Attached files
| file | filename |
|---|---|
| EX-99.1 - EXHIBIT 99.1 - GENOCEA BIOSCIENCES, INC. | gnca0030036monthdata010520.htm |
| 8-K - 8-K - GENOCEA BIOSCIENCES, INC. | gnca-20170105x8k.htm |

Positive 6 Month Clinical
Efficacy Results
GEN-003 Immunotherapy
Candidate for Genital Herpes
Phase 2b Study
5 January 2017
Exhibit 99.2

This presentation contains “forward-looking” statements that are within the meaning of federal securities laws
and are based on our management’s beliefs and assumptions and on information currently available to
management. Forward-looking statements include information concerning our possible or assumed future
results of operations, business strategies, financing plans, competitive position, industry environment, potential
growth opportunities, potential market opportunities and the effects of competition.
Forward-looking statements include all statements that are not historical facts and can be identified by terms
such as “anticipates,” “believes,” “could,” “seeks,” “estimates,” “intends,” “may,” “plans,” “potential,”
“predicts,” “projects,” “should,” “will,” “would” or similar expressions and the negatives of those terms. Forward-
looking statements represent our management’s beliefs and assumptions only as of the date of this
presentation. Our operations involve risks and uncertainties, many of which are outside our control, and any
one of which, or combination of which, could materially affect our results of operations and whether the
forward-looking statements ultimately prove to be correct. Factors that may materially affect our results of
operations include, among other things, those listed in our Annual Report on Form 10-K and other filings with the
Securities and Exchange Commission (“SEC”). Except as required by law, we assume no obligation to update
these forward-looking statements publicly, or to update the reasons actual results could differ materially from
those anticipated in the forward-looking statements, even if new information becomes available in the future.
You may get copies of our Annual Report on Form 10-K, Quarterly Report on Form 10-Q and our other SEC filings
for free by visiting EDGAR on the SEC website at http://www.sec.gov.
2
Safe Harbor Statement

Successful Phase 2b Clinical Results – Advancing
Potential Blockbuster to Phase 3
• Statistically significant improvements in clinical disease versus
placebo across multiple endpoints 6 months post-dosing
• Compelling profile cemented across three clinical studies
– Durable impact on clinical disease
– Significant reduction in viral shedding
– At most once-yearly maintenance dosing
– Safety profile appropriate for therapeutic setting
• Important critical path milestones imminent
– End-of-phase 2 meeting with the FDA: Q1
– Phase 3 start: Q4
• If approved, would be the first new treatment for genital herpes
infections in more than 20 years
• Large unmet patient need in a disease of epidemic proportions
– Potential ~$2 billion global revenue opportunity*
3
*Source: Genocea market research

4
Millions Infected with Genital Herpes Need a New
Treatment Option
•Most patients do not persist
– Loathe “daily reminder”
– Incomplete efficacy
• Little benefit
– No impact on
recurrence frequency
– Small reduction in
duration
2.6
5.2
4.4
Suppressive
(Daily antiviral
use)
Episodic
(Use antivirals
upon
recurrences)
•No benefits
GEN-003 Target Profile
• No pill burden
• Similar disease control
• Potential additive
effect as combo
• Reduce clinical
disease:
• Total lesion days
• Recurrence
number
• Recurrence
duration
• Reduce viral
shedding
• Minimize treatment
burden
* Millions of US patients
Diagnosed,
untreated
Benefits from Antivirals Today
Source: Genocea market research
Treatment Distribution*

6 Month Clinical Readout
GEN-003 Phase 2b Trial
5

• Overall goal
– Evaluate Phase 3-ready formulation of GEN-003 and define dose for
Phase 3 trials
• Primary objective (completed successfully in September 2016(1))
– Compare efficacy of two dose levels of GEN-003 and placebo by
impact on viral shedding
• Secondary objectives
– Evaluate impact on clinical disease vs. placebo at 6 and 12 months
– Evaluate impact on viral shedding vs. placebo at 6 and 12 months
– Immunogenicity
– Safety and tolerability
6
Goals & Objectives
Note (1) Press release - http://ir.genocea.com/releasedetail.cfm?ReleaseID=991389

• Randomized, double-blind, placebo-controlled
• 131 subjects with a history of recurrent genital herpes
• 3 dose groups
– Placebo (n=44)
– 60 µg per antigen / 50 µg of Matrix-M adjuvant (n=43)
– 60 µg per antigen / 75 µg of Matrix-M adjuvant (n=44)
• Most other design elements consistent with prior GEN-003 trials
– Inclusion / exclusion criteria, demographics, sites, dose regimen,
viral shedding swabbing compliance
7
Study Design

• Phase 2b – more comprehensive
– Lesion data collected daily
throughout trial
– Data recorded via smartphone-
based app with reminders
• Previous GEN-003 studies
– Lesion data only during 28-day
observation periods
• Pre-treatment
• Immediately post-treatment
• 6 & 12 months post-treatment
– Data recorded via paper diary
based on recall
8
Improved Lesion Data Collection Strengthens
GEN-003 Clinical Endpoints Assessment
• Capture data on
additional clinical
endpoints:
– Number of recurrences
– Duration of recurrences
• Enables Phase 3-like
analysis across doses after
treatment, rather than
versus baseline
Analytical Enhancements
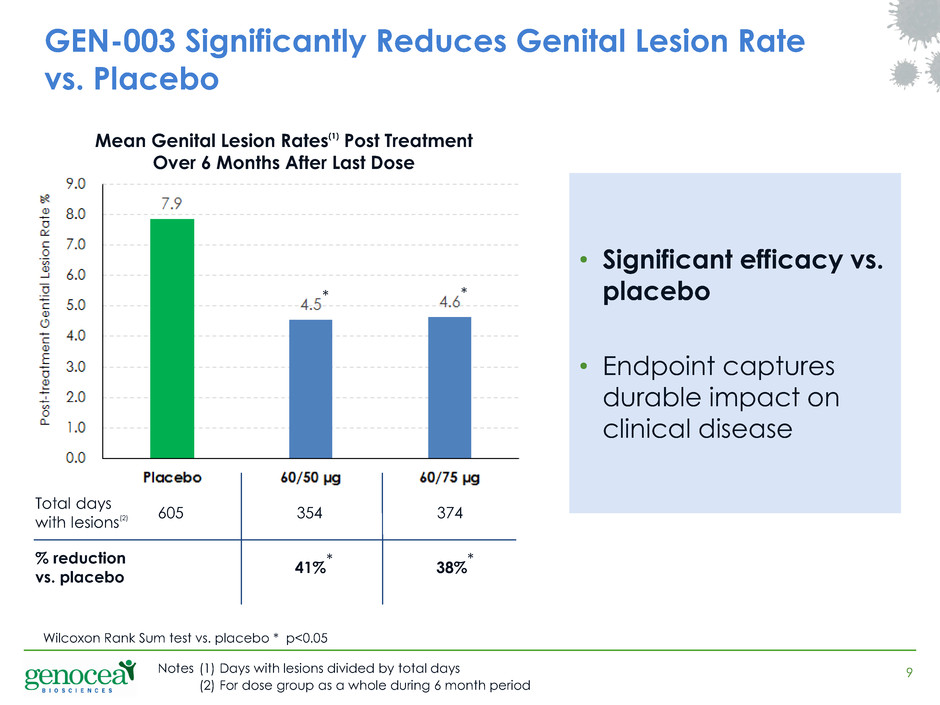
Wilcoxon Rank Sum test vs. placebo * p<0.05
605 354 374
41% 38%
Total days
with lesions(2)
% reduction
vs. placebo
* *
Notes (1) Days with lesions divided by total days
(2) For dose group as a whole during 6 month period
• Significant efficacy vs.
placebo
• Endpoint captures
durable impact on
clinical disease
9
GEN-003 Significantly Reduces Genital Lesion Rate
vs. Placebo
Mean Genital Lesion Rates(1) Post Treatment
Over 6 Months After Last Dose
* *

10
GEN-003 Significantly Reduces both the Number
and Duration of Recurrences Versus Placebo
Mean Number of Recurrences
Over 6 Months Following Last Dose
Mean Duration of Recurrences
Over 6 Months Following Last Dose
• Reducing the frequency and duration of recurrences is important
to both patients and their caregivers
Wilcoxon Rank Sum test vs. placebo * p<0.05
*
*

11
GEN-003 Drives Significant Improvement in Number of
Subjects Recurrence Free at 6 Months
Kaplan-Meier Estimate of % Subjects
Recurrence Free at 6 Months
• GEN-003 patients 2-3
times more likely to be
completely recurrence-
free than placebo at 6
months
• GEN-003 efficacy
consistent with Phase 2
clinical trial
Log rank test vs. placebo * p<0.05
*
*
*

• Statistically significant improvements in clinical disease versus
placebo across multiple endpoints 6 months post-dosing
• Significant reductions in viral shedding
• Prioritizing 60 µg per antigen / 50 µg adjuvant based on clinical &
virologic efficacy and tolerability
• Finalizing Phase 3 program preparations with FDA in Q1
12
Positive GEN-003 Phase 2b Results Provide Strong
Foundation for Phase 3

13
Phase 2b Clinical Efficacy Data Maintains Momentum
to Phase 3 Start
2017
12 month
FDA EoP2
Phase 2b
Phase 3
Program
Regulatory
Phase 2b
maintenance
dosing
Phase 2
extension 24 month
Antiviral
combination
• Could GEN-003 be dosed every 2 years?
• Introduced improved process material
• Demonstrated clinical, virologic efficacy
• Safety and efficacy of
maintenance dosing
• Pending Ph 2 extension
• Safety and benefits of
GEN-003 with daily
antivirals
6 month

• Large unmet patient need in a disease of epidemic proportions
– Potential ~$2 billion global revenue opportunity*
• 3 successful clinical trials to date
– Clinical efficacy demonstrated against multiple endpoints reflecting
patient unmet need
– Durable for at least 1 year; annual maintenance dose possible
– Comprehensive dose exploration; consistent efficacy at selected dose
– Safety profile appropriate for therapeutic setting
• Multiple planned upcoming milestones
– Q1: End of Phase 2 meeting
– H2: 24-month Phase 2 data & 12-month Phase 2b data & combination
study
– Q4: Start Phase 3 trials
• Exploring global development and commercialization partner(s)
14
GEN-003: Phase 3-Ready Program with Blockbuster
Potential
*Source: Genocea market research

Q&A
15

Jonathan Poole
Chief Financial Officer
Phone: +1 617-876-8191
jonathan.poole@genocea.com
Liz Bryan
Spectrum Science
Communications
Phone: +1 202-587-2526
lbryan@spectrumscience.com
Investor inquiries: Media inquiries:
