Attached files
| file | filename |
|---|---|
| EXCEL - IDEA: XBRL DOCUMENT - GERON CORP | Financial_Report.xls |
| EX-32.2 - EX-32.2 - GERON CORP | a2223360zex-32_2.htm |
| EX-31.1 - EX-31.1 - GERON CORP | a2223360zex-31_1.htm |
| EX-31.2 - EX-31.2 - GERON CORP | a2223360zex-31_2.htm |
| EX-12.1 - EX-12.1 - GERON CORP | a2223360zex-12_1.htm |
| EX-32.1 - EX-32.1 - GERON CORP | a2223360zex-32_1.htm |
| EX-23.1 - EX-23.1 - GERON CORP | a2223360zex-23_1.htm |
| EX-10.36 - EX-10.36 - GERON CORP | a2223360zex-10_36.htm |
| EX-10.35 - EX-10.35 - GERON CORP | a2223360zex-10_35.htm |
Use these links to rapidly review the document
TABLE OF CONTENTS
ITEM 8. CONSOLIDATED FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
| (Mark One) | ||
ý |
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
|
For the Fiscal Year Ended December 31, 2014 |
||
or |
||
o |
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
|
For the transition period from to . |
||
Commission File Number: 0-20859
GERON CORPORATION
(Exact name of registrant as specified in its charter)
| Delaware (State or other jurisdiction of incorporation or organization) |
75-2287752 (I.R.S. Employer Identification No.) |
|
149 Commonwealth Drive, Suite 2070, Menlo Park, CA (Address of principal executive offices) |
94025 (Zip Code) |
Registrant's telephone number, including area code: (650) 473-7700
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Name of each exchange on which registered | |
|---|---|---|
| Common Stock, $0.001 par value | The Nasdaq Stock Market LLC |
Securities registered pursuant to Section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes o No ý
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes o No ý
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ý No o
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Website, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes ý No o
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant's knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ý
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of "large accelerated filer", "accelerated filer" and "smaller reporting company" in Rule 12b-2 of the Exchange Act.
| Large accelerated filer o | Accelerated filer ý | Non-accelerated filer o (Do not check if a smaller reporting company) |
Smaller reporting company o |
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes o No ý
The aggregate market value of voting and non-voting common equity held by non-affiliates of the registrant was approximately $501,005,000 based upon the closing price of the registrant's common stock on June 30, 2014 on the Nasdaq Global Select Market. The calculation of the aggregate market value of voting and non-voting common equity held by non-affiliates of the registrant excludes shares of common stock held by each officer, director and stockholder that the registrant concluded were affiliates on that date. This determination of affiliate status is not necessarily a conclusive determination for other purposes.
As of March 6, 2015, there were 157,700,375 shares of common stock outstanding.
DOCUMENTS INCORPORATED BY REFERENCE:
Document
|
Form 10-K Parts |
|
|---|---|---|
Portions of the Registrant's definitive proxy statement for the 2015 annual meeting of stockholders to be filed pursuant to Regulation 14A within 120 days of the Registrant's fiscal year ended December 31, 2014 |
III |
In this report, unless otherwise indicated or the context otherwise requires, "Geron," "the registrant," "we," "us," and "our" refer to Geron Corporation, a Delaware corporation.
2
Forward-Looking Statements
This annual report on Form 10-K, including "Business" in Part I, Item 1 and "Management's Discussion and Analysis of Financial Condition and Results of Operations" in Part II, Item 7, contains forward-looking statements that involve risks and uncertainties, as well as assumptions that, if they never materialize or prove incorrect, could cause the results of Geron Corporation, or Geron or the Company, to differ materially from those expressed or implied by such forward-looking statements. All statements other than statements of historical fact are statements that could be deemed forward-looking statements. In some cases, forward-looking statements can be identified by the use of terminology such as "may," "expects," "plans," "intends," "will," "should," "projects," "believes," "predicts," "anticipates," "estimates," "potential," or "continue" or the negative thereof or other comparable terminology. The risks and uncertainties referred to above include, without limitation, risks related to our research and development efforts, our dependence on Janssen Biotech, Inc. for the development, regulatory approval, manufacture and commercialization of our sole product candidate, imetelstat, need for future capital, uncertainty of clinical trial results or regulatory approvals or clearances, the future development of imetelstat, including any future efficacy or safety results that cause the benefit-risk profile of imetelstat to become unacceptable, enforcement of our patent and proprietary rights, reliance upon investigators, potential competition and other risks that are described herein and that are otherwise described from time to time in our Securities and Exchange Commission reports including, but not limited to, the factors described in Part I, Item 1A, "Risk Factors," of this annual report on Form 10-K. Geron assumes no obligation for and except as required by law, disclaims any obligation to update these forward-looking statements to reflect future information, events or circumstances.
Calculation of Aggregate Market Value of Non-Affiliate Shares
For purposes of calculating the aggregate market value of shares of our common stock held by non-affiliates as set forth on the cover page of this annual report on Form 10-K, we have assumed that all outstanding shares are held by non-affiliates, except for shares held by each of our executive officers, directors and 5% or greater stockholders. In the case of 5% or greater stockholders, we have not deemed such stockholders to be affiliates unless there are facts and circumstances which would indicate that such stockholders exercise any control over our company. These assumptions should not be deemed to constitute an admission that all executive officers, directors and 5% or greater stockholders are, in fact, affiliates of our company, or that there are no other persons who may be deemed to be affiliates of our company. Further information concerning shareholdings of our executive officers, directors and principal stockholders is incorporated by reference in Part III, Item 12 of this annual report on Form 10-K.
Company Overview
We are a clinical stage biopharmaceutical company focused on the development of a telomerase inhibitor, imetelstat, in hematologic myeloid malignancies. The discovery and early development of imetelstat, our sole product candidate, was based on our core expertise in telomerase and telomere biology. Telomerase is an enzyme that enables cancer cells, including malignant progenitor cells, to maintain telomere length, which provides them with the capacity for limitless, uncontrolled proliferation. Using our proprietary nucleic acid chemistry, we designed imetelstat to be an oligonucleotide that targets and binds with high affinity to the active site of telomerase, thereby directly inhibiting telomerase activity and impeding malignant cell proliferation. Molecular responses in essential thrombocythemia, or ET, and remission responses, including reversal of bone marrow fibrosis,
3
in myelofibrosis, or MF, suggest imetelstat has disease-modifying activity by inhibiting the progenitor cells of the malignant clone for the underlying disease in a relatively selective manner.
On November 13, 2014, we entered into an exclusive collaboration and license agreement, or the Collaboration Agreement, with Janssen Biotech, Inc., or Janssen, to develop and commercialize imetelstat worldwide for all indications in oncology, including hematologic myeloid malignancies, and all other human therapeutic uses. The Collaboration Agreement became effective on December 15, 2014 and we received $35 million from Janssen as an upfront payment. Additional consideration under the Collaboration Agreement includes payments up to a potential total of $900 million for the achievement of development, regulatory and commercial milestones, as well as royalties on worldwide net sales.
Under the Collaboration Agreement, Janssen is responsible for the development, manufacturing and commercialization of, and seeking regulatory approval for imetelstat worldwide. Development of imetelstat will proceed under a mutually agreed clinical development plan, which includes two Phase 2 studies to be pursued initially, one in myelofibrosis, referred to as the Initial Phase 2 MF Study, and one in myelodysplastic syndrome, referred to as the Initial Phase 2 MDS Study. We expect Janssen to initiate the Initial Phase 2 MF Study in mid-2015, followed by the Initial Phase 2 MDS Study to be initiated at the end of 2015. In addition, the clinical development plan may also include additional, possible registration studies in MF and myelodysplastic syndrome, or MDS, and possible exploratory Phase 2 and potential follow-on Phase 3 studies in acute myelogenous leukemia, or AML.
We believe our current operational and financial resources, including the upfront payment received from Janssen under the Collaboration Agreement, may enable us to acquire one or more oncology products, programs or companies to diversify our business.
Telomeres and Telomerase in Normal Development
In the human body, normal growth and maintenance of tissues occurs by cell division. However, most cells are only able to divide a limited number of times, and this number of divisions is regulated by telomere length. Telomeres are repetitions of a deoxyribonucleic acid, or DNA, sequence located at the ends of chromosomes. They act as protective caps to maintain stability and integrity of the chromosomes, which contain the cell's genetic material. Normally, every time a cell divides, the telomeres shorten. Eventually, they shrink to a critically short length, and as a result, the cell either dies by apoptosis or stops dividing and senesces.
Telomerase is a naturally occurring enzyme that maintains telomeres and prevents them from shortening during cell division in cells, such as stem cells, that must remain immortalized to support normal health. Telomerase consists of at least two essential components: a ribonucleic acid, or RNA, template (hTR), which binds to the telomere, and a catalytic subunit (hTERT) with reverse transcriptase activity, which adds a specific DNA sequence to the chromosome ends. The 2009 Nobel Prize for Physiology and Medicine was awarded to Drs. Elizabeth H. Blackburn and Carol W. Greider, together with Dr. Jack Szostak, who were former Geron collaborators, for the discovery of how chromosomes are protected by telomeres and the enzyme telomerase.
Telomerase is active during embryonic development, enabling the rapid cell division that supports normal growth. During the latter stages of human fetal development and in adulthood, telomerase is repressed in most cells, and telomere length gradually decreases during a lifetime. In tissues that have a high turnover throughout life, such as blood and gut, telomerase can be transiently upregulated in progenitor cells to enable controlled, self-limited proliferation to replace cells lost through natural cell aging processes. In proliferating progenitor cells, relatively long telomeres are maintained by upregulated telomerase. As the progeny of progenitor cells mature, telomerase is downregulated and telomeres shorten with cell division, preventing uncontrolled proliferation.
4
Telomeres and Telomerase in Cancer
Telomerase is upregulated in many tumor progenitor cells, which enables the continued and uncontrolled proliferation of the malignant cells that drive tumor growth and progression. Telomerase expression has been found to be present in approximately 90% of biopsies taken from a broad range of human cancers. Our nonclinical studies, in which the telomerase gene was artificially introduced and expressed in normal cells grown in culture, have suggested that telomerase does not itself cause a normal cell to become malignant. However, the sustained upregulation of telomerase enables tumor cells to maintain telomere length, providing them with the capacity for limitless proliferation. We believe that the sustained upregulation of telomerase is critical for tumor progression as it enables malignant progenitor cells to acquire cellular immortality and avoid apoptosis, or cell death.
Telomerase Inhibition: Inducing Cancer Cell Death
We believe that inhibiting telomerase may be an attractive approach to treating cancer because it may limit the proliferative capacity of malignant cells. We and others have observed in various in vitro and rodent tumor models that inhibiting telomerase results in telomere shortening and arrests uncontrolled malignant cell proliferation and tumor growth. In vitro studies have suggested that tumor cells with short telomeres may be especially sensitive to the anti-proliferative effects of inhibiting telomerase. Our nonclinical data also suggest that inhibiting telomerase is particularly effective at limiting the proliferation of malignant progenitor cells, which have high levels of telomerase and are believed to be important drivers of tumor growth and progression.
Many hematologic malignancies, such as ET, MF, and polycythemia vera, or PV, are known to arise from malignant progenitor cells in the bone marrow that express higher telomerase activity and have shorter telomeres when compared to normal healthy cells. Recent nonclinical data reported in the journal Cell Stem Cell in December 2014 by Steven Lane, M.D., Ph.D., Queensland Institute of Medical Research, one of our nonclinical collaborators, provided proof-of-concept of the role of telomerase in disease initiation and progression in AML. Leukemic stem cells, or LSCs, are functionally described as cells within AML that are capable of initiating and maintaining the disease. Through their high expression of telomerase, LSCs are believed to be responsible for chemotherapy resistance and relapse in AML which make them an important therapeutic target as a durable treatment for AML. Data from the nonclinical study conducted by Dr. Lane suggest that imetelstat has the potential for disease-modifying activity in AML by targeting LSCs.
Imetelstat: The First Telomerase Inhibitor to Advance to Clinical Development
Imetelstat is a lipid conjugated 13-mer oligonucleotide that is designed to be complementary to and bind with high affinity to the RNA template of telomerase, thereby directly inhibiting telomerase activity. The compound has a proprietary thio-phosphoramidate backbone, which is designed to provide resistance to the effect of cellular nucleases, thus conferring improved stability in plasma and tissues, as well as improved binding affinity to its target. To improve the ability of imetelstat to permeate through cellular membranes, we conjugated the oligonucleotide to a lipid group. Imetelstat's IC50, or half maximal inhibitory concentration, is 0.5-10 nM in cell free assays. The tissue half-life of imetelstat, or the time it takes for the concentration or amount of imetelstat to be reduced by half, in bone marrow, spleen, liver and tumor has been estimated to be 41 hours in humans, based on data from animal studies and clinical trial data. The tissue half-life indicates how long a drug will remain present in the tissues, and a longer tissue half-life may enable a drug to remain at effective doses for a longer period of time.
Imetelstat has been shown in nonclinical studies to exhibit relatively preferential inhibition of the clonal proliferation of malignant progenitor cells compared to normal progenitors. For this reason, imetelstat has been studied as a treatment for malignant diseases.
5
Imetelstat is the first telomerase inhibitor to advance to clinical development. The Phase 1 trials that we completed evaluated the safety, tolerability, pharmacokinetics and pharmacodynamic effects of imetelstat. Doses and dosing schedules were established that were tolerable and achieved target exposures in patients that were consistent with those required for efficacy in animal models. We believe adverse events were generally manageable and reversible. The dose-limiting toxicities were thrombocytopenia, or reduced platelet count, and neutropenia, or reduced neutrophil count. Following intravenous administration of imetelstat using tolerable dosing regimens, clinically relevant and significant inhibition of telomerase activity was observed in various types of tissue in which telomerase activity is measurable, including normal bone marrow hematopoietic cells, malignant plasma cells, hair follicle cells, and peripheral blood mononuclear cells.
Developing Imetelstat to Treat Hematologic Myeloid Malignancies
Proof-of-Concept in Essential Thrombocythemia
Myeloproliferative neoplasms, or MPNs, are hematologic myeloid malignancies that arise from malignant hematopoietic myeloid progenitor cells in the bone marrow, such as the precursor cells of red blood cells, platelets and granulocytes. Proliferation of malignant progenitor cells leads to an overproduction of any combination of myeloid white cells, red blood cells and/or platelets, depending on the disease. These overproduced cells may also be abnormal, leading to additional clinical complications. MPN diseases include PV, ET and MF. ET is an MPN characterized by a high platelet count, often accompanied by a high white cell count, and an increased risk of thrombosis, or bleeding, in higher risk patients.
In January 2011, we initiated a Phase 2 clinical trial of imetelstat in patients with ET. The Phase 2 ET trial was a multi-center, single arm, and open label trial that we designed to provide proof-of-concept for the potential use of imetelstat as a treatment for hematologic myeloid malignancies, such as MF, MDS or AML. The trial leveraged clinical observations from Phase 1 trials suggesting that imetelstat reduces platelet counts, as well as nonclinical observations that imetelstat distributes well to bone marrow in rodent models and selectively inhibits the proliferation of malignant progenitors ex vivo from patients with ET. Hematologic responses were measured by reductions in platelet counts, and molecular responses were measured by reductions in the JAK2 V617F mutant allele burden in circulating granulocytes as assessed by a reduction in the proportion of the abnormal Janus kinase 2, or JAK2, gene compared to the normal, or wild type JAK2 gene. We believe a decrease in the proportion of the JAK2 V617F mutant relative to the wild type JAK2 is consistent with selective inhibition of the malignant progenitor cells responsible for the disease.
Data from the primary efficacy analysis of the Phase 2 ET trial in October 2013 showed that imetelstat induced platelet count reductions in all 18 patients in the trial (a 100% hematologic response rate) and normalizations in 16 out of 18 patients (an 89% complete response rate). The median time on therapy was 17.1 months (range 6.9 months to 2.7 years). The JAK2 V617F gene mutation was detected in eight patients at baseline. Seven out of the eight (88%) patients achieved 72% to 96% reductions in JAK2 V617F allele burden that qualified as partial molecular responses with a median duration of 15.5 months. These data suggest that imetelstat inhibits the progenitor cells of the malignant clone believed to be responsible for the underlying disease in a relatively selective manner.
Adverse events reported in the Phase 2 ET trial have been similar to the adverse events reported in other imetelstat clinical trials, with fatigue, gastrointestinal symptoms (specifically nausea, diarrhea, constipation, and vomiting) and cytopenias being the most frequently observed adverse events. One patient experienced Grade 3 hepatic cirrhosis and encephalopathy which was assessed by the investigator to be possibly attributable to imetelstat, and later died of bleeding esophageal varices. Two patients experienced reversible Grade 3 alanine transaminase, which was assessed by the investigator to be possibly attributable to imetelstat. At least one abnormal liver function test, or LFT, was observed in
6
laboratory findings in all patients in the trial, with some patients experiencing persistent low-grade LFT abnormalities with longer dosing. With longer dosing, Grade 1 increases in alkaline phosphatase were observed, associated with mostly Grade 1 to some Grade 2 unconjugated hyperbilirubinemia. The clinical significance and long-term consequences of such persistent low-grade LFT abnormalities is currently undetermined.
In March 2014, we received written notice from the United States Food and Drug Administration, or FDA, that our Investigational New Drug application, or IND, for imetelstat had been placed on full clinical hold following the FDA's review of safety data in our then ongoing clinical studies. A full clinical hold is an order that the FDA issues to a trial sponsor to suspend all ongoing clinical trials and delay all proposed trials under a given IND. With this clinical hold, any patients in an ongoing Geron-sponsored clinical trial could not receive any further treatment with imetelstat. Therefore, we stopped imetelstat treatment in our Phase 2 Geron-sponsored clinical trials in ET and multiple myeloma, or MM. In our Phase 2 ET trial, imetelstat treatment was stopped in eight patients and in our Phase 2 MM trial, imetelstat treatment was stopped in two patients. See below for discussion of removal of the full clinical hold.
In their notice to us, the FDA cited the following safety issues as the basis for the clinical hold: lack of evidence of reversibility of hepatotoxicity, risk for chronic liver injury and lack of adequate follow-up in patients who experienced hepatotoxicity. To address the clinical hold, we were required to provide clinical follow-up information on patients who experienced LFT abnormalities until LFT abnormalities resolved to normal or baseline and to provide information regarding the reversibility of the liver toxicity after chronic imetelstat administration in animals.
We submitted a complete response to the FDA to seek release of the full clinical hold. In the complete response, we provided clinical follow-up information from patients in the previously ongoing Geron-sponsored Phase 2 trials in ET and MM. Our analysis of these data concluded that in the Phase 2 ET trial, LFT abnormalities resolved to normal or baseline in 14 of 18 follow-up patients. For the remaining four ET patients, at the time of the data cut-off, three showed improvement in LFT abnormalities and one had unresolved LFT abnormalities. In the Phase 2 MM trial, LFT abnormalities resolved to normal or baseline in all nine follow-up patients. In addition, no emergent hepatic adverse events were reported during follow-up for either study. In the complete response, we also provided data from our previously conducted nonclinical toxicology studies, which included a six-month study in mice and a nine-month study in cynomolgus monkeys. In those studies, no clinical pathology changes indicative of hepatocellular injury were observed, and no clear signal of LFT abnormalities were identified.
On October 31, 2014, the FDA removed the full clinical hold on our IND for imetelstat. In addition, the FDA stated that our proposed clinical development plan for imetelstat that is focused on high-risk myeloid malignancies, such as MF, is acceptable. The FDA acknowledged that we do not intend to conduct further studies in, or develop imetelstat for, the treatment of ET or PV, which is consistent with our plans as originally disclosed in April 2013.
Prior to initiation of the Initial Phase 2 MF Study, we plan to transfer our IND for imetelstat to Janssen as required by our Collaboration Agreement with them. For further discussion of the collaboration with Janssen, see the sub-section entitled, "Future Development of Imetelstat in Collaboration with Janssen".
Clinical Development in Myelofibrosis
MF is a myeloproliferative neoplasm among related diseases, such as ET, and is characterized by clonal proliferation of malignant hematopoietic progenitor cells in the bone marrow that causes bone marrow fibrosis, elevation in bone density, known as osteosclerosis, and abnormal rapid proliferation of blood vessels, known as pathological angiogenesis. MF patients may exhibit abnormally low red blood
7
cells/hemoglobin, known as progressive anemia, abnormally low white blood cells, known as leukopenia, abnormally high white blood cells, known as leukocytosis, abnormally low platelets, known as thrombocytopenia, abnormally high platelets, known as thrombocytosis, immature blood cells, known as peripheral blood leukoerythroblastosis, and abnormally high precursor cells in the blood, known as excess circulating blasts. In addition, impaired blood production from the bone marrow causes blood production to shift to other organs such as the spleen and liver, known as extramedullary hematopoiesis, which leads to an enlarged spleen, known as splenomegaly, or an enlarged liver, known as hepatomegaly. MF patients can also suffer from debilitating constitutional symptoms, such as drenching night sweats, fatigue, severe itching, known as pruritus, fever and bone pain. The estimated prevalence of MF in the United States is approximately 13,000 patients, with an annual incidence of approximately 3,000 patients. Approximately 70% of MF patients have two to three risk factors (intermediate-2) or four or more risk factors (high risk), as defined by the Dynamic International Prognostic Scoring System Plus, or DIPSS Plus, described in a 2011 Journal of Clinical Oncology article. These patients have a median survival of approximately one to three years, representing a significant unmet medical need.
Allogeneic hematopoietic cell transplantation, or allo-HCT, is the only current treatment approach for MF that can lead to complete remission of the disease with normalization of peripheral blood counts, regression of bone marrow fibrosis, disappearance of cytogenetic abnormalities, normalization of spleen size and resolution of constitutional symptoms. However, use of allo-HCT is limited to a very small number of eligible patients due to the lack of suitable donors, older age and/or comorbid conditions. In addition, graft vs. host disease and life-threatening infections are other limitations of allo-HCT treatment.
Recent data presented in December 2014 at the American Society of Hematology, or ASH, Annual Meeting by Dr. Ron Hoffman of the Mount Sinai School of Medicine from in vitro translational studies have demonstrated that imetelstat inhibits malignant hematopoiesis and malignant megakaryopoiesis. In one study, hematopoietic stem cells were obtained from spleens of MF patients and normal cord blood. Imetelstat treatment on both in vitro cultures of stem cells showed selective inhibition of the proliferation of hematopoietic stem cells and myeloid progenitor cells and preferential depletion of malignant hematopoietic progenitor cells. In another study, peripheral blood mononuclear cells, or PBMCs, were taken from MF patients and normal patients. Imetelstat treatment on both in vitro cultures of cells showed selective inhibition of the proliferation of malignant megakaryocytic progenitor cells from myelofibrosis PBMCs; a reduction in the number of malignant megakaryocytes from myelofibrosis PBMCs; and inhibition of late-stage megakaryocytic maturation derived from both myelofibrosis and normal PBMCs. These in vitro data support the clinical remission responses observed to date in the investigator-initiated clinical trial of myelofibrosis being conducted at Mayo Clinic, or the MF Pilot Study, and the disease-modifying activity suggested by the MF Pilot Study results.
Pilot Study in Myelofibrosis (MF Pilot Study)
Based on the data from the Phase 2 ET trial, in November 2012, Dr. Ayalew Tefferi, or the investigator, initiated the MF Pilot Study to assess the effect of imetelstat in patients with MF. The MF Pilot Study is an open label trial in patients with primary MF, post-ET MF, or post-PV MF who have two to three risk factors (intermediate-2) or four or more risk factors (high risk) as defined by DIPSS Plus. In the MF Pilot Study, imetelstat is administered as a single agent over a two hour intravenous infusion to patients in multiple patient cohorts. In the first cohort, Cohort A, imetelstat is given once every three weeks. In the second cohort, Cohort B, imetelstat is given weekly for four weeks, followed by one dose every three weeks. Under the protocol, patients in Cohorts A and B may receive an intensified dosing regimen, up to once per week, after the initial six cycles of treatment. The starting dose of imetelstat in Cohorts A and B is 9.4 mg/kg, with dose reductions and dose holds allowed for toxicity. The primary endpoint in the MF Pilot Study is overall response rate, which is defined by the
8
proportion of patients who are classified as responders, which means that they have achieved either a clinical improvement, or CI, partial remission, or PR, or complete remission, or CR, consistent with the criteria of the 2013 International Working Group for Myeloproliferative Neoplasms Research and Treatment, or IWG-MRT criteria, described in a 2013 Blood article. Secondary endpoints include reduction of spleen size by palpation, improvement in anemia or inducement of red blood cell transfusion independence, safety and tolerability.
In January 2014, Mayo Clinic closed the MF Pilot Study to new patient enrollment. Mayo Clinic's notification informing us of its decision to cease new patient enrollment did not indicate any concerns regarding efficacy or safety. In March 2014, we were informed by Mayo Clinic that the investigator's IND for the MF Pilot Study was placed on partial clinical hold by the FDA due to a safety signal of hepatotoxicity that was identified in Geron's Phase 2 clinical trials of imetelstat and that it was unknown if this hepatotoxicity was reversible. In order to resolve the partial clinical hold, the investigator was required to provide follow-up information regarding reversibility of hepatotoxicity for all patients who received imetelstat in the MF Pilot Study. The investigator submitted a complete response to the FDA to seek release of the partial clinical hold, and the partial hold was removed by the FDA in June 2014.
On July 31, 2014, we entered into an agreement with Mayo Clinic under which Mayo Clinic and the investigator agreed to transfer to us certain data and information from the MF Pilot Study, and agreed that we would assume full responsibility for the investigator's IND, as well as responsibility for the conduct of the MF Pilot Study as the trial sponsor. In September 2014, the investigator's IND, under which the MF Pilot Study has been conducted, was transferred to us and we assumed responsibility for the MF Pilot Study as the trial sponsor. Dr. Tefferi continues as the principal investigator for the trial. As of December 5, 2014, 23 patients out of the 80 patients enrolled in the MF Pilot Study continue to receive imetelstat treatment, which includes 17 out of 62 patients with MF, five out of nine patients with refractory anemia with ringed sideroblasts, or RARS, a subpopulation of MDS, and one out of nine patients with blast-phase MF.
Prior to initiation of the Initial Phase 2 MF Study, we plan to transfer the IND for the MF Pilot Study, as well as responsibility for the conduct of the MF Pilot Study as the trial sponsor, to Janssen, and they do not intend to enroll additional patients in the MF Pilot Study. The remaining patients in the MF Pilot Study will continue to receive imetelstat treatment and Janssen will continue to collect data and information from the MF Pilot Study. For further discussion of the collaboration with Janssen under the Collaboration Agreement, see the sub-section entitled, "Future Development of Imetelstat in Collaboration with Janssen".
Updated Preliminary Data from MF Pilot Study
In December 2014, the investigator presented updated preliminary efficacy and safety data (as of September 10, 2014) from Cohorts A and B of the MF Pilot Study (n=33) at the 2014 ASH Annual Meeting. The data presented in December 2014 updated the investigator's previous analysis from the preliminary data he had presented at ASH in December 2013. We believe that the updated preliminary data from the MF Pilot Study continue to suggest that imetelstat has disease-modifying activity in MF, with remissions that have been durable (median 11.1 months; range 6.9 months - 16.2 months as of September 10, 2014). The investigator reported that no new safety signals had been observed and myelosuppression continued to be the principal dose-limiting toxicity.
9
Patient Demographics and Status
Below is a table setting forth the demographics of the first 33 patients enrolled in the MF Pilot Study, including certain disease characteristics and exposure to any prior treatments:
| |
Total (n=33) | |
|---|---|---|
Median Age (range; years) |
67.0 (53.0 - 79.0) | |
Male |
22 (66.7%) | |
Myelofibrosis Subtype |
||
Primary |
18 (54.5%) | |
Post-ET |
5 (15.2%) | |
Post-PV |
10 (30.3%) | |
DIPSS-plus Risk Status |
||
Intermediate-2 Risk |
16 (48.5%) | |
High Risk |
17 (51.5%) | |
Previously Treated |
26 (78.8%) | |
Median # of Prior Treatments (range) |
2 (1 - 6) | |
Prior JAK inhibitors |
19 (57.6%) | |
Abnormal Karyotype |
16 (48.5%) | |
Unfavorable Karyotype per DIPSS-plus |
6 (18.2%) | |
Transfusion Dependent |
13 (39.4%) | |
Constitutional Symptoms± |
21 (63.6%) | |
Palpable Splenomegaly |
23 (69.7%) | |
Median (range; cm) |
15.0 (5.0 - 33.0) |
± DIPSS+ assessment of symptoms at baseline: Includes unexplained persistent fever greater than 38.3°C (or greater than 101°F) during the past six months, unexplained non-menopausal night sweats during past six months, unexplained weight loss greater than 10% body weight in the previous six months and unexplained, non-articular bone pain during past six months.
As of September 10, 2014, the median duration of treatment was 11 cycles (range two cycles - 21 cycles). Median time on treatment was 14.3 months (range 6.5 months - 18.9 months) for patients with a CR, PR or CI response. All other patients had a median time on treatment of 6.9 months (range 1.4 months - 16.4 months).
10
Of these 33 patients, a total of nine patients remained on imetelstat treatment as of September 10, 2014. The following table describes patient status and reason for treatment discontinuation for each of the 33 patients, as reported by the investigator.
Patient Status and Reason for Treatment Discontinuation
|
Total (n=33) | |
|---|---|---|
On Treatment |
9 (27.3%) | |
Discontinued Treatment: |
24 (72.7%) | |
Stable Disease but Insufficient Response |
15 (45.5%) | |
Disease Progression/Relapse |
4 (12.1%) | |
Death(1) |
2 (6.1%) | |
Adverse Event/Side Effects/Complications(2) |
2 (6.1%) | |
Other Complicating Disease(3) |
1 (3.0%) |
- (1)
- One
death due to upper gastrointestinal hemorrhage (deemed unrelated to imetelstat per investigator assessment), the other due to intracranial hemorrhage
with febrile neutropenia after prolonged myelosuppression (deemed possibly related to imetelstat per investigator assessment).
- (2)
- One
case of thrombocytopenia and the other persistent thrombocytopenia.
- (3)
- Pre-existing problems with atrial fibrillation.
Updated Efficacy Data
The following table presents Geron's analysis of updated efficacy data as of September 10, 2014 for the first 33 eligible patients enrolled in the MF Pilot Study, using the IWG-MRT criteria:
Best Response by IWG-MRT
|
Total (n=33) | |
|---|---|---|
Overall Response (CR+PR+CI) |
12 (36.4%) | |
Complete Remission (CR) |
4 (12.1%) | |
Partial Remission (PR) |
3 (9.1%) | |
Clinical Improvement (CI) by Anemia |
1 (3.0%) | |
Clinical Improvement (CI) by Spleen |
4 (12.1%) | |
Stable Disease (SD) |
21 (63.6%) |
Median onset of remission occurred at five cycles (range one cycle - nine cycles). As of September 10, 2014, six of seven CR/PR patients remained in remission with median duration of 11.1 months (range 6.9 months - 16.2 months). All four CR patients achieved reversal of bone marrow fibrosis including three with complete molecular response. Three CR/PR patients who were transfusion dependent at baseline became transfusion independent. Three CR/PR patients with splenomegaly at baseline achieved splenic response.
Additional efficacy results reported by the investigator included spleen response, transfusion independence and resolution of circulating blasts, leukoerythroblastosis, marked leukocytosis and thrombocytosis:
- •
- Eight of 23 (34.8%) patients with splenomegaly achieved spleen responses by palpation, which is defined as either greater than or equal to 50% decrease if the baseline is greater than or equal to 10 centimeters or becoming non palpable if baseline is five to less than 10 centimeters. The median spleen size at baseline was 15 centimeters below the left costal margin (range five centimeters - 33 centimeters).
11
- •
- Four of 13 patients (30.8%) who were transfusion dependent at baseline became transfusion independent which is defined as absence of
any packed red blood cells transfusions during any consecutive 12-week interval with a hemoglobin level of ³8.5 grams per deciliter.
- •
- 17 of 21 (81.0%) patients with circulating blasts, or immature cells, at baseline achieved complete (n=14, 66.7%) or partial (n=3,
14.3%) resolution.
- •
- 22 of 27 (81.5%) patients with leukoerythroblastosis, a condition characterized by circulating immature granulocytes and nucleated red
blood cells, achieved complete (n=13, 48.1%) or partial (n=9, 33.3%) resolution.
- •
- Eight of 10 (80.0%) patients with marked leukocytosis, a condition characterized by very elevated white blood cell counts, achieved
complete (n=3, 30.0%) or partial (n=5, 50.0%) resolution.
- •
- 11 of 11 (100.0%) patients with thrombocytosis, a condition characterized by high platelet counts in blood, achieved complete (n=10, 90.9%) or partial (n=1, 9.1%) resolution.
Updated Safety Data
The following table sets forth the non-hematologic adverse events as of September 10, 2014, which were generally mild to moderate and not dose-limiting, for the first 33 eligible patients enrolled in the MF Pilot Study:
| |
All (n=33) | Related(1) (n=33) | ||
|---|---|---|---|---|
Fatigue |
3 (9.1%) | — | ||
APTT |
2 (6.1%) | — | ||
Atrial Fibrillation |
2 (6.1%) | — | ||
Heart Failure |
2 (6.1%) | — | ||
Hyperkalemia |
2 (6.1%) | — | ||
Ejection Fraction Decreased |
1 (3.0%) | — | ||
Intracranial Hemorrhage(2) |
1 (3.0%)(3) | 1 (3.0%)(3) | ||
Febrile Neutropenia |
1 (3.0%)(3) | 1 (3.0%)(3) | ||
Upper GI Hemorrhage(2) |
1 (3.0%) | — | ||
Hyponatremia |
1 (3.0%) | — | ||
Lipase Increased |
1 (3.0%) | — | ||
Lung Infection |
1 (3.0%) | — | ||
Pain |
1 (3.0%) | — | ||
Pyoderma Gangrenosum(4) |
1 (3.0%) | — | ||
Small Intestinal Obstruction |
1 (3.0%) | — |
- (1)
- Deemed
possibly related to imetelstat per investigator assessment.
- (2)
- Grade
5 event.
- (3)
- Same
patient.
- (4)
- The pyoderma gangrenosum is associated with a post-operative complication of a splenectomy, or spleen removal.
12
The following table sets forth all hematologic adverse events greater than or equal to Grade 3 as of September 10, 2014 for the first 33 patients enrolled in the MF Pilot Study.
| |
Worst Grade* | Cohort A (n=19) |
Cohort B (n=14) |
Total (n=33) |
||||
|---|---|---|---|---|---|---|---|---|
Thrombocytopenia |
3 | 8 (42.1%) | 1 (7.1%) | 9 (27.3%) | ||||
|
4 | 2 (10.5%) | 5 (35.7%) | 7 (21.2%) | ||||
Neutropenia |
3 | 4 (21.1%) | 2 (14.3%) | 6 (18.2%) | ||||
|
4 | 2 (10.5%) | 4 (28.6%) | 6 (18.2%) | ||||
Anemia |
3 | 7 (36.8%) | 9 (64.3%) | 16 (48.5%) | ||||
|
4 | — | — | — | ||||
Leukopenia |
3 | 3 (15.8%) | 6 (42.9%) | 9 (27.3%) | ||||
|
4 | 2 (10.5%) | 1 (7.1%) | 3 (9.1%) |
- *
- Hematologic toxicity is defined as worsening in grade after baseline.
The following table sets forth hematologic adverse events related to imetelstat as reported by the investigator lasting greater than or equal to four weeks as of September 10, 2014. These events mainly were observed in a small number of patients who received weekly dosing initially.
| |
|
Cohort A (n=19) | Cohort B (n=14) | Total (n=33) | ||||
|---|---|---|---|---|---|---|---|---|
Grade 3/4 Laboratory Finding Lasted ³ 4 Weeks |
Thrombocytopenia | 5 (26.3%) | 3 (21.4%) | 8 (24.2%) | ||||
|
Neutropenia | 1 (5.3%) | 2 (14.3%) | 3 (9.1%) | ||||
|
Either | 5 (26.3%) | 5 (35.7%) | 10 (30.3%) | ||||
Grade 4 Laboratory Finding Lasted ³ 4 Weeks |
Thrombocytopenia | 0 | 1 (7.1%) | 1 (3.0%) | ||||
|
Neutropenia | 1 (5.3%) | 1 (7.1%) | 2 (6.1%) | ||||
|
Either | 1 (5.3%) | 2 (14.3%) | 3 (9.1%) |
To mitigate the risk of severe, persistent cytopenias, the protocol for the MF Pilot Study was amended to raise the hematologic threshold for retreatment and include more stringent monitoring and dose adjustment criteria. Since then, no further episodes of significant bleeding events associated with thrombocytopenia, or infections associated with neutropenia, or additional episodes of febrile neutropenia have been reported to us by the investigator. As a result, we believe that the myelosuppressive effect of the drug may be manageable through dose hold rules and dose modifications.
Since the MF Pilot Study is ongoing, additional data from the remaining patients enrolled in the MF Pilot Study continues to be generated and is not reflected in the data discussed above. In this regard, additional and updated safety and efficacy data generated from the MF Pilot Study may be materially different from the data discussed above. Additional or updated data from the MF Pilot Study are also subject to any review or verification procedures that Janssen may conduct as the trial sponsor for the MF Pilot Study after it assumes responsibility for the conduct of the MF Pilot Study, and since this could result in material differences from the data reported by the investigator or us, additional or updated data that may be reported from the MF Pilot Study should be considered carefully and with caution. Analyses performed by Janssen after it becomes the sponsor of the MF Pilot Study may result in conclusions that are materially different from the investigator's analyses or ours, and therefore preliminary data should be considered carefully and with caution. As such, final data from the MF Pilot Study may be materially different from the data discussed above. Accordingly, the data discussed above should be considered carefully and with caution. Please refer to the risk factor
13
entitled "Risks Related to Clinical and Commercialization Activities—Success in early clinical trials may not be indicative of results in subsequent clinical trials. Likewise, preliminary data reported by investigators from time-to-time are subject to review and verification procedures that could result in material differences to final data and may change as more patient data become available" under Part I, Item 1A, "Risk Factors," of this annual report on Form 10-K.
Future Development of Imetelstat in Collaboration with Janssen
On November 13, 2014, we entered into the Collaboration Agreement with Janssen to develop and commercialize imetelstat worldwide for all indications in oncology, including hematologic myeloid malignancies, and all other human therapeutic uses. The Collaboration Agreement became effective on December 15, 2014, and we received $35 million from Janssen as an upfront payment.
Under the Collaboration Agreement, we granted to Janssen exclusive worldwide rights to develop and commercialize imetelstat for all indications, and Janssen is responsible for the development, manufacturing and commercialization of, and seeking regulatory approval for, imetelstat worldwide. Development of imetelstat will proceed under a mutually agreed clinical development plan, which includes the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study. We expect Janssen to initiate the Initial Phase 2 MF Study in mid-2015, followed later by the Initial Phase 2 MDS Study to be initiated at the end of 2015. In addition, the clinical development plan may also include possible registration studies in MF and MDS, and possible exploratory Phase 2 and potential follow-on Phase 3 studies in AML. Development costs for the planned Initial Phase 2 MF Study and the planned Initial Phase 2 MDS Study will be shared between the parties on a 50/50 basis.
Following the protocol-specified primary analysis of the Initial Phase 2 MF Study, which results are referred to in this annual report on Form 10-K as the Initial Phase 2 MF Results, or a certain time period after the initiation of the first Phase 3 MF study, Janssen must notify us of their decision, or a Continuation Decision, as to whether they elect to maintain the license rights granted to them under the Collaboration Agreement and continue to advance the development of imetelstat in any indication. In the event that the Initial Phase 2 MF Study has been terminated early or suspended, Janssen must instead notify us of their Continuation Decision by the date that is the later of 24 months after the initiation of the planned Initial Phase 2 MDS Study or 24 months after the termination of the Initial Phase 2 MF Study or commencement of the suspension period, as applicable.
In the event that Janssen notifies us of an affirmative Continuation Decision, we will then have an option to share further U.S. development and promotion costs, or the U.S. Opt-In Rights, in exchange for higher tiered royalty rates and higher future potential milestone payments if imetelstat is successfully developed and approved. If we exercise the U.S. Opt-In Rights, then we and Janssen will share U.S. development and promotion costs on a 20/80 basis (Geron 20%, Janssen 80%), we will receive a $65 million milestone payment at the time of the Continuation Decision, and will be eligible to receive additional potential payments of up to $470 million in development and regulatory milestones, up to $350 million in sales milestones, and tiered royalties ranging from a mid-teens up to a low twenties percentage rate on worldwide net sales of imetelstat in any countries where regulatory exclusivity exists or there are valid claims under the patent rights exclusively licensed to Janssen. In addition, if we exercise the U.S. Opt-In Rights, we will also have a separate co-promotion option, or the U.S. Co-Promotion Option, to provide 20% of the U.S. selling effort with sales force personnel, in lieu of funding 20% of U.S. promotion costs, upon regulatory approval and commercial launch of imetelstat in the United States. Such co-promotion would be conducted under a Janssen prepared promotion plan, and in accordance with a co-promotion agreement to be agreed by us and Janssen at the time of our exercise of the U.S. Co-Promotion Option. We would be responsible for all costs associated with establishing and maintaining our sales force in any conduct of such co-promotion. All product sales would be booked by Janssen. If we do not exercise the U.S. Opt-In Rights upon an affirmative Continuation Decision by Janssen, then all further development and promotion costs
14
beyond the Initial Phase 2 MF Study or Initial Phase 2 MDS Study will be borne by Janssen, we will receive a $65 million milestone payment at the time of the Continuation Decision plus a $70 million payment for Janssen's retention of full U.S. rights, and will be eligible to receive additional potential payments of up to $415 million in development and regulatory milestones, up to $350 million in sales milestones, and tiered royalties ranging from a double-digit up to a mid-teens percentage rate on worldwide net sales in any countries where regulatory exclusivity exists or there are valid claims under the patent rights exclusively licensed to Janssen.
Under the terms of the Collaboration Agreement, we and Janssen have created a joint governance structure, including joint development and steering committees and working groups, to oversee and manage worldwide regulatory, development and manufacturing work under the joint clinical development plan and promotional activities (assuming we exercise the U.S. Opt-In Rights) for imetelstat, with Janssen responsible for the operational implementation of those activities. In addition, both we and Janssen may propose to the joint development committee imetelstat development for any new indications not then provided for in the joint clinical development plan and if we and Janssen agree such development should be conducted outside of the joint clinical development plan, both we and Janssen would be entitled to independently undertake such development at the developing party's own cost, subject to the other party's obligation to provide reimbursement for its specified portion of the development costs plus a premium following marketing approval of imetelstat in such newly proposed indication as a result of such independent development. In the event that we do not exercise the U.S. Opt-In Rights following Janssen's Continuation Decision, the joint governance structure under the Collaboration Agreement would be dissolved, a joint oversight committee would monitor the progress of the collaboration, and we would have no further rights to conduct any independent imetelstat development.
Research and Development
Since our inception, we have devoted a significant amount of resources to develop our current and former product candidates. For information regarding research and development expenses incurred during 2014, 2013 and 2012, see Item 7, "Management's Discussion and Analysis of Financial Condition and Results of Operations—Research and Development Expenses".
In light of projected reduced operational demands as a result of the Collaboration Agreement with Janssen, on March 3, 2015, we announced an organizational resizing to reduce our workforce from 39 to 21 positions, representing a reduction of approximately 46% of our workforce. As a result of this action, we expect personnel related research and development expenses to decrease in the future. For a further discussion, see Note 15 on Subsequent Event in Notes to Consolidated Financial Statements of this annual report on Form 10-K.
Intellectual Property
Intellectual property, including patent protection, is very important to our business. We file patent applications in the United States and other jurisdictions, and we also rely on trade secret protection and contractual arrangements to protect aspects of our business. An enforceable patent with appropriate claim coverage can provide an advantage over competitors who may seek to employ similar approaches to develop therapeutics, and so our future commercial success will be in part dependent on our intellectual property strategy. The information provided in this section should be reviewed in the context of the section entitled "Risks Related to Protecting Our Intellectual Property" under Item 1A, "Risk Factors".
The development of biotechnology products, including ours, typically includes the early development of a technology, followed by rounds of increasingly focused innovation around a product opportunity, including identification and definition of a specific product candidate and uses thereof,
15
manufacturing processes, product formulation and administration methods. The result of this process is that biotechnology products are often protected by several families of patent filings that are filed at different times during product development and cover different aspects of the product. Consequently, earlier filed, broad technology patents will usually expire ahead of patents covering later developments such as product formulations, so that patent expirations on a product may span several years. Patent coverage may also vary from country to country based on the scope of available patent protection. There are also opportunities to obtain extension of patent coverage for a product in certain countries, which add further complexity to the determination of patent life.
We endeavor to monitor worldwide patent filings by third parties that are relevant to our business. Based on this monitoring, we may determine that an action is appropriate to protect our business interests. Such actions may include negotiating patent licenses where appropriate, filing oppositions or reexaminations against a patent, or filing a request for the declaration of an interference with a United States patent application or issued patent.
Imetelstat
The following table shows the estimated latest expiration dates for the composition of matter patents for our sole product candidate, imetelstat. Composition of matter patents generally provide the most material coverage, and therefore may convey competitive advantages. Because imetelstat is still under development, subsequent innovation and associated patent filings may provide additional patent coverage with later expiration dates. Examination of overseas patent applications typically lags behind U.S. examination particularly where cases are filed first in the United States. The stated U.S. expiration date includes a patent term adjustment for delays in prosecution by the U.S. Patent and Trademark Office, but does not account for a potential patent term extension that may be available to compensate us for delays in FDA regulatory review of a new drug application.
Product Candidate
|
U.S. Patent Status / Expiration Date |
Europe Patent Status / Expiration Date |
Japan Patent Status / Expiration Date |
|||
|---|---|---|---|---|---|---|
Imetelstat |
Issued / 2025 | Issued / 2020* | Issued / 2024 |
- *
- An additional composition of matter patent application for imetelstat has been filed that, if issued, would provide European patent protection until 2024.
Our patent rights relating to imetelstat which have been exclusively licensed to Janssen for all disorders or medical conditions include those covering the nucleic acid sequence of hTR, the RNA component of telomerase, against which the oligonucleotide component of imetelstat is targeted; composition claims to the drug molecule and related telomerase inhibiting molecules; the amidate nucleic acid chemistry used in the oligonucleotide; as well as manufacturing processes for the drug, and method of treatment and kit claims, certain of which are co-owned by us. Our proprietary nucleic acid chemistry is covered by patent families that we acquired in 2002 from Lynx Therapeutics, Inc., as well as in patents that we filed for further developments of this chemistry. Certain of our patent rights for measuring the expression of telomerase activity or the length of telomeres in cells have been non-exclusively licensed to Janssen.
As noted previously, we granted to Janssen exclusive worldwide rights to develop and commercialize imetelstat for all disorders or disease conditions. Under the terms of the Collaboration Agreement with Janssen, we remain responsible for prosecuting, at Janssen's direction, the patents exclusively licensed to Janssen, with costs shared between us and Janssen on a 50/50 basis. For intellectual property developed under the Collaboration Agreement, the party having sole ownership interest in such intellectual property will be responsible for prosecuting any such patents, with Janssen bearing all of the patent costs for such intellectual property solely owned by Janssen and with patent costs for such intellectual property either jointly owned or solely owned by us shared between the parties on a 50/50 basis.
16
Telomerase
Our patent rights relating to telomerase that cover the cloned genes that encode the catalytic protein component (hTERT) of human telomerase and cells that are immortalized by expression of recombinant hTERT are co-owned with and in-licensed exclusively from the University of Colorado. Certain patents for identifying telomerase modulators or diagnosing cancer by measuring the expression of telomerase activity are co-owned and in-licensed from the University of Texas Southwestern Medical Center and the University of California.
Licensing
In addition to the Collaboration Agreement with Janssen (see the section entitled "Future Development of Imetelstat in Collaboration with Janssen" above for further discussion of the Collaboration Agreement with Janssen), we have also granted licenses to a number of other organizations in the ordinary course of our business to utilize aspects of our technologies to develop and commercialize products outside of our imetelstat program. These include:
- •
- licenses to several biotechnology and pharmaceutical companies to use telomerase immortalized cells in drug discovery research;
- •
- licenses to several companies to commercialize telomerase immortalized cells for drug discovery applications;
- •
- licenses to several companies to sell antibodies specific to telomerase for research purposes;
- •
- licenses to several companies to develop and commercialize reagent kits, or to provide services, for the measurement of telomere
length or telomerase activity for research purposes;
- •
- a license to a company to develop and commercialize a particular telomerase based technology for cancer detection; and
- •
- a license to a company for the development of cancer immunotherapies for veterinary applications.
Competition
The pharmaceutical and biotechnology industries are intensely competitive. Other pharmaceutical and biotechnology companies and research organizations currently engage in or have in the past engaged in efforts related to the biological mechanisms related to imetelstat, including the study of telomeres, telomerase and our proprietary oligonucleotide chemistry, and the research and development of therapies for the treatment of hematologic myeloid malignancies. In addition, other products and therapies that could compete directly with imetelstat currently exist or are being developed by pharmaceutical and biopharmaceutical companies and by academic institutions, government agencies and other public and private research organizations.
Our sole product candidate, imetelstat, if approved for marketing, will face significant competition from approved drugs, drugs currently under development and any other drugs that may be subsequently approved. Imetelstat would have to compete successfully based on efficacy, safety, convenience, price, cost-effectiveness and other relevant factors. In addition, imetelstat would have to compete against other drugs with a variety of decision makers, including physicians, patients, government and private third-party payors, technology assessment groups and patient advocacy organizations. We cannot guarantee that we, in collaboration with Janssen, will be able to compete successfully on any of these factors. If we or Janssen cannot compete successfully on any of the factors described previously, Janssen may terminate the Collaboration Agreement and our business may fail.
17
Imetelstat is likely to be in highly competitive markets. We are aware of products in research or development by our competitors that address the diseases we are targeting, and any of these products may compete with imetelstat. Our competitors may succeed in developing their products before we do, obtaining approvals from the FDA or other regulatory agencies for their products more rapidly than we do, or developing products that are more effective than imetelstat. These products or technologies might render our technology obsolete or noncompetitive. There may also be product candidates of which we are not aware at an earlier stage of development that may compete with imetelstat. In addition, imetelstat may need to compete or combine with existing therapies, many with long histories of use.
Many companies are developing alternative therapies to treat hematologic myeloid malignancies and, in this regard, are competitors of ours and Janssen. For example, if approved for commercial sale for the treatment of MF, imetelstat would compete against Incyte Corporation's ruxolitinib, or Jakafi®, which is orally administered. In clinical trials, Jakafi® reduced spleen size, abdominal discomfort, early satiety, bone pain, night sweats and itching in MF patients. Recently, there have also been reports of overall survival benefit as well as improvement in bone marrow fibrosis from Jakafi® treatment. Other treatment modalities for MF include:
- •
- hydroxyurea for the management of splenomegaly, leukocytosis, thrombocytosis and constitutional symptoms;
- •
- splenectomy and splenic irradiation for the management of splenomegaly and co-existing cytopenias, or low blood cell counts; and
- •
- chemotherapy and pegylated interferon.
Drugs for the treatment of MF-associated anemia include erythropoiesis-stimulating agents, androgens, danazol, corticosteroids, thalidomide and lenalidomide. There are other investigational treatments further advanced in development than imetelstat, such as momelotinib by Gilead Sciences, Inc. and pacritinib by Cell Therapeutics, Inc., which are currently in Phase 3 clinical trials, and other inhibitors of the JAK-STAT pathway, as well as several investigational treatments in early phase testing such as histone deacetylase inhibitors, inhibitors of heat shock protein 90, hypomethylating agents, PI3 Kinase and mTOR inhibitors, hedgehog inhibitors, anti-LOX2 inhibitors, recombinant pentraxin 2 protein, KIP-1 activators, TGF-beta inhibitors, FLT inhibitors, and other tyrosine kinase inhibitors.
Independently, Janssen is developing therapies for hematologic malignancies, including AML, MDS, MM and ABC-subtype diffuse large B-cell lymphoma. Molecular and cellular pathways of interest include:
- •
- cell surface targets for immune-directed therapy;
- •
- immune checkpoint inhibition;
- •
- leukemia stem cells;
- •
- pathway addiction (genetic alterations, cell-type specific pathways);
- •
- conditional sensitivity (stress, protein-producing tumors);
- •
- targeting of T-cells and natural killer "NK" cells to tumors;
- •
- identification of novel tumor-specific antigens; and
- •
- progression from early MDS to AML and cancer interception.
18
Success by Janssen in any of these approaches may compete with imetelstat or render imetelstat obsolete or noncompetitive. A decision by Janssen to discontinue the imetelstat program and terminate the Collaboration Agreement would materially and adversely affect our business and business prospects.
Smaller companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. We anticipate increased competition in the future as new companies explore treatments for hematologic myeloid malignancies, which may significantly impact the commercial viability of imetelstat. Academic institutions, government agencies and other public and private research organizations may also conduct research, seek patent protection and establish collaborative arrangements for research, clinical development and marketing of products similar to ours. These companies and institutions compete with us in recruiting and retaining qualified scientific and management personnel as well as in acquiring technologies complementary to our imetelstat program.
In addition to the above factors, we and Janssen expect to face competition in the following areas:
- •
- product efficacy and safety;
- •
- convenience of product administration;
- •
- cost of manufacturing;
- •
- the timing and scope of regulatory consents;
- •
- status of reimbursement coverage;
- •
- price; and
- •
- patent position, including potentially dominant patent positions of others.
As a result of the foregoing, competitors may develop more commercially desirable or affordable products, or achieve earlier patent protection or product commercialization than us or Janssen. Competitors have developed, or are in the process of developing, technologies that are, or in the future may be, competitive to imetelstat. Some of these products may have an entirely different approach or means of accomplishing therapeutic effects similar to those that may be demonstrated by imetelstat. Competitors may develop products that are safer, more effective or less costly than imetelstat, or more convenient to administer to patients and, therefore, present a serious competitive threat to imetelstat. In addition, competitors may price their products below what Janssen may determine to be an acceptable price for imetelstat, may receive better third-party payor coverage and/or reimbursement, or may be more cost effective than imetelstat. Such competitive products or activities by competitors may render imetelstat obsolete, which may cause Janssen to terminate the Collaboration Agreement and which would severely and adversely affect our business prospects.
Government Regulation
Regulation by governmental authorities in the United States and other countries is a significant factor in the development, manufacture and marketing of our sole product candidate, imetelstat, in collaboration with Janssen. We anticipate that imetelstat will require regulatory approval by governmental agencies prior to commercialization. In particular, potential human therapeutic products, such as imetelstat, are subject to rigorous preclinical and clinical testing and other approval procedures of the FDA and similar regulatory authorities in European and other countries. Various governmental statutes and regulations also govern or influence testing, manufacturing, safety, labeling, storage and recordkeeping related to such products and their marketing. In collaboration with Janssen, the process of obtaining these approvals and the subsequent compliance with appropriate statutes and regulations require the expenditure of substantial time and money, and there can be no guarantee that approvals will be granted.
19
United States Food and Drug Administration Regulatory Approval Process
Prior to commencement of clinical trials involving humans, preclinical testing of new pharmaceutical products is generally conducted on animals in the laboratory to evaluate the potential efficacy and safety of a product candidate. The results of these trials are submitted to the FDA as part of an IND application, which must be cleared by the FDA before clinical testing in humans can begin. Typically, clinical evaluation involves a time consuming and costly three phase trial process. In Phase 1, clinical trials are conducted with a small number of healthy volunteers or patients afflicted with a specific disease to assess safety and to evaluate the pattern of drug distribution and metabolism within the body. In Phase 2, clinical trials are conducted with groups of patients afflicted with a specific disease in order to determine preliminary efficacy, optimal dosages and expanded evidence of safety. The Phase 2 trials can be conducted comparing the investigational treatment to a comparator arm, or not. If used, a comparator usually includes standard of care therapy. Safety and efficacy data from Phase 2 clinical trials, even if favorable, may not provide sufficient rationale for proceeding to a Phase 3 clinical trial. In Phase 3, large scale, multi center, comparative trials are conducted with patients afflicted with a target disease to provide sufficient data to demonstrate the efficacy and safety required by the FDA. The FDA closely monitors the progress of each of the three phases of clinical testing and may, at its discretion, re-evaluate, alter, suspend, or terminate the trials.
The results of the preclinical and clinical testing of small molecules and many biologic drugs are submitted to the FDA in the form of a New Drug Application, or NDA, for review and for approval prior to commencement of commercial sales. In the case of blood products, vaccines, or gene and cell therapies, the results of clinical trials are submitted to the FDA as a Biologics License Application, or BLA. In responding to an NDA/BLA submission, the FDA may grant a marketing authorization, impose limitations on a marketing authorization, request additional information, deny the application if it determines that the application does not provide an adequate basis for approval, or refuse to review an application that has been submitted if it determines that the application does not provide an adequate basis for filing and review.
European and Other Regulatory Approval Process
Prior to initiating clinical trials in a region outside of the United States, a clinical trial application must be submitted and reviewed by the appropriate regulatory authority regulating the country in which the trial will be conducted. Whether or not FDA clearance or approval has been obtained, approval of a product by comparable regulatory authorities in Europe and other countries is necessary prior to commencement of marketing the product in such countries. The regulatory authorities in each country may impose their own requirements and may refuse to grant an approval, or may require additional data before granting it, even though the relevant product has been cleared or approved by the FDA or another authority. As with the FDA, the regulatory authorities in the European Union, or EU, and other developed countries have lengthy approval processes for pharmaceutical products. The process for gaining approval in particular countries varies, but generally follows a similar sequence to that described for FDA approval. In Europe, the European Medicine Agency, or EMA, and the European Committee for Proprietary Medicinal Products, or CPMP, provide a mechanism for EU member states to exchange information on all aspects of product licensing. The EU has established the EMA for the evaluation of medical products, with both a centralized procedure with which the marketing authorization is recognized in all EU member states and a decentralized procedure, the latter being based on the principle of licensing within one member country followed by mutual recognition by the other member countries.
20
Other Regulations
We are also subject to various and often changing federal, state, local and international laws, rules, regulations, guidelines and recommendations relating to safe working conditions and manufacturing practices.
Manufacturing
A typical sequence of steps in the manufacture of imetelstat drug product includes the following key components:
- •
- starting materials, which are well-defined raw materials that are used to make bulk drug substance;
- •
- bulk drug substance, which is the active pharmaceutical ingredient in a drug product that provides pharmacological activity or other
direct effect in the treatment of disease; and
- •
- final drug product, which is the finished dosage form that contains the drug substance that is shipped to the clinic for patient treatment.
Under the Collaboration Agreement, after a transition period, Janssen will be responsible for the manufacture and/or supply of imetelstat on a global basis for clinical trials and, after any regulatory approval, all commercial activities. Consequently, we will be, and expect to remain, dependent on Janssen to appropriately supply imetelstat. Currently, third-party contractors perform certain process development and other technical and scientific work with respect to imetelstat, in addition to supplying starting materials and manufacturing drug substance and drug product. We or Janssen do not have direct control over their personnel or operations. These third-party contractors, and/or any other contractors that Janssen may rely upon for the manufacture and/or supply of imetelstat, typically complete their services on a proposal by proposal basis under master supply agreements and may need to make substantial investments to enable sufficient capacity increases and cost reductions, and to implement those regulatory and compliance standards necessary for successful Phase 3 clinical trials and commercial production. These third-party contractors, and/or any other contractors that Janssen may rely upon for the manufacture and/or supply of imetelstat, may not be able to achieve such capacity increases, cost reductions, or regulatory and compliance standards, and even if they do, such achievements may not be at a commercially reasonable cost. Neither we nor Janssen have any long-term commitments or commercial supply agreements with any of the third-party contractors for imetelstat.
We currently have a master service agreement with two third-party contractors for labeling and packaging of imetelstat final drug product and for distribution of imetelstat to clinical sites in North America. In addition, we have agreements with two third-party contractors for release and distribution of imetelstat drug product to clinical sites in Europe. These third-party contractors provide services on a proposal by proposal basis. If requested by Janssen, we may transfer or assign these agreements to Janssen to the extent permissible by the terms of the agreements or agreed with such third-party contractors.
We have also entered into quality agreements with our imetelstat bulk drug substance and final drug product manufacturers, and our labeling, packaging and distribution service providers. The master and quality agreements are designed to ensure product quality, compliance with current Good Manufacturing Practices, or cGMP, and oversight of third parties for all critical aspects of imetelstat production, testing, release, labeling and packaging, storage and distribution. If requested by Janssen, we may transfer or assign these agreements to Janssen to the extent permissible by the terms of the agreements or agreed with such third-party contractors.
21
Concentration of Revenues
In 2014 and 2013, the majority of our revenues were from license fees and royalties under licenses granted to several biotechnology and pharmaceutical companies to use telomerase immortalized cells in drug discovery research and for drug discovery applications. Two customers accounted for approximately 31%, 42% and 59% of our 2014, 2013 and 2012 revenues, respectively. In 2012, the majority of our revenues were from license fees and royalties related to our license and collaboration agreement with GE Healthcare UK, Limited, or GE Healthcare, for the development and commercialization of cellular assay products and our license agreement with Asia Biotech Corporation related to our telomerase activation technology. Upon the closing of the stem cell divestiture under the Contribution Agreement on October 1, 2013, the license agreement with GE Healthcare, including any future revenue payments thereunder, was transferred to Asterias Biotherapeutics, Inc. In December 2012, we assigned our telomerase activation technology to Telomerase Activation Sciences, Inc. and terminated our license agreement with Asia Biotech Corporation. Future royalty obligations by Asia Biotech Corporation under the license agreement have been terminated. We operate in one operating segment and have operations solely in the United States. All of our long-lived assets are maintained in the United States. Information regarding total revenues, net loss and total assets is set forth in our financial statements included in Item 8 of this annual report on Form 10-K.
Stem Cell Divestiture; Asterias Series A Distribution
Background
On October 1, 2013, we closed the transaction to divest our human embryonic stem cell assets and our autologous cellular immunotherapy program pursuant to the terms of the previously disclosed asset contribution agreement, or the Contribution Agreement, that we entered into in January 2013 with BioTime, Inc., or BioTime, and BioTime's wholly owned subsidiary, Asterias Biotherapeutics, Inc., or Asterias (formerly known as BioTime Acquisition Corporation) and received 6,537,779 shares of Asterias Series A common stock. In accordance with our contractual obligations under the Contribution Agreement, we distributed all of the shares of Asterias Series A common stock to our stockholders on a pro rata basis, or distributed cash in lieu thereof, which we refer to as the Series A Distribution. We completed the Series A Distribution to eligible stockholders on August 15, 2014 and have no remaining obligations for the Series A Distribution. See further discussion in Note 7 on Divestiture of Stem Cell Assets in Notes to Consolidated Financial Statements of this annual report on Form 10-K.
In connection with the Contribution Agreement, BioTime made certain contributions to Asterias, including five-year warrants to purchase 8,000,000 shares of BioTime common stock at an exercise price of $5.00 per share, or the BioTime Warrants. Upon the completion of the Series A Distribution, Asterias distributed the BioTime Warrants on a pro rata basis to the holders of Asterias Series A common stock. The BioTime Warrant distribution was completed on October 1, 2014.
Consultants
We have consulting agreements with a number of leading academic scientists, clinicians and regulatory experts. These individuals serve as key consultants, expert witnesses, or as members of clinical advisory panels with respect to our imetelstat program or in legal proceedings. They also serve as important contacts for us throughout the broader scientific and clinical communities. They are distinguished individuals with expertise in numerous fields, including telomere and telomerase biology, cellular biology, molecular biology, oncology and drug regulations.
We retain each consultant according to the terms of a consulting agreement. Under such agreements, we pay them a consulting fee and reimburse them for out-of-pocket expenses incurred in performing their services for us. In addition, some consultants hold options to purchase our common stock and restricted stock awards, subject to the vesting requirements contained in the consulting
22
agreements. Our consultants may be employed by other entities and therefore may have commitments to their employer, or may have other consulting or advisory agreements that may limit their availability to us.
Executive Officers of the Company
The following table sets forth certain information with respect to our executive officers as of January 31, 2015:
Name
|
Age | Position | |||
|---|---|---|---|---|---|
John A. Scarlett, M.D. |
63 | President and Chief Executive Officer | |||
Olivia K. Bloom |
46 | Executive Vice President, Finance, Chief Financial Officer and Treasurer | |||
Melissa A. Kelly Behrs |
51 | Executive Vice President, Business Development and Portfolio and Alliance Management | |||
Andrew J. Grethlein, Ph.D. |
50 | Executive Vice President, Development and Technical Operations | |||
Stephen N. Rosenfield, J.D. |
65 | Executive Vice President, General Counsel and Corporate Secretary | |||
John A. Scarlett, M.D., has served as our Chief Executive Officer and a director since September 2011 and President since January 2012. Prior to joining Geron, Dr. Scarlett served as President, Chief Executive Officer and a member of the board of directors of Proteolix, Inc., a privately held, oncology-oriented biopharmaceutical company, from February 2009 until its acquisition by Onyx Pharmaceuticals, Inc., an oncology-oriented biopharmaceutical company, in November 2009. From February 2002 until its acquisition by Ipsen, S.A. in October 2008, Dr. Scarlett served as the Chief Executive Officer and a member of the board of directors of Tercica, Inc., an endocrinology-oriented biopharmaceutical company, and also as its President from February 2002 through February 2007. From March 1993 to May 2001, Dr. Scarlett served as President and Chief Executive Officer of Sensus Drug Development Corporation. In 1995, he co-founded Covance Biotechnology Services, Inc., a contract biopharmaceutical manufacturing operation, and served as a member of its board of directors from inception to 2000. From 1991 to 1993, Dr. Scarlett headed the North American Clinical Development Center and served as Senior Vice President of Medical and Scientific Affairs at Novo Nordisk Pharmaceuticals, Inc., a wholly owned subsidiary of Novo Nordisk A/S. Dr. Scarlett received his B.A. degree in chemistry from Earlham College and his M.D. from the University of Chicago, Pritzker School of Medicine.
Olivia K. Bloom has served as our Executive Vice President, Finance since February 2014, Chief Financial Officer since December 2012 and Treasurer since February 2011. Ms. Bloom previously served as our Senior Vice President, Finance from December 2012 to February 2014, Chief Accounting Officer from September 2010 to December 2012 and Vice President, Finance from January 2007 to December 2012. Ms. Bloom joined the Company in 1994 as a Senior Financial Analyst and from 1996 to 2011 served as our Controller. Prior to Geron, Ms. Bloom started her career in public accounting at KPMG Peat Marwick and became a Certified Public Accountant in 1994. Ms. Bloom graduated Phi Beta Kappa with a B.S. in Business Administration from the University of California at Berkeley.
Melissa A. Kelly Behrs has served as our Executive Vice President, Business Development and Portfolio & Alliance Management, since July 2014. Prior to that she was our Executive Vice President, Portfolio and Alliance Management, since February 2014 and she was our Senior Vice President, Portfolio and Alliance Management, from September 2012 to February 2014. Ms. Behrs joined Geron in November 1998 as Director of Corporate Development. Since then, she has served in various managerial positions, including General Manager, R&D Technologies; Vice President, Corporate
23
Development; Senior Vice President, Therapeutic Development, Oncology; and Senior Vice President, Strategic Portfolio Management. From 1990 to 1998, Ms. Behrs worked at Genetics Institute, Inc., a biotechnology research and development company, serving initially as Assistant Treasurer and then as Associate Director of Preclinical Operations where she was responsible for all business development, regulatory, and project management activities for the Preclinical Development function. Ms. Behrs received a B.S. from Boston College and an M.B.A. from Babson College.
Andrew J. Grethlein, Ph.D., has served as our Executive Vice President, Development and Technical Operations, since July 2014. Prior to that he served as our Executive Vice President, Technical Operations, since September 2012. Prior to joining Geron, Dr. Grethlein was Executive Vice President and Chief Operating Officer for Inspiration Biopharmaceuticals, a biopharmaceutical company, from January 2010 to September 2012. From October 2008 until January 2010, Dr. Grethlein was Senior Vice President of Biotechnology and Portfolio Management Team Leader for Hematology at Ipsen S.A., a global specialty pharmaceutical company. His responsibilities at Ipsen included planning and execution of worldwide strategy for product and portfolio development in the hematologic therapeutic area. From 2003 to 2008, Dr. Grethlein served as Senior Vice President of Pharmaceutical Operations at Tercica, Inc., an endocrinology-oriented biopharmaceutical company. In this role, he was a member of the senior executive team that governed corporate strategy, business planning and company operations, and had responsibility for all manufacturing and quality functions. Before joining Tercica, Dr. Grethlein served in various positions at Elan Corporation, a biotechnology company, from 1997 to 2003, including as Senior Director, South San Francisco Pharmaceutical Operations, where he had responsibility as site head for commercial manufacturing operations. From 1995 to 1997, Dr. Grethlein served as Manager, Biologics Development and Manufacturing, for Athena Neurosciences, Inc., a pharmaceutical company. Prior to this, he served in various engineering positions for the Michigan Biotechnology Institute, a nonprofit technology research and business development corporation. Dr. Grethlein received his A.A. degree in liberal arts from Simon's Rock Early College, his B.S. in biology from Bates College, and his M.S. and Ph.D. in chemical engineering from Michigan State University.
Stephen N. Rosenfield, J.D., has served as our Executive Vice President, General Counsel and Corporate Secretary since February 2012, General Counsel and Secretary since January 2012 and Secretary since October 2011. From July 2009 to February 2012, Mr. Rosenfield has been a consultant to private companies. From October 2008 until June 2009, Mr. Rosenfield was the General Counsel and Secretary of Tercica, Inc., a U.S. subsidiary of Ipsen, S.A., a global specialty pharmaceutical company. From June 2004 until October 2008, Mr. Rosenfield was the General Counsel and Secretary of Tercica, Inc., an endocrinology-oriented biopharmaceutical company, from January 2006 until October 2008, he was also the Executive Vice President of Legal Affairs, and from June 2004 until January 2006, Mr. Rosenfield was the Senior Vice President of Legal Affairs. Prior to joining Tercica, Mr. Rosenfield served as the Executive Vice President of Legal Affairs, General Counsel and Secretary of InterMune, Inc., a biotechnology company focused in pulmonology and fibrotic diseases. Prior to joining InterMune, Mr. Rosenfield was an attorney at Cooley LLP, an international law firm, where he served as outside counsel for biotechnology and technology clients. Mr. Rosenfield received a B.S. from Hofstra University and a J.D. from Northeastern University School of Law.
Employees
As of December 31, 2014, we had 42 employees, of whom 7 hold Ph.D. degrees and 13 hold other advanced degrees. Of this current total workforce, 22 employees were engaged in, or directly supported, our research and development activities, and 20 employees were engaged in business development, legal, finance and administration. None of our employees are covered by a collective bargaining agreement; nor have we experienced work stoppages. We consider relations with our employees to be good.
On March 3, 2015, we announced an organizational resizing to reduce our workforce from 39 to 21 positions, representing a reduction of approximately 46% of our workforce. For a further discussion, see Note 15 on Subsequent Event in Notes to Consolidated Financial Statements of this annual report on Form 10-K.
24
Corporate Information
Geron Corporation was incorporated in the State of Delaware on November 28, 1990.
Available Information
Our internet address is www.geron.com. Information included on our website is not part of this annual report on Form 10-K. We make available free of charge on our website our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, and all amendments to those reports as soon as reasonably practicable after such material is electronically filed with or furnished to the SEC. In addition, copies of our annual reports are available free of charge upon written request. The SEC also maintains an Internet site that contains reports, proxy and information statements, and other information regarding issuers that file electronically with the SEC. The address of that site is www.sec.gov.
Our business is subject to various risks and uncertainties that may have a material adverse effect on our business, financial condition or results of operations. You should carefully consider the risks and uncertainties described below, together with all of the other information included in this annual report on Form 10-K. Our business faces significant risks and uncertainties, and those described below may not be the only risks and uncertainties we face. Additional risks and uncertainties not presently known to us or that we currently believe are immaterial may also significantly impair our business, financial condition or results of operations. If any of these risks or uncertainties occur, our business, results of operations or financial condition could suffer, the market price of our common stock could decline and you could lose all or part of your investment in our common stock.
We are dependent upon our collaborative relationship with Janssen to further develop, manufacture and commercialize imetelstat, our sole product candidate. If Janssen fails to perform as expected, the potential for us to generate future revenues from milestone payments and royalties from imetelstat would be significantly reduced, the development and/or commercialization of imetelstat may be terminated or substantially delayed, and our business would be severely harmed.
Under the terms of the Collaboration Agreement, we and Janssen have created a joint governance structure, including joint development and steering committees and working groups, to oversee and manage worldwide regulatory, development, manufacturing and commercialization activities for imetelstat; however, Janssen is solely responsible for the operational implementation of those activities. Accordingly, the timely and successful completion by Janssen of those activities will significantly affect the timing and amount of any revenues from milestone payments and royalties we may receive under the Collaboration Agreement, and these activities will be influenced by, among other things, the efforts and allocation of resources by Janssen, none of which we control. If Janssen does not perform in the manner we expect or fulfill its responsibilities in a timely manner, or at all, the clinical development, manufacturing, regulatory approval and/or commercialization efforts related to imetelstat could be delayed or terminated, and it could become necessary for us to assume the responsibilities for the clinical development, manufacturing, regulatory approval and/or commercialization of imetelstat at our own expense. Accordingly, there can be no assurance that any of the development, regulatory or sales milestones will be achieved or that we will receive any future milestone or royalty payments under the Collaboration Agreement.
In addition, because Janssen is solely responsible for the operational implementation of worldwide regulatory, development, manufacturing and commercialization activities related to imetelstat, we are solely dependent on Janssen to provide us with timely and accurate information concerning these
25
activities. If we do not receive accurate information from Janssen in a timely manner, or at all, regarding these activities, including, for example, plans for, and enrollment, efficacy and safety results from, clinical trials, then the timeliness and accuracy of our public disclosures, as well as our governance-related decision-making regarding these activities, may be adversely affected.
Our collaboration with Janssen may be unsuccessful due to other factors, including the following:
- •
- Janssen may choose to terminate the Collaboration Agreement for convenience;
- •
- Janssen may provide a negative Continuation Decision and halt its development of imetelstat;
- •
- the results of the Initial Phase 2 MF Study and/or the Initial Phase 2 MDS Study may be negative or inconclusive, or
Janssen may observe safety issues in either of these studies, which may result in a negative Continuation Decision by Janssen, in which case we would receive no further payments from Janssen under the
Collaboration Agreement;
- •
- Janssen may choose not to develop and commercialize imetelstat in certain markets or for one or more indications, if at all;
- •
- Janssen may take considerably more time advancing imetelstat through the clinical and regulatory process than we currently anticipate,
which could materially delay the achievement of milestones and, consequently the receipt of milestone payments from Janssen, and ultimately, any royalties on worldwide net sales;
- •
- in the event of a dispute between us and Janssen regarding Janssen's performance under the Collaboration Agreement, it may be
difficult for us to prove that Janssen breached its obligation to use "commercially reasonable efforts" with regard to the development, regulatory approval, manufacture and commercialization of
imetelstat under the Collaboration Agreement;
- •
- Janssen may not dedicate the resources necessary to carry imetelstat through clinical development or may not obtain the necessary
regulatory approvals, and this would delay the achievement of development, regulatory or sales milestones;
- •
- Janssen's ability to achieve development and manufacturing objectives or milestones may be delayed or substantially impacted if we
fail to transfer technology and information related to imetelstat to Janssen in a timely manner or at all;
- •
- subject to our election of the U.S. Co-Promotion Option, Janssen will be responsible for all aspects of the commercialization of
imetelstat worldwide, including pricing decisions which would affect the royalties on worldwide net sales we could receive;
- •
- Janssen may change the focus of its commercialization efforts or prioritize other programs more highly and, accordingly, reduce the
efforts and resources allocated to imetelstat, which would have the direct effect of reducing our royalties or share of potential co-promotion activities since the extent of our U.S. Co-Promotion
Option is limited to a percentage of overall promotion activities under the Collaboration Agreement;
- •
- after assuming manufacturing responsibilities for imetelstat, Janssen may fail to manufacture or supply sufficient quantities of
imetelstat for use in planned clinical trials, which could delay, suspend or stop any imetelstat clinical activities;
- •
- Janssen may fail to develop a commercially viable formulation or manufacturing process for imetelstat, and may fail to manufacture or supply sufficient quantities of imetelstat for commercial use, if approved, which would result in lost sales revenue and reduced royalties for us;
26
- •
- Janssen may not comply with all applicable regulatory requirements or may fail to report safety data in accordance with all applicable
regulatory requirements, which could delay, suspend or stop clinical activities being performed by Janssen or by us; and
- •
- if Janssen is acquired during the term of our collaboration, the acquirer may have different strategic priorities that could cause it to terminate the Collaboration Agreement or reduce its commitment to our collaboration.
If our collaboration with Janssen is unsuccessful as a result of any of the above factors, or any other factor, then Janssen may terminate the Collaboration Agreement, and we would not be eligible for any further payments from Janssen under the Collaboration Agreement, which would severely and adversely affect our business and business prospects.
Delays in the initiation of, or the inability to initiate, subsequent clinical trials of imetelstat, such as the Initial Phase 2 MF Study or the Initial Phase 2 MDS Study planned to be conducted under the Collaboration Agreement, could result in increased development costs and would delay our ability to earn revenues from milestone payments or royalties from the Collaboration Agreement with Janssen.
To date, we have not initiated any clinical trials evaluating imetelstat in any hematologic myeloid malignancies (other than essential thrombocythemia), including myelofibrosis, or MF. Advancing clinical development of imetelstat will be influenced by results from existing clinical trials, such as the MF Pilot Study, and potential future clinical trials of imetelstat in hematologic myeloid malignancies, such as the Initial Phase 2 MF Study or the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement. The commencement of potential future clinical trials of imetelstat could be delayed or abandoned for a variety of reasons, including as a result of failures or delays by Janssen in:
- •
- obtaining regulatory clearance to commence subsequent clinical trials of imetelstat in a timely manner, or at all, in the United
States or other countries;
- •
- properly designing, commencing, enrolling, conducting or completing potential future clinical trials, including the Initial
Phase 2 MF Study or the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement, and promptly or adequately reporting data from such trials;
- •
- demonstrating sufficient safety and efficacy in future Phase 2 clinical trials, including the Initial Phase 2 MF Study
or the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement, to obtain regulatory clearance to commence subsequent clinical trials in a timely manner,
or at all, in the United States or other countries;
- •
- properly conducting and/or completing the MF Pilot Study;
- •
- manufacturing sufficient quantities of imetelstat and in a manner that meets the quality standards of the FDA and other regulatory
agencies;
- •
- ensuring the ability to manufacture imetelstat at acceptable costs for Phase 3 clinical trials and commercialization;
- •
- obtaining clearance or approval of proposed trial designs or manufacturing specifications from the FDA and other regulatory
authorities;
- •
- reaching agreement on acceptable terms and on a timely basis, if at all, with collaborators and vendors located in the United States
or foreign jurisdictions, including contract research organizations, laboratory service providers and trial sites, on all aspects of clinical trials;
- •
- obtaining institutional review board or ethics committee approval to conduct clinical trials at prospective clinical trial sites; and
27
- •
- identifying and successfully screening and enrolling appropriate subjects for participation in clinical trials and retaining those subjects in the clinical trials.
Failures or delays with respect to any of these events could adversely affect the ability to initiate, maintain or successfully complete any future clinical trials of imetelstat, which could increase development costs, impair our ability to earn revenues from milestone payments or royalties from the Collaboration Agreement with Janssen or cause Janssen to terminate the Collaboration Agreement, any of which could adversely impact our financial results and would have severe adverse effects on our business and business prospects.
If there are any safety or efficacy results that cause the benefit-risk profile of imetelstat to become unacceptable, the clinical development of imetelstat would be delayed or halted, and Janssen may terminate the Collaboration Agreement, which would severely and adversely affect our business prospects, and may cause us to cease operations.
Imetelstat may prove to have undesirable or unintended side effects or other characteristics adversely affecting its safety, efficacy, cost-effectiveness or marketability that could prevent or limit its approval for marketing and successful commercial use, or that could delay or prevent the commencement and/or completion of clinical trials for imetelstat. For example, although the FDA removed the full clinical hold on our IND for imetelstat, if patients in current or future clinical trials experience similar or more severe hepatotoxicity, including elevated LFTs or severe hepatic adverse events, such IND for imetelstat may again be placed on clinical hold, and we, in collaboration with Janssen, may be precluded from further developing imetelstat. In addition, if regulatory submissions requesting approval to market imetelstat are submitted, after reviewing the data in such submissions, the FDA and regulatory agencies in other countries may conclude that the overall benefit-risk profile of imetelstat treatment, including hepatotoxicity or severe hepatic adverse events, may preclude approval of imetelstat for marketing or further development for any indications, including hematologic malignancies. Any of these events would severely harm our business and prospects, and would likely cause us to cease operations.
Further, in our Phase 1 clinical trials of imetelstat, we observed dose-limiting toxicities, including thrombocytopenia when the drug was used as a single agent, and neutropenia when the drug was used in combination with paclitaxel, as well as a low incidence of severe infusion reactions. In our Phase 2 clinical trials of imetelstat in ET, MM, and solid tumors, we have observed hematologic toxicities as well as gastrointestinal events, infections, muscular and joint pain, fatigue and infusion reactions. In addition, in our Phase 2 clinical trials of imetelstat, we have observed LFT abnormalities, the clinical significance and long-term consequences of which are currently undetermined. In our Phase 2 trial in ET, one patient died of bleeding esophageal varices, a complication of chronic liver disease, for which imetelstat could not be excluded as a causative agent. In the MF Pilot Study, myelosuppression has been the primary dose-limiting toxicity reported to date, consistent with our observations in previous Geron-sponsored imetelstat studies. However, during the MF Pilot Study, more persistent and profound myelosuppression, particularly thrombocytopenia, was observed with imetelstat administered on a weekly basis. This included one case of febrile neutropenia after prolonged myelosuppression with intracranial hemorrhage resulting in patient death, which was assessed as possibly related to imetelstat by the investigator. Since the MF Pilot Study is an ongoing study with additional data being generated, the benefit-risk profile of imetelstat in MF will continue to be assessed, including the risk of hepatotoxicity and severe cytopenias that may be associated with life-threatening clinical outcomes.
Clinical trials by their nature examine the effect of a potential therapy in a sample of the potential future patient population. As such, clinical trials conducted with imetelstat, to date and in the future, may not uncover all possible adverse events that patients treated with imetelstat may experience. In collaboration with Janssen, we may observe or report dose-limiting toxicities or other safety issues in potential future clinical trials of imetelstat, including the Initial Phase 2 MF Study and the Initial
28
Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement. Likewise, because previously enrolled patients continue to receive imetelstat in the MF Pilot Study, additional or more severe toxicities or safety issues in the MF Pilot Study, including additional serious adverse events and clinically significant LFT abnormalities, may be observed or reported as patient treatment continues and more data become available. If such toxicities or other safety issues in any clinical trial of imetelstat result in an unacceptable benefit-risk profile, then:
- •
- the commencement and/or completion of any current or future clinical trials, including the MF Pilot Study and the Initial
Phase 2 MF Study and the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement, would likely be delayed or prevented;
- •
- the MF Pilot Study or any potential future clinical trials, including the Initial Phase 2 MF Study and the Initial
Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement, may be placed on clinical hold or halted by regulatory authorities, such as the previous clinical holds
placed by the FDA on our IND for imetelstat and the IND for the MF Pilot Study; or
- •
- additional, unforeseen trials or preclinical studies may be required to be conducted.
The occurrence of any of these events would likely cause Janssen to abandon their development of imetelstat entirely and terminate the Collaboration Agreement. Any termination of the Collaboration Agreement by Janssen would have a material adverse effect on our results of operations, financial condition, business prospects and the future of imetelstat, any of which may cause us to cease operations.
If Janssen does not elect to continue the development of imetelstat in a timely manner, or at all, our business and business prospects would be severely harmed.
Under the terms of the Collaboration Agreement, Janssen is not obligated to make any additional payments to us until it makes an affirmative Continuation Decision following the results of the Initial Phase 2 MF Study, or, if the Initial Phase 2 MF Study is terminated early or suspended for an extended period of time, within a certain time period thereafter as set forth in the Collaboration Agreement. The timing of Janssen's Continuation Decision also affects the timing and availability of our decision regarding U.S. Opt-In Rights, as well as our election of the U.S. Co-Promotion Option. If the Initial Phase 2 MF Study is terminated early, suspended for an extended period of time, or is otherwise unsuccessful, Janssen may provide a negative Continuation Decision, in which case, the Collaboration Agreement would terminate, we would not be eligible for any further payments from Janssen under that agreement and our business and business prospects would be severely and adversely affected.
In addition, Janssen may terminate the Collaboration Agreement at any time for convenience. If Janssen terminates the Collaboration Agreement, then, depending on the timing of such event:
- •
- we would no longer have the right to receive any milestone payments or royalties under the Collaboration Agreement;
- •
- the development of imetelstat would likely be terminated or significantly delayed;
- •
- we would bear all of the risks and costs related to the further clinical development, manufacturing, regulatory approval and
commercialization of imetelstat;
- •
- we would need to raise additional capital if we were to choose to pursue imetelstat development on our own, or we would need to establish alternative collaborations with third-party collaboration partners, which may not be possible in a timely manner or at all, or may not be possible on terms acceptable to us, in which case it would likely be necessary for us to limit the
29
- •
- we would need to hire additional employees to support the development and commercialization of imetelstat, which would increase our need for additional funding.
size or scope of the imetelstat development program or seek additional funding by other means to accommodate the increased expenditures; and
Any termination of the Collaboration Agreement by Janssen at any time would have a material adverse effect on our results of operations, financial condition, business prospects and the future of imetelstat, any of which would have severe adverse effects on our business and business prospects, and may cause us to cease operations.
Our decision to exercise our U.S. Opt-In Rights under the Collaboration Agreement with Janssen for imetelstat must be made within a limited time after Janssen makes an affirmative Continuation Decision and, as a result, we may be required to make a substantial capital investment based on limited clinical data.
We must elect to exercise our U.S. Opt-In Rights within a short timeframe following Janssen's Continuation Decision. Although we expect to receive information from Janssen regarding data from the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study, proposed future clinical development plans and costs, estimates in timing for commercializing imetelstat and related promotional activities, and calculation of our share of development costs incurred to date by Janssen that we will be required to reimburse if we exercise our U.S. Opt-In Rights, we will be required to rapidly decide whether to make a substantial capital investment in imetelstat prior to the conclusion of any Phase 3 registration-enabling clinical trial. Accordingly, if imetelstat were to become unsuccessful in any Phase 3 registration-enabling clinical trial or fails to receive regulatory approval, we would not receive any financial return on this substantial capital investment. Such an occurrence would negatively impact our financial condition and results of operations.
Our Collaboration Agreement with Janssen prohibits us from developing or commercializing any product that operates through the same mechanism of action as imetelstat, and our U.S. co-promotion rights may be terminated if we market or promote any such products for any oncology indication. As a result of this, or for any other reason, we may not be able to successfully acquire or in-license promising product opportunities for development, which would limit our growth and revenue potential.
We plan to seek to diversify our sole product candidate development risk by identifying promising product opportunities for development, which we may seek to acquire or in-license. However, the Collaboration Agreement with Janssen prohibits us from commercializing, under the intellectual property we have licensed exclusively to Janssen, any substance whose identified or known mechanism of action is telomerase inhibition. Further, if we exercise our U.S. Co-Promotion Option under the Collaboration Agreement, we will be required to certify at the time of exercising our U.S. Co-Promotion Option that we are not marketing or promoting, and have no right to market or promote, any such products for any oncology indication. Our right to co-promote in the U.S. may be terminated by Janssen if we develop or commercialize a product for treating an oncology indication that acts through the same mechanism of action as imetelstat or that is substitutable for imetelstat. Accordingly, our Collaboration Agreement with Janssen could adversely affect our ability to acquire or in-license, or to research, develop or market, promising product candidates.
In addition, we may not be able to identify promising product candidates. The competition to acquire or in-license rights to promising product candidates is fierce, and many of our competitors are large, multinational pharmaceutical and biotechnology companies with considerably more financial, development and commercialization resources and experience than we have. Thus, even if we succeed in identifying promising product candidates, we may not be able to acquire rights to them on acceptable terms, or at all. In any event, any growth through acquisition or in-licensing will depend upon our identifying and obtaining promising product candidates, our ability to develop those product
30
candidates and the availability of funding to complete the development of, obtain regulatory approval for and commercialize these product candidates. If we are unable to identify and acquire promising product candidates, we will be unable to diversify our sole product candidate development risk, and our growth and revenue potential could be limited.
We may not be able to successfully retain key personnel to support our collaboration with Janssen or to manage any future growth.
Under the terms of the Collaboration Agreement, we and Janssen have created a joint governance structure, including committees and working groups, to manage worldwide regulatory, development, manufacturing and commercialization activities for imetelstat, and we will have ongoing responsibilities to oversee and participate in the collaboration with Janssen. In addition, we will remain responsible for prosecuting, at Janssen's direction, the patents we licensed to Janssen, and have sole responsibility for those patents that were not licensed to Janssen. If we are unable to successfully retain, motivate and incentivize our personnel, our ability to support the Collaboration Agreement with Janssen could be impaired, and our business and the price of our common stock would be adversely impacted.
In addition, our future growth and success depend to a significant extent on the skills, experience and efforts of our executive officers and key members of our clinical and scientific staff. We face intense competition for qualified individuals from numerous pharmaceutical, biopharmaceutical and biotechnology companies, as well as academic and other research institutions. The previous restructurings we implemented and the recently announced organizational resizing, as well as our collaboration with Janssen and uncertainties regarding our ability to diversify our sole product candidate development risk, could have an adverse impact on our ability to retain and recruit qualified personnel or we may incur unanticipated inefficiencies caused by our reduced personnel resources. We may be unable to retain our current personnel or attract or assimilate other highly qualified management and development personnel in the future on acceptable terms. The loss of any or all of these individuals could harm our business and could impair our ability to support our collaboration with Janssen or to support future growth.
We and certain of our officers have been named as defendants in three purported securities lawsuits, two of which are securities class action lawsuits, and certain of our officers and directors have been named as defendants in a derivative lawsuit. These, and potential similar or related lawsuits, could result in substantial damages, divert management's time and attention from our business, and have a material adverse effect on our results of operations. These lawsuits and any other lawsuits will be costly to defend or pursue and are uncertain in their outcome.
Securities-related class action lawsuits and derivative litigation has often been brought against companies, including many biotechnology companies, which experience volatility in the market price of their securities. This risk is especially relevant for us because biotechnology and biopharmaceutical companies often experience significant stock price volatility in connection with their product development programs.
On March 14, 2014, a purported securities class action lawsuit was commenced in the United States District Court for the Northern District of California, or the California District Court, naming as defendants us and certain of our officers. The lawsuit alleges violations of the Securities Exchange Act of 1934 in connection with allegedly false and misleading statements made by us related to our Phase 2 trial of imetelstat in patients with ET or PV. The plaintiff alleges, among other things, that we failed to disclose facts related to the occurrence of persistent low-grade LFT abnormalities observed in our Phase 2 trial of imetelstat in ET or PV patients and the potential risk of chronic liver injury following long-term exposure to imetelstat. The plaintiff seeks damages and an award of reasonable costs and expenses, including attorneys' fees.
31
On March 28, 2014, a second purported securities class action lawsuit was commenced in the California District Court, naming as defendants us and certain of our officers. This lawsuit, which is based on the same factual background as the purported securities class action lawsuit that commenced on March 14, 2014, also alleges violations of the Securities Exchange Act of 1934 and seeks damages and an award of reasonable costs and expenses, including attorneys' fees.
On June 30, 2014, both of the foregoing lawsuits, or the Class Action Lawsuits, were consolidated for all purposes, and a lead plaintiff and lead counsel were appointed by the California District Court. On July 21, 2014, the California District Court ordered the lead plaintiff to file its consolidated amended complaint in the Class Action Lawsuits, which was filed on September 19, 2014. We filed our motion to dismiss the consolidated amended complaint on November 18, 2014. The plaintiff's opposition to our motion to dismiss was filed on January 20, 2015, and we filed our reply on February 25, 2015. The court hearing for the motion to dismiss has been scheduled for April 10, 2015.
On June 6, 2014, a purported securities lawsuit, not styled as a class action, was commenced in the United States District Court for the Southern District of Mississippi, or the Mississippi District Court, naming as defendants us and certain of our officers. This lawsuit, which is based on the same factual background as the Class Action Lawsuits, also alleges violations of the Securities Exchange Act of 1934 and seeks damages and an award of reasonable costs and expenses, including attorneys' fees. On August 11, 2014, we filed a motion to transfer the purported securities lawsuit filed in the Mississippi District Court to the California District Court so it could be consolidated with the purported Class Action Lawsuits. On November 4, 2014, the Mississippi District Court granted our motion and transferred the case to the California District Court, and the transferred case has been consolidated by the California District Court with the purported Class Action Lawsuits filed in the California District Court.
On April 21, 2014, a stockholder purporting to act on our behalf filed a derivative lawsuit in the Superior Court of California for the County of San Mateo against certain of our officers and directors. The lawsuit alleges breaches of fiduciary duties by the defendants and other violations of law. In general, the lawsuit alleges that the defendants caused or allowed the dissemination of allegedly false and misleading statements related to our Phase 2 trial of imetelstat in patients with ET or PV. The plaintiff is seeking unspecified monetary damages and other relief, including reforms and improvements to our corporate governance and internal procedures.
It is possible that additional suits will be filed, or allegations received from stockholders, with respect to these same or other matters and also naming us and/or our officers and directors as defendants. These lawsuits and any other related lawsuits are subject to inherent uncertainties, and the actual defense and disposition costs will depend upon many unknown factors. The outcome of these lawsuits is necessarily uncertain. We could be forced to expend significant resources in the defense against these lawsuits and we may not prevail. In addition, we may incur substantial legal fees and costs in connection with these lawsuits. We currently are not able to estimate the possible cost to us from these matters, as these lawsuits are currently at an early stage, and we cannot be certain how long it may take to resolve these matters or the possible amount of any damages that we may be required to pay. We have not established any reserve for any potential liability relating to these lawsuits. It is possible that we could, in the future, incur judgments or enter into settlements of claims for monetary damages. A decision adverse to our interests on these actions could result in the payment of substantial damages, or possibly fines, and could have a material adverse effect on our cash flow, results of operations and financial position.
32
We may also be subject to litigation arising from our proposed or completed strategic transactions or if the results of our business and collaboration activities are not successful.
On October 1, 2013, we closed the transaction to divest our human embryonic stem cell assets and our autologous cellular immunotherapy program pursuant to the terms of the Contribution Agreement that we entered into in January 2013 with BioTime and Asterias. On November 13, 2014, we announced that we had entered into the Collaboration Agreement with Janssen to develop and commercialize imetelstat worldwide. We may face litigation arising from or related to the value received by our stockholders, if any, from our distribution of the Asterias Series A common stock and/or the BioTime Warrants distributed by Asterias under the Contribution Agreement, or our role as a named underwriter with respect to our distribution of the Asterias Series A common stock, including the delays we experienced with respect to completing our distribution of the Asterias Series A common stock, or we may face litigation based on other matters related to the Contribution Agreement and the Collaboration Agreement or the transactions contemplated thereby, including if we are unable to generate substantial value under the Collaboration Agreement with Janssen or such collaboration is not otherwise successful.
As a result of these and other factors, we may be exposed to a number of litigation risks related to the transactions contemplated by the Contribution Agreement and the Collaboration Agreement, including declines or fluctuations in our stock price, additional advisor and legal fees, distractions to our management caused by activities undertaken in connection with resolving any disputes related to the transactions, or the loss of important contractual rights. As another example, some of our investors purchased shares of our common stock because they were interested in the opportunities presented by our human embryonic stem cell programs. Thus, certain stockholders may have attributed substantial financial value to our stem cell assets and may believe that the Asterias Series A common stock, BioTime Warrants and/or cash received in the distributions pursuant to the Contribution Agreement were inadequate consideration for such assets.
Similarly, the announcement and/or completion of these strategic transactions could result in litigation arising out of any claims that our stockholders suffered financial losses due to the transactions, the approval of our stockholders was required under applicable law or otherwise should have been obtained prior to the completion of either or both of these transactions, or that our officers and directors breached their fiduciary duties in connection with the approval and completion of these transactions. Although we believe that stockholder approval was not required under applicable law in order to complete either or both of these transactions and therefore we neither sought nor intend to seek such stockholder approval, it is possible that persons who were stockholders at the time of the applicable transaction may claim that their approval was required, in which case litigation could follow, which could result in substantial damages to us and/or could negatively affect our rights and obligations under either of these agreements or, in the case of the Collaboration Agreement, could result in the termination of that agreement.
Likewise, our stockholders may believe that the financial and other terms of the Collaboration Agreement are not favorable to either us or our stockholders, including any belief that the potential payments we may receive under the Collaboration Agreement are inadequate. Litigation brought by our stockholders challenging the validity of, or financial losses resulting from, these transactions could also result in claims against us by Asterias and/or Janssen, and each of the Contribution Agreement and the Collaboration Agreement provide for indemnification by us of BioTime and Janssen, respectively, against all losses and expenses relating to breaches of our representations, warranties and covenants in the applicable agreement, which could expose us to further financial obligations and damages. The occurrence of any one or more of the above could have a significant adverse impact on our business and financial condition.
33
In addition, if the results of our business and collaboration activities are not successful, including without limitation, if:
- •
- we or Janssen are otherwise unable to continue development of imetelstat due to actions by regulatory authorities, such as the
previous full clinical hold that was placed by the FDA on our IND for imetelstat in March 2014;
- •
- we, Janssen or any investigators ascertain that the use of imetelstat results in significant systemic or organ toxicities, including
hepatotoxicity, or other safety issues resulting in an unacceptable benefit-risk profile;
- •
- the conduct of previous clinical trials, such as the MF Pilot Study, and future clinical trials, such as the Initial Phase 2 MF
Study and the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement, results in patient injury or death, or any failure to meet regulatory and
compliance requirements;
- •
- the final or any preliminary results from the MF Pilot Study, or any subsequent clinical trial of imetelstat, including the Initial
Phase 2 MF Study and the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement, are not deemed to be successful;
- •
- Janssen discontinues the further development of imetelstat and terminates the Collaboration Agreement; or
- •
- Asterias is unable to develop our stem cell assets, and we are not able to receive any royalties from the sale of any potential stem cell products by Asterias,
our stock price would likely decline, and future litigation may result. A decision adverse to our interests in any such lawsuits could result in the payment of substantial damages by us, and could have a material adverse effect on our cash flow, results of operations and financial position or could otherwise severely harm our business.
Our business may also bring us into conflict with our licensees, licensors, or others with whom we have contractual or other business relationships, or with our competitors or others whose interests differ from ours. If we are unable to resolve those conflicts on terms that are satisfactory to all parties, we may become involved in litigation brought by or against us. For example, we are subject to the risk of possible disagreements with Janssen, including those regarding the development and/or commercialization of imetelstat, interpretation of the Collaboration Agreement and ownership of proprietary rights. In addition, in certain circumstances we may believe that we have achieved a particular milestone and Janssen may disagree with our belief. In that case, receipt of that milestone payment may be delayed or may never be received, which would adversely affect our financial condition and may require us to adjust our operating plans. While the Collaboration Agreement provides for a joint governance structure to oversee and manage worldwide regulatory, development, manufacturing and commercialization activities for imetelstat, Janssen generally will, subject to limited exceptions, have the deciding vote in the event of any disagreement. In any event, the joint governance structure contemplated by the Collaboration Agreement will be dissolved in the event that we do not exercise our U.S. Opt-In Rights, which would preclude our ability to participate in any further decision-making for imetelstat. Reliance on a joint governance structure also subjects us to the risk that changes in key management personnel that are members of the various joint committees may materially and adversely affect the functioning of these committees, which could significantly delay or preclude imetelstat development and/or commercialization. As a result of possible disagreements with Janssen, we also may become involved in litigation or arbitration, which would be time-consuming and expensive.
Monitoring, initiating and defending against legal actions, including our currently-pending securities-related lawsuits and derivative litigation, are time-consuming for our management, likely to be expensive and may detract from our ability to fully focus our internal resources on our business
34
activities. The outcome of litigation is always uncertain, and in some cases could include judgments against us that require us to pay damages, enjoin us from certain activities, or otherwise negatively affect our legal or contractual rights, which could have a significant adverse effect on our business. In addition, the inherent uncertainty of such litigation, including our currently-pending securities-related lawsuits and derivative litigation, could lead to increased volatility in our stock price and a decrease in the value of our stockholders' investment in our common stock.
RISKS RELATED TO CLINICAL AND COMMERCIALIZATION ACTIVITIES
The research and development of imetelstat is subject to numerous risks and uncertainties.
The science and technology of telomere biology, telomerase and our proprietary oligonucleotide chemistry are relatively new. There is no precedent for the successful commercialization of a therapeutic product candidate based on these technologies. In collaboration with Janssen, we must undertake significant research and development activities to develop imetelstat, our sole product candidate, based on these technologies, which may take years to accomplish, if at all.
Because of the significant scientific, regulatory and commercial challenges that must be overcome for the research, development and commercialization of imetelstat to be successful, the development of imetelstat in hematologic myeloid malignancies, including MF and MDS, or any other indications, may be delayed or abandoned, even after significant resources have been expended on it. Our decisions to discontinue our Phase 2 clinical trial of imetelstat in metastatic breast cancer in September 2012, and to discontinue our development of imetelstat in solid tumors with short telomeres in April 2013, are examples of this. Any further delay or abandonment of the development of imetelstat in hematologic myeloid malignancies would have a material adverse effect on our collaboration with Janssen which could result in the termination of the Collaboration Agreement. Any of these events would have severe adverse effects on our business and business prospects and likely result in the failure of our business.
Success in early clinical trials may not be indicative of results in subsequent clinical trials. Likewise, preliminary data reported by investigators from time-to-time are subject to review or verification procedures that could result in material differences to final data and may change as more patient data become available.
A number of new drugs and biologics have shown promising results in initial clinical trials, but subsequently failed to establish sufficient safety and efficacy data to obtain necessary regulatory approvals. Data obtained from preclinical and clinical activities are subject to varying interpretations, which may delay, limit or prevent regulatory approval.
Data from our preclinical studies and Phase 1 and Phase 2 clinical trials of imetelstat, as well as preliminary, additional or updated data from the MF Pilot Study, should not be relied upon as evidence that subsequent or larger-scale clinical trials of imetelstat will succeed. The positive efficacy results we have obtained from the Phase 2 clinical trial of imetelstat in ET may not predict the future therapeutic benefit of imetelstat, if any, in other hematologic myeloid malignancies, including MF. For example, the known LFT abnormalities and dose-limiting toxicities associated with imetelstat, such as profound thrombocytopenia and neutropenia and other safety issues, including death, that have been observed in both Geron-sponsored and investigator-sponsored trials, including the MF Pilot Study, could cause complexities in treating patients with MF and could result in the discontinuation of the MF Pilot Study and any future clinical trials of imetelstat, including the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement. Also, the criteria used to assess efficacy in the MF Pilot Study have not been validated for clinical use and may not be considered by the FDA or other regulatory agencies to be accurate predictors of efficacy for different endpoints that may be required by the FDA or other regulatory agencies for Phase 3 clinical trials.
35
The preliminary results of the MF Pilot Study presented by the investigator at the ASH annual meeting in December 2013 and the updated preliminary results presented at ASH in December 2014 will need to be confirmed in one or more larger Phase 2 and Phase 3 trials in MF at multiple treating centers. The results reported by us, Janssen or by the investigator in the MF Pilot Study may not be reproduced in any subsequent imetelstat trials conducted in the future, including the Initial Phase 2 MF Study or the Initial Phase 2 MDS Study planned to be conducted under the Collaboration Agreement, or by any other investigator or group of investigators, or in any trial enrolling a larger number of patients or conducted at multiple treating centers, and thus should not be relied upon as indicative of future clinical results of imetelstat in MF or any other hematologic myeloid malignancy.
In addition, from time-to-time, we or Janssen may report or announce preliminary data from current or potential future clinical trials, such as the Initial Phase 2 MF Study and Initial Phase 2 MDS Study planned to be conducted under the Collaboration Agreement. For example, the investigator for the MF Pilot Study reported preliminary data from the trial in December 2013 and updated preliminary data in December 2014. Since those data were preliminary, the final data from the MF Pilot Study may be materially different than the data reported in December 2013 or December 2014. Since patients previously enrolled in the MF Pilot Study continue to receive imetelstat, safety and efficacy data continue to be generated, and such additional and updated data may materially change the overall conclusions from the preliminary data reported in December 2013 or December 2014. Therefore, such preliminary data should be considered carefully and with caution. Additional and updated data from the MF Pilot Study are also subject to any review or verification procedures that Janssen may conduct as the trial sponsor for the MF Pilot Study after it assumes responsibility for the conduct of the MF Pilot Study, and since this could result in material differences from the data reported by the investigator or us, additional or updated data that may be reported from the MF Pilot Study should be considered carefully and with caution. Analyses performed by Janssen may result in conclusions that are materially different from the investigator's analyses or ours, and therefore such preliminary data should be considered carefully and with caution.
Material adverse changes in final data from the MF Pilot Study could jeopardize our Collaboration Agreement with Janssen and if Janssen were to terminate the Collaboration Agreement, our business prospects would be severely and adversely affected. Even if final safety and efficacy data from the MF Pilot Study are positive, significant additional clinical testing will be necessary for the future development of imetelstat in MF. Any such final safety and efficacy data from the MF Pilot Study may not be reproducible in future clinical trials, including the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement.
Before Janssen can seek to obtain regulatory approval for the commercial sale of imetelstat, multiple clinical trials, including larger-scale Phase 3 clinical trials, will need to be conducted to demonstrate that imetelstat is safe and effective for use in a diverse population. There is typically an extremely high rate of attrition from the failure of drug candidates proceeding through clinical trials. If imetelstat cannot be developed in future clinical trials, including Phase 3 clinical trials, our Collaboration Agreement with Janssen will be negatively impacted and could be terminated altogether, which would have severe adverse effects on our business and business prospects, and likely result in the failure of our business.
36
Conducting and completing potential future clinical trials of imetelstat, including the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement, on a timely basis is subject to risks and uncertainties.
Delays or terminations of potential future clinical trials, including the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement, could be caused by matters such as:
- •
- not receiving timely regulatory clearances or approvals in any jurisdiction, whether within or outside of the United States,
including, if we, Janssen or future investigators do not obtain and maintain regulatory clearance to commence studies of imetelstat in MF, MDS or any additional hematologic myeloid malignancies in a
timely manner or at all, including the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement;
- •
- the inability by Janssen to maintain the INDs for imetelstat that we expect to transfer to Janssen, including the IND for the MF Pilot
Study, without such INDs being placed on full or partial clinical hold by the FDA;
- •
- the inability to properly design, conduct and/or complete current and potential future clinical trials of imetelstat including the MF
Pilot Study and the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study;
- •
- data showing lack of effectiveness of imetelstat during clinical trials, or results that do not demonstrate statistically significant
efficacy;
- •
- safety issues, side effects or dose-limiting toxicities, including any additional or more severe safety issues related to imetelstat
in addition to those which have been observed to date in Geron-sponsored or investigator-sponsored trials, whether or not in the same indications or therapeutic areas;
- •
- disruptions due to drug supply or quality issues;
- •
- not receiving acceptance of new manufacturing specifications or procedures or clinical trial protocol amendments by regulatory
authorities;
- •
- failure by investigators conducting future clinical trials of imetelstat, including the Initial Phase 2 MF Study and the
Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement, to timely commence, enroll, complete or report data from such clinical trials;
- •
- not receiving timely institutional review board or ethics committee approval of clinical trial protocols or protocol amendments;
- •
- delays in patient enrollment due to size or nature of patient population, nature of protocols, proximity of patients to clinical
sites, availability of effective treatments for the relevant disease and eligibility criteria for the trial;
- •
- inability to retain patients to complete clinical trials or to return for post-treatment follow-up;
- •
- difficulty in obtaining or accessing necessary clinical data, including additional and future data from the MF Pilot Study, which may
result in incomplete data sets;
- •
- unavailability of any study-related treatment (including comparator therapy);
- •
- issues or disputes with key vendors of clinical services, such as contract research organizations, clinical trial sites and laboratory service providers; or
37
- •
- governmental or regulatory delays in any jurisdiction, whether within or outside of the United States, information requests, clinical holds, such as the previous clinical holds placed by the FDA on our IND for imetelstat and the IND for the MF Pilot Study, and changes in regulatory requirements, policies and guidelines.
Advancing clinical development of imetelstat in the United States is dependent on obtaining positive results from existing and potential future clinical trials of imetelstat in hematologic myeloid malignancies, including the MF Pilot Study and the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement. Obtaining additional and future data from the MF Pilot Study may provide additional insights into the further development of imetelstat for MF, MDS or AML, including with respect to Janssen's ability to initiate the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement. Accordingly, a delay in the timely completion of the MF Pilot Study, including any delay caused by any future clinical hold placed by the FDA on the INDs for imetelstat, including the IND for the MF Pilot Study, that we plan to transfer to Janssen prior to initiation of the Initial Phase 2 MF Study, could have a material adverse effect on advancing the development of imetelstat to subsequent clinical trials, including the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement. Also, adverse safety results from clinical trials of imetelstat, including those results that have been reported and those that may in the future be reported from the MF Pilot Study or the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement, could delay or prevent the initiation or continuation of further clinical development of imetelstat, whether under the Collaboration Agreement or otherwise. Occurrence of any of these events would delay the timing of any Continuation Decision Janssen could provide to us or could cause Janssen to terminate the Collaboration Agreement which would severely and adversely affect the future of imetelstat and our business prospects.
In addition, enrollment goals for potential future clinical trials of imetelstat, including the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement, may not be met. The inability to retain or treat patients who have enrolled in a clinical trial but may be prone to withdraw due to side effects from imetelstat, lack of efficacy or personal issues, or who are lost to further follow-up, could result in clinical trial delays, the inability to complete clinical trials, or incomplete data sets. Further, any future clinical trials may be overseen by a safety monitoring committee, which may determine to significantly modify, delay or suspend one or more of these trials due to safety or futility findings based on emerging data occurring during a clinical trial. Data that we have received or that we or Janssen may in the future receive from investigators may be flawed or incomplete if the investigators fail to follow appropriate clinical or quality practices. Delays in timely initiation or completion of clinical testing of imetelstat could increase research and development costs and could prevent or would delay obtaining regulatory approval for imetelstat, either of which would delay the timing of any Continuation Decision Janssen could provide to us or could cause Janssen to terminate the Collaboration Agreement which would severely and adversely affect the future of imetelstat and our business prospects.
Obtaining regulatory clearances and approvals to develop and market imetelstat in the United States and other countries is a costly and lengthy process, and we cannot predict whether or when regulatory authorities will permit additional imetelstat development or approve imetelstat for commercial sale.
Federal, state and local governments in the United States and governments in other countries have significant regulations in place that govern drug research and development and may prevent us, in collaboration with Janssen, from successfully conducting development efforts or from commercializing imetelstat. Imetelstat must receive all relevant regulatory approvals before it may be marketed in the United States or other countries. Obtaining regulatory approval is a lengthy, expensive and uncertain
38
process. Because imetelstat involves the application of new technologies and a new therapeutic approach, it may be subject to substantial additional review by various government regulatory authorities, and, as a result, the process of obtaining regulatory approvals for imetelstat may proceed more slowly than for product candidates based upon more conventional technologies, and any approval that may be received could limit the use of imetelstat.
Prior to initiating future clinical trials of imetelstat, including the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement, the clinical trial protocols must be submitted to the FDA or regulatory authorities in other countries. Questions or comments from these agencies that must be addressed would likely delay further clinical development of imetelstat and the timing of any Continuation Decision by Janssen or could cause Janssen to terminate the Collaboration Agreement, which would severely and adversely affect the future of imetelstat and our business prospects.
Prior to submission of any regulatory application seeking approval to commence commercial sales of imetelstat, extensive preclinical and clinical testing will be required to be conducted. If the interpretation of safety and efficacy data obtained from these preclinical and clinical studies varies from interpretations by the FDA or regulatory authorities in other countries, this would likely delay, limit or prevent further development and approval of imetelstat which may cause Janssen to terminate the Collaboration Agreement. For example, the FDA and regulatory authorities in other countries may require more or different data than what has been generated from our preclinical studies and our prior Geron-sponsored Phase 2 clinical trials or the MF Pilot Study or that may be generated by potential future clinical trials, including the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement. In addition, delays or rejections of regulatory approvals, or limitations in marketing authorizations, may be encountered as a result of changes in regulatory environment or regulatory agency policy during the period of product development and/or the period of review of any application for regulatory agency approval for imetelstat. We do not expect imetelstat to be approved for commercial sale for many years, if at all.
Delays in obtaining regulatory agency clearances and approvals or limitations in the scope of such clearances or approvals could:
- •
- significantly harm the commercial potential of imetelstat;
- •
- impose costly procedures upon future development activities;
- •
- diminish any competitive advantages that may have been available; or
- •
- adversely limit the amount of, or affect our ability to receive, any milestone payments or royalties under the Collaboration Agreement with Janssen.
Even if the necessary time and resources are committed by us and Janssen, the required regulatory agency clearances and approvals may not be obtained for imetelstat. Even if regulatory agency clearances and approvals are obtained to commence commercial sales of imetelstat, they may entail limitations on the indicated uses or other aspects of the product label for which imetelstat can be marketed, which could limit the potential commercial use of imetelstat, or an approval might be contingent on the performance of costly additional clinical trials that would be required after approval. The occurrence of any of these events could delay any applications for regulatory approval and therefore delay the payment of potential milestone payments to us, or, if approved for commercial sale, could reduce the market demand for imetelstat and therefore result in decreased sales and reduced royalties for us under the Collaboration Agreement. Occurrence of any of these events could negatively impact our collaboration with Janssen or cause Janssen to terminate the Collaboration Agreement, which would severely and adversely affect our business and business prospects.
39
Failure to achieve continued compliance with government regulation could delay or halt commercialization of imetelstat, our sole product candidate.
Approved products and their manufacturers are subject to continual review, and discovery of previously unknown problems with a product or its manufacturer may result in restrictions on the product or manufacturer, including import restrictions, seizure and withdrawal of the product from the market. If approved for commercial sale, future sales of imetelstat will be subject to government regulation related to numerous matters, including the processes of:
- •
- manufacturing;
- •
- advertising and promoting;
- •
- selling and marketing;
- •
- labeling; and
- •
- distribution.
If, and to the extent that, we are or Janssen is unable to comply with these regulations, our ability to earn potential milestone payments and royalties from worldwide net sales of imetelstat would be materially and adversely impacted.
Failure to comply with regulatory requirements can result in severe civil and criminal penalties, including but not limited to:
- •
- recall or seizure of products;
- •
- injunctions against the import, manufacture, distribution, sales and/or marketing of products; and
- •
- criminal prosecution.
The imposition of any of these penalties or other commercial limitations could negatively impact our collaboration with Janssen or cause Janssen to terminate the Collaboration Agreement, either of which would severely and adversely affect our business and business prospects.
Janssen's development activities conducted under a Janssen Independent Development Plan, or IDP, may create significant reimbursement obligations for us, which could result in reduced cash inflow from future milestone payments and royalties until we have fully paid our obligations.
Under the Collaboration Agreement, Janssen may conduct certain development activities for imetelstat under a Janssen IDP if we and Janssen agree that such activities should be performed outside of the mutually agreed global clinical development plan. Although Janssen would bear all of the costs for such Janssen IDP, if we exercised our U.S. Opt-In Rights and if any data from a Janssen IDP supports approval by a regulatory agency in the United States or other countries, then we would be required to reimburse Janssen for our share of the costs of that Janssen IDP plus a premium pursuant to the terms of the Collaboration Agreement. This cost reimbursement is payable as a lump sum up to a certain threshold upon receipt of regulatory approval for the Janssen IDP. Any remaining amounts in excess of the threshold are payable in installments by offsetting milestone payments or royalties received by us over a certain period of time, at which time any remaining reimbursement amount would be payable in a lump sum. This payment mechanism could result in reduced cash inflow from future milestone payments and royalties, which would adversely affect our results of operations and financial condition.
40
Under the Collaboration Agreement with Janssen, if we develop imetelstat independently under our own IDP, the success of that IDP may depend on providing adequate financial and technical resources. Failure to successfully conduct or fund our own IDP activities may adversely affect our business.
Under the Collaboration Agreement with Janssen, we may conduct certain development activities for imetelstat under a Geron IDP if we and Janssen agree that such activities should be performed outside of the mutually agreed global clinical development plan. In the event we conduct any clinical activities under a Geron IDP, we will be responsible for paying all of the development costs for the Geron IDP. Because the outcome of any clinical activities and/or regulatory approval process is highly uncertain, we cannot reasonably estimate whether any Geron IDP activities we may undertake will succeed. Since we are only eligible for reimbursement from Janssen for their share of the Geron IDP costs plus a premium if any data from a Geron IDP supports approval by a regulatory agency in the United States or other countries, we may not recoup our investment in any Geron IDP, which could adversely affect our financial condition. In addition, we may need additional capital to support any Geron IDP activities and we cannot assure you that our existing capital resources, future interest income, potential milestone payments and royalties under the Collaboration Agreement and potential future sales of our common stock will be sufficient to fund these future activities. If sufficient capital is not available, we may be unable to pursue activities under a Geron IDP, which could adversely affect our business.
To execute activities under a Geron IDP, we likely would be required to collaborate with contract research organizations, investigators, academic institutions, vendors, clinical trial sites, scientific consultants and others. We would be dependent upon the ability of these parties to perform their responsibilities reliably. In addition, we would have limited control over the activities of these organizations, investigators, scientific consultants and vendors. Except as otherwise required by our agreements with them, we could expect only limited amounts of their time to be dedicated to our activities. If any of these third parties were unable or refuse to contribute to projects on which we needed their help, our ability to conduct activities under a Geron IDP could be significantly harmed. Also, if the performance of these services is not of the highest quality, does not achieve necessary regulatory compliance standards, or if such organization or vendor stops or delays its performance for any reason, it would impair and delay our ability to report data from clinical activities under a Geron IDP which would, in turn, hinder our ability to make the necessary representations or provide the necessary information to regulatory authorities, if at all. As a result, we may not obtain regulatory approval and receive any reimbursement from Janssen for their share of the costs for the Geron IDP, which could adversely affect our business and financial condition.
We will be dependent on Janssen and third parties to manufacture clinical and commercial quantities of imetelstat, which could result in a delay of clinical trials or regulatory approval or lost sales.
Under the Collaboration Agreement, after a transition period, Janssen will be responsible for the manufacture and/or manage the supply of imetelstat on a global basis for clinical trials and all commercial activities. Consequently, we will be, and expect to remain, dependent on Janssen to appropriately supply imetelstat. Janssen may encounter difficulties in production scale-up, including problems involving production yields, quality control and quality assurance, and shortage of qualified personnel. Janssen may not perform as agreed or may default in its obligations to supply imetelstat for clinical trials and/or commercial activities. Janssen also may fail to deliver the required quantities of imetelstat on a timely basis. Any such failure by Janssen could delay future clinical trials and any applications for regulatory approval and therefore delay the payment of potential milestone payments to us, or, if approved for commercial sale, could impair Janssen's ability to meet the market demand for imetelstat and therefore result in decreased sales and reduced royalties for us.
Currently, third-party contractors perform certain process development or other technical and scientific work with respect to imetelstat, as well as supply starting materials and manufacture drug
41
substance and drug product. We do not have direct control over their personnel or operations. We rely on these third-party contractors to produce and deliver sufficient quantities of imetelstat to support clinical trials on a timely basis and to comply with applicable regulatory requirements. If requested by Janssen, we may transfer certain of the agreements with these third parties to Janssen, if permitted by the terms and conditions of the respective agreements or otherwise allowed by the third parties. If these companies do not perform the work which they are contracted to perform, fail to comply with applicable cGMP regulations, do not complete the work within the expected timelines, fail to produce materials which are suitable for use in clinical trials or choose to exit the business, the ability to develop or manufacture imetelstat could be significantly harmed. For example, changes to one or more suppliers due to these or other reasons could lead to delays in drug supply. Manufacturing delays could adversely impact the initiation or completion of future clinical trials, such as the Initial Phase 2 MF Study or the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement, which may cause Janssen to terminate the Collaboration Agreement or delay the timing of any Continuation Decision that Janssen could provide to us, either of which would severely and adversely affect our business prospects.
In addition, current third-party contractors and/or any other contractors utilized by Janssen may need to make substantial investments to enable sufficient capacity increases and cost reductions, and to implement those regulatory and compliance standards necessary for successful Phase 3 clinical trials and commercial production. These third-party contractors may not be able to achieve such capacity increases, cost reductions, or regulatory and compliance standards, and even if they do, such achievements may not be at commercially reasonable costs. Neither we nor Janssen have any long-term commitments or commercial supply agreements with any of the third-party contractors for imetelstat, and changing manufacturers may be prolonged and difficult due to inherent technical complexities and because the number of potential manufacturers is limited. It may be difficult or impossible for Janssen to find a replacement manufacturer on acceptable terms, or at all.
There are other risks and uncertainties with respect to manufacturing that could adversely impact the initiation or completion of future clinical trials. For example, certain commonly used reagents and solvents may experience market shortages and, if these shortages occur, such shortages may adversely impact the ability to manufacture imetelstat. If a significant issue arises regarding manufacturing, this may cause Janssen to terminate the Collaboration Agreement or delay the timing of any Continuation Decision that Janssen could provide to us, either of which would severely and adversely affect our business prospects.
Imetelstat may not be able to be manufactured at costs or scales necessary to conduct clinical trials or potential future commercialization activities.
Oligonucleotides are relatively large molecules produced using complex chemistry, and the cost of manufacturing an oligonucleotide like imetelstat is greater than the cost of making typical small-molecule drugs. Therefore, imetelstat for clinical use is more expensive to manufacture than most other treatments currently available today or that may be available in the future. Similarly, the cost of manufacturing imetelstat for commercial use will need to be significantly lower than our current costs in order for imetelstat to become a commercially successful product. However, Janssen may not be able to achieve sufficient scale increases or cost reductions necessary for successful commercial production of imetelstat, which could result in decreased sales and reduced royalties for us.
Manufacturing imetelstat is subject to process and technical challenges and regulatory risks.
We have faced and Janssen will continue to face numerous risks and uncertainties with regard to manufacturing imetelstat. Regulatory requirements for oligonucleotide products are less well-defined than for small-molecule drugs, and there is no guarantee that Janssen will achieve sufficient product quality standards required for Phase 3 clinical trials or for commercial approval and manufacturing of
42
imetelstat. Changes in the manufacturing processes or formulations for imetelstat that may be made during later stages of clinical development, including during Phase 3 clinical trials, may result in regulatory delays, the need for further clinical trials, rejection of a marketing application, or limitation on marketing authorization by regulatory authorities, and therefore delay the payment of potential milestone payments to us, or, if approved for commercial sale, could impair Janssen's ability to meet the market demand for imetelstat and therefore result in decreased sales and reduced royalties for us.
We have not yet negotiated our agreement with Janssen specifying all of the terms for our co-promotion of imetelstat should we exercise our U.S. Co-Promotion Option. In addition, we do not have a sales force and may not develop one.
Pursuant to the Collaboration Agreement with Janssen, we have a U.S. Co-Promotion Option if we exercise our U.S. Opt-In Rights. Assuming we exercise the U.S. Co-Promotion Option, we can elect to provide 20% of the U.S. imetelstat selling effort with sales force personnel, in lieu of funding 20% of U.S. promotion costs upon regulatory approval and commercial launch of imetelstat in the United States. While the Collaboration Agreement includes the material terms of our U.S. Co-Promotion Option, we and Janssen mutually agreed to negotiate a separate agreement specifying the detailed activities and responsibilities with respect to the marketing and co-promotion of imetelstat following our election to exercise our U.S. Co-Promotion Option. We will need to negotiate this separate agreement with Janssen and, as a result, Janssen may place restrictions or additional obligations on us, including financial obligations. Any restrictions or additional obligations may restrict our co-promotion activities or involve more significant financial or other obligations than we currently anticipate. In addition, we have no sales experience as a company, and there are risks involved with establishing our own sales force capabilities. Developing an internal sales force and function will require substantial expenditures and will be time-consuming, may expose us to unforeseen costs and expenses, and we may not be able to effectively recruit, train or retain sales personnel. Accordingly, we may be unable to establish our own sales force which could effectively preclude us from participating in co-promoting imetelstat in the United States. In addition, any sales force we establish may not be effective, or may be less effective than any sales force that Janssen utilizes to promote imetelstat. In such event, the commercialization of imetelstat may be adversely affected, which could materially and adversely affect any sales milestone or royalties we may receive under the Collaboration Agreement.
The Collaboration Agreement limits our ability to transfer our U.S. Co-Promotion Option to a potential acquirer.
Although the Collaboration Agreement permits us to be acquired by any company, our right to transfer our U.S. Co-Promotion Option to a potential acquirer is limited, and subject to Janssen's sole discretion under certain circumstances. If we are acquired under such limited circumstances, then we may not be able to transfer the U.S. Co-Promotion Option to such acquirer as part of the acquisition. This limiting provision may discourage potential acquisition bids for us or lower our value thus preventing holders of our common stock from benefiting from what they may believe are the positive aspects of an acquisition, including the potential realization of a higher rate of return on their investment from this type of transaction.
RISKS RELATED TO PROTECTING OUR INTELLECTUAL PROPERTY
We remain responsible for prosecuting, at Janssen's direction, the patents we have exclusively licensed to Janssen. The success of our collaboration with Janssen will depend on our ability to protect our technologies and imetelstat, through patents and other intellectual property rights.
Protection of our proprietary technology is critically important to our business, especially with respect to our collaboration with Janssen. Our success will depend in part on our ability to obtain, enforce and extend our patents and maintain trade secrets, both in the United States and in other
43
countries. If we are unsuccessful in any of these regards, the value of our technologies and imetelstat will be adversely affected, and we and/or Janssen may be unable to continue development of imetelstat. By way of example, we do not yet have issued compound patent coverage for imetelstat in Europe after 2020. Further, our patents may be challenged, invalidated or circumvented, and our patent rights may not provide proprietary protection or competitive advantages to us or Janssen. In the event that we or our licensors are unsuccessful in obtaining and enforcing patents, we or Janssen may not be able to further develop or commercialize imetelstat, any of which could delay future clinical trials and any applications for regulatory approval and therefore delay the payment of potential milestone payments to us, or, if imetelstat is approved for commercial sale, could impair Janssen's ability to sell imetelstat and therefore result in decreased sales and reduced royalties for us. Occurrence of any of these events could negatively impact our collaboration with Janssen or cause Janssen to terminate the Collaboration Agreement, which would materially and adversely affect our business, and we may be unable to continue our operations.
The patent positions of pharmaceutical and biopharmaceutical companies, including ours, are highly uncertain and involve complex legal and technical questions. In particular, legal principles for biotechnology and pharmaceutical patents in the United States and in other countries are evolving, and the extent to which we will be able to obtain patent coverage to protect our technologies and imetelstat, or enforce issued patents, is uncertain. If we or Janssen infringe the patents of others, we or Janssen may be blocked from continuing development work or be required to obtain licenses on terms that may impact the value of imetelstat or cause it to be commercially impracticable.
A number of significant changes to U.S. patent law occurred when the Leahy-Smith America Invents Act, or the AIA, was signed into law on September 16, 2011. These include provisions that affect the way patent applications will be prosecuted and may affect patent litigation. The United States Patent and Trademark Office, or the Patent Office, has developed new and untested regulations and procedures to govern the full implementation of the AIA. Many of the substantive changes to patent law associated with the AIA, and in particular, the first to file provisions, became effective on March 16, 2013. For example, under the AIA, patent rights are awarded to the first inventor to file a patent application with respect to a particular invention. Since the publication of discoveries in scientific or patent literature tends to lag behind actual discoveries by at least several months and sometimes several years, the persons or entities that we or our licensors name as inventors in our patents and patent applications may not have been the first to invent the inventions disclosed in the patent applications or patents, or the first to file patent applications for these inventions. As a result, we may not be able to obtain patents for discoveries that we otherwise would consider patentable and that we consider to be extremely significant to the future success of imetelstat. Thus, our ability to protect our patentable intellectual property depends, in part, on our ability to be the first to file patent applications with respect to our inventions or joint inventions with Janssen. Delay in the filing of a patent application for any purpose, including further development or refinement of an invention, may result in the risk of loss of patent rights. The AIA and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents. Significant impairment of our imetelstat patent rights would have a material adverse effect on our business and could cause Janssen to terminate the Collaboration Agreement which would severely and adversely affect our business prospects.
The U.S. Supreme Court, or the Court, has also issued decisions affecting patents. On June 13, 2013, in Association for Molecular Pathology v. Myriad Genetics, Inc. the Court held that claims to isolated genomic DNA were not patentable subject matter, but claims to complementary DNA, or cDNA, molecules were patentable subject matter. The effect of the decision on patents for other isolated natural products is uncertain. On March 20, 2012, in Mayo Collaborative Services, DBA Mayo Medical Laboratories, et al. v. Prometheus Laboratories, Inc., the Court held that several claims drawn to measuring drug metabolite levels from patient samples and correlating them to drug doses were not
44
patentable subject matter. The decision has created uncertainty around the ability to patent certain biomarker-related method patents. These decisions have increased the uncertainty with regard to our ability to obtain patents in the future as well as the value of current and future patents, once obtained. Depending on decisions by the U.S. federal courts and the Patent Office, the interpretation of laws and regulations governing patents could change in unpredictable ways that would weaken our ability to obtain new patents or to enforce our existing patents. Occurrence of these events could significantly impair our imetelstat patent rights which would have a material adverse effect on our business and could cause Janssen to terminate the Collaboration Agreement.
Challenges to our patent rights can result in costly and time-consuming legal proceedings that may prevent or limit development of imetelstat.
Our patents may be challenged through administrative or judicial proceedings. Such proceedings are typically lengthy and complex, and an adverse decision can result in the loss of important patent rights. For example, where more than one party seeks U.S. patent protection for the same technology, the Patent Office may declare an interference proceeding in order to ascertain the party to which the patent should be issued. Patent interferences are typically complex, highly contested legal proceedings, subject to appeal. They are usually expensive and prolonged, and can cause significant delay in the issuance of patents. Our pending patent applications, or our issued patents, may be drawn into interference proceedings or be challenged through post-grant review procedures, which may delay or prevent the issuance of patents, or result in the loss of issued patent rights.
Under the AIA, interference proceedings have been eliminated for patent applications filed on or after March 16, 2013, and have been replaced with other types of proceedings, including derivation proceedings. The AIA also includes post-grant review procedures subjecting U.S. patents to post-grant review procedures similar to European oppositions. U.S. patents owned or licensed by us may therefore be subject to post-grant review procedures, as well as other forms of review and re-examination. A decision in such proceedings adverse to our interests could result in the loss of valuable patent rights which would have a material adverse effect on our business and could cause Janssen to terminate the Collaboration Agreement which would severely and adversely affect our business prospects.
Certain jurisdictions, such as Europe, New Zealand and Australia, permit oppositions to be filed against granted patents or patents proposed to be granted. Under the Collaboration Agreement, Janssen could commercialize imetelstat internationally if approved by regulatory authorities for commercial sale. Therefore, securing both proprietary protection and freedom to operate outside of the United States is important to the Collaboration Agreement with Janssen and our business.
We have been involved in both opposing the grant of patents to others through such opposition proceedings and in defending our patent applications against oppositions filed by others. These opposition proceedings required significant time and costs to protect our intellectual property rights. If we are unable to commit these types of resources for our imetelstat patent rights, we and/or Janssen could be prevented or limited in the development and commercialization of imetelstat. Occurrence of any of these events could negatively impact our collaboration with Janssen or cause Janssen to terminate the Collaboration Agreement which would severely and adversely affect our business prospects.
45
As more groups become engaged in scientific research and product development in the areas of telomerase biology, the risk of our patents, or patents that we have in-licensed, being challenged through patent interferences, derivation proceedings, oppositions, re-examinations, litigation or other means will likely increase. Challenges to our patents through these procedures can be extremely expensive and time-consuming, even if the outcome is favorable to us. An adverse outcome in a patent dispute could severely harm our collaboration with Janssen or cause Janssen to terminate the Collaboration Agreement, or could otherwise have a material adverse effect on our business by:
- •
- causing us to lose patent rights in the relevant jurisdiction(s);
- •
- subjecting us to litigation, or otherwise preventing Janssen or us from commercializing imetelstat in the relevant jurisdiction(s);
- •
- requiring Janssen or us to obtain licenses to the disputed patents;
- •
- forcing Janssen or us to cease using the disputed technology; or
- •
- requiring Janssen or us to develop or obtain alternative technologies.
We or Janssen may be subject to infringement claims that are costly to defend, and as to which we may be obligated to indemnify or obtain unblocking licenses, and such claims may limit our or Janssen's ability to use disputed technologies and prevent us or Janssen from pursuing research and development or commercialization of imetelstat.
The commercial success of imetelstat will depend upon our and Janssen's ability to develop, manufacture, market and sell imetelstat without infringing or otherwise violating the intellectual property and other proprietary rights of third parties. There is considerable intellectual property litigation in the biotechnology and pharmaceutical industries, and many pharmaceutical companies, including potential competitors, have substantial patent portfolios. In the event our technologies infringe the rights of others or require the use of discoveries and technologies controlled by third parties, we or Janssen may be prevented from pursuing research, development, manufacturing or commercialization of imetelstat, or may be required to obtain licenses to those patents or other proprietary rights or develop or obtain alternative technologies. For example, we are aware that certain third parties have or may be prosecuting patents and patent estates that may relate to imetelstat, and while we believe these patents will expire before imetelstat is commercialized and/or that these patents are invalid and/or would not be infringed by the manufacture, use or sale of imetelstat, it is possible that the owner(s) of these patents will assert claims against us and/or Janssen in the future. Under the Collaboration Agreement, we are obligated under certain circumstances to indemnify Janssen from any claim of infringement of the patent rights of third parties in Janssen's development, manufacture or commercialization of imetelstat, or to obtain unblocking licenses from such third parties, at our cost.
Since we cannot be aware of all intellectual property rights potentially relating to imetelstat and its uses, we do not know with certainty that imetelstat, or the intended commercialization thereof, does not and will not infringe or otherwise violate any third party's intellectual property. Any infringement claims against us or Janssen would likely be expensive to resolve, and the cost of any indemnification of Janssen or unblocking license that we could be required to obtain under the Collaboration Agreement is unpredictable and could be significant. If we or Janssen are unable to resolve an infringement claim successfully, we or Janssen could be subject to an injunction which would prevent us or Janssen from commercializing imetelstat, and could also require us or Janssen to pay substantial damages. In addition to infringement claims, in the future we or Janssen may also be subject to other claims relating to intellectual property, such as claims that we or Janssen have misappropriated the trade secrets of third parties. We expect that as imetelstat continues to progress in development, we will see more efforts by others to obtain patents that are positioned to cover imetelstat. Our success therefore
46
depends significantly on our and Janssen's ability to operate without infringing patents and the proprietary rights of others.
We or Janssen may become aware of discoveries and technologies controlled by third parties that are advantageous to developing or manufacturing imetelstat. Under such circumstances, we may initiate negotiations for licenses to other technologies as the need or opportunity arises. We, or Janssen, may not be able to obtain a license to a technology required for the research, development, manufacturing or commercialization of imetelstat on commercially favorable terms, or at all, or our licenses may be terminated on certain grounds, including as a result of our failure to comply with our obligations under such licenses. If we or Janssen do not obtain a necessary license or if such a license is terminated, we or Janssen may need to redesign our technologies or obtain rights to alternate technologies, which may not be possible, and even if possible, could cause delays in the development efforts for imetelstat. In cases where we or Janssen are unable to license necessary technologies, we and/or Janssen could be subject to litigation and prevented from developing imetelstat, and in certain circumstances we may be required to indemnify Janssen for infringement claims arising from Janssen's development, manufacture or commercialization of imetelstat, which could materially and adversely impact our business. Failure by us or Janssen to obtain alternative technologies or a license to any technology that may be required to research, develop, manufacture or commercialize imetelstat would delay future clinical trials and any applications for regulatory approval and therefore delay the payment of potential milestone payments to us, or, if imetelstat is approved for commercial sale, could impair Janssen's ability to sell imetelstat and therefore result in decreased sales and reduced royalties for us. Occurrence of any of these events could negatively impact our collaboration with Janssen or cause Janssen to terminate the Collaboration Agreement which would materially and adversely affect our business, and we may be unable to continue our operations.
We may become involved in disputes with Janssen or any past or future collaborator(s) over intellectual property inventorship or ownership, and publications by our investigators, scientific consultants and research collaborators could impair our ability to obtain patent protection or protect our proprietary information, which, in either case, could have a significant impact on our business.
Inventions discovered under research, material transfer or other such collaborative agreements, including our Collaboration Agreement with Janssen, may become jointly owned by us and the other party to such agreements in some cases and the exclusive property of either party in other cases. Under some circumstances, it may be difficult to determine who invents and owns a particular invention, or whether it is jointly owned, and disputes could arise regarding inventorship and ownership of those inventions. These disputes could be costly and time consuming and an unfavorable outcome could have a significant adverse effect on our business if we were not able to protect or license rights to these inventions. In addition, clinical trial investigators, scientific consultants and research collaborators generally have contractual rights to publish data and other proprietary information, subject to review by us and/or Janssen. Publications by investigators, scientific consultants and research collaborators containing such information, either with permission or in contravention of the terms of their agreements, may impair the ability to obtain patent protection or protect proprietary information which would have a material adverse effect on our business and could cause Janssen to terminate the Collaboration Agreement.
Much of the information and know-how that is critical to our business is not patentable, and we may not be able to prevent others from obtaining this information and establishing competitive enterprises.
We sometimes rely on trade secrets to protect our proprietary technology, especially in circumstances in which we believe patent protection is not appropriate or available. We attempt to protect our proprietary technology in part by confidentiality agreements with our employees, consultants, collaborators and contractors. We cannot provide assurance that these agreements will not
47
be breached, that we would have adequate remedies for any breach, or that our trade secrets will not otherwise become known or be independently discovered by competitors, any of which would harm our business significantly.
RISKS RELATED TO OUR FINANCIAL POSITION AND NEED FOR ADDITIONAL FINANCING
We have a history of losses and anticipate continued future losses, and our continued losses could impair our ability to sustain operations.
We have incurred operating losses every year since our operations began in 1990. As of December 31, 2014, our accumulated deficit was approximately $928.4 million. Losses have resulted principally from costs incurred in connection with our research and development activities and from general and administrative costs associated with our operations. We expect to incur additional operating losses and, as clinical development activities continue under our Collaboration Agreement with Janssen, our operating losses may increase in size.
Substantially all of our revenues to date have been research support payments under collaborative agreements and milestones, royalties and other revenues from our licensing arrangements. Any revenues generated from ongoing collaborative agreements and revenues from our licensing arrangements, including the Collaboration Agreement with Janssen, may not be sufficient alone to sustain our operations. In addition, there can be no assurance that we will receive any milestone payments or royalties from Janssen in the future. We may be unsuccessful in entering into any new corporate collaboration, partnership or license agreements that result in revenues, or existing collaborative agreements or license arrangements, such as the Collaboration Agreement with Janssen, may be terminated or expire.
We also expect to experience negative cash flow for the foreseeable future as we fund our operating losses and capital expenditures. This will result in decreases in our working capital, total assets and stockholders' equity, which may not be offset by milestone payments or royalties from Janssen or by future financings. We will need to generate significant revenues to achieve profitability. We may not be able to generate these revenues from the Collaboration Agreement with Janssen through milestone payments or royalties, and we may never achieve profitability. Our failure to achieve profitability could negatively impact the market price of our common stock and our ability to sustain operations. Even if we do become profitable, we may not be able to sustain or increase profitability on a quarterly or annual basis.
We may require additional capital to support development and commercialization of imetelstat in collaboration with Janssen and to otherwise grow our business, and our ability to obtain the necessary funding is uncertain.
We may need additional capital resources in order to support development and commercialization of imetelstat, especially if we elect to exercise our U.S. Opt-In Rights and U.S. Co-Promotion Option under the Collaboration Agreement and potentially independently pursue imetelstat development under our own IDP, and to otherwise support the future growth of our business through the acquisition of other oncology products, programs or companies. We cannot assure you that our existing capital resources, future interest income, potential milestone payments and royalties under the Collaboration Agreement with Janssen and potential future sales of our common stock, including pursuant to our At-The-Market Issuance Sales Agreement, or sales agreement, with MLV & Co. LLC, or MLV, will be sufficient to fund future planned activities. The timing and degree of any future capital requirements will depend on many factors, including:
- •
- the accuracy of the assumptions underlying our estimates for our capital needs;
- •
- whether we elect U.S. Opt-In Rights to share future U.S. development and promotion costs for imetelstat under the Collaboration Agreement with Janssen;
48
- •
- to the extent permitted under the Collaboration Agreement, whether we independently pursue imetelstat development under our own IDP;
- •
- our potential reimbursement obligations to Janssen if any data from a Janssen IDP support approval by a regulatory agency in the
United States or other countries;
- •
- the achievement of development, regulatory and commercial milestones resulting in the payment to us from Janssen under the
Collaboration Agreement and the timing of receipt of such payments, if any;
- •
- changes or delays in our and Janssen's development plans for imetelstat, including changes which may result from any future clinical
holds on the INDs for imetelstat, including the IND for the MF Pilot Study, that we expect to transfer to Janssen;
- •
- Janssen's ability to meaningfully reduce manufacturing costs of imetelstat;
- •
- the progress, timing, magnitude, scope and costs of clinical development, manufacturing and commercialization for imetelstat,
including the number of indications being pursued, subject to permission from the FDA;
- •
- the time and costs involved in obtaining regulatory clearances and approvals in the United States and in other countries;
- •
- Janssen's ability to successfully market and sell imetelstat, upon regulatory approval or clearance, in the United States and other
countries;
- •
- our decision to exercise our U.S. Co-Promotion Option, including the costs and timing of building a U.S. sales force;
- •
- the timing, receipt and amount of royalties under the Collaboration Agreement on worldwide net sales of imetelstat, upon regulatory
approval or clearance, if any;
- •
- the timing, receipt and amount of royalties on sales of any stem cell products by Asterias, upon development, regulatory approval or
clearance, if any;
- •
- the sales price and availability of adequate third-party reimbursement for imetelstat;
- •
- the cost of acquiring and/or licensing any new product candidates, if any;
- •
- expenses associated with the pending and potential additional related purported securities lawsuits and derivative lawsuits, as well
as any other litigation; and
- •
- the costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing patent claims.
In addition, changes in our business may occur that would consume available capital resources sooner than we expect. If our existing capital resources, future interest income, and potential milestone payments and royalties under the Collaboration Agreement with Janssen are insufficient to meet future capital requirements, we will need to raise additional capital to fund our operations. Further, if the Collaboration Agreement is terminated, including as a result of Janssen's failure to provide an affirmative Continuation Decision to us, we would not receive any milestone payments or royalties under the Collaboration Agreement, and we would be required to fund all clinical development, manufacturing and commercial activities for imetelstat ourselves, which would require us to raise additional capital or establish alternative collaborations with third-party collaboration partners, which may not be possible. Additional financing through public or private equity financings, including pursuant to our sales agreement with MLV prior to its expiration in October 2015, capital lease transactions or other financing sources may not be available on acceptable terms, or at all. We may raise equity capital at a stock price or on other terms that could result in substantial dilution of
49
ownership for our stockholders. The receptivity of the public and private equity markets to proposed financings is substantially affected by the general economic, market and political climate and by other factors which are unpredictable and over which we have no control.
Our ability to raise additional funds will be severely impaired in the event of:
- •
- any future clinical holds on any IND for imetelstat;
- •
- failure to show adequate safety or efficacy of imetelstat in existing or potential future clinical trials; or
- •
- a termination of the Collaboration Agreement or our collaboration with Janssen is otherwise unsuccessful.
If sufficient capital is not available, we may be unable to fulfill our funding obligations under the Collaboration Agreement with Janssen resulting in our breach of the Collaboration Agreement which could lead to Janssen paying lower milestone payments and lower royalties to us under a reduced royalty tier which could have a material adverse effect on our results of operations and financial condition.
Moreover, we plan to diversify our sole product candidate development risk by identifying and seeking to acquire or in-license new product opportunities for development. Acquisition or in-licensing opportunities that we may pursue could materially affect our liquidity and capital resources and may require us to incur indebtedness, seek equity capital or both. In addition, there can be no assurance that sufficient additional capital would be available to us in order to pursue any of these opportunities.
Our ability to use our net operating loss carryforwards and certain other tax attributes may be limited.
Under Section 382 of the Internal Revenue Code of 1986, as amended, or the Code, if a corporation undergoes an "ownership change," generally defined as a greater than 50% change (by value) in its equity ownership over a three-year period, the corporation's ability to use its pre-change net operating loss carryforwards and other pre-change tax attributes (such as research tax credits) to offset its post-change taxable income or taxes may be limited. Changes in our stock ownership, some of which are outside of our control, may have resulted or could in the future result in an ownership change. If a limitation were to apply, utilization of a portion of our domestic net operating loss and tax credit carryforwards could be limited in future periods. In addition, a portion of the carryforwards may expire before being available to reduce future income tax liabilities.
RISKS RELATED TO OUR COMMON STOCK AND FINANCIAL REPORTING
Historically, our stock price has been extremely volatile.
Historically, our stock price has been extremely volatile. Between January 1, 2005 and December 31, 2014, our stock has traded as high as $12.18 per share and as low as $0.91 per share. Between January 1, 2012 and December 31, 2014, the price has ranged between a high of $7.79 per share and a low of $0.91 per share. The significant market price fluctuations of our common stock have been due to and may in the future be influenced by a variety of factors, including:
- •
- not receiving timely regulatory clearances or approvals in any jurisdiction, whether within or outside of the United States, including, if we, Janssen or future investigators do not obtain regulatory clearance to commence studies of imetelstat in MF, MDS or any additional hematologic myeloid malignancies in a timely manner or at all, including the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study planned to be conducted by Janssen under the Collaboration Agreement;
50
- •
- developments in our collaboration with Janssen, including the termination or modification of the Collaboration Agreement or disputes
regarding the collaboration;
- •
- announcements regarding the research and development of imetelstat, including results of or delays in any clinical trials of
imetelstat, and investor perceptions thereof;
- •
- announcements regarding the safety of imetelstat, including announcements similar to our March 2014 announcements that the FDA had
placed a full clinical hold on our IND for imetelstat and a partial clinical hold on the investigator's IND for the MF Pilot Study due to safety concerns;
- •
- announcements regarding our plans to discontinue certain programs or clinical trials, such as our prior announcements regarding the
discontinuation of our stem cell programs and certain clinical trials;
- •
- perception by our stockholders about the adequacy of the consideration received for the divestiture of our stem cell assets to
Asterias or potential payments we may receive under the Collaboration Agreement;
- •
- the demand in the market for our common stock;
- •
- the experimental nature of imetelstat;
- •
- fluctuations in our operating results;
- •
- our declining cash balance as a result of operating losses;
- •
- general market conditions or market conditions relating to the biopharmaceutical and pharmaceutical industries;
- •
- announcements of technological innovations, new commercial products, or clinical progress or lack thereof by us, our collaborators,
licensees, partners or our competitors;
- •
- announcements concerning regulatory developments and proprietary rights;
- •
- comments by securities analysts;
- •
- large stockholders exiting their position in our common stock;
- •
- announcements of or developments concerning pending and/or potential future litigation;
- •
- the issuance of common stock to partners, vendors or investors to raise additional capital; and
- •
- the occurrence of any other risks and uncertainties discussed under the heading "Risk Factors."
Stock prices and trading volumes for many biopharmaceutical companies fluctuate widely for a number of reasons, including factors which may be unrelated to their businesses or results of operations, such as media coverage, legislative and regulatory measures and the activities of various interest groups or organizations. In addition to other risk factors described in this section, overall market volatility, as well as general domestic or international economic, market and political conditions, could materially and adversely affect the market price of our common stock and the return on your investment.
If we fail to meet continued listing standards of NASDAQ, our common stock may be delisted, which could have a material adverse effect on the liquidity of our common stock.
Our common stock is currently traded on the Nasdaq Global Select Market. The NASDAQ Stock Market LLC has requirements that a company must meet in order to remain listed on NASDAQ. In particular, NASDAQ rules require us to maintain a minimum bid price of $1.00 per share of our common stock. If the closing bid price of our common stock were to fall below $1.00 per share for 30 consecutive trading days or we do not meet other listing requirements, we would fail to be in
51
compliance with NASDAQ's listing standards. There can be no assurance that we will continue to meet the minimum bid price requirement, or any other requirement in the future. If we fail to meet the minimum bid price requirement, The NASDAQ Stock Market LLC may initiate the delisting process with a notification letter. If we were to receive such a notification, we would be afforded a grace period of 180 calendar days to regain compliance with the minimum bid price requirement. In order to regain compliance, shares of our common stock would need to maintain a minimum closing bid price of at least $1.00 per share for a minimum of 10 consecutive trading days. In addition, we may be unable to meet other applicable NASDAQ listing requirements, including maintaining minimum levels of stockholders' equity or market values of our common stock in which case, our common stock could be delisted. If our common stock were to be delisted, the liquidity of our common stock would be adversely affected and the market price of our common stock could decrease.
The sale of a substantial number of shares may adversely affect the market price of our common stock.
The sale of a substantial number of shares of our common stock in the public market, or the perception that such sales could occur, could significantly and negatively affect the market price of our common stock. As of December 31, 2014, we had 300,000,000 shares of common stock authorized for issuance and 157,429,871 shares of common stock outstanding. In addition, we had reserved 32,327,345 shares of our common stock for future issuance pursuant to our option and equity incentive plans and outstanding warrants as of December 31, 2014. Issuing additional shares could negatively affect the market price of our common stock and the return on your investment.
Future sales of our common stock, including pursuant to our sales agreement with MLV prior to its expiration in October 2015, or the issuance of common stock to satisfy our current or future cash payment obligations or to acquire technology, property, or other businesses, could cause immediate dilution and adversely affect the market price of our common stock. In addition, under the universal shelf registration statement filed by us in July 2012 and declared effective by the SEC in October 2012, we may sell any combination of common stock, preferred stock, debt securities and warrants in one or more offerings, up to a cumulative value of $96.5 million. The sale or issuance of our securities, as well as the existence of outstanding options and shares of common stock reserved for issuance under our option and equity incentive plans and outstanding warrants also may adversely affect the terms upon which we are able to obtain additional capital through the sale of equity securities, which could negatively affect the market price of our common stock and the return on your investment.
Our undesignated preferred stock may inhibit potential acquisition bids; this may adversely affect the market price of our common stock and the voting rights of holders of our common stock.
Our certificate of incorporation provides our board of directors with the authority to issue up to 3,000,000 shares of undesignated preferred stock and to determine or alter the rights, preferences, privileges and restrictions granted to or imported upon these shares without further vote or action by our stockholders. The issuance of shares of preferred stock may delay or prevent a change in control transaction without further action by our stockholders. As a result, the market price of our common stock may be adversely affected.
In addition, if in the future, we issue preferred stock that has preference over our common stock with respect to the payment of dividends or upon our liquidation, dissolution or winding up, or if we issue preferred stock with voting rights that dilute the voting power of our common stock, the rights of holders of our common stock or the market price of our common stock could be adversely affected.
52
Provisions in our charter, bylaws and Delaware law may inhibit potential acquisition bids for us, which may prevent holders of our common stock from benefiting from what they believe may be the positive aspects of acquisitions and takeovers.
Provisions of our charter documents and bylaws may make it substantially more difficult for a third party to acquire control of us and may prevent changes in our management, including provisions that:
- •
- prevent stockholders from taking actions by written consent;
- •
- divide the board of directors into separate classes with terms of office that are structured to prevent all of the directors from
being elected in any one year; and
- •
- set forth procedures for nominating directors and submitting proposals for consideration at stockholders' meetings.
Provisions of Delaware law may also inhibit potential acquisition bids for us or prevent us from engaging in business combinations. In addition, we have severance agreements with several employees and a company-wide severance plan, either of which could require a potential acquirer to pay a higher price. Either collectively or individually, these provisions may prevent holders of our common stock from benefiting from what they may believe are the positive aspects of acquisitions and takeovers, including the potential realization of a higher rate of return on their investment from these types of transactions.
We do not intend to pay cash dividends on our common stock in the foreseeable future.
We do not anticipate paying cash dividends on our common stock in the foreseeable future. Any payment of cash dividends will depend upon our financial condition, results of operations, capital requirements and other factors and will be at the discretion of our board of directors.
Failure to achieve and maintain effective internal controls in accordance with Section 404 of the Sarbanes-Oxley Act of 2002 could have a material adverse effect on our business and stock price.
Section 404 of the Sarbanes-Oxley Act of 2002, or Section 404, requires that we establish and maintain an adequate internal control structure and procedures for financial reporting. Our annual reports on Form 10-K must contain an assessment by management of the effectiveness of our internal control over financial reporting and must include disclosure of any material weaknesses in internal control over financial reporting that we have identified. In addition, our independent registered public accounting firm must annually provide an opinion on the effectiveness of our internal control over financial reporting.
The requirements of Section 404 are ongoing and also apply to future years. We expect that our internal control over financial reporting will continue to evolve as our business develops. Although we are committed to continue to improve our internal control processes and we will continue to diligently and vigorously review our internal control over financial reporting in order to ensure compliance with Section 404 requirements, any control system, regardless of how well designed, operated and evaluated, can provide only reasonable, not absolute, assurance that its objectives will be met. Therefore, we cannot be certain that in the future material weaknesses or significant deficiencies will not exist or otherwise be discovered. If material weaknesses or other significant deficiencies occur, these weaknesses or deficiencies could result in misstatements of our results of operations, restatements of our financial statements, a decline in our stock price, or other material adverse effects on our business, reputation, results of operations, financial condition or liquidity.
53
RISKS RELATED TO COMPETITIVE FACTORS
Competitors may develop technologies that are superior to or more cost-effective than ours, which may significantly impact the commercial viability of imetelstat which could cause Janssen to terminate the Collaboration Agreement and damage our ability to sustain operations.
The pharmaceutical and biotechnology industries are intensely competitive. Other pharmaceutical and biotechnology companies and research organizations currently engage in or have in the past engaged in efforts related to the biological mechanisms related to imetelstat, including the study of telomeres, telomerase and our proprietary oligonucleotide chemistry, and the research and development of therapies for the treatment of hematologic myeloid malignancies. In addition, other products and therapies that could directly compete with imetelstat currently exist or are being developed by pharmaceutical and biopharmaceutical companies and by academic institutions, government agencies and other public and private research organizations.
Many companies are developing alternative therapies to treat hematologic myeloid malignancies and, in this regard, are competitors of ours and Janssen. For example, if approved for commercial sale for the treatment of MF, imetelstat would compete against Incyte Corporation's ruxolitinib, or Jakafi®, which is orally administered. In clinical trials, Jakafi® reduced spleen size, abdominal discomfort, early satiety, bone pain, night sweats and itching in MF patients. Recently, there have also been reports of overall survival benefit as well as improvement in bone marrow fibrosis from Jakafi® treatment. Other treatment modalities for MF include hydroxyurea for the management of splenomegaly, leukocytosis, thrombocytosis and constitutional symptoms; splenectomy and splenic irradiation for the management of splenomegaly and co-existing cytopenias, or low blood cell counts; chemotherapy and pegylated interferon. Drugs for the treatment of MF-associated anemia include erythropoiesis-stimulating agents, androgens, danazol, corticosteroids, thalidomide and lenalidomide. There are other investigational treatments further along in development than imetelstat, such as momelotinib by Gilead Sciences, Inc. and pacritinib by Cell Therapeutics, Inc., which are currently in Phase 3 clinical trials, and other inhibitors of the JAK-STAT pathway, as well as several investigational treatments in early phase testing such as histone deacetylase inhibitors, inhibitors of heat shock protein 90, hypomethylating agents, PI3 Kinase and mTOR inhibitors, hedgehog inhibitors, anti-LOX2 inhibitors, recombinant pentraxin 2 protein, KIP-1 activators, TGF-beta inhibitors, FLT inhibitors, and other tyrosine kinase inhibitors.
Independently, Janssen is developing therapies for hematologic malignancies, including AML, MDS, MM and ABC-subtype diffuse large B-cell lymphoma. Molecular and cellular pathways of interest include:
- •
- cell surface targets for immune-directed therapy;
- •
- immune checkpoint inhibition;
- •
- leukemia stem cells;
- •
- pathway addiction (genetic alterations, cell-type specific pathways);
- •
- conditional sensitivity (stress, protein-producing tumors);
- •
- targeting of T-cells and natural killer "NK" cells to tumors;
- •
- identification of novel tumor-specific antigens; and
- •
- progression from early MDS to AML and cancer interception.
Success by Janssen in any of these approaches may compete with imetelstat or render imetelstat obsolete or noncompetitive, which could lead to a decision by Janssen to discontinue the imetelstat program and terminate the Collaboration Agreement which would materially and adversely affect our business and business prospects.
54
Smaller companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. We anticipate increased competition in the future as new companies explore treatments for hematologic myeloid malignancies, which may significantly impact the commercial viability of imetelstat. Academic institutions, government agencies and other public and private research organizations may also conduct research, seek patent protection and establish collaborative arrangements for research, clinical development and marketing of products similar to ours. These companies and institutions compete with us in recruiting and retaining qualified scientific and management personnel as well as in acquiring technologies complementary to our imetelstat program.
In addition to the above factors, we and Janssen expect to face competition in the following areas:
- •
- product efficacy and safety;
- •
- convenience of product administration;
- •
- cost of manufacturing;
- •
- the timing and scope of regulatory consents;
- •
- status of reimbursement coverage;
- •
- price; and
- •
- patent position, including potentially dominant patent positions of others.
As a result of the foregoing, competitors may develop more commercially desirable or affordable products, or achieve earlier patent protection or product commercialization than us or Janssen. Competitors have developed, or are in the process of developing, technologies that are, or in the future may be, competitive to imetelstat. Some of these products may have an entirely different approach or means of accomplishing therapeutic effects similar to those that may be demonstrated by imetelstat. Competitors may develop products that are safer, more effective or less costly than imetelstat, or more convenient to administer to patients and, therefore, present a serious competitive threat to imetelstat. In addition, competitors may price their products below what Janssen may determine to be an acceptable price for imetelstat, may receive better third-party payor coverage and/or reimbursement, or may be more cost effective than imetelstat. Such competitive products or activities by competitors may render imetelstat obsolete, which may cause Janssen to terminate the Collaboration Agreement which would severely and adversely affect our business prospects.
To be successful, imetelstat must be accepted by the health care community, which can be very slow to adopt or unreceptive to new technologies and products.
If approved for marketing, imetelstat may not achieve market acceptance since hospitals, physicians, patients or the medical community in general may decide not to accept and utilize imetelstat. If approved for commercial sale, imetelstat will compete with a number of conventional and widely accepted drugs and therapies manufactured and marketed by major pharmaceutical companies. The degree of market acceptance of imetelstat will depend on a number of factors, including:
- •
- the establishment and demonstration to the medical community of the clinical efficacy and safety of imetelstat;
- •
- the ability to demonstrate that imetelstat is superior to alternatives currently on the market;
- •
- the ability to establish in the medical community the potential advantage of imetelstat over alternative treatment methods;
- •
- the label and promotional claims allowed by the FDA or other regulatory agencies for imetelstat, if any;
55
- •
- sales, marketing and distribution support for imetelstat; and
- •
- reimbursement policies of government and third-party payors.
The established use of conventional products competitive with imetelstat may limit or preclude the potential for imetelstat to receive market acceptance upon any commercialization. We or Janssen may be unable to demonstrate any pharmacoeconomic advantage for imetelstat compared to established or standard-of-care therapies, or newly developed therapies, for hematologic myeloid malignancies. Third-party payors may decide that any potential improvement that imetelstat may provide to clinical outcomes in hematologic myeloid malignancies is not adequate to justify the costs of treatment with imetelstat. If third-party payors do not view imetelstat as offering a better balance between clinical benefit and treatment cost compared to standard-of-care therapies or other treatment modalities currently in development, imetelstat may not be commercially viable. If the health care community does not accept imetelstat for any of the foregoing reasons, or for any other reason, our ability to earn potential milestone payments and royalties under the Collaboration Agreement with Janssen would be negatively impacted and our business prospects would be severely and adversely affected.
If acceptable prices or adequate reimbursement for imetelstat is not obtained, the use of imetelstat could be severely limited.
The ability to successfully commercialize imetelstat will depend significantly on obtaining acceptable prices and the availability of reimbursement to the patient from third-party payors. In March 2010, the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act, collectively known as the Affordable Care Act, became law and substantially changed the way healthcare will be financed by both governmental and private insurers, and significantly impacted the pharmaceutical industry. The Affordable Care Act contains a number of provisions, including those governing enrollment in federal healthcare programs, reimbursement changes and fraud and abuse, which will impact existing government healthcare programs and will result in the development of new programs, including Medicare payment for performance initiatives and improvements to the physician quality reporting system and feedback program. Additionally, the Affordable Care Act:
- •
- mandates a further shift in the burden of Medicaid payments to the states;
- •
- increases the minimum level of Medicaid rebates payable by manufacturers of brand-name drugs from 15.1% to 23.1%;
- •
- requires collection of rebates for drugs paid by Medicaid managed care organizations;
- •
- requires manufacturers to participate in a coverage gap discount program, under which they must agree to offer 50% point-of-sale
discounts off negotiated prices of applicable brand drugs to eligible beneficiaries during their coverage gap period, as a condition for the manufacturer's outpatient drugs to be covered under
Medicare Part D, beginning January 2011; and
- •
- imposes a non-deductible annual fee on pharmaceutical manufacturers or importers who sell "branded prescription drugs" to specified federal government programs.
While the U.S. Supreme Court upheld the constitutionality of most elements of the Affordable Care Act in June 2012, other legal challenges are still pending final adjudication in several jurisdictions. In addition, the United States Congress has also proposed a number of legislative initiatives, including possible repeal of the Affordable Care Act. At this time, it remains unclear whether there will be any changes made to the Affordable Care Act, whether to certain provisions or its entirety. We cannot assure that the Affordable Care Act, as currently enacted or as amended in the future, will not adversely affect our business and financial results and we cannot predict how future federal or state legislative or administrative changes relating to healthcare reform will affect our business.
56
In addition, other legislative changes have been proposed and adopted since the Affordable Care Act was enacted. The American Taxpayer Relief Act of 2012, signed into law in January 2013, among other things, also reduced Medicare payments to several providers, including hospitals, imaging centers and cancer treatment centers, and increased the statute of limitations period for the government to recover overpayments to providers from three to five years. On March 1, 2013, the President signed an executive order implementing sequestration, and on April 1, 2013, Medicare payment reductions of 2% went into effect and will remain in effect through 2024 unless additional Congressional action is taken.
While the Affordable Care Act has likely increased the number of patients who have insurance coverage for imetelstat, it is uncertain whether its cost containment measures will adversely affect reimbursement for imetelstat. Cost control initiatives could decrease the price that Janssen may receive for imetelstat in the future. If imetelstat is not considered cost-effective or adequate third-party reimbursement for the users of imetelstat cannot be obtained, then Janssen may be unable to maintain price levels sufficient to realize an appropriate return on the investment in imetelstat, which could impair our ability to earn potential milestone payments and royalties under the Collaboration Agreement with Janssen and our financial condition, operating results and business prospects would be severely and adversely affected.
RISKS RELATED TO ENVIRONMENTAL AND PRODUCT LIABILITY
Activities conducted by us or Janssen involve hazardous materials, and improper handling of these materials by employees, contractors, or agents could expose us or Janssen to significant legal and financial penalties.
We, Janssen, or contractors or agents may be required to incur significant costs to comply with current or future environmental laws and regulations and may be adversely affected by the cost of compliance with these laws and regulations. Additionally, an accident could damage the manufacturing facilities and operations of any third-party contracted by us or Janssen to perform services with respect to imetelstat. If we, Janssen or contractors or agents are unable to comply with federal, state and county environmental and safety laws and regulations, including those governing laboratory procedures, exposure to blood-borne pathogens and the handling of biohazardous materials, chemicals and various radioactive compounds, or an accident occurs, considerable additional costs, fines, penalties or liabilities could be assessed which could negatively impact our collaboration with Janssen or cause Janssen to terminate the Collaboration Agreement, either of which would severely and adversely affect our business prospects.
We may not be able to obtain or maintain sufficient insurance on commercially reasonable terms or with adequate coverage against potential liabilities in order to protect ourselves against product liability claims.
Our business exposes us to potential product liability risks that are inherent in the testing, manufacturing and marketing of human therapeutic and diagnostic products. We may become subject to product liability claims if the use of imetelstat is alleged to have injured patients, including any injuries alleged to arise from any hepatotoxicity from imetelstat. We currently have limited clinical trial liability insurance, and we may not be able to maintain this type of insurance for any of our clinical trials or clinical trials that we may conduct in collaboration with Janssen under the Collaboration Agreement. In addition, product liability insurance is becoming increasingly expensive. Being unable to obtain or maintain product liability insurance in the future on acceptable terms or with adequate coverage against potential liabilities could have a material adverse effect on our business.
Significant disruptions of information technology systems or breaches of data security could adversely affect our business.
Our business is increasingly dependent on critical, complex and interdependent information technology systems, including Internet-based systems, to support business processes as well as internal
57
and external communications. The size and complexity of our computer systems make them potentially vulnerable to breakdown, malicious intrusion and computer viruses that may result in the impairment of key business processes.
In addition, our systems are potentially vulnerable to data security breaches—whether by employees or others—that may expose sensitive data to unauthorized persons. Such data security breaches could lead to the loss of trade secrets or other intellectual property, or could lead to the public exposure of personal information (including sensitive personal information) of our employees, collaborators, clinical trial patients, customers and others.
Such disruptions and breaches of security could have a material adverse effect on our business, financial condition and results of operations.
Our headquarters are located near known earthquake fault zones, and the occurrence of an earthquake or other catastrophic disaster could cause damage to our offices and equipment, which could cause delays or even require us to cease or curtail operations.
Our headquarters are located in the San Francisco Bay Area near known earthquake fault zones and are vulnerable to significant damage from earthquakes. We do not carry earthquake insurance. We are also vulnerable to damage from other types of disasters, including fires, floods, power loss, communications failures, terrorism and similar events. If any disaster were to occur, our ability to operate our business at our offices would be seriously, or potentially completely, impaired. The insurance we maintain may not be adequate to cover our losses from such disasters or other business interruptions.
ITEM 1B. UNRESOLVED STAFF COMMENTS
None.
In February 2014, we amended the lease agreement for our premises at 149 Commonwealth Drive, Menlo Park, California to extend the lease term through January 2016 and reduce the space leased by us to approximately 24,000 square feet of office space effective July 2014. Our amended lease at 149 Commonwealth Drive includes an option to extend the lease for one additional period of 18 months. We believe that our facilities are adequate to meet our requirements for the near term.
On March 14, 2014, a purported securities class action lawsuit was commenced in the United States District Court for the Northern District of California, or the California District Court, naming as defendants us and certain of our officers. The lawsuit alleges violations of the Securities Exchange Act of 1934 in connection with allegedly false and misleading statements made by us related to our Phase 2 trial of imetelstat in patients with ET or PV. The plaintiff alleges, among other things, that we failed to disclose facts related to the occurrence of persistent low-grade LFT abnormalities observed in our Phase 2 trial of imetelstat in ET or PV patients and the potential risk of chronic liver injury following long-term exposure to imetelstat. The plaintiff seeks damages and an award of reasonable costs and expenses, including attorneys' fees. On March 28, 2014, a second purported securities class action lawsuit was commenced in the California District Court, and on June 6, 2014, a third purported securities lawsuit, not styled as a class action, was commenced in the United States District Court for the Southern District of Mississippi, or the Mississippi District Court, naming as defendants us and certain of our officers. These lawsuits, which are based on the same factual background as the purported securities class action lawsuit that commenced on March 14, 2014, also allege violations of the Securities Exchange Act of 1934 and seek damages and an award of reasonable costs and expenses,
58
including attorneys' fees. On June 30, 2014, the California District Court consolidated both of the purported class actions filed in the California District Court and appointed a lead plaintiff and lead counsel to represent the purported class. On July 21, 2014, the California District Court ordered the lead plaintiff to file its consolidated amended complaint, which was filed on September 19, 2014. On August 11, 2014, we filed a motion to transfer the purported securities lawsuit filed in the Mississippi District Court to the California District Court. On November 4, 2014, the Mississippi District Court granted our motion and transferred the case to the California District Court, which was thereafter consolidated with the class actions. We filed our motion to dismiss the consolidated amended complaint on November 18, 2014. The plaintiff's opposition was filed on January 20, 2015, and we filed our reply on February 25, 2015. The court hearing for the motion to dismiss has been scheduled for April 10, 2015. It is possible that additional suits will be filed, or allegations made by stockholders, with respect to these same or other matters and also naming us and/or our officers and directors as defendants. We believe that we have meritorious defenses and intend to defend against these lawsuits vigorously.
On April 21, 2014, a stockholder purporting to act on our behalf filed a derivative lawsuit in the Superior Court of California for the County of San Mateo against certain of our officers and directors. The lawsuit alleges breaches of fiduciary duties by the defendants and other violations of law. In general, the lawsuit alleges that the defendants caused or allowed the dissemination of allegedly false and misleading statements related to our Phase 2 trial of imetelstat in patients with ET or PV. The plaintiff is seeking unspecified monetary damages and other relief, including reforms and improvements to our corporate governance and internal procedures. It is possible that additional derivative lawsuits will be filed with respect to these same or other matters and also naming our officers and directors as defendants. We have not yet responded to the derivative lawsuit, but intend to vigorously defend against the claims alleged and to seek dismissal of the lawsuit.
These lawsuits and any other related lawsuits are subject to inherent uncertainties, and the actual defense and disposition costs will depend upon many unknown factors. The outcome of these lawsuits is necessarily uncertain. We could be forced to expend significant resources in the defense against these and any other related lawsuits and we may not prevail. We currently are not able to estimate the possible cost to us from these lawsuits, as they are currently at an early stage, and such amounts could be material to our financial statements even if we prevail in the defense against these lawsuits. We cannot be certain how long it may take to resolve these lawsuits or the possible amount of any damages that we may be required to pay.
ITEM 4. MINE SAFETY DISCLOSURES
Not applicable.
59
ITEM 5. MARKET FOR THE REGISTRANT'S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
Market Information
Our common stock is quoted on the Nasdaq Global Select Market under the symbol GERN. The high and low intraday sales prices as reported by the Nasdaq Global Select Market of our common stock for each of the quarters in the years ended December 31, 2014 and 2013 were as follows:
| |
High | Low | |||||
|---|---|---|---|---|---|---|---|
Year ended December 31, 2014 |
|||||||
First quarter |
$ | 5.92 | $ | 1.39 | |||
Second quarter |
$ | 3.47 | $ | 1.69 | |||
Third quarter |
$ | 3.30 | $ | 1.98 | |||
Fourth quarter |
$ | 3.96 | $ | 1.76 | |||
Year ended December 31, 2013 |
|||||||
First quarter |
$ | 1.78 | $ | 1.05 | |||
Second quarter |
$ | 1.55 | $ | 0.98 | |||
Third quarter |
$ | 3.95 | $ | 1.27 | |||
Fourth quarter |
$ | 7.79 | $ | 2.65 | |||
As of March 6, 2015, there were approximately 654 stockholders of record of our common stock. This number does not include "street name" or beneficial holders, whose shares are held of record by banks, brokers and other financial institutions. We are engaged in a highly dynamic industry, which often results in significant volatility of our common stock price. On March 6, 2015, the closing sales price for our common stock was $3.50 per share.
Dividend Policy
We have never paid cash dividends on our capital stock and do not anticipate paying cash dividends in the foreseeable future, but intend to retain our capital resources for reinvestment in our business. Any future determination to pay cash dividends will be at the discretion of the board of directors and will be dependent upon our financial condition, results of operations, capital requirements and other factors our board of directors deems relevant.
Performance Measurement Comparison(1)
The following graph compares total stockholder returns of Geron Corporation for the last five fiscal years beginning December 31, 2009 to two indices: the Nasdaq CRSP Total Return Index for the Nasdaq Stock Market-U.S. Companies, or the Nasdaq-US, and the Nasdaq Pharmaceutical Index, or the Nasdaq-Pharmaceutical. The total return for our stock and for each index assumes the reinvestment of dividends, although we have never declared cash dividends on Geron stock, and is based on the returns of the component companies weighted according to their capitalizations as of the end of each quarterly period. The Nasdaq-US tracks the aggregate price performance of equity securities of U.S. companies traded on the Nasdaq Global Select Market, or NGSM. The Nasdaq-Pharmaceutical, which is calculated and supplied by Nasdaq, represents pharmaceutical companies, including biotechnology companies, trading on Nasdaq under the Standard Industrial Classification (SIC) Code No. 283 Drugs main category (2833—Medicinals & Botanicals, 2834—Pharmaceutical Preparations, 2835—Diagnostic Substances, 2836—Biological Products). Geron common stock trades on the NGSM and is a component of both the Nasdaq-US and the Nasdaq-Pharmaceutical. The stockholder return shown in the graph below is not necessarily indicative of future performance, and we do not make or endorse any predictions as to future stockholder returns.
60
Comparison of Five Year Cumulative Total Return on Investment Among
Geron Corporation, the Nasdaq-US Index and the Nasdaq-Pharmaceutical Index(2)
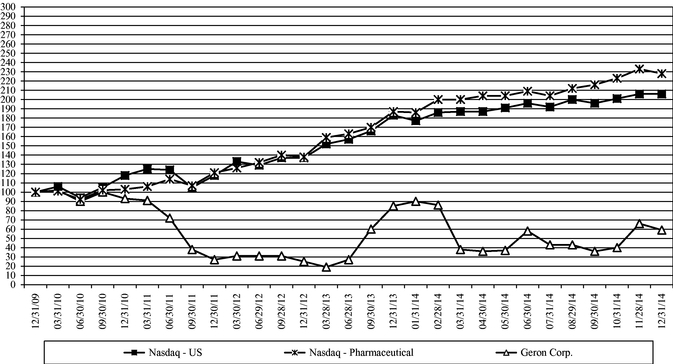
- (1)
- This
Section is not "soliciting material," is not deemed "filed" with the SEC and is not to be incorporated by reference in any filing of Geron Corporation
under the Securities Act, or the Exchange Act, whether made before or after the date hereof and irrespective of any general incorporation language in any such filing.
- (2)
- Shows the cumulative total return on investment assuming an investment of $100 in each of Geron, the Nasdaq-US and the Nasdaq-Pharmaceutical on December 31, 2009. The cumulative total return on Geron stock has been computed based on a price of $5.55 per share, the price at which Geron common stock closed on December 31, 2009.
Recent Sales of Unregistered Securities
In December 2014, we issued an aggregate of 168,039 shares of our common stock pursuant to the net, or cashless, exercise of warrants that were originally issued in connection with a loan agreement with the California Institute for Regenerative Medicine, or CIRM, in November 2011. These warrants were exercisable for an aggregate of 461,382 shares of our common stock and had an exercise price of $2.32 per share.
In issuing the above-mentioned shares, we relied on the exemptions provided by Section 3(a)(9) of the Securities Act of 1933, as amended.
61
ITEM 6. SELECTED FINANCIAL DATA
The following selected consolidated financial data should be read together with our consolidated financial statements and accompanying notes and "Management's Discussion and Analysis of Financial Condition and Results of Operations" appearing elsewhere in this annual report on Form 10-K. The selected consolidated financial data in this section is not intended to replace our consolidated financial statements and the accompanying notes. Our historical results are not necessarily indicative of our future results.
| |
Year Ended December 31, | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
2014 | 2013 | 2012 | 2011 | 2010 | |||||||||||
| |
(In thousands, except share and per share data) |
|||||||||||||||
Consolidated Statements of Operations Data: |
||||||||||||||||
Revenues from collaborative agreements |
$ | — | $ | — | $ | — | $ | 300 | $ | 925 | ||||||
License fees and royalties |
1,153 | 1,283 | 2,709 | 2,138 | 2,638 | |||||||||||
| | | | | | | | | | | | | | | | | |
Total revenues |
1,153 | 1,283 | 2,709 | 2,438 | 3,563 | |||||||||||
Operating expenses: |
||||||||||||||||
Research and development |
20,707 | 23,155 | 51,368 | 69,316 | 61,687 | |||||||||||
Acquired in-process research and development(1) |
— | — | — | — | 35,000 | |||||||||||
Restructuring charges(2) |
— | 1,462 | 2,702 | 5,449 | — | |||||||||||
General and administrative |
16,758 | 15,624 | 20,397 | 23,789 | 18,043 | |||||||||||
| | | | | | | | | | | | | | | | | |
Total operating expenses |
37,465 | 40,241 | 74,467 | 98,554 | 114,730 | |||||||||||
| | | | | | | | | | | | | | | | | |
Loss from operations |
(36,312 | ) | (38,958 | ) | (71,758 | ) | (96,116 | ) | (111,167 | ) | ||||||
Unrealized gain (loss) on derivatives |
351 | (316 | ) | 13 | 643 | 190 | ||||||||||
Interest and other income |
373 | 951 | 3,097 | 1,024 | 2,045 | |||||||||||
Losses recognized under equity method investment |
— | — | — | (503 | ) | (2,347 | ) | |||||||||
Losses recognized from debt extinguishment(3) |
— | — | — | (1,664 | ) | — | ||||||||||
Interest and other expense |
(82 | ) | (56 | ) | (233 | ) | (237 | ) | (98 | ) | ||||||
| | | | | | | | | | | | | | | | | |
Net loss |
$ | (35,670 | ) | $ | (38,379 | ) | $ | (68,881 | ) | $ | (96,853 | ) | $ | (111,377 | ) | |
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | |
Basic and diluted net loss per share: |
||||||||||||||||
Net loss per share |
$ | (0.23 | ) | $ | (0.30 | ) | $ | (0.54 | ) | $ | (0.78 | ) | $ | (1.14 | ) | |
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | |
Shares used in computing net loss per share |
153,540,341 | 128,380,800 | 126,941,024 | 124,506,763 | 97,601,520 | |||||||||||
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | |
- (1)
- In December 2010, we and Angiochem, Inc., or Angiochem, entered into an exclusive license agreement that provided us with a worldwide exclusive license, with the right to grant sublicenses, to Angiochem's proprietary peptide technology that facilitates the transfer of anti-cancer compounds across the blood-brain barrier to be used with tubulin disassembly inhibitors to enable the treatment of primary brain cancers and cancers that have metastasized to the brain. As consideration for the license rights, we paid Angiochem an upfront payment of $7.5 million in cash and issued to Angiochem 5,261,144 shares of common stock on January 5, 2011 as payment of our obligation to issue $27.5 million in stock to Angiochem. Because further clinical and process development of GRN1005 was required before any viable commercial application could be identified or utilized, we concluded that the technology had no alternative future use, and
62
accordingly, expensed the total upfront payment of $35 million as acquired in-process research and development at the time of acquisition in 2010.
- On
December 3, 2012, we provided to Angiochem notice of termination of the exclusive license agreement. We returned the asset to
Angiochem in May 2013 and the license agreement terminated effective June 1, 2013.
- (2)
- In
April 2013, we announced the decision to discontinue our discovery research programs and companion diagnostics program based on telomere length and close
our research laboratory facility located at 200 Constitution Drive, Menlo Park, California. With this decision, a total of 20 positions were eliminated, representing approximately 31% of
our workforce at that time. In connection with this restructuring, we incurred aggregate restructuring charges of approximately $1.4 million in 2013. All actions associated with this
restructuring were completed in 2013, and we do not anticipate incurring any further charges in connection with this restructuring.
- In
December 2012, we announced the decision to discontinue development of GRN1005. With this decision, a total of 43 positions were eliminated,
representing a reduction of approximately 40% of our workforce at that time. In connection with the restructuring, we incurred aggregate restructuring charges of approximately $2.8 million, of
which $2.7 million was recorded in 2012 and $92,000 was recorded in 2013. All actions associated with this restructuring were completed in 2013, and we do not anticipate incurring any further
charges in connection with this restructuring.
- In
November 2011, we discontinued further development of our stem cell programs. With this decision, a total of 66 positions were eliminated,
representing a reduction of approximately 38% of our workforce at that time. In connection with the restructuring, we recorded aggregate restructuring charges of approximately $5.4 million in
2011. All actions associated with this restructuring were completed in 2012, and we do not anticipate incurring any further charges in connection with this restructuring. See Note 6 on
Restructurings in Notes to Consolidated Financial Statements of this annual report on Form 10-K.
- (3)
- In November 2011, we repaid the outstanding principal balance, including accrued interest, or Loan Balance, to CIRM representing our entire Loan Balance under our loan agreement with CIRM. In addition, we relinquished our right to future disbursements from CIRM under the loan agreement and gave notice of termination. With the repayment of the entire outstanding Loan Balance, we have no further amounts owed to CIRM. In connection with the early termination of the loan agreement with CIRM, we recognized a debt extinguishment charge of $1.7 million for the unamortized debt discount associated with the loan.
| |
December 31, | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (In thousands) |
2014 | 2013 | 2012 | 2011 | 2010 | |||||||||||
Consolidated Balance Sheets Data: |
||||||||||||||||
Cash, restricted cash, cash equivalents and marketable securities |
$ | 170,639 | $ | 66,019 | $ | 96,329 | $ | 154,239 | $ | 221,274 | ||||||
Working capital |
111,607 | 59,470 | 84,269 | 112,181 | 154,168 | |||||||||||
Total assets |
172,511 | 67,344 | 99,801 | 160,047 | 233,584 | |||||||||||
Accumulated deficit |
(928,433 | ) | (892,763 | ) | (854,384 | ) | (785,503 | ) | (688,650 | ) | ||||||
Total stockholders' equity |
130,712 | 59,757 | 85,653 | 146,603 | 192,735 | |||||||||||
63
ITEM 7. MANAGEMENT'S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
Overview
The following discussion should be read in conjunction with the audited consolidated financial statements and notes thereto included in Part II, Item 8 of this annual report on Form 10-K.
We are a clinical stage biopharmaceutical company focused on the development of a telomerase inhibitor, imetelstat, in hematologic myeloid malignancies. The discovery and early development of imetelstat, our sole product candidate, was based on our core expertise in telomerase and telomere biology. Telomerase is an enzyme that enables cancer cells, including malignant progenitor cells, to maintain telomere length, which provides them with the capacity for limitless, uncontrolled proliferation. Using our proprietary nucleic acid chemistry, we designed imetelstat to be an oligonucleotide that targets and binds with high affinity to the active site of telomerase, thereby directly inhibiting telomerase activity and impeding malignant cell proliferation. Molecular responses in essential thrombocythemia, or ET, and remission responses, including reversal of bone marrow fibrosis, in myelofibrosis, or MF, suggest imetelstat has disease-modifying activity by inhibiting the progenitor cells of the malignant clone for the underlying disease in a relatively selective manner.
On November 13, 2014, we entered into an exclusive collaboration and license agreement, or the Collaboration Agreement, with Janssen Biotech, Inc., or Janssen, to develop and commercialize imetelstat worldwide for all indications in oncology, including hematologic myeloid malignancies, and all other human therapeutic uses. The Collaboration Agreement became effective on December 15, 2014 and we received $35 million from Janssen as an upfront payment, which has been recorded as deferred revenue on our consolidated balance sheet as of December 31, 2014. Additional consideration under the Collaboration Agreement includes payments up to a potential total of $900 million for the achievement of development, regulatory and commercial milestones, as well as royalties on worldwide net sales.
Under the Collaboration Agreement, Janssen is responsible for the development, manufacturing and commercialization of, and seeking regulatory approval for imetelstat worldwide. Development of imetelstat will proceed under a mutually agreed clinical development plan, which includes two Phase 2 studies to be pursued initially, one in myelofibrosis, referred to as the Initial Phase 2 MF Study, and one in myelodysplastic syndrome, referred to as the Initial Phase 2 MDS Study. We expect Janssen to initiate the Initial Phase 2 MF Study in mid-2015, followed by the Initial Phase 2 MDS Study to be initiated at the end of 2015. In addition, the clinical development plan may also include additional, possible registration studies in MF and myelodysplastic syndrome, or MDS, and possible exploratory Phase 2 and potential follow-on Phase 3 studies in acute myelogenous leukemia, or AML. For a further discussion regarding the Collaboration Agreement, see Note 10 on License Agreements in Notes to Consolidated Financial Statements of this annual report on Form 10-K.
We believe our current operational and financial resources, including the upfront payment received from Janssen under the Collaboration Agreement, may enable us to acquire other oncology products, programs or companies to diversify our business.
We have incurred operating losses every year since our operations began in 1990. Losses have resulted principally from costs incurred in connection with our research and development activities and from general and administrative costs associated with our operations. Substantially all of our revenues to date have been research support payments under collaborative agreements, and milestones, royalties and other revenues from our licensing arrangements. We currently have no source of product revenue. Our revenues for 2015 are expected to primarily consist of revenue from the upfront payment from Janssen upon the completion of technology transfer-related activities in 2015, and future revenues are substantially dependent on Janssen successfully developing and commercializing imetelstat in
64
accordance with the Collaboration Agreement. Since inception, we have primarily financed our operations through the sale of equity securities, interest income on our marketable securities and payments we received under our collaborative and licensing arrangements.
For the years ended December 31, 2014, 2013 and 2012, we incurred net losses of $35.7 million, or $0.23 per share, $38.4 million, or $0.30 per share, and $68.9 million, or $0.54 per share, respectively. As of December 31, 2014, we had an accumulated deficit of $928.4 million. The significance of future losses will depend on whether Janssen continues to develop and advance imetelstat and the clinical and commercial success of imetelstat, which would result in future revenues to us in the form of milestone payments and royalties under the Collaboration Agreement as described above, and whether we acquire other oncology products, programs or companies to diversify our business. There can be no assurance that we will receive any milestone payments or royalties from Janssen in the future, or at all. Imetelstat, which is our sole product candidate, will require significant additional clinical testing prior to possible regulatory approval in the United States and other countries, and we do not expect imetelstat to be commercially available for many years, if at all.
As of December 31, 2014, we had cash, restricted cash, cash equivalents and marketable securities of $170.6 million compared to $66.0 million at December 31, 2013 and $96.3 million at December 31, 2012. The increase in cash, restricted cash, cash equivalents and marketable securities in 2014 was primarily the result of the receipt of net cash proceeds of approximately $96.8 million, after deducting the underwriting discount and offering expenses payable by us, from an underwritten public offering of 25,875,000 shares of our common stock at a public offering price of $4.00 per share that we completed in February 2014 and the receipt of $35.0 million from Janssen as an upfront payment in December 2014 upon the effectiveness of the Collaboration Agreement. We estimate that our existing capital resources and future interest income will be sufficient to fund our current level of operations through at least the next 12 months. However, we may use our available capital resources sooner than we anticipate.
Critical Accounting Policies and Estimates
Our consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America. The preparation of these consolidated financial statements requires management to make estimates and assumptions that affect the reported amounts of assets, liabilities, revenues and expenses. Note 1 of Notes to Consolidated Financial Statements describes the significant accounting policies used in the preparation of the consolidated financial statements. Certain of these significant accounting policies are considered to be critical accounting policies, as defined below.
A critical accounting policy is defined as one that is both material to the presentation of our consolidated financial statements and requires management to make difficult, subjective or complex judgments that could have a material effect on our financial condition and results of operations. Specifically, critical accounting estimates have the following attributes: (i) we are required to make assumptions about matters that are highly uncertain at the time of the estimate; and (ii) different estimates we could reasonably have used, or changes in the estimate that are reasonably likely to occur, would have a material effect on our financial condition or results of operations.
Estimates and assumptions about future events and their effects cannot be determined with certainty. We base our estimates on historical experience and on various other assumptions believed to be applicable and reasonable under the circumstances. These estimates may change as new events occur, as additional information is obtained and as our operating environment changes. These changes have historically been minor and have been included in the consolidated financial statements as soon as they became known. Based on a critical assessment of our accounting policies and the underlying judgments and uncertainties affecting the application of those policies, management believes that our
65
consolidated financial statements are fairly stated in accordance with accounting principles generally accepted in the United States, and meaningfully present our financial condition and results of operations.
We believe the following critical accounting policies reflect our more significant estimates and assumptions used in the preparation of our consolidated financial statements:
Fair Value of Financial Instruments
We categorize financial instruments recorded at fair value on our consolidated balance sheet based upon the level of judgment associated with inputs used to measure their fair value. The categories are as follows:
Level 1—Inputs are unadjusted, quoted prices in active markets for identical assets or liabilities at the measurement date. An active market for the asset or liability is a market in which transactions for the asset or liability occur with sufficient frequency and volume to provide pricing information on an ongoing basis.
Level 2—Inputs (other than quoted market prices included in Level 1) are either directly or indirectly observable for the asset or liability through correlation with market data at the measurement date and for the duration of the instrument's anticipated life.
Level 3—Inputs reflect management's best estimate of what market participants would use in pricing the asset or liability at the measurement date. Consideration is given to the risk inherent in the valuation technique and the risk inherent in the inputs to the model.
A financial instrument's categorization within the valuation hierarchy is based upon the lowest level of input that is significant to the fair value measurement. Following is a description of the valuation methodologies used for instruments measured at fair value on our consolidated balance sheet, including the category for such instruments.
Instruments classified as Level 1 include money market funds, representing 24% of our total financial instruments measured at fair value classified as assets as of December 31, 2014. Instruments classified as Level 2 include U.S. government-sponsored enterprise securities, commercial paper and corporate notes, representing 76% of our total financial instruments measured at fair value classified as assets as of December 31, 2014. The price for each security at the measurement date is derived from various sources. Periodically, we assess the reasonableness of these sourced prices by comparing them to the prices provided by our portfolio managers from broker quotes as well as reviewing the pricing methodologies used by our portfolio managers. Historically, we have not experienced significant deviation between the sourced prices and our portfolio manager's prices.
Warrants to purchase common stock and non-employee options are normally traded less actively, have trade activity that is one way, and/or are traded in less-developed markets and are therefore valued based upon models with significant unobservable market parameters, resulting in Level 3 categorization. Instruments classified as Level 3 include derivative liabilities from non-employee options, representing all of our financial instruments measured at fair value classified as liabilities as of December 31, 2014. The fair value for these instruments is calculated using the Black Scholes option-pricing model. The model's inputs reflect assumptions that market participants would use in pricing the instrument in a current period transaction. Use of this model requires us to make assumptions regarding stock volatility, dividend yields, expected term of the non-employee options and risk-free interest rates. Changes to the model's inputs are not changes to valuation methodologies, but instead reflect direct or indirect impacts from changes in market conditions. Accordingly, results from the valuation model in one period may not be indicative of future period measurements. Expected volatilities are based on historical volatilities of our stock. The expected term of non-employee options represents the remaining contractual term of the instruments. The risk-free interest rate is based on the
66
U.S. Zero Coupon Treasury Strip Yields for the remaining term of the instrument. If factors change and we employ different assumptions in future periods, the fair value of these non-employee options reflected as of each balance sheet date and the resulting change in fair value that we record may differ significantly from what we have recorded in previous periods. As of December 31, 2014, we have not revised the method in which we derive assumptions in order to estimate fair values of non-employee options classified as liabilities, and we do not expect revisions in the future.
For a further discussion regarding fair value measurements, see Note 2 on Fair Value Measurements in Notes to Consolidated Financial Statements of this annual report on Form 10-K.
Revenue Recognition
In general, we recognize revenue for each unit of accounting when all of the following criteria have been met: (a) persuasive evidence of an arrangement exists, (b) delivery has occurred or services have been rendered, (c) the seller's price to the buyer is fixed or determinable, and (d) collectability is reasonably assured. Amounts received prior to satisfying these revenue recognition criteria are recorded as deferred revenue. Amounts expected to be recognized as revenue within the 12 months following the balance sheet date are classified as current deferred revenue. Amounts not expected to be recognized as revenue within the 12 months following the balance sheet date are classified as noncurrent deferred revenue.
Since our inception, substantially all of our revenues have been generated from collaboration agreements and licensing arrangements. Economic terms in these agreements may include non-refundable license payments in cash or equity securities, option payments in cash or equity securities, cost reimbursements, cost-sharing arrangements, milestone payments, royalties on future sales of products, or any combination of these items. In applying the appropriate revenue recognition guidance related to these agreements, we first assess whether the arrangement contains multiple elements. In this evaluation, we consider: (1) the deliverables included in the arrangement and (2) whether the individual deliverables represent separate units of accounting or whether they must be accounted for as a combined unit of accounting. This evaluation involves subjective determinations and requires us to make judgments about the individual deliverables and whether such deliverables are separable from the other aspects of the contractual relationship. Deliverables are considered separate units of accounting provided that: (i) the delivered item(s) has value to the customer on a standalone basis, and if (ii) the arrangement includes a general right of return relative to the delivered item(s), delivery or performance of the undelivered item(s) is considered probable and substantially in our control. In assessing whether an item has standalone value, we consider factors such as the research, manufacturing and commercialization capabilities of the collaboration partner and the availability of the associated expertise in the general marketplace. In addition, we consider whether the collaboration partner can use the other deliverable(s) for their intended purpose without the receipt of the remaining element(s), whether the value of the deliverable is dependent on the undelivered item(s) and whether there are other vendors that can provide the undelivered element(s).
Arrangement consideration that is fixed or determinable is allocated among the separate units of accounting using the relative selling price method. We then apply the applicable revenue recognition criteria noted above to each of the separate units of accounting in determining the appropriate period and pattern of recognition. We determine how to allocate arrangement consideration to identified units of accounting based on the selling price hierarchy provided under relevant accounting guidance. The estimated fair value of deliverables under the arrangement may be derived using a best estimate of selling price if vendor-specific-objective evidence and third-party evidence are not available.
67
Upfront non-refundable signing, license or non-exclusive option fees are recognized as revenue: (i) when rights to use the intellectual property, related to a license that has standalone value from the other deliverables to be provided under the agreement, have been delivered or (ii) over the term of the agreement if we have continuing performance obligations as the arrangement would be accounted for as a single unit of accounting. When payments are received in equity securities, we do not recognize any revenue unless such securities are determined to be realizable in cash.
At the inception of an arrangement that includes milestone payments, we assess whether each milestone is substantive and at risk to both parties on the basis of the contingent nature of the milestone. This evaluation includes an assessment of whether: (i) the consideration is commensurate with either the performance to achieve the milestone or the enhancement of the value of the delivered item(s) as a result of a specific outcome resulting from the performance to achieve the milestone, (ii) the consideration relates solely to past performance and (iii) the consideration is reasonable relative to all of the deliverables and payment terms within the arrangement. We consider various factors such as the scientific, clinical, regulatory, commercial and other risks that must be overcome to achieve the respective milestone and the level of effort and investment required to achieve the respective milestone in making this assessment. There is considerable judgment involved in determining whether a milestone satisfies all of the criteria required to conclude that a milestone is substantive. Milestone payments for milestones that are considered substantive would be recognized as revenue in their entirety upon successful accomplishment of the milestone, assuming all other revenue recognition criteria are met. Milestone payments for milestones that are not considered substantive would be recognized as revenue over the remaining period of performance, assuming all other revenue recognition criteria are met.
Royalties are recognized as earned in accordance with contract terms when royalties from licensees can be reasonably estimated and collectability is reasonably assured. If royalties cannot be reasonably estimated or collectability of a royalty amount is not reasonably assured, royalties are recognized as revenue when the cash is received. Revenue from commercial milestone payments will be accounted for as royalties and recorded as revenue upon achievement of the milestone, assuming all other revenue recognition criteria are met.
Cost-sharing expenses are recorded as earned or owed based on the performance requirements by both parties under the respective contracts. For arrangements in which we and our collaboration partner in the agreement are exposed to significant risks and rewards depending on the commercial success of the activity, we recognize payments between the parties on a net basis and record such amounts as a reduction or addition to research and development expense. For arrangements in which we have agreed to perform certain research and development services for our collaboration partner and are not exposed to significant risks and rewards depending on the commercial success of the activity, we recognize the respective cost reimbursements as revenue under the collaborative agreement as the related research and development services are rendered.
Revenue recognition for licenses and collaboration agreements requires significant judgment. We estimate the projected future term of license agreements over which we recognize revenue. We evaluate the deliverables under an arrangement and estimate the fair value of those deliverables. We also assess the substantive nature of milestones. Our assessments and estimates are based on contractual terms, historical experience and general industry practice. Revisions in these values or estimations have the effect of increasing or decreasing license fee revenue in the period of revision. As of December 31, 2014, we have not made any revisions to revenue recognition estimates and we do not expect revisions to currently active agreements in the future.
Clinical Trial Accruals
Substantial portions of our preclinical studies and all of our clinical trials have been performed by third-party contract research organizations, or CROs, and other vendors. We accrue expenses for
68
preclinical studies performed by our vendors based on certain estimates over the term of the service period and adjust our estimates as required. We accrue expenses for clinical trial activities performed by CROs based upon the estimated amount of work completed on each study. For clinical trial expenses, the significant factors used in estimating accruals include the number of patients enrolled, the number of active clinical sites, and the duration for which the patients will be enrolled in the study. Pass through costs from CROs include, but are not limited to, regulatory expenses, investigator fees, lab fees, travel costs and other miscellaneous costs, including shipping and printing fees. We accrue pass through costs based on estimates of the amount of work completed for the clinical trial. We monitor patient enrollment levels and related activities to the extent possible through internal reviews, review of contractual terms and correspondence with CROs. We base our estimates on the best information available at the time. However, additional information may become available to us which will allow us to make a more accurate estimate in future periods. In this event, we may be required to record adjustments to research and development expenses in future periods when the actual level of activity becomes more certain.
Valuation of Stock-Based Compensation
We measure and recognize compensation expense for all stock-based awards to our employees and directors, including stock options, restricted stock awards and employee stock purchases related to our Employee Stock Purchase Plan, or ESPP, based on estimated fair values. We use the Black Scholes option-pricing model to estimate the grant-date fair value of our stock options and employee stock purchases. Option-pricing valuation model assumptions such as expected volatility, risk-free interest rate and expected term impact the fair value estimate.
Further, the estimated forfeiture rate impacts the amount of aggregate stock-based compensation expense recognized during the period. The fair value of stock options and employee stock purchases is amortized over the vesting period of the awards using a straight-line method.
Expected volatilities are based on historical volatilities of our stock since traded options on Geron common stock do not correspond to option terms and trading volume of options is limited. The expected term of options represents the period of time that options granted are expected to be outstanding. In deriving this assumption, we reviewed actual historical exercise and cancellation data and the remaining outstanding options not yet exercised or cancelled. The expected term of employees' purchase rights under our ESPP is equal to the purchase period. The risk-free interest rate is based on the U.S. Zero Coupon Treasury Strip Yields for the expected term in effect on the date of grant. Forfeiture rates are estimated based on historical data and are adjusted, if necessary, over the requisite service period based on the extent to which actual forfeitures differ, or are expected to differ, from their estimate.
We have granted restricted stock awards to employees and directors with three types of vesting schedules: (i) service-based, (ii) performance-based or (iii) market-based. Service-based restricted stock awards generally vest annually over four years. Performance-based restricted stock awards vest upon achievement of discrete strategic corporate goals within a specified performance period, generally three years. Market-based restricted stock awards vest upon achievement of certain market price thresholds of our common stock within a specified performance period, generally three years.
The fair value for service-based restricted stock awards is determined using the fair value of our common stock on the date of grant. The fair value is amortized as stock-based compensation expense over the requisite service period of the award, which is generally the vesting period, on a straight-line basis and is reduced for estimated forfeitures, as applicable.
The fair value for performance-based restricted stock awards is determined using the fair value of our common stock on the date of grant. Stock-based compensation expense for awards with vesting based on performance conditions is recognized over the period from the date the performance
69
condition is determined to be probable of occurring through the date the applicable condition is expected to be met and is reduced for estimated forfeitures, as applicable. If the performance condition is not considered probable of being achieved, no stock-based compensation expense is recognized until such time as the performance condition is considered probable of being met, if at all. If that assessment of the probability of the performance condition being met changes, the impact of the change in estimate would be recognized in the period of the change. If the requisite service period has been met prior to the change in estimate, the effect of the change in estimate would be immediately recognized. We evaluate whether performance conditions are probable of occurring, as well as the expected performance period, on a quarterly basis.
The fair value for market-based restricted stock awards is determined using a lattice valuation model with a Monte Carlo simulation. The model takes into consideration the historical volatility of our stock and the risk-free interest rate at the date of grant. In addition, the model is used to estimate the derived service period for the awards. The derived service period is the estimated period of time that would be required to satisfy the market condition, assuming the market condition will be satisfied. Stock-based compensation expense is recognized over the derived service period for the awards using the straight-line method and is reduced for estimated forfeitures, as applicable, but is accelerated if the market condition is achieved earlier than estimated. If a market-based restricted stock award is forfeited or expires after completion of the derived service period, any previously recognized stock-based compensation expense is not reversed.
We evaluate the assumptions used in estimating fair values of our stock-based awards by reviewing current trends in comparison to historical data on an annual basis. We have not revised the methods by which we derive assumptions in order to estimate fair values of our stock-based awards. If factors change and we employ different assumptions in future periods, the stock-based compensation expense that we record for awards to employees and directors may differ significantly from what we have recorded in the current period.
Compensation expense recognized for stock-based awards to employees and directors was $7.7 million, $4.4 million and $5.3 million for the years ended December 31, 2014, 2013 and 2012, respectively. As of December 31, 2014, total compensation cost related to unvested stock-based awards not yet recognized, net of estimated forfeitures, was $15.0 million, which is expected to be recognized over the next 27 months on a weighted-average basis.
For our non-employee stock-based awards, the measurement date on which the fair value of the stock-based award is calculated is equal to the earlier of (i) the date at which a commitment for performance by the counterparty to earn the equity instrument is reached or (ii) the date at which the counterparty's performance is complete. We recognized stock-based compensation expense of $94,000, $92,000 and $135,000 for the fair value of the vested portion of non-employee options and restricted stock awards in our consolidated statements of operations for the years ended December 31, 2014, 2013 and 2012, respectively.
Results of Operations
Our results of operations have fluctuated from period to period and may continue to fluctuate in the future, based upon the progress of research and development efforts in collaboration with Janssen and whether we acquire other oncology products, programs or companies to diversify our business. Results of operations for any period may be unrelated to results of operations for any other period. In addition, historical results should not be viewed as indicative of future operating results. We are subject to risks common to companies in our industry and at our stage of development, including, but not limited to, risks inherent in research and development efforts, our dependence on Janssen for the development, regulatory approval, manufacture and commercialization of our sole product candidate, imetelstat, uncertainty of preclinical and clinical trial results or regulatory approvals or clearances, need
70
for future capital, enforcement of our patent and proprietary rights, reliance upon our collaborators, investigators and other third parties, and potential competition. In order for imetelstat to be commercialized based on our research, we and Janssen must conduct preclinical tests and clinical trials, demonstrate the safety and efficacy of imetelstat, obtain regulatory approvals or clearances and enter into manufacturing, distribution and marketing arrangements, as well as obtain market acceptance. We do not expect to receive royalties based on sales of imetelstat for many years, if at all.
Revenues
In addition to the Collaboration Agreement with Janssen, we have entered into several license or collaboration agreements with companies involved with oncology, diagnostics, research tools and biologics production. In each of these agreements, we have granted certain rights to our technologies. In connection with the agreements, we are eligible to receive license fees, option fees, milestone payments and royalties on future sales of products, or any combination thereof. We recognized license fee revenues of $738,000, $933,000 and $1.6 million in 2014, 2013 and 2012, respectively, related to our various agreements. We have not recognized any revenue related to the Collaboration Agreement in 2014 since we have determined that the sole non-contingent deliverable under the Collaboration Agreement is our delivery of the license rights to Janssen and our complete performance of the technology transfer-related activities. We currently expect completion of the technology transfer-related activities to occur by September 30, 2015, at which point we expect to fully recognize the $35.0 million upfront payment from Janssen as license fee revenue. The decrease in license fee revenues in 2014 compared to 2013 primarily reflects the full recognition of a non-refundable up-front license payment in 2013 for an exclusive commercial license using our telomerase promoter technology for oncology-related in vitro assays. The decrease in license fee revenues in 2013 compared to 2012 primarily reflects the full recognition of a license payment from GE Healthcare in 2012 upon the exercise of an option to expand the scope of their original license agreement with us to obtain exclusive global rights to intellectual property and know-how for the development and sale of cellular assays derived from induced pluripotent stem cells. In connection with the closing of the divestiture of our human embryonic stem cell assets in October 2013, our license agreement with GE Healthcare, including any future revenue payments thereunder, was transferred to Asterias.
We recognized royalty revenues of $415,000, $350,000 and $1.1 million in 2014, 2013 and 2012, respectively, on product sales of telomerase detection and telomere measurement kits to the research-use-only market, cell-based research products and nutritional products. The increase in royalty revenues in 2014 compared to 2013 primarily reflects the receipt of a milestone fee in 2014 in connection with the achievement of a net sales milestone by a licensee of our hTERT technology. The decrease in royalty revenues in 2013 compared to 2012 primarily reflects the assignment of our telomerase activation technology to Telomerase Activation Sciences, Inc., or TA Sciences, in December 2012 and termination of our license agreement with Asia Biotech Corporation. See further discussion of the agreement with TA Sciences under the sub-section entitled "Interest and Other Income".
We expect our revenues for 2015 to primarily consist of license fee revenue from the upfront payment from Janssen under the Collaboration Agreement upon our completion of the technology transfer-related activities in 2015, and future license fee and royalty revenues are substantially dependent on Janssen successfully developing and commercializing imetelstat in accordance with the Collaboration Agreement. See further discussion of revenue recognition for the Janssen collaboration in Note 10 on License Agreements in Notes to Consolidated Financial Statements of this annual report on Form 10-K. Future license fee and royalty revenues are also dependent on additional agreements being signed and current agreements being maintained. Current revenues may not be predictive of future revenues.
71
Research and Development Expenses
For each of our research and development programs, we incur direct external, personnel related and other research and development costs. Direct external expenses primarily consist of costs to outside parties to perform laboratory studies, develop manufacturing processes and manufacture raw materials and clinical trial drug materials, conduct and manage clinical trials, including investigator-sponsored clinical trials, and provide advice and consultation for scientific and clinical strategies. Personnel related expenses primarily consist of salaries and wages, stock-based compensation, payroll taxes and benefits for those individuals involved with ongoing research and development efforts. Other research and development expenses primarily consist of laboratory supplies, research-related overhead associated with leasing, operating and maintaining our facilities and equipment depreciation and maintenance. All of these costs apply to our current and historical clinical programs and our historical preclinical programs and discovery research efforts. A product candidate is designated a clinical candidate once an investigational new drug application has been filed with the FDA, or a similar filing with regulatory agencies outside the United States, for the purpose of commencing clinical trials in humans. Preclinical programs include product candidates undergoing toxicology, pharmacology, metabolism and efficacy studies and manufacturing process development required before testing in humans can commence.
Research and development expenses were $20.7 million, $23.2 million and $51.4 million for the years ended December 31, 2014, 2013 and 2012, respectively. As shown in the table below, the decrease in research and development expenses in 2014 compared to 2013 and 2013 compared to 2012 primarily reflects the net result of lower direct external costs due to the wind-down of our GRN1005 trials in patients with brain metastases and imetelstat trials in solid tumors, reduced personnel related costs resulting from previous restructurings and lower costs for scientific supplies and services and other research-related overhead costs due to the discontinuation of our discovery research programs in April 2013. The decrease in research and development expenses in 2014 compared to 2013 was partially offset by an increase in direct external costs for the manufacturing of imetelstat drug product. Overall, in 2015 we expect direct external research and development expenses to increase as we collaborate with Janssen on the development of imetelstat in hematologic myeloid malignancies and personnel related research and development expenses to decrease as a result of the recently announced organizational resizing.
Research and development expenses for the years ended December 31, 2014, 2013 and 2012 were as follows:
| |
Year Ended December 31, | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| (In thousands) |
2014 | 2013 | 2012 | |||||||
Direct external research and development expenses: |
||||||||||
Clinical program: Imetelstat |
$ | 8,901 | $ | 7,665 | $ | 12,907 | ||||
Clinical program: GRN1005(1) |
— | 1,039 | 10,723 | |||||||
Clinical program: GRNOPC1(2) |
— | 202 | 393 | |||||||
Preclinical programs(3) |
— | 228 | 1,155 | |||||||
Personnel related expenses |
9,674 | 10,753 | 19,008 | |||||||
All other research and development expenses |
2,132 | 3,268 | 7,182 | |||||||
| | | | | | | | | | | |
Total |
$ | 20,707 | $ | 23,155 | $ | 51,368 | ||||
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
- (1)
- In
December 2012, we discontinued the GRN1005 program and returned the asset to Angiochem in May 2013.
- (2)
- In October 2013, we closed the transaction to divest our human embryonic stem cell assets and our autologous cellular immunotherapy program to Asterias. Asterias assumed all post-closing liabilities with respect to all of the assets contributed by us, including any liabilities related to the
72
GRNOPC1 and autologous cellular immunotherapy clinical trials. See Note 7 on Divestiture of Stem Cell Assets in Notes to Consolidated Financial Statements of this annual report on Form 10-K for further discussion.
- (3)
- In April 2013, we announced the decision to discontinue our discovery research programs and companion diagnostics program based on telomere length.
At this time, we cannot provide reliable estimates of how much time or investment will be necessary to commercialize imetelstat. For a more complete discussion of the risks and uncertainties associated with the development of imetelstat, see the sub-sections entitled "Risks Related to Our Business" and "Risks Related to Clinical and Commercialization Activities" in Part I, Item 1A entitled "Risk Factors" and elsewhere in this annual report on Form 10-K.
Restructuring Charges
In April 2013, we announced the decision to discontinue our discovery research programs and companion diagnostics program based on telomere length and close our research laboratory facility located at 200 Constitution Drive, Menlo Park, California. With this decision, a total of 20 positions were eliminated. In connection with this restructuring, we incurred aggregate restructuring charges of approximately $1.4 million in 2013, of which $624,000 related to one-time termination benefits, including $28,000 of non-cash stock-based compensation expense relating to the extension of the post-termination exercise period through the end of December 2013 for certain stock options previously granted to terminated employees, $200,000 related to non-cash charges for write-downs of excess equipment and leasehold improvements and $546,000 related to costs associated with the closure of our research laboratory facility. In connection with the decision to close our research laboratory facility, we entered into an amendment to the lease agreement for the 200 Constitution Drive facility under which the lease terminated effective December 31, 2013. As consideration for the early termination of the lease, we paid the landlord the remaining rents due under the original term of the lease as well as certain facility maintenance costs, all of which have been included in restructuring charges. In 2013, we received proceeds of approximately $1.1 million from the sale of excess laboratory equipment in connection with the closure of our research laboratory facility. All actions associated with this restructuring were completed in 2013, and we do not anticipate incurring any further charges in connection with this restructuring.
In December 2012, we announced the decision to discontinue development of GRN1005. With this decision, a total of 43 positions were eliminated. In connection with this restructuring, we incurred aggregate restructuring charges of approximately $2.8 million, of which $2.7 million was recorded in 2012 and $92,000 was recorded in 2013. The aggregate restructuring charges consisted of $2.5 million related to one-time termination benefits, including $107,000 of non-cash stock-based compensation expense relating to the extension of the post-termination exercise period through the end of December 2013 for certain stock options previously granted to terminated employees, and $271,000 related to write-downs of GRN1005 manufacturing equipment. All actions associated with this restructuring were completed in 2013, and we do not anticipate incurring any further charges in connection with this restructuring. See Note 6 on Restructurings in Notes to Consolidated Financial Statements of this annual report on Form 10-K for further discussion of the restructuring charges.
General and Administrative Expenses
General and administrative expenses were $16.8 million, $15.6 million and $20.4 million for the years ended December 31, 2014, 2013 and 2012, respectively. The increase in general and administrative expenses in 2014 compared to 2013 primarily reflects the net result of higher non-cash stock-based compensation expense, increased legal fees associated with the purported securities lawsuits and the derivative lawsuit filed against us and/or certain of our officers and directors and transaction
73
costs associated with the Collaboration Agreement we entered into with Janssen in November 2014, partially offset by reduced patent costs and transaction fees associated with the stem cell divestiture which closed in October 2013. The decrease in general and administrative expenses in 2013 compared to 2012 primarily reflects lower personnel related expenses, primarily resulting from previous restructurings, and reduced legal and consulting fees associated with our intellectual property portfolio and our stem cell divestiture efforts. We expect general and administrative expenses to increase in 2015 as a result of higher anticipated legal fees as we intend to vigorously defend against the lawsuits filed against us.
Unrealized Gain (Loss) on Derivatives
Unrealized gain (loss) on derivatives reflects a non-cash adjustment for changes in fair value of options held by non-employees that are classified as current liabilities. Derivatives classified as liabilities are marked to fair value at each financial reporting date with any resulting unrealized gain (loss) recorded in the consolidated statements of operations. The derivatives continue to be reported as a liability until such time as the instruments are exercised or expire or are otherwise modified to remove the provisions which require them to be recorded as liabilities, at which time these instruments are marked to fair value and reclassified from liabilities to stockholders' equity. We incurred unrealized gains on derivatives of $351,000 and $13,000 for the years ended December 31, 2014 and 2012, respectively, compared to unrealized losses on derivatives of $316,000 for the year ended December 31, 2013. The unrealized gains and losses on derivatives primarily reflect the change in fair values of derivative liabilities as a result of fluctuations in the market value of our stock and changes in other inputs factored into the estimate of their fair value such as the volatility of our stock. See Note 2 on Fair Value Measurements in Notes to Consolidated Financial Statements of this annual report on Form 10-K for further discussion of the fair value of derivatives.
Interest and Other Income
Interest income was $373,000, $219,000 and $597,000 for the years ended December 31, 2014, 2013 and 2012, respectively. The increase in interest income in 2014 compared to 2013 primarily reflects an increase in our cash and investment balances in connection with the receipt of the net cash proceeds from the underwritten public offering of shares of our common stock that we completed in February 2014. The decrease in interest income in 2013 compared to 2012 primarily reflects lower cash and investment balances resulting from the use of cash for operations. Interest earned in future periods will depend on the size of our securities portfolio and prevailing interest rates.
Other income was $732,000 and $2.5 million for the years ended December 31, 2013 and 2012, respectively. No other income was recognized for the year ended December 31, 2014. Other income recognized in 2013 reflects a net gain on the sale of excess laboratory equipment in connection with the closure of our research laboratory facility. Other income recognized in 2012 reflects the receipt a non-refundable upfront payment of $2.5 million for the assignment of our telomerase activation technology to TA Sciences, pursuant to the Termination and Assignment Agreement that we entered into with Asia Biotech Corporation, or Asia Biotech, and TA Sciences in December 2012. See Note 10 on License Agreements in Notes to Consolidated Financial Statements of this annual report on Form 10-K for further discussion of the Termination and Assignment Agreement with Asia Biotech and TA Sciences.
Interest and Other Expense
Interest and other expense was $82,000, $56,000 and $233,000 for the years ended December 31, 2014, 2013 and 2012, respectively. The increase in interest and other expense in 2014 compared to 2013 primarily reflects higher bank charges related to the increase in our cash and investment balances in connection with the receipt of the net cash proceeds from the underwritten public offering of shares of our common stock that we completed in February 2014. The decrease in interest and other expense in 2013 compared to 2012 primarily reflects the recognition of accumulated foreign currency translation adjustments in connection with the dissolution of Geron Bio-Med Ltd. in August 2012 and reduced bank charges as a result of lower cash and investment balances in 2013.
74
Net Loss
Net loss was $35.7 million, $38.4 million and $68.9 million for the years ended December 31, 2014, 2013 and 2012, respectively. The decrease in net loss in 2014 compared to 2013 and 2013 compared to 2012 primarily reflects lower clinical trial costs as a result of the wind-down of our GRN1005 trials in patients with brain metastases and imetelstat trials in solid tumors, decreased personnel related costs resulting from previous restructurings, reduced costs for scientific supplies and services and other research-related overhead costs with the discontinuation of our discovery research programs in April 2013 and lower patent costs and transaction fees associated with the stem cell divestiture which closed in October 2013. The decrease in net loss in 2014 compared to 2013 was partially offset by increased costs for the manufacturing of imetelstat drug product, higher non-cash stock-based compensation expense and increased legal fees associated with the purported securities lawsuits and transaction costs for the Collaboration Agreement we entered into with Janssen in November 2014.
Liquidity and Capital Resources
Cash, restricted cash, cash equivalents and marketable securities at December 31, 2014 were $170.6 million, compared to $66.0 million at December 31, 2013 and $96.3 million at December 31, 2012. We estimate that our existing capital resources and future interest income will be sufficient to fund our current level of operations through at least the next 12 months. We have an investment policy to invest these funds in liquid, investment grade securities, such as interest-bearing money market funds, certificates of deposit, municipal securities, U.S. government and agency securities, corporate notes and commercial paper. Our investment portfolio does not contain securities with exposure to sub-prime mortgages, collateralized debt obligations, asset-backed securities or auction rate securities and, to date, we have not recognized any other-than-temporary impairment on our marketable securities or any significant changes in aggregate fair value that would impact our cash resources or liquidity. To date, we have not experienced lack of access to our invested cash and cash equivalents; however, access to our invested cash and cash equivalents may be impacted by adverse conditions in the financial and credit markets. The increase in cash, restricted cash, cash equivalents and marketable securities in 2014 was primarily the result of the receipt of net cash proceeds of approximately $96.8 million, after deducting the underwriting discount and offering expenses payable by us, from an underwritten public offering of 25,875,000 shares of our common stock at a public offering price of $4.00 per share that we completed in February 2014 and the receipt of $35.0 million from Janssen as an upfront payment in December 2014 upon the effectiveness of the Collaboration Agreement.
In October 2012, we entered into an At-The-Market Issuance Sales Agreement, or sales agreement, with MLV & Co. LLC, or MLV, which provides that, upon the terms and subject to the conditions and limitations set forth in the sales agreement, we may elect to issue and sell shares of our common stock having an aggregate offering price of up to $50.0 million from time to time through MLV as our sales agent. We are not obligated to make any sales of common stock under the sales agreement. To date, we have not sold any common stock pursuant to the sales agreement. The sales agreement will expire in October 2015 unless extended by the parties.
We may need additional capital resources in order to support development and commercialization of imetelstat, especially if we elect to exercise our U.S. Opt-In Rights and U.S. Co-Promotion Option under the Collaboration Agreement and potentially independently pursue imetelstat development under our own IDP, and to otherwise support the future growth of our business through the acquisition of other oncology products, programs or companies. We cannot assure you that our existing capital resources, future interest income, potential milestone payments and royalties under the Collaboration Agreement with Janssen and potential future sales of our common stock, including pursuant to our
75
sales agreement with MLV, will be sufficient to fund future planned activities. The timing and degree of any future capital requirements will depend on many factors, including:
- •
- the accuracy of the assumptions underlying our estimates for our capital needs;
- •
- whether we elect U.S. Opt-In Rights to share future U.S. development and promotion costs for imetelstat under the Collaboration
Agreement with Janssen;
- •
- to the extent permitted under the Collaboration Agreement, whether we independently pursue imetelstat development under our own IDP;
- •
- our potential reimbursement obligations to Janssen if any data from a Janssen IDP support approval by a regulatory agency in the
United States or other countries;
- •
- the achievement of development, regulatory and commercial milestones resulting in the payment to us from Janssen under the
Collaboration Agreement and the timing of receipt of such payments, if any;
- •
- changes or delays in our and Janssen's development plans for imetelstat, including changes which may result from any future clinical
holds on the INDs for imetelstat, including the IND for the MF Pilot Study, that we expect to transfer to Janssen;
- •
- Janssen's ability to meaningfully reduce manufacturing costs of imetelstat;
- •
- the progress, timing, magnitude, scope and costs of clinical development, manufacturing and commercialization for imetelstat,
including the number of indications being pursued, subject to permission from the FDA;
- •
- the time and costs involved in obtaining regulatory clearances and approvals in the United States and in other countries;
- •
- Janssen's ability to successfully market and sell imetelstat, upon regulatory approval or clearance, in the United States and other
countries;
- •
- our decision to exercise our U.S. Co-Promotion Option, including the costs and timing of building a U.S. sales force;
- •
- the timing, receipt and amount of royalties under the Collaboration Agreement on worldwide net sales of imetelstat, upon regulatory
approval or clearance, if any;
- •
- the timing, receipt and amount of royalties on sales of any stem cell products by Asterias, upon development, regulatory approval or
clearance, if any;
- •
- the sales price and availability of adequate third-party reimbursement for imetelstat;
- •
- the cost of acquiring and/or licensing any new product candidates, if any;
- •
- expenses associated with the pending and potential additional related purported securities lawsuits and derivative lawsuits, as well
as any other litigation; and
- •
- the costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing patent claims.
In addition, changes in our business may occur that would consume available capital resources sooner than we expect. If our existing capital resources, future interest income, and potential milestone payments and royalties under the Collaboration Agreement with Janssen are insufficient to meet future capital requirements, we will need to raise additional capital to fund our operations. Further, if the Collaboration Agreement is terminated, including as a result of Janssen's failure to provide an affirmative Continuation Decision to us, we would not receive any milestone payments or royalties under the Collaboration Agreement, and we would be required to fund all clinical development,
76
manufacturing and commercial activities for imetelstat ourselves, which would require us to raise additional capital or establish alternative collaborations with third-party collaboration partners, which may not be possible. Additional financing through public or private equity financings, including pursuant to our sales agreement with MLV, capital lease transactions or other financing sources may not be available on acceptable terms, or at all. We may raise equity capital at a stock price or on other terms that could result in substantial dilution of ownership for our stockholders. The receptivity of the public and private equity markets to proposed financings is substantially affected by the general economic, market and political climate and by other factors which are unpredictable and over which we have no control.
Our ability to raise additional funds will be severely impaired in the event of:
- •
- any future clinical holds on any IND for imetelstat;
- •
- failure to show adequate safety or efficacy of imetelstat in existing or potential future clinical trials; or
- •
- a termination of the Collaboration Agreement or our collaboration with Janssen is otherwise unsuccessful.
If sufficient capital is not available, we may be unable to fulfill our funding obligations under the Collaboration Agreement with Janssen resulting in our breach of the Collaboration Agreement which could lead to Janssen paying lower milestone payments and lower royalties to us under a reduced royalty tier which could have a material adverse effect on our results of operations and financial condition.
Moreover, we plan to diversify our sole product candidate development risk by identifying and seeking to acquire or in-license new product opportunities for development. Acquisition or in-licensing opportunities that we may pursue could materially affect our liquidity and capital resources and may require us to incur indebtedness, seek equity capital or both. In addition, there can be no assurance that sufficient additional capital would be available to us in order to pursue any of these opportunities.
Cash Flows from Operating Activities
Net cash provided by operations was $9.4 million in 2014. Net cash used in operations was $36.7 million and $55.1 million in 2013 and 2012, respectively. The decrease in net cash used in operations in 2014 compared to 2013 primarily reflects the receipt of $35.0 million from Janssen as an upfront payment in December 2014 upon the effectiveness of the Collaboration Agreement. Additionally, the decrease in net cash used in operations in 2014 compared to 2013 and in 2013 compared to 2012 reflects reduced operating expenses due to previous restructurings and the wind-down of our GRN1005 trials in patients with brain metastases and imetelstat trials in solid tumors.
Cash Flows from Investing Activities
Net cash used in investing activities was $77.9 million in 2014. Net cash provided by investing activities was $21.1 million and $61.0 million in 2013 and 2012, respectively. The decrease in net cash provided by investing activities in 2014 compared to 2013 primarily reflects higher purchases of marketable securities with the net cash proceeds received from an underwritten public offering of shares of our common stock that we completed in February 2014. The decrease in net cash provided by investing activities in 2013 compared to 2012 was primarily the result of lower proceeds from maturities of marketable securities relative to purchases of marketable securities.
For the three years ended December 31, 2014, we have purchased approximately $1.0 million in property and equipment, none of which was financed through equipment financing arrangements. In
77
2014, we closed the certificate of deposit for our unused equipment line of credit upon maturity and transferred the cash proceeds to our cash operating accounts. This action also cancelled the availability of the equipment line of credit.
Cash Flows from Financing Activities
Net cash provided by financing activities in 2014, 2013 and 2012 was $98.4 million, $6.6 million and $150,000, respectively. Net cash provided by financing activities in 2014 primarily reflects the receipt of net cash proceeds of approximately $96.8 million, after deducting the underwriting discount and offering expenses payable by us, from the underwritten public offering of 25,875,000 shares of our common stock at a public offering price of $4.00 per share that we completed in February 2014. Net cash provided by financing activities in 2013 and 2012 reflects proceeds from the issuance of common stock under our employee equity plans.
Significant Cash and Contractual Obligations
As of December 31, 2014, our contractual obligations for the next five years and thereafter were as follows:
| |
Payments Due by Period | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Contractual Obligations(1)
|
Total | Less Than 1 Year |
1 - 3 Years | 4 - 5 Years | After 5 Years |
|||||||||||
| |
(In thousands) |
|||||||||||||||
Equipment lease |
$ | 20 | $ | 10 | $ | 10 | $ | — | $ | — | ||||||
Operating leases(2) |
959 | 885 | 74 | — | — | |||||||||||
Research funding and license fees(3) |
313 | 88 | 90 | 90 | 45 | |||||||||||
| | | | | | | | | | | | | | | | | |
Total contractual cash obligations |
$ | 1,292 | $ | 983 | $ | 174 | $ | 90 | $ | 45 | ||||||
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | |
- (1)
- This
table does not include payments under our severance plan if there were a change in control of Geron or severance payments to employees in the event of
an involuntary termination. In addition, this table does not include any royalty obligations under our license agreements as the timing and likelihood of such payments are not known.
- (2)
- In
February 2014, we amended the lease agreement for our premises at 149 Commonwealth Drive to extend the lease term through January 2016. Our amended lease
at 149 Commonwealth Drive includes an option to extend the lease for one additional period of 18 months. Operating lease obligations in the table above do not include payments due under the
amended lease agreement for the extended lease term or assume the exercise by us of any right of termination.
- (3)
- Research funding is comprised of sponsored research commitments at various laboratories around the world. License fees are comprised of minimum annual license payments under our existing license agreements with several universities and companies for the right to use intellectual property related to technologies that we have in-licensed.
Off-Balance Sheet Arrangements
We have not engaged in any off-balance sheet arrangements, including the use of structured finance, special purpose entities or variable interest entities.
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
The following discussion about our market risk disclosures contains forward-looking statements. Actual results could differ materially from those projected in the forward-looking statements. We are exposed to credit risk and interest rate risk. We do not use derivative financial instruments for speculative or trading purposes.
78
Credit Risk. We currently place our cash, restricted cash, cash equivalents and marketable securities with four financial institutions in the United States. Deposits with banks may exceed the amount of insurance provided on such deposits. While we monitor the cash balances in our operating accounts and adjust the cash balances as appropriate, these cash balances could be impacted if the underlying financial institutions fail or could be subject to other adverse conditions in the financial markets. Financial instruments that potentially subject us to concentrations of credit risk consist primarily of cash equivalents and marketable securities. Cash equivalents and marketable securities currently consist of money market funds, U.S. government-sponsored enterprise securities, commercial paper and corporate notes. Our investment policy, approved by the audit committee of our board of directors, limits the amount we may invest in any one type of investment issuer, thereby reducing credit risk concentrations. We limit our credit and liquidity risks through our investment policy and through regular reviews of our portfolio against our policy. To date, we have not experienced any loss or lack of access to cash in our operating accounts or to our cash equivalents and marketable securities in our investment portfolio. The effect of a hypothetical decrease of 10% in the average yield earned on our cash equivalents and marketable securities would have resulted in an immaterial decrease in our interest income for the year ended December 31, 2014.
Interest Rate Risk. The primary objective of our investment activities is to manage our marketable securities portfolio to preserve principal and liquidity while maximizing the return on the investment portfolio through the full investment of available funds without significantly increasing risk. To achieve this objective, we primarily invest in widely diversified investments with fixed interest rates, which carry a degree of interest rate risk. Fixed rate securities may have their fair value adversely impacted due to a rise in interest rates. Due in part to these factors, our future interest income may fall short of expectations due to changes in market conditions and in interest rates or we may suffer losses in principal if forced to sell securities which may have declined in fair value due to changes in interest rates.
The fair value of our cash equivalents and marketable securities at December 31, 2014 was $167.9 million. These investments include $40.4 million of cash equivalents which are due in less than 90 days, $108.6 million of short-term investments which are due in less than one year and $18.9 million of long-term investments which are due in one to two years. We primarily invest our marketable securities portfolio in securities with at least an investment grade rating to minimize interest rate and credit risk as well as to provide for an immediate source of funds. Although changes in interest rates may affect the fair value of the marketable securities portfolio and cause unrealized gains or losses, such gains or losses would not be realized unless the investments are sold. Due to the nature of our investments, which are primarily money market funds, U.S. government-sponsored enterprise securities, commercial paper and corporate notes, we have concluded that there is no material interest rate risk exposure and a 1% movement in market interest rates would not have a significant impact on the total value of our portfolio.
79
ITEM 8. CONSOLIDATED FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
The following consolidated financial statements and the related notes thereto, of Geron Corporation and the Report of Independent Registered Public Accounting Firm, Ernst & Young LLP, are filed as a part of this annual report on Form 10-K.
80
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
The Board of Directors and Stockholders of Geron Corporation
We have audited the accompanying consolidated balance sheets of Geron Corporation as of December 31, 2014 and 2013, and the related consolidated statements of operations, comprehensive loss, stockholders' equity and cash flows for each of the three years in the period ended December 31, 2014. These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the consolidated financial position of Geron Corporation at December 31, 2014 and 2013, and the consolidated results of its operations and its cash flows for each of the three years in the period ended December 31, 2014, in conformity with U.S. generally accepted accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), Geron Corporation's internal control over financial reporting as of December 31, 2014, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework) and our report dated March 11, 2015 expressed an unqualified opinion thereon.
/s/
Ernst & Young LLP
Redwood City, California
March 11, 2015
81
GERON CORPORATION
CONSOLIDATED BALANCE SHEETS
| |
December 31, | ||||||
|---|---|---|---|---|---|---|---|
| |
2014 | 2013 | |||||
| |
(In thousands, except share and per share data) |
||||||
ASSETS |
|||||||
Current assets: |
|||||||
Cash and cash equivalents |
$ | 42,796 | $ | 12,990 | |||
Restricted cash |
266 | 795 | |||||
Marketable securities |
108,645 | 52,234 | |||||
Interest and other receivables |
963 | 564 | |||||
Prepaid assets |
736 | 474 | |||||
| | | | | | | | |
Total current assets |
153,406 | 67,057 | |||||
Noncurrent marketable securities |
18,932 | — | |||||
Property and equipment, net |
173 | 92 | |||||
Deposits and other assets |
— | 195 | |||||
| | | | | | | | |
|
$ | 172,511 | $ | 67,344 | |||
| | | | | | | | |
| | | | | | | | |
| | | | | | | | |
LIABILITIES AND STOCKHOLDERS' EQUITY |
|||||||
Current liabilities: |
|||||||
Accounts payable |
$ | 1,033 | $ | 1,397 | |||
Accrued compensation and benefits |
4,213 | 3,946 | |||||
Accrued restructuring charges |
— | 94 | |||||
Accrued liabilities |
1,537 | 1,783 | |||||
Deferred revenue |
35,000 | — | |||||
Fair value of derivatives |
16 | 367 | |||||
| | | | | | | | |
Total current liabilities |
41,799 | 7,587 | |||||
Commitments and contingencies |
|||||||
Stockholders' equity: |
|||||||
Preferred stock, $0.001 par value; 3,000,000 shares authorized; no shares issued and outstanding at December 31, 2014 and 2013 |
— | — | |||||
Common stock, $0.001 par value; 300,000,000 shares authorized; 157,429,871 and 130,677,949 shares issued and outstanding at December 31, 2014 and 2013, respectively |
157 | 131 | |||||
Additional paid-in capital |
1,059,072 | 952,403 | |||||
Accumulated deficit |
(928,433 | ) | (892,763 | ) | |||
Accumulated other comprehensive loss |
(84 | ) | (14 | ) | |||
| | | | | | | | |
Total stockholders' equity |
130,712 | 59,757 | |||||
| | | | | | | | |
|
$ | 172,511 | $ | 67,344 | |||
| | | | | | | | |
| | | | | | | | |
| | | | | | | | |
See accompanying notes.
82
GERON CORPORATION
CONSOLIDATED STATEMENTS OF OPERATIONS
| |
Year Ended December 31, | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| |
2014 | 2013 | 2012 | |||||||
| |
(In thousands, except share and per share data) |
|||||||||
Revenues: |
||||||||||
License fees and royalties |
$ | 1,153 | $ | 1,283 | $ | 2,709 | ||||
Operating expenses: |
||||||||||
Research and development |
20,707 | 23,155 | 51,368 | |||||||
Restructuring charges |
— | 1,462 | 2,702 | |||||||
General and administrative |
16,758 | 15,624 | 20,397 | |||||||
| | | | | | | | | | | |
Total operating expenses |
37,465 | 40,241 | 74,467 | |||||||
| | | | | | | | | | | |
Loss from operations |
(36,312 | ) | (38,958 | ) | (71,758 | ) | ||||
Unrealized gain (loss) on derivatives |
351 | (316 | ) | 13 | ||||||
Interest and other income |
373 | 951 | 3,097 | |||||||
Interest and other expense |
(82 | ) | (56 | ) | (233 | ) | ||||
| | | | | | | | | | | |
Net loss |
$ | (35,670 | ) | $ | (38,379 | ) | $ | (68,881 | ) | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
Basic and diluted net loss per share |
$ | (0.23 | ) | $ | (0.30 | ) | $ | (0.54 | ) | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
Shares used in computing basic and diluted net loss per share |
153,540,341 | 128,380,800 | 126,941,024 | |||||||
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
See accompanying notes.
83
GERON CORPORATION
CONSOLIDATED STATEMENTS OF COMPREHENSIVE LOSS
| |
Year Ended December 31, | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| |
2014 | 2013 | 2012 | |||||||
| |
(In thousands) |
|||||||||
Net loss |
$ | (35,670 | ) | $ | (38,379 | ) | $ | (68,881 | ) | |
Other comprehensive income (loss): |
||||||||||
Net unrealized loss on marketable securities |
(70 | ) | (54 | ) | (38 | ) | ||||
Foreign currency translation adjustments |
— | — | 16 | |||||||
Reclassification of accumulated foreign currency translation adjustments |
— | — | 153 | |||||||
| | | | | | | | | | | |
Other comprehensive (loss) income |
(70 | ) | (54 | ) | 131 | |||||
| | | | | | | | | | | |
Comprehensive loss |
$ | (35,740 | ) | $ | (38,433 | ) | $ | (68,750 | ) | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
See accompanying notes.
84
GERON CORPORATION
CONSOLIDATED STATEMENTS OF STOCKHOLDERS' EQUITY
| |
Common Stock | |
|
Accumulated Other Comprehensive Income (Loss) |
|
||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
Additional Paid-In Capital |
Accumulated Deficit |
Total Stockholders' Equity |
||||||||||||||||
| |
Shares | Amount | |||||||||||||||||
| |
(In thousands, except share data) |
||||||||||||||||||
Balances at December 31, 2011 |
131,443,148 | $ | 131 | $ | 932,066 | $ | (785,503 | ) | $ | (91 | ) | $ | 146,603 | ||||||
Net loss |
— | — | — | (68,881 | ) | — | (68,881 | ) | |||||||||||
Other comprehensive income |
— | — | — | — | 131 | 131 | |||||||||||||
Stock-based compensation related to issuance of common stock and options in exchange for services |
170,298 | — | 135 | — | — | 135 | |||||||||||||
Cancellations of non-vested restricted stock under equity plans, net of issuances of common stock |
(2,592,375 | ) | (2 | ) | 269 | — | — | 267 | |||||||||||
Stock-based compensation for equity-based awards to employees and directors |
— | — | 5,311 | — | — | 5,311 | |||||||||||||
401(k) contribution |
1,221,624 | 1 | 2,086 | — | — | 2,087 | |||||||||||||
| | | | | | | | | | | | | | | | | | | | |
Balances at December 31, 2012 |
130,242,695 | 130 | 939,867 | (854,384 | ) | 40 | 85,653 | ||||||||||||
Net loss |
— | — | — | (38,379 | ) | — | (38,379 | ) | |||||||||||
Other comprehensive loss |
— | — | — | — | (54 | ) | (54 | ) | |||||||||||
Stock-based compensation related to issuance of common stock and options in exchange for services |
66,853 | — | 252 | — | — | 252 | |||||||||||||
Cancellations of non-vested restricted stock under equity plans, net of issuances of common stock |
(388,056 | ) | — | 6,553 | — | — | 6,553 | ||||||||||||
Stock-based compensation for equity-based awards to employees and directors |
— | — | 4,435 | — | — | 4,435 | |||||||||||||
401(k) contribution |
756,457 | 1 | 1,296 | — | — | 1,297 | |||||||||||||
| | | | | | | | | | | | | | | | | | | | |
Balances at December 31, 2013 |
130,677,949 | 131 | 952,403 | (892,763 | ) | (14 | ) | 59,757 | |||||||||||
Net loss |
— | — | — | (35,670 | ) | — | (35,670 | ) | |||||||||||
Other comprehensive loss |
— | — | — | — | (70 | ) | (70 | ) | |||||||||||
Issuance of common stock in connection with public offering, net of issuance costs of $6,695 |
25,875,000 | 26 | 96,779 | — | — | 96,805 | |||||||||||||
Stock-based compensation related to issuance of common stock and options in exchange for services |
71,239 | — | 253 | — | — | 253 | |||||||||||||
Issuance of common stock upon net exercise of warrants |
168,039 | — | — | — | — | — | |||||||||||||
Issuances of common stock under equity plans, net of cancellations of non-vested restricted stock |
564,950 | — | 1,555 | — | — | 1,555 | |||||||||||||
Stock-based compensation for equity-based awards to employees and directors |
— | — | 7,658 | — | — | 7,658 | |||||||||||||
401(k) contribution |
72,694 | — | 424 | — | — | 424 | |||||||||||||
| | | | | | | | | | | | | | | | | | | | |
Balances at December 31, 2014 |
157,429,871 | $ | 157 | $ | 1,059,072 | $ | (928,433 | ) | $ | (84 | ) | $ | 130,712 | ||||||
| | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | |
See accompanying notes.
85
GERON CORPORATION
CONSOLIDATED STATEMENTS OF CASH FLOWS
| |
Year Ended December 31, | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| |
2014 | 2013 | 2012 | |||||||
| |
(In thousands) |
|||||||||
Cash flows from operating activities: |
||||||||||
Net loss |
$ | (35,670 | ) | $ | (38,379 | ) | $ | (68,881 | ) | |
Adjustments to reconcile net loss to net cash provided by (used in) operating activities: |
||||||||||
Depreciation and amortization |
47 | 320 | 830 | |||||||
Accretion and amortization on investments, net |
2,889 | 1,322 | 2,184 | |||||||
Loss (gain) on retirement/sales of property and equipment, net |
3 | (831 | ) | (142 | ) | |||||
Loss on write-downs of property and equipment |
— | 200 | 271 | |||||||
Stock-based compensation for services by non-employees |
253 | 252 | 183 | |||||||
Stock-based compensation for employees and directors |
7,658 | 4,435 | 5,311 | |||||||
Amortization related to 401(k) contributions |
111 | 458 | 726 | |||||||
Unrealized (gain) loss on derivatives |
(351 | ) | 316 | (13 | ) | |||||
Changes in assets and liabilities: |
||||||||||
Interest and other receivables |
(399 | ) | 188 | 646 | ||||||
Prepaid assets |
(72 | ) | 1,081 | 1,311 | ||||||
Deposits and other assets |
5 | (4 | ) | 112 | ||||||
Accounts payable |
(364 | ) | (2,032 | ) | 449 | |||||
Accrued compensation and benefits |
580 | (431 | ) | 3,548 | ||||||
Accrued restructuring charges |
(94 | ) | (1,878 | ) | (1,758 | ) | ||||
Accrued liabilities |
(246 | ) | (1,697 | ) | (92 | ) | ||||
Deferred revenue |
35,000 | — | — | |||||||
Translation adjustment |
— | — | 169 | |||||||
| | | | | | | | | | | |
Net cash provided by (used in) operating activities |
9,350 | (36,680 | ) | (55,146 | ) | |||||
Cash flows from investing activities: |
||||||||||
Restricted cash transfer |
529 | (1 | ) | (1 | ) | |||||
Purchases of property and equipment |
(131 | ) | (3 | ) | (862 | ) | ||||
Proceeds from sales of property and equipment |
— | 1,196 | 170 | |||||||
Purchases of marketable securities |
(190,263 | ) | (88,977 | ) | (79,369 | ) | ||||
Proceeds from sales/calls of marketable securities |
10,549 | — | — | |||||||
Proceeds from maturities of marketable securities |
101,412 | 108,839 | 141,016 | |||||||
| | | | | | | | | | | |
Net cash (used in) provided by investing activities |
(77,904 | ) | 21,054 | 60,954 | ||||||
Cash flows from financing activities: |
||||||||||
Proceeds from issuance of common stock, net of issuance costs |
98,360 | 6,553 | 150 | |||||||
| | | | | | | | | | | |
Net cash provided by financing activities |
98,360 | 6,553 | 150 | |||||||
| | | | | | | | | | | |
Net increase (decrease) in cash and cash equivalents |
29,806 | (9,073 | ) | 5,958 | ||||||
Cash and cash equivalents, at beginning of year |
12,990 | 22,063 | 16,105 | |||||||
| | | | | | | | | | | |
Cash and cash equivalents, at end of year |
$ | 42,796 | $ | 12,990 | $ | 22,063 | ||||
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
See accompanying notes.
86
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
1. ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Organization
Geron Corporation, or we or Geron, was incorporated in the State of Delaware on November 28, 1990. We are a clinical stage biopharmaceutical company focused on the development of a telomerase inhibitor, imetelstat, in hematologic myeloid malignancies. Principal activities to date have included obtaining financing, securing operating facilities and conducting research and development. In November 2014, we entered into an exclusive collaboration and license agreement, or the Collaboration Agreement, with Janssen Biotech, Inc., or Janssen, to develop and commercialize imetelstat worldwide for oncology, including hematologic myeloid malignancies, and all other human therapeutic uses. The significance of future losses will depend on whether Janssen continues to develop and advance imetelstat and the clinical and commercial success of imetelstat, which would result in future revenues to us in the form of milestone payments and royalties under the Collaboration Agreement, as described below, and whether we acquire other oncology products, programs or companies to diversify our business. There can be no assurance that we will receive any milestone payments or royalties from Janssen in the future. Imetelstat, which is our sole product candidate, will require significant additional clinical testing prior to possible regulatory approval in the United States and other countries, and we do not expect imetelstat to be commercially available for many years, if at all.
Principles of Consolidation
The consolidated financial statements include the accounts of Geron and our former wholly-owned subsidiary, Geron Bio-Med Ltd. (Geron Bio-Med), a United Kingdom company. In March 2012, the board of directors and shareholders of Geron Bio-Med approved actions to commence a voluntary winding up of the company. The full wind up of Geron Bio-Med was completed in August 2012. Prior to 2013, we eliminated intercompany accounts and transactions and prepared the financial statements of Geron Bio-Med using the local currency as the functional currency. We translated the assets and liabilities of Geron Bio-Med at rates of exchange at the balance sheet date and translated income and expense items at average monthly rates of exchange. The resultant translation adjustments were included in accumulated other comprehensive income (loss), a separate component of stockholders' equity.
Net Loss Per Share
Basic earnings (loss) per share is calculated based on the weighted average number of shares of common stock outstanding during the period. Diluted earnings (loss) per share is calculated based on the weighted average number of shares of common stock and potential dilutive securities outstanding during the period. Potential dilutive securities primarily consist of outstanding stock options, restricted stock awards and warrants to purchase common stock and are determined using the treasury stock method at an average market price during the period.
Because we are in a net loss position, diluted loss per share excludes the effects of potential dilutive securities. Had we been in a net income position, diluted earnings per share would have included the shares used in the computation of basic net loss per share as well as an additional 3,072,340, 532,120 and 11,497 shares for 2014, 2013 and 2012, respectively, related to outstanding stock options, restricted stock awards and warrants (as determined using the treasury stock method at the estimated average market value).
87
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
1. ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (Continued)
Use of Estimates
The preparation of financial statements in conformity with accounting principles generally accepted in the United States requires us to make estimates and assumptions that affect the amounts reported in the financial statements and accompanying notes. On a regular basis, management evaluates these estimates and assumptions. Actual results could differ from those estimates.
Fair Value of Financial Instruments
Cash Equivalents and Marketable Securities
We consider all highly liquid investments with an original maturity of three months or less to be cash equivalents. We are subject to credit risk related to our cash equivalents and marketable securities. We place our cash and cash equivalents in money market funds and cash operating accounts. Our marketable securities include U.S. government-sponsored enterprise securities, commercial paper and corporate notes with original maturities ranging from four to 19 months.
We classify our marketable securities as available-for-sale. We record available-for-sale securities at fair value with unrealized gains and losses reported in accumulated other comprehensive income (loss) in stockholders' equity. Realized gains and losses are included in interest and other income and are derived using the specific identification method for determining the cost of securities sold and have been insignificant to date. Dividend and interest income are recognized when earned and included in interest and other income in our consolidated statements of operations. We recognize a charge when the declines in the fair values below the amortized cost basis of our available-for-sale securities are judged to be other-than-temporary. We consider various factors in determining whether to recognize an other-than-temporary charge, including whether we intend to sell the security or whether it is more likely than not that we would be required to sell the security before recovery of the amortized cost basis. Declines in market value associated with credit losses judged as other-than-temporary result in a charge to interest and other income. Other-than-temporary charges not related to credit losses are included in accumulated other comprehensive income (loss) in stockholders' equity. We have not recorded any other-than-temporary impairment charges for our available-for-sale securities for the years ended December 31, 2014, 2013 and 2012. See Note 2 on Fair Value Measurements.
Non-Marketable Equity Investments
Non-marketable equity investments in companies in which we own less than 20% of the outstanding voting stock and do not otherwise have the ability to exert significant influence over the investees are carried at cost, as adjusted for other-than-temporary impairments. We apply the equity method of accounting for investments in non-marketable nonpublic companies in which we own more than 20% of the outstanding voting stock or otherwise have the ability to exert significant influence over the investees. Under this method, we increase (decrease) the carrying value of our investment by a proportionate share of the investee's earnings (losses). If losses exceed the carrying value of the investment, losses are then applied against any advances to the investee, including any commitment to provide financial support, until those amounts are reduced to zero. Commitments to provide financial support include formal guarantees, implicit arrangements, reputational expectations, intercompany relationships or a consistent past history of providing financial support. The equity method is then suspended until the investee has earnings. Any proportionate share of investee earnings is first applied to the share of accumulated losses not recognized during the period the equity method was suspended.
88
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
1. ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (Continued)
We recognize previously suspended losses to the extent additional investment is determined to represent the funding of prior losses. See Note 7 on Divestiture of Stem Cell Assets.
Fair Value of Derivatives
For non-employee options classified as liabilities, the fair value of these instruments is recorded on the consolidated balance sheet at inception and adjusted to fair value at each financial reporting date. The change in fair value of the non-employee options is recorded in the consolidated statements of operations as unrealized gain (loss) on derivatives. Fair value of non-employee options is estimated using the Black Scholes option-pricing model. The non-employee options continue to be reported as a liability until such time as the instruments are exercised or expire or are otherwise modified to remove the provisions which require this treatment, at which time these instruments are marked to fair value and reclassified from liabilities to stockholders' equity. For non-employee options classified as permanent equity, the fair value of the non-employee options is recorded in stockholders' equity as of their respective vesting dates and no further adjustments are made. See Note 2 on Fair Value Measurements.
Nonmonetary Transactions
We account for nonmonetary transactions based on the fair values of the assets (or services) involved. The cost of a nonmonetary asset acquired in exchange for another nonmonetary asset is the fair value of the asset surrendered to obtain it with a gain or loss recognized on the exchange. We use the fair value of the asset received to measure the cost if it is more clearly evident than the fair value of the asset surrendered. If the fair value of neither the assets received nor the assets relinquished is determinable within reasonable limits, we use the recorded amount (or carrying value) of the nonmonetary assets relinquished to account for the exchange. Similarly, we use carrying value for an exchange of controlled assets that do not meet the definition of a business for a non-controlling non-marketable equity interest in a company with no gain or loss recognized on the exchange. See Note 7 on Divestiture of Stem Cell Assets.
Revenue Recognition
In general, we recognize revenue for each unit of accounting when all of the following criteria have been met: (a) persuasive evidence of an arrangement exists, (b) delivery has occurred or services have been rendered, (c) the seller's price to the buyer is fixed or determinable, and (d) collectability is reasonably assured. Amounts received prior to satisfying these revenue recognition criteria are recorded as deferred revenue. Amounts expected to be recognized as revenue within the 12 months following the balance sheet date are classified as current deferred revenue. Amounts not expected to be recognized as revenue within the 12 months following the balance sheet date are classified as noncurrent deferred revenue.
License and/or Collaboration Agreements
In addition to the Collaboration Agreement with Janssen, we have entered into several license or collaboration agreements with various oncology, diagnostics, research tools and biologics production companies. Economic terms in these agreements may include non-refundable license payments in cash or equity securities, option payments in cash or equity securities, cost reimbursements, cost-sharing
89
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
1. ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (Continued)
arrangements, milestone payments, royalties on future sales of products, or any combination of these items. In applying the appropriate revenue recognition guidance related to these agreements, we first assess whether the arrangement contains multiple elements. In this evaluation, we consider: (1) the deliverables included in the arrangement and (2) whether the individual deliverables represent separate units of accounting or whether they must be accounted for as a combined unit of accounting. This evaluation involves subjective determinations and requires us to make judgments about the individual deliverables and whether such deliverables are separable from the other aspects of the contractual relationship. Deliverables are considered separate units of accounting provided that: (i) the delivered item(s) has value to the customer on a standalone basis, and if (ii) the arrangement includes a general right of return relative to the delivered item(s), delivery or performance of the undelivered item(s) is considered probable and substantially in our control. In assessing whether an item has standalone value, we consider factors such as the research, manufacturing and commercialization capabilities of the collaboration partner and the availability of the associated expertise in the general marketplace. In addition, we consider whether the collaboration partner can use the other deliverable(s) for their intended purpose without the receipt of the remaining element(s), whether the value of the deliverable is dependent on the undelivered item(s) and whether there are other vendors that can provide the undelivered element(s).
Arrangement consideration that is fixed or determinable is allocated among the separate units of accounting using the relative selling price method. We then apply the applicable revenue recognition criteria noted above to each of the separate units of accounting in determining the appropriate period and pattern of recognition. We determine how to allocate arrangement consideration to identified units of accounting based on the selling price hierarchy provided under relevant accounting guidance. The estimated fair value of deliverables under the arrangement may be derived using a best estimate of selling price if vendor-specific-objective evidence and third-party evidence are not available.
Upfront non-refundable signing, license or non-exclusive option fees are recognized as revenue: (i) when rights to use the intellectual property, related to a license that has standalone value from the other deliverables to be provided under the agreement, have been delivered or (ii) over the term of the agreement if we have continuing performance obligations as the arrangement would be accounted for as a single unit of accounting. When payments are received in equity securities, we do not recognize any revenue unless such securities are determined to be realizable in cash.
At the inception of an arrangement that includes milestone payments, we assess whether each milestone is substantive and at risk to both parties on the basis of the contingent nature of the milestone. This evaluation includes an assessment of whether: (i) the consideration is commensurate with either the performance to achieve the milestone or the enhancement of the value of the delivered item(s) as a result of a specific outcome resulting from the performance to achieve the milestone, (ii) the consideration relates solely to past performance and (iii) the consideration is reasonable relative to all of the deliverables and payment terms within the arrangement. We consider various factors such as the scientific, clinical, regulatory, commercial and other risks that must be overcome to achieve the respective milestone and the level of effort and investment required to achieve the respective milestone in making this assessment. There is considerable judgment involved in determining whether a milestone satisfies all of the criteria required to conclude that a milestone is substantive. Milestone payments for milestones that are considered substantive would be recognized as revenue in their entirety upon successful accomplishment of the milestone, assuming all other revenue recognition criteria are met.
90
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
1. ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (Continued)
Milestone payments for milestones that are not considered substantive would be recognized as revenue over the remaining period of performance, assuming all other revenue recognition criteria are met.
Royalties are recognized as earned in accordance with contract terms when royalties from licensees can be reasonably estimated and collectability is reasonably assured. If royalties cannot be reasonably estimated or collectability of a royalty amount is not reasonably assured, royalties are recognized as revenue when the cash is received. Revenue from commercial milestone payments will be accounted for as royalties and recorded as revenue upon achievement of the milestone, assuming all other revenue recognition criteria are met.
Cost-sharing expenses are recorded as earned or owed based on the performance requirements by both parties under the respective contracts. For arrangements in which we and our collaboration partner in the agreement are exposed to significant risks and rewards depending on the commercial success of the activity, we recognize payments between the parties on a net basis and record such amounts as a reduction or addition to research and development expense. For arrangements in which we have agreed to perform certain research and development services for our collaboration partner and are not exposed to significant risks and rewards depending on the commercial success of the activity, we recognize the respective cost reimbursements as revenue under the collaborative agreement as the related research and development services are rendered.
Restricted Cash
Restricted cash consists of funds maintained in separate certificate of deposit accounts for specified purposes. The components of restricted cash were as follows:
| |
December 31, | ||||||
|---|---|---|---|---|---|---|---|
| (In thousands) |
2014 | 2013 | |||||
Certificate of deposit for unused equipment line of credit |
$ | — | $ | 530 | |||
Certificate of deposit for credit card purchases |
266 | 265 | |||||
| | | | | | | | |
|
$ | 266 | $ | 795 | |||
| | | | | | | | |
| | | | | | | | |
| | | | | | | | |
In 2014, we closed the certificate of deposit for our unused equipment line of credit upon maturity and transferred the cash proceeds to our cash operating accounts. This action also cancelled the availability of the equipment line of credit.
Research and Development Expenses
Research and development expenses consist of expenses incurred in identifying, developing and testing product candidates resulting from our independent efforts as well as efforts associated with collaborations. These expenses include, but are not limited to, in-process research and development acquired in an asset acquisition and deemed to have no alternative future use, payroll and personnel expense, lab supplies, preclinical studies, clinical trials, including support for investigator-sponsored clinical trials, raw materials to manufacture clinical trial drugs, manufacturing costs for research and clinical trial materials, sponsored research at other labs, consulting, costs to maintain technology licenses and research-related overhead. Research and development costs are expensed as incurred, including payments made under our license agreements.
91
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
1. ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (Continued)
Clinical Trial Costs
A significant component of our research and development expenses has historically been clinical trial costs. Substantial portions of our preclinical studies and all of our clinical trials have been performed by third-party contract research organizations, or CROs, and other vendors. We accrue expenses for preclinical studies performed by our vendors based on certain estimates over the term of the service period and adjust our estimates as required. We accrue expenses for clinical trial activities performed by CROs based upon the estimated amount of work completed on each study. For clinical trial expenses, the significant factors used in estimating accruals include the number of patients enrolled, the number of active clinical sites and the duration for which the patients will be enrolled in the study. Pass through costs from CROs include, but are not limited to, regulatory expenses, investigator fees, lab fees, travel costs and other miscellaneous costs, including shipping and printing fees. We accrue pass through costs based on estimates of the amount of work completed for the clinical trial. We monitor patient enrollment levels and related activities to the extent possible through internal reviews, review of contractual terms and correspondence with CROs. We base our estimates on the best information available at the time. However, additional information may become available to us which would allow us to make a more accurate estimate in future periods. In this event, we may be required to record adjustments to research and development expenses in future periods when the actual level of activity becomes more certain.
Depreciation and Amortization
We record property and equipment at cost and calculate depreciation using the straight-line method over the estimated useful lives of the assets, generally four years. Leasehold improvements are amortized over the shorter of the estimated useful life or remaining term of the lease.
Stock-Based Compensation
We maintain various stock incentive plans under which stock options and restricted stock awards are granted to employees, directors and consultants. We also have an employee stock purchase plan for all eligible employees. We recognize stock-based compensation expense on a straight-line basis over the requisite service period, which is generally the vesting period. For additional information, see Note 9 on Stockholders' Equity.
Stock Options and Employee Stock Purchase Plan
We grant service-based stock options under our equity plans to employees, directors and consultants. The vesting period for employee options is generally four years. We use the Black Scholes option-pricing model to estimate the grant-date fair value of our stock options and employee stock plan purchases. The determination of fair value for these stock-based awards on the date of grant using the Black Scholes option-pricing model is affected by our stock price as well as assumptions regarding a number of complex and subjective variables. For additional information, see Note 9 on Stockholders' Equity.
Restricted Stock Awards
We have granted restricted stock awards to employees and directors with three types of vesting schedules: (i) service-based, (ii) performance-based or (iii) market-based. Service-based restricted stock
92
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
1. ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (Continued)
awards generally vest annually over four years. Performance-based restricted stock awards vest upon achievement of discrete strategic corporate goals within a specified performance period, generally three years. Market-based restricted stock awards vest only upon achievement of certain market price thresholds of our common stock within a specified performance period, generally three years.
The fair value for service-based restricted stock awards is determined using the fair value of our common stock on the date of grant. The fair value is amortized as stock-based compensation expense over the requisite service period of the award, which is generally the vesting period, on a straight-line basis and is reduced for estimated forfeitures, as applicable.
The fair value for performance-based restricted stock awards is determined using the fair value of our common stock on the date of grant. Stock-based compensation expense for awards with vesting based on performance conditions is recognized over the period from the date the performance condition is determined to be probable of occurring through the date the applicable condition is expected to be met and is reduced for estimated forfeitures, as applicable. If the performance condition is not considered probable of being achieved, no stock-based compensation expense is recognized until such time as the performance condition is considered probable of being met, if at all. If that assessment of the probability of the performance condition being met changes, the impact of the change in estimate would be recognized in the period of the change. If the requisite service period has been met prior to the change in estimate, the effect of the change in estimate would be recognized immediately. All previously granted performance-based restricted stock awards have been cancelled unvested as the performance conditions were not achieved within the respective performance periods.
The fair value for market-based restricted stock awards is determined using a lattice valuation model with a Monte Carlo simulation. The model takes into consideration the historical volatility of our stock and the risk-free interest rate at the date of grant. In addition, the model is used to estimate the derived service period for the awards. The derived service period is the estimated period of time that would be required to satisfy the market condition, assuming the market condition will be satisfied. Stock-based compensation expense is recognized over the derived service period for the awards using the straight-line method and is reduced for estimated forfeitures, as applicable, but is accelerated if the market condition is achieved earlier than estimated. If a market-based restricted stock award is forfeited or expires after completion of the derived service period, any previously recognized stock-based compensation expense is not reversed. All previously granted market-based restricted stock awards have been cancelled unvested as the market conditions were not achieved within the specified performance period.
Non-Employee Stock-Based Awards
For our non-employee stock-based awards, the measurement date on which the fair value of the stock-based award is calculated is equal to the earlier of: (i) the date at which a commitment for performance by the counterparty to earn the equity instrument is reached or (ii) the date at which the counterparty's performance is complete. We recognize stock-based compensation expense for the fair value of the vested portion of non-employee awards in our consolidated statements of operations.
93
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
1. ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (Continued)
Accumulated Other Comprehensive Loss
Accumulated other comprehensive loss includes certain changes in stockholders' equity which are excluded from net loss. The components of accumulated other comprehensive loss were as follows:
| |
December 31, | ||||||
|---|---|---|---|---|---|---|---|
| (In thousands) |
2014 | 2013 | |||||
Unrealized loss on marketable securities |
$ | (84 | ) | $ | (14 | ) | |
| | | | | | | | |
| | | | | | | | |
| | | | | | | | |
Income Taxes
We maintain deferred tax assets and liabilities that reflect the net tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes and are subject to tests of recoverability. Our deferred tax assets include net operating loss carryforwards, research credits and capitalized research and development. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. Our net deferred tax asset has been fully offset by a valuation allowance because of our history of losses. Any potential accrued interest and penalties related to unrecognized tax benefits within operations would be recorded as income tax expense.
Concentrations of Customers and Suppliers
The majority of our revenues was earned in the United States. Two customers accounted for approximately 31%, 42% and 59% of our 2014, 2013 and 2012 revenues, respectively.
We contract third-party manufacturers to produce GMP-grade drugs for preclinical and clinical studies. We also contract for starting materials to supply those manufacturers and us. Certain development and clinical activities may be delayed if we or Janssen are unable to obtain sufficient quantities of starting materials or GMP-grade drugs from current third-party suppliers or other third-party sources.
Recent Accounting Pronouncements
In April 2014, the Financial Accounting Standards Board, or FASB, issued Accounting Standard Update No. 2014-08, Presentation of Financial Statements and Property, Plant, and Equipment: Reporting Discontinued Operations and Disclosures of Disposals of Components of an Entity, or ASU 2014-08. ASU 2014-08 raised the threshold for a disposal of assets to qualify as a discontinued operation and requires new disclosures for both discontinued operations and disposals of individually significant components of a business that do not qualify as discontinued operations. Under the new guidance, only disposals of assets representing a strategic shift in operations that has a major effect on the entity's operations and financial results should be presented as discontinued operations. If the disposal does qualify as a discontinued operation, the entity will be required to provide expanded disclosures, as well as disclosure of the pretax income attributable to the disposal of a significant part of an entity that does not qualify as a discontinued operation. ASU 2014-08 is effective for us beginning January 1, 2015 and subsequent interim periods. We do not expect the adoption of ASU 2014-08 to have a material effect on our consolidated financial statements.
94
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
1. ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (Continued)
In May 2014, the FASB issued Accounting Standard Update No. 2014-09, Revenue from Contracts with Customers, or ASU 2014-09. ASU 2014-09 provides a single comprehensive model for entities to use in accounting for revenue arising from contracts with customers and supersedes most current revenue recognition guidance, including industry-specific guidance. ASU 2014-09 will require an entity to recognize revenue when it transfers promised goods or services to customers in an amount that reflects the consideration to which the entity expects to be entitled in exchange for those goods or services. This update creates a five-step model that requires entities to exercise judgment when considering the terms of the contract(s). The five-step model includes: (i) identifying the contract(s) with the customer, (ii) identifying the separate performance obligations in the contract, (iii) determining the transaction price, (iv) allocating the transaction price to the separate performance obligations, and (v) recognizing revenue when each performance obligation is satisfied. ASU 2014-09 also requires additional disclosure about the nature, amount, timing and uncertainty of revenue and cash flows arising from customer contracts, including significant judgments and changes in judgments and assets recognized from costs incurred to obtain or fulfill a contract. ASU 2014-09 will be effective for us beginning January 1, 2017 and subsequent interim periods. We have the option to apply the provisions of ASU 2014-09 either retrospectively to each prior reporting period presented or retrospectively with the cumulative effect of applying this accounting standard recognized at the date of initial application. Early adoption is not permitted. We are currently evaluating the transition method and the impact that the adoption of ASU 2014-09 will have on our consolidated financial statements and related disclosures.
In August 2014, the FASB issued Accounting Standard Update No. 2014-15, Presentation of Financial Statements—Going Concern (Subtopic 205-40): Disclosure of Uncertainties about an Entity's Ability to Continue as a Going Concern, or ASU 2014-15. ASU 2014-15 is intended to define management's responsibility to evaluate whether there is substantial doubt about an organization's ability to continue as a going concern and to provide related footnote disclosures. Substantial doubt about an entity's ability to continue as a going concern exists when relevant conditions and events, considered in the aggregate, indicate that it is probable that the entity will be unable to meet its obligations as they become due within one year after the date that the financial statements are issued (or available to be issued). ASU 2014-15 provides guidance to an organization's management, with principles and definitions that are intended to reduce diversity in the timing and content of disclosures that are commonly provided by organizations in financial statement footnotes. ASU 2014-15 will be effective for us beginning December 31, 2016 and subsequent interim periods. Early adoption is permitted. We are currently in the process of evaluating the impact of adoption of ASU 2014-15 on our consolidated financial statements and related disclosures.
95
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
2. FAIR VALUE MEASUREMENTS
We categorize financial instruments recorded at fair value on our consolidated balance sheets based upon the level of judgment associated with inputs used to measure their fair value. The categories are as follows:
| Level 1— | Inputs are unadjusted, quoted prices in active markets for identical assets or liabilities at the measurement date. An active market for the asset or liability is a market in which transactions for the asset or liability occur with sufficient frequency and volume to provide pricing information on an ongoing basis. | |
Level 2— |
Inputs (other than quoted market prices included in Level 1) are either directly or indirectly observable for the asset or liability through correlation with market data at the measurement date and for the duration of the instrument's anticipated life. |
|
Level 3— |
Inputs reflect management's best estimate of what market participants would use in pricing the asset or liability at the measurement date. Consideration is given to the risk inherent in the valuation technique and the risk inherent in the inputs to the model. |
A financial instrument's categorization within the valuation hierarchy is based upon the lowest level of input that is significant to the fair value measurement. Below is a description of the valuation methodologies used for financial instruments measured at fair value on our consolidated balance sheets, including the category for such financial instruments.
Cash Equivalents and Marketable Securities Available-for-Sale
Certificates of deposit and money market funds are categorized as Level 1 within the fair value hierarchy as their fair values are based on quoted prices available in active markets. U.S. government-sponsored enterprise securities, commercial paper and corporate notes are categorized as Level 2 within the fair value hierarchy as their fair values are estimated by using pricing models, quoted prices of securities with similar characteristics or discounted cash flows.
96
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
2. FAIR VALUE MEASUREMENTS (Continued)
Cash equivalents, restricted cash and marketable securities by security type at December 31, 2014 were as follows:
| (In thousands) |
Amortized Cost | Gross Unrealized Gains | Gross Unrealized Losses | Estimated Fair Value | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Included in cash and cash equivalents: |
|||||||||||||
Money market funds |
$ | 40,342 | $ | — | $ | — | $ | 40,342 | |||||
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
Restricted cash: |
|||||||||||||
Certificate of deposit |
$ | 266 | $ | — | $ | — | $ | 266 | |||||
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
Marketable securities: |
|||||||||||||
Government-sponsored enterprise securities (due in less than 1 year) |
$ | 401 | $ | — | $ | (1 | ) | $ | 400 | ||||
Government-sponsored enterprise securities (due in 1 to 2 years) |
6,556 | — | (7 | ) | 6,549 | ||||||||
Commercial paper (due in less than 1 year) |
10,985 | 14 | — | 10,999 | |||||||||
Corporate notes (due in less than 1 year) |
97,307 | 2 | (63 | ) | 97,246 | ||||||||
Corporate notes (due in 1 to 2 years) |
12,412 | — | (29 | ) | 12,383 | ||||||||
| | | | | | | | | | | | | | |
|
$ | 127,661 | $ | 16 | $ | (100 | ) | $ | 127,577 | ||||
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
Cash equivalents, restricted cash and marketable securities by security type at December 31, 2013 were as follows:
| (In thousands) |
Amortized Cost | Gross Unrealized Gains | Gross Unrealized Losses | Estimated Fair Value | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Included in cash and cash equivalents: |
|||||||||||||
Money market funds |
$ | 8,079 | $ | — | $ | — | $ | 8,079 | |||||
Corporate notes |
2,206 | — | — | 2,206 | |||||||||
| | | | | | | | | | | | | | |
|
$ | 10,285 | $ | — | $ | — | $ | 10,285 | |||||
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
Restricted cash: |
|||||||||||||
Certificates of deposit |
$ | 795 | $ | — | $ | — | $ | 795 | |||||
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
Marketable securities: |
|||||||||||||
Government-sponsored enterprise securities (due in less than 1 year) |
$ | 7,369 | $ | 1 | $ | (1 | ) | $ | 7,369 | ||||
Commercial paper (due in less than 1 year) |
5,496 | 3 | — | 5,499 | |||||||||
Corporate notes (due in less than 1 year) |
39,383 | 1 | (18 | ) | 39,366 | ||||||||
| | | | | | | | | | | | | | |
|
$ | 52,248 | $ | 5 | $ | (19 | ) | $ | 52,234 | ||||
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
97
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
2. FAIR VALUE MEASUREMENTS (Continued)
Marketable securities with unrealized losses at December 31, 2014 and 2013 were as follows:
| |
Less Than 12 Months | 12 Months or Greater | Total | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (In thousands) |
Estimated Fair Value | Gross Unrealized Losses | Estimated Fair Value | Gross Unrealized Losses | Estimated Fair Value | Gross Unrealized Losses | |||||||||||||
As of December 31, 2014: |
|||||||||||||||||||
Government-sponsored enterprise securities (due in less than 1 year) |
$ | 400 | $ | (1 | ) | $ | — | $ | — | $ | 400 | $ | (1 | ) | |||||
Government-sponsored enterprise securities (due in 1 to 2 years) |
5,549 | (7 | ) | — | — | 5,549 | (7 | ) | |||||||||||
Corporate notes (due in less than 1 year) |
92,989 | (63 | ) | — | — | 92,989 | (63 | ) | |||||||||||
Corporate notes (due in 1 to 2 years) |
12,383 | (29 | ) | — | — | 12,383 | (29 | ) | |||||||||||
| | | | | | | | | | | | | | | | | | | | |
|
$ | 111,321 | $ | (100 | ) | $ | — | $ | — | $ | 111,321 | $ | (100 | ) | |||||
| | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | |
As of December 31, 2013: |
|||||||||||||||||||
Government-sponsored enterprise securities (due in less than 1 year) |
$ | 3,947 | $ | (1 | ) | $ | — | $ | — | $ | 3,947 | $ | (1 | ) | |||||
Corporate notes (due in less than 1 year) |
37,060 | (18 | ) | — | — | 37,060 | (18 | ) | |||||||||||
| | | | | | | | | | | | | | | | | | | | |
|
$ | 41,007 | $ | (19 | ) | $ | — | $ | — | $ | 41,007 | $ | (19 | ) | |||||
| | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | |
The gross unrealized losses related to government-sponsored enterprise securities and corporate notes as of December 31, 2014 and 2013 were due to changes in interest rates. We determined that the gross unrealized losses on our marketable securities as of December 31, 2014 and 2013 were temporary in nature. We review our investments quarterly to identify and evaluate whether any investments have indications of possible impairment. Factors considered in determining whether a loss is temporary include the length of time and extent to which the fair value has been less than the cost basis and whether we intend to sell the security or whether it is more likely than not that we would be required to sell the security before recovery of the amortized cost basis. We currently do not intend to sell these securities before recovery of their amortized cost basis.
Derivatives
Non-employee options are normally traded less actively, have trade activity that is one way, and/or are traded in less-developed markets and are therefore valued based upon models with significant unobservable market parameters, resulting in Level 3 categorization.
Options held by non-employees whose performance obligations are complete are classified as derivative liabilities on our consolidated balance sheets. These options are marked to fair value at each reporting period, and upon the exercise of these options, the instruments are marked to fair value and reclassified from derivative liabilities to stockholders' equity. We have not recorded any reclassifications from current liabilities to stockholders' equity for non-employee option exercises in 2014 and 2013.
98
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
2. FAIR VALUE MEASUREMENTS (Continued)
As of December 31, 2014 and 2013, the following non-employee options to purchase common stock were considered derivatives and classified as current liabilities:
| |
|
Number of Shares at December 31, | |
|
Fair Value at December 31, | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
Exercise Price |
Exercisable Date |
Expiration Date |
|||||||||||||||||
Issuance Date
|
2014 | 2013 | 2014 | 2013 | ||||||||||||||||
| |
|
|
|
|
|
(In thousands) |
||||||||||||||
March 2005 |
$ | 6.39 | 284,600 | 284,600 | January 2007 | March 2015 | $ | 16 | $ | 367 | ||||||||||
| | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | |
The fair value of derivatives has been calculated at each reporting date using the Black Scholes option-pricing model with the following assumptions:
| |
December 31, | |||
|---|---|---|---|---|
| |
2014 | 2013 | ||
Dividend yield |
0% | 0% | ||
Expected volatility |
0.895 | 0.844 | ||
Risk-free interest rate |
0.04% | 0.13% | ||
Expected term |
0.25 yr | 1 yr | ||
Dividend yield is based on historical cash dividend payments. The expected volatility is based on historical volatilities of our stock since traded options on Geron common stock do not correspond to derivatives' terms and trading volume of Geron options is limited. The risk-free interest rate is based on the U.S. Zero Coupon Treasury Strip Yields for the expected term in effect on the reporting date. The expected term of derivatives is equal to the remaining contractual term of the instruments.
Fair Value on a Recurring Basis
The following table presents information about our financial instruments that are measured at fair value on a recurring basis as of December 31, 2014 and indicates the fair value category assigned.
| |
Fair Value Measurements at Reporting Date Using | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
Quoted Prices in Active Markets for Identical Assets / Liabilities |
Significant Other Observable Inputs |
Significant Unobservable Inputs |
|
|||||||||
| (In thousands) |
Level 1 | Level 2 | Level 3 | Total | |||||||||
Assets |
|||||||||||||
Money market funds(1) |
$ | 40,342 | $ | — | $ | — | $ | 40,342 | |||||
Government-sponsored enterprise securities(2)(3) |
— | 6,949 | — | 6,949 | |||||||||
Commercial paper(2) |
— | 10,999 | — | 10,999 | |||||||||
Corporate notes(2)(3) |
— | 109,629 | — | 109,629 | |||||||||
| | | | | | | | | | | | | | |
Total |
$ | 40,342 | $ | 127,577 | $ | — | $ | 167,919 | |||||
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
Liabilities |
|||||||||||||
Derivatives(4) |
$ | — | $ | — | $ | 16 | $ | 16 | |||||
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
99
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
2. FAIR VALUE MEASUREMENTS (Continued)
The following table presents information about our financial instruments that are measured at fair value on a recurring basis as of December 31, 2013 and indicates the fair value category assigned.
| |
Fair Value Measurements at Reporting Date Using | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
Quoted Prices in Active Markets for Identical Assets / Liabilities |
Significant Other Observable Inputs |
Significant Unobservable Inputs |
|
|||||||||
| (In thousands) |
Level 1 | Level 2 | Level 3 | Total | |||||||||
Assets |
|||||||||||||
Money market funds(1) |
$ | 8,079 | $ | — | $ | — | $ | 8,079 | |||||
Government-sponsored enterprise securities(2) |
— | 7,369 | — | 7,369 | |||||||||
Commercial paper(2) |
— | 5,499 | — | 5,499 | |||||||||
Corporate notes(1)(2) |
— | 41,572 | — | 41,572 | |||||||||
| | | | | | | | | | | | | | |
Total |
$ | 8,079 | $ | 54,440 | $ | — | $ | 62,519 | |||||
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
Liabilities |
|||||||||||||
Derivatives(4) |
$ | — | $ | — | $ | 367 | $ | 367 | |||||
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
| | | | | | | | | | | | | | |
- (1)
- Included
in cash and cash equivalents on our consolidated balance sheets.
- (2)
- Included
in current portion of marketable securities on our consolidated balance sheets.
- (3)
- Included
in noncurrent portion of marketable securities on our consolidated balance sheets.
- (4)
- Included in fair value of derivatives on our consolidated balance sheets.
Changes in Level 3 Recurring Fair Value Measurements
The table below includes a rollforward of the balance sheet amounts for the year ended December 31, 2014, including the change in fair value, for financial instruments in the Level 3 category. When a determination is made to classify a financial instrument within Level 3, the determination is based upon the significance of the unobservable parameters to the overall fair value measurement. However, Level 3 financial instruments typically include, in addition to the unobservable components, observable components (that is, components that are actively quoted and can be validated to external
100
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
2. FAIR VALUE MEASUREMENTS (Continued)
sources). Accordingly, the gain in the table below includes changes in fair value due in part to observable factors that are part of the methodology.
| |
Fair Value Measurements Using Significant Unobservable Inputs (Level 3) Year Ended December 31, 2014 |
||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (In thousands) |
Fair Value at December 31, 2013 |
Total Unrealized Gain Included in Earnings(1) |
Purchases, Sales, Issuances, Settlements |
Transfers In and/or Out of Level 3 |
Fair Value at December 31, 2014 |
Change in Unrealized Gain Related to Financial Instruments Held at December 31, 2014(1) |
|||||||||||||
Derivative liabilities |
$ | 367 | $ | (351 | ) | $ | — | $ | — | $ | 16 | $ | (351 | ) | |||||
- (1)
- Reported as unrealized gain on derivatives in our consolidated statements of operations.
Credit Risk
We currently place our cash, restricted cash, cash equivalents and marketable securities with four financial institutions in the United States. Generally, these deposits may be redeemed upon demand and therefore, bear minimal risk. Deposits with banks may exceed the amount of insurance provided on such deposits. Financial instruments that potentially subject us to concentrations of credit risk consist primarily of cash equivalents and marketable securities. Cash equivalents and marketable securities currently consist of money market funds, U.S. government-sponsored enterprise securities, commercial paper and corporate notes. Our investment policy, approved by the audit committee of our board of directors, limits the amount we may invest in any one type of investment issuer, thereby reducing credit risk concentrations.
3. PROPERTY AND EQUIPMENT
Property and equipment, stated at cost, is comprised of the following:
| |
December 31, | ||||||
|---|---|---|---|---|---|---|---|
| (In thousands) |
2014 | 2013 | |||||
Furniture and computer equipment |
$ | 1,158 | $ | 1,092 | |||
Lab equipment |
130 | 118 | |||||
Leasehold improvements |
74 | 74 | |||||
| | | | | | | | |
|
1,362 | 1,284 | |||||
Less accumulated depreciation and amortization |
(1,189 | ) | (1,192 | ) | |||
| | | | | | | | |
|
$ | 173 | $ | 92 | |||
| | | | | | | | |
| | | | | | | | |
| | | | | | | | |
4. EQUIPMENT LINE
In 2009, we renewed our equipment financing facility and had approximately $500,000 available for borrowing. This facility was secured by a certificate of deposit. In 2014, we closed the certificate of deposit for our unused equipment line of credit upon maturity and transferred the cash proceeds to our cash operating accounts. This action also cancelled the availability of the equipment line of credit. We had no amounts due under this facility as of December 31, 2013.
101
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
5. ACCRUED LIABILITIES
Accrued liabilities consisted of the following:
| |
December 31, | ||||||
|---|---|---|---|---|---|---|---|
| (In thousands) |
2014 | 2013 | |||||
Service provider obligations |
$ | 408 | $ | 840 | |||
Clinical trial costs |
513 | 326 | |||||
Other |
616 | 617 | |||||
| | | | | | | | |
|
$ | 1,537 | $ | 1,783 | |||
| | | | | | | | |
| | | | | | | | |
| | | | | | | | |
6. RESTRUCTURINGS
April 2013 Restructuring
On April 25, 2013, we announced the decision to discontinue our discovery research programs and companion diagnostics program based on telomere length and close our research laboratory facility located at 200 Constitution Drive, Menlo Park, California. With this decision, a total of 20 positions were eliminated. In connection with this restructuring, we incurred aggregate restructuring charges of $1,370,000 for the year ended December 31, 2013, of which $624,000 related to one-time termination benefits, including $28,000 of non-cash stock-based compensation expense relating to the extension of the post-termination exercise period through the end of December 2013 for certain stock options previously granted to terminated employees, $200,000 related to non-cash charges for write-downs of excess equipment and leasehold improvements and $546,000 related to costs associated with the closure of our research laboratory facility. In connection with the decision to close our research laboratory facility, we entered into an amendment to the lease agreement for the 200 Constitution Drive facility under which the lease terminated effective December 31, 2013. As consideration for the early termination of the lease, we paid the landlord the remaining rents due under the original term of the lease as well as certain facility maintenance costs, all of which have been included in restructuring charges. The restructuring resulted in aggregate cash expenditures of $1,085,000 after adjustments and non-cash credits. In 2013, we received proceeds of $1,080,000 from the sale of excess laboratory equipment in connection with the closure of our research laboratory facility. All actions associated with this restructuring were completed in 2013, and we do not anticipate incurring any further charges in connection with this restructuring.
The components of the accrued restructuring charges included on our consolidated balance sheet relating to the April 2013 restructuring are summarized in the following table. As of December 31, 2014, we had no remaining obligations related to the April 2013 restructuring:
| (In thousands) |
Employee Severance and Other Benefits |
Facility Related Charges |
Total | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
Beginning accrual balance as of December 31, 2013 |
$ | 21 | $ | 73 | $ | 94 | ||||
Cash payments |
(19 | ) | (73 | ) | (92 | ) | ||||
Adjustments or non-cash credits |
(2 | ) | — | (2 | ) | |||||
| | | | | | | | | | | |
Ending accrual balance as of December 31, 2014 |
$ | — | $ | — | $ | — | ||||
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
102
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
6. RESTRUCTURINGS (Continued)
December 2012 Restructuring
On December 3, 2012, we announced the decision to discontinue development of GRN1005. With this decision, a total of 43 positions were eliminated. In connection with this restructuring, we incurred aggregate restructuring charges of $2,794,000, of which $2,702,000 was recognized for the year ended December 31, 2012 and $92,000 was recognized for the year ended December 31, 2013. The aggregate restructuring charges consisted of $2,523,000 related to one-time termination benefits, including $107,000 of non-cash stock-based compensation expense relating to the extension of the post-termination exercise period through the end of December 2013 for certain stock options previously granted to terminated employees, and $271,000 related to non-cash charges for write-downs of GRN1005 manufacturing equipment. The restructuring resulted in aggregate cash expenditures of $2,271,000 after adjustments and non-cash credits. All actions associated with this restructuring were completed in 2013, and we do not anticipate incurring any further charges in connection with this restructuring.
See Note 15 on Subsequent Event for discussion of an organizational resizing announced in March 2015.
7. DIVESTITURE OF STEM CELL ASSETS
On October 1, 2013, we closed the transaction to divest our human embryonic stem cell assets and our autologous cellular immunotherapy program pursuant to the terms of the previously disclosed asset contribution agreement, or the Contribution Agreement, that we entered into in January 2013 with BioTime, Inc., or BioTime, and BioTime's subsidiary, Asterias Biotherapeutics, Inc., or Asterias (formerly known as BioTime Acquisition Corporation).
In accordance with the terms of the Contribution Agreement, on October 1, 2013 we received 6,537,779 shares of Asterias Series A common stock representing 21.4% of Asterias' outstanding common stock as a class as of that date. We are also entitled to receive royalties from Asterias on the sale of products that are commercialized, if any, in reliance upon the patents we contributed to Asterias. In accordance with our contractual obligations under the Contribution Agreement, we distributed all of the shares of Asterias Series A common stock we received from Asterias to our stockholders on a pro rata basis, other than with respect to fractional shares and shares that would otherwise have been distributed to Geron stockholders residing in certain excluded jurisdictions, which shares, as required by the Contribution Agreement, were sold with the net cash proceeds therefrom distributed ratably to the stockholders who would otherwise have been entitled to receive such shares. We refer to the distribution by us of the Asterias Series A common stock, or cash in lieu thereof, as the Series A Distribution. We completed the Series A Distribution to eligible stockholders on August 15, 2014. As of December 31, 2014, we had no remaining obligations with respect to the Series A Distribution.
We accounted for the divestiture of the stem cell assets as a nonmonetary transaction since we transferred intangible assets in exchange for a non-controlling interest in Asterias. The stem cell assets we contributed consisted primarily of intellectual property and know-how and did not meet the definition of a business for accounting purposes. A business consists of three elements: (i) inputs, (ii) processes and (iii) outputs. To be considered a business, only inputs and processes are required, which together form an integrated set of activities used to create outputs. Since we did not contribute any processes, such as operational processes or an organized workforce with the skills and experience
103
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
7. DIVESTITURE OF STEM CELL ASSETS (Continued)
to provide the necessary processes capable of being applied to inputs to create outputs, we determined the stem cell assets only represented inputs and therefore were not considered an integrated set of activities. Due to the significant research and development necessary to realize the commercial potential of the stem cell assets, we expensed all research and development costs associated with the stem cell assets as incurred and therefore, there was no recorded amount, or carrying value, for the stem cell assets on our consolidated balance sheets. Since the divestiture of the stem cell assets represented the transfer of nonfinancial assets that do not meet the definition of a business in exchange for a non-controlling equity interest in Asterias, we accounted for the transaction using the carrying amount, or book value, of the assets surrendered with no gain or loss recognized on the exchange, consistent with our accounting policy for such transactions. Because the stem cell assets had a carrying amount of zero, we applied a carrying amount of zero to the Asterias Series A common stock received in the divestiture.
We applied the equity method of accounting to our investment in Asterias Series A common stock during the period of ownership from October 1, 2013 through August 15, 2014. Since our investment in Asterias had an initial carrying amount of zero upon the closing of the transactions contemplated by the Contribution Agreement on October 1, 2013 and we had no commitments to provide financial support or obligations to perform services or other activities for Asterias, we suspended the equity method of accounting on October 1, 2013. In addition, since Asterias incurred net losses during our period of ownership, no additional value has been recognized for Asterias Series A common stock. Accordingly, the completion of the Series A Distribution had no impact on our consolidated financial statements.
8. COMMITMENTS AND CONTINGENCIES
Purported Securities and Derivative Lawsuits
On March 14, 2014, a purported securities class action lawsuit was commenced in the United States District Court for the Northern District of California, or the California District Court, naming as defendants us and certain of our officers. The lawsuit alleges violations of the Securities Exchange Act of 1934 in connection with allegedly false and misleading statements made by us related to our Phase 2 trial of imetelstat in patients with ET or PV. The plaintiff alleges, among other things, that we failed to disclose facts related to the occurrence of persistent low-grade LFT abnormalities observed in our Phase 2 trial of imetelstat in ET or PV patients and the potential risk of chronic liver injury following long-term exposure to imetelstat. The plaintiff seeks damages and an award of reasonable costs and expenses, including attorneys' fees. On March 28, 2014, a second purported securities class action lawsuit was commenced in the California District Court, and on June 6, 2014, a third purported securities lawsuit, not styled as a class action, was commenced in the United States District Court for the Southern District of Mississippi, or the Mississippi District Court, naming as defendants us and certain of our officers. These lawsuits, which are based on the same factual background as the purported securities class action lawsuit that commenced on March 14, 2014, also allege violations of the Securities Exchange Act of 1934 and seek damages and an award of reasonable costs and expenses, including attorneys' fees. On June 30, 2014, the California District Court consolidated both of the purported class actions filed in the California District Court and appointed a lead plaintiff and lead counsel to represent the purported class. On July 21, 2014, the California District Court ordered the lead plaintiff to file its consolidated amended complaint, which was filed on September 19, 2014. On August 11, 2014, we filed a motion to transfer the purported securities lawsuit filed in the Mississippi
104
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
8. COMMITMENTS AND CONTINGENCIES (Continued)
District Court to the California District Court. On November 4, 2014, the Mississippi District Court granted our motion and transferred the case to the California District Court, which was thereafter consolidated with the class actions. On November 18, 2014, we filed a motion with the California District Court to dismiss the consolidated amended complaint. The plaintiff's opposition to our motion to dismiss was filed on January 20, 2015, and we filed our reply on February 25, 2015. The court hearing for the motion to dismiss has been scheduled for April 10, 2015. We believe that we have meritorious defenses and intend to defend against these lawsuits vigorously.
On April 21, 2014, a stockholder purporting to act on our behalf filed a derivative lawsuit in the Superior Court of California for the County of San Mateo against certain of our officers and directors. The lawsuit alleges breaches of fiduciary duties by the defendants and other violations of law. In general, the lawsuit alleges that the defendants caused or allowed the dissemination of allegedly false and misleading statements related to our Phase 2 trial of imetelstat in patients with ET or PV. The plaintiff is seeking unspecified monetary damages and other relief, including reforms and improvements to our corporate governance and internal procedures. It is possible that additional derivative lawsuits will be filed with respect to these same or other matters and also naming our officers and directors as defendants. We have not yet responded to the derivative lawsuit, but intend to vigorously defend against the claims alleged and to seek dismissal of the lawsuit.
For a further discussion of these ongoing lawsuits, refer to the section entitled "Legal Proceedings" in Part I, Item 3 of this annual report Form 10-K. These lawsuits and any other related lawsuits are subject to inherent uncertainties, and the actual defense and disposition costs will depend upon many unknown factors. The outcome of these lawsuits is necessarily uncertain. We could be forced to expend significant resources in the defense against these and any other related lawsuits and we may not prevail. We currently are not able to estimate the possible cost to us from these lawsuits, as they are currently at an early stage, and we cannot be certain how long it may take to resolve these lawsuits or the possible amount of any damages that we may be required to pay. Such amounts could be material to our consolidated financial statements even if we prevail in the defense against these lawsuits. We have not established any reserves for any potential liability relating to these lawsuits. It is possible that we could, in the future, incur judgments or enter into settlements of claims for monetary damages.
Indemnifications to Officers and Directors
Our corporate bylaws require that we indemnify our officers and directors, as well as those who act as directors and officers of other entities at our request, against expenses, judgments, fines, settlements and other amounts actually and reasonably incurred in connection with any proceedings arising out of their services to Geron. In addition, we have entered into separate indemnification agreements with each of our directors and officers which provide for indemnification of these directors and officers under similar circumstances and under additional circumstances. The indemnification obligations are more fully described in our bylaws and the indemnification agreements. We purchase standard insurance to cover claims or a portion of the claims made against our directors and officers. Since a maximum obligation is not explicitly stated in our bylaws or in our indemnification agreements and will depend on the facts and circumstances that arise out of any future claims, the overall maximum amount of the obligations cannot be reasonably estimated. We have no such obligations on our consolidated balance sheets as of December 31, 2014 and 2013.
105
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
8. COMMITMENTS AND CONTINGENCIES (Continued)
Operating Lease Commitment
In February 2014, we amended the lease agreement for our premises at 149 Commonwealth Drive to extend the lease term through January 2016. As of December 31, 2014, future minimum payments under our operating lease for our premises at 149 Commonwealth Drive were approximately $959,000. Rent expense under our operating leases was approximately $936,000, $1,422,000 and $1,474,000 for the years ended December 31, 2014, 2013 and 2012, respectively.
Severance Plan
We have an Amended and Restated Severance Plan, or Severance Plan, that applies to all employees that are not subject to performance improvement plans, and most significantly provides for, among other benefits: (i) a severance payment upon a Change of Control Triggering Event and Separation from Service (as defined in the Severance Plan) and (ii) a severance payment for each non-executive employee upon a Non-Change of Control Triggering Event and Separation from Service (as defined in the Severance Plan). A Change of Control Triggering Event is defined as an event where: (i) an employee is terminated by us without cause in connection with a change of control or within 12 months following a change of control provided, however, that if an employee is terminated by us in connection with a change of control but immediately accepts employment with our successor or acquirer, the employee will not be eligible for the benefits outlined in the Severance Plan, (ii) an employee resigns because in connection with a change of control, the offered terms of employment (new or continuing) by us or our successor or acquirer within 30 days after the change of control results in a material change in the terms of employment, or (iii) after accepting (or continuing) employment with us after a change of control, an employee resigns within 12 months following a change of control due to a material change in the terms of employment. A Non-Change of Control Triggering Event is defined as an event where a non-executive employee is terminated by us without cause. Severance payments range from two to 18 months of base salary, depending on the employee's position with us, payable in a lump sum payment. The Severance Plan also provides that the provisions of employment agreements entered into between us and executive or non-executive employees supersede the provisions of the Severance Plan. As of December 31, 2014, all our executive officers have employment agreements with provisions that may provide greater severance benefits than those in the Severance Plan.
9. STOCKHOLDERS' EQUITY
Public Offering
On February 4, 2014, we completed an underwritten public offering of 25,875,000 shares of our common stock at a public offering price of $4.00 per share, resulting in net cash proceeds of approximately $96,805,000 after deducting the underwriting discount and offering expenses payable by us.
106
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
9. STOCKHOLDERS' EQUITY (Continued)
Warrants
As of December 31, 2014, the following warrants to purchase our common stock were outstanding and classified as equity:
Issuance Date
|
Exercise Price |
Number of Shares |
Exercisable Date |
Expiration Date |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
August 2011(1) |
$ | 3.98 | 537,893 | August 2011 | August 2021 | |||||
April 2005 |
$ | 3.75 | 470,000 | April 2005 | April 2015 | |||||
| | | | | | | | | | | |
|
1,007,893 | |||||||||
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
- (1)
- In connection with each disbursement under the loan agreement with CIRM, we were obligated to issue to CIRM a warrant to purchase Geron common stock. Such warrants and the underlying common stock were unregistered. We have no further obligations to issue any additional warrants to CIRM. In December 2014, CIRM exercised a warrant to purchase 461,382 shares of our common stock utilizing the net exercise provision in the warrant resulting in the issuance of 168,039 shares of our common stock.
Equity Plans
2002 Equity Incentive Plan
The 2002 Equity Incentive Plan, or 2002 Plan, expired in May 2012. Upon the adoption of the 2011 Incentive Award Plan in May 2011 (see below), no further grants of options or stock purchase rights were made from the 2002 Plan. Options granted under the 2002 Plan expire no later than ten years from the date of grant. Option exercise prices were equal to 100% of the fair market value of the underlying common stock on the date of grant. Service-based stock options under the 2002 Plan generally vested over a period of four years from the date of the option grant. Other stock awards (restricted stock awards and restricted stock units) had variable vesting schedules which were determined by our board of directors on the date of grant.
2011 Incentive Award Plan
In May 2011, our stockholders approved the adoption of the 2011 Incentive Award Plan, or 2011 Plan. Our board of directors administers the 2011 Plan. The 2011 Plan provides for grants to employees (including officers and employee directors) of either incentive stock or nonstatutory stock options and stock purchase rights to employees (including officers and employee directors) and consultants (including non-employee directors). As of December 31, 2014, an aggregate of 13,384,883 shares of our common stock were available for future grants of equity awards under the 2011 Plan. Pursuant to the terms of the 2011 Plan, any shares subject to outstanding stock options originally granted under the 2002 Plan or 1996 Directors' Stock Option Plan, or outstanding unvested restricted stock awards originally granted under the 2002 Plan, that expire or terminate for any reason prior to exercise or settlement or are forfeited because of the failure to meet a contingency or condition required to vest such shares shall become available for issuance under the 2011 Plan. Options granted under the 2011 Plan expire no later than ten years from the date of grant. Option exercise prices shall be equal to 100% of the fair market value of the underlying common stock on the date of grant. If, at the time we
107
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
9. STOCKHOLDERS' EQUITY (Continued)
grant an option, the optionee directly or by attribution owns stock possessing more than 10% of the total combined voting power of all classes of our stock, the option price shall be at least 110% of the fair market value of the underlying common stock and shall not be exercisable more than five years after the date of grant.
We grant service-based stock options to employees under our 2011 Plan that generally vest over a period of four years from the date of the option grant. Other stock awards (restricted stock awards and restricted stock units) have variable vesting schedules as determined by our board of directors on the date of grant.
Under certain circumstances, options may be exercised prior to vesting, subject to our right to repurchase shares subject to such option at the exercise price paid per share. Our repurchase rights would generally terminate on a vesting schedule identical to the vesting schedule of the exercised option. During 2014, we have not repurchased any shares under the 2011 Plan. As of December 31, 2014, we have no shares outstanding subject to repurchase.
Our Non-Employee Director Compensation Policy adopted by our board of directors in March 2014 provides for the automatic grant of the following types of equity awards under the 2011 Plan:
First Director Option. Each person who becomes a non-employee director, whether by election by our stockholders or by appointment by our board of directors to fill a vacancy, will automatically be granted an option to purchase 70,000 shares of common stock on the date such person first becomes a non-employee director, or First Director Option. The First Director Option shall vest annually over three years upon each anniversary date of appointment to our board of directors.
Subsequent Director Option. Each non-employee director (other than any director receiving a First Director Option on the date of the annual meeting) will automatically be granted a subsequent option to purchase 35,000 shares of common stock, a Subsequent Director Option, on the date of the annual meeting of stockholders in each year during such director's service on our board of directors. The Subsequent Director Option vests one year from the date of grant.
1996 Directors' Stock Option Plan
The 1996 Directors' Stock Option Plan, or 1996 Directors Plan, expired in July 2006 upon which no further option grants were made from the 1996 Directors Plan. The options granted under the 1996 Directors Plan were nonstatutory stock options and expire no later than ten years from the date of grant. The option exercise price was equal to the fair market value of the underlying common stock on the date of grant. Options to purchase shares of common stock generally were 100% vested upon grant, except for options granted upon first appointment to the board of directors. These initial options vested annually over three years upon each anniversary date of appointment to the board of directors.
108
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
9. STOCKHOLDERS' EQUITY (Continued)
2006 Directors' Stock Option Plan
The 2006 Directors' Stock Option Plan, or 2006 Directors Plan, was terminated by our board of directors and replaced by the 2011 Plan in March 2014. No further grants of options were made from the 2006 Directors Plan upon the 2006 Directors Plan's termination. All outstanding awards granted under the 2006 Directors Plan remain subject to the terms of the 2006 Directors Plan and the individual grants made thereunder.
The options granted under the 2006 Directors Plan were nonstatutory stock options, and they expire no later than ten years from the date of grant. The option exercise price was equal to the fair market value of the underlying common stock on the date of grant. The First Director Option granted to non-employee members of the board of directors under the 2006 Directors Plan vested annually over three years upon each anniversary date of appointment to the board of directors. The Subsequent Director Option granted to non-employee members of the board of directors on the date of the annual meeting of stockholders in each year during such director's service on our board of directors under the 2006 Directors Plan vested one year from the date of grant.
Aggregate option and award activity for the 2002 Plan, 2011 Plan, 1996 Directors Plan and 2006 Directors Plan is as follows:
| |
|
Outstanding Options | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
Shares Available For Grant |
Number of Shares |
Weighted Average Exercise Price Per Share |
Weighted Average Remaining Contractual Life (In years) |
Aggregate Intrinsic Value (In thousands) |
|||||||||||
Balance at December 31, 2013 |
16,207,250 | 15,576,216 | $ | 3.04 | $ | 33,798 | ||||||||||
Options granted |
(5,658,931 | ) | 5,658,931 | $ | 4.85 | |||||||||||
Awards granted |
(59,330 | ) | — | $ | — | |||||||||||
Options exercised |
— | (662,626 | ) | $ | 1.94 | |||||||||||
Options cancelled/forfeited |
3,613,577 | (3,613,577 | ) | $ | 5.48 | |||||||||||
Awards cancelled/forfeited |
142,375 | — | $ | — | ||||||||||||
2006 Directors Plan termination |
(860,058 | ) | — | $ | — | |||||||||||
| | | | | | | | | | | | | | | | | |
Balance at December 31, 2014 |
13,384,883 | 16,958,944 | $ | 3.16 | 7.38 | $ | 16,038 | |||||||||
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | |
Options exercisable at December 31, 2014 |
9,129,576 | $ | 3.12 | 6.47 | $ | 9,231 | ||||||||||
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | |
Options fully vested and expected to vest at December 31, 2014 |
16,225,022 | $ | 3.14 | 7.32 | $ | 15,546 | ||||||||||
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | |
The aggregate intrinsic value in the preceding table represents the total intrinsic value, based on Geron's closing stock price of $3.25 per share as of December 31, 2014, which would have been received by the option holders had all the option holders exercised their options as of that date.
There were no options granted with an exercise price below or greater than fair market value of our common stock on the date of grant in 2014, 2013 or 2012. As of December 31, 2014, 2013 and 2012, there were 9,129,576, 8,144,040 and 10,410,194 exercisable options outstanding at weighted average exercise prices per share of $3.12, $4.26 and $5.49, respectively.
109
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
9. STOCKHOLDERS' EQUITY (Continued)
The total pretax intrinsic value of stock options exercised during 2014, 2013 and 2012 was $989,000, $2,787,000 and $100, respectively. Cash received from the exercise of options in 2014, 2013 and 2012 totaled approximately $1,286,000, $6,567,000 and $1,000, respectively. No income tax benefit was realized from stock options exercised in 2014 since we reported an operating loss.
Information about stock options outstanding as of December 31, 2014 is as follows:
| |
Options Outstanding | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
Exercise Price Range
|
Number of Shares |
Weighted Average Exercise Price Per Share |
Weighted Average Remaining Contractual Life (In years) |
|||||||
$1.10 - $1.50 |
4,284,937 | $ | 1.42 | 7.41 | ||||||
$1.51 - $2.14 |
4,278,382 | $ | 1.64 | 7.84 | ||||||
$2.16 - $5.01 |
5,376,639 | $ | 4.29 | 7.91 | ||||||
$5.05 - $9.32 |
3,018,986 | $ | 5.80 | 5.72 | ||||||
| | | | | | | | | | | |
$1.10 - $9.32 |
16,958,944 | $ | 3.16 | 7.38 | ||||||
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
Aggregate restricted stock activity for the 2002 Plan, 2011 Plan and 2006 Directors Plan is as follows:
| |
Number of Shares |
Weighted Average Grant Date Fair Value Per Share |
Weighted Average Remaining Contractual Term (In years) |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
Non-vested restricted stock at December 31, 2013 |
409,437 | $ | 4.84 | 0.82 | ||||||
Granted |
59,330 | $ | 2.67 | |||||||
Vested |
(265,422 | ) | $ | 4.47 | ||||||
Cancelled/forfeited |
(142,375 | ) | $ | 4.66 | ||||||
| | | | | | | | | | | |
Non-vested restricted stock at December 31, 2014 |
60,970 | $ | 4.73 | 0.37 | ||||||
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
The total fair value of restricted stock that vested during 2014, 2013 and 2012 was $782,000, $252,000 and $936,000, respectively.
Employee Stock Purchase Plan
In March 2014, our board of directors adopted the 2014 Employee Stock Purchase Plan, or 2014 Purchase Plan. The 2014 Purchase Plan was approved by our stockholders in May 2014. The 2014 Purchase Plan replaced the 1996 Employee Stock Purchase Plan, or 1996 Purchase Plan, which was terminated effective as of the date the 2014 Purchase Plan was approved by our stockholders. However, outstanding purchase rights granted under the 1996 Purchase Plan prior to its termination remained subject to the terms of the 1996 Purchase Plan. A total of 968,829 shares of our common stock were issued under the 1996 Purchase Plan since its adoption in July 1996 and reserves of 231,171 shares of our common stock for future issuance under the 1996 Purchase Plan were cancelled as of the date of termination and became available for future issuance for other corporate purposes. Under the 2014 Purchase Plan, we are authorized to sell to eligible employees up to an aggregate of 1,000,000 shares of
110
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
9. STOCKHOLDERS' EQUITY (Continued)
Geron common stock. As of December 31, 2014, 24,375 shares of our common stock have been issued under the 2014 Purchase Plan since its adoption.
Under the terms of the 2014 Purchase Plan, employees can choose to have up to 10% of their annual salary withheld to purchase our common stock. An employee may not make additional payments into such account or increase the withholding percentage during the offering period.
The 2014 Purchase Plan is comprised of a series of offering periods, each with a maximum duration (not to exceed 12 months) with new offering periods commencing on January 1st and July 1st of each year. The date an employee enters the offering period will be designated as the entry date for purposes of that offering period. An employee may only participate in one offering period at a time. Each offering period consists of two consecutive purchase periods of six months' duration, with the last day of such period designated a purchase date.
The purchase price per share at which common stock is purchased by the employee on each purchase date within the offering period is equal to 85% of the lower of (i) the fair market value per share of Geron's common stock on the employee's entry date into that offering period or (ii) the fair market value per share of Geron's common stock on the purchase date. If the fair market value of Geron's common stock on the purchase date is less than the fair market value at the beginning of the offering period, a new 12 month offering period will automatically begin on the first business day following the purchase date with a new fair market value.
Stock-Based Compensation for Employees and Directors
We measure and recognize compensation expense for all share-based payment awards made to employees and directors, including employee stock options, restricted stock awards and employee stock purchases, based on grant-date fair values for these instruments. We grant service-based stock options and restricted stock awards under our equity plans to employees, directors and consultants. The vesting period for employee options is generally four years. We use the Black Scholes option-pricing model to estimate the grant-date fair value of our stock options and employee stock purchases. The fair value for service-based restricted stock awards is determined using the fair value of our common stock on the date of grant.
In the past, our board of directors has awarded to our employees and directors performance-based restricted stock awards and market-based restricted stock awards. We have not recognized any stock-based compensation expense for performance-based restricted stock awards in our consolidated statements of operations for the years ended December 31, 2014, 2013 and 2012 as the achievement of the specified performance criteria was not considered probable during that time. All of these awards have been cancelled unvested as the performance conditions were not achieved within the respective performance periods. The fair value for market-based restricted stock awards was determined using a lattice valuation model with a Monte Carlo simulation. All previously granted market-based restricted stock awards have been cancelled unvested as the market conditions were not achieved within the specified performance period.
As stock-based compensation expense recognized in the consolidated statements of operations for the years ended December 31, 2014, 2013 and 2012 is based on awards ultimately expected to vest, it has been reduced for estimated forfeitures but at a minimum, reflects the grant-date fair value of those awards that actually vested in the period. Forfeitures have been estimated at the time of grant based on
111
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
9. STOCKHOLDERS' EQUITY (Continued)
historical data and revised, if necessary, in subsequent periods if actual forfeitures differ from those estimates.
We recognize stock-based compensation expense on a straight-line basis over the requisite service period, which is generally the vesting period. The following table summarizes the stock-based compensation expense related to stock options, restricted stock awards and employee stock purchases for the years ended December 31, 2014, 2013 and 2012 which was allocated as follows:
| |
Year Ended December 31, | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| (In thousands) |
2014 | 2013 | 2012 | |||||||
Research and development |
$ | 2,545 | $ | 1,741 | $ | 2,336 | ||||
Restructuring charges |
— | 28 | 107 | |||||||
General and administrative |
5,113 | 2,666 | 2,868 | |||||||
| | | | | | | | | | | |
Stock-based compensation expense included in operating expenses |
$ | 7,658 | $ | 4,435 | $ | 5,311 | ||||
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
Modifications to the post-termination exercise period of outstanding options held by certain members of our executive management team resulted in additional stock-based compensation expense of $205,000 for the year ended December 31, 2013 and have been reflected in the above table. In addition, stock-based compensation expense has been recognized for the modification of the post-termination exercise period for certain stock options previously granted to employees affected by the April 2013 and December 2012 restructurings, which has been included in restructuring charges in our consolidated statements of operations. See Note 6 on Restructurings for further discussion of the restructurings.
The fair value of stock options granted in 2014, 2013 and 2012 has been estimated at the date of grant using the Black Scholes option-pricing model with the following assumptions:
| |
2014 | 2013 | 2012 | |||
|---|---|---|---|---|---|---|
Dividend yield |
0% | 0% | 0% | |||
Expected volatility range |
0.898 to 0.922 | 0.742 to 0.792 | 0.631 to 0.740 | |||
Risk-free interest rate range |
1.64% to 1.92% | 0.80% to 1.97% | 0.81% to 1.25% | |||
Expected term |
5.5 yrs | 6 yrs | 6 yrs |
The fair value of employee stock purchases in 2014, 2013 and 2012 under the 2014 Purchase Plan and 1996 Purchase Plan has been estimated using the Black Scholes option-pricing model with the following assumptions:
| |
2014 | 2013 | 2012 | |||
|---|---|---|---|---|---|---|
Dividend yield |
0% | 0% | 0% | |||
Expected volatility range |
0.835 to 1.666 | 0.506 to 1.391 | 0.458 to 0.774 | |||
Risk-free interest rate range |
0.06% to 0.15% | 0.09% to 0.21% | 0.06% to 0.21% | |||
Expected term range |
6 mos to 12 mos | 6 mos to 12 mos | 6 mos to 12 mos |
Dividend yield is based on historical cash dividend payments and Geron has paid no dividends to date. The expected volatility range is based on historical volatilities of our stock since traded options on
112
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
9. STOCKHOLDERS' EQUITY (Continued)
Geron stock do not correspond to option terms and the trading volume of options is limited. The risk-free interest rate range is based on the U.S. Zero Coupon Treasury Strip Yields for the expected term in effect on the date of grant for an award. The expected term of options is derived from actual historical exercise and post-vesting cancellation data and represents the period of time that options granted are expected to be outstanding. The expected term of employees' purchase rights is equal to the purchase period.
Based on the Black Scholes option-pricing model, the weighted average estimated fair value of employee stock options granted during the years ended December 31, 2014, 2013 and 2012 was $3.57, $1.03 and $0.89 per share, respectively. The weighted average estimated fair value of employees' purchase rights for the years ended December 31, 2014, 2013 and 2012 was $2.10, $0.75 and $0.59 per share, respectively. As of December 31, 2014, total compensation cost related to unvested share-based payment awards not yet recognized, net of estimated forfeitures, was $15,032,000, which is expected to be recognized over the next 27 months on a weighted-average basis.
Stock-Based Compensation to Service Providers
We grant stock options and restricted stock awards to consultants from time to time in exchange for services performed for us. In general, the stock options and restricted stock awards vest over the contractual period of the consulting arrangement. We granted stock options to purchase 75,000, 80,000 and 50,000 shares of our common stock to consultants in 2014, 2013 and 2012, respectively. The fair value of stock options and restricted stock awards held by consultants is recorded as operating expenses over the vesting term of the respective equity awards. In addition, we will record any increase in the fair value of the stock options and restricted stock awards as the respective equity award vests. We recorded stock-based compensation expense of $94,000, $92,000 and $135,000 for the vested portion of the fair value of stock options and restricted stock awards held by consultants in 2014, 2013 and 2012, respectively.
We have also issued common stock to consultants and vendors in exchange for services either performed or to be performed for us. For these stock issuances, we record a prepaid asset equal to the fair market value of the shares on the date of issuance and amortize the fair value of the shares to our operating expenses on a pro-rata basis as services are performed or goods are received. In 2014, 2013 and 2012, we issued 71,239, 66,853 and 170,298 shares of common stock, respectively, in exchange for goods or services. In 2014, 2013 and 2012, we recognized approximately $158,000, $202,000 and $1,010,000, respectively, of expense in connection with stock grants to consultants and vendors. As of December 31, 2014, $7,000 related to consultant and vendor stock issuances remained as a prepaid asset which is being amortized to our operating expenses on a pro-rata basis as services are incurred or goods are received.
113
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
9. STOCKHOLDERS' EQUITY (Continued)
Common Stock Reserved for Future Issuance
Common stock reserved for future issuance as of December 31, 2014 is as follows:
Outstanding stock options |
16,958,944 | |||
Options and awards available for grant |
13,384,883 | |||
Employee stock purchase plan |
975,625 | |||
Warrants outstanding |
1,007,893 | |||
| | | | | |
Total |
32,327,345 | |||
| | | | | |
| | | | | |
| | | | | |
401(k) Plan
We sponsor a defined-contribution savings plan under Section 401(k) of the Internal Revenue Code covering all full-time U.S. employees, or the Geron 401K Plan. Participating employees may contribute up to the annual Internal Revenue Service contribution limit. The Geron 401K Plan also permits us to provide discretionary matching and profit sharing contributions. The Geron 401K Plan is intended to qualify under Section 401 of the Internal Revenue Code so that contributions by employees or by us, and income earned on the contributions, are not taxable to employees until withdrawn from the Geron 401K Plan. Our contributions, if any, will be deductible by us when made.
In 2014, our board of directors approved a cash matching contribution equal to 50% of each employee's contributions, which was fully vested when paid. We provided the 2014 matching contribution in February 2015. In 2013 and 2012, our board of directors approved a matching contribution equal to 75% and 100% of each employee's contributions, respectively. Those matching contributions were made in our common stock and vest ratably over four years for each year of service completed by the employee, commencing from the date of hire, until they are fully vested when the employee has completed four years of service.
For the vested portion of the 2014 match, we recorded $175,000 as research and development expense and $143,000 as general and administrative expense. For the vested portion of the 2013 match, we recorded $156,000 as research and development expense and $157,000 as general and administrative expense. For the vested portion of the 2012 match, we recorded $616,000 as research and development expense and $259,000 as general and administrative expense. Due to the number of positions eliminated in the previous restructurings, a partial plan termination was triggered in both 2013 and 2012. We accelerated the vesting of unvested prior employer matches for employees affected by the respective restructurings, which resulted in $266,000 and $370,000 of operating expenses in 2013 and 2012, respectively. As of December 31, 2014, approximately $273,000 remained unvested for the 2013, 2012 and 2011 matches which will be amortized to operating expenses as the corresponding years of service are completed by the employees.
Sales Agreement
On October 8, 2012, we entered into an At-the-Market Issuance Sales Agreement, or sales agreement, with MLV & Co. LLC, or MLV, pursuant to which we may elect to issue and sell shares of our common stock having an aggregate offering price of up to $50,000,000 from time to time into the open market at prevailing prices through MLV as our sales agent. We will pay MLV an aggregate commission rate equal to up to 3.0% of the gross proceeds of the sales price per share for common
114
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
9. STOCKHOLDERS' EQUITY (Continued)
stock sold through MLV under the sales agreement. Pursuant to the sales agreement, sales of common stock will be made in such quantities and on such minimum price terms as we may set from time to time. We are not obligated to make any sales of common stock under the sales agreement. As of December 31, 2014, we had not sold any common stock pursuant to the sales agreement. The sales agreement will expire in October 2015 unless extended by the parties.
10. LICENSE AGREEMENTS
Janssen Biotech, Inc.
In November 2014, we and Janssen Biotech, Inc., or Janssen, entered into an exclusive collaboration and license agreement, or Collaboration Agreement, to develop and commercialize imetelstat worldwide for oncology, including hematologic myeloid malignancies, and all other human therapeutic uses. Upon the early termination of the waiting periods under the Hart-Scott-Rodino Antitrust Improvements Act of 1976, the Collaboration Agreement became effective on December 15, 2014. Upon the effectiveness of the Collaboration Agreement, we received $35,000,000 from Janssen as an upfront payment.
Under the Collaboration Agreement, we granted to Janssen exclusive worldwide rights to develop and commercialize imetelstat for all indications, and Janssen is responsible for the development, manufacturing and commercialization of, and seeking regulatory approval for, imetelstat worldwide. Under the Collaboration Agreement, development of imetelstat will initially proceed under a mutually agreed joint clinical development plan, or CDP, which includes two agreed upon Phase 2 studies to be pursued initially, one in myelofibrosis, or the Initial Phase 2 MF Study, and one in myelodysplastic syndrome, or the Initial Phase 2 MDS Study, as well as additional, possible registration studies in myelofibrosis, or MF, and myelodysplastic syndrome, or MDS, and possible exploratory Phase 2 and potential follow-on Phase 3 studies in acute myelogenous leukemia, or AML. Development costs for the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study will be shared between the parties on a 50/50 basis. We expect Janssen to initiate the Initial Phase 2 MF Study in mid-2015, followed later by the Initial Phase 2 MDS Study to be initiated at the end of 2015.
Following the protocol-specified primary analysis of the Initial Phase 2 MF Study or after a certain time period after the initiation of the first Phase 3 MF study, Janssen must notify us whether it elects to maintain its license rights and continue to advance the development of imetelstat in any indication. In the event that the Initial Phase 2 MF Study has been terminated early or suspended, Janssen must instead notify us of its election by the date that is the later of 24 months from the initiation of the planned Initial Phase 2 MDS Study or 24 months from the termination of the planned Initial Phase 2 MF Study or commencement of the suspension period, as applicable.
In the event that Janssen elects to continue to maintain its license rights and advance the development of imetelstat in any indication within the applicable timeframe set forth in the Collaboration Agreement (such election, the Continuation Election), we then would have an option, or the U.S. Opt-In Rights, to share further U.S. development and promotion costs in exchange for higher tiered royalty rates and higher future milestone payments if imetelstat is successfully developed and approved. If we exercise our U.S. Opt-In Rights, then the parties would share U.S. development and promotion costs on a 20/80 basis (Geron 20%, Janssen 80%), we would receive a $65,000,000 milestone payment, or Continuation Fee, at the time of the Continuation Election, and would be eligible to receive additional potential payments of up to $470,000,000 in development and regulatory milestones,
115
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
10. LICENSE AGREEMENTS (Continued)
up to $350,000,000 in sales milestones, and tiered royalties ranging from a mid-teens up to low twenties percentage rate on worldwide net sales of imetelstat in any countries where regulatory exclusivity exists or there are valid claims under the patent rights exclusively licensed to Janssen. In addition, if we exercise our U.S. Opt-In Rights, we then would also have a separate option, or the Co-Promotion Option, to provide 20% of the U.S. selling effort with sales force personnel, in lieu of funding 20% of U.S. promotion costs, upon regulatory approval and commercial launch of imetelstat in the United States. Such co-promotion would be conducted under a Janssen prepared promotion plan, and in accordance with a co-promotion agreement to be agreed by the parties at the time of our exercise of our Co-Promotion Option. We would be responsible for all costs associated with establishing and maintaining a sales force in any conduct of such co-promotion. All product sales would be booked by Janssen. If we do not exercise our U.S. Opt-In Rights, then all further development and promotion costs beyond the Initial Phase 2 MF Study or Initial Phase 2 MDS Study would be borne by Janssen, we would receive the $65,000,000 Continuation Fee at the time of the Continuation Election plus a $70,000,000 payment, or Full U.S. Rights Fee, for Janssen's retention of full U.S. rights to imetelstat, and would be eligible to receive additional potential payments of up to $415,000,000 in development and regulatory milestones, up to $350,000,000 in sales milestones, and tiered royalties ranging from a double-digit up to mid-teens percentage rate on worldwide net sales of imetelstat in any countries where regulatory exclusivity exists or there are valid claims under the patent rights exclusively licensed to Janssen.
Under the terms of the Collaboration Agreement, we remain responsible for prosecuting, at Janssen's direction, the patents licensed to Janssen at the time we entered into the Collaboration Agreement, with costs shared between us and Janssen on a 50/50 basis. For intellectual property developed under the Collaboration Agreement, or Development IP, the party having sole ownership interest in such Development IP would be responsible for prosecuting the patents, with Janssen bearing all of the costs for Development IP solely owned by Janssen and costs shared between the parties on a 50/50 basis for Development IP either jointly owned or solely owned by us.
Under the terms of the Collaboration Agreement, we and Janssen created a joint governance structure, including joint development and steering committees and working groups, to oversee and manage worldwide regulatory, development and manufacturing work under the joint CDP and promotional activities (assuming we exercises our U.S. Opt-In Rights) for imetelstat, with Janssen responsible for the operational implementation of those activities. In addition, either of the parties may propose to the joint development committee imetelstat development for any new indications not then provided for in the joint CDP and if the parties agree such development should be conducted outside of the joint CDP, each of Geron and Janssen would be entitled to independently undertake such development at its own cost, subject to the other party's obligation to provide reimbursement for its specified portion of the costs plus a premium for such independent development following marketing approval of imetelstat in such newly proposed indication as a result of such independent development. In the event that we do not exercise our U.S. Opt-In Rights following Janssen's Continuation Election, the joint governance structure under the Collaboration Agreement would be dissolved, a joint oversight committee would monitor the progress of the collaboration, and we would have no further rights to conduct any independent imetelstat development.
After a Continuation Election by Janssen, the Collaboration Agreement would remain in effect until the expiration of the last-to-expire patent or the royalty obligations on sales of imetelstat cease, unless terminated earlier. If Janssen does not effect a Continuation Election, then the Collaboration
116
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
10. LICENSE AGREEMENTS (Continued)
Agreement would terminate and all rights would revert to us. Janssen may terminate the Collaboration Agreement at any time for convenience and due to a safety-related concern. If a notice of termination from Janssen occurs, we would be entitled to certain continued operational support and cost-sharing under various circumstances and all rights would revert to us.
The terms of the Janssen Collaboration Agreement contain multiple deliverables, which include at inception: (i) exclusive worldwide rights to develop and commercialize imetelstat for all indications, (ii) transfer of know-how and intellectual property, including our obligation to procure supply for manufacturing imetelstat for up to nine months after the effective date of the Collaboration Agreement, (iii) participation on the joint steering committees and working groups and (iv) potential participation in selling imetelstat in the United States, if approved for commercial sale. We concluded the license for exclusive worldwide rights to develop and commercialize imetelstat has standalone value to Janssen based on the technical and financial resources of Janssen, including Janssen's drug development experience, sizeable employee base with specific experience in hematologic malignancies, and sufficient capital to independently develop imetelstat on a global basis. Since Janssen has final decision-making authority in the event a unanimous decision cannot be reached by the joint steering committees, we determined our participation on the joint steering committees does not represent a non-contingent deliverable under the Collaboration Agreement. In addition, we determined our potential participation in selling imetelstat in the United States does not represent a non-contingent deliverable because such participation is uncertain and dependent on the drug being approved for commercial sale, which is not within our control. Accordingly, we have determined delivery of the license rights granted by us to Janssen, together with our performance of the technology transfer-related activities, represents the sole non-contingent deliverable under the Collaboration Agreement. Therefore, we will account for our delivery of the license rights and our performance of the technology transfer-related activities as a single unit of accounting. We currently expect completion of the technology transfer-related activities to occur by September 30, 2015 at which point we expect to fully recognize the $35,000,000 upfront payment from Janssen as license fee revenue. As a result, we have not recognized any revenue related to the Collaboration Agreement in 2014. We have determined that each of the additional potential milestone payments to us under the Collaboration Agreement, including: (i) the Continuation Fee at the time of the Continuation Election, (ii) the Full U.S. Rights Fee if we do not exercise our U.S. Opt-In Rights and (iii) payments based on the achievement of certain development, regulatory or commercial milestones, represent substantive milestones. Consequently, we will recognize revenue for these payments in their entirety upon successful accomplishment of the respective milestone. Royalties on future product sales of imetelstat, if successfully commercialized under the Collaboration Agreement, will be recognized as revenue when earned.
The cost-sharing arrangement with Janssen for the Initial Phase 2 MF Study and the Initial Phase 2 MDS Study began in 2015. Therefore, we have not recorded any payables to Janssen or receivables from Janssen in 2014.
GE Healthcare UK Limited
In June 2009, we entered into a worldwide exclusive license and alliance agreement with GE Healthcare UK, Limited, or GEHC, to develop and commercialize cellular assay products derived from human embryonic stem cells, or hESCs, for use in drug discovery, development and toxicity screening. In connection with the GEHC agreement, we recognized $825,000 as license fee revenue in our
117
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
10. LICENSE AGREEMENTS (Continued)
consolidated statements of operations for the year ended December 31, 2012 which reflects the full recognition of a license payment from GEHC related to the exercise of an option to expand the scope of their original 2009 license agreement to include exclusive global rights to our intellectual property and know-how for the development and sale of cellular assays derived from induced pluripotent stem cells. Upon the closing of the divestiture of our stem cell assets on October 1, 2013, the GEHC agreement, including any future revenue payments thereunder, was transferred to Asterias. No license fee revenue was recognized under the GEHC agreement in 2013. For a further discussion of the divestiture of our stem cell assets, see Note 7 on Divestiture of Stem Cell Assets.
Telomerase Activation Sciences, Inc.
In December 2012, we entered into a Termination and Assignment Agreement, or the Assignment Agreement, with Asia Biotech Corporation, or Asia Biotech, and Telomerase Activation Sciences, Inc., or TA Sciences, pursuant to which we agreed to assign to TA Sciences the intellectual property, including patents previously licensed to Asia Biotech, related to our telomerase activation technology. As consideration for the assignment and fulfillment of the obligations set forth in the Assignment Agreement, we received a non-refundable, upfront payment of $2,500,000 from TA Sciences, which we recognized in full as other income in our consolidated statements of operations for the year ended December 31, 2012, and TA Sciences does not have further payment obligations to us. In addition, Asia Biotech's future royalty obligations under the original license agreement have been terminated.
11. INCOME TAXES
Deferred income taxes reflect the net tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes. Significant components of our deferred tax assets are as follows:
| |
December 31, | ||||||
|---|---|---|---|---|---|---|---|
| |
2014 | 2013 | |||||
| |
(In thousands) |
||||||
Net operating loss carryforwards |
$ | 281,300 | $ | 271,800 | |||
Purchased technology |
— | 6,300 | |||||
Research credits |
23,400 | 22,700 | |||||
Capitalized research and development |
2,100 | 6,600 | |||||
License fees |
500 | 700 | |||||
Other—net |
7,600 | 10,100 | |||||
| | | | | | | | |
Total deferred tax assets |
314,900 | 318,200 | |||||
Valuation allowance for deferred tax assets |
(314,900 | ) | (318,200 | ) | |||
| | | | | | | | |
Net deferred tax assets |
$ | — | $ | — | |||
| | | | | | | | |
| | | | | | | | |
| | | | | | | | |
118
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
11. INCOME TAXES (Continued)
We record net deferred tax assets to the extent we believe these assets will more likely than not be realized. In making such determination, we consider all available positive and negative evidence, including scheduled reversals of deferred tax liabilities, projected future taxable income, tax planning strategies and recent financial performance. Forming a conclusion that a valuation allowance is not required is difficult when there is negative evidence such as cumulative losses in recent years. Because of our history of losses, the net deferred tax assets have been fully offset by a valuation allowance. The valuation allowance decreased by $3,300,000 and $5,500,000 during the years ended December 31, 2014 and 2013, respectively, and increased by $14,200,000 during the year ended December 31, 2012. Approximately $4,900,000 of the valuation allowance for deferred tax assets relates to benefits of stock option deductions which, when recognized, will be allocated directly to contributed capital.
As of December 31, 2014, we had domestic federal net operating loss carryforwards of approximately $774,000,000 expiring at various dates beginning in 2018 through 2034, and state net operating loss carryforwards of approximately $395,000,000 expiring at various dates beginning in 2015 through 2034, if not utilized. We also had federal research and development tax credit carryforwards of approximately $14,900,000 expiring at various dates beginning in 2018 through 2034, if not utilized. Our state research and development tax credit carryforwards of approximately $13,000,000 carry forward indefinitely.
Due to the change of ownership provisions of the Tax Reform Act of 1986, utilization of a portion of our domestic net operating loss and tax credit carryforwards may be limited in future periods. Further, a portion of the carryforwards may expire before being applied to reduce future income tax liabilities.
We adopted the provision of the standard for accounting for uncertainties in income taxes on January 1, 2007. Upon adoption, we recognized no material adjustment in the liability for unrecognized tax benefits. At December 31, 2014, we had approximately $17,100,000 of unrecognized tax benefits, none of which would currently affect our effective tax rate if recognized due to our deferred tax assets being fully offset by a valuation allowance.
A reconciliation of the beginning and ending amounts of unrecognized tax benefits is as follows (in thousands):
Balance as of December 31, 2013 |
$ | 11,600 | ||
Increase related to prior year tax positions |
5,100 | |||
Increase related to current year tax positions |
400 | |||
Settlements |
— | |||
Reductions due to lapse of applicable statute of limitations |
— | |||
| | | | | |
Balance as of December 31, 2014 |
$ | 17,100 | ||
| | | | | |
| | | | | |
| | | | | |
If applicable, we would classify interest and penalties related to uncertain tax positions in income tax expense. Through December 31, 2014, there has been no interest expense or penalties related to unrecognized tax benefits.
We do not currently expect any significant changes to unrecognized tax benefits during the fiscal year ended December 31, 2015. In certain cases, our uncertain tax positions are related to tax years that remain subject to examination by the relevant tax authorities. Tax years for which we have
119
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
11. INCOME TAXES (Continued)
carryforward net operating loss and credit attributes remain subject to examination by federal and most state tax authorities.
12. SEGMENT INFORMATION
Our executive management team represents our chief decision maker. We view our operations as one segment, the discovery and development of therapeutic products for oncology. As a result, the financial information disclosed herein materially represents all of the financial information related to our principal operating segment.
13. CONSOLIDATED STATEMENTS OF CASH FLOWS DATA
| |
Year Ended December 31, | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| |
2014 | 2013 | 2012 | |||||||
| |
(In thousands) |
|||||||||
Supplemental operating activities: |
||||||||||
Issuance of common stock for services rendered to date or to be received in future periods |
$ | — | $ | — | $ | 69 | ||||
Issuance of common stock for 401(k) matching contributions |
$ | 313 | $ | 839 | $ | 1,361 | ||||
Reclassification between deposits and other current assets |
$ | 190 | $ | 219 | $ | 526 | ||||
Supplemental investing activities: |
||||||||||
Net unrealized loss on marketable securities |
$ | (70 | ) | $ | (54 | ) | $ | (38 | ) | |
We have not made any cash payments for taxes or interest for the years ended December 31, 2014, 2013 and 2012.
14. SELECTED QUARTERLY FINANCIAL INFORMATION (UNAUDITED)
| |
First Quarter |
Second Quarter |
Third Quarter |
Fourth Quarter |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
(In thousands, except per share amounts) |
||||||||||||
Year Ended December 31, 2014 |
|||||||||||||
Revenues |
$ | 474 | $ | 341 | $ | 160 | $ | 178 | |||||
Operating expenses |
9,205 | 9,004 | 10,067 | 9,189 | |||||||||
Net loss |
(8,440 | ) | (8,734 | ) | (9,549 | ) | (8,947 | ) | |||||
Basic and diluted net loss per share |
$ | (0.06 | ) | $ | (0.06 | ) | $ | (0.06 | ) | $ | (0.06 | ) | |
Year Ended December 31, 2013 |
|||||||||||||
Revenues |
$ | 765 | $ | 112 | $ | 181 | $ | 225 | |||||
Operating expenses(1) |
12,750 | 9,077 | 8,914 | 9,500 | |||||||||
Net loss |
(11,897 | ) | (8,947 | ) | (8,254 | ) | (9,281 | ) | |||||
Basic and diluted net loss per share |
$ | (0.09 | ) | $ | (0.07 | ) | $ | (0.06 | ) | $ | (0.07 | ) | |
- (1)
- The fourth quarter of 2013 includes approximately $430,000 in restructuring charges in connection with the closure our research laboratory facility located at 200 Constitution Drive, Menlo Park, California. See Note 6 on Restructurings.
120
GERON CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (Continued)
14. SELECTED QUARTERLY FINANCIAL INFORMATION (UNAUDITED) (Continued)
Basic and diluted net losses per share are computed independently for each of the quarters presented. Therefore, the sum of the quarters may not be equal to the full year net loss per share amounts.
15. SUBSEQUENT EVENT
In light of projected reduced operational demands as a result of the Collaboration Agreement with Janssen, on March 3, 2015, we announced an organizational resizing to reduce our workforce from 39 to 21 positions, representing a reduction of approximately 46% of our workforce. We expect the majority of the reduction in our workforce to be completed by the end of the third quarter of 2015. In connection with the resizing, we anticipate incurring aggregate restructuring charges of approximately $1,900,000, of which approximately $1,500,000 is expected be paid in cash during 2015. The aggregate projected restructuring charges represent one-time termination benefits, comprised principally of severance, benefit continuation costs, outplacement services and non-cash stock-based compensation expense associated with the elimination of 18 positions. The majority of these charges are expected to be recognized in the first half of 2015. We may incur other charges and will record these expenses in the appropriate period as they are determined.
121
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING
AND FINANCIAL DISCLOSURE
None.
ITEM 9A. CONTROLS AND PROCEDURES
(I) Evaluation of Disclosure Controls and Procedures
We have carried out an evaluation under the supervision and with the participation of management, including our principal executive officer and principal financial officer, of our disclosure controls and procedures (as defined in Rule 13a-15(e) of the Securities Exchange Act of 1934, as amended) as of the end of the period covered by this annual report on Form 10-K. Based on their evaluation, our principal executive officer and principal financial officer concluded that our disclosure controls and procedures were effective as of December 31, 2014.
It should be noted that any system of controls, however well designed and operated, can provide only reasonable assurance, and not absolute assurance, that the objectives of the system are met. In addition, the design of any control system is based in part upon certain assumptions about the likelihood of future events. Because of these and other inherent limitations of control systems, there can be no assurance that any design will succeed in achieving its stated goals in all future circumstances. Accordingly, our disclosure controls and procedures are designed to provide reasonable, not absolute, assurance that the objectives of our disclosure control system are met and, as set forth above, our principal executive officer and principal financial officer have concluded, based on their evaluation as of the end of the period covered by this report, that our disclosure controls and procedures were effective to provide reasonable assurance that the objectives of our disclosure control system were met.
(II) Changes in Internal Control over Financial Reporting
There was no change in our internal control over financial reporting during the quarter ended December 31, 2014 that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
122
(III) Management's Report on Internal Control over Financial Reporting
Internal control over financial reporting refers to the process designed by, or under the supervision of, our Chief Executive Officer and Chief Financial Officer, and effected by our board of directors, management and other personnel, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles, and includes those policies and procedures that:
- (1)
- Pertain
to the maintenance of records that in reasonable detail accurately and fairly reflect the transactions and dispositions of our assets;
- (2)
- Provide
reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted
accounting principles, and that our receipts and expenditures are being made only in accordance with authorizations of our management and directors; and
- (3)
- Provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of our assets that could have a material effect on the financial statements.
Management is responsible for establishing and maintaining an adequate internal control over financial reporting for us. Internal control over financial reporting cannot provide absolute assurance of achieving financial reporting objectives because of its inherent limitations. Internal control over financial reporting is a process that involves human diligence and compliance and is subject to lapses in judgment and breakdowns resulting from human failures. Internal control over financial reporting also can be circumvented by collusion or improper management override. Because of such limitations, there is a risk that material misstatements may not be prevented or detected on a timely basis by internal control over financial reporting. However, these inherent limitations are known features of the financial reporting process. Therefore, it is possible to design into the process safeguards to reduce, though not eliminate, this risk.
Under the supervision and with the participation of our management, including our principal executive officer and principal financial officer, we conducted an evaluation of the effectiveness of our internal control over financial reporting based on the framework set forth in "Internal Control—Integrated Framework" issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework). Based on our evaluation under the framework set forth in "Internal Control—Integrated Framework," our management concluded that our internal control over financial reporting was effective as of December 31, 2014. The effectiveness of our internal control over financial reporting as of December 31, 2014 has been audited by Ernst & Young LLP, an independent registered public accounting firm, as stated in their report which is included herein.
123
(IV) Report of Independent Registered Public Accounting Firm
The Board of Directors and Stockholders of Geron Corporation
We have audited Geron Corporation's internal control over financial reporting as of December 31, 2014, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework) (the COSO criteria). Geron Corporation's management is responsible for maintaining effective internal control over financial reporting, and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management's Report on Internal Control over Financial Reporting. Our responsibility is to express an opinion on the company's internal control over financial reporting based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
A company's internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company's internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company's assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
In our opinion, Geron Corporation maintained, in all material respects, effective internal control over financial reporting as of December 31, 2014 based on the COSO criteria.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), the consolidated balance sheets of Geron Corporation as of December 31, 2014 and 2013, and the related consolidated statements of operations, comprehensive loss, stockholders' equity and cash flows for each of the three years in the period ended December 31, 2014 of Geron Corporation and our report dated March 11, 2015 expressed an unqualified opinion thereon.
/s/ Ernst & Young LLP
Redwood
City, California
March 11, 2015
None.
124
Certain information required by Part III is omitted from this annual report on Form 10-K because we will file with the U.S. Securities and Exchange Commission a definitive proxy statement pursuant to Regulation 14A in connection with the solicitation of proxies for Geron's Annual Meeting of Stockholders expected to be held in May 2015, or the Proxy Statement, not later than 120 days after the end of the fiscal year covered by this annual report on Form 10-K, and certain information included therein is incorporated herein by reference.
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
Identification of Directors and Nominees for Director
The information required by this item concerning our directors and nominees for director is incorporated by reference from the section captioned "Proposal 1: Election of Directors" contained in our Proxy Statement.
Identification of Executive Officers
The information required by this item concerning our executive officers is set forth in Part I, Item 1 of this annual report on Form 10-K.
Code of Ethics
We have adopted a Code of Conduct with which every person who works for Geron, including our board of directors, is expected to comply. The Code of Conduct is publicly available on our website under the Investor Relations section at www.geron.com. This website address is intended to be an inactive, textual reference only; none of the material on this website is part of this annual report on Form 10-K. If any substantive amendments are made to the Code of Conduct or any waiver granted, including any implicit waiver, from a provision of the Code to our Chief Executive Officer, Chief Financial Officer or Corporate Controller, we will disclose the nature of such amendment or waiver on that website or in a report on Form 8-K.
Copies of the Code of Conduct will be furnished without charge to any person who submits a written request directed to the attention of our Corporate Secretary, at our offices located at 149 Commonwealth Drive, Suite 2070, Menlo Park, California, 94025.
Section 16(a) Compliance
Information concerning Section 16(a) beneficial ownership reporting compliance is incorporated by reference from the section captioned "Section 16(a) Beneficial Ownership Reporting Compliance" contained in the Proxy Statement.
Certain Corporate Governance Matters
The information required by this item concerning our audit committee, audit committee financial expert and procedures by which stockholders may recommend nominees to our board of directors, may be found under the section captioned "Corporate Governance Matters" contained in the Proxy Statement.
ITEM 11. EXECUTIVE COMPENSATION
The information required by this item is incorporated by reference from the sections captioned "Compensation Discussion and Analysis," "Compensation Committee Report," "Executive Compensation Tables," "Compensation of Directors" and "Compensation Committee Interlocks and Insider Participation" contained in the Proxy Statement.
125
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
AND RELATED STOCKHOLDER MATTERS
The information required by this item is incorporated by reference from the sections captioned "Equity Compensation Plans" and "Security Ownership of Certain Beneficial Owners and Management" contained in the Proxy Statement.
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND
DIRECTOR INDEPENDENCE
The information required by this item is incorporated by reference from the sections captioned "Proposal 1: Election of Directors" and "Certain Transactions" contained in the Proxy Statement.
ITEM 14. PRINCIPAL ACCOUNTING FEES AND SERVICES
The information required by this item is incorporated by reference from the section captioned "Principal Accountant Fees and Services" contained in the Proxy Statement.
ITEM 15. EXHIBITS, FINANCIAL STATEMENT SCHEDULES
- (a)
- (1) Consolidated Financial Statements
Included in Part II, Item 8 of this Report:
- (2)
- Financial Statement Schedules
Financial statement schedules are omitted because they are not required or the information is disclosed in the financial statements listed in Item 15(a)(1) above.
- (3)
- Exhibits
See Exhibit Index.
126
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| GERON CORPORATION | ||||
Date: March 11, 2015 |
By: |
/s/ OLIVIA K. BLOOM OLIVIA K. BLOOM Executive Vice President, Finance, Chief Financial Officer and Treasurer |
||
127
KNOW BY ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints, jointly and severally, John A. Scarlett, M.D., and Olivia K. Bloom, and each one of them, attorneys-in-fact for the undersigned, each with the power of substitution, for the undersigned in any and all capacities, to sign any and all amendments to this annual report on Form 10-K, and to file the same, with exhibits thereto and other documents in connection therewith, with the Securities and Exchange Commission, hereby ratifying and confirming all that each of said attorneys-in-fact, or his or her substitutes, may do or cause to be done by virtue hereof.
IN WITNESS WHEREOF, each of the undersigned has executed this Power of Attorney as of the date indicated opposite his/her name.
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the Registrant and in the capacities and on the dates indicated.
Signature
|
Title
|
Date
|
||
|---|---|---|---|---|
| /s/ JOHN A. SCARLETT JOHN A. SCARLETT |
President, Chief Executive Officer and Director (Principal Executive Officer) | March 11, 2015 | ||
/s/ OLIVIA K. BLOOM OLIVIA K. BLOOM |
Executive Vice President, Finance, Chief Financial Officer and Treasurer (Principal Financial and Accounting Officer) |
March 11, 2015 |
||
/s/ DANIEL M. BRADBURY DANIEL M. BRADBURY |
Director |
March 11, 2015 |
||
/s/ KARIN EASTHAM KARIN EASTHAM |
Director |
March 11, 2015 |
||
/s/ THOMAS HOFSTAETTER THOMAS HOFSTAETTER |
Director |
March 11, 2015 |
||
/s/ HOYOUNG HUH HOYOUNG HUH |
Director |
March 11, 2015 |
||
/s/ V. BRYAN LAWLIS V. BRYAN LAWLIS |
Director |
March 11, 2015 |
||
/s/ SUSAN M. MOLINEAUX SUSAN M. MOLINEAUX |
Director |
March 11, 2015 |
||
/s/ ROBERT J. SPIEGEL ROBERT J. SPIEGEL |
Director |
March 11, 2015 |
128
| |
|
Incorporation by Reference | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Exhibit Number |
Description |
Exhibit Number |
Filing |
Filing Date |
File No. |
||||||
2.1 |
Asset Contribution Agreement by and among Geron Corporation, BioTime, Inc. and Asterias Biotherapeutics, Inc. (formerly known as BioTime Acquisition Corporation) | 2.1 | 8-K | January 8, 2013 | 000-20859 | ||||||
3.1 |
Restated Certificate of Incorporation | 3.3 | 8-K | May 18, 2012 | 000-20859 | ||||||
3.2 |
Certificate of Amendment of the Restated Certificate of Incorporation | 3.1 | 8-K | May 18, 2012 | 000-20859 | ||||||
3.3 |
Amended and Restated Bylaws of Registrant | 3.1 | 8-K | March 19, 2010 | 000-20859 | ||||||
4.1 |
Form of Common Stock Certificate | 4.1 | 10-K | March 15, 2013 | 000-20859 | ||||||
4.2 |
Form of 2005 Warrant | 4.2 | 8-K | April 25, 2005 | 000-20859 | ||||||
4.3 |
Form of 2011 Warrant | Attachment to 10.1 | 10-Q | November 3, 2011 | 000-20859 | ||||||
10.1 |
Form of Indemnification Agreement | 10.1 | 10-K | March 7, 2012 | 000-20859 | ||||||
10.2 |
Amended and Restated 1996 Employee Stock Purchase Plan* | 10.2 | 10-Q | July 31, 2009 | 000-20859 | ||||||
10.3 |
1996 Directors' Stock Option Plan, as amended* | Appendix B | Def 14A | April 15, 2003 | 000-20859 | ||||||
10.4 |
Amended and Restated 2002 Equity Incentive Plan* | 4.1 | S-8 | June 4, 2010 | 333-167349 | ||||||
10.5 |
Form of Stock Option Agreement under 2002 Equity Incentive Plan* | 10.6 | 10-K | March 15, 2013 | 000-20859 | ||||||
10.6 |
Form of Restricted Stock Award Agreement under 2002 Equity Incentive Plan* | 10.7 | 10-K | March 15, 2013 | 000-20859 | ||||||
10.7 |
Amended and Restated 2006 Directors' Stock Option Plan* | 10.5 | 10-Q | November 7, 2013 | 000-20859 | ||||||
10.8 |
2011 Incentive Award Plan* | 10.1 | 8-K | May 16, 2011 | 000-20859 | ||||||
10.9 |
Form of Stock Option Agreement under 2011 Incentive Award Plan* | 10.11 | 10-K | March 15, 2013 | 000-20859 | ||||||
10.10 |
Form of Restricted Stock Award Agreement under 2011 Incentive Award Plan* | 10.12 | 10-K | March 15, 2013 | 000-20859 | ||||||
10.11 |
Form of Non-Employee Director Stock Option Agreement under 2011 Incentive Award Plan* | 10.37 | 10-K | March 17, 2014 | 000-20859 | ||||||
| |
|
Incorporation by Reference | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Exhibit Number |
Description |
Exhibit Number |
Filing |
Filing Date |
File No. |
||||||
10.12 |
2014 Employee Stock Purchase Plan* | 10.1 | 8-K | May 23, 2014 | 000-20859 | ||||||
10.13 |
Stock Purchase Agreement between the Registrant and Angiochem, Inc., effective as of January 5, 2011 | 10.1 | 8-K | January 7, 2011 | 000-20859 | ||||||
10.14 |
† | California Institute for Regenerative Medicine Notice of Loan Award | 10.1 | 10-Q | November 3, 2011 | 000-20859 | |||||
10.15 |
Amended and Restated Severance Plan, effective as of May 23, 2013* | 10.1 | 8-K | May 24, 2013 | 000-20859 | ||||||
10.16 |
Employment agreement between the Registrant and John A. Scarlett, M.D., effective as of September 29, 2011* | 10.2 | 10-Q | November 3, 2011 | 000-20859 | ||||||
10.17 |
First Amendment to Employment Agreement between the Registrant and John A. Scarlett, M.D., effective as of February 11, 2014* | 10.5 | 8-K | February 14, 2014 | 000-20859 | ||||||
10.18 |
Employment agreement between the Registrant and Stephen N. Rosenfield, effective as of February 16, 2012* | 10.32 | 10-K | March 7, 2012 | 000-20859 | ||||||
10.19 |
First Amendment to Employment Agreement between the Registrant and Stephen N. Rosenfield, effective as of September 24, 2013* | 10.4 | 8-K | September 27, 2013 | 000-20859 | ||||||
10.20 |
Employment agreement between the Registrant and Andrew J. Grethlein, effective as of September 17, 2012* | 10.2 | 10-Q | November 2, 2012 | 000-20859 | ||||||
10.21 |
First Amendment to Employment Agreement between the Registrant and Andrew J. Grethlein, effective as of February 11, 2014* | 10.4 | 8-K | February 14, 2014 | 000-20859 | ||||||
10.22 |
Employment agreement between the Registrant and Craig C. Parker, effective as of December 3, 2012* | 10.25 | 10-K | March 15, 2013 | 000-20859 | ||||||
10.23 |
First Amendment to Employment Agreement between the Registrant and Craig C. Parker, effective as of September 24, 2013* | 10.3 | 8-K | September 27, 2013 | 000-20859 | ||||||
10.24 |
Second Amendment to Employment Agreement between the Registrant and Craig C. Parker, effective as of February 11, 2014* | 10.3 | 8-K | February 14, 2014 | 000-20859 | ||||||
| |
|
Incorporation by Reference | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Exhibit Number |
Description |
Exhibit Number |
Filing |
Filing Date |
File No. |
||||||
10.25 |
Employment agreement between the Registrant and Olivia K. Bloom, effective as of December 7, 2012* | 10.26 | 10-K | March 15, 2013 | 000-20859 | ||||||
10.26 |
First Amendment to Employment Agreement between the Registrant and Olivia K. Bloom, effective as of September 24, 2013* | 10.2 | 8-K | September 27, 2013 | 000-20859 | ||||||
10.27 |
Second Amendment to Employment Agreement between the Registrant and Olivia K. Bloom, effective as of February 11, 2014* | 10.1 | 8-K | February 14, 2014 | 000-20859 | ||||||
10.28 |
Employment agreement between the Registrant and Melissa A. Kelly Behrs, effective as of January 31, 2013* | 10.28 | 10-K | March 15, 2013 | 000-20859 | ||||||
10.29 |
First Amendment to Employment Agreement between the Registrant and Melissa A. Kelly Behrs, effective as of September 24, 2013* | 10.1 | 8-K | September 27, 2013 | 000-20859 | ||||||
10.30 |
Second Amendment to Employment Agreement between the Registrant and Melissa A. Kelly Behrs, effective as of February 11, 2014* | 10.2 | 8-K | February 14, 2014 | 000-20859 | ||||||
10.31 |
† | Office Lease Agreement by and between the Registrant and Exponent Realty, LLC, effective as of February 29, 2012 | 10.36 | 10-K/A | March 27, 2012 | 000-20859 | |||||
10.32 |
Third Amendment to Office Lease Agreement by and between the Registrant and Exponent Realty, LLC, effective as of February 27, 2014 | 10.1 | 8-K | March 4, 2014 | 000-20859 | ||||||
10.33 |
At-the-Market Issuance Sales Agreement, dated October 8, 2012, by and between the Registrant and MLV & Co. LLC | 10.1 | 8-K | October 9, 2012 | 000-20859 | ||||||
10.34 |
Transfer Agreement by and between Registrant and Mayo Clinic, effective July 31, 2014 | 10.1 | 10-Q | November 5, 2014 | 000-20859 | ||||||
10.35 |
Non-Employee Director Compensation Policy, as amended* | ||||||||||
10.36 |
†† | Collaboration and License Agreement by and between the Registrant and Janssen Biotech, Inc., dated November 13, 2014 | |||||||||
| |
|
Incorporation by Reference | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Exhibit Number |
Description |
Exhibit Number |
Filing |
Filing Date |
File No. |
||||||
12.1 |
Computation of Ratio of Earnings to Fixed Charges | ||||||||||
23.1 |
Consent of Independent Registered Public Accounting Firm | ||||||||||
24.1 |
Power of Attorney (see signature page) | ||||||||||
31.1 |
Certification of Chief Executive Officer pursuant to Form of Rule 13a-14(a), as Adopted Pursuant to Section 302(a) of the Sarbanes-Oxley Act of 2002, dated March 11, 2015 | ||||||||||
31.2 |
Certification of Chief Financial Officer pursuant to Form of Rule 13a-14(a), as Adopted Pursuant to Section 302(a) of the Sarbanes-Oxley Act of 2002, dated March 11, 2015 | ||||||||||
32.1 |
Certification of Chief Executive Officer pursuant to 18 U.S.C. Section 1350, as Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, dated March 11, 2015** | ||||||||||
32.2 |
Certification of Chief Financial Officer pursuant to 18 U.S.C. Section 1350, as Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, dated March 11, 2015** | ||||||||||
101 |
The following materials from the Registrant's annual report on Form 10-K for the year ended December 31, 2014, formatted in Extensible Business Reporting Language (XBRL) include: (i) Consolidated Balance Sheets as of December 31, 2014 and 2013, (ii) Consolidated Statements of Operations, Comprehensive Loss, Stockholders' Equity and Cash Flows for each of the three years in the period ended December 31, 2014, and (iii) Notes to Consolidated Financial Statements. | ||||||||||
- †
- Confidential
treatment has been granted for certain portions of this exhibit. Omitted information has been filed separately with the Securities
and Exchange Commission.
- ††
- Confidential
treatment has been requested for certain portions of this exhibit. Omitted information has been filed separately with
the Securities and Exchange Commission.
- *
- Management contract or compensation plan or arrangement.
- **
- The certifications attached as Exhibits 32.1 and 32.2 that accompany this annual report on Form 10-K, are not deemed filed with the Securities and Exchange Commission and are not to be incorporated by reference into any filing of the Company under the Securities Act of 1933, as amended, or the Securities Exchange Act of 1934, as amended (whether made before or after the date of this Form 10-K), irrespective of any general incorporation language contained in such filing.
