Attached files
Table of Contents
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
| x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(D) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2010
or
| ¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(D) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission file number 1-15103
INVACARE CORPORATION
(Exact name of Registrant as specified in its charter)
| Ohio | 95-2680965 | |
| (State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer Identification Number) |
One Invacare Way, P.O. Box 4028, Elyria, Ohio 44036
(Address of principal executive offices) (Zip Code)
Registrant’s telephone number, including area code: (440) 329-6000
Securities registered pursuant to Section 12(b) of the Act:
| Title of Each Class |
Name of Exchange on which Registered | |
| Common Shares, without par value Rights to Purchase Preferred Shares, without par value |
New York Stock Exchange New York Stock Exchange |
Securities registered pursuant to Section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined by Rule 405 of the Securities Act. Yes ¨ No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ¨ No x
Indicate by check mark whether the Registrant (1) has filed all reports to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the Registrant was required to file such reports) and (2) has been subject to the filing requirements for the past 90 days. Yes x No ¨
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such short period that the registrant was required to submit and post such files). Yes ¨ No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (Section229.405) is not contained herein, and will not be contained, to the best of the Registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See definitions of “large accelerated filer”, “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer ¨ | Accelerated filer x | |
| Non-accelerated filer ¨ | Smaller reporting company ¨ |
Indicate by check mark whether the registrant is a shell company (as defined by Rule 12b-2 of the Act). Yes ¨ No x
As of June 30, 2010, the aggregate market value of the 28,334,691 Common Shares of the Registrant held by non-affiliates was $587,661,491 and the aggregate market value of the 17,342 Class B Common Shares of the Registrant held by non-affiliates was $359,673. While the Class B Common Shares are not listed for public trading on any exchange or market system, shares of that class are convertible into Common Shares at any time on a share-for-share basis. The market values indicated were calculated based upon the last sale price of the Common Shares as reported by The New York Stock Exchange on June 30, 2010, which was $20.74. For purposes of this information, the 2,954,236 Common Shares and 1,080,174 Class B Common Shares which were held by Executive Officers and Directors of the Registrant were deemed to be the Common Shares and Class B Common Shares held by affiliates.
As of February 23, 2011, 31,316,801 Common Shares and 1,084,947 Class B Common Shares were outstanding.
Documents Incorporated By Reference
Portions of the Registrant’s definitive Proxy Statement to be filed in connection with its 2011 Annual Meeting of Shareholders are incorporated by reference into Part III (Items 10, 11, 12, 13 and 14) of this report.
Except as otherwise stated, the information contained in this Annual Report on Form 10-K is as of December 31, 2010.
Table of Contents
2010 ANNUAL REPORT ON FORM 10-K CONTENTS
| Item |
Page | |||||
| PART I: | ||||||
| 1. |
I-3 | |||||
| 1A. |
I-17 | |||||
| 1B. |
I-29 | |||||
| 2. |
I-29 | |||||
| 3. |
I-32 | |||||
| I-32 | ||||||
| PART II: | ||||||
| 5. |
I-34 | |||||
| 6. |
I-37 | |||||
| 7. |
Management’s Discussion and Analysis of Financial Condition and Results of Operations |
I-38 | ||||
| 7A. |
I-52 | |||||
| 8. |
I-52 | |||||
| 9. |
Changes in and Disagreements with Accountants on Accounting and Financial Disclosure |
I-52 | ||||
| 9A. |
I-52 | |||||
| 9B. |
I-53 | |||||
| PART III: | ||||||
| 10. |
I-54 | |||||
| 11. |
I-54 | |||||
| 12. |
Security Ownership of Certain Beneficial Owners and Management and Related Shareholder Matters |
I-54 | ||||
| 13. |
I-54 | |||||
| 14. |
I-54 | |||||
| PART IV: | ||||||
| 15. |
I-55 | |||||
| I-56 | ||||||
I-2
Table of Contents
PART I
| Item 1. | Business. |
GENERAL
Invacare Corporation is the world’s leading manufacturer and distributor in the estimated $11.0 billion worldwide market for medical equipment and supplies used in the home based upon its distribution channels, breadth of product line and net sales. The company designs, manufactures and distributes an extensive line of health care products for the non-acute care environment, including the home health care and extended care markets. The company continuously revises and expands its product lines to meet changing market demands and currently offers numerous product lines. The company sells its products principally to over 25,000 home health care and medical equipment providers, distributors and government locations in the United States, Australia, Canada, Europe, New Zealand and Asia. Invacare’s products are sold through its worldwide distribution network by its sales force, telesales associates and various organizations of independent manufacturers’ representatives and distributors. The company also distributes medical equipment and disposable medical supplies manufactured by others.
Invacare is committed to design and deliver the best value in medical products, which promote recovery and active lifestyles for people requiring home and other non-acute health care. Invacare pursues this vision by:
| • | designing and developing innovative and technologically superior products; |
| • | ensuring continued focus on the company’s primary market—the non-acute health care market; |
| • | marketing the company’s broad range of products; |
| • | driving efficiency and innovation through the use of the company’s global resources; |
| • | providing a professional and cost-effective sales, customer service and distribution organization; |
| • | supplying innovative provider support and aggressive product line extensions; |
| • | building a strong referral base among health care professionals; |
| • | continuously advancing and recruiting top management candidates; |
| • | empowering all employees; |
| • | providing a performance-based reward environment; and |
| • | continually striving for total quality throughout the organization. |
When the company was acquired in December 1979 by a group of investors, including some of its current officers and Directors, it had $19.5 million in net sales and a limited product line of standard wheelchairs and patient aids. In 2010, Invacare reached approximately $1.7 billion in net sales, representing a 16% compound average sales growth rate since 1979, and, based upon the company’s distribution channels, breadth of product line and net sales, currently is the leading company in each of the following major, non-acute, medical equipment categories: power and manual wheelchairs, home care bed systems and home oxygen systems.
The company’s executive offices are located at One Invacare Way, Elyria, Ohio, 44036 and its telephone number is (440) 329-6000. In this report, “Invacare” and the “company” refer to Invacare Corporation and, unless the context otherwise indicates, its consolidated subsidiaries.
I-3
Table of Contents
THE HOME MEDICAL EQUIPMENT INDUSTRY
North America Market
The home medical equipment (HME) market includes home health care products, physical rehabilitation products and other non-disposable products used for the recovery and long-term care of patients. The company believes that patients overwhelmingly prefer care and treatment in their home. There is a growing body of evidence that homecare generally results in faster recovery and better outcomes. Homecare is often more cost-effective and comfortable than institutional care by a considerable factor. A principal reason is that homecare patients are not exposed to today’s increasingly virulent strains of hospital-borne pathogens. Invacare, through its diverse product and service offerings, delivers what the company refers to as a “medical trifecta”: patient satisfaction; better outcomes; and lower costs. The company’s view is an adequately equipped home is a better recovery option for a significant number of patients who face hospitalization. Accordingly, demand for domestic home medical equipment products is expected to grow during the next decade and beyond as a result of the factors mentioned above and more, including:
Growth in Population over Age 65. Globally, overall life expectancy continues to increase. Recent reports from the U.S. Department of Health and Human Services (DHHS) state that the average life expectancy in the United States for men and women who reach the age of 65 is now 82 and 85, respectively. Furthermore, life expectancy in the United States at birth is now an average of 78 for men and women together, a record high. The DHHS also reports that people age 75 or older represent the vast majority of home health care patients and will increase to 12% of the population by the year 2050. The oldest of the “Baby Boomer” generation, which numbers roughly 78 million people, will begin to turn 65 in 2011 and for the next 18 years.
Treatment Trends. The company believes that many medical professionals and patients prefer home health care over institutional care because home health care results in greater patient independence, increased patient responsibility and improved responsiveness to treatment. Further, health care professionals, public payors and private payors appear to favor home care as a cost-effective, clinically appropriate alternative to facility-based care. Recent surveys show that approximately 70% of adults would rather recover from an accident or illness in their home, and approximately 90% of the population aged 65 and over showed a preference for home-based, long-term care. In addition, the number of hospital beds per capita has fallen over the past twenty-five years in the United States, a trend which is expected to continue. This decline has coincided with the reduction in average length of stays in hospitals.
Technological Trends. Technological advances have made medical equipment increasingly adaptable for use in the home. Current hospital procedures often allow for earlier patient discharge, thereby lengthening recuperation periods outside of the traditional institutional setting. In addition, continuing medical advances prolong the lives of adults and children, thus increasing the demand for home medical care equipment.
Health Care Cost Containment Trends. Health care expenditures in the United States for 2009 were estimated to be $2.5 trillion dollars or approximately 17.6% of the Gross Domestic Product (GDP), the highest among industrialized countries. It is now estimated that federal, state and local government spending on health care in the U.S. will soon exceed private health care spending for the first time. By 2019, the nation’s health care spending is projected to increase to $4.5 trillion, growing at an average annual rate of 7.0%. Over this same period, spending on health care is expected to be approximately 19.3% of GDP. The rising cost of health care has caused many payors of health care expenses to look for ways to contain costs. The company believes that home health care and home medical equipment will play a significant role in reducing health care costs. In fact, a recent study conducted by Frank Lichtenberg, the Courtney C. Brown Professor of Business at the Columbia University Graduate School of Business and a Research Associate with the National Bureau of Economic Research, found that a nationwide increase in the use of home health care can save the U.S. billions of dollars in hospital costs. The study estimates the United States may have
I-4
Table of Contents
saved as much as $25 billion in total hospital payroll costs in 2008 alone thanks to the growth of the home health care sector during the previous 10 years. The study mentions that “it is a reasonable calculation” that further savings will be realized in the years ahead if the use of home care continues to grow.
Society’s Mainstreaming of People with Disabilities. People with disabilities are increasingly a part of the fabric of society, in part due to the 1991 Americans with Disabilities Act, or the “ADA.” This legislation provides mainstream opportunities to people with disabilities. The ADA imposes requirements on certain components of society to make reasonable accommodations to integrate people with disabilities into the community and the workplace.
Distribution Channels. The changing home health care market continues to provide new ways of reaching the consumer. The distribution network for products has expanded to include not only specialized home health care providers and extended care facilities but also retail drug stores, surgical supply houses, rental, hospital and HMO-based stores, home health agencies, mass merchandisers and the Internet.
Europe/Asia/Pacific Market
The company believes that, while many of the market factors influencing demand in the U.S. are also present in Europe and Asia/Pacific—aging of the population, technological trends and society’s acceptance of people with disabilities—each of the markets of Europe and in Asia/Pacific have distinctive characteristics. The health care industry tends to be more heavily socialized and, therefore, is more influenced by government regulation and fiscal policy. Variations in product specifications, regulatory approval processes, distribution requirements and reimbursement policies require the company to tailor its approach to the local market. Management believes that as the European markets develop more common product requirements and the company continues to refine its distribution channels, the company can more effectively penetrate these markets. Likewise, the company expects to increase its sales in the highly fragmented Australian, New Zealand and Asian markets as these markets, and the company’s distribution within them, develop.
United States/Europe Market
The company is directly affected by government regulation and reimbursement policies in virtually every country in which the company operates. In the United States, the growth of health care costs has increased at rates in excess of the rate of inflation and as a percentage of GDP for several decades. A number of efforts to control the federal deficit have impacted reimbursement levels for government sponsored health care programs, and private insurance companies and state Medicaid programs peg their reimbursement levels to Medicare.
Reimbursement guidelines in the home health care industry have a substantial impact on the nature and type of equipment an end-user can obtain and, thus, affect the product mix, pricing and payment patterns of the company’s customers who are medical equipment providers. The company believes its strong market position and technical expertise will allow it to respond to ongoing regulatory changes. However, the issues described above will likely continue to have significant impacts on the pricing of the company’s products.
GEOGRAPHICAL SEGMENTS AND PRODUCT CATEGORIES
North America
North America includes: North America/Home Medical Equipment (NA/HME), Invacare Supply Group (ISG) and Institutional Products Group (IPG).
NA/HME
This segment includes: Rehab, Standard and Respiratory product lines as discussed below.
I-5
Table of Contents
REHAB PRODUCTS
Power Wheelchairs. Invacare manufactures a complete line of power wheelchairs for individuals who require independent powered mobility. The range includes products that can be significantly customized to meet an individual’s specific needs, as well as products that are inherently versatile and meet a broad range of individual requirements. Center-wheel drive power wheelchair lines are marketed under the Invacare® TDX® brand names and include a full range of powered mobility products. The TDX® line of power wheelchairs offer an unprecedented combination of power, stability and maneuverability. The Pronto® Series Power Wheelchairs with SureStep® Stability feature center-wheel drive performance for exceptional maneuverability and intuitive driving. Power tilt and recline systems are offered as well.
Custom Manual Wheelchairs. Invacare manufactures and markets a range of custom manual wheelchairs for everyday, sports and recreational uses. These lightweight chairs are marketed under the Invacare® and Invacare Top End® brand names. The chairs provide mobility for people with moderate to severe disabilities in their everyday activities as well as for use in various sports such as basketball, racing and tennis.
Personal Mobility. Invacare manufactures and distributes personal mobility products, including compact scooters available in three-wheel and four-wheel versions.
Seating and Positioning Products. Invacare markets seat cushions, back supports and accessories under three series: the Invacare® Absolute™ Series provides simple seating solutions for comfort, fit and function; the Invacare InTouch™ Series includes versatile modular seating, providing optimal rehab solutions; and the Invacare PinDot® Series offers custom seating solutions personalized for the most challenged clients. The company also markets specialty seating products, pediatric seating and wheelchairs as well as various standers that allow people to stand that otherwise would be unable.
STANDARD PRODUCTS
Manual Wheelchairs. Invacare’s manual wheelchairs are sold for use inside and outside the home, institutional settings or public places. Clients include people who are chronically or temporarily disabled and require basic mobility performance with little or no frame modification. Examples of the company’s manual wheelchair lines, which are marketed under the Invacare® brand name, include the 9000 and the Tracer® product lines. These wheelchairs are designed to accommodate the diverse capabilities and unique needs of the individual, from petite to bariatric sizes.
Personal Care. Invacare is principally a distributor of a full line of personal care products, including ambulatory aids such as crutches, canes, walkers, knee walkers and wheeled walkers. Also available are safety aids such as tub transfer benches, shower chairs and grab bars, and patient care products such as commodes and other toilet assist aids.
Home Care Beds. Invacare manufactures and distributes a wide variety of manual, semi-electric and fully-electric beds for home use under the Invacare® brand name. Home care bed accessories include bedside rails, mattresses, overbed tables and trapeze bars. Also available are bariatric beds and accompanying accessories to serve the special needs of bariatric patients.
Low Air Loss Therapy Products. Invacare distributes a complete line of mattress overlays and replacement products, under the Invacare® Solace® and microAIR® brand names. These products, which use either pressure reducing foam or air flotation to redistribute weight and move moisture away from patients, assist in the total care of those who are immobile and spend a great deal of time in bed.
Patient Transport. Invacare manufactures and/or distributes products needed to assist in transferring individuals from surface to surface (bed to chair) or transporting from room to room. Designed for use in the home and institutional settings, these products include patient lifts and slings, and a series of mobile, multi-functional recliners.
I-6
Table of Contents
RESPIRATORY PRODUCTS
Non-Delivery Oxygen. Trends in the industry continue to be towards a non-delivery oxygen model. The Invacare® HomeFill® Oxygen System is the standard in ambulatory oxygen technology. Approaching 200,000 units in the field, it is the basis for a non-delivery model and allows patients to fill their own high-pressure cylinders from a concentrator in their home. With some upfront investment, the long-term benefits are unmatched, allowing providers to virtually eliminate time-consuming and costly service calls associated with cylinder and/or liquid oxygen deliveries.
Rounding out Invacare’s non-delivery oxygen offerings are the Invacare® SOLO2® Transportable Concentrator and the Invacare® XPO2™ Portable Concentrator, which are now both approved by the FAA for use in flight. The SOLO2® offers continuous flow oxygen up to 3 LPM or pulse dose oxygen in settings 1-5. It is a flexible, reliable and clinically robust system that is easy to operate. Named for its extreme portability, the XPO2™ weighs just 6 pounds with pulse dose settings 1-5 to meet the needs of a broad range of patients.
Stationary Oxygen Concentrators. Invacare oxygen concentrators are manufactured under the Perfecto2™ name and are available in five and 10 liter models. All Invacare stationary concentrators provide patients with durable equipment and reliable oxygen either at home or in a healthcare setting.
Aerosol Products and Oxygen Accessories. Invacare offers a family of aerosol compressors under the Stratos™ name. Invacare also has an expanded line of conservers and regulators to maximize the efficiency of oxygen cylinders.
OTHER PRODUCTS
Other products include various services, including repair services, equipment rentals, accounts receivable collections and external contracting.
Invacare Supply Group (ISG)
Invacare distributes numerous lines of branded medical supplies including ostomy, incontinence, diabetic, interals, wound care and urology products as wells as home medical equipment, including aids for daily living.
Institutional Products Group (IPG)
Invacare, operating as Invacare Continuing Care, Invacare Continuing Care Canada and Champion, is a manufacturer and marketer of healthcare furnishings including beds, case goods and patient handling equipment for the long-term care markets, specialty clinical recliners for dialysis and oncology clinics and certain other home medical equipment and accessory products.
Asia/Pacific
The company’s Asia/Pacific operations consist of Invacare Australia, which distributes the Invacare range of products which includes: manual and power wheelchairs, lifts, ramps, beds, furniture and pressure care products; Dynamic Controls, a manufacturer of electronic operating components used in power wheelchairs, scooters, respiratory and other products; and Invacare New Zealand, a distributor of a wide range of home medical equipment.
Europe
The company’s European operations operate as a “common market” company with sales throughout Europe. The European operations currently sell a line of products providing room for growth as Invacare continues to broaden its product line offerings in line with its globalization strategy.
I-7
Table of Contents
Most wheelchair products sold in Europe are designed locally to meet specific market requirements. The company manufactures and/or assembles both manual and power wheelchair products in the following countries: United Kingdom, France and Germany. Manual wheelchair products are also manufactured and/or assembled in Portugal, Switzerland and Sweden. Beds are assembled in Denmark and Portugal. Personal care products are manufactured in Germany; and Dolomite products are manufactured in Sweden. Oxygen products such as concentrators and HomeFill® Oxygen Systems are imported from Invacare U.S. or China operations.
For information relating to net sales by product group, see Business Segments in the Notes to the Consolidated Financial Statements included in this report.
WARRANTY
Generally, the company’s products are covered from the date of sale to the customer by warranties against defects in material and workmanship for various periods depending on the product. Certain components carry a lifetime warranty.
COMPETITION
North America and Asia/Pacific
The home medical equipment market is highly competitive and Invacare products face significant competition from other well-established manufacturers and distributors. The company believes that its success in increasing market share is dependent on providing value to the customer based on the quality, performance and price of the company products, the range of products offered, the technical expertise of the sales force, the effectiveness of the company distribution system, the strength of the dealer and distributor network and the availability of prompt and reliable service for its products. Various competitors, from time to time, have instituted price-cutting programs in an effort to gain market share and may do so again in the future.
Europe
As a result of the differences encountered in the European marketplace, competition generally varies from one country to another. The company typically encounters one or two strong competitors in each country, some of them becoming regional leaders in specific product lines.
MARKETING AND DISTRIBUTION
North America and Asia/Pacific
Invacare products are marketed in the United States and Asia/Pacific primarily to providers who in turn sell or rent these products directly to consumers within the non-acute care setting. Invacare’s primary customer is the home medical equipment (HME) provider. The company also employs a “pull-through” marketing strategy to medical professionals, including physical and occupational therapists, who refer their patients to HME providers to obtain specific types of home medical equipment.
Invacare’s domestic sales and marketing organization consists primarily of a homecare sales force, which markets and sells Invacare® branded products to HME providers. Each member of Invacare’s home care sales force functions as a Territory Business Manager (TBM) and handles all product and service needs for an account, thus saving customers’ valuable time. The TBM also provides training and servicing information to providers, as well as product literature, point-of-sale materials and other advertising and merchandising aids. In Canada, products are sold by a sales force and distributed through regional distribution centers to health care providers throughout Canada.
I-8
Table of Contents
The Inside Sales Department provides increased sales coverage of smaller accounts and complements the efforts of the field sales force. Inside Sales offers cost-effective sales coverage through a targeted telesales effort, and has delivered solid sales growth since its existence.
Invacare’s Technical Education department offers education programs that continue to place emphasis on improving the productivity of repair technicians. The Service Referral Network includes numerous providers who honor the company’s product warranties regardless of where the product was purchased. This network of servicing providers seeks to ensure that all consumers using Invacare products receive quality service and support that is consistent with the Invacare brand promise.
Additionally, Invacare is the only manufacturer with a breadth of service offerings that includes the ability to assist providers in the collection of outstanding co-pays, rental capabilities, software and technology to streamline efficiencies, repair services and replacement parts. These tools and resources assist home and long-term care providers in maximizing efficiency and furthering their business success.
The company markets products and services to the institutional care market through IPG. IPG products include beds and furnishings, patient handling, bathing, durable medical equipment and clinical therapies, such as therapeutic support surfaces. IPG sales and marketing organizations consist of field sales representatives and independent rep agencies supported by a marketing group that generates awareness and demand at institutions for Invacare products and services. IPG also provides interior design services for nursing homes and assisted living facilities involved with renovation and new construction.
In 2010, Invacare continued to focus on a growing suite of programs and services designed to simplify business for HME providers, reduce their costs, optimize their resources and improve their bottom line. Invacare is working to help HME providers respond to the challenges associated with competitive bidding, escalating operating costs and changes in Medicare reimbursement through products, services and business consulting.
The company sells distributed products, primarily soft goods and disposable medical supplies, through ISG. ISG products include ostomy, incontinence, wound care and diabetic supplies, as well as 40 other categories of other soft goods and disposables. ISG markets its products through field account managers, inside telesales, a customer service department and the Internet. Additionally, ISG entered the long-term care market on a regional basis and markets to those nursing homes utilizing independent manufacturer representatives. ISG also markets a Home Delivery Program to home medical equipment providers through which ISG drop ships supplies in the provider’s name to the customer’s address. Thus, providers have no products to stock, no minimum order requirements and delivery is made within 24 to 48 hours nationwide. ISG also offers many customized marketing programs as well as business to consumer and business to business website development, designed to help its customers create awareness, grow companion and cash sales and assist in patient retention.
Invacare continues to improve performance and usability on www.invacare.com. In 2010, the company implemented a new global website platform with the goal of creating a highly usable web presence and one central destination for all Invacare web users. Invacare also increased participation in online forums and engaged customers by utilizing social media tools, including a corporate blog (www.invacareconnects.com), Facebook page and YouTube channel. These moves toward a more customer-centric approach allow the company to provide a user interface that better addresses customer needs.
Also in 2010, the company continued its strategic advertising campaign in key business to business publications that reach Invacare’s respective customers. The company contributed extensively to editorial coverage in trade publications concerning the products the company manufactures; and company representatives attended numerous trade shows and conferences on a national and regional basis in which Invacare products were displayed to providers, health care professionals, managed care professionals and consumers. “Yes, you can®” continues to be Invacare’s global tagline, and it remains steadfast in company ads and on the Invacare global website and is indicative of the company’s “can do” attitude.
I-9
Table of Contents
The company continues to generate greater consumer awareness of its products. This was evidenced by the company’s sponsorship of a variety of wheelchair sporting events and support of various philanthropic causes benefiting the consumers of the company’s products. The company continued its sponsorships of individual wheelchair athletes and teams, including several of the top-ranked male and female racers, hand cyclists, and wheelchair tennis players in the world. The company also continued its support of disabled veterans through its sponsorship of the 30th National Veterans Wheelchair Games, the largest annual wheelchair sports event in the world. The games bring a competitive and recreational sports experience to military service veterans who use wheelchairs for their mobility needs due to spinal cord injury, neurological conditions or amputation.
Europe
The company’s European operations consist primarily of manufacturing, marketing and distribution operations in Western Europe and export sales activities through local distributors elsewhere in the world. The company has a sales force and where appropriate, distribution centers, in Austria, Belgium, Denmark, France, Germany, Ireland, Italy, Netherlands, Norway, Portugal, Spain, Sweden, Switzerland and the United Kingdom, and sells through distributors elsewhere in Europe and in the Middle East. In markets where the company has its own sales force, product sales are typically made through dealers of medical equipment and, in certain markets, directly to government agencies. In 2010, the continued consolidation of big buying groups tending to develop their business on a European scale has continued. As a result, Invacare is generalizing the application of pan-European pricing policies.
In 2010, Invacare was the title sponsor for the fifteenth year in a row of the “Invacare World Team Cup,” a wheelchair tennis tournament, which was in Antalya, Turkey.
PRODUCT LIABILITY COSTS
The company’s captive insurance company, Invatection Insurance Company, currently has a policy year that runs from September 1 to August 31 and insures annual policy losses of $10,000,000 per occurrence and $13,000,000 in the aggregate of the company’s North American product liability exposure. The company also has additional layers of external insurance coverage insuring up to $75,000,000 in annual aggregate losses arising from individual claims anywhere in the world that exceed the captive insurance company policy limits or the limits of the company’s per country foreign liability limits, as applicable. There can be no assurance that Invacare’s current insurance levels will continue to be adequate or available at affordable rates.
Product liability reserves are recorded for individual claims based upon historical experience, industry expertise and indications from the third-party actuary. Additional reserves, in excess of the specific individual case reserves, are provided for incurred but not reported claims based upon actuarial valuations at the time such valuations are conducted. Historical claims experience and other assumptions are taken into consideration to estimate the ultimate reserves. For example, the actuarial analysis assumes that historical loss experience is an indicator of future experience, that the distribution of exposures by geographic area and nature of operations for ongoing operations is expected to be very similar to historical operations with no dramatic changes and that the government indices used to trend losses and exposures are appropriate. Estimates made are adjusted on a regular basis and can be impacted by actual loss awards and settlements on claims. While actuarial analysis is used to help determine adequate reserves, the company is responsible for the determination and recording of adequate reserves in accordance with accepted loss reserving standards and practices.
PRODUCT DEVELOPMENT AND ENGINEERING
Invacare is committed to continuously improving upon and renewing its product offerings. Invacare’s key globalization initiative is moving from a local product development approach to address local markets, to a global product development approach, aimed at developing global product platforms. This strategy is designed to
I-10
Table of Contents
enable the company to increase the number of new products it introduces and offer more innovative product solutions, while at the same time reducing complexity within the business. By leveraging its engineering and product development capabilities on a global basis, Invacare expects to further increase its industry leadership in the broadest range of product offerings in both home care and continuing care medical device equipment.
2010’s marquee global product was the Invacare® FDX® Front-Wheel Drive Power Wheelchair. Launched in May in the United States and July in Europe, this wheelchair completes Invacare’s power wheelchair offerings with a solution for substantially all custom rehab needs. Invacare now offers its customers a full range of power bases and drive wheel configurations, including center-wheel, rear-wheel and now front-wheel drive. The FDX® Wheelchair features core technologies such as the Single Stage Drive (SSD) motor-gearbox combination, Invacare® MK6i™ Electronics and Invacare® G-Trac™ true-tracking technology.
Also globally, Invacare expanded the presence of its European Jazz Rollator by introducing it as the Invacare® FR300™ Rollator into the United States. This rollator combines a high-end, streamlined look with the clinical benefits associated with walking more inside the rollator frame. Other features include a curb climber, or integrated “pedal” that makes it easier to negotiate obstacles and a unique x-brace and folding mechanism which allows the rollator to be folded easily and with one hand for portability.
The following are some of Invacare’s other notable product developments and updates:
Global Products
The new Single Stage Drive (SSD) motor-gearbox combination has several inherent advantages over current drive technology. The new SSD drive system is more efficient than previous designs and consolidates motor-gearbox combinations globally, reducing SKUs. As a two piece unit, the new SSD motor is easier to service because the motor- gearbox modules can be replaced separately. In addition, the SSD drive technology will be completely designed and manufactured by Invacare, further enhancing overall quality.
The Invacare® PCS (positioning-comfort-stability) back is designed for optimal comfort and function. Single-point mounting hardware with quick-release latch provides secure mounting, while allowing height, depth and back angle adjustments with easy installation and removal. The spacer fabric cover improves airflow between the user and the back, increasing comfort and preventing heat and moisture build-up. Three inches of contour depth allow for centering and postural stability without interfering with hip placement. For customizable support, the PCS back comes with optional pelvic stabilizers and thoracic lateral supports to provide additional pelvic and trunk control and stability.
The Invacare® Leo™ Scooter offers a stable four-wheel base that provides a smooth, safe drive and handles varying surfaces with ease. Features include a full lighting package, built-in splash guards to protect the electronics and transaxle, comfortable seating that swivels and slides and flat free tires. There are many add-on accessories available such as a rear basket or oxygen holder.
Made from robust, yet lightweight plastic, the new Invacare® Bathboard has been developed to fit most types of baths and provides a stable and ergonomic surface for showering and personal hygiene.
The Invacare® Roze™ Stand Assist Lift is for clients who require help in standing up or in transfers from one location to another. With the ability to handle up to 200 kg, the Roze lift™ perfectly fits the requirements for a lift system for everyday use. In addition to delivering safe client handling, it reduces the burden of lifting for the caregiver. Of particular note is the wide-opening base, which together with a removable footplate and an intelligent control box makes it easy for any caregiver to accomplish transfers, quickly and safely, on a daily basis.
The Invacare® Jasmine Mobile Lift is a flexible solution that offers high comfort for all clients. The lift provides safe patient handling with up to 200 kg weight capacity. With its expansive, wide-opening base
I-11
Table of Contents
and high lifting mechanism, Jasmine makes life easier for caregivers operating this new Invacare lift system. The control box features an intelligent service monitoring system that enables a safe operating environment for the caregiver, while allowing maximum comfort for the client during each transfer.
The new Invacare® Top End® Pro chair is designed for aspiring athletes who want durability in a chair, but also need to be able to make quick and easy adjustments. Available in two versions, as suitable for basketball or tennis, the new Top End® Pro chair is available in both a short and tall frame to accommodate a range of differing body shapes.
The Invacare® Top End® Twirl wheelchair allows one to have fun, exercise and compete in wheelchair dancing with its specially developed dancing anti-tip ability and robust center-of-gravity positioning.
Local Products
The Invacare® TDX® SI-2 power wheelchair in Europe provides excellent TDX power and performance. It has 14” drive wheels to handle aggressive terrain with ease, while its integrated rear suspension offers a smooth ride. Center wheel drive and a narrow footprint provide excellent indoor maneuverability, while its clean lines and brightly painted frame combine for a simple yet elegant design. The end result is an eye-catching power wheelchair with a go-anywhere attitude.
With the new Invacare Storm®4 X-plore power wheelchair in Europe, the demanding user can enjoy all the benefits of Storm®4, combined with a four-wheel-suspension for enhanced outdoor performance, increased shock absorption / comfort and better traction on uneven ground. The G-Trac™ option is also available for the ultimate driving performance. The new Storm®4 X-plore offers the same advantages as the Storm®4 in terms of configurability, adaptability and functionality. The stylish look of Storm®4 has been conserved, with its modularity, flexible seat concept and the trouble-free servicing.
In the United States, Invacare introduced the Invacare® Insignia® Wheelchair. This high-strength lightweight chair offers height adjustable arms that convert from desk to full length, allowing providers to stock one chair instead of two, as well as a robust wheel lock, simple adjustable head tube design, sleek caster fork, adjustable angle back and quick release axles. It is a stylish, lightweight chair that is easy to use and adjusts to a patient’s specific needs.
The European küschall® K-Series family has grown with the latest addition: the K-Series Titanium. The newest K-Series features a rigid titanium wheelchair, is available in a 75° or 90° front frame angle. Available with titanium footrest, backrest and hand rims as well as a carbon axle to experience a highly active and dynamic wheelchair which is ideal for the active user.
The Invacare® Rea Spirea4NG in Europe is a dynamic and durable lightweight folding wheelchair ideal for everyday use. By incorporating user feedback into the design, the Spirea4NG offers light handling, minimal adjustment requirements, high technical quality and low maintenance, making it a perfect choice for users and care givers.
The European Invacare® SoftAIR dynamic mattresses are designed for patients at very high risk of developing pressure ulcers. There are two options available within the SoftAIR range, the SoftAIR Super, and the SoftAIR Excellence.
The newly designed Invacare® Knee Walker from the United States is a great alternative for those who are weary of discomfort related to crutches. The new design enhances stability, tracking control and patient comfort and sets a new standard for the market. The Knee Walker comes with a convenient basket and folds easily for transport and storage.
Invacare added a low-cost stationary concentrator to its product portfolio with the Invacare® Perfecto2™ V Concentrator. This concentrator meets a patient’s oxygen needs easily and effectively and is equipped with an oxygen sensor to ensure oxygen purity levels are monitored and appropriate. An alarm will also alert the patient of low flow or any issues with kinked or loose tubing. It also features easy-to-access and clean filters, which prolong the life of the equipment, as well as easy-to-access circuit breakers which allow the system to be reset easily after power surges or outages.
I-12
Table of Contents
MANUFACTURING AND SUPPLIERS
The company’s objective is to continue to reduce costs through cost reductions and possibly facility consolidation while maintaining the highest quality supply chain in the industry. The company seeks to achieve this objective through a strategic combination of Invacare manufacturing facilities, contract manufacturing facilities and key suppliers. The operational strategy further supports the marketing strategy with flexible providers of new and modified products that respond to the demands of the market.
The supply chain is focused on providing custom-configured, made-to-order manufactured products as well as high-quality, cost-effective solutions for standard stock products. As strategic choices are made globally, the company will continue to be focused on providing quick product delivery to the market as a specific competitive advantage to the marketing and sales teams in these regions.
The company continues to emphasize reducing the costs of its global manufacturing and distribution operations. Access to sourcing opportunities has been facilitated by the company’s establishment of a test and design engineering facility in the company’s Suzhou, China location. In Asia, Invacare manufactures products that serve regional market opportunities through the company’s wholly-owned factory in Suzhou, Jiangsu Province, China. The Suzhou facility supplies products to the major geographic regions of the world served by Invacare: North America, Europe and Asia/Pacific.
Best practices in lean manufacturing are used throughout the company’s operations to eliminate waste, shorten lead times, optimize inventory, improve productivity, drive quality and engage supply chain associates in the defining and implementation of needed change.
The company purchases raw materials, components, sub-assemblies and finished goods from a variety of suppliers around the world. The company’s Hong Kong-based Asian sourcing and purchasing office has proven to be a significant asset to the company’s supply chain through the identification, development and management of suppliers across Asia. Where appropriate, Invacare utilizes contracts with suppliers in all regions to increase the guarantees of delivery, cost, quality and responsiveness. In those situations where contracts are not advantageous, Invacare works to manage multiple sources of supply and relationships that provide increased flexibility to the supply chain.
North America
The company has focused its factories in North America on the production of powered mobility and custom manual wheelchairs and seating products, the fully integrated manufacture of homecare and institutional care beds, the final assembly of respiratory products and the integrated component fabrication, painting and final assembly of a variety of standard manual wheelchairs and personal care products. The company operates four major factories located in Elyria, Ohio; Sanford, Florida; London, Ontario and Reynosa, Mexico.
Asia/Pacific
The Asia/Pacific region is focused on improving its customer delivery effectiveness, expanding its reach into all customer channels in all major metropolitan centers and integrating its distribution operations across the region.
Europe
The company has nine manufacturing/assembly facilities spread throughout Europe with the capability to manufacture patient aid, wheelchair, powered mobility, bath safety, beds and patient transport products. The European manufacturing and logistics facilities are focused on accelerating opportunities for streamlining to gain productivity improvements in cost and quality over the next few years.
I-13
Table of Contents
GOVERNMENT REGULATION
The company is directly affected by government regulation and reimbursement policies in virtually every country in which it operates. Government regulations and health care policy differ from country to country, and within some countries (most notably the U.S., European Union, Australia and Canada), from state to state or province to province. Changes in regulations and health care policy take place frequently and can impact the size, growth potential and profitability of products sold in each market.
In the U.S., the growth of health care costs has increased at rates in excess of the rate of inflation and as a percentage of GDP for several decades. A number of efforts to control the federal deficit have impacted reimbursement levels for government sponsored health care programs and private insurance companies often imitate changes made in federal programs. Reimbursement guidelines in the home health care industry have a substantial impact on the nature and type of equipment an end user can obtain and thus, affect the product mix, pricing and payment patterns of the company’s customers who are the HME providers.
The company continues its pro-active efforts to shape public policy that impacts home and community-based, non-acute health care. The company is currently very active with federal legislation and regulatory policy makers. Invacare believes that these efforts give the company a competitive advantage in two ways. First, customers frequently express appreciation for the company’s efforts on behalf of the entire industry. Second, sometimes the company has the ability to anticipate and plan for changes in public policy, unlike most other HME manufacturers who must react to change after it occurs.
The United States Food and Drug Administration (the “FDA”) regulates the manufacture and sale of medical devices. Under such regulation, medical devices are classified as Class I, Class II or Class III devices. The company’s principal products are designated as Class I or Class II devices. In general, Class I devices must comply with labeling and record keeping requirements and are subject to other general controls. In addition to general controls, certain Class II devices must comply with product design and manufacturing controls established by the FDA. Domestic and foreign manufacturers of medical devices distributed commercially in the U.S. are subject to periodic inspections by the FDA. Furthermore, state, local and foreign governments have adopted regulations relating to the design, manufacture and marketing of health care products.
As part of its regulatory function, the FDA routinely inspects the sites of medical device companies, and in 2010, the FDA inspected certain of the company’s facilities. In December 2010, the company received a warning letter from the FDA related to documentation and procedures at the company’s Sanford, Florida facility. The letter does not call into question the safety or efficacy of Invacare products, and production has not been impacted. The company is taking these issues very seriously and has added resources to ensure it is addressing all of the FDA’s concerns in a timely manner.
The quality management system of all locations required to meet ISO 13485 requirements for the US, Canada, Europe and other foreign markets were inspected by a third party quality system registrar during 2010. All facilities were found to be in compliance and were issued new quality system certificates.
From time to time, the company may undertake voluntary recalls or field corrective actions of the company’s products to maintain ongoing customer relationships and to enhance the company’s reputation for adhering to high standards of quality and safety. None of the company’s actions has been classified by the FDA as high risk. The company continues to strengthen its programs to better ensure compliance with applicable regulations and actively keeps abreast of proposed regulations, particularly those which could have a material adverse effect on the company.
The company occasionally sponsors clinical studies, usually involving its respiratory products. These studies have historically been non-significant risk studies with human subjects. Such studies, their protocols, participant criteria and all results are registered in the Clinical Registry managed by the National Institutes of Health and available to the public via the Internet.
I-14
Table of Contents
In regards to reimbursement, the company is mindful of three key issues. In the United States, the Centers for Medicaid and Medicare Services is moving forward with National Competitive Bidding in the first nine metropolitan areas. While the company expects this to be neutral to earnings in 2011, it will remain judicious in its extension of credit to customers and it will monitor whether other payors begin to model their payments on this system. The company also will closely watch State Medicaid budgets and how deficits may impact coverage and payments for home medical equipment and institutional care products. In the European segment, there is discussion by the French government of reduced wheelchair reimbursement in the second half of 2011. This issue was originally anticipated to occur in 2010, but it was delayed.
Last year’s health care reform law in the U.S., the Patient Protection and Accountable Care Act, included a number of provisions affecting the HME industry. First, the health care law expanded Round 2 of the Medicare competitive bid program from 70 to 91 bid areas. Second, the law eliminated the Medicare program’s first month purchase option for standard power wheelchairs effective January 1, 2011. Instead, Medicare now makes rental payments for 13 months before the beneficiary assumes ownership of the standard power wheelchair. Finally, the new health care law imposed a “productivity adjustment” to the annual fee schedules of all Medicare providers, including HME providers, that limits any annual cost of living increases applied to the fee schedules. The 2010 health care reform law also includes a new tax on U.S. sales of medical device manufacturers or importers, such as Invacare. The law will impose a yearly 2.3% sales-based excise tax on medical device manufacturers starting in 2013. The excise tax will be deductible by the manufacturer on its federal tax return. The excise tax will not apply to medical devices that the Secretary of Treasury determines are generally purchased by the general public at retail for individual use. At this point, it is unclear whether any of Invacare products will be determined to be exempt from the tax by the Department of Treasury.
Although these reductions in Medicare payments are not beneficial to the home care industry, the company believes that it can still grow and thrive in this environment. No significant cost-of-living adjustments have been made over the last few years to the reimbursement and payment amounts permitted under Medicare with respect to the company’s products, but the company will continue to try to respond with improved productivity to address the lack of support from Congress. In addition, the company’s respiratory products (for example, the low-cost HomeFill® oxygen delivery system) can help offset the Medicare reimbursement cuts to the home care provider. The company will continue to focus on developing products that help the provider improve profitability. Additionally, the company continues to focus on low-cost country sourcing and/or manufacturing to help ensure that the company is one of the lowest cost manufacturers and distributors to the home care provider.
BACKLOG
The company generally manufactures most of its products to meet near-term demands by shipping from stock or by building to order based on the specialty nature of certain products. Therefore, the company does not have substantial backlog of orders of any particular product nor does it believe that backlog is a significant factor for its business.
EMPLOYEES
As of December 31, 2010, the company had approximately 6,300 employees.
FOREIGN OPERATIONS AND EXPORT SALES
The company also markets its products for export to other foreign countries. In 2010, the company had product sales in over 80 countries worldwide. For information relating to net sales, operating income and identifiable assets of the company’s foreign operations, see Business Segments in the Notes to the Consolidated Financial Statements.
I-15
Table of Contents
AVAILABLE INFORMATION
The company files Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K, and any amendments thereto, as well as proxy statements and other documents with the Securities and Exchange Commission (SEC). The public may read and copy any material that the company files with the SEC at the SEC’s Public Reference Room located at 100 F Street, NE, Washington D.C. 20549. The public may obtain information on the operation of the Public Reference Room by calling the SEC at 1-800-SEC-0330. The SEC also maintains a website, www.sec.gov, which contains all reports, proxy statements and other information filed by the company with the SEC.
Additionally, Invacare’s filings with the SEC are available on or through the company’s website, www.invacare.com, as soon as reasonably practicable after they are filed electronically with, or furnished to, the SEC. Copies of the company’s filings also can be requested, free of charge, by writing to: Shareholder Relations Department, Invacare Corporation, One Invacare Way, P.O. Box 4028, Elyria, OH 44036-2125.
FORWARD-LOOKING INFORMATION
This Form 10-K contains forward-looking statements within the meaning of the “Safe Harbor” provisions of the Private Securities Litigation Reform Act of 1995. Terms such as “will,” “should,” “could”, “plan,” “intend,” “expect,” “continue,” “forecast,” “believe,” “anticipate” and “seek,” as well as similar comments, are forward-looking in nature. Because forward-looking statements relate to the future, they are subject to inherent uncertainties, risks and changes in circumstances that are difficult to predict. Actual results and events may differ significantly from those expressed or anticipated as a result of risks and uncertainties which include, but are not limited to, the following: adverse changes in government and other third-party payor reimbursement levels and practices (such as, for example, the Medicare bidding program covering nine metropolitan areas beginning in 2011 and an additional 91 metropolitan areas beginning in 2013), impacts of the U.S. health care reform legislation that was recently enacted (such as, for example, the excise tax beginning in 2013 on medical devices, together with further regulations to be promulgated by the U.S. Secretary of Treasury, if adopted); the uncertain impact on the Company’s providers, on the Company’s suppliers and on the demand for the Company’s products resulting from the current global economic conditions and general volatility in the credit and stock markets; loss of key health care providers; exchange rate and tax rate fluctuations; inability to design, manufacture, distribute and achieve market acceptance of new products with higher functionality and lower costs; consolidation of health care providers and the Company’s competitors; lower cost imports; uncollectible accounts receivable; difficulties in implementing/upgrading Enterprise Resource Planning systems; risks inherent in managing and operating businesses in many different foreign jurisdictions; ineffective cost reduction and restructuring efforts; potential product recalls; legal actions or regulatory proceedings and governmental investigations (including, for example, compliance costs or other adverse effects arising from FDA or other regulatory enforcement actions); product liability claims; possible adverse effects of being leveraged, which could impact the Company’s ability to raise capital, limit its ability to react to changes in the economy or the health care industry or expose the Company to interest rate or event of default risks; increased freight costs; inadequate patents or other intellectual property protection; extensive government regulation of the Company’s products; failure to comply with regulatory requirements or receive regulatory clearance or approval for the Company’s products or operations in the United States or abroad; incorrect assumptions concerning demographic trends that impact the market for the Company’s products; decreased availability or increased costs of materials which could increase the Company’s costs of producing or acquiring the Company’s products, including possible increases in commodity costs; the loss of the services of the Company’s key management and personnel; inability to acquire strategic acquisition candidates because of limited financing alternatives; increased security concerns and potential business interruption risks associated with political and/or social unrest in foreign countries where the Company’s facilities or assets are located; provisions of Ohio law or in the Company’s debt agreements, shareholder rights plan or charter documents that may prevent or delay a change in control, as well as the risks described from time to time in Invacare’s reports as filed with the Securities and
I-16
Table of Contents
Exchange Commission. Except to the extent required by law, we do not undertake and specifically decline any obligation to review or update any forward-looking statements or to publicly announce the results of any revisions to any of such statements to reflect future events or developments or otherwise.
| Item 1A. | Risk Factors. |
The company’s business, operations and financial condition are subject to various risks and uncertainties. One should carefully consider the risks and uncertainties described below, together with all of the other information in this annual report on Form 10-K and in the company’s other filings with the SEC, before making any investment decision with respect to the company’s securities. The risks and uncertainties described below may not be the only ones the company faces. Additional risks and uncertainties not presently known by the company or that the company currently deems immaterial may also affect the company’s business. If any of these known or unknown risks or uncertainties actually occur, develop or worsen, the company’s business, financial condition, results of operations and future growth prospects could change substantially.
The adoption of healthcare reform and other legislative developments in the United States may adversely affect the company’s business, results of operations and/or financial condition.
In March 2010, significant reforms to the healthcare system were adopted as law in the United States. The law includes provisions that, among other things, reduce and/or limit Medicare reimbursement, require all individuals to have health insurance (with limited exceptions) and impose new and/or increased taxes. Specifically, the law imposes a 2.3% excise tax on U.S. sales of most medical devices beginning in 2013. The company is still evaluating the impact of this tax on its overall business. Various healthcare reform proposals have also emerged at the state level. The new law and these proposals could impact the demand for the company’s products or the prices at which the company sells its products. In addition, the excise tax will increase the company’s cost of doing business. The impact of this law and these proposals could have a material adverse effect on the company’s business, results of operations and/or financial condition.
The recently enacted Dodd-Frank Wall Street Reform and Consumer Protection Act (the “Act”) institutes a wide range of reforms, some of which may impact the company. Among other things, the Act contains significant corporate governance and executive compensation-related provisions that authorize or require the SEC to adopt additional rules and regulations in these areas, such as shareholder “say on pay” voting and proxy access. The impact of these provisions on the company’s business is uncertain. The Act also provides for new statutory and regulatory requirements for derivative transactions, including foreign exchange and interest rate hedging transactions. Certain transactions will be required to be cleared on exchanges, and cash collateral will be required for those transactions. While the Act provides for a potential exception from these clearing and cash collateral requirements for commercial end-users such as the company, the exception is subject to future rule making and interpretation by regulatory authorities. The company enters into foreign exchange contracts, interest rate swaps and foreign currency forward contracts from time to time to manage its exposure to commodity price risk, foreign currency exchange risk and interest rate risk. If, in the future, the company is required to provide cash collateral for its hedging transactions, it could reduce the company’s ability to execute strategic hedges. In addition, the contractual counterparties in hedging arrangements will be required to comply with the Act’s new requirements, which could ultimately result in increased costs of these arrangements to customers such as the company.
Changes in government and other third-party payor reimbursement levels and practices have negatively impacted and could continue to negatively impact the company’s revenues and profitability.
The company’s products are sold primarily through a network of medical equipment and home health care providers, extended care facilities, hospital and HMO-based stores and other providers. In addition, the company sells directly to various government providers throughout the world. Many of these providers (the company’s customers) are reimbursed for the products and services provided to their customers and patients by third-party
I-17
Table of Contents
payors, such as government programs, including Medicare and Medicaid, private insurance plans and managed care programs. Most of these programs set maximum reimbursement levels for some of the products sold by the company in the United States and abroad. If third-party payors deny coverage, make the reimbursement process or documentation requirements more uncertain or further reduce their current levels of reimbursement (i.e., beyond the reductions described below), or if the company’s costs of production increase faster than increases in reimbursement levels, the company may be unable to sell the affected product(s) through its distribution channels on a profitable basis.
Reduced government reimbursement levels and changes in reimbursement policies have in the past added, and could continue to add, significant pressure to the company’s revenues and profitability. For example, CMS also introduced competitive bidding for nine metropolitan areas in the U.S., which went into effect in January 2011. The reimbursement rates for nine product categories were reduced by an average of 32 percent in these nine metropolitan areas. CMS is currently scheduled to expand the NCB program to an additional 91 metropolitan areas in January 2013.
Similar trends and concerns are occurring in state Medicaid programs. These recent changes to reimbursement policies, and any additional unfavorable reimbursement policies or budgetary cuts that may be adopted in the future, could adversely affect the demand for the company’s products by customers who depend on reimbursement from the government-funded programs. The percentage of the company’s overall sales that are dependent on Medicare or other insurance programs may increase as the portion of the U.S. population over age 65 continues to grow, making the company more vulnerable to reimbursement level reductions by these organizations. Reduced government reimbursement levels also could result in reduced private payor reimbursement levels because some third-party payors index their reimbursement schedules to Medicare fee schedules. Reductions in reimbursement levels also may affect the profitability of the company’s customers and ultimately force some customers without strong financial resources to go out of business. The reductions that went into effect recently may prove to be so dramatic that some of the company’s customers may not be able to adapt quickly enough to survive. The company is the industry’s largest creditor and an increase in bankruptcies in the company’s customer base could have an adverse effect on the company’s financial results.
Outside the United States, reimbursement systems vary significantly by country. Many foreign markets have government-managed health care systems that govern reimbursement for new home health care products. The ability of hospitals and other providers supported by such systems to purchase the company’s products is dependent, in part, upon public budgetary constraints. Various countries have tightened reimbursement rates and other countries may follow. If adequate levels of reimbursement from third-party payors outside of the United States are not obtained, international sales of the company’s products may decline, which could adversely affect the company’s net sales and would have a material adverse effect on the company’s business, financial condition and results of operations.
The impact of all the changes discussed above is uncertain and could have a material adverse effect on the company’s business, financial condition and results of operations.
The company is subject to risks arising out of the continuing global economic uncertainty.
As is the case for many companies operating in the current economic environment, the company is exposed to a number of risks. These risks include the possibility that: one or more of the lenders participating in the company’s revolving credit facility may be unable or unwilling to extend credit to the company; the third party company that provides lease financing to the company’s customers may refuse or be unable to fulfill its financing obligations or extend credit to the company’s customers; one or more customers of the company may be unable to pay for purchases of the company’s products on a timely basis; one or more key suppliers may be unable or unwilling to provide critical goods or services to the company; and one or more of the counterparties to the company’s hedging arrangements may be unable to fulfill its obligations to the company. Although the company
I-18
Table of Contents
has taken actions in an effort to mitigate these risks, during periods of economic downturn, the company’s exposure to these risks increases. Events of this nature may adversely affect the company’s liquidity or sales and revenues, and therefore have an adverse effect on the company’s business and results of operations.
If the company’s cost reduction efforts are ineffective, the company’s revenues and profitability could be negatively impacted.
In response to reimbursement reductions, including competitive pricing pressures, the company continues to initiate numerous cost reduction and organizational efficiency efforts, including globalization of its product lines. The company may not be successful in achieving the operating efficiencies and operating cost reductions expected from these efforts and the company may experience business disruptions associated with the restructuring and cost reduction activities. These efforts may not produce the full efficiency and cost reduction benefits that the company expects. Further, these benefits may be realized later than expected, and the costs of implementing these measures may be greater than anticipated. If these measures are not successful, the company may undertake additional cost reduction efforts, which could result in future charges. Moreover, the company’s ability to achieve other strategic goals and business plans and the company’s financial performance may be adversely affected and the company could experience business disruptions with customers and elsewhere if the company’s cost reduction and restructuring efforts prove ineffective.
The company’s revenues and profits are subject to exchange rate and interest rate fluctuations that could adversely affect its results of operations or financial position.
Currency exchange rates are subject to fluctuation due to, among other things, changes in local, regional or global economic conditions, the imposition of currency exchange restrictions and unexpected changes in regulatory or taxation environments. The functional currency of the company’s subsidiaries outside the United States is the predominant currency used by the subsidiaries to transact business. Through the company’s international operations, the company is exposed to foreign currency fluctuations, and changes in exchange rates can have a significant impact on net sales and elements of cost. The company conducts a significant number of transactions in currencies other than the U.S. dollar. In addition, because certain of the company’s costs and revenues are denominated in other currencies, the company’s results of operations are exposed to foreign exchange rate fluctuations as the financial results of those operations are translated from local currency into U.S. dollars upon consolidation.
The company uses forward contracts primarily to help reduce its exposure to transactional exchange rate risk. Despite the company’s efforts to mitigate these risks, however, the company’s revenues and profitability may be materially adversely affected by exchange rate fluctuations. The company does not have a meaningful way to hedge translation.
The company also is exposed to market risk through various financial instruments, including fixed rate and floating rate debt instruments. The company does at times use interest swap agreements to mitigate its exposure to interest rate fluctuations, but those efforts may not adequately protect the company from significant interest rate risks. Interest on much of the company’s debt is based on LIBOR, which is currently historically low. Increases in LIBOR could have a significant impact on the company’s reported interest expense.
The industry in which the company operates is highly competitive and some of the company’s competitors may be larger and may have greater financial resources than the company does.
The home medical equipment market is highly competitive and the company’s products face significant competition from other well-established manufacturers. Reduced government reimbursement levels and changes in reimbursement policies, such as the competitive bidding program implemented by CMS, may drive competitors that have greater financial resources than the company to offer drastically reduced pricing terms in an effort to secure government acceptance of their products and pricing. Any increase in competition may cause the company to lose market share or compel the company to reduce prices to remain competitive, which could have a material adverse affect on the company’s results of operations.
I-19
Table of Contents
The company’s success depends on the company’s ability to design, manufacture, distribute and achieve market acceptance of new products with higher functionality and lower costs.
The company sells products to customers primarily in markets that are characterized by technological change, product innovation and evolving industry standards, yet in which product price is increasingly a primary consideration in customers’ purchasing decisions. The company is continually engaged in product development and improvement programs. The company must continue to design and improve innovative products, effectively distribute and achieve market acceptance of those products, and reduce the costs of producing the company’s products, in order to compete successfully with the company’s competitors. If competitors’ product development capabilities become more effective than the company’s product development capabilities, if competitors’ new or improved products are accepted by the market before the company’s products or if competitors are able to produce products at a lower cost and thus offer products for sale at a lower price, the company’s business, financial condition and results of operation could be adversely affected.
The consolidation of health care customers and the company’s competitors could result in a loss of customers or in additional competitive pricing pressures.
Numerous initiatives and reforms instituted by legislators, regulators and third-party payors to reduce home medical equipment costs have resulted in a consolidation trend in the home medical equipment industry as well as among the company’s customers, including home health care providers. In the past, some of the company’s competitors have been lowering the purchase prices of their products in an effort to attract customers. This in turn has resulted in greater pricing pressures, including pressure to offer customers more competitive pricing terms, and the exclusion of certain suppliers from important market segments as group purchasing organizations, independent delivery networks and large single accounts continue to consolidate purchasing decisions for some of the company’s customers. Further consolidation could result in a loss of customers, in increased collectability risks, or in increased competitive pricing pressures.
The company may be adversely affected by legal actions or regulatory proceedings.
The company may be subject to claims, litigation or other liabilities as a result of injuries caused by allegedly defective products, acquisitions the company has completed or in the intellectual property area. Any such claims or litigation against the company, regardless of the merits, could result in substantial costs and could harm the company’s business or its reputation. Intellectual property litigation or claims also could require the company to:
| • | cease manufacturing and selling any of the company’s products that incorporate the challenged intellectual property; |
| • | obtain a license from the holder of the infringed intellectual property right alleged to have been infringed, which license may not be available on commercially reasonable terms, if at all; or |
| • | redesign or rename the company’s products, which may not be possible, and could be costly and time consuming and could result in lost revenues and market share. |
The results of legal proceedings are difficult to predict and the company cannot provide any assurance that an action or proceeding will not be commenced against the company, or that the company will prevail in any such action or proceeding. An unfavorable resolution of any legal action or proceeding could materially and adversely affect the company’s business, results of operations, liquidity or financial condition or its reputation.
Product liability claims may harm the company’s business, particularly if the number of claims increases significantly or the company’s product liability insurance proves inadequate.
The manufacture and sale of home health care devices and related products exposes the company to a significant risk of product liability claims. From time to time, the company has been, and is currently, subject to a number of product liability claims alleging that the use of the company’s products has resulted in serious injury or even death.
I-20
Table of Contents
Even if the company is successful in defending against any liability claims, these claims could nevertheless distract the company’s management, result in substantial costs, harm the company’s reputation, adversely affect the sales of all the company’s products and otherwise harm the company’s business. If there is a significant increase in the number of product liability claims, the company’s business could be adversely affected.
The company’s captive insurance company, Invatection Insurance Company, currently has a policy year that runs from September 1 to August 31 and insures annual policy losses of $10,000,000 per occurrence and $13,000,000 in the aggregate of the company’s North American product liability exposure. The company also has additional layers of external insurance coverage insuring up to $75,000,000 in annual aggregate losses arising from individual claims anywhere in the world that exceed the captive insurance company policy limits or the limits of the company’s per country foreign liability limits as applicable. There can be no assurance that the company’s current insurance levels will continue to be adequate or available at affordable rates.
Product liability reserves are recorded for individual claims based upon historical experience, industry expertise and indications from the third-party actuary. Additional reserves, in excess of the specific individual case reserves, are provided for incurred but not reported claims based upon actuarial valuations at the time such valuations are conducted. Historical claims experience and other assumptions are taken into consideration to estimate the ultimate reserves. For example, the actuarial analysis assumes that historical loss experience is an indicator of future experience, that the distribution of exposures by geographic area and nature of operations for ongoing operations is expected to be very similar to historical operations with no dramatic changes and that the government indices used to trend losses and exposures are appropriate. Estimates made are adjusted on a regular basis and can be impacted by actual loss awards and settlements on claims. While actuarial analysis is used to help determine adequate reserves, the company is responsible for the determination and recording of adequate reserves in accordance with accepted loss reserving standards and practices.
In addition, as a result of a product liability claim or if the company’s products are alleged to be defective, the company may have to recall some of its products, may have to incur significant costs or may suffer harm to its business reputation.
The company’s products are subject to recalls, which could harm the company’s reputation and business.
The company is subject to ongoing medical device reporting regulations that require the company to report to the FDA or similar governmental authorities in other countries if the company’s products cause, or contribute to, death or serious injury, or if they malfunction and would be likely to cause, or contribute to, death or serious injury if the malfunction were to recur. The FDA and similar governmental authorities in other countries have the authority to require the company to do a field correction or recall the company’s products in the event of material deficiencies or defects in design or manufacturing. In addition, in light of a deficiency, defect in design or manufacturing or defect in labeling, the company may voluntarily elect to recall or correct the company’s products. A government mandated or voluntary recall/field correction by the company could occur as a result of component failures, manufacturing errors or design defects, including defects in labeling. Any recall/field correction would divert managerial and financial resources and could harm the company’s reputation with its customers, product users and the health care professionals that use, prescribe and recommend the company’s products. The company could have product recalls or field actions that result in significant costs to the company in the future, and these actions could have a material adverse effect on the company’s business.
The company is subject to extensive government regulation, and if the company fails to comply with applicable laws or regulations, the company could suffer severe civil or criminal sanctions or be required to make significant changes to the company’s operations that could have a material adverse effect on the company’s results of operations.
The company sells its products principally to medical equipment and home health care providers who resell or rent those products to consumers. Many of those providers (the company’s customers) are reimbursed for the
I-21
Table of Contents
Invacare products sold to their customers and patients by third-party payors, including Medicare and Medicaid. The U.S. federal government and the governments in the states and other countries in which the company operates regulate many aspects of the company’s business. As a medical device manufacturer, the company is subject to extensive government regulation, including numerous laws directed at preventing fraud and abuse and laws regulating reimbursement under various government programs. The marketing, invoicing, documenting and other practices of health care suppliers and manufacturers are all subject to government scrutiny. Government agencies periodically open investigations and obtain information from health care suppliers and manufacturers pursuant to the legal process. Violations of law or regulations can result in severe administrative, civil and criminal penalties and sanctions, including disqualification from Medicare and other reimbursement programs, which could have a material adverse effect on the company’s business. The company has established numerous policies and procedures that the company believes are sufficient to ensure that the company will operate in substantial compliance with these laws and regulations. In addition, the company employs a Director of Compliance and Internal Audit to continue to develop, implement, monitor and manage these policies and procedures, including internal controls, to comply with applicable legal, regulatory and company standards. The company cannot guarantee that its efforts will be effective to prevent a material adverse effect on the company’s business from noncompliance issues.
The company received a subpoena in 2006 from the U.S. Department of Justice seeking documents relating to three long-standing and well-known promotional and rebate programs maintained by the company. The company believes that the programs described in the subpoena are in compliance with all applicable laws and the company has cooperated fully with the government investigation. As of February 2011, the subpoena remains pending.
Health care is an area of rapid regulatory change. Changes in the law and new interpretations of existing laws may affect permissible activities, the costs associated with doing business, and reimbursement amounts paid by federal, state and other third-party payors. The company cannot predict the future of federal, state and local regulation or legislation, including Medicare and Medicaid statutes and regulations, or possible changes in health care policies in any country in which the company conducts business. Future legislation and regulatory changes could have a material adverse effect on the company’s business.
The company’s research and development and manufacturing processes are subject to federal, state, local and foreign environmental requirements.
The company’s research and development and manufacturing processes are subject to federal, state, local and foreign environmental requirements, including requirements governing the discharge of pollutants into the air or water, the use, handling, storage and disposal of hazardous substances and the responsibility to investigate and clean up contaminated sites. Under some of these laws, the company also could be held responsible for costs relating to any contamination at the company’s past or present facilities and at third-party waste disposal sites. These could include costs relating to contamination that did not result from any violation of law and, in some circumstances, contamination that the company did not cause. The company may incur significant expenses relating to the failure to comply with environmental laws. The enactment of stricter laws or regulations, the stricter interpretation of existing laws and regulations or the requirement to undertake the investigation or remediation of currently unknown environmental contamination at the company’s own or third party sites may require the company to make additional expenditures, which could be material.
The company’s failure to comply with regulatory requirements or receive regulatory clearance or approval for the company’s products or operations in the United States or abroad could adversely affect the company’s business.
The company’s medical devices are subject to extensive regulation in the United States by the Food and Drug Administration, or the “FDA,” and by similar governmental authorities in the foreign countries where the company does business. The FDA regulates virtually all aspects of a medical device’s development, testing,
I-22
Table of Contents
manufacturing, labeling, promotion, distribution and marketing. In addition, the company is required to file reports with the FDA if the company’s products cause, or contribute to, death or serious injury, or if they malfunction and would be likely to cause, or contribute to, death or serious injury if the malfunction were to recur. In general, unless an exemption applies, the company’s wheelchair and respiratory medical devices must receive a pre-marketing clearance from the FDA before they can be marketed in the United States. The FDA also regulates the export of medical devices to foreign countries. The company cannot be assured that any of the company’s devices, to the extent required, will be cleared by the FDA through the pre-market clearance process or that the FDA will provide export certificates that are necessary to export certain of the company’s products. If FDA issues a warning letter as a result of its findings from their inspections, the FDA could refuse to provide export certificates until the matters covered in the warning letter are resolved.
Additionally, the company may be required to obtain pre-marketing clearances to market modifications to the company’s existing products or market its existing products for new indications. The FDA requires device manufacturers themselves to make and document a determination as to whether or not a modification requires a new clearance; however, the FDA can review and disagree with a manufacturer’s decision. The company has applied for, and received, a number of such clearances in the past. The company may not be successful in receiving clearances in the future or the FDA may not agree with the company’s decisions not to seek clearances for any particular device modification. The FDA may require a clearance for any past or future modification or a new indication for the company’s existing products. Such submissions may require the submission of additional data and may be time consuming and costly, and ultimately may not be cleared by the FDA.
If the FDA requires the company to obtain pre-marketing clearances for any modification to a previously cleared device, the company may be required to cease manufacturing and marketing the modified device or to recall the modified device until the company obtains FDA clearance and the company may be subject to significant regulatory fines or penalties. In addition, the FDA may not clear these submissions in a timely manner, if at all. The FDA also may change its policies, adopt additional regulations or revise existing regulations, each of which could prevent or delay pre-market clearance of the company’s devices, or could impact the company’s ability to market a device that was previously cleared. Any of the foregoing could adversely affect the company’s business.
As part of its regulatory function, the FDA routinely inspects the sites of medical device companies, and in 2010, the FDA inspected certain of the company’s facilities. In December 2010, the company received a warning letter from the FDA related to documentation and procedures at the company’s Sanford, Florida facility. The letter does not call into question the safety or efficacy of Invacare products, and production has not been impacted. The company is taking these issues very seriously and has added resources to ensure it is addressing all of the FDA’s concerns in a timely manner. However, the results of regulatory claims, proceedings, investigations, or litigation are difficult to predict. An unfavorable resolution or outcome of an FDA inspection or investigation could materially and adversely affect the company’s business, financial condition, and results of operations.
The company’s failure to comply with the regulatory requirements of the FDA and other applicable U.S. regulatory requirements may subject the company to administrative or judicially imposed sanctions. These sanctions include warning letters, civil penalties, criminal penalties, injunctions, consent decrees, product seizure or detention, product recalls and total or partial suspension of production.
In many of the foreign countries in which the company markets its products, the company is subject to extensive regulations that are similar to those of the FDA, including those in Europe. The regulation of the company’s products in Europe falls primarily within the European Economic Area, which consists of the 27 member states of the European Union, as well as Iceland, Liechtenstein and Norway. Only medical devices that comply with certain conformity requirements of the Medical Device Directive are allowed to be marketed within the European Economic Area. In addition, the national health or social security organizations of certain foreign countries, including those outside Europe, require the company’s products to be qualified before they can be marketed in those countries. Failure to receive or delays in the receipt of, relevant foreign qualifications in the European Economic Area or other foreign countries could have a material adverse effect on the company’s business.
I-23
Table of Contents
Decreased availability or increased costs of raw materials could increase the company’s costs of producing its products.
The company purchases raw materials, fabricated components, some finished goods and services from a variety of suppliers. Raw materials such as plastics, steel, and aluminum are considered key raw materials. Where appropriate, the company employs contracts with its suppliers, both domestic and international. In those situations in which contracts are not advantageous, the company believes that its relationships with its suppliers are satisfactory and that alternative sources of supply are readily available. From time to time, however, the prices and availability of these raw materials fluctuate due to global market demands, which could impair the company’s ability to procure necessary materials, or increase the cost of these materials. Inflationary and other increases in costs of these raw materials have occurred in the past and may recur from time to time. In addition, freight costs associated with shipping and receiving product and sales are impacted by fluctuations in the cost of oil and gas. A reduction in the supply or increase in the cost of those raw materials could impact the company’s ability to manufacture its products and could increase the cost of production. As an example, inflation in China has in the past and will probably in the future increase costs and an appreciation of the Yuan or an increase in labor rates could have an unfavorable impact on the cost of key components and some finished goods. Demand in China and other developing countries for raw materials may result in increases in the cost of key commodities and could have a negative impact on the profits of the company if these increases cannot be passed onto the company’s customers.
Lower cost imports could negatively impact the company’s profitability.
Lower cost imports sourced from Asia may negatively impact the company’s sales volumes. In the past, competition from certain of these products has caused the company to lower its prices, cutting into the company’s profit margins and reducing the company’s overall profitability.
Difficulties in implementing or upgrading the company’s Enterprise Resource Planning systems may disrupt the company’s business.
The company is continuously upgrading its Enterprise Resource Planning (ERP) systems which results in various complexities and business process changes that can negatively affect the company’s ability to handle transactions, such as the processing of orders, and can create customer disruptions and or loss of some business. While the company believes that the difficulties associated with implementing and upgrading the company’s ERP systems are manageable, there can be no assurance that the company will not experience disruptions or inefficiencies in the company’s business operations as a result of new system implementations or upgrades.
The company’s reported results may be adversely affected by increases in reserves for uncollectible accounts receivable.
The company has a large balance of accounts receivable and has established a reserve for the portion of such accounts receivable that the company estimates will not be collected because of the company’s customers’ non-payment. The specific reserve is based on historical trends and current relationships with the company’s customers and providers. Changes in the company’s collection rates can result from a number of factors, including turnover in personnel, changes in the payment policies or practices of payors, changes in industry rates or pace of reimbursement or changes in the financial health of the company’s customers. As a result of past changes in Medicare reimbursement regulations, specifically changes to the qualification processes and reimbursement levels of consumer power wheelchairs and custom power wheelchairs, the business viability of several of the company’s customers had become questionable and several have failed. Further, as national competitive bidding is implemented in additional areas, the number of start-up or new providers who have three-year contracted pricing will increase. The company’s reserve for uncollectible receivables has fluctuated in the past and will continue to fluctuate in the future. Changes in rates of collection, even if they are small in absolute terms, could require the company to increase its reserve for uncollectible receivables beyond its current level. The company has reviewed the accounts receivables, including those receivables financed through DLL,
I-24
Table of Contents
associated with many of its customers that are most exposed to these issues. If the business viability of certain of the company’s customers deteriorates or if the company’s credit policies are ineffective in reducing the company’s exposures to credit risk, additional increases in reserves for uncollectible accounts may be necessary, which could adversely affect the company’s financial results.
Failure to properly manage the distribution of the company’s products may result in reduced revenue and profitability.
The company uses a variety of distribution methods to sell its products and services. The company’s distribution network includes various customers such as specialized home health care providers and extended care facilities, hospital and HMO-based stores, home health agencies, mass merchandisers and the Internet. As the company reaches more customers worldwide through an increasing number of new distribution channels, inventory management becomes more challenging. If the company is unable to properly manage and balance inventory levels and potential conflicts among these various distribution methods, its operating results could be harmed.
The company is subject to certain risks inherent in managing and operating businesses in many different foreign jurisdictions.
The company has significant international operations, including operations in Australia, Canada, New Zealand, Mexico, Asia (primarily China) and Europe. There are risks inherent in operating and selling products internationally, including:
| • | different regulatory environments and reimbursement systems; |
| • | difficulties in enforcing agreements and collecting receivables through certain foreign legal systems; |
| • | foreign customers who may have longer payment cycles than customers in the United States; |
| • | tax rates in certain foreign countries that may exceed those in the United States and foreign earnings that may be subject to withholding requirements; |
| • | the imposition of tariffs, exchange controls or other trade restrictions including transfer pricing restrictions when products produced in one country are sold to an affiliated entity in another country; |
| • | general economic and political conditions in countries where the company operates or where end users of the company’s products reside; |
| • | security concerns and potential business interruption risks associated with political and/or social unrest in foreign countries where the company’s facilities or assets are located; |
| • | difficulties associated with managing a large organization spread throughout various countries; |
| • | difficulties in enforcing intellectual property rights and weaker intellectual property rights protection in some countries; |
| • | required compliance with a variety of foreign laws and regulations; and |
| • | differing consumer product preferences. |
The factors described above also could disrupt the company’s product manufacturing/assembling and key suppliers located outside of the United States. For example, the company increasingly relies on its manufacturing and sourcing operations in China for the production of its products. Disruptions in the company’s foreign operations, particularly those in China or Mexico, may impact the company’s revenues and profitability.
The company’s debt may limit the company’s flexibility in operating its business.
The company has substantial outstanding indebtedness. This indebtedness requires a significant portion of cash flow from operations to be dedicated to the payment of principal and or interest, thus reducing the
I-25
Table of Contents
company’s ability to use its cash flow to fund its operations, capital expenditures and future business opportunities. The company’s indebtedness also may limit the company’s ability to react to changes in the economy or its industry.
The company’s revolving credit facility contains various covenants that limit the company’s ability to engage in specified types of transactions. In addition, under the company’s revolving credit facility, it is required to satisfy and maintain specified financial ratios and other financial condition tests. These covenants could materially and adversely affect the company’s ability to finance its future operations or capital needs. Furthermore, they may restrict the company’s ability to conduct and expand its business and pursue its business strategies. The company’s ability to meet these financial ratios and financial condition tests can be affected by events beyond its control, including changes in general economic and business conditions.
If the company’s patents and other intellectual property rights do not adequately protect the company’s products, the company may lose market share to its competitors and may not be able to operate profitably.
The company relies on a combination of patents, trade secrets and trademarks to establish and protect the company’s intellectual property rights in its products and the processes for the development, manufacture and marketing of the company’s products.
The company uses non-patented proprietary know-how, trade secrets, undisclosed internal processes and other proprietary information and currently employs various methods to protect this proprietary information, including confidentiality agreements, invention assignment agreements and proprietary information agreements with various vendors, employees, independent sales agents, distributors, consultants and others. However, these agreements may be breached. The FDA or another governmental agency may require the disclosure of this information in order for the company to have the right to market a product. Trade secrets, know-how and other unpatented proprietary technology also may otherwise become known to, or independently developed by, the company’s competitors.
In addition, the company holds U.S. and foreign patents relating to a number of its components and products and has patent applications pending with respect to other components and products. The company also applies for additional patents in the ordinary course of its business, as the company deems appropriate. However, these precautions offer only limited protection, and the company’s proprietary information may become known to, or be independently developed by, competitors, or the company’s proprietary rights in intellectual property may be challenged, any of which could have a material adverse effect on the company’s business, financial condition and results of operations. Additionally, the company cannot assure that its existing or future patents, if any, will afford the company adequate protection or any competitive advantage, that any future patent applications will result in issued patents or that the company’s patents will not be circumvented, invalidated or declared unenforceable.
Any proceedings before the U.S. Patent and Trademark Office could result in adverse decisions as to the priority of the company’s inventions and the narrowing or invalidation of claims in issued patents. The company also could incur substantial costs in any proceeding. In addition, the laws of some of the countries in which the company’s products are or may be sold may not protect the company’s products and intellectual property to the same extent as U.S. laws, if at all. The company also may be unable to protect the company’s rights in trade secrets and unpatented proprietary technology in these countries.
In addition, the company holds patent and other intellectual property licenses from third parties for some of its products and on technologies that are necessary in the design and manufacture of some of the company’s products. The loss of these licenses could prevent the company from, or could cause additional disruption or expense in, manufacturing, marketing and selling these products, which could harm the company’s business.
I-26
Table of Contents
The company’s operating results and financial condition could be adversely affected if the company becomes involved in litigation regarding its patents or other intellectual property rights.
Litigation involving patents and other intellectual property rights is common in the company’s industry, and other companies within the company’s industry have used intellectual property litigation in an attempt to gain a competitive advantage. The company currently is, and in the future may become, a party to lawsuits involving patents or other intellectual property. If the company loses any of these proceedings, a court or a similar foreign governing body could invalidate or render unenforceable the company’s owned or licensed patents, require the company to pay significant damages, seek licenses and/or pay ongoing royalties to third parties, require the company to redesign its products, or prevent the company from manufacturing, using or selling its products, any of which would have an adverse effect on the company’s results of operations and financial condition. The company has brought, and may in the future also bring, actions against third parties for infringement of the company’s intellectual property rights. The company may not succeed in these actions. The defense and prosecution of intellectual property suits, proceedings before the U.S. Patent and Trademark Office or its foreign equivalents and related legal and administrative proceedings are both costly and time consuming. Protracted litigation to defend or prosecute the company’s intellectual property rights could seriously detract from the time the company’s management would otherwise devote to running its business. Intellectual property litigation relating to the company’s products could cause its customers or potential customers to defer or limit their purchase or use of the affected products until resolution of the litigation.
The company’s business strategy relies on certain assumptions concerning demographic trends that impact the market for its products. If these assumptions prove to be incorrect, demand for the company’s products may be lower than expected.
The company’s ability to achieve its business objectives is subject to a variety of factors, including the relative increase in the aging of the general population. The company believes that these trends will increase the need for its products. The projected demand for the company’s products could materially differ from actual demand if the company’s assumptions regarding these trends and acceptance of its products by health care professionals and patients prove to be incorrect or do not materialize. If the company’s assumptions regarding these factors prove to be incorrect, the company may not be able to successfully implement the company’s business strategy, which could adversely affect the company’s results of operations. In addition, the perceived benefits of these trends may be offset by competitive or business factors, such as the introduction of new products by the company’s competitors or the emergence of other countervailing trends, including lower reimbursement and pricing.
The loss of the services of the company’s key management and personnel could adversely affect its ability to operate the company’s business.
The company’s future success will depend, in part, upon the continued service of key managerial, research and development staff and sales and technical personnel. In addition, the company’s future success will depend on its ability to continue to attract and retain other highly qualified personnel. The company may not be successful in retaining its current personnel or in hiring or retaining qualified personnel in the future. The company’s failure to do so could have a material adverse effect on the company’s business. The company’s executive officers have substantial experience and expertise in the company’s industry. The company’s future success depends, to a significant extent, on the abilities and efforts of its executive officers and other members of its management team. If the company loses the services of any of its management team, the company’s business may be adversely affected.
I-27
Table of Contents
The company’s Chairman of the Board of Directors and certain members of management own shares representing a substantial percentage of the company’s voting power and their interests may differ from other shareholders.
The company has two classes of common stock. The Common Shares have one vote per share and the Class B Common Shares have 10 votes per share. As of January 1, 2011, the company’s chairman, Mr. A. Malachi Mixon, III, and certain members of management beneficially own approximately 33% of the combined voting power of the company’s Common Shares and Class B Common Shares and could influence the outcome of any corporate transaction or other matter submitted to the shareholders for approval, including mergers, consolidations and the sale of all or substantially all of the company’s assets. They also will have the power to influence or make more difficult a change in control. The interests of Mr. Mixon and his relatives may differ from the interests of the other shareholders and they may take actions with which some shareholders may disagree.
Since the company’s ability to obtain further financing may be limited, the company may be unable to acquire strategic acquisition candidates.
The company’s plans typically include identifying, acquiring, and integrating other strategic businesses. There are various reasons for the company to acquire businesses or product lines, including providing new products or new manufacturing and service capabilities, to add new customers, to increase penetration with existing customers, and to expand into new geographic markets. The company’s ability to successfully grow through acquisitions depends upon its ability to identify, negotiate, complete and integrate suitable acquisitions and to obtain any necessary financing. The costs of acquiring other businesses could increase if competition for acquisition candidates increases. Further, the provisions of the company’s existing credit facility impose limitations regarding acquisitions, which could prevent significant acquisitions, without entering into amendments with regard to those provisions. If the company is unable to obtain the necessary financing, it may miss opportunities to grow its business through strategic acquisitions.
Additionally, the success of the company’s acquisition strategy is subject to other risks and costs, including the following:
| • | the company’s ability to realize operating efficiencies, synergies, or other benefits expected from an acquisition, and possible delays in realizing the benefits of the acquired company or products; |
| • | diversion of management’s time and attention from other business concerns; |
| • | difficulties in retaining key employees of the acquired businesses who are necessary to manage these businesses; |
| • | difficulties in maintaining uniform standards, controls, procedures and policies throughout acquired companies; |
| • | adverse effects on existing business relationships with suppliers or customers; |
| • | the risks associated with the assumption of contingent or undisclosed liabilities of acquisition targets; and |
| • | ability to generate future cash flows or the availability of financing. |
In addition, an acquisition could materially impair the company’s operating results by causing the company to incur debt or requiring the amortization of acquisition expenses and acquired assets.
Armed hostilities, terrorism, natural disasters, political unrest or public health issues could harm the company’s business.
Armed hostilities, terrorism, natural disasters, political unrest or public health issues, whether in the U.S. or abroad, could cause damage or disruption to the company, its suppliers or customers, or could create political or
I-28
Table of Contents
economic instability, any of which could harm the company’s business. These events could cause a decrease in demand for the company’s products, could make it difficult or impossible for the company to deliver products or for the company’s suppliers to deliver materials, and could create delays and inefficiencies in the company’s manufacturing operations.
Certain provisions of the company’s debt agreements, its charter documents, its shareholder rights plan and Ohio law could delay or prevent the sale of the company.
Provisions of the company’s debt agreements, its charter documents, its shareholder rights plan and Ohio law may make it more difficult for a third party to acquire, or attempt to acquire, control of the company even if a change in control would result in the purchase of shares of the company at a premium to market price. In addition, these provisions may limit the ability of shareholders of the company to approve transactions that they may deem to be in their best interest.
| Item 1B. | Unresolved Staff Comments. |
None.
| Item 2. | Properties. |
The company owns or leases its warehouses, offices and manufacturing facilities and believes that these facilities are well maintained, adequately insured and suitable for their present and intended uses. Information concerning certain leased facilities of the company as of December 31, 2010 is set forth in Leases and Commitments in the Notes to the Consolidated Financial Statements of the company included in this report and in the table below:
| North American/HME Operations |
Square Feet |
Ownership Or Expiration Date of Lease |
Renewal Options |
Use | ||||||
| Akron, Ohio |
17,477 | April 2012 | One (5 yr.) | Offices | ||||||
| Alexandria, Virginia |
230 | September 2011 | None | Offices | ||||||
| Alpharetta, Georgia |
11,665 | March 2014 | None | Warehouse and Offices | ||||||
| Arlington, Texas |
63,626 | June 2011 | One (3 yr.) | Warehouse | ||||||
| Atlanta, Georgia |
91,418 | April 2011 | None | Warehouse and Offices | ||||||
| Beijing, China |
1,399 | January 2013 | None | Offices | ||||||
| Brookfield, Wisconsin |
3,200 | January 2013 | Two (3 yr.) | Warehouse and Offices | ||||||
| Chicopee, Massachusetts |
4,800 | November 2015 | Two (3 yr.) | Warehouse and Offices | ||||||
| Cranbury, New Jersey |
111,987 | April 2018 | Two (3 yr.) | Warehouse and Offices | ||||||
| Eden Prairie, Minnesota |
3,764 | September 2013 | Two (3 yr.) | Warehouse and Offices | ||||||
| Elyria, Ohio |
||||||||||
| —1200 Taylor Street |
251,656 | Own | — | Manufacturing and Offices | ||||||
| —899 Cleveland Street |
126,657 | November 2013 | None | Warehouse | ||||||
| —One Invacare Way |
50,000 | Own | — | Headquarters | ||||||
| —1320 Taylor Street |
30,000 | January 2015 | One (5 yr.) | Offices | ||||||
| —1160 Taylor Street |
4,800 | Own | — | Warehouse and Offices | ||||||
| —56 Ternes Avenue |
12,001 | December 2011 | Two (1 yr.) | Warehouse | ||||||
| Hampden, Maine |
4,800 | September 2011 | Three (1 yr.) | Warehouse and Offices | ||||||
| Hong Kong, China |
2,236 | November 2012 | None | Offices | ||||||
| Kansas City, Missouri |
2,822 | February 2013 | One (3 yr.) | Warehouse and Offices | ||||||
| Kirkland, Quebec |
26,196 | November 2015 | None | Manufacturing, Warehouse and Offices | ||||||
| Knoxville, Tennessee |
2,400 | May 2012 | One (1 yr.) | Warehouse and Offices | ||||||
I-29
Table of Contents
| North American/HME Operations |
Square Feet |
Ownership Or Expiration Date of Lease |
Renewal Options |
Use | ||||||
| Lithia Springs, Georgia |
4,000 | December 2011 | None | Warehouse and Offices | ||||||
| Marlboro, New Jersey |
2,800 | June 2012 | None | Offices | ||||||
| Mississauga, Ontario |
61,375 | February 2016 | None | Warehouse and Offices | ||||||
| Modesto, California |
3,675 | January 2013 | Two (3 yr.) | Warehouse and Offices | ||||||
| Morton, Minnesota |
28,400 | May 2012 | Two (3 yr.) | Manufacturing, Warehouse and Offices | ||||||
| Norristown, Pennsylvania |
3,790 | January 2013 | None | Warehouse and Offices | ||||||
| North Ridgeville, Ohio |
152,861 | Own | — | Manufacturing, Warehouse and Offices | ||||||
| Norwood, Massachusetts |
15,000 | February 2014 | One (3 yr.) | Warehouse and Offices | ||||||
| Pharr, Texas |
4,375 | November 2012 | None | Warehouse and Offices | ||||||
| Pinellas Park, Florida |
11,400 | July 2011 | None | Manufacturing and Offices | ||||||
| Pinellas Park, Florida |
3,200 | June 2011 | Two (1 yr.) | Manufacturing | ||||||
| Reynosa, Mexico |
152,256 | Own | — | Manufacturing and Offices | ||||||
| Sacramento, California |
26,900 | May 2011 | None | Manufacturing, Warehouse and Offices | ||||||
| Sanford, Florida |
116,272 | Own | — | Manufacturing and Offices | ||||||
| Scarborough, Ontario |
5,428 | February 2014 | None | Manufacturing and Offices | ||||||
| Shenzhen, China |
4,020 | September 2012 | None | Offices | ||||||
| Simi Valley, California |
38,501 | February 2014 | One (5 yr.) | Manufacturing, Warehouse and Offices | ||||||
| Spicewood, Texas |
6,500 | Month to Month | None | Manufacturing and Offices | ||||||
| Suzhou, China |
11,841 | December 2012 | None | Manufacturing and Offices | ||||||
| Suzhou, China |
86,863 | October 2012 | None | Manufacturing and Offices | ||||||
| Tonawanda, New York |
7,515 | March 2013 | None | Warehouse and Offices | ||||||
| Vaughan, Ontario |
26,637 | December 2015 | None | Manufacturing and Offices | ||||||
| Wallingford, Connecticut |
4,000 | December 2013 | One (3 yr.) | Warehouse and Offices | ||||||
| Warwick, Rhode Island |
3,100 | Month to Month | One (1 yr.) | Warehouse and Offices | ||||||
| Woburn, Massachusetts |
5,200 | Month to Month | None | Warehouse and Offices | ||||||
| Invacare Supply Group |
||||||||||
| Grand Prairie, Texas |
87,508 | August 2015 | One (5 yr.) | Warehouse and Offices | ||||||
| Jacksonville, Florida |
79,652 | September 2014 | Two (3 yr.) | Warehouse and Offices | ||||||
| Jamesburg, New Jersey |
83,200 | Month to Month | None | Warehouse and Offices | ||||||
| Milford, Massachusetts |
29,582 | December 2015 | None | Offices | ||||||
| Rancho Cucamonga, California |
55,890 | February 2012 | None | Warehouse and Offices | ||||||
| South Bend, Indiana |
68,121 | September 2011 | None | Warehouse and Offices | ||||||
| Institutional Products Group |
||||||||||
| Elkhart, Indiana |
44,718 | Month to Month | Two (5 yr.) | Manufacturing, Warehouse and Offices | ||||||
| Elkhart, Indiana |
12,000 | April 2011 | One (1 yr.) | Manufacturing | ||||||
| London, Ontario |
103,200 | Own | — | Manufacturing and Offices | ||||||
| St. Louis, Missouri |
8,196 | July 2013 | Two (3 yr.) | Offices | ||||||
| Asia/Pacific Operations |
||||||||||
| Auckland, New Zealand |
30,518 | September 2014 | None | Manufacturing, Warehouse and Offices | ||||||
| Banyo, QLD, Australia |
26,791 | July 2013 | One (5 yr.) | Warehouse and Offices | ||||||
| Beverley, SA, Australia |
9,601 | December 2013 | One (3 yr.) | Warehouse and Offices | ||||||
| Broadview, SA, Australia |
16,146 | October 2011 | One (5 yr.) | Warehouse and Offices | ||||||
I-30
Table of Contents
| Asia/Pacific Operations |
Square Feet |
Ownership Or Expiration Date of Lease |
Renewal Options |
Use | ||||||
| Carrum Downs, VIC, Australia |
16,006 | December 2012 | One (5 yr.) | Warehouse and Offices | ||||||
| Christchurch, New Zealand |
13,691 | December 2014 | Two (6 yr.) | Offices | ||||||
| Christchurch, New Zealand |
22,027 | December 2014 | One (3 yr.) | Manufacturing, Warehouse and Offices | ||||||
| Kidderminster, United Kingdom |
6,200 | January 2018 | None | Warehouse and Offices | ||||||
| Malaga, WA, Australia |
8,396 | April 2011 | None | Warehouse and Offices | ||||||
| North Olmsted, Ohio |
2,280 | October 2013 | One (3 yr.) | Warehouse and Offices | ||||||
| Southport, QLD, Australia |
1,119 | Month to Month | None | Retail | ||||||
| Suzhou, China |
41,290 | September 2013 | None | Warehouse and Offices | ||||||
| North Rocks, NSW, Australia |
45,712 | August 2012 | Two (3 yr.) | Warehouse and Offices | ||||||
| European Operations |
||||||||||
| Albstadt, Germany |
78,523 | February 2018 | Two (5 yr.) | Manufacturing, Warehouse and Offices | ||||||
| Anderstorp, Sweden |
47,576 | Own | — | Manufacturing, Warehouse and Offices | ||||||
| Bergen, Norway |
1,076 | November 2012 | One (5 yr.) | Warehouse and Offices | ||||||
| Brondby, Denmark |
17,922 | June 2011 | One (1 yr.) | Warehouse and Offices | ||||||
| Dio, Sweden |
110,524 | Own | — | Manufacturing, Warehouse and Offices | ||||||
| Dublin, Ireland |
5,000 | December 2024 | Three (5 yr.) | Warehouse and Offices | ||||||
| Ede, The Netherlands |
12,917 | November 2011 | One (5 yr.) | Warehouse | ||||||
| Ede, The Netherlands |
9,257 | November 2011 | One (5 yr.) | Warehouse and Offices | ||||||
| Fondettes, France |
191,856 | Own | — | Manufacturing and Warehouse | ||||||
| Girona, Spain |
14,639 | January 2012 | One (1 yr.) | Warehouse and Offices | ||||||
| Gland, Switzerland |
5,586 | September 2012 | One (5 yr.) | Offices | ||||||
| Gland, Switzerland |
1,184 | September 2012 | One (4 yr.) | Offices | ||||||
| Goteborg, Sweden |
7,502 | September 2012 | One (3 yr.) | Warehouse and Offices | ||||||
| Hong, Denmark |
155,541 | Own | — | Manufacturing, Warehouse and Offices | ||||||
| Isny, Germany |
47,232 | Own | — | Manufacturing, Warehouse and Offices | ||||||
| Isny, Germany |
1,615 | Own | — | Warehouse | ||||||
| Loppem, Belgium |
4,036 | March 2015 | One (3 yr.) | Warehouse and Offices | ||||||
| Mondsee, Austria |
2,153 | March 2011 | One (3 yr.) | Warehouse and Offices | ||||||
| Odense, Denmark |
1,776 | June 2011 | One (1 yr.) | Warehouse and Offices | ||||||
| Oporto, Portugal |
88,270 | December 2015 | One (1 yr.) | Manufacturing, Warehouse and Offices | ||||||
| Oskarshamn, Sweden |
3,552 | April 2011 | One (1 yr.) | Warehouse | ||||||
| Oslo, Norway |
36,414 | August 2011 | None | Manufacturing, Warehouse and Offices | ||||||
| Pencoed, United Kingdom |
150,000 | December 2019 | None | Manufacturing and Offices | ||||||
| Porta Westfalica, Germany |
134,563 | October 2021 | Two (5yr.) | Manufacturing, Warehouse and Offices | ||||||
| Spanga, Sweden |
3,229 | December 2013 | One (3 yr.) | Warehouse and Offices | ||||||
| Spanga, Sweden |
16,146 | Own | — | Warehouse and Offices | ||||||
| Thiene, Italy |
21,528 | Own | — | Warehouse and Offices | ||||||
| Thiene, Italy |
10,764 | October 2012 | None | Warehouse | ||||||
| Trondheim, Norway |
3,229 | May 2011 | One (3 yr.) | Services and Offices | ||||||
| Witterswil, Switzerland |
40,343 | March 2015 | One (5 yr.) | Manufacturing, Warehouse and Offices | ||||||
| Witterswil, Switzerland |
2,319 | June 2011 | None | Warehouse | ||||||
| Witterswil, Switzerland |
4,080 | June 2011 | None | Warehouse | ||||||
I-31
Table of Contents
| Item 3. | Legal Proceedings. |
In the ordinary course of its business, Invacare is a defendant in a number of lawsuits, primarily product liability actions in which various plaintiffs seek damages for injuries allegedly caused by defective products. All of the product liability lawsuits have been referred to the company’s captive insurance company and/or excess insurance carriers and generally are contested vigorously. The coverage territory of the company’s insurance is worldwide with the exception of those countries with respect to which, at the time the product is sold for use or at the time a claim is made, the U.S. government has suspended or prohibited diplomatic or trade relations. Management does not believe that the outcome of any of these actions will have a material adverse effect upon the company’s business or financial condition.
As part of its regulatory function, the FDA routinely inspects the sites of medical device companies, and in 2010, the FDA inspected certain of the company’s facilities. In December 2010, the company received a warning letter from the FDA related to documentation and procedures at the company’s Sanford, Florida facility. The letter does not call into question the safety or efficacy of Invacare products, and production has not been impacted. The company is taking these issues very seriously and has added resources to ensure it is addressing all of the FDA’s concerns in a timely manner. The costs associated with making the process improvements indicated in the FDA’s letter are currently not expected to be material; however, at the time of this filing the matter remains pending.
The company received a subpoena in 2006 from the U.S. Department of Justice seeking documents relating to three long-standing and well-known promotional and rebate programs maintained by the company. The company believes that the programs described in the subpoena are in compliance with all applicable laws and the company has cooperated fully with the government investigation. As of February 2011, the subpoena remains pending.
Executive Officers of the Registrant.*
The following table sets forth the names of the executive officers of Invacare, each of whom serves at the pleasure of the Board of Directors, as well as certain other information.
| Name |
Age | Position | ||
| A. Malachi Mixon, III |
70 | Chairman of the Board of Directors | ||
| Gerald B. Blouch |
64 | President and Chief Executive Officer and Director | ||
| Robert K. Gudbranson |
47 | Senior Vice President, Chief Financial Officer and Treasurer | ||
| Anthony C. LaPlaca |
52 | Senior Vice President—General Counsel and Secretary | ||
| Joseph B. Richey, II |
74 | President—Invacare Technologies, Senior Vice | ||
| Louis F.J. Slangen |
63 | Senior Vice President—Corporate Marketing and Chief Product Officer | ||
| Patricia A. Stumpp |
49 | Senior Vice President—Human Resources | ||
| Carl E. Will |
40 | Senior Vice President—Global Commercial Operations |
| * | The description of executive officers is included pursuant to Instruction 3 to Section (b) of Item 401 of Regulation S-K. |
A. Malachi Mixon, III has been a director since 1979. Mr. Mixon served as Chief Executive Officer from 1979 through 2010 and as President until 1996. He has served as Chairman of the Board since 1983. Mr. Mixon serves on the Board of Directors of The Sherwin-Williams Company (NYSE), Cleveland, Ohio, a manufacturer and distributor of coatings and related products and Park-Ohio Holdings Corp. (NASDAQ), Cleveland, Ohio, a diversified manufacturing services and products holding company. Mr. Mixon serves as Chairman Emeritus of the Board of Trustees of The Cleveland Clinic Foundation, Cleveland, Ohio, one of the world’s leading academic medical centers. Mr. Mixon previously served on the Board of Directors of Lamson & Sessions from 1990 until it was sold in November 2007.
I-32
Table of Contents
Gerald B. Blouch has been President and a director of Invacare since November 1996. Effective January 1, 2011, Mr. Blouch became Chief Executive Officer of Invacare, after serving as interim Chief Executive Officer from April 2010 through December 2010. Mr. Blouch served as Chief Operating Officer from December 1994 through December 2010 and has served as Chairman—Invacare International since December 1993. Previously, Mr. Blouch was President—Homecare Division from March 1994 to December 1994 and Senior Vice President—Homecare Division from September 1992 to March 1994. Mr. Blouch served as Chief Financial Officer of Invacare from May 1990 to May 1993 and Treasurer of Invacare from March 1991 to May 1993.
Robert K. Gudbranson was appointed Senior Vice President and Chief Financial Officer in April 2008. From October 2005 until his appointment at Invacare, Mr. Gudbranson served as Vice President of Strategic Planning and Acquisitions at Lincoln Electric Holdings, Inc. (NASDAQ: LECO), a $2.0 billion global manufacturer of welding, brazing and soldering products located in Cleveland, Ohio. Prior to joining Lincoln Electric, Mr. Gudbranson served as Director of Business Development and Investor Relations at Invacare from June 2002 to October 2005. Mr. Gudbranson has also served as Invacare’s Assistant Treasurer and as the European Finance Director.
Anthony C. LaPlaca was appointed Senior Vice President, General Counsel and Secretary effective January 2009. Previously, Mr. LaPlaca served as Vice President and General Counsel for six and a half years with Bendix Commercial Vehicle Systems LLC, a member of the Knorr-Bremse group. Prior to that, he served as Vice President and General Counsel to Honeywell Transportation & Power Systems and General Counsel to Honeywell Commercial Vehicle Systems LLC.
Joseph B. Richey, II has been a director since 1980 and in September 1992 was named President—Invacare Technologies Division and Senior Vice President—Electronic and Design Engineering. Previously, Mr. Richey was Senior Vice President of Product Development from July 1984 to September 1992 and Senior Vice President and General Manager of North American Operations from September 1989 to September 1992. Mr. Richey is also a member of the Board of Trustees for Case Western Reserve University and The Cleveland Clinic Foundation. Mr. Richey previously served on the Board of Directors of Steris Corporation from 1987 to July 2009.
Louis F. J. Slangen was named Senior Vice President—Corporate Marketing and Chief Product Officer in September 2010. Previously, Mr. Slangen served as Senior Vice President—Global Market Development from June 2004 to September 2010; Senior Vice President—Sales & Marketing from December 1994 to June 2004 and from September 1989 to December 1994 was Vice President—Sales and Marketing. Mr. Slangen was also President—Rehab Division from March 1994 to December 1994 and Vice President and General Manager—Rehab Division from September 1992 to March 1994.
Patricia A. Stumpp has been the Senior Vice President—Human Resources since September 2009. Mrs. Stumpp joined Invacare in 1991 and was promoted to her current position in 2009. Prior to her promotion, Mrs. Stumpp served as Director of Compensation & Benefits from January 2001 to August 2009 and as Director of Human Resources Group from August 2006 until August 2009. She also has prior experience in healthcare, small business and the services industry. She holds a B.A. in Psychology and M.B.A. from The University of Toledo.
Carl E. Will has been Senior Vice President—Global Commercial Operations since September 2010. Prior to his September 2010 promotion, Mr. Will served as Senior Vice President—North American Homecare from January 2007 through September 2010 having previously serving as Group Vice President of Standard Products and IPG. Mr. Will is responsible for revenue and earnings across all lines of business, channels and geographies, as well as expanding Invacare’s global market share. Prior to joining Invacare, Mr. Will was responsible for commercial operations at General Electric in the Light Emitting Diode (LED) division and served as a strategic consultant at McKinsey and Company. He received a B.S. degree in Accounting from The Ohio State University and an M.B.A. from the Fuqua School of Business at Duke University.
I-33
Table of Contents
PART II
| Item 5. | Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities. |
Invacare’s Common Shares, without par value, trade on the New York Stock Exchange (NYSE) under the symbol “IVC.” Ownership of the company’s Class B Common Shares (which are not listed on NYSE) cannot be transferred, except, in general, to family members without first being converted into Common Shares. Class B Common Shares may be converted into Common Shares at any time on a share-for-share basis. The number of record holders of the company Common Shares and Class B Common Shares at February 23, 2011 was 3,087 and 22, respectively. The closing sale price for the Common Shares on February 23, 2011 as reported by NYSE was $29.50. The prices set forth below do not include retail markups, markdowns or commissions.
The range of high and low quarterly prices of the Common Shares and dividends in each of the two most recent fiscal years were as follows:
| 2010 | 2009 | |||||||||||||||||||||||
| High | Low | Cash Dividends Declared |
High | Low | Cash Dividends Declared |
|||||||||||||||||||
| Quarter Ended: |
||||||||||||||||||||||||
| December 31 |
$ | 30.71 | $ | 26.52 | $ | 0.0125 | $ | 26.19 | $ | 21.22 | $ | 0.0125 | ||||||||||||
| September 30 |
26.51 | 20.00 | 0.0125 | 23.55 | 17.02 | 0.0125 | ||||||||||||||||||
| June 30 |
27.50 | 21.02 | 0.0125 | 17.70 | 15.06 | 0.0125 | ||||||||||||||||||
| March 31 |
30.16 | 24.52 | 0.0125 | 19.81 | 14.67 | 0.0125 | ||||||||||||||||||
During 2010 and 2009, the Board of Directors also declared annualized dividends of $0.045 per Class B Common Share. For information regarding limitations on the payment of dividends in the company loan and note agreements, see Long Term Debt in the Notes to the Consolidated Financial Statements included in this report. The Common Shares are entitled to receive cash dividends at a rate of at least 110% of cash dividends paid on the Class B Common Shares.
I-34
Table of Contents
SHAREHOLDER RETURN PERFORMANCE GRAPH
The following graph compares the yearly cumulative total return on Invacare’s common shares against the yearly cumulative total return of the companies listed on the Standard & Poor’s 500 Stock Index, the Russell 2000 Stock Index and the S&P Healthcare Equipment & Supplies Index*.
COMPARISON OF 5 YEAR CUMULATIVE TOTAL RETURN*
Among Invacare Corporation, The S&P 500 Index,
The Russell 2000 Index And S&P Healthcare Equipment & Supplies
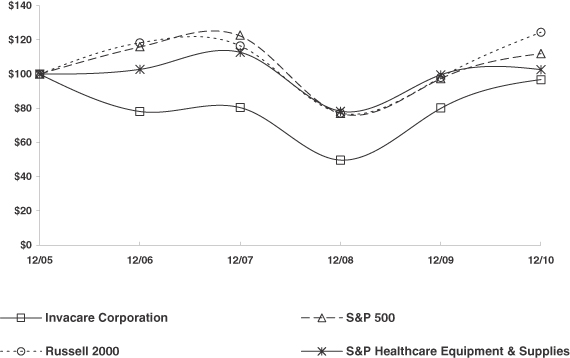
| 12/05 | 12/06 | 12/07 | 12/08 | 12/09 | 12/10 | |||||||||||||||||||
| Invacare Corporation |
$ | 100.00 | $ | 78.11 | $ | 80.38 | $ | 49.63 | $ | 79.95 | $ | 96.83 | ||||||||||||
| S&P 500 |
100.00 | 115.80 | 122.16 | 76.96 | 97.33 | 111.99 | ||||||||||||||||||
| Russell 2000 |
100.00 | 118.37 | 116.51 | 77.15 | 98.11 | 124.46 | ||||||||||||||||||
| S&P Healthcare Equipment & Supplies |
100.00 | 102.69 | 112.74 | 78.33 | 99.52 | 102.60 | ||||||||||||||||||
Copyright © 2011 S&P, a division of The McGraw-Hill Companies Inc. All rights reserved.
| * | The S&P Healthcare Equipment & Supplies Index is a capitalization-weighted average index comprised of health care companies in the S&P 500 Index. |
The graph assumes $100 invested on December 31, 2005 in the common shares of Invacare Corporation, S&P 500 Index, Russell 2000 Index and the S&P Healthcare Equipment & Supplies Index, including reinvestment of dividends, through December 31, 2010.
I-35
Table of Contents
The following table presents information with respect to repurchases of common shares made by the company during the three months ended December 31, 2010.
| Period |
Total Number of Shares Purchased (1) |
Average Price Paid Per Share |
Total Number of Shares Purchased as Part of Publicly Announced Plans or Programs |
Maximum Number of Shares That May Yet Be Purchased Under the Plans or Programs (2) |
||||||||||||
| 10/1/2010-10/31/10 |
— | $ | — | — | 1,362,900 | |||||||||||
| 11/1/2010-11/30/10 |
233,990 | 26.59 | 185,000 | 1,177,900 | ||||||||||||
| 12/1/2010-12/31/10 |
20,435 | 29.00 | 20,000 | 1,157,900 | ||||||||||||
| Total |
254,425 | $ | 26.78 | 205,000 | 1,157,900 | |||||||||||
| (1) | Includes 48,990 shares repurchased between November 1, 2010 and November 30, 2010 and 435 shares repurchased between December 1, 2010 and December 31, 2010 that were surrendered to the company by employees for tax withholding purposes in conjunction with the vesting of restricted shares held by the employees under the company’s 2003 Performance Plan. |
| (2) | On August 17, 2001, the Board of Directors authorized the company to purchase up to 2,000,000 Common Shares, excluding any shares acquired from employees or directors as a result of the exercise of options or vesting of restricted shares pursuant to the company’s performance plans. The Board of Directors reaffirmed its authorization of this repurchase program on November 5, 2010. To date, the company has purchased 842,100 shares with authorization remaining to purchase 1,157,900 more shares. The company purchased 205,000 shares pursuant to this Board authorized program during 2010. |
During 2010, the company purchased a total of $57,790,000 in principal amount of its outstanding 4.125% Convertible Senior Subordinated Debentures due 2027 in open market transactions for an aggregate of approximately $69,242,000, plus accrued and unpaid interest. During the first nine months of 2010, the company purchased a total of $29,000,000 in principal amount of its outstanding 9 3/4% Senior Notes due 2015 in open market transactions for an aggregate of approximately $31,213,000. On November 1, 2010, the company purchased an aggregate of $142,945,000 in principal amount of the 9 3/4% Senior Notes due 2015 in a tender offer conducted by the company. The company paid $1,075.00 for each $1,000 principal amount of the 9 3/4% Senior Notes due 2015 validly tendered in the tender offer, which included a consent payment of $30.00 per $1,000 principal amount of the 9 3/4% Senior Notes due 2015. In the tender offer, the company also paid accrued and unpaid interest on the purchased 9 3/4% Senior Notes due 2015 up to, but not including, November 1, 2010. On December 31, 2010, the company redeemed the remaining $3,055,000 principal amount of 9 3/4% Senior Notes due 2015 for an aggregate amount of approximately $3,237,000, plus accrued and unpaid interest. The company may continue from time to time seek to retire or purchase the Company’s outstanding 4.125% Convertible Senior Subordinated Debentures due 2027, in open market purchases, privately negotiated transactions or otherwise.
I-36
Table of Contents
| Item 6. | Selected Financial Data. |
The selected consolidated financial data set forth below with respect to the company’s consolidated statements of operations, cash flows and shareholders’ equity for the fiscal years ended December 31, 2010, 2009 and 2008, and the consolidated balance sheets as of December 31, 2010 and 2009 are derived from the Consolidated Financial Statements included elsewhere in this Form 10-K. The consolidated statements of earnings, cash flows and shareholders’ equity data for the fiscal years ended December 31, 2007 and 2006 and consolidated balance sheet data for the fiscal years ended December 31, 2008, 2007 and 2006 are derived from the company’s previously filed Consolidated Financial Statements. The data set forth below should be read in conjunction with Item 7—“Management’s Discussion and Analysis of Financial Condition and Results of Operations” and the company’s Consolidated Financial Statements and Notes thereto included elsewhere in this Form 10-K.
| 2010 * | 2009 ** | 2008 *** | 2007 **** | 2006 ***** | ||||||||||||||||
| (In thousands, except per share and ratio data) | ||||||||||||||||||||
| Earnings |
||||||||||||||||||||
| Net Sales |
$ | 1,722,081 | $ | 1,693,136 | $ | 1,755,694 | $ | 1,602,237 | $ | 1,498,035 | ||||||||||
| Net Earnings (loss) |
25,341 | 41,179 | 34,857 | (1,714 | ) | (317,774 | ) | |||||||||||||
| Net Earnings (loss) per Share—Basic |
.78 | 1.29 | 1.09 | (0.05 | ) | (10.00 | ) | |||||||||||||
| Net Earnings (loss) per Share—Assuming Dilution |
.78 | 1.29 | 1.09 | (0.05 | ) | (10.00 | ) | |||||||||||||
| Dividends per Common Share |
0.05 | 0.05 | 0.05 | 0.05 | 0.05 | |||||||||||||||
| Dividends per Class B Common Share |
0.04545 | 0.04545 | 0.04545 | 0.04545 | 0.04545 | |||||||||||||||
| Balance Sheet |
||||||||||||||||||||
| Current Assets |
$ | 526,159 | $ | 528,464 | $ | 551,058 | $ | 591,085 | $ | 655,758 | ||||||||||
| Total Assets |
1,280,400 | 1,359,501 | 1,314,473 | 1,500,042 | 1,490,451 | |||||||||||||||
| Current Liabilities |
290,308 | 290,327 | 284,998 | 326,611 | 447,976 | |||||||||||||||
| Working Capital |
235,851 | 238,137 | 266,060 | 264,474 | 207,782 | |||||||||||||||
| Long-Term Debt |
238,090 | 272,234 | 407,707 | 457,233 | 448,883 | |||||||||||||||
| Other Long-Term Obligations |
99,591 | 95,703 | 88,826 | 106,046 | 107,223 | |||||||||||||||
| Shareholders’ Equity |
652,411 | 701,237 | 532,942 | 610,152 | 486,369 | |||||||||||||||
| Other Data |
||||||||||||||||||||
| Research and Development Expenditures |
$ | 25,954 | $ | 25,725 | $ | 24,764 | $ | 22,491 | $ | 22,146 | ||||||||||
| Capital Expenditures |
17,353 | 17,999 | 19,957 | 20,068 | 21,789 | |||||||||||||||
| Depreciation and Amortization |
36,804 | 40,562 | 43,744 | 43,717 | 39,892 | |||||||||||||||
| Key Ratios |
||||||||||||||||||||
| Return on Sales % |
1.5 | 2.4 | 2.0 | (.1 | ) | (21.2 | ) | |||||||||||||
| Return on Average Assets % |
1.9 | 3.1 | 2.5 | (.1 | ) | (20.3 | ) | |||||||||||||
| Return on Beginning Shareholders’ Equity % |
3.6 | 7.7 | 5.7 | (.4 | ) | (42.2 | ) | |||||||||||||
| Current Ratio |
1.8:1 | 1.8:1 | 1.9:1 | 1.8:1 | 1.5:1 | |||||||||||||||
| Debt-to-Equity Ratio |
0.4:1 | 0.4:1 | 0.8:1 | 0.7:1 | 0.9:1 | |||||||||||||||
| * | Reflects loss on debt extinguishment including debt finance charges and associated fees of $40,164 ($40,164 after tax or $1.23 per share assuming dilution) as a result of the company’s decision to extinguish higher interest rate debt. |
| ** | Reflects restructuring charge of $4,804 ($4,124 after tax or $.13 per share assuming dilution); loss on debt extinguishment including debt fees $2,878 ($2,878 after tax or $.09 per share assuming dilution); asset write-downs for intangibles and investments of $8,409 ($7,909 after tax or $.25 per share assuming dilution). |
| *** | Reflects restructuring charge of $4,766 ($4,516 after tax or $.14 per share assuming dilution). |
| **** | Reflects restructuring charge of $11,408 ($10,478 after tax or $.33 per share assuming dilution) and $13,408 expense related to finance charges, interest and fees associated with the company’s previously reported debt covenant violations ($13,408 after tax or $.42 per share assuming dilution). |
| ***** | Reflects restructuring charge of $21,250 ($18,700 after tax or $.59 per share assuming dilution), $3,745 expense related to finance charges, interest and fees associated with the company’s previously reported debt covenant violations ($3,300 after tax or $.10 per share assuming dilution), $26,775 expense related to accounts receivable collectability issues arising primarily from Medicare reimbursement reductions for power wheelchairs announced on November 15, 2006 ($26,775 after tax or $.84 per share assuming dilution), $300,417 expense for an impairment charge related to the write-down of goodwill and other intangible assets ($300,417 after tax or $9.45 per share assuming dilution). |
I-37
Table of Contents
| Item 7. | Management’s Discussion and Analysis of Financial Condition and Results of Operations. |
OUTLOOK
Invacare returned to organic net sales growth in 2010 and plans to increase the growth rate in 2011. As the company shifts to a global operating model, it intends to leverage its regional product strengths into its other geographic markets. For example, the company will be introducing a new bath lift in the United States that was developed and has already enjoyed success in Europe. The company also has identified a number of internal opportunities to reduce complexity and drive organic net sales. For instance, the company is integrating its Motion Concepts seating and positioning subsidiary with its existing Invacare® branded seating products. The combined efforts of the Invacare and Motion Concepts teams are expected to lead to improved product design through one product development center, an enhanced sales organization that is focused on one united goal and greater ease in sales and technical support for customers. Similarly, the company intends to deploy its research and development efforts more efficiently to accelerate new product introductions that are expected to show benefits during the year. For instance, the new Invacare® FDX® power wheelchair, launched in Europe and the United States in 2010, is the company’s first global power wheelchair platform and it has already started to gain traction with clinicians and providers.
The company will benefit from interest savings in 2011, as a result of paying down higher interest rate debt, namely the $146 million of 9 3/4% senior notes retired in the fourth quarter of 2010. However, this benefit will be partially offset by a number of items. First, the new revolving credit facility entered into by the company in October 2010 bears a higher interest rate than the previous revolving credit facility. Second, the company’s indebtedness has increased as a result of the debt finance charges, premium and fees paid during 2010 to extinguish the previous debt structure. In addition, the new credit facility affords Invacare the opportunity to pursue acquisitions or buy back company stock.
The company’s organic net sales growth and interest savings will likely be partially offset by the potential for continued increases in freight and commodity costs, particularly in aluminum and steel, which are already being incurred in 2011. Additionally, as Invacare plans for its business in the United States to continue to improve, the company’s overall effective tax rate on adjusted pre-tax earnings is expected to increase, since the United States tax rate is the highest of the countries in which it does business. The company anticipates continued volatility related to foreign exchange rates which could be a benefit or a detriment. The company may also manage potential increases in LIBOR rates by entering into interest rate swap agreements to fix some of its exposure.
In regards to reimbursement, the company is mindful of three key issues. In the United States, the Centers for Medicaid and Medicare Services is moving forward with National Competitive Bidding in the first nine metropolitan areas. While the company expects this to be neutral to earnings in 2011, it will remain judicious in its extension of credit to customers and it will monitor whether other payors begin to model their payments on this system. The company also will closely watch State Medicaid budgets and how deficits may impact coverage and payments for home medical equipment and institutional care products. In the European segment, there is discussion by the French government of reduced wheelchair reimbursement in the second half of 2011. This issue was originally anticipated to occur in 2010, but it was delayed.
As part of its regulatory function, the FDA routinely inspects the sites of medical device companies, and in 2010, the FDA inspected certain of the company’s facilities. In December 2010, the company received a warning letter from the Food and Drug Administration (FDA) related to documentation and procedures at the company’s Sanford, Florida facility. The letter does not call into question the safety or efficacy of Invacare products, and production has not been impacted. The company is taking these issues very seriously and has added resources to ensure it is addressing all of the FDA’s concerns in a timely manner. The costs related to making the process improvements are not expected to be material and have been included in the company’s 2011 operating plan and guidance.
I-38
Table of Contents
Organic sales growth, earnings and cash flow for 2011 are expected to be consistent with the guidance provided in the company’s February 3, 2011 press release. The guidance should be read in conjunction with the information contained herein under “Risk Factors” and “Forward-Looking Information.”
RESULTS OF OPERATIONS
2010 Versus 2009
Net Sales. Consolidated net sales for 2010 increased 1.7% for the year, to $1,722,081,000 from $1,693,136,000 in 2009. Foreign currency translation increased net sales by less 0.3 of a percentage point while acquisitions increased sales by 0.4 of a percentage point. The organic net sales increase was 1.0% which was driven by growth in ISG, Europe and Asia/Pacific segments.
North America/Home Medical Equipment
NA/HME net sales decreased 0.1% in 2010 versus the prior year to $747,599,000 from $748,401,000 with an acquisition increasing net sales by 0.9 of a percentage point while foreign currency translation increased net sales by 0.7 of a percentage point. The organic net sales decline of 1.7% was driven by a decline in the Respiratory product line partially offset by increases in Standard and Rehab product lines. Respiratory product line net sales decreased by 16.9% in 2010, primarily driven by lower sales of both concentrators and HomeFill® oxygen delivery systems to national providers. Standard product line net sales improved by 2.6% in 2010, driven by increased volumes in standard wheelchairs, beds and therapeutic support surfaces. Rehab product line net sales increased by 2.0% in 2010 primarily driven by increases in custom power products.
Invacare Supply Group
ISG net sales increased 6.1% in 2010 over the prior year to $297,517,000 from $280,295,000. The net sales increase was primarily in the result of volume increases in diabetic, incontinence, ostomy and urological products.
Institutional Products Group
IPG net sales decreased 1.3% in 2010 over the prior year to $88,261,000 from $89,423,000. Foreign currency translation increased net sales by 0.7 of a percentage point. The organic net sales decrease of 2.0% was largely driven by continued weakness in capital expenditures by nursing home customers, due primarily to budgetary pressures in state Medicaid programs.
Europe
European net sales increased 0.6% in 2010 compared to the prior year to $506,069,000 from $503,084,000 with foreign currency translation decreasing net sales by 1.9 percentage points. Organic net sales increased 2.5% attributable to increases in France, U.K., Germany and Sweden and increases in Standard and Respiratory product lines.
Asia/Pacific
Asia/Pacific net sales increased 14.9% in 2010 from the prior year to $82,635,000 from $71,933,000. Foreign currency translation increased net sales by 12.9 percentage points. The organic net sales growth of 2.0% was driven by the Company’s New Zealand distribution business and increased demand for product from the Company’s subsidiary which produces microprocessor controllers. Changes in exchange rates, particularly with the Euro and U.S. Dollar, have had, and may continue to have, a significant impact on sales in this segment.
I-39
Table of Contents
Gross Profit. Consolidated gross profit as a percentage of net sales was 29.6% in 2010 as compared to 29.1% in 2009. The margin improvement was primarily the result of volume increases and cost reduction activities, including warranty costs. Gross profit as a percentage of net sales for NA/HME, IPG and Asia/Pacific segments were favorable as compared to the prior year with ISG and European segments unfavorable to the prior year.
NA/HME gross profit as a percentage of net sales increased by 1.5 percentage points in 2010 versus 2009. The improvement in margins was primarily a result of cost reduction initiatives including freight and warranty expenses.
ISG gross profit as a percentage of net sales decreased 0.6 percentage points in comparison to the prior year. The decrease was primarily as a result of unfavorable product mix to lower margin diabetic and ostomy products partially offset by volume increases and cost reduction programs including freight costs.
IPG gross profit as a percentage of net sales increased 0.8 percentage points in 2010 from the prior year. The increase in margin is primarily attributable to cost reduction activities including freight costs partially offset by reduced volumes.
Gross profit in Europe as a percentage of net sales declined 0.4 percentage points in 2010 from the prior year. The decrease was primarily a result of unfavorable product mix toward lower margin product and unfavorable foreign currency transactions partially offset by cost reduction activities associated with commodity costs.
Gross profit in Asia/Pacific as a percentage of net sales increased by 4.3 percentage points in 2010 from the prior year. The improvement was primarily as a result of increased volumes and favorable foreign currency impact principally due to the strengthening of the U.S. dollar.
Selling, General and Administrative. Consolidated selling, general and administrative expenses as a percentage of net sales were 23.9% in 2010 and 23.5% in 2009. The overall dollar increase was $12,867,000 or 3.2%, with foreign currency translation increasing expenses by $4,869,000 or 1.2 percentage points and an acquisition increasing expenses by approximately $4,455,000 or 1.1 percentage points. Excluding acquisitions and the impact of foreign currency translation, selling, general and administrative (SG&A) expenses increased $3,543,000 or 0.9%. This increase is primarily attributable to increased associate costs and higher legal expenses related to enforcement of intellectual property rights.
SG&A expenses for NA/HME increased 4.8% or $9,950,000 in 2010 compared to 2009. An acquisition increased these expenses by approximately $4,455,000 while foreign currency increased SG&A expense by $1,672,000. Excluding the acquisition and foreign currency translation, SG&A expense increased $3,823,000 or 1.8% primarily due to increased associate costs, and higher legal expenses related to enforcement of intellectual property rights. In addition, the SG&A expenses for 2010 include an impairment charge related to a customer list of $248,000 recorded as a result of the company’s 2010 intangible impairment review.
SG&A expenses for ISG decreased by 4.8% or $1,357,000 in 2010 compared to 2009. The decrease is primarily attributable to a decrease in distribution and marketing costs partially offset by increased bad debt expense.
SG&A expenses for IPG increased by 16.8% or $2,599,000 in 2010 compared to 2009. Foreign currency translation increased SG&A expenses by 1.6 percentage points or $242,000. Excluding the impact of foreign currency translation, SG&A expenses increased by $2,357,000 due to increased associate costs and unfavorable currency transaction effects associated with the Canadian Dollar versus the U.S. Dollar. In addition, the SG&A expenses for 2010 include an impairment charge related to a trademark of $336,000 recorded as a result of the company’s 2010 intangible impairment review.
I-40
Table of Contents
European SG&A expenses decreased by 0.5% or $568,000 in 2010 compared to 2009. Foreign currency translation decreased SG&A expenses by approximately $390,000. Excluding the foreign currency translation impact, SG&A expenses decreased by $178,000.
Asia/Pacific SG&A expenses increased 8.8% or $2,243,000 in 2010 compared to 2009. Foreign currency translation increased expenses by $3,345,000. Excluding the foreign currency translation impact, SG&A expenses decreased $1,102,000 or 4.3% primarily due to favorable currency transactions partially offset by increased associate costs.
Debt Finance Charges and Fees Associated with Debt Refinancing. In 2010, as part of the company’s refinancing, proceeds of the refinancing were used by the company to repay amounts outstanding on its $150,000,000 revolving credit facility which was not due to expire until February 2012 and repurchase all of its outstanding 9.75% Senior Notes which were not due until February 2015. During 2010, the company also extinguished $57,799,000 in principal amount of its outstanding 4.125% convertible senior subordinated debentures due in February 2027. This early debt extinguishment resulted in debt fees and premium expenses of $40,164,000 for all of these debt instruments.
Related to the revolving credit facility, the company expensed $1,228,000 of deferred financing fees, which were previously capitalized. Related to the senior notes, the company incurred the following debt fees and premium expenses: debt deferred financing fees of $3,764,000, which were previously capitalized and premiums and fees associated with the early extinguishment of the debt of $14,907,000. Related to the convertible senior subordinated debentures, the company incurred $18,763,000 of premiums paid and losses recorded as a result of early debt extinguishment and expensed deferred financing fees of $1,502,000, which were previously capitalized.
In 2009, the company fully repaid its $250,000,000 term loan facility which was not due to expire until February 2013. As a result, deferred financing fees of $2,878,000, which were previously capitalized, were expensed. All of these charges in 2010 and 2009 are included in the All Other segment.
Asset write-downs to intangibles and investments. The company has made other investments in limited partnerships and non-marketable equity securities, which are accounted for using the cost method, adjusted for any estimated declines in value. These investments were acquired in private placements and there are no quoted market prices or stated rates of return and the company does not have the ability to easily sell these investments. In 2009, the company recognized an impairment charge totaling $6,713,000 on investments along with an impairment charge of $1,696,000 on its intangibles. The company completed an evaluation of the residual value related to its investments in the fourth quarter of 2010 and recognized an immaterial loss. These charges are included in the All Other Segment.
Interest. Interest expense decreased to $20,647,000 in 2010 from $33,150,000 in 2009, representing a 37.7% decrease. This decrease was attributable to debt reduction during the year and, to a lesser extent, decreased borrowing rates in 2010 compared to 2009. Interest income in 2010 was $724,000, which was lower than the prior year amount of $1,674,000, primarily due to decreased volume of financing provided to customers.
Income Taxes. The company had an effective tax rate of 33.4% in 2010 and 12.9% in 2009. The company’s effective tax rate is lower than the expected U.S. federal statutory rate due to earnings abroad being taxed at rates lower than the U.S. statutory rate. In both years, the company’s rate was higher than it otherwise would have been due to losses without benefit, and due to valuation allowances in the United States, Australia and New Zealand. In addition, the 2009 tax rate was lower than the 2010 rate primarily due to a loss carryback, resulting from a tax law change in the United States, which previously was fully offset by a valuation allowance. See “Income Taxes” in the Notes to the Consolidated Financial Statements included elsewhere in this report for more detail.
I-41
Table of Contents
Research and Development. The company continues to invest in research and development activities to maintain its competitive advantage. The company dedicates funds to applied research activities to ensure that new and enhanced design concepts are available to its businesses. Research and development expenditures, which are included in costs of products sold, increased to $25,954,000 in 2010 from $25,725,000 in 2009. The expenditures, as a percentage of net sales, were 1.5% and 1.5% in 2010 and 2009, respectively.
2009 Versus 2008
Charge Related to Restructuring Activities. Throughout 2009, the company continued its cost reduction and profit improvement initiatives, which now are substantially complete as related to restructuring activities. The company has achieved tremendous benefits from its cost reduction initiatives, principally related to product sourcing savings, headcount reductions and manufacturing consolidation. However, as was expected, a significant portion of this benefit was offset by continued pricing pressures and product mix shift toward lower margin product, primarily in the U.S. and Europe, as a result of reimbursement changes.
Restructuring charges of $4,804,000 were incurred during 2009 of which $298,000 was recorded in cost of goods sold, since it relates to inventory markdowns, and the remaining charge amount was included in the Charge Related to Restructuring Activities in the Consolidated Statement of Earnings. The costs incurred during 2009 were principally for severance expenses.
Net Sales. Consolidated net sales for 2009 decreased 3.6% for the year, to $1,693,136,000 from $1,755,694,000 in 2008. Foreign currency translation decreased net sales by four percentage points while acquisitions increased sales by less than one percentage point. The remaining increase was driven by performance in NA/HME, ISG and Europe.
North America/Home Medical Equipment
NA/HME net sales increased 0.9% in 2009 versus the prior year to $748,401,000 from $741,502,000 with acquisitions increasing net sales by one percentage point while foreign currency translation decreased net sales by one percentage point. Standard product line net sales improved by 5.5% in 2009, driven by increased volumes in beds, patient transport and therapeutic support surfaces products. Rehab product line net sales decreased by 0.6% in 2009, despite volume increases in custom power products. Respiratory product line net sales decreased by 8.1% in 2009, primarily driven by lower sales of HomeFill® oxygen delivery systems to national providers.
Invacare Supply Group
ISG net sales increased 5.4% in 2009 over the prior year to $280,295,000 from $265,818,000. Acquisitions and foreign currency translation had no impact on the sales increase. The net sales increase was primarily in diabetic, incontinence and wound care products.
Institutional Products Group
IPG net sales decreased 10.3% in 2009 over the prior year to $89,423,000 from $99,662,000. Foreign currency translation decreased net sales by approximately one percentage point. The net sales decrease was largely driven by continued weakness in capital expenditures by nursing home customers, due primarily to budgetary pressures in state Medicaid programs.
Europe
European net sales decreased 9.2% in 2009 compared to the prior year to $503,084,000 from $553,845,000 with foreign currency translation decreasing net sales by nine percentage points. The net sales decrease was the result of sales declines primarily in France, where sales of beds and wheelchairs into nursing homes weakened as a result of changes in reimbursement rules. This decline was partially offset by favorable net sales performance in the U.K. region.
I-42
Table of Contents
Asia/Pacific
Asia/Pacific net sales decreased 24.2% in 2009 from the prior year to $71,933,000 from $94,867,000. Foreign currency translation decreased net sales by eight percentage points. The sales decline at the company’s subsidiary, which manufactures controllers, was largely due to external customers whose demand for inventory remained weak in the current economic environment. The company’s Australian distribution business had lower sales due in large part to weak demand from long-term care facilities which continue to delay capital purchases. Changes in exchange rates, particularly with the Euro and U.S. Dollar, have had, and may continue to have, a significant impact on sales in this segment.
Gross Profit. Consolidated gross profit as a percentage of net sales was 29.1% in 2009 as compared to 27.8% in 2008. The margin improvement compared to the prior year for all segments except Asia/Pacific and Europe was primarily the result of volume increases and cost reduction activities, including commodity cost and freight reductions.
NA/HME gross profit as a percentage of net sales was 34.1% in 2009 versus 30.5% in 2008. The significant improvement in margins was primarily a result of increased volumes, selective price increases implemented in the second half of 2008 and cost reduction initiatives.
ISG gross profit as a percentage of net sales increased 1.1 percentage points in comparison to the prior year. The improvement was primarily as a result of volume increases, freight reduction programs and reduced discounts associated with lower sales to larger providers.
IPG gross profit as a percentage of net sales increased 5.0 percentage points in 2009 from the prior year. The increase in margin is primarily attributable to selective price increases introduced in the second half of 2008 and cost reduction activities associated with commodity and freight costs.
Gross profit in Europe as a percentage of net sales declined 0.7 percentage points in 2009 from the prior year. The decrease was primarily a result of unfavorable product mix toward lower margin product and unfavorable foreign currency transactions partially offset by cost reduction activities associated with commodity and freight costs.
Gross profit in Asia/Pacific as a percentage of net sales decreased by 7.6 percentage points in 2009 from the prior year. The decrease was primarily as a result of volume declines and unfavorable foreign currency impact principally due to the strengthening of the U.S. dollar.
Selling, General and Administrative. Consolidated selling, general and administrative expenses as a percentage of net sales were 23.5% in 2009 and 22.7% in 2008. The overall dollar increase was $392,000 or 0.1%, with foreign currency translation decreasing expenses by $14,143,000 or four percentage points and acquisitions increasing expenses by approximately $1,804,000 or one percentage point. Excluding acquisitions and foreign currency translation impact, selling, general and administrative (SG&A) expenses increased $12,731,000 or 3.2%. This increase is primarily attributable to higher bad debt expense and unfavorable foreign currency transactions.
SG&A expenses for NA/HME increased 5.4% or $10,604,000 in 2009 compared to 2008. Acquisitions increased these expenses by approximately $1,804,000 while foreign currency decreased SG&A expense by $969,000. Excluding foreign currency translation, SG&A expense increased $9,769,000 or 4.9% primarily due to higher bad debt expense.
SG&A expenses for ISG increased by 6.7% or $1,754,000 in 2009 compared to 2008. The increase is primarily attributable to higher bad debt expense.
I-43
Table of Contents
SG&A expenses for IPG increased by 6.3% or $922,000 in 2009 compared to 2008. Foreign currency translation decreased SG&A expenses by approximately one percentage point or $185,000. Excluding the impact of foreign currency translation, SG&A expenses increased by $1,107,000 due to unfavorable currency transaction effects associated with the Canadian Dollar versus the U.S. Dollar.
European SG&A expenses decreased by 8.0% or $10,593,000 in 2009 compared to 2008. Foreign currency translation decreased SG&A expenses by approximately $9,812,000. Excluding the foreign currency translation impact, SG&A expenses decreased by $781,000.
Asia/Pacific SG&A expenses decreased 8.3% or $2,295,000 in 2009 compared to 2008. Foreign currency translation decreased expenses by $3,177,000. Excluding the foreign currency translation impact, SG&A expenses increased $882,000 or 3.2% primarily due to unfavorable currency transaction effects.
Debt Finance Charges and Fees Associated with Debt Refinancing. In 2009, the company fully repaid its $250,000,000 term loan facility which was not due to expire until February 2013. As a result, deferred financing fees of $2,878,000 pre-tax, which were previously capitalized, were expensed in the All Other operating segment.
Asset write-downs to intangibles and investments. The company has made other investments in limited partnerships and non-marketable equity securities, which are accounted for using the cost method, adjusted for any estimated declines in value. These investments were acquired in private placements and there are no quoted market prices or stated rates of return and the company does not have the ability to easily sell these investments. The company completed an evaluation of the residual value related to these investments in the fourth quarter of 2009 which considered the weakening in the commercial real estate market as well as the redemption of one of the investments for a nominal amount and as a result, the company recognized impairment charges totaling $6,713,000 pre-tax which is included in the All Other segment.
In accordance with ASC 350, Intangibles—Goodwill and Other, the company reviews intangibles for impairment. As a result of the company’s 2009 intangible impairment review, impairment charges of $896,000 and $800,000 were recorded related to trademarks for Europe and a customer list for NA/HME, respectively as the actual cash flows associated with these intangibles were less than what was originally used to value the intangibles.
Interest. Interest expense decreased to $33,150,000 in 2009 from $42,927,000 in 2008, representing a 22.8% decrease. This decrease was attributable to debt reduction during the year and, to a lesser extent, decreased borrowing rates in 2009 compared to 2008. Interest income in 2009 was $1,674,000, which was lower than the prior year amount of $3,045,000, primarily due to decreased volume of financing provided to customers. As a result of the company’s adoption, effective January 1, 2009, of FASB Staff Position APB 14-1, Accounting for Convertible Debt Instruments That May Be Settled in Cash upon Conversion (Including Partial Cash Settlement) (FSP APB 14-1) as codified in Debt with Conversion and Other Options, ASC 470-20, the company’s 2009 financial statements contain restated amounts for 2008 and 2007 that reflect an increase in interest expense of $3,694,000 and $2,904,000 for 2008 and 2007, respectively. See “Accounting Policies” in the Notes to Consolidated Financial Statements included elsewhere in this report.
Income Taxes. The company had an effective tax rate of 12.9% in 2009 and 27.1% in 2008. The company’s effective tax rate is lower than the expected U.S. federal statutory rate due to earnings abroad being taxed at rates lower than the U.S. statutory rate. In both years, the company’s rate was higher than it otherwise would have been due to losses without benefit, and due to valuation allowances in the United States, Australia and New Zealand. In addition, the 2009 tax rate was lower than the 2008 tax rate primarily due to a loss carryback, resulting from a tax law change in the United States, which previously was fully offset by a valuation allowance. See “Income Taxes” in the Notes to the Consolidated Financial Statements included elsewhere in this report for more detail.
I-44
Table of Contents
Research and Development. The company continues to invest in research and development activities to maintain its competitive advantage. The company dedicates funds to applied research activities to ensure that new and enhanced design concepts are available to its businesses. Research and development expenditures, which are included in costs of products sold, increased to $25,725,000 in 2009 from $24,764,000 in 2008. The expenditures, as a percentage of net sales, were 1.5% and 1.4% in 2009 and 2008, respectively.
INFLATION
Although the company cannot determine the precise effects of inflation, management believes that inflation does continue to have an influence on the cost of materials, salaries and benefits, utilities and outside services. The company attempts to minimize or offset the effects through increased sales volumes, capital expenditure programs designed to improve productivity, alternative sourcing of material and other cost control measures. In 2010, 2009 and 2008, the company was able to offset the majority of the impact of price increases from suppliers by productivity improvements, increasing prices to customers and other cost reduction activities.
LIQUIDITY AND CAPITAL RESOURCES
The company continues to maintain an adequate liquidity position through its unused bank lines of credit (see Long-Term Debt in the Notes to Consolidated Financial Statements included in this report) and working capital management.
The company’s debt, including the debt discount (related to the recording of convertible debt in accordance with ASC 470-20 which requires the recording of a debt discount, reducing debt and increasing equity) as described below, decreased by $50,396,000 from $321,597,000 at December 31, 2009 to $271,201,000 at December 31, 2010 as a result of improved cash flow generation. The company’s recorded debt discount was $25,137,000 as of December 31, 2010 and $48,272,000 as of December 31, 2009, respectively. The decrease in the recorded discount during 2010 was principally the result of extinguishing $57,799,000 of convertible debt during the year.
On October 28, 2010, the company entered into a new senior secured revolving credit agreement (the “New Credit Agreement”) which provides for a $400 million senior secured revolving credit facility maturing in October 2015. Pursuant to the terms of the New Credit Agreement, the company may from time to time borrow, repay and re-borrow up to an aggregate outstanding amount at any one time of $400 million, subject to customary conditions. The New Credit Agreement also provides for the issuance of swing line loans and letters of credit.
Borrowings under the New Credit Agreement bear interest, at the company’s election, at (i) the London Inter-Bank Offer Rate (“LIBOR”) plus a margin; or (ii) a Base Rate Option plus a margin. The applicable margin is based on the company’s leverage ratio and at the time of entry into the New Credit Agreement, the applicable margin was 2.50% per annum for LIBOR loans and 1.50% for the Base Rate Option loans. In addition to interest, the company is required to pay commitment fees on the unused portion of the New Credit Agreement. The commitment fee rate is initially 0.40% per annum and, like the interest rate spreads, is subject to adjustment thereafter based on the company’s leverage ratio. The obligations of the borrowers under the New Credit Agreement are secured by substantially all of the company’s U.S. assets and are guaranteed by substantially all of the company’s material domestic and foreign subsidiaries.
The company may from time to time seek to retire or purchase its 4.125% Convertible Senior Subordinated Debentures due 2027, in open market purchases, privately negotiated transactions or otherwise. Such purchases or exchanges, if any, will depend on prevailing market conditions, the company’s liquidity requirements, contractual restrictions and other factors. The amounts involved in any such transactions, individually or in the aggregate, may be material. During 2010, the company extinguished $57,799,000 in convertible senior subordinated debentures.
I-45
Table of Contents
The New Credit Agreement contains certain covenants that are customary for similar credit arrangements, including covenants relating to, among other things, financial reporting and notification, compliance with laws, preservation of existence, maintenance of books and records, use of proceeds, maintenance of properties and insurance, and limitations on liens, dispositions, issuance of debt, investments, payment of dividends, repurchases of capital stock, acquisitions, transactions with affiliates, and capital expenditures. There also are financial covenants that require the company to maintain a maximum leverage ratio (consolidated funded indebtedness to consolidated EBITDA, each as defined in the New Credit Agreement) of no greater than 3.50 to 1, and a minimum interest coverage ratio (consolidated EBITDA to consolidated interest charges, each as defined in the New Credit Agreement) of no less than 3.50 to 1. As of December 31, 2010, the company’s leverage ratio was 1.89 and the company’s interest coverage ratio was 8.40 and the company was in compliance with all covenant requirements. Under the most restrictive covenant of the company’s borrowing arrangements as of December 31, 2010, the company had the capacity to borrow up to an additional $215,068,000.
While there is general concern about the potential for rising interest rates, the company believes that its exposure to interest rate fluctuations is manageable given that portions of the company’s debt are at fixed rates for extended periods of time, the company has the ability to utilize swaps to exchange variable rate debt to fixed rate debt, if needed, and the company’s free cash flow should allow it to absorb any modest rate increases in the months ahead without any material impact on its liquidity or capital resources. As of December 31, 2010, the weighted average floating interest rate on borrowings was 3.29%.
As is the case for many companies operating in the current economic environment, the company is exposed to a number of risks. These risks include the possibility that: one or more of the lenders participating in the company’s revolving credit facility may be unable or unwilling to extend credit to the company; the third party company that provides lease financing to the company’s customers may refuse or be unable to fulfill its financing obligations or extend credit to the company’s customers; one or more customers of the company may be unable to pay for purchases of the company’s products on a timely basis; one or more key suppliers may be unable or unwilling to provide critical goods or services to the company; and one or more of the counterparties to the company’s hedging arrangements may be unable to fulfill its obligations to the company. Although the company has taken actions in an effort to mitigate these risks, during periods of economic downturn, the company’s exposure to these risks increases. Events of this nature may adversely affect the company’s liquidity or sales and revenues, and therefore have an adverse effect on the company’s business and results of operations.
CAPITAL EXPENDITURES
There are no individually material capital expenditure commitments outstanding as of December 31, 2010. The company estimates that capital investments for 2011 could approximate between $25,000,000 and $30,000,000, compared to actual capital expenditures of $17,353,000 in 2010. The company believes that its balances of cash and cash equivalents, together with funds generated from operations and existing borrowing facilities, will be sufficient to meet its operating cash requirements and fund required capital expenditures for the foreseeable future.
CASH FLOWS
Cash flows provided by operating activities were $122,207,000 in 2010, compared to $155,663,000 in the previous year. The decline in operating cash flows in 2010 was primarily attributable to an increase in net working capital assets specifically trade receivables and inventories. Trade receivables increased due to strong fourth quarter 2010 sales.
Cash flows used for investing activities were $30,617,000 in 2010, compared to $16,682,000 in 2009. The increase in cash used was primarily attributable to an acquisition in the amount of $13,725,000 in the NA/HME segment in 2010.
I-46
Table of Contents
Cash flows required by financing activities in 2010 were $77,634,000, compared to cash flows required of $153,290,000 in 2009. The decrease in cash used was primarily attributable to the company paying down less debt in 2010 as compared to 2009. This was partially offset by increase in the use of cash for the payment of debt financing costs related to the company’s early extinguishment of debt and refinancing of $30,329,000.
During 2010, the company generated free cash flow of $104,890,000 compared to free cash flow of $141,598,000 in 2009. The decrease is due primarily to an increase in net working capital assets as noted above. Free cash flow is a non-GAAP financial measure that is comprised of net cash provided by operating activities, excluding net cash impact related to restructuring activities, less net purchases of property and equipment, net of proceeds from sales of property and equipment. Management believes that this financial measure provides meaningful information for evaluating the overall financial performance of the company and its ability to repay debt or make future investments (including acquisitions, etc.).
The non-GAAP financial measure is reconciled to the GAAP measure as follows (in thousands):
| Twelve Months Ended December 31, |
||||||||
| 2010 | 2009 | |||||||
| Net cash provided by operating activities |
$ | 122,207 | $ | 155,663 | ||||
| Plus: Net cash impact related to restructuring activities |
— | 2,771 | ||||||
| Less: Purchases of property and equipment—net |
(17,317 | ) | (16,836 | ) | ||||
| Free Cash Flow |
$ | 104,890 | $ | 141,598 | ||||
CONTRACTUAL OBLIGATIONS
The company’s contractual obligations as of December 31, 2010 are as follows (in thousands):
| Payments due by period | ||||||||||||||||||||
| Total | Less than 1 year |
1-3 years | 3-5 years | More than 5 years |
||||||||||||||||
| 4.125% Convertible Senior Subordinated Debentures due 2027 |
$ | 128,552 | $ | 3,185 | $ | 6,369 | $ | 6,369 | $ | 112,629 | ||||||||||
| Revolving Credit Agreements due 2015 |
184,932 | 6,950 | — | 177,982 | — | |||||||||||||||
| Operating lease obligations |
57,750 | 19,374 | 21,433 | 9,357 | 7,586 | |||||||||||||||
| Capital lease obligations |
12,090 | 1,575 | 2,850 | 2,727 | 4,938 | |||||||||||||||
| Purchase obligations (primarily computer systems contracts) |
9,232 | 3,676 | 3,567 | 1,989 | — | |||||||||||||||
| Product liability |
24,160 | 4,134 | 9,414 | 4,558 | 6,054 | |||||||||||||||
| SERP |
26,524 | 391 | 2,030 | 2,302 | 21,801 | |||||||||||||||
| Other, principally deferred compensation |
9,308 | 66 | 928 | 288 | 8,026 | |||||||||||||||
| Total |
$ | 452,548 | $ | 39,351 | $ | 46,591 | $ | 205,572 | $ | 161,034 | ||||||||||
“Other” includes an estimated payment of $700,000 in years 1-3 for liabilities recorded for uncertain tax positions. The table does not include any other payments related to liabilities recorded for uncertain tax positions as the company cannot make a reasonably reliable estimate as to any other payments. See Income Taxes in the Notes to the Consolidated Financial Statements included in this report.
I-47
Table of Contents
DIVIDEND POLICY
It is the company’s policy to pay a nominal dividend in order for its stock to be more attractive to a broader range of investors. The current annual dividend rate remains at $0.05 per Common Share and $0.045 per Class B Common Share. It is not anticipated that this will change materially as the company continues to have available significant growth opportunities through internal development and acquisitions. For 2010, annualized dividends of $0.05 per Common Share and $0.045 per Class B Common Share were declared and paid.
CRITICAL ACCOUNTING POLICIES
The Consolidated Financial Statements included in the report include accounts of the company and all majority-owned subsidiaries. The preparation of financial statements in conformity with accounting principles generally accepted in the United States requires management to make estimates and assumptions in certain circumstances that affect amounts reported in the accompanying Consolidated Financial Statements and related footnotes. In preparing the financial statements, management has made its best estimates and judgments of certain amounts included in the financial statements, giving due consideration to materiality. However, application of these accounting policies involves the exercise of judgment and use of assumptions as to future uncertainties and, as a result, actual results could differ from these estimates.
The following critical accounting policies, among others, affect the more significant judgments and estimates used in preparation of the company’s consolidated financial statements.
Revenue Recognition
Invacare’s revenues are recognized when products are shipped to unaffiliated customers. Revenue Recognition, ASC 605, provides guidance on the application of generally accepted accounting principles to selected revenue recognition issues. The company has concluded that its revenue recognition policy is appropriate and in accordance with GAAP and ASC 605. Shipping and handling costs are included in cost of goods sold.
Sales are made only to customers with whom the company believes collection is reasonably assured based upon a credit analysis, which may include obtaining a credit application, a signed security agreement, personal guarantee and/or a cross corporate guarantee depending on the credit history of the customer. Credit lines are established for new customers after an evaluation of their credit report and/or other relevant financial information. Existing credit lines are regularly reviewed and adjusted with consideration given to any outstanding past due amounts.
The company offers discounts and rebates, which are accounted for as reductions to revenue in the period in which the sale is recognized. Discounts offered include: cash discounts for prompt payment, base and trade discounts based on contract level for specific classes of customers. Volume discounts and rebates are given based on large purchases and the achievement of certain sales volumes. Product returns are accounted for as a reduction to reported sales with estimates recorded for anticipated returns at the time of sale. The company does not ship any goods on consignment.
Distributed products sold by the company are accounted for in accordance with the revenue recognition guidance in ASC 605-45-05. The company records distributed product sales gross as a principal since the company takes title to the products and has the risks of loss for collections, delivery and returns.
Product sales that give rise to installment receivables are recorded at the time of sale when the risks and rewards of ownership are transferred. In December 2000, the company entered into an agreement with De Lage Landen, Inc. (“DLL”), a third party financing company, to provide the majority of future lease financing to
I-48
Table of Contents
Invacare customers. As such, interest income is recognized based on the terms of the installment agreements. Installment accounts are monitored and if a customer defaults on payments, interest income is no longer recognized. All installment accounts are accounted for using the same methodology, regardless of duration of the installment agreements.
Allowance for Uncollectible Accounts Receivable
The estimated allowance for uncollectible amounts is based primarily on management’s evaluation of the financial condition of the customer. In addition, as a result of the third party financing arrangement, management monitors the collection status of these contracts in accordance with the company’s limited recourse obligations and provides amounts necessary for estimated losses in the allowance for doubtful accounts and establishing reserves for specific customers as needed.
The company continues to closely monitor the credit-worthiness of its customers and adhere to tight credit policies. During the second quarter of 2010, the Centers for Medicare and Medicaid Services announced the single payment amounts for Round 1 of the Competitive Bidding Program which commenced on January 1, 2011 in nine metropolitan statistical areas (MSAs). The single payment amounts will be used to determine the price that Medicare pays for certain durable medical equipment, prosthetics, orthotics and supplies. The program replaces Medicare’s existing fee schedule amounts with market-based prices. The company believes the changes announced could have a significant impact on the collectability of accounts receivable for those customers which are in the MSA locations impacted and which have a portion of their revenues tied to Medicare reimbursement. As a result, this is an additional risk factor which the company considers when assessing the collectability of accounts receivable.
Invacare has an agreement with DLL, a third party financing company, to provide the majority of future lease financing to Invacare’s North America customers. The DLL agreement provides for direct leasing between DLL and the Invacare customer. The company retains a recourse obligation for events of default under the contracts. The company monitors the collections status of these contracts and has provided amounts for estimated losses in its allowances for doubtful accounts.
Inventories and Related Allowance for Obsolete and Excess Inventory
Inventories are stated at the lower of cost or market with cost determined by the first-in, first-out method. Inventories have been reduced by an allowance for excess and obsolete inventories. The estimated allowance is based on management’s review of inventories on hand compared to estimated future usage and sales. A provision for excess and obsolete inventory is recorded as needed based upon the discontinuation of products, redesigning of existing products, new product introductions, market changes and safety issues. Both raw materials and finished goods are reserved for on the balance sheet.
In general, Invacare reviews inventory turns as an indicator of obsolescence or slow moving product as well as the impact of new product introductions. Depending on the situation, the company may partially or fully reserve for the individual item. The company continues to increase its overseas sourcing efforts, increase its emphasis on the development and introduction of new products, and decrease the cycle time to bring new product offerings to market. These initiatives are sources of inventory obsolescence for both raw material and finished goods.
Goodwill, Intangible and Other Long-Lived Assets
Property, equipment, certain intangibles and certain other long-lived assets are amortized over their useful lives. Useful lives are based on management’s estimates of the period that the assets will generate revenue. Under Intangibles—Goodwill and Other, ASC 350, goodwill and intangible assets deemed to have indefinite lives are subject to annual impairment tests. Furthermore, goodwill and other long-lived assets are reviewed for
I-49
Table of Contents
impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. The company completes its annual impairment tests in the fourth quarter of each year. The discount rates used have a significant impact upon the discounted cash flow methodology utilized in the company’s annual impairment testing as higher discount rates decrease the fair value estimates.
The company utilizes a discounted cash flow method model to analyze reporting units for impairment in which the company forecasts income statement and balance sheet amounts based on assumptions regarding future sales growth, profitability, inventory turns, days’ sales outstanding, etc. to forecast future cash flows. The cash flows are discounted using a weighted average cost of capital discount rate where the cost of debt is based on quoted rates for 20-year debt of companies of similar credit risk and the cost of equity is based upon the 20-year treasury rate for the risk free rate, a market risk premium, the industry average beta and a small cap stock adjustment. The assumptions used are based on a market participant’s point of view and yielded a discount rate of 9.59% in 2010 compared to 10.74% in 2009 and 8.90% to 9.90% in 2008.
The company also utilizes an EV (Enterprise Value) to EBITDA (Earnings Before Interest, Taxes, Depreciation and Amortization) Method to compute the fair value of its reporting units which considers potential acquirers and their EV to EBITDA multiples adjusted by an estimated premium. While more weight is given to the discounted cash flow method, the EV to EBITDA Method does provide corroborative evidence of the reasonableness of the discounted cash flow method results.
While there was no indication of impairment in 2010 related to goodwill, a future potential impairment is possible for any of the company’s reporting units should actual results differ materially from forecasted results used in the valuation analysis. Furthermore, the company’s annual valuation of goodwill can differ materially if the market inputs used to determine the discount rate change significantly. For instance, higher interest rates or greater stock price volatility would increase the discount rate and thus increase the chance of impairment. For example, if the discount rate used were 100 basis points higher for the 2010 impairment analysis, there still would not be any indicator of potential impairment for any of the reporting units.
Product Liability
The company’s captive insurance company, Invatection Insurance Co., currently has a policy year that runs from September 1 to August 31 and insures annual policy losses of $10,000,000 per occurrence and $13,000,000 in the aggregate of the company’s North American product liability exposure. The company also has additional layers of external insurance coverage insuring up to $75,000,000 in annual aggregate losses arising from individual claims anywhere in the world that exceed the captive insurance company policy limits or the limits of the company’s per country foreign liability limits, as applicable. There can be no assurance that Invacare’s current insurance levels will continue to be adequate or available at affordable rates.
Product liability reserves are recorded for individual claims based upon historical experience, industry expertise and indications from the third-party actuary. Additional reserves, in excess of the specific individual case reserves, are provided for incurred but not reported claims based upon third-party actuarial valuations at the time such valuations are conducted. Historical claims experience and other assumptions are taken into consideration by the third-party actuary to estimate the ultimate reserves. For example, the actuarial analysis assumes that historical loss experience is an indicator of future experience, that the distribution of exposures by geographic area and nature of operations for ongoing operations is expected to be very similar to historical operations with no dramatic changes and that the government indices used to trend losses and exposures are appropriate.
Estimates made are adjusted on a regular basis and can be impacted by actual loss awards and settlements on claims. While actuarial analysis is used to help determine adequate reserves, the company is responsible for the determination and recording of adequate reserves in accordance with accepted loss reserving standards and practices.
I-50
Table of Contents
Warranty
Generally, the company’s products are covered from the date of sale to the customer by warranties against defects in material and workmanship for various periods depending on the product. Certain components carry a lifetime warranty. A provision for estimated warranty cost is recorded at the time of sale based upon actual experience. The company continuously assesses the adequacy of its product warranty accrual and makes adjustments as needed. Historical analysis is primarily used to determine the company’s warranty reserves. Claims history is reviewed and provisions are adjusted as needed. However, the company does consider other events, such as a product recall, which could warrant additional warranty reserve provision. No material adjustments to warranty reserves were necessary in the current year. See Warranty Costs in the Notes to the Condensed Consolidated Financial Statements included in this report for a reconciliation of the changes in the warranty accrual.
Accounting for Stock-Based Compensation
The company accounts for share based compensation under the provisions of Compensation—Stock Compensation, ASC 718. The company has not made any modifications to the terms of any previously granted options and no changes have been made regarding the valuation methodologies or assumptions used to determine the fair value of options granted since 2005 and the company continues to use a Black-Scholes valuation model. As of December 31, 2010, there was $15,539,000 of total unrecognized compensation cost from stock-based compensation arrangements granted under the 2003 Plan, which is related to non-vested options and shares, and includes $5,190,000 related to restricted stock awards. The company expects the compensation expense to be recognized over a four-year period for a weighted-average period of approximately two years.
The substantial majority of the options awarded have been granted at exercise prices equal to the market value of the underlying stock on the date of grant. Restricted stock awards granted without cost to the recipients are expensed on a straight-line basis over the vesting periods.
Income Taxes
As part of the process of preparing its financial statements, the company is required to estimate income taxes in various jurisdictions. The process requires estimating the company’s current tax exposure, including assessing the risks associated with tax audits, as well as estimating temporary differences due to the different treatment of items for tax and accounting policies. The temporary differences are reported as deferred tax assets and or liabilities. Substantially all of the company’s U.S. deferred tax assets are offset by a valuation allowance. The company also must estimate the likelihood that its deferred tax assets will be recovered from future taxable income and whether or not valuation allowances should be established. In the event that actual results differ from its estimates, the company’s provision for income taxes could be materially impacted. The company does not believe that there is a substantial likelihood that materially different amounts would be reported related to its critical accounting policies.
RECENTLY ISSUED ACCOUNTING PRONOUNCEMENTS
On January 21, 2010, the Financial Accounting Standards Board (FASB) issued Accounting Standards Update No. 2010-06, Improving Disclosures about Fair Value Measurements (ASU 2010-06 or the ASU). The ASU amends ASC 820 to require a number of additional disclosures regarding fair value measurements. The amended guidance requires entities to disclose additional information regarding assets and liabilities that are transferred between levels of the fair value hierarchy. Entities are also required to disclose information in the Level 3 roll forward about purchases, sales, issuances and settlements on a gross basis. In addition to these new disclosure requirements, ASU 2010-06 also amends Topic 820 to further clarify existing guidance pertaining to the level of disaggregation at which fair value disclosures should be made and the requirements to disclose
I-51
Table of Contents
information about the valuation techniques and inputs used in estimating Level 2 and Level 3 fair value measurements. The company adopted ASU 2010-06 effective January 1, 2010 and the standard was utilized in preparing the fair value measurement disclosures.
On July 21, 2010, the FASB issued Accounting Standards Update No. 2010-20, Disclosures about the Credit Quality of Financing Receivables and the Allowance for Credit Losses (ASU 2010-20). ASU 2010-20 requires entities to provide additional disclosures regarding credit-risk exposures, including how credit risk is analyzed and assessed, and allowances for credit losses, including reasons for changes each period. The company adopted ASU 2010-20 effective December 31, 2010 and the company’s receivable disclosures in this 2010 Form 10-K reflect the required disclosures. The adoption of ASU 2010-20 did not have any material impact on the company’s financial position, results of operations or cash flows.
| Item 7A. | Quantitative and Qualitative Disclosure about Market Risk. |
The company is exposed to market risk through various financial instruments, including fixed rate and floating rate debt instruments. The company does at times use interest swap agreements to mitigate its exposure to interest rate fluctuations. Based on December 31, 2010 debt levels, a 1% change in interest rates would impact annual interest expense by approximately $1,849,000. Additionally, the company operates internationally and, as a result, is exposed to foreign currency fluctuations. Specifically, the exposure results from intercompany loans, intercompany sales or payments and third party sales or payments. In an attempt to reduce this exposure, foreign currency forward contracts are utilized to hedge intercompany purchases and sales as well as third party purchases and sales. The company does not believe that any potential loss related to these financial instruments would have a material adverse effect on the company’s financial condition or results of operations.
On October 28, 2010, the company entered into the New Credit Agreement which provides for a $400,000,000 senior secured revolving credit facility maturing in October 2015 at variable rates. As of December 31, 2010, the company had outstanding $77,201,000 in principal amount of 4.125% Convertible Senior Subordinated Debentures due in February 2027, of which $25,137,000 is included in equity. Accordingly, while the company is exposed to increases in interest rates, its exposure to the volatility of the current market environment is limited as the company does not currently need to re-finance any of its debt. However, the company’s New Credit Agreement contains covenants with respect to, among other items, consolidated funded indebtedness to consolidated earnings before interest, taxes, depreciation and amortization (EBITDA) and interest coverage, as defined in the agreement. The company is in compliance with all covenant requirements, but should it fall out of compliance with these requirements, the company would have to attempt to obtain alternative financing and thus likely be required to pay much higher interest rates.
| Item 8. | Financial Statements and Supplementary Data. |
Reference is made to the Report of Independent Registered Public Accounting Firm, Consolidated Balance Sheets, Consolidated Statement of Earnings, Consolidated Statement of Cash Flows, Consolidated Statement of Shareholders’ Equity, Notes to Consolidated Financial Statements and Financial Statement Schedule, which appear on pages FS-1 to FS-50 of this Annual Report on Form 10-K.
| Item 9. | Changes in and Disagreements with Accountants on Accounting and Financial Disclosure. |
None.
| Item 9A. | Controls and Procedures. |
(a) Evaluation of Disclosure Controls and Procedures
As of December 31, 2010, an evaluation was performed, under the supervision and with the participation of the company’s management, including the Chief Executive Officer and Chief Financial Officer, of the effectiveness of the design and operation of the company’s disclosure controls and procedures (as defined in
I-52
Table of Contents
Exchange Act Rules 13a-15(e) and 15d-15(e)). Based on that evaluation, the company’s management, including the Chief Executive Officer and Chief Financial Officer, concluded that the company’s disclosure controls and procedures were effective as of December 31, 2010, in ensuring that information required to be disclosed by the company in the reports it files and submits under the Exchange Act is (1) recorded, processed, summarized and reported, within the time periods specified in the Commission’s rules and forms and (2) accumulated and communicated to the company’s management, including the Chief Executive Officer and the Chief Financial Officer, as appropriate to allow for timely decisions regarding required disclosure.
(b) Management’s Annual Report on Internal Control Over Financial Reporting
Management is responsible for establishing and maintaining a system of adequate internal control over financial reporting that provides reasonable assurance that assets are safeguarded and that transactions are authorized, recorded and reported properly. The system includes self-monitoring mechanisms; regular testing by the company’s internal auditors; a Code of Conduct; written policies and procedures; and a careful selection and training of employees. Actions are taken to correct deficiencies as they are identified. An effective internal control system, no matter how well designed, has inherent limitations—including the possibility of the circumvention or overriding of controls—and therefore can provide only reasonable assurance that errors and fraud that can be material to the financial statements are prevented or would be detected on a timely basis. Further, because of changes in conditions, internal control system effectiveness may vary over time.
Management’s assessment of the effectiveness of the company’s internal control over financial reporting is based on the Internal Control—Integrated Framework published by the Committee of Sponsoring Organizations of the Treadway Commission.
In management’s opinion, internal control over financial reporting is effective as of December 31, 2010.
(c) Attestation Report of the Registered Public Accounting Firm
The company’s independent registered public accounting firm, Ernst & Young LLP, audited the company’s internal control over financial reporting and, based on that audit, issued an attestation report regarding the company’s internal control over financial reporting, which is included in this Annual Report on Form 10-K on page FS-2.
(d) Changes in Internal Control Over Financial Reporting
There have been no changes in the company’s internal control over financial reporting that occurred during the company’s last fiscal quarter that have materially affected, or are reasonably likely to materially affect, the company’s internal control over financial reporting.
| Item 9B. | Other Information. |
None.
I-53
Table of Contents
PART III
| Item 10. | Directors and Executive Officers of the Registrant. |
Information required by Item 10 as to the executive officers of the company is included in Part I of this Annual Report on Form 10-K. The other information required by Item 10 as to the directors of the company, the Audit Committee, the audit committee financial expert, the procedures for recommending nominees to the Board of Directors, compliance with Section 16(a) of the Exchange Act and corporate governance is incorporated herein by reference to the information set forth under the captions “Election of Directors,” “Corporate Governance,” and “Section 16(a) Beneficial Ownership Compliance” in the company’s definitive Proxy Statement for the 2011 Annual Meeting of Shareholders.
| Item 11. | Executive Compensation. |
The information required by Item 11 is incorporated by reference to the information set forth under the captions “Executive Compensation” and “Corporate Governance” in the company’s definitive Proxy Statement for the 2011 Annual Meeting of Shareholders.
| Item 12. | Security Ownership of Certain Beneficial Owners and Management. |
The information required by Item 12 is incorporated by reference to the information set forth under the caption “Share Ownership of Principal Holders and Management” in the company’s definitive Proxy Statement for the 2011 Annual Meeting of Shareholders.
Information regarding the securities authorized for issuance under the company’s equity compensation plans is incorporated by reference to the information set forth under the captions “Compensation of Executive Officers” and “Compensation of Directors” in the company’s definitive Proxy Statement for the 2011 Annual Meeting of Shareholders.
| Item 13. | Certain Relationships and Related Transactions. |
The information required by Item 13 is incorporated by reference to the information set forth under the caption “Certain Relationships and Related Transactions” in the company’s definitive Proxy Statement for the 2011 Annual Meeting of Shareholders.
| Item 14. | Principal Accounting Fees and Services. |
The information required by Item 14 is incorporated by reference to the information set forth under the caption “Independent Auditors” and “Pre-Approval Policies and Procedures” in the company’s definitive Proxy Statement for the 2011 Annual Meeting of Shareholders.
I-54
Table of Contents
PART IV
| Item 15. | Exhibits and Financial Statement Schedules. |
(a)(1) Financial Statements.
The following financial statements of the company are included in Part II, Item 8:
Consolidated Statement of Earnings—years ended December 31, 2010, 2009 and 2008
Consolidated Balance Sheet—December 31, 2010 and 2009
Consolidated Statement of Cash Flows—years ended December 31, 2010, 2009 and 2008
Consolidated Statement of Shareholders’ Equity—years ended December 31, 2010, 2009 and 2008
Notes to Consolidated Financial Statements
(a)(2) Financial Statement Schedules.
The following financial statement schedule of the company is included in Part II, Item 8:
Schedule II—Valuation and Qualifying Accounts
All other schedules have been omitted because they are not applicable or not required, or because the required information is included in the Consolidated Financial Statements or notes thereto.
(a)(3) Exhibits.
See Exhibit Index at page number I-57 of this Report on Form 10-K.
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this Report to be signed on its behalf by the undersigned, thereunto duly authorized as of February 25, 2011.
| INVACARE CORPORATION | ||
| By: | /s/ GERALD B. BLOUCH | |
| Gerald B. Blouch | ||
| President and Chief Executive Officer | ||
I-55
Table of Contents
Pursuant to the requirements of the Securities Exchange Act of 1934, this Report has been signed below by the following persons on behalf of the Registrant and in the capacities indicated as of February 25, 2011.
| Signature |
Title | |
| /s/ A. MALACHI MIXON, III A. Malachi Mixon, III |
Chairman of the Board of Directors | |
| /s/ GERALD B. BLOUCH Gerald B. Blouch |
President and Chief Executive Officer and Director (Principal Executive Officer) | |
| /s/ ROBERT K. GUDBRANSON Robert K. Gudbranson |
Senior Vice President, Chief Financial Officer (Principal Financial and Accounting Officer) | |
| /s/ JAMES C. BOLAND James C. Boland |
Director | |
| /s/ MICHAEL F. DELANEY Michael F. Delaney |
Director | |
| /s/ C. MARTIN HARRIS, M.D. C. Martin Harris, M.D. |
Director | |
| /s/ JAMES L. JONES James L. Jones |
Director | |
| /s/ DALE C. LAPORTE Dale C. LaPorte |
Director | |
| /s/ DAN T. MOORE, III Dan T. Moore, III |
Director | |
| /s/ JOSEPH B. RICHEY, II Joseph B. Richey, II |
President—Invacare Technologies, Senior Vice President—Electronics and Design Engineering and Director | |
| /s/ CHARLES S. ROBB Charles S. Robb |
Director | |
| /s/ WILLIAM M. WEBER William M. Weber |
Director | |
I-56
Table of Contents
INVACARE CORPORATION
Report on Form 10-K for the fiscal year ended December 31, 2010.
Exhibit Index
| Official Exhibit No. |
Description |
Sequential Page No. |
||||
| 2.1 | Sale and Purchase Agreement Regarding the Sale and Purchase of All Shares in WP Domus GmbH by and among WP Domus LLC, Mr. Peter Schultz and Mr. Wilhelm Kaiser, Invacare GmbH & Co. KG and Invacare Corporation dated as of July 31, 2004 | (A | ) | |||
| 2.2 | Guarantee Letter Agreement of Warburg, Pincus Ventures, L.P. and Warburg, Pincus International, L.P. dated as of September 9, 2004 | (A | ) | |||
| 3(a) | Second Amended and Restated Articles of Incorporation | (L | ) | |||
| 3(b) | Code of Regulations, as amended on May 21, 2009 | (N | ) | |||
| 3(c) | Amendment to Code of Regulations, adopted May 20, 2010 | (S | ) | |||
| 4(a) | Specimen Share Certificate for Common Shares | (G | ) | |||
| 4(b) | Specimen Share Certificate for Class B Common Shares | (G | ) | |||
| 4(c) | Rights agreement between Invacare Corporation and National City Bank (as predecessor in interest to Wells Fargo Bank, N.A.) dated as of July 8, 2005 | (F | ) | |||
| 4(d) | Indenture, dated as of February 12, 2007, by and among Invacare Corporation, the Guarantors named therein and Wells Fargo Bank, N.A., as trustee (including the Form of 4.125% Convertible Senior Subordinated Debenture due 2027 and related Guarantee attached as Exhibit A) | (I | ) | |||
| 4(f) | Amendment No. 1 to Rights agreement between Invacare Corporation and Wells Fargo Bank, N.A. dated as of October 28, 2009 | (O | ) | |||
| 10(a) | 1992 Non-Employee Directors Stock Option Plan adopted in May 1992 | (E | ) | |||
| 10(b) | Deferred Compensation Plan for Non-Employee Directors, adopted in May 1992 | (E | ) | |||
| 10(c) | Invacare Corporation 1994 Performance Plan approved January 28, 1994 | (E | )* | |||
| 10(d) | Amendment No. 1 to the Invacare Corporation 1994 Performance Plan approved May 28, 1998 | (E | )* | |||
| 10(e) | Amendment No. 2 to the Invacare Corporation 1994 Performance Plan approved May 24, 2000 | (B | )* | |||
| 10(f) | Amendment No. 3 to the Invacare Corporation 1994 Performance Plan approved March 13, 2003 | (C | )* | |||
| 10(g) | Invacare Retirement Savings Plan, effective January 1, 2001, as amended | (J | )* | |||
| 10(h) | Agreement entered into by and between the company and its Chief Financial Officer | (D | )* | |||
| 10(i) | Invacare Corporation 401(K) Plus Benefit Equalization Plan, effective January 1, 2003, as amended and restated | (J | )* | |||
| 10(j) | Invacare Corporation Amended and Restated 2003 Performance Plan | (M | )* | |||
| 10(k)** | Form of Change of Control Agreement entered into by and between the company and certain of its executive officers and schedule of all such agreements with current executive officers | * | ||||
I-57
Table of Contents
| Official Exhibit No. |
Description |
Sequential Page No. |
||||
| 10(l)** | Form of Indemnity Agreement entered into by and between the company and its directors and certain of its executive officers and schedule of all such agreements with directors and executive officers | * | ||||
| 10(m)** | Invacare Corporation Deferred Compensation Plus Plan, effective January 1, 2005, as amended August 19, 2009 and on November 23, 2010 | * | ||||
| 10(n) | Invacare Corporation Death Benefit Only Plan, effective January 1, 2005, as amended | (J | )* | |||
| 10(o) | Supplemental Executive Retirement Plan, as amended and restated effective February 1, 2000 | (E | )* | |||
| 10(p) | Form of Director Stock Option Award under Invacare Corporation 1994 Performance Plan | (E | )* | |||
| 10(q) | Form of Director Stock Option Award under Invacare Corporation 2003 Performance Plan | (J | )* | |||
| 10(r)** | Form of Director Deferred Option Award under Invacare Corporation 2003 Performance Plan | * | ||||
| 10(s) | Form of Restricted Stock Option Award under Invacare Corporation 2003 Performance Plan | (J | )* | |||
| 10(t) | Form of Stock Option Award under Invacare Corporation 2003 Performance Plan | (J | )* | |||
| 10(u) | Form of Executive Stock Option Award under Invacare Corporation 2003 Performance Plan | (J | )* | |||
| 10(v) | Form of Switzerland Stock Option Award under Invacare Corporation 2003 Performance Plan | (J | )* | |||
| 10(w) | Form of Switzerland Executive Stock Option Award under Invacare Corporation 2003 Performance Plan | (J | )* | |||
| 10(x)** | Director Compensation Schedule | |||||
| 10(y) | Invacare Corporation Executive Incentive Bonus Plan, as amended March 9, 2010 | (Q | )* | |||
| 10(aa) | Purchase Agreement by and among Invacare Corporation, the Subsidiary Guarantors named therein, and the Initial Purchasers named therein dated as of February 5, 2007 | (H | ) | |||
| 10(ab)** | Form of Rule 10b5-1 Sales Plan entered into between the company and certain of its executive officers and other employees and a schedule of all such agreements with executive officers and other employees | |||||
| 10(ac) | A. Malachi Mixon, III Retirement Benefit Agreement | (J | )* | |||
| 10(ad) | Cash Balance Supplemental Executive Retirement Plan, as amended and restated, effective December 31, 2008 | (K | )* | |||
| 10(ae) | Form of Participation Agreement, for current participants in the Cash Balance Supplemental Executive Retirement Plan, as of December 31, 2008, entered into by and between the company and certain participants and a schedule of all such agreements with participants | (K | )* | |||
| 10(af) | Amended and Restated Severance Protection Agreement, between the company and Gerald B. Blouch, effective December 31, 2008 | (K | )* | |||
I-58
Table of Contents
| Official Exhibit No. |
Description |
Sequential Page No. |
||||
| 10(ag) | Amendment No. 1 to the Cash Balance Supplemental Executive Retirement Plan, effective August 19, 2009 | (P | )* | |||
| 10(ah) | $400,000,000 Revolving Credit Facility Credit Agreement by and among Invacare Corporation, the other borrowers, guarantors and lenders thereto; PNC Bank, National Association, as Administrative Agent; Keybank National Association and Bank of America, N.A. as Co-Syndication Agents; and RBS Citizens, N.A. as Documentation Agent. | (R | )* | |||
| 21** | Subsidiaries of the company | |||||
| 23** | Consent of Independent Registered Public Accounting Firm | |||||
| 31.1** | Certification of the Chief Executive Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 | |||||
| 31.2** | Certification of the Chief Financial Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 | |||||
| 32.1** | Certification of the Chief Executive Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 | |||||
| 32.2** | Certification of the Chief Financial Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 | |||||
| * | Management contract, compensatory plan or arrangement |
| ** | Filed herewith |
| (A) | Reference is made to the appropriate Exhibit to the company report on Form 8-K, dated September 9, 2004, which Exhibit is incorporated herein by reference. | |
| (B) | Reference is made to the appropriate Exhibit of the company report on Form S-8, dated March 30, 2001, which Exhibit is incorporated herein by reference. | |
| (C) | Reference is made to the appropriate Exhibit of the company report on Form 10-Q for the quarter ended March 31, 2003, which Exhibit is incorporated herein by reference. | |
| (D) | Reference is made to Exhibit 10.1 of the company report on Form 8-K, dated March 6, 2008. | |
| (E) | Reference is made to the appropriate Exhibit of the company report on Form 10-K for the fiscal year ended December 31, 2004, which Exhibit is incorporated herein by reference. | |
| (F) | Reference is made to the appropriate Exhibit of the company report on Form 8-K, dated July 8, 2005, which is incorporated herein by reference. | |
| (G) | Reference is made to the appropriate Exhibit of the company report on Form 10-K for the fiscal year ended December 31, 2005, which Exhibit is incorporated herein by reference. | |
| (H) | Reference is made to the appropriate Exhibit of the company report on Form 8-K, dated February 5, 2007, which is incorporated herein by reference. | |
| (I) | Reference is made to the appropriate Exhibit of the company report on Form 8-K, dated February 12, 2007, which is incorporated herein by reference. | |
| (J) | Reference is made to the appropriate Exhibit of the company report on Form 10-K for the fiscal year ended December 31, 2007, which Exhibit is incorporated herein by reference. | |
| (K) | Reference is made to the appropriate Exhibit of the company report on Form 8-K, dated December 31, 2008, which is incorporated herein by reference. | |
I-59
Table of Contents
| (L) | Reference is made to the appropriate Exhibit of the company report on Form 10-K for the fiscal year ended December 31, 2008, which Exhibit is incorporated herein by reference. | |
| (M) | Reference is made to the appropriate Exhibit of the company report on Form 8-K, dated May 21, 2009, which is incorporated herein by reference. | |
| (N) | Reference is made to the appropriate Exhibit of the company report on Form 10-Q, dated June 30, 2009, which is incorporated herein by reference. | |
| (O) | Reference is made to the appropriate Exhibit of the company report on Form 8-A, dated October 30, 2009, which is incorporated herein by reference. | |
| (P) | Reference is made to the appropriate Exhibit of the company report on Form 10-Q, dated September 30, 2009, which is incorporated herein by reference. | |
| (Q) | Reference is made to the appropriate Exhibit to Appendix A of the company Definitive Proxy Statement on Schedule 14A, dated April 8, 2005, which is incorporated herein by reference. | |
| (R) | Reference is made to the appropriate Exhibit of the company report on Form 8-K, dated October 28, 2010, which is incorporated herein by reference. | |
| (S) | Reference is made to Appendix A to the company’s definitive proxy statement on Schedule 14A dated April 7, 2010, which is incorporated herein by reference. | |
I-60
Table of Contents
Report of Independent Registered Public Accounting Firm
The Board of Directors and Shareholders
Invacare Corporation
We have audited the accompanying consolidated balance sheets of Invacare Corporation and subsidiaries as of December 31, 2010 and 2009, and the related consolidated statements of earnings, cash flows and shareholders’ equity for each of the three years in the period ended December 31, 2010. Our audits also included the financial statement schedule listed in the Index at Item 15(a)(2). These financial statements and schedule are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements and schedule based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the consolidated financial position of Invacare Corporation and subsidiaries at December 31, 2010 and 2009, and the consolidated results of their operations and their cash flows for each of the three years in the period ended December 31, 2010, in conformity with U. S. generally accepted accounting principles. Also, in our opinion, the related financial statement schedule, when considered in relation to the basic financial statements taken as a whole, presents fairly in all material respects the information set forth therein.
We have also audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), Invacare Corporation’s internal control over financial reporting as of December 31, 2010, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission and our report dated February 25, 2011 expressed an unqualified opinion thereon.
/s/ ERNST & YOUNG LLP
Cleveland, Ohio
February 25, 2011
FS-1
Table of Contents
Report of Independent Registered Public Accounting Firm
The Board of Directors and Shareholders
Invacare Corporation
We have audited Invacare Corporation’s internal control over financial reporting as of December 31, 2010, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (the COSO criteria). Invacare Corporation’s management is responsible for maintaining effective internal control over financial reporting, and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying “Management’s Report on Internal Control over Financial Reporting” which is included in Item 9A. Our responsibility is to express an opinion on the company’s internal control over financial reporting based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
In our opinion, Invacare Corporation maintained, in all material respects, effective internal control over financial reporting as of December 31, 2010, based on the COSO criteria.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), the consolidated balance sheets of Invacare Corporation and subsidiaries as of December 31, 2010 and 2009 and the related consolidated statements of earnings, cash flows and shareholders’ equity for each of the three years in the period ended December 31, 2010 of Invacare Corporation, and the financial statement schedule for the three years in the period ended December 31, 2010 and our report dated February 25, 2011 expressed an unqualified opinion thereon.
/s/ ERNST & YOUNG LLP
Cleveland, Ohio
February 25, 2011
FS-2
Table of Contents
CONSOLIDATED STATEMENT OF EARNINGS
INVACARE CORPORATION AND SUBSIDIARIES
| Years Ended December 31, | ||||||||||||
| 2010 | 2009 | 2008 | ||||||||||
| (In thousands, except per share data) | ||||||||||||
| Net sales |
$ | 1,722,081 | $ | 1,693,136 | $ | 1,755,694 | ||||||
| Cost of products sold |
1,212,440 | 1,199,942 | 1,266,802 | |||||||||
| Gross Profit |
509,641 | 493,194 | 488,892 | |||||||||
| Selling, general and administrative expenses |
411,513 | 398,646 | 398,254 | |||||||||
| Charges related to restructuring activities |
— | 4,506 | 2,949 | |||||||||
| Loss on debt extinguishment including debt finance charges and associated fees |
40,164 | 2,878 | — | |||||||||
| Asset write-downs to intangibles and investments |
— | 8,409 | — | |||||||||
| Interest expense |
20,647 | 33,150 | 42,927 | |||||||||
| Interest income |
(724 | ) | (1,674 | ) | (3,045 | ) | ||||||
| Earnings before Income Taxes |
38,041 | 47,279 | 47,807 | |||||||||
| Income taxes |
12,700 | 6,100 | 12,950 | |||||||||
| Net Earnings |
$ | 25,341 | $ | 41,179 | $ | 34,857 | ||||||
| Net Earnings per Share—Basic |
$ | 0.78 | $ | 1.29 | $ | 1.09 | ||||||
| Weighted Average Shares Outstanding—Basic |
32,393 | 31,969 | 31,902 | |||||||||
| Net Earnings per Share—Assuming Dilution |
$ | 0.78 | $ | 1.29 | $ | 1.09 | ||||||
| Weighted Average Shares Outstanding—Assuming Dilution |
32,694 | 31,996 | 31,953 | |||||||||
See notes to consolidated financial statements.
FS-3
Table of Contents
CONSOLIDATED BALANCE SHEETS
INVACARE CORPORATION AND SUBSIDIARIES
| December 31, 2010 |
December 31, 2009 |
|||||||
| (In thousands) | ||||||||
| Assets | ||||||||
| Current Assets |
||||||||
| Cash and cash equivalents |
$ | 48,462 | $ | 37,501 | ||||
| Trade receivables, net |
252,004 | 263,014 | ||||||
| Installment receivables, net |
3,959 | 3,565 | ||||||
| Inventories, net |
174,375 | 172,222 | ||||||
| Deferred income taxes |
5,778 | 390 | ||||||
| Other current assets |
41,581 | 51,772 | ||||||
| Total Current Assets |
526,159 | 528,464 | ||||||
| Other Assets |
45,484 | 48,006 | ||||||
| Other Intangibles |
70,911 | 85,305 | ||||||
| Property and Equipment, net |
130,763 | 141,633 | ||||||
| Goodwill |
507,083 | 556,093 | ||||||
| Total Assets |
$ | 1,280,400 | $ | 1,359,501 | ||||
| Liabilities and Shareholders’ Equity | ||||||||
| Current Liabilities |
||||||||
| Accounts payable |
$ | 143,753 | $ | 141,059 | ||||
| Accrued expenses |
130,079 | 142,293 | ||||||
| Accrued income taxes |
8,502 | 5,884 | ||||||
| Short-term debt and current maturities of long-term obligations |
7,974 | 1,091 | ||||||
| Total Current Liabilities |
290,308 | 290,327 | ||||||
| Long-Term Debt |
238,090 | 272,234 | ||||||
| Other Long-Term Obligations |
99,591 | 95,703 | ||||||
| Shareholders’ Equity |
||||||||
| Preferred Shares (Authorized 300 shares; none outstanding) |
— | — | ||||||
| Common Shares (Authorized 100,000 shares; 33,559 and 33,048 issued in 2010 and 2009, respectively)—no par |
8,401 | 8,273 | ||||||
| Class B Common Shares (Authorized 12,000 shares; 1,086 and 1,111, issued and outstanding in 2010 and 2009, respectively)—no par |
272 | 278 | ||||||
| Additional paid-in-capital |
231,685 | 229,272 | ||||||
| Retained earnings |
370,001 | 346,272 | ||||||
| Accumulated other comprehensive earnings |
112,631 | 174,204 | ||||||
| Treasury shares (2,319 and 1,834 shares in 2010 and 2009, respectively) |
(70,579 | ) | (57,062 | ) | ||||
| Total Shareholders’ Equity |
652,411 | 701,237 | ||||||
| Total Liabilities and Shareholders’ Equity |
$ | 1,280,400 | $ | 1,359,501 | ||||
See notes to consolidated financial statements.
FS-4
Table of Contents
CONSOLIDATED STATEMENT OF CASH FLOWS
INVACARE CORPORATION AND SUBSIDIARIES
| Years Ended December 31, | ||||||||||||
| 2010 | 2009 | 2008 | ||||||||||
| (In thousands) | ||||||||||||
| Operating Activities |
||||||||||||
| Net earnings |
$ | 25,341 | $ | 41,179 | $ | 34,857 | ||||||
| Adjustments to reconcile net earnings to net cash provided by operating activities: |
||||||||||||
| Depreciation and amortization |
36,804 | 40,562 | 43,744 | |||||||||
| Provision for losses on trade and installment receivables |
16,979 | 19,281 | 14,284 | |||||||||
| (Benefit) provision for deferred income taxes |
(2,467 | ) | 1,785 | 1,420 | ||||||||
| Provision for other deferred liabilities |
2,781 | 2,573 | 2,930 | |||||||||
| Provision for stock-based compensation |
6,135 | 4,495 | 3,299 | |||||||||
| Loss on disposals of property and equipment |
233 | 1,237 | 145 | |||||||||
| Loss on debt extinguishment including debt finance charges and associated fees |
40,164 | 2,878 | — | |||||||||
| Asset write-downs to intangibles and investments |
— | 8,409 | — | |||||||||
| Amortization of convertible debt discount |
3,198 | 4,142 | 3,694 | |||||||||
| Changes in operating assets and liabilities: |
||||||||||||
| Trade receivables |
(5,839 | ) | 6,452 | (15,031 | ) | |||||||
| Installment sales contracts, net |
(2,423 | ) | (3,356 | ) | (3,788 | ) | ||||||
| Inventories |
(6,352 | ) | 20,515 | (292 | ) | |||||||
| Other current assets |
3,181 | 11,628 | 4,754 | |||||||||
| Accounts payable |
5,534 | 12,532 | (20,440 | ) | ||||||||
| Accrued expenses |
(6,980 | ) | (18,012 | ) | 5,479 | |||||||
| Other long-term liabilities |
5,918 | (637 | ) | 1,359 | ||||||||
| Net Cash Provided by Operating Activities |
122,207 | 155,663 | 76,414 | |||||||||
| Investing Activities |
||||||||||||
| Purchases of property and equipment |
(17,353 | ) | (17,999 | ) | (19,957 | ) | ||||||
| Proceeds from sale of property and equipment |
36 | 1,163 | 211 | |||||||||
| Business acquisitions, net of cash acquired |
(13,725 | ) | — | (8,420 | ) | |||||||
| Decrease in other long-term assets |
801 | 601 | 4,882 | |||||||||
| Other |
(376 | ) | (447 | ) | 799 | |||||||
| Net Cash Used for Investing Activities |
(30,617 | ) | (16,682 | ) | (22,485 | ) | ||||||
| Financing Activities |
||||||||||||
| Proceeds from revolving lines of credit and long-term borrowings |
708,742 | 400,123 | 356,261 | |||||||||
| Payments on revolving lines of credit and long-term borrowings |
(751,660 | ) | (553,436 | ) | (417,182 | ) | ||||||
| Proceeds from exercise of stock options |
2,912 | 1,628 | 834 | |||||||||
| Payment of financing costs |
(30,329 | ) | — | — | ||||||||
| Payment of dividends |
(1,612 | ) | (1,605 | ) | (1,599 | ) | ||||||
| Purchase of treasury stock |
(5,687 | ) | — | — | ||||||||
| Net Cash Used by Financing Activities |
(77,634 | ) | (153,290 | ) | (61,686 | ) | ||||||
| Effect of exchange rate changes on cash |
(2,995 | ) | 4,294 | (6,927 | ) | |||||||
| Increase (decrease) in cash and cash equivalents |
10,961 | (10,015 | ) | (14,684 | ) | |||||||
| Cash and cash equivalents at beginning of year |
37,501 | 47,516 | 62,200 | |||||||||
| Cash and cash equivalents at end of year |
$ | 48,462 | $ | 37,501 | $ | 47,516 | ||||||
See notes to consolidated financial statements.
FS-5
Table of Contents
CONSOLIDATED STATEMENT OF SHAREHOLDERS’ EQUITY
INVACARE CORPORATION AND SUBSIDIARIES
| Common Stock |
Class B Stock |
Additional Paid-in- Capital |
Retained Earnings |
Accumulated Other Comprehensive Earnings |
Treasury Stock |
Total | ||||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||||||||||
| January 1, 2008 Balance |
$ | 8,034 | $ | 278 | $ | 206,307 | $ | 273,440 | $ | 164,969 | $ | (42,877 | ) | $ | 610,151 | |||||||||||||
| Exercise of stock options |
61 | 5,697 | (5,011 | ) | 747 | |||||||||||||||||||||||
| Non-qualified stock option expense |
1,961 | 1,961 | ||||||||||||||||||||||||||
| Restricted stock awards |
24 | 1,314 | (333 | ) | 1,005 | |||||||||||||||||||||||
| Net earnings |
34,857 | 34,857 | ||||||||||||||||||||||||||
| Foreign currency translation adjustments |
(124,361 | ) | (124,361 | ) | ||||||||||||||||||||||||
| Unrealized loss on cash flow hedges |
(387 | ) | (387 | ) | ||||||||||||||||||||||||
| Defined benefit plans: |
||||||||||||||||||||||||||||
| Amortization of prior service costs and unrecognized losses and credits |
2,513 | 2,513 | ||||||||||||||||||||||||||
| Plan amendment giving rise to prior service credit |
12,455 | 12,455 | ||||||||||||||||||||||||||
| Amounts arising during the year, primarily due to the addition of new participants |
(4,287 | ) | (4,287 | ) | ||||||||||||||||||||||||
| Marketable securities holding loss |
(113 | ) | (113 | ) | ||||||||||||||||||||||||
| Total comprehensive loss |
(79,323 | ) | ||||||||||||||||||||||||||
| Dividends |
(1,599 | ) | (1,599 | ) | ||||||||||||||||||||||||
| December 31, 2008 Balance |
$ | 8,119 | $ | 278 | $ | 215,279 | $ | 306,698 | $ | 50,789 | $ | (48,221 | ) | $ | 532,942 | |||||||||||||
| Exercise of stock options |
123 | 9,529 | (8,297 | ) | 1,355 | |||||||||||||||||||||||
| Non-qualified stock option expense |
2,713 | 2,713 | ||||||||||||||||||||||||||
| Restricted stock awards |
31 | 1,751 | (544 | ) | 1,238 | |||||||||||||||||||||||
| Net earnings |
41,179 | 41,179 | ||||||||||||||||||||||||||
| Foreign currency translation adjustments |
119,453 | 119,453 | ||||||||||||||||||||||||||
| Unrealized gain on cash flow hedges |
3,329 | 3,329 | ||||||||||||||||||||||||||
| Defined benefit plans: |
||||||||||||||||||||||||||||
| Amortization of prior service costs and unrecognized losses and credits |
537 | 537 | ||||||||||||||||||||||||||
| Marketable securities holding gain |
96 | 96 | ||||||||||||||||||||||||||
| Total comprehensive income |
164,594 | |||||||||||||||||||||||||||
| Dividends |
(1,605 | ) | (1,605 | ) | ||||||||||||||||||||||||
| December 31, 2009 Balance |
$ | 8,273 | $ | 278 | $ | 229,272 | $ | 346,272 | $ | 174,204 | $ | (57,062 | ) | $ | 701,237 | |||||||||||||
| Exercise of stock options |
99 | 9,108 | (6,909 | ) | 2,298 | |||||||||||||||||||||||
| Non-qualified stock option expense |
4,113 | 4,113 | ||||||||||||||||||||||||||
| Restricted stock awards |
23 | 1,999 | (921 | ) | 1,101 | |||||||||||||||||||||||
| Conversion from Class B Stock to Common Stock |
6 | (6 | ) | — | ||||||||||||||||||||||||
| Net earnings |
25,341 | 25,341 | ||||||||||||||||||||||||||
| Foreign currency translation adjustments |
(59,823 | ) | (59,823 | ) | ||||||||||||||||||||||||
| Unrealized gain on cash flow hedges |
245 | 245 | ||||||||||||||||||||||||||
| Defined benefit plans: |
||||||||||||||||||||||||||||
| Amortization of prior service costs and unrecognized losses and credits |
549 | 549 | ||||||||||||||||||||||||||
| Amounts arising during the year, primarily due to the addition of new participants |
(1,860 | ) | (1,860 | ) | ||||||||||||||||||||||||
| Marketable securities holding loss |
(684 | ) | (684 | ) | ||||||||||||||||||||||||
| Total comprehensive loss |
(36,232 | ) | ||||||||||||||||||||||||||
| Extinguishment of Convertible Debt |
(12,807 | ) | (12,807 | ) | ||||||||||||||||||||||||
| Dividends |
(1,612 | ) | (1,612 | ) | ||||||||||||||||||||||||
| Purchase of treasury shares |
(5,687 | ) | (5,687 | ) | ||||||||||||||||||||||||
| December 31, 2010 Balance |
$ | 8,401 | $ | 272 | $ | 231,685 | $ | 370,001 | $ | 112,631 | $ | (70,579 | ) | $ | 652,411 | |||||||||||||
See notes to consolidated financial statements.
FS-6
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Accounting Policies
Nature of Operations: Invacare Corporation is the world’s leading manufacturer and distributor in the estimated $11.0 billion worldwide market for medical equipment and supplies used in the home based upon the company’s distribution channels, breadth of product line and net sales. The company designs, manufactures and distributes an extensive line of health care products for the non-acute care environment, including the home health care, retail and extended care markets.
Principles of Consolidation: The consolidated financial statements include the accounts of the company and its wholly owned subsidiaries. Certain foreign subsidiaries, represented by the European segment, are consolidated using a November 30 fiscal year end in order to meet filing deadlines. No material subsequent events have occurred related to the European segment, which would require disclosure or adjustment to the company’s financial statements. All significant intercompany transactions are eliminated.
Use of Estimates: The consolidated financial statements are prepared in conformity with accounting principles generally accepted in the United States, which require management to make estimates and assumptions that affect the amounts reported in the financial statements and accompanying notes. Actual results may differ from these estimates.
Inventories: Inventories are stated at the lower of cost or market with cost determined by the first-in, first-out method. Market values are based on the lower of replacement cost or estimated net realizable value. Inventories have been reduced by an allowance for excess and obsolete inventories. The estimated allowance is based on management’s review of inventories on hand compared to estimated future usage and sales.
Property and Equipment: Property and equipment are stated on the basis of cost. The company principally uses the straight-line method of depreciation for financial reporting purposes based on annual rates sufficient to amortize the cost of the assets over their estimated useful lives. Machinery and equipment as well as furniture and fixtures are generally depreciated using lives of 3 to 10 years, while buildings and improvements are depreciated using lives of 5 to 40 years. Accelerated methods of depreciation are used for federal income tax purposes. Expenditures for maintenance and repairs are charged to expense as incurred. Amortization of assets under capital leases is included in depreciation expense.
Long-lived assets are reviewed for impairment whenever events or changes in circumstances indicate the carrying amount may not be recoverable. The asset would be considered impaired when the future net undiscounted cash flows generated by the asset are less than its carrying value. An impairment loss would be recognized based on the amount by which the carrying value of the asset exceeds its fair value.
Goodwill and Other Intangibles: In accordance with Intangibles—Goodwill and Other, ASC 350, goodwill and indefinite lived intangibles are subject to annual impairment testing. For purposes of the impairment test, the fair value of each reporting unit is estimated by forecasting cash flows and discounting those cash flows using appropriate discount rates. The fair values are then compared to the carrying value of the net assets of each reporting unit. In 2010, the company recorded impairment charges, included in amortization expense, of $336,000 and $248,000 related to intangible assets for the IPG and the NA/HME segments, respectively, as the actual and future projected cash flows associated with these intangibles were less than what was originally used to value the intangibles. In 2009, the company recorded impairment charges related to intangible assets for Europe of $896,000 and NA/HME of $800,000 as the actual and future projected cash flows associated with these intangibles were less than what was originally used to value the intangibles. No impairments were recognized in 2008.
FS-7
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Accounting Policies—Continued
Accrued Warranty Cost: Generally, the company’s products are covered by warranties against defects in material and workmanship for various periods depending on the product from the date of sale to the customer. Certain components carry a lifetime warranty. A provision for estimated warranty cost is recorded at the time of sale based upon actual experience. The company continuously assesses the adequacy of its product warranty accrual and makes adjustments as needed. Historical analysis is primarily used to determine the company’s warranty reserves. Claims history is reviewed and provisions are adjusted as needed. However, the company does consider other events, such as a product recall, which could warrant additional warranty reserve provision. No material adjustments to warranty reserves were necessary in the current year. See Current Liabilities in the Notes to the Consolidated Financial Statements for a reconciliation of the changes in the warranty accrual.
Product Liability Cost: The company’s captive insurance company, Invatection Insurance Co., currently has a policy year that runs from September 1 to August 31 and insures annual policy losses of $10,000,000 per occurrence and $13,000,000 in the aggregate of the company’s North American product liability exposure. The company also has additional layers of external insurance coverage insuring up to $75,000,000 in annual aggregate losses arising from individual claims anywhere in the world that exceed the captive insurance company policy limits or the limits of the company’s per country foreign liability limits, as applicable. There can be no assurance that Invacare’s current insurance levels will continue to be adequate or available at affordable rates.
Product liability reserves are recorded for individual claims based upon historical experience, industry expertise and other indicators. Additional reserves, in excess of the specific individual case reserves, are provided for incurred but not reported claims based upon actuarial valuations at the time such valuations are conducted. Historical claims experience and other assumptions are taken into consideration by the company in estimating the ultimate reserves. For example, the actuarial analysis assumes that historical loss experience is an indicator of future experience, that the distribution of exposures by geographic area and nature of operations for ongoing operations is expected to be very similar to historical operations with no dramatic changes and that the government indices used to trend losses and exposures are appropriate. Estimates made are adjusted on a regular basis and can be impacted by actual loss awards and settlements on claims. While actuarial analysis is used to help determine adequate reserves, the company is responsible for the determination and recording of adequate reserves in accordance with accepted loss reserving standards and practices.
Revenue Recognition: Invacare’s revenues are recognized when products are shipped to unaffiliated customers, risk of loss is passed and title is transferred. Revenue Recognition, ASC 605, provides guidance on the application of generally accepted accounting principles to selected revenue recognition issues. Shipping and handling costs are included in cost of goods sold.
Sales are made only to customers with whom the company believes collection is reasonably assured based upon a credit analysis, which may include obtaining a credit application, a signed security agreement, personal guarantee and/or a cross corporate guarantee depending on the credit history of the customer. Credit lines are established for new customers after an evaluation of their credit report and/or other relevant financial information. Existing credit lines are regularly reviewed and adjusted with consideration given to any outstanding past due amounts.
The company offers discounts and rebates, which are accounted for as reductions to revenue in the period in which the sale is recognized. Discounts offered include: cash discounts for prompt payment, base and trade discounts based on contract level for specific classes of customers. Volume discounts and rebates are given based on large purchases and the achievement of certain sales volumes. Product returns are accounted for as a reduction to reported sales with estimates recorded for anticipated returns at the time of sale. The company does not sell any goods on consignment.
FS-8
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Accounting Policies—Continued
Distributed products sold by the company are accounted for in accordance with the revenue recognition guidance in ASC 605-45-05. The company records distributed product sales gross as a principal since the company takes title to the products and has the risks of loss for collections, delivery and returns.
Product sales that give rise to installment receivables are recorded at the time of sale when the risks and rewards of ownership are transferred. As such, interest income is recognized based on the terms of the installment agreements. Installment accounts are monitored and if a customer defaults on payments, interest income is no longer recognized. All installment accounts are accounted for using the same methodology, regardless of duration of the installment agreements. The company has entered into an agreement with De Lage Landen, Inc. (“DLL”), a third party financing company, to provide the majority of future lease financing to Invacare customers.
Research and Development: Research and development costs are expensed as incurred and included in cost of products sold. The company’s annual expenditures for product development and engineering were approximately $25,954,000, $25,725,000 and $24,764,000 for 2010, 2009 and 2008, respectively.
Advertising: Advertising costs are expensed as incurred and included in selling, general and administrative expenses. The company has a co-op advertising program in which the company reimburses customers up to 50% of their costs of qualifying advertising expenditures. Invacare product and brand logos must appear in all advertising. Invacare requires customers to submit proof of advertising with their claims for reimbursement. The company’s cost of the program is included in SG&A expense in the consolidated statement of earnings at the time the liability is estimated. Reimbursement is made on an annual basis and within 3 months of submission and approval of the documentation. The company receives monthly reporting from those in the program of their qualified advertising dollars spent and accrues based upon information received. Advertising expenses amounted to $20,119,000, $16,519,000 and $16,224,000 for 2010, 2009 and 2008, respectively, the majority of which is incurred for advertising in the United States.
Stock-Based Compensation Plans: The company accounts for share based compensation under the provisions of the Compensation—Stock Compensation, ASC 718. The amounts of stock-based compensation expense recognized were as follows (in thousands):
| 2010 | 2009 | 2008 | ||||||||||
| Stock-based compensation expense recognized as part of selling, general and administrative expense |
$ | 6,135 | $ | 4,495 | $ | 3,299 | ||||||
The amounts above reflect compensation expense related to restricted stock awards and nonqualified stock options awarded under the 2003 Performance Plan. Stock-based compensation is not allocated to the business segments, but is reported as part of All Other as shown in the company’s Business Segment Note to the Consolidated Financial Statements.
Income Taxes: The company uses the liability method in measuring the provision for income taxes and recognizing deferred tax assets and liabilities on the balance sheet. The liability method requires that deferred income taxes reflect the tax consequences of currently enacted rates for differences between the tax and financial reporting bases of assets and liabilities. With the exception of two subsidiaries, undistributed earnings of the company’s foreign subsidiaries are considered to be indefinitely reinvested and, accordingly with the exception of the two subsidiaries, no provision for income taxes has been provided for unremitted earnings of these foreign subsidiaries. The amount of the unrecognized deferred tax liability for temporary differences related to investments in these foreign subsidiaries that are permanently reinvested is not practically determinable. The
FS-9
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Accounting Policies—Continued
company has established a deferred tax liability of $625,000 for the unremitted earnings of the two subsidiaries for which the company intends to remit earnings when available under local statutory laws.
Derivative Instruments: Derivatives and Hedging, ASC 815, requires companies to recognize all derivative instruments in the consolidated balance sheet as either assets or liabilities at fair value. The accounting for changes in fair value of a derivative is dependent upon whether or not the derivative has been designated and qualifies for hedge accounting treatment and the type of hedging relationship. For derivatives designated and qualifying as hedging instruments, the company must designate the hedging instrument, based upon the exposure being hedged, as a fair value hedge, cash flow hedge, or a hedge of a net investment in a foreign operation.
The company recognizes its derivative instruments as assets or liabilities in the consolidated balance sheet measured at fair value. A majority of the company’s derivative instruments are designated and qualify as cash flow hedges. Accordingly, the effective portion of the gain or loss on the derivative instrument is reported as a component of other comprehensive income and reclassified into earnings in the same period or periods during which the hedged transaction affects earnings. The remaining gain or loss on the derivative instrument in excess of the cumulative change in the fair value of the hedged item, if any, is recognized in current earnings during the period of change.
Foreign Currency Translation: The functional currency of the company’s subsidiaries outside the United States is the applicable local currency. The assets and liabilities of the company’s foreign subsidiaries are translated into U.S. dollars at year-end exchange rates. Revenues and expenses are translated at monthly average exchange rates. Gains and losses resulting from translation of balance sheet items are included in accumulated other comprehensive earnings.
Net Earnings Per Share: Basic earnings per share are computed based on the weighted-average number of Common Shares and Class B Common Shares outstanding during the year. Diluted earnings per share are computed based on the weighted-average number of Common Shares and Class B Common Shares outstanding plus the effects of dilutive stock options and awards outstanding during the year. Diluted earnings per share can potentially be impacted by the convertible notes should the conditions be met to make the notes convertible or if average market price of company stock for the period exceeds the conversion price of $24.79.
Defined Benefit Plans: The company’s benefit plans are accounted for in accordance with Compensation-Retirement Benefits, ASC 715 which requires plan sponsors to recognize the funded status of their defined benefit postretirement benefit plans in the consolidated balance sheet, measure the fair value of plan assets and benefit obligations as of the balance sheet date and to recognize changes in that funded status in the year in which the changes occur through comprehensive income.
Recent Accounting Pronouncements: On January 21, 2010, the Financial Accounting Standards Board (FASB) issued Accounting Standards Update No. 2010-06, Improving Disclosures about Fair Value Measurements (ASU 2010-06). ASU 2010-06 amends ASC 820 to require a number of additional disclosures regarding fair value measurements. The amended guidance requires entities to disclose additional information regarding assets and liabilities that are transferred between levels of the fair value hierarchy. Entities are also required to disclose information in the Level 3 roll forward about purchases, sales, issuances and settlements on a gross basis. In addition to these new disclosure requirements, ASU 2010-06 also amends Topic 820 to further clarify existing guidance pertaining to the level of disaggregation at which fair value disclosures should be made and the requirements to disclose information about the valuation techniques and inputs used in estimating Level II and Level III fair value measurements. The company adopted ASU 2010-06 effective January 1, 2010 and it was utilized in preparing the fair value measurement disclosures.
FS-10
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Accounting Policies—Continued
On July 21, 2010, the FASB issued Accounting Standards Update No. 2010-20, Disclosures about the Credit Quality of Financing Receivables and the Allowance for Credit Losses (ASU 2010-20). ASU 2010-20 requires entities to provide additional disclosures regarding credit-risk exposures, including how credit risk is analyzed and assessed, and allowances for credit losses, including reasons for changes each period. The company adopted ASU 2010-20 effective December 31, 2010 and the company’s receivable disclosures were prepared considering the additional disclosures. The adoption of ASU 2010-20 did not have any material impact on the company’s financial position, results of operations or cash flows.
Receivables
Accounts receivable are reduced by an allowance for amounts that may become uncollectible in the future. Substantially all of the company’s receivables are due from health care, medical equipment providers and long term care facilities located throughout the United States, Australia, Canada, New Zealand and Europe. A significant portion of products sold to providers, both foreign and domestic, is ultimately funded through government reimbursement programs such as Medicare and Medicaid in the U.S. As a consequence, changes in these programs can have an adverse impact on dealer liquidity and profitability. The estimated allowance for uncollectible amounts ($25,327,000 in 2010 and $21,995,000 in 2009) is based primarily on management’s evaluation of the financial condition of specific customers. In addition, as a result of the third party financing arrangement with DLL, a third party financing company which the company has worked with since 2000, management monitors the collection status of these contracts in accordance with the company’s limited recourse obligations and provides amounts necessary for estimated losses in the allowance for doubtful accounts and establishing reserves for specific customers as needed. The company charges off uncollectible trade accounts receivable after such receivables are moved to collection status and legal remedies are exhausted. See Concentration of Credit Risk in the Notes to the Consolidated Financial Statements for a description of the financing arrangement. Long-term installment receivables are included in “Other Assets” on the consolidated balance sheet.
The company’s U.S. customers electing to finance their purchases can do so using DLL. In addition, Invacare often provides financing directly for its Canadian customers for which DLL is not an option, as DLL typically provides financing to Canadian customers only on a limited basis. The installment receivables recorded on the books of the company represent a single portfolio segment of finance receivables to the independent provider channel. The portfolio segment is comprised of two classes of receivables distinguished by geography and credit quality. The U.S. installment receivables are the first class and represent installment receivables re-purchased from DLL because the customers were in default. Default with DLL is defined as a customer being delinquent by three payments. The Canadian installment receivables represent the second class of installment receivables which were originally financed by Invacare because third party financing was not available to the HME providers. The Canadian installment receivables are typically financed for twelve months and historically have had a very low risk of default.
The estimated allowance for uncollectible amounts and evaluation for impairment for both classes of installment receivables is based on the company’s quarterly review of the financial condition of each individual customer with the allowance for doubtful accounts adjusted accordingly. Installments are individually and not collectively reviewed for impairment. The company assesses the bad debt reserve levels based upon the status of the customer’s adherence to a legally negotiated payment schedule and the company’s ability to enforce judgments, liens, etc.
For purposes of granting or extending credit, the company utilizes a scoring model to generate a composite score that considers each customer’s consumer credit score and or D&B credit rating, payment history, security
FS-11
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Receivables—Continued
collateral and time in business. Additional analysis is performed for customers desiring credit greater than $250,000 which includes a detailed review of the customer’s financials as well as consideration of other factors such as exposure to changing reimbursement laws.
Interest income is recognized on installment receivables based on the terms of the installment agreements. Installment accounts are monitored and if a customer defaults on payments and is moved to collection, interest income is no longer recognized. Subsequent payments received once an account is put on non-accrual status are generally first applied to the principal balance and then to the interest. Accruing of interest on collection accounts does not occur and accruing of interest would only be restarted if the account became current again. All installment accounts are accounted for using the same methodology regardless of the duration of the installment agreements. When an account is placed in collection status, the company goes through a legal process of adjudication which typically approximates 18 months. Any write-offs are made after the legal process has been completed. The company has not made any changes to either its accounting policies or methodology to estimation allowances for doubtful accounts in the last twelve months.
Installment receivables as of December 31, 2010 and 2009 consist of the following (in thousands):
| 2010 | 2009 | |||||||||||||||||||||||
| Current | Long- Term |
Total | Current | Long- Term |
Total | |||||||||||||||||||
| Installment receivables |
$ | 5,777 | $ | 4,854 | $ | 10,631 | $ | 5,015 | $ | 8,268 | $ | 13,283 | ||||||||||||
| Less: |
||||||||||||||||||||||||
| Unearned interest |
(118 | ) | — | (118 | ) | (97 | ) | — | (97 | ) | ||||||||||||||
| 5,659 | 4,854 | 10,513 | 4,918 | 8,268 | 13,186 | |||||||||||||||||||
| Allowance for doubtful accounts |
(1,700 | ) | (3,141 | ) | (4,841 | ) | (1,353 | ) | (4,727 | ) | (6,080 | ) | ||||||||||||
| $ | 3,959 | $ | 1,713 | $ | 5,672 | $ | 3,565 | $ | 3,541 | $ | 7,106 | |||||||||||||
Installment receivable purchased from DLL during the twelve months ended December 31, 2010 increased the gross installment receivables balance by $4,799,000 during the year compared to $5,242,000 in 2009. No sales of installment receivables were made by the company during the year.
The movement in the installment receivables allowance for doubtful accounts was as follows (in thousands):
| 2010 | ||||
| Balance as of January 1 |
$ | 6,080 | ||
| Current period provision |
4,022 | |||
| Direct write-offs charged against the allowance |
(5,261 | ) | ||
| Balance as of December 31 |
$ | 4,841 | ||
FS-12
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Receivables—Continued
Installment receivables by class as of December 31, 2010 consist of the following (in thousands):
| 2010 | ||||||||||||||||
| Total Installment Receivables |
Unpaid Principal Balance |
Related Allowance for Doubtful Accounts |
Interest Income Recognized |
|||||||||||||
| U.S. |
||||||||||||||||
| Impaired Installment receivables with a related allowance recorded |
$ | 7,153 | $ | 7,153 | $ | 4,822 | $ | — | ||||||||
| Canada |
||||||||||||||||
| Non-Impaired Installment receivables with no related allowance recorded |
3,222 | 3,104 | — | 109 | ||||||||||||
| Impaired Installment receivables with a related allowance recorded |
256 | 256 | 19 | — | ||||||||||||
| Total Canadian Installment Receivables |
$ | 3,478 | $ | 3,360 | $ | 19 | $ | 109 | ||||||||
| Total |
||||||||||||||||
| Non-Impaired Installment receivables with no related allowance recorded |
3,222 | 3,104 | — | 109 | ||||||||||||
| Impaired Installment receivables with a related allowance recorded |
7,409 | 7,409 | 4,841 | — | ||||||||||||
| Total Installment Receivables |
$ | 10,631 | $ | 10,513 | $ | 4,841 | $ | 109 | ||||||||
Installment receivables with a related allowance recorded as noted in the table above represent those installment receivables on a non-accrual basis in accordance with ASU 2010-20. As of December 31, 2010, the company had no U.S. installment receivables past due of 90 days or more for which the company is still accruing interest. Individually, all U.S. installment receivables are assigned a specific allowance for doubtful accounts based on management’s review when the company does not expect to receive both the contractual principal and interest payments as specified in the loan agreement. However, while the full balance may be deemed to be impaired, the company does historically collect a large percentage of the principal of its U.S. installment receivables.
The company had Canadian installment receivables which were past due of 90 days or more totaling $7,000 for which the company is still accruing interest.
The aging of the company’s installment receivables was as follows as of December 31, 2010 (in thousands):
| Total | U.S. | Canada | ||||||||||
| Current |
$ | 3,097 | $ | — | $ | 3,097 | ||||||
| 0-30 Days Past Due |
89 | — | 89 | |||||||||
| 31-60 Days Past Due |
31 | — | 31 | |||||||||
| 61-90 Days Past Due |
5 | — | 5 | |||||||||
| 90+ Days Past Due |
7,409 | 7,153 | 256 | |||||||||
| $ | 10,631 | $ | 7,153 | $ | 3,478 | |||||||
FS-13
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Inventories
Inventories as of December 31, 2010 and 2009 consist of the following (in thousands):
| 2010 | 2009 | |||||||
| Finished goods |
$ | 101,243 | $ | 99,701 | ||||
| Raw materials |
59,921 | 59,451 | ||||||
| Work in process |
13,211 | 13,070 | ||||||
| $ | 174,375 | $ | 172,222 | |||||
Other Current Assets
Other current assets as of December 31, 2010 and 2009 consist of the following (in thousands):
| 2010 | 2009 | |||||||
| Value added tax receivables |
$ | 13,829 | $ | 14,347 | ||||
| Supplier receivables |
5,703 | 5,500 | ||||||
| Recoverable income taxes |
3,708 | 13,195 | ||||||
| Derivatives (forward exchange contracts) |
2,884 | 1,907 | ||||||
| Prepaid insurance |
2,222 | 2,105 | ||||||
| Prepaids and other current assets |
13,235 | 14,718 | ||||||
| $ | 41,581 | $ | 51,772 | |||||
Property and Equipment
Property and equipment as of December 31, 2010 and 2009 consist of the following (in thousands):
| 2010 | 2009 | |||||||
| Machinery and equipment |
$ | 332,687 | $ | 329,181 | ||||
| Land, buildings and improvements |
91,956 | 98,160 | ||||||
| Furniture and fixtures |
27,775 | 26,635 | ||||||
| Leasehold improvements |
15,705 | 14,744 | ||||||
| 468,123 | 468,720 | |||||||
| Less allowance for depreciation |
(337,360 | ) | (327,087 | ) | ||||
| $ | 130,763 | $ | 141,633 | |||||
Acquisitions
In June 2010, Invacare Corporation acquired Centralized Medical Equipment LLC and the majority of the assets of Specialty Medical Equipment LLC, both Massachusetts limited liability companies, collectively referred to as Boston Rentals, which rent equipment to skilled nursing and long-term care providers, for $13,725,000, which was paid in cash. The results of the acquisitions are included in the Institutional Products Group segment since the date of acquisition.
In March 2008, Invacare Corporation acquired the assets of Naylor Medical Sales & Rentals, Inc. (Naylor), a Tennessee corporation specializing in renting product, for $2,152,000, which was paid in cash. In October 2008, Invacare Corporation purchased a billing company operating as Homecare Collection Services (HCS) for
FS-14
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Acquisitions—Continued
$6,268,000. Both of these acquisitions were made to expand the company’s services business. The company’s results of operations include the results of Naylor and HCS since their respective dates of acquisition. Pursuant to the HCS purchase agreement, the company agreed to pay contingent consideration based upon earnings before interest, taxes and depreciation over the three years subsequent to the acquisition up to a maximum of $3,000,000. During 2010, the company settled the contingency for the minimum amount of $500,000.
Goodwill
The carrying amount of goodwill by operating segment is as follows (in thousands):
| North America/ HME |
Invacare Supply Group |
Institutional Products Group |
Europe | Asia/ Pacific |
Consolidated | |||||||||||||||||||
| Balance at January 1, 2009 |
$ | 9,162 | $ | 23,073 | $ | 17,511 | $ | 396,632 | $ | 28,308 | $ | 474,686 | ||||||||||||
| Foreign currency translation adjustments |
— | — | 2,756 | 70,753 | 7,509 | 81,018 | ||||||||||||||||||
| Purchase accounting adjustments |
389 | — | — | — | — | 389 | ||||||||||||||||||
| Balance at December 31, 2009 |
$ | 9,551 | $ | 23,073 | $ | 20,267 | $ | 467,385 | $ | 35,817 | $ | 556,093 | ||||||||||||
| Foreign currency translation adjustments |
— | — | 1,238 | (60,870 | ) | 4,330 | (55,302 | ) | ||||||||||||||||
| Acquisitions |
6,292 | — | — | — | — | 6,292 | ||||||||||||||||||
| Balance at December 31, 2010 |
$ | 15,843 | $ | 23,073 | $ | 21,505 | $ | 406,515 | $ | 40,147 | $ | 507,083 | ||||||||||||
As a result of the Boston Rentals acquisition in 2010, additional goodwill of $6,292,000 was recorded, which is deductible for tax purposes. In accordance with Intangibles—Goodwill and Other, ASC 350, goodwill is reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. The company completes its annual impairment tests in the fourth quarter of each year. The discount rates used have a significant impact upon the discounted cash flow methodology utilized in the company’s annual impairment testing as higher discount rates decrease the fair value estimates used in the company’s testing. For purposes of Step I of the impairment test, the fair value of each reporting unit is estimated by forecasting cash flows and discounting those cash flows using appropriate discount rates. The fair values are then compared to the carrying value of the net assets of each reporting unit. Step II of the impairment test requires a more detailed assessment of the fair values associated with the net assets of a reporting unit that fails the Step I test, including a review for impairment in accordance with Property, Plant and Equipment, ASC 360.
The company utilizes a discounted cash flow method model to analyze reporting units for impairment in which the company forecasts income statement and balance sheet amounts based on assumptions regarding future sales growth, profitability, inventory turns, days’ sales outstanding, etc. to forecast future cash flows. The cash flows are discounted using a weighted average cost of capital discount rate where the cost of debt is based on quoted rates for 20-year debt of companies of similar credit risk and the cost of equity is based upon the 20-year treasury rate for the risk free rate, a market risk premium, the industry average beta and a small cap stock adjustment. The assumptions used are based on a market participant’s point of view and yielded a discount rate of 9.59% in 2010 compared to 10.74% in 2009 and 8.90% to 9.90% in 2008.
The company also utilizes an EV (Enterprise Value) to EBITDA (Earnings Before Interest Taxes Depreciation and Amortization) Method to compute the fair value of its reporting units which considers potential acquirers and their EV to EBITDA multiples adjusted by an estimated premium. While more weight is given to the discounted cash flow method, the EV to EBITDA Method does provide corroborative evidence of the reasonableness of the discounted cash flow method results.
FS-15
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Goodwill—Continued
While there was no indication of impairment in 2010 related to goodwill, a future potential impairment is possible for any of the company’s reporting units should actual results differ materially from forecasted results used in the valuation analysis. Furthermore, the company’s annual valuation of goodwill can differ materially if the market inputs used to determine the discount rate change significantly. For instance, higher interest rates or greater stock price volatility would increase the discount rate and thus increase the chance of impairment. For example, if the discount rate used were 100 basis points higher for the 2010 impairment analysis, there still would not be any indicator of potential impairment for any of the reporting units.
Other Intangibles
All of the company’s other intangible assets have been assigned definite lives and continue to be amortized over their useful lives, except for $31,246,000 related to trademarks, which have indefinite lives. The changes in intangible balances reflected on the balance sheet from December 31, 2009 to December 31, 2010 were the result of foreign currency translation and amortization except for intangible write-downs, noted below, which totaled $584,000 and the addition of a customer list intangible of $2,430,000, which was assigned a five-year life, as a result of the Boston Rentals acquisition. The company’s intangibles consist of the following (in thousands):
| December 31, 2010 | December 31, 2009 | |||||||||||||||
| Historical Cost |
Accumulated Amortization |
Historical Cost |
Accumulated Amortization |
|||||||||||||
| Customer Lists |
$ | 72,998 | $ | 40,071 | $ | 78,780 | $ | 36,359 | ||||||||
| Trademarks |
31,246 | — | 34,953 | — | ||||||||||||
| License agreements |
3,183 | 2,958 | 4,326 | 4,051 | ||||||||||||
| Developed Technology |
8,521 | 3,988 | 7,409 | 2,434 | ||||||||||||
| Patents |
7,518 | 5,863 | 7,020 | 5,246 | ||||||||||||
| Other |
6,092 | 5,767 | 5,905 | 4,998 | ||||||||||||
| $ | 129,558 | $ | 58,647 | $ | 138,393 | $ | 53,088 | |||||||||
Amortization expense related to other intangibles was $8,451,000, $8,671,000 and $9,634,000 for 2010, 2009 and 2008, respectively. Estimated amortization expense for each of the next five years is expected to be $8,659,000 for 2011, $7,837,000 in 2012, $6,970,000 in 2013, $6,704,000 in 2014 and $5,615,000 in 2015. Amortized intangibles are being amortized on a straight-line basis for periods from 3 to 20 years with the majority of the intangibles being amortized over a life of between 10 and 13 years.
In accordance with ASC 350, Intangibles—Goodwill and Other, the company reviews intangibles for impairment. For purposes of the impairment test, the fair value of each indefinite lived intangible is estimated by forecasting cash flows and discounting those cash flows using appropriate discount rates. The fair values are then compared to the carrying value of the intangible. For amortized intangibles, the forecasted undiscounted cash flows were compared to the carrying value, and if impairment results, the impairment is measured based on the estimated fair value of the intangibles. In 2010, the company recorded impairment charges of $336,000 and $248,000 related to certain intangible assets in the IPG and the NA/HME segments, respectively, as the actual and remaining forecasted cash flows associated with these intangibles were less than what was originally used to value the intangibles. As a result of the company’s 2009 intangible impairment review, impairment charges of $896,000 and $800,000 were recorded related to trademarks for Europe and a customer list for NA/HME, respectively, as the actual and remaining forecasted cash flows associated with these intangibles were less than the cash flows originally used to value the intangibles.
FS-16
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Current Liabilities
Accrued expenses as of December 31, 2010 and 2009 consisted of accruals for the following (in thousands):
| 2010 | 2009 | |||||||
| Salaries and wages |
$ | 46,658 | $ | 45,252 | ||||
| Taxes other than income taxes, primarily Value Added Taxes |
19,981 | 19,390 | ||||||
| Warranty cost |
18,252 | 21,506 | ||||||
| Freight |
11,189 | 13,058 | ||||||
| Professional |
7,333 | 5,888 | ||||||
| Product liability, current portion |
4,134 | 4,232 | ||||||
| Rebates |
3,320 | 3,488 | ||||||
| Insurance |
2,393 | 2,270 | ||||||
| Interest |
2,273 | 9,822 | ||||||
| Derivative liability (foreign forward exchange contracts) |
1,929 | 2,173 | ||||||
| Severance |
524 | 1,507 | ||||||
| Other items, principally trade accruals |
12,093 | 13,707 | ||||||
| $ | 130,079 | $ | 142,293 | |||||
Accrued rebates relate to several volume incentive programs the company offers its customers. The company accounts for these rebates as a reduction of revenue when the products are sold in accordance with the guidance in ASC 605-50, Customer Payments and Incentives.
Changes in accrued warranty costs were as follows (in thousands):
| 2010 | 2009 | |||||||
| Balance as of January 1 |
$ | 21,506 | $ | 16,798 | ||||
| Warranties provided during the period |
5,996 | 12,186 | ||||||
| Settlements made during the period |
(9,681 | ) | (9,404 | ) | ||||
| Changes in liability for pre-existing warranties during the period, including expirations |
431 | 1,926 | ||||||
| Balance as of December 31 |
$ | 18,252 | $ | 21,506 | ||||
Long-Term Debt
Debt as of December 31, 2010 and 2009 consisted of the following (in thousands):
| 2010 | 2009 | |||||||
| $400,000,000 senior secured revolving credit facility, due in October 2015 |
$ | 184,932 | $ | — | ||||
| Revolving credit agreements, due in February 2012 |
— | 1,725 | ||||||
| Convertible senior subordinated debentures at 4.125%, due in February 2027 |
52,064 | 86,728 | ||||||
| Senior notes at 9.75%, due in February 2015 |
— | 173,490 | ||||||
| Other notes and lease obligations |
9,068 | 11,382 | ||||||
| 246,064 | 273,325 | |||||||
| Less current maturities of long-term debt |
(7,974 | ) | (1,091 | ) | ||||
| $ | 238,090 | $ | 272,234 | |||||
FS-17
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Long-Term Debt—Continued
On October 28, 2010, the company entered into a new credit agreement (the “New Credit Agreement”) which provides for a $400,000,000 senior secured revolving credit facility maturing in October 2015. Pursuant to the terms of the New Credit Agreement, the company may from time to time borrow, repay and re-borrow up to an aggregate outstanding at any one time of $400,000,000 under the new senior secured revolving credit facility, subject to customary conditions. The New Credit Agreement also provides for the issuance of swing line loans and letters of credit.
Borrowings under the new senior secured revolving credit facility bear interest, at the company’s election, at (i) the London Inter-Bank Offer Rate (“LIBOR”) plus a margin; or (ii) a Base Rate Option plus a margin. The applicable margin is based on the company’s leverage ratio and at the time of entry into the New Credit Agreement, the applicable margin was 2.50% per annum for LIBOR loans and 1.50% for the Base Rate Option loans. In addition to interest, the company is required to pay commitment fees on the unused portion of the senior secured revolving credit facility. The commitment fee rate is initially 0.40% per annum and, like the interest rate spreads, is subject to adjustment thereafter based on the company’s leverage ratio. The obligations of the borrowers under the New Credit Agreement are secured by substantially all of the company’s U.S. assets and are guaranteed by substantially all of the company’s material domestic and foreign subsidiaries.
The New Credit Agreement contains certain covenants that are customary for similar credit arrangements, including covenants relating to, among other things, financial reporting and notification, compliance with laws, preservation of existence, maintenance of books and records, use of proceeds, maintenance of properties and insurance, and limitations on liens, dispositions, issuance of debt, investments, payment of dividends, repurchases of capital stock, acquisitions, transactions with affiliates, and capital expenditures. There also are financial covenants that require the company to maintain a maximum leverage ratio (consolidated funded indebtedness to consolidated EBITDA, each as defined in the New Credit Agreement) of no greater than 3.50 to 1, and a minimum interest coverage ratio (consolidated EBITDA to consolidated interest charges, each as defined in the New Credit Agreement) of no less than 3.50 to 1. As of December 31, 2010, the company’s leverage ratio was 1.89 and the company’s interest coverage ratio was 8.40 and the company was in compliance with all covenant requirements. Under the most restrictive covenant of the company’s borrowing arrangements as of December 31, 2010, the company had the capacity to borrow up to an additional $215,068,000.
The New Credit Agreement required the company to redeem, purchase or repurchase no less than $100 million in principal amount of the 9.75% Senior Notes due 2015 (the “Senior Notes”) and/or the company’s 4.25% Convertible Senior Subordinated Debentures due 2027 (the “Convertible Notes”) by February 28, 2011. This was completed by December 31, 2010. After February 28, 2011, the company may redeem, purchase or repurchase the Convertible Notes so long as no event of default is then occurring or would be caused thereby and the company’s leverage ratio after such redemption, purchase or repurchase is not more than 3.00 to 1. The New Credit Agreement provides for customary events of default with corresponding grace periods, including, among other things, failure to pay any principal or interest when due, failure to perform or observe covenants, bankruptcy or insolvency events and change of control.
On February 12, 2007, the company entered into a $400,000,000 senior secured credit facility (“revolving credit agreement”) consisting of a $250,000,000 term loan facility and a $150,000,000 revolving credit facility. The company’s obligations under the revolving credit facility were secured by substantially all of the company’s assets and were guaranteed by its material domestic subsidiaries, with certain obligations also guaranteed by its material foreign subsidiaries. Borrowings under the revolving credit facility were at LIBOR plus a margin of 1.25%, including a facility fee of 0.25% per annum on the facility. During 2009, the company fully repaid its $250,000,000 term loan facility which was not due to expire until February 2013. As a result, $2,878,000 of
FS-18
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Long-Term Debt—Continued
deferred financing fees, which were previously capitalized, were expensed in the All Other operating segment in 2009. As part of the refinancing done on October 28, 2010, the outstanding amount owed on the existing revolving credit agreement entered into in 2007 was repaid, and as a result, $1,228,000 of deferred financing fees, which were previously capitalized, were expensed in the All Other segment.
In February 2007, the company issued $175,000,000 principal amount of 9.75% Senior Notes due 2015. The notes were unsecured senior obligations of the company guaranteed by substantially all of the company’s domestic subsidiaries, and paid interest at 9.75% per annum on each February 15 and August 15. During 2010, the company retired all of its outstanding Senior Notes at a premium above par and recognized a loss of $18,671,000, including $3,764,000 of deferred financing fees, which were previously capitalized.
Also, in 2007, the company issued $135,000,000 principal amount of Convertible Senior Subordinated Debentures due 2027. The debentures are unsecured senior subordinated obligations of the company guaranteed by substantially all of the company’s domestic subsidiaries, pay interest at 4.125% per annum on each February 1 and August 1, and are convertible upon satisfaction of certain conditions into cash, common shares of the company, or a combination of cash and common shares of the company, subject to certain conditions, and at the company’s discretion. The debentures allow the company to satisfy the conversion using any combination of cash or stock. The company intends to satisfy the accreted value of the debentures using cash. Assuming adequate cash on hand at the time of conversion, the company also intends to satisfy the conversion spread using cash, as opposed to stock. As of December 31, 2010, the if-converted value of the company’s Convertible Notes exceeded the principal amount of those notes by $16,721,000. During 2010, the company retired $57,799,000 in principal amount of Convertible Notes at a premium above par. In accordance with ASC 470-20, Convertible Debt, the company utilized the inducement method of accounting to calculate the loss associated with the early retirement of the convertible debt. For the year ended December 31, 2010, the company recorded expense of $20,265,000 related to the loss on the debt extinguishment including the write-off of $1,502,000 of deferred financing fees, which were previously capitalized.
The company includes the dilutive effect of shares necessary to settle the conversion spread in the Net Earnings per Share—Assuming Dilution calculation unless such amounts are anti-dilutive. The initial conversion rate is 40.3323 shares per $1,000 principal amount of debentures, which represents an initial conversion price of approximately $24.79 per share. Holders of the debentures can convert the debt to common stock if the company’s common stock price is at a level in excess of $32.23, a 30% premium to the initial conversion price for at least 20 trading days during a period of 30 consecutive trading days preceding the date on which the notice of conversion is given. At a conversion price of $32.23 (30% premium over $24.79), the full conversion of the convertible debt equates to 3,114,000 shares. The debentures are redeemable at the company’s option, subject to specified conditions, on or after February 6, 2012 through and including February 1, 2017. The company may redeem some or all of the debentures for cash on or after February 1, 2017. Holders have the right to require the company to repurchase all or some of their debentures upon the occurrence of certain circumstances on February 1, 2017 and 2022. The company evaluated the terms of the call, redemption and conversion features under the applicable accounting literature, including Derivatives and Hedging, ASC 815, and determined that the features did not require separate accounting as derivatives. The notes, debentures and common shares issuable upon conversion of the debentures have been registered under the Securities Act.
FS-19
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Long-Term Debt—Continued
The components of the company’s convertible debt as of December 31, 2010 and 2009 consist of the following (in thousands):
| 2010 | 2009 | |||||||
| Carrying amount of equity component |
$ | 46,205 | $ | 59,012 | ||||
| Principal amount of liability component |
$ | 77,201 | $ | 135,000 | ||||
| Unamortized discount |
(25,137 | ) | (48,272 | ) | ||||
| Net carrying amount of liability component |
$ | 52,064 | $ | 86,728 | ||||
The unamortized discount of $25,137,000 is to be amortized through February 2017. The effective interest rate on the liability component was 11.5% for 2007 through 2010. Non-cash interest expense of $3,198,000, $4,142,000 and $3,694,000 was recognized in 2010, 2009 and 2008, respectively, in comparison to actual interest expense paid of $4,178,000, $5,569,000 and $5,569,000 based on the stated coupon rate of 4.125%, for each of the same periods. The convertible debt was not convertible as of December 31, 2010 nor was the convertible debt conversion price threshold of $32.23, as noted above, met.
Included in the $400,000,000 senior secured revolving credit facility, there was $12,982,000 of borrowings denominated in foreign currencies as of December 31, 2010 while there were none as of December 31, 2009. For 2010 and 2009, the weighted average interest rate on borrowings was 6.06% and 6.67%, respectively.
In July 2009, cash flow hedges entered into in July 2007 that exchanged the LIBOR variable rate on $125,000,000 of term loan debt for a fixed rate of 5.0525% expired. As of December 31, 2010, the company was not a party to any interest rate swap agreements.
The aggregate minimum maturities of long-term debt for each of the next five years are as follows: $7,974,000 in 2011, $947,000 in 2012, $918,000 in 2013, $972,000 in 2014, and $179,025,000 in 2015. Interest paid on borrowings was $28,341,000, $33,188,000 and $40,547,000 in 2010, 2009 and 2008, respectively.
Other Long-Term Obligations
Other long-term obligations as of December 31, 2010 and 2009 consist of the following (in thousands):
| 2010 | 2009 | |||||||
| Supplemental Executive Retirement Plan liability |
$ | 26,133 | $ | 25,677 | ||||
| Product liability |
20,026 | 19,757 | ||||||
| Deferred income taxes |
32,559 | 30,276 | ||||||
| Deferred compensation |
8,542 | 7,253 | ||||||
| Other |
12,331 | 12,740 | ||||||
| Total long-term obligations |
$ | 99,591 | $ | 95,703 | ||||
Leases and Commitments
The company leases a portion of its facilities, transportation equipment, data processing equipment and certain other equipment. These leases have terms from 1 to 14 years and provide for renewal options. Generally, the company is required to pay taxes and normal expenses of operating the facilities and equipment. As of
FS-20
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Leases and Commitments—Continued
December 31, 2010, the company is committed under non-cancelable operating leases, which have initial or remaining terms in excess of one year and expire on various dates through 2024. Lease expenses were approximately $23,094,000 in 2010, $23,966,000 in 2009, and $23,363,000 in 2008.
The amount of buildings and equipment capitalized in connection with capital leases was $14,197,000 and $17,637,000 at December 31, 2010 and 2009, respectively. At December 31, 2010 and 2009, accumulated amortization was $5,201,000 and $6,295,000, respectively, which is included in depreciation expense.
Future minimum operating and capital lease commitments, as of December 31, 2010, are as follows (in thousands):
| Year |
Capital Leases | Operating Leases | ||||||
| 2011 |
$ | 1,575 | $ | 19,374 | ||||
| 2012 |
1,467 | 12,695 | ||||||
| 2013 |
1,383 | 8,738 | ||||||
| 2014 |
1,363 | 5,906 | ||||||
| 2015 |
1,364 | 3,451 | ||||||
| Thereafter |
4,938 | 7,586 | ||||||
| Total future minimum lease payments |
12,090 | $ | 57,750 | |||||
| Amounts representing interest |
(3,086 | ) | ||||||
| Present value of minimum lease payments |
$ | 9,004 | ||||||
Retirement and Benefit Plans
Substantially all full-time salaried and hourly domestic employees are included in the Invacare Retirement Savings Plan sponsored by the company. The company makes matching cash contributions up to 66.7% of employees’ contributions up to 3% of compensation, quarterly contributions equal to 4% of qualified wages and may make discretionary contributions to the domestic plans based on an annual resolution of the Board of Directors. Contribution expense for the Invacare Retirement Savings Plan in 2010, 2009 and 2008 was $7,153,000, $6,681,000, and $6,140,000, respectively.
The company sponsors a Deferred Compensation Plus Plan covering certain employees, which provides for elective deferrals and the company retirement deferrals so that the total retirement deferrals equal amounts that would have contributed to the company’s principal retirement plans if it were not for limitations imposed by income tax regulations.
The company also sponsors a non-qualified defined benefit Supplemental Executive Retirement Plan (SERP) for certain key executives. Effective December 31, 2008, the SERP was amended, in part to comply with IRS Section 409A. As a result of the amendment, the plan became a defined benefit cash balance plan for the non-retired participants and thus, future payments by the company will be made based upon a cash balance formula with interest credited at a rate determined annually by the Compensation Committee of the Board of Directors, currently 6%. The plan continues to be unfunded with individual hypothetical accounts maintained for each participant. Future company expense will be equal to the hypothetical contributions made for each participant plus the crediting of interest.
The projected benefit obligation related to this unfunded plan was $26,524,000 and $26,068,000 at December 31, 2010 and 2009, respectively, and the accumulated benefit obligation was $26,524,000 and
FS-21
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Retirement and Benefit Plans—Continued
$25,941,000 at December 31, 2010 and 2009, respectively. The projected benefit obligations were calculated using an assumed future salary increase of 4% at both December 31, 2010 and 2009. The assumed discount rate, relevant for three participants unaffected by plan conversion as explained below, for both 2010 and 2009 was 6% based upon the discount rate on high-quality fixed-income investments without adjustment. The retirement age was 65 for both 2010 and 2009. Expense for the plan in 2010, 2009 and 2008 was $2,176,000, $2,128,000, and $2,391,000, respectively of which $1,535,000, $1,454,000, and $1,294,000 was related to interest cost with the remaining portion related to service costs, prior service costs and other gains/losses. Benefit payments in 2010, 2009 and 2008 were $1,592,000, $517,000 and $424,000, respectively. In 2010, benefit payments included a lump sum distribution to a plan participant.
In 2005, the company began sponsoring a Death Benefit Only Plan for certain key executives that provides a benefit equal to three times the participant’s final target earnings should the participant’s death occur while an employee and a benefit equal to one times the participant’s final earnings upon the participant’s death after normal retirement or post-employment. Expense for the plan in 2010, 2009 and 2008 was $399,000, $190,000, and $121,000, respectively of which $235,000, $131,000, and $72,000 was related to service cost and accrual adjustments with the remaining portion related to interest costs. There were no benefit payments in 2010, 2009 or 2008.
In Switzerland, the company also maintains a statutory pension plan with a private insurance company and, in accordance with Swiss law, the plan functions as a defined contribution plan whereby employee and employer contributions are defined as a percentage of individual salary depending on the age of the employee and a guaranteed interest rate, which is annually defined by the Swiss Pension Fund. Under U.S. GAAP, the plan is treated as a defined benefit plan. Expense for the plan was $23,000 and $498,000 in 2010 and 2009, respectively.
Accumulated other comprehensive income associated with the SERP, Swiss pension plan and Death Benefit Only Plan (Defined Benefit Plans) was $2,332,000 and $1,021,000 as of December 31, 2010 and 2009, respectively for a net change of $1,331,000 with $2,598,000 in net periodic benefit costs recognized during the year.
In conjunction with these non-qualified U.S. defined benefit plans, the company has invested in life insurance policies related to certain employees to help satisfy these future obligations. The current cash surrender value of these policies approximates the current benefit obligations.
Shareholders’ Equity Transactions
The company’s Common Shares have a $.25 stated value. The Common Shares and the Class B Common Shares generally have identical rights, terms and conditions and vote together as a single class on most issues, except that the Class B Common Shares have ten votes per share, carry a 10% lower cash dividend rate and, in general, can only be transferred to family members. Holders of Class B Common Shares are entitled to convert their shares into Common Shares at any time on a share-for-share basis.
The 2003 Performance Plan, as amended (the “2003 Plan”), allows the Compensation and Management Development Committee of the Board of Directors (the “Committee”) to grant up to 6,800,000 Common Shares in connection with incentive stock options, non-qualified stock options, stock appreciation rights and stock awards (including the use of restricted stock), which includes the addition of 3,000,000 Common Shares authorized for issuance under the 2003 Plan, as approved by the company’s shareholders on May 21, 2009. The maximum aggregate number of Common Shares that may be granted during the term of the 2003 Plan pursuant to all awards, other than stock options, is 1,300,000 Common Shares. The Committee has the authority to
FS-22
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Shareholders’ Equity Transactions—Continued
determine which participants will receive awards, the amount of the awards and the other terms and conditions of the awards. During 2010, 2009 and 2008, the Committee granted 646,797, 754,581 and 701,594 non-qualified stock options, respectively, each having a term of ten years and generally granted at the fair market value of the company’s Common Shares on the date of grant under the 2003 Plan. There were no stock appreciation rights outstanding at December 31, 2010, 2009 or 2008.
Restricted stock awards for 92,900, 125,840, and 96,800 shares were granted in years 2010, 2009 and 2008 without cost to the recipients. The 2010 weighted average fair value of the 2010 restricted stock awards was $25.26. The restricted stock awards vest ratably over the four years after the award date. There were 91,491 restricted stock awards with a weighted average fair value of $23.32 that vested in 2010 and 17,325 restricted stock awards with a weighted average fair value of $24.23 that were forfeited in 2010.
At December 31, 2010 and 2009, there were 243,770 and 247,961 shares, respectively, for restricted stock awards that were unvested. Unearned restricted stock compensation of $5,190,000 in 2010, $4,866,000 in 2009 and $4,505,000 in 2008, determined as the market value of the shares at the date of grant, is being amortized on a straight-line basis over the vesting period. Compensation expense of $2,023,000, $1,783,000 and $1,338,000 was recognized in 2010, 2009 and 2008, respectively, related to restricted stock awards granted since 2004.
The 2003 Plan and the 1994 Performance Plan have provisions that allow employees to exchange mature shares to pay the exercise price and surrender shares for the options or restricted awards to cover the minimum tax withholding obligation. Under these provisions, the company acquired approximately 280,000 treasury shares for $7,830,000 in 2010, 410,000 shares for $8,841,000 in 2009 and 224,000 shares for $5,344,000 in 2008.
The following table summarizes information about stock option activity for the three years ended December 31, 2010, 2009 and 2008:
| 2010 | Weighted Average Exercise Price |
2009 | Weighted Average Exercise Price |
2008 | Weighted Average Exercise Price |
|||||||||||||||||||
| Options outstanding at January 1 |
4,619,528 | $ | 29.28 | 4,910,547 | $ | 29.37 | 4,732,965 | $ | 30.02 | |||||||||||||||
| Granted |
646,797 | 25.22 | 754,581 | 20.38 | 701,594 | 24.82 | ||||||||||||||||||
| Exercised |
(399,144 | ) | 23.08 | (490,325 | ) | 19.68 | (243,982 | ) | 23.60 | |||||||||||||||
| Canceled |
(382,986 | ) | 25.07 | (555,275 | ) | 26.27 | (280,030 | ) | 33.89 | |||||||||||||||
| Options outstanding at December 31 |
4,484,195 | $ | 29.60 | 4,619,528 | $ | 29.28 | 4,910,547 | $ | 29.37 | |||||||||||||||
| Options exercise price range at December 31 |
$ | 10.70 to | $ | 10.70 to | $ | 10.70 to | ||||||||||||||||||
| $ | 47.80 | $ | 47.80 | $ | 47.80 | |||||||||||||||||||
| Options exercisable at December 31 |
2,941,772 | 3,099,092 | 3,654,689 | |||||||||||||||||||||
| Options available for grant at December 31* |
2,478,905 | 3,132,623 | 746,320 | |||||||||||||||||||||
| * | Options available for grant as of December 31, 2010 reduced by net restricted stock award activity of 487,578. |
FS-23
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Shareholders’ Equity Transactions—Continued
The following table summarizes information about stock options outstanding at December 31, 2010:
| Options Outstanding | Options Exercisable | |||||||||||||||||||
| Exercise Prices |
Number Outstanding At 12/31/10 |
Weighted Average Remaining Contractual Life |
Weighted Average Exercise Price |
Number Exercisable At 12/31/10 |
Weighted Average Exercise Price |
|||||||||||||||
| $ 10.70 – $15.00 |
20,175 | 1.8 years | $ | 10.85 | 19,425 | $ | 10.70 | |||||||||||||
| $ 15.01 – $25.00 |
1,446,742 | 7.4 | $ | 21.66 | 784,436 | $ | 22.30 | |||||||||||||
| $ 25.01 – $35.00 |
1,636,332 | 6.5 | $ | 27.50 | 756,965 | $ | 29.88 | |||||||||||||
| $ 35.01 – $47.80 |
1,380,946 | 3.3 | $ | 40.67 | 1,380,946 | $ | 40.67 | |||||||||||||
| Total |
4,484,195 | 5.8 | $ | 29.60 | 2,941,772 | $ | 32.80 | |||||||||||||
The plans provide that shares granted come from the company’s authorized but unissued Common Shares or treasury shares. In addition, the company’s stock-based compensation plans allow participants to exchange mature shares for the exercise price and surrender shares for minimum withholding taxes, which results in the company acquiring treasury shares. Pursuant to the plans, the Committee has established that the majority of the 2010 grants may not be exercised within one year from the date granted and options must be exercised within ten years from the date granted. Accordingly, for the stock options issued in 2010, 2009 and 2008, 25% of such options vested in the year following issuance. The stock options awarded during such years provided a four-year vesting period whereby options vest equally in each year. The 2010, 2009 and 2008 expense has been adjusted for estimated forfeitures of awards that will not vest because service or employment requirements have not been met.
The fair value of each option grant is estimated on the date of grant using the Black-Scholes option-pricing model with the following weighted-average assumptions:
| 2010 | 2009 | 2008 | ||||||||||
| Expected dividend yield |
.21 | % | .21 | % | .21 | % | ||||||
| Expected stock price volatility |
39.6 | % | 39.9 | % | 31.5 | % | ||||||
| Risk-free interest rate |
1.57 | % | 1.81 | % | 2.65 | % | ||||||
| Expected life in years |
3.9 | 3.7 | 3.7 | |||||||||
| Forfeiture percentage |
10.5 | % | 12.7 | % | 5.7 | % | ||||||
Expected stock price volatility is calculated at each date of grant based on historical stock prices for a period of time commensurate with the expected life of the option. The weighted-average fair value of options granted during 2010, 2009 and 2008 was $7.83, $6.84 and $6.91, respectively. The weighted-average remaining contractual life of options outstanding at December 31, 2010, 2009 and 2008 was 5.8, 5.5 and 5.0 years, respectively. The weighted-average contractual life of options exercisable at December 31, 2010 was 4.3 years. The total intrinsic value of stock awards exercised in 2010, 2009 and 2008 was $1,928,000, $962,000 and $263,000, respectively. As of December 31, 2010, the intrinsic value of all options outstanding and of all options exercisable was $18,136,000 and $7,864,000, respectively.
The exercise of stock awards in 2010, 2009 and 2008 resulted in cash received by the company totaling $2,912,000, $1,628,000 and $834,000 for each period, respectively with no tax benefits for any period. The total fair value of awards vested during 2010, 2009 and 2008 was $5,261,000, $1,716,000 and $1,771,000, respectively.
As of December 31, 2010, there was $15,539,000 of total unrecognized compensation cost from stock-based compensation arrangements granted under the plans, which is related to non-vested options and shares, which
FS-24
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Shareholders’ Equity Transactions—Continued
includes $5,190,000 related to restricted stock awards. The company expects the compensation expense to be recognized over a weighted-average period of approximately two years. Prior to the adoption of ASC 718, Compensation—Stock Compensation, the company presented all tax benefit deductions resulting from the exercise of stock options as a component of operating cash flows in the Consolidated Statement of Cash Flows. In accordance with ASC 718, any tax benefits resulting from tax deductions in excess of the compensation expense recognized for those options is classified as a component of financing cash flows.
Effective July 8, 2005, the company adopted a new Rights Agreement to replace the company’s previous shareholder rights plan, which expired on July 7, 2005. In order to implement the new Rights Agreement, the Board of Directors declared a dividend of one Right for each outstanding share of the company’s Common Shares and Class B Common Shares to shareholders of record at the close of business on July 19, 2005. Each Right entitles the registered holder to purchase from the company one one-thousandth of a Series A Participating Serial Preferred Share, without par value, at a Purchase Price of $180.00 in cash, subject to adjustment. The Rights will not become exercisable until after a person (an “Acquiring Party”) has acquired, or obtained the right to acquire, or commences a tender offer to acquire, shares representing 30% or more of the company’s outstanding voting power, subject to deferral by the Board of Directors. After the Rights become exercisable, under certain circumstances, the Rights may be exercisable to purchase Common Shares of the company, or common shares of an acquiring company, at a price equal to the exercise price of the Right divided by 50% of the then current market price per Common Share or acquiring company common share, as the case may be. The Rights will expire on July 18, 2015 unless previously redeemed or exchanged by the company. The company may redeem and terminate the Rights in whole, but not in part, at a price of $0.001 per Right at any time prior to 10 days following a public announcement that an Acquiring Party has acquired beneficial ownership of shares representing 30% or more of the company’s outstanding voting power, and in certain other circumstances described in the Rights Agreement.
Capital Stock
Capital stock activity for 2010, 2009 and 2008 consisted of the following (in thousands of shares):
| Common Stock Shares |
Class B Shares |
Treasury Shares |
||||||||||
| January 1, 2008 Balance |
32,126 | 1,112 | (1,200 | ) | ||||||||
| Conversion of Class B to Common |
1 | (1 | ) | — | ||||||||
| Exercise of stock options |
242 | — | (204 | ) | ||||||||
| Restricted stock awards |
80 | — | (20 | ) | ||||||||
| December 31, 2008 Balance |
32,449 | 1,111 | (1,424 | ) | ||||||||
| Exercise of stock options |
490 | — | (386 | ) | ||||||||
| Restricted stock awards |
109 | — | (24 | ) | ||||||||
| December 31, 2009 Balance |
33,048 | 1,111 | (1,834 | ) | ||||||||
| Exercise of stock options |
399 | — | (247 | ) | ||||||||
| Restricted stock awards |
87 | — | (33 | ) | ||||||||
| Purchase of shares for treasury |
— | — | (205 | ) | ||||||||
| Conversion of Class B to Common |
25 | (25 | ) | — | ||||||||
| December 31, 2010 Balance |
33,559 | 1,086 | (2,319 | ) | ||||||||
Stock awards for 5,600 and 17,325 shares were cancelled in 2010 and 2009. There were no stock award cancellations in 2008.
FS-25
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Other Comprehensive Earnings
The components of accumulated other comprehensive earnings are as follows (in thousands):
| Currency Translation Adjustments |
Unrealized Gain (Loss) on Available- for-Sale Securities |
Defined Benefit Plans |
Unrealized Gain (Loss) on Derivative Financial Instruments |
Total | ||||||||||||||||
| Balance at January 1, 2008 |
$ | 179,053 | $ | 701 | $ | (12,239 | ) | $ | (2,546 | ) | $ | 164,969 | ||||||||
| Foreign currency translation adjustments |
(124,361 | ) | (124,361 | ) | ||||||||||||||||
| Unrealized loss on available for sale securities |
(113 | ) | (113 | ) | ||||||||||||||||
| Deferred tax asset relating to unrealized gain on available for sale securities |
40 | 40 | ||||||||||||||||||
| Valuation reserve reduction relating to unrealized loss on available for sale securities |
(40 | ) | (40 | ) | ||||||||||||||||
| Amortization of prior service costs and unrecognized losses |
2,513 | 2,513 | ||||||||||||||||||
| Plan amendment giving rise to prior service credit |
12,455 | 12,455 | ||||||||||||||||||
| Amounts arising during the year, primarily due to the addition of new participants |
(4,287 | ) | (4,287 | ) | ||||||||||||||||
| Deferred tax expense resulting from amortization of prior service costs and unrecognized losses, prior service credit and other amounts arising during the year |
(3,738 | ) | (3,738 | ) | ||||||||||||||||
| Valuation reserve reduction resulting from amortization of prior service costs and unrecognized losses, prior service credit and other amounts arising during the year |
3,738 | 3,738 | ||||||||||||||||||
| Current period unrealized loss on cash flow hedges, net of reclassifications |
(470 | ) | (470 | ) | ||||||||||||||||
| Deferred tax benefits relating to unrealized loss on derivative financial instruments |
83 | 83 | ||||||||||||||||||
| Balance at December 31, 2008 |
$ | 54,692 | $ | 588 | $ | (1,558 | ) | $ | (2,933 | ) | $ | 50,789 | ||||||||
| Foreign currency translation adjustments |
119,453 | 119,453 | ||||||||||||||||||
| Unrealized gain on available for sale securities |
96 | 96 | ||||||||||||||||||
| Deferred tax liability relating to unrealized loss on available for sale securities |
(34 | ) | (34 | ) | ||||||||||||||||
| Valuation reserve reduction relating to unrealized loss on available for sale securities |
34 | 34 | ||||||||||||||||||
| Defined Benefit Plans: |
||||||||||||||||||||
| Amortization of prior service costs and unrecognized losses |
537 | 537 | ||||||||||||||||||
| Deferred tax expense resulting from amortization of prior service costs and unrecognized losses, prior service credit and other amounts arising during the year |
(188 | ) | (188 | ) | ||||||||||||||||
| Valuation reserve reduction resulting from amortization of prior service costs and unrecognized losses, prior service credit and other amounts arising during the year |
188 | 188 | ||||||||||||||||||
| Current period unrealized gain on cash flow hedges, net of reclassifications |
3,360 | 3,360 | ||||||||||||||||||
| Deferred tax loss relating to unrealized loss on derivative financial instruments |
(31 | ) | (31 | ) | ||||||||||||||||
| Balance at December 31, 2009 |
$ | 174,145 | $ | 684 | $ | (1,021 | ) | $ | 396 | $ | 174,204 | |||||||||
| Foreign currency translation adjustments |
(59,823 | ) | (59,823 | ) | ||||||||||||||||
| Unrealized loss on available for sale securities |
(684 | ) | (684 | ) | ||||||||||||||||
| Deferred tax liability relating to unrealized gain on available for sale securities |
239 | 239 | ||||||||||||||||||
| Valuation reserve reduction relating to unrealized gain on available for sale securities |
(239 | ) | (239 | ) | ||||||||||||||||
| Defined Benefit Plans: |
||||||||||||||||||||
| Amortization of prior service costs and unrecognized losses |
549 | 549 | ||||||||||||||||||
| Amounts arising during the year, primarily due to the addition of new participants |
(1,860 | ) | (1,860 | ) | ||||||||||||||||
| Deferred tax adjustment resulting from Defined benefit plan amortization of prior service costs and unrecognized losses |
459 | 459 | ||||||||||||||||||
| Valuation reserve increase resulting from amortization of prior service costs, unrecognized losses and other adjustments related to Defined benefit plans |
(459 | ) | (459 | ) | ||||||||||||||||
| Current period unrealized gain on cash flow hedges, net of reclassifications |
273 | 273 | ||||||||||||||||||
| Deferred tax loss relating to unrealized gain on derivative financial instruments |
(28 | ) | (28 | ) | ||||||||||||||||
| Balance at December 31, 2010 |
$ | 114,322 | $ | — | $ | (2,332 | ) | $ | 641 | $ | 112,631 | |||||||||
FS-26
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Other Comprehensive Earnings—Continued
A $2,803,000 net gain in 2010 and net losses of $3,158,000 in 2009 and $26,000 in 2008 were reclassified into earnings related to derivative instruments designated and qualifying as cash flow hedges.
Charges Related to Restructuring Activities
On July 28, 2005, the company announced multi-year cost reductions and profit improvement actions, which included: reducing global headcount, outsourcing improvements utilizing the company’s China manufacturing capability and third parties, shifting substantial resources from product development to manufacturing cost reduction activities and product rationalization, reducing freight exposure through freight auctions and changing the freight policy, general expense reductions and exiting four facilities. The restructuring was necessitated by the continued decline in reimbursement by the U.S. government as well as similar reimbursement pressures abroad and continued pricing pressures faced by the company as a result of outsourcing by competitors to lower cost locations.
The company’s previous restructuring activities concluded in the fourth quarter of 2009 thus no additional charges were incurred in 2010. The company did record restructuring charges of $4,804,000, $4,766,000, $11,408,000 and $21,250,000 in 2009, 2008, 2007 and 2006, respectively, of which $298,000, $1,817,000, $1,817,000, and $3,973,000, respectively is recorded in cost of products sold as it relates to inventory markdowns. There have been no material changes in accrued balances related to the charge, either as a result of revisions in the plan or changes in estimates. A progression by reporting segment of the accruals recorded as a result of the restructuring is as follows (in thousands):
| Severance | Product Line Discontinuance |
Contract Terminations |
Other | Total | ||||||||||||||||
| December 31, 2009 Balance |
||||||||||||||||||||
| NA/HME |
46 | 1 | 23 | — | 70 | |||||||||||||||
| IPG |
5 | — | — | — | 5 | |||||||||||||||
| Europe |
816 | — | — | 343 | 1,159 | |||||||||||||||
| Asia/Pacific |
42 | — | — | — | 42 | |||||||||||||||
| Total |
$ | 909 | $ | 1 | $ | 23 | $ | 343 | $ | 1,276 | ||||||||||
| Payments |
||||||||||||||||||||
| NA/HME |
(46 | ) | (1 | ) | (23 | ) | — | (70 | ) | |||||||||||
| IPG |
(5 | ) | — | — | — | (5 | ) | |||||||||||||
| Europe |
(816 | ) | — | — | (343 | ) | (1,159 | ) | ||||||||||||
| Asia/Pacific |
(42 | ) | — | — | — | (42 | ) | |||||||||||||
| Total |
$ | (909 | ) | $ | (1 | ) | $ | (23 | ) | $ | (343 | ) | $ | (1,276 | ) | |||||
| December 31, 2010 Balance |
||||||||||||||||||||
| NA/HME |
— | — | — | — | — | |||||||||||||||
| ISG |
— | — | — | — | — | |||||||||||||||
| Europe |
— | — | — | — | — | |||||||||||||||
| Asia/Pacific |
— | — | — | — | — | |||||||||||||||
| Total |
$ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||
FS-27
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Income Taxes
Earnings (loss) before income taxes consist of the following (in thousands):
| 2010 | 2009 | 2008 | ||||||||||
| Domestic |
$ | (16,115 | ) | $ | (797 | ) | $ | (10,138 | ) | |||
| Foreign |
54,156 | 48,076 | 57,945 | |||||||||
| $ | 38,041 | $ | 47,279 | $ | 47,807 | |||||||
The company has provided for income taxes (benefits) as follows (in thousands):
| 2010 | 2009 | 2008 | ||||||||||
| Current: |
||||||||||||
| Federal |
$ | 4,749 | $ | (8,310 | ) | $ | 560 | |||||
| State |
689 | 1,775 | (600 | ) | ||||||||
| Foreign |
9,729 | 10,850 | 11,570 | |||||||||
| 15,167 | 4,315 | 11,530 | ||||||||||
| Deferred: |
||||||||||||
| Federal |
(1,696 | ) | — | 190 | ||||||||
| Foreign |
(771 | ) | 1,785 | 1,230 | ||||||||
| (2,467 | ) | 1,785 | 1,420 | |||||||||
| Income Taxes |
$ | 12,700 | $ | 6,100 | $ | 12,950 | ||||||
Included in the 2009 Federal current tax benefit is a benefit of $7,750,000 resulting from the carryback of the 2008 Federal domestic net operating loss as a result of the Worker, Homeownership and Business Assistance Act of 2009, which became effective in November of 2009. The deferred tax asset previously recorded by the company, related to the loss carryforward, was fully offset by a tax valuation allowance.
A reconciliation to the effective income tax rate from the federal statutory rate is as follows:
| 2010 | 2009 | 2008 | ||||||||||
| Statutory federal income tax rate |
35.0 | % | 35.0 | % | 35.0 | % | ||||||
| State and local income taxes, net of federal income tax benefit |
1.2 | 2.4 | (0.8 | ) | ||||||||
| Tax credits |
(41.1 | ) | (146.4 | ) | (3.0 | ) | ||||||
| Foreign taxes at less than the federal statutory rate excluding valuation allowances |
(24.4 | ) | (12.2 | ) | (15.9 | ) | ||||||
| Federal and foreign valuation allowance |
4.6 | 13.3 | 9.3 | |||||||||
| Non-deductible extinguishment and debt finance costs |
8.5 | — | — | |||||||||
| Withholding taxes |
(0.4 | ) | 2.4 | 1.6 | ||||||||
| Compensation |
(0.3 | ) | 0.6 | 0.7 | ||||||||
| Dividends |
54.8 | 129.3 | 4.0 | |||||||||
| Life insurance |
(1.1 | ) | (1.0 | ) | 2.3 | |||||||
| Foreign branch activity |
(3.4 | ) | (5.2 | ) | (7.3 | ) | ||||||
| Uncertain tax positions |
(1.7 | ) | (2.5 | ) | (0.4 | ) | ||||||
| Other, net |
1.7 | (2.8 | ) | 1.6 | ||||||||
| 33.4 | % | 12.9 | % | 27.1 | % | |||||||
FS-28
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Income Taxes—Continued
At December 31, 2010, total deferred tax assets were $108,850,000, total deferred tax liabilities were $53,650,000 and the tax valuation allowance total was $81,981,000 for a net deferred income tax liability of $26,781,000 compared to total deferred tax assets of $96,495,000, total deferred tax liabilities of $61,331,000 and a tax valuation allowance total of $65,050,000 for a net deferred income tax liability of $29,886,000 at December 31, 2009. Significant components of deferred income tax assets and liabilities at December 31, 2010 and 2009 are as follows (in thousands):
| 2010 | 2009 | |||||||
| Current deferred income tax assets (liabilities), net: |
||||||||
| Loss carryforwards |
$ | 5,853 | $ | 907 | ||||
| Bad debt |
9,398 | 8,657 | ||||||
| Warranty |
4,338 | 5,167 | ||||||
| State and local taxes |
2,699 | 2,628 | ||||||
| Other accrued expenses and reserves |
5,535 | 1,932 | ||||||
| Inventory |
2,742 | 3,984 | ||||||
| Compensation and benefits |
1,182 | 2,089 | ||||||
| Product liability |
292 | 292 | ||||||
| Valuation allowance |
(21,657 | ) | (23,229 | ) | ||||
| Other, net |
(4,604 | ) | (2,037 | ) | ||||
| $ | 5,778 | $ | 390 | |||||
| Long-term deferred income tax assets (liabilities), net: |
||||||||
| Goodwill & intangibles |
(24,478 | ) | (27,176 | ) | ||||
| Convertible debt |
(8,798 | ) | (16,895 | ) | ||||
| Fixed assets |
(15,770 | ) | (15,223 | ) | ||||
| Compensation and benefits |
13,416 | 12,300 | ||||||
| Loss and credit carryforwards |
45,519 | 44,116 | ||||||
| Product liability |
4,428 | 4,203 | ||||||
| State and local taxes |
9,480 | 6,559 | ||||||
| Valuation allowance |
(60,324 | ) | (41,821 | ) | ||||
| Other, net |
3,968 | 3,661 | ||||||
| $ | (32,559 | ) | $ | (30,276 | ) | |||
| Net Deferred Income Taxes |
$ | (26,781 | ) | $ | (29,886 | ) | ||
The company recorded a valuation allowance for its domestic net deferred tax assets due to a domestic loss recognized in each year from 2006 through 2009 and based upon near term domestic projections. For 2010 the company estimates a domestic current tax return liability of approximately $3,200,000 and has recorded a deferred tax asset equal to this amount. In addition, during 2007 through 2010, the company also recorded valuation allowances for certain foreign country net deferred tax assets where recent performance results in a three year cumulative loss and near term projections do not warrant substantial positive evidence to overcome the past losses. The company made net payments for income taxes of $2,600,000, $12,340,000, and $10,564,000 during the years ended December 31, 2010, 2009 and 2008, respectively.
At December 31, 2010, the company had foreign tax loss carryforwards of approximately $42,385,000 of which $26,770,000 are non-expiring, $5,868,000 expire in 2026, and $9,747,000 expire in 2027, of which $24,796,000 are offset by valuation allowances. At December 31, 2010 the company also had a $12,324,000 domestic capital loss carryforward of which $8,526,000 expires in 2011 and $3,798,000 expires in 2012, and
FS-29
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Income Taxes—Continued
$380,000,000 of domestic state and local tax loss carryforwards, of which $201,000,000 expire between 2011 and 2014, $91,000,000 expire between 2015 and 2024 and $88,000,000 expire after 2024. The company has domestic federal tax credit carryforwards of $35,075,000 of which $12,953,000 expire between 2014 and 2018 and $21,664,000 expire between 2019 and 2029 and $458,000 is indefinite.
As of December 31, 2010 and 2009, the company had a liability for uncertain tax positions, excluding interest and penalties of $3,420,000 and $5,770,000, respectively. The company does not believe there will be a material change in its unrecognized tax positions over the next twelve months.
The total liabilities associated with unrecognized tax benefits that, if recognized, would impact the effective tax rates were $3,420,000 and $5,770,000 at December 31, 2010 and 2009, respectively.
A reconciliation of the beginning and ending balance of unrecognized tax benefits is as follows (in thousands):
| 2010 | 2009 | |||||||
| Balance at beginning of year |
$ | 6,710 | $ | 6,400 | ||||
| Additions to: |
||||||||
| Positions taken during the current year |
1,400 | 1,130 | ||||||
| Positions taken during a prior year |
265 | 2,340 | ||||||
| Deductions due to: |
||||||||
| Exchange rate impact |
(65 | ) | 280 | |||||
| Positions taken during a prior year |
(15 | ) | (95 | ) | ||||
| Settlements with taxing authorities |
(3,185 | ) | (2,365 | ) | ||||
| Lapse of statute of limitations |
(610 | ) | (980 | ) | ||||
| Balance at end of year |
$ | 4,500 | $ | 6,710 | ||||
The company recognizes interest and penalties associated with uncertain tax positions in income tax expense. During 2010, 2009 and 2008 the benefit for interest and penalties was $1,150,000, $490,000 and $155,000, respectively. The Company had approximately $740,000 and $2,035,000 of accrued interest and penalties as of December 31, 2010 and 2009, respectively.
The company and its subsidiaries file income tax returns in the U.S. and certain foreign jurisdictions. The company is subject to U.S. federal income tax examinations for calendar year 2009 and 2010, and is subject to various U.S. state income tax examinations for 2005 to 2010. With regards to foreign income tax jurisdictions, the company is generally subject to examinations for the periods 2005 to 2010.
FS-30
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Net Earnings Per Common Share
The following table sets forth the computation of basic and diluted net earnings per common share.
| 2010 | 2009 | 2008 | ||||||||||
| (In thousands except per share data) | ||||||||||||
| Basic |
||||||||||||
| Average common shares outstanding |
32,393 | 31,969 | 31,902 | |||||||||
| Net earnings |
$ | 25,341 | $ | 41,179 | $ | 34,857 | ||||||
| Net earnings per common share |
$ | 0.78 | $ | 1.29 | $ | 1.09 | ||||||
| Diluted |
||||||||||||
| Average common shares outstanding |
32,393 | 31,969 | 31,902 | |||||||||
| Shares related to convertible debt |
163 | — | — | |||||||||
| Stock options and awards |
138 | 27 | 51 | |||||||||
| Average common shares assuming dilution |
32,694 | 31,996 | 31,953 | |||||||||
| Net earnings |
$ | 25,341 | $ | 41,179 | $ | 34,857 | ||||||
| Net earnings per common share |
$ | 0.78 | $ | 1.29 | $ | 1.09 | ||||||
At December 31, 2010, 2009, and 2008, 2,396,061, 4,230,630 and 4,337,838 shares associated with stock options, respectively were excluded from the average common shares assuming dilution, as they were anti-dilutive. At December 31, 2010, the majority of the anti-dilutive shares were granted at an exercise price of $41.87, which was higher than the average fair market value price of $25.82 for 2010. In 2009, the majority of the anti-dilutive shares were granted at an exercise price of $41.87, which was higher than the average fair market value price of $19.42 for 2009. In 2008, the majority of the anti-dilutive shares were granted at an exercise price of $25.79, which was higher than the average fair market value price of $20.99 for 2008. Shares necessary to settle a conversion spread on the convertible notes were included in the common shares assuming dilution as the average market price of the company stock for 2010 did exceed the conversion price, which was not the case in 2009 and 2008.
Concentration of Credit Risk
The company manufactures and distributes durable medical equipment and supplies to the home health care, retail and extended care markets. The company performs credit evaluations of its customers’ financial condition. In December 2000, Invacare entered into an agreement with De Lage Landen, Inc. (“DLL”), a third party financing company, to provide the majority of future lease financing to Invacare’s North America customers. The DLL agreement provides for direct leasing between DLL and the Invacare customer. The company retains a recourse obligation of $25,829,000 at December 31, 2010 to DLL for events of default under the contracts, which total $69,430,000 at December 31, 2010. Guarantees, ASC 460, requires the company to record a guarantee liability as it relates to the limited recourse obligation. As such, the company has recorded a liability of $655,000 for this guarantee obligation within accrued expenses. The company monitors the collections status of these contracts and has provided amounts for estimated losses in its allowances for doubtful accounts in accordance with Receivables, ASC 310-10-05-4. Credit losses are provided for in the financial statements.
Substantially all of the company’s receivables are due from health care, medical equipment providers and long term care facilities located throughout the United States, Australia, Canada, New Zealand and Europe. A significant portion of products sold to dealers, both foreign and domestic, is ultimately funded through government reimbursement programs such as Medicare and Medicaid. In addition, the company has also seen a significant shift in reimbursement to customers from managed care entities. As a consequence, changes in these programs can have an adverse impact on dealer liquidity and profitability. In addition, reimbursement guidelines
FS-31
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Concentration of Credit Risk—Continued
in the home health care industry have a substantial impact on the nature and type of equipment an end user can obtain as well as the timing of reimbursement and, thus, affect the product mix, pricing and payment patterns of the company’s customers.
The company’s top 10 customers accounted for approximately 12.4% of 2010 net sales. The loss of business of one or more of these customers may have a significant impact on the company, although no single customer accounted for more than 3.3% of the company’s 2010 net sales. Providers who are part of a buying group generally make individual purchasing decisions and are invoiced directly by the company.
Derivatives
ASC 815 requires companies to recognize all derivative instruments in the consolidated balance sheet as either assets or liabilities at fair value. The accounting for changes in fair value of a derivative is dependent upon whether or not the derivative has been designated and qualifies for hedge accounting treatment and the type of hedging relationship. For derivatives designated and qualifying as hedging instruments, the company must designate the hedging instrument, based upon the exposure being hedged, as a fair value hedge, cash flow hedge, or a hedge of a net investment in a foreign operation.
Cash Flow Hedging Strategy
The company uses derivative instruments in an attempt to manage its exposure to foreign currency exchange risk and interest rate risk. Foreign forward exchange contracts are used to manage the price risk associated with forecasted sales denominated in foreign currencies and the price risk associated with forecasted purchases of inventory over the next twelve months. Interest rate swaps are, at times, utilized to manage interest rate risk associated with the company’s fixed and floating-rate borrowings.
The company recognizes its derivative instruments as assets or liabilities in the consolidated balance sheet measured at fair value. A majority of the company’s derivative instruments are designated and qualify as cash flow hedges. Accordingly, the effective portion of the gain or loss on the derivative instrument is reported as a component of other comprehensive income and reclassified into earnings in the same period or periods during which the hedged transaction affects earnings. The remaining gain or loss on the derivative instrument in excess of the cumulative change in the fair value of the hedged item, if any, is recognized in current earnings during the period of change.
The company was not a party to any interest rate swap agreements during 2010. During 2009, the company was a party to interest rate swap agreements that qualified as cash flow hedges and effectively converted floating-rate debt to fixed-rate debt, so the company could avoid the risk of changes in market interest rates. The gains or losses on interest rate swaps are reflected in interest expense on the consolidated statement of earnings.
To protect against increases/decreases in forecasted foreign currency cash flows resulting from inventory purchases/sales over the next year, the company utilizes foreign currency forward contracts to hedge portions of its forecasted purchases/sales denominated in foreign currencies. The gains and losses are included in cost of products sold and selling, general and administrative expenses on the consolidated statement of earnings. If it is later determined that a hedged forecasted transaction is unlikely to occur, any prospective gains or losses on the forward contracts would be recognized in earnings. The company does not expect any material amount of hedge ineffectiveness related to forward contract cash flow hedges during the next twelve months.
The company has historically not recognized any material amount of ineffectiveness related to forward contract cash flow hedges because the company generally limits it hedges to between 60% and 90% of total
FS-32
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Derivatives—Continued
forecasted transactions for a given entity’s exposure to currency rate changes and the transactions hedged are recurring in nature. Furthermore, the majority of the hedged transactions are related to intercompany sales and purchases for which settlement occurs on a specific day each month. Forward contracts with a total notional amount in USD of $173,337,000 and $180,664,000 matured during the twelve months ended December 31, 2010 and 2009, respectively. Foreign exchange forward contracts qualifying and designated for hedge accounting treatment were as follows (in thousands USD):
| December 31, 2010 | December 31, 2009 | |||||||||||||||
| Notional Amount |
Unrealized Net Gain (Loss) |
Notional Amount |
Unrealized Net Gain (Loss) |
|||||||||||||
| USD / AUD |
$ | 3,072 | $ | (223 | ) | $ | 3,294 | $ | (41 | ) | ||||||
| USD / CAD |
32,974 | (14 | ) | 49,345 | 202 | |||||||||||
| USD / EUR |
32,419 | 927 | 22,119 | (526 | ) | |||||||||||
| USD / GBP |
4,212 | 86 | 3,640 | (72 | ) | |||||||||||
| USD / NZD |
9,577 | 202 | 8,286 | 130 | ||||||||||||
| USD / SEK |
10,395 | 95 | 8,965 | (100 | ) | |||||||||||
| USD / MXP |
— | — | 2,520 | 217 | ||||||||||||
| EUR / CHF |
8,768 | 54 | 2,755 | (9 | ) | |||||||||||
| EUR / GBP |
18,068 | (577 | ) | 22,258 | 27 | |||||||||||
| EUR / SEK |
8,045 | 92 | 3,800 | 15 | ||||||||||||
| EUR / NZD |
2,630 | 5 | 8,029 | 359 | ||||||||||||
| GBP / CHF |
770 | (3 | ) | 501 | 14 | |||||||||||
| GBP / SEK |
2,014 | (43 | ) | 2,169 | 37 | |||||||||||
| GBP / DKK |
1,016 | (27 | ) | 765 | 17 | |||||||||||
| CHF / SEK |
6,937 | (3 | ) | — | — | |||||||||||
| DKK / CHF |
514 | 1 | — | — | ||||||||||||
| DKK / SEK |
1,465 | 18 | 7,439 | 52 | ||||||||||||
| DKK / NOK |
— | — | 2,236 | 19 | ||||||||||||
| NOK / EUR |
— | — | 342 | 6 | ||||||||||||
| NOK / CHF |
— | — | 592 | (9 | ) | |||||||||||
| NOK / SEK |
— | — | 1,190 | (21 | ) | |||||||||||
| $ | 142,876 | $ | 590 | $ | 150,245 | $ | 317 | |||||||||
Fair Value Hedging Strategy
In 2010 and 2009, the company did not utilize any derivatives designated as fair value hedges. However, the company has in the past utilized fair value hedges in the form of forward contracts to manage the foreign exchange risk associated with certain firm commitments and has entered into interest rate swaps to effectively convert fixed-rate debt to floating-rate debt in an attempt to avoid paying higher than market interest rates. For derivative instruments designated and qualifying as fair value hedges, the gain or loss on the derivative instrument as well as the offsetting gain or loss on the hedged item associated with the hedged risk are recognized in the same line item associated with the hedged item in earnings.
Derivatives Not Qualifying or Designated for Hedge Accounting Treatment
The company utilizes foreign currency forward or option contracts that do not qualify for hedge accounting treatment in an attempt to manage the risk associated with the conversion of earnings in foreign currencies into U.S. Dollars. While these derivative instruments do not qualify for hedge accounting treatment in accordance with ASC 815, these derivatives do provide the company with a means to manage the risk associated with currency translation. These instruments are recorded at fair value in the consolidated balance sheet and any gains
FS-33
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Derivatives—Continued
or losses are recorded as part of earnings in the current period. A gain of $28,000 and a loss of $68,000 were recorded by the company for the year ended December 31, 2010 and 2009, respectively, related to derivatives not qualifying for hedge accounting treatment.
The company also utilizes foreign currency forward contracts that are not designated as hedges in accordance with ASC 815. These contracts are entered into to eliminate the risk associated with the settlement of short-term intercompany trading receivables and payables between Invacare Corporation and its foreign subsidiaries. The currency forward contracts are entered into at the same time as the intercompany receivables or payables are created so that upon settlement, the gain/loss on the settlement is offset by the gain/loss on the foreign currency forward contract. No material net gain or loss was realized by the company in 2010 or 2009 related to these forward contracts and the associated short-term intercompany trading receivables and payables.
Foreign exchange forward contracts not qualifying or designated for hedge accounting treatment entered into in 2010 and 2009, respectively, and outstanding were as follows (in thousands USD):
| December 31, 2010 | December 31, 2009 | |||||||||||||||
| Notional Amount |
Gain (Loss) |
Notional Amount |
Gain (Loss) |
|||||||||||||
| CAD / USD |
$ | 14,636 | $ | 337 | $ | 2,194 | $ | (3 | ) | |||||||
| EUR / USD |
1,394 | 28 | — | — | ||||||||||||
| CHF / USD |
— | — | 1,102 | (39 | ) | |||||||||||
| DKK / USD |
— | — | 7,580 | (77 | ) | |||||||||||
| GBP / USD |
— | — | 3,304 | (73 | ) | |||||||||||
| NZD / USD |
— | — | 1,756 | 59 | ||||||||||||
| SEK / USD |
— | — | 9,899 | (126 | ) | |||||||||||
| EUR / NZD |
— | — | 7,457 | (324 | ) | |||||||||||
| $ | 16,030 | $ | 365 | $ | 33,292 | $ | (583 | ) | ||||||||
The fair values of the company’s derivative instruments were as follows (in thousands):
| December 31, 2010 | December 31, 2009 | |||||||||||||||
| Assets | Liabilities | Assets | Liabilities | |||||||||||||
| Derivatives designated as hedging instruments under ASC 815 |
||||||||||||||||
| Foreign currency forward contracts |
$ | 2,518 | $ | 1,928 | $ | 1,815 | $ | 1,498 | ||||||||
| Derivatives not designated as hedging instruments under ASC 815 |
||||||||||||||||
| Foreign currency forward contracts |
366 | 1 | 92 | 675 | ||||||||||||
| Total derivatives |
$ | 2,884 | $ | 1,929 | $ | 1,907 | $ | 2,173 | ||||||||
The fair values of the company’s foreign currency forward assets and liabilities are included in Other Current Assets and Accrued Expenses, respectively in the Consolidated Balance Sheets.
FS-34
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Derivatives—Continued
The effect of derivative instruments on the Statement of Earnings and Other Comprehensive Income (OCI) was as follows (in thousands):
| Derivatives in ASC 815 cash flow hedge |
Amount of Gain (Loss) Recognized in OCI on Derivatives (Effective Portion) |
Amount of Gain (Loss) Reclassified from Accumulated OCI into Income (Effective Portion) |
Amount of Gain (Loss) Recognized in Income on Derivatives (Ineffective Portion and Amount Excluded from Effectiveness Testing) |
|||||||||
| Year ended December 31, 2010 |
||||||||||||
| Foreign currency forward contracts |
$ | (2,530 | ) | $ | 2,803 | $ | (134 | ) | ||||
| Interest rate swap contracts |
— | — | — | |||||||||
| $ | (2,530 | ) | $ | 2,803 | $ | (134 | ) | |||||
| Year ended December 31, 2009 |
||||||||||||
| Foreign currency forward contracts |
$ | 962 | $ | (339 | ) | $ | — | |||||
| Interest rate swap contracts |
5,556 | (2,819 | ) | — | ||||||||
| $ | 6,518 | $ | (3,158 | ) | $ | — | ||||||
| Derivatives not designated as hedging |
Amount of Gain Recognized in Income on Derivatives |
|||||||||||
| Year ended December 31, 2010 |
||||||||||||
| Foreign currency forward contracts |
$ | 3,800 | ||||||||||
| Year ended December 31, 2009 |
||||||||||||
| Foreign currency forward contracts |
$ | 2,899 | ||||||||||
The gains or losses recognized as the result of the settlement of cash flow hedge foreign currency forward contracts are recognized in net sales for hedges of inventory sales or cost of product sold for hedges of inventory purchases. In 2010, net sales were increased by $1,605,000 and cost of product sold was decreased by $1,198,000 for a net realized gain of $2,803,000. In 2009, net sales were increased by $3,093,000 and cost of product sold was increased by $3,432,000 for a net realized loss of $339,000 compared to a net loss of $26,000 in 2008.
The company recognized net losses of $2,819,000 and $2,684,000 in 2009 and 2008, respectively related to interest rate swap agreements which are reflected in interest expense on the consolidated statement of earnings. Gains of $3,800,000 and $2,899,000 were recognized in selling, general and administrative (SG&A) expenses in 2010 and 2009, respectively, on foreign currency forward contracts not designated as hedging instruments which were offset by losses of comparable amounts also recorded in SG&A expenses on the intercompany trade payables for which the derivatives were entered into to offset.
Fair Values of Financial Instruments
Pursuant to ASC 820, the inputs used to derive the fair value of assets and liabilities are analyzed and assigned a level I, II or III priority, with level I being the highest and level III being the lowest in the hierarchy. Level I inputs are quoted prices in active markets for identical assets or liabilities. Level II inputs are quoted prices for similar assets or liabilities in active markets: quoted prices for identical or similar instruments in markets that are not active; and model-derived valuations in which all significant inputs are observable in active markets. Level III inputs are based on valuations derived from valuation techniques in which one or more significant inputs are unobservable.
FS-35
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Fair Values of Financial Instruments—Continued
The following table provides a summary of the company’s assets and liabilities that are measured on a recurring basis (in thousands).
| Basis for Fair Value Measurements at Reporting Date | ||||||||||||||||
| Quoted Prices in Active Markets for Identical Assets / (Liabilities) |
Significant Other Observable Inputs |
Significant Other Unobservable Inputs |
||||||||||||||
| Total | Level I | Level II | Level III | |||||||||||||
| December 31, 2010: |
||||||||||||||||
| Forward Exchange Contracts—net |
$ | 955 | — | $ | 955 | — | ||||||||||
| December 31, 2009: |
||||||||||||||||
| Forward Exchange Contracts—net |
(266 | ) | — | (266 | ) | — | ||||||||||
Forward Contracts: The company operates internationally and as a result is exposed to foreign currency fluctuations. Specifically, the exposure includes intercompany loans and third party sales or payments. In an attempt to reduce this exposure, foreign currency forward contracts are utilized and accounted for as hedging instruments. The forward contracts are used to hedge the following currencies: AUD, CAD, CHF, CNY, DKK, EUR, GBP, MXP, NOK, NZD, SEK and USD. The company does not use derivative financial instruments for speculative purposes. Fair values for the company’s foreign exchange forward contracts are based on quoted market prices for contracts with similar maturities.
The gains and losses that result from the majority of the forward contracts are deferred and recognized when the offsetting gains and losses for the identified transactions are recognized. The company recognized a net gain of $2,803,000 in 2010, a net loss of $339,000 in 2009 and a net loss of $26,000 in 2008 on ASC 815 designated derivatives. Gains or losses recognized as the result of the settlement of forward contracts are recognized in cost of products sold for hedges of inventory transactions, sales for hedges of forecasted sales or selling, general and administrative expenses for other hedged transactions. The company’s forward contracts are included in Other Current Assets or Accrued Expenses in the Consolidated Balance Sheets.
The carrying amounts and fair values of the company’s financial instruments at December 31, 2010 and 2009 are as follows (in thousands):
| 2010 | 2009 | |||||||||||||||
| Carrying Value |
Fair Value | Carrying Value |
Fair Value | |||||||||||||
| Cash and cash equivalents |
$ | 48,462 | $ | 48,462 | $ | 37,501 | $ | 37,501 | ||||||||
| Other investments |
1,588 | 1,588 | 1,521 | 1,521 | ||||||||||||
| Installment receivables, net of reserves |
5,672 | 5,672 | 7,106 | 7,106 | ||||||||||||
| Long-term debt (including current maturities of long-term debt) |
(246,064 | ) | (264,382 | ) | (273,325 | ) | (293,133 | ) | ||||||||
| Forward contracts in Other Current Assets |
2,884 | 2,884 | 1,907 | 1,907 | ||||||||||||
| Forward contracts in Accrued Expenses |
(1,929 | ) | (1,929 | ) | (2,173 | ) | (2,173 | ) | ||||||||
The company, in estimating its fair value disclosures for financial instruments, used the following methods and assumptions:
Cash, cash equivalents: The carrying amount reported in the balance sheet for cash, cash equivalents equals its fair value.
FS-36
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Fair Values of Financial Instruments—Continued
Installment receivables: The carrying amount reported in the balance sheet for installment receivables approximates its fair value. The interest rates associated with these receivables have not varied significantly since inception. Management believes that after consideration of the credit risk, the net book value of the installment receivables approximates market value.
Long-term debt: Fair values for the company’s senior notes and convertible debt are based on quoted market prices as of the end of the year, while the revolving credit facility fair values are based upon the company’s estimate of the market for similar borrowing arrangements.
Other investments: The company has made other investments in limited partnerships and non-marketable equity securities, which are accounted for using the cost method, adjusted for any estimated declines in value. These investments were acquired in private placements and there are no quoted market prices or stated rates of return and the company does not have the ability to easily sell these investments. The company completed an evaluation of the residual value related to these investments in the fourth quarter of 2010 and recognized an immaterial loss. In the fourth quarter 2009, the company recognized impairment charges totaling $6,713,000 pre-tax, which is included in the All Other segment, as a result of an evaluation of the residual value related to these investments which considered the weakening in the commercial real estate market as well as the redemption of one of the investments for a nominal amount.
Other Intangibles and Goodwill: Under Intangibles—Goodwill and Other, ASC 350, goodwill and intangible assets deemed to have indefinite lives are subject to annual impairment tests. Furthermore, goodwill and other long-lived assets are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. The company completes its annual impairment tests in the fourth quarter of each year. The discount rates used have a significant impact upon the discounted cash flow methodology utilized in the company’s annual impairment testing as higher discount rates decrease the fair value estimates.
The company utilizes a discounted cash flow method model to analyze reporting units for impairment in which the company forecasts income statement and balance sheet amounts based on assumptions regarding future sales growth, profitability, inventory turns, days’ sales outstanding, etc. to forecast future cash flows. The cash flows are discounted using a weighted average cost of capital discount rate where the cost of debt is based on quoted rates for 20-year debt of companies of similar credit risk and the cost of equity is based upon the 20-year treasury rate for the risk free rate, a market risk premium, the industry average beta, a small cap stock adjustment and company specific risk premiums. The assumptions used are based on a market participant’s point of view and yielded a discount rate of 9.59% in 2010 compared to 10.74% in 2009 and 8.90% to 9.90% in 2008.
The company also utilizes an EV (Enterprise Value) to EBITDA (Earnings Before Interest Taxes Depreciation and Amortization) Method to compute the fair value of its reporting units which considers potential acquirers and their EV to EBITDA multiples adjusted by an estimated premium. While more weight is given to the discounted cash flow method, the EV to EBITDA Method does provide corroborative evidence of the reasonableness of the discounted cash flow method results.
While there was no indication of impairment in 2010 related to goodwill, a future potential impairment is possible for any of the company’s reporting units should actual results differ materially from forecasted results used in the valuation analysis. Furthermore, the company’s annual valuation of goodwill can differ materially if the market inputs used to determine the discount rate change significantly. For instance, higher interest rates or greater stock price volatility would increase the discount rate and thus increase the chance of impairment. For example, if the discount rate used were 100 basis points higher for the 2010 impairment analysis, there still would not be any indicator of potential impairment for any of the reporting units.
FS-37
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Fair Values of Financial Instruments—Continued
For purposes of testing intangibles for impairment, the fair value of each unamortized intangible is estimated by forecasting cash flows and discounting those cash flows using appropriate discount rates and using market participant assumptions regarding taxes, impact of contributory assets in the valuation models, etc. The fair values are then compared to the carrying value of the intangible. For amortized intangibles, the forecasted undiscounted cash flows were compared to the carrying value, and if impairment results, the impairment is measured based on the estimated fair value of the intangibles.
As a result of the company’s 2010 intangible impairment review, the company calculated the fair value of an IPG segment indefinite-lived trademark and a NA/HME segment customer list with a remaining life of eight years as each had indicators of impairment, principally net sales less than forecasted. The fair value of the trademark was calculated using a relief from royalty payment methodology which requires applying an estimated market royalty rate to forecasted net sales and discounting the resulting cash flows to determine fair value. The calculated fair value was $3,930,000 compared to a carrying value of $4,266,000 for a resulting impairment charge of $336,000. The fair value of the customer list was calculated using an excess earnings method, using a discounted cash flow model. Estimated cash flow returns to the customer relationship were reduced by the cash flows required to satisfy the return requirements of each of the assets employed with the residual cash flow then discounted to value the customer relationship. The calculated fair value was $500,000 compared to a carrying value of $748,000 for a resulting impairment charge of $248,000. Both fair values were calculated using inputs that are not observable in the market and included management’s own estimates regarding the assumptions that market participants would use and thus these inputs are deemed Level III inputs in regards to the fair value hierarchy.
Business Segments
The company operates in five primary business segments: North America/Home Medical Equipment (NA/HME), Invacare Supply Group (ISG), Institutional Products Group (IPG), Europe and Asia/Pacific.
The NA/HME segment sells each of three primary product lines, which includes: standard, rehab and respiratory products. Invacare Supply Group sells distributed product and the Institutional Products Group sells health care furnishings and accessory products. Europe and Asia/Pacific sell the same product lines. Each business segment sells to the home health care, retail and extended care markets.
The company evaluates performance and allocates resources based on profit or loss from operations before income taxes for each reportable segment. The accounting policies of each segment are the same as those described in the summary of significant accounting policies for the company’s consolidated financial statements. Intersegment sales and transfers are based on the costs to manufacture plus a reasonable profit element. Therefore, intercompany profit or loss on intersegment sales and transfers is not considered in evaluating segment performance except for Asia/Pacific due to its significant intercompany sales volume.
In 2010, management changed how it views segment earnings before taxes and accordingly reclassifications have been made to the company’s segment disclosure of earnings (loss) before income tax amounts for 2009 to be consistent with 2010 presentation. As a result, 2009 earnings before taxes increased for NA/HME by $2,878,000 and the loss before income taxes for All Other increased by $2,878,000. For product sales, All Other was revised to exclude parts sales which have been allocated based on major product categories to which the parts sales relate and prior periods were restated accordingly.
FS-38
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Business Segments—Continued
The information by segment is as follows (in thousands):
| 2010 | 2009 | 2008 | ||||||||||
| Revenues from external customers |
||||||||||||
| North America/HME |
$ | 747,599 | $ | 748,401 | $ | 741,502 | ||||||
| Invacare Supply Group |
297,517 | 280,295 | 265,818 | |||||||||
| Institutional Products Group |
88,261 | 89,423 | 99,662 | |||||||||
| Europe |
506,069 | 503,084 | 553,845 | |||||||||
| Asia/Pacific |
82,635 | 71,933 | 94,867 | |||||||||
| Consolidated |
$ | 1,722,081 | $ | 1,693,136 | $ | 1,755,694 | ||||||
| Intersegment revenues |
||||||||||||
| North America/HME |
$ | 83,316 | $ | 72,273 | $ | 56,826 | ||||||
| Invacare Supply Group |
75 | 232 | 527 | |||||||||
| Institutional Products Group |
5,571 | 2,639 | 2,668 | |||||||||
| Europe |
10,165 | 9,719 | 12,482 | |||||||||
| Asia/Pacific |
33,616 | 31,143 | 31,132 | |||||||||
| Consolidated |
$ | 132,743 | $ | 116,006 | $ | 103,635 | ||||||
| Depreciation and amortization |
||||||||||||
| North America/HME |
$ | 16,514 | $ | 17,905 | $ | 19,478 | ||||||
| Invacare Supply Group |
383 | 403 | 377 | |||||||||
| Institutional Products Group |
1,155 | 1,306 | 1,670 | |||||||||
| Europe |
13,620 | 15,285 | 17,198 | |||||||||
| Asia/Pacific |
4,941 | 5,555 | 4,987 | |||||||||
| All Other(1) |
191 | 108 | 34 | |||||||||
| Consolidated |
$ | 36,804 | $ | 40,562 | $ | 43,744 | ||||||
| Net interest expense (income) |
||||||||||||
| North America/HME |
$ | 12,841 | $ | 26,687 | $ | 25,934 | ||||||
| Invacare Supply Group |
3,058 | 3,153 | 3,531 | |||||||||
| Institutional Products Group |
513 | 2,525 | 3,865 | |||||||||
| Europe |
721 | (1,876 | ) | 6,027 | ||||||||
| Asia/Pacific |
2,790 | 987 | 525 | |||||||||
| Consolidated |
$ | 19,923 | $ | 31,476 | $ | 39,882 | ||||||
| Earnings (loss) before income taxes |
||||||||||||
| North America/HME |
$ | 54,586 | $ | 39,115 | $ | 17,655 | ||||||
| Invacare Supply Group |
7,547 | 5,374 | 2,192 | |||||||||
| Institutional Products Group |
9,258 | 9,213 | 6,725 | |||||||||
| Europe |
39,344 | 34,685 | 44,675 | |||||||||
| Asia/Pacific |
6,754 | 1,639 | 8,705 | |||||||||
| All Other(1) |
(79,448 | ) | (42,747 | ) | (32,145 | ) | ||||||
| Consolidated |
$ | 38,041 | $ | 47,279 | $ | 47,807 | ||||||
FS-39
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Business Segments—Continued
| 2010 | 2009 | 2008 | ||||||||||
| Assets |
||||||||||||
| North America/HME |
$ | 356,678 | $ | 310,404 | $ | 359,364 | ||||||
| Invacare Supply Group |
88,678 | 86,469 | 88,540 | |||||||||
| Institutional Products Group |
45,561 | 45,518 | 33,491 | |||||||||
| Europe |
660,620 | 761,992 | 683,870 | |||||||||
| Asia/Pacific |
92,322 | 90,318 | 90,062 | |||||||||
| All Other(1) |
36,541 | 64,800 | 59,146 | |||||||||
| Consolidated |
$ | 1,280,400 | $ | 1,359,501 | $ | 1,314,473 | ||||||
| Long-lived assets |
||||||||||||
| North America/HME |
$ | 98,651 | $ | 72,527 | $ | 99,709 | ||||||
| Invacare Supply Group |
24,126 | 24,085 | 24,312 | |||||||||
| Institutional Products Group |
32,066 | 31,191 | 28,103 | |||||||||
| Europe |
510,728 | 596,142 | 517,319 | |||||||||
| Asia/Pacific |
52,565 | 50,323 | 43,163 | |||||||||
| All Other(1) |
36,105 | 56,769 | 50,809 | |||||||||
| Consolidated |
$ | 754,241 | $ | 831,037 | $ | 763,415 | ||||||
| Expenditures for assets |
||||||||||||
| North America/HME |
$ | 9,836 | $ | 8,110 | $ | 6,590 | ||||||
| Invacare Supply Group |
404 | 196 | 506 | |||||||||
| Institutional Products Group |
234 | 245 | 962 | |||||||||
| Europe |
4,448 | 5,268 | 6,311 | |||||||||
| Asia/Pacific |
2,224 | 3,433 | 5,567 | |||||||||
| All Other(1) |
207 | 747 | 21 | |||||||||
| Consolidated |
$ | 17,353 | $ | 17,999 | $ | 19,957 | ||||||
| (1) | Consists of un-allocated corporate selling, general and administrative costs and intercompany profits, which do not meet the quantitative criteria for determining reportable segments. In addition, the “All Other” earnings (loss) before income taxes includes loss on debt extinguishment including debt finance charges, interest and fees and impairment charges recognized related to limited partnership investments. |
Net sales by product, are as follows (in thousands):
| 2010 | 2009 | 2008 | ||||||||||
| North America/HME |
||||||||||||
| Standard |
$ | 303,798 | $ | 296,068 | $ | 280,662 | ||||||
| Rehab |
288,756 | 283,214 | 284,793 | |||||||||
| Respiratory |
111,242 | 133,821 | 145,627 | |||||||||
| Other(1) |
43,803 | 35,298 | 30,420 | |||||||||
| $ | 747,599 | $ | 748,401 | $ | 741,502 | |||||||
| Invacare Supply Group |
||||||||||||
| Distributed |
$ | 297,517 | $ | 280,295 | $ | 265,818 | ||||||
| Institutional Products Group |
||||||||||||
| Continuing Care |
$ | 88,261 | $ | 89,423 | $ | 99,662 | ||||||
FS-40
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Business Segments—Continued
| 2010 | 2009 | 2008 | ||||||||||
| Europe |
||||||||||||
| Standard |
$ | 289,577 | $ | 285,253 | $ | 325,218 | ||||||
| Rehab |
183,271 | 185,186 | 196,340 | |||||||||
| Respiratory |
20,493 | 17,137 | 16,901 | |||||||||
| Other(1) |
12,728 | 15,508 | 15,386 | |||||||||
| $ | 506,069 | $ | 503,084 | $ | 553,845 | |||||||
| Asia/Pacific |
||||||||||||
| Rehab |
$ | 38,226 | $ | 31,428 | $ | 39,171 | ||||||
| Standard |
21,216 | 28,363 | 31,695 | |||||||||
| Respiratory |
1,021 | 626 | 1,290 | |||||||||
| Other(1) |
22,172 | 11,516 | 22,711 | |||||||||
| $ | 82,635 | $ | 71,933 | $ | 94,867 | |||||||
| Total Consolidated |
$ | 1,722,081 | $ | 1,693,136 | $ | 1,755,694 | ||||||
| (1) | Includes various services, including repair services, equipment rentals and external contracting. |
No single customer accounted for more than 3.3% of the company’s sales.
Contingencies
In the ordinary course of its business, the company is a defendant in a number of lawsuits, primarily product liability actions in which various plaintiffs seek damages for injuries allegedly caused by defective products. All of the product liability lawsuits have been referred to the company’s captive insurance company and/or excess insurance carriers and generally are contested vigorously. The coverage territory of the company’s insurance is worldwide with the exception of those countries with respect to which, at the time the product is sold for use or at the time a claim is made, the U.S. government has suspended or prohibited diplomatic or trade relations. The amount recorded for identified contingent liabilities is based on estimates. Amounts recorded are reviewed periodically and adjusted to reflect additional technical and legal information that becomes available. Actual costs to be incurred in future periods may vary from the estimates, given the inherent uncertainties in evaluating certain exposures. Subject to the imprecision in estimating future contingent liability costs, the company does not expect that any sum it may have to pay in connection with these matters in excess of the amounts recorded will have a materially adverse effect on its financial position, results of operations or liquidity.
As a medical device manufacturer, the company is subject to extensive government regulation, including numerous laws directed at preventing fraud and abuse and laws regulating reimbursement under various government programs. The marketing, invoicing, documenting and other practices of health care suppliers and manufacturers are all subject to government scrutiny. Violations of law or regulations can result in administrative, civil and criminal penalties and sanctions, including disqualification from Medicare and other reimbursement programs, which could have a material adverse effect on the company’s business. The company has established numerous policies and procedures that the company believes are sufficient to ensure that the company will operate in substantial compliance with these laws and regulations. Further, the FDA regulates virtually all aspects of a medical device’s development, testing, manufacturing, labeling, promotion, distribution and marketing. The company’s failure to comply with the regulatory requirements of the FDA and other applicable U.S. regulatory requirements may subject the company to administrative or judicially imposed sanctions. These sanctions include warning letters, civil penalties, criminal penalties, injunctions, consent decrees, product seizure or detention, product recalls and total or partial suspension of production.
FS-41
Table of Contents
Supplemental Guarantor Information
Effective February 12, 2007, substantially all of the domestic subsidiaries (the “Guarantor Subsidiaries”) of the company became guarantors of the indebtedness of Invacare Corporation under its 4.125% Convertible Senior Subordinated Debentures due 2027 (the “Debentures”) with an original aggregate principal amount of $135,000,000. The majority of the company’s subsidiaries are not guaranteeing the indebtedness of the Debentures (the “Non-Guarantor Subsidiaries”). Each of the Guarantor Subsidiaries has fully and unconditionally guaranteed, on a joint and several basis, to pay principal, premium, and interest related to the Debentures and each of the Guarantor Subsidiaries are directly or indirectly wholly-owned subsidiaries of the company.
Presented below are the consolidating condensed financial statements of Invacare Corporation (Parent), its combined Guarantor Subsidiaries and combined Non-Guarantor Subsidiaries with their investments in subsidiaries accounted for using the equity method. The company does not believe that separate financial statements of the Guarantor Subsidiaries are material to investors and accordingly, separate financial statements and other disclosures related to the Guarantor Subsidiaries are not presented.
FS-42
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Supplemental Guarantor Information—Continued
CONSOLIDATING CONDENSED STATEMENTS OF OPERATIONS
| The Company (Parent) |
Combined Guarantor Subsidiaries |
Combined Non-Guarantor Subsidiaries |
Eliminations | Total | ||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| Year ended December 31, 2010 |
||||||||||||||||||||
| Net sales |
$ | 403,227 | $ | 723,402 | $ | 693,463 | $ | (98,011 | ) | $ | 1,722,081 | |||||||||
| Cost of products sold |
283,859 | 563,837 | 462,776 | (98,032 | ) | 1,212,440 | ||||||||||||||
| Gross Profit |
119,368 | 159,565 | 230,687 | 21 | 509,641 | |||||||||||||||
| Selling, general and administrative expenses |
132,177 | 70,902 | 169,114 | 39,320 | 411,513 | |||||||||||||||
| Loss on debt extinguishment including debt finance charges and associated fees |
40,164 | — | — | — | 40,164 | |||||||||||||||
| Income (loss) from equity investee |
97,602 | 37,438 | (591 | ) | (134,449 | ) | — | |||||||||||||
| Interest expense—net |
16,208 | 729 | 2,986 | — | 19,923 | |||||||||||||||
| Earnings (loss) before Income Taxes |
28,421 | 125,372 | 57,996 | (173,748 | ) | 38,041 | ||||||||||||||
| Income taxes |
3,080 | — | 9,620 | — | 12,700 | |||||||||||||||
| Net Earnings (loss) |
$ | 25,341 | $ | 125,372 | $ | 48,376 | $ | (173,748 | ) | $ | 25,341 | |||||||||
| Year ended December 31, 2009 |
||||||||||||||||||||
| Net sales |
$ | 388,141 | $ | 707,618 | $ | 681,374 | $ | (83,997 | ) | $ | 1,693,136 | |||||||||
| Cost of products sold |
275,089 | 555,503 | 453,464 | (84,114 | ) | 1,199,942 | ||||||||||||||
| Gross Profit |
113,052 | 152,115 | 227,910 | 117 | 493,194 | |||||||||||||||
| Selling, general and administrative expenses |
16,813 | 118,940 | 156,791 | 106,102 | 398,646 | |||||||||||||||
| Charges related to restructuring activities |
301 | 60 | 4,145 | — | 4,506 | |||||||||||||||
| Loss on debt extinguishment including debt finance charges and associated fees |
2,878 | — | — | — | 2,878 | |||||||||||||||
| Asset write-downs to intangibles and investments |
8,409 | — | — | — | 8,409 | |||||||||||||||
| Income (loss) from equity investee |
(22,580 | ) | 25,508 | (13,445 | ) | 10,517 | — | |||||||||||||
| Interest expense (income)—net |
27,021 | (2,897 | ) | 7,352 | — | 31,476 | ||||||||||||||
| Earnings (loss) before Income Taxes |
35,050 | 61,520 | 46,177 | (95,468 | ) | 47,279 | ||||||||||||||
| Income taxes (benefit) |
(6,129 | ) | 99 | 12,130 | — | 6,100 | ||||||||||||||
| Net Earnings (loss) |
$ | 41,179 | $ | 61,421 | $ | 34,047 | $ | (95,468 | ) | $ | 41,179 | |||||||||
| Year ended December 31, 2008 |
||||||||||||||||||||
| Net sales |
$ | 368,574 | $ | 683,773 | $ | 776,405 | $ | (73,058 | ) | $ | 1,755,694 | |||||||||
| Cost of products sold |
274,948 | 547,193 | 517,861 | (73,200 | ) | 1,266,802 | ||||||||||||||
| Gross Profit |
93,626 | 136,580 | 258,544 | 142 | 488,892 | |||||||||||||||
| Selling, general and administrative expenses |
112,554 | 117,195 | 157,639 | 10,866 | 398,254 | |||||||||||||||
| Charge related to restructuring activities |
217 | — | 2,732 | — | 2,949 | |||||||||||||||
| Income (loss) from equity investee |
83,013 | 48,405 | 5,518 | (136,936 | ) | — | ||||||||||||||
| Interest expense (income)—net |
31,173 | (1,065 | ) | 9,774 | — | 39,882 | ||||||||||||||
| Earnings (loss) before Income Taxes |
32,695 | 68,855 | 93,917 | (147,660 | ) | 47,807 | ||||||||||||||
| Income taxes (benefit) |
(2,162 | ) | 194 | 14,918 | — | 12,950 | ||||||||||||||
| Net Earnings (loss) |
$ | 34,857 | $ | 68,661 | $ | 78,999 | $ | (147,660 | ) | $ | 34,857 | |||||||||
FS-43
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Supplemental Guarantor Information—Continued
CONSOLIDATING CONDENSED BALANCE SHEETS
| The Company (Parent) |
Combined Guarantor Subsidiaries |
Combined Non-Guarantor Subsidiaries |
Eliminations | Total | ||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| December 31, 2010 |
||||||||||||||||||||
| Assets |
||||||||||||||||||||
| Current Assets |
||||||||||||||||||||
| Cash and cash equivalents |
$ | 4,036 | $ | 2,476 | $ | 41,950 | $ | — | $ | 48,462 | ||||||||||
| Trade receivables, net |
95,673 | 68,504 | 87,827 | — | 252,004 | |||||||||||||||
| Installment receivables, net |
— | 876 | 3,083 | — | 3,959 | |||||||||||||||
| Inventories, net |
72,499 | 39,299 | 63,873 | (1,296 | ) | 174,375 | ||||||||||||||
| Deferred income taxes |
3,289 | — | 2,489 | — | 5,778 | |||||||||||||||
| Other current assets |
12,274 | 6,895 | 27,685 | (5,273 | ) | 41,581 | ||||||||||||||
| Total Current Assets |
187,771 | 118,050 | 226,907 | (6,569 | ) | 526,159 | ||||||||||||||
| Investment in subsidiaries |
1,489,732 | 594,690 | — | (2,084,422 | ) | — | ||||||||||||||
| Intercompany advances, net |
77,990 | 745,991 | 226,421 | (1,050,402 | ) | — | ||||||||||||||
| Other Assets |
42,782 | 1,881 | 821 | — | 45,484 | |||||||||||||||
| Other Intangibles |
1,241 | 8,590 | 61,080 | — | 70,911 | |||||||||||||||
| Property and Equipment, net |
46,791 | 12,093 | 71,879 | — | 130,763 | |||||||||||||||
| Goodwill |
5,023 | 34,388 | 467,672 | — | 507,083 | |||||||||||||||
| Total Assets |
$ | 1,851,330 | $ | 1,515,683 | $ | 1,054,780 | $ | (3,141,393 | ) | $ | 1,280,400 | |||||||||
| Liabilities and Shareholders’ Equity |
||||||||||||||||||||
| Current Liabilities |
||||||||||||||||||||
| Accounts payable |
$ | 73,468 | $ | 14,923 | $ | 55,362 | $ | — | $ | 143,753 | ||||||||||
| Accrued expenses |
39,090 | 20,690 | 75,572 | (5,273 | ) | 130,079 | ||||||||||||||
| Accrued income taxes |
5,633 | — | 2,869 | — | 8,502 | |||||||||||||||
| Short-term debt and current maturities of long-term obligations |
7,149 | 83 | 742 | — | 7,974 | |||||||||||||||
| Total Current Liabilities |
125,340 | 35,696 | 134,545 | (5,273 | ) | 290,308 | ||||||||||||||
| Long-Term Debt |
217,164 | — | 20,926 | — | 238,090 | |||||||||||||||
| Other Long-Term Obligations |
48,645 | 1,123 | 49,823 | — | 99,591 | |||||||||||||||
| Intercompany advances, net |
807,770 | 180,743 | 61,889 | (1,050,402 | ) | — | ||||||||||||||
| Total Shareholders’ Equity |
652,411 | 1,298,121 | 787,597 | (2,085,718 | ) | 652,411 | ||||||||||||||
| Total Liabilities and Shareholders’ Equity |
$ | 1,851,330 | $ | 1,515,683 | $ | 1,054,780 | $ | (3,141,393 | ) | $ | 1,280,400 | |||||||||
FS-44
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Supplemental Guarantor Information—Continued
CONSOLIDATING CONDENSED BALANCE SHEETS
| The Company (Parent) |
Combined Guarantor Subsidiaries |
Combined Non-Guarantor Subsidiaries |
Eliminations | Total | ||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| December 31, 2009 |
||||||||||||||||||||
| Assets |
||||||||||||||||||||
| Current Assets |
||||||||||||||||||||
| Cash and cash equivalents |
$ | 6,569 | $ | 2,526 | $ | 28,406 | $ | — | $ | 37,501 | ||||||||||
| Trade receivables, net |
101,416 | 64,451 | 101,312 | (4,165 | ) | 263,014 | ||||||||||||||
| Installment receivables, net |
— | 954 | 2,611 | — | 3,565 | |||||||||||||||
| Inventories, net |
42,512 | 39,114 | 91,916 | (1,320 | ) | 172,222 | ||||||||||||||
| Deferred income taxes |
— | — | 390 | — | 390 | |||||||||||||||
| Other current assets |
15,608 | 6,307 | 31,245 | (1,388 | ) | 51,772 | ||||||||||||||
| Total Current Assets |
166,105 | 113,352 | 255,880 | (6,873 | ) | 528,464 | ||||||||||||||
| Investment in subsidiaries |
1,447,759 | 594,024 | — | (2,041,783 | ) | — | ||||||||||||||
| Intercompany advances, net |
115,510 | 1,057,341 | 196,323 | (1,369,174 | ) | — | ||||||||||||||
| Other Assets |
43,246 | 3,420 | 1,340 | — | 48,006 | |||||||||||||||
| Other Intangibles |
1,604 | 8,023 | 75,678 | — | 85,305 | |||||||||||||||
| Property and Equipment, net |
49,608 | 9,344 | 82,681 | — | 141,633 | |||||||||||||||
| Goodwill |
5,023 | 24,634 | 526,436 | — | 556,093 | |||||||||||||||
| Total Assets |
$ | 1,828,855 | $ | 1,810,138 | $ | 1,138,338 | $ | (3,417,830 | ) | $ | 1,359,501 | |||||||||
| Liabilities and Shareholders’ Equity |
||||||||||||||||||||
| Current Liabilities |
||||||||||||||||||||
| Accounts payable |
$ | 70,867 | $ | 12,986 | $ | 57,206 | $ | — | $ | 141,059 | ||||||||||
| Accrued expenses |
45,309 | 24,137 | 78,400 | (5,553 | ) | 142,293 | ||||||||||||||
| Accrued income taxes |
— | — | 5,884 | — | 5,884 | |||||||||||||||
| Short-term debt and current maturities of long-term obligations |
173 | — | 918 | — | 1,091 | |||||||||||||||
| Total Current Liabilities |
116,349 | 37,123 | 142,408 | (5,553 | ) | 290,327 | ||||||||||||||
| Long-Term Debt |
262,188 | — | 10,046 | — | 272,234 | |||||||||||||||
| Other Long-Term Obligations |
45,156 | 2,040 | 48,507 | — | 95,703 | |||||||||||||||
| Intercompany advances, net |
703,925 | 564,582 | 100,667 | (1,369,174 | ) | — | ||||||||||||||
| Total Shareholders’ Equity |
701,237 | 1,206,393 | 836,710 | (2,043,103 | ) | 701,237 | ||||||||||||||
| Total Liabilities and Shareholders’ Equity |
$ | 1,828,855 | $ | 1,810,138 | $ | 1,138,338 | $ | (3,417,830 | ) | $ | 1,359,501 | |||||||||
FS-45
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Supplemental Guarantor Information—Continued
CONSOLIDATING CONDENSED STATEMENTS OF CASH FLOWS
| The Company (Parent) |
Combined Guarantor Subsidiaries |
Combined Non-Guarantor Subsidiaries |
Eliminations | Total | ||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| Year ended December 31, 2010 |
||||||||||||||||||||
| Net Cash Provided (Used) by Operating Activities |
$ | 101,658 | $ | 15,427 | $ | 44,442 | $ | (39,320 | ) | $ | 122,207 | |||||||||
| Investing Activities |
||||||||||||||||||||
| Purchases of property and equipment |
(7,281 | ) | (1,567 | ) | (8,505 | ) | — | (17,353 | ) | |||||||||||
| Proceeds from sale of property and equipment |
— | — | 36 | — | 36 | |||||||||||||||
| Business acquisitions, net of cash acquired |
— | (13,725 | ) | — | — | (13,725 | ) | |||||||||||||
| Decrease (increase) in other long-term assets |
291 | (11 | ) | 521 | — | 801 | ||||||||||||||
| Other |
153 | (174 | ) | (355 | ) | — | (376 | ) | ||||||||||||
| Net Cash Used for Investing Activities |
(6,837 | ) | (15,477 | ) | (8,303 | ) | — | (30,617 | ) | |||||||||||
| Financing Activities |
||||||||||||||||||||
| Proceeds from revolving lines of credit and long-term borrowings |
689,022 | — | 19,720 | — | 708,742 | |||||||||||||||
| Payments on revolving lines of credit and long-term borrowings |
(751,660 | ) | — | — | — | (751,660 | ) | |||||||||||||
| Proceeds from exercise of stock options |
2,912 | — | — | — | 2,912 | |||||||||||||||
| Payment of financing costs |
(30,329 | ) | — | — | — | (30,329 | ) | |||||||||||||
| Payment of dividends |
(1,612 | ) | — | (39,320 | ) | 39,320 | (1,612 | ) | ||||||||||||
| Purchase of treasury stock |
(5,687 | ) | — | — | — | (5,687 | ) | |||||||||||||
| Net Cash Provided (Used) by Financing Activities |
(97,354 | ) | — | (19,600 | ) | 39,320 | (77,634 | ) | ||||||||||||
| Effect of exchange rate changes on cash |
— | — | (2,995 | ) | — | (2,995 | ) | |||||||||||||
| Increase (Decrease) in cash and cash equivalents |
(2,533 | ) | (50 | ) | 13,544 | — | 10,961 | |||||||||||||
| Cash and cash equivalents at beginning of year |
6,569 | 2,526 | 28,406 | — | 37,501 | |||||||||||||||
| Cash and cash equivalents at end of year |
$ | 4,036 | $ | 2,476 | $ | 41,950 | $ | — | $ | 48,462 | ||||||||||
FS-46
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Supplemental Guarantor Information—Continued
CONSOLIDATING CONDENSED STATEMENTS OF CASH FLOWS
| The Company (Parent) |
Combined Guarantor Subsidiaries |
Combined Non-Guarantor Subsidiaries |
Eliminations | Total | ||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| Year ended December 31, 2009 |
||||||||||||||||||||
| Net Cash Provided (Used) by Operating Activities |
$ | 154,367 | $ | 1,823 | $ | 105,575 | $ | (106,102 | ) | $ | 155,663 | |||||||||
| Investing Activities |
||||||||||||||||||||
| Purchases of property and equipment |
(6,733 | ) | (1,875 | ) | (9,391 | ) | — | (17,999 | ) | |||||||||||
| Proceeds from sale of property and equipment |
5 | — | 1,158 | — | 1,163 | |||||||||||||||
| Decrease (increase) in other long-term assets |
737 | (122 | ) | (14 | ) | — | 601 | |||||||||||||
| Other |
(579 | ) | 416 | (284 | ) | — | (447 | ) | ||||||||||||
| Net Cash Used for Investing Activities |
(6,570 | ) | (1,581 | ) | (8,531 | ) | — | (16,682 | ) | |||||||||||
| Financing Activities |
||||||||||||||||||||
| Proceeds from revolving lines of credit and long-term borrowings |
400,123 | — | — | — | 400,123 | |||||||||||||||
| Payments on revolving lines of credit and long-term borrowings |
(552,294 | ) | — | (1,142 | ) | — | (553,436 | ) | ||||||||||||
| Proceeds from exercise of stock options |
1,628 | — | — | — | 1,628 | |||||||||||||||
| Payment of dividends |
(1,605 | ) | — | (106,102 | ) | 106,102 | (1,605 | ) | ||||||||||||
| Net Cash Provided (Used) by Financing Activities |
(152,148 | ) | — | (107,244 | ) | 106,102 | (153,290 | ) | ||||||||||||
| Effect of exchange rate changes on cash |
— | — | 4,294 | — | 4,294 | |||||||||||||||
| Increase (Decrease) in cash and cash equivalents |
(4,351 | ) | 242 | (5,906 | ) | — | (10,015 | ) | ||||||||||||
| Cash and cash equivalents at beginning of year |
10,920 | 2,284 | 34,312 | — | 47,516 | |||||||||||||||
| Cash and cash equivalents at end of year |
$ | 6,569 | $ | 2,526 | $ | 28,406 | $ | — | $ | 37,501 | ||||||||||
FS-47
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Supplemental Guarantor Information—Continued
CONSOLIDATING CONDENSED STATEMENTS OF CASH FLOWS
| The Company (Parent) |
Combined Guarantor Subsidiaries |
Combined Non-Guarantor Subsidiaries |
Eliminations | Total | ||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| Year ended December 31, 2008 |
||||||||||||||||||||
| Net Cash Provided (Used) by Operating Activities |
$ | 33,365 | $ | 2,248 | $ | 51,667 | $ | (10,866 | ) | $ | 76,414 | |||||||||
| Investing Activities |
||||||||||||||||||||
| Purchases of property and equipment |
(5,377 | ) | (1,246 | ) | (13,334 | ) | — | (19,957 | ) | |||||||||||
| Proceeds from sale of property and equipment |
— | 2 | 209 | — | 211 | |||||||||||||||
| Business acquisitions, net of cash acquired |
(6,268 | ) | (2,152 | ) | — | — | (8,420 | ) | ||||||||||||
| Decrease in other long-term assets |
4,882 | — | — | — | 4,882 | |||||||||||||||
| Other |
(620 | ) | 1,666 | (247 | ) | — | 799 | |||||||||||||
| Net Cash Used for Investing Activities |
(7,383 | ) | (1,730 | ) | (13,372 | ) | — | (22,485 | ) | |||||||||||
| Financing Activities |
||||||||||||||||||||
| Proceeds from revolving lines of credit and long-term borrowings |
334,680 | — | 21,581 | — | 356,261 | |||||||||||||||
| Payments on revolving lines of credit and long-term borrowings |
(376,110 | ) | (7 | ) | (41,065 | ) | — | (417,182 | ) | |||||||||||
| Proceeds from exercise of stock options |
834 | — | — | — | 834 | |||||||||||||||
| Payment of dividends |
(1,599 | ) | — | (10,866 | ) | 10,866 | (1,599 | ) | ||||||||||||
| Net Cash Provided (Used) by Financing Activities |
(42,195 | ) | (7 | ) | (30,350 | ) | 10,866 | (61,686 | ) | |||||||||||
| Effect of exchange rate changes on cash |
— | — | (6,927 | ) | — | (6,927 | ) | |||||||||||||
| Increase (Decrease) in cash and cash equivalents |
(16,213 | ) | 511 | 1,018 | — | (14,684 | ) | |||||||||||||
| Cash and cash equivalents at beginning of year |
27,133 | 1,773 | 33,294 | — | 62,200 | |||||||||||||||
| Cash and cash equivalents at end of year |
$ | 10,920 | $ | 2,284 | $ | 34,312 | $ | — | $ | 47,516 | ||||||||||
FS-48
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Interim Financial Information (unaudited)
| QUARTER ENDED (In thousands, except per share data) |
||||||||||||||||
| March 31, | June 30, | September 30, | December 31, | |||||||||||||
| 2010 |
||||||||||||||||
| Net sales |
$ | 402,240 | $ | 430,828 | $ | 437,476 | $ | 451,537 | ||||||||
| Gross profit |
117,713 | 126,490 | 131,567 | 133,871 | ||||||||||||
| Earnings before income taxes |
5,306 | 2,414 | 20,923 | 9,398 | ||||||||||||
| Net earnings (loss) |
3,106 | (611 | ) | 15,598 | 7,248 | |||||||||||
| Net earnings (loss) per share—basic |
.10 | (.02 | ) | .48 | .22 | |||||||||||
| Net earnings (loss) per share—assuming dilution |
.09 | (.02 | ) | .48 | .22 | |||||||||||
| March 31, | June 30, | September 30, | December 31, | |||||||||||||
| 2009 |
||||||||||||||||
| Net sales |
$ | 397,995 | $ | 412,541 | $ | 434,031 | $ | 448,569 | ||||||||
| Gross profit |
108,468 | 118,055 | 131,454 | 135,217 | ||||||||||||
| Earnings before income taxes |
4,447 | 10,561 | 17,776 | 14,495 | ||||||||||||
| Net earnings |
2,397 | 7,661 | 13,476 | 17,645 | ||||||||||||
| Net earnings per share—basic |
.08 | .24 | .42 | .55 | ||||||||||||
| Net earnings per share—assuming dilution |
.08 | .24 | .42 | .55 | ||||||||||||
FS-49
Table of Contents
INVACARE CORPORATION AND SUBSIDIARIES
SCHEDULE II—VALUATION AND QUALIFYING ACCOUNTS
| COL A. | COL B. | COL C. | COL D. | |||||||||||||
| Balance At Beginning of Period |
Charged To Cost And Expenses |
Additions (Deductions) Describe |
Balance At End of Period |
|||||||||||||
| (In thousands) | ||||||||||||||||
| Year Ended December 31, 2010 |
||||||||||||||||
| Deducted from asset accounts— |
||||||||||||||||
| Allowance for doubtful accounts |
$ | 28,075 | $ | 16,979 | $ | (14,886 | )(A) | $ | 30,168 | |||||||
| Inventory obsolescence reserve |
15,009 | 5,350 | (6,117 | )(B) | 14,242 | |||||||||||
| Tax valuation allowances |
65,050 | 4,526 | 12,405 | (D) | 81,981 | |||||||||||
| Accrued warranty cost |
21,506 | 6,427 | (9,681 | )(B) | 18,252 | |||||||||||
| Accrued product liability |
23,989 | 8,523 | (8,352 | )(C) | 24,160 | |||||||||||
| Year Ended December 31, 2009 |
||||||||||||||||
| Deducted from asset accounts— |
||||||||||||||||
| Allowance for doubtful accounts |
$ | 23,090 | $ | 19,281 | $ | (14,296 | )(A) | $ | 28,075 | |||||||
| Inventory obsolescence reserve |
12,419 | 6,497 | (3,907 | )(B) | 15,009 | |||||||||||
| Tax valuation allowances |
75,507 | 6,275 | (16,732 | )(D) | 65,050 | |||||||||||
| Accrued warranty cost |
16,798 | 14,112 | (9,404 | )(B) | 21,506 | |||||||||||
| Accrued product liability |
23,758 | 7,880 | (7,649 | )(C) | 23,989 | |||||||||||
| Year Ended December 31, 2008 |
||||||||||||||||
| Deducted from asset accounts— |
||||||||||||||||
| Allowance for doubtful accounts |
$ | 42,960 | $ | 14,284 | $ | (34,154 | )(A) | $ | 23,090 | |||||||
| Inventory obsolescence reserve |
12,501 | 8,469 | (8,551 | )(B) | 12,419 | |||||||||||
| Tax valuation allowances |
70,084 | 5,721 | (298 | )(D) | 75,507 | |||||||||||
| Accrued warranty cost |
16,616 | 12,546 | (12,364 | )(B) | 16,798 | |||||||||||
| Accrued product liability |
21,136 | 8,083 | (5,461 | )(C) | 23,758 | |||||||||||
Note (A)—Uncollectible accounts written off, net of recoveries.
Note (B)—Amounts written off or payments incurred.
Note (C)—Loss and loss adjustment.
Note (D)—Other activity not affecting federal or foreign tax expense.
FS-50
