Attached files
| file | filename |
|---|---|
| EX-31.2 - EXHIBIT 31.2 - STANDARD DIVERSIFIED INC. | ex31-2.htm |
| EX-32.2 - EXHIBIT 32.2 - STANDARD DIVERSIFIED INC. | ex32-2.htm |
| EX-31.1 - EXHIBIT 31.1 - STANDARD DIVERSIFIED INC. | ex31-1.htm |
| EX-32.1 - EXHIBIT 32.1 - STANDARD DIVERSIFIED INC. | ex32-1.htm |
| EX-23.1 - EXHIBIT 23.1 - STANDARD DIVERSIFIED INC. | ex23-1.htm |
| EX-10.41 - EXHIBIT 10.41 - STANDARD DIVERSIFIED INC. | ex10-41.htm |
| EX-10.42 - EXHIBIT 10.42 - STANDARD DIVERSIFIED INC. | ex10-42.htm |
UNITED STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
|
FORM
10-K
|
||
|
(Mark
One)
|
|
|
x
|
ANNUAL
REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF
1934
|
|
For
the Fiscal Year Ended December 31, 2009
|
|
|
o
|
TRANSITION
REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF
1934
|
For
the Transition Period From ____________ to ____________
Commission
File No. 000-22400
STRATEGIC
DIAGNOSTICS INC.
(Exact
name of Registrant as specified in its charter)
|
Delaware
|
56-1581761
|
|||
|
(State
or other jurisdiction of
|
(I.R.S.
Employer
|
|||
|
incorporation
or organization)
|
identification
no.)
|
|||
|
111
Pencader Drive
|
||||
|
Newark,
Delaware
|
19702
|
|||
|
(Address
of principal executive offices)
|
(Zip
Code)
|
Registrant’s
telephone number, including area code: (302) 456-6789
Securities
registered pursuant to Section 12(b) of the Act: None
|
Title
of Each Class
|
Name
of Each Exchange on Which Registered
|
|||||
|
Common
Stock, $0.01 par value
|
The
NASDAQ Stock Market LLC
|
|||||
Securities
registered pursuant to Section 12(g) of the Act:
None
Indicate by check mark if
the Registrant is a well-known seasoned issuer, as defined in Rule 405 of the
Securities Act. Yes
o No x
Indicate by check mark if
the Registrant is not required to file reports pursuant to Section 13 or Section
15(d) of the Exchange
Act. Yes o No x
Indicate by check mark
whether the Registrant: (1) has filed all reports required to be filed by
Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding
12 months (or for such shorter period that the registrant was required to file
such reports), and (2) has been subject to such filing requirements for the past
90 days. Yes x No o
Indicate by check mark whether the registrant has submitted
electronically and posted on its corporate Web site, if any, every Interactive
Data File required to be submitted and posted pursuant to Rule 405 of Regulation
S-T (§
232.405 of this chapter) during the preceding 12 months (or for such shorter
period that the registrant was required to submit and post such files).
Yes o No o
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405
of Regulation S-K is not contained herein, and will not be contained, to the
best of Registrant’s knowledge, in definitive proxy or information statements
incorporated by reference in Part III of this Form 10-K or any amendment to this
Form 10-K. o
Indicate
by check mark whether the Registrant is a large accelerated filer, an
accelerated filer, a non-accelerated filer or a smaller reporting company. See
the definitions of “large accelerated filer,” “accelerated filer” and “smaller
reporting company” in Rule 12b-2 of the Exchange Act. (Check one):
|
Large Accelerated
Filer o
|
Accelerated Filer
o
|
Non-Accelerated
Filer o
|
Smaller Reporting
Company x
|
|
(Do
not check if a smaller
|
|||
|
reporting
company)
|
Indicate by check mark
whether the Registrant is a shell company (as defined in Rule 12b-2 of the
Exchange Act). Yes o No x
The
aggregate market value of the common stock held by non-affiliates of the
Registrant was $17,970,578, calculated by using the number of shares outstanding
and the closing price of the common stock on June 30, 2009 (the last business
day of the Registrant’s most recently completed second fiscal
quarter).
As of
March 22, 2010 there were 20,795,126 shares outstanding of the Registrant’s
common stock, par value $0.01 per share.
DOCUMENTS
INCORPORATED BY REFERENCE
Portions
of the definitive proxy statement (the “Definitive Proxy Statement”) to be filed
no later than April 30, 2010 with the Securities and Exchange Commission
relative to the Company’s 2010 Annual Meeting of Stockholders are incorporated
by reference into Part III of this Report.
|
PART
I
|
1
|
|||
|
ITEM
1.
|
BUSINESS
|
1
|
||
|
Overview
|
1
|
|||
|
Life
Sciences
|
1
|
|||
|
Industrial BioDetection
Tests
|
2
|
|||
|
Life Sciences Products and
Services
|
3
|
|||
|
Kit
Products
|
3
|
|||
|
Food
Safety Products
|
3
|
|||
|
Agricultural
Testing
|
4
|
|||
|
Water
Quality
|
5
|
|||
|
Environmental Contamination
Detection Products
|
5
|
|||
|
Sales and Marketing
Strategy
|
6
|
|||
|
Competition
|
6
|
|||
|
Markets and
Products
|
7
|
|||
|
Geographic and Customer
Information
|
7
|
|||
|
Regulatory
Approvals
|
7
|
|||
|
Manufacturing
|
8
|
|||
|
Research and
Development
|
9
|
|||
|
Proprietary Technology and
Patents
|
9
|
|||
|
Employees
|
10
|
|||
|
Organizational
History
|
10
|
|||
|
ITEM
1A.
|
RISK
FACTORS
|
11
|
||
|
ITEM
1B.
|
UNRESOLVED
STAFF COMMENTS
|
13
|
||
|
ITEM
2.
|
PROPERTIES
|
13
|
||
|
ITEM
3.
|
LEGAL
PROCEEDINGS
|
14
|
||
|
ITEM
4.
|
RESERVED
|
14
|
||
|
PART II
|
15
|
|||
|
ITEM
5.
|
MARKET
FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER
PURCHASES OF EQUITY SECURITIES
|
15
|
||
|
Stock
Performance Graph
|
16
|
|||
|
ITEM
6.
|
SELECTED
FINANCIAL DATA
|
17
|
||
|
ITEM
7.
|
MANAGEMENT’S
DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF
OPERATIONS
|
18
|
||
|
Forward Looking
Statements
|
18
|
|||
|
Overview
|
18
|
|||
|
Results of
Operations
|
19
|
|||
|
Year ended December 31, 2009
versus year ended December 31, 2008
|
19
|
|||
|
Year ended December 31, 2008
versus year ended December 31, 2007
|
20
|
|||
|
Liquidity and Capital
Resources
|
21
|
|||
|
Off-Balance Sheet
Arrangements
|
23
|
|||
|
Contractual
Obligations
|
23
|
|||
|
Critical
Accounting Policies
|
23
|
|||
|
New
Accounting Standards and Disclosures
|
25
|
|||
|
ITEM
7A.
|
QUANTITATIVE
AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
|
26
|
||
|
ITEM
8.
|
FINANCIAL
STATEMENTS AND SUPPLEMENTARY DATA
|
26
|
||
|
ITEM
9.
|
CHANGES
IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL
DISCLOSURE
|
26
|
||
|
ITEM
9A.
|
CONTROLS
AND PROCEDURES
|
26
|
||
|
ITEM
9B.
|
OTHER
INFORMATION
|
26
|
||
|
PART
III
|
27
|
|||
|
ITEM
10.
|
DIRECTORS,
EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
|
27
|
||
|
ITEM
11.
|
EXECUTIVE
COMPENSATION
|
27
|
||
|
ITEM
12.
|
SECURITY
OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED
STOCKHOLDER MATTERS
|
27
|
||
|
ITEM
13.
|
CERTAIN
RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR
INDEPENDENCE
|
28
|
||
|
ITEM
14.
|
PRINCIPAL
ACCOUNTING FEES AND SERVICES
|
28
|
||
|
PART
IV
|
29
|
|||
|
ITEM
15.
|
EXHIBITS
AND FINANCIAL STATEMENT SCHEDULES
|
29
|
||
Item
1. Business
Overview
Strategic
Diagnostics Inc., now doing business as SDIX (“SDIX” or the “Company”), is a
biotechnology company with a core mission of developing, commercializing, and
marketing innovative, effective, and proprietary products, and solutions that
preserve and enhance the quality of human health and wellness. The
Company serves the pharmaceutical, biotechnology, diagnostics, food safety and
environmental markets.
In
February 2010, the Company announced it would be doing business as
SDIX. The SDIX name and logo were introduced to better reflect
the Company’s evolution from its historical beginnings as an industrial
diagnostics products and antibody production company to the company it is today,
with all of its core capabilities and expanded expertise in developing and
delivering a full suite of innovative immuno-solutions. The new name
also aligned the Company's name with its website address and NASDAQ listing,
making the Company easier to find and connect to.
SDIX is a customer-centric
organization. Our goals are to consistently deliver increased value to our
customers through products and services that facilitate business results, reduce
costs, and help manage risk. SDIX sales professionals focus on
delivering a quantifiable “return on investment” to their customers,
demonstrating to them how to reduce time and total costs associated with
applications for which the Company’s products are used. In addition, the Company
believes its tests and immuno-solutions provide high levels of accuracy and
reliability, delivering more actionable results to the customer compared to
alternative products.
The
Company is focused on sustaining profitable growth by leveraging its expertise
in antibodies and immuno-technologies to successfully develop proprietary
products and services that enhance the competitive advantage of our
customers.
The
Company believes that our competitive position has been enhanced through the
combination of talent, technology, and resources resulting from the business
development activities we have pursued since our inception. The Company has
achieved meaningful economies of scale for the products it offers through the
utilization of its facilities in Newark, Delaware for the manufacture of test
kits and antibodies and its facilities located in Windham, Maine for the
manufacture of antibodies.
The
Company’s Life Sciences product portfolio includes a full suite of integrated
immuno-solution capabilities including assay design, development, and
production. These capabilities combined with its proprietary Genomic
Antibody Technology™ (“GAT™”) are being used today to help discover the
mechanisms of disease, facilitate the development of new drugs, and
provide the means for rapid diagnosis.
The
Company’s Food Safety portfolio includes immunoassays which represent advanced
technology for rapid, cost-effective, easy-to-use, and accurate detection of
food pathogens. SDIX’s RapidChek® and SELECT ™ test kits are experiencing
growing adoption for the detection of pathogens such as E. coli, Salmonella and Listeria in the production, processing, and
manufacturing of food and beverages.
SDIX has
been developing antibodies which advance our customers’ immuno-based work
for 20 years. By applying its core competencies of creating
proprietary, high-quality antibodies and assay development solutions, the
Company has produced sophisticated testing and reagent systems that are
responsive to each customer’s analytical information needs.
The
Company segregates its business into two areas: Life Sciences and Kit
Products, which are described below.
Life
Sciences
SDIX
is a leading provider of a wide range of life sciences products and services,
including custom antibodies, pre-made catalog antibodies, in vitro
diagnostic-grade antibodies, proprietary critical reagent products, associated
bio-processing services, and custom assay design and development services. The
Company’s products and services are sold to, and often embedded in other
commercial products used by a wide range of customers including pharmaceutical,
biotechnology and diagnostic companies, and major biomedical research centers
both domestically and internationally. The Company is fully integrated to
deliver a wide range of services encompassing its customers’
immuno-solution needs from antigen design and antibody development through large
scale production and post production bio-processing and immunoassay design and
development. Customer service, innovation, and expertise are the
foundation of the Company’s competitive advantage. The Company’s Certified Good
Manufacturing Practices (cGMP) and ISO9001:2008 accredited facilities employ
sophisticated production processes that are reliable and deliver high quality to
its customers, and its Sandy Drive and Maine facilities are certified and
accredited by the Association for the Assessment and Accreditation of Laboratory
Animal Care (“AAALAC”), the highest standard in laboratory animal care. The
Company is licensed by the U.S. Department of Agriculture.
1
GAT™.
Innovation is a key element of the Company’s Life Science strategy for
establishing and maintaining sustainable differentiation in key markets. The
study of gene and protein functionality has created a growing demand for
antibody reagents; commercially available antibodies exist to less than 10% of
the human proteome. GAT™ was developed to address this growing need for high
quality reagents in the Life Science industry. GAT™ products and services
utilize sophisticated bioinformatics and immunization strategies to produce high
value antibody reagents and biomolecules. SDIX’s application of powerful
proprietary algorithms provides GAT™ the ability to “dial in” the precise gene
or protein sequence to produce a recombinant protein inside the host
animal that in turn elicits an immune response to the encoded protein.
This “specificity by design” approach generates antibodies that recognize
the conformational epitopes on the native protein. The ability of any antibody
to recognize a protein’s naturally folded state has the potential to expand a
biomolecule’s utility to advance platforms like flow cytometry. A
recognized advantage is the technology’s ability to produce reagents
against traditionally difficult cellular targets, such as highly conserved and
transmembrane proteins.
Immunoassay
Technology. An immunoassay is an analytical test that uses antibodies to
detect the presence of a target in a complex biological sample with high degrees
of sensitivity, precision and accuracy. Immunoassays play a central role in the
detection and quantitation of proteins associated with disease diagnosis,
prognosis and progression, and therapeutic toxicity, efficacy and outcome. The
Company’s scientists are experts in the design and development of antibodies and
immunoassays. The Company’s scientific expertise with multiple immunoassay
formats, coupled with a thorough understanding of the needs of markets and
specific customer applications, has allowed the Company to develop a diverse
array of immunoassay products. Recent activities by Company scientists have been
focused on developing multiplexed immunoassay tests employing the Company’s
genomic antibodies in the field of biomarker discovery, especially relating to
cancer research.
Industrial
BioDetection Tests
The
Company’s detection technologies allow industrial customers to rapidly and
cost-effectively identify the presence of adulterants, such as chemical toxins,
biological pathogens and other contaminants, that can compromise human or
environmental safety, and/or financially impact efficiencies of production
processes. Many of the Company’s products are in the form of single use test
devices, sample prep materials and reagents, thus creating recurring revenue
opportunities. Specific industry applications include:
|
●
|
Food
and Beverage Manufacturing: Systems for high efficiency testing for
the identification of pathogens and toxins in food, water and the
manufacturing environment.
|
|
●
|
Water
Utilities: Drinking water facilities test for chemical toxins and
pathogens. Wastewater treatment facilities manage pollution control by
monitoring efficiency maintenance in biological processing systems,
specifically testing for influent and effluent toxic chemicals and
pesticides.
|
|
●
|
Environmental
Management: On-site testing systems to increase the speed and
accuracy of environmental remediation of soil and ground water
pollutants.
|
|
●
|
Agriculture
and Agro-science: Systems for the detection, identity preservation,
and quantification testing of genetically modified organisms, and test
systems for feed and grain safety testing, including for the presence of
mycotoxins.
|
By
leveraging its expertise in immunology, proteomics, bio-luminescence and other
bio-reactive technologies with innovative application and production
capabilities, the Company is able to provide sophisticated diagnostic testing
and reagent systems to a diverse customer base serving multiple vertical
markets.
Bacteriophage
Technology. Bacteriophage, or phage, are viruses that infect bacteria.
They are highly specific for the type of bacteria that they infect and do not
infect any other living cell from any other organism including animal, plant,
fungus or yeast. Because lytic bacteriophage specifically kill their bacterial
hosts and not other living cells, purified preparations of phage have been used
medicinally to treat bacterial infections of plants and animals, including
humans. The use of bacteriophage as a human therapeutic attests to the
biological specificity and safety of these viruses. In the last two years, the
U.S. Food and Drug Administration (“FDA”) has approved the use of bacteriophage
products for direct application to ready-to-eat foods for reduction of Listeria
bacteria based on the determination that phage are “generally recognized as
safe” (“GRAS”).
The
Company is applying its bacteriophage technology in its test kit products for
the detection of bacterial food pathogens, including its Salmonella
SELECT ™ product. The Company has been awarded a U.S. patent claiming the
use of bacteriophage to control competing and cross-reacting bacteria, thereby
reducing false positive and negative results and improving analytical test
performance.
The
Company has also filed patent applications claiming the use of specific lytic
bacteriophage to control contaminating bacteria in large scale industrial
fermentation processes such as ethanol and lysine production. The Company
believes that the use of bacteriophage is a significant improvement over the use
of antibiotics and may have an impact on yield and cost associated with the
production of ethanol from feedstock.
2
Bioluminescence
Technology.
The Company’s Microtox®
and DeltaTox®
tests use a specific strain of luminescent bacteria as biosensors of toxicity,
especially in water samples. These bacteria, when exposed to certain chemicals,
undergo a chemical reaction resulting in the emission of visible light. Light
output is inversely proportional to the toxicity of the sample being
tested.
SDIX’s
solutions include the instrumentation, reagents and technology necessary to
employ testing. The Company has developed proprietary technology to analyze the
results and calculate toxicity according to industry standard and regulatory
methods. These solutions are highly reliable and offer significantly greater
precision than other commonly applied measures of toxicity employing small
numbers of living organisms (e.g., fish). The Company’s products, reagent kits,
instruments and software provide for rapid and inexpensive assessment of
toxicity in multiple applications including approved regulatory methods in
many countries worldwide.
Life
Sciences Products and Services
The
Life Sciences market is experiencing growth due to the expansion of research in
the genomic era into understanding of the role of proteins in biology and
medicine. According
to Frost & Sullivan and other market researchers, the global market for
antibody-based reagents and tools in 2008 was approximately $1.8 billion with
estimated growth in the range of 5-7%. Custom reagent development and production
account for approximately $450 million with an estimated 10-12% growth rate and
premade reagent products comprised approximately $1.4 billion with an estimated
growth rate in the 5-7% range. Customers in these markets regard the Company as
a leader in the design, development and production of critical tools used to
target, differentiate, quantify, and profile the vast number of proteins related
to human health. The Company links its historical expertise in
immunotools and immunoassays with the speed and agility of its proprietary GAT™
platform. Post-genomics drug development is a rapidly emerging sector for
proteomic immunotools. Within this market, investment has largely shifted from
discovery activities into more information-rich integrated development
activities. Within the past two years, the Company has supplied 14 of the top 20
pharmaceutical and biopharmaceutical companies with proteomic immunotools to
further their drug development programs as well as initial clinical candidates
for monoclonal antibody therapeutics. The Company produces antibodies to targets
and biomarkers of interest allowing customers to quickly assess the feasibility,
efficacy and safety of compounds in their developmental pipelines.
Protein
biomarkers as predictive, prognostic, diagnostic, and reporters of activity
throughout the drug discovery and development workflow have created increased
needs for protein identification and quantitation tools. The Company sees rapid
advances in the use of antibodies as tools to measure biomarkers. BCC Research
[‘Biomarkers: The Expanding Global Market’] projects market size for the entire
biomarker services industry to be at $13 billion by 2012. The
biomarker assay testing market has a proven record of revenue generation. The
market was $612 MM in 2007 and is estimated to have an annual growth rate of
23.5% and this estimate is based solely on assays and products that are
currently available. Currently, high-quality biomarker assays exist for less
than 500 proteins (across all species), a fraction (1-2%) of the total number of
proteins encoded by the genomes of key species (e.g., human and rodent).
Customers in these markets view the Company as a key provider of critical
antibody reagents and immunoassay design and development.
The
Company’s experience in antibody generation and immunoassay design together
with its proprietary GAT™ platform puts it in a strong position to address these
needs. In 2009, SDIX was selected for a third consecutive time through an open
Request for Proposal by Science Applications International Corporation (“SAIC”)
in cooperation with the National Cancer Institute to generate a library of
monoclonal reagents against cancer biomarkers. These antibodies will become part
of a reference set of validated tools for researchers. The Company also has a
portfolio of catalog antibodies made using its GAT™ platform available for sale
online. Within the past year, many new customers have benefitted from these
oncology-focused research reagents. In many cases, a singular product has been
selected by a client to become a critical testing reagent in long term projects,
precipitating the transition of a per-unit sale into a critical reagent supply
agreement. These antibodies are now a resource for the Company to assess
application in novel platforms, assays, and multiplex applications.
The
Company is a supplier to many major manufacturers of antibody-based diagnostic
tests. The Company maintains regulatory compliance, industrial scale and
efficiencies, and requisite quality systems to assure a secure supply of
critical reagents to its partners. The Company provides proprietary reagents as
well as large scale Original Equipment Manufacturer (OEM) production of custom
antibodies.
Kit
Products
Food
Safety Products
The
Company’s food safety product line includes enrichment media and rapid tests to
detect food pathogens, including E.
coli O157 (including H7), Listeria
and Salmonella.
The Company is a leader in tests for targeted traits in genetically engineered
plants, tests to detect Genetically Modified (GM) traits in grain, seeds, food
ingredients and food fractions and tests to detect naturally occurring fungi in
grains (mycotoxins).
3
Food
Pathogen Testing
Pathogen
specific testing is an increasingly important part of microbiology testing
performed in the global food industry. The worldwide market for pathogen tests
and media is estimated to be between $850 million and $1 billion according to
independent studies and the Company’s own market research. According to several
independent studies, the market for pathogen tests grew at a rate of 5
to 7% in 2009 and this growth rate is expected to continue for the next
several years. Growth in pathogen testing is driven primarily by regulatory
changes, customer testing trends, industry consolidation, and globalization of
the world’s food supply.
Since
2001, the Company has invested in the development and market introduction of
products for the detection of pathogenic microorganisms in food. In 2002, the
Company introduced its first test method for the pathogen E.
coli O157 (including H7). The RapidChek®
E.
coli O157:H7 test strips and proprietary media system have received
ongoing market acceptance in the United States. In 2005, the Company was
notified that its RapidChek®
E.
coli O157:H7 assay had been selected as the assay method of choice for
the National School Lunch Program for screening raw and frozen beef for the
organism. The National School Lunch Program is a federally-assisted meal program
that operates in over 97,000 public and non-profit private schools and
residential childcare institutions. The United Stated Department of Agriculture
(“USDA”) is responsible for determining that the meat produced for the National
School Lunch Program is safe. In addition, the RapidChek®
test for detection of E.
coli O157:H7 was selected by the Food Safety Inspection Service (“FSIS”),
the public health agency in the USDA, as an approved methodology for screening
of the organism in raw beef samples. The FSIS section of the USDA conducted a
rigorous evaluation of rapid methods that are currently on the market for
screening pathogens, including polymerase chain reaction, and automated/manual
immunoassays and benchmarked kit performance against the current USDA
traditional cultural method. The RapidChek®
E.
coli O157:H7 method was evaluated and determined to be the “best in
class” against the other immunological methods tested. RapidChek®
has been included in the USDA Microbiological Laboratory Guidelines as one of
only two immunoassays that are recognized for use in screening raw beef for
E.
coli O157 (including H7). The Company believes that the acceptance of its
method by the agencies regulating food safety has increased sales as producers
seek to use methods that have been evaluated by the regulatory agencies. The
RapidChek®
E.
coli O157:H7 test system has also received international recognition with
regulatory approvals in Canada (Canadian Food Inspection Agency) and Australia
(Australian Quarantine and Inspection Service). In 2009, the Company
received AOAC Research Institute Performance Tested Method certification for
improvements made to the RapidChek E.
coli O157 test method including validation of the testing of
composite 375g ground beef and beef trim sample.
In June
2004, the Company launched its test for detection of Listeria.
This test system received AOAC Research Institute (“AOAC”, “AOAC-RI”) approval
for both food and environmental samples, in contrast to several
competitive methods on the market that have AOAC approval for food samples only.
As a result of new regulations enacted by the USDA in 2003, environmental
samples account for approximately 80% of all Listeria
testing. The Listeria
test incorporates the use of a proprietary enrichment procedure that provides
results in 40 hours, which is 8-12 hours faster than most other methods on the
market. In addition, the proprietary enrichment system does not require a
transfer step, providing significant labor savings compared to other methods on
the market. As with all pathogen systems, food companies require internal
evaluations prior to adoption. In these evaluations, the Company’s Listeria
test system demonstrated superior performance and improvements in efficiency and
productivity compared to most competitive methods on the market. As a result of
improvements in performance and cost-in-use, the Company has had the Listeria
product adopted by a number of very large food processors.
In August
2006, the Company launched its new RapidChek®
SELECT™ Salmonella
test with AOAC-RI approval at the International Association of Food Protection
Meeting in Calgary, Canada. This novel test is based on a patented phage
technology combined with the Company's next generation lateral flow
technology and has revolutionized the Salmonella
testing arena. The RapidChek®
SELECT ™ test was developed to meet some of the challenges faced in Salmonella
testing, including high false positive and negative rates, which can be
particularly prevalent in high burden samples. The patent claims technology that
increases both the specificity and sensitivity of rapid pathogen tests. In
September 2006, the RapidChek®
SELECT ™ Salmonella
test was the first lateral flow test approved for the National Poultry
Improvement Plan, and will provide an attractive alternative to current methods
used such as labor intensive cultural methodologies. The RapidChek®
SELECT ™ Salmonella
test was evaluated and adopted by several of the top poultry and beef processors
in 2007. The launch and acceptability of RapidChek®
SELECT ™ in the market has also facilitated the increase in sales of the
RapidChek®
Listeria
system, as most processors prefer to utilize one platform for multiple testing
needs. Customers have cited the use of SELECT ™ contributing to improved
laboratory efficiencies and significant savings as compared to what they were
previously using to test.
Agricultural
Testing
Genetically
Modified Crops
Tests for
GM traits are generally used to determine whether the sample tested contains the
protein associated with the genetic modification. Seeds, grain or leaf tissue
are typically tested. The tests may be employed by users desiring to ensure that
seed or grain lots are either GM-free or, in other cases, that they contain a
specified amount of the GM material in order to meet certain GM requirements.
Among the commodities typically tested with the Company’s products are corn,
soybeans, rice and cotton. The Company estimates that the worldwide demand for
protein based testing of genetically modified crops is $15 million per year. To
address this market, the Company maintains a small U.S. sales force and
distribution in the five principal countries that, in addition to the United
States, are responsible for 96% of the GM crop area worldwide.
4
The
Company has developed a simple “one-step” test that is used at the point of
testing to determine if an individual plant contains the targeted genetic trait.
Commercial seed producers use these products to ensure the quality of their
products. This type of test also can be used in crops for enforcement purposes
to expose unlicensed application of the genetic technology.
Acceptance
of GM crops has increased and as the development of new traits has risen, some
countries have adopted regulations on biotech crops. In 2004, the European Union
(“EU”) adopted regulations regarding labeling and traceability of GM food and
feed with enforcement beginning in April 2004. The regulatory tolerance for
EU-authorized GM traits is 0.9%, and 0.5% for unauthorized GM traits that have
already received a favorable risk assessment from various U.S. regulatory
agencies. Traceability systems must be in place and must demonstrate that any
traces of GM traits are adventitious and are technically avoidable. The Company
no longer believes that the impact of regulations will result in stricter
testing of grain and grain exports from countries growing GM crops, or increases
in testing to meet these new regulations. Conversely, widespread acceptance of
GM crops is generally reducing the practice of grain testing as GM traits are
increasingly ubiquitous in the environment.
Water
Quality
The
Company’s water quality product line includes industrial bio-detection kits for
water and soil contaminants such as pesticides, explosives, petroleum related
products and polychlorinated biphenyls (PCBs); Microtox®
toxicity tests used in a wide array of market segments; and products for
detecting polymers and corrosion in water. In addition to use by water utilities
and related government agencies, the product line is used in many industrial
manufacturing segments, environmental remediation, research and ecological
studies. The global market for analytical testing associated with the water and
environmental industries is estimated at $1.4 billion based on a compilation of
market research studies. The overall growth rate in developed markets is
estimated at 2-3%, while the growth rate in developing markets, primarily Asia,
is estimated at 7-9%. The biggest driver for growth is government regulations
associated with water quality and environmental protection.
Toxicity
Testing
In 2001,
the Company acquired AZUR Environmental Limited to add the Microtox®
product line to its portfolio. Microtox®
is a unique rapid acute toxicity test that detects a broad range of
toxins and chemical agents. The Microtox®
brand is the global reference standard for rapid acute toxicity testing.
Microtox®
makes toxicity analysis simple and easy to perform and results can be
generated in as little as 30 minutes. The Company also markets a portable
version of the Microtox®
technology known as DeltaTox®.
Many water utilities and emergency response teams are using DeltaTox®
technology as part of their Water Fit for Use and emergency response programs.
Microtox®
has been widely accepted by the wastewater treatment industry where managing and
controlling costs by accurately assessing the mechanical, operational and
chemical performance of these facilities is critical. Microtox®
delivers value by helping to improve operating efficiency and by helping
facilities stay in compliance with their discharge permits.
In
February 2006, the Company announced that its Microtox®
bioassay technology was awarded the Designation and Certification as an
“Approved Product for Homeland Security” by the Department of Homeland Security.
In December 2007, the Company was awarded a Federal Supply Schedule GSA
contract. The contract further expands the Company’s reach into federal, state
and local agencies, in addition to making it easier for these agencies to do
business with the Company.
The
Company is currently developing its next generation DeltaTox® instrument, for
expected market introduction in the first half of 2010. This
new instrument, DeltaTox® II, will include features to further enhance business
results for customers implementing strong Water Fit for Use
programs.
Environmental
Contamination Detection Products
The
entrance of pesticides into the water supply is a result of agricultural and
residential runoff. In areas of substantial agricultural activity, drinking
water is tested for pesticides to protect supplies and to comply with federal
and state regulations. The Company’s pesticide test kits are used in situations
where field testing, or the testing of one specific pesticide gives the test kit
much greater utility than a lab-based analyzer. Users include federal agencies
such as the U.S. Geological Survey and USDA, state environmental and health
departments, water utilities and environmental engineering companies. The
Company also sells immunoassay products in the environmental market. The Company
offers three different test formats, each with performance characteristics that
make them well suited for a particular customer application. All of the
Company’s environmental test kits are capable of analyzing multiple samples in
parallel. The Company is currently marketing kits for a variety of contaminant
classes and has been able to expand its product offerings through distribution
agreements to accommodate new technologies.
5
Sales
and Marketing Strategy
The
Company markets and sells products in the life sciences, food safety,
agricultural testing and water quality product categories through a U.S. direct
sales force, Internet presence and a network of over 50 distributors in Canada,
Mexico, Latin America, Europe and Asia and through the Company’s corporate
partners. The Company also has a European office and sales operation near
London, England. The Company evaluates various sales and service models that can
contribute to the profitable growth of business. Identifying the most effective
channels to market will allow the Company to better allocate resources to both
new and existing growth opportunities.
In the
United States, the primary sales channel is through a direct sales force
comprised of geographically based field sales professionals, key segment
managers, and inside sales associates. The sales force is augmented by customer
service and project management organizations, and applied technical marketing
specialists which assure that all elements of the customer’s buying experience
meet and exceed their performance expectations.
On the
basis of its strengthening market position, the Company continues to develop
channels to market and accelerate predictability and sustainability of revenues.
The Company is investing in its direct sales force through the addition of new
sales representatives and focused sales and technical training. The Company
continually measures sales performance and maintains discipline in the balance
between the addition of new sales resources and ongoing efforts to continually
improve sales efficiency and effectiveness of existing resources.
The
Company is also focusing on its network of quality channel partners. In 2006,
the Company added its first distributor for its custom antibody offering. The
Company is working to add additional channel partners for both its custom and
catalog offerings nationally and internationally.
In 2005,
the Company signed an exclusive distribution agreement with DuPont Qualicon for
the representation of the Company’s immunoassays for food pathogen detection. In
2007, the Company moved to a non-exclusive distribution agreement with DuPont
Qualicon and expanded the agreement in 2009 to include RapidChek®
SELECT products. In 2007, SDIX began to add new distribution partners
for this product line. The Company expanded its international distribution
network for food safety pathogen products, adding and training a total of 15
independent distributors to sell the RapidChek®
product line in high growth markets globally, including Southeast Asia, Europe
and Latin America. The Company also took a much more aggressive role in
marketing these methods. It is anticipated that the additional distributors and
international expansions of promotion/sales of the products will increase
revenues as they gain acceptability.
Competition
Many of
the Company’s potential competitors are large companies with substantially
greater financial and other resources than the Company. To the extent that any
such companies enter into one or more of the Company’s markets, the Company’s
operations could be materially adversely affected. The Company anticipates
increased competition as potential competitors perceive that the Company’s
products have become commercially proven, or if the Company cannot maintain
competitive differentiation.
In the
Ag/GMO market, the Company competes with several small, privately held companies
(Agdia, Envirologix) that market very similar, if not identical
products.
In food
pathogen testing, the Company is among the more recent entrants to the market
and faces a broad base of competition. The worldwide market for pathogen tests
is estimated to be between $850 million and $1 billion annually and as such has
drawn many competitive products. The Company’s RapidChek®
E.
coli O157:H7, Salmonella
and Listeria
tests compete globally with numerous competitive rapid testing systems.
Instrument-based tests are offered by bioMerieux SA and DuPont Qualicon among
others. Competitive strip based tests are offered by Neogen Corp., BioControl
Systems, Inc. and others. In addition, traditional lab culture methods offer
indirect competition. The Company hopes to gain market share from competitive
methods and with new users due to key product advantages such as speed of
result, ease-of-use, accuracy and by providing overall cost
savings.
The
Company believes there are no meaningful direct competitors for the Company’s
Microtox®
product line in the United States. In Europe and other parts of the world, the
Company primarily competes against Checklight, Ltd., an Israeli-based company,
and one other instrument-based test method produced by Hach Lange, an affiliate
of The Danaher Corporation. The Company believes its products have a number of
competitive advantages including the comprehensive screening for general
toxicity and competes effectively on superior features and
functions.
6
With
respect to the environmental contaminant test products, the Company currently
receives the greatest competition from fixed site environmental laboratories and
several small privately held companies. Traditional analytical methods for
environmental contamination are often utilized for confirmation and closure of
environmental sites. The Company believes it has detection products which are
user friendly and provide greater value in use than competitive
offerings.
In the
antibody product category, the competitive landscape is rapidly changing as the
Company continues to shift its emphasis to earlier activities in drug and
biomarker discovery. The Company will increasingly compete with technology
companies that offer products and services for the discovery and advancement of
novel antibodies. The Company believes that its proprietary GAT™ platform
coupled with its expertise in assay development provides differentiated access
to the high value application markets it is targeting.
The
Company also competes in its traditional antibody markets with the internal
capabilities of some of the Company’s large pharmaceutical, research and
diagnostics customers. These customers often have significantly greater revenues
than the Company. Generally these customers produce some products internally and
purchase similar products from the Company.
Competitors
in the market as third party providers of custom, large scale antibody reagent
production include Covance (public), Harlan (private), Lampire (private) and
Scantibodies (private). Additionally, there are a number of smaller companies
that offer competing products. In the custom research reagent market, the
Company has identified 49 companies offering some form of traditional antibody
production from customer-provided antigens. The Company believes that its
innovation, expertise, and fully integrated suite of immune-solutions plus the
scale of its operations are significant competitive advantages against both
large and small competitors. In the catalog antibody space, there are over 130
companies competing for this $1.35 billion market.
Markets
and Products
The
Company sells products in the life sciences, food safety, agricultural testing
and water quality market categories through its U.S. direct sales force, a
network of over 50 distributors in Canada, Mexico, Latin America, Europe and
Asia and the Company’s corporate partners.
Geographic
and Customer Information
The
following table sets forth sales by geographic region:
|
Year
|
||||||||||||
|
Ended
December 31,
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
United
States
|
$ | 19,739 | $ | 20,744 | $ | 21,154 | ||||||
|
Rest
of the world
|
7,415 | 6,915 | 6,053 | |||||||||
|
Total
|
$ | 27,154 | $ | 27,659 | $ | 27,207 | ||||||
The
Company’s basis for identifying sales by country is the ship-to location. There
were no individual countries outside of the United States that represented more
than 10% of the total revenues of the Company. There are no significant
long-lived assets located outside the United States.
No
single customer accounted for 10% or more of the Company’s revenues in
2009, 2008 or 2007.
Regulatory
Approvals
The
Company is engaged in the development of antibody and immunoassay products for
use in the medical and human healthcare fields. Its current products in this
market are intended for “research use only.” The Company also manufactures tests
for bacterial food pathogens, mycotoxins, genetically engineered traits in
plants and water treatment polymers, which are currently unregulated. However,
agencies such as the Environmental Protection Agency (“EPA”), the FDA, and the
FSIS are engaged in testing and, together with organizations like the AOAC,
maintain compilations of official methods for use in testing in certain market
segments. Some of these organizations also issue procedures and guidelines for
validating new methods. Although not required, official methods adopted by these
agencies sometimes have the commercial impact of regulations because the
industry and the Company’s customers tend to follow the practices of regulatory
agencies.
The
Company maintains compliance to 21 CFR 820 cGMP, and International
Organization for Standardization (“ISO”) 9001:2008
certification for all three of its facilities from an ANSI-ASQ National
Accreditation Board (“ANAB”) Accredited International Registrar for the ISO 9001
standards. Recognized and respected worldwide, the ISO 9001:2008 standards are
put forth by the ISO. This certification demonstrates the Company’s
commitment to excellence in product and service quality, and a continued focus
on improving the customer experience.
7
The
Company has maintained AAALAC (Association for the Assessment and Accreditation
of Laboratory Animal Care) accreditation at its Delaware facility since 1992 and
at its Maine facility since 2000. The Company volunteers to participate in the
AAALAC program in addition to complying with the local, state and federal laws
that regulate animal research. In order to maintain these accreditations, the
Company undergoes regular inspections and reviews. The Company also holds
approvals from the USDA, OLAW (Office of Laboratory Animal Welfare), and the
NIH, further validating the stewardship of the Company in proper laboratory
animal care.
The
Company believes that the validation and acceptance of its environmental
products by regulatory agencies plays a significant role in market acceptance.
EPA SW-846 is the compendium of test methods published by the EPA’s Office of
Solid Waste listing those analytical methods that have been validated by the EPA
for a stated purpose. The vast majority of the Company’s analytical methods for
environmental soil sample analysis are listed in EPA SW-846. Many federal, state
and local environmental programs often refer to and rely on EPA SW-846 methods
for purposes of remediation and monitoring.
The
legislation and regulations that the Company believes are most applicable to its
environmental business are the Research Conservation and Recovery Act (“RCRA”),
Comprehensive Environmental Response, Compensation and Liability Act (“CERCLA”),
Toxic Substances Control Act (“TSCA”), Federal Insecticide, Fungicide and
Rodenticide Act (“FIFRA”) and the Pure Food and Drug Act. For analysis of water
and wastewater, the Safe Drinking Water Act, the Clean Water Act and the
National Pollution Discharge Elimination System (“NPDES”) permitting program
acceptance under the Clean Water Act also will be significant to the Company’s
business. As the utility of the Company’s Microtox®
products continues to be widely recognized in drinking water security
applications, regulations and mandates associated with Homeland Security
programs may also have an impact on the Company’s business. Collectively, these
programs regulate the management, disposal and clean-up of hazardous substances
and protect the nation’s ground and surface water and drinking water
supplies.
Manufacturing
The
Company manufactures test kits for the detection of a wide array of analytes in
five immunoassay formats and one bioluminescence format. The five formats are:
one step lateral flow tests; coated tubes; latex particles; magnetic particles;
and micro-titer plates. The Company manufactures a biological supplement that
enhances the detection of certain analytes and improves overall performance of
certain assay formats. In addition to test kits, the Company supplies ancillary
equipment and supplies including test evaluation instruments, reagents, sample
media, spectrophotometers, pipettes, balances and timers.
The key
critical reagent manufacturing technologies are conjugation chemistries,
antibody formulations, calibrator preparation, lateral flow strip production,
microbiological and immunoassay processes. Reagent production processes include
filling and dispensing liquids, subcomponent and finished goods assembly,
in-process testing, quality control, packaging and shipping. The critical
reagents and production assembly groups produce test kits in the Newark,
Delaware laboratories. Biological materials are primarily developed and produced
in-house; however, some reagents are licensed from third parties or purchased
from commercial sources. A crucial step in the Company’s manufacturing process
is the stabilization of the immunoreagents utilizing proprietary lyophilization
techniques. In general, raw materials used by the Company in its products are
obtainable from multiple sources. The Company purchases instruments and
ancillary equipment from outside vendors. A number of the instruments sold by
the Company were developed to be used exclusively with the Company’s products
and are subject to specific supply agreements. The Company believes that the raw
materials, instruments and equipment used in the manufacture of its products are
sufficiently available for the Company’s current and foreseeable manufacturing
needs.
The
Company manufactures its products in accordance with the FDA’s Good
Manufacturing Practices guidelines and has implemented data-driven problem
solving, measurement and statistical process controls to troubleshoot and
continuously improve quality and output performance. Capital investment and
equipment automation have reduced key parameter variation, improved production
efficiencies and lowered manufacturing costs. The Company utilizes planning
tools to control all elements of the supply chain and manufacturing processes,
including raw material procurement, inventory management, capacity planning and
production scheduling, work-in-process tracking, order processing and
fulfillment, shipping and customer invoicing. The Company believes the existing
facilities and equipment are sufficient to support a significantly larger
production demand.
The
Company also supplies a wide array of custom antibody products and services to
the in-vitro diagnostic, academic,
pharmaceutical and medical research industries. Antibodies are developed and
produced using animals or cell culture methods. Laboratories are maintained to
prepare immunogens, perform chemical conjugations, purify antibodies, and
perform a range of quality control procedures. The cell culture laboratories
support the development of hybridomas and manufacture of monoclonal antibodies.
The cell culture laboratories also provide services to enhance the productivity
of cell lines, establish Master Cell Banks, and store cell lines in secure
fail-safe cryogenic systems. In 2009, the Company consolidated the operations
performed in the Dallas facility into the Sandy Drive technology center in
Newark, Delaware, resulting in overhead cost savings and improved technical
collaboration. Animal facilities house specific-pathogen-free animals that are
tested routinely to assure they are maintained under the highest health
standards. Current capacity utilization in antibody production is approximately
70%, and there is additional land and zoning clearance on the 64-acre site in
Windham, Maine to double polyclonal operations.
8
Research
and Development
The
Company engages in substantial research and development activities (R&D)
involving development of products, services and technology platforms for its two
primary markets, Life Sciences and Food Safety. In the years ended December
31, 2009, 2008 and 2007, the Company incurred approximately $2.9 million, $3.6
million, and $2.9 million, respectively, in research and development
expenditures. Research and development on the Company’s proprietary GAT™ product
offering, and food safety products accounted for 71% of the total R&D
effort for the year ended December 31, 2009. Also in
2009, the Company invested 12% of its R&D effort in the development of its
new instrument for the detection of toxins in water in support of its Microtox™
product line and scheduled to launch in the second quarter of
2010.
The
Company’s primary laboratory facilities located in Newark, Delaware were
designed and built specifically for conducting research and development relating
to antibody and immunoassay technology. These facilities include state-of-the
art, cGMP antibody development and large-scale production facilities. The
Company has assembled a scientific staff with extensive experience in the
development, production and purification of monoclonal and polyclonal
antibodies. The Company also has extensive expertise in the development and
production of reagents from the antibodies it produces, as well as commercial
immunoassays employing those reagents.
In
2009, the Company continued development of its proprietary GAT™ platform,
focusing on advanced methods for development of antibodies to high value
proteins that are the targets of pharmaceutical and biotechnology companies.
Consistent with the vision of applying its GAT™ to the field of
proteomics and biomarker discovery, the Company is testing multiplex
immunoassays employing the novel antibodies developed using the GAT™ platform.
In addition, in 2009 the Company entered into an agreement with the Fred
Hutchinson Cancer Research Center (the “Hutchinson Center”) to collaborate to
discover biomarkers for use in the early detection of a variety of
cancers. Initial studies will center on pancreatic
cancer. The Company has provided approximately 1,300 antibodies
for the collaborative studies from its unique collection of cancer antibodies
and
the Hutchinson Center will test the antibodies, with a unique set of
patient samples collected prior to disease diagnosis.
In the
food safety market, the Company completed the development of and
commercially launched its new RapidChek® E. coli O157 (including H7) test, and
earned Performance-Tested Methods™ certification from the AOAC Research
Institute for testing composite samples of raw beef, including ground beef and
boneless beef trim. This new
test is the first product to be validated for analysis of the increased sample
size being implemented across the beef processing industry. Also in
the food safety market, the Company received a Certificate of Validation for its
RapidChek®
SELECT™ Salmonella
system from the AOAC Research Institute.
The
Company is currently developing its next generation DeltaTox® instrument,
for expected market introduction in the first half of 2010. This new
instrument, DeltaTox® II, will
include features to further exhance business results for customers implementing
strong water fit for use programs.
In 2009,
the Company was awarded a U.S. patent for the use of its proprietary
bacteriophage technology in microbiological assay tests and
processes. This technology is a rapid bacterial detection method that
reduces or eliminates the growth of undesirable bacteria, resulting in improved
test performance. The use of bacteriophage to improve the specificity
and sensitivity of testing methods is unique to the Company. Also in
2009, the Company received a U.S. patent for a novel method of detecting water
treatment polymers using raman spectroscopy.
The
Company’s research and development personnel are experts in many advanced
research disciplines in life sciences including immunology, immunochemistry,
molecular biology, protein chemistry, biochemistry, microbiology and synthetic
organic chemistry. In addition to the technical expertise resident within the
research and development organization, the Company’s technical manufacturing
organization is expert in large-scale cGMP production, bioprocessing,
purification and quality control of antibodies and reagents. The Company’s core
expertise is in antibody and immunoassay development and it is a major developer
and producer of monoclonal antibodies.
Research
and development activities are focused on developing proprietary technology and
products to expand the Company’s differentiated market position in Life Science
and food safety markets. The Company is a recognized leader in the field of
contract antibody and assay development services primarily for large
pharmaceutical, biotech, diagnostic and chemical companies, and the development
of rapid test kits in the food, water quality and agricultural sectors based on
immunoassay technology. In addition, the Company has extensive expertise,
facilities and equipment relating to the development and manufacture of one-step
lateral flow tests.
The
Company’s research and development organization consists of 15 individuals, 11
of whom hold advanced academic degrees. In addition, approximately one-third of
the Company’s employees are involved in technical job functions.
Proprietary
Technology and Patents
The
Company’s products are based on the use of proprietary reagents, technology and
test systems developed by Company scientists or acquired externally.
Accordingly, the Company has implemented a number of procedures to safeguard the
proprietary nature of its technology. The Company requires its employees and
consultants to execute confidentiality agreements upon the commencement of an
employment or consulting relationship with the Company and all employees are
required to assign to the Company all rights to any inventions made during their
employment or relating to the Company’s activities. Additionally, the Company
seeks to protect its technology and processes through the patent process. The
Company currently holds 24 issued U.S. patents, as well as one U.S.
patent licensed for exclusive use by the Company.
9
There can
be no assurance that the Company’s patent applications will result in the
issuance of any patent or that any patents issued to the Company would provide
protection that is sufficiently broad to protect the Company’s technology and
products. In addition, the Company cannot be certain that it was the first
creator of inventions covered by pending patent applications or that it was the
first to file patent applications for such inventions. In addition to seeking
patent protection for the Company’s proprietary information, the Company also
relies upon trade secrets, know-how and continuing technical innovation to
maintain competitiveness for its products and services. The Company has
developed a number of proprietary technologies which it has chosen not to
patent, including immunization protocols, DNA and plasmid constructs,
stabilization systems for reagents, chemical syntheses, and strategies relating
to antibody development.
|
U.S.
Patent
|
Title
|
|
|
5,426,035
|
Method
for compensating toxicity test data for the measured toxicity of a
reference sample
|
|
|
5,449,611
|
Polyaromatic
hydrocarbon (PAH) immunoassay method, its components and a kit for use in
performing the same
|
|
|
5,541,079
|
Monoclonal
and polyclonal antibodies and test method for determination of
organophosphates (license)
|
|
|
5,547,877
|
Methods
for the rapid detection of toxic halogenated hydrocarbons and kits useful
in performing the same
|
|
|
5,593,850
|
Monitoring
of industrial water quality using monoclonal antibodies to
polymers
|
|
|
5,618,681
|
Polyaromatic
hydrocarbon (PAH) immunoassay method, its components and a kit for use in
performing the same
|
|
|
5,780,250
|
Immunoassay
standards for polyaromatic hydrocarbon detection
|
|
|
5,834,222
|
Polychlorinated
Biphenyls (PCB) immunoassay method
|
|
|
5,858,692
|
PCB
immunoassay
|
|
|
5,874,216
|
Indirect
label assay device for detecting small molecules and method of use
thereof
|
|
|
5,891,657
|
Immunoassay
standards for volatile analytes with benzene rings
|
|
|
5,919,645
|
Method
for the direct determination of the toxicity of particulate
solids
|
|
|
6,096,563
|
Dual
particle immunoassay method & kit
|
|
|
6,146,903
|
Method
for determination of water treatment polymers
|
|
|
6,376,195
|
Indirect
label assay device for detecting small molecules and method of use
thereof
|
|
|
6,420,530
|
Determination
method
|
|
|
6,524,810
|
Method
of making bioluminescent assay reagent based on non-viable E.
coli
|
|
|
6,663,833
|
Integrated
Assay Device and Methods of Production and Use
|
|
|
6,750,328
|
Antibodies
for detection of water treatment polymers
|
|
|
6,911,534
|
Method
for determination of water treatment polymers
|
|
|
7,189,520
|
Compositions
and methods for detecting animal byproduct in feed
|
|
|
7,214,505
|
Cell-based
assay for the detection of toxic analytes
|
|
|
7,241,626
|
Isolation
and confirmation of analytes from test devices
|
|
|
7,521,201
|
Bacteriophages
as Selective Agents
|
|
|
7,532,321
|
Compositions
and methods for the detection of water treatment
polymers
|
Employees
As of
December 31, 2009, the Company employed 150 full time and four part time
employees. The workforce was supplemented by six agency-provided contractors.
All of the Company’s employees have executed agreements with the Company
agreeing not to disclose the Company’s proprietary information and assigning to
the Company all rights to inventions made during their employment. Key personnel
have signed agreements prohibiting them from competing with the Company. None of
the Company’s employees are covered by collective bargaining agreements. The
Company believes that its relations with its employees are good.
Organizational
History
Strategic Diagnostics Inc.
is a Delaware corporation formed in 1990.
10
Item
1A. Risk Factors
If
we do not produce future taxable income, our ability to realize the benefits of
our net operating loss carryforwards could be significantly
reduced.
As
of December 31, 2009, the Company had U.S. federal net operating loss
carryforwards, including those acquired in the Company’s past acquisitions, of
approximately $17.4 million, which, if not utilized, begin to expire as
follows:
|
Year
|
Net
Operating
Loss
(in
thousands)
|
|||
|
2010
|
$ | 4,536 | ||
|
2017
|
760 | |||
|
2018
|
1,327 | |||
|
2019
|
550 | |||
|
2020
|
66 | |||
|
2021
|
56 | |||
|
2022
|
2,268 | |||
|
2024
|
2,033 | |||
|
2025
|
3 | |||
|
2026
|
1 | |||
|
2027
|
1 | |||
|
2028
|
3,492 | |||
|
2029
|
2,287 | |||
|
Total
|
$ | 17,380 | ||
The
Tax Reform Act of 1986 (the “Act”) limits the annual use of net operating loss
and income tax credit carryforwards (after certain ownership changes, as defined
by the Act). The application of these limits could significantly restrict our
ability to utilize carryforwards. Certain of our total net operating loss
carryforwards from 2001 and prior years are subject to limitations on their
annual use since a cumulative change in ownership of more than 50% has occurred
within a three-year period with respect to those net operating loss
carryforwards. The Company is currently evaluating recent changes in ownership.
If it is determined that an ownership change of more than 50% within a
three-year period did occur, as determined pursuant to the Internal Revenue Code
and Regulations, substantially all of the net operating loss carryforwards and
income tax credit carryforwards could be subject to annual limitations on usage.
Because U.S. tax laws limit the time period during which these carryforwards may
be applied against future taxable income, we may not be able to take full
advantage of these attributes for federal and state income tax purposes due to
the annual limitation usage.
Based
on the best information available to us today, we may not have sufficient future
taxable income to utilize the net operating loss carryforwards and income tax
credit carryforwards prior to their expiration, and we have established a full
valuation allowance against these net operating loss and income tax credit
carryforwards for financial reporting purposes.
Our
results of operations may fluctuate, which could cause volatility in our stock
price.
Our
results of operations may fluctuate significantly in the future as a result of a
number of factors, many of which are outside of our control. These factors
include, but are not limited to:
|
●
|
unanticipated
events associated with regulatory changes;
|
|
●
|
general
economic conditions;
|
|
●
|
acceptance
of our products;
|
|
●
|
the
success of products competitive with ours;
|
|
●
|
expenses
associated with development and protection of intellectual property
matters;
|
|
●
|
establishing
or maintaining commercial scale manufacturing
capabilities;
|
|
●
|
the
timing of expenses related to commercialization of new
products;
|
|
●
|
seasonality;
and
|
|
●
|
the
timing and success in building our distribution
channels.
|
11
The
results of our operations may fluctuate significantly from quarter to quarter
and may not meet expectations of securities analysts and investors. This may
cause our stock price to be volatile.
If
we use hazardous materials in a manner that causes injury or violates laws, we
may be liable for damages.
Our
research and development activities involve the controlled use of potentially
harmful biological materials as well as hazardous materials, chemicals and
various radioactive compounds. We use radioactivity in conducting biological
assays and we use solvents that could be flammable in conducting our research
and development activities. We cannot completely eliminate the risk of
accidental contamination or injury from the use, storage, handling or disposal
of these materials. We do not maintain a separate insurance policy for these
types of risks. In the event of contamination or injury, we could be held liable
for damages that result, and any liability could exceed our resources. We are
subject to federal, state and local laws and regulations governing the use,
storage, handling and disposal of these materials and specified waste products.
The cost of compliance with these laws and regulations could be
significant.
Our
antibody production process utilizes various species of animals that could
contract disease or die, interrupting business operations.
Our
antibody production process utilizes animals to produce antibodies. We cannot
completely eliminate the risks of animals contracting disease or a disaster that
could cause death to valuable production animals. Disease or death on a broad
scale could interrupt business operations as animals are a key part of the
antibody production operation.
The
difficulties of operating in international markets may harm sales of our
products.
Customers
outside of the United States accounted for 27% and 25% of our revenues for the
years ended December 31, 2009 and 2008, respectively. The majority of our sales
transactions are in U.S. dollars, however, we received payments in British
pounds sterling for approximately $3.0 million in sales from our foreign
subsidiary.
The
international nature of our business subjects us and our representatives, agents
and distributors to the laws and regulations of the jurisdictions in which they
operate, and in which our products are sold. The types of risks that we face in
international operations include, but are not limited to:
|
●
|
the
imposition of governmental controls;
|
|
●
|
logistical
difficulties in managing international operations; and
|
|
●
|
fluctuations
in foreign currency exchange rates.
|
Our
international sales and operations may be limited or disrupted if we cannot
successfully meet the challenges of operating internationally.
Future
acquisitions and business combinations that we consummate may be difficult to
integrate, disrupt our business, dilute stockholder value or divert management
attention.
From time to time, we have considered
and may in the future consider expanding our operations and market presence by
making acquisitions and entering into business combinations, investments, joint
ventures or other strategic alliances with other companies. We may have to issue
debt or equity securities to pay for future acquisitions, which could be
dilutive to our then current stockholders. We cannot assure you that we will
consummate any transactions in the future. However, these transactions create
risks, such as:
|
●
|
difficulty
assimilating the operations, technology and personnel of the combined
companies;
|
|
●
|
disrupting
our ongoing business;
|
|
●
|
problems
retaining key technical and managerial personnel;
|
|
●
|
additional
operating losses and expenses of acquired businesses;
and
|
|
●
|
impairment
of relationships with existing employees, customers and business
partners.
|
Any of the events described in the
foregoing paragraph could have an adverse effect on our business, financial
condition and results of operations and could cause our stock price to
decline.
The
leases for our headquarters and a property where we conduct manufacturing and
research will expire no later than December 31, 2011. If we are
unable to negotiate new leases for our existing space, or find replacement
space, in either case on terms that are attractive to us, or if we are required
to make substantial capital improvements in either the existing or new space,
our results of operations could be harmed.
Our
headquarters and 25,000 square feet of manufacturing and research space that we
lease in Newark, Delaware are leased under operating leases that will expire no
later than December 2011. We are currently considering our options
with respect to renewal or replacement of this space. We may be
unable to negotiate new leases for our existing space, or to find replacement
space, in either case on terms that are attractive to us. In
addition, should we elect to attempt to negotiate new leases in our current
facilities, we may have to make investments in capital improvements to enhance
the space. In any such case, our results of operations could be
harmed.
12
Certain
of our shareholders are able to significantly influence proposals for a change
in control or other matters requiring a shareholder vote.
Directly,
or through entities that they control, members of our Board of Directors as of
December 31, 2009 controlled approximately 29% of our common stock. Through
entities that he controls, Steven R. Becker, who joined our Board effective
March 12, 2008, controlled approximately 11% of our outstanding common stock as
of December 31, 2009. Due to this concentration of ownership, members of our
Board, acting together or, in some cases, individually, can substantially
influence all matters requiring a stockholder vote, including, without
limitation:
|
●
|
the
election of directors;
|
|
●
|
the
amendment of our organizational documents; or
|
|
●
|
the
approval of a merger, sale of assets, or other major corporate
transaction.
|
Provisions
in our organizational documents could prevent or frustrate attempts by
stockholders to replace our current management.
Our
certificate of incorporation and our bylaws contain provisions that could make
it more difficult for a third party to acquire us without the consent of our
Board. Our certificate of incorporation provides for a staggered board and
removal of directors only for cause. Accordingly, stockholders may elect only a
portion of our board at any annual meeting, which may have the effect of
delaying or preventing changes in management. In addition, under our certificate
of incorporation, our Board of Directors may issue additional shares of
preferred stock and determine the terms of those shares of stock without any
further action by our stockholders. Our issuance of additional preferred stock
could make it more difficult for a third party to acquire a majority of our
outstanding voting stock and thereby effect a change in the composition of our
Board of Directors. Our bylaws require advance notice of stockholder proposals
and director nominations and permit only our President or a majority of the
Board of Directors to call a special stockholder meeting. These provisions may
have the effect of preventing or hindering attempts by our stockholders to
replace our current management. In addition, our certificate of incorporation
contains provisions that limit our ability to engage in a business combination
with any holder of 15% or more of our capital stock unless, among other
possibilities, the Board of Directors approves the transaction. These provisions
may have the effect of preventing or hindering a change of control of our
company.
Our
stock has generally had low trading volume, and its public trading price has
been volatile.
During
the year ended December 31, 2009, the price of our common stock fluctuated
between $0.85 and $2.25 per share, with an average daily trading volume for the
year of approximately 17,000 shares. The market may experience significant price
and volume fluctuations that are often unrelated to the operating performance of
individual companies.
Our
common stock may be delisted from the NASDAQ Global Market, which could
negatively impact the price of our common stock and our ability to access the
capital markets.
Our
common stock is listed on the NASDAQ Global Market. The delisting of our common
stock would significantly affect the ability of investors to trade our
securities and would significantly negatively affect the value and liquidity of
our common stock. In addition, the delisting of our common stock could
materially adversely affect our ability to raise capital on terms acceptable to
us or at all. Delisting from the NASDAQ Global Market could also have other
negative results, including the potential loss of confidence by our suppliers,
customers and employees, the loss of institutional investor interest, and fewer
business development opportunities.
Item
1B. Unresolved Staff Comments
None.
Item
2. Properties
The
Company is headquartered in Newark, Delaware, and occupies approximately 28,000
square feet of space under an operating lease expiring in June 2011. The Company
also leases approximately 25,000 square feet of manufacturing and research space
in Newark, Delaware, under an operating lease expiring in December 2011. The
Company owns and occupies approximately 75,000 square feet of manufacturing,
research and animal facility space and approximately 88 acres of farmland in
Windham, Maine. The Company leases regional sales offices near London,
England expiring in June 2011. The Company believes that its equipment and
facilities are adequate for its present purposes.
The
Company’s inactive subsidiary, AZUR Environmental Limited, is the lessee for two
real property leases located in the United Kingdom. In 2001, the landlord of the
two properties gave AZUR Environmental Limited its consent to allow AZUR to
assign the lease and its related obligations to a third party. As inducement to
the landlord to grant the assignment, AZUR was required to guarantee performance
under the original lease terms if the third party fails to perform. Both lease
terms expire in November 2016 and provide for annual principal rent payments of
approximately $300,000 in the aggregate. The Company believes that based on its
assessment of the current financial strength of the third party, no liability is
required to be recorded with regard to the guarantee or lease
obligation.
13
Item
3. Legal Proceedings
The
Company is not a party to any material legal proceedings.
Item
4. Reserved
14
PART
II
Item
5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer
Purchases of Equity Securities
The
Company’s common stock is traded on The NASDAQ Global Market under the symbol
“SDIX.” Set forth below are the quarterly high and low bid prices for the shares
of common stock of the Company as reported by The NASDAQ Global Market without
retail mark-up, mark-down or commission and may not necessarily represent actual
transactions:
|
Common
Stock
|
||||||||
|
Price
Range
|
||||||||
|
Fiscal
Year Ended
|
High
|
Low
|
||||||
|
December
31, 2009:
|
||||||||
|
Fourth
Quarter
|
$ | 2.02 | $ | 1.00 | ||||
|
Third
Quarter
|
2.19 | 0.88 | ||||||
|
Second
Quarter
|
1.67 | 0.79 | ||||||
|
First
Quarter
|
1.36 | 0.69 | ||||||
|
December
31, 2008:
|
||||||||
|
Fourth
Quarter
|
$ | 2.45 | $ | 0.71 | ||||
|
Third
Quarter
|
3.70 | 1.62 | ||||||
|
Second
Quarter
|
4.26 | 3.26 | ||||||
|
First
Quarter
|
5.42 | 3.48 | ||||||
On March
22, 2010, there were approximately 26,000 holders (354 holders of record) of the
common stock of the Company. The Company has never paid any cash dividends on
its common stock.
15
Stock
Performance Graph
The
following line graph compares for the fiscal years ended December 31, 2004
through 2009 (i) the yearly cumulative total shareholder return on the common
stock with (ii) the cumulative total return of the NASDAQ Composite Index and
with (iii) a Peer Group Index consisting of NASDAQ Medical Equipment
Stocks.
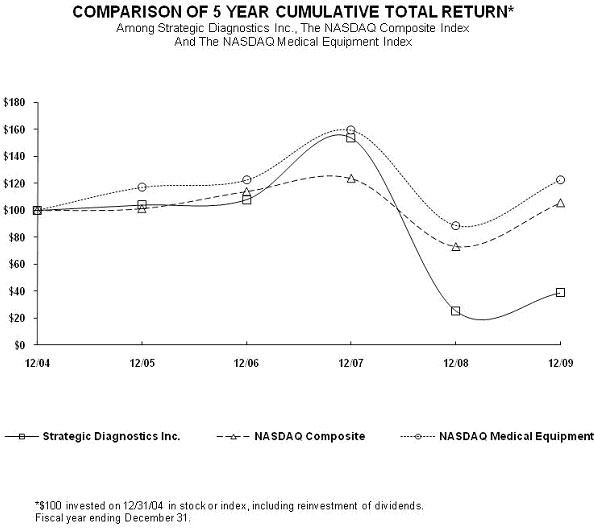
COMPARISON
OF CUMULATIVE TOTAL RETURN
Strategic
Diagnostics Inc., NASDAQ Composite Index and NASDAQ Medical Equipment Peer Group
Index
|
12/04
|
12/05
|
12/06
|
12/07
|
12/08
|
12/09
|
|||||||||||||||||||
|
Strategic
Diagnostics Inc.
|
100.00
|
104.00
|
108.00
|
153.71
|
25.43
|
38.86
|
||||||||||||||||||
|
NASDAQ
Composite
|
100.00
|
101.33
|
114.01
|
123.71
|
73.11
|
105.61
|
||||||||||||||||||
|
NASDAQ
Medical Equipment
|
100.00
|
117.06
|
122.50
|
159.63
|
88.67
|
122.59
|
||||||||||||||||||
16
Item
6. Selected Financial Data
|
Year
Ended December 31,
|
||||||||||||||||||||
|
2009
|
2008
|
2007
|
2006
|
2005
|
||||||||||||||||
|
(in
thousands, except share and per share data)
|
||||||||||||||||||||
|
Revenues
|
$ | 27,154 | $ | 27,659 | $ | 27,207 | $ | 25,522 | $ | 24,845 | ||||||||||
|
Operating
expenses:
|
||||||||||||||||||||
| Manufacturing | 12,416 | 13,091 | 10,766 | 11,715 | 11,416 | |||||||||||||||
| Research and development | 2,894 | 3,576 | 2,938 | 2,630 | 3,034 | |||||||||||||||
| Selling, general and administrative | 13,593 | 14,425 | 11,990 | 10,555 | 9,722 | |||||||||||||||
| Goodwill impairment | - | 4,150 | - | - | - | |||||||||||||||
| (Gain) loss on disposal of assets | (1 | ) | (17 | ) | 110 | 42 | - | |||||||||||||
| Total operating expenses | 28,902 | 35,225 | 25,804 | 24,942 | 24,172 | |||||||||||||||
|
Operating
income (loss)
|
(1,748 | ) | (7,566 | ) | 1,403 | 580 | 673 | |||||||||||||
|
Interest
income (expense), net
|
(15 | ) | 157 | 433 | 386 | 207 | ||||||||||||||
|
Income
(loss) before taxes
|
(1,763 | ) | (7,409 | ) | 1,836 | 966 | 880 | |||||||||||||
|
Income
tax expense (benefit)
|
(112 | ) | 8,386 | 976 | 282 | 296 | ||||||||||||||
|
Net
income (loss)
|
$ | (1,651 | ) | $ | (15,795 | ) | $ | 860 | $ | 684 | $ | 584 | ||||||||
|
Basic
net income (loss) per share
|
$ | (0.08 | ) | $ | (0.78 | ) | $ | 0.04 | $ | 0.03 | $ | 0.03 | ||||||||
|
Shares
used in computing basic net income (loss)
|
||||||||||||||||||||
|
per
share
|
20,113,659 | 20,312,707 | 20,325,285 | 20,031,833 | 19,741,223 | |||||||||||||||
|
Diluted
net income (loss) per share
|
$ | (0.08 | ) | $ | (0.78 | ) | $ | 0.04 | $ | 0.03 | $ | 0.03 | ||||||||
|
Shares
used in computing diluted net income (loss)
|
||||||||||||||||||||
|
per
share
|
20,113,659 | 20,312,707 | 20,562,645 | 20,108,688 | 19,868,956 | |||||||||||||||
|
December
31,
|
||||||||||||||||||||
| 2009 | 2008 | 2007 | 2006 | 2005 | ||||||||||||||||
|
BALANCE
SHEET DATA:
|
||||||||||||||||||||
|
Cash
and cash equivalents
|
$ | 7,937 | $ | 9,980 | $ | 12,988 | $ | 10,892 | $ | 10,009 | ||||||||||
|
Working
capital
|
14,671 | 14,233 | 19,973 | 16,731 | 15,552 | |||||||||||||||
|
Total
assets
|
23,225 | 25,521 | 41,949 | 37,953 | 36,661 | |||||||||||||||
|
Current
portion of long-term debt
|
400 | 1,658 | 611 | 211 | 211 | |||||||||||||||
|
Long-term
debt
|
700 | - | 1,640 | 351 | 562 | |||||||||||||||
|
Stockholders’
equity
|
20,093 | 21,248 | 37,128 | 35,262 | 33,778 | |||||||||||||||
17
Item
7. Management’s Discussion and Analysis of Financial Condition and Results of
Operations
Forward
Looking Statements
This
annual report contains certain forward-looking statements reflecting the current
expectations of Strategic Diagnostics Inc. and its subsidiaries (the “Company”
or “SDIX”). In addition, when used in this annual report, the words
“anticipate,” “enable,” “estimate,” “intend,” “expect,” “believe,” “potential,”
“may,” “will,” “should,” “project” and similar expressions as they relate to the
Company are intended to identify said forward-looking statements. Investors are
cautioned that all forward-looking statements involve risks and uncertainties,
which may cause actual results to differ from those anticipated at this time.
Such risks and uncertainties include, without limitation, changes in demand for
products, delays in product development, delays in market acceptance of new
products, retention of customers, attraction and retention of management and key
employees, adequate supply of raw materials, inability to obtain or delays in
obtaining third party approvals or required government approvals, the ability to
meet increased market demand, competition, protection of intellectual property,
non-infringement of intellectual property, seasonality, the ability to obtain
financing and other factors more fully described in the Company’s public filings
with the U.S. Securities and Exchange Commission.
Overview
SDIX is a
biotechnology company with a core mission of developing, commercializing and
marketing innovative and proprietary products, services and solutions that
preserve and enhance the quality of human health and wellness.
The
Company believes that its competitive position has been enhanced through the
combination of talent, technology and resources resulting from the business
development activities it has pursued since its inception. The Company has
achieved meaningful economies of scale for the products it offers through the
utilization of its consolidated facilities in Newark, Delaware for the
manufacture of test kits and antibodies, and its facility located in Windham,
Maine for the manufacture of antibodies.
The
Company believes that by applying its core competency of creating custom
antibodies to assay development, it produces sophisticated diagnostic testing
and reagent systems that are responsive to customer diagnostic and information
needs. Customers benefit from a quantifiable “return on investment” by reducing
time, labor and/or material costs associated with applications for which the
Company’s products are used. In addition, the Company believes its tests provide
high levels of accuracy, reliability and actionability of essential test results
as compared to alternative products. The Company is focused on sustaining this
competitive advantage by leveraging its expertise in immunology, proteomics,
bio-luminescence and other bio-reactive technologies to continue its successful
customer-focused research and development efforts. The Company believes that an
established product base, quality manufacturing expertise, experienced sales and
marketing organization, established network of distributors, corporate partner
relationships and proven research and development expertise will be critical
elements of its potential future success.
In 2009,
the Company continued the transition from a fragmented product offering and
marketing strategy to becoming a focused organization, with proven, proprietary
technologies tied directly to its customers’ needs. The transition is most
evident in the Life Science immuno-solutions initiative and food pathogen
detection products, where the Company believes significant progress is being
made.
The
Company continues to develop and introduce new methods for the detection of food
pathogens that deliver a strong competitive advantage to its customers. In 2005,
the Company filed a patent for new technology to be used in proprietary
enrichments of its food pathogen testing methods. The patent covers technology
for increasing the specificity and sensitivity of the Company’s immunoassay test
methods. The patent also makes claims for the application of the technology in
large scale bio-production/bio-fermentation processes, such as those used in the
production of amino acids, ethanol, enzymes and other processes using
microbiological production methods.
The
Company continued to develop multiple channels to market worldwide through an
approach that includes direct sales, inside sales, distributors and agents. The
Company increased distribution for its food pathogen products in Europe and Asia
where there is growing demand for the Company’s product line.
The
Company believes it is making progress in most of its business efforts. As the
deployment of new initiatives is accelerated, building on the Company’s
leadership position in food pathogens and expanding its strong positioning in
the emerging area of genomic antibodies, the Company anticipates that the
revenue lost to market changes in its legacy businesses will be replaced and the
Company will develop a stronger, more predictable revenue base.
The
Company expects the life science and food pathogen products to be its primary
growth drivers in the future, and that the Company’s competencies and
competitive positions in these two areas are strong.
18
Results
of Operations
Year
ended December 31, 2009 versus year ended December 31, 2008
Revenues
Revenues for the year
ended December 31, 2009 decreased 2% to $27.2 million compared to $27.7 million
for the year ended December 31, 2008. The decrease in revenues was
primarily the result of a 7% decrease in sales of kit products, partially offset
by a 4% increase in sales of life science products. The
following table sets out revenues by product
category:
|
Year
Ended
|
||||||||||||||||
|
December
31,
|
Increase
(Decrease)
|
Percent
Change
|
||||||||||||||
|
2009
|
2008
|
|||||||||||||||
|
(in
thousands, except percentages)
|
||||||||||||||||
|
Life
sciences
|
$ | 14,321 | $ | 13,821 | $ | 500 | 4 | % | ||||||||
|
Kit
products
|
12,738 | 13,634 | (896 | ) | (7 | %) | ||||||||||
|
Contract
|
95 | 204 | (109 | ) | (53 | %) | ||||||||||
|
Revenues
|
$ | 27,154 | $ | 27,659 | $ | (505 | ) | (2.0 | %) | |||||||
Life
Sciences Products
Life
science revenues increased 4% to $14.3 million for the year ended December 31,
2009, compared to $13.8 million for the year ended December 31,
2008. Sales of bulk antibody products increased 10% to $3.4 million,
sales of products utilizing the Company’s GAT ™ platform increased 8% to $1.6
million and sales of custom polyclonal products increased 2% to $5.7 million all
as a result of increased sales to the Company’s IVD (In-vitro Diagnostics)
customers. These increases were partially offset by a decline in
sales of custom monoclonal products of 4% to $3.3 million,
Kit
Products
Kit products revenues
decreased 7% to $12.7 million for the year ended December 31, 2009 compared to
$13.6 million for 2008. Sales of food pathogen products were $5.5 million in
both 2009 and 2008. Water quality product sales decreased 7% to $4.8
million for 2009 compared to 2008, which was primarily attributable to
decreased demand for the Company’s water testing equipment in China. Ag-GMO
products decreased 18% to $2.4 million for 2009 compared to 2008, primarily
attributable to decreased demand for the Company’s testing products in Brazil as
well as the elimination of required testing for the Starlink TM
trait in grains in the United States.
Gross
profit (defined as revenues less manufacturing costs) increased to $14.7 million
for the year ended December 31, 2009 from $14.6 million for the year ended
December 31, 2008. Gross margins improved to 54% for 2009 compared to
53% in 2008. The increase in margin was primarily attributable to
increased sales of the Life Science products which have a lower per unit
cost of sale.
Research and development
expenses were $2.9 million for the year ended December 31, 2009, compared to
$3.6 million, or 13% of revenues, for the year ended December 31,
2008. This decrease was primarily due to decreased spending and
effort on development of the Company’s proprietary SEQer™ antibodies, which are
produced by the GAT ™ platform and are being sold through the Company’s antibody
catalog and
decreased spending and effort on development of the Company’s proprietary phage
technology for use in the production of
ethanol.
Selling,
general and administrative expenses (SG&A) were $13.6 million for the year
ended December 31, 2009, compared to $14.4 million for the year ended December
31, 2008. This decrease is primarily the result of decreased professional fees
related to the recruitment of new management, reductions in severance costs
related to the replacement of prior management and an insurance settlement
related to employee fraud.
Interest
income (expense), net
The
Company recorded $15,000 in net interest expense for the year ended December 31,
2009 compared to net interest income of $157,000 for the year ended December 31,
2008, due to lower interest rates received on decreased levels of invested cash
and cash equivalents during 2009.
19
Income
taxes
The
Company recorded an income tax benefit of $112,000 for the year ended December
31, 2009 compared to an income tax expense of $8.4 million for the year ended
December 31, 2008. The 2009 federal income tax benefit is primarily
the result of changes in federal tax law that provided the ability to realize
research and experimentation credits and alternative minimum tax credits that
previously had a valuation allowance placed against them. The tax
provision for 2008 is due to valuation allowances placed against U.S. federal
and state deferred tax assets in 2008, as a result of uncertainty as to the
realization of the net deferred tax assets prior to their
expiration.
Net
loss
Net loss
for the year ended December 31, 2009 was $1.7 million, or $0.08 per diluted
share, compared to a net loss of $15.8 million, or $0.78 per diluted share, for
the year ended December 31, 2008. Diluted shares utilized in these computations
were 20.1 million and 20.3 million for the 2009 and 2008 periods,
respectively.
Year
ended December 31, 2008 versus year ended December 31, 2007
Revenues
Revenues
for the year ended December 31, 2008 increased 2% to $27.7 million, compared to
$27.2 million for 2007. The increase in revenues was primarily the result of a
7% increase in the sale of kit products partially offset by a 3% decline in the
sale of life science products. The following table sets out revenues
by product category:
|
Year
Ended
|
||||||||||||||||
|
December
31,
|
Increase
(Decrease)
|
Percent
Change
|
||||||||||||||
|
2008
|
2007
|
|||||||||||||||
|
(in
thousands, except percentages)
|
||||||||||||||||
|
Life
sciences
|
$ | 13,821 | $ | 14,281 | $ | (460 | ) | (3 | %) | |||||||
|
Kit
products
|
13,634 | 12,770 | 864 | 7 | % | |||||||||||
|
Contract
|
204 | 156 | 48 | 31 | % | |||||||||||
|
Revenues
|
$ | 27,659 | $ | 27,207 | $ | 452 | 2 | % | ||||||||
Life
Sciences Products
Life
science revenues decreased 3% to $13.8 million for the year ended December 31,
2008, compared to $14.3 million for 2007. Revenues from the Company’s GAT™
platform grew by 155% to $1.4 million. This increase is primarily attributable
to continued adoption by our clients of the Company’s GAT™ platform. This
increase was offset by declines in bulk antibody sales of 19% to $3.1 million,
custom monoclonal antibodies of approximately 3% to $3.4 million, and a decline
in custom polyclonal antibodies of 9% to $5.6 million. These decreases are
primarily attributable to decreased orders by customers that had previously
accumulated excess inventories.
Kit
Products
Kit
products revenues increased 7% to $13.6 million for the year ended December 31,
2008, compared to $12.8 million for 2007.
Sales of
tests for food pathogens increased 20%, to $5.5 million in 2008, from $4.6
million in 2007, as the Company continued to see increasing acceptance of its
line of products to detect pathogens in food. Water quality products revenue
increased by 2% to $5.2 million for the year ended December 31, 2008, compared
to $5.1 million for the year ended December 31, 2007. This increase was
primarily the result of increased water quality equipment sales to
China. Ag-GMO revenues declined 6%, to $2.9 million in 2008 from $3.1
million in 2007, due to a shrinking marketplace for products that detect genetic
traits in grain and seed.
Gross
profit (defined as revenue less manufacturing costs) for the year ended December
31, 2008 was $14.6 million, compared to $16.4 million for 2007. The gross margin
was 53% in 2008, compared to 60% in 2007. This decrease in margin
was primarily the result of underutilized capacity in the antibody product
category and increased write-offs of obsolete inventory in both the food safety
(specifically Ag-GMO) and antibody product categories.
20
For the
year ended December 31, 2008, operating expenses increased 37% to $35.2 million,
compared to $25.8 million for 2007. This increase is primarily due to
approximately $4.2 million of expense related to goodwill impairment and an
increase of approximately $2.4 million in selling, general and administrative
costs.
For the
year ended December 31, 2008, research and development spending was $3.6
million, compared to $2.9 million for 2007. This increase was primarily due to
increased spending and effort on development of the Company’s proprietary
antibodies, which are produced by the Company’s GAT™ platform and are being sold
through the Company’s antibody catalog, and increased spending and effort on
development of the Company’s proprietary phage technology for use in the
production of ethanol.
During
the fourth quarter of 2008, the Company performed the annual goodwill impairment
analysis and recorded a non-cash goodwill impairment charge of $4.2 million.
Refer to Note 3 of the Notes to the Consolidated Financial Statements for
further information.
Selling,
general and administrative expenses were $14.4 million for the year ended
December 31, 2008, compared to $12.0 million for 2007. The 20% increase is
primarily attributable to severance costs for the Company’s former CEO,
recruiting costs associated with management and personnel changes that were
initiated during 2008 and continued expansion of the Company’s sales and
marketing efforts.
Interest
income, net
The
Company recorded net interest income of $157,000 during 2008 compared to net
interest income of $433,000 during 2007. This decrease is primarily due to
increased debt and related interest expense on our $2 million term loan executed
in August 2007, and decreased interest rates received on lower levels of
invested cash balances.
Income
taxes
The
Company recorded income tax expense of $8.4 million in 2008, which includes a
$10.7 million increase in the valuation allowance, recorded against net deferred
tax assets of approximately $12.5 million. The valuation allowance was recorded
primarily due to being in a cumulative three year pre-tax loss position as of
December 31, 2008 and uncertainty in overall economic conditions, which may not
allow the Company to realize the net deferred tax assets prior to their
expiration. These non-cash charges do not impact the Company’s ongoing business
operations. In 2007, the Company recorded income tax expense of $976,000,
representing an effective tax rate of 53%.
Net
Income (loss)
Net loss
for the year ended December 31, 2008 was $15.8 million, or $0.78 per diluted
share, compared to net income of $860,000, or $0.04 per diluted share for 2007.
Diluted shares totaling 20.3 million and 20.6 million were used in the
computations for 2008 and 2007, respectively.
Liquidity
and Capital Resources
Liquidity
is our ability to generate sufficient cash flows from operating activities to
meet the Company’s obligations and commitments, or obtain appropriate financing.
Currently our liquidity needs arise primarily from debt service on indebtedness,
working capital requirements and capital expenditures.
21
The
following is a summary of selected cash flow information:
|
Year
Ended
|
||||||||||||
|
December
31,
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
(in
thousands)
|
||||||||||||
|
Net
cash provided by (used in) operating activities
|
$ | 366 | $ | (749 | ) | $ | 2,302 | |||||
|
Net
cash used in investing activities
|
(499 | ) | (895 | ) | (2,482 | ) | ||||||
|
Net
cash provided by (used in) financing activities
|
(1,791 | ) | (1,082 | ) | 2,262 | |||||||
|
Effect
of exchange rate changes on cash
|
(119 | ) | (282 | ) | 14 | |||||||
|
Net
increase (decrease) in cash and cash equivalents
|
$ | (2,043 | ) | $ | (3,008 | ) | $ | 2,096 | ||||
Net cash
provided by operating activities in 2009 primarily relates to the net loss for
the year, offset by depreciation, amortization and stock based compensation
charges, decreases in accounts receivable and inventories and decreases in
accounts payable and accruals. For 2008, net cash used in operating
activities primarily relates to the net loss incurred for the year offset by
depreciation, amortization and stock based compensation charges, the goodwill
impairment charge and deferred income taxes.
Net cash
used in investing activities for 2009 was $499,000 compared to $895,000 for
2008. These cash outflows were primarily the result of capital purchases which
were $500,000 in 2009 versus $929,000 in 2008. The capital expenditures in 2009
were primarily related to the purchase of computer and electronic equipment. The
capital expenditures in 2008 were primarily related to the purchase of lab and
manufacturing equipment.
Net cash
used in financing activities was $1.8 million for 2009, primarily related to a
$1.25 million restricted cash requirement from the Company’s lender, and
$558,000 in debt repayments. In 2008, net cash used in financing
activities was $1.1 million, primarily the result of $593,000 in debt repayments
and $555,000 in purchases of treasury stock.
The
Company’s working capital (current assets less current liabilities) increased to
$14.7 million at December 31, 2009 from $14.2 million at December 31, 2008,
primarily due to the reclassification of $700,000 of debt to long-term from
current as a result of our amended credit agreement described
below. Outstanding debt decreased to $1.1 million at December 31,
2009 from $1.7 million at December 31, 2008 due to scheduled debt
payments.
On May 5,
2000, the Company entered into a financing agreement with a commercial bank
which was amended on August 12, 2009 (as amended, the “Credit
Agreement”). The Credit Agreement was amended to eliminate the
revolving line of credit, remove financial covenants requiring minimum EBITDA
and tangible net worth amounts, and reduce the restricted cash requirement from
$2.7 million to $1.25 million.
On August
21, 2007, the Company received a term loan under the Credit Agreement to finance
the construction of new facilities at its Windham, Maine location. This
agreement provided for up to $2 million in financing, $1.1 million of which was
outstanding at December 31, 2009, and is repayable over five years, with
principal payments that began on October 1, 2007. The loan bears a fixed
interest rate of 5.96% with equal amortization of principal payments plus
interest. This indebtedness is secured by $1.25 million in restricted cash as
required by the Credit Agreement.
For the
year ended December 31, 2009, the Company satisfied all of its cash requirements
from cash and cash equivalents on-hand. At December 31, 2009, the Company had
$1.1 million in debt and stockholders’ equity of $20.1 million.
Based
upon its cash and cash equivalents on hand, credit facilities, current product
sales and the anticipated sales of new products, the Company believes it has, or
has access to, sufficient resources to meet its operating requirements at least
through the next 12 months.
The
Company’s ability to meet its long-term capital needs will depend on a number of
factors, including compliance with existing and new loan covenants, the success
of its current and future products, the focus and direction of its research and
development program, competitive and technological advances, future
relationships with corporate partners, government regulation, the Company’s
marketing and distribution strategy, its successful sale of additional common
stock and/or the Company successfully locating and obtaining other financing,
and the success of the Company’s plan to make future acquisitions. Accordingly,
no assurance can be given that the Company will be able to meet the long-term
liquidity requirements that may arise from these inherent and similar
uncertainties.
22
Off-Balance
Sheet Arrangements
As of
December 31, 2009, the Company did not have any off-balance sheet arrangements
as defined in Item 304(a) (4) (ii) of Regulation S-K.
Contractual
Obligations
The
Company is committed to making cash payments in the future on two types of
contracts: its long-term indebtedness and leases. The Company has no off-balance
sheet debt or other such unrecorded obligations. Below is a schedule of the
future payments that the Company was obligated to make based on agreements in
place as of December 31, 2009.
|
Payments
Due by Year
|
||||||||||||||||||||||||||||
|
2015
and
|
||||||||||||||||||||||||||||
|
2010
|
2011
|
2012
|
2013
|
2014
|
Beyond
|
Total
|
||||||||||||||||||||||
|
(in
thousands)
|
||||||||||||||||||||||||||||
|
Long-term
debt (1)
|
$ | 400 | 400 | 300 | - | - | - | 1,100 | ||||||||||||||||||||
|
Operating
leases (2)
|
$ | 1,169 | 845 | 330 | 300 | 300 | 600 | 3,544 | ||||||||||||||||||||
|
Total
contractual
|
||||||||||||||||||||||||||||
|
cash
obligations
|
$ | 1,569 | 1,245 | 630 | 300 | 300 | 600 | 4,644 | ||||||||||||||||||||
|
(1)
|
See
Note 5 to the Consolidated Financial Statements for a discussion of
long-term debt
|
|
(2)
|
See
Note 8 to the Consolidated Financial Statements for a discussion of
operating leases
|
Critical
Accounting Policies
The
Company’s accounting policies are described in Note 1 of the Notes to the
Consolidated Financial Statements. The Consolidated Financial Statements are
prepared in conformity with U.S. generally accepted accounting principles
(“GAAP”), which require the Company to make estimates and assumptions that
affect the reported amounts of assets and liabilities and disclosures of
contingent assets and liabilities at the date of the financial statements and
the reported amounts of revenues and expenses during the year. On an on-going
basis, the Company evaluates its estimates, including those related to bad
debts, inventories, deferred taxes, long-lived assets and stock-based
compensation. The Company bases its estimates on historical experience and on
various other assumptions that the Company believes are reasonable under the
circumstances. The results form the basis for making judgments about the
carrying values of assets and liabilities that are not readily apparent from
other sources. Actual results could differ from those estimates. The Company
considers the following policies to be most critical in understanding the
judgments that are involved in preparing the Consolidated Financial Statements
and the uncertainties that could impact the consolidated results of operations,
financial condition and cash flows.
Valuation
of Accounts Receivable – Accounts receivable as of December 31, 2009 and
December 31, 2008, were net of an allowance for doubtful accounts of $38,000 and
$70,000, respectively. The recorded allowance is continually evaluated based on
current market conditions, an analysis of customer-specific facts and
circumstances, and the size and composition of the overall portfolio. The
current state of the economy could cause longer sales cycles resulting in
increased risk that outstanding balances could become uncollectible. If
receivables are in dispute with the customer or otherwise deemed uncollectible,
the corresponding amounts are written off and are charged against the
allowance.
Valuation
of Inventories – Inventories are valued at the lower of cost or
market.
For
inventories that consist primarily of test kit components, bulk antibody serum
and antibody products, cost is determined using the first in, first out method.
Realization of inventories is dependent upon the successful marketing of our
products. Judgments are made regarding the carrying value of inventory based on
current market conditions. Market conditions may change depending upon
competitive product introductions and customer demand. If market conditions
change or if the introduction of new products by the Company impacts the market
for previously released products, the Company may be required to write-down the
cost of its inventory.
For
inventories that consist of costs associated with the production of custom
antibodies, cost is determined using the specific identification method.
Realization of such inventories is dependent upon the successful completion of a
project in accordance with customer specifications. Losses on projects in
progress are recorded in the period such losses become probable and
estimable.
23
Deferred
Taxes –
In assessing the realizability of deferred tax assets, management
considers whether it is more likely than not that some portion or all of the
deferred tax assets will not be realized. The ultimate realization of deferred
tax assets is dependent upon the generation of future taxable income during the
period in which those temporary differences become deductible. In making their
assessment, management considers positive evidence, negative evidence, and
possible tax planning strategies that could be
implemented. Management also considers the future reversal of
existing taxable temporary differences, recent earnings history, history of or
potential for tax attributes such as net operating losses to expire and the
ability to project future taxable income. The Company’s history of cumulative
pre-tax losses over the most recent three-year period, including 2009, is
significant negative evidence that is difficult to overcome. In light
of this negative evidence, coupled with the current economic conditions,
management has concluded that it is more likely than not that the Company will
not realize the benefits of these tax deductible differences and has continued
to provide a full valuation allowance offsetting its U.S. federal and state net
deferred tax assets at December 31, 2009.
At
December 31, 2009, the Company had approximately $4.9 million of net operating
loss carryforwards for tax purposes, which have no expiration and which
correspond to a $1.4 million deferred tax asset, related to operations in the
United Kingdom (“UK”). Management considered positive and negative indicators in
concluding that a substantial valuation allowance of approximately $1.32 million
was necessary for the foreign deferred tax assets of $1.37 million. The positive
indicators include the contribution to income before taxes by the foreign
operations in the UK for 2009, 2008 and 2007, and the expected income before
taxes in the UK for 2010 and 2011. The negative indicators include a history of
substantial net operating losses in the UK, the lack of income before taxes
until 2004 and limitations with regard to estimating income in the UK beyond
2011 resulting from a year-to-year evaluation of the future need for a UK
subsidiary.
As of
December 31, 2009, the Company had U.S. federal net operating loss
carryforwards, of $17.4 million, including those of acquired companies, which
begin to expire as follows:
|
Year
|
Net
Operating
Loss (in thousands) |
||||
|
2010
|
$ | 4,536 | |||
|
2017
|
760 | ||||
|
2018
|
1,327 | ||||
|
2019
|
550 | ||||
|
2020
|
66 | ||||
|
2021
|
56 | ||||
|
2022
|
2,268 | ||||
|
2024
|
2,033 | ||||
|
2025
|
3 | ||||
|
2026
|
1 | ||||
|
2027
|
1 | ||||
|
2028
|
3,492 | ||||
|
2029
|
2,287 | ||||
|
Total
|
$ | 17,380 | |||
Revenue
Recognition — Revenues composed of sales of immunoassay-based test kits
and certain antibodies and immunochemical reagents are recognized upon the
shipment of the product and transfer of title or when related services are
provided. Revenues associated with such products or services are recognized when
persuasive evidence of an order exists, shipment of product has occurred or
services have been provided, the price is fixed or determinable, and
collectability is reasonably assured. Management is required to make judgments
based on actual experience about whether or not collectability is reasonably
assured.
The
Company enters into contracts related to the production of custom antibodies,
which provide for the performance of defined tasks for a fixed price, with
delivery of the product upon completion of production. The standard time to
complete a project is typically longer than 30 days but less than 12 months and
effort is expended over the life of the project. Revenues related to sales of
custom antibody projects are recognized when a project’s specifications have
been met and/or the related materials have been shipped.
24
Fees
associated with products and services added on to a custom antibody project
subsequent to delivery of the initial project are billed monthly and recognized
as revenue as the services and other deliverables are provided.
Valuation
of Goodwill and Long-Lived Assets — Long-lived assets, such as property
and equipment, and purchased intangibles subject to amortization, are reviewed
for impairment whenever events or changes in circumstances indicate that the
carrying amount of an asset may not be recoverable. Recoverability of assets to
be held and used is measured by a comparison of the carrying amount of an asset
to its estimated undiscounted future cash flows expected to be generated by the
asset. If the carrying amount of an asset exceeds its estimated future cash
flows, an impairment charge is recognized by the amount by which the carrying
amount of the asset exceeds the fair value of the asset. Goodwill and intangible
assets not subject to amortization are tested annually for impairment, and are
tested for impairment more frequently if events and circumstances indicate that
the asset might be impaired. An impairment loss is recognized to the extent that
the carrying amount exceeds an asset’s fair value.
The
Company tests goodwill annually in a two-step approach. The first step is the
comparison of the carrying value of the Company, including goodwill, to the fair
value of the Company at the testing date. If the carrying amount exceeds the
fair value of the Company, a second test is performed to measure the amount of
an impairment charge, if any. To measure the amount of any impairment charge,
the Company determines the implied fair value of goodwill in the same manner as
if the Company were being acquired in a business combination.
In the
fourth quarter of 2008, the Company recorded an impairment charge of
approximately $4.2 million to writeoff goodwill and other intangible assets. For
additional information, refer to Note 3 of the Notes to the Consolidated
Financial Statements.
Share–Based
Compensation — The Company accounts for stock-based compensation in
accordance with the fair value method of accounting, which requires the Company
to measure all employee share–based compensation awards at the date of grant and
recognize such expense in our consolidated financial
statements.
The grant
date fair value of the awards is recognized as compensation expense over the
vesting period of the awards and is included in selling, general and
administrative expenses. Management is required to make estimates and
assumptions to determine the grant date fair value of stock options, including
the expected term of stock options and the volatility of our stock price in the
future. In addition, assumptions related to expected future forfeitures and
performance-based vesting features all impact expense recognition. These
assumptions have an impact on the valuation assigned to equity awards and the
associated recognition of expense.
New
Accounting Standards and Disclosures
In June
2009, the Financial Accounting Standards Board (“FASB”) issued Statement of
Financial Accounting Standards (SFAS) No. 168, The FASB Accounting
Standards Codification and the Hierarchy of Generally Accepted Accounting
Principles (“SFAS 168”). SFAS 168 establishes the FASB Accounting Standards
Codification (“ASC”) as the single source of authoritative nongovernmental
GAAP. The ASC only changes the referencing of financial
accounting standards and does not change or alter existing GAAP. The
Codification is effective for interim and annual periods ending after
September 15, 2009. The adoption of this standard did not have a material
impact on the Company’s financial statements. SFAS 168 has been
incorporated into the ASC as ASC-105, Generally Accepted Accounting Principles
or ASC 105.
In
December 2007, the FASB issued SFAS No. 141(Revised), Business
Combinations SFAS No. 141(R), which replaces SFAS No. 141, Business
Combinations, and requires an acquirer to recognize the assets acquired, the
liabilities assumed, and any non-controlling interest in the acquired at the
acquisition date, measured at their fair values as of that date, with limited
exceptions. This statement also requires the acquirer in a business combination
achieved in stages to recognize the identifiable assets and liabilities, as well
as the non-controlling interest in the acquired, at the full amounts of their
fair values. SFAS No. 141(R) makes various other amendments to
authoritative literature intended to provide additional guidance or to confirm
the guidance in that literature to that provided in this statement. This
statement applies prospectively to business combinations for which the
acquisition date is on or after the beginning of the first annual reporting
period beginning on or after December 15, 2008. SFAS
No. 141(R)’s impact on accounting for business combinations is dependent
upon future acquisitions the Company may make. SFAS 141(R) has been
incorporated into the ASC as ASC-805, Business Combinations (“ASC
805”).
In April
2008, the FASB issued FASB Staff Position No. FAS 142-3, Determination of the
Useful Life of Intangible Assets (“FSP 142-3”). FSP 142-3 amends the
factors an entity should consider in developing renewal or extension assumptions
used in determining the useful life of recognized intangible assets under SFAS
No. 142, Goodwill and Other Intangible Assets. This new guidance
applies prospectively to intangible assets that are acquired individually or
with a group of other assets in business combinations and asset
acquisitions. FSP 142-3 is effective for financial statements issued
for fiscal years and interim periods beginning after December 15,
2008. FSP 142-3’s impact on the Company is dependent upon future
acquisitions the Company may make. FSP 142-3 has been incorporated into the ASC
as ASC-350, Intangibles (“ASC 350”).
25
In
September 2009, the FASB issued Accounting Standard Update (“ASU”)
No. 2009-13,
Revenue Recognition (Topic 605) – Multi-Deliverables Revenue
Arrangements, a Consensus of the FASB Emerging Issues Task Force, to address the
accounting for multiple-deliverable arrangements to enable vendors to account
for products or services (deliverables) separately rather than as a combined
unit. It establishes the accounting and reporting guidance for arrangements
under which the vendor will perform multiple revenue-generating activities,
specifically, how to separate deliverables and how to measure and allocate
arrangement consideration to one or more units of accounting. The update will be
effective prospectively for revenue arrangements entered into or materially
modified in fiscal years beginning on or after June 15, 2010. The Company
is currently evaluating the impact, if any, of adopting the update.
Item
7A. Quantitative and Qualitative Disclosures About Market Risk
The
Company has limited exposure to changing interest rates, and is currently not
engaged in hedging activities. Interest on approximately $1.1 million of debt,
is at a fixed annual rate of interest of 5.961%.
The
Company conducts operations in the United Kingdom. The consolidated financial
statements of the Company are denominated in U.S. dollars and changes in
exchange rates between foreign countries and the U.S. dollar will affect the
translation of financial results of foreign subsidiaries into U.S. dollars for
purposes of recording the Company’s consolidated financial results.
Historically, the effects of translation have not been material to the
consolidated financial results.
Item
8. Financial Statements and Supplementary Data
The
following consolidated financial statements and supplemental quarterly financial
data of the Company and its subsidiaries are included as part of this Form
10-K:
|
Page
|
||
|
Management’s
Report on Internal Control over Financial Reporting
|
32
|
|
|
Report
of Independent Registered Public Accounting Firm
|
33
|
|
|
Consolidated
Balance Sheets as of December 31, 2009 and 2008
|
34
|
|
|
Consolidated
Statements of Operations for each of the years in the three-year period
ended December 31, 2009
|
35
|
|
|
Consolidated
Statements of Stockholders’ Equity and Comprehensive Income (Loss) for
each of the years in the three-year period ended December 31,
2009
|
36
|
|
|
Consolidated
Statements of Cash Flows for each of the years in the three-year period
ended December 31, 2009
|
37
|
|
|
Notes
to Consolidated Financial Statements
|
38
|
Item
9. Changes in and Disagreements with Accountants on Accounting and Financial
Disclosure
None.
Item
9A. Controls and Procedures
(a)
Evaluation of Disclosure Controls and Procedures
Our
management, with the participation of our Chief Executive Officer and Chief
Financial Officer, evaluated the effectiveness of our disclosure controls and
procedures, as defined in Rule 13a-15(e) under the Securities Exchange Act of
1934, as amended, as of the end of the period covered by this report. Based upon
that evaluation, the Chief Executive Officer and Chief Financial Officer
concluded that our disclosure controls and procedures as of December 31, 2009,
were functioning effectively to provide reasonable assurance that the
information required to be disclosed by us in reports filed or submitted under
the Securities Exchange Act of 1934, as amended, is (i) recorded, processed,
summarized and reported within the time periods specified in the SEC’s rules and
forms, and (ii) accumulated and communicated to our management, including our
principal executive and financial officers, or persons performing similar
functions, as appropriate to allow timely decisions regarding required
disclosure.
(b)
Change in Internal Control over Financial Reporting
No change
in our internal control over financial reporting occurred during the quarter
ended December 31, 2009 that has materially affected, or is reasonably likely to
materially affect, our internal control over financial reporting.
Management’s
Report on Internal Control over Financial Reporting
The
report of management on our internal control over financial reporting is set
forth in Item 8 of this report and is incorporated herein by
reference.
Item
9B. Other Information
None.
26
PART
III
Item
10. Directors, Executive Officers and Corporate Governance
The
applicable information set forth in the Company’s Definitive Proxy Statement is
incorporated herein by reference.
Identification
of Executive Officers and Certain Significant Employees
The
executive officers of the Company, their positions with the Company, their ages
and a brief biography for each are as follows:
|
Name
|
Age
|
Position
|
|||||
|
Francis
M. DiNuzzo
|
54
|
President
and Chief Executive Officer
|
|||||
|
Klaus
Lindpaintner
|
55
|
VP
– Research and Development and Chief Science Officer
|
|||||
|
(effective
February 1, 2010)
|
|||||||
|
Kevin
J. Bratton
|
60
|
VP
– Finance, Chief Financial Officer and Corporate
Secretary
|
|||||
Francis
M. DiNuzzo has served as President and Chief Executive Officer since
October 13, 2008. Mr. DiNuzzo joined the Company in February 2008 as Executive
Vice President – Marketing and Chief Commercial Officer, and on June 6, 2008, he
was appointed interim President and Chief Executive Officer. Prior to joining
SDIX, Mr. DiNuzzo spent 26 years at Agilent Technologies / Hewlett Packard. He
began his career in research and development in 1981 and advanced through a
series of functional management roles over the next 18 years. In 1999, Mr.
DiNuzzo became General Manager of Agilent’s Chemical Solutions Business Unit
where he had global responsibility for analytical equipment, consumables and
service products serving the chemical, environmental, food and forensics
markets. In 2001, Mr. DiNuzzo became General Manager of the Consumable and
Services Business Unit, with global responsibility for all consumables and
services across all Life Science and Chemical Analysis markets. In 2004, Mr.
DiNuzzo became Vice President and General Manager of the Integrated Biology
Solutions unit, a role where he formed a biotechnology business focused on
Genomics, Proteomics and BioInformatics. Mr. DiNuzzo holds B.S and M.S. degrees
in Engineering with a minor in Business Administration from the University of
New Hampshire.
Klaus
Lindpaintner joined SDIX in February 2010 as Vice President – Research
and Development and Chief Scientific Officer. Prior to joining SDIX,
Dr. Lindpaintner was with F. Hoffmann-La Roche Ltd. for 13 years and most
recently served as Director of the “Roche Molecular Medicine Laboratories” at
the company’s headquarters in Basel, Switzerland, and as Roche’s Global Head,
Molecular Medicines Policy and External Affairs, coordinating the company’s
efforts and activities in implementing biomarker research based on genetics,
genomics, proteomics, and associated disciplines from early discovery to
late-stage clinical trials. Dr. Lindpaintner graduated from the
Innsbruck University Medical School with a degree in Medicine and from Harvard
University with a degree in Public Health.
Kevin
J. Bratton joined SDIX in June 2009 as Vice President – Finance and Chief
Financial Officer. Mr. Bratton was most recently Senior Vice
President Business Operations for EUSA Pharma (USA), Inc. in Langhorne,
Pennsylvania. Mr. Bratton had been Senior Vice President and Chief
Financial Officer of Cytogen Corporation in Princeton, New Jersey prior to its
acquisition by EUSA Pharma, Inc. in May 2008. Mr. Bratton has over 35
years of experience in all phases of multi-national financial operations across
the healthcare, biotechnology and technology industries, including developing
strategic plans and annual budgets as well as financing negotiations and merger
& acquisition transactions. Prior to joining Cytogen, Mr. Bratton
was Chief Financial Officer at Metrologic Instruments, Inc., a global technology
company, where he directed the company’s finance operations during a period of
significant growth in sales, net income, cash flow from operations, and working
capital. Previously, Mr. Bratton began his career with the public accounting
firm Touche Ross & Co. (now Deloitte & Touche LLP). He has a bachelor of
science in business and accounting from Northeastern
University.
Item
11. Executive Compensation
The
applicable information set forth in the Company’s Definitive Proxy Statement is
incorporated herein by reference.
Item
12. Security Ownership of Certain Beneficial Owners and Management and Related
Stockholder Matters
The
applicable information set forth in the Company’s Definitive Proxy Statement is
incorporated herein by reference.
27
Equity
Compensation
The table
below presents certain information as of December 31, 2009 concerning securities
issuable in connection with equity compensation plans that have been approved by
the Company’s stockholders and that have not been approved by the Company’s
stockholders.
|
Plan
Category
|
Number
of
securities
to be
issued
upon
exercise
of
outstanding
options,
warrants
and
rights
(a)
|
Weighted-average
exercise
price of
outstanding
options,
warrants
and
rights
(b)
|
Number
of securities
remaining
available
for
issuance under
equity
compensation
plans
(excluding
securities
reflected in
column
(a))
(c)
|
|||||||||
|
Equity
compensation plan
|
||||||||||||
|
approved
by security
|
||||||||||||
|
holders
|
1,541,800 | $ | 2.90 | 1,906,731 | ||||||||
|
Equity
compensation
|
||||||||||||
| not approved by | ||||||||||||
| security holders | 160,000 | $ | 1.50 | - | ||||||||
|
Total
|
1,701,800 | $ | 2.77 | 1,906,731 | ||||||||
The
160,000 shares underlying options granted under equity compensation not approved
by security holders were granted in connection with the Company’s hiring, on
January 28, 2009, of its Vice-President Marketing and Sales, Monette Greenway,
85,000 shares, and in connection with the Company’s hiring on June 1, 2009, of
its Chief Financial Officer, Kevin Bratton, 75,000 shares. The grants to Ms.
Greenway and Mr. Bratton are ten year non-qualified stock option grants at an
exercise price of $1.50 per share.
Item
13. Certain Relationships and Related Transactions, and Director
Independence
The
applicable information set forth in the Company’s Definitive Proxy Statement is
incorporated herein by reference.
Item
14. Principal Accounting Fees and Services
The
applicable information set forth in the Company’s Definitive Proxy Statement is
incorporated herein by reference.
28
PART
IV
Item
15. Exhibits and Financial Statement Schedules
1.
Financial Statements
(a) See
the Consolidated Financial Statements which begin on page 43 of this
report.
2.
Financial Statement Schedules
Financial
statement schedules are omitted because they are either not required or not
applicable or the required information is reflected in the financial statements
or notes thereto.
3.
Exhibits
|
Exhibit
Number
|
Description
|
Reference
|
||
|
2.1
|
Agreement
and Plan of Merger among the Company, AZUR Acquisition Corp. and AZUR
Environmental dated May 4, 2001
|
(1)
|
||
|
3.1
|
Fourth
Amended and Restated Certificate of Incorporation of the
Company
|
(2)
|
||
|
3.2
|
Amended
and Restated Bylaws of the Company
|
(2)
|
||
|
4.1
|
Reference
is made to Exhibits 3.1 and 3.2
|
|||
|
4.2
|
Forms
of Warrants to Purchase Common Stock of the Company
|
(2)
|
||
|
10.1
|
Stock
Purchase Agreement among the Company and its outside directors and certain
of their affiliates dated August 16, 2002
|
(12)
|
||
|
10.2
|
Demand
Registration Rights Agreement among the Company and its outside directors
and certain of their affiliates dated August 16, 2002
|
(12)
|
||
|
10.3
|
EnSys
Environmental Products, Inc. 1993 Stock Incentive Plan*
|
(3)
|
||
|
10.4
|
Amended
and Restated EnSys Environmental Products, Inc. 1995 Stock Incentive
Plan*
|
(4)
|
||
|
10.5
|
EnSys
Environmental Products, Inc. 401(k) Plan Adoption
Agreement
|
(3)
|
||
|
10.11
|
Agreement
and Plan of Merger by and between EnSys and Strategic Diagnostics Inc.
dated as of October 11, 1996
|
(2)
|
||
|
10.18
|
Industrial
Lease dated October 26, 1993, by and between Tober & Agnew Properties,
Inc. and Strategic Diagnostics Inc.
|
(6)
|
||
|
10.21
|
Lease
agreement dated October 29, 1997 by and between Pencader Courtyard, L.P.
and Strategic Diagnostics Inc.
|
(7)
|
||
|
10.22
|
1998
Employee Stock Purchase Plan
|
(11)
|
||
|
10.23
|
2000
Stock Incentive Plan*
|
(18)
|
|
10.27
|
Loan
Agreement between the Company and PNC Bank, Delaware, dated May 5,
2000
|
(10)
|
||
|
10.28
|
Line
of Credit Note between the Company and PNC Bank, Delaware, dated May 5,
2000
|
(10)
|
||
|
10.29
|
Term
Note between the Company and PNC Bank, Delaware, dated May 5,
2000
|
(10)
|
||
|
10.30
|
Employment
Agreement dated September 2, 2003, by and between Matthew H. Knight and
the Company*
|
(13)
|
||
|
10.31
|
Nonqualified
Stock Option Agreement dated September 2, 2003, by and between Matthew H.
Knight and the Company*
|
(13)
|
29
|
10.32
|
Restricted
Stock Grant Agreement dated September 2, 2003, by and between Matthew H.
Knight and the Company*
|
(13)
|
||
|
10.33
|
Employment
Agreement dated January 1, 1997 by and between James W. Stave and the
Company*
|
(14)
|
||
|
10.34
|
Amended
and Restated Distribution Agreement, dated as of August 31, 2009, by and
between the Registrant and the DuPont Qualicon division of E.I. du Pont de
Nemours and Company
|
(15)
(16) |
||
|
10.35
|
Strategic
Diagnostics Inc. Change of Control Severance Agreement*
|
(17)
|
||
|
10.36
|
Agreement,
dated as of March 12, 2008, by and among the Company and Steven R. Becker,
BC Advisors, LLC, SRB Management, L.P. and Richard van den
Broek
|
(19)
|
||
|
10.37
|
Separation
Agreement, dated as of June 6, 2008, between Strategic Diagnostics Inc.
and Matthew H. Knight*
|
(20)
|
||
|
10.38
|
Employment
Agreement, dated as of October 13, 2008, between Strategic Diagnostics
Inc. and Francis M. DiNuzzo*
|
(21)
|
||
|
10.39
|
Separation
Agreement, dated as of December 9, 2008, between Strategic Diagnostics
Inc. and Stanley Fronczkowski*
|
(22)
|
||
|
10.40
|
Amendment
No. 1 to Separation Agreement and General Release, dated as of April 3,
2009, between Strategic Diagnostics Inc. and Stanley
Fronczkowski*
|
(23)
|
||
|
10.41
|
Form
of Nonqualified Stock Option Agreement
|
|||
|
10.42
|
Form
of Restricted Stock Grant Agreement
|
|||
|
21.1
|
Subsidiaries
of the Company
|
|||
|
23.1
|
Consent
of KPMG LLP, Independent Registered Public Accounting Firm
|
|||
|
31.1
|
Certifications
of the Chief Executive Officer of Strategic Diagnostics Inc. required by
Rule 13a-14(a) under the Securities Exchange Act of 1934
|
|||
|
31.2
|
Certifications
of the Chief Financial Officer of Strategic Diagnostics Inc. required by
Rule 13a-14(a) under the Securities Exchange Act of 1934
|
|||
|
32.1
|
Certification
of Francis M. DiNuzzo pursuant to 18 U.S.C. Section 1350, as adopted
pursuant to Section 906 of Sarbanes-Oxley Act of 2002
|
|||
|
32.2
|
Certification
of Kevin J. Bratton pursuant to 18 U.S.C. Section 1350, as
adopted pursuant to Section 906 of Sarbanes-Oxley Act
of 2002
|
|
(1)
|
Incorporated
by reference to the designated exhibit of the Company’s 10-Q for the
fiscal quarter ended September 30, 2001.
|
|
(2)
|
Incorporated
by reference to the designated exhibit of the EnSys Registration Statement
on Form S-4 (No. 333-17505) filed on December 9, 1996.
|
|
(3)
|
Incorporated
by reference to the designated exhibit of the EnSys Registration Statement
on Form S-1 (No. 33-68440) filed on September 3, 1993.
|
|
(4)
|
Incorporated
by reference to Appendix F to the Joint Proxy Statement/Prospectus
contained in the EnSys Registration Statement on Form S-4 (No. 333-17505)
filed on December 9, 1996.
|
|
(5)
|
Incorporated
by reference to the designated exhibit of the EnSys Form 10-K for the
fiscal year ended December 31, 1994.
|
|
(6)
|
Incorporated
by reference to the designated exhibit of the EnSys Form 10-Q for the
fiscal quarter ended March 31, 1996.
|
|
(7)
|
Incorporated
by reference to the designated exhibit of the Company’s Form 10-K for the
fiscal year ended December 31, 1996.
|
|
(8)
|
Incorporated
by reference to the designated exhibit of the Company’s Form 10-K for the
fiscal year ended December 31, 1997.
|
30
|
(9)
|
Incorporated
by reference to the identically numbered exhibit contained in the
Company’s Form 8-K filed on May 26, 1999.
|
|
(10)
|
Incorporated
by reference to the identically numbered exhibit contained in the
Company’s Form 8-K filed on March 15, 1999.
|
|
(11)
|
Incorporated
by reference to the designated exhibit of the Company’s Registration
Statement on Form S-8 (No. 333- 68107) filed on November 30,
1998.
|
|
(12)
|
Incorporated
by reference to the designated exhibit of the Company’s 10-Q for the
fiscal quarter ended September 30, 2002.
|
|
(13)
|
Incorporated
by reference to the designated exhibit of the Company’s Form 10-Q for the
fiscal quarter ended September 30, 2003.
|
|
(14)
|
Incorporated
by reference to the designated exhibit of the Company’s Form 10-K for the
fiscal year ended December 31, 2004.
|
|
(15)
|
Confidential
treatment has been requested as to certain portions of this exhibit
pursuant to Rule 24b-2 under the Securities Exchange Act of 1934, as
amended.
|
|
(16)
|
Incorporated
by reference to the designated exhibit of the Company’s Form 10-Q for the
fiscal quarter ended September 30, 2009, as amended.
|
|
(17)
|
Incorporated
by reference to the designated exhibit of the Company’s Form 10-Q for the
fiscal quarter ended September 30, 2005.
|
|
(18)
|
Incorporated
by reference to Appendix A of the Company’s Definitive Proxy Statement
filed with the Securities and Exchange Commission on March 24,
2004.
|
|
(19)
|
Incorporated
by reference to Exhibit 99.1 of the Company’s Form 8-K filed March 18,
2008.
|
|
(20)
|
Incorporated
by reference to Exhibit 99.1 of the Company’s Form 8-K filed June 6,
2008.
|
|
(21)
|
Incorporated
by reference to Exhibit 99.1 of the Company’s Form 8-K filed October 16,
2008.
|
|
(22)
|
Incorporated
by reference to Exhibit 99.1 of the Company’s Form 8-K filed December 9,
2008.
|
|
(23)
|
Incorporated
by reference to Exhibit 10.1 of the Company’s Form 10-Q for the fiscal
quarter ended March 31, 2009.
|
|
*
|
Management
contract or compensatory
plan.
|
31
MANAGEMENT’S
REPORT ON INTERNAL CONTROL OVER FINANCIAL REPORTING
Our
management is responsible for establishing and maintaining adequate internal
control over financial reporting for the Company. Internal control over
financial reporting is a process designed to provide reasonable assurance
regarding the reliability of financial reporting and the preparation of
financial statements for external purposes in accordance with generally accepted
accounting principles. Our system of internal control over financial reporting
includes those policies and procedures that (i) pertain to the maintenance of
records that, in reasonable detail, accurately and fairly reflect the
transactions and dispositions of the assets of the Company; (ii) provide
reasonable assurance that transactions are recorded as necessary to permit
preparation of financial statements in accordance with generally accepted
accounting principles, and that receipts and expenditures of the Company are
being made only in accordance with authorizations of management and directors of
the Company; and (iii) provide reasonable assurance regarding prevention or
timely detection of unauthorized acquisition, use or disposition of the
Company’s assets that could have a material effect on the financial
statements.
Management
performed an assessment of the effectiveness of our internal control over
financial reporting as of December 31, 2009 based upon criteria in Internal
Control — Integrated Framework issued by the Committee of Sponsoring
Organizations of the Treadway Commission (“COSO”). Based on this assessment,
management determined that the Company’s internal control over financial
reporting was effective as of December 31, 2009, based on the criteria in
Internal Control-Integrated Framework issued by COSO.
This
annual report does not include an attestation report of the Company’s registered
public accounting firm regarding internal control over financial reporting
pursuant to temporary rules of the Securities and Exchange Commission that
permit the Company to provide only management’s report in this annual
report.
This
report shall not be deemed “filed” for purposes of Section 18 of the Securities
Exchange Act of 1934, as amended, or otherwise subject to the liability of that
section. Further, this report shall not be deemed to be incorporated by
reference into any filing under the Securities Act of 1933, as amended, or the
Securities Exchange Act of 1934, as amended.
|
/s/
Francis M. DiNuzzo
|
/s/
Kevin J. Bratton
|
|||
|
Francis
M. DiNuzzo
|
Kevin
J. Bratton
|
|||
|
President
and Chief Executive Officer
|
Vice
President – Finance and Chief
|
|||
|
Financial
Officer
|
||||
Dated:
March 26, 2010
32
REPORT OF
INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
Board of
Directors and Stockholders
Strategic
Diagnostics Inc.:
We have
audited the accompanying consolidated balance sheets of Strategic Diagnostics
Inc. and subsidiaries as of December 31, 2009 and 2008, and the related
consolidated statements of operations, stockholders’ equity and comprehensive
income (loss) and cash flows for each of the years in the three-year period
ended December 31, 2009. These consolidated financial statements are the
responsibility of the Company’s management. Our responsibility is to express an
opinion on these consolidated financial statements based on our
audits.
We
conducted our audits in accordance with the standards of the Public Company
Accounting Oversight Board (United States). Those standards require that we plan
and perform the audit to obtain reasonable assurance about whether the financial
statements are free of material misstatement. An audit includes examining, on a
test basis, evidence supporting the amounts and disclosures in the financial
statements. An audit also includes assessing the accounting principles used and
significant estimates made by management, as well as evaluating the overall
financial statement presentation. We believe that our audits provide a
reasonable basis for our opinion.
In our
opinion, the consolidated financial statements referred to above present fairly,
in all material respects, the financial position of Strategic Diagnostics Inc.
and subsidiaries as of December 31, 2009 and 2008, and the results of their
operations and their cash flows for each of the years in the three-year period
ended December 31, 2009 in conformity with U.S. generally accepted accounting
principles.
/s/
KPMG LLP
Philadelphia,
Pennsylvania
March 26,
2010
33
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
CONSOLIDATED
BALANCE SHEETS
(in
thousands, except share and per share data)
|
December
31,
|
December
31,
|
|||||||
|
2009
|
2008
|
|||||||
|
ASSETS
|
||||||||
|
Current
Assets :
|
||||||||
|
Cash
and cash equivalents
|
$ | 7,937 | $ | 9,980 | ||||
|
Restricted
cash
|
1,250 | - | ||||||
|
Receivables,
net
|
3,650 | 4,099 | ||||||
|
Inventories
|
3,714 | 3,890 | ||||||
|
Deferred
tax asset
|
1 | 3 | ||||||
|
Other
current assets
|
551 | 534 | ||||||
|
Total
current assets
|
17,103 | 18,506 | ||||||
|
Property
and equipment, net
|
4,626 | 5,275 | ||||||
|
Other
assets
|
10 | 107 | ||||||
|
Deferred
tax asset
|
51 | 71 | ||||||
|
Intangible
assets, net
|
1,435 | 1,562 | ||||||
|
Total
assets
|
$ | 23,225 | $ | 25,521 | ||||
|
LIABILITIES
AND STOCKHOLDERS’ EQUITY
|
||||||||
|
Current
Liabilities :
|
||||||||
|
Current
portion of long-term debt
|
$ | 400 | $ | 1,658 | ||||
|
Accounts
payable
|
571 | 691 | ||||||
|
Accrued
expenses
|
1,386 | 1,860 | ||||||
|
Deferred
revenue
|
75 | 64 | ||||||
|
Total
current liabilities
|
2,432 | 4,273 | ||||||
|
Long-term
debt
|
700 | - | ||||||
|
Commitments
and Contingencies (Note 8)
|
||||||||
|
Stockholders’
Equity:
|
||||||||
|
Preferred stock, $.01 par value, 20,920,648 shares
authorized,
|
||||||||
|
no
shares issued or outstanding
|
- | - | ||||||
|
Common stock, $.01 par value, 35,000,000 shares
authorized,
|
||||||||
|
20,786,515
and 20,680,522 issued
|
||||||||
|
at
December 31, 2009 and December 31, 2008, respectively
|
208 | 206 | ||||||
|
Additional
paid-in capital
|
40,958 | 40,345 | ||||||
|
Treasury stock, 406,627 common shares at cost
|
||||||||
|
at
December 31, 2009 and December 31, 2008
|
(555 | ) | (555 | ) | ||||
|
Accumulated
deficit
|
(20,276 | ) | (18,625 | ) | ||||
|
Cumulative
translation adjustments
|
(242 | ) | (123 | ) | ||||
|
Total
stockholders’ equity
|
20,093 | 21,248 | ||||||
|
Total
liabilities and stockholders’ equity
|
$ | 23,225 | $ | 25,521 | ||||
The
accompanying notes are an integral part of these statements
34
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
CONSOLIDATED
STATEMENTS OF OPERATIONS
(in
thousands, except share and per share data)
|
Year
Ended December 31,
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
Revenues
|
$ | 27,154 | $ | 27,659 | $ | 27,207 | ||||||
|
Operating
expenses:
|
||||||||||||
|
Manufacturing
|
12,416 | 13,091 | 10,766 | |||||||||
|
Research
and development
|
2,894 | 3,576 | 2,938 | |||||||||
|
Selling,
general and administrative
|
13,593 | 14,425 | 11,990 | |||||||||
|
Goodwill
impairment
|
- | 4,150 | - | |||||||||
|
(Gain)
loss on disposal of assets
|
(1 | ) | (17 | ) | 110 | |||||||
|
Total
operating expenses
|
28,902 | 35,225 | 25,804 | |||||||||
|
Operating
income (loss)
|
(1,748 | ) | (7,566 | ) | 1,403 | |||||||
|
Interest
income (expense), net
|
(15 | ) | 157 | 433 | ||||||||
|
Income
(loss) before taxes
|
(1,763 | ) | (7,409 | ) | 1,836 | |||||||
|
Income
tax expense (benefit)
|
(112 | ) | 8,386 | 976 | ||||||||
|
Net
income (loss)
|
$ | (1,651 | ) | $ | (15,795 | ) | $ | 860 | ||||
|
Basic
net income (loss) per share
|
$ | (0.08 | ) | $ | (0.78 | ) | $ | 0.04 | ||||
|
Shares
used in computing basic net income (loss)
|
||||||||||||
|
per
share
|
20,113,659 | 20,312,707 | 20,325,285 | |||||||||
|
Diluted
net income (loss) per share
|
$ | (0.08 | ) | $ | (0.78 | ) | $ | 0.04 | ||||
|
Shares
used in computing diluted net income (loss)
|
||||||||||||
|
per
share
|
20,113,659 | 20,312,707 | 20,562,645 | |||||||||
The
accompanying notes are an integral part of these statements.
35
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
CONSOLIDATED
STATEMENTS OF STOCKHOLDERS’ EQUITY
AND
COMPREHENSIVE INCOME (LOSS)
(in
thousands)
|
Additional
|
Cumulative
|
|||||||||||||||||||||||||||
|
Preferred
|
Common
|
Paid-In
|
Treasury
|
Accumulated
|
Translation
|
|||||||||||||||||||||||
|
Stock
|
Stock
|
Capital
|
Stock
|
Deficit
|
Adjustments
|
Total
|
||||||||||||||||||||||
|
Balance
January 1, 2007
|
$ | - | $ | 202 | $ | 38,605 | $ | - | $ | (3,690 | ) | $ | 145 | $ | 35,262 | |||||||||||||
|
Comprehensive
income:
|
||||||||||||||||||||||||||||
|
Net
income
|
- | - | - | - | 860 | - | 860 | |||||||||||||||||||||
|
Currency
translation adjustment
|
- | - | - | - | - | 14 | 14 | |||||||||||||||||||||
|
Total
comprehensive income
|
874 | |||||||||||||||||||||||||||
|
Exercises
of stock options
|
- | 3 | 534 | - | - | - | 537 | |||||||||||||||||||||
|
Employee
stock purchase plan
|
- | - | 33 | - | - | - | 33 | |||||||||||||||||||||
|
Stock-based
compensation
|
- | - | 422 | - | - | - | 422 | |||||||||||||||||||||
|
Balance
December 31, 2007
|
$ | - | $ | 205 | $ | 39,594 | $ | - | $ | (2,830 | ) | $ | 159 | $ | 37,128 | |||||||||||||
|
Comprehensive
loss:
|
||||||||||||||||||||||||||||
|
Net
loss
|
- | - | - | - | (15,795 | ) | - | (15,795 | ) | |||||||||||||||||||
|
Currency
translation adjustment
|
- | - | - | - | - | (282 | ) | (282 | ) | |||||||||||||||||||
|
Total
comprehensive loss
|
(16,077 | ) | ||||||||||||||||||||||||||
|
Company
share buyback
|
- | - | - | (555 | ) | - | - | (555 | ) | |||||||||||||||||||
|
Employee
stock purchase plan
|
- | 1 | 69 | - | - | - | 70 | |||||||||||||||||||||
|
Stock-based
compensation
|
- | - | 682 | - | - | - | 682 | |||||||||||||||||||||
|
Balance
December 31, 2008
|
$ | - | $ | 206 | $ | 40,345 | $ | (555 | ) | $ | (18,625 | ) | $ | (123 | ) | $ | 21,248 | |||||||||||
|
Comprehensive
loss:
|
||||||||||||||||||||||||||||
|
Net
loss
|
- | - | - | - | (1,651 | ) | - | (1,651 | ) | |||||||||||||||||||
|
Currency
translation adjustment
|
- | - | - | - | - | (119 | ) | (119 | ) | |||||||||||||||||||
|
Total
comprehensive loss
|
(1,770 | ) | ||||||||||||||||||||||||||
|
Employee
stock purchase plan
|
- | 1 | 27 | - | - | - | 28 | |||||||||||||||||||||
|
Stock-based
compensation
|
- | 1 | 586 | - | - | - | 587 | |||||||||||||||||||||
|
Balance
December 31, 2009
|
$ | - | $ | 208 | $ | 40,958 | $ | (555 | ) | $ | (20,276 | ) | $ | (242 | ) | $ | 20,093 | |||||||||||
The
accompanying notes are an integral part of these statements.
36
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
CONSOLIDATED
STATEMENTS OF CASH FLOWS
(in
thousands)
|
Year
Ended December 31,
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
Cash
Flows from Operating Activities :
|
||||||||||||
|
Net
income (loss)
|
$ | (1,651 | ) | $ | (15,795 | ) | $ | 860 | ||||
|
Adjustments
to reconcile net income (loss) to net
|
||||||||||||
|
cash
provided by (used in) operating activities :
|
||||||||||||
|
Goodwill
impairment charge
|
- | 4,150 | - | |||||||||
|
Depreciation,
amortization and other intangibles impairment
|
1,277 | 1,401 | 1,206 | |||||||||
|
Stock-based
compensation expense
|
591 | 682 | 430 | |||||||||
|
Deferred
income taxes
|
22 | 8,566 | 769 | |||||||||
|
(Gain)
loss on disposal of fixed assets
|
(1 | ) | (17 | ) | 93 | |||||||
|
(Increase)
decrease in :
|
||||||||||||
|
Receivables
|
449 | 11 | (432 | ) | ||||||||
|
Inventories
|
176 | 314 | (1,026 | ) | ||||||||
|
Other
current assets
|
(17 | ) | (13 | ) | (29 | ) | ||||||
|
Other
assets
|
103 | (100 | ) | (14 | ) | |||||||
|
Increase
(decrease) in :
|
||||||||||||
|
Accounts
payable
|
(120 | ) | 122 | 32 | ||||||||
|
Accrued
expenses
|
(474 | ) | 1 | 411 | ||||||||
|
Deferred
revenue
|
11 | 59 | (128 | ) | ||||||||
|
Other
non-current liabilities
|
- | (130 | ) | 130 | ||||||||
|
Net
cash provided by (used in) operating activities
|
366 | (749 | ) | 2,302 | ||||||||
|
Cash
Flows from Investing Activities :
|
||||||||||||
|
Purchase
of property and equipment
|
(500 | ) | (929 | ) | (2,484 | ) | ||||||
|
Proceeds
from sale / disposal of assets
|
1 | 34 | 2 | |||||||||
|
Net
cash used in investing activities
|
(499 | ) | (895 | ) | (2,482 | ) | ||||||
|
Cash
Flows from Financing Activities :
|
||||||||||||
|
Proceeds
from exercise of stock options
|
- | - | 537 | |||||||||
|
Proceeds
from employee stock purchase plan
|
17 | 66 | 36 | |||||||||
|
Proceeds
from issuance of long and short term debt
|
- | - | 2,000 | |||||||||
|
Purchase
of treasury stock
|
- | (555 | ) | - | ||||||||
|
Restricted
cash requirement
|
(1,250 | ) | - | - | ||||||||
|
Repayments
on financing obligations
|
(558 | ) | (593 | ) | (311 | ) | ||||||
|
Net
cash provided by (used in) financing activities
|
(1,791 | ) | (1,082 | ) | 2,262 | |||||||
|
Effect
of exchange rate changes on cash
|
(119 | ) | (282 | ) | 14 | |||||||
|
Net
increase (decrease) in cash and cash equivalents
|
(2,043 | ) | (3,008 | ) | 2,096 | |||||||
|
Cash
and Cash Equivalents, Beginning of Year
|
9,980 | 12,988 | 10,892 | |||||||||
|
Cash
and Cash Equivalents, End of Year
|
$ | 7,937 | $ | 9,980 | $ | 12,988 | ||||||
|
Supplemental
Cash Flow Disclosure :
|
||||||||||||
|
Cash
paid (received) for taxes and tax refunds
|
$ | (54 | ) | $ | 11 | $ | 233 | |||||
|
Cash
paid for interest
|
84 | 119 | 69 | |||||||||
The
accompanying notes are an integral part of these statements
37
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER
31, 2009
(in
thousands, except share and per share data)
1.
SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES AND CERTAIN BALANCE SHEET
INFORMATION
Business
Strategic
Diagnostics Inc. and its subsidiaries (“SDIX” or the “Company”) is a
biotechnology company with a core mission of developing, commercializing and
marketing innovative and proprietary products, services and solutions that
preserve and enhance the quality of human health and wellness.
The
Company’s Life Science portfolio includes products and custom services that
supply critical reagents used across the life science research and development
markets. The Company’s Genomic Antibody Technology™ (“GAT™”) is
gaining wide adoption in proteomic research, disease understanding and
drug/biomarker discovery among academic, biotech, in-vitro diagnostic and large
pharmaceutical customers.
The
Company’s Kit products portfolio includes immunoassays, which represent advanced
technology for rapid, cost-effective detection of food pathogens as well as
water and soil contaminants. SDIX’s RapidChek® and
SELECT ™ kits are experiencing growing adoption for the detection of pathogens
such as E.
coli, Salmonella
and Listeria
in the production, processing and manufacturing of food and
beverages.
By
applying its core competencies of creating proprietary antibodies and assay
development, the Company has produced sophisticated testing and reagent systems
that are responsive to each customer’s analytical information
needs.
SDIX is a
customer-centric organization. The Company’s goals are to consistently deliver
increased value to its customers that facilitate their business results, reduce
costs and help in the management of risk. SDI sales professionals focus on
delivering a quantifiable “return on investment” to their customers by reducing
time and total costs associated with applications for which the Company’s
products are used. In addition, the Company believes its tests provide high
levels of accuracy and reliability, which deliver more actionable test results
to the customer as compared to alternative products. The Company is focused on
sustaining profitable growth by leveraging its expertise in antibodies and
immuno-technologies to successfully develop proprietary products and services
that enhance the competitive advantage of its customers.
The
Company believes that its competitive position has been enhanced through the
combination of talent, technology and resources resulting from the business
development activities it has pursued since its inception. The Company has
achieved meaningful economies of scale for the products it offers through the
utilization of its facilities in Newark, Delaware for the manufacture of test
kits and antibodies, and its facility in Windham, Maine for the manufacture of
antibodies.
The
continued economic downturn, including disruptions in the capital and credit
markets, may continue indefinitely and intensify, and could adversely affect our
results of operations, cash flows and financial condition or those of our
customers and suppliers. These circumstances could adversely affect our access
to liquidity needed to conduct or expand our business or conduct acquisitions or
make other discretionary investments. These circumstances may also adversely
impact the capital needs of our customers and suppliers, which, in turn, could
adversely affect their ability to purchase our products or supply us with
necessary equipment and raw materials. This could adversely affect our results
of operations, cash flows and financial condition. A weakening business climate
could cause longer sales cycles and slower growth, and could expose us to
increased business or credit risk in dealing with customers or suppliers
adversely affected by economic conditions. Our ability to collect accounts
receivable may be delayed or precluded if our customers are unable to pay their
obligations.
Basis
of Presentation
The
historical financial statements presented herein include the consolidated
financial statements of Strategic Diagnostics Inc. and its subsidiaries. All
significant intercompany balances and transactions have been eliminated in
consolidation.
Accounts
Receivable
As of
December 31, 2009 and 2008, the allowance for doubtful accounts was $38 and $70,
respectively. If receivables are in dispute with the customer or otherwise
deemed uncollectible, the Company’s policy is to charge these write-offs against
the allowance. The Company continually reviews the realizability of its
receivables and charges current period earnings for the amount deemed
unrealizable. At December 31, 2009 and 2008, net accounts receivable were $3,650
and $4,099, respectively.
38
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER
31, 2009
(in
thousands, except share and per share data)
A summary
of the activity in the allowance for doubtful accounts for the years ended
December 31, 2009, 2008 and 2007 is as follows:
|
2009
|
2008
|
2007
|
||||||||||
|
Balance,
January 1
|
$ | 70 | $ | 82 | $ | 134 | ||||||
|
Additions
(reductions) to allowance for doubtful accounts charged (credited) to
costs and expenses
|
12 | 70 | (49 | ) | ||||||||
|
Deductions-written
off as uncollectible
|
(44 | ) | (82 | ) | (3 | ) | ||||||
|
Balance,
December 31
|
$ | 38 | $ | 70 | $ | 82 | ||||||
Inventories
The
Company’s inventories are valued at the lower of cost or market. For
inventories that consist primarily of test kit components, bulk antibody serum
and antibody products, cost is determined using the first in, first out
method.
For
inventories that consist of costs associated with the production of custom
antibodies, cost is determined using the specific identification method.
Realization of such inventories is dependent upon the successful completion of a
project in accordance with customer specifications. Losses on projects in
progress are recorded in the period such losses become
probable.
At
December 31, inventories consisted of the following:
|
2009
|
2008
|
||||||||
|
Raw
materials
|
$ | 807 | $ | 1,216 | |||||
|
Work
in progress
|
1,228 | 946 | |||||||
|
Finished
goods
|
1,679 | 1,728 | |||||||
|
Inventories
|
$ | 3,714 | $ | 3,890 | |||||
Property
and Equipment
Property
and equipment are stated at cost. Depreciation and amortization are computed
using the straight-line method over the estimated useful lives (generally three
to five years) of the assets. Leasehold improvements are depreciated over the
shorter of the lease term or the estimated useful life.
Impairment
of Goodwill and Long-Lived Assets
Long-lived
assets, such as property and equipment, and purchased intangibles subject to
amortization, are reviewed for impairment whenever events or changes in
circumstances indicate that the carrying amount of an asset may not be
recoverable. Recoverability of assets to be held and used is measured by a
comparison of the carrying amount of an asset to estimated undiscounted future
cash flows expected to be generated by the asset. If the carrying amount of an
asset exceeds its estimated future cash flows, an impairment charge is
recognized in the amount by which the carrying amount of the asset exceeds the
fair value of the asset. Goodwill and intangible assets not subject to
amortization are tested annually for impairment, and are tested for impairment
more frequently if events and circumstances indicate that the asset might be
impaired. An impairment loss is recognized to the extent that the carrying
amount exceeds the asset’s fair value.
The
Company tests its goodwill annually in a two-step approach. The first step is
the comparison of the carrying value of the Company, including goodwill, to the
fair value of the Company at the testing date. If the carrying amount exceeds
the fair value of the Company, a second test is performed to measure the amount
of an impairment charge, if any. To measure the amount of any impairment charge,
the Company determines the implied fair value of goodwill in the same manner as
if the Company were being acquired in a business combination. See Note 3 for
additional information.
Revenue
Recognition
Revenues
composed of sales of immunoassay-based test kits and certain antibodies and
immunochemical reagents are recognized upon the shipment of the product and
transfer of title, or when related services are provided. Revenues associated
with such products or services are recognized when persuasive evidence of an
order exists, shipment of product has occurred or services have been provided,
the price is fixed or determinable and, collectability is reasonably assured.
Management is required to make judgments based on actual experience about
whether or not collectability is reasonably assured.
39
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER
31, 2009
(in
thousands, except share and per share data)
The
Company enters into contracts related to the production of custom antibodies,
which provide for the performance of defined tasks for a fixed price, with
delivery of the product upon completion of production. The standard time to
complete a project is typically longer than 30 days but less than 12 months and
effort is expended over the life of the project. Revenues related to sales of
custom antibody projects are recognized when a project’s specifications have
been met and/or the related materials have been shipped.
Fees
associated with products and services added on to a custom antibody project
subsequent to delivery of the initial project are billed monthly and recognized
as revenue as the services and other deliverables are provided.
Stock-Based
Compensation
The
Company accounts for stock-based compensation using the fair value method, which
requires that compensation costs related to employee share-based payment
transactions are measured in the financial statements at fair value on the date
of grant and are recognized over the vesting period of the award.
Research
and Development
Research
and development costs are charged to expense as incurred.
Accounting
for Income Taxes
Deferred
income tax assets and liabilities are determined based on the differences
between the financial statement and tax basis of assets and liabilities and are
measured using the enacted tax rates and laws that will be in effect when the
differences are expected to reverse. The measurement of deferred tax assets is
reduced, if necessary, by a valuation allowance for any tax benefits which are
not expected to be realized. The effect on deferred income tax assets and
liabilities of a change in tax rates is recognized in the period that such
changes are enacted.
The
Company utilizes a recognition threshold and measurement attribute for the
financial statement recognition and measurement of a tax position taken or
expected to be taken in a tax return. As a result of the
implementation of authoritative guidance on January 1, 2007 related to the
accounting for uncertainty in income taxes, the Company recognizes in
its financial statements the impact of a tax position if that position is more
likely than not of being sustained on audit, based on the technical merits of
the position. The authoritative guidance also provides guidance on
derecognition, classification, interest and penalties, accounting in interim
periods and disclosure. The Company includes interest and penalties related to
unrecognized tax benefits as a component of income tax expense. See Note 10 for
further information.
Basic
and Diluted Income (Loss) per Share
Basic
earnings (loss) per share (EPS) are computed by dividing net income or loss
available for common stockholders by the weighted-average number of common
shares outstanding during the period. Diluted EPS is similar to basic EPS,
except that the dilutive effect of converting or exercising all potentially
dilutive securities is also included in the denominator. The Company’s
calculation of diluted EPS includes the dilutive effect of exercising stock
options into common shares and the inclusion of unvested restricted stock
awards. Basic loss per share excludes potentially dilutive securities. For the
years 2009, 2008 and 2007, conversion of stock options and unvested restricted
shares totaling 208,679, 339,656 and 96,116 into common share equivalents were
excluded from this calculation because they were anti-dilutive.
Listed
below are the basic and diluted share calculations for the years ended December
31, 2009, 2008 and 2007:
|
2009
|
2008
|
2007
|
|||||||||||
|
Weighted
average common shares outstanding
|
20,113,659 | 20,312,707 | 20,325,285 | ||||||||||
|
Shares
used in computing basic net
|
|||||||||||||
|
income
(loss) per share
|
20,113,659 | 20,312,707 | 20,325,285 | ||||||||||
|
Dilutive
effect of stock options and
|
|||||||||||||
|
unvested
restricted stock awards
|
- | - | 237,360 | ||||||||||
|
Shares
used in computing diluted net
|
|||||||||||||
|
income
(loss) per share
|
20,113,659 | 20,312,707 | 20,562,645 | ||||||||||
40
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER
31, 2009
(in
thousands, except share and per share data)
Treasury
Stock
Shares of
common stock repurchased by the Company are recorded at cost as treasury stock
in the stockholders’ equity section of the consolidated balance sheet, and as a
use of cash in the financing activities section of the consolidated statement of
cash flows.
Foreign
Currency Translation
The
functional currency for the Company’s United Kingdom branch operation is the
British pound. Assets and liabilities related to this foreign operation are
translated at the current exchange rates at the end of each period. The
resulting translation adjustments are accumulated as a separate component of
stockholders’ equity. Revenues and expenses are translated at average exchange
rates in effect during the period with foreign currency transaction gains and
losses, if any, included in results of operations.
Comprehensive
Income (Loss)
Comprehensive
income (loss) is comprised of net income (loss) and currency translation
adjustments and is presented in the consolidated statements of stockholders’
equity.
Use
of Estimates
The
preparation of the consolidated financial statements requires the management of
the Company to make a number of estimates and assumptions relating to the
reported amount of assets and liabilities and disclosure of contingent assets
and liabilities at the date of the consolidated financial statements and the
reported amounts of revenues and expenses during the period. These estimates
include those made in connection with assessing the valuation of accounts
receivable, inventories, deferred tax assets, goodwill and long lived assets.
Actual results could differ from those estimates.
Statements
of Cash Flows
The
Company considers all highly liquid instruments purchased with an original
maturity of three months or less to be cash equivalents.
New
Accounting Pronouncements
In June
2009, the Financial Accounting Standards Board (“FASB”) issued SFAS
No. 168, The FASB Accounting Standards Codification and the Hierarchy of
Generally Accepted Accounting Principles (“SFAS 168”). SFAS 168 establishes the
FASB Accounting Standards Codification (“ASC”) as the single source of
authoritative nongovernmental GAAP. The ASC only changes the
referencing of financial accounting standards and does not change or alter
existing GAAP. The Codification is effective for interim and annual
periods ending after September 15, 2009. The adoption of this standard did
not have a material impact on the Company’s financial
statements. SFAS 168 has been incorporated into the ASC as ASC-105,
Generally Accepted Accounting Principles, or ASC 105.
In
December 2007, the FASB issued SFAS No. 141(Revised), Business
Combinations SFAS No. 141(R), which replaces SFAS No. 141, Business
Combinations, and requires an acquirer to recognize the assets acquired, the
liabilities assumed, and any non-controlling interest in the acquired at the
acquisition date, measured at their fair values as of that date, with limited
exceptions. This statement also requires the acquirer in a business combination
achieved in stages to recognize the identifiable assets and liabilities, as well
as the non-controlling interest in the acquired, at the full amounts of their
fair values. SFAS No. 141(R) makes various other amendments to
authoritative literature intended to provide additional guidance or to confirm
the guidance in that literature to that provided in this statement. This
statement applies prospectively to business combinations for which the
acquisition date is on or after the beginning of the first annual reporting
period beginning on or after December 15, 2008. SFAS
No. 141(R)’s impact on accounting for business combinations is dependent
upon future acquisitions the Company may make. SFAS 141(R) has been
incorporated into the ASC as ASC-805, Business Combinations (“ASC
805”).
In April
2008, the FASB issued FASB Staff Position No. FAS 142-3, Determination of the
Useful Life of Intangible Assets (“FSP 142-3”). FSP 142-3 amends the
factors an entity should consider in developing renewal or extension assumptions
used in determining the useful life of recognized intangible assets under SFAS
No. 142, Goodwill and Other Intangible Assets. This new guidance
applies prospectively to intangible assets that are acquired individually or
with a group of other assets in business combinations and asset
acquisitions. FSP 142-3 is effective for financial statements issued
for fiscal years and interim periods
beginning after December 15, 2008. FSP 142-3’s impact on the Company
is dependent upon future acquisitions the Company may make. FSP 142-3 has been
incorporated into the ASC as ASC-350, Intangibles (“ASC 350”).
41
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER
31, 2009
(in
thousands, except share and per share data)
In
September 2009, the FASB issued Accounting Standard Update (“ASU”)
No. 2009-13,
Revenue Recognition (Topic 605) – Multi-Deliverables Revenue
Arrangements, a Consensus of the FASB Emerging Issues Task Force, to address the
accounting for multiple-deliverable arrangements to enable vendors to account
for products or services (deliverables) separately rather than as a combined
unit. It establishes the accounting and reporting guidance for arrangements
under which the vendor will perform multiple revenue-generating activities,
specifically, how to separate deliverables and how to measure and allocate
arrangement consideration to one or more units of accounting. The update will be
effective prospectively for revenue arrangements entered into or materially
modified in fiscal years beginning on or after June 15, 2010. The Company
is currently evaluating the impact, if any, of adopting the update.
2.
PROPERTY AND EQUIPMENT
As of
December 31, property and equipment consisted of the following:
|
2009
|
2008
|
||||||||
|
Equipment
|
$ | 6,595 | $ | 6,256 | |||||
|
Building
improvements
|
4,105 | 4,088 | |||||||
|
Furniture
and fixtures
|
188 | 175 | |||||||
|
Land
|
1,452 | 1,380 | |||||||
|
Leasehold
improvements
|
972 | 904 | |||||||
|
Total
property and equipment
|
13,312 | 12,803 | |||||||
|
Less
- accumulated depreciation and amortization
|
(8,686 | ) | (7,528 | ) | |||||
|
Net
property and equipment
|
$ | 4,626 | $ | 5,275 | |||||
Depreciation
expense was $1,150, $1,114 and $966 in 2009, 2008 and 2007,
respectively.
3.
INTANGIBLE ASSETS
As of
December 31, intangible assets consisted of the following:
|
2009
|
2008
|
Lives
|
|||||||||||
|
Intangible
assets
|
$ | 2,614 | $ | 3,085 | 2-20 | ||||||||
|
Less
- accumulated amortization
|
(1,179 | ) | (1,523 | ) | |||||||||
|
Net
intangible assets
|
$ | 1,435 | $ | 1,562 | |||||||||
In the
fourth quarter of 2008, as economic conditions continued to decline, the Company
experienced a sustained and significant decline in its stock price. As a result
of this decline, the Company’s market capitalization was significantly below the
book value of the Company’s stockholders’ equity at December 31, 2008. The
Company performed an impairment test and the resulting fair value of goodwill
was zero. Accordingly, the Company recorded a charge of $4,150 to write off the
entire balance of goodwill during the year ended December 31,
2008.
The
remaining intangible assets principally relate to intellectual and property
rights acquired from Molecular Circuitry Inc. (“MCI”). The technology acquired
from MCI primarily relates to proprietary growth media used by the Company in
conjunction with the Company’s E.
coli and Salmonella
test kits, and also technology used in the Company’s ruminant feed test
kit. The Company launched sales of these products during the year ended December
31, 2003 and expects a continued revenue stream from the sale of these
products.
A former
director and current large shareholder of the Company was a majority shareholder
of MCI. MCI will continue to receive royalties from the Company until July of
2012. Royalties paid to MCI in 2009, 2008 and 2007 were $167, $166 and $172,
respectively.
Amortization
of these intangible assets is on a straight line basis over their useful lives
and was $127, $237 and $240 in 2009, 2008 and 2007, respectively. The following
table is a schedule of the expected amortization expense in each of the next
five years:
42
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER
31, 2009
(in
thousands, except share and per share data)
|
Year
|
Amount
|
||||
|
2010
|
$ | 114 | |||
|
2011
|
114 | ||||
|
2012
|
114 | ||||
|
2013
|
114 | ||||
|
2014
|
114 | ||||
4. ACCRUED EXPENSES
As of
December 31, accrued expenses consisted of the following:
|
2009
|
2008
|
||||||||
|
Royalties
|
$ | 88 | $ | 143 | |||||
|
Compensation
|
798 | 1,245 | |||||||
|
Professional
fees
|
256 | 303 | |||||||
|
Purchases
|
244 | 169 | |||||||
|
Total
accrued expenses
|
$ | 1,386 | $ | 1,860 | |||||
5.
LONG-TERM DEBT
On May 5,
2000, the Company entered into a financing agreement with a commercial bank
which was amended on August 12, 2009 (as amended, the “Credit
Agreement”).
On August
21, 2007, the Company received a term loan under the Credit Agreement to finance
the construction of new facilities at its Windham, Maine location. This
agreement provided for up to $2,000 in financing, $1,100 of which was
outstanding at December 31, 2009, and is repayable over five years, with
principal payments that began on October 1, 2007. The loan bears a fixed
interest rate of 5.96% with equal amortization of principal payments plus
interest.
This
indebtedness is secured by $1,250 in restricted cash as required by the
Credit Agreement.
The
following table is a schedule of the principal payments required under the
Company’s long-term indebtedness:
|
2010
|
$ | 400 | |||
|
2011
|
400 | ||||
|
2012
|
300 | ||||
|
Total
debt
|
$ | 1,100 | |||
Interest
expense was $82, $117 and $78 in 2009, 2008 and 2007, respectively.
6. SHARE-BASED
COMPENSATION
Under
various plans, executives, key employees and outside directors receive awards of
options to purchase common stock. The Company has a stock option plan (the “2000
Plan”) which authorizes the granting of incentive and nonqualified stock options
and restricted stock awards. Incentive stock options are granted at not less
than 100% of fair market value at the date of grant (110% for stockholders
owning more than 10% of the Company’s common stock). Nonqualified stock options
are granted at not less than 85% of fair market value at the date of grant. A
maximum of 6,000,000 shares of common stock are issuable under the 2000 Plan.
Certain additional options have been granted outside the 2000 Plan. These
options generally follow the provisions of the 2000 Plan. The Company issues new
shares to satisfy option exercises and the vesting of restricted stock
awards.
The
Company also has an Employee Stock Purchase Plan (the “ESPP”). The ESPP allows
eligible full-time employees to purchase shares of common stock at 90 percent of
the lower of the fair market value of a share of common stock on the first or
last day of the quarter. Eligible employees are provided the opportunity to
acquire Company common stock during each quarter. No more than 661,157 shares of
common stock may be issued under the ESPP. Such stock may be unissued shares or
treasury shares of the Company or may be outstanding shares purchased in the
open market or otherwise on behalf of the ESPP. For financial reporting
purposes, the Company’s ESPP is compensatory. Therefore, the Company is required
to recognize compensation expense related to the discount from market value of
shares sold under the ESPP.
43
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER
31, 2009
(in
thousands, except share and per share data)
On
December 27, 2005, the Board of Directors of the Company approved the
accelerated vesting, effective as of December
31, 2005, of all unvested stock options granted to employees and non-employee
directors from 2002 through 2005 under the 2000 Plan, as well as options granted
to the Company’s former Chief Executive Officer under his original employment
agreement. The acceleration of vesting of these options reduced non-cash
compensation expense that would have been recorded in the Company’s statements
of operations in 2006, 2007 and 2008, in anticipation of the adoption of the
fair value method of accounting for stock-based compensation in January 2006.
The Board’s decision to accelerate the vesting was based on such factors as: 48%
of the options were “out of the money”; the options generally vested over the
next three years; to reduce compensation expense that would be recorded in
future periods following the adoption of the fair value method and, the Company
decided to rely, to a substantial degree, on restricted stock as opposed to
options in future incentive compensation awards.
As a
result of the acceleration, options to purchase approximately 381,000 shares of
the Company’s common stock (which represented 23% of the Company’s outstanding
stock options) became exercisable on December 31, 2005. The accelerated options
ranged in exercise price from $2.51 to $4.12 per share and the weighted average
exercise price of the accelerated options was $3.41 per share. Of the 381,000
shares that became exercisable, approximately 199,000 of these shares were “in
the money” as of December 27, 2005, meaning that the exercise price was at or
below the price of the Company’s common stock on that date. The weighted average
exercise price of the “in the money” shares on that date was $3.02. The options
subject to acceleration included options to purchase approximately 222,000
shares held in the aggregate by executive officers and approximately 48,000
shares held in the aggregate by non-employee directors of the Company. Of these
270,000 shares, approximately 147,000 were “in the money” as of December 27,
2005. The grant date fair value of the unvested portion of accelerated options
at December 31, 2005 totaled $975,000 and would have been recognized as
compensation expense in accordance with FASB ASC Topic 718 – Stock Compensation,
in future years as follows: $682,000 in 2006, $228,000 in 2007 and $65,000 in
2008.
Share-based
compensation expense recorded in 2009, 2008 and 2007 is summarized as
follows:
|
2009
|
2008
|
2007
|
||||||||||
|
Stock
options
|
$ | 307 | $ | 384 | $ | 249 | ||||||
|
Employee
stock purchase plan
|
4 | 7 | 8 | |||||||||
|
Restricted
stock awards
|
280 | 291 | 173 | |||||||||
|
Total
share-based compensation expense
|
$ | 591 | $ | 682 | $ | 430 | ||||||
The
deferred income tax benefit related to share-based compensation expense for the
years ended December 31, 2009 and 2008 was $0 due to the full valuation
allowance recorded against deferred tax assets (see Note 10). The deferred
income tax benefit for the year ended December 31, 2007 was
$120. Share-based compensation expense is a component of selling,
general and administrative expense, and is recorded as a non-cash expense in the
operating activities section of the consolidated statement of cash
flows.
44
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER
31, 2009
(in
thousands, except share and per share data)
Information
with respect to the stock options granted under the 2000 Plan and options
granted separately from the 2000 Plan is summarized as follows:
|
Weighted
|
Aggregate
|
|||||||||||||||||
|
Number
|
Average
Remaining
|
Instrinsic
|
||||||||||||||||
|
of
Shares
|
Price
Range
|
Contractual
term
|
Value
|
|||||||||||||||
|
Balance,
January 1, 2007
|
1,469,729 | $ | 1.88 | - | $ | 6.94 | ||||||||||||
|
Granted
|
180,575 | $ | 3.46 | - | $ | 4.65 | ||||||||||||
|
Cancelled
/ forefeited
|
(224,284 | ) | $ | 2.50 | - | $ | 6.94 | |||||||||||
|
Exercised
|
(174,400 | ) | $ | 1.88 | - | $ | 4.20 | |||||||||||
|
Balance,
December 31, 2007
|
1,251,620 | $ | 1.88 | - | $ | 6.94 | ||||||||||||
|
Granted
|
574,420 | $ | 1.50 | - | $ | 4.60 | ||||||||||||
|
Cancelled
/ forfeited
|
(106,068 | ) | $ | 3.05 | - | $ | 5.50 | |||||||||||
|
Balance,
December 31, 2008
|
1,719,972 | $ | 1.88 | - | $ | 6.94 | ||||||||||||
|
Granted
|
483,000 | $ | 1.10 | - | $ | 1.50 | ||||||||||||
|
Cancelled
/ forfeited
|
(501,172 | ) | $ | 2.88 | - | $ | 4.69 | |||||||||||
|
Balance,
December 31, 2009
|
1,701,800 | $ | 1.10 | - | $ | 6.94 |
6.1
years
|
$ | 4 | |||||||||
|
Vested
and excercisable at
|
||||||||||||||||||
|
December
31, 2009
|
924,150 | $ | 1.50 | - | $ | 6.94 |
4.7
years
|
$ | - | |||||||||
|
Expected
to vest as of
|
||||||||||||||||||
|
December
31, 2009
|
1,630,903 | $ | 1.10 | - | $ | 6.94 |
6.5
years
|
$ | 4 | |||||||||
As of
December 31, 2009, options covering 924,150 shares were exercisable with a
weighted average exercise price of $3.75 per share, and 1,906,731 shares were
available for future grant under the 2000 Plan.
As of
December 31, 2009 there was $446 of unrecognized compensation expense related to
non-vested stock options that is expected to be recognized over a weighted
average period of 3.3 years.
The total
aggregate intrinsic value of options exercised during the years ended December
31, 2009, 2008 and 2007 was $0, $0 and $257 respectively. Cash received from the
exercises during the years ended December 31, 2009, 2008 and 2007 was $0, $0 and
$537 and are included within the financing activity section of the consolidated
statements of cash flows.
The
weighted average fair value at the date of grant for options granted during
2009, 2008 and 2007 was estimated at $0.62, $1.39 and $2.55 per share,
respectively, using the Black-Scholes pricing model. The weighted-average
assumptions used in the Black-Scholes model were as follows: dividend yield of
0%, expected volatility of 51% in 2009, 49% in 2008 and 59% in 2007, risk-free
interest rate of 2.50% in 2009, 3.20% in 2008 and 4.25% in 2007, and an expected
option life of 6 years in 2009, 6 years in 2008, and 5.5 years in 2007. The
expected option life was computed using the sum of the average vesting period
and the contractual life of the option and dividing by 2, for all periods
presented.
45
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER
31, 2009
(in
thousands, except share and per share data)
The
following table provides additional information about the Company’s stock
options outstanding at December 31, 2009:
|
Options
Outstanding
|
Options
Exercisable
|
|||||||||||||||||||||||||
|
Weighted
Average
|
Wtd.
Average
Exercise
Price
|
|||||||||||||||||||||||||
| Range of Exercise Prices |
Number
of
Shares
|
Remaining Contractual
Life |
Exercise Price |
Number
of Shares |
||||||||||||||||||||||
| $ | 1.10 | - | $ | 2.61 | 846,700 | 8.6 |
Years
|
$ | 1.57 | 158,150 | $ | 1.90 | ||||||||||||||
| $ | 3.05 | - | $ | 3.57 | 206,400 | 3.6 |
Years
|
$ | 3.32 | 206,400 | $ | 3.32 | ||||||||||||||
| $ | 3.69 | - | $ | 6.94 | 648,700 | 5.0 |
Years
|
$ | 4.17 | 559,600 | $ | 4.14 | ||||||||||||||
| $ | 1.50 | - | $ | 6.94 | 1,701,800 | 6.1 |
Years
|
$ | 3.41 | 924,150 | $ | 3.75 | ||||||||||||||
The
Company grants restricted stock awards (RSA) which is the right to receive
shares. The fair value of RSAs is based on the market price for the stock at the
date of grant. RSAs generally vest over periods of two to five
years.
The
following table summarizes the changes in non-vested restricted stock units for
the three year period ending December 31, 2009:
|
Weighted
Average
|
||||||||||||
|
Grant
Date
|
Aggregate
|
|||||||||||
|
Shares
|
Fair
Value
|
Intrinsic
Value
|
||||||||||
|
Non-vested
RSAs at January 1, 2007
|
85,222 | $ | 3.71 | |||||||||
|
Granted
|
70,193 | $ | 4.61 | |||||||||
|
Vested
|
(43,549 | ) | $ | 3.54 | ||||||||
|
Cancelled
/ forfeited
|
(15,750 | ) | $ | 4.00 | ||||||||
|
Non-vested
RSAs at December 31, 2007
|
96,116 | $ | 4.36 | |||||||||
|
Granted
|
266,619 | $ | 1.99 | |||||||||
|
Vested
|
(52,117 | ) | $ | 4.02 | ||||||||
|
Cancelled
/ forfeited
|
(14,212 | ) | $ | 4.71 | ||||||||
|
Non-vested
RSAs at December 31, 2008
|
296,406 | $ | 2.28 | |||||||||
|
Granted
|
64,500 | $ | 1.26 | |||||||||
|
Vested
|
(142,133 | ) | $ | 2.26 | ||||||||
|
Cancelled
/ forfeited
|
(12,747 | ) | $ | 3.78 | ||||||||
|
Non-vested
RSAs at December 31, 2009
|
206,026 | $ | 1.77 | $ | 280 | |||||||
|
Expected
to vest at December 31, 2009
|
196,111 | $ | 1.72 | $ | 267 | |||||||
The
Company recorded compensation expense of $280, $291 and $173 for the years ended
December 31, 2009, 2008 and 2007, for RSAs. This expense is a component of
selling, general and administrative expenses, and is recorded as a non-cash
expense in the operating activities section of the consolidated statement of
cash flows. As of December 31, 2009, 196,111 of the above non-vested RSAs are
expected to vest and there is approximately $256 of unrecognized compensation
expense related to non-vested RSAs that are expected to vest over a weighted
average of 2.1 years.
46
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER
31, 2009
(in
thousands, except share and per share data)
7.
SEGMENT, GEOGRAPHIC AND CUSTOMER INFORMATION
The
Company develops and manufactures antibodies in its life sciences division. Such
antibodies are incorporated into test kits manufactured by the Company’s
industrial BioDetection (Kit products) services division for the detection of a
wide variety of substances related to food safety and water quality. In
addition, antibodies from the life sciences division are sold to medical
diagnostic and pharmaceutical companies and research institutions. The Company
does not provide segment disclosures as discrete financial information is not
prepared for the Life Sciences or BioDetection division for review by the
Company’s Chief Operating Decision Maker.
Geographic:
The
following table sets forth revenues by geographic region:
|
Year
Ended December 31,
|
|||||||||||||
|
2009
|
2008
|
2007
|
|||||||||||
|
United
States
|
$ | 19,739 | $ | 20,744 | $ | 21,154 | |||||||
|
Rest
of the world
|
7,415 | 6,915 | 6,053 | ||||||||||
|
Total
|
$ | 27,154 | $ | 27,659 | $ | 27,207 | |||||||
The
Company’s basis for identifying sales by country is the ship-to location. There
were no individual countries outside of the United States that represented more
than 10% of the total revenues of the Company. There are no significant
long-lived assets located outside the United States.
Products
and Services:
The
following table sets out revenues by product category.
|
Year
Ended December 31,
|
|||||||||||||
|
2009
|
2008
|
2007
|
|||||||||||
|
Life
science
|
$ | 14,321 | $ | 13,821 | $ | 14,281 | |||||||
|
Kit
products
|
12,738 | 13,634 | 12,770 | ||||||||||
|
Contract
revenue
|
95 | 204 | 156 | ||||||||||
|
Revenues
|
$ | 27,154 | $ | 27,659 | $ | 27,207 | |||||||
47
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER
31, 2009
(in
thousands, except share and per share data)
8.
COMMITMENTS AND CONTINGENCIES
The
Company leases its office and manufacturing facilities and other equipment under
operating leases. Rent expense for the years ended December 31, 2009, 2008 and
2007, was $1,157, $991 and $843, respectively. Future commitments under
non-cancelable leases at December 31, 2009 are as follows:
|
2010
|
$ | 1,169 | |||
|
2011
|
845 | ||||
|
2012
|
330 | ||||
|
2013
|
300 | ||||
|
2014
|
300 | ||||
|
2015
and thereafter
|
600 | ||||
| $ | 3,544 |
The
Company’s subsidiary, AZUR Environmental Limited, is the lessee for two real
property leases located in the United Kingdom. In 2001, the landlord of the two
properties gave AZUR Environmental Limited its consent to allow AZUR to assign
the lease and its related obligations to a third party. As inducement to the
landlord to grant the assignment, AZUR was required to guarantee performance
under the original lease terms if the third party fails to perform. Both lease
terms expire in November 2016 and provide for annual principal rent payments of
approximately $300 per year in the aggregate and these amounts have been
included in the table above.
The
Company is subject to various claims arising in the ordinary course of business.
Although the ultimate outcome of these matters is presently not determinable,
management does not believe that the outcome of these matters will have a
material adverse effect on the Company’s financial position or results of
operations.
9.
RETIREMENT SAVINGS PLAN
The
Company maintains a retirement savings plan qualified under Section 401(k) of
the Internal Revenue Code. The plan allows for eligible employees to contribute
a portion of their gross wages to the plan. The Company matches employees’
contributions on a 100% basis up to 1% of gross wages and on a 50% basis up to
the next 5% of gross wages. In 2009, 2008 and 2007, the Company recognized
expense of $245, $242 and $172, respectively, associated with this
plan.
10.
INCOME TAXES
The
components of income (loss) before tax expense as of December 31 are as
follows:
|
2009
|
2008
|
2007
|
||||||||||
|
United
States
|
$ | (1,900 | ) | $ | (7,587 | ) | $ | 1,673 | ||||
|
Rest
of the world
|
137 | 178 | 163 | |||||||||
|
Total
|
$ | (1,763 | ) | $ | (7,409 | ) | $ | 1,836 | ||||
The
income tax expense (benefit) consists of the following:
|
2009
|
2008
|
2007
|
||||||||||||
|
Federal
|
current
|
$ | (147 | ) | $ | (20 | ) | $ | 95 | |||||
|
deferred
|
7,249 | 710 | ||||||||||||
| (147 | ) | 7,229 | 805 | |||||||||||
|
State
|
current
|
7 | (135 | ) | 63 | |||||||||
|
deferred
|
- | 1,242 | 59 | |||||||||||
| 7 | 1,107 | 122 | ||||||||||||
|
Foreign
|
current
|
- | - | - | ||||||||||
|
deferred
|
28 | 50 | 49 | |||||||||||
| 28 | 50 | 49 | ||||||||||||
|
Total
|
$ | (112 | ) | $ | 8,386 | $ | 976 | |||||||
48
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER
31, 2009
(in
thousands, except share and per share data)
The
following table summarizes the significant differences between the U.S. Federal
statutory rate and the Company’s effective tax rate for financial statement
purposes:
|
2009
|
2008
|
2007
|
||||||||||
|
Statutory
tax rate
|
34.0 | % | 34.0 | % | 34.0 | % | ||||||
|
State
taxes, net of federal benefit
|
(0.3 | ) | (9.9 | ) | 4.4 | |||||||
|
Valuation
allowance, federal
|
(22.7 | ) | (135.8 | ) | - | |||||||
|
Goodwill
impairment
|
- | (2.5 | ) | - | ||||||||
|
Provision
for uncertain tax positions
|
- | - | 10.4 | |||||||||
|
Additional
tax for reconciliation to return
|
- | - | 6.2 | |||||||||
|
Research
and development credits
|
(0.4 | ) | 1.6 | (6.7 | ) | |||||||
|
Other,
net
|
(4.3 | ) | (0.6 | ) | 4.9 | |||||||
|
Total
|
6.3 | % | (113.2 | %) | 53.2 | % | ||||||
Significant
components of the Company’s deferred tax assets as of December 31 are as
follows:
|
2009
|
2008
|
|||||||
|
Net
operating loss carryforwards
|
$ | 7,856 | $ | 6,875 | ||||
|
Credit
carryforwards
|
884 | 1,048 | ||||||
|
Amortization
and depreciation
|
3,647 | 4,033 | ||||||
|
Deferred
compensation
|
240 | 188 | ||||||
|
Non-deductible
reserves
|
141 | 69 | ||||||
|
Inventory
costs not currently deductible
|
323 | 299 | ||||||
|
Total
deferred tax assets
|
13,091 | 12,512 | ||||||
|
Valuation
allowance
|
(13,039 | ) | (12,438 | ) | ||||
|
Net
deferred tax assets
|
$ | 52 | $ | 74 | ||||
For the
year ended December 31, 2009, the Company recorded income tax benefit of $112,
which included a $601 increase in the valuation allowance recorded against
deferred tax assets of $13,091. The current federal income tax
benefit is primarily the result of changes in federal tax law that provided the
ability to realize research and experimentation credits and alternative minimum
tax credits that previously had a valuation allowance provided against them. The
increase in the valuation allowance is primarily due to the continuation of the
conditions that led to the conclusion that a full valuation allowance against
U.S. federal and state deferred tax assets was required
FASB ASC
740, Accounting for Income Taxes (“FASB ASC 740”), requires a company to
evaluate its deferred tax assets on a regular basis to determine if a valuation
allowance against the net deferred tax assets is required. Pursuant to FASB ASC
740, a cumulative pre-tax loss in recent years is significant negative evidence
that is difficult to overcome in considering whether deferred tax assets are
more likely than not realizable. The Company has evaluated the possibility of
potential tax planning strategies and determined that none currently exist that
the Company would conclude are prudent and feasible. The Company has concluded,
based upon the evaluation of all available evidence, that it is more likely than
not that the U.S. federal and state net deferred tax assets will not be realized
and has recorded a full valuation allowance on its U.S. federal and state net
deferred tax assets, as of December 31, 2009.
At
December 31, 2009, the Company had approximately $4,905 of net operating loss
carryforwards for tax purposes which have no expiration related to operations in
the United Kingdom (“UK”). Management considered positive and negative
indicators, as well as potential tax planning strategies, and has concluded that
a substantial valuation allowance of approximately $1,321 was necessary for the
foreign deferred tax assets of approximately $1,373. The positive indicators
include the contribution to income before taxes by the foreign operations in the
UK for 2009 and 2008, and the forecasted income before taxes in the UK for 2010
and 2011. The negative indicators include a history of substantial net operating
losses in the UK, the lack of income before taxes prior to 2004, limited income
before taxes in the recent years and limitations with regard to estimating
income in the UK beyond 2011 resulting from a year-to-year evaluation of the
future need for a UK subsidiary.
49
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER
31, 2009
(in
thousands, except share and per share data)
At
December 31, 2009, the Company had U.S. federal net operating loss carryforwards
of $17,380 including those of acquired companies, which will expire as
follows:
|
Year
|
Net
Operating
Loss (in
thousands)
|
||||
|
2010
|
$ | 4,536 | |||
|
2017
|
760 | ||||
|
2018
|
1,327 | ||||
|
2019
|
550 | ||||
|
2020
|
66 | ||||
|
2021
|
56 | ||||
|
2022
|
2,268 | ||||
|
2024
|
2,033 | ||||
|
2025
|
3 | ||||
|
2026
|
1 | ||||
|
2027
|
1 | ||||
|
2028
|
3,492 | ||||
|
2029
|
2,287 | ||||
|
Total
|
$ | 17,380 | |||
The Tax
Reform Act of 1986 (the “Act”) limits the annual use of net operating loss and
income tax credit carryforwards (after certain ownership changes, as defined by
the Act). The application of these limits could significantly restrict the
Company’s ability to utilize these carryforwards. Certain of the Company’s total
net operating loss carryforwards from 2001 and prior years are subject to
limitations on their annual use since a cumulative change in ownership of more
than 50% has occurred within a three-year period with respect to those net
operating loss carryforwards. The Company continues to evaluate recent changes
in ownership. If it is determined that an ownership change of more than 50%
within a three-year period did occur, as determined pursuant to the Internal
Revenue Code and Regulations, substantially all the net operating loss
carryforwards and income tax credit carryforwards could be subject to annual
limitations on usage. Because U.S. tax laws limit the time period during which
these carryforwards may be applied against future taxable income, the Company
may not be able to take full advantage of these attributes for federal and state
income tax purposes due to the annual limitation usage.
The
Company has federal research and experimentation credit carryforwards of $730 as
of December 31, 2009, which are set to expire in years 2019 through
2029. The Company also has federal alternative minimum tax credit
carryforwards of $11 which have indefinite lives. In accordance with Internal
Revenue Code §168(k)(4)) the Company intends to elect out of bonus depreciation
on the filing of its 2009 federal income tax return which will result in a
federal tax refund related to certain of the research and experimentation
credits and the alternative minimum tax credits. Accordingly, the
research and experimentation credits and alternative minimum tax credits were
reduced in the current year by $31 and $1, respectively.
For the
year ended December 31, 2009, the Company increased its unrecognized tax
benefits increased by $23 (net of decreases).
50
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER
31, 2009
(in
thousands, except share and per share data)
The
following table is a reconciliation of the gross unrecognized tax benefits
during the years ended December 31:
|
2009
|
2008
|
2007
|
||||||||||
|
Gross
unrecognized tax benefits as of January 1
|
$ | 487 | $ | 580 | $ | 362 | ||||||
|
Increases
from positions taken in prior periods
|
- | - | 128 | |||||||||
|
Decreases
from positions taken in prior periods
|
(5 | ) | (130 | ) | - | |||||||
|
Increases
from positions taken in current period
|
28 | 37 | 92 | |||||||||
|
Decreases
from unrecognized tax benefits for settlements
|
||||||||||||
|
with
taxing authorities
|
- | - | (2 | ) | ||||||||
|
Gross
unrecognized tax benefits as of December 31
|
$ | 510 | $ | 487 | $ | 580 | ||||||
The
unrecognized tax benefits at December 31, 2009 of $510, if recognized in a
period where there was not a full valuation allowance, would affect the
effective tax rate.
The
Company is subject to U.S. federal and UK income tax, as well as income taxes of
multiple state jurisdictions.
The
Company recognizes accrued interest expense and penalties related to uncertain
tax benefits in income tax expense. The Company had no payments or
accruals of interest or penalties that were required to be recognized for
unrecognized tax benefits during the year ended December 31,
2009. The Company had no payments or accruals of interest or
penalties for the years ended December 31, 2008 and 2007, due to the available
net operating loss carryforwards. Additionally there was no interest or
penalties required to be recognized in the statement of operations during the
year ended December 31, 2009 and no interest or penalties required to be
recognized in the statement of operations during the years ended December 31,
2008 and 2007.
For
federal purposes, post-1992 tax years remain open to examination as a result of
net operating loss carryforwards. For state purposes, the statute of
limitations remains open in a similar manner for states that have generated net
operating losses. The Company does not expect that the total amount of
unrecognized tax benefits related to positions taken in prior periods will
change significantly during the next twelve months.
51
STRATEGIC
DIAGNOSTICS INC. AND SUBSIDIARIES
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER
31, 2009
(in
thousands, except share and per share data)
12.
QUARTERLY FINANCIAL DATA (unaudited)
|
Three
Months Ended,
|
||||||||||||||||
|
March
31
|
June
30
|
September
30
|
December
31
|
|||||||||||||
|
(In
thousands except per share data)
|
||||||||||||||||
|
2009
|
||||||||||||||||
|
Revenues
|
$ | 6,902 | $ | 6,853 | $ | 7,196 | $ | 6,203 | ||||||||
|
Gross
profit (1)
|
3,805 | 3,630 | 4,113 | 3,190 | ||||||||||||
|
Net
loss
|
(561 | ) | (734 | ) | (291 | ) | (65 | ) | ||||||||
|
Basic
loss per share
|
(0.03 | ) | (0.04 | ) | (0.01 | ) | - | |||||||||
|
Diluted
loss per share
|
(0.03 | ) | (0.04 | ) | (0.01 | ) | - | |||||||||
|
2008
|
||||||||||||||||
|
Revenues
|
$ | 7,155 | $ | 6,584 | $ | 6,892 | $ | 7,028 | ||||||||
|
Gross
profit (1)
|
4,033 | 3,400 | 3,337 | 3,798 | ||||||||||||
|
Net
loss
|
(12 | ) | (1,081 | ) | (2,218 | ) | (12,484 | ) | ||||||||
|
Basic
loss per share
|
- | (0.05 | ) | (0.11 | ) | (0.62 | ) | |||||||||
|
Diluted
loss per share
|
- | (0.05 | ) | (0.11 | ) | (0.62 | ) | |||||||||
|
(1) Gross
profit is revenues less manufacturing expenses.
|
||||||||||||||||
52
SIGNATURES
Pursuant
to the requirements of Section 13 or 15(d) of the Securities Exchange Act of
1934, the Registrant has duly caused this report to be signed on its behalf by
the undersigned, thereunto duly authorized.
|
STRATEGIC
DIAGNOSTICS INC.
|
|||
|
Date:
|
March
26, 2010
|
/s/
Francis M. DiNuzzo
|
|
|
Francis
M. DiNuzzo
|
|||
|
President,
Chief Executive Officer and
Director
|
|||
Pursuant
to the requirements of the Securities Exchange Act of 1934, this report has been
signed below by the following persons on behalf of the Registrant and in the
capacities and on the dates indicated.
|
Date:
|
March
26, 2010
|
/s/
Richard van den Broek
|
|
|
Richard
van den Broek
|
|||
|
Lead
Director
|
|||
|
Date:
|
March
26, 2010
|
/s/
Francis M. DiNuzzo
|
|
|
Francis
M. DiNuzzo
|
|||
|
President,
Chief Executive Officer and Director
|
|||
|
(Principal
Executive Officer)
|
|||
|
Date:
|
March
26, 2010
|
/s/
Kevin J. Bratton
|
|
|
Kevin
J. Bratton
|
|||
|
Vice
President – Finance and Chief Financial Officer
|
|||
|
(Principal
Financial and Accounting Officer)
|
|||
|
Date:
|
March
26, 2010
|
/s/
Steven Becker
|
|
|
Steven
Becker
|
|||
|
Director
|
|||
|
Date:
|
March
26, 2010
|
/s/
Thomas A. Bologna
|
|
|
Thomas
A. Bologna
|
|||
|
Director
|
|||
|
Date:
|
March
26, 2010
|
/s/
Geoffrey Davis
|
|
|
Geoffrey
Davis
|
|||
|
Director
|
|||
|
Date:
|
March
26, 2010
|
/s/
Clifford L. Spiro
|
|
|
Clifford
L. Spiro
|
|||
|
Director
|
|||
|
Date:
|
March
26, 2010
|
/s/
Stephen L. Waechter
|
|
|
Stephen
L. Waechter
|
|||
|
Director
|
|||
|
Date:
|
March
26, 2010
|
/s/
David M. Wurzer
|
|
|
David
M. Wurzer
|
|||
|
Director
|
|||
53
