Attached files
| file | filename |
|---|---|
| 8-K - 8-K - EAGLE PHARMACEUTICALS, INC. | a8-k081016.htm |

August 2016
Corporate Overview

2
Forward Looking Statements
• This presentation contains forward-looking information within the meaning of the Private Securities Litigation Reform Act of 1995, as
amended, and other securities laws. Forward-looking statements are statements that are not historical facts. Words such as “will,”,
“determined”, “confirming”, “underway,” ramping up,” “agreement,” “allows,” “well positioned,” “expect,” “may,” “intends,” “anticipate(s),”
“plan,” “enables,” “potentially,” “look forward,” “on track,” and similar expressions are intended to identify forward-looking statements. These
statements include, but are not limited to, statements regarding future events including, but not limited to: the continued commercial
performance of our marketed products, including BENDEKA, which is marketed by our partner Cephalon, and RYANODEX, which we
market ourselves as well as our ability to replicate our marketing successes for our other product candidates either through joint or direct
marketing efforts; the lack of a need for human safety and efficacy data for the submission of an NDA for Ryanodex and the adequacy of the
regulatory pathway to complete an NDA submission; the Company’s share repurchase authorization and timing and ability to repurchase
shares of the Company’s common stock under a share repurchase program; the business path forward for the Company beyond 2020; the
label expansions of Ryanodex for EHS patients and for the treatment of ecstasy and methamphetamine intoxication; the strength of the
Company’s cash position and the ability to optimize the deployment of capital and take advantage of market opportunities; the potential of the
Company’s pipeline to drive value beyond 2020; the contribution of the Ryanodex portfolio to the Company’s growth; the timing of Ryanodex
for EHS entering the market; and the advancement of the Company’s product candidates through the development process and the ability to
access significant new markets. All of such statements are subject to certain risks and uncertainties, many of which are difficult to predict and
generally beyond Eagle’s control, that could cause actual results to differ materially from those expressed in, or implied or projected by, the
forward-looking information and statements. Such risks include, but are not limited to: whether our animal studies will support the safety and
efficacy of Ryanodex for the treatment of EHS and ecstasy and methamphetamine intoxication; whether the FDA will ultimately approve
Ryanodex for these indications; whether the Company will adequately evaluate and respond to the FDA’s Complete Response Letter on RTU
bivalirudin; fluctuations in the trading volume and market price of shares of the Company's common stock, general business and market
conditions and management's determination of alternative needs and uses of the Company's cash resources which may affect the Company's
share repurchase program; the success of our commercial relationship with Teva and the parties’ ability to work effectively together; whether
Eagle and Teva will successfully perform their respective obligations under the license agreement; difficulties or delays in manufacturing; the
availability and pricing of third party sourced products and materials; the outcome of litigation involving any of our products or that may have
an impact on any of our products, successful compliance with FDA and other governmental regulations applicable to product approvals,
manufacturing facilities, products and/or businesses; general economic conditions; the strength and enforceability of our intellectual property
rights or the rights of third parties; competition from other pharmaceutical and biotechnology companies; the timing of product launches; the
successful marketing of our products; the risks inherent in the early stages of drug development and in conducting clinical trials; and other
factors that are discussed in Eagle’s Annual Report on Form 10-K for the year ended December 31, 2015, and its other filings with the U.S.
Securities and Exchange Commission. Readers are cautioned not to place undue reliance on these forward-looking statements that speak
only as of the date hereof, and we do not undertake any obligation to revise and disseminate forward-looking statements to reflect events or
circumstances after the date hereof, or to reflect the occurrence of or non-occurrence of any events.

3
About Eagle Pharmaceuticals
Fully commercial specialty pharmaceutical company
focused on hospitals and infusion centers
• Developing & commercializing injectable products that improve upon existing
formulations
• 5 products in-market delivering profits
• Key driver of profit through 2019
• Bendeka: line extension developed for Teva to replace Treanda
• 80% market share of ~$750mm market since Q1 2016 launch
• 20% royalty (~$125-$150mm annually) through 2019
• Next big product: Ryanodex
• Approved and selling in niche market to treat malignant hyperthermia
• Large worldwide market opportunity in treating Exertional Heat Stroke
• Expect to file NDA shortly
• Potential to treat brain hyperthermia in ecstasy and methamphetamine intoxication
• 125K ER cases annually in US alone
• Exciting additional pipeline programs
• Solid financial position: profitable, growing cash position, no debt
• Confident in future: purchased Ryanodex royalty obligation and authorized $75mm
stock buyback

4
• Profitable fully commercial specialty pharmaceutical company with a broad portfolio and
deep near-term pipeline
• Strong YoY growth anticipated beyond 2020 from product sales and royalty income in
multiple attractive markets
• 40+ salesforce focused on infusion centers, hospitals and oncology purchasing groups
• Solid balance sheet: $127.6 million in cash and receivables, no debt; $75 million share
repurchase
Investment Highlights
• 5 in-market products generating revenue and positive earnings
• Royalty Income: Bendeka™, Argatroban, Diclofenac
• Product Sales: Ryanodex®, Docetaxel
Growing
Commercialized
Portfolio
• Potential Ryanodex label expansion to treat Exertional Heat Stroke
and Ecstasy and Methamphetamine intoxication
• Potential Pemetrexed launch ahead of other generics
• Internal and AMRI development programs for 4+ product candidates
Pipeline with
near-term
catalysts driving
additional growth

5
Growing Product Portfolio in Attractive Markets
Mkt Size: $XX
Products Market Size
Bendamustine Bendeka™
• Launched January 2016; 80% market share
• Exclusive marketing license with Teva
• Orange book patents thru 2033
Bendamustine 500mL
• Right to launch as of May 2016
~$750M
Ryanodex Malignant hyperthermia (current indication)
• Dantrolene sodium injectable suspension
• Currently in-market
Exertional Heat Stroke (potential label expansion)
• Clinical trial completed
• FDA agreement for NDA filing
Ecstasy and Methamphetamine intoxication (potential label expansion)
• NIDA/NIH study underway
$75M1
$400M WW
125K+ ER cases in
2011
Bivalirudin • NDA filed May 2015
• Complete Response Letter received from FDA in March 2016
• In discussion with FDA regarding human immunogenicity study
$635M2
Additional Pipeline
Programs
RTU Pemetrexed (Lilly’s Alimta)
• NDA to be filed late 2016
• May launch ahead of other generics as early as mid 2019
AMRI development program
Eagle internal development
$1.1B
Additional In-Market
Products
• Docetaxel
• Argatroban (royalty)
• Diclofenac/misoprostol (royalty)
Expansion outside
US markets
• Multiple product expansion potential
in addition to US markets
1 Over 2 year stocking period.
2 Worldwide Angiomax/Angiox sales in 2014 of $635.7 million, source: The Medicines Company press release dated 2/18/2015.
6
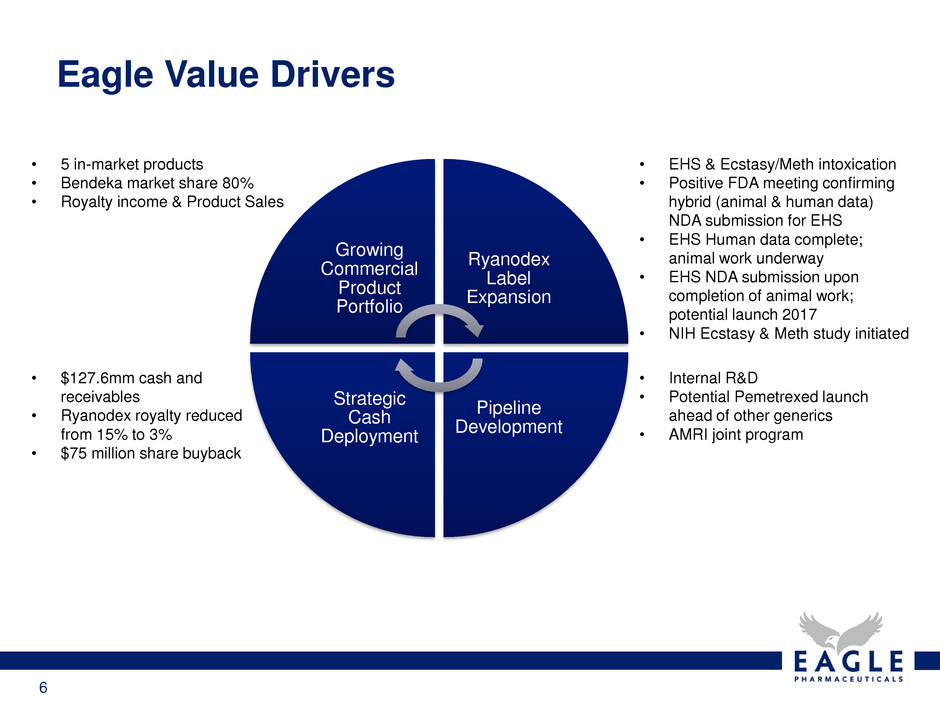
Eagle Value Drivers
Growing
Commercial
Product
Portfolio
Ryanodex
Label
Expansion
Pipeline
Development
Strategic
Cash
Deployment
• EHS & Ecstasy/Meth intoxication
• Positive FDA meeting confirming
hybrid (animal & human data)
NDA submission for EHS
• EHS Human data complete;
animal work underway
• EHS NDA submission upon
completion of animal work;
potential launch 2017
• NIH Ecstasy & Meth study initiated
• 5 in-market products
• Bendeka market share 80%
• Royalty income & Product Sales
• Internal R&D
• Potential Pemetrexed launch
ahead of other generics
• AMRI joint program
• $127.6mm cash and
receivables
• Ryanodex royalty reduced
from 15% to 3%
• $75 million share buyback

7
• $750 million market previously dominated by Teva’s Treanda®
• Option to launch tentatively approved Bendamustine RTD (500mL) in the U.S. at any
time
• Final j-code ruling expected Q4 2016; pursuing legal action on FDA denial of seven
years of orphan drug marketing exclusivity
• Six patents issued and listed in Orange Book covering periods ending within 2026-
2033 protecting the longevity of the bendamustine franchise
Bendamustine Opportunity
• Exclusive marketing license with Teva for BendekaTM
(bendamustine 50mL)
– Eagle receives 20% royalty on net sales of Bendeka
– Teva receives 20% royalty on profit if we take Bendeka back
– Option to terminate agreement when competitive product launches
– $25 million in additional milestone payments subject to achievement of certain
sales goals
• Market share goal with Teva is 90%
– Achieved 80% total market share as of August 5, 2016
• Eagle has option to take back Bendeka in 2019
– Upon generic bendamustine entrant and no unique J Code for Bendeka

8
Multiple Benefits of Bendeka™ (50mL in 10 min)
• Less chair time: 30 or 60 min. reduced to 10 min.
• Less volume & potential related issues: 50mL vs. 500mL
admixture
Patient
• Less nursing time requiredNurse
• Sodium-free administration for patients requiring restricted
sodium intake (e.g. cardiovascular and renal disorders)Clinical
• Additional patients treated in the cancer clinic enabled by
shorter infusion time
Economic
• Lower incidence of observed treatment emergent adverse
events (TEAEs)Safety
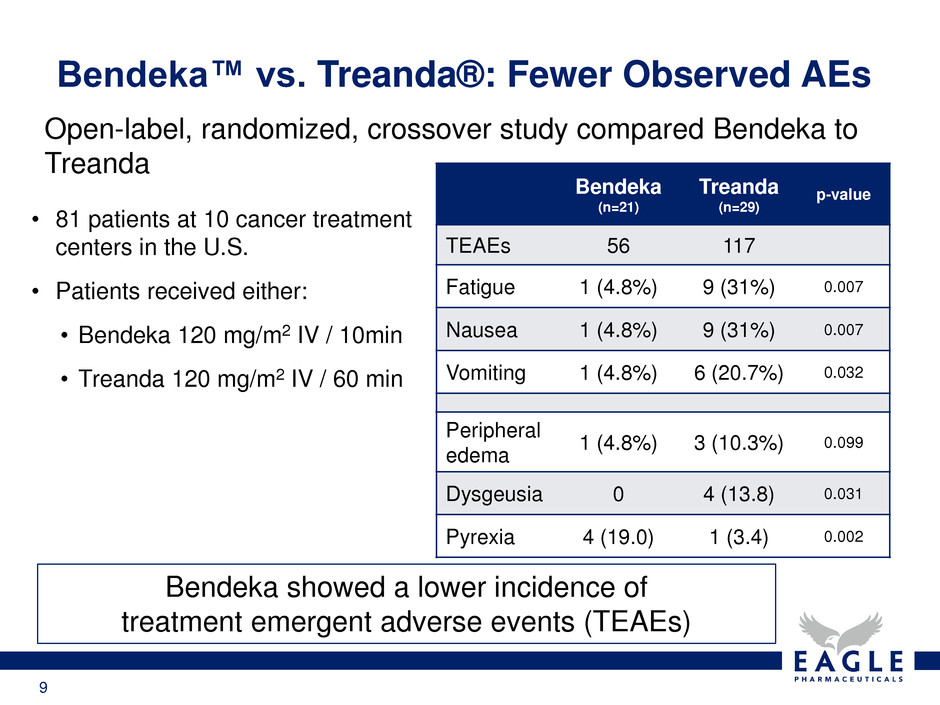
9
Bendeka showed a lower incidence of
treatment emergent adverse events (TEAEs)
Bendeka™ vs. Treanda®: Fewer Observed AEs
Open-label, randomized, crossover study compared Bendeka to
Treanda
Bendeka
(n=21)
Treanda
(n=29)
p-value
TEAEs 56 117
Fatigue 1 (4.8%) 9 (31%) 0.007
Nausea 1 (4.8%) 9 (31%) 0.007
Vomiting 1 (4.8%) 6 (20.7%) 0.032
Peripheral
edema
1 (4.8%) 3 (10.3%) 0.099
Dysgeusia 0 4 (13.8) 0.031
Pyrexia 4 (19.0) 1 (3.4) 0.002
• 81 patients at 10 cancer treatment
centers in the U.S.
• Patients received either:
• Bendeka 120 mg/m2 IV / 10min
• Treanda 120 mg/m2 IV / 60 min
10
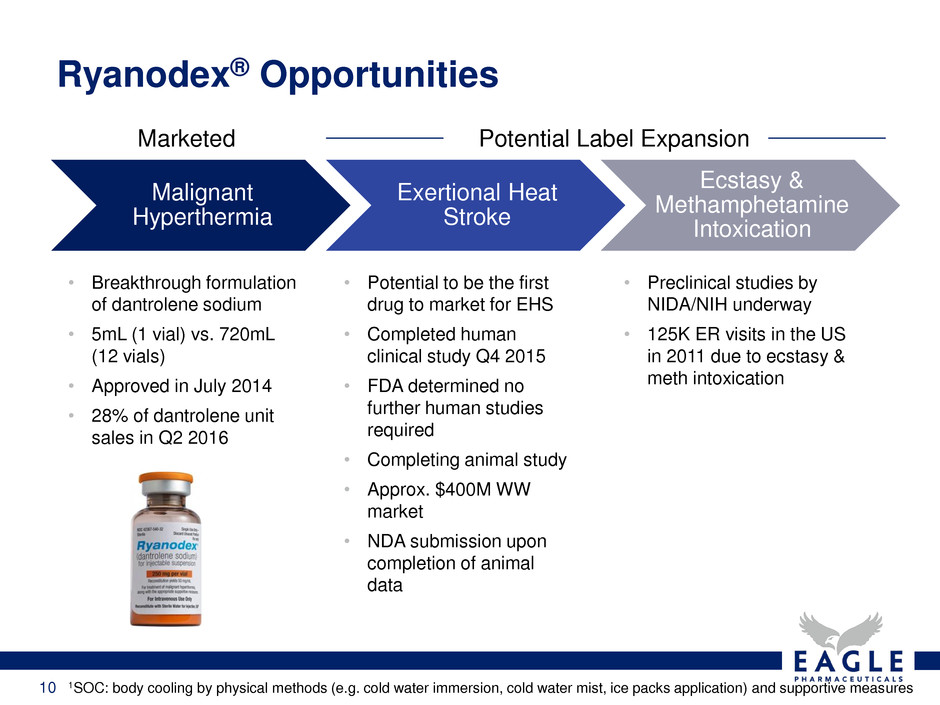
Ryanodex® Opportunities
1SOC: body cooling by physical methods (e.g. cold water immersion, cold water mist, ice packs application) and supportive measures
Malignant
Hyperthermia
Exertional Heat
Stroke
Ecstasy &
Methamphetamine
Intoxication
• Breakthrough formulation
of dantrolene sodium
• 5mL (1 vial) vs. 720mL
(12 vials)
• Approved in July 2014
• 28% of dantrolene unit
sales in Q2 2016
Marketed Potential Label Expansion
• Potential to be the first
drug to market for EHS
• Completed human
clinical study Q4 2015
• FDA determined no
further human studies
required
• Completing animal study
• Approx. $400M WW
market
• NDA submission upon
completion of animal
data
• Preclinical studies by
NIDA/NIH underway
• 125K ER visits in the US
in 2011 due to ecstasy &
meth intoxication

11
Ryanodex for Malignant Hyperthermia (MH)
1SOC: body cooling by physical methods (e.g. cold water immersion, cold water mist, ice packs application) and supportive measures
No change to Standard of Care (SOC)1 for MH
treatment in 30+ years
Optimized breakthrough formulation of dantrolene
sodium
Reduces to 5mL (1 vial) vs. 720 mL (12 vials) for
old product
Approved July 2014
Protected market position
• 5 patents issued + 2 filed
Orphan drug designation for MH (US & EU)
• Granted 7 years of market exclusivity in US

12
• Exertional Heat Stroke (EHS) unmet medical need
‒ Sudden, unpredictable and life-threatening condition
‒ Patient population impacted: military, student athletes, athletes,
construction workers, migrant workers, and firemen
‒ A leading cause of student athlete death (US) & non-combat military deaths
‒ Similarities to MH
• Potential for Ryanodex to be first to market for EHS
– Granted fast track and orphan drug designation
– Human study completed; animal study underway
– Potential for 7 years of exclusivity
• $400 million worldwide market
Ryanodex for EHS

13
• Safety & Efficacy study to evaluate Ryanodex for treatment of EHS
during the Hajj pilgrimage in Saudi Arabia (Sep 2015)
• 34 patients randomized 1:1 to receive SOC or SOC + Ryanodex
• Inclusion protocol criteria required that patients showed hallmark
clinical features of EHS including:
– 18-45 years of age who experienced exertional physical activity within previous 24
hours;
– Presence of neurological impairment, evaluated using the Glasgow Coma Scale;
– Core body temperature of at least 104 degrees Fahrenheit; and
– Tachycardia (at least 100 heart beats per minute)
EHS Human Clinical Study
July 2016 - FDA determined
NO additional human safety & efficacy studies required
14
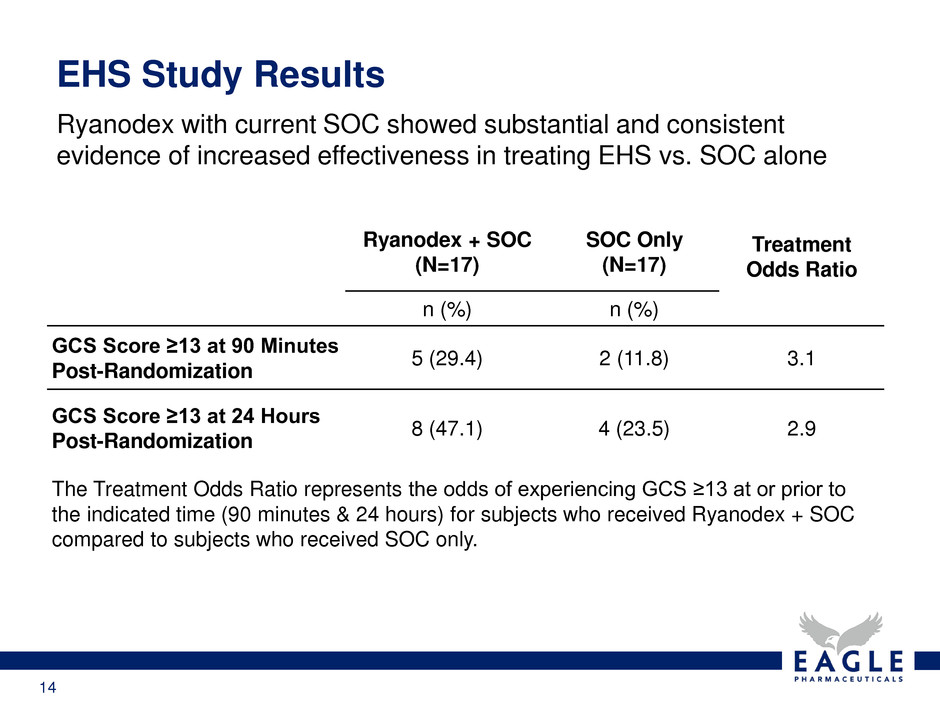
Ryanodex with current SOC showed substantial and consistent
evidence of increased effectiveness in treating EHS vs. SOC alone
EHS Study Results
Ryanodex + SOC
(N=17)
SOC Only
(N=17)
Treatment
Odds Ratio
n (%) n (%)
GCS Score ≥13 at 90 Minutes
Post-Randomization
5 (29.4) 2 (11.8) 3.1
GCS Score ≥13 at 24 Hours
Post-Randomization
8 (47.1) 4 (23.5) 2.9
The Treatment Odds Ratio represents the odds of experiencing GCS ≥13 at or prior to
the indicated time (90 minutes & 24 hours) for subjects who received Ryanodex + SOC
compared to subjects who received SOC only.
15
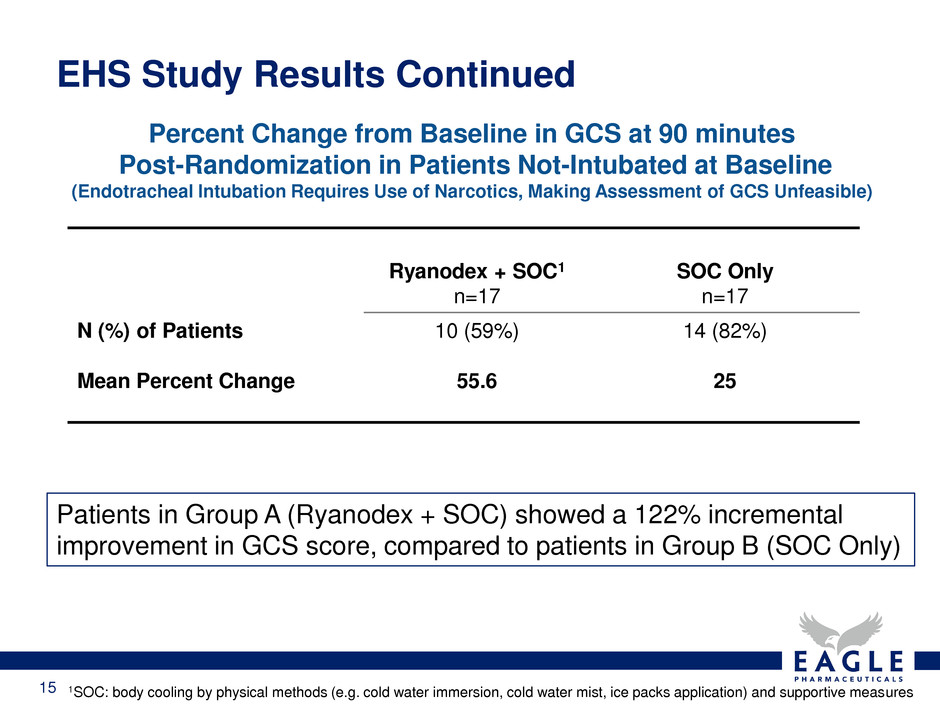
Ryanodex + SOC1
n=17
SOC Only
n=17
N (%) of Patients
Mean Percent Change
10 (59%)
55.6
14 (82%)
25
Percent Change from Baseline in GCS at 90 minutes
Post-Randomization in Patients Not-Intubated at Baseline
(Endotracheal Intubation Requires Use of Narcotics, Making Assessment of GCS Unfeasible)
Patients in Group A (Ryanodex + SOC) showed a 122% incremental
improvement in GCS score, compared to patients in Group B (SOC Only)
1SOC: body cooling by physical methods (e.g. cold water immersion, cold water mist, ice packs application) and supportive measures
EHS Study Results Continued
16
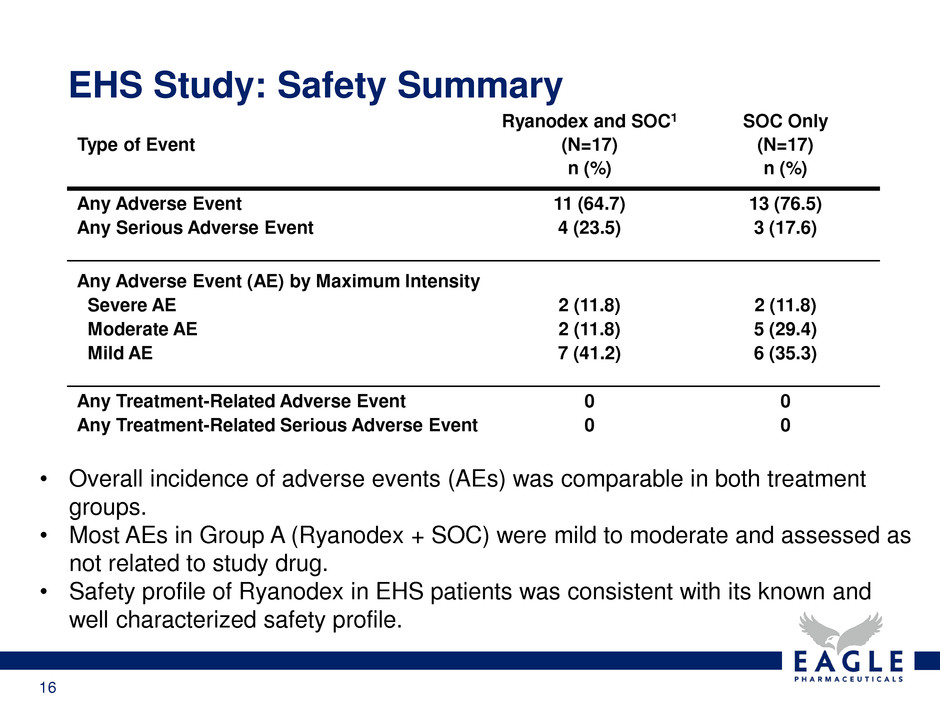
EHS Study: Safety Summary
• Overall incidence of adverse events (AEs) was comparable in both treatment
groups.
• Most AEs in Group A (Ryanodex + SOC) were mild to moderate and assessed as
not related to study drug.
• Safety profile of Ryanodex in EHS patients was consistent with its known and
well characterized safety profile.
Type of Event
Ryanodex and SOC1
(N=17)
n (%)
SOC Only
(N=17)
n (%)
Any Adverse Event 11 (64.7) 13 (76.5)
Any Serious Adverse Event 4 (23.5) 3 (17.6)
Any Adverse Event (AE) by Maximum Intensity
Severe AE 2 (11.8) 2 (11.8)
Moderate AE 2 (11.8) 5 (29.4)
Mild AE 7 (41.2) 6 (35.3)
Any Treatment-Related Adverse Event 0 0
Any Treatment-Related Serious Adverse Event 0 0

17
• Joint effort with the NIH to explore the potential of Ryanodex to treat
hyperthermia related to Ecstasy and Methamphetamine intoxication
– 125,000 emergency room visits in the US in 2011 due to Ecstasy and
Methamphetamine use1
– Brain hyperthermia is one of the leading causes of death in Ecstasy and
Methamphetamine intoxication
• Preclinical studies to be conducted by NIDA beginning summer 2016
– Well-characterized animal model to be used
• Positive preclinical results would facilitate FDA meeting and potentially
lead to a rapid transition to short-duration pivotal clinical trials
Ryanodex for Ecstasy & Meth Intoxication
1Source Drug Abuse Warning Network

18
• Lilly’s Alimta patent infringement lawsuit win should prevent current
ANDA filers from launching until May 24, 2022
• Eagle plans to file Pemetrexed RTU NDA in late 2016
• Registration batches have been produced
• We believe our patent position may enable us to bring the product to
market as early as Q4 2017
• 30 month stay to expire 1st half of 2019
• $1.1B market opportunity1
Pemetrexed
1 Alimta® (pemetrexed) (Eli Lilly & Co.). Source: Eli Lilly & Co. Q2 2016 earnings for MAT 12 mos ending 6/30/13: (U.S. sales)

19
RTU Bivalirudin Opportunity
• Complete Response Letter received March 2016
• In discussions with FDA regarding human study
1 Angiomax® (bivalirudin) / Angiox® (bivalirudin) (The Medicines Company)

20
Building Pipeline for Long Term Growth
• Ryanodex for the treatment of Ecstasy and Methamphetamine
intoxication
• AMRI Development Program
• Joint development program for several new product candidates
• AMRI to provide drug development and manufacturing once approved
by the FDA
• Eagle responsible clinical trials, regulatory submissions, and
commercial distribution in the U.S.
• New NDA under development targeted to reduce number of
injections of $400M branded product in a growing market
• Undisclosed next project

21
Strategic Cash Deployment
• $127.6 million in cash and receivables as of June 30, 2016
• Focused on prudent deployment of cash on behalf of shareholders
• Ryanodex royalty
• Reduced royalty obligation from 15% to 3%
• $15 million in cash
• Share repurchase
• Board authorized $75 million share buyback program
• Completed over time at market price
22
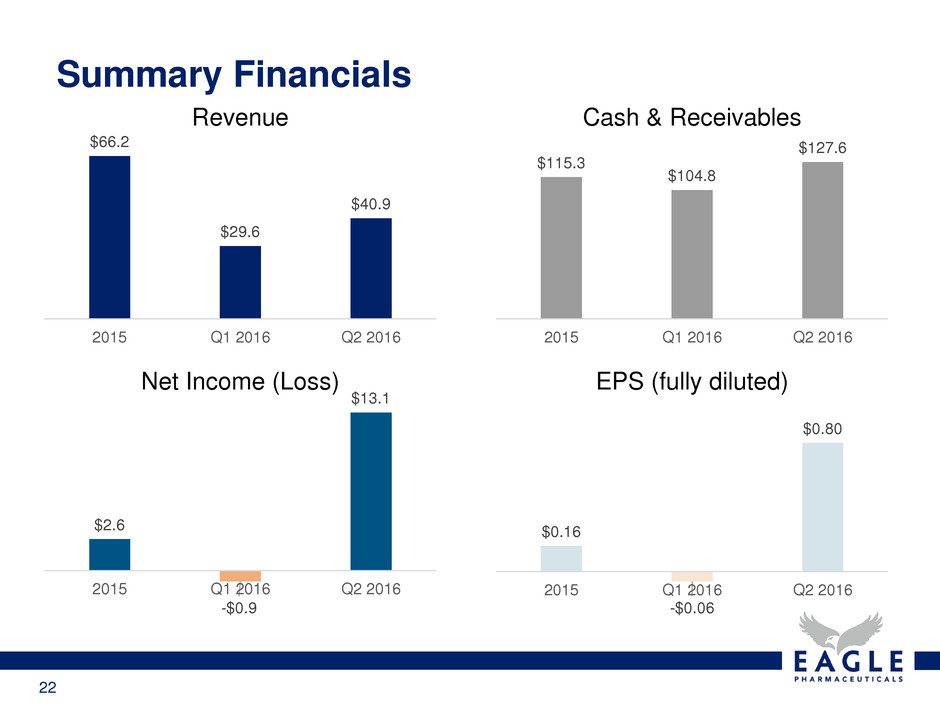
$66.2
$29.6
$40.9
2015 Q1 2016 Q2 2016
Revenue
Summary Financials
$115.3
$104.8
$127.6
2015 Q1 2016 Q2 2016
Cash & Receivables
$2.6
-$0.9
$13.1
2015 Q1 2016 Q2 2016
Net Income (Loss)
$0.16
-$0.06
$0.80
2015 Q1 2016 Q2 2016
EPS (fully diluted)

23
Why Eagle? Why Now?
• Fully commercial specialty pharmaceutical company with 5 in-market products
• Bendeka expected to deliver steady royalty stream driving near term profitability
• 80% market share (and growing) of $750mm market
• 20% royalty (~$125-$150mm+ annually) through 2019
• Next big product: Ryanodex
• Selling in niche market to treat malignant hyperthermia
• Large market opportunity in treating Exertional Heat Stroke
• Expect to file NDA shortly
• Potential to treat brain hyperthermia in ecstasy and methamphetamine intoxication
• 125K ER cases annually in the US
• Exciting additional pipeline programs
• Profitable, growing cash position, no debt
• Confident in future
• Purchased Ryanodex royalty obligation
• Authorized $75mm stock buyback
