Attached files
| file | filename |
|---|---|
| EX-32.1 - EXHIBIT 32.1 - TITAN PHARMACEUTICALS INC | v404713_ex32-1.htm |
| EX-23.1 - EXHIBIT 23.1 - TITAN PHARMACEUTICALS INC | v404713_ex23-1.htm |
| EX-31.1 - EXHIBIT 31.1 - TITAN PHARMACEUTICALS INC | v404713_ex31-1.htm |
| EXCEL - IDEA: XBRL DOCUMENT - TITAN PHARMACEUTICALS INC | Financial_Report.xls |
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
(Mark One)
| x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2014
or
| ¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to .
Commission file number 000-27436
TITAN PHARMACEUTICALS, INC.
(Exact name of registrant as specified in its charter)
| Delaware | 94-3171940 |
|
(State or Other Jurisdiction of Incorporation or Organization) |
(I.R.S. Employer Identification Number) |
|
400 Oyster Point Blvd., Suite 505, South San Francisco, California |
94080 |
| (Address of principal executive offices) | (Zip code) |
Registrant’s telephone number, including area code: (650) 244-4990
Securities registered pursuant to Section 12(b) of the Act: None
Securities registered pursuant to Section 12(g) of the Act:
Common Stock, $0.001 par value
Indicate by check mark if the registrant is a well-known seasoned issuer as defined in Rule 405 of the Securities Act. Yes ¨ No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Exchange Act. Yes ¨ No x
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Exchange Act during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to the filing requirements for the past 90 days. Yes x No ¨
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). x Yes ¨ No
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§ 229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. x
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated filer”, “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act. (Check one):
| Large accelerated filer | ¨ | Accelerated filer | ¨ |
| Non-accelerated filer | ¨ | Smaller Reporting Company | x |
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ¨ No x
The aggregate market value of the 77,845,432 shares of voting and non-voting common equity held by non-affiliates of the registrant based on the closing price on June 30, 2014 was $60.7 million.
As of March 24, 2015, 110,327,707 shares of common stock, $0.001 par value, of the registrant were issued and outstanding.
| 2 |
DOCUMENTS INCORPORATED BY REFERENCE:
NONE
PART I
NOTE REGARDING FORWARD-LOOKING STATEMENTS
Statements in this Annual Report on Form 10-K or in the documents incorporated by reference herein may contain “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933 (the “Securities Act”) and Section 21E of the Securities Exchange Act of 1934 (the “Exchange Act”). Reference is made in particular to the description of our plans and objectives for future operations, assumptions underlying such plans and objectives and other forward-looking terminology such as “may,” “expects,” “believes,” “anticipates,” “intends,” “expects,” “projects,” or similar terms, variations of such terms or the negative of such terms. Forward-looking statements are based on management’s current expectations. Actual results could differ materially from those currently anticipated due to a number of factors, including but not limited to, uncertainties relating to financing and strategic agreements and relationships; difficulties or delays in the regulatory approval process; uncertainties relating to sales, marketing and distribution of our drug candidates that may be successfully developed and approved for commercialization; adverse side effects or inadequate therapeutic efficacy of our drug candidates that could slow or prevent product development or commercialization; dependence on third party suppliers; the uncertainty of protection for our patents and other intellectual property or trade secrets; and competition.
We expressly disclaim any obligation or undertaking to release publicly any updates or revisions to any forward-looking statements contained herein to reflect any change in our expectations or any changes in events, conditions or circumstances on which any such statement is based.
References herein to “we,” “us,” “Titan,” and “our company” refer to Titan Pharmaceuticals, Inc. unless the context otherwise requires.
Probuphine® and ProNeura™ are trademarks of our company. This Annual Report on Form 10-K also includes trade names and trademarks of companies other than Titan.
Item 1. Business
Overview
We are a specialty pharmaceutical company developing proprietary therapeutics for the treatment of serious medical disorders. Our product development programs utilize our proprietary long-term drug delivery platform, ProNeura®, and focus primarily on innovative treatments for select chronic diseases for which steady state delivery of a drug provides an efficacy and/or safety benefit.
Probuphine®, our first product candidate to utilize ProNeura, is being developed for the long term maintenance treatment of opioid dependence and is designed to maintain a stable, around the clock blood level of the medicine buprenorphine in patients for six months following a single treatment. We have licensed the rights to commercialize Probuphine in the U.S. and Canada to Braeburn Pharmaceuticals Sprl (“Braeburn”) and during 2014 we have been supporting Braeburn and a team of experts to implement the program developed in cooperation with the FDA to address the items in the Complete Response letter (“CRL”) issued in April 2013. This includes conducting a double blind, double dummy clinical study of a four implant dose of Probuphine in clinically stable patients who are receiving maintenance treatment with an approved sublingual formulation containing buprenorphine at a daily dose of 8mg or less. This clinical study, which is being funded and managed by Braeburn, completed patient enrollment in November 2014 and study completion is anticipated by the end of the second quarter of 2015 followed by resubmission of the NDA later in the year, with a potential PDUFA date for the Probuphine NDA in the first half of 2016. Pursuant to our license agreement with Braeburn, as amended to date, we are entitled to receive a $15 million milestone payment upon FDA approval of the Probuphine NDA and royalties on net sales of Probuphine ranging in percentage from the mid-teens to the low twenties. The agreement also provides for up to $165 million in sales milestones and $35 million in regulatory milestones and entitles us to royalty rates in the low single digit on sales by Braeburn, if any, of other future products in the addiction market.
| 3 |
We believe that our ProNeura technology has the potential to be used in the treatment of other chronic conditions, such as Parkinson’s disease (PD), where maintaining stable, around the clock blood levels of a dopamine agonist may benefit the patient and improve medical outcomes. We have commenced initial work on an implant formulation with ropinirole, a dopamine agonist approved for the treatment of PD. Our goal is to complete the non-clinical studies required in support of an Investigational New Drug (“IND”) application by early next year and enable commencement of a ‘proof of concept’ clinical study following the potential approval of Probuphine. We are also currently evaluating drugs and disease settings for opportunities to develop our drug delivery technology for other potential treatment applications in situations where conventional treatment is limited by variability in blood drug levels and poor patient compliance.
We operate in only one business segment, the development of pharmaceutical products.
ProNeura Continuous Drug Delivery Technology
Our ProNeura continuous drug delivery system consists of a small, solid rod made from a mixture of ethylene-vinyl acetate (“EVA”) and a drug substance. The resulting product is a solid matrix that is placed subdermally, normally in the inside part of the upper arm in a simple office procedure, and is removed in a similar manner at the end of the treatment period. The drug substance is released continuously through the process of dissolution. This results in a steady rate of release generally similar to intravenous administration. We believe that such long-term, linear release characteristics are desirable by avoiding peak and trough level dosing that may pose problems for many disease settings.
The ProNeura technology was developed to address the need for a simple, practical method to achieve continuous long-term drug delivery, and potentially can provide treatment on an outpatient basis over extended periods of up to 6 – 12 months. We believe that the benefits of this technology have been demonstrated by the clinical results to date with Probuphine. We believe that this technology has the potential to be useful in the treatment of other diseases. Accordingly, we have been evaluating opportunities to develop this drug delivery technology for other potential treatment applications in which conventional treatment is limited by variability in blood drug levels and poor patient compliance and where existing therapeutic compounds have sufficient potency to be effective at low doses. In furtherance of these efforts, during 2012, with the support of a National Institute of Health Small Business Innovation Research grant, we completed a non-clinical study with long-term delivery of ropinirole (Requip TM), a dopamine agonist marketed in the U.S. by GlaxoSmithKline for the treatment of Parkinson’s disease.
Our Product Pipeline
Probuphine
We are developing Probuphine for the maintenance treatment of opioid dependence. Probuphine utilizes ProNeura, our novel, proprietary, long-term drug delivery technology. Upon subdermal insertion in a patient, Probuphine is designed to release medication continuously and maintain a stable, around the clock blood level of the drug buprenorphine, an approved agent for the treatment of opioid dependence. If approved, Probuphine is expected to provide six months of medication following a single treatment. In addition to the ongoing Phase 3 clinical study, Probuphine has been evaluated in the following Phase 3 clinical studies:
| · | Two six-month, double-blind, placebo-controlled safety and efficacy trials; one of which included an open label, active control (Suboxone). In both studies, Probuphine demonstrated superiority to placebo implants, and in the second study, established non-inferiority in comparison to Suboxone; |
| · | Two six-month, open-label re-treatment safety trials; and |
| · | A pharmacokinetic (relative bioavailability) safety study. |
| 4 |
The goal of any therapy for an addictive disorder is to reduce the use of the addictive substance over time and to engage the patient in treatment long enough for therapeutic gains to be consolidated. In a clinical study, the effectiveness of a treatment for opioid dependence is primarily evaluated by testing a patient’s urine samples for the presence of illicit opioids over the treatment period. In both placebo-controlled Phase 3 studies of Probuphine, every participant was required to provide urine samples three times a week, essentially on alternate days. Any missed sample was considered a positive result (i.e. urine testing positive for illicit opioid). In these studies, the primary effectiveness of the treatment with Probuphine (i.e. the primary endpoint) was established by comparing the negative urine results (i.e. urine testing negative for illicit opioid) between the Probuphine and placebo arms using a statistical technique, specifically ‘the cumulative distribution function of negative urines’, which basically performs a comparative analysis on the relative proportions of negative urines between treatment groups over the time period of treatment. The patients in the Probuphine arm showed statistically significant difference in the negative urines as compared to the placebo arm in both studies, i.e. the Probuphine patients had statistically more negative results than the placebo arm, demonstrating that the treatment with Probuphine was successful in reducing their usage of illicit opioids as compared to the treatment with placebo. These favorable results for Probuphine were also confirmed by a significant difference over the placebo arm in other secondary measures such as retention in treatment, withdrawal symptoms and craving for opioids, all of which are monitored by clinicians to see if a treatment is providing benefit to the patients.
Results for the first double-blind, placebo-controlled safety and efficacy study have been published in the Journal of the American Medical Association (JAMA, October 2010) and results of the follow-on randomized three arm study with Probuphine, placebo and sublingual treatment have been published in the journal Addiction (Addiction, September 2013).
Patients who completed the controlled studies were eligible for enrollment in six-month re-treatment studies, which provided data on up to one full year of treatment. The pharmacokinetic safety study has provided important data on the level of buprenorphine in the blood during the treatment period and gives a good profile of the safety of Probuphine. Data from all of these studies was presented at several scientific meetings, including the International Society of Addiction Medicine Annual Meetings in November 2008 and September 2011, the American Society of Addiction Medicine Annual Meetings in May 2009 and 2012, American Society of Addiction Medicine Education Forum in October 2011, and the American College of Neuropharmacology in November 2009 and 2012.
These studies are part of a registration directed program intended to obtain marketing approval of Probuphine for the treatment of opioid dependence in the U.S. and other countries. We met with the FDA in October 2011 for a pre-NDA meeting and reviewed the clinical development program as well as the chemistry, manufacturing and controls (“CMC”) aspects of the NDA. Based on this interaction we completed the requirements for an NDA and subsequently prepared and submitted the NDA in October 2012. ”) On March 21, 2013, the Psychopharmacologic Drugs Advisory Committee (PDAC) of the FDA met to review and discuss the briefing material provided by the FDA and Titan, and at the end of the meeting voted in favor of approval of Probuphine. On April 30, 2013, the FDA issued a complete response letter to our NDA stating that it cannot approve the application in its present form and outlining the FDA’s request for additional clinical data demonstrating adequate clinical benefit to patients from this treatment, data from human factors testing of the training program for insertion and removal of the implant, as well as recommendations regarding product labeling, REMS and non-clinical safety data.
Our efforts since receipt of the CRL have focused on supporting Braeburn and a team of expert clinical and regulatory advisors to establish a path forward that addresses the questions in the CRL and is acceptable to the FDA, and leads to the potential resubmission of the NDA with the additional information. Following a meeting with the FDA on November 19, 2013 and subsequent communications, there is general agreement on the next steps, which includes conducting an additional clinical study. This study is a randomized, double blind, double dummy design of approximately 180 patients enrolled into two parallel treatment arms. The study population is clinically stable patients who are receiving maintenance treatment with an approved sublingual formulation containing buprenorphine at a daily dose of 8mg or less. Patients will be randomized to receive either four Probuphine implants, or to continue the daily sublingual buprenorphine therapy. The patients are expected to be treated for six months, and the primary analysis will be a non-inferiority comparison of responders in the two arms. Patient enrollment in this study was completed in November 2014 and study completion is anticipated by the end of the second quarter of 2015 followed by resubmission of the NDA later in the year and a possible PDUFA date in the first half of 2016.
| 5 |
Pursuant to the license agreement with Braeburn, as amended to date, we are entitled to receive a $15 million milestone payment upon FDA approval of the Probuphine NDA and royalties on net sales of Probuphine ranging in percentage from the mid-teens to the low twenties. The agreement also provides for up to $165 million in sales milestones and $35 million in regulatory milestones and entitles us to low single digit royalties on sales by Braeburn, if any, of other future products in the addiction market.
ProNeura-Ropinirole for Parkinson’s Disease
Parkinson’s disease (PD) is a disease of the central nervous system characterized by the loss of dopaminergic neurons, which leads to increasing activity in the brain region that influences movement and motor function. According to the Parkinson’s Disease Foundation, more than one million people in the U.S. suffer from PD, and this number is projected to double by 2030. Early stage PD patients are treated with daily doses of drugs designed to replace dopamine in the brain. However, these therapeutics typically lose their benefits after several years of chronic treatment, and trigger serious side effect. About one-third of the treated patients develop motor response fluctuations and/or drug-induced dyskinesias within only 3 − 5 years of treatment, and these symptoms are present in almost all patients after 10 − 12 years. Clinical and nonclinical research indicates that these motor side effects arise from the pulsatile dopaminergic stimulation resulting from current oral treatment. Continuous dopaminergic stimulation (CDS) by subcutaneous infusion has been shown to palliate these motor complications, as well as to delay or prevent the onset of dyskinesias. We believe our ProNeura drug delivery technology provides a clinically-validated platform to safely and conveniently provide CDS for several months from a single treatment. Further, the subdermal placement of these implants eliminates many of the device-related complications associated with existing treatment modalities.
During the past couple of years we conducted preliminary experiments to investigate the use of our ProNeura technology to continuously deliver dopamine agonists for the treatment of PD. We have conducted a non-clinical study in an MPTP Parkinsonian monkey model and demonstrated that a sustained non-fluctuating plasma level of ropinirole could be delivered safely for several months following implantation and could control PD symptoms without triggering dyskenesias in severely lesioned primates. We have committed resources to this product development program to enable the following during 2015 and 2016:
| • | Optimize the implant formulation of ropinirole |
| • | Develop the non-clinical study plan to support Investigational New Drug (IND) application |
| • | Design a proof of concept clinical study |
| • | Conduct a pre-IND meeting with the FDA |
| • | Complete the non-clinical studies to enable timely submission of IND and commence ‘proof of concept’ clinical study |
Our goal is to complete the non-clinical studies necessary to enable timely submission of the IND and commence a ‘proof of concept’ clinical study in 2016 following the potential approval of Probuphine.
Fanapt® (iloperidone)
Fanapt (iloperidone) is an atypical antipsychotic approved in 2009 by the FDA for the treatment of schizophrenia and in 2014 was marketed by Novartis in the U.S. Under a sublicense agreement with Novartis, we are entitled to a royalty of 8 – 10% of net sales, based on a U.S. patent that we licensed from Sanofi-Aventis. The U.S. patent expires in October 2016 (excluding a six-month pediatric extension). On December 31, 2014 Vanda Pharmaceuticals, Inc. (“Vanda”) acquired the rights to Fanapt for the U.S. and Canada from Novartis and is now marketing Fanapt in the U.S. Vanda already owned the development and commercialization rights to the oral and depot formulations of this product for the rest of the world, and by acquiring the U.S. and Canadian rights from Novartis, effectively replaces Novartis in the sublicense agreement with Titan. Patent coverage on the compound has now expired in the significant markets outside of the U.S. and no patent term extensions are possible since the product was not approved in these countries prior to patent expiration, and hence we do not expect any royalties on any future sales in such markets.
| 6 |
We have entered into several agreements with Deerfield, which entitle Deerfield to most of the future royalty revenues related to Fanapt in exchange for cash and debt considerations, the proceeds of which have been used to advance the development of Probuphine and for general corporate purposes. We have retained a portion of the royalty revenue from net sales of Fanapt in excess of specified annual threshold levels; however, based on sales levels to date, it is unlikely that we will ever receive any revenue from Fanapt. We do not incur any ongoing expenses associated with this product.
License Agreements
In December 2012, we entered into a license agreement (the “Agreement”) with Braeburn pursuant to which we granted Braeburn an exclusive right and license to commercialize Probuphine in the United States of America and its territories, including Puerto Rico, and Canada (the “Territory”). Under the Agreement, Braeburn made a non-refundable up-front license fee payment of $15.75 million and agreed to pay us tiered royalties on a percentage of net sales of Probuphine ranging from the mid-teens to the low twenties. Additionally, the Agreement provided for us to receive $45 million upon FDA approval of the NDA for Probuphine and at such time ownership of the NDA will transfer to Braeburn, as well as up to an additional $130 million upon the achievement of specified sales milestones and up to $35 million in regulatory milestones. We will retain all of the rights to Probuphine outside the Territory. Unless earlier terminated, the Agreement will expire on the later of (i) the 15 th anniversary of the date of product launch in the Territory or (ii) the expiration of the last to expire patent in the Territory covered by the Agreement (the “Term”). Either party may terminate the Agreement prior to the expiration of the Term in the event of a material breach by the other party that remains uncured or in the event of the other party’s bankruptcy. We may terminate the Agreement if, for reasons other than force majeure, regulatory, safety, manufacturing or product quality issues, Braeburn discontinues commercial sale of the product and fails to resume sales within 30 days following notice or in the event Braeburn or any of its affiliates or sublicensees commences any legal proceeding seeking to challenge or dispute the validity or ownership of the licensed patents. Braeburn may terminate the Agreement in the event that Braeburn, notwithstanding good faith efforts to do so, is unable to enter into an agreement for the supply of EVA or if such a supply agreement is terminated by Braeburn due to a material breach by the supplier or the supplier fails to provide EVA to Braeburn for a period of at least three months. Braeburn may also terminate the Agreement (i) on a country by country basis upon six months’ notice following the occurrence of any “significant competition” in such country, as such term is defined in the Agreement; (ii) immediately upon notice if Braeburn determines in good faith that it is inadvisable to continue commercialization as a result of any actual or perceived safety issues.
In May 2013, we entered into an amendment to the Agreement (the “Amendment”) primarily to modify certain of the termination provisions of the Agreement. The Amendment gives Braeburn the right to terminate the Agreement in the event that (A) after May 28, 2013, based on written or oral communications from or with the FDA, Braeburn reasonably determines either that the FDA will require significant development to be performed before approval of the Probuphine TM NDA can be given, such as, but not limited to, one or more additional controlled clinical studies with a clinical efficacy endpoint, or substantial post-approval commitments that may materially impact the products financial returns or that the FDA will require one or more changes in the proposed label, which change(s) Braeburn reasonably determines will materially reduce the authorized prescribed patient base, or (B) the NDA has not been approved by the FDA on or before June 30, 2014. The Amendment also provides that we will share in legal and consulting expenses in excess of a specified amount prior to approval of the NDA.
In July 2013, we entered into a second amendment to the Agreement (the “Second Amendment”) primarily to establish and provide the parameters for a committee comprised of representatives of Titan and Braeburn responsible for and with the authority to make all decisions regarding the development and implementation of a strategic plan to seek approval from the FDA of Probuphine® for subdermal use in the maintenance treatment of adult patients with opioid dependence, including development of the strategy for all written and oral communications with the FDA. The Second Amendment also makes Braeburn the primary contact for FDA communications regarding the Probuphine NDA.
In November 2013, we entered into a stock purchase agreement pursuant to which Braeburn made a $5 million equity investment in our company and a third amendment to the Agreement (the “Third Amendment”) primarily to modify the amount and timing of the approval and sales milestone payments payable under the Agreement. Under the Third Amendment, we are entitled to receive a $15 million payment upon FDA approval of the NDA and royalties on net sales of Probuphine ranging in percentage from the mid-teens to the low twenties. The agreement also provides for up to $165 million in sales milestones and $35 in regulatory milestones. In addition, we are entitled to receive royalties on a percentage of sales in the low single digit by Braeburn, if any, of other continuous delivery treatments for opioid dependence as defined in the Third Amendment and can elect to receive low single digit royalties on sales by Braeburn, if any, of other products in the addiction market in exchange for a similar reduction in our royalties on Probuphine.
| 7 |
In January 1997, we acquired an exclusive worldwide license under U.S. and foreign patents and patent applications relating to the use of iloperidone for the treatment of psychiatric and psychotic disorders and analgesia from Sanofi-Aventis SA (“Sanofi-Aventis”) (formerly Hoechst Marion Roussel, Inc.). The Sanofi-Aventis agreement provides for the payment of royalties on future net sales. In November 1997, we granted a worldwide sublicense, exclusive of Japan, to Novartis under which Novartis continued, at its expense, all further development of iloperidone. In April 2001, that sublicense was extended to include Japan. Under this agreement, Novartis agreed to pay Titan a royalty on future net sales of the product equal to 8% of annual worldwide net sales up to $200 million and 10% of annual worldwide net sales above $200 million, in addition to royalty payments owed by us to Sanofi-Aventis. In June 2004, Novartis granted Vanda the worldwide rights to develop and commercialize iloperidone. In October 2009, Vanda and Novartis amended and restated their sub-license agreement whereby Novartis acquired the U.S. and Canadian rights to commercialize Fanapt, the oral formulation of iloperidone approved in the U.S. Novartis also acquired the U.S. and Canadian development and commercialization rights to the depot formulation previously under development by Vanda and retained the right of first negotiation to co-market Fanapt and the depot formulation in the rest of the world. On December 31, 2014, Vanda, via a settlement transaction, reacquired from Novartis its rights and obligations relating to Fanapt in the U.S. and Canada. All of our rights and economic interests in iloperidone, including royalties on sales, remained essentially unchanged under these agreements and, as previously stated, we have entered into several agreements with Deerfield, which entitle Deerfield to the future royalty revenues related to Fanapt in exchange for cash and debt considerations.
Intellectual Property
Our goal is to obtain, maintain and enforce patent protection for our product candidates, formulations, processes, methods and any other proprietary technologies, preserve our trade secrets, and operate without infringing on the proprietary rights of other parties, both in the United States and in other countries. Our policy is to actively seek to obtain, where appropriate, the broadest intellectual property protection possible for our current product candidates and any future product candidates, proprietary information and proprietary technology through a combination of contractual arrangements and patents, both in the United States and abroad. However, patent protection may not afford us with complete protection against competitors who seek to circumvent our patents.
We also depend upon the skills, knowledge, experience and know-how of our management and research and development personnel, as well as that of our advisors, consultants and other contractors. To help protect our proprietary know-how, which is not patentable, and for inventions for which patents may be difficult to enforce, we currently rely and will in the future rely on trade secret protection and confidentiality agreements to protect our interests. To this end, we require all of our employees, consultants, advisors and other contractors to enter into confidentiality agreements that prohibit the disclosure of confidential information and, where applicable, require disclosure and assignment to us of the ideas, developments, discoveries and inventions important to our business.
In June 2010, the United States Patent and Trademark Office (“USPTO”) issued a patent covering methods of using Probuphine for the treatment of opiate addiction. Titan is the owner of this patent which claims a method for treating opiate addiction with a subcutaneously implanted device comprising buprenorphine and EVA, a biocompatible copolymer that releases buprenorphine continuously for extended periods of time. This patent will expire in June 2024. Four U.S. continuation applications are currently pending which incorporate the use of buprenorphine with the continuous delivery technology. These patent applications include claims related to Probuphine for the potential treatment of opioid addiction and chronic pain. Related patents covering use of Probuphine with the continuous delivery technology for the treatment of opiate addiction have also issued in Australia, Canada, India, Japan, Mexico and New Zealand. Further prosecution of Probuphine applications is currently proceeding at the USPTO and corresponding agencies in Europe, Canada, India and Hong Kong. Patents covering certain dopamine agonist implants have already been issued or allowed in the United States, Europe, Japan, Australia, Canada, South Korea, Mexico, New Zealand, South Africa, and Hong Kong, while prosecution of the patent application continues in the United States, Israel, India, Japan, and China.
| 8 |
We have filed additional patent applications for a heterogenous implant designed with some unique properties that may provide benefits to the structural integrity of the implants and potentially enhance drug delivery.
We hold a license from Sanofi-Aventis under certain issued U.S. patents and certain issued foreign patents relating to iloperidone and its methods of use in the treatment of psychiatric disorders, psychotic disorders and analgesia. The term of the U.S. patent that covers certain aspects of our iloperidone product expires in October 2016, excluding a six month extension possible if an approval of pediatric indication is obtained.
Future court decisions or changes in patent law might materially affect the patents or patent applications, including, but not limited to, their expiration dates.
Competition
The pharmaceutical and biotechnology industries are characterized by rapidly evolving technology and intense competition. Many companies of all sizes, including major pharmaceutical companies and specialized biotechnology companies, are engaged in the development and commercialization of therapeutic agents designed for the treatment of the same diseases and disorders that we target. Many of our competitors have substantially greater financial and other resources, larger research and development staff and more experience in the regulatory approval process. Moreover, potential competitors have or may have patents or other rights that conflict with patents covering our technologies.
With respect to Probuphine, there are no six-month implant formulations of buprenorphine on the market or in development, and the primary competition will be Indivior, PLC (formerly the Pharmaceutical business of Reckitt Benckiser Group, PLC (“Reckitt”) which markets globally a sublingual buprenorphine product (tablet and film formulations) for the treatment of opioid dependence. This product (Subutex®, Suboxone®), which is administered daily, holds the dominant market share of global sales and it will compete with our six-month implantable product for the maintenance treatment of opioid dependence. Additionally, two proprietary daily dosed formulations have been approved by the FDA in the past two years; the first is a sublingual tablet called Zubsolv marketed by Orexo and the second is a buccal patch called Bunavail marketed by Bio Delivery Sciences International, which, if successful in capturing market share, would compete with Probuphine. Also, during 2013 and 2014, several generic sublingual tablet formulations of buprenorphine similar to Suboxone and Subutex were approved by the FDA which are expected to compete in the opioid addiction treatment market. Other forms of buprenorphine are also in development by other companies, including intramuscular and intradermal one week and one month depot injections which, if approved, will also compete with our product. Braeburn has licensed rights to certain of such potential products and Titan is entitled to a low single digit royalty on net sales if these are commercialized. The one-month depot formulations of buprenorphine are in mid-stage clinical development for the treatment of opioid dependence, and are likely to take another 2-3 years prior to approval In 2010, Alkermes, Inc. received FDA approval to market Vivitrol®, a one-month depot injection of naltrexone as a maintenance treatment for opioid dependent patients who have successfully achieved abstinence.
With respect to our potential ProNeura ropinirole product for Parkinson’s disease, the competition is primarily from numerous daily dosed dopamine agonist treatments currently in use that provide symptom relief from disease related immobility, and the complications associated with long-term levodopa therapy (e.g. dyskinesias, tolerance). Approved products in the U.S. in addition to Requip XL TM , which is marketed by GlaxoSmithKline, include Apokyn® (US WorldMeds LLC), Parlodel® (Novartis Pharmaceuticals Inc.), Mirapex ER® (Boehringer Ingelheim Pharmaceuticals Inc.) and Neupro® (UCB Inc.). There is a strong need for products delivering continuous, stable, long term delivery of dopamine and dopamine agonists and, recently, the U.S. FDA approved a product called Duopa™ which is the first and only treatment delivered via catheters directly into the duodenum which is capable of providing 16 continuous hours of carbidopa and levodopa for treatment of motor fluctuations in advanced Parkinson's disease. Duopa is marketed globally by the company Abbvie. Also, we are aware of products in development that are capable of short to medium term subcutaneous and subdermal delivery of levodopa/carbidopa using pumps, however these are still in mid stage clinical development.
| 9 |
Manufacturing
The manufacturing of Probuphine has primarily been conducted at DPT Laboratories, Inc., or DPT, and we have expanded the manufacturing facility at this contract manufacturer to establish commercial scale capability to support the future market launch of Probuphine and ongoing demand following potential approval by the FDA. To date, we have been operating with DPT under an arrangement pursuant to which batches of product needed for validation studies, stability testing or clinical trial purposes are acquired pursuant to purchase orders on a time and product cost basis. We have entered into a commercial manufacturing agreement with DPT that will govern the terms of the production and supply of Probuphine at such time, if ever, as the product is launched commercially. We anticipate that at or prior to such time, such agreement will be assigned to Braeburn as licensee or a replacement agreement entered into between Braeburn and DPT.
To date, we have obtained the supply of bupenorphine from Teva Pharmaceuticals, Inc., or Teva, under an arrangement similar to the one with DPT. We have entered into a commercial supply agreement with Teva; however, we anticipate that at or prior to such time if ever, as the product is launched commercially, such agreement will be assigned to Braeburn as licensee or a replacement agreement entered into between Braeburn and Teva.
Sales and Marketing
We do not currently have and do not intend to establish any sales and marketing capability. As licensee, Braeburn will have sole responsibility for sales and marketing of Probuphine within the United States and Canada. We intend to seek comparable partnering arrangements for Probuphine outside the Territory, as well as for any additional products we may successfully develop based on our ProNeura technology.
Government Regulation and Product Approval
Government authorities in the United States, at the federal, state and local level, and other countries extensively regulate, among other things, the research, development, testing, manufacture, quality control, approval, labeling, packaging, storage, record-keeping, promotion, advertising, distribution, post-approval monitoring and reporting, marketing and export and import of products such as those we are developing.
FDA Approval Process
In the United States, pharmaceutical products are subject to extensive regulation by the FDA. The Federal Food, Drug, and Cosmetic Act, or the FDC Act, and other federal and state statutes and regulations, govern, among other things, the research, development, testing, manufacture, storage, recordkeeping, approval, labeling, promotion and marketing, distribution, post-approval monitoring and reporting, sampling, and import and export of pharmaceutical products. Failure to comply with applicable U.S. requirements may subject a company to a variety of administrative or judicial sanctions, such as FDA refusal to approve pending new drug applications, or NDAs, warning letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, civil penalties, and criminal prosecution.
Pharmaceutical product development in the U.S. typically involves preclinical laboratory and animal tests, the submission to the FDA of either a notice of claimed investigational exemption or an investigational new drug application, or IND, which must become effective before clinical testing may commence, and adequate and well-controlled clinical trials to establish the safety and effectiveness of the drug for each indication for which FDA approval is sought. Satisfaction of FDA pre-market approval requirements typically takes many years and the actual time required may vary substantially based upon the type, complexity, and novelty of the product or disease.
Preclinical tests include laboratory evaluation of product chemistry, formulation, and toxicity, as well as animal trials to assess the characteristics and potential safety and efficacy of the product. The conduct of the preclinical tests must comply with federal regulations and requirements, including good laboratory practices. The results of preclinical testing are submitted to the FDA as part of an IND along with other information, including information about product chemistry, manufacturing and controls, and a proposed clinical trial protocol. Long term preclinical tests, such as animal tests of reproductive toxicity and carcinogenicity, may continue after the IND is submitted.
| 10 |
Clinical trials involve the administration of the investigational new drug to healthy volunteers or patients under the supervision of a qualified investigator. Clinical trials must be conducted: (i) in compliance with federal regulations; (ii) in compliance with good clinical practice, or GCP, an international standard meant to protect the rights and health of patients and to define the roles of clinical trial sponsors, administrators, and monitors; as well as (iii) under protocols detailing the objectives of the trial, the parameters to be used in monitoring safety, and the effectiveness criteria to be evaluated. Each protocol involving testing on U.S. patients and subsequent protocol amendments must be submitted to the FDA as part of the IND.
The FDA may order the temporary, or permanent, discontinuation of a clinical trial at any time, or impose other sanctions if it believes that the clinical trial either is not being conducted in accordance with FDA requirements or presents an unacceptable risk to the clinical trial patients. The study protocol and informed consent information for patients in clinical trials must also be submitted to an institutional review board, or IRB, for approval. An IRB may also require the clinical trial at the site to be halted, either temporarily or permanently, for failure to comply with the IRB’s requirements, or may impose other conditions.
Clinical trials to support NDAs for marketing approval are typically conducted in three sequential phases, but the phases may overlap. In Phase 1, the initial introduction of the drug into healthy human subjects or patients, the drug is tested to assess metabolism, pharmacokinetics, pharmacological actions, side effects associated with increasing doses, and, if possible, early evidence on effectiveness. Phase 2 usually involves trials in a limited patient population to determine the effectiveness of the drug for a particular indication, dosage tolerance, and optimum dosage, and to identify common adverse effects and safety risks. If a compound demonstrates evidence of effectiveness and an acceptable safety profile in Phase 2 evaluations, Phase 3 trials are undertaken to obtain the additional information about clinical efficacy and safety in a larger number of patients, typically at geographically dispersed clinical trial sites, to permit FDA to evaluate the overall benefit-risk relationship of the drug and to provide adequate information for the labeling of the drug.
After completion of the required clinical testing, an NDA is prepared and submitted to the FDA. FDA approval of the NDA is required before marketing of the product may begin in the U.S. The NDA must include the results of all preclinical, clinical, and other testing and a compilation of data relating to the product’s pharmacology, chemistry, manufacture, and controls. The cost of preparing and submitting an NDA is substantial.
Once the submission is accepted for filing, the FDA begins an in-depth review. Priority review can be applied to drugs that the FDA determines offer major advances in treatment, or provide a treatment where no adequate therapy exists. The FDA may refer applications for novel drug products, or drug products which present difficult questions of safety or efficacy, to an advisory committee — typically a panel that includes clinicians and other experts — for review, evaluation, and a recommendation as to whether the application should be approved. The FDA is not bound by the recommendation of an advisory committee, but it generally follows such recommendations. Before approving an NDA, the FDA will typically inspect one, or more, clinical sites to assure compliance with GCP. Additionally, the FDA will inspect the facility or the facilities at which the drug is manufactured. FDA will not approve the product unless compliance with current good manufacturing practices, or GMP — a quality system regulating manufacturing — is satisfactory and the NDA contains data that provide substantial evidence that the drug is safe and effective in the indication studied.
After the FDA evaluates the NDA and the manufacturing facilities, it issues either an approval letter or a complete response letter. A complete response letter generally outlines the deficiencies in the submission and may require substantial additional testing, or information, in order for the FDA to reconsider the application. If, or when, those deficiencies have been addressed to the FDA’s satisfaction in a resubmission of the NDA, the FDA will issue an approval letter. An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications. As a condition of NDA approval, the FDA may require a risk evaluation and mitigation strategy, or REMS, to help ensure that the benefits of the drug outweigh the potential risks. REMS can include medication guides, communication plans for healthcare professionals, and elements to assure safe use, or ETASU. ETASU can include, but are not limited to, special training or certification for prescribing or dispensing, dispensing only under certain circumstances, special monitoring, and the use of patient registries. The requirement for a REMS can materially affect the potential market and profitability of the drug. Moreover, product approval may require substantial post-approval testing and surveillance to monitor the drug’s safety or efficacy. Once granted, product approvals may be withdrawn if compliance with regulatory standards is not maintained or problems are identified following initial marketing.
| 11 |
The Hatch-Waxman Act
In seeking approval for a drug through an NDA, applicants are required to list with the FDA each patent whose claims cover the applicant’s product. Upon approval of a drug, each of the patents listed in the application for the drug is then published in the FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations, commonly known as the Orange Book. Drugs listed in the Orange Book can, in turn, be cited by potential competitors in support of approval of an abbreviated new drug application, or ANDA. An ANDA provides for marketing of a drug product that has the same active ingredients in the same strengths and dosage form as the listed drug and has been shown through bioequivalence testing to be therapeutically equivalent to the listed drug. Other than the requirement for bioequivalence testing, ANDA applicants are not required to conduct, or submit results of, pre-clinical or clinical tests to prove the safety or effectiveness of their drug product. Drugs approved in this way are commonly referred to as “generic equivalents” to the listed drug, and can often be substituted by pharmacists under prescriptions written for the original listed drug.
The ANDA applicant is required to certify to the FDA concerning any patents listed for the approved product in the FDA’s Orange Book. Specifically, the applicant must certify that: (i) the required patent information has not been filed; (ii) the listed patent has expired; (iii) the listed patent has not expired, but will expire on a particular date and approval is sought after patent expiration; or (iv) the listed patent is invalid or will not be infringed by the new product. A certification that the new product will not infringe the already approved product’s listed patents, or that such patents are invalid, is called a Paragraph IV certification. If the applicant does not challenge the listed patents, the ANDA application will not be approved until all the listed patents claiming the referenced product have expired. The ANDA application also will not be approved until any non-patent exclusivity listed in the Orange Book for the referenced product has expired. Federal law provides a period of five years following approval of a drug containing no previously approved active ingredients during which ANDAs for generic versions of those drugs cannot be submitted, unless the submission contains a Paragraph IV challenge to a listed patent — in which case the submission may be made four years following the original product approval. Federal law provides for a period of three years of exclusivity during which the FDA cannot grant effective approval of an ANDA based on the approval of a listed drug that contains previously approved active ingredients but is approved in a new dosage form, route of administration or combination, or for a new use; the approval of which was required to be supported by new clinical trials conducted by, or for, the applicant.
Section 505(b)(2) New Drug Applications
Most drug products obtain FDA marketing approval pursuant to an NDA or an ANDA. A third alternative is a special type of NDA, commonly referred to as a Section 505(b)(2) NDA, that enables the applicant to rely, in part, on the FDA’s previous approval of a similar product, or published literature, in support of its application. Our NDA for Probuphine was submitted under Section 505(b)(2) and we anticipate that we will pursue this pathway for any additional therapeutic products we may develop based on our ProNeura technology. Section 505(b)(2) permits the filing of an NDA where at least some of the information required for approval comes from studies not conducted by, or for, the applicant and for which the applicant has not obtained a right of reference. If the 505(b)(2) applicant can establish that reliance on the FDA’s previous approval is scientifically appropriate, it may eliminate the need to conduct certain preclinical or clinical studies of the new product. The FDA may also require companies to perform additional studies or measurements to support the change from the approved product. The FDA may then approve the new product candidate for all, or some, of the label indications for which the referenced product has been approved, as well as for any new indication sought by the Section 505(b)(2) applicant.
| 12 |
Advertising and Promotion
Once an NDA is approved, a product will be subject to certain post-approval requirements. For instance, the FDA closely regulates the post-approval marketing and promotion of drugs, including standards and regulations for direct-to-consumer advertising, off-label promotion, industry-sponsored scientific and educational activities and promotional activities involving the internet.
Drugs may be marketed only for the approved indications and in accordance with the provisions of the approved labeling. Changes to some of the conditions established in an approved application, including changes in indications, labeling, or manufacturing processes or facilities, require submission and FDA approval of a new NDA or NDA supplement before the change can be implemented. An NDA supplement for a new indication typically requires clinical data similar to that in the original application, and the FDA uses the same procedures and actions in reviewing NDA supplements as it does in reviewing NDAs.
Adverse Event Reporting and GMP Compliance
Adverse event reporting and submission of periodic reports is required following FDA approval of an NDA. The FDA also may require post-marketing testing, known as Phase 4 testing, risk minimization action plans, and surveillance to monitor the effects of an approved product, or the FDA may place conditions on an approval that could restrict the distribution or use of the product. In addition, quality-control, drug manufacture, packaging, and labeling procedures must continue to conform to current good manufacturing practices, or cGMPs, after approval. Drug manufacturers and certain of their subcontractors are required to register their establishments with FDA and certain state agencies. Registration with the FDA subjects entities to periodic unannounced inspections by the FDA, during which the agency inspects manufacturing facilities to assess compliance with cGMPs. Accordingly, manufacturers must continue to expend time, money, and effort in the areas of production and quality-control to maintain compliance with cGMPs. Regulatory authorities may withdraw product approvals or request product recalls if a company fails to comply with regulatory standards, if it encounters problems following initial marketing, or if previously unrecognized problems are subsequently discovered.
Pediatric Information
Under the Pediatric Research Equity Act, or PREA, NDAs or supplements to NDAs must contain data to assess the safety and effectiveness of the drug for the claimed indications in all relevant pediatric subpopulations and to support dosing and administration for each pediatric subpopulation for which the drug is safe and effective. The FDA may grant full or partial waivers, or deferrals, for submission of data. Unless otherwise required by regulation, PREA does not apply to any drug for an indication for which orphan designation has been granted.
The Best Pharmaceuticals for Children Act, or BPCA, provides NDA holders a six-month extension of any exclusivity — patent or non-patent — for a drug if certain conditions are met. Conditions for exclusivity include the FDA’s determination that information relating to the use of a new drug in the pediatric population may produce health benefits in that population, the FDA making a written request for pediatric studies, and the applicant agreeing to perform, and reporting on, the requested studies within the statutory timeframe. Applications under the BPCA are treated as priority applications, with all of the benefits that designation confers.
Physician Drug Samples
As part of the sales and marketing process, pharmaceutical companies frequently provide samples of approved drugs to physicians. The Prescription Drug Marketing Act, or the PDMA, imposes requirements and limitations upon the provision of drug samples to physicians, as well as prohibits states from licensing distributors of prescription drugs unless the state licensing program meets certain federal guidelines that include minimum standards for storage, handling, and record keeping. In addition, the PDMA sets forth civil and criminal penalties for violations.
| 13 |
Controlled Substances
Manufacturers of controlled substances, including buprenorphine, are also subject to the licensing, quota, and regulatory requirements of the Controlled Substances Act. Failure to comply with the Controlled Substances Act and the regulations promulgated thereunder could subject companies to loss or suspension of those licenses and to civil or criminal penalties.
Anti-Kickback, False Claims Laws & The Prescription Drug Marketing Act
In addition to FDA restrictions on marketing of pharmaceutical products, several other types of state and federal laws have been applied to restrict certain marketing practices in the pharmaceutical industry in recent years. These laws include anti-kickback statutes and false claims statutes. The federal healthcare program anti-kickback statute prohibits, among other things, knowingly and willfully offering, paying, soliciting or receiving remuneration to induce; or in return for; purchasing, leasing, ordering or arranging for the purchase, lease or order of any healthcare item or service reimbursable under Medicare, Medicaid, or other federally financed healthcare programs. This statute has been interpreted to apply to arrangements between pharmaceutical manufacturers on the one hand and prescribers, purchasers, and formulary managers on the other. Violations of the anti-kickback statute are punishable by imprisonment, criminal fines, civil monetary penalties, and exclusion from participation in federal healthcare programs. Although there are a number of statutory exemptions and regulatory safe harbors protecting certain common activities from prosecution or other regulatory sanctions, the exemptions and safe harbors are drawn narrowly, and practices that involve remuneration intended to induce prescribing, purchases, or recommendations may be subject to scrutiny if they do not qualify for an exemption or safe harbor.
Federal false claims laws prohibit any person from knowingly presenting, or causing to be presented, a false claim for payment to the federal government, or knowingly making, or causing to be made, a false statement to have a false claim paid. Recently, several pharmaceutical and other healthcare companies have been prosecuted under these laws for allegedly inflating drug prices they report to pricing services, which in turn were used by the government to set Medicare and Medicaid reimbursement rates, and for allegedly providing free product to customers with the expectation that the customers would bill federal programs for the product. In addition, certain marketing practices, including off-label promotion, may also violate false claims laws. The majority of states also have statutes or regulations similar to the federal anti-kickback law and false claims laws, which apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply regardless of the payor.
Foreign Regulatory Issues
Sales of pharmaceutical products outside the United States are subject to foreign regulatory requirements that vary widely from country to country. Whether or not FDA approval has been obtained, approval of a product by a comparable regulatory authority of a foreign country must generally be obtained prior to the commencement of marketing in that country. Although the time required to obtain such approval may be longer or shorter than that required for FDA approval, the requirements for FDA approval are among the most detailed in the world and FDA approval generally takes longer than foreign regulatory approvals.
Employees
As of December 31, 2014, we had 13 full-time employees.
| 14 |
Item 1A. Risk Factors
Further delays in the FDA approval process for Probuphine or termination of the license agreement by Braeburn could materially adversely impact our liquidity and financial condition.
While Braeburn has commenced the clinical study and patient enrollment is underway, we cannot predict the timing of commencement or completion of the study. At December 31, 2014, we had cash of approximately $15.5 million, which we believe is sufficient to fund our planned operations into fourth quarter of 2016. Under our license agreement, as amended, Braeburn currently has the technical right to terminate the agreement. If Braeburn were to exercise this right, we would not have sufficient funds available to us to complete the FDA regulatory process and, in the event of ultimate approval, commercialize Probuphine without raising additional capital. If we are unable to complete a debt or equity offering, or otherwise obtain sufficient financing in such event, our business and prospects would be materially adversely impacted. We cannot assure you that the financing we need will be available on acceptable terms.
FDA approval of Probuphine may be denied.
While Titan and Braeburn have agreed in principal with the FDA on a path forward for Probuphine, which along with other steps includes conducting an additional clinical study which is expected to be completed by the middle of 2015, there can be no assurance that the FDA will ultimately approve the NDA. The FDA may deny approval of Probuphine for many reasons, including:
| · | we may be unable to demonstrate to the satisfaction of the FDA that Probuphine is safe and effective for the treatment of opioid dependence in the targeted patient population; |
| · | the FDA may disagree with our interpretation of data from the clinical trial; |
| · | we may be unable to demonstrate that Probuphine’s clinical and other benefits outweigh any safety or other perceived risks; or |
| · | we may not be able to successfully address the other issues raised by the FDA in the CRL. |
If Probuphine fails to receive FDA approval, our business and prospects will be materially adversely impacted.
Even if we obtain FDA approval of Probuphine, we may never obtain approval or commercialize our products outside of the United States, which would limit our ability to realize their full market potential.
In order to market Probuphine outside of the United States, we must establish and comply with numerous and varying regulatory requirements of other countries regarding safety and efficacy. Clinical trials conducted in one country may not be accepted by regulatory authorities in other countries, and regulatory approval in one country does not mean that regulatory approval will be obtained in any other country. Approval procedures vary among countries and can involve additional product testing and validation and additional administrative review periods. Seeking foreign regulatory approvals could result in significant delays, difficulties and costs for us and may require additional pre-clinical studies or clinical trials which would be costly and time consuming. Regulatory requirements can vary widely from country to country and could delay or prevent the introduction of our products in those countries. Satisfying these and other regulatory requirements is costly, time consuming, uncertain and subject to unanticipated delays. In addition, our failure to obtain regulatory approval in any country may delay or have negative effects on the process for regulatory approval in other countries. We do not have any product candidates approved for sale in any jurisdiction, including international markets, and we do not have experience in obtaining regulatory approval in international markets. If we fail to comply with regulatory requirements in international markets or to obtain and maintain required approvals, our target market will be reduced and our ability to realize the full market potential of our products will be harmed.
| 15 |
We have granted an exclusive license to Braeburn for the commercialization of Probuphine in the United States and Canada (the “Territory”). If approved by the FDA, Braeburn will be solely responsible for the marketing, manufacture and commercialization of Probuphine in the Territory and, accordingly, the timing and amount of any royalty revenues or sales milestones we receive from this product will be wholly dependent upon Braeburn’s ability to successfully launch and commercialize this product in the Territory. Braeburn is a recently formed company and does not have a track record upon which investors can rely on making an investment decision. Additionally, our ability to generate revenues in the Territory from any additional indications for Probuphine, including chronic pain, depends on Braeburn’s ability to successfully develop, obtain regulatory approvals for and commercialize the product for additional indications. We do not have control over the amount and timing of resources that Braeburn will dedicate to these efforts, none of which have commenced to date. We will be similarly dependent on the development, regulatory and marketing efforts of third parties with respect to revenues, if any, from sales of Probuphine outside the Territory. To date, we have not entered into any collaborative arrangements or granted any rights with respect to Probuphine in the rest of the world.
Our ProNeura development programs are at very early stages and will require substantial additional resources that may not be available to us.
To date, we have conducted limited research and development activities based on our ProNeura delivery system beyond Probuphine. We will require substantial additional funds to support our research and development activities, and the anticipated costs of preclinical studies and clinical trials, regulatory approvals and eventual commercialization of ProNeura for PD or any therapeutic based on our ProNeura platform technology. If we are unable to generate sufficient revenues from royalties from the sale of Probuphine or other payments under our license agreement with Braeburn, we will need to seek additional sources of financing, which may not be available on favorable terms, if at all. If we do not succeed in raising the requisite financing on acceptable terms, we may be unable to initiate clinical trials or obtain approval of any product candidates from the FDA and other regulatory authorities. In addition, we could be forced to discontinue product development, forego sales and marketing efforts and forego attractive business opportunities.
To the extent we raise additional capital through the sale of equity securities, the issuance of those securities could result in dilution to our shareholders. In addition, if we obtain debt financing, a substantial portion of our operating cash flow may be dedicated to the payment of principal and interest on such indebtedness, thus limiting funds available for our business activities. If adequate funds are not available, we may be required to delay, reduce the scope of or eliminate our research and development programs, reduce our commercialization efforts or curtail our operations. In addition, we may be required to obtain funds through arrangements with collaborative partners or others that may require us to relinquish rights to technologies, product candidates or products that we would otherwise seek to develop or commercialize ourselves or license rights to technologies, product candidates or products on terms that are less favorable to us than might otherwise be available.
Our ProNeura program for PD is at a very early stage and we may not be able to successfully develop this product or any other product based on our ProNeura drug delivery technology.
Our ability to successfully develop any future product candidates based on our ProNeura drug delivery technology is subject to the risks of failure and delay inherent in the development of new pharmaceutical products, including: delays in product development, clinical testing, or manufacturing; unplanned expenditures in product development, clinical testing, or manufacturing; failure to receive regulatory approvals; emergence of superior or equivalent products; inability to manufacture on its own, or through any others, product candidates on a commercial scale; and failure to achieve market acceptance.
Because of these risks, our research and development efforts may not result in any commercially viable products. If a significant portion of these development efforts are not successfully completed, required regulatory approvals are not obtained or any approved products are not commercially successfully, our business, financial condition, and results of operations may be materially harmed.
| 16 |
Our development and commercialization strategy for ProNeura for PD depends, in part, upon the FDA’s prior findings regarding the safety and efficacy of ropinirole based on data not developed by us, but upon which the FDA may rely in reviewing our NDA.
We are developing ProNeura for PD with the expectation that it will be eligible for approval through the regulatory pathway under Section 505(b)(2) of the FDCA. Section 505(b)(2) of the FDCA allows an NDA to rely in part on data in the public domain or the FDA’s prior conclusions regarding the safety and effectiveness of an approved drug product, which could expedite the development program for ProNeura for PD by potentially decreasing the amount of clinical data that would need to be generated in order to obtain FDA approval. If the FDA does not allow us to pursue the Section 505(b)(2) regulatory pathway as anticipated, we may need to conduct additional clinical trials, provide additional data and information, and meet additional standards for product approval. If this were to occur, the time and financial resources required to obtain FDA approval for ProNeura for PD, and complications and risks associated with regulatory approval, would likely substantially increase. Moreover, inability to pursue the Section 505(b)(2) regulatory pathway may result in new competitive products reaching the market more quickly than ProNeura for PD, which would adversely impact our competitive position and prospects. Even if we are able to utilize the Section 505(b)(2) regulatory pathway, there is no guarantee that this regulatory pathway will ultimately lead to accelerated product development or earlier approval for ProNeura for PD. Moreover, notwithstanding the approval of many products by the FDA pursuant to Section 505(b)(2), over the last few years, some pharmaceutical companies and others have objected to the FDA’s interpretation of Section 505(b)(2). If the FDA changes its interpretation of Section 505(b)(2), or if the FDA’s interpretation is successfully challenged in court, this result could delay or even prevent the FDA from approving any Section 505(b)(2) NDAs that we submit. Such a result could require us to conduct additional testing and costly clinical trials, which could substantially delay or prevent the approval and launch of ProNeura for PD. The FDA may require us to perform additional studies or measurements to support any changes in our product as compared to the approved product. If we utilize Section 505(b)(2), the FDA may approve our new product for all or some of the label indications for which the referenced product has been approved, as well as for any new indication sought by us.
Clinical trials required for new product candidates are expensive and time-consuming, and their outcome is uncertain.
In order to obtain FDA approval to market a new drug product based on our ProNeura drug delivery technology, we must demonstrate proof of safety and effectiveness in humans. To meet these requirements, we must conduct “adequate and well controlled” clinical trials. Conducting clinical trials is a lengthy, time-consuming, and expensive process. The length of time may vary substantially according to the type, complexity, novelty, and intended use of the product candidate, and often can be several years or more per trial. Delays associated with products for which we are directly conducting clinical trials may cause us to incur additional operating expenses. The commencement and rate of completion of clinical trials may be delayed by many factors, including, for example: inability to manufacture sufficient quantities of qualified materials under cGMP, for use in clinical trials; slower than expected rates of patient recruitment; failure to recruit a sufficient number of patients; modification of clinical trial protocols; changes in regulatory requirements for clinical trials; the lack of effectiveness during clinical trials; the emergence of unforeseen safety issues; delays, suspension, or termination of the clinical trials due to the institutional review board responsible for overseeing the study at a particular study site; and government or regulatory delays or “clinical holds” requiring suspension or termination of the trials.
The results from early clinical trials are not necessarily predictive of results obtained in later clinical trials. Accordingly, even if we obtain positive results from early clinical trials, we may not achieve the same success in future clinical trials. Clinical trials may not demonstrate statistically significant safety and effectiveness to obtain the requisite regulatory approvals for product candidates.
The failure of clinical trials to demonstrate safety and effectiveness for the desired indications could harm the development of that product candidate and other product candidates. This failure could cause us to abandon a product candidate and could delay development of other product candidates. Any delay in, or termination of, our clinical trials would delay the filing of our NDAs with the FDA and, ultimately, our ability to commercialize our product candidates and generate product revenues. Any change in, or termination of, our clinical trials could materially harm our business, financial condition, and results of operations.
| 17 |
If Probuphine or any other product candidate that we may successfully develop does not achieve broad market acceptance among physicians, patients, healthcare payors and the medical community, the revenues that it generates from their sales will be limited.
Even if Probuphine or any other product candidate we may in the future develop receives regulatory approval, they may not gain market acceptance among physicians, patients, healthcare payors and the medical community. Coverage and reimbursement of our product candidates by third-party payors, including government payors, generally is also necessary for commercial success. The degree of market acceptance of any approved products will depend on a number of factors, including:
| · | the efficacy and safety as demonstrated in clinical trials; |
| · | the clinical indications for which the product is approved; |
| · | acceptance by physicians, operators of hospitals and clinics and patients of the product as a safe and effective product; |
| · | the potential and perceived advantages of the product over alternative treatments; |
| · | the safety of the product in broader patient groups, including its use outside of approved indications; |
| · | the cost of treatment in relation to alternative treatments; |
| · | the availability of adequate reimbursement and pricing by third parties and government authorities; |
| · | the prevalence and severity of adverse events; |
| · | the effectiveness of sales and marketing efforts; and |
| · | unfavorable publicity relating to the product. |
If any product candidate is approved but does not achieve an adequate level of acceptance by physicians, hospitals and clinics, healthcare payors and patients, we may not generate significant revenue from such products.
We must comply with extensive government regulations.
The research, development, manufacture labeling, storage, record-keeping, advertising, promotion, import, export, marketing and distribution of pharmaceutical products are subject to an extensive regulatory approval process by the FDA in the U.S. and comparable health authorities in foreign markets. The process of obtaining required regulatory approvals for drugs is lengthy, expensive and uncertain. Approval policies or regulations may change and the FDA and foreign authorities have substantial discretion in the pharmaceutical approval process, including the ability to delay, limit or deny approval of a product candidate for many reasons. Despite the time and expense invested in clinical development of product candidates, regulatory approval is never guaranteed. Regulatory approval may entail limitations on the indicated usage of a drug, which may reduce the drug’s market potential. Even if regulatory clearance is obtained, post-market evaluation of the products, if required, could result in restrictions on a product’s marketing or withdrawal of the product from the market, as well as possible civil and criminal sanctions. Of the large number of drugs in development, only a small percentage successfully complete the regulatory approval process and are commercialized.
We are dependent upon key collaborative relationships and license agreements.
We will rely significantly on the resources of third parties to market and commercialize Probuphine, if approved, as well as any other products we may develop. For example, our ability to ultimately derive revenues from Probuphine in the United States and Canada is dependent upon Braeburn implementing a successful marketing program for the treatment of opioid dependence in adults and pursuing development and commercialization of the product for other indications. Beyond any contractual rights, we cannot control the amount or timing of resources that any existing or future corporate partner devotes to product development and commercialization efforts for our product candidates. We depend on our ability to maintain existing collaborative relationships, to develop new collaborative relationships with third parties and potentially to acquire or in-license additional products and technologies for the development of new product candidates.
| 18 |
Our dependence on third party collaborators and license agreements subjects us to a number of risks, including:
| · | our collaborators may not comply with applicable regulatory guidelines with respect to developing or commercializing our products, which could adversely impact sales or future development of our products; |
| · | we and our collaborators could disagree as to future development plans and our collaborators may delay, fail to commence or stop future clinical trials or other development; and |
| · | there may be disputes between us and our collaborators, including disagreements regarding the license agreements, that may result in the delay of or failure to achieve developmental, regulatory and commercial objectives that would result in milestone or royalty payments and/or the delay or termination of any future development or commercialization of our products. |
In addition, collaborators may, to the extent permitted by our agreements, develop products that divert resources from our products, preclude us from entering into collaborations with their competitors or terminate their agreements with us prematurely. For example, Braeburn recently announced that it has obtained an exclusive license from Camurus for its long-acting bupenorphine injectables under development, which, if approved, could in the future divert resources from Probuphine. Moreover, disagreements could arise with our collaborators or strategic partners over rights to our intellectual property and our rights to share in any of the future revenues from products or technologies resulting from use of our technologies, or our activities in separate fields may conflict with other business plans of our collaborators.
We face risks associated with third parties conducting preclinical studies and clinical trials of our products; as well as our dependence on third parties to manufacture any products that we may successfully develop.
We depend on third-party laboratories and medical institutions to conduct preclinical studies and clinical trials for our products and other third-party organizations to perform data collection and analysis, all of which must maintain both good laboratory and good clinical practices. We also depend upon third party manufacturers for the production of any products we may successfully develop to comply with current Good Manufacturing Practices of the FDA, which are similarly outside our direct control. If third party laboratories and medical institutions conducting studies of our products fail to maintain both good laboratory and clinical practices, the studies could be delayed or have to be repeated. Similarly, if the manufacturers of any products we develop in the future fail to comply with current Good Manufacturing Practices of the FDA, we may be forced to cease manufacturing such product until we have found another third party to manufacture the product.
We face risks associated with product liability lawsuits that could be brought against us.
The testing, manufacturing, marketing and sale of human therapeutic products entail an inherent risk of product liability claims. We currently have a limited amount of product liability insurance, which may not be sufficient to cover claims that may be made against us in the event that the use or misuse of our product candidates causes, or merely appears to have caused, personal injury or death. In the event we are forced to expend significant funds on defending product liability actions, and in the event those funds come from operating capital, we will be required to reduce our business activities, which could lead to significant losses. Adequate insurance coverage may not be available in the future on acceptable terms, if at all. If available, we may not be able to maintain any such insurance at sufficient levels of coverage and any such insurance may not provide adequate protection against potential liabilities. Whether or not a product liability insurance policy is obtained or maintained in the future, any claims against us, regardless of their merit, could severely harm our financial condition, strain our management and other resources or destroy the prospects for commercialization of the product which is the subject of any such claim.
| 19 |
We may be unable to protect our patents and proprietary rights.
Our future success will depend to a significant extent on our ability to:
| · | obtain and keep patent protection for our products and technologies on an international basis; |
| · | enforce our patents to prevent others from using our inventions; |
| · | maintain and prevent others from using our trade secrets; and |
| · | operate and commercialize products without infringing on the patents or proprietary rights of others. |
We cannot assure you that our patent rights will afford any competitive advantages, and these rights may be challenged or circumvented by third parties. Further, patents may not be issued on any of our pending patent applications in the U.S. or abroad. Because of the extensive time required for development, testing and regulatory review of a potential product, it is possible that before a potential product can be commercialized, any related patent may expire or remain in existence for only a short period following commercialization, reducing or eliminating any advantage of the patent. If we sue others for infringing our patents, a court may determine that such patents are invalid or unenforceable. Even if the validity of our patent rights is upheld by a court, a court may not prevent the alleged infringement of our patent rights on the grounds that such activity is not covered by our patent claims.
In addition, third parties may sue us for infringing their patents. In the event of a successful claim of infringement against us, we may be required to:
| · | pay substantial damages; |
| · | stop using our technologies and methods; |
| · | stop certain research and development efforts; |
| · | develop non-infringing products or methods; and |
| · | obtain one or more licenses from third parties. |
If required, we cannot assure you that we will be able to obtain such licenses on acceptable terms, or at all. If we are sued for infringement, we could encounter substantial delays in development, manufacture and commercialization of our product candidates. Any litigation, whether to enforce our patent rights or to defend against allegations that we infringe third party rights, will be costly, time consuming, and may distract management from other important tasks.
We also rely in our business on trade secrets, know-how and other proprietary information. We seek to protect this information, in part, through the use of confidentiality agreements with employees, consultants, advisors and others. Nonetheless, we cannot assure you that those agreements will provide adequate protection for our trade secrets, know-how or other proprietary information and prevent their unauthorized use or disclosure. To the extent that consultants, key employees or other third parties apply technological information independently developed by them or by others to our proposed products, disputes may arise as to the proprietary rights to such information, which may not be resolved in our favor.
We face intense competition.
Competition in the pharmaceutical and biotechnology industries is intense. We face, and will continue to face, competition from numerous companies that currently market, or are developing, products for the treatment of the diseases and disorders we have targeted. Many of these entities have significantly greater research and development capabilities, experience in obtaining regulatory approvals and manufacturing, marketing, financial and managerial resources than we have. We also compete with universities and other research institutions in the development of products, technologies and processes, as well as the recruitment of highly qualified personnel. Our competitors may succeed in developing technologies or products that are more effective than the ones we have under development or that render our proposed products or technologies noncompetitive or obsolete. In addition, our competitors may achieve product commercialization or patent protection earlier than we will.
| 20 |
Healthcare reform and restrictions on reimbursements may limit our financial returns.
Braeburn’s ability to commercialize Probuphine in the Territory and our ability or the ability of any future collaborators to commercialize Probuphine outside the Territory or to commercialize any other products we may successfully develop will depend in part on the extent to which government health administration authorities, private health insurers and other organizations will reimburse consumers for the cost of these products. These third parties are increasingly challenging both the need for and the price of new drug products. Significant uncertainty exists as to the reimbursement status of newly approved therapeutics. Adequate third party reimbursement may not be available for our own or our collaborator’s drug products to enable us or them to maintain price levels sufficient to realize an appropriate return on their and our investments in research and product development.
Health care reform measures and changes in policies, funding, staffing and leadership at the FDA and other agencies could hinder or prevent the commercial success of our products.
In the United States, there have been a number of legislative and regulatory changes to the healthcare system in ways that could affect our future results of operations and the future results of operations of our potential customers. For example, the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 established a new Part D prescription drug benefit, which became effective January 1, 2006. Under the prescription drug benefit, Medicare beneficiaries can obtain prescription drug coverage from private sector plans that are permitted to limit the number of prescription drugs that are covered in each therapeutic category and class on their formularies. If our products are not widely included on the formularies of these plans, our ability to market our products may be adversely affected.
Furthermore, there have been and continue to be a number of initiatives at the federal and state levels that seek to reduce healthcare costs. In March 2010, President Obama signed into law the Patient Protection and Affordable Health Care Act of 2010, as amended by the Health Care and Education Affordability Reconciliation Act of 2010 (jointly, the “PPACA”), which includes measures to significantly change the way health care is financed by both governmental and private insurers.
In addition, other legislative changes have been proposed and adopted since the PPACA was enacted. On August 2, 2011, the Budget Control Act of 2011, among other things, created measures for spending reductions by Congress. This includes aggregate reductions to Medicare payments to providers of up to 2% per fiscal year. On January 2, 2013, President Obama signed into law the American Taxpayer Relief Act of 2012 (the “ATRA”), which, among other things, reduced Medicare payments to several providers, including hospitals, imaging centers and cancer treatment centers, and increased the statute of limitations period for the government to recover overpayments to providers from three to five years. These laws may result in additional reductions in Medicare and other health care funding, which could have a material adverse effect on our customers and accordingly, our financial operations.
Additionally, individual states have become increasingly aggressive in passing legislation and implementing regulations designed to control pharmaceutical product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access, and marketing cost disclosure and transparency measures, and designed to encourage importation from other countries and bulk purchasing. Legally-mandated price controls on payment amounts by third-party payors or other restrictions could harm our business, results of operations, financial condition and prospects.
In addition, regional healthcare authorities and individual hospitals are increasingly using bidding procedures to determine what pharmaceutical products and which suppliers will be included in their prescription drug and other healthcare programs. This can reduce demand for our products or put pressure on our product pricing, which could negatively affect our business, results of operations, financial condition and prospects.
| 21 |
Additionally, given recent federal and state government initiatives directed at lowering the total cost of healthcare, Congress and state legislatures will likely continue to focus on healthcare reform, the cost of prescription drugs and the reform of the Medicare and Medicaid programs. While we cannot predict the full outcome of any such legislation, it may result in decreased reimbursement for prescription drugs, which may further exacerbate industry-wide pressure to reduce prescription drug prices. This could harm our ability to market our products and generate revenues. In addition, legislation has been introduced in Congress that, if enacted, would permit more widespread importation or re-importation of pharmaceutical products from foreign countries into the United States, including from countries where the products are sold at lower prices than in the United States. Such legislation, or similar regulatory changes, could lead to a decision to decrease our prices to better compete, which, in turn, could adversely affect our business, results of operations, financial condition and prospects. It is also possible that other legislative proposals having similar effects will be adopted.
Furthermore, regulatory authorities’ assessment of the data and results required to demonstrate safety and efficacy can change over time and can be affected by many factors, such as the emergence of new information, including on other products, changing policies and agency funding, staffing and leadership. We cannot be sure whether future changes to the regulatory environment will be favorable or unfavorable to our business prospects.
We may not be able to retain our key management and scientific personnel, and a loss of certain key personnel could significantly hinder our ability to move forward with our business plan.
As a company with a limited number of personnel, we are highly dependent on the services of our executive management and scientific staff, in particular Sunil Bhonsle and Marc Rubin, our President and Executive Chairman, respectively, and Katherine Glassman-Beebe our Executive Vice President and Chief Development Officer. The loss of one or more of such individuals could substantially impair ongoing research and development programs and could hinder our ability to obtain corporate partners. Our success depends in large part upon our ability to attract and retain highly qualified personnel. We compete in our hiring efforts with other pharmaceutical and biotechnology companies, as well as universities and nonprofit research organizations, and we may not be successful in our efforts to attract and retain personnel.
Our net operating losses and research and development tax credits may not be available to reduce future federal and state income tax payments.
At December 31, 2014, we had federal net operating loss and tax credit carryforwards of $232.5 million and $8.3 million, respectively, and state net operating loss and tax credit carryforwards of $154.7 million and $8.1 million, respectively, available to offset future taxable income, if any. Current federal and state tax laws include substantial restrictions on the utilization of net operating loss and tax credits in the event of an ownership change and we cannot assure you that our net operating loss and tax carryforwards will continue to be available.
We have a limited number of authorized shares of common stock available for issuance and will need to seek stockholder approval to amend our charter to either effect an increase in our authorized shares of common stock or a reverse split.
At March 24, 2015, we had 14,672,293 authorized but unissued shares of our common stock, substantially all of which were reserved for issuance upon the exercise of outstanding stock options and warrants, and did not have a sufficient number of authorized shares to permit exercise of the Class A Warrants and Underwriter Warrants we issued in connection with our October 2014 public offering. Furthermore, we will need to continue to seek additional financing in order to fund our product development programs until such time, if ever, as the Probuphine NDA is approved by the FDA and royalty and milestone payments are sufficient to fund our operations. We have agreed to seek stockholder approval of an amendment to our certificate of incorporation to effect either an increase in the number of authorized shares of common stock or a reverse split, in either case in an amount sufficient to permit the exercise in full of the Class A Warrants. An increase in the authorized number of shares of common stock and the subsequent issuance of such shares could have the effect of delaying or preventing a change in control of our company without further action by our stockholders. Shares of authorized and unissued common stock could, within the limits imposed by applicable law, be issued in one or more transactions which would make a change in control of our company more difficult, and therefore less likely. Furthermore, there are risks associated with effecting a reverse split, including a decline in the market price of our common stock and the possibility of certain shareholders owning “odd lots” of less than 100 shares, which may be more difficult to sell, or require greater transaction costs per share to sell, than shares in “round lots” of even multiples of 100 shares. In addition, because holders of our common stock have no preemptive rights to purchase or subscribe for any unissued stock of our company, the availability of a greater number of authorized shares, whether as a result of a reverse split or an increase in the authorized number, could result in additional dilution to existing shareholders and investors.
| 22 |
Our stock price has been and will likely continue to be volatile.
Our stock price has experienced substantial fluctuations and could continue to fluctuate significantly due to a number of factors, including:
| · | variations in our anticipated or actual operating results or prospects; |
| · | sales of substantial amounts of our common stock; |
| · | announcements about us or about our competitors, including introductions of new products; |
| · | litigation and other developments relating to our patents or other proprietary rights or those of our competitors; |
| · | conditions in the pharmaceutical or biotechnology industries; |
| · | governmental regulation and legislation; and |
| · | change in securities analysts’ estimates of our performance, or our failure to meet analysts’ expectations. |
Our common stock is not listed on a national securities exchange and we may not be able to obtain an uplisting to a national exchange in the foreseeable future, if ever.
Our common stock is currently listed on the OTCBB. Trading on the OTC Market is characterized by wide fluctuations in bid and asked prices and periods of inactive or limited trading. We expect to commence efforts to seek an uplisting to the Nasdaq Stock Market or another national securities exchange, however, we do not know whether we will be able to meet the initial listing criteria to enable us to obtain an uplisting of our common stock in the foreseeable future, if ever.
Our common stock is deemed to be a “penny stock,” which may make it more difficult for investors to sell their shares due to suitability requirements.
Our common stock is subject to Rule 15g-1 through 15g-9 under the Exchange Act, which imposes certain sales practice requirements on broker-dealers which sell our common stock to persons other than established customers and “accredited investors” (generally, individuals with a net worth in excess of $1,000,000 or annual incomes exceeding $200,000 (or $300,000 together with their spouses)). For transactions covered by this rule, a broker-dealer must make a special suitability determination for the purchaser and have received the purchaser’s written consent to the transaction prior to the sale. This rule adversely affects the ability of broker-dealers to sell our common stock and the ability of our stockholders to sell their shares of common stock.
Additionally, our common stock is subject to the SEC regulations for “penny stock.” Penny stock includes any equity security that is not listed on a national exchange and has a market price of less than $5.00 per share, subject to certain exceptions. The regulations require that prior to any non-exempt buy/sell transaction in a penny stock, a disclosure schedule set forth by the SEC relating to the penny stock market must be delivered to the purchaser of such penny stock. This disclosure must include the amount of commissions payable to both the broker-dealer and the registered representative and current price quotations for the common stock. The regulations also require that monthly statements be sent to holders of penny stock that disclose recent price information for the penny stock and information of the limited market for penny stocks. These requirements adversely affect the market liquidity of our common stock.
| 23 |
We have never paid and do not intend to pay cash dividends. As a result, capital appreciation, if any, will be your sole source of gain.
We have never paid cash dividends on any of our capital stock and we currently intend to retain future earnings, if any, to fund the development and growth of our business. In addition, the terms of existing and future debt agreements may preclude us from paying dividends. As a result, capital appreciation, if any, of our common stock will be your sole source of gain for the foreseeable future.
Provisions in our certificate of incorporation, our by-laws and Delaware law might discourage, delay or prevent a change in control of our company or changes in our management and, therefore, depress the trading price of our common stock.
Provisions of our certificate of incorporation, our by-laws and Delaware law may have the effect of deterring unsolicited takeovers or delaying or preventing a change in control of our company or changes in our management, including transactions in which our stockholders might otherwise receive a premium for their shares over then current market prices. In addition, these provisions may limit the ability of stockholders to approve transactions that they may deem to be in their best interests. These provisions include:
| · | the inability of stockholders to call special meetings; and |
| · | the ability of our Board of Directors to designate the terms of and issue new series of preferred stock without stockholder approval, which could include the right to approve an acquisition or other |
| · | change in our control or could be used to institute a rights plan, also known as a poison pill, that would work to dilute the stock ownership of a potential hostile acquirer, likely preventing acquisitions that have not been approved by our Board of Directors. |
In addition, Section 203 of the Delaware General Corporation Law prohibits a publicly-held Delaware corporation from engaging in a business combination with an interested stockholder, generally a person which together with its affiliates owns, or within the last three years, has owned 15% of our voting stock, for a period of three years after the date of the transaction in which the person became an interested stockholder, unless the business combination is approved in a prescribed manner.
The existence of the forgoing provisions and anti-takeover measures could limit the price that investors might be willing to pay in the future for shares of our common stock. They could also deter potential acquirers of our company, thereby reducing the likelihood that you could receive a premium for your common stock in an acquisition.
Item 2. Properties
Our executive offices are located in approximately 9,255 square feet of office space in South San Francisco, California that we occupy under a three-year operating lease expiring in June 2016. It is our intention to continue to be based in South San Francisco.
Item 3. Legal Proceedings
We are currently not a party to any material legal or administrative proceedings and are not aware of any pending or threatened legal or administrative proceedings against us.
| 24 |
PART II
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities.
(a) Price Range of Securities
Our common stock is traded in the over-the-counter market and has been quoted through the Over-The-Counter Bulletin Board under the symbol “TTNP” since June 2010. The following table sets forth, for the periods indicated, the high and low sale prices for our common stock as reported by the OTCBB. Quotations on the OTCBB reflect inter-dealer prices, without retail mark-up, mark-down or commissions, and may not represent actual transactions. For current price information, stockholders are urged to consult publicly available sources.
| High | Low | |||||||
| Fiscal 2014 | ||||||||
| Fourth Quarter | $ | 0.58 | $ | 0.44 | ||||
| Third Quarter | $ | 0.87 | $ | 0.52 | ||||
| Second Quarter | $ | 0.86 | $ | 0.55 | ||||
| First Quarter | $ | 0.84 | $ | 0.60 | ||||
| Fiscal 2013 | ||||||||
| Fourth Quarter | $ | 1.17 | $ | 0.58 | ||||
| Third Quarter | $ | 0.70 | $ | 0.46 | ||||
| Second Quarter | $ | 1.95 | $ | 0.43 | ||||
| First Quarter | $ | 2.48 | $ | 1.19 | ||||
(b) Approximate Number of Equity Security Holders
At March 24, 2015, there were 110,327,707 shares of our common stock outstanding held by 145 holders of record. The number of record holders was determined from the records of our transfer agent and does not include beneficial owners of common stock whose shares are held in the names of various security brokers, dealers, and registered clearing agencies.
(c) Dividends
We have never declared or paid any cash dividends on our common stock and we do not anticipate paying any cash dividends to shareholders in the foreseeable future. Any future determination to pay cash dividends will be at the discretion of the Board of Directors and will be dependent upon our financial condition, results of operations, capital requirements, and such other factors as the Board of Directors deem relevant.
(d)
Equity Compensation Plan Information
The following table sets forth aggregate information regarding our equity compensation plans in effect as of December 31, 2014:
| 25 |
| Plan category | Number of securities to be issued upon exercise of outstanding options, warrant and rights (a) | Weighted-average exercise price of outstanding options, warrants and rights (b) | Number of securities remaining available for future issuance under equity compensation plans (c) | |||||||||
| Equity compensation plans approved by security holders | 3,808,553 | $ | 1.23 | — | ||||||||
| Equity compensation plans not approved by security holders(1)(2)(3) | 3,170,000 | $ | 1.09 | 1,558,000 | ||||||||
| Total | 6,978,553 | $ | 1.16 | 1,558,000 | ||||||||
| (1) | In August 2002, we amended our 2001 Employee Non-Qualified Stock Option Plan. Pursuant to this amendment, a total of 1,750,000 shares of common stock were reserved and authorized for issuance for option grants to employees and consultants who are not officers or directors of Titan. At December 31, 2014, 1,124,000 of these non-qualified stock options remained outstanding. |
| (2) | In October 2007, we granted 1,500,000 non-qualified stock options outside of our stock option plans to our Chief Executive Officer, at an exercise price of $2.40, vesting equally over 48 months from the date of grant. At December 31, 2014, 437,500 of these non-qualified stock options remained outstanding. |
| (3) | In May 2009, we granted 615,000 and 310,000 non-qualified stock options outside of our stock option plans to our Executive Chairman and President, respectively, at an exercise price of $0.79, vesting equally over 48 months from the date of grant. |
| (4) | In February 2014, we established our 2014 Incentive Plan. A total of 2,500,000 shares of common stock were reserved and authorized for issuance for option grants and restricted share awards to employees, directors and consultants. At December 31, 2014, 683,500 non-qualified stock options and restricted share awards remained outstanding. |
| 26 |
Performance Graph
The information contained in the Performance Graph shall not be deemed to be “soliciting material” or “filed” with the SEC or subject to the liabilities of Section 18 of the Exchange Act, except to the extent that we specifically incorporate it by reference into a document filed under the Securities Act or the Exchange Act.
The following graph compares the cumulative total stockholder return on our common stock with the cumulative total stockholder return of (i) the NYSE MKT Index, and (ii) a peer group index consisting of companies reporting under the Standard Industrial Classification Code 2834 (Pharmaceutical Preparations). The graph assumes $100 invested on December 31, 2009 and assumes dividends reinvested. Measurement points are at the last trading day of the fiscal years ended December 31, 2010, 2011, 2012, 2013 and 2014. The stock price performance on the following graph is not necessarily indicative of future stock price performance.
COMPARE CUMULATIVE TOTAL RETURN
AMONG TITAN PHARMACEUTICALS, INC., NYSE MKT INDEX AND
SIC CODE INDEX
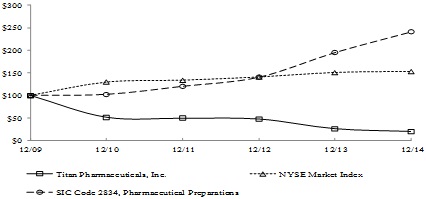
| 27 |
Item 6. Selected Financial Data.
The selected financial data presented below summarizes certain financial data which has been derived from and should be read in conjunction with our financial statements and notes thereto included in the section beginning on page F-1. See also “Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations.”
| Years Ended December 31, | ||||||||||||||||||||
| 2014 | 2013 | 2012 | 2011 | 2010 | ||||||||||||||||
| (in thousands, except per share data) | ||||||||||||||||||||
| Statement of Operations Data: | ||||||||||||||||||||
| Total revenue | $ | 3,646 | $ | 10,481 | $ | 7,117 | $ | 4,068 | $ | 10,093 | ||||||||||
| Operating expenses: | ||||||||||||||||||||
| Research and development | 4,075 | 8,309 | 10,610 | 11,206 | 12,855 | |||||||||||||||
| General and administrative | 3,046 | 3,063 | 4,877 | 3,368 | 3,263 | |||||||||||||||
| Other income (expense), net | 1,072 | 10,602 | (6,810 | ) | (4,697 | ) | (809 | ) | ||||||||||||
| Net income (loss) | (2,403 | ) | 9,711 | (15,180 | ) | (15,203 | ) | (6,834 | ) | |||||||||||
| Gain on retirement of preferred stock upon dissolution of subsidiary | — | — | — | — | 1,241 | |||||||||||||||
| Net income (loss) applicable to common stockholders | $ | (2,403 | ) | $ | 9,711 | $ | (15,180 | ) | $ | (15,203 | ) | $ | (5,593 | ) | ||||||
| Basic net income (loss) per common share | $ | (0.03 | ) | $ | 0.12 | $ | (0.23 | ) | $ | (0.26 | ) | $ | (0.09 | ) | ||||||
| Diluted net income (loss) per common share | $ | (0.04 | ) | $ | 0.10 | $ | (0.23 | ) | $ | (0.28 | ) | $ | (0.09 | ) | ||||||
| Shares used in computing: | ||||||||||||||||||||
| Basic net income (loss) per common share | 93,814 | 82,099 | 66,509 | 59,324 | 59,248 | |||||||||||||||
| Diluted net income (loss) per common share | 93,832 | 82,659 | 66,509 | 60,392 | 59,248 | |||||||||||||||
| As of December 31, | ||||||||||||||||||||
| 2014 | 2013 | 2012 | 2011 | 2010 | ||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| Balance Sheet Data: | ||||||||||||||||||||
| Cash | $ | 15,470 | $ | 11,798 | $ | 18,102 | $ | 5,406 | $ | 3,180 | ||||||||||
| Working capital (deficit) | 12,921 | 5,974 | 2,042 | 4,839 | (706 | ) | ||||||||||||||
| Total assets | 20,851 | 18,423 | 24,827 | 10,217 | 4,752 | |||||||||||||||
| Total stockholders’ equity (deficit) | 8,611 | 5,760 | (23,128 | ) | (20,079 | ) | (6,053 | ) | ||||||||||||
| 28 |
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations.
Forward-Looking Statements
Statements in the following discussion and throughout this report that are not historical in nature are “forward-looking statements” within the meaning of Section 27A of the Securities Act and Section 21E of the Exchange Act. You can identify forward-looking statements by the use of words such as “expect,” “anticipate,” “estimate,” “may,” “will,” “should,” “intend,” “believe,” and similar expressions. Although we believe the expectations reflected in these forward-looking statements are reasonable, such statements are inherently subject to risk and we can give no assurances that our expectations will prove to be correct. Actual results could differ from those described in this report because of numerous factors, many of which are beyond our control. These factors include, without limitation, those described under Item 1A “Risk Factors.” We undertake no obligation to update these forward-looking statements to reflect events or circumstances after the date of this report or to reflect actual outcomes. Please see “Note Regarding Forward-Looking Statements” at the beginning of this Annual Report on Form 10-K.
The following discussion of our financial condition and results of operations should be read in conjunction with our financial statements and the related notes thereto and other financial information appearing elsewhere in this Annual Report on Form 10-K.
Overview
We are a specialty pharmaceutical company developing proprietary therapeutics for the treatment of serious medical disorders. Our product development programs utilize our proprietary long-term drug delivery platform, ProNeura®, and focus primarily on innovative treatments for select chronic diseases for which steady state delivery of a drug provides an efficacy and/or safety benefit.
Probuphine®, our first product candidate to utilize ProNeura, is being developed for the long term maintenance treatment of opioid dependence and is designed to maintain a stable, around the clock blood level of the medicine buprenorphine in patients for six months following a single treatment. We have licensed the rights to commercialize Probuphine in the U.S. and Canada to Braeburn Pharmaceuticals Sprl (“Braeburn”)and during 2014 we have been supporting Braeburn and a team of experts to implement the program developed in cooperation with the FDA to address the items in the Complete Response letter (“CRL”) issued in April 2013. This includes conducting a double blind, double dummy clinical study of a four implant dose of Probuphine in clinically stable patients who are receiving maintenance treatment with an approved sublingual formulation containing buprenorphine at a daily dose of 8mg or less. This clinical study, which is being funded and managed by Braeburn, completed patient enrollment in November 2014 and study completion is anticipated by the end of the second quarter of 2015 followed by resubmission of the NDA later in the year, with a potential PDUFA date for the Probuphine NDA in the first half of 2016. Pursuant to our license agreement with Braeburn, as amended to date, we are entitled to receive a $15 million milestone payment upon FDA approval of the Probuphine NDA and royalties on net sales of Probuphine ranging in percentage from the mid-teens to the low twenties. The agreement also provides for up to $165 million in sales milestones and $35 million in regulatory milestones and entitles us to royalty in the low single digit percentage on sales by Braeburn, if any, of other future products in the addiction market.
We believe that our ProNeura technology has the potential to be used in the treatment of other chronic conditions, such as Parkinson’s disease (PD), where maintaining stable, around the clock blood levels of a dopamine agonist may benefit the patient and improve medical outcomes. We have commenced initial work on an implant formulation with ropinirole, a dopamine agonist approved for the treatment of PD. Our goal is to complete the non-clinical studies required in support of an Investigational New Drug (“IND”) application by early next year and enable commencement of a ‘proof of concept’ clinical study following the potential approval of Probuphine. We are also currently evaluating drugs and disease settings for opportunities to develop our drug delivery technology for other potential treatment applications in situations where conventional treatment is limited by variability in blood drug levels and poor patient compliance.
We operate in only one business segment, the development of pharmaceutical products.
| 29 |
Critical Accounting Policies and the Use of Estimates
Critical Accounting Policies and Use of Estimates
The preparation of our financial statements in conformity with accounting principles generally accepted in the United States requires management to make estimates and assumptions that affect the amounts reported in our financial statements and accompanying notes. Actual results could differ materially from those estimates. We believe the following accounting policies for the years ended December 31, 2014 and 2013 to be applicable:
Revenue Recognition
We generate revenue principally from collaborative research and development arrangements, technology licenses, and government grants. Consideration received for revenue arrangements with multiple components is allocated among the separate units of accounting based on their respective selling prices. The selling price for each unit is based on vendor-specific objective evidence, or VSOE, if available, third party evidence if VSOE is not available, or estimated selling price if neither VSOE nor third party evidence is available. The applicable revenue recognition criteria are then applied to each of the units.
Revenue is recognized when the four basic criteria of revenue recognition are met: (1) a contractual agreement exists; (2) transfer of technology has been completed or services have been rendered; (3) the fee is fixed or determinable; and (4) collectibility is reasonably assured. For each source of revenue, we comply with the above revenue recognition criteria in the following manner:
| · | Technology license agreements typically consist of non-refundable upfront license fees, annual minimum access fees or royalty payments. Non-refundable upfront license fees and annual minimum payments received with separable stand-alone values are recognized when the technology is transferred or accessed, provided that the technology transferred or accessed is not dependent on the outcome of our continuing research and development efforts. |
| · | Royalties earned are based on third-party sales of licensed products and are recorded in accordance with contract terms when third-party results are reliably measurable and collectibility is reasonably assured. We no longer recognize royalty income related to the Fanapt royalty payments received from Novartis unless Fanapt sales exceed certain thresholds (see Note 8, “Royalty Liability” for further discussion). |
| · | Government grants, which support our research efforts in specific projects, generally provide for reimbursement of approved costs as defined in the notices of grants. Grant revenue is recognized when associated project costs are incurred. |
| · | Collaborative arrangements typically consist of non-refundable and/or exclusive technology access fees, cost reimbursements for specific research and development spending, and various milestone and future product royalty payments. If the delivered technology does not have stand-alone value, the amount of revenue allocable to the delivered technology is deferred. Non-refundable upfront fees with stand-alone value that are not dependent on future performance under these agreements are recognized as revenue when received, and are deferred if we have continuing performance obligations and have no evidence of fair value of those obligations. Cost reimbursements for research and development spending are recognized when the related costs are incurred and when collections are reasonably expected. Payments received related to substantive, performance-based “at-risk” milestones are recognized as revenue upon achievement of the clinical success or regulatory event specified in the underlying contracts, which represent the culmination of the earnings process. Amounts received in advance are recorded as deferred revenue until the technology is transferred, costs are incurred, or a milestone is reached. |
Share-Based Payments
We recognize compensation expense for all share-based awards made to employees and directors. The fair value of share-based awards is estimated at the grant date based on the fair value of the award and is recognized as expense, net of estimated pre-vesting forfeitures, ratably over the vesting period of the award.
| 30 |
We use the Black-Scholes option pricing model to estimate the fair value method of our awards. Calculating stock-based compensation expense requires the input of highly subjective assumptions, including the expected term of the share-based awards, stock price volatility, and pre-vesting forfeitures. We estimate the expected term of stock options granted for the years ended December 31, 2014 and 2013 based on the historical experience of similar awards, giving consideration to the contractual terms of the share-based awards, vesting schedules and the expectations of future employee behavior. We estimate the volatility of our common stock at the date of grant based on the historical volatility of our common stock. The assumptions used in calculating the fair value of stock-based awards represent our best estimates, but these estimates involve inherent uncertainties and the application of management judgment. As a result, if factors change and we use different assumptions, our stock-based compensation expense could be materially different in the future. In addition, we are required to estimate the expected pre-vesting forfeiture rate and only recognize expense for those shares expected to vest. We estimate the pre-vesting forfeiture rate based on historical experience. If our actual forfeiture rate is materially different from our estimate, our stock-based compensation expense could be significantly different from what we have recorded in the current period.
Income Taxes
We make certain estimates and judgments in determining income tax expense for financial statement purposes. These estimates and judgments occur in the calculation of certain tax assets and liabilities, which arise from differences in the timing of recognition of revenue and expense for tax and financial statement purposes.
As part of the process of preparing our financial statements, we are required to estimate our income taxes in each of the jurisdictions in which we operate. This process involves us estimating our current tax exposure under the most recent tax laws and assessing temporary differences resulting from differing treatment of items for tax and accounting purposes.
We assess the likelihood that we will be able to recover our deferred tax assets. We consider all available evidence, both positive and negative, expectations and risks associated with estimates of future taxable income and ongoing prudent and feasible tax planning strategies in assessing the need for a valuation allowance. If it is not more likely than not that we will recover our deferred tax assets, we will increase our provision for taxes by recording a valuation allowance against the deferred tax assets that we estimate will not ultimately be recoverable.
Clinical Trial Accruals
We also record accruals for estimated ongoing clinical trial costs. Clinical trial costs represent costs incurred by clinical research organizations (“CROs”) and clinical sites. These costs are recorded as a component of research and development expenses. Under our agreements, progress payments are typically made to investigators, clinical sites and CROs. We analyze the progress of the clinical trials, including levels of patient enrollment, invoices received and contracted costs when evaluating the adequacy of accrued liabilities. Significant judgments and estimates must be made and used in determining the accrued balance in any accounting period. Actual results could differ from those estimates under different assumptions. Revisions are charged to expense in the period in which the facts that give rise to the revision become known. The actual clinical trial costs for the Probuphine studies conducted in the past three years have not differed materially from the estimated projection of expenses.
Warrants Issued in Connection with Equity Financing
We generally account for warrants issued in connection with equity financings as a component of equity, unless there is a deemed possibility that we may have to settle warrants in cash. For warrants issued with deemed possibility of cash settlement, we record the fair value of the issued warrants as a liability at each reporting period and record changes in the estimated fair value as a non-cash gain or loss in the Statements of Operations and Comprehensive Income (Loss).
| 31 |
Liquidity and Capital Resources
| 2014 | 2013 | 2012 | ||||||||||
| (in thousands) | ||||||||||||
| As of December 31: | ||||||||||||
| Cash | $ | 15,470 | $ | 11,798 | $ | 18,102 | ||||||
| Working capital | $ | 12,921 | $ | 5,974 | $ | 2,042 | ||||||
| Current ratio | 2.9:1 | 1.6:1 | 1.1:1 | |||||||||
| Years Ended December 31: | ||||||||||||
| Cash (used in) provided by operating activities | $ | (5,865 | ) | $ | (9,799 | ) | $ | 1,830 | ||||
| Cash used in investing activities | $ | (18 | ) | $ | (318 | ) | $ | (1,154 | ) | |||
| Cash provided by financing activities | $ | 9,555 | $ | 3,813 | $ | 12,020 | ||||||
Liquidity and Capital Resources
We have funded our operations since inception primarily through the sale of our securities and the issuance of debt, as well as with proceeds from warrant and option exercises, corporate licensing and collaborative agreements, the sale of royalty rights and government-sponsored research grants. At December 31, 2014, we had working capital of approximately $12.9 million compared to working capital of approximately $6.0 million at December 31, 2013.
Our operating activities used approximately $5.9 million of cash during the year ended December 31, 2014. This consisted primarily of the net loss for the period of approximately $2.4 million, approximately $1.1 million related to non-cash gains resulting from changes in the fair value of warrants and $3.3 million related to net changes in other operating assets and liabilities. This was offset in part by non-cash charges of approximately $0.5 million related to share-based compensation expenses and approximately $0.4 million related to depreciation and amortization. Uses of cash in operating activities were primarily to fund product development programs and administrative expenses.
Net cash used in investing activities of approximately $18,000 during the year ended December 31, 2014 and $0.3 million during the year ended December 31, 2013 was primarily related to purchases of equipment.
Our financing activities provided approximately $9.6 million, net of underwriting discounts, commissions, and other related fees, during the year ended December 31, 2014. This was primarily related to sale of common stock.
In March 2011, we entered into several agreements with entities affiliated with Deerfield pursuant to which Deerfield agreed to provide $20.0 million in funding to us. Pursuant to the terms of a facility agreement, we issued Deerfield 8.5% promissory notes in the aggregate principal amount of $20.0 million. We paid Deerfield a facility fee of $0.5 million and issued them the Deerfield Warrants to purchase 6,000,000 shares of our common stock. Under a royalty agreement, in exchange for $3.0 million that was recorded as royalty liability, we agreed to pay Deerfield 2.5% of the aggregate royalties on net sales of Fanapt, subsequent to the funding date, constituting a portion of the royalty revenue we receive from Novartis. The agreements with Deerfield also provided us with the option to repurchase the royalty rights for $40.0 million.
In November 2011, we entered into several agreements with Deerfield pursuant to which we agreed to pay them a substantial portion of the remaining future royalties on the sales of Fanapt in exchange for $5.0 million in cash that was recorded as royalty liability, a $10.0 million reduction in the principal amount owed to Deerfield under the existing facility agreement and a revised principal repayment schedule of $2.5 million per year for four years commencing in April 2013 to retire the remaining long-term debt of $10.0 million. Deerfield is entitled to the balance of our portion of the royalties on Fanapt (5.5% to 7.5% of net sales, net of the 2.5% we previously agreed to pay to Deerfield) up to specified threshold levels of net sales of Fanapt and 40% of the royalties above the threshold level.
In February 2013, we amended the terms of the Deerfield Warrants to permit payment of the exercise price through the reduction of the outstanding loan. In February and March 2013, Deerfield exercised all of the Deerfield Warrants resulting in a $7.5 million reduction of our outstanding indebtedness. In April 2013, we made the last $2.5 million installment payment and our debt obligation to Deerfield was satisfied in full.
| 32 |
In March 2013, we terminated our option to repurchase the royalty rights. As a result, we recognized a gain on the extinguishment of the royalty liability of $9.0 million, which was recorded in other income, because we are no longer required to account for it as a liability. Additionally, we no longer recognize royalty income related to the Fanapt royalty payments received from Novartis.
In November 2013, we entered into (i) a stock purchase agreement pursuant to which Braeburn made a $5.0 million equity investment in our company and (ii) an amendment to the license agreement with Braeburn primarily to modify the amount and timing of the approval and sales milestone payments payable under the license agreement.
In October 2014, we completed an underwritten public offering of 21,000,000 units at an offering price of $0.50 per unit, with each unit consisting of one share of common stock and 0.75 of a warrant, each full warrant to purchase one share of common stock at an exercise price of $0.60 per share (the “Public Offering”). Net proceeds were approximately $9.6 million after deducting underwriting discounts, commissions and other related expenses.
At December 31, 2014, we had cash of approximately $15.5 million, which we believe is sufficient to fund our planned operations into the fourth quarter of 2016.
Although Braeburn has commenced the clinical study and is proceeding with plans for completing the clinical study expeditiously, under our December 2012 license agreement with Braeburn, as amended, Braeburn currently has the technical right to terminate the Agreement. If Braeburn were to exercise its right to terminate the Agreement, we would not have sufficient funds available to us to complete the FDA regulatory process and, in the event of ultimate approval, commercialize Probuphine without raising additional capital. If we are unable to complete a debt or equity offering, or otherwise obtain sufficient financing in such event, our business and prospects would be materially adversely impacted. Furthermore, in order to advance our current ProNeura development program for Parkinson’s disease to later stage clinical studies, we will require additional funds, either through payments from Braeburn under the license agreement in the event the Probuphine NDA is ultimately approved or through other financing arrangements, to complete the clinical studies and regulatory approval process necessary to commercialize any additional products we might develop.
The following table sets forth the aggregate contractual cash obligations as of December 31, 2014 (in thousands):
| Payments Due by Period | ||||||||||||||||||||
| Contractual obligations | Total | < 1 year | 1-3 years | 3-5 years | 5years+ | |||||||||||||||
| Operating leases | $ | 317 | $ | 211 | $ | 106 | $ | — | $ | — | ||||||||||
| Total contractual cash obligations | $ | 317 | $ | 211 | $ | 106 | $ | — | $ | — | ||||||||||
Results of Operations
Year Ended December 31, 2014 Compared to Year Ended December 31, 2013
License revenues of approximately $3.6 million and $9.1 million for the years ended December 31, 2014 and 2013 reflect the amortization of the upfront license fee received from Braeburn in December 2012. Royalty revenues for the year ended December 31, 2013 reflects royalties on sales of Fanapt, all of which were paid to Deerfield in accordance with our royalty sales agreement. We no longer recognize Fanapt royalty revenues since all of such royalties are paid to third parties.
Research and development expenses for 2014 were approximately $4.1 million compared to approximately $8.3 million in 2013, a decrease of approximately $4.2 million, or 51%. The decrease in research and development costs was primarily associated with a decrease in external research and development expenses related to completion of the product development program and preparation and review of the NDA for our Probuphine product with the FDA. External research and development expenses include direct expenses such as CRO charges, investigator and review board fees, patient expense reimbursements, expenses for NDA preparation and contract manufacturing expenses. During 2014, our external research and development expenses relating to our Probuphine product development program were approximately $0.9 million compared to approximately $3.5 million for 2013. Other research and development expenses include internal operating costs such as clinical research and development personnel-related expenses, clinical trials related travel expenses, and allocation of facility and corporate costs. As a result of the risks and uncertainties inherently associated with pharmaceutical research and development activities described elsewhere in this document, we are unable to estimate the specific timing and future costs of our clinical development programs or the timing of material cash inflows, if any, from our product candidates. However, we anticipate that our research and development expenses will increase in connection with our ProNeura for PD development activities and any other ProNeura technology based product development program we may pursue.
| 33 |
General and administrative expenses for 2014 and 2013 remained constant at approximately $3.1 million.
Net other income for the year ended December 31, 2014 was approximately $1.1 million, compared to net other income of approximately $10.6 million in the comparable period in 2013. Net other income in 2014 consisted primarily of $1.1 million related to non-cash gains on changes in the fair value of warrants. Net other income in 2013 consisted primarily of approximately $9.0 million in other income generated by the termination of Titan’s royalty repurchase agreement with Deerfield, an approximately $1.9 million gain resulting from the settlement of indebtedness to Deerfield as a result of the exercise of all of the Deerfield Warrants and non-cash gains on changes in the fair value of warrants of approximately $1.7 million, which amounts were offset in part by interest expense of approximately $1.6 million related to the Deerfield loans and approximately $0.5 million in other expenses related to unamortized transaction fees related to the initial Deerfield debt transaction.
Our net loss applicable to common stockholders for the year ended December 31, 2014 was approximately $2.4 million, or approximately $0.03 per share, compared to our net income applicable to common stockholders of approximately $9.7 million, or approximately $0.12 per share, for the comparable period in 2013.
Year Ended December 31, 2013 Compared to Year Ended December 31, 2012
License revenues of approximately $9.1 million and $2.3 million for the years ended December 31, 2013 and 2012 reflect the amortization of the upfront license fee received from Braeburn in December 2012. Royalty revenues for the years ended December 31, 2013 and 2012 reflect royalties paid on sales of Fanapt, all of which were paid to Deerfield in accordance with our royalty sales agreement. We no longer recognize Fanapt royalty revenues since all of such royalties are paid to third parties. We generated no grant revenue during the year ended December 31, 2013 compared with $42,000 of NIH grant revenue during the year ended December 31, 2012 relating to our Probuphine program.
Research and development expenses for 2013 were approximately $8.3 million compared to approximately $10.6 million in 2012, a decrease of approximately $2.3 million, or 22%. The decrease in research and development costs was primarily associated with a decrease in external research and development expenses related to completion of the product development program and preparation and review of the NDA for our Probuphine product with the FDA. External research and development expenses include direct expenses such as CRO charges, investigator and review board fees, patient expense reimbursements, expenses for NDA preparation and contract manufacturing expenses. During 2013, our external research and development expenses relating to our Probuphine product development program were approximately $3.5 million compared to approximately $5.4 million for 2012. Other research and development expenses include internal operating costs such as clinical research and development personnel-related expenses, clinical trials related travel expenses, and allocation of facility and corporate costs. As a result of the risks and uncertainties inherently associated with pharmaceutical research and development activities described elsewhere in this report, we are unable to estimate the specific timing and future costs of our clinical development programs or the timing of material cash inflows, if any, from our product candidates.
General and administrative expenses for 2013 were approximately $3.1 million, compared to approximately $4.9 million in 2012, a decrease of approximately $1.8 million, or 37%. The decrease in general and administrative expenses was primarily related to decreases in non-cash stock compensation costs of approximately $1.3 million, employee-related costs of approximately $0.2 million and consulting and professional fees of approximately $0.3 million.
| 34 |
Net other income for the year ended December 31, 2013 was approximately $10.6 million, compared to net other expense of approximately $6.8 million in the comparable period in 2012. The increase in net other income during the year ended December 31, 2013 was primarily related to approximately $9.0 million of other income generated by the termination of our royalty repurchase agreement with Deerfield, an approximately $1.9 million gain resulting from the $7.5 million settlement of our indebtedness to Deerfield as a result of Deerfield’s exercise of all of the Deerfield Warrants, a decrease in interest expense of approximately $3.3 million related to the Deerfield loans and approximately $3.5 million related to non-cash gains on changes in the fair value of warrants. This was offset in part by approximately $0.5 million of other expense related to unamortized transaction fees related to the initial Deerfield debt transaction.
Our net income applicable to common stockholders for the year ended December 31, 2013 was approximately $9.7 million, or approximately $0.12 per share, compared to our net loss applicable to common stockholders of approximately $15.2 million, or approximately $0.23 per share, for the comparable period in 2012.
Off-Balance Sheet Arrangements
We have never entered into any off-balance sheet financing arrangements and we have never established any special purpose entities. We have not guaranteed any debt or commitments of other entities or entered into any options on non-financial assets.
| Item 7A. | Quantitative and Qualitative Disclosures About Market Risk |
We held no marketable securities at December 31, 2014 and 2013.
| Item 8. | Financial Statements and Supplementary Data. |
The response to this item is included in a separate section of this Report. See “Index to Financial Statements” on Page F-1.
| Item 9. | Changes and Disagreements with Accountants on Accounting and Financial Disclosure. |
None.
| Item 9A. | Controls and Procedures. |
(a) Evaluation of Disclosure Controls and Procedures: Our principal executive and financial officers reviewed and evaluated the effectiveness of our disclosure controls and procedures (as defined in Exchange Act Rule 13a-15(e)) as of the end of the period covered by this Annual Report on Form 10-K. Based on that evaluation, our principal executive and financial officers concluded that our disclosure controls and procedures are effective in timely providing them with material information relating to the Company, as required to be disclosed in the reports we file under the Exchange Act.
(b) Management’s Annual Report on Internal Control Over Financial Reporting:
Internal control over financial reporting refers to the process designed by, or under the supervision of, our Principal Executive Officer and Principal Financial Officer, and effected by our board of directors, management and other personnel, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles, and includes those policies and procedures that:
(1) Pertain to the maintenance of records that in reasonable detail accurately and fairly reflect the transactions and dispositions of our assets;
| 35 |
(2) Provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that our receipts and expenditures are being made only in accordance with authorization of our management and directors; and
(3) Provide reasonable assurance regarding prevention or timely detection of unauthorized acquisitions, use or disposition of our assets that could have a material effect on the financial statements.
Internal control over financial reporting cannot provide absolute assurance of achieving financial reporting objectives because of its inherent limitations. Internal control over financial reporting is a process that involves human diligence and compliance and is subject to lapses in judgment and breakdowns resulting from human failures. Internal control over financial reporting also can be circumvented by collusion or improper management overrides. Due to such limitations, there is a risk that material misstatements may not be prevented or detected on a timely basis by internal control over financial reporting. However, these inherent limitations are known features of the financial reporting process. Therefore, it is possible to design into the process safeguards to reduce, though not eliminate, this risk. Management is responsible for establishing and maintaining adequate internal control over financial reporting for the company.
Management has used the framework set forth in the report entitled Internal Control—Integrated Framework published by the Committee of Sponsoring Organizations of the Treadway Commission, known as COSO, to evaluate the effectiveness of the Company’s internal control over financial reporting. Based on this assessment, management has concluded that our internal control over financial reporting was effective as of December 31, 2014.
(c) Changes in Internal Control Over Financial Reporting: There were no changes in our internal control over financial reporting (as defined in Rules 13(a)-15(f) and 15(d)-15(f) under the Securities Act) during our most recent fiscal quarter that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Item 9B. Other Information.
None.
| 36 |
PART III
| Item 10. | Directors; Executive Officers and Corporate Governance |
Set forth below are the name, age and position and a brief account of the business experience of each of our executive officers and directors:
| Name | Age | Office | Director Since | |||
| Marc Rubin (1) | 60 | Executive Chairman of the Board | November 2007 | |||
| Sunil Bhonsle | 65 | President and Director | February 2004 | |||
| Joseph A. Akers (2) | 69 | Director | November 2014 | |||
| Victor J. Bauer (2)(3) | 79 | Director | November 1997 | |||
| Eurelio M. Cavalier (1)(3)(4) | 82 | Director | September 1998 | |||
| M. David MacFarlane (2)(4) | 74 | Director | May 2002 | |||
| James R. McNab, Jr. (4) | 71 | Director | November 2014 | |||
| Ley S. Smith (1)(2)(4) | 80 | Director | July 2000 |
| (1) | Member of Executive Committee |
| (2) | Member of Audit Committee |
| (3) | Member of Compensation Committee |
| (4) | Member of Nominating Committee |
Marc Rubin, M.D. served as our President and Chief Executive from October 2007 until December 2008 and was re-engaged as our Executive Chairman in May 2009. Until February 2007, Dr. Rubin served as Head of Global Research and Development for Bayer Schering Pharma, as well as a member of the Executive Committee of Bayer Healthcare and the Board of Management of Bayer Schering Pharma. Prior to the merger of Bayer Pharmaceuticals and Schering AG in June 2006, Dr. Rubin was a member of the Executive Board of Schering AG since joining the Company in October 2003, as well as Chairman of Schering Berlin Inc. and President of Berlex Pharmaceuticals, a division of Schering AG. From 1990 until August 2003, Dr. Rubin was employed by GlaxoSmithKline where he held positions of increasing responsibility in global clinical and commercial development overseeing programs in the United States, Europe, Asia and Latin America. From 2001 through 2003, he was Senior Vice President of Global Clinical Pharmacology & Discovery Medicine. Dr. Rubin holds an M.D. from Cornell University Medical College. Dr. Rubin currently serves on the board of directors of Curis Inc. and Galectin Therapeutics. Based on Dr. Rubin’s position as the executive chairman, his extensive senior management experience and service on boards of directors in the biotechnology and pharmaceutical industries and his medical background, our Board believes that Dr. Rubin has the appropriate set of skills to serve as a member of the Board.
Sunil Bhonsle served as our Executive Vice President and Chief Operating Officer from September 1995 until December 2008 and was re-engaged as our President in May 2009. Mr. Bhonsle served in various positions, including Vice President and General Manager — Plasma Supply and Manager — Inventory and Technical Planning, at Bayer Corporation from July 1975 until April 1995. Mr. Bhonsle holds an M.B.A. from the University of California at Berkeley and a B.Tech. in chemical engineering from the Indian Institute of Technology. Based on Mr. Bhonsle’s position as the president and his substantial experience in the pharmaceutical industry, particularly in the areas of clinical development and manufacturing, our Board believes that Mr. Bhonsle has the appropriate set of skills to serve as a member of the Board.
Joseph A. Akers was employed in various capacities by Bayer Corporation, Bayer Healthcare and certain related entities, including as president of the Hematology/Cardiology Business Unit from 2004 to 2007, president and chief executive officer of Bayer Business and Corporate Services from July 2002 through 2003 and executive vice president and chief administrative and financial officer from 1999 to July 2002. Mr. Akers received a B.S. in marketing and an M.B.A. in finance from the University of California at Berkeley. Based on Mr. Akers’ extensive management experience in the pharmaceutical industry, particularly in the areas of administration and finance, our Board believes that Mr. Akers has the appropriate set of skills to serve as a member of the Board.
| 37 |
Victor J. Bauer, Ph.D. serves as the President of Concordia Pharmaceuticals, LLC, a biopharmaceutical company he co-founded in 2004. From February 1997 through March 2003, Dr. Bauer was employed by Titan, most recently as our Executive Director of Corporate Development. From April 1996 until its merger into Titan, Dr. Bauer also served as a director and Chairman of Theracell. Since December 1992 Dr. Bauer has been a self-employed consultant to companies in the pharmaceutical and biotechnology industries. Prior to that time, Dr. Bauer was with Hoechst-Roussel Pharmaceuticals Inc., where he served as President from 1988 through 1992. Dr. Bauer holds an SB from MIT and a Ph.D. from the University of Wisconsin, and served as a Research Fellow at Harvard University. Based on Dr. Bauer’s extensive management and consulting experience in the biotechnology and pharmaceutical industries, particularly in the areas of research and product development, our Board believes that Dr. Bauer has the appropriate set of skills to serve as a member of the Board.
Eurelio M. Cavalier was employed in various capacities by Eli Lilly & Co. from 1958 until his retirement in 1994, serving as Vice President Sales from 1976 to 1982 and Group Vice President U.S. Pharmaceutical Business Unit from 1982 to 1993. Based on Mr. Cavalier’s management experience in the pharmaceutical industry, particularly in the area of sales and marketing, our Board of directors believes that Mr. Cavalier has the appropriate set of skills to serve as a member of the Board.
M. David MacFarlane, Ph.D. served as Vice President and Responsible Head of Regulatory Affairs of Genentech, Inc. from 1989 until his retirement in August 1999. Prior to joining Genentech, Inc., he served in various positions with Glaxo Inc., last as Vice President of Regulatory Affairs. Based on Dr. MacFarlane’s management experience in the pharmaceutical industry, particularly in the area of clinical and regulatory affairs, our Board believes that Dr. MacFarlane has the appropriate set of skills to serve as a member of the Board.
James R. McNab, Jr. has served since 1998 as chief executive officer and chairman of Palmetto Pharmaceuticals, Inc., a privately-held drug discovery company he founded. He has been a chairman of the board of directors of Curis, Inc. (Nasdaq:CRIS), an oncology focused biotechnology company, since May 2002. Since 2009, Mr. McNab has served as executive chairman of FirstString Research, Inc., a privately-held biopharmaceutical company, and as chief executive officer of JT Pharmaceuticals, Inc., a privately-held drug discovery company. Mr. McNab has co-founded several privately-held companies, including Sontra Medical Corporation, a drug delivery company, and Parker Medical Associates, a manufacturer and worldwide supplier of orthopedic and sports-related products. He received a B.A. in economics from Davidson College and an M.B.A. from the University of North Carolina at Chapel Hill. Based on Mr. McNab’s extensive management experience in the pharmaceutical industry, our Board believes that Mr. McNab has the appropriate set of skills to serve as a member of the Board.
Ley S. Smith served in various positions with The Upjohn Company and Pharmacia & Upjohn from 1958 until his retirement in November 1997. From 1991 to 1993 he served as Vice Chairman of the Board of The Upjohn Company, and from 1993 to 1995 he was President and Chief Operating Officer of The Upjohn Company. At the time of his retirement, Mr. Smith was Executive Vice President of Pharmacia & Upjohn, and President of Pharmacia & Upjohn’s U.S. Pharma Product Center. Based on Mr. Smith’s management experience in the pharmaceutical industry, our Board believes that Mr. Smith has the appropriate set of skills to serve as a member of the Board.
As indicated above, each of our directors has extensive management and operational experience in one or more facets of the pharmaceutical industry, including research, product development, clinical and regulatory affairs, manufacturing and sales and marketing, providing our company with the leadership needed by a biotechnology company in all stages of its development.
Directors serve until the next annual meeting or until their successors are elected and qualified. Officers serve at the discretion of the board of directors, subject to rights, if any, under contracts of employment. See “Item 6. Executive Compensation—Employment Agreements.”
Board Leadership Structure
Currently, our principal executive officer and chairman of the Board positions are held separately by Sunil Bhonsle and Marc Rubin, respectively.
| 38 |
Section 16(a) Beneficial Ownership Reporting Compliance
Section 16(a) of the Exchange Act, requires our executive officers, directors and persons who beneficially own more than 10% of a registered class of our equity securities to file with the Securities and Exchange Commission initial reports of ownership and reports of changes in ownership of our common stock and other equity securities. Such executive officers, directors, and greater than 10% beneficial owners are required by SEC regulation to furnish us with copies of all Section 16(a) forms filed by such reporting persons.
Based solely on our review of such forms furnished to us and written representations from certain reporting persons, we believe that all filing requirements applicable to our executive officers, directors and greater than 10% beneficial owners were complied with during 2014.
Code of Ethics
We adopted a Code of Business Conduct and Ethics (the “Code”) in February 2013 that applies to all directors, officers and employees. The Code was filed as an exhibit to our Annual Report on Form 10-K for the year ended December 31, 2012 and is available on our website at www.titanpharm.com. A copy of our code of ethics will also be provided to any person without charge, upon written request sent to us at our offices located at 400 Oyster Point Blvd, Suite 505, South San Francisco, California 94080.
Changes in Director Nomination Process for Stockholders
None.
| 39 |
| Item 11. | Executive Compensation |
Overview
During 2014, Dr. Rubin and Mr. Bhonsle continued as our Executive Chairman and President, respectively, with compensation packages structured to reflect our current level of operations and resources. The key objectives for 2014 were to support the review by the FDA of the Probuphine NDA, and if approved, support Braeburn in the commercial launch of the product. The FDA issued a CRL on April 30, 2013 thereby delaying potential approval of Probuphine until additional clinical and other requirements are met (See Item 7: Management’s Discussion and Analysis of Financial Condition and Results of Operations). Efforts subsequent to receipt of the CRL have focused on working with Braeburn and the FDA to establish a path forward for potential resubmission of the NDA with the additional information requested by the FDA. This compensation discussion describes the material elements of compensation awarded to, earned by, or paid to each of our executive officers who served as named executive officers during the year ended December 31, 2014. This compensation discussion focuses on the information contained in the following tables and related footnotes and narrative for primarily the last completed fiscal year; however, we also describe compensation actions taken before or after the last completed fiscal year to the extent it enhances the understanding of our executive compensation disclosure.
Compensation Program Objectives and Philosophy
Our Compensation Committee currently oversees the design and administration of our executive compensation program. It reviews and approves all elements of compensation for each of our named executive officers taking into consideration recommendations from our principal executive officer (for compensation other than his own), as well as competitive market guidance. We define our competitive markets for executive talent to be the pharmaceutical and biotechnology industries in northern California. To date, we have utilized the Radford Biotechnology Surveys, a third party market specific compensation survey, and, when applicable, other independent third-party compensation consultants to benchmark our executive compensation.
The principal elements of our executive compensation program have historically been base salary, annual cash incentives, long-term equity incentives in the form of stock options, other benefits and perquisites, post-termination severance and acceleration of stock option vesting for certain named executive officers upon termination and/or a change in control. Our other benefits and perquisites have consisted of life, health and disability insurance benefits, and a qualified 401(k) savings plan. Our philosophy has been to position the aggregate of these elements at a level that is competitive within the industry and commensurate with our size and performance recognizing operational needs and limited financial resources during this period.
During 2014, our operations continued to focus on efforts to realize maximum shareholder value from activities associated with our Probuphine and ProNeura development programs. Accordingly, our Compensation Committee continued a compensation plan which provides base salary and potential earnings through stock option and restricted stock awards.
Base Salaries
During 2014, the base salary of our named executives was reflective of the availability of resources and level of continuing operations. Dr. Rubin received an annual salary of $210,000 and Mr. Bhonsle received an annual salary of $300,000
As we continue to evaluate the strategic alternatives for us going forward and our related human resource requirements, our Compensation Committee will continue to review appropriate base salaries for our executive officers. In making its determination, the Compensation Committee will consider the time commitment necessary and the roles our executives will play in implementing our plans.
| 40 |
Long-term Equity Incentives
We provide the opportunity for our named executive officers and other executives to earn a long-term equity incentive award. Long-term incentive awards provide employees with the incentive to stay with us for longer periods of time, which in turn, provides us with greater stability. Equity awards also are less costly to us in the short term than cash compensation. We review long-term equity incentives for our named executive officers and other executives annually.
Historically, for our named executive officers, our stock option grants were of a size and term determined and approved by the Compensation Committee in consideration of the range of grants in the Radford Survey, generally falling within the 50-75% range outlined in the survey. We have traditionally used stock options as our form of equity compensation because stock options provide a relatively straightforward incentive for our executives, result in less immediate dilution of existing shareholders’ interests and, prior to our adoption of FAS 123(R), resulted in less compensation expense for us relative to other types of equity awards. Generally, all grants of stock options to our employees were granted with exercise prices equal to or greater than the fair market value of our common stock on the respective grant dates. For a discussion of the determination of the fair market value of these grants, see “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Critical Accounting Policies and the Use of Estimates.”
We do not time stock option grants to executives in coordination with the release of material non-public information. Our stock option grants have a 10-year contractual exercise term. In general, the option grants are also subject to the following post-termination and change in control provisions:
| Event | Award Vesting | Exercise Term | ||
| • Termination by us for Reason Other than Cause, Disability or Death | • Forfeit Unvested Options | • Earlier of: (1) 90 days or (2) Remaining Option Period | ||
| • Termination for Disability, Death or Retirement | • Forfeit Unvested Options | • Earlier of: (1) 2 years or (2) Remaining Option Period | ||
| • Termination for Cause | • Forfeit Vested and Unvested Options | • Expire | ||
| • Other Termination | • Forfeit Unvested Options | • Earlier of: (1) 90 days or (2) Remaining Option Period | ||
| • Change in Control | • Accelerated* | • * |
| * | The Compensation Committee may provide that, in the event of a change in control, any outstanding awards that are unexercisable or otherwise unvested will become fully vested and immediately exercisable. If there is a termination of employment, the applicable termination provisions regarding exercise term will apply. |
In February 2014, Dr. Rubin and Mr. Bhonsle each received a restricted stock award of 100,000 shares, of which 25,000 vested immediately and the balance will vest on the first anniversary of the grant date.
Compensation Committee Interlocks and Insider Participation
Members of our Compensation Committee of the board of directors are Eurelio M. Cavalier and Victor J. Bauer. No member of our Compensation Committee was, or has been at any time in the last 10 years, an officer or employee of Titan or any of our former subsidiaries.
No member of the Compensation Committee has a relationship that would constitute an interlocking relationship with executive officers or directors of the Company or another entity.
| 41 |
SUMMARY COMPENSATION TABLE
The following table shows information concerning the annual compensation for services provided to us by our Chief Executive Officer, our Chief Financial Officer and our other executive officers for the periods set forth.
| Name and Principal Position | Year | Salary ($) | Bonus ($) |
Options Awards |
Stock ($) (1) |
All Other Compensation ($) |
Total Compensation ($) |
|||||||||||||||||||
| Marc Rubin, M.D. | 2014 | $ | 210,000 | $ | — | $ | — | $ | 66,000 | $ | — | $ | 276,000 | |||||||||||||
| Executive Chairman | 2013 | $ | 210,000 | — | — | — | — | $ | 210,000 | |||||||||||||||||
| Sunil Bhonsle | 2014 | 300,000 | — | — | 66,000 | — | 366,000 | |||||||||||||||||||
| President and Principal Financial Officer | 2013 | 300,000 | — | — | — | — | 300,000 | |||||||||||||||||||
| (1) | Amounts shown represent the grant date fair value computed in accordance with FASB ASC 718. The assumptions used by us with respect to the valuation of option grants and stock awards are set forth in “Titan Pharmaceuticals, Inc. Financial Statements—Notes to Financial Statements—Note 12—Stock Plans.” |
GRANTS OF PLAN-BASED AWARDS
The following table shows information concerning grants of plan based awards to named executive officers during the year ended December 31, 2014.
Name | Grant | Approval | Number of | Exercise or | Grant Date | |||||||||||||||
| Marc Rubin, M.D. | 2/12/2014 | 2/11/2014 | 100,000 | (3) | $ | — | $ | 66,000 | ||||||||||||
| Sunil Bhonsle | 2/12/2014 | 2/11/2014 | 100,000 | (3) | $ | — | $ | 66,000 | ||||||||||||
| (1) | All grants were approved by the Compensation Committee on the dates indicated. |
| (2) | Valuation assumptions are found under “Titan Pharmaceuticals, Inc. Consolidated Financial Statements—Notes to Consolidated Financial Statements—Note 12—Stock Plans.” |
| (3) | These restricted stock awards were 25% vested on the grant date with the balance vesting on the first anniversary of the grant date. |
Employee Benefits Plans
The principal purpose of our stock incentive plans is to attract, motivate, reward and retain selected employees, consultants and directors through the granting of stock-based compensation awards. The stock option plans provides for a variety of awards, including non-qualified stock options, incentive stock options (within the meaning of Section 422 of the Code), stock appreciation rights, restricted stock awards, performance-based awards and other stock-based awards.
2002 Stock Incentive Plan
In July 2002, we adopted the 2002 Stock Incentive Plan, or the 2002 Plan. Under the 2002 Plan, as amended, a total of approximately 7.4 million shares of our common stock were authorized for issuance to employees, officers, directors, consultants, and advisers. The 2002 Plan expired by its terms in July 2012. On December 31, 2014, options to purchase an aggregate of 3,808,553 shares of our common stock were outstanding under the 2002 Plan.
| 42 |
2001 Stock Option Plan
In August 2001, we adopted the 2001 Employee Non-Qualified Stock Option Plan, or the 2001 NQ Plan, pursuant to which 1,750,000 shares of common stock were authorized for issuance for option grants to employees and consultants who are not officers or directors of Titan. The 2001 NQ Plan expired by its terms in August 2011. On December 31, 2014, options to purchase an aggregate of 1,124,000 shares of our common stock were outstanding under the 2001 NQ Plan.
2014 Incentive Plan
On February 11, 2014, our Board adopted the 2014 Incentive Plan, or the 2014 Plan, pursuant to which 2,500,000 shares of our common stock were authorized for issuance to employees, directors, officers, consultants and advisors. On December 31, 2014, restricted stock awards and options to purchase 683,500 shares of our common stock were outstanding under the 2014 Plan.
OUTSTANDING EQUITY AWARDS AT FISCAL YEAR-END
The following table summarizes the number of securities underlying outstanding plan awards for each named executive officer as of December 31, 2014.
| Awards | ||||||||||||||
| Name | Number of Securities Underlying Unexercised Awards (#) Exercisable |
Number of Securities Underlying Unexercised Awards (#) Unexercisable |
Exercise Price ($) |
Expiration Date | ||||||||||
| Marc Rubin, M.D. | 437,500 | — | $ | 2.40 | 10/01/2017 | |||||||||
| 7,500 | — | 1.52 | 5/30/2018 | |||||||||||
| 100,000 | — | 0.79 | 5/17/2019 | |||||||||||
| 15,000 | — | 0.79 | 5/17/2019 | |||||||||||
| 285,000 | — | 0.79 | 5/17/2019 | |||||||||||
| 615,000 | — | 0.79 | 5/17/2019 | |||||||||||
| 150,000 | — | 1.40 | 4/15/2021 | |||||||||||
| 250,000 | — | 1.15 | 1/3/2022 | |||||||||||
| — | 75,000 | — | 2/12/2024 | |||||||||||
| Sunil Bhonsle | 70,000 | — | 2.62 | 2/7/2015 | ||||||||||
| 10,137 | — | 1.40 | 1/3/2016 | |||||||||||
| 70,000 | — | 1.40 | 1/3/2016 | |||||||||||
| 11,250 | — | 2.35 | 8/29/2016 | |||||||||||
| 30,737 | — | 3.13 | 1/3/2017 | |||||||||||
| 45,929 | — | 3.13 | 1/3/2017 | |||||||||||
| 5,000 | — | 1.52 | 5/30/2018 | |||||||||||
| 100,000 | — | 0.79 | 5/17/2019 | |||||||||||
| 10,000 | — | 0.79 | 5/17/2019 | |||||||||||
| 390,000 | — | 0.79 | 5/17/2019 | |||||||||||
| 310,000 | — | 0.79 | 5/17/2019 | |||||||||||
| 200,000 | — | 1.40 | 4/15/2021 | |||||||||||
| 300,000 | — | 1.15 | 1/3/2022 | |||||||||||
| — | 75,000 | — | 2/12/2024 | |||||||||||
There were no option exercises by our named executive officers during 2014.
Pension Benefits
We do not sponsor any qualified or non-qualified defined benefit plans.
| 43 |
Nonqualified Deferred Compensation
We do not maintain any non-qualified defined contribution or deferred compensation plans. The Compensation Committee, which is comprised solely of “outside directors” as defined for purposes of Section 162(m) of the Code, may elect to provide our officers and other employees with non-qualified defined contribution or deferred compensation benefits if the Compensation Committee determines that doing so is in our best interests. We sponsor a tax qualified defined contribution 401(k) plan in which Dr. Rubin and Mr. Bhonsle participated.
Employment Agreements
During the year ended December 31, 2014, we were not parties to employment agreements with Dr. Rubin and Mr. Bhonsle as such agreements expired by their terms on December 31, 2013.
DIRECTOR COMPENSATION
Summary of Director Compensation
The following table summarizes compensation that our directors earned during 2014 for services as members of our Board.
| Name | Fees Earned or Paid in Cash ($) | Stock Awards ($) | Options Awards ($) | Non-Equity Incentive Plan Compensation ($) | Nonqualified Deferred Compensation Earnings ($) | All Other Compensation ($) | Total ($) | |||||||||||||||||||||
| Joseph A. Akers (1) | $ | 12,500 | $ | — | $ | 10,863 | $ | — | $ | — | $ | — | $ | 23,363 | ||||||||||||||
| Victor J. Bauer, Ph.D. | 55,000 | — | 15,953 | — | — | — | 70,953 | |||||||||||||||||||||
| Eurelio M. Cavalier | 62,500 | — | 15,953 | — | — | — | 78,453 | |||||||||||||||||||||
| M. David MacFarlane, Ph.D. | 55,000 | — | 15,953 | — | — | — | 70,953 | |||||||||||||||||||||
| James R. McNab, Jr. (1) | 12,500 | — | 10,863 | — | — | — | 23,263 | |||||||||||||||||||||
| Ley S. Smith | 62,500 | — | 15,953 | — | — | — | 78,453 | |||||||||||||||||||||
| (1) | Mr. Akers and Mr. McNab joined the Board on November 10, 2014. |
| Item 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters. |
The following table sets forth
as of March 24, 2015, the number of shares of our common stock beneficially owned by (i) each person who is known by us to be the
beneficial owner of more than five percent of our common stock; (ii) each director and director nominee; (iii) each of the named
executive officers in the Summary Compensation Table; and (iv) all directors and executive officers as a group. As of March 24,
2015, we had 110,327,707 shares of common stock issued and outstanding.
Beneficial ownership is determined in accordance with the rules of the Securities and Exchange Commission (the “SEC”) and generally includes voting or investment power with respect to securities. Unless otherwise indicated, the stockholders listed in the table have sole voting and investment power with respect to the shares indicated.
| Name and Address of Beneficial Owner (1) | Shares Beneficially Owned (2) | Percent of Shares Beneficially Owned | ||||||
| Joseph A. Akers | 37,500 | (3) | * | |||||
| Victor J. Bauer, Ph.D. | 321,144 | (4) | * | |||||
| Sunil Bhonsle | 2,335,310 | (5) | 2.1 | % | ||||
| Eurelio M. Cavalier | 447,500 | (6) | * | |||||
| M. David MacFarlane, Ph.D. | 342,500 | (7) | * | |||||
| James R. McNab, Jr. | 387,500 | (8) | * | |||||
| Marc Rubin, M.D. | 2,742,200 | (9) | 2.4 | |||||
| Ley S. Smith | 377,500 | (10) | * | |||||
| Braeburn Pharmaceuticals BVBA SPRL | 9,650,000 | (11) | 8.7 | % | ||||
| Broadfin Capital, LLC | 10,872,500 | (12) | 9.9 | % | ||||
| Robert E. Mead | 6,377,885 | (13) | 5.8 | % | ||||
| All executive officers and directors as a group (6) persons | 6,991,154 | 6.1 | % | |||||
| 44 |
| * | Less than one percent. |
| (1) | Unless otherwise indicated, the address of such individual is c/o Titan Pharmaceuticals, Inc., 400 Oyster Point Boulevard, Suite 505, South San Francisco, California 94080. |
| (2) | In computing the number of shares beneficially owned by a person and the percentage ownership of a person, shares of our common stock subject to options held by that person that are currently exercisable or exercisable within 60 days of March 24, 2015 are deemed outstanding. Such shares, however, are not deemed outstanding for purposes of computing the percentage ownership of each other person. Except as indicated in the footnotes to this table and pursuant to applicable community property laws, the persons named in the table have sole voting and investment power with respect to all shares of common stock. |
| (3) | Includes 37,500 shares issuable upon exercise of outstanding options. |
| (4) | Includes 285,000 shares issuable upon exercise of outstanding options. |
| (5) | Includes (i) 1,603,053 shares issuable upon exercise of outstanding options and (ii) 300,757 shares held in a family trust for which he serves as trustee. |
| (6) | Includes 265,000 shares issuable upon exercise of outstanding options. |
| (7) | Includes 220,000 shares issuable upon exercise of outstanding options. |
| (8) | Includes 37,500 shares issuable upon exercise of outstanding options. | |
| (9) | Includes 1,960,000 shares issuable upon exercise of outstanding options. | |
| (10) | Includes 265,000 shares issuable upon exercise of outstanding options. |
| (11) | Derived from a Schedule 13D filed by Braeburn, Apple Tree Consolidated BVBA Sprl (“ATC”), Apple Tree Investments S.a.r.l (“ATI”), Apple Tree Partners IV, L.P. (“ATP IV”), ATP III GP, Ltd. (“ATP GP”) and Seth L. Harrison (“Harrison”). ATP GP is the sole general partner of ATP IV. Harrison is the sole owner and director of ATP GP. As the sole owner of Braeburn, ATC may be deemed to own beneficially such shares. As the sole owner of ATC, ATI may be deemed to own beneficially such shares. As the sole owner of ATI, ATP IV may be deemed to own beneficially such shares. As the sole general partner of ATP IV, ATP GP may be deemed to own beneficially such shares. As the sole owner and director of ATP GP, Harrison may be deemed to own beneficially such shares. Each of the foregoing persons except Braeburn, disclaims beneficial ownership of such shares except to the extent of their pecuniary interest therein, if any. The address of the principal business office of Braeburn is Brugmannlaan 147, 1190 Vorst, Belgium. |
| (12) | Derived from a Schedule 13G filed by Broadfin Capital, LLC, Broadfin Healthcare Master Fund, Ltd. And Kevin Kotler. The address of the principal business office of Broadfin Capital, LLC is 237 Park Avenue, Suite 900, New York, New York 10017. Includes 4,500,000 shares issuable upon exercise of outstanding warrants. | |
| (13) | Derived from a Schedule 13G filed by Mr. Mead. The address of Mr. Mead’s principal business office is 3653 Maplewood Ave., Dallas, TX 75205. |
| Item 13. | Certain Relationships and Related Transactions, and Director Independence |
Certain Relationships and Related Transactions.
None.
Independence of Directors
The following members of our Board meet the independence requirements and standards currently established by the NYSE MKT: Joseph A. Akers, Victor J. Bauer, Eurelio M. Cavalier, M. David MacFarlane, James R. McNab, Jr. and Ley S. Smith.
| 45 |
Board Committees
Our Board has established the following three standing committees: audit committee; compensation committee; and nominating and governance committee, or nominating committee.
The audit committee was formed in compliance with Section 3(a)(58)(A) of the Exchange Act and consists of Joseph A. Akers, Ley S. Smith, M. David MacFarlane and Victor J. Bauer, each of whom meets the independence requirements and standards currently established by the NYSE MKT and the SEC. In addition, the Board has determined that Messrs. Akers and Smith are “audit committee financial experts” and “independent” as defined under the relevant rules of the SEC and the NYSE MKT. The audit committee assists the Board by overseeing the performance of the independent auditors and the quality and integrity of Titan’s internal accounting, auditing and financial reporting practices. The audit committee is responsible for retaining (subject to stockholder ratification) and, as necessary, terminating, the independent auditors, annually reviews the qualifications, performance and independence of the independent auditors and the audit plan, fees and audit results, and pre-approves audit and non-audit services to be performed by the auditors and related fees. During the fiscal year ended December 31, 2014, the audit committee met four times.
The compensation committee makes recommendations to the Board concerning salaries and incentive compensation for our officers, including our Principal Executive Officer, and employees and administers our stock option plans. The compensation committee consists of Eurelio M. Cavalier and Victor J. Bauer, each of whom meets the independence requirements and standards currently established by the NYSE MKT. The compensation committee did not meet as a separate committee, but did take action by written consent one time during the fiscal year ended December 31, 2014.
The purpose of the nominating committee is to assist the Board in identifying qualified individuals to become Board members, in determining the composition of the Board and in monitoring the process to assess Board effectiveness. The nominating committee consists of Eurelio M. Cavalier, M. David MacFarlane, James R. McNab, Jr. and Ley S. Smith, each of whom meets the independence requirements and standards currently established by the NYSE MKT. The nominating committee met two times as a separate committee and took action by written consent one time during the fiscal year ended December 31, 2014.
The charters for the audit, compensation and nominating committees, which have been adopted by our Board, contain detailed descriptions of the committees’ duties and responsibilities and are available in the Investor Relations section of our website at www.titanpharm.com.
Role of the Board in Risk Oversight
Our audit committee is primarily responsible for overseeing our risk management processes on behalf of the full Board. The audit committee receives reports from management at least quarterly regarding our assessment of risks. In addition, the audit committee reports regularly to the full Board, which also considers our risk profile. The audit committee and the full Board focus on the most significant risks we face and our general risk management strategies. While the Board oversees our risk management, management is responsible for day-to-day risk management processes. Our Board expects management to consider risk and risk management in each business decision, to proactively develop and monitor risk management strategies and processes for day-to-day activities and to effectively implement risk management strategies adopted by the audit committee and the Board. We believe this division of responsibilities is the most effective approach for addressing the risks we face and that our Board leadership structure, which also emphasizes the independence of the Board in its oversight of its business and affairs, supports this approach.
Board Meetings
Our business and affairs are managed under the direction of our Board, which is currently composed of eight members. The primary responsibilities of the Board are to provide oversight, strategic guidance, counseling and direction to our management. During the fiscal year ended December 31, 2014, the Board met nine times and no director attended fewer than 75% of the meetings of the Board and Board committees of which the director was a member.
| Item 14. | Principal Accounting Fees and Services. |
Aggregate fees billed by OUM & Co. LLP, an independent registered public accounting firm, during the fiscal years ended December 31, 2014 and 2013 were as follows:
| 46 |
| 2014 | 2013 | |||||||
| Audit Fees | $ | 162,382 | $ | 161,500 | ||||
| Audit-Related Fees | — | — | ||||||
| Tax Fees | 18,455 | 18,990 | ||||||
| All Other Fees | — | — | ||||||
| Total | $ | 180,837 | $ | 180,490 | ||||
Audit Fees—This category includes aggregate fees billed by our independent auditors for the audit of our annual financial statements, audit of management’s assessment and effectiveness of internal controls over financial reporting, review of financial statements included in our quarterly reports on Form 10-Q and services that are normally provided by the auditor in connection with statutory and regulatory filings for those fiscal years.
Audit-Related Fees—This category consists of services by our independent auditors that, including accounting consultations on transaction related matters, are reasonably related to the performance of the audit or review of our financial statements and are not reported above under Audit Fees.
Tax Fees—This category consists of professional services rendered for tax compliance and preparation of our corporate tax returns and other tax advice.
All Other Fees—During the years ended December 31, 2014 and 2013, OUM & Co. LLP did not incur any fees for other professional services.
The audit committee reviewed and approved all audit and non-audit services provided by OUM & Co. LLP and concluded that these services were compatible with maintaining its independence. The audit committee approved the provision of all non-audit services by OUM & Co. LLP. Of the total number of hours expended during OUM & Co. LLP’s engagement to audit our financial statements for the year ended December 31, 2013, none of the hours were attributed to work performed by persons other than permanent, full-time employees of OUM & Co. LLP.
Pre-Approval Policies and Procedures
In accordance with the SEC’s auditor independence rules, the audit committee has established the following policies and procedures by which it approves in advance any audit or permissible non-audit services to be provided to us by our independent auditor.
Prior to the engagement of the independent auditors for any fiscal year’s audit, management submits to the audit committee for approval lists of recurring audit, audit-related, tax and other services expected to be provided by the independent auditors during that fiscal year. The audit committee adopts pre-approval schedules describing the recurring services that it has pre-approved, and is informed on a timely basis, and in any event by the next scheduled meeting, of any such services rendered by the independent auditor and the related fees.
The fees for any services listed in a pre-approval schedule are budgeted, and the audit committee requires the independent auditor and management to report actual fees versus the budget periodically throughout the year. The audit committee will require additional pre-approval if circumstances arise where it becomes necessary to engage the independent auditor for additional services above the amount of fees originally pre-approved. Any audit or non-audit service not listed in a pre-approval schedule must be separately pre-approved by the audit committee on a case-by-case basis.
Every request to adopt or amend a pre-approval schedule or to provide services that are not listed in a pre-approval schedule must include a statement by the independent auditors as to whether, in their view, the request is consistent with the SEC’s rules on auditor independence.
The audit committee will not grant approval for:
| • | any services prohibited by applicable law or by any rule or regulation of the SEC or other regulatory body applicable to us; |
| 47 |
| • | provision by the independent auditors to us of strategic consulting services of the type typically provided by management consulting firms; or |
| • | the retention of the independent auditors in connection with a transaction initially recommended by the independent auditors, the tax treatment of which may not be clear under the Internal Revenue Code and related regulations and which it is reasonable to conclude will be subject to audit procedures during an audit of our financial statements. |
Tax services proposed to be provided by the auditor to any director, officer or employee of Titan who is in an accounting role or financial reporting oversight role must be approved by the audit committee on a case-by-case basis where such services are to be paid for by us, and the audit committee will be informed of any services to be provided to such individuals that are not to be paid for by us.
In determining whether to grant pre-approval of any non-audit services in the “all other” category, the audit committee will consider all relevant facts and circumstances, including the following four basic guidelines:
| • | whether the service creates a mutual or conflicting interest between the auditor and us; |
| • | whether the service places the auditor in the position of auditing his or her own work; |
| • | whether the service results in the auditor acting as management or an employee of our company; and |
| • | whether the service places the auditor in a position of being an advocate for our company. |
PART IV
| Item 15. | Exhibits and Financial Statements Schedules. |
| (a) | 1. Financial Statements |
An index to Financial Statements appears on page F-1.
| 2. Schedules |
All financial statement schedules are omitted because they are not applicable, not required under the instructions or all the information required is set forth in the financial statements or notes thereto.
| 48 |
TITAN PHARMACEUTICALS, INC.
INDEX TO FINANCIAL STATEMENTS
| F-1 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
The Board of Directors and Stockholders of
Titan Pharmaceuticals, Inc.
We have audited the accompanying balance sheets of Titan Pharmaceuticals, Inc. as of December 31, 2014 and 2013, the related statements of operations and comprehensive income (loss), stockholders’ equity (deficit), and cash flows for each of the three years in the period ended December 31, 2014. These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements audited by us present fairly, in all material respects, the financial position of Titan Pharmaceuticals, Inc. at December 31, 2014 and 2013, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2014, in conformity with accounting principles generally accepted in the United States of America.
/s/ OUM & CO. LLP
San Francisco, California
March 31, 2015
| F-2 |
TITAN PHARMACEUTICALS, INC.
| December 31, | ||||||||
| 2014 | 2013 | |||||||
| (in thousands, except share and per share data ) | ||||||||
| Assets | ||||||||
| Current assets: | ||||||||
| Cash | $ | 15,470 | $ | 11,798 | ||||
| Receivables | 3,968 | 4,818 | ||||||
| Prepaid expenses and other current assets | 145 | 204 | ||||||
| Total current assets | 19,583 | 16,820 | ||||||
| Property and equipment, net | 1,268 | 1,603 | ||||||
| Total Assets | $ | 20,851 | $ | 18,423 | ||||
| Liabilities and Stockholders’ Equity (Deficit) | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 4,408 | $ | 5,118 | ||||
| Accrued clinical trials expenses | 254 | 118 | ||||||
| Other accrued liabilities | 329 | 293 | ||||||
| Deferred contract revenue | 1,671 | 5,317 | ||||||
| Total current liabilities | 6,662 | 10,846 | ||||||
| Warrant liability | 5,578 | 1,817 | ||||||
| Total Liabilities | 12,240 | 12,663 | ||||||
| Commitments and contingencies | ||||||||
| Stockholders’ equity: | ||||||||
| Preferred stock, $0.001 par value per share; 5,000,000 shares authorized, none issued and outstanding at December 31, 2014 and 2013. | — | — | ||||||
| Common stock, at amounts paid-in, $0.001 par value per share; 125,000,000 shares authorized, 109,997,533 and 88,794,222 shares issued and outstanding at December 31, 2014 and 2013, respectively. | 289,196 | 284,485 | ||||||
| Additional paid-in capital | 22,235 | 21,692 | ||||||
| Accumulated deficit | (302,820 | ) | (300,417 | ) | ||||
| Total stockholders’ equity | 8,611 | 5,760 | ||||||
| Total Liabilities and Stockholders’ Equity | $ | 20,851 | $ | 18,423 | ||||
See accompanying notes to financial statements.
| F-3 |
TITAN PHARMACEUTICALS, INC.
STATEMENTS OF OPERATIONS AND COMPREHENSIVE INCOME (LOSS)
| Years ended December 31, | ||||||||||||
| 2014 | 2013 | 2012 | ||||||||||
| (in thousands, except per share amount) | ||||||||||||
| Revenue: | ||||||||||||
| License revenue | $ | 3,646 | $ | 9,057 | $ | 2,325 | ||||||
| Royalty revenue | — | 1,424 | 4,750 | |||||||||
| Grant revenue | — | — | 42 | |||||||||
| Total revenue | 3,646 | 10,481 | 7,117 | |||||||||
| Operating expenses: | ||||||||||||
| Research and development | 4,075 | 8,309 | 10,610 | |||||||||
| General and administrative | 3,046 | 3,063 | 4,877 | |||||||||
| Total operating expenses | 7,121 | 11,372 | 15,487 | |||||||||
| Loss from operations | (3,475 | ) | (891 | ) | (8,370 | ) | ||||||
| Other income (expense): | ||||||||||||
| Interest expense, net | — | (1,568 | ) | (4,861 | ) | |||||||
| Other income (expense), net | (11 | ) | 10,433 | (183 | ) | |||||||
| Non-cash gain (loss) on changes in the fair value of warrants | 1,083 | 1,737 | (1,766 | ) | ||||||||
| Other income (expense), net | 1,072 | 10,602 | (6,810 | ) | ||||||||
| Net income (loss) and comprehensive income (loss) applicable to common stockholders | $ | (2,403 | ) | $ | 9,711 | $ | (15,180 | ) | ||||
| Basic net income (loss) per common share | $ | (0.03 | ) | $ | 0.12 | $ | (0.23 | ) | ||||
| Diluted net income (loss) per common share | $ | (0.04 | ) | $ | 0.10 | $ | (0.23 | ) | ||||
| Weighted average shares used in computing basic net income (loss) per common share | 93,814 | 82,099 | 66,509 | |||||||||
| Weighted average shares used in computing diluted net income (loss) per common share | 93,832 | 82,659 | 66,509 | |||||||||
See accompanying notes to financial statements.
| F-4 |
TITAN PHARMACEUTICALS, INC
STATEMENTS OF STOCKHOLDERS’ EQUITY (DEFICIT)
(in thousands)
| Common Stock | Additional Paid-In | Accumulated | Accumulated Other Comprehensive | Total Stockholders’ | ||||||||||||||||||||
| Shares | Amount | Capital | Deficit | Income (Loss) | Equity (Deficit) | |||||||||||||||||||
| Balances at December 31, 2011 | 59,387 | $ | 256,436 | $ | 18,433 | $ | (294,948 | ) | $ | — | $ | (20,079 | ) | |||||||||||
| Net loss | (15,180 | ) | (15,180 | ) | ||||||||||||||||||||
| Issuance of common stock, net of issuance costs | 9,917 | 4,653 | 4,653 | |||||||||||||||||||||
| Issuance of common stock upon exercise of warrants | 5,762 | 4,897 | 4,897 | |||||||||||||||||||||
| Issuance of common stock upon vesting of restricted stock awards, net | 150 | — | ||||||||||||||||||||||
| Compensation related to stock options | 2,581 | 2,581 | ||||||||||||||||||||||
| Balances at December 31, 2012 | 75,216 | 265,986 | 21,014 | (310,128 | ) | — | (23,128 | ) | ||||||||||||||||
| Net income | 9,711 | 9,711 | ||||||||||||||||||||||
| Issuance of common stock, net of issuance costs | 6,250 | 4,925 | 4,925 | |||||||||||||||||||||
| Issuance of common stock upon exercise of options | 75 | 113 | 113 | |||||||||||||||||||||
| Issuance of common stock upon exercise of warrants | 7,253 | 13,461 | 13,461 | |||||||||||||||||||||
| Compensation related to stock options | 678 | 678 | ||||||||||||||||||||||
| Balances at December 31, 2013 | 88,794 | $ | 284,485 | $ | 21,692 | $ | (300,417 | ) | $ | — | $ | 5,760 | ||||||||||||
| Net loss | (2,403 | ) | (2,403 | ) | ||||||||||||||||||||
| Issuance of common stock, net of issuance costs | 21,000 | 4,747 | 4,747 | |||||||||||||||||||||
| Issuance of common stock upon vesting of restricted stock awards, net | 204 | (36 | ) | (36 | ) | |||||||||||||||||||
| Compensation related to stock options | 543 | 543 | ||||||||||||||||||||||
| Balances at December 31, 2014 | 109,998 | $ | 289,196 | $ | 22,235 | $ | (302,820 | ) | $ | — | $ | 8,611 | ||||||||||||
See accompanying notes to financial statements.
| F-5 |
TITAN PHARMACEUTICALS, INC.
| Years ended December 31, | ||||||||||||
| 2014 | 2013 | 2012 | ||||||||||
| (in thousands) | ||||||||||||
| Cash flows from operating activities: | ||||||||||||
| Net income (loss) | $ | (2,403 | ) | $ | 9,711 | $ | (15,180 | ) | ||||
| Adjustments to reconcile net income (loss) to net cash used in operating activities: | ||||||||||||
| Depreciation and amortization | 353 | 107 | 17 | |||||||||
| Non-cash gain on settlement of long-term debt | — | (1,860 | ) | — | ||||||||
| Non-cash gain on termination of royalty purchase agreement | — | (8,962 | ) | — | ||||||||
| Interest on royalty liability | — | — | (347 | ) | ||||||||
| Non-cash (gain) loss on changes in fair value of warrants | (1,083 | ) | (1,737 | ) | 1,766 | |||||||
| Stock-based compensation | 543 | 678 | 2,581 | |||||||||
| Changes in operating assets and liabilities: | ||||||||||||
| Receivables | 850 | (172 | ) | (926 | ) | |||||||
| Prepaid expenses and other assets | 59 | 483 | 149 | |||||||||
| Accounts payable | (710 | ) | 1,351 | (1,022 | ) | |||||||
| Other accrued liabilities | 172 | (340 | ) | 417 | ||||||||
| Deferred contract revenue | (3,646 | ) | (9,058 | ) | 14,375 | |||||||
| Net cash provided by (used in) operating activities | (5,865 | ) | (9,799 | ) | 1,830 | |||||||
| Cash flows from investing activities: | ||||||||||||
| Purchases of furniture and equipment | (18 | ) | (318 | ) | (1,154 | ) | ||||||
| Net cash used in investing activities | (18 | ) | (318 | ) | (1,154 | ) | ||||||
| Cash flows from financing activities: | ||||||||||||
| Proceeds from issuance of common stock from the exercise of stock options | — | 113 | — | |||||||||
| Proceeds from issuance of common stock and warrants, net of issuance costs | 9,591 | 4,925 | 7,516 | |||||||||
| Issuance of common stock from the vesting of restricted shares | (36 | ) | — | — | ||||||||
| Proceeds from the exercise of warrants, net of issuance costs | — | 1,275 | 4,897 | |||||||||
| Payments on long-term debt | — | (2,500 | ) | (393 | ) | |||||||
| Net cash provided by financing activities | 9,555 | 3,813 | 12,020 | |||||||||
| Net increase (decrease) in cash | 3,672 | (6,304 | ) | 12,696 | ||||||||
| Cash at beginning of period | 11,798 | 18,102 | 5,406 | |||||||||
| Cash at end of period | $ | 15,470 | $ | 11,798 | $ | 18,102 | ||||||
| Supplemental disclosure of cash flow information | ||||||||||||
| Interest paid | $ | — | $ | 1,568 | $ | 2,576 | ||||||
| Schedule of non-cash transactions | ||||||||||||
| Settlement of long-term debt | $ | — | $ | 7,500 | $ | — | ||||||
| Fair value of warrants at the time of exercise | $ | — | $ | 4,686 | $ | — | ||||||
See accompanying notes to financial statements.
| F-6 |
TITAN PHARMACEUTICALS, INC.
1. Organization and Summary of Significant Accounting Policies
The Company
We are a specialty pharmaceutical company developing proprietary therapeutics for the treatment of serious medical disorders. Our product development programs utilize our proprietary long-term drug delivery platform, ProNeura®, and focus primarily on innovative treatments for select chronic diseases for which steady state delivery of a drug provides an efficacy and/or safety benefit. We are directly developing our product candidates and also utilize corporate, academic and government partnerships as appropriate. We operate in only one business segment, the development of pharmaceutical products.
The accompanying financial statements have been prepared assuming we will continue as a going concern. At December 31, 2014, we had cash of approximately $15.5 million, which we believe is sufficient to fund our planned operations into the fourth quarter of 2016.
Although Braeburn has commenced the clinical study and is proceeding with plans for completing the clinical study expeditiously, under our December 2012 license agreement with Braeburn, as amended, Braeburn currently has the technical right to terminate the Agreement. If Braeburn were to exercise its right to terminate the Agreement, we would not have sufficient funds available to us to complete the FDA regulatory process and, in the event of ultimate approval, commercialize Probuphine without raising additional capital. If we are unable to complete a debt or equity offering, or otherwise obtain sufficient financing in such event, our business and prospects would be materially adversely impacted. Furthermore, in order to advance our current ProNeura development program for Parkinson’s disease to later stage clinical studies, we will require additional funds, either through payments from Braeburn under the license agreement in the event the Probuphine NDA is ultimately approved or through other financing arrangements, to complete the clinical studies and regulatory approval process necessary to commercialize any additional products we might develop.
Use of Estimates
The preparation of financial statements in conformity with accounting principles generally accepted in the United States requires management to make estimates and assumptions that affect the amounts reported in the financial statements and accompanying notes. Actual results could differ from those estimates.
Stock-Based Compensation
We recognize compensation expense using a fair-value based method, for all stock-based payments including stock options and restricted stock awards and stock issued under an employee stock purchase plan. These standards require companies to estimate the fair value of stock-based payment awards on the date of grant using an option pricing model. See Note 12 “Stock Plans,” for a discussion of our stock-based compensation plans. Our non-cash stock-based compensation expense related to employees and non-employee members of our board of directors totaled $0.5 million, $0.7 million and $2.6 million for the years ended December 31, 2014, 2013 and 2012, respectively.
Warrants Issued in Connection with Equity Financing
We generally account for warrants issued in connection with equity financings as a component of equity, unless there is a deemed possibility that we may have to settle warrants in cash. For warrants issued with deemed possibility of cash settlement, we record the fair value of the issued warrants as a liability at each reporting period and record changes in the estimated fair value as a non-cash gain or loss in the Statements of Operations and Comprehensive Income (Loss).
| F-7 |
Cash, Cash Equivalents and Marketable Securities
Our investment policy emphasizes liquidity and preservation of principal over other portfolio considerations. We select investments that maximize interest income to the extent possible given these two constraints. We satisfy liquidity requirements by investing excess cash in securities with different maturities to match projected cash needs and limit concentration of credit risk by diversifying our investments among a variety of high credit-quality issuers and limit the amount of credit exposure to any one issuer. The estimated fair values have been determined using available market information. We do not use derivative financial instruments in our investment portfolio.
All investments with original maturities of three months or less are considered to be cash equivalents. Marketable securities, consisting primarily of high-grade debt securities including money market funds, U.S. government and corporate notes and bonds, and commercial paper, are classified as available-for-sale at time of purchase and carried at fair value. If the fair value of a security is below its amortized cost and we plan to sell the security before recovering its cost, the impairment is considered to be other-than-temporary. Other-than-temporary declines in fair value of our marketable securities are charged against interest income. We did not have cash equivalents or marketable securities as of December 31, 2014 and 2013 and for any of the periods presented.
Property and Equipment
Property and equipment are recorded at cost and depreciated using the straight-line method over the estimated useful lives of the assets ranging from three to five years. Leasehold improvements are amortized over the shorter of the lease term or the estimated useful life of the assets.
Revenue Recognition
We generate revenue principally from collaborative research and development arrangements, technology licenses, and government grants. Consideration received for revenue arrangements with multiple components is allocated among the separate units of accounting based on their respective selling prices. The selling price for each unit is based on vendor-specific objective evidence, or VSOE, if available, third party evidence if VSOE is not available, or estimated selling price if neither VSOE nor third party evidence is available. The applicable revenue recognition criteria are then applied to each of the units.
Revenue is recognized when the four basic criteria of revenue recognition are met: (1) a contractual agreement exists; (2) transfer of technology has been completed or services have been rendered; (3) the fee is fixed or determinable; and (4) collectibility is reasonably assured. For each source of revenue, we comply with the above revenue recognition criteria in the following manner:
| · | Technology license agreements typically consist of non-refundable upfront license fees, annual minimum access fees or royalty payments. Non-refundable upfront license fees and annual minimum payments received with separable stand-alone values are recognized when the technology is transferred or accessed, provided that the technology transferred or accessed is not dependent on the outcome of our continuing research and development efforts. |
| · | Royalties earned are based on third-party sales of licensed products and are recorded in accordance with contract terms when third-party results are reliably measurable and collectibility is reasonably assured. We no longer recognize royalty income related to the Fanapt royalty payments received from Novartis unless Fanapt sales exceed certain thresholds (see Note 8, “Royalty Liability” for further discussion). |
| · | Government grants, which support our research efforts in specific projects, generally provide for reimbursement of approved costs as defined in the notices of grants. Grant revenue is recognized when associated project costs are incurred. |
| · | Collaborative arrangements typically consist of non-refundable and/or exclusive technology access fees, cost reimbursements for specific research and development spending, and various milestone and future product royalty payments. If the delivered technology does not have stand-alone value, the amount of revenue allocable to the delivered technology is deferred. Non-refundable upfront fees with stand-alone value that are not dependent on future performance under these agreements are recognized as revenue when received, and are deferred if we have continuing performance obligations and have no evidence of fair value of those obligations. Cost reimbursements for research and development spending are recognized when the related costs are incurred and when collections are reasonably expected. Payments received related to substantive, performance-based “at-risk” milestones are recognized as revenue upon achievement of the clinical success or regulatory event specified in the underlying contracts, which represent the culmination of the earnings process. Amounts received in advance are recorded as deferred revenue until the technology is transferred, costs are incurred, or a milestone is reached. |
| F-8 |
Research and Development Costs and Related Accrual
Research and development expenses include internal and external costs. Internal costs include salaries and employment related expenses, facility costs, administrative expenses and allocations of corporate costs. External expenses consist of costs associated with outsourced clinical research organization activities, sponsored research studies, product registration, patent application and prosecution, and investigator sponsored trials. We also record accruals for estimated ongoing clinical trial costs. Clinical trial costs represent costs incurred by CROs, and clinical sites. These costs are recorded as a component of research and development expenses. Under our agreements, progress payments are typically made to investigators, clinical sites and CROs. We analyze the progress of the clinical trials, including levels of patient enrollment, invoices received and contracted costs when evaluating the adequacy of accrued liabilities. Significant judgments and estimates must be made and used in determining the accrued balance in any accounting period. Actual results could differ from those estimates under different assumptions. Revisions are charged to expense in the period in which the facts that give rise to the revision become known.
Net Income (Loss) Per Share
Basic net income (loss) per share excludes the effect of dilution and is computed by dividing net income (loss) by the weighted-average number of shares outstanding for the period. Diluted net income (loss) per share reflects the potential dilution that could occur if securities or other contracts to issue shares were exercised into shares. In calculating diluted net income (loss) per share, the numerator is adjusted for the change in the fair value of the warrant liability (only if dilutive) and the denominator is increased to include the number of potentially dilutive common shares assumed to be outstanding during the period using the treasury stock method.
The following table sets forth the reconciliation of the numerator and denominator used in the computation of basic and diluted net income (loss) per common share for the years ended December 31, 2014, 2013 and 2012:
| Years ended December 31, | ||||||||||||
| (in thousands, except per share amounts) | 2014 | 2013 | 2012 | |||||||||
| Numerator: | ||||||||||||
| Net income (loss) used for basic earnings per share | $ | (2,403 | ) | $ | 9,711 | $ | (15,180 | ) | ||||
| Less change in fair value of warrant liability | 1,083 | 1,737 | — | |||||||||
| Net (loss) income used for diluted earnings per share | $ | (3,486 | ) | $ | 7,974 | $ | (15,180 | ) | ||||
| Denominator: | ||||||||||||
| Basic weighted-average outstanding common shares | 93,814 | 82,099 | 66,509 | |||||||||
| Effect of dilutive potential common shares resulting from options | 18 | 493 | — | |||||||||
| Effect of dilutive potential common shares resulting from warrants | — | 67 | — | |||||||||
| Weighted-average shares outstanding—diluted | 93,832 | 82,659 | 66,509 | |||||||||
| Net income (loss) per common share: | ||||||||||||
| Basic | $ | (0.03 | ) | $ | 0.12 | $ | (0.23 | ) | ||||
| Diluted | $ | (0.04 | ) | $ | 0.10 | $ | (0.23 | ) | ||||
| F-9 |
The table below presents common shares underlying stock options and warrants that are excluded from the calculation of the weighted average number of shares of common stock outstanding used for the calculation of diluted net income (loss) per common share. These are excluded from the calculation due to their anti-dilutive effect for the years ended December 31, 2014, 2013 and 2012:
| Years ended December 31, | ||||||||||||
| (in thousands) | 2014 | 2013 | 2012 | |||||||||
| Weighted-average anti-dilutive common shares resulting from options and awards | 6,894 | 2,628 | 4,213 | |||||||||
| Weighted-average anti-dilutive common shares resulting from warrants | 2,333 | 675 | 3,011 | |||||||||
| 9,227 | 3,303 | 7,224 | ||||||||||
Comprehensive Income (Loss)
Comprehensive income and loss for the periods presented is comprised solely of our net income and loss. Comprehensive loss for the year ended December 31, 2014 was $2.4 million. Comprehensive income for the year ended December 31, 2013 was $9.7 million. Comprehensive loss for the year ended December 31, 2012 was $15.2 million.
Recent Accounting Pronouncements
In July 2013, the FASB issued ASU No. 2013-11, Income Taxes (Topic 740): Presentation of an Unrecognized Tax Benefit When a Net Operating Loss Carryforward, a Similar Tax Loss, or a Tax Credit Carryforward Exists, providing guidance on the presentation of unrecognized tax benefits in the financial statements as either a reduction to a deferred tax asset or a liability to better reflect the manner in which an entity would settle at the reporting date any additional income taxes that would result from the disallowance of a tax position when net operating loss carryforwards, similar tax losses or tax credit carryforwards exist. The amendments in this ASU do not require new recurring disclosures. The amendments in this ASU are effective for fiscal years, and interim periods within those years, beginning after December 15, 2013. The amendments in this ASU should be applied prospectively to all unrecognized tax benefits that exist at the effective date. The adoption of the amendments in this ASU did not have a significant impact on our financial statements.
In May 2014, the FASB issued ASU No. 2014-09, Revenue from Contracts with Customers (“ASU 2014-09”), which supersedes nearly all existing revenue recognition guidance under U.S. GAAP. The core principle of ASU 2014-09 is to recognize revenues when promised goods or services are transferred to customers in an amount that reflects the consideration to which an entity expects to be entitled for those goods or services. ASU 2014-09 defines a five step process to achieve this core principle and, in doing so, more judgment and estimates may be required within the revenue recognition process than are required under existing U.S. GAAP.
The standard is effective for annual periods beginning after December 15, 2016, and interim periods therein, using either of the following transition methods: (i) a full retrospective approach reflecting the application of the standard in each prior reporting period with the option to elect certain practical expedients, or (ii) a retrospective approach with the cumulative effect of initially adopting ASU 2014-09 recognized at the date of adoption (which includes additional footnote disclosures). We are currently evaluating the impact of our pending adoption of ASU 2014-09 on our financial statements and have not yet determined the method by which we will adopt the standard.
In June 2014, the FASB issued ASU No. 2014-12, Accounting for Share-Based Payments When the Terms of an Award Provide That a Performance Target Could Be Achieved after the Requisite Service Period (“ASU 2014-12”). The standard provides guidance that a performance target that affects vesting of a share-based payment and that could be achieved after the requisite service condition is a performance condition. As a result, the target is not reflected in the estimation of the award’s grant date fair value. Compensation cost for such award would be recognized over the required service period, if it is probable that the performance condition will be achieved. ASU 2014-12 is effective for annual reporting periods beginning after December 15, 2015. Early adoption is permitted. The guidance should be applied on a prospective basis to awards that are granted or modified on or after the effective date. Companies also have the option to apply the amendments on a modified retrospective basis for performance targets outstanding on or after the beginning of the first annual period presented as of the adoption date. We are currently evaluating the impact of our pending adoption of ASU 2014-12 on our financial statements and the method by which we will adopt the standard.
| F-10 |
Subsequent Events
We have evaluated events that have occurred subsequent to December 31, 2014 and through the date that the financial statements are issued.
Fair Value Measurements
We measure the fair value of financial assets and liabilities based on authoritative guidance which defines fair value, establishes a framework consisting of three levels for measuring fair value, and requires disclosures about fair value measurements. Fair value is defined as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. There are three levels of inputs that may be used to measure fair value:
Level 1 – quoted prices in active markets for identical assets or liabilities;
Level 2 – quoted prices for similar assets and liabilities in active markets or inputs that are observable;
Level 3 – inputs that are unobservable (for example cash flow modeling inputs based on assumptions).
Financial instruments, including receivables, accounts payable and accrued liabilities are carried at cost, which we believe approximates fair value due to the short-term nature of these instruments. Our warrant liabilities are classified within level 3 of the fair value hierarchy because the value is calculated using significant judgment based on our own assumptions in the valuation of these liabilities.
During the years ended December 31, 2014 and 2013, as a result of the fair value adjustment of the warrant liabilities, we recorded non-cash gains on decreases in the fair value of $1,083,000 and $1,737,000, respectively, in our statements of operations and comprehensive income (loss). See Note 9, “Warrant Liability” for further discussion on the calculation of the fair value of the warrant liability.
The following table rolls forward the fair value of the Company’s warrant liability, the fair value of which is determined by Level 3 inputs for the years ended December 31, 2014 and 2013 (in thousands):
| December 31, | ||||||||
| 2014 | 2013 | |||||||
| Fair value, beginning of period | $ | 1,817 | $ | 8,240 | ||||
| Issuance of warrants | 4,844 | — | ||||||
| Exercise of warrants | — | (4,686 | ) | |||||
| Change in fair value | (1,083 | ) | (1,737 | ) | ||||
| Fair value, end of period | $ | 5,578 | $ | 1,817 | ||||
2. Property and Equipment
Property and equipment consisted of the following at December 31, 2014 and 2013 (in thousands):
| 2014 | 2013 | |||||||
| Furniture and office equipment | $ | 388 | $ | 388 | ||||
| Leasehold improvements | 408 | 408 | ||||||
| Laboratory equipment | 2,333 | 2,318 | ||||||
| Computer equipment | 1,046 | 1,043 | ||||||
| 4,175 | 4,157 | |||||||
| Less accumulated depreciation and amortization | (2,907 | ) | (2,554 | ) | ||||
| Property and equipment, net | $ | 1,268 | $ | 1,603 | ||||
| F-11 |
Depreciation and amortization expense was $353,000, $107,000 and $17,000 for the years ended December 31, 2014, 2013 and 2012, respectively.
3. Research and License Agreements
We have entered into various agreements with research institutions, universities, clinical research organizations and other entities for the performance of research and development activities and for the acquisition of licenses related to those activities. Expenses under these agreements totaled approximately $3,000 in the years ended December 31, 2014, 2013 and 2012.
We have no annual payment requirements to maintain our current licenses after 2015. Certain licenses provide for the payment of royalties by us on future product sales, if any. In addition, in order to maintain these licenses and other rights during product development, we must comply with various conditions including the payment of patent-related costs.
4. Agreement with Sanofi-Aventis SA
In 1997, we entered into an exclusive license agreement with Sanofi-Aventis. The agreement gave us a worldwide license to the patent rights and know-how related to the antipsychotic agent iloperidone, including the ability to develop, use, sublicense, manufacture and sell products and processes claimed in the patent rights. We are required to make additional benchmark payments as specific milestones are met. Upon commercialization of the product, the license agreement provides that we will pay royalties based on net sales.
5. Iloperidone Sublicense to Novartis Pharma AG
We are party to an agreement with Novartis, which, as amended, granted Novartis a worldwide sublicense to iloperidone (Fanapt®) in exchange for tiered royalties on net sales ranging from 8% to 10% and assumption of responsibility for all clinical development, registration, manufacturing and marketing of the product. Novartis had the right to commercialize Fanapt in the United States and Canada. In December 2014, Novartis transferred all rights to commercialize Fanapt in the United States and Canada to Vanda Pharmaceuticals, Inc. Our rights under the agreements have not changed. Pursuant to agreements entered into during 2011, we sold substantially all of our remaining future royalties on the sales of Fanapt® to Deerfield, and accordingly the future royalty payments owed to us by Novartis will continue to be transmitted to Deerfield upon receipt from Novartis per the terms of the agreement with Deerfield. See Note 8, “Royalty Liability” for further discussion of our royalty liabilities.
6. Braeburn License
In December 2012, we entered into the Agreement with Braeburn granting Braeburn exclusive commercialization rights to Probuphine in the United States and its territories, including Puerto Rico, and Canada. As part of the Agreement, we received a non-refundable up-front license fee of $15.75 million (approximately $15.0 million, net of expenses), and would have received $45.0 million upon approval by the FDA of the NDA as well as up to an additional $130.0 million upon achievement of specified sales milestones and up to $35.0 million in regulatory milestones for additional indications, including chronic pain. We would have received tiered royalties on net sales of Probuphine ranging from the mid-teens to the low twenties.
On May 28, 2013, we entered into the Amendment to the Agreement primarily to modify certain of the termination provisions of the Agreement. The Amendment gives Braeburn the right to terminate the Agreement in the event that (A) after May 28, 2013, based on written or oral communications from or with the FDA, Braeburn reasonably determines either that the FDA will require significant development to be performed before approval of the Probuphine™ NDA can be given, such as, but not limited to, one or more additional controlled clinical studies with a clinical efficacy endpoint, or substantial post-approval commitments that may materially impact the product’s financial returns or that the FDA will require one or more changes in the proposed label, which change(s) Braeburn reasonably determines will materially reduce the authorized prescribed patient base, or (B) the NDA has not been approved by the FDA on or before June 30, 2014. The Amendment also provides that we will share in legal and consulting expenses in excess of a specified amount prior to approval of the NDA.
| F-12 |
On July 2, 2013, we entered into the Second Amendment to the Agreement primarily to establish and provide the parameters for a committee comprised of representatives of Titan and Braeburn responsible for and with the authority to make all decisions regarding the development and implementation of a strategic plan to seek approval from the FDA of Probuphine® for subdermal use in the maintenance treatment of adult patients with opioid dependence, including development of the strategy for all written and oral communications with the FDA. The Second Amendment also makes Braeburn the primary contact for FDA communications regarding the Probuphine NDA.
On November 12, 2013, we entered into the stock purchase agreement pursuant to which Braeburn made a $5 million equity investment in our company and the Third Amendment primarily to modify the amount and timing of the approval and sales milestone payments payable under the Agreement. Under the Third Amendment, we are entitled to receive a $15 million payment upon FDA approval of the NDA and royalties on net sales of Probuphine ranging in percentage from the mid-teens to the low twenties. The agreement also provides for up to $165 million in sales milestones and $35 million in regulatory milestones. In addition, we are entitled to receive a low single digit royalty on sales by Braeburn, if any, of other continuous delivery treatments for opioid dependence as defined in the Third Amendment and can elect to receive a low single digit royalty on sales by Braeburn, if any, of other products in the addiction market in exchange for a similar reduction in our royalties on Probuphine.
We have evaluated the revenue components of the agreement, which includes multiple elements, to determine whether the components of the arrangement represent separate units of accounting. We have determined that the non-refundable, up-front license fee of $15.75 million (approximately $15.0 million, net of expenses) and our costs up to the PDUFA date to be one deliverable which will be accounted for as a single unit of accounting. This amount will be recognized on a straight-line basis over the estimated period during which we expect to meet the contract deliverables. Based on our understanding of subsequent steps to be performed following the PDUFA date related to the completion of the transition of production and supply services to Braeburn, we estimated the revenue recognition period from the up-front payment to be approximately 12 months from the date of the agreement. Accordingly, we recognized revenue for the up-front payment ratably from December 14, 2012, the date of the agreement, through March 31, 2013 at an amount equal to approximately $1.25 million per month. Following the receipt of the CRL in April 2013, we estimated the revenue recognition period for the up-front payment would be approximately 18 months from the date of the agreement. Accordingly, we recognized the remaining revenue from the up-front payment ratably from April 1, 2013 through September 30, 2013 at an amount equal to approximately $733,000 per month. Following our meeting with the FDA in November 2013 and subsequent discussions in which an agreement in principle with respect to a path forward has been reached with the FDA, we estimate the revenue recognition period for the up-front payment to be approximately 30 months from the date of the agreement. Accordingly, we will recognize the remaining revenue from the up-front payment ratably from September 30, 2013 at an amount equal to approximately $304,000 per month. As of December 31, 2014, we have recognized approximately $13.3 million in license revenue and recorded deferred revenues of $1.7 million related to the up-front payment. Internal and external research and development costs related to this product will be expensed in the period incurred.
Under the Agreement, we will receive a $15.0 million milestone payment from Braeburn within 10 days following the achievement of FDA approval of the product NDA. As such, upon receipt of FDA approval our obligation will be fulfilled. As the milestone payment relates solely to past performance, i.e. FDA approval, we will recognize the $15.0 million regulatory milestone payment from Braeburn on the date of achievement of FDA approval in accordance with the milestone method of revenue recognition. Following FDA approval, we will be reimbursed by Braeburn for any development services and activities performed by us at Braeburn’s request.
The Agreement also provides for a development committee. The duties of the development committee are to periodically report to each other, exchange information, and confer with and review the clinical development of the product and matters pertaining to regulatory approval. The development committee has no authority to approve or direct either party to take action, approve or withhold approval for any plan, budget, timeline or strategies, amend, modify or waive compliance with the Agreement, create new obligations or alter, increase or expand, or waive compliance with the Agreement, create new obligations not specified in the Agreement, or alter, increase or expand, or waive compliance by a party with obligations under the Agreement. The development committee can be disbanded upon mutual agreement of the parties and shall automatically disband six years after the NDA transfer date. Based on the above, we have determined that participation in the development committee is perfunctory and inconsequential, and is not considered a separate deliverable in the Agreement.
| F-13 |
7. Commitments and Contingencies
Financing Agreements
On March 15, 2011, we entered into several agreements pursuant to which Deerfield agreed to provide $20.0 million in funding to us. Funding occurred on April 5, 2011 and we used approximately $7.6 million of proceeds from the Deerfield funding to repay a prior lender in full, including required final payments aggregating $480,000. Pursuant to the terms of a facility agreement, we issued Deerfield promissory notes in the aggregate principal amount of $20.0 million. The long-term debt bears interest at 8.5% per annum, payable quarterly, and was originally repayable over five years, with 10% of the principal amount due on the first anniversary, 15% due on the second anniversary, and 25% due on each of the next three anniversaries. We paid Deerfield a facility fee of $0.5 million. The long-term debt is secured by our assets and has a provision for pre-payment. Deerfield has the right to have the long-term debt repaid at 110% of the principal amount in the event we complete a major transaction, which includes, but was not limited to, a merger or sale of our company or the sale of Probuphine. In connection with the facility agreement, we issued Deerfield six-year warrants to purchase 6,000,000 shares of our common stock at an exercise price of $1.57 per share (See Note 9, “Warrant Liability” for further discussion). As a result of our April 2012 subscription agreements, and pursuant to the terms of the Deerfield Warrants, the exercise price of the Deerfield Warrants was adjusted to $1.25 per share. (see Note 11, “Stockholders’ Equity (Deficit)” for further discussion). We also entered into a royalty agreement with Deerfield in exchange for $3.0 million (see Note 8, “Royalty Liability” for further discussion).
We recorded the promissory notes with an aggregate principal amount of $20.0 million at its face value less a note discount consisting of (i) $3.0 million cash discount, (ii) a $500,000 loan fee, and (iii) the $5.5 million fair value of the associated warrants. The note discount totaling $9.0 million was amortized using the interest method.
On November 14, 2011, we entered into several agreements with Deerfield pursuant to which we agreed to pay a substantial portion of the remaining future royalties on the sales of Fanapt to Deerfield in exchange for $5.0 million in cash that was recorded as royalty liability (see Note 8, “Royalty Liability” for further discussion), a $10.0 million reduction in the principal amount owed to Deerfield under the existing facility agreement and a revised principal repayment schedule of $2.5 million per year for four years commencing in April 2013 to retire the remaining long-term debt of $10.0 million. We evaluated the November 2011 principal reduction and other amendments to the $20.0 million facility agreement and determined that the modifications should be accounted for as a troubled debt restructuring on a prospective basis. As a result, we recognized the difference between the carrying value of the long-term debt and the total required future principal and interest payments as interest expense over the remaining term using the interest method.
On February 6, 2013, the facility agreement was amended to provide that the exercise price of the Deerfield Warrants could be satisfied through a reduction in the principal amount of our outstanding indebtedness to Deerfield. In February and March 2013, Deerfield exercised all of the Deerfield Warrants resulting in a reduction of our outstanding indebtedness to Deerfield of $7.5 million and, accordingly, cancellation of our obligation to make the 2014, 2015 and 2016 installment payments under the Facility Agreement. This resulted in a gain of $1.9 million which was recorded in Other Income (Expense). On April 1, 2013, we made the final principal payment of $2.5 million under the facility agreement.
Lease Commitments
We lease facilities under operating leases that expire at various dates through June 2016. Rent expense was $209,000, $210,000, and $203,000 for years ended December 31, 2014, 2013, and 2012, respectively.
The following is a schedule of future minimum lease payments at December 31, 2014 (in thousands):
| 2015 | $ | 211 | ||
| 2016 and thereafter | 106 | |||
| $ | 317 |
| F-14 |
Legal Proceedings
There are no ongoing legal proceedings against our company.
8. Royalty Liability
On March 15, 2011, under the royalty agreement with Deerfield, in exchange for $3.0 million that was recorded as royalty liability, we agreed to pay Deerfield 2.5% of the net sales of Fanapt, constituting a portion of the royalty revenue that we are entitled to under our sublicense agreement with Novartis. The agreements with Deerfield also provided us with the option to repurchase the royalty rights for $40.0 million.
The $3.0 million received under the royalty agreement was recorded as a royalty liability in accordance with the appropriate accounting guidance as the related agreement includes a provision which allowed us to repurchase the royalty rights from Deerfield through a payment of a lump sum. Interest on the royalty liability was recognized using the interest method based on the estimated future royalties expected to be paid under the Royalty Agreement.
Under the November 14, 2011 amended and restated royalty agreement, in exchange for an additional $5.0 million royalty liability, Deerfield is entitled to our portion of the royalties on Fanapt (5.5% to 7.5% of net sales, net of the 2.5% previously agreed to have been provided to Deerfield) up to specified threshold levels of net sales of Fanapt and 40% of the royalties above the threshold level. We retain 60% of the royalties on net sales of Fanapt above the threshold levels. The $5.0 million received was recorded as a royalty liability in accordance with the appropriate accounting guidance as the related agreement included a provision which allowed us to repurchase the royalty rights from Deerfield through a payment of a lump sum. Interest on this royalty obligation was recognized using the interest method based on the estimated future royalties expected to be paid under the royalty agreement.
On March 28, 2013, we amended the agreements with Deerfield terminating our option to repurchase the royalty rights. As a result, we recognized a gain on the extinguishment of the royalty liability of approximately $9.0 million, which was recorded in other income, because we are no longer required to account for it as a liability. Additionally, we will no longer recognize royalty income related to the Fanapt royalty payments received from Novartis unless Fanapt sales exceed certain thresholds.
9. Warrant Liability
On March 15, 2011, in connection with the facility agreement, we issued Deerfield six-year warrants to purchase 6,000,000 shares of our common stock at an initial exercise price of $1.57 per share. As a result of our April 2012 sale of equity, and pursuant to the terms of the Deerfield Warrants, the exercise price of the Deerfield Warrants was adjusted to $1.25 per share. The Deerfield Warrants contain a provision where the warrant holder has the option to receive cash, equal to the Black-Scholes fair value of the remaining unexercised portion of the warrant, as cash settlement in the event that there is a fundamental transaction (contractually defined to include various merger, acquisition or stock transfer activities). Due to this provision, ASC 480, Distinguishing Liabilities from Equity requires that these warrants be classified as liabilities. The fair values of these warrants have been determined using the Binomial Lattice (“Lattice”) valuation model, and the changes in the fair value are recorded in the Statements of Operations and Comprehensive Income (Loss). The Lattice model provides for assumptions regarding volatility and risk-free interest rates within the total period to maturity.
On February 6, 2013, the facility agreement was amended to provide that the exercise price of the Deerfield Warrants could be satisfied through a reduction in the principal amount of our outstanding indebtedness to Deerfield. In February and March 2013, Deerfield exercised all of the Deerfield Warrants resulting in a $7.5 million reduction in the amount owed to Deerfield.
On April 9, 2012, in connection with subscription agreements with certain institutional investors for the purchase and sale of 6,517,648 shares of our common stock, we issued (i) six-year warrants (“Series A Warrants”) to purchase 6,517,648 shares of common stock at an exercise price of $1.15 per share and (ii) six-month warrants (“Series B Warrants”) to purchase 6,517,648 shares of common stock at an exercise price of $0.85 per share. As a result of our public offering in October 2014 and anti-dilution provisions contained in the outstanding Series A warrants, the exercise price of such warrants was reduced from $1.15 to $0.89 per share. The Series A Warrants and Series B Warrants contain a provision where the warrant holder has the option to receive cash, equal to the Black Scholes fair value of the remaining unexercised portion of the warrant, as cash settlement in the event that there is a fundamental transaction (contractually defined to include various merger, acquisition or stock transfer activities). Due to this provision, ASC 480, Distinguishing Liabilities from Equity requires that these warrants be classified as liabilities. The fair values of these warrants have been determined using the Lattice valuation model, and the changes in the fair value are recorded in the Statements of Operations and Comprehensive Income (Loss). The Lattice model provides for assumptions regarding volatility and risk-free interest rates within the total period to maturity.
| F-15 |
During the year ended December 31, 2012, Series B Warrants to purchase 5,761,765 shares of common stock were exercised at a price of $0.85 per share. The remaining Series B Warrants to purchase 755,883 shares of common stock expired in October 2012.
During the year ended December 31, 2013, Series A Warrants to purchase 1,109,010 shares of common stock were exercised resulting in gross proceeds of approximately $1,275,000. The remaining Series A Warrants to purchase 5,408,638 shares of common stock will expire in April 2018.
The key assumptions used to value the Series A Warrants were as follows:
| Assumption | December 31, 2014 | |||
| Expected price volatility | 90 | % | ||
| Expected term (in years) | 3.27 | |||
| Risk-free interest rate | 1.18 | % | ||
| Dividend yield | 0.00 | % | ||
| Weighted-average fair value of warrants | $ | 0.16 | ||
In October 2014, we completed an underwritten public offering of 21,000,000 units at an offering price of $0.50 per unit, with each unit consisting of one share of common stock and 0.75 of a warrant (“Class A Warrant”), each full warrant to purchase one share of common stock at an exercise price of $0.60 per share. The Class A Warrants will entitle the holders thereof to purchase an aggregate of 15,750,000 shares of our common stock at an initial exercise price of $0.60 per share of common stock. The Class A Warrants will be exercisable beginning on the later of (i) one year and one day from the date of issuance and (ii) the date our stockholders approve either an increase in the number of our authorized shares of common stock or a reverse stock split, in either case in an amount sufficient to permit the exercise in full of the Class A Warrants and will expire on the fifth anniversary of the date they first become exercisable.
We agreed to hold a stockholders meeting no later than March 31, 2015 in order to seek stockholder approval for an amendment to our certificate of incorporation to either (i) increase the number of shares of common stock we are authorized to issue or (ii) effect a reverse split of the common stock, in either case in an amount sufficient to permit the exercise in full of the Class A Warrants in accordance with their terms. In the event that we are unable to effect an increase in our authorized shares of common stock or effect a reverse split of our common stock prior to October 9, 2015, we will be required to pay liquidated damages in the aggregate amount of $2,500,000. In February 2015, the Class A Warrants were amended to extend the date of the required stockholders’ meeting to August 31, 2015 (see Note 15, “Subsequent Events” for further discussion).
We also agreed to issue to the underwriter warrants to purchase 630,000 shares of common stock (“Underwriter Warrant”). The underwriter’s warrants have an exercise price per share of $0.60 and may be exercised on a cashless basis. The underwriter’s warrants are not redeemable by us. The underwriter’s warrants are substantially the same form as the Class A Warrants included in the units except that the underwriter’s warrants do not include certain liquidated damages rights contained in the Class A Warrants and will expire on the fifth anniversary of the date of effectiveness of the registration statement.
| F-16 |
The Class A Warrants and Underwriter Warrants contain a provision where the warrant holder has the option to receive cash, equal to the fair value of the remaining unexercised portion of the warrant, as cash settlement in the event that there is a fundamental transaction (contractually defined to include various merger, acquisition or stock transfer activities). Due to this provision, ASC 480, Distinguishing Liabilities from Equity requires that these warrants be classified as liabilities. The fair values of these warrants have been determined using the Binomial Lattice (“Lattice”) valuation model, and the changes in the fair value are recorded in the Statements of Operations and Comprehensive Income (Loss). The Lattice model provides for assumptions regarding volatility and risk-free interest rates within the total period to maturity.
The key assumptions used to value the Class A Warrants were as follows:
| Assumption | December 31, 2014 | |||
| Expected price volatility | 90 | % | ||
| Expected term (in years) | 5.8 | |||
| Risk-free interest rate | 1.73 | % | ||
| Dividend yield | 0.00 | % | ||
| Weighted-average fair value of warrants | $ | 0.29 | ||
The key assumptions used to value the Underwriter Warrants were as follows:
| Assumption | December 31, 2014 | |||
| Expected price volatility | 90 | % | ||
| Expected term (in years) | 4.8 | |||
| Risk-free interest rate | 1.59 | % | ||
| Dividend yield | 0.00 | % | ||
| Weighted-average fair value of warrants | $ | 0.24 | ||
10. Guarantees and Indemnifications
As permitted under Delaware law and in accordance with our Bylaws, we indemnify our officers and directors for certain events or occurrences while the officer or director is or was serving at our request in such capacity. The term of the indemnification period is for the officer’s or director’s lifetime. The maximum amount of potential future indemnification is unlimited; however, we have a director and officer insurance policy that limits our exposure and may enable us to recover a portion of any future amounts paid. We believe the fair value of these indemnification agreements is minimal. Accordingly, we have not recorded any liabilities for these agreements as of December 31, 2014.
In the normal course of business, we have commitments to make certain milestone payments to various clinical research organizations in connection with our clinical trial activities. Payments are contingent upon the achievement of specific milestones or events as defined in the agreements, and we have made appropriate accruals in our financial statements for those milestones that were achieved as of December 31, 2014. We also provide indemnifications of varying scope to our CROs and investigators against claims made by third parties arising from the use of our products and processes in clinical trials. Historically, costs related to these indemnification provisions were immaterial. We also maintain various liability insurance policies that limit our exposure. We are unable to estimate the maximum potential impact of these indemnification provisions on our future results of operations.
11. Stockholders’ Equity (Deficit)
Common Stock
In October 2014, we completed an underwritten public offering of 21,000,000 units at an offering price of $0.50 per unit, with each unit consisting of one share of common stock and 0.75 of a warrant, each full warrant to purchase one share of common stock at an exercise price of $0.60 per share. Net proceeds were approximately $9.6 million after deducting underwriting discounts, commissions and other related expenses. As a result of the public offering, and pursuant to the terms of the existing Series A Warrants, the exercise price of the Series A Warrants (See Note 9, “Warrant Liability” for further discussion) was adjusted to $0.89 per share.
| F-17 |
In November 2013, we entered into a stock purchase agreement with Braeburn pursuant to which we sold 6,250,000 shares of our common stock for an aggregate purchase price of $5.0 million, or $0.80 per share.
In April 2013, 144,499 shares of common stock were issued to a former lender upon the cashless net exercise of 287,356 warrants in accordance with the terms of the warrants.
In January and March 2013, Series A Warrants to purchase 1,109,010 shares of common stock were exercised resulting in gross proceeds of approximately $1,275,000.
On February 6, 2013, the facility agreement with Deerfield was amended to provide that the exercise price of the Deerfield Warrants could be satisfied through a reduction in the principal amount of our outstanding indebtedness to Deerfield. In February and March 2013, Deerfield exercised the 6,000,000 Deerfield Warrants resulting in a $7.5 million reduction in the amount owed to Deerfield.
In October 2012, Series B Warrants to purchase 4,627,941 shares of common stock were exercised resulting in gross proceeds of approximately $3,934,000.
In September 2012, Series B Warrants to purchase 1,133,824 shares of common stock were exercised resulting in gross proceeds of approximately $964,000.
In September 2012, we entered into a stock purchase and option agreement with an affiliate of Braeburn pursuant to which we sold 3,400,000 shares of our common stock for an aggregate purchase price of $4.25 million, or $1.25 per share, and agreed to an exclusive option period for execution of the proposed license agreement. The $1.7 million premium, or $0.50 per share, has been allocated to the fair value of the option agreement and was recorded as license revenue in 2012.
In April 2012, we entered into subscription agreements with certain institutional investors for the purchase and sale, in a registered direct offering, of (i) 6,517,648 shares of our common stock, (ii) 6,517,648 Series A Warrants and (iii) 6,517,648 Series B Warrants for gross proceeds of $5,540,000 (the “Offering”). As a result of the Offering, and pursuant to the terms of the Deerfield Warrants, the exercise price of the Deerfield Warrants (See Note 9, “Warrant Liability” for further discussion) was adjusted to $1.25 per share.
We recorded the gross proceeds from the Offering, net of (i) issuance costs of $0.5 million and (ii) the fair value of the Warrants of $2.9 million (see Note 9, “Warrant Liability”), as common stock paid-in in the Balance Sheets.
As of December 31, 2014, warrants to purchase shares of common stock consisted of the following (in thousands, except per share price):
| Date Issued | Expiration Date | Exercise Price | Outstanding at December 31, 2014 |
|||||||
| 04/13/2012 | 04/13/2018 | $ | 0.89 | 5,409 | ||||||
| 10/08/2014 | 10/08/2019 | $ | 0.60 | 630 | ||||||
| 10/08/2014 | 10/08/2020 | $ | 0.60 | 15,750 | ||||||
| 21,789 | ||||||||||
Shares Reserved for Future Issuance
As of December 31, 2014, shares of common stock reserved by us for future issuance consisted of the following (in thousands):
| Stock options outstanding | 6,979 | |||
| Shares issuable upon the exercise of warrants | 5,409 | |||
| 12,388 |
| F-18 |
12. Stock Plans
On February 11, 2014, our Board adopted the 2014 Incentive Plan, or the 2014 Plan, pursuant to which 2,500,000 shares of our common stock were authorized for issuance to employees, directors, officers, consultants and advisors. On December 31, 2014, restricted stock awards and options to purchase 683,500 shares of our common stock were outstanding under the 2014 Plan.
In July 2002, we adopted the 2002 Stock Incentive Plan (“2002 Plan”). The 2002 Plan assumed the options which remained available for grant under our option plans previously approved by stockholders. Under the 2002 Plan and predecessor plans, a total of 7.4 million shares of our common stock were authorized for issuance to employees, officers, directors, consultants, and advisers. In August 2005, we adopted an amendment to the 2002 Stock Incentive Plan (“2002 Plan”) to (i) permit the issuance of shares of restricted stock and stock appreciation rights to participants under the 2002 Plan, and (ii) increase the number of shares issuable pursuant to grants under the 2002 Plan from 2,000,000 to 3,000,000. Options granted under the 2002 Plan and predecessor plans may either be incentive stock options within the meaning of Section 422 of the Internal Revenue Code and/or options that do not qualify as incentive stock options; however, only employees are eligible to receive incentive stock options. Options granted under the option plans generally expire no later than ten years from the date of grant. Option vesting schedule and exercise price are determined at time of grant by the board of directors or compensation committee. Historically, the exercise prices of options granted under the 2002 Plan were 100% of the fair market value of our common stock on the date of grant. The 2002 Plan expired by its terms in July 2012. On December 31, 2014, options to purchase an aggregate of 3,808,553 shares of our common stock were outstanding under the 2002 Plan.
In August 2001, we adopted the 2001 Employee Non-Qualified Stock Option Plan (“2001 NQ Plan”) pursuant to which 1,750,000 shares of common stock were authorized for issuance for option grants to employees and consultants who are not officers or directors of Titan. Options granted under the option plans generally expire no later than ten years from the date of grant. Option vesting schedule and exercise price were determined at time of grant by the board of directors or compensation committee. Historically, the exercise prices of options granted under the 2001 NQ Plan were 100% of the fair market value of our common stock on the date of grant. The 2001 Stock Option Plan expired by its terms in August 2011. On December 31, 2014, options to purchase an aggregate of 1,124,000 shares of our common stock were outstanding under the 2001 NQ Plan.
Activity under our stock plans, as well as non-plan activity, is summarized below (shares in thousands):
| Shares or Awards Available For Grant | Number of Options and Awards Outstanding | Weighted Average Exercise Price | ||||||||||
| Balance at December 31, 2011 | 2,578 | 5,595 | $ | 1.56 | ||||||||
| Options granted | (1,718 | ) | 1,718 | $ | 1.14 | |||||||
| Options cancelled and expired | 290 | (290 | ) | $ | 5.54 | |||||||
| Awards issued | — | (181 | ) | $ | — | |||||||
| Expiration of option plan | (1,150 | ) | — | $ | — | |||||||
| Balance at December 31, 2012 | — | 6,842 | $ | 1.33 | ||||||||
| Options exercised | — | (75 | ) | $ | 1.50 | |||||||
| Options cancelled and expired | — | (35 | ) | $ | 3.29 | |||||||
| Balance at December 31, 2013 | — | 6,732 | $ | 1.31 | ||||||||
| Increase in shares reserved | 2,500 | — | — | |||||||||
| Options granted | (325 | ) | 325 | $ | 0.63 | |||||||
| Options cancelled and forfeited | — | (27 | ) | $ | 1.66 | |||||||
| Options expired | — | (410 | ) | $ | 2.12 | |||||||
| Awards granted | (617 | ) | 617 | $ | — | |||||||
| Awards issued | — | (258 | ) | $ | — | |||||||
| Balance at December 31, 2014 | 1,558 | 6,979 | $ | 1.16 | ||||||||
| F-19 |
The 2002 Plan and the 2001 NQ Plan allow for stock options issued as the result of a merger or consolidation of another entity, including the acquisition of the minority interest of our former subsidiaries, to be added to the maximum number of shares provided for in the plan (“Substitute Options”). Consequently, Substitute Options are not returned to the shares reserved under the plan when cancelled. During 2014, 2013 and 2012, the number of Substitute Options cancelled was immaterial.
Options to purchase approximately 6.6 million and 6.7 million shares were exercisable at December 31, 2014 and 2013, respectively. The options outstanding at December 31, 2014 have been segregated into four ranges for additional disclosure as follows (options in thousands):
| Options Outstanding | Options Exercisable | |||||||||||||||||||
| Range of Exercise Prices | Number Outstanding | Weighted Average Remaining Life (Years) | Weighted Average Exercise Price | Number Exercisable | Weighted Average Exercise Price | |||||||||||||||
| $0.45 - $0.92 | 2,615 | 4.95 | $ | 0.77 | 2,490 | $ | 0.78 | |||||||||||||
| $0.93 - $1.19 | 1,668 | 6.97 | $ | 1.15 | 1,668 | $ | 1.15 | |||||||||||||
| $1.20 - $1.46 | 1,194 | 5.03 | $ | 1.33 | 1,194 | $ | 1.33 | |||||||||||||
| $1.47 - $3.13 | 1,143 | 3.00 | $ | 2.29 | 1,143 | $ | 2.29 | |||||||||||||
| $0.45 - $3.13 | 6,620 | 5.14 | $ | 1.23 | 6,495 | $ | 1.24 | |||||||||||||
We use the Black-Scholes-Merton option-pricing model with the following assumptions to estimate the stock-based compensation expense for the years ended December 31, 2014, 2013 and 2012:
| Years Ended December 31, | ||||||||||||
| 2014 | 2013 | 2012 | ||||||||||
| Weighted-average risk-free interest rate | 2.04 | % | 0.92 | % | 0.91 | % | ||||||
| Expected dividend payments | — | — | — | |||||||||
| Expected holding period (years)(1) | 6.46 | 3.9 | 5.1 | |||||||||
| Weighted-average volatility factor(2) | 1.65 | 1.38 | 1.75 | |||||||||
| Estimated forfeiture rates for options granted | 31 | % | 31 | % | 31 | % | ||||||
| (1) | Expected holding period is based on historical experience of similar awards, giving consideration to the contractual terms of the stock-based awards, vesting schedules and the expectations of future employee behavior. |
| (2) | Weighted average volatility is based on the historical volatility of our common stock. |
| (3) | Estimated forfeiture rates are based on historical data. |
During the year ended December 31, 2014, 325,000 options and 617,000 awards were granted. Based upon the above methodology, the weighted-average fair value of options and awards granted during the years ended December 31, 2014 and 2012 was $0.64 and $1.09, respectively. No options or awards were granted during the year ended December 31, 2013.
The following table summarizes the stock-based compensation expense and impact on our basic and diluted loss per share for the years ended December 31, 2013, 2012 and 2011:
| Years Ended December 31, | ||||||||||||
| (in thousands, except per share amounts) | 2014 | 2013 | 2012 | |||||||||
| Research and development | $ | 245 | $ | 378 | $ | 1,021 | ||||||
| General and administrative | 298 | 300 | 1,560 | |||||||||
| Total stock-based compensation expenses | $ | 543 | $ | 678 | $ | 2,581 | ||||||
| Increase in basic net income (loss) per share | $ | (0.01 | ) | $ | (0.01 | ) | $ | (0.04 | ) | |||
| Increase in diluted net income (loss) per share | $ | (0.01 | ) | $ | (0.01 | ) | $ | (0.04 | ) | |||
| F-20 |
No tax benefit was recognized related to stock-based compensation expense since we have incurred operating losses and we have established a full valuation allowance to offset all the potential tax benefits associated with our deferred tax assets.
During the year ended December 31, 2014, 325,000 options were granted to employees, directors and consultants. The following table summarizes option activity for the year ended December 31, 2014:
| (in thousands, except per share amounts) | Shares | Weighted Average Exercise Price | Weighted Average Remaining Contractual Term | Aggregate Intrinsic Value | ||||||||||||
| Outstanding at January 1, 2014 | 6,732 | $ | 1.31 | |||||||||||||
| Granted | 325 | 0.63 | ||||||||||||||
| Cancelled and forfeited | (27 | ) | 1.66 | |||||||||||||
| Expired | (410 | ) | 2.12 | |||||||||||||
| Outstanding at December 31, 2014 | 6,620 | $ | 1.23 | 5.14 | $ | 1 | ||||||||||
| Exercisable at December 31, 2014 | 6,495 | $ | 1.24 | 5.05 | $ | — | ||||||||||
As of December 31, 2014, there was approximately $19,000 of total unrecognized compensation expense related to non-vested stock options. This expense is expected to be recognized over a weighted-average period of 0.65 years.
During the year ended December 31, 2014, 617,000 restricted stock awards were granted to employees, directors and consultants. The following table summarizes restricted stock award activity for the year ended December 31, 2014:
| (in thousands, except per share amounts) | Shares | Weighted Average Exercise Price | Weighted Average Remaining Contractual Term | Aggregate Intrinsic Value | ||||||||||||
| Outstanding at January 1, 2014 | — | $ | — | |||||||||||||
| Granted | 617 | — | ||||||||||||||
| Released | (258 | ) | — | |||||||||||||
| Outstanding at December 31, 2014 | 359 | $ | — | 9.12 | $ | 166 | ||||||||||
| Exercisable at December 31, 2014 | — | $ | — | — | $ | — | ||||||||||
Of the 258,000 released restricted stock awards during 2014, approximately 54,000 were withheld to satisfy tax withholding requirements.
As of December 31, 2014, there was approximately $26,000 of total unrecognized compensation expense related to non-vested stock options. This expense is expected to be recognized over a weighted-average period of 0.11 years.
13. Income Taxes
As of December 31, 2014, we had net operating loss carryforwards for federal income tax purposes of approximately $232.5 million that expire at various dates through 2034, and federal research and development tax credits of approximately $8.3 million that expire at various dates through 2034. We also had net operating loss carryforwards for California income tax purposes of approximately $154.7 million that expire at various dates through 2034 and state research and development tax credits of approximately $8.1 million which do not expire. Approximately $12.4 million of federal and state net operating loss carryforwards represent stock option deductions arising from activity under our stock option plans, the benefit of which will increase additional paid in capital when realized.
| F-21 |
Current federal and California tax laws include substantial restrictions on the utilization of net operating losses and tax credits in the event of an ownership change of a corporation. We have performed a change in ownership analysis through December 31, 2014 and, accordingly, all of our net operating loss and tax credit carryforwards are available to offset future taxable income, if any.
Deferred income taxes reflect the net tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes and operating loss and credit carryforwards. Significant components of our deferred tax assets are as follows (in thousands):
| December 31, | ||||||||
| 2014 | 2013 | |||||||
| Deferred tax assets: | ||||||||
| Net operating loss carryforwards | $ | 88,086 | $ | 85,912 | ||||
| Research credit carryforwards | 13,645 | 13,481 | ||||||
| Other, net | 3,529 | 3,962 | ||||||
| Deferred revenue | 666 | 2,116 | ||||||
| Total deferred tax assets | 105,926 | 105,471 | ||||||
| Valuation allowance | (105,926 | ) | (105,471 | ) | ||||
| Net deferred tax assets | $ | — | $ | — | ||||
Realization of deferred tax assets is dependent upon future earnings, if any, the timing and amount of which are uncertain. Accordingly, the net deferred tax assets have been fully offset by a valuation allowance. The valuation allowance increased by $0.5 million during 2014, increased by $1.7 million during 2013 and increased by $3.1 million during 2012.
Under ASC 718, the deferred tax asset for net operating losses as of December 31, 2014 excludes deductions for excess tax benefits related to stock based compensation.
| F-22 |
TITAN PHARMACEUTICALS, INC.
NOTES TO FINANCIAL STATEMENTS—(Continued)
The provision for income taxes consists of state minimum taxes due. The effective tax rate of our provision (benefit) for income taxes differs from the federal statutory rate as follows (in thousands):
| Year Ending December 31, | ||||||||||||
| 2014 | 2013 | 2012 | ||||||||||
| Computed at 34% | $ | (839 | ) | $ | 3,301 | $ | (5,134 | ) | ||||
| State taxes | 592 | 213 | (234 | ) | ||||||||
| Book gains (losses) not currently benefited | 454 | 1,656 | 3,120 | |||||||||
| Other | 235 | (476 | ) | 1,901 | ||||||||
| Disallowed interest expense | — | 160 | 1,363 | |||||||||
| Income from debt restructuring | — | — | (1,615 | ) | ||||||||
| Revaluation of warrant liability | (346 | ) | (591 | ) | 600 | |||||||
| Research and development credits | (97 | ) | (583 | ) | — | |||||||
| Non-cash gain from termination of royalty purchase agreement | — | (3,047 | ) | — | ||||||||
| Non-cash gain on settlement of long-term debt | — | (632 | ) | — | ||||||||
| Total | $ | (1 | ) | $ | 1 | $ | 1 | |||||
We had no unrecognized tax benefits or any amounts accrued for interest and penalties for the three year period ended December 31, 2014. Our policy is to recognize interest and penalties related to income taxes as a component of income tax expense.
We file tax returns in the U.S. Federal jurisdiction and some state jurisdictions. We are subject to the U.S. federal and state income tax examination by tax authorities for such years 1995 through 2014, due to net operating losses that are being carried forward for tax purposes.
The Credit for Increasing Research Activities expired for amounts incurred after December 31, 2011. However, The American Taxpayer Relief Act of 2012, which was signed into law on January 2, 2013, extended the credit for amounts incurred before January 1, 2014. The Act also retroactively restored the credit for amounts incurred in 2012. However, since the Act was not signed until January 2, 2013 the amount of credit generated in 2012 was not reflected in the deferred tax amounts as of December 31, 2012. The amount of this credit that was generated in 2012 was approximately $340,000. The deferred tax asset for this credit was increased by this amount in 2013.
14. Quarterly Financial Data (Unaudited)
| First Quarter | Second Quarter | Third Quarter | Fourth Quarter | |||||||||||||
| (in thousands, except per share amount) | ||||||||||||||||
| 2014 | ||||||||||||||||
| Total revenue | $ | 911 | $ | 911 | $ | 911 | $ | 912 | ||||||||
| Net income (loss) | $ | (1,804 | ) | $ | (842 | ) | $ | 716 | $ | (473 | ) | |||||
| Basic net income (loss) per share | $ | (0.02 | ) | $ | (0.01 | ) | $ | 0.01 | $ | (0.00 | ) | |||||
| Diluted net income (loss) per share | $ | (0.02 | ) | $ | (0.01 | ) | $ | (0.01 | ) | $ | (0.01 | ) | ||||
| 2013 | ||||||||||||||||
| Total revenue | $ | 5,174 | $ | 2,198 | $ | 2,198 | $ | 911 | ||||||||
| Net income (loss) | $ | 6,001 | $ | 5,064 | $ | (1,145 | ) | $ | (209 | ) | ||||||
| Basic net income (loss) per share | $ | 0.08 | $ | 0.06 | $ | (0.01 | ) | $ | (0.00 | ) | ||||||
| Diluted net income (loss) per share | $ | 0.07 | $ | 0.00 | $ | (0.01 | ) | $ | (0.00 | ) | ||||||
| F-23 |
15. Subsequent Events
In February 2015, we amended the Class A Warrants issued in connection with our public offering in October 2014 to extend the date by which we must hold a stockholders meeting for the purpose of seeking approval of our stockholders for an amendment to our certificate of incorporation to either (i) increase the number of shares of common stock we are authorized to issue or (ii) effect a reverse split of the common stock to August 31, 2015.
| F-24 |
(b) Exhibits
| No. | Description | |
| 3.1 | Amended and Restated Certificate of Incorporation of the Registrant, as amended9 | |
| 3.2 | By-laws of the Registrant1 | |
| 3.3 | Certificate of Designations of Junior Participating Preferred Stock of Titan Pharmaceuticals, Inc.15 | |
| 4.1 | Registration Rights Agreement dated as of December 17, 20072 | |
| 4.2 | Registration Rights Agreement dated as of December 8, 20099 | |
| 4.3 | Warrant to Purchase Common Stock dated December 23, 2009 issued to Oxford Finance Corporation9 | |
| 4.4 | Form of Warrant13 | |
| 4.5 | Registration Rights Agreement, dated as of March 15, 201113 | |
| 4.6 | Form of Series A Warrant18 | |
| 4.6 | Form of Class A Warrant26 | |
| 4.6 | Form of Underwriter Warrant26 | |
| 10.1 | 1998 Stock Option Plan3 | |
| 10.2 | 2001 Non-Qualified Employee Stock Option Plan4 | |
| 10.3 | 2002 Stock Option Plan5 | |
| 10.4 | Employment Agreement between the Registrant and Sunil Bhonsle, dated May 16, 2009, as amended by agreements dated February 17, 2010, December 30, 2011 and December 31, 20129, 16, 19 | |
| 10.5 | Employment Agreement between the Registrant and Marc Rubin, dated May 16, 2009, as amended by agreements dated February 17, 2010, December 30, 2011 and December 31, 2012,9 16, 19 | |
| 10.6 | Lease for the Registrant’s facilities, amended as of October 1, 20046 | |
| 10.7 | Amendments to lease for Registrant’s facilities dated May 21, 2007 and March 12, 20099 | |
| 10.8* | License Agreement between the Registrant and Sanofi-Aventis SA effective as of December 31, 19967 | |
| 10.9* | Sublicense Agreement between the Registrant and Novartis Pharma AG dated November 20, 19978 | |
| 10.10 | Loan and Security Agreement between the Registrant and Oxford Finance Corporation dated December 18, 20099 | |
| 10.11 | Stock Purchase Agreement between the Registrant and certain investors dated December 8, 20099 | |
| 10.12 | Amendment to Employment Agreement dated June 15, 2010 between the Registrant and Marc Rubin10 | |
| 10.13 | Amendment to Employment Agreement dated June 15, 2010 between the Registrant and Sunil Bhonsle10 | |
| 10.14 | Amendment to lease for Registrant’s facilities dated June 15, 201011 |
| 49 |
| 10.15 | Amended and Restated Loan and Security Agreement between the Registrant and Oxford Finance Corporation dated September 27, 201012 | |
| 10.16 | Facility Agreement, dated as of March 15, 2011, by and among the Company, Deerfield Private Design Fund II, L.P., Deerfield Private Design International II, L.P., Deerfield Special Situations Fund, L.P., and Deerfield Special Situations Fund International Limited, as amended on February 6, 201313, 27 | |
| 10.17 | Security Agreement, dated as of March 15, 2011, by and among the Company, Deerfield Private Design Fund II, L.P., Deerfield Private Design International II, L.P., Deerfield Special Situations Fund, L.P., and Deerfield Special Situations Fund International Limited13 | |
| 10.18 | Royalty Purchase Agreement, dated November 14, 2011, by and among the Company, Deerfield Private Design Fund II, L.P., Deerfield Special Situations Fund, L.P. and Horizon Sante TTNP SARL14 | |
| 10.19 | Amended and Restated Royalty Agreement, dated November 14, 2011 by and among the Company, Deerfield Private Design Fund II, L.P., Deerfield Special Situations Fund, L.P. and Horizon Sante TTNP SARL14 | |
| 10.20 | Amended and Restated Royalty Repurchase Agreement, dated November 14, 2011, by and among the Company, Deerfield Private Design Fund II, L.P., and Horizon Sante TTNP SARL14 | |
| 10.21 | Cash Management Agreement, dated November 14, 2011, by and among the Company, Deerfield Private Design Fund II, L.P., Deerfield Special Situations Fund, L.P. and Horizon Sante TTNP SARL14 | |
| 10.22 | Paying Agent Agreement, dated November 14, 2011, by and among the Company, Deerfield Management Company, L.P. and U.S. Bank National Association14 | |
| 10.23 | Agreement, dated as of November 14, 2011, by and among the Company, Deerfield Private Design Fund II, L.P., Deerfield Private Design International II, L.P., Deerfield Special Situations Fund, L.P., and Deerfield Special Situations Fund International Limited14 | |
| 10.24 | Form of Subscription Agreement dated April 9, 201218 | |
| 10.25* | License Agreement by and between Titan Pharmaceuticals, Inc. and Braeburn Pharmaceuticals Sprl, dated December 14, 201220 | |
| 10.26 | Amendment dated May 28, 2013 to License Agreement by and between Titan Pharmaceuticals, Inc. and Braeburn Pharmaceuticals Sprl 21 | |
| 10.27 | Second Amendment dated July 2, 2013 to License Agreement by and between Titan Pharmaceuticals, Inc. and Braeburn Pharmaceuticals Sprl 22 | |
| 10.28 | Third Amendment dated November 12, 2013 to License Agreement by and between Titan Pharmaceuticals, Inc. and Braeburn Pharmaceuticals Sprl 23 | |
| 10.29 | Stock Purchase Agreement dated November 12, 2013 by and between Titan Pharmaceuticals, Inc. and Braeburn Pharmaceuticals Sprl 23 | |
| 10.30 | 2014 Incentive Plan24 | |
| 14.1 | Code of Business Conduct and Ethics 25 |
| 50 |
| 23.1 | Consent of OUM & Co., LLP, Independent Registered Public Accounting Firm | |
| 31.1 | Certification of the Principal Executive and Financial Officer pursuant to Rule 13(a)-14(a) of the Securities Exchange Act of 1934 | |
| 32.1 | Certification of the Principal Executive and Financial Officer pursuant to 18 U.S.C. 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
| 101.INS** | XBRL Instance Document | |
| 101.SCH** | XBRL Taxonomy Extension Schema Document | |
| 101.CAL** | XBRL Taxonomy Extension Calculation Linkbase Document | |
| 101.DEF** | XBRL Taxonomy Extension Definition Linkbase Document | |
| 101.LAB** | XBRL Taxonomy Extension Label Linkbase Document | |
| 101.PRE** | XBRL Taxonomy Extension Presentation Linkbase Document |
| (1) | Incorporated by reference from the Registrant’s Registration Statement on Form SB-2 (File No. 33-99386). |
| (2) | Incorporated by reference from the Registrant’s Current Report on Form 8-K dated December 27, 2007. |
| (3) | Incorporated by reference from the Registrant’s definitive Proxy Statement filed on July 28, 2000. |
| (4) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2001. |
| (5) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2002. |
| (6) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2005. |
| (7) | Incorporated by reference from the Registrant’s Annual Report on Form 10-KSB for the year ended December 31, 1996. |
| (8) | Incorporated by reference from the Registrant’s Registration Statement on Form S-3 (File No. 333-42367). |
| (9) | Incorporated by reference from the Registrant’s Registration Statement on Form 10. |
| (10) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2003. |
| (11) | Incorporated by reference from the Registrant’s Quarterly Report on Form 10-Q for the period ended June 30, 2010. |
| (12) | Incorporated by reference from the Registrant’s Quarterly Report on Form 10-Q for the period ended September 30, 2010. |
| (13) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on March 18, 2011. |
| (14) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on November 17, 2011. |
| (15) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on December 21, 2011. |
| (16) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on January 4, 2012. |
| (17) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2011. |
| (18) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on April 10, 2013. |
| (19) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on January 2, 2013. |
| (20) | Incorporated by reference from the Registrant’s Current Report on Form 8-K/A filed on February 28, 2013. |
| (21) | Incorporated by reference from the Registrant’s Current Report on Form 8-K dated May 29, 2013. |
| (22) | Incorporated by reference from the Registrant’s Current Report on Form 8-K dated July 5, 2013. |
| (23) | Incorporated by reference from the Registrant’s Current Report on Form 8-K dated November 13, 2013. |
| (24) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2013. |
| (25) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2012. |
| 51 |
| (26) | Incorporated by reference from the Registrant’s Registration Statement on Form S-1/A (File No. 333-198476). |
| (27) | Incorporated by reference from the Registrant’s Current Report on Form 8-K dated February 7, 2013. |
| * | Confidential treatment has been granted with respect to portions of this exhibit. |
| ** | Pursuant to Rule 406T of Regulation S-T, the interactive files on Exhibit 101.1 hereto are deemed not filed or part of a registration statement or prospectus for purposes of Sections 11 or 12 of the Securities Act of 1933, as amended, are deemed not filed for purposes of Section 18 of the Securities Exchange Act of 1934, as amended, and otherwise are not subject to liability under those sections. |
| 52 |
SIGNATURES
Pursuant to the requirements of Section 12 of the Securities Exchange Act of 1934, the registrant has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized.
| Date: March 31, 2015 | TITAN PHARMACEUTICALS, INC. | |
| By: | /S/ SUNIL BHONSLE | |
| Name: | Sunil Bhonsle | |
| Title: | President | |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons in the capacities and on the dates stated.
| Signature | Title | Date | ||
| /s/ Marc Rubin, M.D. | Executive Chairman | March 31, 2015 | ||
| Marc Rubin, M.D. | ||||
| /s/ Sunil Bhonsle | President and Director | March 31, 2015 | ||
| Sunil Bhonsle | (principal executive officer and principal financial officer) | |||
| /s/ Joseph A. Akers | Director | March 31, 2015 | ||
| Joseph A. Akers | ||||
| /s/ Victor J. Bauer, Ph.D. | Director | March 31, 2015 | ||
| Victor J. Bauer, Ph.D. | ||||
| /s/ Eurelio M. Cavalier | Director | March 31, 2015 | ||
| Eurelio M. Cavalier | ||||
| /s/ M. David MacFarlane, Ph.D. | Director | March 31, 2015 | ||
| M. David MacFarlane, Ph.D. | ||||
| /s/ James R. McNab, Jr. | Director | March 31, 2015 | ||
| James R. McNab, Jr. | ||||
| /s/ Ley S. Smith | Director | March 31, 2015 | ||
| Ley S. Smith | ||||
| /s/ Brian E. Crowley | Vice President, Finance | March 31, 2015 | ||
| Brian E. Crowley | (principal accounting officer) |
| 53 |
EXHIBIT INDEX
Exhibits
| No. | Description | |
| 3.1 | Amended and Restated Certificate of Incorporation of the Registrant, as amended9 | |
| 3.2 | By-laws of the Registrant1 | |
| 3.3 | Certificate of Designations of Junior Participating Preferred Stock of Titan Pharmaceuticals, Inc.15 | |
| 4.1 | Registration Rights Agreement dated as of December 17, 20072 | |
| 4.2 | Registration Rights Agreement dated as of December 8, 20099 | |
| 4.3 | Warrant to Purchase Common Stock dated December 23, 2009 issued to Oxford Finance Corporation9 | |
| 4.4 | Form of Warrant13 | |
| 4.5 | Registration Rights Agreement, dated as of March 15, 201113 | |
| 4.6 | Form of Series A Warrant18 | |
| 4.6 | Form of Class A Warrant26 | |
| 4.6 | Form of Underwriter Warrant26 | |
| 10.1 | 1998 Stock Option Plan3 | |
| 10.2 | 2001 Non-Qualified Employee Stock Option Plan4 | |
| 10.3 | 2002 Stock Option Plan5 | |
| 10.4 | Employment Agreement between the Registrant and Sunil Bhonsle, dated May 16, 2009, as amended by agreements dated February 17, 2010, December 30, 2011 and December 31, 20129, 16, 19 | |
| 10.5 | Employment Agreement between the Registrant and Marc Rubin, dated May 16, 2009, as amended by agreements dated February 17, 2010, December 30, 2011 and December 31, 2012,9 16, 19 | |
| 10.6 | Lease for the Registrant’s facilities, amended as of October 1, 20046 | |
| 10.7 | Amendments to lease for Registrant’s facilities dated May 21, 2007 and March 12, 20099 | |
| 10.8* | License Agreement between the Registrant and Sanofi-Aventis SA effective as of December 31, 19967 | |
| 10.9* | Sublicense Agreement between the Registrant and Novartis Pharma AG dated November 20, 19978 | |
| 10.10 | Loan and Security Agreement between the Registrant and Oxford Finance Corporation dated December 18, 20099 | |
| 10.11 | Stock Purchase Agreement between the Registrant and certain investors dated December 8, 20099 | |
| 10.12 | Amendment to Employment Agreement dated June 15, 2010 between the Registrant and Marc Rubin10 |
| 54 |
| 10.13 | Amendment to Employment Agreement dated June 15, 2010 between the Registrant and Sunil Bhonsle10 | |
| 10.14 | Amendment to lease for Registrant’s facilities dated June 15, 201011 | |
| 10.15 | Amended and Restated Loan and Security Agreement between the Registrant and Oxford Finance Corporation dated September 27, 201012 | |
| 10.16 | Facility Agreement, dated as of March 15, 2011, by and among the Company, Deerfield Private Design Fund II, L.P., Deerfield Private Design International II, L.P., Deerfield Special Situations Fund, L.P., and Deerfield Special Situations Fund International Limited, as amended on February 6, 201313, 27 | |
| 10.17 | Security Agreement, dated as of March 15, 2011, by and among the Company, Deerfield Private Design Fund II, L.P., Deerfield Private Design International II, L.P., Deerfield Special Situations Fund, L.P., and Deerfield Special Situations Fund International Limited13 | |
| 10.18 | Royalty Purchase Agreement, dated November 14, 2011, by and among the Company, Deerfield Private Design Fund II, L.P., Deerfield Special Situations Fund, L.P. and Horizon Sante TTNP SARL14 | |
| 10.19 | Amended and Restated Royalty Agreement, dated November 14, 2011 by and among the Company, Deerfield Private Design Fund II, L.P., Deerfield Special Situations Fund, L.P. and Horizon Sante TTNP SARL14 | |
| 10.20 | Amended and Restated Royalty Repurchase Agreement, dated November 14, 2011, by and among the Company, Deerfield Private Design Fund II, L.P., and Horizon Sante TTNP SARL14 | |
| 10.21 | Cash Management Agreement, dated November 14, 2011, by and among the Company, Deerfield Private Design Fund II, L.P., Deerfield Special Situations Fund, L.P. and Horizon Sante TTNP SARL14 | |
| 10.22 | Paying Agent Agreement, dated November 14, 2011, by and among the Company, Deerfield Management Company, L.P. and U.S. Bank National Association14 | |
| 10.23 | Agreement, dated as of November 14, 2011, by and among the Company, Deerfield Private Design Fund II, L.P., Deerfield Private Design International II, L.P., Deerfield Special Situations Fund, L.P., and Deerfield Special Situations Fund International Limited14 | |
| 10.24 | Form of Subscription Agreement dated April 9, 201218 | |
| 10.25* | License Agreement by and between Titan Pharmaceuticals, Inc. and Braeburn Pharmaceuticals Sprl, dated December 14, 201220 | |
| 10.26 | Amendment dated May 28, 2013 to License Agreement by and between Titan Pharmaceuticals, Inc. and Braeburn Pharmaceuticals Sprl 21 | |
| 10.27 | Second Amendment dated July 2, 2013 to License Agreement by and between Titan Pharmaceuticals, Inc. and Braeburn Pharmaceuticals Sprl 22 | |
| 10.28 | Third Amendment dated November 12, 2013 to License Agreement by and between Titan Pharmaceuticals, Inc. and Braeburn Pharmaceuticals Sprl 23 | |
| 10.29 | Stock Purchase Agreement dated November 12, 2013 by and between Titan Pharmaceuticals, Inc. and Braeburn Pharmaceuticals Sprl 23 | |
| 10.30 | 2014 Incentive Plan24 | |
| 14.1 | Code of Business Conduct and Ethics 25 |
| 55 |
| 23.1 | Consent of OUM & Co., LLP, Independent Registered Public Accounting Firm | |
| 31.1 | Certification of the Principal Executive and Financial Officer pursuant to Rule 13(a)-14(a) of the Securities Exchange Act of 1934 | |
| 32.1 | Certification of the Principal Executive and Financial Officer pursuant to 18 U.S.C. 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
| 101.INS** | XBRL Instance Document | |
| 101.SCH** | XBRL Taxonomy Extension Schema Document | |
| 101.CAL** | XBRL Taxonomy Extension Calculation Linkbase Document | |
| 101.DEF** | XBRL Taxonomy Extension Definition Linkbase Document | |
| 101.LAB** | XBRL Taxonomy Extension Label Linkbase Document | |
| 101.PRE** | XBRL Taxonomy Extension Presentation Linkbase Document |
| (1) | Incorporated by reference from the Registrant’s Registration Statement on Form SB-2 (File No. 33-99386). |
| (2) | Incorporated by reference from the Registrant’s Current Report on Form 8-K dated December 27, 2007. |
| (3) | Incorporated by reference from the Registrant’s definitive Proxy Statement filed on July 28, 2000. |
| (4) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2001. |
| (5) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2002. |
| (6) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2005. |
| (7) | Incorporated by reference from the Registrant’s Annual Report on Form 10-KSB for the year ended December 31, 1996. |
| (8) | Incorporated by reference from the Registrant’s Registration Statement on Form S-3 (File No. 333-42367). |
| (9) | Incorporated by reference from the Registrant’s Registration Statement on Form 10. |
| (10) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2003. |
| (11) | Incorporated by reference from the Registrant’s Quarterly Report on Form 10-Q for the period ended June 30, 2010. |
| (12) | Incorporated by reference from the Registrant’s Quarterly Report on Form 10-Q for the period ended September 30, 2010. |
| (13) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on March 18, 2011. |
| (14) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on November 17, 2011. |
| (15) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on December 21, 2011. |
| (16) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on January 4, 2012. |
| (17) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2011. |
| (18) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on April 10, 2013. |
| (19) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on January 2, 2013. |
| (20) | Incorporated by reference from the Registrant’s Current Report on Form 8-K/A filed on February 28, 2013. |
| (21) | Incorporated by reference from the Registrant’s Current Report on Form 8-K dated May 29, 2013. |
| (22) | Incorporated by reference from the Registrant’s Current Report on Form 8-K dated July 5, 2013. |
| (23) | Incorporated by reference from the Registrant’s Current Report on Form 8-K dated November 13, 2013. |
| (24) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2013. |
| (25) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2012. |
| 56 |
| (26) | Incorporated by reference from the Registrant’s Registration Statement on Form S-1/A (File No. 333-198476). |
| (27) | Incorporated by reference from the Registrant’s Current Report on Form 8-K dated February 7, 2013. |
| * | Confidential treatment has been granted with respect to portions of this exhibit. |
| ** | Pursuant to Rule 406T of Regulation S-T, the interactive files on Exhibit 101.1 hereto are deemed not filed or part of a registration statement or prospectus for purposes of Sections 11 or 12 of the Securities Act of 1933, as amended, are deemed not filed for purposes of Section 18 of the Securities Exchange Act of 1934, as amended, and otherwise are not subject to liability under those sections. |
| 57 |
