Attached files
| file | filename |
|---|---|
| EX-23.1 - CONSENTS OF EXPERTS AND COUNSEL - ENDRA Life Sciences Inc. | end_ex231.htm |
| EX-10.7 - NON-EMPLOYEE DIRECTOR COMPENSATION POLICY - ENDRA Life Sciences Inc. | end_ex107.htm |
As filed with the Securities and Exchange Commission on
January 20, 2017
Registration No. 333-214724
UNITED STATES SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
AMENDMENT NO. 5
TO
FORM S-1
REGISTRATION STATEMENT UNDER THE SECURITIES ACT OF
1933
ENDRA LIFE SCIENCES INC.
(Exact name of registrant as
specified in its charter)
|
Delaware
|
|
3845
|
|
26-0579295
|
|
(State or other jurisdiction
of
|
|
(Primary Standard
Industrial
|
|
(I.R.S. Employer
|
|
incorporation or organization)
|
|
Classification Code Number)
|
|
Identification No.)
|
ENDRA
Life Sciences Inc.
3600
Green Court, Suite 350
Ann
Arbor, MI 48105
(734)
335-0468
(Address, including zip code,
and telephone number, including area code, of registrant’s
principal executive offices)
Francois
Michelon
Chief
Executive Officer
ENDRA
Life Sciences Inc.
3600
Green Court, Suite 350
Ann
Arbor, MI 48105
(734)
335-0468
(Name, address, including zip
code, and telephone number, including area code, of agent for
service)
Copies to:
|
Mark R.
Busch
K&L
Gates LLP
214
North Tryon St., 47th Floor
Charlotte,
North Carolina 28202
Telephone:
(704) 331-7440
|
Jonathan R. Zimmerman
Ben A.
Stacke
Faegre Baker Daniels LLP
2200 Wells Fargo Center
90 South Seventh Street
Minneapolis, MN 55402-3901
Telephone:
(612) 766-7000
|
As soon as practicable after the effective date of this
Registration Statement.
(Approximate date of
commencement of proposed sale to the public)
If any of the securities being registered on this
Form are to be offered on a delayed or continuous basis pursuant to
Rule 415 under the Securities Act of 1933 check the following
box. [X]
If
this Form is filed to register additional securities for an
offering pursuant to Rule 462(b) under the Securities Act, please
check the following box and list the Securities Act registration
statement number of the earlier effective registration statement
for the same offering. [ ]
If
this Form is a post-effective amendment filed pursuant to Rule
462(c) under the Securities Act, check the following box and list
the Securities Act registration statement number of the earlier
effective registration statement for the same offering.
[ ]
If
this Form is a post-effective amendment filed pursuant to Rule
462(d) under the Securities Act, check the following box and list
the Securities Act registration statement number of the earlier
effective registration statement for the same offering.
[ ]
Indicate
by check mark whether the registrant is a large accelerated filer,
an accelerated filer, a non-accelerated filer, or a smaller
reporting company. See the definitions of “large accelerated
filer,” “accelerated filer” and “smaller
reporting company” in Rule 12b-2 of the Exchange Act (check
one):
|
Large
accelerated filer [ ]
|
|
Accelerated
filer
[ ]
|
|
Non-accelerated
filer [ ]
|
|
Smaller
reporting company [X]
|
|
(Do not check if a smaller reporting
company)
|
|
|
CALCULATION
OF REGISTRATION FEE
|
Title
of Each Class of Securities to be Registered
|
Amount to be
Registered
|
Proposed Maximum Aggregate
Offering Price Per Share
|
Proposed
Maximum
Aggregate
Offering Price(1)
|
Amount
of
Registration
Fee
|
|
Units, each
consisting of one share of common stock, par
value $0.0001 per share, and a warrant to purchase one share
of common stock(2)
|
2,300,000
|
$ 5.50
|
$12,650,000
|
$1,466.14
|
|
Common Stock
included in the units(3)
|
--
|
--
|
$--
|
$--
|
|
Warrants to
purchase Common Stock included in the
units(3)
|
--
|
--
|
$--
|
$--
|
|
Common Stock
underlying Warrants included in the
units(2)
|
2,300,000
|
$ 6.60
|
$15,180,000
|
$1,759.36
|
|
Underwriters'
warrants(3)(4)
|
--
|
--
|
$--
|
$--
|
|
Shares of Common
Stock underlying Underwriters'
Warrants(4)
|
184,000
|
$6.60
|
$ 1,214,000
|
$ 140.75
|
|
Total Registration
Fee(5)
|
|
|
$29,044,000
|
$ 3,366.25
|
(1)
Estimated solely
for the purpose of calculating the amount of the registration fee
pursuant to Rule 457(o) under the Securities Act of 1933, as
amended. Includes the offering price of the shares and warrants
that the underwriters have the option to purchase to cover
over-allotments, if any.
(2)
Pursuant to Rule
416 under the Securities Act of 1933, as amended, there is also
being registered hereby such indeterminate number of additional
shares of common stock of the registrant as may be issued or
issuable because of stock splits, stock dividends, stock
distributions, and similar transactions.
(3)
No separate
registration fee required pursuant to Rule 457(g) under the
Securities Act of 1933, as amended.
(4)
Represents
warrants granted to the underwriters to
purchase shares of common stock in an amount up to 8% of the number
of shares sold to the public in this offering. See
“Underwriting” contained within this Registration
Statement for information on underwriting arrangements relating to
this offering.
(5)
The
registrant previously paid $2,486.56 in registration fees in
connection with a previous filing of this Registration
Statement. The
registrant accordingly has paid the difference of $879.69 with this
filing.
The
registrant hereby amends this Registration Statement on such date
or dates as may be necessary to delay its effective date until the
registrant shall file a further amendment, which specifically
states that this Registration Statement shall thereafter become
effective in accordance with Section 8(a) of the Securities Act of
1933, as amended, or until this Registration Statement shall become
effective on such date as the Commission, acting pursuant to said
Section 8(a), may determine.
THE INFORMATION IN THIS PROSPECTUS IS NOT COMPLETE AND MAY BE
CHANGED. THESE SECURITIES MAY NOT BE SOLD UNTIL THE REGISTRATION
STATEMENT FILED WITH THE SECURITIES AND EXCHANGE COMMISSION IS
EFFECTIVE. THIS PRELIMINARY PROSPECTUS IS NOT AN OFFER TO SELL
THESE SECURITIES AND WE ARE NOT SOLICITING AN OFFER TO BUY THESE
SECURITIES IN ANY STATE WHERE THE OFFER OR SALE IS NOT
PERMITTED.
PRELIMINARY PROSPECTUS SUBJECT TO COMPLETION, DATED JANUARY
20, 2017

ENDRA Life
Sciences Inc.
2,000,000 Units
This is
our initial public offering. We are offering 2,000,000 units
consisting of one share of our common stock and
a warrant to purchase one share of our
common stock, which numbers reflect our proposed one
for 2.50 reverse stock split described in this prospectus
(the “reverse
stock split”).
The
warrants will have an exercise price equal to 120% of the initial
public offering price per unit set forth on the cover
page of this prospectus and will expire on the fifth anniversary of
the original issuance date. The shares of our common stock and
warrants will trade together as units during the first 60
days following the date of this prospectus and, thereafter, the
units will automatically separate and the common stock and warrants
will trade separately, unless Dougherty & Company LLC
determines that an earlier separation date is
acceptable.
Prior
to this offering, there has been no public market for our
securities. We have been authorized to list on the
Nasdaq Capital Market our common stock under the symbol
“NDRA.” We have applied to list our
units and warrants on the Nasdaq Capital Market under the symbols
“NDRAU” and “NDRAW,” respectively.
We expect that the public offering price will be between $5.00 and
$5.50 per unit. After the effectiveness of the
registration statement of which this prospectus is a part, and
concurrently with the pricing of this offering, we will effect the
reverse stock split.
We are
an “emerging growth company” as that term is used in
the Jumpstart Our Business Startups Act of 2012 and, as such, have
elected to comply with certain reduced public company reporting
requirements for this prospectus and future filings. See
“Prospectus Summary – Implications of Being an Emerging
Growth Company.”
Investing
in our securities involves a high degree of risk. See “Risk
Factors” beginning on page 8 for a discussion of information
that should be considered in connection with an investment in our
securities.
|
|
Per
Unit
|
Total
|
|
Public offering
price
|
$
|
$
|
|
Underwriting
discount(1)
|
$
|
$
|
|
Proceeds, before
expenses, to us(2)
|
$
|
$
|
________
(1) We
have also granted warrants to the underwriters in connection with
this offering and agreed to reimburse the underwriters for certain
expenses incurred by them. See “Underwriting” for a
description of the compensation payable by us to the
underwriters.
(2) We
estimate the total expenses payable by us, excluding the
underwriting discount, will be approximately
$600,000.
We have
granted the underwriters a 30-day option to purchase up to 300,000
additional units to cover over-allotments, if
any, provided that in no event may exercise of this option
occur after separation of the units occurs.
Neither
the Securities and Exchange Commission nor any state securities
commission has approved or disapproved of these securities or
determined if this prospectus is truthful or
complete. Any representation to the contrary is a
criminal offense.
The
underwriters expect to deliver the units to investors
on or about ,
2017.
______________________________________
Dougherty & Company
Dawson
James Securities, Inc.
______________________________________
The
date of this prospectus is ,
2017.
TABLE
OF CONTENTS
|
|
Page
|
|
PROSPECTUS SUMMARY
|
1
|
|
THE OFFERING
|
6
|
|
SUMMARY SELECTED FINANCIAL DATA
|
7
|
|
RISK FACTORS
|
8
|
|
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS AND OTHER
INFORMATION CONTAINED IN THIS PROSPECTUS
|
32
|
|
USE OF PROCEEDS
|
33
|
|
DIVIDEND POLICY
|
34
|
|
CAPITALIZATION
|
35
|
|
DILUTION
|
36
|
|
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION
AND RESULTS OF OPERATIONS
|
38
|
|
BUSINESS
|
47
|
|
EXECUTIVE OFFICERS, DIRECTORS AND CORPORATE GOVERNANCE
|
62
|
|
EXECUTIVE COMPENSATION
|
66
|
|
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND
MANAGEMENT
|
69
|
|
CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR
INDEPENDENCE
|
72
|
|
DESCRIPTION OF THE SECURITIES WE ARE OFFERING
|
73
|
|
SHARES ELIGIBLE FOR FUTURE SALE
|
76
|
|
UNDERWRITING
|
78
|
|
NOTICE TO INVESTORS
|
81
|
|
LEGAL MATTERS
|
83
|
|
EXPERTS
|
83
|
|
WHERE YOU CAN FIND MORE INFORMATION
|
83
|
|
INDEX TO FINANCIAL STATEMENTS
|
F-1
|
Unless
otherwise stated or the context otherwise requires, the terms
“ENDRA,” “we,” “us,”
“our” and the “Company” refer to
ENDRA
Life Sciences Inc., a Delaware
corporation.
You
should rely only on the information contained in this prospectus
and any related free writing prospectus that we may provide to you
in connection with this offering. We have not, and the
underwriters have not, authorized anyone to provide you with
additional or different information. If anyone provides
you with different or inconsistent information, you should not rely
on it. We are not, and the underwriters are not, making an offer to
sell these securities in any jurisdiction where the offer or sale
is not permitted. You should assume that the information appearing
in this prospectus is accurate only as of the date on the front
cover of this prospectus. Our business, financial condition,
results of operations and prospects may have changed since that
date.
For
investors outside the United States: neither we nor the
underwriters have done anything that would permit this offering or
possession or distribution of this prospectus or any free writing
prospectus we may provide to you in connection with this offering
in any jurisdiction where action for that purpose is required,
other than in the United States. You are required to inform
yourselves about and to observe any restrictions relating to this
offering and the distribution of this prospectus and any such free
writing prospectus outside of the United States.
MARKET AND INDUSTRY DATA
Unless otherwise indicated, information contained
in this prospectus concerning our industry and the markets in which
we operate is based on information from independent industry and
research organizations, other third-party sources (including
industry publications, surveys and forecasts), and management
estimates. Management estimates are derived from publicly available
information released by independent industry analysts and
third-party sources, as well as data from our internal research,
and are based on assumptions made by us
upon reviewing such data and our knowledge of such industry and
markets which we believe to be reasonable. Although we believe the
data from these third-party sources is reliable, we have not
independently verified any third-party information. In addition,
projections, assumptions and estimates of the future performance of
the industry in which we operate and our future performance are
necessarily subject to uncertainty and risk due to a variety of
factors, including those described in “Risk Factors”
and “Special Note Regarding Forward-Looking Statements and
Other Information Contained In This Prospectus.” These and
other factors could cause results to differ materially from those
expressed in the estimates made by the independent parties and by
us.
TRADEMARKS
We operate under a number of trademarks,
including, among others, “ENDRA” and
“TAEUS,” all of which are registered under applicable
intellectual property laws. This prospectus contains references to
our trademarks and service marks and to those belonging to other
entities. Solely for convenience, trademarks and trade names
referred to in this prospectus may appear without
the ® or TM symbols,
but such references are not intended to indicate, in any way, that
we will not assert, to the fullest extent possible under applicable
law, our rights or the rights of the applicable licensor to these
trademarks and trade names. We do not intend our use or display of
other companies’ trade names, trademarks or service marks to
imply a relationship with, or endorsement or sponsorship of us by,
any other companies.
Prospectus
Summary
|
This summary highlights selected information
contained elsewhere in this prospectus and does not contain all of
the information that you should consider before investing in our
securities. You should carefully read this
prospectus and the registration statement of which this prospectus
is a part in their entirety before investing in our
securities, including the information discussed under
“Risk Factors” and our financial statements and
notes thereto that appear elsewhere in this
prospectus.
Our
Company
We have
commercialized an enhanced ultrasound technology for the
pre-clinical research market and are leveraging that expertise to
develop technology for increasing the capabilities of clinical
diagnostic ultrasound, to broaden patient access to the safe
diagnosis and treatment of a number of significant medical
conditions in circumstances where expensive X-ray computed
tomography, or CT, and magnetic resonance imaging, or MRI,
technology is unavailable or impractical.
Since
2010, we have marketed and sold our Nexus 128 system, which
combines light-based thermoacoustics and ultrasound, to address the
imaging needs of researchers studying disease models in
pre-clinical applications. Sales of the Nexus 128 system were
approximately $560,000 in 2014 and $1.4 million in
2015. We have not yet completed preparation of financial
statements for the quarter and year ended December 31, 2016, but,
as described below under “Recent Financial Results,”
based on preliminary data available to us, we expect to report
total revenues of approximately $515,000 for the quarter and year
ended December 31, 2016, all of which revenue was attributable to
sales of our Nexus 128 system. Our Nexus 128 system is used
in a number of leading global academic research centers, including
Stanford University, The University of Michigan, Shanghai Jiao Tong
University, and Purdue University. We expect to continue to sell
our Nexus 128 system to maintain a base level of revenue, but
believe the market potential for our clinical systems is much
higher.
Building on our
expertise in thermoacoustics, we have developed a next-generation
technology platform — Thermo Acoustic Enhanced Ultrasound, or
TAEUS — which is intended to enhance the capability of
clinical ultrasound technology and support the diagnosis and
treatment of a number of significant medical conditions that
currently require the use of expensive CT or MRI imaging or where
imaging is not practical using existing technology.
Unlike
the near-infrared light pulses used in our Nexus 128 system, our
TAEUS technology uses radio frequency, or RF, pulses to stimulate
tissues, using a small fraction of the energy transmitted into the
body during an MRI scan. The use of RF energy allows our TAEUS
technology to penetrate deep into tissue, enabling the imaging of
human anatomy at depths equivalent to those of conventional
ultrasound. The RF pulses are absorbed by tissue and converted into
ultrasound signals, which are detected by an external ultrasound
receiver and a digital acquisition system that is part of the TAEUS
system. The detected ultrasound is processed into images using our
proprietary algorithms and overlaid in real time onto conventional
gray-scale ultrasound images.
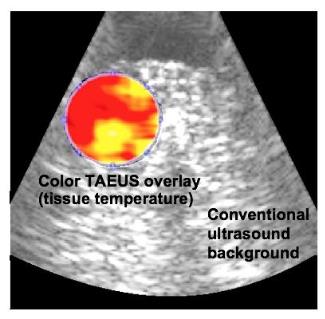
Image below: Real-time ex-vivo bovine tissue temperature analysis
overlaid on traditional ultrasound image.
 |
1
|
We
believe that our TAEUS technology has the potential to add a number
of new capabilities to conventional ultrasound and thereby enhance
the utility of both existing and new ultrasound
systems.
Our
TAEUS platform is not intended to replace CT and MRI systems, both
of which are versatile imaging technologies with capabilities and
uses beyond the focus of our business. However, they are also
expensive, with a CT system costing approximately $1 million and an
MRI system costing up to $3 million. In addition, and in contrast
to ultrasound systems, due to their limited number and the fact
that they are usually fixed-in-place at major medical facilities,
CT and MRI systems are frequently inaccessible to
patients.
We
believe that our TAEUS platform can extend the use of ultrasound
technology to a number of important applications that either
require the use of expensive CT or MRI imaging systems or where
imaging is not practical using existing
technology. To demonstrate the
capabilities of our TAEUS platform, we have conducted various
internal ex-vivo laboratory experiments and have also conducted
limited internal in-vivo large animal studies. However, we have not
yet conducted any human studies and these capabilities are not
supported by clinical data that we have gathered in pursuit of
obtaining regulatory approvals or that was subject to regulatory
oversight and guidance. In our ex-vivo and in-vivo testing,
we have demonstrated that the TAEUS platform has the following
capabilities and potential clinical applications:
●
Tissue Composition:
Our TAEUS technology enables ultrasound to distinguish fat from
lean tissue. This capability would enable the use of TAEUS-enhanced
ultrasound for the early identification, staging and monitoring of
Non-Alcoholic Fatty Liver Disease, or NAFLD, which affects an
estimated 1.4 billion people worldwide and is a precursor to liver
fibrosis, cirrhosis and liver cancer.
●
Temperature
Monitoring: Our TAEUS technology enables traditional ultrasound to
visualize changes in tissue temperature, in real time. This
capability would enable the use of TAEUS-enhanced ultrasound to
guide thermoablative therapy, which uses heat or cold to remove
tissue, such as in the treatment of cardiac atrial fibrillation, or
removal of cancerous liver and kidney lesions, with greater
accuracy.
●
Vascular Imaging:
Our TAEUS technology enables ultrasound to view blood vessels from
any angle, using only a saline solution contrasting agent, unlike
Doppler ultrasound which requires precise viewing angles. This
capability would enable the use of TAEUS-enhanced ultrasound to
easily identify arterial plaque or malformed
vessels.
●
Tissue Perfusion:
Our TAEUS technology enables ultrasound to image blood flow at the
capillary level in a region, organ or tissue. This capability could
be used to assist physicians in characterizing microvasculature
fluid flows symptomatic of damaged tissue, such as internal
bleeding from trauma, or diseased tissue, such as certain
cancers.
|
2
|
Sales
of ultrasound diagnostic equipment were approximately $4 billion
globally in 2014 and are expected to grow at approximately 4.4%
annually. There are approximately 800,000 installed systems
generating over 400 million annual diagnostic ultrasound procedures
globally. Additionally, an estimated 30,000 to 50,000 new and
replacement systems are sold into the market each year. These
numbers include both portable and cart-based ultrasound systems,
and cover all types of diagnostic ultrasound procedures, including
systems intended for cardiology, prenatal and abdominal use. We do
not intend to address low-cost, portable ultrasound systems and
systems focused on applications, such as prenatal care, where we
believe our TAEUS technology will not substantially impact patient
care. Accordingly, we define our addressable market for one or more
of our TAEUS applications at approximately 300,000 cart-based
ultrasound systems currently in use throughout the
world.
After
approval, our TAEUS technology can be added as an accessory to
existing ultrasound systems, helping to improve clinical
decision-making on the front lines of patient care, without
requiring new clinical workflows or large capital investments. We
are also developing TAEUS for incorporation into new ultrasound
systems, primarily through our collaboration with GE Healthcare,
described more fully below. We are not aware of any other
ultrasound devices in development that include the anticipated
functionality of our planned TAEUS applications.
Based
on our design work and our understanding of the ultrasound
accessory market, we intend to price our initial NAFLD TAEUS
application at a price point approximating one-half of the price of
a new cart-based ultrasound system, which should enable purchasers
to recoup their investment in less than one year by performing a
relatively small number of additional ultrasound procedures. We
further believe that clinicians will be attracted to our technology
because it will enable them to perform more procedures with their
existing ultrasound equipment, thereby retaining more imaging
patients in their clinics rather than referring patients out to a
regional medical center for a CT or MRI scan.
We
expect that the first-generation TAEUS application will be a
standalone ultrasound accessory designed to cost-effectively
quantify fat in the liver and stage progression of NAFLD, which can
only be achieved today with impractical surgical biopsies or MRI
scans. Subsequent TAEUS applications are expected to be implemented
via a second generation hardware platform that can run multiple
clinical software applications that we will offer TAEUS users for a
one-time licensing fee – adding ongoing customer value to the
TAEUS platform and a growing software revenue stream for
ENDRA.
Each of
our TAEUS platform applications will require regulatory approvals
before we are able to sell or license the application. Based on
certain factors, such as the installed base of ultrasound systems,
availability of other imaging technologies, such as CT and MRI,
economic strength and applicable regulatory requirements, we intend
to seek initial approval of our applications for sale in the
European Union, followed by the United States and
China.
We
believe that our NAFLD TAEUS application will qualify for sale in
the European Union as a Class IIa medical device. As a result, we
will be required to obtain a CE mark for our NAFLD TAEUS
application before we can sell the application in the European
Union. Existing regulations would not require us to conduct a
clinical trial to obtain a CE mark for this application.
Nonetheless, for commercial reasons and to support our CE mark
application, we plan to conduct a limited (less than 10 person)
trial to demonstrate our NAFLD TAEUS application’s ability to
distinguish fat from lean tissue. Based on our understanding of
applicable regulations and consultations with medical device
regulatory consulting firms and medical device contract engineering
firms, we expect to receive a CE mark for our NAFLD TAEUS
application approximately ten months after the completion of this
offering. However, this estimate is subject to uncertainty and
there can be no assurance that this process will not take longer or
be more costly than we expect. While we are seeking a CE mark for
our NAFLD TAEUS application, we also plan to expand our sales,
marketing, customer support and contract manufacturing
capabilities, so that we can commence commercial sales of the
application in the European Union promptly following receipt of
this regulatory approval. Following receipt of such CE mark and
placement of initial systems with researchers and universities, we
plan to conduct one or more clinical studies to further demonstrate
this product’s capabilities.
While
the process of obtaining a CE mark for our NAFLD TAEUS application
is underway, we also intend to prepare for submission to the U.S.
Food and Drug Administration, or the FDA, an application under the
Food, Drug and Cosmetic Act, or the FD&C Act, to sell our NAFLD
TAEUS application in the U.S. We anticipate that the application,
as well as those for our other TAEUS applications, will be
submitted for approval under Section 510(k) of the FD&C Act. In
connection with our initial submission to the FDA, we believe we
will be required to provide imaging verification and validation
testing data, as well as the data from the limited trial we plan to
conduct to support our CE mark application. We expect that our
initial FDA clearance will allow us to sell the NAFLD TAEUS
application in the U.S. with general imaging claims. However, we
will need to obtain additional FDA clearances to be able to make
diagnostic claims for fatty tissue content determination.
Accordingly, to support our commercialization efforts we expect
that, following receipt of our initial FDA clearance, we will
submit one or more additional applications to the FDA, each of
which will need to include additional clinical trial data, so that
following receipt of the necessary clearances we may make those
diagnostic claims. Based on our understanding of applicable
regulations and consultations with medical device regulatory
consulting firms and medical device contract engineering firms, we
expect to submit our initial FDA application to the FDA
approximately eleven months after the completion of this offering
and that the FDA will make a final determination on our application
approximately eleven months after it is submitted. However, these
estimates are subject to uncertainty and there can be no assurance
that these processes will not take longer or be more costly than we
expect.
In
April 2016, we entered into a Collaborative Research Agreement with
General Electric Company, acting through its GE Healthcare business
unit and the GE Global Research Center, or GE Healthcare. Under the
terms of the agreement, GE Healthcare has agreed to assist us in
our efforts to commercialize our TAEUS technology for use in a
fatty liver application by, among other things, providing equipment
and technical advice, and facilitating introductions to GE
Healthcare clinical ultrasound customers. In return for this
assistance, we have agreed to afford GE Healthcare certain rights
of first offer with respect to manufacturing and licensing rights
for the target application. More specifically, we have agreed
that, prior to commercially releasing our NAFLD TAEUS application,
we will offer to negotiate an exclusive ultrasound manufacturer
relationship with GE Healthcare for a period of at least one year
of commercial sales. The commercial sales would involve, within our
sole discretion, either our Company commercially selling GE
Healthcare ultrasound systems as the exclusive ultrasound system
with our TAEUS fatty liver application embedded, or GE Healthcare
being the exclusive ultrasound manufacturer to sell ultrasound
systems with our TAEUS fatty liver application embedded. The
agreement has a term of one year and is subject to termination by
either party upon not less than 60 days’ notice.
|
3
|
Risks
Related to Our Business
An
investment in our securities involves a high degree of
risk. You should carefully consider the risks summarized below.
These risks are discussed more fully in the “Risk
Factors” section of this prospectus immediately following
this prospectus summary. These risks include, but are not limited
to, the following:
●
We have a history
of operating losses, and we may never achieve or maintain
profitability.
●
Our limited
operating history makes it difficult to evaluate our current
business, predict our future results or forecast our financial
performance and growth.
●
Our efforts may
never result in the successful development of commercial
applications based on our TAEUS technology.
●
If we fail to
obtain and maintain necessary regulatory clearances or approvals
for our TAEUS applications, or if clearances or approvals for
future applications and indications are delayed or not issued, our
commercial operations will be harmed.
●
We will depend on
third parties to design, manufacture and seek regulatory approval
of our TAEUS applications. If any third party fails to successfully
design, manufacture and gain regulatory approval of our TAEUS
applications, our business will be materially harmed.
●
Competition in the
medical imaging market is intense and we may be unable to
successfully compete.
●
We intend to market
our TAEUS applications, if approved, globally, in which case we
will be subject to the risks of doing business outside of the
United States.
●
If we are unable to
protect our intellectual property, then our financial condition,
results of operations and the value of our technology and products
could be adversely affected.
●
There
are no established public trading markets
for the securities being offered in this offering and
active trading markets for these securities may not
develop.
Reverse Stock Split
In
December 2016, our board of directors and stockholders holding a
majority of the outstanding shares of our common stock approved
resolutions authorizing our board of directors to effect a reverse
split of our common stock at certain exchange ratios ranging from
1:1.50 to
1:2.50, with our board of directors
retaining the discretion as to whether to implement the reverse
stock split and which exchange ratio to implement. Following the
effectiveness of the registration statement of which this
prospectus is a part, and prior to the closing of this offering, we
anticipate that we will effect the reverse stock split at a ratio
of 1 share for each 2.50 shares.
Except
as otherwise indicated and except in our financial statements, all
information regarding share amounts of common stock and prices per
share of common stock contained in this prospectus assume the
consummation of the reverse stock split to be effected following
effectiveness of the registration statement of which this
prospectus is a part, and prior to the closing of this
offering.
Recent
Financial Results
We have not yet
completed preparation of financial statements for the year ended
December 31, 2016, but based on preliminary data available to us,
we expect to report total revenue of approximately $515,000 for the
quarter and year ended December 31, 2016.
This preliminary
financial information is based on current expectations and is
subject to year-end closing adjustments. Actual results may differ.
This preliminary financial data has been prepared by and is the
responsibility of our management. RBSM LLP has not audited,
reviewed, compiled or performed any procedures with respect to this
preliminary financial data, and accordingly, RBSM LLP does not
express an opinion or any other form of assurance with respect
thereto. For a discussion of certain risks that may cause our
results of operations to differ from our expectations, see
“Risk Factors” elsewhere in this
prospectus.
Implications
of Being an Emerging Growth Company
We are
an “emerging growth company,” as defined in the
Jumpstart Our Business Startups Act of 2012, or the JOBS Act, and,
for as long as we continue to be an “emerging growth
company,” we may choose to take advantage of exemptions from
various reporting requirements applicable to other public companies
but not to “emerging growth companies,” including, but
not limited to, not being required to comply with the auditor
attestation requirements of Section 404 of the Sarbanes-Oxley Act
of 2002, reduced disclosure obligations regarding executive
compensation in our periodic reports and proxy statements, and
exemptions from the requirements of holding a nonbinding advisory
vote on executive compensation and stockholder approval of any
golden parachute payments not previously approved. We could be an
“emerging growth company” for up to five years, or
until the earliest of (i) the last day of the first fiscal year in
which our annual gross revenues exceed $1 billion, (ii) the date
that we become a “large accelerated filer” as defined
in Rule 12b-2 under the Securities Exchange Act of 1934, as
amended, which would occur if the market value of our common stock
that is held by non-affiliates exceeds $700 million as of the last
business day of our most recently completed second fiscal quarter,
or (iii) the date on which we have issued more than $1 billion in
non-convertible debt during the preceding three-year period. We are
choosing to “opt out” of the extended transition
periods available under the JOBS Act for complying with new or
revised accounting standards, but intend to take advantage of the
other exemptions discussed above.
|
4
|
We are
also currently considered a “smaller reporting
company,” which generally means that we have a public float
of less than $75 million and had annual revenues of less than $50
million during the most recently completed fiscal year. If we are
still considered a “smaller reporting company” at such
time as we cease to be an “emerging growth company,” we
will be subject to increased disclosure requirements. However, the
disclosure requirements will still be less than they would be if we
were not considered either an “emerging growth company”
or a “smaller reporting company.” Specifically, similar
to emerging growth companies”, “smaller reporting
companies” are able to provide simplified executive
compensation disclosures in their filings; are exempt from the
provisions of Section 404(b) of the Sarbanes-Oxley Act requiring
that independent registered public accounting firms provide an
attestation report on the effectiveness of internal control over
financial reporting; and have certain other decreased disclosure
obligations in their Securities and Exchange Commission filings,
including, among other things, being required to provide only two
years of audited financial statements in annual
reports.
Corporate
Information
We were
incorporated in Delaware in July 2007. Our corporate headquarters
is located at 3600 Green Court, Suite 350, Ann Arbor, Michigan
48105-1570. Our website can be accessed at
www.endrainc.com. The information contained on, or that
may be obtained from, our website is not, and shall not be deemed
to be, a part of this prospectus.
|
5
THE OFFERING
|
Units offered by us
|
2,000,000
units (or 2,300,000
units if the underwriters exercise their option to
purchase additional units in full), each
consiting of one share of common stock and a warrant to purchase
one share of common stock.
|
|
Warrants
|
Up to
2,000,000 shares of our common stock
issuable upon exercise of the warrants to be issued in this
offering (or up to
2,300,000 shares of our common stock if the
underwriters exercise their option to purchase additional
units in full). The
warrants will have an exercise price equal to 120% of the initial
public offering price per unit set forth on the cover
of this prospectus and will expire on the fifth anniversary of the
original issuance date. Each warrant will be
exercisable commencing upon separation of the unit of which it was
a part.
|
|
Separation of shares and warrants included in
the units offered hereby
|
The units
will begin trading on, or promptly after, the date of this
prospectus. The units will separate at the earlier of
(i) the first trading day following the 60th day after the date of
this prospectus, or (ii) such earlier date as may be determined by
Dougherty & Company LLC.
|
|
Common stock outstanding after this offering
|
4,443,213
shares (or 4,743,213 shares if
the underwriters exercise their option to purchase additional
units in full), and a total of
6,443,213 shares of our
common stock outstanding if the warrants issued in
this offering are exercised in full (or
7,043,213 shares if the
underwriters exercise their option to purchase additional
units in full and the warrants included in such
units are exercised).
(1)(2)
|
|
Over-allotment option
|
We
have granted the underwriters a 30-day option to purchase up to
300,000 additional units to cover over-allotments, if
any, provided that in no event may exercise of this option
occur after separation of the units
occurs.
|
|
Use of proceeds
|
We
intend to use the net proceeds from this offering to fund the
development, regulatory approval and the commercialization of our
Non-Alcoholic Fatty Liver Disease, or NAFLD, TAEUS application in
the European Union and the United States and for working capital
and other general corporate purposes. See “Use of
Proceeds” for additional information.
|
|
Risk factors
|
See
the section entitled “Risk Factors” and other
information included in this prospectus for a discussion of factors
you should carefully consider before deciding to invest in our
securities.
|
|
Proposed Nasdaq Capital Market symbol
|
We
have received approval for listing on the Nasdaq Capital Market our
common stock under the symbol “NDRA.”
We have applied to list our units and warrants on the Nasdaq
Capital Market under the symbols “NDRAU” and
“NDRAW,” respectively.
|
|
(1)
The number of
shares of our common stock to be outstanding after this offering is
based on 2,443,213 shares of common stock outstanding as of
September 30,
2016, assumes the conversion of the outstanding principal and
accrued interest on our outstanding convertible promissory notes at
September 30, 2016 into an aggregate of 1,430,490 shares of our
common stock at a conversion price of $1.00 per share immediately
prior to the completion of this offering, and excludes the
following:
●
213,937 shares of common stock issuable
upon the exercise of outstanding warrants, at a weighted average
exercise price of $13.53 per
share;
●
213,452 shares of
our common stock issuable upon the exercise of outstanding stock
options issued pursuant to our 2016 Omnibus Incentive Plan, or our
Incentive Plan, at a weighted average exercise price of
$7.15 per share and an
estimated 792,782 shares of our common stock issuable
upon the exercise of stock options expected to be granted to our
directors and certain of our officers upon the completion of this
offering at an exercise price equal to the public offering price
per unit set forth on the cover of this
prospectus;
●
an estimated 517,643 shares of
our common stock that will be reserved for future issuance under
our Incentive Plan;
●
up to
2,300,000 shares of our common stock that may be
issued under warrants to be issued to the public in
this offering; and
●
up to 184,000
shares of our common stock issuable upon exercise of the
underwriters' warrants.
(2) Except as
otherwise indicated herein, all information in this prospectus
assumes or gives effect to:
●
the reverse stock
split;
●
the conversion of
the outstanding principal and accrued interest on our outstanding
convertible promissory notes as of September 30, 2016 into an
aggregate of 1,430,490 shares of
our common stock at a conversion price of $1.00 per share
immediately prior to the completion of this
offering;
●
the
adoption of our Fourth Amended and Restated Certificate of
Incorporation and Amended and Restated Bylaws in connection with
the consummation of this offering; and
●
no
exercise of the underwriters’ over-allotment
option.
|
|
6
|
SUMMARY
SELECTED FINANCIAL DATA
The
following tables set forth a summary of our historical financial
data at, and for the period ended on, the dates indicated. We have
derived the statements of operations data for the years ended
December 31, 2015 and 2014 from our audited financial statements
included in this prospectus. We have derived the statements of
operations data for the nine month periods ended September 30, 2016
and 2015, and the balance sheet data as of September 30, 2016 from
our unaudited financial statements included in
this prospectus. The unaudited financial data includes, in the
opinion of our management, all adjustments, consisting of normal
recurring adjustments that are necessary for a fair presentation of
our financial position and results of operations for these periods.
Our historical results for any prior period are not necessarily
indicative of results to be expected in any other period or for the
year ended December 31, 2016. You should read this data together
with our financial statements and related notes appearing elsewhere
in this prospectus and the section of this prospectus entitled
“Management’s Discussion and Analysis of Financial
Condition and Results of Operations.”
Income Statement Data
|
||||
|
|
|
|
Nine
Months
|
Nine
Months
|
|
|
Year
Ended
|
Year
Ended
|
Ended
|
Ended
|
|
|
December
31,
2015
|
December
31,
2014
|
September
30,
2016
|
September
30,
2015
|
|
Revenue
|
$1,410,064
|
$559,355
|
$0
|
$1,155,065
|
|
Gross
Profit
|
$743,831
|
$300,210
|
$0
|
$615,256
|
|
Operating
Expenses
|
$2,312,402
|
$1,826,391
|
$1,474,878
|
$1,728,411
|
|
Other
Expenses
|
$(710,634)
|
$(654,697)
|
$(613,612)
|
$(707,316)
|
|
Net
Loss
|
$(2,279,204)
|
$(2,180,878)
|
$(2,088,491)
|
$(1,820,471)
|
|
Basic
and diluted net loss per common share
|
$ (0.98)
|
$ (1.35)
|
$ (0.82)
|
$ (0.81)
|
|
Weighted
average shares outstanding, basic and diluted
|
2,320,045
|
1,612,569
|
2,531,566
|
2,250,011
|
|
Pro
forma net loss per share, basic and diluted
(unaudited)(1)
|
$ (2.46)
|
$ (3.38)
|
$ (2.06)
|
$ (2.03)
|
|
Pro
forma weighted average shares outstanding
(unaudited)(1)
|
928,018
|
645,028
|
1,012,626
|
900,004
|
|
|
|
|
|
|
Balance Sheet Data
|
|||
|
|
As
of
|
As
of
|
As
of
|
|
|
December
31,
|
December
31,
|
September
30,
|
|
|
2015
|
2014
|
2016
|
|
Assets
|
|
|
|
|
Current
Assets
|
$ 126,618
|
$ 404,416
|
$ 362,196
|
|
Fixed
Assets, net
|
$ 360,104
|
$ 298,518
|
$ 311,492
|
|
Total
Assets
|
$ 486,722
|
$ 702,934
|
$ 673,688
|
|
Liabilities and
Stockholders' Equity
|
|
|
|
|
Current
Liabilities
|
$ 236,421
|
$ 97,601
|
$ 914,883
|
|
Stockholders'
Equity/(Deficit)
|
$ 250,300
|
$ 605,333
|
$ (241,196)
|
|
Total Liabilities
and Stockholders' Equity
|
$ 486,722
|
$ 702,934
|
$ 673,688
|
|
|
|
|
|
|
|
Statements of Cash Flows Data
|
||||
|
|
|
|
Nine Months
|
Nine Months
|
|
|
Year Ended
|
Year Ended
|
Ended
|
Ended
|
|
|
December 31,
2015
|
December 31,
2014
|
September 30,
2016
|
September 30,
2015
|
|
Cash Flows from
Operating Activities
|
$(842,727)
|
$(1,397,229)
|
$(1,385,033)
|
$(875,363)
|
|
Cash Flows from
Investing Activities
|
$(133,811)
|
$(275,371)
|
$--
|
$(133,811)
|
|
Cash Flows from
Financing Activities
|
$839,224
|
$1,751,015
|
$1,441,448
|
$955,617
|
|
Cash, end of
period
|
$19,128
|
$156,442
|
$75,543
|
$102,885
|
|
|
|
|
|
|
|
(1)
The number of weighted average common shares used in computing pro
forma net loss per share gives effect to the
contemplated reverse stock split at a ratio of 1 share
for each 2.5 shares to be effected following the effectiveness of
the registration statement of which this prospectus is a part, and
prior to the closing of this offering.
|
||||
|
|
|
|
|
|
7
An investment in our securities involves a high degree of risk. You
should carefully consider the following information about these
risks, together with the other information appearing elsewhere in
this prospectus, before deciding to invest in our
securities.
If any of the events described in the following risk factors
actually occurs, or if additional risks and uncertainties that are
not presently known to us or that we currently deem immaterial
later materialize, then our business, prospects, results of
operations and financial condition could be materially adversely
affected. In that event, the trading price of our
securities could decline, and you may lose all or part
of your investment in our securities. The risks
discussed below include forward-looking statements, and our actual
results may differ substantially from those discussed in these
forward-looking statements. See “Special Note Regarding
Forward-Looking Statements and Other Information Contained in this
Prospectus.”
Risks
Related to Our Business
We have a history of operating losses, and we may never achieve or
maintain profitability.
We have
a limited operating history upon which investors may evaluate our
prospects. We have only generated limited revenues to date and have
a history of losses from operations. As of September 30, 2016, we
had an accumulated deficit of approximately
$11,831,594. Even following the sale of
units in this offering, we may require additional
capital to complete the commercialization of our planned TAEUS
applications and to meet our growth and profitability targets. We
expect to expend significant resources on hiring of personnel,
payroll and benefits, continued scientific and potential product
research and development, potential product testing and
pre-clinical and clinical investigations, intellectual property
development and prosecution, marketing and promotion, capital
expenditures, working capital, general and administrative expenses,
and fees and expenses associated with this offering. We also expect
to incur costs and expenses related to consulting, laboratory
development, hiring of scientists and other operational personnel,
and expenses associated with the development of relationships with
strategic partners.
Our
Thermo Acoustic Enhanced Ultrasound, or TAEUS, technology is still
in development and we do not have any applications for our TAEUS
technology approved for sale. Applications for our TAEUS technology
may never be approved, generate significant revenue or become
commercially viable. Our ability to generate significant revenues
and, ultimately, achieve profitability will depend on whether we
can obtain additional capital when we need it, complete the
development of our technology, receive required regulatory
approvals for our TAEUS applications and find
customers who will purchase our future products or strategic
partners that will incorporate our technology into their products.
Even if we develop commercially viable applications for our TAEUS
technology, which may include licensing, we may never recover our
research and development expenses and we may never be able to
produce material revenues or operate on a profitable
basis.
Our efforts may never
result in the successful development of commercial
applications based on our TAEUS
technology.
Due to
the limited tissue penetration capability of light-based
thermoacoustic technology, we believe that there is a limited
clinical market for our current Nexus 128 product, which is focused
on laboratory specimen analysis. As a result, we are currently
focused on the development of products based on our TAEUS
technology. We have not yet completed the development of any
applications
based on such technology. Our research and development efforts
remain subject to all of the risks associated with the development
of new products
based on emerging technologies, including, without limitation,
unanticipated technical or other problems, the inability to develop
a product that may be sold at an acceptable price point and the
possible insufficiency of funds needed in order to complete
development of these products. Technical problems may result in
delays and cause us to incur additional expenses that would
increase our losses. If we cannot complete, or if we experience
significant delays in developing applications based on, our
TAEUS technology, particularly after incurring significant
expenditures, our business may fail.
8
Our success is substantially dependent on the success of
applications for our TAEUS platform.
To date
we have generated only limited sales of our existing Nexus 128
product and our ability to generate meaningful revenues in the
future will depend on the successful development and
commercialization of our TAEUS platform applications. The
commercial success of our TAEUS platform applications and our
ability to generate revenues will depend on many factors, including
the following:
●
our
successful development of applications for our TAEUS technology,
such as those we intend to pursue for the diagnosis of Non-Alcohol
Fatty Liver Disease, or NAFLD, and the monitoring of thermal
ablation surgery, and the acceptance in the marketplace by
physicians and patients of such applications;
●
the
successful design and manufacturing of a device or devices which
enable the use of our TAEUS technology by physicians on their
patients;
●
receipt
of necessary regulatory approvals;
●
sufficient
coverage or reimbursement by third-party payors;
●
our
ability to successfully market our products;
●
our ability to demonstrate that our TAEUS
applications have advantages over
competing products and procedures;
●
the
amount and nature of competition from competing or alternative
imaging products; and
●
our
ability to establish and maintain commercial manufacturing,
distribution and sales force capabilities.
We have limited data regarding the efficacy of our TAEUS platform
applications. If our applications do not perform in accordance with
our expectations, we are unlikely to successfully commercialize our
applications, even if they receive regulatory
approval.
Our
success depends in large part on the medical and third-party payor
community’s acceptance of our TAEUS applications. As a
result, even if we receive regulatory approval for our
applications, we believe that we will need to obtain additional
clinical data from users of our applications to persuade medical
professions to use our applications. We may conduct post-approval
clinical testing to obtain such additional data. Clinical testing
is expensive, can take a significant amount of time to complete and
can have uncertain outcomes. We have not yet conducted any clinical
studies and there can be no assurance that the results of any such
studies will be positive. Our failure to conduct successful
clinical studies could have a material, adverse impact on our
business.
Our limited operating history makes it difficult to evaluate our
business, predict our future results or forecast our financial
performance and growth.
We were
incorporated in 2007 and began commercializing our initial
pre-clinical Nexus 128 products in 2010. Our limited operating
history makes it difficult to evaluate our business, predict our
future results or forecast our financial performance and growth. If
our assumptions regarding the risks and uncertainties we face,
which we use to plan our business, are incorrect or change due to
circumstances in our business or our markets, or if we do not
address these risks successfully, our operating and financial
results could differ materially from our expectations and our
business could suffer.
9
We may not remain commercially viable if there is an inadequate
level of reimbursement by governmental programs and other
third-party payors.
Medical
imaging products are purchased principally by hospitals, physicians
and other healthcare providers around the world that typically bill
various third-party payors, including governmental programs
(e.g., Medicare and
Medicaid in the United States), private insurance plans and managed
care programs, for the services provided to their
patients.
Third-party payors
and governments may approve or deny coverage for certain
technologies and associated procedures based on independently
determined assessment criteria. Reimbursement decisions by payors
for these services are based on a wide range of methodologies that
may reflect the services' assessed resource costs, clinical
outcomes and economic value. These reimbursement methodologies and
decisions confer different, and sometimes conflicting, levels of
financial risk and incentives to healthcare providers and patients,
and these methodologies and decisions are subject to frequent
refinements. Third-party payors are also increasingly adjusting
reimbursement rates, often downwards, indirectly challenging the
prices charged for medical products and services. There can be no
assurance that our products will be covered by third-party payors,
that adequate reimbursement will be available or, even if payment
is available, that third-party payors' coverage policies will not
adversely affect our ability to sell our products
profitably.
We will depend on third parties to design, manufacture and seek
regulatory approval of our TAEUS applications. If any third
party fails to successfully design, manufacture and gain regulatory
approval of our TAEUS applications, our business
will be materially harmed.
We
currently intend to outsource the design and manufacturing of
applications utilizing our TAEUS technology rather than manufacture
our TAEUS applications ourselves. We will have limited control over
the efforts and resources that any third-party original equipment
manufacturer, or OEM, will devote to developing and manufacturing
our TAEUS applications. In addition, we currently expect to depend
on OEMs to acquire CE marks for the device or devices that they
develop and manufacture which are necessary to permit marketing of
those devices in the European Union.
An OEM
may not be able to successfully design and manufacture the products
it develops based on our TAEUS technology, may not devote
sufficient time and resources to support these efforts or may fail
in gaining the required regulatory approvals of our TAEUS
applications. The failure by an OEM in its efforts to design,
manufacture or gain regulatory approval of our TAEUS applications
could substantially harm the value of our TAEUS technology, brand
and business.
If we are unable to manage the anticipated growth of our business,
our future revenues and operating results may be
harmed.
Because
of our small size, growth in accordance with our business plan, if
achieved, will place a significant strain on our financial,
technical, operational and management resources. As we expand our
activities, there will be additional demands on these resources.
The failure to continue to upgrade our technical, administrative,
operating and financial control systems or the occurrence of
unexpected expansion difficulties, including issues relating to our
research and development activities and retention of experienced
scientists, managers and technicians, could have a material adverse
effect on our business, financial condition and results of
operations and our ability to timely execute our business plan. If
we are unable to implement these actions in a timely manner, our
results may be adversely affected.
10
Competition in the medical imaging market is intense and we may be
unable to successfully compete.
In
general, competition in the medical imaging market is very
significant and characterized by extensive research and development
and rapid technological change. Competitors in this market include
very large companies with significantly greater resources than we
have. To successfully compete in this market we will need to
develop TAEUS applications for particular applications that offer
significant advantages over alternative imaging products and
procedures for such applications.
Developments by
other medical imaging companies of new or improved products,
processes or technologies may make our products or proposed
products obsolete or less competitive. Alternative medical imaging
devices may be more accepted or cost-effective than our products.
Competition from these companies for employees with experience in
the medical imaging industry could result in higher turnover of our
employees. If we are unable to respond to these competitive
pressures, we could experience delayed or reduced market acceptance
of our products, higher expenses and lower revenue. If we are
unable to compete effectively with current or new entrants to these
markets, we will be unable to generate sufficient revenue to
maintain our business.
For
additional information regarding our competition, see the section
of this prospectus captioned
“Business-Competition.”
Changes in the healthcare industry could result in a reduction in
the size of the market for our products or may require us to
decrease the selling price for our products, either of which could
have a negative impact on our financial performance.
Trends
toward managed care, healthcare cost containment, and other changes
in government and private sector initiatives in Europe, the United
States and China are placing increased emphasis on lowering the
cost of medical services, which could adversely affect the demand
for or the prices of our products. For example:
●
major
third-party payors of hospital and non-hospital based healthcare
services could revise their payment methodologies and impose
stricter standards for reimbursement of imaging procedures charges
and/or a lower or more bundled reimbursement;
●
there
has been a consolidation among healthcare facilities and purchasers
of medical devices who prefer to limit the number of suppliers from
whom they purchase medical products, and these entities may decide
to stop purchasing our products or demand discounts on our
prices;
●
there
is economic pressure to contain healthcare costs in markets
throughout the world; and
●
there
are proposed and existing laws and regulations in international and
domestic markets regulating pricing and profitability of companies
in the healthcare industry.
These
trends could lead to pressure to reduce prices for our products and
could cause a decrease in the demand for our products in any given
market that could adversely affect our revenue and profitability,
which could harm our business.
We have limited selling, marketing and manufacturing resources,
which may restrict our success in commercializing our TAEUS
technology.
We
currently do not have a sales, marketing, customer support or
manufacturing team dedicated to our TAEUS clinical applications. To
grow our business as planned, we must expand our sales, marketing,
customer support and manufacturing capabilities. We must also
establish satisfactory arrangements for the manufacture and
distribution of our TAEUS applications, which will involve the
development of our commercial infrastructure and/or collaborative
commercial arrangements and partnerships. We may be unable to
attract, retain and manage the specialized workforce and
collaborative manufacturing and distribution arrangements necessary
to successfully commercialize our products. In addition, developing
these functions is time consuming and expensive. We intend to
partner with others to assist us with some or all of these
functions. However, we may be unable to find appropriate third
parties with whom to enter into these arrangements. Furthermore, if
we do enter into these arrangements, these third parties may not
perform as expected.
11
If we experience problems in our relationships with our
distributors, our ability to sell our products could be
limited.
Because
we are a small company with limited resources, we expect to depend
on distributors to help promote market acceptance and demand for
our products. Distributors that are in the business of selling
other medical products may not devote a sufficient level of
resources and support required to generate awareness of our
products and grow or maintain product sales. If our distributors
are unwilling or unable to market and sell our products, or if they
do not perform to our expectations, we could experience delayed or
reduced market acceptance and sales of our products. In addition,
disagreements with our distributors or non-performance by these
third parties could lead to costly and time-consuming litigation or
arbitration and disrupt distribution channels for a period of time
and require us to re-establish a distribution channel.
We have and may in the future form or seek additional strategic
alliances, collaborations or enter into licensing arrangements, and
we may not realize the benefits of such alliances, collaborations
or licensing arrangements.
In
April 2016, we entered into a Collaborative Research Agreement with
GE Healthcare under which GE Healthcare has agreed to support our
efforts to commercialize our TAEUS technology for use in an NAFLD
application by, among other things, providing equipment and
technical advice, and facilitating introductions to GE Healthcare
clinical ultrasound customers. This agreement does not commit GE
Healthcare to a long-term relationship and it may disengage with us
at any time.
We
may in the future form or seek additional strategic alliances,
create joint ventures or collaborations or enter into licensing
arrangements with third parties that we believe will complement or
augment our development and commercialization efforts with respect
to our technologies and applications.
Any
of these relationships may require us to incur non-recurring and
other charges, increase our near- and long-term expenditures, issue
securities that dilute our existing stockholders or disrupt our
management and business. In addition, we face significant
competition in seeking appropriate strategic partners and the
negotiation process is time-consuming and complex. If we license
technologies or applications, we may not be able to realize the
benefit of such transactions. Further, strategic alliances and
collaborations are subject to numerous risks, which may include the
following:
●
collaborators
have significant discretion in determining the efforts and
resources that they will apply to a collaboration;
●
collaborators
may not pursue development and commercialization of our
technologies and applications or may elect not to continue or renew
development or commercialization programs based on clinical trial
results, changes in their strategic focus due to the acquisition of
competitive products, availability of funding, or other external
factors, such as a business combination that diverts resources or
creates competing priorities;
●
collaborators
may delay clinical trials, provide insufficient funding for a
clinical trial, stop a clinical trial, abandon the development of
an application, repeat or conduct new clinical trials, or require a
new formulation of an application for clinical
testing;
●
collaborators
could independently develop, or develop with third parties,
products that compete directly or indirectly with our applications
and technologies;
●
a
collaborator with marketing and distribution rights to one or more
applications may not commit sufficient resources to their marketing
and distribution;
●
collaborators
may not properly maintain or defend our intellectual property
rights or may use our intellectual property or proprietary
information in a way that gives rise to actual or threatened
litigation that could jeopardize or invalidate our intellectual
property or proprietary information or expose us to potential
liability;
●
disputes
may arise between us and a collaborator that cause the delay or
termination of the research, development or commercialization of
our technologies and applications, or that result in costly
litigation or arbitration that diverts management attention and
resources;
●
collaborations
may be terminated and, if terminated, may result in a need for
additional capital to pursue further development or
commercialization of the applicable applications or technologies;
and
●
collaborators
may own or co-own intellectual property covering our products that
results from our collaborating with them, and in such cases, we
would not have the exclusive right to commercialize such
intellectual property.
As
a result, if we enter into collaboration agreements and strategic
partnerships or license our applications or technologies, we may
not be able to realize the benefit of such transactions if we are
unable to successfully integrate them with our existing operations
and company culture, which could delay our timelines or otherwise
adversely affect our business. We also cannot be certain that,
following a strategic transaction or license, we will achieve the
revenue or specific net income that justifies such transaction. Any
delays in entering into new strategic partnership agreements
related to our applications could delay the development and
commercialization of our technologies and applications in certain
geographies or for certain applications, which would harm our
business prospects, financial condition and results of
operations.
We intend to market our TAEUS applications, if approved, globally,
in which case we will be subject to the risks of doing business
outside of the United States.
Because
we intend to market our TAEUS applications, if approved, globally,
our business may be subject to risks associated with doing business
globally. Accordingly, our business and financial results in the
future could be adversely affected due to a variety of factors,
including:
●
changes
in a specific country’s or region’s political and
cultural climate or economic condition;
●
unexpected
changes in laws and regulatory requirements in local
jurisdictions;
●
difficulty
of effective enforcement of contractual provisions in local
jurisdictions;
●
inadequate
intellectual property protection in certain countries;
●
trade-protection
measures, import or export licensing requirements such as Export
Administration Regulations promulgated by the United States
Department of Commerce and fines, penalties or suspension or
revocation of export privileges;
●
effects
of applicable local tax structures and potentially adverse tax
consequences; and
●
significant
adverse changes in currency exchange rates.
We depend on our senior management team and the loss of one or more
key employees or an inability to attract and retain highly skilled
employees could harm our business.
Our
success largely depends upon the continued services of our
executive management team and key employees and the loss of one or
more of our executive officers or key employees could harm us and
directly impact our financial results. Our employees may terminate
their employment with us at any time. Our executive management team
has significant experience and knowledge of medical devices and
ultrasound systems, and the loss of any team member could impair
our ability to design, identify, and develop new intellectual
property and new scientific or product ideas. Additionally, if we
lose the services of any of these persons, we would likely be
forced to expend significant time and money in the pursuit of
replacements, which may result in a delay in the implementation of
our business plan and plan of operations. We can give no assurance
that we could find satisfactory replacements for these individuals
on terms that would not be unduly expensive or burdensome to
us.
To
execute our growth plan, we must attract and retain highly
qualified personnel. Competition for skilled personnel is intense,
especially for engineers with high levels of experience in
designing and developing medical devices and for sales executives,
especially with industry expertise. We may experience difficulty in
hiring and retaining employees with appropriate qualifications.
Many of the companies with which we compete for experienced
personnel have greater resources than we have. In addition, we
invest significant time and expense in training our employees,
which increases their value to competitors who may seek to recruit
them. If we fail to attract new personnel or fail to retain and
motivate our current personnel, our business and future growth
prospects would be harmed.
12
Our employees, independent contractors, consultants, commercial
partners and vendors may engage in misconduct or other improper
activities, including noncompliance with regulatory standards and
requirements.
We are
exposed to the risk of fraud, misconduct or other illegal activity
by our employees, independent contractors, consultants, commercial
partners and vendors. Misconduct by these parties could include
intentional, reckless and negligent conduct that fails to: comply
with the U.S. Food, Drug and Cosmetics Act, or the FD&C Act,
and similar laws of other countries, and rules and regulations of
the U.S. Food and Drug Administration, or the FDA, and other
similar foreign regulatory bodies; provide true, complete and
accurate information to the FDA and other similar foreign
regulatory bodies; comply with manufacturing standards we
establish; comply with healthcare fraud and abuse laws in the
United States and similar foreign fraudulent misconduct laws; or
report financial information or data accurately or to disclose
unauthorized activities to us. If we obtain European, Chinese or
FDA approval of any of our products and begin commercializing those
products in Europe, China or the United States, respectively, our
potential exposure under such laws will increase significantly, and
our costs associated with compliance with such laws are also likely
to increase. In particular, the promotion, sales and marketing of
healthcare items and services, as well as certain business
arrangements in the healthcare industry, are subject to extensive
laws designed to prevent fraud, kickbacks, self-dealing and other
abusive practices. These laws and regulations may restrict or
prohibit a wide range of pricing, discounting, marketing and
promotion, structuring and commissions, certain customer incentive
programs and other business arrangements generally. It is not
always possible to identify and deter misconduct by employees and
other parties, and the precautions we take to detect and prevent
this activity may not be effective in controlling unknown or
unmanaged risks or losses or in protecting us from governmental
investigations or other actions or lawsuits stemming from a failure
to comply with these laws or regulations. If any such actions are
instituted against us, and we are not successful in defending
ourselves or asserting our rights, those actions could have a
significant impact on our business, including the imposition of
significant fines or other sanctions.
Misdiagnosis, warranty and other claims initiated against us and
product field actions could increase our costs, delay or reduce our
sales and damage our reputation, adversely affecting our financial
condition.
Our
business exposes us to the risk of malpractice, warranty or product
liability claims inherent in the sale and support of medical device
products, including those based on claims that the use or failure
of one of our products resulted in a misdiagnosis or harm to a
patient. Such claims may cause financial loss, damage our
reputation by raising questions about our products’ safety
and efficacy, adversely affect regulatory approvals and interfere
with our efforts to market our products. Although to date we have
not been involved in any medical malpractice or product liability
litigation, we may incur significant liability if such litigation
were to occur. We may also face adverse publicity resulting from
product field actions or regulatory proceedings brought against us.
Claims could also be asserted under state consumer protection acts.
If we cannot successfully defend ourselves against product
liability or related claims, we may incur substantial liabilities
or be required to limit distribution of our products. Even a
successful defense would require significant financial and
management resources. Regardless of the merits or eventual outcome,
liability claims may result in:
●
decreased
demand for our products;
●
injury
to our reputation and negative media attention;
●
initiation
of investigations by regulators;
13
●
costs
to defend the related litigation;
●
a
diversion of management’s time and our
resources;
●
substantial
monetary awards to trial participants or
patients;
●
product
recalls, withdrawals or labeling, marketing or promotional
restrictions;
●
loss
of revenue;
●
exhaustion
of any available insurance and our capital
resources;
●
the
inability to commercialize a product at all or for particular
applications; and
●
a
decline in the price of our securities.
Although we
currently maintain liability insurance in amounts we believe are
commercially reasonable, any liability we incur may exceed our
insurance coverage. Our insurance policies may also have various
exclusions, and we may be subject to a claim for which we have no
coverage. Liability insurance is expensive and may cease to be
available on acceptable terms, if at all. A malpractice, warranty,
product liability or other claim or product field action not
covered by our insurance or exceeding our coverage could
significantly impair our financial condition. In addition, a
product field action or a liability claim against us could
significantly harm our reputation and make it more difficult to
obtain the funding and commercial relationships necessary to
maintain our business.
Our internal computer systems, or those used by third-party
manufacturers or other contractors or consultants, may fail or
suffer security breaches.
Despite
the implementation of security measures, our internal computer
systems and those of our future manufacturers and other contractors
and consultants are vulnerable to damage from computer viruses and
unauthorized access. Although to our knowledge we have not
experienced any such material system failure or security breach to
date, if such an event were to occur and cause interruptions in our
operations, it could result in a material disruption of our
development programs and our business operations. To the extent
that any disruption or security breach were to result in a loss of,
or damage to, our data or applications, or inappropriate disclosure
of confidential or proprietary information, we could incur
liability and the further development and commercialization of our
products could be delayed.
Unfavorable economic conditions may have an adverse impact on our
business.
Unfavorable changes
in economic conditions, including inflation, recession, or other
changes in economic conditions, may result in lower consumer
healthcare spending as well as physician and hospital spending and
availability of credit. If demand for medical devices or budgets
for capital improvements decline, our revenue could be adversely
affected. Additionally, if our suppliers face challenges in
obtaining credit, in selling their products or otherwise in
operating their businesses, they may become unable to continue to
offer the materials we use to manufacture our products, which could
result in sales disruption.
We may
face significant challenges if global economic conditions remain
unstable or worsen, including reduced demand for our products and
services, increased order cancellations, longer sales cycles and
slower adoption of new technologies; increased difficulty in
collecting accounts receivable and increased risk of excess and
obsolete inventories; increased price competition in our served
markets; increased prices in components as a result of higher
commodities prices; supply chain interruptions, which could disrupt
our ability to produce our products; and increased risk of
impairment of investments, goodwill and intangible and long-lived
assets.
14
The United Kingdom’s vote to leave the European Union will
have uncertain effects and could adversely affect us.
The
United Kingdom held a referendum on June 23, 2016 in which a
majority of voters voted to exit the European Union, commonly
referred to as “Brexit”. Negotiations are expected to
commence to determine the future terms of the United
Kingdom’s relationship with the European Union, including,
among other things, the terms of trade between the United Kingdom
and the European Union. The effects of Brexit will depend on any
agreements the United Kingdom makes to retain access to E.U.
markets either during a transitional period or more permanently.
Brexit could adversely affect European and worldwide economic and
market conditions and could contribute to instability in global
financial and foreign exchange markets, including volatility in the
value of the Sterling and Euro. In addition, Brexit could lead to
legal uncertainty and potentially divergent national laws and
regulations as the United Kingdom determines which E.U. laws to
replace or replicate. Any of these effects of Brexit, and others we
cannot anticipate, could adversely affect our business, results of
operations, financial condition and cash flows.
Risks
Related to Intellectual Property and Other Legal
Matters
If we are unable to protect our intellectual property, then our
financial condition, results of operations and the value of our
technology and products could be adversely affected.
Much of
our value arises out of our proprietary technology and intellectual
property for the design, manufacture and use of medical imaging
systems, including development of our TAEUS applications. We rely
on patent, copyright, trade secret and trademark laws to protect
our proprietary technology and limit the ability of others to
compete with us using the same or similar technology. Third parties
may infringe or misappropriate our intellectual property, which
could harm our business.
We
currently maintain a patent portfolio consisting of two granted
patents and thirteen pending patent applications in the United
States and foreign jurisdictions relating to our technology. In
addition, we currently license eight granted patents and three
pending patent applications in the United States and foreign
jurisdictions. We or our licensor may fail to maintain these
patents, may determine not to pursue litigation against entities
that are infringing upon these patents, or may pursue such
enforcement less aggressively than we ordinarily
would.
Policing
unauthorized use of our intellectual property is difficult, costly
and time-intensive. We may fail to stop or prevent misappropriation
of our technology, particularly in countries where the laws may not
protect our proprietary rights to the same extent as do the laws of
the United States. Proceedings to enforce our patent and other
intellectual property rights in non-U.S. jurisdictions could result
in substantial costs and divert our efforts and attention from
other aspects of our business. If we cannot prevent other companies
from using our proprietary technology or if our patents are found
invalid or otherwise unenforceable, we may be unable to compete
effectively against other manufacturers of ultrasound systems,
which could decrease our market share. In addition, the breach of a
patent licensing agreement by us may result in termination of a
patent license.
If we are unable to protect the confidentiality of our proprietary
information and know-how, the value of our technology and products
could be adversely affected.
In
addition to our patent activities, we rely upon, among other
things, unpatented proprietary technology, processes, trade secrets
and know-how. Any involuntary disclosure to or misappropriation by
third parties of our confidential or proprietary information could
enable competitors to duplicate or surpass our technological
achievements, potentially eroding our competitive position in our
market. We seek to protect confidential or proprietary information
in part by confidentiality agreements with our employees,
consultants and third parties. While we require all of our
employees, consultants, advisors and any third parties who have
access to our proprietary know-how, information and technology to
enter into confidentiality agreements, we cannot be certain that
this know-how, information and technology will not be disclosed or
that competitors will not otherwise gain access to our trade
secrets or independently develop substantially equivalent
information and techniques. These agreements may be terminated or
breached, and we may not have adequate remedies for any such
termination or breach. Furthermore, these agreements may not
provide meaningful protection for our trade secrets and know-how in
the event of unauthorized use or disclosure. To the extent that any
of our staff was previously employed by other pharmaceutical,
medical technology or biotechnology companies, those employers may
allege violations of trade secrets and other similar claims in
relation to their former employee’s therapeutic development
activities for us.
15
We may in the future be a party to intellectual property litigation
or administrative proceedings that could be costly and could
interfere with our ability to sell our TAEUS
applications.
The
medical device industry has been characterized by extensive
litigation regarding patents, trademarks, trade secrets, and other
intellectual property rights, and companies in the industry have
used intellectual property litigation to gain a competitive
advantage. It is possible that U.S. and foreign patents and pending
patent applications or trademarks controlled by third parties may
be alleged to cover our products, or that we may be accused of
misappropriating third parties’ trade secrets. Other medical
imaging market participants, many of which have substantially
greater resources and have made substantial investments in patent
portfolios, trade secrets, trademarks, and competing technologies,
may have applied for or obtained or may in the future apply for or
obtain, patents or trademarks that will prevent, limit or otherwise
interfere with our ability to make, use, sell and/or export our
products or to use product names. We may become a party to patent
or trademark infringement or trade secret claims and litigation as
a result of these and other third party intellectual property
rights being asserted against us. The defense and prosecution of
these matters are both costly and time consuming. Vendors from whom
we purchase hardware or software may not indemnify us in the event
that such hardware or software is accused of infringing a third
party's patent or trademark or of misappropriating a third
party’s trade secret.
Further, if such
patents, trademarks, or trade secrets are successfully asserted
against us, this may harm our business and result in injunctions
preventing us from selling our products, license fees, damages and
the payment of attorney fees and court costs. In addition, if we
are found to willfully infringe third-party patents or trademarks
or to have misappropriated trade secrets, we could be required to
pay treble damages in addition to other penalties. Although patent,
trademark, trade secret, and other intellectual property disputes
in the medical device area have often been settled through
licensing or similar arrangements, costs associated with such
arrangements may be substantial and could include ongoing
royalties. We may be unable to obtain necessary licenses on
satisfactory terms, if at all. If we do not obtain necessary
licenses, we may not be able to redesign our TAEUS applications to
avoid infringement.
Similarly,
interference or derivation proceedings provoked by third parties or
brought by the U.S. Patent and Trademark Office, or USPTO, may be
necessary to determine the priority of inventions or other matters
of inventorship with respect to our patents or patent applications.
We may also become involved in other proceedings, such as
re-examination, inter partes review, or opposition proceedings,
before the USPTO or other jurisdictional body relating to our
intellectual property rights or the intellectual property rights of
others. Adverse determinations in a judicial or administrative
proceeding or failure to obtain necessary licenses could prevent us
from manufacturing and selling our TAEUS applications or using
product names, which would have a significant adverse impact on our
business.
Additionally, we
may need to commence proceedings against others to enforce our
patents or trademarks, to protect our trade secrets or know-how, or
to determine the enforceability, scope and validity of the
proprietary rights of others. These proceedings would result in
substantial expense to us and significant diversion of effort by
our technical and management personnel. We may not prevail in any
lawsuits that we initiate and the damages or other remedies
awarded, if any, may not be commercially meaningful. We may not be
able to stop a competitor from marketing and selling products that
are the same or similar to our products or from using product names
that are the same or similar to our product names, and our business
may be harmed as a result.
16
Intellectual
property rights may not provide adequate protection, which may
permit third parties to compete against us more
effectively.
In
order to remain competitive, we must develop and maintain
protection of the proprietary aspects of our technologies. We rely
on a combination of patents, copyrights, trademarks, trade secret
laws and confidentiality and invention assignment agreements to
protect our intellectual property rights. Any patents issued to us
may be challenged by third parties as being invalid, or third
parties may independently develop similar or competing technology
that avoids our patents. Should such challenges be successful,
competitors might be able to market products and use manufacturing
processes that are substantially similar to ours. We may not be
able to prevent the unauthorized disclosure or use of our technical
knowledge or other trade secrets by consultants, vendors or former
or current employees, despite the existence generally of
confidentiality agreements and other contractual restrictions.
Monitoring unauthorized use and disclosure of our intellectual
property is difficult, and we do not know whether the steps we have
taken to protect our intellectual property will be adequate. In
addition, the laws of many foreign countries will not protect our
intellectual property rights to the same extent as the laws of the
United States. Consequently, we may be unable to prevent our
proprietary technology from being exploited abroad, which could
affect our ability to expand to international markets or require
costly efforts to protect our technology. To the extent our
intellectual property protection is incomplete, we are exposed to a
greater risk of direct competition. In addition, competitors could
purchase our products and attempt to replicate some or all of the
competitive advantages we derive from our development efforts or
design around our protected technology. Our failure to secure,
protect and enforce our intellectual property rights could
substantially harm the value of our TAEUS platform, brand and
business.
Risks
Related to Government Regulation
Failure to comply with laws and regulations could harm our
business.
Our
business is or in the future may be subject to regulation by
various federal, state, local and foreign governmental agencies,
including agencies responsible for monitoring and enforcing
employment and labor laws, workplace safety, environmental laws,
consumer protection laws, anti-bribery laws, import/export
controls, securities laws and tax laws and regulations. In certain
jurisdictions, these regulatory requirements may be more stringent
than those in the United States. Noncompliance with applicable
regulations or requirements could subject us to investigations,
sanctions, mandatory recalls, enforcement actions, adverse
publicity, disgorgement of profits, fines, damages, civil and
criminal penalties or injunctions and administrative actions. If
any governmental sanctions, fines or penalties are imposed, or if
we do not prevail in any possible civil or criminal litigation, our
business, operating results and financial condition could be
harmed. In addition, responding to any action will likely result in
a significant diversion of management's attention and our resources
and substantial costs. Enforcement actions and sanctions could
further harm our business, operating results and financial
condition.
17
If we fail to obtain and maintain necessary regulatory clearances
or approvals for our TAEUS applications, or if clearances or
approvals for future applications and indications are delayed or
not issued, our commercial operations will be harmed.
The
medical devices that we manufacture and market are subject to
regulation by numerous worldwide regulatory bodies, including the
FDA and comparable international regulatory agencies. These
agencies require manufacturers of medical devices to comply with
applicable laws and regulations governing development, testing,
manufacturing, labeling, marketing and distribution of medical
devices. Devices are generally subject to varying levels of
regulatory control, based on the risk level of the device.
Governmental regulations specific to medical devices are
wide-ranging and govern, among other things:
●
product
design, development and manufacture;
●
laboratory,
pre-clinical and clinical testing, labeling, packaging storage and
distribution;
●
premarketing
clearance or approval;
●
record
keeping;
●
product
marketing, promotion and advertising, sales and distribution;
and
●
post-marketing
surveillance, including reporting of deaths or serious injuries and
recalls and correction and removals.
In the
European Union, we will be required to comply with applicable
medical device directives (including the Medical Devices Directive
and the Active Implantable Medical Devices Directive) and obtain CE
mark certification in order to market medical devices. The CE mark
is applied following approval from an independent notified body or
declaration of conformity. It is an international symbol of
adherence to quality assurance standards and compliance with
applicable European Medical Devices Directives. We believe that our
TAEUS applications will qualify for sale in the European Union as
Class IIa medical devices. Existing regulations do not
require clinical trials to obtain CE marks for Class IIa medical
devices. However, in 2012 the European Commission proposed a new
regulatory scheme that, if implemented, will impose significant
additional obligations on medical device companies. Expected
changes include stricter requirements for clinical evidence and
pre-market assessment of safety and performance, new
classifications to indicate risk levels, requirements for third
party testing by government accredited groups for some types of
medical devices, and tightened and streamlined quality management
system assessment procedures. It is anticipated that this new
regulatory scheme may be implemented prior to receipt of the CE
mark for our NAFLD TAEUS application but we believe that applicable
transition rules should allow us to avoid their application in that
case. However, such new rules could impose additional requirements,
such as a requirement to conduct clinical trials, on future CE mark
applications we make.
We are
also required to comply with the regulations of each other country
where we commercialize products, such as the requirement that we
obtain approval from the FDA and the China Food and Drug
Administration before we can launch new products in the United
States and China, respectively.
International sales
of medical devices manufactured in the United States that are not
approved by the FDA for use in the United States, or that are
banned or deviate from lawful performance standards, are subject to
FDA export requirements. Exported devices are subject to the
regulatory requirements of each country to which the device is
exported. Frequently, regulatory approval may first be obtained in
a foreign country prior to application in the United States due to
differing regulatory requirements; however, other countries, such
as China for example, require approval in the country of origin
first.
Before
a new medical device or a new intended use for an existing product
can be marketed in the United States, a company must first submit
and receive either 510(k) clearance or premarketing approval, or
PMA, from the FDA, unless an exemption applies. The typical
duration to receive a 510(k) approval is approximately nine to
twelve months from the date of the initial 510(k) submission and
the typical duration to receive a PMA approval is approximately two
years from the date of submission of the initial PMA application,
although there is no guarantee that the timing will not be
longer.
We expect all of
our products to be classified as Class II medical devices that may
be approved by means of a 510(k) clearance. In the past, the 510(k)
pathway for product marketing has required only proof of
substantial equivalence in technology for a given indication with a
previously cleared device. Recently, there has been a trend of the
FDA requiring additional clinical work to prove efficacy in
addition to technological equivalence and basic safety. Whether
clinical data is provided or not, the FDA may decide to reject the
substantial equivalence argument we present. If that happens, the
device is automatically designated as a Class III device. The
device sponsor must then fulfill more rigorous PMA requirements, or
can request a risk-based classification determination for the
device in accordance with the “de novo” process, which
may determine that the new device is of low to moderate risk and
that it can be appropriately be regulated as a Class I or II
device. Thus, although at this time we do not anticipate that we
will be required to do so, it is possible that one or more of our
other products may require approval through the 510(K) de novo
process or by means of a PMA.
We may not be able
to obtain the necessary clearances or approvals or may be unduly
delayed in doing so, which could harm our business. Furthermore,
even if we are granted regulatory clearances or approvals, they may
include significant limitations on the indicated uses for the
product, which may limit the market for the product. Therefore,
even if we believe we have successfully developed our TAEUS
technology, we may not be permitted to market TAEUS applications in
the United States if we do not obtain FDA regulatory clearance to
market such applications. Delays in obtaining clearance or approval
could increase our costs and harm our revenues and
growth.
18
In
addition, we are required to timely file various reports with the
FDA, including reports required by the medical device reporting
regulations that require us to report to certain regulatory
authorities if our devices may have caused or contributed to a
death or serious injury or malfunctioned in a way that would likely
cause or contribute to a death or serious injury if the malfunction
were to recur. If these reports are not filed timely, regulators
may impose sanctions and sales of our products may suffer, and we
may be subject to product liability or regulatory enforcement
actions, all of which could harm our business.
If we
initiate a correction or removal for one of our devices to reduce a
risk to health posed by the device, we would be required to submit
a publically available Correction and Removal report to the FDA
and, in many cases, similar reports to other regulatory agencies.
This report could be classified by the FDA as a device recall which
could lead to increased scrutiny by the FDA, other international
regulatory agencies and our customers regarding the quality and
safety of our devices. Furthermore, the submission of these reports
has been and could be used by competitors against us in competitive
situations and cause customers to delay purchase decisions or
cancel orders and would harm our reputation.
The FDA
and the Federal Trade Commission, or FTC, also regulate the
advertising and promotion of our products to ensure that the claims
we make are consistent with our regulatory clearances, that there
are adequate and reasonable data to substantiate the claims and
that our promotional labeling and advertising is neither false nor
misleading in any respect. If the FDA or FTC determines that any of
our advertising or promotional claims are misleading, not
substantiated or not permissible, we may be subject to enforcement
actions, including warning letters, and we may be required to
revise our promotional claims and make other corrections or
restitutions.
The FDA
and state authorities have broad enforcement powers. Our failure to
comply with applicable regulatory requirements could result in
enforcement action by the FDA or state agencies, which may include
any of the following sanctions:
●
adverse
publicity, warning letters, fines, injunctions, consent decrees and
civil penalties;
●
repair,
replacement, refunds, recall or seizure of our
products;
●
operating
restrictions, partial suspension or total shutdown of
production;
●
refusing
our requests for 510(k) clearance or premarket approval of new
products, new intended uses or modifications to existing
products;
●
withdrawing
510(k) clearance or premarket approvals that have already been
granted; and
●
criminal
prosecution.
If any
of these events were to occur, our business and financial condition
would be harmed.
Our TAEUS applications may require recertification or new
regulatory clearances or premarket approvals and we may be required
to recall or cease marketing our TAEUS applications until such
recertification or clearances are obtained.
Most
countries outside of the United States require that product
approvals be recertified on a regular basis, generally every five
years. The recertification process requires that we evaluate any
device changes and any new regulations or standards relevant to the
device and, where needed, conduct appropriate testing to document
continued compliance. Where recertification applications are
required, they must be approved in order to continue selling our
products in those countries.
19
In the
United States, material modifications to the intended use or
technological characteristics of our TAEUS applications will
require new 510(k) clearances or premarket approvals or require us
to recall or cease marketing the modified devices until these
clearances or approvals are obtained. Based on FDA published
guidelines, the FDA requires device manufacturers to initially make
and document a determination of whether or not a modification
requires a new approval, supplement or clearance; however, the FDA
can review a manufacturer’s decision. Any modification to an
FDA-cleared device that would significantly affect its safety or
efficacy or that would constitute a major change in its intended
use would require a new 510(k) clearance or possibly a premarket
approval.
We may
not be able to obtain recertification or additional 510(k)
clearances or premarket approvals for our applications or for
modifications to, or additional indications for, our TAEUS
technology in a timely fashion, or at all. Delays in obtaining
required future governmental approvals would harm our ability to
introduce new or enhanced products in a timely manner, which in
turn would harm our future growth. If foreign regulatory
authorities or the FDA require additional approvals, we may be
required to recall and to stop selling or marketing our TAEUS
applications, which could harm our operating results and require us
to redesign our applications. In these circumstances, we may be
subject to significant enforcement actions.
If we or our suppliers fail to comply with the FDA’s Quality
System Regulations or other regulatory bodies’ equivalent
regulations, our manufacturing operations could be delayed or shut
down and TAEUS platform sales could suffer.
Our
manufacturing processes and those of our third party suppliers will
be required to comply with the FDA’s Quality System
Regulations and other regulatory bodies’ equivalent
regulations, which cover the procedures and documentation of the
design, testing, production, control, quality assurance, labeling,
packaging, storage and shipping of our TAEUS applications. We will
also be subject to similar state requirements and licenses. In
addition, we must engage in extensive recordkeeping and reporting
and must make available our manufacturing facilities and records
for periodic unannounced inspections by governmental agencies,
including the FDA, state authorities and comparable agencies in
other countries. If we fail such an inspection, our operations
could be disrupted and our manufacturing interrupted. Failure to
take adequate corrective action in response to an adverse
inspection could result in, among other things, a shut-down of our
manufacturing operations, significant fines, suspension of
marketing clearances and approvals, seizures or recalls of our
products, operating restrictions and criminal prosecutions, any of
which would cause our business to suffer. Furthermore, our key
component suppliers may not currently be or may not continue to be
in compliance with applicable regulatory requirements, which may
result in manufacturing delays for our product and cause our
results of operations to suffer.
Our TAEUS applications may in the future be subject to product
recalls that could harm our reputation.
Governmental
authorities in Europe, the United States and China have the
authority to require the recall of commercialized products in the
event of material regulatory deficiencies or defects in design or
manufacture. A government-mandated or voluntary recall by us could
occur as a result of component failures, manufacturing errors or
design or labeling defects. Recalls of our TAEUS applications would
divert managerial attention, be expensive, harm our reputation with
customers and harm our financial condition and results of
operations. A recall announcement would negatively affect the
price of our securities.
Healthcare reform measures could hinder or prevent our planned
products' commercial success.
There
have been, and we expect there will continue to be, a number of
legislative and regulatory changes to the healthcare system in ways
that could harm our future revenues and profitability and the
future revenues and profitability of our potential customers. In
the European Union, although there have not been any recent
amendments to the relevant regulatory legislation, there are
ongoing discussions regarding amending the current regulatory
framework for medical devices. Moreover, because the Medical
Devices Directive requires only minimum harmonization in the
European Union, member countries may alter their enforcement of the
directives or amend their national regulatory rules. We cannot
predict what healthcare initiatives, if any, will be implemented by
the European Union or E.U. member countries, or the effect any
future legislation or regulation will have on us.
20
In the
United States, federal and state lawmakers regularly propose and,
at times, enact legislation that would result in significant
changes to the healthcare system, some of which are intended to
contain or reduce the costs of medical products and services. For
example, one of the most significant healthcare reform measures in
decades, the Patient Protection and Affordable Care Act, as amended
by the Health Care and Education Affordability Reconciliation Act,
or Affordable Care Act, was enacted in 2010. The Affordable Care
Act contains a number of provisions, including those governing
enrollment in federal healthcare programs, reimbursement changes
and fraud and abuse measures, all of which will impact existing
government healthcare programs and will result in the development
of new programs. The Affordable Care Act, among other things,
imposes an excise tax of 2.3% on the sale of most medical devices,
including ours, and any failure to pay this amount could result in
the imposition of an injunction on the sale of our products, fines
and penalties.
It
remains unclear whether changes will be made to the Affordable Care
Act, or whether it will be repealed or materially modified. We
cannot assure you that the Affordable Care Act, as currently
enacted or as amended or discontinued in the future, will not harm
our business and financial results and we cannot predict how future
federal or state legislative or administrative changes relating to
healthcare reform will affect our business.
There
likely will continue to be legislative and regulatory proposals at
the federal and state levels directed at containing or lowering the
cost of healthcare. We cannot predict the initiatives that may be
adopted in the future or their full impact. The continuing efforts
of the government, insurance companies, managed care organizations
and other payors of healthcare services to contain or reduce costs
of healthcare may harm:
●
our
ability to set a price that we believe is fair for our
products;
●
our
ability to generate revenues and achieve or maintain profitability;
and
●
the
availability of capital.
If we fail to comply with healthcare regulations, we could face
substantial penalties and our business, operations and financial
condition could be adversely affected.
Even
though we do not and will not control referrals of healthcare
services or bill directly to Medicare, Medicaid or other third
party payors, certain federal and state healthcare laws and
regulations pertaining to fraud and abuse and patients’
rights are and will be applicable to our business. We could be
subject to healthcare fraud and abuse and patient privacy
regulation by both the federal government and the states in which
we conduct our business. Other jurisdictions such as the European
Union have similar laws. The regulations that will affect how we
operate include:
●
the
federal healthcare program Anti-Kickback Statute, which prohibits,
among other things, any person from knowingly and willfully
offering, soliciting, receiving or providing remuneration, directly
or indirectly, in exchange for or to induce either the referral of
an individual for, or the purchase, order or recommendation of, any
good or service for which payment may be made under federal
healthcare programs, such as the Medicare and Medicaid
programs;
21
●
the
federal False Claims Act, which prohibits, among other things,
individuals or entities from knowingly presenting, or causing to be
presented, false claims, or knowingly using false statements, to
obtain payment from the federal government;
●
federal
criminal laws that prohibit executing a scheme to defraud any
healthcare benefit program or making false statements relating to
healthcare matters;
●
the
federal Physician Payment Sunshine Act, created under the
Affordable Care Act, and its implementing regulations, which
require manufacturers of drugs, medical devices, biologicals and
medical supplies for which payment is available under Medicare,
Medicaid, or the Children's Health Insurance Program to report
annually to the U.S. Department of Health and Human Services, or
HHS, information related to payments or other transfers of value
made to physicians and teaching hospitals, as well as ownership and
investment interests held by physicians and their immediate family
members;
●
HIPAA,
as amended by the Health Information Technology for Economic and
Clinical Health Act, which governs the conduct of certain
electronic healthcare transactions and protects the security and
privacy of protected health information; and
●
state
law equivalents of each of the above federal laws, such as
anti-kickback and false claims laws which may apply to items or
services reimbursed by any third-party payor, including commercial
insurers.
The
Affordable Care Act, among other things, amends the intent
requirement of the Federal Anti-Kickback Statute and criminal
healthcare fraud statutes. A person or entity no longer needs to
have actual knowledge of this statute or specific intent to violate
it. In addition, the Affordable Care Act provides that the
government may assert that a claim including items or services
resulting from a violation of the Federal Anti-Kickback Statute
constitutes a false or fraudulent claim for purposes of the False
Claims Act.
Efforts
to ensure that our business arrangements will comply with
applicable healthcare laws may involve substantial costs. It is
possible that governmental and enforcement authorities will
conclude that our business practices do not comply with current or
future statutes, regulations or case law interpreting applicable
fraud and abuse or other healthcare laws and regulations. If any
such actions are instituted against us, and we are not successful
in defending ourselves or asserting our rights, those actions could
have a significant impact on our business, including the imposition
of civil, criminal and administrative penalties, damages,
disgorgement, monetary fines, possible exclusion from participation
in Medicare, Medicaid and other federal and similar foreign
healthcare programs, contractual damages, reputational harm,
diminished profits and future earnings, and curtailment of our
operations, any of which could harm our ability to operate our
business and our results of operations.
Compliance with environmental laws and regulations could be
expensive. Failure to comply with environmental laws and
regulations could subject us to significant liability.
Our
research and development and manufacturing operations may involve
the use of hazardous substances and are subject to a variety of
federal, state, local and foreign environmental laws and
regulations relating to the storage, use, discharge, disposal,
remediation of, and human exposure to, hazardous substances and the
sale, labeling, collection, recycling, treatment and disposal of
products containing hazardous substances. In addition, our research
and development and manufacturing operations produce biological
waste materials, such as human and animal tissue, and waste
solvents, such as isopropyl alcohol. These operations are permitted
by regulatory authorities, and the resultant waste materials are
disposed of in material compliance with environmental laws and
regulations. Liability under environmental laws and regulations can
be joint and several and without regard to fault or negligence.
Compliance with environmental laws and regulations may be expensive
and non-compliance could result in substantial liabilities, fines
and penalties, personal injury and third part property damage
claims and substantial investigation and remediation costs.
Environmental laws and regulations could become more stringent over
time, imposing greater compliance costs and increasing risks and
penalties associated with violations. We cannot assure you that
violations of these laws and regulations will not occur in the
future or have not occurred in the past as a result of human error,
accidents, equipment failure or other causes. The expense
associated with environmental regulation and remediation could harm
our financial condition and operating results.
22
Risks
Related to this Offering and Owning Our Securities,
Our Financial Results and Our Need for Financing
The preliminary financial data contained herein are based on
information existing as of the date hereof and there can be no
assurance that actual financial results will not differ,
potentially materially so.
The estimated
financial results included herein are based on information existing
as of the date hereof. During the preparation of our consolidated
financial statements for the year ended December 31, 2016, we may
identify items that would require us to make adjustments, which may
be material to the estimates described above. This preliminary
financial data has been prepared by and is the responsibility of
our management. RBSM LLP has not audited, reviewed, compiled or
performed any procedures with respect to this preliminary financial
data, and accordingly, RBSM LLP does not express an opinion or any
other form of assurance with respect thereto. Accordingly, there
can be no assurance that the projected results will be realized or
that actual results will not be significantly lower than
projected.
Our quarterly and annual results may fluctuate significantly, may
not fully reflect the underlying performance of our business and
may result in decreases in the price of our
securities.
Our
operating results will be affected by numerous factors such
as:
●
variations
in the level of expenses related to our proposed
products;
●
status
of our product development efforts;
●
execution
of collaborative, licensing or other arrangements, and the timing
of payments received or made under those arrangements;
●
intellectual
property prosecution and any infringement lawsuits to which we may
become a party;
●
regulatory
developments affecting our products or those of our
competitors;
●
our
ability to obtain and maintain FDA clearance and approval from
foreign regulatory authorities for our products, which have not yet
been approved for marketing;
●
market acceptance of our TAEUS
applications;
●
the availability of reimbursement for our
TAEUS applications;
●
our
ability to attract new customers and grow our business with
existing customers;
●
the
timing and success of new product and feature introductions by us
or our competitors or any other change in the competitive dynamics
of our industry, including consolidation among competitors,
customers or strategic partners;
●
the
amount and timing of costs and expenses related to the maintenance
and expansion of our business and operations;
●
changes
in our pricing policies or those of our competitors;
●
general
economic, industry and market conditions;
●
the
hiring, training and retention of key employees, including our
ability to expand our sales team;
●
litigation
or other claims against us;
●
our
ability to obtain additional financing; and
●
advances
and trends in new technologies and industry standards.
Any or
all of these factors could adversely affect our cash position
requiring us to raise additional capital which may be on
unfavorable terms and result in substantial dilution.
23
The forecasts of market growth included in this prospectus may
prove to be inaccurate, and even if the markets in which we compete
achieve the forecasted growth, our business may not grow at similar
rates, if at all.
Growth
forecasts are subject to significant uncertainty and are based on
assumptions and estimates that may not prove to be accurate. The
forecasts in this prospectus relating to, among other things, the
expected growth and need for diagnosis of NAFLD and the growth and
need for temperature monitoring of thermal ablation procedures may
prove to be inaccurate.
Even if
these markets experience the forecasted growth described in this
prospectus, we may not grow our business at similar rates, or at
all. Our growth is subject to many factors, including whether the
markets for NAFLD diagnosis and thermal ablation continue to grow,
the rate of market acceptance of our TAEUS applications versus the
products of our competitors and our success in implementing our
business strategies, each of which is subject to many risks and
uncertainties. Accordingly, the forecasts of market growth included
in this prospectus should not be taken as indicative of our future
growth.
If we are unable to implement and maintain effective internal
control over financial reporting in the future, investors may lose
confidence in the accuracy and completeness of our financial
reports and the market price of our securities may
decrease.
As a
public company, we will be required to maintain internal control
over financial reporting and to report any material weaknesses in
such internal controls. Section 404 of the Sarbanes-Oxley Act
of 2002, or the Sarbanes-Oxley Act, requires that we evaluate and
determine the effectiveness of our internal control over financial
reporting and, beginning with our annual report for the year ending
December 31, 2016, provide a management report on our internal
control over financial reporting, which must be attested to by our
independent registered public accounting firm to the extent we are
no longer an “emerging growth company,” as defined by
the Jumpstart Our Businesses Act of 2012, or the JOBS Act, or a
smaller reporting company under the Securities Act.
Currently and in
the future, if we have material weaknesses in our internal control
over financial reporting, we may not detect errors on a timely
basis and our financial statements may be materially misstated. We
are in the process of designing and implementing our internal
control over financial reporting, which process will be time
consuming, costly and complicated. Until such time as we are no
longer an "emerging growth company" or a smaller reporting company,
our auditors will not be required to attest as to our internal
control over financial reporting. If we continue to identify
material weaknesses in our internal control over financial
reporting, if we are unable to comply with the requirements of
Section 404 in a timely manner, if we are unable to assert
that our internal control over financial reporting is effective or,
once required, if our independent registered public accounting firm
is unable to attest that our internal control over financial
reporting is effective, investors may lose confidence in the
accuracy and completeness of our financial reports and the market
price of our common stock could decrease. We could also become
subject to stockholder or other third-party litigation as well as
investigations by the stock exchange on which our securities are
listed, the Securities and Exchange Commission, or the SEC, or
other regulatory authorities, which could require additional
financial and management resources and could result in fines,
trading suspensions or other remedies.
We may not be able to secure additional financing on favorable
terms, or at all, to meet our future capital needs and our failure
to obtain additional financing when needed could force us to delay,
reduce or eliminate our product development programs and
commercialization efforts.
We
believe that the net proceeds from this offering, together with our
current cash and expected revenues from operations, will be
sufficient for us to fund the development, regulatory approval and
the commercialization of our NAFLD TAEUS application in the
European Union and the United States. However, the expected net
proceeds from this offering are not expected to be sufficient for
us to complete the full commercialization of this application or to
complete the development of any other TAEUS application. As a
result, we expect that we will need to finance our future cash
needs through public or private equity offerings, debt financings,
corporate collaboration and licensing arrangements or other
financing alternatives.
24
To
date, we have financed our operations primarily through sales of
our Nexus 128 system and net proceeds from the issuance of equity
and debt securities. We do not know when or if our operations will
generate sufficient cash to fund our ongoing operations. In the
future, we may require additional capital in order to
(i) continue to conduct research and development activities;
(ii) conduct clinical studies; (iii) fund the costs of seeking
regulatory approval of TAEUS applications; (iv) expand our
sales and marketing infrastructure; (v) acquire complementary
business technology or products; or (vi) respond to business
opportunities, challenges, increased regulatory obligations or
unforeseen circumstances. Our forecast of the period of time
through which our financial resources will be adequate to support
our operations is a forward-looking statement and involves risks
and uncertainties, and actual results could vary as a result of a
number of factors, including the factors discussed elsewhere in
this “Risk Factors” section. We have based this
estimate on assumptions that may prove to be wrong, and we could
utilize our available capital resources sooner than we currently
expect. Our future funding requirements will depend on many
factors, including, but not limited to:
●
the costs, timing and outcomes of regulatory
reviews associated with our future products, including TAEUS
applications;
●
the
costs and expenses of expanding our sales and marketing
infrastructure;
●
the costs and timing of developing variations of
our TAEUS applications and, if
necessary, obtaining regulatory clearance of such
variations;
●
the degree of success we experience in
commercializing our products, particularly our TAEUS
applications;
●
the extent to which our TAEUS applications
are adopted by hospitals for use by
primary care physicians, hepatologists, radiologists and
oncologists for diagnosis of fatty liver disease and the thermal
ablation of lesions;
●
the
number and types of future products we develop and
commercialize;
●
the
costs of preparing, filing and prosecuting patent applications and
maintaining, enforcing and defending intellectual property-related
claims;
●
the
extent and scope of our general and administrative
expenses;
●
the
progress, timing, scope and costs of our clinical trials, including
the ability to timely enroll patients in our planned and potential
future clinical trials;
●
the
outcome, timing and cost of regulatory approvals, including the
potential that the FDA or comparable regulatory authorities may
require that we perform more studies than those that we currently
expect;
●
the
amount of sales and other revenues from technologies and products
that we may commercialize, if any, including the selling prices for
such potential products and the availability of adequate
third-party reimbursement;
●
selling
and marketing costs associated with our potential products,
including the cost and timing of expanding our marketing and sales
capabilities;
25
●
the
terms and timing of any potential future collaborations, licensing
or other arrangements that we may establish;
●
cash
requirements of any future acquisitions and/or the development of
other products;
●
the
costs of operating as a public company;
●
the
cost and timing of completion of commercial-scale, outsourced
manufacturing activities; and
●
the
time and cost necessary to respond to technological and market
developments.
We may
raise funds in equity or debt financings following our initial
public offering or enter into credit facilities in order to access
funds for our capital needs. Any debt financing obtained by us in
the future would cause us to incur debt service expenses and could
include restrictive covenants relating to our capital raising
activities and other financial and operational matters, which may
make it more difficult for us to obtain additional capital and
pursue business opportunities. If we raise additional funds through
further issuances of equity or convertible debt securities, our
existing stockholders could suffer significant dilution in their
percentage ownership of our Company, and any new equity securities
we issue could have rights, preferences and privileges senior to
those of holders of our common stock. General market conditions or
the market price of our common stock may not support capital
raising transactions such as an additional public or private
offering of our common stock or other securities. If we are unable
to obtain adequate financing or financing on terms satisfactory to
us when we require it, we may terminate or delay the development of
one or more of our products, or delay establishment of sales and
marketing capabilities or other activities necessary to
commercialize our products. In addition, if we raise additional
funds through collaborations, strategic alliances or marketing,
distribution or licensing arrangements with third parties, we may
have to relinquish valuable rights to our technologies, future
revenue streams or products or to grant licenses on terms that may
not be favorable to us.
If we
are unable to raise additional capital when needed, we may be
required to curtail the development of our technology or materially
curtail or reduce our operations. We could be forced to sell or
dispose of our rights or assets. Any inability to raise adequate
funds on commercially reasonable terms could have a material
adverse effect on our business, results of operation and financial
condition, including the possibility that a lack of funds could
cause our business to fail and liquidate with little or no return
to investors.
We may be subject to securities litigation, which is expensive and
could divert management attention.
The
price of our securities may be volatile, and in the
past companies that have experienced volatility in the market price
of their securities have been subject to an increased
incidence of securities class action litigation. We may be the
target of this type of litigation in the future. Securities
litigation against us could result in substantial costs and divert
our management’s attention from other business concerns,
which could seriously harm our business.
As an investor, you may lose all of your investment.
Investing in our
securities involves a high degree of risk. As an
investor, you may never recoup all, or even part, of your
investment and you may never realize any return on your investment.
You must be prepared to lose all of your investment.
26
Prior to the completion of our initial public offering, there will
have been no public trading market for our units, common stock or
warrants. An active public trading market for our securities may
not develop and our securities may trade below the public offering
price.
The
offering under this prospectus is an initial public offering of our
securities. Prior to the closing of the offering, there
will have been no public market for our units, common
stock or warrants. An active public trading market for our
units, common stock and warrants may not develop after
the completion of the offering. If an active trading market
for our units, common stock or warrants does not
develop after this offering, the market price and liquidity of our
units, common stock and warrants may be materially and
adversely affected. The public offering price for our
units has been determined by negotiation among us and
the underwriters based upon several factors, and the price at which
our units trade after this offering may decline below
the public offering price. Investors in our units may
experience a significant decrease in the value of their
units regardless of our operating performance or
prospects.
If a public market for units, our common stock or
warrants develops, it may be volatile. This may affect
the ability of our investors to sell their securities
as well as the price at which they sell their
securities.
If a
market for our units, common stock or warrants
develops, the market price for the units, shares and
warrants may be significantly affected by factors such as
variations in quarterly and yearly operating results, general
trends in the medical imaging industry, and changes in state or
federal regulations affecting us and our
industry. Furthermore, in recent years the stock market
has experienced extreme price and volume fluctuations that are
unrelated or disproportionate to the operating performance of the
affected companies. Such broad market fluctuations may
adversely affect the market price of our units, common
stock and warrants, if markets for them develop.
In
addition to market and industry factors, the price and trading
volume for our units, common stock and warrants may be
highly volatile for specific business reasons,
including:
●
announcements
of regulatory approval or a complete response letter, or specific
label indications or patient populations for its use, or changes or
delays in the regulatory review process;
●
announcements
of innovations or new products by us or other participants in the
medical imaging market;
●
adverse
actions taken by regulatory agencies with respect to our clinical
trials, manufacturing supply chain or sales and marketing
activities;
●
any
adverse changes to our relationship with manufacturers or
suppliers;
●
results
of our testing and clinical trials;
●
results
of our efforts to acquire or license additional
products;
●
variations
in the level of expenses related to our existing products or
pre-clinical and clinical development programs;
●
any
intellectual property infringement actions in which we may become
involved;
●
announcements
concerning our competitors or the medical imaging industry in
general;
●
achievement
of expected product sales and profitability;
●
manufacture,
supply or distribution shortages;
●
variations
in our results of operations;
●
announcements
about our earnings that are not in line with analyst
expectations;
27
●
publication
of operating or industry metrics by third parties, including
government statistical agencies, that differ from expectations of
industry or financial analysts;
●
changes
in financial estimates by securities research
analysts;
●
press
reports, whether or not true, about our
business;
●
additions
to or departures of our management;
●
release
or expiry of lock-up or other transfer restrictions on our
outstanding securities;
●
sales
or perceived potential sales of additional
securities;
●
sales
of our securities by us, our executive officers and
directors or our stockholders in the future;
●
general
economic and market conditions and overall fluctuations in the U.S.
equity markets; and
●
changes
in accounting principles.
Any of
these factors may result in large and sudden changes in the volume
and trading price of our securities. In addition, the
stock market, in general, and smaller companies like ours, in
particular, have experienced extreme price and volume fluctuations
that have often been unrelated or disproportionate to the operating
performance of these companies. Broad market and industry factors
may negatively affect the market price of our units,
common stock and warrants, regardless of our actual operating
performance.
The warrants may not have any value.
The
warrants will be exercisable beginning after
separation from the units and until the five
year anniversary
of the date of initial issuance of the units at an initial
exercise price equal to 120% of the initial offering price per
unit set forth on the cover page of this prospectus.
In the event that our common stock price does not exceed the
exercise price of the warrants during the period when the warrants
are exercisable, the warrants may not have any value.
A warrant does not entitle the holder to any rights as common
stockholders until the holder exercises the warrant for
a share of our common stock.
Until
you acquire shares upon exercise of your warrants, the warrants
will not provide you any rights as a common stockholder. Upon
exercise of your warrants, you will be entitled to exercise the
rights of a common stockholder only as to matters for which the
record date occurs after the exercise date.
We are an “emerging growth company” under the JOBS Act
and we cannot be certain if the reduced disclosure requirements
applicable to emerging growth companies will make our
securities less attractive to investors.
We are
an “emerging growth company,” as defined in the
Jumpstart Our Business Startups Act of 2012, or the JOBS Act, and
we may take advantage of certain exemptions from various reporting
requirements that are applicable to other public companies that are
not “emerging growth companies” including, but not
limited to, not being required to comply with the auditor
attestation requirements of section 404 of the Sarbanes-Oxley Act,
reduced disclosure obligations regarding executive compensation in
our periodic reports and proxy statements, and exemptions from the
requirements of holding a nonbinding advisory vote on executive
compensation and stockholder approval of any golden parachute
payments not previously approved. We cannot predict if
investors will find our securities less attractive
because we may rely on these exemptions. If some
investors find our securities less attractive as a
result, there may be a less active trading market for our
securities and the price of our
securities may be more volatile.
We will
remain an “emerging growth company” for up to five
years, although we will lose that status sooner if our annual
revenues exceed $1 billion, if we issue more than $1 billion in
non-convertible debt in a three year period, or if the market value
of our common stock that is held by non-affiliates exceeds $700
million as of any June 30.
If securities or industry analysts do not publish research reports
about our business, or if they issue an adverse opinion about our
business, the price of our securities and
trading volume could decline.
The
trading market for our securities will be influenced
by the research and reports that industry or securities analysts
publish about us or our business. We do not currently have and may
never obtain research coverage by securities and industry analysts.
If no or few analysts commence research coverage of us, or one or
more of the analysts who cover us issues an adverse opinion about
our company, the price of our securities
would likely decline. If one or more of these analysts ceases
research coverage of us or fails to regularly publish reports on
us, we could lose visibility in the financial markets, which in
turn could cause the price of our
securities or trading volume to decline.
28
We have not paid dividends in the past and have no immediate plans
to pay dividends.
We plan
to reinvest all of our earnings, to the extent we have earnings, in
order to further develop our technology and potential products and
to cover operating costs. We do not plan to pay any cash
dividends with respect to our securities in the foreseeable
future. We cannot assure you that we would, at any time,
generate sufficient surplus cash that would be available for
distribution to the holders of our common stock as a
dividend. Therefore, you should not expect to receive
cash dividends on the common stock we are
offering.
Concentration of ownership among our existing executive officers,
directors and significant stockholders may prevent new investors
from influencing significant corporate decisions.
All
decisions with respect to the management of the Company will be
made by our board of directors and our officers, who, before this
offering, beneficially own approximately 25.2% of our common stock,
as calculated in accordance with Rule 13d-3 promulgated under the
Securities Exchange Act of 1934, as amended, or the Exchange
Act. After the issuance of our common stock in this
offering, management will beneficially own approximately 9.8% of
our common stock, as calculated in accordance with Rule 13d-3. In
addition, before this offering, Blue Earth Fund, LP, Jeffrey and
Margaret Padnos, Erick Richardson, Benjamin Padnos and Robert
Clifford and their affiliates beneficially own approximately 22.9%,
16.5%, 15.9%, 12.9% and 10.0% of our common stock, respectively,
and after this offering will beneficially own approximately 6.1%,
4.1%, 4.1%, 3.1% and 2.3% of our common stock, respectively, as
calculated in accordance with Rule 13d-3 promulgated under the
Exchange Act. As a result, these stockholders will be
able to exercise a substantial level of control over all matters
requiring stockholder approval, including the election of
directors, amendment of our Fourth Amended and Restated Certificate
of Incorporation, or the Certificate of Incorporation, and approval
of significant corporate transactions. This control could have the
effect of delaying or preventing a change of control of the Company
or changes in management and will make the approval of certain
transactions difficult or impossible without the support of these
stockholders.
We will incur significant increased costs as a result of becoming a
public company that reports to the SEC and our management will be
required to devote substantial time to meet compliance
obligations.
As a
public company listed in the United States, we will incur
significant legal, accounting and other expenses that we did not
incur as a private company. We will be subject to
reporting requirements of the Exchange Act and the Sarbanes-Oxley
Act, as well as rules subsequently implemented by the SEC and
Nasdaq that impose significant requirements on public companies,
including requiring the establishment and maintenance of effective
disclosure and financial controls and changes in corporate
governance practices. In addition, in 2010, the
Dodd-Frank Wall Street Reform and Protection Act, or the Dodd-Frank
Act, was enacted. There are significant corporate
governance and executive compensation-related provisions in the
Dodd-Frank Act that will increase our legal and financial
compliance costs, make some activities more difficult,
time-consuming or costly and may also place undue strain on our
personnel, systems and resources. Our management and
other personnel will need to devote a substantial amount of time to
these compliance initiatives. In addition, these rules
and regulations may make it more difficult and more expensive for
us to obtain director and officer liability insurance, and we may
be required to accept reduced policy limits and coverage or incur
substantially higher costs to obtain the same or similar
coverage. As a result, it may be more difficult for us
to attract and retain qualified people to serve on our board of
directors, our board committees or as executive
officers.
29
A
significant portion of our total outstanding shares of common stock
is restricted from immediate resale but may be sold into the market
in the near future. This could cause the market price of our
securities to drop significantly, even if our business is doing
well.
Sales
of a substantial number of shares of our common stock in the public
market could occur in the future. These sales, or the perception in
the market that the holders of a large number of
securities intend to sell shares, could reduce the
market price of our common stock. After this offering, we will have
outstanding 4,443,213 shares of common stock (or 4,743,213
shares if the underwriters exercise their option to purchase
additional units in full). Of these
outstanding shares, all of the 2,000,000 shares of common stock
(or
2,300,000 shares if the underwriters exercise their option to
purchase additional units in full) and
warrants exercisable for
2,000,000 shares of common stock
(2,300,000 shares if the underwriters exercise their
option to purchase additional units in full)
issued in this offering will be freely tradable. In
addition, we expect that the shares issued upon exercise of the
warrants issued in this offering will be freely tradeable. The
remaining outstanding shares of our common stock will be
restricted under securities laws or as a result of lock-up
agreements but will be able to be resold after the offering as
described in the “Shares Eligible for Future Sale”
section of this prospectus. We also intend to register all shares
of common stock that we may issue under our equity compensation
plans. Once we register these shares, they can be freely sold in
the public market upon issuance and once vested, subject to the
#180- and 365-day lock-up periods under the lock-up
agreements described in the “Underwriting” section of
this prospectus.
Future sales and issuances of our common stock or rights to
purchase common stock, including pursuant to our equity incentive
plan, could result in additional dilution of the percentage
ownership of our stockholders and could cause the
price of our securities to fall.
We
expect that significant additional capital will be needed in the
future to continue our planned operations. To the extent we raise
additional capital by issuing equity securities, our stockholders
may experience substantial dilution. We may sell common stock,
convertible securities or other equity securities. If we sell
common stock, convertible securities or other equity securities,
your investment in our common stock will be diluted. These sales
may also result in material dilution to our existing stockholders,
and new investors could gain rights superior to our existing
stockholders.
We may allocate the net proceeds from this offering in ways that
differ from the estimates discussed in the section titled
“Use of Proceeds” and with which you may not
agree.
The
allocation of net proceeds of this offering set forth in the
“Use of Proceeds” section below represents our
estimates based upon our current plans and assumptions regarding
industry and general economic conditions, and our future revenues
and expenditures. The amounts and timing of our actual
expenditures will depend on numerous factors, including market
conditions, cash generated by our operations, business developments
and related rate of growth. We may find it necessary or
advisable to use portions of the proceeds from this offering for
other purposes. Circumstances that may give rise to a
change in the use of proceeds and the alternate purposes for which
the proceeds may be used are discussed in the section entitled
“Use of Proceeds” below. You may not have an
opportunity to evaluate the economic, financial or other
information on which we base our decisions on how to use our
proceeds. As a result, you and other stockholders may
not agree with our decisions. See “Use of
Proceeds” for additional information.
You will experience immediate dilution in the book value per share
of the common stock you purchase.
The
initial public offering price per unit (and our common stock
forming part of such unit) will be substantially higher than
the net tangible book value (deficit) per share of our common stock
immediately prior to the offering. After giving effect to the
assumed sale of units in this offering, at the assumed
initial public offering price of $5.25 per unit, the
mid-point of the range set forth on the cover page of this
prospectus, and after deducting the underwriting discount and
estimated offering expenses payable by us and attributing no value
to the warrants included in the units, purchasers of
our units in this offering will incur immediate
dilution of $2.97 per share in the net tangible book value
(deficit) of the common stock they acquire. In the event that you
exercise your warrants, you will experience additional dilution to
the extent that the exercise price of the warrants is higher than
the tangible book value (deficit) per share of our common stock.
For a further description of the dilution that investors in this
offering will experience, see “Dilution.”
Our charter documents and Delaware law may inhibit a takeover that
stockholders consider favorable.
Upon
the closing of this offering, provisions of our Certificate of
Incorporation and bylaws and applicable provisions of Delaware law
may delay or discourage transactions involving an actual or
potential change in control or change in our management, including
transactions in which stockholders might otherwise receive a
premium for their shares, or transactions that our stockholders
might otherwise deem to be in their best interests. The
provisions in our Certificate of Incorporation and
bylaws:
30
●
authorize
our board of directors to issue preferred stock without stockholder
approval and to designate the rights, preferences and privileges of
each class; if issued, such preferred stock would increase the
number of outstanding shares of our common stock and could include
terms that may deter an acquisition of us;
●
limit
who may call stockholder meetings;
●
do
not provide for cumulative voting rights; and
●
provide
that all vacancies may be filled by the affirmative vote of a
majority of directors then in office, even if less than a
quorum.
In
addition, once we become a publicly traded corporation, section 203
of the Delaware General Corporation Law may limit our ability to
engage in any business combination with a person who beneficially
owns 15% or more of our outstanding voting stock unless certain
conditions are satisfied. This restriction lasts for a
period of three years following the share
acquisition. These provisions may have the effect of
entrenching our management team and may deprive you of the
opportunity to sell your shares to potential acquirers at a premium
over prevailing prices. This potential inability to
obtain a control premium could reduce the price of our common
stock. See “Anti-Takeover Effects of Certain Provisions of
Delaware Law and Our Charter Documents” for additional
information.
Our disclosure controls and procedures may not prevent or detect
all errors or acts of fraud.
Upon
completion of this offering, we will become subject to the periodic
reporting requirements of the Exchange Act. Our disclosure controls
and procedures are designed to reasonably assure that information
required to be disclosed by us in reports we file or submit under
the Exchange Act is accumulated and communicated to management, and
recorded, processed, summarized and reported within the time
periods specified by the rules and forms of the SEC. We believe
that any disclosure controls and procedures or internal controls
and procedures, no matter how well conceived and operated, can
provide only reasonable, not absolute, assurance that the
objectives of the control system are met.
These
inherent limitations include the realities that judgments in
decision-making can be faulty, and that breakdowns can occur
because of simple error or mistake. Additionally, controls can be
circumvented by the individual acts of some persons, by collusion
of two or more people or by an unauthorized override of the
controls. Accordingly, because of the inherent limitations in our
control system, misstatements due to error or fraud may occur and
not be detected.
31
SPECIAL NOTE REGARDING
FORWARD-LOOKING STATEMENTS AND OTHER INFORMATION CONTAINED IN THIS
PROSPECTUS
This
prospectus contains forward-looking statements. All statements
other than statements of historical facts contained in this
prospectus, including statements regarding the timing of our
clinical trials, our strategy, future operations, future financial
position, future revenue, projected costs, prospects, plans,
objectives of management and expected market growth
are forward-looking
statements. In addition, our preliminary financial data for
the quarter and year ended December 31, 2016 contained in this
prospectus are forward-looking
statements. Forward-looking statements give our current
expectations or forecasts of future events. You can find many (but
not all) of these statements by looking for words such as
“approximates,” “believes,”
“hopes,” “expects,”
“anticipates,” “estimates,”
“projects,” “intends,” “plans,”
“would,” “should,” “could,”
“may” or other similar expressions in this
prospectus. These statements may be found principally
under the sections entitled “Prospectus Summary,”
“Risk Factors,” “Use of Proceeds,”
“Management’s Discussion and Analysis of Financial
Condition and Results of Operations” and
“Business.” These forward-looking statements are
subject to certain risks and uncertainties that could cause actual
results to differ materially from our historical experience and our
present expectations or projections. Actual results may differ materially from those
discussed as a result of various factors, including, but not
limited to:
●
our
need to secure required regulatory approvals from governmental
authorities in the European Union, United States and other
jurisdictions;
●
our dependence on
third parties to design, manufacture, obtain required regulatory
approvals of, market and distribute our TAEUS applications;
●
our ability to commercialize any applications of
our TAEUS technology and the
pricing of any such applications;
●
our
limited operating history and our ability to achieve
profitability;
●
our
need for and ability to obtain adequate financing in the
future;
●
the
impact of competitive or alternative products, technologies and
pricing;
●
the
adequacy of protections afforded to us by the patents that we own
and the success we may have in, and the cost to us of, maintaining,
enforcing and defending those patents;
●
our
ability to obtain, expand and maintain patent protection in the
future, and to protect our non-patented intellectual
property;
●
general
economic conditions and events and the impact they may have on us
and our potential customers;
●
our
ability to continue as a going concern;
●
our success at managing the risks involved in the
foregoing items; and
●
other factors
discussed in the “Risk Factors” section of this
prospectus.
These
statements reflect our views with respect to future events as of
the date of this prospectus and are based on assumptions and
subject to risks and uncertainties. Given these uncertainties, you
should not place undue reliance on these forward-looking
statements. These forward-looking statements represent our
estimates and assumptions only as of the date of this prospectus
and, except as required by law, we undertake no obligation to
update or review publicly any forward-looking statements, whether
as a result of new information, future events or otherwise after
the date of this prospectus. We anticipate that subsequent events
and developments will cause our views to change. You should read
this prospectus and the documents referenced in this prospectus and
filed as exhibits to the registration statement, of which this
prospectus is a part, completely and with the understanding that
our actual future results may be materially different from what we
expect. Our forward-looking statements do not reflect the potential
impact of any future acquisitions, merger, dispositions, joint
ventures or investments we may undertake. We qualify all of our
forward-looking statements by these cautionary
statements.
32
We
estimate that we will receive net proceeds of approximately
$9,060,000 from this offering, or approximately $10,509,000 if the
underwriters exercise their over-allotment option in full, based on
an assumed initial public offering price of $5.25, the mid-point of
the range set forth on the cover page of this prospectus, and after
deducting the underwriting discount and estimated offering expenses
payable by us and excluding the proceeds, if any, from the exercise
of the warrants.
A $0.25
increase (decrease) in the initial public offering price of $5.25
per unit would increase (decrease) the net proceeds to
us from this offering by approximately $460,000, assuming the
number of units offered by us, as set forth on the
cover page of this prospectus, remains the same and after deducting
the underwriting discount and estimated offering expenses payable
by us. Similarly, each increase (decrease) of one hundred thousand
in the number of units offered by us, as set forth on
the cover page of this prospectus, would increase (decrease) the
net proceeds to us from this offering by approximately $483,000,
assuming the initial public offering price of $5.25 per
unit, the mid-point of the price range set forth on
the cover page of this prospectus, remains the same and after
deducting the underwriting discount and estimated offering expenses
payable by us.
We
intend to use the net proceeds from this offering to fund the
development, regulatory approval and the initial commercialization
of our NAFLD TAEUS application in the European Union and the United
States and for working capital and other general corporate
purposes. Our initial target market for this application is the
European Union. We believe that our NAFLD TAEUS application will
qualify for sale in the European Union as a Class IIa medical
device. As a result, we will be required to obtain a CE mark for
our NAFLD TAEUS application before we can sell the application in
the European Union.
Existing
regulations would not require us to conduct a clinical trial to
obtain a CE mark for this application. Nonetheless, for commercial
reasons and to support our CE mark application, we plan to conduct
a limited (less than 10 person) trial to demonstrate our NAFLD
TAEUS application’s ability to distinguish fat from lean
tissue. We intend to engage a medical device contract engineering
firm to perform commercial product engineering, and to obtain a CE
mark, for this application. Based on our understanding of
applicable regulations and consultations with medical device
regulatory consulting firms and medical device contract engineering
firms, we expect that the development of our NAFLD TAEUS
application, including the receipt of the necessary CE mark, will
be complete approximately ten months after the completion of this
offering and that we will use approximately $1,000,000 of the net proceeds from this offering on such
activities.
While we are seeking a CE mark for our NAFLD TAEUS
application, we also plan to
expand our sales, marketing, customer support and
contract manufacturing capabilities,
so that we can commence commercial sales of the application in the
European Union promptly following receipt of this regulatory
approval. We estimate that we will use approximately $700,000
of the net proceeds from this offering on such
activities.
Additionally, to
enhance our commercialization efforts in the European Union,
following receipt of such CE mark and placement of initial systems
with researchers and universities, we plan to conduct one or more
clinical studies to demonstrate this product’s capabilities,
and that we will use approximately $700,000 of the net proceeds
from this offering on such activities. However, these estimates are
subject to uncertainty and there can be no assurance that these
processes will not take longer or be more costly than we
expect.
While
the process of obtaining a CE mark for our NAFLD TAEUS application
is underway, we also intend to prepare for submission to the U.S.
Food and Drug Administration, or the FDA, an application under the
Food, Drug and Cosmetic Act, or the FD&C Act, to sell our NAFLD
TAEUS application in the U.S. We anticipate that the application,
as well as those for our other TAEUS applications, will be
submitted for approval under Section 510(k) of the FD&C Act.
Based on our understanding of applicable regulations and
consultations with medical device regulatory consulting firms and
medical device contract engineering firms, we expect to submit this
application to the FDA approximately eleven months after the
completion of this offering and for the FDA to make a final
determination on our application approximately eleven months after
that application is submitted. We estimate that we will use
approximately $100,000 of the net proceeds from this offering on
such activities. However, these estimates are subject to
uncertainty and there can be no assurance that these processes will
not take longer or be more costly than we expect.
We
expect to use the balance of the net proceeds from this offering on
research and development, additional regulatory activities, sales
and marketing activities for general and administrative expense and
other general corporate purposes.
We
expect that our initial FDA clearance will allow us to sell the
NAFLD TAEUS application in the U.S. with general imaging claims.
However, we will need to obtain additional FDA clearances to be
able to make diagnostic claims for fatty tissue content
determination. Accordingly, to support our commercialization
efforts we expect that, following receipt of our initial FDA
clearance, we will submit one or more additional applications to
the FDA, each of which will need to include additional clinical
trial data, so that following receipt of the necessary clearances
we may make those diagnostic claims.
The
amounts and timing of our actual expenditures will depend on
numerous factors, including market conditions, results from our
research and development efforts, business developments and related
sales and marketing activities. Therefore, as of the date of this
prospectus, we cannot specify with certainty the specific
allocation of the net proceeds to be received upon the completion
of this offering. Our management will have broad discretion in the
application of the net proceeds, and investors will be relying on
the judgment of our management regarding the application of the
proceeds from this offering.
We
believe that the net proceeds of this offering, together with our
existing cash, will be sufficient for us to fund the development of
our NAFLD TAEUS application through the expected receipt of
regulatory approval in the European Union and the United States and
to commercialize this application in those markets. It is possible
that we will not achieve the progress that we expect because of
unforeseen costs or factors impacting timely completion of the
regulatory approvals for a new medical device, which are difficult
to predict and are subject to risks and delays. We have no other
committed external sources of funds. The expected net proceeds from
this offering are not expected to be sufficient for us to complete
the full commercialization of our NAFLD TAEUS application or to
complete the development of any other TAEUS application. As a
result, we expect that we will need to finance our future cash
needs through public or private equity offerings, debt financings,
corporate collaboration and licensing arrangements or other
financing alternatives.
Pending
our use of the net proceeds from this offering, we intend to invest
the net proceeds in a variety of capital preservation investments,
including short-term, investment-grade, interest-bearing
instruments and U.S. government securities.
33
We have
never declared or paid any dividends on our capital stock, and do
not plan to do so for the foreseeable future. We expect that we
will retain all of our available funds and future earnings, if any,
for use in the operation and expansion of our business. The terms
of any loan agreement we enter into or any debt securities we may
issue are likely to contain restrictions on our ability to pay
dividends on our capital stock. Subject to the foregoing, the
payment of dividends in the future, if any, will be at the
discretion of our board of directors and will depend upon such
factors as earnings levels, capital requirements, restrictions
imposed by applicable law, our overall financial condition and any
other factors deemed relevant by our board of
directors.
34
The
following table sets forth our actual cash and capitalization, each
as of September 30, 2016:
●
on an actual
basis;
●
on a pro forma
basis to give effect to the reverse stock split, to reflect the
filing of our Fourth Amended and Restated Certificate of
Incorporation in connection with this offering and to give effect to the assumed
conversion of the outstanding principal and accrued interest on our
outstanding convertible promissory notes as of September 30, 2016
into an aggregate of
1,430,490 shares of our common stock at a conversion price of $1.00
per share immediately prior to the completion of this offering;
and
●
on a pro forma as
adjusted basis, to further reflect the sale by us of units at
an initial public offering price of $5.25 per
unit, the mid-point of the price range set forth on
the cover page of this prospectus, and after deducting the
underwriting discount and estimated offering expenses payable by us
and the receipt by us of the expected net proceeds of such
sale.
The pro
forma and pro forma as adjusted information below is illustrative
only, and our capitalization following the closing of this offering
may differ from that shown below based on the actual initial public
offering price and other terms of this offering determined at
pricing. You should read this information together with the
sections entitled “Summary Financial Data” and
“Management’s Discussion and Analysis of Financial
Condition and Results of Operations” and our financial
statements and the related notes, which appear elsewhere in this
prospectus.
|
|
As of September 30, 2016
|
||
|
|
Actual
|
Pro Forma
|
Pro Forma As Adjusted (1)(2)
|
|
|
(unaudited)
|
||
|
Cash
|
$75,543
|
$75,543
|
$ 9,175,543
|
|
|
|
|
|
|
Capitalization
|
|
|
|
|
Debt:
|
|
--
|
--
|
|
Senior
convertible notes
|
1,430,490
|
--
|
--
|
|
Total
debt
|
$ 1,430,490
|
$--
|
$--
|
|
|
|
|
|
|
Stockholders
Equity:
|
|
|
|
|
Preferred
stock, $0.0001 par value; 34,861,927 shares authorized; no shares
issued or outstanding, actual; 10,000,000 shares authorized pro
forma; no shares issued or outstanding pro forma and pro forma as
adjusted
|
--
|
--
|
--
|
|
Common stock, $0.0001 par value; 45,000,000 shares
authorized; 2,531,808 shares issued and
outstanding actual; 50,000,000 shares authorized and 2,443,213
shares issued and outstanding, pro forma, and 50,000,000 shares
authorized and 4,443,213 shares issued and outstanding, pro forma
as adjusted
|
253
|
244
|
444
|
|
Stock
payable(3)
|
72,000
|
72,000
|
72,000
|
|
Additional
paid-in capital
|
11,518,145
|
12,948,644
|
22,048,444
|
|
Accumulated
deficit
|
(11,831,594)
|
(11,831,594)
|
(11,831,594)
|
|
Total
stockholders equity
|
(241,196)
|
1,189,294
|
10,289,294
|
|
|
|
|
|
|
Total
capitalization
|
$ 1,189,294
|
$ 1,189,294
|
10,289,294
|
__________
(1)
A $0.25 increase
(decrease) in the assumed initial public offering price of
$5.25 per unit,
the mid-point of the price range set forth on the cover page of
this prospectus, would increase or decrease the amount of cash,
cash equivalents and short-term investments, additional paid-in
capital and total capitalization by approximately $460,000,
assuming the number of units offered by us, as set
forth on the cover page of this prospectus, remains the same and
after deducting the underwriting discount and estimated offering
expenses payable by us. Similarly, a one hundred thousand
unit increase (decrease) in the number of
units offered by us, as set forth on the cover page of
this prospectus, would increase (decrease) cash and cash
equivalents, working capital, total assets and total
stockholders’ equity by approximately $483,000, assuming the assumed initial public
offering price of $5.25 per
unit, the mid-point of the price range set forth on
the cover page of this prospectus, remains the same, and after
deducting the underwriting discount and estimated offering expenses
payable by us.
(2)
The number of shares of our common stock to be
outstanding after this offering is based on 1,012,723
shares of common stock outstanding as
of September 30, 2016, assumes the conversion of the principal and
accrued interest on our outstanding convertible promissory notes as
of September 30, 2016 into an aggregate of 1,430,490 shares of our
common stock at a conversion price of $1.00 per share immediately
prior to the completion of this offering, and excludes the
following:
●
213,937 shares of common stock issuable
upon the exercise of outstanding warrants, at a weighted average
exercise price of $13.53 per
share;
●
213,452
shares of our common stock issuable
upon the exercise of outstanding stock options issued pursuant to
our 2016
Omnibus Incentive Plan,
or our
Incentive Plan, at a weighted average
exercise price of $7.15 per
share and an
estimated 792,782 shares of our common stock issuable
upon the exercise of stock options expected to be granted to our
directors and certain of our officers upon the completion of this
offering at an exercise price equal to the public offering price
set forth on the cover of this
prospectus;
●
an
estimated 517,643 shares of our common
stock that will be reserved for future issuance under our Incentive
Plan;
●
up to
2,300,000 shares of our common stock that may be
issued under warrants to be sold in this offering;
and
●
up to 184,000
shares of our common stock issuable upon exercise of the
underwriters' warrants.
(3)
Stock Payable is to be issued at the closing of this offering to
StoryCorp Consulting (d/b/a Wells Compliance Group) in an amount of
13,714 shares of common stock.
35
If you
purchase units in this offering, you will experience
dilution to the extent of the difference between the public
offering price per unit in this offering and our pro
forma as adjusted net tangible book value per share immediately
after this offering, assuming no value is attributed
to the warrants and such warrants are accounted for and classified
as equity.
Net
tangible book value per share represents total tangible assets less
total liabilities, divided by the number of shares of common stock
outstanding. Our historical net tangible book value as of September
30, 2016 (assuming the reverse stock split) was
$(241,196), or $(0.10) per share of
common stock. On a pro forma basis assuming the conversion of the
principal and accrued interest on our outstanding convertible
promissory notes as of September 30, 2016 into an aggregate of
1,430,490 shares of our common stock at a conversion price of $1.00
per share immediately prior to the completion of this offering, our
pro forma net tangible book value as of September 30, 2016 would
have been $1,189,294, or $0.49 per share.
After giving effect to the assumed sale of 2,000,000
units in this offering at the assumed initial public
offering price of $5.25 per unit, the mid-point of the
price range set forth on the cover page of this prospectus, after
deducting the underwriting discount and estimated offering expenses
payable by us, our pro forma as adjusted net tangible book value as
of September 30, 2016 would have been $10,289,294, or
$2.32 per share. This represents an immediate increase
in pro forma as adjusted net tangible book value of
$1.83 per share to existing stockholders and an
immediate dilution in pro forma as adjusted net tangible book value
of $2.93 per share to investors in this offering. The
following table illustrates this dilution on a per share
basis:
|
Assumed initial
public offering price per unit
|
|
$ 5.25
|
|
Historical net
tangible book value per share as of September 30, 2016
|
$ (0.10)
|
|
|
Pro forma increase
in net tangible book value per share attributable to conversion of
convertible promissory notes
|
$0.58
|
|
|
Pro forma net
tangible book value per share as of September 30, 2016
|
$ 0.49
|
|
|
Pro forma increase
in net tangible book value per share attributable to new
investors
|
$ 1.83
|
|
|
Pro forma as
adjusted tangible book value per share, after giving effect to this
offering
|
|
2.32
|
|
Dilution of pro
forma as adjusted net tangible book value per share to new
investors
|
|
$ 2.93
|
If the
underwriters exercise in full their option to purchase 300,000
additional units at the initial public offering price
of $5.25 per unit, the pro forma as adjusted net
tangible book value per share after giving effect to this offering
would be $2.50 per share, which amount represents an
immediate increase in pro forma as adjusted net tangible book value
of $2.01 per share of our common stock to existing
stockholders and an immediate dilution in pro forma as adjusted net
tangible book value of $2.75 per share of our common
stock to new investors purchasing units in this
offering.
The
following table summarizes, on a pro forma as adjusted basis as
described above as of September 30, 2016 and with respect shares
acquired since 2013 (and excluding shares acquired prior to 2013,
for which it was impracticable to determine cost basis
information), the number of shares of common stock purchased from
us, the total consideration paid to us and the average price per share
paid to us by our existing stockholders (including holders
of our convertible promissory notes upon the conversion thereof)
and by new investors purchasing units in this offering
at the assumed initial public offering price of $5.25 per
unit, the mid-point of the price range set forth on
the cover page of this prospectus, attributing no value to the
warrants and assuming no exercise of the warrants, and before the
deduction of the underwriting discount and estimated offering
expenses payable by us. Investors purchasing units in
this offering will pay an average price per share substantially
higher than our existing stockholders paid.
|
|
Shares
Acquired
|
Total Consideration
|
Average Price
|
||
|
|
Number
|
Percent
|
Amount
|
Percent
|
Per Share
|
|
Existing
stockholders
|
2,162,891
|
52.0
|
$ 5,802,928
|
35.6
|
$ 2.68
|
|
New
investors
|
2,000,000
|
48.0
|
$ 10,500,000
|
64.4
|
$ 5.25
|
|
Total
|
|
100%
|
100%
|
|
|
36
In the
event that you exercise your warrants, you will experience
additional dilution to the extent that the exercise price of the
warrants is higher than the average price per share paid to us by
our existing stockholders. If any shares are issued upon exercise
of outstanding options or warrants, you may experience further
dilution.
The above
discussion and tables are based on 2,443,213 shares of common stock
outstanding as of September 30, 2016, assumes the conversion of the
principal and accrued interest on our outstanding convertible
promissory notes outstanding as of September 30, 2016 into an
aggregate of 1,430,490 shares of our common stock at a conversion
price of $1.00 per share immediately prior to the completion of
this offering, and excludes the
following:
●
213,937
shares of common stock issuable upon the exercise of outstanding
warrants, at a weighted average exercise price of $13.53 per
share;
●
213,452 shares of our common stock issuable upon
the exercise of outstanding stock options issued pursuant to our
2016 Omnibus Incentive Plan, or our Incentive Plan, at a weighted
average exercise price of $7.15 per share and an estimated
792,782 shares of our
common stock issuable upon the exercise of stock options expected
to be granted to our directors and certain of our officers upon the
completion of this offering at an exercise price equal to the
public offering price set forth on the cover of this
prospectus;
●
an
estimated 517,643 shares of our common stock that will
be reserved for future issuance under our Incentive
Plan;
●
up to
2,300,000 shares of our common stock that may be
issued under warrants to be issued to the public in
this offering; and
●
up
to 184,000 shares of our common stock issuable upon exercise of the
underwriters' warrants.
A $0.25
increase (decrease) in the assumed initial public offering price of
$5.25 per unit, the mid-point of the price range set
forth on the cover page of this prospectus, would increase
(decrease) the pro forma as adjusted net tangible book value per
share after this offering by approximately $0.11 per share and the
dilution of pro forma as adjusted net tangible book value per share
to new investors by approximately $0.14 per share, assuming that
the number of units offered by us, as set forth on the
cover page of this prospectus, remains the same and after deducting
the underwriting discount and estimated offering expenses payable
by us. An increase (decrease) of one hundred thousand in the number
of units offered by us, as set forth on the cover of
this prospectus, would increase (decrease) the pro forma as
adjusted net tangible book value per share after this offering by
approximately $0.06 and $(0.07), respectively and decrease
(increase) the dilution of pro forma as adjusted net tangible book
value per share to new investors by approximately $0.06 and
$(0.07), respectively, assuming the assumed initial public offering
price of $5.25 per unit, the mid-point of the price
range set forth on the cover page of this prospectus, remains the
same, and after deducting the underwriting discount and estimated
offering expenses payable by us.
We may choose to raise additional capital through the sale of
equity or convertible debt securities due to market conditions or
strategic considerations even if we believe we have sufficient
funds for our current or future operating plans. To the extent that
any of these options or warrants are exercised, new options are
issued under our Incentive Plan or we issue additional shares of
common stock or other equity securities in the future, there may be
further dilution to new investors participating in this
offering.
37
MANAGEMENT’S
DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF
OPERATIONS
You should read the following discussion and analysis of our
financial condition and results of operations together with the
section entitled “Summary Financial Data” and our
financial statements and related notes included elsewhere in this
prospectus. Some of the information contained in this discussion
and analysis or set forth elsewhere in this prospectus, including
information with respect to our plans and strategy for our business
and related financing, includes forward-looking statements that
involve risks and uncertainties. See “Cautionary Note
Regarding Forward-Looking Statements.” Our actual results may
differ materially from those described below. You should read the
“Risk Factors” section of this prospectus for a
discussion of important factors that could cause actual results to
differ materially from the results described in or implied by the
forward-looking statements contained in the following discussion
and analysis.
Overview
We have
commercialized an enhanced ultrasound technology for the
pre-clinical research market and are leveraging that expertise to
develop technology for increasing the capabilities of clinical
diagnostic ultrasound, to broaden patient access to the safe
diagnosis and treatment of a number of significant medical
conditions in circumstances where expensive X-ray computed
tomography, or CT, and magnetic resonance imaging, or MRI,
technology is unavailable or impractical.
Since
2010, we have marketed and sold our Nexus 128 system, which
combines light-based thermoacoustics and ultrasound, to address the
imaging needs of researchers studying disease models in
pre-clinical applications. Sales of the Nexus 128 system were
approximately
$560,000 in 2014 and $1.4 million in 2015. We
have not yet completed preparation of financial statements for the
quarter and year ended December 31, 2016, but, as described below
under “Recent Financial Results,” based on preliminary
data available to us, we expect to report total revenues of
approximately $515,000 for the quarter and year ended December 31,
2016, all of which revenue was attributable to sales of our Nexus
128 system. Our Nexus 128 system is used in a number of
leading global academic research centers, including Stanford
University, The University of Michigan, Shanghai Jiao Tong
University, and Purdue University. We expect to continue to sell
our Nexus 128 system to maintain a base level of revenue, but
believe the market potential for our clinical systems is much
higher.
We
believe that our TAEUS technology has the potential to add a number
of new capabilities to conventional ultrasound and thereby enhance
the utility of both existing and new ultrasound systems. Our TAEUS
platform is not intended to replace CT and MRI systems, both of
which are versatile imaging technologies with capabilities and uses
beyond the focus of our business. However, they are also expensive,
with a CT system costing approximately $1 million and an MRI system
costing up to $3 million. In addition, and in contrast to
ultrasound systems, due to their limited number and the fact that
they are usually fixed-in-place at major medical facilities, CT and
MRI systems are frequently inaccessible to patients.
We
believe that our TAEUS platform can extend the use of ultrasound
technology to a number of important applications that either
require the use of expensive CT or MRI imaging systems or where
imaging is not practical using existing technology. To demonstrate the
capabilities of our TAEUS platform, we have conducted various
internal ex-vivo laboratory experiments and have also conducted
limited internal in-vivo large animal studies. However, we have not
yet conducted any human studies and these capabilities are not
supported by clinical data that we have gathered in pursuit of
obtaining regulatory approvals or that was subject to regulatory
oversight and guidance. In our ex-vivo and in-vivo testing,
we have demonstrated that the TAEUS platform has the following
capabilities and potential clinical applications:
●
Tissue Composition:
Our TAEUS technology enables ultrasound to distinguish fat from
lean tissue. This capability would enable the use of TAEUS-enhanced
ultrasound for the identification, staging and monitoring of NAFLD,
a precursor to liver fibrosis, cirrhosis and liver
cancer.
●
Temperature
Monitoring: Our TAEUS technology enables traditional ultrasound to
visualize changes in tissue temperature, in real time. This
capability would enable the use of TAEUS-enhanced ultrasound to
guide thermoablative therapy, such as in the treatment of cardiac
atrial fibrillation, or removal of cancerous liver and kidney
lesions, with greater accuracy.
●
Vascular Imaging:
Our TAEUS technology enables ultrasound to view blood vessels from
any angle, using only a saline solution contrasting agent, unlike
Doppler ultrasound which requires precise viewing angles. This
capability would enable the use of TAEUS-enhanced ultrasound to
easily identify arterial plaque or malformed vessels.
●
Tissue Perfusion:
Our TAEUS technology enables ultrasound to image blood flow at the
capillary level in a region, organ or tissue. This capability could
be used to assist doctors in characterizing microvasculature fluid
flows symptomatic of damaged tissue, such as internal bleeding from
trauma, or diseased tissue, such as certain cancers.
38
After
approval, our TAEUS technology can be added as an accessory to
existing ultrasound systems, helping to improve clinical
decision-making on the front lines of patient care, without
requiring new clinical workflows or large capital investments. We
are also developing TAEUS for incorporation into new ultrasound
systems, primarily through our collaboration with GE
Healthcare.
We are not aware of any other
ultrasound devices in development that include the anticipated
functionality of our planned TAEUS
applications.
Based on our design work and our understanding of
the ultrasound accessory market, we intend to price our
initial NAFLD TAEUS application
at a price point approximating one-half of the price
of a new cart-based ultrasound system, which should enable
purchasers to recoup their investment in less than one year by
performing a relatively small number of additional ultrasound
procedures. We further believe that clinicians will be attracted to
our technology because it will enable them to perform more
procedures with their existing ultrasound equipment, thereby
retaining more imaging patients in their clinics rather than
referring patients out to a regional medical center for a CT or MRI
scan.
We
expect that the first-generation TAEUS application will be a
standalone ultrasound accessory designed to cost-effectively
quantify fat in the liver and stage progression of NAFLD, which can
only be achieved today with impractical surgical biopsies or MRI
scans. Subsequent TAEUS offerings are expected to be implemented
via a second generation hardware platform that can run multiple
clinical software applications that we will offer TAEUS users for a
one-time licensing fee – adding ongoing customer value to the
TAEUS platform and a growing software revenue stream for our
Company.
Each of
our TAEUS platform applications will require regulatory approvals
before we are able to sell or license the application. Based on
certain factors, such as the installed base of ultrasound systems,
availability of other imaging technologies, such as CT and MRI,
economic strength and applicable regulatory requirements, we intend
to seek initial approval of our applications for sale in the
European Union, followed by the United States, and
China.
We
believe that our NAFLD TAEUS application will qualify for sale in
the European Union as a Class IIa medical device. As a result, we
will be required to obtain a CE mark for our NAFLD TAEUS
application before we can sell the application in the European
Union. Existing regulations would not require us to conduct a
clinical trial to obtain a CE mark for this application.
Nonetheless, for commercial reasons and to support our CE mark
application we plan to conduct a limited (less than 10 person)
trial to demonstrate our NAFLD TAEUS application’s ability to
distinguish fat from lean tissue. Based on our understanding of
applicable regulations and consultations with medical device
regulatory consulting firms and medical device contract engineering
firms, we expect to receive a CE mark for our NAFLD TAEUS
application approximately ten months after the completion of this
offering. However, this estimate is subject to uncertainty and
there can be no assurance that this process will not take longer or
be more costly than we expect. While
we are seeking a CE mark for our NAFLD TAEUS application, we also
plan to expand our sales, marketing, customer support and contract
manufacturing capabilities, so that we can commence commercial
sales of the application in the European Union promptly following
receipt of this regulatory approval. Following receipt of
such CE mark and placement of initial systems with researchers and
universities, we plan to conduct one or more clinical studies to
further demonstrate this application’s
capabilities.
While
the process of obtaining a CE mark for our NAFLD TAEUS application
is underway, we also intend to prepare for submission to the U.S.
Food and Drug Administration, or the FDA, an application under the
Food, Drug and Cosmetic Act, or the FD&C Act, to sell our NAFLD
TAEUS application in the U.S. We anticipate that the application,
as well as those for our other TAEUS applications, will be
submitted for approval under Section 510(k) of the FD&C Act. In
connection with our initial submission to the FDA, we believe we
will be required to provide imaging verification and validation
testing data, as well as the data from the limited trial we plan to
conduct to support our CE mark application. We expect that our
initial FDA clearance will allow us to sell the NAFLD TAEUS
application in the U.S. with general imaging claims. However, we
will need to obtain additional FDA clearances to be able to make
diagnostic claims for fatty tissue content determination.
Accordingly, to support our commercialization efforts we expect
that, following receipt of our initial FDA clearance, we will
submit one or more additional applications to the FDA, each of
which will need to include additional clinical trial data, so that
following receipt of the necessary clearances we may make those
diagnostic claims. Based on our understanding of applicable
regulations and consultations with medical device regulatory
consulting firms and medical device contract engineering firms, we
expect to submit our initial FDA application to the FDA
approximately eleven months after the completion of this offering
and that the FDA will make a final determination on our application
approximately eleven months after it is submitted. However, these
estimates are subject to uncertainty and there can be no assurance
that these processes will not take longer or be more costly than we
expect.
39
Financial Operations
Overview
Revenue
To date
our revenue has been generated by the placement and sale of our
Nexus 128 system for use in pre-clinical applications.
Cost of Goods Sold
Our
cost of goods sold is related to our direct costs associated with
the development and shipment of our thermoacoustic imaging systems
placed in pre-clinical settings.
Research and Development Expenses
Our
research and development expenses primarily include wages, fees and
equipment for the development of our TAEUS technology platform and
our proposed applications. Additionally, we incur certain costs
associated with the protection of our products and inventions
through a combination of patents, licenses, applications and
disclosures.
Sales and Marketing Expenses
Sales
and marketing expenses consist primarily of advertising, marketing
and consulting expenses and headcount. Currently, our marketing
efforts for our pre-clinical business are through distributors in
China, the European Union, Australia, Korea and the United Kingdom,
our website, and attendance of key industry meetings. In connection
with the commercialization of our TAEUS applications, we expect to
build a small sales and marketing team to train and support global
ultrasound distributors, as well as execute traditional marketing
activities such as promotional materials, electronic media and
participation in industry conferences.
General and Administrative Expenses
General
and administrative expenses consist primarily of salaries and
related expenses for our management and personnel, and professional
fees, such as accounting, consulting and legal.
Critical
Accounting Policies and Estimates
Use of Estimates
The
preparation of the financial statements in conformity with
accounting principles generally accepted in the United States
requires management to make estimates and assumptions that affect
the reported amounts of assets and liabilities, and disclosure of
contingent liabilities at the date of the financial statements and
the reported amounts of expenses during the reporting period.
Actual results could differ from those estimates.
Management makes
estimates that affect certain accounts including deferred income
tax assets, accrued expenses, fair value of equity instruments and
reserves for any other commitments or contingencies. Any
adjustments applied to estimates are recognized in the period in
which such adjustments are determined.
Share-based Compensation
Our 2016 Omnibus
Incentive Plan, or our Incentive Plan, which has been approved by
our board of directors, permits the grant of share options and
shares to our employees, consultants and non-employee members of
our board of directors for up to an estimated
1,496,448 shares of common stock (to be an amount
equal to 18% of our total issued and outstanding shares of common
stock following the completion of this offering on a fully diluted
basis, including shares issuable under our Incentive Plan, shares
issuable upon the conversion into shares of common stock of all
outstanding securities that are convertible by their terms into
shares of common stock and the exercise of all options and warrants
exercisable for shares of common stock, and shares issuable upon
exercise in full of the underwriters’ warrants to purchase
shares of common stock and the underwriters’ over-allotment
option). We record share-based compensation in accordance with the
provisions of the Share-based Compensation Topic of the FASB
Codification. The guidance requires the use of option-pricing
models that require the input of highly subjective assumptions,
including the option’s expected life and the price volatility
of the underlying stock. The fair value of each option grant is
estimated on the date of grant using the Black-Scholes option
valuation model, and the resulting charge is expensed using the
straight-line attribution method over the vesting period. We have
elected to use the calculated value method to account for the
options we issued in 2014. A nonpublic entity that is unable to
estimate the expected volatility of the price of its underlying
shares may measure awards based on a “calculated
value,” which substitutes the volatility of appropriate
public companies (representative of the company’s size and
industry) as a bench mark for the volatility of the entity’s
own share price. There is no existing active market for our common
stock. We have used the historical closing values of these
companies to estimate volatility, which was calculated to be
90%.
40
Stock
compensation expense recognized during the period is based on the
value of share-based awards that were expected to vest during the
period adjusted for estimated forfeitures. The estimated fair value
of grants of stock options and warrants to non-employees is charged
to expense, if applicable, in the financial
statements.
Recent Accounting Pronouncements
See
Footnote 2 of the financial statements for a discussion of recently
issued accounting standards.
Recent
Financial Results
We have not yet
completed preparation of financial statements for the year ended
December 31, 2016, but based on preliminary data available to us,
we expect to report total revenue of approximately $515,000 for the
quarter and year ended December 31, 2016.
This preliminary
financial information is based on current expectations and is
subject to year-end closing adjustments. Actual results may differ.
This preliminary financial data has been prepared by and is the
responsibility of our management. RBSM LLP has not audited,
reviewed, compiled or performed any procedures with respect to this
preliminary financial data, and accordingly, RBSM LLP does not
express an opinion or any other form of assurance with respect
thereto. For a discussion of certain risks that may cause our
results of operations to differ from our expectations, see
“Risk Factors” elsewhere in this
prospectus.
Results
of Operations
Years ended December 31, 2015 and 2014
Revenues
Revenues were
$1,410,064 for the year ended December 31, 2015, an increase of
$850,709, or 152%, as compared to $559,355 for the year ended
December 31, 2014. The increase in revenue was due to increased
unit sales of our Nexus 128 system. As described above
under “Recent
Financial Results,”
we expect revenues for 2016 to decline to
$515,000 compared to revenues in 2015 of
$1.4 million as we focused our limited resources on
developing our TAEUS applications.
Cost of Goods Sold
Cost of
goods sold was $666,233 for the year ended December
31, 2015, an increase of $407,088, or
157%, as compared to $259,145 for the
year ended December 31, 2014. Cost of goods sold increased as a
result of an increase in units sold during the year. Our gross
margin was approximately 53% for the year ended
December 31, 2015, compared to 54% for the prior year.
The slight decrease in gross margin resulted from
a slight increase in the cost of certain parts used to
assemble the systems sold. We expect increased margins in
2016 due to increased retail prices.
Research and Development
Research and
development expenses were $1,038,878 for the year ended December
31, 2015, as compared to $873,167 for the year ended December 31,
2014, an increase of $165,711, or 19%. The increase was primarily
due to additional personnel dedicated to our research and
development activities. We expect to continue to dedicate a
substantial amount of our resources to research and development
related to the development of our TAEUS applications.
Sales and Marketing
Sales
and marketing expenses were $50,635 for the year ended December 31,
2015, as compared to $56,298 for the year ended December 31, 2014,
a decrease of $5,663, or 10%. The decrease was primarily due to
reduced commissions paid on the sale of our Nexus 128 system.
Following completion of the offering we expect to dedicate
additional resources to our sales efforts for both our Nexus 128
product line and our TAEUS applications.
41
General and Administrative
Our
general and administrative expenses for the year ended December 31,
2015 were $1,222,888, an increase of
$325,962, or 36%, compared to $896,926
for the year ended December 31, 2014. Our wage and related expenses for the
year ended December 31, 2015 were $774,779, an increase of
$302,464, or 69%, compared to $445,315 for the year ended December
31, 2014. The increase in wages and related expenses was due
primarily to increased headcount related to the development of our
TAEUS technology platform. Wage and related expenses in 2015
included $236,364 of stock compensation expense related to the
issuance and vesting of common stock, options and warrants issued
for services, compared to $188,287 of stock compensation expense
for 2014. We expect that our general and administrative expenses
will increase significantly as a result of our becoming a public
company.
Net loss
As a
result of the foregoing, for the year ended December 31, 2015, we
recorded a net loss of $2,279,204 compared to a net
loss of $2,180,878 for the year ended December 31,
2014.
Three Months ended September 30, 2016 and 2015
Revenues
We had
no revenue for the three months ended September 30, 2016, as
compared to $326,085 for the three months ended September 30, 2015.
Revenues for 2016 declined as compared to our revenues for 2015 due
to our limited resources and our decision to focus those resources
on developing our TAEUS applications. As described
under “Recent
Financial Results,”
we expect sales of approximately $515,000 from our
Nexus 128 product during the fourth quarter of 2016.
Cost of Goods Sold
Cost of
goods sold was $189,535 for the three months ended
September 30, 2015. Gross margin was approximately 42%
for the three months ended September 30, 2015.
Research and Development
Research and
development expenses were $139,540 for the three months ended
September 30, 2016, as compared to $265,025 for the three months
ended September 30, 2015, a decrease of $125,485, or 47%. These
costs include primary wages, fees and equipment for the development
of our TAEUS product line. Research and development expenses
declined due to a reduction in spending to conserve cash. Following
completion of the offering we expect that our research and
development expenses will increase significantly due to our efforts
to develop our TAEUS applications.
Sales and Marketing
Sales
and marketing expenses were $16,040 for the three months ended
September 30, 2016, as compared to $20,743 for the three months
ended September 30, 2015, a decrease of $4,703, or 23%. The
decrease was primarily due to reduced commissions paid on the sale
of our Nexus 128 system. Currently our marketing efforts for our
pre-clinical business are through distributors in China, the
European Union, Australia and the United Kingdom, our website and
attendance of key industry meetings. Our future clinical business
will involve hiring and training additional staff to support our
sales efforts. As we seek to complete the development and
commercialization of our TAEUS applications, we intend to build a
small sales and marketing team to train and support global
ultrasound distributors, as well as execute traditional marketing
activities such as promotional materials, electronic media and
participation in industry conferences.
42
General and Administrative
Our
general and administrative expenses for the three months ended
September 30, 2016 were $451,531, an increase of
$55,770, or 14%, compared to $395,761 for
the three months ended September 30, 2015. This increase was due
primarily to increased professional fees associated with business
advisory consulting services. Our wage and related expenses for the
three months ended September 30, 2016 were $231,918, compared to
$272,478 for the three months ended September 30, 2015. Wage and
related expenses in 2015 included $103,483 of stock compensation
expense related to the issuance and vesting of options, compared to
$115,125 of stock compensation expense for 2015. Our professional
fees for the three months ended September 30, 2016 were $149,454,
an increase of $88,976, or 147%, compared to $60,478 for the three
months ended September 30, 2015. We expect that our general and
administrative expenses will increase significantly as a result of
our becoming a public company.
Net loss
As a
result of the foregoing, for the three months ended September 30,
2016, we recorded a net loss of $979,899 compared to a
net loss of $540,987 for the three months ended
September 30, 2015.
Nine Months ended September 30, 2016 and 2015
Revenues
We had
no revenue for the nine months ended September 30, 2016, as
compared to $1,155,065 for the nine months ended September 30, 2015
due to our limited resources and our decision to focus those
resources on developing our TAEUS applications. As described
above under “Recent
Financial Results,”
we expect sales of approximately $515,000 from our
Nexus 128 product during the fourth quarter of 2016.
Cost of Goods Sold
Cost of
goods sold was $539,809 for the nine months ended
September 30, 2015. Gross margin was approximately
53.3% for the nine months ended September 30,
2015.
Research and Development
Research and
development expenses were $338,417 for the nine months ended
September 30, 2016, as compared to $768,215 for the nine months
ended September 30, 2015, a decrease of $429,798, or 56%. These
costs include primary wages, fees and equipment for the development
of our TAEUS product line. Research and development expenses
declined due to a reduction in spending to conserve cash. Following
completion of the offering we expect that our research and
development expenses will increase significantly due to our efforts
to develop our TAEUS applications.
Sales and Marketing
Sales
and marketing expenses were $26,197 for the nine months ended
September 30, 2016, as compared to $41,422 for the nine months
ended September 30, 2015, a decrease of $15,225, or 37%. The
decrease was primarily due to reduced commissions paid on the sale
of our Nexus 128 system. Currently our marketing efforts for our
pre-clinical business are through distributors in China, the
European Union, Australia and the United Kingdom, our website and
attendance of key industry meetings. Our future clinical business
will involve hiring and training additional staff to support our
sales efforts. As we seek to complete the development and
commercialization of our TAEUS applications, we intend to build a
small sales and marketing team to train and support global
ultrasound distributors, as well as execute traditional marketing
activities such as promotional materials, electronic media and
participation in industry conferences.
43
General and Administrative
Our
general and administrative expenses for the nine months ended
September 30, 2016 were $1,110,264, an increase of
$191,490, or 21%, compared to $918,774
for the nine months ended September 30, 2015. Our wage and related expenses for the
nine months ended September 30, 2016 were $549,430, compared to
$530,918 for the nine months ended September 30, 2015. Wage and
related expenses in 2016 included $172,723 of stock compensation
expense related to the issuance and vesting of options, compared to
$140,569 of stock compensation expense for 2015. Our professional
fees for the nine months ended September 30, 2016 were $384,399, an
increase of $194,849, or 103%, compared to $189,550 for the nine
months ended September 30, 2015. We expect that our general and
administrative expenses will increase significantly as a result of
our becoming a public company.
Net loss
As a
result of the foregoing, for the nine months ended September 30,
2016, we recorded a net loss of $2,088,491 compared to
a net loss of $1,820,471 for the nine months ended
September 30, 2015.
Liquidity
and Capital Resources
To
date, we have generated only limited revenues from sales of our
Nexus 128 system. We have funded our operations to date through the
private sale of our equity securities. As of September 30, 2016, we
had $75,543 in cash.
The
consolidated financial statements included in this prospectus have
been prepared assuming the Company will continue as a going
concern, which contemplates the realization of assets and the
settlement of liabilities and commitments in the normal course of
business. As reflected in the accompanying consolidated financial
statements, during the year ended December 31, 2015, the Company
incurred a net loss of $2,279,204, used cash in
operations of $842,727, and at December 31, 2015, the
Company had a stockholders’ equity of $250,300.
Also, as reflected in the accompanying consolidated financial
statements, during the nine months ended September 30, 2016, the
Company incurred net loss of $2,088,491, and used cash
in operations of $1,385,033. At September 30, 2016, the Company had
a stockholders’ deficit of $241,196. These and
other factors raise substantial doubt about the Company’s
ability to continue as a going concern. The financial statements do
not include any adjustments that might be necessary should the
Company be unable to continue as a going concern.
Years ended December 31, 2015 and 2014
Operating Activities
During
the year ended December 31, 2015, we used $842,727 of
cash in operating activities primarily as a result of our net loss
of $2,279,204, offset by additional warrants issued
during the warrant exchange program of $686,343, share-based
compensation of $309,837, net changes in operating assets and
liabilities of $323,272, $72,225 in
depreciation and amortization expenses, and loss on warrant
exercise of $25,000.
During
the year ended December 31, 2014, we used $1,397,229
of cash in operating activities primarily as a result of our net
loss of $2,180,878, offset in part by a loss on common
stock issued for note conversion of $639,178, $259,493 in non-cash
stock compensation expense, net changes in operating assets and
liabilities of $152,294, and $37,242 in depreciation
and amortization expense.
44
Investing
Activities
During
the year ended December 31, 2015, we acquired equipment in the
aggregate amount of $133,811. During the year ended
December 31, 2014, we acquired equipment in the aggregate amount of
$275,371.
Financing
Activities
During
the year ended December 31, 2015, financing activities provided
$839,224 from common stock issued for cash. Financing activities
provided $1,751,015 to us during the year ended December 31, 2014
from the issuance of common stock.
Nine Months ended
September 30, 2016 and 2015
Operating
Activities
During
the nine months ended September 30, 2016, we used $1,385,033 of
cash in operating activities primarily as a result of our net loss
of $2,088,491, offset by amortization of discount of
convertible debt of $561,812, share-based compensation of $199,723,
$48,613 in depreciation and amortization expenses,
additional warrants issued during the warrant exchange program of
$5,823, and net changes in operating assets and liabilities of
$(112,513).
During
the nine months ended September 30, 2015, we used
$875,363 of cash in operating activities primarily as
a result of our net loss of $1,820,471, offset in part
by additional warrants issued during the warrant exchange program
of $686,343, net changes in operating assets and liabilities of
$(144,155), $55,841 in depreciation and
amortization expense, $25,444 in non-cash stock compensation
expense, and loss on warrant exercise of $25,000.
Investing
Activities
During
the nine months ended September 30, 2015, we acquired equipment in
the aggregate amount of $133,811. There were no
investing activities for the nine months ended September 30,
2016.
Financing
Activities
During
the nine months ended September 30, 2016, financing activities
provided $1,441,448 including $5,000 from common stock issued for
cash, $50,000 in proceeds from notes payable, and $1,386,448 in
proceeds from issuance of convertible notes. Financing activities
provided $955,617 to us during the nine months ended September 30,
2015 from the issuance of common stock.
Funding Requirements
We have
not completed development of our TAEUS technology platform
applications. We expect to continue to incur significant expenses
for the foreseeable future. We anticipate that our expenses will
increase substantially as we:
●
advance
the engineering design and development of our NAFLD TAEUS
application;
●
prepare
applications required for marketing approval of our NAFLD TAEUS
application in the European Union and the United
States;
●
seek to
hire a small internal marketing team to engage and support channel
partners and clinical customers for our NAFLD TAEUS
application;
●
commence marketing
of our NAFLD TAEUS application;
●
advance
development of our other TAEUS applications; and
●
add
operational, financial and management information systems and
personnel, including personnel to support our product development,
planned commercialization efforts and our operation as a public
company.
45
We
believe that the net proceeds of this offering, together with our
existing cash, will be sufficient for us fund the development,
regulatory approval and initial commercialization of our NAFLD
TAEUS application in the European Union and the United States. It
is possible that we will not achieve the progress that we expect
because the actual costs and timing of completing the development
and regulatory approvals for a new medical device are difficult to
predict and are subject to substantial risks and delays. We have no
committed external sources of funds. The expected net proceeds of
this offering are not expected to be sufficient for us to complete
the commercialization of our NAFLD TAEUS application or to complete
the development of any other TAEUS application and we will need to
raise substantial additional capital for those purposes. As a
result, we will need to finance our future cash needs through
public or private equity offerings, debt financings, corporate
collaboration and licensing arrangements or other financing
alternatives. Our forecast of the period of time through which our
financial resources will be adequate to support our operations is a
forward-looking statement and involves risks and uncertainties, and
actual results could vary as a result of a number of factors,
including the factors discussed in the section entitled “Risk
Factors” and elsewhere in this prospectus. We have based this
estimate on assumptions that may prove to be wrong, and we could
utilize our available capital resources sooner than we currently
expect.
Until
we can generate a sufficient amount of revenue from our TAEUS
platform applications, if ever, we expect to finance future cash
needs through public or private equity offerings, debt financings
or corporate collaborations and licensing arrangements. Additional
funds may not be available when we need them on terms that are
acceptable to us, or at all. If adequate funds are not available,
we may be required to delay, reduce the scope of or eliminate one
or more of our research or development programs or our
commercialization efforts. To the extent that we raise additional
funds by issuing equity securities, our stockholders may experience
additional dilution, and debt financing, if available, may involve
restrictive covenants. To the extent that we raise additional funds
through collaborations and licensing arrangements, it may be
necessary to relinquish some rights to our technologies or
applications or grant licenses on terms that may not be favorable
to us. We may seek to access the public or private capital markets
whenever conditions are favorable, even if we do not have an
immediate need for additional capital at that time.
Off
Balance
Sheet Transactions
We do
not have any off balance sheet transactions.
46
Overview
We
have
commercialized an enhanced ultrasound technology for the
pre-clinical research market and are leveraging that expertise to
develop technology for increasing the capabilities of clinical
diagnostic ultrasound to broaden patient access to the safe
diagnosis and treatment of a number of significant medical
conditions in circumstances where expensive X-ray computed
tomography, or CT, and magnetic resonance imaging, or MRI,
technology is unavailable or impractical.
Since
2010, we have marketed and sold our Nexus 128 system, which
combines light-based thermoacoustics and ultrasound, to address the
imaging needs of researchers studying disease models in
pre-clinical applications. Building on our expertise in
thermoacoustics, we have developed a next-generation technology
platform — Thermo Acoustic Enhanced Ultrasound, or TAEUS
— which is intended to enhance the capability of clinical
ultrasound technology and support the diagnosis and treatment of a
number of significant medical conditions that require the use of
expensive CT or MRI imaging or where imaging is not practical using
existing technology. We believe that our TAEUS technology, which
can be used with existing ultrasound equipment and incorporated
into next-generation ultrasound systems, has the potential to make
advanced imaging available in certain applications to a wider range
of patients on a more cost-effective basis than is possible using
existing CT and MRI technology. We expect to continue to sell our
Nexus 128 system to maintain a base level of revenue, but believe
the market potential for our clinical systems is much
higher.
Diagnostic
Imaging Technologies
Diagnostic imaging
technologies such as CT, MRI and ultrasound allow physicians to
look inside a person’s body to guide treatment or gather
information about medical conditions such as broken bones, cancers,
signs of heart disease or internal bleeding. The type of imaging
technology a physician uses depends on a patient’s symptoms
and the part of the body being examined. CT technology is well
suited for viewing bone injuries, diagnosing lung and chest
problems, and detecting cancers. MRI technology excels at examining
soft tissue in ligament and tendon injuries, spinal cord injuries,
and brain tumors. CT scans can take as little as 5 minutes, while
an MRI scan can take up to 30 minutes.
Unfortunately,
while CT and MRI systems are versatile and create high quality
images, they are also expensive and not always accessible to
patients. A CT system costs approximately $1 million and an MRI
system can cost up to $3 million. CT and MRI systems are large and
can weigh several tons, typically requiring significant
modifications to existing healthcare facilities to safely handle
the load. Because of their size and weight, CT and MRI systems are
usually fixed-in-place at major medical facilities. As a result,
they are less accessible to primary care and rural clinics,
economically developing markets, and patient bedsides. There are
only approximately 64,000 CT systems and 32,000 MRI systems in the
world, approximately 50% of which are located in the U.S. and
Japan.
While
CT and MRI systems create high quality images, their use is not
always practical. For example, the diagnosis and treatment of the
estimated 1.4 billion patients suffering from Non-Alcoholic Fatty
Liver Disease, or NAFLD, requires ongoing surveillance of the
patients’ livers to assess the progression of the disease and
the efficacy of treatment. However, the use of CT and MRI systems
to perform that surveillance is impractical for a number of
reasons, including the high cost of the scan, the limited
availability of CT and MRI systems and the required use of contrast
agents, including those containing radioactive substances, that can
cause allergic reactions and reduced kidney functions. Patient
exposure to the ionizing radiation generated by a CT system must be
limited for safety reasons. Similarly, because of the strong
magnetic field created by an MRI machine, patients with metal joint
replacements or cardiac pacemakers cannot be imaged with an MRI
system.
Because
of CT and MRI’s limited availability and practical
limitations, a patient who would otherwise be a candidate for CT or
MRI scanning must often rely on less effective or less practical
methods. For example, MRI scans are not typically used to measure
tissue temperature during thermoablative (temperature based)
surgery. Instead, physicians use printed manufacturer guidelines to
time the thermal surgery or insert surgical temperature probes in
an attempt to guide treatment. As a result, the treatment is often
imprecise or comes with additional risks, such as
infection.
47
These
limitations have led to a decrease in the number of CT scans.
According to the American College of Radiology, the overall number
of CT scans performed in the United States under Medicare Part B
fell approximately 8% from 2009 to 2014. The decline in CT scans
has been accompanied by increased use of alternative scanning
technologies. The American College of Radiology reported that the
overall number of ultrasound scans performed in the United States
under Medicare Part B increased approximately 6% from 2009 to 2014.
During the same period MRI usage increased by 5%, but remains
significantly below the use of ultrasound technology, even in the
United States.
Ultrasound
Technology
An
ultrasound machine transmits sound waves, which bounce off tissues,
organs and blood in the body. The ultrasound machine captures these
echoes and uses them to create an image. Ultrasound technology
excels at imaging the structure of internal organs, muscles and
bone surfaces.
Ultrasound systems
are more broadly available to patients than either CT or MRI
systems. There are approximately 800,000 ultrasound systems
globally in use today. Ultrasound systems are relatively
inexpensive compared to CT and MRI systems, with smaller portable
ultrasound systems costing as little as $10,000 and new cart-based ultrasound systems
costing between $75,000 and
$125,000. Ultrasound systems are also more mobile than CT
and MRI systems and many are designed to be moved by an operator
from room to room, or closer to patients. Ultrasound technology
does not present the same safety concerns as CT and MRI technology,
since ultrasound does not emit ionizing radiation and ultrasound
contrast agents are considered to be generally safe.
Due to
its utility, cost-effectiveness and safety profile, ultrasound
imaging is frequently used in a physician’s examination room
or at a patient’s bedside as a first-line diagnostic tool,
which has resulted in an overall increase in the number of
ultrasound scans performed. According to the American College of
Radiology, the overall number of ultrasound scans performed in the
United States under Medicare Part B increased 6% from 2009 to 2014
(while CT exams declined 8% during the same period).
However,
ultrasound’s imaging capabilities are more limited compared
to CT and MRI technology. For example, ultrasound systems cannot
measure tissue temperature during thermal ablation surgery, or
quantify fat to diagnose early stage liver disease -- instances
where CT and MRI systems are commonly used.
Unmet
Need
We
believe that the limited availability of high-utility and
cost-effective imaging technology represents a significant unmet
medical need. We believe that expanding the capability of
ultrasound technology to perform more of the imaging tasks
presently available only on expensive CT and MRI systems will
satisfy this unmet need.
Our
Solution – Thermo-Acoustic Enhanced Ultrasound, or
TAEUS
Our
commercially available Nexus 128 system and our Thermo-Acoustic
Enhanced Ultrasound, or TAEUS technology, each use a pulsed energy
source – near-infrared light and radio-frequency, or RF,
respectively – to generate ultrasonic waves in tissue. These
waves are then detected with ultrasound equipment and used to
create high-contrast images using our proprietary algorithms.
Unlike conventional ultrasound, which creates images based on the
scattering properties of tissue, thermoacoustic imaging provides
tissue absorption maps of the pulsed energy, similar to those
generated by CT scans. Ultrasound is only utilized to transmit the
absorption signal to the imaging system outside of the
body.
48
Since
2010 we have marketed our Nexus 128 system to address the imaging
needs of researchers studying disease models in pre-clinical applications. The
Nexus 128 uses near-infrared light combined with ultrasound to
generate 3D images of tumors in laboratory mice. We believe the
Nexus 128 is the only commercially available fully 3D
thermoacoustic imaging system.
Sales
of the Nexus 128 system were approximately $560,000 in 2014
and $1.4 million in 2015. We have not yet completed
preparation of financial statements for the quarter and year ended
December 31, 2016, but, as described above in the section of this
prospectus entitled “Prospectus Summary—Recent
Financial Results,” based on preliminary data available to
us, we expect to report total revenues of approximately $515,000
for the quarter and year ended December 31, 2016, all of which
revenue was attributable to sales of our Nexus 128 system. Our
Nexus 128 system is used in a number of leading global
academic research centers, including Stanford University, The
University of Michigan, Shanghai Jiao Tong University, and Purdue
University.
While
our Nexus 128 system is suited for small animal research, the
near-infrared light energy used in our Nexus 128 system only
penetrates tissues up to 3cm, limiting its utility beyond
shallow-depth human dermatological or breast applications.
Additionally, blood-filled organs, such as the liver, absorb most
of the near-infrared light, making it difficult to generate an
accurate image.
Our
TAEUS Technology Platform
To
increase the utility of our thermoacoustic technology, in 2013 we
began to develop our TAEUS technology platform. Unlike the
near-infrared light pulses used in our Nexus 128 system, our TAEUS
technology uses RF pulses to stimulate tissues, using a small
fraction of the energy transmitted into the body during an MRI
scan. Using RF energy enables our TAEUS technology to penetrate
deep into tissue, enabling the imaging of human anatomy at depths
equivalent to those of conventional ultrasound. The RF pulses are
absorbed by tissue and converted into ultrasound signals, which are
detected by an external ultrasound receiver and a digital
acquisition system that is part of the TAEUS system. The detected
ultrasound is processed into images using our proprietary
algorithms and overlaid in real time onto conventional gray-scale
ultrasound images. An example of a TAEUS image overlay is shown
below:
Image below: Real-time ex-vivo bovine tissue temperature analysis
overlaid on traditional ultrasound image.

Our
RF-based thermoacoustics are not adversely affected by blood-filled
organs, enabling our TAEUS technology to be used in clinical liver
applications, among others.
After
approval, our TAEUS technology can be added as an accessory to
existing ultrasound systems, helping to improve clinical
decision-making on the front lines of patient care, without
requiring new clinical workflows or large capital investments. We
are also developing TAEUS for incorporation into new ultrasound
systems, primarily through our collaboration with GE Healthcare,
described more fully below.
We
believe that our TAEUS technology has the potential to add a number
of new capabilities to conventional ultrasound and thereby enhance
the utility of both existing and new ultrasound systems and extend
the use of ultrasound technology to circumstances that either
require the use of expensive CT or MRI imaging systems or where
imaging is not practical using existing technology.
To
demonstrate the capabilities of our TAEUS platform, we have
conducted various internal ex-vivo laboratory experiments and have
also conducted limited internal in-vivo large animal studies.
However, we have not yet conducted any human studies and these
capabilities are not supported by clinical data that we have
gathered in pursuit of obtaining regulatory approvals or that was
subject to regulatory oversight and guidance. In our ex-vivo
and in-vivo testing, we have demonstrated that the TAEUS platform
has the following capabilities and potential clinical
applications:
49
●
Tissue Composition:
Our TAEUS technology enables ultrasound to distinguish fat from
lean tissue. This capability would enable the use of TAEUS-enhanced
ultrasound for the early identification, staging and monitoring of
NAFLD, a precursor to liver fibrosis, cirrhosis and liver
cancer.
●
Temperature
Monitoring: Our TAEUS technology enables traditional ultrasound to
visualize changes in tissue temperature, in real time. This
capability would enable the use of TAEUS-enhanced ultrasound to
guide thermoablative therapy, which uses heat or cold to remove
tissue, such as in the treatment of cardiac atrial fibrillation, or
removal of cancerous liver and kidney lesions, with greater
accuracy.
●
Vascular Imaging:
Our TAEUS technology enables ultrasound to view blood vessels from
any angle, using only a saline solution contrasting agent, unlike
Doppler ultrasound, which requires precise viewing angles. This
capability would enable the use of TAEUS-enhanced ultrasound to
easily identify arterial plaque or malformed vessels.
●
Tissue Perfusion:
Our TAEUS technology enables ultrasound to image blood flow at the
capillary level in a region, organ or tissue. This capability could
be used to assist physicians in characterizing microvasculature
fluid flows symptomatic of damaged tissue, such as internal
bleeding from trauma, or diseased tissue, such as certain
cancers.
Because
of the large number of traditional ultrasound systems currently in
global use, we are first developing our TAEUS technology for sale
as an aftermarket accessory that works with existing ultrasound
systems. Because our TAEUS technology is designed to enhance the
utility of, not replace, conventional ultrasound, we believe
healthcare providers will be able to increase the utilization of,
and generate new revenue from, their existing ultrasound systems
once we obtain required regulatory approval for specific
applications. Based on our design work
and our understanding of the ultrasound accessory market, we
intend to price our initial
NAFLD TAEUS application at a price point approximating one-half of the price of a
new cart-based ultrasound system, or approximately $40,000 to
$50,000, which should enable purchasers to recoup their investment
in less than one year by performing a relatively small number of
additional ultrasound procedures. We further believe that
clinicians will be attracted to our technology because it will
enable them to perform more procedures with their existing
ultrasound equipment, thereby retaining more imaging patients in
their clinics rather than referring patients out to a regional
medical center for a CT or MRI scan.
Endra’s
first clinical product will interface with a conventional
ultrasound scanner, utilizing the scanner’s B-mode imaging to
guide the selected region for assessment of liver fat content. The
following sub-systems will comprise Endra’s first generation
product.
Radio frequency (RF) source and computer:
The
RF source consists of a low power waveform generator and an
amplifier. Together, these components provide the characteristic
pulses required to excite thermoacoustic signals in tissue. The
computer provides processing capability to both utilize the
conventional ultrasound data for navigation to the measurement site
of interest, and the calculations required to convert digitized
thermoacoustic signals to measurements of fat in liver tissue. The
entire sub-system is expected to be approximately the size of a
desk-side computer. The entire sub-system will reside in a single
enclosure, on wheels, and sit adjacent to the ultrasound imaging
system.
Specialized Transducer:
A
single channel ‘receive only’ ultrasound transducer is
specifically designed and optimized for thermoacoustic imaging. The
transducer sub-system will detect thermoacoustic signals excited by
the RF source within the liver. The transducer assembly includes
electronics for signal amplification, digitization, and signal
processing. The specialized transducer will attach to the
conventional ultrasound probe used for liver imaging.
RF Applicator:
The
RF applicator transmits pulses of energy, provided by the RF
source, into tissue. The applicator contacts the patient’s
skin in proximity of the target region for
measurement.
A
second generation product is expected to provide two dimensional
imaging with a transducer composed of multiple receive elements.
The RF source and applicator will be similar to those in the first
generation product but the multi-element transducer will allow for
multiple applications including: reading tissue composition,
temperature, vascular flow, tissue perfusion, and other potential
applications. Ultimately, we expect our technology will be
incorporated into conventional ultrasound systems and our business
model will transition from producing stand-alone systems to
licensing our technology, IP and specialized components to
ultrasound OEMs. Existing ultrasound equipment already includes
power supplies, computation, high speed electronics, and ultrasound
transducers, which may be leveraged by our thermoacoustic imaging
applications. The RF source and applicator are the principal
hardware components that will be added to OEM ultrasound systems
for the OEM fully integrated form of our product.
We are following a model that mirrors the approach used by
companies in the past to introduce new ultrasound imaging
capabilities to existing conventional ultrasound scanners. Color
Doppler, elastography, 3-D imaging, and high channel count systems
were all introduced by new companies (not already involved in
conventional ultrasound imaging). Historically, ultrasound imaging
has grown through the introduction of unique technology and
capabilities that expanded the applications and use of clinical
ultrasound in a form that often added separate hardware to existing
ultrasound systems. Ultimately, as these new technologies gained
acceptance in the marketplace they were incorporated into OEM
designed and built systems that were sold by the leading ultrasound
imaging vendors.
50
Image:
Illustration of a typical cart-based ultrasound system
(left) with our TAEUS technology depicted to the
right.
Ultrasound
Market
Sales
of ultrasound diagnostic equipment were approximately $4 billion
globally in 2014 and are expected to grow at approximately 4.4%
annually. There are approximately 800,000 installed systems
generating over 400 million annual diagnostic ultrasound procedures
globally. Additionally, an estimated 30,000 to 50,000 new and
replacement systems are sold into the market each year. These
numbers include both portable and cart-based ultrasound systems,
and cover all types of diagnostic ultrasound procedures, including
systems intended for cardiology, prenatal and abdominal use. We do
not intend to address low-cost, portable ultrasound systems and
systems focused on applications, such as prenatal care, where we
believe our TAEUS technology will not substantially impact patient
care. Accordingly, we define our addressable market for one or more
of our TAEUS applications at approximately 300,000 cart-based
ultrasound systems currently in use throughout the
world.
We
believe that demand for ultrasound systems is driven primarily by
the following factors:
●
Population growth
and age demographics that increase the demand for diagnostic
screening for cancer, cardiology, and prenatal
applications.
●
Economic
development broadening investment in healthcare in previously
underserved markets such as China and Latin America, where
ultrasound technology has significant appeal due to its price point
and flexibility at point-of-care.
●
Expanding
ultrasound applications and improving image quality that drive
demand for new ultrasound technologies, such as software
enhancements, bi-axial probes, and dedicated single application
systems.
●
Positive insurance
reimbursement rate trends for ultrasound diagnostics due to the
technology’s safety and cost-effectiveness.
51
Potential
Clinical Applications for our TAEUS Technology
Early Diagnosis and Monitoring of Non-Alcoholic Fatty Liver
Disease, or NAFLD
Our
first TAEUS platform application will focus on quantifying fat in
the liver and stage progression of NAFLD which, untreated, can
progress to Non-Alcoholic Steato-Hepatitis, or NASH, cirrhosis and
liver cancer. In 2011, over 1.4 billion people were affected by
NAFLD/NASH. The World Gastroenterology Organisation considers
NAFLD/NASH a global pandemic affecting rich and poor countries
alike. Obesity, hepatitis, and diabetes are leading contributors to
the development of NAFLD.
Untreated, an
estimated 20% of NAFLD cases progress to NASH, a condition in which
liver fat causes inflammation and decreased liver function,
resulting in fatigue, weight loss, muscle pain and abdominal
pain.
Approximately 25%
of NASH cases progress to liver cirrhosis, in which liver
inflammation causes scar tissue which eventually prevents the liver
from functioning properly. The scar tissue blocks the flow of blood
through the liver and slows the processing of nutrients, hormones,
drugs, and naturally produced toxins. It also slows the production
of proteins and other substances made by the liver. Once a patient
develops cirrhosis of the liver, the only life-saving therapy is a
liver transplant. Additionally, cirrhosis patients may develop
liver cancer. In 2015, the World Health Organization ranked liver
cancer as the second highest cause of cancer death, after lung
cancer, killing 745,000 people annually. Because of the increased
incidence of obesity, hepatitis and diabetes throughout the world,
NAFLD has become the most common chronic liver disease and an
important cause of cirrhosis and liver cancer
worldwide.
Despite
the increased incidence of NAFLD and its role in the development of
NASH, cirrhosis and liver cancer, we believe that no low-cost,
accurate and safe method exists for measuring fat in the liver.
Current liver enzyme blood tests are indicative, but cannot
reliably confirm early stage NAFLD or NASH, and liver enzyme levels
are normal in a large percentage of patients with NAFLD. Existing
ultrasound technology can only measure fat qualitatively in the
liver at moderate to severe levels, typically greater than 30%
liver fat, and ultrasound has low accuracy when used on obese
patients. While early stage NAFLD and NASH can be confirmed by an
MRI scan, an MRI scan is expensive, and MRI systems are not widely
available or practical for many patients. A surgical biopsy can be
used to confirm NAFLD and NASH, but is also expensive, involves a
painful procedure and exposes patients to the risk of infection.
Furthermore, MRIs and surgical biopsies are impractical for
repeated screening and monitoring of liver disease. We believe
these limitations negatively impact the diagnosis and treatment of
patients with NAFLD.
Patients
diagnosed with NAFLD and related liver diseases are typically
treated with therapies such as statins, insulin sensitizers and
other compounds and are encouraged to adopt lifestyle changes to
improve their overall health.
A
significant number of pharmaceutical compounds are in development
by companies such as Bristol-Myers Squibb Company, Intercept
Pharmaceuticals, Inc., Gilead Sciences, Inc., Genfit SA, Galectin
Therapeutics Inc., Conatus Pharmaceuticals Inc., NuSirt Sciences
Inc., Tobira Therapeutics, Inc. and Immuron Limited.
Billions of dollars
are spent annually on the diagnosis and treatment of NAFLD and
related liver diseases. Identification and staging of NAFLD is
central to determining the course of treatment. In addition,
patients receiving treatment for NAFLD-spectrum liver diseases must
continue to be monitored to assess disease progression and the
efficacy of treatment. Because of the high cost and limited global
availability, CT and MRI technology is not typically used for this
function.
52
We
believe our TAEUS technology will enable primary care physicians,
radiologists and hepatologists to diagnose NAFLD earlier and
monitor patients with NAFLD-spectrum liver diseases more accurately
and cost-effectively than is possible with existing
technology.
Image below: Ex-vivo TAEUS tissue composition analysis overlaid on
traditional ultrasound image.
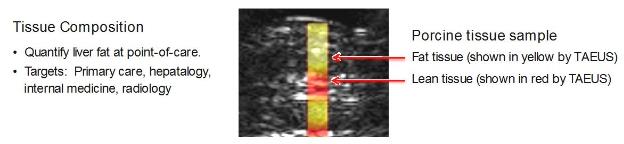
Temperature Monitoring of Thermoablative Surgery
We also
intend to develop a TAEUS platform application to guide thermal
ablation surgery, such as in the treatment of cardiac atrial
fibrillation, chronic pain and lesions of the liver, thyroid,
kidneys and other soft tissues. We plan to target clinical users of
thermoablative technology, including interventional radiologists,
cardiologists, gynecologists and surgical oncologists.
Thermoablation
involves the use of heat or cold to remove malfunctioning or
diseased tissue in surgical oncology, cardiology, neurology,
gynecology, and urology applications. Thermoablative technologies
include RF, microwave, laser and cryogenic ablation. The worldwide
market for RF surgical ablation procedures alone was estimated in
2015 to be $3.7 billion per annum, generating over 5 million annual
RF ablation procedures and growing at approximately 18% annually.
We believe that the growth of this market is driven primarily by
the aging global population requiring more cardiac and cancer
procedures, as well as the relative ease-of-use and low cost of
thermoablative technologies when compared to open
surgery.
However,
RF and other thermoablative surgery technologies pose risks,
including under-treatment of diseased tissue and unintended thermal
damage to areas outside the treatment area. For example, it has
been reported that patients receiving RF ablation of liver tumors
have experienced thermal injury to the diaphragm, gallbladder, bile
ducts and gastrointestinal tract, some of which have resulted in
patient deaths.
Clinicians
must rely on printed manufacturer guidelines to plan procedures using thermal ablation
technologies or, when available, monitor tissue temperature changes
in real-time with MRI imaging or surgical temperature probes. We
believe these existing methods either lack real-time precision or
are impractical due to cost, poor availability and other
factors.
We
believe that the ability to visualize changes in tissue temperature
in real time could potentially enhance the effectiveness and safety
of thermoablation therapies and that our TAEUS technology platform
combined with traditional ultrasound has the potential to guide
thermoablation surgery more cost-effectively and more accurately
than existing methods.
53
Image below: Real-time ex-vivo TAEUS tissue temperature analysis
overlaid on traditional ultrasound image.
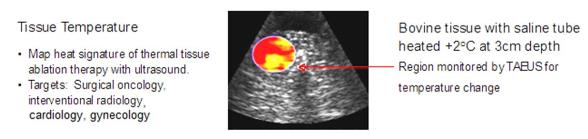
Vascular Imaging
We
believe that our TAEUS technology can be used to image blood
vessels and distinguish them from the surrounding tissue. In
addition to our NAFLD and thermoablation applications, we intend to
develop a cardiovascular application based on our TAEUS technology
that, with the use of a standard saline contrast agent, can enable
existing ultrasound systems to perform a number of cardiovascular
diagnostic functions, such as identifying arterial plaque or
blocked or malformed vessels, as well as safely guiding biopsies
away from vital vasculature.
Conventional
ultrasound imaging systems use Doppler imaging in a variety of
vascular applications. Doppler ultrasound, which images the
velocity of blood, is effective in larger vessels and regions where
blood velocity is high. However, Doppler ultrasound is not
sufficiently sensitive for use in very small vessels or in vascular
imaging applications where blood velocities are very low. For these
applications, contrast enhanced CT and MRI angiography is used
which requires the patient to be injected with a contrast agent,
iodinated compounds and gadolinium, respectively. Contrast-enhanced
CT and MRI scans both require referral for examination after
initial screening with ultrasound and carry risks associated with
their respective contrast agents. We believe that our TAEUS
platform application has the potential to offer the advantages of
CT and MR contrast enhanced imaging at the point of care using only
a safe electrolyte solution as the contrast agent.
Tissue Perfusion or “Leakiness”
We
believe that our TAEUS technology can be used to image tissue
perfusion, or the absorption of fluids into an organ or tissue. We
intend to develop an application for our TAEUS platform that would
enable ultrasound detection of microvasculature fluid flows
symptomatic of tissue compromised by trauma or
disease.
When a
person’s body is affected by disease or trauma, blood and
other fluids may leak from damaged tissues in subtle ways.
Traditional ultrasound cannot effectively image these disruptions
in microvascular permeability, but we believe ultrasound combined
with our TAEUS technology can.
We
believe that using our TAEUS technology physicians will be able to
quickly and clearly see tissue compromised by disease, such as
cancer, or trauma, especially with the use of a standard saline
contrast agent, when CT or MRI is not readily
available.
Collaboration
with GE Healthcare
In
April 2016, we entered into a Collaborative Research Agreement with
General Electric Company, acting through its GE Healthcare business
unit and the GE Global Research Center, or GE Healthcare. Under the
terms of the agreement, GE Healthcare has agreed to assist us in
our efforts to commercialize our TAEUS technology for use in a
fatty liver application by, among other things, providing equipment
and technical advice, and facilitating introductions to GE
Healthcare clinical ultrasound customers. In return for this
assistance, we have agreed to afford GE Healthcare certain rights
of first offer with respect to manufacturing and licensing rights
for the target application. More specifically, we have agreed that,
prior to commercially releasing our TAEUS technology for a fatty
liver application, we will offer to negotiate an exclusive
ultrasound manufacturer relationship with GE Healthcare for a
period of at least one year of commercial sales. The commercial
sales would involve, within our sole discretion, either our company
commercially selling GE Healthcare ultrasound systems as the
exclusive ultrasound system with their TAEUS fatty liver
application embedded, or GE Healthcare being the exclusive
ultrasound manufacturer to sell ultrasound systems with the TAEUS
fatty liver application technology embedded.
54
The
agreement with GE Healthcare does not prevent us from selling our
TAEUS fatty liver application technology to distributors or
directly to non-manufacturer purchasers.
Additionally, the
agreement provides that prior to offering to license any of our
TAEUS fatty liver application intellectual property to a third
party, we will first offer to negotiate to license our TAEUS fatty
liver application intellectual property to GE
Healthcare.
Finally, we agreed
that prior to selling any equity interests in our company to a
healthcare device manufacturer, we will first offer to negotiate in
good faith to sell such equity interests to GE
Healthcare.
The
agreement has a term of one year and is subject to termination by
either party upon not less than 60 days’ notice.
Intellectual
Property
We rely
on a combination of patent, copyright, trademark and trade secret
laws and other agreements with employees and third parties to
establish and protect our proprietary intellectual property rights.
We require our officers, employees and consultants to enter into
standard agreements containing provisions requiring confidentiality
of proprietary information and assignment to us of all inventions
made during the course of their employment or consulting
relationship. We also enter into nondisclosure agreements with our
commercial counterparties and limit access to, and distribution of,
our proprietary information.
We are
committed to developing and protecting our intellectual property
and, where appropriate, filing patent applications to protect our
technology. Our issued and pending patents claims are directed at
the following areas related to our technology:
●
Methods to induce
and enhance thermoacoustic signal generation;
●
System
configurations, devices and novel hardware for transmission of RF
pulses into tissue and detection of acoustic signals;
●
Methods for
integrating our devices with existing conventional ultrasound
systems; and
●
Methods and
algorithms for signal processing, image formation and
analysis.
We
currently maintain a patent portfolio consisting of two patents
issued in foreign jurisdictions, eight patent applications pending
in the United States and five patent applications pending in
foreign jurisdictions. These patents and patent applications cover
certain innovations relating to contrast-enhanced imaging as well
as several aspects of fat imaging and fat quantitation in the liver
and other tissues.
In
addition, we have in-licensed five U.S. patents, three foreign
patents, one patent application pending in the United States and
two patent applications pending in foreign jurisdictions. These
patents protect a number of key design attributes that are specific
to our Nexus 128 product.
Each of
our patents generally has a term of 20 years from its respective
priority filing date. Among our issued patents, the first patents
are set to expire in 2018 and the last patents expire in
2031.
55
Sales and Marketing
We
currently market our Nexus 128 pre-clinical system through a small
internal marketing team and a global network of distributors in the
United Kingdom, the European Union, Australia, China and Korea. We
use our corporate website, sales materials and key industry
meetings to drive customer awareness, interest and trial of our
products.
We
currently do not have a sales and marketing team dedicated to our
TAEUS clinical applications. In parallel to securing all necessary
government marketing approvals, we intend to hire a small internal
marketing team to engage and support channel partners and clinical
customers. As we have done with our Nexus 128 system, we intend to
partner with several geographically-focused independent clinical
ultrasound equipment distributors to market and sell our TAEUS
applications. We believe that these distributors have existing
customer relationships, a strong knowledge of diagnostic imaging
technology and the capabilities to support the installation,
customer training and post-sale service of capital equipment and
software.
We also
intend to work with original equipment manufacturers, or OEMs, of
ultrasound and thermal ablation equipment to sell our TAEUS
applications alongside their own new systems and into their
existing installed base systems. We believe that these OEMs will
find our applications attractive as they will enable them to
generate additional revenue from their installed systems – as
they currently do with aftermarket accessory portfolios. We believe
our relationship with GE Healthcare will facilitate this
strategy.
Based
on our design work and our understanding of the ultrasound
accessory market, we intend to price our initial NAFLD TAEUS
application at a price point approximating one-half of the price of
a new cart-based ultrasound system, which should enable purchasers
to recoup their investment in less than one year by performing a
relatively small number of additional ultrasound
procedures.
Some of
our TAEUS offerings are expected to be implemented via a hardware
platform that can run multiple individual software applications
that we will offer TAEUS users for a one-time licensing fee,
enabling users to perform more procedures with their existing
ultrasound equipment and retaining more patients in their clinics
rather than referring them out to a regional imaging medical center
for a CT or MRI scan.
We also
intend to license our TAEUS technology to OEMs, such as GE
Healthcare, for incorporation in their new ultrasound
systems.
Manufacturing
We
assemble our Nexus 128 products from components provided to us by
third-party component suppliers and manufacturers. While many of
the components are off-the-shelf components available from multiple suppliers, our
proprietary receiver array is specially manufactured to our
specifications by one manufacturer. To date, we have not
experienced any component shortages. We do not have any long-term
supply or manufacturing agreements related to our Nexus 128
products and components are obtained on a purchase order basis when
required.
We
intend to contract with a medical device contract engineering firm
to perform the commercial product engineering for our NAFLD TAEUS
application, as well as any other application we decide to
commercialize. We expect that the selected contractor will have
quality systems and processes in place, commensurate with
productizing devices with CE mark certification that will meet FDA
requirements for approval. We believe that our contractor will have
the ability to provide product design, development and
documentation necessary to support a CE mark that will enable us to
sell the application in the European Union as a Class IIa medical
device once a final design has been developed and tested. We also
expect that this contractor, with support from a medical device
regulatory consulting firm, will lead the preparation of
documentation for regulatory approval submission both in the
European Union and in the United States. In order to foster
collaboration with and supervision of our contractor, we intend to
locate one or more of our employees at the medical device contract
engineering firm during the TAEUS application manufacturing
process. We have identified several medical device contract
engineering firms that have the capability to provide these
services. However, as of the date hereof, we have not entered into
a contract with any of them. We expect that our contract
manufacturers will either supply necessary components internally or
obtain them from third-party sources. At this time, we do not know
whether any components will be single sourced.
56
Regulatory
Approval Pathway
Each of our TAEUS
platform applications will require regulatory approvals before we
are able to sell or license the application. Based on certain
factors, such as the installed base of ultrasound systems,
availability of other imaging technologies, such as CT and MRI,
economic strength and applicable regulatory requirements, we intend
to seek initial approval of our applications for sale in the
European Union, followed by the United States and
China.
The
first TAEUS application we intend to commercialize is our NAFLD
TAEUS application. Our initial target market for this application
is the European Union. We believe that our NAFLD TAEUS application
will qualify for sale in the European Union as a Class IIa medical
device. As a result, we will be required to obtain a CE mark for
our NAFLD TAEUS application before we can sell the application in
the European Union. We have not yet initiated the process for
obtaining this CE mark. The first step we plan to take in this
regard is to contract with a medical device contract engineering
firm to perform the commercial product engineering for our NAFLD
TAEUS application, as well as any other application we decide to
commercialize. We expect that the selected contractor will have
quality systems and processes in place, commensurate with
productizing devices with CE mark and FDA certification and
approvals. We believe that our contractor will have the ability to
provide product design, development and documentation necessary to
support a CE mark that will enable us to sell the application in
the European Union as a Class IIa medical device once a final
design has been developed and tested. We also expect that this
contractor, with support from a medical device regulatory
consulting firm, will lead the preparation of documentation for
regulatory approval submission both in the European Union and in
the United States. We have identified several medical device
contract engineering firms that have the capability to provide
these services. However, as of the date hereof, we have not entered
into a contract with any of them. Existing regulations would not
require us to conduct a clinical trial to obtain a CE mark for this
application. Nonetheless, for commercial reasons and to support our
CE mark application we plan to conduct a limited (less than 10
person) trial to demonstrate our NAFLD TAEUS application’s
ability to distinguish fat from lean tissue. Based on our
understanding of applicable regulations and consultations with
medical device regulatory consulting firms and medical device
contract engineering firms, we expect that the development of our
NAFLD TAEUS application, including the receipt of the necessary CE
mark, will be complete approximately ten months after the
completion of this offering, and that we will use approximately
$1,000,000 of the net proceeds from this offering on such
activities. Additionally, to enhance our commercialization efforts
in the European Union, following receipt of such CE mark and
placement of initial systems with researchers and universities, we
plan to conduct one or more clinical studies to demonstrate this
product’s capabilities, and that we will use approximately
$700,000 of the net proceeds from this offering on such activities.
However, these estimates are subject to uncertainty and there can
be no assurance that these processes will not take longer or be
more costly than we expect. In 2012 the European Commission
proposed a new regulatory scheme that, if implemented, will impose
significant additional obligations on medical device companies.
Expected changes include stricter requirements for clinical
evidence and pre-market assessment of safety and performance, new
classifications to indicate risk levels, requirements for third
party testing by government accredited groups for some types of
medical devices, and tightened and streamlined quality management
system assessment procedures. It is anticipated that this new
regulatory scheme may be implemented prior to receipt of the CE
mark for our NAFLD TAEUS application but we believe that applicable
transition rules should allow us to avoid their application in that
case. However, such new rules could impose additional requirements,
such as a requirement to conduct clinical trials, on future CE mark
applications we make.
While
the process of obtaining a CE mark for our NAFLD TAEUS application
is underway, we also intend to prepare for submission to the U.S.
Food and Drug Administration, or the FDA, an application under the
Food, Drug and Cosmetic Act, or the FD&C Act, to sell our NAFLD
TAEUS application in the U.S. We anticipate that the application,
as well as those for our other TAEUS applications, will be
submitted for approval under Section 510(k) of the FD&C Act.
Based on our understanding of applicable regulations and
consultations with medical device regulatory consulting firms and
medical device contract engineering firms, we expect to submit this
application to the FDA approximately eleven months after the
completion of this offering and for the FDA to make a final
determination on our application approximately eleven months after
that application is submitted. We estimate that we will use
approximately $100,000 of the net proceeds from this offering on
such activities. However, these estimates are subject to
uncertainty and there can be no assurance that these processes will
not take longer or be more costly than we expect. We expect that
our initial FDA clearance will allow us to sell the NAFLD TAEUS
application in the U.S. with general imaging claims. However, we
will need to obtain additional FDA clearances to be able to make
diagnostic claims for fatty tissue content determination.
Accordingly, to support our commercialization efforts we expect
that, following receipt of our initial FDA clearance, we will
submit one or more additional applications to the FDA, each of
which will need to include additional clinical trial data, so that
following receipt of the necessary clearances we may make those
diagnostic claims.
Regulation
European Union
The
primary regulatory environment in Europe is the European Union,
which consists of 28 member states encompassing most of the major
countries in Europe. We believe that in the European Union
applications
incorporating our TAEUS technology will be regulated as Class IIa
medical devices by the European Medicines Agency, or EMA, and the
European Union Commission. As described above, we expect our
applications will
receive a CE mark from an appropriate Competent Authority as a
result of successful review of one or more submissions prepared by
our contract engineering and manufacturer(s), so that such
applications can
be marketed and distributed within the European Economic Area. Each
of our applications will be required
to be recertified each year for CE marking, which recertification
may require an annual audit. The audit procedure, which will
include on-site visits at our facility, and the contract
manufacturer’s(s’) facility(ies), will require us to
provide the contract manufacturer(s) with information and
documentation concerning our quality management system and all
applicable documents, policies, procedures, manuals, and other
information.
57
In the
European Union, the manufacturer of medical devices is subject to
current Good Manufacturing Practice, or cGMP, as set forth in the
relevant laws and guidelines of the European Union and its member
states. Compliance with cGMP is generally assessed by a Notified
Body accredited by a Competent Authority. For a Class IIa device,
typically, quality system evaluation is performed by the Notified
Body, which also recommends to the relevant Competent Authority for
the European community whether a device will receive a CE mark. The
Notified Body may conduct inspections of relevant facilities, and
review manufacturing procedures, operating systems and personnel
qualifications. In addition to obtaining approval for each
application, in
many cases each device manufacturing facility must be audited on a
periodic basis by the Notified Body. Further inspections may occur
over the life of the application.
FDA Regulation
Each of
our products must be approved or cleared by the FDA before it is
marketed in the United States Before and after approval or
clearance in the United States, our applications are subject to
extensive regulation by the FDA under the FD&C Act and/or the Public Health
Service Act, as well as by other regulatory bodies. The FDA
regulations govern, among other things, the development, testing,
manufacturing, labeling, safety, storage, record-keeping, market
clearance or approval, advertising and promotion, import and
export, marketing and sales, and distribution of medical devices
and pharmaceutical products.
FDA Approval or Clearance of Medical Devices
In the
United States, medical devices are subject to varying degrees of
regulatory control and are classified in one of three classes
depending on the extent of controls the FDA determines are
necessary to reasonably ensure their safety and
efficacy:
●
Class I: general
controls, such as labeling and adherence to quality system
regulations;
●
Class II: special
controls, premarket notification (510(k)), specific controls such
as performance standards, patient registries and post-market
surveillance and additional controls such as labeling and adherence
to quality system regulations; and
●
Class III: special
controls and approval of a premarket approval, or PMA,
application.
We
expect all of our products to be classified as Class II medical
devices and require FDA authorization prior to marketing by means
of a 510(k) clearance.
To
request marketing authorization by means of a 510(k) clearance, we
must submit a premarket notification demonstrating that the
proposed device is substantially equivalent to another legally
marketed medical device, has the same intended use, and is as safe
and effective as a legally marketed device and does not raise
different questions of safety and effectiveness than a legally
marketed device. 510(k) submissions generally include, among other
things, a description of the device and its manufacturing, device
labeling, medical devices to which the device is substantially
equivalent, safety and biocompatibility information and the results
of performance testing. In some cases, a 510(k) submission must
include data from human clinical studies. Marketing may commence
only when the FDA issues a clearance letter finding substantial
equivalence. The typical duration to receive a 510(k) approval is
approximately nine to twelve months from the date of the initial
510(k) submission, although there is no guarantee that the timing
will not be longer.
In the past, the
510(k) pathway for product marketing has required only proof of
substantial equivalence in technology for a given indication with a
previously cleared device. Recently, there has been a trend of the
FDA requiring additional clinical work to prove efficacy in
addition to technological equivalence and basic safety. Whether
clinical data is provided or not, the FDA may decide to reject the
substantial equivalence argument we present. If that happens, the
device is automatically designated as a Class III device. The
device sponsor must then fulfill more rigorous PMA requirements, or
can request a risk-based classification determination for the
device in accordance with the “de novo” process, which
may determine that the new device is of low to moderate risk and
that it can be appropriately be regulated as a Class I or II
device. If a de novo request is granted, the device may be legally
marketed and a new classification is established. If the device is
classified as Class II, the device may serve as a predicate for
future 510(k) submissions. If the device is not approved through de
novo review, then it must go through the standard PMA process for
Class III devices.
58
After a
device receives 510(k) clearance, any product modification that
could significantly affect the safety or effectiveness of the
product, or that would constitute a significant change in intended
use, requires a new 510(k) clearance or, if the device would no
longer be substantially equivalent, a PMA. If the FDA determines
that the product does not qualify for 510(k) clearance, then a
company must submit, and the FDA must approve, a PMA before
marketing can begin.
A PMA
application must provide a demonstration of safety and
effectiveness, which generally requires extensive pre-clinical and
clinical trial data. Information about the device and its
components, device design, manufacturing and labeling, among other
information, must also be included in the PMA. As part of the PMA
review, the FDA will inspect the manufacturer’s facilities
for compliance with quality system regulation requirements, which
govern testing, control, documentation and other aspects of quality
assurance with respect to manufacturing. If the FDA determines the
application or manufacturing facilities are not acceptable, the FDA
may outline the deficiencies in the submission and often will
request additional testing or information. Notwithstanding the
submission of any requested additional information, the FDA
ultimately may decide that the application does not satisfy the
regulatory criteria for approval. During the review period, a FDA
advisory committee, typically a panel of clinicians and
statisticians, is likely to be convened to review the application
and recommend to the FDA whether, or upon what conditions, the
device should be approved. The FDA is not bound by the advisory
panel decision. While the FDA often follows the panel’s
recommendation, there have been instances in which the FDA has not.
The FDA must find the information to be satisfactory in order to
approve the PMA. The PMA approval can include post-approval
conditions, including, among other things, restrictions on
labeling, promotion, sale and distribution, or requirements to do
additional clinical studies after approval. Even after approval of
a PMA, a new PMA or PMA supplement is required to authorize certain
modifications to the device, its labeling or its manufacturing
process. Supplements to a PMA often require the submission of the
same type of information required for an original PMA, except that
the supplement is generally limited to that information needed to
support the proposed change from the product covered by the
original PMA. The typical duration to receive PMA approval is
approximately two years from the date of submission of the initial
PMA application, although there is no guarantee that the timing
will not be longer.
Clinical Trials of Medical Devices
One or
more clinical trials are generally required to support a PMA
application and more recently are becoming necessary to support a
510(k) submission. Clinical studies of unapproved or uncleared
medical devices or devices being studied for uses for which they
are not approved or cleared (investigational devices) must be
conducted in compliance with FDA requirements. If an
investigational device could pose a significant risk to patients,
the sponsor company must submit an investigational device exemption
application to the FDA prior to initiation of the clinical study.
An investigational device exemption application must be supported
by appropriate data, such as animal and laboratory test results,
showing that it is safe to test the device on humans and that the
testing protocol is scientifically sound. The investigational
device exemption will automatically become effective 30 days after
receipt by the FDA unless the FDA notifies the company that the
investigation may not begin. Clinical studies of investigational
devices may not begin until an institutional review board has
approved the study.
59
During
the study, the sponsor must comply with the FDA’s
investigational device exemption requirements. These requirements
include investigator selection, trial monitoring, adverse event
reporting, and record keeping. The investigators must obtain
patient informed consent, rigorously follow the investigational
plan and study protocol, control the disposition of investigational
devices, and comply with reporting and record keeping requirements.
The sponsor, the FDA, or the institutional review board at each
institution at which a clinical trial is being conducted may
suspend a clinical trial at any time for various reasons, including
a belief that the subjects are being exposed to an unacceptable
risk. During the approval or clearance process, the FDA typically
inspects the records relating to the conduct of one or more
investigational sites participating in the study supporting the
application.
Post-Approval Regulation of Medical Devices
After a
device is cleared or approved for marketing, numerous and pervasive
regulatory requirements continue to apply. These
include:
●
the FDA quality
systems regulation, which governs, among other things, how
manufacturers design, test, manufacture, exercise quality control
over, and document manufacturing of their products;
●
labeling and claims
regulations, which prohibit the promotion of products for
unapproved or “off-label” uses and impose other
restrictions on labeling; and
●
the Medical Device
Reporting regulation, which requires reporting to the FDA of
certain adverse experiences associated with use of the
product.
Good Manufacturing Practices Requirements
Manufacturers of
medical devices are required to comply with the good manufacturing
practices set forth in the quality system regulation promulgated
under Section 520 of the FD&C Act. Current good
manufacturing practices regulations require, among other things,
quality control and quality assurance as well as the corresponding
maintenance of records and documentation. The manufacturing
facility for an approved product must be registered with the FDA
and meet current good manufacturing practices requirements to the
satisfaction of the FDA pursuant to a pre-PMA approval inspection
before the facility can be used. Manufacturers, including third
party contract manufacturers, are also subject to periodic
inspections by the FDA and other authorities to assess compliance
with applicable regulations. Failure to comply with statutory and
regulatory requirements subjects a manufacturer to possible legal
or regulatory action, including the seizure or recall of products,
injunctions, consent decrees placing significant restrictions on or
suspending manufacturing operations, and civil and criminal
penalties. Adverse experiences with the product must be reported to
the FDA and could result in the imposition of marketing
restrictions through labeling changes or in product withdrawal.
Product approvals may be withdrawn if compliance with regulatory
requirements is not maintained or if problems concerning safety or
efficacy of the product occur following the approval.
China
Regulation
China’s
regulatory approval framework includes nationwide approval based on
a showing that the device for which approval is sought has been
previously approved in the country of origin. Alternatively, we
understand it is also possible to receive approval at the
provincial level or to work exclusively with hospitals that do not
require such nationwide or provincial approval. We intend to
explore these potential paths to regulatory compliance in
China.
Other Regulations
We will
become subject to regulations and product registration requirements
in many foreign countries in which we may sell our products,
including in the areas of product standards, packaging
requirements, labeling requirements, import and export restrictions
and tariff regulations, duties and tax requirements. The time
required to obtain clearance required by foreign countries may be
longer or shorter than that required for EMA or FDA clearance, and
requirements for licensing a product in a foreign country may
differ significantly from EMA and FDA requirements.
60
Competition
While
we believe that we are the only company developing RF-based
thermoacoustic ultrasound products, we will face direct and
indirect competition from a number of competitors, many of whom
have greater financial, sales and marketing and other resources
than we do.
Manufacturers of CT
and MRI systems include multi-national corporations such as Royal
Philips, Siemens AG and Hitachi, Ltd., many of whom also
manufacture and sell ultrasound equipment. In the NAFLD diagnosis
market we will compete with makers of surgical biopsy tools, such
as Cook Medical and Sterylab S.r.l. In the thermal ablation market,
we will compete with manufacturers of surgical temperature probes,
such as Medtronic plc and St. Jude Medical, Inc.
Research
and Development
Our
research and development expenses were approximately $1,038,878 and
$873,167 for the years ended December 31, 2015 and 2014,
respectively.
Employees
As of
September 30, 2016, we had eight
employees, five of whom are employed on a full-time basis. Three
full-time employees and two part-time employees were engaged in
research and development activities, one full-time employee was
engaged in administrative activities, one full-time employee was
engaged in product assembly and one part-time employee was engaged
in marketing activities. After the closing of the offering, we
intend to employ certain of our part-time employees on a full-time
basis and to hire a full-time Chief Financial Officer. None of our
employees is covered by a collective bargaining agreement, and we
believe our relationship with our employees is good.
We also
employ technical advisors, on an as-needed basis, to supplement
existing staff. We believe that these technical advisors provide us
with necessary expertise in clinical ultrasound applications,
ultrasound technology, and intellectual property.
Properties
Our
principal office is located at 3600 Green Court, Suite 350, Ann
Arbor, Michigan 48105-1570. We currently lease approximately 3,657
square feet of office and light industrial/research space under a
lease that is due to expire in 2020. The rent is approximately
$6,135 per month, subject to moderate annual increases. We believe
that equivalent suitable space is available at similar
rents.
Legal
Proceedings
We are
not a party to any pending legal proceedings.
61
The
following table sets forth the names and ages of all of our
executive officers and directors. Our officers are appointed
by, and serve at the pleasure of, the board of
directors.
|
Name
|
Age
|
Position
|
|
Francois
Michelon
|
50
|
Chief
Executive Officer and Chairman
|
|
Michael
Thornton
|
48
|
Chief
Technology Officer
|
|
David
Wells
|
54
|
Chief
Financial Officer (Interim)
|
|
Anthony
DiGiandomenico
|
50
|
Director
|
|
Dr.
Sanjiv Sam Gambhir
|
54
|
Director
|
|
Michael
Harsh
|
62
|
Director
|
|
Alexander
Tokman
|
55
|
Director
|
Biographical
information with respect to our executive officers and directors is
provided below. There are no family relationships
between any of our executive officers or directors.
Francois
Michelon − Chief Executive Officer and Chairman
Francois Michelon
joined ENDRA as Chief Executive Officer and Chairman of the Board
of Directors in 2015. He has 18 years of healthcare technology
experience in general management, operations, strategy and
marketing across the diagnostic imaging, surgical instrument and
dental sectors.
From
2012 to 2014, Mr. Michelon served as Vice President of Global
Marketing for the 3i division of Biomet, Inc. (now Zimmer Biomet
Holdings, Inc.), a provider of oral reconstruction technologies,
where he was responsible for the upstream and downstream
development of the division’s global portfolio. From 2004 to
2011, Mr. Michelon served as Group Director of Global Services and
Visualization for Smith & Nephew plc’s Advanced Surgical
Devices division, where he led P&L’s in the B2B service
and capital equipment sectors. From 1997 to 2004, Mr. Michelon
worked at GE Healthcare in a variety of global upstream and
downstream marketing roles.
Mr.
Michelon received an MBA from Carnegie-Mellon University and a BA
in Economics from the University of Chicago. He has also earned his
Six Sigma Black Belt certification. Mr. Michelon’s extensive
industry and executive experience position him well to serve as our
Chief Executive Officer and a member of our board of
directors.
Michael
Thornton − Chief Technology Officer
Prior
to joining ENDRA as Chief Technology Officer in 2007, Michael
Thornton was a founder and President of Enhanced Vision Systems
Corp., or EVS, a developer and supplier of medical imaging
equipment to the pharmaceutical, biotech, and academic
sectors.
In
2002, EVS was acquired by General Electric Company and was
integrated into the Functional and Molecular Imaging business unit
of GE Medical Systems (now GE Healthcare, a subsidiary of General
Electric Company). Following the acquisition of EVS by GE Medical
Systems, Mr. Thornton held a number of positions at GE Healthcare,
including Sales Manager, Global Product Manager, and Site Leader.
He was a member of the leadership team that expanded the
pre-clinical imaging business to include: computed tomography,
optical, and positron emission tomography imaging technologies,
with global market reach. He is also a founder of Volumetrics
Medical Corp., a developer and manufacturer of quality assurance
devices for diagnostic imaging.
Prior
to founding EVS, Mr. Thornton developed medical imaging related
technologies at the Robarts Research Institute (London, Ontario,
Canada) for which he obtained an MSc in Electrical Engineering from
the University of Western Ontario. Mr. Thornton also holds a BASc
in Electrical Engineering from the University of Toronto and is a
member of the American Association of Physicists in
Medicine.
62
David
Wells − Chief Financial Officer (Interim)
David
Wells became our Chief Financial Officer on an interim basis in
2014. He possesses 30 years of experience in finance, operations
and administrative positions. While mainly focused on technology
companies, Mr. Wells has also worked in the water treatment,
supply-chain management, manufacturing and professional services
industries.
Mr.
Wells is the founder of Wells Compliance Group, a technology-based
services firm supporting the financial reporting needs of publicly
traded companies and privately held firms whose investor or
shareholder base requires timely GAAP-compliant financial
reporting. Mr. Wells has been the Chief Executive Officer of
StoryCorp Consulting (d/b/a/ Wells Compliance Group) since March
2013. In this role, Mr. Wells has served as the principal financial
officer of several emerging growth companies, including Mount Tam
Biotechnologies, Inc., a biopharmaceutical company (August 2015 to
April 2016), Content Checked Holdings, Inc., a technology company
(April 2015 to present), and Loton, Corp., a media company
(February 2016 to present). From 2009 to 2013, he was the
President, CFO and a Director of Sionix Corporation, a publicly
traded water treatment company.
Mr.
Wells holds an MBA from Pepperdine University and a BS in Finance
and Entrepreneurship from Seattle Pacific University.
Anthony
DiGiandomenico − Director
Anthony
DiGiandomenico joined ENDRA’s board of directors in 2013. A
co-founder of MDB Capital Group LLC, Mr. DiGiandomenico focuses on
corporate finance and capital formation for growth-oriented
companies. He has participated in all areas of corporate finance
including private capital, public offerings, PIPEs, business
consulting and strategic planning, and mergers and
acquisitions.
Mr.
DiGiandomenico has also worked on a wide range of transactions for
growth-oriented companies in biotechnology, nutritional
supplements, manufacturing and entertainment industries. Prior to
forming MDB Capital Group LLC in 1997, Mr. DiGiandomenico served as
President and CEO of the Digian Company, a real estate development
company.
Mr.
DiGiandomenico holds an MBA from the Haas School of Business at the
University of California, Berkeley and a BS in Finance from the
University of Colorado. Mr. DiGiandomenico’s financial
expertise, general business acumen and significant executive
leadership experience position him well to make valuable
contributions to our board of directors.
Dr.
Sanjiv Sam Gambhir − Director
Dr.
Sanjiv Sam Gambhir joined our board of directors in 2008. He is the
Virginia & D.K. Ludwig Professor of Cancer Research and the
Chair of Radiology at Stanford University School of Medicine. He
also heads the Canary Center at Stanford for Cancer Early Detection
and directs the Molecular Imaging Program at Stanford
(MIPS).
He
received an MD/PhD from the UCLA Medical Scientist Training
Program. He has many publications in the field and numerous patents
pending or granted. He has developed and clinically translated
several multimodality molecular imaging strategies including
imaging of gene and cell therapies. He has also pioneered imaging
areas such as Bioluminescence Resonance Energy Transfer (BRET),
split-reporter technology, Raman imaging in vivo, Molecular
Photoacoustic imaging, PET reporter genes, and novel in vitro and
in vivo strategies for the early detection of cancer.
Dr.
Gambhir serves on numerous academic advisory boards for
universities around the world and also served as a member of the
Board of Scientific Advisors of the National Cancer Institute from
2004 to 2012. He has also founded or co-founded several startups in
the diagnostics space. Among his many awards are the George Von
Hevesy Prize and the Paul C. Aebersold Award for outstanding
achievement in basic nuclear medicine science from the Society of
Nuclear Medicine, Outstanding Researcher Award from the
Radiological Society of Northern America, the Distinguished
Clinical Scientist Award from the Doris Duke Charitable Foundation,
the Holst Medal, the Tesla Medal, and the Hounsfield Medal from
Imperial College, London. He was elected to the Institute of
Medicine of the U.S. National Academies in 2008. Dr.
Gambhir’s unique and extensive scientific and technical
expertise positions him well to serve on our board of
directors.
63
Michael
Harsh − Director
Michael
Harsh joined ENDRA’s board of directors in 2015. He has 36
years’ experience in healthcare technology, focused on
diagnostic imaging. Mr. Harsh was most recently GE
Healthcare’s Vice President and Chief Technology Officer,
leading its global science and technology organization and research
and development teams in diagnostics, healthcare IT and life
sciences.
In
2004, Mr. Harsh was named Global Technology Leader – Imaging
Technologies Lab at the GE Global Research Center, where he
led the research for imaging technologies across the company as
well as the research associated with computer visualization/image
analysis and superconducting systems. He led the Engineering
division for GE Industrial and Enterprise Solutions from 2006
to 2009. Mr. Harsh was named an officer of General Electric Company
in November 2006. Mr. Harsh is a co-founder and current Chief
Product Officer of Terapede Systems Inc., a digital x-ray detector
startup, a member of the board of directors of FloDesign Sonics,
Inc., a member of the Scientific Advisory Board of Phoenix Nuclear
Labs, LLC and a consultant to start-ups in the medical device
industry.
Mr.
Harsh is a graduate of Marquette University, where he earned a
bachelor’s degree in Electrical Engineering. He holds
numerous U.S. patents in the field of medical imaging and
instrumentation. In 2008, Mr. Harsh was elected to the American
Institute for Medical and Biological Engineering College of
Fellows for his significant contributions to the medical and
biological engineering field. Mr. Harsh’s extensive industry,
executive and board experience position him well to serve on our
board of directors.
Alexander
Tokman − Director
Alexander Tokman
joined ENDRA’s board of directors in 2008. He has served as
President, Chief Executive Officer, and a director of Microvision,
Inc., a publicly traded laser beam scanning projection and imaging
company, since January 2006.
Previously, Mr.
Tokman completed a 10+ year tenure as an executive with GE
Healthcare, where he led several global businesses, most recently
as a General Manager of its Global Molecular Imaging and
Radiopharmacy multi-technology business unit from 2003 to
2005.
Between
1995 and 2003, Mr. Tokman served in various leadership roles at GE
Healthcare, where he led the definition and successful
commercialization of several product segments, including PET/CT,
which generated over $500 million of revenue within the first three
years of its launch.
Mr.
Tokman is a certified Six Sigma and Design for Six Sigma (DFSS)
Black Belt and Master Black Belt and as one of General Electric
Company’s Six Sigma pioneers, he drove the quality culture
change across GE Healthcare in the late 1990s. From 1989 to 1995,
Mr. Tokman served as development programs lead and a head of
Industry and Regional Development at Tracor Applied Sciences. Mr.
Tokman has both an MS and BS in Electrical Engineering from the
University of Massachusetts, Dartmouth. Mr. Tokman’s industry
expertise and significant executive leadership and director
experience position him well to make valuable contributions to our
board of directors.
Director
Independence
Our
board of directors has determined that Anthony DiGiandomenico, Dr.
Sanjiv Sam Gambhir, Michael Harsh and Alexander Tokman are “independent
directors” as such term is defined by Nasdaq Marketplace Rule
5605(a)(2). We have established an Audit Committee, a
Compensation Committee and a Nominating and Corporate Governance
Committee. Each of Mr. DiGiandomenico, Mr. Harsh and Mr.
Tokman serve as members of the Audit Committee and Compensation
Committee. Mr. Gambhir, Mr. Harsh and Mr. Tokman serve as members
of the Nominating and Corporate Governance Committee. Our board of
directors has determined that Mr.
DiGiandomenico is an audit committee financial expert, as
defined under the applicable rules of the SEC, and that all members
of the Audit Committee are “independent” within the
meaning of the applicable Nasdaq listing standards and the
independence standards of Rule 10A-3 of the Exchange Act. Each of
the members of the Audit Committee meets the requirements for
financial literacy under the applicable rules and regulations of
the SEC and The Nasdaq Stock Market.
64
Scientific
Advisory Board
Our
Scientific Advisory Board members work with our management team in
the planning, development and execution of scientific and business
strategies. It reviews, and advises management on our progress in
research and clinical development as well as new scientific
perspectives.
Jonathan
Rubin, MD, PhD − Scientific Advisor
Dr.
Jonathan Rubin is the Martel Collegiate Professor of Radiology and
Section Head for Ultrasound and Abdominal Interventional Radiology
at the University of Michigan Medical School.
Dr.
Rubin has over 200 peer-reviewed publications, over 125 invited
presentations, and 10 patents. In 2005 he was awarded the
University of Michigan Medical School Innovation Award. In 2007 he
won the American Institute of Ultrasound in Medicine Joseph H.
Holmes Clinical Pioneer Award. In 2011 he received the Society of
Radiologists in Ultrasound Lawrence Mack Lifetime Achievement
Award.
Dr.
Rubin received a BA in Chemistry from the University of Utah. He
received an MD from the University of Chicago Pritzker School of
Medicine and a PhD in Biophysics and Theoretical Biology from the
University of Chicago. From 1979 to 1984, Dr. Rubin was the
director of the Section of Body Computed Tomography and Ultrasound
Imaging in the Department of Radiology at the University of
Chicago.
Dr. Jing Gao, MD − Scientific Advisor
Dr.
Jing Gao is currently Research Assistant Professor of Radiology at
Weill Cornell Medicine in New York, NY. Dr. Gao brings over 30
years of clinical and research experience in abdominal ultrasound,
in both the United States and China.
Dr. Gao
completed her medical education at Changchun and Dalian Medical
Colleges in China. Besides her post at Cornell, Dr. Gao is also
Deputy President and guest professor at the Dalian University
International Institute of Medical Imaging in China.
Her
numerous honors and professional affiliations include being named
one of China’s Top 100 Ultrasound Physicians by the Chinese
Association of Medical Imaging Technology. She is a Fellow of the
Chinese Association of Ultrasound in Medicine and Biology, a Fellow
of the American Institute of Ultrasound in Medicine and an
Editorial Board Member of Clinical Imaging (Elsevier).
Dr. Gao
has numerous peer reviewed publications in the areas of liver,
spleen and kidney diseases and quantitative ultrasound
imaging.
65
Our
compensation philosophy is to offer our executive officers
compensation and benefits that are competitive and meet our goals
of attracting, retaining and motivating highly skilled management,
which is necessary to achieve our financial and strategic
objectives and create long-term value for our stockholders. We
believe the levels of compensation we provide should be
competitive, reasonable and appropriate for our business needs and
circumstances and our board of directors uses benchmark
compensation studies in determining compensation elements and
levels. The principal elements of our executive compensation
program have to date included base salary, annual bonus opportunity
and long-term equity compensation in the form of stock options. We
believe successful long-term Company performance is more critical
to enhancing stockholder value than short-term results. For this
reason and to conserve cash and better align the interests of
management and our stockholders, we emphasize long-term
performance-based equity compensation over base annual
salaries.
The
following table sets forth information concerning the compensation
earned by the individual that served as our Principal Executive
Officer during 2016 and our two most highly
compensated executive officers other than the individual who served
as our Principal Executive Officer during 2016
(collectively, the “named executive
officers”):
2016
Summary Compensation Table
|
Name &
Position
|
Fiscal
Year
|
Salary
($)
|
Bonus ($)
|
Option
Awards($)(1)
|
All Other Compensation
($)
|
Total
($)
|
|
Francois
Michelon
|
2016
|
262,152
|
-
|
-
|
-
|
262,152
|
|
Chief Executive Officer
|
2015(2)
|
177,083
|
-
|
248,359
|
-
|
425,442
|
|
Michael
Thornton
|
2016
|
218,056
|
-
|
-
|
-
|
218,056
|
|
Chief Technology Officer
|
2015
|
200,000(3)
|
-
|
-
|
-
|
200,000
|
|
David R.
Wells
|
2016
|
96,000(4)
|
-
|
-
|
-
|
96,000
|
|
Chief Financial Officer (Interim)
|
2015
|
96,000(5)
|
-
|
-
|
-
|
96,000
|
________
(1)
The amounts shown
in this column indicate the grant date fair value of option awards
granted in the subject year computed in accordance with FASB ASC
Topic 718. For additional information regarding the assumptions
made in calculating these amounts, see notes 2 and 7
to our audited financial statements included herein.
(2)
Represents a
partial year of employment. Mr. Michelon joined us on April 16,
2015.
(3)
Includes $33,403 of
accrued compensation settled for 5,556 shares of common
stock.
(4)
Represents fees earned by StoryCorp
Consulting (d/b/a Wells Compliance Group) pursuant to the
consulting agreement described below. $60,000 of this was paid in
cash and the balance will be paid in shares of restricted stock
upon the completion of this offering.
(5)
Represents fees
earned by StoryCorp Consulting (d/b/a Wells Compliance Group)
pursuant to the consulting agreement described below.
$60,000 of this was paid in cash and the balance will
be paid in shares of restricted stock upon the
completion of this offering.
66
Outstanding
Equity Awards at 2016 Fiscal Year-End
The
following table provides information regarding equity awards held
by the named executive officers as of December 31,
2016.
|
Name
and Principal Position
|
Number of Securities Underlying Unexercised Options Exercisable
(#)
|
Number of Securities Underlying Unexercised Options Unexercisable
(#)
|
Option Exercise Price ($)
|
Option Expiration Date
|
|
Francois
Michelon
|
16,566
|
33,133(1)
|
$ 7.15
|
July 1,
2020
|
|
Chief Executive Officer
|
|
|
|
|
|
Michael
Thornton
|
41,260
|
-
|
$ 7.15
|
November
1, 2018
|
|
Chief Technology Officer
|
|
|
|
|
|
David
R. Wells
|
-
|
-
|
-
|
-
|
|
Chief Financial Officer (Interim)
|
|
|
|
|
________
(1)
These options vest in two equal annual installments on
July 1 of 2017 and 2018.
Employment
Agreements and Change of Control Arrangements
Employment Agreements
The
following is a summary of the employment arrangements with our
executive officers as currently in effect.
Francois
Michelon. On July 21, 2016, our board of
directors approved an amended and restated employment agreement
with Francois Michelon, our Chief Executive Officer and Chairman of
our board of directors, which shall become effective upon the
closing of this offering. The term of the employment agreement runs
through December 31, 2019. The employment agreement provides for an
annual base salary of $325,000. Under the employment agreement, Mr.
Michelon is eligible for an annual cash bonus (in 2016, up to 35%
of his base salary then in effect) based upon achievement of
performance-based objectives established by our board of directors.
Pursuant to Mr. Michelon’s employment agreement, upon the
closing of this offering he is entitled to be granted options to
purchase a number of shares of common stock that, taken together
with the option to purchase 49,699 shares of common stock he
already holds, equals 5.0% of the Company’s total issued and
outstanding shares of common stock on the date of grant on a fully
diluted basis. The options will have an exercise price equal to the
price at which our common stock is offered to investors in this
offering and will vest in three equal annual installments beginning
on the first anniversary of its grant date. Upon termination
without cause, any portion of Mr. Michelon’s options
scheduled to vest within 12 months will automatically vest, and
upon termination without cause within 12 months following a change
of control, the entire unvested portion of the option will
automatically vest. Upon termination for any other reason, the
entire unvested portion of the option will terminate.
If Mr.
Michelon’s employment is terminated by the Company without
cause, Mr. Michelon will be entitled to receive 12 months’
continuation of his current base salary and a lump sum payment
equal to 12 months of continued healthcare coverage (or 24
months’ continuation of his current base salary and a lump
sum payment equal to 24 months of continued healthcare coverage if
such termination occurs within one year following a change in
control).
Under
his employment agreement, Mr. Michelon is eligible to receive
benefits that are substantially similar to those of the
Company’s other senior executive officers.
Michael
Thornton. On July 21, 2016, our board of
directors approved an amended and restated employment agreement
with Michael Thornton, our Chief Technology Officer, which shall
become effective upon the closing of this offering. Under the
employment agreement, Mr. Thornton’s title will be Chief
Technology Officer. The term of the employment agreement runs
through December 31, 2019. The employment agreement provides for an
annual base salary of $245,000. Under the employment agreement, Mr.
Thornton is eligible for an annual cash bonus (in 2016, up to 22%
of his base salary then in effect) based upon achievement of
performance-based objectives established by our board of directors.
Pursuant to Mr. Thornton’s employment agreement, upon the
closing of this offering he is entitled to be granted options to
purchase a number of shares of common stock that, taken together
with the option to purchase 41,260 shares of common stock he
already holds, equals 5.0% of the Company’s total issued and
outstanding shares of common stock on the date of grant on a fully
diluted basis. The options will have an exercise price equal to the
price at which our common stock is offered to investors in this
offering and will vest in three equal annual installments beginning
on the first anniversary of its grant date. Upon termination
without cause, any portion of Mr. Thornton’s option scheduled
to vest within 12 months will automatically vest, and upon
termination without cause within 12 months following a change of
control, the entire unvested portion of the option will
automatically vest. Upon termination for any other reason, the
entire unvested portion of the option will terminate
If Mr.
Thornton’s employment is terminated by the Company without
cause, Mr. Thornton will be entitled to receive 12 months’
continuation of his current base salary and a lump sum payment
equal to 12 months of continued healthcare coverage (or 24
months’ continuation of his current base salary and a lump
sum payment equal to 24 months of continued healthcare coverage if
such termination occurs within one year following a change in
control).
67
Under
his employment agreement, Mr. Thornton is eligible to receive
benefits that are substantially similar to those of the
Company’s other senior executive officers.
David R. Wells. We
entered into a consulting agreement with StoryCorp Consulting
(d/b/a Wells Compliance Group),or StoryCorp, in July 2014 for
services provided to the Company by David R. Wells, our Chief
Financial Officer (interim). Under this consulting agreement, the
Company pays to StoryCorp a monthly fee of $8,000, of which $5,000
is payable in cash and $3,000 is payable in shares of restricted
stock of the Company at the closing of this offering in an amount
based on the per share price of our common stock sold in this
offering. Additionally, StoryCorp may issue invoices to the Company
for services provided outside of those described in the consulting
agreement at a rate of $250 per hour, payable in cash, and the
Company will reimburse StoryCorp for reasonable and necessary
expenses incurred in connection with the performance of its
services under the consulting agreement. The consulting
agreement’s term renews monthly and the agreement may be
terminated by the Company with or without cause immediately and
without prior notice to StoryCorp. On July 21, 2016, our board of
directors approved a one-time grant to Mr. Wells effective upon the
closing of this offering of options to purchase $35,000 worth of
shares of our common stock with an exercise price equal to the
price at which our common stock is offered to investors in this
offering.
Director
Compensation
Members
of our board of directors received a one-time grant of fully vested
stock options in January 2016 for their service as directors for
the year ended December 31, 2015. On July 21, 2016, we adopted a
non-employee director compensation policy that will become
effective upon the closing of this offering pursuant to which our
non-employee directors will receive on an annual basis a $36,000
retainer paid in cash and an annual equity award with a value of
$30,000. The equity award will consist of a stock option grant made
on the first trading day following December 31 of each year
covering a number of shares of common stock equal to $30,000
divided by the closing price of our common stock on such date and
that vests in full on the one year anniversary of
grant; provided, the grant
for 2017 will be made upon the closing of this offering and the
number of shares covered will be equal to $30,000 divided by the
price at which our securities are offered to investors in this
offering. Because our non-employee directors
did not receive any compensation for their service
during 2016, the non-employee director policy provides that,
in addition, upon the closing of this offering each
non-employee director is entitled to a stock option award covering
a number of shares of securities equal to $30,000
divided by the price at which our securities are
offered to investors in this offering and that will be
immediately exercisable.
The following table
sets forth information with respect to compensation earned by or
awarded to each of our non-employee directors who served on our
board of directors during the fiscal year ended December 31,
2016:
|
Name
|
Fees Earned or
Paid in Cash ($)
|
Option Awards
($)(1)
|
All Other
Compensation ($)
|
Total
($)
|
|
Anthony
DiGiandomenico
|
-
|
12,589
|
-
|
12,589
|
|
Dr. Sanjiv Sam
Gambhir
|
-
|
12,589
|
-
|
12,589
|
|
Michael
Harsh
|
-
|
12,589
|
-
|
12,589
|
|
Alexander
Tokman
|
-
|
12,589
|
-
|
12,589
|
(1)
The amounts shown
in this column indicate the grant date fair value of option awards
granted in the subject year computed in accordance with FASB ASC
Topic 718. For additional information regarding the assumptions
made in calculating these amounts, see notes 2 and 6 to our audited
financial statements included herein. The following table shows the
number of shares subject to outstanding option awards held by each
non-employee director as of December 31, 2016:
|
Name
|
Shares subject
to Outstanding Stock Option Awards (#)
|
|
Anthony
DiGiandomenico
|
11,329
|
|
Dr. Sanjiv Sam
Gambhir
|
27,759
|
|
Michael
Harsh
|
7,518
|
|
Alexander
Tokman
|
17,031
|
2016 Omnibus Incentive Plan
In
September 2016, our board of directors and stockholders approved
the 2016 Omnibus Incentive Plan, or the Incentive Plan, pursuant to
which, effective following completion of the offering, the total
number shares available for issuance under such plan shall equal
18% of the total number of shares of common stock outstanding
immediately following the completion of the offering (assuming for
this purpose the issuance of all shares issuable under the
Company’s equity plan, the conversion into common stock of
all outstanding securities that are convertible by their terms into
common stock and the exercise of all options and warrants
exercisable for shares of common stock and including shares and
warrants included in the units issued to the
underwriters pursuant to the offering
upon exercise of its over-allotment option, if any) (i.e. on a
“fully diluted basis”). Concurrently with the closing
of the offering and as described above, we expect to grant stock
options to our executive officers and directors covering a
significant number of shares. Following such grants we estimate
that the shares remaining available for grant under the Incentive
Plan will be approximately 6.2% of the
total number of shares of common stock outstanding on a fully
diluted basis following completion of the offering.
68
We have
set forth in the following table certain information regarding our
common stock beneficially owned by (i) each stockholder we know to
be the beneficial owner of 5% or more of our outstanding common
stock, (ii) each of our directors and named executive officers, and
(iii) all executive officers and directors as a
group. Generally, a person is deemed to be a
“beneficial owner” of a security if that person has or
shares the power to dispose or to direct the disposition of such
security. A person is also deemed to be a beneficial
owner of any securities of which the person has the right to
acquire beneficial ownership within 60 days pursuant to options,
warrants, conversion privileges or similar
rights. Unless otherwise indicated, ownership
information is as of September 30, 2016, and is based on
1,012,723 shares of common stock outstanding on that date (adjusted
for the reverse stock split). The percentage ownership
after the offering is based on shares of common stock outstanding,
including shares of common stock issuable upon the conversion of
our promissory notes and the issuance of the common stock
included in the units being sold in the
offering.
|
Name
of Beneficial Owner (1)
|
Number
of Shares Beneficially Owned (2)
|
Percentage
Owned Prior to the Offering
|
Percentage
Owned After the Offering
|
|
|
|
|
|
|
Francois
Michelon
|
52,019 (3)(4)
|
4.9%
|
1.2%
|
|
Michael
Thornton
|
123,125 (4)(5)
|
10.9%
|
2.7%
|
|
David
R. Wells
|
- (6)
|
*
|
*
|
|
Dr.
Sanjiv Sam Gambhir
|
27,759 (7)
|
2.7%
|
*
|
|
Michael
Harsh
|
7,518
(8)
|
*
|
*
|
|
Alexander
Tokman
|
17,032
(9)
|
1.7%
|
*
|
|
Anthony
DiGiandomenico
|
93,582
(4)(10)
|
8.9%
|
2.1%
|
|
All
directors and named executive officers as a group (7
individuals)
|
321,034
|
25.2%
|
6.8%
|
|
|
|
|
|
|
5%
or More Shareholders
|
|
|
|
|
Blue
Earth Fund, LP (11)
|
283,807
(4)(12)
|
22.9%
|
6.1%
|
|
Jeffrey
S. Padnos and Margaret M. Padnos (13)
|
188,442
(4)(14)
|
16.4%
|
4.1%
|
|
Benjamin L. Padnos (15)
|
186,532 (4)(16)
|
15.9%
|
4.1%
|
|
Erick
Richardson (17)
|
139,566
(4)(18)
|
12.9%
|
3.1%
|
|
Robert
C. Clifford (19)
|
104,876
(4)(20)
|
10.0%
|
2.3%
|
|
Peter
Appel (21)
|
96,926
(4)(22)
|
9.0%
|
2.2%
|
|
Daniel
Landry (23)
|
91,638
(24)
|
8.7%
|
2.0%
|
|
Don
Miloni (25)
|
86,752
(4)(26)
|
8.1%
|
1.9%
|
|
ENDRA
Holdings LLC (27)
|
82,338
|
8.1%
|
1.9%
|
|
Andreas
Typaldos (28)
|
85,115(4)(29)
|
8.0%
|
1.9%
|
|
Mark
L. Baum (30)
|
74,286
(31)
|
7.3%
|
1.7%
|
* Less
than one percent.
(1)
The
address of each officer and director is 3600 Green Court, Suite
350, Ann Arbor, MI 48105-1570.
(2)
Beneficial
ownership is determined in accordance with Rule 13d-3 under the
Exchange Act and is generally determined by voting powers and/or
investment powers with respect to securities. Unless otherwise
noted, the shares of common stock listed above are owned as of
September 30, 2016, and
are owned of record by each individual named as beneficial owner
and such individual has sole voting and dispositive power with
respect to the shares of common stock owned by each of
them.
(3)
Consists of 16,566
shares of common stock issuable upon the exercise of options held
directly that are presently exercisable and 35,453 shares of common stock
issuable upon the conversion of a convertible promissory
note.
(4)
Amounts of shares
of common stock issuable upon the conversion of outstanding
convertible promissory notes assume that such notes are converted
immediately prior to the offering at a conversion price of $1.00
per share pursuant to the terms thereof. These amounts exclude
shares to be issued with respect to interest accrued on such
convertible promissory notes after September 30, 2016. See
“Description of Our Capital Stock Convertible Promissory
Notes” for a description of the terms of our convertible
promissory notes.
69
(5)
Consists of (a)
6,954 shares of common stock held directly; (b) 41,260 shares of
common stock issuable upon the exercise of options held directly
that are presently exercisable; (c) 1,399 shares of common stock
issuable upon the exercise of warrants held directly that are
presently exercisable; and (d) 73,512 shares of common stock
issuable upon the conversion of convertible promissory
notes.
(6)
Does
not include shares issuable upon the exercise of options awarded
upon the closing of this offering pursuant to our consulting
agreement with StoryCorp. See “Executive Compensation
Employment Agreements and Change of Control
Arrangements.”
(7)
Consists of 27,759
shares of common stock issuable upon the exercise of options held
directly that are presently exercisable.
(8)
Consists of 7,518
shares of common stock issuable upon the exercise of options held
directly that are presently exercisable.
(9)
Consists
of 17,032 shares of
common stock issuable upon the exercise of options held directly
that are presently exercisable.
(10)
Consists of (a)
54,879 shares of common stock held directly; (b) 11,329 shares of
common stock issuable upon the exercise of options held directly
that are presently exercisable; (c) 1,399 shares of common stock
issuable upon the exercise of warrants held directly that are
presently exercisable; and (d) 25,975 shares of common stock
issuable upon the conversion of a convertible promissory note held
directly.
(11)
The
address of Blue Earth Fund, LP is 1312 Cedar Street, Santa Monica,
CA 90405. Sole voting and dispositive power with respect to all of
Blue Earth Fund, LP's 283,806 shares of common stock is held by
Brett Conrad, the manager of Blue Earth Fund, LP, whose address is
also 1312 Cedar Street, Santa Monica, CA 90405.
(12)
Consists of (a)
59,442 shares of common stock held directly; (b) 17,483 shares of
common stock issuable upon the exercise of warrants held directly
that are presently exercisable; and (c) 206,882 shares of common
stock issuable upon the conversion of a convertible promissory
note.
(13)
The
address of Jeffrey S. Padnos and Margaret M. Padnos is 1088 West
27th Street, Holland, MI 49423.
(14)
Consists of (a)
25,210 shares of common stock held jointly by Mr. and Mrs. Padnos;
(b) 10,804 shares of common stock issuable upon the exercise of
warrants held jointly by Mr. and Mrs. Padnos that are presently
exercisable; (c) 150,959 shares of common stock issuable upon the
conversion of a convertible promissory note held jointly by Mr. and
Mrs. Padnos; (d) 12,727 shares of common stock held by Jeffrey
& Margaret Padnos 2010 Generation Trust fbo Benjamin Padnos (as
to which Mr. and Mrs. Padnos have shared voting and investment
power); (e) 5,454 shares of common stock issuable upon the exercise
of warrants held by Jeffrey & Margaret Padnos 2010 Generation
Trust fbo Benjamin Padnos (as to which Mr. and Mrs. Padnos have
shared voting and investment power) that are presently exercisable;
(f) 13,968 shares of common stock issuable upon the conversion of a
convertible promissory note held by Jeffrey & Margaret Padnos
2010 Generation Trust fbo Benjamin Padnos (as to which Mr. and Mrs.
Padnos have shared voting and investment power); (g) 7,955 shares
of common stock held by Jeffrey & Margaret Padnos 2010
Generation Trust fbo Rebecca Padnos (as to which Mr. and Mrs.
Padnos have shared voting and investment power); (h) 3,409 shares
of common stock issuable upon the exercise of warrants held by
Jeffrey & Margaret Padnos 2010 Generation Trust fbo Rebecca
Padnos (as to which Mr. and Mrs. Padnos have shared voting and
investment power) that are presently exercisable; (i) 8,600 shares
of common stock issuable upon the conversion of a convertible
promissory note held by Jeffrey & Margaret Padnos 2010
Generation Trust fbo Rebecca Padnos (as to which Mr. and Mrs.
Padnos have shared voting and investment power); (j) 7,955 shares
of common stock held by Jeffrey & Margaret Padnos 2010
Generation Trust fbo Joshua Padnos (as to which Mr. and Mrs. Padnos
have shared voting and investment power); (k) 3,409 shares of
common stock issuable upon the exercise of warrants held by Jeffrey
& Margaret Padnos 2010 Generation Trust fbo Joshua Padnos (as
to which Mr. and Mrs. Padnos have shared voting and investment
power) that are presently exercisable; (l) 8,600 shares of common
stock issuable upon the conversion of a convertible promissory note
held by Jeffrey & Margaret Padnos 2010 Generation Trust fbo
Joshua Padnos (as to which Mr. and Mrs. Padnos have shared voting
and investment power); (m) 7,955 shares of common stock held by
Jeffrey & Margaret Padnos 2010 Generation Trust fbo Samuel
Padnos (as to which Mr. and Mrs. Padnos have shared voting and
investment power); (n) 3,409 shares of common stock issuable upon
the exercise of warrants held by Jeffrey & Margaret Padnos 2010
Generation Trust fbo Samuel Padnos (as to which Mr. and Mrs. Padnos
have shared voting and investment power) that are presently
exercisable; and (o) 8,600 shares of common stock issuable upon the
conversion of a convertible promissory note held by Jeffrey &
Margaret Padnos 2010 Generation Trust fbo Samuel Padnos (as to
which Mr. and Mrs. Padnos have shared voting and investment
power).
(15)
The
address of Benjamin L. Padnos is 221 34th Street, Manhattan Beach,
CA 90266.
(16)
(17)
The address of Erick Richardson is 11290 Chalon Road, Los Angeles,
CA 90049.
(18)
Consists of (a) 50,468 shares of common stock held directly; (b)
15,735 shares of common stock held jointly by Erick and Molly
Richardson; (c) 11,364 shares of common stock issuable upon the
exercise of warrants held jointly by Mr. and Mrs. Richardson; and
(d) 61,999 shares issuable upon the conversion of a promissory note
held directly.
(19)
The
address of Robert C. Clifford is 1057 Corsica Drive, Pacific
Palisades, CA 90272.
70
(20)
Consists of (a)
52,082 shares of common stock held by 1999 Clifford Family Trust,
dated 12/22/1999 (as to which Mr. Clifford has shared voting and
investment power); (b) 19,714 shares of common stock held by The
Kingdom Trust Company Custodian fbo Robert C. Clifford (as to which
Mr. Clifford has voting and investment power); (c) 12,944 shares of
common stock issuable upon the exercise of warrants held by The
Kingdom Trust Company Custodian fbo Robert C. Clifford (as to which
Mr. Clifford has voting and investment power); and (d) 20,136
shares of common stock issuable upon the conversion of a
convertible promissory note held by The Kingdom Trust Company
Custodian fbo Robert C. Clifford (as to which Mr. Clifford has
voting and investment power).
(21)
The
address of Peter Appel is 77 Oregon Road, Bedford Corners, NY
10549.
(22)
Consists of (a)
34,965 shares of common stock held by Lone Wolf Holdings, LLC (as
to which Mr. Appel has voting and investment power); (b) 26,224
shares of common stock issuable upon the exercise of warrants held
by Lone Wolf Holdings, LLC (as to which Mr. Appel has voting and
investment power); and (c) 35,737 shares of common stock issuable
upon the conversion of a promissory note held by Lone Wolf
Holdings, LLC (as to which Mr. Appel has voting and investment
power).
(23)
The
address of Daniel Landry is 216 Avenue B, Redondo Beach, CA
90277.
(24)
Consists of (a)
54,880 shares of common stock held directly; (b) 8,811 shares of
common stock issuable upon the exercise of options held directly
that are presently exercisable; (c) 2,098 shares of common stock
issuable upon the exercise of warrants held directly that are
presently exercisable; and (d) 25,849 shares of common stock
issuable upon the conversion of a convertible promissory note held
by The Kingdom Trust Company Custodian fbo Daniel Landry (as to
which Mr. Landry has voting and investment power).
(25)
The
address of Don Miloni is 1425 E. Greenwood Lane, Greenwood Village,
CO 80121.
(26)
Consists of (a)
24,476 shares of common stock held by RCHER Financial LLC (as to
which Mr. Miloni has voting and investment power); (b) 10,490
shares of common stock issuable upon the exercise of warrants held
by RCHER Financial LLC (as to which Mr. Miloni has voting and
investment power); and (c) 51,786 shares of common stock issuable
upon the conversion of a convertible promissory note held
directly.
(27)
The
address of ENDRA Holdings LLC is 500 Boylston Street, Suite 1600,
Boston, MA 02116.The manager of
ENDRA Holdings LLC is Enlight Biosciences LLC, which also has an
address of 500 Boylston Street, Suite 1600, Boston MA 02116. Daphne
Zohar, chief executive officer of Enlight Biosciences LLC, has sole
voting and dispositive power with respect to all of Endra Holdings
LLC's 82,338 shares of common stock.
(28)
The
address of Andreas Typaldos is 666 Greenwich Street, Suite 734, New
York, NY 10014.
(29)
Consists of (a)
27,972 shares of common stock held by Andreas Typaldos LTD
Partnership (as to which Mr. Typaldos has voting and investment
power); and (b) 57,143 shares of common stock issuable upon the
conversion of a convertible promissory note held by Andreas
Typaldos LTD Partnership (as to which Mr. Typaldos has voting and
investment power).
(30)
The
address of Mark L. Baum is 1127 Cuchara Drive, Del Mar, CA
92014.
(31)
Consists of (a)
65,474 shares of common stock held by Mark Baum Trust dated May 17,
2011 (as to which Mr. Baum has voting and investment power); and
(b) 8,811 shares of common stock issuable upon the exercise of
options held by Mr. Baum that are presently
exercisable.
71
CERTAIN
RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR
INDEPENDENCE
We have
been authorized to list our common stock,
and have applied for the listing of our units and
warrants, on the Nasdaq Capital Market, therefore, our
determination of the independence of directors is made using the
definition of “independent” contained in the listing
standards of the Nasdaq Stock Market. On the basis of information
solicited from each director, the board has determined that each of
Anthony DiGiandomenico, Dr. Sanjiv Sam Gambhir, Michael Harsh and
Alexander Tokman has no material relationship with the Company and
is independent within the meaning of such rules.
SEC
regulations define the related person transactions that require
disclosure to include any transaction, arrangement or relationship
in which the amount involved exceeds the lesser of $120,000 or one
percent of the average of the Company’s total assets at year
end for the last two completed fiscal years in which we were or are
to be a participant and in which a related person had or will have
a direct or indirect material interest. A related person is: (i) an
executive officer, director or director nominee of the Company,
(ii) a beneficial owner of more than 5% of our common stock, (iii)
an immediate family member of an executive officer, director or
director nominee or beneficial owner of more than 5% of our common
stock, or (iv) any entity that is owned or controlled by any of the
foregoing persons or in which any of the foregoing persons has a
substantial ownership interest or control.
For the
period from January 1, 2015 through the date of this
prospectus (the “Reporting Period”), described below
are certain transactions or series of transactions between us and
certain related persons.
In July
2015, Mr. Thornton received 5,556 shares of the Company's common
stock for accrued salary of approximately $33,000.
On
November 31, 2015, Kevin Cotter, an ENDRA common stockholder,
transferred to Mr. Thornton warrants to purchase 1,399 shares of
the Company's common stock at an exercise price of $3.58. On
January 19, 2016, Mr. Thornton exercised these warrants, and
received an additional 1,399 warrants at an exercise price of
$14.30 as a part of the warrant exchange program.
On
January 28, 2016, we issued convertible promissory notes to Sanjiv
Gambhir (the “Gambhir Note”), Michael Harsh (the
“Harsh Note”) and Alexander Tokman (the “Tokman
Note”), each a member of our board of directors. The Gambhir
Note and the Tokman Note are each in the principal sum of $20,000
and the Harsh Note is in the principal sum of $10,000. None of the
notes accrue interest and all three are payable upon the earlier of
(1) completion by the Company of an equity financing of $4.0
million or more and (2) the one-year anniversary of the issuance
date.
In April 2016, we issued convertible promissory
notes to the following related persons: (i) Anthony DiGiandomenico,
a director of the Company, in the principal sum of $25,000, (ii) a
trust beneficially owned by Robert C. Clifford, a beneficial owner
of more than 5% of our common stock, in the principal sum of
$19,474, (iii) a trust beneficially owned by Daniel Landry, a
beneficial owner of more than 5% of our common stock, in the
principal sum of $25,000, (iv) Benjamin L. Padnos, a beneficial
owner of more than 5% of our common stock, in the principal sum of
$54,500, (v) Cynthia Padnos, an immediate family member of a
beneficial owner of more than 5% of our common stock, in the
principal sum of $12,096, (vi) Daniel Padnos, an immediate family
member of a beneficial owner of more than 5% of our common stock,
in the principal sum of $7,258, (vii) Jeffrey S. Padnos and Margaret M. Padnos
(including trusts which they beneficially own), joint beneficial
owners of more than 5% of our common stock, in the aggregate
principal sum of $96,811, (viii) Jonathan Padnos, an immediate
family member of a beneficial owner of more than 5% of our common
stock, in the principal sum of $17,258, (ix) Sivan Padnos Caspi, an
immediate family member of a beneficial owner of more than 5% of
our common stock, in the principal sum of $7,258, (x) Michael
Thornton, our Chief Technology Officer, in the principal sum of
$20,000, and (xi) Conal Thornton, the father of Michael Thornton,
our Chief Technology Officer, in the principal sum of $20,000.
These convertible promissory notes mature one year from the issue
date, accrue interest at the rate of 8% per annum, are payable at
maturity, are secured by all assets of the Company, now owned or
hereafter acquired, and automatically convert into the same
security issued by the Company, on the same terms, including price,
upon a qualified financing in the amount of $3.0 million or more.
Separately, upon any individual noteholder’s election, such
noteholder’s convertible promissory note is convertible into
shares of the Company’s common stock, and upon the election
of noteholders holding a majority of the outstanding principal
amount of the convertible promissory notes, all outstanding
convertible promissory notes are convertible into shares of the
Company’s common stock, in each case at a conversion price of
$1.00 per share. While we believe this offering will constitute a
qualified financing as described above, because the conversion
price is significantly lower than the expected public offering
price of the common stock sold in this offering, we have assumed
that the noteholders will convert all of the outstanding principal
and accrued interest on the convertible promissory notes into
shares of common stock of the Company at the lower conversion price
immediately prior to the completion of the
offering.
On
July 21, 2016, we issued convertible promissory notes to Francois
Michelon, our Chief Executive Officer, or the Michelon Note, and
Michael Thornton, our Chief Technology Officer, or the Thornton
Note. The Michelon Note and the Thornton Note are in the principal
sums of $35,000 and $52,000, respectively. The Michelon Note and
the Thornton Note mature one year from the issue date, accrue
interest at the rate of 8% per annum and are payable at maturity,
are secured by all assets of the Company, now owned or hereafter
acquired, and automatically convert into the same security issued
by the Company, on the same terms, including price, upon a
qualified financing in the amount of $3.0 million or more.
Separately, upon any individual noteholder’s election, such
noteholder’s convertible promissory note is convertible into
shares of the Company’s common stock, and upon the election
of noteholders holding a majority of the outstanding principal
amount of the convertible promissory notes, all outstanding
convertible promissory notes are convertible into shares of the
Company’s common stock, in each case at a conversion price of
$1.00 per share. While we believe this offering will constitute a
qualified financing as described above, because the conversion
price is significantly lower than the expected public offering
price of the common stock sold in this offering, we have assumed
that the noteholders will convert all of the outstanding principal
and accrued interest on the convertible promissory notes into
shares of common stock of the Company at the lower conversion price
immediately prior to the completion of the offering.
72
DESCRIPTION OF THE
SECURITIES WE ARE OFFERING
The
following is a brief description of our capital stock. This summary
does not purport to be complete in all respects. This description
is subject to and qualified entirely by the terms of our Fourth
Amended and Restated Certificate of Incorporation (the
“Certificate of Incorporation”), and our amended and
restated bylaws, each of which we plan to adopt prior to the
completion of this offering and copies of which have been filed
with the SEC and are also available upon request from
us.
Authorized Capitalization
We have
60,000,000 shares of
capital stock authorized under our Certificate of Incorporation,
consisting of 50,000,000 shares of common stock with a par value of
$0.0001 per share and
10,000,000 shares of preferred stock with a par value of
$0.0001 per share. As
of September 30, 2016, we had
1,012,723 shares of common stock outstanding held of record by 72
stockholders and no shares of preferred stock outstanding. Our
authorized but unissued shares of common and preferred stock are
available for issuance without further action by our stockholders,
unless such action is required by applicable law or the rules of
any stock exchange or automated quotation system on which our
securities may be listed or traded.
Units
Each
unit consists of one share of common stock, $0.0001 par value per
share, and a warrant to purchase one share of our common stock.
The common stock and warrants comprising each unit are not
immediately separable. The units are expected to begin
trading on or promptly after the date of this prospectus. The
common stock and warrants comprising the units will begin trading
separately on the first day following the 60th day after the
date of this prospectus, or such earlier date as may be determined
by Dougherty & Company LLC, as representative of the
underwriters, at which time trading of the units will be suspended,
the units will be delisted and only our common stock and warrants
will continue to be listed for trading on the Nasdaq Capital
Market.
Common Stock
Based
on the 1,012,723 shares of common stock outstanding as of September
30, 2016, and assuming (1) the conversion of $1,386,448 aggregate principal amount of
our convertible notes (plus accrued interest thereon as of
September 30, 2016 into
1,430,490 shares of our common stock) and a conversion date of
September 30, 2016 and (2) the
issuance by us of 2,000,000 shares of common stock in this
offering, there will be 4,443,213 shares of common stock
outstanding upon the closing of this offering (or 4,743,213
shares if the underwriters exercise their option to purchase
additional units in full).
Holders
of our common stock are entitled to such dividends as may be
declared by our board of directors out of funds legally available
for such purpose. The shares of common stock are neither redeemable
nor convertible. Holders of common stock have no preemptive or
subscription rights to purchase any of our securities.
Each
holder of our common stock is entitled to one vote for each such
share outstanding in the holder’s name. No holder
of common stock is entitled to cumulate votes in voting for
directors.
In the
event of our liquidation, dissolution or winding up, the holders of
our common stock are entitled to receive pro rata our assets, which
are legally available for distribution, after payments of all debts
and other liabilities. All of the outstanding shares of
our common stock are fully paid and non-assessable. The
shares of common stock offered by this prospectus will also be
fully paid and non-assessable.
Description
of Warrants
The following summary of certain terms and provisions of the
warrants included in the units offered hereby is not
complete and is subject to, and qualified in its entirety by, the
provisions of the warrant, the form of which has been filed as an
exhibit to the registration statement of which this prospectus is a
part. Prospective investors should carefully review the terms and
provisions of the form of warrant for a complete description of the
terms and conditions of the warrants.
Form. The warrants will be issued as
individual warrants to the investors, all of which will be governed
by a warrant agreement.
Exercisability. The warrants will
be exercisable on
the first trading day following the 60th day after the date of this
prospectus or on such date Dougherty & Company LLC, as
representative of the underwriters, determines to separate the
units, whichever date is earlier, and
following such
separation at any time up to the date that is five
years after their original issuance date. The warrants
will be exercisable, at the option of each holder, in whole or in
part by delivering to us a duly executed exercise notice and, at
any time a registration statement registering the issuance of the
shares of common stock underlying the warrants under the Securities
Act is effective and available for the issuance of such shares, or
an exemption from registration under the Securities Act is
available for the issuance of such shares, by payment in full in
immediately available funds for the number of shares of common
stock purchased upon such exercise. If a registration statement
registering the issuance of the shares of common stock underlying
the warrants under the Securities Act is not effective or available
and an exemption from registration under the Securities Act is not
available for the issuance of such shares, the holder may, in its
sole discretion, elect to exercise the warrant through a cashless
exercise, in which case the holder would receive upon such exercise
the net number of shares of common stock determined according to
the formula set forth in the warrant. No fractional shares of
common stock will be issued in connection with the exercise of a
warrant. In lieu of fractional shares, we will pay the holder an
amount in cash equal to the fractional amount multiplied by the
exercise price.
Exercise Limitation. A holder will not
have the right to exercise any portion of the warrant if the holder
(together with its affiliates) would beneficially own in excess of
4.99% of the number of shares of our common stock outstanding
immediately after giving effect to the exercise, as such percentage
ownership is determined in accordance with the terms of the
warrants. However, any holder may increase or decrease such
percentage to any other percentage not in excess of 9.99% upon at
least 61 days’ prior notice from the holder to
us.
Exercise Price. The warrants will have
an exercise price equal to 120% of the initial public offering
price per unit set forth on the cover page of this
prospectus. The exercise price is subject to appropriate adjustment
in the event of certain stock dividends and distributions, stock
splits, stock combinations, reclassifications or similar events
affecting our common stock and also upon any distributions of
assets, including cash, stock or other property to our
stockholders.
Transferability. Subject to applicable
laws, the warrants may be offered for sale, sold, transferred or
assigned without our consent.
Fundamental Transactions. In the event
of a fundamental transaction, as described in the warrants and
generally including any reorganization, recapitalization or
reclassification of our common stock, the sale, transfer or other
disposition of all or substantially all of our properties or
assets, our consolidation or merger with or into another person,
the acquisition of more than 50% of our outstanding common stock,
or any person or group becoming the beneficial owner of 50% of the
voting power represented by our outstanding common stock, the
holders of the warrants will be entitled to receive upon exercise
of the warrants the kind and amount of securities, cash or other
property that the holders would have received had they exercised
the warrants immediately prior to such fundamental
transaction.
Rights as a Stockholder. Except as
otherwise provided in the warrants or by virtue of such
holder’s ownership of shares of our common stock, the holder
of a warrant does not have the rights or privileges of a holder of
our common stock, including any voting rights, until the holder
exercises the warrant.
Waivers and Amendments. Subject to
certain exceptions, any term of the warrants may be amended or
waived with our written consent and the written consent of the
holders of at least two-thirds of the then-outstanding
warrants.
Stock
Options and Warrants
As of
September 30, 2016, we
had reserved the following shares of common stock for issuance
pursuant to stock options, warrants and equity plans:
●
213,937 shares of common stock issuable upon the
exercise of outstanding warrants, at a weighted average exercise
price of $13.53 per share;
●
213,452 shares of our common stock issuable
upon the exercise of outstanding stock options issued pursuant to
our 2016 Omnibus Incentive Plan, or our Incentive Plan, at a
weighted average exercise price of $7.15 per share and an
estimated 792,782 shares of our common stock issuable
upon the exercise of stock options expected to be granted to our
directors and certain of our officers upon the completion of this
offering at an exercise price equal to the public offering price
set forth on the cover of this prospectus; and
●
an estimated 517,643 shares of
our common stock that will be reserved for future issuance under
our Incentive Plan.
73
Convertible Promissory Notes
As of
September 30, 2016, we had reserved an estimated 1,430,490 shares
of our common stock for future issuance under convertible
promissory notes. From April to August 2016, we issued convertible
promissory notes in the aggregate principal amount of $1,386,448 to
62 accredited investors. These convertible promissory notes mature
one year from the issue date, accrue interest at the rate of 8% per
annum, are payable at maturity, are secured by all assets of the
Company, now owned or hereafter acquired, and automatically convert
into the same security issued by the Company, on the same terms,
including price, upon a qualified financing in the amount of $3.0
million or more. Separately, upon any individual noteholder’s
election, such noteholder’s convertible promissory note is
convertible into shares of the Company’s common stock, and
upon the election of noteholders holding a majority of the
outstanding principal amount of the convertible promissory notes,
all outstanding convertible promissory notes are convertible into
shares of the Company’s common stock, in each case at a
conversion price of $1.00 per share. While we believe this offering
will constitute a qualified financing as described above, because
the conversion price is significantly lower than the expected
public offering price of the common stock sold in this offering, we
have assumed that the noteholders will convert all of the
outstanding principal and accrued interest on the convertible
promissory notes into shares of common stock of the Company at the
lower conversion price immediately prior to the completion of the
offering.
Preferred Stock
Our
board of directors will have the authority, without further action
by the stockholders, to issue up to 10,000,000 shares of preferred
stock in one or more series and to fix the designations, powers,
rights, preferences, qualifications, limitations and restrictions
thereof. These designations, powers, rights and preferences could
include voting rights, dividend rights, dissolution rights,
conversion rights, exchange rights, redemption rights, liquidation
preferences, and the number of shares constituting any series or
the designation of such series, any or all of which may be greater
than the rights of common stock. The issuance of preferred stock
could adversely affect the voting power of holders of common stock
and the likelihood that such holders will receive dividend payments
and payments upon liquidation. In addition, the issuance of
preferred stock could have the effect of delaying, deferring or
preventing change in our control or other corporate action. No
shares of preferred stock are outstanding, and we have no present
plan to issue any shares of preferred stock.
GE
Healthcare Right
In
April 2016, we entered into a Collaborative Research Agreement with
General Electric Company, acting through its GE Healthcare business
unit and the GE Global Research Center, or GE Healthcare. The
agreement provides that prior to selling any equity interests in
our company to a healthcare device manufacturer, we will first
offer to negotiate in good faith to sell such equity interests to
GE Healthcare.
Anti-Takeover
Effects of Certain Provisions of Delaware Law and Our Charter
Documents
The
following is a summary of certain provisions of Delaware law, our
Certificate of Incorporation and our bylaws. This summary does not
purport to be complete and is qualified in its entirety by
reference to the corporate law of Delaware and our Certificate of
Incorporation and bylaws.
Effect of Delaware Anti-Takeover
Statute. We are subject to Section 203 of the
Delaware General Corporation Law, an anti-takeover
law. In general, Section 203 prohibits a Delaware
corporation from engaging in any business combination (as defined
below) with any interested stockholder (as defined below) for a
period of three years following the date that the stockholder
became an interested stockholder, unless:
●
prior to that date,
the board of directors of the corporation approved either the
business combination or the transaction that resulted in the
stockholder becoming an interested stockholder;
●
upon consummation
of the transaction that resulted in the stockholder becoming an
interested stockholder, the interested stockholder owned at least
85% of the voting stock of the corporation outstanding at the time
the transaction commenced, excluding for purposes of determining
the number of shares of voting stock outstanding (but not the
voting stock owned by the interested stockholder) those shares
owned by persons who are directors and officers and by excluding
employee stock plans in which employee participants do not have the
right to determine whether shares held subject to the plan will be
tendered in a tender or exchange offer; or
●
on or subsequent to
that date, the business combination is approved by the board of
directors of the corporation and authorized at an annual or special
meeting of stockholders, and not by written consent, by the
affirmative vote of at least 66 2/3% of the outstanding voting
stock that is not owned by the interested stockholder.
74
Section
203 defines “business combination” to include the
following:
●
any merger or
consolidation involving the corporation and the interested
stockholder;
●
any sale, transfer,
pledge or other disposition of 10% or more of the assets of the
corporation involving the interested stockholder;
●
subject to certain
exceptions, any transaction that results in the issuance or
transfer by the corporation of any stock of the corporation to the
interested stockholder;
●
subject to limited
exceptions, any transaction involving the corporation that has the
effect of increasing the proportionate share of the stock of any
class or series of the corporation beneficially owned by the
interested stockholder; or
●
the receipt by the
interested stockholder of the benefit of any loans, advances,
guarantees, pledges or other financial benefits provided by or
through the corporation.
In
general, Section 203 defines an interested stockholder as any
entity or person beneficially owning 15% or more of the outstanding
voting stock of the corporation, or who beneficially owns 15% or
more of the outstanding voting stock of the corporation at any time
within a three-year period immediately prior to the date of
determining whether such person is an interested stockholder, and
any entity or person affiliated with or controlling or controlled
by any of these entities or persons.
Our Charter Documents. Our
charter documents include provisions that may have the effect of
discouraging, delaying or preventing a change in control or an
unsolicited acquisition proposal that a stockholder might consider
favorable, including a proposal that might result in the payment of
a premium over the market price for the shares held by our
stockholders. Certain of these provisions are summarized
in the following paragraphs.
Effects of authorized but unissued common
stock. One of the effects of the existence of
authorized but unissued common stock may be to enable our board of
directors to make more difficult or to discourage an attempt to
obtain control of our Company by means of a merger, tender offer,
proxy contest or otherwise, and thereby to protect the continuity
of management. If, in the due exercise of its fiduciary
obligations, the board of directors were to determine that a
takeover proposal was not in our best interest, such shares could
be issued by the board of directors without stockholder approval in
one or more transactions that might prevent or render more
difficult or costly the completion of the takeover transaction by
diluting the voting or other rights of the proposed acquirer or
insurgent stockholder group, by putting a substantial voting block
in institutional or other hands that might undertake to support the
position of the incumbent board of directors, by effecting an
acquisition that might complicate or preclude the takeover, or
otherwise.
Cumulative Voting. Our
Certificate of Incorporation does not provide for cumulative voting
in the election of directors, which would allow holders of less
than a majority of the stock to elect some directors.
Vacancies. Our Certificate
of Incorporation provides that all vacancies may be
filled by the
affirmative vote of a majority of directors then in office, even if
less than a quorum.
Special Meeting of
Stockholders. A
special meeting of stockholders may only be called by the Chairman
of the board of directors, the President, the Chief Executive
Officer, or the board of directors at any time and for any purpose
or purposes as shall be stated in the notice of the meeting, or by
request of the holders of record of at least 20% of the outstanding
shares of common stock. This provision could prevent
stockholders from calling a special meeting because, unless certain
significant stockholders were to join with them, they might not
obtain the percentage necessary to request the
meeting. Therefore, stockholders holding less than 20%
of the issued and outstanding common stock, without the assistance
of management, may be unable to propose a vote on any transaction
that would delay, defer or prevent a change of control, even if the
transaction were in the best interests of our
stockholders.
Transfer
Agent and Warrant Agent
The
transfer agent of our units and common stock and
warrant agent for our warrants included in our units offered
hereby is Corporate Stock Transfer, Inc., 3200 Cherry Creek
Dr. South, Suite 430, Denver, CO 80209. Its telephone number is
(303) 282-4800.
75
SHARES
ELIGIBLE FOR FUTURE SALE
Prior
to this offering, there has been no public market for our
securities, and we cannot predict the effect, if any, that market
sales of our securities or the availability of our securities for
sale will have on the market price of our securities prevailing
from time to time. Nevertheless, sales of substantial amounts of
our common stock, including shares issued upon exercise of
outstanding options and warrants, in the public market following
this offering could adversely affect market prices prevailing from
time to time and could impair our ability to raise capital through
the sale of our equity securities.
Upon
the closing of this offering, we will have a total of 4,443,213
shares of our common stock outstanding (or 4,743,213 shares if the
underwriters exercise their option to purchase additional
units in full), and a total of 6,443,213
shares of our common stock outstanding if the warrants sold in this
offering are exercised in full (or 7,043,213 shares if
the underwriters exercise their option to purchase additional
units in full and the warrants included in such
units are exercised) based on the 2,443,213 shares of our
common stock outstanding as of September 30, 2016, assuming the
conversion immediately prior to the closing of this offering of all
convertible promissory notes outstanding as of September 30, 2016
into an aggregate of 1,430,490 shares of common stock at a
conversion price of $1.00 per share. Of these outstanding shares,
all of the 2,000,000 units sold in this offering will
be freely tradable, except that any units purchased in
this offering by our affiliates, as that term is defined in Rule
144 under the Securities Act of 1933, as amended, or the Securities
Act, would only be able to be sold in compliance with the Rule 144
limitations described below. In addition, we expect that the
warrants and the shares issued upon exercise of the warrants issued
in this offering will be freely tradeable except for any such
warrants or shares issued to our affiliates, as that term is
defined in Rule 144 under the Securities Act, which would only be
able to be sold in compliance with the Rule 144 limitations
described below.
The
remaining outstanding shares of our common stock will be deemed
“restricted securities” as defined in Rule 144.
Restricted securities may be sold in the public market only if they
are registered under the Securities Act or if they qualify for an
exemption from registration under Rule 144 or Rule 701 promulgated
under the Securities Act, which rules are summarized below. In
addition, our executive officers, directors and substantially all
of our existing stockholders have entered into lock-up agreements
with the underwriters under which they have agreed,
subject to specific exceptions, not to sell any of our common stock
or warrants for at least 180 or 365 days following the date of this
prospectus, as described below. As a result of these agreements,
subject to the provisions of Rule 144 or Rule 701, based on an
assumed offering date of September 30, 2016,
securities will be available for sale in the public
market as follows:
●
Beginning on the
date of this prospectus, all of the units sold in this
offering will be immediately available for sale in the public
market;
●
Beginning 181 days
after the date of this prospectus, approximately 350,000 shares of
common stock will become eligible for sale in the public market;
and
●
The remainder of
the shares will be eligible for sale in the public market from time
to time thereafter, subject in some cases to the volume and other
restrictions of Rule 144, as described below.
Rule
144
In
general, under Rule 144 as currently in effect, once we have been
subject to public company reporting requirements for at least 90
days, a person who is not deemed to have been one of our affiliates
for purposes of the Securities Act at any time during the 90 days
preceding a sale and who has beneficially owned the shares proposed
to be sold for at least six months, including the holding period of
any prior owner other than our affiliates, is entitled to sell
those shares without complying with the manner of sale, volume
limitation or notice provisions of Rule 144, subject to compliance
with the public information requirements of Rule 144. If such a
person has beneficially owned the shares proposed to be sold for at
least one year, including the holding period of any prior owner
other than our affiliates, then that person would be entitled to
sell those shares upon expiration of the lock-up agreements
described below, without complying with any of the requirements of
Rule 144.
76
In
general, under Rule 144, as currently in effect, our affiliates or
persons selling shares on behalf of our affiliates are entitled to
sell upon expiration of the lock-up agreements described above,
within any three-month period, a number of shares that does not
exceed the greater of:
●
1% of the number of
shares of common stock then outstanding, which will equal
approximately 44,432 shares immediately after this offering;
or
●
the average weekly
trading volume of common stock on the Nasdaq Capital Market during
the four calendar weeks preceding the filing of a notice on Form
144 with respect to that sale.
Sales
under Rule 144 by our affiliates or persons selling shares on
behalf of our affiliates are also subject to certain manner of sale
provisions and notice requirements and to the availability of
current public information about us.
Rule
701
Rule
701 generally allows a stockholder who purchased shares of our
common stock pursuant to a written compensatory plan or contract
and who is not deemed to have been an affiliate of our company
during the immediately preceding 90 days to sell these shares in
reliance upon Rule 144, but without being required to comply with
the public information, holding period, volume limitation or notice
provisions of Rule 144. Rule 701 also permits affiliates of our
company to sell their Rule 701 shares under Rule 144 without
complying with the holding period requirements of Rule 144. All
holders of Rule 701 shares, however, are required by that rule to
wait until 90 days after the date of this prospectus before selling
those shares pursuant to Rule 701, subject to the market standoff
agreements and lock-up agreements described above.
Stock
Options
As soon
as practicable after the closing of this offering, we intend to
file one or more registration statements on Form S-8 under the
Securities Act covering all of the shares of our common stock
subject to outstanding options and the shares of our common stock
reserved for issuance under our stock plans. In addition, we intend
to file a registration statement on Form S-8 or such other form as
may be required under the Securities Act for the resale of shares
of our common stock issued upon the exercise of options that were
not granted under Rule 701. We expect to file this registration
statement as soon as permitted under the Securities Act and the
terms of the lock-up agreements described below. However, the
shares registered on Form S-8 may be subject to the volume
limitations and the manner of sale, notice and public information
requirements of Rule 144 and will not be eligible for resale until
expiration of the lock-up and market standoff agreements to which
they are subject.
Lock-up
Agreements
For a
description of the lock-up agreements with the
underwriters that restrict sales of shares and
warrants by us, our executive officers, and our directors and
substantially all our shareholders see the information under the
heading “Underwriting.”
77
UNDERWRITING
We have entered
into an underwriting agreement with Dougherty &
Company LLC, acting as the representative of the several
underwriters named below, with respect to the units
subject to this offering. Subject to certain conditions, we have
agreed to sell to the underwriters, and the underwriters have
severally agreed to purchase, the number of units
provided below opposite their respective names.
|
Underwriters
|
Number of
Units
|
|
Dougherty & Company LLC
|
|
| Dawson James Securities, Inc. |
|
|
Total
|
2,000,000
|
The
underwriters are offering the units subject to their
acceptance of the units from us and subject to prior
sale. The underwriting agreement provides that the obligations of
the several underwriters to pay for and accept delivery of the
units offered by this prospectus are subject to the
approval of certain legal matters by their counsel and to certain
other conditions. The underwriters are obligated to take and pay
for all of the units if any such units
are taken. However, the underwriters are not required to take or
pay for the units covered by the underwriters’
over-allotment option described below.
Over-Allotment
Option
We have
granted the underwriters an option, exercisable for 30 days from
the date of this prospectus, to purchase up to an additional
300,000 units to cover over-allotments, if any, of the
units offered by this prospectus. If the underwriters
exercise this option, each underwriter will be obligated, subject
to certain conditions, to purchase a number of additional
units proportionate to that underwriter’s
initial purchase commitment as indicated in the table above for
which the option has been exercised.
Discount,
Commissions and Expenses
The
underwriters have advised us that they propose to offer the
units to the public at the initial public offering
price set forth on the cover page of this prospectus and to certain
dealers at that price less a concession not in excess of
$ per
unit. The underwriters may allow, and certain dealers
may reallow, a discount from the concession not in excess of
$ per
unit to certain brokers and dealers. After this
offering, the initial public offering price, concession and
reallowance to dealers may be changed by the representative. No
such change shall change the amount of proceeds to be received by
us as set forth on the cover page of this prospectus. The
units are offered by the underwriters as stated
herein, subject to receipt and acceptance by them and subject to
their right to reject any order in whole or in part. The
underwriters have informed us that they do not intend to confirm
sales to any accounts over which they exercise discretionary
authority.
We have granted
Dougherty & Company LLC a right of first refusal to serve as
co-manager or co-placement agent in connection with any future
public or private offering of securities we undertake or as agent
or advisor in the event of a sale or merger of all or a portion of
the Company occurring within one year following the effective date
of this offering. In accordance with applicable rules of FINRA,
Dougherty & Company LLC does not have more than one opportunity
to waive or terminate the right of first refusal in consideration
of any payment or fee, and any payment or fee to waive or terminate
the right of first refusal must be paid in cash and have a value
not in excess of the greater of 1% of the proceeds in this offering
(or, if greater, the maximum amount permitted by FINRA rules for
compensation in connection with this offering) or 5% of the
underwriting discounts or commissions paid in connection with any
future financing subject to the right of first refusal (including
any over-allotment option that may be exercised). This right of
first refusal is not reflected in the table
below.
The
following table shows the underwriting discount payable to the
underwriters by us in connection with this offering. Such amounts
are shown assuming both no exercise and full exercise of the
underwriters’ over-allotment option to purchase additional
units.
78
|
|
Per
Unit(1)
|
Total Without
Exercise of Over-Allotment Option
|
Total With Exercise
of Over-Allotment Option
|
|
Initial
public offering price
|
$
|
$
|
$
|
|
Underwriting
discount
|
$
|
$
|
$
|
__________________________
(1)
Does not include
the warrants to purchase shares of common stock equal to 8% of the
number of units issued in the offering to
be issued to the underwriters at the
closing.
We have
agreed to reimburse the underwriters for all accountable expenses,
subject to a cap of $150,000. Such accountable
expenses include (i) the fees and expenses of underwriters’
counsel, excluding “blue sky” fees; (ii) the fees and
expenses of the underwriters related to the road show; and (iii)
the fees, expenses and disbursements relating to background checks
of the Company's officers and directors. We estimate that
expenses payable by us in connection with this offering, other than
the underwriting discount referred to above but including the
reimbursement of the underwriters’ expenses, will be
approximately $600,000.
Underwriters’
Warrants
We have
also agreed to issue underwriters’ warrants to
purchase a number of our shares of common stock equal to an
aggregate of 8% of the number of units
issued in this offering. The warrants
will have an exercise price equal to 120% of the initial public
offering price of the units issued in
this offering and may be exercised on a cashless basis. The
warrants are not redeemable by us.
Each warrant also provides for one demand registration
of the shares of common stock underlying the warrant at our expense
and an additional demand at the warrant holder's expense during the
five year period commencing on the date of effectiveness of this
offering. The warrants will provide for adjustment in
the number and price of such warrants (and the shares
of common stock underlying such warrants) in the event
of recapitalization, merger or other fundamental transaction. The
warrants and the underlying shares of common stock
have been deemed compensation by FINRA and are therefore subject to
FINRA Rule 5110(g)(1). In accordance with FINRA Rule 5110(g)(1),
neither the underwriters’ warrants nor any
shares of our common stock issued upon exercise of the
underwriters’ warrants may be sold, transferred, assigned,
pledged, or hypothecated, or be the subject of any hedging, short
sale, derivative, put, or call transaction that would result in the
effective economic disposition of such securities by any person for
a period of 180 days immediately following the date of
effectiveness or commencement of sales of the offering pursuant to
which the underwriters’ warrants are being
issued, except the transfer of any security:
●
by operation of law
or by reason of reorganization of the Company;
●
to any FINRA member
firm participating in this offering and the officers or partners
thereof, if all securities so transferred remain subject to the
lock-up restriction described above for the remainder of the time
period;
●
if the aggregate
amount of securities of the Company held by either an underwriter
or a related person do not exceed 1% of the securities being
offered;
●
that is
beneficially owned on a pro-rata basis by all equity owners of an
investment fund, provided that no participating member manages or
otherwise directs investments by the fund, and participating
members in the aggregate do not own more than 10% of the equity in
the fund; or
●
the exercise or
conversion of any security, if all securities received remain
subject to the lock-up restriction set forth above for the
remainder of the time period.
79
No
Public Market
Prior
to this offering, there has not been a public market for our
units, common stock or warrants in the United States
and the initial public offering price for our units
will be determined through negotiations between us and the
underwriters. Among the factors to be considered in
these negotiations will be prevailing market conditions, our
financial information, market valuations of other companies that we
and the underwriters believe to be comparable to us, estimates of
our business potential, the present state of our development and
other factors deemed relevant.
No
assurance can be given that the initial public offering price will
correspond to the price at which our units will trade
in the public market subsequent to this offering or that an active
trading market for our units, common stock or warrants
will develop and continue after this offering.
Indemnification
We have
agreed to indemnify the underwriters against certain liabilities,
including liabilities under the Securities Act of 1933, as amended,
or the Securities Act, or to contribute to payments that the
underwriters may be required to make in respect of
those liabilities.
Lock-up
Agreements
We, our
officers, directors and substantially all of our existing
stockholders have agreed, subject to limited exceptions, for a
period of 365 days after the date of the underwriting agreement,
not to offer, sell, contract to sell, pledge, grant any option to
purchase, make any short sale or otherwise dispose of, directly or
indirectly any units, shares of common stock or any
securities convertible into or exchangeable for our common stock,
including warrants, either owned as of the date of the underwriting
agreement or thereafter acquired without the prior written consent
of the representative; provided, that the lock-up period applicable
to persons other than our officers and directors with respect to
shares purchased from us prior to the date hereof for a purchase
price per share of $7.15 or more is 180 days. The representative
may, in its sole discretion and at any time or from time to time
before the termination of the lock-up period release all or any
portion of the securities subject to lock-up agreements; provided,
however, that, subject to limited exceptions, at least three
business days before the release or waiver or any lock-up
agreement, the representative must notify us of the impending
release or waiver and we will be required to announce the impending
release or waiver through a major news service at least two
business days before the release or waiver.
Price
Stabilization, Short Positions and Penalty Bids
In
connection with the offering the underwriters may engage in
stabilizing transactions, over-allotment transactions, syndicate
covering transactions and penalty bids in accordance with
Regulation M under the Exchange Act:
●
Stabilizing
transactions permit bids to purchase the underlying security so
long as the stabilizing bids do not exceed a specified
maximum.
●
Over-allotment
involves sales by the underwriters of securities in
excess of the number of securities the underwriters
are obligated to purchase, which creates a syndicate short
position. The short position may be either a covered short position
or a naked short position. In a covered short position, the number
of securities over-allotted by the underwriters is not
greater than the number of securities that they may
purchase in the over-allotment option. In a naked short position,
the number of securities involved is greater than the
number of securities in the over-allotment option. The
underwriters may close out any covered short position by either
exercising their over-allotment option and/or purchasing
securities in the open market.
●
Syndicate covering
transactions involve purchases of securities in the
open market after the distribution has been completed in order to
cover syndicate short positions. In determining the source of
securities to close out the short position, the
underwriters will consider, among other things, the price of
securities available for purchase in the open market
as compared to the price at which it may purchase
securities through the over-allotment option. If the
underwriters sell more securities than could be
covered by the over-allotment option, a naked short position, the
position can only be closed out by buying securities
in the open market. A naked short position is more likely to be
created if the underwriters are concerned that there could be
downward pressure on the price of the securities in
the open market after pricing that could adversely affect investors
who purchase in the offering.
●
Penalty bids permit
the representative to reclaim a selling concession from a syndicate
member when the securities originally sold by the
syndicate member is purchased in a stabilizing or syndicate
covering transaction to cover syndicate short
positions.
80
These
stabilizing transactions, syndicate covering transactions and
penalty bids may have the effect of raising or maintaining the
market price of our securities or preventing or
retarding a decline in the market price of the
securities. As a result, the price of our
securities may be higher than the price that might
otherwise exist in the open market. Neither we nor the underwriters
make any representation or prediction as to the direction or
magnitude of any effect that the transactions described above may
have on the price of the securities. In addition,
neither we nor the underwriters make any representations that the
underwriters will engage in these stabilizing transactions or that
any transaction, once commenced, will not be discontinued without
notice.
Listing
and Transfer and Warrant Agent
Our
common stock has been approved for listing on the
Nasdaq Capital Market under the trading symbol
"NDRA". We
have applied to list our units and warrants on the Nasdaq Capital
Market under the symbols “NDRAU” and
“NDRAW,” respectively. The transfer agent
of our units and common stock is Corporate Stock
Transfer, Inc., 3200 Cherry Creek Dr. South, Suite 430, Denver, CO
80209. Its telephone number is (303) 282-4800. Corporate Stock
Transfer, Inc. will also act as the warrant agent for the
warrants.
Electronic
Distribution
This
prospectus in electronic format may be made available on websites
or through other online services maintained by the
underwriters, or by their affiliates.
Other than this prospectus in electronic format, the information on
any underwriter’s website and any information
contained in any other website maintained by any
underwriter is not part of this prospectus or the registration
statement of which this prospectus forms a part, has not been
approved and/or endorsed by us or the underwriters in
their capacity as underwriters, and
should not be relied upon by investors.
Other
From
time to time, certain of the underwriters and/or their affiliates
have provided, and may in the future provide, various investment
banking and other financial services for us for which services they
have received and, may in the future receive, customary fees. In
the course of their businesses, the underwriters and their
affiliates may actively trade our securities or loans for their own
account or for the accounts of customers, and, accordingly, the
underwriters and their affiliates may at any time hold long or
short positions in such securities or loans. Except for services
provided in connection with this offering, no underwriter has
provided any investment banking or other financial services to us
during the 180-day period preceding the date of this prospectus and
we do not expect to retain any underwriter to perform any
investment banking or other financial services for at least 90 days
after the date of this prospectus.
Notice
to Investors
Notice
to Investors in the United Kingdom
In
relation to each Member State of the European Economic Area which
has implemented the Prospectus Directive, each a Relevant Member
State, an offer to the public of any securities which are the
subject of the offering contemplated by this prospectus may not be
made in that Relevant Member State except that an offer to the
public in that Relevant Member State of any such securities may be
made at any time under the following exemptions under the
Prospectus Directive, if they have been implemented in that
Relevant Member State:
81
(a)
to legal entities
which are authorized or regulated to operate in the financial
markets or, if not so authorized or regulated, whose corporate
purpose is solely to invest in securities;
(b)
to any legal entity
which has two or more of (1) an average of at least 250 employees
during the last financial year; (2) a total balance sheet of more
than €43,000,000 and (3) an annual net turnover of more than
€50,000,000, as shown in its last annual or consolidated
accounts;
(c)
by the underwriter
to fewer than 100 natural or legal persons (other than qualified
investors as defined in the Prospectus Directive); or
(d)
in any other
circumstances falling within Article 3(2) of the Prospectus
Directive, provided that no such offer of these securities shall
result in a requirement for the publication by the issuer or the
underwriter of a prospectus pursuant to Article 3 of the Prospectus
Directive.
For the
purposes of this provision, the expression an “offer to the
public” in relation to any of the securities in any Relevant
Member State means the communication in any form and by any means
of sufficient information on the terms of the offer and any such
securities to be offered so as to enable an investor to decide to
purchase any such securities, as the same may be varied in that
Member State by any measure implementing the Prospectus Directive
in that Member State and the expression “Prospectus
Directive” means Directive 2003/71/EC and includes any
relevant implementing measure in each Relevant Member
State.
Each
underwriter has represented, warranted and agreed
that:
(a)
it has only
communicated or caused to be communicated and will only communicate
or cause to be communicated any invitation or inducement to engage
in investment activity (within the meaning of section 21 of the
Financial Services and Markets Act 2000 (the FSMA)) received by it
in connection with the issue or sale of any of the securities in
circumstances in which section 21(1) of the FSMA does not apply to
the issuer; and
(b)
it has complied
with and will comply with all applicable provisions of the FSMA
with respect to anything done by it in relation to the securities
in, from or otherwise involving the United Kingdom.
European Economic Area
In
particular, this document does not constitute an approved
prospectus in accordance with European Commission’s
Regulation on Prospectuses no. 809/2004 and no such prospectus is
to be prepared and approved in connection with this offering.
Accordingly, in relation to each Member State of the European
Economic Area which has implemented the Prospectus Directive (being
the Directive of the European Parliament and of the Council
2003/71/EC and including any relevant implementing measure in each
Relevant Member State) (each, a Relevant Member State), with effect
from and including the date on which the Prospectus Directive is
implemented in that Relevant Member State (the Relevant
Implementation Date) an offer of securities to the public may not
be made in that Relevant Member State prior to the publication of a
prospectus in relation to such securities which has been approved
by the competent authority in that Relevant Member State or, where
appropriate, approved in another Relevant Member State and notified
to the competent authority in that Relevant Member State, all in
accordance with the Prospectus Directive, except that it may, with
effect from and including the Relevant Implementation Date, make an
offer of securities to the public in that Relevant Member State at
any time:
●
to legal entities
which are authorized or regulated to operate in the financial
markets or, if not so authorized or regulated, whose corporate
purpose is solely to invest in securities;
●
to any legal entity
which has two or more of (1) an average of at least 250 employees
during the last financial year; (2) a total balance sheet of more
than €43,000,000; and (3) an annual net turnover of more than
€50,000,000, as shown in the last annual or consolidated
accounts; or
●
in any other
circumstances which do not require the publication by the Issuer of
a prospectus pursuant to Article 3 of the Prospectus
Directive.
For the
purposes of this provision, the expression an “offer of
securities to the public” in relation to any of the
securities in any Relevant Member State means the communication in
any form and by any means of sufficient information on the terms of
the offer and the securities to be offered so as to enable an
investor to decide to purchase or subscribe for the securities, as
the same may be varied in that Member State by any measure
implementing the Prospectus Directive in that Member State. For
these purposes the shares of our common stock and warrants offered
hereby are “securities.”
Notice to Prospective Investors in
Canada
The units may be sold only to purchasers purchasing,
or deemed to be purchasing, as principal that are accredited
investors, as defined in National Instrument
45-106 Prospectus
Exemptions or subsection
73.3(1) of the Securities
Act (Ontario), and are
permitted clients, as defined in National Instrument
31-103 Registration Requirements,
Exemptions and Ongoing Registrant Obligations. Any resale of the units must be
made in accordance with an exemption from, or in a transaction not
subject to, the prospectus requirements of applicable securities
laws.
Securities legislation in certain provinces or territories of
Canada may provide a purchaser with remedies for rescission or
damages if this prospectus (including any amendment thereto)
contains a misrepresentation, provided that the remedies for
rescission or damages are exercised by the purchaser within the
time limit prescribed by the securities legislation of the
purchaser’s province or territory. The purchaser should refer
to any applicable provisions of the securities legislation of the
purchaser’s province or territory for particulars of these
rights or consult with a legal advisor.
82
LEGAL MATTERS
The
validity of the units offered hereby will be passed
upon for us by K&L Gates LLP, Charlotte, North Carolina. Faegre
Baker Daniels LLP, Minneapolis, Minnesota, has acted as counsel for
the underwriters in connection with certain legal matters related
to this offering.
EXPERTS
The
financial statements of ENDRA
Life Sciences Inc. as of December 31, 2015 and December
31, 2014 included in this prospectus have been audited by RBSM LLP,
independent registered public accounting firm. We have included
these financial statements in this prospectus in reliance upon the
report of RBSM LLP, given on their authority as experts in
accounting and auditing.
WHERE YOU CAN FIND MORE
INFORMATION
We have
filed with the SEC a registration statement on Form S-1, including
exhibits, under the Securities Act that registers the
units be sold in this offering. This prospectus does
not contain all the information contained in the registration
statement and the exhibits filed as part of the registration
statement. For further information with respect to us and our
securities, we refer you to the registration statement
and the exhibits filed as part of the registration statement.
Statements contained in this prospectus as to the contents of any
contract or other document are not necessarily complete. If a
contract or document has been filed as an exhibit to the
registration statement, we refer you to the copies of the contract
or document that has been filed. Each statement in this prospectus
relating to a contract or document filed as an exhibit is qualified
in all respects by the filed exhibit.
Upon
the consummation of this offering, we will file annual, quarterly
and current reports, proxy statements and other information with
the SEC under the Exchange Act. You can read our SEC filings,
including the registration statement, at the SEC’s website at
www.sec.gov.
You may
read and copy this information at the SEC’s Public Reference
Room at 100 F Street, N.E., Washington D.C. 20549, at prescribed
rates. You may obtain information regarding the operation of the
public reference room by calling the SEC at
1-800-SEC-0330.
The SEC
also maintains a website (http://www.sec.gov) that contains
reports, proxy and information statements and other information
regarding issuers that file electronically with the
SEC.
Our
website can be accessed at www.endrainc.com. The information
contained on, or that may be obtained from, our website is not, and
shall not be deemed to be, a part of this prospectus.
The
representations, warranties and covenants made by us in any
agreement that is filed as an exhibit to the registration statement
of which this prospectus is a part were made solely for the benefit
of the parties to such agreement, including, in some cases, for the
purpose of allocating risk among the parties to such agreements,
and should not be deemed to be a representation, warranty or
covenant to you. Moreover, such representations, warranties or
covenants were made as of an earlier date. Accordingly, such
representations, warranties and covenants should not be relied on
as accurately representing the current state of our
affairs.
This
prospectus includes statistical and other industry and market data
that we obtained from industry publications and research, surveys
and studies conducted by third parties. Industry publications and
third-party research, surveys and studies generally indicate that
they have gathered their information from sources they believe to
be reliable, although they do not guarantee the accuracy or
completeness of such information. While we believe that these
industry publications and third-party research, surveys and studies
are reliable, we have not independently verified such
data.
83
ENDRA Life
Sciences Inc.
|
Report
of Independent Registered Public Accounting Firm
|
F-2
|
|
Balance sheets as
of December 31, 2015 and 2014
|
F-3
|
|
Statements of
operations for the years ended December 31, 2015 and
2014
|
F-4
|
|
Statement of
stockholders’ equity for the years ended December 31, 2015
and 2014
|
F-5
|
|
Statements of cash
flows for the years ended December 31, 2015 and 2014
|
F-6
|
|
Notes
to the unaudited financial statements
|
F-7
|
F-1
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING
FIRM
To the Board of
Directors and Stockholders of
ENDRA Life Sciences
Inc.
We have audited the
accompanying balance sheets of ENDRA Life Sciences Inc. (the
“Company”) as of December 31, 2015 and 2014 and the
related statements of operations, stockholders’ equity and
cash flows for each of the years in the two-year period ended
December 31, 2015. The Company’s management is responsible
for these financial statements. Our responsibility is to express an
opinion on these financial statements based on our
audits.
We conducted our
audits in accordance with the standards of the Public Company
Accounting Oversight Board (United States). Those standards require
that we plan and perform the audit to obtain reasonable assurance
about whether the financial statements are free of material
misstatement. The Company is not required to have, nor were we
engaged to perform an audit of the Company’s internal control
over financial reporting. Our audit included consideration of
internal control over financial reporting as a basis for designing
audit procedures that are appropriate in the circumstances, but not
for the purpose of expressing an opinion on the effectiveness of
the Company’s internal control over financial reporting.
Accordingly, we express no such opinion. An audit includes
examining, on a test basis, evidence supporting the amounts and
disclosures in the consolidated financial statements, assessing the
accounting principles used and significant estimates made by
management, as well as evaluating the overall financial statement
presentation. We believe that our audits provide a reasonable basis
for our opinion.
In our opinion, the
financial statements referred to above present fairly, in all
material respects, the financial position of ENDRA Life Sciences
Inc. as of December 31, 2015 and 2014 and the results of its
operations and cash flows for each of the years in the two-year
period ended December 31, 2015 in conformity with accounting
principles generally accepted in the United States of
America.
The accompanying
financial statements have been prepared assuming that the Company
will continue as a going concern. As discussed in Note 2 to the
financial statements, the Company has suffered losses from
operations, which raise substantial doubt about the Company’s
ability to continue as a going concern. Management’s plans in
regard to these matters are also described in Note 2. The financial
statements do not include any adjustments that might result from
the outcome of this uncertainty.
As described in
Note 3, the financial statements for the years ended December 31,
2015 and 2014 have been restated. We audited the adjustments
described in Note 3 that were applied to restate 2015 and 2014
financial statements. In our opinion, such adjustments are
appropriate and have been properly applied.
/s/ RBSM
LLP
Henderson,
Nevada
July 15, 2016,
except for the effect of the restatements discussed in Note 3 to
the financial statements, for which the date is January 19, 2017
and except for Note 11, as to which the date is December 16,
2016.
F-2
ENDRA
LIFE SCIENCES
INC.
BALANCE
SHEETS
|
|
December
31,
|
December
31,
|
|
Assets
|
2015
|
2014
|
|
Assets
|
(Restated)
|
(Restated)
|
|
Cash
|
$ 19,128
|
$ 156,442
|
|
Inventory
|
99,004
|
238,988
|
|
Other current
asset
|
8,486
|
8,986
|
|
Total Current
Assets
|
126,618
|
404,416
|
|
Other
Assets
|
|
|
|
Fixed assets,
net
|
360,104
|
298,518
|
|
Total
Assets
|
$ 486,722
|
$ 702,934
|
|
|
|
|
|
Liabilities
and Stockholders’ Equity
|
|
|
|
Current
Liabilities
|
|
|
|
Accounts payable
and accrued liabilities
|
$ 236,421
|
$ 97,601
|
|
Total Current
Liabilities
|
236,421
|
97,601
|
|
Total
Liabilities
|
236,421
|
97,601
|
|
|
|
|
|
Stockholders’
Equity
|
|
|
|
Preferred stock,
$0.0001 par value; 10,000,000 shares authorized; no shares issued
or outstanding
|
--
|
--
|
|
Common stock,
$0.0001 par value; 50,000,000 shares authorized; 2,528,311 and
2,002,336 shares
issued and
outstanding
|
253
|
200
|
|
Additional paid in
capital
|
9,948,151
|
8,060,032
|
|
Stock
payable
|
45,000
|
9,000
|
|
Accumulated
deficit
|
(9,743,104)
|
(7,463,899)
|
|
Total
Stockholders’ Equity
|
250,300
|
605,333
|
|
Total
Liabilities and Stockholders’ Equity
|
$ 486,722
|
$ 702,934
|
The
accompanying notes are an integral part of these financial
statements.
F-3
ENDRA LIFE SCIENCES
INC.
STATEMENTS
OF OPERATIONS
|
|
Year
Ended
|
Year
Ended
|
|
|
December
31,
2015
|
December
31,
2014
|
|
|
(Restated)
|
(Restated)
|
|
Revenue
|
$ 1,410,064
|
$ 559,355
|
|
|
|
|
|
Cost of Goods
Sold
|
666,233
|
259,145
|
|
|
|
|
|
Gross
Profit
|
743,831
|
300,210
|
|
|
|
|
|
Operating
Expenses
|
|
|
|
Research and
development
|
1,038,878
|
873,167
|
|
Sales and
marketing
|
50,635
|
56,298
|
|
General and
administrative
|
1,222,888
|
896,926
|
|
Total operating
expenses
|
2,312,402
|
1,826,391
|
|
|
|
|
|
Operating
loss
|
(1,568,571)
|
(1,526,181)
|
|
|
|
|
|
Other
Expenses
|
|
|
|
Loss on warrant
exercise
|
(711,343)
|
--
|
|
Loss on notes
conversion
|
--
|
(639,178)
|
|
Other income
(expense)
|
709
|
(15,519)
|
|
Total other income
(expense)
|
(710,634)
|
(654,697)
|
|
|
|
|
|
Net Loss before
Taxes
|
(2,279,204)
|
(2,180,878)
|
|
|
|
|
|
Provision for
income taxes
|
--
|
--
|
|
|
|
|
|
Net
Loss
|
$ (2,279,204)
|
$ (2,180,878)
|
|
|
|
|
|
Net
loss per share – basic and diluted
|
$ (0.98)
|
$ (1.35)
|
|
|
|
|
|
Weighted
average common shares – basic and diluted
|
2,320,045
|
1,612,569
|
The
accompanying notes are an integral part of these financial
statements.
F-4
ENDRA
LIFE SCIENCES
INC.
STATEMENT
OF STOCKHOLDERS’ EQUITY
(Restated)
|
|
Common
stock
|
Additional
|
|
|
Total
|
|
|
|
Shares
|
Amount
|
Paid
in
Capital
|
Stock to be
Issued
|
Accumulated
Deficit
|
Stockholders'
Equity
|
|
Balance
as of December 31, 2013
|
1,050,469
|
$105
|
$5,281,355
|
$-
|
$(5,283,021)
|
$(1,561)
|
|
Common stock
issued for cash
|
611,900
|
61
|
1,750,954
|
-
|
-
|
1,751,015
|
|
Common stock
issued to placement agent
|
68,182
|
7
|
(7)
|
-
|
-
|
-
|
|
Common stock
issued for notes conversion
|
271,785
|
27
|
777,237
|
-
|
-
|
777,264
|
|
Common stock
to be issued for services
|
-
|
-
|
-
|
9,000
|
-
|
9,000
|
|
Fair value of
vested stock options
|
-
|
-
|
250,493
|
-
|
-
|
250,493
|
|
Net
loss
|
-
|
-
|
-
|
-
|
(2,180,878)
|
(2,180,878)
|
|
Balance
as of December 31, 2014
|
2,002,336
|
200
|
8,060,032
|
9,000
|
(7,463,899)
|
605,333
|
|
Common stock
issued for cash
|
87,415
|
9
|
249,991
|
-
|
-
|
250,000
|
|
Common stock
issued for exercise of warrants
|
412,045
|
41
|
589,183
|
-
|
-
|
589,224
|
|
Common stock
issued for accrued salaries - related parties
|
26,515
|
3
|
63,765
|
-
|
-
|
63,768
|
|
Common stock
to be issued for services
|
-
|
-
|
-
|
36,000
|
-
|
36,000
|
|
Additional
warrants issued during exchange
|
-
|
-
|
686,343
|
-
|
-
|
686,343
|
|
Loss on
exercise of warrant
|
-
|
-
|
25,000
|
-
|
-
|
25,000
|
|
Fair value of
vested stock options
|
-
|
-
|
273,837
|
-
|
-
|
273,837
|
|
Net
loss
|
-
|
-
|
-
|
-
|
(2,279,204)
|
(2,279,204)
|
|
Balance
as of December 31, 2015 (restated)
|
2,528,311
|
$253
|
$9,948,151
|
$45,000
|
$(9,743,104)
|
$250,300
|
The
accompanying notes are an integral part of these financial
statements.
F-5
ENDRA
LIFE SCIENCES
INC.
STATEMENTS
OF CASH FLOWS
|
|
Year
Ended
|
Year
Ended
|
|
|
December
31,
|
December
31,
|
|
|
2015
|
2014
|
|
Cash
Flows from Operating Activities
|
(Restated)
|
(Restated)
|
|
Net
loss
|
$ (2,279,204)
|
$ (2,180,878)
|
|
Adjustments to
reconcile net loss to net cash used in operating
activities:
|
|
|
|
Depreciation and
amortization
|
72,225
|
37,242
|
|
Common stock issued
for services
|
309,837
|
259,493
|
|
Additional warrants
issued during exchange
|
686,343
|
-
|
|
Loss on warrant
exercise
|
25,000
|
-
|
|
Loss on common
stock issued for note conversion
|
-
|
639,178
|
|
Changes in
operating assets and liabilities:
|
|
|
|
(Increase)/Decrease
in inventory
|
139,984
|
(60,212)
|
|
(Increase)/Decrease
in other asset
|
500
|
(5,986)
|
|
Increase/(Decrease)
in accounts payable and accrued liabilities
|
202,588
|
(86,006)
|
|
Net cash used in
operating activities
|
(842,727)
|
(1,397,229)
|
|
|
|
|
|
Cash
Flows from Investing Activities
|
|
|
|
Purchases of fixed
assets
|
(133,811)
|
(275,371)
|
|
Net cash used in
investing activities
|
(133,811)
|
(275,371)
|
|
|
|
|
|
Cash
Flows from Financing Activities
|
|
|
|
Proceeds from
issuance of common stock
|
839,224
|
1,751,015
|
|
Net cash provided
by financing activities
|
839,224
|
1,751,015
|
|
|
|
|
|
Net
Increase/(Decrease) in cash
|
(137,314)
|
78,415
|
|
|
|
|
|
Cash, beginning of
period
|
156,442
|
78,027
|
|
|
|
|
|
Cash,
end of period
|
$19,128
|
$156,442
|
|
|
|
|
|
Supplemental
disclosures:
|
|
|
|
Interest
paid
|
$-
|
$-
|
|
Income
tax paid
|
$-
|
$-
|
|
|
|
|
|
Supplemental
disclosures of non-cash Items:
|
|
|
|
Common shares
issued for notes conversion
|
$-
|
$777,264
|
|
Common shares
issued for accrued salaries - related parties
|
$63,768
|
$-
|
The
accompanying notes are an integral part of these financial
statements.
F-6
ENDRA LIFE SCIENCES INC.
NOTES TO FINANCIAL STATEMENTS
For the
years ended December 31, 2015 (restated) and December 31, 2014
(restated)
Note 1 – Nature of the Business
ENDRA
Life Sciences Inc. (“Endra” or the
“Company”) was incorporated on July 18, 2007 as a
Delaware corporation.
Endra
has developed a medical imaging technology based on the
thermoacoustic effect that significantly improves the sensitivity
and specificity of clinical ultrasound.
Note 2 – Summary of Significant Accounting
Policies
The
significant accounting policies applied in the annual financial
statements of the Company as of December 31, 2015 and 2014 are
applied consistently in these financial statements.
Basis of Presentation
The
accompanying financial statements have been prepared in accordance
with the U.S. generally accepted accounting
principles.
Use of Estimates
The
preparation of the financial statements in conformity with
accounting principles generally accepted in the United States
requires management to make estimates and assumptions that affect
the reported amounts of assets and liabilities, and disclosure of
contingent liabilities at the date of the financial statements and
the reported amounts of expenses during the reporting period.
Actual results could differ from those estimates.
Management
makes estimates that affect certain accounts including deferred
income tax assets, accrued expenses, fair value of equity
instruments and reserves for any other commitments or
contingencies. Any adjustments applied to estimates are recognized
in the period in which such adjustments are
determined.
Cash and Cash Equivalents
The
Company considers all cash on hand and in banks, including accounts
in book overdraft positions, certificates of deposit and other
highly-liquid investments with maturities of three months or less,
when purchased, to be cash and cash equivalents. As of December 31,
2015 and 2014 the Company had no cash equivalents.
Inventory
The Company’s
inventory is stated at the lower of cost or market, with cost being
determined on the first-in, first-out (“FIFO”) method.
The Company continuously evaluates the composition of its
inventory, assessing slow-turning product. As of December 31, 2015,
an inventory reserve had not been deemed necessary, and therefore,
not recorded. As of December 31, 2015 and December 31, 2014,
inventory balances were $99,004 and $238,988
respectively.
Capitalization of Fixed Assets
The
Company capitalizes expenditures related to property and equipment,
subject to a minimum rule, that have a useful life greater than one
year for: (1) assets purchased; (2) existing assets that are
replaced, improved or the useful lives have been extended; or (3)
all land, regardless of cost. Acquisitions of new assets,
additions, replacements and improvements (other than land) costing
less than the minimum rule in addition to maintenance and repair
costs, including any planned major maintenance activities, are
expensed as incurred.
Capitalization of Intangible Assets
The
Company records the purchase of intangible assets not purchased in
a business combination in accordance with the ASC Topic
350.
Revenue Recognition
The
Company’s recognizes revenue in accordance with the
requirements of ASC 605-10-599, which directs that it should
recognize revenue when (1) persuasive evidence of an arrangement
exists (contracts); (2) delivery has occurred; (3) the
seller’s price is fixed or determinable (per the
customer’s contract); and (4) collectability is reasonably
assured (based upon our credit policy). For products sold to end
users revenue is recognized when title has passed to the customer
and collectability is reasonably assured; and no further efforts
are required. Future revenue from anticipated new products will
follow this same policy.
F-7
For our preclinical
NEXUS product line, our distributors have no rights of return or
price protection, or similar rights. For our TAEUS product line, we
expect to arrange similar distributor agreements and terms and
conditions with distributors for our clinical products, as we have
for our preclinical products.
Advertising Expense
The
cost of advertising is expensed as
incurred. Advertising expense for the year ended December
31, 2015 were approximately $12,894. Advertising expense for
the year ended December 31, 2014 were approximately
$7,223.
Income Taxes
The
Company utilizes ASC 740, “Income Taxes,” which
requires the recognition of deferred tax assets and liabilities for
the expected future tax consequences of events that have been
included in the financial statements or tax returns. Under this
method, deferred tax assets and liabilities are determined based on
the difference between the tax basis of assets and liabilities and
their financial reporting amounts based on enacted tax laws and
statutory tax rates applicable to the periods in which the
differences are expected to affect taxable income. A valuation
allowance is recorded when it is “more likely-than-not”
that a deferred tax asset will not be realized.
The
Company generated a deferred tax asset through net operating loss
carry-forwards. However, a valuation allowance of 100% has been
established due to the uncertainty of the Company’s
realization of the net operating loss carry forward prior to its
expiration.
Research and Development Costs
The
Company follows ASC 730-10, “Research and Development”.
Research and development costs are charged to the statement of
operations as incurred. During the years ended December 31, 2015
and December 31, 2014 the Company incurred $1,038,878 and $873,167
of expenses related to research and development costs,
respectively.
Net Earnings (Loss) Per Common Share
The
Company computes earnings per share under ASC Subtopic 260-10,
Earnings Per Share (“ASC 260-10”). Basic earnings
(loss) per share is computed by dividing the net income (loss)
attributable to the common stockholders (the numerator) by the
weighted average number of shares of common stock outstanding (the
denominator) during the reporting periods. Diluted loss per
share is computed by increasing the denominator by the weighted
average number of additional shares that could have been
outstanding from securities convertible into common stock (using
the “treasury stock” method), unless their effect on
net loss per share is anti-dilutive. There were 1,332,921 and
1,180,970 potentially dilutive shares, which include outstanding
common stock options and warrants, for the years ended December 31,
2015 and 2014.
The
potential shares, which are excluded from the determination of
basic and diluted net loss per share as their effect is
anti-dilutive, are as follows:
|
|
December 31,
2015
|
December 31,
2014
|
|
Options to purchase
common stock
|
499,012
|
301,165
|
|
Warrants to
purchase common stock
|
833,909
|
879,805
|
|
Potential
equivalent shares excluded
|
1,332,921
|
1,180,970
|
F-8
Fair Value Measurements
Disclosures about
fair value of financial instruments require disclosure of the fair
value information, whether or not recognized in our consolidated
balance sheet, where it is practicable to estimate that value. As
of December 31, 2014, the amounts reported for cash, accrued
liabilities and accrued interest approximated fair value because of
their short maturities.
In
accordance with ASC Topic 820, “Fair Value Measurements and
Disclosures,” we measure certain financial instruments at
fair value on a recurring basis. ASC Topic 820 defines fair value,
established a framework for measuring fair value in accordance with
accounting principles generally accepted in the United States, and
expands disclosures about fair value measurements.
Fair
value is defined as the price that would be received to sell an
asset or paid to transfer a liability in an orderly transaction
between market participants at the measurement date. ASC Topic 820
established a three-tier fair value hierarchy, which prioritizes
the inputs used in measuring fair value. The hierarchy gives the
highest priority to unadjusted quoted prices in active markets for
identical assets or liabilities (level 1 measurements) and the
lowest priority to unobservable inputs (level 3 measurements).
These tiers include:
|
|
●
|
Level
1, defined as observable inputs such as quoted prices for identical
instruments in active markets;
|
|
|
|
|
|
|
●
|
Level
2, defined as inputs other than quoted prices in active markets
that are either directly or indirectly observable such as quoted
prices for similar instruments in active markets or quoted prices
for identical or similar instruments in markets that are not
active; and
|
|
|
|
|
|
|
●
|
Level
3, defined as unobservable inputs in which little or no market data
exists, therefore requiring an entity to develop its own
assumptions, such as valuations derived from valuation techniques
in which one or more significant inputs or significant value
drivers are unobservable.
|
Share-based Compensation
The
Company’s 2016 Omnibus Incentive Plan, which has been approved by its board of directors,
permits the grant of share options and shares to its employees and
consultants for up to an estimated 1,318,182 shares of common stock. The
Company records share-based compensation in accordance with the
provisions of the Share-based Compensation Topic of the FASB
Codification. The guidance requires the use of option-pricing
models that require the input of highly subjective assumptions,
including the option’s expected life and the price volatility
of the underlying stock. The fair value of each option grant is
estimated on the date of grant using the Black-Scholes option
valuation model, and the resulting charge is expensed using the
straight-line attribution method over the vesting period. The
Company has elected to use the calculated value method to account
for the options it issued in 2014. A nonpublic entity that is
unable to estimate the expected volatility of the price of its
underlying share may measure awards based on a “calculated
value,” which substitutes the volatility of appropriate
public companies (representative of the company’s size and
industry) as a bench mark for the volatility of the entity’s
own share price. Currently, there is no active market for the
company’s common shares. The Company has used the historical
closing values of these companies to estimate volatility, which was
calculated to be 90%.
Stock
compensation expense recognized during the period is based on the
value of share-based awards that were expected to vest during the
period adjusted for estimated forfeitures. The estimated fair value
of grants of stock options and warrants to non-employees of the
Company is charged to expense, if applicable, in the financial
statements. These options vest in the same manner as the employee
options granted under each of the option plans as described
above.
F-9
Beneficial Conversion Feature
If the
conversion features of conventional convertible debt provides for a
rate of conversion that is below market value, this feature is
characterized as a beneficial conversion feature
(“BCF”). A BCF is recorded by the Company as a debt
discount pursuant to ASC Topic 470-20 “Debt with Conversion
and Other Options.” In those circumstances, the convertible
debt is recorded net of the discount related to the BCF and the
Company amortizes the discount to interest expense over the life of
the debt using the effective interest method.
Debt Discount
The
Company determines if the convertible debenture should be accounted
for as liability or equity under ASC 480, Liabilities —
Distinguishing Liabilities from Equity. ASC 480, applies to certain
contracts involving a company’s own equity, and requires that
issuers classify the following freestanding financial instruments
as liabilities. Mandatorily redeemable financial instruments,
obligations that require or may require repurchase of the
issuer’s equity shares by transferring assets (e.g., written
put options and forward purchase contracts), and certain
obligations where at inception the monetary value of the obligation
is based solely or predominantly on:
●
A fixed monetary
amount known at inception, for example, a payable settleable with a
variable number of the issuer’s equity shares with an
issuance date fair value equal to a fixed dollar
amount;
●
Variations in
something other than the fair value of the issuer’s equity
shares, for example, a financial instrument indexed to the S&P
500 and settleable with a variable number of the issuer’s
equity shares; or
●
Variations
inversely related to changes in the fair value of the
issuer’s equity shares, for example, a written put that could
be net share settled.
If the
entity determined the instrument meets the guidance under ASC 480
the instrument is accounted for as a liability with a respective
debt discount. The Company records debt discounts in connection
with raising funds through the issuance of promissory notes (see
Note 8). These costs are amortized to non-cash interest expense
over the life of the debt. If a conversion of the underlying debt
occurs, a proportionate share of the unamortized amounts is
immediately expensed.
Going Concern
The
Company’s financial statements are prepared using U.S. GAAP
applicable to a going concern, which contemplates the realization
of assets and liquidation of liabilities in the normal course of
business. The Company has limited operating history and had a
cumulative net loss from inception to December 31, 2015 of
$9,743,104. The Company has a working deficit of
$109,803 as of December 31, 2015. The Company has not
yet established an ongoing source of revenue sufficient to cover
its operating costs and to allow it to continue as a going concern.
The accompanying financial statements for the period ended December
31, 2015, have been prepared assuming the Company will continue as
a going concern. The Company believes its cash resources are
insufficient to meet its anticipated needs during the next twelve
months. The Company will require additional financing to fund its
future planned operations, including research and development and
commercialization of its products.
The
ability of the Company to continue as a going concern is dependent
on the Company obtaining adequate capital to fund operating losses
until it establishes a revenue stream and becomes profitable.
Management’s plans to continue as a going concern include
raising additional capital through borrowing and sales of common
stock. However, management cannot provide any assurances that the
Company will be successful in accomplishing any of its plans. If
the Company is not able to obtain the necessary additional
financing on a timely basis, the Company will be forced to delay or
scale down some or all of its development activities or perhaps
even cease the operation of its business. The ability of the
Company to continue as a going concern is dependent upon its
ability to successfully secure other sources of financing and
attain profitable operations. The accompanying financial statements
do not include any adjustments that might be necessary if the
Company is unable to continue as a going concern.
F-10
Reclassifications
Certain
reclassifications have been made to the 2015 financial statement
amounts and disclosures. A portion of wages and related expenses
for the years ended December 31, 2015 and 2014 of $665,501 and
$607,823, respectively, have been reclassified in the financial
statements from general and administrative to research and
development expenses. In addition, accrued salaried of $78,565 were
reclassified from stock payable to accrued
liabilities.
Note
3 – Restatement
●
Due to additional accrued salary and
reclassification of accrued salary from stock payable to accrued
liabilities, the Company has restated its balance sheet,
statement of operations, statement of stockholders’ equity
and statement of cash flows for the year ended December 31, 2015 to
account for the following:
o
Increase of accrued
liabilities of $150,734 in 2015 and
decrease of accrued liabilities of $13,695 in 2014;
and
o
Reduction of stock
payable by $78,565.
●
Due to a revision of its inventory and cost of
goods sold, the Company has restated its balance sheets,
statements of operations, statement of stockholders’ equity
and statements of cash flows for the years ended December 31, 2015
and December 31, 2014 to account for the
following:
o
Increase of
inventory of $99,004 and $238,988,
respectively;
o
Increase to
fixed assets, net, of $85,278 in 2015 and decrease to fixed
assets of $9,000 in 2014; and
o
Increase to cost of
goods sold by $55,936 and a decrease of $51,182,
respectively.
A
summary of the effect of the restatements for December 31,
2015 is as follows:
|
|
As
Previously Reported(1)
|
Restatement
Adjustments
|
As
Restated
|
|
Balance
Sheet – December 31, 2015
|
|
|
|
|
Inventory
|
$ -
|
$ 99,004
|
$ 99,004
|
|
Total
current assets
|
$ 27,614
|
$ 99,004
|
$ 126,618
|
|
Fixed
assets, net
|
$ 274,826
|
$ 85,278
|
$ 360,104
|
|
Total
assets
|
$ 302,440
|
$ 184,282
|
$ 486,722
|
|
|
|
|
|
|
Accounts
payable and accrued liabilities
|
$ 85,687
|
$ 150,734
|
$ 236,421
|
|
Total
liabilities
|
$ 85,687
|
$ 150,734
|
$ 236,421
|
|
Stock
payable
|
$ 123,565
|
$ (78,565)
|
$ 45,000
|
|
Accumulated
deficit
|
$ (9,855,216)
|
$ 112,112
|
$ (9,743,104)
|
|
Total
stockholders’ equity
|
$ 216,753
|
$ 33,547
|
$ 250,300
|
|
|
|
|
|
|
Statement
of Operations - For the Year Ended December 31,
2015
|
|
|
|
|
Cost
of goods sold
|
$ 610,297
|
$ 55,936
|
$ 666,233
|
|
General
and administrative
|
$ 375,885
|
$ 847,004
|
$ 1,222,888
|
|
Depreciation
and amortization
|
$ 62,655
|
$ 9,570
|
$ 72,225
|
|
Net
loss
|
$ (2,147,634)
|
$ (131,570)
|
$ (2,279,204)
|
|
|
|
|
|
|
Statement
of Cash Flows - For the Year Ended December 31,
2015
|
|
|
|
|
Net
loss
|
$ (2,147,634)
|
$ (131,570)
|
$ (2,279,204)
|
|
Increase/(Decrease)
in inventory
|
$ -
|
$ 139,984
|
$ 139,984
|
|
Decrease/(Increase)
in accounts payable and accrued liabilities
|
$ 52,956
|
$ 149,632
|
$ 202,588
|
|
Purchases
of fixed assets
|
$ (29,963)
|
$ (103,848)
|
$ (133,811)
|
(1)
Certain reclassifications have been made to the 2015 financial
statement amounts and disclosures. A portion of wages and related
expenses for the years ended December 31, 2015 and 2014 have been
reclassified in the financial statements from general and
administrative to research and development expenses.
A
summary of the effect of the restatements for December 31, 2014 is
as follows:
|
|
As
Previously Reported
|
Restatement
Adjustments
|
As
Restated
|
|
Balance
Sheet – December 31, 2014
|
|
|
|
|
Inventory
|
$ -
|
$ 238,988
|
$ 238,988
|
|
Total
current assets
|
$ 165,428
|
$ 238,988
|
$ 404,416
|
|
Fixed
assets, net
|
$ 307,518
|
$ (9,000)
|
$ 298,518
|
|
Total
assets
|
$ 472,946
|
$ 229,988
|
$ 702,934
|
|
|
|
|
|
|
Accounts
payable and accrued liabilities
|
$ 111,296
|
$ (13,695)
|
$ 97,601
|
|
Total
liabilities
|
$ 111,296
|
$ (13,695)
|
$ 97,601
|
|
Stock
payable
|
$ 9,000
|
$ -
|
$ 9,000
|
|
Accumulated
deficit
|
$ (7,707,582)
|
$ 243,683
|
$ (7,463,899)
|
|
Total
stockholders' equity
|
$ 361,650
|
$ 243,683
|
$ 605,333
|
|
|
|
|
|
|
Statement
of Operations - For the Year Ended December 31,
2014
|
|
|
|
|
Cost
of goods sold
|
$ 310,327
|
$ (51,182)
|
$ 259,145
|
|
Depreciation
and amortization
|
$ 37,242
|
$ -
|
$ 37,242
|
|
Net
loss
|
$ (2,232,060)
|
$ 51,182
|
$ (2,180,878)
|
|
|
|
|
|
|
Statement
of Cash Flows - For the Year Ended December 31,
2014
|
|
|
|
|
Net
loss
|
$ (2,232,060)
|
$ 51,182
|
$ (2,180,878)
|
|
Increase/(Decrease)
in inventory
|
$ -
|
$ (60,212)
|
$ (60,212)
|
|
Purchases
of fixed assets
|
$ (284,371)
|
$9,000
|
$ (275,371)
|
Refer to Note
4, Note 5 and Note 9, which were updated related to the restatement
described above.
F-11
Note
4 – Fixed Assets
As of
December 31, 2015 and 2014, fixed assets consisted of the
following:
|
|
December
31,
2015
|
December
31,
2014
|
|
Computer equipment and fixtures
|
$ 568,198
|
$ 487,187
|
|
Accumulated depreciation
|
(208,094)
|
(188,669)
|
|
Fixed assets,
net
|
$ 360,104
|
$ 298,518
|
Depreciation and
amortization expense for the years ended December 31, 2015 and 2014
was $72,225 and $37,242, respectively.
Note
5 – Current Liabilities
As of
December 31, 2015 and December 31, 2014, current liabilities
consisted of the following:
|
|
December
31,
2015
|
December
31,
2014
|
|
Accounts
payable
|
$ 80,945
|
$ 11,500
|
|
Accrued
payroll
|
144,629
|
63,768
|
|
Accrued employee
benefits
|
10,847
|
22,333
|
|
Total
|
$ 236,421
|
$ 97,601
|
During
the period ended December 31, 2015 the Company issued 26,515 shares
of common stock as payment for accrued and unpaid wages to two
officers of the Company.
During
the year ended December 31, 2015, the Company issued 10% promissory
notes in the aggregate principal amount of $32,000. As of December 31, 2015, the notes and
accrued interest of $3,200 were paid in full.
Note
6 – Capital Stock
At
December 31, 2015, the authorized capital of the Company consists
of 60,000,000 shares of
capital stock, consisting of 50,000,000 shares of common stock with
a par value of $0.0001 per share, and 10,000,000 shares
of preferred stock with a par value of $0.0001 per share.
On
December 8, 2014, the Company’s board of directors approved
the Second Amended and Restated 2013 Stock Incentive Plan, and the
Company reserved 1,318,182 shares of Common Stock
thereunder.
Effective January
22, 2015 ENDRA’s board of directors approved a 1 for 22
reverse stock split, which has been retroactively
stated in the accompanying financial statements As well the
board of directors authorized the elimination of the authorized but
unissued Series A Preferred Stock.
During
the year ended December 31, 2014, we issued 951,867 shares of
common stock, including 611,900 shares of common stock issued in
exchange for $1,751,015, 68,182 shares of common stock issued to
placement agent, and 271,785 shares issued for note conversions
valued at $777,264. There was $9,000 stock to be issued for
services during the year ended December 31, 2014.
During
the year ended December 31, 2015, we issued 525,975 shares of
common stock, including 87,415 shares of common stock issued in
exchange for $250,000, 26,515 shares of common stock issued for
accrued salaries of $63,768, and 412,045 shares issued for warrants
valued at $589,224. There was $114,565 stock to be issued during
the year ended December 31, 2014, including $36,000 for services
and $78,565 for accrued salaried to related parties.
F-12
As of
December 31, 2015 and 2014, there were 2,528,311 and 2,002,336
shares of Common Stock issued and outstanding, and no Preferred
Stock outstanding for either period.
Note
7 – Stock Options and Warrants
As of
December 31, 2015, the Company granted options to purchase 499,012
shares of common stock with exercise of $2.86 per share to
employees of the Company. The stock options vest between one and
three years. The fair value of these options was determined to be
$977,312 using the Black-Scholes-Merton option-pricing model based
on the following assumptions: (i) volatility rate of 90%, (ii)
discount rate of 0%, (iii) zero expected dividend yield, and (iv)
expected life of 5 years.
Stock-based
compensation expense related to vested options was $273,837 and
$250,493 during the years ended December 31, 2015 and 2014,
respectively. The Company determined the value of share-based
compensation using the Black-Scholes fair value option-pricing
model. A summary of option activity under the Company option plans
as of December 31, 2015, and changes during the period then ended
is presented below:
|
|
Number of
Options
|
Weighted Average
Exercise Price
|
Weighted Average
Remaining Contractual Term
|
|
Balance outstanding
at December 31, 2014
|
301,165
|
$2.86
|
3.62
|
|
Granted
|
197,847
|
2.84
|
4.51
|
|
Exercised
|
-
|
-
|
-
|
|
Forfeited
|
-
|
-
|
-
|
|
Expired
|
-
|
-
|
-
|
|
Balance outstanding
at December 31, 2015
|
499,012
|
$2.85
|
3.36
|
|
Exercisable at
December 31, 2015
|
413,907
|
$2.85
|
3.13
|
As of
December 31, 2015, the Company granted warrants to purchase
2,307,155 shares of common stock with exercise prices ranging from
$1.43 to $6.60 per share to employees of the Company and
consultants. The warrants generally vest immediately. The fair
value of these warrants was determined to be $686,343 using the
Black-Scholes-Merton option-pricing model based on the following
assumptions: (i) volatility rate of 90%, (ii) discount rate of 0%,
(iii) zero expected dividend yield, and (iv) expected life of 5
years. 480,227 warrants valued at $686,343 were issued during
warrant exchange program for exercised warrants. The Company
incurred a loss on warrant exercise of $25,000 during the year
ended December 31, 2015.
The
following table summarizes all stock warrant activity for the year
ended December 31, 2015:
|
|
Number of
Warrants
|
Weighted Average
Exercise Price
|
Weighted Average
Remaining Contractual Term
|
|
Balance outstanding
at December 31, 2014
|
879,805
|
$1.82
|
1.52
|
|
Granted
|
502,082
|
5.21
|
3.81
|
|
Exercised
|
(412,046)
|
1.49
|
0.05
|
|
Forfeited
|
-
|
-
|
-
|
|
Expired
|
(135,932)
|
1.43
|
-
|
|
Balance outstanding
at December 31, 2015
|
833,909
|
$4.08
|
2.87
|
|
Exercisable at
December 31, 2015
|
599,527
|
$4.08
|
2.87
|
F-13
Between July 31, 2013 and April 11, 2014, the
Company issued (i) warrants with an exercise price of $1.43
covering a total of 764,877 (adjusted for the
Company’s 2015 reverse stock split) shares of common stock and (ii) warrants with an
exercise price of $5.72 covering a total of 87,421 (adjusted for the Company’s 2015 reverse
stock split) shares of common stock. In 2015 in order to bring in
funds for working capital purposes, the Company issued 412,045
warrants under an exchange program to holders who exercised their
warrants. The warrants issued under the exchange program have an
exercise price of $5.72 (subject to a onetime downward adjustment
with a floor of $2.86 in the event the Company consummates an
equity financing resulting in gross proceeds of at least $1 million
with a per share valuation below $5.72), a term of five years, and
allow for cashless exercise after the Company has completed an
initial public offering. Stock-based compensation expense related
to exchange warrants was $686,343 during the year ended December
31, 2015 for the new warrants issued under this exchange program.
The Company determined the value of share-based compensation using
the Black-Scholes fair value option-pricing
model.
Note
8 – Material Contracts
On
September 30, 2015 the Company received a fully executed Purchase
Order from the University of Illinois at Urbana-Champaign for the
purchase of a Nexus 128 CT Scanner. The purchase price was $275,000
and was delivered during 2015.
During
September 2015 the Company received a fully executed Purchase Order
from a non-US based distributor for the purchase of a Nexus 128 CT
Scanner. The purchase price is $255,000 and was delivered during
2015.
Note
9
– Income Taxes
Deferred income
taxes reflect the net tax effects of temporary differences between
the carrying amounts of assets and liabilities for financial
reporting purposes and the amounts used for income tax purposes.
Significant components of the Company’s deferred tax assets
as of December 31, 2015 and 2014 are summarized below.
|
|
2014
|
2015
|
|
Net operating loss
carryforward
|
$ (2,542,038)
|
$ (3,294,333)
|
|
Stock based
compensation
|
217,321
|
241,857
|
|
Fair value of
options
|
85,168
|
93,105
|
|
Total deferred tax
assets
|
(2,239,550)
|
(2,959,372)
|
|
Valuation
allowance
|
(2,239,550)
|
(2,959,372)
|
|
Net deferred tax
asset
|
-
|
-
|
In
assessing the potential realization of deferred tax assets,
management considers whether it is more likely than not that some
portion or all of the deferred tax assets will be realized. The
ultimate realization of deferred tax assets is dependent upon the
Company attaining future taxable income during the periods in which
those temporary differences become deductible. As of December 31,
2015 and 2014, management was unable to determine if it is more
likely than not that the Company’s deferred tax assets will
be realized, and has therefore recorded an appropriate valuation
allowance against deferred tax assets at such dates.
No
federal tax provision has been provided for the years ended
December 31, 2015 and 2014 due to the losses incurred during such
periods. Reconciled below is the difference between the income tax
rate computed by applying the U.S. federal statutory rate and the
effective tax rate for the years ended December 31, 2015 and
2014.
|
|
2014
|
2015
|
|
U.S.
federal statutory income tax
|
-34.00%
|
-34.00%
|
|
State tax, net of
federal tax benefit
|
-5.80%
|
-5.80%
|
|
Stock based
compensation
|
0.00%
|
0.00%
|
|
Change in valuation
allowance
|
39.80%
|
39.80%
|
|
Effective tax
rate
|
0.00%
|
0.00%
|
At
December 31, 2015 and 2014, the Company has available
net operating loss carryforwards for federal and state income tax
purposes of approximately $9.7 million and
$7.5 million, respectively, which, if not utilized
earlier, expire through 2035.
Note
10 – Commitments and Contingencies
From
time to time the Company may become a party to litigation in the
normal course of business. Management believes that there are no
current legal matters that would have a material effect on the
Company’s financial position or results of
operations.
F-14
On
November 11, 2007, the Company entered into an at-will employment
agreement with its Chief Operating Officer. The employment
agreement requires annual base salary payments of $200,000 per
year, with a bonus potential of 20% of the then current base
salary. In addition, the executive has been granted an option to
purchase 103,000 shares of Company's common stock exercisable at
$2.86 per share, vesting in 3 equal annual installments on each
anniversary of its three year term. The agreement also provides for
severance compensation if terminated other than for cause (as
defined) of 6 months of the then applicable base salary if the CEO
has been employed at least 6 months, and compensation equal to 12
months of the then applicable base salary if employed over 12
months.
On
August 28, 2014, the Company entered into a services agreement with
StoryCorp Consulting dba Wells Compliance Group for financial
reporting and compliance services. David R. Wells is the owner of
this firm and is the Company’s Interim Chief Financial
Officer. The services agreement calls for payments of $5,000, and
accrues an additional $3,000 per month in fees to be paid by common
stock at the time of a public offering. The accrued balance due
under the cash portion as of December 31, 2015 and December 31,
2014 was $7,500 and $0, respectively, and the accrued balance due
under the stock portion was $45,000 and $9,000, respectively. The
Company can cancel the contract at any time without
notice.
Effective January
1, 2015, we entered into office lease agreement with Green Court,
LLC, a Michigan limited liability company, for approximately 3,657
rentable square feet of space, for the initial monthly rent of
$5,986, which commenced on January 1, 2015 for an initial term of
60 months. Under the terms of the lease the Company has an option
on the same space for an additional 60 month term. Future minimum
payments under this lease are as follows:
|
2016
|
$73,476
|
|
2017
|
75,302
|
|
2018
|
77,190
|
|
2019
|
79,125
|
|
2020
|
81,460
|
|
Total
|
$386,553
|
On
April 16, 2015, the Company entered into an at-will employment
agreement with its Chief Executive Officer. The employment
agreement requires annual base salary payments of $250,000 per year
with a bonus potential of 50% of the then current base salary. In
addition, the executive has been granted an option to purchase
124,248 shares of Company's common stock at exercisable at $2.86
per share, vesting in 3 equal annual installments on each
anniversary of its three year term. The agreement also provides for
severance compensation if terminated other than for cause (as
defined) of 6 months of the then applicable base salary if the CEO
has been employed at least 6 months, and compensation equal to 12
months of the then applicable base salary if employed over 12
months.
Note
11 – Subsequent Events
On
January 29, 2016 the Company received a fully executed Purchase
Order from United Well (ENDRA's Chinese Distributor) for the
purchase of two (2) Nexus 128 CT Scanners. The purchase price is
$500,000 and the systems are scheduled for delivery during the
second half of 2016. In accordance with the Company’s revenue
recognition policy, it has determined that persuasive evidence of
an arrangement exists, the price is fixed or determinable per the
customer’s contract and collectability is reasonably assured.
Accordingly, the Company expects to recognize the revenue from this
sale upon shipment of the completed units to its customer which is
when title to the unit passes.
Subsequent
to December 31, 2015, the Company issued 3,497 shares of common
stock for warrants valued at $5,000. The Company also agreed to
issue $69,910 shares of its common stock, including $9,000 for
services and $60,910 for accrued salaries to related
parties.
F-15
On April 22, 2016 the Company entered into
convertible promissory notes with approximately 60 investors for a
total amount of $1,199,448 (“April 2016 Notes”).
The April 2016 Notes mature one year from the issue date,
accrue interest at the rate of 8% per annum and are payable at
maturity, are secured by all assets of the Company, now owned or
hereafter acquired, and automatically convert into the same
security issued by the Company, on the same terms, including price,
upon a qualified financing in the amount of $3.0 million or more.
Separately, upon any individual noteholder’s election, such
noteholder’s convertible promissory note is convertible into
shares of the Company’s common stock, and upon the election
of noteholders holding a majority of the outstanding principal
amount of the convertible promissory notes, all outstanding April
2016 Notes are convertible into shares of the Company’s
common stock, in each case at a conversion price of $0.40 per
share, or 2,998,624 shares of the Company’s common stock.
While we believe this offering will constitute a qualified
financing as described above, because the conversion price is
significantly lower than the expected public offering price of the
common stock sold in this offering, we have assumed that the
noteholders will convert all of the outstanding principal and
accrued interest on the convertible promissory notes into shares of
common stock of the Company at the lower conversion price
immediately prior to the completion of the
offering.
The
Company has filed a registration statement on Form S-1 with
the U.S. Securities and Exchange Commission in connection with a
proposed offering of its securities. On December 3, 2016, the board
of directors of the Company and stockholders holding a majority of
the outstanding shares of the Company’s common stock approved
resolutions authorizing the board of directors to effect a reverse
split of the Company’s common stock at certain exchange
ratios ranging from 1:1.50 to
1:2.50, with the board of directors
retaining the discretion as to whether to implement the reverse
stock split and which exchange ratio to implement. Following the
effectiveness of the registration statement of which this
prospectus is a part, and prior to the closing of this offering,
the Company anticipates that it will effect the reverse stock split
at a ratio of 1 share for each 2.50 shares.
Since
the 1 for 2.50 reverse stock split is to be effected
after the effectiveness of the registration statement, the
historical share information included in the accompanying financial
statements and notes hereto do not assume the 1 for
2.50 reverse stock split, and accordingly have not been
adjusted.
On a
pro forma basis, taking into account the anticipated reverse stock
split at a ratio of 1 share for each 2.50 shares outstanding as of
the following dates of presentation, the Company’s earnings
per share would change as follows:
As
Presented:
|
|
Year Ended
|
Year Ended
|
|
|
December 31,
|
December 31,
|
|
|
2015
|
2014
|
|
Net
Loss
|
$ (2,279,204)
|
$ (2,180,878)
|
|
Basic
and diluted net loss per common share
|
$ (0.98)
|
$ (1.35)
|
|
Weighted
average shares outstanding, basic and diluted
|
2,320,045
|
1,612,569
|
Pro
forma:
|
|
Year Ended
|
Year Ended
|
|
|
December 31,
|
December 31,
|
|
|
2015
|
2014
|
|
Net
Loss
|
$(2,279,204)
|
$(2,180,878)
|
|
Basic
and diluted net loss per common share
|
$(2.46)
|
$(3.38)
|
|
Weighted
average shares outstanding, basic and diluted
|
928,018
|
645,028
|
F-16
ENDRA
LIFE SCIENCES
INC.
FINANCIAL
STATEMENTS
Table
of Contents
|
F-18
|
|
|
Condensed
Statements of
Operations for the Three and Nine Months Ended
September 30, 2016 and 2015 (unaudited)
|
F-19
|
|
Condensed
Statements of Cash Flows for the
Nine Months Ended September 30, 2016 and 2015
(unaudited)
|
F-20
|
|
Notes
to the unaudited condensed financial statements
|
F-21
|
F-17
ENDRA LIFE SCIENCES INC.
CONDENSED BALANCE SHEETS
CONDENSED BALANCE SHEETS
|
|
September
30,
|
December
31,
|
|
Assets
|
2016
|
2015
|
|
Assets
|
(unaudited)
(Restated)
|
(Restated)
|
|
Cash
|
$ 75,543
|
$ 19,128
|
|
Inventory
|
276,856
|
99,004
|
|
Other current
asset
|
9,797
|
8,486
|
|
Total Current
Assets
|
362,196
|
126,618
|
|
Other
Assets
|
|
|
|
Fixed assets,
net
|
311,492
|
360,104
|
|
Total
Assets
|
$ 673,688
|
$ 486,722
|
|
|
|
|
|
Liabilities
and Stockholders’ (Deficit) Equity
|
|
|
|
Current
Liabilities
|
|
|
|
Accounts payable
and accrued liabilities
|
$ 303,071
|
$ 236,421
|
|
Notes
payable
|
50,000
|
-
|
|
Convertible notes
payable, net of discount
|
561,812
|
-
|
|
Total Current
Liabilities
|
914,883
|
236,421
|
|
Total
Liabilities
|
914,883
|
236,421
|
|
|
|
|
|
Stockholders’
(Deficit) Equity
|
|
|
|
Preferred stock,
$0.0001 par value; 10,000,000 shares authorized; no shares issued
or outstanding
|
-
|
-
|
|
Common stock,
$0.0001 par value; 50,000,000 shares authorized; 2,531,808 and
2,528,311 shares issued and outstanding
|
253
|
253
|
|
Stock
payable
|
72,000
|
45,000
|
|
Additional paid in
capital
|
11,518,145
|
9,948,151
|
|
Accumulated
deficit
|
(11,831,594)
|
(9,746,103)
|
|
Total
Stockholders’ (Deficit) Equity
|
(241,196)
|
250,301
|
|
Total
Liabilities and Stockholders’ (Deficit)
Equity
|
$ 673,688
|
$ 486,722
|
The
accompanying notes are an integral part of these unaudited
condensed financial statements.
F-18
ENDRA
LIFE SCIENCES INC.
CONDENSED
STATEMENTS OF OPERATIONS
(Unaudited)
|
|
Three Months Ended September 30,
2016
(Restated)
|
Three Months Ended September 30,
2015
(Restated)
|
Nine Months Ended September 30,
2016
(Restated)
|
Nine Months Ended September 30, 2015
(Restated)
|
|
Revenue
|
$ -
|
$ 326,085
|
$ -
|
$ 1,155,065
|
|
|
|
|
|
|
|
Cost
of Goods Sold
|
-
|
189,535
|
-
|
539,809
|
|
|
|
|
|
|
|
Gross Profit
|
$ -
|
$ 136,550
|
$ -
|
$ 615,256
|
|
|
|
|
|
|
|
Operating Expenses
|
|
|
|
|
|
Research
and development
|
139,540
|
265,025
|
338,417
|
768,215
|
|
Sales
and marketing
|
16,040
|
20,743
|
26,197
|
41,422
|
|
General
and administrative
|
451,531
|
395,761
|
1,110,264
|
918,774
|
|
Total
operating expenses
|
607,111
|
681,528
|
1,474,878
|
1,728,411
|
|
|
|
|
|
|
|
Operating
loss
|
(607,111)
|
(544,979)
|
(1,474,878)
|
(1,113,155)
|
|
|
|
|
|
|
|
Other Expenses
|
|
|
|
|
|
Loss
on warrant exercise
|
-
|
-
|
(5,823)
|
-
|
|
Other
income (expense)
|
(372,789)
|
3,991
|
(607,789)
|
(707,316)
|
|
Total
other expenses
|
(372,789)
|
3,991
|
(613,612)
|
(707,316)
|
|
|
|
|
|
|
|
Loss
from operations before taxes
|
(979,899)
|
(540,987)
|
(2,088,491)
|
(1,820,471)
|
|
|
|
|
|
|
|
Provision
for income taxes
|
-
|
-
|
-
|
-
|
|
|
|
|
|
|
|
Net Loss
|
$ (979,899)
|
$ (540,987)
|
$ (2,088,491)
|
$ (1,820,471)
|
|
|
|
|
|
|
|
Net loss per share – basic and diluted
|
$ (0.39)
|
$ (0.22)
|
$ (0.82)
|
$ (0.81)
|
|
|
|
|
|
|
|
Weighted average common shares – basic and
diluted
|
2,531,808
|
2,458,090
|
2,531,566
|
2,250,011
|
The
accompanying notes are an integral part of these unaudited
condensed financial statements.
F-19
ENDRA
LIFE SCIENCES INC.
CONDENSED
STATEMENTS OF CASH FLOWS
(Unaudited)
|
|
Nine Months Ended
|
|
|
|
September 30,
|
September 30,
|
|
|
2016
|
2015
|
|
Cash
Flows from Operating Activities
|
(Restated)
|
(Restated)
|
|
Net
loss
|
$ (2,088,491)
|
$ (1,820,471)
|
|
Adjustments to
reconcile net loss to net cash used in operating
activities:
|
|
|
|
Depreciation and
amortization
|
48,613
|
55,841
|
|
Common stock and
options issued for services
|
199,723
|
25,444
|
|
Additional warrants
issued during exchange
|
5,823
|
686,343
|
|
Interest on
discount of convertible debt
|
561,812
|
-
|
|
Loss on warrant
exercise
|
-
|
25,000
|
|
Changes in
operating assets and liabilities:
|
|
|
|
Increase in
accounts receivable
|
-
|
(125,000)
|
|
Increase in
inventory
|
(177,852)
|
169,259
|
|
Increase in other
asset
|
(1,311)
|
500
|
|
Increase in
accounts payable and accrued liabilities
|
66,650
|
107,721
|
|
Net cash used in
operating activities
|
(1,385,033)
|
(875,363)
|
|
|
|
|
|
Cash
Flows from Investing Activities
|
|
|
|
Purchases of fixed
assets
|
-
|
(133,811)
|
|
Net cash used in
investing activities
|
-
|
(133,811)
|
|
|
|
|
|
Cash
Flows from Financing Activities
|
|
|
|
Proceeds from
issuance of common stock
|
5,000
|
955,617
|
|
Proceeds from notes
payable
|
50,000
|
-
|
|
Proceeds from
convertible notes
|
1,386,448
|
-
|
|
Net cash provided
by financing activities
|
1,441,448
|
955,617
|
|
|
|
|
|
Net
Increase/(Decrease) in cash
|
56,415
|
(53,557)
|
|
|
|
|
|
Cash, beginning of
period
|
19,128
|
156,442
|
|
|
|
|
|
Cash,
end of period
|
$75,543
|
$102,885
|
|
|
|
|
|
Supplemental
disclosures:
|
|
|
|
Interest
paid
|
$-
|
$-
|
|
Income
tax paid
|
$-
|
$-
|
|
Discount
on convertible notes
|
$1,386,448
|
$-
|
The
accompanying notes are an integral part of these unaudited
condensed financial statements.
F-20
ENDRA LIFE SCIENCES INC.
NOTES TO CONDENSED UNAUDITED FINANCIAL STATEMENTS
For the nine months ended September 30, 2016 and 2015
Note 1 – Nature of the Business
ENDRA
Life Sciences Inc. (“ENDRA” or the
“Company”) was incorporated on July 18, 2007 as a
Delaware corporation.
ENDRA
has developed a medical imaging technology based on the
thermoacoustic effect that significantly improves the sensitivity
and specificity of clinical ultrasound.
Note 2 – Summary of Significant Accounting
Policies
The
significant accounting policies applied in the annual financial
statements of the Company as of December 31, 2015 are applied
consistently in these financial statements.
Basis
of Presentation
The
accompanying condensed financial statements are unaudited. The
unaudited interim financial statements have been prepared in
accordance with accounting principles generally accepted in the
United States ("GAAP") and pursuant to the rules and regulations of
the Securities and Exchange Commission (the "SEC"). Certain
information and note disclosures normally included in annual
financial statements prepared in accordance with GAAP have been
condensed or omitted pursuant to those rules and regulations,
although the Company believes that the disclosures made are
adequate to make the information not misleading
In the
opinion of management, all adjustments (consisting of normal
recurring accruals) considered necessary for a fair presentation
have been included. Operating results for the nine month period
ended September 30, 2016 are not necessarily indicative of the
results that may be expected for the year ended December 31,
2016.
The
condensed balance sheet as of December 31, 2015 has been derived
from the audited financial statements at that date but do not
include all disclosures required by the accounting principles
generally accepted in the United States of America.
Use of Estimates
The
preparation of the financial statements in conformity with
accounting principles generally accepted in the United States
requires management to make estimates and assumptions that affect
the reported amounts of assets and liabilities, and disclosure of
contingent liabilities at the date of the financial statements and
the reported amounts of expenses during the reporting period.
Actual results could differ from those estimates.
Management
makes estimates that affect certain accounts including deferred
income tax assets, accrued expenses, fair value of equity
instruments and reserves for any other commitments or
contingencies. Any adjustments applied to estimates are recognized
in the period in which such adjustments are
determined.
Cash and Cash Equivalents
The
Company considers all cash on hand and in banks, including accounts
in book overdraft positions, certificates of deposit and other
highly-liquid investments with maturities of three
months or less, when purchased, to be cash and cash equivalents. As
of September 30, 2016 and
December 31, 2015 the Company had no cash
equivalents.
Capitalization of Fixed Assets
F-21
The
Company capitalizes expenditures related to property and equipment,
subject to a minimum rule, that have a useful life greater than one
year for: (1) assets purchased; (2) existing assets that are
replaced, improved or the useful lives have been extended; or (3)
all land, regardless of cost. Acquisitions of new assets,
additions, replacements and improvements (other than land) costing
less than the minimum rule in addition to maintenance and repair
costs, including any planned major maintenance activities, are
expensed as incurred.
Capitalization of Intangible Assets
The
Company records the purchase of intangible assets not purchased in
a business combination in accordance with the ASC Topic
350.
Inventory
The
Company’s inventory is stated at the lower of cost or
market, with cost being determined on the
first-in, first-out (“FIFO”) method. As
of September 30, 2016 and December 31, 2015 the Company had
inventory of $276,856 and $99,004,
respectively.
Revenue Recognition
The Company recognizes revenue in accordance with the requirements
of ASC 605-10-599, which directs that it should recognize revenue
when (1) persuasive evidence of an arrangement exists (contracts);
(2) delivery has occurred; (3) the seller’s price is fixed or
determinable (per the customer’s contract); and (4)
collectability is reasonably assured (based upon our credit
policy). For products sold to end users revenue is recognized when
title has passed to the customer and collectability is reasonably
assured; and no further efforts are required. Future revenue from
anticipated new products will follow this same policy.
Advertising Expense
The
cost of advertising is expensed as
incurred. Advertising expense for the nine months ended
September 30, 2016 were approximately
$3,641. Advertising expense for the three and nine months
ended September 30, 2015 were approximately $690 and $3,641,
respectively.
Income Taxes
The
Company utilizes ASC 740, “Income Taxes,” which
requires the recognition of deferred tax assets and liabilities for
the expected future tax consequences of events that have been
included in the financial statements or tax returns. Under this
method, deferred tax assets and liabilities are determined based on
the difference between the tax basis of assets and liabilities and
their financial reporting amounts based on enacted tax laws and
statutory tax rates applicable to the periods in which the
differences are expected to affect taxable income. A valuation
allowance is recorded when it is “more likely-than-not”
that a deferred tax asset will not be realized.
The
Company generated a deferred tax asset through net operating loss
carry-forwards. However, a valuation allowance of 100% has been
established due to the uncertainty of the Company’s
realization of the net operating loss carry forward prior to its
expiration.
Research and Development Costs
The
Company follows ASC 730-10, “Research and Development”.
Research and development costs are charged to the statement of
operations as incurred. During the nine months ended September 30, 2016 and 2015
the Company incurred $338,417 and $768,215 of expenses related to
research and development costs, respectively. During the three
months ended September 30, 2016 and 2015 the Company incurred
$139,540 and $265,025 of expenses related to research and
development costs, respectively.
Net Earnings (Loss) Per Common Share
The
Company computes earnings per share under ASC Subtopic 260-10,
Earnings Per Share (“ASC 260-10”). Basic earnings
(loss) per share is computed by dividing the net income (loss)
attributable to the common stockholders (the numerator) by the
weighted average number of shares of common stock outstanding (the
denominator) during the reporting periods. Diluted loss per
share is computed by increasing the denominator by the weighted
average number of additional shares that could have been
outstanding from securities convertible into common stock (using
the “treasury stock” method), unless their effect on
net loss per share is anti-dilutive. There were 4,642,651 and 1,332,921 potentially
dilutive shares, which include outstanding common stock options,
warrants, and convertible notes, as of September 30, 2016 and December 31,
2015.
F-22
The
potential shares, which are excluded from the determination of
basic and diluted net loss per share as their effect is
anti-dilutive, are as follows:
|
|
September
30,
2016
|
December
31,
2015
|
|
Options to purchase
common stock
|
531,584
|
499,012
|
|
Warrants to
purchase common stock
|
534,842
|
833,909
|
|
Convertible
notes
|
3,576,225
|
-
|
|
Potential
equivalent shares excluded
|
4,642,651
|
1,332,921
|
Fair Value Measurements
Disclosures
about fair value of financial instruments require disclosure of the
fair value information, whether or not recognized in our balance
sheet, where it is practicable to estimate that value. As of
September 30, 2016 and December 31, 2015, the amounts reported for
cash, accrued liabilities and accrued interest approximated fair
value because of their short maturities.
In accordance with ASC Topic 820, “Fair Value Measurements
and Disclosures,” we measure certain financial instruments at
fair value on a recurring basis. ASC Topic 820 defines fair value,
established a framework for measuring fair value in accordance with
accounting principles generally accepted in the United States, and
expands disclosures about fair value measurements.
Fair value is defined as the price that would be received to sell
an asset or paid to transfer a liability in an orderly transaction
between market participants at the measurement date. ASC Topic 820
established a three-tier fair value hierarchy, which prioritizes
the inputs used in measuring fair value. The hierarchy gives the
highest priority to unadjusted quoted prices in active markets for
identical assets or liabilities (level 1 measurements) and the
lowest priority to unobservable inputs (level 3 measurements).
These tiers include:
|
|
●
|
Level
1, defined as observable inputs such as quoted prices for identical
instruments in active markets;
|
|
|
|
|
|
|
●
|
Level
2, defined as inputs other than quoted prices in active markets
that are either directly or indirectly observable such as quoted
prices for similar instruments in active markets or quoted prices
for identical or similar instruments in markets that are not
active; and
|
|
|
|
|
|
|
●
|
Level
3, defined as unobservable inputs in which little or no market data
exists, therefore requiring an entity to develop its own
assumptions, such as valuations derived from valuation techniques
in which one or more significant inputs or significant value
drivers are unobservable.
|
Share-based Compensation
The
Company’s Second Amended and Restated 2013 Stock Incentive
Plan (the “Plan”), which is approved by its board of
directors, permits the grant of share options and shares to its
employees and consultants for up to 1,318,182 shares of common
stock. The Company records share-based compensation in accordance
with the provisions of the Share-based Compensation Topic of the
FASB Codification. The guidance requires the use of option-pricing
models that require the input of highly subjective assumptions,
including the option’s expected life and the price volatility
of the underlying stock. The fair value of each option grant is
estimated on the date of grant using the Black-Scholes option
valuation model, and the resulting charge is expensed using the
straight-line attribution method over the vesting period. The
Company has elected to use the calculated value method to account
for the options it issued in 2016 and 2015. A nonpublic entity that
is unable to estimate the expected volatility of the price of its
underlying share may measure awards based on a “calculated
value,” which substitutes the volatility of appropriate
public companies (representative of the company’s size and
industry) as a bench mark for the volatility of the entity’s
own share price. Currently, there is no active market for the
company’s common shares. The Company has used the historical
closing values of these companies to estimate volatility, which was
calculated to be 90%.
F-23
Stock
compensation expense recognized during the period is based on the
value of share-based awards that were expected to vest during the
period adjusted for estimated forfeitures. The estimated fair value
of grants of stock options and warrants to non-employees of the
Company is charged to expense, if applicable, in the financial
statements. These options vest in the same manner as the employee
options granted under each of the option plans as described
above.
Going Concern
The
Company’s financial statements are prepared using U.S. GAAP
applicable to a going concern, which contemplates the realization
of assets and liquidation of liabilities in the normal course of
business. The Company has limited operating history and had a
cumulative net loss from inception to September 30, 2016 of
$11,999,600. The Company has a working capital deficit of $645,586
as of September 30, 2016. The Company has not yet established an
ongoing source of revenue sufficient to cover its operating costs
and to allow it to continue as a going concern. The accompanying
condensed financial statements for the period ended September 30,
2016, have been prepared assuming the Company will continue as a
going concern. The Company believes its cash resources are
insufficient to meet its anticipated needs during the next twelve
months. The Company will require additional financing to fund its
future planned operations, including research and development and
commercialization of its products.
The
ability of the Company to continue as a going concern is dependent
on the Company obtaining adequate capital to fund operating losses
until it establishes a revenue stream and becomes profitable.
Management’s plans to continue as a going concern include
raising additional capital through borrowing and sales of common
stock. However, management cannot provide any assurances that the
Company will be successful in accomplishing any of its plans. If
the Company is not able to obtain the necessary additional
financing on a timely basis, the Company will be forced to delay or
scale down some or all of its development activities or perhaps
even cease the operation of its business. The ability of the
Company to continue as a going concern is dependent upon its
ability to successfully secure other sources of financing and
attain profitable operations. The accompanying financial statements
do not include any adjustments that might be necessary if the
Company is unable to continue as a going concern.
Note
3 – Restatement
The
Company determined that a restatement of its originally issued
financial statements for the periods ended December 31, 2015 and
September 31, 2016 was required for the following
reason:
●
Due to a revision
of its inventory and cost of goods sold, the Company has restated
its Balance Sheets as of September 30, 2016 and December 31, 2015,
and statements of operations for the three and nine months ended
September 30, 2016 and 2015, and statements of cash flows for the
nine months ended September 30, 2016 and 2015 to account for the
following:
o
Increase of
inventory of $99,004 and $99,004,
respectively;
o
Increase to
fixed assets, net, of $85,278 in 2015 and $75,108 for the
nine months ended September 30, 2016;
and
o
Increase to cost of
goods sold by $73,736 for the
nine months ended September 30, 2015.
|
|
As
Previously Reported
|
Restatement
Adjustments
|
As
Restated
|
|
Balance
Sheet – December 31, 2015
|
|
|
|
|
Inventory
|
$ -
|
$ 99,004
|
$ 99,004
|
|
Total
current assets
|
$ 27,614
|
$ 99,004
|
$ 126,618
|
|
Fixed
assets, net
|
$ 274,826
|
$ 85,278
|
$ 360,104
|
|
Total
assets
|
$ 302,440
|
$ 184,282
|
$ 486,722
|
|
|
As Previously Reported
|
Restatement Adjustments
|
As Restated
|
|
Balance
Sheet – September 30, 2016
|
|
|
|
|
Inventory
|
$ 177,852
|
$ 99,004
|
$ 276,856
|
|
Total
current assets
|
$ 263,192
|
$ 99,004
|
$ 362,196
|
|
Fixed
assets, net
|
$ 236,384
|
$ 75,108
|
$ 311,492
|
|
Total
assets
|
$ 499,576
|
$ 174,112
|
$ 673,688
|
|
|
|
|
|
|
|
|
|
|
|
Statement
of Operations - For the Nine Months Ended September 30,
2016
|
|
|
|
|
Cost
of goods sold
|
$-
|
$-
|
$-
|
|
Depreciation
and amortization
|
$ 38,442
|
$ 10,171
|
$ 48,613
|
|
Net
loss
|
$ (2,078,320)
|
$ (10,171)
|
$ (2,088,491)
|
|
|
|
|
|
|
Statement
of Cash Flows - Nine Months Ended September 30,
2016
|
|
|
|
|
Net
loss
|
$ (2,078,320)
|
$ (10,171)
|
$ (2,088,491)
|
|
Increase/(Decrease)
in inventory
|
$ (177,852)
|
$-
|
$ (177,852)
|
|
Purchases
or fixed assets
|
$-
|
$-
|
$-
|
|
|
As Previously
Reported
|
Restatement
Adjustments
|
As
Restated
|
|
Statement
of Operations - For the Three Months Ended September 30,
2016
|
|
|
|
|
Cost
of goods sold
|
$-
|
$-
|
$-
|
|
Net
loss
|
$(976,484)
|
$(3,415)
|
$(979,899)
|
|
|
|
|
|
|
Statement
of Cash Flows - For the Three Months Ended September 30,
2016
|
|
|
|
|
Net
loss
|
$(976,484)
|
$(3,415)
|
$(979,899)
|
|
|
As Previously
Reported
|
Restatement
Adjustments
|
As
Restated
|
|
Statement of
Operations - For the Nine Months Ended September 30,
2015
|
|
|
|
|
Cost of goods
sold
|
$466,073
|
$73,736
|
$539,809
|
|
Depreciation
and amortization
|
$49,686
|
$6,155
|
$55,841
|
|
Net
loss
|
$(1,740,580)
|
$(79,891)
|
$(1,820,471)
|
|
|
|
|
|
|
Statement of
Cash Flows - For the Nine Months Ended September 30,
2015
|
|
|
|
|
Net
loss
|
$(1,740,580)
|
$(79,891)
|
$(1,820,471)
|
|
Depreciation
and amortization
|
$49,686
|
$6,155
|
$55,841
|
|
Increase/(Decrease) in inventory
|
$-
|
$169,259
|
$169,259
|
|
Purchases of
fixed assets
|
$(29,963)
|
$(103,848)
|
$(133,811)
|
|
|
As Previously
Reported
|
Restatement
Adjustments
|
As
Restated
|
|
Statement
of Operations - For the Three Months Ended September 30,
2015
|
|
|
|
|
Cost
of goods sold
|
$265,056
|
$(75,521)
|
$189,535
|
|
Net
loss
|
$(613,093)
|
$72,106
|
$(540,987)
|
|
|
|
|
|
|
Statement
of Cash Flows - For the Three Months Ended September 30,
2015
|
|
|
|
|
Net
loss
|
$(613,093)
|
$72,106
|
$(540,987)
|
Refer
to Note 4, which was updated related to the restatement described
above.
Note 4 – Fixed Assets
As of
September 30, 2016 and December 31, 2015, fixed assets consisted of
the following:
|
|
September
30, 2016
|
December 31,
2015
|
|
Computer equipment
and fixtures
|
$ 571,318
|
$ 571,318
|
|
Accumulated
depreciation
|
(259,827)
|
(211,214)
|
|
Fixed
assets, net
|
$ 311,492
|
$ 360,104
|
F-24
Depreciation
expense for the nine months ended September 30, 2016 and September
30, 2015 was $48,613 and $55,841,
respectively. Depreciation expense for the three months ended
September 30, 2016 and September 30, 2015 was $16,341
and $18,744, respectively.
Note
5 – Current Liabilities
|
|
September 30, 2016
|
December 31,
2015
|
|
Accounts
payable
|
$133,411
|
$80,945
|
|
Accrued
payroll
|
105,258
|
144,629
|
|
Accrued employee
benefits
|
20,361
|
10,847
|
|
Note
payable
|
50,000
|
-
|
|
Convertible notes,
net of discount
|
561,812
|
-
|
|
Total
|
$914,883
|
$236,421
|
On January 28, 2016 the Company
entered into promissory notes with three investors for a total
amount of $50,000. The notes mature one year from the issue
date, accrue no interest and are payable at
maturity,
On
April 22, 2016 the Company entered into convertible promissory
notes with approximately 60 investors for a total amount of
$1,386,448 (“April 2016 Notes”). The April 2016
Notes mature one year from the issue date, accrue interest at the
rate of 8% per annum and are payable at maturity, are secured by
all assets of the Company, now owned or hereafter acquired, and
automatically convert into the same security issued by the Company,
on the same terms, including price, upon a qualified financing in
the amount of $3.0 million or more. Separately, upon any individual
noteholder’s election, such noteholder’s convertible
promissory note is convertible into shares of the Company’s
common stock, and upon the election of noteholders holding a
majority of the outstanding principal amount of the convertible
promissory notes, all outstanding April 2016 Notes are convertible
into shares of the Company’s common stock, in each case at a
conversion price of $0.40 per share, or 3,576,225 shares of the
Company’s common stock. In connection with the issuance of
these notes, the Company recorded a debt discount at an initial
aggregate value of $1,386,448. $561,812 was amortized during the
nine months ended September 30, 2016, resulting in a debt discount
balance of $824,637 as of September 30, 2016.
Note
6 – Capital Stock
At
September 30, 2016, the authorized capital of the Company consists
of 60,000,000 shares of
capital stock, consisting of 50,000,000 shares of common stock with
a par value of $0.0001 per share, and 10,000,000 shares
of preferred stock with a par value of $0.0001 per share.
During
the nine months ended September 30, 2016, we issued 3,497 shares of
common stock for warrants exercised for $5,000. There was $27,000
stock to be issued during the nine months ended September 30, 2016
for services. There was $72,000 in stock
payable as of September 30, 2016.
As of
September 30, 2016, there were 2,531,808 shares of Common Stock
issued and outstanding, and no Preferred Stock outstanding for
either period.
Note
7 – Stock Options and Warrants
During
the nine months ended September 30, 2016, the Company granted
options to purchase 34,617 shares of common stock with an exercise
price of $2.86 per share to employees of the Company. The stock
options vest immediately. The fair value of these options was
determined to be $172,273 using the Black-Scholes-Merton
option-pricing model based on the following assumptions: (i)
volatility rate of 90%, (ii) discount rate of 0%, (iii) zero
expected dividend yield, and (iv) expected life of 5 years. A
summary of option activity under the Company option plans as of
September 30, 2016, and changes during the period then ended is
presented below:
F-25
|
|
Number of
Options
|
Weighted Average
Exercise Price
|
Weighted Average
Remaining Contractual Term
|
|
Balance outstanding
at December 31, 2015
|
499,012
|
$2.86
|
3.62
|
|
Granted
|
34,617
|
2.86
|
4.26
|
|
Exercised
|
-
|
-
|
-
|
|
Forfeited
|
-
|
-
|
-
|
|
Cancelled or
expired
|
(2,045)
|
2.86
|
-
|
|
Balance outstanding
at September 30, 2016
|
531,584
|
$2.86
|
2.72
|
|
Exercisable at
September 30, 2016
|
557,843
|
$2.86
|
2.52
|
During
the nine months ended September 30, 2016, the Company granted
warrants to purchase 3,497 shares of common stock with an exercise
price of $5.72 per share during the warrant exchange program for
exercised warrants. The warrants vest immediately. The fair value
of these warrants was determined to be $5,823 using the
Black-Scholes-Merton option-pricing model based on the following
assumptions: (i) volatility rate of 90%, (ii) discount rate of 0%,
(iii) zero expected dividend yield, and (iv) expected life of 5
years.
The
following table summarizes all stock warrant activity for the nine
months ended September 30, 2016:
|
|
Number of
Warrants
|
Weighted
Average
Exercise
Price
|
Weighted
Average
Remaining
Contractual
Term
|
|
Balance outstanding
at December 31, 2015
|
837,406
|
$4.08
|
2.87
|
|
Granted
|
3,497
|
5.72
|
4.31
|
|
Exercised
|
(3,497)
|
1.43
|
-
|
|
Forfeited
|
-
|
-
|
-
|
|
Expired
|
(302,564)
|
1.75
|
-
|
|
Balance outstanding
at September 30, 2016
|
534,842
|
$5.41
|
3.56
|
|
Exercisable at
September 30, 2016
|
534,842
|
$5.41
|
3.56
|
Note
8 – Commitments and Contingencies
From
time to time the Company may become a party to litigation in the
normal course of business. Management believes that there are no
current legal matters that would have a material effect on the
Company’s financial position or results of
operations.
On
November 11, 2007, the Company entered into an at-will employment
agreement with its Chief Operating Officer. The employment
agreement requires annual base salary payments of $200,000 per
year, with a bonus potential of 20% of the then current base
salary. In addition, the executive has
been granted an option to purchase 103,000 shares of Company's
common stock exercisable at $2.86 per share, vesting in 3 equal
annual installments on each anniversary of its three year term. The
agreement also provides for severance compensation if terminated
other than for cause (as defined) of 6 months of the then
applicable base salary if the CEO has been employed at least 6
months, and compensation equal to 12 months of the then applicable
base salary if employed over 12 months.
On
August 28, 2014, the Company entered into a services agreement with
StoryCorp Consulting dba Wells Compliance Group for financial
reporting and compliance services. David R. Wells is the owner of
this firm and is the Company’s Interim Chief Financial
Officer. The services agreement calls for payments of $5,000, and
accrues an additional $3,000 per month in fees to be paid by common
stock at the time of a public offering. The accrued balance due
under the cash portion as of September 30, 2016 and December 31,
2015 was $25,000 and $10,000 respectively, and the accrued balance
due under the stock portion was $72,000 and $45,000, respectively.
The Company can cancel the contract at any time without
notice.
F-26
Effective
January 1, 2015, we entered into an office lease agreement with
Green Court, LLC, a Michigan limited liability company, for
approximately 3,657 rentable square feet of space, for the initial
monthly rent of $5,986, which commenced on January 1, 2015 for an
initial term of 60 months. Under the terms of the lease the Company
has an option on the same space for an additional 60 month term.
Future minimum payments under this lease are as
follows:
|
2016
|
$ 17,958
|
|
2017
|
75,302
|
|
2018
|
77,190
|
|
2019
|
79,125
|
|
2020
|
81,460
|
|
Total
|
$ 331,035
|
On
April 16, 2015, the Company entered into an at-will employment
agreement with its Chief Executive Officer. The employment
agreement requires annual base salary payments of $250,000 per year
with a bonus potential of 50% of the then current base salary. In
addition, the executive has been granted an option to purchase
124,248 shares of Company's common stock exercisable at $2.86 per
share, vesting in 3 equal annual installments on each anniversary
of its three year term. The agreement also provides for severance
compensation if terminated other than for cause (as defined) of 6
months of the then applicable base salary if the CEO has been
employed at least 6 months, and compensation equal to 12 months of
the then applicable base salary if employed over 12
months.
Note 9 – Subsequent Events
The
Company has filed a registration statement on Form S-1 with
the U.S. Securities and Exchange Commission in connection with a
proposed offering of its securities. On December 3, 2016, the board
of directors of the Company and stockholders holding a majority of
the outstanding shares of the Company’s common stock approved
resolutions authorizing the board of directors to effect a reverse
split of the Company’s common stock at certain exchange
ratios ranging from 1:1.50 to
1:2.50, with the board of directors
retaining the discretion as to whether to implement the reverse
stock split and which exchange ratio to implement. Following the
effectiveness of the registration statement of which this
prospectus is a part, and prior to the closing of this offering,
the Company anticipates that it will effect the reverse stock split
at a ratio of 1 share for each 2.50 shares.
Since
the 1 for 2.50 reverse stock split is to be effected
after the effectiveness of the registration statement, the
historical share information included in the accompanying financial
statements and notes hereto do not assume the 1 for 2.50
reverse stock split, and accordingly have not been
adjusted.
On a
pro forma basis, taking into account the anticipated reverse stock
split at a ratio of 1 share for each 2.50 shares outstanding as of
the following dates of presentation, the Company’s earnings
per share would change as follows:
As
Presented:
|
|
Nine Months
|
Nine Months
|
Three Months
|
Three Months
|
|
|
Ended
|
Ended
|
Ended
|
Ended
|
|
|
September 30, 2016
|
September 30, 2015
|
September 30, 2016
|
September 30, 2015
|
|
Net
Loss
|
$(2,088,491)
|
$(1,820,471)
|
$(979,899)
|
$(540,987)
|
|
Basic
and diluted net loss per common share
|
$(0.82)
|
$(0.81)
|
$(0.39)
|
$(0.22 )
|
|
Weighted
average shares outstanding, basic and diluted
|
2,531,566
|
2,250,011
|
2,531,808
|
2,458,090
|
Pro
forma:
|
|
Nine Months
|
Nine Months
|
Three Months
|
Three Months
|
|
|
Ended
|
Ended
|
Ended
|
Ended
|
|
|
September 30, 2016
|
September 30, 2015
|
September 30, 2016
|
September 30, 2015
|
|
Net
Loss
|
$(2,088,491)
|
$(1,820,471)
|
$(979,899)
|
$(540,987)
|
|
Basic
and diluted net loss per common share
|
$(2.06)
|
$(2.05)
|
$(0.97)
|
$(0.55)
|
|
Weighted
average shares outstanding, basic and diluted
|
1,012,626
|
900,004
|
1,012,723
|
983,236
|
F-27
2,000,000
Units

PROSPECTUS
Dougherty & Company
Dawson
James Securities, Inc.
Until
, 2017, all dealers that effect transactions in these
securities, whether or not participating in this offering, may be
required to deliver a prospectus. This is in addition to
the dealers’ obligation to deliver a prospectus when acting
as underwriters and with respect to their unsold allotments or
subscriptions.
PART
II
INFORMATION
NOT REQUIRED IN PROSPECTUS
ITEM
13. OTHER EXPENSES OF ISSUANCE AND DISTRIBUTION
The
following table sets forth the costs and expenses, other than the
underwriting discount, payable by us in connection with the sale
and distribution of the securities being registered. All amounts
are estimated except the SEC registration fee, the FINRA filing fee
and the initial listing fee for The Nasdaq Capital
Market.
|
SEC
Filing Fee
|
$3,366
|
|
FINRA
Fee
|
$4,857
|
|
Underwriters'
Legal Fees and Expenses
|
$ 150,000
|
|
Nasdaq
Fee
|
$50,000
|
|
Printing
Expenses
|
$35,000
|
|
Accounting
Fees and Expenses
|
$50,000
|
|
Legal
Fees and Expenses
|
$250,000
|
|
Transfer
Agent and Registrar Expenses
|
$10,000
|
|
Miscellaneous
|
$46,777
|
|
|
|
|
Total
|
$600,000
|
ITEM
14. INDEMNIFICATION OF DIRECTORS AND OFFICERS
The
following summary is qualified in its entirety by reference to the
complete text of any statutes referred to below and the Fourth
Amended and Restated Certificate of Incorporation of ENDRA Life Sciences Inc., a Delaware
corporation.
Section
145 of the General Corporation Law of the State of Delaware (the
“DGCL”) permits a Delaware corporation to indemnify any
person who was or is a party or is threatened to be made a party to
any threatened, pending or completed action, suit or proceeding,
whether civil, criminal, administrative or investigative (other
than an action by or in the right of the corporation) by reason of
the fact that the person is or was a director, officer, employee or
agent of the corporation, or is or was serving at the request of
the corporation as a director, officer, employee or agent of
another corporation, partnership, joint venture, trust or other
enterprise, against expenses (including attorneys’ fees),
judgments, fines and amounts paid in settlement actually and
reasonably incurred by the person in connection with such action,
suit or proceeding if the person acted in good faith and in a
manner the person reasonably believed to be in or not opposed to
the best interests of the corporation, and, with respect to any
criminal action or proceeding, had no reasonable cause to believe
the person’s conduct was unlawful.
In the
case of an action by or in the right of the corporation, Section
145 of the DGCL permits a Delaware corporation to indemnify any
person who was or is a party or is threatened to be made a party to
any threatened, pending or completed action or suit by reason of
the fact that the person is or was a director, officer, employee or
agent of the corporation, or is or was serving at the request of
the corporation as a director, officer, employee or agent of
another corporation, partnership, joint venture, trust or other
enterprise, against expenses (including attorneys’ fees)
actually and reasonably incurred by the person in connection with
such action, suit or proceeding if the person acted in good faith
and in a manner the person reasonably believed to be in or not
opposed to the best interests of the corporation and except that no
indemnification shall be made in respect of any claim, issue or
matter as to which such person shall have been adjudged to be
liable to the corporation unless and only to the extent that the
Court of Chancery or the court in which such action or suit was
brought shall determine upon application that, despite the
adjudication of liability but in view of all the circumstances of
the case, such person is fairly and reasonably entitled to
indemnity for such expenses that the Court of Chancery or such
other court shall deem proper.
II-1
Section
145 of the DGCL also permits a Delaware corporation to purchase and
maintain insurance on behalf of any person who is or was a
director, officer, employee or agent of the corporation, or is or
was serving at the request of the corporation as a director,
officer, employee or agent of another corporation, partnership,
joint venture, trust or other enterprise against any liability
asserted against such person and incurred by such person in any
such capacity, or arising out of such person’s status as
such, whether or not the corporation would have the power to
indemnify such person against such liability under Section 145 of
the DGCL.
Article
NINTH of our Fourth Amended and Restated Certificate of
Incorporation states that our directors shall not be personally
liable to us or to our stockholders for monetary damages for any
breach of fiduciary duty as a director, notwithstanding any
provision of law imposing such liability. Under Section 102(b)(7)
of the DGCL, the personal liability of a director to the
corporation or its stockholders for monetary damages for breach of
fiduciary duty can be limited or eliminated except (i) for any
breach of the director’s duty of loyalty to the corporation
or its stockholders; (ii) for acts or omissions not in good faith
or which involve intentional misconduct or a knowing violation of
law; (iii) under Section 174 of the DGCL (relating to unlawful
payment of dividend or unlawful stock purchase or redemption); or
(iv) for any transaction from which the director derived an
improper personal benefit.
Article
EIGHTH of our Fourth Amended and Restated Certificate of
Incorporation provides that we shall indemnify (and advance
expenses to) our officers and directors to the full extent
permitted by the DGCL.
Effective upon the
closing of this offering, we will have directors’ and
officers’ liability insurance insuring our directors and
officers against liability for acts or omissions in their
capacities as directors or officers, subject to certain exclusions.
Such insurance also insures us against losses which we may incur in
indemnifying our officers and directors.
As
permitted by the DGCL, prior to the closing of the offering we plan
to enter into indemnification agreements with each of our directors
and executive officers that require us to indemnify such persons
against various actions including, but not limited to, third-party
actions where such director or executive officer, by reason of his
or her corporate status, is a party or is threatened to be made a
party to an action, or by reason of anything done or not done by
such director in any such capacity. We intend to indemnify
directors and executive officers against all costs, judgments,
penalties, fines, liabilities, amounts paid in settlement by or on
behalf of such directors or executive officers and for any expenses
actually and reasonably incurred by such directors or executive
officers in connection with such action, if such directors or
executive officers acted in good faith and in a manner they
reasonably believed to be in or not opposed to our best interests,
and with respect to any criminal proceeding, had no reasonable
cause to believe their conduct was unlawful. We also intend to
advance to our directors and executive officers expenses (including
attorney’s fees) incurred by or on behalf of such directors
and executive officers in advance of the final disposition of any
action after our receipt of a statement or statements from
directors or executive officers requesting such payment or payments
from time to time, provided that such statement or statements are
preceded or accompanied by a written undertaking, by or on behalf
of such directors or executive officers, to repay such amount if it
shall ultimately be determined that they are not entitled to be
indemnified against such expenses by us.
The
indemnification agreements will also set forth certain procedures
that will apply in the event of a claim for indemnification or
advancement of expenses, including, among others, provisions about
submitting a written request to us that includes such documentation
and information as is reasonably available to the director or
executive officer and is reasonably necessary to determine
entitlement to indemnification and provisions. Prior to the closing
of this offering we plan to enter into an underwriting agreement,
which will provide that the underwriters are obligated, under some
circumstances, to indemnify our directors, officers and controlling
persons against specified liabilities.
II-2
ITEM
15. RECENT SALES OF UNREGISTERED SECURITIES
During
the past three years, we issued the following securities without
registration under the Securities Act of 1933, as amended (the
“Securities Act”).
Stock, Warrants and Convertible Notes
From
July 2013 through April 2014, we issued 384,625 shares of our
common stock at a price of $7.15 per share, for gross proceeds of
$2,750,015. The shares were sold to 64 investors.
From
July 2013 through April 2014, we issued warrants to purchase 34,968
shares of common stock at an exercise price of $14.30 and warrants
to purchase 305,951 shares of common stock at an exercise price of
$3.58 per share.
In
September 2014, we issued to certain accredited investors an
aggregate of 108,714 shares of our common stock at a price of $7.15
per share in exchange for the cancellation of outstanding principal
and accrued interest on senior promissory notes issued in July 2013
and for the cancellation of certain ten-year warrants issued to
these same investors. In addition, as consideration for its
consulting services, we issued to DALA LLC (i) 27,273 shares of our
common stock and (ii) a warrant to purchase 27,273 shares of common
stock at an exercise price of $7.15 per share, exercisable upon
consummation of a change-in-control transaction or an initial
public offering of our common stock.
In
January 2015, we issued 34,966 shares of our common stock at a
price of $7.15 per share, for gross proceeds of $250,000, to 3
accredited investors. Additionally, we issued warrants to purchase
8,742 shares of common stock at an exercise price of $7.15 per
share.
From
May 2015 through January 2016, pursuant to a warrant exercise
program, we issued 166,217 shares of our common stock at a price of
$3.58 per share, for aggregate gross proceeds of $594,224 in
connection with the exercise of outstanding warrants held by 41
accredited investors. As an inducement to exercise their warrants,
we issued to the investors exercising their warrants new warrants
to purchase 166,217 shares of common stock at an exercise price of
$14.30 per share in connection with a warrant exercise
program.
On June
23, 2015, we issued an aggregate of 10,606 shares of common stock
to two Company employees to satisfy accrued, but unpaid salary
obligations to the two employees.
In
January 2016, we issued promissory notes in the aggregate principal
amount of $50,000 to three accredited investors.
In
April and May 2016, we issued convertible promissory notes in the
aggregate principal amount of $1,199,448 to 62 accredited
investors.
In July
2016 we issued convertible promissory notes in the aggregate
principal amount of $186,389 to three accredited
investors.
The
offers, sales and issuances of the securities described above were
deemed to be exempt from registration under the Securities Act in
reliance on the exemption provided by Section 4(a)(2) of the
Securities Act. Each of the recipients of securities in these
transactions was an accredited investor and there was no form of
general solicitation or general advertising relating to the
offer.
II-3
Stock
Options
In
November 2013, we granted options to purchase an aggregate of
92,808 shares of our common stock under our Amended and Restated
2013 Stock Incentive Plan at an exercise price of $7.15 per share,
to our executive officers, employees, certain advisors and members
of our Board of Directors. 78,932 options have a term of five years
and vests immediately or over 3 years in 3 equal installments,
remaining options were fully vested on the date of
issuance.
In May
2014, we granted to each member of our Board of Directors, as
consideration for their agreement to serve on the Board of
Directors, stock options to purchase an aggregate of 27,658 shares
of our common stock at an exercise price of $7.15 per share. The
options have a term of five years and were fully vested on the date
of issuance.
In May
2015, we granted stock options to purchase an aggregate of 1,364
shares of our common stock under our Second Amended and Restated
2013 Stock Incentive Plan at an exercise price of $7.15 per share,
to two employees. Each option has a term of five years and vests
over three years in three equal installments.
In July
2015, we granted stock options to purchase 49,699 shares of our
common stock under our Second Amended and Restated 2013 Stock
Incentive Plan at an exercise price of $7.15 per share, to a
Company employee. The options have a term of five years and vest
over three years in three equal installments. We also granted stock
options to purchase 5,000 shares of our common stock under our
Second Amended and Restated 2013 Stock Incentive Plan, which
options have an exercise price of $7.15 per share to a new member
of our Board of Directors.
In July
2015, we granted stock options to purchase 23,076 shares of common
stock under our Second Amended and Restated 2013 Stock Incentive
Plan at an exercise price of $7.15 per share, to each existing
independent member of our Board of Directors. Each option has a
term of five years and was fully vested on the date of
issuance.
In
January 2016, we granted stock options to purchase 13,847 shares of
common stock under our Second Amended and Restated 2013 Stock
Incentive Plan at an exercise price of $7.15 per share, to each
existing independent member of our Board of Directors and to each
member of our Scientific Advisory Board. Each option has a term of
five years and was fully vested on the date of
issuance.
All of
the stock options described above were granted in reliance upon an
available exemption from the registration requirements of the
Securities Act, including those contained in Rule 701 promulgated
under Section 3(b) of the Securities Act. Among other things, we
relied on the fact that, under Rule 701, companies that are not
subject to the reporting requirements of Section 13 or Section
15(d) of the Exchange Act are exempt from registration under the
Securities Act with respect to certain offers and sales of
securities pursuant to “compensatory benefit plans” as
defined under that rule. We believe that all of the options
described above were issued pursuant qualifying “compensatory
benefit plans”.
II-4
ITEM
16. EXHIBITS
|
Exhibit No.
|
|
Description of Document
|
|
1.1
|
|
Form of Underwriting Agreement**
|
|
3.1
|
|
Third Amended and Restated Certificate of Incorporation of the
Registrant, as currently in effect*
|
|
3.2
|
|
Bylaws of the Registrant, as currently in effect*
|
|
3.3
|
|
Form of Fourth Amended and Restated Certificate of Incorporation of
the Registrant, to be in effect upon completion of this
offering*
|
|
3.4
|
|
Form of Amended and Restated Bylaws of the Registrant, to be in
effect upon completion of this offering*
|
|
3.5
|
|
Certificate
of Amendment to Certificate of Incorporation of the Registrant, as
currently in effect*
|
|
3.6
|
|
Form of
Certificate of Amendment to Certificate of Incorporation of the
Registrant, to be in effect upon completion of this
offering*
|
|
4.1
|
|
Specimen Certificate representing shares of common stock of the
Registrant*
|
|
4.2
|
|
Form of Warrant Agreement and Warrant**
|
|
4.3
|
|
Form of
Underwriters' Warrants*
|
|
4.4
|
|
Form of Warrant to Purchase Common Stock issued to the placement
agent in the Registrant's 2014 private placement
offering*
|
|
4.5
|
|
Form of Warrant to Purchase Common Stock issued pursuant to
2013-2014 Bridge Financing*
|
|
4.6
|
|
Form of Warrant to Purchase Common Stock issued pursuant to 2015
Warrant Exercise Program*
|
|
4.7
|
|
Form of Senior Promissory Note issued pursuant to Securities
Purchase Agreement dated July 10, 2013*
|
|
4.8
|
|
Form of Convertible Promissory Note*
|
|
4.9
|
|
Form of Promissory Note issued by the Registrant to certain members
of the Board of Directors*
|
|
4.10
|
|
Form of Unit Certificate**
|
|
5.1
|
|
Form of Opinion of K&L Gates LLP**
|
|
10.1
|
|
ENDRA Life Sciences Inc. Second Amended and Restated 2013 Stock
Incentive Plan*†
|
|
10.2
|
|
Form of Non-Qualified Stock Option Award under Second Amended and
Restated 2013 Stock Incentive Plan*†
|
|
10.3
|
|
Form of Incentive Stock Option Agreement under Second Amended and
Restated 2013 Stock Incentive Plan*†
|
|
10.4
|
|
Form of ENDRA Life Sciences Inc. 2016 Omnibus Incentive Plan, to be
in effect upon completion of this offering*†
|
|
10.5
|
|
Form of Stock Option Award under 2016 Omnibus Incentive
Plan*†
|
|
10.6
|
|
Form of Restricted Stock Unit Award under 2016 Omnibus Incentive
Plan*†
|
|
|
Non-Employee Director Compensation Policy, to be in effect upon
completion of this offering†
|
|
|
10.8
|
|
Form of Indemnification Agreement by and between the Registrant and
each of its directors and executive officers*
|
|
10.9
|
|
Form of Amended and Restated Employment Agreement, by and between
the Registrant and Francois Michelon, to be in effect upon
completion of this offering*†
|
|
10.10
|
|
Form of Amended and Restated Employment Agreement, by and between
the Registrant and Michael Thornton, to be in effect upon
completion of this offering*†
|
|
10.11
|
|
Consulting Agreement, dated July 23, 2014, by and between the
Registrant and StoryCorp Consulting*†
|
|
10.12
|
|
Form of Securities Purchase Agreement, dated July 10, 2013, by and
between the Registrant and the purchasers named
therein*
|
|
10.13
|
|
Form of Securities Purchase Agreement, dated July 10, 2013, by and
between the Registrant and the purchasers named
therein*
|
|
10.14
|
|
Form of Exchange Agreement, dated September 26, 2014, between the
Registrant and certain security holders*
|
|
10.15
|
|
Form of Subscription Agreement between the Registrant and investors
in the Registrant's 2013 private placement offering*
|
|
10.16
|
|
Form of Subscription Agreement between the Registrant and investors
in the Registrant's 2014 private placement offering*
|
|
10.17
|
|
Collaborative Research Agreement, dated April 22, 2016, by and
between the Registrant and General Electric Company*
|
|
10.18
|
|
Gross Lease, dated January 1, 2015, between the Registrant and
Green Court LLC*
|
|
10.19
|
|
Sublicense Agreement, dated August 2, 2007, by and between the
Registrant and Optosonics, Inc.*
|
|
10.20
|
|
Amendment to Sublicense Agreement, dated January 18, 2011, by and
between the Registrant and Optosonics, Inc.*
|
|
14.1
|
|
Code of Ethics, to be in effect upon completion of this
offering*
|
|
|
Consent of RBSM LLP, Independent Registered Public Accounting
Firm
|
|
|
23.2
|
|
Consent of K&L Gates LLP (included in Exhibit
5.1)*
|
|
24.1
|
|
Power of Attorney*
|
|
|
|
|
|
*Previously filed.
**To be
filed by amendment.
†Indicates management compensatory plan, contract or
arrangement.
|
||
II-5
ITEM
17. UNDERTAKINGS
The
undersigned registrant hereby undertakes:
(1) To
file, during any period in which offers or sales are being made, a
post-effective amendment to this registration
statement:
(i) To
include any prospectus required by section 10(a)(3) of the
Securities Act of 1933;
(ii) To
reflect in the prospectus any facts or events arising after the
effective date of the registration statement (or the most recent
post-effective amendment thereof) which, individually or in the
aggregate, represent a fundamental change in the information set
forth in the registration statement. Notwithstanding the foregoing,
any increase or decrease in volume of securities offered (if the
total dollar value of securities offered would not exceed that
which was registered) and any deviation from the low or high end of
the estimated maximum offering range may be reflected in the form
of prospectus filed with the Commission pursuant to Rule 424(b) if,
in the aggregate, the changes in volume and price represent no more
than a 20% change in the maximum aggregate offering price set forth
in the “Calculation of Registration Fee” table in the
effective registration statement;
(iii) To
include any material information with respect to the plan of
distribution not previously disclosed in the registration statement
or any material change to such information in the registration
statement.
(2) That,
for the purpose of determining any liability under the Securities
Act of 1933, each such post-effective amendment shall be deemed to
be a new registration statement relating to the securities offered
therein, and the offering of such securities at that time shall be
deemed to be the initial bona fide offering thereof.
(3) To
remove from registration by means of a post-effective amendment any
of the securities being registered that remain unsold at the
termination of the offering.
(4) That,
for the purpose of determining liability of the registrant under
the Securities Act to any purchaser in the initial distribution of
the securities: The undersigned registrant undertakes that in a
primary offering of securities of the undersigned registrant
pursuant to this registration statement, regardless of the
underwriting method used to sell the securities to the purchaser,
if the securities are offered or sold to such purchaser by means of
any of the following communications, the undersigned registrant
will be a seller to the purchaser and will be considered to offer
or sell such securities to such purchaser: (i) any preliminary
prospectus or prospectus of the undersigned registrant relating to
the offering required to be filed pursuant to Rule 424; (ii) any
free writing prospectus relating to the offering prepared by or on
behalf of the undersigned registrant or used or referred to by the
undersigned registrant; (iii) the portion of any other free writing
prospectus relating to the offering containing material information
about the undersigned registrant or its securities provided by or
on behalf of the undersigned registrant; and (iv) any other
communication that is an offer in the offering made by the
undersigned registrant to the purchaser.
(5) To
provide to the underwriter at the closing specified in the
underwriting agreements certificates in such denominations and
registered in such names as required by the underwriter to permit
prompt delivery to each purchaser.
(6) For
purposes of determining any liability under the Securities Act of
1933, the information omitted from the form of prospectus as filed
as part of this registration statement in reliance upon Rule 430A
and contained in a form of prospectus filed by the registrant
pursuant to Rule 424(b)(1) or (4) or 497(h) under the Securities
Act shall be deemed to be part of this registration statement as of
the time it was declared effective.
(7) For
the purpose of determining any liability under the Securities Act
of 1933, each post-effective amendment that contains a form of
prospectus shall be deemed to be a new registration statement
relating to the securities offered therein, and the offering of
such securities at that time shall be deemed to be the initial bona
fide offering thereof.
(8) Insofar
as indemnification for liabilities arising under the Securities Act
of 1933 may be permitted to directors, officers and controlling
persons of the registrant pursuant to the foregoing provisions, or
otherwise, the registrant has been advised that in the opinion of
the Securities and Exchange Commission such indemnification is
against public policy as expressed in the Securities Act and is,
therefore, unenforceable. In the event that a claim for
indemnification against such liabilities (other than the payment by
the registrant of expenses incurred or paid by a director, officer
or controlling person of the registrant in the successful defense
of any action, suit or proceeding) is asserted by such director,
officer or controlling person in connection with the securities
being registered, the registrant will, unless in the opinion of its
counsel the matter has been settled by controlling precedent,
submit to a court of appropriate jurisdiction the question whether
such indemnification by it is against public policy as expressed in
the Securities Act and will be governed by the final adjudication
of such issue.
II-6
SIGNATURES
Pursuant to the
requirements of the Securities Act of 1933, the registrant has duly
caused this Amendment No. 5 to the registration
statement to be signed on its behalf by the undersigned, thereunto
duly authorized, in the City of Ann Arbor, State of Michigan, on
this 20th
day of January, 2017.
|
|
ENDRA
Life Sciences Inc.
/s/Francois
Michelon
Francois
Michelon
Chief
Executive Officer and Director
(Principal
Executive Officer)
|
Pursuant to the
requirements of the Securities Act of 1933, this Amendment No.
5 to the registration statement has been signed
by the following persons in the capacities and on the dates
indicated.
|
Dated:
January 20, 2017
|
/s/Francois
Michelon
Francois
Michelon
Chief
Executive Officer and Director
(Principal
Executive Officer)
|
|
Dated: January
20, 2017
|
/s/David R.
Wells
David
R. Wells
Interim
Chief Financial Officer
(Principal
Financial and Accounting Officer)
|
|
Dated: January
20, 2017
|
/s/***
Anthony
DiGiandomenico, Director
|
|
Dated: January
20, 2017
|
/s/***
Sanjiv
Gambhir, M.D., Ph.D, Director
|
|
Dated: January
20, 2017
|
/s/***
Michael
Harsh, Director
|
|
Dated: January
20, 2017
|
/s/***
Alexander Tokman,
Director
|
|
***
By: /s/
Francois Michelon
Francois Michelon Attorney-in-fact |
|
II-7
