Attached files
| file | filename |
|---|---|
| EX-32.2 - CERTIFICATION - SHINECO, INC. | f10k2018ex32-2_shinecoinc.htm |
| EX-32.1 - CERTIFICATION - SHINECO, INC. | f10k2018ex32-1_shinecoinc.htm |
| EX-31.2 - CERTIFICATION - SHINECO, INC. | f10k2018ex31-2_shinecoinc.htm |
| EX-31.1 - CERTIFICATION - SHINECO, INC. | f10k2018ex31-1_shinecoinc.htm |
| EX-23.1 - CONSENT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM - SHINECO, INC. | f10k2018ex23-1_shinecoinc.htm |
| EX-21.1 - LIST OF SUBSIDIARIES OF THE COMPANY - SHINECO, INC. | f10k2018ex21-1_shinecoinc.htm |
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, DC 20549
FORM 10-K
☒ annual report pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
For the fiscal year ended June 30, 2018
or
☐ Transition report pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
For the transition period from _________ to _________
Commission File Number: 001-37776

| SHINECO, INC. |
| (Exact name of issuer as specified in its charter) |
| Delaware | 52-2175898 | |
| (State or other jurisdiction of | (I.R.S. employer | |
| incorporation or organization) | identification number) | |
| Room 1001, Building T5, DaZu Square, | ||
| Daxing District, Beijing | ||
| People’s Republic of China | 100176 | |
| (Address of principal executive offices) | (Zip Code) |
Registrant’s telephone number, including area code (+86) 10-87227366
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Name of each exchange on which registered | |
| Common stock, $0.001 par value | NASDAQ Capital Market |
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐ No þ
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐ No þ
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes þ No ☐
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every, Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (Sec. 232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes þ No ☐
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§ 229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer | ☐ | Accelerated filer | ☐ |
| Non-accelerated filer | ☐ (Do not check if a smaller reporting company) | Smaller reporting company | þ |
| Emerging growth company | þ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ☐ No þ
The aggregate market value of 16,676,070 shares of voting and non-voting common equity stock held by non-affiliates of the registrant was approximately $38,354,961 as of December 29, 2017, the last business day of the registrant’s most recently completed second fiscal quarter, based on the last sale price of the registrant’s common stock on such date of $2.3 per share, as reported on the Nasdaq Capital Market.
As of October 15, 2018, the registrant had 22,871,772 shares of common stock outstanding.
TABLE OF CONTENTS
TO ANNUAL REPORT ON FORM 10-K
FOR YEAR ENDED JUNE 30, 2018
| i |
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This annual report on Form 10-K (the “Report”) and other reports (collectively the “Filings”) filed by the registrant from time to time with the Securities and Exchange Commission (the “SEC”) contain or may contain forward looking statements and information that are based upon beliefs of, and information currently available to, the registrant’s management as well as estimates and assumptions made by the registrant’s management. When used in the filings the words “anticipate,” “believe,” “estimate,” “expect,” “future,” “intend,” “plan” or the negative of these terms and similar expressions as they relate to the registrant or the registrant’s management identify forward looking statements. Such statements reflect the current view of the registrant with respect to future events and are subject to risks, uncertainties, assumptions and other factors (including the risks contained in the section of this Report entitled “Risk Factors”) relating to the registrant’s industry, the registrant’s operations and results of operations and any businesses that may be acquired by the registrant. Should one or more of these risks or uncertainties materialize, or should the underlying assumptions prove incorrect, actual results may differ significantly from those anticipated, believed, estimated, expected, intended or planned.
Although the registrant believes that the expectations reflected in the forward-looking statements are reasonable, the registrant cannot guarantee future results, levels of activity, performance or achievements. Except as required by applicable law, including the securities laws of the United States, the registrant does not intend to update any of the forward-looking statements to conform these statements to actual results. The following discussion should be read in conjunction with the registrant’s financial statements and the related notes thereto included in this Report.
All references to “we,” “us,” “our,” “TYHT,” “Company,” “registrant” or similar terms used in this report refer to Shineco, Inc., a Delaware corporation (“TYHT”), including its consolidated subsidiaries and variable interest entities (“VIEs”), unless the context otherwise indicates.
| ii |
| ITEM 1. | Business |
General Overview
We are a Delaware holding company that uses our subsidiaries’ and variable interest entities’ vertically and horizontally integrated production, distribution and sales channels to provide health and well-being focused plant-based products. Our products are only sold domestically in China. We utilize modern engineering technologies and biotechnologies to produce, among other products, Chinese herbal medicines, organic agricultural produce and specialized textiles. Our health and well-being focused plant-based products business is divided into three major segments:
1. Processing and distributing traditional Chinese herbal medicine products as well as other pharmaceutical products. This segment is conducted through Ankang Longevity Group, which operates 66 cooperative retail pharmacies throughout Ankang, a city in southern Shaanxi province, China, through which we sell directly to individual customers traditional Chinese medicinal products produced by us as well as by third parties. Ankang Longevity Group also owns a factory specializing in decoction, which is the process by which solid materials are heated or boiled in order to extract liquids, and distributes decoction products to wholesalers and pharmaceutical companies around China. This segment accounted for approximately 32% of our revenues for the year ended June 30, 2018.
2. Processing and distributing green and organic agricultural produce as well as growing and cultivating yew trees (taxus media). We currently cultivate and sell yew mainly to large group and corporate customers, but do not currently process yew trees into Chinese or Western medicines. This segment is conducted through the Company’s variable interest entities: the Zhisheng Group, which comprises the following Chinese companies participating in our yew tree business: Shineco Zhisheng (Beijing) Bio-Technology Co. (“Zhisheng Bio-Tech”), Yantai Zhisheng International Freight Forwarding Co., Ltd (“Zhisheng Freight”), Yantai Zhisheng International Trade Co., Ltd (“Zhisheng Trade”), Yantai Mouping District Zhisheng Agricultural Produce Cooperative (“Zhisheng Agricultural”), and Qingdao Zhihesheng Agricultural Produce Services, Ltd (“Qingdao Zhihesheng”). This segment accounted for approximately 43% of our revenues for the year ended June 30, 2018. Zhisheng Agricultural has not had any significant business activities and thus we have deregistered it in 2017. We have transferred all assets, rights and liabilities to an affiliated entity, Zhisheng Freight.
3. Developing and distributing specialized fabrics, textiles and other byproducts derived from an indigenous Chinese plant Apocynum Venetum, grown in the Xinjiang region of China, and known in Chinese as “Luobuma” or “bluish dogbane”. Our Luobuma products are specialized textile and health supplement products designed to incorporate traditional Eastern medicines with modern scientific methods. These products are predicated on centuries-old traditions of Eastern herbal remedies derived from the Luobuma raw material. This segment is channeled through the Company’s directly-owned subsidiary, Beijing Tenet-Jove Technological Development Co., Ltd. (“Tenet-Jove”). This segment accounted for approximately 25% of our revenues for the year ended June 30, 2018.
We primarily market our health and wellbeing-focused products in China. At present, we do not sell any of our products in the United States or Canada. China’s domestic pharmaceutical and healthcare products market is fast-growing but, in our opinion, underdeveloped. We believe China’s healthcare sector has the capacity to develop even further. From pharmaceuticals to medical products to general consumer health, China remains among the world’s most attractive markets, and by far the fastest-growing of all the large emerging ones. Driving this growth is China’s aging population, increased incidence of chronic diseases, and a material increase in investment from both domestic and foreign corporations. The growth also reflects the Chinese government’s focus on healthcare as both a social priority (as witnessed in its late 2000s healthcare reforms) and a strategic priority (as evidenced in the 12th five-year plan’s stated focus on growing the biomedical industry in the future).
| 1 |
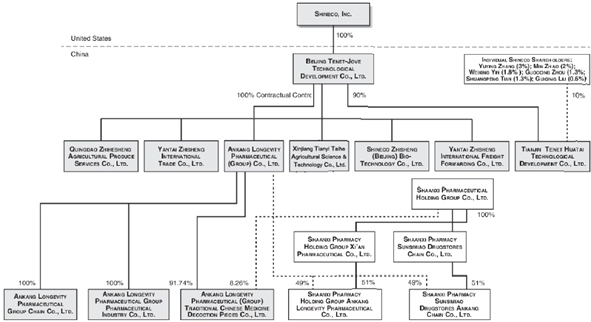
History and Corporate Structure
Shineco, Inc. was incorporated under the laws of the State of Delaware on August 20, 1997 as Supcor, Inc. From 1997 to 2004, the Company’s only activities were organizational ones, directed at developing its business plan and raising initial capital. On December 30, 2004, the Company acquired all of the issued and outstanding shares of Tenet-Jove, a company organized under the laws of the People’s Republic of China on December 15, 2003 that operated in the fabrics and textile business, in exchange for restricted shares of Shineco’s common stock; as a result, Tenent-Jove became our wholly-owned subsidiary. At that point, the sole operating business of Tenet-Jove then became that of the Company. On June 9, 2005, we changed our name to Shineco, Inc.
On April 19, 2017, Tenet-Jove established Xinjiang Tiankunrunze Biological Engineering Co., Ltd. (“Tiankunrunze”) and owned a 65% equity interest in Tiankunrunze. On April 28, 2017, Tiankunrunze established Xinjiang Tianzhuo Technology Development Co., Ltd. (“Tianzhuo”) with registered capital of RMB 10.0 million ($1,450,233). On May 22, 2017, Tiankunrunze established Xinjiang Tianhuihechuang Agriculture Development Co., Ltd. (“Tianhuihechuang”) with registered capital of RMB 10.0 million ($1,452,294). On May 23, 2017, Tiankunrunze established Xinjiang Tianxintongye Biotechnology Development Co., Ltd. (“Tianxintongye”) with registered capital of RMB 10.0 million ($1,451,615). Tianzhuo, Tianhuihechuang and Tianxintongye became subsidiaries of Tenet-Jove.
On June 9, 2017, the Company and its subsidiary Tiankunrunze have entered into a Strategic Cooperation Agreement with Beijing Zhongke Biorefinery Engineering Technology Co., Ltd. (“Biorefinery”), a leading high-tech biomass refining company financially backed by the Chinese Academy of Sciences Institute of Process Engineering, to establish the Institute of Chinese Apocynum Industrial Technology Research (“ICAITR”). Pursuant to the Strategic Cooperation Agreement entered into on May 2, 2017 among the three parties, Tiankunrunze, Shineco and Biorefinery agreed to establish the ICAITR and each will own 45%, 35% and 20% of the equity interests of ICAITR, respectively. Shineco and Tiankunrunze will invest RMB 5.0 million ($737,745) as the registered capital, and Biorefinery will invest technology such as the patent for “Steam Explosion Degumming” as well as other resources.
As of June 30, 2018, Tenet-Jove, through a series of contractual relationships, effectively controls and manages:
| ● | Ankang Longevity Pharmaceutical (Group) Co., Ltd. (“Ankang Longevity Group”), which owns a controlling interest in the following Chinese companies participating in our traditional Chinese medicine business: Ankang Longevity Pharmaceutical (Group) Traditional Chinese Medicine Decoction Pieces Co., Ltd. (“Ankang Longevity Decoction Pieces”), Ankang Longevity Pharmaceutical Group Chain Co., Ltd. (“Ankang Longevity Chain”), and Ankang Longevity Pharmaceutical Group Pharmaceutical Industry Co., Ltd. (“Ankang Longevity Industry”); and |
| ● | Zhisheng Group, which comprises the following Chinese companies participating in our yew tree business: Shineco Zhisheng (Beijing) Bio-Technology Co., Yantai Zhisheng International Freight Forwarding Co., Ltd, Yantai Zhisheng International Trade Co., Ltd, and Qingdao Zhihesheng Agricultural Produce Services, Ltd. |
| 2 |
The Company is also a majority shareholder of Tiankunrunze, Tianjin Tenet Huatai Technological Development Co., Ltd. (“Tianjin Tenet Huatai”), and it owns 49% of Shaanxi Pharmacy Holding Group Ankang Longevity Pharmaceutical Co., Ltd. and Shaanxi Pharmacy Sunsimiao Drugstores Ankang Chain Co., Ltd. through a joint venture with Shaanxi Pharmaceutical Group Pai’ang Medicine Co. Ltd. (“Shaanxi Pharmaceutical Group”), a Chinese state-owned pharmaceutical enterprise.
Zhisheng Agricultural has not had any significant business activities and thus we have deregistered it in 2017. We have transferred all assets, rights and liabilities to an affiliated entity, Zhisheng Freight.
On September 30, 2017, Tenet-Jove established Xinjiang Tianyitaihe Agriculture Development Co., Ltd. (“Tianyitaihe”) with registered capital of RMB 10.0 million ($1,502,652). On September 30, 2017, Tenet-Jove also established Xinjiang Tianyirunze Biotechnology Development Co., Ltd. (“Tianyirunze”) with registered capital of RMB 10.0 million ($1,502,652). Tianyitaihe and Tianyirunze are wholly owned subsidiaries of Tenet-Jove.
Our current corporate structure as of June 30, 2018 is as follows:
Contractual Arrangements with Ankang Longevity Group or Zhisheng Group and their Owners
We conduct our business through a combination of contractual arrangements with PRC operating companies and equity ownership of PRC subsidiaries. In most cases, we have had to use contractual relationships because direct investment by foreign-owned companies like our Delaware company is prohibited or restricted. In other cases we have elected to do so in spite of being permitted to own such operating company directly. Where we operate our business through such contractual relationships, we are subject to risks related to such operation.
The principal regulation governing foreign ownership of businesses in the PRC is the Foreign Investment Industrial Guidance Catalogue, effective as of April 10, 2015 (the “Catalogue”). The Catalogue classifies various industries into three categories: encouraged, restricted and prohibited. Shineco is engaged in business in industries where direct foreign investment is expressly prohibited: the preparation of traditional Chinese medicines in small pieces ready for decoction.
| 3 |
Due, in part, to the regulations on foreign ownership of PRC businesses, neither we nor our subsidiaries own any equity interest in Ankang Longevity Group or the Zhisheng Group. The foregoing companies are referred to herein as the “Controlled Companies.” Instead, we control and receive the economic benefits of the Controlled Companies’ business operations through a series of contractual arrangements. Tenet-Jove, each of the Controlled Companies and its shareholders have entered into a series of contractual arrangements, also known as VIE Agreements. The VIE Agreements are designed to provide Tenet-Jove with the power, rights and obligations equivalent in all material respects to those it would possess as the sole equity holder of each Controlled Company, including absolute control rights and the rights to the assets, property and revenue and net income of each Controlled Company. Based on a legal opinion issued by Dentons LLP to Tenet-Jove, the VIE Agreements constitute valid and binding obligations of the parties to such agreements and are enforceable and valid in accordance with the laws of the PRC.
Each of the types of VIE Agreements is described below and consist of, for each of Ankang Longevity Group and the Zhisheng Group, (a) exclusive business cooperation agreements, (b) timely reporting agreements, (c) equity interest pledge agreements, (d) exclusive option agreements, and (e) powers of attorney. As an overview, these agreements taken together are designed to allow the Company to manage the operations of each of the Controlled Companies and to receive all of the net income of such Controlled Companies in return therefor. To secure our interest in the Controlled Companies, the equity interest pledges and option agreements and the powers of attorney are designed to allow us to step in and convert our contractual interest into an equity interest in the event we determine that doing so is warranted. Finally, the timely reporting agreement is designed to ensure that we have timely access to the financial and other information from the Controlled Companies that we require in order to prepare regulatory and other filings.
The controlling shareholders of the Company and the VIEs could cancel these agreements or permit them to expire at the end of the agreement terms, as a result of which the Company would not retain control of the VIEs.
The following is a summary of the common contractual arrangements that provide us with effective control of our VIEs and that enable us to receive substantially all of the economic benefits from their operations.
Exclusive Business Cooperation Agreements
Tenet-Jove entered into an Exclusive Business Cooperation Agreement with Zhisheng Bio-Tech, Ankang Longevity Group, and Qingdao Zhihesheng on February 24, 2014, December 31, 2008, and May 24, 2012, respectively, and with each of Zhisheng Freight, Zhisheng Trade, and Zhisheng Agricultural on June 16, 2011. Zhisheng Agricultural has not had any significant business activities and thus we have deregistered it in 2017. We have transferred all assets, rights and liabilities to an affiliated entity, Zhisheng Freight.
Tenet-Jove is currently managing each Controlled Company pursuant to the terms of an Exclusive Business Cooperation Agreement.
| 4 |
Pursuant to substantially identical Exclusive Business Cooperation Agreements between each Controlled Company and Tenet-Jove, Tenet-Jove provides such Controlled Company with technical support, consulting services and other management services relating to its day-to-day business operations and management, on an exclusive basis, utilizing its advantages in technology, human resources, and information. Additionally, each Controlled Company has granted an irrevocable and exclusive option to Tenet-Jove to purchase from such Controlled Company, any or all of its assets, to the extent permitted under applicable PRC law. Tenet-Jove may exercise, at its sole discretion, the option to purchase from each Controlled Company any or all of such Controlled Company’s assets at the lowest purchase price permitted by PRC law. Should Tenet-Jove exercise such option, the parties shall enter into a separate asset transfer or similar agreement. Tenet-Jove shall own all intellectual property rights that are developed during the course of each Exclusive Business Cooperation Agreement. For services rendered to each Controlled Company by Tenet-Jove under the agreement to which such Controlled Company is a party, Tenet-Jove is entitled to collect a service fee calculated based on the time of services rendered multiplied by the corresponding rate, which is approximately equal to the net income of such Controlled Company.
Each Exclusive Business Cooperation Agreement shall remain in effect for ten years until it is extended or terminated by Tenet-Jove, which may be done unilaterally, except in the case of gross negligence or fraud, in which case the Controlled Companies may terminate the agreements. Pursuant to each such agreement, Tenet-Jove has absolute authority relating to the management of each Controlled Company, including but not limited to decisions with regard to expenses, salary raises and bonuses, hiring, firing and other operational functions. Although the Exclusive Business Cooperation Agreements do not prohibit related party transactions, the audit committee of Shineco will be required to review and approve in advance any related party transactions, including transactions involving Tenet-Jove or any Controlled Company.
Timely Reporting Agreements
To ensure each Controlled Company promptly provides all of the information that Tenet-Jove and the Company need to file various reports with the SEC and other applicable regulatory authorities, a Timely Reporting Agreement was entered between each Controlled Company and Shineco on July 3, 2014.
Under the Timely Reporting Agreements, each Controlled Company agrees that it is obligated to make its officers and directors available to Shineco and promptly provide all information required by Shineco so that Shineco can file all necessary SEC and other regulatory reports as required.
Equity Interest Pledge Agreements
Under the Equity Interest Pledge Agreements among each Controlled Company (other than Zhisheng Agricultural, which is a cooperative and thus has no equity interests that can be pledged), the shareholders of each such Controlled Company and Tenet-Jove, the shareholders pledged all of their equity interests in each such Controlled Company to Tenet-Jove to guarantee the performance of such Controlled Company’s obligations under the respective Exclusive Business Cooperation Agreement. Under the terms of each agreement, in the event that the Controlled Company or its shareholders breach their respective contractual obligations under the Exclusive Business Cooperation Agreement to which they are a party, Tenet-Jove, as pledgee, will be entitled to certain rights, including, but not limited to, the right to collect dividends generated by the pledged equity interests. Each Controlled Company’s shareholders also agreed that upon occurrence of any event of default, as set forth in the applicable Equity Interest Pledge Agreement, Tenet-Jove is entitled to dispose of the pledged equity interest in accordance with applicable PRC laws. Each Controlled Company’s shareholders further agree not to dispose of the pledged equity interests or take any actions that would prejudice Tenet-Jove’s interest in the applicable Controlled Company.
| 5 |
Each Equity Interest Pledge Agreement shall be effective until all payments due under the related Exclusive Business Cooperation Agreement have been paid by the Controlled Company party thereto. Tenet-Jove shall cancel or terminate an Equity Interest Pledge Agreement upon a Controlled Company’s full payment of fees payable under its applicable Exclusive Business Cooperation Agreement.
Exclusive Option Agreements
Under the Exclusive Option Agreements, shareholders of each of the Controlled Companies (other than Zhisheng Agricultural, which is a cooperative and thus has no equity holders) irrevocably granted Tenet-Jove (or its designee) an exclusive option to purchase, to the extent permitted under PRC law, once or at multiple times, at any time, part or all of their equity interests in each Controlled Company. The option price is equal to the capital paid in by the applicable Controlled Company shareholders subject to any appraisal or restrictions required by applicable PRC laws and regulations. The option purchase price shall increase in case the applicable Controlled Company shareholders make additional capital contributions to such Controlled Company.
Each agreement remains effective for a term of ten years and may be unilaterally renewed at Tenet-Jove’s election.
Powers of Attorney
Under the Powers of Attorney, the shareholders of each Controlled Company authorize Tenet-Jove to act on their behalf as their exclusive agent and attorney with respect to all rights as shareholders of the respective Controlled Companies, including but not limited to: (a) attending shareholders’ meetings; (b) exercising all the shareholder’s rights, including voting, that shareholders are entitled to under the laws of China and the Articles of Association, including but not limited to the sale or transfer or pledge or disposition of shares in part or in whole; and (c) designating and appointing on behalf of shareholders the legal representative, the executive director, supervisor, the chief executive officer and other senior management members of the respective Controlled Companies.
Our Products and Operations
Our health and well-being focused plant-based products business is divided into three major segments:
| 1. | Processing and distributing traditional Chinese herbal medicine products as well as other pharmaceutical products. This segment is conducted by the Company’s VIE, Ankang Longevity Group. |
| 2. | Planting, processing and distributing green and organic agricultural produce as well as growing and cultivating yew trees (taxus media). This segment is conducted through the Company’s VIEs, the Zhisheng Group. |
| 3. | Developing and distributing specialized fabrics, textiles and other byproducts derived from an indigenous Chinese plant Apocynum Venetum, known in Chinese as “Luobuma” or “bluish dogbane”. This segment is channeled through the Company’s directly-owned subsidiary, Tenet-Jove. |
| 6 |
The details of each business segment are described as follows:
Ankang Longevity Group
The companies of this segment, Ankang Longevity Group, operates 66 cooperative retail pharmacies throughout Ankang, a city in southern Shaanxi province, PRC, through which we sell directly to individual customers traditional Chinese medicinal products produced by us as well as by third parties. This group also processes more than 600 kinds of Chinese medicinal herbal products such as medicines for bone and joint pain, arthritis, respiratory infections and insomnia, and distribute such products through an established Chinese domestic sales and distribution network, including more than 20 pharmaceutical companies and more than 50 hospitals throughout China. In May, 2013, Ankang Longevity Group has established a joint venture with a large state-owned domestic pharmacy chain group, Shaanxi Pharmaceutical Group Pai’ang Medicine Co. Ltd.. As part of the joint venture, Ankang Longevity Group contributed 13 retail pharmacies— operating as Sunsimiao Pharmacies— to the joint venture while retaining 66 retail pharmacies whereby Ankang Longevity Group acts as a franchisor. We also provide evaluation and diagnostic services by on-site doctors at pharmacies to support and drive our Chinese medicine product sales. The operations of this segment are focused in the northwest region of Mainland China, particularly Shaanxi province. This segment accounts for approximately 32% of our revenues.
Ankang Longevity Group has built a scalable decocting-free Chinese herbal medicine manufacturing facility that passed the Good Manufacturing Practice (“GMP”) certification in 2009 and was recognized as a Key Agricultural Industrialization Companies by the Shaanxi Provincial Government. The facility covers an area of over 4,000 square meters and is equipped with an advanced toxic herbal medicine treatment production line and herbal medicine testing instruments.
Ankang Longevity Group has recently acquired land use rights to use 8,200 acres of selenium-rich farmland and woodland in Ziyang County, China to grow our Chinese medicine and other plant products.
Zhisheng Group
The Company’s other VIEs, which form the Zhisheng Group and include Zhisheng Bio-Tech, Zhisheng Freight, Zhisheng Trade and Qingdao Zhihesheng, engage in the business of organic agricultural products, principally yew trees, as well as providing logistics services for all of the agricultural products we produce. Since 2013, this segment is focusing its efforts on the growing and cultivation of yew trees (taxus media), small evergreen trees that can be used for the production of anti-cancer medication as well as ornamental bonsai trees, which are known to have the effect of purifying indoor air quality. We currently cultivate and sell yew trees but do not currently process yew into Chinese or Western medicines. The entities composing the Zhisheng Group are currently focusing on researching, developing and cultivating organic produce, yew ecological products and other native plants. The operations of this segment are focused in the East region of Mainland China, principally Shandong Province, and in Beijing where we have newly developed over 100 acres of modern greenhouses for cultivating yew and other plants. This segment accounts for approximately 43% of our revenues.
| 7 |
Tenet-Jove
Through Tenet-Jove and Tianjin Tenet Huatai, the Company develops and distributes specialized textiles and health supplements derived from a native Chinese plant Apocynum venetum, grown in the Xinjiang region of China and known in Chinese as “Luobuma” or “bluish dogbane” and referred to herein as Luobuma. This plant has traditionally been used in China both internally and externally for centuries to treat high blood pressure, depression, dizziness, pain, insomnia, and other common ailments. The stems of Luobuma serve as raw material for fiber used in textile production, and the leaves serve as raw material for pharmaceutical drugs. This segment accounts for approximately 25% of our revenues.
The companies of this segment, Tenet-Jove and Tianjin Tenet Huatai, specialize in Luobuma sourcing and developing Luobuma byproducts. With rich experience and broad channels in the Chinese domestic market, we believe that we are one of the leaders in Luobuma textile sales in China. This segment’s operations are focused in the north region of Mainland China, mostly carried out in Xinjiang and Tianjin. Our Luobuma products are specialized textile and health supplement products designed to incorporate traditional Eastern medicines with modern scientific methods. These products are predicated on centuries-old traditions of Eastern herbal remedies derived from the Luobuma raw material.
In addition to developing textile products, we expect to use our high-pressure steam degumming process to extract other Luobuma byproducts we intend to commercialize and distribute: flavonoids, xylooligosaccharides (XOS), edible pectin, fiberboard, and organic fertilizer. The traditional method of degumming Luobuma only produces Luobuma fiber, whereas our high-pressure steam degumming process produces these five additional Luobuma byproducts. Flavonoids are organic compounds widely distributed in plants, and flavonoid-rich Luobuma extract can be used in the manufacture of many pharmaceuticals. Xylooligosaccharides, or XOS, is a sugar that can be used as a food additive that provides various health benefits like lowering glucose levels. Pectin is a thickener and stabilizer used in food, beverages and cosmetics, as well as a gelling agent for jellies. Fiberboard is a type of engineered wood alternative that is made out of Luobuma fibers; it is used widely for furniture manufacturing and packaging.
Product Descriptions
Traditional Chinese Medicines
Our Ankang Longevity Group manufactures hundreds of Chinese medicinal herbal products and decoction pieces such as medicines for bone and joint pain, arthritis, respiratory infections, insomnia, as well as many other common ailments. We distribute such products through an established Chinese domestic sales and distribution network, including through more than 20 pharmaceutical wholesale companies and more than 50 hospitals throughout China, as well as through our own and cooperative retail pharmacies. In addition to distributing Chinese medicinal herbal products, we also distribute many popular Western medicines we purchase from outside parties through our wholesale and retail channels so that we can offer a good variety of products to meet customer demands. In the second quarter of 2013, this group entered the pharmaceutical retailing industry through a joint venture with another large domestic pharmacy chain group, Shaanxi Pharmaceutical Group. Ankang’s main products include the following traditional Chinese medicines:
| ● | Polygonum cuspidatum or Japanese knotweed, which is ingested to treat colds, digestive diseases, hepatitis and Cholecystitis (gallbladder inflammation); |
| 8 |
| ● | Salvia mint, which is ingested to improve microcirculation; | |
| ● | Tianma, which is ingested to treat Rheumatoid arthritis; | |
| ● | Eucommia, which is ingested for bone and join pain; | |
| ● | Radix, which is ingested to treat respiratory infections; | |
| ● | Schisandra shrub, which is ingested to treat insomnia; and | |
| ● | Berberis shrub, which is used as a raw material for antibiotics. |
Yew Trees
Currently, through our Zhisheng Group VIEs, we sell ornamental yew trees and yew cuttings to third parties. We also rent ornamental yew trees to companies who desire the environmental benefits of natural plants in their workplaces. Until recently we were primarily engaged in the production, distribution and sale of agricultural products, including the planting and processing of organic fruits and vegetables, such as tomatoes, eggplants, string beans, peppers as well as certain popular fruits in China like blueberries and wine grapes, but those operations have been temporarily scaled back due to stiff competition and a change of our internal policy in favor of the expansion of our yew tree business.
As our inventories of young yew trees mature, our long-term goals are particularly focused on the extraction of paclitaxel or taxol, which is derived from certain species of yew trees including those we grow. Taxol, a broad-spectrum mitotic inhibitor used in cancer chemotherapy, can be extracted from mature yew trees. As a mitotic inhibitor, taxol adheres to rapidly dividing cancerous cells during mitosis (cell division) and interferes with the division process. It may suppress tumor growth through regulating microtubule stabilization, inducing apoptosis and adjusting immunologic mechanism. Taxol is also used for the prevention of restenosis, which is the narrowing of blood vessels. In the treatment of certain soft tissue cancers, such as breast cancer, taxol is given for early stage and metastatic breast cancer after combination anthracycline and cytoxan therapy and is also given as treatment to shrink a tumor before surgery. It can also be used together with a drug called Cisplatin to treat advanced ovarian cancer and non-small cell lung cancer, or “NSCLC.” The U.S. Food and Drug Administration approved taxol as the primary and secondary treatment for NSCLC. There are other generally accepted protocols for the use of taxol as a cancer drug alone or in combination with other drugs depending upon the diagnosis, staging and type of cancer, as well as a patient’s medical history, tolerances and allergies, among other relevant factors. Taxol is usually sold to large pharmaceutical companies to be used in their products, which can be used to treat patients with lung, ovarian, breast, head and neck cancer, and advanced forms of Kaposi’s sarcoma.
Tenet-Jove Textiles
Our company’s scientists and other Chinese researchers have brought modern scientific methods to the study of Luobuma, and have determined that Luobuma fibers have an increased tendency to radiate light at the “far infrared” end of the light spectrum, with wavelengths measuring between 8-15 microns (referred to as “FIR”). Based on Chinese scientific studies some believe that Luobuma’s FIR-radiating qualities exert a positive effect on various functions of the human body, including cellular metabolism. For this reason, we have marketed and sold these products utilizing such technology. These products are popular with Chinese customers seeking the perceived benefits of traditional Chinese medicine.
For example, according to a report by the College of Science of Tianjin University, tests conducted by the PRC’s National Institute of Metrology have reported that the radiance rate of far infrared light from Luobuma fiber is 84%, 2 to 4 times higher than that from cotton and other natural fibers. The same tests found that the FIR radiance rate from our proprietary bio-ceramic powder reaches 91%. Healthful benefits have been observed at radiance rate levels above 70%. Based on these observations about FIR radiance, we have developed textiles that our customers can wear and from which we believe they can receive those health benefits commonly associated with Chinese herbal remedies.
| 9 |
Tenet-Jove first commercially developed the natural FIR-radiant properties of the Luobuma plant in 1997. We refer to this natural Luobuma fiber as a “Second Generation” FIR textile. The “First Generation” of FIR-radiant textiles initially became popular in China around 1989, when manufacturers learned to add 3% of a FIR-radiant inorganic material to synthetic fibers comparable to nylon or polyester. This “First Generation” FIR material employs a relatively low level of technology and has relatively few perceived or measurable health benefits. The “Second Generation” FIR textiles we have developed are softer, smoother and more breathable natural fibers that are not as prone to static electricity as the low technology “First Generation” FIR-radiant textiles.
Our Luobuma fabrics have been a success in the Chinese domestic market and have also received numerous awards. The technology applied to our Luobuma-based FIR Therapeutic Clothing and Textile Products has received a “Special Golden Award” from the China National Intellectual Property Bureau at China’s National Patent and Brand Expo. Our products under the brand name of “Tenethealth” have also been honored with the title of “Consumer’s Favorite Products” by the Chinese Consumer Association.
The fibers of natural Luobuma FIR materials can contain up to 32 medicinal compounds, many of which are familiar to practitioners of traditional Chinese medicine. In addition, our processes for manufacturing Luobuma textiles produce a fabric that is smooth, air-permeable, and soft. By combining a product that is familiar to PRC consumers seeking the benefits of traditional Chinese medicine with quality and comfort, we believe we are innovative and have chosen a product that has great commercial potential in the Chinese textile market.
Tenet-Jove Product Development
We have developed what we term a “Third Generation” of FIR textiles under a contract with the Institute of Process Engineering at the Chinese Academy of Sciences, one of the leading scientific institutions in China. Our research and development has focused on adding nanotechnology enhancements to our Luobuma textile products, in which we use small-scale nanotechnology to embed or impregnate our Luobuma-fiber textiles with other FIR-radiant materials, bio-ceramic materials, or other Chinese herbal remedies. Using these nanotechnology methods, we have developed and marketed health-promoting textile goods that are impregnated with FIR-radiant materials or other Chinese herbal remedies, which are then absorbed through the wearer’s skin. We believe these “Third Generation” FIR textiles will better combine the health benefits of Luobuma with an even softer, more natural cotton-like fabric that will be popular with Chinese consumers.
The Company presently produces approximately 100 “Third Generation” FIR textile products. These textile products include:
| ● | Far Infrared bedding sets (including various pillows, comforters, and sheets); | |
| ● | Far Infrared underwear, T-shirts, and socks; | |
| ● | Far Infrared knee and shin pads, waist supports and other protective clothing; and | |
| ● | Far Infrared body wraps or protectors (for the ankle, elbow, wrist, and knee). |
All our textile products are made of Luobuma-based fibers and are impregnated with bio-ceramic powder, which contains various minerals such as halloysite. Both the fiber and the bio-ceramic powder are developed with the Company’s patented, proprietary techniques.
| 10 |
Manufacturing and Production Facilities
We have formed strategic alliances with several certified knitting and clothing manufacturers throughout China in order to produce our Luobuma products. We assign them limited manufacturing jobs and require certain conditions, including protecting our proprietary techniques and meeting our rigid quality standards. We are in preliminary negotiations to build a new facility in Kuerle, Xinjiang Uyghur Autonomous Region, China to exploit our steam explosion Luobuma fiber production technology.
In 2013, we began large-scale operations in newly leased, energy efficient greenhouses aggregating 50,000 square meters in Beijing and have temporarily scaled back our operations in our former greenhouses located in Shandong Province. At this time, we rent our greenhouses in Shandong Province to local farmers. Our current greenhouse facilities are used primarily for the cultivation of yew trees and other decorative plants and trees. These state-of-the-art facilities allow us to better regulate light, temperate, humidity and other conditions within the greenhouse as well as to significantly increase the number of plants that can be housed in a particular greenhouse footprint. To a lesser degree, we expect these current greenhouses will reduce our manual labor costs associated with our greenhouse operations by approximately 25%. Rather than competing on price, which has become increasingly difficult in the Chinese market, we expect that technological and productivity advances from our new greenhouses will improve our competitive position within our agricultural segment, but there can be no guarantee that we will experience such advances, or if we do, that such advances would improve our competitive position.
Ankang Longevity Group has an approximately 4,000 square meter production factory and an approximately 2,000 square meter production facility in Ankang City, Shaanxi Provice, China, which are used for production of decoction pieces.
Our Strategy for Research and Development
| ● | To keep our products proprietary and patented; |
| ● | To focus on our core existing product lines: Chinese herbal medicines and pharmaceutical sales, yew cultivation, Luobuma-based products, and FIR technology; |
| ● | To commit to further development of our Luobuma byproducts, houpu magnolia products, and selenium-enriched herbs and plants; and |
| ● | To build strategic alliances with universities and scientific institutions, which will allow us exposure to advanced technologies, excellent researchers and scientists and we believe will lower the costs and timing of the development of new products. |
Tenet-Jove specializes in developing Luobuma products and combining FIR technology with natural herbal medicines. We estimate that there are large supplies of Luobuma in China, especially Xinjiang Province. In China, Luobuma can grow as high as 3.6 meters. In the first year after planting, Luobuma can be harvested once during that year; thereafter, it can be harvested twice per year before or at the beginning of the flowering period in June and a second time around September. Currently, we believe China’s Luobuma supplies are largely undeveloped. The Company’s future success will depend on improving its techniques to industrialize Luobuma by developing new Luobuma-derived products such as improved Luobuma functional fiber and various Luobuma nutritional supplements, which can be marketed and distributed through the Company’s Ankang Longevity business segment.
| 11 |
High-Pressure Steam Degumming Process
We currently produce an extensive line of Luobuma-based textile products; we have an exclusive patent on a Luobuma fiber yarn spinning method. Large-scale production of Luobuma fiber products is generally difficult due to the limited yield of Luobuma fiber and the high cost of obtaining that fiber. The current mainstream technology used to produce Luobuma fiber mainly uses chemical agents to remove gum from Luobuma, which destroys Luobuma fiber and causes lower Luobuma fiber production and pollution and makes it very difficult to extract other valuable by-products. A central technical challenge is how to quickly and efficiently remove the gum and sap from the Luobuma plants so that only the natural fiber remains.
To solve this technical challenge, in August 2006 we signed a technology development contract with the Institute of Process Engineering at the Chinese Academy of Science (the “Institute of Process Engineering”), one of China’s leading scientific institutes. Pursuant to our contract, we and the Institute of Process Engineering worked together to develop a high-pressure steam degumming process for the mass production of Luobuma fiber, as well as its byproducts, which was completed in 2008. A literal translation of the project name is “The Stream Steam Explosion Project.” Essentially, the project will develop a modern material engineering method of quickly blasting a large amount of high-pressure steam into a large container filled with raw Luobuma plants. The steam will remove the gum and sap and leave the fiber for use in textile production. The gum and sap and other Luobuma byproducts will be washed out with the steam and collected in a separate place for other uses.
If we can successfully implement our high-pressure steam degumming process, we expect that our annual yield of Luobuma fiber can rise by over one hundredfold— from less than 200 tons to approximately 27,000 tons per year, and the cost of extracting the fiber will be reduced by approximately 50%. For example, the older method of production might require 150 laborers to produce one ton of Luobuma fiber in one day, whereas our high-pressure steam degumming process can utilize just 30 laborers to produce 15 tons of Luobuma fiber in one day. During the process, a certain amount of steam will be used, but unlike the traditional method of degumming, our high-pressure steam degumming process discharges no hazardous byproducts of any kind, because the fiber will be collected in one place, and the liquefied gum and sap and other material will be collected in another place to further separate flavonoids, xylooligosaccharides (XOS), and edible pectin through biological reverse osmosis membrane. The remaining material could be used to produce fiberboard and organic fertilizer, leaving no hazardous discharge or waste. We expect that this technique will be relatively simple and convenient for us to operate with our advanced technology. Luobuma fiber produced by this technique is more like cotton and more spinnable than before.
Intellectual Property
Trademarks
We regard our trademarks as an important part of our business due to the name recognition of our customers. Our subsidiary, Tenet-Jove, has currently obtained 18 trademark registrations at the China Trademark Office and applying for another 7 trademarks covering various categories of the company’s products. As of June 30, 2018, we are not aware of any valid claim or challenges to our right to use our registered trademark or any counterfeit or other infringement to our registered trademark.
| 12 |
Patents
Currently, the Company holds a patent in the People’s Republic of China for Luobuma fiber yarn preparation and an application method (patent number: 201110429362.9), which serves as our core technology and its derivative applications for our Luobuma business.
Distribution Network
We sell our products through various distribution networks. Our traditional Chinese medicinal products and Western medicines are largely sold through either our wholesale customers or our Ankang retail pharmacies — 13 pharmacies operating as Sunsimiao Pharmacies and 66 pharmacies operated by third parties as Ankang Longevity Group Pharmacy cooperatives. Additionally, we sell decoction pieces on the Anhui Bozhou Chinese medicine transaction market, to medical materials company, such as Qianhe Pharmaceutical Industry Co. and to Chinese patent medicine factories, such as Wanxi Pharmaceutical Factory.
Our Luobuma product distribution networks consists of four distributors who distribute our products to approximately 21 outlets, including flagship stores, retail stores and sales counters. These distributors sell our products throughout mainland China, under our proprietary brand name and “Tenethealth®” trademark. We also sell our Luobuma textile products online through third party e-commerce websites, such as Taobao, Tmall and JD. Our yew trees and agricultural products are primarily sold through our sales personnel and group and institutional sales. In 2013, the Company placed its Luobuma, traditional Chinese medicine and yew products in a total of 144 retail stores and sales counters throughout China and on four e-commerce websites.
Our sales and distribution strategy for our products focuses on expanding our distribution network of retail stores and sales counters into all major provinces and cities of China. We also plan to use our current distribution network to introduce our newly developed products into target markets more efficiently and effectively.
All of these certified outlets operate independently, but they prominently display products mainly carrying our trade name “Tenethealth®”. These independent retailers sell our products as well as other products.
The location and number of retail stores and sales counters selling our products, including all sales outlets (both those connected with our major distributors (i.e., our most frequently used distributors) and other independent sellers), as of June 30, 2018 are listed below:
| Location | Number of Major Distributors | Number of Sales Outlets | ||||||
| Liaoning | 1 | 1 | ||||||
| Jilin | 1 | 1 | ||||||
| Shandong | 2 | 16 | ||||||
| Jiangsu | 2 | 6 | ||||||
| Anhui | 2 | 4 | ||||||
| Shaanxi | 5 | 94 | ||||||
| Xinjiang | 1 | 2 | ||||||
| Sichuan | 1 | 5 | ||||||
| Guangdong | 1 | 3 | ||||||
| Tianjin | 1 | 2 | ||||||
| Beijing | 1 | 4 | ||||||
| Chongqing | 1 | 3 | ||||||
| Hubei | 1 | 3 | ||||||
| TOTALS | 20 | 144 | ||||||
| 13 |
Sales and Marketing
We market Luobuma to consumers primarily by highlighting its unique characteristics— the material is soft like cotton, breathable like hemp and is smooth to the touch like silk, and its FIR-radiating qualities are believed by some to exert a positive effect on various functions of the human body. Very few other companies in China are involved with Luobuma fiber production, so we are chiefly able to market our products against products of natural and man-made fibers that do not have the perceived advantages of Luobuma. The small number of companies that are involved in Luobuma fiber production are still using the traditional, outdated methods of producing Luobuma. We are the only company using advanced technologies. Tenet-Jove’s overall marketing strategy includes:
| ● | Brand marketing strategy, primarily through media publicity, product- and market-oriented strategy; | |
| ● | Distinguishing Luobuma as a high-end, technologically advanced native Chinese product; and | |
| ● | Online advertising, which includes online advertisements appearing on the sites where we sell our products, as well as social media advertising, including Wechat, and direct e-mail solicitations. |
Ankang Longevity Group’s overall marketing strategy of its traditional Chinese medicine products focuses on promoting the Ankang district’s place in traditional Chinese medicine history and its geographic location. First, Ankang City is located in the Han River area between Qinling Mountain and Ba Mountain. Because of its geographic location and favorable climate, it is the principal production location of the ancient Qin medicine, which dates from the Qin dynasty, the first imperial dynasty of China. This area contains more than 1,200 types of herbs and plants used in traditional Chinese medicine and is home to 176 of the 282 types of herbal medicine set forth in The Pharmacopoeia of the People’s Republic of China (PPRC), compiled by the Pharmacopoeia Commission of the Ministry of Health of the PRC. The PPRC is the PRC’s official compendium of drugs, covering traditional Chinese and western medicines. Second, the soil in Ankang City, especially in Ziyang County, which is located in Ziyang District of Ankang City, contains large quantities of selenium. Because the selenium content of food is largely dependent on location and soil conditions, which can vary widely, the tea, traditional Chinese medicines and raw materials produced and cultivated in Ankang contain great amounts of selenium, which is a beneficial nutrient in foods and an important raw material for medicines. Ankang Longevity Group has entered into an agreement with the local government in Ziyang County for the use of an approximately 8,200 acre selenium-rich parcel of farmland and woodlands for the cultivation of herbs and plants — principally houpu magnolia and eucommia — used in traditional Chinese medicine. Selenium has also attracted attention because of its antioxidant properties; antioxidants protect cells from damage.
Ankang Longevity emphasizes the following marketing strategies:
| ● | A focus on the excellent quality of its selenium Chinese herbal medicines; | |
| ● | Establishment of a long-term supply relationships with pharmaceutical companies and hospitals nationwide; and | |
| ● | Developing its Chinese herbal medicine wholesale market and e-commerce platform. |
| 14 |
Finally, the Zhisheng Group emphasizes the following marketing strategies:
| ● | Focusing on the advanced growing conditions provided by our modern greenhouse operations and the potential pharmaceutical byproducts of yew, especially paclitaxel or taxol; and | |
| ● | Brand marketing to focus on our yew’s brand positioning. |
In October 2012, the Company, through its VIEs, Zhisheng Freight and Zhisheng Agricultural, entered into an agreement with an unrelated third party, Zhejiang Zhen’Ai Network Warehousing Services Co., Ltd. (“Zhen’Ai Network”), to invest RMB 14.5 million (approximately $2.4 million) into the Tiancang Systematic Warehousing Project (“Tiancang Project”) operated by Zhen’Ai Network in exchange for a 29% equity interest in this project upon completion. The Tiancang Project is an online platform aiming to provide comprehensive warehousing and logistic solutions for various companies’ e-commerce needs. The Company expects to increase its distribution channels through this platform as well as to benefit from boosting its own revenue by providing synergetic logistic and warehousing services using its existing facilities and a service team in the Eastern port city of Qingdao.
Currently, the Company’s sales are generated through the following five major channels:
| 1. | Retail stores and sales counters. We mainly sell our Luobuma related products through sales counters and medicine through our pharmacy chain stores. | |
| 2. | Sales to group or institutional customers. We mainly sell our organic agricultural products and yew trees to group or corporate customers. | |
| 3. | Seminars and conferences. Because a majority of new consumers need to learn about our new products before buying them, it becomes very important and effective for us to organize or sponsor seminars and events to present healthcare knowledge while introducing and selling our products to new users. | |
| 4. | Wholesale. We mainly sell our decocting-free Chinese herbal medicines to large Chinese medicine resellers and pharmaceutical companies. | |
| 5. | E-commerce. We mainly sell the Luobuma related products through Tmall and Taobao to underdeveloped regions in China, Taiwan and Macau. We are currently one of only three certified online sellers of Luobuma textile products on China’s largest online sales platform, Tmall run by Alibaba. Selling through the Internet has become increasingly important to our sales in undeveloped regions and developed cities. |
The Market
We primarily market our health and wellbeing-focused products in China. At present, we do not sell any of our products in the United States or Canada. On the demand side, we believe that the following four forces drive market growth in all three of our business segments:
| 1. | The rapid growth of China’s economy, which has produced one of the largest groups of middle-class families in the world, with the largest collective purchasing power in the world. The Brookings Institution estimates that by 2030, over 70 percent of China’s population could be middle class, consuming approximately $10 trillion in goods and services. | |
| 2. | The increase of China’s aging population. The China Census Bureau predicts that the majority of the China “baby boom” population (representing 40% of China’s total population) will be 65 or older by 2020, which represents over 500 million potential consumers of our pharmaceutical and healthcare products, the majority of which are sold to older customers. | |
| 3. | Chinese people’s increasing attention and awareness to healthy and active lifestyles, especially in urban areas. | |
| 4. | Chinese healthcare reforms. |
| 15 |
We believe China’s healthcare sector has the capacity to grow in the coming years. From pharmaceuticals to medical products to general consumer health, China remains among the world’s most attractive markets, and by far the fastest-growing of all the large emerging ones. This growth is being driven by China’s aging population, increased incidence of chronic diseases, and a material increase in investment from both domestic and foreign corporations.
China’s healthcare market is being shaped by positive economic and demographic trends, further healthcare reform efforts, and the policies set forth in the government’s 12th five-year plan. We believe that improvements in infrastructure, the broadening of insurance coverage, and government encouragement and support for innovation will have positive implications for us and other healthcare companies.
Strong growth in the Chinese healthcare sector has been fueled by favorable demographic trends, continued urbanization throughout China, the overall Chinese economy’s expansion, and income growth (which encourages a greater awareness of and access to healthcare among Chinese consumers). It also reflects the Chinese government’s focus on healthcare as both a social priority (as witnessed in its late 2000s healthcare reforms) and a strategic priority (as witnessed in the 12th five-year plan’s stated focus on growing the Chinese biomedical industry). From pharmaceuticals to medical devices to traditional Chinese medicine, almost every health sector has benefited.
Competition
We compete with other top-tier pharmaceutical and healthcare companies in China. Many of them are more established than we are and have significantly greater financial, technical, marketing and other resources than we presently possess. Some of our competitors have greater name recognition and a larger customer base. Those competitors may be able to respond more quickly to new or changing opportunities and customer requirements and may be able to undertake more extensive promotional activities, offer more attractive terms to customers, and adopt more aggressive pricing policies. Some of our competitors have also developed similar products that compete with ours.
Our most prominent competitors in China’s textile products market are primarily large-scale textile companies, such as Luolai Home Textile Co., Fuanna Bedding and Furnishing Co., Ltd., Violet Home Textile Co., and Shuixing Home Textile Co., Ltd, as well as Bauerfeind Sports and Albert Medical, makers of protective clothing products similar to our protective clothing products. Our most prominent competitors in our pharmaceutical sales market include Ankang City Zhenning Chinese Medicine Decoction Pieces Co. Ltd. and Zhenping County Chinese Medicine Decoction Pieces Co. Ltd. Our most prominent competitors in China’s agricultural market are Beijing Jinfu Yinong Agricultural Technology Group Co., Ltd. for vegetables and other produce and Shenyang Xincheng Garden Engineering Co., Ltd. for yew trees. In the pharmaceutical sales market, we believe that our competitive position is strong because Shaanxi Pharmacy Holding in which Ankang Longevity Group has a 49% ownership stake, is a participant in the Ankang municipal government’s “Three Unities of Medicine” program, which is designed to facilitate the purchase, sale and delivery of pharmaceuticals. The “Three Unities of Medicine” program refers to “unified procurement price, unified selling price, and unified logistics”. This project is promoted by the Shaanxi Government to regulate the price of medicines in the local market. Under the program, the Shaanxi Pharmacy Holding joint venture directly sells medicines to local institutional purchasers, like hospitals; these institutional purchasers are only permitted to purchase pharmaceuticals from selected preferred providers. After a bidding process, Shaanxi Pharmacy Holding, along with other two companies, were selected by the Ankang municipal government as preferred providers under the program. However, Shaanxi Pharmacy Holding is entitled to cover all counties and districts of Ankang City, and the other two providers only cover portions of Ankang City. This program is valid until 2020, and the participants are subject to an ongoing review of their qualifications by the local pharmaceutical supervision authority every three years until 2020.
| 16 |
Ankang Longevity Group
Ankang Longevity Group competes within its traditional Chinese medicinal products and Western medicine wholesale and retail business primarily against Ankang City Zhenning Chinese Medicine Decoction Pieces Co. Ltd. and Zhenping County Chinese Medicine Decoction Pieces Co. Ltd., which conduct their wholesale pharmaceuticals business in the Ankang area, but Ankang Longevity Group has greater revenues than any of its competitors; its two main competitors’ output value and sales combined are just one-third of Ankang Longevity Group’s. The Ankang Longevity Group has formed a new pharmaceutical company and chain of drug stores with two entities wholly owned by the state-owned Shaanxi Pharmaceutical Group that has resulted in a favorable market position in the Ankang area.
Numerous competitors nationwide, including Ankang City Zhenning Chinese Medicine Decoction Pieces Co. Ltd. and Zhenping County Chinese Medicine Decoction Pieces Co. Ltd., participate in the sale of Chinese medicinal herbs and Chinese medicine decoction pieces; among them are some high-profile and large-scale companies along with some companies that have huge production and storage capacity to influence the market price. Because we believe we are able to obtain early access to and to occupy natural resources for producing selenium Chinese herbal medicines, the Group is especially focusing on those products in order to gain market share.
Zhisheng Group
There are dozens of companies planting and cultivating yew trees in China, some of which are large-scale companies. Shenyang Xincheng Garden Engineering Co., Ltd. is a large agricultural competitor whose main product is yew. Their nurseries have the most mature yew trees in northeast China, and the average age of their yew trees is more than eleven years old. Another competitor, Chongqing Jiangjin District Mansheng Agricultural Development Co., Ltd., has the biggest nursery for young plants in Southwest China. And Jingyin City Hengtu Town Green Industry Yew Base specializes in cultivating, planting, gardening, and technological development of yew trees. They were the first company to introduce taxus media yew trees in China.
Tenet-Jove
There are few viable competitors producing advanced technology textile products with health benefits like our Luobuma textile products. Principally, our competitors are those that market and sell traditional textile products, such as Luolai Home Textile Co., Fuanna Bedding and Furnishing Co., Ltd., Violet Home Textile Co., and Shuixing Home Textile Co., Ltd, as well as those companies that market and sell protective clothing, like Bauerfeind Sports and Albert Medical. Luobuma is native to China, thus our ability to source raw materials locally greatly enhances our competitive position in the Chinese market for high quality textile products with perceived health benefits.
| 17 |
Employees
As of June 30, 2018, we employed a total of 425 full-time and no part-time employees in the following functions.
| Department | June 30, 2018 | |||
| Senior Management | 30 | |||
| Human Resource & Administration | 25 | |||
| Finance | 35 | |||
| Research & Development | 18 | |||
| Production & Procurement | 152 | |||
| Sales & Marketing | 165 | |||
| Total | 425 | |||
Our employees are not represented by a labor organization or covered by a collective bargaining agreement. We have not experienced any work stoppages.
The Company plans to hire additional employees as required. Its management and employees enjoy both compensation and welfare benefits pursuant to Chinese laws. We are required under PRC law to make contributions to employee benefit plans at specified percentages of our after-tax profit. In addition, we are required by PRC law to cover employees in China with various types of social insurance. In 2018, 2017, and 2016, we contributed approximately $146,312, $139,736, and $128,763, respectively, to employee social insurance. The effect on our liquidity by the payments for these contributions is immaterial. We believe that we are in material compliance with the relevant PRC employment laws.
Relevant PRC Regulations
Laws and Regulations in China Regarding Agriculture and Pharmaceutical Products and Distribution
Laws regulating pharmaceutical and healthcare products and agriculture cover a broad array of subjects. We must comply with numerous additional provincial and local laws relating to matters such as safe working conditions, manufacturing practices, environmental protection and fire hazard control. We believe we are in compliance with these laws and regulations in the future in all material respects. We may be required to incur significant costs to comply with these laws and regulations. Unanticipated changes in existing regulatory requirements or adoption of new requirements could materially adversely affect our business, financial condition and results of operations.
Pharmaceutical Administration Law of The People’s Republic of China and its detailed implementation provisions
According to Pharmaceutical Administration Law of The People’s Republic of China effective on December 1, 2001, a Pharmaceutical Trade License is required in order to sell pharmaceutical products and a Pharmaceutical Manufacturer License shall be obtained to manufacture pharmaceutical products. Our license currently expires in April 2019. The regulations indicate the specific procedure to obtain and maintain other licenses, packaging, price control, advertising and relevant penalty.
| 18 |
Notice on Supervision of Manufacture of Chinese Medicine in Pieces
According to Notice on Supervision of Manufacture of Chinese medicine in Pieces on February 1, 2008 and effective on June 1, 2008, companies processing raw Chinese medicine shall be a GMP certified company. Our GMP license currently expires on April 17, 2019.
Standards of Preparation for Chinese Medicine in Pieces
Shaanxi province has issued standards of preparation for Chinese Medicine in Pieces, which became effective on July 1, 2011. Our company is required to follow the procedures designated in the standards during the process of preparing the pieces of herbs.
Food Safety Law of the People’s Republic of China
The Food Safety Law of the People’s Republic of China as adopted at the 7th Session of the Standing Committee of the 11th National People’s Congress of the People’s Republic of China and effective on June 1, 2009, governs the food safety in food production and business operation activities. Pursuant to the Food Safety Law of the People’s Republic of China, food producers must establish an internal inspection and record system for raw materials and pre-delivery products, and food distributors must also establish internal systems to record and inspect food products procured from suppliers. In addition, any food additives that are not in the approved government catalog must not be used and no food products can be sold inspection-free.
Regulations on the Implementation of the Food Safety Law of the People’s Republic of China
The Regulations on the Implementation of the Food Safety Law of the People’s Republic of China as adopted at the 73rd Standing Committee Meeting of the State Council on July 8, 2009 and effective on July 20, 2009, are promulgated in accordance with the Food Safety Law of the People’s Republic of China. The Regulations require that the local People’s Government at or above the county level shall perform the responsibility specified in the Food Safety Law of the People’s Republic of China, improve the ability for supervision and administration of food safety, ensure supervision and administration of food safety; establish and improve the coordination mechanism between food safety regulatory authorities, integrate and improve the food safety information network, and realize the sharing of food safety and food inspection information and other technical resources.
Law of the People’s Republic of China on Quality and Safety of Agricultural Products
The Law of the People’s Republic of China on Quality and Safety of Agricultural Products was adopted at the 21st Meeting of the Standing Committee of the Tenth National People’s Congress on April 29, 2006. This Law was enacted in order to ensure the quality and safety of agricultural products, maintain the health of the general public, and promote the development agriculture and rural economy. Pursuant to this Law, agricultural products distribution enterprises shall establish a sound system of inspection and acceptance for their purchases. In addition, agricultural products that fail to pass the inspection based on the quality and safety standards of agricultural products cannot be marketed.
| 19 |
Regulation on Product Advertisements and Promotion
Article 5 of the Provisions for Health Food Management provides that foods claimed to have health function shall be approved by the Chinese Ministry of Health. The developer or manufacturer shall submit an application to the provincial level health administrative departments where such developer or manufacturer is located. After preliminary examination and approval by Ministry of Health, the Ministry of Health may issue a health food license to the qualified health food. Under Article 21, the label and package insert of health foods shall conform to national standards and requirements and indicate, among other things, its function and suitable users; dosage and administration; storage methods; and active ingredients.
When promoting health foods, the advertisement of health food shall conform to the other regulations. Article 19 of The Advertisement Law of People’s Republic of China provides that “an advertisement for foods, alcoholic drinks or cosmetics must meet requirements for public health, and shall not employ medical jargon or terms liable to confuse them with pharmaceuticals.” In Interim Provisions on Health Food Advertisements Review, Article 4 provides that prior to advertising health foods, developers or manufacturers should first submit an application to the food and drug administration departments on the provincial, autonomous, municipal level under the Central Government. Article 8 provides that publicizing of health functions, active ingredients, content, suitable users, dosage in health food advertisements shall be subject to prior review of the package insert ratified by the food and drug administration departments in the State Council and cannot be changed without permission. Certain content may not appear in health food advertisements, including: a guarantee of its functions; exaggerated claims; jargon, mysterious terms and technical content; promises such as “safe” or “no side effects”; or comprehensive assessment information such as efficiency, cure rate, ranking and awards.
Laws and Regulations Regarding Promotion and Advertisement of Health Textiles
In The Model Code of Health Textiles, “health textiles” refer to textiles without toxic side effects that have far infrared functions, magnetic functions and/or antibacterial effects, and which aim at regulating the body, but not healing illnesses. The Code requires that the effect of health textiles cannot be exaggerated in any form of advertising.
In addition, the promotion of health textiles should comply with The Advertisement Law of People’s Republic of China. Where there are statements in an advertisement on the performance, place of origin, usage, quality, price, producer or manufacturer, or on the items, forms, quality, price and promise of service, such statements shall be clear and explicit.
Regulation on Product Liability
Manufacturers and vendors of defective products in the PRC may incur liability for losses and injuries caused by such products. Under the General Principles of the Civil Laws of the PRC, which became effective on January 1, 1987 and were amended on August 27, 2009, manufacturers or retailers of defective products that cause property damage or physical injury to any person will be subject to civil liability.
In 1993, the General Principles of the PRC Civil Law were supplemented by the Product Quality Law of the PRC (as amended in 2000 and 2009) and the Law of the PRC on the Protection of the Rights and Interests of Consumers (as amended in 2009), which were enacted to protect the legitimate rights and interests of end-users and consumers and to strengthen the supervision and control of the quality of products. If our products are defective and cause any personal injuries or damage to assets, our customers have the right to claim compensation from us.
| 20 |
The PRC Tort Law was promulgated on December 26, 2009 and became effective from July 1, 2010. Under this law, a patient who suffers injury from a defective product can claim damages from either the hospital or medical institution or the manufacturer of the defective product. If our pharmaceutical products injure a patient, for example, and if the patient claims damages from the medical institution, the medical institution is entitled to claim repayment from us. Pursuant to the PRC Tort Law, where a personal injury is caused by a tort, the tortfeasor shall compensate the victim for the reasonable costs and expenses for treatment and rehabilitation, as well as death compensation and funeral costs and expenses if it causes the death of the victim. There is no cap on monetary damages the plaintiffs may seek under the PRC Tort Law.
Regulation of Work Safety
On June 29, 2002, the Work Safety Law of the PRC was adopted by the Standing Committee of the 9th National People’s Congress and came into effect on November 1, 2002, as amended on August 27, 2009. The Work Safety Law provides general work safety requirements for entities engaging in manufacturing and business activities within the PRC. Additionally, Regulation on Work Safety Licenses, as adopted by the State Council on January 7, 2004 and effective on January 13, 2004, requires enterprises engaging in the manufacture of dangerous chemicals to obtain a work safety license with a term of three years. If a work safety license needs to be extended, the enterprise must go through extension procedures with authorities three months prior to its expiration. In addition, on May 17, 2004, the Measures for Implementation of Work Safety Licenses of Dangerous Chemicals Production was promulgated as implementing measures to the Regulation on Work Safety Licenses which provides that entities producing dangerous chemicals are required to obtain work safety licenses pursuant to specific requirements. Without work safety licenses, no entity may engage in the formal manufacture of dangerous chemicals.
The Regulations on the Safety Administration of Dangerous Chemicals was promulgated by the State Council on January 26, 2002, and effective as of March 15, 2002. It sets forth general requirements for manufacturing and the storage of dangerous chemicals in China. The Regulations on the Safety Administration of Dangerous Chemicals requires that companies manufacturing dangerous chemicals establish and strengthen their internal regulations and rules on safety control and fulfill the national standards and other relevant provisions of the State. In addition, according to the Regulations on the Safety Administration of Dangerous Chemicals, companies that manufacture, store, transport or use dangerous chemicals shall be required to obtain corresponding approvals or licenses with the State Administration of Work Safety and its local branches and other proper authorities. Companies that manufacture or store dangerous chemicals without approval or registration with the proper authorities can be shut down, ordered to stop manufacturing or ordered to destroy the dangerous chemicals. Such companies can also be subject to fines. If criminal law is violated, the persons chiefly liable, along with other personnel directly responsible for such impropriety, shall be subject to relevant criminal liability.
Regulation of Foreign Currency Exchange
Foreign currency exchange in the PRC is governed by a series of regulations, including the Foreign Currency Administrative Rules (1996), as amended, and the Administrative Regulations Regarding Settlement, Sale and Payment of Foreign Exchange (1996), as amended. Under these regulations, the Renminbi is freely convertible for trade and service-related foreign exchange transactions, but not for direct investment, loans or investments in securities outside the PRC without the prior approval of China’s State Administration of Foreign Exchange. Pursuant to the Administrative Regulations Regarding Settlement, Sale and Payment of Foreign Exchange (1996), Foreign Investment Entities may purchase foreign exchange without the approval of the State Administration of Foreign Exchange for trade and service-related foreign exchange transactions by providing commercial documents evidencing these transactions. They may also retain foreign exchange, subject to a cap approved by the State Administration of Foreign Exchange, to satisfy foreign exchange liabilities or to pay dividends. However, the relevant Chinese government authorities may limit or eliminate the ability of foreign investment entities to purchase and retain foreign currencies in the future. In addition, foreign exchange transactions for direct investment, loan and investment in securities outside the PRC are still subject to limitations and require approvals from the State Administration of Foreign Exchange.
| 21 |
China has allowed for a more market-based exchange rate to influence the valuation of the Renmenbi versus global currencies, and supported devaluation consistently over the seven months prior to the date of this prospectus. To the extent any of our future revenues are denominated in Renmenbi or other currencies other than the United States dollar, we would be subject to increased risks relating to foreign currency exchange rate fluctuations which could have a material adverse effect on our financial condition and operating results since operating results are reported in United States dollars and significant changes in the exchange rate could materially impact our reported earnings.
Regulation of Foreign Exchange in Certain Onshore and Offshore Transactions
In October 2005, the State Administration of Foreign Exchange issued the Notice on Issues Relating to the Administration of Foreign Exchange in Fund-raising and Return Investment Activities of Domestic Residents Conducted via Offshore Special Purpose Companies, or the State Administration of Foreign Exchange Notice 75, which became effective as of November 1, 2005, and was further supplemented by three implementation notices issued by the State Administration of Foreign Exchange on November 24, 2005, May 29, 2007 and May 20, 2011, respectively. On July 4th, 2014, SAFE amended it as Circular 37 (i.e., SAFE Circular on Issues Relating to the Administration of Foreign Exchange in Offshore Investment and Financing and Round-trip Investment Activities of Domestic Residents Conducted via Offshore Special Purpose Companies issued by SAFE on July 4, 2014). The State Administration of Foreign Exchange Circular states that PRC residents, whether natural or legal persons, must register with the relevant local State Administration of Foreign Exchange branch prior to establishing or taking control of an offshore entity established for the purpose of overseas equity financing involving onshore assets or equity interests held by them. The term “PRC legal person residents” as used in the State Administration of Foreign Exchange Circular 37 refers to those entities with legal person status or other economic organizations established within the territory of the PRC. The term “PRC natural person residents” as used in the State Administration of Foreign Exchange Circular 37 includes all PRC citizens and all other natural persons, including foreigners, who habitually reside in the PRC for economic benefit.
PRC residents are required to complete amended registrations with the local State Administration of Foreign Exchange branch upon: (i) injection of equity interests or assets of an onshore enterprise to the offshore entity, or (ii) subsequent overseas equity financing by such offshore entity. PRC residents are also required to complete amended registrations or filing with the local State Administration of Foreign Exchange branch within 30 days of any material change in the his shareholding or capital of the offshore entity, such as changes in share capital, share transfers and long-term equity or debt investments and merger and split activities.
Regulation on Dividend Distributions
Our PRC subsidiaries are wholly foreign-owned and joint venture enterprises under the PRC law. The principal regulations governing the distribution of dividends paid by wholly foreign-owned enterprises include:
| ● | Corporate Law (1993), as amended in 2005 and 2013; | |
| ● | The Wholly Foreign-Owned Enterprise Law (1986), as amended in 2000; | |
| ● | The Wholly Foreign-Owned Enterprise Law Implementation Regulations (1990), as amended in 2001; and | |
| ● | The Enterprise Income Tax Law (2007) and its Implementation Regulations (2007). |
| 22 |
Under these regulations, wholly foreign-owned and joint venture enterprises in China may pay dividends only out of their accumulated profits, if any, as determined in accordance with PRC accounting standards and regulations. In addition, an enterprise in China is required to set aside at least 10% of its after-tax profit based on PRC accounting standards each year to its general reserves until its cumulative total reserve funds reaches 50% of its registered capital. The board of directors of a wholly foreign-owned enterprise has the discretion to allocate a portion of its after-tax profits to its employee welfare and bonus funds. These reserve funds, however, may not be distributed as cash dividends.
On March 16, 2007, the National People’s Congress enacted the Enterprise Income Tax Law, and on December 6, 2007, the State Council issued the Implementation Regulations on the Enterprise Income Tax Law, both of which became effective on January 1, 2008. Under this law and its implementation regulations, dividends payable by a foreign-invested enterprise in the PRC to its foreign investor who is a non-resident enterprise will be subject to a 10% withholding tax, unless any such foreign investor’s jurisdiction of incorporation has a tax treaty with the PRC that provides for a lower withholding tax rate.
M&A Rules and Regulation on Overseas Listings
On August 8, 2006, six PRC regulatory agencies, the Ministry of Commerce, the State Assets Supervision and Administration Commission, the State Administration for Taxation, the State Administration for Industry and Commerce, the China Securities Regulatory Commission (“CSRC”) and the SAFE, jointly adopted the Regulation on Mergers and Acquisitions of Domestic Enterprises by Foreign Investors, or the M&A Rules, which became effective on September 8, 2006. The M&A Rules purport, among other things, to require that offshore SPVs that are controlled by PRC companies or individuals and that have been formed for overseas listing purposes through acquisitions of PRC domestic interests held by such PRC companies or individuals, obtain the approval of the CSRC prior to publicly listing their securities on an overseas stock exchange. On September 21, 2006, the CSRC published a notice on its official website specifying documents and materials required to be submitted to it by SPVs seeking CSRC approval of their overseas listings.
While the application of the M&A Rules remains unclear, our PRC counsel have advised us that, based on their understanding of the current PRC laws and regulations as well as the notice announced on September 21, 2006:
| ● | the CSRC currently has not issued any definitive rule or interpretation concerning whether offerings such as our initial public offering are subject to the CSRC approval procedures under the M&A Rules; | |
| ● | despite the lack of any definitive rule or interpretation from CSRC, the main purpose of the M&A rule is for national security and national industrial policy and so far none of the Chinese companies that have completed their public listing in the U.S. have obtained such approval; and | |
| ● | our Delaware company is not controlled by a Chinese citizen. Accordingly, although the purpose of Delaware incorporation is for overseas listing, the M&A rule should not apply to us. |
| 23 |
Our PRC counsel also advises us, however, that there is still uncertainty as to how the M&A Rules will be interpreted and implemented. If the CSRC or other PRC regulatory agencies, subsequently determine that CSRC approval was required for our initial public offering, we may need to apply for remedial approval from the CSRC and we may be subject to penalties and administrative sanctions administered by these regulatory agencies. These regulatory agencies may impose fines and penalties on our operations in the PRC, limit our operating privileges in the PRC, delay or restrict the repatriation of the proceeds from our initial public offering into the PRC, or take other actions that could materially adversely affect our business, financial condition, results of operations, reputation and prospects, as well as the trading price of our common stock.
In addition, if the CSRC later requires that we obtain its approval for our initial public offering, we may be unable to obtain a waiver of the CSRC approval requirements, if and when procedures are established to obtain such a waiver. Any uncertainties or negative publicity regarding the CSRC approval requirements could have a material adverse effect on the trading price of our common stock.
Restriction on Foreign Ownership
The Foreign Investment Industrial Catalogue jointly issued by the Ministry of Commerce for the People’s Republic of China and the National Development and Reform Commission in 2011 classified various industries/business into three different categories: (i) encouraged for foreign investment; (ii) restricted to foreign investment; and (iii) prohibited from foreign investment. For any industry/business not covered by any of these three categories, they will be deemed industries/business permitted to have foreign investment. Except for those expressly provided restrictions, encouraged and permitted industries/business are usually 100% open to foreign investment and ownership. With regard to those industries/business restricted to or prohibited from foreign investment, there is always a limitation on foreign investment and ownership. The decoction business conducted by Ankang Longevity Group falls under the industry categories that are prohibited from foreign investment and thus is subject to limitation on foreign investment and ownership. None of the other business activities conducted by us fall under industry categories that are restricted to, or prohibited from foreign investment.
Regulations on Offshore Parent Holding Companies’ Direct Investment in and Loans to Their PRC Subsidiaries
An offshore company may invest equity in a PRC company, which will become the PRC subsidiary of the offshore holding company after investment. Such equity investment is subject to a series of laws and regulations generally applicable to any foreign-invested enterprise in China, which include the Wholly Foreign Owned Enterprise Law, the Sino-foreign Equity Joint Venture Enterprise Law, the Sino-foreign Contractual Joint Venture Enterprise Law, all as amended from time to time, and their respective implementing rules; the Tentative Provisions on the Foreign Exchange Registration Administration of Foreign-Invested Enterprise; and the Notice on Certain Matters Relating to the Change of Registered Capital of Foreign-Invested Enterprises.
Under the aforesaid laws and regulations, the increase of the registered capital of a foreign-invested enterprise is subject to the prior approval by the original approval authority of its establishment. In addition, the increase of registered capital and total investment amount shall both be registered with the State Administration for Industry and Commerce (“SAIC”) and SAFE.
Shareholder loans made by offshore parent holding companies to their PRC subsidiaries are regarded as foreign debts in China for regulatory purposes, which debts are subject to a number of PRC laws and regulations, including the PRC Foreign Exchange Administration Regulations, the Interim Measures on Administration on Foreign Debts, the Tentative Provisions on the Statistics Monitoring of Foreign Debts and its implementation rules, and the Administration Rules on the Settlement, Sale and Payment of Foreign Exchange.
| 24 |
Under these regulations, the shareholder loans made by offshore parent holding companies to their PRC subsidiaries shall be registered with SAFE. Furthermore, the total amount of foreign debts that can be incurred by such PRC subsidiaries, including any shareholder loans, shall not exceed the difference between the total investment amount and the registered capital amount of the PRC subsidiaries, both of which are subject to governmental approval.
Regulations on Trademarks
Trademarks are protected by the PRC Trademark Law adopted in 1982, as subsequently amended, as well as the Implementation Regulations of the PRC Trademark Law adopted by the State Council in 2002 and 2013. The Trademark Office under the SAIC handles trademark registrations. Trademarks can be registered for a term of ten years and can be extended for another ten years if requested upon expiration of the first or any renewed ten-year term. The PRC Trademark Law has adopted a “first-to-file” principle with respect to trademark registration. Where a trademark for which a registration application has been made is identical or similar to another trademark which has already been registered or been subject to a preliminary examination and approval for use on the same type of or similar commodities or services, the application for such trademark registration may be rejected. Any person applying for the registration of a trademark may not prejudice the existing right first obtained by others, nor may any person register in advance a trademark that has already been used by another party and has already gained a “sufficient degree of reputation” through such other party’s use. Trademark license agreements must be filed with the Trademark Office or its regional offices.
Regulations on Patents
The PRC Patent Law provides for patentable inventions, utility models and designs, which must meet three conditions: novelty, inventiveness and practical applicability. The State Intellectual Property Office is responsible for examining and approving patent applications. A patent is valid for a term of twenty years in the case of an invention and a term of ten years in the case of utility models and designs. Presently the Company holds a patent in the People’s Republic of China for Luobuma fiber yarn preparation and an application method (patent number: 201110429362.9), which patent is a critical patent used for our high-pressure steam degumming process project.
PRC Enterprise Income Tax Law and Individual Income Tax Law
Under the Enterprise Income Tax Law or EIT Law, enterprises are classified as resident enterprises and non-resident enterprises. PRC resident enterprises typically pay an enterprise income tax at the rate of 25%. An enterprise established outside of the PRC with its “de facto management bodies” located within the PRC is considered a “resident enterprise,” meaning that it can be treated in a manner similar to a PRC domestic enterprise for enterprise income tax purposes. The implementation rules of the EIT Law define “de facto management body” as a managing body that in practice exercises “substantial and overall management and control over the production and operations, personnel, accounting, and properties” of the enterprise.
The SAT Circular 82 issued by the SAT in April 2009 provides certain specific criteria for determining whether the “de facto management body” of a PRC-controlled offshore incorporated enterprise is located in China. Pursuant to the SAT Circular 82, a PRC-controlled offshore incorporated enterprise has its “de facto management body” in China only if all of the following conditions are met: (a) the senior management and core management departments in charge of its daily operations function have their presence mainly in the PRC; (b) its financial and human resources decisions are subject to determination or approval by persons or bodies in the PRC; (c) its major assets, accounting books, company seals, and minutes and files of its board and shareholders’ meetings are located or kept in the PRC; and (d) more than half of the enterprise’s directors or senior management with voting rights habitually reside in the PRC. The SAT Bulletin 45, in effect from September 2011, provides more guidance on the implementation of the SAT Circular 82 and provides for procedures and administration details on determining resident status and administration on post-determination matters. Although the SAT Circular 82 and the SAT Bulletin 45 only apply to offshore enterprises controlled by PRC enterprises or PRC enterprise groups, not those controlled by PRC individuals or foreign individuals, the determining criteria set forth there may reflect the SAT’s general position on how the “de facto management body” test should be applied in determining the tax resident status of offshore enterprises, regardless of whether they are controlled by PRC enterprises or PRC enterprise groups or by PRC or foreign individuals.
| 25 |
Due to the lack of applicable legal precedents, it remains unclear how the PRC tax authorities will determine the PRC tax resident treatment of a foreign company controlled by individuals in the PRC. We may be classified as a PRC “resident enterprise” for PRC enterprise income tax purposes. Such classification would likely result in unfavorable tax consequences to us and our non-PRC shareholders, and would have a material adverse effect on our results of operations and the value of your investment.
PRC Value Added Tax
Pursuant to the Provisional Regulation of China on Value Added Tax, all entities and individuals that are engaged in the businesses of sales of goods, provision of repair and placement services and importation of goods into China are generally subject to a VAT at a rate of 17% (with the exception of certain goods which are subject to a rate of 13%) of the gross sales proceeds received, less any VAT already paid or borne by the taxpayer on the goods or services purchased by it and utilized in the production of goods or provisions of services that have generated the gross sales proceeds.
PRC Business Tax
Companies in China were generally subject to business tax and related surcharges by various local tax authorities at rates ranging from 3% to 20% on revenue generated from providing services and revenue generated from the transfer of intangibles. Beginning on May 1, 2016, value added tax replaced the existing business tax in China, and there is no longer business tax imposed in China.
Employment Laws
In accordance with the PRC National Labor Law and the PRC Labor Contract Law, employers must execute written labor contracts with full-time employees in order to establish an employment relationship. All employers must compensate their employees equal to at least the local minimum wage standards. All employers are required to establish a system for labor safety and sanitation, strictly abide by state rules and standards and provide employees with appropriate workplace safety training. In addition, employers in China are obliged to pay contributions to the social insurance plan and the housing fund plan for employees.
We have not entered into employment agreements with any of our executive officers. We have contributed to the basic and minimum social insurance plan. While we believe we have made adequate provision of such outstanding amounts of contributions to such plans in our audited financial statements, any failure to make sufficient payments to such plans would be in violation of applicable PRC laws and regulations and, if we are found to be in violation of such laws and regulations, we could be required to make up the contributions for such plans as well as to pay late fees and fines.
Regulations on Environmental Protection
According to the Prevention and Control of Water Pollution Law, China adopted a licensing system for pollutant discharge. Companies directly or indirectly responsible for discharge of industrial waste water or medical sewage to waters shall be required to obtain a pollutant discharge license. All companies are prohibited from discharging wastewater and sewage to waters without or in violation of the terms of the pollutant discharge license.
The Regulations on the Administration of Construction Projects Environmental Protection governs construction projects and the impact such projects will have on the environment. Pursuant to the Regulations on the Administration of Construction Projects Environmental Protection, the governing body is responsible for supervising the implementation of a three tiered system that includes (i) reviewing and approving a construction project, (ii) overseeing the construction project and (iii) to inspect the finished construction project and ensure that all harmful pollutants are disposed of correctly. Manufacturing companies are required to apply for inspection with environment protection authorities upon completion of a construction project.
| 26 |
| Item 1a. | Risk Factors |
The Company is not required to provide the information required by this Item because the Company is a smaller reporting company.
| Item 1b. | Unresolved Staff Comments |
None.
| Item 2. | Properties |
According to Chinese laws and regulations regarding land usage rights, land in urban districts is owned by the State, while land in the rural areas and suburban areas, except as otherwise provided for by the State, is collectively owned by individuals designated as resident farmers by the State. Also, in accordance with the legal principle that land ownership is separate from the right to the use of the land, the State assigns land usage rights to land users for a certain number of years in return for the payment of fees. The maximum term with respect to the assigned land usage right is 50 years for industrial purposes and 40 years for commercial purposes.
Because the period of land usage is quite long, can be renewed, enables its users to transfer, lease, or mortgage the land usage right, or use it for other economic activities, and the lawful rights and interests are protected by the laws of the State, in common practice, we consider or refer to the right of land usage below for certain properties as an asset “owned” by the company. None of our properties are encumbered by debt, and we are not aware of any environmental concerns or limitations on the use of our properties for the purposes we currently use them or intend to use them in the future. Following is a list of our properties, all of which we lease or for which we have land use rights:
| Property Description | Address | Rental/ownership Term |
Space | |||
| Office—leased out to an unrelated third party—Tenet-Jove serves as the lessor. | Room B-3106, Jianwai SOHO, 39 East Middle Third Ring Road, Chaoyang District, Beijing | Company owns the property right | 280 square meters | |||
| Office— Tenet-Jove serves as the lessee. | T5 1001, Dazu Plaza, No.2, South of Ronghua Road , Yizhuang Economic and technological development zone, Beijing | 3 years (October 1o, 2016 - October 9, 2019) | 480.20 square meters | |||
| Office— Qingdao Zhihesheng Agricultural Produce Services Co., Ltd (General office); Yantai Zhisheng International Trade Co., Ltd. | 766-43 Wangsha Road, Chengyang District, Qingdao City | 5
years (March 1,2014- February 28, 2019) |
234.16 square meters | |||
| Office— Qingdao Zhihesheng Agricultural Produce Services Co., Ltd (General office); Yantai Zhisheng International Trade Co., Ltd. | 766-43 Wangsha Road, Chengyang District, Qingdao City | 5
years (March 1,2014- February 28, 2019) |
234.16 square meters | |||
| Office— Qingdao Zhihesheng Agricultural Produce Services Co., Ltd. (Farmland) | Qingfeng
Community, Xifu Town Chengyang District, Qingdao City |
8
years (October 1, 2013 - September 30, 2018) |
282,141 square meters | |||
| Factory— Yantai Mouping District Zhisheng Agricultural Produce Cooperative* | Gaoling Village, Muping District, Yantai City | 30
years (April 27, 2011 - April 26, 2041) |
13,333 square meters | |||
| Office— Ankang Longevity Pharmaceutical (Group) Co., Ltd. | 31 Daqiao Road, Hanbin District, Ankang City | 3
year (June 20, 2016 – June 19, 2019) |
1,300 square meters | |||
| Warehouse— Ankang Longevity —Pharmaceutical Group Pharmaceutical Industry Co., Ltd. (Medicine Logistics Warehouse) (Construction of medicine logistics and warehouse project) | Chenjiagou Village, New City Office, Hanbin District, Ankang City | Company owns the property right | 5,530 square meters | |||
| Office— Ankang Longevity Pharmaceutical Group Pharmaceutical Industry Co., Ltd. | Second floor, 31 Daqiao Road, Hanbin District, Ankang City | 3
year (June 20, 2016 – June 19, 2019) |
400 square meters | |||
| 27 |
| Property Description | Address | Rental/ownership Term |
Space | |||
| Dormitory— Ankang Longevity Pharmaceutical Group Chain Co. Ltd. (Land) (General) | 15 East Xing’an Road, Ankang City | Company owns the property right | 2,750 square meters | |||
| Office— Ankang Longevity Pharmaceutical Group Chain Co. Ltd. (Land) (Commercial land) | 36 Shidi Street, Ankang City | Company owns the property right | 1,543 square meters | |||
| Office— Ankang Longevity Pharmaceutical Group Chain Co. Ltd. | 31 Daqiao Road, Hanbin District, Ankang City
|
3
year (June 20, 2016 – June 19, 2019) |
500 square meters | |||
| Office— Ankang Longevity Pharmaceutical (Group) Traditional Chinese Medicine Decoction Pieces Co., Ltd. (Offices) (Land) | Minxing Village, Wuli Town, Hanbin District, Ankang City | Company owns the property right | 1,733 square meters | |||
| Production facility— Ankang Longevity Pharmaceutical (Group) Traditional Chinese Medicine Decoction Pieces Co., Ltd. (Industrial use) (Land) | Minxing Village, Wuli Town, Hanbin District, Ankang City | Company owns the property right | 33,545 square meters | |||
| Office— Ankang Longevity Pharmaceutical (Group) Traditional Chinese Medicine Decoction Pieces Co., Ltd. (Offices) (Buildings) (Mixed type, five-story) | Minxing Village, Wuli Town, Hanbin District, Ankang City | Company owns the property right | 3,672 square meters | |||
| Production facility— Ankang Longevity Pharmaceutical (Group) Traditional Chinese Medicine Decoction Pieces Co., Ltd. (Industrial use) (Buildings) (Mixed type, one-story) | Minxing Village, Wuli Town, Hanbin District, Ankang City | Company owns the property right | 3,596 square meters | |||
| Production facility— Qingdao Zhihesheng Agricultural Produce Services Co., Ltd., Yantai Mouping District Zhisheng Agricultural Produce Cooperative (Agricultural use)* | Mafang Town, Pinggu District, Beijing | 18
years (August 31, 2012- August 31, 2030) |
26,666 square meters | |||
| Production facility— Qingdao Zhihesheng Agricultural Produce Services Co., Ltd., Yantai Mouping District Zhisheng Agricultural Produce Cooperative (Agricultural use) | South of Bridge, Jixiang Temple, Xiangnaixi Village, Cuigezhuang, Chaoyang District, Beijing | 12 years (August 1, 2012- July 31, 2024) |
73,333 square meters | |||
| * | Zhisheng Freight took over the lease following the deregistration of Zhisheng Agricultural in 2017. |
| Item 3. | Legal Proceedings |
Other than ordinary routine litigation (of which we are not currently involved), we know of no material, existing or pending legal proceedings against us, nor are we involved as a plaintiff in any material proceeding or pending litigation, and there are no proceedings in which any of our directors, officers or affiliates, or any registered or beneficial stockholder, is an adverse party or has a material interest adverse to our company except as set forth below:
On May 16, 2017, Bonwick Capital Partners, LLC (“Plaintiff”) commenced a lawsuit (Case No. 1:17-cv-03681-PGG) against the Company in the United States District Court for the Southern District of New York. Plaintiff alleges that the Company entered into an agreement with Plaintiff (the “Agreement”), pursuant to which Plaintiff was to provide the Company with financial advisory services in connection with the Company’s initial public offering in the United States. Plaintiff alleges that the Company breached the Agreement and seeks money damages up to $6 million. The Company believes that these claims are without merit and intends to vigorously defend itself.
| Item 4. | Mine Safety Disclosures |
The information required by Item 4 is not applicable to us, as we have no mining operations in the United States.
| 28 |
| Item 5. | Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchase of Equity Securities |
Market Information
On September 27, 2016, we completed an initial public offering of 1,713,190 shares of common stock at a $4.50 offering price. Our common stock started trading on the NASDAQ Capital Market under the symbol of TYHT on September 28, 2016. The quotations reflect inter-dealer prices, without retail mark-up, mark-down or commission, and may not represent actual transactions.
| FISCAL YEAR 2018 | HIGH | LOW | ||||||
| First Quarter | $ | 3.70 | $ | 1.72 | ||||
| Second Quarter | $ | 4.60 | $ | 2.11 | ||||
| Third Quarter | $ | 2.58 | $ | 1.31 | ||||
| Fourth Quarter | $ | 2.47 | $ | 1.40 | ||||
| FISCAL YEAR 2017 | ||||||||
| First Quarter | $ | 34.88 | $ | 7.36 | ||||
| Second Quarter | $ | 11.00 | $ | 3.39 | ||||
| Third Quarter | $ | 5.29 | $ | 3.02 | ||||
| Fourth Quarter | $ | 5.19 | $ | 2.90 | ||||
| 29 |
Holders
As of October 15, 2018, there were 269 registered holders of record of our common stock.
Dividends
We anticipate that we will retain any earnings to support operations and to finance the growth and development of our business. Therefore, we do not expect to pay cash dividends in the foreseeable future. Any future determination relating to our dividend policy will be made at the discretion of our Board of Directors and will depend on a number of factors, including future earnings, capital requirements, financial conditions and future prospects and other factors the Board of Directors may deem relevant. Furthermore, our ability to pay dividends is limited by the Delaware General Corporation Law, which provides that a corporation may only pay dividends out of existing “surplus,” which is defined as the amount by which a corporation’s net assets exceeds its stated capital.
During the current fiscal year and the two most recent completed fiscal years, we did not declare or pay any cash dividends on our shares of common stock, and we do not expect to pay cash dividends in the foreseeable future. If we determine to pay dividends on any of our common stock in the future, as a holding company, we will be dependent principally on receipt of funds from our operating subsidiaries. Current PRC regulations permit our PRC subsidiaries to pay dividends to Shineco only out of their accumulated profits, if any, determined in accordance with Chinese accounting standards and regulations. In addition, each of our subsidiaries in China is required to set aside at least 10% of its after-tax profits each year, if any, to fund a statutory reserve until such reserve reaches 50% of its registered capital. Each of such entity in China is also required to further set aside a portion of its after-tax profits to fund the employee welfare fund, although the amount to be set aside, if any, is determined at the discretion of its board of directors. Although the statutory reserves can be used, among other ways, to increase the registered capital and eliminate future losses in excess of retained earnings of the respective companies, the reserve funds are not distributable as cash dividends except in the event of liquidation.
In addition, pursuant to the EIT Law and its implementation rules, dividends generated after January 1, 2008 and distributed to us by our PRC subsidiaries are subject to withholding tax at a rate of 10% unless otherwise exempted or reduced according to treaties or arrangements between the PRC central government and governments of other countries or regions where the non-PRC-resident enterprises are incorporated.
Under existing PRC foreign exchange regulations, payments of current account items, including profit distributions, interest payments and trade and service-related foreign exchange transactions, can be made in foreign currencies without prior approval of SAFE, by complying with certain procedural requirements. Specifically, under the existing exchange restrictions, without prior approval of SAFE, cash generated from the operations in China may be used to pay dividends to our company.
Securities Authorized for Issuance under Equity Compensation Plans
During the fiscal year ended June 30, 2018, the Company has not adopted any incentive plan.
Recent Sales of Unregistered Securities
We have not sold any equity securities during the period covered by this Report.
| 30 |
| Item 6. | Selected Financial Data |
The Company is not required to provide the information required by this Item because the Company is a smaller reporting company.
| ITEM 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITIONS AND RESULTS OF OPERATIONS |
The following discussion and analysis of the results of our operations and financial condition for the years ended June 30, 2018 and 2017 should be read in conjunction with our consolidated financial statements, and the notes to those consolidated financial statements that are included elsewhere in this Report All monetary figures are presented in U.S. dollars, unless otherwise indicated.
Forward-Looking Statements
The statements in this discussion that are not historical facts are “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended, and are subject to the “safe harbor” created by those sections The words “may,” “will,” “expect,” “believe,” “anticipate,” “intend,” “could,” “estimate,” “continue,” the negative forms thereof, or similar expressions, are intended to identify forward-looking statements, although not all forward-looking statements are identified by those words or expressions. Forward-looking statements by their nature involve substantial risks and uncertainties, certain of which are beyond our control. Actual results, performance or achievements may differ materially from those expressed or implied by forward-looking statements depending on a variety of important factors, including, but not limited to, weather, local, regional, national and global Luobuma and herbal medicines price fluctuations, availability of financing and interest rates, competition, changes in, or failure to comply with, government regulations, costs, uncertainties and other effects of legal and other administrative proceedings, and other risks and uncertainties. . Actual results and the timing of the events may differ materially from those contained in these forward looking statements due to many factors, including those discussed in the “Forward-Looking Statements” set forth elsewhere in this quarterly report on Form 10-Q. We are not undertaking to update or revise any forward-looking statement, whether as a result of new information, future events or circumstances or otherwise.
Business Overview and Corporate Structure
Shineco, Inc. (the “Company”, “we”, “us” and “our”) was incorporated in the State of Delaware on August 20, 1997. On December 30, 2004, the Company acquired all of the issued and outstanding shares of Beijing Tenet-Jove Technological Development Co., Ltd. (“Tenet-Jove”), a PRC company, in exchange for our restricted shares of common stock. Consequently, Tenet-Jove became our 100% owned subsidiary and its operating business became that of the Company. Tenet-Jove was incorporated on December 15, 2003 under the laws of China and was officially granted the status of a Wholly Foreign-Owned Entity (“WFOE”) by Chinese authorities on July 14, 2006. This transaction was accounted for as a recapitalization. Tenet-Jove owns a 90% interest of Tianjin Tenet Huatai Technological Development Co., Ltd. (“Tenet Huatai”).
On December 31, 2008, June 11, 2011 and May 24, 2012, Tenet-Jove entered into a series of contractual agreements including an Executive Business Cooperation Agreement, a Timely Reporting Agreement, an Equity Interest Pledge Agreement and Executive Option Agreement (collectively, the “VIE Agreements”), with each one of the following entities, Ankang Longevity Pharmaceutical (Group) Co., Ltd. (“Ankang Longevity Group”), Yantai Zhisheng International Freight Forwarding Co., Ltd. (“Zhisheng Freight”), Yantai Zhisheng International Trade Co., Ltd. (“Zhisheng Trade”), Yantai Mouping District Zhisheng Agricultural Produce Cooperative (“Zhisheng Agricultural”) and Qingdao Zhihesheng Agricultural Produce Services., Ltd. (“Qingdao Zhihesheng”). On February 24, 2014, Tenet-Jove entered into the same series of contractual agreements with Shineco Zhisheng (Beijing) Bio-Technology Co., Ltd. (“Zhisheng Bio-Tech”), which was incorporated in 2014. Zhisheng Bio-Tech, Zhisheng Freight, Zhisheng Trade, Zhisheng Agricultural, and Qingdao Zhihesheng are collectively referred to herein as the “Zhisheng Group”. Zhisheng Agricultural has not had any significant business activities and thus we have deregistered it in 2017. We have transferred all assets, rights and liabilities to an affiliated entity, Zhisheng Freight.
| 31 |
Pursuant to the VIE Agreements, Tenet-Jove has the exclusive right to provide to each of the Zhisheng Group entities and Ankang Longevity Group consulting services related to their business operations and management. All these contractual agreements obligate Tenet-Jove to absorb a majority of the risk of loss from each of the Zhisheng Group entities and Ankang Longevity Group’s activities and entitle Tenet-Jove to receive a majority of their residual returns. In essence, Tenet-Jove has gained effective control over each of the Zhisheng Group and Ankang Longevity Group. Based on these contractual arrangements, the Zhisheng Group and Ankang Longevity Group are treated as Variable Interest Entities (“VIEs”) under Financial Accounting Standards Board (“FASB”) Accounting Standards Codification (“ASC”) 810 “Consolidation”. Accordingly, the accounts of each of the Zhisheng Group entities and Ankang Longevity Group are consolidated with those of Tenet-Jove. Ankang Longevity Group has several subsidiaries. We carry out all of our business in China through our PRC subsidiaries, our VIEs and their subsidiaries.
On April 19, 2017, Tenet-Jove established Xinjiang Tiankunrunze Biological Engineering Co., Ltd. (“Tiankunrunze”) with registered capital of RMB 50.0 million (US$ 7,262,000) and owns 65% interest of Tiankunrunze. On April 28, 2017, Tiankunrunze established Xinjiang Tianzhuo Technology Development Co., Ltd. (“Tianzhuo”) with registered capital of RMB 10.0 million (US$ 1,450,233). On May 22, 2017, Tiankunrunze established Xinjiang Tianhuihechuang Agriculture Development Co., Ltd. (“Tianhuihechuang”) with registered capital of RMB 10.0 million (US$ 1,452,294). On May 23, 2017, Tiankunrunze established Xinjiang Tianxintongye Biotechnology Development Co., Ltd. (“Tianxintongye”) with registered capital of RMB 10.0 million (US$ 1,451,615). Therefore, Tenet-Jove controls Tiankunrunze and its wholly owned subsidiaries.
On May 2, 2017, the Company entered into a Strategic Cooperation Agreement with Beijing Zhongke Biorefinery Engineering Technology Co., Ltd. (“Biorefinery”), a leading high-tech biomass refining company financially backed by the Chinese Academy of Sciences Institute of Process Engineering, to establish the Institute of Chinese Apocynum Industrial Technology Research (“ICAITR”). Pursuant to the Strategic Cooperation Agreement the two parties agreed to establish the ICAITR and each will own 80% and 20% of the equity interests of ICAITR, respectively. Shineco invested RMB 5.0 million (US$ 737,745) as the registered capital, and Biorefinery will invest a technology patent for “Steam Explosion Degumming”.
On September 21, 2017, the Company, through its wholly owned subsidiary Tenet-Jove, entered into a Strategic Cooperation Agreement (the “Agreement”) with Mr. Jianjun Wang, who is experienced in apocynum planting, manufacturing and knowledgeable in apocynum market and administration procedures with relevant authorities in apocynum industry in China, to establish an Apocynum Industrial Park in Xinjiang, China. Pursuant to the Agreement entered into on September 21, 2017, both parties have agreed to establish a new company, namely, Xinjiang Shineco Taihe Agriculture Technology Ltd. (“Xinjiang Taihe”) to hold and operate the Apocynum Industrial Park, with a total investment of RMB 50 million (approximately US$ 7.57 million), of which the Company will invest RMB 47.5 million and Mr. Wang will invest RMB 2.5 million. Upon the closing of the Agreement, Shineco will own 95% of the equity interest of Xinjiang Taihe.
On September 30, 2017, Tenet-Jove established Xinjiang Shineco Taihe Agriculture Technology Ltd. (“Xinjiang Taihe”) with registered capital of RMB 10.0 million (US$ 1,502,650). On September 30, 2017, Tenet-Jove established Xinjiang Tianyi Runze Bioengineering Co., Ltd. (“Runze”) with registered capital of RMB 10.0 million (US$ 1,502,650). Xinjiang Taihe and Runze became wholly-owned subsidiaries of Tenet-Jove.
On December 10, 2016, Tenet-Jove entered into a purchase agreement with Tianjin Tajite, an online e-commerce company based in Tianjin, China, specializing in distributing Luobuma related products and branded products of Daiso 100-yen shops, pursuant to which Tenet-Jove would acquire a 51% equity interest in Tianjin Tajite for cash consideration of RMB 14,000,000 (approximately US$ 2.1 million). On December 25, 2016, the Company paid the full amount as the deposit to secure the deal. In May, 2017, the Company amended the agreement that required Tianjin Tajite to satisfy certain preconditions related to product introductions into China. On October 26, 2017, the Company completed the acquisition for 51% of the equity interest in Tianjin Tajite.
| 32 |
On October 27, 2017, the Company, through its subsidiary Tianjin Tajite E-Commerce Co., Ltd. (“Tianjin Tajite”), obtained contractual rights to distribute branded products of Daiso Industries Co., Ltd. (“Daiso”), a large franchise of 100-yen shops founded in Japan, via JD.com (“JD”), one of the largest e-commerce companies and one of the largest retailers in China. On November 3, 2017, the Company further developed the cooperation with Daiso by entering into a supply and purchase agreement (the “Daiso Agreement”) for the purpose of establishing a continuous supply and sale of Daiso’s products in China. Pursuant to the Daiso Agreement, the Company planned to purchase Daiso Products in the amount of approximately RMB 20 million by August, 2018 and add orders as circumstance requires. The term of the Daiso Agreement is for one year, and it renews for an additional one-year at the end of each term unless terminated by written notice by either Tianjin Tajite or Daiso. Due to the policy of China Customs, many of the bestselling products of Daiso are not allowed to be imported through general form of trade model, but only through cross-border e-commence business model. As a result, the Company suspends the cooperation with Daiso temporarily and waits for the opening of the China-Japan-South Korea Free Trade Zone.
On November 1, 2017, the Company established an Apocynum Industrial Park in Xinjiang, China.
The Company, through its subsidiary, Xinjiang Taihe has entered into a definitive Share Exchange and Acquisition Agreement (the “Xinjiang Tiansheng Agreement”) with Western Xinjiang Tiansheng Agricultural Development Co., Ltd (“Xinjiang Tiansheng”). Pursuant to the Xinjiang Tiansheng Agreement, Xinjiang Taihe will receive 51% equity ownership in Xinjiang Tiansheng for further investment in apocynum business expansion in Xinjiang, China, in exchange for a combination of 14% equity ownership in Xinjiang Taihe and cash payments in three separate installments (the “Acquisition Consideration”). The first installment in the amount of RMB 810,000 was paid to Xinjiang Tiansheng (the “Xinjiang Tiansheng Deposit”). The Acquisition Consideration in the aggregate is valued at RMB 23.8 million contingent upon certain milestones in the next years. The Company and Xinjiang Tiansheng terminated the Xinjiang Tiansheng Agreement on July 10, 2018 and Xinjiang Tiansheng returned the full Xinjiang Tiansheng Deposit following such termination by the end of July 2018.
Currently, we have three main business segments: (i) Tenet-Jove is engaged in the developing, manufacturing and selling of Bluish Dogbane and related products, also known in Chinese as “Luobuma,” including therapeutic clothing and textile products made from Luobuma , as well as purchasing Luoboma raw materials processing; (ii) Zhisheng Group is engaged in the business of planting, processing and distributing of green agricultural produce as well as providing domestic and international logistic services for agricultural products (“Agricultural Products”); and, (iii) Ankang Longevity manufactures traditional Chinese medicinal herbal products as well as other retail pharmaceutical products. These different business activities and products can potentially be integrated and benefit from one and other.
Financing Activities and Subsequent Events
On January 23, 2018, the Company entered into a Common Stock Purchase Agreement (the “Purchase Agreement”) with IFG OPPORTUNITY FUND LLC (“IFG Fund”) whereby, upon the terms and subject to the conditions and limitations set forth therein, the Company has the right, from time to time in its sole discretion during the 24-month term of the Purchase Agreement, to direct IFG Fund to purchase up to a total of $15,000,000 of shares of Common Stock and an additional 200,000 shares of Common Stock (the “Commitment Shares”) as consideration for IFG to enter into the Purchase Agreement. The Company and IFG Fund, on January 23, 2018, entered into a Registration Rights Agreement for certain registration rights in connection with the Purchase Agreement (the “Registration Rights Agreement”). The IFG Fund offering was made pursuant to a prospectus supplement dated and filed with the Securities and Exchange Commission (“SEC”) on January 26, 2018 (the “Prospectus Supplement”) and an accompanying prospectus dated November 21, 2017, under the Company’s shelf registration statement on Form S-3 declared effective by the SEC on December 19, 2017 (File No. 333-221711) (the “Registration Statement”). On January 23, 2018, the Company issued the Commitment Shares to IFG Fund. On July 3, 2018, the Company and IFG Fund entered into a termination agreement, dated July 3, 2018 (the “Termination Agreement”) effective as of July 3, 2018, to terminate the Purchase Agreement and the Registration Rights Agreement. As of the date of this annual report, shares pursuant to the Equity Line have been issued to IFG by the Company (except for the Commitment Shares).
On September 27, 2018, the Company entered into a securities purchase agreement with selected investors whereby the Company agreed to sell up to 1,637,700 of common stock at a purchase price of $1 per Share, for gross proceeds to the Company of approximately $1,637,700 (the “2018 Offering”). The 2018 Offering closed on September 28, 2018. The 2018 Offering was made pursuant to the Company’s effective registration statement on Form S-3 (Registration Statement No. 333-221711) previously filed with the Securities and Exchange Commission and a prospectus supplement thereunder.
| 33 |
Factors Affecting Financial Performance
We believe that the following factors will affect our financial performance:
Increasing demand for our products - The increasing demand for our Chinese medicinal herbal products and our agricultural products will have a positive impact on our financial position. We plan to develop new products and expand our distribution network as well as to grow our business through possible mergers and acquisitions of similar or synergetic businesses, all aimed at increasing awareness of our brand, developing customer loyalty, meeting customer demands in various markets and providing solid foundations for our continuous growth. As of the date of this Report however, we do not have any agreements, undertakings or understandings to acquire any such entities and there can be no guarantee that we ever will.
Expansion of our sources of supply, production capacity and sales network - To meet the increasing demand for our products, we need to expand our sources of supply and production capacity. We plan to make capital improvements in our existing production facilities, which would improve both their efficiency and capacity. In the short-run, we intend to increase our investment in our reliable supply network, personnel training, information technology applications and logistic system upgrades. We also participate in two non-equity investment opportunities through a VIE, both of which we expect to provide us with new networks and platforms.
Maintaining effective control of our costs and expenses - Successful cost control depends upon our ability to obtain and maintain adequate material supplies as required by our operations at competitive prices. We will focus on improving our long-term cost control strategies including establishing long-term alliances with certain suppliers to ensure adequate supply is maintained. We will carry forward the economies of scale and advantages from our nationwide distribution network and diversified offerings. Moreover, we will step up our efforts in higher value added products of Luobuma by using an exclusive and patented technology, to optimize quality management, procurement processes and cost control, and give full play to the strong production capacity and trustworthy sales teams to maximize our profit and bring better long-term return for our shareholders.
Economic and Political Risks
Our operations are conducted primarily in the PRC. Accordingly, our business, financial conditions and results may be influenced by the political, economic and legal environment in the PRC, and by the general state of the PRC economy.
Our operations in the PRC are subject to special considerations and significant risks not typically associated with companies in North America and Western Europe. These include risks with, among others, the political, economic and legal environment and foreign currency exchange. Our Company’s results may be adversely affected by changes in the political and social conditions in the PRC, and by changes in governmental policies with respect to laws and regulations, anti-inflationary measures, currency conversions, remittances abroad, and rates and methods of taxation, among other things.
Critical Accounting Policies and Estimates
The preparation of financial statements in conformity with U.S. generally accepted accounting principles (“U.S. GAAP”) requires the use of estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the consolidated financial statements as well as the reported amounts of revenue and expenses during the reporting period. Critical accounting policies are those accounting policies that may be material due to the levels of subjectivity and judgment necessary to account for highly uncertain matters or the susceptibility of such matters to change, and that have a material impact on financial condition or operating performance. While we base our estimates and judgments on our experience and on various other factors that we believe to be reasonable under the circumstances, actual results may differ from these estimates under different assumptions or conditions. We believe the following critical accounting policies used in the preparation of our consolidated financial statements require significant judgments and estimates. For additional information relating to these and other accounting policies, see Note 2 to our consolidated financial statements included elsewhere in this Report.
| 34 |
Consolidation of Variable Interest Entities
In accordance with accounting standards regarding consolidation of VIEs, VIEs are generally entities that lack sufficient equity to finance their activities without additional financial support from other parties or whose equity holders lack adequate decision-making ability. All VIEs and their subsidiaries with which the Company is involved must be evaluated to determine the primary beneficiary of the risks and rewards of the VIE. The primary beneficiary is required to consolidate the VIE for financial reporting purposes.
Use of Estimates
The preparation of financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the consolidated financial statements as well as the reported amounts of revenue and expenses during the reporting period. Significant estimates required to be made by management include, but are not limited to, useful lives of property, plant, and equipment, and intangible assets, the recoverability of long-lived assets and the valuation of accounts receivable, deferred taxes and inventory reserves. Actual results could differ from those estimates.
Accounts Receivable
Accounts receivable are recorded at net realizable value consisting of the carrying amount less an allowance for uncollectible accounts, as necessary. The Company reviews the accounts receivable on a periodic basis and makes general and specific allowances when there is doubt as to the collectability of individual balances. In evaluating the collectability of individual receivable balances, the Company considers many factors, including the age of the balance, the customers’ historical payment history, their current credit-worthiness and current economic trends. Accounts are written off against the allowance after efforts at collection prove unsuccessful.
Inventories
Inventories, which are stated at the lower of cost or current market value, consist of raw materials, work-in-progress, and finished goods related to the Company’s products. Cost is determined using the first in first out (“FIFO”) method. Market value is the lower of replacement cost or net realizable value. Agricultural products that the Company farms are recorded at cost, which includes direct costs such as seed selection, fertilizer, labor cost and contract fees that are spent in growing agricultural products on the leased farmland, and indirect costs which include amortization of prepayments of farmland leases and farmland development cost. All the costs are accumulated until the time of harvest and then allocated to the harvested crops costs when they are sold. The Company periodically evaluates its inventory and records an inventory reserve for certain inventories that may not be saleable or whose cost exceeds market prices.
Revenue Recognition
The Company recognizes revenue from sales of Luobuma products, Chinese medicinal herbal products and agricultural products, as well as providing logistic service and other processing services to external customers. The Company recognizes revenue when all of the following have occurred: (i) there is persuasive evidence of an arrangement with a customer, (ii) delivery has occurred or services have been rendered, (iii) the sales price is fixed or determinable, and (iv) the Company’s collection of such fees is reasonably assured. These criteria, as related to the Company’s revenue, are considered to have been met as follows:
Sales of products: the Company recognizes revenue from the sale of products when the goods are delivered and title to the goods passes to the customer provided that there are no uncertainties regarding customer acceptance; persuasive evidence of an arrangement exists; the sales price is fixed or determinable; and collectability is deemed probable.
| 35 |
Revenue from the rendering of services: Revenue from international freight forwarding, domestic air and overland freight forwarding services is recognized upon the performance of services as stipulated in the underlying contract or when commodities are being released from the customer’s warehouse; the service price is fixed or determinable; and collectability is deemed probable.
Fair Value of Financial Instruments
The Company follows the provisions of ASC 820, “Fair Value Measurements and Disclosures.” ASC 820 clarifies the definition of fair value, prescribes methods for measuring fair value, and establishes a fair value hierarchy to classify the inputs used in measuring fair value as follows:
Level 1 applies to assets or liabilities for which there are quoted prices in active markets for identical assets or liabilities.
Level 2 applies to assets or liabilities for which there are inputs other than those in Level 1 such as quoted prices for similar assets or liabilities in active markets; quoted prices for identical assets or liabilities in markets with insufficient volume or infrequent transactions (less active markets); or model-derived valuations in which significant inputs are observable or can be derived principally from, or corroborated by, observable market data.
Level 3 applies to assets or liabilities for which there are unobservable inputs to the valuation methodology that are significant to the measurement of the fair value of the assets or liabilities.
The carrying value of financial instruments included in current assets and liabilities approximate their fair values because of the short-term nature of these instruments.
Results of Operations for the Years Ended June 30, 2018 and 2017
Overview
The following table summarizes our results of operations for the years ended June 30, 2018 and 2017:
| Years Ended June 30, | Variance | |||||||||||||||
| 2018 | 2017 | Amount | % | |||||||||||||
| Revenue | $ | 43,897,618 | $ | 33,592,337 | $ | 10,305,281 | 30.68 | % | ||||||||
| Cost of revenue | 29,110,326 | 22,852,009 | 6,258,317 | 27.39 | % | |||||||||||
| Gross profit | 14,787,292 | 10,740,328 | 4,046,964 | 37.68 | % | |||||||||||
| General and administrative expenses | 3,985,604 | 1,813,402 | 2,172,202 | 119.79 | % | |||||||||||
| Selling expenses | 1,530,005 | 1,480,855 | 49,150 | 3.32 | % | |||||||||||
| Income from operations | 9,271,683 | 7,446,071 | 1,825,612 | 24.52 | % | |||||||||||
| Impairment loss on goodwill | (2,153,298 | ) | - | (2,153,298 | ) | 100.00 | % | |||||||||
| Income from equity method investments | 907,794 | 927,697 | (19,903 | ) | (2.15 | )% | ||||||||||
| Purchase rebate income | 1,377,108 | 1,136,162 | 240,946 | 21.21 | % | |||||||||||
| Other income | 307,637 | 348,181 | (40,544 | ) | (11.64 | )% | ||||||||||
| Interest income (expense), net | (58,775 | ) | 14,171 | (72,946 | ) | (514.76 | )% | |||||||||
| Income before income tax provision | 9,652,149 | 9,872,282 | (220,133 | ) | (2.23 | )% | ||||||||||
| Provision for income taxes | 2,123,587 | 1,252,637 | 870,950 | 69.53 | % | |||||||||||
| Net income | $ | 7,528,562 | $ | 8,619,645 | $ | (1,091,083 | ) | (12.66 | )% | |||||||
| Comprehensive income attributable to Shineco Inc. | $ | 9,219,686 | $ | 7,216,601 | $ | 2,003,085 | 27.76 | % | ||||||||
| 36 |
Revenue
Currently, we have three revenue streams derived from our three major business segments. First, developing, manufacturing and distributing specialized fabrics, textiles and other by-products derived from an indigenous Chinese plant Apocynum Venetum, known in Chinese as “Luobuma” or “Bluish Dogbane”, as well as purchasing and Luoboma raw materials processing, this segment is channeled through our wholly owned subsidiary, Tenet-Jove. Second, processing and distributing traditional Chinese medicinal herbal products as well as other pharmaceutical products; this segment is conducted via our VIE, Ankang Longevity Group and its subsidiaries. Third, planting, processing and distributing green and organic agricultural produce as well as growing and cultivation of yew trees; this segment is conducted through our VIEs, the Zhisheng Group.
The following table sets forth the breakdown of our revenue for each of our three segments, for the years ended June 30, 2018 and 2017, respectively:
| Years ended June 30, | Variance | |||||||||||||||||||||||
| 2018 | % | 2017 | % | Amount | % | |||||||||||||||||||
| Sales of Luobuma products | $ | 10,884,901 | 24.80 | % | $ | 3,624,993 | 10.79 | % | $ | 7,259,908 | 200.27 | % | ||||||||||||
| Sales of Chinese medicinal herbal products | 14,179,603 | 32.30 | % | 13,245,236 | 39.43 | % | 934,367 | 7.05 | % | |||||||||||||||
| Sales of other agricultural products | 18,833,114 | 42.90 | % | 16,722,108 | 49.78 | % | 2,111,006 | 12.62 | % | |||||||||||||||
| Total Amount | $ | 43,897,618 | 100.00 | % | $ | 33,592,337 | 100.00 | % | $ | 10,305,281 | 30.68 | % | ||||||||||||
For the years ended June 30, 2018 and 2017, revenue from sales of Luobuma products was US$ 10,884,901 and US$ 3,624,993, respectively, which represented a significant increase of US$ 7,259,908 or 200.27%. The significant increase of revenue from this segment was mainly due to revenue generated by a new subsidiary, Xinjiang Taihe, of US$ 8,206,556 for the year ended June 30, 2018. Moreover, the increase of revenue from this segment was due to increased sales volume of our health awareness related products. The Company also enhanced online sales promotions during the year ended June 30, 2018, which contributed to more sales revenue overall. The increase was partially offset by the decrease in revenue of US$ 1,592,159 from Tenet-Jove Xuzhou branch, as the business operation of this branch ceased in November 2017.
For the years ended June 30, 2018 and 2017, revenue from sales of Chinese medicinal herbal products was US$14,179,603 and US$13,245,236, respectively, representing an increase of US$934,367 or 7.05%. The increase was mainly due to the increased unit sales price in the herbal market from October to December 2017, as well as an increase in fulfilled sales orders from customers for the year ended June 30, 2018 than the same period in 2017.
For the years ended June 30, 2018 and 2017, revenue from sales of other agricultural products was US$ 18,833,114 and US$ 16,722,108, respectively, representing an increase of US$ 2,111,006 or 12.62%. The increase was mainly due to the increase in sales volume of yew trees since the PRC general public started to acknowledge the air purification function associated with the yew trees for year ended June 30, 2018 as compared to the same period in 2017.
| 37 |
Cost of Revenue and related tax
The following table sets forth the breakdown of the Company’s cost of revenue for each of our three segments, for the years ended June 30, 2018 and 2017, respectively:
| Years ended June 30, | Variance | |||||||||||||||||||||||
| 2018 | % | 2017 | % | Amount | % | |||||||||||||||||||
| Luobuma products | $ | 4,871,723 | 16.73 | % | $ | 1,695,938 | 7.42 | % | $ | 3,175,785 | 187.26 | % | ||||||||||||
| Chinese medicinal herbal products | 10,703,386 | 36.77 | % | 10,124,063 | 44.30 | % | 579,323 | 5.72 | % | |||||||||||||||
| Other agricultural products | 13,430,550 | 46.14 | % | 10,956,034 | 47.95 | % | 2,474,516 | 22.59 | % | |||||||||||||||
| Business and sales related tax | 104,667 | 0.36 | % | 75,974 | 0.33 | % | 28,693 | 37.77 | % | |||||||||||||||
| Total Amount | $ | 29,110,326 | 100.00 | % | $ | 22,852,009 | 100.00 | % | $ | 6,258,317 | 27.39 | % | ||||||||||||
For the years ended June 30, 2018 and 2017, cost of revenue from sales of our Luobuma products was US$ 4,871,723 and US$ 1,695,938, respectively, representing a significant increase of US$ 3,175,785 or 187.26%. The percentage of the increase in cost of revenue was lower than the increase in sales during this period, primarily due to the lower raw material costs ratio from our new subsidiary, Xinjiang Taihe for the year ended June 30, 2018 as compared to the corresponding period in 2017.
For the years ended June 30, 2018 and 2017, cost of revenue from sales of Chinese medicinal herbal products was US$ 10,703,386 and US$ 10,124,063, respectively, representing an increase of US$ 579,323 or 5.72%. The percentage of the variance in cost of revenue was proportional to the percentage of the variance in revenue due to the stable gross margin of our products.
For the years ended June 30, 2018 and 2017, cost of revenue from sales of other agricultural products was US$ 13,430,550 and US$10,956,034, respectively, representing an increase of US$ 2,474,516 or 22.59%. The increase was mainly due to the increase in freight cost of yew trees since we sold more yew trees in the year ended June 30, 2018, as compared to the same period in 2017. Additionally, beginning in the second quarter of fiscal year 2017, we started offering storage services, which has higher cost of revenue.
Gross Profit
The following table sets forth the breakdown of the Company’s gross profit for each of our three segments, for the years ended June 30, 2018 and 2017, respectively:
| Years ended June 30, | Variance | |||||||||||||||||||||||
| 2018 | % | 2017 | % | Amount | % | |||||||||||||||||||
| Luobuma products | $ | 5,995,619 | 40.55 | % | $ | 1,912,979 | 17.81 | % | $ | 4,082,640 | 213.42 | % | ||||||||||||
| Chinese medicinal herbal products | 3,417,825 | 23.11 | % | 3,061,275 | 28.50 | % | 356,550 | 11.65 | % | |||||||||||||||
| Other agricultural products | 5,373,848 | 36.34 | % | 5,766,074 | 53.69 | % | (392,226 | ) | (6.80 | )% | ||||||||||||||
| Total Amount | $ | 14,787,292 | 100.00 | % | $ | 10,740,328 | 100.00 | % | $ | 4,046,964 | 37.68 | % | ||||||||||||
Gross profit from Luobuma product sales increased by US$ 5,995,619 and gross profit contribution percentage increased by 213.42% for the year ended June 30, 2018 as compared to the same period of 2017. The increase in gross profit was primarily due to an increase in sales resulting from revenues generated by the new subsidiary Xinjiang Taihe, as mentioned above. The percentage of increase in gross profit was higher than the percentage of increase in sales mainly due to the higher gross margin from Xinjiang Taihe for the year ended June 30, 2018 as compared to the corresponding period in 2017.
| 38 |
Gross profit from sales of Chinese medicinal herbal products increased by US$ 356,550 or 11.65% for the year ended June 30, 2018 as compared to the same period of 2017. The percentage of the variance in gross profit was proportional to the percentage of the variance in revenue due to the stable gross margin of our products.
Gross profit from sales of other agricultural products decreased by US$ 392,226 or 6.80% for the year ended June 30, 2018 as compared to the same period of 2017. The decrease was mainly due to that we started new storage services with lower gross margin in the second quarter of fiscal year 2017, as compared to our traditional freight services provided in 2017.
Expenses
The following table sets forth the breakdown of our operating expenses for the years ended June 30, 2018 and 2017, respectively:
| Years ended June 30, | Variance | |||||||||||||||||||||||
| 2018 | % | 2017 | % | Amount | % | |||||||||||||||||||
| General and administrative expenses | $ | 3,985,604 | 72.26 | % | $ | 1,813,402 | 55.05 | % | $ | 2,172,202 | 119.79 | % | ||||||||||||
| Selling expenses | 1,530,005 | 27.74 | % | 1,480,855 | 44.95 | % | 49,150 | 3.32 | % | |||||||||||||||
| Total Amount | $ | 5,515,609 | 100.00 | % | $ | 3,294,257 | 100.00 | % | $ | 2,221,352 | 67.43 | % | ||||||||||||
General and Administrative Expenses
For the year ended June 30, 2018, our general and administrative expenses were US$ 3,985,604, representing an increase of US$ 2,172,202 or 119.79%, as compared to the same period of 2017. The increase was primarily due to the incorporation and acquisition of new subsidiaries, Tiankunrunze in second quarter of fiscal year 2017, and Xinjiang Taihe, Runze and Tianjin Tajite in fiscal year 2018. The increase in general and administrative expenses was also a result of increased bad debt expense, salary expenses as well as entertainment expense.
Selling Expenses
For the year ended June 30, 2018, our selling and distribution expenses were US$ 1,530,005, representing a slight increase of US$ 49,150, or 3.32%, as compared to the same period of 2017. The increase was primarily due to the acquisition of a new subsidiary, Tianjin Tajite, in October 2017. The increase in selling and distribution expenses was also a result of increased promotion expenses as the Company enhanced its online sales promotions, partially offset by decreased rent expense of warehouse and salary expenses due to more effective cost control during the years ended June 30, 2018 compared to the same period of 2017.
Impairment loss on goodwill
For the year ended June 30, 2018, our impairment loss on goodwill were US$ 2,153,298, representing an increase of US$ 2,153,298 or 100.00%, as compared to the same period of 2017. In conjunction with the preparation of our consolidated financial statement for year ended June 30, 2018, the management performed evaluation on the impairment of goodwill. Due to the lower than expected revenue and profit, and unfavorable business environment, the management fully recorded impairment loss on goodwill of Tianjin Tajite.
| 39 |
Income from Equity Method Investments
We are 49% participants in two equity investment companies with Shaanxi Pharmaceutical Group Pai’ang Medicine Co. Ltd. (“Shaanxi Pharmaceutical Group”): Shaanxi Pharmaceutical Sunsimiao Drugstores Ankang Retail Chain Co., Ltd. (“Sunsimiao Drugstores”), and Shaanxi Pharmaceutical Holding Group Longevity Pharmacy Co., Ltd. (“Shaanxi Longevity Pharmacy”). We recorded net income of US$ 754,099 and US$ 817,603 from these equity method investments for the years ended June 30, 2018 and 2017, respectively. The decrease in net income was primarily due to lower net profit in the two 49% equity investment companies in the current period.
We invested RMB 14.5 million (approximately US$ 2.2 million) into Tiancang Systematic Warehousing project (“Tiancang Project”) operated by Zhen’Ai Network. The Tiancang Project is currently operational. For the years ended June 30, 2018 and 2017, we recorded investment income distributions of US$ 153,695 and US$ 110,094 from Zhen’Ai Network, respectively.
On November 21, 2016, the Company entered into an agreement with Original Lab Inc., a California corporation (the “Investee”), pursuant to which the Company made a payment of US$ 200,000 to Original Lab in exchange for the right to acquire certain shares of its common stock and preferred stock. For the years ended June 30, 2018 and 2017, the Company did not record investment income from this investment.
Purchase Rebate Income
We are party to a supplemental agreement with Shaanxi Pharmaceutical Group. According to the supplemental agreement, the participants in the 49% equity investment companies established by Shaanxi Pharmaceutical Group and Ankang Longevity Group are required to purchase certain raw materials and drug products exclusively from Shaanxi Pharmaceutical Group. In return, Shaanxi Pharmaceutical Group has agreed to compensate Ankang Longevity Group with a purchase rebate of 7% of the total purchases made from Shaanxi Pharmaceutical Group. For the year ended June 30, 2018, total income of US$ 1,377,108 was recognized by Ankang Longevity Group from this supplemental agreement, compared to US$ 1,136,162 in the same period in 2017 due to the increase in purchases in the current period.
Interest Income (Expense), net
For the year ended June 30, 2018, our net interest expense was US$ 58,775 as compared to interest income of US$ 14,171 in the same period of 2017. The decrease in net interest income was primarily due to decreased interest income on the loan to Xinyang Yifangyuan Garden Technology Co., Ltd. (“Xinyang Yifangyuan”). Xinyang Yifangyuan paid off the loan of RMB 3,000,000 in December, 2016. Interest income of nil and US$ 86,355 was recognized on the loans for the years ended June 30, 2018 and 2017, respectively.
Provision for Income Taxes
For the years ended June 30, 2018 and 2017, the Company’s provision for income taxes increased by US$ 870,950 or 69.53% to US$ 2,123,587 for the year ended June 30, 2018 from US$ 1,252,637 for the year ended June 30, 2017. On December 22, 2017, the “Tax Cuts and Jobs Act” (“The Act”) was enacted, this Tax Act imposes a one-time transition tax on deemed repatriation of historical earnings of foreign subsidiaries, and future foreign earnings are subject to U.S. taxation. The change in rate has caused us to re-measure the Company’s income tax liability and record an estimate income tax expense of US$ 744,766 for the year ended June 30, 2018. Meanwhile, the provision for income taxes of Zhisheng Group increased by US$ 250,754 for the year ended June 30, 2018 due to the less amount of income tax exemption the Company received from the local tax authority, which also resulted a higher income taxes for the year ended June 30, 2018.
Net Income
Our net income decreased by US$ 1,091,083 or 12.66% for the year ended June 30, 2018 as compared to the same period of 2017. The decrease in net income was primarily a result of the increase in general and administrative expenses and impairment loss on goodwill, offset by the increase in gross profit.
| 40 |
Comprehensive Income
The comprehensive income was US$ 9,187,220 for the year ended June 30, 2018, an increase of US$ 1,838,611 from comprehensive income of US$ 7,348,609 for the year ended June 30, 2017. After deduction of non-controlling interest, the comprehensive income attributable to the Company was US$ 9,219,686 for the year ended June 30, 2018, compared to comprehensive income attributable to the Company of US$ 7,216,601 for the year ended June 30, 2017. The reason of the significant increase of comprehensive income was due to the increase in other comprehensive income due to the increase in the recorded gains of foreign currency translation where the financial statements denominated in RMB were translated to the USD denomination, partially offset by the decrease in net income as mentioned above.
Treasury Policies
We have established treasury policies with the objectives of achieving effective control of treasury operations and of lowering cost of funds. Therefore, funding for all operations and foreign exchange exposure have been centrally reviewed and monitored from the top level. To manage our exposure to fluctuations in exchange rates and interest rates on specific transactions and foreign currency borrowings, currency structured instruments and other appropriate financial instruments will be used to hedge material exposure, if any.
Our policy precludes us from entering into any derivative contracts purely for speculative activities. Through our treasury policies, we aim to:
(a) Minimize interest risk
This is accomplished by loan re-financing and negotiation. We will continue to closely monitor the total loan portfolio and compare the loan margin spread under our existing agreements against the current borrowing interest rates under different currencies and new offers from banks.
(b) Minimize currency risk
In view of the current volatile currency market, we will closely monitor the foreign currency borrowings at the company level. As of June 30, 2018 and June 30, 2017, we do not engage in any foreign currency borrowings or loan contracts.
Liquidity and Capital Resources
We currently finance our business operations primarily through cash flows from operations and proceeds from our initial public offering, as well as from short-term loans. Our current cash primarily consists of cash on hand and cash in bank, which is unrestricted as to withdrawal and use and is deposited with banks in China.
On September 28, 2016, we completed the initial public offering of 1,713,190 shares of the Company’s common stock at a price of US$ 4.50 per share for gross proceeds of US$ 7.7 million and net proceeds of approximately US$ 5.4 million.
Management believes that our current cash, cash flows from current and future operations, and access to loans will be sufficient to meet our working capital needs for at least the next 12 months. We intend to continue to carefully execute our growth plans and manage market risk
| 41 |
Working Capital
The following table provides the information about our working capital at June 30, 2018 and June 30, 2017:
| June 30, 2018 | June 30, 2017 | |||||||
| Current Assets | $ | 56,164,395 | $ | 44,725,927 | ||||
| Current Liabilities | 9,529,899 | 5,031,048 | ||||||
| Working Capital | $ | 46,634,496 | $ | 39,694,879 | ||||
The working capital increased by US$ 6,939,617 or 17.5% at June 30, 2018 from June 30, 2017, primarily as a result of an increase in cash due to better operating results during the years ended June 30, 2018. We believe that we currently have sufficient working capital to operate our business.
As of June 30, 2018 and June 30, 2017, the other major component of our working capital is accounts receivable.
The accounts receivable as of June 30, 2018 were US$ 15,478,336 an increase of approximately 6.9% from US$ 14,480,004 as of June 30, 2017, mainly due to an increase of revenue receivable from Xinjiang Taihe of US$ 4,531,858, partially offset by a decrease of revenue receivable from Qingdao Zhihesheng of US$ 2,254,563 and Ankang Longevity Group of US$ 538,532. In August 2018, the Company entered into three accounts receivable repayment plans with three major customers, Horgos Huajing Tencel Technology Development Co., Ltd., Qingdao Shipping Service Association and Qingdao Ship Owners Association, who each agreed to pay RMB 3.0 million (US$ 453,186 as of June 30, 2018), RMB 1.2 million (US$ 181,274 as of June 30, 2018) and RMB 1.2 million (US$ 181,274 as of June 30, 2018) per month starting from September 2018, and the balance of account receivable from them as of June 30, 2018 will be paid off before January 2020, June 2019 and June 2019, respectively. In the meanwhile, the Company continues to make sales transactions with these three customers.
Capital Commitments and Contingencies
Capital commitments refer to the allocation of funds for the possible purchase in the near future for fixed assets or investment. Contingency refers to a condition that arises from past transactions or events, the outcome of which will be confirmed only by the occurrence or non-occurrence of uncertain futures events.
As of June 30, 2018 and June 30, 2017, we had no material capital commitments or contingent liabilities.
Cash Flows
The following table provides detailed information about our net cash flows for the years ended June 30, 2018 and 2017.
| For the years ended June 30, | ||||||||
| 2018 | 2017 | |||||||
| Net cash provided by (used in) operating activities | $ | 9,846,719 | $ | (2,744,136 | ) | |||
| Net cash used in investing activities | (748,960 | ) | (725,832 | ) | ||||
| Net cash (used in) provided by financing activities | (485,483 | ) | 5,383,975 | |||||
| Effect of exchange rate changes on cash | (279,774 | ) | (768,830 | ) | ||||
| Net increase in cash | 8,332,502 | 1,145,177 | ||||||
| Cash, beginning of period | 23,154,551 | 22,009,374 | ||||||
| Cash, end of period | $ | 31,487,053 | $ | 23,154,551 | ||||
| 42 |
Operating Activities
Net cash provided by operating activities during the year ended June 30, 2018 was approximately US$ 9.8 million, consisting of net income of US$ 7.5 million, impairment loss on goodwill of US$ 2.2 million and net changes in our operating assets and liabilities, which mainly included an increase in accounts payable of US$ 2.1 million, an increase in taxes payable of US$ 2.0 million and an increase in other payable of US$ 1.3 million, partially offset by an increase in account receivables of US$ 3.6 million, an increase in advances to suppliers of US$ 2.6 million. Net cash used in operating activities during the year ended June 30, 2017 was approximately US$ 2.7 million, consisting of net income of US$ 8.6 million, income from equity method investments of US$ 0.9 million and net changes in our operating assets and liabilities, which mainly included an increase in accounts receivable of US$ 8.1 million, an increase in advances to suppliers of US$ 2.3 million and repayment in other payables of US$ 1.6 million, partially offset by reduction in inventory of US$ 2.1 million.
Investing Activities
For the year ended June 30, 2018, net cash used in investing activities was approximately US$ 748,960 as compared to net cash used in investing activities of US$725,832 for the same period of 2017. The slight increase in net cash used in investing activities was primarily due to an increase in acquisitions of property and equipment of US$ 1.7 million, a decrease in income received from investments in unconsolidated entities of US$ 0.8 million and a decrease in repayments of loans from related parties of US$ 0.6 million, partially offset by a decrease in payments of deposit for business acquisition of US$ 2.1 million and increase in repayment of loans from third parties of US$ 0.8 million for the year ended June 30, 2018, as compared to the same period in 2017.
Financing Activities
For the year ended June 30, 2018, net cash used in financing activities amounted to US$ 0.5 million, as opposed to net cash provided by financing activities of US$ 5.4 million for the same period of 2017. The decrease of US$ 5.9 million in net cash provided by financing activities was primarily due to absence of proceeds from our initial public offering for the year ended June 30, 2018, since we completed our initial public offering on September 28, 2016, as compared to the same period in 2017.
| Item 7a. | Quantitative and Qualitative Disclosures About Market Risk |
The Company is not required to provide the information required by this Item because the Company is a smaller reporting company.
| Item 8. | Financial Statements and Supplementary Data |
The consolidated financial statements are included in Part III, Item 15 (a) (1) and (2) of this Report.
| Item 9. | Changes in and Disagreements With Accountants on Accounting and Financial Disclosure |
None.
| 43 |
| Item 9A. | Controls and ProcedureS |
| (a) | Evaluation of Controls and Procedures |
We maintain disclosure controls and procedures designed to provide reasonable assurance that material information required to be disclosed by us in the reports filed or submitted under the Securities Exchange Act of 1934, as amended, is recorded, processed, summarized and reported within the time periods specified in the Securities and Exchange Commission rules and forms, and that the information is accumulated and communicated to our management, including our Chief Executive Officer and Chief Financial Officer, as appropriate to allow timely decisions regarding required disclosure. Based on our review, our management, including our Chief Executive Officer and Chief Financial Officer, concluded that the Company’s disclosure controls and procedures were not effective at the reasonable assurance level as of the end of the period covered by this Report due to following material weaknesses:
| ● | The Company does not have U.S. GAAP full-time qualified personnel in the accounting department to monitor the recording of the daily transactions; |
| ● | Lack of segregation of duties for accounting personnel who prepared and reviewed the journal entries. |
In order to address the above material weaknesses, our management plans to take the following steps:
| ● | Recruiting sufficient qualified professionals with appropriate levels of knowledge of U.S. GAAP and experience to assist in reviewing and resolving accounting issues in routine or complex transactions. To mitigate the reporting risks, we engaged an outside professional consulting firm to supplement our efforts to improve our internal control over financial reporting; |
| ● | Improving the communication between management, board of directors and the Chief Financial Officer; and |
| ● | Obtaining proper approval for other significant and non-routine transactions from the Board of Directors. |
The Company believes the foregoing measures will remediate the identified material weaknesses in future periods. The Company is committed to monitoring the effectiveness of these measures and making any changes that are necessary and appropriate.
| (b) | Changes in Internal Control over Financial Reporting |
Other than described above, there have been no changes in our internal control over financial reporting that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting during our fourth fiscal quarter of 2018. Because of its inherent limitations, a system of internal control over financial reporting can provide only reasonable assurance and may not prevent or detect misstatements. Further, because of changes in conditions, effectiveness of internal controls over financial reporting may vary over time. Our system contains self-monitoring mechanisms, and actions are taken to correct deficiencies as they are identified.
| 44 |
| (c) | Management’s Annual Report on Internal Control over Financial Reporting |
Management is responsible for establishing and maintaining adequate internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act) for the Company. These controls are designed and implemented under the supervision of our Chief Executive Officer and Chief Financial Officer to provide reasonable assurance to the management and our Board of Directors regarding the reliability of financial reporting and the preparation of our financial statements for external reporting purposes in accordance with U.S. GAAP. Our internal control over financial reporting includes those policies and procedures that:
| 1. | Pertain to the maintenance of records that in reasonable detail, accurately and fairly reflect the transactions and dispositions of our assets; | |
| 2. | Provide reasonable assurance that transactions are recorded properly to permit preparation of financial statements in accordance with U.S. GAAP, and that our receipts and expenditures are being made only in accordance with authorization of our management and directors; and | |
| 3. | Provide reasonable assurance regarding prevention or timely detection of unauthorized acquisitions, use or disposition of our assets that could have a material effect on the financial statements. |
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate. All internal control systems, no matter how well designed, have inherent limitations. Therefore, even those systems determined to be effective can provide only reasonable assurance with respect to financial statement preparation and presentation. Because of the inherent limitations of internal control, there is a risk that material misstatements may not be prevented or detected on a timely basis by internal control over financial reporting. However, these inherent limitations are known features of the financial reporting process. Therefore, it is possible to design into the process safeguards to reduce, though not eliminate, this risk.
As of June 30, 2018, management assessed the effectiveness of its internal control over financial reporting based on the criteria for effective internal control over financial reporting established in Internal Control—2013 Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (“COSO”) and SEC guidance on conducting such assessments. Based on such evaluation, management identified deficiencies that were determined to be material weaknesses.
A material weakness is a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of the company’s annual or interim financial statements will not be prevented or detected on a timely basis. Because of the following material weaknesses, management concluded that our internal controls over financial reporting were ineffective as of June 30, 2018:
| 1. | We did not have sufficient skilled accounting personnel that are either qualified as Certified Public Accountants in the United States or that have received education from U.S. institutions or other educational programs that would provide adequate relevant education relating to U.S. GAAP. Our Chief Financial Officer has limited experience with U.S. GAAP and are not U.S. Certified Public Accountants. Furthermore, our operating subsidiaries are based in China and are therefore required to comply with PRC GAAP, rather than U.S. GAAP. Thus, the accounting skills and understanding necessary to fulfill the requirements of U.S. GAAP-based reporting, including the preparation of consolidated financial statements, remain inadequate and thus constitute a material weakness. |
| 45 |
| 2. | Lack of segregation of duties for accounting personnel who prepared and reviewed the journal entries. | |
| 3. | In addition, since we only completed the design of our internal controls and assessments for all of our financial reporting cycles in March 2012, we are not yet able to declare our controls as effective over a sufficient period of time in order to demonstrate the operating effectiveness of our controls as of June 30, 2018. Therefore, we have determined that such lack of time to evaluate the design and operating effectiveness of our controls is also a material weakness. |
In an effort to remedy the foregoing material weaknesses in the future, we have started the second and third approaches, and we intend to continue to do the following:
| · | Develop a comprehensive training and development plan for our finance, accounting and internal audit personnel, including our Chief Financial Officer and Controller, in the principles and rules of U.S. GAAP, SEC reporting requirements and the application thereof; | |
| · | Design and implement a program to provide ongoing company-wide training regarding our internal controls, with particular emphasis on our finance and accounting staff; | |
| · | Implement an internal review process over financial reporting to review all recent accounting pronouncements and to verify that any accounting treatment identified in such report has been fully implemented and confirmed by our third-party consultant, and to continue to improve our ongoing review and supervision of our internal control over financial reporting; and | |
| · | Hire a full-time employee who possesses the requisite U.S. GAAP experience and education. |
Despite the material weaknesses and deficiencies reported above, our management believes that our consolidated financial statements included in this Report fairly present in all material respects our financial condition, results of operations and cash flows for the periods presented and that this Report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this annual Report.
This annual report does not include an attestation report of the Company’s independent registered public accounting firm regarding internal control over financial reporting. Management’s report was not subject to attestation by the Company’s independent registered public accounting firm pursuant to rules of the SEC that permit the Company to provide only management’s report in this annual report.
| Item 9b. | Other Information |
None.
| 46 |
| Item 10. | Directors, Executive Officers and Corporate Governance |
Executive Officers and Directors
The following table and text set forth the names and ages of all directors and executive officers as of June 30, 2018. There are no family relationships among our directors and executive officers. Each director is elected at our annual meeting of shareholders and holds office until the next annual meeting of shareholders, or until his successor is elected and qualified. Also provided herein are brief descriptions of the business experience of each director, executive officer and advisor during the past five years and an indication of directorships held by each director in other companies subject to the reporting requirements under the Federal securities laws. None of our officers or directors is a party adverse to us or has a material interest adverse to us.
| Name | Age | Role | Since | |||
| Yuying Zhang | 65 | Chair of the Board, Chief Executive Officer and Director | 2011 | |||
| Sai (Sam) Wang | 32 | Chief Financial Officer and Director | 2015* | |||
| Jiping Chen | 68 | Director | 2011 | |||
| Ying (Teresa) Zhang | 39 | Director (Independent) | 2014 | |||
| Yajun Shi | 40 | Director (Independent) | 2014 | |||
| Leiger Yongmin Yang | 41 | Director (Independent) | 2015 | |||
| Hua Yang | 47 | Director (Independent) | 2016 |
| * | Mr. Sai (Sam) Wang has been our CFO since 2015 and director since 2016. |
YuYing Zhang, age 65, has been Chairman of Shineco since 2011 and is the Chairman and CEO of the Company. He was the principal founder of Tenet-Jove, which was established in 1995 with his research and development of Luobuma functional fiber healthcare products. He has been the Chairman and CEO of Tenet-Jove since December 2003; under his leadership, the company has worked with more than 20 research institutions and enterprises and has obtained numerous national invention and new product patent rights for Luobuma product development. He also serves as a director in Tianjin Tenet Huatai Technological Development Co., Ltd. since 2003. From April 2014 to December 2014, he was the Chairman of the Board of Beijing Huiyin Ansheng Asset Management Co., Ltd. From 1995 to December 2003, he served as general manager of Tianjin Balas Technological Development Co., Ltd. Prior to starting Tenet-Jove in 1995, he was the deputy director at the Army Institute of Integrative Medicine. From 1991 to 1994, he was the Executive Director and Deputy General Manager at Shan Haidan Pharmaceutical Group, where he was responsible for strategic development planning and marketing. Mr. Zhang is a senior economist with a bachelor degree from China Central Radio and Television University in China. Mr. Zhang was chosen as director because his knowledge and extensive experience in research and development and management.
| 47 |
Sai (Sam) Wang, age 32, became our Chief Financial Officer in February 2015 and Director since 2016. Mr. Wang has worked for Shineco, Inc. since 2011 where he served as Financial Controller until his appointment as our Chief Financial Officer. Mr. Wang has been the supervisory director of Shineco Zhisheng (Beijing) Bio-Technology Co., Ltd. since 2014. He serves as the General Manager of Qingdao Yinghuanhai International Logistics Co., Ltd. since 2012. Prior to joining Shineco, he worked for Citibank in Shenzhen from 2008 until 2011, where he served as Manager of Corporate Finance. Mr. Wang obtained a Masters in Commerce with a concentration in applied finance from The University of Queensland in 2010. In 2008, he received a bachelor’s degree in Accounting from Griffith University in Australia. Mr. Wang was chosen as a director because he has profound knowledge of our industry and he is experienced with our financial matters.
Jiping Chen, age 68, has been a director for Shineco since 2011. Mr. Chen has served as Chairman, General Manager and Legal Representative of Ankang Longevity Group since 2006. From 1985 to 1992, Mr. Chen worked at Ankang Longevity Group where he a marketing manager. He has also served as a supervisor of Ankang Longevity Pharmaceutical (Group) Traditional Chinese Medicine Management Co., Ltd. since 2015. Since 2014, he has been Executive Director of Ankang Longevity Pharmaceutical (Group) Ziyang Longevity Traditional Chinese Medicine Materials Planting Co., Ltd. Since 2011 he has served as a supervisor of Ankang Longevity Real Estate Development Co., Ltd. He has also served as the Chairman of Board and General Manager of Ankang Longevity Pharmaceutical Group Pharmaceutical Industry Co., Ltd. since 2008. During 2008 to 2014, he served as a director of Ankang Longevity Pharmaceutical Group Breeding and Planting Co., Ltd. Since 2004, he has been the legal representative and Chairman of Board of Ankang Longevity Pharmaceutical (Group) Traditional Chinese Medicine Decoction Pieces Co., Ltd. He serves as legal representative and General Manager of Ankang Longevity Pharmaceutical Group Chain Co., Ltd. since 2003. Since 2002, Mr. Chen has been the Chairman of Board and General Manager of Ankang Longevity Pharmaceutical Group Purchasing Station Co., Ltd. From February 1975 to September 1985, Mr. Chen worked at the Ankang Area Public Bus Company where he served as company staff. From February 1973 to January 1975, he worked for Ankang County. Mr. Chen completed his three-year college education in Chinese Traditional Medicine major in Shannxi College of Chinese Traditional Medicine in 1997. Mr. Chen was chosen as director because of his experience in the traditional Chinese medicine business and his familiarity with Ankang Longevity Group’s operations.
Ying (Teresa) Zhang, age 39, has been a director for Shineco since October 2014. From 2014 to July 2016, she served as the director in Beijing Huiyin Ansheng Assets Management Co., Ltd., which is a related party to Shineco. Since October 2010, Ms. Zhang has served as a director for Mixbox Co. Ltd., an international chain store management company. From January 2010 through December 2010 she served as the chief financial officer and a director of Cleantech Solutions International, Inc., a U.S. public company (NASDAQ: CLNT) that manufactures wind power equipment in China. Ms. Zhang has served as a director in Shiqiao (Tianjin) Investment Consulting Co., Ltd. since 2009. Ms. Zhang was chosen to serve as a member of the board of directors because of her experience with U.S. GAAP, as well as her extensive prior work experience and educational background in the accounting field. Ms. Zhang was previously an auditing manager at GC Alliance HK CPA in Beijing from July 2005 until January 2010, where she provided auditing services to China-based companies. From January 2003 through June 2005, Ms. Zhang served as a liaison officer for the Australian-Chinese Friendship Business Association, a trade organization, and from July 2000 to September 2002 she was an auditor at Ernst & Young in Beijing. None of these companies that Ms. Zhang worked with is related to or affiliated with us. Ms. Zhang is a certified practicing accountant in Australia. She received a bachelor degree in international accounting from Renmin University in China in 1996 and a master’s degree in accounting from Macquarie University in Australia in 2005.
| 48 |
Yajun Shi, age 40, has been a director for Shineco since October 2014. He is an associate professor and supervisor of postgraduate education at Shaanxi University of Chinese Medicine, is currently the vice president of the College of Pharmacy of Shaanxi University of Chinese Medicine, and is the director of instrument center and committee member of the Traditional Chinese Medicine Chinese Drugs Pharmaceutics Association. Mr. Shi graduated from Shaanxi University of Chinese Medicine in June 1999 with a doctor degree, and has been engaging in new medicine development successively in The Fourth Military Medical University, the Pharmaceutical Research Institute and Shaanxi Tiansen Medicine Development Company. In July 2003, Mr. Shi received Master’s Degree from the College of Pharmacy of Shaanxi University of Chinese Medicine. In June 2012, Mr. Shi received Doctor of Medicine degree (M.D.) from Chengdu University of Traditional Chinese Medicine where he specialized in traditional Chinese medicine preparation. Mr. Shi’s major research efforts are focused on traditional Chinese medicine and health products, focusing on the basic study and application of a Chinese medicine nasal drug application system, the study of traditional Chinese medicine powder characterization, and the modification and the adaptability, as well as the research and development of, healthy foods. Mr. Shi has published more than 40 academic papers, and compiled and published six professional books. Mr. Shi was chosen as a director because of his extensive knowledge and research of traditional Chinese medicines.
Leiger Yongmin Yang, age 41, has been a director for Shineco since January 2015. He has served as a director in Beijing Lanzhong Time Internet Science and Technology Co., Ltd. since 2014. Mr. Yang founded in 2012 China Offshore Financial Group (COFG), a professional offshore financial services provider, focused on financial services for private equity and venture capital firms, outbound and inbound investments, international trading, family trusts, and tax planning. Prior to founding COFG, Yang served as the General Manager of Offshore Incorporations Limited Group & Vistra Group (OV Group) beginning in 2010. The OV Group focuses on the formation and maintenance of offshore companies, fund formation and fund administration, immigration and trust services. Mr. Yang has also worked as Chief Editor of the Chinese Asian Venture Capital Journal since 2006, where he is responsible for content of the weekly Chinese Asian Venture Capital Journal. From 2003 to 2011, he was the director of Zhiyuan Rongtong Investment Management Consulting (Beijing) Co., Ltd. Mr. Yang has spent the major part of his career in the private equity and venture capital sectors. Yang holds a Bachelor of Journalism degree from Jilin University in China. He was chosen as a director because of his hands-on experience in the private equity and venture capital sectors and his strong network with leading venture capital and private equity firms, law firms, accounting firms and banks in China.
Hua Yang, age 47, has been a director for Shineco since 2016. Ms. Yang has been a Partner and attorney in Grandall Law Firm in Beijing since March 2007. From 2006 to 2011, she served as a director of Beijing Shichen International Consulting Co., Ltd. Her practice area covers Corporation Law, foreign investments, international litigations and arbitrations. She provides services in the industries including civil aviation, agriculture biotechnology and nuclear power. From 2001 to 2007, Ms. Yang was a lawyer in Beijing Weiheng Law Firm. She was also a visiting attorney for Canada Fraser Milner Casgrain LLP and Pothier Delisle Law Office during 2002 to 2003. Before that, she was the Legal Manager of Weihai International Trust and Investment Corporation during 1995 to 1999. Ms. Yang received her degree of Master of Laws in Indiana University in 2006. She also graduated from Renmin University of China with a degree of Master of Laws in 2001. She obtained a Bachelor’s degree of Laws from Northwest University of Political Science and Law in 1995 in China. We have chosen Ms. Yang to serve as a director because of the perspective she brings to legal matters in China and her rich experience in Corporate Law and foreign investments.
| 49 |
Identification of Significant Employees
We do not have employees who are not executive officers, but who are expected to make significant contributions to our business.
Involvement in Certain Legal Proceedings
To the best of the Company’s knowledge, none of the following events occurred during the past ten years that are material to an evaluation of the ability or integrity of any of our executive officers, directors or promoters:
(1) A petition under the Federal bankruptcy laws or any state insolvency law was filed by or against, or a receiver, fiscal agent or similar officer was appointed by a court for the business or property of such person, or any partnership in which he was a general partner at or within two years before the time of such filing, or any corporation or business association of which he was an executive officer at or within two years before the time of such filing;
(2) Convicted in a criminal proceeding or is a named subject of a pending criminal proceeding (excluding traffic violations and other minor offenses);
(3) Subject of any order, judgment, or decree, not subsequently reversed, suspended or vacated, of any court of competent jurisdiction, permanently or temporarily enjoining him from, or otherwise limiting, the following activities:
(i) Acting as a futures commission merchant, introducing broker, commodity trading advisor, commodity pool operator, floor broker, leverage transaction merchant, any other person regulated by the Commodity Futures Trading Commission, or an associated person of any of the foregoing, or as an investment adviser, underwriter, broker or dealer in securities, or as an affiliated person, director or employee of any investment company, bank, savings and loan association or insurance company, or engaging in or continuing any conduct or practice in connection with such activity;
(ii) Engaging in any type of business practice; or
(iii) Engaging in any activity in connection with the purchase or sale of any security or commodity or in connection with any violation of Federal or State securities laws or Federal commodities laws;
(4) Subject of any order, judgment or decree, not subsequently reversed, suspended or vacated, of any Federal or State authority barring, suspending or otherwise limiting for more than 60 days the right of such person to engage in any activity described in paragraph (3)(i) above, or to be associated with persons engaged in any such activity;
(5) Found by a court of competent jurisdiction in a civil action or by the Commission to have violated any Federal or State securities law, and the judgment in such civil action or finding by the Commission has not been subsequently reversed, suspended, or vacated;
| 50 |
(6) Found by a court of competent jurisdiction in a civil action or by the Commodity Futures Trading Commission to have violated any Federal commodities law, and the judgment in such civil action or finding by the Commodity Futures Trading Commission has not been subsequently reversed, suspended or vacated;
(7) Subject of, or a party to, any Federal or State judicial or administrative order, judgment, decree, or finding, not subsequently reversed, suspended or vacated, relating to an alleged violation of:
(i) Any Federal or State securities or commodities law or regulation; or
(ii) Any law or regulation respecting financial institutions or insurance companies including, but not limited to, a temporary or permanent injunction, order of disgorgement or restitution, civil money penalty or temporary or permanent cease-and-desist order, or removal or prohibition order; or
(iii) Any law or regulation prohibiting mail or wire fraud or fraud in connection with any business entity; or
(8) Subject of, or a party to, any sanction or order, not subsequently reversed, suspended or vacated, of any self-regulatory organization, any registered entity (as defined in Section 1(a)(29) of the Commodity Exchange Act (7 U.S.C. 1(a)(29))), or any equivalent exchange, association, entity or organization that has disciplinary authority over its members or persons associated with a member.
Promoters and Certain Control Persons
None of our management or other control persons were “promoters” (within the meaning of Rule 405 under the Securities Act), and none of such persons took the initiative in the formation of our business or received any of our debt or equity securities or any of the proceeds from the sale of such securities in exchange for the contribution of property or services, during the last five years.
Board of Directors and Board Committees
Our board of directors currently consists of seven directors, four of whom — Ying (Teresa) Zhang, Yajun Shi, Leiger Yongmin Yang and Hua Yang — are independent, as such term is defined by The NASDAQ Capital Market.
Mr. Yuying Zhang currently holds both the positions of Chief Executive Officer and Chairman of the Board. These two positions have not been consolidated into one position; Mr. Zhang simply holds both positions at this time.
The Board believes that the most effective leadership structure at this time is not to separate the roles of Chairman and Chief Executive Officer. A combined structure provides us with a single leader who represents us to our stockholders, regulators, business partners and other stakeholders, among other reasons set forth below. Should the Board conclude otherwise, the Board will separate the roles and appoint an independent director. This structure creates efficacy in the preparation of the meeting agendas and related Board materials as our Chief Executive Officer works directly with those individuals preparing the necessary Board materials and is more connected to our overall daily operations. Agendas are also prepared with the permitted input of the full Board of Directors allowing for any concerns or risks of any individual director to be discussed as deemed appropriate. The Board believes that we have benefited from this structure, and Ms. Zhang’s continuation in the combined role of the Chairman and Chief Executive Officer is in the best interest of the stockholders.
As a smaller reporting company with a small board of directors, we believe it is appropriate to have the involvement and input of all of our directors in risk oversight matters.
| 51 |
Board Committees
We have established three standing committees under the board: the audit committee, the compensation committee and the nominating committee. Each committee has three members, and each member is independent, as such term is defined by The Nasdaq Capital Market. The audit committee is responsible for overseeing the accounting and financial reporting processes of our company and audits of the financial statements of our company, including the appointment, compensation and oversight of the work of our independent auditors. The compensation committee of the board of directors reviews and makes recommendations to the board regarding our compensation policies for our officers and all forms of compensation, and also administers our incentive compensation plans and equity-based plans (but our board retains the authority to interpret those plans). The nominating committee of the board of directors is responsible for the assessment of the performance of the board, considering and making recommendations to the board with respect to the nominations or elections of directors and other governance issues. The nominating committee considers diversity of opinion and experience when nominating directors.
The members of the audit committee, the compensation committee and the nominating committee are set forth below. All such members qualify as independent under the rules of The Nasdaq Capital Market.
| Director | Audit Committee |
Compensation Committee |
Nominating Committee |
|||||||||
| Ying (Teresa) Zhang | (1)(2)(3) | (1) | (1) | |||||||||
| Hua Yang | (1) | (1)(2) | ||||||||||
| Leiger Yongmin Yang | (1) | (1) | ||||||||||
| Yajun Shi | (1)(2) | (1) | ||||||||||
| (1) | Committee member |
| (2) | Committee chair |
| (3) | Our board has determined that we have at least one “audit committee financial expert,” as defined by the rules and regulations of the SEC and that is Ying (Teresa) Zhang. |
Section 16(a) of the Exchange Act
Section 16(a) of the Exchange Act, as amended, requires our directors and certain of our officers, as well as persons who own more than 10% of a registered class of our equity securities (“Reporting Persons”), to file reports with the SEC. To our knowledge, based solely on review of the copies of such reports furnished to us and written representations that no other reports were required, all Section 16(a) filing requirements applicable to officers, directors and greater than ten percent shareholders were complied with during the fiscal year ended June 30, 2018.
| 52 |
Code of Ethics
We have adopted a code of ethics that applies to our principal executive officer, principal financial officer, principal accounting officer or controller, or persons performing similar functions. We will provide a copy of our code of ethics to any person who requests a copy in writing to the Secretary of the Company, including the e-mail address or facsimile number of the requesting party. Any written requests should be mailed to us at Shineco, Inc., Room 1001, Building T5, DaZu Square, Daxing District, Beijing, People’s Republic of China.
| Item 11. | Executive Compensation |
The following table shows the annual compensation paid by us for the years ended June 30, 2018 and 2017 to YuYing Zhang, our principal executive officer. We are required to include the compensation of our CEO, regardless of his compensation.
Summary Compensation Table
| Name and Principal Position | Fiscal Year | Salary (1) ($) | Bonus ($) | Stock Awards ($) | Option Awards ($) | Other Compensation ($) | Total ($) | ||||||||||||||||||||
| YuYing Zhang (CEO) (1) | 2018 | 120,000 | - | - | - | 18,180 | (2) | 138,180 | |||||||||||||||||||
| 2017 | 120,000 | - | - | - | 18,180 | (2) | 138,180 | ||||||||||||||||||||
| (1) | Salaries were paid in RMB. |
| (2) | Mr. Zhang receives monthly payments for rent for his personal home and parking. |
Employment Agreements
Generally
Under Chinese law, we may only terminate employment agreements without cause and without penalty by providing notice of non-renewal one month prior to the date on which the employment agreement is scheduled to expire. If we fail to provide this notice or if we wish to terminate an employment agreement in the absence of cause, then we are obligated to pay the employee one month’s salary for each year we have employed the employee. We are, however, permitted to terminate an employee for cause without penalty to our company, where the employee has committed a crime or the employee’s actions or inactions have resulted in a material adverse effect to us. At this time, we have no employment agreements with any of our executive officers.
| 53 |
Outstanding Equity Awards
There was no equity awards granted to our officers or directors in the year ended June 30, 2018.
Retirement Plans
We currently have no plans that provide for the payment of retirement benefits, or benefits that will be paid primarily following retirement, including but not limited to tax-qualified defined benefit plans, supplemental executive retirement plans, tax-qualified defined contribution plans and nonqualified defined contribution plans.
Potential Payments upon Termination or Change-in-Control
We currently have no contract, agreement, plan or arrangement, whether written or unwritten, that provides for payments to a named executive officer at, following, or in connection with any termination, including without limitation resignation, severance, retirement or a constructive termination of a named executive officer, or a change in control of the Company or a change in the named executive officer’s responsibilities, with respect to each named executive officer.
Director Compensation
During the year ended June 30, 2018, we paid our independent directors an annual cash retainer of $10,000. In the future, we may also provide stock, option or other equity-based incentives to our directors for their service. We also reimbursed our directors for any out-of-pocket expenses incurred by them in connection with their services provided in such capacity.
The following table reflects all compensation awarded to, earned by or paid to our directors for the fiscal year ended June 30, 2018. Directors who are also officers do not receive any additional compensation for their services as directors.
| Name | Fees Earned or Paid in Cash ($) | Stock Awards ($) | Options Awards ($) | Non-Equity Incentive Plan Compensation ($) | Non-Qualified Deferred Compensation Earnings ($) | All Other Compensation ($) | Total ($) (1) | |||||||||||||||||||||
| YuYing Zhang | 10,000 | — | — | — | — | — | 10,000 | |||||||||||||||||||||
| Sai (Sam) Wang | 10,000 | — | — | — | — | — | 10,000 | |||||||||||||||||||||
| Jiping Chen | 10,000 | — | — | — | — | — | 10,000 | |||||||||||||||||||||
| Ying (Teresa) Zhang | 10,000 | — | — | — | — | — | 10,000 | |||||||||||||||||||||
| Yajun Shi | 10,000 | — | — | — | — | — | 10,000 | |||||||||||||||||||||
| Leiger Yongmin Yang | 10,000 | — | — | — | — | — | 10,000 | |||||||||||||||||||||
| Hua Yang | 10,000 | — | — | — | — | — | 10,000 | |||||||||||||||||||||
| (1) | All cash compensation was paid in RMB. The amounts in the foregoing table have been converted into U.S. Dollar at the conversion rate at 1 RMB to 0.1475 USD. |
| 54 |
| Item 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters |
The following table sets forth information, as of October 11, 2018, regarding the beneficial ownership of our common stock by any person known to us to be the beneficial owner of more than 5% of the outstanding common stock, by directors and certain executive officers, and by all of our directors and executive officers as a group. Unless otherwise noted, our officers and directors utilize the following address for correspondence purposes: Shineco, Inc., Room 1001, Building T5, DaZu Square, Daxing District, Beijing, People’s Republic of China.
| Name and Address | Title of Class | Amount and Nature of Beneficial Ownership | Percent (%) of Class | |||||||
| Yuying Zhang | common | 1,343,140 | (1) | 9.632 | % | |||||
| Sai (Sam) Wang | common | 749,645 | 3.564 | % | ||||||
| Jiping Chen | common | 2,194,115 | 10.431 | % | ||||||
| Ying (Teresa) Zhang | common | - | - | |||||||
| Leiger Leiger Yongmin Yang | common | - | - | |||||||
| Hua Yang | common | - | - | |||||||
| Yajun Shi | common | - | - | |||||||
| All Officers and Directors as a Group (7 total) | common | 4,286,900 | 23.63 | % | ||||||
| 5% Shareholders Not Mentioned Above: | ||||||||||
| Xiaoyan Chen | common | 1,088,067 | 5.173 | % | ||||||
| Qiwei Wang | common | 1,109,908 | 5.277 | % | ||||||
| (1) | Includes 200,000 shares owned by Min Zhao, the wife of Yuying Zhang. By virtue of this relationship, each of Ms. Zhao and Mr. Zhang may be deemed to share beneficial ownership of the shares of our company held by each of them. Mr. Zhang disclaims beneficial ownership of these shares. |
| 55 |
| Item 13. | Certain Relationships and Related Transactions |
REVIEW, APPROVAL OR RATIFICATION OF RELATED PARTY TRANSACTIONS
Our audit committee is responsible for reviewing and approving all related party transactions that are required to be disclosed under the applicable rules of the SEC and NASDAQ, when appropriate, and authorizing or ratifying all such transactions in accordance with written policies and procedures established by our board of directors from time to time. The audit committee may approve or ratify related party transaction only if it determines in good faith that under all the circumstances, the transaction is fair to us.
A director may vote in respect of any contract or transaction in which he is interested, provided, however that the nature of the interest of any director in any such contract or transaction shall be disclosed by him at or prior to its consideration and any vote on that matter. A general notice or disclosure to the directors or otherwise contained in the minutes of a meeting or a written resolution of the directors or any committee thereof of the nature of a director’s interest shall be sufficient disclosure and after such general notice it shall not be necessary to give special notice relating to any particular transaction. A director may be counted for a quorum upon a motion in respect of any contract or arrangement which he shall make with our company, or in which he is so interested and may vote on such motion.
We have a policy under which we are prohibited from making or renewing any personal loan to our executive officers or directors in accordance with Section 13(k) of the Exchange Act. The related party transactions with YuYing Zhang, our Chief Executive Officer and director, described in this section occurred prior to adoption of this policy, and as such, these transactions were not subject to such prohibition. As of date of this Report, all outstanding amounts due from any loans to executive officers or directors have been collected in full.
TRANSACTIONS
Members of the current management team are the owners of the VIEs in the PRC. (For additional details regarding the structure and ownership of our VIEs, please refer to Item 1 “Business”.)
DUE FROM RELATED PARTIES
The Company had previously made temporary advances to certain shareholders (listed below) of the Company and to other entities that are either owned by family members of those shareholders or to other entities that the Company has investments in. Those advances are due on demand, non-interest bearing
As of June 30, 2018, the outstanding amounts due from related parties consist of the following:
| June 30, 2018 | ||||
| Yang Bin | $ | 151,063 | ||
| Zhang Xin | 93,658 | |||
| Chang Song | 59,669 | |||
| Zhang Xinyu | - | |||
| Zhang Hua | - | |||
| Beijing Huiyinansheng Asset Management Co., Ltd | 22,690 | |||
| Zhang Yuying | - | |||
| Wang Qiwei | 61,181 | |||
| Tian Shuangpeng | - | |||
| $ | 388,261 | |||
Zhang Xinyu and Zhang Hua, paid off their outstanding amounts in the year ended June 30, 2018, $61,441 and $28,034, respectively.
| 56 |
DUE TO RELATED PARTIES
As of June 30, 2018, the Company had related party payables of US$ 197,617, mainly due to the principal shareholders or certain relatives of the shareholders of the Company who lend funds for the Company’s operations. The payables are unsecured, non-interest bearing and due on demand.
| June 30, 2018 | ||||
| Wu Yang (1) | $ | 96,755 | ||
| Wang Sai (2) | - | |||
| Zhao Min (3) | 100,862 | |||
| $ | 197,617 | |||
| (1) | Yang Wu is the wife of Yin Weixing, one of our Directors. |
| (2) | Sai (Sam) WangSai (Sam) Wang is our Chief Financial Officer and Director. The Company paid to Wang Sai $61,441 for the related party payables in the year ended June 30, 2018. |
| (3) | Min Zhao is the wife of Yuying Zhang, our Chief Executive Officer and Chair of the Board. |
SALES TO RELATED PARTIES
For the years ended June 30, 2018 and 2017, the Company recorded sales to Shaanxi Pharmaceutical Group, a related party of US$ 3,285,311 and US$ 3,038,195, respectively. As of June 30, 2018 and 2017, the balance of accounts receivable due from Shaanxi Pharmaceutical Group was US$ 1,526,351 and US$ 2,205,453, respectively. We are 49% participants in two equity investment companies with Shaanxi Pharmaceutical Group Pai’ang Medicine Co. Ltd. (“Shaanxi Pharmaceutical Group”): Shaanxi Pharmaceutical Sunsimiao Drugstores Ankang Retail Chain Co., Ltd. (“Sunsimiao Drugstores”), and Shaanxi Pharmaceutical Holding Group Longevity Pharmacy Co., Ltd. (“Shaanxi Longevity Pharmacy”).
| 57 |
| Item 14. | Principal Accounting Fees and Services |
The following table shows the fees that were billed for audit and other services provided by Wei Wei & Co., LLP and Friedman LLP, our independent accountants, respectively, for the fiscal year ended June 30, 2018 and 2017:
| Fiscal Year Ended June 30, | ||||||||
| 2018 | 2017 | |||||||
| Audit Fees(1) | $ | 210,000 | $ | 300,000 | ||||
| Audit-related Fees(2) | - | - | ||||||
| Tax Fees(3) | - | - | ||||||
| All Other Fees(4) | - | - | ||||||
| Total | $ | 210,000 | $ | 300,000 | ||||
| (1) | Audit Fees –This category includes the audit of our annual financial statements, review of financial statements included in our Quarterly Reports on Form 10-Q, and services that are normally provided by independent auditors in connection with statutory and regulatory filings or the engagement for fiscal years. This category also includes advice on audit and accounting matters that arose during, or as a result of, the audit or the review of interim financial statements. |
| (2) | Audit-Related Fees – This category consists of assurance and related services by our independent auditor that are reasonably related to the performance of the audit or review of our financial statements and are not reported above under “Audit Fees.” The services for the fees disclosed under this category include consultation regarding our correspondence with the SEC. |
| (3) | Tax Fees – This category consists of professional services rendered by our independent auditors for tax compliance and tax advice. The services for the fees disclosed under this category include tax return preparation and technical tax advice. |
| (4) | All Other Fees –This category consists of fees for other miscellaneous items such as travel and out-of-pocket expenses. |
Pre-Approval Policies and Procedures of the Audit Committee
Our Audit Committee approves the engagement of our independent auditors and is also required to pre-approve all audit and non-audit expenses. Prior to engaging its accountants to perform particular services, our Audit Committee obtains an estimate for the service to be performed. All of the services described above were approved by the Audit Committee in accordance with its procedure.
| 58 |
| Item 15. | Exhibits and Financial Statement Schedules |
(1) Financial Statements
The financial statements for the years ended June 30, 2018 and 2017 are included in Part II, Item 8 of this Report.
(2) Financial Statement Schedules
Schedules are omitted because the required information is not present or is not present in amounts sufficient to require submission of the schedule or because the information required is given in the consolidated financial statements or the notes thereto.
(3) Exhibits
EXHIBIT INDEX
The following documents are filed herewith:
| 59 |
| 60 |
| 61 |
| 62 |
| 63 |
| * | Filed herewith. |
| ** | Furnished but not filed. |
| † | Previously filed. |
| (1) | Incorporated by reference to the Company’s Registration Statement on Form S-1 filed with the SEC on July 1, 2015 (Registration No. 333-202803). |
| (2) | Incorporated by reference to the Company’s Annual Report on Form 10-K filed with the SEC September 28, 2016. |
| (3) | Incorporated by reference to the Company’s Registration Statement on Form S-1 filed with the SEC on January 27, 2016 (Registration No. 333-202803). |
| Item 16. | FORM 10-K SUMMARY |
None.
| 64 |
SIGNATURES
Pursuant to the requirements of section 13 or 15 (d) of the Securities Exchange Act of 1934, the Registrant has duly caused this Report to be signed on its behalf by the undersigned, thereunto duly authorized.
| SHINECO, INC. (Registrant) | ||
| Date: October 15, 2018 | By: | /s/ YuYing Zhang |
| YuYing Zhang | ||
| Chief Executive Officer | ||
| Date: October 15, 2018 | By: | /s/ Sai (Sam) Wang |
| Sai (Sam) Wang | ||
| Chief Financial Officer |
Pursuant to the requirements of the Securities Act of 1933, this Report has been signed below by the following persons on behalf of the Registrant and in the capacities and on the dates indicated:
| Signature | Title | Date | ||
| /s/ YuYing Zhang | Chief Executive Officer and Director | October 15, 2018 | ||
| YuYing Zhang | (Principal Executive Officer) | |||
| /s/ Sai (Sam) Wang | Chief Financial Officer and Director | October 15, 2018 | ||
| Sai (Sam) Wang | (Principal Accounting and Financial Officer) | |||
| /s/ Jiping Chen | Director | October 15, 2018 | ||
| Jiping Chen | ||||
| /s/ Ying (Teresa) Zhang | Director | October 15, 2018 | ||
| Ying (Teresa) Zhang | ||||
| /s/ Yajun Shi | Director | October 15, 2018 | ||
| Yajun Shi | ||||
| /s/ Leiger Yongmin Yang | Director | October 15, 2018 | ||
| Leiger Yongmin Yang | ||||
| /s/ Hua Yang | Director | October 15, 2018 | ||
| Hua Yang | ||||
| 65 |

 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders of Shineco, Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheet of Shineco, Inc. (the “Company”) as of June 30, 2018 and 2017, and the related consolidated statements of income and comprehensive income, changes in stockholders’ equity, and cash flows for each of the years in the two-year period ended June 30, 2018, and the related notes (collectively referred to as the financial statements). In our opinion, the financial statements present fairly, in all material respects, the consolidated financial position of the Company as of June 30, 2018 and 2017, and the consolidated results of its operations and its cash flows for each of the years in the two-year period ended June 30, 2018, in conformity with accounting principles generally accepted in the United States of America.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. |
F-1

Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ Wei, Wei & Co., LLP
We have served as the Company’s auditor since 2017. Flushing, New York
October 15, 2018 |
F-2
SHINECO, INC.
CONSOLIDATED BALANCE SHEETS
| June 30, | June 30, | |||||||
| 2018 | 2017 | |||||||
| ASSETS | ||||||||
| CURRENT ASSETS: | ||||||||
| Cash | $ | 31,487,053 | $ | 23,154,551 | ||||
| Accounts receivable, net | 15,478,336 | 14,480,004 | ||||||
| Due from related parties | 388,261 | 448,833 | ||||||
| Inventories | 2,364,558 | 2,346,273 | ||||||
| Advances to suppliers, net | 4,977,407 | 2,396,123 | ||||||
| Deferred issuance cost | 434,000 | - | ||||||
| Other current assets | 1,034,780 | 1,900,143 | ||||||
| TOTAL CURRENT ASSETS | 56,164,395 | 44,725,927 | ||||||
| Property and equipment, net | 11,697,304 | 10,320,396 | ||||||
| Land use right, net of accumulated amortization | 1,345,088 | 1,346,631 | ||||||
| Investments | 6,567,090 | 5,695,080 | ||||||
| Deposit for business acquisition | - | 2,065,686 | ||||||
| Distribution rights | 1,114,837 | - | ||||||
| Long-term deposit and other noncurrent assets | 113,764 | 112,883 | ||||||
| Long-term accounts receivable, net | 2,700,367 | - | ||||||
| Prepaid leases | 3,397,572 | 3,784,533 | ||||||
| Deferred tax assets | - | 233,834 | ||||||
| TOTAL ASSETS | $ | 83,100,417 | $ | 68,284,970 | ||||
| LIABILITIES AND EQUITY | ||||||||
| CURRENT LIABILITIES: | ||||||||
| Short-term loans | $ | 2,316,283 | $ | 2,663,628 | ||||
| Accounts payable | 2,270,140 | 158,068 | ||||||
| Advances from customers | 17,500 | 5,439 | ||||||
| Due to related parties | 197,617 | 257,880 | ||||||
| Other payables and accrued expenses | 1,736,735 | 337,107 | ||||||
| Taxes payable | 2,991,624 | 1,608,926 | ||||||
| TOTAL CURRENT LIABILITIES | 9,529,899 | 5,031,048 | ||||||
| Income tax payable - noncurrent portion | 685,185 | - | ||||||
| Deferred tax liability | 11,652 | - | ||||||
| TOTAL LIABILITIES | 10,226,736 | 5,031,048 | ||||||
| Commitments and contingencies | - | - | ||||||
| EQUITY: | ||||||||
| Common stock; par value $0.001, 100,000,000 shares authorized; 21,234,072 and 21,034,072 shares issued and outstanding at June 30, 2018 and June 30, 2017 | 21,234 | 21,034 | ||||||
| Additional paid-in capital | 23,171,102 | 22,737,302 | ||||||
| Statutory reserve | 4,085,819 | 3,484,449 | ||||||
| Retained earnings | 46,051,289 | 39,064,743 | ||||||
| Accumulated other comprehensive loss | (1,509,212 | ) | (3,140,982 | ) | ||||
| Total Stockholders' equity of Shineco, Inc. | 71,820,232 | 62,166,546 | ||||||
| Non-controlling interest | 1,053,449 | 1,087,376 | ||||||
| TOTAL EQUITY | 72,873,681 | 63,253,922 | ||||||
| TOTAL LIABILITIES AND EQUITY | $ | 83,100,417 | $ | 68,284,970 | ||||
The accompanying notes are an integral part of these consolidated financial statements
F-3
SHINECO, INC.
CONSOLIDATED STATEMENTS OF INCOME AND COMPREHENSIVE INCOME
| For the Years Ended June 30, | ||||||||
| 2018 | 2017 | |||||||
| REVENUE | $ | 43,897,618 | $ | 33,592,337 | ||||
| COST OF REVENUE | ||||||||
| Cost of product and services | 29,005,659 | 22,776,035 | ||||||
| Business and sales related tax | 104,667 | 75,974 | ||||||
| Total cost of revenue | 29,110,326 | 22,852,009 | ||||||
| GROSS PROFIT | 14,787,292 | 10,740,328 | ||||||
| OPERATING EXPENSES | ||||||||
| General and administrative expenses | 3,985,604 | 1,813,402 | ||||||
| Selling expenses | 1,530,005 | 1,480,855 | ||||||
| Total operating expenses | 5,515,609 | 3,294,257 | ||||||
| INCOME FROM OPERATIONS | 9,271,683 | 7,446,071 | ||||||
| OTHER INCOME | ||||||||
| Impairment loss on goodwill | (2,153,298 | ) | - | |||||
| Income from equity method investments | 907,794 | 927,697 | ||||||
| Purchase rebate income | 1,377,108 | 1,136,162 | ||||||
| Other income | 307,637 | 348,181 | ||||||
| Interest income (expense), net | (58,775 | ) | 14,171 | |||||
| Total other income | 380,466 | 2,426,211 | ||||||
| INCOME BEFORE PROVISION FOR INCOME TAXES | 9,652,149 | 9,872,282 | ||||||
| PROVISION FOR INCOME TAXES | 2,123,587 | 1,252,637 | ||||||
| NET INCOME | 7,528,562 | 8,619,645 | ||||||
| Net (loss) income attributable to non-controlling interest | (59,354 | ) | 149,991 | |||||
| NET INCOME ATTRIBUTABLE TO SHINECO, INC. | $ | 7,587,916 | $ | 8,469,654 | ||||
| COMPREHENSIVE INCOME | ||||||||
| Net income | $ | 7,528,562 | $ | 8,619,645 | ||||
| Other comprehensive income (loss): foreign currency translation gain (loss) | 1,658,658 | (1,271,036 | ) | |||||
| Total comprehensive income | 9,187,220 | 7,348,609 | ||||||
| Less: comprehensive (loss) income attributable to non-controlling interest | (32,466 | ) | 132,008 | |||||
| COMPREHENSIVE INCOME ATTRIBUTABLE TO SHINECO, INC. | $ | 9,219,686 | $ | 7,216,601 | ||||
| Weighted average number of shares basic and diluted | 21,119,004 | 20,616,335 | ||||||
| Earnings per common share | $ | 0.36 | $ | 0.41 | ||||
The accompanying notes are an integral part of these consolidated financial statements
F-4
SHINECO, INC.
CONSOLIDATED STATEMENTS OF CHANGES IN EQUITY
FOR THE YEARS ENDED JUNE 30, 2018 AND 2017
| ACCUMULATED | ||||||||||||||||||||||||||||||||
| ADDITIONAL | OTHER | NON- | ||||||||||||||||||||||||||||||
| COMMON STOCK | PAID-IN | STATUTORY | RETAINED | COMPREHENSIVE | CONTROLLING | TOTAL | ||||||||||||||||||||||||||
| SHARES | AMOUNT | CAPITAL | RESERVE | EARNINGS | INCOME (LOSS) | INTEREST | EQUITY | |||||||||||||||||||||||||
| Balance at June 30, 2016 | 19,320,882 | $ | 19,321 | $ | 17,344,466 | $ | 3,242,139 | $ | 30,837,399 | $ | (1,887,929 | ) | $ | 955,368 | $ | 50,510,764 | ||||||||||||||||
| Stock issuance | 1,713,190 | 1,713 | 5,392,836 | 5,394,549 | ||||||||||||||||||||||||||||
| Net income for the year | 8,469,654 | 149,991 | 8,619,645 | |||||||||||||||||||||||||||||
| Appropriation of statutory reserve | 242,310 | (242,310 | ) | - | ||||||||||||||||||||||||||||
| Foreign currency translation loss | (1,253,053 | ) | (17,983 | ) | (1,271,036 | ) | ||||||||||||||||||||||||||
| Balance at June 30, 2017 | 21,034,072 | 21,034 | 22,737,302 | 3,484,449 | 39,064,743 | (3,140,982 | ) | 1,087,376 | 63,253,922 | |||||||||||||||||||||||
| Acquisition of Tajite | (1,461 | ) | (1,461 | ) | ||||||||||||||||||||||||||||
| Stock issuance | 200,000 | 200 | 433,800 | 434,000 | ||||||||||||||||||||||||||||
| Net income (loss) for the year | 7,587,916 | (59,354 | ) | 7,528,562 | ||||||||||||||||||||||||||||
| Appropriation of statutory reserve | 601,370 | (601,370 | ) | - | ||||||||||||||||||||||||||||
| Foreign currency translation gain | 1,631,770 | 26,888 | 1,658,658 | |||||||||||||||||||||||||||||
| Balance at June 30, 2018 | 21,234,072 | $ | 21,234 | $ | 23,171,102 | $ | 4,085,819 | $ | 46,051,289 | $ | (1,509,212 | ) | $ | 1,053,449 | $ | 72,873,681 | ||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements
F-5
SHINECO, INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
| For the Years Ended June 30, | ||||||||
| 2018 | 2017 | |||||||
| CASH FLOWS FROM OPERATING ACTIVITIES: | ||||||||
| Net income | $ | 7,528,562 | $ | 8,619,645 | ||||
| Adjustments to reconcile net income to net cash provided by (used in) operating activities: | ||||||||
| Depreciation and amortization | 698,232 | 575,196 | ||||||
| Loss (gain) from disposal of property and equipment | 5,557 | (8,063 | ) | |||||
| Provision (recovery of) for doubtful accounts | 262,013 | (342,041 | ) | |||||
| Increase in inventory reserve | 124,601 | 37,292 | ||||||
| Deferred tax (benefit) provision | (28,138 | ) | 86,780 | |||||
| Income from equity method investments | (907,794 | ) | (927,697 | ) | ||||
| Interest income from loans to related parties | - | (86,355 | ) | |||||
| Impairment loss on goodwill | 2,153,298 | - | ||||||
| Changes in operating assets and liabilities: | ||||||||
| Accounts receivable | (3,569,821 | ) | (8,136,668 | ) | ||||
| Advances to suppliers | (2,563,943 | ) | (2,339,757 | ) | ||||
| Inventories | (25,031 | ) | 2,122,982 | |||||
| Other receivables | 170,125 | (72,891 | ) | |||||
| Prepaid expense and other assets | 4,442 | (401,755 | ) | |||||
| Due from related parties | 126,293 | (976,937 | ) | |||||
| Prepaid leases | 485,382 | 466,759 | ||||||
| Accounts payable | 2,145,058 | (96,137 | ) | |||||
| Advances from customers | (70,459 | ) | (3,950 | ) | ||||
| Other payables | 1,272,263 | (1,614,992 | ) | |||||
| Taxes payable | 2,036,079 | 354,453 | ||||||
| NET CASH PROVIDED BY (USED IN) OPERATING ACTIVITIES | 9,846,719 | (2,744,136 | ) | |||||
| CASH FLOWS FROM INVESTING ACTIVITIES: | ||||||||
| Acquisitions of property and equipment | (1,763,160 | ) | (49,863 | ) | ||||
| Proceeds from disposal of property and equipment | 607 | 17,688 | ||||||
| Payment for construction in progress | 58,671 | - | ||||||
| Repayments of loans from third parties | 831,716 | 4,839 | ||||||
| Loan advances to related party | (53,793 | ) | - | |||||
| Repayments of loans from related parties | - | 565,739 | ||||||
| Income received from investments in unconsolidated entities | 153,695 | 990,839 | ||||||
| Deposit for business acquisition | - | (2,055,074 | ) | |||||
| Deposit for potential investment | - | (200,000 | ) | |||||
| Cash of subsidiary acquired | 23,304 | - | ||||||
| NET CASH USED IN INVESTING ACTIVITIES | (748,960 | ) | (725,832 | ) | ||||
| CASH FLOWS FROM FINANCING ACTIVITIES: | ||||||||
| Proceeds from short-term loans | 2,459,122 | 2,673,064 | ||||||
| Repayment of short-term loans | (2,877,044 | ) | (2,701,321 | ) | ||||
| Proceeds from initial public offering, net of offering costs | - | 5,394,549 | ||||||
| Repayments of advances from related parties | (67,561 | ) | 17,683 | |||||
| NET CASH (USED IN) PROVIDED BY FINANCING ACTIVITIES | (485,483 | ) | 5,383,975 | |||||
| EFFECT OF EXCHANGE RATE CHANGE ON CASH | (279,774 | ) | (768,830 | ) | ||||
| NET INCREASE IN CASH | 8,332,502 | 1,145,177 | ||||||
| CASH - Beginning of the Period | 23,154,551 | 22,009,374 | ||||||
| CASH - End of the Period | $ | 31,487,053 | $ | 23,154,551 | ||||
| SUPPLEMENTAL CASH FLOW DISCLOSURES: | ||||||||
| Cash paid for income taxes | $ | 857,201 | $ | 845,792 | ||||
| Cash paid for interest | $ | 133,930 | $ | 150,175 | ||||
| SUPPLEMENTAL NON-CASH INVESTING ACTIVITY: | ||||||||
| Issued 200,000 shares of deferred issuance cost | $ | 434,000 | $ | - | ||||
The accompanying notes are an integral part of these consolidated financial statements
F-6
NOTE 1 - ORGANIZATION AND NATURE OF OPERATIONS
Shineco, Inc. (“Shineco” or the “Company”) was incorporated in the State of Delaware on August 20, 1997. The Company is a holding company whose primary purpose is to develop business opportunities in the People’s Republic of China (“PRC” or “China”).
On December 30, 2004, the Company acquired all of the issued and outstanding shares of Beijing Tenet-Jove Technological Development Co., Ltd. (“Tenet-Jove”), a PRC company, in exchange for restricted shares of the Company’s common stock, and the sole operating business of the Company became that of its subsidiary, Tenet-Jove. Tenet-Jove was incorporated on December 15, 2003 under the laws of China. Consequently, Tenet-Jove became a 100% owned subsidiary of Shineco and was officially granted the status of a Wholly Foreign-Owned Entity (“WFOE”) by Chinese authorities on July 14, 2006. This transaction was accounted for as a recapitalization. Tenet-Jove owns 90% interest of Tianjin Tenet Huatai Technological Development Co., Ltd. (“Tenet Huatai”).
On December 31, 2008, June 11, 2011 and May 24, 2012, Tenet-Jove entered into a series of contractual agreements including an Executive Business Cooperation Agreement, a Timely Reporting Agreement, an Equity Interest Pledge Agreement and Executive Option Agreement (collectively, the “VIE Agreements”), with each one of the following entities, Ankang Longevity Pharmaceutical (Group) Co., Ltd. (“Ankang Longevity Group”), Yantai Zhisheng International Freight Forwarding Co., Ltd. (“Zhisheng Freight”), Yantai Zhisheng International Trade Co., Ltd. (“Zhisheng Trade”), Yantai Mouping District Zhisheng Agricultural Produce Cooperative (“Zhisheng Agricultural”) and Qingdao Zhihesheng Agricultural Produce Services., Ltd. (“Qingdao Zhihesheng”). On February 24, 2014, Tenet-Jove entered into the same series of contractual agreements with Shineco Zhisheng (Beijing) Bio-Technology Co., Ltd. (“Zhisheng Bio-Tech”), which was incorporated in 2014. Zhisheng Bio-Tech, Zhisheng Freight, Zhisheng Trade, Zhisheng Agricultural, and Qingdao Zhihesheng are collectively referred to herein as the “Zhisheng Group”.
Pursuant to the VIE Agreements, Tenet-Jove has the exclusive right to provide to Zhisheng Group and Ankang Longevity Group consulting services related to their business operations and management. All the above contractual agreements obligate Tenet-Jove to absorb a majority of the risk of loss from the Zhisheng Group and Ankang Longevity Group’s activities and entitle Tenet-Jove to receive a majority of their residual returns. In essence, Tenet-Jove has gained effective control over the Zhisheng Group and Ankang Longevity Group. Therefore, the Zhisheng Group and Ankang Longevity Group are treated as Variable Interest Entities (“VIEs”) under the Financial Accounting Standards Board (“FASB”) Accounting Standards Codification (“ASC”) 810 “Consolidation”. Accordingly, the accounts of these entities are consolidated with those of Tenet-Jove.
Since Shineco is effectively controlled by the majority shareholders of the Zhisheng Group and Ankang Longevity Group, Shineco owns 100% of Tenet-Jove. Accordingly, Shineco, Tenet-Jove, and its VIEs, the Zhisheng Group and Ankang Longevity Group are effectively controlled by the same majority shareholders. Therefore, Shineco, Tenet-Jove and its VIEs are considered under common control. The consolidation of Tenet-Jove and its VIEs into Shineco was accounted for at historical cost and prepared on the basis as if the aforementioned exclusive contractual agreements between Tenet-Jove and its VIEs had become effective as of the beginning of the first period presented in the accompanying consolidated financial statements.
On April 19, 2017, Tenet-Jove established Xinjiang Tiankunrunze Biological Engineering Co., Ltd. (“Tiankunrunze”) with registered capital of RMB 50.0 million (US$ 7,262,000) and owns 65% interest of Tiankunrunze. On April 28, 2017, Tiankunrunze established Xinjiang Tianzhuo Technology Development Co., Ltd. (“Tianzhuo”) with registered capital of RMB 10.0 million (US$ 1,450,233). On May 22, 2017, Tiankunrunze established Xinjiang Tianhuihechuang Agriculture Development Co., Ltd. (“Tianhuihechuang”) with registered capital of RMB 10.0 million (US$ 1,452,294). On May 23, 2017, Tiankunrunze established Xinjiang Tianxintongye Biotechnology Development Co., Ltd. (“Tianxintongye”) with registered capital of RMB 10.0 million (US$ 1,451,615). Therefore, Tenet-Jove controls Tiankunrunze and its wholly owned subsidiaries.
F-7
On May 2, 2017, the Company entered into a Strategic Cooperation Agreement with Beijing Zhongke Biorefinery Engineering Technology Co., Ltd. (“Biorefinery”), a leading high-tech biomass refining company financially backed by the Chinese Academy of Sciences Institute of Process Engineering, to establish the Institute of Chinese Apocynum Industrial Technology Research (“ICAITR”). Pursuant to the Strategic Cooperation Agreement the two parties agreed to establish the ICAITR and each will own 80% and 20% of the equity interests of ICAITR, respectively. Shineco invested RMB 5.0 million (US$ 737,745) as the registered capital, and Biorefinery will invest a technology patent named “Steam Explosion Degumming”.
On September 30, 2017, Tenet-Jove established Xinjiang Shineco Taihe Agriculture Technology Ltd. (“Xinjiang Taihe”) with registered capital of RMB 10.0 million (US$ 1,502,650). On September 30, 2017, Tenet-Jove established Xinjiang Tianyi Runze Bioengineering Co., Ltd. (“Runze”) with registered capital of RMB 10.0 million (US$ 1,502,650). Xinjiang Taihe and Runze became wholly-owned subsidiaries of Tenet-Jove.
On December 10, 2016, Tenet-Jove entered into a purchase agreement with Tianjin Tajite E-Commerce Co., Ltd. (“Tianjin Tajite”), an online e-commerce company based in Tianjin, China, specializing in distributing Luobuma related products and branded products of Daiso 100-yen shops, pursuant to which Tenet-Jove would acquire a 51% equity interest in Tianjin Tajite for cash consideration of RMB 14,000,000 (approximately US$ 2.1 million). On December 25, 2016, the Company paid the full amount as the deposit to secure the deal. In May, 2017, the Company amended the agreement that required Tianjin Tajite to satisfy certain preconditions related to product introductions into China. On October 26, 2017, the Company completed the acquisition for 51% of the shares in Tianjin Tajite.
On October 27, 2017, the Company, through its subsidiary Tianjin Tajite E-Commerce Co., Ltd. (“Tianjin Tajite”), obtained contractual rights to distribute branded products of Daiso Industries Co., Ltd. (“Daiso”), a large franchise of 100-yen shops founded in Japan, via JD.com (“JD”), one of the largest e-commerce companies and one of the largest retailers in China. On November 3, 2017, the Company further developed the cooperation with Daiso by entering into a supply and purchase agreement (the “Daiso Agreement”) for the purpose of establishing a continuous supply and sale of Daiso’s products in China. Pursuant to the Daiso Agreement, the Company planned to purchase Daiso Products in the amount of approximately RMB 20 million by August, 2018 and add orders as circumstance requires. The term of the Daiso Agreement is for one year, and it renews for an additional one-year at the end of each term unless terminated by written notice by either Tianjin Tajite or Daiso. Due to the policy of China Customs, many of the bestselling products of Daiso are not allowed to be imported through general form of trade model, but only through cross-border e-commence business model. As a result, the Company suspends the cooperation with Daiso temporarily and waits for the opening of the China-Japan-South Korea Free Trade Zone.
On November 1, 2017, the Company established an Apocynum Industrial Park in Xinjiang, China. The industrial park is focusing on planting and purchasing Bluish Dogbane, processing and distributing Bluish Dogbane preliminary products.
The Company, its subsidiaries, its VIEs and its VIEs’ subsidiaries (collectively the “Group”) operate three main business segments: 1) Tenet-Jove is engaged in manufacturing and selling of Bluish Dogbane and related products, also known in Chinese as “Luobuma”, including therapeutic clothing and textile products made from Luobuma; 2) Zhisheng Group is engaged in the business of planting, processing and distributing of green agricultural produce as well as providing domestic and international logistic services for agricultural products (“Agricultural Products”); and, 3) Ankang Longevity Group manufactures traditional Chinese medicinal herbal products as well as other retail pharmaceutical products. These different business activities and products can potentially be integrated and benefit from one another.
F-8
NOTE 2 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation and Principles of Consolidation
The accompanying consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America (“US GAAP”).
The consolidated financial statements of the Company reflect the principal activities of the Company, its subsidiaries, its VIEs and its VIEs’ subsidiaries. The non-controlling interest represents the minority shareholders’ interest in the Company’s majority owned subsidiaries and VIEs. All intercompany accounts and transactions have been eliminated in consolidation.
Consolidation of Variable Interest Entities
VIEs are generally entities that lack sufficient equity to finance their activities without additional financial support from other parties or whose equity holders lack adequate decision making ability. All VIEs and their subsidiaries with which the Company is involved must be evaluated to determine the primary beneficiary of the risks and rewards of the VIE. The primary beneficiary is required to consolidate the VIE for financial reporting purposes.
The carrying amount of the VIEs and their subsidiaries’ consolidated assets and liabilities are as follows:
| June 30, 2018 | June 30, 2017 | |||||||
| Current assets | $ | 49,812,314 | $ | 40,584,817 | ||||
| Plant and equipment, net | 9,818,518 | 8,958,282 | ||||||
| Other noncurrent assets | 11,194,017 | 10,707,344 | ||||||
| Total assets | 70,824,849 | 60,250,443 | ||||||
| Total liabilities | (5,014,036 | ) | (4,662,387 | ) | ||||
| Net assets | $ | 65,810,813 | $ | 55,588,056 | ||||
Non-controlling Interests
US GAAP requires that non-controlling interests in subsidiaries and affiliates be reported in the equity section of a company’s balance sheet. In addition, the amounts attributable to the non-controlling interests in the net income (loss) of these entities are reported separately in the consolidated statements of income and comprehensive income.
Risks and Uncertainties
The operations of the Company are located in the PRC. Accordingly, the Company’s business, financial condition, and results of operations may be influenced by the political, economic, and legal environment in the PRC, as well as by the general state of the PRC economy. The Company’s operations in the PRC are subject to special considerations and significant risks not typically associated with companies in North America and Western Europe. These include risks associated with, among other factors, the political, economic and legal environment and foreign currency exchange. The Company’s results may be adversely affected by changes in the political, regulatory and social conditions in the PRC, and by changes in governmental policies or interpretations with respect to laws and regulations, anti-inflationary measures, currency conversion, remittances abroad, and rates and methods of taxation, among other things. Although the Company has not experienced losses from these situations and believes that it is in compliance with existing laws and regulations, changes could affect the Company’s interest in these entities.
Members of the current management team own controlling interests in the Company and are also the owners of the VIEs in the PRC. The Company only controls the VIEs through contractual arrangements which obligate it to absorb the risk of loss and to receive the residual expected returns. As such, the controlling shareholders of the Company and the VIEs could cancel these agreements or permit them to expire at the end of the agreement terms, as a result of which the Company would not retain control of the VIEs. In addition, should these agreements be challenged or litigated, they would also be subject to the laws and courts of the PRC legal system which could make enforcing the Company’s rights difficult.
F-9
Use of Estimates
The preparation of the consolidated financial statements in conformity with US GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the consolidated financial statements as well as the reported amounts of revenue and expenses during the reporting periods. Significant estimates required to be made by management include, but are not limited to, useful lives of property, plant, and equipment, and intangible assets, the recoverability of long-lived assets and the valuation of accounts receivable, deferred taxes and inventory reserves. Actual results could differ from those estimates.
Revenue Recognition
The Company recognizes revenue from sales of Luobuma products, Chinese medicinal herbal products and agricultural products, as well as providing logistic services and other processing services to external customers. The Company recognizes revenue when all of the following have occurred: (i) there is persuasive evidence of an arrangement with a customer; (ii) delivery has occurred or services have been rendered; (iii) the sales price is fixed or determinable; and (iv) the Company’s collection of such fees is reasonably assured. These criteria, as related to the Company’s revenue, are considered to have been met as follows:
Sales of products: The Company recognizes revenue from the sale of products when the goods are delivered and title to the goods passes to the customer provided that there are no uncertainties regarding customer acceptance; persuasive evidence of an arrangement exists; the sales price is fixed or determinable; and collectability is deemed probable.
Revenue from the rendering of services: Revenue from international freight forwarding, domestic air and overland freight forwarding services is recognized upon the performance of services as stipulated in the underlying contract or when commodities are being released from the customer’s warehouse; the service price is fixed or determinable; and collectability is deemed probable.
Cash and Cash Equivalents
Cash and cash equivalents consist of cash on hand, cash on deposit and other highly liquid investments which are unrestricted as to withdrawal or use, and which have original maturities of three months or less when purchased. The Company maintains cash with various financial institutions mainly in the PRC. As of June 30, 2018 and 2017, the Company had no cash equivalents.
Under PRC law, it is generally required that a commercial bank in the PRC that holds third party cash deposits protect the depositors’ rights over and interests in their deposited money. PRC banks are subject to a series of risk control regulatory standards, and PRC bank regulatory authorities are empowered to take over the operation and management of any PRC bank that faces a material credit crisis.
Accounts Receivable
Accounts receivable are recorded at net realizable value consisting of the carrying amount less an allowance for uncollectible accounts, as necessary. The Company reviews the accounts receivable on a periodic basis and makes general and specific allowances when there is doubt as to the collectability of individual balances. In evaluating the collectability of individual receivable balances, the Company considers many factors, including the age of the balance, the customers’ historical payment history, their current credit-worthiness and current economic trends. As of June 30, 2018 and 2017, the allowance for doubtful accounts was US$ 232,355 and US$ 48,450, respectively. Accounts are written off against the allowance after efforts at collection prove unsuccessful.
F-10
Inventories
Inventories, which are stated at the lower of cost or current market value, consist of raw materials, work-in-progress, and finished goods related to the Company’s products. Cost is determined using the first in first out (“FIFO”) method. Market value is the lower of replacement cost or net realizable value. Agricultural products that the Company farms are recorded at cost, which includes direct costs such as seed selection, fertilizer, labor cost and contract fees that are spent in growing agricultural products on the leased farmland, and indirect costs which include amortization of prepayments of farmland leases and farmland development costs. All the costs are accumulated until the time of harvest and then allocated to the harvested crops costs when they are sold. The Company periodically evaluates its inventory and records an inventory reserve for certain inventories that may not be saleable or whose cost exceeds market prices.
Advances to Suppliers
Advances to suppliers consist of payments to suppliers for materials that have not been received. Advances to suppliers are reviewed periodically to determine whether their carrying value has become impaired. As of June 30, 2018 and 2017, the Company had an allowance for uncollectible advances to suppliers of US$ 13,819 and US$ 17,618, respectively.
Loans to Third Parties
Loans to third parties consist of various cash advances to unrelated companies and individuals, with whom the Company has business relationships. The loans are due within one year with no interest. Loans to third parties are reviewed periodically as to whether their carrying values remain realizable.
Business Acquisitions
Business acquisitions are accounted for under the acquisition method. The acquisition method requires the reporting entity to identify the acquirer, determine the acquisition date, recognize and measure the identifiable assets acquired, the liabilities assumed and any non-controlling interest in the acquired entity, and recognize and measure goodwill or a bargain gain from the purchase. The acquiree’s results are included in the Company’s consolidated financial statements from the date of acquisition. Assets acquired and liabilities assumed are recorded at their fair values on the date acquired and the excess of the purchase price over the amounts assigned is recorded as goodwill, or if the fair value of the net assets acquired exceeds the purchase price consideration, a bargain purchase gain is recorded. Adjustments to fair value assessments are generally recorded to goodwill over the measurement period (not longer than twelve months). The acquisition method also requires that acquisition-related transaction and post-acquisition restructuring costs be charged to expense as committed, and requires the Company to recognize and measure certain assets and liabilities including those arising from contingencies and contingent consideration in a business combination.
Goodwill
Goodwill represents the excess of the purchase price over the fair value of assets acquired. The goodwill impairment test compares the fair value of a reporting unit with its carrying amount, including goodwill. If the carrying amount of a reporting unit exceeds its fair value, goodwill of the reporting unit would be considered impaired. To measure the amount of the impairment loss, the implied fair value of a reporting unit’s goodwill is compared to the carrying amount of that goodwill. The implied fair value of goodwill is determined in the same manner as the amount of goodwill recognized in a business combination. If the carrying amount of a reporting unit’s goodwill exceeds the implied fair value of that goodwill, an impairment loss is recognized in an amount equal to that excess. For each of these tests, the fair value of each of the Company’s reporting units is determined using a combination of valuation techniques, including a discounted cash flow methodology. To corroborate the discounted cash flow analysis performed at each reporting unit, a market approach is utilized using observable market data such as comparable companies in similar lines of business that are publicly traded or which are part of a public or private transaction (to the extent available).
F-11
Property and Equipment
Property and equipment are stated at cost, less accumulated depreciation and amortization. Expenditures for additions, major renewals and betterments are capitalized, and expenditures for maintenance and repairs are charged to expense as incurred. Depreciation is provided on a straight-line basis, less estimated residual value, if any, over an asset’s estimated useful life. Farmland leasehold improvements are amortized over the shorter of lease term or estimated useful lives of the underlying assets. The estimated useful lives of the Company’s property and equipment are as follows:
| Estimated
useful lives | |||
| Buildings | 20-50 years | ||
| Machinery equipment | 5-10 years | ||
| Motor vehicles | 5-10 years | ||
| Office equipment | 5-10 years | ||
| Farmland leasehold improvements | 12-18 years |
Land Use Rights
According to the Chinese laws and regulations regarding land use rights, land in urban districts is owned by the State, while land in the rural areas and suburban areas, except otherwise provided for by the State, is collectively owned by individuals designated as resident farmers by the State. In accordance with the legal principle that land ownership is separate from the right to the use of the land, the government grants individuals and companies the rights to use parcels of land for a specified period of time. Land use rights, which are usually prepaid, are stated at cost less accumulated amortization. Amortization is provided over the life of the land use rights, using the straight-line method. The estimated useful life is 50 years, based on the term of the land use rights.
Long-lived Assets
Finite-lived assets and intangibles are reviewed for impairment testing when circumstances require. For purposes of evaluating the recoverability of long-lived assets, when undiscounted future cash flows will not be sufficient to recover an asset’s carrying amount, the asset is written down to its fair value. The long-lived assets of the Company that are subject to evaluation consist primarily of property, plant and equipment, land use rights, investments and long-term prepaid leases. For the years ended June 30, 2018 and 2017, the Company did not recognize any impairment of its long-lived assets.
Fair Value of Financial Instruments
The Company follows the provisions of ASC 820, “Fair Value Measurements and Disclosures.” ASC 820 clarifies the definition of fair value, prescribes methods for measuring fair value, and establishes a fair value hierarchy to classify the inputs used in measuring fair value as follows:
Level 1 applies to assets or liabilities for which there are quoted prices in active markets for identical assets or liabilities.
Level 2 applies to assets or liabilities for which there are inputs other than quoted prices in level 1 that are observable for the asset or liability such as quoted prices for similar assets or liabilities in active markets; quoted prices for identical assets or liabilities in markets with insufficient volume or infrequent transactions (less active markets); or model-derived valuations in which significant inputs are observable or can be derived principally from, or corroborated by, observable market data.
Level 3 applies to assets or liabilities for which there are unobservable inputs to the valuation methodology that are significant to the measurement of the fair value of the asset or liability.
The carrying value of financial instruments included in current assets and liabilities approximate their fair values because of the short-term nature of these instruments.
F-12
Income Taxes
Deferred tax assets and liabilities are recognized for the future tax consequences attributable to differences between the consolidated financial statement carrying amounts of existing assets and liabilities and their respective tax bases. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in the results of operations in the period that includes the enactment date. A valuation allowance is established, when necessary, to reduce deferred tax assets to the amount expected to be realized.
The provisions of ASC 740-10-25, “Accounting for Uncertainty in Income Taxes,” prescribe a more-likely-than-not threshold for consolidated financial statement recognition and measurement of a tax position taken (or expected to be taken) in a tax return. This ASC also provides guidance on the recognition of income tax assets and liabilities, classification of current and deferred income tax assets and liabilities, accounting for interest and penalties associated with tax positions, and related disclosures. The Company does not have any uncertain tax positions at June 30, 2018 and 2017. The Company has not provided deferred taxes for undistributed earnings of non-U.S. subsidiaries at June 30, 2018, as it is the Company’s policy to indefinitely reinvest these earnings in non-U.S. operations. Quantification of the deferred tax liability, if any, associated with indefinitely reinvested earnings is not practicable.
The statute of limitations for the Company’s U.S. federal income tax returns and certain state income tax returns remains open for tax years 2015 and after. As of June 30, 2018, the tax years ended June 30, 2013 through June 30, 2018 for the Company’s People’s Republic of China (“PRC”) subsidiaries remain open for statutory examination by PRC tax authorities.
On December 22, 2017, the “Tax Cuts and Jobs Act” (“The Act”) was enacted. Under the provisions of The Act, the U.S. corporate tax rate decreased from 35% to 21%. As the Company has a June 30 fiscal year end, the lower corporate income tax rate will be phased in, resulting in a U.S. statutory federal rate of approximately 28% for our fiscal year ended June 30, 2018, and 21% for subsequent fiscal years. Additionally, The Act imposes a one-time transition tax on deemed repatriation of historical earnings of foreign subsidiaries, and future foreign earnings are subject to U.S. taxation. The change in rate has caused us to re-measure the Company’s income tax liability and record an estimated income tax expense of US$ 744,766 for the year ended June 30, 2018. On December 22, 2017, Staff Accounting Bulletin No. 118 (“SAB 118”) was issued to address the application of US GAAP in situations when a registrant does not have the necessary information available, prepared, or analyzed (including computations) in reasonable detail to complete the accounting for certain income tax effects of the Act. In accordance with SAB 118, additional work is necessary to do a more detailed analysis of the Act as well as potential correlative adjustments. Any subsequent adjustment to these amounts will be recorded to current tax expense in fiscal 2019 when the analysis is complete. The Company elects to pay the transition tax over an eight-year period using specified percentages (eight percent per year for the first five years, 15 percent in year six, 20 percent in year seven, and 25 percent in year eight).
Value Added Tax
Sales revenue represents the invoiced value of goods, net of a Value-Added Tax (“VAT”). Before May 1, 2018, all of the Company’s products that are sold in the PRC are subject to a Chinese value-added tax at a rate of 17% of the gross sales price, and after May 1, 2018, the Company subject a tax rate of 16% based on the newly Chinese tax law. This VAT may be offset by VAT paid by the Company on raw materials and other materials included in the cost of producing finished products or acquiring finished products. The Company records a VAT payable or VAT receivable in the accompanying consolidated financial statements.
Foreign Currency Translation
The Company uses the United States dollar (“U.S. dollars”, “USD” or “US$”) for financial reporting purposes. The Company’s subsidiaries and VIEs maintain their books and records in their functional currency of Renminbi (“RMB”), the currency of the PRC.
F-13
In general, for consolidation purposes, the Company translates the assets and liabilities of its subsidiaries and VIEs into U.S. dollars using the applicable exchange rates prevailing at the balance sheet date, and the statements of income and cash flows are translated at average exchange rates during the reporting periods. As a result, amounts related to assets and liabilities reported on the statement of cash flows will not necessarily agree with changes in the corresponding balances on the balance sheet. Equity accounts are translated at historical rates. Adjustments resulting from the translation of the financial statements of the subsidiaries and VIEs are recorded as accumulated other comprehensive income (loss).
The balance sheet amounts, with the exception of equity, at June 30, 2018 and 2017 were translated at 1 RMB to 0.1511 USD and at 1 RMB to 0.1475 USD, respectively. The average translation rates applied to the income and cash flow statement amounts for the years ended June 30, 2018 and 2017 were at 1 RMB to 0.1537 USD and at 1 RMB to 0.1468 USD, respectively.
Comprehensive Income
Comprehensive income consists of two components, net income and other comprehensive income (loss). The foreign currency translation gain or loss resulting from translation of the financial statements expressed in RMB to USD is reported in other comprehensive income (loss) in the consolidated statements of income and comprehensive income.
Equity Investment
An investment in which the Company has the ability to exercise significant influence, but does not have a controlling interest, is accounted for using the equity method. Significant influence is generally considered to exist when the Company has an ownership interest in the voting stock between 20% and 50%, and other factors, such as representation on the Board of Directors, voting rights and the impact of commercial arrangements, are considered in determining whether the equity method of accounting is appropriate.
Earnings per Share
The Company computes earnings per share (“EPS”) in accordance with ASC 260, “Earnings per Share” (“ASC 260”). ASC 260 requires companies with complex capital structures to present basic and diluted EPS. Basic EPS is measured as net income divided by the weighted average common shares outstanding for the period. Diluted EPS is similar to basic EPS but presents the dilutive effect on a per share basis of potential common shares (e.g., outstanding convertible securities, options and warrants) as if they had been converted at the beginning of the periods presented, or issuance date, if later. Potential common shares that have an anti-dilutive effect (i.e., those that increase income per share or decrease loss per share) are excluded from the calculation of diluted EPS. There was no anti-dilutive effect for the years ended June 30, 2018 and 2017.
New Accounting Pronouncements
In February 2016, the FASB issued ASU 2016-02, Amendments to the ASC 842 Leases. This update requires a lessee to recognize the assets and liability (the lease liability) arising from operating leases on the balance sheet for the lease term. When measuring assets and liabilities arising from a lease, a lessee (and a lessor) should include payments to be made in optional periods only if the lessee is reasonably certain to exercise an option to extend the lease or not to exercise an option to terminate the lease. Within a twelve months or less lease term, a lessee is permitted to make an accounting policy election not to recognize lease assets and liabilities. If a lessee makes this election, it should recognize lease expense on a straight-line basis over the lease term. In transition, this update will be effective for public entities for fiscal years beginning after December 15, 2018, including interim periods within those fiscal years. The Company does not currently expect the adoption of ASU 2016-02 to have a material impact on the Company’s financial statements unless it enters into any new long-term leases.
F-14
In April 2016, the FASB issued ASU 2016-10, Revenue from Contracts with Customers (ASC 606): Identifying Performance Obligations and Licensing. The objective is to clarify the two aspects of Topic 606: identifying performance obligations and the licensing implementation guidance, while retaining the related principles for these areas. The ASU affects the guidance in ASU 2014-09, Revenue from Contracts with Customers (ASC 606), which is not yet effective. The effective date and transition requirements for this ASU are the same as the effective date and transition requirements in ASC 606 (and any other Topic amended by ASU 2014-09). ASU 2015-14, Revenue from Contracts with Customers (ASC 606): Deferral of the Effective Date, defers the effective date of ASU 2014-09 by one year. The Company does not expect the adoption of ASU 2016-10 to have a material impact on the Company’s financial statements.
In May 2016, the FASB issued ASU 2016-12, Revenue from Contracts with Customers (ASC 606): Narrow-Scope Improvements and Practical Expedients. The objective is to address certain issues identified by the FASB-IASB Joint Transition Resource Group for Revenue Recognition. The amendments in this Update affect the guidance in Accounting Standards Update 2014-09, Revenue from Contracts with Customers (ASC 606), which is not yet effective. The effective date and transition requirements for the amendments in this Update are the same as the effective date and transition requirements for ASC 606 (and any other Topic amended by Update 2014-09). Accounting Standards Update 2015-14, Revenue from Contracts with Customers (ASC 606): Deferral of the Effective Date, defers the effective date of Update 2014-09 by one year. The Company does not expect the adoption of ASU 2016-12 to have a material impact on the Company’s financial statements.
In June 2016 the FASB issued ASU 2016-13, Financial Instruments - Credit Losses (Topic 326): Measurement of Credit Losses on Financial Instruments, which eliminates the probable initial recognition threshold for credit losses in current U.S. GAAP, and instead requires an organization to record a current estimate of all expected credit losses over the contractual term for financial assets carried at amortized cost. This is commonly referred to as the current expected credit losses (“CECL”) methodology. Expected credit losses for financial assets held at the reporting date will be measured based on historical experience, current conditions, and reasonable and supportable forecasts. ASU 2016-13 does not change the existing write-off principle in U.S. GAAP or current nonaccrual practices, nor does it change accounting requirements for loans held for sale or certain other financial assets which are measured at the lower of amortized cost or fair value. As a public business entity that is an SEC filer, ASU 2016-13 becomes effective for the Company on January 1, 2020, although early application is permitted for 2019. The Company is currently evaluating the potential effects on the Company’s financial statements, if any.
In August 2016, the FASB issued ASU 2016-15, Statement of Cash Flows (Topic 230): Classification of Certain Cash Receipts and Cash Payments, to address diversity in how certain cash receipts and cash payments are presented and classified in the statement of cash flows. The amendments provide guidance on the following eight specific cash flow issues: (1) Debt Prepayment or Debt Extinguishment Costs; (2) Settlement of Zero-Coupon Debt Instruments or Other Debt Instruments with Coupon Interest Rates That Are Insignificant in Relation to the Effective Interest Rate of the Borrowing; (3) Contingent Consideration Payments Made after a Business Combination; (4) Proceeds from the Settlement of Insurance Claims; (5) Proceeds from the Settlement of Corporate-Owned Life Insurance Policies, including Bank-Owned; (6) Life Insurance Policies; (7) Distributions Received from Equity Method Investees; (8) Beneficial Interests in Securitization Transactions; and Separately Identifiable Cash Flows and Application of the Predominance Principle. The amendments are effective for public business entities for fiscal years beginning after December 15, 2017, and interim periods within those fiscal years. Early adoption is permitted, including adoption in an interim period. The amendments should be applied using a retrospective transition method to each period presented. If it is impracticable to apply the amendments retrospectively for some of the issues, the amendments for those issues would be applied prospectively as of the earliest date practicable. The Company does not expect the adoption of ASU 2016-15 to have a material impact on the Company’s financial statements.
In November 2016, the FASB issued Accounting Standards Update 2016-18, Statement of Cash Flows (Topic 230): Restricted Cash (ASU 2016-18), which requires companies to include amounts generally described as restricted cash and restricted cash equivalents in cash and cash equivalents when reconciling beginning-of-period and end-of-period total amounts shown on the statement of cash flows. This guidance is effective for fiscal year beginning after December 31, 2018 and early adoption is permitted. The Company does not expect the adoption of ASU 2016-18 to have a material impact on the Company’s financial statements.
F-15
In May 2017, the FASB issued ASU 2017-09, Scope of Modification Accounting, which amends the scope of modification accounting for share-based payment arrangements and provides guidance on the types of changes to the terms or conditions of share-based payment awards to which an entity would be required to apply modification accounting under ASC 718. For all entities, this ASU is effective for annual reporting periods, including interim periods within those annual reporting periods, beginning after December 15, 2017. Early adoption is permitted, including adoption in any interim period. The adoption of this ASU is not expected to have a material effect on the Company’s financial statements.
In July 2017, the FASB issued ASU 2017-11, Earnings Per Share (Topic 260), Distinguishing Liabilities from Equity (Topic 480), Derivatives and Hedging (Topic 815). The amendments in Part I of this Update change the classification analysis of certain equity-linked financial instruments (or embedded features) with down round features. When determining whether certain financial instruments should be classified as liabilities or equity instruments, a down round feature no longer precludes equity classification when assessing whether the instrument is indexed to an entity’s own stock. The amendments also clarify existing disclosure requirements for equity-classified instruments. The amendments in Part II of this Update recharacterize the indefinite deferral of certain provisions of Topic 480 that now are presented as pending content in the Codification, to a scope exception. Those amendments do not have an accounting effect. For public business entities, the amendments in Part I of this Update are effective for fiscal years, and interim periods within those fiscal years, beginning after December 15, 2018. Early adoption is permitted for all entities, including adoption in an interim period. If an entity early adopts the amendments in an interim period, any adjustments should be reflected as of the beginning of the fiscal year that includes that interim period. The Company believes that the adoption of this ASU will not have a material impact on its financial statements.
In September 2017, the FASB issued ASU 2017-13, Revenue from Contracts with Customers (Topic 606) and Leases (Topic 842). The main objective of this pronouncement is to clarify the effective date of the adoption of ASC Topic 606 and ASC Topic 842 and the definition of public business entity as stipulated in ASU 2014-09 and ASU 2016-02. ASU 2014-09 provides that a public business entity and certain other specified entities adopt ASC Topic 606 for annual reporting periods beginning after December 15, 2017, including interim reporting periods within that reporting period. All other entities are required to adopt ASC Topic 606 for annual reporting periods beginning after December 15, 2018, and interim reporting periods within annual reporting periods beginning after December 15, 2019. ASU 2016-12 requires that “a public business entity and certain other specified entities adopt ASC Topic 842 for fiscal years beginning after December 15, 2018, and interim periods within those fiscal years. All other entities are required to adopt ASC Topic 842 for fiscal years beginning after December 15, 2019, and interim periods within fiscal years beginning after December 15, 2020”. ASU 2017-13 clarifies that the SEC would not object to certain public business entities electing to use the non-public business entities effective dates for applying ASC 606 and ASC 842. ASU 2017-13, however, limits such election to certain public business entities that “otherwise would not meet the definition of a public business entity except for a requirement to include or inclusion of its financial statements or financial information in another entity’s filings with the SEC”. The Company expects that the adoption of this ASU will not have a material impact on its financial statements.
In February 2018, the FASB issued ASU 2018-02, Income Statement—Reporting Comprehensive Income (Topic 220). The amendments in this update allow a reclassification from accumulated other comprehensive income to retained earnings for stranded tax effects resulting from the Tax Cuts and Jobs Act. Consequently, the amendments eliminate the stranded tax effects resulting from the Tax Cuts and Jobs Act and will improve the usefulness of information reported to financial statement users. However, because the amendments only relate to the reclassification of the income tax effects of the Tax Cuts and Jobs Act, the underlying guidance that requires that the effect of a change in tax laws or rates be included in income from continuing operations is not affected. The amendments in this update also require certain disclosures about stranded tax effects. The guidance is effective for fiscal years beginning after December 15, 2018 with early adoption permitted, including interim periods within those years. The Company expects that the adoption of this ASU will not have a material impact on its financial statements.
F-16
In June 2018, the FASB issued ASU No. 2018-07, Compensation – Stock Compensation (Topic 718): Improvements to Employee Share-Based Payment Accounting, or ASU 2018-07. ASU 2018-07 simplifies the accounting for share-based payments made to nonemployees so the accounting for such payments is substantially the same as those made to employees. Under this ASU, share based awards to nonemployees will be measured at fair value on the grant date of the awards, entities will need to assess the probability of satisfying performance conditions if any are present, and awards will continue to be classified according to Accounting Standards Codification (“ASC”) 718 upon vesting which eliminates the need to reassess classification upon vesting, consistent with awards granted to employees. This ASU is effective for fiscal years beginning after December 15, 2018, including interim periods within those fiscal years. The Company expects that the adoption of this ASU will not have a material impact on its financial statements.
The Company believes that other recent accounting pronouncement updates will not have a material effect on the Company’s consolidated financial statements.
NOTE 3 - INVENTORIES
The inventories consist of the following:
| June
30, 2018 | June 30, 2017 | |||||||
| Raw materials | $ | 1,225,830 | $ | 1,167,553 | ||||
| Work-in-process | 766,119 | 672,966 | ||||||
| Finished goods | 1,355,774 | 1,346,437 | ||||||
| Less: inventory reserve for raw materials | (983,165 | ) | (840,683 | ) | ||||
| Total | $ | 2,364,558 | $ | 2,346,273 | ||||
Work-in-process includes direct costs such as seed selection, fertilizer, labor cost and subcontractor fees that are spent in growing agricultural products on the leased farmland, and indirect costs which include amortization of the prepayment of the farmland lease fees and farmland development costs. All the costs are accumulated until the time of harvest and then allocated to harvested crop costs when they are sold.
NOTE 4 - PROPERTY AND EQUIPMENT
Property and equipment consist of the following:
| June 30, 2018 | June 30, 2017 | |||||||
| Buildings | $ | 12,433,157 | $ | 10,516,245 | ||||
| Building improvements | 82,599 | 51,797 | ||||||
| Machinery and equipment | 922,065 | 474,888 | ||||||
| Motor vehicles | 84,583 | 48,651 | ||||||
| Construction in progress | 40,524 | 442,646 | ||||||
| Office equipment | 179,624 | 153,836 | ||||||
| Farmland leasehold improvements | 3,176,677 | 3,102,803 | ||||||
| 16,919,229 | 14,790,866 | |||||||
| Less: accumulated depreciation and amortization | (5,221,925 | ) | (4,470,470 | ) | ||||
| Property and equipment, net | $ | 11,697,304 | $ | 10,320,396 | ||||
Depreciation and amortization expense charged to operations was US$ 658,268 and US$ 535,806 for the years ended June 30, 2018 and 2017, respectively.
F-17
Farmland leasehold improvements consist of following:
| June 30, 2018 | June 30, 2017 | |||||||
| Blueberry farmland leasehold reconstruction | $ | 2,440,465 | $ | 2,383,711 | ||||
| Yew tree planting base reconstruction | 273,422 | 267,064 | ||||||
| Greenhouse renovation | 462,790 | 452,028 | ||||||
| Total farmland leasehold improvements | $ | 3,176,677 | $ | 3,102,803 | ||||
NOTE 5 - LAND USE RIGHTS
Land use rights are recognized at cost less accumulated amortization. According to the Chinese laws and regulations regarding land use rights, land in urban districts is owned by the State, while land in the rural areas and suburban areas, except otherwise provided for by the State, is collectively owned by individuals designated as resident farmers by the State. However, in accordance with the legal principle that land ownership is separate from the right to the use of the land, the government grants the user a “land use right” (the “Right”) to use the land. The Company has the Right to use the land for 50 years and amortizes the rights on a straight-line basis over the period of 50 years.
| June 30, 2018 | June 30, 2017 | |||||||
| Land use rights | $ | 1,680,259 | $ | 1,641,181 | ||||
| Less: accumulated amortization | (335,171 | ) | (294,550 | ) | ||||
| Land use rights, net | $ | 1,345,088 | $ | 1,346,631 | ||||
For the years ended June 30, 2018 and 2017, the Company recognized amortization expense of US$ 39,964 and US$ 39,390, respectively.
The estimated future amortization expenses are as follows:
| Twelve months ending June 30: | ||||
| 2019 | $ | 33,605 | ||
| 2020 | 33,605 | |||
| 2021 | 33,605 | |||
| 2022 | 33,605 | |||
| 2023 | 33,605 | |||
| Thereafter | 1,177,063 | |||
| Total | $ | 1,345,088 | ||
NOTE 6 - DISTRIBUTION RIGHTS
The Company acquired distribution rights to distribute branded products of Daiso 100-yen shops through the acquisition of Tianjin Tajite. As this distribution right is difficult to acquire and will contribute significant revenue to Tianjin Tajite, such distribution rights were identified and valued as an intangible asset in the acquisition of Tianjin Tajite. The distribution rights, which have no expiration date, have been determined to have an indefinite life. Since the distribution rights have an indefinite life, the Company will evaluate them for impairment at least annually or earlier if determined necessary. As of June 30, 2018, the distribution rights were evaluated at RMB 7,380,000 (US$ 1,114,837).
F-18
NOTE 7 - INVESTMENTS
Ankang Longevity Group entered into two equity investment agreements with Shaanxi Pharmaceutical Group Pai’ang Medicine Co. Ltd. (“Shaanxi Pharmaceutical Group”), a Chinese state-owned pharmaceutical enterprise to invest a total of RMB 6.8 million (approximately US$ 1.0 million) for a 49% equity interest in a pharmacy retail company called Shaanxi Pharmaceutical Sunsimiao Drugstores Ankang Retail Chain Co., Ltd. (“Sunsimiao Drugstores”), and a 49% equity interest in a pharmaceutical wholesale distribution company named Shaanxi Pharmaceutical Holding Group Longevity Pharmacy Co., Ltd. (“Shaanxi Longevity Pharmacy”). These two equity investments were formed as new business entities to collaborate with Shaanxi Pharmaceutical Group to expand sales to regional hospitals and clinics and to establish the presence of retail pharmacies under the Brand name “Sunsimiao”. The investments are accounted for using the equity method because Ankang Longevity Group has significant influence, but no control of these two entities. Ankang Longevity Group recorded income of US$ 754,099 and US$ 817,603 for the years ended June 30, 2018 and 2017, respectively, from the investments, which was included in “Income from equity method investments” in the consolidated statements of income and comprehensive income (see Note 12).
Ankang Longevity Group entered into a supplemental agreement with Shaanxi Pharmaceutical Group. According to the supplemental agreement, the new 49% equity investment companies established by Shaanxi Pharmaceutical Group and Ankang Longevity Group are required to exclusively purchase certain raw materials and drug products from Shaanxi Pharmaceutical Group. In return, Shaanxi Pharmaceutical Group has agreed to compensate Ankang Longevity Group with a purchase rebate of 7% of the total purchases made from Shaanxi Pharmaceutical Group. For the years ended June 30, 2018 and 2017 a total of US$ 1,377,108 and US$ 1,136,162 was recognized by Ankang Longevity Group from this supplemental agreement in addition to its 49% share of the income from the equity investment companies, respectively.
On October 21, 2013, the Company, through its controlled subsidiaries, Zhisheng Freight and Zhisheng Agricultural, entered into an agreement with an unrelated third party, Zhejiang Zhen’Ai Network Warehousing Services Co., Ltd. (“Zhen’Ai Network”), and invested RMB 14.5 million (approximately US$ 2.2 million) into Tiancang Systematic Warehousing project (“Tiancang Project”) operated by Zhen’Ai Network. The Tiancang Project is an online platform established to provide comprehensive warehousing and logistic solutions to companies involved in E-commerce. The Company is entitled to 29% of Tiancang Project’s after-tax net income annually, less 30% statutory reserve and a 10 % employee welfare fund contribution. When the amount of the accumulated statutory reserve reaches 30% of the total investment for the Tiancang Project, no additional appropriation to the statutory reserve is required. For the years ended June 30, 2018 and 2017, the Company recorded investment income distributions of US$ 153,695 and US$ 110,094 from Zhen’Ai Network, respectively.
On November 21, 2016, the Company (the “Investor”) entered into an agreement with Original Lab Inc., a California corporation (the “Investee”), and made a payment of US$ 200,000 in exchange for the right to acquire certain shares of the Investee’s common and preferred stock. For the years ended June 30, 2018 and 2017, the Company did not record investment income from this investment.
The Company’s investments in unconsolidated entities consist of the following:
| June 30, 2018 | June 30, 2017 | |||||||
| Shaanxi Pharmaceutical Holding Group Longevity Pharmacy Co., Ltd. (Ankang Longevity Pharmacy) | $ | 3,439,793 | $ | 2,744,391 | ||||
| Shaanxi Pharmaceutical Sunsimiao Drugstores Ankang Chain Co., Ltd. | 736,898 | 611,228 | ||||||
| Zhejiang Zhen’Ai Network Warehousing Services Co., Ltd. | 2,190,399 | 2,139,461 | ||||||
| Original Lab Inc. | 200,000 | 200,000 | ||||||
| Total | $ | 6,567,090 | $ | 5,695,080 | ||||
F-19
Summarized financial information of unconsolidated entities is as follows:
| June 30, 2018 | June 30, 2017 | |||||||
| Current assets | $ | 38,079,702 | $ | 32,880,168 | ||||
| Noncurrent assets | 291,267 | 281,162 | ||||||
| Current liabilities | 29,862,664 | 26,328,322 | ||||||
| For
the years ended June 30, | ||||||||
| 2018 | 2017 | |||||||
| Net sales | $ | 36,788,909 | $ | 29,869,223 | ||||
| Gross profit | 4,720,094 | 4,011,307 | ||||||
| Income from operations | 1,644,747 | 1,671,820 | ||||||
| Net income | 1,538,977 | 1,668,578 | ||||||
NOTE 8 - DEPOSIT FOR BUSINESS ACQUISITION
On November 16, 2017, Xinjiang Taihe entered into a Strategic Cooperation Agreement (the “Agreement”) with Western Xinjiang Tiansheng Agricultural Development Co., Ltd. (“Xinjiang Tiansheng”), a leading nursery and agricultural company with extensive industry experience in Xinjiang, China. Pursuant to the Agreement, Xinjiang Taihe intends to acquire 51% equity interest in Xinjiang Tiansheng, in exchange for a combination of 14% equity ownership in Xinjiang Taihe and for cash consideration of RMB 23.8 million (approximately US$ 3,657,000). On December 20, 2017, the Company paid RMB 810,000 as a deposit, however, the Company and Xinjiang Tiansheng entered into an agreement to terminate the Agreement on July 10, 2018. The Company has received a refund of its deposit of RMB 810,000 from Xinjiang Tiansheng by the ended of July 2018.
NOTE 9 - PREPAID LEASES
One of the Company’s controlled subsidiaries, Zhisheng Group entered into several farmland lease contracts with farmer cooperatives to lease farmland in order to plant and grow organic vegetables, fruit and Chinese yew trees. The lease terms vary from 5 years to 24 years. The aggregate prepaid lease payments on these leases was approximately RMB 36.7 million (approximately US$ 5.5 million). Zhisheng Group was required to prepay the leases plus transfer fees at the beginning of the lease.
These leases are accounted for as operating leases and will be amortized each year on a straight-line basis over the lease terms. The amortization expense is initially recorded as work in process in the inventory account during the growing period and then transferred to harvested crops costs at the time of harvest and then allocated to cost of sales when they are sold.
Future amortization expense will be recognized as follows:
| Twelve months ending June 30: | ||||
| 2019 | $ | 477,066 | ||
| 2020 | 477,066 | |||
| 2021 | 455,414 | |||
| 2022 | 217,240 | |||
| 2023 | 217,240 | |||
| Thereafter | 1,553,546 | |||
| Total | $ | 3,397,572 | ||
F-20
NOTE 10 - SHORT-TERM LOANS
Short-term loans consist of the following:
| Lender | June
30, 2018 |
Maturity
Date |
Int.
Rate/Year |
|||||||
| MY Bank-a | 50,354 | 2018-10-20 | 11.84 | % | ||||||
| Agricultural Bank of China-d | 302,124 | 2018-7-3 | * | 5.22 | % | |||||
| Agricultural Bank of China-d | 755,310 | 2018-10-12 | 5.66 | % | ||||||
| Agricultural Bank of China-d | 1,208,495 | 2019-1-30 | 5.66 | % | ||||||
| Total | $ | 2,316,283 | ||||||||
| Lender | June
30, 2017 |
Maturity
Date |
Int.
Rate/Year |
|||||||
| Wanxiang Trust Co., Ltd-a | 7,746 | 2017-9-9 | * | 13.48 | % | |||||
| Agricultural Bank of China-b | 295,098 | 2017-10-16 | * | 5.22 | % | |||||
| Agricultural Bank of China-c | 737,745 | 2017-8-17 | * | 5.66 | % | |||||
| Agricultural Bank of China-d | 1,180,392 | 2017-12-7 | * | 5.22 | % | |||||
| Agricultural Bank of China-e | 442,647 | 2017-11-15 | * | 5.22 | % | |||||
| Total | $ | 2,663,628 | ||||||||
The loans outstanding were guaranteed by the following properties, entities or individuals:
| a. | Not collateralized or guaranteed. |
| b. | Collateralized by the building owned by Xiaoyan Chen and Jing Chen, who are both related parties of the Company. Xiaoyan Chen is one of the shareholders of Ankang Longevity Group. Jing Chen is the sister of the Xiaoyan Chen but not a shareholder of Ankang Longevity Group. |
| c. | Guaranteed by commercial credit guaranty companies unrelated to the Company. |
| d. | Guaranteed by a commercial credit guaranty company, unrelated to the Company and also by Jiping Chen, a shareholder of the Company. |
| e. | Guaranteed by a third-party company and also by Jiping Chen, a shareholder of the Company. |
| * | The Company repaid the loan in full on maturity date. |
The Company recorded interest expense of US$ 133,930 and US$ 148,793 for the years ended June 30, 2018 and 2017, respectively. The annual weighted average interest rates are 5.64% and 5.36% for the years ended June 30, 2018 and 2017, respectively.
Note 11 - ACQUISITION
On December 12, 2016, the Company entered into a merger and acquisition agreement with Tianjin Tajite E-Commerce Co., Ltd. (“Tianjin Tajite”), a professional e-commerce company distributing Luobuma fabric commodities and branded products of Daiso 100-yen shops, based in Tianjin, China, to acquire a 51 % equity interest of Tianjin Tajite.
Pursuant to the agreement, the Company made a payment of RMB 14,000,000 (approximately US$ 2.1 million) at the end of December, 2016 as the total consideration for the acquisition of Tianjin Tajite.
F-21
On October 26, 2017, the Company completed the acquisition of Tianjin Tajite. The acquisition provides a unique opportunity for the Company to enter the market of Luobuma fabric commodities and branded products of Daiso 100-yen shops.
The transaction was accounted for in accordance with the provisions of ASC 805-10, Business Combinations. The Company retained independent appraisers to advise management in the determination of the fair value of the various assets acquired and liabilities assumed. The values assigned in these financial statements represent management’s best estimate of fair values as of the Acquisition Date.
As required by ASC 805-20, Business Combinations—Identifiable Assets and Liabilities, and Any Noncontrolling Interest, management conducted a review to reassess whether they identified all the assets acquired and all the liabilities assumed, and followed ASC 805-20’s measurement procedures for recognition of the fair value of net assets acquired.
The following table summarizes the allocation of estimated fair values of net assets acquired and liabilities assumed:
| Accounts receivable, net | 28,036 | |||
| Inventory | 60,288 | |||
| Other current assets | 191,632 | |||
| Distribution rights | 1,114,837 | |||
| Property, plant and equipment | 14,594 | |||
| Advance from customers | (81,184 | ) | ||
| Tax payable | (17,523 | ) | ||
| Deferred tax liabilities | (278,709 | ) | ||
| Salary payable | (26,057 | ) | ||
| Accrued liabilities and other current liabilities | (1,031,839 | ) | ||
| Non-controlling interest | 1,480 | |||
| Goodwill | 2,116,408 | |||
| Total purchase price for acquisition, net of US$ 22,905 of cash | $ | 2,091,963 |
The excess of the purchase price over the aggregate fair value of assets acquired was allocated to goodwill. The results of operations of Tianjin Tajite have been included in the consolidated statements of operations from the date of acquisition.
In conjunction with the preparation of our consolidated financial statement for year ended June 30, 2018, the management performed evaluation on the impairment of goodwill. Due to the lower than expected revenue and profit, and unfavorable business environment, the management fully recorded a US$ 2,116,408 impairment loss on goodwill of Tianjin Tajite.
The fair value of distribution rights and its estimated useful lives is as follows:
| Preliminary
Fair Value | Weighted
Average Useful Life (in Years) | |||||||
| Distribution rights | $ | 1,114,837 | (a) | |||||
| (a) | The distribution rights with no expiration date has been determined to have an indefinite life. |
F-22
Under ASC 805-10, acquisition-related costs (i.e., advisory, legal, valuation and other professional fees) are not included as a component of consideration transferred, but are expensed in the periods in which the costs are incurred. Acquisition-related costs were nil in the year ended June 30, 2018.
The Company has included the operating results of Tianjin Tajite in its consolidated financial statements since the Acquisition Date, which includes US$ 279,989 in net sales and US$ 405,247 in net loss of Tianjin Tajite.
The following unaudited pro forma condensed financial information presents the combined results of operations for the years ended June 30, 2018 and 2017 of Shineco, Inc and Tianjin Tajite as if the acquisition had occurred as of the beginning of each period presented (in thousands except per share amounts):
| Pro
Forma Combined Years Ended June 30, | ||||||||
| 2018 | 2017 | |||||||
| Net sales | $ | 44,058 | $ | 33,922 | ||||
| Net income | 7,327 | 8,008 | ||||||
| Net income per common share, basic and diluted | $ | 0.35 | $ | 0.41 | ||||
| Shares outstanding, basic and diluted | 21,119 | 20,616 | ||||||
The unaudited pro forma condensed financial information is not intended to represent or be indicative of the consolidated results of operations of the Company that would have been reported had the acquisition been completed as of the beginning of the period presented, and should not be taken as being representative of the future consolidated results of operations of the Company.
NOTE 12 - RELATED PARTY TRANSACTIONS
DUE FROM RELATED PARTIES
The Company had previously made temporary advances to certain shareholders of the Company and to other entities that are either owned by family members of those shareholders or to other entities that the Company has investments in. Those advances are due on demand, non-interest bearing.
As of June 30, 2018 and 2017, the outstanding amounts due from related parties consist of the following
| June 30, 2018 | June 30, 2017 | |||||||
| Yang Bin | $ | 151,063 | $ | 147,550 | ||||
| Zhang Xin | 93,658 | 91,480 | ||||||
| Chang Song | 59,669 | 73,037 | ||||||
| Zhang Xinyu | - | 61,441 | ||||||
| Zhang Hua | - | 28,034 | ||||||
| Beijing Huiyinansheng Asset Management Co., Ltd | 22,690 | 22,132 | ||||||
| Zhang Yuying | - | 15,567 | ||||||
| Wang Qiwei | 61,181 | 8,117 | ||||||
| Tian Shuangpeng | - | 1,475 | ||||||
| $ | 388,261 | $ | 448,833 | |||||
F-23
DUE TO RELATED PARTIES
As of June 30, 2018 and 2017, the Company had related party payables of US$ 197,617 and US$ 257,880, respectively, mainly due to the principal shareholders or certain relatives of the shareholders of the Company who lend funds for the Company’s operations. The payables are unsecured, non-interest bearing and due on demand.
| June
30, 2018 | June 30, 2017 | |||||||
| Wu Yang | $ | 96,755 | $ | 94,505 | ||||
| Wang Sai | - | 71,942 | ||||||
| Zhao Min | 100,862 | 91,433 | ||||||
| $ | 197,617 | $ | 257,880 | |||||
SALES TO RELATED PARTIES
For the years ended June 30, 2018 and 2017, the Company recorded sales to Shaanxi Pharmaceutical Group, a related party (see Note 7), of US$ 3,285,311 and US$ 3,038,195, respectively. As of June 30, 2018 and 2017, the balance of accounts receivable due from Shaanxi Pharmaceutical Group was US$ 1,526,351 and US$ 2,205,453, respectively.
NOTE 13 - TAXES
(a) Corporate Income Taxes
The Company is subject to income taxes on an entity basis on income arising in or derived from the location in which each entity is domiciled.
Shineco is incorporated in the United States and has no operating activities. Tenet-Jove and its VIEs entities are governed by the Income Tax Laws of the PRC, and are currently subject to tax at a statutory rate of 25% on taxable income. Two VIE entities and Xinjiang Taihe receive a full income tax exemption from the local tax authority of the PRC as agricultural enterprises as long as the favorable tax policy remains unchanged.
On December 22, 2017, the “Tax Cuts and Jobs Act” (“The Act”) was enacted, The Act imposes a one-time transition tax on deemed repatriation of historical earnings of foreign subsidiaries, and future foreign earnings are subject to U.S. taxation. The change in rate has caused us to re-measure the Company’s income tax liability and record an estimated income tax expense of US$ 744,766 for the year ended June 30, 2018. In accordance with SAB 118, additional work is necessary to do a more detailed analysis of the Act as well as potential correlative adjustments. Any subsequent adjustment to these amounts will be recorded to current tax expense in fiscal 2019 when the analysis is complete. The Company elects to pay the transition tax over an eight year period using specified percentages (eight percent per year for the first five years, 15 percent in year six, 20 percent in year seven, and 25 percent in year eight).
| i) | The components of the income tax expense are as follows: |
| For
the years ended June 30, | ||||||||
| 2018 | 2017 | |||||||
| Current income tax provision | $ | 2,151,725 | $ | 1,165,857 | ||||
| Deferred income tax provision (benefit) | (28,138 | ) | 86,780 | |||||
| Total | $ | 2,123,587 | $ | 1,252,637 | ||||
| ii) | The following table summarizes deferred tax assets resulting from differences between the financial reporting basis and tax basis of assets and liabilities: |
F-24
| June
30, 2018 | June 30, 2017 | |||||||
| Deferred tax assets: | ||||||||
| Allowance for doubtful accounts | $ | 22,225 | $ | 24,598 | ||||
| Inventory reserve | 244,832 | 209,236 | ||||||
| Net operating loss carry-forwards | 539,061 | 111,882 | ||||||
| Total | 806,118 | 345,716 | ||||||
| Valuation allowance | (539,061 | ) | (111,882 | ) | ||||
| Total deferred tax assets | 267,057 | 233,834 | ||||||
| Deferred tax liability: | ||||||||
| Distribution rights | (278,709 | ) | - | |||||
| Total deferred tax liability | (278,709 | ) | - | |||||
| Deferred tax assets (liability), net | $ | (11,652 | ) | $ | 233,834 | |||
Movement of the valuation allowance:
| June
30, 2018 | June 30, 2017 | |||||||
| Beginning balance | $ | 111,882 | $ | 114,122 | ||||
| Current year addition | 424,517 | - | ||||||
| Exchange difference | 2,662 | (2,240 | ) | |||||
| Ending balance | $ | 539,061 | $ | 111,882 | ||||
(b) Value Added Tax
The Company is subject to a value added tax (“VAT”) for selling merchandise. The applicable VAT rate is 17% before May 1, 2018 for products sold in the PRC and decreased to 16% thereafter. The amount of VAT liability is determined by applying the applicable tax rate to the invoiced amount of goods sold (output VAT) less VAT paid on purchases made with the relevant supporting invoices (input VAT). Under commercial practice in the PRC, the Company pays VAT based on tax invoices issued. The tax invoices may be issued subsequent to the date on which revenue is recognized, and there may be a considerable delay between the date on which the revenue is recognized and the date on which the tax invoice is issued.
In the event that the PRC tax authorities dispute the date on which revenue is recognized for tax purposes, the PRC tax office has the right to assess a penalty based on the amount of the taxes which are determined to be late or deficient, and will be expensed in the period if and when a determination is made by the tax authorities. There were no assessed penalties during the years ended June 30, 2018 and 2017.
(c) Taxes Payable
Taxes payable consists of the following:
| June 30, 2018 | June 30, 2017 | |||||||
| Income tax payable | $ | 3,106,642 | $ | 1,541,548 | ||||
| Value added tax payable | 562,960 | 60,685 | ||||||
| Business tax and other taxes payable | 7,207 | 6,693 | ||||||
| Total | 3,676,809 | 1,608,926 | ||||||
| Less: current portion | 2,991,624 | 1,608,926 | ||||||
| Income tax payable - noncurrent portion | $ | 685,185 | $ | - | ||||
F-25
NOTE 14 - SHAREHOLDERS’ EQUITY
Initial Public Offering
On September 28, 2016, the Company completed its initial public offering of 1,713,190 shares of common stock at a price of US$ 4.50 per share for gross proceeds of US$ 7.7 million and net proceeds of approximately US$ 5.4 million. The Company’s common shares began trading on September 28, 2016 on the NASDAQ Capital Market under the symbol “TYHT.”
Statutory Reserve
The Company is required to make appropriations to reserve funds, comprising the statutory surplus reserve and discretionary surplus reserve, based on after-tax net income determined in accordance with generally accepted accounting principles of the PRC (“PRC GAAP”).
Appropriations to the statutory surplus reserve are required to be at least 10% of the after tax net income determined in accordance with PRC GAAP until the reserve is equal to 50% of the entities’ registered capital. Appropriations to the discretionary surplus reserve are made at the discretion of the Board of Directors. As of June 30, 2018 and 2017, the balance of the required statutory reserves were US$ 4,085,819 and US$ 3,484,449, respectively.
On January 23, 2018, Shineco, Inc. entered into a Common Stock Purchase Agreement (“Purchase Agreement”) with IFG Opportunity Fund LLC (“IFG Fund”) whereby, upon the terms and subject to the conditions and limitations set forth therein, the Company has the right, from time to time in its sole discretion during the 24-month term of the Purchase Agreement, to direct IFG Fund to purchase up to a total of US$ 15,000,000 worth of shares of common stock. As consideration for IFG Fund to enter into the Purchase Agreement, the Company agreed to issue 200,000 shares of the Company’s Common Stock (the “Commitment Shares”) to IFG Fund. The Purchase Shares are being offered in an indirect primary offering consisting of an equity line of credit, in accordance with the terms and conditions of the Purchase Agreement. The total number of Purchase Shares shall not exceed 4,000,000. On January 23, 2018, the Company issued the Commitment Shares to IFG Fund.
NOTE 15 - CONCENTRATIONS AND RISKS
The Company maintains principally all bank accounts in the PRC. The cash balance held in the PRC bank accounts was US$ 31,423,686 and US$ 23,112,124 as of June 30, 2018 and 2017, respectively.
During the years ended June 30, 2018 and 2017, almost 100% of the Company’s assets were located in the PRC and 100% of the Company’s revenues were derived from its subsidiaries and VIEs located in the PRC.
For the year ended June 30, 2018, two customers accounted for approximately 21% and 10% of the Company’s total sales, respectively. At June 30, 2018, five customers accounted for approximately 76% of the Company’s accounts receivable.
For the year ended June 30, 2017, two customers accounted for approximately 11% and 10% of the Company’s total sales, respectively. At June 30, 2017, four customers accounted for approximately 54% of the Company’s accounts receivable.
For the year ended June 30, 2018, three vendors accounted for approximately 36%, 16% and 11% of the Company’s total purchases, respectively. For the year ended June 30, 2017, two vendors accounted for approximately 23% and 14% of the Company’s total purchases, respectively.
F-26
NOTE 16 - COMMITMENTS AND CONTIGENCIES
Lease Commitments
The Company leases four main office spaces under non-cancelable operating lease agreements through December 31, 2020. The Company also leases farmland under a non-cancelable operating lease agreement through April 26, 2041. Most of those operating lease payments are scheduled on a quarterly basis. The future minimum rental payments are as follows:
| Twelve months ending June 30: | ||||
| 2019 | $ | 569,778 | ||
| 2020 | 449,576 | |||
| 2021 | 288,530 | |||
| 2022 | 221,321 | |||
| 2023 | 221,321 | |||
| Thereafter | 3,946,890 | |||
| Total | $ | 5,697,416 | ||
Rent expense totaled US$ 546,098 and US$ 613,811 for the years ended June 30, 2018 and 2017, respectively.
In addition, the Company sublets the above-mentioned farmland to a third party under a non-cancelable operating lease agreement through May 31, 2020. The future minimum sublease rental income to be received is as follows:
| Twelve months ending June 30: | ||||
| 2019 | $ | 221,321 | ||
| 2020 | 202,878 | |||
| Total | $ | 424,199 | ||
Sublease rental income totaled US$ 221,321 and US$ 211,379 for the years ended June 30, 2018 and 2017, respectively.
Legal Contingencies
On May 16, 2017, Bonwick Capital Partners, LLC (“Plaintiff”) commenced a lawsuit (Case No. 1:17-cv-03681-PGG) against the Company in the United States District Court for the Southern District of New York. Plaintiff alleges that the Company entered into an agreement with Plaintiff (the “Agreement”), pursuant to which Plaintiff was to provide the Company with financial advisory services in connection with the Company’s initial public offering in the United States. Plaintiff alleges that the Company breached the Agreement and seeks money damages up to US$ 6 million. The Company believes that these claims are without merit and intends to vigorously defend its position.
NOTE 17 - SEGMENT REPORTING
ASC 280, “Segment Reporting”, establishes standards for reporting information about operating segments on a basis consistent with the Group’s internal organizational management structure as well as information about geographical areas, business segments and major customers in for details on the Group’s business segments.
The Company’s chief operating decision maker has been identified as the Chief Executive Officer who reviews the financial information of separate operating segments when making decisions about allocating resources and assessing performance of the Group. Based on management’s assessment, the Company has determined that it has three operating segments according to its major products and locations as follows:
| ● | Developing, manufacturing and distributing of specialized fabrics, textile products and other by-products derived from an indigenous Chinese plant called Apocynum Venetum, commonly known as “Bluish Dogbane” or known in Chinese as “Luobuma” (referred to herein as Luobuma): |
The operating companies of this segment, namely Tenet-Jove and Tenet Huatai, specialize in Luobuma growing, development and manufacturing of relevant products, as well as purchasing Luobuma raw materials processing.
This segment’s operations are focused in the north region of Mainland China, mostly carried out in Beijing, Tianjin and Xinjiang City.
F-27
| ● | Processing and distributing of traditional Chinese medicinal herbal products as well as other pharmaceutical products (“Herbal products”): |
The operating companies of this segment, namely AnKang Longevity Group and its subsidiaries, process more than 600 kinds of Chinese medicinal herbal products with an established domestic sales and distribution network.
Ankang Longevity Group is also engaged in the retail pharmacy business and the operating revenue, which is not material, is also included in this segment.
| ● | Planting, processing and distributing of green and organic agricultural produce as well as growing and cultivating of Chinese Yew trees (“Other agricultural products”): |
The operating companies of this segment, the Zhisheng Group, is engaged in the business of growing and distributing green and organic vegetables and fruits as well as providing logistics services for distributing agricultural products. This segment has been focusing its efforts on the growing and cultivating of Chinese yew trees (formally known as “taxus media”), a small evergreen tree whose branches can be used for the production of medications believed to be anti-cancer and the tree itself can be used as an ornamental indoor bonsai tree, which are known to have the effect of purifying air quality.
The operations of this segment are located in the East and North regions of Mainland China, mostly carried out in Shandong Province and in Beijing where the Zhisheng Group has newly developed over 100 acres of modern greenhouses for cultivating yew trees and other plants.
The following table presents summarized information by segment for the year ended June 30, 2018:
| For the year ended June 30, 2018 | ||||||||||||||||
| Luobuma | Herbal | Other agricultural | ||||||||||||||
| products | products | products | Total | |||||||||||||
| Segment revenue | $ | 10,884,901 | $ | 14,179,603 | $ | 18,833,114 | $ | 43,897,618 | ||||||||
| Cost of revenue and related business and sales tax | 4,889,282 | 10,761,778 | 13,459,266 | 29,110,326 | ||||||||||||
| Gross profit | 5,995,619 | 3,417,825 | 5,373,848 | 14,787,292 | ||||||||||||
| Gross profit % | 55.2 | % | 24.1 | % | 28.5 | % | 33.7 | % | ||||||||
The following table presents summarized information by segment for the year ended June 30, 2017:
| For the year ended June 30, 2017 | ||||||||||||||||
| Luobuma | Herbal | Other agricultural | ||||||||||||||
| products | products | products | Total | |||||||||||||
| Segment revenue | $ | 3,624,993 | $ | 13,245,236 | $ | 16,722,108 | $ | 33,592,337 | ||||||||
| Cost of revenue and related business and sales tax | 1,712,014 | 10,183,961 | 10,956,034 | 22,852,009 | ||||||||||||
| Gross profit | 1,912,979 | 3,061,275 | 5,766,074 | 10,740,328 | ||||||||||||
| Gross profit % | 52.8 | % | 23.1 | % | 34.5 | % | 32.0 | % | ||||||||
Total Assets as of
| June 30, 2018 | June 30, 2017 | |||||||
| Luobuma products | $ | 11,927,928 | $ | 6,983,551 | ||||
| Herbal products | 40,904,909 | 35,222,278 | ||||||
| Other agricultural products | 30,267,580 | 26,079,141 | ||||||
| $ | 83,100,417 | $ | 68,284,970 | |||||
NOTE 18 – SUBSEQUENT EVENTS
These consolidated financial statements were approved by management and available for issuance on October 15, 2018,
and the Company has evaluated subsequent events through this date. No subsequent events required adjustments to or disclosure
in these consolidated financial statements.
F-28
