Attached files
| file | filename |
|---|---|
| 8-K - 8-K - Kindred Biosciences, Inc. | a8k-december2016investorpr.htm |

1 KindredBio
KindredBio
Best Medicines for Our Best Friends

2 KindredBio
Forward Looking Statements
This presentation contains forward-looking statements, including but not limited to statements regarding the
timing of development for our product candidates, expected commencement and completion of pivotal trials,
prospective product candidates, anticipated regulatory approvals for our product candidates, anticipated
commercialization of our product candidates, our financial position, business strategy, plans and objectives of
management for future operations and other similar statements. These forward-looking statements are based
on our current expectations and beliefs, as well as assumptions concerning future events. These statements
are subject to risks, uncertainties, and other factors, many of which are outside of our control, that could cause
actual results to differ materially from the results discussed in the forward-looking statements, including, but not
limited to, our limited operating history and lack of profitability, our lack of product revenue and potential need
to raise additional capital to achieve our goals, our dependence upon the success of our lead product
candidates, other companies’ ability to develop substantially similar products that may compete with our
product candidates, any inability to obtain regulatory approval for our existing or future product candidates, any
delay or discontinuance of our current or future pivotal trials, any inability to achieve market acceptance or
commercial success for our product candidates even if they are approved, inability to obtain adequate
intellectual property protection covering our product candidates, our dependence on third-party manufacturers
for supplies, and any inability to successfully identify, develop and commercialize additional product
candidates.
Any forward-looking statement made by us in this presentation speaks only as of the date of this presentation
and represents our estimates and assumptions only as of the date of this presentation. Except as required by
law, we assume no obligation to update these statements publicly, or to update the reasons actual results
could differ materially from those anticipated in these statements, even if new information becomes available in
the future.
November 7, 2016

3 KindredBio
$15.7 billion on veterinary care
$1.5 billion on dog knee
surgeries
$370 million on pet Halloween
costumes
$700 million on Valentine’s Day
gifts
Annually, U.S. pet parents estimated to spend:
We Love Our Pet Family Members

4 KindredBio
Bring the very best science and medicine
to our animal family members.
KindredBio
5 KindredBio
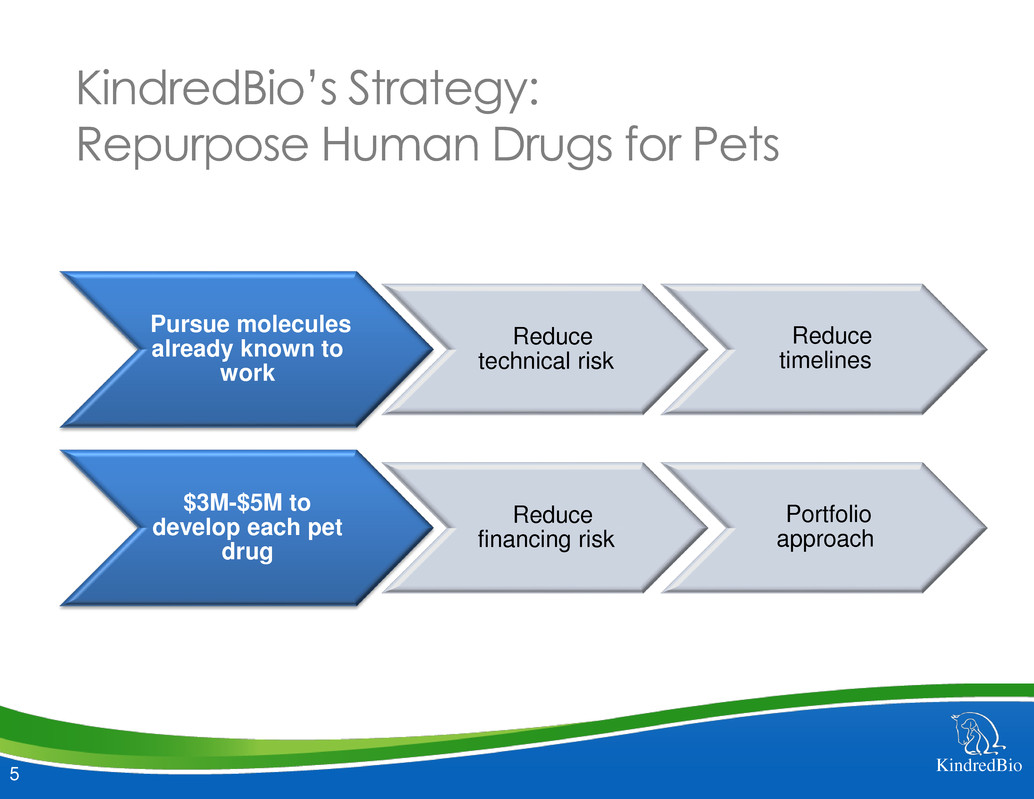
KindredBio’s Strategy:
Repurpose Human Drugs for Pets
Pursue molecules
already known to
work
Reduce
technical risk
Reduce
timelines
$3M-$5M to
develop each pet
drug
Reduce
financing risk
Portfolio
approach

6 KindredBio
KindredBio Highlights
Two launches
by 2017
Zimeta™ for fever in horses
Mirataz™ for mgmt. of weight loss in cats
~2 launches per year thereafter
Attractive
Markets
Rapidly growing
Few current competitors
World Class
Team
Extensive drug development experience
Human and veterinary experience
Deep Pipeline ~20 small molecule and biologic candidates
Strategic
Approach
Reduces technical risk
Reduces financing risk

7 KindredBio
Richard Chin, M.D.
Founder and Chief Executive Officer
Former Head of Clinical Research, BioTherapeutics, Genentech
Rhodes Scholar
World Class Leadership Team
7
Denise Bevers
Founder and Chief Operating Officer
Founder, SD Scientific; 25 years in biotech/pharma
Stephen Sundlof, D.V.M, Ph.D.
Chief Scientific Officer and Executive Vice President, Regulatory Affairs & Quality
Former Director, Center for Veterinary Medicine, FDA
Wendy Wee
Vice President, Finance
16 years of biotechnology finance experience
Hangjun Zhan, Ph.D.
Vice President, Biologics Research
20 years of drug discovery experience

8 KindredBio
Molecule Indication Discovery/Process Development
Pilot/Pivotal
Clinical Studies
NADA & Launch
Preparation
Biologic Product Candidates
epoCat™ (feline erythropoietin) Anemia in cats
Anti-Interleukin Antibodies Atopic dermatitis in dogs
Checkpoint Inhibitors Cancer in dogs
Anti-CD20 antibody
Cancer and autoimmune
diseases in dogs
KIND-Bodies Multiple indications
Anti-IgE antibody Allergic diseases in dogs
Anti-VEGF antibody Cancer in dogs
Anti-TNF Sick newborn foals
Molecule Indication Formulation
Laboratory
Pilot Studies
Field Pilot
Studies
Pivotal
Clinical
Study
NADA &
Launch
Preparation
Small Molecule Product Candidates
Mirataz™
(mirtazapine transdermal ointment)
Management of weight loss in
cats
NADA
Zimeta™
(dipyrone injection)
Fever in horses
KIND-014* Equine gastric ulcers
KIND-015* Metabolic syndrome in horses
Deep Product Pipeline
Rolling NADA
Being Filed
Rolling NADA
Being Filed
Not all programs are listed. Some are not disclosed for competitive reasons.
* Initial pilot studies completed. Final formulation being developed.

9 KindredBio
Market Opportunity

10 KindredBio
$9.3
[VALUE]
$0
$2
$4
$6
$8
$10
$12
$14
$16
$18
2006 2015
Sp
e
n
d
in
g
in
B
ill
ion
s
Veterinary Market is Growing Rapidly
The veterinary care market
grew 70% from 2006 to 2015
Source: APPA Pet Industry Market Size & Ownership Statistics 2015 &
TriMark Vet Health Market, June 2013
Estimated
11 KindredBio
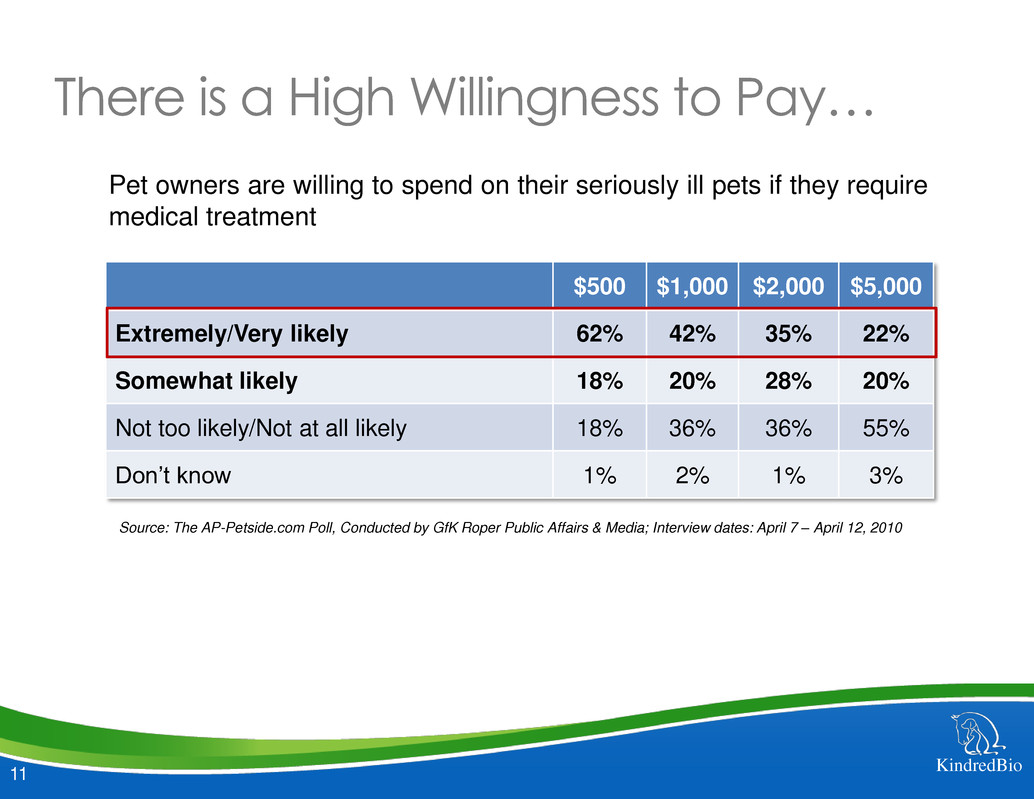
There is a High Willingness to Pay…
$500 $1,000 $2,000 $5,000
Extremely/Very likely 62% 42% 35% 22%
Somewhat likely 18% 20% 28% 20%
Not too likely/Not at all likely 18% 36% 36% 55%
Don’t know 1% 2% 1% 3%
Pet owners are willing to spend on their seriously ill pets if they require
medical treatment
Source: The AP-Petside.com Poll, Conducted by GfK Roper Public Affairs & Media; Interview dates: April 7 – April 12, 2010

12 KindredBio
…But Treatment Options are Limited
Underserved market with attractive growth
opportunities
Few competing biotechs
Large pharma focused on blockbusters
On average, less than a dozen pet drugs
are approved annually by the FDA
In 2015 the FDA approved:
0 novel pet drugs
45 novel humans drugs

13 KindredBio
Veterinary Pharmaceuticals Field
We believe there are similarities between veterinary pharmaceutical field
now and the human pharmaceutical field in its early stages:
Similar regulatory standards
Similar commercial and reimbursement landscape
Similar development costs
Numerous untapped opportunities (low hanging fruit)
Many years behind Pet
Therapeutics
Human
Pharmaceuticals

14 KindredBio
Veterinary vs. Human Markets
Can reach
market in
3-5 years Faster
Development
Lower
Development
Cost
Almost no
biotechs,
almost no
generics
Lower
Competition Self-Pay Very few
reimbursement
hurdles
Can develop
for $3M-$5M
per product

15 KindredBio
Programs

16 KindredBio
KindredBio Strategy
Already validated in
humans &
established
manufacturing
Customize species-
specific dosage and
formulate
flavored/convenient
delivery
Targets based on
approved human
drugs (e.g., Enbrel
and Orencia)
Create canine/
feline/equine
versions of biologics
with the same or
similar target
Small
Molecules
Biologics

17 KindredBio
Equine Products
• High willingness to pay
• Similar to human orphan drug market
• Efficient commercial structure
Biologics
• Atopic dermatitis
• Cancer
KindredBio’s Priority Areas

18 KindredBio
Equine Market
Fewer horses than cats or dogs, but higher willingness to pay –
similar to human orphan drug markets
Only 10-12 sales representatives required for full national
coverage
Underappreciated opportunity
KindredBio’s goal is to become dominant company in the equine
sector

19 KindredBio
Zimeta™ (dipyrone injection)
IV and oral drug for treatment of pyrexia (fever) in horses
High unmet medical need
U.S. equine veterinarians report seeing 12 cases (median) of
fever in a typical month
Currently, no FDA-approved treatment for fever in horses
8-9 million
horses in
the U.S
690,000
horses
treated
for fever
annually
Sources: The Economic Impact of the Horse Industry on the United States, 2005,
American Horse Council Foundation (n=18,648 U.S. horse owners/industry suppliers).
Zimeta Pricing Research, Ipsos Ag & Animal Health, May 2016
(n=160 U.S. equine veterinarians) . Data on file at Kindred Biosciences.

20 KindredBio
Zimeta Development Program
IV:
Positive pivotal field study completed
CMC technical section of NADA filed Dec 2015
Safety technical section of NADA filed Mar 2016
Effectiveness technical section of NADA approved by FDA Oct
2016
Expect approval 2H/2017, if review proceeds as expected
Oral:
Initial pilot studies completed
Final formulation development underway
21 KindredBio
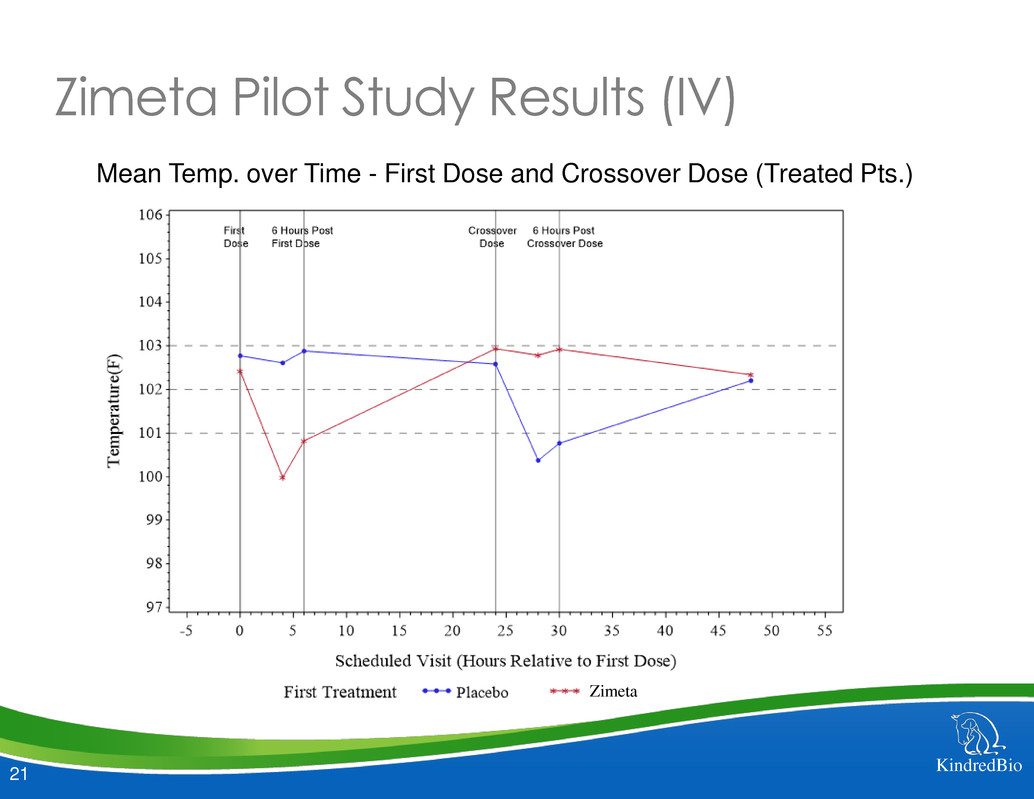
Zimeta Pilot Study Results (IV)
Mean Temp. over Time - First Dose and Crossover Dose (Treated Pts.)
Zimeta

22 KindredBio
Zimeta Pivotal Field Study Results (IV)
KB0120 study:
Randomized, double-blind, placebo-controlled (N=138)
Primary endpoint was improvement or resolution of fever
Placebo Zimeta
n 34 104
Success Rate (%) 7 (20.6) 78 (75.0)
p-value < 0.0001

23 KindredBio
Mirataz™
(mirtazapine transdermal ointment)
Transdermal drug for the management of weight loss in cats
Innovative Accusorb™ transdermal technology
High unmet medical need
Current drugs often not effective
90% of veterinarians treat cats with inappetence
(average of 7 cats per week)
9 million
cats are
inappetent
3 million cats
are treated
for
inappetence
References: 2012 U.S. Pet Ownership & Demographics Sourcebook
American Veterinary Medical Association (n=50,000 U.S. Households)
2016 U.S. Veterinarian Mirtazapine Research, Wise Insights
May 2016 (n=89 U.S. small animal veterinarians). Data on file at Kindred Biosciences.

24 KindredBio
Mirataz Development Program
Positive pivotal field study completed
CMC technical section of NADA filed May 2016
Effectiveness technical section of NADA filed Aug 2016
Safety technical section of NADA filed Sep 2016
Expect approval 2H/2017 if review proceeds as expected

25 KindredBio
Mirataz Pivotal Field Study Results
Randomized, double-blind, placebo-controlled study (N=231)
Primary endpoint was percentage change in body weight from
Day 1 to Week 2
Placebo Mirataz
Primary analysis population n=97 n=90
Mean % increase in body weight
from Day 1 to Week 2 (SE)
0.29 (0.35) 4.07 (0.56)
p-value <0.0001

26 KindredBio
Biologics
Highly experienced biologics team
Extensive experience developing Lucentis®, Xolair®, Tysabri®,
Avastin®, Rituxan®, Herceptin®, Enbrel®, and multiple other biologics
Internal caninization/felinization/equinization expertise
Promising biologics candidates
Feline erythropoietin
Checkpoint inhibitors
Interleukin antibodies
New technologies, including KIND-Bodies

27 KindredBio
New Scaffold Technology: KIND-Body
A proprietary, non-antibody scaffold technology being developed
by KindredBio
Binding affinity similar to antibodies
Bispecific binding possible
Could allow KindredBio to pursue biologics targets before other
parties’ antibody patents expire

28 KindredBio
KIND-510: Feline Erythropoietin
Proprietary recombinant feline erythropoietin (epoCat™)
Strong internal expertise in erythropoietin biology and
engineering
Initial laboratory study completed - positive efficacy signal, as
evidenced by increased reticulocyte formation
Pilot field study ongoing
High unmet medical need
Up to 30% of elderly cats (over 15 years) develop kidney failure,
leading to anemia
Human erythropoietin is immunogenic in cats

29 KindredBio
KIND-511: Anti-TNF for Newborn Foals
Sick newborn foals are challenging and difficult to treat
Approximately 50% mortality
Pilot field study for Anti-Tumor Necrosis Factor (Anti-TNF)
underway
Sick foals defined as sepsis score ≥ 11 or positive blood
culture
Of 5 foals enrolled, all survived to day 7 (primary endpoint of
study)

30 KindredBio
Key Focus Area: Atopic Dermatitis
Atopic dermatitis is a large market
Apoquel® projected peak sales >$200M
Multiple antibody candidates
Anti-IgE antibody
Anti-IL17a antibody
Anti-IL4Ra antibody
Anti-IL31 antibody
Anti-CD20 antibody
Multi-center, pilot field study underway to assess safety and
efficacy of several molecules

31 KindredBio
Key Focus Area: Cancer
Multiple antibody candidates
Anti-CTLA4 antibody
Anti-PD1 antibody
Anti-PDL1 antibody
Anti-CD20 antibody

32 KindredBio
Biologics Manufacturing Plant
In-house biologics manufacturing plant nearly complete
Single-use, state of the art system
Will allow full GMP commercial production of feline epo (epoCat™)
Will allow full GMP clinical and early commercial production of
antibodies

33 KindredBio
Commercialization

34 KindredBio
Commercialization
Equine Commercialization:
Launch and commercialize our U.S. products with
~10 person direct sales force
Dog/Cat Commercialization Options:
Self-Commercialization
Out-license
Partner for 3 – 5 years, and then transition to KindredBio
salesforce
35 KindredBio
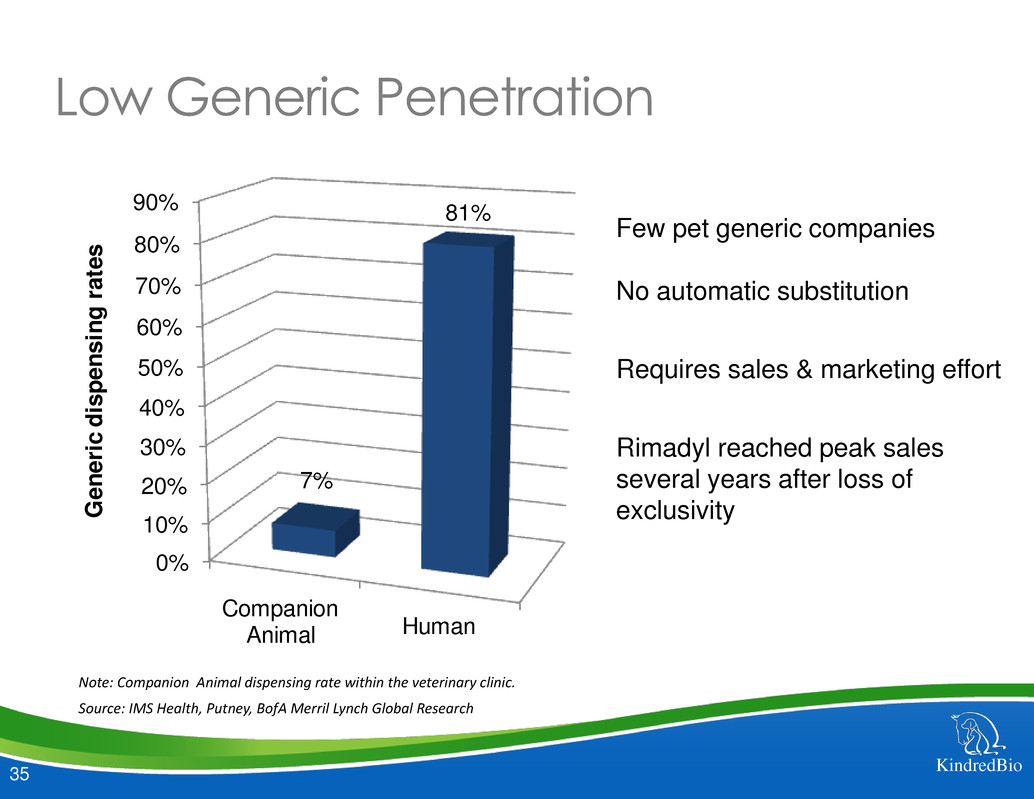
Few pet generic companies
No automatic substitution
Requires sales & marketing effort
Rimadyl reached peak sales
several years after loss of
exclusivity
Low Generic Penetration
Note: Companion Animal dispensing rate within the veterinary clinic.
Source: IMS Health, Putney, BofA Merril Lynch Global Research
0%
10%
20%
30%
40%
50%
60%
70%
80%
90%
Companion
Animal Human
7%
81%
G
e
n
e
ri
c
d
is
p
e
n
s
in
g
r
a
te
s

36 KindredBio
Exclusivity and IP Position
Full intellectual property protection anticipated for
antibody portfolio
Use and formulation patents for small molecules
20 years of patent protection from date of filing
Regulatory Exclusivity
5 years in U.S.
10 years in E.U.
Lifecycle Management
New formulations, combinations and derivatives

37 KindredBio
Business Development
In active discussions about acquisitions of businesses
and/or assets, particularly of equine assets
Ideal candidate:
Revenue generating/accretive
Commercial infrastructure
Complementary assets

38 KindredBio
Financials

39 KindredBio
Select Summary Financials
$ millions
For the quarter
ended
September 30, 2016
Operating expenses:
Research and development $3.8
General and administrative $2.0
Total cash operating expenses
(excluding stock-based compensation)
$4.8
Total operating expenses
(including stock-based compensation)
$5.8
Total cash, cash equivalents and investments
(As of September 30, 2016)
$62.5
We believe that our cash and equivalents are sufficient to fund operations until
we start generating significant revenues.

40 KindredBio
Share Structure & 6 Month Stock History
19.9 M
Shares
Outstanding
3.6 M Options1
US$107.4 M Market Cap2
US$58.7 M
Working
Capital3
1 As of September 30, 2016; avg. $6.35
2 As of close of market October 31, 2016
3 As of September 30, 2016
Analyst Coverage
Alex Arfaei, BMO Capital Markets
Alex.Arfaei@BMO.com
Kevin DeGeeter, Ladenburg Thalmann
kdegeeter@Ladenburg.com

41 KindredBio
Summary
Validated Drugs and
Targets
$3M-$5M to Market
Multiple Approvals
Starting within 1 year
World Class Team

42 KindredBio
Thank You
