Attached files
| file | filename |
|---|---|
| 8-K - FORM 8-K - Adamis Pharmaceuticals Corp | admp-8k_030221.htm |
Adamis Pharmaceuticals Corporation 8-K
Exhibit 99.1

Corporate Presentation with Overview of Products & Development Pipeline March 2021

2

Adamis Pharmaceuticals Corporation (Adamis) is a specialty biopharmaceutical company primarily focused on developing products in various therapeutic areas, including allergy, opioid overdose, respiratory and inflammatory disease Adamis’ epinephrine injection products are approved by the U.S. Food & Drug Administration (FDA) for use in the emergency treatment of acute allergic reactions, including anaphylaxis and launched in 2019 Adamis’ high - dose naloxone injection product candidate is intended for the reversal of opioid overdose; the Company has submitted responses to the deficiencies identified in the CRL received in November 2020 The Company has other pipeline products in varying stages of development 3

FDA cleared the Company to proceed with the clinical investigation of Tempol for the treatment of coronavirus in COVID - 19 positive patients Positive data from Stanford University study showing Tempol suppressed cytokine production in cells from COVID - 19 patients Walgreens added both SYMJEPI products to its Prescription Savings Club, offering members the lowest price for epinephrine devices ($99.99) in the U.S. Submitted responses to issues raised in CRL for ZIMHI NDA and awaiting Type A meeting with the FDA Announced intent to sell substantially all of the assets of US Compounding to a strategic buyer 4

Begin patient enrollment for the clinical study of Tempol as a treatment for COVID - 19 Publish data from Stanford study showing Tempol suppressed cytokine production from COVID - 19 positive human cells Increased revenue for SYMJEPI as USWM continues to ramp sales and marketing efforts Obtain a favorable outcome from FDA Type A meeting for naloxone NDA Obtain government and/or non - government funding for additional clinical studies of Tempol for the treatment of radiation dermatitis 5

Injectable Products - Using patented, FDA - approved device ▪ SYMJEPI ® (epinephrine) Injection 0.3mg & 0.15mg • Both doses FDA approved, launched in 2019 • Licensed U.S. commercial rights to USWM ▪ ZIMHI TM (naloxone) Injection 5mg • Intended for rapid reversal of opioid overdose • Licensed U.S. commercial rights to USWM Specialty Products ▪ APC - 400 (tempol) Topical Gel • In development for treating radiation induced dermatitis ▪ APC - 410 (tempol) Oral Capsule ▪ In development for treating acute respiratory diseases, including COVID - 19 6

7 Footnotes: 1 Licensed U.S. commercial rights to US WorldMeds 2 Submitted responses to issues raised in CRL 3 Phase II completed, Phase III ready 4 Phase II/III ready PRODUCT DELIVERY POTENTIAL INDICATION PHASE I PHASE II PHASE III NDA APPROVED SYMJEPI ® (epinephrine) Injection 0.3mg Injectable Anaphylaxis (1) SYMJEPI® (epinephrine) Injection 0.15mg Injectable Anaphylaxis (1) ZIMHI ™ (naloxone) Injection 5mg Injectable Opioid Overdose (1)(2) APC - 400 (tempol) Topical Gel Topical Radiation Dermatitis (3) APC-410 (tempol) Oral Capsule Oral Respiratory Disease / Anti-Inflammatory (4)

In 2020, Adamis reacquired U.S. commercial rights for SYMJEPI from Sandoz and licensed U.S. rights for both SYMJEPI and ZIMHI to US WorldMeds (USWM) for milestones and 50% of profits USWM is a privately held specialty pharmaceuticals company focused on developing and commercializing unique branded drug products Built a product portfolio over more than 20 years in the areas of malignant hyperthermia, opioid withdrawal, oral mucositis, and CNS Promoting SYMJEPI through 50+ sales professionals, including 30 outside sales reps, with plans to increase following ZIMHI approval Preparing for a commercial launch of ZIMHI following approval 8

9 1 Source: Bloomberg Intelligence First products developed on patented, FDA - approved injection platform Both doses approved in U.S. and under regulatory review in Australia High dose (66 lbs. or more) Low dose (33 - 66 lbs.) Indication: Anaphylaxis Status: Marketing (US); Under Review (AU) Dose/Route: 0.3mg & 0.15mg IM or SC injection Market (US): $1.5 billion (1) Commercial partners: US WorldMeds (US) ; Chiesi Australia (AU & NZ) ; terms 50% of net profits and potential performance milestones Continuing to explore additional ex - US opportunities
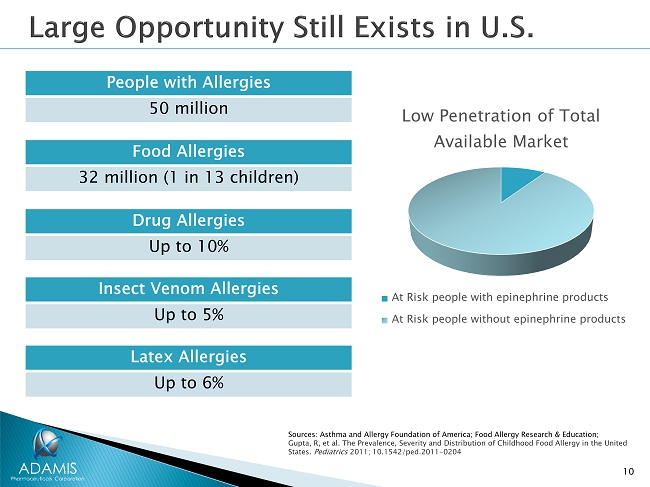
10 Food Allergies 32 million (1 in 13 children) Low Penetration of Total Available Market At Risk people with epinephrine products At Risk people without epinephrine products Sources: Asthma and Allergy Foundation of America; Food Allergy Research & Education; Gupta, R, et al. The Prevalence, Severity and Distribution of Childhood Food Allergy in the United States. Pediatrics 2011; 10.1542/ped.2011 - 0204 Latex Allergies Up to 6% Drug Allergies Up to 10% Insect Venom Allergies Up to 5% People with Allergies 50 million

11 Third product developed on proprietary injection platform Naloxone is a fast - acting opioid antagonist Overdose epidemic: highest number of overdose deaths (81K) ever recorded in a year (ending May 2020) in US; greater than 40% caused by more potent synthetic opioids like fentanyl (2) Higher dose of naloxone needed because more potent opioids cause high mortality and require repeat dosing - incidences up to 83% noted in multiple studies (3) 1 Source: Bloomberg Intelligence 2 Source: CDC – Centers for Disease Control and Prevention 3 Source: Morbidity and Mortality Weekly Report, April 14, 2017 Indication: Opioid Overdose Status: Type A Meeting pending Dose/Route: 5mg IM or SC injection Market (US): $330 million (1) 5mg Dose (IM Injection)

Market growing in the midst of opioid and COVID - 19 crises ▪ Over the last five years, annual naloxone sales rose from $44mm to $330mm and units sold grew 28% year - over - year (1) ▪ COVID - 19 impact – 81K overdose fatalities in the U.S. between 6/2019 and 5/2020 – highest one year overdose deaths ever recorded (2) Market is in transition ▪ Historically a non - retail market composed of non - branded injectables ▪ New consumer - oriented products and enhanced awareness have created an emerging retail market channel ▪ FDA now recommends HCPs co - prescribe naloxone with opioids Limited competitive activity ▪ Only approved intramuscular injection ( Evzio ®) was discontinued ▪ Remaining approved product (Narcan®) is a lower dosage, which may be sub - optimal due to the increasing strength of synthetic opioids (3) 12 1 Source: Bloomberg Intelligence 2 Source: CDC – Centers for Disease Control and Prevention 3 Source: Moss RB, et. al. (2020) Higher naloxone dosing in a quantitative systems pharmacology model that predicts naloxone - fentanyl competition at the opioid mu receptor level. PLoS ONE 15(6): e0234683. https://doi.org/10.1371/journal.pone.0234683

13 Novel anti - oxidant and anti - inflammatory synthetic compound which has shown efficacy in treatment of RID 1.08 million new cancer cases are diagnosed annually in the U.S. and 60% of those patients will undergo radiation treatment (1) 85% of patients will experience moderate to severe skin reactions (2) Currently no approved treatment for RID Phase II study of Tempol in treatment of RID completed; currently Phase III ready 1 Source: American Cancer Society - 2020 Estimates 2 Source: Salvo N, et. al. Prophylaxis and Management of Acute R adiation - Induced S kin R eactions: A S ystematic R eview of the L iterature. Current Oncology. 2010 Aug;17(4):94 - 112 Indication: Radiation Induced Dermatitis (RID) Status: Phase III Ready Route: Topical Gel Market (US): ~1.08 million patients annually (1)

14 Main role intended as an adjuvant treatment to chemo and radiation therapies in cancer patients making treatment much more manageable and improving quality of life Radiation therapy is a common treatment for cancer patients and had a global market size of about $5.6 billion in 2018 (1) One of the most common side effects of radiation is acute skin reaction (radiation dermatitis or RID) that ranges from a mild rash to severe ulceration Approximately 85% of patients treated with radiation therapy will experience an adverse skin reaction (2) 1 Source: Radiotherapy Market by Type (External (IGRT, IMRT, 3D - CRT, Stereotactic), Brachytherapy (LDR, HDR)), Product (LINAC, CyberKnife , Gamma Knife, Tomotherapy , Particle Therapy, Cyclotron), Application (Prostate, Breast), End User (Hospital) - Forecasts to 2023 2 Source: Salvo N, et. al. Prophylaxis and Management of Acute R adiation - Induced S kin R eactions: A S ystematic R eview of the L iterature. Current Oncology. 2010 Aug;17(4):94 - 112

15 Tempol is a novel synthetic compound which reduces oxidative stress and inflammatory cytokines associated with the cytokine storm In animal models of LPS induced ARDS, Tempol demonstrated decreased lung pathology (2) In an animal model of a betacoronavirus , Tempol showed increased survival and decreased virus levels (3) Cleared by FDA to begin clinical studies of Tempol as a treatment for COVID - 19 1 Source: CDC - Average New Cases Reported as of February 12, 2021 2 Source: El - Sayed NS, et al. European Journal of Pharmacology, 2011 August 1; 663 (1 - 3): 68 - 73 3 Source: Tsuhako MH, et al. Free Radical Biology and Medicine. 2010 March 1; 48(5): 704 - 712 Indication: Respiratory Disease Status: Phase II/III ready Route: Oral Capsule Market (US): 14,647 daily COVID - 19 cases (1)

Begin patient enrollment for the clinical study of Tempol as a treatment for COVID - 19 Publish data from Stanford study showing Tempol suppressed cytokine production from COVID - 19 positive human cells Increased revenue for SYMJEPI as USWM continues to ramp sales and marketing efforts Obtain a favorable outcome from FDA Type A meeting for naloxone NDA Obtain government and/or non - government funding for additional clinical studies of Tempol for the treatment of radiation dermatitis 16

