23) | |
| 101.PRE+ | XBRL Taxonomy Extension Presentation Linkbase Document(23) | |
| † | Confidential treatment has been granted with respect to certain portions of this exhibit. This exhibit omits the information subject to this confidentiality request. Omitted portions have been filed separately with the SEC. |
| * | Indicates a management contract or compensation plan or arrangement. |
| + | Filed herewith |
| (1) | Filed as Exhibit 4.1 to the Registrant’s registration statement on Form S-3 (File No. 333-127175), filed with the SEC on August 4, 2005, and incorporated herein by reference. |
| (2) | Filed as Exhibit 3.2 to the Registrant’s Current Report on Form 8-K (File No. 000-50549), filed with the SEC on July 26, 2007 and incorporated herein by reference. |
| (3) | Filed as the like numbered Exhibit to the Registrant’s registration statement on Form S-1 (File No. 333-109700), filed with the SEC on October 15, 2003, as amended, and incorporated herein by reference. |
| (4) | Filed as the like numbered Exhibit to the Registrant’s registration statement on Form S-3 (File No. 333-148321), filed with the SEC on December 26, 2007, and incorporated herein by reference. |
| (5) | Filed as the like numbered Exhibit to the Registrant’s Annual Report on Form 10-K (File No. 000-50549), filed with the SEC on March 15, 2010, and incorporated herein by reference. |
| (6) | Filed as the like numbered Exhibit to the Registrant’s Current Report on Form 8-K (File No. 000-50549), filed with the SEC on May 6, 2008, and incorporated herein by reference. |
| (7) | Filed as the like numbered Exhibit to the Registrant’s Annual Report on Form 10-K (File No. 000-50549), filed with the SEC on March 3, 2009, and incorporated herein by reference. |
| (8) | Filed as the like numbered Exhibit to the Registrant’s Quarterly Report on Form 10-Q (File No. 000-50549), filed with the SEC on November 9, 2007, and incorporated herein by reference. |
| (9) | Filed as Exhibit 10.6 to the Registrant’s registration statement on Form S-1 (File No. 333-109700), filed with the SEC on October 15, 2003, as amended, and incorporated herein by reference. |
56
Table of Contents
| (10) | Filed as Exhibit 10.1 to the Registrant’s Current Report on Form 8-K/A (File No. 000-50549), filed with the SEC on March 7, 2005, and incorporated herein by reference. |
| (11) | Filed as Exhibit 10.2 to the Registrant’s Current Report on Form 8-K/A (File No. 000-50549), filed with the SEC on March 7, 2005, and incorporated herein by reference. |
| (12) | Filed as Exhibit 10.27 to the Registrant’s Quarterly Report on Form 10-Q (File No. 000-50549), filed with the SEC on July 27, 2005, and incorporated herein by reference. |
| (13) | Filed as Exhibit 10.1 to the Registrant’s Current Report on Form 8-K (File No. 000-50549), filed with the SEC on April 27, 2006, and incorporated herein by reference. |
| (14) | Filed as Exhibit 10.33 to the Registrant’s Quarterly Report on Form 10-Q (File No. 000-50549), filed with the SEC on August 9, 2006, and incorporated herein by reference. |
| (15) | Filed as Exhibit 10.34 to the Registrant’s Quarterly Report on Form 10-Q (File No. 000-50549), filed with the SEC on August 9, 2006, and incorporated herein by reference. |
| (16) | Filed as Exhibit 10.35 to the Registrant’s Quarterly Report on Form 10-Q (File No. 000-50549), filed with the SEC on August 9, 2006, and incorporated herein by reference. |
| (17) | Filed as the like numbered Exhibit to the Registrant’s Annual Report on Form 10-K (File No. 000-50549), filed with the SEC on March 11, 2008, and incorporated herein by reference. |
| (18) | Filed as the like numbered Exhibit to the Registrant’s Current Report on Form 8-K (File No. 000-50549), filed with the SEC on May 6, 2011, and incorporated herein by reference. |
| (19) | Filed as the like numbered Exhibit to the Registrant’s Quarterly Report on Form 10-Q (File No. 000-50549), filed with the SEC on May 9, 2011, and incorporated herein by reference. |
| (20) | Filed as the like numbered Exhibit to the Registrant’s Annual Report on Form 10-K (File No. 000-50549), filed with the SEC on March 8, 2011, and incorporated herein by reference. |
| (21) | Filed as the like numbered Exhibit to the Registrant’s Quarterly Report on Form 10-Q (File No. 000-50549), filed with the SEC on August 9, 2011, and incorporated herein by reference. |
| (22) | This certification accompanies the Form 10-K to which it relates, is not deemed filed with the Securities and Exchange Commission and is not to be incorporated by reference into any filing of the Registrant under the Securities Act of 1933, as amended, or the Securities Exchange Act of 1934, as amended (whether made before or after the date of the Form 10-K), irrespective of any general incorporation language contained in such filing. |
| (23) | Pursuant to applicable securities laws and regulations, the Registrant is deemed to have complied with the reporting obligation relating to the submission of interactive data files in such exhibits and is not subject to liability under any anti-fraud provisions of the federal securities laws as long as the Registrant has made a good faith attempt to comply with the submission requirements and promptly amends the interactive data files after becoming aware that the interactive data files fail to comply with the submission requirements. These interactive data files are deemed not filed or part of a registration statement or prospectus for purposes of sections 11 or 12 of the Securities Act of 1933, as amended, are deemed not filed for purposes of section 18 of the Securities Exchange Act of 1934, as amended, and otherwise are not subject to liability under these sections. |
57
Table of Contents
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| GTx, Inc. | ||||
| By |
/s/ Mitchell S. Steiner | |||
| Mitchell S. Steiner, M.D., F.A.C.S. Chief Executive Officer, Vice Chairman and Director |
Date: March 2, 2012 | |||
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENT, that each person whose signature appears below constitutes and appoints Mitchell S. Steiner and Mark E. Mosteller, and each of them, acting individually, as his attorney-in-fact, each with full power of substitution and resubstitution, for him or her and in his or her name, place and stead, in any and all capacities, to sign any and all amendments to this Annual Report on Form 10-K, and to file the same, with all exhibits thereto, and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, and each of them, full power and authority to do and perform each and every act and thing requisite and necessary to be done in connection therewith and about the premises, as fully to all intents and purposes as he or she might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact and agents, or any of them, or their or his or her substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| Date | ||||
| /s/ J. R. Hyde, III J. R. Hyde, III |
Chairman of the Board of Directors | March 2, 2012 | ||
| /s/ Mitchell S. Steiner Mitchell S. Steiner, M.D., F.A.C.S. |
Chief Executive Officer, Vice Chairman and Director (Principal Executive Officer) | March 2, 2012 | ||
| /s/ Mark E. Mosteller Mark E. Mosteller, CPA |
Vice President and Chief Financial Officer (Principal Financial and Accounting Officer) |
March 2, 2012 | ||
| /s/ Michael G. Carter Michael G. Carter, M. D. |
Director | March 2, 2012 | ||
| /s/ Barrington J. A. Furr Barrington J. A. Furr |
Director | March 2, 2012 | ||
| /s/ J. Kenneth Glass J. Kenneth Glass |
Director | March 2, 2012 | ||
| /s/ Kenneth S. Robinson Kenneth S. Robinson, M.D. |
Director | March 2, 2012 | ||
| /s/ Timothy R. G. Sear Timothy R. G. Sear |
Director | March 2, 2012 | ||
58
Table of Contents
GTx, Inc.
F-1
Table of Contents
INTERNAL CONTROL OVER FINANCIAL REPORTING
We, as management of GTx, Inc., are responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Securities Exchange Act Rule 13a-15(f). Internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with United States generally accepted accounting principles. Any system of internal control, no matter how well designed, has inherent limitations, including the possibility that a control can be circumvented or overridden and misstatements due to error or fraud may occur and not be detected. Also, because of changes in conditions, internal control effectiveness may vary over time. Accordingly, even an effective system of internal control will provide only reasonable assurance that the objectives of the internal control system are met.
Under the supervision and with the participation of management, including our principal executive officer and principal financial officer, we conducted an evaluation of the effectiveness of our internal control over financial reporting as of December 31, 2011 using the criteria for effective internal control over financial reporting as described in “Internal Control – Integrated Framework,” issued by the Committee of Sponsoring Organizations of the Treadway Commission. Based on this evaluation, we concluded that, as of December 31, 2011, our internal control over financial reporting was effective. The effectiveness of our internal control over financial reporting has been audited by Ernst & Young LLP, independent registered public accounting firm.
| /s/ Mitchell S. Steiner | /s/ Mark E. Mosteller | |||
| Mitchell S. Steiner, M.D., F.A.C.S. | Mark E. Mosteller, CPA | |||
| Vice Chairman and | Vice President and Chief Financial Officer | |||
| Chief Executive Officer |
Memphis, Tennessee
March 2, 2012
F-2
Table of Contents
Report of Independent Registered Public Accounting Firm
The Board of Directors and Stockholders of GTx, Inc.
We have audited GTx, Inc.’s internal control over financial reporting as of December 31, 2011, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (the COSO criteria). GTx Inc.’s management is responsible for maintaining effective internal control over financial reporting, and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management’s Report on Internal Control Over Financial Reporting. Our responsibility is to express an opinion on the Company’s internal control over financial reporting based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
In our opinion, GTx, Inc. maintained, in all material respects, effective internal control over financial reporting as of December 31, 2011, based on the COSO criteria.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), the accompanying balance sheets of GTx, Inc. as of December 31, 2011 and 2010, and the related statements of operations, stockholders’ equity (deficit) and cash flows for each of the three years in the period ended December 31, 2011 and our report dated March 2, 2012 expressed an unqualified opinion thereon.
/s/ Ernst & Young LLP
Memphis, Tennessee
March 2, 2012
F-3
Table of Contents
Report of Independent Registered Public Accounting Firm
The Board of Directors and Stockholders of GTx, Inc.
We have audited the accompanying balance sheets of GTx, Inc. as of December 31, 2011 and 2010, and the related statements of operations, stockholders’ equity (deficit) and cash flows for each of the three years in the period ended December 31, 2011. These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the financial position of GTx, Inc. at December 31, 2011 and 2010, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2011, in conformity with U.S. generally accepted accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), GTx, Inc.’s internal control over financial reporting as of December 31, 2011, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission and our report dated March 2, 2012 expressed an unqualified opinion thereon.
/s/ Ernst & Young LLP
Memphis, Tennessee
March 2, 2012
F-4
Table of Contents
GTx, Inc.
(in thousands, except share and per share data)
| December 31, | ||||||||
| 2011 | 2010 | |||||||
| ASSETS |
||||||||
| Current assets: |
||||||||
| Cash and cash equivalents |
$ | 63,745 | $ | 58,181 | ||||
| Short-term investments |
10,695 | 450 | ||||||
| Accounts receivable, net |
981 | 683 | ||||||
| Inventory |
161 | 171 | ||||||
| Prepaid expenses and other current assets |
1,266 | 875 | ||||||
|
|
|
|
|
|||||
| Total current assets |
76,848 | 60,360 | ||||||
| Property and equipment, net |
1,096 | 2,040 | ||||||
| Intangible and other assets, net |
240 | 1,850 | ||||||
|
|
|
|
|
|||||
| Total assets |
$ | 78,184 | $ | 64,250 | ||||
|
|
|
|
|
|||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY |
||||||||
| Current liabilities: |
||||||||
| Accounts payable |
$ | 1,219 | $ | 848 | ||||
| Accrued expenses and other current liabilities |
4,857 | 3,112 | ||||||
| Deferred revenue – current portion |
— | 1,345 | ||||||
|
|
|
|
|
|||||
| Total current liabilities |
6,076 | 5,305 | ||||||
| Deferred revenue, less current portion |
— | 6,721 | ||||||
| Other long-term liabilities |
234 | 497 | ||||||
| Commitments and contingencies |
||||||||
| Stockholders’ equity: |
||||||||
| Common stock, $0.001 par value: 120,000,000 and 60,000,000 shares authorized at December 31, 2011 and December 31, 2010, respectively; 62,790,223 and 51,719,187 shares issued and outstanding at December 31, 2011 and December 31, 2010, respectively |
63 | 52 | ||||||
| Additional paid-in capital |
457,985 | 404,555 | ||||||
| Accumulated deficit |
(386,174 | ) | (352,880 | ) | ||||
|
|
|
|
|
|||||
| Total stockholders’ equity |
71,874 | 51,727 | ||||||
|
|
|
|
|
|||||
| Total liabilities and stockholders’ equity |
$ | 78,184 | $ | 64,250 | ||||
|
|
|
|
|
|||||
The accompanying notes are an integral part of these financial statements.
F-5
Table of Contents
GTx, Inc.
(in thousands, except share and per share data)
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Revenues: |
||||||||||||
| Product sales, net |
$ | 6,673 | $ | 3,827 | $ | 3,289 | ||||||
| Collaboration revenue |
8,066 | 56,786 | 11,441 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total revenues |
14,739 | 60,613 | 14,730 | |||||||||
| Costs and expenses: |
||||||||||||
| Cost of product sales |
1,055 | 768 | 1,290 | |||||||||
| Research and development expenses |
31,938 | 28,495 | 32,344 | |||||||||
| General and administrative expenses |
15,438 | 17,419 | 27,778 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total costs and expenses |
48,431 | 46,682 | 61,412 | |||||||||
|
|
|
|
|
|
|
|||||||
| (Loss) income from operations |
(33,692 | ) | 13,931 | (46,682 | ) | |||||||
| Other income, net |
398 | 1,363 | 188 | |||||||||
|
|
|
|
|
|
|
|||||||
| (Loss) income before income taxes |
(33,294 | ) | 15,294 | (46,494 | ) | |||||||
| Income tax benefit |
— | — | 238 | |||||||||
|
|
|
|
|
|
|
|||||||
| Net (loss) income |
$ | (33,294 | ) | $ | 15,294 | $ | (46,256 | ) | ||||
|
|
|
|
|
|
|
|||||||
| Net (loss) income per share: |
||||||||||||
| Basic and diluted |
$ | (0.58 | ) | $ | 0.39 | $ | (1.27 | ) | ||||
|
|
|
|
|
|
|
|||||||
| Weighted average shares used in computing net (loss) income per share: |
||||||||||||
| Basic and diluted |
57,359,466 | 38,874,721 | 36,415,379 | |||||||||
|
|
|
|
|
|
|
|||||||
The accompanying notes are an integral part of these financial statements.
F-6
Table of Contents
GTx, Inc.
STATEMENTS OF STOCKHOLDERS’ EQUITY (DEFICIT)
For the Years Ended December 31, 2011, 2010 and 2009
(in thousands, except share data)
| Stockholders’ Equity (Deficit) | ||||||||||||||||||||
| Additional | Total Stockholders’ | |||||||||||||||||||
| Common Stock | Paid-in | Accumulated | ||||||||||||||||||
| Shares | Amount | Capital | Deficit | Equity (Deficit) | ||||||||||||||||
| Balances at January 1, 2009 |
36,392,443 | $ | 36 | $ | 353,900 | $ | (321,918 | ) | $ | 32,018 | ||||||||||
| Exercise of employee stock options |
18,434 | — | 136 | — | 136 | |||||||||||||||
| Directors’ deferred compensation |
— | — | 178 | — | 178 | |||||||||||||||
| Issuance of common stock under deferred compensation arrangements |
10,024 | — | — | — | — | |||||||||||||||
| Share-based compensation |
— | — | 5,174 | — | 5,174 | |||||||||||||||
| Net loss and comprehensive loss |
— | — | — | (46,256 | ) | (46,256 | ) | |||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Balances at December 31, 2009 |
36,420,901 | 36 | 359,388 | (368,174 | ) | (8,750 | ) | |||||||||||||
| Issuance of common stock, net of offering costs |
15,285,715 | 15 | 40,273 | — | 40,288 | |||||||||||||||
| Issuance of common stock under deferred compensation arrangements |
8,321 | — | — | — | — | |||||||||||||||
| Exercise of employee stock options |
4,250 | — | 9 | — | 9 | |||||||||||||||
| Directors’ deferred compensation |
— | 1 | 186 | — | 187 | |||||||||||||||
| Share-based compensation |
— | — | 4,699 | — | 4,699 | |||||||||||||||
| Net income and comprehensive income |
— | — | — | 15,294 | 15,294 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Balances at December 31, 2010 |
51,719,187 | 52 | 404,555 | (352,880 | ) | 51,727 | ||||||||||||||
| Issuance of common stock, net of offering costs |
11,023,000 | 11 | 48,971 | — | 48,982 | |||||||||||||||
| Issuance of common stock under deferred compensation arrangements |
35,036 | — | — | — | — | |||||||||||||||
| Exercise of employee stock options |
13,000 | — | 55 | — | 55 | |||||||||||||||
| Directors’ deferred compensation |
— | — | 178 | — | 178 | |||||||||||||||
| Share-based compensation |
— | — | 4,226 | — | 4,226 | |||||||||||||||
| Net loss and comprehensive loss |
— | — | — | (33,294 | ) | (33,294 | ) | |||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Balances at December 31, 2011 |
62,790,223 | $ | 63 | $ | 457,985 | $ | (386,174 | ) | $ | 71,874 | ||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
The accompanying notes are an integral part of these financial statements.
F-7
Table of Contents
GTx, Inc.
(in thousands)
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Cash flows from operating activities: |
||||||||||||
| Net (loss) income |
$ | (33,294 | ) | $ | 15,294 | $ | (46,256 | ) | ||||
| Adjustments to reconcile net (loss) income to net cash used in operating activities: |
||||||||||||
| Depreciation and amortization |
1,040 | 1,573 | 1,785 | |||||||||
| Share-based compensation |
4,226 | 4,699 | 5,174 | |||||||||
| Directors’ deferred compensation |
178 | 187 | 178 | |||||||||
| Deferred revenue amortization |
(8,066 | ) | (51,786 | ) | (11,441 | ) | ||||||
| Impairment of intangible assets |
1,598 | 1,687 | — | |||||||||
| Write off of property and equipment |
— | — | 114 | |||||||||
| Changes in assets and liabilities: |
||||||||||||
| Short-term investments, trading |
— | — | 2,157 | |||||||||
| Accounts receivable, net |
(298 | ) | (277 | ) | 81 | |||||||
| Inventory |
(24 | ) | (55 | ) | (24 | ) | ||||||
| Receivable from collaboration partners |
— | 189 | 588 | |||||||||
| Prepaid expenses and other assets |
(388 | ) | 36 | 85 | ||||||||
| Accounts payable |
371 | (420 | ) | (1,553 | ) | |||||||
| Accrued expenses and other liabilities |
1,568 | (1,654 | ) | (1,956 | ) | |||||||
| Deferred revenue |
— | — | 5,071 | |||||||||
|
|
|
|
|
|
|
|||||||
| Net cash used in operating activities |
(33,089 | ) | (30,527 | ) | (45,997 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| Cash flows from investing activities: |
||||||||||||
| Purchase of property and equipment |
(54 | ) | (95 | ) | (600 | ) | ||||||
| Purchase of short-term investments, held to maturity |
(15,145 | ) | (8,569 | ) | (11,275 | ) | ||||||
| Proceeds from maturities of short-term investments, held to maturity |
4,900 | 16,944 | 2,450 | |||||||||
|
|
|
|
|
|
|
|||||||
| Net cash (used in) provided by investing activities |
(10,299 | ) | 8,280 | (9,425 | ) | |||||||
|
|
|
|
|
|
|
|||||||
| Cash flows from financing activities: |
||||||||||||
| Proceeds from issuance of common stock |
48,982 | 40,288 | — | |||||||||
| Proceeds from exercise of employee stock options |
55 | 9 | 136 | |||||||||
| Payments on capital lease and financed equipment obligations |
(85 | ) | (88 | ) | (5 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| Net cash provided by financing activities |
48,952 | 40,209 | 131 | |||||||||
|
|
|
|
|
|
|
|||||||
| Net increase (decrease) in cash and cash equivalents |
5,564 | 17,962 | (55,291 | ) | ||||||||
| Cash and cash equivalents, beginning of period |
58,181 | 40,219 | 95,510 | |||||||||
|
|
|
|
|
|
|
|||||||
| Cash and cash equivalents, end of period |
$ | 63,745 | $ | 58,181 | $ | 40,219 | ||||||
|
|
|
|
|
|
|
|||||||
| Supplemental schedule of non-cash investing and financing activities: |
||||||||||||
| Equipment purchased under debt or capital lease |
$ | — | $ | — | $ | 268 | ||||||
|
|
|
|
|
|
|
|||||||
The accompanying notes are an integral part of these financial statements.
F-8
Table of Contents
GTx, Inc.
(in thousands, except share and per share data)
1. Business
GTx, Inc. (“GTx” or the “Company”), a Delaware corporation incorporated on September 24, 1997 and headquartered in Memphis, Tennessee, is a biopharmaceutical company dedicated to the discovery, development and commercialization of small molecules that selectively target hormone pathways for the treatment of cancer, cancer supportive care, and other serious medical conditions.
The Company is developing selective androgen receptor modulators (“SARMs”), including enobosarm (also known as OstarineTM or GTx-024). SARMs are a new class of drugs with the potential to prevent and treat muscle wasting in patients with cancer and other musculoskeletal wasting or muscle loss conditions, including chronic sarcopenia (age related muscle loss). The Company is conducting two pivotal Phase III clinical trials evaluating enobosarm for the prevention and treatment of muscle wasting in patients with non-small cell lung cancer.
Additionally, the Company has been developing Capesaris® (GTx-758), an oral nonsteroidal selective estrogen receptor alpha agonist, for primary (first line) androgen deprivation therapy for advanced prostate cancer and secondary (second line) hormonal treatment. On February 21, 2012, the Company announced that the United States Food and Drug Administration (“FDA”) had placed a clinical hold on the Company’s three clinical trials evaluating Capesaris®. See Note 13, Subsequent Events, for further discussion.
The Company sells FARESTON® (toremifene citrate) 60 mg tablets, approved for the treatment of advanced metastatic breast cancer in postmenopausal women, in the United States. The Company previously determined to discontinue its toremifene 80 mg and toremifene 20 mg development programs.
2. Significant Accounting Policies
Basis of Presentation
The accompanying financial statements have been prepared in accordance with accounting principles generally accepted in the United States (“U.S. GAAP”). Additionally, GTx operates in one business segment.
Use of Estimates
The preparation of financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities, the disclosure of contingent assets and liabilities at the date of the financial statements, and the reported amounts of revenues and expenses during the reporting period. Actual amounts and results could differ from those estimates.
Cash and Cash Equivalents
The Company considers highly liquid investments with initial maturities of three months or less to be cash equivalents.
Short-term Investments
At December 31, 2011 and 2010, short-term investments consisted of certificates of deposit with original maturities of greater than three months and less than one year. As the Company has the positive intent and ability to hold the certificates of deposit until maturity, these investments have been classified as held to maturity investments and are stated at cost, which approximates fair value.
Accounts Receivable
Accounts receivable are recorded net of allowances for cash discounts for prompt payment. The Company makes judgments as to its ability to collect outstanding receivables and will provide allowances for the portion of receivables if and when collection becomes doubtful. The Company has not recorded reserves related to the collectability of its accounts receivable for the years ended December 31, 2011 and 2010.
F-9
Table of Contents
GTx, Inc.
NOTES TO FINANCIAL STATEMENTS
(in thousands, except share and per share data)
Inventory
Inventory consists of FARESTON® tablets that are manufactured by Orion Corporation (“Orion”) and delivered to the Company as finished goods. Inventory is stated at the lower of cost (first-in, first-out method) or market. The Company analyzes its current inventory levels and will write down inventory if it has become un-saleable or has a cost basis in excess of its expected net realizable value. To date, there have been no inventory write-downs.
Property and Equipment
Property and equipment is stated at cost. Amortization of leasehold improvements is recognized over the shorter of the estimated useful life of the leasehold improvement or the lease term. Depreciation is computed using the straight-line method over the estimated useful lives as follows:
| Laboratory and office equipment |
3 to 5 years | |
| Leasehold improvements |
3 to 7 years | |
| Furniture and fixtures |
5 years | |
| Computer equipment and software |
3 years | |
Intangible Assets
The Company amortizes its purchased intangible assets with finite lives over their estimated economic lives. The Company’s purchased intangible assets, license fees, represent the value of each license acquired by the Company pursuant to the agreements described in Note 6, Intangible Assets. License fees are amortized on a straight-line basis over the respective terms of the agreements.
Impairment of Long-Lived Assets
The Company reviews long-lived assets for impairment whenever events or changes in facts and circumstances are present, both internally and externally, that may indicate impairment of long-lived assets. An impairment loss is recognized when estimated future cash flows are less than the carrying amount. The cash flow estimates are based on management’s best estimates, using appropriate and customary assumptions and projections at the time. See Note 6, Intangible Assets, for further discussion.
Fair Value of Financial Instruments
The carrying amounts of the Company’s financial instruments, which include cash, cash equivalents, short-term investments, accounts receivable and accounts payable approximate their fair values. The method of determining the fair value for the Company’s short-term investments is discussed above (See Short-term Investments).
Concentration of Risk
Financial instruments that potentially subject the Company to a concentration of credit risk consist of cash and cash equivalents, short-term investments and accounts receivable. The Company has established guidelines relating to diversification and maturities of its cash equivalents and short-term investments which are designed to manage risk. The Company’s cash and cash equivalents consist of bank deposits, certificates of deposit, and money market mutual funds. Bank deposits may at times be in excess of FDIC insurance limits. The Company’s short-term investments consist of investments in certificates of deposit with original maturities of greater than 3 months and less than 1 year as discussed above (See Short-term Investments).
F-10
Table of Contents
GTx, Inc.
NOTES TO FINANCIAL STATEMENTS
(in thousands, except share and per share data)
Three wholesale drug distributors individually comprised 25%, 40% and 33%, respectively, of the Company’s accounts receivable as of December 31, 2011. These same three distributors represented 22%, 40% and 33%, respectively, of the Company’s product sales for the year ended December 31, 2011.
Revenue Recognition
The Company recognizes revenue from product sales of FARESTON® less deductions for estimated sales discounts and sales returns. Revenue from product sales is recognized when persuasive evidence of an arrangement exists, title passes, the price is fixed or determinable, and collectability is reasonably assured. The Company accounts for rebates to certain governmental agencies as a reduction of product sales. The Company allows customers to return product within a specified time period prior to and subsequent to the product’s labeled expiration date. The Company estimates an accrual for product returns, which is recorded as a reduction of product sales, based on factors which include historical product returns and estimated product in the distribution channel which is expected to exceed its expiration date. At December 31, 2011 and 2010, the Company’s accrual for product returns was $1,114 and $802, respectively.
Collaboration revenue consisted of non-refundable upfront payments, license fees, reimbursements for research and development activities, and milestone payments associated with the Company’s former collaboration and license agreements. Revenues from the Company’s prior collaboration and license agreements were recognized based on the performance requirements of the specific agreements. The Company analyzed agreements with multiple element arrangements to determine whether the deliverables under the agreement, including license and performance obligations such as joint steering committee participation and research and development activities, could have been separated or whether all of the deliverables must have been accounted for as a single unit of accounting. Revenues from milestone payments for which the Company had no continuing performance obligations were recognized upon achievement of the performance milestone, as defined in the related agreement, provided the milestone was substantive and a culmination of the earnings process had occurred. See Note 8, Collaboration and License Agreements, for further discussion.
Research and Development Expenses
Research and development expenses include, but are not limited to, the Company’s expenses for personnel, supplies, and facilities associated with research activities, screening and identification of product candidates, formulation and synthesis activities, manufacturing, preclinical studies, toxicology studies, clinical trials, regulatory and medical affairs activities, quality assurance activities and license fees. The Company expenses these costs in the period in which they are incurred. The Company estimates its liabilities for research and development expenses in order to match the recognition of expenses to the period in which the actual services are received. As such, accrued liabilities related to third party research and development activities are recognized based upon the Company’s estimate of services received and degree of completion of the services in accordance with the specific third party contract.
Patent Costs
The Company expenses patent costs, including legal expenses, in the period in which they are incurred. Patent expenses are included in general and administrative expenses in the Company’s statements of operations.
Income Taxes
The Company accounts for deferred taxes by recognition of deferred tax assets and liabilities for the expected future tax consequences of events that have been included in the financial statements or tax returns. Under this method, deferred tax assets and liabilities are determined based on the difference between the financial statement and tax basis of assets and liabilities using enacted tax rates in effect for the year in which the differences are expected to reverse. A valuation allowance is provided when it is more likely than not that some portion or all of the deferred tax assets will not be realized. Accordingly, at December 31, 2011 and 2010, net of the valuation allowance, the net deferred tax assets were reduced to zero. See Note 9, Income Taxes, for further discussion.
F-11
Table of Contents
GTx, Inc.
NOTES TO FINANCIAL STATEMENTS
(in thousands, except share and per share data)
Share-Based Compensation
The Company has stock option and equity incentive plans that provide for the purchase or acquisition of the Company’s common stock by certain of the Company’s employees and non-employee directors. The Company recognizes compensation expense for its share-based payments based on the fair value of the awards over the period during which an employee or non-employee director is required to provide service in exchange for the award. See Note 3, Share-Based Compensation, for further discussion.
Other Income, Net
Other income, net consists of interest earned on the Company’s cash, cash equivalents and short-term investments, interest expense, foreign currency transaction gains and losses, and other non-operating income or expense.
For the year ended December 31, 2010, other income, net included income from grants totaling $1,220 awarded to the Company by the United States Government under the Qualifying Therapeutic Discovery Project Program, which was established under the Patient Protection and Affordable Care Act. The Company was granted $244 for each of five applications submitted for the Company’s cancer and cancer supportive care research and development programs.
Basic and Diluted Net (Loss) Income Per Share
Basic net (loss) income per share is calculated based on the weighted average number of common shares outstanding during the period. Diluted net (loss) income per share gives effect to the dilutive potential of common stock consisting of stock options.
The following table sets forth the computation of the Company’s basic and diluted net (loss) income per share for the years ended December 31, 2011, 2010 and 2009:
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Basic and diluted net (loss) income per share |
||||||||||||
| Numerator: |
||||||||||||
| Net (loss) income |
$ | (33,294 | ) | $ | 15,294 | $ | (46,256 | ) | ||||
|
|
|
|
|
|
|
|||||||
| Denominator: |
||||||||||||
| Weighted average shares used in computing basic and diluted net (loss) income per share |
57,359,466 | 38,874,721 | 36,415,379 | |||||||||
|
|
|
|
|
|
|
|||||||
| Basic and diluted net (loss) income per share |
$ | (0.58 | ) | $ | 0.39 | $ | (1.27 | ) | ||||
|
|
|
|
|
|
|
|||||||
Weighted average options outstanding to purchase shares of common stock of 5,327,752, 4,473,576, and 3,597,716 were excluded from the calculation of diluted net (loss) income per share for the years ended December 31, 2011, 2010 and 2009, respectively, as inclusion of the options would have had an anti-dilutive effect on the net (loss) income per share for the periods. At December 31, 2011, the Company had outstanding 62,790,223 shares of common stock.
Comprehensive (Loss) Income
For all periods presented, there were no differences between net (loss) income and comprehensive (loss) income.
F-12
Table of Contents
GTx, Inc.
NOTES TO FINANCIAL STATEMENTS
(in thousands, except share and per share data)
Reduction in Force
In June 2011, the Company implemented a reduction in its workforce in connection with its decision to discontinue development and commercialization of its toremifene 80 mg and toremifene 20 mg product candidates. The reduction in force was effective immediately and represented approximately 15% of the Company’s total workforce, including three non-executive officers of the Company. As a result of the workforce reduction, the Company incurred severance related cash expenses of approximately $681, of which $633 was included in general and administrative expenses and $48 was included in research and development expenses for the year ended December 31, 2011. The Company paid these expenses in their entirety in 2011 and had no recorded liabilities related to the workforce reduction as of December 31, 2011. Additionally, the Company incurred a one-time, non-cash share-based compensation charge of $481 related to the modification of certain stock option provisions for the affected non-executive officers, which was included in general and administrative expenses for the year ended December 31, 2011. This charge was offset by the reversal of $704 of previously recognized share-based compensation expense for non-vested stock options that were cancelled in conjunction with the workforce reduction. Of this amount, $646 was included in general and administrative expenses and $58 was included in research and development expenses for the year ended December 31, 2011.
Recent Accounting Pronouncements
In May 2011, the Financial Accounting Standards Board issued Accounting Standard Update 2011-04, Amendments to Achieve Common Fair Value Measurement and Disclosure Requirements in U.S. GAAP and IFRS. The new guidance results in a consistent definition of fair value and common requirements for measurement of and disclosure about fair value between U.S. GAAP and International Financial Reporting Standards. While many of the amendments to U.S. GAAP are not expected to have a significant effect on practice, the new guidance changes some fair value measurement principles and disclosure requirements. This new guidance is effective for fiscal years and interim periods beginning after December 15, 2011. The Company does not expect the adoption of the standard update to have a significant impact on its financial position or results of operations.
3. Share-Based Compensation
Share-based payments include stock option grants under the Company’s stock option and equity incentive plans and deferred compensation arrangements for the Company’s non-employee directors.
The Company grants to employees and non-employee directors options to purchase common stock under various plans at prices equal to the fair market value of the stock on the dates the options are granted as determined in accordance with the terms of the applicable plan. The options have a term of ten years from the grant date and vest over three years from the grant date for director options and over periods of up to five years from the grant date for employee options. Employees generally have three months after the employment relationship ends to exercise all vested options except in the case of voluntary retirement, disability or death, where exercise periods are generally longer. The Company issues new shares of common stock upon the exercise of options. The Company estimates the fair value of certain stock option awards as of the date of the grant by applying the Black-Scholes-Merton option pricing valuation model. The application of this valuation model involves assumptions that are judgmental and highly sensitive in the determination of compensation expense.
The fair value of each option is amortized into compensation expense on a straight-line basis between the grant date for the award and each vesting date. The amount of share-based compensation expense recognized is reduced ratably over the vesting period by an estimate of the percentage of options granted that are expected to be forfeited or canceled before becoming fully vested.
F-13
Table of Contents
GTx, Inc.
NOTES TO FINANCIAL STATEMENTS
(in thousands, except share and per share data)
The following table summarizes share-based compensation expense included within the statements of operations for each of the three years in the period ended December 31, 2011:
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Research and development expenses |
$ | 1,972 | $ | 2,340 | $ | 2,143 | ||||||
| General and administrative expenses |
2,432 | 2,546 | 3,209 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total share-based compensation |
$ | 4,404 | $ | 4,886 | $ | 5,352 | ||||||
|
|
|
|
|
|
|
|||||||
Share-based compensation expense recorded in the statement of operations as general and administrative expense for the years ended December 31, 2011, 2010 and 2009 included share-based compensation expense related to deferred compensation arrangements for the Company’s non-employee directors of $178, $187 and $178, respectively. See Note 10, Directors’ Deferred Compensation Plan, for further discussion of deferred compensation arrangements for the Company’s non-employee directors. Additionally, as part of the June 2011 workforce reduction, the Company modified certain stock options of three terminated non-executive officers to accelerate the vesting of certain outstanding non-vested stock options and to extend the post-termination exercise period of their vested stock options. As a result of these modifications, the Company incurred a one-time share-based compensation charge of $481, which was included in general and administrative expenses for the year ended December 31, 2011. This charge was offset by the reversal of $704 of previously recognized share-based compensation expense for non-vested stock options that were canceled in conjunction with the total workforce reduction. Of this amount, $646 was included in general and administrative expenses and $58 was included in research and development expenses for the year ended December 31, 2011.
For the years ended December 31, 2011, 2010 and 2009, the weighted average grant date fair value per share of options granted was $1.75, $2.62 and $8.45, respectively. The weighted average for key assumptions used in determining the grant date fair value of options granted in 2011, 2010 and 2009, and a summary of the methodology applied to develop each assumption were as follows:
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Expected price volatility |
64.9 | % | 64.6 | % | 55.0 | % | ||||||
| Risk-free interest rate |
2.54 | % | 3.36 | % | 2.04 | % | ||||||
| Weighted average expected life in years |
6.5 years | 6.5 years | 6.9 years | |||||||||
| Dividend yield |
0 | % | 0 | % | 0 | % | ||||||
Expected Price Volatility—This is a measure of the amount by which a price has fluctuated or is expected to fluctuate. The Company based its determination of expected volatility on its historical stock price volatility. An increase in the expected price volatility will increase compensation expense.
Risk-Free Interest Rate—This is determined using U.S. Treasury rates where the term is consistent with the expected life of the stock options. An increase in the risk-free interest rate will increase compensation expense.
Expected Life—This is the period of time over which the options granted are expected to remain outstanding and is determined by calculating the average of the vesting term and the contractual term of the options. The Company has utilized this method due to the lack of historical option exercise information related to the Company’s stock option and equity incentive plans. Options granted have a maximum term of ten years. An increase in the expected life will increase compensation expense.
Dividend Yield—The Company has not made any dividend payments nor does it have plans to pay dividends in the foreseeable future. An increase in the dividend yield will decrease compensation expense.
F-14
Table of Contents
GTx, Inc.
NOTES TO FINANCIAL STATEMENTS
(in thousands, except share and per share data)
The following is a summary of stock option transactions for all of the Company’s stock option and equity incentive plans for the three year period ended December 31, 2011:
| Number of Shares |
Weighted Average Exercise Price Per Share |
|||||||
| Options outstanding at January 1, 2009 |
2,673,976 | 13.01 | ||||||
| Options granted |
1,118,150 | 15.07 | ||||||
| Options forfeited or expired |
(408,821 | ) | 14.52 | |||||
| Options exercised |
(18,434 | ) | 7.37 | |||||
|
|
|
|||||||
| Options outstanding at December 31, 2009 |
3,364,871 | 13.55 | ||||||
| Options granted |
1,320,500 | 4.14 | ||||||
| Options forfeited or expired |
(250,626 | ) | 10.68 | |||||
| Options exercised |
(4,250 | ) | 2.24 | |||||
|
|
|
|||||||
| Options outstanding at December 31, 2010 |
4,430,495 | 10.91 | ||||||
| Options granted |
1,395,000 | 2.82 | ||||||
| Options forfeited or expired |
(866,930 | ) | 8.23 | |||||
| Options exercised |
(13,000 | ) | 4.20 | |||||
|
|
|
|||||||
| Options outstanding at December 31, 2011 |
4,945,565 | 9.12 | ||||||
|
|
|
|||||||
| Options vested and expected to vest at December 31, 2011 |
4,821,162 | 9.20 | ||||||
|
|
|
|||||||
The following table summarizes information about stock options outstanding at December 31, 2011:
| Options Outstanding |
Options Exercisable | |||||||||||||||||||
| Exercise Price |
Number Outstanding |
Weighted Average Remaining Contractual Life (years) |
Weighted Average Exercise Price |
Number Exercisable |
Weighted Average Exercise Price |
|||||||||||||||
| $2.65 - $4.20 |
2,133,100 | 8.26 | $ | 3.40 | 297,636 | $ | 3.95 | |||||||||||||
| $5.17 - $15.59 |
1,649,047 | 4.29 | 10.62 | 1,151,572 | 9.81 | |||||||||||||||
| $15.64 - $20.40 |
1,163,418 | 6.03 | 17.48 | 406,021 | 18.15 | |||||||||||||||
|
|
|
|
|
|||||||||||||||||
| 4,945,565 | 6.41 | 9.12 | 1,855,229 | 10.69 | ||||||||||||||||
|
|
|
|
|
|||||||||||||||||
At December 31, 2011, the aggregate intrinsic value of all outstanding options was $759 with a weighted average remaining contractual term of 6.41 years. Of the Company’s outstanding options, 1,855,229 options were exercisable and had a weighted average remaining contractual term of 4.12 years and an aggregate intrinsic value of $23. Additionally, the Company’s vested and expected to vest options had a weighted average remaining contractual term of 6.36 years and an intrinsic value of $717.
Options to purchase 13,000 shares were exercised during the year ended December 31, 2011. The total intrinsic value of options exercised during the years ended December 31, 2011, 2010 and 2009 was $15, $6 and $99, respectively. At December 31, 2011, the total compensation cost related to non-vested options not yet recognized was $6,341, with a weighted average expense recognition period of 2.78 years. Shares available for future issuance under the Company’s stock option and equity incentive plans were 1,825,241 at December 31, 2011. On January 1, 2012, shares available for future issuance under the Company’s stock option and equity incentive plans increased to 5,059,752 shares in accordance with the provisions of such plans.
F-15
Table of Contents
GTx, Inc.
NOTES TO FINANCIAL STATEMENTS
(in thousands, except share and per share data)
4. Property and Equipment, Net
Property and equipment, net consisted of the following:
| December 31, | ||||||||
| 2011 | 2010 | |||||||
| Laboratory and office equipment |
$ | 4,173 | $ | 4,183 | ||||
| Computer equipment and software |
2,646 | 2,612 | ||||||
| Furniture and fixtures |
1,361 | 1,361 | ||||||
| Leasehold improvements |
1,024 | 1,024 | ||||||
|
|
|
|
|
|||||
| 9,204 | 9,180 | |||||||
| Less: accumulated depreciation |
(8,108 | ) | (7,140 | ) | ||||
|
|
|
|
|
|||||
| $ | 1,096 | $ | 2,040 | |||||
|
|
|
|
|
|||||
Depreciation and amortization expense for the years ended December 31, 2011, 2010 and 2009 was $998, $1,346 and $1,447, respectively. Of these amounts, $425, $543 and $619, respectively, were included in research and development expenses in the statements of operations.
5. Accrued Expenses and Other Current Liabilities
Accrued expenses and other current liabilities consisted of the following:
| December 31, | ||||||||
| 2011 | 2010 | |||||||
| Clinical trials |
$ | 2,286 | $ | 189 | ||||
| Product returns |
1,114 | 802 | ||||||
| Selling, general and administrative |
1,037 | 1,331 | ||||||
| Research and development |
73 | 379 | ||||||
| Employee compensation |
265 | 326 | ||||||
| Current portion of capital lease and financed equipment liabilities |
82 | 85 | ||||||
|
|
|
|
|
|||||
| $ | 4,857 | $ | 3,112 | |||||
|
|
|
|
|
|||||
6. Intangible Assets, Net
Intangible assets, net consisted of the following:
| December 31, | ||||||||
| 2011 | 2010 | |||||||
| License fees |
$ | 290 | $ | 2,703 | ||||
| Less: accumulated amortization |
(89 | ) | (862 | ) | ||||
|
|
|
|
|
|||||
| $ | 201 | $ | 1,841 | |||||
|
|
|
|
|
|||||
In accordance with the terms of the Amended and Restated License and Supply Agreement that the Company entered into with Orion effective January 1, 2005 (“Orion License and Supply Agreement”), the Company paid a license fee to Orion of $4,826. In accordance with the terms of the Consolidated, Amended, and Restated License Agreement (“SARM License Agreement”) and the Amended and Restated License Agreement (“SERM License Agreement”) that the Company entered into with the University of Tennessee Research Foundation (“UTRF”) in July 2007 and September 2007, respectively, the Company paid a one-time up-front fee of $290 per agreement.
Based upon the Company’s decision to discontinue toremifene 80 mg development and after analyzing future cash flows and estimates of fair market value from a market participant perspective, the Company determined that its toremifene 80 mg intangible asset was impaired and recorded an impairment charge of $1,598 during the first quarter of 2011. The impaired intangible asset consisted of capitalized license fees paid to Orion related to the Company’s toremifene 80 mg program. The impairment charge was included in research and development expenses in the statement of operations for the year ended December 31, 2011.
F-16
Table of Contents
GTx, Inc.
NOTES TO FINANCIAL STATEMENTS
(in thousands, except share and per share data)
In the second quarter of 2010, the Company determined that it would discontinue its toremifene 20 mg development program and, as a result, recorded an impairment charge of $1,687. The impaired intangible assets consisted of capitalized license fees related to the Company’s toremifene 20 mg program paid under the Orion License and Supply Agreement and the SERM License Agreement. The impairment charge was included in research and development expenses in the statement of operations for the year ended December 31, 2010.
The Company’s remaining intangible asset, net at December 31, 2011 consisted of $201 related to the SARM License Agreement. This intangible asset is being amortized on a straight-line basis over the term of the agreement, which the Company estimates to be approximately 14 years.
Amortization expense for the years ended December 31, 2011, 2010 and 2009 was $42, $227 and $338, respectively. Estimated future intangible asset amortization expense as of December 31, 2011 is as follows:
| Years Ending December 31, |
||||
| 2012 |
$ | 21 | ||
| 2013 |
21 | |||
| 2014 |
21 | |||
| 2015 |
21 | |||
| 2016 |
21 | |||
| Thereafter |
96 | |||
|
|
|
|||
| Total |
$ | 201 | ||
|
|
|
|||
7. Common and Preferred Stock
The Company’s certificate of incorporation authorizes the Company to issue 120,000,000 shares of common stock, $0.001 par value per share, and 5,000,000 shares of preferred stock, $0.001 par value per share.
On December 18, 2007, the Company completed a private placement of 1,285,347 shares of common stock to Merck at a per share price of $23.34. See Note 8, Collaboration and License Agreements, for further discussion.
On November 1, 2010, the Company completed an underwritten public offering of 14,285,715 shares of its common stock at a price to the public of $2.80 per share. Net cash proceeds from the public offering were approximately $37,700 after deducting underwriting discounts and commissions and other offering expenses. The Company also granted the underwriter a 30-day option to purchase up to an additional 2,142,857 shares of common stock to cover over-allotments, if any. On November 24, 2010, the underwriter exercised its option and purchased an additional 1,000,000 shares of the Company’s common stock at a price of $2.80 per share. Net cash proceeds from the exercise of the over-allotment option were approximately $2,600 after deducting underwriting discounts and commissions and other offering expenses.
On May 6, 2011, the Company filed a Certificate of Amendment to the Company’s Restated Certificate of Incorporation with the Secretary of State of the State of Delaware to increase the number of authorized shares of the Company’s common stock, par value $0.001 per share, from 60,000,000 shares to 120,000,000 shares. The amendment was approved by the Company’s stockholders at the Company’s 2011 Annual Meeting of Stockholders held on May 5, 2011.
On June 28, 2011, the Company completed an underwritten public offering of 10,000,000 shares of its common stock at a price to the public of $4.75 per share. The Company also granted the underwriters a 30-day option to purchase up to an additional 1,500,000 shares of common stock to cover over-allotments, if any. The underwriters exercised this option and purchased an additional 1,023,000 shares of the Company’s common stock on June 28, 2011 at a price of $4.75 per share. Net cash proceeds from the public offering were approximately $49,000 after deducting the underwriting discount and offering expenses.
F-17
Table of Contents
GTx, Inc.
NOTES TO FINANCIAL STATEMENTS
(in thousands, except share and per share data)
8. Collaboration and License Agreements
Orion Corporation License and Supply Agreement
In March 2000, the Company entered into a license and supply agreement with Orion to develop and commercialize products containing toremifene, which was subsequently amended in 2001 and 2003 (“Original Orion License”). The Company’s rights under the Original Orion License were limited to specific disease fields pertaining to prostate cancer. In December 2004, the Company entered into an agreement with Orion to purchase specified FARESTON® related assets which Orion had re-acquired from another licensee. The Company also entered into the Orion License and Supply Agreement, effective January 2005, with Orion which replaced the Original Orion License and provided the Company an exclusive license from Orion to develop and commercialize toremifene-based products for all human indications worldwide, except breast cancer outside of the United States. Under the Orion License and Supply Agreement, the Company was required to pay a license fee of $4,826 and the Company is obligated to pay a royalty in the low-teens to Orion on sales by the Company, its affiliates and third-party sublicensees of toremifene-based products. The term of the Orion License and Supply Agreement lasts, on a country-by-country basis, until the later of expiration of the Company’s own patents claiming the manufacturing or the methods of use of toremifene for prostate cancer or the end of all marketing or regulatory exclusivity which the Company may obtain for toremifene-based products. Orion may terminate the Orion License and Supply Agreement, in its entirety or in some cases on a country-by-country basis, as a result of the Company’s uncured material breach or the Company’s bankruptcy.
Ipsen Collaboration and License Agreement
In September 2006, the Company entered into a collaboration and license agreement with Ipsen (the “Ipsen Collaboration Agreement”) pursuant to which the Company granted Ipsen exclusive rights in the European Union, Switzerland, Norway, Iceland, Lichtenstein, and the Commonwealth of Independent States (the “European Territory”) to develop and commercialize toremifene in all indications which the Company has licensed from Orion, which include all indications in humans except the treatment and prevention of breast cancer outside of the United States.
In accordance with the terms of the Ipsen Collaboration Agreement, Ipsen paid the Company €23,000 as a license fee and expense reimbursement. In February 2008, the Company earned a milestone of €1,000 (approximately $1,482) with the achievement of the primary endpoint in the Company’s completed pivotal Phase III clinical trial evaluating toremifene 80 mg to reduce fractures in men with prostate cancer on androgen deprivation therapy. This amount was recognized as collaboration revenue in the first quarter of 2008. Under the Ipsen Collaboration Agreement, the Company recorded deferred revenue of $29,330 related to the Ipsen upfront license fee and expense reimbursement which was being amortized into revenue on a straight-line basis over the estimated development period for toremifene in the European Territory.
In the first quarter of 2011, the Company and Ipsen mutually agreed to terminate the Ipsen Collaboration Agreement, as then amended. During the first quarter of 2011, the Company recognized as collaboration revenue all of the remaining $8,066 unamortized revenue that was deferred as of December 31, 2010. This amount is included in collaboration revenue for the year ended December 31, 2011. The Company recognized as collaboration revenue $1,930 and $5,777 for the years ended December 31, 2010 and 2009, respectively, from the amortization of the Ipsen deferred revenue.
Merck & Co., Inc. Collaboration and License Agreement
In December 2007, GTx and Merck & Co., Inc. (“Merck”) entered into a global exclusive license and collaboration agreement (the “Merck Collaboration Agreement”) governing the Company’s and Merck’s joint research, development and commercialization of SARM compounds and related SARM products for all potential indications of interest. In March 2010, the Company reacquired full rights to its SARM program, including enobosarm, following the termination by the Company and Merck of the Merck Collaboration Agreement.
F-18
Table of Contents
GTx, Inc.
NOTES TO FINANCIAL STATEMENTS
(in thousands, except share and per share data)
Under the Merck Collaboration Agreement, the Company granted Merck an exclusive worldwide license under its SARM-related patents and know-how. The Company conducted preclinical research of SARM compounds and products, and Merck was primarily responsible under the terms of the agreement for conducting and funding development and commercialization of products developed under the Merck Collaboration Agreement. Merck paid the Company an upfront licensing fee of $40,000 and purchased approximately $30,000 of the Company’s common stock. In addition, Merck paid the Company $15,000 in cost reimbursements for research and development activities in equal annual installments over a three year period.
The Company deferred the recognition of the upfront licensing fee of $40,000 and the $10,800 in equity premium received that represented the difference between the purchase price and the closing price of the Company’s common stock on the date the stock was purchased by Merck. These amounts were being recognized as collaboration revenue over the period of the Company’s performance obligation, which the Company estimated to be ten years. The $5,000 of cost reimbursements received in both December 2008 and December 2009 were being recognized as collaboration revenue over the remaining period of the Company’s performance obligation. In March 2010, the Company reacquired full rights to the Company’s SARM program following the termination by the Company and Merck of the Merck Collaboration Agreement. In the first quarter of 2010, the Company recognized $54,856 as collaboration revenue, which consisted of the remaining $49,856 unamortized revenue that was deferred as of December 31, 2009, as well as the final $5,000 research and development activities cost reimbursement due under the terms of the Merck Collaboration Agreement in December 2010 for which the Company had no further performance obligation. The Company recognized as collaboration revenue $5,664 for the year ended December 31, 2009 from the amortization of the Merck deferred revenue.
University of Tennessee Research Foundation License Agreements
The Company and UTRF are parties to the SARM License Agreement pursuant to which the Company was granted exclusive worldwide rights in all existing SARM technologies owned or controlled by UTRF, and certain improvements thereto, and exclusive rights to certain future SARM technology that may be developed by certain scientists at the University of Tennessee or subsequently licensed to UTRF under certain existing inter-institutional agreements with The Ohio State University. Under the SARM License Agreement, the Company is obligated to pay UTRF annual license maintenance fees, low single-digit royalties on net sales of products and mid single-digit royalties on sublicense revenues.
Additionally, the Company and UTRF previously entered into the SERM License Agreement pursuant to which the Company was granted exclusive worldwide rights to UTRF’s method of use patents relating to SERMs, including toremifene for chemoprevention of prostate cancer. In light of the Company’s decision to discontinue further clinical development of toremifene 20 mg, the Company exercised its right to terminate the SERM License Agreement with UTRF during the first quarter of 2011.
F-19
Table of Contents
GTx, Inc.
NOTES TO FINANCIAL STATEMENTS
(in thousands, except share and per share data)
9. Income Taxes
Deferred income taxes reflect the net tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes. The principal components of the Company’s net deferred income tax assets and liabilities consisted of the following:
| December 31, | ||||||||
| 2011 | 2010 | |||||||
| Deferred income tax assets: |
||||||||
| Net federal and state operating loss carryforwards |
$ | 101,777 | $ | 87,766 | ||||
| Research and development credits |
10,921 | 9,776 | ||||||
| Deferred revenue |
— | 3,215 | ||||||
| Share-based compensation |
8,307 | 7,332 | ||||||
| Depreciation and amortization |
1,334 | 776 | ||||||
| Other |
368 | 624 | ||||||
|
|
|
|
|
|||||
| Total deferred tax assets |
122,707 | 109,489 | ||||||
|
|
|
|
|
|||||
| Valuation allowance |
(122,707 | ) | (109,489 | ) | ||||
|
|
|
|
|
|||||
| Net deferred tax assets and liabilities |
$ | — | $ | — | ||||
|
|
|
|
|
|||||
Realization of deferred tax assets is dependent upon future earnings, if any, the timing and amount of which are uncertain. Accordingly, due to the Company’s history of net operating losses, the net deferred tax assets have been fully offset by a valuation allowance. The valuation allowance increased by $13,218 in 2011, decreased by $3,878 in 2010 and increased by $17,979 in 2009.
At December 31, 2011, the Company had net federal operating loss carryforwards of approximately $262,874, which expire from 2018 to 2031 if not utilized. The Company had state operating loss carryforwards of approximately $247,421, which expire from 2012 to 2026 if not utilized. The Company also had research and development credits of approximately $10,921, which expire from 2020 to 2031 if not utilized.
Both of the net federal and state operating loss carryforwards include approximately $2,141 of deductions related to the exercise of stock options. This amount represents an excess tax benefit and has not been included in the gross deferred tax asset reflected for net federal and state operating loss carryforwards. If utilized, the benefits from these deductions will be recorded as an adjustment to additional paid in capital.
The Company will recognize the impact of a tax position in the financial statements if that position is more likely than not of being sustained on audit based on the technical merits of the position. As of December 31, 2011, the Company had no unrecognized tax benefits. Utilization of the Company’s net operating loss carryforwards may be subject to a substantial annual limitation due to ownership change limitations provided by the Internal Revenue Code of 1986, as amended and similar state provisions. The annual limitations may result in the expiration of net operating loss carryforwards before utilization. The Company has not yet performed a Section 382 change in control study in order to determine if there is a limitation of its net operating loss carryforwards. Until this study is performed, the Company cannot be certain of the use of these loss carryforwards. Additionally, the Company has not yet conducted an in depth study of its research and development credits. This study may result in an increase or decrease to the Company’s research and development credits. Until studies are conducted of the Company’s net operating loss carryforwards and research and development credits, no amounts are being presented as an uncertain tax position. The Company’s net deferred tax assets have been fully offset by a valuation allowance. Therefore, future changes to the Company’s unrecognized tax benefits would be offset by an adjustment to the valuation allowance and there would be no impact on the Company’s balance sheet, statement of operations, or cash flows. The Company does not expect its unrecognized tax benefits to change significantly over the next 12 months.
The Company is currently open to audit under the statute of limitations by the Internal Revenue Service and the appropriate state income taxing authorities for all years due to the net loss carryforwards from those years. The Company is currently not under examination by the Internal Revenue Service or any other taxing authorities. The Company has not recorded any interest and penalties on any unrecognized tax benefits since its inception.
F-20
Table of Contents
GTx, Inc.
NOTES TO FINANCIAL STATEMENTS
(in thousands, except share and per share data)
For the year ended December 31, 2009, the Company recognized a federal income tax benefit of $238 due to the adoption of a provision in the Housing and Economic Recovery Act of 2008 that allowed the Company to claim refunds for portions of its pre-2006 research and development tax credits.
10. Directors’ Deferred Compensation Plan
Non-employee directors may defer all or a portion of their fees under the Company’s Directors’ Deferred Compensation Plan until termination of their status as directors. Deferrals can be made into a cash account, a stock account, or a combination of both. Stock accounts will be paid out in the form of Company common stock, except that any fractional shares will be paid out in cash valued at the then current market price of the Company’s common stock. Cash accounts and stock accounts under the Directors’ Deferred Compensation Plan are credited with interest or the value of any cash and stock dividends, respectively. Non-employee directors are fully vested in any amounts that they elect to defer under the Directors’ Deferred Compensation Plan.
For the years ended December 31, 2011, 2010 and 2009, the Company incurred non-employee director fee expense of $260, $279 and $298, respectively, of which $178, $187 and $178 was deferred into stock accounts and will be paid in common stock following separation from service as a director. At December 31, 2011, 131,552 shares of the Company’s common stock had been credited to individual director stock accounts under the Directors’ Deferred Compensation Plan, and no amounts had been credited to individual director cash accounts under the Directors’ Deferred Compensation Plan.
11. 401(k) Plan
The Company sponsors a 401(k) retirement savings plan that is available to all eligible employees. The plan is intended to qualify under Section 401(k) of the Internal Revenue Code of 1986, as amended. The plan provides that each participant may contribute up to a statutory limit of their pre-tax compensation which was $16.5 for employees under age 50 and $22 for employees 50 and older in calendar year 2011. Employee contributions are held in the employees’ name and invested by the plan’s trustee. The plan also permits the Company to make matching contributions, subject to established limits. The Company elected to match a portion of employee’s contributions to the plan in the amount of $388, $433 and $551 in 2011, 2010 and 2009, respectively.
12. Commitments and Contingencies
Operating Lease Commitments
The Company leases laboratory facilities and office space pursuant to a sublease, which is accounted for as an operating lease. The sublease expires on December 31, 2012, with an option to extend the sublease for an additional two years. The Company subleases additional office space under a sublease that is accounted for as an operating lease. This sublease has escalating rent payments and expires on April 30, 2015. Total rent expense under these operating leases was approximately $933, $1,508 and $1,458 for the years ended December 31, 2011, 2010 and 2009, respectively.
As of December 31, 2011, annual minimum payments under operating lease arrangements were as follows:
| 2012 |
$ | 933 | ||
| 2013 |
538 | |||
| 2014 |
553 | |||
| 2015 |
185 | |||
|
|
|
|||
| Total |
$ | 2,209 | ||
|
|
|
F-21
Table of Contents
GTx, Inc.
NOTES TO FINANCIAL STATEMENTS
(in thousands, except share and per share data)
Equipment Financing and Capital Lease Obligations
As of December 31, 2011, 2010 and 2009, the Company had approximately $268 of property and equipment that was financed or acquired through a capital lease. Amortization expense for these assets is included in depreciation expense, with accumulated amortization of $194, $128 and $24 at December 31, 2011, 2010 and 2009, respectively.
As of December 31, 2011, the annual minimum payments under these financing and capital lease arrangements were as follows:
| 2012 |
$ | 85 | ||
| 2013 |
7 | |||
| 2014 |
2 | |||
|
|
|
|||
| Total |
$ | 94 | ||
|
|
|
Purchase Commitments
The Company had outstanding contractual purchase obligations of $12 and $7 at December 31, 2011 and 2010, respectively. These outstanding contractual purchase obligations are not recorded in the accompanying financial statements as the amounts represent future obligations, not liabilities, at December 31, 2011 and 2010, respectively.
13. Subsequent Events
The Company has evaluated all events or transactions that occurred after December 31, 2011 up through the date the financial statements were issued.
On February 21, 2012, the Company announced that the FDA had placed a clinical hold on the Company’s clinical trials evaluating Capesaris® for primary (first line) androgen deprivation therapy for advanced prostate cancer and secondary (second line) hormonal treatment. A clinical hold is a notification issued by the FDA to the trial sponsor to delay a clinical trial or suspend an ongoing clinical trial. The clinical hold, which was effective as of February 17, 2012, affects the Company’s Phase II maintenance dose finding clinical trial and its Phase II loading dose finding clinical trial, as well as its Phase II clinical trial in men with castration resistant prostate cancer. The clinical hold followed the Company’s reports to the FDA of venous thromboembolic events, or blood clots, in subjects treated with Capesaris® at the doses being studied in the trials (1000 mg and higher). There were two deaths in subjects treated with Capesaris® and two deaths in subjects treated with Lupron Depot®. As a result of the clinical hold, the Company suspended further enrollment into these three studies and notified clinical sites to discontinue treatment of subjects with Capesaris®.
Subject to further input from the FDA, the Company believes there may be a path forward to develop Capesaris® at lower doses to treat men with metastatic hormone sensitive prostate cancer and men with castration resistant prostate cancer. The Company plans to work with the FDA to determine the appropriate course of action, if any, to evaluate Capesaris® in these patient populations. This subsequent event does not materially impact the valuation of the Company’s assets.
There were no other material recognizable or nonrecognizable subsequent events during the period evaluated.
F-22
Table of Contents
GTx, Inc.
NOTES TO FINANCIAL STATEMENTS
(in thousands, except share and per share data)
14. Quarterly Financial Data (Unaudited)
The following is a summary of the quarterly results of operations for the years ended December 31, 2011 and 2010:
| 2011 Quarters Ended | ||||||||||||||||
| March 31 | June 30 | September 30 | December 31 | |||||||||||||
| Revenues: |
||||||||||||||||
| Product sales, net |
$ | 1,229 | $ | 1,645 | $ | 2,029 | $ | 1,770 | ||||||||
| Collaboration revenue |
8,066 | — | — | — | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total revenues |
9,295 | 1,645 | 2,029 | 1,770 | ||||||||||||
| Costs and expenses: |
||||||||||||||||
| Cost of product sales |
205 | 264 | 311 | 275 | ||||||||||||
| Research and development expenses |
7,303 | 7,591 | 8,181 | 8,863 | ||||||||||||
| General and administrative expenses |
4,684 | 4,470 | 2,904 | 3,380 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total costs and expenses |
12,192 | 12,325 | 11,396 | 12,518 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Loss from operations |
(2,897 | ) | (10,680 | ) | (9,367 | ) | (10,748 | ) | ||||||||
| Other income, net |
302 | 7 | 23 | 66 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net loss |
$ | (2,595 | ) | $ | (10,673 | ) | $ | (9,344 | ) | $ | (10,682 | ) | ||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net loss per share: |
||||||||||||||||
| Basic and diluted |
$ | (0.05 | ) | $ | (0.21 | ) | $ | (0.15 | ) | $ | (0.17 | ) | ||||
|
|
|
|
|
|
|
|
|
|||||||||
| 2010 Quarters Ended | ||||||||||||||||
| March 31 | June 30 | September 30 | December 31 | |||||||||||||
| Revenues: |
||||||||||||||||
| Product sales, net |
$ | 799 | $ | 599 | $ | 960 | $ | 1,469 | ||||||||
| Collaboration revenue |
55,778 | 336 | 336 | 336 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total revenues |
56,577 | 935 | 1,296 | 1,805 | ||||||||||||
| Costs and expenses: |
||||||||||||||||
| Cost of product sales |
151 | 134 | 216 | 267 | ||||||||||||
| Research and development expenses |
7,650 | 9,477 | 5,593 | 5,775 | ||||||||||||
| General and administrative expenses |
4,509 | 4,325 | 4,066 | 4,519 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total costs and expenses |
12,310 | 13,936 | 9,875 | 10,561 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Income (loss) from operations |
44,267 | (13,001 | ) | (8,579 | ) | (8,756 | ) | |||||||||
| Other income, net |
72 | 60 | 4 | 1,227 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net income (loss) |
$ | 44,339 | $ | (12,941 | ) | $ | (8,575 | ) | $ | (7,529 | ) | |||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net income (loss) per share: |
||||||||||||||||
| Basic and diluted |
$ | 1.22 | $ | (0.36 | ) | $ | (0.24 | ) | $ | (0.16 | ) | |||||
|
|
|
|
|
|
|
|
|
|||||||||
F-23
