Attached files
| file | filename |
|---|---|
| EX-99.1 - EX-99.1 - ABAXIS INC | d553493dex991.htm |
| 8-K - 8-K - ABAXIS INC | d553493d8k.htm |
Exhibit 99.2

|
INTRODUCING YOU TO ZOETIS
May 16, 2018

|
FORWARD-LOOKING STATEMENTS
Statements included in this communication which are not historical in nature or do not relate to current facts are intended to be, and are hereby identified as, forward-looking
statements for purposes of the safe harbor provided by Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. The words “may,” “will,” “anticipate,”
“could,” “should,” “would,” “believe,” “contemplate,” “expect,” “estimate,” “continue,” “plan,” “project” and “intend,” as well as other
similar words and expressions of the future, are intended to identify forward-looking statements. Zoetis Inc. (“Zoetis”) and Abaxis, Inc. (“Abaxis”) caution readers that forward-looking statements are subject to certain risks and
uncertainties that could cause actual results to differ materially from anticipated results. Such risks and uncertainties, include, among others, the following possibilities: the occurrence of any event, change or other circumstances that could give
rise to the right of one or both of the parties to terminate the definitive merger agreement between Zoetis and Abaxis; the outcome of any legal proceedings that may be instituted against Zoetis or Abaxis; the failure to obtain necessary regulatory
approvals (and the risk that such approvals may result in the imposition of conditions that could adversely affect the combined company or the expected benefits of the transaction) or Abaxis shareholder approval or to satisfy any of the other
conditions to the transaction on a timely basis or at all; the possibility that the anticipated benefits of the transaction are not realized when expected or at all, including as a result of the impact of, or problems arising from, the integration
of the two companies or as a result of the strength of the economy and competitive factors in the areas where Zoetis and Abaxis do business; the possibility that the transaction may be more expensive to complete than anticipated, including as a
result of unexpected factors or events; diversion of management’s attention from ongoing business operations and opportunities; potential adverse reactions or changes to business or employee relationships, including those resulting from the
announcement or completion of the transaction; the ability to complete the acquisition and integration of Abaxis successfully; and other factors that may affect future results of Zoetis and Abaxis. Additional factors that could cause results to
differ materially from those described above can be found in Zoetis’ Annual Report on Form 10-K for the year ended December 31, 2017, which is on file with the Securities and Exchange Commission (the
“SEC”) and in other documents Zoetis files with the SEC, and in Abaxis’s Annual Report on Form 10-K for the year ended March 31, 2017 and Quarterly Report on Form 10-Q for the quarter ended December 31, 2017, which are on file with the SEC and in other2 documents Abaxis files with the SEC.

|
IMPORTANT ADDITIONAL INFORMATION
In connection with the proposed transaction between Zoetis and Abaxis, Abaxis expects to file with the SEC a proxy statement of Abaxis, as well as other relevant documents
concerning the proposed transaction. This communication is not a substitute for the proxy statement or for any other document that Abaxis may file with the SEC and send to its shareholders in connection with the proposed transaction. The proposed
transaction will be submitted to Abaxis’ shareholders for their consideration. Before making any voting decision, Shareholders of Abaxis are urged to read the proxy statement regarding the transaction when it becomes available and any other
relevant documents filed with the SEC, as well as any amendments or supplements to those documents, because they will contain important information about the proposed transaction.
Shareholders of Abaxis will be able to obtain a free copy of the proxy statement, as well as other filings containing information about Zoetis and Abaxis, without charge, at the
SEC’s website (http://www.sec.gov). Copies of the proxy statement and the filings with the SEC that will be incorporated by reference therein can also be obtained, without charge, by directing a request to Abaxis, Inc., 3240 Whipple Road, Union
City, CA 94587, Attention: Corporate Secretary; telephone: (510) 675-6500, or from Abaxis’ website, www.abaxis.com.
PARTICIPANTS IN THE SOLICITATION
Abaxis, Zoetis, and certain of their
respective directors, executive officers and employees may be deemed to be participants in the solicitation of proxies in respect of the proposed transaction. Information regarding Abaxis’ directors and executive officers is available in
Abaxis’ definitive proxy statement, which was filed with the SEC on September 19, 2017, and certain of its Current Reports on Form 8-K. Information regarding Zoetis’ directors is available in
Zoetis’ definitive proxy statement, which was filed with the SEC on April 2, 2018, and information regarding Zoets’ executive officers is available in its Annual Report on Form 10-K for the year
ended December 31, 2017, which was filed with the SEC on February 15, 2018. Other information regarding the participants in the proxy solicitation and a description of their direct and indirect interests, by security holdings or otherwise,
will be contained in the proxy statement and other relevant materials to be filed with the SEC in connection with the proposed transaction. Free copies of this document may be obtained as described in the preceding paragraph.

|
Building on more than 60 years of experience, Zoetis discovers, develops, manufactures and markets veterinary vaccines and medicines, complemented by diagnostic products, genetic tests, biodevices and a range of services.

|
OUR VISION
OUR VISION GUIDES OUR ASPIRATIONS
Our products, services and people will be the most valued by
animal health customers around the world.
OUR MISSION
OUR MISSION GUIDES OUR
ACTIONS
We build on a six-decade history and singular focus on animal health to bring customers quality products, services
and a commitment to their business.

|
MANUFACTURING ANIMAL CORE
SITES $
25 8 SPECIES 5.3
APPROXIMATE
APPROXIMATE COLLEAGUES R&D NUMBER OF BILLION
300 PRODUCT LINES
ANNUAL REVENUE
1,000 WE PROVIDE
VACCINES MEDICINES DIAGNOSTICS
MAJOR PRODUCT GENETIC TESTS YEARS OF CATEGORIES BIODEVICES
5 SERVICES 60+ EXPERIENCE
MARKET PRESENCE IN APPROXIMATE WORLDWIDE COLLEAGUES OUR FOCUS
42%1 2,900
COMPANION
9,000 ANIMAL HEALTH 57%1 APPROXIMATE FIELD FORCE MEMBERS
100+ LIVESTOCK
COUNTRIES HEALTH
Note: Facts and figures shown are as of Dec. 31, 2017 | 1Excludes revenue associated with Client Supply Services, which represented 1% of total 2017 revenue.

|
ADDRESSING
CUSTOMER NEEDS
THE ZOETIS ADVANTAGE
• Global Leadership and Scale
• Diverse Portfolio
• Interconnected Capabilities:
• Direct Customer Relationships
• Innovation
• High Quality Manufacturing
• Singular Focus
|
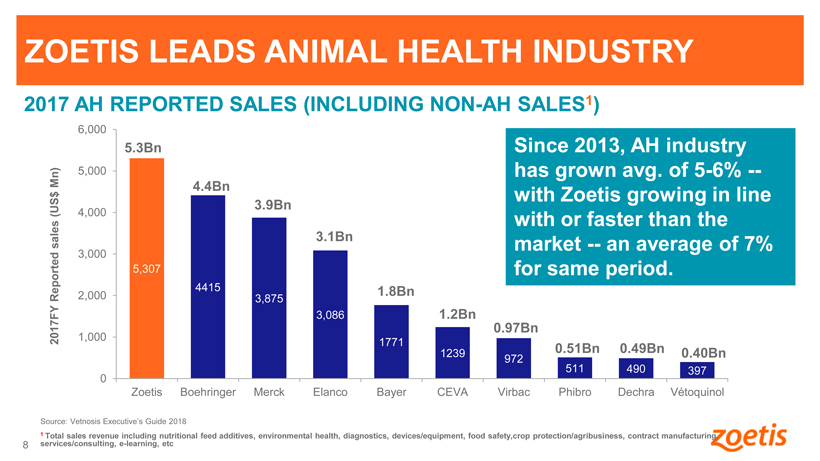
ZOETIS LEADS ANIMAL HEALTH INDUSTRY
2017 AH REPORTED SALES (INCLUDING NON-AH SALES1)
6,000
5.3Bn Since 2013, AH industry Mn) 5,000 has grown avg. of 5-6% —
4.4Bn with Zoetis growing in line
3.9Bn
(US$ 4,000
with or faster than the sales 3.1Bn market — an average of 7%
3,000
5,307 for same period.
4415 1.8Bn
Reported 2,000 3,875
3,086 1.2Bn
0.97Bn
2017FY 1,000
1771 0.51Bn 0.49Bn
1239 0.40Bn
972 511 490
397 0
8 Zoetis Boehringer Merck Elanco Bayer CEVA Virbac Phibro Dechra Vétoquinol
Source: Vetnosis Executive’s Guide 2018
1 Total sales revenue including
nutritional feed additives, environmental health, diagnostics, devices/equipment, food safety,crop protection/agribusiness, contract manufacturing, services/consulting, e-learning, etc
|
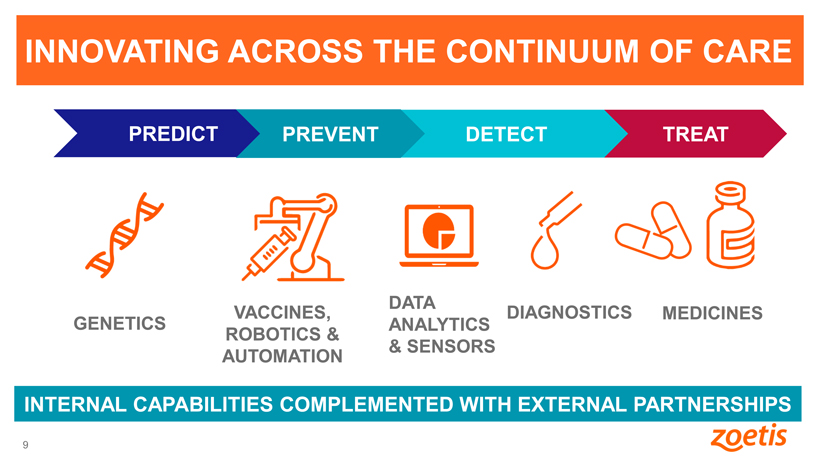
INNOVATING ACROSS THE CONTINUUM OF CARE
PREDICT PREVENT DETECT TREAT
DATA
VACCINES, DIAGNOSTICS MEDICINES GENETICS ANALYTICS
ROBOTICS &
& SENSORS
AUTOMATION
INTERNAL CAPABILITIES COMPLEMENTED WITH EXTERNAL PARTNERSHIPS

|
EXPERIENCED MANAGEMENT TEAM
It is our vision to have the products, services — and people — most valued by animal health customers around the world. We are proud to be led by some of the most
passionate, experienced experts in the industry.
JUAN RAMÓN ALAIX
Chief Executive Officer
HEIDI CHEN
Executive Vice President and General Counsel
GLENN DAVID
Executive Vice President and Chief Financial Officer
ANDREW FENTON
Executive Vice President and Chief Information Officer
CATHY KNUPP
Executive Vice President and President, Research and Development
ROXANNE LAGANO
Executive Vice President and Chief Human Resources Officer
CLINT LEWIS
Executive Vice President and Group President, International Operations, Commercial Development, Genetics and PHARMAQ
KRISTIN PECK
Executive Vice President and Group President, U.S.
Operations, Business Development and Strategy
ROMAN TRAWICKI
Executive Vice President and President, Global Manufacturing and Supply

|
OUR CULTURE
OUR CORE BELIEFS FORM THE FOUNDATION OF OUR CULTURE
AWARD-WINNING WORKPLACE

|
ACCELERATING GROWTH TOGETHER
|
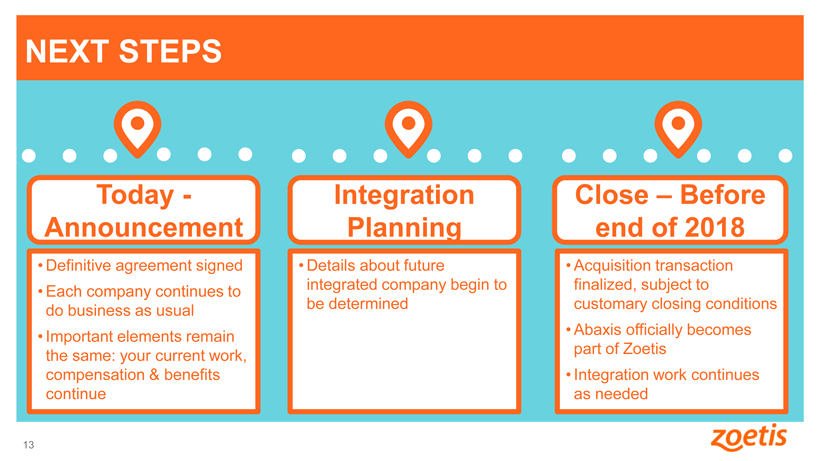
NEXT STEPS
Today -Announcement
• Definitive agreement signed
• Each company continues to do business as usual
• Important elements remain the
same: your current work, compensation & benefits continue
Integration Planning
• Details about future integrated company begin to be determined
Close – Before end
of 2018
• Acquisition transaction finalized, subject to customary closing conditions
• Abaxis officially becomes part of Zoetis
• Integration work continues as needed

|
ON YOUR MIND
Future Compensation,
Business Benefits, Employment Structure & Policies With Zoetis
Leadership

|
QUESTIONS?

|
www.zoetis.com
zoetis.com
