Attached files
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
________________________________________________________
FORM 10-K
x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the Fiscal Year Ended December 31, 2016 | |
or | |
o | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to | |
Commission file number 000-19319
____________________________________________
Vertex Pharmaceuticals Incorporated
(Exact name of registrant as specified in its charter)
Massachusetts | 04-3039129 |
(State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) |
50 Northern Avenue, Boston, Massachusetts | 02210 |
(Address of principal executive offices) | (Zip Code) |
Registrant’s telephone number, including area code (617) 341-6100
____________________________________________
Securities registered pursuant to Section 12(b) of the Exchange Act:
Title of Each Class | Name of Each Exchange on Which Registered | |||
Common Stock, $0.01 Par Value Per Share | The NASDAQ Global Select Market | |||
Securities registered pursuant to Section 12(g) of the Exchange Act: None
______________________________________________
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes x No o
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Exchange Act. Yes o No x
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No o
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes x No o
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§229.405 of this chapter) is not contained herein, and will not be contained, to the best of the registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10 K. x
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer or a smaller reporting company. See definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act. (Check one):
Large accelerated filer x | Accelerated filer o | Non-accelerated filer o | Smaller reporting company o |
(Do not check if a smaller reporting company) | |||
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes o No x
The aggregate market value of the registrant’s common stock held by non-affiliates of the registrant (without admitting that any person whose shares are not included in such calculation is an affiliate) based on the last reported sale price of the common stock on June 30, 2016 (the last trading day of the registrant’s second fiscal quarter of 2016) was $21.1 billion. As of February 10, 2017, the registrant had 248,438,127 shares of common stock outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the definitive Proxy Statement for the 2017 Annual Meeting of Shareholders to be held on June 8, 2017 are incorporated by reference into Part III of this Annual Report on Form 10-K.
VERTEX PHARMACEUTICALS INCORPORATED
ANNUAL REPORT ON FORM 10-K
TABLE OF CONTENTS
“We,” “us,” “Vertex” and the “Company” as used in this Annual Report on Form 10-K refer to Vertex Pharmaceuticals Incorporated, a Massachusetts corporation, and its subsidiaries.
“Vertex,” “KALYDECO®” and “ORKAMBI®” are registered trademarks of Vertex. Other brands, names and trademarks contained in this Annual Report on Form 10-K are the property of their respective owners.
PART I
ITEM 1. | BUSINESS |
OVERVIEW
We are in the business of discovering, developing, manufacturing and commercializing medicines for serious diseases. We use precision medicine approaches with the goal of creating transformative drugs for patients in specialty markets. Our business is focused on developing and commercializing therapies for the treatment of cystic fibrosis, or CF, and advancing our research and development programs in other indications, while maintaining our financial strength.
Cystic Fibrosis
Our two marketed medicines are ORKAMBI and KALYDECO, which together are approved to treat approximately 40% of the 75,000 CF patients in North America, Europe and Australia. ORKAMBI (lumacaftor in combination with ivacaftor) is approved as a treatment for approximately 25,000 patients who have two copies (homozygous) of the F508del mutation in their cystic fibrosis transmembrane conductance regulator, or CFTR, gene. KALYDECO (ivacaftor) is approved for the treatment of approximately 4,000 CF patients who have the G551D mutation or other specified mutations in their CFTR gene. Our goal is to develop treatment regimens that will provide benefits to as many patients with CF as possible and will enhance the benefits that currently are being provided to patients taking our medicines.
CF Development Programs
We have multiple development programs in the field of CF, including:
• | Tezacaftor (VX-661) is a corrector compound that we are evaluating in a Phase 3 development program in combination with ivacaftor in multiple CF patient populations who have at least one copy of the F508del mutation in their CFTR gene. We expect data from this Phase 3 development program in the first half of 2017. If supported by data from the Phase 3 development program, we plan to submit a New Drug Application, or NDA, to the United States Food and Drug Administration, or FDA for tezacaftor in combination with ivacaftor in the second half of 2017. |
• | VX-152, VX-440, VX-659 and VX-445 are next-generation CFTR corrector compounds that we are evaluating as part of combination treatment regimens. We have initiated Phase 2 clinical trials of VX-152 and VX-440 and expect data from these clinical trials in the second half of 2017. We have initiated Phase 1 clinical trials of VX-659 and VX-445. |
• | VX-371 is an investigational epithelial sodium channel, or ENaC, inhibitor that is being evaluated in a Phase 2 development program and which we exclusively licensed from Parion Sciences, Inc., or Parion, in 2015. |
Research and Development Programs
We are engaged in a number of other research and mid- and early-stage development programs, including programs in the areas of pain and neurology. We also have entered into third-party collaborations pursuant to which we are engaged in the discovery and development of nucleic acid-based therapies for a variety of diseases, including CF. We plan to continue investing in our research programs and fostering scientific innovation in order to identify and develop transformative medicines. Our current research programs include programs targeting cystic fibrosis, adrenoleukodystrophy, alpha-1 antritrypsin deficiency, sickle cell disease and polycystic kidney disease. We believe that pursuing research in diverse areas allows us to balance the risks inherent in drug development and may provide drug candidates that will form our pipeline in future years.
CYSTIC FIBROSIS
Background
CF is a rare, life-threatening genetic disease affecting approximately 75,000 people in North America, Europe and Australia. CF is caused by a defective or missing CFTR protein resulting from mutations in the CFTR gene. To develop CF, children must inherit two defective CFTR genes, which are referred to as alleles - one from each parent. There are more than 1,900 known mutations in the CFTR gene, some of which result in CF. The vast majority of patients with CF carry at least one of the two of the most prevalent mutations, the F508del mutation and the G551D mutation.
1
The F508del mutation results in a defect in the CFTR protein in which the CFTR protein does not reach the surface of cells in sufficient quantities. The G551D mutation results in a defect in the CFTR protein in which the defective CFTR protein reaches the surface of a cell but does not efficiently transport chloride ions across the cell membrane. The absence of working CFTR proteins results in poor flow of salt and water into and out of cells in a number of organs, including the lungs. As a result, thick, sticky mucus builds up and blocks the passages in many organs, leading to a variety of symptoms. In particular, mucus builds up and clogs the airways in the lungs, causing chronic lung infections and progressive lung damage. Ivacaftor, a CFTR potentiator, increases the open probability of the CFTR protein channels on the cell surface increasing the flow of salt and water into and out of the cell. CFTR correctors, such as lumacaftor, tezacaftor, VX-152, VX-440, VX-659 and VX-445 help CFTR proteins reach the cell surface. We believe that ENaC inhibitors, such as VX-371, may help maintain mucus hydration and accelerate pulmonary mucus clearance.
We use the brand name for our products when we refer to the product that has been approved and with respect to the indications on the approved label. Otherwise, including in discussions of our CF development programs, we refer to our compounds by their scientific (or generic) name.
ORKAMBI (lumacaftor in combination with ivacaftor)
ORKAMBI is an orally-administered combination therapy comprised of lumacaftor, a CFTR corrector, and ivacaftor, a CFTR potentiator, that is approved in the United States, European Union, Australia and Canada for the treatment of patients with CF twelve years of age and older who are homozygous for the F508del mutation in their CFTR gene. In the United States, ORKAMBI is also approved for the treatment of patients with CF six to eleven years of age who are homozygous for the F508del mutation in their CFTR gene.
In the fourth quarter of 2016, we announced the results of a Phase 3 clinical trial designed to support European approval of lumacaftor in combination with ivacaftor in patients with CF six to eleven years of age who are homozygous for the F508del mutation in their CFTR gene. The clinical trial met its primary endpoint of absolute change in lung clearance index, or LCI2.5, through 24 weeks of treatment, demonstrating a statistically significant improvement in LCI2.5 among patients treated with ORKAMBI compared to placebo. Based on these results, we plan to submit a Marketing Authorization Application, or MAA, line extension to the European Medicines Agency, or EMA, seeking approval of lumacaftor in combination with ivacaftor in this patient population in the European Union in the first half of 2017.
We are conducting a Phase 3 clinical trial for lumacaftor in combination with ivacaftor in patients with CF two to five years of age who are homozygous for the F508del mutation in their CFTR gene. We expect enrollment in the first part of this clinical trial to be complete in mid-2017. The first part of the two-part clinical trial is evaluating safety and pharmacokinetics to inform dose selection for the second part of the clinical trial. The primary endpoint of the second part of the clinical trial is safety and tolerability, with multiple efficacy measurements as secondary endpoints.
KALYDECO (ivacaftor)
KALYDECO (ivacaftor) is an orally-administered CFTR potentiator that is approved in the United States, European Union, Australia and Canada for the treatment of certain patients with CF who have specific mutations in their CFTR gene. In the United States, KALYDECO is approved for the treatment of patients with CF two years of age and older who have one of the following mutations in their CFTR gene: G551D, G178R, S549N, S549R, G551S, G1244E, S1251N, S1255P, G1349D and R117H. In the European Union, KALYDECO is approved for the treatment of patients with CF (i) two years of age and older who have one of the following mutations in their CFTR gene: G551D, G178R, S549N, S549R, G551S, G1244E, S1251N, S1255P and G1349D and (ii) eighteen years of age and older who have the R117H mutation in their CFTR gene.
We have initiated a Phase 3 clinical trial for ivacaftor in patients with CF less than two years of age to evaluate the effect of ivacaftor on markers of CF disease in young children. The clinical trial utilizes a weight-based dose of ivacaftor granules that can be mixed in soft foods or liquids. The clinical trial is enrolling patients with one of the ten CFTR gene mutations for which KALYDECO is currently approved.
Tezacaftor (VX-661) in Combination with Ivacaftor
Tezacaftor is an orally-administered CFTR corrector drug candidate that we are evaluating in a Phase 3 development program in combination with ivacaftor in multiple CF patient populations who have at least one copy of the F508del mutation in their CFTR gene. We expect data from this Phase 3 development program in the first half of 2017. If supported
2
by data from the Phase 3 development program, we plan to submit a NDA to the FDA for tezacaftor in combination with ivacaftor in the second half of 2017.
The ongoing clinical trials in this Phase 3 development program are:
• | Two copies of the F508del in their CFTR gene: We have completed enrollment in this clinical trial and expect data from this clinical trial to be available in the first half of 2017; |
• | One copy of the F508del mutation in their CFTR gene and a second mutation in their CFTR gene that results in residual CFTR function: We have completed enrollment in this clinical trial and expect data from this clinical trial to be available in the first half of 2017; and |
• | One copy of the F508del mutation in their CFTR gene and a second mutation in their CFTR gene that results in a gating defect in the CFTR protein: We plan to complete enrollment in this clinical trial in the first half of 2017. Data from this clinical trial are not expected to be included in the initial regulatory applications for tezacaftor in combination with ivacaftor. |
In the third quarter of 2016, we completed an interim futility analysis of efficacy data from the first part of a fourth clinical trial in patients with one copy of the F508del mutation in their CFTR gene and a second mutation that results in minimal CFTR function. The analysis showed that the combination of tezacaftor and ivacaftor did not result in a pre-specified improvement in lung function in this patient group. The independent Data Safety Monitoring Board, or DSMB, recommended that we stop the clinical trial and not initiate enrollment in the second part of this clinical trial. There were no safety concerns noted in the DSMB's review of the data. We subsequently reviewed the data and concurred with the DSMB’s assessment. We have closed this clinical trial based on the recommendation of the DSMB. The planned NDA for tezacaftor in combination with ivacaftor will include safety data from this clinical trial.
In addition to evaluating the efficacy of the combination regimen, these Phase 3 clinical trials will provide safety data on the combination of tezacaftor and ivacaftor to support the planned development of a triple combination regimen that includes a next-generation corrector in combination with tezacaftor and ivacaftor.
In addition to the Phase 3 development program described above, we also are conducting a Phase 3 open-label clinical trial evaluating the safety and tolerability of tezacaftor in combination with ivacaftor in patients with CF six to eleven years of age who have either (i) two copies of the F508del mutation in their CFTR gene or (ii) one copy of the F508del mutation in their CFTR gene and a second mutation in their CFTR gene that has clinically demonstrated to be responsive to ivacaftor, including gating and residual function mutations.
Next-generation CFTR Corrector Compounds
We are developing next-generation CFTR corrector compounds that we plan to evaluate as part of triple combination treatment regimens. We have initiated Phase 2 clinical trials of:
• | VX-440 to evaluate the safety and efficacy of four-week dosing of VX-440 in combination with tezacaftor and ivacaftor in approximately 40 patients with CF who have one copy of the F508del mutation in their CFTR gene and a second mutation that results in minimal CFTR function and approximately 25 patients with CF who have two copies of the F508del mutation in their CFTR gene; and |
• | VX-152 to evaluate the safety and efficacy of two-week dosing of VX-152 in combination with tezacaftor and ivacaftor in approximately 35 patients with CF who have one copy of the F508del mutation in their CFTR gene and a second mutation that results in minimal CFTR function and approximately 25 patients with CF who have two copies of the F508del mutation in their CFTR gene. |
Data from these clinical trials are expected in the second half of 2017 and if successful, are intended to support the potential initiation of Phase 3 development of VX-440 and a longer-duration Phase 2b or registrational program for VX-152.
We have initiated a Phase 1 clinical trial of VX-659, an additional next-generation corrector, to evaluate single ascending doses, multiple ascending doses and triple combination dosing in healthy volunteers and will include an arm to evaluate triple combination dosing in patients with CF who have one copy of the F508del mutation in their CFTR gene and a second mutation that results in minimal CFTR function. This clinical trial is enrolling healthy volunteers, and we expect to begin
3
enrolling CF patients in this clinical trial in the first half of 2017. Dosing of a fourth next generation corrector, VX-445, began in the first quarter of 2017. Pending data from these Phase 1 clinical trials, we plan to begin Phase 2 development for one or both of these additional next-generation correctors in the second half of 2017.
ENaC Inhibition
In 2015, we entered into a collaboration with Parion to develop investigational ENaC inhibitors, including VX-371, for the potential treatment of CF and other pulmonary diseases. Preclinical evaluation in human bronchial epithelial, or HBE, cells from patients with CF who have two copies of the F508del mutation in their CFTR gene showed that the addition of VX-371 to lumacaftor in combination with ivacaftor resulted in an additional increase in both airway surface liquid and cilia beat frequency compared to baseline and to the use of VX-371 or lumacaftor in combination with ivacaftor alone. Improvements in airway surface liquid height and cilia beat frequency are believed to be measures of increased hydration of the cell surface.
In the second quarter of 2016, Parion completed a Phase 2 clinical trial in 142 patients with CF with no restriction on the mutations in their CFTR gene. The primary endpoint of the clinical trial was safety as compared to patients on placebo. Secondary endpoints evaluated the effect on mean absolute forced expiratory volume in one second, or FEV1 and patient-reported respiratory symptoms as reported in the CF questionnaire-revised, or CFQ-R. The clinical trial met its primary safety endpoint and data from the clinical trial showed that VX-371 was generally well-tolerated. There were no statistically significant changes in FEV1 or CFQ-R for patients who received VX-371.
In the first quarter of 2016, we initiated a Phase 2a clinical trial evaluating VX-371 in combination with ORKAMBI, both with and without the addition of hypertonic saline, in patients with CF twelve years of age and older who have two copies of the F508del mutation in their CFTR gene. The primary endpoints of this clinical trial are safety and mean absolute change from baseline in FEV1 at day 28 as compared to patients on placebo. We expect data from this clinical trial to be available in the second half of 2017.
RESEARCH AND DEVELOPMENT PROGRAMS
Our approach to project selection and drug design aims to enhance our ability to discover and develop drug candidates by combining transformative insights into the causes of serious diseases with innovative approaches to therapeutics. Historically, our approach to drug discovery has focused on the research and development of small molecule drugs, which has been validated through our success in moving novel small molecule drug candidates into clinical trials and obtaining marketing approvals for ORKAMBI, KALYDECO and INCIVEK (telapravir). Recently, we have expanded our research capabilities to include additional innovative therapeutic approaches with a focus on nucleic acid-based therapies. We believe that attempting to identify multiple approaches to the treatment of diseases enhances our potential to develop treatment options that, either as monotherapies or combination therapies, are transformational in nature.
We focus our research activities on developing products that would be prescribed by specialist physicians for the treatment of rare or life-threatening diseases. We begin by applying our knowledge of human genetics and human biology and focus on validated targets that have shown a causal relationship with respect to serious diseases. We generate biological assays to query the underlying biology of disease and utilize clinical biomarkers as tools to predict clinical response. We leverage our expertise in assay automation, medicinal and process chemistry, modeling and informatics, drug metabolism and pharmacokinetics, toxicology, material sciences and formulation to develop, select and advance drug candidates that have the potential to offer transformative benefits and have an efficient path to approval. Our current research programs include programs targeting cystic fibrosis, pain, adrenoleukodystrophy, alpha-1 antritrypsin deficiency, sickle cell disease and polycystic kidney disease.
To augment our internal research programs, we seek to collaborate with biopharmaceutical and technology companies, leading academic research institutions, government laboratories, foundations and other organizations as needed to advance research in our areas of therapeutic interest as well as to access technologies needed to execute on our strategy. We have established such relationships with organizations around the world and intend to extend and leverage that experience to further our research efforts to discover transformational medicines for serious diseases.
In addition to our continuing research efforts in CF, our research and mid- and early-stage development programs currently include VX-371 for the treatment of primary ciliary dyskinesia, VX-150 for the treatment of pain and VX-210 for the treatment of acute cervical spinal cord injury. We are also collaborating with CRISPR Therapeutics AG, or CRISPR, on
4
the discovery and development of potential new treatments aimed at the underlying genetic causes of human diseases using CRISPR-Cas9 gene editing technology and Moderna Therapeutics, Inc., or Moderna, to identify and develop messenger ribonucleic acid, or mRNA, therapeutics for the treatment of CF.
COMMERCIAL ORGANIZATION
Our commercial organization focuses on supporting sales of ORKAMBI and KALYDECO in the markets where such products are approved. Our sales and marketing organizations are responsible for promoting products to health care providers and obtaining reimbursement for products from third-party payors, including governmental organizations in the United States and non-U.S. markets.
Our U.S. field-based CF commercial team is comprised of a small number of individuals whom we believe will be sufficient to support future needs. We focus our CF marketing efforts in the United States on a relatively small number of physicians and health care professionals who write most of the prescriptions for CF medicines. Many of these physicians and health care professionals are located at a limited number of accredited centers in the United States focused on the treatment of CF. In international markets, we have a small sales force that promotes KALYDECO and ORKAMBI in jurisdictions where these products are approved.
We market our products through personal interactions with individual physicians, advertising, sending direct mail, public relations activities and other activities. In addition, our government affairs and public policy group advocates for policies that promote life sciences innovation and increase awareness of the diseases on which we are focusing, with state and federal legislatures, government agencies, public health officials and other policy-makers. We also have established programs in the United States that provide our products to qualified uninsured or underinsured patients at no charge or at a reduced charge, based on specific eligibility criteria.
COLLABORATIONS
We have entered into collaborations with pharmaceutical and other companies and organizations that provide us financial and other resources, including capabilities in research, development, manufacturing and sales and marketing, and licenses to intellectual property. These collaborations have provided us with drug candidates and/or important financial and non-financial resources that have contributed to our products and a number of the drug candidates in our current development pipeline. We may seek to license or acquire drugs, drug candidates and other technologies that have the potential to add to our pipeline or to provide us with new commercial opportunities. In particular, we are focusing on drug candidates for the treatment of patients with CF and other third-party drug candidates that could be developed for specialty markets. Furthermore, we may seek collaborators to support, develop and/or commercialize some of our current drug candidates and/or additional drug candidates that may emerge from our research activities.
Cystic Fibrosis Foundation Therapeutics Incorporated
We began working with the Cystic Fibrosis Therapeutics Incorporated, or CFFT, in 1998. We entered into a collaboration agreement with CFFT in 2004 and have amended it several times to support research and development activities. Pursuant to the collaboration agreement, as amended, we have agreed to pay tiered royalties ranging from single digits to sub-teens on any approved drugs first synthesized and/or tested during a research term on or before February 28, 2014, including KALYDECO, ORKAMBI, lumacaftor and tezacaftor and royalties ranging from low single digits to mid-single digits on potential sales of certain compounds first synthesized and/or tested between March 1, 2014 and August 31, 2016. For combination products, such as ORKAMBI, sales are allocated equally to each of the active pharmaceutical ingredients in the combination product. Pursuant to an amendment entered into in the fourth quarter of 2016, the CFFT paid us an upfront program award of $75.0 million and agreed to provide development funding to us of up to $6.0 million annually.
For ivacaftor, lumacaftor and tezacaftor, we will have royalty obligations to CFFT until the expiration of patents covering that compound. We have patents in the United States and European Union covering the composition-of-matter of ivacaftor that expire in 2027 and 2025, respectively, subject to potential patent life extensions. We have patents in the United States and European Union covering the composition-of-matter of lumacaftor that expire in 2030 and 2026, respectively, subject to potential patent life extensions. We have patents in the United States and European Union covering the composition-of-matter of tezacaftor that expire in 2027 and 2028, respectively, subject to potential patent life extensions.
5
Parion Sciences, Inc.
In 2015, we entered into a strategic collaboration and license agreement with Parion pursuant to which we are collaborating with Parion to develop ENaC inhibitors, including VX-371 and VX-551, for the potential treatment of CF and other pulmonary diseases.
We are leading development activities for VX-371 and are responsible for all costs, subject to certain exceptions, related to its development and commercialization. We also will lead development activities for VX-551, which is in pre-clinical development. Under the terms of the agreement, we received worldwide development and commercial rights to VX-371 and VX-551 for the potential treatment of CF and all other pulmonary diseases and have the option to select additional compounds discovered in Parion’s research program. Parion received an $80.0 million up-front payment and has the potential to receive up to an additional (i) $490.0 million in development and regulatory milestone payments for development of ENaC inhibitors in CF, including $360.0 million related to global filing and approval milestones, (ii) $370.0 million in development and regulatory milestones for VX-371 and VX-551 in non-CF pulmonary indications and (iii) $230.0 million in development and regulatory milestones if we elect to develop an additional ENaC inhibitor from Parion’s research program. Parion will receive tiered royalties on potential sales of licensed products that range from the low double digits to mid-teens as a percentage of sales. In the second quarter of 2016, we paid Parion a $5.0 million development milestone related to VX-371.
We may terminate the agreement upon 90 days’ notice to Parion prior to any licensed product receiving marketing approval or upon 180 days’ notice after a licensed product has received marketing approval. Parion may terminate the agreement upon 30 days’ notice if Vertex experiences a change of control prior to the initiation of the first Phase 3 clinical trial for a licensed product, subject to our right to receive specified royalties on any subsequent commercialization of licensed products. The agreement also may be terminated by either party for a material breach by the other, subject to notice and cure provisions. Unless earlier terminated, the agreement will continue in effect until the expiration of our royalty obligations, which expire on a country-by-country basis on the later of (i) the date the last-to-expire patent covering a licensed product expires or (ii) ten years after the first commercial sale in the country.
CRISPR Therapeutics AG
In 2015, we entered into a strategic collaboration, option and license agreement with CRISPR to collaborate on the discovery and development of potential new treatments aimed at the underlying genetic causes of human diseases using CRISPR-Cas9 gene editing technology. We have the exclusive right to license up to six CRISPR-Cas9-based targets. In connection with the CRISPR agreement, we paid CRISPR an upfront payment of $75.0 million and made a $30.0 million investment in CRISPR pursuant to a convertible loan agreement that converted into preferred stock in the first quarter of 2016. In the second quarter of 2016, we made an additional preferred stock investment in CRISPR of approximately $3.1 million. In connection with CRISPR's initial public offering in October 2016, we made an additional $10.0 million common share investment in CRISPR and our preferred stock investment in CRISPR converted into common shares.
We fund all of the discovery activities conducted pursuant to the agreement. For potential hemoglobinapathy treatments, including treatments for sickle cell disease, we share equally with CRISPR all research and development costs and worldwide revenues. For other targets that we elect to license, we would lead all development and global commercialization activities. For each of up to six targets that we elect to license, other than hemoglobinapathy targets, CRISPR has the potential to receive up to $420.0 million in development, regulatory and commercial milestones and royalties on net sales.
We may terminate the agreement upon 90 days’ notice to CRISPR prior to any product receiving marketing approval or upon 270 days’ notice after a product has received marketing approval. The agreement also may be terminated by either party for a material breach by the other, subject to notice and cure provisions. Unless earlier terminated, the agreement will continue in effect until the expiration of our payment obligations under the agreement.
Other Collaborations
Moderna Therapeutics, Inc.
In July 2016, we entered into a strategic collaboration and licensing agreement with Moderna pursuant to which the parties are seeking to identify and develop messenger ribonucleic acid, or mRNA, therapeutics for the treatment of CF. In connection with the Moderna agreement, we made an upfront payment to Moderna of $20.0 million and a $20.0 million investment in Moderna pursuant to a convertible promissory note that converted into preferred stock in August 2016.
6
Moderna has the potential to receive future development and regulatory milestones of up to $275.0 million, including $220.0 million in approval and reimbursement milestones, as well as tiered royalty payments on future sales.
Under the terms of the agreement, Moderna is leading discovery efforts and we are leading all preclinical, development and commercialization activities associated with the advancement of mRNA therapeutics that result from this collaboration and we will fund all expenses related to the collaboration.
We may terminate the agreement by providing advanced notice to Moderna, with the required length of notice dependent on whether any product developed under the agreement has received marketing approval. The agreement also may be terminated by either party for a material breach by the other, subject to notice and cure provisions. Unless earlier terminated, the agreement will continue in effect until the expiration of our payment obligations under the agreement.
BioAxone Biosciences, Inc.
In 2014, we entered into a license and collaboration agreement with BioAxone. Pursuant to this agreement, we are collaborating with BioAxone on the research, development and commercialization of VX-210 (formerly referred to as Cethrin), a Rho inhibitor controlled by BioAxone, for the treatment of patients who have spinal cord injuries.
We paid BioAxone initial payments of $10.0 million and BioAxone has the potential to receive up to $90.0 million in milestones and fees, including development and regulatory milestone payments and a license continuation fee. In addition, BioAxone would receive royalties and commercial milestones based on future net product sales, if any. We hold an option to purchase BioAxone at a predetermined price. The option expires on the earliest of (a) the day the FDA accepts a Biologics License Application submission for VX-210, (b) the day we elect to continue the license instead of exercising the option to purchase BioAxone and (c) March 15, 2018, subject to our option to extend this date by one year. We may terminate our agreement with BioAxone upon 90 days’ notice or immediately if we determine that a licensed product is unsafe for administration to humans. The agreement also may be terminated by either party for a material breach by the other or by BioAxone for our inactivity with respect to VX-210, in each case subject to notice and cure provisions. Unless earlier terminated, the agreement will continue until the expiration of our royalty obligations.
Outlicense Arrangements
We have entered into various agreements pursuant to which we have outlicensed rights to certain drug candidates to third-party collaborators. Pursuant to these outlicense arrangements, our collaborators become responsible for all costs related to the continued development of such drug candidates and obtain development and commercialization rights to these drug candidates. Depending on the terms of the arrangements, our collaborators may be required to make upfront payments, milestone payments upon the achievement of certain product research and development objectives and/or pay royalties on future sales, if any, of commercial products resulting from the collaboration.
Merck KGaA
On January 10, 2017, we entered into a Strategic Collaboration and License Agreement with Merck KGaA, Darmstadt, Germany, or Merck KGaA. Pursuant to the agreement, we granted Merck KGaA an exclusive worldwide license to research, develop and commercialize four oncology research and development programs. Under the agreement, we granted Merck KGaA exclusive, worldwide rights to our two clinical-stage programs targeting DNA damage repair: our ataxia telangiectasia and Rad3-related protein inhibitor, or ATR program, including VX-970 and VX-803, and our DNA-dependent protein kinase inhibitor, or DNA-PK program, including VX-984. In addition, we granted Merck KGaA exclusive, worldwide rights to two pre-clinical programs.
Under the agreement, we will receive an up-front payment of $230.0 million. In addition, we will receive tiered royalties on potential sales of licensed products, calculated as a percentage of net sales, that range from (i) mid-single digits to mid-twenties for clinical-stage programs and (ii) mid-single digits to high single digits for the pre-clinical research programs. Merck KGaA will assume full responsibility for development and commercialization costs for all programs. The licenses granted pursuant to the agreement and the up-front payment are subject to the expiration of the waiting period under the Hart-Scott-Rodino Antitrust Improvements Act.
Merck KGaA may terminate the agreement or any individual program by providing 90 days’ notice, or, in the case of termination of a program with a product that has received marketing approval, 180 days’ notice. The agreement may also be
7
terminated by either party for a material breach by the other party, subject to notice and cure provisions. Unless earlier terminated, the agreement will continue in effect until the date on which the royalty term and all payment obligations with respect to all products in all countries have expired.
Janssen Pharmaceuticals, Inc.
In 2014, we entered into an agreement with Janssen Pharmaceuticals, Inc., or Janssen Inc. Pursuant to this agreement, Janssen Inc. has an exclusive worldwide license to develop and commercialize certain drug candidates for the treatment of influenza, including VX-787. We received non-refundable payments of $35.0 million from Janssen Inc. in 2014 and have the potential to receive development, regulatory and commercial milestone payments as well as royalties on future product sales, if any. Janssen Inc. is responsible for costs related to the development and commercialization of the compounds. Janssen Inc. may terminate the agreement, subject to certain exceptions, upon six months’ notice.
INTELLECTUAL PROPERTY
We actively seek protection for our products and proprietary information by means of U.S. and foreign patents, trademarks and copyrights, as appropriate. In addition, we rely upon trade secret protection and contractual arrangements to protect certain of our proprietary information and products. We have patents and pending patent applications that relate to potential drug targets, compounds we are developing to modulate those targets, methods of making or using those compounds and proprietary elements of our drug discovery platform.
Much of our technology and many of our processes depend upon the knowledge, experience and skills of key scientific and technical personnel. To protect our rights to our proprietary know-how and technology, we require all employees, as well as our consultants and advisors when feasible, to enter into confidentiality agreements that require disclosure and assignment to us of ideas, developments, discoveries and inventions made by these employees, consultants and advisors in the course of their service to us.
While we have numerous issued patents and pending patent applications in our patent portfolio, we believe that the patents and patent applications in the United States and the European Union that are the most important to our business are those that claim the composition-of-matter of our drugs and drug candidates that have progressed at least into Phase 3 clinical trials. The following table sets forth the status of such primary patents and patent applications in the United States and the European Union covering the composition-of-matter of these drugs and drug candidates:
Drug/Drug Candidate | Status of United States Patent (Anticipated Expiration, Subject to Potential Extensions) | Status of European Union Patent (Anticipated Expiration, Subject to Potential Extensions) |
Ivacaftor | Granted (2027) | Granted (2025) |
Lumacaftor | Granted (2030) | Granted (2026) |
Tezacaftor | Granted (2027) | Granted (2028) |
We hold issued patents and pending patent applications in the United States, and in foreign countries we deem appropriate, claiming intellectual property developed as part of our research and development programs. In addition to the composition-of-matter patents and patent applications listed above, we hold or have exclusive licenses to the following intellectual property:
• | U.S. and foreign patent applications covering CF potentiators, correctors and ENaC inhibitors, including ivacaftor, lumacaftor, tezacaftor, VX-371, VX-152, VX-440, VX-659 and VX-445 and many other related compounds, and the use of those potentiators, correctors and ENaC inhibitors to treat CF. |
• | U.S. and foreign patents and patent applications covering VX-150 and VX-241 and the use of VX-150 and VX-241 to treat pain indications. |
• | U.S. and foreign patents and patent applications covering VX-210 and the use of VX-210 to treat neurology indications. |
8
• | U.S. and foreign patents and patent applications covering the manufacture, pharmaceutical compositions, related solid forms, formulations, dosing regimens and methods of use of these compounds, including ivacaftor and lumacaftor. |
We cannot be certain, however, that issued patents will be enforceable or provide adequate protection or that pending patent applications will result in issued patents.
From time to time we enter into non-exclusive license agreements for proprietary third-party technology used in connection with our research activities. These license agreements typically provide for the payment by us of a license fee, but may also include terms providing for milestone payments or royalties for the development and/or commercialization of our drug products arising from the related research.
Ivacaftor was granted orphan drug status in the United States and the European Union. We have a U.S. patent that covers the composition-of-matter of ivacaftor that we expect will provide intellectual property protection in the United States through its expiration date in 2027. We have a European patent that covers the composition-of-matter of ivacaftor that we expect will provide intellectual property protection in the European Union through its expiration date in 2025, subject to potential extension. We are entitled to orphan drug exclusivity for ivacaftor in the United States and the European Union, which means that the FDA may not approve another application to market ivacaftor for the same indication for a period of seven years following approval, and the EMA cannot accept an MAA for a drug similar to ivacaftor for a period of ten years following approval. As a result of the orphan drug exclusivity, even if a competitor successfully challenges the ivacaftor patents, it could not obtain approval from the FDA to market ivacaftor in the United States for the treatment of patients who have one of the mutations to the CFTR gene for which KALYDECO is currently approved until 2019, or submit an MAA in the European Union for the treatment of patients who have one of the mutations to the CFTR gene for which KALYDECO is currently approved until 2022, except in very limited circumstances.
Lumacaftor was granted orphan drug designation in the United States and the European Union and the fixed dose combination of lumacaftor and ivacaftor was granted orphan drug status in the United States. We have patents in the United States and European Union that cover the composition of matter of lumacaftor that we expect will provide intellectual property protection in these jurisdictions through their expiration dates in 2030 and 2026, respectively, subject to potential extension.
Tezacaftor was granted orphan drug designation in the United States and the European Union. We have patents in the United States and European Union that cover the composition of matter of tezacaftor that we expect will provide intellectual property protection in these jurisdictions through their expiration dates in 2027 and 2028, respectively, subject to potential extension.
MANUFACTURING
Manufacturing Approach and Philosophy
As we market and sell our approved products and advance our drug candidates through clinical development toward commercialization, we continue to build and maintain our supply chain and quality assurance resources. We rely on internal capabilities and an international network of third parties to manufacture and distribute our products for commercial sale and post-approval clinical trials and to manufacture and distribute our drug candidates for clinical trials. Wherever possible, we seek to establish multiple suppliers for each raw material and step in the manufacturing process, however our supply chain includes a single-source manufacturer for (i) one step in the ivacaftor manufacturing process and (ii) the manufacture of the oral granule formulation of KALYDECO that is used for patients with CF two to five years of age.
We expect that we will continue for the foreseeable future to rely on third parties to meet most of our commercial supply needs and a significant portion of our clinical supply needs. We have established our own small-scale manufacturing capabilities, which we use for clinical trial supplies and as an additional source for commercial supplies.
Our supply chain for sourcing raw materials and manufacturing drug product ready for distribution is a multi-step international endeavor. Third-party contract manufacturers, including some in China, supply us with raw materials, and convert these raw materials into drug substance, and convert the drug substance into final dosage form. Establishing and managing this global supply chain for each of our drugs and drug candidates requires a significant financial commitment and the creation and maintenance of numerous third-party contractual relationships.
9
We have developed systems and processes to track, monitor and oversee our third-party manufacturers’ activities, including a quality assurance program intended to ensure that our third-party manufacturers comply with current Good Manufacturing Practices, or cGMP. We regularly evaluate the performance of our third-party manufacturers with the objective of confirming their continuing capabilities to meet our needs efficiently and economically. Manufacturing facilities, both foreign and domestic, are subject to inspections by or under the authority of the FDA and other U.S. and foreign government authorities.
Manufacture of KALYDECO (ivacaftor)
We obtain ivacaftor to meet our commercial and clinical supply needs through a third-party manufacturing network. A disruption in the commercial supply of KALYDECO would have a significant effect on patients, our business and our product revenues. A disruption in the clinical supply of ivacaftor could delay the completion of clinical trials and/or affect timelines for submitting regulatory filings.
Manufacture of ORKAMBI (lumacaftor/ivacaftor)
We have developed several manufacturing processes to produce commercial quantities of ORKAMBI, including a process utilizing continuous manufacturing technology as well as a batch manufacturing process. We have established manufacturing capabilities at our third-party manufacturer in the United Kingdom that is producing commercial quantities of ORKAMBI using a batch manufacturing process we designed. We have established continuous manufacturing capabilities and completed validation for these capabilities at our facility located in Boston, Massachusetts. While continuous process manufacturing has been used in many industries, we believe that we are the first company to obtain FDA approval for a fully-continuous drug product manufacturing process.
Manufacture of Tezacaftor/Ivacaftor
We are using both a batch manufacturing process and a continuous drug product manufacturing process to obtain a supply of tezacaftor tablets being used in our Phase 3 clinical trials of tezacaftor in combination with ivacaftor. If we successfully complete development and obtain approval for tezacaftor in combination with ivacaftor, we plan to produce our commercial supply of tezacaftor using a continuous drug product manufacturing process.
COMPETITION
The pharmaceutical industry is characterized by extensive research efforts, rapid technological progress and intense competition. There are many public and private companies, including pharmaceutical companies and biotechnology companies, engaged in developing products for the indications our drugs are approved to treat and the therapeutic areas we are targeting with our research and development activities. Potential competitors also include academic institutions, government agencies, other public and private research organizations and charitable venture philanthropy organizations that conduct research, seek patent protection and/or establish collaborative arrangements for research, development, manufacturing and commercialization. Many of our competitors have substantially greater financial, technical and human resources than we do. We face competition based on the safety and efficacy of our products and drug candidates, the timing and scope of regulatory approvals, the availability and cost of supply, marketing and sales capabilities, reimbursement coverage, price, patent protection and other factors. Our competitors may develop or commercialize more effective, safer or more affordable products than we are able to develop or commercialize or obtain more effective patent protection. As a result, our competitors may commercialize products more rapidly or effectively than we do, which would adversely affect our competitive position, the likelihood that our drug candidates, if approved, would achieve and maintain market acceptance and our ability to generate meaningful revenues from our products. Future competitive products may render our products, or future products, obsolete or noncompetitive.
Cystic Fibrosis
An increasing number of companies are seeking to identify and develop drug candidates for the treatment of CF, including companies such as Concert Pharmaceuticals, Galapagos NV in collaboration with Abbvie, Genzyme, which is a division of Sanofi, Novartis, Pfizer, ProQR Therapeutics, Proteostasis Therapeutics, PTC Therapeutics and several private companies. Although we are the first company to successfully develop drugs that treat the underlying cause of CF, ORKAMBI and KALYDECO are collectively approved to treat only a portion of patients with CF. Our competitors have research and development programs directed at identifying and developing CFTR potentiators, CFTR correctors, ENaC inhibitors and drug candidates with other mechanisms of action or that utilize new therapeutic approaches that seek to address
10
the underlying cause of CF. Our competitors are exploring the development of drug candidates both as monotherapies and as part of combination regimens. Our success in rapidly developing and commercializing KALYDECO and ORKAMBI may increase the resources that our competitors allocate to the development of these potential treatments for CF. If one or more competing therapies are successfully developed as a treatment for patients with CF, our revenues from ORKAMBI, KALYDECO and/or our other CF drug candidates, if then approved, could face significant competitive pressure.
GOVERNMENT REGULATION
The research, development, testing, manufacture, packaging, quality control, approval, labeling, packaging, storage, safety monitoring, record keeping, promotion, advertising, distribution import, export and marketing of our products and drug candidates are subject to extensive regulation by United States and foreign governmental authorities.
United States Government Regulation
New Drug Application Approval Processes
In the United States, the FDA regulates drugs, including small molecules, under the Federal Food, Drug and Cosmetic Act, or the FDCA, and implementing regulations. The process of obtaining regulatory approvals and the subsequent compliance with applicable federal, state, local and foreign statutes and regulations require the expenditure of substantial time and financial resources. Failure to comply with the applicable U.S. requirements at any time during the drug development process, approval process or after approval, may subject us to administrative or judicial sanctions, any of which could have a material adverse effect on us. These sanctions could include:
• | refusal to approve or delay in review of pending applications; |
• | withdrawal of an approval or the implementation of limitations on a previously approved indication for use; |
• | imposition of a clinical hold, a risk mitigation and evaluation strategy or other safety-related limitations; |
• | warning letters or “untitled letters”; |
• | product seizures; |
• | total or partial suspension of production or distribution; or |
• | injunctions, fines, disgorgement, refusals of government contracts, or civil or criminal penalties. |
The process required by the FDA before a drug may be marketed in the United States generally involves the following:
• | completion of preclinical laboratory tests, animal studies and formulation studies conducted according to Good Laboratory Practices, or GLP, and other applicable regulations; |
• | submission to the FDA of an investigational new drug, or IND, application, which must become effective before clinical trials in the United States may begin; |
• | performance of adequate and well-controlled clinical trials according to Good Clinical Practices, or GCP, to establish the safety and efficacy of the proposed drug for its intended use; |
• | submission to the FDA of an NDA; |
• | satisfactory completion of an FDA inspection of the manufacturing facility or facilities at which the product will be produced to assess compliance with cGMP to assure that the facilities, methods and controls are adequate to preserve the product’s identity, strength, quality and purity; and |
• | FDA review and approval of the NDA. |
Once a drug candidate is identified for development, it enters the preclinical testing stage. Preclinical tests include laboratory evaluations of product chemistry, toxicity and formulation, as well as animal pharmacology and toxicology studies. An IND sponsor must submit the results of the preclinical tests, together with manufacturing information and analytical data, to the FDA as part of the IND. Preclinical or nonclinical testing typically continues even after the IND is
11
submitted. In addition to including the results of the preclinical studies, the IND also will include a protocol detailing, among other things, the objectives of the initial clinical trial and the parameters to be used in monitoring safety. The IND automatically becomes effective 30 days after receipt by the FDA, unless the FDA, within the 30-day time period, places the IND on clinical hold. If an IND is placed on clinical hold, the IND sponsor and the FDA must resolve any outstanding concerns before clinical trials can begin. A clinical hold may occur at any time during the life of an IND, and may affect one or more specific clinical trials or all clinical trials conducted under the IND.
All clinical trials must be conducted under the supervision of one or more qualified investigators in accordance with GCP. They must be conducted under protocols detailing the objectives of the trial, dosing procedures, subject selection and exclusion criteria and the safety and effectiveness criteria to be evaluated. Each protocol and any amendments must be submitted to the FDA as part of the IND, and progress reports detailing the results of the clinical trials must be submitted at least annually to the FDA and more frequently in other situations, including the occurrence of serious adverse events. An institutional review board, or IRB, at each institution participating in the clinical trial must review and approve the protocol and any amendments before a clinical trial commences or continues at that institution, approve the information regarding the clinical trial and the consent form that must be provided to each trial subject or his or her legal representative, and monitor the clinical trial until completed and otherwise comply with IRB regulations. Information about certain clinical trials must be submitted within specific timeframes to the National Institutes of Health for public dissemination on the www.clinicaltrials.gov website.
Clinical trials typically are conducted in three sequential phases that may overlap or be combined:
• | Phase 1. The drug initially is introduced into healthy human subjects and tested for safety, dosage tolerance, absorption, metabolism, distribution and elimination. In the case of some drug candidates for severe or life-threatening diseases, such as cancer, especially when the drug candidate may be inherently too toxic to ethically administer to healthy volunteers, the initial human testing is often conducted in patients. |
• | Phase 2. Clinical trials are initiated in a limited patient population intended to identify possible adverse effects and safety risks, to preliminarily evaluate the efficacy of the drug candidate for specific targeted diseases and to determine dosage tolerance and optimal dosage. |
• | Phase 3. Clinical trials are undertaken to further evaluate dosage, clinical efficacy and safety in an expanded patient population at geographically dispersed clinical trial sites. These clinical trials are intended to establish the overall risk-benefit ratio of the drug candidate and provide an adequate basis for regulatory approval and product labeling. |
Phase 1, Phase 2 and Phase 3 testing may not be completed successfully within any specified period, if at all. The FDA or the sponsor may suspend a clinical trial at any time for a variety of reasons, including a finding that the healthy volunteers or patients are being exposed to an unacceptable health risk. Similarly, an IRB can suspend or terminate approval of a clinical trial at its institution if the clinical trial is not being conducted in accordance with the IRB’s requirements or if the drug candidate has been associated with unexpected serious harm to healthy volunteers or patients.
We estimate that it generally takes 10 to 15 years, or possibly longer, to discover, develop and bring to market a new pharmaceutical product in the United States, as outlined below:
Phase | Estimated Duration | |
Discovery | 2 to 4 years | |
Preclinical | 1 to 2 years | |
Phase 1 | 1 to 2 years | |
Phase 2 | 2 to 4 years | |
Phase 3 | 2 to 4 years | |
FDA approval | 8 months to 2 years | |
During the development of a new drug, sponsors are given opportunities to meet with the FDA at certain points. These points may be prior to submission of an IND, at the end of Phase 2 testing, and before an NDA is submitted. Meetings at other times may be requested. These meetings can provide an opportunity for the sponsor to share information about the data gathered to date, for the FDA to provide advice, and for the sponsor and FDA to reach agreement on the next phase of development. Sponsors typically use the end of Phase 2 meeting to discuss their Phase 2 clinical results and present their plans for the pivotal Phase 3 clinical trial that they believe will support approval of the drug candidate.
12
As part of the development process, companies usually complete animal safety studies and develop additional information about the chemistry and physical characteristics of the drug and finalize a process for manufacturing the product in accordance with cGMP requirements. The manufacturing process must be capable of consistently producing quality batches of the drug candidate, and the manufacturer must develop methods for testing the quality, purity and potency of the final products. Additionally, appropriate packaging must be selected and tested and stability studies must be conducted to demonstrate that the drug candidate does not undergo unacceptable deterioration over its shelf-life.
The results of drug development, preclinical studies and clinical trials, along with descriptions of the manufacturing process, analytical tests conducted on the chemistry of the drug candidate, proposed labeling and other relevant information are submitted to the FDA as part of an NDA requesting approval to market the drug candidate. The FDA reviews each NDA submitted to ensure that it is sufficiently complete for substantive review before it accepts it for filing. It may request additional information rather than accept an NDA for filing. The FDA has approximately two months to make a decision on whether to accept an NDA for filing.
Once the submission is accepted for filing, the FDA begins an in-depth review. The FDA may not approve an NDA if the applicable regulatory criteria are not satisfied or may require additional clinical or other data. Even if such data are submitted, the FDA may ultimately decide that the NDA does not satisfy the criteria for approval. The FDA reviews an NDA to determine, among other things, whether a drug candidate is safe and effective for its intended use and whether its manufacturing is cGMP-compliant to assure and preserve the drug candidate’s identity, strength, quality and purity. The FDA may refer the NDA to an advisory committee for review and recommendation as to whether the NDA should be approved and under what conditions. The FDA is not bound by the recommendation of an advisory committee, but it generally follows such recommendations. Before approving an NDA, the FDA will inspect the facility or facilities where the drug candidate is manufactured and tested. Additionally, before approving an NDA, the FDA may inspect one or more clinical trial sites to assure compliance with GCP requirements.
The FDA may require, as a condition of approval, restricted distribution and use, enhanced labeling, special packaging or labeling, expedited reporting of certain adverse events, pre-approval of promotional materials, restrictions on direct-to-consumer advertising or commitments to conduct additional research post-approval. The FDA will issue a complete response letter if the agency decides not to approve the NDA in its present form. The complete response letter usually describes all of the specific deficiencies in the NDA identified by the FDA. If a complete response letter is issued, the applicant may either resubmit the NDA, addressing all of the deficiencies identified in the letter, or withdraw the application.
Biologics License Application Process
Certain of our drug candidates may be regulated by the FDA under the FDCA and the Public Health Service Act as biologics. Biologics can present special safety, efficacy and manufacturing challenges that may differ from those present in the regulation of small molecule drugs. As such, while similar to the NDA review process described above, in lieu of filing an NDA, biologics require the submission of a Biologics License Application, or BLA, and approval of such BLA by the FDA prior to being marketed in the U.S.
Expedited Review and Approval
The FDA has various programs, including Fast Track, priority review, and accelerated approval, that are intended to expedite or simplify the process for reviewing drug candidates, and/or provide for approval on the basis of surrogate endpoints. Even if a drug candidate qualifies for one or more of these programs, the FDA may later decide that the drug candidate no longer meets the conditions for qualification or that the time period for FDA review or approval will not be shortened. Generally, drug candidates that may be eligible for these programs are those for serious or life-threatening conditions, those with the potential to address unmet medical needs, and those that offer meaningful benefits over existing treatments. For example, Fast Track is a process designed to facilitate the development, and expedite the review of drug candidates to treat serious diseases and fill an unmet medical need. Priority review is designed to give drug candidates that offer significant improvements in safety or effectiveness or provide a treatment where no adequate therapy exists an initial review within six months after acceptance for filing, as compared to a standard review time of ten months after acceptance for filing. Although Fast Track and priority review do not affect the standards for approval, the FDA will attempt to facilitate early and frequent meetings with a sponsor of a Fast Track designated drug candidate and expedite review of the application for a drug candidate designated for priority review. Accelerated approval provides an earlier approval of drugs that treat serious diseases, and that fill an unmet medical need based on a surrogate endpoint, which is a laboratory measurement or
13
physical sign that the FDA determines is reasonably likely to predict a clinical benefit. As a condition of approval, the FDA may require that a sponsor of a product receiving accelerated approval perform post-marketing confirmatory clinical trials.
In July 2012, the Food and Drug Administration Safety and Innovation Act, or FDASIA, was enacted, amending the FDCA. As part of FDASIA, Congress created a drug designation called “Breakthrough Therapy.” This designation is intended to facilitate expedited development and review of a compound which, alone or in combination with one or more other compounds, is intended to treat a serious or life-threatening disease or condition and for which preliminary clinical evidence indicates that the compound may demonstrate substantial clinical improvement over existing therapies. Breakthrough Therapy designation may be requested at the filing of, or as an amendment to, an IND based on criteria established by the FDA.
Actions identified in FDASIA that may expedite the development and review of a Breakthrough Therapy include, as appropriate: holding meetings with the sponsor and the review team throughout the development of the drug; involving senior managers and experienced review staff, as appropriate, in a collaborative, cross-disciplinary review; and assigning a cross-disciplinary project lead for the FDA review team to facilitate efficient review of the development program and serve as a scientific liaison between the review team and the sponsor.
Post-approval Requirements
Once an approval is granted, the FDA may withdraw the approval if compliance with regulatory standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product may result in restrictions on the product or complete withdrawal of the product from the market. In addition, under the FDCA the sponsor of an approved drug in the United States may not promote that drug for unapproved, or off-label, uses, although a physician may prescribe a drug for an off-label use in accordance with the practice of medicine. After approval, some types of changes to the approved product, such as adding new indications, manufacturing changes and additional labeling claims, are subject to further FDA review and approval. In addition, the FDA may require testing and surveillance programs to monitor the effect of approved products that have been commercialized, and the FDA has the power to prevent or limit further marketing of a product based on the results of these post-marketing programs.
Products manufactured or distributed by us pursuant to FDA approvals are subject to continuing regulation by the FDA, including, among other things:
• | record-keeping requirements; |
• | reporting of adverse experiences with the product; |
• | providing the FDA with updated safety and efficacy information; |
• | drug sampling and distribution requirements; |
• | notifying the FDA and gaining its approval of specified manufacturing or labeling changes; |
• | complying with certain electronic records and signature requirements; and |
• | complying with FDA promotion and advertising requirements. |
Drug manufacturers and other entities involved in the manufacture and distribution of approved products are required to register with the FDA and certain state agencies, and are subject to periodic unannounced inspections by the FDA and some state agencies for compliance with cGMP and other laws.
We rely, and expect to continue to rely, on third parties for the production of our products. Future FDA and state inspections may identify compliance issues at the facilities of our contract manufacturers that may disrupt manufacture or distribution of our products, or require substantial resources to correct.
From time to time, new legislation is enacted that changes the statutory provisions governing the approval, manufacturing and marketing of products regulated by the FDA. In addition, FDA regulations and guidance often are revised or reinterpreted by the agency in ways that may significantly affect our business and our products. It is impossible to predict whether legislative changes will be enacted, or FDA regulations, guidance or interpretations changed.
14
Patent Term Restoration and Data Exclusivity
Depending upon the timing, duration and specifics of FDA approval of the use of our drugs, some of our United States patents may be eligible for limited patent term extension under the Drug Price Competition and Patent Term Restoration Act of 1984, referred to as the Hatch-Waxman Amendments. The Hatch-Waxman Amendments permit a patent restoration term of up to five years as compensation for patent term lost during product development and the FDA regulatory review process. However, patent term restoration cannot extend the remaining term of a patent beyond a total of 14 years from the product’s approval date. The patent term restoration period is generally one-half the time between the later of the effective date of an IND or the issuance date of the patent, and the submission date of an NDA, plus the time between the submission date of an NDA and the approval of that application. Only one patent applicable to an approved product is eligible for the extension and the extension must be applied for prior to expiration of the patent. The United States Patent and Trademark Office, in consultation with the FDA, reviews and approves the application for any patent term extension or restoration. In the future, we may apply for restorations of patent term for some of our currently owned or licensed patents to add patent life beyond their current expiration date, depending on the expected length of clinical trials and other factors involved in the submission of the relevant NDA.
Data exclusivity provisions under the FDCA also can delay the submission or the approval of certain applications. The FDCA provides a five-year period of non-patent data exclusivity within the United States to the first applicant to gain approval of an NDA for a new chemical entity. A product is a new chemical entity if the FDA has not previously approved any other new product containing the same active moiety, which is the molecule responsible for the action of the drug substance. During the data exclusivity period for a new chemical entity, the FDA may not accept for review an abbreviated new drug application, or ANDA, or a 505(b)(2) NDA submitted by another company for another version of such product where the applicant does not own or have a legal right of reference to all the data required for approval. However, an application may be submitted after four years if it contains a certification of patent invalidity or non-infringement. The FDCA also provides three years of data exclusivity for an NDA, 505(b)(2) NDA or supplement to an existing NDA if new clinical investigations, other than bioavailability studies, that were conducted or sponsored by the applicant are deemed by the FDA to be essential to the approval of the application, for example, for new indications, dosages, or strengths of an existing drug. This three-year exclusivity covers only the conditions associated with the new clinical investigations and does not prohibit the FDA from approving ANDAs for drugs containing the original active agent.
Pediatric Exclusivity
Section 505A of the FDCA, as amended by the FDA Amendments Act of 2007, provides for pediatric exclusivity which, if granted, provides for the attachment of an additional six months to the term of any existing regulatory exclusivity, including non-patent and orphan exclusivity. If the sponsor submits information requested in writing by the FDA, in the form of a written request, relating to the use of the drug in children and such information is accepted by FDA within the statutory time limits, whatever regulatory periods of exclusivity or patent protection covering the product are extended by six months. The FDA may not issue a written request for clinical trials on unapproved or approved indications or where it determines that information relating to the use of a drug in a pediatric population, or part of the pediatric population, may not produce health benefits in that population.
Exclusivity of Biologics
Biologics are entitled to exclusivity under the Biologics Price Competition and Innovation Act, which was passed as Title VII to the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act of 2010, which we refer to as the ACA. The law provides a pathway for approval of biosimilars following the expiration of 12 years of exclusivity for the innovator biologic plus any extension term for pediatrics as discussed above. Historically, a biologic approved under a BLA was not subject to the generic drug review and approval provisions of the FDCA. However, the ACA created a regulatory pathway for the abbreviated approval for biological products that are demonstrated to be “biosimilar” or “interchangeable” with an FDA-approved biological product. In order to meet the standard of interchangeability, a sponsor must demonstrate that the biosimilar product can be expected to produce the same clinical result as the reference product, and for a product that is administered more than once, that the risk of switching between the reference product and biosimilar product is not greater than the risk of maintaining the patient on the reference product. Such biosimilars would reference biological products approved in the United States. The law establishes a period of 12 years of data exclusivity for reference products, which protects the data in the original BLA by prohibiting sponsors of biosimilars from gaining FDA approval based in part on reference to data in the original BLA.
15
Foreign Regulation
In addition to regulations in the United States, we are subject to a variety of foreign regulations governing clinical trials and commercial sales and distribution of our products. Whether or not we obtain FDA approval for a drug candidate, we must obtain approval by the comparable regulatory authorities of foreign countries or economic areas, such as the European Union, before we can commence clinical trials or market products in those countries or areas. The approval process and requirements governing the conduct of clinical trials, product licensing, pricing and reimbursement vary greatly from place to place, and the time may be longer or shorter than that required for FDA approval.
Under European Union regulatory systems, a company may submit marketing authorization applications either under a centralized or decentralized procedure. The centralized procedure, which is compulsory for medicines produced by biotechnology or those medicines intended to treat AIDS, cancer, neurodegenerative disorders, or diabetes and optional for those medicines that are highly innovative, provides for the grant of a single marketing authorization that is valid for all European Union member states. The decentralized procedure provides for approval by one or more “concerned” member states based on an assessment of an application performed by one member state, known as the “reference” member state. Under the decentralized approval procedure, an applicant submits an application, or dossier, and related materials to the reference member state and concerned member states. The reference member state prepares a draft assessment and drafts of the related materials within 120 days after receipt of a valid application. Within 90 days of receiving the reference member state’s assessment report, each concerned member state must decide whether or not to approve the assessment report and related materials. If a member state does not recognize the marketing authorization, the disputed points are eventually referred to the European Commission, whose decision is binding on all member states.
Orphan Drug Designation
Under the Orphan Drug Act, the FDA may grant orphan drug designation to drug candidates intended to treat a rare disease or condition, which is generally a disease or condition that affects fewer than 200,000 people in the United States, or more than 200,000 people in the United States and for which there is no reasonable expectation that the cost of developing and making available in the United States a drug for this type of disease or condition will be recovered from sales in the United States for that drug. Orphan drug designation must be requested before submitting an NDA or BLA. After the FDA grants orphan drug designation, the identity of the therapeutic agent and its potential orphan use are disclosed publicly by the FDA. Orphan drug designation does not convey any advantage in or shorten the duration of the regulatory review and approval process. KALYDECO, ORKAMBI and tezacaftor have been granted designation as orphan drugs by the FDA.
If a drug candidate that has orphan drug designation subsequently receives the first FDA approval for that drug for the disease for which it has such designation, the product is entitled to orphan drug exclusivity, which means that the FDA may not approve any other applications to market the same drug for the same indication, except in very limited circumstances, for seven years. Orphan drug exclusivity, however, also could block the approval of our drug candidates for seven years if a competitor first obtains approval of the same product as defined by the FDA or if our drug candidate is determined to be contained within the competitor’s product for the same indication or disease.
As in the United States, we may apply for designation of a drug candidate as an orphan drug for the treatment of a specific indication in the European Union before the application for marketing authorization is made. Orphan drugs in Europe enjoy economic and marketing benefits, including up to 10 years of market exclusivity for the approved indication unless another applicant can show that its product is safer, more effective or otherwise clinically superior to the orphan-designated product.
The FDA and foreign regulators expect holders of exclusivity for orphan drugs, such as KALYDECO and ORKAMBI, to ensure the availability of sufficient quantities of their orphan drugs to meet the needs of patients. Failure to do so could result in the withdrawal of marketing exclusivity for the orphan drug.
Reimbursement
Sales of our products depend, to a large degree, on the extent to which our products will be covered by third-party payors, such as government health programs, commercial insurance and managed health care organizations. These third-party payors increasingly are reducing reimbursements for medical products and services. Additionally, the containment of health care costs has become a priority of federal and state governments, and the prices of drugs have been a focus in this effort. The U.S. government, state legislatures and foreign governments have shown significant interest in implementing cost-containment programs, including price controls, restrictions on reimbursement and requirements for substitution of
16
generic products. Adoption of price controls and cost-containment measures, and adoption of more restrictive policies in jurisdictions with existing controls and measures, could limit our revenues. Decreases in third-party reimbursement for a product or a decision by a third-party payor to not cover a product could reduce physician usage of the product.
The Medicare Prescription Drug, Improvement, and Modernization Act of 2003, or the MMA, established the Medicare Part D program to provide a voluntary prescription drug benefit to Medicare beneficiaries. Under Part D, Medicare beneficiaries may enroll in prescription drug plans offered by private entities, which will provide coverage of outpatient prescription drugs. Unlike Medicare Part A and B, Part D coverage is not standardized. Part D prescription drug plan sponsors are not required to pay for all covered Part D drugs, and each drug plan can develop its own drug formulary that identifies which drugs it will cover and at what tier or level. However, Part D prescription drug formularies must include drugs within each therapeutic category and class of covered Part D drugs, though not necessarily all the drugs in each category or class. Any formulary used by a Part D prescription drug plan must be developed and reviewed by a pharmacy and therapeutic committee. Government payment for some of the costs of prescription drugs may increase demand for products for which we receive marketing approval. However, any negotiated prices for our products covered by a Part D prescription drug plan likely will be lower than the prices we might otherwise obtain. Moreover, while the MMA applies only to drug benefits for Medicare beneficiaries, private payors often follow Medicare coverage policy and payment limitations in setting their own payment rates. Any reduction in payment that results from the MMA may result in a similar reduction in payments from non-governmental payors.
The American Recovery and Reinvestment Act of 2009 provides funding for the federal government to compare the effectiveness of different treatments for the same illness. A plan for the research will be developed by the Department of Health and Human Services, or HHS, the Agency for Healthcare Research and Quality and the National Institutes of Health, and periodic reports on the status of the research and related expenditures will be made to the U.S. Congress. Although the results of the comparative effectiveness studies are not intended to mandate coverage policies for public or private payors, it is not clear what effect, if any, the research will have on the sales of our products. It is possible that comparative effectiveness research demonstrating benefits of a competitor’s product could adversely affect the sales of our products. If third-party payors do not consider our products to be cost-effective compared to other available therapies, they may not cover our products as a benefit under their plans or, if they do, the level of payment may not be sufficient to allow us to sell our products on a profitable basis.
The ACA was enacted in March 2010 and is designed to expand coverage for the uninsured while at the same time containing overall health care costs. With regard to pharmaceutical products, among other things, the ACA is designed to expand and increase industry rebates for drugs covered under Medicaid programs, impose an annual fee on branded pharmaceutical manufacturers and make changes to the coverage requirements under the Medicare Part D program. The branded prescription drug fee is not tax deductible. We cannot predict all of the effects of the ACA on pharmaceutical companies as many of the ACA reforms require the promulgation of detailed regulations implementing the statutory provisions, which has not yet occurred.
In Europe and many other foreign countries, the success of ORKAMBI, KALYDECO and of any other drug candidates we may develop, depends largely on obtaining and maintaining government reimbursement, because in many foreign countries patients are unable to access prescription pharmaceutical products that are not reimbursed by their governments. Negotiating reimbursement rates in foreign countries can delay the commercialization of a pharmaceutical product and generally results in a reimbursement rate that is lower than the net price that companies can obtain for the same product in the United States.
In some countries, such as Germany and France, commercial sales of a new product can occasionally begin while the reimbursement rate that a company will receive is under discussion. In other countries, a company must complete the reimbursement discussions prior to the commencement of commercial sales of the pharmaceutical product. The requirements governing drug pricing vary widely from country to country. For example, the member states of the European Union can restrict the range of drugs for which their national health insurance systems provide reimbursement and to control the prices of drugs for human use. A member state may approve a specific price for the drug or it may instead adopt a system of direct or indirect controls on the profitability of the company placing the drug on the market. Recently, many countries in the European Union have increased the amount of discounts required on pharmaceuticals and these efforts could continue as countries attempt to manage healthcare expenditures, especially in light of the severe fiscal and debt crises experienced by many countries in the European Union. There can be no assurance that any country that has price controls or reimbursement limitations for pharmaceutical products will provide for reimbursement of our products, or such countries may only provide
17
for reimbursement on terms that we do not deem adequate. Additionally, reimbursement discussions in ex-U.S. markets may take a significant period of time.
Other United States Regulations
Pharmaceutical companies also are subject to various federal and state laws pertaining to health care “fraud and abuse,” including anti-kickback laws and false claims laws, and the reporting of payments to physicians and teaching hospitals.
Anti-kickback Laws
U.S. federal laws prohibit fraud and abuse involving state and federal health care programs, such as Medicare and Medicaid. These laws are interpreted broadly and enforced aggressively by various state and federal agencies, including the Centers for Medicare & Medicaid Services, or CMS, the Department of Justice, the Office of Inspector General for HHS and various state agencies. These anti-kickback laws prohibit, among other things, knowingly and willfully offering, paying, soliciting, receiving or providing remuneration, directly or indirectly, in exchange for or to induce either the referral of an individual, or the furnishing, arranging for or recommending of an item or service that is reimbursable, in whole or in part, by a federal health care program. Remuneration is broadly defined to include anything of value, such as, cash payments, gifts or gift certificates, discounts, or the furnishing of services, supplies or equipment. The anti-kickback laws are broad and prohibit many arrangements and practices that are lawful in businesses outside of the health care industry.
The penalties for violating the anti-kickback laws can be severe. The sanctions include criminal and civil penalties, and possible exclusion from the federal health care programs. Many states have adopted laws similar to the federal anti-kickback laws, and some apply to items and services reimbursable by any payor, including third-party payors.
State and Federal Prohibitions on False Claims
The federal False Claims Act imposes liability on any person or entity that, among other things, knowingly presents, or causes to be presented, a false or fraudulent claim for payment to the federal government. Under the False Claims Act, a person acts knowingly if he has actual knowledge of the information or acts in deliberate ignorance or in reckless disregard of the truth or falsity of the information. Specific intent to defraud is not required. Provisions of the False Claims Act allow a private individual to bring an action on behalf of the federal government and to share in any amounts paid by the defendant to the government in connection with the action. The number of filings under these provisions has increased significantly in recent years. When an entity is determined to have violated the False Claims Act, it may be required to pay up to three times the actual damages sustained by the government, plus civil penalties for each false claim. Conduct that violates the False Claims Act may also lead to exclusion from the federal health care programs. Given the number of claims likely to be at issue, potential damages under the False Claims Act for even a single inappropriate arrangement could be significant. In addition, various states have enacted similar laws modeled after the False Claims Act that apply to items and services reimbursed under Medicaid and other state health care programs, and, in several states, such laws apply to claims submitted to all payors.
Federal Prohibitions on Health Care Fraud and False Statements Related to Health Care Matters
Under the administrative simplification provisions of the Health Insurance Portability and Accountability Act of 1996, or HIPAA, and state laws there are numerous regulations for protecting the privacy and security of protected health information. Additional administrative simplification provisions created the following federal crimes: health care fraud, false statements relating to health care matters, theft or embezzlement in connection with a health benefit program and obstruction of criminal investigation of health care offenses. The health care fraud statute prohibits knowingly and willfully executing a scheme to defraud any health care benefit program, including a private insurer. The false statements statute prohibits knowingly and willfully falsifying, concealing, or covering up a material fact or making any materially false, fictitious, or fraudulent statement in connection with the delivery of or payment for health care benefits, items, or services. The theft or embezzlement statute prohibits knowingly and willfully embezzling, stealing or otherwise converting or misapplying the money or property of a health care benefit program. The obstruction of criminal investigations of health care offenses statute prohibits willfully preventing, obstructing, misleading or delaying the communication of information and records relating to a violation of a federal health care offense to a criminal investigator. A violation of any of these laws is a felony and may result in fines, or exclusion from the federal health care programs.
18
US Open Payments (formerly Physician Payment Sunshine Act)
Open Payments (commonly known as the Sunshine Act) is a federal program, required by the ACA, that requires pharmaceutical manufacturers to report annually to the Centers for Medicare and Medicaid Services payments or other transfers of value made by that entity to physicians and teaching hospitals. In February 2013, regulations were released that contain detailed guidance regarding the information that must be collected and reported. We were required to collect information regarding such payments starting in August 2013 and were required to begin reporting such information in March 2014. Over the next several years, we will need to continue to dedicate significant resources to enhance our systems and processes in order to comply with these regulations. Failure to comply with the reporting requirements could result in significant civil monetary penalties. Similar laws have been enacted or are under consideration in foreign jurisdictions, including France which has adopted the Loi Bertrand, or French Sunshine Act, which became effective in 2013.
The Foreign Corrupt Practices Act
The Foreign Corrupt Practices Act prohibits U.S. companies and their representatives from offering, promising, authorizing or making payments to foreign officials for the purpose of obtaining or retaining business abroad. In many countries, the health care professionals we regularly interact with may meet the definition of a foreign government official for purposes of the Foreign Corrupt Practices Act.
Other Regulations
In addition to the statutes and regulations described above, we also are subject to regulation in the United States under the Occupational Safety and Health Act, the Environmental Protection Act, the Toxic Substances Control Act, the Resource Conservation and Recovery Act and other federal, state, local and foreign statutes and regulations, now or hereafter in effect.
EMPLOYEES
As of December 31, 2016, we had approximately 2,150 employees, as compared to approximately 1,950 employees as of December 31, 2015. Of these employees, approximately 1,725 were based in the United States, approximately 350 were based in Europe and approximately 75 were based in Canada. In February 2017, we decided to consolidate our research activities into our Boston, Milton Park and San Diego locations and are in the process of closing our research site in Canada. Our scientific staff members have diversified experience and expertise in molecular and cell biology, genetics, biochemistry, synthetic organic chemistry, protein X-ray crystallography, protein nuclear magnetic resonance spectroscopy, microbiology, computational chemistry and computational biology, biophysical chemistry, medicinal chemistry, clinical pharmacology and clinical medicine. Our clinical development personnel have extensive expertise in designing and executing clinical trials. Employees in our commercial organization have extensive experience in selling and marketing pharmaceutical products as well as seeking reimbursement from government and third-party payors for pharmaceutical products. Our employees are not covered by a collective bargaining agreement, except for a small number of employees outside the U.S. Science magazine named Vertex as one of its top employers in the life sciences in each of the last five years. We consider our relations with our employees to be good.
OTHER MATTERS
Financial Information and Significant Customers
Financial information about (i) our net product revenues and other revenues generated in the principal geographic regions in which we operate and our significant customers is set forth in Note T, “Segment Information,” to our consolidated financial statements included in this Annual Report on Form 10-K, (ii) net income (loss) per share attributable to Vertex common shareholders and our total assets are provided in our consolidated financial statements included in this Annual Report on Form 10-K and (iii) our research and development expenses in each of the last three fiscal years and our deconsolidation of Alios as of December 31, 2014 is provided in Item 7, “Management’s Discussion and Analysis of Financial Condition and Results of Operations.” A discussion of the risks attendant to our international operations is set forth in the “Risk Factors” section of this Annual Report on Form 10-K.
Information Available on the Internet
Our internet address is www.vrtx.com. Our annual reports on Form 10-K, quarterly reports on Form 10-Q and current reports on Form 8-K, and all amendments to those reports, are available to you free of charge through the “Investors-SEC
19
Filings” section of our website as soon as reasonably practicable after those materials have been electronically filed with, or furnished to, the Securities and Exchange Commission.
Corporate Information
Vertex was incorporated in Massachusetts in 1989, and our principal executive offices are located at 50 Northern Avenue Boston, Massachusetts 02210.
20
DIRECTORS AND EXECUTIVE OFFICERS OF THE REGISTRANT
The names, ages and positions held by our executive officers and directors are as follows:
Name | Age | Position | |
Jeffrey M. Leiden, M.D., Ph.D. | 61 | Chairman of the Board, Chief Executive Officer and President | |
David Altshuler, M.D., Ph.D. | 52 | Executive Vice President, Global Research and Chief Scientific Officer | |
Stuart A. Arbuckle | 51 | Executive Vice President and Chief Commercial Officer | |
Jeffrey A. Chodakewitz, M.D. | 61 | Executive Vice President, Global Medicines Development and Medical Affairs, and Chief Medical Officer | |
Michael Parini, J.D. | 42 | Executive Vice President and Chief Legal and Administrative Officer | |
Amit K. Sachdev, J.D. | 49 | Executive Vice President, Chief Regulatory Officer and Chief of Staff to the CEO | |
Ian F. Smith | 51 | Executive Vice President, Chief Operating Officer and Chief Financial Officer | |
Paul M. Silva | 51 | Senior Vice President and Corporate Controller | |
Sangeeta M. Bhatia, M.D., Ph.D. | 48 | Director | |
Joshua S. Boger, Ph.D. | 65 | Director | |
Terrence C. Kearney | 62 | Director | |
Yuchun Lee | 51 | Director | |
Margaret G. McGlynn | 57 | Director | |
Bruce I. Sachs | 57 | Director | |
Elaine S. Ullian | 69 | Director | |
William Young | 72 | Director | |
Dr. Leiden is our Chairman, Chief Executive Officer and President. He has held the positions of Chief Executive Officer and President since February 2012 after joining us as CEO Designee in December 2011. He has been a member of our Board of Directors since July 2009, the Chairman of our Board of Directors since May 2012, and served as our lead independent director from October 2010 through December 2011. Dr. Leiden was a Managing Director at Clarus Ventures, a life sciences venture capital firm, from 2006 through January 2012. Dr. Leiden was President and Chief Operating Officer of Abbott Laboratories, Pharmaceuticals Products Group, and a member of the Board of Directors of Abbott Laboratories from 2001 to 2006. From 1987 to 2000, Dr. Leiden held several academic appointments, including the Rawson Professor of Medicine and Pathology and Chief of Cardiology and Director of the Cardiovascular Research Institute at the University of Chicago, the Elkan R. Blout Professor of Biological Sciences at the Harvard School of Public Health, and Professor of Medicine at Harvard Medical School. He is an elected member of both the American Academy of Arts and Sciences, and the Institute of Medicine of the National Academy of Sciences. Dr. Leiden is a senior advisor to Clarus Ventures. Dr. Leiden serves as a director of Quest Diagnostics Inc., a medical diagnostics company, and Massachusetts Mutual Life Insurance Company, an insurance company. Dr. Leiden was a director and the non-executive Vice Chairman of the board of Shire plc, a specialty biopharmaceutical company, from 2006 to January 2012. Dr. Leiden received his M.D., Ph.D. and B.A. degrees from the University of Chicago.
Dr. Altshuler has been our Executive Vice President, Global Research and Chief Scientific Officer since January 2015 and was a member of our Board of Directors from May 2012 through December 2014. Dr. Altshuler was one of four founding members of the Broad Institute, a research collaboration of Harvard, MIT, The Whitehead Institute and the Harvard Hospitals. He served as the Director of the Institute’s Program in Medical and Population Genetics from 2003 through December 2014 and as the Institute’s Deputy Director and Chief Academic Officer from 2009 through December 2014. Dr. Altshuler joined the faculty at Harvard Medical School and the Massachusetts General Hospital in 2000 and held the academic rank of Professor of Genetics and Medicine from 2008 through December 2014. He served as Adjunct Professor of Biology at MIT from 2012 through December 2014. Dr. Altshuler earned a B.S. from MIT, a Ph.D. from Harvard University and an M.D. from Harvard Medical School. Dr. Altshuler completed his clinical training in Internal Medicine, and in Endocrinology, Diabetes and Metabolism, at the Massachusetts General Hospital.
21
Mr. Arbuckle is our Executive Vice President and Chief Commercial Officer, a position he has held since September 2012. Prior to joining us, Mr. Arbuckle held multiple commercial leadership roles at Amgen, Inc., a 17,000 person biotechnology company, from July 2004 through August 2012. Mr. Arbuckle has worked in the biopharmaceuticals industry since 1986, including more than 15 years at GlaxoSmithKline plc, where he held sales and marketing roles of increasing responsibility for medicines aimed at treating respiratory, metabolic, musculoskeletal, cardiovascular and other diseases. He has served as a member of the Board of Directors of Cerulean Pharma, Inc. since June 2015. Mr. Arbuckle holds a BSc in pharmacology and physiology from the University of Leeds.
Dr. Chodakewitz is our Executive Vice President, Global Medicines Development and Medical Affairs and Chief Medical Officer. Dr. Chodakewitz joined Vertex as a Senior Vice President in January 2014 and became an Executive Vice President in October 2014. Prior to joining us, Dr. Chodakewitz spent more than 20 years at Merck & Co., Inc., where he held a variety of roles including Vice President of Clinical Research – Infectious Diseases & Vaccines, Vice President of Clinical Pharmacology/Early Stage Development, Senior Vice President of Late Stage Development, and Senior Vice President of Global Scientific Strategy (Infectious Diseases, Respiratory/Immunology). Prior to his tenure at Merck, he served as the Director of the HIV Outpatient Clinic at the Veterans Administration Medical Center in West Haven, Connecticut and held various academic positions at Yale University and New York University Schools of Medicine. Dr. Chodakewitz serves as a member of the Board of Directors of Tetraphase Pharmaceuticals, Inc., a pharmaceutical company. Dr. Chodakewitz holds B.S. in Biochemistry from Yale University, and an M.D. from the Yale University School of Medicine.
Mr. Parini is our Executive Vice President and Chief Legal and Administrative Officer, a position he has held since January 2017. From January 2016 to January 2017, he was our Executive Vice President and Chief Legal Officer. From 2004 until he joined Vertex, Mr. Parini served in various roles of increasing responsibility at Pfizer Inc., most recently as Senior Vice President and Associate General Counsel. Prior to Pfizer, Mr. Parini was an attorney at Akin, Gump, Strauss, Hauer & Feld, L.L.P. Mr. Parini holds a B.A. from Georgetown University and a J.D. from the Georgetown University Law Center.
Mr. Sachdev is our Executive Vice President, Chief Regulatory Officer and Chief of Staff to our CEO, a role he assumed in January 2017. He served as our Executive Vice President, Policy, Access and Value, from October 2014 through December 2016. In 2007, he joined us as a Senior Vice President, and has led our government affairs and public policy activities, as well as our patient advocacy programs. From 2010 through 2013 he established our first international commercial operations in Canada. Prior to joining us, Mr. Sachdev served as Executive Vice President, Health of the Biotechnology Industry Organization (BIO) and was the Deputy Commissioner for Policy at the FDA where he also served in several other senior positions within the FDA. Prior to the FDA, Mr. Sachdev served as Majority Counsel to the Committee on Energy and Commerce in the United States House of Representatives and practiced law at the Chemical Manufacturers Association, and subsequently at the law firm of Ropes & Gray LLP. Mr. Sachdev holds a B.S from Carnegie Mellon University, and a J.D. from Emory University School of Law.
Mr. Smith is our Executive Vice President, Chief Financial Officer and Chief Operating Officer, a role he assumed in January 2017. He was our Executive Vice President and Chief Financial Officer from February 2006 until January 2017, our Senior Vice President and Chief Financial Officer from November 2003 to February 2006, and our Vice President and Chief Financial Officer from October 2001 to November 2003. Prior to joining us, Mr. Smith served as a partner in the Life Science and Technology Practice Group of Ernst & Young LLP, an accounting firm, from 1999 to 2001. Mr. Smith initially joined Ernst & Young’s U.K. firm in 1987, and then joined its Boston office in 1995. Mr. Smith has served as a member of the Boards of Directors of Acorda Therapeutics, Inc., a drug development company, since February 2007, and Infinity Pharmaceuticals, Inc., a drug development company, since May 2008. In August 2016, Mr. Smith joined the Board of Directors of Ophthotech Corporation, a biopharmaceutical company, and was also appointed to serve as Chairman of the Audit Committee of Ophthotech Corporation’s Board. Mr. Smith holds a B.A. in accounting and finance from Manchester Metropolitan University, U.K., is a member of the American Institute of Certified Public Accountants and is a Chartered Accountant of England and Wales.
Mr. Silva is our Senior Vice President and Corporate Controller, a position he has held since April 2011. Mr. Silva joined us in August 2007 as Senior Director, Accounting Operations and was our Vice President and Corporate Controller from September 2008 through April 2011. Prior to joining us, he was the Vice President, Internal Reporting at Iron Mountain Incorporated from July 2006 until August 2007 and a consultant to Iron Mountain’s financing department from April 2005 until July 2006. He was the Finance Director of the Bioscience Technologies Division of Thermo Electron Corporation from 2002 to April 2005. Mr. Silva holds a B.S. in accounting from Assumption College.
22
Dr. Bhatia has been a member of our Board of Directors since June 2015. Dr. Bhatia is a professor at the Massachusetts Institute of Technology, where she currently serves as the John J. and Dorothy Wilson Professor of Health Sciences & Technology/Electrical Engineering & Computer Science. Prior to joining the Massachusetts Institute of Technology in 2005, Dr. Bhatia was a professor of bioengineering and medicine at the University of California at San Diego from 1998 through 2005. Dr. Bhatia also is an investigator for the Howard Hughes Medical Institute, a member of the Department of Medicine at Brigham and Women’s Hospital, a member of the Broad Institute and a member of the Koch Institute for Integrative Cancer Research. Dr. Bhatia holds an Sc.B. in biomedical engineering from Brown University, an S.M. and Ph.D. in Mechanical Engineering from the Massachusetts Institute of Technology and an M.D. from Harvard Medical School.
Dr. Boger is the founder of Vertex and has been a director since our inception in 1989. He was our Chief Executive Officer from 1992 through May 2009. He was our Chairman of our Board of Directors from 1997 until May 2006 and our President from our inception until December 2000, and from 2005 through February 2009. He was our Chief Scientific Officer from 1989 until May 1992. Prior to founding Vertex in 1989, Dr. Boger held the position of Senior Director of Basic Chemistry at Merck Sharp & Dohme Research Laboratories in Rahway, New Jersey, where he headed both the Department of Medicinal Chemistry of Immunology & Inflammation and the Department of Biophysical Chemistry. Dr. Boger holds a B.A. in chemistry and philosophy from Wesleyan University and M.S. and Ph.D. degrees in chemistry from Harvard University.
Mr. Kearney has been a member of our Board of Directors since May 2011. Mr. Kearney served as the Chief Operating Officer of Hospira, Inc., a specialty pharmaceutical and medication delivery company, from April 2006 to January 2011. From April 2004 to April 2006, he served as Hospira’s Senior Vice President, Finance, and Chief Financial Officer, and he served as Acting Chief Financial Officer through August 2006. Mr. Kearney served as Vice President and Treasurer of Abbott Laboratories from 2001 to April 2004. From 1996 to 2001, Mr. Kearney was Divisional Vice President and Controller for Abbott’s International Division. Mr. Kearney serves as a member of the Board of Directors at Acceleron Pharma Inc., a biopharmaceutical company, and AveXis, Inc., a gene therapy company, and served as a member of the Board of Directors at Innoviva, Inc. (formerly known as Theravance, Inc.), a royalty management company, until April 2016. He received his B.S. in biology from the University of Illinois and his M.B.A. from the University of Denver.
Mr. Lee has been a member of our Board of Directors since September 2012. Mr. Lee serves as an Executive in Residence (XIR) and Partner of General Catalyst Partners, a venture capital firm, positions he has held since April of 2013. Mr. Lee also serves as the Chief Executive Officer of Allego, Inc. and is Executive Chairman of Clarabridge, Inc. Mr. Lee was the Vice President of IBM’s Enterprise Marketing Management Group from November 2010 through January 2013. Mr. Lee co-founded Unica Corporation, a provider of software and services used to automate marketing processes, in 1992, and was Unica’s President and/or Chief Executive Officer from 1992 through November 2010, when Unica was acquired by IBM. From 1989 to 1992, Mr. Lee was a senior consultant at Digital Equipment Corporation, a supplier of general computing technology and consulting services. Mr. Lee holds a B.S. and an M.S. in electrical engineering and computer science from the Massachusetts Institute of Technology and an M.B.A. from Babson College.
Ms. McGlynn has been a member of our Board of Directors since May 2011. Ms. McGlynn served as the President and Chief Executive Officer of the International AIDS Vaccine Initiative, a global not-for-profit organization whose mission is to ensure the development of safe, effective and accessible HIV vaccines for use throughout the world, from July 2011 until September 2015. Ms. McGlynn served as President, Vaccines and Infectious Diseases of Merck & Co., Inc. from 2005 until 2009. Ms. McGlynn joined Merck in 1983 and served in a variety of marketing, sales and managed care roles. Ms. McGlynn serves as a member of the Board of Directors for Air Products and Chemicals, Inc., a company specializing in gases and chemicals for industrial uses, and Amicus Therapeutics, Inc., a biopharmaceutical company. She is also a member of the National Industrial Advisory Committee at the University at Buffalo School of Pharmacy and Pharmaceutical Sciences. Ms. McGlynn holds a B.S. in Pharmacy and an M.B.A. in Marketing from the State University of New York at Buffalo.
Mr. Sachs has been a member of our Board of Directors since 1998. Mr. Sachs is a General Partner at Charles River Ventures, a venture capital firm he joined in 1999. From 1998 to 1999, he served as Executive Vice President and General Manager of Ascend Communications, Inc. From 1997 until 1998, Mr. Sachs served as President and Chief Executive Officer of Stratus Computer, Inc. From 1995 to 1997, he served as Executive Vice President and General Manager of the Internet Telecom Business Group at Bay Networks, Inc. From 1993 to 1995, he served as President and Chief Executive Officer of Xylogics, Inc. Mr. Sachs holds a B.S.E.E. in electrical engineering from Bucknell University, an M.E.E. in electrical engineering from Cornell University, and an M.B.A. from Northeastern University.
Ms. Ullian has been a member of our Board of Directors since 1997. Ms. Ullian served as President and Chief Executive Officer of Boston Medical Center, a private, not-for-profit, 626-bed, academic medical center with a community-based focus,
23
from 1996 through January 2010. From 1994 to 1996, she served as President and Chief Executive Officer of Boston University Medical Center Hospital. From 1987 to 1994, Ms. Ullian served as President and Chief Executive Officer of Faulkner Hospital. She also serves as a director of Thermo Fisher Scientific Inc. and Hologic, Inc. Ms. Ullian holds a B.A. in political science from Tufts University and an M.P.H. from the University of Michigan.
Mr. Young has been a member of our Board of Directors since May 2014. Mr. Young is a Venture Partner at Clarus Ventures, a life sciences venture capital firm, which he joined in 2010. Prior to Clarus Ventures, Mr. Young served from 1999 until June 2009 as the Chairman and Chief Executive Officer of Monogram Biosciences, Inc., a biotechnology company acquired by Laboratory Corporation of America in June 2009. From 1980 to 1999, Mr. Young was employed at Genentech, Inc. in positions of increasing responsibility, including as Chief Operating Officer from 1997 to 1999, where he was responsible for all product development, manufacturing and commercial functions. Prior to joining Genentech, Mr. Young was with Eli Lilly & Co. for 14 years. Mr. Young currently serves as the Chairman of the Board of Directors of NanoString Technologies, Inc., and as a member of the Board of Directors of Theravance BioPharma Inc. Mr. Young retired from BioMarin Pharmaceutical Inc.’s Board of Directors in November 2015 and from Biogen’s Board of Directors in June 2014. Mr. Young holds a B.S. in Chemical Engineering from Purdue University, an M.B.A. from Indiana University and an Honorary Doctorate in Engineering from Purdue University. Mr. Young was elected to the National Academy of Engineering in 1993 for his contributions to biotechnology.
24
ITEM 1A. RISK FACTORS
RISK FACTORS
Investing in our common stock involves a high degree of risk, and you should carefully consider the risks and uncertainties described below in addition to the other information included or incorporated by reference in this Annual Report on Form 10-K. If any of the following risks or uncertainties actually occurs, our business, financial condition or results of operations would likely suffer, possibly materially. In that case, the trading price of our common stock could decline.
Risks Related to Our Business
Our business and future revenues depend heavily on ORKAMBI net product revenues. If we are unable to increase our ORKAMBI net product revenues or if we do not meet the expectations of investors or public equity market analysts, our business will be materially harmed and the market price of our common stock would likely decline.
ORKAMBI was approved by the FDA in July 2015 and by the European Commission in November 2015, for the treatment of patients with CF twelve years of age and older who are homozygous for the F508del mutation in their CFTR gene. In September 2016, the FDA approved ORKAMBI for the treatment of patients with CF six to eleven years of age who are homozygous for the F508del mutation in their CFTR gene. Most of our ORKAMBI net product revenues have come from the United States and, except with respect to Germany, we have not recognized significant ex-U.S. net product revenues due to the time it takes to complete reimbursement discussions in many ex-U.S. countries. We have experienced challenges in the commercialization of ORKAMBI, including discontinuations by patients who had previously initiated treatment with ORKAMBI and a slower than anticipated launch in Germany. Our ability to increase ORKAMBI net product revenues will be dependent on:
• | our ability to obtain, and the timing and terms of obtaining, reimbursement for ORKAMBI in ex-U.S. markets; |
• | the number of additional patients who begin treatment with ORKAMBI, including patients six to eleven years of age in the United States; |
• | the rate at which additional patients initiate treatment; |
• | the proportion of initiated patients who remain on treatment; and |
• | the compliance rate for patients who remain on treatment. |
If we continue to experience challenges with the commercialization of ORKAMBI or if ORKAMBI were to become subject to problems such as safety or efficacy issues, the introduction or greater acceptance of competing products, changes in reimbursement policies of payors and other third parties, or adverse legal, administrative, regulatory or legislative developments, our ability to commercialize ORKAMBI would be impaired and our stock price would likely decline. Since the regulations that govern pricing, coverage and reimbursement for drugs vary widely from country to country, there is no assurance that coverage and reimbursement will be available outside of the United States and, even if it is available, the timing or the level of reimbursement may not be satisfactory. Adverse pricing limitations or a delay in obtaining coverage and reimbursement would decrease our future net product revenues and harm our business.
Our business is dependent on KALYDECO net product revenues and if we are unable to sustain our KALYDECO net product revenues, our business will be materially harmed and the market price of our common stock would decline.
KALYDECO net product revenues represented approximately 41% and 61% of our total revenues in 2016 and 2015, respectively, and we expect KALYDECO net product revenues to continue to represent a substantial portion of our total revenues in future periods. We are seeking to expand the label for KALYDECO and to increase the number of patients eligible and reimbursed for treatment with KALYDECO in the United States and ex-U.S. markets. We have initiated a clinical trial for ivacaftor in patients with CF less than two years of age to evaluate the effect of ivacaftor on markers of CF disease in young children. There can be no assurance that the data from this clinical trial will be sufficient to obtain approval for ivacaftor in this patient population. Additionally, in February 2016, we received a Complete Response Letter from the FDA regarding our sNDA for ivacaftor for patients with CF two years of age and older who have one of 23 residual function mutations. The FDA determined that it cannot approve the sNDA in its present form. While we have had discussions with
25
the FDA regarding the sNDA to determine an appropriate path forward, there can be no assurance that the outcome from these discussions will be sufficient to obtain approval for ivacaftor in this patient population.
If we are unable to sustain KALYDECO net products revenues for any reason, such as safety or efficacy issues, the introduction or greater acceptance of competing products, changes in reimbursement policies of payors and other third parties, or adverse legal, administrative, regulatory or legislative developments, our ability to commercialize KALYDECO would be impaired and our product revenues would decrease and our financial position and stock price would be materially harmed.
If we are unable to successfully develop additional drug candidates, and in particular tezacaftor in combination with ivacaftor and triple combination regimens including next-generation CFTR corrector compounds, our business will be materially harmed.
Our long-term success and revenue growth will depend upon our ability to successfully develop new products. We believe that a significant portion of the value attributed to our company by investors is based on the potential of the combination regimens that we are developing for the treatment of CF, including tezacaftor in combination with ivacaftor, which is Phase 3 development, and triple combination regimens that include our next-generation CFTR corrector compounds, including VX-152 and VX-440, each of which are in Phase 2 development, VX-659, which is in Phase 1 development, and VX-445, which we expect will enter Phase 1 development in the first quarter of 2017. Furthermore, our success will be dependent on our ability to successfully develop and ultimately commercialize drug candidates in therapeutic areas outside of CF, including drug candidates resulting from our internal research programs and drug candidates that have been or may in the future be licensed or acquired from third parties.
Drug development is inherently risky and we will be required to undertake additional clinical trials before such candidates become eligible for marketing approval. The results of clinical trials and findings from our nonclinical studies for these drug candidates, including toxicology findings in nonclinical studies conducted concurrently with clinical trials, could lead to abrupt changes in our development activities, including the possible cessation of development activities associated with a particular drug candidate or program. Many companies in the pharmaceutical and biotechnology industries, including our company, have suffered significant setbacks in later-stage clinical trials even after achieving promising results in earlier-stage clinical trials. Accordingly, the results from completed preclinical studies and clinical trials may not be replicated in later clinical trials, and ongoing clinical trials for our drug candidates may not be predictive of the results we may obtain in later-stage clinical trials or of the likelihood of approval of a drug candidate for commercial sale.
We expect data from our Phase 3 development program evaluating tezacaftor in combination with ivacaftor in the first half of 2017. If supported by data from the Phase 3 development program, we plan to submit an NDA to the FDA for tezacaftor in combination with ivacaftor in the second half of 2017. We expect to begin receiving data from clinical trials evaluating triple combination treatment regimens in the second half of 2017. If our ongoing or planned clinical trials for our drug candidates, are not successful, or if we fail to expand our pipeline outside of CF, our business will be materially harmed.
We have a history of incurring losses, and we cannot predict the extent of our future profitability.
We have incurred operating losses in each of the last three years. While our revenues have increased and our operating losses have decreased in each of the last three years, we have not achieved annual profitability on a GAAP basis. Our ability to achieve and sustain profitability depends on the extent to which we can continue to increase our revenue and control our costs in order to, among other things, counter any unforeseen difficulties, complications or other unknown factors that may impair future revenue or require additional expenditures. Our ability to increase our revenues is dependent on our ability to increase our sales of ORKAMBI, to maintain sales of KALYDECO and to develop and commercialize additional products. Our operating expenses may increase due to, among other factors, additional investments to support or accelerate our research and development activities, the expansion of our organization, and/or costs associated with business development activities, including costs to acquire assets or programs, integration costs and the costs to develop drug candidates that are acquired. Because of the numerous risks and uncertainties associated with pharmaceutical product development, we are unable to predict the extent of our future profitability or losses. If we are unable to increase sales of ORKAMBI, sustain sales of KALYDECO and develop additional products, we may not achieve and/or sustain profitability.
26
If our competitors bring drugs with superior product profiles to market, our drugs may not be competitive and our revenues could decline.
ORKAMBI, KALYDECO and any drugs that we develop in the future may not be able to compete effectively with marketed drugs or new drugs that may be developed by competitors. The risk of competition is specifically important to our company because substantially all of our revenues as well as our most advanced drug candidates are related to the treatment of patients with CF. There are many other companies developing drugs for the same indications that we are pursuing. In order to compete successfully in these areas, we must demonstrate improved safety, efficacy and/or tolerability, and ease of manufacturing, and gain and maintain market acceptance over competing drugs. Many of our competitors, including major pharmaceutical companies such as Abbvie, Bristol-Myers Squibb, Gilead, Johnson & Johnson, Merck, Merck KGaA, Novartis, Pfizer, Sanofi and Roche, possess substantially greater financial, technical and human resources than we possess. Potential competitors also include other public and private companies, academic institutions, government agencies, other public and private research organizations and charitable venture philanthropy organizations that conduct research, seek patent protection and/or establish collaborative arrangements for research, development, manufacturing and commercialization. As an example, in 2013 and 2014 we experienced a rapid decline in the number of patients being treated with INCIVEK, a product we previously marketed for the treatment of hepatitis C virus infection.
Mergers and acquisitions in the pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of our competitors. Smaller and other early-stage companies also may prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These third parties compete with us in recruiting and retaining qualified scientific and management personnel, establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our programs.
A number of companies are seeking to identify and develop drug candidates for the treatment of CF, including Galapagos NV in collaboration with Abbvie, Concert Pharmaceuticals, Genzyme, which is a division of Sanofi, Novartis, Pfizer, ProQR Therapeutics, Proteostasis Therapeutics, PTC Therapeutics and several private companies. Our competitors have research and development programs directed at identifying CFTR potentiators, CFTR correctors, ENaC inhibitors and drug candidates with other mechanisms of action or that utilize new therapeutic approaches that seek to address the underlying cause of CF. Our success in rapidly developing and commercializing ORKAMBI and KALYDECO may increase the resources that our competitors allocate to the development of these potential treatments for CF. Our competitors are exploring the development of drug candidates both as monotherapies and as part of combination regimens. If one or more competing therapies are successfully developed as a treatment for patients with CF, our revenues from ORKAMBI, KALYDECO and/or other compounds, if then approved, could face competitive pressures. If one or more competing therapies prove to be superior to our existing products and/or drug candidates for the treatment of CF, our business would be materially adversely affected.
If we discover safety issues with any of our products or if we fail to comply with continuing U.S. and applicable foreign regulations, commercialization efforts for the product could be negatively affected, the approved product could lose its approval or sales could be suspended, and our business could be materially harmed.
Our products are subject to continuing regulatory oversight, including the review of additional safety information. Drugs are more widely used by patients once approval has been obtained and therefore side-effects and other problems may be observed after approval that were not seen or anticipated, or were not as prevalent or severe, during pre-approval clinical trials or nonclinical studies. The subsequent discovery of previously unknown problems with a product could negatively affect commercial sales of the product, result in restrictions on the product or lead to the withdrawal of the product from the market. Each of our two commercial products and our most advanced drug candidates, contain ivacaftor, either alone or in combination with one or more other compounds. As a result, if either of our products were to experience safety issues, both ORKAMBI and KALYDECO, as well as one or more of our drug candidates, may be adversely affected. The reporting of adverse safety events involving our products or public speculation about such events could cause our stock price to decline or experience periods of volatility.
If we or our collaborators fail to comply with applicable continuing regulatory requirements, we or our collaborators may be subject to fines, suspension or withdrawal of regulatory approvals for specific products, product recalls and seizures, operating restrictions and/or criminal prosecutions. In addition, the manufacturers we engage to make our products and the manufacturing facilities in which our products are made are subject to periodic review and inspection by the FDA and foreign regulatory authorities. If problems are identified during the review or inspection of these manufacturers or manufacturing
27
facilities, it could result in our inability to use the facility to make our product or a determination that inventories are not safe for commercial sale.
If physicians and patients do not accept our drugs, or if patients do not remain on treatment or comply with the prescribed dosing regimen, our product revenues would be materially harmed in future periods.
Our drugs may not gain or maintain market acceptance among physicians and patients. Effectively marketing our drugs and any of our drug candidates, if approved, requires substantial efforts, both prior to launch and after approval. Physicians may elect not to prescribe our drugs, and patients may elect not to take them or may discontinue use of our drugs after initiation of treatment, for a variety of reasons including:
• | prevalence and severity of adverse side-effects; |
• | lack of reimbursement availability from third-party payors; |
• | lower demonstrated efficacy, safety and/or tolerability compared to alternative treatment methods; |
• | lack of cost-effectiveness; |
• | a decision to wait for the approval of other therapies in development that have significant perceived advantages over our drug; |
• | convenience and ease of administration; |
• | other potential advantages of alternative treatment methods; and |
• | ineffective sales, marketing and/or distribution support. |
For example, our net product revenues from ORKAMBI during 2016 were affected by discontinuations by patients who had previously initiated treatment with ORKAMBI. If the discontinuation rate for ORKAMBI or any of our other drug products increases, or if our drugs otherwise fail to achieve or maintain market acceptance, we may not be able to generate significant revenues in future periods.
Government and other third-party payors seek to contain costs of health care through legislative and other means. If they fail to provide coverage and adequate reimbursement rates for our products, our revenues will be harmed.
In both domestic and foreign markets, our sales of products depend in part upon the availability of reimbursement from third-party payors. Third-party payors include government health programs such as Medicare and Medicaid in the United States and the national health care systems in many international markets, managed care providers, private health insurers and other organizations. The trend in the health care industry is cost containment and efforts of third-party payors to contain or reduce health care costs that may adversely affect our ability to establish or maintain appropriate prices for our products or any drugs that we may develop and commercialize. In most ex-U.S. markets, pricing or profitability of therapeutic and other pharmaceutical products is subject to governmental control and such government authorities are increasingly attempting to limit or regulate the price of drug products, particularly under recent global economic pressures and geopolitical uncertainty particularly in Europe. Reimbursement agencies in Europe are often more conservative than those in the United States and the reimbursement process is often slower since reimbursement decisions are made on a country-by-country basis. In the United States, there have been, and we expect that there will continue to be, a number of federal and state proposals to implement similar governmental control as currently exists in Europe. The ACA requires discounts under the Medicare drug benefit program and increased the rebates paid by pharmaceutical companies on drugs covered by Medicaid. The ACA also imposes an annual fee, which increases annually, on sales by branded pharmaceutical manufacturers.
In addition, third-party payors attempt to contain health care costs by demanding price discounts or rebates and limiting both the types and variety of drugs that they will cover and the amounts that they will pay for drugs. As a result, they may not cover or provide adequate payment for our products. We might need to conduct post-marketing studies in order to demonstrate the cost-effectiveness of our products or any other future products to such payors’ satisfaction. Such studies might require us to commit a significant amount of management’s time and our financial and other resources. Our products might not ultimately be considered cost-effective. Adequate third-party reimbursement might not be available to enable us to maintain price levels sufficient to realize an appropriate return on our investment in product development.
28
Reimbursement rates vary according to the use of the drug and the clinical setting in which it is used, may be based on payments allowed for lower-cost products that already are reimbursed, may be incorporated into existing payments for other products or services and may reflect budgetary constraints and/or imperfections in the data used to calculate these rates. Net prices for products are reduced by mandatory discounts or rebates required by government health care programs and privately-negotiated discounts. While we have implemented policies in an effort to comply with mandated reimbursement rates, the U.S. federal government, state governments and private payors frequently pursue actions against pharmaceutical and biotechnology companies alleging that the companies have overstated prices in order to inflate reimbursement rates. Any such action could adversely affect the pricing of and revenues from our products.
In the United States and some foreign jurisdictions, there have been a number of legislative and regulatory proposals and initiatives to change the health care system in ways that could affect our ability to sell products. For example the ACA was enacted under the prior U.S. administration, and there is significant uncertainty regarding changes in the laws and regulations applicable to the health care system that may be made under the new administration, and, in particular, the effect any such changes may have on our business. Some of these proposed and implemented reforms have resulted, or could result, in reduced reimbursement rates and/or more limited access for our current or future products, which would adversely affect our business, operations and financial results.
Specialty pharmaceuticals are drugs that are prescribed by specialist physicians to treat rare or life-threatening conditions and typically address smaller patient populations. Each of ORKAMBI and KALYDECO is a specialty pharmaceutical product and our research and development programs are primarily focused on developing additional specialty pharmaceutical products. The increasing availability and use of innovative specialty pharmaceuticals, combined with their relative higher cost as compared to other types of pharmaceutical products, is beginning to generate significant third-party payor interest in developing cost-containment strategies targeted to this sector. Government regulations in both non-U.S. and U.S. markets could limit the prices that can be charged for our products and may limit our commercial opportunity. The increasing use of health technology assessments in markets around the world and the financial challenges faced by many governments may lead to significant adverse effects on our business.
Any legislation or regulatory changes or relaxation of laws that restrict imports of drugs from other countries also could reduce the net price we receive for our products.
If regulatory authorities interpret any of our conduct, including our marketing practices, as being in violation of applicable health care laws, including fraud and abuse laws, laws prohibiting off-label promotion, disclosure laws or other similar laws, we may be subject to civil or criminal penalties.
We are subject to health care fraud and abuse laws, such as the federal False Claims Act and the anti-kickback provisions of the federal Social Security Act, laws prohibiting off-label product promotion and other similar laws and regulations both in United States and in non-U.S. markets. While we have a corporate compliance program designed to actively identify, prevent and mitigate risk through the implementation of compliance policies and systems and the promotion of a culture of compliance, if we are found not to be in full compliance with these laws our business could be materially harmed.
The federal anti-kickback law prohibits knowingly and willfully offering, paying, soliciting, receiving or providing remuneration, directly or indirectly, in exchange for or to induce either the referral of an individual, or the ordering, furnishing, arranging for or recommending of an item or service that is reimbursable, in whole or in part, by a federal health care program, such as Medicare or Medicaid. The federal statute has been interpreted to apply to arrangements between pharmaceutical manufacturers on the one hand and prescribers, patients, purchasers and formulary managers on the other hand, and therefore constrains our marketing practices and our various service arrangements with physicians, including physicians who make clinical decisions to use our products. Although there are a number of statutory exemptions and regulatory safe harbors protecting certain common activities from prosecution, the exemptions and safe harbors are drawn narrowly and have been interpreted by courts as such.
Federal false claims laws prohibit any person from knowingly presenting, or causing to be presented, a false claim for payment to the federal government, or knowingly making, or causing to be made, a false statement to get a false claim paid. Pharmaceutical companies have been prosecuted under these laws for a variety of alleged promotional and marketing activities, such as providing free product to customers with the expectation that the customers would bill federal programs for the product; reporting to pricing services inflated average wholesale prices that were then used by federal programs to set reimbursement rates; engaging in promotion for uses that the FDA has not approved, known as “off-label” uses, that caused claims to be submitted to Medicaid for non-covered off-label uses; submitting inflated “best price” information to the
29
Medicaid Rebate Program; and certain manufacturing-related violations. The scope of this and other laws may expand in ways that make compliance more difficult and expensive.
Although physicians are permitted, based on their medical judgment, to prescribe products for indications other than those approved by the FDA, manufacturers are prohibited from promoting their products for such off-label uses. We market ORKAMBI and KALYDECO to eligible CF patients for whom the applicable product has been approved and provide promotional materials and training programs to physicians regarding the use of ORKAMBI and KALYDECO in these patient populations. These eligible patients represent only a portion of the total patients with CF. If the FDA determines that our promotional materials, training or other activities constitute off-label promotion, it could request that we modify our training or promotional materials or other activities, conduct corrective advertising or subject us to regulatory enforcement actions, including the issuance of a warning letter, injunction, seizure, civil fine and criminal penalties. It also is possible that other federal, state or foreign enforcement authorities might take action if they believe that the alleged improper promotion led to the submission and payment of claims for an off-label use, which could result in significant fines or penalties under other statutory authorities, such as laws prohibiting false claims for reimbursement. Even if it is later determined we were not in violation of these laws, we may be faced with negative publicity, incur significant expenses defending our actions and have to divert significant management resources from other matters.
Also applicable to some of our practices is the Health Insurance Portability and Accountability Act, or HIPAA, and its implementing regulations, which created federal criminal laws that prohibit executing a scheme to defraud any health care benefit program or making false statements relating to health care matters and which also imposes certain regulatory and contractual requirements regarding the privacy, security and transmission of individually identifiable health information. In addition, the ACA includes various provisions designed to strengthen significantly fraud and abuse enforcement, such as increased funding for enforcement efforts and the lowering of the intent requirement of the federal anti-kickback statute and criminal health care fraud statute such that a person or entity no longer needs to have actual knowledge of this statute or specific intent to violate it.
In recent years, legislation has been adopted at the federal, state and local level requiring pharmaceutical companies to establish marketing compliance programs, file periodic reports or make periodic public disclosures on sales, marketing, pricing, clinical trials, health care provider payments and other activities. For example, as part of the ACA, the federal government enacted the Open Payments (commonly known as the Sunshine Act) provisions. Open Payments requires pharmaceutical manufacturers to report annually to the Centers for Medicare and Medicaid Services payments or other transfers of value made by that entity to physicians and teaching hospitals. We also now have similar reporting obligations throughout the European Union. We expended significant efforts to establish, and are continuing to devote significant resources to maintain and enhance, systems and processes in order to comply with these regulations. Failure to comply with the reporting requirements would result in significant civil monetary penalties.
The sales and marketing practices of our industry have been the subject of increased scrutiny from governmental entities in the United Sates and other countries in which we market our products, and we believe that this trend will continue. The risk of our being found in violation of these laws is increased by the fact that many of them have not been fully interpreted by the regulatory authorities or the courts, and their provisions are subject to a variety of interpretations. If our past or present operations are found to be in violation of any such laws or any other governmental regulations that may apply to us, we may be subject to penalties, including civil and criminal penalties, damages, fines, exclusion from federal health care programs and/or the curtailment or restructuring of our operations. Any action against us for violation of these laws, even if we successfully defend against them, also could cause us to incur significant legal expenses and divert our management’s attention from the operation of our business.
Changes in laws and regulations governing the privacy and protection of data and personal information could adversely affect our business.
We are subject to data privacy and protection laws and regulations that apply to the collection, transmission, storage and use of proprietary information and personally-identifying information, including HIPAA, which among other things, imposes certain requirements relating to the privacy, security and transmission of individually identifiable health information. In addition to HIPAA, numerous other federal and state laws, including state security breach notification laws, state health information privacy laws and federal and state consumer protection laws, govern the collection, use and disclosure of personal information. Various foreign countries also have, or are developing, laws governing the collection, use and transmission of personal information. The legislative and regulatory landscape for privacy and data protection continues to evolve, and there has been an increasing amount of focus on privacy and data protection issues with the potential to affect our
30
business. We have in the past relied on adherence to the U.S.-EU Safe Harbor Framework as agreed to and set forth by the U.S. Department of Commerce and the European Union, which established a means for legitimizing the transfer of personal information by U.S. companies doing business in Europe from the European Economic Area to the United States. As a result of a 2015 opinion of the European Union Court of Justice, the U.S.-EU Safe Harbor Framework was deemed to be an invalid method of compliance with restrictions regarding the transfer of data outside of the European Economic Area. In response to the invalidation of the U.S.-EU Safe Harbor Framework, we have utilized other sanctioned approaches for transferring personal information from the European Union to the United States, such as standard contractual clauses that have been approved by the European Commission. While we continue to address the implications of changes to European Union data privacy regulations, the area remains an evolving landscape with new regulations coming into effect and continued legal challenges and our efforts to comply with the evolving data protection rules may be unsuccessful. Failure to comply with laws regarding data protection would expose us to risk of enforcement actions taken by data protection authorities in the European Union and the potential for significant penalties if we are found to be non-compliant. Even if we are not determined to have violated these laws, government investigations into these issues typically require the expenditure of significant resources and generate negative publicity, which could harm our business.
On January 1, 2015, the EMA adopted a new policy on publication of clinical data whereby it will publish clinical reports submitted as part of MAAs for drugs. The policy applies to all clinical reports submitted after January 1, 2015 and the reports will be released as soon as a decision on the application has been made by the EMA. While implementation of this policy is ongoing and its full effect on our business is not yet known, the ability of third-parties to review and/or analyze the raw data from our clinical trials may increase the risk of patient confidentiality breaches and could result in enhanced scrutiny of our clinical trials results. Such scrutiny could result in misconceptions being spread about our drugs and drug candidates, even if the underlying analysis of such review turns out to be flawed. These publications could also result in the disclosure of information to our competitors that we might otherwise deem confidential, which could harm our competitive position.
The increasing use of social media platforms presents risks and challenges.
Social media increasingly is being used by third parties to communicate about our products and drug candidates and the diseases our therapies are designed to treat. We believe that members of the CF community may be more active on social media as compared to other patient populations due to the demographics of this patient population. Social media practices in the pharmaceutical and biotechnology industries are evolving, which creates uncertainty and risk of noncompliance with regulations applicable to our business. For example, patients may use social media platforms to comment on the effectiveness of, or adverse experiences with, a drug or a drug candidate, which could result in reporting obligations. In addition, there is a risk of inappropriate disclosure of sensitive information or negative or inaccurate posts or comments about us on any social networking website. If any of these events were to occur or we otherwise fail to comply with applicable regulations, we could incur liability, face restrictive regulatory actions or incur other harm to our business.
Risks Related to Development, Clinical Testing and Regulation of Our Products and Drug Candidates
Our drug candidates remain subject to clinical testing and regulatory approval. If we are unable to successfully develop additional drug candidates, and in particular tezacaftor in combination with ivacaftor and triple combination regiments including next-generation CFTR corrector compounds, our business will be materially harmed.
Our business depends upon the successful development and commercialization of drug candidates. These drug candidates are in various stages of development and must satisfy rigorous standards of safety and efficacy before they can be approved for sale by the FDA or comparable foreign regulatory authorities. To satisfy these standards, we must allocate resources among our various development programs and must engage in expensive and lengthy testing of our drug candidates. Discovery and development efforts for new pharmaceutical products, including new combination therapies, are resource-intensive and may take 10 to 15 years or longer for each drug candidate. Despite our efforts, our drug candidates may not:
• | offer therapeutic or other improvement over existing competitive therapies; |
• | be proven safe and effective in clinical trials; |
• | meet applicable regulatory standards; |
• | be capable of being produced in commercial quantities at acceptable costs; or |
31
• | if approved for commercial sale, be successfully marketed as pharmaceutical products. |
We have recently completed and/or have ongoing or planned clinical trials for several of our drug candidates, including drug candidates for the treatment of CF, oncology, pain and neurology. The strength of our company’s product portfolio and pipeline will depend in large part upon the outcomes of these clinical trials and our ability to develop and commercialize combination treatments for CF that include ivacaftor in combination with (i) tezacaftor and/or (ii) our next-generation CFTR corrector compounds, including VX-152, VX-440, VX-659 and VX-445. Results of our clinical trials and findings from our nonclinical studies, including toxicology findings in nonclinical studies conducted concurrently with clinical trials, could lead to abrupt changes in our development activities, including the possible cessation of development activities associated with a particular drug candidate or program. Moreover, clinical data are often susceptible of varying interpretations and analyses, and many companies that have believed their drug candidates performed satisfactorily in clinical trials have nonetheless failed to obtain marketing approval of their drug candidate. Furthermore, results from our clinical trials may not meet the level of statistical significance required by the FDA or other regulatory authorities for approval of a drug candidate.
Many companies in the pharmaceutical and biotechnology industries, including our company, have suffered significant setbacks in later-stage clinical trials even after achieving promising results in earlier-stage clinical trials. Accordingly, the results from completed preclinical studies and clinical trials may not be replicated in later clinical trials, and ongoing clinical trials for our drug candidates may not be predictive of the results we may obtain in later-stage clinical trials or of the likelihood of approval of a drug candidate for commercial sale. In addition, from time to time we report interim data from our clinical trials. Interim data from a clinical trial may not be predictive of final results from the clinical trial.
If we are unable to obtain regulatory approval, we will be unable to commercialize our drug candidates.
The time required to complete clinical trials and to satisfy the FDA and other countries’ regulatory review processes is uncertain and typically takes many years. Our analysis of data obtained from nonclinical and clinical activities is subject to confirmation and interpretation by regulatory authorities, which could delay, limit or prevent regulatory approval. We also may encounter unanticipated delays or increased costs due to government regulation from future legislation or administrative action or changes in governmental policy during the period of drug development, clinical trials and governmental regulatory review.
We may seek a Fast Track and/or Breakthrough Therapy designation for some of our drug candidates. For example, a number of our drugs and drug candidates, including ivacaftor, lumacaftor, tezacaftor, VX-371, VX-440, VX-152, VX-445 and VX-220 have been granted Fast Track designation in the United States. Ivacaftor and the combination regimens of lumacaftor with ivacaftor and tezacaftor with ivacaftor were designated as Breakthrough Therapies. Drug candidates that receive one or both of these designations may be eligible for, among other things, a priority regulatory review. Each of these designations is within the discretion of the FDA. Accordingly, even if we believe one of our drug candidates meets the criteria for Fast Track and/or Breakthrough Therapy designation, the FDA may disagree and instead determine not to make such designation. The receipt of one or both of these designations for a drug candidate does not guarantee a faster development process, review or approval compared to drugs considered for approval under conventional FDA procedures and does not assure ultimate approval by the FDA. In addition, even if one or more of our drugs or drug candidates qualifies for Fast Track and/or Breakthrough Therapy designation, the FDA may later decide to withdraw such designation if it determines that the drug or drug candidate no longer meets the conditions for qualification.
Any failure to obtain regulatory approvals for a drug candidate would prevent us from commercializing that drug candidate. Any delay in obtaining required regulatory approvals could materially adversely affect our ability to successfully commercialize a drug candidate. Furthermore, any regulatory approval to market a drug may be subject to limitations that we do not expect on the indicated uses for which we may market the drug. Any such limitations could reduce the size of the market for the drug.
We also are subject to numerous foreign regulatory requirements governing the conduct of clinical trials, manufacturing and marketing authorization, pricing and third-party reimbursement. Non-U.S. jurisdictions have different approval procedures than those required by the FDA, and these jurisdictions may impose additional testing requirements for our drug candidates. The foreign regulatory approval process includes all of the risks associated with the FDA approval process described above, as well as risks attributable to the satisfaction of foreign requirements. Approval by the FDA does not ensure approval by regulatory authorities outside the United States and approval by a foreign regulatory authority does not ensure approval by the FDA. In addition, although the FDA may accept data from clinical trials conducted outside the United States, acceptance of this data is subject to conditions imposed by the FDA. For example, the clinical trial must be well
32
designed and conducted and performed by qualified investigators in accordance with ethical principles. The trial population also must adequately represent the U.S. population, and the data must be applicable to the U.S. population and U.S. medical practice in ways that the FDA deems clinically meaningful. In addition, while these clinical trials are subject to the applicable local laws, FDA acceptance of the data will depend on its determination that the trials also complied with all applicable U.S. laws and regulations. If the FDA does not accept the data from any trial that we conduct outside the United States, it would likely result in the need for additional trials, which would be costly and time-consuming and delay or permanently halt our development of the applicable drug candidate.
If clinical trials are prolonged or delayed, our development timelines for the affected development program could be extended, our costs to develop the drug candidate could increase and the competitive position of the drug candidate could be adversely affected.
We cannot predict whether or not we will encounter problems with any of our completed, ongoing or planned clinical trials that will cause us or regulatory authorities to delay or suspend clinical trials, or delay the analysis of data from our completed or ongoing clinical trials. Among the factors that could delay our development programs are:
• | ongoing discussions with the FDA or comparable foreign authorities regarding the scope or design of our clinical trials and the number of clinical trials we must conduct; |
• | delays in enrolling volunteers or patients into clinical trials, including as a result of low numbers of patients that meet the eligibility criteria for the trial; |
• | a lower than anticipated retention rate of volunteers or patients in clinical trials; |
• | the need to repeat clinical trials as a result of inconclusive results, unforeseen complications in testing or clinical investigator error; |
• | inadequate supply or deficient quality of drug candidate materials or other materials necessary for the conduct of our clinical trials; |
• | unfavorable FDA or foreign regulatory authority inspection and review of a manufacturing facility that supplied clinical trial materials or its relevant manufacturing records or a clinical trial site or records of any clinical or preclinical investigation; |
• | unfavorable scientific results from clinical trials; |
• | serious and unexpected drug-related side-effects experienced by participants in our clinical trials or by participants in clinical trials being conducted by our competitors to evaluate drug candidates with similar mechanisms of action or structures to drug candidates that we are developing; |
• | favorable results in testing of our competitors’ drug candidates, or FDA or foreign regulatory authority approval of our competitors’ drug candidates; or |
• | action by the FDA or a foreign regulatory authority to place a clinical hold or partial clinical hold on a trial or compound or deeming the clinical trial conduct as problematic. |
Our ability to enroll patients in our clinical trials in sufficient numbers and on a timely basis is subject to a number of factors, including the size of the patient population, the nature of the protocol, the proximity of patients to clinical sites, the availability of effective treatments for the relevant disease, the number of other clinical trials ongoing and competing for patients in the same indication and the eligibility criteria for the clinical trial. In addition, patients may drop out of our clinical trials or may be lost to follow-up medical evaluation after treatment ends, and this could impair the validity or statistical significance of the trials. Delays in patient enrollment or unforeseen drop-out rates may result in increased costs and longer development times.
We, our collaborators, the FDA or other applicable regulatory authorities may suspend clinical trials of a drug candidate at any time if we or they believe the healthy volunteers or patients participating in such clinical trials are being exposed to unacceptable health risks or for other reasons. Any such suspension could materially adversely affect the development of a particular drug candidate and our business.
33
If our processes and systems are not compliant with regulatory requirements, we could be subject to restrictions on marketing our products or could be delayed in submitting regulatory filings seeking approvals for our drug candidates.
We have a number of regulated processes and systems that are required to obtain and maintain regulatory approval for our drugs and drug candidates. These processes and systems are subject to continual review and periodic inspection by the FDA and other regulatory bodies. In addition, the clinical research organizations and other third parties that we work with in our non-clinical studies and clinical trials and our oversight of such parties are subject to similar reviews and periodic inspection by the FDA and other regulatory bodies. If compliance issues are identified at any point in the development and approval process, we may experience delays in filing for regulatory approval for our drug candidates, or delays in obtaining regulatory approval after filing. Any later discovery of previously unknown problems or safety issues with approved drugs or manufacturing processes, or failure to comply with regulatory requirements, may result in restrictions on such drugs or manufacturing processes, withdrawal of drugs from the market, the imposition of civil or criminal penalties or a refusal by the FDA and/or other regulatory bodies to approve pending applications for marketing approval of new drugs or supplements to approved applications, any of which could have a material adverse effect on our business. In addition, we are a party to agreements that transfer responsibility for complying with specified regulatory requirements, such as filing and maintenance of marketing authorizations and safety reporting or compliance with manufacturing requirements, to our collaborators and third-party manufacturers. If our collaborators or third-party manufacturers do not fulfill these regulatory obligations, any drugs for which we or they obtain approval may be subject to later restrictions on manufacturing or sale, which could have a material adverse effect on our business.
Risks Related to Collaborations and other Business Development Activities
Our ability to execute on our long-term strategy depends in part on our ability to acquire rights to additional drugs, drug candidates and other technologies that have the potential to add to our pipeline or provide us with new commercial opportunities.
In order to achieve our long-term business objectives, our strategy is to supplement our internal pipeline by acquiring rights to additional drugs, drug candidates and other technologies that have the potential to provide us with new commercial opportunities, including in the field of treating CF and in therapeutic areas outside of CF. We may not be able to acquire, in-license or otherwise obtain rights to additional drugs, drug candidates or other technologies on acceptable terms or at all. We have faced and will continue to face significant competition for these types of drugs, drug candidates and other technologies from a variety of other companies with interests in the specialty pharmaceutical marketplace, many of which have significantly more financial resources and experience in business development activities than we have. In addition, non-profit organizations may be willing to provide capital to the companies that control additional drugs, drug candidates or technologies, which may provide incentives for companies to advance these drugs, drug candidates or technologies independently. Because of these competitive pressures, the cost of acquiring, in-licensing or otherwise obtaining rights to such drugs, drug candidates or other technologies has grown dramatically in recent years and may be at levels that we cannot afford or that we believe are not justified by market potential. This competition is most intense for approved drugs and late-stage drug candidates, which have the lowest risk and would have the most immediate effect on our financial performance.
We may not realize the anticipated benefits of potential acquisitions or licenses to businesses, drugs, drug candidates and other technologies, and the integration following any such acquisition or license may disrupt our business and management.
We may acquire a business or the rights to drugs, drug candidates or other technologies. In recent years we have entered into numerous collaboration agreements, including our collaboration with Parion pursuant to which we exclusively licensed investigational ENaC inhibitors, including VX-371, our agreement with CRISPR to collaborate on the discovery and development of potential new treatments aimed at the underlying genetic causes of human diseases using CRISPR-Cas9 gene editing technology and our agreement with Moderna pursuant to which we are seeking to identify and develop mRNA therapeutics for the treatment of CF. With respect to each of these transactions and any additional acquisition of a business or rights to drugs, drug candidates or other technologies, we may not realize the anticipated benefits of such transaction, each of which involves numerous risks. These risks include:
• | failure to successfully further develop the acquired or licensed drugs or technology or to achieve strategic objectives, including successfully developing and commercializing the drugs, drug candidates or technologies that we acquire or license; |
34
• | inadequate or unfavorable data from clinical trials evaluating the acquired or licensed drug or drug candidates; |
• | entry into markets in which we have no or limited direct prior experience or where competitors in such markets have stronger market positions; |
• | disruption of our ongoing business and distraction of our management and employees from other opportunities and challenges; |
• | potential failure of the due diligence processes to identify significant problems, liabilities or other shortcomings or challenges of an acquired company, or acquired or licensed product or technology, including but not limited to, problems, liabilities or other shortcomings or challenges with respect to intellectual property, product quality, safety, accounting practices, employee, customer or third party relations and other known and unknown liabilities; |
• | liability for activities of the acquired company or licensor before the acquisition or license, including intellectual property infringement claims, violations of laws, commercial disputes, tax liabilities, and other known and unknown liabilities; |
• | exposure to litigation or other claims in connection with, or inheritance of claims or litigation risk as a result of an acquisition or license, including but not limited to, claims from terminated employees, customers, former equity holders or other third-parties; |
• | difficulty in integrating the drugs, drug candidates, technologies, business operations and personnel of an acquired company; and |
• | difficulties in the integration of the acquired company’s departments, systems, including accounting, human resource and other administrative systems, technologies, books and records, and procedures, as well as in maintaining uniform standards, controls, including internal control over financial reporting required by the Sarbanes-Oxley Act of 2002 and related procedures and policies. |
Acquisitions and licensing arrangements are inherently risky, and ultimately, if we do not complete an announced acquisition or license transaction or integrate an acquired business, or an acquired or licensed drug, drug candidate or other technology successfully and in a timely manner, we may not realize the benefits of the acquisition or license to the extent anticipated and the perception of the effectiveness of our management team and our company may suffer in the marketplace. Additionally, we may later incur impairment charges related to assets acquired in any such transaction. For example, we acquired or licensed several drug candidates for the treatment of HCV infection, but due to adverse clinical data regarding these drug candidates and competitive pressures, we incurred significant costs and impairment charges but did not realize the expected benefits from these transactions. In addition, even if we achieve the long-term benefits associated with strategic transactions, our expenses and short-term costs may increase materially and adversely affect our liquidity and short-term net income (loss). Future licenses or acquisitions could result in potentially dilutive issuances of equity securities, the incurrence of debt, the creation of contingent liabilities, impairment expenses related to goodwill, and impairment or amortization expenses related to other intangible assets, which could harm our financial condition.
We face risks in connection with existing and future collaborations with respect to the development, manufacture and commercialization of our products and drug candidates.
The risks that we face in connection with our current collaborations, including with Parion, CRISPR, Merck KGaA, Moderna and Janssen Inc. and any future collaborations include the following:
• | Our collaborators may change the focus of their development and commercialization efforts or may have insufficient resources to effectively develop our drug candidates. The ability of some of our products and drug candidates to reach their potential could be limited if collaborators decrease or fail to increase development or commercialization efforts related to those products or drug candidates. Our collaboration agreements provide our collaborators with a level of discretion in determining the amount and timing of efforts and resources that they will apply to these collaborations. |
• | Any future collaboration agreements may have the effect of limiting the areas of research and development that we may pursue, either alone or in collaboration with third parties. |
35
• | Collaborators may develop and commercialize, either alone or with others, drugs that are similar to or competitive with the drugs or drug candidates that are the subject of their collaborations with us. |
• | Disagreements with collaborators, including disagreements over proprietary rights, contract interpretation or the preferred course of development, might cause delays or termination of the research, development or commercialization of drug candidates, might lead to additional responsibilities for us with respect to drug candidates, or might result in litigation or arbitration. Any such disagreements would divert management attention and resources and be time-consuming and expensive. |
• | Collaborators may not properly maintain or defend our intellectual property rights or may use our proprietary information in such a way as to invite litigation that could jeopardize or invalidate our intellectual property or proprietary information or expose us to potential litigation. |
• | Collaborators may infringe the intellectual property rights of third parties, which may expose us to litigation and potential liability. |
• | Investigations and/or compliance or enforcement actions against a collaborator, which may expose us to indirect liability as a result of our partnership with such collaborator. |
• | Our collaboration agreements are subject to termination under various circumstances. |
Additionally, if a collaborator were to be involved in a business combination, it might deemphasize or terminate the development or commercialization of any drug candidate licensed to it by us. If one of our collaborators terminates its agreement with us, we may find it more difficult to attract new collaborators and our perception in the business and financial communities could be harmed.
We may not be able to attract collaborators or external funding for the development and commercialization of certain of our drug candidates.
As part of our ongoing strategy, we may seek additional collaborative arrangements or external funding for certain of our development programs and/or seek to expand existing collaborations to cover additional commercialization and/or development activities. We have a number of research programs and early-stage clinical development programs, some of which are being developed in collaboration with a third party. For example, in January 2017, we granted Merck KGaA an exclusive worldwide license to research, develop and commercialize four of our oncology research and development programs. At any time, we may determine that in order to continue development of a drug candidate or program or successfully commercialize a drug we need to identify a collaborator or amend or expand an existing collaboration. Whether we reach a definitive agreement for a collaboration will depend, among other things, upon our assessment of the collaborator’s resources and expertise, the terms and conditions of the proposed collaboration and the proposed collaborator’s evaluation of a number of factors. Those factors may include the design or results of clinical trials, the likelihood of approval by the FDA or similar regulatory authorities outside the United States, the potential market for the subject drug candidate, the costs and complexities of manufacturing and delivering such drug candidate to patients, the potential of competing products, the existence of uncertainty with respect to our ownership of the applicable intellectual property, which can exist if there is a challenge to such ownership without regard to the merits of the challenge, and industry and market conditions generally. Potentially, and depending on the circumstances, we may desire that a collaborator either agree to fund portions of a drug development program led by us, or agree to provide all of the funding and directly lead the development and commercialization of a program. No assurance can be given that any efforts we make to seek additional collaborative arrangements will be successfully completed on a timely basis or at all. If we elect to fund and undertake development or commercialization activities on our own, we may need to obtain additional expertise and additional capital, which may not be available to us on acceptable terms or at all. If we are unable to enter into acceptable collaborative relationships, one or more of our development programs could be delayed or terminated and the possibility of our receiving a return on our investment in the program could be impaired.
36
Risks Related to Third-Party Manufacturing and Reliance on Third Parties
We depend on third-party manufacturers to manufacture our products and the materials we require for our clinical trials. We may not be able to maintain these relationships and could experience supply disruptions outside of our control.
We rely on a worldwide network of third-party manufacturers and in rare circumstances, compounders, to manufacture some of our drugs for commercial use and our drug candidates for clinical trials. As a result of our reliance on these third-party manufacturers and suppliers, we could be subject to significant supply disruptions outside of our control. Our supply chain for sourcing raw materials and manufacturing drug product ready for distribution is a multi-step international endeavor. Third-party contract manufacturers, including some in China, supply us with raw materials, and convert these raw materials into drug substance and convert the drug substance into final dosage form. Establishing and managing this global supply chain requires a significant financial commitment and the creation and maintenance of numerous third-party contractual relationships. Although we attempt to manage the business relationships with companies in our supply chain, we do not have control over their operations. Supply disruptions may result from a number of factors, including shortages in product raw materials, labor or technical difficulties, regulatory inspections or restrictions, shipping or customs delays or any other performance failure by any third-party manufacturer on which we rely. Any supply disruptions could disrupt sales of our products and/or the timing of our clinical trials.
We require a supply of ivacaftor and lumacaftor for commercial sale (as KALYDECO and/or ORKAMBI). We also require a supply of our drug candidates for use in our clinical trials. We obtain ivacaftor and lumacaftor (and the combinations thereof) to meet our commercial and clinical supply needs through a third-party manufacturing network. Our supply chain includes a single-source manufacturer for (i) one step in the ivacaftor manufacturing process and (ii) the manufacture of the oral granule formulation of KALYDECO that is used for patients with CF two to five years of age. As a result, if these manufacturers become unable or unwilling to continue manufacturing product on our behalf and we are not able to promptly identify another manufacturer, we would experience a disruption in the commercial supply of ORKAMBI and/or KALYDECO, which would have a significant effect on patients, our business and our product revenues. Similarly, a disruption in the clinical supply of drug products could delay the completion of clinical trials and affect timelines for regulatory filings. There can be no assurance that we will be able to establish and maintain secondary manufacturers for all of our ivacaftor or lumacaftor supply needs on a timely basis or at all.
In the course of providing its services, a contract manufacturer may develop process technology related to the manufacture of our products or drug candidates that the manufacturer owns, either independently or jointly with us. This would increase our reliance on that manufacturer or require us to obtain a license from that manufacturer in order to have our products or drug candidates manufactured by other suppliers utilizing the same process.
We rely on third parties to conduct certain pre-clinical work and clinical trials, and those third parties may not perform satisfactorily, including failing to meet established deadlines for the completion of such studies and/or trials or failing to satisfy regulatory requirements.
We rely on third parties such as contract research organizations to help manage certain pre-clinical work and our clinical trials and on medical institutions, clinical investigators and clinical research organizations such as the Therapeutic Development Network, which is primarily funded by the CFFT, to assist in the design and review of, and to conduct our clinical trials, including enrolling qualified patients. Our reliance on these third parties for clinical development activities reduces our control over these activities but does not relieve us of our responsibilities. For example, we remain responsible for ensuring that each of our clinical trials is conducted in accordance with the general investigational plan and protocols for the clinical trial. Moreover, the FDA requires us to comply with standards, commonly referred to as good laboratory practices and good clinical practices for conducting, recording and reporting the results of pre-clinical and clinical trials to assure that data and reported results are credible and accurate and that the rights, integrity and confidentiality of trial participants are protected. If these third parties do not successfully carry out their contractual duties or meet expected deadlines, we may be required to replace them. Although we believe that there are a number of other third-party contractors we could engage to continue these activities, it may result in a delay of the affected clinical trial or drug development program. If clinical trials are not conducted in accordance with our contractual expectations or regulatory requirements, action by regulatory authorities might significantly and adversely affect the conduct or progress of these clinical trials or in specific circumstances might result in a requirement that a clinical trial be redone. Accordingly, our efforts to obtain regulatory approvals for and commercialize our drug candidates could be delayed.
37
Risks Related to Intellectual Property
If our patents do not protect our drugs or our drugs infringe third-party patents, we could be subject to litigation and substantial liabilities.
We have numerous issued patents and pending patent applications in the United States, as well as counterparts in other countries. Our success will depend, in significant part, on our ability to obtain and defend U.S. and foreign patents covering our drugs, their uses and our processes, to preserve our trade secrets and to operate without infringing the proprietary rights of third parties. We cannot be certain that any patents will issue from our pending patent applications or, even if patents issue or have issued, that the issued claims will provide us with adequate protection against competitive products or otherwise be commercially valuable.
Due to evolving legal standards relating to the patentability, validity and enforceability of patents covering pharmaceutical inventions and the scope of claims made under these patents, our ability to obtain, maintain and enforce patents is uncertain and involves complex legal and factual questions. Recent patent reform legislation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents in the U.S. The Leahy-Smith America Invents Act, or the Leahy-Smith Act, includes a number of significant changes to United States patent law. These include provisions that affect the way patent applications are prosecuted and may also affect patent litigation. The United States Patent Office developed new regulations and procedures to govern administration of the Leahy-Smith Act, and many of the substantive changes to patent law associated with the Leahy-Smith Act, and in particular, the first to file provisions, became effective in March 2013. The first to file provisions limit the rights of an inventor who is the first to invent an invention but is not the first to file an application claiming that invention. U.S. and foreign patent applications typically are maintained in confidence for a period of time after they initially are filed with the applicable patent office. Consequently, we cannot be certain that we were the first to invent, or the first to file patent applications on, our products or drug candidates or their use. If a third party also has filed a U.S. patent application relating to our drugs or drug candidates, their uses, or a similar invention, we may have to participate in legal or administrative proceedings to determine priority of invention. For applications governed by the Lahey-Smith Act, if a third-party has an earlier filed U.S. patent application relating to our drugs or drug candidates, their uses, or a similar invention, we may be unable to obtain an issued patent from our application.
The issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability. Our patents may be challenged by third parties, resulting in the patent being deemed invalid, unenforceable or narrowed in scope, or the third party may circumvent any such issued patents. Also, our pending patent applications may not issue, and we may not receive any additional patents. Our patents might not contain claims that are sufficiently broad to prevent others from utilizing our technologies. For instance, the issued patents relating to our drugs or drug candidates may be limited to a particular molecule or molecules and may not cover similar molecules that have similar clinical properties. Consequently, our competitors may independently develop competing products that do not infringe our patents or other intellectual property. In addition, if the breadth or strength of protection provided by our patents and patent applications is threatened, it could dissuade companies from collaborating with us to license, develop or commercialize current or future products.
The laws of many foreign jurisdictions do not protect intellectual property rights to the same extent as in the United States and many companies in our segment of the pharmaceutical industry have encountered significant difficulties in protecting and defending such rights in foreign jurisdictions. If we encounter such difficulties in protecting or are otherwise precluded from effectively protecting our intellectual property rights in foreign jurisdictions, our business could be substantially harmed.
Because of the extensive time required for the discovery, development, testing and regulatory review of drug candidates, it is possible that, a patent may expire before a drug candidate can be commercialized, or a patent may expire or remain in force for only a short period following commercialization of such drug candidate resulting in a minimal, if any, period of patent exclusivity. To the extent our drug candidates are not commercialized significantly ahead of the expiration date of any applicable patent, or to the extent we have no patent protection on such drug candidates, then, to the extent available we would rely on other forms of exclusivity, such as regulatory exclusivity provided by the FDCA and its counterpart agencies in various jurisdictions, and/or orphan drug exclusivity.
38
Uncertainty over intellectual property in the pharmaceutical and biotechnology industry has been the source of litigation and other disputes, which is inherently costly and unpredictable.
There is considerable uncertainty within our industry about the validity, scope and enforceability of many issued patents in the United States and elsewhere in the world, and, to date, the law and practice remains in substantial flux both in the agencies that grant patents and in the courts. We cannot currently determine the ultimate scope and validity of patents which may be granted to third parties in the future or which patents might be asserted as being infringed by the manufacture, use and sale of our products.
There has been, and we expect that there may continue to be, significant litigation in the industry regarding patents and other intellectual property rights. Litigation, arbitrations, administrative proceedings and other legal actions with private parties and governmental authorities concerning patents and other intellectual property rights may be protracted, expensive and distracting to management. Competitors may sue us as a way of delaying the introduction of our drugs or to remove our drugs from the market. Any litigation, including litigation related to Abbreviated New Drug Applications, or ANDA, litigation related to 505(b)(2) applications, interference proceedings to determine priority of inventions, derivations proceedings, inter partes review, oppositions to patents in foreign countries, litigation against our collaborators or similar actions, may be costly and time consuming and could harm our business. We expect that litigation may be necessary in some instances to determine the validity and scope of certain of our proprietary rights. Litigation may be necessary in other instances to determine the validity, scope or non-infringement of certain patent rights claimed by third parties to be pertinent to the manufacture, use or sale of our products. Ultimately, the outcome of such litigation could adversely affect the validity and scope of our patent or other proprietary rights, hinder our ability to manufacture and market our products, or result in the assessment of significant monetary damages against us that may exceed amounts, if any, accrued in our financial statements.
To the extent that valid present or future third-party patents or other intellectual property rights cover our drugs, drug candidates or technologies, we or our strategic collaborators may seek licenses or other agreements from the holders of such rights in order to avoid or settle legal claims. Such licenses may not be available on acceptable terms, which may hinder our ability to, or prevent us from being able to, manufacture and market our drugs. Payments under any licenses that we are able to obtain would reduce our profits derived from the covered products.
We may be subject to claims by third parties asserting that our employees or we have misappropriated their intellectual property, or claiming ownership of what we regard as our own intellectual property.
Many of our employees were previously employed at universities or other biotechnology or pharmaceutical companies, including our competitors or potential competitors. Although we try to ensure that our employees do not use the proprietary information or know-how of others in their work for us, we may be subject to claims that these employees or we have used or disclosed intellectual property, including trade secrets or other proprietary information, of any such employee’s former employer. Litigation may be necessary to defend against these claims.
In addition, while it is our policy to require our employees and contractors who may be involved in the development of intellectual property to execute agreements assigning such intellectual property to us, we may be unsuccessful in executing such an agreement with each party who in fact develops intellectual property that we regard as our own. Our and their assignment agreements may not be self-executing or may be breached, and we may be forced to bring claims against third parties, or defend claims they may bring against us, to determine the ownership of what we regard as our intellectual property.
If we fail in prosecuting or defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel. Even if we are successful in prosecuting or defending against such claims, litigation could result in substantial costs and be a distraction to management.
Risks Related To Our Operations
Risks associated with operating in foreign countries could materially adversely affect our business.
We have expanded our international operations over the past several years in order to market ORKAMBI and KALYDECO and expand our research and development capabilities. In 2016, a significant portion of our revenues and expenses were associated with our foreign operations and we expect that portion to increase over time as we complete reimbursement discussions in Europe for ORKAMBI. New laws and industry codes in the European Union and elsewhere have expanded transparency requirements regarding payments and transfers of value as well as patient-level clinical trial
39
data. New laws in the European Union also have expanded protections related to personal data and provided for increased sanctions for violations. Collectively, our expansion and these new requirements are adding to our compliance costs and expose us to potential sanctions for failing to meet the enhanced safeguards and reporting demands in these jurisdictions. In addition, a significant portion of our commercial supply chain, including sourcing of raw materials and manufacturing, is located in China and the European Union. Consequently, we are, and will continue to be, subject to risks related to operating in foreign countries. Risks associated with conducting operations in foreign countries include:
• | differing regulatory requirements for drug approvals and regulation of approved drugs in foreign countries; |
• | varying reimbursement regimes and difficulties or the inability to obtain reimbursement for our products in a timely manner; |
• | differing patient treatment infrastructures, particularly since our business is focused on the treatment of rare diseases that are typically prescribed by specialist physicians; |
• | collectibility of accounts receivable; |
• | changes in tariffs, trade barriers and regulatory requirements, the risks of which appear to have increased in the current political environment; |
• | economic weakness, including recession and inflation, or political instability in particular foreign economies and markets; |
• | differing levels of enforcement and/or recognition of contractual and intellectual property rights; |
• | complying with local laws and regulations, which are interpreted and enforced differently across jurisdictions and which can change significantly over time; |
• | foreign taxes, including withholding of payroll taxes; |
• | foreign currency fluctuations, which could result in reduced revenues or increased operating expenses, and other obligations incident to doing business or operating in another country; |
• | workforce uncertainty in countries where labor unrest is more common than in the United States; |
• | import and export licensing requirements, tariffs, and other trade and travel restrictions; |
• | production shortages resulting from any events affecting raw material supply or manufacturing capabilities abroad; and |
• | business interruptions resulting from geo-political actions, including war and terrorism. |
Our revenues are subject to foreign exchange rate fluctuations due to the global nature of our operations. Although we have foreign currency forward contracts to hedge forecasted product revenues denominated in foreign currencies, our efforts to reduce currency exchange losses may not be successful. As a result, currency fluctuations among our reporting currency, the U.S. dollar, and the currencies in which we do business will affect our operating results, often in unpredictable ways.
In addition, our international operations are subject to regulation under U.S. law. For example, the Foreign Corrupt Practices Act prohibits U.S. companies and their representatives from offering, promising, authorizing or making payments to foreign officials for the purpose of obtaining or retaining business abroad. In many countries, the health care professionals we regularly interact with may meet the definition of a foreign government official for purposes of the Foreign Corrupt Practices Act. We also are subject to import/export control laws. Failure to comply with domestic or foreign laws could result in various adverse consequences, including the possible delay in approval or refusal to approve a product, recalls, seizures, withdrawal of an approved product from the market, the imposition of civil or criminal sanctions, the prosecution of executives overseeing our international operations and corresponding bad publicity and negative perception of our company in foreign countries.
40
If we fail to manage our operations effectively, our business may suffer.
We have expanded and are continuing to expand our global operations and capabilities, which has placed, and will continue to place, significant demands on our management and our operational, research and development and financial infrastructure. To effectively manage our business, we need to:
• | implement and clearly communicate our corporate-wide strategies; |
• | enhance our operational and financial infrastructure, including our controls over records and information; |
• | enhance our operational, financial and management processes, including our cross-functional decision-making processes and our budget prioritization systems; |
• | train and manage our global employee base; |
• | transition from a U.S.-centric company into an organization capable of developing and commercializing multiple drug candidates in international markets; and |
• | enhance our compliance and legal resources. |
Risk Relating to the Referendum of the United Kingdom’s Membership of the European Union.
Our European headquarters and European research facility are located in the United Kingdom, and a significant portion of our ex-U.S. net product revenues are derived from sales in the United Kingdom. In June 2016, the United Kingdom, or the U.K., held a referendum in which voters approved an exit from the European Union commonly referred to as “Brexit.” It is expected that the U.K. government will provide official notice in the first half of 2017, and after notice is provided the parties will negotiate the terms of the U.K.’s withdrawal from the European Union. The withdrawal could, among other outcomes, disrupt the free movement of goods, services and people between the U.K. and the E.U., undermine bilateral cooperation in key policy areas and significantly disrupt trade between the U.K. and the E.U. In addition, Brexit could lead to legal uncertainty and potentially divergent national laws and regulations as the U.K. determines which E.U. laws to replace or replicate. Given the lack of comparable precedent, it is unclear what financial, trade, regulatory and legal implications the withdrawal of the U.K. from the E.U. would have and how such withdrawal would affect us. The announcement of Brexit caused significant volatility in global stock markets and currency exchange rate fluctuations that resulted in the strengthening of the U.S. dollar against foreign currencies in which we conduct business. The announcement of Brexit and the withdrawal of the U.K. from the E.U. may also create global economic uncertainty, which may cause third-party payors, including governmental organizations, to closely monitor their costs and reduce their spending budgets. Any of these effects of Brexit, among others, could adversely affect our business, financial condition and operating results.
Our business has a substantial risk of product liability claims and other litigation liability. If we do not obtain appropriate levels of insurance, any potential claims could adversely affect our business.
We are or may be involved in various legal proceedings, including securities class action lawsuits and claims related to product liability, intellectual property and breach of contract. Such proceedings may involve claims for, or the possibility of, fines and penalties involving substantial amounts of money or other relief, including but not limited to civil or criminal fines and penalties. If any of these legal proceedings were to result in an adverse outcome, it could have a material adverse effect on our business.
With respect to product liability and clinical trial risks, in the ordinary course of business we are subject to liability claims and lawsuits, including potential class actions, alleging that our products or drug candidates have caused, or could cause, serious adverse events or other injury. We have product liability insurance and clinical trial insurance in amounts that we believe are adequate to cover this risk. However, our insurance may not provide adequate coverage against all potential liabilities. If a claim is brought against us, we might be required to pay legal and other expenses to defend the claim, as well as pay uncovered damage awards resulting from a claim brought successfully against us and these damages could be significant and have a material adverse effect on our financial condition. Furthermore, whether or not we are ultimately successful in defending any such claims, we might be required to direct significant financial and managerial resources to such defense and adverse publicity is likely to result.
41
A breakdown or breach of our information technology systems could subject us to liability or interrupt the operation of our business.
We maintain and rely extensively on information technology systems and network infrastructures for the effective operation of our business. In the course of our business, we collect, store and transmit confidential information (including personal information and intellectual property), and it is critical that we do so in a secure manner to maintain the confidentiality and integrity of such confidential information. The size and complexity of our information technology and information security systems makes such systems potentially vulnerable to service interruptions or to security breaches. A disruption, infiltration or failure of our information technology systems or any of our data centers as a result of software or hardware malfunctions, computer viruses, cyber attacks, employee theft or misuse, power disruptions, natural disasters, floods or accidents could cause breaches of data security and loss of critical data, which in turn could materially adversely affect our business and subject us to both private and governmental causes of action. While we have implemented security measures in an attempt to minimize these risks to our data and information technology systems and have adopted a business continuity plan to deal with a disruption to our information technology systems, there can be no assurance that our efforts will prevent breakdowns or breaches in our systems that could adversely affect our business.
If we fail to attract and retain skilled employees, our business could be materially harmed.
Because our drug discovery and development activities are highly technical in nature, we require the services of highly qualified and trained scientists who have the skills necessary to conduct these activities. In addition, we need to attract and retain employees with experience in marketing and commercialization of medicines. We face intense competition for our personnel from our competitors and other companies throughout our industry. We also experience competition for the hiring of scientific and clinical personnel from universities and research institutions. Moreover, the growth of local biotechnology companies and the expansion of major pharmaceutical companies into the Boston area have increased competition for the available pool of skilled employees, especially in technical fields, and the high cost of living in Massachusetts makes it difficult to attract employees from other parts of the country to Massachusetts. In addition, the available pool of skilled employees would be further reduced if immigration laws change in a manner that increases restrictions on immigration. Our ability to commercialize our products, and achieve our research and development objectives, depends on our ability to respond effectively to these demands. If we are unable to hire and retain qualified personnel, there could be a material adverse effect on our business.
The loss of the services of key employees or the failure to effectively integrate key employees could negatively affect our business.
Our future success will depend in large part on our ability to retain the services of our key scientific and management personnel and to integrate new scientific and management personnel into our business. A loss of key personnel or a failure to properly integrate new personnel could be disruptive. We have entered into employment agreements with some executives and provide compensation-related benefits to all of our key employees that vest over time and therefore induce them to remain with us. However, the employment agreements can be terminated by the executive on relatively short notice. The value to employees of stock-related benefits that vest over time—such as options, restricted stock and restricted stock units—is significantly affected by movements in our stock price, and may at any point in time be insufficient to counteract more lucrative offers from other companies. A failure to retain, as well as hire, train and effectively integrate into our organization a sufficient number of qualified scientists, professionals, sales personnel and senior management would negatively affect our business.
If we do not comply with laws regulating the protection of the environment and health and human safety, our business could be adversely affected.
Our research and development efforts involve the regulated use of hazardous materials, chemicals and various controlled and radioactive compounds. Although we believe that our safety procedures for handling and disposing of these materials comply with the standards prescribed by state, federal and foreign regulations, the risk of loss of, or accidental contamination or injury from, these materials cannot be eliminated. If an accident occurs, we could be held liable for resulting damages, which could be substantial. We also are subject to numerous environmental, health and workplace safety laws and regulations, including those governing laboratory procedures, exposure to blood-borne pathogens and the handling of biohazardous materials. Although we maintain workers’ compensation insurance to cover us for costs we may incur due to injuries to our employees resulting from the use of these materials, this insurance may not provide adequate coverage against potential liabilities. We maintain insurance to cover pollution conditions or other extraordinary or unanticipated events
42
relating to our use and disposal of hazardous materials that we believe is appropriate based on the small amount of hazardous materials we generate. Additional federal, state and local laws and regulations affecting our operations may be adopted in the future. We may incur substantial costs to comply with, and substantial fines or penalties if we violate, any of these laws or regulations.
If our facilities were to experience a catastrophic loss, our operations would be seriously harmed.
Most of our operations, including our research and development activities, are conducted in a limited number of facilities. If any of our major facilities were to experience a catastrophic loss, due to a fire, earthquake or similar event, our operations could be seriously harmed. For example, our corporate headquarters, as well as additional leased space that we use for certain logistical and laboratory operations and manufacturing, are located in a flood zone along the Massachusetts coast. We have adopted a business continuity plan to address most crises. However, if we are unable to fully implement our disaster recovery plans, we may experience delays in recovery of data and/or an inability to perform vital corporate functions, which could result in a significant disruption in our research, development, manufacturing and/or commercial activities, the loss or critical data and/or large expenses to repair or replace the facility, which would have a material adverse effect on our business.
Risks Related to Holding Our Common Stock
Our stock price may fluctuate.
Market prices for securities of companies such as ours are highly volatile. From January 1, 2016 to December 31, 2016, our common stock traded between $71.46 and $124.96 per share. The market for our stock, like that of other companies in the biotechnology industry, has experienced significant price and volume fluctuations. The future market price of our securities could be significantly and adversely affected by factors such as:
• | the information contained in our quarterly earnings releases, including our net product revenues and operating expenses for completed periods and guidance regarding future periods; |
• | announcements of FDA actions with respect to our drugs or our competitors’ drugs, or regulatory filings for our drug candidates or those of our competitors, or announcements of interim or final results of clinical trials or nonclinical studies relating to our drugs, drug candidates or those of our competitors; |
• | prescription data and other information disclosed by third parties regarding our business or products; |
• | technological innovations or the introduction of new drugs by our competitors; |
• | government regulatory action; |
• | public concern as to the safety of drugs developed by us or our competitors; |
• | developments in patent or other intellectual property rights or announcements relating to these matters; |
• | developments in domestic and international governmental policy or regulation, for example, relating to intellectual property rights; |
• | developments relating specifically to other companies and market conditions for pharmaceutical and biotechnology stocks or stocks in general; |
• | business development, capital structuring or financing activities; and |
• | general worldwide or national economic, political and capital market conditions. |
Following periods of volatility in the market price of a company's securities, stockholder derivative lawsuits and securities class action litigation are common. Such litigation, if instituted against us or our officers and directors, could result in substantial costs and a diversion of management's attention and resources.
43
Our indebtedness could materially and adversely affect our financial condition, and the terms of our credit agreement impose restrictions on our business, reducing our operational flexibility and creating default risks.
In October 2016, we entered into a credit agreement providing for a $500 million revolving facility, $300 million of which was drawn at closing. All outstanding borrowings under the credit agreement mature on October 13, 2021. Our indebtedness could have important consequences to our business, including increasing our vulnerability to general adverse financial, business, economic and industry conditions, as well as other factors that are beyond our control. The credit agreement requires that we comply with certain financial covenants, including (i) a consolidated leverage ratio covenant and (ii) a consolidated EBITDA covenant, in each case to be measured on a quarterly basis. Further, the credit agreement includes negative covenants, subject to exceptions, restricting or limiting our ability and the ability of our subsidiaries to, among other things, incur additional indebtedness, grant liens, engage in certain investment, acquisition and disposition transactions, pay dividends, repurchase capital stock and enter into transactions with affiliates. As a result, we may be restricted from engaging in business activities that may otherwise improve our business. Failure to comply with the covenants could result in an event of default that could trigger acceleration of our indebtedness, which would require us to repay all amounts owing under the credit agreement and/or our capital leases and could have a material adverse effect on our business. Additionally, our obligations under the credit agreement are unconditionally guaranteed by certain of our domestic subsidiaries. All obligations under the credit agreement, and the guarantees of those obligations, are secured by substantially all of our assets and the assets of all guarantors (excluding intellectual property, owned and leased real property and certain other excluded property), including the pledge of all or a portion of the equity interests of certain of our subsidiaries. If we fail to satisfy our obligations under the credit agreement or are unable to obtain sufficient funds to make payments, the lenders could foreclose on our pledged collateral.
Our quarterly operating results are subject to significant fluctuation.
Our operating results have fluctuated from quarter to quarter in the past, and we expect that they will continue to do so in the future. Our revenues are primarily dependent on the level of net product revenues from sales of ORKAMBI and KALYDECO and our total net product revenues could vary on a quarterly basis. Our total net product revenues may be affected by, among other factors, the timing of orders from our significant customers. Additional factors that have caused quarterly fluctuations in recent years include variable amounts of revenues, impairment charges, charges for excess and obsolete inventories, changes in the fair value of derivative instruments and the consolidation or deconsolidation of variable interest entities. Our revenues also are subject to foreign exchange rate fluctuations due to the global nature of our operations. Although we have foreign currency forward contracts to hedge forecasted product revenues denominated in foreign currencies, our efforts to reduce currency exchange losses may not be successful. As a result, currency fluctuations among our reporting currency, the U.S. dollar, and the currencies in which we do business may affect our operating results, often in unpredictable ways. Our quarterly results also could be materially affected by significant charges, which may or may not be similar to charges we have experienced in the past. Most of our operating expenses relate to our research and development activities, do not vary directly with the amount of revenues and are difficult to adjust in the short term. As a result, if revenues in a particular quarter are below expectations, we are unlikely to reduce operating expenses proportionately for that quarter. These examples are only illustrative and other risks, including those discussed in these “Risk Factors,” could also cause fluctuations in our reported financial results. Our operating results during any one period do not necessarily suggest the results of future periods.
We expect that results from our clinical development activities and the clinical development activities of our competitors will continue to be released periodically, and may result in significant volatility in the price of our common stock.
Any new information regarding our products and drug candidates or competitive products or potentially competitive drug candidates can substantially affect investors’ perceptions regarding our future prospects. We, our collaborators and our competitors periodically provide updates regarding drug development programs, typically through press releases, conference calls and presentations at medical conferences. These periodic updates often include interim or final results from clinical trials conducted by us or our competitors and/or information about our or our competitors’ expectations regarding regulatory filings and submissions as well as future clinical development of our products or drug candidates, competitive products or potentially competitive drug candidates. The timing of the release of information by us regarding our drug development programs is often beyond our control and is influenced by the timing of receipt of data from our clinical trials and by the general preference among pharmaceutical companies to disclose clinical data during medical conferences. In addition, the information disclosed about our clinical trials, or our competitors’ clinical trials, may be based on interim rather than final data that may involve interpretation difficulties and may in any event not accurately predict final results.
44
We could be negatively affected by government investigations.
In the third quarter of 2015, we received a subpoena from the United States Department of Justice related to our marketed medicines. This subpoena requested documents relating primarily to our Good Laboratory Practices in a bioanalytical laboratory. We have responded to the subpoena. If this matter is not resolved in a satisfactory manner, our business could be adversely affected.
Changes in tax laws, regulations and treaties could affect our future taxable income.
A change in tax laws, treaties or regulations, or their interpretation, of any country in which we operate could materially affect us if we generate taxable income in a future period. We continue to assess the impact of various tax reform proposals and modifications to existing tax treaties in all jurisdictions where we have operations to determine the potential effect on our business and any assumptions we have made about our future taxable income. We cannot predict whether any specific proposals will be enacted, the terms of any such proposals or what effect, if any, such proposals would have on our business if they were to be enacted.
We may need to raise additional capital that may not be available.
We have a history of operating losses and may in the future need to raise additional capital. In recent periods, we also have received significant proceeds from the issuance of common stock under our employee benefit plans, but we received limited proceeds from employee benefit plans in 2016 and the amount and timing of future proceeds from employee benefits plans is uncertain. Any potential public offering, private placement or debt financing may or may not be similar to the transactions that we entered into in the past. Any debt financing may be on terms that, among other things, include conversion features that could result in dilution to our then-existing security holders and restrict our ability to pay interest and dividends—although we do not intend to pay dividends for the foreseeable future. Additionally, our pledge of specified assets as collateral to secure our obligations under our credit agreement may limit our ability to obtain additional debt financing. Any equity financings would result in dilution to our then-existing security holders. If adequate funds are not available on acceptable terms, or at all, we may be required to curtail significantly or discontinue one or more of our research, drug discovery or development programs, including clinical trials, incur significant cash exit costs, or attempt to obtain funds through arrangements with collaborators or others that may require us to relinquish rights to certain of our technologies, drugs or drug candidates. Based on many factors, including general economic conditions, additional financing may not be available on acceptable terms, if at all.
Issuances of additional shares of our common stock could cause the price of our common stock to decline.
As of December 31, 2016, we had 248.3 million shares of common stock issued and outstanding. As of December 31, 2016, we also had outstanding options to purchase 12.6 million shares of common stock with a weighted-average exercise price of $81.41 per share. Outstanding vested options are likely to be exercised if the market price of our common stock exceeds the applicable exercise price, and, in the future, we expect to issue additional options, restricted stock and restricted stock units to directors and employees. In addition, we may issue additional common stock or restricted securities in the future as part of financing activities or business development activities and any such issuances may have a dilutive effect on our then-existing shareholders. Sales of substantial amounts of our common stock in the open market, or the availability of such shares for sale, could adversely affect the price of our common stock. The issuance of restricted common stock or common stock upon exercise of any outstanding options would be dilutive, and may cause the market price for a share of our common stock to decline.
We have adopted anti-takeover provisions and are subject to Massachusetts corporate laws that may frustrate any attempt to remove or replace our current management or effectuate a business combination involving Vertex.
Our corporate charter and by-law provisions and Massachusetts state laws may discourage certain types of transactions involving an actual or potential change of control of Vertex that might be beneficial to us or our security holders. Our charter provides for staggered terms for the members of the Board of Directors. Our by-laws grant the directors a right to adjourn annual meetings of shareholders, and certain provisions of our by-laws may be amended only with an 80% shareholder vote. We may issue shares of any class or series of preferred stock in the future without shareholder approval and upon such terms as our Board of Directors may determine. The rights of the holders of common stock will be subject to, and may be adversely affected by, the rights of the holders of any class or series of preferred stock that may be issued in the future. Massachusetts state law prohibits us from engaging in specified business combinations, unless the combination is approved or consummated in a prescribed manner, and prohibits voting by any shareholder who acquires 20% or more of our voting
45
stock without shareholder approval. As a result, shareholders or other parties may find it more difficult to remove or replace our current management. Additionally, one of our collaboration agreements includes a change in control provision that could reduce the potential acquisition price an acquirer is willing to pay or discourage a takeover attempt that may otherwise be viewed as beneficial to shareholders.
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K and, in particular, the description of our Business set forth in Item 1, the Risk Factors set forth in this Item 1A and our Management’s Discussion and Analysis of Financial Condition and Results of Operations set forth in Item 7 contain or incorporate a number of forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended, including statements regarding:
• | our expectations regarding the amount of, timing of and trends with respect to our revenues, costs and expenses and other gains and losses, including those related to net product revenues from KALYDECO and ORKAMBI; |
• | our expectations regarding clinical trials, development timelines and regulatory authority filings and submissions for ivacaftor, lumacaftor, tezacaftor, VX-371, VX-440, VX-152, VX-659, VX-445, VX-150 and VX-210, as well as the MAA for ORKAMBI for patients with CF six to eleven years of age who are homozygous for the F508del mutation in their CFTR gene and the NDA for tezacaftor in combination with ivacaftor; |
• | our ability to obtain reimbursement for ORKAMBI in ex-U.S. markets and our ability to otherwise successfully market ORKAMBI and KALYDECO or any of our other drug candidates for which we obtain regulatory approval; |
• | our expectations regarding the timing and structure of clinical trials of our drugs and drug candidates, including ivacaftor, lumacaftor, tezacaftor, VX-371, VX-440, VX-152, VX-659, VX-445, VX-150 and VX-210, and the expected timing of our receipt of data from our ongoing and planned clinical trials; |
• | the data that will be generated by ongoing and planned clinical trials and the ability to use that data to advance compounds, continue development or support regulatory filings; |
• | our beliefs regarding the support provided by clinical trials and preclinical and nonclinical studies of our drug candidates for further investigation, clinical trials or potential use as a treatment; |
• | our plan to continue investing in our research and development programs and our strategy to develop our drug candidates, alone or with third party-collaborators; |
• | the establishment, development and maintenance of collaborative relationships; |
• | potential business development activities; |
• | potential fluctuations in foreign currency exchange rates; |
• | our ability to use our research programs to identify and develop new drug candidates to address serious diseases and significant unmet medical needs; and |
• | our liquidity and our expectations regarding the possibility of raising additional capital. |
Any or all of our forward-looking statements in this Annual Report on Form 10-K may turn out to be wrong. They can be affected by inaccurate assumptions or by known or unknown risks and uncertainties. Many factors mentioned in this Annual Report on Form 10-K will be important in determining future results. Consequently, no forward-looking statement can be guaranteed. Actual future results may vary materially from expected results. We also provide a cautionary discussion of risks and uncertainties under “Risk Factors” above in this Item 1A. These are factors and uncertainties that we think could cause our actual results to differ materially from expected results. Other factors and uncertainties besides those listed there could also adversely affect us.
46
Without limiting the foregoing, the words “believes,” “anticipates,” “plans,” “intends,” “expects” and similar expressions are intended to identify forward-looking statements. There are a number of factors and uncertainties that could cause actual events or results to differ materially from those indicated by such forward-looking statements, many of which are beyond our control, including the factors and uncertainties set forth under “Risk Factors” above in this Item 1A. In addition, the forward-looking statements contained herein represent our estimate only as of the date of this filing and should not be relied upon as representing our estimate as of any subsequent date. While we may elect to update these forward-looking statements at some point in the future, we specifically disclaim any obligation to do so to reflect actual results, changes in assumptions or changes in other factors affecting such forward-looking statements.
ITEM 1B. | UNRESOLVED STAFF COMMENTS |
We did not receive any written comments from the Securities and Exchange Commission prior to the date 180 days before the end of the fiscal year ended December 31, 2016 regarding our filings under the Securities Exchange Act of 1934, as amended, that have not been resolved.
ITEM 2. | PROPERTIES |
Corporate Headquarters
We lease approximately 1.1 million square feet of office and laboratory space at our corporate headquarters in Boston, Massachusetts in two buildings pursuant to two leases that we entered into in May 2011. The leases commenced in December 2013 and will extend until December 2028. We have an option to extend the term of the leases for an additional ten years. In addition, in connection with our relocation to Boston, we entered into a lease in June 2012 for approximately 100,000 square feet of space in the Boston Marine Industrial Park, in close proximity to our corporate headquarters. We are using this additional space for certain logistical and laboratory operations and manufacturing equipment that will complement the office and laboratory facilities at our corporate headquarters.
Facility in Cambridge, Massachusetts
We currently lease approximately 290,000 square feet of laboratory and office space at our former Kendall Square facility in Cambridge, Massachusetts that will expire in 2018. We have subleased approximately 267,000 square feet of the approximately 290,000 square feet of the Kendall Square facility under subleases, each with terms ending in 2018.
Additional United States and Worldwide Locations
In addition to our facilities in Massachusetts, we lease an aggregate of approximately 300,000 square feet of space. This includes laboratory and office space to support our research and development organizations in San Diego, California and Milton Park, Abingdon, England, London, England and office space in many of the countries in which we sell our products. In addition, in December 2015, we entered into a lease for approximately 170,000 square feet of office and laboratory space under construction in San Diego, California, which will replace our existing facility in San Diego. The lease will commence upon completion of the building, scheduled for the first half of 2018, and will extend for 16 years from the commencement date.
ITEM 3. | LEGAL PROCEEDINGS |
Local No. 8 IBEW Retirement Plan & Trust v. Vertex Pharmaceuticals Incorporated, et al.
On May 28, 2014, a purported shareholder class action Local No. 8 IBEW Retirement Plan & Trust v. Vertex Pharmaceuticals Incorporated, et al. was filed in the United States District Court for the District of Massachusetts, naming us and certain of our current and former officers and directors as defendants. The lawsuit alleged that we made material misrepresentations and/or omissions of material fact in our disclosures during the period from May 7, 2012 through May 29, 2012, all in violation of Section 10(b) of the Securities Exchange Act of 1934, as amended, and Rule 10b-5 promulgated thereunder. The purported class consisted of all persons (excluding defendants) who purchased our common stock between May 7, 2012 and May 29, 2012. The plaintiffs sought unspecified monetary damages, costs and attorneys’ fees as well as disgorgement of the proceeds from certain individual defendants’ sales of our stock. On October 8, 2014, the Court approved Local No. 8 IBEW Retirement Fund as lead plaintiff, and Scott and Scott LLP as lead counsel for the plaintiff and the putative class. On September 30, 2015, the court granted our motion to dismiss. On October 15, 2015, the plaintiff filed a notice of appeal. In 2016, the parties filed briefs with, and presented oral arguments to, the First Circuit Court of Appeals.
47
On October 3, 2016, the First Circuit Court of Appeals affirmed the district court’s dismissal of the plaintiff’s complaint. The times for petitioning the U.S. Court of Appeals for the First Circuit for an en banc rehearing as well as filing a petition for certiorari to the U.S. Supreme Court both have passed.
In the third quarter of 2015, we received a subpoena from the United States Department of Justice related to our marketed medicines. This subpoena requested documents relating primarily to our Good Laboratory Practices in a bioanalytical laboratory. We have responded to the subpoena and intend to continue to cooperate.
ITEM 4. | MINE SAFETY DISCLOSURES |
Not applicable.
48
PART II
ITEM 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES |
Market Information
Our common stock is traded on The NASDAQ Global Select Market under the symbol “VRTX.” The following table sets forth for the periods indicated the high and low sale prices per share of our common stock as reported by NASDAQ Stock Market LLC:
Year Ended December 31, 2016: | High | Low | ||||||
First quarter | $ | 124.96 | $ | 75.90 | ||||
Second quarter | 96.49 | 75.92 | ||||||
Third quarter | 103.73 | 83.50 | ||||||
Fourth quarter | 97.93 | 71.46 | ||||||
Year Ended December 31, 2015: | High | Low | ||||||
First quarter | $ | 136.33 | $ | 103.75 | ||||
Second quarter | 137.50 | 113.68 | ||||||
Third quarter | 143.45 | 97.45 | ||||||
Fourth quarter | 134.71 | 101.49 | ||||||
Shareholders
As of February 10, 2017, there were 1,629 holders of record of our common stock.
Performance Graph
CUMULATIVE TOTAL RETURN
Based on Initial Investment of $100 on December 31, 2011
with dividends reinvested (fiscal years ended December 31)
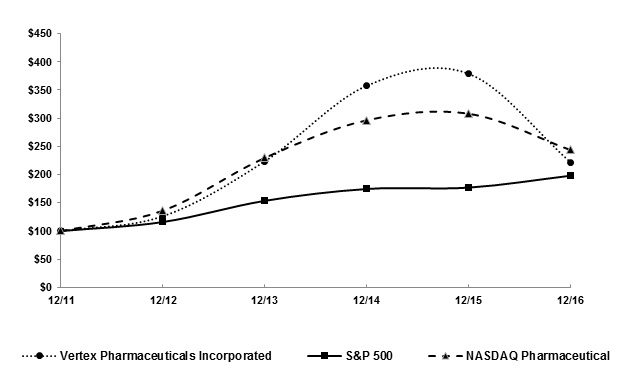
We became part of the Standard & Poor’s 500 (“S&P 500®”) Stock Index in 2013.
49
Dividends
We have never declared or paid any cash dividends on our common stock, and we currently expect that any future earnings will be retained for use in our business. Any future determination to declare cash dividends will be subject to the discretion of our board of directors and applicable law and will depend on various factors, including our results of operations, financial condition, prospects and any other factors deemed relevant by our board of directors. In addition, our credit agreement limits our ability to pay cash dividends on our common stock.
Issuer Repurchases of Equity Securities
The table set forth below shows all repurchases of securities by us during the three months ended December 31, 2016:
Period | Total Number of Shares Purchased | Average Price Paid per Share | Total Number of Shares Purchased as Part of Publicly Announced Plans or Programs | Maximum Number of Shares that May Yet be Purchased Under the Plans or Programs | ||||
Oct. 1, 2016 to Oct. 31, 2016 | 23,701 | $ | 0.01 | — | — | |||
Nov. 1, 2016 to Nov. 30, 2016 | 16,880 | $ | 0.01 | — | — | |||
Dec. 1, 2016 to Dec. 31, 2016 | 10,946 | $ | 0.01 | — | — | |||
The repurchases were made under the terms of our Amended and Restated 2006 Stock and Option Plan and Amended and Restated 2013 Stock and Option Plan. Under these plans, we award shares of restricted stock to our employees that typically are subject to a lapsing right of repurchase by us. We may exercise this right of repurchase if a restricted stock recipient’s service to us is terminated. If we exercise this right, we are required to repay the purchase price paid by or on behalf of the recipient for the repurchased restricted shares, which typically is the par value per share of $0.01. Repurchased shares are returned and are available for future awards under the terms of our Amended and Restated 2013 Stock and Option Plan.
50
ITEM 6. | SELECTED FINANCIAL DATA |
The following unaudited selected consolidated financial data are derived from our audited consolidated financial statements and have been revised to reflect discontinued operations. These data should be read in conjunction with our audited consolidated financial statements and related notes that are included elsewhere in this Annual Report on Form 10-K and with “Management’s Discussion and Analysis of Financial Condition and Results of Operations” included in Item 7.
Year Ended December 31, | |||||||||||||||||||
2016 | 2015 | 2014 | 2013 | 2012 | |||||||||||||||
(in thousands, except per share amounts) | |||||||||||||||||||
Consolidated Statements of Operations Data: | |||||||||||||||||||
Product revenues, net | |||||||||||||||||||
KALYDECO product revenues, net | $ | 703,432 | $ | 631,674 | $ | 463,750 | $ | 371,285 | $ | 171,645 | |||||||||
ORKAMBI product revenues, net | 979,590 | 350,663 | — | — | — | ||||||||||||||
INCIVEK product revenues, net | 610 | 17,987 | 24,071 | 466,360 | 1,161,813 | ||||||||||||||
Total product revenues, net | 1,683,632 | 1,000,324 | 487,821 | 837,645 | 1,333,458 | ||||||||||||||
Royalty revenues | 16,600 | 23,959 | 40,919 | 156,592 | 141,498 | ||||||||||||||
Collaborative revenues (1) | 1,945 | 8,053 | 51,675 | 217,738 | 52,086 | ||||||||||||||
Total revenues | 1,702,177 | 1,032,336 | 580,415 | 1,211,975 | 1,527,042 | ||||||||||||||
Total costs and expenses (2) | 1,692,241 | 1,499,215 | 1,272,827 | 1,821,983 | 1,480,315 | ||||||||||||||
(Loss) income from continuing operations attributable to Vertex | (112,052 | ) | (556,334 | ) | (737,643 | ) | (503,622 | ) | 32,271 | ||||||||||
(Loss) income from discontinued operations attributable to Vertex (3) | — | — | (912 | ) | 58,594 | (139,303 | ) | ||||||||||||
Net (loss) income attributable to Vertex | $ | (112,052 | ) | $ | (556,334 | ) | $ | (738,555 | ) | $ | (445,028 | ) | $ | (107,032 | ) | ||||
Diluted (loss) income from continuing operations attributable to Vertex per common share | $ | (0.46 | ) | $ | (2.31 | ) | $ | (3.14 | ) | $ | (2.24 | ) | $ | 0.15 | |||||
Shares used in per diluted share calculations | 244,685 | 241,312 | 235,307 | 224,906 | 215,262 | ||||||||||||||
As of December 31, | |||||||||||||||||||
2016 | 2015 | 2014 | 2013 | 2012 | |||||||||||||||
(in thousands) | |||||||||||||||||||
Consolidated Balance Sheet Data: | |||||||||||||||||||
Cash, cash equivalents and marketable securities | $ | 1,434,557 | $ | 1,042,462 | $ | 1,387,106 | $ | 1,465,076 | $ | 1,321,215 | |||||||||
Total assets | 2,896,787 | 2,498,587 | 2,334,679 | 2,319,041 | 2,759,288 | ||||||||||||||
Total current liabilities (4) | 792,537 | 506,167 | 368,254 | 397,829 | 432,624 | ||||||||||||||
Long-term debt obligations, excluding current portion (5) | — | 223,863 | 280,569 | — | 400,000 | ||||||||||||||
Construction financing lease obligation, excluding current portion (6) | 486,359 | 472,611 | 473,073 | 440,937 | 268,031 | ||||||||||||||
Other long-term obligations | 279,700 | 202,318 | 116,600 | 123,870 | 424,251 | ||||||||||||||
(1) | In 2013, we recorded $203.4 million of collaborative revenues from Janssen NV, which were primarily attributable to a 2013 amendment to our collaboration agreement with Janssen NV. See Note B, “Collaborative Arrangements.” |
(2) | Total costs and expenses included (i) in 2013 and 2012, an aggregate of $10.4 million and $133.2 million, respectively, of write-offs for excess and obsolete inventories, (ii) in 2013 and 2012, total costs and expenses included intangible asset impairment charges of $412.9 million and $105.8 million, respectively and (iii) in 2016, 2015, 2014 and 2013, $1.3 million, $2.2 million, $50.9 million and $40.5 million, respectively, of restructuring charges. See Note H, “Inventories,” Note J, “Intangible Assets and Goodwill” and Note Q, “Restructuring Expenses.” |
(3) | (Loss) income from discontinued operations attributable to Vertex relates to our collaboration with Alios BioPharma, Inc., in 2012 through 2013, which we deconsolidated as of December 31, 2013. See Note B, “Collaborative Arrangements.” |
(4) | In 2016, we borrowed $300.0 million pursuant to a revolving credit facility that matures in October 2021. In February 2017, we repaid the $300.0 million that was outstanding under our revolving credit facility. See Note L, “Long Term Obligations.” |
(5) | In 2013, our convertible senior subordinated notes (due 2015) with an aggregate principal amount of $400.0 million were converted into common stock or redeemed. During 2016, we terminated and repaid all outstanding obligations under our term loan. See Note L, “Long Term Obligations.” |
(6) | In 2011, we entered into two leases for our corporate headquarters, which we occupied in December 2013. We are deemed for accounting purposes to be the owner of the buildings. See Note L, “Long Term Obligations.” |
51
ITEM 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
OVERVIEW
We are in the business of discovering, developing, manufacturing and commercializing medicines for serious diseases. We use precision medicine approaches with the goal of creating transformative medicines for patients in specialty markets. Our business is focused on developing and commercializing therapies for the treatment of cystic fibrosis, or CF, and advancing our research and development programs in other indications, while maintaining our financial strength. Our two marketed products are ORKAMBI (lumacaftor in combination with ivacaftor) and KALYDECO (ivacaftor).
Our total net product revenues were $1.7 billion in 2016, an increase of 68% over net product revenues of $1.0 billion in 2015, primarily due to increased ORKAMBI net product revenues, which commenced in the third quarter of 2015, and an increase in KALYDECO net product revenues. We expect our net income (loss) and total net product revenues in 2017 will be largely dependent on the level of ORKAMBI net product revenues.
Cystic Fibrosis
ORKAMBI and KALYDECO are approved to treat approximately 40% of the 75,000 CF patients in North America, Europe, Australia and Canada. ORKAMBI (lumacaftor in combination with ivacaftor) is approved as a treatment for approximately 25,000 patients who have two copies (homozygous) of the F508del mutation in their cystic fibrosis transmembrane conductance regulator, or CFTR, gene. KALYDECO (ivacaftor) is approved for the treatment of approximately 4,000 CF patients who have the G551D mutation or other specified mutations in their CFTR gene. Our goal is to develop treatment regimens that will provide benefits to as many patients with CF as possible and will enhance the benefits that currently are being provided to patients taking our medicines.
CF Development Programs
We have multiple development programs in the field of CF, including:
• | Tezacaftor (VX-661) is a corrector compound that we are evaluating in a Phase 3 development program in combination with ivacaftor in multiple CF patient populations who have at least one copy of the F508del mutation in their CFTR gene. We expect data from this Phase 3 development program in the first half of 2017. If supported by data from the Phase 3 development program, we plan to submit a New Drug Application, or NDA, to the United States Food and Drug Administration, or FDA for tezacaftor in combination with ivacaftor in the second half of 2017. |
• | VX-152, VX-440, VX-659 and VX-445 are next-generation CFTR corrector compounds that we are evaluating as part of combination treatment regimens. We have initiated Phase 2 clinical trials of VX-152 and VX-440 and expect data from these clinical trials in the second half of 2017. We have initiated Phase 1 clinical trials of VX-659 and VX-445. |
• | VX-371, an investigational epithelial sodium channel, or ENaC, inhibitor, is being evaluated in a Phase 2 development program and which we exclusively licensed from Parion Sciences, Inc., or Parion, in 2015. |
Research and Development
We are engaged in a number of other research and mid- and early-stage development programs, including in the areas of pain and neurology. We have also entered into third-party collaborations pursuant to which we are engaged in the discovery and development of nucleic acid-based therapies for a variety of diseases, including CF. We plan to continue investing in our research programs and fostering scientific innovation in order to identify and develop transformative medicines. Our current research programs include programs targeting cystic fibrosis, adrenoleukodystrophy, alpha-1 antritrypsin deficiency, sickle cell disease and polycystic kidney disease. We believe that pursuing research in diverse areas allows us to balance the risks inherent in drug development and may provide drug candidates that will form our pipeline in future years.
52
Collaboration Arrangements
We have entered into collaborations with biotechnology and pharmaceutical companies in order to acquire rights or to license drug candidates or technologies that enhance our pipeline and/or our research capabilities. Over the last several years, we entered into collaboration agreements with:
• | CRISPR Therapeutics AG, or CRISPR, pursuant to which we are collaborating on the discovery and development of potential new treatments aimed at the underlying genetic causes of human diseases using CRISPR-Cas9 gene editing technology; |
• | Parion Sciences, Inc., or Parion, pursuant to which we are developing epithelial sodium channel, or ENaC, inhibitors for the treatment of pulmonary diseases; |
• | Moderna Therapeutics, Inc., or Moderna, pursuant to which we are seeking to identify and develop mRNA therapeutics for the treatment of CF; and |
• | BioAxone Biosciences, Inc., or BioAxone, pursuant to which we are evaluating VX-210 as a potential treatment for patients who have spinal cord injuries. |
Generally, when we in-license a technology or drug candidate, we make upfront payments to the collaborator, assume the costs of the program and agree to make contingent payments, which could consist of milestone, royalty and option payments. Depending on many factors, including the structure of the collaboration, the significance of the drug candidate that we license to the collaborator’s operations and the other activities in which our collaborators are engaged, the accounting for these transactions can vary significantly. For example, the upfront payments and expenses incurred in connection with our CRISPR and Moderna collaborations are being expensed as research expenses because the collaboration represents a small portion of these collaborators overall business. CRISPR and Moderna’s activities unrelated to our collaborations have no effect on our consolidated financial statements. Parion and BioAxone are being accounted for as variable interest entities, or VIEs, and are included in our consolidated financial statements due to (i) the significance of the respective licensed programs to Parion and BioAxone as a whole, (ii) our power to control the significant activities under each collaboration and (iii) our obligation to absorb losses and right to receive benefits that potentially could be significant. Each of our consolidated VIEs are engaging in activities unrelated to our collaboration, including in the case of Parion, seeking to develop novel treatments for pulmonary and ocular diseases. The revenues and expenses unrelated to the programs we in-license from our VIEs are immaterial to our consolidated financial statements. In each case, the activities unrelated to our collaboration represent less than 1% of our total revenues and total expenses. Because we consolidate our VIEs, we evaluate the fair value of the contingent payments payable by us on a quarterly basis. Changes in the fair value of these contingent future payments affect net income attributable to Vertex on a dollar-for-dollar basis, with increases in the fair value of contingent payments payable by us to a VIE resulting in a decrease in net income attributable to Vertex (or an increase in net loss attributable to Vertex) and decreases in the fair value of contingent payments payable by us to a VIE resulting in an increase in net income attributable to Vertex (or decrease in net loss attributable to Vertex). For additional information regarding our VIEs see Note B “Collaborative Arrangements” and our critical accounting policies “Collaborations; Variable Interest Entities.”
We have also out-licensed internally developed programs to collaborators who are leading the development of these programs. These outlicense arrangements include our collaboration agreements with:
• | Merck KGaA, pursuant to which Merck KGaA will, subject to regulatory approval, obtain rights to four oncology research and development programs; and |
• | Janssen Pharmaceuticals, Inc. which is developing JNJ-3872 (formerly VX-787) for the treatment of influenza. |
Pursuant to these outlicensing arrangements, our collaborators are responsible for the research, development and commercialization costs associated with these programs and we are entitled to receive contingent milestone and/or royalty payments. As a result, we do not expect to incur significant expenses in connection with these programs and have the potential for future collaborative and/or royalty revenues resulting from these programs.
Drug Discovery and Development
Discovery and development of a new pharmaceutical product is a difficult and lengthy process that requires significant financial resources along with extensive technical and regulatory expertise and can take 10 to 15 years or more. Potential drug candidates are subjected to rigorous evaluations, driven in part by stringent regulatory considerations, designed to
53
generate information concerning efficacy, side-effects, proper dosage levels and a variety of other physical and chemical characteristics that are important in determining whether a drug candidate should be approved for marketing as a pharmaceutical product. Most chemical compounds that are investigated as potential drug candidates never progress into development, and most drug candidates that do advance into development never receive marketing approval. Because our investments in drug candidates are subject to considerable risks, we closely monitor the results of our discovery, research, clinical trials and nonclinical studies and frequently evaluate our drug development programs in light of new data and scientific, business and commercial insights, with the objective of balancing risk and potential. This process can result in abrupt changes in focus and priorities as new information becomes available and as we gain additional understanding of our ongoing programs and potential new programs, as well as those of our competitors.
If we believe that data from a completed registration program support approval of a drug candidate, we submit an NDA to the FDA requesting approval to market the drug candidate in the United States and seek analogous approvals from comparable regulatory authorities in foreign jurisdictions. To obtain approval, we must, among other things, demonstrate with evidence gathered in nonclinical studies and well-controlled clinical trials that the drug candidate is safe and effective for the disease it is intended to treat and that the manufacturing facilities, processes and controls for the manufacture of the drug candidate are adequate. The FDA and foreign regulatory authorities have substantial discretion in deciding whether or not a drug candidate should be granted approval based on the benefits and risks of the drug candidate in the treatment of a particular disease, and could delay, limit or deny regulatory approval. If regulatory delays are significant or regulatory approval is limited or denied altogether, our financial results and the commercial prospects for the drug candidate involved will be harmed.
Regulatory Compliance
Our marketing of pharmaceutical products is subject to extensive and complex laws and regulations. We have a corporate compliance program designed to actively identify, prevent and mitigate risk through the implementation of compliance policies and systems, and through the promotion of a culture of compliance. Among other laws, regulations and standards, we are subject to various U.S. federal and state laws, and comparable foreign laws pertaining to health care fraud and abuse, including anti-kickback and false claims statutes, and laws prohibiting the promotion of drugs for unapproved or off-label uses. Anti-kickback laws make it illegal for a prescription drug manufacturer to solicit, offer, receive or pay any remuneration to induce the referral of business, including the purchase or prescription of a particular drug. False claims laws prohibit anyone from presenting for payment to third-party payors, including Medicare and Medicaid, claims for reimbursed drugs or services that are false or fraudulent, claims for items or services not provided as claimed, or claims for medically unnecessary items or services. We expect to continue to devote substantial resources to maintain, administer and expand these compliance programs globally.
Reimbursement
Sales of our products depend, to a large degree, on the extent to which our products are covered by third-party payors, such as government health programs, commercial insurance and managed health care organizations. We dedicate substantial management and other resources in order to obtain and maintain appropriate levels of reimbursement for our products from third-party payors, including governmental organizations in the United States and ex-U.S. markets. In the United States, we continue to engage in discussions with numerous commercial insurers and managed health care organizations, along with government health programs that are typically managed by authorities in the individual states. In Europe and other ex-U.S. markets, we are working to obtain government reimbursement for ORKAMBI on a country-by-country basis, because in many foreign countries patients are unable to access prescription pharmaceutical products that are not reimbursed by their governments. In the fourth quarter of 2016, we reached a pricing and reimbursement agreement for ORKAMBI with the German Federal Association of the Statutory Health Insurances. Consistent with our experience with KALYDECO when it was first approved, we expect reimbursement discussions in ex-U.S. markets may take a significant period of time.
Recent Transaction
In January 2017, we entered into a Strategic Collaboration and License Agreement with Merck KGaA, Darmstadt, Germany, or Merck KGaA. Pursuant to the agreement, we granted Merck KGaA an exclusive worldwide license to research, develop and commercialize four oncology research and development programs, including our ataxia telangiectasia and Rad3-related protein inhibitor, or ATR program, including VX-970 and VX-803, and our DNA-dependent protein kinase inhibitor, or DNA-PK program, including VX-984. Under the agreement, we expect to receive an up-front payment of $230.0 million, subject to Hart-Scott Rodino clearance, and tiered royalties on potential sales of licensed products.
54
RESULTS OF OPERATIONS
2016/2015 Comparison | 2015/2014 Comparison | ||||||||||||||||||||||||
Increase/(Decrease) | Increase/(Decrease) | ||||||||||||||||||||||||
2016 | 2015 | 2014 | $ | % | $ | % | |||||||||||||||||||
(in thousands) | (in thousands, except percentages) | ||||||||||||||||||||||||
Revenues | $ | 1,702,177 | $ | 1,032,336 | $ | 580,415 | $ | 669,841 | 65 | % | $ | 451,921 | 78 | % | |||||||||||
Operating costs and expenses | 1,692,241 | 1,499,215 | 1,272,827 | 193,026 | 13 | % | 226,388 | 18 | % | ||||||||||||||||
Other items, net | (121,988 | ) | (89,455 | ) | (45,231 | ) | $ | 32,533 | 36 | % | 44,224 | 98 | % | ||||||||||||
Loss from continuing operations attributable to Vertex | (112,052 | ) | (556,334 | ) | (737,643 | ) | (444,282 | ) | (80 | )% | (181,309 | ) | (25 | )% | |||||||||||
Loss from discontinued operations attributable to Vertex | — | — | (912 | ) | n/a | n/a | n/a | n/a | |||||||||||||||||
Net loss attributable to Vertex | $ | (112,052 | ) | $ | (556,334 | ) | $ | (738,555 | ) | $ | (444,282 | ) | (80 | )% | $ | (182,221 | ) | (25 | )% | ||||||
Net Loss Attributable to Vertex
Comparison of Net Loss Attributable to Vertex 2016 vs. 2015
Net loss attributable to Vertex was $112.1 million in 2016 as compared to a net loss attributable to Vertex of $556.3 million in 2015. Our revenues increased significantly in 2016 as compared to 2015 primarily due to a $628.9 million increase in ORKAMBI net product revenues, which we began selling in mid-2015, and a $71.8 million increase in KALYDECO net product revenues, partially offset by decreases in our royalty revenues and collaborative revenues. Our operating costs and expenses increased in 2016 as compared to 2015 primarily due to increases in cost of product revenues, research and development expenses, sales, general and administrative expenses.
The change in our other items, net was primarily due to a $54.9 million increase in the fair value of contingent payments related to our consolidated VIEs, which results in an increase in net loss attributable to Vertex.
Comparison of Net Loss Attributable to Vertex 2015 vs. 2014
Net loss attributable to Vertex was $556.3 million in 2015 as compared to a net loss attributable to Vertex of $738.6 million in 2014. Our revenues increased significantly in 2015 as compared to 2014 primarily due to ORKAMBI net product revenues, which commenced in the third quarter of 2015, and a $167.9 million increase in KALYDECO net product revenues, partially offset by a $43.6 million decrease in our collaborative revenues. Our operating costs and expenses increased in 2015 as compared to 2014 primarily due to increases in research and development expenses, sales, general and administrative expenses and cost of product revenues, partially offset by decreased restructuring expenses and royalty expenses.
Our other items, net in 2015 and 2014, included $4.5 million and $0.5 million increases in the fair value of contingent payments related to our consolidated VIEs, which resulted in corresponding increases in net loss attributable to Vertex.
Earnings Per Share
In 2016, 2015 and 2014, net loss attributable to Vertex was $0.46, $2.31 and $3.14, respectively, per diluted share. In 2016, 2015 and 2014, net loss from continuing operations attributable to Vertex was $0.46, $2.31 and $3.14, respectively, per diluted share.
Common Shares Outstanding
Our shares of outstanding common stock increased from 246.3 million shares on December 31, 2015 to 248.3 million shares on December 31, 2016 due to our issuance in 2016 of approximately 2.0 million shares of common stock pursuant to our employee equity programs. Our shares of outstanding common stock increased from 241.8 million shares on December 31, 2014 to 246.3 million shares on December 31, 2015 due to our issuance in 2015 of approximately 4.5 million shares of common stock issued pursuant to our employee equity programs.
55
Stock-based Compensation
Stock-based compensation expense was $237.7 million, $231.0 million and $177.5 million in 2016, 2015 and 2014, respectively. The increase in stock-based compensation expense has primarily been due to increases in the grant-date fair value of equity awards granted in recent years, partially offset by changes we have made in our equity compensation program.
Revenues
2016/2015 Comparison | 2015/2014 Comparison | ||||||||||||||||||||||||
Increase/(Decrease) | Increase/(Decrease) | ||||||||||||||||||||||||
2016 | 2015 | 2014 | $ | % | $ | % | |||||||||||||||||||
(in thousands) | (in thousands, except percentages) | ||||||||||||||||||||||||
Product revenues, net | $ | 1,683,632 | $ | 1,000,324 | $ | 487,821 | $ | 683,308 | 68 | % | $ | 512,503 | 105 | % | |||||||||||
Royalty revenues | 16,600 | 23,959 | 40,919 | (7,359 | ) | (31 | )% | (16,960 | ) | (41 | )% | ||||||||||||||
Collaborative revenues | 1,945 | 8,053 | 51,675 | (6,108 | ) | (76 | )% | (43,622 | ) | (84 | )% | ||||||||||||||
Total revenues | $ | 1,702,177 | $ | 1,032,336 | $ | 580,415 | $ | 669,841 | 65 | % | $ | 451,921 | 78 | % | |||||||||||
Product Revenues, Net
2016 | 2015 | 2014 | |||||||||
(in thousands) | |||||||||||
ORKAMBI | $ | 979,590 | $ | 350,663 | $ | — | |||||
KALYDECO | $ | 703,432 | $ | 631,674 | $ | 463,750 | |||||
INCIVEK | 610 | 17,987 | 24,071 | ||||||||
Total product revenues, net | $ | 1,683,632 | $ | 1,000,324 | $ | 487,821 | |||||
Our total net product revenues increased by 68% in 2016 as compared to 2015 due to increased net product revenues from ORKAMBI, which was approved by the FDA in July 2015, and increased KALYDECO net product revenues.
ORKAMBI sales commenced in mid-2015 and ORKAMBI net product revenues increased from $350.7 million in 2015 to $979.6 million in 2016. In 2015 and 2016, we recognized approximately $1.6 million and $76.4 million, respectively, in ex-U.S. ORKAMBI net product revenues, which were mainly from Germany. We also are distributing ORKAMBI in France pursuant to an early access program, but are not recognizing any revenues on ORKAMBI sales in France because the price is not fixed or determinable. Our consolidated balance sheets include $73.4 million collected as of December 31, 2016 in France related to ORKAMBI. We believe that the level of our ORKAMBI revenues during 2017 will be dependent whether, when and on what terms we are able to obtain reimbursement in additional ex-U.S. markets, the number and rate at which additional patients begin treatment with ORKAMBI, the proportion of initiated patients who remain on treatment and the compliance rates for patients who remain on treatment.
In 2016, KALYDECO net product revenues were $703.4 million, including $303.9 million of net product revenues from ex-U.S. markets, compared to KALYDECO net product revenues of $631.7 million in 2015, including $266.1 million of net product revenues from ex-U.S. markets. In 2017, we expect our KALYDECO net product revenues will be similar to our KALYDECO net product revenues in 2016. In 2014, KALYDECO net product revenues were $463.8 million, including $201.4 million of net product revenues from ex-U.S. markets. The increases were primarily due to additional patients being treated with KALYDECO as we completed reimbursement discussions in various jurisdictions and to the increased number of patients eligible to receive KALYDECO through label expansions.
INCIVEK net product revenues were $0.6 million, $18.0 million and $24.1 million in 2016, 2015 and 2014, respectively. We have withdrawn INCIVEK from the market and may continue to have small adjustments to INCIVEK revenues over the next several quarters as we adjust our INCIVEK reserves for rebates, chargebacks and discounts.
Royalty Revenues
Our royalty revenues were $16.6 million, $24.0 million and $40.9 million in 2016, 2015 and 2014, respectively. Since the beginning of 2014, our royalty revenues have consisted of (i) revenues related to a cash payment we received in 2008
56
when we sold our rights to certain HIV royalties and (ii) revenues related to certain third-party royalties payable by our collaborators on sales of HIV drugs and telaprevir that also result in corresponding royalty expenses.
Collaborative Revenues
Our collaborative revenues were $1.9 million, $8.1 million and $51.7 million in 2016, 2015 and 2014, respectively. In 2014, the majority of our collaborative revenues related to $35.0 million in payments we received from Janssen Inc. related to our outlicense of VX-787. Our collaborative revenues have historically fluctuated significantly from one period to another and may continue to fluctuate in the future. We expect our collaborative revenues to increase significantly in 2017 as a result of the $230.0 million upfront payment we expect to receive pursuant to the collaboration agreement with Merck KGaA that we entered into in January 2017.
Operating Costs and Expenses
2016/2015 Comparison | 2015/2014 Comparison | ||||||||||||||||||||||||
Increase/(Decrease) | Increase/(Decrease) | ||||||||||||||||||||||||
2016 | 2015 | 2014 | $ | % | $ | % | |||||||||||||||||||
(in thousands) | (in thousands, except percentages) | ||||||||||||||||||||||||
Cost of product revenues | $ | 206,811 | $ | 117,151 | $ | 39,725 | $ | 89,660 | 77 | % | $ | 77,426 | 195 | % | |||||||||||
Royalty expenses | 3,649 | 7,361 | 21,262 | (3,712 | ) | (50 | )% | (13,901 | ) | (65 | )% | ||||||||||||||
Research and development expenses | 1,047,690 | 995,922 | 855,506 | 51,768 | 5 | % | 140,416 | 16 | % | ||||||||||||||||
Sales, general and administrative expenses | 432,829 | 376,575 | 305,409 | 56,254 | 15 | % | 71,166 | 23 | % | ||||||||||||||||
Restructuring expenses | 1,262 | 2,206 | 50,925 | (944 | ) | (43 | )% | (48,719 | ) | (96 | )% | ||||||||||||||
Total costs and expenses | $ | 1,692,241 | $ | 1,499,215 | $ | 1,272,827 | $ | 193,026 | 13 | % | $ | 226,388 | 18 | % | |||||||||||
Cost of Product Revenues
Our cost of product revenues includes the cost of producing inventories that corresponded to product revenues for the reporting period, plus the third-party royalties payable on our net sales of our products. Pursuant to our agreement with Cystic Fibrosis Foundation Therapeutics Incorporated, or CFFT, our tiered third-party royalties on sales of KALYDECO and ORKAMBI, calculated as a percentage of net sales, range from the single digits to the sub-teens.
Our cost of product revenues have been increasing due primarily to increased net product revenues. In each of 2016 and 2015, our cost of product revenues included a $13.9 million commercial milestone that was earned by CFFT related to sales of ORKAMBI. There are no further commercial milestones payable to CFFT. In future periods, our cost of product revenues will not be affected by commercial milestones on ORKAMBI, with our cost of product revenues generally tracking our net product revenues.
Royalty Expenses
Royalty expenses include third-party royalties payable upon net sales of telaprevir by our collaborators in their territories and expenses related to a subroyalty payable to a third party on net sales of an HIV protease inhibitor sold by GlaxoSmithKline. Royalty expenses do not include royalties we pay to CFFT on sales of KALYDECO and ORKAMBI, which instead are included in cost of product revenues.
57
Research and Development Expenses
2016/2015 Comparison | 2015/2014 Comparison | ||||||||||||||||||||||||
Increase/(Decrease) | Increase/(Decrease) | ||||||||||||||||||||||||
2016 | 2015 | 2014 | $ | % | $ | % | |||||||||||||||||||
(in thousands) | (in thousands, except percentages) | ||||||||||||||||||||||||
Research expenses | $ | 314,602 | $ | 337,797 | $ | 257,483 | $ | (23,195 | ) | (7 | )% | $ | 80,314 | 31 | % | ||||||||||
Development expenses | 733,088 | 658,125 | 598,023 | 74,963 | 11 | % | 60,102 | 10 | % | ||||||||||||||||
Total research and development expenses | $ | 1,047,690 | $ | 995,922 | $ | 855,506 | $ | 51,768 | 5 | % | $ | 140,416 | 16 | % | |||||||||||
Our research and development expenses include internal and external costs incurred for research and development of our drugs and drug candidates. We do not assign our internal costs, such as salary and benefits, stock-based compensation expense, laboratory supplies and other direct expenses and infrastructure costs, to individual drugs or drug candidates, because the employees within our research and development groups typically are deployed across multiple research and development programs. These internal costs are significantly greater than our external costs, such as the costs of services provided to us by clinical research organizations and other outsourced research, which we allocate by individual program. All research and development costs for our drugs and drug candidates are expensed as incurred.
Over the past three years, we have incurred $2.9 billion in research and development expenses associated with drug discovery and development. The successful development of our drug candidates is highly uncertain and subject to a number of risks. In addition, the duration of clinical trials may vary substantially according to the type, complexity and novelty of the drug candidate and the disease indication being targeted. The FDA and comparable agencies in foreign countries impose substantial requirements on the introduction of therapeutic pharmaceutical products, typically requiring lengthy and detailed laboratory and clinical testing procedures, sampling activities and other costly and time-consuming procedures. Data obtained from nonclinical and clinical activities at any step in the testing process may be adverse and lead to discontinuation or redirection of development activities. Data obtained from these activities also are susceptible of varying interpretations, which could delay, limit or prevent regulatory approval. The duration and cost of discovery, nonclinical studies and clinical trials may vary significantly over the life of a project and are difficult to predict. Therefore, accurate and meaningful estimates of the ultimate costs to bring our drug candidates to market are not available.
In 2014, 2015 and 2016, costs related to our CF programs represented the largest portion of our development costs. Any estimates regarding development and regulatory timelines for our drug candidates are highly subjective and subject to change. We cannot make a meaningful estimate when, if ever, our other clinical development programs will generate revenues and cash flows.
Research Expenses
2016/2015 Comparison | 2015/2014 Comparison | ||||||||||||||||||||||||
Increase/(Decrease) | Increase/(Decrease) | ||||||||||||||||||||||||
2016 | 2015 | 2014 | $ | % | $ | % | |||||||||||||||||||
(in thousands) | (in thousands, except percentages) | ||||||||||||||||||||||||
Research Expenses: | |||||||||||||||||||||||||
Salary and benefits | $ | 80,845 | $ | 81,752 | $ | 82,975 | $ | (907 | ) | (1 | )% | $ | (1,223 | ) | (1 | )% | |||||||||
Stock-based compensation expense | 51,034 | 49,744 | 40,531 | 1,290 | 3 | % | 9,213 | 23 | % | ||||||||||||||||
Laboratory supplies and other direct expenses | 43,151 | 37,058 | 38,082 | 6,093 | 16 | % | (1,024 | ) | (3 | )% | |||||||||||||||
Outsourced services | 33,682 | 24,210 | 17,401 | 9,472 | 39 | % | 6,809 | 39 | % | ||||||||||||||||
Collaboration payments | 33,000 | 75,000 | — | (42,000 | ) | (56 | )% | n/a | n/a | ||||||||||||||||
Infrastructure costs | 72,890 | 70,033 | 78,494 | 2,857 | 4 | % | (8,461 | ) | (11 | )% | |||||||||||||||
Total research expenses | $ | 314,602 | $ | 337,797 | $ | 257,483 | $ | (23,195 | ) | (7 | )% | $ | 5,314 | 2 | % | ||||||||||
Over the past three years we have maintained a substantial and consistent investment in our internal research activities. Our total research expenses in 2016 and 2015 have been affected by research expenses associated with our business
58
development activities, including in 2016 a $20.0 million upfront payment to Moderna Therapeutics, Inc. and approximately $10.0 million in expenses related to the acquisition of early-stage research assets, and in 2015 a $75.0 million upfront payment we made to CRISPR Therapeutics AG, or CRISPR. We expect to continue to invest in our research programs with a focus on identifying drug candidates with the goal of creating transformative medicines.
Development Expenses
2016/2015 Comparison | 2015/2014 Comparison | ||||||||||||||||||||||||
Increase/(Decrease) | Increase/(Decrease) | ||||||||||||||||||||||||
2016 | 2015 | 2014 | $ | % | $ | % | |||||||||||||||||||
(in thousands) | (in thousands, except percentages) | ||||||||||||||||||||||||
Development Expenses: | |||||||||||||||||||||||||
Salary and benefits | $ | 177,399 | $ | 164,466 | $ | 161,718 | $ | 12,933 | 8 | % | $ | 2,748 | 2 | % | |||||||||||
Stock-based compensation expense | 102,417 | 103,211 | 76,467 | (794 | ) | (1 | )% | 26,744 | 35 | % | |||||||||||||||
Laboratory supplies and other direct expenses | 42,861 | 30,611 | 34,689 | 12,250 | 40 | % | (4,078 | ) | (12 | )% | |||||||||||||||
Outsourced services | 282,137 | 248,506 | 197,743 | 33,631 | 14 | % | 50,763 | 26 | % | ||||||||||||||||
Drug supply costs | 12,510 | 9,799 | 10,026 | 2,711 | 28 | % | (227 | ) | (2 | )% | |||||||||||||||
Infrastructure costs | 115,764 | 101,532 | 117,380 | 14,232 | 14 | % | (15,848 | ) | (14 | )% | |||||||||||||||
Total development expenses | $ | 733,088 | $ | 658,125 | $ | 598,023 | $ | 74,963 | 11 | % | $ | 60,102 | 10 | % | |||||||||||
Our development expenses increased by $75.0 million, or 11%, in 2016 as compared to 2015 and increased by $60.1 million, or 10%, in 2015 as compared to 2014. The increase in 2016 as compared to 2015 was primarily due to an increase in outsourced services related to ongoing clinical trials, including our Phase 3 development program for tezacaftor in combination with ivacaftor and increases in salary and benefits, laboratory supplies and other direct expenses and infrastructure costs. We expect our development expenses to increase in 2017 as compared to 2016 due to activities related to clinical trials, including continuation of the Phase 3 development program for tezacaftor in combination with ivacaftor as well as clinical trials associated with our next-generation correctors.
The increased development expenses in 2015 as compared to 2014 were primarily due to an increase in outsourced services related to ongoing clinical trials, including our Phase 3 development program for tezacaftor in combination with ivacaftor and an increase in stock-based compensation expense, partially offset by decreased infrastructure costs and decreased laboratory supplies and other direct expenses.
Sales, General and Administrative Expenses
2016/2015 Comparison | 2015/2014 Comparison | ||||||||||||||||||||||||
Increase/(Decrease) | Increase/(Decrease) | ||||||||||||||||||||||||
2016 | 2015 | 2014 | $ | % | $ | % | |||||||||||||||||||
(in thousands) | (in thousands, except percentages) | ||||||||||||||||||||||||
Sales, general and administrative expenses | $ | 432,829 | $ | 376,575 | $ | 305,409 | $ | 56,254 | 15 | % | $ | 71,166 | 23 | % | |||||||||||
Sales, general and administrative expenses increased by 15% in 2016 as compared to 2015, and by 23% in 2015 as compared to 2014. These increases were primarily due to increased investment in commercial support for ORKAMBI in the United States and ex-U.S. markets.
Restructuring Expense
In 2016, 2015 and 2014, we recorded restructuring expenses of $1.3 million, $2.2 million and $50.9 million, respectively. Our restructuring expenses in 2014 were primarily related to the relocation of our corporate headquarters in Massachusetts to Boston from Cambridge. As of December 31, 2016, our accrued restructuring liability related to our lease obligation in Cambridge was $8.0 million. This lease obligation expires in April 2018.
59
Other Items, Net
Interest Expense, Net
In 2016, 2015 and 2014, interest expense, net was $81.4 million, $84.2 million and $72.9 million, respectively. The decrease in interest expense, net in 2016 as compared to 2015 was primarily due to a lower interest rate on the $300.0 million of outstanding borrowings following the refinancing of our credit agreement in October 2016. The increase in interest expense, net in 2015 as compared to 2014 was primarily due to the interest expense we incurred for the full fiscal year in 2015 on the $300.0 million that we borrowed in mid-2014 pursuant to our prior credit agreement. We expect our interest expense, net in 2017 to be dependent on the amounts we borrow pursuant to our revolving credit agreement during 2017.
Other Income (Expense), Net
In 2016, net other income was $4.1 million and primarily due to foreign exchange gains. In 2015, net other expense was $6.7 million primarily due to foreign exchange losses. In 2014, we recorded net other income of $30.4 million primarily due to a credit of $36.7 million related to a one-time cash payment we received in 2014 from our landlord pursuant to leases for our corporate headquarters in Boston, Massachusetts.
Income Taxes
In 2016, we recorded a provision for income taxes of $16.7 million, principally related to income taxes payable by our VIEs. In 2015, we recorded a provision for income taxes of $30.4 million, principally due to the consolidation of Parion as a VIE into our consolidated financial statements in the second quarter of 2015. In 2014, our provision for income taxes was $7.0 million, of which approximately $3.9 million was due to the consolidation of BioAxone as a VIE into our consolidated financial statements in the fourth quarter of 2014.
Noncontrolling Interest (VIEs)
The net (income) loss attributable to noncontrolling interest (VIEs) recorded on our consolidated statements of operations reflects Parion and BioAxone's net loss (income) for the reporting period, adjusted for any changes in the noncontrolling interest holders’ claim to net assets, including contingent milestone, royalty and option payments. A summary of net (income) loss attributable to noncontrolling interest related to our VIEs for the three years ended December 31, 2016 is as follows:
2016 | 2015 | 2014 | |||||||||
(in thousands) | |||||||||||
Loss attributable to noncontrolling interest before provision for income taxes | $ | 10,086 | $ | 6,646 | $ | 764 | |||||
Provision for income taxes | 16,743 | 29,731 | 3,876 | ||||||||
Increase in fair value of contingent payments | (54,850 | ) | (4,530 | ) | (450 | ) | |||||
Net (income) loss attributable to noncontrolling interest | $ | (28,021 | ) | $ | 31,847 | $ | 4,190 | ||||
Discontinued Operations
In 2014, we recorded a loss from discontinued operations attributable to Vertex of $0.9 million. Our loss from discontinued operations in this period included losses due to the deconsolidation of Alios BioPharma, Inc, or Alios, our former collaborator, prior to the termination of our collaboration agreement with Alios in the fourth quarter of 2014.
LIQUIDITY AND CAPITAL RESOURCES
As of December 31, 2016, we had cash, cash equivalents and marketable securities of $1.43 billion, which represented an increase of $392.1 million from approximately $1.04 billion as of December 31, 2015. The increase in our cash, cash equivalents and marketable securities balance was primarily due to increased cash receipts in 2016 from product sales, partially offset by increased cash expenditures in 2016 related to, among other things, research and development expenses and sales, general and administrative expenses.
Our future cash flows will be substantially dependent on product sales of ORKAMBI and KALYDECO.
60
Sources of Liquidity
We intend to rely on our existing cash, cash equivalents and marketable securities together with cash flows from product sales as our primary source of liquidity. We are receiving cash flows from sales of ORKAMBI and KALYDECO from the United States and ex-U.S. markets. Future net product revenues for ORKAMBI from ex-U.S. markets will be dependent on, among other things, the timing of and ability to complete reimbursement discussions in European countries. Under the agreement we entered into in the first quarter of 2016 with Merck KGaA, we expect to receive an up-front payment of $230.0 million in 2017, subject to Hart-Scott Rodino clearance.
As of December 31, 2016, we had borrowed $300.0 million under a five-year $500.0 million revolving credit facility that we entered into in October 2016. We may repay and reborrow amounts under the revolving credit agreement without penalty. Subject to certain conditions, we may request that the borrowing capacity under this credit agreement be increased by an additional $300.0 million. In February 2017, we repaid the $300.0 million that we had borrowed pursuant to the revolving credit agreement plus accrued interest using existing cash resources.
In 2014 and 2015, we also received significant proceeds from the issuance of common stock under our employee benefit plans, but we received limited proceeds from employee benefit plans in 2016 and the amount and timing of future proceeds from employee benefits plans is uncertain. Other possible sources of liquidity include strategic collaborative agreements that include research and/or development funding, commercial debt, public and private offerings of our equity and debt securities, development milestones and royalties on sales of products, software and equipment leases, strategic sales of assets or businesses and financial transactions. Negative covenants in our credit agreement may prohibit or limit our ability to access these sources of liquidity.
Future Capital Requirements
We incur substantial operating expenses to conduct research and development activities and to operate our organization. Under the terms of our credit agreement entered into in October 2016, we are required to repay all outstanding principal amounts in 2021. We also have substantial facility and capital lease obligations, including leases for two buildings in Boston, Massachusetts that continue through 2028. In addition, we have entered into certain collaboration agreements with third parties that include the funding of certain research, development and commercialization efforts with the potential for future milestone and royalty payments by us upon the achievement of pre-established developmental and regulatory targets.
We expect that cash flows from ORKAMBI and KALYDECO, together with our current cash, cash equivalents and marketable securities will be sufficient to fund our operations for at least the next twelve months. The adequacy of our available funds to meet our future operating and capital requirements will depend on many factors, including the amounts of future revenues generated by ORKAMBI and KALYDECO and the potential introduction of one or more of our other drug candidates to the market, the level of our business development activities and the number, breadth, cost and prospects of our research and development programs.
Financing Strategy
We have a $500.0 million revolving credit facility that we entered into in October 2016. We may repay and reborrow amounts under the revolving credit agreement without penalty. In addition, subject to certain conditions, we may request that the borrowing capacity under this credit agreement be increased by an additional $300.0 million. We may raise additional capital through public offerings or private placements of our securities or securing new collaborative agreements or other methods of financing. We will continue to manage our capital structure and will consider all financing opportunities, whenever they may occur, that could strengthen our long-term liquidity profile. There can be no assurance that any such financing opportunities will be available on acceptable terms, if at all.
61
CONTRACTUAL COMMITMENTS AND OBLIGATIONS
The following table sets forth our commitments and obligations as of December 31, 2016:
Payments Due by Period | |||||||||||||||||||
2017 | 2018-2019 | 2020-2021 | 2022 and later | Total | |||||||||||||||
(in thousands) | |||||||||||||||||||
Fan Pier Leases | $ | 67,206 | $ | 139,795 | $ | 145,178 | $ | 535,032 | $ | 887,211 | |||||||||
Facility leases, excluding Fan Pier Leases | 36,391 | 41,568 | 39,816 | 200,626 | 318,401 | ||||||||||||||
Capital lease obligations | 21,995 | 30,171 | 5,793 | 543 | 58,502 | ||||||||||||||
Revolving credit facility | 300,000 | — | — | — | 300,000 | ||||||||||||||
Research, development and drug supply costs | 24,061 | — | — | — | 24,061 | ||||||||||||||
Other | 3,119 | 3,004 | 226 | 6,108 | 12,457 | ||||||||||||||
Total contractual commitments and obligations | $ | 452,772 | $ | 214,538 | $ | 191,013 | $ | 742,309 | $ | 1,600,632 | |||||||||
Leases
We lease two buildings that are located at Fan Pier in Boston, Massachusetts. We commenced lease payments on these two buildings in December 2013 and the initial lease periods end in December 2028.
In December 2015, we entered into a lease agreement, pursuant to which we agreed to lease approximately 170,000 square feet of office and laboratory space in a building under construction in San Diego, California. The lease will commence upon completion of the building, scheduled for the first half of 2018 and will extend for 16 years from the commencement date. The future minimum rental payments that we are obligated to pay after taking occupancy are included in “Facility leases, excluding Fan Pier Leases.”
We currently lease laboratory and office space at our former Kendall Square facility in Cambridge, Massachusetts that will expire in 2018. Our future minimum commitments under our Kendall Square lease are included in “Facility leases, excluding Fan Pier Leases.” We have entered into three subleases for a portion of the rentable square footage at the Kendall Square facility to offset our on-going contractual lease obligations. The future minimum committed income from the subleases is $15.7 million for 2017 and $5.2 million total for 2018. These amounts are not offset against our obligations set forth in the table above.
The table also reflects leases of equipment, leasehold improvements and software licenses that are accounted for as capital leases.
Revolving Credit Facility
In October 2016, we entered into a revolving credit agreement and borrowed $300.0 million under a $500.0 million revolving credit facility. Subject to certain conditions, we may request that the borrowing capacity under this credit agreement be increased by an additional $300.0 million. The outstanding loan bears interest at a rate of LIBOR plus an applicable margin ranging from 1.75% to 2.50%, based on our consolidated leverage ratio (the ratio of our total consolidated funded indebtedness to our consolidated EBITDA for the most recently completed four fiscal quarter period). The table above reflects the $300.0 million outstanding as of December 31, 2016. The above table assumes the repayment of the $300.0 million in 2017 because we repaid the outstanding amounts under the credit agreement in February 2017. We may reborrow and repay amounts under the revolving credit agreement without penalty.
Research, Development and Drug Supply Costs
Research, development and drug supply costs, does not include certain payments we are obligated to make to clinical research organizations, or CROs, because these contracts are cancelable, at our option, with notice. However, we historically have not cancelled such contracts. As of December 31, 2016, we had accrued $25.1 million related to these contracts for costs incurred for services provided through December 31, 2016, and we have approximately $175.1 million in cancelable future commitments based on existing contracts as of December 31, 2016. These amounts reflect planned expenditures based on existing contracts and do not reflect any future modifications to, or terminations of, existing contracts or anticipated or potential new contracts.
62
Collaborative Arrangements
We have entered into certain research and development collaboration agreements with third parties that include the funding of certain development, manufacturing and commercialization efforts with the potential for future milestone and royalty payments by us upon the achievement of pre-established developmental, regulatory and/or commercial targets. Our obligation to fund these efforts is contingent upon continued involvement in the programs and/or the lack of any adverse events that could cause the discontinuance of the programs. Our payment obligations under these collaborations include the following:
• | CFFT: CFFT has the right to tiered royalties ranging from single digits to sub-teens on any approved drugs first synthesized and/or tested during a research term on or before February 28, 2014, including KALYDECO, ORKAMBI, lumacaftor and tezacaftor and royalties ranging from low single digits to mid-single digits on potential sales of certain compounds first synthesized and/or tested between March 1, 2014 and August 31, 2016. |
• | Parion: Parion has the potential to receive milestone and royalty payments, including up to $490.0 million in development and regulatory milestone payments for the development of VX-371 and/or VX-551 to treat CF. |
• | CRISPR: CRISPR has the potential to receive milestone and royalty payments, including up to $420.0 million in development, regulatory and commercial milestone payments for each of up to six targets pursuant to the collaboration. |
• | Moderna: Moderna has the potential to receive milestone and royalty payments, including up to $275.0 million in development and regulatory milestones. |
• | BioAxone: BioAxone has the potential to receive milestone and royalty payments, including up to $90.0 million in development and regulatory milestone payments (including a license continuation fee). |
Contingent payments under these agreements become due and payable only upon achievement of certain milestones and are not included in the contractual obligations table above.
Tax-related Obligations
We exclude liabilities pertaining to uncertain tax positions from our summary of contractual obligations as we cannot make a reliable estimate of the period of cash settlement with the respective taxing authorities. As of December 31, 2016, we did not have any liabilities associated with uncertain tax positions. As of December 31, 2016, we cannot reasonably estimate the amount we expect to pay within the next twelve months in connection with any such settlements.
Other Funding Commitments
Our table detailing contractual commitments and obligations does not include severance payment obligations to certain of our executive officers in the event of a not-for-cause employment termination under existing employment contracts. We provide information regarding these obligations annually in our proxy statement for our annual meeting of shareholders.
CRITICAL ACCOUNTING POLICIES AND ESTIMATES
Our discussion and analysis of our financial condition and results of operations are based upon our consolidated financial statements prepared in accordance with generally accepted accounting principles in the United States. The preparation of these financial statements requires us to make certain estimates and assumptions that affect the reported amounts of assets and liabilities, the disclosure of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts of revenues and expenses during the reported periods. These items are monitored and analyzed by management for changes in facts and circumstances, and material changes in these estimates could occur in the future. Changes in estimates are reflected in reported results for the period in which the change occurs. We base our estimates on historical experience and various other assumptions that we believe to be reasonable under the circumstances. Actual results may differ from our estimates if past experience or other assumptions do not turn out to be substantially accurate.
63
We believe that our application of the following accounting policies, each of which requires significant judgments and estimates on the part of management, are the most critical to aid in fully understanding and evaluating our reported financial results:
• | revenue recognition; |
• | intangible assets; |
• | collaborations and variable interest entities; |
• | research and development accruals; |
• | commercial supplies and inventories; |
• | income taxes; |
• | leases; |
• | restructuring expenses; and |
• | stock-based compensation expense. |
Our accounting policies, including the ones discussed below, are more fully described in the Notes to our consolidated financial statements, including Note A, “Nature of Business and Accounting Policies,” included in this Annual Report on Form 10-K.
Revenue Recognition
Product Revenues, Net
We generate product revenues from sales in the United States and in international markets. We sell our products principally to a limited number of specialty pharmacy providers and selected regional wholesalers in North America as well as government-owned and supported customers in international markets, collectively, our customers. Our customers in North America subsequently resell our products to patients and health care providers. We contract with government agencies and various private organizations so that our products will be eligible for purchase by, or partial or full reimbursement from, such third-party payors. We recognize net product revenues from sales of our products upon delivery to our customers as long as:
• | there is persuasive evidence that an arrangement exists between us and our customer; |
• | collectability is reasonably assured; and |
• | the price is fixed or determinable. |
In order to conclude that the price is fixed or determinable, we must be able to calculate our gross product revenues from our customers and reasonably estimate our net product revenues upon delivery to our customers’ locations. Our gross product revenues are based on the fixed price for our products that we charge our customers. We estimate our net product revenues by deducting from our gross product revenues (i) trade allowances, such as invoice discounts for prompt payment and customer fees, (ii) estimated government and private payor rebates, chargebacks and discounts, (iii) estimated reserves for expected product returns and (iv) estimated costs of co-pay assistance programs for patients, as well as other incentives for certain indirect customers. We make significant estimates and judgments that materially affect our recognition of net product revenues. Changes in our estimates of net product revenues could have a material effect on net product revenues recorded in the period in which we determine that change occurs.
In certain instances, we may be unable to reasonably conclude that the price is fixed or determinable at the time of delivery, in which case we defer the recognition of revenues. For example, we are distributing ORKAMBI in France pursuant to early access programs but have not recognized any product revenues based on these sales because the price was not fixed or determinable due to the ongoing negotiations regarding the reimbursement rate for ORKAMBI in France. Once we are able to determine that the price is fixed or determinable, we recognize the revenues associated with the units in which revenue recognition was deferred. Our consolidated balance sheets include $73.4 million collected as of December 31, 2016 in France related to ORKAMBI.
The value of the rebates, chargebacks and discounts provided to third-party payors per course of treatment vary significantly and are based on government-mandated discounts and our arrangements with other third-party payors. In order to estimate our total rebates, chargebacks and discounts, we estimate the percentage of prescriptions that will be covered by each third-party payor, which is referred to as the payor mix. We track available information regarding changes, if any, to the
64
payor mix for our products, to our contractual terms with third-party payors and to applicable governmental programs and regulations and levels of our products in the distribution channel. We adjust our estimated rebates, chargebacks and discounts based on new information, including information regarding actual rebates, chargebacks and discounts for our products, as it becomes available. Claims by third-party payors for rebates, chargebacks and discounts frequently are submitted to us significantly after the related sales, potentially resulting in adjustments in the period in which the new information becomes known.
Our customers generally have the right to return unopened unprescribed packages subject to contractual limitations. To date returns have been minimal and, based on inventory levels held by our customers and our distribution model, we believe that returns of products will continue to be minimal. We track actual returns by individual production lots and will continue to monitor inventory levels in the distribution channel. If necessary, we will adjust our estimated product returns based on new information as it becomes available.
Collaborative Revenues
We recognize revenues generated through collaborative research, development and/or commercialization agreements. The terms of these agreements typically include payment to us of one or more of the following: nonrefundable, up-front license fees; development and commercial milestone payments; funding of research and/or development activities; payments for services we provide through our third-party manufacturing network; and royalties on net sales of licensed products. Each of these types of payments results in collaborative revenues, except for revenues from royalties on net sales of licensed products, which are classified as royalty revenues.
For each collaborative research, development and/or commercialization agreement that results in revenues, we determine (i) whether multiple deliverables exist, (ii) whether the undelivered elements have value to the customer on a stand-alone basis, (iii) how the deliverables should be separated and (iv) how the consideration should be allocated to the deliverables. We allocate consideration in an arrangement using the relative selling price method based on our best estimate of selling price of deliverables if we do not have vendor-specific objective evidence or third-party evidence. As part of the accounting for these agreements, we must develop assumptions that require judgment to determine the best estimate of selling price. We utilize key assumptions to determine the best estimate of selling price, which may include patient enrollment requirements from regulatory authorities, development timelines, reimbursement rates for personnel costs, discount rates, and estimated third-party development costs.
Intangible Assets
We maintain an indefinite-lived in-process research and development asset on our consolidated balance sheet until either the research and development project underlying it is completed or the asset becomes impaired. When we determine that an asset has become impaired or we abandon a project, we write down the carrying value of the related intangible asset to its fair value and take an impairment charge in the period in which the impairment occurs.
We assess the fair value of assets, including intangible assets such as in-process research and development assets, using a variety of methods, including present-value models that are based upon multiple probability-weighted scenarios involving the development and potential commercialization of the acquired drug candidates. The present-value models require us to make significant assumptions regarding the estimates that market participants would make in evaluating a drug candidate, including the probability of successfully completing clinical trials and obtaining regulatory approval to market the drug candidate, the timing of and the expected costs to complete in-process research and development projects, future net cash flows from potential drug sales, which are based on estimates of the sales price of the drug, the number of patients who will be diagnosed and treated and our competitive position in the marketplace, and appropriate discount and tax rates.
We test our intangible assets for impairment on an annual basis as of October 1, and more frequently if indicators are present or changes in circumstance suggest that impairment may exist. Events that could result in an impairment, or trigger an interim impairment assessment, include the receipt of additional clinical or nonclinical data regarding our drug candidate or a potentially competitive drug candidate, changes in the clinical development program for a drug candidate or new information regarding potential sales for the drug. In connection with each annual impairment assessment and any interim impairment assessment, we compare the fair value of the asset as of the date of the assessment with the carrying value of the asset on our consolidated balance sheet.
As of December 31, 2016, we had $284.3 million of indefinite-lived intangible assets recorded on our balance sheet related to Parion and BioAxone, our variable interest entities, or VIEs.
65
Collaborations; Variable Interest Entities
Our collaborations require us to apply accounting policies that involve significant judgments and that have a material effect on our consolidated financial statements. We review each collaboration agreement pursuant to which we license assets owned by a collaborator in order to determine whether we have a variable interest via the license agreement with the collaborator and if the variable interest is a variable interest in the collaborator as a whole. In connection with this assessment, we consider and make judgments regarding the following, among other factors: (1) whether the collaborator is a business; (2) the purpose and design of the collaborator; (3) the value of the asset as compared to the value of the collaborator as a whole; and (4) which party has the power to direct the activities that most significantly affect the collaborator’s economic performance. For example, in connection with the Parion collaboration we determined that (a) Parion is a business; (b) the purpose and redesign of Parion was to advance the development and commercialization of the licensed assets with a company that is able to effectively develop and commercialize products for the treatment of cystic fibrosis and other pulmonary diseases; (c) the licensed assets represented significantly more than half the value of Parion; and (d) through the joint steering committee, we have the power to direct the development and commercialization of Parion’s ENaC inhibitors, which are the activities that most significantly affect the economic performance of Parion. Based on this analysis, we consolidate Parion’s financial statements into our financial statements. Similarly, we have determined that BioAxone is a VIE that we consolidate into our financial statements.
We evaluate on a quarterly basis if we continue to have a variable interest in each VIE and are the primary beneficiary of the VIE, and if we later determine that we no longer have a variable interest or are no longer the primary beneficiary, we deconsolidate the applicable VIE. This evaluation involves an assessment of the activities being conducted pursuant to our collaboration agreement with the collaborator, the collaborator’s financial statements, discussions with the collaborator’s management regarding its other activities, including any new collaborations, financing activities, clinical data and the collaborator’s other programs.
We believe that the following effects of the consolidation and deconsolidation of VIEs on our consolidated financial statements are the most significant:
• | In each period, we record net income (loss) attributable to our VIEs noncontrolling interest. This net income (loss) reflects our VIEs net income (loss) for the period as adjusted for gains and losses in the fair value of the contingent payments, which consist of milestone, royalty and option payments, payable by us to our VIEs. Determining the fair value of the contingent payments payable by us to our VIEs requires us to make significant estimates regarding the probability and potential timing of achieving each of the milestones pursuant to the agreement, future potential net sales of licensed products and appropriate discount rates. We expect that the net income (loss) attributed to noncontrolling interest will continue to be affected by changes in the fair value of the contingent payments. In 2016, 2015 and 2014, the fair value of contingent payments payable by us increased by $54.9 million, $4.5 million and $0.5 million, respectively. The increase in fair value of the contingent payments in 2016 primarily related to a Phase 2 clinical trial of VX-371, a compound in-licensed from Parion, achieving its primary safety endpoint in the second quarter of 2016. The increases in the fair value of contingent payments increased our net loss attributable to Vertex on a dollar-for-dollar basis. |
• | We recorded $255.3 million and $29.0 million, respectively, of intangible assets on our consolidated balance sheet based on our estimate of the fair value of Parion’s and BioAxone’s in-process research and development assets as of the transaction date and made significant estimates regarding: the probability of obtaining regulatory approval of licensed products; the timing and expected costs of clinical trials and other development activities; future potential cash flows from sales of drugs and the appropriate discount rates. If we are successful in developing a drug candidate, we will amortize the carrying value of the relevant intangible asset as part of cost of product revenues. We test these in-process research and development assets for impairment on an annual basis as of October 1, and more frequently if indicators are present or changes in circumstances suggest that impairment may exist. If the fair value of a licensed program becomes impaired as the result of safety or efficacy data from any ongoing or future clinical trial conducted by us or our competitors or because of any other information regarding the prospects of successfully developing or commercializing the licensed drug candidate, we could incur significant charges in the period in which the impairment occurs. We determined the fair value of these in-process research and development assets using probability-weighted present-value models. |
66
• | The revenues and expenses of our VIEs that are unrelated to the programs that we in-license from our VIEs and that are consolidated into our financial statements are set forth in the table below and represent less than 1% of our revenues and expenses in each period: |
2016 | 2015 | 2014 | |||||||||
(in thousands) | |||||||||||
Revenues | $ | 944 | $ | 2,888 | $ | — | |||||
Research and development expenses | (6,762 | ) | (3,642 | ) | (286 | ) | |||||
Sales, general and administrative expenses | (4,160 | ) | (5,836 | ) | (491 | ) | |||||
Other (expenses) income, net | (108 | ) | (56 | ) | 13 | ||||||
Loss attributable to noncontrolling interest before provision for income taxes | $ | (10,086 | ) | $ | (6,646 | ) | $ | (764 | ) | ||
To the extent that our VIEs pursue other programs, expenses related to those activities would be reflected in our research and development expenses and our sales, general and administrative expenses as a result of the financial statement consolidation. We would not be entitled to any benefits from those activities. In future periods, our VIEs could increase their operating expenses related to other activities and any such increases would affect our operating expenses as presented in our consolidated financial statements.
• | We reflect all of our VIEs' cash and cash equivalents under the heading “restricted cash and cash equivalents (VIE)” on our consolidated balance sheets. We do not have any rights to our VIEs cash or cash equivalents, these resources are not available to fund research and development programs pursuant to the collaborations and these amounts do not provide us with any additional liquidity. Our VIEs have control over the restricted cash and cash equivalents (VIE), including the ability to distribute the restricted cash and cash equivalents to their equity holders, and as a result, these assets, although carried on our consolidated balance sheets, are not included in the discussion of our liquidity and should be disregarded when evaluating our financial condition. |
In order to account for the fair value of the intangible assets and contingent payments related to collaborations with our VIEs under GAAP, we use present-value models based on assumptions regarding the probability of achieving the relevant milestones, estimates regarding the timing of achieving the milestones, estimates of future product sales and the appropriate discount rates. We base our estimates of the probability of achieving the relevant milestones on industry data for similar assets and our own experience. The discount rates used in the valuation model represent a measure of credit risk and market risk associated with settling the liabilities. Significant judgment is used in determining the appropriateness of these assumptions during each reporting period. Changes in these assumptions could have a material effect on the fair value of the contingent payments and affect the analysis of whether or not an intangible asset is impaired.
Research and Development Accruals
Research and development expenses, including amounts funded through research and development collaborations, are expensed as incurred. When third-party service providers’ billing terms do not coincide with our period-end, we are required to make estimates of our obligations to those third parties, including clinical trial and pharmaceutical development costs, contractual services costs, costs for drug supply, marketing expenses and infrastructure expenses incurred in a given accounting period and record accruals at the end of the period. We base our estimates on our knowledge of the research and development programs, services performed for the period, experience with related activities and the expected duration of the third-party service contract, where applicable.
Commercial Supplies and Inventories
We began capitalizing the costs of our ORKAMBI inventories on July 1, 2014. We capitalize inventories produced in preparation for initiating sales of a drug candidate when the related drug candidate is considered to have a high likelihood of regulatory approval and the related costs are expected to be recoverable through sale of the inventories. In determining whether or not to capitalize such inventories, we evaluate, among other factors, information regarding the drug candidate’s safety and efficacy, the status of regulatory submissions and communications with regulatory authorities and the outlook for commercial sales, including the existence of current or anticipated competitive drugs and the availability of reimbursement. In addition, we evaluate risks associated with manufacturing the drug candidate and the remaining shelf life of the inventories. After we begin capitalizing inventories, we perform an assessment of the recoverability of capitalized inventory during each reporting period, and write down any excess and obsolete inventories to their net realizable value in the period in which the impairment is first identified. Periodic assessments of the recoverability of capitalized costs involve significant estimates and judgments on the part of management.
As of December 31, 2016, all of our inventories are related to KALYDECO and ORKAMBI.
67
Income Taxes
We maintain a valuation allowance on the majority of our net operating losses and other deferred tax assets because we have an extended history of annual losses. Our U.S. federal net operating loss carryforwards totaled approximately $4.1 billion as of December 31, 2016. On an annual basis, we reassess the valuation allowance for deferred income tax assets. After consideration of all the evidence, both positive and negative, we continue to maintain a valuation allowance on the deferred tax asset as of December 31, 2016 because it is more likely than not that the deferred tax asset will not be realized. In future periods, if we determine that it is more likely than not that the deferred tax asset will be realized, (i) the valuation allowance would be decreased, (ii) the deferred tax asset would be reflected on our consolidated balance sheet and (iii) we would record non-cash benefits in our consolidated statements of operations related to the reflection of the deferred tax asset on our consolidated balance sheet.
Leases
In 2011, we entered into two leases for our corporate headquarters. Our corporate headquarters were built during the period from 2011 through December 2013. We lease our corporate headquarters pursuant to leases that expire in 2028, subject to our right to extend the leases for an additional 10 years. Because we were involved in the construction project, we were deemed for accounting purposes to be the owner of the buildings during the construction period. Accordingly, we record project construction costs incurred by the landlord as an asset and a related financing obligation in “Property and equipment, net” and “Construction financing lease obligation,” respectively, on our consolidated balance sheets.
Upon completion of the construction of the corporate headquarters buildings, we evaluated the leases and determined that the leases did not meet the criteria for “sale-leaseback” treatment. Accordingly, we depreciate the asset and incur interest expense related to the financing obligation recorded on our balance sheet. We bifurcate our lease payments pursuant to the leases into (i) a portion that is allocated to the buildings and (ii) a portion that is allocated to the land on which the buildings were constructed. The portion of the lease obligations allocated to the land is treated as an operating lease. In connection with the leases for our corporate headquarters, we incurred $60.2 million in interest expense, $13.3 million in depreciation expense and $6.5 million in operating expense in 2016. In 2017, we expect interest expense, depreciation expense and operating expenses related to the leases for our corporate headquarters to be approximately consistent with that from 2016.
In 2015, we entered into a lease agreement for a research and development facility to be built in San Diego. Because we are involved in the construction project, we are deemed for accounting purposes to be the owner of the building during the construction period and are recording project construction costs incurred by the landlord. We will need to evaluate this lease based on “sale-leaseback” criteria upon completion of the construction. We currently expect this lease will not meet the criteria and will be accounted for in the same manner as we have accounted for the leases for our corporate headquarters.
Restructuring Expenses
We have adopted several plans to restructure our facility operations for which we have incurred restructuring expenses in the three years ended December 31, 2016. In particular, in 2014, we recorded $50.9 million in costs associated with exit and disposal activities related to the relocation of our headquarters in Massachusetts from Cambridge to Boston and maintained a liability related to these activities of $3.6 million as of December 31, 2016. Our initial estimate of our liabilities for net ongoing costs associated with these facility obligations are recorded at fair value. In estimating the expenses and liabilities related to these facilities, we utilize probability-weighted discounted cash-flows of our ongoing lease obligations. In estimating the expense and liability under our lease obligations, we estimate (i) the costs to be incurred to satisfy rental and build-out commitments under the lease (including operating costs), (ii) the lead-time necessary to sublease the space, (iii) the projected sublease rental rates and (iv) the anticipated durations of subleases. We use a credit-adjusted risk-free rate to discount the estimated cash flows.
We review our estimates and assumptions on at least a quarterly basis. We intend to continue such reviews until the termination of these facility lease obligations and will make whatever modifications we believe are necessary, based on our best judgment, to reflect any changed circumstances. Our estimates have changed in the past, and may change in the future, resulting in additional adjustments to the estimate of these liabilities. Changes to our estimate of these liabilities are recorded as additional restructuring expenses (credits). In addition, because our estimate of these liabilities includes the application of a discount rate to reflect the time-value of money, we record imputed interest costs related to these liabilities each quarter. These costs are included in restructuring expenses on our consolidated statements of operations.
Stock-based Compensation Expense
Stock-based compensation expense is determined based on the fair value of the equity award at the grant date, net of estimated forfeitures, and is adjusted each period to reflect actual forfeitures and the outcomes of certain performance
68
conditions. For awards with performance conditions that accelerate vesting of the award, we estimate the likelihood of satisfaction of the performance conditions, which affects the period over which the expense is recognized, and recognize the expense using the accelerated attribution model. For awards with performance conditions in which the award does not vest unless the performance condition is met, we recognize expense only if we estimate that achievement of the performance condition is probable. If we conclude that vesting is probable, we recognize expense from the date that we reach this conclusion through the estimated vesting date. During 2016, we also granted awards with a variable number of awards. Threshold, target and maximum parameters were established for the metric based half on financial and half on non-financial goals, and will be used to calculate the number of shares that will be issuable when the award vests, which may range from zero to 200% of the target amount. Since 2014, we have provided to employees who have rendered a certain number of years of service and meet certain age requirements, partial or full acceleration of vesting of their equity awards, subject to certain conditions including a notification period, upon a termination of employment other than for cause. If actual forfeitures differ significantly from our estimates, if our estimates regarding the employees who will be eligible for partial or full acceleration of their equity awards, if the likelihood of achievement of a performance conditions changes or if any of our other assumptions or estimates prove incorrect, our stock-based compensation expense, or the period over which our stock-based compensation is recognized, could be materially affected.
RECENT ACCOUNTING PRONOUNCEMENTS
Refer to Note A, “Nature of Business and Accounting Policies,” in the accompanying notes to the consolidated financial statements for a discussion of recent accounting pronouncements. There were no new accounting pronouncements adopted during 2016 that had a material effect on our financial statements.
ITEM 7A. | QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK |
As part of our investment portfolio, we own financial instruments that are sensitive to market risks. The investment portfolio is used to preserve our capital until it is required to fund operations, including our research and development activities. None of these market risk-sensitive instruments is held for trading purposes. We do not have derivative financial instruments in our investment portfolio.
Interest Rate Risk
We invest our cash in a variety of financial instruments, principally securities issued by the U.S. government and its agencies, investment-grade corporate bonds and commercial paper, and money market funds. These investments are denominated in U.S. dollars. All of our interest-bearing securities are subject to interest rate risk and could decline in value if interest rates fluctuate. Substantially all of our investment portfolio consists of marketable securities with active secondary or resale markets to help ensure portfolio liquidity, and we have implemented guidelines limiting the term-to-maturity of our investment instruments. Due to the conservative nature of these instruments, we do not believe that we have a material exposure to interest rate risk. If interest rates were to increase or decrease by 1%, the fair value of our investment portfolio would increase or decrease by an immaterial amount.
In October 2016, we entered into a credit agreement. Loans under the credit agreement will bear interest, at our option, at either a base rate or a Eurodollar rate, in each case plus an applicable margin. The applicable margin on base rate loans ranges from 0.75% to 1.50% and the applicable margin on Eurodollar loans ranges from 1.75% to 2.50%, in each case, based on our consolidated leverage ratio (as defined in the credit agreement). We do not believe that changes in interest rates related to the credit agreement would have a material effect on our financial statements. As of December 31, 2016, we had approximately $300.7 million of principle and interest outstanding. If interest rates were to increase or decrease by 1% for the year, annual interest expense would increase or decrease by approximately $3.0 million.
Foreign Exchange Market Risk
As a result of our foreign operations, we face exposure to movements in foreign currency exchange rates, primarily the Euro, Swiss Franc, British Pound, Australian Dollar and Canadian Dollar against the U.S. dollar. The current exposures arise primarily from cash, accounts receivable, intercompany receivables, payables and inventories. Both positive and negative affects to our net revenues from international product sales from movements in foreign currency exchange rates are partially mitigated by the natural, opposite affect that foreign currency exchange rates have on our international operating costs and expenses.
We have a foreign currency management program with the objective of reducing the effect of exchange rate fluctuations on our operating results and forecasted revenues and expenses denominated in foreign currencies. We currently have hedges for Euro, British Pound and Australian Dollar. These cash flow hedges qualify for hedge accounting. As of December 31,
69
2016, we held foreign exchange forward contracts with notional amounts totaling $253.4 million. As of December 31, 2016, our outstanding foreign exchange forward contracts had a net fair value of $15.4 million.
Based on our foreign currency exchange rate exposures at December 31, 2016, a hypothetical 10% adverse fluctuation in exchange rates would decrease the fair value of our foreign exchange forward contracts that are designated as cash flow hedges by approximately $25.3 million at December 31, 2016. The resulting loss on these forward contracts would be offset by the gain on the underlying transactions and therefore would have minimal impact on future anticipated earnings and cash flows. Similarly, adverse fluctuations in exchange rates that would decrease the fair value of our foreign exchange forward contracts that are not designated as hedge instruments would be offset by a positive impact of the underlying monetary assets and liabilities.
ITEM 8. | FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA |
The information required by this Item 8 is contained on pages F-1 through F-50 of this Annual Report on Form 10-K.
ITEM 9. | CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE |
Not applicable.
ITEM 9A. | CONTROLS AND PROCEDURES |
(1) Evaluation of Disclosure Controls and Procedures. The Company’s chief executive officer and chief financial officer, after evaluating the effectiveness of the Company’s disclosure controls and procedures (as defined in Rule 13a-15(e) and Rule 15d-15(e) promulgated under the Securities Exchange Act of 1934, as amended) as of the end of the period covered by this Annual Report on Form 10-K, have concluded that, based on such evaluation, the Company’s disclosure controls and procedures were effective. In designing and evaluating the disclosure controls and procedures, the Company’s management recognized that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired control objectives, and the Company’s management necessarily was required to apply its judgment in evaluating the cost-benefit relationship of possible controls and procedures.
(2) Management’s Annual Report on Internal Control Over Financial Reporting. The management of the Company is responsible for establishing and maintaining adequate internal control over financial reporting. Internal control over financial reporting is defined in Rule 13a-15(f) and Rule 15d-15(f) promulgated under the Securities Exchange Act of 1934, as amended, as a process designed by, or under the supervision of, the Company’s principal executive and principal financial officers and effected by the Company’s Board of Directors, management and other personnel, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. The Company’s internal control over financial reporting includes those policies and procedures that:
• | pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the Company; |
• | provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the Company are being made only in accordance with authorizations of management and directors of the Company; and |
• | provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the Company’s assets that could have a material effect on the financial statements. |
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
The Company’s management assessed the effectiveness of the Company’s internal control over financial reporting as of December 31, 2016. In making this assessment, it used the criteria set forth in the Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework)(COSO). Based on its
70
assessment, the Company’s management has concluded that, as of December 31, 2016, the Company’s internal control over financial reporting is effective based on those criteria.
The Company’s independent registered public accounting firm, Ernst & Young LLP, issued an attestation report on the Company’s internal control over financial reporting. See Section 4 below.
(3) Changes in Internal Controls. During the quarter ended December 31, 2016, there were no changes in the Company’s internal control over financial reporting that materially affected, or are reasonably likely to materially affect, the Company’s internal control over financial reporting.
71
(4) Report of Independent Registered Public Accounting Firm
The Board of Directors and Shareholders of
Vertex Pharmaceuticals Incorporated
We have audited Vertex Pharmaceuticals Incorporated’s internal control over financial reporting as of December 31, 2016, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework) (the COSO criteria). Vertex Pharmaceuticals Incorporated’s management is responsible for maintaining effective internal control over financial reporting, and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management’s Annual Report on Internal Control Over Financial Reporting. Our responsibility is to express an opinion on the company’s internal control over financial reporting based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
In our opinion, Vertex Pharmaceuticals Incorporated maintained, in all material respects, effective internal control over financial reporting as of December 31, 2016, based on the COSO criteria.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), the consolidated balance sheets of Vertex Pharmaceuticals Incorporated as of December 31, 2016 and 2015, and the related consolidated statements of operations, comprehensive loss, shareholders’ equity and noncontrolling interest, and cash flows for each of the three years in the period ended December 31, 2016 of Vertex Pharmaceuticals Incorporated and our report dated February 23, 2017 expressed an unqualified opinion thereon.
/s/ Ernst & Young LLP
Boston, Massachusetts
February 23, 2017
ITEM 9B. | OTHER INFORMATION |
Not applicable.
72
PART III
Portions of our definitive Proxy Statement for the 2017 Annual Meeting of Shareholders, or 2017 Proxy Statement, are incorporated by reference into this Part III of our Annual Report on Form 10-K.
ITEM 10. | DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE |
The information regarding directors required by this Item 10 will be included in our 2017 Proxy Statement and is incorporated herein by reference. We expect this information to be provided under “Election of Directors,” “Corporate Governance and Risk Management,” “Shareholder Proposals for the 2017 Annual Meeting and Nominations for Director,” “Section 16(a) Beneficial Ownership Reporting Compliance” and “Code of Conduct.” The information regarding executive officers required by this Item 10 as well as certain information regarding our directors is included in Part I of this Annual Report on Form 10-K.
ITEM 11. | EXECUTIVE COMPENSATION |
The information required by this Item 11 will be included in the 2017 Proxy Statement and is incorporated herein by reference. We expect this information to be provided under “Compensation Committee Interlocks and Insider Participation,” “Compensation Discussion and Analysis,” “Compensation and Equity Tables,” “Director Compensation,” “Management Development and Compensation Committee Report” and/or “Corporate Governance and Risk Management.”
ITEM 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS |
The information required by this Item 12 will be included in the 2017 Proxy Statement and is incorporated herein by reference. We expect this information to be provided under “Security Ownership of Certain Beneficial Owners and Management” and “Equity Compensation Plan Information.”
ITEM 13. | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE |
The information required by this Item 13 will be included in the 2017 Proxy Statement and is incorporated herein by reference. We expect this information to be provided under “ Election of Directors,” “Corporate Governance and Risk Management,” “Approval of Related Person Transactions” and “Transactions with Related Persons.”
ITEM 14. | PRINCIPAL ACCOUNTANT FEES AND SERVICES |
The information required by this Item 14 will be included in the 2017 Proxy Statement and is incorporated herein by reference. We expect this information to be provided under “Ratification of the Appointment of Independent Registered Public Accounting Firm.”
73
PART IV
ITEM 15. | EXHIBITS AND FINANCIAL STATEMENT SCHEDULES |
(a)(1) The Financial Statements required to be filed by Items 8 and 15(c) of Form 10-K, and filed herewith, are as follows:
Page Number in this Form 10-K | |
Report of Independent Registered Public Accounting Firm | |
Consolidated Statements of Operations for the years ended December 31, 2016, 2015 and 2014 | |
Consolidated Statements of Comprehensive Loss for the years ended December 31, 2016, 2015 and 2014 | |
Consolidated Balance Sheets as of December 31, 2016 and 2015 | |
Consolidated Statements of Shareholders’ Equity and Noncontrolling Interest for the years ended December 31, 2016, 2015 and 2014 | |
Consolidated Statements of Cash Flows for the years ended December 31, 2016, 2015 and 2014 | |
Notes to Consolidated Financial Statements | |
(a)(2) Financial Statement Schedules have been omitted because they are either not applicable or the required information is included in the consolidated financial statements or notes thereto listed in (a)(1) above.
(a)(3) Exhibits.
The following is a list of exhibits filed as part of this Annual Report on Form 10-K.
Exhibit Number | Exhibit Description | Filed with this report | Incorporated by Reference herein from—Form or Schedule | Filing Date/ Period Covered | SEC File/ Reg. Number |
3.1 | Restated Articles of Organization of Vertex Pharmaceuticals Incorporated, as amended. | 10-Q (Exhibit 3.1) | August 4, 2015 | 000-19319 | |
3.2 | Amended and Restated By-Laws of Vertex Pharmaceuticals Incorporated, as subsequently amended on April 26, 2016. | 10-Q (Exhibit 3.1) | May 3, 2016 | 000-19319 | |
4.1 | Specimen stock certificate. | S-1 (Exhibit 4.1) | July 18, 1991 | 33-40966 | |
Collaboration Agreements | |||||
10.1 | Research, Development and Commercialization Agreement, dated as of May 24, 2004, between Vertex Pharmaceuticals Incorporated and Cystic Fibrosis Foundation Therapeutics Incorporated.† | 10-Q/A (Exhibit 10.2) | August 19, 2011 | 000-19319 | |
10.2 | Amendment No. 1 to Research, Development and Commercialization Agreement, dated as of January 6, 2006, between Vertex Pharmaceuticals Incorporated and Cystic Fibrosis Foundation Therapeutics Incorporated.† | 10-K (Exhibit 10.9) | March 16, 2006 | 000-19319 | |
10.3 | Amendment No. 2 to Research, Development and Commercialization Agreement, dated as of March 17, 2006, between Vertex Pharmaceuticals Incorporated and Cystic Fibrosis Foundation Therapeutics Incorporated. | 10-Q/A (Exhibit 10.6) | August 19, 2011 | 000-19319 | |
10.4 | Amendment No. 5 to Research, Development and Commercialization Agreement, effective as of April 1, 2011, between Vertex Pharmaceuticals Incorporated and Cystic Fibrosis Foundation Therapeutics Incorporated.† | 10-Q (Exhibit 10.3) | August 9, 2011 | 000-19319 | |
10.5 | Amendment No. 7 to Research, Development and Commercialization Agreement, dated October 13, 2016, between Vertex Pharmaceuticals Incorporated and Cystic Fibrosis Foundation Therapeutics Incorporated. † | X | |||
10.6 | Strategic Collaboration and License Agreement, dated as of June 4, 2015, by and among Parion Sciences, Inc., Vertex Pharmaceuticals Incorporated and Vertex Pharmaceuticals (Europe) Limited.† | 10-Q (Exhibit 10.2) | August 4, 2015 | 000-19319 | |
74
Exhibit Number | Exhibit Description | Filed with this report | Incorporated by Reference herein from—Form or Schedule | Filing Date/ Period Covered | SEC File/ Reg. Number |
10.7 | Strategic Collaboration, Option and License Agreement, dated October 26, 2015, by and among CRISPR Therapeutics AG, CRISPR Therapeutics Limited, CRISPR Therapeutics, Inc., Tracr Hematology Ltd., Vertex Pharmaceuticals Incorporated and Vertex Pharmaceuticals (Europe) Limited.† | 10-K (Exhibit 10.6) | February 16, 2016 | 000-19319 | |
Leases | |||||
10.8 | Lease, dated May 5, 2011, between Fifty Northern Avenue LLC and Vertex Pharmaceuticals Incorporated.† | 10-Q (Exhibit 10.4) | August 9, 2011 | 000-19319 | |
10.9 | Lease, dated May 5, 2011, between Eleven Fan Pier Boulevard LLC and Vertex Pharmaceuticals Incorporated.† | 10-Q (Exhibit 10.5) | August 9, 2011 | 000-19319 | |
10.10 | Lease, dated as of January 18, 2001, between Kendall Square, LLC and Vertex Pharmaceuticals Incorporated.† | 10-K (Exhibit 10.16) | March 26, 2001 | 000-19319 | |
10.11 | Lease, dated December 2, 2015, between ARE-SD Region No. 23, LLC and Vertex Pharmaceuticals Incorporated. | 10-K (Exhibit 10.10) | February 16, 2016 | 000-19319 | |
Financing Agreements | |||||
10.12 | Credit Agreement, dated as of October 13, 2016, among Vertex Pharmaceuticals Incorporated, Bank of America, N.A. and the other lenders party thereto. | X | |||
10.13 | First Amendment to Credit Agreement, dated as of February 9, 2017, among Vertex Pharmaceuticals Incorporated, Bank of America, N.A. and the other lenders party thereto. | X | |||
Equity Plans | |||||
10.14 | 1996 Stock and Option Plan, as amended and restated as of March 14, 2005.* | 10-K (Exhibit 10.3) | March 16, 2005 | 000-19319 | |
10.15 | Form of Stock Option Grant under 1996 Stock and Option Plan.* | 8-K (Exhibit 10.1) | February 9, 2005 | 000-19319 | |
10.16 | Amended and Restated 2006 Stock and Option Plan.* | 10-Q (Exhibit 10.3) | August 8, 2012 | 000-19319 | |
10.17 | Form of Stock Option Agreement under Amended and Restated 2006 Stock and Option Plan (granted prior to July 30, 2013).* | 8-K (Exhibit 10.2) | May 15, 2006 | 000-19319 | |
10.18 | Form of Restricted Stock Agreement under Amended and Restated 2006 Stock and Option Plan (granted prior to July 30, 2013).* | 8-K (Exhibit 10.3) | May 15, 2006 | 000-19319 | |
10.19 | Form of Restricted Stock Agreement (Performance Accelerated Restricted Stock) under Amended and Restated 2006 Stock and Option Plan (granted prior to July 30, 2013).* | 8-K (Exhibit 10.4) | May 15, 2006 | 000-19319 | |
10.20 | Form of Stock Option Agreement under Amended and Restated 2006 Stock and Option Plan (granted on or after July 30, 2013).* | 10-K (Exhibit 10.20) | February 13, 2015 | 000-19319 | |
10.21 | Form of Restricted Stock Agreement under Amended and Restated 2006 Stock and Option Plan (granted on or after July 30, 2013).* | 10-K (Exhibit 10.21) | February 13, 2015 | 000-19319 | |
10.22 | Form of Restricted Stock Unit Agreement under Amended and Restated 2006 Stock and Option Plan (granted on or after July 30, 2013).* | 10-K (Exhibit 10.22) | February 13, 2015 | 000-19319 | |
10.23 | Amended and Restated 2013 Stock and Option Plan.* | DEF 14A (Appendix A) | April 30, 2015 | 000-19319 | |
10.24 | Form of Non-Qualified Stock Option Agreement under 2013 Stock and Option Plan.* | 10-K (Exhibit 10.17) | February 13, 2015 | 000-19319 | |
10.25 | Form of Restricted Stock Agreement under 2013 Stock and Option Plan.* | 10-K (Exhibit 10.18) | February 13, 2015 | 000-19319 | |
10.26 | Form of Restricted Stock Unit Agreement under 2013 Stock and Option Plan (U.S.).* | 10-K (Exhibit 10.25) | February 16, 2016 | 000-19319 | |
10.27 | Form of Restricted Stock Unit Agreement under 2013 Stock and Option Plan (International).* | 10-K (Exhibit 10.19) | February 13, 2015 | 000-19319 | |
10.28 | Non-Employee Director Deferred Compensation Plan.* | 10-K (Exhibit 10.27) | February 16, 2016 | 000-19319 | |
10.29 | Vertex Pharmaceuticals Incorporated Employee Stock Purchase Plan, as amended and restated as of July 12, 2016.* | 10-Q (Exhibit 10.1) | August 1, 2016 | 000-19319 | |
Agreements with Executive Officers and Directors | |||||
10.30 | Amended and Restated Employment Agreement, dated November 30, 2016, by and between Vertex Pharmaceuticals Incorporated and Jeffrey M. Leiden, M.D., Ph.D* | 8-K (Exhibit 10.1) | November 29, 2016 | 000-19319 | |
75
Exhibit Number | Exhibit Description | Filed with this report | Incorporated by Reference herein from—Form or Schedule | Filing Date/ Period Covered | SEC File/ Reg. Number |
10.31 | Employee Non-disclosure, Non-competition and Inventions Agreement between Jeffrey M. Leiden and Vertex, dated December 14, 2011.* | 10-K (Exhibit 10.35) | February 22, 2012 | 000-19319 | |
10.32 | Employment Agreement, dated as of August 27, 2012, between Vertex Pharmaceuticals Incorporated and Stuart Arbuckle.* | 10-Q (Exhibit 10.1) | November 6, 2012 | 000-19319 | |
10.33 | Change of Control Agreement, dated as of August 27, 2012, between Vertex Pharmaceuticals Incorporated and Stuart Arbuckle.* | 10-Q (Exhibit 10.2) | November 6, 2012 | 000-19319 | |
10.34 | Employment Agreement, dated as of December 12, 2014, between Vertex Pharmaceuticals Incorporated and David Altshuler.* | 10-K (Exhibit 10.34) | February 16, 2016 | 000-19319 | |
10.35 | Change of Control Agreement, dated as of December 12, 2014, between Vertex Pharmaceuticals Incorporated and David Altshuler.* | 10-K (Exhibit 10.35) | February 16, 2016 | 000-19319 | |
10.36 | Amended and Restated Employment Agreement, dated as of November 8, 2004, between Vertex Pharmaceuticals Incorporated and Ian F. Smith.* | 10-Q (Exhibit 10.13) | November 9, 2004 | 000-19319 | |
10.37 | Amendment No. 1 to Amended and Restated Employment Agreement between Ian F. Smith and Vertex Pharmaceuticals Incorporated, dated December 29, 2008.* | 10-K (Exhibit 10.66) | February 17, 2009 | 000-19319 | |
10.38 | Employment Agreement, dated as of December 2, 2013, between Vertex Pharmaceuticals Incorporated and Jeffrey Chodakewicz.* | 10-Q (Exhibit 10.1) | March 31, 2015 | 000-19319 | |
10.39 | Change of Control Agreement, dated as of December 2, 2013, between Vertex Pharmaceuticals Incorporated and Jeffrey Chodakewicz.* | 10-Q (Exhibit 10.2) | March 31, 2015 | 000-19319 | |
10.40 | Employment Agreement, dated as of November 14, 2015, between Vertex Pharmaceuticals Incorporated and Michael Parini.* | X | |||
10.41 | Change of Control Agreement, dated as of November 9, 2015, between Vertex Pharmaceuticals Incorporated and Michael Parini.* | X | |||
10.42 | Third Amended and Restated Employment Agreement, dated as of February 26, 2013, between Vertex Pharmaceuticals Incorporated and Amit Sachdev.* | X | |||
10.43 | Third Amended and Restated Change of Control Agreement, dated as of February 26, 2013, between Vertex Pharmaceuticals Incorporated and Amit Sachdev.* | X | |||
10.44 | Form of Employee Non-Disclosure and Inventions Agreement.* | S-1 (Exhibit 10.4) | May 30, 1991 | 33-40966 | |
10.45 | Vertex Employee Compensation Plan.* | 10-K (Exhibit 10.41) | February 16, 2016 | 000-19319 | |
10.46 | Vertex Pharmaceuticals Non-Employee Board Compensation.* | 10-K (Exhibit 10.42) | February 16, 2016 | 000-19319 | |
Subsidiaries | |||||
21.1 | Subsidiaries of Vertex Pharmaceuticals Incorporated. | X | |||
Consent | |||||
23.1 | Consent of Independent Registered Public Accounting Firm, Ernst & Young LLP. | X | |||
Certifications | |||||
31.1 | Certification of the Chief Executive Officer under Section 302 of the Sarbanes-Oxley Act of 2002. | X | |||
31.2 | Certification of the Chief Financial Officer under Section 302 of the Sarbanes-Oxley Act of 2002. | X | |||
32.1 | Certification of the Chief Executive Officer and the Chief Financial Officer under Section 906 of the Sarbanes-Oxley Act of 2002. | X | |||
101.INS | XBRL Instance | X | |||
101.SCH | XBRL Taxonomy Extension Schema | X | |||
101.CAL | XBRL Taxonomy Extension Calculation | X | |||
101.LAB | XBRL Taxonomy Extension Labels | X | |||
101.PRE | XBRL Taxonomy Extension Presentation | X | |||
101.DEF | XBRL Taxonomy Extension Definition | X | |||
* Management contract, compensatory plan or agreement.
† | Confidential portions of this document have been filed separately with the Securities and Exchange Commission pursuant to a request for confidential treatment. |
76
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
Vertex Pharmaceuticals Incorporated | ||
February 23, 2017 | By: | /s/ Jeffrey M. Leiden |
Jeffrey M. Leiden Chief Executive Officer | ||
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
Name | Title | Date | |||||||||||
/s/ Jeffrey M. Leiden | Chair of the Board, President and Chief Executive Officer (Principal Executive Officer) | ||||||||||||
Jeffrey M. Leiden | February 23, 2017 | ||||||||||||
/s/ Ian F. Smith | Executive Vice President, Chief Operating Officer and Chief Financial Officer (Principal Financial Officer) | ||||||||||||
Ian F. Smith | February 23, 2017 | ||||||||||||
/s/ Paul M. Silva | Senior Vice President and Corporate Controller (Principal Accounting Officer) | ||||||||||||
Paul M. Silva | February 23, 2017 | ||||||||||||
/s/ Sangeeta N. Bhatia | Director | ||||||||||||
Sangeeta N. Bhatia | February 23, 2017 | ||||||||||||
/s/ Joshua S. Boger | Director | ||||||||||||
Joshua S. Boger | February 23, 2017 | ||||||||||||
/s/ Terrence C. Kearney | Director | ||||||||||||
Terrence C. Kearney | February 23, 2017 | ||||||||||||
/s/ Yuchun Lee | Director | ||||||||||||
Yuchun Lee | February 23, 2017 | ||||||||||||
/s/ Margaret G. McGlynn | Director | ||||||||||||
Margaret G. McGlynn | February 23, 2017 | ||||||||||||
/s/ Bruce I. Sachs | Director | ||||||||||||
Bruce I. Sachs | February 23, 2017 | ||||||||||||
/s/ Elaine S. Ullian | Director | ||||||||||||
Elaine S. Ullian | February 23, 2017 | ||||||||||||
/s/ William D. Young | Director | ||||||||||||
William D. Young | February 23, 2017 | ||||||||||||
77
Report of Independent Registered Public Accounting Firm
The Board of Directors and Shareholders of
Vertex Pharmaceuticals Incorporated
We have audited the accompanying consolidated balance sheets of Vertex Pharmaceuticals Incorporated as of December 31, 2016 and 2015, and the related consolidated statements of operations, comprehensive loss, shareholders’ equity and noncontrolling interest, and cash flows for each of the three years in the period ended December 31, 2016. These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the consolidated financial position of Vertex Pharmaceuticals Incorporated at December 31, 2016 and 2015, and the consolidated results of its operations and its cash flows for each of the three years in the period ended December 31, 2016, in conformity with U.S. generally accepted accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), Vertex Pharmaceuticals Incorporated’s internal control over financial reporting as of December 31, 2016, based on the criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework) and our report dated February 23, 2017 expressed an unqualified opinion thereon.
/s/ Ernst & Young LLP | |
Boston, Massachusetts
February 23, 2017
F-1
VERTEX PHARMACEUTICALS INCORPORATED Consolidated Statements of Operations (in thousands, except per share amounts) | |||||||||||
Year Ended December 31, | |||||||||||
2016 | 2015 | 2014 | |||||||||
Revenues: | |||||||||||
Product revenues, net | $ | 1,683,632 | $ | 1,000,324 | $ | 487,821 | |||||
Royalty revenues | 16,600 | 23,959 | 40,919 | ||||||||
Collaborative revenues | 1,945 | 8,053 | 51,675 | ||||||||
Total revenues | 1,702,177 | 1,032,336 | 580,415 | ||||||||
Costs and expenses: | |||||||||||
Cost of product revenues | 206,811 | 117,151 | 39,725 | ||||||||
Royalty expenses | 3,649 | 7,361 | 21,262 | ||||||||
Research and development expenses | 1,047,690 | 995,922 | 855,506 | ||||||||
Sales, general and administrative expenses | 432,829 | 376,575 | 305,409 | ||||||||
Restructuring expenses | 1,262 | 2,206 | 50,925 | ||||||||
Total costs and expenses | 1,692,241 | 1,499,215 | 1,272,827 | ||||||||
Income (loss) from operations | 9,936 | (466,879 | ) | (692,412 | ) | ||||||
Interest expense, net | (81,432 | ) | (84,206 | ) | (72,863 | ) | |||||
Other income (expense), net | 4,130 | (6,715 | ) | 30,400 | |||||||
Loss from continuing operations before provision for income taxes | (67,366 | ) | (557,800 | ) | (734,875 | ) | |||||
Provision for income taxes | 16,665 | 30,381 | 6,958 | ||||||||
Loss from continuing operations | (84,031 | ) | (588,181 | ) | (741,833 | ) | |||||
Loss from discontinued operations, net of tax benefit of $0, $0 and $0, respectively | — | — | (912 | ) | |||||||
Net loss | (84,031 | ) | (588,181 | ) | (742,745 | ) | |||||
(Income) loss attributable to noncontrolling interest | (28,021 | ) | 31,847 | 4,190 | |||||||
Net loss attributable to Vertex | $ | (112,052 | ) | $ | (556,334 | ) | $ | (738,555 | ) | ||
Amounts attributable to Vertex: | |||||||||||
Loss from continuing operations | $ | (112,052 | ) | $ | (556,334 | ) | $ | (737,643 | ) | ||
Loss from discontinued operations | — | — | (912 | ) | |||||||
Net loss attributable to Vertex | $ | (112,052 | ) | $ | (556,334 | ) | $ | (738,555 | ) | ||
Amounts per share attributable to Vertex common shareholders: | |||||||||||
Net loss from continuing operations: | |||||||||||
Basic | $ | (0.46 | ) | $ | (2.31 | ) | $ | (3.14 | ) | ||
Diluted | $ | (0.46 | ) | $ | (2.31 | ) | $ | (3.14 | ) | ||
Net loss from discontinued operations: | |||||||||||
Basic | $ | — | $ | — | $ | — | |||||
Diluted | $ | — | $ | — | $ | — | |||||
Net loss: | |||||||||||
Basic | $ | (0.46 | ) | $ | (2.31 | ) | $ | (3.14 | ) | ||
Diluted | $ | (0.46 | ) | $ | (2.31 | ) | $ | (3.14 | ) | ||
Shares used in per share calculations: | |||||||||||
Basic | 244,685 | 241,312 | 235,307 | ||||||||
Diluted | 244,685 | 241,312 | 235,307 | ||||||||
The accompanying notes are an integral part of the consolidated financial statements.
F-2
VERTEX PHARMACEUTICALS INCORPORATED Consolidated Statements of Comprehensive Loss (in thousands) | |||||||||||
Year ended December 31, | |||||||||||
2016 | 2015 | 2014 | |||||||||
Net loss | $ | (84,031 | ) | $ | (588,181 | ) | $ | (742,745 | ) | ||
Changes in other comprehensive income (loss): | |||||||||||
Unrealized holding gains (losses) on marketable securities, net of tax | 17,395 | 249 | (165 | ) | |||||||
Unrealized gains on foreign currency forward contracts, net of tax | 7,736 | 1,767 | 2,034 | ||||||||
Foreign currency translation adjustment | (5,782 | ) | (1,109 | ) | (646 | ) | |||||
Total changes in other comprehensive income (loss) | 19,349 | 907 | 1,223 | ||||||||
Comprehensive loss | (64,682 | ) | (587,274 | ) | (741,522 | ) | |||||
Comprehensive (income) loss attributable to noncontrolling interest | (28,021 | ) | 31,847 | 4,190 | |||||||
Comprehensive loss attributable to Vertex | $ | (92,703 | ) | $ | (555,427 | ) | $ | (737,332 | ) | ||
The accompanying notes are an integral part of the consolidated financial statements.
F-3
VERTEX PHARMACEUTICALS INCORPORATED Consolidated Balance Sheets (in thousands, except share and per share amounts) | |||||||
December 31, | |||||||
2016 | 2015 | ||||||
Assets | |||||||
Current assets: | |||||||
Cash and cash equivalents | $ | 1,183,945 | $ | 714,768 | |||
Marketable securities, available-for-sale | 250,612 | 327,694 | |||||
Restricted cash and cash equivalents (VIE) | 47,762 | 78,910 | |||||
Accounts receivable, net | 201,083 | 173,838 | |||||
Inventories | 77,604 | 57,207 | |||||
Prepaid expenses and other current assets | 70,534 | 54,736 | |||||
Total current assets | 1,831,540 | 1,407,153 | |||||
Property and equipment, net | 698,362 | 697,715 | |||||
Intangible assets | 284,340 | 284,340 | |||||
Goodwill | 50,384 | 50,384 | |||||
Cost method investments | 20,276 | — | |||||
Note receivable | — | 30,000 | |||||
Restricted cash | 52 | 22,083 | |||||
Other assets | 11,833 | 6,912 | |||||
Total assets | $ | 2,896,787 | $ | 2,498,587 | |||
Liabilities and Shareholders’ Equity | |||||||
Current liabilities: | |||||||
Accounts payable | $ | 61,451 | $ | 74,942 | |||
Accrued expenses | 315,249 | 305,820 | |||||
Deferred revenues, current portion | 6,005 | 16,296 | |||||
Accrued restructuring expense, current portion | 6,047 | 7,894 | |||||
Capital lease obligations, current portion | 19,426 | 15,545 | |||||
Senior secured term loan, current portion | — | 71,296 | |||||
Customer deposits | 73,416 | — | |||||
Credit facility | 300,000 | — | |||||
Other liabilities, current portion | 10,943 | 14,374 | |||||
Total current liabilities | 792,537 | 506,167 | |||||
Deferred revenues, excluding current portion | 6,632 | 9,714 | |||||
Accrued restructuring expense, excluding current portion | 1,907 | 7,464 | |||||
Capital lease obligations, excluding current portion | 34,976 | 42,923 | |||||
Deferred tax liability | 134,063 | 110,439 | |||||
Construction financing lease obligation, excluding current portion | 486,359 | 472,611 | |||||
Senior secured term loan, excluding current portion | — | 223,863 | |||||
Other liabilities, excluding current portion | 102,122 | 31,778 | |||||
Total liabilities | 1,558,596 | 1,404,959 | |||||
Commitments and contingencies | |||||||
Shareholders’ equity: | |||||||
Preferred stock, $0.01 par value; 1,000,000 shares authorized; none issued and outstanding at December 31, 2015 and 2014 | — | — | |||||
Common stock, $0.01 par value; 500,000,000 shares authorized at December 31, 2016 and 2015; 248,300,517 and 246,306,818 shares issued and outstanding at December 31, 2016 and 2015, respectively | 2,450 | 2,427 | |||||
Additional paid-in capital | 6,506,795 | 6,197,500 | |||||
Accumulated other comprehensive income | 21,173 | 1,824 | |||||
Accumulated deficit | (5,373,836 | ) | (5,261,784 | ) | |||
Total Vertex shareholders’ equity | 1,156,582 | 939,967 | |||||
Noncontrolling interest | 181,609 | 153,661 | |||||
Total shareholders’ equity | 1,338,191 | 1,093,628 | |||||
Total liabilities and shareholders’ equity | $ | 2,896,787 | $ | 2,498,587 | |||
The accompanying notes are an integral part of the consolidated financial statements.
F-4
VERTEX PHARMACEUTICALS INCORPORATED Consolidated Statements of Shareholders’ Equity and Noncontrolling Interest (in thousands) | ||||||||||||||||||||||||||||||
Common Stock | Additional Paid-in Capital | Accumulated Other Comprehensive Loss | Accumulated Deficit | Total Vertex Shareholders’ Equity | Noncontrolling Interest | Total Shareholders’ Equity | ||||||||||||||||||||||||
Shares | Amount | |||||||||||||||||||||||||||||
Balance, December 31, 2013 | 233,789 | $ | 2,320 | $ | 5,321,286 | $ | (306 | ) | $ | (3,966,895 | ) | $ | 1,356,405 | $ | — | $ | 1,356,405 | |||||||||||||
Other comprehensive income, net of tax | 1,223 | 1,223 | 1,223 | |||||||||||||||||||||||||||
Net (loss) income | (738,555 | ) | (738,555 | ) | (4,190 | ) | (742,745 | ) | ||||||||||||||||||||||
Issuance of common stock under benefit plans | 7,975 | 65 | 274,743 | 274,808 | 274,808 | |||||||||||||||||||||||||
Stock-based compensation expense | 178,965 | 178,965 | 178,965 | |||||||||||||||||||||||||||
Tax benefit from equity compensation | 2,160 | 2,160 | 2,160 | |||||||||||||||||||||||||||
Noncontrolling interest upon consolidation | — | 25,367 | 25,367 | |||||||||||||||||||||||||||
Balance, December 31, 2014 | 241,764 | $ | 2,385 | $ | 5,777,154 | $ | 917 | $ | (4,705,450 | ) | $ | 1,075,006 | $ | 21,177 | $ | 1,096,183 | ||||||||||||||
Other comprehensive income, net of tax | 907 | 907 | 907 | |||||||||||||||||||||||||||
Net loss | (556,334 | ) | (556,334 | ) | (31,847 | ) | (588,181 | ) | ||||||||||||||||||||||
Issuance of common stock under benefit plans | 4,543 | 42 | 185,234 | 185,276 | 14 | 185,290 | ||||||||||||||||||||||||
Stock-based compensation expense | 235,112 | 235,112 | 235,112 | |||||||||||||||||||||||||||
Tax benefit from equity compensation | — | — | — | |||||||||||||||||||||||||||
Noncontrolling interest upon consolidation | — | 164,317 | 164,317 | |||||||||||||||||||||||||||
Balance, December 31, 2015 | 246,307 | $ | 2,427 | $ | 6,197,500 | $ | 1,824 | $ | (5,261,784 | ) | $ | 939,967 | $ | 153,661 | $ | 1,093,628 | ||||||||||||||
Other comprehensive income, net of tax | 19,349 | 19,349 | 19,349 | |||||||||||||||||||||||||||
Net loss | (112,052 | ) | (112,052 | ) | 28,021 | (84,031 | ) | |||||||||||||||||||||||
Issuance of common stock under benefit plans | 1,994 | 23 | 67,983 | 68,006 | — | 68,006 | ||||||||||||||||||||||||
Stock-based compensation expense | 241,312 | 241,312 | (73 | ) | 241,239 | |||||||||||||||||||||||||
Balance, December 31, 2016 | 248,301 | $ | 2,450 | $ | 6,506,795 | $ | 21,173 | $ | (5,373,836 | ) | $ | 1,156,582 | $ | 181,609 | $ | 1,338,191 | ||||||||||||||
The accompanying notes are an integral part of the consolidated financial statements.
F-5
VERTEX PHARMACEUTICALS INCORPORATED Consolidated Statements of Cash Flows (in thousands) | |||||||||||
Year Ended December 31, | |||||||||||
2016 | 2015 | 2014 | |||||||||
Cash flows from operating activities: | |||||||||||
Net loss | $ | (84,031 | ) | $ | (588,181 | ) | $ | (742,745 | ) | ||
Adjustments to reconcile net loss to net cash provided by (used in) operating activities: | |||||||||||
Stock-based compensation expense | 237,705 | 231,025 | 177,542 | ||||||||
Depreciation and amortization expense | 61,398 | 62,343 | 63,257 | ||||||||
Deferred income taxes | 16,961 | 3,283 | 281 | ||||||||
Impairment of property and equipment | — | 2,516 | 1,689 | ||||||||
Excess tax benefit from share-based payment arrangements | — | — | (2,160 | ) | |||||||
Other non-cash items, net | 6,140 | 9,532 | — | ||||||||
Changes in operating assets and liabilities, excluding the effects of the acquisition and deconsolidation of variable interest entities: | |||||||||||
Accounts receivable, net | (33,027 | ) | (104,847 | ) | 7,428 | ||||||
Inventories | (16,450 | ) | (23,146 | ) | (16,469 | ) | |||||
Prepaid expenses and other assets | (8,699 | ) | (9,260 | ) | (15,771 | ) | |||||
Accounts payable | (11,745 | ) | (1,709 | ) | 25,048 | ||||||
Accrued expenses and other liabilities | 88,649 | 102,746 | (63,183 | ) | |||||||
Accrued restructuring expense | (7,426 | ) | (30,492 | ) | 17,502 | ||||||
Deferred revenues | (13,372 | ) | (19,242 | ) | (25,531 | ) | |||||
Net cash provided by provided by (used in) operating activities | 236,103 | (365,432 | ) | (573,112 | ) | ||||||
Cash flows from investing activities: | |||||||||||
Maturities of marketable securities | 757,562 | 1,067,443 | 1,557,938 | ||||||||
Purchases of marketable securities | (616,625 | ) | (633,041 | ) | (1,424,172 | ) | |||||
Payment for acquisition of variable interest entity | — | (80,000 | ) | (10,000 | ) | ||||||
Expenditures for property and equipment | (56,563 | ) | (45,302 | ) | (51,201 | ) | |||||
Investment in note receivable | (20,000 | ) | (30,000 | ) | — | ||||||
Investment in CRISPR | (13,075 | ) | — | — | |||||||
(Decrease) increase in restricted cash and cash equivalents | 22,029 | (21,981 | ) | — | |||||||
Decrease in restricted cash and cash equivalents (VIE) | 31,148 | 11,685 | 1,638 | ||||||||
Increase (decrease) in other assets | (7 | ) | 52 | (244 | ) | ||||||
Payments returned related to construction financing lease obligation | — | — | 8,050 | ||||||||
Net cash provided by investing activities | 104,469 | 268,856 | 82,009 | ||||||||
Cash flows from financing activities: | |||||||||||
Issuances of common stock under benefit plans | 68,230 | 185,592 | 274,615 | ||||||||
Payments on construction financing lease obligation | (432 | ) | (381 | ) | (336 | ) | |||||
Proceeds from lease financing | 11,208 | 23,662 | — | ||||||||
Payments on capital lease financing | (17,597 | ) | (19,954 | ) | (21,443 | ) | |||||
Proceeds from senior secured term loan | — | — | 294,243 | ||||||||
Payments on senior secured term loan | (75,000 | ) | — | — | |||||||
Proceeds from revolving credit facility | 74,965 | — | — | ||||||||
Payments of debt issuance costs | (3,103 | ) | — | — | |||||||
Advance from CFFT | 75,000 | — | — | ||||||||
Excess tax benefit from share-based payment arrangements | — | — | 2,160 | ||||||||
Net cash provided by financing activities | 133,271 | 188,919 | 549,239 | ||||||||
Effect of changes in exchange rates on cash | (4,666 | ) | (2,834 | ) | (2,176 | ) | |||||
Net increase in cash and cash equivalents | 469,177 | 89,509 | 55,960 | ||||||||
Cash and cash equivalents—beginning of period | 714,768 | 625,259 | 569,299 | ||||||||
Cash and cash equivalents—end of period | $ | 1,183,945 | $ | 714,768 | $ | 625,259 | |||||
Supplemental disclosure of cash flow information: | |||||||||||
Cash paid for interest | $ | 83,656 | $ | 85,613 | $ | 68,963 | |||||
Cash (received from) paid for income taxes | $ | (2,579 | ) | $ | 1,806 | $ | 1,210 | ||||
Non-cash investing and financing activities: | |||||||||||
Capitalization of costs related to construction financing lease obligation | $ | 14,238 | $ | — | $ | 25,564 | |||||
Assets acquired under capital lease obligations | $ | — | $ | — | $ | 9,188 | |||||
Issuances of common stock exercises from employee benefit plans receivable | $ | 68 | $ | 361 | $ | 637 | |||||
Proceeds from revolving credit facility directly paid to settle all outstanding obligations under the term loan | $ | 225,000 | $ | — | $ | — | |||||
The accompanying notes are an integral part of the consolidated financial statements.
F-6
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements
A. | Nature of Business and Accounting Policies |
Business
Vertex Pharmaceuticals Incorporated (“Vertex” or the “Company”) is in the business of discovering, developing, manufacturing and commercializing medicines for serious diseases. The Company uses precision medicine approaches with the goal of creating transformative medicines for patients in specialty markets. The Company is focused on developing and commercializing therapies for the treatment of cystic fibrosis (“CF”) and advancing its research and development programs. The Company’s two marketed medicines are ORKAMBI and KALYDECO, which are approved to treat patients with CF who have specific mutations in their cystic fibrosis transmembrane conductance regulator (“CFTR”) gene.
The Company’s net loss attributable to Vertex for 2016 was $112.1 million, or $0.46 per share. As of December 31, 2016, the Company had cash, cash equivalents and marketable securities of $1.43 billion. The Company expects that cash flows from the sales of its products, together with the Company’s cash, cash equivalents and marketable securities, will be sufficient to fund its operations for at least the next twelve months.
Vertex is subject to risks common to companies in its industry including, but not limited to, the dependence on revenues from ORKAMBI and KALYDECO, competition, uncertainty about clinical trial outcomes and regulatory approvals, uncertainties relating to pharmaceutical pricing and reimbursement, uncertainty related to international expansion, uncertain protection of proprietary technology, the need to comply with government regulations, share price volatility, dependence on collaborative relationships and potential product liability.
Basis of Presentation
The consolidated financial statements reflect the operations of (i) the Company, (ii) its wholly-owned subsidiaries and (iii) consolidated variable interest entities (VIEs). In addition, the consolidated financial statements for 2014 reflect the operations of Alios BioPharma, Inc. (“Alios”), a former collaborator, as well as direct expenses Vertex incurred as a result of the Company’s agreement with Alios, as discontinued operations. All material intercompany balances and transactions have been eliminated. The Company operates in one segment, pharmaceuticals. Please refer to Note T, “Segment Information,” for enterprise-wide disclosures regarding the Company’s revenues, major customers and long-lived assets by geographic area. The Company has reclassified certain amounts in the consolidated balance sheets for the period ended December 31, 2015 between Accounts receivable, net and Prepaid expenses and other current assets to conform to the current year presentation.
Use of Estimates
The preparation of consolidated financial statements in accordance with accounting principles generally accepted in the United States of America (“GAAP”) requires management to make certain estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the consolidated financial statements, and the amounts of revenues and expenses during the reported periods. Significant estimates in these consolidated financial statements have been made in connection with the calculation of revenues, inventories, research and development expenses, stock-based compensation expense, restructuring expense, the fair value of intangible assets, goodwill, contingent consideration, noncontrolling interest, the consolidation of VIEs, leases, the fair value of cash flow hedges and the provision for or benefit from income taxes. The Company bases its estimates on historical experience and various other assumptions, including in certain circumstances future projections, that management believes to be reasonable under the circumstances. Actual results could differ from those estimates. Changes in estimates are reflected in reported results in the period in which they become known.
Revenue Recognition
Product Revenues, Net
The Company sells its products principally to a limited number of specialty pharmacy providers and selected regional wholesalers in North America as well as government-owned and supported customers in international markets (collectively, its “Customers”). The Company’s Customers in North America subsequently resell the products to patients and health care
F-7
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
providers. The Company recognizes net revenues from product sales upon delivery as long as (i) there is persuasive evidence that an arrangement exists between the Company and the Customer, (ii) collectibility is reasonably assured and (iii) the price is fixed or determinable.
In order to conclude that the price is fixed or determinable, the Company must be able to (i) calculate its gross product revenues from sales to Customers and (ii) reasonably estimate its net product revenues upon delivery to its Customer’s locations. The Company calculates gross product revenues based on the price that the Company charges its Customers. The Company estimates its net product revenues by deducting from its gross product revenues (a) trade allowances, such as invoice discounts for prompt payment and Customer fees, (b) estimated government and private payor rebates, chargebacks and discounts, (c) estimated reserves for expected product returns and (d) estimated costs of co-pay assistance programs for patients, as well as other incentives for certain indirect customers.
Trade Allowances: The Company generally provides invoice discounts on product sales to its Customers for prompt payment and pays fees for distribution services, such as fees for certain data that Customers provide to the Company. The payment terms for sales to Customers in the United States generally include a discount for payment within 30 days. The Company expects that, based on its experience, its Customers will earn these discounts and fees, and deducts the full amount of these discounts and fees from its gross product revenues and accounts receivable at the time such revenues are recognized.
Rebates, Chargebacks and Discounts: The Company contracts with government agencies and various private organizations (collectively, its “Third-party Payors”) so that products will be eligible for purchase by, or partial or full reimbursement from, such Third-party Payors. The Company estimates the rebates, chargebacks and discounts it will provide to Third-party Payors and deducts these estimated amounts from its gross product revenues at the time the revenues are recognized. For each product, the Company estimates the aggregate rebates, chargebacks and discounts that it will provide to Third-party Payors based upon (i) the Company’s contracts with these Third-party Payors, (ii) the government-mandated discounts applicable to government-funded programs, (iii) information obtained from the Company’s Customers and other third-party data regarding the payor mix for such product and (iv) historical experience.
Product Returns: The Company estimates the amount of each product that will be returned and deducts these estimated amounts from its gross revenues at the time the revenues are recognized. The Company’s Customers have the right to return unopened unprescribed packages, subject to contractual limitations. To date product returns have been minimal and, based on inventory levels held by its Customers and its distribution model, the Company believes that returns of its products will continue to be minimal.
Other Incentives: Other incentives that the Company offers include co-pay mitigation rebates provided by the Company to commercially insured patients who have coverage and who reside in states that permit co-pay mitigation programs. The Company’s co-pay mitigation programs are intended to reduce each participating patient’s portion of the financial responsibility for a product’s purchase price to a specified dollar amount. Based upon the terms of the Company’s co-pay mitigation programs, the Company estimates average co-pay mitigation amounts for each of its products in order to establish its accruals for co-pay mitigation rebates and deducts these estimated amounts from its gross product revenues at the later of the date (i) the revenues are recognized or (ii) the incentive is offered. The Company’s co-pay mitigation rebates are subject to expiration.
The Company makes significant estimates and judgments that materially affect the Company’s recognition of net product revenues. In certain instances, the Company may be unable to reasonably conclude that the price is fixed or determinable at the time of delivery, in which case it defers the recognition of revenues. Once the Company is able to determine that the price is fixed or determinable, it recognizes the revenues associated with the units in which revenue recognition was deferred. ORKAMBI net product revenues do not include any revenues from product sales in France. The Company began distributing ORKAMBI through early access programs in the fourth quarter of 2015. The Company’s consolidated balance sheet includes $73.4 million collected as of December 31, 2016 in France related to ORKAMBI that is classified as customer deposits. The Company expects that revenues from these early access programs will be recognized in the period that a formal reimbursement agreement in France is reached based on the terms of such agreement.
F-8
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
The following table summarizes activity in each of the product revenue allowance and reserve categories for the three years ended December 31, 2016:
Trade Allowances | Rebates, Chargebacks and Discounts | Product Returns | Other Incentives | Total | |||||||||||||||
(in thousands) | |||||||||||||||||||
2016 | |||||||||||||||||||
Beginning Balance | $ | 2,089 | $ | 44,669 | $ | 1,228 | $ | 1,310 | $ | 49,296 | |||||||||
Provision related to current period sales | 20,075 | 134,198 | 3,047 | 6,602 | 163,922 | ||||||||||||||
Adjustments related to prior period sales | (90 | ) | 154 | (17 | ) | (151 | ) | (104 | ) | ||||||||||
Credits/payments made | (19,506 | ) | (97,094 | ) | (766 | ) | (6,547 | ) | (123,913 | ) | |||||||||
Ending Balance | $ | 2,568 | $ | 81,927 | $ | 3,492 | $ | 1,214 | $ | 89,201 | |||||||||
2015 | |||||||||||||||||||
Beginning Balance | $ | 1,463 | $ | 29,102 | $ | 4,713 | $ | 745 | $ | 36,023 | |||||||||
Provision related to current period sales | 10,890 | 65,781 | 779 | 3,755 | 81,205 | ||||||||||||||
Adjustments related to prior period sales | (214 | ) | (19,410 | ) | (993 | ) | (235 | ) | (20,852 | ) | |||||||||
Credits/payments made | (10,050 | ) | (30,804 | ) | (3,271 | ) | (2,955 | ) | (47,080 | ) | |||||||||
Ending Balance | $ | 2,089 | $ | 44,669 | $ | 1,228 | $ | 1,310 | $ | 49,296 | |||||||||
2014 | |||||||||||||||||||
Beginning Balance | $ | 1,535 | $ | 68,244 | $ | 15,799 | $ | 1,555 | $ | 87,133 | |||||||||
Provision related to current period sales | 8,468 | 35,713 | 2,478 | 1,347 | 48,006 | ||||||||||||||
Adjustments related to prior period sales | (43 | ) | 329 | 3,056 | (72 | ) | 3,270 | ||||||||||||
Credits/payments made | (8,497 | ) | (75,184 | ) | (16,620 | ) | (2,085 | ) | (102,386 | ) | |||||||||
Ending Balance | $ | 1,463 | $ | 29,102 | $ | 4,713 | $ | 745 | $ | 36,023 | |||||||||
The Company adjusts its estimated rebates, chargebacks and discounts based on new information, including information regarding actual rebates, chargebacks and discounts for its products, as it becomes available. Claims by third-party payors for rebates, chargebacks and discounts frequently are submitted to the Company significantly after the related sales, potentially resulting in adjustments in the period in which the new information becomes known. In each of the periods presented, the Company’s adjustments relating to prior period sales principally related to the Company’s estimates for INCIVEK. During the fourth quarter of 2014, the Company withdrew INCIVEK from the market in the United States.
Royalty Revenues
The Company’s royalty revenues on commercial sales of INCIVO (telaprevir) by Janssen NV were based on net sales of licensed products in licensed territories as provided by Janssen NV. The Company recognized royalty revenues in the period the sales occured.
The Company has sold its rights to receive certain royalties on sales of an HIV protease inhibitor (fosamprenavir) and recognizes the revenues related to this sale as royalty revenues. In the circumstance where the Company has sold its rights to future royalties under a license agreement and also maintains continuing involvement in the royalty arrangement (but not significant continuing involvement in the generation of the cash flows payable to the purchaser of the future royalty rights), the Company defers recognition of the proceeds it receives for the royalty stream and recognizes these deferred revenues over the life of the license agreement pursuant to the units-of-revenue method. The Company’s estimates regarding the estimated remaining royalty payments due to the purchaser have changed in the past and may change in the future.
Collaborative Revenues
The Company recognizes revenues generated through collaborative research, development and/or commercialization agreements. The terms of these agreements typically include payment to the Company of one or more of the following: nonrefundable, up-front license fees; development and commercial milestone payments; funding of research and/or
F-9
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
development activities; payments for services the Company provides through its third-party manufacturing network; and royalties on net sales of licensed products. Each of these types of payments results in collaborative revenues, except for revenues from royalties on net sales of licensed products, which are classified as royalty revenues.
For each collaborative research, development and/or commercialization agreement that result in revenues, the Company determines (i) whether multiple deliverables exist, (ii) whether the undelivered elements have value to the customer on a stand-alone basis, (iii) how the deliverables should be separated and (iv) how the consideration should be allocated to the deliverables. For arrangements entered into or materially modified after January 1, 2011, the Company allocates consideration in an arrangement using the relative selling price method based on management’s best estimate of selling price of deliverables if it does not have vendor-specific objective evidence or third-party evidence. As part of the accounting for these agreements, the Company must develop assumptions that require judgment to determine the best estimate of selling price. Key assumptions utilized by the Company to determine the best estimate of selling price may include forecasted revenues, patient enrollment requirements from regulatory authorities, development timelines, reimbursement rates for personnel costs, discount rates, and estimated third-party development costs.
The Company evaluates amendments to its existing arrangements to determine whether they have been materially modified. In making its determination that an arrangement has been materially modified, the Company considers whether there have been significant changes to the consideration under the arrangement, the deliverables under the arrangement, the timing of deliverables and the period of the arrangement. If the arrangement is determined to have been materially modified, the Company allocates fixed consideration under the arrangement using its best estimate of selling price to the remaining undelivered elements at the date of material modification. Any consideration remaining after the allocation is recognized as revenue.
Up-front License Fees: If the license to the Company’s intellectual property was determined to have stand-alone value from the other deliverables identified in the arrangement, the Company recognized revenues from nonrefundable, up-front license fees upon delivery. If these licenses did not have stand-alone value, the Company recognized revenues from nonrefundable, up-front license fees on a straight-line basis over the contracted or estimated period of performance. The Company evaluates the period of performance each reporting period and adjusts the period of performance on a prospective basis if there are changes to be made.
Milestone Payments: At the inception of each agreement that included research and development milestone payments, the Company evaluated whether each milestone was substantive. The Company recognized revenues related to substantive milestones in full in the period in which the substantive milestone is achieved if payment is reasonably assured. If a milestone is not considered substantive, the Company recognized the applicable milestone payment over the period of performance.
Research and Development Activities/Manufacturing Services: If the Company was entitled to reimbursement from its collaborators for specified research and development expenses and/or was entitled to payments for specified manufacturing services that the Company provided through its third-party manufacturing network, the Company determines whether the research and development funding would result in collaborative revenues or an offset to research and development expenses in accordance with the provisions of gross or net revenue presentation.
Concentration of Credit Risk
Financial instruments that potentially subject the Company to concentration of credit risk consist principally of money market funds and marketable securities. The Company places these investments with highly rated financial institutions, and, by policy, limits the amounts of credit exposure to any one financial institution. These amounts at times may exceed federally insured limits. The Company also maintains a foreign currency hedging program that includes foreign currency forward contracts with several counterparties. The Company has not experienced any credit losses related to these financial instruments and does not believe it is exposed to any significant credit risk related to these instruments.
The Company also is subject to credit risk from its accounts receivable related to its product sales and collaborators. The Company evaluates the creditworthiness of each of its customers and has determined that all of its material customers are creditworthy. To date, the Company has not experienced significant losses with respect to the collection of its accounts receivable. The Company’s receivables from Greece, Italy, Portugal and Spain were not material at December 31, 2016. The
F-10
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
Company believes that its allowance for doubtful accounts was adequate at December 31, 2016. Please refer to Note T, “Segment Information,” for further information.
Cash and Cash Equivalents
The Company considers all highly liquid investments with original maturities of three months or less at the date of purchase to be cash equivalents.
Marketable Securities
The Company’s marketable securities consist of investments in government-sponsored enterprise securities, corporate debt securities, corporate equity securities and commercial paper that are classified as available-for-sale. The Company classifies marketable securities available to fund current operations as current assets on its consolidated balance sheets. Marketable securities are classified as long-term assets on the consolidated balance sheets if (i) they have been in an unrealized loss position for longer than one year and (ii) the Company has the ability and intent to hold them (a) until the carrying value is recovered and (b) such holding period may be longer than one year. The Company’s marketable securities are stated at fair value with their unrealized gains and losses included as a component of accumulated other comprehensive income (loss), which is a separate component of shareholders’ equity, until such gains and losses are realized. The fair value of these securities is based on quoted prices for identical or similar assets.
The Company reviews investments in marketable securities for other-than-temporary impairment whenever the fair value of an investment is less than the amortized cost and evidence indicates that an investment’s carrying amount is not recoverable within a reasonable period of time. To determine whether an impairment is other-than-temporary, the Company considers whether it has an intent to sell, or whether it is more likely than not that the Company will be required to sell, the investment before recovery of the investment’s amortized cost basis. Evidence considered in this assessment includes reasons for the impairment, compliance with the Company’s investment policy, the severity and the duration of the impairment and changes in value subsequent to year-end. If a decline in the fair value is considered other-than-temporary, based on available evidence, the unrealized loss is transferred from other comprehensive income (loss) to the consolidated statements of operations.
Realized gains and losses are determined using the specific identification method and are included in other income (expense), net in the consolidated statements of operations.
Accounts Receivable
The Company deducts trade allowances for prompt payment and fees for distribution services from its accounts receivable based on its experience that the Company’s Customers will earn these discounts and fees. The Company’s estimates for its allowance for doubtful accounts, which have not been significant to date, are determined based on existing contractual payment terms and historical payment patterns.
Stock-based Compensation Expense
The Company expenses the fair value of employee stock options and other forms of stock-based employee compensation over the associated employee service period on a straight-line basis. Stock-based compensation expense is determined based on the fair value of the award at the grant date, net of estimated forfeitures, and is adjusted each period to reflect actual forfeitures and the outcomes of certain performance conditions.
For awards with performance conditions that accelerate vesting of the award, the Company estimates the likelihood of satisfaction of the performance conditions, which affects the period over which the expense is recognized, and recognizes the expense using the accelerated attribution model. For awards with performance conditions in which the award does not vest unless the performance condition is met, the Company recognizes expense if, and to the extent that, the Company estimates that achievement of the performance condition is probable. If the Company concludes that vesting is probable, it recognizes expense from the date it reaches this conclusion through the estimated vesting date.
Effective for equity awards granted on or after February 5, 2014, the Company provides to employees who have rendered a certain number of years’ to the Company and meet certain age requirements, partial or full acceleration of vesting
F-11
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
of these equity awards, subject to certain conditions including a notification period, upon a termination of employment other than for cause. Approximately 5% of the Company’s employees were eligible for partial or full acceleration of any of their equity awards as of December 31, 2016. The Company recognizes stock-based compensation expense related to these awards over a service period reflecting qualified employees eligibility for partial or full acceleration of vesting.
Research and Development Expenses
The Company expenses as incurred all research and development expenses, including amounts funded by research and development collaborations. The Company capitalizes nonrefundable advance payments made by the Company for research and development activities and expenses the payments as the related goods are delivered or the related services are performed.
Research and development expenses are comprised of costs incurred by the Company in performing research and development activities, including salary and benefits; stock-based compensation expense; laboratory supplies and other direct expenses; outsourced services, including clinical trial and pharmaceutical development costs; expenses associated with drug supplies that are not being capitalized; and infrastructure costs, including facilities costs and depreciation expense.
Advertising Expenses
The Company expenses the costs of advertising, including promotional expenses, as incurred. Advertising expenses, recorded in sales, general and administrative expenses, were $31.4 million, $24.5 million and $16.2 million in 2016, 2015 and 2014, respectively.
Inventories
The Company values its inventories at the lower-of-cost or market. The Company determines the cost of its inventories, which includes amounts related to materials and manufacturing overhead, on a first-in, first-out basis. The Company performs an assessment of the recoverability of capitalized inventory during each reporting period, and writes down any excess and obsolete inventories to their realizable value in the period in which the impairment is first identified. Shipping and handling costs incurred for inventory purchases are capitalized and recorded upon sale in cost of product revenues in the consolidated statements of operations. Shipping and handling costs incurred for product shipments are recorded as incurred in cost of product revenues in the consolidated statements of operations.
The Company capitalizes inventories produced in preparation for initiating sales of a drug candidate when the related drug candidate is considered to have a high likelihood of regulatory approval and the related costs are expected to be recoverable through sales of the inventories. In determining whether or not to capitalize such inventories, the Company evaluates, among other factors, information regarding the drug candidate’s safety and efficacy, the status of regulatory submissions and communications with regulatory authorities and the outlook for commercial sales, including the existence of current or anticipated competitive drugs and the availability of reimbursement. In addition, the Company evaluates risks associated with manufacturing the drug candidate and the remaining shelf-life of the inventories.
Property and Equipment
Property and equipment are recorded at cost. Depreciation expense is recorded using the straight-line method over the estimated useful life of the related asset, generally seven to ten years for furniture and equipment, three to five years for computers and software, 40 years for buildings and for leasehold improvements, the shorter of the useful life of the improvements or the estimated remaining life of the associated lease. Amortization expense of assets acquired under capital leases is included in depreciation expense. Maintenance and repairs to an asset that do not improve or extend its life are charged to operations. When assets are retired or otherwise disposed of, the assets and related accumulated depreciation are eliminated from the accounts and any resulting gain or loss is reflected in the Company’s consolidated statements of operations. The Company performs an assessment of the fair value of the assets if indicators of impairment are identified during a reporting period and records the assets at the lower of the net book value or the fair value of the assets.
The Company capitalizes internal costs incurred to develop software for internal use during the application development stage. The Company expenses costs related to the planning and post-implementation phases of development of software for
F-12
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
internal use as these costs are incurred. Maintenance and enhancement costs (including costs in the post-implementation stages) are expensed as incurred, unless such costs relate to substantial upgrades and enhancements to the software resulting in added functionality, in which case the costs are capitalized. Amortization of capitalized internally developed software costs is recorded in depreciation expense over the useful life of the related asset.
The Company records certain construction costs incurred by a landlord as an asset and a corresponding financing obligation on the Company’s consolidated balance sheets when the Company is determined to be the owner of the buildings during construction for accounting purposes. Upon completion of the project, the Company performs a sale-leaseback analysis to determine if the Company can remove the assets from its consolidated balance sheet.
Capital Leases
The assets and liabilities associated with capital lease agreements are recorded at the present value of the minimum lease payments at the inception of the lease agreement. The assets are depreciated using the straight-line method over the shorter of the useful life of the related asset or the remaining life of the associated lease. Amortization of assets that the Company leases pursuant to a capital lease is included in depreciation expense. The Company performs an assessment of the fair value of the assets if indicators of impairment are identified during a reporting period and records the assets at the lower of the net book value or the fair value of the assets. Assets recorded under capital leases are recorded within “Property and equipment, net” and liabilities related to those assets are recorded within “Capital lease obligations, current portion” and “Capital lease obligations, excluding current portion” on the Company’s consolidated balance sheets.
Income Taxes
Deferred tax assets and liabilities are recognized for the expected future tax consequences of temporary differences between the financial statement carrying amounts and the income tax bases of assets and liabilities. A valuation allowance is applied against any net deferred tax asset if, based on the available evidence, it is more likely than not that some or all of the deferred tax assets will not be realized.
The Company records liabilities related to uncertain tax positions by prescribing a minimum recognition threshold and measurement attribute for the financial statement recognition and measurement of a tax position taken or expected to be taken in a tax return. The Company does not believe any such uncertain tax positions currently pending will have a material adverse effect on its consolidated financial statements.
Variable Interest Entities
The Company reviews each collaboration agreement pursuant to which the Company licenses assets owned by a collaborator in order to determine whether or not the Company has a variable interest via the license agreement with the collaborator and if the variable interest is a variable interest in the collaborator as a whole. In assessing whether the Company has a variable interest in the collaborator as a whole, the Company considers and makes judgments regarding the purpose and design of the entity, the value of the licensed assets to the collaborator and the significant activities of the collaborator. If the Company has a variable interest in the collaborator as a whole, the Company assesses whether or not the Company is the primary beneficiary of that VIE based on a number of factors, including (i) which party has the power to direct the activities that most significantly affect the VIE’s economic performance, (ii) the parties’ contractual rights and responsibilities pursuant to the collaboration agreement and (iii) which party has the obligation to absorb losses of or the right to receive benefits from the VIE that could be significant to the VIE. If the Company determines it is the primary beneficiary of a VIE at the onset of the collaboration agreement, the collaboration is treated as a business combination and the Company consolidates the financial statements of the VIE into the Company’s consolidated financial statements. The Company evaluates whether it continues to be the primary beneficiary of any consolidated VIEs on a quarterly basis. If the Company determines that it is no longer the primary beneficiary of a consolidated VIE, or no longer has a variable interest in the VIE, it deconsolidates the VIE in the period that the determination is made.
Assets recorded as a result of consolidating VIEs’ financial results into the Company’s consolidated balance sheet do not represent additional assets that could be used to satisfy claims against the Company’s general assets. With respect to the Company’s VIEs, the VIEs’ assets are not significant, except for the VIEs’ cash and cash equivalents. The Company records the cash and cash equivalents of consolidated VIEs as restricted cash because the Company does not have control over the
F-13
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
VIEs’ cash and cash equivalents. The Company also has recorded the liabilities of its consolidated VIEs for which creditors do not have recourse to the Company’s general assets outside of the VIE.
Fair Value of In-process Research and Development Assets and Contingent Payments
The present-value models used to estimate the fair values of research and development assets and contingent payments pursuant to collaborations incorporate significant assumptions, including: assumptions regarding the probability of obtaining marketing approval and/or achieving relevant development milestones for a drug candidate; estimates regarding the timing of and the expected costs to develop a drug candidate; estimates of future cash flows from potential product sales and/or the potential to achieve certain commercial milestones with respect to a drug candidate; and the appropriate discount and tax rates.
In-process Research and Development Assets
The Company records the fair value of in-process research and development assets as of the transaction date of a business combination. Each of these assets is accounted for as an indefinite-lived intangible asset and is maintained on the Company’s consolidated balance sheet until either the project underlying it is completed or the asset becomes impaired. If the asset becomes impaired or is abandoned, the carrying value of the related intangible asset is written down to its fair value, and an impairment charge is recorded in the period in which the impairment occurs. If a project is completed, the carrying value of the related intangible asset is amortized as a part of cost of product revenues over the remaining estimated life of the asset beginning in the period in which the project is completed. In-process research and development assets are tested for impairment on an annual basis as of October 1, and more frequently if indicators are present or changes in circumstances suggest that impairment may exist.
Goodwill
The difference between the purchase price and the fair value of assets acquired and liabilities assumed in a business combination is allocated to goodwill. Goodwill is evaluated for impairment on an annual basis as of October 1, and more frequently if indicators are present or changes in circumstances suggest that impairment may exist.
Noncontrolling Interest
The Company records noncontrolling interest, which has historically related to consolidated VIEs, on its consolidated balance sheets. The Company records net loss (income) attributable to noncontrolling interest on its consolidated statements of operations, reflecting the VIEs’ net loss (income) for the reporting period, adjusted for changes in the noncontrolling interest holders’ claim to net assets, including contingent milestone, royalty and option payments, is evaluated each reporting period.
Deconsolidation and Discontinued Operations
Upon the occurrence of certain events and on a regular basis, the Company evaluates whether it no longer has a controlling interest in its subsidiaries, including consolidated VIEs. If the Company determines it no longer has a controlling interest, the subsidiary is deconsolidated. The Company records a gain or loss on deconsolidation based on the difference on the deconsolidation date between (i) the aggregate of (a) the fair value of any consideration received, (b) the fair value of any retained noncontrolling investment in the former subsidiary and (c) the carrying amount of any noncontrolling interest in the subsidiary being deconsolidated, less (ii) the carrying amount of the former subsidiary’s assets and liabilities.
The Company assesses whether a deconsolidation is required to be presented as discontinued operations in its consolidated financial statements on the deconsolidation date. This assessment is based on whether or not the deconsolidation represents a strategic shift that has or will have a major effect on the Company’s operations or financial results. If the Company determines that a deconsolidation requires presentation as a discontinued operation on the deconsolidation date, or at any point during the one year period following such date, it will present the former subsidiary as a discontinued operation in current and comparative period financial statements.
F-14
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
Derivative Instruments, Embedded Derivatives and Hedging Activities
The Company has entered into financial transactions involving free-standing derivative instruments and embedded derivatives in the past. Embedded derivatives are required to be bifurcated from the host instruments if the derivatives are not clearly and closely related to the host instruments. The Company determines the fair value of each derivative instrument or embedded derivative that is identified on the date of issuance and at the end of each quarterly period. The estimates of the fair value of the derivatives include significant assumptions regarding the estimates market participants would make in order to evaluate these derivatives.
The Company recognizes the fair value of hedging instruments that are designated and qualify as hedging instruments pursuant to GAAP, primarily foreign currency forward contracts, as either assets or liabilities on the consolidated balance sheets. Changes in the fair value of hedging instruments are recorded each period in accumulated other comprehensive income (loss) as unrealized gains and losses until the forecasted underlying transaction occurs. Unrealized gains and losses on these foreign currency forward contracts are included in (i) “Prepaid expenses and other current assets,” (ii) Other assets,” (iii) “Other liabilities, current portion” and (iv) “Other liabilities, excluding current portion,” respectively, on the Company’s consolidated balance sheets. Realized gains and losses for the effective portion of such contracts are recognized in “Product revenues, net” in the consolidated statement of operations when the contract is settled with the counterparty. The Company classifies the cash flows from hedging instruments in the same category as the cash flows from the hedged items.
Certain of the Company’s hedging instruments are subject to master netting arrangements to reduce the risk arising from such transactions with its counterparties. The Company presents unrealized gains and losses on its foreign currency forward contracts on a gross basis within its consolidated balance sheets.
The Company assesses, both at inception and on an ongoing basis, whether the foreign currency forward contracts used in hedging transactions are highly effective in offsetting the changes in cash flows of the hedged items. The Company also assesses hedge ineffectiveness quarterly and, if determined to be ineffective, records the gain or loss related to the ineffective portion to earnings in “Other income (expense), net” in its consolidated statements of operations.
Restructuring Expenses
The Company records costs and liabilities associated with exit and disposal activities based on estimates of fair value in the period the liabilities are incurred. In periods subsequent to the initial measurement, the Company measures changes to the liability using the credit-adjusted risk-free discount rate applied in the initial period. The Company evaluates and adjusts these liabilities as appropriate for changes in circumstances at least on a quarterly basis.
Comprehensive Income (Loss)
Comprehensive income (loss) consists of net income (loss) and other comprehensive income (loss), which includes foreign currency translation adjustments and unrealized gains and losses on foreign currency forward contracts and certain marketable securities. For purposes of comprehensive income (loss) disclosures, the Company records tax provisions or benefits related to the unrealized gains and losses on foreign currency forward contracts and certain marketable securities. The Company does not record tax provisions or benefits related to the cumulative translation adjustment, as the Company intends to permanently reinvest undistributed earnings in its foreign subsidiaries.
Foreign Currency Translation and Transactions
The Company primarily operates with entities that have the U.S. dollar denominated as their functional currency. Non-U.S. dollar denominated functional currency subsidiaries have assets and liabilities translated into U.S. dollars at rates of exchange in effect at the end of the year. Revenue and expense amounts are translated using the average exchange rates for the period. Net unrealized gains and losses resulting from foreign currency translation are included in accumulated other comprehensive income (loss), which is a separate component of shareholders’ equity. Included in accumulated other comprehensive income (loss) are net unrealized losses related to foreign currency translation of $7.9 million, $2.1 million and $1.0 million at December 31, 2016, 2015 and 2014, respectively. Net foreign currency exchange transaction gains or losses are included in “net loss” on the Company’s consolidated statement of operations. Net transaction gains were $4.0 million for 2016 and net transaction losses were $6.8 million and $6.4 million for 2015 and 2014, respectively.
F-15
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
Net Loss Per Share Attributable to Vertex Common Shareholders
Basic and diluted net loss per share attributable to Vertex common shareholders are presented in conformity with the two-class method required for participating securities. Under the two-class method, earnings are allocated to (i) Vertex common shares, excluding unvested restricted stock, and (ii) participating securities, based on their respective weighted-average shares outstanding for the period. Shares of unvested restricted stock granted under the Company’s Amended and Restated 2006 Stock and Option Plan have the non-forfeitable right to receive dividends on an equal basis with other outstanding common stock. As a result, these unvested shares of restricted stock are considered participating securities under the two-class method. Potentially dilutive shares result from the assumed exercise of outstanding stock options (the proceeds of which are then assumed to have been used to repurchase outstanding stock using the treasury stock method).
Basic net loss per share attributable to Vertex common shareholders is based upon the weighted-average number of common shares outstanding during the period, excluding restricted stock that has been issued but is not yet vested. Diluted net loss per share attributable to Vertex common shareholders is based upon the weighted-average number of common shares outstanding during the period plus additional weighted-average common equivalent shares outstanding during the period when the effect is dilutive.
The Company utilizes income (loss) from continuing operations attributable to Vertex to determine whether potentially outstanding stock options and the assumed conversion of convertible notes are dilutive.
Recent Accounting Pronouncements
In 2014, the Financial Accounting Standards Board (“FASB”) issued amended guidance applicable to revenue recognition that will be effective for the year ending December 31, 2018. Early adoption is permitted for the year-ending December 31, 2017. The new guidance applies a more principles based approach to recognizing revenue. Under the new guidance, revenue is recognized when a customer obtains control of promised goods or services and is recognized in an amount that reflects the consideration that an entity expects to receive in exchange for those goods or services. In addition, the standard requires disclosure of the nature, amount, timing, and uncertainty of revenue and cash flows arising from contracts with customers. The new guidance must be adopted using either a modified retrospective approach or a full retrospective approach for all periods presented. Under the modified retrospective method, the cumulative effect of applying the standard would be recognized at the date of initial application within retained earnings. Under the full retrospective approach, the standard would be applied to each prior reporting period presented. The Company expects to adopt the new guidance using the modified retrospective method. The Company is in the process of evaluating the new guidance and determining whether the expected effect is material to its consolidated financial statements. The process includes identifying and analyzing the impact of the standard by reviewing the Company’s current accounting policies and practices to identify potential differences that would result from applying the requirements of the new standard to each revenue contract associated with all of the Company’s revenue streams. The new guidance could impact the Company’s accounting for product shipments to countries through early access programs, for example the French early access programs, whereby the associated product has received regulatory approval but the reimbursement rate has not been finalized.
In 2016, the FASB issued amended guidance applicable to leases that will be effective for the year ending December 31, 2019. Early adoption is permitted. This update requires an entity to recognize assets and liabilities for leases with lease terms of more than 12 months on the balance sheet. The Company is in the process of evaluating the new guidance and determining the expected effect on its consolidated financial statements.
In 2016, the FASB issued amended guidance applicable to share-based compensation to employees that will be effective for the year ending December 31, 2017 with early adoption permitted. This guidance simplifies the accounting for employee share-based payment transactions, including the accounting for income taxes, forfeitures, and statutory tax withholding requirements, as well as classification in the statement of cash flows. The Company plans to change how it accounts for forfeitures upon adoption and is currently quantifying the adjustment to be recorded to retained earnings related to this change in policy. In addition, the Company will include certain net operating losses and tax credits, offset with a full valuation allowance, as a component of deferred taxes upon adoption of this guidance that were previously not included in deferred taxes. The Company will also record a corresponding valuation allowance against these increased net operating loss carryforwards upon adoption of this new guidance.
F-16
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
In 2016, the FASB issued amended guidance for the classification of certain cash receipts and cash payments on the statement of cash flows to reduce existing diversity in practice. The new accounting guidance is effective for the year ending December 31, 2017. Early adoption is permitted. The Company does not expect a significant effect on its consolidated financial statements upon adoption of this new guidance.
In January 2017, the FASB issued amended guidance related to business combinations. The new guidance clarifies the definition of a business with the objective of adding guidance to assist entities with evaluating whether transactions should be accounted for as acquisitions (or disposals) of assets or businesses. The new accounting guidance is effective for annual periods beginning after December 15, 2017, including interim periods within those periods. Early adoption is permitted. The Company plans to apply this new guidance to future acquisitions.
In 2014, the FASB issued new guidance on management’s responsibility in evaluating whether or not there is substantial doubt about a company’s ability to continue as a going concern within one year from the date the financial statements are issued each reporting period. This new accounting guidance became effective for the Company for the year ended December 31, 2016. The adoption of the new guidance did not have a material effect on the Company’s consolidated financial statements.
B. | Collaborative Arrangements |
Cystic Fibrosis Foundation Therapeutics Incorporated
The Company has a research, development and commercialization agreement with Cystic Fibrosis Foundation Therapeutics Incorporated (“CFFT”) that was originally entered into in May 2004, and was most recently amended on October 13, 2016 (the “2016 Amendment”). Pursuant to the agreement, as amended, the Company has agreed to pay royalties ranging from low single digits to mid-single digits on potential sales of certain compounds first synthesized and/or tested between March 1, 2014 and August 31, 2016 and tiered royalties ranging from single digits to sub-teens on any approved drugs first synthesized and/or tested during a research term on or before February 28, 2014, including KALYDECO (ivacaftor), ORKAMBI (lumacaftor in combination with ivacaftor), lumacaftor and tezacaftor. For combination products, such as ORKAMBI, sales will be allocated equally to each of the active pharmaceutical ingredients in the combination product.
In each of the fourth quarter of 2015 and first quarter of 2016, CFFT earned a commercial milestone payment of $13.9 million from the Company upon achievement of certain sales levels of lumacaftor. There are no additional commercial milestone payments payable by the Company to CFFT pursuant to the agreement. Pursuant to the 2016 Amendment, the CFFT provided the Company an upfront payment of $75.0 million and agreed to provide development funding to the Company of up to $6.0 million annually. The upfront payment plus any future development funding represent a form of financing pursuant to Accounting Standards Codification (ASC) 730, Research and Development, and thus the amounts are recorded as a liability on the consolidated balance sheet, primarily reflected in Other liabilities, excluding current portion. The liability is reduced over the estimated royalty term of the agreement. Reductions in the liability are reflected as an offset to cost of product revenues and as interest expense.
The Company began marketing KALYDECO in the United States and certain countries in the European Union in 2012 and began marketing ORKAMBI in the United States in 2015. The Company received approval for ORKAMBI In the European Union in 2015 and in Canada and Australia in 2016. The Company has royalty obligations to CFFT for ivacaftor, lumacaftor and tezacaftor until the expiration of patents covering those compounds. The Company has patents in the United States and European Union covering the composition-of-matter of ivacaftor that expire in 2027 and 2025, respectively, subject to potential patent extensions. The Company has patents in the United States and European Union covering the composition-of-matter of lumacaftor that expire in 2030 and 2026, respectively, subject to potential extension. The Company has patents in the United States and European Union covering the composition-of-matter of tezacaftor that expire in 2027 and 2028, respectively, subject to potential extension.
F-17
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
CRISPR Therapeutics AG
On October 26, 2015, the Company entered into a strategic collaboration, option and license agreement (the “CRISPR Agreement”) with CRISPR Therapeutics AG and its affiliates (“CRISPR”) to collaborate on the discovery and development of potential new treatments aimed at the underlying genetic causes of human diseases using CRISPR-Cas9 gene editing technology. The Company has the exclusive right to license up to six CRISPR-Cas9-based targets. In connection with the CRISPR Agreement, the Company made an upfront payment to CRISPR of $75.0 million and a $30.0 million investment in CRISPR pursuant to a convertible loan agreement that converted into preferred stock in January 2016. The Company expensed $75.0 million to research and development, and the $30.0 million investment was recorded at cost and was classified as a long-term asset on the Company’s consolidated balance sheet in 2015. In the second quarter of 2016, the Company made an additional preferred stock investment in CRISPR of approximately $3.1 million. In connection with CRISPR's initial public offering in October 2016, the Company purchased $10.0 million of common shares at the public offering price and the Company's preferred stock investments in CRISPR converted into common shares. Pursuant to the terms of a lockup agreement between the Company and the underwriters of CRISPR’s initial public offering, the Company agreed not to sell or otherwise dispose of its shares in CRISPR through April 17, 2017. As of December 31, 2016, the Company recorded the CRISPR common shares it holds at fair value and included the fair value of the common shares in its marketable securities and the unrecognized gain related to these common shares in accumulated other comprehensive income (loss) on the consolidated balance sheet.
The Company will fund all of the discovery activities conducted pursuant to the CRISPR Agreement. For potential hemoglobinapathy treatments, including treatments for sickle cell disease, the Company and CRISPR will share equally all research and development costs and worldwide revenues. For other targets that the Company elects to license, the Company would lead all development and global commercialization activities. For each of up to six targets that the Company elects to license, other than hemoglobinapathy targets, CRISPR has the potential to receive up to $420.0 million in development, regulatory and commercial milestones and royalties on net product sales.
The Company may terminate the CRISPR Agreement upon 90 days’ notice to CRISPR prior to any product receiving marketing approval or upon 270 days’ notice after a product has received marketing approval. The CRISPR Agreement also may be terminated by either party for a material breach by the other, subject to notice and cure provisions. Unless earlier terminated, the CRISPR Agreement will continue in effect until the expiration of the Company’s payment obligations under the CRISPR Agreement.
Variable Interest Entities (VIE)
The Company has entered into several agreements pursuant to which it has licensed rights to certain drug candidates from third-party collaborators, which has resulted in the consolidation of the third parties' financial statements into the Company's consolidated financial statements as VIEs. In order to account for the fair value of the contingent payments, which consist of milestone, royalty and option payments, related to these collaborations under GAAP, the Company uses present-value models based on assumptions regarding the probability of achieving the relevant milestones, estimates regarding the timing of achieving the milestones, estimates of future product sales and the appropriate discount rates. The Company bases its estimate of the probability of achieving the relevant milestones on industry data for similar assets and its own experience. The discount rates used in the valuation model represent a measure of credit risk and market risk associated with settling the liabilities. Significant judgment is used in determining the appropriateness of these assumptions at each reporting period. Changes in these assumptions could have a material effect on the fair value of the contingent payments. The following collaborations are reflected in the Company's financial statements as consolidated VIEs:
Parion Sciences, Inc.
License and Collaboration Agreement
In June 2015, the Company entered into a strategic collaboration and license agreement (the “Parion Agreement”) with Parion Sciences, Inc. (“Parion”). Pursuant to the agreement, the Company is collaborating with Parion to develop investigational epithelial sodium channel (“ENaC”) inhibitors, including VX-371 (formerly P-1037) and VX-551 (formerly P-1055), for the potential treatment of CF and all other pulmonary diseases. The Company is leading development activities
F-18
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
for VX-371 and VX-551 and is responsible for all costs, subject to certain exceptions, related to development and commercialization of the compounds.
Pursuant to the Parion Agreement, the Company has worldwide development and commercial rights to Parion’s lead investigational ENaC inhibitors, VX-371 and VX-551, for the potential treatment of CF and all other pulmonary diseases and has the option to select additional compounds discovered in Parion’s research program. Parion received an $80.0 million up-front payment and has the potential to receive up to an additional (i) $490.0 million in development and regulatory milestone payments for development of ENaC inhibitors in CF, including $360.0 million related to global filing and approval milestones, (ii) $370.0 million in development and regulatory milestones for VX-371 and VX-551 in non-CF pulmonary indications and (iii) $230.0 million in development and regulatory milestones should the Company elect to develop an additional ENaC inhibitor from Parion’s research program. The Company has agreed to pay Parion tiered royalties that range from the low double digits to mid-teens as a percentage of potential sales of licensed products. In the second quarter of 2016, Parion earned a milestone payment of $5.0 million based upon the achievement of a specified milestone under the Parion Agreement.
The Company may terminate the Parion Agreement upon 90 days’ notice to Parion prior to any licensed product receiving marketing approval or upon 180 days’ notice after a licensed product has received marketing approval. If the Company experiences a change of control prior to the initiation of the first Phase 3 clinical trial for a licensed product, Parion may terminate the Parion Agreement upon 30 days’ notice, subject to the Company’s right to receive specified royalties on any subsequent commercialization of licensed products. The Parion Agreement also may be terminated by either party for a material breach by the other, subject to notice and cure provisions. Unless earlier terminated, the Parion Agreement will continue in effect until the expiration of the Company’s royalty obligations, which expire on a country-by-country basis on the later of (i) the date the last-to-expire patent covering a licensed product expires or (ii) ten years after the first commercial sale in the country.
The Company determined that it has a variable interest in Parion via the Parion Agreement, and that the variable interest represents a variable interest in Parion as a whole since the fair value of the ENaC inhibitors represents more than half of the total fair value of Parion’s assets. The Company also concluded that it is the primary beneficiary as it has the power to direct the activities that most significantly affect the economic performance of Parion and it has the obligation to absorb losses and right to receive benefits that potentially could be significant to Parion. Accordingly, the Company consolidated Parion's financial statements beginning on June 4, 2015. However, the Company's interests in Parion are limited to those accorded to the Company in the Parion Agreement.
Consideration for the Parion Agreement
The Company determined that the fair value of the consideration to be transferred from the Company to Parion was $255.3 million as of June 4, 2015, which consisted of (i) an $80.0 million up-front payment, (ii) the estimated fair value of the contingent research and development milestones and (iii) the estimated fair value of potential royalty payments. The Company valued the contingent payments using (a) discount rates ranging from 4.1% to 5.9% for the development milestones and (b) a discount rate of 6.6% for royalties. The up-front payment made and the fair value of the contingent payments payable by the Company pursuant to the Parion Agreement are set forth in the table below:
June 4, 2015 | |||
(in thousands) | |||
Up-front payment | $ | 80,000 | |
Fair value of contingent payments | 175,340 | ||
Total | $ | 255,340 | |
F-19
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
Allocation of Assets and Liabilities
The Company recorded the fair value of the assets and liabilities of Parion on the effective date of the agreement as follows:
June 4, 2015 | |||
(in thousands) | |||
Consideration transferred | $ | — | |
Noncontrolling interest | 164,317 | ||
Intangible assets | (255,340 | ) | |
Net other liabilities | 10,468 | ||
Deferred tax liability | 91,023 | ||
Goodwill | $ | 10,468 | |
While there was a transfer of $80.0 million to Parion, the cash remained within the Company’s consolidated financial statements since Parion is part of the consolidated entity. The cash is classified as restricted cash and cash equivalents (VIE) within the consolidated balance sheet as it is attributed to the noncontrolling interest holders of Parion. When determining the valuation of goodwill, the fair value of consideration for the license is zero since there was no consideration transferred outside the consolidated financial statements. The Company recorded $255.3 million of intangible assets on the Company's consolidated balance sheets for Parion's in-process research and development assets. These in-process research and development assets relate to Parion's pulmonary ENaC platform, including the intellectual property related to VX-371 and VX-551, that are licensed by Parion to the Company. The Company also recorded the fair value of the net assets attributable to noncontrolling interest and deferred tax liability resulting from a basis difference in the intangible assets and certain other net liabilities held by Parion. The difference between the fair values of the consideration and noncontrolling interest and the fair value of Parion’s net assets was recorded as goodwill.
BioAxone Biosciences, Inc.
In October 2014, the Company entered into a license and collaboration agreement (the “BioAxone Agreement”) with BioAxone Biosciences, Inc. (“BioAxone”), which resulted in the consolidation of BioAxone as a VIE beginning on October 1, 2014. The Company determined that BioAxone is a VIE based on, among other factors, the significance to BioAxone of VX-210, which was licensed to the Company pursuant to the BioAxone Agreement, and on the Company’s power to direct the activities that most significantly affect the economic performance of BioAxone. Accordingly, the Company consolidated BioAxone’s financial statements beginning in October 2014. The Company paid BioAxone initial payments of $10.0 million in the fourth quarter of 2014.
BioAxone has the potential to receive up to $90.0 million in milestones and fees, including development, regulatory and milestone payments and a license continuation fee. In addition, BioAxone would receive royalties and commercial milestones on future net product sales of VX-210, if any. The Company recorded an in-process research and development intangible asset of $29.0 million for VX-210 and a corresponding deferred tax liability of $11.3 million attributable to BioAxone. The Company holds an option to purchase BioAxone at a predetermined price. The option expires on the earliest of (a) the day the FDA accepts the Biologics License Application submission for VX-210, (b) the day the Company elects to continue the license instead of exercising the option to purchase BioAxone and (c) March 15, 2018, subject to the Company’s option to extend this date by one year.
F-20
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
Alios BioPharma, Inc.
In 2011, the Company entered into a license and collaboration agreement (the “Alios Agreement”) with Alios, which terminated in the fourth quarter of 2014. Pursuant to the Alios Agreement, which resulted in the consolidation of Alios as a VIE through December 31, 2013, the Company and Alios collaborated on the research, development and commercialization of HCV nucleotide analogues discovered by Alios. As of September 30, 2014, the Company concluded that it no longer had significant continuing involvement with Alios due to its intent and ability to terminate the Alios Agreement, among other factors, and the operations of Alios are presented as discontinued operations in these consolidated financial statements for 2014.
Aggregate VIE Financial Information
An aggregate summary of net loss attributable to noncontrolling interest related to the Company’s VIEs for the three years ended December 31, 2016 was as follows:
2016 | 2015 | 2014 | |||||||||
(in thousands) | |||||||||||
Loss attributable to noncontrolling interest before provision for income taxes | $ | 10,086 | $ | 6,646 | $ | 764 | |||||
Provision for income taxes | 16,743 | 29,731 | 3,876 | ||||||||
Increase in fair value of contingent payments | (54,850 | ) | (4,530 | ) | (450 | ) | |||||
Net (income) loss attributable to noncontrolling interest | $ | (28,021 | ) | $ | 31,847 | $ | 4,190 | ||||
During the years ended December 31, 2016 and 2015, the noncontrolling interest holders’ claim to net assets with respect to the contingent payments related to the Parion Agreement, increased by $64.8 million and $3.6 million, respectively. The increase in the fair value of the contingent payments related to the Parion Agreement in 2016 was primarily due to a Phase 2 clinical trial of VX-371 achieving its primary safety endpoint in the second quarter of 2016 offset by a payment of $5.0 million related to the achievement of a specified milestone under the Parion Agreement. The changes in the fair value of the contingent payments were also due to the changes in market interest rates and the time value of money. As of December 31, 2016 and 2015, the fair value of the contingent payments related to the Parion Agreement was $238.8 million and $179.0 million, respectively.
During the year ended December 31, 2016, the noncontrolling interest holders’ claim to net assets with respect to the contingent payments related to the BioAxone Agreement decreased by $10.0 million. The decrease in the fair value of the contingent payments was due to changes in certain assumptions used in establishing the fair value including revenue assumptions and the development timeline. During the year ended December 31, 2015 and 2014, the fair value of the contingent payments related to the BioAxone Agreement increased by $0.9 million and $0.5 million, respectively. As of December 31, 2016 and 2015, the fair value of the contingent payments related to the BioAxone Agreement was $18.0 million and $28.0 million, respectively.
F-21
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
The following table summarizes items related to the Company’s VIEs included in the Company’s consolidated balance sheets as of the dates set forth in the table:
December 31, 2016 | December 31, 2015 | ||||||
(in thousands) | |||||||
Restricted cash and cash equivalents (VIE) | $ | 47,762 | $ | 78,910 | |||
Prepaid expenses and other current assets | 6,812 | 3,138 | |||||
Intangible assets | 284,340 | 284,340 | |||||
Goodwill | 19,391 | 19,391 | |||||
Other assets | 399 | 455 | |||||
Accounts payable | 415 | 676 | |||||
Taxes payable | 1,330 | 24,554 | |||||
Other current liabilities | 2,137 | 7,100 | |||||
Deferred tax liability, net | 131,446 | 110,438 | |||||
Other liabilities | 300 | 300 | |||||
Noncontrolling interest | 181,609 | 153,661 | |||||
The Company has recorded the VIEs’ cash and cash equivalents as restricted cash and cash equivalents (VIE) because (i) the Company does not have any interest in or control over the VIEs’ cash and cash equivalents and (ii) the Company’s agreements with each VIE do not provide for the VIEs’ cash and cash equivalents to be used for the development of the assets that the Company licensed from the applicable VIE. Assets recorded as a result of consolidating the Company’s VIEs’ financial condition into the Company’s balance sheets do not represent additional assets that could be used to satisfy claims against the Company’s general assets.
Other Collaborations
The Company has entered into various agreements pursuant to which it collaborates with third parties, including inlicensing and outlicensing arrangements. Although the Company does not consider any of these arrangements to be material, the most notable of these arrangements are described below.
Moderna Therapeutics, Inc.
In July 2016, the Company entered into a strategic collaboration and licensing agreement (the "Moderna Agreement") with Moderna Therapeutics, Inc. ("Moderna") pursuant to which the parties are seeking to identify and develop messenger Ribonucleic Acid ("mRNA") Therapeutics for the treatment of CF. In connection with the Moderna Agreement in the third quarter of 2016, the Company made an upfront payment to Moderna of $20.0 million and a $20.0 million cost-method investment in Moderna pursuant to a convertible promissory note that converted into preferred stock in August 2016. Moderna has the potential to receive future development and regulatory milestones of up to $275.0 million, including $220.0 million in approval and reimbursement milestones, as well as tiered royalty payments on future sales.
Under the terms of the Moderna Agreement, Moderna will lead discovery efforts and the Company will lead all preclinical, development and commercialization activities associated with the advancement of mRNA Therapeutics that result from this collaboration and will fund all expenses related to the collaboration.
The Company may terminate the Moderna Agreement by providing advanced notice to Moderna, with the required length of notice dependent on whether any product developed under the Moderna Agreement has received marketing approval. The Moderna Agreement also may be terminated by either party for a material breach by the other, subject to notice and cure provisions. Unless earlier terminated, the Moderna Agreement will continue in effect until the expiration of the Company's payment obligations under the Moderna Agreement.
F-22
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
Janssen Pharmaceuticals, Inc.
In June 2014, the Company entered into an agreement (the “Janssen Influenza Agreement”) with Janssen Pharmaceuticals, Inc. (“Janssen Inc.”), which was amended in October 2014 to clarify certain roles and responsibilities of the parties.
Pursuant to the Janssen Influenza Agreement, Janssen Inc. has an exclusive worldwide license to develop and commercialize certain drug candidates for the treatment of influenza, including JNJ-3872 (formerly VX-787). The Company received non-refundable payments of $35.0 million from Janssen Inc. in 2014, which were recorded as collaborative revenues. The Company has the potential to receive development, regulatory and commercial milestone payments as well as royalties on future product sales, if any.
Janssen Inc. is responsible for costs related to the development and commercialization of the compounds. The Company recorded reimbursement for these development activities of $14.7 million, $22.8 million and $9.1 million in 2016, 2015 and 2014, respectively. The reimbursements are recorded as a reduction to development expense in the Company’s consolidated statements of operations primarily due to the fact that Janssen Inc. directs the activities and selects the suppliers associated with these activities. Janssen Inc. may terminate the Janssen Influenza Agreement, subject to certain exceptions, upon six months’ notice.
Janssen Pharmaceutica NV
The Company has a collaboration agreement (the “Janssen HCV Agreement”) with Janssen Pharmaceutica NV (“Janssen NV”) for the development, manufacture and commercialization of telaprevir, which Janssen NV began marketing under the brand name INCIVO in certain of its territories in September 2011. Pursuant to the Janssen HCV Agreement, as amended, Janssen NV has a fully-paid license to manufacture and commercialize INCIVO in its territories including Europe, South America, the Middle East, Africa and Australia, subject to the payment of third-party royalties on net sales of INCIVO. In addition to the collaborative revenues, the Company recorded royalty revenues and corresponding royalty expenses related to third-party royalties that Janssen NV remains responsible for based on INCIVO net sales.
During the three years ended December 31, 2016, the Company recognized the following revenues attributable to the Janssen HCV collaboration:
2016 | 2015 | 2014 | |||||||||
(in thousands) | |||||||||||
Royalty revenues | $ | 71 | $ | 1,518 | $ | 13,481 | |||||
Collaborative revenues | $ | (155 | ) | $ | 1,946 | $ | 7,104 | ||||
Total revenues attributable to the Janssen HCV collaboration | $ | (84 | ) | $ | 3,464 | $ | 20,585 | ||||
Subsequent Event
Merck KGaA
On January 10, 2017, the Company entered into a Strategic Collaboration and License Agreement (the “Merck KGaA Agreement”) with Merck KGaA, Darmstadt, Germany (“Merck KGaA”). Pursuant to the Merck KGaA Agreement, the Company granted Merck KGaA an exclusive worldwide license to research, develop and commercialize four oncology research and development programs. Under the Merck KGaA Agreement, the Company granted Merck KGaA exclusive, worldwide rights to our two clinical-stage programs targeting DNA damage repair: its ataxia telangiectasia and Rad3-related protein inhibitor program, including VX-970 and VX-803, and its DNA-dependent protein kinase inhibitor program, including VX-984. In addition, the Company granted Merck KGaA exclusive, worldwide rights to two pre-clinical programs.
Under the Merck KGaA Agreement, the Company will receive an up-front payment of $230.0 million. In addition, it will receive tiered royalties on potential sales of licensed products, calculated as a percentage of net sales, that range from (i)
F-23
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
mid-single digits to mid-twenties for clinical-stage programs and (ii) mid-single digits to high single digits for the pre-clinical research programs. Merck KGaA will assume full responsibility for development and commercialization costs for all programs. The licenses granted pursuant to the Merck KGaA Agreement and the up-front payment are subject to the expiration of the waiting period under the Hart-Scott-Rodino Antitrust Improvements Act.
Merck KGaA may terminate the Merck KGaA Agreement or any individual program by providing 90 days’ notice, or, in the case of termination of a program with a product that has received marketing approval, 180 days’ notice. The Merck KGaA Agreement may also be terminated by either party for a material breach by the other party, subject to notice and cure provisions. Unless earlier terminated, the Merck KGaA Agreement will continue in effect until the date on which the royalty term and all payment obligations with respect to all products in all countries have expired.
C. | Earnings Per Share |
Basic net loss per share attributable to Vertex common shareholders is based upon the weighted-average number of common shares outstanding during the period, excluding restricted stock and restricted stock units that have been issued but are not yet vested. Diluted net loss per share attributable to Vertex common shareholders is based upon the weighted-average number of common shares outstanding during the period plus additional weighted-average common equivalent shares outstanding during the period when the effect is dilutive.
The Company did not include the securities in the following table in the computation of the net loss from continuing operations per share attributable to Vertex common shareholders calculations because the effect would have been anti-dilutive during each period.
2016 | 2015 | 2014 | ||||||
(in thousands) | ||||||||
Stock options | 12,642 | 11,145 | 12,003 | |||||
Unvested restricted stock and restricted stock units | 3,546 | 3,024 | 3,091 | |||||
D. | Fair Value Measurements |
The fair value of the Company’s financial assets and liabilities reflects the Company’s estimate of amounts that it would have received in connection with the sale of the assets or paid in connection with the transfer of the liabilities in an orderly transaction between market participants at the measurement date. In connection with measuring the fair value of its assets and liabilities, the Company seeks to maximize the use of observable inputs (market data obtained from sources independent from the Company) and to minimize the use of unobservable inputs (the Company’s assumptions about how market participants would price assets and liabilities). The following fair value hierarchy is used to classify assets and liabilities based on the observable inputs and unobservable inputs used in order to value the assets and liabilities:
Level 1: | Quoted prices in active markets for identical assets or liabilities. An active market for an asset or liability is a market in which transactions for the asset or liability occur with sufficient frequency and volume to provide pricing information on an ongoing basis. |
Level 2: | Observable inputs other than Level 1 inputs. Examples of Level 2 inputs include quoted prices in active markets for similar assets or liabilities and quoted prices for identical assets or liabilities in markets that are not active. |
Level 3: | Unobservable inputs based on the Company’s assessment of the assumptions that market participants would use in pricing the asset or liability. |
The Company’s investment strategy is focused on capital preservation. The Company invests in instruments that meet the credit quality standards outlined in the Company’s investment policy. This policy also limits the amount of credit exposure to any one issue or type of instrument. As of December 31, 2016, the Company’s investments were in money market funds, government-sponsored enterprise securities, corporate equity securities, corporate debt securities and commercial paper.
F-24
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
As of December 31, 2016, all of the Company’s financial assets that were subject to fair value measurements were valued using observable inputs. The Company’s financial assets valued based on Level 1 inputs consisted of a money market funds, corporate equity securities and government-sponsored enterprise securities. The Company’s financial assets valued based on Level 2 inputs consisted of corporate debt securities and commercial paper, which consisted of investments in highly-rated investment-grade corporations. The fair value of the Company’s foreign currency forward contracts was based on Level 2 inputs using third party pricing services. During 2016, 2015 and 2014, the Company did not record an other-than-temporary impairment charge related to its financial assets.
The following table sets forth the Company’s financial assets (excluding VIE cash and cash equivalents) subject to fair value measurements:
Fair Value Measurements as of December 31, 2016 | |||||||||||||||
Fair Value Hierarchy | |||||||||||||||
Total | Level 1 | Level 2 | Level 3 | ||||||||||||
(in thousands) | |||||||||||||||
Financial instruments carried at fair value (asset position): | |||||||||||||||
Cash equivalents: | |||||||||||||||
Money market funds | $ | 280,560 | $ | 280,560 | $ | — | $ | — | |||||||
Marketable securities: | |||||||||||||||
Government-sponsored enterprise securities | 15,508 | 15,508 | — | — | |||||||||||
Corporate equity securities | 64,560 | 64,560 | — | — | |||||||||||
Commercial paper | 59,404 | — | 59,404 | — | |||||||||||
Corporate debt securities | 111,140 | — | 111,140 | — | |||||||||||
Prepaid and other current assets: | |||||||||||||||
Foreign currency forward contracts | 14,407 | — | 14,407 | — | |||||||||||
Other assets: | |||||||||||||||
Foreign currency forward contracts | 1,186 | $ | — | 1,186 | $ | — | |||||||||
Total financial assets | $ | 546,765 | $ | 360,628 | $ | 186,137 | $ | — | |||||||
Financial instruments carried at fair value (liability position): | |||||||||||||||
Other liabilities, current portion: | |||||||||||||||
Foreign currency forward contracts | $ | (144 | ) | $ | — | $ | (144 | ) | $ | — | |||||
Total financial liabilities | $ | (144 | ) | $ | — | $ | (144 | ) | $ | — | |||||
F-25
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
Fair Value Measurements as of December 31, 2015 | |||||||||||||||
Fair Value Hierarchy | |||||||||||||||
Total | Level 1 | Level 2 | Level 3 | ||||||||||||
(in thousands) | |||||||||||||||
Financial instruments carried at fair value (asset position): | |||||||||||||||
Cash equivalents: | |||||||||||||||
Money market funds | $ | 199,507 | $ | 199,507 | $ | — | $ | — | |||||||
Government-sponsored enterprise securities | 85,994 | 85,994 | — | — | |||||||||||
Commercial paper | 34,889 | — | 34,889 | — | |||||||||||
Corporate debt securities | 11,533 | — | 11,533 | — | |||||||||||
Marketable securities: | |||||||||||||||
Government-sponsored enterprise securities | 87,162 | 87,162 | — | — | |||||||||||
Commercial paper | 99,123 | — | 99,123 | — | |||||||||||
Corporate debt securities | 141,409 | — | 141,409 | — | |||||||||||
Prepaid and other current assets: | |||||||||||||||
Foreign currency forward contracts | 5,161 | — | 5,161 | — | |||||||||||
Other assets: | |||||||||||||||
Foreign currency forward contracts | 605 | $ | — | 605 | $ | — | |||||||||
Total financial assets | $ | 665,383 | $ | 372,663 | $ | 292,720 | $ | — | |||||||
Financial instruments carried at fair value (liability position): | |||||||||||||||
Other liabilities, current portion: | |||||||||||||||
Foreign currency forward contracts | $ | (769 | ) | $ | — | $ | (769 | ) | $ | — | |||||
Other liabilities, excluding current portion: | |||||||||||||||
Foreign currency forward contracts | (132 | ) | — | (132 | ) | — | |||||||||
Total financial liabilities | $ | (901 | ) | $ | — | $ | (901 | ) | $ | — | |||||
VIEs had cash equivalents of $46.1 million as of December 31, 2016 that consisted of money market funds, which are valued based on Level 1 inputs. These cash equivalents are not included in the table above. The Company’s noncontrolling interest related to VIEs includes the fair value of the contingent payments, which are valued based on Level 3 inputs. Please refer to Note B, “Collaborative Arrangements,” for further information.
The Company’s Credit Facility carries a variable interest rate set at current market rates, and as such, the carrying value approximates fair values. As of December 31, 2016, the fair value and carrying value of the Company’s Credit Facility was $300.0 million.
F-26
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
E. | Marketable Securities |
A summary of the Company’s cash, cash equivalents and marketable securities is shown below:
Amortized Cost | Gross Unrealized Gains | Gross Unrealized Losses | Fair Value | ||||||||||||
(in thousands) | |||||||||||||||
December 31, 2016 | |||||||||||||||
Cash and cash equivalents: | |||||||||||||||
Cash and money market funds | $ | 1,183,945 | $ | — | $ | — | $ | 1,183,945 | |||||||
Total cash and cash equivalents | $ | 1,183,945 | $ | — | $ | — | $ | 1,183,945 | |||||||
Marketable securities: | |||||||||||||||
Government-sponsored enterprise securities (matures within 1 year) | $ | 15,506 | $ | 2 | $ | — | $ | 15,508 | |||||||
Corporate equity securities (matures within 1 year) | 43,213 | 21,347 | — | 64,560 | |||||||||||
Commercial paper (matures within 1 year) | 59,331 | 73 | — | 59,404 | |||||||||||
Corporate debt securities (matures within 1 year) | 111,225 | — | (85 | ) | 111,140 | ||||||||||
Total marketable securities | 229,275 | 21,422 | (85 | ) | 250,612 | ||||||||||
Total cash, cash equivalents and marketable securities | $ | 1,413,220 | $ | 21,422 | $ | (85 | ) | $ | 1,434,557 | ||||||
December 31, 2015 | |||||||||||||||
Cash and cash equivalents: | |||||||||||||||
Cash and money market funds | $ | 582,352 | $ | — | $ | — | $ | 582,352 | |||||||
Government-sponsored enterprise securities | 85,994 | — | — | 85,994 | |||||||||||
Commercial paper | 34,889 | — | — | 34,889 | |||||||||||
Corporate debt securities | 11,533 | — | — | 11,533 | |||||||||||
Total cash and cash equivalents | $ | 714,768 | $ | — | $ | — | $ | 714,768 | |||||||
Marketable securities: | |||||||||||||||
Government-sponsored enterprise securities (matures within 1 year) | $ | 87,176 | $ | — | $ | (14 | ) | $ | 87,162 | ||||||
Commercial paper (matures within 1 year) | 98,877 | 246 | — | 99,123 | |||||||||||
Corporate debt securities (matures within 1 year) | 141,515 | — | (106 | ) | 141,409 | ||||||||||
Total marketable securities | 327,568 | 246 | (120 | ) | 327,694 | ||||||||||
Total cash, cash equivalents and marketable securities | 1,042,336 | 246 | (120 | ) | 1,042,462 | ||||||||||
The Company has a limited number of marketable securities in insignificant loss positions as of December 31, 2016, which the Company does not intend to sell and has concluded it will not be required to sell before recovery of the amortized costs for the investment at maturity. There were no charges recorded for other-than-temporary declines in fair value of marketable securities nor gross realized gains or losses recognized in 2016, 2015 or 2014.
F-27
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
F. | Accumulated Other Comprehensive Income |
The following table summarizes the changes in accumulated other comprehensive income by component:
Foreign currency translation adjustment | Unrealized holding gains (losses) on marketable securities, net of tax | Unrealized (losses) gains on foreign currency forward contracts, net of tax | Total | ||||||||||||
(in thousands) | |||||||||||||||
Balance at December 31, 2013 | $ | (325 | ) | $ | 42 | $ | (23 | ) | $ | (306 | ) | ||||
Other comprehensive (loss) income before reclassifications | (646 | ) | (165 | ) | 3,591 | 2,780 | |||||||||
Amounts reclassified from accumulated other comprehensive loss | — | — | (1,557 | ) | (1,557 | ) | |||||||||
Net current period other comprehensive (loss) income | (646 | ) | (165 | ) | 2,034 | 1,223 | |||||||||
Balance at December 31, 2014 | $ | (971 | ) | $ | (123 | ) | $ | 2,011 | $ | 917 | |||||
Other comprehensive (loss) income before reclassifications | (1,109 | ) | 249 | 6,493 | 5,633 | ||||||||||
Amounts reclassified from accumulated other comprehensive loss | — | — | (4,726 | ) | (4,726 | ) | |||||||||
Net current period other comprehensive (loss) income | (1,109 | ) | 249 | 1,767 | 907 | ||||||||||
Balance at December 31, 2015 | $ | (2,080 | ) | $ | 126 | $ | 3,778 | $ | 1,824 | ||||||
Other comprehensive (loss) income before reclassifications | (5,782 | ) | 17,395 | 17,383 | 28,996 | ||||||||||
Amounts reclassified from accumulated other comprehensive loss | — | — | (9,647 | ) | (9,647 | ) | |||||||||
Net current period other comprehensive (loss) income | (5,782 | ) | 17,395 | 7,736 | 19,349 | ||||||||||
Balance at December 31, 2016 | $ | (7,862 | ) | $ | 17,521 | $ | 11,514 | $ | 21,173 | ||||||
G. | Hedging |
The Company maintains a hedging program intended to mitigate the effect of changes in foreign exchange rates for a portion of the Company’s forecasted product revenues denominated in certain foreign currencies. The program includes foreign currency forward contracts that are designated as cash flow hedges under GAAP having contractual durations from one to eighteen months.
The Company formally documents the relationship between foreign currency forward contracts (hedging instruments) and forecasted product revenues (hedged items), as well as the Company’s risk management objective and strategy for undertaking various hedging activities, which includes matching all foreign currency forward contracts that are designated as cash flow hedges to forecasted transactions. The Company also formally assesses, both at the hedge’s inception and on an ongoing basis, whether the foreign currency forward contracts are highly effective in offsetting changes in cash flows of hedged items on a prospective and retrospective basis. If the Company determines that a (i) foreign currency forward contract is not highly effective as a cash flow hedge, (ii) foreign currency forward contract has ceased to be a highly effective hedge or (iii) forecasted transaction is no longer probable of occurring, the Company would discontinue hedge accounting treatment prospectively. The Company measures effectiveness based on the change in fair value of the forward contracts and the fair value of the hypothetical foreign currency forward contracts with terms that match the critical terms of the risk being hedged. As of December 31, 2016, all hedges were determined to be highly effective and the Company had not recorded any ineffectiveness related to the hedging program.
F-28
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
The following table summarizes the notional amount of the Company’s outstanding foreign currency forward contracts designated as cash flow hedges:
As of December 31, 2016 | As of December 31, 2015 | ||||||
Foreign Currency | (in thousands) | ||||||
Euro | $ | 164,368 | $ | 103,362 | |||
British pound sterling | 65,237 | 78,756 | |||||
Australian dollar | 23,776 | 27,167 | |||||
Total foreign currency forward contracts | $ | 253,381 | $ | 209,285 | |||
The following table summarizes the fair value of the Company’s outstanding foreign currency forward contracts designated as cash flow hedges under GAAP included on the Company’s consolidated balance sheets:
As of December 31, 2016 | ||||||||||
Assets | Liabilities | |||||||||
Classification | Fair Value | Classification | Fair Value | |||||||
(in thousands) | ||||||||||
Prepaid and other current assets | $ | 14,407 | Other liabilities, current portion | $ | (144 | ) | ||||
Other assets | 1,186 | Other liabilities, excluding current portion | — | |||||||
Total assets | $ | 15,593 | Total liabilities | $ | (144 | ) | ||||
As of December 31, 2015 | ||||||||||
Assets | Liabilities | |||||||||
Classification | Fair Value | Classification | Fair Value | |||||||
(in thousands) | ||||||||||
Prepaid and other current assets | $ | 5,161 | Other liabilities, current portion | $ | (769 | ) | ||||
Other assets | 605 | Other liabilities, excluding current portion | (132 | ) | ||||||
Total assets | $ | 5,766 | Total liabilities | $ | (901 | ) | ||||
The following table summarizes the potential effect of offsetting derivatives by type of financial instrument on the Company’s consolidated balance sheets:
As of December 31, 2016 | |||||||||||||||||||
Gross Amounts Recognized | Gross Amounts Offset | Gross Amount Presented | Gross Amount Not Offset | Legal Offset | |||||||||||||||
Foreign currency forward contracts | (in thousands) | ||||||||||||||||||
Total assets | $ | 15,593 | $ | — | $ | 15,593 | $ | (144 | ) | $ | 15,449 | ||||||||
Total liabilities | $ | (144 | ) | $ | — | $ | (144 | ) | $ | 144 | $ | — | |||||||
As of December 31, 2015 | |||||||||||||||||||
Gross Amounts Recognized | Gross Amounts Offset | Gross Amount Presented | Gross Amount Not Offset | Legal Offset | |||||||||||||||
Foreign currency forward contracts | (in thousands) | ||||||||||||||||||
Total assets | $ | 5,766 | $ | — | $ | 5,766 | $ | (901 | ) | $ | 4,865 | ||||||||
Total liabilities | (901 | ) | — | (901 | ) | 901 | — | ||||||||||||
F-29
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
H. | Inventories |
Inventories consisted of the following:
As of December 31, | |||||||
2016 | 2015 | ||||||
(in thousands) | |||||||
Raw materials | $ | 6,348 | $ | 8,696 | |||
Work-in-process | 56,672 | 40,695 | |||||
Finished goods | 14,584 | 7,816 | |||||
Total | $ | 77,604 | $ | 57,207 | |||
I. | Property and Equipment |
Property and equipment, net consisted of the following:
As of December 31, | |||||||
2016 | 2015 | ||||||
(in thousands) | |||||||
Buildings | $ | 548,232 | $ | 531,627 | |||
Furniture and equipment | 236,634 | 218,623 | |||||
Software | 134,321 | 124,469 | |||||
Leasehold improvements | 108,702 | 106,768 | |||||
Computers | 58,271 | 52,295 | |||||
Total property and equipment, gross | 1,086,160 | 1,033,782 | |||||
Less: accumulated depreciation | (387,798 | ) | (336,067 | ) | |||
Total property and equipment, net | $ | 698,362 | $ | 697,715 | |||
Total property and equipment, gross, as of December 31, 2016 and 2015, included $101.3 million and $106.8 million, respectively, for property and equipment recorded under capital leases. Accumulated depreciation, as of December 31, 2016 and 2015, included $37.9 million and $30.4 million, respectively, for property and equipment recorded under capital leases.
As of December 31, 2016, included in property and equipment, net were $17.8 million and $9.2 million in capitalized internally developed software costs and related amortization, respectively. As of December 31, 2015, included in property and equipment, net were $15.4 million and $4.1 million in capitalized internally developed software costs and related amortization, respectively.
The Company recorded depreciation expense of $60.8 million, $60.0 million and $62.3 million in 2016, 2015 and 2014, respectively.
J. | Intangible Assets and Goodwill |
Intangible Assets
As of each of December 31, 2016 and December 31, 2015, in-process research and development intangible assets of $284.3 million were recorded on the Company’s consolidated balance sheets.
In June 2015, in connection with entering into the Parion Agreement, the Company recorded an in-process research and development intangible asset of $255.3 million based on the Company’s estimate of the fair value of Parion’s lead investigational ENaC inhibitors, including VX-371 and VX-551, that were licensed by the Company from Parion. The Company aggregated the fair value of the ENaC inhibitors into a single intangible asset because the phase, nature and risks of development as well as the amount and timing of benefits associated with the assets were similar. The Company used a discount rate of 7.1% in the present-value models to estimate the fair values of the ENaC inhibitors intangible assets. The Company also conducted an evaluation of Parion’s other programs at the effective date of the Parion Agreement and
F-30
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
determined that market participants would not have ascribed value to those programs because of the stage of development of the assets in each program and uncertainties related to the potential development and commercialization of the programs.
Goodwill
As of each of December 31, 2016 and December 31, 2015, goodwill of $50.4 million was recorded on the Company’s consolidated balance sheet.
K. | Additional Balance Sheet Detail |
Prepaid and other current assets consisted of the following:
As of December 31, | |||||||
2016 | 2015 | ||||||
(in thousands) | |||||||
Prepaid expenses | $ | 36,134 | $ | 22,058 | |||
Fair value foreign currency forward contracts | 14,407 | 5,161 | |||||
Taxes receivable | 3,213 | 14,682 | |||||
Other | 16,780 | 12,835 | |||||
Total | $ | 70,534 | $ | 54,736 | |||
Accrued expenses consisted of the following:
As of December 31, | |||||||
2016 | 2015 | ||||||
(in thousands) | |||||||
Payroll and benefits | $ | 86,387 | $ | 87,873 | |||
Research, development and commercial contract costs | 62,756 | 55,677 | |||||
Product revenue allowances | 86,533 | 47,209 | |||||
Royalty payable | 52,845 | 60,191 | |||||
Taxes payable and reserves (including VIE taxes payable) | 6,883 | 30,953 | |||||
Professional fees | 6,512 | 7,455 | |||||
Interest | 1,390 | 4,642 | |||||
Other | 11,943 | 11,820 | |||||
Total | $ | 315,249 | $ | 305,820 | |||
Other liabilities, excluding current portion consisted of the following:
As of December 31, | |||||||
2016 | 2015 | ||||||
(in thousands) | |||||||
Advance from CFFT | $ | 73,423 | $ | — | |||
Deferred rent | 19,551 | 22,235 | |||||
Other | 9,148 | 9,543 | |||||
Total | $ | 102,122 | $ | 31,778 | |||
L. | Long Term Obligations |
Fan Pier Leases
In 2011, the Company entered into two lease agreements, pursuant to which the Company leases approximately 1.1 million square feet of office and laboratory space in two buildings (the “Buildings”) at Fan Pier in Boston, Massachusetts (the
F-31
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
“Fan Pier Leases”). The Company commenced lease payments in December 2013, and will make lease payments pursuant to the Fan Pier Leases through December 2028. The Company has an option to extend the term of the Fan Pier Leases for an additional 10 years.
Because the Company was involved in the construction project, the Company was deemed for accounting purposes to be the owner of the Buildings during the construction period and recorded project construction costs incurred by the landlord. Upon completion of the Buildings, the Company evaluated the Fan Pier Leases and determined that the Fan Pier Leases did not meet the criteria for “sale-leaseback” treatment. Accordingly, the Company began depreciating the asset and incurring interest expense related to the financing obligation in 2013. The Company bifurcates its lease payments pursuant to the Fan Pier Leases into (i) a portion that is allocated to the Buildings and (ii) a portion that is allocated to the land on which the Buildings were constructed. The portion of the lease obligations allocated to the land is treated as an operating lease that commenced in 2011. The Company recorded interest expense of $60.2 million in each of 2016, 2015 and 2014. The Company recorded depreciation expense of $13.3 million in each of 2016, 2015 and $13.4 million in 2014, respectively. In each of 2016, 2015 and 2014, the Company recorded rent expense of $6.5 million.
Property and equipment, net, included $489.0 million and $502.3 million as of December 31, 2016 and 2015, respectively, related to construction costs for the Buildings. The carrying value of the construction financing lease obligation related to the Buildings, which excludes interest that will be imputed over the course of the Company’s lease agreement for the Buildings, was $472.6 million and $473.0 million, as of December 31, 2016 and 2015, respectively.
San Diego Lease
On December 2, 2015, the Company entered into a lease agreement for 3215 Merryfield Row, San Diego, California with ARE-SD Region No. 23, LLC. Pursuant to this agreement, the Company agreed to lease approximately 170,000 square feet of office and laboratory space in a building under construction in San Diego, California (“San Diego Lease”). Lease payments pursuant to the San Diego Lease will commence upon completion of the building, scheduled for the first half of 2018, and will extend for 16 years from the commencement date. Pursuant to the San Diego Lease, during the initial 16-year term, the Company will pay an average of approximately $10.2 million per year in aggregate rent, exclusive of operating expenses. The Company has the option to extend the lease term for up to two additional five-year terms.
Because the Company is involved in the construction project, the Company is deemed for accounting purposes to be the owner of the San Diego building during the construction period and recorded project construction costs incurred by the landlord. The Company bifurcates its lease payments pursuant to the San Diego Lease into (i) a portion that is allocated to the San Diego building and (ii) a portion that is allocated to the land on which the San Diego building was constructed. Although the Company will not begin making lease payments pursuant to the San Diego Lease until the commencement date, the portion of the lease obligation allocated to the land is treated for accounting purposes as an operating lease that commenced in the fourth quarter of 2016. Upon completion of the San Diego building, the Company will evaluate the San Diego Lease and determine if the San Diego Lease meets the criteria for “sale-leaseback” treatment. If the San Diego Lease meets the “sale-leaseback” criteria, the Company will remove the asset and the related liability from its consolidated balance sheet and treat the San Diego Lease as either an operating or a capital lease based on the Company’s assessment of the accounting guidance. The Company expects that upon completion of construction of the San Diego building the San Diego Lease will not meet the “sale-leaseback” criteria. If the San Diego Lease does not meet “sale-leaseback” criteria, the Company will treat the San Diego Lease as a financing obligation and will depreciate the asset over its estimated useful life.
Revolving Credit Facility
In October 2016, the Company entered into a Credit Agreement (the “Credit Agreement”) with Bank of America, N.A., as administrative agent and the lenders referred to therein. The Credit Agreement provides for a $500.0 million revolving facility, $300.0 million of which was drawn at closing (the “Loans”). The Credit Agreement also provides that, subject to satisfaction of certain conditions, the Company may request that the borrowing capacity under the Credit Agreement be increased by an additional $300.0 million. The Credit Agreement matures on October 13, 2021.
The proceeds of the borrowing under the Credit Agreement were used primarily to repay the Company's existing indebtedness under the Macquarie Loan. The Loans will bear interest, at the Company's option, at either a base rate or a
F-32
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
Eurodollar rate, in each case plus an applicable margin. Under the Credit Agreement, the applicable margins on base rate loans range from 0.75% to 1.50% and the applicable margins on Eurodollar loans range from 1.75% to 2.50%, in each case based on the Company's consolidated leverage ratio (the ratio of the Company's total consolidated debt to the Company's trailing twelve-month EBITDA).
The Loans are guaranteed by certain of the Company's domestic subsidiaries and secured by substantially all of the Company's assets and the assets of the Company's domestic subsidiaries (excluding intellectual property, owned and leased real property and certain other excluded property) and by the equity interests of the Company's subsidiaries, subject to certain exceptions. Under the terms of the Credit Agreement, the Company must maintain, subject to certain limited exceptions, a consolidated leverage ratio of 3.00 to 1.00 and consolidated EBITDA of at least $200.0 million, in each case to be measured on a quarterly basis.
The Credit Agreement contains customary representations and warranties and usual and customary affirmative and negative covenants. The Credit Agreement also contains customary events of default. In the case of a continuing event of default, the administrative agent would be entitled to exercise various remedies, including the acceleration of amounts due under outstanding loans.
Term Loan
On July 9, 2014, the Company entered into a credit agreement with the lenders party thereto, and Macquarie US Trading LLC (“Macquarie”), as administrative agent. The credit agreement provided for a $300.0 million senior secured term loan (“Macquarie Loan”). On October 13, 2016, the Company terminated and repaid all outstanding obligations under the Macquarie Loan. The Company incurred a charge of $2.2 million in the fourth quarter of 2016 related to a loss on extinguishment attributable to the Macquarie Loan that was recorded as Interest Expense in the Company’s consolidated statements of operations.
The Macquarie Loan initially bore interest at a rate of 7.2% per annum, which was reduced to 6.2% per annum based on the FDA’s approval of ORKAMBI. The Macquarie Loan bore interest at a rate of LIBOR plus 5.0% per annum during the third year of the term. If the Company had not terminated and repaid all outstanding obligations, the maturity date of all loans under the facilities would have been July 9, 2017.
Based on the Company's evaluation of the Macquarie Loan, the Company determined that the Macquarie Loan contained several embedded derivatives. These embedded derivatives were clearly and closely related to the host instrument because they related to the Company's credit risk; therefore, they did not require bifurcation from the host instrument, the Macquarie Loan.
The Company incurred $5.3 million in fees paid to Macquarie that were recorded as a discount on the Term Loan and that were recorded as additional interest expense using the effective interest method over the term of the loan in the Company’s consolidated statements of operations. As of December 31, 2016 and 2015, the unamortized discount associated with the Term Loan that was included in the senior secured term loan caption on the Company’s consolidated balance sheets was zero and $4.6 million, respectively.
Subsequent Event
In February 2017, the Company repaid all $300.0 million outstanding under the Credit Agreement. The Company may reborrow and repay amounts under the revolving credit agreement without penalty.
M. | Common Stock, Preferred Stock and Equity Plans |
The Company is authorized to issue 500,000,000 shares of common stock. Holders of common stock are entitled to one vote per share. Holders of common stock are entitled to receive dividends, if and when declared by the Company’s Board of Directors, and to share ratably in the Company’s assets legally available for distribution to the Company’s shareholders in the event of liquidation. Holders of common stock have no preemptive, subscription, redemption or conversion rights. The holders of common stock do not have cumulative voting rights.
F-33
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
The Company is authorized to issue 1,000,000 shares of preferred stock in one or more series and to fix the powers, designations, preferences and relative participating, option or other rights thereof, including dividend rights, conversion rights, voting rights, redemption terms, liquidation preferences and the number of shares constituting any series, without any further vote or action by the Company’s shareholders. As of December 31, 2016 and 2015, the Company had no shares of preferred stock issued or outstanding.
Stock and Option Plans
The purpose of each of the Company’s stock and option plans is to attract, retain and motivate its employees, consultants and directors. Awards granted under these plans can be incentive stock options (“ISOs”), nonstatutory stock options (“NSOs”), restricted stock (“RSs”), restricted stock units (“RSUs”) or other equity-based awards, as specified in the individual plans.
Shares issued under all of the Company’s plans are funded through the issuance of new shares. The following table contains information about the Company’s equity plans:
As of December 31, 2016 | ||||||||||
Title of Plan | Group Eligible | Type of Award Granted | Awards Outstanding | Additional Awards Authorized for Grant | ||||||
2013 Stock and Option Plan | Employees, Non-employee Directors and Consultants | NSO, RS and RSU | 9,832,269 | 9,180,002 | ||||||
2006 Stock and Option Plan | Employees, Non-employee Directors and Consultants | NSO, RS and RSU | 6,355,357 | — | ||||||
Total | 16,187,626 | 9,180,002 | ||||||||
All options granted under the Company’s 2013 Stock and Option Plan (“2013 Plan”) and 2006 Stock and Option Plan (“2006 Plan”) were granted with an exercise price equal to the fair value of the underlying common stock on the date of grant. As of December 31, 2016, the stock and option plan under which the Company makes new equity awards is the Company’s 2013 Plan. Under the 2013 Plan, no stock options can be awarded with an exercise price less than the fair market value on the date of grant. The Company’s shareholders (i) approved an increase in the number of shares authorized for issuance pursuant to the 2013 Plan of 7,800,000 shares, plus the number of shares that remained available for issuance under the Company’s 2006 Stock and Option Plan, which rolled-over into the 2013 Stock and Option Plan in 2015 and (ii) approved an increase in the number of shares authorized for issuance pursuant to the 2013 Plan of 9,500,000 shares in 2014.
During the three years ended December 31, 2016, grants to current employees and directors primarily had a grant date that was the same as the date the award was approved by the Company’s Board of Directors. During the three years ended December 31, 2016, for grants to new employees and directors, the date of grant for awards was the employee’s first day of employment or the date the director was elected to the Company’s Board of Directors. All options awarded under the Company’s stock and option plans expire not more than 10 years from the grant date.
Historically, all shares of restricted stock and restricted stock units have been granted at a price equal to $0.01, the par value of the Company’s common stock. Beginning with awards approved by the Company’s Board of Directors in July 2016, the Company stopped granting restricted stock units at par value and instead grants the awards at a purchase price equal to $0.00. Vesting of options, restricted stock and restricted stock units generally is ratable over specified periods and is determined by the Company’s Board of Directors.
F-34
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
The following table summarizes information related to the outstanding and exercisable options during the year ended December 31, 2016:
Stock Options | Weighted-average Exercise Price | Weighted-average Remaining Contractual Life | Aggregate Intrinsic Value | |||||||||
(in thousands) | (per share) | (in years) | (in thousands) | |||||||||
Outstanding at December 31, 2015 | 11,145 | $ | 75.99 | |||||||||
Granted | 3,183 | $ | 91.36 | |||||||||
Exercised | (1,064 | ) | $ | 45.61 | ||||||||
Forfeited | (544 | ) | $ | 94.62 | ||||||||
Expired | (78 | ) | $ | 107.51 | ||||||||
Outstanding at December 31, 2016 | 12,642 | $ | 81.41 | 7.06 | $ | 124,939 | ||||||
Exercisable at December 31, 2016 | 7,323 | $ | 68.92 | 6.00 | $ | 121,671 | ||||||
Exercisable and Expected to Vest at December 31, 2016 | 12,200 | $ | 80.76 | 7.00 | $ | 124,892 | ||||||
The aggregate intrinsic value in the table above represents the total pre-tax amount, net of exercise price, that would have been received by option holders if all option holders had exercised all options with an exercise price lower than the market price on December 30, 2016, which was $74.11 based on the average of the high and low price of the Company’s common stock on that date.
The total intrinsic value (the amount by which the fair market value exceeded the exercise price) of stock options exercised during 2016, 2015 and 2014 was $48.6 million, $252.9 million and $316.5 million, respectively. The total cash received by the Company as a result of employee stock option exercises during 2016, 2015 and 2014 was $48.5 million, $165.6 million and $255.5 million, respectively.
The following table summarizes information about stock options outstanding and exercisable at December 31, 2016:
Options Outstanding | Options Exercisable | |||||||||||||||
Range of Exercise Prices | Number Outstanding | Weighted-average Remaining Contractual Life | Weighted-average Exercise Price | Number Exercisable | Weighted-average Exercise Price | |||||||||||
(in thousands) | (in years) | (per share) | (in thousands) | (per share) | ||||||||||||
$18.93–$20.00 | 137 | 1.10 | $ | 18.93 | 137 | $ | 18.93 | |||||||||
$20.01–$40.00 | 1,696 | 3.19 | $ | 33.94 | 1,696 | $ | 33.94 | |||||||||
$40.01–$60.00 | 1,867 | 5.59 | $ | 48.26 | 1,762 | $ | 48.44 | |||||||||
$60.01–$80.00 | 1,345 | 7.11 | $ | 75.90 | 891 | $ | 75.60 | |||||||||
$80.01–$100.00 | 4,529 | 8.45 | $ | 90.60 | 1,548 | $ | 89.61 | |||||||||
$100.01–$120.00 | 1,604 | 8.05 | $ | 109.33 | 702 | $ | 109.29 | |||||||||
$120.01–$134.69 | 1,464 | 8.53 | $ | 130.58 | 587 | $ | 130.17 | |||||||||
Total | 12,642 | 7.06 | $ | 81.41 | 7,323 | $ | 68.92 | |||||||||
F-35
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
The following table summarizes the restricted stock and restricted stock unit activity of the Company during the year ended December 31, 2016:
Restricted Stock | Restricted Stock Units | ||||||||||||
Number of Units | Weighted-average Grant-date Fair Value | Number of Shares | Weighted-average Grant-date Fair Value | ||||||||||
(in thousands) | (per share) | (in thousands) | (per share) | ||||||||||
Unvested at December 31, 2015 | 2,831 | $ | 98.80 | 193 | $ | 98.36 | |||||||
Granted | 857 | $ | 91.49 | 847 | $ | 90.46 | |||||||
Vested | (817 | ) | $ | 79.28 | (59 | ) | $ | 83.13 | |||||
Cancelled | (258 | ) | $ | 98.56 | (48 | ) | $ | 94.54 | |||||
Unvested at December 31, 2016 | 2,613 | $ | 102.54 | 933 | $ | 92.35 | |||||||
The total fair value of restricted stock that vested during 2016, 2015 and 2014 (measured on the date of vesting) was $74.1 million, $124.0 million and $54.5 million, respectively. The total fair value of restricted stock units that vested during 2016, 2015 and 2014 (measured on the date of vesting) was $5.3 million, $8.0 million and $2.9 million, respectively.
Employee Stock Purchase Plan
The Company has an employee stock purchase plan (the “ESPP”). The ESPP permits eligible employees to enroll in a twelve-month offering period comprising two six-month purchase periods. Participants may purchase shares of the Company’s common stock, through payroll deductions, at a price equal to 85% of the fair market value of the common stock on the first day of the applicable twelve-month offering period, or the last day of the applicable six-month purchase period, whichever is lower. Purchase dates under the ESPP occur on or about May 14 and November 14 of each year. As of December 31, 2016, there were 891,353 shares of common stock authorized for issuance pursuant to the ESPP.
In 2016, the following shares were issued to employees under the ESPP:
Year Ended December 31, 2016 | |||
(in thousands, except per share amount) | |||
Number of shares | 272 | ||
Average price paid per share | $ | 70.70 | |
N. | Stock-based Compensation Expense |
The Company recognizes share-based payments to employees as compensation expense using the fair value method. The fair value of stock options and shares purchased pursuant to the ESPP is calculated using the Black-Scholes option pricing model. The fair value of restricted stock and restricted stock units is based on the intrinsic value on the date of grant. Stock-based compensation, measured at the grant date based on the fair value of the award, is typically recognized as expense ratably over the requisite service period. The expense recognized over the requisite service period includes an estimate of awards that will be forfeited.
The effect of stock-based compensation expense during the three years ended December 31, 2016 was as follows:
2016 | 2015 | 2014 | |||||||||
(in thousands) | |||||||||||
Stock-based compensation expense by line item: | |||||||||||
Research and development expenses | $ | 153,451 | $ | 152,955 | $ | 116,998 | |||||
Sales, general and administrative expenses | 84,254 | 78,070 | 60,544 | ||||||||
Total stock-based compensation expense included in costs and expenses | $ | 237,705 | $ | 231,025 | $ | 177,542 | |||||
F-36
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
The stock-based compensation expense by type of award during the three years ended December 31, 2016 was as follows:
2016 | 2015 | 2014 | |||||||||
(in thousands) | |||||||||||
Stock-based compensation expense by type of award: | |||||||||||
Stock options | $ | 114,768 | $ | 129,276 | $ | 99,961 | |||||
Restricted stock and restricted stock units | 118,709 | 98,811 | 70,678 | ||||||||
ESPP share issuances | 7,835 | 7,025 | 8,326 | ||||||||
Less: stock-based compensation expense capitalized to inventories | (3,607 | ) | (4,087 | ) | (1,423 | ) | |||||
Total stock-based compensation expense included in costs and expenses | $ | 237,705 | $ | 231,025 | $ | 177,542 | |||||
The Company capitalizes stock-based compensation expense to inventories, all of which is attributable to employees who support the Company’s manufacturing operations for the Company’s products.
The following table sets forth the Company’s unrecognized stock-based compensation expense, net of estimated forfeitures, as of December 31, 2016, by type of award and the weighted-average period over which that expense is expected to be recognized:
As of December 31, 2016 | |||||
Unrecognized Expense Net of Estimated Forfeitures | Weighted-average Recognition Period | ||||
(in thousands) | (in years) | ||||
Type of award: | |||||
Stock options | $ | 157,819 | 2.50 | ||
Restricted stock and restricted stock units | $ | 176,972 | 2.39 | ||
ESPP share issuances | $ | 4,080 | 0.58 | ||
Stock Options
The Company issues stock options with service conditions, which are generally the vesting periods of the awards. The Company uses the Black-Scholes option pricing model to estimate the fair value of stock options at the grant date. The Black-Scholes option pricing model uses the option exercise price as well as estimates and assumptions related to the expected price volatility of the Company’s stock, the rate of return on risk-free investments, the expected period during which the options will be outstanding, and the expected dividend yield for the Company’s stock to estimate the fair value of a stock option on the grant date. The options granted during 2016, 2015 and 2014 had a weighted-average grant-date fair value per share of $37.93, $52.16 and $39.95, respectively.
The fair value of each option granted during 2016, 2015 and 2014 was estimated on the date of grant using the Black-Scholes option pricing model with the following weighted-average assumptions:
2016 | 2015 | 2014 | ||||||
Expected stock price volatility | 46.77 | % | 47.29 | % | 50.86 | % | ||
Risk-free interest rate | 1.32 | % | 1.61 | % | 1.77 | % | ||
Expected term of options (in years) | 4.91 | 5.28 | 5.47 | |||||
Expected annual dividends | — | — | — | |||||
The weighted-average valuation assumptions were determined as follows:
• | Expected stock price volatility: Expected stock price volatility is calculated using the trailing one month average of daily implied volatilities prior to grant date. Implied volatility is based on options to purchase the Company’s stock with remaining terms of greater than one year that are regularly traded in the market. |
F-37
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
• | Risk-free interest rate: The Company bases the risk-free interest rate on the interest rate payable on U.S. Treasury securities in effect at the time of grant for a period that is commensurate with the assumed expected option term. |
• | Expected term of options: The expected term of options represents the period of time options are expected to be outstanding. The Company uses historical data to estimate employee exercise and post-vest termination behavior. The Company believes that all groups of employees exhibit similar exercise and post-vest termination behavior and therefore does not stratify employees into multiple groups in determining the expected term of options. |
• | Expected annual dividends: The estimate for annual dividends is $0.00 because the Company has not historically paid, and does not intend for the foreseeable future to pay, a dividend. |
Restricted Stock and Restricted Stock Units
The Company awards restricted stock and restricted stock units with service conditions, which are generally the vesting periods of the awards. Until 2017, the Company also awarded, to certain members of senior management, on an annual basis restricted stock and restricted stock units that vest upon the earlier of the satisfaction of (i) a performance condition or (ii) a service condition.
In February 2016, the Company began granting performance-based restricted stock units (“PSUs”) to certain members of senior management. Threshold, target and maximum parameters were established for the financial and half on non-financial goals, and will be used to calculate the number of shares that will be issuable when the award vests, which may range from zero to 200% of the target amount. The financial-based PSUs vest in three equal installments over a three-year period and are expensed ratably over that same period based upon an assessment of the likelihood of achievement. The non-financial based PSUs cliff vest at the end of the three-year performance period and are expensed on a straight-line basis over that same period based upon an assessment of the likelihood of achievement.
In addition, in 2015 and 2014, the Company issued, pursuant to a retention program, restricted stock awards to certain members of senior management that will vest upon the satisfaction of both (i) a performance condition and (ii) a service condition.
Employee Stock Purchase Plan
The weighted-average fair value of each purchase right granted during 2016, 2015 and 2014 was $26.86, $37.84 and $29.59, respectively. The following table reflects the weighted-average assumptions used in the Black-Scholes option pricing model for 2016, 2015 and 2014:
2016 | 2015 | 2014 | ||||||
Expected stock price volatility | 48.22 | % | 47.20 | % | 60.32 | % | ||
Risk-free interest rate | 0.56 | % | 0.40 | % | 0.09 | % | ||
Expected term (in years) | 0.75 | 0.72 | 0.75 | |||||
Expected annual dividends | — | — | — | |||||
The expected stock price volatility for ESPP offerings is based on implied volatility. The Company bases the risk-free interest rate on the interest rate payable on U.S. Treasury securities in effect at the time of grant for a period that is commensurate with the assumed expected term. The expected term represents purchases and purchase periods that take place within the offering period. The expected annual dividends estimate is $0.00 because the Company has not historically paid, and does not for the foreseeable future intend to pay, a dividend.
O. | Other Arrangements |
Sale of HIV Protease Inhibitor Royalty Stream
In 2008, the Company sold to a third party its rights to receive royalty payments from GlaxoSmithKline plc, net of royalty amounts to be earned by and due to a third party, for a one-time cash payment of $160.0 million. These royalty
F-38
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
payments relate to net sales of HIV protease inhibitors, which had been developed pursuant to a collaboration agreement between the Company and GlaxoSmithKline plc. As of December 31, 2016, the Company had $12.6 million in deferred revenues related to the one-time cash payment, which it is recognizing over the life of the collaboration agreement with GlaxoSmithKline plc based on the units-of-revenue method. In addition, the Company continues to recognize royalty revenues equal to the amount of the third-party subroyalty and an offsetting royalty expense for the third-party subroyalty payment.
Other income (expense), net
In April 2014, the Company received a one-time cash payment of $36.7 million from its landlord pursuant to the Fan Pier Leases. This payment related to bonds issued pursuant to an Infrastructure Development Assistance Agreement between The Commonwealth of Massachusetts and the Company’s landlord. The bonds were issued in connection with the landlord’s contribution to infrastructure improvements and also were dependent upon employment levels at the Company through the bond issuance date. The Company accounted for the cash payment as a government grant as it was provided in part related to the Company’s employment level in Massachusetts. Such grants are recognized in income in the period in which the conditions of the grant are met and there is reasonable assurance that the grant will be received, provided it is not subject to refund. In the second quarter of 2014, the Company recorded $36.7 million as a credit to other income (expense), net in its consolidated statements of operations because the Company’s employment obligations related to these funds were satisfied as of the date of issuance of the bonds and the payment received is not subject to refund.
P. | Income Taxes |
The components of loss from continuing operations before provision for income taxes during the three years ended December 31, 2016 consisted of the following:
2016 | 2015 | 2014 | |||||||||
(in thousands) | |||||||||||
United States | $ | (147,860 | ) | $ | (272,326 | ) | $ | (645,465 | ) | ||
Foreign | 80,494 | (285,474 | ) | (89,410 | ) | ||||||
Loss from continuing operations before provision for income taxes | $ | (67,366 | ) | $ | (557,800 | ) | $ | (734,875 | ) | ||
The components of the provision for income taxes from continuing operations during the three years ended December 31, 2016 consisted of the following:
2016 | 2015 | 2014 | |||||||||
(in thousands) | |||||||||||
Current taxes: | |||||||||||
United States | $ | (3,821 | ) | $ | 25,623 | $ | 2,853 | ||||
Foreign | 1,794 | 831 | 2,457 | ||||||||
State | 1,836 | 3,629 | 1,366 | ||||||||
Total current taxes | $ | (191 | ) | $ | 30,083 | $ | 6,676 | ||||
Deferred taxes: | |||||||||||
United States | $ | 18,659 | $ | 497 | $ | 244 | |||||
Foreign | (3,359 | ) | (355 | ) | — | ||||||
State | 1,556 | 156 | 38 | ||||||||
Total deferred taxes | $ | 16,856 | $ | 298 | $ | 282 | |||||
Provision for income taxes | $ | 16,665 | $ | 30,381 | $ | 6,958 | |||||
F-39
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
The difference between the Company’s “expected” tax provision (benefit), as computed by applying the U.S. federal corporate tax rate of 35% to loss from continuing operations before provision for income taxes, and actual tax is reconciled as follows:
2016 | 2015 | 2014 | |||||||||
(in thousands) | |||||||||||
Loss from continuing operations before provision for income taxes | $ | (67,366 | ) | $ | (557,800 | ) | $ | (734,875 | ) | ||
Expected tax provision (benefit) | (23,578 | ) | (195,230 | ) | (257,206 | ) | |||||
State taxes, net of federal benefit | 3,621 | 3,800 | 1,124 | ||||||||
Foreign rate differential | 21,346 | 47,402 | 39,335 | ||||||||
Tax credits | (47,773 | ) | (55,696 | ) | (33,788 | ) | |||||
Unbenefitted operating losses (gains) | 14,837 | 226,169 | 241,037 | ||||||||
Non-deductible expenses | 24,749 | 5,817 | 18,756 | ||||||||
Rate change | 12,836 | (1,224 | ) | (1,826 | ) | ||||||
Tax attribute expiration | 9,947 | — | — | ||||||||
Other | 680 | (657 | ) | (474 | ) | ||||||
Provision for income taxes | $ | 16,665 | $ | 30,381 | $ | 6,958 | |||||
The foreign rate differential in the tax rate reconciliation table reflects the effect of operations in jurisdictions with tax rates that are different from the United States. As set forth in the components of loss before provision for income taxes, the Company had income in 2016 and losses in 2015 and 2014 in foreign jurisdictions in each year presented. Due to lower foreign tax rates, particularly in the United Kingdom, the Company’s tax expense (benefit) in foreign jurisdictions is less than the “expected” tax expense (benefit) that would have resulted from income (losses) in these jurisdictions at corporate tax rates in the United States. The difference between the tax expense (benefit) at foreign corporate tax rates and the “expected” expense (benefit) based on corporate tax rates in the United States is reflected in the tax reconciliation table under the caption “foreign rate differential.”
The unbenefitted operating losses in the tax rate reconciliation table primarily reflect a change in the valuation allowance on deferred tax assets related to the United States, United Kingdom and Switzerland. In 2016, the valuation allowance increased primarily due to an increase in tax credits in the United States and an increase in the net operating loss in the United Kingdom, both due to the uncertainty in the Company’s ability to use them in future periods. In 2015 and 2014, the valuation allowance increased primarily related to an increase in net operating losses that have been incurred with no corresponding benefit due to the uncertainty in the Company’s ability to use them in future periods.
In 2016, the effect of non-deductible expenses is related to equity compensation, Orphan Drug Credits, foreign amortization and partial disallowance of expenses related to dissolution of a foreign subsidiary.
F-40
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
Deferred tax assets and liabilities are determined based on the difference between financial statement and tax bases using enacted tax rates in effect for the year in which the differences are expected to reverse. The components of the deferred taxes were as follows:
As of December 31, | |||||||
2016 | 2015 | ||||||
(in thousands) | |||||||
Deferred tax assets: | |||||||
Net operating loss | $ | 1,232,399 | $ | 1,250,642 | |||
Tax credit carryforwards | 367,402 | 315,535 | |||||
Property and equipment | 22 | — | |||||
Intangible assets | 34,938 | 14,673 | |||||
Deferred revenues | 31,205 | 9,341 | |||||
Stock-based compensation | 110,446 | 93,404 | |||||
Inventories | 4,705 | 5,913 | |||||
Accrued expenses | 23,078 | 27,236 | |||||
Currency translation adjustment | — | 222 | |||||
Unrealized loss | 5 | — | |||||
Construction financing lease obligation | 177,735 | 176,250 | |||||
Gross deferred tax assets | 1,981,935 | 1,893,216 | |||||
Valuation allowance | (1,731,186 | ) | (1,716,349 | ) | |||
Total deferred tax assets | 250,749 | 176,867 | |||||
Deferred tax liabilities: | |||||||
Property and equipment | (169,089 | ) | (175,424 | ) | |||
Acquired intangibles | (134,063 | ) | (110,439 | ) | |||
Deferred revenue | $ | (73,357 | ) | $ | — | ||
Unrealized gain | $ | (7,967 | ) | $ | (1,088 | ) | |
Net deferred tax liabilities | $ | (133,727 | ) | $ | (110,084 | ) | |
The Company presents its deferred tax assets and deferred tax liabilities gross on its consolidated balance sheets. As of December 31, 2016, $134.0 million of the deferred tax liabilities are attributable to the Company’s collaborations with BioAxone and Parion. As of December 31, 2015, $110.4 million of the deferred tax liabilities are attributable to the Company’s collaborations with BioAxone and Parion.
For federal income tax purposes, as of December 31, 2016, the Company has net operating loss carryforwards of approximately $4.1 billion and tax credits of $262.9 million, which may be used to offset future federal income and tax liability, respectively. In addition, the Company will record an increase of approximately $1.2 billion of the federal net operating loss carryforward with a corresponding valuation allowance upon adoption of the new stock compensation guidance in the first quarter 2017.
For state income tax purposes, the Company has net operating loss carryforwards of approximately $975.8 million and tax credits of $103.8 million, which may be used to offset future state income and tax liability, respectively. In addition, the Company will record an increase of approximately $190.0 million of the state net operating loss carryforward with a corresponding valuation allowance upon adoption of the new stock compensation guidance in the first quarter 2017.
These federal and state operating loss carryforwards and tax credits expire at various dates through 2036. After consideration of all the evidence, both positive and negative, the Company continues to maintain a valuation allowance for the majority of the 2016 deferred tax asset because it is more likely than not that the deferred tax asset will not be realized. In future periods, if management determines that it is more likely than not that the deferred tax asset will be realized, (i) the valuation allowance would be decreased, (ii) the deferred tax asset would be reflected on the Company’s consolidated balance sheet and (iii) the Company would record non-cash benefits in its consolidated statements of operations related to the reflection of the deferred tax asset on its consolidated balance sheets.
F-41
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
The valuation allowance increased by $14.8 million from December 31, 2015 to December 31, 2016 primarily due to an increase in research tax credit carry forwards in the United States and other timing items.
Unrecognized tax benefits during the two years ended December 31, 2016 consisted of the following:
2016 | 2015 | ||||||
(in thousands) | |||||||
Unrecognized tax benefits beginning of year | $ | 425 | $ | 880 | |||
Decrease due to statute of limitations expiring | (425 | ) | — | ||||
Decrease due to settlements and payments | — | (455 | ) | ||||
Unrecognized tax benefits end of year | $ | — | $ | 425 | |||
The Company had gross unrecognized tax benefits of zero and $0.4 million, respectively, as of December 31, 2016 and 2015. The Company recognizes interest and penalties related to income taxes as a component of income tax expense. As of December 31, 2016, no interest and penalties have been accrued.
The Company files United States federal income tax returns and income tax returns in various state, local and foreign jurisdictions. The Company is no longer subject to any tax assessment from an income tax examination in the United States or any other major taxing jurisdiction for years before 2011, except where the Company has net operating losses or tax credit carryforwards that originate before 2011. The Company currently is under examination by Canada Revenue Agency for the years ending December 31, 2011 through December 31, 2013. No adjustments have been reported. The Company is not under examination by any other jurisdictions for any tax year. The Company concluded audits with Internal Revenue Service, Delaware, Pennsylvania, Texas and Revenue Quebec during 2016, and Massachusetts and New York during 2015, with no material adjustments.
At December 31, 2016, foreign earnings, which were not significant, have been retained indefinitely by foreign subsidiary companies for reinvestment; therefore, no provision has been made for income taxes that would be payable upon the distribution of such earnings and it would not be practicable to determine the amount of the related unrecognized deferred income tax liability. Upon repatriation of those earnings, in the form of dividends or otherwise, the Company would be subject to U.S. federal income taxes (subject to an adjustment for foreign tax credits) and withholding taxes payable to the various foreign countries.
Q. | Restructuring Expenses |
Facility Lease Obligations
The Company has adopted several plans to restructure its facility operations for which it has incurred restructuring expenses in the three years ended December 31, 2016. The Company’s initial estimate of its liabilities for net ongoing costs associated with these facility obligations are recorded at fair value on the cease use date. In estimating the expenses and liabilities related to these facilities, the Company utilizes the probability-weighted discounted cash-flows of the Company’s ongoing lease obligations. In estimating the expense and liability under its lease obligations, the Company estimated (i) the costs to be incurred to satisfy rental and build-out commitments under the lease (including operating costs), (ii) the lead-time necessary to sublease the space, (iii) the projected sublease rental rates and (iv) the anticipated durations of subleases. The Company uses a credit-adjusted risk-free rate to discount the estimated cash flows.
The Company reviews its estimates and assumptions on at least a quarterly basis, intends to continue such reviews until the termination of these facility lease obligations, and will make whatever modifications the Company believes necessary, based on the Company’s best judgment, to reflect any changed circumstances. The Company’s estimates have changed in the past, and may change in the future, resulting in additional adjustments to the estimate of these liabilities. Changes to the Company’s estimate of these liabilities are recorded as additional restructuring expenses (credits). In addition, because the Company’s estimate of these liabilities includes the application of a discount rate to reflect the time-value of money, the Company records imputed interest costs related to these liabilities each quarter. These costs are included in restructuring expenses on the Company’s consolidated statements of operations.
F-42
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
2003 Kendall Restructuring
In 2003, the Company adopted a plan to restructure its operations (the “2003 Kendall Restructuring”) to coincide with its increasing internal emphasis on advancing drug candidates through clinical development to commercialization. The restructuring was designed to re-balance the Company’s relative investments in research and development to better support the Company’s long-term strategy. At that time, the restructuring plan included a workforce reduction, write-offs of certain assets and a decision not to occupy approximately 290,000 square feet of specialized laboratory and office space in Cambridge, Massachusetts under lease to Vertex (the “Kendall Square Lease”). The Kendall Square Lease commenced in January 2003 and has a 15-year term. In 2005, the Company revised its assessment of its real estate requirements and decided to use approximately 120,000 square feet of the facility subject to the Kendall Square Lease (the “Kendall Square Facility”) for its operations, beginning in 2006. The rentable square footage of the Kendall Square Facility related to the 2003 Kendall Restructuring currently is subleased to third parties.
The restructuring expense incurred from the second quarter of 2003 through the end of the first quarter of 2005 (i.e., immediately prior to the Company’s decision to use a portion of the Kendall Square Facility for its operations) related to the estimated incremental net ongoing lease obligations associated with the entire Kendall Square Facility, together with imputed interest costs relating to the restructuring liability. The restructuring expense incurred in the period beginning in the second quarter of 2005 relates only to the portion of the Kendall Square Facility that the Company was not occupying and did not intend to occupy for its operations. The Company uses a discount rate of 10% related to this restructuring activity.
The remaining lease obligations, which are associated with the 120,000 square foot portion of the Kendall Square Facility that the Company occupied and used for its operations, were recorded as rental expense in the period incurred until the Company incurred a cease use charge related to this portion of the Kendall Square Facility in the third quarter of 2014 in connection with transitioning its Massachusetts operations to Fan Pier in Boston, Massachusetts (the “Fan Pier Move Restructuring”).
The activity related to restructuring and other liability for 2003 was as follows:
Restructuring Expense | Cash Payments | Non-cash Expense | Liability as of December 31, 2003 | ||||||||||||
(in thousands) | |||||||||||||||
Lease restructuring and other operating lease expense | $ | 84,726 | $ | (15,200 | ) | $ | — | $ | 69,526 | ||||||
Employee severance, benefits and related costs | 2,616 | (2,616 | ) | — | — | ||||||||||
Leasehold improvements and asset impairments | 4,482 | — | (4,482 | ) | — | ||||||||||
Total | $ | 91,824 | $ | (17,816 | ) | $ | (4,482 | ) | $ | 69,526 | |||||
In 2003, the lease restructuring and other operating lease expense included $78.7 million of lease restructuring expense and $6.0 million of lease operating expense incurred prior to the decision not to occupy the Kendall Square Facility. The restructuring accrual as of December 31, 2003 related only to the lease restructuring expense.
The activities related to 2003 restructuring liability for 2004 through 2016 were as follows:
2016 | 2015 | 2014 | 2004-2016 | ||||||||||||
(in thousands) | |||||||||||||||
Liability, beginning of the period | $ | 7,944 | $ | 11,596 | $ | 19,115 | $ | 69,526 | |||||||
Cash payments | (15,841 | ) | (14,625 | ) | (17,494 | ) | (226,912 | ) | |||||||
Cash received from subleases | 11,892 | 11,089 | 12,912 | 111,601 | |||||||||||
Credit for portion of facility Vertex decided to occupy in 2005 | — | — | — | (10,018 | ) | ||||||||||
Restructuring expense | 333 | (116 | ) | (2,937 | ) | 60,131 | |||||||||
Liability, end of the period | $ | 4,328 | $ | 7,944 | $ | 11,596 | $ | 4,328 | |||||||
F-43
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
Fan Pier Move Restructuring
In connection with the relocation of its Massachusetts operations to Fan Pier in Boston, Massachusetts, which commenced in 2013, the Company is incurring restructuring charges related to its remaining lease obligations at its facilities in Cambridge, Massachusetts. The majority of these restructuring charges were recorded in the third quarter of 2014 upon decommissioning three facilities in Cambridge. The Company discounted the estimated cash flows related to the facilities at a discount rate of 9%. The Company will continue to incur charges through April 2018 related to the difference between the Company’s estimated future cash flows related to its lease obligations, which include an estimate for sublease income to be received if applicable, and its actual cash flows. The Fan Pier Move Restructuring included lease obligations related to the 120,000 square feet of the Kendall Square Facility that the Company continued to use for its operations following its 2013 Kendall Restructuring. The remaining rentable square footage of the Kendall Square Facility related to the Fan Pier Move Restructuring was subleased to a third party in February 2015.
The activities related to the Fan Pier relocation restructuring liability for the three years ended December 31, 2016 were as follows:
2016 | 2015 | 2014 | |||||||||
(in thousands) | |||||||||||
Liability, beginning of the period | $ | 5,964 | $ | 33,390 | $ | 797 | |||||
Cash payments | (12,674 | ) | (30,022 | ) | (18,271 | ) | |||||
Cash received from subleases | 9,751 | 4,229 | — | ||||||||
Restructuring expense | 585 | (1,633 | ) | 50,864 | |||||||
Liability, end of the period | $ | 3,626 | $ | 5,964 | $ | 33,390 | |||||
Other Restructuring Activities
The Company has engaged in several other restructuring activities that are unrelated to its 2003 Kendall Restructuring and the Fan Pier Move Restructuring. The most significant activity commenced in October 2013 when the Company adopted a restructuring plan that included (i) a workforce reduction primarily related to the commercial support of INCIVEK following the continued and rapid decline in the number of patients being treated with INCIVEK as new medicines for the treatment of HCV infection neared approval and (ii) the write-off of certain assets. This action resulted from the Company’s decision to focus its investment on future opportunities in CF and other research and development programs.
The activities related to the Company’s other restructuring liabilities for the three years ended December 31, 2016 were as follows:
2016 | 2015 | 2014 | |||||||||
(in thousands) | |||||||||||
Liability, beginning of the period | $ | 1,450 | $ | 869 | $ | 8,441 | |||||
Cash payments | (1,794 | ) | (3,374 | ) | (10,570 | ) | |||||
Restructuring expense | 344 | 3,955 | 2,998 | ||||||||
Liability, end of the period | $ | — | $ | 1,450 | $ | 869 | |||||
R. | Employee Benefits |
The Company has a 401(k) retirement plan (the “Vertex 401(k) Plan”) in which substantially all of its permanent U.S. employees are eligible to participate. Participants may contribute up to 60% of their annual compensation to the Vertex 401(k) Plan, subject to statutory limitations. The Company may declare discretionary matching contributions to the Vertex 401(k) Plan. Beginning in mid-2013, the Company began paying matching contributions in the form of cash. For the years ended December 31, 2016, 2015 and 2014, the Company contributed approximately $11.8 million, $12.8 million and $12.0 million to the plan, respectively. As of December 31, 2016, 755,000 shares of common stock remained available for grant under the Vertex 401(k) Plan.
F-44
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
S. | Commitments and Contingencies |
Lease Obligations
The Company moved into its corporate headquarters to Boston, Massachusetts in January 2014. In December 2015, the Company entered into a lease agreement for 3215 Merryfield Row, San Diego, California. Please refer to Note L, “Long Term Obligations,” for additional information regarding both of these commitments.
The Kendall Square Lease began in January 2003 and will expire in April 2018. The Company occupied and used for its operations approximately 120,000 square feet of the Kendall Square Facility until 2014 when it moved its operations to Fan Pier. The Company has sublease arrangements in place for the remaining rentable square footage of the Kendall Square Facility, with terms that expire concurrently with the Kendall Square Lease. Please refer to Note Q, “Restructuring Expenses,” for further information.
As of December 31, 2016, future minimum commitments under the facility leases with terms of more than one year and contractual sublease income under the Company’s subleases for the Kendall Square Facility were as follows:
Year | Fan Pier Leases | San Diego Leases | Kendall Square Lease | Kendall Sublease Income | Other Leases | Total Lease Commitments (Net of Sublease Income) | ||||||||||||||||||
(in thousands) | ||||||||||||||||||||||||
2017 | $ | 67,206 | $ | 3,147 | $ | 20,088 | $ | (15,687 | ) | $ | 13,156 | $ | 87,910 | |||||||||||
2018 | 67,206 | 3,245 | 6,696 | (5,236 | ) | 12,975 | 84,886 | |||||||||||||||||
2019 | 72,589 | 6,906 | — | — | 11,746 | 91,241 | ||||||||||||||||||
2020 | 72,589 | 9,208 | — | — | 11,100 | 92,897 | ||||||||||||||||||
2021 | 72,589 | 9,208 | — | — | 10,300 | 92,097 | ||||||||||||||||||
Thereafter | 535,032 | 138,217 | — | — | 62,409 | 735,658 | ||||||||||||||||||
Total minimum lease payments | $ | 887,211 | $ | 169,931 | $ | 26,784 | $ | (20,923 | ) | $ | 121,686 | $ | 1,184,689 | |||||||||||
During 2016, 2015 and 2014, rental expense was $19.1 million, $18.1 million and $38.9 million, respectively. The majority of the Company’s lease payments related to the Fan Pier Leases are recorded as interest expense because the Company is deemed for accounting purposes to be the owner of the Buildings. Please refer to Note L, “Long Term Obligations,” for further information.
The Company has outstanding leases, which are accounted for as capital leases, for equipment, leasehold improvements and software licenses. The capital leases bear interest at rates ranging from less than 1% to 9% per year. The following table sets forth the Company’s future minimum payments due under capital leases as of December 31, 2016:
Year | (in thousands) | |||
2017 | $ | 21,995 | ||
2018 | 21,393 | |||
2019 | 8,778 | |||
2020 | 3,336 | |||
2021 | 2,457 | |||
Thereafter | 543 | |||
Total payments | 58,502 | |||
Less: amount representing interest | (4,100 | ) | ||
Present value of payments | $ | 54,402 | ||
In addition, the Company has committed to make potential future milestone and royalty payments pursuant to certain collaboration agreements. Payments generally become due and payable upon the achievement of certain developmental, regulatory and/or commercial milestones. Please refer to Note B, “Collaborative Arrangements,” for further information.
F-45
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
Litigation
On May 28, 2014, a purported shareholder class action Local No. 8 IBEW Retirement Plan & Trust v. Vertex Pharmaceuticals Incorporated, et al. was filed in the United States District Court for the District of Massachusetts, naming the Company and certain of the Company’s current and former officers and directors as defendants. The lawsuit alleged that the Company made material misrepresentations and/or omissions of material fact in the Company’s disclosures during the period from May 7, 2012 through May 29, 2012, all in violation of Section 10(b) of the Securities Exchange Act of 1934, as amended, and Rule 10b-5 promulgated thereunder. The purported class consists of all persons (excluding defendants) who purchased the Company’s common stock between May 7, 2012 and May 29, 2012. The plaintiffs sought unspecified monetary damages, costs and attorneys’ fees as well as disgorgement of the proceeds from certain individual defendants’ sales of the Company’s stock. On October 8, 2014, the Court approved Local No. 8 IBEW Retirement Fund as lead plaintiff, and Scott and Scott LLP as lead counsel for the plaintiff and the putative class. On September 30, 2015, the court granted the Company’s motion to dismiss. On October 15, 2015, the plaintiff filed a notice of appeal. In 2016, the parties filed briefs with, and presented oral arguments to, the First Circuit Court of Appeals. On October 3, 2016, the First Circuit Court of Appeals affirmed the district court’s dismissal of the plaintiff’s complaint. The times for petitioning the U.S. Court of Appeals for the First Circuit for an en banc rehearing as well as filing a petition for certiorari to the U.S. Supreme Court both have passed. As of December 31, 2016, the Company has not recorded any reserves for this purported class action.
Guaranties and Indemnifications
As permitted under Massachusetts law, the Company’s Articles of Organization and By-laws provide that the Company will indemnify certain of its officers and directors for certain claims asserted against them in connection with their service as an officer or director. The maximum potential amount of future payments that the Company could be required to make under these indemnification provisions is unlimited. However, the Company has purchased directors’ and officers’ liability insurance policies that could reduce its monetary exposure and enable it to recover a portion of any future amounts paid. No indemnification claims currently are outstanding, and the Company believes the estimated fair value of these indemnification arrangements is minimal.
The Company customarily agrees in the ordinary course of its business to indemnification provisions in agreements with clinical trial investigators and sites in its drug development programs, sponsored research agreements with academic and not-for-profit institutions, various comparable agreements involving parties performing services for the Company, and its real estate leases. The Company also customarily agrees to certain indemnification provisions in its drug discovery, development and commercialization collaboration agreements. With respect to the Company’s clinical trials and sponsored research agreements, these indemnification provisions typically apply to any claim asserted against the investigator or the investigator’s institution relating to personal injury or property damage, violations of law or certain breaches of the Company’s contractual obligations arising out of the research or clinical testing of the Company’s compounds or drug candidates. With respect to lease agreements, the indemnification provisions typically apply to claims asserted against the landlord relating to personal injury or property damage caused by the Company, to violations of law by the Company or to certain breaches of the Company’s contractual obligations. The indemnification provisions appearing in the Company’s collaboration agreements are similar to those for the other agreements discussed above, but in addition provide some limited indemnification for its collaborator in the event of third-party claims alleging infringement of intellectual property rights. In each of the cases above, the indemnification obligation generally survives the termination of the agreement for some extended period, although the Company believes the obligation typically has the most relevance during the contract term and for a short period of time thereafter. The maximum potential amount of future payments that the Company could be required to make under these provisions is generally unlimited. The Company has purchased insurance policies covering personal injury, property damage and general liability that reduce its exposure for indemnification and would enable it in many cases to recover all or a portion of any future amounts paid. The Company has never paid any material amounts to defend lawsuits or settle claims related to these indemnification provisions. Accordingly, the Company believes the estimated fair value of these indemnification arrangements is minimal.
F-46
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
Other Contingencies
The Company has certain contingent liabilities that arise in the ordinary course of its business activities. The Company accrues a reserve for contingent liabilities when it is probable that future expenditures will be made and such expenditures can be reasonably estimated. There were no material contingent liabilities accrued as of December 31, 2016 or 2015.
T. | Segment Information |
Segment reporting is prepared on the same basis that the Company’s chief executive officer, who is the Company’s chief operating decision maker, manages the business, makes operating decisions and assesses performance. The Company operates in one segment, pharmaceuticals. Enterprise-wide disclosures about revenues, significant customers, and property and equipment, net by location are presented below.
Revenues by Product
Product revenues, net consisted of the following:
2016 | 2015 | 2014 | |||||||||
(in thousands) | |||||||||||
KALYDECO | $ | 703,432 | $ | 631,674 | $ | 463,750 | |||||
ORKAMBI | 979,590 | 350,663 | — | ||||||||
INCIVEK | 610 | 17,987 | 24,071 | ||||||||
Total product revenues, net | $ | 1,683,632 | $ | 1,000,324 | $ | 487,821 | |||||
Revenues by Geographic Location
Total revenues from external customers and collaborators by geographic region consisted of the following. Product revenues are attributed to countries based on the location of the customer. Collaborative revenues are attributed to the operations of the Company in the United States. Royalty revenues are attributed to countries based on the location of the collaborator.
2016 | 2015 | 2014 | |||||||||
(in thousands) | |||||||||||
United States | $ | 1,321,807 | $ | 763,316 | $ | 361,074 | |||||
Outside of the United States | |||||||||||
Europe | 320,456 | 219,596 | 197,611 | ||||||||
Other | 59,914 | 49,424 | 21,730 | ||||||||
Total revenues outside of the United States | 380,370 | 269,020 | 219,341 | ||||||||
Total revenues | $ | 1,702,177 | $ | 1,032,336 | $ | 580,415 | |||||
In 2016, 2015 and 2014, revenues attributable to the United Kingdom were the largest contributor to the Company’s European revenues.
F-47
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
Significant Customers
Gross revenues and accounts receivable from each of the Company’s customers who individually accounted for 10% or more of total gross revenues and/or 10% or more of total gross accounts receivable consisted of the following:
Percent of Total Gross Revenues | Percent of Gross Accounts Receivable | |||||||||||||
Year Ended December 31, | As of December 31, | |||||||||||||
2016 | 2015 | 2014 | 2016 | 2015 | ||||||||||
Walgreen Co. | 19 | % | 20 | % | 12 | % | 15 | % | 15 | % | ||||
CVS/Caremark | 19 | % | 17 | % | <10 | % | 17 | % | 17 | % | ||||
Accredo/Curascript | 15 | % | 15 | % | <10 | % | 10 | % | 16 | % | ||||
Property and Equipment, Net by Location
Property and equipment, net by location consisted of the following:
As of December 31, | |||||||
2016 | 2015 | ||||||
(in thousands) | |||||||
United States | $ | 665,552 | $ | 661,421 | |||
Outside of the United States | |||||||
United Kingdom | 26,921 | 32,793 | |||||
Other | 5,889 | 3,501 | |||||
Total property and equipment, net outside of the United States | 32,810 | 36,294 | |||||
Total property and equipment, net | $ | 698,362 | $ | 697,715 | |||
F-48
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
U. | Quarterly Financial Data (unaudited) |
The following table sets forth the Company’s quarterly financial data for the two years ended December 31, 2016.
Three Months Ended | |||||||||||||||
March 31, 2016 | June 30, 2016 | September 30, 2016 | December 31, 2016 | ||||||||||||
(in thousands, except per share amounts) | |||||||||||||||
Revenues: | |||||||||||||||
Product revenues, net | $ | 394,410 | $ | 425,651 | $ | 409,689 | $ | 453,882 | |||||||
Royalty revenues | 3,596 | 5,282 | 3,835 | 3,887 | |||||||||||
Collaborative revenues | 74 | 675 | 259 | 937 | |||||||||||
Total revenues | 398,080 | 431,608 | 413,783 | 458,706 | |||||||||||
Costs and expenses: | |||||||||||||||
Cost of product revenues | 49,789 | 44,154 | 53,222 | 59,646 | |||||||||||
Royalty expenses | 860 | 1,098 | 855 | 836 | |||||||||||
Research and development expenses (1) | 255,860 | 271,008 | 272,370 | 248,452 | |||||||||||
Sales, general and administrative expenses | 105,214 | 111,652 | 106,055 | 109,908 | |||||||||||
Restructuring expenses | 687 | 343 | 8 | 224 | |||||||||||
Total costs and expenses | 412,410 | 428,255 | 432,510 | 419,066 | |||||||||||
(Loss) income from operations | (14,330 | ) | 3,353 | (18,727 | ) | 39,640 | |||||||||
Interest expense, net | (20,698 | ) | (20,155 | ) | (20,140 | ) | (20,439 | ) | |||||||
Other income (expense), net | 4,411 | (1,219 | ) | (167 | ) | 1,105 | |||||||||
(Loss) income before provision for income taxes | (30,617 | ) | (18,021 | ) | (39,034 | ) | 20,306 | ||||||||
Provision for (benefit from) income taxes | 5,485 | 18,130 | 503 | (7,453 | ) | ||||||||||
Net (loss) income | (36,102 | ) | (36,151 | ) | (39,537 | ) | 27,759 | ||||||||
(Income) loss attributable to noncontrolling interest | (5,529 | ) | (28,374 | ) | 696 | 5,186 | |||||||||
Net (loss) income attributable to Vertex | $ | (41,631 | ) | $ | (64,525 | ) | $ | (38,841 | ) | $ | 32,945 | ||||
Amounts per share attributable to Vertex common shareholders: | |||||||||||||||
Net (loss) income: | |||||||||||||||
Basic | $ | (0.17 | ) | $ | (0.26 | ) | $ | (0.16 | ) | $ | 0.13 | ||||
Diluted | $ | (0.17 | ) | $ | (0.26 | ) | $ | (0.16 | ) | $ | 0.13 | ||||
Shares used in per share calculations: | |||||||||||||||
Basic | 243,831 | 244,482 | 244,920 | 245,454 | |||||||||||
Diluted | 243,831 | 244,482 | 244,920 | 247,757 | |||||||||||
F-49
VERTEX PHARMACEUTICALS INCORPORATED
Notes to Consolidated Financial Statements (Continued)
Three Months Ended | |||||||||||||||
March 31, 2015 | June 30, 2015 | September 30, 2015 | December 31, 2015 | ||||||||||||
(in thousands, except per share amounts) | |||||||||||||||
Revenues: | |||||||||||||||
Product revenues, net | $ | 130,875 | $ | 160,388 | $ | 302,511 | $ | 406,550 | |||||||
Royalty revenues | 6,792 | 5,077 | 5,759 | 6,331 | |||||||||||
Collaborative revenues | 842 | 611 | 1,546 | 5,054 | |||||||||||
Total revenues | 138,509 | 166,076 | 309,816 | 417,935 | |||||||||||
Costs and expenses: | |||||||||||||||
Cost of product revenues | 9,381 | 15,409 | 30,269 | 62,092 | |||||||||||
Royalty expenses | 2,926 | 1,451 | 1,691 | 1,293 | |||||||||||
Research and development expenses (2) | 215,599 | 223,858 | 246,284 | 310,181 | |||||||||||
Sales, general and administrative expenses | 85,860 | 94,394 | 99,772 | 96,549 | |||||||||||
Restructuring (income) expenses | (3,272 | ) | 2,128 | 1,826 | 1,524 | ||||||||||
Total costs and expenses | 310,494 | 337,240 | 379,842 | 471,639 | |||||||||||
Loss from operations | (171,985 | ) | (171,164 | ) | (70,026 | ) | (53,704 | ) | |||||||
Interest expense, net | (21,307 | ) | (21,111 | ) | (21,134 | ) | (20,654 | ) | |||||||
Other (expense) income, net | (5,113 | ) | 1,414 | (1,326 | ) | (1,690 | ) | ||||||||
Loss before provision for (benefit from) income taxes | (198,405 | ) | (190,861 | ) | (92,486 | ) | (76,048 | ) | |||||||
Provision for (benefit from) income taxes | 299 | 30,131 | 1,330 | (1,379 | ) | ||||||||||
Net loss | (198,704 | ) | (220,992 | ) | (93,816 | ) | (74,669 | ) | |||||||
Loss (income) attributable to noncontrolling interest | 98 | 32,144 | (1,333 | ) | 938 | ||||||||||
Net loss attributable to Vertex | $ | (198,606 | ) | $ | (188,848 | ) | $ | (95,149 | ) | $ | (73,731 | ) | |||
Amounts per share attributable to Vertex common shareholders: | |||||||||||||||
Net loss: | |||||||||||||||
Basic and diluted | $ | (0.83 | ) | $ | (0.78 | ) | $ | (0.39 | ) | $ | (0.30 | ) | |||
Shares used in per share calculations: | |||||||||||||||
Basic and diluted | 239,493 | 240,757 | 241,969 | 242,987 | |||||||||||
1. | In the second quarter of 2016, the Company incurred research and development expenses of approximately $10.0 million to acquire certain early-stage research assets. In the third quarter of 2016, the Company incurred research and development expenses related to a $20.0 million upfront payment to Moderna Therapeutics, Inc. See Note B, “Collaborative Arrangements,” for further information. |
2. | In the fourth quarter of 2015, the Company made a $75.0 million upfront payment to CRISPR Therapeutics in connection with the collaboration, which was recorded as a research and development expense. See Note B, “Collaborative Arrangements,” for further information. |
V. | Subsequent Events |
In February 2017, the Company decided to consolidate its research activities into its Boston, Milton Park and San Diego locations. As a result, the Company plans to close its Laval, Canada site. In connection with this decision, approximately 70 positions were affected and the Company estimates that it will incur aggregate restructuring expenses of approximately $10 million in the first quarter of 2017.
In January 2017, the Company entered into the Merck KGaA Agreement. A description of the Merck KGaA Agreement is set forth in Note B, “Collaborative Arrangements.” In February 2017, the Company repaid all outstanding borrowings under the Credit Agreement. Further information on the Credit Agreement, including the February 2017 repayment, is set forth in Note L, “Long Term Obligations.”
F-50
