Attached files
| file | filename |
|---|---|
| 8-K - FORM 8-K - ANGIODYNAMICS INC | an56071525-8ki.htm |
Exhibit 99.1

04 | 12 - 15 | 2021 ANGIODYNAMICS The 20th Annual Needham Virtual Healthcare ConferenceJim Clemmer,
President & CEOStephen Trowbridge, EVP & CFO

2 Forward-Looking Statement Notice Regarding Forward-Looking StatementsThis presentation contains
forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements regarding AngioDynamics’ expected future financial position, results of operations, cash flows, business strategy, budgets,
projected costs, capital expenditures, products, competitive positions, growth opportunities, plans and objectives of management for future operations, as well as statements that include the words such as “expects,” “reaffirms,” “intends,”
“anticipates,” “plans,” “projects,” “believes,” “seeks,” “estimates,” “optimistic,” or variations of such words and similar expressions, are forward-looking statements. These forward-looking statements are not guarantees of future performance
and are subject to risks and uncertainties. Investors are cautioned that actual events or results may differ materially from AngioDynamics’ expectations, expressed or implied. Factors that may affect the actual results achieved by AngioDynamics
include, without limitation, the scale and scope of the COVID-19 global pandemic, the ability of AngioDynamics to develop its existing and new products, technological advances and patents attained by competitors, infringement of AngioDynamics’
technology or assertions that AngioDynamics’ technology infringes the technology of third parties, the ability of AngioDynamics to effectively compete against competitors that have substantially greater resources, future actions by the FDA or
other regulatory agencies, domestic and foreign health care reforms and government regulations, results of pending or future clinical trials, overall economic conditions, the results of on-going litigation, challenges with respect to
third-party distributors or joint venture partners or collaborators, the results of sales efforts, the effects of product recalls and product liability claims, changes in key personnel, the ability of AngioDynamics to execute on strategic
initiatives, the effects of economic, credit and capital market conditions, general market conditions, market acceptance, foreign currency exchange rate fluctuations, the effects on pricing from group purchasing organizations and competition,
the ability of AngioDynamics to integrate acquired businesses, as well as the risk factors listed from time to time in AngioDynamics’ SEC filings, including but not limited to its Annual Report on Form 10-K for the year ended May 31, 2020.
AngioDynamics does not assume any obligation to publicly update or revise any forward-looking statements for any reason.In the United States, the NanoKnife System has received a 510(k) clearance by the Food and Drug Administration for use in
the surgical ablation of soft tissue, and is similarly approved for commercialization in Canada, the European Union and Australia. The NanoKnife System has not been cleared for the treatment or therapy of a specific disease or condition.Notice
Regarding Non-GAAP Financial MeasuresManagement uses non-GAAP measures to establish operational goals and believes that non-GAAP measures may assist investors in analyzing the underlying trends in AngioDynamics’ business over time. Investors
should consider these non-GAAP measures in addition to, not as a substitute for or as superior to, financial reporting measures prepared in accordance with GAAP. In this presentation, AngioDynamics has reported adjusted EBITDA (income before
interest, taxes, depreciation and amortization and stock-based compensation); adjusted net income and adjusted earnings per share. Management uses these measures in its internal analysis and review of operational performance. Management
believes that these measures provide investors with useful information in comparing AngioDynamics’ performance over different periods. By using these non-GAAP measures, management believes that investors get a better picture of the performance
of AngioDynamics’ underlying business. Management encourages investors to review AngioDynamics’ financial results prepared in accordance with GAAP to understand AngioDynamics’ performance taking into account all relevant factors, including
those that may only occur from time to time but have a material impact on AngioDynamics’ financial results. Please see the tables that follow for a reconciliation of non-GAAP measures to measures prepared in accordance with GAAP.

PURSUIT OF LARGER MARKETSActive portfolio management enables us to compete in larger, growing markets
relying on technology & innovation to produce measurable patient outcomes FOCUSED RESOURCE DEVELOPMENTResource deployment is focused in areas that offer best opportunities for success PORTFOLIO TRANSFORMATIONPortfolio
transformation & strength is driven by R&D, M&A, and Clinical & Regulatory TOP TALENT Portfolio combined with talent drives value ANGIODYNAMICSStrategic Transformation 3

4 FOCUSED TRANSFORMATIONU.S. Total Addressable Markets Our strategic initiative to become a growth
company begins 2018 Launch of the Auryon System gives us access to the peripheral atherectomy market New Thrombectomy & NanoKnife System portfolio additions & indications increase market access $1.3B 0-3% Mkt
CAGR $3.2B 2-5%Mkt CAGR $7.8B 3-7%Mkt CAGR 2021 2025 The planned portfolio additions are not guarantees of future performance and are subject to risks and uncertainties. Investors are cautioned that actual events or results may
differ from AngioDynamics’ expectations.

5 Diagnostic Catheters, Guidewires and Kits Vascular Access Catheters and Accessories Microwave &
Radiofrequency Tumor Ablation Endovenous Laser Treatment Lung Biopsy Safety Radiation Treatment Stabilization Balloons INVEST FOR GROWTH MAINTAIN POSITIONING Thrombus Management Irreversible Electroporation Peripheral Atherectomy

6 FOCUSED INVESTMENT Healthy Blood Flow to the Heart Expanded Treatment Options in
Oncology Leveraging 3 main drivers to carve out our space in large, growing markets through innovative, disruptive technologies that treat patients with cancer, promote healthy blood flow and deliver critical therapies. Healthy Blood Flow
from the Heart M & A R & D Clinical and Regulatory Pathway Expansion

PERIPHERAL ATHERECTOMY 04 | 12 - 15 | 2021 7

2.35 mmAspiration and Off-Center capabilities and indicated for Peripheral Atherectomy and In-Stent
Restenosis (ISR) 2.0 mmAspiration capability and indicated for Peripheral Atherectomy and ISR 1.5 mmIndicated for Peripheral Atherectomy Why wavelength mattersEach type of tissue interacts differently with a given wavelength Why pulse width
and amplitude matterGreater amplitude is achieved with shorter pulses, which can deposit energy before thermal diffusion occurs The Auryon System produces a photon energy of 3.5 eV, which is low enough to be nonreactive to vessel
endothelium, but high enough to vaporize calcium. b, c The Auryon System has a pulse width of 10 to 25 ns, ensuring enough power to target the lesion and spare the vessel. a 0.9 mmIndicated for Peripheral Atherectomy 8 a. Auryon.
Instructions for use. AngioDynamics; 2019. b. Herzog A, Bogdan S, Glikson M, Ishaaya AA, Love C. Selective tissue ablation using laser radiation at 355 nm in lead extraction by a hybrid catheter; a preliminary report. Lasers Surg Med.
2016;48(3):281-287. c. Spectranetics Corporation. CVX-300 Excimer Laser System: Operator’s Manual. Version 28. 2019:1-56.

PERIPHERAL
ATHERECTOMYUS Addressable Markets & Competitive Landscape MECHANICAL LASER $555M 23%1 1. Peripheral Vascular Devices Medtech 360 Market Analysis US 2017. (2016, December). Millennium Research Group, Inc. 77%1 2020 Total Addressable
Market (TAM) 9 Company Product Thrombus Plaque Fibrotic ISR Calcified ANGO Auryon Philips Excimer Laser MDT TurboHawk BSC Jetstream CSI Diamonback
360

10 PERIPHERAL ATHERECTOMYContinuing our momentum of growth PRODUCT DEVELOPMENT Product development
pipelinePortfolio and product indication expansionContinuous voice of customer CLINICAL COMPENDIUMExpand awareness by creating a strong podium and clinical publication presence Clinically differentiated validation through new users, data
collection (PATHFINDER I & II), and clinical outcomes COMMERCIAL SCALETargeted commercial & clinical expansionMeaningful physician training programsIncrease physician ambassador programs

THROMBUS MANAGEMENT 04 | 12 - 15 | 2021 11

12 Deep Vein ThrombosisDVT + Pulmonary EmbolismPE = Venous ThromboembolismVTE A blood clot that
forms in a deep vein, usually the leg, groin or arm A DVT breaks free from a vein wall and travels to the lungs blocking some or all of the blood supply DVT and PE are collectively referred to as VTE 208,000 Iliofemoral Cases1 171,000
High-risk & intermediate-risk PE Cases1 100,000 – 300,000 VTE-Related Deaths in the USA Annually2 Stages of Clot Clot in Transit (traveling through the heart) Clot in Pulmonary Arteries (PE) 1. Plovanic, W. J., &
Furlong, C. (2020, June). Inari Medical Biomedical Devices and Services. Canaccord Genuity Capital Markets.2. “Venous Thromboembolism (VTE).” World Thrombosis Day, www.worldthrombosisday.org/issue/vte. Illustrations and Images not Produced by
AngioDynamics Include:https://www.vascularmedcure.com/disease-backgroundDVT (Blood Clot In the Leg): 7 Warning Signs and Symptoms (emedicinehealth.com)

13 DVT & PE TREATMENT OPTIONSPercutaneous Thrombectomy CDT & US-CDTCatheter-Directed
Thrombolysis (with or without the assistance of ultrasound) PCMTPharmacomechanical Thrombectomy AngioVacContinuous Aspiration Suction Thrombectomy MTMechanical Retrieval Thrombectomy MAMechanical Aspiration(Small & Large) Lytic
Based Non-Lytic Based Rx AnticoagulationPrescription Medication SIMPLE COMPLEX MODERATE Treatment “Type” Spectrum Illustrations and Images not Produced by AngioDynamics Include:https://youtu.be/0QUUBZ6BxSo
https://www.penumbrainc.com/indigo-lightning/ https://www.vascularmedcure.com/ourproducts
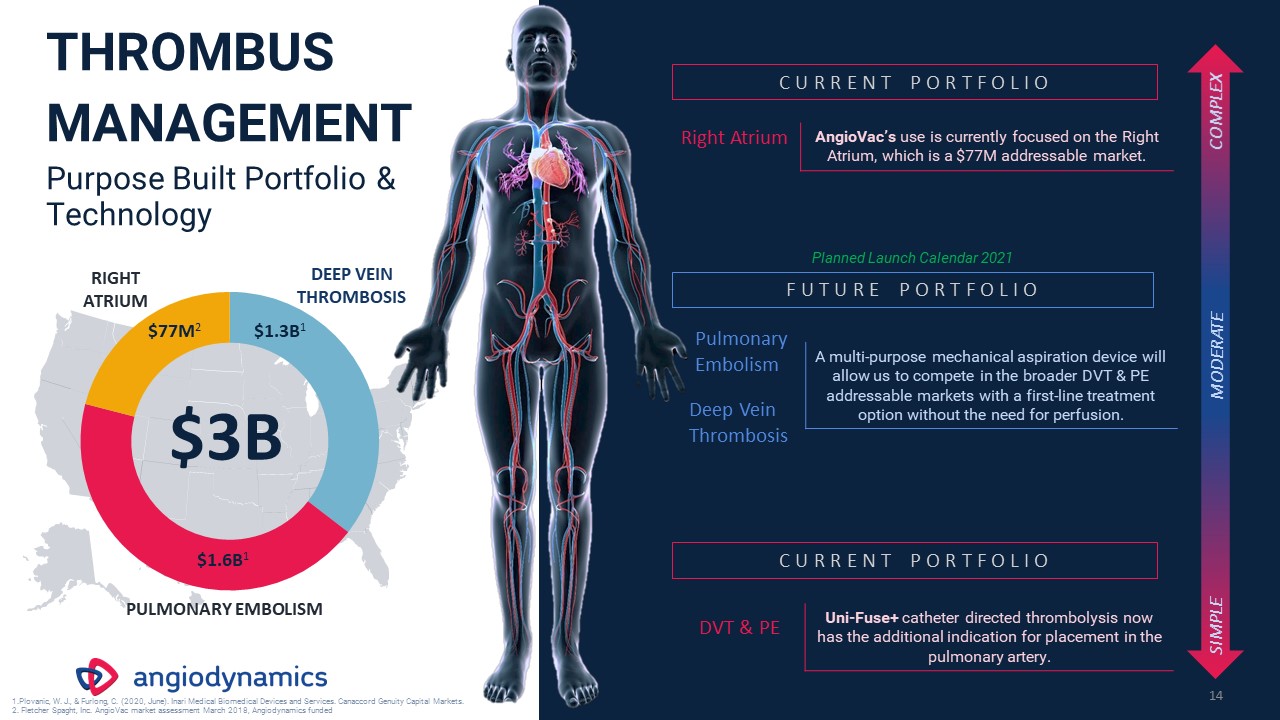
THROMBUS
MANAGEMENTPurpose Built Portfolio & Technology 14 Right Atrium AngioVac’s use is currently focused on the Right Atrium, which is a $77M addressable market. Pulmonary Embolism Deep Vein Thrombosis A multi-purpose mechanical aspiration
device will allow us to compete in the broader DVT & PE addressable markets with a first-line treatment option without the need for perfusion. C U R R E N T P O R T F O L I O F U T U R E P O R T F O L I O DVT & PE Uni-Fuse+
catheter directed thrombolysis now has the additional indication for placement in the pulmonary artery. C U R R E N T P O R T F O L I O SIMPLE COMPLEX MODERATE $3B $77M2 $1.3B1 $1.6B1 PULMONARY EMBOLISM RIGHT ATRIUM DEEP VEIN
THROMBOSIS Plovanic, W. J., & Furlong, C. (2020, June). Inari Medical Biomedical Devices and Services. Canaccord Genuity Capital Markets. Fletcher Spaght, Inc. AngioVac market assessment March 2018, Angiodynamics funded Planned Launch
Calendar 2021

The Difference The AngioVac System allows for the continuous aspiration of embolic material such as
fresh, soft thrombi or vegetation from the venous systemUtilizing a self-expanding, nitinol reinforced funnel tipSimultaneously reinfusing the patient’s own filtered blood to limit procedural blood loss Centrifugal Pump Console Waste
Collection System Filter Saline AngioVac Cannula Reinfusion Cannula AngioVac Circuit Individual experience may not be indicative of all procedure results. 15

THE NEXT GENERATION OF ANGIOVACPhysician requests for use in DVT drive new product
development $1.6B Controlled Powerful Versatile THE NEXT PORTFOLIO INNOVATION A purpose-built, innovative product leveraging the strengths of the AngioVac cannula technology with off-circuit manual aspiration control Proven, funnel tip
design allows efficient aspiration and compression of large clot burden Designed to allow the end-user command and control of the mechanical aspiration Broadens our Thrombus Management portfolio and designed to provide an intuitive,
first-line treatment option without the need for lytics and advanced procedural support 16 Not approved by the Food and Drug Administration (FDA). These statements and the subject product have not been evaluated by the Food and Drug
Administration (FDA). This product is not intended to diagnose, treat, cure, or prevent any disease. The device is not currently being marketed, nor is it available for sale in any country

THROMBUS MANAGEMENTPlanned Portfolio Additions & U.S. Addressable Markets
Expansion 17 $1.3B FY 2021 FY 2022 FY 2023 FY 2025 FY 2024 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 $140M $700M $3.6B $1.5B 1 2 The planned portfolio additions are not guarantees of future
performance and are subject to risks and uncertainties. Investors are cautioned that actual events or results may differ from AngioDynamics’ expectations. (22 Fr)Right Atrium, Illio-caval DVT (22 & 18 Fr)Entry into Ilio-femoral DVT
(13 Fr)Access to full Ilio-femoral DVT market (18 Fr) Smaller size should drive further adoption in addressable market PE Indication Purpose Built, Comprehensive, Thrombus Portfolio AV3 AV4 C20 C85 C120 Continuous Aspiration with
Simultaneous Reinfusion Multi-purpose Mechanical/Manual Aspiration AV3 AV4 C20 C85 C120 Catheter Directed Thrombolysis with PE Indication

IRREVERSIBLE ELECTROPORATION 04 | 12 - 15 | 2021 18

19 Probe placement Placement of probes around a target area provides
effective treatment coverage a-d Electrical pulses cause pores to form within the cellular membrane leading to non-thermal cell death a-d Electrical field Decellularization Non-thermal Revascularization The NanoKnife System’s unique
technology enables physicians to provide localized treatments in locations and at times previously unavailable to them a-d By preserving those underlying structures the potential for revascularization of treated tissue is maintained
a-d Target tissue undergoes complete decellularization while preserving the underlying structure of blood vessels, nerves, ducts, and tissue a-d IRREVERSIBLE ELECTROPORATIONNon-thermal energy destroys cells while preserving critical
structures a. Maor E. et al., The effect of irreversible electroporation on blood vessels, Technol. Cancer Res. Treat. 6(4), 307–312 (2007).10.1177/153303460700600407.b. Rubinsky, B., Onik, G., and Mikus, P., Irreversible electroporation: a
new ablation modality–clinical implications. Technol. Cancer Res. Treat. 6, 37–48. (2007). doi:10.1177/153303460700600106.c. Onik G., Mikus P., and Rubinsky B., Irreversible electroporation: implications for prostate ablation, Technol. Cancer
Res. Treat. 6(4), 295–300(2007).10.1177/153303460700600405.d. Lee EW, Chen C, Prieto VE, Dry SM, Loh CT, Kee ST., Advanced hepatic ablation technique for creating complete cell death: irreversible electroporation. Radiology 255:426–433. (2010).
doi:1148/radiol.10090337

20 Localized Treatment Strong safety profile Maintains potential for future
treatments Provides options for patients PROVIDES THE INNOVATION DOCTORS NEEDExpands treatment options and helps preserve patient's quality of life Spares critical structures and nerves Treated tissue can revascularize Preserves patient's
quality of life Non-thermal energy decellularizes target tissue d W J a-d a-d a-d a-d a. Maor E. et al., The effect of irreversible electroporation on blood vessels, Technol. Cancer Res. Treat. 6(4), 307–312
(2007).10.1177/153303460700600407.b. Rubinsky, B., Onik, G., and Mikus, P., Irreversible electroporation: a new ablation modality–clinical implications. Technol. Cancer Res. Treat. 6, 37–48. (2007). doi:10.1177/153303460700600106.c. Onik G.,
Mikus P., and Rubinsky B., Irreversible electroporation: implications for prostate ablation, Technol. Cancer Res. Treat. 6(4), 295–300(2007).10.1177/153303460700600405.d. Lee EW, Chen C, Prieto VE, Dry SM, Loh CT, Kee ST., Advanced hepatic
ablation technique for creating complete cell death: irreversible electroporation. Radiology 255:426–433. (2010). doi:1148/radiol.10090337 a-d

21 DIRECT StudyThis study is evaluating the use of the NanoKnife System as a potential treatment for
stage III pancreatic cancer. Prostate IDEThis safety study will evaluate the use of the NanoKnife System as a focal therapy option for prostate tissue. Liver Oral
Cavity Pancreas Kidney Bladder Melanoma Colorectal Prostate Lung Breast New Cancer Cases 2020: 1,256,970 22% 18% 15% 12% 8% 6% 6% 5% 4% 3% a. Cancer Facts & Figures 2020. American Cancer Society.
https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2020.html. Accessed January 7, 2021. a Illustrations and Images not Produced by AngioDynamics
Include:https://seenamagowitzfoundation.org/pancreas-transplants/https://www.youtube.com/watch?v=9LPYb87Gjgk&feature=emb_title
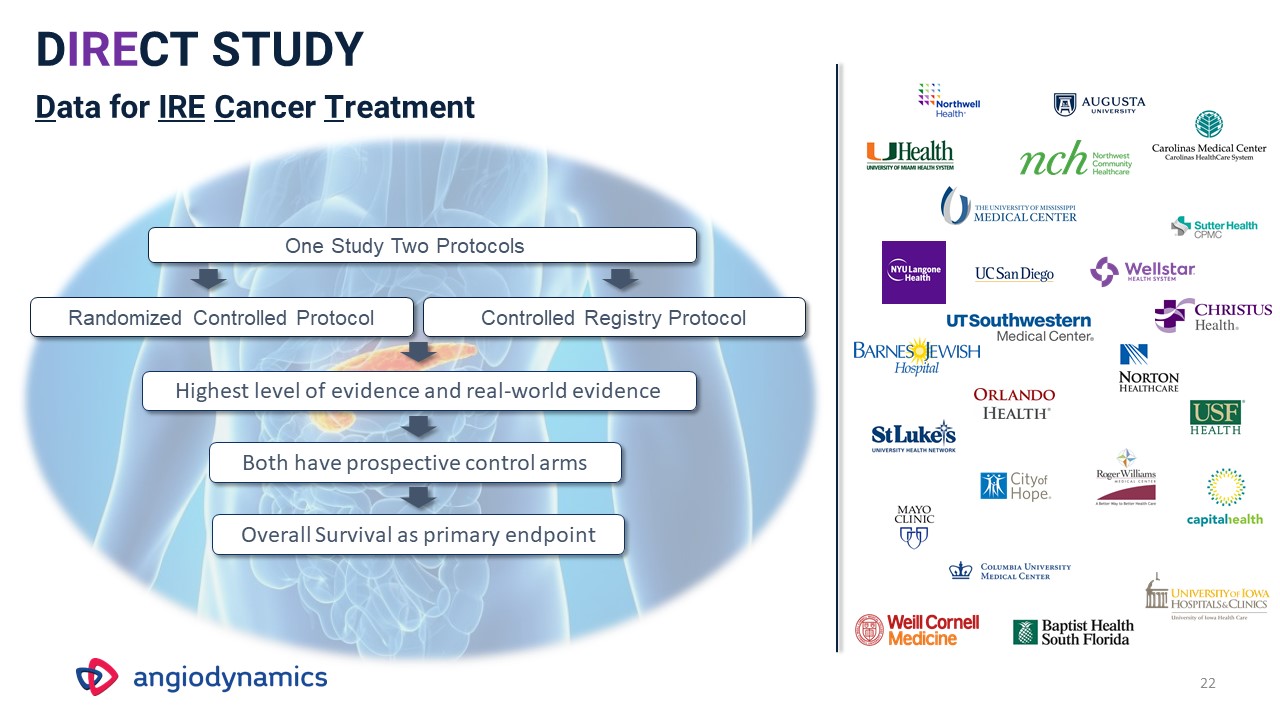
22 DIRECT STUDYData for IRE Cancer Treatment Randomized Controlled Protocol Controlled Registry
Protocol One Study Two Protocols Highest level of evidence and real-world evidence Overall Survival as primary endpoint Both have prospective control arms
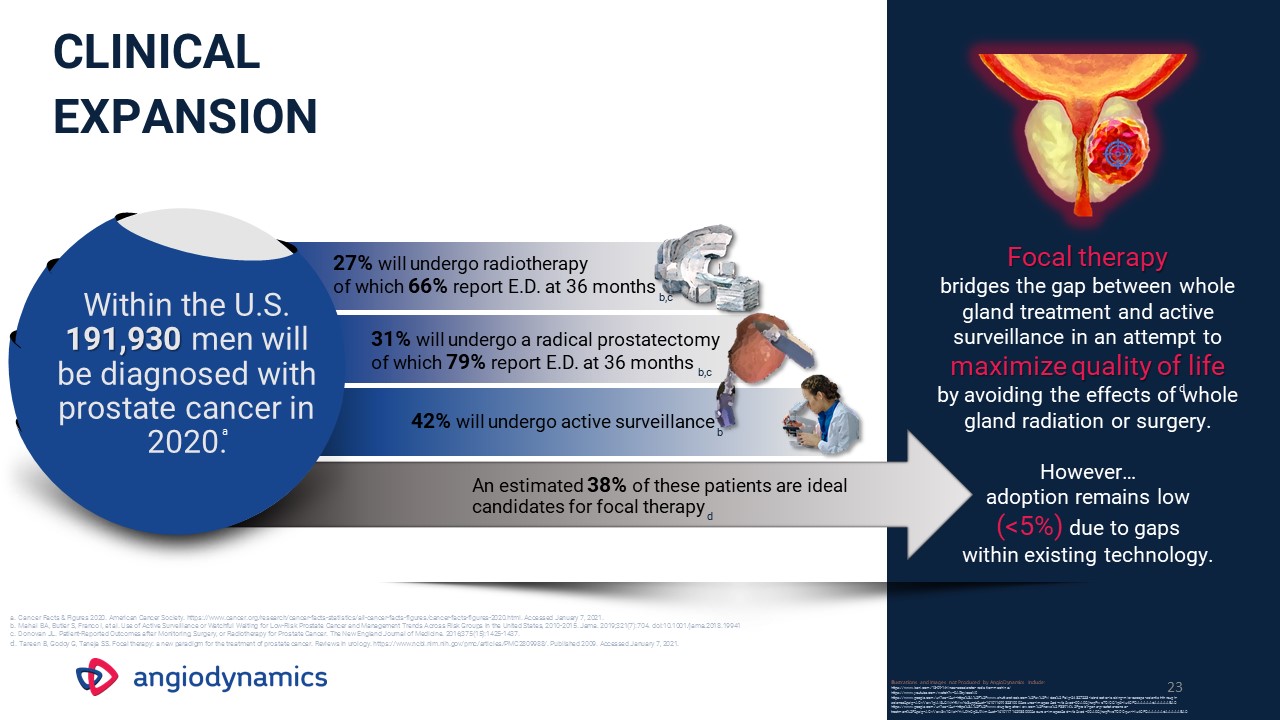
23 Focal therapy bridges the gap between whole gland treatment and active surveillance in an attempt to
maximize quality of life by avoiding the effects of whole gland radiation or surgery.However…adoption remains low (<5%) due to gaps within existing technology. 27% will undergo radiotherapy of which 66% report
E.D. at 36 months Within the U.S. 191,930 men will be diagnosed with prostate cancer in 2020. 31% will undergo a radical prostatectomy of which 79% report E.D. at 36 months 42% will undergo active surveillance An estimated 38% of these
patients are ideal candidates for focal therapy Clinical Expansion a. Cancer Facts & Figures 2020. American Cancer Society. https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2020.html.
Accessed January 7, 2021. b. Mahal BA, Butler S, Franco I, et al. Use of Active Surveillance or Watchful Waiting for Low-Risk Prostate Cancer and Management Trends Across Risk Groups in the United States, 2010-2015. Jama. 2019;321(7):704.
doi:10.1001/jama.2018.19941c. Donovan JL. Patient-Reported Outcomes after Monitoring, Surgery, or Radiotherapy for Prostate Cancer. The New England Journal of Medicine. 2016;375(15):1425-1437. d. Tareen B, Godoy G, Taneja SS. Focal therapy: a
new paradigm for the treatment of prostate cancer. Reviews in urology. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2809988/. Published 2009. Accessed January 7, 2021. a b,c b,c b d d Illustrations and Images not Produced by
AngioDynamics
Include:https://www.keni.com/18-09-14-linear-accelerator-radiation-machine/https://www.youtube.com/watch?v=0ARkyIcacN0https://www.google.com/url?sa=i&url=https%3A%2F%2Fwww.shutterstock.com%2Fsv%2Fvideo%2Fclip-24827228-lab-doctor-looking-microscope-scientist-through-science&psig=AOvVaw1giAl5LQINhYtMwYx8upqb&ust=1610116993281000&source=images&cd=vfe&ved=0CA0QjhxqFwoTCICQ1q6Hiu4CFQAAAAAdAAAAABADhttps://www.google.com/url?sa=i&url=https%3A%2F%2Fwww.drugtargetreview.com%2Fnews%2F38914%2Fgreb1-gene-prostate-cancer-treatment%2F&psig=AOvVaw3w1QlisHYviL0fnOq2L9Nm&ust=1610117162985000&source=images&cd=vfe&ved=0CA0QjhxqFwoTCPCquv-Hiu4CFQAAAAAdAAAAABAD

24 The Team Expanding Indications Pancreas pivotal study underway (DIRECT)Prostate
safety study underwayFDA Pre-Sub meeting regarding prostate tissue indication complete InpatientICD-10 Code (2018)DRGs (2019)Organ Agnostic CPT Code (2020)OutpatientOutpatient (2021)ASC (2021) ExpandedReimbursement NANOKNIFE
PLATFORM Current Indications Surgical ablation of soft tissue Clinical & Regulatory R&D Sales

VASCULAR ACCESS 04 | 12 - 15 | 2021 25

26 VASCULAR ACCESSSafely delivering medication to patients PORTFOLIO MARKET
ACCESS PERFORMANCE PICCs Midlines Tip Location Ports Dialysis Point of Care Ultrasound Delivering on our product road map including development of an integrated navigation & tip location technology that enables the use of our BioFlo
PICC. Maximize clinical differentiation by reducing thrombus accumulation through the utilization of our BioFlo family of catheters Maintain a strong culture of execution and collaboration through disciplined sales & marketing
plans CONNECT / CAPTURE / CONFIRM
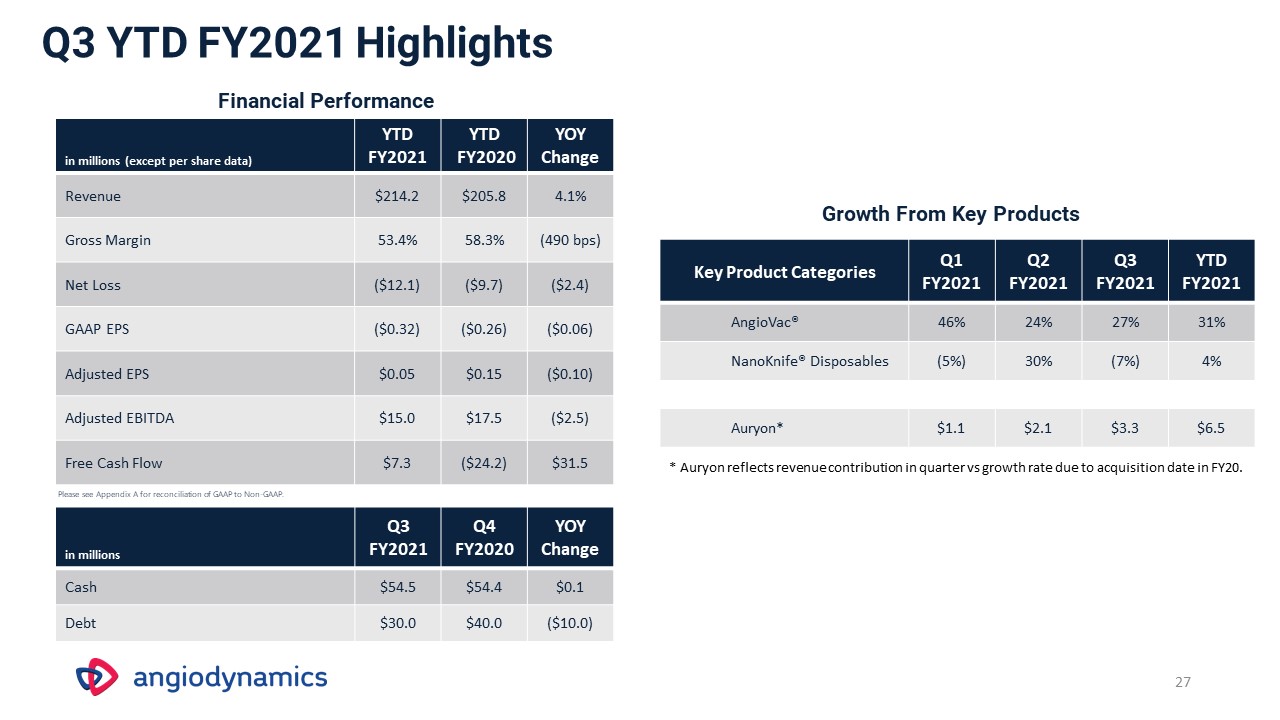
Q3 YTD FY2021 Highlights in millions (except per share data) YTD FY2021 YTD FY2020 YOY
Change Revenue $214.2 $205.8 4.1% Gross Margin 53.4% 58.3% (490 bps) Net Loss ($12.1) ($9.7) ($2.4) GAAP EPS ($0.32) ($0.26) ($0.06) Adjusted EPS $0.05 $0.15 ($0.10) Adjusted EBITDA $15.0 $17.5 ($2.5) Free Cash
Flow $7.3 ($24.2) $31.5 Financial Performance Key Product Categories Q1 FY2021 Q2 FY2021 Q3FY2021 YTD FY2021 AngioVac® 46% 24% 27% 31% NanoKnife® Disposables (5%) 30% (7%) 4% Auryon* $1.1 $2.1 $3.3 $6.5 Growth
From Key Products * Auryon reflects revenue contribution in quarter vs growth rate due to acquisition date in FY20. in millions Q3 FY2021 Q4 FY2020 YOY Change Cash $54.5 $54.4 $0.1 Debt $30.0 $40.0 ($10.0) 27 Please see Appendix
A for reconciliation of GAAP to Non-GAAP.

PURSUIT OF LARGER MARKETSActive portfolio management enables us to compete in larger, growing markets
relying on technology & innovation to produce measurable patient outcomes FOCUSED RESOURCE DEVELOPMENTResource deployment is focused in areas that offer best opportunities for success PORTFOLIO TRANSFORMATIONPortfolio
transformation & strength is driven by R&D, M&A, and Clinical & Regulatory TOP TALENT Portfolio combined with talent drives value ANGIODYNAMICSStrategic Transformation 28

04 | 12 - 15 | 2021 ANGIODYNAMICS The 20th Annual Needham Virtual Healthcare ConferenceJim Clemmer,
President & CEOStephen Trowbridge, EVP & CFO

Appendix A 30

Reconciliation of GAAP to Non-GAAP Net Income and EPS Reconciliation of Net Loss to Adjusted
EBITDA Includes costs related to merger and acquisition activities, restructurings, and unusual items, including asset impairments and write-offs, certain litigation, and other items.Deferred financing fees related to the old credit agreement
were written off during the second quarter of fiscal year 2020.Adjustment to reflect the income tax provision on a non-GAAP basis has been calculated assuming no valuation allowance on the Company's U.S. deferred tax assets and an effective tax
rate of 23% for the periods ended February 28, 2021 and February 29, 2020.Diluted shares may differ for non-GAAP measures as compared to GAAP due to a GAAP loss. 31 Includes costs related to merger and acquisition activities, restructurings,
and unusual items, including asset impairments and write-offs, certain litigation, and other items. (in thousands, except per share data) (in thousands) Reconciliation of Free Cash Flow (in thousands)
