Attached files
| file | filename |
|---|---|
| EXCEL - IDEA: XBRL DOCUMENT - NXT Nutritionals Holdings, Inc. | Financial_Report.xls |
| EX-32.1 - CERTIFICATION PURSUANT TO SECTION 906 OF THE SARBANES-OXLEY ACT - NXT Nutritionals Holdings, Inc. | f10k2011ex32i_nxt.htm |
| EX-32.2 - CERTIFICATION PURSUANT TO SECTION 906 OF THE SARBANES-OXLEY ACT - NXT Nutritionals Holdings, Inc. | f10k2011ex32ii_nxt.htm |
| EX-31.2 - CERTIFICATION PURSUANT TO SECTION 302 OF THE SARBANES-OXLEY ACT - NXT Nutritionals Holdings, Inc. | f10k2011ex31ii_nxt.htm |
| EX-31.1 - CERTIFICATION PURSUANT TO SECTION 302 OF THE SARBANES-OXLEY ACT - NXT Nutritionals Holdings, Inc. | f10k2011ex31i_nxt.htm |
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
|
x ANNUAL REPORT UNDER SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
|
For the fiscal year ended December 31, 2011
|
o TRANSITION REPORT UNDER SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
|
Commission File No. 333-147631
NXT Nutritionals Holdings, Inc.
(Exact name of registrant as specified in its charter)
|
Delaware
|
26 -3186713
|
|
|
(State or other jurisdiction of
incorporation or organization)
|
(IRS Employer Identification No.)
|
|
|
933 E. Columbus Avenue
Suite C
Springfield, MA
|
01105
|
|
|
(Address of principal executive offices)
|
(Zip Code)
|
(413) 533-9300
(Registrant’s telephone number, including area code)
|
Securities registered under Section 12(b) of the Exchange Act:
|
||
|
Title of each class registered:
|
Name of each exchange on which registered:
|
|
|
None
|
None
|
|
|
Securities registered under Section 12(g) of the Exchange Act:
Common stock, par value $.001
|
||
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes o No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes o No x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No o
1
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T(§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). x Yes o No
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§ 229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act.
|
Large accelerated filer
|
o
|
Accelerated filer
|
o
|
|
|
Non-accelerated filer
(Do not check if a smaller reporting company)
|
o
|
Smaller reporting company
|
x
|
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act).
Yes o No x
State the aggregate market value of the voting and non-voting common equity held by non-affiliates computed by reference to the price at which the common equity was last sold, or the average bid and ask price of such common equity, as of the last business day of the registrant’s most recently completed second fiscal quarter: $3,415,993.
As of April 16, 2012, the registrant had 73,953,106 shares of its common stock issued and outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
None
2
TABLE OF CONTENTS
|
PAGE
|
|||||
|
Forward Looking Statements
|
|||||
|
PART I
|
|||||
| 4 | |||||
| 12 | |||||
| 19 | |||||
| 19 | |||||
| 19 | |||||
| 20 | |||||
|
PART II
|
|||||
| 21 | |||||
| 22 | |||||
| 22 | |||||
| 28 | |||||
| 29 | |||||
| 30 | |||||
| 30 | |||||
|
PART III
|
|||||
| 33 | |||||
| 38 | |||||
| 41 | |||||
| 44 | |||||
| 45 | |||||
|
PART IV
|
|||||
| 46 | |||||
| 48 | |||||
3
FORWARD-LOOKING STATEMENTS
This report contains information that may constitute "forward-looking statements." Generally, the words "believe," "expect," "intend," "estimate," "anticipate," "project," "will" and similar expressions identify forward-looking statements, which generally are not historical in nature. However, the absence of these words or similar expressions does not mean that a statement is not forward-looking. All statements that address operating performance, events or developments that we expect or anticipate will occur in the future — including statements relating to volume growth, share of sales and earnings per share growth, and statements expressing general views about future operating results — are forward-looking statements. Management believes that these forward-looking statements are reasonable as and when made. However, caution should be taken not to place undue reliance on any such forward-looking statements because such statements speak only as of the date when made. We undertake no obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law. In addition, forward-looking statements are subject to certain risks and uncertainties that could cause actual results to differ materially from our historical experience and our present expectations or projections. These risks and uncertainties include, but are not limited to, those described in Part I, "Item 1A. Risk Factors" and elsewhere in this report and those described from time to time in our future reports filed with the Securities and Exchange Commission.
PART I
ITEM 1. BUSINESS
OVERVIEW
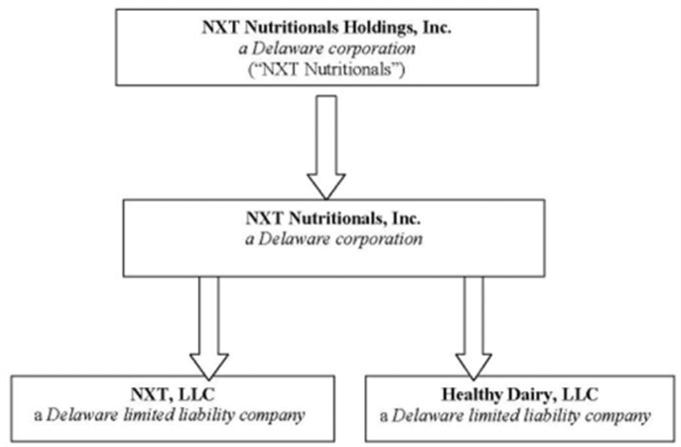
NXT Nutritionals Holdings, Inc. is a Delaware corporation incorporated in 2006 under the name Goldvale Resources, Inc. We were originally organized to search for mining properties in British Columbia.
In February 2009, we completed a share exchange with NXT Nutritionals, Inc. and the shareholders of NXT Nutritionals, pursuant to which we acquired all of the issued and outstanding common stock of NXT Nutritionals from the NXT Shareholders. In exchange, we issued to the NXT Shareholders 22,480,000 shares of our common stock, representing 63.06% of our outstanding shares of common stock (the “Share Exchange”).
As a result of the closing of the Share Exchange, NXT Nutritionals became our wholly-owned subsidiary. We also became of the indirect owner of NXT, LLC and Healthy Dairy, LLC, two subsidiaries of NXT Nutritionals.
Operating through NXT Nutritionals, we are engaged in developing and marketing of a proprietary, patent-pending, all–natural, healthy sweetener sold under the brand name SUSTA™ and other food and beverage products. SUSTA™ is being sold as a stand-alone product and it is the common ingredient for substantially all of our products. We also market and sell Healthy Dairy® non-fat yogurt smoothies, which are enhanced by the revolutionary taste and nutritious ingredients contained in SUSTA™. Our mission is to provide consumers with unique, healthy and delicious products that promote a healthier lifestyle, especially for those concerned with obesity and diabetes.
Our current goal is to bring SUSTA™ to the retail marketplace nationwide, expand the Healthy Diary® product line by focusing on the food service channel, and eventually to expand the Healthy Dairy® to include product lines such as cup yogurt and ice cream.
Our current corporate structure is illustrated in the figure below:
4
PRODUCTS
SUSTA™ Natural Sweetener
SUSTA™ is a natural, healthy sweetener that has minimal calories and low glycemic index. It is a proprietary blend of inulin fiber, fructose, botanical extracts, natural flavors, vitamins, minerals, and probiotics that is patented in New Zealand and patent-pending in the U.S. and Canada.
SUSTA ™ is used in coffees, teas, other beverages, cereals, and any other food that requires a sweetener. SUSTA™ is targeted at individuals craving sweeteners but not the calories from sugar, and is a better option than sugar for people with diabetes because it has approximately one-half gram per serving of fructose and a glycemic index that is one-third of regular sugar.
SUSTA™ sweetens the taste of food without the concerns with table sugar or chemicals. It also contains healthy probiotics consisting of 100 million bacillus coagulas spores per serving. In addition, SUSTA ™ contains a vital dietary fiber and antioxidants and key cellular nutrients such as B-Vitamins, Chromium, and Fiber which help to increase metabolism by making insulin more effective through the direction of sugars to energy rather than fat, and steering sugar into energy metabolism. The Dietary Fiber in SUSTA ™ is soluble with over 1 gram per serving and the antioxidant sources are from nutrients consisting primarily of Vitamin-C, Mineral Selenium, Chromium, Cinnamon, Bitter Melon, Goji, and Grape Seed. These nutrients provide a full spectrum of Flavanol and bioactive compounds at a 20:1 ratio of concentration. Lastly, SUSTA™ simultaneously supports a healthy heart, bones, and immune system by converting Fiber into short chain fatty acids which further accelerates the absorption of important minerals such as Zinc, Calcium, and Magnesium from food.
5
We are seeking to market SUSTA™ in three primary categories:
• SUSTA™ as a table top sweetener alternative to sugar and other sweeteners,
• SUSTA™ as an ingredient used in beverages, cereals, baked goods, dairy products, candy and chewing gum, and
• NXT/SUSTA™ branded products including Healthy Dairy® and other SUSTA™ branded products to be launched by the Company.
Brand awareness of SUSTA™ will be driven by an aggressive marketing campaign and substantial incentive trial program. Additionally, more traditional levers in the retail sales channel like advertising, trade incentives, price promotions, couponing, and demonstrations will be employed. We target consumer food and beverage companies to incorporate SUSTA™ into their products to provide a healthy alternative to sugar, artificial sweeteners and other natural sweeteners that do not provide the nutritional and health benefits of SUSTA™.
Healthy Dairy® Yogurt Smoothies
We have developed a line of SUSTA™ -enhanced, non-fat reduced-calorie yogurt smoothies, which are marketed as Healthy Dairy®. Healthy Dairy® products combine appealing packaging, the health benefits of SUSTA™, and great taste. They are offered in 5 different flavors: strawberry, peach, mixed berry, tropical fruit, and strawberry-banana. Healthy Dairy® contains cultured pasteurized skim milk, real fruit, pure cane sugar, soluble fiber, tapioca starch, natural flavors, phytosterols (plant sterols), SUSTA™, antioxidant botanicals, and many beneficial vitamins totaling 170 calories per bottle for our 10-ounce bottle. Healthy Dairy® contains SUSTA™ and important nutrients that promote health and wellness with outstanding taste and sweetness with significantly fewer calories than the Company’s key competitors. We have shifted our focus on sales of Healthy Dairy® products from grocery chains to the food service category. We have developed a 7-ounce version of our Healthy Dairy® smoothie line as well as a line of 4-ounce yogurt cups branded under the Health Dairy trademark. These products and the 10-ounce Healthy Dairy® smoothies are being marketed and sold to the food service industry including but not limited to college campuses, the U.S. Military, airlines, elementary schools and restaurants. Our management believes this change in our business model will make us profitable in the future.
Product Sales Below is a breakdown of the gross sales for NXT Nutritionals for the 2011 and 2010 annual reporting periods. The gross sales amounts are broken down by product, Healthy Dairy ("HD") and SUSTA for each period when applicable.
|
HD 2010 Sales:
|
$
|
93,683
|
||
|
SUSTA 2010 Sales:
|
386,157
|
|||
|
Total 2010 Sales:
|
$
|
477,840
|
|
HD 2011 Sales:
|
$
|
151,802
|
||
|
SUSTA 2011 Sales:
|
150,993
|
|||
|
Total 2011 Sales:
|
$
|
302,795
|
||
OPERATIONS
The Company operates as a sales and marketing company that out-sources the distribution and manufacturing of its products. Our officers and directors are seasoned veterans of the food and beverage industry that support and supervise an in house staff and a national broker system. We drive sales to the grocery system and create marketing programs to drive consumer trial and purchase. Additionally we have hired a public relations company to drive brand awareness.
Research and development is performed by contract with one of the Company’s founders Dr. Richard Kozenko and Vickie Babcock, a food technologist. The R&D team also performs quality control of the products for the Company.
6
Our internal staff manages our accounts receivable and accounts payable under the supervision of the CEO, COO, and CFO. They also arrange for the purchase of raw materials and the production of products.
SUSTA Natural sweetener is blended and packaged by a contract packer Subco Foods, Inc., which operates three production facilities in the Midwestern part of the United States. The Healthy Dairy Smoothies are made at Neoga Dairy, Inc. in Farmingdale New York.
SUSTA is distributed by a group of distributors that include C&S Wholesale Grocers, Inc. (“C&S”), White Rose Food (“White Rose”), Bozzuto’s, Inc. (“Bozzuto’s”) and United Natural Foods, Inc. (“UNFI”). The Company also sells direct to several grocery chains. We expect that the Healthy Dairy® branded products will be distributed by large scale distribution companies.
MARKET OPPORTUNITIES
With a significant growth market, SUSTA™ is much more than a next generation standalone sweetener. We will use this natural product as the basis for a growing line of healthy food products. Healthy Dairy® is a SUSTA™-enhanced product. With SUSTA™ as the base sweetener, we believe that there is an almost unlimited number of products that we can produce or license that will have immediate appeal to the growing number of refined averse and health conscious consumers.
We have encountered a decline since 2008 in the sales of Healthy Dairy® smoothies due to the change in our business focus from sales to grocery stores to the food service category. Our management believes this change in our business model will make us profitable in the future.
SUSTA™ Natural Sweetener
SUSTA™ is well-positioned to be a competitive natural alternative sweetener in the U.S. table top sweetener market. The rise in type-II diabetes and the epidemic proportion of obesity have resulted in an increased market demand for minimal calorie, natural and healthy sweeteners. SUSTA™ is uniquely formulated to take advantage of this demand, and is targeted at individuals craving sweeteners but neither not the calories from sugar nor the use of common artificial sweeteners.. It is a better option than sugar for people with diabetes because it has approximately one-half gram per serving of fructose and a glycemic index that is one-third of regular sugar. Our target demographics include:
|
·
|
Diabetics, individuals on diets, and those proactively managing obesity. Given today’s environment, with the increase in obesity and diabetes, this is a mass market product.
|
|
·
|
Head-of-household moms who bring healthy choices into the household. These moms, who range in age from 20 to 50 years old, make the key food purchase decisions for the household, are employed, and are concerned about health and fitness for their families.
|
|
·
|
Consumers who shun synthetic sweeteners. This list is headed by consumers who have an affinity for natural products, many of whom are fanatical as it relates to purchasing natural products. Consumers are increasingly aware that “you are what you eat” – this awareness has led to a change in consumer behavior as evidenced by the growth of the organic and all natural food sector.
|
|
·
|
Individuals that have active lifestyles and are wellness advocates who tend to be sports enthusiasts, such as runners, bikers, climbers, walkers and devoted exercisers.
|
Healthy Dairy® Yogurt Smoothies
Healthy Dairy®, was previously sold in thousands of stores in 14 states. Currently, Healthy Dairy® yogurt cups, and smoothies in both 10 and 7 ounce portions are being marketed to the food service channel which includes the military, airlines, school systems and restaurants. We have shifted our focus on sales of Healthy Dairy products from grocery chains to the food service category because our management believes this change in our business model will make us profitable in the future. The drinkable yogurt market is a 6% share ($221 Million) of the $3.5 Billion yogurt market, and is growing annually by 5% in the U.S. and 10% globally. In addition, the Healthy Dairy® production line can be extended to launch new products such as Healthy Dairy® ice creams which will generate more market opportunities for the Company.
7
MARKETING STRATEGY
Our initial efforts are focused on launching SUSTA™ into the U.S. table top sweetener market. We have targeted the natural sweeteners segment because it is less entrenched and has less formidable competitors. Brand awareness of SUSTA™ will be driven by an aggressive marketing and public relations campaign and a substantial sampling program. Additionally, more traditional levers like advertising, trade incentives, price promotions, couponing, and demonstrations will be employed. Because of the health benefits of SUSTA™, especially for diabetics, we will partner with a number of health organizations. Consumers will be able to buy SUSTA™ products online with a percentage of each sale allocated toward such charitable organizations with whom we partner. We have engaged three high profile celebrities, including Eddie George, Blair Underwood and Dara Torres, as our spokespersons who will help drive awareness of SUSTA™ by appearing in commercials, making public appearances, heading our cause marketing campaign and appearing on popular television shows.
Because an increasing number of consumers are learning and searching for information on the internet rather than via traditional advertising on TV or in print media, we will take advantage of social networking sites to enhance SUSTA™ brand awareness. Health Forum and SUSTA™ recipes contests will be part of our on-line strategy.
We have also developed a medical advisory board consisting of Dr. Paul S. Auerbach, James R. Gavin III, MD, PhD and Camillo Ricordi, M.D., whose mission is to advise us on scientific advances regarding our products as well as to communicate the health benefits of SUSTA™ to consumers.
DISTRIBUTION
We initially distributed our Healthy Dairy® through the mainstream grocery channel and eventually offered the products through several thousand locations up and down the East Coast. However, during the 2010 fiscal year, we have changed our business model from selling Healthy Dairy® to grocery stores to focusing on selling Healthy Dairy® to the food service category. We have begun shipments to the United States Naval Academy and the United States Air Force Academy. Currently, we do not have our Healthy Dairy product in any grocery stores.
We are seeking to distribute SUSTA™ through grocery stores. Leading brokers in the country, including C&S, White Rose, Bozzuto’s and UNIF, have agreed to carry SUSTA™. We believes that we can utilize the relationship developed through our sales of Healthy Dairy® to allow us to quickly scale up our marketing efforts with SUSTA™. We will continue to sell SUSTA™ natural sweetener and SUSTABOWL™ (for baking) to grocery chains and to sell SUSTA™ and SUSTABOWL™ to the food service market.
The chart below indicates the stores currently carrying SUSTA™ :
8

SUSTA™ and SUSTABOWL™ continue to be marketed to and sold in grocery stores and we continue to expand the number of locations in which these products are sold.
COMPETITION
SUSTA™ Natural Sweetener
SUSTA™’s major competitors in the table top sweetener market are:
|
·
|
Splenda (61% market share)
|
|
·
|
Equal (13% market share)
|
|
·
|
Sweet’n Low (13% market share)
|
|
·
|
Other (13% market share). This includes a variety of competitors – best known brands/producers in this segment include NutraSweet, ADM, & Cargill
|
(Source: IRI – Information Resources International)
Although we compete against all of the products and companies listed, there are major differences between our SUSTA™ and the competitive field in the retail marketplace for alternative sweeteners. SUSTA™ is an all-natural, low calorie sweetener that contains a proprietary blend of probiotics with 100 million Lacto Bacillus Coagulas Spores per 2 gram serving. SUSTA™ also contains botanical extracts including Cinnamon, Bitter Melon, Goji and Grape Seed, which provide a full spectrum of Flavanol and bioactive compounds at a 20:1 ratio of concentration. SUSTA™ also contains Dietary Soluble Fiber at over 1 gram per serving and antioxidant sources from nutrients such as Vitamin-C primarily with Mineral Selenium and Chromium that is unique in the retail marketplace today.
SUSTA™ as a nutritional sweetener aids in digestion and supports the immune system, is formulated with fiber, nutrients, antioxidants and probiotics and is low glycemic. SUSTA™ is like a sugar-shield in the food we eat because the presence of fiber will create less fatty lipids and make blood sugar more stable. The name of fiber used in SUSTA™ is an oligofructose (OFS), or similar forms named as insulin FOS, or oligosaccharides. Recent studies have indicated that a high fructose diet has a pro-oxidant effect in rats compared with a starch-based diet and OFS has already been shown to decrease plasma lipids in rats. Feeding rats a diet supplemented with OFS lowered TG plasma concentrations and prevented TG accumulation induced by fructose in the liver. Dietary OFS also modified the kinetic absorption of dietary carbohydrate, leading to modifications in lipid and glucose concentrations. Thus, lower glucose and insulin concentrations may contribute to the reduction in hepatic fatty acid and TG synthesis and thus to the hypolipidemic effect of OFS.(Source: Oligofructose Protects again the Hypertriglyceridemic and Pro-oxidative Effects of a High Fructose Diet in Rats, The American Society for Nutritional Sciences J. Nutr., June 2003). SUSTA™ works with the body naturally to help tame and transform problem-making, fast absorbing sugars and carbohydrates, into slower absorbing, healthier protected energy because the fiber in SUSTA™ changes the nature of how sugar is metabolized in the liver by reducing levels of blood lipids as compared to controls. The SUSTA™ research funded by us in 2004 at University of Scranton and performed by Joe Vinson, Ph.D. from University of Scranton Department of Chemistry and Food Science, demonstrated key ratios of agent to sugar in modifying blood sugar under the delta curve and the rate of return to baseline blood sugar levels. The research was performed with six double blind cross-over, healthy fasted student objects, who consumed a 70g sugar loaded test beverage with 6 blood draws over three hours. Dr. Joe Vinson was not affiliated with us. The University of Scranton Department of Chemistry and Food Science was a nationally respected research department in food science. The research demonstrated that there was average of 331/3% less area for blood glucose under the delta curve in a 70g sugar beverage with SUSTA™ agent at an 8:1 ratio of sugar to agent as compared to control. In production formula at a 3:1 sugar to fiber ratio, there was 88% less area blood glucose under the delta curve as compared to control, and return to baseline blood sugar occurred in 15 minutes as compared to 30 minutes for control, which suggests less time of circulating insulin to clear lingering elevated blood glucose. These findings are the basis of structuring SUSTA™ which is a 2g serving size, with a ratio of twice or more fiber to fructose.
9
In addition, the fiber in SUSTA™ influences gut hormones to reduce the release of gut glucogen which in turn modulates the release of glucogen which is designed to raise blood sugar. Intestinal hormones stimulate more than 50% of the insulin response after oral glucose administration. Short chain fatty acids stimulate mucosal adaptation and may alter proglucagon messenger RNA and release of the insulin secretagogue, glucagon-like peptide-1 (GLP-1). Sprague- Dawley rats ingested a fiber-free elemental diet or an elemental diet supplemented with 30% fiber providing similar energy and nutrients for 14 days. The cecal and colonic short chain fatty acids contents were significantly higher in the 30% fiber group. Ileal proglucagon messenger RNA levels were significantly higher in the 30% group vs. the 0% group (11.47 +/- 0.87 vs. 6.52 +/- 0.87 densitometer units), respectively. Similar trends were seen in the colon (13.36 +/- 1.0 vs. 10.90 +/- 0.77 densitometer units; P = 0.07). Plasma GLP-1, insulin, and C peptide levels 30 min postoral glucose were significantly higher in the 30% fiber group vs. the 0% group (19.8 +/- 1.2 vs. 15.4 +/- 1.2 pg/ml, 2.67 +/- 0.4 vs. 1.29 +/- 0.5 ng/ml, and 964.4 +/- 94.4 vs. 530.2 +/- 120.4 pM, respectively). Plasma glucose and glucagon did not differ between groups. A diet supplemented with fiber is able to significantly alter proglucagon gene expression and modulate GLP-1 and insulin secretion. These novel findings deepen our understanding of the beneficial role of fiber in improving glucose homeostasis. (Source: Dietary fiber modulates intestinal proglucagon messenger ribonucleic acid and postpradndial secretion of glucagon-like peptide-1 and insulin in rats, Endocrinology, Vol 1996).
SUSTA™ provides vital dietary fiber, antioxidants and key cellular nutrients that are needed for a smoother running, more calorie efficient metabolism. And importantly, SUSTA™ nutrition supports the health of the bones, heart, and immune system by converting fiber into short chain fatty acids which further accelerates the absorption of important minerals such as zinc, calcium, and magnesium from food. The gut bacteria will ferment the fiber provided by SUSTA™ into short chain fatty acids, the fermentation process not only helps to create healthy gut flora, thus calling the fiber a “periodic,” but in the process, creates a slightly acidic environment of organic acids in the gut that help facilitate the absorption process of dietary minerals related to the nutritional dynamics of bones and teeth. As mentioned above, soluble fiber, as OFS in SUSTA™, has a modulation effect on the de novo synthesis of triglycerides and cholesterol which are important markers in heart disease. In addition, SUSTA™, contains Vitamin C along with the key B-vitamins noted in enhancing of homocysteine control-folic acid, B12.B6, Homocysteine is a risk factor in a growing number of diseases including heart disease and various forms of bone diseases
Management views Splenda, Equal, Sweet’N Low and Truvia (the most prominent of the all natural brands based on the Stevia plant) as its main competitors. We will position SUSTA™ as a healthy, all natural, low calorie alternative to sugar and artificial sweeteners with our “Naturally Healthy, Naturally Sweet” campaign.
While McNeil Nutritionals has established Splenda as the dominant brand in the space, our management believes that with our differentiation marketing plan, which includes celebrity marketing, public relations, a major cause marketing program, and a massive sampling program, we will be ideally positioned to a) start off by taking market share from other all-natural sweeteners that do not have the features and health benefits of SUSTA™, and b) after establishing SUSTA™, we will begin to take market share from Splenda and the other major artificial sweetener brands. Executing this plan on a nationwide basis will be contingent on the Company’s ability to continue to raise capital.
10
Healthy Dairy® Yogurt Smoothies
Healthy Dairy® contains plant sterols, which may reduce the risk of heart disease and help lower cholesterol when combined with a low saturated fat and cholesterol diet.
We believe that today’s food service industry is concerned with living longer and providing healthier lives. As a result, we believe the food service industry will gradually switch to Healthy Dairy® as consumer awareness of our products increases upon the launch of our advertising, celebrity marketing and public relations campaigns. Our product has high nutritional content, no fat and is low in calories.
Claims from Competitor Regarding SUSTA
We have received a letter from one of our competitors in which the competitor claimed that a sample of the SUSTA product contained a non-natural product and therefore was misbranded within the meaning of the Federal Food, Drug, and Cosmetic Act. In particular, the competitor claimed that it tested a sample of the SUSTA product and found that the sample contained sucralose. Sucralose is an artificial sweetener which is found in a wide variety of consumer food products and is approved by the Food and Drug Administration.
The Company formulated SUSTA to be ”natural,” without any added synthetic ingredients. This formulation of SUSTA does not include sucralose. The Company requires all of its suppliers to certify that the ingredients that they provide to the Company are free of any non-natural ingredients, including sucralose and the Company has received written assurances from its suppliers that all of the ingredients that comprise SUSTA and SUSTABOWL are natural.
The Company has attempted to investigate the claim made by the competitor. As part of this investigation, the Company requested a copy of the test results obtained by the competitor from its in-house laboratory. However, the competitor has refused to provide the data that supports the alleged test results. As such, the Company is unable to fully investigate the accuracy of the claim.
The Company has also undertaken independent testing of samples of its products to determine whether any of them contain sucralose. Based on that preliminary testing, which have been shared with the competitor, SUSTA and SUSTABOWL are not mislabeled. The Company contacted its suppliers which have confirmed that there is no sucralose added to any part of the ingredients provided by the suppliers. One supplier is continuing to review its internal procedures to ascertain why any trace of sucralose may have been found in any specific samples. At the present time, the Company has been unable to conclude whether there is any trace of sucralose in any of the Company’s products which have previously been sold to customers as well as in the Company’s existing inventory. Further scientific testing under the supervision and direction of expert legal counsel is underway.
If the Company determines that SUSTA products are mislabeled, the Company would take appropriate steps to remedy the matter, which may include a recall of some or all of such products at some level of distribution. If the Company were required to undertake a recall, the recall could have a material adverse effect on the Company’s financial condition and results of operations, including potential loss of reputation and future sales.
INTELLECTUAL PROPERTY
Patent
Following years of research and development, SUSTA™ has been developed into a proprietary formulation that is patented in New Zealand and patent-pending in the United States and Canada. A patent for the formulation was granted in Australia but has subsequently lapsed. The Company intends to pay the fee to have the patent reinstated in the near future. The patent for SUSTA™ is held by NXT, LLC, a wholly owned subsidiary of NXT Nutritionals, Inc.
The SUSTA™ provides a carbohydrate modifying formulation or agent of synergistic ingredients, pertaining to the metabolism of mono and disaccharides. Metabolically, the formulation of the invention slows the absorption of sugars, modifies the release of insulin, and stabilizes blood sugar response. Additionally, the oral ingestion of the formulation of the SUSTA™ prevents or reduces the formation of dental caries by inhibiting the metabolic capability of dental plaque-forming bacteria to convert sugars into erosive, tooth-decaying acids.
The formulations of SUSTA™ provide direct and indirect positive effects on sugar metabolism and blood sugar response. Thus, the formulations of the invention, when consumed in normal amounts, do not adversely contribute or aggravate such conditions as obesity, diabetes, or dietary-based, hormone related hyperactivity such as that often described in young children.
A formulation of SUSTA™ may be in liquid or dry form. That is, it may be in the form of a powder that comprises or contains the formulation, or in the form of a liquid, either an aqueous liquid or a non-aqueous liquid. In one preferred aspect, the invention provides a finished, water-based beverage, into which the formulation of the invention is incorporated. Moreover, SUSTA™ provides a finished water-based beverage, which is acidified and which includes a formulation of the invention.
SUSTA™ also includes a method of slowing absorption of sugars, for instance, from the intestine of a subject (including but not limited to a human individual), that comprises administering to the subject, or making available for ingestion by the mammal, a formulation of the invention. The formulation becomes effective when in an aqueous medium, which may be provided extrinsically, for instance by oral or intravenous administration or ingestion of an aqueous liquid containing the formulation, or intrinsically, for instance by ingestion of a solid formulation of the invention which is acted on by the body’s digestive secretions and conveyed to and through the body’s digestive system (an aqueous environment).
We believe that the additional objects and advantages of SUSTA™ include the following::
The SUSTA™ provides a formulation having desirable properties built upon synergistic ingredients; maintaining low simple sugar levels; and slowing down the normally rapid absorption of simple sugars from the gut. This objective best optimizes energy levels by thwarting the potential destabilizing effects on blood sugar and inulin response, by preferably utilizing a polysaccharide matrix of complex carbohydrates and soluble gum fibers.
SUSTA™ provides numerous advantages not found in other agents including, but not limited to, limiting the effects of excessive use of ingredients, such as sugar, that may promote greater oxidative stress and actually reduce energy. Ingredients are preferably chosen from among those that neutralize and inhibit free radical production and oxidative stress and, therefore, help to protect the cellular energy generating mechanisms. Moreover, presently preferred ingredients are those that assist in the cellular utilization and burning of fuels for energy. The composition of SUSTA™ also provides multiple tiered uses of various timed caloric energy fuels plus the sweetness system disclosed herein for longer, sustained energy.
Trademark
NXT Nutritionals also owns several registered trademarks, including Healthy Dairy®, which applies to its all-natural line of dairy products including our Healthy Dairy® non-fat yogurt smoothies, and SUSTA: Turns Calories into Healthy Energy®. The Healthy Dairy® trademark is held by Healthy Dairy, LLC, a wholly owned subsidiary of NXT Nutritionals. The SUSTA: Turns Calories into Healthy Energy® trademark is held by NXT, LLC, a wholly owned subsidiary of NXT Nutritionals.
11
ITEM 1A. RISK FACTORS
In addition to the other information set forth in this report, you should carefully consider the following discussion of “risk factors” which identifies the most significant factors that may adversely affect our business, operations, financial position or future financial performance. The risks described below are not the only risks facing our Company. Additional risks not currently known to us or that we currently deem to be immaterial also may materially adversely affect our business, financial condition or results of operations in future periods.
Risks Relating to Our Business
OUR LIMITED OPERATING HISTORY MAY NOT SERVE AS AN ADEQUATE BASIS TO JUDGE OUR FUTURE PROSPECTS AND RESULTS OF OPERATIONS.
We have a relatively limited operating history. Although we were incorporated on April 25, 2006, we did not begin operation in our current business until we entered into a Share Exchange Agreement with NXT Nutritionals and its shareholders on February 12, 2009. Such limited operating history and the unpredictability of the sweetener, and food and beverage industry makes it difficult for investors to evaluate our businesses and future operating results. An investor in our securities must consider the risks, uncertainties, and difficulties frequently encountered by companies in new and rapidly evolving markets. The risks and difficulties we face include challenges in accurate financial planning as a result of limited historical data and the uncertainties resulting from having had a relatively limited time period in which to implement and evaluate our business strategies as compared to older companies with longer operating histories.
OUR INDEPENDENT AUDITOR HAS RAISED DOUBT ABOUT OUR ABILITY TO CONTINUE AS A GOING CONCERN.
As reflected in our audited financial statement for the year ended December 31, 2011, we generated a net loss of $(1,335,706) during the year ended December 31, 2011, and as of that date, our current liabilities exceeded our current assets resulting in a working capital deficit of $(2,287,655). In addition, we also had a stockholder’s deficit of $(11,777,534) at December 31, 2011. As a result, our independent auditor expressed substantial doubt as to our ability to continue as a going concern.
WE NEED ADDITIONAL FINANCING TO EXECUTE OUR BUSINESS PLAN. IF WE DO NOT OBTAIN ADDITIONAL FINANCING IT COULD HAVE A MATERIAL ADVERSE EFFECT ON OUR BUSINESS.
Our cash from operations is not adequate to support our expansion and product development programs at this time. We will need substantial additional funds to:
|
·
|
effectuate our business plan and expand our product line;
|
|
·
|
file, prosecute, defend and enforce our intellectual property rights; and
|
|
·
|
produce and market our products.
|
There are no assurances that future funding will be available on favorable terms or at all. If additional funding of up to $500,000 is not obtained, we will need to reduce, defer or cancel development programs, planned initiatives, or overhead expenditures to the extent necessary. The failure to fund our capital requirements could have a material adverse effect on our business, financial condition and results of operations. Moreover, the sale of additional equity securities to raise financing could result in additional dilution to our stockholders and the incurrence of indebtedness would result in increased debt service obligations that could result in operating and financing covenants that would restrict our operations.
12
NEWLY DEVELOPED PRODUCTS MAY NOT BE COMPATIBLE WITH MARKET NEEDS RESULTING IN AN ADVERSE EFFECT ON OUR SALES AND EARNINGS.
Our business is particularly subject to changing consumer trends and preferences. Our continued success depends in part on our ability to anticipate and respond to these changes, and we may not respond in a timely or commercially appropriate manner to such changes. If we fail to invest in extensive market research on consumer health needs in each market we target, we may face limited market acceptance of our products, which could have a material adverse effect on our sales and earnings.
WE HAVE SHIFTED OUR MARKETING AND SALES FOCUS OF HEALTHY DAIRY®, OUR SUSTA™ ENHANCED NON-FAT REDUCED CALORIE YOGURT SMOOTHIES AND YOGURT CUPS, FROM SALE IN GROCERY CHAINS TO THE FOOD SERVICE CATEGORY.
We have shifted our focus from sales of Healthy Dairy® products from grocery chains to the food service category. However, to date we have only begun to sell Healthy Dairy® to the U.S. Naval Academy and the U.S. Air Force Academy and sales are low as we develop this distribution channel. We had previously generated all of our revenues of Healthy Dairy® from the sales to grocery chains. Although our management believes that this change in our business model will make us profitable in the future, there is no guarantee that we will be able to implement this change successfully. If we fail to succeed with this new business model it would have a material adverse effect on our sales and earnings
WE MAY ENCOUNTER SUBSTANTIAL COMPETITION IN OUR BUSINESS AND FAILURE TO COMPETE EFFECTIVELY MAY ADVERSELY AFFECT OUR ABILITY TO GENERATE REVENUE.
We believe that existing and new competitors will continue to improve their products and to introduce new products with competitive price and performance characteristics. We expect that we will be required to continue to invest in product development and productivity improvements to compete effectively in our markets. Our competitors could develop more efficient products or undertake more aggressive and costly marketing campaigns than ours, which may adversely affect our marketing strategies and could have a material adverse effect on our business, results of operations and financial condition.
Important factors affecting our ability to compete successfully include:
|
·
|
the taste and flavor of products;
|
|
·
|
trade and consumer promotions;
|
|
·
|
rapid and effective development of new, unique cutting edge products;
|
|
·
|
attractive and different packaging;
|
|
·
|
branded product advertising; and
|
|
·
|
pricing and promotion.
|
In periods of reduced demand for our products, we can either choose to maintain market share by reducing our selling prices to meet competition or maintain selling prices, which would likely sacrifice market share. Sales and overall profitability would be reduced in either case. In addition, we cannot assure that additional competitors will not enter our existing markets, or that we will be able to compete successfully against existing or new competition.
13
WE RELY ON THE SERVICES OF CERTAIN KEY PERSONNEL. IF WE FAIL TO KEEP THEM EMPLOYED IT MAY HAVE A MATERIAL ADVERSE EFFECT ON FULFILLING OUR BUSINESS PLAN.
Our business relies on the efforts and talents of our President and Chief Executive Officer, Francis McCarthy. The loss of Mr. McCarthy’s service could adversely affect the operations of our business. Although we have entered into a two year employment agreement with Mr. McCarthy and Mr. McCarthy has not indicated any intention of leaving us, the loss of his service for any reason could have a very negative impact on our ability to fulfill on our business plan.
WE MAY NOT BE ABLE TO HIRE AND RETAIN QUALIFIED PERSONNEL TO SUPPORT OUR GROWTH AND IF WE ARE UNABLE TO RETAIN OR HIRE SUCH PERSONNEL IN THE FUTURE, OUR ABILITY TO IMPROVE OUR PRODUCTS AND IMPLEMENT OUR BUSINESS OBJECTIVES COULD BE ADVERSELY AFFECTED.
If one or more of our senior executives or other key personnel are unable or unwilling to continue in their present positions, we may not be able to replace them easily or at all, and our business may be disrupted and our financial condition and results of operations may be materially and adversely affected. Competition for senior management and senior technology personnel is intense, the pool of qualified candidates is very limited, and we may not be able to retain the services of our senior executives or senior technology personnel, or attract and retain high-quality senior executives or senior technology personnel in the future. Such failure could materially and adversely affect our future growth and financial condition.
OUR MANAGEMENT CONCLUDES THAT OUR DISCLOSURE CONTROLS AND PROCEDURES WERE NOT EFFECTIVE, WHICH MAY PREVENT US FROM FILING REPORTS REQUIRED BY APPLICABLE LAWS AND REGULATIONS WITHIN THE TIME FRAME REQUIRED BY THE APPLICABLE RULES OF THE SECURITIES AND EXCHANGE COMMISSION.
Our management concludes that our disclosure controls and procedures are not effective to ensure that information required to be disclosed by us in the responses we file or submit under the Exchange Act is recorded, processed, summarized, and reported within the time periods specified in the SEC’s rules due to that the fact that we have not completed the process of formally documenting internal controls. Effective internal controls are necessary for us to provide reliable financial reports and effectively prevent fraud. If we cannot provide reliable financial reports timely or prevent fraud, our business reputation and operating results could be harmed. Failure to achieve and maintain an effective internal control environment could also cause investors to lose confidence in our reported financial information, which could have a material adverse effect on the price of our common stock,
PRODUCT LIABILITY CLAIMS AGAINST US COULD RESULT IN ADVERSE PUBLICITY AND POTENTIALLY SIGNIFICANT MONETARY DAMAGES.
As with other food producers, we are exposed to risks associated with product liability claims if the consumption of our products results in injury or illness. We cannot predict what impact such product liability claims or resulting negative publicity would have on our business or on our brand image. The successful assertion of product liability claims against us could result in potentially significant monetary damages, diversion of management resources and require us to make significant payments and incur substantial legal expenses, although we do carry product liability insurance for potential product liability claims. Even if a product liability claim is not successfully pursued to judgment by a claimant, we may still incur substantial legal expenses defending against such a claim. Finally, serious product quality concerns could result in governmental action against us, which, among other things, could result in the suspension of production or distribution of our products, loss of certain licenses, or other governmental penalties.
14
MARKETING CLAIMS AGAINST US COULD RESULT IN ADVERSE PUBLICITY.
We are exposed to risks associated with marketing claims from our competitors. In this connection, we have received a letter from a competitor claiming that a sample of our SUSTA product contains a non-natural ingredient. This claim is more fully described in Item 1-Business- Claims from Competitor. We are currently investigating this claim and intend to take appropriate remedial action if this claim has any validity. We cannot predict what impact such claim or resulting negative publicity would have on our business or on our brand image. If the claim were accurate, we may need to recall some or all of our product at some level of distribution. If the Company were required to undertake a recall, the recall could have a material adverse effect on the Company's financial condition and results of operations, including potential loss of reputation and future sales.
WE COMPETE IN AN INDUSTRY THAT IS BRAND-CONSCIOUS, AND UNLESS WE ARE ABLE TO ESTABLISH AND MAINTAIN BRAND NAME RECOGNITION OUR SALES MAY BE NEGATIVELY IMPACTED.
Our business is substantially dependent upon awareness and market acceptance of our products and brand by our targeted consumers. In addition, our business depends on acceptance by our independent distributors and consumers of our brand. Although we believe that we have made progress towards establishing market recognition for our brand in the industry, it is too early in the product life cycle of the brand to determine whether our products and brand will achieve and maintain satisfactory levels of acceptance by independent distributors, grocery stores, retailers and consumers.
WE RELY PRIMARILY ON THIRD-PARTY DISTRIBUTORS AND INDEPENDENT RETAILERS, AND THIS COULD NEGATIVELY AFFECT OUR ABILITY TO EFFICIENTLY AND PROFITABLY DISTRIBUTE AND MARKET OUR PRODUCTS, MAINTAIN OUR EXISTING MARKETS AND EXPAND OUR BUSINESS INTO OTHER GEOGRAPHIC MARKETS.
Except for the sales through the internet, we do not sell our products directly to our end customers. We primarily rely on third-party distributors and retailers for the sale and distribution of our products. To the extent that our distributors are distracted from selling our products or do not expend sufficient efforts in managing and selling our products, our sales will be adversely affected. Our ability to maintain our distribution network and attract additional distributors will depend on a number of factors, many of which are outside our control. Some of these factors include: (i) the level of demand for our brand and products in a particular distribution area; (ii) our ability to price our products at levels competitive with those offered by competing products and (iii) our ability to deliver products in the quantity and at the time ordered by distributors.
There can be no assurance that we will be able to meet all or any of these factors in any of our current or prospective geographic areas of distribution. Furthermore, shortage of adequate working capital may make it impossible for us to do so. Our inability to achieve any of these factors in a geographic distribution area will have a material adverse effect on our relationships with our distributors in that particular geographic area, thus limiting our ability to maintain and expand our market, which will likely adversely affect our revenues and financial results.
OUR FUTURE SUCCESS RELIES UPON SUSTA™, ALL-NATURAL, PATENT-PENDING, HEALTHY SWEETENER. THERE IS NO ASSURANCE THAT THESE PATENTS WILL BE GRANTED. EVEN IF THEY ARE GRANTED, THERE IS NO ASSURANCE THAT WE WILL HAVE THE RESOURCES TO ENFORCE THE PATENTS THROUGH LITIGATION OR OTHERWISE. THE LOSS OF EXCLUSIVE RIGHT TO SUSTA™ COULD HAVE A MATERIAL ADVERSE EFFECT ON OUR BUSINESS, FINANCIAL CONDITION AND RESULTS OF OPERATIONS.
We believe that our business does not infringe upon the valid proprietary rights of others, but there can be no assurance that third parties will not assert infringement claims against us. If infringement claims are brought against us, there can be no assurance that we will have the financial resources to defend against such claims or prevent an adverse judgment against us. In the event of an unfavorable ruling on any such claim, there can be no assurance that a license or similar agreement to utilize the intellectual property rights in question relied upon by us in the conduct of our business will be available to us on reasonable terms, if at all. In the event of an unfavorable ruling on any such claim, there can be no assurance that a license or similar agreement to utilize the intellectual property rights in question relied upon by us in the conduct of our business will be available to us on reasonable terms, if at all.
15
SUSTA™ is patent-pending in the United States and Canada. There can be no assurance any of these pending patents will be granted or, even if they are, that we will have the resources to enforce these patents through litigation or otherwise. In addition, patents granted by the United States Patent Office do not guarantee that competitors in overseas locations will not imitate our products, or patent similar products in other nations. The failure to obtain the patent to SUSTA™ may have a material adverse effect on our business operations.
WE MAY NEED ADDITIONAL CAPITAL TO FUND OUR FUTURE OPERATIONS AND, IF IT IS NOT AVAILABLE WHEN WE NEED IT, WE MAY NEED TO REDUCE OUR PLANNED DEVELOPMENT AND MARKETING EFFORTS, WHICH MAY REDUCE OUR SALES REVENUES.
We believe that our existing working capital and cash available from operations will enable us to meet our working capital requirements for at least the next 3 months. The development and marketing of new products and the expansion of distribution channels and associated support personnel requires a significant commitment of resources. In addition, if the markets for our products develop more slowly than anticipated, or if we fail to establish significant market share and achieve sufficient net revenues, we may continue to consume significant amounts of capital. As a result, we could be required to raise additional capital of up to $5,000,000. We cannot assure that additional capital, if required, will be available on acceptable terms, or at all. If we are unable to obtain sufficient amounts of additional capital, we may be required to reduce the scope of our planned product development and marketing efforts, which could harm our business, financial condition and operating results
WE MAY NEVER PAY ANY DIVIDENDS TO SHAREHOLDERS.
We have never declared or paid any cash dividends or distributions on our capital stock. We currently intend to retain our future earnings, if any, to support operations and to finance expansion and therefore we do not anticipate paying any cash dividends on our Common Stock in the foreseeable future.
The declaration, payment and amount of any future dividends will be made at the discretion of the board of directors, and will depend upon, among other things, the results of our operations, cash flows and financial condition, operating and capital requirements, and other factors as the board of directors considers relevant. There is no assurance that future dividends will be paid, and, if dividends are paid, there is no assurance with respect to the amount of any such dividend.
Risks Related to Our Common Stock
OUR COMMON STOCK IS QUOTED ON THE OTC BULLETIN BOARD WHICH MAY HAVE AN UNFAVORABLE IMPACT ON OUR STOCK PRICE AND LIQUIDITY.
Our Common Stock is quoted on the OTC Bulletin Board. The OTC Bulletin Board is a significantly more limited market than the New York Stock Exchange or Nasdaq system. The quotation of our shares on the OTC Bulletin Board may result in a less liquid market available for existing and potential stockholders to trade shares of our Common Stock, could depress the trading price of our Common Stock and could have a long-term adverse impact on our ability to raise capital in the future.
THERE IS LIMITED LIQUIDITY ON THE OTCBB.
When fewer shares of a security are being traded on the OTCBB, volatility of prices may increase and price movement may outpace the ability to deliver accurate quote information. Due to lower trading volumes in shares of our Common Stock, there may be a lower likelihood of one's orders for shares of our Common Stock being executed, and current prices may differ significantly from the price one was quoted at the time of one's order entry.
16
OUR COMMON STOCK IS THINLY TRADED, SO CURRENT SHAREHOLDERS MAY BE UNABLE TO SELL THEIR SHARES AT OR NEAR ASKING PRICES OR AT ALL.
Currently our Common Stock is quoted in the OTC Bulletin Board market and the trading volume we will develop may be limited by the fact that many major institutional investment funds, including mutual funds, as well as individual investors follow a policy of not investing in Bulletin Board stocks and certain major brokerage firms restrict their brokers from recommending Bulletin Board stocks because they are considered speculative, volatile and thinly traded. The OTC Bulletin Board market is an inter-dealer market much less regulated than the major exchanges and our Common Stock is subject to abuses, volatility and shorting. Thus there is currently no broadly followed and established trading market for our Common Stock. An established trading market may never develop or be maintained. Active trading markets generally result in lower price volatility and more efficient execution of buy and sell orders. Absence of an active trading market reduces the liquidity of the shares traded there.
The trading volume of our Common Stock has been and may continue to be limited and sporadic. As a result of such trading activity, the quoted price for our Common Stock on the OTC Bulletin Board may not necessarily be a reliable indicator of its fair market value. Further, if we cease to be quoted, holders would find it more difficult to dispose of, or to obtain accurate quotations as to the market value of our Common Stock and as a result, the market value of our Common Stock likely would decline.
OUR COMMON STOCK IS SUBJECT TO PRICE VOLATILITY UNRELATED TO OUR OPERATIONS.
The market price of our common stock could fluctuate substantially due to a variety of factors, including market perception of our ability to achieve our planned growth, quarterly operating results of other companies in the same industry, trading volume in our common stock, changes in general conditions in the economy and the financial markets or other developments affecting our competitors or us. In addition, the stock market is subject to extreme price and volume fluctuations. This volatility has had a significant effect on the market price of securities issued by many companies for reasons unrelated to their operating performance and could have the same effect on our Common Stock.
IF WE FAIL TO REMAIN CURRENT ON OUR REPORTING REQUIREMENTS, WE COULD BE REMOVED FROM THE OTC BULLETIN BOARD, WHICH WOULD LIMIT THE ABILITY OF BROKER-DEALERS TO SELL OUR SECURITIES AND THE ABILITY OF SHAREHOLDERS TO SELL THEIR SECURITIES IN THE SECONDARY MARKET.
Companies trading on the OTC Bulletin Board, such as NXT Nutritionals, must be reporting issuers under Section 12 of the Exchange Act, and must be current in their reports under Section 13 of the Exchange Act, in order to maintain price quotation privileges on the OTC Bulletin Board. If we fail to remain current on our reporting requirements, we could be removed from the OTC Bulletin Board. As a result, the market liquidity for our securities could be adversely affected by limiting the ability of broker-dealers to sell our securities and the ability of shareholders to sell their securities in the secondary market.
CURRENT SHAREHOLDERS MAY BE SUBJECT TO DILUTION CAUSED BY CONVERSION OF OUR OUTSTANDING CONVERTIBLE DEBENTURE AND/OR EXERCISE OF OUR OUTSTANDING SERIES A, SERIES B AND SERIES C WARRANTS.
In connection with a private offering closed on August 27, 2009, we issued a number of 3-year convertible debentures, representing an aggregate principal amount of $3,173,000, which are convertible into shares of our Common Stock at $0.40 per share at any time during the term of the convertible debentures, a number of Series A Warrants to purchase 100% of the underlying shares of Common Stock of the convertible debenture at an exercise price of $0.40 per share and a number of Series B Warrants to purchase 100% of the underlying shares of Common Stock of the convertible debenture at an exercise price of $0.60 per share. Upon conversion or exercise of the convertible debenture or the Series A and Series B Warrants, in whole or in part, current shareholders will be subject to significant dilution.
17
In connection with a private offering closed on February 26, 2010, we issued a number of 15 month convertible debentures, representing an aggregate principal amount of $5,667,743 with a face amount of $6,517,943, and subsequently modified to $8,438,684, which are convertible into shares of our Common Stock at a modified rate of $0.40 per share at any time during the term of the convertible debentures, a number of Series C Warrants to purchase 100% of the underlying shares of Common Stock of the convertible debenture at a modified exercise price of $0.40 per share. Upon conversion or exercise of the convertible debenture or the Series C Warrants, in whole or in part, current shareholders will be subject to significant dilution.
On November 21, 2011, NXT Nutritionals Holdings, Inc. (the “Company” or the “Registrant”) and NXT Investment Partners, LLC, a Delaware limited liability company (“NIP”) executed a Securities Purchase Agreement (the “SPA”) pursuant to which NIP agreed to make investments (the “Investment”) in the Company in the form of a senior secured loan to the Company in the aggregate principal amount of at least $1,000,000 and up to $1,500,000 (the amount loaned shall be referred to as the “Principal Amount of the 2011 Note”), bearing interest at 13% per annum, in exchange for a four-year 13% Senior Secured promissory note (“2011 Note”). On November 21, 2011, pursuant to the SPA, the Company received an initial investment of $1,000,000 and issued the 2011 Note in the original principal amount of $1,000,000 along with 13,075,468 shares of Series A Convertible Preferred Stock (“Preferred Stock”) of the Company to NXT. The sum of the $1,000,000 initial investment was attributed 20% for the purchase of the 2011 Note and 80% for the purchase of the Preferred Stock. Pursuant to the terms of the SPA, the Company and NXT may agree to an additional investment of up to $500,000. A Preferred Stock Certificate of Designation as attached to the SPA. The Preferred Stock issued under the SPA carries an annual dividend equal to the greater of: (a) 10% of the then outstanding Principal Amount of the 2011 Note as of December 31st of the applicable fiscal year for which the annual dividend is being paid; or (b) 10% of the Net Income (as defined in the 2011 Note) of the Company in excess of $500,000 for the applicable fiscal year of the Company and is convertible at the option of NIP into an equal number of shares of common stock of the Company representing 20% of the Company’s fully diluted capital stock at the time of conversion.
In connection with the closing of the Investment, the Company granted to NIP a security interest in the Company’s assets as collateral as set forth in the Security Agreement dated November 21, 2011, as attached to the SPA. Furthermore, on November 21, 2011 the Company’s subsidiaries also entered into a Subsidiary Guarantee Agreement, as attached to the SPA, to guarantee the Company’s prompt and complete payment and performance when the obligations set forth in the Subsidiary Guarantee are due.
OUR COMMON STOCK IS CLASSIFIED AS A “PENNY STOCK” AS THAT TERM IS GENERALLY DEFINED IN THE SECURITIES EXCHANGE ACT OF 1934 TO MEAN EQUITY SECURITIES WITH A PRICE OF LESS THAN $5.00. OUR COMMON STOCK WILL BE SUBJECT TO RULES THAT IMPOSE SALES PRACTICE AND DISCLOSURE REQUIREMENTS ON BROKER-DEALERS WHO ENGAGE IN CERTAIN TRANSACTIONS INVOLVING A PENNY STOCK.
The SEC has adopted regulations which generally define so-called "penny stocks" to be an equity security that has a market price less than $5.00 per share or an exercise price of less than $5.00 per share, subject to certain exemptions. As a "penny stock," our Common Stock may become subject to Rule 15g-9 under the Exchange Act of 1934, or the "Penny Stock Rule." This rule imposes additional sales practice requirements on broker-dealers that sell such securities to persons other than established customers and "accredited investors" (generally, individuals with a net worth in excess of $1,000,000 or annual incomes exceeding $200,000, or $300,000 together with their spouses).
For any transaction involving a penny stock, unless exempt, the penny stock rules require that a broker or dealer approve a person's account for transactions in penny stocks and the broker or dealer receive from the investor a written agreement to the transaction setting forth the identity and quantity of the penny stock to be purchased. In order to approve a person's account for transactions in penny stocks, the broker or dealer must obtain financial information and investment experience and objectives of the person and make a reasonable determination that the transactions in penny stocks are suitable for that person and that that person has sufficient knowledge and experience in financial matters to be capable of evaluating the risks of transactions in penny stocks.
18
The broker or dealer must also deliver, prior to any transaction in a penny stock, a disclosure schedule prepared by the SEC relating to the penny stock market, which, in highlight form, sets forth:
The basis on which the broker or dealer made the suitability determination, and that the broker or dealer received a signed, written agreement from the investor prior to the transaction.
Disclosure also has to be made about the risks of investing in penny stocks in both public offerings and in secondary trading and commissions payable to both the broker-dealer and the registered representative, current quotations for the securities and the rights and remedies available to an investor in cases of fraud in penny stock transactions. Finally, monthly statements have to be sent disclosing recent price information for the penny stock held in the account and information on the limited market in penny stocks.
Because of these regulations, broker-dealers may not wish to engage in the above-referenced necessary paperwork and disclosures and/or may encounter difficulties in their attempt to sell shares of our Common Stock, which may affect the ability of selling shareholders or other holders to sell their shares in any secondary market and have the effect of reducing the level of trading activity in any secondary market. These additional sales practice and disclosure requirements could impede the sale of our Common Stock. In addition, the liquidity for our Common Stock may decrease, with a corresponding decrease in the price of our Common Stock. Our Common Stock, in all probability, will be subject to such penny stock rules for the foreseeable future and our shareholders will, in all likelihood, find it difficult to sell their common stock.
There can be no assurance that our Common Stock will qualify for exemption from the Penny Stock Rule. In any event, even if our Common Stock were exempt from the Penny Stock Rule, we would remain subject to Section 15(b)(6) of the Exchange Act, which gives the SEC the authority to restrict any person from participating in a distribution of a penny stock if the SEC determines that such a restriction would be in the public interest.
ITEM 1B. UNRESOLVED STAFF COMMENTS
We have received no written comments regarding our periodic or current reports from the staff of the SEC that were issued 180 days or more preceding the end of our fiscal year 2011 that remain unresolved.
ITEM 2. PROPERTIES
Our principal executive offices are located at 933 E. Columbus Avenue, Suite C, Springfield, MA 01105. Currently we do not own any real property. Our office rental expense on a monthly basis is $450. Healthy Dairy, LLC, and NXT, LLC operate from our principal executive office.
ITEM 3. LEGAL PROCEEDINGS
On November 5, 2004, our subsidiary NXT Nutritionals, filed an application to register the SUSTA trademark on the United States Patent and Trademark Office (“USPTO”) Principal Register, which the USPTO approved and published for opposition.
On November 23, 2009, ConAgra Foods Food Ingredients Company, Inc. filed an opposition proceeding before the USPTO’s Trademark Trial and Appeal Board seeking to prevent such registration. In the opposition proceeding, ConAgra claims that the SUSTA mark is almost identical in sight, sound, and commercial impression to ConAgra’s SUSTAGRAIN trademark, such that a likelihood of confusion exists between these two marks. On October 28, 2010, we responded to ConAgra’s Notice of Opposition. As of April 1, 2012, the companies have reached an agreement in principle pursuant to which NXT will have trademark rights in SUSTA Natural Sweetener and SustaBowl natural sweetener. The proceeding pending before the USPTO has been extended by the parties.
19
ITEM 4. MINE SAFETY DISCLOSURES
Not applicable
20
PART II
ITEM 5. MARKET FOR COMMON EQUITY AND RELATED SHAREHOLDER MATTERS
Our shares of common stock are trading on the Over the Counter Bulletin Board (“OTC Bulletin Board”) under the trading symbol “NXTH.OB.” The OTC Bulletin Board is a significantly more limited market than the New York Stock Exchange or Nasdaq system. The quotation of our shares on the OTC Bulletin Board may result in a less liquid market available for existing and potential stockholders to trade shares of our common stock, could depress the trading price of our common stock and could have a long-term adverse impact on our ability to raise capital in the future.
The following table sets forth, for the periods indicated, the high and low bid prices for our common stock on the OTC Bulletin Board as reported by various OTCBB market makers. The quotations do not reflect adjustments for retail mark-ups, mark-downs, or commissions and may not necessarily reflect actual transactions.
|
Quarter Ended
|
High Bid ($)
|
Low Bid ($)
|
||||||
|
Fourth Quarter ended December 31, 2011
|
0.06
|
0.01
|
||||||
|
Third Quarter ended September 30, 2011
|
0.07
|
0.02
|
||||||
|
Second Quarter ended June 30, 2011
|
0.18
|
0.05
|
||||||
|
First Quarter ended March 31, 2011
|
0.27
|
0.14
|
||||||
|
Fourth Quarter ended December 31, 2010
|
0.32
|
0.17
|
||||||
|
Third Quarter ended September 30, 2010
|
0.30
|
0.17
|
||||||
|
Second Quarter ended June 30, 2010
|
0.62
|
0.16
|
||||||
|
First Quarter ended March 31, 2010
|
3.22
|
0.52
|
||||||
Holders
As of April, 2012, there were approximately 68 holders of record of our common stock. This does not reflect the number of persons or entities who held stock in nominee or "street" name through various brokerage firms.
Dividends
To date, we have never declared or paid any cash dividends on our capital stock. We currently intend to retain any future earnings for funding growth and therefore, do not expect to pay any dividends in the foreseeable future. Payment of future dividends, if any, will be at the discretion of our Board of Directors after taking into account various factors, including our financial condition, operating results, current and anticipated cash needs and plans for expansion.
Transfer Agent and Registrar
Our independent stock transfer agent is Corporate Stock Transfer, Inc. at 3200 Cherry Creek South Drive, Suite 430, Denver, Colorado 80209.
21
ITEM 6. SELECTED FINANCIAL DATA
Not applicable.
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITIONS AND RESULTS OF OPERATIONS.
The following discussion is provided as a supplement to – and should be read in conjunction with - the Consolidated Financial Statements and Notes thereto appearing elsewhere in this Form 10-K. The following discussion contains “forward-looking statements” as described in the introduction to this Form 10-K.
BUSINESS
Operating through NXT Nutritionals, we are engaged in developing and marketing of a proprietary, patent-pending, natural, healthy sweetener sold under the brand name SUSTA™ and other food and beverage products. SUSTA™ is being sold as a stand-alone product and it is the common ingredient for all of our products. We also market and sell Healthy Dairy® which is enhanced by the revolutionary taste and nutritious ingredients contained in SUSTA™. Our mission is to provide consumers with unique, healthy, delicious products that promote a healthier lifestyle and combat obesity and diabetes.
We have previously been focused on expanding the distribution of SUSTA™ to the retail marketplace nationwide and, expanding the Healthy Diary® product line from the east coast to nationwide reach, and eventually expanding the Healthy Dairy® to include product lines such as cup yogurt and ice cream. Currently we have changed our business focus of Healthy Dairy away from selling to the grocery chains and to focus on the food service category.
We have undertaken an aggressive marketing campaign and trial program. Additionally, more traditional levers in the retail sales channel like advertising, trade incentives, price promotions, couponing, and demonstrations are being employed. We are targeting consumer food and beverage companies to incorporate SUSTA™ into their products to provide a healthy alternative to sugar, artificial sweeteners and other natural sweeteners that do not provide the nutritional and health benefits of SUSTA™. We also plan to continue to utilize our three high profile celebrity spokespersons, including Eddie George, Blair Underwood and Dara Torres, to help drive awareness of SUSTA™ by appearing in commercials, making public appearances, heading our cause marketing campaign and appearing on popular television shows.
We have funded our operations to date on private placement offerings of our securities. On August 27, 2009, we completed a private offering of an aggregate subscription amount of $3,173,000 through the issuance of investment units to certain accredited investors. Each investment unit had a purchase price of $50,000 and consisted of (i) a three year Debentures in the amount of $65,000 convertible into shares of our common stock at a conversion price of $0.40 per share, (ii) five year Series A Warrants to purchase 100% of the common stock underlying the Debenture at an exercise price of $0.40 per share, and (iii) five year Series B Warrants to purchase 100% of the common stock underlying the Debenture at an exercise price of $0.60 per share.
On February 26, 2010, we closed on a private placement offering by raising total gross proceeds of $5,667,743, through the sale of (i) 0% Original Issue Discount Senior Secured Convertible Notes convertible into shares of our common stock at a conversion price of $1.00 per share, and (ii) a number of five-year Warrants exercisable into a number of shares of common stock equal to 100% of the number of common shares underlying the Notes at an exercise price of $1.25 per share to certain accredited investors. The principal amount of each Note is 115% of the subscription proceeds received.
On September 1, 2010, we entered into a modification and amendment agreement (the “Modification Agreement”) with purchasers holding approximately 87% of the aggregate number of (1) the Notes, (2) the Warrants, and (3) the shares of common stock underlying the Notes and the Warrants, pursuant to which the commencement of monthly redemption date of the Notes is extended to December 1, 2010 and the holders of the Notes and the Warrants, we may now pay the monthly redemption of the Notes in common stock even if the monthly redemption price described in the Notes is less than $0.40. In addition, pursuant to the Modification Agreement, the conversion price of the Notes and the exercise price of the Warrants are both reduced to $0.40 per share.
22
On December 6, 2010 we entered into a second modification and amendment agreement (the “Second Modification Agreement”) with the Purchasers (the “Purchasers”) holding approximately 91% of the aggregate number of (1) the Notes, (2) Series C warrants and (3) the shares of common stock underlying the Notes and the Series C Warrants. Pursuant to the Amendment, the commencement of monthly redemption date of the Notes is extended to September 1, 2011, the maturity date of the Notes is extended to December 31, 2011 and the original issue discount is amended such that the principal amount equals each investor’s subscription amount multiplied by 1.60. In addition the conversion price can be adjusted on the following events:
|
(i)
|
First Quarter 2011 Form 10-Q. If the Company’s filing of its March 31, 2011 Form 10-Q with the Securities and Exchange Commission does not disclose revenue of at least $5 million for the first three months of 2011, then the Conversion Price of the Notes will decrease by $.03 on the fifth (5th) trading day after the Company files its March 31, 2011 Form 10-Q. Notwithstanding the foregoing, if, during the five (5) trading days following the filing of the March 31, 2011 Form 10-Q, the average closing bid price is $.60 or better, the aggregate trading volume of Company common stock is at least 1.5 million shares and all of the shares underlying the Notes may be sold pursuant to an effective registration statement or Rule 144 (and the Company is then in compliance with the current public information required under Rule 144), then no adjustment to the Conversion Price will be made hereunder.
|
|
(ii)
|
Second Quarter 2011 Form 10-Q. If the Company’s filing of its June 30, 2011 Form 10-Q with the Securities and Exchange Commission does not disclose revenue of at least $8 million for the first six months of 2011, then the Conversion Price of the Notes will be adjusted to equal the lesser of (i) the then effective Conversion Price and (ii) ninety (90%) percent of the average closing bid price during the five (5) trading Days following the filing of the June 30, 2011 Form 10-Q, such adjustment, if any, to occur on the fifth (5th) trading day following the Company’s filing of its June 30, 2011 Form 10-Q. Notwithstanding the foregoing, if, during the five (5) trading Days following the filing of the June 30, 2011 Form 10-Q, the average closing bid price is $.60 or better, the aggregate trading volume of Company common stock is at least 1.5 million shares and all of the shares underlying the Notes may be sold pursuant to an effective registration statement or Rule 144 (and the Company is then in compliance with the current public information required under Rule 144), then no adjustment to the Conversion Price will be made hereunder.
|
On November 21, 2011, NXT Nutritionals Holdings, Inc. (the “Company” or the “Registrant”) and NXT Investment Partners, LLC, a Delaware limited liability company (“NIP”) executed a Securities Purchase Agreement (the “SPA”) pursuant to which NIP agreed to make investments (the “Investment”) in the Company in the form of a senior secured loan to the Company in the aggregate principal amount of at least $1,000,000 and up to $1,500,000 (the amount loaned shall be referred to as the “Principal Amount of the 2011 Note”), bearing interest at 13% per annum, in exchange for a four-year 13% Senior Secured promissory note (“2011 Note”). On November 21, 2011, pursuant to the SPA, the Company received an initial investment of $1,000,000 and issued the 2011 Note in the original principal amount of $1,000,000 along with 13,075,468 shares of Series A Convertible Redeemable Preferred Stock (“Preferred Stock”) of the Company to NXT. The sum of the $1,000,000 initial investment was attributed 20% for the purchase of the 2011 Note and 80% for the purchase of the Preferred Stock. Pursuant to the terms of the SPA, the Company and NXT may agree to an additional investment of up to $500,000. The rights and preferences of the Preferred Stock are set forth in the Series A Preferred Stock Certificate of Designation as attached to the SPA. The Preferred Stock issued under the SPA carries an annual dividend equal to the greater of: (a) 10% of the then outstanding Principal Amount of the 2011 Note as of December 31st of the applicable fiscal year for which the annual dividend is being paid; or (b) 10% of the Net Income (as defined in the 2011 Note) of the Company in excess of $500,000 for the applicable fiscal year of the Company and is convertible at the option of NIP into an equal number of shares of common stock of the Company representing 20% of the Company’s fully diluted capital stock at the time of conversion.
23
In connection with the closing of the Investment, the Company granted to NIP a security interest in the Company’s assets as collateral as set forth in the Security Agreement dated November 21, 2011, as attached to the SPA. Furthermore, on November 21, 2011 the Company’s subsidiaries also entered into a Subsidiary Guarantee Agreement, as attached to the SPA, to guarantee the Company’s prompt and complete payment and performance when the obligations set forth in the Subsidiary Guarantee are due.
Additionally, pursuant to a Registration Rights Agreement dated November 21, 2011 delivered pursuant to the SPA, the Company agreed to register 100% of the common shares issuable upon conversion of the Preferred Stock (the “Registrable Securities”) on a Form S-1 (the “Registration Statement”) to be filed with the Securities and Exchange Commission (the “SEC”) on or prior to the 60th calendar day following the date the Company is required to file its Form 10-K for the fiscal year ending December 31, 2011 (the “Filing Date”) and use its best efforts to have it declared effective within 180 calendar days after November 21, 2011 (the “Effectiveness Date”). In the event that the total number of the Registrable Securities exceeds the limitation imposed by the SEC staff under Rule 415, the number of Registrable Securities to be registered will be subject to cut back as provided in the Registration Rights Agreement. Subject to the terms of the Registration Rights Agreement, upon the occurrence of any event that shall incur liquidated damages (each an “Event” and the date of the occurrence of any such Event, the “Event Date”), including, but not limited to, that the initial Registration Statement is not filed prior to the Filing Date or the initial Registration Statement including the Registrable Securities permitted by Rule 415 is not declared effective by the Effectiveness Date, (A) the Company shall pay to NIP an amount in cash, as partial liquidated damages, equal to 0.5% of the aggregate subscription amount paid by NIP pursuant to the SPA for any unregisterable securities then held by NIP, and (B) on each monthly anniversary of the Event Date until the Event is cured, the shall pay to NIP an amount in cash, equal to 1.0% of the aggregate subscription amount paid by NIP pursuant to the SPA for any unregisterable securities then held by NIP, subject to a maximum amount of 6.0% of the aggregate subscription amount paid by NXT pursuant to the SPA.
The term of the SPA additionally requires the holders of at least 65% of the outstanding principal amount of the Company’s 2009 convertible debentures (the “2009 Debentures”) and five year Series A and Series B warrants (collectively, the “2009 Warrants” and together with the 2009 Debentures are referred to as the “2009 Securities”) to agree to and execute the First Amendment (as described below) and the holders of at least 67% of the outstanding principal amount of the Company’s 0% Original Issue Discount Senior Secured Convertible Notes (the “2010 Notes”) to agree to and execute the Fourth Modification Agreement (as described below).
On November 4, 2011, the Company entered into a Fourth Modification and Amendment Agreement (the “Fourth Modification Agreement”) with the holders holding approximately 93.5% percent of the outstanding principal amount of (1) the 2010 Notes, (2) the related Series C warrants (the “Series C Warrants”) and (3) the shares of common stock underlying the Notes and the Series C Warrants. Pursuant to the Fourth Modification Agreement, the holders, upon closing of the Investment, among other things, have agreed to waived their right to monthly redemptions of the Notes and extend the maturity date of the Notes to the date that is 48-months from the Closing Date (as defined in the SPA) of the Investment. In addition, the holders have agreed (i) that the conversion price will be lowered to $0.25, (ii) to a waiver of the anti-dilution provisions and future participation rights set forth in the Notes and Series C Warrants and (iii) to subordinate their security interests securing the 2010 Notes to the senior security interests to be granted by the Company to secure the Investment and to third parties that provide financing for accounts receivables, inventory and raw materials of the Company.
Also on November 4, 2011, approximately 70% of the holders of the outstanding principal amount of the Company’s 2009 Securities entered into a First Modification and Amendment Agreement (the “First Amendment”) pursuant to which, upon closing of the Investment, the 2009 Debentures and the 2009 Warrants of the approving holders were amended and modified, among other things, (i) to reduce the Conversion Price (as defined in the 2009 Debentures) and the Exercise Price (as defined in the 2009 Warrants) to $0.25 per share of common stock, (ii) to waive the anti-dilution provisions of the 2009 Debentures and the 2009 Warrants and (iii) and extend the Maturity Date under the 2009 Debentures to the date that is 48-months from the Closing Date (as defined in the SPA) of the Investment. As a result of the closing of Investment, those holders of the 2009 Securities who did not execute the First Amendment will incur a reduction of the Conversion Price (as defined in the 2009 Debentures) and the Exercise Price (as defined in the 2009 Warrants) of their 2009 Debentures and 2009 Warrants to $0.09 per share of common stock. The Maturity Date under the 2009 Debentures remains unmodified for those holders of the 2009 Securities who did not execute the First Amendment.
24
The Company continues to explore potential expansion opportunities in the industry in order to boost sales, while leveraging distribution systems to consolidate lower costs.
RESULTS OF OPERATIONS
Results of Operations
Summary of Statement of Operations for the Year Ended December 31, 2011 and 2010:
|
Year ended
|
||||||||
|
December 31, 2011
|
December 31, 2010
|
|||||||
|
Sales – net of slotting fees and discounts
|
$
|
237,989
|
$
|
187,516
|
||||
|
Gross Loss
|
$
|
(912,716
|
)
|
$
|
(246,083
|
)
|
||
|
General and Administrative Expenses
|
$
|
(3,535,854
|
)
|
$
|
(3,591,276
|
)
|
||
|
Other Income (Expense) - Net
|
$
|
3,112,864
|
$
|
(13,565,377
|
)
|
|||
|
Net Loss
|
$
|
(1,335,706
|
)
|
$
|
(17,402,736
|
)
|
||
|
Net Loss per Common Share – Basic
|
$
|
(0.04
|
)
|
$
|
(0.38
|
)
|
||
For the year ended December 31, 2011 and 2010, the Company reported a net loss of $(1,335,706) or $(0.04) per share and a net loss of $(17,402,736) or $(0.38) per share, respectively. The change in loss between the year ended December 31, 2011 and 2010 was primarily attributable to the following:
|
·
|
The Company has shifted its Healthy Dairy sales focus from sales to grocery chains to the food service category. We have only recorded a limited number of Healthy Dairy sales since this shift in focus. The Company also launched the natural sweetener product (SUSTA) on April 30, 2009. The Company has experienced limited sales on the SUSTA product. The mix in sales between the yogurt smoothie and the natural sweetener was approximately 50% each during the year ended December 31, 2011 as compared to 80% of sales during the year ended December 31, 2010 was for the natural sweetener. This increase in sales composition toward the yogurt smoothies is due to selling into the military channel during the year ended December 31, 2011.
|
|
·
|
Sales for the year ended December 31, 2011 as compared to the year ended December 31, 2010 increased by 26%. The increase is primarily attributable to selling into the military channel during the year ended December 31, 2011.
|
|
·
|
General and administrative expenses decreased by approximately 1% during the year ended December 31, 2011 as compared to the corresponding year ended December 31, 2010.
|
Other income (expense) - net increased significantly during the year ended December 31, 2011 as compared to the corresponding year ended December 31, 2010. The increase is primarily attributable to an approximately $8.0 million decrease in interest expense. The Company fully accreted the debt discount recorded on the 2010 convertible note offering during 2010. The Company also recorded a loss on debt extinguishment of $9.8 million during the year ended December 31, 2010. The decreases were offset by a major decrease in the change in fair market value of the derivative liability during the year ended December 31, 2011 as compared to the corresponding year ended December 31, 2010.
25
Liquidity and Capital Resources
Going Concern: As reflected in our audited financial statement for the year ended December 31, 2011, we generated a net loss of $(1,335,706) during the year ended December 31, 2011, and as of that date, our current liabilities exceeded our current assets resulting in a working capital deficit of $(2,287,655). In addition, we also had a stockholder’s deficit of $(11,777,534) at December 31, 2011. As a result, our independent auditor expressed substantial doubt as to our ability to continue as a going concern.
The ability of the Company to continue its operations is dependent on management's plans, which include the raising of capital through debt and/or equity markets with some additional funding from other traditional financing sources, including term notes, until such time that funds provided by operations are sufficient to fund working capital requirements.
The Company will require additional funding to meet its working capital obligations and to finance the growth of its current and expected future operations. The Company believes its current available cash along with anticipated revenues are insufficient to meet its working capital needs unless the Company’s obtains additional funding in the near future. There can be no assurance that financing will be available in amounts or terms acceptable to the Company, if at all.
The ability of the Company to continue as a going concern is dependent on its ability to do all or most of the above listed steps. In the event that the Company were unable to obtain additional financing, it is likely that the Company would be required to discontinue operations.
The following table summarizes total current assets, liabilities and working capital at December 31, 2011 compared to December 31, 2010.
|
December 31,
2011
|
December 31,
2010
|
Increase/
Decrease
|
||||||||||
|
Current Assets
|
$
|
467,227
|
$
|
2,212,843
|
$
|
(1,745,616
|
)
|
|||||
|
Current Liabilities
|
$
|
2,754,882
|
$
|
5,377,489
|
$
|
(2,622,607
|
)
|
|||||
|
Working Capital (Deficit)
|
$
|
(2,287,655
|
)
|
$
|
(3,164,646
|
)
|
$
|
(876,991
|
)
|
|||
As of December 31, 2011, we had a working capital deficit of $(2,287,655) as compared to a working capital deficit of $(3,164,646) as of December 31, 2010, a decrease of $(876,991).
The decrease is primarily a result of a decrease in current liabilities, specifically a decrease in derivative liabilities and a decrease in convertible notes, primarily due to the restructure of the 2010 Notes and 2009 Debentures, which extended the maturity dates to 4 years on a significant portion of those instruments, as well as convertible holders of those instruments converting to common stock during the year ended December 31, 2011. These decreases in current liabilities were partially offset by a decrease in current assets, specifically a decrease in cash by approximately $1,217,808.
Net cash used for operating activities for the year ended December 31, 2011 and 2010 was $(2,151,557) and $(3,035,079), respectively. During the year ended December 31, 2011, the Company used cash to build inventories and to further develop the 4 ounce yogurt cup and to market and build brand awareness of the product line. The Company fully reserved for the inventory balance as of December 31, 2011 due to the difficulties with moving the inventory.
Net cash obtained through all financing activities for the year ended December 31, 2011 was $933,749 as compared to $4,628,755 for the year ended December 31, 2010, specifically attributable to the 2010 Convertible Note Offering. During 2011, the Company raised $1,000,000 through the sale and issuance of the 2011 convertible redeemable preferred stock and note. The proceeds were offset by $78,750 in issuance costs.
26
The Company continues to explore potential expansion opportunities in the industry in order to boost sales, while leveraging distribution systems to effect lower costs.
Recent Accounting Pronouncements
In May 2011, the FASB issued ASU No. 2011-04, Fair Value Measurement (Topic 820): Amendments to Achieve Common Fair Value Measurement and Disclosure Requirements in U.S. GAAP and IFRSs. The guidance in ASU 2011-04 changes the wording used to describe the requirements in U.S. GAAP for measuring fair value and for disclosing information about fair value measurements, including clarification of the FASB's intent about the application of existing fair value and disclosure requirements and changing a particular principle or requirement for measuring fair value or for disclosing information about fair value measurements. The amendments in this ASU should be applied prospectively and are effective for interim and annual periods beginning after December 15, 2011. Early adoption by public entities is not permitted. The adoption of this guidance is not expected to have a material impact on the Company’s financial position or results of operations.
Critical Accounting Policies
Our financial statements and related public financial information are based on the application of accounting principles generally accepted in the United States (“GAAP”). GAAP requires the use of estimates; assumptions, judgments and subjective interpretations of accounting principles that have an impact on the assets, liabilities, revenues and expense amounts reported. These estimates can also affect supplemental information contained in our external disclosures including information regarding contingencies, risk and financial condition. We believe our use of estimates and underlying accounting assumptions adhere to GAAP and are consistently and conservatively applied. We base our estimates on historical experience and on various other assumptions that we believe to be reasonable under the circumstances. Actual results may differ materially from these estimates under different assumptions or conditions. We continue to monitor significant estimates made during the preparation of our financial statements.
Our significant accounting policies are summarized in Note 2 of the consolidated financial statements. While all these significant accounting policies impact our financial condition and results of operations, we view certain of these policies as critical. Policies determined to be critical are those policies that have the most significant impact on our financial statements and require management to use a greater degree of judgment and estimates. Actual results may differ from those estimates. Our management believes that given current facts and circumstances, it is unlikely that applying any other reasonable judgments or estimate methodologies would cause effect on our consolidated results of operations, financial position or liquidity for the periods presented in this report.
We believe the following critical accounting policies and procedures, among others, affect our more significant judgments and estimates used in the preparation of our consolidated financial statements:
Use of Estimates, Going Concern Consideration – The preparation of financial statements in conformity with generally accepted accounting principles in the United States of America requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenue and expenses during the reporting period. Actual results could differ from those estimates. Among the estimates we have made in the preparation of the financial statements is an estimate of our projected revenues, expenses and cash flows in making the disclosures about our liquidity in this report. As an early stage company, many variables may affect our estimates of cash flows that could materially alter our view of our liquidity and capital requirements as our business develops. Our consolidated financial statements have been prepared assuming we are a “going concern”. No adjustment has been made in the consolidated financial statements which could result should we be unable to continue as a going concern.
Share-Based Compensation - US GAAP requires public companies to expense employee share-based payments (including options, warrants, restricted stock units and performance stock units) based on fair value. We must use our judgment to determine key factors in determining the fair value of the share-based payment, such as volatility, forfeiture rates and the expected term in which the award will be outstanding.
27
Derivative Financial Instruments - Fair value accounting requires bifurcation of embedded derivative instruments such as conversion features in convertible debt or equity instruments, and measurement of their fair value for accounting purposes. In determining the appropriate fair value, the Company uses the Binomial Lattice Model. In assessing the convertible debt instruments, management determines if the convertible debt host instrument is conventional convertible debt and further if there is a beneficial conversion feature requiring measurement. If the instrument is not considered conventional convertible debt, the Company will continue its evaluation process of these instruments as derivative financial instruments.
Once determined, derivative liabilities are adjusted to reflect fair value at each reporting period end, with any increase or decrease in the fair value being recorded in results of operations as an adjustment to fair value of derivatives. In addition, the fair value of freestanding derivative instruments such as warrants, are also valued using the binomial option-pricing model.
Debt Issue Costs and Debt Discount -These items are amortized over the life of the debt to interest expense. If a conversion, extinguishment or repayment of the underlying debt occurs, a proportionate share of these amounts is immediately expensed.
Beneficial Conversion Feature - For convertible debt issued in 2008/2009, the convertible feature of the convertible notes indicated a rate of conversion that was below market value. As a result, the Company recorded a "beneficial conversion feature" ("BCF") and related debt discount.
When the Company records a BCF, the relative fair value of the BCF is recorded as a debt discount from the face amount of the respective debt instrument. The discount is amortized to interest expense over the life of the debt. Upon issuance, the convertible debt instruments had an effective conversion rate per share in excess of the market price per share.
Revenue recognition - The Company follows the guidance of the Securities and Exchange Commission’s Staff Accounting Bulletin No. 104 for revenue recognition and records revenue when all of the following have occurred: (1) persuasive evidence of an arrangement exists, (2) the product is delivered, (3) the sales price to the customer is fixed or determinable, and (4) collectability of the related customer receivable is reasonably assured. There is no stated right of return for products.
Sales are recognized upon delivery of products to customers. The Company allows deductions in the form of credits for products unsold during its shelf life which is on average 3 to 4 months. The Company’s reserve for accounts receivable takes these potential future credits into consideration. Expenses such as slotting fees, sales discounts, and reclamation are accounted for as a direct reduction to revenues.
OFF-BALANCE SHEET ARRANGEMENTS:
We do not have any off-balance sheet arrangements, financings, or other relationships with unconsolidated entities or other persons, also known as “special purpose entities” (SPEs).
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK.
Not applicable.
28
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA.
NXT NUTRITIONALS HOLDINGS, INC. AND SUBSIDIARIES
CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2011 AND 2010
|
Page(s)
|
|
|
Report of Independent Registered Public Accounting Firm
|
F-1
|
|
Consolidated Balance Sheets December 31, 2011 and 2010
|
F-2
|
|
Consolidated Statements of Operations Years Ended December 31, 2011 and 2010
|
F-3
|
|
Consolidated Statement of Stockholders’ Deficit Years Ended December 31, 2011 and 2010
|
F-4
|
|
Consolidated Statements of Cash Flows Years Ended December 31, 2011 and, 2010
|
F-5
|
|
Notes to Consolidated Financial Statements
|
F-6 - F-26
|
29

REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors of:
NXT Nutritionals Holdings, Inc.
We have audited the accompanying consolidated balance sheets of NXT Nutritionals Holdings, Inc. and Subsidiaries, as of December 31, 2011 and 2010, and the related consolidated statements of operations, stockholders' deficit and cash flows for the years then ended. These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audits included considerations of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the financial position of NXT Nutritionals Holdings, Inc. and Subsidiaries as of December 31, 2011 and 2010, and the results of its operations and its cash flows for the years then ended, in conformity with accounting principles generally accepted in the United States of America.
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 3 to the financial statements, the Company had a net loss of $1,335,706 and net cash used in operations of $2,151,557 for the year ended December 31, 2011; and a working capital deficit and stockholders' deficit of $2,287,655 and $11,777,534, respectively, at December 31, 2011. These factors raise substantial doubt about the Company's ability to continue as a going concern. Management's plan in regards to these matters is also described in Note 3. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Berman & Company, P.A.
Boca Raton, Florida
April 16, 2012
551 NW 77th Street Suite 201 • Boca Raton, FL 33487
Phone: (561) 864-4444 • Fax: (561) 892-3715
www.berrnancpas.com • info@bermancpas.com
Registered with the PCAOB • Member AICPA Center for Audit Quality
Member American Institute of Certifiedd Public Accountants
Member Florida Institute of Certified Public Accountants
F-1
|
Consolidated Balance Sheets
|
||||||||
|
December 31, 2011
|
December 31, 2010
|
|||||||
|
Assets
|
||||||||
|
Current Assets
|
||||||||
|
Cash
|
$ | 444,322 | $ | 1,662,130 | ||||
|
Accounts receivable - net
|
11,021 | 119,070 | ||||||
|
Inventories - net
|
- | 431,643 | ||||||
|
Other
|
11,884 | - | ||||||
|
Total Current Assets
|
467,227 | 2,212,843 | ||||||
|
Debt issuance costs - net
|
48,076 | 25,548 | ||||||
|
Total Assets
|
$ | 515,303 | $ | 2,238,391 | ||||
|
Liabilities and Stockholders' Deficit
|
||||||||
|
Current Liabilities
|
||||||||
|
Accounts payable and accrued expenses
|
$ | 944,472 | $ | 654,962 | ||||
|
Loans payable - related parties
|
344,624 | 332,126 | ||||||
|
Loans payable - other
|
208,500 | 208,500 | ||||||
|
Accrued interest payable - related parties
|
71,545 | 59,035 | ||||||
|
Accrued interest payable - convertible notes
|
14,247 | - | ||||||
|
Registration rights payable
|
362,453 | 608,840 | ||||||
|
Convertible notes payable - net of debt discount
|
419,375 | 527,126 | ||||||
|
Derivative liabilities
|
389,666 | 2,986,900 | ||||||
|
Total Current Liabilities
|
2,754,882 | 5,377,489 | ||||||
|
Long-Term Liabilities
|
||||||||
|
Note payable - net
|
538,702 | - | ||||||
|
Convertible notes payable - net of debt discount
|
8,524,961 | 9,408,517 | ||||||
|
Total Long-Term Liabilities
|
9,063,663 | 9,408,517 | ||||||
|
Total Liabilities
|
11,818,545 | 14,786,006 | ||||||
|
Series A, convertible redeemable preferred stock, $0.001 par value, 50,000,000 shares
|
||||||||
|
authorized, 13,075,468 and 0 shares issued and outstanding, respectively (no stated value)
|
474,292 | - | ||||||
|
Stockholders' Deficit
|
||||||||
|
Common stock, $0.001 par value, 200,000,000 shares authorized,
|
||||||||
|
72,838,322 and 49,408,068 shares issued and outstanding, respectively
|
72,839 | 49,408 | ||||||
|
Additional paid in capital
|
35,783,011 | 32,822,477 | ||||||
|
Accumulated deficit
|
(47,633,384 | ) | (45,419,501 | ) | ||||
|
Total Stockholders' Deficit
|
(11,777,534 | ) | (12,547,616 | ) | ||||
|
Total Liabilities, Redeemable Preferred Stock, and Stockholders' Deficit
|
$ | 515,303 | $ | 2,238,391 | ||||
See accompanying notes to financial statements
F-2
|
NXT Nutritionals Holdings, Inc. and Subsidiaries
|
||||||||
|
Consolidated Statements of Operations
|
||||||||
|
Years Ended December 31,
|
||||||||
|
2011
|
2010
|
|||||||
|
Sales - net of slotting fees and discounts
|
$ | 237,989 | $ | 187,516 | ||||
|
Cost of sales
|
1,150,705 | 433,599 | ||||||
|
Gross loss
|
(912,716 | ) | (246,083 | ) | ||||
|
General and administrative expenses
|
3,535,854 | 3,591,276 | ||||||
|
Loss from operations
|
(4,448,570 | ) | (3,837,359 | ) | ||||
|
Other income (expense)
|
||||||||
|
Interest expense
|
(420,043 | ) | (8,616,218 | ) | ||||
|
Interest income
|
181 | 7,499 | ||||||
|
Loss on extinguishment of debt
|
- | (9,845,860 | ) | |||||
|
Forgiveness of registration rights payable
|
246,387 | - | ||||||
|
Derivative expense
|
- | (8,590,802 | ) | |||||
|
Change in fair market value of derivative liability
|
3,286,339 | 13,763,391 | ||||||
|
Registration rights expense
|
- | (283,387 | ) | |||||
|
Total other income (expense) - net
|
3,112,864 | (13,565,377 | ) | |||||
|
Net loss
|
$ | (1,335,706 | ) | $ | (17,402,736 | ) | ||
|
Series A, convertible redeemable preferred stock dividend
|
(878,177 | ) | - | |||||
|
Net loss applicable to common stockholders
|
$ | (2,213,883 | ) | $ | (17,402,736 | ) | ||
|
Net loss per common share - basic and diluted
|
$ | (0.04 | ) | $ | (0.38 | ) | ||
|
Weighted average number of common shares outstanding - basic and diluted
|
53,728,392 | 45,787,226 | ||||||
See accompanying notes to financial statements
F-3
|
Consolidated Statement of Stockholders' Deficit
|
||||||||||||||||||||
|
Years Ended December 31, 2011 and 2010
|
||||||||||||||||||||
|
Common Stock
|
||||||||||||||||||||
|
Shares
|
Amount
|
Additional Paid-In Capital
|
Accumulated Deficit
|
Stockholders' Deficit
|
||||||||||||||||
|
Balance, December 31, 2009
|
39,971,745 | $ | 39,972 | $ | 24,657,084 | $ | (28,016,765 | ) | $ | (3,319,708 | ) | |||||||||
|
Conversion of convertible notes to common stock
|
5,656,524 | 5,656 | 2,256,954 | - | 2,262,610 | |||||||||||||||
|
Exercise of warrants
|
1,509,799 | 1,510 | (1,510 | ) | - | - | ||||||||||||||
|
Stock issued for services
|
2,270,000 | 2,270 | 607,790 | - | 610,060 | |||||||||||||||
|
Stock options issued for services
|
- | - | 467,225 | - | 467,225 | |||||||||||||||
|
Reclassification to additional paid in capital when derivative ceases to exist
|
- | - | 4,834,934 | - | 4,834,934 | |||||||||||||||
|
Net loss - 2010
|
- | - | - | (17,402,736 | ) | (17,402,736 | ) | |||||||||||||
|
Balance - December 31, 2010
|
49,408,068 | 49,408 | 32,822,477 | (45,419,501 | ) | (12,547,616 | ) | |||||||||||||
|
Stock issuance costs
|
- | - | (37,012 | ) | - | (37,012 | ) | |||||||||||||
|
Conversion of convertible notes to common stock
|
3,554,806 | 3,555 | 1,348,834 | - | 1,352,389 | |||||||||||||||
|
Stock issued for services
|
19,875,448 | 19,876 | 592,849 | - | 612,725 | |||||||||||||||
|
Stock options issued for services
|
- | - | 866,790 | - | 866,790 | |||||||||||||||
|
Reclassification to additional paid in capital when derivative ceases to exist
|
- | - | 189,073 | - | 189,073 | |||||||||||||||
|
Series A, convertible redeemable preferred stock dividend
|
- | - | - | (878,177 | ) | (878,177 | ) | |||||||||||||
|
Net loss - 2011
|
- | - | - | (1,335,706 | ) | (1,335,706 | ) | |||||||||||||
|
Balance - December 31, 2011
|
72,838,322 | $ | 72,839 | $ | 35,783,011 | $ | (47,633,384 | ) | $ | (11,777,534 | ) | |||||||||
See accompanying notes to financial statements
F-4
|
Consolidated Statements of Cash Flows
|
||||||||
|
Years Ended December 31,
|
||||||||
|
2011
|
2010
|
|||||||
|
CASH FLOWS FROM OPERATING ACTIVITIES:
|
||||||||
|
Net loss
|
$ | (1,335,706 | ) | $ | (17,402,736 | ) | ||
|
Adjustments to reconcile net loss to net cash used in operating activities:
|
||||||||
|
Amortization of debt issue costs
|
19,210 | 748,030 | ||||||
|
Amortization of debt discount
|
374,076 | 7,848,508 | ||||||
|
Bad debt expense
|
4,102 | - | ||||||
|
Inventory reserve
|
897,213 | - | ||||||
|
Stock based compensation
|
1,479,516 | 1,077,284 | ||||||
|
Loss on extinguishment of debt
|
- | 9,845,860 | ||||||
|
Derivative expense
|
- | 8,590,802 | ||||||
|
Change in fair market value of derivative liabilities
|
(3,286,339 | ) | (13,763,391 | ) | ||||
|
Registration rights expense
|
- | 283,387 | ||||||
|
Forgiveness of registration rights payable
|
(246,387 | ) | - | |||||
|
Changes in operating assets and liabilities:
|
||||||||
|
(Increase) Decrease in:
|
||||||||
|
Accounts receivable
|
103,947 | (46,428 | ) | |||||
|
Prepaid expenses
|
(11,884 | ) | - | |||||
|
Inventories
|
(465,570 | ) | (281,533 | ) | ||||
|
Increase (Decrease) in:
|
||||||||
|
Accounts payable and accrued expenses
|
289,508 | 47,228 | ||||||
|
Accrued interest payable - related parties
|
12,510 | 17,910 | ||||||
|
Accrued interest payable - convertible notes
|
14,247 | - | ||||||
|
Net Cash Used in Operating Activities
|
(2,151,557 | ) | (3,035,079 | ) | ||||
|
CASH FLOWS FROM FINANCING ACTIVITIES:
|
||||||||
|
Proceeds from loans payable - related parties
|
12,498 | - | ||||||
|
Proceeds from issuance of convertible notes
|
- | 5,667,743 | ||||||
|
Debt issuance costs paid in cash
|
(41,737 | ) | (726,988 | ) | ||||
|
Proceeds from issuance of preferred stock and promissory note
|
1,000,000 | - | ||||||
|
Stock issuance costs paid in cash
|
(37,012 | ) | - | |||||
|
Repayment on loans - related parties
|
- | (45,000 | ) | |||||
|
Repayment on loans - other
|
- | (230,000 | ) | |||||
|
Liquidated damage payment on registration rights
|
- | (37,000 | ) | |||||
|
Net Cash Provided By Financing Activities
|
933,749 | 4,628,755 | ||||||
|
Net Increase (Decrease) in Cash
|
(1,217,808 | ) | 1,593,676 | |||||
|
Cash - Beginning of Year
|
1,662,130 | 68,454 | ||||||
|
Cash - End of Year
|
$ | 444,322 | $ | 1,662,130 | ||||
|
SUPPLEMENTARY CASH FLOW INFORMATION:
|
||||||||
|
Cash Paid During the Year for:
|
||||||||
|
Income taxes
|
$ | - | $ | - | ||||
|
Interest
|
$ | - | $ | - | ||||
|
SUPPLEMENTARY DISCLOSURE OF NON-CASH INVESTING AND FINANCING ACTIVITIES:
|
||||||||
|
Debt discount recorded on convertible notes
|
$ | 474,292 | $ | 5,667,743 | ||||
|
Series A, convertible redeemable preferred stock dividend
|
$ | 878,178 | $ | - | ||||
|
Original issue discount
|
$ | - | $ | 850,201 | ||||
|
Conversion of convertible note payable to common stock
|
$ | 1,352,389 | $ | 2,262,610 | ||||
|
Reclassification to additional paid in capital when derivative ceases to exist
|
$ | 189,073 | $ | 4,834,934 | ||||
|
Exercise of cashless warrants
|
$ | - | $ | 1,510 | ||||
See accompanying notes to financial statements
F-5
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
Note 1 Nature of Operations and Basis of Presentation
Nature of Operations
NXT Nutritionals Holdings, Inc. ("Holdings) is a Delaware corporation incorporated in 2006.
The Company is a developer of proprietary, patent pending, healthy alternative sweeteners. The foundation and common ingredient for all of the Company’s products is the all-natural sweetener SUSTA®. The Company also sells non-fat yogurt and yogurt smoothie products.
Note 2 Summary of Significant Accounting Policies
Principles of consolidation
All significant intercompany accounts and transactions have been eliminated in consolidation.
Use of estimates
The preparation of consolidated financial statements in conformity with U.S. generally accepted accounting principles requires management to make estimates and assumptions that affect the amounts reported in the consolidated financial statements and accompanying notes. Such estimates and assumptions impact, among others, the following: allowance for bad debt, inventory obsolescence, the fair value of share-based payments, fair value of derivative liabilities, estimates of the probability and potential magnitude of contingent liabilities and the valuation allowance for deferred tax assets due to continuing operating losses.
Making estimates requires management to exercise significant judgment. It is at least reasonably possible that the estimate of the effect of a condition, situation or set of circumstances that existed at the date of the consolidated financial statements, which management considered in formulating its estimate could change in the near term due to one or more future confirming events. Accordingly, the actual results could differ significantly from our estimates.
Risks and uncertainties
The Company operates in an industry that is subject to intense competition and change in consumer demand. The Company's operations are subject to significant risk and uncertainties including financial and operational risks including the potential risk of business failure.
The Company has experienced, and in the future expects to continue to experience, variability in sales and earnings. The factors expected to contribute to this variability include, among others, (i) general economic conditions in the various local markets in which the Company competes, including the general downturn in the economy, (ii) the volatility of prices pertaining to food and beverages in connection with the Company’s distribution of product, and (iii) the Company allocating resources effectively and efficienctly for expenses such as slotting fees and advertising due to the Company’s limited resources. These factors, among others, make it difficult to project the Company’s operating results on a consistent basis.
Cash and cash equivalents
The Company considers all highly liquid instruments purchased with a maturity of three months or less to be cash equivalents. There were no cash equivalents at December 31, 2011 and 2010, respectively.
The Company minimizes its credit risk associated with cash by periodically evaluating the credit quality of its primary financial institution.
F-6
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
Accounts receivable and allowance for doubtful accounts
Accounts receivable are stated at the amount management expects to collect from outstanding balances. The Company generally does not require collateral to support customer receivables. The Company provides an allowance for doubtful accounts based upon a review of the outstanding accounts receivable, historical collection information and existing economic conditions. The Company determines if receivables are past due based on days outstanding, and amounts are written off when determined to be uncollectible by management. The maximum accounting loss from the credit risk associated with accounts receivable is the amount of the receivable recorded, which is the face amount of the receivable net of the allowance for doubtful accounts.
As of December 31, 2011 and 2010, the allowance for doubtful accounts was $4,102 and $0, respectively.
Inventories
Inventories are stated at the lower of cost or market using the first-in, first-out (FIFO) valuation method.
|
December 31, 2011
|
December 31, 2010
|
|||||||
|
Raw materials
|
$ | 713,502 | $ | 285,769 | ||||
|
Finished goods
|
183,712 | 145,874 | ||||||
|
Reserve for obsolete inventory
|
(897,213 | ) | - | |||||
| $ | - | $ | 431,643 | |||||
Raw materials consist of ingredients used to make and package the Company’s all natural sweetener and the Company’s yogurt smoothie products. Finished goods consists of cases of both the sweetener and the yogurt smoothies.
At December 31, 2011, the Company fully reserved inventory due to the decline in sales and the delay in implementing the business plan. In addition, the Company has a gross loss from its product sales, which also is an indication of the product’s value.
Debt Issue Costs and Debt Discount
Amortized over the life of the debt to interest expense. If a conversion, extinguishment or repayment of the underlying debt occurs, a proportionate share is expensed.
Original Issue Discount
For certain convertible debt issued in 2010 and 2009, the Company provided the debt holder with an original issue discount. The original issue discount was equal to three years of simple interest at 10% of the proceeds raised. The original issue discount was recorded to debt discount reducing the face amount of the note and is being amortized to interest expense over the maturity period of the debt.
Derivative Financial Instruments
Fair value accounting requires bifurcation of embedded derivative instruments such as ratchet provisions or conversion features in convertible debt or equity instruments, and measurement of their fair value for accounting purposes. In determining the appropriate fair value, the Company uses the Binomial Lattice Valuation Model (“BLVM”). In assessing the Company’s convertible debt instruments, management determines if the convertible debt host instrument is conventional convertible debt and further if there is a beneficial conversion feature requiring measurement. If the instrument is not considered conventional convertible debt, the Company will continue its evaluation process of these instruments as derivative financial instruments.
Once determined, the derivative liabilities are adjusted to reflect fair value at each reporting period end, with any increase or decrease in the fair value being recorded in results of operations as an adjustment to fair value of derivatives. In addition, the fair value of freestanding derivative instruments such as warrants, are also valued using the BLVM.
F-7
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
Revenue recognition
The Company records revenue for yogurt, yogurt smoothies and for the natural sweetener when all of the following have occurred: (1) persuasive evidence of an arrangement exists, (2) the product is shipped or delivered, depending on the specific nature of the contract, (3) the sales price to the customer is fixed or determinable, and (4) collectability of the related customer receivable is reasonably assured. There is no stated right of return for products.
The Company allows deductions in the form of credits for products unsold during its shelf life which is on average 3 to 4 months. The Company’s reserve for accounts receivable takes these potential future credits into consideration.
Expenses such as slotting fees and sales discounts are accounted for as a direct reduction of revenues as follows:
|
Year Ended
December 31, 2011
|
Year Ended
December 31, 2010
|
|||||||
|
Gross sales
|
$ | 302,795 | $ | 477,840 | ||||
|
Less: slotting fees, discounts, allowances
|
64,806 | 290.324 | ||||||
|
Net sales
|
$ | 237,989 | $ | 187,516 | ||||
Cost of sales
Cost of sales represents costs directly related to the production and manufacturing of the Company’s products. Costs include product development, freight, packaging, and print production costs.
Advertising
Costs incurred for producing and communicating advertising for the Company are charged to operations as incurred as follows:
|
Year Ended
December 31, 2011
|
Year Ended
December 31, 2010
|
||||||
| $ |
708,041
|
$ |
617,787
|
Share-based payments
Generally, all forms of share-based payments, including stock option grants, warrants, restricted stock grants and stock appreciation rights are measured at their fair value utilizing an option pricing model on the awards’ grant date, based on the estimated number of awards that are ultimately expected to vest. Share-based compensation awards issued to non-employees for services rendered are recorded at either the fair value of the services rendered or the fair value of the share-based payment, whichever is more readily determinable. The expense resulting from share-based payments are recorded in cost of goods sold or general and administrative expense in the consolidated statement of operations, depending on the nature of the services provided.
Income Taxes
The Company accounts for income taxes in accordance with accounting guidance now codified as FASB ASC Topic 740, “Income Taxes,” which requires that the Company recognize deferred tax liabilities and assets based on the differences between the financial statement carrying amounts and the tax bases of assets and liabilities, using enacted tax rates in effect in the years the differences are expected to reverse. Deferred income tax benefit (expense) results from the change in net deferred tax assets or deferred tax liabilities. A valuation allowance is recorded when it is more likely than not that some or all deferred tax assets will not be realized.
F-8
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
Accounting guidance now codified as FASB ASC Topic 740-20, “Income Taxes – Intraperiod Tax Allocation,” clarifies the accounting for uncertainties in income taxes recognized in accordance with FASB ASC Topic 740-20 by prescribing guidance for the recognition, de-recognition and measurement in financial statements of income tax positions taken in previously filed tax returns or tax positions expected to be taken in tax returns, including a decision whether to file or not to file in a particular jurisdiction. FASB ASC Topic 740-20 requires that any liability created for unrecognized tax benefits is disclosed. The application of FASB ASC Topic 740-20 may also affect the tax bases of assets and liabilities and therefore may change or create deferred tax liabilities or assets. The Company would recognize interest and penalties related to unrecognized tax benefits in income tax expense. At December 31, 2011 and 2010, respectively, the Company did not record any liabilities for uncertain tax positions.
Earnings per share
Basic earnings (loss) per share is computed by dividing net income (loss) by weighted average number of shares of common stock outstanding during each period. Diluted earnings (loss) per share is computed by dividing net income (loss), adjusted for changes in income or loss that resulted from the assumed conversion of convertible shares, by the weighted average number of shares of common stock, common stock equivalents and potentially dilutive securities outstanding during the period.
The Company had the following potential common stock equivalents at December 31, 2011:
|
Series A, convertible redeemable preferred stock (see note 8)
|
36,396,684 | |||
|
Convertible debt – face amount of $8,650,965, conversion price of $0.25
|
34,603,860 | |||
|
Convertible debt – face amount of $436,458, conversion price of $0.09
|
4,849,534 | |||
|
Common stock warrants, exercise price of $0.25 (Series “A”, “B” and “C”)
|
27,231,269 | |||
|
Stock options, exercise price $0.22
|
6,063,750 | |||
|
Total common stock equivalents
|
109,145,097 | |||
The Company had the following potential common stock equivalents at December 31, 2010:
|
Convertible debt – face amount of $2,001,128, conversion price of $0.40
|
5,002,820 | |||
|
Convertible debt – face amount of $8,438,684, conversion price of $0.40
|
21,096,710 | |||
|
Common stock warrants, conversion price of $0.40 (Series “A”) and $0.60 (Series “B”)
|
19,735,634 | |||
|
Common stock warrants, conversion price of $0.40 (Series “C”)
|
7,495,635 | |||
|
Stock options, exercise price $0.22
|
2,123,750 | |||
|
Total common stock equivalents
|
55,454,549 |
Since the Company reflected a net loss in 2011 and 2010, the effect of considering any common stock equivalents, would have been anti-dilutive. A separate computation of diluted earnings (loss) per share is not presented.
Reclassification
Certain items in the 2010 financial statement presentation have been reclassified to conform to the 2011 presentation. Such reclassifications have no effect on previously reported financial condition, operations or cash flows.
Recent accounting pronouncements
In May 2011, the FASB issued ASU No. 2011-04, Fair Value Measurement (Topic 820): Amendments to Achieve Common Fair Value Measurement and Disclosure Requirements in U.S. GAAP and IFRSs. The guidance in ASU 2011-04 changes the wording used to describe the requirements in U.S. GAAP for measuring fair value and for disclosing information about fair value measurements, including clarification of the FASB's intent about the application of existing fair value and disclosure requirements and changing a particular principle or requirement for measuring fair value or for disclosing information about fair value measurements. The amendments in this ASU should be applied prospectively and are effective for interim and annual periods beginning after December 15, 2011. Early adoption by public entities is not permitted. The adoption of this guidance is not expected to have a material impact on the Company’s financial position or results of operations.
F-9
Note 3 Going Concern
As reflected in the accompanying consolidated financial statements, the Company has a net loss of $1,335,706 and net cash used in operations of $2,151,557 for the year ended December 31, 2011; and has a working capital deficit of $2,287,655 and a stockholders’ deficit of $11,777,534.
The ability of the Company to continue its operations is dependent on Management's plans, which include the raising of capital through debt and/or equity markets with some additional funding from other traditional financing sources, including term notes, until such time that funds provided by operations are sufficient to fund working capital requirements. The Company may need to incur additional liabilities with certain related parties to sustain the Company’s existence.
The Company will require additional funding to finance the growth of its current and expected future operations as well as to achieve its strategic objectives. The Company believes its current available cash along with anticipated revenues may be insufficient to meet its cash needs for the near future. There can be no assurance that financing will be available in amounts or terms acceptable to the Company, if at all (See Note 5 financing).
The accompanying consolidated financial statements have been prepared on a going concern basis, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business. These financial statements do not include any adjustments relating to the recovery of the recorded assets or the classification of the liabilities that might be necessary should the Company be unable to continue as a going concern.
Note 4 Loans Payable
|
(A)
|
Loans Payable – Related Parties
|
As part of the 2008 Unit Purchase Agreement, the Company agreed to pay $600,000 to the unitholders of Healthy Dairy, LLC and NXT, LLC within 90 days from the close of the reverse acquisition, which occurred in February 2009, with a public shell, and an additional $600,000 within 180 days post closing. There are no penalties or interest. Payments will be made once the Company begins to become profitable. As of December 31, 2009, the Company had a balance due of $352,126. The Company paid $20,000 during 2010.
In December 2009, a Director loaned the Company $25,000. The loan is unsecured, bore no interest and was due on demand. The Company repaid the $25,000 during February 2010.
During 2011, 3 Directors loaned the Company $12,498. The loans are unsecured, bear no interest and are due on demand.
The following is a summary of loans payable – related parties:
|
Balance – December 31, 2009
|
377,126
|
|||
|
Repayments
|
(45,000
|
)
|
||
|
Balance – December 31, 2010
|
$
|
332,126
|
||
|
Loans from Directors
|
12,498
|
|||
|
Balance - December 31, 2011
|
$ |
344,624
|
F-10
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
|
(B)
|
Loans Payable – Other
|
On December 30, 2009, the Company issued a $100,000 unsecured promissory note to a third party. The note bore 10% interest and was repaid in February 2010.
In 2008, the Company had outstanding loans for $388,500. These loans bear interest at 6%. The loans are secured by all assets of the Company. These loans are due on demand. The Company repaid $90,000 of these advances in May 2009, and an additional $130,000 was repaid in 2010.
At December 31, 2011 and 2010, the Company has accrued interest on this loan of $71,545 and $59,035, respectively.
|
Balance – December 31, 2009
|
$
|
438,500
|
||
|
Repayments
|
(230,000
|
)
|
||
|
Balance – December 31, 2010
|
208,500
|
|||
|
Repayments
|
(-)
|
|||
|
Balance – December 31, 2011
|
$
|
208,500
|
Note 5 Note Payable
On November 21, 2011, the Company issued a 13% senior secured note with a face amount of $1,000,000, due on the earlier of November 21, 2015 or closing of a change in control transaction. (See Note 8).
In connection with the issuance of the note and Series A, convertible redeemable preferred stock, the Company determined that an allocation of fair value was needed to derive amounts that would be recorded as debt, temporary equity and debt issue costs.
Utilizing the relative fair value method, the Company allocated 53% of the $1,000,000 proceeds to the note. In addition, the Company allocated 53% of the $78,750 ($41,737) in transaction issuance costs as debt issuance costs on the balance sheet and is amortizing these costs over the life of the note. The note is secured by all assets of the Company. The Company allocated 47% of the $1,000,000 proceeds to the series A, convertible redeemable preferred stock. In addition, the Company allocated 47% of the $78,750 ($37,012) in issuance costs as stock issuance costs on the balance sheet.
The Company recorded a debt discount, in the amount of $474,292, which represents the allocation of the gross proceeds of $1,000,000 (47%) that was allocated to the Series A, convertible redeemable preferred stock. During the year ended December 31, 2011, the company amortized $12,994 of the debt discount to interest expense.
At December 31, 2011, the Company has accrued interest on this note in the amounts of $14,247.
The notes carry an additional payment provision in which the Company upon the earlier of (i) the date of prepayment of the principal amount of note, or (ii) the payment of the principal amount of the note on the maturity date, whichever occurs first, shall pay to the holder an additional $200,000; provided that (i) in the event of one or more payments of a portion but not the entire principal amount of this note either as a prepayment or on the maturity date, such payment shall be prorated based upon the portion of the principal amount of this Note then being paid to the Holder divided by the aggregate Principal Amount of this Note on the date hereof; and (ii) no additional payment shall be due and owing to the Holder if the average closing price per share of the Common Stock equals $0.20 or higher per share for the 90 consecutive Trading Days on the OTCBB (or such other then applicable principal Trading Market of the Company’s shares of Common Stock) immediately preceding any payment with respect to the principal amount of the note.
F-11
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
Note 6 Convertible Notes Payable
At December 31, 2011 and December 31, 2010, convertible debt consists of the following:
|
December 31,
2011
|
December 31,
2010
|
|||||||
|
Convertible debt – secured – derivative liabilities (A)
|
$
|
7,575,095
|
$ |
8,438,684
|
||||
|
Conventional convertible debt – secured (B)
|
1,512,328
|
2,001,128
|
||||||
|
Less: debt discount
|
(143,087
|
)
|
(504,169)
|
|||||
|
Convertible debt – net
|
8,944,336
|
9,935,643
|
||||||
|
Less: current portion
|
(419,375
|
)
|
(527,126)
|
|||||
|
Long term debt
|
$
|
8,524,961
|
$ |
9,408,517
|
||||
|
(A)
|
2010 Original Issue Discount Senior Secured Convertible Note Offering
|
On February 26, 2010, the Company completed a secured convertible notes and warrants offerings, which was modified and amended on September 1, 2010, December 6, 2010 and November 4, 2011.
The original principal of the notes was $6,517,944 with an original issue discount of $850,201. The gross proceeds raised were $5,667,743, of which the Company received approximately $4,940,755 in net proceeds. The Company paid $726,988 in debt issuance costs. The new face amount of the secured convertible notes after the second amendment is $8,438,684.
|
i.
|
In accordance with the December 6, 2010 Modification and Amendment Agreement, the Conversion price was modified from $0.40 to $0.37.
|
|
ii.
|
Registration rights – The notes contained a registration rights provision
|
|
iii.
|
Original issue discount- 60% of the cash proceeds received. The discount was fully amortized to interest expense as of December 31, 2010.
|
|
iv.
|
Full ratchet provision – The notes contain a provision in which the conversion price can be reduced in any event the Company issues any security or debt instrument with a lower consideration per share in any future offering.
|
|
v.
|
Secured by all assets of the Company
|
F-12
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
The Company also issued the note holders (“2010 holders”) one stock purchase warrant with a maturity of 5 years. The stock purchase warrants contain cashless exercise provisions. There are currently 7,495,635 warrants associated with this offering outstanding.
The Company recorded the fair value of the derivative liabilities to debt discount to the extent of the face amount of the notes and expensed immediately the remaining value of the derivative as it exceeded the face amount of the note. The Company recorded a derivative expense at issuance in the amount of $8,590,802.
The fair value of the derivative liabilities at issuance was based upon the following management assumptions:
|
Exercise price
|
$1 - $1.25
|
|
Expected dividends
|
0%
|
|
Expected volatility
|
200%
|
|
Expected term: conversion feature
|
1.25 years
|
|
Expected term: warrants
|
5 years
|
|
Risk free interest rate
|
0.35% - 2.3%
|
Modification and Amendment of 2010 Original Issue Discount Senior Secured Convertible Note (Extinguishment Accounting)
The Company determined that the First and Second Modification and Amendment of the debt instruments on September 1, 2010 and December 6, 2010, resulted in debt instruments being exchanged with substantially different terms and applied extinguishment accounting resulting in a loss on extinguishment of debt as follows:
|
Description
|
Date of Modification
|
|||||
|
First Modification
|
September 1, 2010
|
$ | 3,726,410 | |||
|
Second Modification
|
December 6, 2010
|
6,119,450 | ||||
|
Loss on extinguishment of debt
|
$ | 9,845,860 | ||||
The Company compared the present value of both old and new debt as well as the fair value of the warrants granted in the new offering. The Company determined that the present value of the cash flows associated with the new debt instruments exceeded the present value of the old debt instruments by more than 10% and accounted for the modification as an extinguishment.
On September 6, 2011, the Company entered into a third modification and amendment agreement. This agreement acted as a stand-still agreement. All rights and privleges under the second modification and amendment agreement were extended for 30 days.
On November 4, 2011, the Company entered into a fourth modification agreement with the 2010 Secured Convertible Note and Warrant holders. Accordingly, the Company reclassified the debt to long-term liabilities.
Under the modification, the 2010 Holders:
|
·
|
Waived their right to monthly redemptions on the notes,
|
|
·
|
Extended the maturity date to November 21, 2015, ratcheted the exercise price to $0.25 per share for both the notes and warrants,
|
F-13
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
|
·
|
Waived anti-dilution provisions for the note and warrants (The derivative liability ceased to exist),
|
|
·
|
Waived registration right penalties of $246,387, the company recorded as a gain on forgiveness of debt,
|
|
·
|
Agreed to subordinate their security interests securing the Notes to the senior security interests to be granted by the Company to secure the investors in the November 21, 2011 financing and to third parties that provide financing for accounts receivables, inventory and raw materials of the Company.
|
The Company analyzed the fourth modification and amendment agreement. The Company then assessed if the modification and amendments qualified for modification or extinguishment accounting. Because the fair value of the instrument prior to modification and amendment was less than ten percent of the instrument post modification, the Company concluded that extinguishment accounting was not applicable, and the Company applied modification accounting.
In connection with the ASC 815, “Determining Whether an Instrument (or Embedded Feature) Is Indexed to an Entity’s Own Stock,” the Company determined that the embedded conversion feature and the warrant issuances (ratchet down of exercise price based upon lower exercise price in future offerings) are not indexed to the Company’s own stock and, therefore, is an embedded derivative financial liability (the “Embedded Derivative”), which requires bifurcation and to be separately accounted for.
The Company measured the fair value of the derivative liabilities using a BLVM.
The fair value of the derivative liabilities are summarized as follows:
|
Fair value at the issuance dates for conversion feature and warrants issued on the 2010 Original Issue Discount Senior Secured Convertible Note Offering
|
$ | 14,258,545 | ||
|
Mark to market adjustments for the year ended December 31, 2010:
|
(13,763,391 | ) | ||
|
Reclassification to paid in capital when derivative liability ceases to exist
|
(4,834,934 | ) | ||
|
Fair value of the derivative as modified
|
7,326,680 |
|
Derivative liability balance at December 31, 2010
|
$ | 2,986,900 | ||
|
Reclassification to paid in capital when derivative liability ceases to exist
|
( 189,073 | ) | ||
|
Fair value mark to market adjustment
|
(2,797,827 | ) | ||
|
Derivative liability balance at December 31, 2011
|
$ | - |
Mark to Market
At the time of each debt conversion, modification and at year end, the Company re-measured the derivative liabilities at fair value. As a term of the fourth modification, the investors waived the ratchet provision, and the derivative ceased to exist (see Note 8 for the Company's only outstanding derivative liability). The following management assumptions were considered in connection with the computation of fair value of debt and warrants and related reclassification of derivative liabilities to additional paid in capital as well as the period end fair value re-measurement:
F-14
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
|
Exercise price
|
$0.25 - $0.40
|
|
Expected dividends
|
0%
|
|
Expected volatility
|
137% - 360%
|
|
Risk fee interest rate
|
0.20% – 2.24%
|
|
Suboptimal exercise factor
|
1.5
|
|
Expected life of conversion features
|
0.50 years
|
|
Expected life of warrants
|
3.4 years
|
Conversions
During the year ended December 31, 2011, note holders converted principal of $863,589 into 2,332,806 shares of common stock, at a conversion rate of $0.37 - $0.40 per share.
During the year ended December 31, 2010, note holders converted principal in the amount of $598,400 into 1,496,000 shares of the Company’s common stock at a conversion rate of $0.40.
The 2010 Original Issue Discount Senior Secured Convertible Notes are summarized as follow:
|
Fair value at the issuance dates for conversion feature and warrants issued on the 2010 Original Principal amount at issuance
|
$
|
6,517,944
|
||
|
Less debt discount at issuance
|
(6,517,944
|
)
|
||
|
Amortization of debt discount
|
6,517,944
|
|||
|
Increase in principal, as modified in the December 6, 2010 Modification and Amended Agreement
|
2,519,140
|
|||
|
Conversion of note payable to common stock
|
(598,400)
|
|
Convertible Notes Payable at December 31, 2010
|
$ | 8,438,684 | ||
|
Conversion of notes into common stock
|
(863,589 | ) | ||
|
Convertible Notes Payable December 31, 2011
|
$ | 7,575,095 |
|
(B)
|
Convertible Debt and Warrants
|
During 2008 and 2009, the Company entered into a convertible note and warrant offering. The terms are as follows:
|
i.
|
Exercise price - ranging from $0.09 - $0.25
|
|
ii.
|
Three year maturity
|
|
iii.
|
Original issue discount of 30%
|
|
iv.
|
Secured by all assets of the Company
|
|
v.
|
Registration rights – the Company was required to have a registration statement filed within 60 days after the offering closed. To date, the company has not filed a registration statement and has accrued the maximum liquidated damages penalty of 9% of the offering totaling $362,453.
|
|
vi.
|
Full ratchet provision – The notes contain a provision in which the conversion price can be reduced in any event the Company issues any security or debt instrument with a lower consideration per share in any future offering.
|
F-15
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
|
vii.
|
1 Series “A”, 5 Year, $0.40 stock purchase warrant, and 1 Series “B”, 5 Year, $0.60 stock purchase warrant, for each dollar invested into convertible debt
|
On November 4, 2011, approximately 70% of the 2009 Convertible Debenture Holders (“2009 Holders”) entered into a modification agreement.
The 2009 Holders:
|
·
|
Ratcheted the exercise price to $0.25 per share for both the note and warrants,
|
|
·
|
Waived anti-dilution provisions for both the note and warrants (ceases to be a derivative liability); and
|
|
·
|
Extended the maturity date to November 21, 2015
|
A summary of the Convertible Debt Principal is as follows:
|
Convertible Debt, net of debt discount as of December 31, 2009
|
$ | 1,830,605 | ||
|
Conversion of debt into common stock
|
(1,664,210 | ) | ||
|
Amortization of debt discount for the year ended December 31, 2009
|
1,330,564 |
|
Convertible Debt, net of debt discount as of December 31, 2010
|
$ | 1,496,959 | ||
|
Conversion of debt into common stock
|
(488,800 | ) | ||
|
Amortization of debt discount for the year ended December 31, 2011
|
361,081 | |||
|
Convertible Debt, net of debt discount as of December 31, 2011
|
$ | 1,369,240 |
Conversions
During the year ended December 31, 2011, note holders converted principal of $488,800 into 1,222,000 shares of common stock, at a conversion rate of $0.40 per share
During the year ended December 31, 2010, note holders converted principal in the amount of $1,664,210 into 4,160,524 shares of the Company’s common stock at a conversion rate of $0.40.
|
(C)
|
Debt Issuance Costs
|
Debt issuance costs, net are as follows:
|
Balance - December 31, 2009
|
$ | 46,590 | ||
|
Debt issue costs paid in 2010
|
726,988 | |||
|
Amortization of debt issue costs
|
(748,030 | ) | ||
|
Balance - December 31, 2010
|
25,548 | |||
|
Debt issue costs paid in 2011
|
41,737 | |||
|
Amortization of debt issue costs
|
(19,209 | ) | ||
|
Balance – December 31, 2011
|
$ | 48,076 |
F-16
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
|
(D)
|
Registration Rights Penalty
|
The Company incurred liquidated damages for failing to file timely registration statements in 2009 and 2010. The Company recorded liquidated damages as outlined below. In accordance with the November 4, 2011 modification agreement, the 2010 noteholders waived their respective liquidated damages in the amount of $246,387. The 2008/2009 noteholders did not forgive their liquidated damages.
|
Balance as of December 31, 2009
|
$ | 362,453 | ||||||
|
Gross 2010 proceeds raised
|
$ | 5,667,743 | ||||||
|
Penalty for late filing of registration statement
|
0.50 | % | 28,339 | |||||
|
Penalty for not being declared effective
|
0.045 | % | 255,048 | |||||
|
Less: payments made
|
(37,000 | ) | ||||||
|
Balance as of December 31, 2010
|
$ | 608,840 | ||||||
|
Forgiveness of registration rights payable
|
(246,387 | ) | ||||||
|
Balance as of December 31, 2011
|
$ | 362,453 | ||||||
Note 7 Fair Value
Disclosures of information about the fair value of certain financial instruments for which it is practicable to estimate the value. For purpose of this disclosure, the fair value of a financial instrument is the amount at which the instrument could be exchanged in a current transaction between willing parties, other than in a forced sale or liquidation.
The Company has categorized its assets and liabilities recorded at fair value based upon the fair value hierarchy specified by GAAP.
The levels of fair value hierarchy are as follows:
|
|
·
|
Level 1 inputs utilize unadjusted quoted prices in active markets for identical assets or liabilities that the Company has the ability to access;
|
|
|
·
|
Level 2 inputs utilize other-than-quoted prices that are observable, either directly or indirectly. Level 2 inputs include quoted prices for similar assets and liabilities in active markets, and inputs such as interest rates and yield curves that are observable at commonly quoted intervals; and
|
|
|
·
|
Level 3 inputs are unobservable and are typically based on our own assumptions, including situations where there is little, if any, market activity.
|
F-17
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
In certain cases, the inputs used to measure fair value may fall into different levels of the fair value hierarchy. In such cases, the Company categorizes such financial asset or liability based on the lowest level input that is significant to the fair value measurement in its entirety. Our assessment of the significance of a particular input to the fair value measurement in its entirety requires judgment and considers factors specific to the asset or liability.
Both observable and unobservable inputs may be used to determine the fair value of positions that are classified within the Level 3 category. As a result, the unrealized gains and losses for assets within the Level 3 category presented in the tables below may include changes in fair value that were attributable to both observable and unobservable inputs.
The Company recorded derivative liabilities of $389,666 and $2,986,900 December 31, 2011 and December 31, 2010. These derivative liabilities are level 2.
Derivative activity is summarized as follows:
|
Fair value at the issuance dates for conversion feature and warrants issued on the 2010 Original Issue Discount Senior Secured Convertible Note Offering (Note 6A)
|
$ | 14,258,545 | ||
|
Mark to market adjustments for the year ended December 31, 2010 (Note 6A)
|
(13,763,391 | ) | ||
|
Reclassification to paid in capital when derivative liability ceases to exist (Note 6A)
|
(4,834,934 | ) | ||
|
Fair value of the Modified and Amended Agreements (Note 6A)
|
7,326,680 | |||
|
Derivative liability balance at December 31, 2010 (Note 6A)
|
2,986,900 | |||
|
Reclassification to paid in capital when derivative liability ceases to exist (Note 6A)
|
(189,072 | ) | ||
|
Fair value mark to market adjustment (debt) (Note 6A)
|
(2,797,828 | ) | ||
|
Recognition of derivative at commitment date (Note 8)
|
878,177 | |||
|
Fair value mark to market adjustment (Series A, convertible redeemable preferred stock (Note 8)
|
(488,511 | ) | ||
|
Derivative liability balance at December 31, 2011
|
$ | 389,666 |
Note 8 Series A, Convertible Redeemable Preferred Stock
On November 21, 2011, the Company sold 13,075,468 shares of Series A, Convertible Redeemable Preferred Stock with full voting rights equivalent to a fixed 20% voting right. The preferred stock carries the following features:
|
1.
|
Preferred stock is convertible at the option of the holder into an equal number of shares of common stock of the Company representing 20% of the Company’s fully diluted capital stock at the time of conversion. The redemption is triggered upon a change in control or November 21, 2015. The Company concluded the variable conversion feature in the form of a guaranteed 20% ownership right would be classified and accounted for as a derivative liability, The preferred stock is convertible into 20% of the fully diluted capital structure of the Company. It is unknown if the Company will have enough authorized shares to settle this conversion in common stock. Due to this uncertainty, the conversion feature is accounted as a derivative liability. The Company measured the fair value of the derivative liabilities using a BLVM.
|
|
2.
|
Annual dividend equal to the greater of (a) 10% of the then outstanding principal ($1,000,000 at December 31, 2011) amount of the 2011 note payable or (b) 10% of net income in excess of $500,000; Dividends accrue commencing on January 1, 2012 and are payable annually in arrears.
|
F-18
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
|
3.
|
A first priority lien in the assets of the Company was granted to the holders of the 2011 Note. The holders of the 2010 Notes subordinated their interests to the security in the assets of the Company. See discussion of Fourth Modification.
|
|
4.
|
The investors may invest an additional $500,000.
|
|
|
|
|
5.
|
Registration Rights- the Company agreed to register 100% of the common shares issuable upon conversion of the preferred stock, which equates to 20% of the fully dilutive capital structure of the Company, which represents 36,396,684 shares on December 31, 2011. The registration statement must be filed within 60 days following the filing of the Company’s 2011 10K and must be declared effective within 180 calendar days from November 21, 2011, which is on or about May 31, 2012.
|
|
6.
|
If the Company fails the registration rights provisions, liquidated damages will be assessed up to a maximum of 6% of the amount financed.
|
Under Regulation S-X, Rule 5-02-28, preferred stock must be classified outside shareholders' equity when the stock is:
●Redeemable at a fixed or determinable price on a fixed or determinable date,
●Redeemable at the option of the holder, or
●Redeemable based on conditions outside the control of the issuer.
Since the preferred stock is redeemable on November 21, 2015 it is presented on the balance sheets as “Series A, Convertible Redeemable Preferred Stock” in a manner consistent with temporary equity.
There are no other features associated with this class of redeemable preferred stock, which require disclosure. The carrying amount and redemption amount are $474,292 (see note 5, which value of approximately 47% of the $1,000,000 offering). The carrying amount of the redeemable preferred stock will increase at the time when dividends are declared.
The preferred stock is convertible into 20% of the fully diluted capital structure of the Company. It is unknown if the Company will have enough authorized shares to settle this conversion in common stock. Due to this uncertainty, the conversion feature is accounted as a derivative liability. The Company measured the fair value of the derivative liabilities using a BLVM.
At issuance and at December 31, 2011, the Company measured the derivative liabilities at fair value. The following management assumptions were considered in connection with the computation of fair value:
|
Exercise price
|
$0.015 - $0.04
|
|
Expected dividends
|
0%
|
|
Expected volatility
|
137%
|
|
Risk fee interest rate
|
0.41%
|
|
Suboptimal exercise factor
|
1
|
|
Expected life of conversion features
|
4 years
|
F-19
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
The fair value of the derivative liabilities embedded in the Series A, Convertible Redeemable Preferred Stock are summarized as follow:
|
Derivative liability balance at December 31, 2010
|
$ | - | ||
|
Recognition of derivative at commitment date (1)
|
878,177 | |||
|
Fair value mark to market adjustment
|
(488,511 | ) | ||
|
Derivative liability balance at December 31, 2011
|
389,666 |
(1) The fair value of this transaction was also recorded as a deemed preferred stock dividend and changed to accumulated deficit.
Note 9 Stockholders Deficit
|
(A)
|
Common Stock
|
During the year ended December 31, 2011, the Company issued the following common stock:
|
Transaction Type
|
Quantity of Shares
|
Valuation
|
Range of Value per Share
|
|||||||||
|
Services rendered – officers
|
1,660,000 | $ | 143,000 | $ | 0.027 - $0.24 | |||||||
|
Services rendered – consultants
|
18,215,448 | $ | 469,725 | $ | 0.016 - $0.24 | |||||||
|
Conversion of debt
|
3,554,806 | $ | 1,352,389 | $ | 0.25 - $0.40 | |||||||
|
Total
|
23,430,254 | $ | 1,965,114 | $ | 0.016 - $0.40 | |||||||
The fair value of all stock issuances above is based upon the quoted closing trading price on the date of issuance,
F-20
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
During the year ended December 31, 2010, the Company issued the following common stock:
|
Transaction Type
|
Quantity of Shares
|
Valuation
|
Range of Value per Share
|
|||||||||
|
Services rendered – officers
|
605,000 | $ | 127,290 | $ | 0.01 - $0.61 | |||||||
|
Services rendered – consultants
|
1,365,000 | 299,770 | $ | 0.01 - $1.09 | ||||||||
|
Conversion of debt
|
5,656,524 | 2,262,610 | $ | 0.40 | ||||||||
|
Settlement Agreement
|
300,000 | 183,000 | $ | 0.61 | ||||||||
|
Cashless exercise of warrants (1)
|
1,509,799 | - | $ | - | ||||||||
|
Total
|
9,436,323 | $ | 2,872,670 | $ | 0.01 - $1.09 | |||||||
The fair value of all stock issuances above is based upon the quoted closing trading price on the date of issuance,
(1) During the year ended December 31, 2010, note holders exercised 2,171,147 stock purchase warrants utilizing the cashless exercise provision (at $0.61/share), for 1,509,799 shares of the Company’s common stock.
(B) Stock Options
On November 12, 2010, the Company adopted the 2010 Incentive Stock Plan (“the Plan”). The total number of shares of stock which may be purchased or granted directly by options, stock awards or restricted stock purchase offers, or purchased indirectly through exercise of options granted under the Plan shall not exceed 30,000,000 plus an increase of an the first day of each fiscal year, beginning in 2010. The Plan indicates that the exercise price of an award is equivalent to the market value of the Company’s common stock on the grant date.
On November 12, 2010, the Company's board of directors authorized the issuance of 8,495,000 and 7,265,000 stock options for fiscal year 2010 and fiscal year 2011, respectively, having a total fair value of $3,467,194, which vest over a 4 year term. These options expire between November 12, 2020 and November 12, 2021. The 2011 options were granted on November 12, 2010 but the requisite service period does not begin until November 2011, at which time the vesting begins. The Company began expensing the 2011 options as of November 12, 2011. During the years ended December 31, 2011 and 2010, the Company expensed $866,790 and $467,225, respectively.
F-21
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
The following is a summary of the Company’s stock option activity:
|
Options
|
Weighted
Average
Exercise
Price
|
Weighted
Average
Remaining Contractual Life
|
Aggregate
Intrinsic
Value
|
||||||||||
|
Balance – December 31, 2010
|
- | - | |||||||||||
|
Granted
|
15,760,000 | $ | 0.22 | ||||||||||
|
Exercised
|
- | ||||||||||||
|
Forfeited
|
- | ||||||||||||
|
Balance – December 31, 2010 – outstanding
|
15,760,000 | $ | 0.22 | $ | - | ||||||||
|
Balance – December 31, 2010 – exercisable
|
2,123,750 | $ | 0.22 | $ | - | ||||||||
|
Granted
|
- | $ | - | ||||||||||
|
Exercised
|
- | $ | - | ||||||||||
|
Forfeited
|
- | $ | - | ||||||||||
|
Balance – December 31, 2011 – outstanding
|
15,760,000 | $ | 0.22 |
8.90 years
|
$ | - | |||||||
|
Balance - December 31, 2011 – exercisable
|
6,063,750 | $ | 0.22 |
8.90 years
|
$ | - | |||||||
|
Outstanding options held by related parties – 2011
|
15,760,000 | ||||||||||||
|
Exercisable options held by related parties – 2011
|
6,063,750 | ||||||||||||
On the date of grant in 2010 (for the 2010 and 2011 options), the Company valued these issuances at fair value, utilizing a Black Scholes Option Pricing Model. The Company utilized the following management assumptions:
|
Exercise price
|
$0.22
|
|
Expected dividends
|
0%
|
|
Expected volatility
|
360%
|
|
Risk fee interest rate
|
2.8%
|
|
Expected life of stock options
|
7 years
|
|
Expected forfeitures
|
0%
|
F-22
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
(C)Warrants
The following is a summary of the Company’s warrant activity:
|
Warrants
|
Weighted
Average
Exercise Price
|
|||||||
|
Outstanding – December 31, 2009
|
21,906,781 | $ | 0.40 | |||||
|
Exercisable – December 31, 2009
|
21,906,781 | $ | 0.40 | |||||
|
Granted
|
7,495,635 | $ | 0.40 | |||||
|
Exercised
|
(2,171,147 | ) | $ | 0.40 | ||||
|
Forfeited/Cancelled
|
- | $ | - | |||||
|
Outstanding – December 31, 2010
|
27,231,269 | $ | 0.40 | |||||
|
Exercisable – December 31, 2010
|
27,231,269 | $ | 0.40 | |||||
|
Granted
|
- | $ | - | |||||
|
Exercised
|
- | $ | - | |||||
|
Forfeited/Cancelled
|
- | $ | - | |||||
|
Outstanding – December 31, 2011
|
27,231,269 | $ | 0.25 | |||||
|
Exercisable – December 31, 2011
|
27,231,269 | $ | 0.25 | |||||
|
Warrants Outstanding
|
Warrants Exercisable
|
||||||||||||||||||
|
Range of
exercise price |
Number
Outstanding
|
Weighted Average
Remaining
Contractual Life
(in years)
|
Weighted Average Exercise Price
|
Number Exercisable
|
Weighted Average Exercise Price
|
||||||||||||||
| $ | 0.25 | 27,231,269 |
2.62 years
|
$ | 0.25 | 27,231,269 | $ | 0.25 | |||||||||||
As part of the modifications of debt on November 4, 2011, all exercise prices of the warrants were ratcheted to $0.25. The above table retrospectively reflects the ratchet. At December 31, 2011 and 2010, the total intrinsic value of warrants outstanding and exercisable was $0 and $0, respectively.
Note 10 Commitments and Contingencies
Litigations, Claims and Assessments
From time to time, the Company may become involved in various lawsuits and legal proceedings, which arise in the ordinary course of business. However, litigation is subject to inherent uncertainties, and an adverse result in these or other matters may arise that may harm its business. The Company is currently not aware of any such legal proceedings or claims that they believe will have, individually or in the aggregate, a material adverse affect on its business, financial condition or operating results.
F-23
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
Note 11 Income Taxes
At December 31, 2011, the Company has a net operating loss carry-forward of approximately $7,499,000 available to offset future taxable income expiring through 2031. Utilization of these net operating losses may be limited due to potential ownership changes under Section 382 of the Internal Revenue Code.
The valuation allowance at December 31, 2011 was $3,390,000. The net change in valuation allowance during the year ended December 31, 2011 was an increase of $1,082,000. In assessing the realizability of deferred tax assets, management considers whether it is more likely than not that some portion or all of the deferred income tax assets will not be realized. The ultimate realization of deferred income tax assets is dependent upon the generation of future taxable income during the periods in which those temporary differences become deductible. Management considers the scheduled reversal of deferred income tax liabilities, projected future taxable income, and tax planning strategies in making this assessment. Based on consideration of these items, Management has determined that enough uncertainty exists relative to the realization of the deferred income tax asset balances to warrant the application of a full valuation allowance as of December 31, 2011.
Significant deferred tax assets at December 31, 2011 and 2010 are as follows:
|
2011
|
2010
|
|||||||
|
Gross deferred tax assets:
|
||||||||
|
Net operating loss carryforward
|
$
|
2,998,000
|
$
|
1,945,000
|
||||
|
Accrued salary
|
229,000
|
103,000
|
||||||
|
Registration rights
|
|
163,000
|
260,000
|
|||||
|
Total deferred tax assets
|
3,390,000
|
2,308,000
|
||||||
|
Less: valuation allowance
|
(3,390,000
|
)
|
(2,308,000
|
)
|
||||
|
Deferred tax asset – net
|
$
|
-
|
$
|
-
|
||||
There was no income tax expense for the years ended December 31, 2011 and 2010 due to the Company’s net losses.
The Company’s tax expense differs from the “expected” tax expense for the years ended December 31, 2011 and 2010, (computed by applying the Federal Corporate tax rate of 34% to loss before taxes and 8.25% and 9.5% for State Corporate taxes for the years ended December 31, 2011 and 2010, respectively, the blended rate used was 39% and 41% for the years ended December 31, 2011 and 2010, respectively), as follows:
|
2010
|
2010
|
|||||||
|
Expected tax expense (benefit) – Federal
|
$
|
(417,000
|
)
|
$
|
(5,355,000
|
)
|
||
|
Expected tax expense (benefit) – State
|
(110,000
|
)
|
( 1,653,000
|
)
|
||||
|
Non-deductible stock compensation
|
583,000
|
434,000
|
||||||
|
Non-deductible meals and entertainment
|
3,000
|
4,000
|
||||||
|
Derivative Expense
|
-
|
3,460,000
|
||||||
|
Change in fair value of derivative expense
|
(1,296,000)
|
(5,543,000
|
)
|
|||||
|
Amortization of debt discount
|
155,000
|
3,462,000
|
||||||
|
Loss on extinguishment of debt
|
-
|
3,965,000
|
||||||
|
Change in Valuation Allowance
|
1,082,000
|
1,266,000
|
||||||
|
Actual tax expense (benefit)
|
$
|
-
|
$
|
-
|
||||
F-24
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
The following is a reconciliation of the provision for income taxes at the United States federal income tax rate to the income taxes reflected in the statement of operations:
|
|
December 31, 2011
|
December 31, 2010
|
||||||
|
Tax expense (credit) at statutory rate-federal
|
(31%)
|
(31%)
|
||||||
|
State tax expense net of federal tax
|
(8%)
|
(10%)
|
||||||
|
Changes in valuation allowance
|
39%
|
41%
|
||||||
|
Tax expense at actual rate
|
-%
|
-%
|
||||||
Note 12 Concentrations
The Company has the following concentrations:
|
(A)
|
Accounts Receivable
|
|
Customer
|
December 31, 2011
|
December 31, 2010
|
||
|
A
|
-%
|
51%
|
||
|
B
|
-%
|
37%
|
||
|
C
|
72%
|
8%
|
||
|
D
|
28%
|
2%
|
|
(B)
|
Sales
|
|
Customer
|
December 31, 2011
|
December 31, 2010
|
||
|
A
|
-%
|
40%
|
||
|
B
|
20%
|
15%
|
||
|
c
|
49%
|
- %
|
||
|
D
|
13%
|
-%
|
|
(C)
|
Accounts Payable
|
|
Vendor
|
December 31, 2011
|
December 31, 2010
|
||
|
A
|
34%
|
29%
|
||
|
B
|
-%
|
10%
|
F-25
NXT Nutritionals Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
December 31, 2011 and 2010
|
(D)
|
Purchases
|
|
Vendor
|
December 31, 2011
|
December 31, 2010
|
||
|
A
|
32%
|
59%
|
||
|
B
|
24%
|
44%
|
||
|
C
|
11%
|
16%
|
||
|
D
|
-%
|
16%
|
||
|
E
|
-%
|
11%
|
Note 13 Subsequent Event
On March 12, 2012, the Company converted principal and interest of $278,696 into 1,114,784 shares of the Company’s common stock at a conversion rate of $0.25.
F-26
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
Not applicable.
(a) EVALUATION OF DISCLOSURE CONTROLS AND PROCEDURES
Based on their evaluation as of the end of the period covered by this Annual Report on Form 10-K, our Chief Executive Officer and Chief Financial Officer have concluded that our disclosure controls and procedures (as defined in Rules 13a-15(c) and 15d-15(e) under the Exchange Act) are not effective to ensure that information required to be disclosed by us in report that we file or submit under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the Securities and Exchange Commission’s rules and forms and to ensure that information required to be disclosed by us in the reports that we file or submit under the Exchange Act is accumulated and communicated to our management, including our chief executive officer and chief financial officer, as appropriate to allow timely decisions regarding required disclosure.
(b) MANAGEMENT’S REPORT ON INTERNAL CONTROL OVER FINANCIAL REPORTING
This company’s management is responsible for establishing and maintaining internal controls over financial reporting and disclosure controls. Internal Control Over Financial Reporting is a process designed by, or under the supervision of, the Company’s principal executive and principal financial officers, or persons performing similar functions, and effected by the issuer’s board of directors, management and other personnel, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles and includes those policies and procedures that:
1. Pertain to the maintenance of records that in reasonable detail accurately and fairly reflect the transactions and dispositions of the assets of the issuer;
2. Provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the issuer are being made only in accordance with authorizations of management and directors of the registrant; and
3. Provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the issuer’s assets that could have a material effect on the financial statements.
Disclosure controls and procedures are designed to ensure that information required to be disclosed in reports filed under the Securities Exchange Act of 1934, as amended, is appropriately recorded, processed, summarized and reported within the specified time periods.
Management has conducted an evaluation of the effectiveness of our internal control over financial reporting as of December 31, 2009 based on the framework established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (“COSO”).
Based on this assessment, management concluded that as of December 31, 2011 it had material weaknesses in its internal control procedures.
A material weakness is a control deficiency, or combination of control deficiencies, in internal control over financial reporting such that there is a reasonable possibility that a material misstatement of our annual or interim financial statements would not be prevented or detected on a timely basis.
30
As of December 31, 2011, we have concluded that our internal control over financial reporting was ineffective as of December 31, 2011.
The Company’s assessment identified certain material weaknesses which are set forth below:
Financial Statement Close Process
There are insufficient written policies and procedures for accounting and financial reporting with respect to the requirements and application of US GAAP and SEC disclosure requirements;
There is a lack of formal process and timeline for closing the books and records at the end of each reporting period.
The Company currently has an insufficient level of monitoring and oversight controls for review and recording of stock issuances, agreements and contracts, including insufficient documentation and review of the selection and application of generally accepted accounting principles to significant non-routine transactions. In addition this has resulted in a lack of controls over the issuance of the Company’s stock which resulted in several instances of extra or duplicate shares being issued.
These weaknesses restrict the Company’s ability to timely gather, analyze and report information relative to the financial statements.
Functional Controls and Segregation of Duties
Because of the company’s limited resources, there are limited controls over information processing, and no internal controls over the accuracy, completeness and authorization of transactions.
There is an inadequate segregation of duties consistent with control objectives. Our company’s management is composed of a small number of individuals resulting in a situation where limitations on segregation of duties exist. In order to remedy this situation we would need to hire additional staff to provide greater segregation of duties. Currently, it is not feasible to hire additional staff to obtain optimal segregation of duties. Management will reassess this matter in the following year to determine whether improvement in segregation of duty is feasible.
There is a lack of top level reviews in place to review targets, product development, joint ventures or financing. All major business decisions are carried out by the officers with board of director approval when needed.
Accordingly, as the result of identifying the above material weaknesses we have concluded that these control deficiencies resulted in a reasonable possibility that a material misstatement of the annual or interim financial statements will not be prevented or detected on a timely basis by the company’s internal controls.
Management believes that the material weaknesses set forth above were the result of the scale of our operations and are intrinsic to our small size. Management believes these weaknesses did not have a material effect on our financial results and intends to take remedial actions upon receiving funding for the company’s business operations.
This annual report does not include an attestation report of our registered public accounting firm regarding internal control over financial reporting. Management’s report was not subject to attestation by our registered public accounting firm pursuant to temporary rules of the Securities and Exchange Commission that permit the Company to provide only management’s report herein.
(c) CHANGES IN INTERNAL CONTROLS OVER FINANCIAL REPORTING
We are committed to improving our financial organization. As part of this commitment, we will create a position to segregate duties consistent with control objectives and will increase our personnel resources and technical accounting expertise within the accounting function when funds are available to us by preparing and implementing sufficient written policies and checklists which will set forth procedures for accounting and financial reporting with respect to the requirements and application of US GAAP and SEC disclosure requirements.
31
Management believes that preparing and implementing sufficient written policies and checklists will remedy the material weaknesses pertaining to insufficient written policies and procedures for accounting and financial reporting with respect to the requirements and application of US GAAP and SEC disclosure requirements. We intend to take appropriate and reasonable steps to make the necessary improvements to remediate these deficiencies, including:
(1) We will document a formal code of ethics.
(2) We will revise processes to provide for a greater role of independent board members in the oversight and review until such time that we are adequately capitalized to permit hiring additional personnel to address segregation of duties issues, ineffective controls over the revenue cycle and insufficient supervision and review by our corporate management.
(3) We will continue to update the documentation of our internal control processes, including formal risk assessment of our financial reporting processes.
We intend to consider the results of our remediation efforts and related testing as part of our year-end 2010 assessment of the effectiveness of our internal control over financial reporting.
Subsequent to December 31 2011, we have undertaken the following steps to address the deficiencies stated above:
|
·
|
Continued the development and documentation of internal controls and procedures surrounding the financial reporting process, primarily through the use of account reconciliations, and supervision.
|
32
PART III
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
The following table lists certain information about the directors and executive officers of the Company:
|
Name
|
Age
|
Director
Since
|
Officer
Since
|
Position with the Company
|
|
Francis McCarthy
|
64
|
2009
|
2009
|
Director, President, Secretary and Chief Executive Officer
|
|
Mark A. Giresi
|
53
|
2009
|
2010
|
Director and Chief Operating Officer
|
|
David Briones
|
36
|
-
|
2009
|
Chief Financial Officer
|
|
Richard M. Jordan
|
56
|
2009
|
2009
|
Director, Executive Vice President and General Manager
|
|
Theodore Mandes, II
|
62
|
2009
|
-
|
Director
|
|
David Deno
|
53
|
2009
|
-
|
Director
|
|
Dr. Paul S. Auerbach
|
60
|
2009
|
-
|
Director
|
|
Ann McBrien
|
51
|
2009
|
-
|
Director
|
|
Philip Sheibley
|
53
|
2012
|
-
|
Director
|
The background of each of the Company’s directors and executive officers is set forth below.
Francis (Michael) McCarthy, President, Secretary, Chief Executive Officer/ Director
NXT Nutritionals, Inc. is led by entrepreneur and food and beverage industry veteran Francis McCarthy, an executive with over 25 years of experience in the sector. He is the former Vice President of Pepsi-Cola’s Fountain Solutions from 1992 to 1995. While at Pepsi, he was responsible for the development and integration of their Citrus Hill juice products. Michael was also instrumental in the negotiation and subsequent integration of Hawaiian Punch into the Pepsi-Cola system. The results of this assimilation increased fountain sales more than five hundred percent. He was also a key component in a national sales effort that was successful in landing several large national accounts. He was also part of a special acquisition team that was working to increase Pepsi-Cola’s fountain presence.
As an entrepreneur, Mr. McCarthy has been the driving force behind the development of numerous beverage products. He was responsible for the development of Juice Creations, a product that was successfully launched in Massachusetts and then expanded nationally. After selling his business, Juice Creations, to Pepsi-Cola, Mr. McCarthy built the Juice Creations product line into a $63 million business in the first year after Pepsi Cola’s acquisition.
After his career at Pepsi-Cola, from 1996 to 2001 he was instrumental in the growth of Al’s Beverage Company from a small independent manufacturer to one of the largest independent Royal Crown Cola fountain manufacturers in the United States. In his position as Chief Operating Officer of Al’s Beverage Company, Mr. McCarthy was responsible for building and operation of one of the most modern independent fountain plants in the country.
Mr. McCarthy continued to work as an independent consultant for Proctor & Gamble and Pepsi-Cola from 2001 until 2003, when he began work with NXT, LLC and Healthy Dairy, LLC, the subsidiaries of NXT Nutritionals.
33
Mark A. Giresi, Chief Operating Officer, General Counsel and Director
Mark A. Giresi is a retail executive with 25 years of experience in various operations and legal roles in the United States and internationally. From September 2007 through February 2008, he served as Executive Vice President, for Limited Brands, Inc., a $9 billion dollar specialty retail business trading under the Victoria’s Secret, Bath & Body Works, White Barn Candle Co., and Henri Bendel brands. In that role, he led the development of the Company’s International growth strategy and the day-to-day management of the growth of Victoria’s Secret and Bath & Body Works outside of the United States. Between May 2005 and August 2007, he served as Executive Vice President of Retail Operations, responsible for the Company’s Real Estate, Store Design and Construction, Visual Merchandising, Store Operations, and Loss Prevention and Brand Protection functions. He was on the Executive Committee of the Company, responsible for its strategy and the overall business performance of its branded specialty retail businesses. Mr. Giresi joined Limited Brands in February, 2000 as Vice President of Store Operations. From August, 2001 until May, 2005 he served as Senior Vice President, Chief Stores Officer for the Company’s almost 4,000 retail stores.
Prior to joining Limited Brands, Mr. Giresi was Senior Vice President of U.S. Franchise Operations and Development for Burger King Corporation, responsible for the restaurant operations and support to almost 8,000 franchise-owned and operated stores together with all real estate investments in the U.S. business. From 1993 through 1998 he held the position of Senior Vice President, Worldwide General Counsel and Secretary for Burger King. During that time, he was also a member of the Board of Directors of Restaurant Services, Inc., the independent purchasing cooperative for the U.S. Burger King System. He began his career with Burger King as a real estate attorney in 1985 and has published numerous articles and spoken on various franchise and intellectual property law topics. Mr. Giresi was a member of the first United States – South Africa Commercial Law Delegation established by the United States Department of Commerce and the government of South Africa.
Mr. Giresi has served on several philanthropic and business association boards, including the Board of Directors of the Beacon Council, the business development agency for Miami-Dade County, Florida, the Miami Philharmonic, the International Franchise Association and the Boys and Girls Clubs of Columbus, Ohio where he served as the Treasurer and a member of the Executive and Human Resources Committees. Since 2005 he has served on the Board of Directors of UFood Grille, a publicly-traded, franchised restaurant business, and since 1995 on the audit committee of Fiduciary Trust International of the South, an investment management firm.
Mr. Giresi is a past recipient of the Italian-American businessman of the year by the National Italian American Foundation in South Florida. In 2004, he received the Champions Award from Safe Horizons, a leading domestic violence prevention organization in New York City. He is an attorney at law of the State of New Jersey. He earned a law degree in 1983 from Seton Hall University and a Bachelor of Science degree in accounting in 1980 from Villanova University.
David Briones, Chief Financial Officer
Since October 1, 2010, Mr. Briones has acted as the managing member of Brio Financial Group, LLC, a financial reporting consulting firm. From January 2006 through September 2010, Mr. Briones had managed the public company and hedge fund practices at Bartolomei Pucciarelli, LLC (“BP”). Within that capacity, Mr. Briones performed audit services, outsourced CFO functions, and/or consulted clients through difficult SEC comment periods particularly through application of complex accounting principles for a vast public company client base. BP is a registered firm with the Public Company Accounting Oversight Board. BP is an independent member of the BDO Seidman Alliance.
Mr. Briones also served as the chief financial officer of Clear-Lite Holdings, Inc. from August 3, 2009 to March 21, 2011. Prior to joining BP, Mr. Briones was an auditor with PricewaterhouseCoopers LLP in New York, New York. Mr. Briones specialized in the financial services group, and most notably worked on the MONY Group, Prudential Financial, and MetLife initial public offerings.
Mr. Briones has a Bachelor of Science in Accounting from Fairfield University, Fairfield, Connecticut. David's professional interests are complemented by a strong commitment to philanthropy and humanitarian causes. His volunteer efforts have benefited such organizations as Habitat for Humanity, New York Cares and Junior Achievement.
34
Richard M. Jordan, Executive Vice President, General Manager and Director
Richard Jordan is a second-generation member in the food industry, now with over twenty four years experience. He received a Bachelor of Science Degree in Advertising and Marketing from Arizona State University, and holds a Masters Degree from Cornell University in Business and Supermarketing.
After graduating from college, Richard began his career in the food industry with Nabisco Foods, Inc. After three years with Nabisco, Richard was promoted through various positions, achieving the position of Sales Manager, was honored as Salesman of the year for Nabisco Brands and served as assistant to the Vice President of Sales and Marketing.
In 1983 Richard joined The New York State Food Merchants Association, a lobbying and trade relations firm, accepting the position of Director of Trade Relations. In 1987 he was promoted to the office of Executive Vice President and General Manager, he held this position until 1991.
Richard then served at Jordan Associates Inc., a food brokerage firm, where he oversaw the Northeast and Middle Atlantic States. Since 2005 Mr. Jordan was Director of Sales for Healthy Dairy, LLC, now a wholly owned subsidiary of NXT Nutritionals, Inc. In addition to being past President of The Catholic Institute of the Food Industry, Richard chairs a committee on the Food Industry for Covenant House. He also sits on the advisory committee for the Deborah Heart and Lung Foundation and the Calvary Hospital.
Theodore Mandes, II, Director
Theodore Mandes, II is an experienced entrepreneur, executive and investor. Mr. Mandes played professional golf until 1975, and then entered the business world as an account executive in the ladies apparel business. After a successful stint in New York, he founded Mandes and Associates. It was during this time that Mr. Mandes honed his marketing skills to make his company one of the largest agents to the trade. During his career in the apparel business, Mr. Mandes developed divisions of Federated Department Stores, May Co., JC Penny and other multiple store groups into multi-million dollar accounts for many of the major wholesale companies that his firm represented.
As an investor, Director and entrepreneur, Mr. Mandes has consistently recognized and understood trends well ahead of the rest of the market, from new innovations in apparel to developing inserts and needs for diabetic patients with conditions like peripheral neuropathy.
In 1994, he founded Bioform, Inc. with Stephen Warner, a pedorthic foot care company that dealt with orthopedic foot problems but also addressed the needs of diabetics. While developing this opportunity, the urgent need to address the alarming increase in the incidence of obesity and diabetes in the United States became apparent to Mr. Mandes.
Mr. Mandes sits on the Board of Directors of the Raymond F. Kravis Center and the Palm Beach County performing arts center. He also developed and chairs the Palm Beach Wine Auction, the most productive fundraising event for the performing arts center's award winning education department. His mission "is to further advance the educational experience of our youth - it is more than just a cause it is an ongoing obligation." Over 85,000 students have attended live performance camps and other workshops every year for the past 16 years.
35
David Deno, Director
Since December of 2009, David Deno has served as the Chief Financial Officer of the international division of Best Buy Co., Inc., a specialty retailer of consumer-electronics and related products and services, since December 2009. Prior to that he was a Managing Director with Obelysk Capital, a private equity firm, from May 2009 to December 2009. From January 2008 to February 2009, Mr. Deno was the President and then CEO of Quiznos LLC, an operator of quickserve restaurants. From June 2006 to February 2009, he was a Managing Director with CCMP Capital Advisors, LLC, a private equity firm. Prior to this, he had a 15-year career with YUM! Brands, Inc. where between 1999 and 2004 he served first as Chief Financial Officer and later as Chief Operating Officer. Mr. Deno was instrumental in growing YUM Brands and building shareholder value after its spinoff from PepsiCo in 1997. Mr. Deno also held senior positions in YUM's International and Pizza Hut divisions. Prior to joining YUM, David worked for Burger King Corporation for 8 years in various finance, real estate, and operations assignments.
David Deno has also served on the Board of Directors for Peet’s Coffee since August 2006.
Dr. Paul S. Auerbach, Director
Since 2005, Dr. Paul S. Auerbach has served as Professor of Surgery in the Division of Emergency Medicine at Stanford University School of Medicine. Between 2003 through 2004, Dr. Auerbach served as the Chief Operating Officer of KAI Pharmaceuticals, Inc., a developmental and discovery pharmaceutical company. From 1999 through 2003 he was a partner at Delphi Ventures, a venture capital firm which specializes in early-stage healthcare investing. Between 1997 and 1999, Dr. Auerbach was the Chief Operating Officer of MedAmerica, a medical practice support and consulting company. Dr. Auerbach currently serves as a member of the Board of Directors for Pacific Partners Management Services, Inc. From 2007 through 2009, Dr. Auerbach was a Member on the Board of Directors for VitalWear and from 2008 through 2009, Dr. Auerbach also served on the Board of Directors for Med-Vantage.
In addition Dr. Auerbach is a founder and past president of the Wilderness Medical Society, editor of the definitive medical reference text Wilderness Medicine, 5th Edition, and author of Medicine for the Outdoors, which is a leading book on outdoor health for laypersons. Dr. Auerbach practices emergency medicine, teaches, performs research, and advises numerous agencies and organizations, including serving as an advisory board member to the AARP “Fat 2 Fit” Community Challenge. Dr. Auerbach has been hailed as a Hero of Emergency Medicine by the American College of Emergency Physicians. He also serves on the National Medical Committee for the National Ski Patrol. Dr. Auerbach is a recipient of numerous awards including the DAN America Award from the Divers Alert Network, Outstanding Contribution in Education Award from the American College of Emergency Physicians, a NOGI Award in 2006 from the Academy of Underwater Arts and Sciences, Diver of the Year for Science in 2008 from Beneath the Sea, and DAN/Rolex Diver of the Year in 2009.
Dr. Auerbach is a graduate of Duke University and Duke University School of Medicine, as well as the Stanford Graduate School of Business.
Ms. Ann McBrien, Director
Ms. McBrien has over 25 years of experience as an entrepreneur, and as a leader in national and international organizations. Ms. McBrien was the first female Vice President of Sales in Procter & Gamble history (1999). Ms. McBrien was a Vice President for Global Operations and Global Health Care Sales for Procter & Gamble for 20 years. She was responsible for the development of global pricing and promotion plans linking the largest customers and brands with a winning strategy. She was responsible for over $8 billion in sales and over 300 people globally. She also led the development of merchandising programs for global customers and led major global restructure of Customer Business Development organization.
From 2009 to the present, Ms. McBrien has worked as a Tremor Consultant with Procter & Gamble, connecting corporations with word of mouth marketing. Since 2008, Ms. McBrien has also worked for AM Marketing & Sales Concepts, LLC, where she coaches senior managers in delivery of concepts and ideas, strategic plan development and sales presentation.
36
From 2002 to 2006, Ms. McBrien co-founded OT OverTime, LLC, where she initiated the acquisition of a brand from Procter & Gamble, secured funding to launch a line of products nationally, and secured sales to Target, Kroger and regional chains. The company’s first year sales were over $2 million.
Ms. McBrien also served on the board of directors of Marriott Grande Ocean from 2001 through 2007.
Ms. McBrien received her BS in finance at the University of Illinois. She was part of the Mortar Board Honor program, president of Illini Pride and president of Kappa Kappa Gamma.
Mr. Philip B. Sheibley, Director
Mr. Sheibley is an experienced executive and venture capitalist. He spent 30 years as a management consultant with Accenture, where he focused on the Life Sciences area, holding a variety of leadership positions including North American industry lead and Global Lead for Management Consulting. He also served as a member of the Products Operating Group Executive Committee.
Mr. Sheibley’s consulting work focused on defining and executing strategies to create leading-edge business capabilities, working with U.S. and internationally-headquartered pharmaceutical companies, biotech, and medical product producers, as well as contract research organizations. Mr. Sheibley also played a leadership role in a number of joint-venture and new venture start-up engagements where company operating model and capability strategies needed to be defined for successful launch.
Since leaving Accenture, Mr. Sheibley has focused on venture capital investment and consulting, helping to structure a number of financing vehicles for early-stage companies, and participating as a board chair/member of several.
Code of Ethics
The Company has adopted a Code of Ethics which applies to the Company’s officers, directors and employees. A copy of the Company’s Code of Ethics is available on the Company’s website: nxtnutritionals.com.
The Code of Ethics has been adopted by the Board of Directors and any exceptions to the policies set forth in the Code of Ethics must be requested in writing addressed to the Audit Committee of the Board of Directors. If an executive officer requests an exception, the request must be delivered to the Chairman of the Audit Committee and no exceptions shall be effective unless approved by the Audit Committee.
37
ITEM 11. EXECUTIVE COMPENSATION
Summary Compensation Table
The following table sets forth information concerning the compensation of the Company’s (a) Principal Executive Officer, (b) Principal Financial Officer, and (c) the other three most highly compensated executive officers as specified by SEC rules (the “named executive officers”) for 2011, 2010 and 2009.
|
Salary ($)
|
Bonus ($)
|
Stock Awards ($)
|
Option Awards ($)(3)
|
Non-Equity Incentive Plan
|
Non-Qualified($)
|
All Other
Comp($) (4)
|
Total($)
|
||||||||||||||||||||||||||
|
Francis McCarthy,
|
2011
|
120,000 | 40,080 | 143,000 | 135,600 | 438,680 | |||||||||||||||||||||||||||
|
President,
|
2010
|
119,269 | - | 322,920 | - | 20,517 | 462,706 | ||||||||||||||||||||||||||
|
CEO
|
2009
|
121,250 | 18,000 | 139,250 | |||||||||||||||||||||||||||||
| - | |||||||||||||||||||||||||||||||||
|
Mark Giresi,
|
2011
|
120,000 | 79,280 | 286,000 | 145,000 | 630,280 | |||||||||||||||||||||||||||
|
COO and Director
|
2010
|
120,000 | - | 59,140 | 645,840 | 120,000 | 24,402 | 969,382 | |||||||||||||||||||||||||
|
2009
|
- | - | - | - | - | - | - | - | |||||||||||||||||||||||||
| - | |||||||||||||||||||||||||||||||||
|
David Briones,
|
2011
|
- | - | - | - | - | - | 72,000 | 72,000 | ||||||||||||||||||||||||
|
CFO (2)
|
2010
|
- | - | - | - | - | - | 141,500 | 141,500 | ||||||||||||||||||||||||
|
2009
|
- | - | - | - | - | - | 49,971 | 49,971 | |||||||||||||||||||||||||
(1) On August 18, 2008, we issued 610,000 shares of our common stock valued at $.01 per share in a share exchange of the above mentioned officers and directors as compensation for founder services.
(2) The CFO’s former accounting firm, Bartolomei Pucciarelli, LLC (“BP”), was paid cash for services rendered at a fixed rate of $39,000 per year and 200,000 shares of the Company’s common stock, vesting 25,000 shares per quarter for services related to the 2010 and 2009 financial statement preparation process. BP is entitled to additional compensation billed hourly for additional services. BP billed the Company $129,808 during the year ended December 31, 2009, of which $49,971 was paid by the Company. The overage of $90,808 was broken out between $75,000 for prior 2008 and prior restatement work and $15,000 for tax work. During 2010, BP billed the Company $65,163 and the Company paid $135,000. The overage of $26,163 was broken out between additional services required for work related to the Company’s SEC filings and tax return work. During the years ended December 31, 2010 and 2009, the full 200,000 shares of the stock award vested. The Company is yet to issue the shares to Mr. Briones, the shares have been accrued for in the financial statements, but the value is not presented in the executive compensation table. On October 1, 2010, Mr. Briones started his own consulting firm, Brio Financial Group, LLC (“Brio”). During 2010, the Company paid Brio $6,500. During 2011, the Company paid Brio $72,000
(3) The amounts set forth represent the aggregate fair value of the awards as of the grant date, computed in accordance with FASB ASC Topic 718, “Compensation-Stock Compensation.”
(4) Other compensation (for McCarthy and Giresi) includes: medical insurance, car allowance, reimbursed medical expenses, and FSA allotments. In addition, other compensation includes deferred salary.
38
Option Grants
Grants of Plan Based Awards in Fiscal Year 2011
The following table shows for the fiscal year ending December 31, 2011, certain information regarding grants of option awards to the named executive officers and directors:
|
Name
|
Grant Date
|
All Other Number of Securities Underlying Option
Awards
Number of Securities
UnderlyingOptions (#)
|
Exercise or
Base
Price of
Option
Awards($/share)
|
Grant Date Fair
Value of
Stock and
Option
Awards
($/share)
|
|||||||||
|
Francis McCarthy
|
-
|
-
|
-
|
-
|
|||||||||
|
Mark Giresi
|
- |
-
|
-
|
-
|
|||||||||
|
Richard Jordan
|
-
|
-
|
-
|
-
|
|||||||||
|
David Deno
|
-
|
-
|
-
|
-
|
|||||||||
|
Theodore Mandes
|
-
|
-
|
-
|
-
|
|||||||||
|
Paul Auerbach
|
-
|
-
|
-
|
-
|
|||||||||
|
Ann McBrien
|
-
|
-
|
-
|
-
|
|||||||||
There were no options granted during the year ended December 31, 2011.
Employment Agreement with Mr. Giresi
On January 26, 2010, we entered into a three year employment agreement with Mark Giresi pursuant to which he will serve as our Chief Operating Officer. Pursuant to the agreement, Mr. Giresi is entitled to receive monthly based compensation of $20,000, with 50% of this amount to be deferred and payable in its entirety on the earlier to occur of (1) the termination of the employment term or (2) a change in control.
Under the employment agreement, we agreed to grant Mr. Giresi the following restricted stock awards (the “Stock Awards”):
|
·
|
On or before February 28, 2010, (A) 400,000 shares of our common stock of which 40,000 shall fully vest on July 1, 2010 and an additional 40,000 shares shall fully vest on the first day of each calendar month over a ten (10) month period until April 1, 2011; and (B) 500,000 shares of our common stock, of which 40,000 shall fully vest on May 1, 2011, an additional 40,000 shares shall vest on each of June 1, August 1, September 1, October 1, 2011; and the remaining 220,000 shares shall fully vest on December 1, 2011.
|
|
·
|
On or before January 1, 2011, 500,000 shares of common stock which shall fully vest on December 31, 2011.
|
|
·
|
On or about January 1, 2012, 500,000 shares of common stock which shall fully vest on December 31, 2012.
|
39
Prior to December 1, 2011, the Stock Awards granted to Mr. Giresi shall be subject to a resale restriction as follows: (A) no more than 40,000 shares of the Stock Awards may be sold in any one month period and (B) no more than 10,000 shares of the Stock Award may be sold on any day during the Employment Term. After that date, there will be no resale restriction imposed on executive. In the event of a change of control of the Company, this resale restriction shall no longer be in effect.
In addition to the Stock Awards, we shall also be eligible to participate in and to receive annual stock options pursuant to our option plan in such amount as determined by the Board at a level commensurate with the Mr. Giresi’s position with us.
Employment Agreement with Francis McCarthy
On December 20, 2010, we entered into a three year employment agreement with Francis McCarthy pursuant to which he will serve as our Chief Operating Officer. Pursuant to the agreement, Mr. McCarthy is entitled to receive monthly based compensation of $20,000, with 50% of this amount to be deferred and payable in its entirety on the earlier to occur of (1) the termination of the employment term or (2) a change in control.
During the first year of the term, 360,000 shares of our common stock of which 30,000 shall fully vest monthly
During the second year of the term, 300,000 shares of common stock which shall fully vest on December 31, 2012.
During the third year of the term, 300,000 shares of common stock which shall fully vest on December 31, 2013.
Employment Agreement with Richard Jordan
On September 23, 2010, the Company entered into an employment agreement with Richard Jordan, our Executive Vice President. The employment agreement had an effective date of January 1, 2010. The terms of the agreements are as follows:
|
·
|
3 Year Term
|
|
·
|
$9,500 monthly base salary
|
|
·
|
Participation in bonus compensation program, maximum bonus of 100% annual salary
|
|
·
|
Stock compensation as follows:
|
|
o
|
25,000 shares monthly from August 1, 2010 through October 31, 2010; of which the first two 25,000 share issuances were granted during the year ended December 31, 2010.
|
|
o
|
Additional 180,000 shares of the Company’s common stock shall be granted on December 31, 2010
|
|
o
|
180,000 shares shall be granted during the year ended December 31, 2011
|
|
o
|
180,000 shares shall be granted during the year ended December 31, 2012
|
40
Potential Payments and Benefits upon Change of Control
Each of the officers are entitled to full payment of their respective deferred salary and vesting of any stock based grants as provided for in each of their employment contracts in the event of a change in control.
The following table shows information regarding the compensation earned during the fiscal year ended December 31, 2011 by our directors.
|
Fees ($)
|
Stock
Awards($)
|
Option Awards ($) (1)
|
Non-Equity Incentive Plan
Compensation ($)
|
Non-qualified Deferred Compen-sation Earnings ($)
|
All other Compen-sation ($)
|
Total ($)
|
||||||||||||||
|
David Deno
|
2,000
|
-
|
- | - |
-
|
- |
2,000
|
|||||||||||||
|
Ann Mcbrien
|
2,000
|
-
|
- | - |
-
|
- |
2,000
|
|||||||||||||
|
Paul S. Auerbach
|
2,000
|
-
|
- | - |
-
|
- |
2,000
|
|||||||||||||
|
Francis McCarthy
|
2,000
|
-
|
- | - |
-
|
- |
2,000
|
|||||||||||||
|
Mark Giresi
|
2,000
|
-
|
- | - |
-
|
- |
2,000
|
|||||||||||||
|
Theodore Mandes II
|
2,000
|
- | - | - |
-
|
- |
2,000
|
|||||||||||||
|
Richard M. Jordan
|
2,000
|
-
|
- | - |
-
|
- |
2,000
|
|||||||||||||
(1) The amounts set forth represent the aggregate fair value of the awards as of the grant date, computed in accordance with FASB ASC Topic 718, “Compensation-Stock Compensation.”
We do not currently have an established policy to provide compensation to members of our Board of Directors for services rendered in that capacity. We plan to develop such a policy in the near future.
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
The following table sets forth certain information concerning the beneficial ownership of the Company’s common stock as of April 16, 2011, by:
• each person known by the Company to own more than 5% of the Company’s common stock,
• each Director and nominee for Director of the Company,
• each of the Company’s executive officers named in the “Summary Compensation Table” and
• all of the Company’s executive officers and Directors of the Company as a group.
41
Except as otherwise indicated, all shares are owned directly and the shareholders listed possesses sole voting and investment power with respect to the shares shown. As of April 16, 2012, we have 73,953,106 shares of Common Stock issued and outstanding.
|
Name
|
Number of Shares
Beneficially Owned (1)
|
Percent of Shares
|
|||
|
Francis McCarthy, President, CEO and Director
|
4,496,000
|
*
|
4.07%
|
||
|
David Briones, CFO
|
200,000
|
**
|
-
|
||
|
Richard M. Jordan, Director
|
430,000
|
**
|
-
|
||
|
David Deno, Director
|
1,447,780
|
(2)
|
1.31%
|
||
|
Mark A. Giresi, Chief Operating Officer and Director
|
2,033,750
|
(3)
|
1.84%
|
||
|
Dr. Paul S. Auerbach, Director
|
542,500
|
(4)
|
-
|
||
|
Theodore Mandes, II, Director
|
1,065,000
|
*
|
1.0%
|
||
|
Ann McBrien, Director
|
250,000
|
-
|
|||
|
Officers and Directors as a Group (8)
|
10,465,030
|
9.48%
|
|||
|
NXT Investment Partners, LLC (5)
|
36,396,684
|
33.0%
|
|||
* Subject to a Lock-Up Agreement dated February 12, 2009, pursuant to which these stockholders have agreed not to sell or otherwise dispose of their shares of our Common Stock for a period of 18 months from the close of the Share Exchange Agreement on February 12, 2009.
** less than 1%
(1) A person is considered to beneficially own any shares: (i) over which such person, directly or indirectly, exercises sole or shared voting or investment power, or (ii) of which such person has the right to acquire beneficial ownership at any time within 60 days (such as through exercise of stock options or warrants). Unless otherwise indicated, voting and investment power relating to the shares shown in the table for our directors and executive officers is exercised solely by the beneficial owner or shared by the owner and the owner’s spouse or children.
(2) Includes 547,780 shares of Common Stock issueable upon the conversion of certain convertible debentures, 325,000 shares of Common Stock, issueable upon the conversion of certain Series A Warrant, and 325,000 shares of Common Stock issueable upon the conversion of certain Series B Warrant, issued to Mr. Deno in the private placement closed on August 27, 2009.
(3) Includes 130,000 shares of Common Stock issueable upon the conversion of certain convertible debentures, 81,250 shares of Common Stock issueable upon the conversion of certain Series A Warrant, and 81,250 shares of Common Stock issueable upon the conversion of certain Series B Warrant, issued to Mr. Giresi in the private placement closed on August 27, 2009. The remaining 1,741,250 shares are subject to the Lock-Up Agreement dated February 12, 2009, pursuant to which Mr. Giresi agreed not to sell the shares for a period of 18 months from the close of the Share Exchange Agreement dated February 12, 2009.
(4) Includes 130,000 shares of Common Stock issueable upon the conversion of certain convertible debentures, 81,250 shares of Common Stock issueable upon the conversion of certain Series A Warrant, and 81,250 shares of Common Stock issueable upon the conversion of certain Series B Warrant, issued to Mr. Auerbach in the private placement closed on August 27, 2009.
(5) Includes the percentage NXT Investment Partners, LLC is convertible into as of April 16, 2012.
42
Equity Compensation Plans
The following table provides information generally as of December 31, 2011, the last day of fiscal 2011, regarding securities to be issued on exercise of stock options, and securities remaining available for issuance under our equity compensation plans that were in effect during fiscal 2011. No other issuances were made
|
Plan Category
|
Number of securities to be issued upon exercise of outstanding options, warrants and rights
|
Weighted average exercise price of outstanding options, warrants and rights
|
Number of securities remaining available for future issuance under equity compensation plans
|
|||||||
|
Equity compensation plans approved by shareholders
|
-
|
-
|
-
|
|||||||
|
Equity compensation plans not approved by shareholders
|
15,760,000
|
|
$ |
0.22
|
14,240,000
|
|||||
|
Total
|
15,760,000
|
|
$ |
0.22
|
14,240,000
|
|||||
43
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
In November 2011, three directors loaned the Company $12,498 in aggregate. The loans bore no interest, was not secured and is due on demand. As of April 15, 2012, the loans remain outstanding.
In December 2009, Mark Giresi, a Director loaned the Company $25,000. The loan bore no interest, was not secured and was due on demand. The Company repaid the $25,000 in February 2010.
On March 7, 2008, the Company entered into a loan agreement with Ed Tubel, a former related party to borrow an amount up to $388,500, of which $120,274 was advanced, with no stated terms during December 2007. An additional $268,226 was advanced during 2008. These loans bear interest at 6% with a default rate of interest of 12%. These loans were initially due on March 7, 2010. The Company repaid $90,000 of these advances in May 2009, and an additional $90,000 was repaid in February 2010. In November 2010, $40,000 was paid on the loan and Ed Tubel agreed to defer receipt of the balance due on the loan until such time as the company achieves profitability or raises a minimum of $10,000,000 in capital to operate the Company.
44
ITEM 14. PRINCIPAL ACCOUNTING FEES AND SERVICES
Berman & Company, P.A. the Company’s independent registered public accountants for the fiscal years ending December 31, 2011 and 2010, performed the review of each of the Company's quarterly reports for the 2011 and 2010 fiscal years and the audit of the Company's financial statements for the years ended December 31, 2011and 2010.
The following table presents fees billed for professional audit and other services rendered by Berman and Co. and for the periods presented.
|
Fiscal 2011
|
Fiscal 2010
|
|||||||
|
Audit Fees(1)
|
$
|
94,000
|
$
|
118,250
|
||||
|
Audit Related Fees
|
-
|
-
|
||||||
|
Tax Fees
|
-
|
-
|
||||||
|
All Other Fees
|
-
|
-
|
||||||
|
Total
|
$
|
94,000
|
$
|
118,250
|
||||
(1) Audit Fees consist of fees billed for professional services rendered for the audit of the Company's consolidated annual financial statements and review of the interim consolidated financial statements included in quarterly reports and services normally provided in connection with statutory and regulatory filings or engagements.
The Audit Committee's policy is to pre-approve all audit and non-audit services provided by the independent auditors. These services may include audit services, audit-related services, tax services and other services. Pre-approval is generally provided for up to one year and any pre-approval is detailed as to the particular service or category of services and is generally subject to a specific budget. During the 2010 fiscal year, 100% of the audit and tax services were pre-approved by the Audit Committee.
45
ITEM 15. EXHIBITS, FINANCIAL STATEMENT SCHEDULES.
(a) Documents filed as part of this Annual Report
1. Financial Statements
2. Financial Statement Schedules
3. Exhibits
|
EXHIBIT NO.
|
TITLE OF DOCUMENT
|
|
2.1
|
Share Exchange Agreement dated February 12, 2009 by and among NXT Nutritionals Holdings, Inc. , NXT Nutritionals, Inc. and the shareholders of NXT Nutritionals, Inc. (1)
|
|
3.1
|
Articles of Incorporation (7)
|
|
3.2
|
By-Laws (5)
|
|
10.1
|
Employment Agreement by and between NXT Nutritionals Holdings, Inc. and Francis McCarthy (1)
|
|
10.2
|
Employment Agreement by and between NXT Nutritionals Holdings. Inc. and David Briones ( 2 )
|
|
10.3
|
Employment Agreement by and between NXT Nutritionals Holdings, Inc. and Mark Giresi (9)
|
|
10.4
|
Lock-Up Agreement with Management Shareholders (1)
|
|
10.5
|
Licensing Agreement by and between NXT Nutritionals Holdings, Inc, and Mine O’Mine, Inc. dated November 30, 2009 (3)
|
|
10.6
|
Endorsement Agreement by and between NXT Nutritionals Holdings, Inc, and Eddie George dated March 3, 2010. (7)
|
|
10.7
|
Securities Purchase Agreement dated February 26, 2010 (4)
|
|
10.8
|
Security Agreement dated February 26, 2010 (4)
|
|
10.9
|
Subsidiaries Guarantee Agreement dated February 26, 2010 (4)
|
|
10.10
|
Registration Rights Agreement dated February 26, 2010 (4)
|
|
10.11
|
Form of Debenture (6)
|
|
10.12
|
Form of Series A Warrant (6)
|
|
10.13
|
Form of Series B Warrant(6)
|
|
10.14
|
Endorsement Agreement by and between NXT Nutritionals Holdings, Inc. and Blair Underwood dated April 22, 2009. (7)
|
|
10.15
|
Director Agreement by and between NXT Nutritionals Holdings, Inc. and David Deno dated August 30, 2009. (8)
|
|
10.16
|
Director Agreement by and between NXT Nutritionals Holdings, Inc. and Ann McBrien dated October 19, 2009. (8)
|
|
10.17
|
Director Agreement between NXT Nutritionals Holdings, Inc. and Dr. Paul S. Auerbach dated June 30, 2009. (8)
|
|
10.18
|
Modification and Amendment Agreement by and among NXT Nutritional Holdings, Inc. and Purchasers identified on the signature page thereto, dated September 1, 2010. (10)
|
|
10.19
|
Endorsement and Service Agreement by and between NXT Nutritional Holdings, Inc. and Dara Torres, dated September 21, 2010 (11)
|
|
10.20
|
Consulting Agreement by and between NXT Nutritionals Holdings, Inc., and Richard Kozlenko dated June 1, 2010 (12)
|
|
10.21
|
Employment Agreement by and between NXT Nutritionals Holdings, Inc., and Richard M. Jordan dated January 1, 2010. (12)
|
|
10.22
|
Bartolomei Pucciarelli Services Agreement (13)
|
|
10.23
|
2010 Stock Option Plan (13)
|
46
|
10.24
|
Form of Stock Option Agreement (13)
|
|
10.25
|
Securities Purchase Agreement dated November 21, 2011 (14)
|
|
10.26
|
2011 Note (14)
|
|
10.27
|
Subsidiary Guarantee (14)
|
|
10.28
|
Security Agreement (14)
|
|
10.29
|
Certificate of Designation relating to Series A Convertible Preferred Stock (14)
|
|
10.30
|
Lock-Up Agreement (14)
|
|
10.31
|
Registration Rights Agreement dated November 21, 2011 (14)
|
|
10.32
|
Fourth Modification Agreement (15)
|
|
10.33
|
First Amendment (15)
|
|
31.1
|
Certification of CEO pursuant to 18 U.S.C. Section 1350 as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. (16)
|
|
31.2
|
Certification of CFO pursuant to 18 U.S.C. Section 1350 as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. (16)
|
|
32.1
|
Certification of CEO pursuant to 18 U.S.C. Section 1350 as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. (16)
|
|
32.1
|
Certification of CFO pursuant to 18 U.S.C. Section 1350 as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. (16)
|
(1) Included as exhibits to the current report on Form 8-K filed on January 27, 2010.
(2) Included as exhibit to the current report on Form 8-K filed on February 12, 2009.
(3) Included as exhibit to the registration statement on Form S-8 filed on December 1, 2009.
(4) Included as exhibits to the current report on Form 8-K filed on March 1, 2010.
(5) Included as exhibit to the registration statement on Form SB-2 filed on November 27, 2007.
(6) Included as exhibits to the current report on Form 8-K filed on September 2, 2009.
(7) Included as exhibits to Amendment No. 1 to the registration statement on Form S-1 filed on June 17, 2010.
(8) Included as exhibits to Amendment No.2 to the registration statement on Form S-1 filed on August 4, 2010.
(9) Included as exhibit to current report on Form 8-K filed on January 27, 2010.
(10) Included as exhibit to current report on Form 8-K filed on September 2, 2010.
(11) Included as exhibit to current report on Form 8-K filed on September 22, 2010.
(12) Included as exhibit to current report on Form S-1 filed on December 30, 2010.
(13) Included as exhibit to current report on Form S-1/A filed on February 10, 2011.
(14) Included as exhibit to current report on Form 8-K filed on November 21, 2011.
(15) Included as exhibit to current report on Form 8-K filed on November 11, 2011.
(16) Filed herewith
47
SIGNATURES
In accordance with Section 13 or 15(d) of the Exchange Act, the registrant caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
NXT Nutritionals Holdings, Inc.
|
|||
|
Date: April 16, 2012
|
By:
|
/s/Francis McCarthy
|
|
|
Name: Francis McCarthy
|
|||
|
Title: President and Chief Executive Officer
|
|||
|
Date: April 16, 2012
|
By:
|
/s/ David Briones
|
|
|
Name: David Briones
|
|||
|
Title: Chief Financial Officer and Principal Accounting Officer
|
|||
In accordance with the requirements of the Securities Exchange Act of 1934, this report has been signed by the following persons on behalf of the registrant and in the capacities and on April 16, 2012.
|
Signature
|
Title
|
Date
|
||
|
/s/Francis McCarthy
|
President, CEO, Secretary and Director
|
Date: April 16, 2012
|
||
|
Francis McCarthy
|
||||
|
/s/ David Briones
|
Chief Financial Officer
|
Date: April 16, 2012
|
||
|
David Briones
|
||||
|
/s/ Richard M. Jordan
|
Executive Vice President, General Manager, and Director
|
Date: April 16, 2012
|
||
|
Richard M. Jordan
|
||||
|
/s/ Mark A. Giresi
|
Director and Chief Operating Officer
|
Date: April 16, 2012
|
||
|
Mark A. Giresi
|
||||
|
/s/ David Deno
|
Director
|
Date: April 16, 2012
|
||
|
David Deno
|
||||
|
/s/ Theodore Mandes, II
|
Director
|
Date: April 16, 2012
|
||
|
Theodore Mandes, II
|
||||
|
/s/ Paul S. Auerbach
|
Director
|
Date: April 16, 2012
|
||
|
Paul S. Auerbach
|
||||
|
/s/ Ann McBrien
|
Director
|
Date: April 16, 2012
|
||
|
Ann McBrien
|
||||
| /s/ Philip Sheibley | Director | Date: April 16, 2012 | ||
| Philip Sheibley | ||||
48
