Attached files
| file | filename |
|---|---|
| 8-K - 8-K - PDS Biotechnology Corp | form8k.htm |
Exhibit 99.1

CORPORATE PRESENTATION Frank Bedu-Addo Ph.D. President & CEO JUNE 2020

2 Forward-Looking Statements This presentation contains forward-looking statements about PDS
Biotechnology Corporation (“PDSB”), and its businesses, business prospects, strategies and plans, including but not limited to statements regarding anticipated pre-clinical and clinical drug development activities and timelines and market
opportunities. All statements other than statements of historical facts included in this presentation are forward-looking statements. The words “anticipates,” “may,” “can,” “plans,” “believes,” “estimates,” “expects,” “projects,” “intends,”
“likely,” “will,” “should,” “to be,” and any similar expressions or other words of similar meaning are intended to identify those assertions as forward-looking statements. These forward-looking statements involve substantial risks and
uncertainties that could cause actual results to differ materially from those anticipated.Factors that may cause actual results to differ materially from such forward-looking statements include those identified under the caption “Risk Factors”
in the documents filed with the Securities and Exchange Commission from time to time, including its Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K. You are cautioned not to place undue reliance on
these forward-looking statements, which speak only as of the date of this presentation. Except to the extent required by applicable law or regulation, PDSB undertakes no obligation to update the forward-looking statements included in this
presentation to reflect subsequent events or circumstances.

PDS Biotechnology leadership team has demonstrated success in the development and commercialization
of leading pharmaceutical products 3 Frank Bedu-Addo, PhDChief Executive Officer Lauren V. Wood, MDChief Medical Officer Gregory Conn, PhDChief Scientific Officer Senior executive experience with management of strategy and execution at
both large pharma and biotechsNotable drug development:Abelcet® (Liposome Company/ Elan)PEG-Intron® (Schering-Plough/ Merck) >30 years of translational clinical research experienceFormer Director of Clinical Research at the National Cancer
Institute Center for Cancer Research (Cancer Vaccine Branch) Co-founder>35 years of drug development experience In-depth experience with biotech drug discovery, product development and manufacturing

PDS Biotech is well-poised to transform vaccines and cancer treatment by fulfilling the promise of
immunotherapy 4 Clinical studies in areas of high unmet medical need supported by leaders in the field 4 Diversified pipeline focused on oncology and infectious disease 3 Demonstrated potential for strong clinical efficacy and durability
of response with minimal toxicity 2 Powerful immunotherapy platform that activates therapeutic and preventive immunological pathways 1

PDS Biotech is a clinical stage biotechnology company developing a pipeline of immunotherapies based on
the proprietary Versamune® platform 5 Versatile and potent T-cell-activating platformClinically validated induction of active antigen-specific killer helper T-cells as well as antibodies in vivoPromising clinical efficacy in early trials of
PDS0101 monotherapy with good safety and no dose limiting toxicities Publicly listed on NASDAQ: PDSB~15 employees with headquarters in Princeton, NJ15.3M shares outstanding with $21.0M in cash* OncologyPDS0101 (Phase 2): HPV-associated
cancersPDS0102: Prostate, breast cancersPDS0103: Ovarian, breast, colorectal and lung cancersPDS0104: MelanomaInfectious DiseasePDS0201: Tuberculosis vaccinePDS0202: Universal influenza vaccinePDS0203: COVID-19
vaccine Pipeline Versamune® Platform Corporate Overview * March 31, 2020

Versamune® Platform

v Versamune® is based on proprietary, positively charged and immune activating lipids that form
spherical nanoparticles in aqueous media The nanoparticles are sized to mimic viruses, which promotes excellent uptake by dendritic cells of the immune systemActivates the important Type I interferon immunological signaling pathwayVersamune®
promotes the activation and maturation of dendritic cells, which then migrate to the lymph nodes Versamune® is a proprietary T-cell activating platform engineered to induce a robust, targeted anti-tumor response in
vivo 7 Water-insolubleFatty acids/hydrocarbon chains Water-soluble and positively charged head-group coats the particle surface R-enantiomer of 1,2-dioleoyl-trimethyl-ammonium-propane (R-DOTAP)

Inability to perform the necessary steps to induce a strong therapeutic killer T-cell response
in-vivo Versamune® design and novel immunological mechanisms of action promote a powerful disease-specific killer T-cell response Versamune® has demonstrated the potential to overcome the challenges of immunotherapy in oncology and
infectious disease 8 Challenges of Immunotherapy How Versamune® May Overcome the Challenge Mechanistic limitations have resulted in lack of therapeutic benefit in human studies Mechanism of action associated with regression of disease
in human studies (PDS0101 monotherapy) Potential for systemic toxicities Mechanism of action results in a lack of clinically relevant toxicities, even at the highest dose, in human studies Reference: Gandhapudi SK, Ward M, Bush
JPC, Bedu-Addo F, Conn G, Woodward JG. 2019. Antigen priming with enantiospecific cationic lipid nanoparticles induces potent antitumor CTL responses through novel induction of a Type I IFN response. J Immunol. 202 (12): 3524-3536.

Greater quantity and quality of Versamune®-induced killer T-cells may result in unique ability to
eradicate HPV-positive tumors after a single dose 9 Produces > 10-fold number of highly potent (polyfunctional) killer T-cells vs. other T-cell technologies Single treatment dose Results typical of current topclinical-stage HPV cancer
vaccines Tumor rechallenge at Day 60; complete and sustained cure of cancer *Adjuvant = cytokine GMCSFReferences: J. Immunology, 2019 (202), 1215; Studies in TC-1 tumor model with other immunotherapies reported in: Vaccine 2009, January
14, 27 (3): 431; Science Translational Medicine 2016, 13 April, Vol 8 Issue 334; Vaccine 2009, August 3, 27 (33): 5706

10 The combination of Versamune® and a proprietary antigen is engineered for simplicity and ease of
administration Vials of HPV16 mix (L)and Versamune® (R) Versamune® formulationis mixed before injection* Delivered viasubcutaneous injection *Electron microscopy picture

Oncology

12 Reference: Data on file. PDS Biotech’s immuno-oncology pipeline combines the Versamune® platform
with proprietary tumor antigens across several cancer types

13 PDS0101 is designed to treat cancers caused by human papillomavirus (HPV) Approximately 43,000
patients are diagnosed with HPV-associated cancers each year, a number unlikely to be impacted by increased use of HPV preventive vaccines in the next decade References: Markowitz et al. 2016. Centers for Disease Control and Prevention. 2018.
Oropharyngeal (head & neck) cancers >18,000 cases annuallyMost common HPV-cancer in men,90% of cases are HPV16-specificIncidence increasingCervical cancer~12,000 cases annuallyMost common HPV-cancer in women,50-60% of cases are
HPV16-specificIncidence steadyInitial market research suggests market penetration of ~20% is reasonable for PDS0101 PDS0101 combines the utility of the Versamune® platform with a proprietary mix of HPV16 antigens, the most virulent high-risk
HPV type and by far the most prevalent in patients with HPV-associated cancer Females (24,391) Males (18,280)

14 PDS0101 Phase 1 clinical trial: Unique in vivo demonstration of high levels of HPV-specific killer
T-cells in circulating blood 14x 24x Total Activated T-Cells 26x Order of magnitude increase over baseline Versamune® Dose Reference: Data on file. INF-γ Elispot Granzyme-b Elispot Clinical Study Results in Patients with
CINImmunogenicity at Day 14Defined dose for Phase 2 studies (3mg)No dose-limiting toxicities Clinical study results successfully demonstrate translation of Versamune®’s multi-functional mechanism of action between pre-clinical models and
humans

15 Follow-up of patients in PDS0101 Phase 1 study demonstrated promising clinical responses at all three
tested doses A post-hoc, retrospective analysis, demonstrated complete lesion regression in at least 60% of evaluable patients (6/10) as early as 1-3 months after treatmentNo lesion recurrence occurred within the 2-year evaluation
periodSpontaneous regression of CIN1 occurs in about 44% of patients over a 2-year duration* These results were remarkably positive as most patients were infected with multiple high-risk HPV typesTwo patients who had regression by cytology were
not considered clinical responders:The first regressed to atypical cells of undetermined significance at the first post-treatment evaluation (3 months) but HPV detectedThe second had complete regression by cytology at the first post-treatment
evaluation (3 months) but had residual CIN by colposcopy Reference: Stefani C. et al, 2014, European Review for Medical and Pharmacological Sciences, 18: 728-733

Checkpoint inhibitors have shown confirmed clinical efficacy and have demonstrated clinical benefit in
late stage cancerCheckpoint inhibitors block a key immunological defense mechanism for cancer cells, and are reported to work primarily in patients whose immune systems are already generating tumor-attacking CD8+ killer T-cells
pre-treatmentUsing various tumor-specific proteins (antigens), Versamune® has demonstrated the unique ability to generate large and superior numbers of CD8+ killer T-cells that effectively recognize and kill antigen-expressing cancer cells in
pre-clinical and human clinical studies PDS Biotech clinical strategy in advanced cancer is to focus on efficiency and risk mitigation to proof of concept 16 Versamune®-based immunotherapies are being developed as combination therapies to
exploit the demonstrated synergies between Versamune® and other anti-cancer agents PDS Biotech is developing a new generation of advanced cancer treatments combining Versamune®-based immunotherapies with checkpoint inhibitors and other
standard of care therapies

The robust T-cell response induced by Versamune® results in the potential for enhancement of efficacy of
checkpoint inhibitors in immune suppressive B16 melanoma 17 Early clinical studies showed the checkpoint inhibitor to be ineffective in treating B16 melanoma, a notoriously difficult model to treat. Versamune® + TRP2 melanoma antigen
(sub-optimal levels) promotes infiltration of active killer T-cells into tumors, strong synergy with the checkpoint inhibitor, and significantly enhanced anti-tumor efficacy Reference: Gandhapudi SK, Ward M, Bush JPC, Bedu-Addo F, Conn G,
Woodward JG. 2019. Antigen priming with enantiospecific cationic lipid nanoparticles induces potent antitumor CTL responses through novel induction of a Type I IFN response. J Immunol. 202 (12): 3524-3536. CD4+ helper T-cell CD8+ killer
T-cell

18 PDS0101 is the only compound selected by Merck for evaluation in combination with KEYTRUDA® as first
line cancer therapy PDS0101 + KEYTRUDA®Keytruda® first immunotherapy approved as standard of care for first line treatment of cancer (recurrent or metastatic head and neck cancer)PDS0101 first T-cell activating immunotherapy to demonstrate
both high levels of circulating CD8+ killer T-cells and therapeutic benefit as monotherapyUnique immuno-oncology combination addressing first-line treatment of cancerValidation of both efficacy and safety of PDS0101Anticipated advantages of
combining PDS0101 with standard of care: Mitigated riskPotential enhanced rates of recruitmentPotential for rapid market penetration and market leadership Initiation TBD pending easing of COVID-19 restrictions

19 PDS Biotechnology-sponsored study with KEYTRUDA ® supplied by MerckPrimary endpoints: Efficacy,
safety and tolerabilityStudy design: Phase 2 open-label studyInclusion criteria: Recurrent/metastatic head and neck cancer and HPV16 infectionClinical Trial Identifier: NCT04260126 A Phase 2 study of PDS0101 in combination with KEYTRUDA ® in
first-line treatment of recurrent/metastatic head and neck cancer is planned Followed by open label SOC with KEYTRUDA® until disease progression or intolerance KEYTRUDA® alone Combination of PDS0101 and
KEYTRUDA® 200 mg IV KEYTRUDA® every 21 days in combinationwith 3 mg SC PDS0101 at cycles 1, 2, 3, 4 and 12 Expected initiation: TBD pending easing of COVID-19 restrictions

20 Investigator-Led Phase 2 studies of PDS0101 in combination therapy will evaluate efficacy and safety
in treatment of advanced HPV cancers Funded By Phase 2 Open Label Study (Safety and Efficacy) Important Considerations Expected Initiation Advanced HPV-associated malignancies – all typesTriple combination with EMD Serono’s M7824 and
NHS-IL1234 subjectsClinical Trial Identifier: NCT04287868 NCI selection and confirmation of synergies with PDS0101All three agents have demonstrated efficacy as monotherapies in early trials TBD pending easing of COVID-19 restrictions
Advanced, localized cervical cancer (Stage IIb-IVa)Combination with chemo-radiotherapy (CRT-standard of care) 35 subjects T-cell induction has strong potential to enhance CRT anti-cancer efficacyMitigated riskPotential for rapid market
penetration and market leadership TBD pending easing of COVID-19 restrictions

Infectious Disease
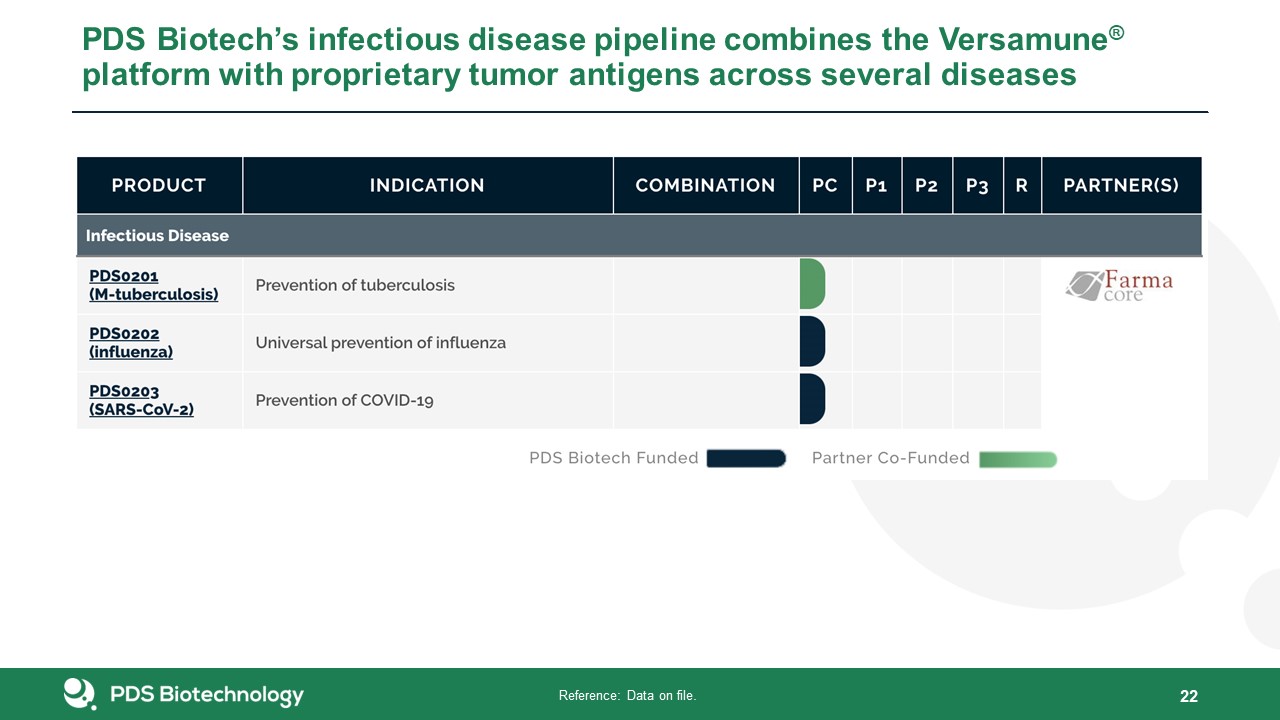
22 Reference: Data on file. PDS Biotech’s infectious disease pipeline combines the Versamune® platform
with proprietary tumor antigens across several diseases
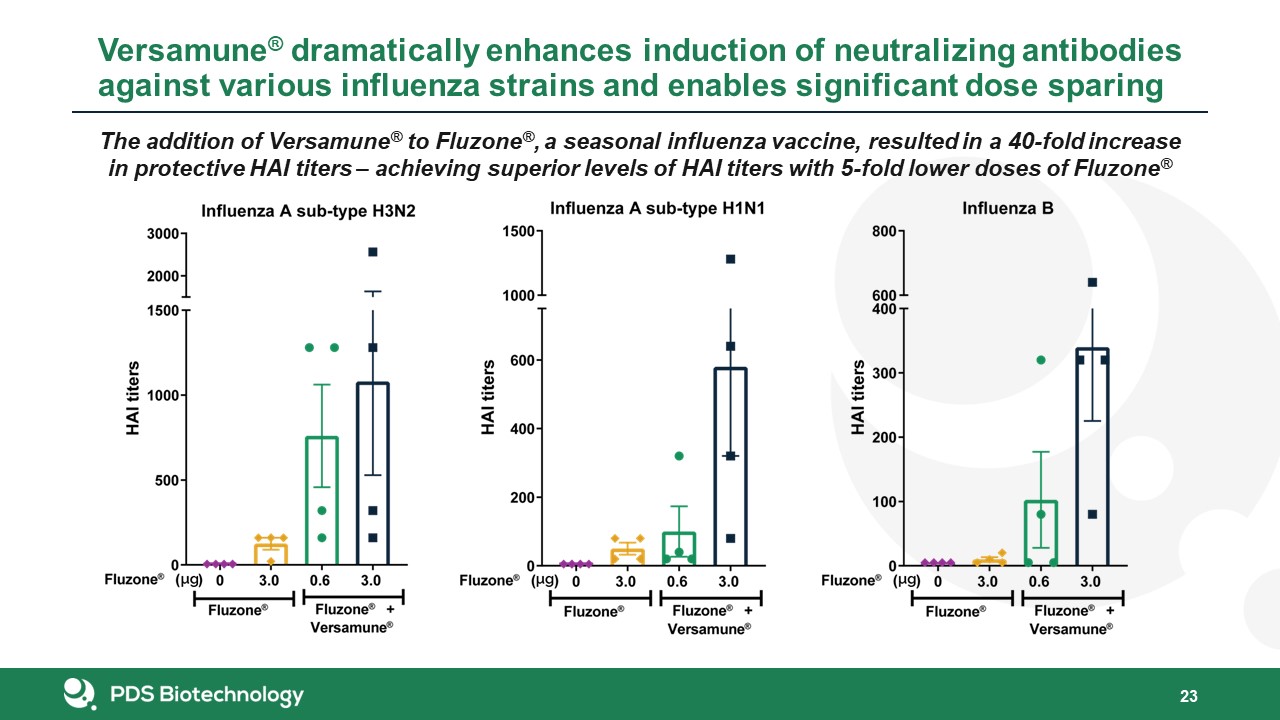
23 The addition of Versamune® to Fluzone®, a seasonal influenza vaccine, resulted in a 40-fold increase
in protective HAI titers – achieving superior levels of HAI titers with 5-fold lower doses of Fluzone® Versamune® dramatically enhances induction of neutralizing antibodies against various influenza strains and enables significant dose
sparing

24 Versamune® uniquely induces both potent antibody mediated responses and high levels of helper and
killer T-cells vs. CFA when both are administered with the same recombinant protein antigen *Adjuvant = Complete Freund’s Adjuvant (CFA) – A powerful immunological adjuvant that is too toxic for human use Versamune®-based vaccines uniquely
induce strong antibody, helper and killer T-cell responses against an antigen to provide superior protection Antibody Response (Day 15) CD4+ Helper T-Cell Response Control Adjuvant*+ Antigen Versamune® + Antigen CD8+ Killer T-Cell
Response Control Versamune® + Antigen Adjuvant*+ Antigen

Preclinical testing of PDS Biotech’s Versamune®-based COVID-19 vaccine candidates are underway with a
clear target profile 25 Poised for rapid commercial scale up 4 No safety signals 3 Demonstrate high levels of both SARS-CoV-2-specific T-cell and antibody response after a single dose 2 Induction of highly-potent, SARS-CoV-2-specific
killer T-cells 1

Intellectual Property and Financials

27 Multiple layers of technology and product protection for Versamune®-related products through
mid-2030s are secured Versamune® and associated patents are owned and licensed by PDS BiotechPatents cover methods and compositions stimulating/promoting an immune response with Versamune® technology in various forms and mechanisms through
2034Use of specific cationic lipids to induce an immune responseCompositions and use of any cationic lipid to activate MAP kinaseCompositions and use of R-DOTAP to induce immune responseMicellar antigen + cationic lipids compositions (US still
ongoing) Compositions of R-DOTAP with GM-CSF to reduce immune suppressive myeloid derived suppressor cells in the tumorFive issued international patent families (including Europe and Japan)

PDS Biotech is in a strong financial position to support near-term milestones 28 Nasdaq PDSB Shares
Outstanding* 15.3M Cash* $21.0M Share Price** $1.31 Market Cap** $19.7M Debt* --- *March 31, 2020 **May 31, 2020 PDS0201 (M. tuberculosis): Complete development and feasibility testingPDS0202 (influenza): Complete development and
feasibility testingPDS0203 (SARS-CoV-2): Complete development and feasibility testingPDS0101 (HPV): Initiation of PDS Biotech-NCI Phase 2 combination study in advanced HPV-associated cancersPDS0101 (HPV): Initiation of Partnered Phase 2
combination study in advanced cervical-cancer Timing of the 2020 milestones will be impacted by the COVID-19 pandemic

PDS Biotech is well-poised to transform vaccines and cancer treatment by fulfilling the promise of
immunotherapy 29 Clinical studies in areas of high unmet medical need supported by leaders in the field 4 Diversified pipeline focused on oncology and infectious disease 3 Demonstrated potential for strong clinical efficacy and durability
of response with minimal toxicity 2 Powerful immunotherapy platform that activates therapeutic and preventive immunological pathways 1

