Attached files
| file | filename |
|---|---|
| EXCEL - IDEA: XBRL DOCUMENT - Protalix BioTherapeutics, Inc. | Financial_Report.xls |
| EX-32.1 - EXHIBIT 32.1 - Protalix BioTherapeutics, Inc. | v303493_ex32-1.htm |
| EX-23.1 - EXHIBIT 23.1 - Protalix BioTherapeutics, Inc. | v303493_ex23-1.htm |
| EX-31.1 - EXHIBIT 31.1 - Protalix BioTherapeutics, Inc. | v303493_ex31-1.htm |
| EX-31.2 - EXHIBIT 31.2 - Protalix BioTherapeutics, Inc. | v303493_ex31-2.htm |
| EX-32.2 - EXHIBIT 32.2 - Protalix BioTherapeutics, Inc. | v303493_ex32-2.htm |
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
FOR ANNUAL AND TRANSITION REPORTS PURSUANT TO SECTIONS 13 OR 15(d)
OF THE SECURITIES EXCHANGE ACT OF 1934
(Mark One)
| x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2011
OR
| ¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
001-33357
(Commission file number)
PROTALIX BIOTHERAPEUTICS, INC.
(Exact name of registrant as specified in its charter)
| Florida | 65-0643773 | |
|
State or other jurisdiction of incorporation or organization |
(I.R.S. Employer Identification No.) | |
|
2 Snunit Street Science Park POB 455 Carmiel, Israel |
20100
| |
| (Address of principal executive offices) | (Zip Code) |
972-4-988-9488
Registrant’s telephone number, including area code
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Name of each exchange on which registered | |
| Common stock, par value $0.001 per share | NYSE AMEX |
Securities registered pursuant to Section 12(g) of the Act:
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ¨ No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ¨ No x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No ¨
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes x No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. þ
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See definition of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act. (check one):
| Large accelerated filer | ¨ | Accelerated filer | x | |
| Non-accelerated filer | ¨ | (Do not check if a smaller reporting company) | Smaller reporting company | ¨ |
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ¨ No x
The aggregate market value of the voting common equity held by non-affiliates of the Registrant, as of June 30, 2011 was approximately $365 million (based upon a per share price equal to $6.26, the closing price for shares of the Registrant’s common stock reported by the NYSE Amex for such date). Shares of common stock held by each officer, director and holder of 5% or more of the outstanding common stock have been excluded in that such persons may be deemed to be affiliates. This determination of affiliate status is not necessarily a conclusive determination for other purposes.
On February 23, 2012, approximately 90,815,396 shares of the Registrant’s common stock, par value $0.001 per share, were outstanding.
FORM 10-K
TABLE OF CONTENTS
| Page | ||
| PART I | ||
| Cautionary Statement Regarding Forward-Looking Statements | 1 | |
| Item 1. | Business | 3 |
| Item 1A. | Risk Factors | 38 |
| Item 1B. | Unresolved Staff Comments | 59 |
| Item 2. | Properties | 59 |
| Item 3. | Legal Proceedings | 59 |
| Item 4. | Mine Safety Disclosures | 59 |
| PART II | ||
| Item 5. | Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities | 60 |
| Item 6. | Selected Financial Data | 62 |
| Item 7. | Management’s Discussion and Analysis of Financial Condition and Results of Operations | 63 |
| Item 7A. | Quantitative and Qualitative Disclosures About Market Risk | 73 |
| Item 8. | Financial Statements and Supplementary Data | 74 |
| Item 9. | Changes in and Disagreements with Accountants on Accounting and Financial Disclosure | 74 |
| Item 9A. | Controls and Procedures | 74 |
| Item 9B. | Other Information | 75 |
| PART III | ||
| Item 10. | Directors, Executive Officers and Corporate Governance | 76 |
| Item 11. | Executive Compensation | 79 |
| Item 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters | 89 |
| Item 13. | Certain Relationships and Related Transactions, and Director Independence | 91 |
| Item 14. | Principal Accountant Fees and Services | 93 |
| PART IV | ||
| Item 15. | Exhibits and Financial Statement Schedules | 94 |
| Signatures | 98 | |
| i |
PART I
Except where the context otherwise requires, the terms, “we,” “us,” “our” or “the Company,” refer to the business of Protalix BioTherapeutics, Inc. and its consolidated subsidiaries, and “Protalix” or “Protalix Ltd.” refers to the business of Protalix Ltd., our wholly-owned subsidiary and sole operating unit.
CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS
The statements set forth under the captions “Business,” “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and “Risk Factors,” and other statements included elsewhere in this Annual Report on Form 10-K, which are not historical, constitute “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933, as amended, or the Securities Act, and Section 21E of the Securities Exchange Act of 1934, as amended, or the Exchange Act, including statements regarding expectations, beliefs, intentions or strategies for the future. When used in this report, the terms “anticipate,” “believe,” “estimate,” “expect” and “intend” and words or phrases of similar import, as they relate to our company or our subsidiaries or our management, are intended to identify forward-looking statements. We intend that all forward-looking statements be subject to the safe-harbor provisions of the Private Securities Litigation Reform Act of 1995. These forward-looking statements are only predictions and reflect our views as of the date they are made with respect to future events and financial performance, and we undertake no obligation to update or revise, nor do we have a policy of updating or revising, any forward-looking statement to reflect events or circumstances after the date on which the statement is made or to reflect the occurrence of unanticipated events, except as may be required under applicable law. Forward-looking statements are subject to many risks and uncertainties that could cause our actual results to differ materially from any future results expressed or implied by the forward-looking statements.
Examples of the risks and uncertainties include, but are not limited to, the following:
| · | delays in the review by the U.S. Food and Drug Administration, or FDA, of our response to the Complete Response Letter, or CRL, we received from the FDA relating to our New Drug Application (NDA) for taliglucerase alfa; |
| · | delays in the approval or the potential rejection of any application we file with the FDA, or any other regulatory authority, including the NDA we filed with the FDA, the marketing application we submitted to the Israeli Ministry of Health, or the Israeli MOH, and the Marketing Authorization Application (MAA) we submitted to each of the European Medicines Agency, or the EMA, the National Sanitary Vigilance Agency, or ANVISA, an agency of the Ministry of Health of Brazil, and the Australian Therapeutic Goods Administration, or the Australian TGA, for taliglucerase alfa; |
| · | risks relating to our ability to finance our research programs, the expansion of our manufacturing capabilities and the ongoing costs in the case of delays in regulatory approvals for taliglucerase alfa; |
| · | risks relating to potential restrictions on the marketing and sale of certain of our product candidates in certain territories due to the orphan drug status that may be granted to competing products; |
| · | the impact of development of competing therapies and/or technologies by other companies; |
| · | the effect of the orphan drug designation granted by the EMA/European Commission to VPRIV® in the European Union on the marketing of taliglucerase alfa in the European Union; |
| · | the availability of reimbursement to patients from health care payors for taliglucerase alfa or any of our other product candidates, if approved; |
| · | delays in our preparation and filing of applications for regulatory approval in the United States, the European Union, Israel, Brazil, Australia and elsewhere; |
| · | any lack of progress of our research and development (including the results of our clinical trials); |
| · | our ability to establish and maintain strategic license, collaboration and distribution arrangements, and to manage our relationships with Pfizer Inc., or Pfizer, Teva Ltd., or Teva, or with any other collaborator, distributor or partner; |
| 1 |
| · | our ability to obtain, on a timely basis, if at all, sufficient patient enrollment in our clinical trials; |
| · | risks relating to biosimilar legislation and/or healthcare reform in the United States or elsewhere; |
| · | the inherent risks and uncertainties in developing the types of drug platforms and products we are developing; |
| · | the risk that we will not be able to develop a successful sales and marketing organization for taliglucerase alfa in Israel or for any other product candidate in a timely manner, if at all; |
| · | our ability to enter into supply arrangements with the Ministry of Health of Brazil or other parties and to supply drug product pursuant to such arrangements; |
| · | potential product liability risks, and risks of securing adequate levels of product liability and clinical trial insurance coverage; |
| · | the possibility of infringing a third party’s patents or other intellectual property rights; |
| · | the uncertainty of obtaining patents covering our products and processes and in successfully enforcing our intellectual property rights against third parties; |
| · | the possible disruption of our operations due to terrorist activities and armed conflict, including as a result of the disruption of the operations of regulatory authorities, our subsidiaries, our manufacturing facilities and our customers, suppliers, distributors, collaborative partners, licensees and clinical trial sites; and |
| · | other risks and uncertainties detailed in Section 1A of this Annual Report on Form 10-K. |
Companies in the pharmaceutical and biotechnology industries have suffered significant setbacks in advanced or late-stage clinical trials, even after obtaining promising earlier trial results or preliminary findings for such clinical trials. Even if favorable testing data is generated from clinical trials of a drug product, the FDA or foreign regulatory authorities may not accept or approve an NDA or MAA, as applicable, filed by a pharmaceutical or biotechnology company for the drug product. These and other risks and uncertainties are detailed under the heading “Risk Factors” in this Annual Report on Form 10-K and are described from time to time in the reports we file with the Securities and Exchange Commission, or the Commission.
| 2 |
| Item 1. | Business |
We are a biopharmaceutical company focused on the development and commercialization of recombinant therapeutic proteins based on our proprietary ProCellEx® protein expression system, or ProCellEx. Using our ProCellEx system, we are developing a pipeline of proprietary and biosimilar versions of recombinant therapeutic proteins based on our plant cell-based expression technology that target large, established pharmaceutical markets and that rely upon known biological mechanisms of action. Our initial commercial focus has been on complex therapeutic proteins, including proteins for the treatment of genetic disorders, such as Gaucher disease and Fabry disease. We believe our ProCellEx protein expression system will enable us to develop proprietary recombinant proteins that are therapeutically equivalent or superior to existing recombinant proteins currently marketed for the same indications. Because we are primarily targeting biologically equivalent versions of highly active, well-tolerated and commercially successful therapeutic proteins, we believe our development process is associated with relatively less risk compared to other biopharmaceutical development processes for completely novel therapeutic proteins.
Taliglucerase alfa, our proprietary, lead product candidate, is a recombinant form of glucocerebrosidase (GCD) that we are developing for the treatment of Gaucher disease using ProCellEx. Gaucher disease is a rare and serious lysosomal storage disorder with severe and debilitating symptoms. Gaucher disease patients suffer from mutations in or deficiencies of GCD, which is an enzyme that is naturally found in human cells. The U.S. Food and Drug Administration, or the FDA, has granted taliglucerase alfa a Prescription Drug User Fee Act (PDUFA) date of May 1, 2012. We first filed a New Drug Application, or NDA, for taliglucerase alfa with the FDA on December 9, 2009, and on February 25, 2011, the FDA issued a complete response letter, or CRL, indicating that the FDA's review was complete but questions remain that preclude the approval of the NDA in its then-current form. The main questions raised by the FDA regarding the NDA related to the clinical and chemistry, manufacturing and controls (CMC) sections. In the clinical section of the CRL, the FDA requested additional data from our ongoing switchover trial and our long-term extension trial. At the time the NDA was submitted, full data from these trials was not available. In the CMC section of the CRL, the FDA requested information regarding testing specifications and assay validation. The FDA did not request additional clinical studies in the CRL. We met with the FDA in May 2011 to clarify the FDA's requests, and in August 2011, we submitted our reply to the CRL. In August 2011, the FDA notified us that it had accepted for review the resubmission of the taliglucerase alfa NDA, had deemed the resubmission a class 2, or 6-month, response and established February 1, 2012 as the new PDUFA date. In December 2011, the FDA notified us that it had decided to extend the PDUFA date to May 1, 2012. In its notification, the FDA stated that its decision related to certain clinical information regarding taliglucerase alfa we had submitted in November 2011 in response to an FDA request. The requested information related mainly to the presentation of select data provided in the NDA. As this information was requested and provided within 90 days of the February 1, 2012 PDUFA goal date, the FDA had the option to extend the PDUFA goal date to provide adequate time for the FDA to complete its review. A three-month extension cycle is the standard period granted. No additional data was requested by the FDA in the notification, nor were we notified of any specific deficiency in the taliglucerase alfa NDA.
In the fourth quarter of 2010, Pfizer, our commercialization partner, filed, with our cooperation, a Marketing Authorization Application, or MAA, for taliglucerase alfa with the European Medicines Agency, or EMA. As part of its ongoing review of the MAA, the EMA delivered a list of outstanding points to be addressed by the applicant. Among the topics currently in discussion, is the orphan drug designation and exclusivity granted by the EMA/European Commission to VPRIV, Shire plc's, or Shire's, Gaucher disease treatment, which could prevent the marketing authorization of taliglucerase alfa in the European Union for a 10-year market exclusivity period commencing as of the August 2010 marketing authorization of VPRIV in the European Union. As part of the MAA procedure, Pfizer, with our cooperation, is challenging VPRIV's orphan market protection with respect to taliglucerase alfa pursuant to the EU orphan drug regulation. The EU orphan drug regulation provides for the possibility of such a challenge, and for an exception to this exclusivity to be granted, based on a number of factors, including contribution to patient care, clinical, supply, capacity and others.
Following the submission of the taliglucerase alfa MAA to the EMA, a good manufacturing practices, or GMP, audit of our manufacturing facility was performed and it was found compliant with EMA GMP in December 2011.
In January 2010, the EMA's Committee for Orphan Medicinal Products, or COMP, after reviewing all relevant clinical data, recommended that the European Commission grant orphan drug designation to taliglucerase alfa for the treatment of Gaucher disease and in March 2010, the European Commission granted orphan designation to taliglucerase alfa.
In addition, marketing applications have been submitted for taliglucerase alfa with each of the Israeli Ministry of Health, or Israeli MOH, the National Sanitary Vigilance Agency, or ANVISA, an agency of the Ministry of Health of Brazil, and the Australian Therapeutic Goods Administration, or Australian TGA.
| 3 |
In February 2010, the Israeli MOH completed a successful GMP audit of our manufacturing facilities in Carmiel, Israel. The audit was performed as part of the Israeli MOH's evaluation of our manufacturing process for taliglucerase alfa. On February 20, 2011, we received a letter from the FDA notifying us that the FDA had completed its review of the Establishment Inspection Report in connection with the FDA's inspection of our facility in Carmiel, Israel, and that the FDA had classified our facility as acceptable. In June 2011, ANVISA completed a successful GMP audit of the facility and determined that the facility is acceptable. In December 2011, following the submission of the taliglucerase alfa MAA to the EMA, the Irish Medicines Board, or IMB, completed a successful GMP audit of our facility and issued a Certificate of GMP Compliance of a Manufacturer for the facility. The IMB certificate is accepted by all health authorities in the European Union under the European Union's centralized marketing authorization procedure, and by authorities of several other countries that recognize EU certification.
In addition to the completed phase III clinical trial we completed in September 2009, we initiated a double-blind, follow-on extension study as part of the trial during the second quarter of 2008. Eligible patients who completed nine months of treatment in our phase III clinical trial were offered the opportunity to participate in the extension study and continue to receive taliglucerase alfa at the same dose they received in the phase III clinical trial for an additional 15 months in a blinded manner. In February 2012, we announced data from 26 adult patients enrolled in the trial. According to the data, after 24 months, patients continued to demonstrate statistically significant improvement in all parameters with a similar safety profile as seen in the phase III clinical trial. Furthermore, those patients who were followed specifically for their bone parameters using Quantitative Chemical Shift Imaging (QCSI) MRI continued to show bone marrow improvement over time. We have also initiated a home care treatment program for patients enrolled in the extension study.
In the second quarter of 2011, we successfully completed a nine-month, worldwide, multi-center, open-label, switch-over clinical study evaluating the safety and efficacy of switching Gaucher patients currently treated with Cerezyme®, which is produced by Genzyme Corporation, or Genzyme (which was acquired by Sanofi-Aventis, or Sanofi, in April 2011), with taliglucerase alfa. The results of the switchover trial demonstrate that over the nine-month treatment period, patients remained stable with regard to spleen volume, liver volume, platelet count and hemoglobin concentration, the efficacy endpoints of the switchover trial, after switching from Cerezyme to taliglucerase alfa. The safety analysis of the switchover trial demonstrated that taliglucerase alfa was well tolerated, and no drug-related serious adverse events were reported. In December 2009, we filed a proposed pediatric investigation plan to the Pediatric Committee of the EMA, which was approved during the first quarter of 2010. We have since initiated the pediatric study and have enrolled all of the naïve pediatric patients required according to the study protocol. All of the enrolled patients are in the final stages of pediatric patient treatment. Patients in the extension trial and the switchover trial are still being treated with taliglucerase alfa.
The current standard of care for Gaucher patients is enzyme replacement therapy with either Cerezyme or, since its U.S. approval in February 2010, VPRIV. Enzyme replacement therapy is a medical treatment in which recombinant enzymes are infused into patients in whom the enzyme is lacking or dysfunctional. Taliglucerase alfa has an amino acid, glycan and three-dimensional structure that is very similar to Cerezyme, which is a mammalian cell expressed version of the same protein. We believe taliglucerase alfa may prove more cost-effective than the currently marketed alternatives due to the cost benefits of expression through our ProCellEx protein expression system. In July 2007, we reached an agreement with the FDA on the final design of our pivotal phase III clinical trial of taliglucerase alfa through the FDA's special protocol assessment (SPA) process. The FDA did not originally require our switch-over study in the SPA as a prerequisite for approval of taliglucerase alfa. However, the FDA requested data from the switchover trial in the CRL which had not been available when the NDA was initially filed.
On November 30, 2009, Protalix Ltd., our wholly-owned subsidiary, and Pfizer entered into an exclusive license and supply agreement, which we refer to as the Pfizer Agreement, pursuant to which Pfizer was granted an exclusive, worldwide license to develop and commercialize taliglucerase alfa. Under the terms and conditions of the Pfizer Agreement, Protalix Ltd. retained the right to commercialize taliglucerase alfa in Israel. In connection with the execution of the Pfizer Agreement, Pfizer made an upfront payment to Protalix Ltd. of $60.0 million in connection with the execution of the agreement and subsequently paid Protalix Ltd. an additional $5.0 million upon its filing of a proposed pediatric investigation plan to the Pediatric Committee of the EMA. Protalix Ltd. is also eligible to receive potential milestone payments totaling $50.0 million for the successful achievement of other regulatory milestones. According to the Pfizer Agreement, Pfizer and Protalix Ltd. are required to share future revenues and expenses for the development and commercialization of taliglucerase alfa on a 60% and 40% basis, respectively, and have agreed to a specific allocation of the responsibilities for the continued development efforts for taliglucerase alfa.
| 4 |
Although taliglucerase alfa has not yet been approved for commercial sale, patients are being treated with taliglucerase alfa, both in the framework of our clinical trials and related studies and in compassionate use programs, special access agreements, named patient provisions and other programs designed to ensure that treatments are available to Gaucher patients in light of recent shortages of approved treatments. In July 2009, following a request by the FDA, we submitted a treatment protocol to the FDA in order to address an expected shortage of the current enzyme replacement therapy approved for Gaucher disease. The treatment protocol was approved by the FDA in August 2009, and we are continuing to treat patients in the United States under this protocol. On July 13, 2010, we announced that the French regulatory authority had granted an Autorisation Temporaire d'Utilisation (ATU), or Temporary Authorization for Use, for taliglucerase alfa for the treatment of Gaucher disease. An ATU is the regulatory mechanism used by the French Health Products and Safety Agency to make non-approved drugs available to patients in France when a genuine public health need exists. This ATU allows Gaucher disease patients in France to receive treatment with taliglucerase alfa before marketing authorization for the product is granted in the European Union. Payment for taliglucerase alfa has been secured through government allocations to hospitals. In addition to the United States and France, taliglucerase alfa is currently being provided to Gaucher disease patients under special access agreements or Named Patient provisions in Brazil and in other countries. Hundreds of patients, in the aggregate, have been treated with taliglucerase alfa.
On August 10, 2010, Pfizer entered into a $30 million short-term supply agreement with the Ministry of Health of Brazil pursuant to which Protalix and Pfizer have provided taliglucerase alfa to the Ministry of Health of Brazil for the treatment of patients with Gaucher disease. During the first quarter of 2011, we and Pfizer supplied the remaining products deliverable under the short-term supply agreement. Revenue generated from the Ministry of Health of Brazil was recorded by Pfizer, and we recorded our share of the revenue in accordance with the terms and conditions of the Pfizer Agreement. During the second quarter of 2011, the Ministry of Health of Brazil requested that Pfizer consider the replacement of certain vials that might expire during 2012. As a result, Pfizer recorded an allowance for sales return. If and when such vials are replaced, revenues will be recorded upon the supply of such replaced vials, and if it is determined that there is no longer a need for the replacement of vials, the allowance will be reversed and the revenues will be recognized accordingly. In addition, we and the Ministry of Health of Brazil are in discussions relating to a possible long-term supply agreement that contemplates, among other matters, providing certain components of our manufacturing technology to the Ministry of Health of Brazil for implementation by it in Brazil. We are currently unable to assess whether these discussions will result in an agreement and we can make no assurance that we will be able to enter into such an agreement on favorable terms, if at all. In any event, we do not expect to enter into a long-term supply agreement with the Ministry of Health of Brazil until we receive marketing approval of taliglucerase alfa from the FDA or ANVISA, if at all.
In September 2009, the FDA’s Office of Orphan Product Development granted taliglucerase alfa Orphan Drug Status. The Orphan Drug designation in the United States for taliglucerase alfa for the treatment of Gaucher disease provides special status to taliglucerase alfa provided that it meets certain criteria. As a result of the Orphan Drug designation, we are qualified for the tax credit and marketing incentives of the Orphan Drug Act of 1983. A marketing application for a prescription drug product that has been designated as a drug for a rare disease or condition is not subject to a prescription drug user fee unless the application includes an indication for other than a rare disease or condition.
Although Gaucher disease is a relatively rare disease, it represents a large commercial market due to the severity of the symptoms and the chronic nature of the disease. According to public reports by Sanofi, consolidated sales of Cerezyme from April 1, 2011 through the end of the year were €441.0 million (or $579.6 million), a growth of 11.1% compared to the same period in 2010. According to Genzyme, it has suffered interruptions and other limitations in its Cerezyme production and release since September 2009 and, as a result, Cerezyme supplies have been limited. Shire reported annual worldwide sales of VPRIV of approximately $256 million in 2011, a growth of 79% compared to the same period in 2010 (VPRIV was approved for sale in the United States in February 2010 and in the European Union in August 2010).
In addition to taliglucerase alfa, we are developing an innovative product pipeline using our ProCellEx protein expression system. Our product pipeline currently includes, among other candidates: (1) PRX-102, a therapeutic protein candidate for the treatment of Fabry disease, a rare, genetic lysosomal disorder in humans; (2) PRX-105, a plant cell expressed pegylated recombinant acetylcholinesterase product candidate for biodefense and other indications; (3) PRX-106, or pr-antiTNF, a plant cell expressed recombinant fusion protein made from the soluble form of the human TNF receptor (TNFR) and an antibody portion, which is being developed as a treatment of certain immune diseases such as rheumatoid arthritis, juvenile idiopathic arthritis, spondylitis, psoriatic arthritis and plaque psoriasis; (4) an orally-administered glucocerebrosidase enzyme for the treatment of Gaucher disease patients utilizing oral delivery of the recombinant enzyme produced within carrot cells; and (5) two additional undisclosed therapeutic proteins, both of which are being evaluated in animal studies. We participated in a pre-investigational new drug, or IND, meeting with respect to one of the undisclosed product candidates in the first quarter of 2012 and a pre-IND meeting for the second candidate is planned for later this year. In March 2010, we initiated a preliminary phase I clinical trial of PRX-105 which we completed in June 2010. We are currently preparing for further efficacy trials of this product candidate in larger animals. In our preclinical studies we utilized an analogue to nerve gas. However, we anticipate that we will use live nerve gas rather than an analogue in the proposed additional efficacy trials in animals. In December 2010, we held a pre-IND meeting with the FDA with respect to PRX-102. We completed preclinical trials in small and large animals with respect to PRX-102, and we expect to submit an IND to the FDA during the second quarter of 2012 in connection with an anticipated phase I/II clinical study of PRX-102 in Fabry patients, and to initiate the trial once the IND is approved, if at all. In December 2011 we held a pre-IND meeting with respect to PRX-106 and we expect to submit an IND around the fourth quarter of 2012. We are also conducting pre-clinical trials of our orally-administrated glucocerebrosidase enzyme for the treatment of Gaucher disease in small and large animals.
| 5 |
Except for the rights to commercialize taliglucerase alfa worldwide (other than Israel) which we licensed to Pfizer, we hold the worldwide commercialization rights to our proprietary development candidates, and we intend to establish an internal, commercial infrastructure and targeted sales force to market taliglucerase alfa in Israel and our other products, if approved, in North America, the European Union and in other significant markets, including Israel. In addition, we are continuously evaluating potential strategic marketing partnerships.
Our pr-antiTNF product candidate has an amino acid sequence that is similar to Enbrel® which is one of the treatments for patients of those diseases. We believe that we may be able to reduce the development risks and time to market for our product candidates as our product candidates are based on well-understood proteins with known biological mechanisms of action. Based on our pre IND meeting with the FDA held in December 2011, we believe we may submit an IND around year end and, if approved, commence a phase I clinical trial shortly thereafter with no need for an external partner.
Our ProCellEx protein expression system consists of a comprehensive set of technologies and capabilities for the development of recombinant proteins, including advanced genetic engineering technology and plant cell-based protein expression methods. Through our ProCellEx protein expression system, we can develop highly complex recombinant therapeutic proteins all the way to the scale-up of a purified product produced in compliance with current. We believe that our plant cell-based expression technology will enable us, in certain cases, to develop and commercialize recombinant proteins without infringing upon the method-based patents or other intellectual property rights of third parties. The major elements of our ProCellEx system are patent protected in most major countries. Moreover, we expect to enjoy method-based patent protection for the proteins we develop using our proprietary ProCellEx protein expression technology, although there can be no assurance that any such patents will be granted. In some cases, we may be able to obtain patent protection for the compositions of the proteins themselves. We have filed for United States and international composition of matter patents for taliglucerase alfa.
Our ProCellEx protein expression system is built on flexible custom-designed bioreactors made of polyethylene and optimized for the development of complex proteins in plant cell cultures. These bioreactors entail low initial capital investment, are rapidly scalable at a low cost and require less hands-on maintenance between cycles, compared to the highly complex, expensive, stainless steel bioreactors typically used in mammalian cell-based production systems. As a result, through our ProCellEx protein expression system, we believe that we can develop recombinant therapeutic proteins yielding substantial cost advantages, accelerated development and other competitive benefits as compared to mammalian cell-based protein expression systems.
We have successfully demonstrated the feasibility of our ProCellEx system through: the clinical and preclinical studies performed by us to date, including the positive efficacy and safety data in our phase III study for taliglucerase alfa, our switchover study and our extension study; preclinical results in well-known models in our enzyme for each of Fabry disease and pr-antiTNF; extensive animal studies for our acetylcholinesterase enzyme; and by expressing, on an exploratory, research scale, many additional complex therapeutic proteins belonging to different drug classes, such as enzymes, hormones, monoclonal antibodies, cytokines and vaccines. The therapeutic proteins we have expressed to date in research models have produced the intended composition and similar biological activity compared to their respective human-equivalent proteins. Moreover, several of such proteins demonstrated advantageous biological activity when compared to the biotherapeutics currently available in the market to treat the applicable disease or disorder. We believe that the clinical success of taliglucerase alfa represents a strong proof-of-concept for our ProCellEx protein expression system and plant cell-based protein expression technology. We also believe that the significant benefits of our ProCellEx protein expression system, if further substantiated in clinical trials and in the commercialization of our product candidates, have the potential to transform the industry standard for the development of complex therapeutic proteins.
Our goal is to become a leading fully integrated biopharmaceutical company focused on the development and commercialization of proprietary and biosimilar or generic versions of recombinant therapeutic proteins. To that end, we are leveraging our ProCellEx protein expression system to develop a pipeline of proprietary and biosimilar versions of recombinant therapeutic proteins. In addition to the product candidates that we are developing internally, we have entered into agreements for additional compounds with academic institutions, including a licensing agreement with the technology transfer arm of Israel’s Weizmann Institute of Science and an agreement with the technology transfer arm of the Hebrew University of Jerusalem. In addition, we are collaborating with other pharmaceutical companies to develop therapeutic proteins that can benefit from the significant cost, intellectual property and other competitive advantages of our ProCellEx protein expression system. We entered into an agreement with Teva in September 2006 under which we have agreed to collaborate on several matters. We and Teva identified two proteins for research and development activities under the agreement, but in 2009 both of the projects were terminated for commercial reasons. Other elements of our collaboration with Teva, as well as the exploration of new collaborations, are currently ongoing. We also continuously review and consider additional development and commercialization alliances with other pharmaceutical companies and academic institutions.
| 6 |
Industry Overview
Recombinant proteins have revolutionized the treatment of a variety of diseases and disorders. Recombinant proteins are forms of human proteins that are produced, or expressed, using a mammalian, plant, bacterial or yeast cell as a production engine. In the early 1970s, a number of key scientific breakthroughs, including, among others, the demonstration of genetic engineering and genetic sequencing techniques, as well as the synthesis of genes, led to the advancement of recombinant protein technology. As a result, the market for pharmaceutical therapeutics has undergone a transformation as recombinant proteins and other biologic products have become an increasingly significant portion of the global drug market and the focus of research worldwide. The IMS Institute for Healthcare Informatics reports that global biologic spending was $138 billion in 2010 (Report by the IMS Institute for Healthcare Informatics, May 2011).
Mammalian cell-based systems are the current industry standard for expression of recombinant therapeutic glycoproteins (complex proteins that contain sugar residues), including catalytic enzymes and monoclonal antibodies. Mammalian cell-based systems were first introduced in the late 1980s and are currently used to produce many of the biotechnology industry’s largest and most successful therapeutic proteins, including Epogen®, Neupogen®, Cerezyme, Rituxan®, Enbrel, Neulasta®, Remicade and Herceptin®. Mammalian cell-based expression technology is based on the introduction of a human gene encoding for a specific therapeutic protein into the genome of a mammalian cell. The cells most often used in connection with mammalian cell-based protein expression are Chinese hamster ovary (CHO) cells.
Mammalian cell-based expression systems have become the dominant system for the expression of recombinant proteins due to their capacity for sophisticated, proper protein folding (which is necessary for proteins to carry out their intended biological activity), assembly and post-expression modification, such as glycosilation (the addition of sugar residues to a protein which is necessary to enable specific biological activity by the protein). While bacterial and yeast cell-based expression systems were the first protein expression systems developed by the biotechnology industry and remain cost-effective compared to mammalian cell-based production methodologies, proteins expressed in bacterial and yeast cell-based systems lack the capacity for sophisticated protein folding, assembly and post-expression modifications, which are key factors of mammalian cell-based systems. Accordingly, such systems cannot be used to produce glycoproteins or other complex proteins and, therefore, bacterial and yeast cell-based systems are limited to the expression of the most basic, simple proteins, such as insulin and growth hormones. Due to their significant advantages, mammalian cell-based expression systems can produce proteins with superior quality and efficacy compared to proteins expressed in bacteria and yeast cell-based systems. As a result, the majority of currently approved therapeutic proteins, as well as those under development, are produced in mammalian cell-based systems.
Despite the utility and widespread use of mammalian cell-based systems, they are subject to a number of disadvantages. CHO cells and other mammalian cells are highly sensitive and can only be grown under near perfect conditions, requiring highly complex, expensive, stainless steel bioreactors which tightly regulate the required temperature, pH and oxygen levels. As a result, such bioreactor systems are very costly and complicated to operate. CHO cells and other mammalian cells are also susceptible to viral infections, including human viruses, and several cases of viral contamination have occurred recently. The FDA and other regulatory authorities require viral inactivation and other rigorous and detailed procedures for mammalian cell-based manufacturing processes in order to address these potential hazards, thereby increasing the cost and time demands of such expression systems. Furthermore, the current FDA and other procedures only ensure screening for scientifically identified, known viruses. Accordingly, compliance with current FDA and other procedures does not fully guarantee that patients are protected against transmission of unknown or new potentially fatal viruses that may infect mammalian cells. In addition, mammalian cell-based expression systems require large quantities of sophisticated and expensive growth medium to accelerate the expression process.
Several companies and research institutions have explored alternatives to mammalian cell-based production technologies that overcome some of these disadvantages, focusing primarily on the expression of human proteins in genetically-modified organisms, or GMOs, such as transgenic field-grown, whole plants and transgenic animals. However, these alternate techniques may be restricted by regulatory and environmental risks regarding contamination of agricultural crops and by the difficulty in applying cGMP standards of the pharmaceutical industry to these expression technologies.
| 7 |
ProCellEx: Our Proprietary Protein Expression System
ProCellEx is our proprietary production system. We have developed ProCellEx based on our plant cell culture technology for the development, expression and manufacture of recombinant proteins. Our expression system consists of a comprehensive set of capabilities and proprietary technologies, including advanced genetic engineering and plant cell culture technology, which enables us to produce complex, proprietary and biologically equivalent proteins for a variety of human diseases. Our protein expression system facilitates the creation and selection of high expressing, genetically stable cell lines capable of expressing recombinant proteins. The entire protein expression process, from initial nucleotide cloning to large-scale production of the protein product, occurs under cGMP-compliant, controlled processes. Our plant cell culture technology uses plant cells, such as carrot and tobacco cells, which undergo advanced genetic engineering and are grown on an industrial scale in a flexible bioreactor system. Cell growth, from scale up through large-scale production, takes place in flexible, sterile, polyethylene bioreactors which are confined to a clean-room environment. Our bioreactors are well-suited for plant cell growth using a simple, inexpensive, chemically-defined growth medium as a catalyst for growth. The reactors are custom-designed and optimized for plant cell cultures, easy to use, entail low initial capital investment, are rapidly scalable at a low cost and require less hands-on maintenance between cycles. Our protein expression system does not involve mammalian or animal components or transgenic field-grown, whole plants at any point in the production process.
Our ProCellEx system is capable of producing proteins with an amino acid structure practically equivalent to that of the desired human protein, and with a very similar, although not identical, glycan, or sugar, structure. Our internal research and external laboratory studies have demonstrated that ProCellEx is capable of producing recombinant proteins that exhibit a glycan and amino acid structure similar to their naturally-produced human counterparts. In collaboration with the Weizmann Institute of Science, we have demonstrated that the three-dimensional structure of a protein expressed in our proprietary plant cell-based expression system retains the same three-dimensional structure as exhibited by the mammalian cell-based expressed version of the same protein. In addition, proteins produced by our ProCellEx system maintain the biological activity that characterize that of the naturally-produced proteins. Based on these results, we believe that proteins developed using our ProCellEx protein expression system have the intended composition and correct biological activity of their human equivalent proteins.
To date, our manufacturing facility, which uses our ProCellEx system, was determined to be acceptable by each of the FDA, EMA, ANVISA and the Israeli MOH after GMP inspections performed as part of their respective reviews for marketing approval of taliglucerase alfa.
Competitive Advantages of Our ProCellEx Protein Expression System
We believe that our ProCellEx protein expression system, including our advanced genetic engineering technology and plant cell-based protein expression methods, affords us a number of significant advantages over mammalian, bacterial, yeast and transgenic cell-based expression technologies, including the following:
Ability to Penetrate Certain Patent-Protected Markets. We seek to develop recombinant proteins that we believe we can produce and commercialize without infringing upon the method-based patents or other intellectual property rights of third parties. Certain biotherapeutic proteins available for commercial sale are not protected by patents that cover the compound and are available for use in the public domain. Rather, the process of expressing the protein product in mammalian or bacterial cell systems is protected by method-based patents. Using our plant cell-based protein expression technology, we are able to express an equivalent protein without infringing upon these method-based patents. Moreover, we expect to enjoy method-based patent protection for the proteins we develop using our proprietary ProCellEx protein expression technology, although there can be no assurance that any such patents will be granted. In some cases, we may be able to obtain patent protection for the compositions of the proteins themselves. We have filed for United States and international composition of matter patents for taliglucerase alfa, PRX-102 and certain of our other product candidates.
Significantly Lower Capital and Production Costs. Plant cells have a number of dynamic qualities that make them well-suited for the production of therapeutic proteins. Plant cells grow rapidly under a variety of conditions and are not as sensitive to temperature, pH and oxygen levels as mammalian cells. Our ProCellEx protein expression system, therefore, requires significantly less upfront capital expenditures as it does not use the highly complex, expensive, stainless steel bioreactors typically used in mammalian cell-based production systems to maintain very specific temperature, pH and oxygen levels. Instead, we use simple polyethylene bioreactors that can be maintained at the room temperature of the clean-room in which they are placed. This system also reduces ongoing production and monitoring costs typically incurred by companies using mammalian cell-based expression technologies. Furthermore, while mammalian cell-based systems require very costly growth media at various stages of the production process to achieve target yields of their proteins, plant cells require only simple and much less expensive solutions based on sugar, water and microelements at infrequent intervals to achieve target yields. We believe that these factors will potentially result in lower capital and production costs for the commercial scale production of proteins by our ProCellEx system thereby providing us with a competitive advantage over competing protein expression technologies.
| 8 |
Elimination of the Risk of Viral Transmission or Infection by Mammalian Components. By nature, plant cells do not carry the risk of infection by human or other animal viruses. As a result, the risk of contamination of our products under development and the potential risk of viral transmission from our products under development to future patients, whether from known or unknown viruses, is eliminated. Because our product candidates do not bear the risk of viral transmission, we are not required by the FDA or other regulatory authorities to perform the constant monitoring procedures for mammalian viruses during the protein expression process that mammalian cell-based manufacturers are required to undertake. In addition, the production process of our ProCellEx protein expression system is void of any mammalian components which are susceptible to the transmission of prions, such as those related to bovine spongiform encephalopathy (commonly known as “mad-cow disease”). These factors further reduce the risks and operating costs of our ProCellEx system compared to mammalian cell-based expression systems.
More Effective and Potent End Product Relative to Mammalian Based Systems. Our ProCellEx protein expression system produces enzymes which have uniform glycosilation patterns and therefore do not require the lengthy and expensive post-expression modifications that are required for certain proteins produced by mammalian cell-based systems, including the proteins for the treatment of Gaucher disease. Such post-expression modifications in mammalian cell-produced proteins are made in order to expose the terminal mannose sugar residues, which are structures on a protein that are key elements in allowing the produced protein to bind to a target cell and subsequently be taken into the target cell for therapeutic benefit. In the production of Cerezyme, exposing these terminal mannose sugar residues involves a multitude of highly technical steps which add time and cost to the production process. In addition, these steps do not guarantee the exposure of all of the required terminal mannose sugar residues, resulting in potentially lower effective yields and inconsistency in potency from batch to batch. Our ProCellEx protein expression system, by contrast, produces taliglucerase alfa in a “ready to use” form that does not require additional glycosilation or other modifications to make taliglucerase alfa suitable for use in enzyme replacement therapy for Gaucher disease. We believe this quality increases the potency and consistency of the expressed proteins, thereby further increasing the cost advantages of our ProCellEx protein expression system over competing protein expression methodologies.
Broad Range of Expression Capabilities. Our ProCellEx protein expression system is able to produce a broad array of complex glycosilated proteins, which differentiates our system from bacterial and yeast cell-based systems which are unable to produce complex proteins. We have successfully demonstrated the feasibility of our ProCellEx system by producing, on an exploratory, research scale, a variety of therapeutic proteins belonging to different classes of recombinant drugs, such as enzymes, hormones, monoclonal antibodies, cytokines and vaccines. We have demonstrated that the recombinant proteins we have expressed to date have the intended composition and correct biological activity of their human-equivalent protein, with several of such proteins demonstrating advantageous biological activity compared to the currently available biotherapeutics. In specific cases, we have been successful in expressing proteins that have not been successfully expressed in other production systems.
Our Strategy
Our goal is to become a leading fully integrated biopharmaceutical company focused on the development and commercialization of proprietary and biosimilar or generic versions of recombinant therapeutic proteins. To achieve our goal, we intend to:
Facilitate the successful development and commercialization of taliglucerase alfa by Pfizer. We intend to work with our licensee, Pfizer, to develop and commercialize taliglucerase alfa. We have been collaborating with Pfizer to facilitate the transition of certain of our taliglucerase alfa assets to Pfizer’s organization. We are cooperating with Pfizer with respect to our Expanded Access protocol for taliglucerase alfa in order to facilitate the participation of additional physicians in additional sites in the protocol. Pfizer is promoting the protocol to new clinical sites and is recruiting additional patients. Pfizer has also taken an active, collaborative role in our ATU in France and similar programs in Brazil and other countries. We have also begun to facilitate relationships between Pfizer and the Gaucher community and third-party payors. We intend to actively participate and provide our expertise in Pfizer’s development and commercialization efforts with respect to taliglucerase alfa.
| 9 |
Obtain Regulatory Approval for Taliglucerase Alfa for the Treatment of Gaucher Disease. We completed successfully our pivotal phase III clinical trial of taliglucerase alfa in September 2009 and announced the positive top-line study results in October 2009 and full study results in February 2010. We filed an NDA with the FDA in December 2009 and taliglucerase alfa was originally granted a PDUFA date of February 25, 2011. On February 24, 2011, the FDA issued a CRL indicating that the FDA’s review was complete but questions remain that preclude the approval of the NDA in its then current form. In July 2011 we submitted our reply to the CRL and in August 2011, the FDA notified us that it had established February 1, 2012 as the new PDUFA date. In December 2011, the FDA notified us that it had decided to extend the PDUFA date to May 1, 2012. In its notification, the FDA stated that its decision related to certain clinical information regarding taliglucerase alfa we had submitted in November 2011 in response to an FDA request. The requested information related mainly to the presentation of select data provided in the NDA. As this information was requested and provided within 90 days of the February 1, 2012 PDUFA goal date, the FDA had the option to extend the PDUFA goal date to provide adequate time for the FDA to complete its review. A three-month extension cycle is the standard period granted. No additional data was requested by the FDA in the notification, nor were we notified of any specific deficiency in the taliglucerase alfa NDA. In addition, in November 2010 we submitted a marketing application to the Israeli MOH, and with our partner Pfizer, an MAA to each of the EMA, ANVISA and the Australian TGA for taliglucerase alfa.
Develop a Pipeline of Innovative or Biosimilar Versions of Recombinant Therapeutic Proteins. We are leveraging our ProCellEx protein expression system to develop a pipeline of innovative or biosimilar versions of recombinant proteins, with an emphasis on therapeutic treatments with large market opportunities. We select additional therapeutic candidates for development through in-house testing, licensing agreements with academic institutions and collaborations with pharmaceutical partners. We have currently identified several product candidates that are mainly oriented towards the specialty disease and therapeutic market segments, including treatments for Fabry disease and an acetylcholinesterase enzyme based therapy for biodefense and nerve gas toxicity treatments. We have also identified several other product candidates that are chemical equivalents of approved therapeutic products that will no longer be patent protected within the next couple of years, such as pr-antiTNF, our proprietary product candidate for the treatment of certain immune diseases such as rheumatoid arthritis. We believe our cost-effective technology will be an important asset for the commercialization of such drug candidates. We believe that the clinical and regulatory pathway for many of our pipeline product programs candidates is already established, and that this may reduce the risks and costs associated with our clinical development programs. Furthermore, established markets already exist for the development of most of our current product candidates. We plan to apply the manufacturing, clinical and regulatory experience we have gained from the development of our lead product candidate to advance a number of our preclinical product candidates into clinical trials over the next few years.
Collaborate with Third Party Pharmaceutical Suppliers and Build a Targeted Sales and Marketing Infrastructure. We have licensed to Pfizer the right to commercialize taliglucerase alfa worldwide, except in Israel. We plan to establish our own, internal sales and marketing capabilities for taliglucerase alfa in Israel, and, for our other product candidates, in North America, the European Union and in other significant markets, including Israel. We believe that the focus of our current clinical pipeline is mainly on relatively rare genetic disorders with small patient populations and a highly concentrated group of physicians focused on treating patients with such disorders may facilitate our creation of a targeted internal sales force. In addition we are continuously evaluating potential strategic marketing partnerships with respect to our other product candidates.
Establish Development and Commercialization Alliances with Corporate Partners. We believe that our technology and know-how has broad applicability to many classes of proteins and can be used to develop and potentially enhance numerous existing marketed protein therapeutics. We intend to leverage our technology and know-how by pursuing development and commercialization alliances with corporate partners for specific products and territories in order to enable us to optimize our resources and effectively penetrate a wider range of target diseases and therapeutic markets. In November 2009, we entered into a license and supply agreement with Pfizer for the development and commercialization of taliglucerase alfa. We entered into an agreement with Teva in September 2006 for the development of two proteins. Although programs relating to two proteins to be developed under the agreement were terminated for commercial reasons in 2009, other elements of our collaboration are still ongoing and we are exploring other potential ways to collaborate. Last, we are engaged in confidential discussions with a number of multinational pharmaceutical companies to discuss additional potential collaboration arrangements.
Acquire or In-License New Technologies, Products or Companies. We continuously seek attractive product candidates and innovative technologies to in-license or acquire. We intend to focus on product candidates that would be synergistic with our ProCellEx protein expression system and expertise and that represent large potential market opportunities. We believe that by pursuing selective acquisitions of technologies in businesses that complement our own, we will be able to enhance our competitiveness and strengthen our market position.
Leverage Strength and Experience of Our Management Team and Board of Directors. Our management team has extensive experience in the biotechnology and pharmaceutical industry. The Interim Chairman of our Board of Directors, Mr. Zeev Bronfeld, is a highly experienced Israeli health care investor. In February 2008, we appointed Professor Roger D. Kornberg, a renowned biochemist and laureate of the Nobel Prize in Chemistry, to our Board of Directors. We will continue to leverage their experience and established track record as well as their relationships across the biotechnology and pharmaceutical industries.
| 10 |
Our Pipeline Drug Candidates
Our Lead Product Candidate, Taliglucerase Alfa
Taliglucerase alfa, our lead proprietary product candidate, is a plant cell expressed recombinant glucocerebrosidase enzyme (GCD) for the treatment of Gaucher disease. In July 2007, we reached an agreement with the FDA on the final design of our pivotal phase III clinical trial of taliglucerase alfa through the FDA’s special protocol assessment (SPA) process. We successfully completed our phase III pivotal clinical trial of taliglucerase alfa in September 2009 and announced positive top-line results of the clinical trial in October 2009 and full study results in February 2010. We submitted an NDA to the FDA in December 2009. In January 2010, the FDA requested additional data regarding the CMC section of the NDA. We provided the requested data to the FDA in April 2010 and in July 2010 we received notification from the FDA that it had accepted the filing of our NDA and assigned it a PDUFA date of February 25, 2011. On that date, the FDA issued a CRL indicating that the FDA’s review was complete but questions remain that preclude the approval of the NDA in its then current form. The main questions raised by the FDA regarding the NDA related to the clinical and CMC sections. In the clinical section of the CRL, the FDA requested additional data from our ongoing switchover trial and our long-term extension trial. At the time the NDA was submitted, full data from these trials was not available. In the CMC section of the CRL, the FDA requested information regarding testing specifications and assay validation. The FDA did not request additional clinical studies in the CRL. We met with the FDA in May 2011 to clarify the FDA’s requests, and in July 2011 we submitted our reply to the CRL. In August 2011, the FDA notified us that it had accepted for review the resubmission of the taliglucerase alfa NDA, had deemed the resubmission a class 2, or 6-month, response and established February 1, 2012 as the new PDUFA date. In December 2011, the FDA notified us that it had decided to extend the PDUFA date to May 1, 2012. In its notification, the FDA stated that its decision related to certain clinical information regarding taliglucerase alfa we had submitted in November 2011 in response to an FDA request. The requested information related mainly to the presentation of select data provided in the NDA. As this information was requested and provided within 90 days of the February 1, 2012 PDUFA goal date, the FDA had the option to extend the PDUFA goal date to provide adequate time for the FDA to complete its review. A three-month extension cycle is the standard period granted. No additional data was requested by the FDA in the notification, nor were we notified of any specific deficiency in the taliglucerase alfa NDA.
In the fourth quarter of 2010, Pfizer, our commercialization partner, filed, with our cooperation, an MAA for taliglucerase alfa with the EMA. As part of its ongoing review of the MAA, the EMA delivered a list of outstanding points to be addressed by the applicant. Among the topics currently in discussion, is the orphan drug designation and exclusivity granted by the EMA/European Commission to VPRIV, which could prevent the marketing authorization of taliglucerase alfa in the European Union for a 10-year market exclusivity period commencing as of the August 2010 marketing authorization of VPRIV in the European Union. As part of the MAA procedure, Pfizer, with our cooperation, is challenging VPRIV's orphan market protection with respect to taliglucerase alfa pursuant to the EU orphan drug regulation. The EU orphan drug regulation provides for the possibility of such a challenge, and for an exception to this exclusivity to be granted, based on a number of factors, including contribution to patient care, clinical, supply, capacity and others.
Following the submission of the taliglucerase alfa MAA to the EMA, a GMP audit of our manufacturing facility was performed and it was found compliant with EMA GMP in December 2011.
In January 2010, COMP, after reviewing all relevant clinical data, recommended that the European Commission grant orphan drug designation to taliglucerase alfa for the treatment of Gaucher disease and in March 2010, the European Commission granted orphan designation to taliglucerase alfa.
In addition, marketing applications for taliglucerase alfa have been submitted with each of the Israeli MOH, ANVISA and the Australian TGA.
In September 2009, the FDA’s Office of Orphan Product Development granted taliglucerase alfa Orphan Drug Status. The Orphan Drug designation in the United States for taliglucerase alfa for the treatment of Gaucher disease provides special status to taliglucerase alfa provided that it meets certain criteria. As a result of the Orphan Drug designation, we are qualified for the tax credit and marketing incentives of the Orphan Drug Act of 1983. A marketing application for a prescription drug product that has been designated as a drug for a rare disease or condition is not subject to a prescription drug user fee unless the application includes an indication for other than a rare disease or condition.
| 11 |
In July 2009, following a request by the FDA, we submitted a treatment protocol to the FDA in order to address an expected shortage of the current enzyme replacement therapy approved for Gaucher disease. The treatment protocol was approved by the FDA in August 2009.
During the third quarter of 2008, we initiated a double blind, follow-on extension study as part of our phase III clinical trial of taliglucerase alfa. Eligible patients who successfully completed nine months of treatment in our phase III clinical trial were offered the opportunity to participate in the extension study and continue to receive taliglucerase alfa at the same dose they received in the phase III clinical trial for an additional 15 months in a blinded manner. In February 2012, we announced data from 26 adult patients enrolled in the trial. According to the data, after 24 months, patients continued to demonstrate a significant improvement in all parameters with a similar safety profile as seen in the phase III clinical trial. Furthermore, those patients who were followed specifically for their bone parameters using Quantitative Chemical Shift Imaging (QCSI) MRI continued to show bone marrow improvement over time. We have also initiated a home care treatment program for patients enrolled in the extension study.
In February 2010, the Israeli MOH completed a successful good manufacturing practices, or GMP, audit of our manufacturing facilities in Carmiel, Israel. The audit was performed as part of the Israeli MOH’s evaluation of our manufacturing process for taliglucerase alfa. On February 20, 2011, we received a letter from the FDA notifying us that the FDA had completed its review of the Establishment Inspection Report in connection with the FDA’s inspection of our facility in Carmiel, Israel, and that the FDA had classified our facility as acceptable. In June 2011, ANVISA completed a successful GMP audit of the facility and determined that the facility is acceptable and in December 2011, the Irish Medicines Board, or IMB, completed a successful GMP audit of our facility and issued a Certificate of GMP Compliance of a Manufacturer for the facility. The IMB certificate is accepted by all health authorities in the European Union under the European Union's centralized marketing authorization procedure, and by authorities of several other countries that recognize EU certification.
In the fourth quarter of 2008 we announced the enrollment of the first patient in a worldwide, multi-center, open-label, switch-over trial which has been reviewed by the FDA and is designed to assess the safety and efficacy of taliglucerase alfa. The switch-over trial, which was not a prerequisite for approval, was originally designed to include 15 patients with Gaucher disease that are currently undergoing enzyme replacement therapy with imiglucerase (Cerezyme). Due to the then shortage of the currently available enzyme replacement therapy for Gaucher disease, after fully enrolling 15 patients, we extended the trial to include additional patients. In August 2011, we announced top-line results from the switch-over trial. A total of 26 adult patients were enrolled in the switchover trial. Patients enrolled in the trial were switched from imiglucerase (doses ranging from around 10-60 U/kg every other week) to an equivalent dose using the same number of units of taliglucerase alfa over a nine-month period. The results of the switchover trial, which were announced in February 2012, demonstrate that over the nine-month treatment period, patients remained stable with regard to spleen volume, liver volume, platelet count and hemoglobin concentration, the efficacy endpoints of the switchover trial, after switching from imiglucerase to taliglucerase alfa. The safety analysis of the switchover trial demonstrated that taliglucerase alfa was well tolerated, and no drug related serious adverse events were reported. Patients in the extension trial and the switchover trial are still being treated with taliglucerase alfa.
In addition, in December 2009, we filed a proposed pediatric investigation plan to the Pediatric Committee of the EMA, which was approved during the first quarter of 2010. We have since initiated the pediatric study and have enrolled all of the naïve pediatric patients required according to the study protocol. All of the enrolled patients are in the final stages of pediatric patient treatment.
In clinical trials of healthy subjects and in vivo primate studies, taliglucerase alfa has demonstrated an increased half-life and prolonged presence of the enzyme in the blood serum of the subjects as compared to Cerezyme, the only enzyme replacement therapy currently marketed to treat Gaucher disease.
We believe that taliglucerase alfa, if approved, has the potential to offer patients and healthcare payors a more effective and cost efficient treatment of Gaucher disease compared to the currently available enzyme replacement therapies because of the following advantages:
Increased Glycan Efficacy and Consistency. We believe that our ProCellEx protein expression system produces recombinant proteins that exhibit consistent enzymatic activity from batch to batch. This results in a highly active product that may achieve a desired therapeutic effect more effectively than the activity demonstrated in proteins produced through mammalian cell-based expression systems due to its greater glycan efficacy and consistency. This quality increases the effective consistency in potency and further increases the cost advantages from using our plant cell-based expression technology compared to competing protein expression methodologies.
| 12 |
Longer Half-Life. The data generated in preclinical and human clinical trials relating to the half-life of taliglucerase alfa in the subjects’ blood serum after infusion showed that the half-life of taliglucerase alfa is significantly longer than that of Cerezyme when measured and compared to publicly available data on Cerezyme.
Cost-Effective. Taliglucerase alfa is potentially less expensive to produce as the manufacturing process does not require the large initial set-up investments involved in mammalian cell-based protein production, the extensive ongoing costs associated with growth media and monitoring throughout the production process nor any of the post-expression modification costs in order to modify the glycosilation of the proteins produced through the mammalian cell-based methodologies.
We intend to pursue post-marketing studies to confirm these advantages. Although Gaucher disease is a relatively rare disease, it represents a substantial commercial market due to the severity of the symptoms and the chronic nature of the disease. We believe that the approval of taliglucerase alfa as a treatment for Gaucher disease, if at all, with its potentially longer acting profile and more cost-effective development process, may lead to an increase in the number of patients who will be able to have access to and afford such treatment, thereby expanding the size of the market for Gaucher disease treatments.
In addition, we are developing a new method for delivering active recombinant proteins systemically through oral administration of transgenic plant cells expressing biotherapeutic proteins. We have commenced pre-clinical studies of an oral form of taliglucerase alfa. If proven effective, our experimental oral taliglucerase alfa would be the first protein to be administered orally rather than through intravenous therapy. Our oral taliglucerase alfa is a plant cell expressed form of GCD that is naturally encapsulated within carrot cells genetically engineered to express the GCD enzyme. Pre-clinical studies of oral taliglucerase alfa demonstrate the stability of the enzyme in the cell and the capacity of the cell’s cellulose wall to protect the enzyme against degradation in the digestive tract in an in-vitro model of the stomach and intestines. Additionally, rats fed with lyophilized carrot cells expressing GCD have accumulated the active enzyme in the target organs; the spleen and liver.
We believe that oral delivery of taliglucerase alfa presents a number of advantages. First, the development of oral taliglucerase alfa has the advantage of leveraging the well-characterized mechanism of action of our intravenously-administered taliglucerase alfa product candidate. In addition, we believe that oral delivery of taliglucerase alfa may dramatically change the treatment paradigm for Gaucher patients, compared to the intravenous delivery of taliglucerase alfa, and contribute to increased compliance and the facilitation of treatment management. Further, oral delivery of GCD targets the disease-specific organs without the need for lifetime dependence on repeated intravenous infusions. Last, our oral enzyme replacement therapy does not have the unpredictable long term untoward effects of the inhibition of other non-disease-specific compounds that are common in oral substrate reduction therapy.
Our oral taliglucerase alfa product candidate is a recombinant form of the GCD enzyme, not a small molecule. This differentiates our oral product candidate from other early clinical stage, experimental, small molecule, oral drugs which are being developed for the treatment of Gaucher disease by Genzyme and Amicus Therapeutics, Inc., or Amicus Therapeutics. Small molecule based treatments for Gaucher disease, such as Zavesca, have different mechanisms of action than those associated with enzyme replacement therapy, and may be associated with a number of side effects. We have filed patent applications with respect to this new protein delivery mechanism in other countries with commercially significant markets. Currently, we are the exclusive owners of all rights to this technology.
Gaucher Disease Background
Gaucher disease, a hereditary, genetic disorder with severe and debilitating symptoms, is the most prevalent lysosomal storage disorder in humans. Lysosomal storage disorders are metabolic disorders in which a lysosomal enzyme, a protein that degrades cellular substrates in the lysosomes of cells, is mutated or deficient. Lysosomes are small membrane-bound cellular structures within cells that contain enzymes necessary for intracellular digestion. Gaucher disease is caused by mutations or deficiencies in the gene encoding GCD, a lysosomal enzyme that catalyzes the degradation of the fatty substrate, glucosylceramide (GlcCer). The normal degradation products of GlcCer are glucose and ceramide, which are easily excreted by the cells through normal biological processes. Patients with Gaucher disease lack or otherwise have dysfunctional GCD and, accordingly, are not able to break down GlcCer. The absence of an active GCD enzyme leads to the accumulation of GlcCer in lysosomes of certain white blood cells called macrophages. Macrophages affected by the disease become highly enlarged due to the accumulation of GlcCer and are referred to as “Gaucher cells.” Gaucher cells accumulate in the spleen, liver, lungs, bone marrow and brain. Signs and symptoms of Gaucher disease may include enlarged liver and spleen, abnormally low levels of red blood cells and platelets and skeletal complications. In some cases, the patient may suffer an impairment of the central nervous system.
| 13 |
Current Treatments for Gaucher Disease
The standard of care for Gaucher disease is enzyme replacement therapy using recombinant GCD to replace the mutated or deficient natural GCD enzyme. The latest studies estimate that there are approximately 12,000 patients suffering from Gaucher disease worldwide, but only approximately 6,000 patients are undergoing treatment. Enzyme replacement therapy is a medical treatment in which recombinant enzymes are injected into patients in whom the enzyme is lacking or dysfunctional. Cerezyme and VPRIV, enzyme replacement therapies commercialized by Genzyme and Shire, respectively, are the only recombinant GCDs currently available on the market for the treatment of Gaucher disease. As enzyme replacement therapy does not cure the genetic disorder, but rather provides an external source for transfusion of the missing or mutated enzyme, Gaucher disease patients generally receive the treatment over their entire lifetime. According to public reports by Sanofi, consolidated sales of Cerezyme from April 1, 2011 through the end of the year were €441.0 million (or $579.6 million), a growth of 11.1% compared to the same period in 2010. According to Genzyme, it has suffered interruptions and other limitations in its Cerezyme production and release since September 2009 and, as a result, Cerezyme supplies have been limited. Shire reported annual worldwide sales of VPRIV of approximately $256 million in 2011, a growth of 79% compared to the same period in 2010 (VPRIV was approved for sale in the United States in February 2010 and in the European Union in August 2010).
Cerezyme is produced through a mammalian cell-based protein expression process in CHO cells and VPRIV is produced using a human cancer cell line. There are no known severe side effects to the use of Cerezyme or VPRIV, and Cerezyme's approved use over the past decade suggests that it is an effective treatment of Gaucher disease. However, Cerezyme and VPRIV are both subject to the limitations of most mammalian cell-based therapeutic proteins, including lengthy and costly production processes and contamination risks.
Zavesca (miglustat), which is marketed by Actelion Ltd., or Actelion, is a small molecule drug for the treatment of Gaucher disease. Zavesca has been approved by the FDA for use in the United States as an oral treatment. However, it has many side effects and the FDA has approved it only for administration to those patients who cannot be treated through enzyme replacement therapy, and, accordingly, have no other treatment alternative. As a result, Zavesca’s use has been extremely limited. Actelion has reported sales of Zavesca of approximately CHF 68.4 million (approximately $74.4 million) in 2011.
Taliglucerase Alfa Development Program
We believe the clinical development and regulatory path for taliglucerase alfa will be similar to that followed by the existing enzyme replacement therapy currently on the market. After completion of laboratory testing and preclinical studies of taliglucerase alfa, we commenced a phase I clinical trial which was completed in June 2006. After the conclusion of the phase I clinical trial and discussions with the FDA, we proceeded directly with a pivotal phase III clinical trial of taliglucerase alfa without undergoing a phase II clinical trial. We reported positive top-line results of the phase III clinical trial in October 2009 and full study results in February 2010. This study was summarized in a recent publication in BLOOD, the Journal of the American Hematological Society (Zimran, et. al., September 2011). In February 2012, we announced positive results from our extension study in naïve patients and from our switchover study.
Laboratory Testing and Preclinical Studies of Taliglucerase Alfa
We conducted several in vitro tests and in vivo preclinical studies of taliglucerase alfa. Our preclinical rodent and primate trials generated extensive toxicological and safety data that demonstrated no adverse effects, even with very high doses of taliglucerase alfa being administered via intravenous infusions. In short term repeat dose studies in rodents and primates and nine-month repeat dose studies in primates, no toxicity was observed at dosage levels of up to 10 times the current dose recommended for GCD in clinical use. Furthermore, no neutralizing antibodies were detected in any of the primates treated in the studies. The presence of neutralizing antibodies would have implied a likelihood of the host rejecting the therapeutic enzyme or reacting to it in a less efficient manner.
Our laboratory and preclinical data demonstrate that taliglucerase alfa has the potential to be an efficacious enzyme replacement therapy for the treatment of Gaucher disease. Data produced from these preliminary development studies show that, relative to Cerezyme, taliglucerase alfa has:
| · | an equivalent to superior level of enzymatic activity; |
| · | enhanced uptake based on observed GlcCer substrate degradation; and |
| · | a prolonged half-life. |
| 14 |
Phase I Clinical Trial
The phase I clinical trial, which we completed in June 2006, was a single-center, non-randomized, open label, dose ranging study designed to evaluate the safety and pharmacokinetics of taliglucerase alfa in healthy subjects. The trial was conducted on healthy subjects over a four-week period in which subjects received three single escalating doses of taliglucerase alfa administered via intravenous infusions.
All doses administered to subjects in the phase I clinical trial, including the highest dose, which was the same dosage currently suggested with respect to the treatment by Cerezyme, demonstrated a strong safety profile. The data from our phase I clinical trial showed that taliglucerase alfa was safe and well tolerated at all doses. There were no serious adverse events and no subjects withdrew from the trial or discontinued treatment due to an adverse event.
In addition, the half-life of taliglucerase alfa was found to be significantly longer than that of Cerezyme, based upon data disclosed publicly by Genzyme, which was consistent with our preclinical data.
Further, as in the laboratory and preclinical testing phase, no neutralizing antibodies or adverse immunological responses were detected in any of the subjects treated in the phase I clinical trial.
Phase III Clinical Trial and Related Studies
After the conclusion of the phase I clinical trial and discussions with the FDA, we applied to commence a pivotal phase III clinical trial of taliglucerase alfa without the requirement to first complete a phase II clinical trial. In April 2007, we received approval from the FDA to initiate a pivotal phase III clinical trial. We submitted to the FDA a request for a special protocol assessment (SPA) of the final design of our pivotal phase III clinical trial for taliglucerase alfa. In July 2007, we reached an agreement with the FDA on the design that we submitted in the SPA request, and in the third quarter of 2007 we initiated enrollment and treatment of naive patients in the phase III clinical trial. In accordance with the terms of the SPA, the phase III clinical trial was a multi-center, world-wide, randomized, double-blind, parallel group, dose-ranging study to assess the safety and efficacy of taliglucerase alfa in 31 treatment-naive patients suffering from Gaucher disease. In the trial, patients were selected randomly for one of two dosing arms (60 U/kg or 30 U/kg) and received intravenous infusions of taliglucerase alfa once every two weeks for a nine-month period. The primary endpoint of the study was a 20% mean reduction from baseline in spleen volume after nine months, as measured by MRI. Major secondary endpoints were an increase in hemoglobin, decrease in liver volume and increase in platelet count. Patients enrolled in the trial were treated in 11 selected leading medical centers throughout Europe, Israel, North America, South America and South Africa. We commenced enrollment and treatment of patients in our phase III clinical trial in the third quarter of 2007 and completed enrollment in the fourth quarter of 2008. We successfully completed the trial in September 2009 and announced positive top-line results of the trial in October 2009 and full study results in February 2010. We submitted an NDA to the FDA in December 2009.
During the third quarter of 2008, we initiated a double blind, follow-on extension study as part of our phase III clinical trial. Eligible patients who successfully completed nine months of treatment in our phase III clinical trial were offered the opportunity to participate in the extension study and continue to receive taliglucerase alfa at the same dose they received in the phase III clinical trial for an additional 15 months in a blinded manner. Accordingly, the extension trial included two treatment groups; one treated with a 60 U/kg dose and the other with a 30 U/kg dose. The major endpoints of the study were spleen volume, liver volume, hemoglobin concentration, platelet count, and chitotriosidase activity. Twenty-six patients were enrolled in the extension trial which was performed in centers throughout Europe, Israel, North America, South America and South Africa. We have also initiated a home care treatment program for patients enrolled in the extension study.
In addition, in the fourth quarter of 2008, we announced the enrollment of the first patient in a worldwide, multi-center, open-label, switch-over trial to assess the safety and efficacy of taliglucerase alfa. The switch-over trial, which was not a prerequisite for approval, was originally designed to include 15 patients with Gaucher disease that are currently undergoing enzyme replacement therapy with imiglucerase (Cerezyme). Due to the shortage of Cerezyme in 2009, after fully enrolling 15 patients, we extended the trial to include additional patients. A total of 26 adult patients were enrolled in the switchover trial. Patients enrolled in the trial were switched from imiglucerase (doses ranging from approximately 10-60 U/kg every other week) to an equivalent dose using the same number of units of taliglucerase alfa over a nine-month period. In February 2012 we reported full results from the trial.
| 15 |
Results of our Phase III Clinical Trial and Extension Trial
We reported positive top-line results of our phase III clinical trial of taliglucerase alfa in October 2009 and full study results in February 2010. In the clinical trial, taliglucerase alfa significantly reduced mean spleen volume after nine months compared with baseline in both treatment groups, thereby meeting the trial's primary endpoint of reduced mean spleen volume after nine months. In February 2012, we announced data from 26 adult patients enrolled in our extension trial. According to the data, after 24 months (nine months in the phase III clinical trial and 15 months in the extension trial), patients continued to demonstrate a significant improvement in all parameters with a similar safety profile as seen in the phase III clinical trial. Furthermore, those patients who were followed specifically for their bone parameters using Quantitative Chemical Shift Imaging (QCSI) MRI continued to show bone marrow improvement over time.
In the phase III clinical trial, the 60 U/kg group demonstrated a statistically significant mean reduction in spleen volume of 38.0% (p<0.0001) and the 30 U/kg group demonstrated a significant mean reduction in spleen volume of 26.9% (p<0.0001). In addition, the primary endpoint was achieved in both treatment groups after only six months of therapy. Patients treated with taliglucerase alfa in the extension trial continued to demonstrate a statistically significant reduction in mean spleen volume after 24 months, compared with baseline, in both treatment groups; reductions of 54.0% (p<0.0001) in the 60 U/kg group and of 41.0% (p<0.0001) in the 30 U/kg group. See Figure 1.
Figure 1. Naive Patient Study: Spleen Volume at 24 Months (30 units/kg (n=12) ; 60 units/kg (n=14)); Mean Change from Baseline (%)
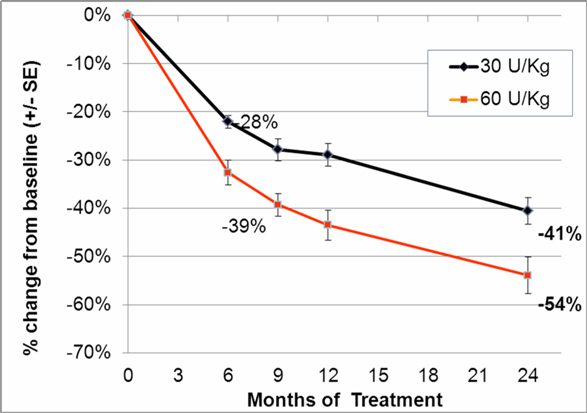
P-value<0.0001
In the phase III clinical trial, statistically significant improvements were also observed for the secondary endpoints, including increase in hemoglobin level, decrease in liver size and increase in platelet count, after nine months when compared to baseline for the 60 U/kg dose. These improvements continued in the extension trial. In the phase III clinical trial, patients in both dosage groups demonstrated mean increases in hemoglobin from baseline; mean increases from 11.4 g/DL to 13.6 g/DL (p<0.0001) (22.2%) in the 60 U/kg group and from 12.2 g/DL to 14.0 g/DL (p<0.0010) (14.8%). In the extension trial, statistically significant mean increases in hemoglobin concentration were also demonstrated by both groups; mean increases from baseline (from 11.6 g/DL to 13.9 g/DL (p<0.0020)) (19.8%) in the 60 U/kg group and mean increases from baseline (from 12.4 g/DL to 13.6 g/DL (p<0.0314)) (9.7%) in the 30 U/kg group. Anemic patients demonstrated a mean increase from baseline in hemoglobin concentration (from 9.5 g/DL to 12.9 g/DL (p<0.0013) (35.8%)). See Figure 2.
| 16 |
Figure 2. Naive Patient Study: Hemoglobin Concentration at 24 Months (30 U/kg n=12, 60 U/kg n=14, Anemic patients n=10); Mean Change from Baseline (%)
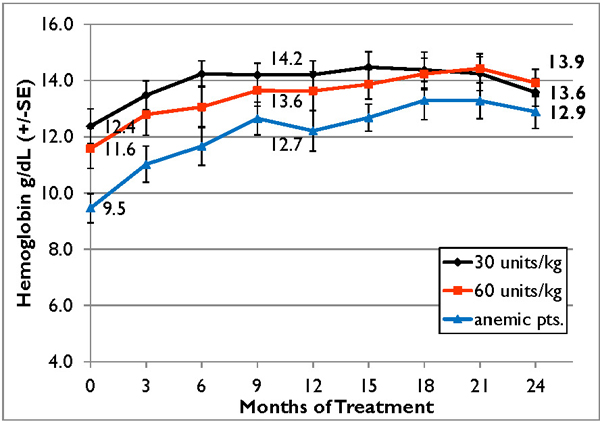
P value change from baseline for 30U dose <0.0314
P value change from baseline for 60U dose <0.0020

In the phase III clinical trial, patients in both dosage groups demonstrated mean increases in liver volume; reductions of 11.1% (p<0.0001) in the 60 U/kg group and of 10.48% (p<0.0041) in the 30 U/kg group. In the extension trial, both groups demonstrated statistically significant mean reductions in liver volume; reductions of 17.5% (p<0.0003) in the 60 U/kg group and of 20.6% (p<0.0001) in the 30 U/kg group; patients with Hepatomegaly demonstrated a reduction in liver volume of 25.2% (p<0.0001). See Figure 3.
| 17 |
Figure 3. Naive Patient Study: Liver Volume at 24 Months (30 units/kg (n=12), 60 units/kg (n=14), Hepatomegaly (n=12)); Mean Change from Baseline (%)
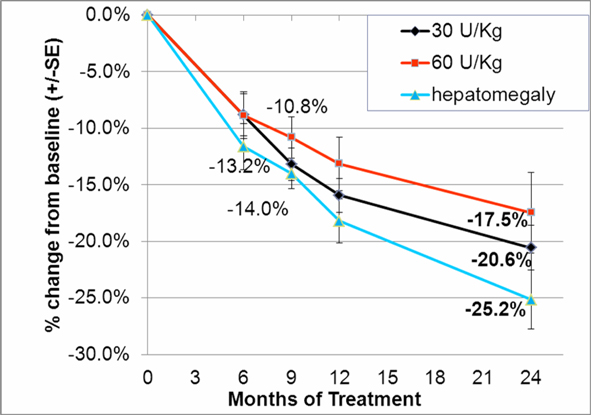
In the phase III clinical trial, patients in both dosage groups demonstrated mean increases in platelet count; an increase in platelet count of 41,494 ml or 72.1% (p=0.0031) in the 60 U/kg group and a nominal increase of 11,427 ml or 13.7% (p=0.0460) in the 30 U/kg group. Accordingly, the lower dose group did not meet the secondary endpoint relating to platelet count. However, in the extension trial, both groups demonstrated statistically significant mean increases in platelet count for both groups; increases from 69,043 to 141,071 (p<0.0016) in the 60 U/kg group and from 64,900 to 93,333 (p<0.0105) in the 30 U/kg group. See Figure 4.
| 18 |
Figure 4. Naive Patient Study: Platelet Count at 24 Months (30 U/kg (n=12), 60 U/kg (n=14)); Mean Change from Baseline (%)
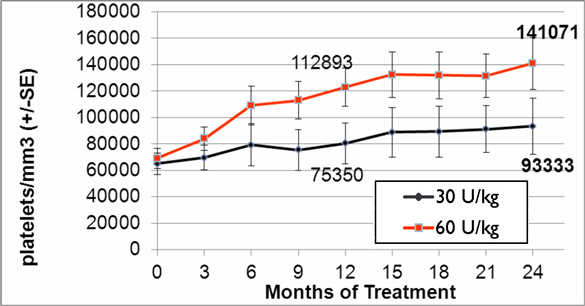
P value change from baseline for 30U dose <0.0105
P value change from baseline for 60U dose <0.0016
Last, 30 patients in the phase III clinical trial had chitotriosidase measurements, a biomarker for clinical symptoms of Gaucher disease. In these patients, chitotriosidase decreased from baseline in both the 30U/kg and 60U/kg groups by 47.3% and 58.4%, respectively. In the extension trial, statistically significant mean reductions in chitotriosidase activity were demonstrated by both groups; a reduction of 76% (p<0.0001)) in the 60 U/kg group and of 61% (p<0.0001) in the 30 U/kg group. See Figure 5.
Figure 5. Naive Patient Study: Chitotriosidase Activity at 24 Months (30 U/kg (n=12), 60 U/kg (n=13*)); Mean Change from Baseline (%)
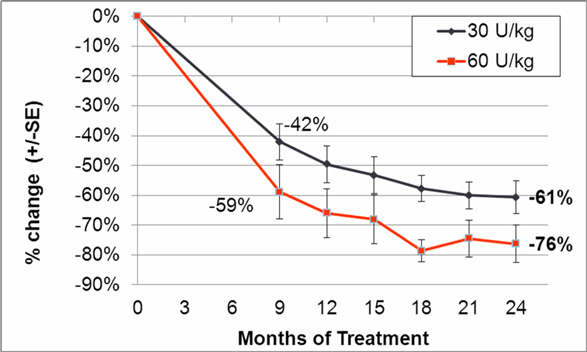
P value for both dose groups <0.0001
*1 patient did not have chitotriosidase activity; tested for CCL18 which showed improvement
| 19 |
The safety analysis for both treatment groups in the phase III clinical trial showed that taliglucerase alfa was well tolerated and no serious or severe adverse events were reported. Two patients in the trial developed antibodies to taliglucerase alfa and no patients developed neutralizing antibodies. In addition, two patients experienced hypersensitivity reactions to taliglucerase alfa. No anti-taliglucerase antibodies were detected in these patients and both reactions were treated in the physicians’ clinic and reversed. The safety analysis in the extension trial for both treatment groups also demonstrates that taliglucerase alfa was well tolerated, and no drug related serious adverse events were reported. Two patients who participated in the extension trial developed neutralizing IgG antibodies that were determined to be positive in an in vitro assay, but were determined to be negative in a cell-based assay. In addition, one patient in the 60 U/kg dose group experienced a hypersensitivity reaction during month 10 of treatment. Treatment of this patient has been continued with premedication for an additional 32 months without any treatment related adverse event reported.
In the phase III clinical trial, most adverse events were considered unrelated to taliglucerase alfa. The most frequent mild to moderate adverse event was headache. Other mild to moderate adverse events included dizziness, muscle spasm, chest discomfort, nausea, skin irritation and arthalgia. Adverse events in the extension trial included headache, pruritus, abdominal pain, infusion related reactions (dizziness, chills, nausea), muscle spasm and fixed drug eruption.
Results of Our Switchover Trial
In November 2010, we announced positive preliminary data from the first 15 patients that completed our switchover trial of taliglucerase alfa, and in February 2012 we reported full results from the trial. The results of the trial demonstrate that over a nine-month treatment period of the study, patients remained stable with regard to the efficacy endpoints—spleen volume, liver volume, platelet count and hemoglobin concentration—after switching to taliglucerase alfa from imiglucerase. See Figures 6-9.
| 20 |
| Figure 6. Platelet Count (Stratified by Dose) | Figure 7. Hemoglobin Concentration (Stratified by Dose) | |
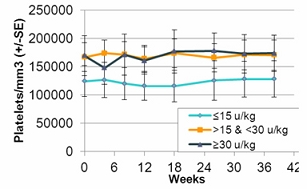 |
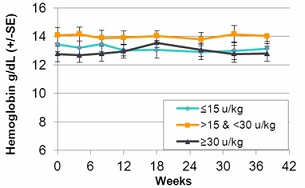 | |
| Figure 8. Spleen Volume (Stratified by Dose) | Figure 7. Liver Volume (Stratified by Dose) | |
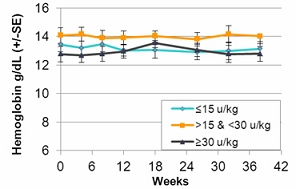 |
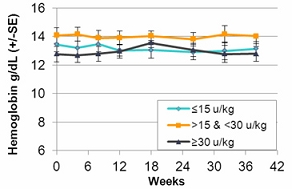 |
The safety analysis presented for the switchover trial demonstrates that taliglucerase alfa was well tolerated, and no drug related serious adverse events were reported. One patient developed neutralizing IgG antibodies that were determined to be positive in an in vitro assay, and were determined to be negative in a cell-based assay. Another patient experienced a hypersensitivity reaction, which was treated in a physician’s office and resolved. The patient declined to continue infusions with premedication. Additionally, all drug-related adverse effects were mild or moderate and transient in nature.
Other Drug Candidates in Our Pipeline
We are developing other innovative recombinant therapeutic proteins to be expressed by our ProCellEx protein expression system, with an emphasis on treatments for which there are large, established pharmaceutical markets and where our proprietary protein expression system enables us to develop and commercialize recombinant proteins that are patent-protected and therapeutically equivalent or superior to the existing treatments. We select additional therapeutic candidates for development by testing candidates in-house and through collaborations with academic partners. We have identified several product candidates oriented towards specialty disease and therapeutic market segments, including: (1) PRX-102, a therapeutic protein candidate for the treatment of Fabry disease, a rare, genetic lysosomal disorder in humans; (2) PRX-105, a plant cell expressed pegylated recombinant acetylcholinesterase product candidate for biodefense and other indications; (3) PRX-106, or pr-antiTNF, a plant cell expressed recombinant fusion protein made from the soluble form of the human TNF receptor (TNFR) and an antibody portion, which is being developed as a treatment of certain immune diseases such as rheumatoid arthritis, juvenile idiopathic arthritis, ankylosing, spondylitis, psoriatic arthritis and plaque psoriasis; (4) an orally administrated glucocerebrosidase enzyme for treating Gaucher disease patients utilizing the oral delivery of the recombinant enzyme produced within carrot cells; and (5) two additional undisclosed therapeutic proteins, both of which are being evaluated in animal studies. We participated in a pre IND meeting with respect to one of the undisclosed product candidates in the first quarter of 2012 and a pre IND meeting for the second candidate is planned for later this year. In March 2010, we initiated a preliminary phase I clinical trial of PRX-105 which we completed in June 2010. We are currently preparing for further efficacy trials of this product candidate in larger animals. In our preclinical studies we utilized an analogue to nerve gas. However, we anticipate that we will use live nerve gas rather than an analogue in the proposed additional efficacy trials in animals. In December 2010, we held a pre- IND meeting with the FDA with respect to PRX-102. We completed preclinical trials in small and large animals with respect to PRX-102, and we expect to submit an IND to the FDA during the second quarter of 2012 in connection with an anticipated phase I/II clinical study of PX-102 in Fabry patients, and to initiate the trial once the IND is approved, if at all. In December 2011 we held a pre IND meeting with respect to PRX 106 and we expect to submit an IND around the fourth quarter of 2012. We are also conducting pre clinical trials of our orally-administrated glucocerebrosidase enzyme for the treatment of Gaucher disease in small and large animals. Last, we are conducting research programs in the fields of enzymes, monoclonal antibodies, cytokines and vaccines.
| 21 |
PRX-102
We are developing PRX-102, our proprietary plant cell expressed modified version of the recombinant alpha-GAL-A protein, a therapeutic enzyme for the treatment of Fabry disease. Fabry disease is a rare, hereditary, genetic lysosomal storage disorder in humans caused by an X-lined deficiency of the alpha-GAL-A enzyme. Fabry disease causes harmful accumulations of lipids in the kidneys, automatic nervous system and cardiovascular system that result in the risk of heart attack and stroke, and can be life-threatening. Fabry disease affects more than 8,000 people globally. We believe that the treatment of Fabry disease is a specialty clinical niche with the potential for high growth. Currently there are two drugs available on the market to treat Fabry disease. Fabrazyme, made by Genzyme (acquired by Sanofi), was approved for the treatment of Fabry disease in the European Union in 2001 and the United States in 2003. Sanofi reported €109 million (approximately $143.2 million) in worldwide sales of Fabrazyme in 2011, a growth of 9.4% compared to 2010. According to Genzyme, supplies of Fabrazyme have also been affected by the production interruptions that have occurred since 2009. The other approved drug for the treatment of Fabry disease in the European Union is Replagal, which is sold by Shire. Shire reported $475.2 million in sales of Replagal in 2011, compared to $351.3 million in sales of Replagal in 2010. According to public reports by Shire, it filed a BLA with the FDA for Replagal in the United States in December 2009. Shire subsequently received Fast Track designation for Replagal, withdrew the BLA in 2010 and in October 2010, initiated a rolling BLA submission. Shire has reported that it withdrew the December 2009 filing in August 2010 in order to update the filing with additional clinical information. According to public reports by Shire, it submitted a a rolling BLA with the FDA for Replagal in the United States in November 2011, which the FDA has accepted and a priority review. The FDA has assigned Replagal an action date of May 17, 2012.
We completed the animal evaluation testing phase of the development of PRX-102, which tests are based on a well established mouse model for Fabry disease. In pre-clinical studies, PRX-102 demonstrated preliminary efficacy in a Fabry animal model. Chemical modifications made to PRX-102 improved the enzyme’s activity and stability resulting in prolonged activity profiles and enhanced bioavailability in animals. The modifications also have the potential to decrease the immunogenicity of the enzyme, which is a major drawback of currently approved therapies for Fabry disease. We currently expect to submit an IND to the FDA during the second quarter of 2012 in connection with an anticipated phase I/II clinical study of PRX-102 and to initiate the trial once the IND is approved, if at all. As was the case in our development of taliglucerase alfa, our development of PRX-102 involves the expression by our proprietary protein expression system of a naturally occurring enzyme to be used in enzyme replacement therapy for the treatment of Fabry disease. Based on the pre IND meeting we held with FDA in 2010, the experience with taliglucerase alfa and the experience of other companies developing enzyme replacement therapies for Fabry disease, we have reason to believe that, if favorable data is accumulated in preclinical and phase I/II clinical trials, the FDA may allow us to proceed directly with a pivotal phase III clinical trial. However, there can be no assurance that we will initiate phase I/II clinical trials and if we do, that such trials will result in favorable data. In addition, there can be no assurance that the FDA will allow us to proceed directly with a phase III clinical trial after completion of a phase I/II clinical trial.
Acetylcholinesterase
In August 2007, Protalix Ltd. licensed the rights to certain technology under a research and license agreement with Yissum Research and Development Company, or Yissum, and the Boyce Thompson Institute, Inc., or Boyce Thompson. Pursuant to the agreement, we are developing PRX-105, a proprietary plant cell-based acetylcholinesterase (AChE) and its molecular variants for the use in several therapeutic and prophylactic indications, as well as in a biodefense program and an organophosphate-based pesticide treatment program. To date, our in vitro experiments of PRX-105 have shown that the acetylcholinesterase enzyme expressed in our ProCellEx protein expression system demonstrates promising biological activity on biochemical and cellular levels. In addition, early animal studies demonstrated that the acetylcholinesterase expressed in our ProCellEx protein expression system was able to successfully treat animals exposed to the nerve gas agent analogues, both when injected with our acetylcholinesterase product candidate immediately before exposure or when injected after exposure. We filed an IND with the FDA for PRX-105, our plant cell expressed pegylated recombinant acetylcholinesterase enzyme (AChE) product candidate, during the last quarter of 2009, and in March 2010 we initiated a preliminary phase I clinical trial of PRX-105. The trial established the pharmokinetics of PRX-105 and demonstrated that single dose intravenous administration of PRX-105 is safe and well tolerated. We are currently preparing for further efficacy trials of PRX-105 in larger animals. In our preclinical studies we utilized an analogue to nerve gas. However, we anticipate that we will use live nerve gas in the proposed additional efficacy trials in animals. In March 2010 we initiated a preliminary phase I clinical trial of PRX-105 which we completed in June 2010. In December 2010, we held a pre-investigational new drug, or IND, meeting with the FDA with respect to PRX-105.
| 22 |
Under the terms of the agreement, Yissum and Boyce Thompson granted us an exclusive, worldwide right and license to certain technology, including patents and certain patent applications relating to AChE for the therapeutic and prophylactic indications as well as an exclusive license not limited to such indications with respect to certain of those patents and patent applications. As consideration for the license, we are obligated to pay Yissum and Boyce Thompson, collectively, an annual, non-refundable initial maintenance fee of $20,000, commencing on the fourth anniversary of the execution of the agreement, which is subject to a 12% annual increase. In addition, we are obligated to make royalty payments equal to varying low, single-digit percentages of net sales of products under the agreement. These royalty rates are evaluated on a country-by-country basis, and are subject to reduction if a third party commercializes a competing product or commercializes an authorized generic version of the applicable product, subject to certain conditions. We also have the right to grant sublicenses relating to the licensed technology under the agreement, subject to the payment of sublicensing fees. The fees payable in connection with any sublicense are equal to varying percentages, in the low-teens through the low-twenties, of the consideration we receive in connection with the sublicense, depending on the level of clinical development of the product at the time we enter into the sublicense. Last, we are obligated to pay Yissum and Boyce Thompson, collectively, milestone payments equal to $700,000, in the aggregate, upon the achievement of certain milestones under the license agreement.
The license agreement remains in effect until the expiration of all obligations to Yissum and Boyce Thompson under the agreement, determined on a country-by-country basis. We have the right to terminate the agreement for any reason upon 60 days’ prior written notice to Yissum and Boyce Thompson. Subject to certain conditions, Yissum and Boyce Thompson may terminate the agreement immediately upon written notice to us in connection with certain events relating to bankruptcy, lapses in our insurance coverage, failures to defend against third party claims or claims we may make regarding the validity or enforceability of any licensed patent. We or Yissum and Boyce Thompson may terminate the agreement within 60 days after receiving written notice if the non-terminating party passes a resolution for a voluntary wind up, if a receiver or liquidator is appointed for the non-terminating party, or the non-terminating party enters into an insolvency or bankruptcy proceeding. In addition, either party may terminate the agreement due to a material breach by the other party if the breaching party is unable to cure the breach within 60 days after receiving written notice of the breach from the non-breaching party. Any termination of the agreement will result in a loss of our rights to the licensed technology, which will revert back to Yissum and Boyce Thompson.
We are currently in discussions with different U.S. civil and military organizations regarding certain grants for which our acetylcholinesterase program is eligible. We anticipate applying for specific U.S. grants during 2012 to support the further development of our acetylcholinesterase program.
In addition, we are researching other indications for which PRX-105 may serve as a therapy. A paper entitled: "Adaptive Alternative Splicing Correlates with Less Environmental Risk of Parkinsonism," has been published in the Journal of Neurodegenerative Diseases (Neurodegener Dis. 2012;9(2):87-98. Epub 2011 Oct 27). The research includes an investigation of the effect of PRX-105 on alternative splicing patterns in the striatum, which may confer protection in Parkinson's disease. The research highlights that a splice shift, either inherited or induced by environmental factors (e.g. neurotoxins), from the more common 'synaptic form' of acetylcholinesterase (AChE-S) to the monomeric 'read-through' variant of AChE (AChE-R), is important in conferring protection against Parkinson-like symptoms. Furthermore, according to the article, intravenous injection with PRX-105 induced a protective gene expression profile in the striatum of the brain. We recently filed a joint PCT patent application entitled: "Methods for Treating or Preventing Parkinson's Disease," with the Hebrew University. These filings are part of our continuing collaboration with Yissum.
| 23 |
pr-antiTNF
pr-antiTNF is an antiTNF (Tumor, Necrosis Factor) protein that we are expressing through our proprietary ProCellEx system. We have designed this antiTNF as pr-antiTNF. pr-antiTNF is a candidate for the treatment of certain autoimmune diseases such as rheumatoid arthritis, juvenile idiopathic arthritis, ankylosing, spondylitis, psoriatic arthritis and plaque psoriasis. Our pr-antiTNF product candidate has an amino acid sequence that is similar to Enbrel which is one of the treatments for patients of those diseases. Amgen Inc. reported total sales of Enbrel of $3.7 billion in the United States and Canada for 2011 and Pfizer has reported total sales of Enbrel outside of the United States and Canada of $3.7 billion for 2011.
pr-antiTNF is a plant cell-expressed recombinant fusion protein made from the soluble form of the human TNF receptor (TNFR), fused to the Fc component of a human antibody domain. pr-antiTNF has an identical amino acid sequence to Enbrel and our in vitro and preclinical animal studies have demonstrated that pr-antiTNF exhibits similar activity to Enbrel. Specifically, pr-antiTNF binds TNF thereby inhibiting it from binding to cellular surface TNF receptors and protects L929 cells from TNF-induced apoptosis in a dose-dependent manner. In a proof-of-concept in vivo study using an established arthritis animal model, pr-antiTNF, when injected in mice, significantly improved the clinical arthritis parameters associated with this accepted arthritis mouse model, including joint inflammation, swelling and tissue degradation. We intend to conduct additional animal studies to collect additional data to form a basis for a discussion with the FDA to explore the regulatory pathway for our antiTNF program. Patents for the Enbrel start to expire as early as 2012, and we expect to use our cost effective manufacturing platform to facilitate entry into this market upon approval of our pr-antiTNF product, if at all. In December 2011, we held a pre IND meeting with respect to pr-antiTNF, and we expect to submit an IND around year end.
Oral Glucocerebrosidase
We are developing an orally delivered glucocerebrosidase (GCD) enzyme for the potential treatment of Gaucher disease. Oral GCD is a plant cell-expressed form of GCD that is naturally encapsulated within carrot cells that are genetically engineered to express the GCD enzyme. Plant cells have the unique attribute of a cellulose cell wall which makes them resistant to enzyme degradation when passing through the digestive tract. The plant cell itself serves as a delivery vehicle to transport the enzyme in active form to the bloodstream. We are currently conducting large animal studies of oral GCD to demonstrate the stability of the enzyme in the cell and the capacity of the cellulose wall to protect the enzyme against degradation in the digestive tract in an in vitro model of the stomach and intestines. We intend to plan a phase I clinical trial of oral glucocerebrosidase in healthy individuals who are carriers of Gaucher disease and who show reduced enzymatic activity at baseline based on the final results of the animal studies.
Commercialization Agreement
On November 30, 2009, Protalix Ltd. and Pfizer entered into a license and supply agreement pursuant to which Pfizer was granted an exclusive, worldwide license to develop and commercialize taliglucerase alfa. Under the terms and conditions of the Pfizer Agreement, Protalix Ltd. retained the right to commercialize taliglucerase alfa in Israel. In connection with the execution of the Pfizer Agreement, Pfizer made an upfront payment to Protalix Ltd. of $60.0 million in connection with the execution of the agreement and subsequently paid to Protalix Ltd. an additional $5.0 million upon our filing of a proposed pediatric investigation plan to the Pediatric Committee of the EMA. Protalix Ltd. is also eligible to receive potential milestone payments of up to $50.0 million, in the aggregate, for the successful achievement of other regulatory-related milestones and to payments equal to 40% of the net profits earned by Pfizer on sales of taliglucerase alfa. In calculating the net profits, there are certain agreed upon limits on the amounts that may be deducted from gross sales for certain expenses and costs of goods sold. Protalix Ltd. retained the manufacturing rights to taliglucerase alfa and Pfizer and Protalix Ltd. have agreed to a specific allocation of the responsibilities for the continued development efforts for taliglucerase alfa. Protalix Ltd. will manufacture all of the taliglucerase alfa needed for all purposes under the agreement and Pfizer will purchase the taliglucerase alfa from Protalix Ltd., subject to certain terms and conditions. The Pfizer Agreement also provides for reimbursement by Pfizer of certain costs to be incurred by Protalix Ltd.
In connection with the upfront and milestone payments made under the Pfizer Agreement, Protalix Ltd. has paid a sublicense fee equal to $1.6 million to the academic institution from who it licensed certain technology relating to taliglucerase alfa. Future milestone payments will be subject to a 2.5% royalty, and all of revenues generated under the agreement will be subject to a 0.75% royalty payable to the same institution until 2016, when a patent related to taliglucerase alfa licensed to us will expire. We are also required to pay a royalty equal to 3% of the revenues of taliglucerase alfa Pfizer records under the Pfizer Agreement to the OCS.
On August 10, 2010, Pfizer entered into a $30 million short-term supply agreement with the Ministry of Health of Brazil pursuant to which Protalix and Pfizer have provided taliglucerase alfa to the Ministry of Health of Brazil for the treatment of patients with Gaucher disease. During the second and fourth quarters of 2011, the Ministry of Health of Brazil requested that Pfizer consider the replacement of certain vials that might expire during 2012. As a result, Pfizer recorded an allowance for sales returns. If and when such vials are replaced, revenues will be recorded upon the supply of such replaced vials, and if it is determined that there is no longer a need for the replacement of vials, the allowance will be reversed and the revenues will be recognized accordingly.
| 24 |
In addition, we and the Ministry of Health of Brazil are in discussions relating to a possible long-term supply agreement that contemplates, among other matters, providing certain components of our manufacturing technology to the Ministry of Health of Brazil for implementation by it in Brazil. At this time, we are unable to assess whether these discussions will result in an agreement and there can be no assurance that we will be able to enter into such an agreement on favorable terms, if at all. In any event, we do not expect to enter into a long-term supply agreement with the Ministry of Health of Brazil until we receive marketing approval of taliglucerase alfa from the FDA or ANVISA, if at all.
We will be subject to a withholding tax on the U.S. revenue source portion of the payments made to us for our share of Pfizer’s in net profits under the Pfizer Agreement. Currently, the withholding tax rate is 15%.
Strategic Collaborations
Teva Pharmaceutical Industries
In September 2006, we entered into a Collaboration and Licensing Agreement with Teva for the development and manufacture of two proteins to be identified by Teva and us using our ProCellEx protein expression system. The agreement also identifies additional matters for collaboration between Teva and us. Subsequently, two proteins were identified to be researched and developed under the agreement but in 2009, both of the projects were terminated for commercial reasons. Pursuant to the agreement, we have agreed to collaborate on certain additional matters regarding proteins, including the research and development of proteins utilizing our ProCellEx protein expression system. See “Risk Factors—Our strategy, in many cases, is to enter into collaboration agreements with third parties to leverage our ProCellEx system to develop product candidates. If we fail to enter into these agreements or if we or the third parties do not perform under such agreements or terminate or elect to discontinue the collaboration, it could have a material adverse affect on our revenues.”
Weizmann Institute of Science
In March 2006, Protalix Ltd. entered into a research and license agreement with the Yeda Research and Development Company Limited, or Yeda, the technology transfer arm of the Weizmann Institute of Science. Under the terms of the agreement, Yeda agreed to use its technology to design a next generation of glucocerebrosidase (GCD) for the treatment of Gaucher disease that can be expressed using our ProCellEx protein expression system and that may have certain benefits over the first generation treatments used today. The technology licensed from Yeda provides a methodology for the rational design of an improved drug for the treatment of Gaucher disease by enzyme replacement therapy, based on the three-dimensional crystal structure of glucocerebrosidase that was solved by scientists from the Weizmann Institute of Science. Yeda has granted us an exclusive worldwide license to use their technology and discoveries for the development, production and sale of enzymatically active mutations of glucocerebrosidase (GCD) and derivatives thereof for the treatment of Gaucher disease. Under the terms of the agreement, we are required to take all necessary steps to develop and commercialize the products subject to the agreement.
As consideration for the license, we agreed to pay Yeda a fixed research budget amount, subject to certain conditions. We have since completed the research phase of the arrangement with Yeda. Accordingly, we are no longer making any research-related payments to Yeda under the agreement. In addition, we are obligated to make an annual non-refundable license fee of $10,000 during the term of the agreement, commencing on the fifth anniversary of the execution of the agreement until, and including, the 19th anniversary thereof. We are also obligated to make royalty payments equal to varying low, single-digit percentages of net sales of products under the agreement. Sublicenses relating to the licensed technology may be granted under the agreement, subject to the payment of sublicensing fees. The fee for any sublicense is equal to a percentage, ranging from the low-teens through the low-twenties, of the consideration we receive in connection with the sublicense, depending on the level of clinical and regulatory development of the products under the agreement at the time we enter into the sublicense.
The license agreement remains in effect until the earlier of the expiration of the last patent licensed under the agreement or if there are no commercial sales of any products for a continuous period of 20 years. Yeda may modify the exclusivity component of the agreement by written notice to us and without our consent. Yeda may terminate the agreement by written notice to us if we fail to satisfy any one or more specified milestones, and we fail to cure any such failure within a certain time period after we receive the notice. Yeda is not entitled to exercise this termination right if we demonstrate that we are making all necessary efforts to achieve such milestones, that our inability to satisfy the milestones is due to factors beyond our control, and that the total delay with respect to any one milestone does not exceed 12 months and the total cumulative delay in respect of all milestones has not exceeded 30 months. Yeda may also terminate the agreement if we contest the validity of any of the patents included in the agreement. We or Yeda may terminate the agreement due to a material breach by the other party if the breach is unable to be cured or, if curable, the breach is not cured within 21 days after the breaching party’s receipt of written notice of the breach from the non-breaching party. In addition, either party may terminate the agreement in connection with certain events relating to a wind up or bankruptcy.
| 25 |
Intellectual Property
We maintain a proactive intellectual property strategy which includes patent filings in multiple jurisdictions, including the United States and other commercially significant markets. As of December 31, 2011, we hold, or have license rights to, 31 patents and 81 pending patent applications with respect to various compositions, methods of production and methods of use relating to our ProCellEx protein expression system and our proprietary product pipeline. As of December 31, 2011, we also hold, with a third party, one joint patent and one joint patent application, and licensed rights to six patents and seven patent applications.
Our competitive position and future success depend in part on our ability, and that of our licensees, to obtain and leverage the intellectual property covering our product candidates, know-how, methods, processes and other technologies, to protect our trade secrets, to prevent others from using our intellectual property and to operate without infringing the intellectual property of third parties. We seek to protect our competitive position by filing United States, European Union, Israeli and other foreign patent applications covering our technology, including both new technology and improvements to existing technology. Our patent strategy includes obtaining patents, where possible, on methods of production, compositions of matter and methods of use. We also rely on know-how, continuing technological innovation, licensing and partnership opportunities to develop and maintain our competitive position.
As of December 31, 2011, our patent portfolio consists of several patent families (consisting of patents and/or patent applications) covering our technology, protein expression methodologies and system and product candidates, as follows:
| · | With respect to our ProCellEx protein expression system, we have been issued, and hold licensed rights to, patents in the United States, Australia, the European Union, Israel, Canada, the Czech Republic, Hungary, Japan, Poland, Mexico, Hong Kong and India, and to eight pending patent applications. Among other things, the patents cover the methods that we use for culturing and harvesting plant cells and/or tissues in consecutive cycles. The issued patents in this patent family are expected to expire in 2017. |
| · | With respect to our ProCellEx protein expression system, we also hold 14 patent applications relating to the large scale production of proteins in cultured plant cells. The patents to issue in the future based on the pending patent applications in this patent family are expected to expire in 2028. |
| · | We hold a patent family containing nine granted patents in India, South Africa, Russia, Australia, China, the United States and the Ukraine, and 35 patent applications, relating to the production of glycosylated lysosomal proteins in our plant culture platform, particularly proteins having a terminal mannose glycosylation, including taliglucerase alfa. The issued patents and any patents to issue in the future based on pending patent applications in this patent family are expected to expire in 2024 and 2028. |
| · | We hold a patent family containing two granted and two pending patent applications relating to a system and method for production of antibodies in a plant cell culture, and antibodies produced in such a system. The patents to issue in the future based on the patent applications in this patent family are expected to expire in 2025. |
| · | We hold a patent family containing two issued patent in South Africa and Singapore and 12 pending patent applications relating to a new method for delivering active recombinant proteins systemically through oral administration of transgenic plant cells. The issued patents and any patents to issue in the future based on patent applications in this patent family are expected to expire in 2026. |
| · | We hold a patent family containing one granted patent in South Africa and six pending patent applications relating to saccharide containing protein conjugates. The patents to issue in the future based on the patent applications in this patent family will expire in 2028. |
| · | Our patent portfolio includes a patent that we co-own that covers human glycoprotein hormone and chain splice variants, including isolated nucleic acids encoding these variants. More specifically, this patent covers a new splice variant of human FSH. This patent was issued in the United States and is expected to expire in 2026. |
| 26 |
| · | With respect to taliglucerase alfa, we have licensed the rights to two patents from Virginia Tech Intellectual Properties, Inc., or Virginia Tech, that are expected to expire in 2016. We also hold the licensed rights from Yeda to one granted and two pending patent applications with respect to the research and development of the glucocerebrosidase protein which are expected to expire in 2024. |
| · | With respect to acetylcholinesterase, we have licensed the rights to three patents issued in the United States that are expected to expire in 2014, 2017 and 2021, and to five patent applications from Yissum. |
We are aware of U.S. patents, and corresponding international counterparts of such patents, owned by third parties that contain claims covering methods of producing GCD. We do not believe that, if any claim of infringement were to be asserted against us based upon such patents, taliglucerase alfa would be found to infringe any valid claim under such patents. However, there can be no assurance that a court would find in our favor or that, if we choose or are required to seek a license to any one or more of such patents, a license would be available to us on acceptable terms or at all.
In April 2005, Protalix Ltd. entered into a license agreement with Icon Genetics AG, or Icon, pursuant to which we received an exclusive worldwide license to develop, test, use and commercialize Icon’s technology to express certain proteins in our ProCellEx protein expression system. Under the terms of the agreement, we are also entitled to a non-exclusive worldwide license to make and have made other proteins expressed by using Icon’s technology in our technology. As consideration for the license, we are obligated to make royalty payments equal to varying low, single-digit percentages of net sales of products by us, our affiliates, or any sublicensees under the agreement. In addition, we are obligated to make milestone payments equal to $350,000, in the aggregate, upon the achievement of certain milestones.
Our license agreement with Icon remains in effect until the earlier of the expiration of the last patent under the agreement or, if all of the patents under the agreement expire, 20 years after the first commercial sale of any product under the agreement. Icon may terminate the agreement upon written notice to us that we are in material breach of our obligations under the agreement and we are unable to remedy such material breach within 30 days after we receive such notice. Further, Icon may terminate the agreement in connection with certain events relating to a wind up or bankruptcy, if we make a general assignment for the benefit of our creditors, or if we cease to conduct operations for a certain period. Icon may also terminate the exclusivity granted to us by written notice if we fail to reach certain milestones within a designated period of time. Notwithstanding the termination date of the agreement, our obligation to pay royalties to Icon under the agreement may expire prior to the termination of the agreement, subject to certain conditions.
In January 2005, Protalix Ltd. entered into a license agreement with Virginia Tech, pursuant to which we received a non-exclusive worldwide license to make, have made, use, sell, offer for sale and import certain of Virginia Tech’s patents. As consideration for the license, we made a one-time license fee payment to Virginia Tech, and we are obligated to make royalty payments equal to varying low, single-digit percentages of net sales of licensed products by Protalix Ltd., its subsidiaries and/or their affiliates. Upon commercialization of a licensed product, the royalty payment is subject to a low, annual minimum amount. In addition, we are obligated to make milestone payments equal to $150,000, in the aggregate, upon the achievement of certain milestones. We have the right to grant sublicenses under the agreement.
Our license agreement with Virginia Tech remains in effect until the earlier of the expiration of the last patent under the agreement or 10 years after the first commercial sale of any licensed product. Virginia Tech may terminate the agreement upon written notice to us that we are in material breach of our obligations under the agreement if we are unable to remedy such material breach within a fixed number of days after we receive such notice, which number may be doubled if we are making good faith efforts to achieve a cure and the extension will not increase the damages suffered by Virginia Tech. We have the right to terminate the agreement at any time upon prior written notice delivered an agreed-upon number of days prior to the date of termination.
Manufacturing
We are obligated to manufacture all of the taliglucerase alfa drug product needed under the Pfizer Agreement, subject to certain terms and conditions. Our drug product candidates, including taliglucerase alfa, must be manufactured in a sterile environment and in compliance with cGMPs set by the FDA and other relevant foreign regulatory authorities. We use our current facility, which has approximately 20,000 sq/ft of clean rooms built according to industry standards, to develop, process and manufacture taliglucerase alfa and other recombinant proteins We intend to use our current manufacturing space to produce all of the taliglucerase alfa we need in the near future, included the taliglucerase alfa to be purchased by Pfizer. Current capacity of our facility can serve approximately 30% of the Gaucher disease patients that are currently under treatment. We intend to expand our current facility in order to reach a capacity of approximately 80% of the Gaucher disease patients that are currently under treatment and to house the laboratory space necessary for further development of other product candidates in our pipeline. Total expected cost for such expansion is currently estimated to be approximately $25 million and the process is expected to be completed during 2013.
| 27 |
In February 2010, the Israeli MOH completed a successful GMP audit of our manufacturing facilities in Carmiel, Israel. The audit was performed as part of the Israeli MOH’s evaluation of our manufacturing process for taliglucerase alfa. On February 20, 2011, we received a letter from the FDA notifying us that the FDA had completed its review of the Establishment Inspection Report in connection with the FDA’s inspection of our Carmiel facility and that the FDA had classified our facility as acceptable. In June 2011, ANVISA completed a successful GMP audit of the facility and determined that the facility is acceptable. In December 2011, after the taliglucerase alfa NDA was filed with the EMA, the IMB completed a successful GMP audit of our facility and issued a Certificate of GMP Compliance of a Manufacturer for the facility. The IMB certificate is accepted by all health authorities in the European Union under the European Union's centralized marketing authorization procedure, and by authorities of several other countries that recognize EU certification.
We have engaged a contract manufacturer in Europe to act as an additional source of fill and finish activities for taliglucerase alfa. According to our agreement with Pfizer, Pfizer will be responsible for the fill and finish activities for taliglucerase alfa.
Our current facility in Israel has been granted “Approved Enterprise” status, and we have elected to participate in the alternative benefits program. Our facility is located in a Zone A location, and, therefore, our income from the Approved Enterprise will be tax exempt in Israel for a 10-year period commencing with the year in which we first generate taxable income from the relevant Approved Enterprise. We currently anticipate that the benefits of this program will be available to Protalix Ltd. until 2017 and, accordingly, we expect to be entitled to similar tax benefits for a number of years thereafter. To remain eligible for these tax benefits, we must continue to meet certain conditions, and if we increase our activities outside of Israel, for example, by future acquisitions, such increased activities generally may not be eligible for inclusion in Israeli tax benefit programs. In addition, our technology is subject to certain restrictions with respect to the transfer of technology and manufacturing rights. “See Risk Factors—The manufacture of our products is an exacting and complex process, and if we or one of our materials suppliers encounter problems manufacturing our products, it will have a material adverse effect on our business and results of operations.”
Raw Materials and Suppliers
We believe that the raw materials that we require throughout the manufacturing process of our current and potential drug product candidates are widely available from numerous suppliers and are generally considered to be generic industrial biological supplies. We rely on a single approved supplier for certain materials relating to the current expression of our proprietary biotherapeutic proteins through ProCellEx. We have identified additional suppliers for most of the materials required for the production of our product candidates.
Development and regulatory approval of our pharmaceutical products are dependent upon our ability to procure active ingredients and certain packaging materials from sources approved by the FDA and other regulatory authorities. Since the FDA and other regulatory approval processes require manufacturers to specify their proposed suppliers of active ingredients and certain packaging materials in their applications, FDA approval of a supplemental application to use a new supplier in connection with any drug candidate or approved product, if any, would be required if active ingredients or such packaging materials were no longer available from the specified supplier, which could result in manufacturing delays. From time to time, we intend to continue to identify alternative FDA-approved suppliers to ensure the continued supply of necessary raw materials.
Competition
The biotechnology and pharmaceutical industries are characterized by rapidly evolving technology and significant competition. Competition from numerous existing companies and others entering the fields in which we operate is intense and expected to increase. Most of these companies have substantially greater research and development, manufacturing, marketing, financial, technological personnel and managerial resources than we do. In addition, many specialized biotechnology companies have formed collaborations with large, established companies to support research, development and commercialization of products that may be competitive with our current and future product candidates and technologies. Acquisitions of competing companies by large pharmaceutical or biotechnology companies could further enhance such competitors’ financial, marketing and other resources. Academic institutions, governmental agencies and other public and private research organizations are also conducting research activities and seeking patent protection and may commercialize competitive products or technologies on their own or through collaborations with pharmaceutical and biotechnology companies.
| 28 |
We specifically face competition from companies with approved treatments of Gaucher disease, including Genzyme, which was recently acquired by Sanofi, and to a much lesser extent, Actelion. In addition, in February 2010, the FDA approved VPRIV, Shire’s enzyme replacement therapy for the treatment of Gaucher disease and the European Commission granted marketing authorization to VPRIV in August 2010. In addition, we are aware of other early clinical stage, experimental, small molecule, oral drugs which are being developed for the treatment of Gaucher disease by Genzyme and Amicus Therapeutics. Amicus Therapeutics reports that it has suspended its development of its Gaucher disease treatment. We also face competition from companies with approved enzyme treatments of Fabry disease, including Genzyme and Shire, and we are aware of other early stage drugs which are being developed for the treatment of Fabry disease, including a drug being developed by Amicus Therapeutics.
We also face competition from companies that are developing other platforms for the expression of recombinant therapeutic pharmaceuticals. We are aware of companies that are developing alternative technologies to develop and produce therapeutic proteins in anticipation of the expiration of certain patent claims covering marketed proteins. Competitors developing alternative expression technologies include Crucell N.V. (which was acquired by Johnson & Johnson during 2010), Shire and GlycoFi, Inc. (which was acquired by Merck & Co. Inc.). Other companies are developing alternate plant-based technologies, include Biolex, Inc., Chlorogen, Inc., iBIO, Medicago, Greenovation Biotech GmbH, and Symbiosys, none of which are cell-based. Rather, such companies base their product development on transgenic plants or whole plants.
Several biogeneric companies are pursuing the opportunity to develop and commercialize follow-on versions of other currently marketed biologic products, including growth factors, hormones, enzymes, cytokines and monoclonal antibodies, which are areas that interest us. These companies include, among others, Novartis AG/Sandoz Pharmaceuticals, BioGeneriX AG, Stada Arzneimittel AG, BioPartners GmbH and Teva.
Key differentiating elements affecting the success of our product candidates are likely to be their potency and efficacy profiles, as well as their cost-effectiveness as compared to other existing therapies. See “Risk Factors—Developments by competitors may render our products or technologies obsolete or non-competitive which would have a material adverse effect on our business and results of operations.”
Scientific Advisory Board
Members of our scientific advisory board, who are experts in the fields of plant molecular and cell biology as well as Gaucher disease and various hematological and genetic disorders, consult with our management within their professional areas of expertise; exchange strategic and business development ideas with our management; attend scientific, medical and business meetings with our management, such as meetings with the FDA and comparable foreign regulatory authorities, meetings with strategic or potential strategic partners and other meetings relevant to their areas of expertise; and attend meetings of our scientific advisory board. We expect our scientific advisory board to convene at least twice annually, and we frequently consult with the individual members of our Scientific Advisory Board. Our scientific advisory board currently includes the following people:
| Name | Affiliation | |
| Professor Aaron Ciechanover, M.D., D.Sc. |
Laureate of the Nobel Prize in Chemistry
Distinguished research Professor at the Cancer and Vascular Biology Research Center of the Rappaport Research Institute and Faculty of Medicine at the Technion
American Academy of Arts and Sciences, Member | |
| Professor Gad Galili, Ph.D. | Former Chairman of the Department of Plant Sciences, The Weizmann Institute of Science, Rehovot, Israel | |
| Professor Ari Zimran, M.D. |
Director of the Gaucher Clinic, Shaare Zedek Medical Center, Jerusalem, Israel
Associate Professor of Medicine, Hebrew University-Hadassah Medical School, Jerusalem, Israel |
| 29 |
Government Regulation
The testing, manufacture, distribution, advertising and marketing of drug products are subject to extensive regulation by federal, state and local governmental authorities in the United States, including the FDA, and by similar authorities in other countries. Any product that we develop must receive all relevant regulatory approvals or clearances, as the case may be, before it may be marketed in a particular country.
The regulatory process, which includes overseeing preclinical studies and clinical trials of each pharmaceutical compound to establish its safety and efficacy and confirmation by the FDA that good laboratory, clinical and manufacturing practices were maintained during testing and manufacturing, can take many years, requires the expenditure of substantial resources and gives larger companies with greater financial resources a competitive advantage over us. Delays or terminations of clinical trials that we undertake would likely impair our development of product candidates. Delays or terminations could result from a number of factors, including stringent enrollment criteria, slow rate of enrollment, size of patient population, having to compete with other clinical trials for eligible patients, geographical considerations and others.
The FDA review process can be lengthy and unpredictable, and we may encounter delays or rejections of our applications when submitted. Generally, in order to gain FDA approval, we must first conduct preclinical studies in a laboratory and in animal models to obtain preliminary information on a compound and to identify any potential safety problems. The results of these studies are submitted as part of an IND application that the FDA must review before human clinical trials of an investigational drug can commence. Clinical trials may be terminated by the clinical trial site, sponsor or the FDA if toxicities appear that are either worse than expected or unexpected.
Clinical trials are normally performed in three sequential phases and generally take two to five years, or longer, to complete. Phase I consists of testing the drug product in a small number of humans, normally healthy volunteers, to determine preliminary safety and tolerable dose range. Phase II usually involves studies in a limited patient population to evaluate the effectiveness of the drug product in humans having the disease or medical condition for which the product is indicated, determine dosage tolerance and optimal dosage and identify possible common adverse effects and safety risks. Phase III consists of additional controlled testing at multiple clinical sites to establish clinical safety and effectiveness in an expanded patient population of geographically dispersed test sites to evaluate the overall benefit-risk relationship for administering the product and to provide an adequate basis for product labeling. Phase IV clinical trials may be conducted after approval to gain additional experience from the treatment of patients in the intended therapeutic indication.
After completion of clinical trials of a new drug product, FDA and foreign regulatory authority marketing approval must be obtained. Assuming that the clinical data support the product’s safety and effectiveness for its intended use, an NDA is submitted to the FDA for its review. Generally, it takes one to three years to obtain approval. If questions arise during the FDA review process, approval may take a significantly longer period of time. The testing and approval processes require substantial time and effort and approval on a timely basis, if at all, or the approval that we receive may be for a narrower indication than we had originally sought, potentially undermining the commercial viability of the product. Even if regulatory approvals are obtained, approved products are subject to continual review and holders of an approved product are required, for example, to report certain adverse reactions and production problems, if any, to the FDA, and to comply with certain requirements concerning advertising and promotional labeling for the product. Also, quality control and manufacturing procedures relating to a product must continue to conform to cGMP after approval, and the FDA periodically inspects manufacturing facilities to assess compliance with cGMP. Accordingly, manufacturers must continue to expend time, money and effort in the area of production and quality control to comply with cGMP and other aspects of regulatory compliance. The later discovery of previously unknown problems or failure to comply with the applicable regulatory requirements with respect to any product may result in restrictions on the marketing of the product or withdrawal of the product from the market as well as possible civil or criminal sanctions. See also “—International Regulation.”
Under the Orphan Drug Act of 1983, the FDA may grant orphan drug designation to drugs and biological products intended to treat a rare disease or condition, which is generally a disease or condition that affects fewer than 200,000 individuals in the United States. In September 2009, we received orphan drug designation for taliglucerase alfa for the treatment of Gaucher disease. The FDA grants orphan drug designation to drugs that may provide a significant therapeutic advantage over existing treatments and target conditions affecting 200,000 or fewer U.S. patients per year. Orphan drug designation does not convey any advantage in or shorten the duration of the regulatory review and approval process. Among the other benefits of orphan drug designation are possible funding and tax savings to support clinical trials and for other financial incentives and a waiver of the marketing application user fee and most likely priority review. If a significant therapeutic advantage over existing treatments is shown in the marketing application, FDA may grant orphan drug approval and provide a seven-year period of marketing exclusivity.
| 30 |
The FDA has a fast track program that is intended to expedite or facilitate the process for reviewing new drugs and biological products that meet certain criteria. Specifically, new drugs and biological products are eligible for fast track designation if they are intended to treat a serious or life-threatening condition and demonstrate the potential to address unmet medical needs for the condition. Fast track designation applies to the combination of the product and the specific indication for which it is being studied. For a fast track product, the FDA may consider for review on a rolling basis sections of the NDA before the complete application is submitted, if the sponsor provides a schedule for the submission of the sections of the NDA, the FDA agrees to accept sections of the NDA as they become available and determines that the schedule is acceptable, and the sponsor pays any required user fees upon submission of the first section of the NDA. We used the rolling submission option for our NDA for our lead product candidate, taliglucerase alfa, which we completed in April 2010.
None of our products under development has been approved for marketing in the United States or elsewhere. We may not be able to obtain regulatory approval for any of our products under development in a timely manner, if at all. Failure to obtain requisite governmental approvals or failure to obtain approvals of the scope requested will delay or preclude us, or our licensees or marketing partners, from marketing our products, or limit the commercial use of our products, and thereby would have a material adverse effect on our business, financial condition and results of operations. See “Risk Factors—We may not obtain the necessary U.S. or worldwide regulatory approvals to commercialize our drug candidates in a timely manner, if at all, which would have a material adverse effect on our business and results of operations.”
The United States federal government regulates healthcare through various agencies, including but not limited to the following: (i) the FDA, which administers the Federal Food, Drug, and Cosmetic Act (FDCA), as well as other relevant laws; (ii) the Center for Medicare & Medicaid Services (CMS), which administers the Medicare and Medicaid programs; (iii) the Office of Inspector General (OIG) which enforces various laws aimed at curtailing fraudulent or abusive practices, including by way of example, the Anti-Kickback Law, the Anti-Physician Referral Law, commonly referred to as Stark, the Anti-Inducement Law, the Civil Money Penalty Law and the laws that authorize the OIG to exclude healthcare providers and others from participating in federal healthcare programs; and (iv) the Office of Civil Rights, which administers the privacy aspects of the Health Insurance Portability and Accountability Act of 1996 (HIPAA). All of the aforementioned are agencies within the Department of Health and Human Services (HHS). Healthcare is also provided or regulated, as the case may be, by the Department of Defense through its TriCare program, the Department of Veterans Affairs, especially through the Veterans Health Care Act of 1992, the Public Health Service within HHS under Public Health Service Act § 340B (42 U.S.C. § 256b), the Department of Justice through the Federal False Claims Act and various criminal statutes, and state governments under the Medicaid and other state sponsored or funded programs and their internal laws regulating all healthcare activities. Many states also have anti-kickback and anti-physician referral laws that are similar to the federal laws, but may be applicable in situations where federal laws do not apply.
Medicare is the federal healthcare program for those who are (i) over 65 years of age, (ii) disabled, (iii) suffering from end-stage renal disease or (iv) suffering from Lou Gehrig’s disease. Medicare consists of part A, which covers inpatient costs, part B, which covers services by physicians and laboratories, durable medical equipment and certain drugs, primarily those administered by physicians, and part D, which provides drug coverage for most prescription drugs other than those covered under part B. Medicare also offers a managed care option under part C. Medicare is administered by CMS. In contrast, Medicaid is a state-federal healthcare program for the poor and is administered by the states pursuant to an agreement with the Secretary of Health and Human Services. Most state Medicaid programs cover most outpatient prescription drugs.
In March 2010, the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act, or collectively, PPACA, became law in the United States. PPACA substantially changes the way healthcare is financed by both governmental and private insurers and significantly affects the pharmaceutical industry. Key provisions of PPACA specific to the pharmaceutical industry, among others, include the following:
| · | An annual, nondeductible fee on any entity that manufactures or imports certain branded prescription drugs and biologic agents into the United States, apportioned among these entities according to their market share in certain federal government healthcare programs (excluding sales of any drug or biologic product marketed for an orphan indication), beginning in 2011; | |
| · | An increase in the rebates a manufacturer must pay under the Medicaid Drug Rebate Program, retroactive to January 1, 2010, to 23.1% and 13% of the average manufacturer price for branded and generic drugs, respectively; | |
| · | A new Medicare Part D coverage gap discount program, in which manufacturers must agree to offer 50% point-of sale discounts off negotiated prices of applicable brand drugs to eligible beneficiaries during their coverage gap period, as a condition for the manufacturer’s outpatient drugs to be covered under Medicare Part D, beginning in 2011; |
| 31 |
| · | Extension of manufacturers’ Medicaid rebate liability to covered drugs dispensed to individuals who are enrolled in Medicaid managed care organizations, effective March 23, 2010; | |
| · | Expansion of eligibility criteria for Medicaid programs by, among other things, allowing states to offer Medicaid coverage to additional individuals beginning in April 2010 and by adding new mandatory eligibility categories for certain individuals with income at or below 133% of the Federal Poverty Level beginning in 2014, thereby potentially increasing both the volume of sales and manufacturers’ Medicaid rebate liability; | |
| · | Expansion of the entities eligible for discounts under the Public Health Service pharmaceutical pricing program, effective January 2010; | |
| · | New requirements to report certain financial arrangements with physicians and others, including reporting any “transfer of value” made or distributed to prescribers and other healthcare providers and reporting any investment interests held by physicians and their immediate family members during each calendar year beginning in 2012, with reporting starting in 2013; | |
| · | A new requirement to annually report drug samples that manufacturers and distributors provide to physicians, effective April 1, 2012; | |
| · | A licensure framework for follow-on biologic products; and | |
| · | A new Patient-Centered Outcomes Research Institute to oversee, identify priorities in, and conduct comparative clinical effectiveness research, along with funding for such research. |
International Regulation
We are subject to regulations and product registration requirements in many foreign countries in which we may sell our products, including in the areas of product standards, packaging requirements, labeling requirements, import and export restrictions and tariff regulations, duties and tax requirements. The time required to obtain clearance required by foreign countries may be longer or shorter than that required for FDA clearance, and requirements for licensing a product in a foreign country may differ significantly from FDA requirements.
Pharmaceutical products may not be imported into, or manufactured or marketed in, the State of Israel absent drug registration. The three basic criteria for the registration of pharmaceuticals in Israel is quality, safety and efficacy of the pharmaceutical product and the Israeli MOH requires pharmaceutical companies to conform to international developments and standards. Regulatory requirements are constantly changing in accordance with scientific advances as well as social and ethical values.
The relevant legislation of the European Union requires that medicinal products, including generic versions of previously approved products, and new strengths, dosage forms and formulations, of previously approved products, shall have a marketing authorization before they are placed on the market in the European Union. Authorizations are granted after the assessment of quality, safety and efficacy by the respective health authorities. In order to obtain an authorization, an application must be made to the competent authority of the member state concerned or in a centralized procedure to the EMA. Besides various formal requirements, the application must contain the results of pharmaceutical (physico-chemical, biological or microbiological) tests, of preclinical (toxicological and pharmacological) tests as well as of clinical trials. All of these tests must have been conducted in accordance with relevant European Union regulations and must allow the reviewer to evaluate the quality, safety and efficacy of the medicinal product. On January 2010, the Committee for Orphan Medicinal Products (COMP) of the EMA recommended that the European Commission grant orphan drug designation to taliglucerase alfa. Orphan drug designation in the European Union is granted to medicinal products intended for the diagnosis, prevention and treatment of life-threatening diseases and very serious conditions that affect not more than five in 10,000 people in the European Union. Orphan drug designation is generally given to medicinal products that treat conditions for which no current therapy exists or are expected to bring a significant benefit to patients over existing therapies. If granted by the European Commission, orphan drug designation will provide us a centralized procedure for obtaining marketing authorization for taliglucerase alfa, with a single marketing authorization valid throughout all EU Member States. We may also be eligible for a number of additional incentives including protocol assistance, reduction in registration fees and eligibility for grants and initiatives supporting research and development related to the orphan drug designation.
| 32 |
Israeli Government Programs
The following is a summary of the current principal Israeli tax laws applicable to us and Protalix Ltd., and of the Israeli Government programs from which Protalix Ltd. benefits. Some parts of this discussion are based on new tax legislation that has not been subject to judicial or administrative interpretation. Therefore, the views expressed in the discussion may not be accepted by the tax authorities in question. The discussion should not be construed as legal or professional tax advice and does not cover all possible tax considerations.
General Corporate Tax Structure in Israel
Protalix Ltd.'s income, other than income from Approved Enterprises, is taxed in Israel at the regular rate. According to the provisions of the Law for Amending the Israel Income Tax Ordinance, 2005, corporate tax rates will be gradually lowered, resulting in a corporate tax rate for 2009 and thereafter: 2009 – 26% and for 2010 and thereafter – 25%.
On July 14, 2009, the Israel Economic Efficiency Law (Legislation Amendments for Applying the Economic Plan for 2009 and 2010), 2009, became effective, stipulating, among other things, an additional gradual decrease in tax rates in 2011 and thereafter, as follows: 2011 – 24%, 2012 – 23%, 2013 – 22%, 2014 – 21%, 2015 – 20% and 2016 and thereafter – 18%.
On December 5, 2011, the Tax Burden Distribution Law was officially published, discontinuing a previously approved gradual decrease in corporate tax provided in the 2009 amendment, and setting the corporate tax rate in Israel for 2012 and thereafter to 25%.
Capital gains on the sale of assets are subject to capital gains tax according to the corporate tax rate in effect for the year the assets are sold.
In addition to the corporate taxes in Israel, we are subject to a withholding tax on the U.S. revenue source portion of the payments made to us for our share of Pfizer’s net profits under the Pfizer Agreement. The withholding tax rate is 15%. See “Business—Commercialization Agreement.”
Law for the Encouragement of Capital Investments, 1959
The Law for the Encouragement of Capital Investments, 1959, or the Investment Law, provides certain incentives for capital investments in a production facility (or other eligible assets). Generally, an investment program that is implemented in accordance with the provisions of the Investment Law, referred to as an “Approved Enterprise,” is entitled to benefits. These benefits may include cash grants from the Israeli government and tax benefits, based upon, among other things, the location of the facility in which the investment is made and specific elections made by the grantee.
The Investment Law was significantly amended effective in April 2005. Protalix Ltd. will continue to enjoy the tax benefits under the pre-revision provisions of the Investment Law. If any new benefits are granted to Protalix Ltd. in the future, Protalix Ltd. will be subject to the provisions of the amended Investment Law with respect to these new benefits. Therefore, the following discussion is a summary of the Investment Law prior to its amendment as well as the relevant changes contained in the new legislation.
Under the Investment Law prior to its amendment, a company that wished to receive benefits had to receive approval from the “Investment Center” of the Israeli Ministry of Industry, Trade and Labor, or the Investment Center. Each certificate of approval for an Approved Enterprise relates to a specific investment program in the Approved Enterprise, delineated both by the financial scope of the investment and by the physical characteristics of the facility or the asset, e.g., the equipment to be purchased and utilized pursuant to the program.
An Approved Enterprise may elect to forego any entitlement to the grants otherwise available under the Investment Law and, instead, participate in an alternative benefits program under which the undistributed income from the Approved Enterprise is fully exempt from corporate tax for a defined period of time. Under the alternative package of benefits, a company’s undistributed income derived from an Approved Enterprise will be exempt from corporate tax for a period of between two and 10 years from the first year of taxable income, depending upon the geographic location within Israel of the Approved Enterprise. Upon expiration of the exemption period, the Approved Enterprise is eligible for the reduced tax rates otherwise applicable under the Investment Law for any remainder of the otherwise applicable benefits period (up to an aggregate benefits period of either seven or 10 years, depending on the location of the company or its definition as a foreign investors’ company). If a company has more than one Approved Enterprise program or if only a portion of its capital investments are approved, its effective tax rate is the result of a weighted combination of the applicable rates. The tax benefits from any certificate of approval relate only to taxable profits attributable to the specific Approved Enterprise. Income from activity that is derived from different Approved Enterprises does not enjoy these tax benefits.
A company that has an Approved Enterprise program is eligible for further tax benefits if it qualifies as a foreign investors’ company. A foreign investors’ company eligible for benefits is essentially a company in which more than 25% of the share capital (in terms of shares, rights to profit, voting and appointment of directors) is owned (measured by both share capital and combined share and loan capital) by non-Israeli residents. A company that qualifies as a foreign investors’ company and has an Approved Enterprise program is eligible for tax benefits for a 10-year benefit period and may enjoy a reduced corporate tax rate of 10% to 25%, depending on the amount of the company’s shares held by non-Israeli shareholders.
| 33 |
If a company that has an Approved Enterprise program is a wholly owned subsidiary of another company, then the percentage of foreign investments is determined based on the percentage of foreign investment in the parent company. The tax rates and related levels of foreign investments are set forth in the following table:
| Percent of Foreign Ownership | Rate of Reduced Tax | |||
| 0-49% | 25% | |||
| 49-74% | 20% | |||
| 74-90% | 15% | |||
| 90-100% | 10% | |||
Our original facility in Israel has been granted “Approved Enterprise” status, and it has elected to participate in the alternative benefits program. Under the terms of its Approved Enterprise program, the facility is located in a top priority location, or “Zone A,” and, therefore, the income from that Approved Enterprise will be tax exempt in Israel for a period of 10 years, commencing with the year in which taxable income is first generated from the relevant Approved Enterprise. The current benefits program may not continue to be available and Protalix Ltd. may not continue to qualify for its benefits.
A company that has elected to participate in the alternative benefits program and that subsequently pays a dividend out of the income derived from the Approved Enterprise during the tax exemption period will be subject to corporate tax in respect of the amount distributed at the rate that would have been applicable had the company not elected the alternative benefits program (generally 10% to 25%, depending on the extent to which non-Israeli shareholders hold such company’s shares). If the dividend is distributed within 12 years after the commencement of the benefits period (or, in the case of a foreign investor’s company, without time limitation), the dividend recipient is taxed at the reduced withholding tax rate of 15% applicable to dividends from approved enterprises, or at the lower rate under an applicable tax treaty. After this period, the withholding tax rate is 25%, or at the lower rate under an applicable tax treaty. In the case of a company with a foreign investment level (as defined by the Investment Law) of 25% or more, the 12-year limitation on reduced withholding tax on dividends does not apply. The company must withhold this tax at its source, regardless of whether the dividend is converted into foreign currency.
The Investment Law also provides that an Approved Enterprise is entitled to accelerated depreciation on its property and equipment that are included in an approved investment program. This benefit is an incentive granted by the Israeli government regardless of whether the alternative benefits program is elected.
The benefits available to an Approved Enterprise are conditioned upon terms stipulated in the Investment Law and regulations and the criteria set forth in the applicable certificate of approval. If Protalix Ltd. does not fulfill these conditions in whole or in part, the benefits can be canceled and Protalix Ltd. may be required to refund the received benefits, linked to the Israeli consumer price index with the addition of interest or alternatively with an additional penalty payment. We believe that Protalix Ltd. currently operates in compliance with all applicable conditions and criteria, but there can be no assurance that Protalix Ltd. will continue to do so. Furthermore, there can be no assurance that any Approved Enterprise status granted to Protalix Ltd.’s facilities will entitle Protalix Ltd. to the same benefits to which it is currently entitled.
Pursuant to the March 2005 amendment to the Investment Law, the approval of the Investment Center is required only for Approved Enterprises that receive cash grants. Approved Enterprises that do not receive benefits in the form of governmental cash grants, but only tax benefits, are no longer required to obtain this approval. Instead, these Approved Enterprises are required to make certain investments as specified in the Investment Law.
The amended Investment Law specifies certain conditions for an Approved Enterprise to be entitled to benefits. These conditions include:
| · | the Approved Enterprise’s revenues from any single country or a separate customs territory may not exceed 75% of the Approved Enterprise’s total revenues; or | |
| · | at least 25% of the Approved Enterprise’s revenues during the benefits period must be derived from sales into a single country or a separate customs territory with a population of at least 12 million. |
There can be no assurance that Protalix Ltd. will comply with the above conditions in the future or that Protalix Ltd. will be entitled to any additional benefits under the Investment Law. In addition, it is possible that Protalix Ltd. may not be able to operate in a manner that maximizes utilization of the potential benefits available under the Investment Law.
| 34 |
From time to time, the Israeli Government has considered reducing the benefits available to companies under the Investment Law. The termination or substantial reduction of any of the benefits available under the Investment Law could materially impact the cost of our future investments.
Encouragement of Industrial Research and Development Law, 1984
To date, Protalix Ltd. has received grants from the Office of the Chief Scientist of the Israeli Ministry of Industry, Trade and Labor, the OCS, for the financing of a portion of its research and development expenditures in Israel. As of December 31, 2011, the OCS approved grants in respect of Protalix Ltd.’s continuing operations totaling approximately $23.9 million, measured from inception. Protalix Ltd. is required to repay up to 100% of grants actually received (plus interest at the LIBOR rate applied to the grants received on or after January 1, 1999) to the OCS through payments of royalties at a rate of 3% to 6% of the revenues generated from an OCS-funded project, depending on the period in which revenues were generated. As of December 31, 2011, Protalix Ltd. either paid or accrued royalties payable of $2.8 million and Protalix Ltd.’s contingent liability to the OCS with respect to grants received was approximately $19.8 million.
Under the Israeli Law for the Encouragement of Industrial Research and Development, 1984, and related regulations, or the Research Law, recipients of grants from the OCS are prohibited from manufacturing products developed using these grants outside of the State of Israel without special approvals, although the Research Law does enable companies to seek prior approval for conducting manufacturing activities outside of Israel without being subject to increased royalties. If Protalix Ltd. receives approval to manufacture the products developed with government grants outside of Israel, it will be required to pay an increased total amount of royalties (possibly up to 300% of the grant amounts plus interest), depending on the manufacturing volume that is performed outside of Israel, as well as at a possibly increased royalty rate.
Additionally, under the Research Law, Protalix Ltd. is prohibited from transferring the OCS-financed technologies and related intellectual property rights outside of the State of Israel, except under limited circumstances and only with the approval of the OCS' Research Committee. Protalix Ltd. may not receive the required approvals for any proposed transfer and, if received, Protalix Ltd. may be required to pay the OCS a portion of the consideration that it receives upon any sale of such technology by a non-Israeli entity. The scope of the support received, the royalties that Protalix Ltd. has already paid to the OCS, the amount of time that has elapsed between the date on which the know-how was transferred and the date on which the OCS grants were received and the sale price and the form of transaction will be taken into account in order to calculate the amount of the payment to the OCS. Approval of the transfer of technology to residents of the State of Israel is required, and may be granted in specific circumstances only if the recipient abides by the provisions of applicable laws, including the restrictions on the transfer of know-how and the obligation to pay royalties. No assurance can be made that approval to any such transfer, if requested, will be granted.
In March 2005, Research Law was amended. One of the main modifications included in the amendment was an authorization of the Research Committee to allow the transfer outside of Israel of know-how derived from an approved program and the related manufacturing rights. In general, the Research Committee may approve transfer of know-how in limited circumstances as follows:
| · | in the event of a sale of know-how itself to a non affiliated third party, provided that upon such sale the owner of the know-how pays to the OCS an amount, in cash, as set forth in the Research Law. In addition, the amendment provides that if the purchaser of the know-how gives the selling Israeli company the right to exploit the know-how by way of an exclusive, irrevocable and unlimited license, the research committee may approve such transfer in special cases without requiring a cash payment. | |
| · | in the event of a sale of a company which is the owner of know-how, pursuant to which the company ceases to be an Israeli company, provided that upon such sale, the owner of the know-how makes a cash payment to the OCS as set forth in the Research Law. | |
| · | in the event of an exchange of know-how such that in exchange for the transfer of know-how outside of Israel, the recipient of the know-how transfers other know-how to the company in Israel in a manner in which the OCS is convinced that the Israeli economy realizes a greater, overall benefit from the exchange of know-how. |
Another provision in the amendment concerns the transfer of manufacturing rights. The research committee may, in special cases, approve the transfer of manufacture or of manufacturing rights of a product developed within the framework of the approved program or which results therefrom, outside of Israel.
| 35 |
The State of Israel does not own intellectual property rights in technology developed with OCS funding and there is no restriction on the export of products manufactured using technology developed with OCS funding. The technology is, however, subject to transfer of technology and manufacturing rights restrictions as described above. For a description of such restrictions, please see “Risk Factors—Risks Relating to Our Operations in Israel.” OCS approval is not required for the export of any products resulting from the research or development or for the licensing of any technology in the ordinary course of business.
Special Provisions Relating to Taxation under Inflationary Conditions
Protalix Ltd. is subject to Israel's Income Tax Law (Inflationary Adjustments), 1985, or the Inflationary Adjustments Law. The Inflationary Adjustments Law is highly complex, and represents an attempt to overcome the problems presented to a traditional tax system by an economy undergoing rapid inflation. The provisions that are material to us are summarized below:
| · | Where a company’s equity, as calculated under the Inflationary Adjustments Law, exceeds the depreciated cost of its fixed assets (as defined in the Inflationary Adjustments Law), a deduction from taxable income is permitted equal to this excess multiplied by the applicable annual rate of inflation. The maximum deduction permitted under this provision in any single tax year is 70% of taxable income. The unused portion linked to the Israeli consumer price index, may be carried forward. | |
| · | Where a company’s depreciated cost of fixed assets exceeds its equity, the excess multiplied by the applicable annual rate of inflation is added to taxable income. | |
| · | Subject to specified limitations, depreciation deductions carryforwards on fixed assets and losses are adjusted for inflation based on the change in the consumer price index. |
Under the Inflationary Adjustments Law, results for tax purposes are measured in real terms, in accordance with changes in the Israeli consumer price index. The difference between the change in the Israeli consumer price index and the exchange rate of Israeli currency in relation to the U.S. dollar may in future periods cause significant differences between taxable income and the income measured in dollars as reflected in our consolidated financial statements.
Law for the Encouragement of Industry (Taxes), 1969
We believe that Protalix Ltd. currently qualifies as an “Industrial Company” within the meaning of the Law for the Encouragement of Industry (Taxes), 1969, or the Industry Encouragement Law. The Industry Encouragement Law defines “Industrial Company” as a company resident in Israel that derives 90% or more of its income in any tax year (other than specified kinds of passive income such as capital gains, interest and dividends) from an “Industrial Enterprise” that it owns. An “Industrial Enterprise” is defined as an enterprise whose major activity in a given tax year is industrial production.
The following corporate tax benefits, among others, are available to Industrial Companies:
| · | amortization of the cost of purchased know-how and patents over an eight-year period for tax purposes; | |
| · | accelerated depreciation rates on equipment and buildings; | |
| · | under specified conditions, an election to file consolidated tax returns with other related Israeli Industrial Companies; and | |
| · | expenses related to a public offering are deductible in equal amounts over three years. |
Eligibility for the benefits under the Industry Encouragement Law is not subject to receipt of prior approval from any governmental authority. It is possible that Protalix Ltd. may fail to qualify or may not continue to qualify as an “Industrial Company” or that the benefits described above will not be available in the future.
Tax Benefits for Research and Development
Under specified conditions, Israeli tax laws allow a tax deduction by a company for research and development expenditures, including capital expenditures, for the year in which such expenditures are incurred. These expenditures must relate to scientific research and development projects and must be approved by the OCS. Furthermore, the research and development projects must be for the promotion of the company and carried out by or on behalf of the company seeking such tax deduction. However, the amount of such deductible expenditures is reduced by the sum of any funds received through government grants for the finance of such scientific research and development projects. Expenditures not so approved are deductible over a three-year period.
| 36 |
Employees
As of December 31, 2011, we had 235 employees, of whom 34 have an M.D. or a Ph.D. in their respective scientific fields. We believe that our relations with these employees are good. We intend to continue to hire additional employees in research and development, manufacturing and administration in order to meet our operating plans. We believe that our success will greatly depend on our ability to identify, attract and retain capable employees. The Israeli Ministry of Labor and Welfare is authorized to make certain industry-wide collective bargaining agreements that apply to types of industries or employees including ours (“Expansion Orders”). These agreements affect matters such as cost of living adjustments to salaries, length of working hours and week, recuperation, travel expenses, and pension rights. Otherwise, our employees are not represented by a labor union or represented under a collective bargaining agreement. See “Risk Factors—We depend upon key employees and consultants in a competitive market for skilled personnel. If we are unable to attract and retain key personnel, it could adversely affect our ability to develop and market our products.”
Company Background
Our principal business address is set forth below. Our executive offices and our main research manufacturing facility are located at that address. Our telephone number is +972-4-988-9488. From May 2001 through December 31, 2006, our company had no operations. We were originally formed as Embassy Acquisition Corp., a Florida corporation, in November 2005 and changed our name to Orthodontix, Inc., in April, 1992. On December 31, 2006, we acquired, through a merger with our wholly-owned subsidiary, Protalix Acquisition Co. Ltd., all of the outstanding shares of Protalix Ltd., in exchange for shares of our common stock. As a result, Protalix Ltd. is now our wholly-owned subsidiary. In connection with the merger, we completed a one-for-ten reverse stock split and on February 26, 2007, we changed our name to Protalix BioTherapeutics, Inc. Unless otherwise indicated, all share numbers in this Annual Report on Form 10-K give effect to such reverse stock split.
Our wholly-owned subsidiary and sole operating unit, Protalix Ltd., is an Israeli corporation and was originally incorporated in Israel as Metabogal Ltd. on December 27, 1993. During 1999, Protalix Ltd. changed its focus from plant secondary metabolites to the expression of recombinant therapeutic proteins in plant cells, and in April 2004 changed its name to Protalix Ltd.
ProCellEx® is our registered trademark. Each of the other trademarks, trade names or service marks appearing in this Annual Report on Form 10-K belongs to its respective holder.
Available Information
Our corporate website is www.protalix.com. We make available on our website, free of charge, our Commission, filings, including our Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and any amendments to these reports, as soon as reasonably practicable after we electronically file these documents with, or furnish them to, the Commission. Additionally, from time to time, we provide notifications of material news including press releases and conferences on our website. Webcasts of presentations made by our company at certain conferences may also be available from time to time on our website, to the extent the webcasts are available. The content of our website is not intended to be incorporated by reference into this report or in any other report or document we file and any references to these websites are intended to be inactive textual references only.
We are also listed on the Tel Aviv Stock Exchange, or the TASE, and, accordingly, we submit copies of all our filings with the Commission to the Israeli Securities Authority and the TASE. Such copies can be retrieved electronically through the TASE’s internet messaging system (www.maya.tase.co.il) and through the MAGNA distribution site of the Israeli Securities Authority (www.magna.isa.gov.il).
Our website also includes printable versions of our Code of Business Conduct and Ethics and the charters for each of the Audit, Compensation and Nominating Committees of our Board of Directors. Each of these documents is also available in print, free of charge, to any shareholder who requests a copy by addressing a request to:
Protalix BioTherapeutics, Inc.
2 Snunit Street
Science Park
POB 455
Carmiel 20100, Israel
Attn: Mr. Yossi Maimon, Chief Financial Officer
| 37 |
Item 1A. Risk Factors
You should carefully consider the risks described below together with the other information included in this Annual Report on Form 10-K. Our business, financial condition or results of operations could be adversely affected by any of these risks. If any of these risks occur, the value of our common stock could decline.
Risks Related to Our Financial Condition and Capital Requirements
We currently have no significant product revenues and will need to raise additional capital to operate our business, which may not be available on favorable terms, or at all, and which will have a dilutive effect on our shareholders.
To date, we have generated no significant revenues from product sales and only minimal revenues from research and development services and other fees, other than the milestone payments we received in connection with our license and supply agreement with Pfizer. For the years ended December 31, 2011, 2010 and 2009, we had net losses of $36.5 million, $29.0 million and $31.4 million, respectively, primarily as a result of expenses incurred through a combination of research and development activities and expenses supporting those activities, which includes share-based compensation expense. Drug development and commercialization is very capital intensive. Until we receive approval from the FDA and other regulatory authorities for our drug candidates, we cannot generate significant sales from our drugs and will not have product revenues, except for certain regulatory-related milestone payments under the Pfizer Agreement which we expect to earn prior to any sales of taliglucerase alfa. Therefore, until we generate significant sales, we will have to fund all of our operations and capital expenditures from our cash on hand, potential regulatory-related milestone payments under the Pfizer Agreement, other licensing fees and grants and the net proceeds of any equity or debt offerings. Over the next 12 months, we expect to spend a minimum of approximately $30.0 million building an internal sales and marketing force for the sale of taliglucerase alfa in Israel and on preclinical and clinical development for our products candidates. Based on our current plans and capital resources, we believe that our cash and cash equivalents together with the net proceeds from our public underwritten offering of February 2012 will be sufficient to enable us to meet our planned operating needs for at least 12 months. However, changes may occur that could consume our existing capital at a faster rate than projected, including, among others, the cost and timing of regulatory approvals, changes in the progress of our research and development efforts and the costs of protecting our intellectual property rights.
We may seek additional financing to implement and fund product development, preclinical studies and clinical trials for the drugs in our pipeline, as well as additional drug candidates and other research and development projects. If we are unable to secure additional financing in the future on acceptable terms, or at all, we may be unable to commence or complete planned preclinical and clinical trials or obtain approval of our drug candidates from the FDA and other regulatory authorities. In addition, we may be forced to reduce or discontinue product development or product licensing, reduce or forego sales and marketing efforts and other commercialization activities or forego attractive business opportunities in order to improve our liquidity and to enable us to continue operations which would have a material adverse effect on our business and results of operations. Any additional source of financing will likely involve the issuance of our equity securities, which will have a dilutive effect on our shareholders.
We are not currently profitable and may never become profitable which would have a material adverse effect on our business and results of operations and could negatively impact the value of our common stock.
We expect to incur substantial losses for the foreseeable future and may never become profitable. We also expect to continue to incur significant operating and capital expenditures, and we anticipate that our expenses will increase substantially in the foreseeable future as we:
| · | seek regulatory approvals for our drug candidates; |
| · | continue to undertake preclinical development and clinical trials for our current and new drug candidates; |
| · | hire additional personnel; |
| · | implement additional internal systems and infrastructure; and |
| · | seek to license-in additional technologies to develop. |
We also expect to continue to experience negative cash flow for the foreseeable future as we fund our operating losses and capital expenditures. As a result, we will need to generate significant revenues in order to achieve and maintain profitability. We may not be able to generate these revenues or achieve profitability in the future. Any failure to achieve or maintain profitability may have a material adverse effect on our business and results of operations and may negatively impact the value of our common stock.
| 38 |
Risks Related to Our Business
We have a limited operating history which may limit the ability of investors to make an informed investment decision.
We are a clinical stage biopharmaceutical company. To date, we have not commercialized any of our drug candidates or received any FDA or other approval to market any drug. The successful commercialization of our drug candidates will require us to perform a variety of functions, including:
| · | continuing to undertake preclinical development and clinical trials; |
| · | participating in regulatory approval processes; |
| · | formulating and manufacturing products; and |
| · | conducting sales and marketing activities. |
Our operations have been limited to organizing and staffing our company, acquiring, developing and securing our proprietary technology and undertaking, through third parties, preclinical trials and clinical trials of our principal drug candidates. To date, we have commenced a phase III clinical trial in connection with only one drug candidate, taliglucerase alfa, which trial was completed in August 2009, and we have not commenced any additional clinical trial for any of our other drug candidates, except for our acetylcholinesterase product candidate. These operations provide a limited basis for investors to assess our ability to commercialize our drug candidates and whether to invest in our company.
Our ProCellEx protein expression system is based on our proprietary plant cell-based expression technology which has a limited history and any material problems with the system, which may be unforeseen, may have a material adverse effect on our business and results of operations.
Our ProCellEx protein expression system is based on our proprietary plant cell-based expression technology. Our business is dependent upon the successful development and approval of our product candidates produced through our protein expression system. Our ProCellEx protein expression system is novel and is still in the early stages of development and optimization, and, accordingly, is subject to certain risks. Mammalian cell-based protein expression systems have been used in connection with recombinant therapeutic protein expression for more than 20 years and are the subject of a wealth of data; in contrast, there is not a significant amount of data generated regarding plant cell-based protein expression and, accordingly, plant cell-based protein expression systems may be subject to unknown risks. In addition, the protein glycosilation pattern created by our protein expression system is not identical to the natural human glycosilation pattern and its long term effect on human patients is still unknown. Lastly, as our protein expression system is a new technology, we cannot always rely on existing equipment; rather, there is a need to design custom-made equipment and to generate specific growth media for the plant cells, which may not be available at favorable prices, if at all. Any material problems with the technology underlying our plant cell-based protein expression system may have a material adverse effect on our business, results of operations and financial condition.
We currently depend heavily on the success of taliglucerase alfa, our lead product candidate. Any failure to commercialize taliglucerase alfa, or the experience of significant delays in doing so, will have a material adverse effect on our business, results of operations and financial condition.
We have invested a significant portion of our efforts and financial resources in the development of taliglucerase alfa. Our ability to generate product revenue, depends heavily on the successful development and commercialization of taliglucerase alfa. In November 2009, we granted to Pfizer an exclusive worldwide license to develop and commercialize taliglucerase alfa except in Israel. We retained such rights in Israel. The successful commercialization of taliglucerase alfa will depend on several factors, including the following:
| · | obtaining marketing approvals from the FDA and other foreign regulatory authorities; |
| · | successful completion of our ongoing studies of taliglucerase alfa; |
| · | the successful audit of our facilities by additional regulatory authorities; |
| · | maintaining the cGMP compliance of our manufacturing facility or establishing manufacturing arrangements with third parties; |
| · | Pfizer’s efforts under the Pfizer Agreement; |
| · | our development of a successful sales and marketing organization for taliglucerase in Israel; |
| · | a continued acceptable safety and efficacy profile of our product candidates following approval; |
| · | the availability of reimbursement to patients from healthcare payors for our drug products, if approved; and |
| · | other risks described in these Risk Factors. |
| 39 |
Any failure to commercialize taliglucerase alfa or the experience of significant delays in doing so will have a material adverse effect on our business, results of operations and financial condition.
Our strategy, in certain cases, is to enter into collaboration agreements with third parties to leverage our ProCellEx system to develop product candidates. If we fail to enter into these agreements or if we or the third parties do not perform under such agreements or terminate or elect to discontinue the collaboration, it could have a material adverse affect on our revenues.
Our strategy, in certain cases, is to enter into arrangements with pharmaceutical companies to leverage our ProCellEx system to develop additional product candidates. Under these arrangements, we may grant to our partners rights to license and commercialize pharmaceutical products developed under the applicable agreements. Our partners may control key decisions relating to the development of the products and we may depend on our partners’ expertise and dedication of sufficient resources to develop and commercialize our product candidates. The rights of our partners limit our flexibility in considering alternatives for the commercialization of our product candidates. To date, we have entered into a license and supply agreement with Pfizer relating to the development and commercialization of taliglucerase alfa and an agreement with Teva, which relates to the development by us of two proteins, and the licensing by Teva of such proteins in consideration for royalties and milestone payments. Subsequently, two proteins were identified to be researched and developed under the agreement but in 2009, both of the projects were terminated for commercial reasons. We may not identify any additional proteins to be developed through a collaboration between us and Teva under the agreement, which may have a material adverse effect on our business, results of operations and financial condition. If we or any of our partners breach or terminate the agreements that make up such arrangements, our partners otherwise fail to conduct their obligations under such arrangements in a timely manner, there is a dispute about their obligations or if either party terminates the applicable agreement or elects not to continue the arrangement, we may not enjoy the benefits of the agreements or receive a sufficient amount of royalty or milestone payments from them, if any, which may have a material adverse effect on our business, results of operations and financial condition.
All of our product candidates other than taliglucerase alfa and our acetylcholinesterase product are in preclinical or research stages. If we are unable to develop and commercialize our product candidates, our business will be adversely affected.
A key element of our strategy is to develop and commercialize a portfolio of new products in addition to taliglucerase alfa. We are seeking to do so through our internal research programs and strategic collaborations for the development of new products. Research programs to identify new product candidates require substantial technical, financial and human resources, whether or not any product candidates are ultimately identified. Our research programs may initially show promise in identifying potential product candidates, yet fail to yield product candidates for clinical development for many reasons, including the following:
| · | a product candidate is not capable of being produced in commercial quantities at an acceptable cost, or at all; |
| · | a product candidate may not be accepted by patients, the medical community or third-party payors; |
| · | competitors may develop alternatives that render our product candidates obsolete; |
| · | the research methodology used may not be successful in identifying potential product candidates; or |
| · | a product candidate may on further study be shown to have harmful side effects or other characteristics that indicate it is unlikely to be effective or otherwise does not meet applicable regulatory approval. |
Any failure to develop or commercialize any of our other product candidates may have a material adverse effect on our business, results of operations and financial condition.
Clinical trials are very expensive, time-consuming and difficult to design and implement and may result in unforeseen costs which may have a material adverse effect on our business and results of operations.
Human clinical trials are very expensive and difficult to design and implement, in part because they are subject to rigorous regulatory requirements. The clinical trial process is also time-consuming. Other than taliglucerase alfa and our acetylcholinesterase product, our drug candidates are in the preclinical studies or research stages. Other, ongoing clinical trials of taliglucerase alfa and our acetylcholinesterase product, and anticipated clinical trials of our other potential drug candidates which have not yet been initiated, will take at least several years to complete. Preliminary and initial results from a clinical trial do not necessarily predict final results, and failure can occur at any stage of the trials. We may encounter problems that cause us to abandon or repeat preclinical studies or clinical trials. Companies in the pharmaceutical and biotechnology industries have suffered significant setbacks in advanced clinical trials, even after obtaining promising results in earlier trials. Data obtained from tests are susceptible to varying interpretations which may delay, limit or prevent regulatory approval. Failure or delay in the commencement or completion of our clinical trials may be caused by several factors, including:
| 40 |
| · | unforeseen safety issues; |
| · | determination of dosing issues; |
| · | lack of effectiveness during clinical trials; |
| · | slower than expected rates of patient recruitment; |
| · | inability to monitor patients adequately during or after treatment; |
| · | inability or unwillingness of medical investigators and institutional review boards to follow our clinical protocols; and |
| · | lack of sufficient funding to finance the clinical trials. |
Any failure or delay in commencement or completion of any clinical trials may have a material adverse effect on our business, results of operations and financial condition. In addition, we or the FDA or other regulatory authorities may suspend any clinical trial at any time if it appears that we are exposing participants in the trial to unacceptable safety or health risks or if the FDA or such other regulatory authorities, as applicable, find deficiencies in our IND submissions or the conduct of the trial. Any suspension of a clinical trial may have a material adverse effect on our business, results of operations and financial condition.
If the results of our clinical trials do not support our claims relating to any drug candidate or if serious side effects are identified, the completion of development of such drug candidate may be significantly delayed or we may be forced to abandon development altogether, which will significantly impair our ability to generate product revenues.
The results of our clinical trials with respect to any drug candidate might not support our claims of safety or efficacy, the effects of our drug candidates may not be the desired effects or may include undesirable side effects or the drug candidates may have other unexpected characteristics. Further, success in preclinical testing and early clinical trials does not ensure that later clinical trials will be successful, and the results of later clinical trials may not replicate the results of prior clinical trials and preclinical testing. The clinical trial process may fail to demonstrate that our drug candidates are safe for humans and effective for indicated uses. In addition, our clinical trials may involve a specific and small patient population. Results of early clinical trials conducted on a small patient population may not be indicative of future results. Adverse or inconclusive results may cause us to abandon a drug candidate and may delay development of other drug candidates. Any delay in, or termination of, our clinical trials will delay the filing of NDAs with the FDA, or other filings with other foreign regulatory authorities, and, ultimately, significantly impair our ability to commercialize our drug candidates and generate product revenues which would have a material adverse effect on our business, results of operations and financial condition.
We may find it difficult to enroll patients in our clinical trials, which could cause significant delays in the completion of such trials or may cause us to abandon one or more clinical trials.
Many of the diseases or disorders that our drug candidates are intended to treat are relatively rare and we expect only a subset of the patients with these diseases to be eligible for our clinical trials. Given that each of our drug candidates, other than taliglucerase alfa and our acetylcholinesterase product, is in the preclinical or research stages, we may not be able to initiate clinical trials for any of our drug candidates if we are unable to locate a sufficient number of eligible subjects to participate in the clinical trials required by the FDA and/or other foreign regulatory authorities. The requirements of our clinical trials generally mandate that a patient cannot be involved in another clinical trial for the same indication. We are aware of certain companies that have ongoing clinical trials for products that are competitive with our drug candidates, and subjects who would otherwise be eligible for our clinical trials may be involved in such trials, rendering them unavailable for testing of our drug candidates. Our inability to enroll a sufficient number of patients for any of our current or future clinical trials would result in significant delays or may require us to abandon one or more clinical trials altogether, which will have a material adverse effect on our business, results of operations and financial condition.
Patients may discontinue their participation in our clinical trials, which may negatively impact the results of these studies and extend the timeline for completion of our development programs.
Patients enrolled in our clinical studies may discontinue their participation at any time during the study as a result of a number of factors, including withdrawing their consent or experiencing adverse clinical events, which may or may not be judged related to our drug candidates under evaluation. If a large number of patients in any one of our studies discontinue their participation in the study, the results from that study may not be positive or may not support a filing for regulatory approval of the applicable drug candidate, which would have a material adverse effect on our business, results of operations and financial condition.
| 41 |
Because our clinical trials depend upon third-party researchers, the results of our clinical trials and such research activities are subject to delays and other risks which are, to a certain extent, beyond our control, which could impair our clinical development programs and our competitive position.
We depend upon independent investigators and collaborators, such as universities and medical institutions, to conduct our preclinical and clinical trials. These collaborators are not our employees, and we cannot control the amount or timing of resources that they devote to our clinical development programs. The investigators may not assign as great a priority to our clinical development programs or pursue them as diligently as we would if we were undertaking such programs directly. If outside collaborators fail to devote sufficient time and resources to our clinical development programs, or if their performance is substandard, the approval of our NDA, MAAs and other applications, and our introduction of new drugs, if any, may be delayed which could impair our clinical development programs and would have a material adverse effect on our business and results of operations. The collaborators may also have relationships with other commercial entities, some of whom may compete with us. If our collaborators also assist our competitors, our competitive position could be harmed.
The manufacture of our products is an exacting and complex process, and if we or one of our materials suppliers encounter problems manufacturing our products, it will have a material adverse effect on our business and results of operations.
The FDA and foreign regulators require manufacturers to register manufacturing facilities. The FDA and foreign regulators also inspect these facilities to confirm compliance with cGMP or similar requirements that the FDA or foreign regulators establish. We or our materials suppliers may face manufacturing or quality control problems causing product production and shipment delays or a situation where we or the supplier may not be able to maintain compliance with the FDA’s cGMP requirements, or those of foreign regulators, necessary to continue manufacturing our drug candidates. Any failure to comply with cGMP requirements or other FDA or foreign regulatory requirements could adversely affect our clinical research activities and our ability to market and develop our products. To date, our current facility has passed audits by the FDA, the Israeli MOH, ANVISA and the IMB on behalf of the EMA but remains subject to audit by other foreign regulatory authorities. There can be no assurance that we will be able to comply with FDA or foreign regulatory manufacturing requirements for our current facility or any facility we may establish in the future, which would have a material adverse effect on our business, results of operations and financial condition.
We rely on third parties for final processing of taliglucerase alfa, which exposes us to a number of risks that may delay development, regulatory approval and commercialization of our product candidates or result in higher product costs.
We have no experience in the final filling and freeze drying steps of the drug manufacturing process. According to our license and supply agreement with Pfizer, we currently are responsible for the fill and finish activities for taliglucerase alfa, but such functions will be transferred to Pfizer in the future. In addition, we have engaged a European contract manufacturer to act as an additional source of fill and finish activities for taliglucerase alfa. We currently rely primarily on other third-party contractors to perform the final manufacturing steps for taliglucerase alfa on a commercial scale. We may be unable to identify manufacturers and/or replacement manufacturers on acceptable terms or at all because the number of potential manufacturers is limited and the FDA and other regulatory authorities, as applicable, must approve any manufacturer and/or replacement manufacturer, including us, and we or any such third party manufacturer might be unable to formulate and manufacture our drug products in the volume and of the quality required to meet our clinical and commercial needs. If we engage any contract manufacturers, such manufacturers may not perform as agreed or may not remain in the contract manufacturing business for the time required to supply our clinical or commercial needs. In addition, contract manufacturers are subject to the rules and regulations of the FDA and comparable foreign regulatory authorities and face the risk that any of those authorities may find that they are not in compliance with applicable regulations. Each of these risks could delay our clinical trials, the approval, if any, of taliglucerase alfa and our other potential drug candidates by the FDA and other regulatory authorities, or the commercialization of taliglucerase alfa and our other drug candidates or could result in higher product costs or otherwise deprive us of potential product revenues.
If physicians, patients, third party payors and others in the medical community do not accept and use our drugs, our ability to generate revenue from sales of our products under development will be materially impaired.
Even if the FDA or other foreign regulatory authorities approve any of our drug candidates for commercialization, physicians and patients, and other healthcare providers, may not accept and use such candidates. Future acceptance and use of our products will depend upon a number of factors including:
| 42 |
| · | perceptions by physicians, patients, third party payors and others in the medical community about the safety and effectiveness of our drug candidates; |
| · | the willingness of the target patient population to try new therapies and of physicians to prescribe these therapies; |
| · | the prevalence and severity of any side effects, including any limitations or warnings contained in our products’ approved labeling; |
| · | pharmacological benefits of our products relative to competing products and products under development; |
| · | the efficacy and potential advantages relative to competing products and products under development; |
| · | relative convenience and ease of administration; |
| · | effectiveness of education, marketing and distribution efforts by us and our licensees and distributors, if any; |
| · | publicity concerning our products or competing products and treatments; |
| · | coverage and reimbursement of our products by third party payors; and |
| · | the price for our products and competing products. |
Because we expect sales of our current drug candidates, if approved, to generate substantially all of our product revenues for the foreseeable future, any lack of market acceptance of our drug candidates would have a material adverse effect on our business and financial condition, and revenues from sales of our products would be materially impaired.
We have no experience selling, marketing or distributing products and no internal capability to do so.
We currently have very limited sales, marketing or distribution capabilities and no experience in building a sales force and distribution capabilities. To be able to commercialize taliglucerase alfa upon approval, if at all, in Israel, and to commercialize any of our other product candidates, we must either develop internal sales, marketing and distribution capabilities, which will be expensive and time consuming, or make arrangements with third parties to perform these services. In November 2009, we granted to Pfizer an exclusive, worldwide right to develop and commercialize taliglucerase alfa, but retained such rights in Israel. If we decide to market any of our products directly, we must commit significant financial and managerial resources to develop a marketing and sales force with technical expertise and with supporting distribution capabilities. Factors that may inhibit our efforts to commercialize our products directly and without strategic partners include:
| · | our inability to recruit and retain adequate numbers of effective sales and marketing personnel; |
| · | the inability of sales personnel to obtain access to an adequate numbers of physicians or to pursuance them to prescribe our products; |
| · | the lack of complementary products to be offered by sales personnel, which may put us at a competitive disadvantage relative to companies with more extensive product lines; and |
| · | unforeseen costs and expenses associated with creating and sustaining an independent sales and marketing organization. |
We may not be successful in recruiting the sales and marketing personnel necessary to sell any of our products upon approval, if at all, and even if we do build a sales force, it may not be successful in marketing our products, which would have a material adverse effect on our business, results of operations and financial condition.
We may enter into distribution arrangements and marketing alliances for certain products and any failure to successfully identify and implement these arrangements on favorable terms, if at all, may impair our ability to commercialize our product candidates.
While we intend to build a sales force to market taliglucerase alfa in Israel and other product candidates worldwide, we do not anticipate having the resources in the foreseeable future to develop global sales and marketing capabilities for all of the products we develop, if any. We may pursue arrangements regarding the sales and marketing and distribution of one or more of our product candidates, such as our license and supply agreement with Pfizer, and our future revenues may depend, in part, on our ability to enter into and maintain arrangements with other companies having sales, marketing and distribution capabilities and the ability of such companies to successfully market and sell any such products. Any failure to enter into such arrangements and marketing alliances on favorable terms, if at all, could delay or impair our ability to commercialize our product candidates and could increase our costs of commercialization. Any use of distribution arrangements and marketing alliances to commercialize our product candidates will subject us to a number of risks, including the following:
| · | we may be required to relinquish important rights to our products or product candidates; |
| · | we may not be able to control the amount and timing of resources that our distributors or collaborators may devote to the commercialization of our product candidates; |
| 43 |
| · | our distributors or collaborators may experience financial difficulties; |
| · | our distributors or collaborators may not devote sufficient time to the marketing and sales of our products; and |
| · | business combinations or significant changes in a collaborator’s business strategy may adversely affect a collaborator’s willingness or ability to complete its obligations under any arrangement. |
We may need to enter into additional co-promotion arrangements with third parties where our own sales force is neither well situated nor large enough to achieve maximum penetration in the market. We may not be successful in entering into any co-promotion arrangements, and the terms of any co-promotion arrangements we enter into may not be favorable to us.
If the market opportunities for our current product candidates are smaller than we believe they are, our revenues may be adversely affected and our business may suffer.
A substantial focus of our current clinical pipeline is on relatively rare disorders with small patient populations, in particular Gaucher disease and Fabry disease. Currently, most reported estimates of the prevalence of these diseases are based on studies of small subsets of the population of specific geographic areas, which are then extrapolated to estimate the prevalence of the diseases in the broader world population. As new studies are performed, the estimated prevalence of these diseases may change. There can be no assurance that the prevalence of Gaucher disease or Fabry disease in the study populations, particularly in these newer studies, accurately reflect the prevalence of these diseases in the broader world population. If the market opportunities for our current product candidates are smaller than we believe they are, our revenues may be adversely affected and our business may suffer.
Developments by competitors may render our products or technologies obsolete or non-competitive which would have a material adverse effect on our business, results of operations and financial condition.
We compete against fully integrated pharmaceutical companies and smaller companies that are collaborating with larger pharmaceutical companies, academic institutions, government agencies and other public and private research organizations. Our drug candidates will have to compete with existing therapies and therapies under development by our competitors. In addition, our commercial opportunities may be reduced or eliminated if our competitors develop and market products that are less expensive, more effective or safer than our drug products. Other companies have drug candidates in various stages of preclinical or clinical development to treat diseases for which we are also seeking to develop drug products. Some of these potential competing drugs are further advanced in development than our drug candidates and may be commercialized earlier. Even if we are successful in developing effective drugs, our products may not compete successfully with products produced by our competitors.
We specifically face competition from companies with approved treatments of Gaucher disease, including Genzyme, Shire and to a much lesser extent, Actelion. In February 2010, the FDA approved VPRIV, Shire’s enzyme replacement therapy for the treatment of Gaucher disease, and the European Commission granted marketing authorization to VPRIV in August 2010. In addition, we are aware of other early stage, experimental, small molecule, oral drugs which are being developed for the treatment of Gaucher disease by each of Genzyme and Amicus Therapeutics. According to Amicus Therapeutics, its trial of a small molecule, oral drug for the treatment of Gaucher disease has been suspended. We also face competition from companies with approved treatments of Fabry disease, including Genzyme and Shire, and we are aware of other early stage drugs which are being developed for the treatment of Fabry disease, including a drug being developed by Amicus Therapeutics. Amicus Therapeutics reports that it has suspended its development of its Gaucher disease treatment. We also face competition from companies with approved enzyme treatments of Fabry disease, including Genzyme and Shire, and we are aware of other early stage drugs which are being developed for the treatment of Fabry disease, including a drug being developed by Amicus Therapeutics.
We also face competition from companies that are developing other platforms for the expression of recombinant therapeutic pharmaceuticals. We are aware of companies that are developing alternative technologies to develop and produce therapeutic proteins in anticipation of the expiration of certain patent claims covering marketed proteins. Competitors developing alternative expression technologies include Crucell N.V. (which was acquired by Johnson & Johnson), Shire and GlycoFi Inc. (which was acquired by Merck). Other companies are developing alternate plant-based technologies, include Biolex, Inc., Chlorogen, Inc., iBIO, Medicago, Greenovation Biotech GmbH and Dow Agroscience.
Several biogeneric companies are pursuing the opportunity to develop and commercialize follow-on versions of other currently marketed biologic products, including growth factors, hormones, enzymes, cytokines and monoclonal antibodies, which are areas that interest us. These companies include, among others, Novartis AG/Sandoz Pharmaceuticals, BioGeneriX AG, Stada Arzneimittel AG, BioPartners GmbH and Teva.
| 44 |
Most of our competitors, either alone or together with their collaborative partners, operate larger research and development programs, staff and facilities and have substantially greater financial resources than we do, as well as significantly greater experience in:
| · | developing drugs; |
| · | undertaking preclinical testing and human clinical trials; |
| · | obtaining marketing approvals from the FDA and other regulatory authorities; |
| · | formulating and manufacturing drugs; and |
| · | launching, marketing and selling drugs. |
These organizations also compete with us to attract qualified personnel, acquisitions and joint ventures candidates and for other collaborations. Activities of our competitors may impose unanticipated costs on our business which would have a material adverse effect on our business, results of operations and financial condition.
If we in-license drug candidates, we may delay or otherwise adversely affect the development of our existing drug candidates, which may negatively impact our business, results of operations and financial condition.
In addition to our own internally developed drug candidates, we proactively seek opportunities to in-license and advance other drug candidates that are strategic and have value-creating potential to take advantage of our development know-how and technology. If we in-license any additional drug candidate, our capital requirements may increase significantly. In addition, in-licensing additional drug candidates may place a strain on the time of our existing personnel, which may delay or otherwise adversely affect the development of our existing drug candidates or cause us to re-prioritize our drug pipeline if we do not have the necessary capital resources to develop all of our drug candidates, which may delay the development of our drug candidates and materially and adversely impact our business, results of operations and financial condition.
If we are unable to successfully manage our growth, there could be a material adverse impact on our business, results of operations and financial condition.
We have grown rapidly and expect to continue to grow. We expect to hire more employees, particularly in the areas of drug development, manufacturing, regulatory affairs and sales and marketing, and increase our facilities and corporate infrastructure, further increasing the size of our organization and related expenses. To manage our anticipated future growth, we must continue to implement and improve our managerial, operational and financial systems, expand our facilities and continue to recruit and train additional qualified personnel. Due to our limited resources, we may not be able to effectively manage the expansion of our operations or recruit and train additional qualified personnel. The expansion of our operations may lead to significant costs and may divert our management and business development resources. Any inability on the part of our management to manage growth could delay the execution of our business plans or disrupt our operations. If we are unable to manage our growth effectively, we may not use our resources in an efficient manner, which may delay the development of our drug candidates and materially and adversely impact our business, results of operations and financial condition.
If we acquire companies, products or technologies, we may face integration risks and costs associated with those acquisitions that could negatively impact our business, results of operations and financial condition.
If we are presented with appropriate opportunities, we may acquire or make investments in complementary companies, products or technologies. We may not realize the anticipated benefit of any acquisition or investment. If we acquire companies or technologies, we will face risks, uncertainties and disruptions associated with the integration process, including difficulties in the integration of the operations of an acquired company, integration of acquired technology with our products, diversion of our management’s attention from other business concerns, the potential loss of key employees or customers of the acquired business and impairment charges if future acquisitions are not as successful as we originally anticipate. In addition, our operating results may suffer because of acquisition-related costs or amortization expenses or charges relating to acquired intangible assets. Any failure to successfully integrate other companies, products or technologies that we may acquire may have a material adverse effect on our business and results of operations. Furthermore, we may have to incur debt or issue equity securities to pay for any additional future acquisitions or investments, the issuance of which could be dilutive to our existing shareholders.
| 45 |
We depend upon key employees and consultants in a competitive market for skilled personnel. If we are unable to attract and retain key personnel, it could adversely affect our ability to develop and market our products.
We are highly dependent upon the principal members of our management team, especially our President and Chief Executive Officer, Dr. David Aviezer, Ph.D., as well as the Interim Chairman of our Board of Directors, Zev Bronfeld, our other directors, our scientific advisory board members, consultants and collaborating scientists. Many of these people have been involved with us for many years and have played integral roles in our progress, and we believe that they will continue to provide value to us. A loss of any of these personnel may have a material adverse effect on aspects of our business, clinical development and regulatory programs. We have employment agreements with Dr. Aviezer and five other officers that may be terminated by us or the applicable officer at any time with varying notice periods of 60 to 90 days. Although these employment agreements generally include non-competition covenants and provide for severance payments that are contingent upon the applicable employee’s refraining from competition with us, the applicable noncompetition provisions can be difficult and costly to monitor and enforce. The loss of any of these persons’ services may adversely affect our ability to develop and market our products and obtain necessary regulatory approvals. Further, we do not maintain key-man life insurance.
We also depend in part on the continued service of our key scientific personnel and our ability to identify, hire and retain additional personnel, including marketing and sales staff. We experience intense competition for qualified personnel, and the existence of non-competition agreements between prospective employees and their former employers may prevent us from hiring those individuals or subject us to suit from their former employers. While we attempt to provide competitive compensation packages to attract and retain key personnel, many of our competitors are likely to have greater resources and more experience than we have, making it difficult for us to compete successfully for key personnel.
Our collaborations with outside scientists and consultants may be subject to restriction and change.
We work with chemists, biologists and other scientists at academic and other institutions, and consultants who assist us in our research, development, regulatory and commercial efforts, including the members of our scientific advisory board. These scientists and consultants have provided, and we expect that they will continue to provide, valuable advice regarding our programs. These scientists and consultants are not our employees, may have other commitments that would limit their future availability to us and typically will not enter into non-compete agreements with us. If a conflict of interest arises between their work for us and their work for another entity, we may lose their services. In addition, we will be unable to prevent them from establishing competing businesses or developing competing products. For example, if a key scientist acting as a principal investigator in any of our clinical trials identifies a potential product or compound that is more scientifically interesting to his or her professional interests, his or her availability to remain involved in our clinical trials could be restricted or eliminated.
Under current U.S. and Israeli law, we may not be able to enforce employees’ covenants not to compete and therefore may be unable to prevent our competitors from benefiting from the expertise of some of our former employees.
We have entered into non-competition agreements with all of our employees. These agreements prohibit our employees, if they cease working for us, from competing directly with us or working for our competitors for a limited period. Under current U.S. and Israeli law, we may be unable to enforce these agreements against most of our employees and it may be difficult for us to restrict our competitors from gaining the expertise our former employees gained while working for us. If we cannot enforce our employees’ non-compete agreements, we may be unable to prevent our competitors from benefiting from the expertise of our former employees.
If product liability claims are brought against us, it may result in reduced demand for our products and product candidates or damages that exceed our insurance coverage.
The clinical testing, marketing and use of our products and product candidates exposes us to product liability claims if the use or misuse of those products or product candidates cause injury, disease or results in adverse effects. Use of our products or product candidates, whether in clinical trials or post approval, could result in product liability claims. We presently carry clinical trial liability insurance with coverages of up to $10.0 million per occurrence and $10.0 million in the aggregate, an amount we consider reasonable and customary . However, this insurance coverage includes various deductibles, limitations and exclusions from coverage, and in any event might not fully cover any potential claims. We may need to obtain additional clinical trial liability coverage prior to initiating additional clinical trials. We expect to obtain product liability insurance coverage before commercialization of our product candidates; however, such insurance is expensive and insurance companies may not issue this type of insurance when we need it. We may not be able to obtain adequate insurance in the future at an acceptable cost. Any product liability claim, even one that was not in excess of our insurance coverage or one that is meritless and/or unsuccessful, may adversely affect our cash available for other purposes, such as research and development, which may have a material adverse effect on our business, results of operations and financial condition. Product liability claims, even if without merit, may result in reduced demand for our products, if approved, which would have a material adverse effect on our business, financial condition and results of operations. In addition, the existence of a product liability claim could affect the market price of our common stock.
| 46 |
Coverage and reimbursement may not be available for our product candidates, which could diminish our sales or affect our ability to sell our products profitably.
Market acceptance and sales of our product candidates will depend on worldwide coverage and reimbursement policies. Government authorities and third-party payors, such as private health insurers and health maintenance organizations, decide which drugs they will pay for and establish reimbursement levels. We cannot be sure that coverage and reimbursement will be available for any of our product candidates, if approved for marketing and sale. Obtaining reimbursement approval for an approved product from governments and other third party payors is a time consuming and costly process that requires us to provide supporting scientific, clinical and cost-effectiveness data for the use of our products, if and when approved, to every payor. We may not be able to provide data sufficient to gain acceptance with respect to coverage and reimbursement or we might need to conduct post-marketing studies in order to demonstrate the cost-effectiveness of approved products, if any, to such payors’ satisfaction. Such studies might require us to commit a significant amount of management time and financial and other resources. Even if a payor determines that an approved product is eligible for reimbursement, the payor may impose coverage limitations that preclude payment for some uses that are approved by the FDA or other regulatory authorities. In addition, there is a risk that full reimbursement may not be available for high priced products. Moreover, eligibility for coverage does not imply that any approved product will be reimbursed in all cases or at a rate that allows us to make a profit or even cover our costs. Also, limited reimbursement amounts may reduce the demand for, or the price of, our product candidates. Except with respect to taliglucerase alfa, we have not commenced efforts to have our product candidates covered and reimbursed by government or third-party payors. If coverage and reimbursement are not available or are available only to limited levels, the sales of our products, if approved may be diminished or we may not be able to sell such products profitably.
Reforms in the healthcare industry and the uncertainty associated with pharmaceutical pricing, reimbursement and related matters could adversely affect the marketing, pricing and demand for our products, if approved.
Increasing healthcare expenditures have been the subject of considerable public attention in the United States. Both private and government entities are seeking ways to reduce or contain healthcare costs. Numerous proposals that would affect changes in the U.S. healthcare system have been introduced or proposed in the U.S. Congress and in some state legislatures within the United States, including reductions in the pricing of prescription products and changes in the levels at which consumers and healthcare providers are reimbursed for purchases of pharmaceutical products. Legislation passed in recent years has imposed certain changes to the way in which drugs, including our product candidates, are covered and reimbursed in the United States. For example, federal legislation and regulations have implemented new reimbursement methodologies for certain drugs, created a voluntary prescription drug benefit, Medicare Part D, and have imposed significant revisions to the Medicaid Drug Rebate Program. The recently enacted Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act (collectively, the “PPACA”), imposes yet additional changes to these programs. We believe that legislation that reduces reimbursement for our product candidates could adversely impact how much or under what circumstances healthcare providers will prescribe or administer our products, if approved. This could materially and adversely impact our business by reducing our ability to generate revenue, raise capital, obtain additional collaborators and market our products, if approved. In addition, we believe the increasing emphasis on managed care in the United States has and will continue to put pressure on the price and usage of pharmaceutical products, which may adversely impact product sales, upon approval, if at all.
We are subject to healthcare reform measures, including legislation, regulatory proposals and healthcare payor initiatives that may increase our costs and adversely affect our profitability and/or ability to obtain adequate reimbursement for our product candidates.
The United States and some foreign jurisdictions are considering or have enacted a number of legislative and regulatory proposals to change the healthcare system in ways that could affect our ability to sell our products profitably. Among policymakers and payors in the United States and elsewhere, there is significant interest in promoting changes in healthcare systems with the stated goals of containing healthcare costs, improving quality and/or expanding access. In the United States, the pharmaceutical industry has been a particular focus of these efforts and has been significantly affected by major legislative initiatives. For example, the PPACA which was enacted in the United States in March 2010 substantially changes the way healthcare is financed in the United States by both governmental and private insurers and significantly affects the pharmaceutical industry. Although we cannot predict their full impact, we anticipate that PPACA, as well as other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage and reimbursement criteria and in additional downward pressure on the price that we receive for any approved product, and could adversely affect our profits and our business generally.
| 47 |
We may be subject, directly or indirectly, to federal and state healthcare fraud and abuse and false claims laws and regulations. If we are unable to comply, or have not fully complied, with such laws, we could face substantial penalties.
If we obtain FDA approval for any of our product candidates and begin commercializing those products in the United States, our operations may be directly, or indirectly through our customers, subject to various state and federal fraud and abuse laws, including, without limitation, the federal Anti-Kickback Statute, the federal False Claims Act and HIPAA. These laws may impact, among other things, our proposed sales, marketing and education programs.
The federal Anti-Kickback Statute prohibits persons from knowingly and willfully soliciting, receiving, offering or paying remuneration, directly or indirectly, to induce either the referral of an individual, or the furnishing, recommending, or arranging for a good or service, for which payment may be made under a federal healthcare program, such as the Medicare and Medicaid programs. The term “remuneration” has been broadly interpreted to include anything of value, including for example, gifts, discounts, the furnishing of supplies or equipment, credit arrangements, payments of cash, waivers of co-payments and deductibles, ownership interests and providing anything at less than its fair market value. The reach of the Anti-Kickback Statute was also broadened by PPACA, which, among other things, amends the intent requirement of the federal Anti-Kickback Statute and the applicable criminal healthcare fraud statutes contained within 42 U.S.C. § 1320a-7b, effective March 23, 2010. Pursuant to the statutory amendment, a person or entity no longer needs to have actual knowledge of this statute or specific intent to violate it in order to have committed a violation. In addition, PPACA provides that the government may assert that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the civil False Claims Act (discussed below) or the civil monetary penalties statute, which imposes penalties against any person who is determined to have presented or caused to be presented a claim to a federal health program that the person knows or should know is for an item or service that was not provided as claimed or is false or fraudulent. The federal Anti-Kickback Statute is broad, and despite a series of narrow safe harbors, prohibits many arrangements and practices that are lawful in businesses outside of the healthcare industry. Penalties for violations of the federal Anti-Kickback Statute include criminal penalties and civil sanctions such as fines, imprisonment and possible exclusion from Medicare, Medicaid and other state or federal healthcare programs. Many states have also adopted laws similar to the federal Anti-Kickback Statute, some of which apply to the referral of patients for healthcare items or services reimbursed by any source, not only the Medicare and Medicaid programs, and do not contain identical safe harbors.
The federal False Claims Act imposes liability on any person who, among other things, knowingly presents, or causes to be presented, a false or fraudulent claim for payment by a federal healthcare program. The “qui tam” provisions of the False Claims Act allow a private individual to bring civil actions on behalf of the federal government alleging that the defendant has submitted a false claim to the federal government, and to share in any monetary recovery. In addition, various states have enacted false claims laws analogous to the False Claims Act. Many of these state laws apply where a claim is submitted to any third-party payer and not merely a federal healthcare program. When an entity is determined to have violated the False Claims Act, it may be required to pay up to three times the actual damages sustained by the government, plus civil penalties of $5,500 to $11,000 for each separate false claim.
Also, the Health Insurance Portability and Accountability Act of 1996, or HIPAA, created several new federal crimes, including health care fraud, and false statements relating to health care matters. The health care fraud statute prohibits knowingly and willfully executing a scheme to defraud any health care benefit program, including private third-party payers. The false statements statute prohibits knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for health care benefits, items or services.
We are unable to predict whether we could be subject to actions under any of these or other fraud and abuse laws, or the impact of such actions. Moreover, to the extent that our products will be sold in a foreign country, we may be subject to similar foreign laws and regulations. If we are found to be in violation of any of the laws described above and other applicable state and federal fraud and abuse laws, we may be subject to penalties, including civil and criminal penalties, damages, fines, exclusion from government healthcare reimbursement programs and the curtailment or restructuring or our operations, all of which could have a material adverse effect on our business, results of operations and financial condition.
| 48 |
Governments outside the United States tend to impose strict price controls and reimbursement approval policies, which may adversely affect our prospects for generating revenue.
In some countries, particularly European Union member states, the pricing of prescription pharmaceuticals is subject to governmental control. In these countries, pricing negotiations with governmental authorities can take considerable time (six to 12 months or longer) after the receipt of marketing approval for a product. To obtain reimbursement or pricing approval in some countries with respect to any product candidate that achieves regulatory approval, we may be required to conduct a clinical trial that compares the cost-effectiveness of our product candidate to other available therapies. If reimbursement of our products upon approval, if at all, is unavailable or limited in scope or amount, or if pricing is set at unsatisfactory levels, our prospects for generating revenue, if any, could be adversely affected which would have a material adverse effect on our business, results of operations and financial condition. Further, if we achieve regulatory approval of any product, we must successfully negotiate product pricing for such product in individual countries. As a result, the pricing of our products, if approved, in different countries may vary widely, thus creating the potential for third-party trade in our products in an attempt to exploit price differences between countries. This third-party trade of our products could undermine our sales in markets with higher prices which could have a material adverse effect on our business, results of operations and financial condition.
Our ability to utilize net operating loss carryforwards may be limited.
The net operating loss carryforwards, or “NOLs”, of the Company as of December 31, 2011, equal to approximately $11.7 million, may be restricted under Section 382 of the Internal Revenue Code of 1986, as amended, or the “Code.” Section 382 of the Code imposes limitations on a corporation’s ability to utilize NOLs to offset taxable income if the corporation experiences an “ownership change.” In general terms, an “ownership change” may result from transactions increasing the ownership of certain stockholders in the stock of a corporation by more than 50% over a three-year period. In the event that an ownership change has occurred, or were to occur, utilization of our NOLs would be subject to an annual limitation under Section 382, which is generally the fair market value of the pre-change entity multiplied by the long-term tax exempt rate, which is published monthly by the Internal Revenue Service.
All liabilities of our company have survived the merger and there may be undisclosed liabilities that could harm our revenues, business, prospects, financial condition and results of operations.
Protalix Ltd. and its counsel conducted due diligence on us that was customary and appropriate for the reverse merger transaction consummated on December 31, 2006. However, the due diligence process may not have revealed all our material liabilities then existing or that could be asserted in the future against us relating to our activities before the consummation of the merger. Any such potential liabilities survive the merger and could harm our revenues, business, prospects, results of operations and financial condition.
We are a holding company with no operations of our own.
We are a holding company with no operations of our own. Accordingly, our ability to conduct our operations, service any debt that we may incur in the future and pay dividends, if any, is dependent upon the earnings from the business conducted by Protalix Ltd. The distribution of those earnings or advances or other distributions of funds by our subsidiary to us, as well as our receipt of such funds, are contingent upon the earnings of our subsidiary and are subject to various business considerations and U.S. and Israeli law. If Protalix Ltd. is unable to make sufficient distributions or advances to us, or if there are limitations on our ability to receive such distributions or advances, we may not have the cash resources necessary to conduct our corporate operations which would have a material adverse effect on our business, results of operations and financial condition.
Risks Related to Regulatory Matters
We are subject to extensive governmental regulation including the requirement of FDA approval before our drug candidates may be marketed.
Both before and after approval of our drug candidates, we, our drug candidates, our suppliers, our contract manufacturers and our contract testing laboratories are subject to extensive regulation by the FDA or comparable foreign regulatory authorities. Failure to comply with applicable requirements of the FDA or comparable foreign regulatory authorities could result in, among other things, any of the following actions:
| · | warning letters; |
| · | fines and other monetary penalties; |
| 49 |
| · | unanticipated expenditures; |
| · | delays in the FDA’s or other foreign regulatory authorities’ approving, or the refusal of any regulatory authority to approve, any drug candidate; |
| · | product recall or seizure; |
| · | interruption of manufacturing or clinical trials; |
| · | operating restrictions; |
| · | injunctions; and |
| · | criminal prosecutions. |
In addition to the approval requirements, other numerous and pervasive regulatory requirements apply, both before and after approval, to us, our drug candidates, and our suppliers, contract manufacturers, and contract laboratories. These include requirements related to:
| · | testing; |
| · | manufacturing; |
| · | quality control; |
| · | labeling; |
| · | advertising; |
| · | promotion; |
| · | distribution; |
| · | export; |
| · | reporting to the FDA certain adverse experiences associated with use of the drug candidate; and |
| · | obtaining additional approvals for certain modifications to the drug candidate or its labeling or claims. |
We also are subject to inspection by the FDA and comparable foreign regulatory authorities, to determine our compliance with regulatory requirements, as are our suppliers, contract manufacturers, and contract testing laboratories, and there can be no assurance that the FDA or any other comparable foreign regulatory authority, will not identify compliance issues that may disrupt production or distribution, or require substantial resources to correct. We may be required to make modifications to our manufacturing operations in response to these inspections which may require significant resources and may have a material adverse effect upon our business, results of operations and financial condition.
The approval process for any drug candidate may also be delayed by changes in government regulation, future legislation or administrative action or changes in policy of the FDA and comparable foreign authorities that occur prior to or during their respective regulatory reviews of such drug candidate. Delays in obtaining regulatory approvals with respect to any drug candidate may:
| · | delay commercialization of, and our ability to derive product revenues from, such drug candidate; |
| · | delay the regulatory-related milestone payments we anticipate receiving from Pfizer; |
| · | require us to perform costly procedures with respect to such drug candidate; or |
| · | otherwise diminish any competitive advantages that we may have with respect to such drug candidate. |
Delays in the approval process for any drug candidate may have a material adverse effect upon our business, financial condition and results of operations.
We may not obtain the necessary U.S., EMA or other worldwide regulatory approvals to commercialize our drug candidates in a timely manner, if at all, which would have a material adverse effect on our business, financial condition and results of operations.
We need FDA approval to commercialize our drug candidates in the United States, EMA approval to commercialize our drug candidates in the European Union and approvals from foreign regulators to commercialize our drug candidates elsewhere. In order to obtain FDA approval of any of our drug candidates, we must submit to the FDA an NDA or a Biologic License Application (BLA) demonstrating that the drug candidate is safe for humans and effective for its intended use. This demonstration requires significant research and animal tests, which are referred to as preclinical studies, as well as human tests, which are referred to as clinical trials. In the European Union, we must submit an MAA to the EMA. Satisfaction of the FDA’s, the EMA's and foreign regulatory authorities’ regulatory requirements typically takes many years, depends upon the type, complexity and novelty of the drug candidate and requires substantial resources for research, development and testing. In December 2009, we completed the filing of an NDA for taliglucerase alfa for the treatment of Gaucher disease and received a PDUFA date of February 25, 2011. In addition, we have submitted a marketing application for taliglucerase alfa to the Israeli MOH, and MAAs have been submitted to each of the EMA, ANVISA and the Australian TGA.
| 50 |
In February 2011, we received a CRL from the FDA regarding our NDA for taliglucerase alfa for the treatment of Gaucher disease. The main questions raised by the FDA regarding the NDA related to clinical and CMC. In the clinical section of the CRL, the FDA requested additional data from the ongoing switchover trial and the long-term extension trial relating to taliglucerase alfa. At the time the NDA was submitted, full data from these trials was not available. In the CMC section of the CRL, the FDA requested information regarding testing specifications and assay validation. The FDA did not request additional clinical studies in the CRL. In May 2011, we had a meeting with the FDA to clarify the FDA’s requests and in July 2011, we submitted a reply to the CRL. In August 2011, the FDA notified us that it had accepted for review the resubmission of the taliglucerase alfa NDA, had deemed the resubmission a class 2, or 6-month, response and established February 1, 2012 as the new PDUFA date. In December 2011, the FDA notified us that it had decided to extend the PDUFA date to May 1, 2012. In its notification, the FDA stated that its decision related to certain clinical information regarding taliglucerase alfa we had submitted in November 2011 in response to an FDA request. The requested information related mainly to the presentation of select data provided in the NDA. As this information was requested and provided within 90 days of the February 1, 2012 PDUFA goal date, the FDA had the option to extend the PDUFA goal date to provide adequate time for the FDA to complete its review. A three-month extension cycle is the standard period granted. No additional data was requested by the FDA in the notification, nor were we notified of any specific deficiency in the taliglucerase alfa NDA. There can be no assurance that the FDA will not make any additional request regarding our NDA. In the past, the FDA has made additional requests to other applicants after the delivery of a CRL. Any additional requests from the FDA relating to the NDA may delay or preclude the FDA’s review of our reply to the CRL. Even if we comply with all of the FDA’s requests in the CRL or otherwise, if any, the FDA may ultimately reject the NDA, or fail to approve the NDA in a timely manner, which would have a material adverse effect on our business, financial condition and results of operations.
Even if we comply with all the requests of the FDA, EMA and comparable foreign authorities, the authorities may ultimately reject the NDA, MAA or other filing or submission we filed for taliglucerase alfa or one or more of the NDAs or other filings or submissions we file in the future, if any, or we might not obtain regulatory clearance in a timely manner for taliglucerase alfa or any of our other drug candidates. Companies in the pharmaceutical and biotechnology industries have suffered significant setbacks in advanced or late-stage clinical trials, even after obtaining promising earlier trial results or in preliminary findings or other comparable authorities for such clinical trials. Further, even if favorable testing data is generated by the clinical trials of a drug candidate, the FDA, EMA or other regulatory authority may not accept or approve an NDA, MAA or other comparable submission, as applicable, filed by a pharmaceutical or biotechnology company for the drug candidate. Failure to obtain approval of the FDA, EMA or comparable foreign authorities of any of our drug candidates in a timely manner, if at all, will severely undermine our business, financial condition and results of operation by reducing our potential marketable products and our ability to generate corresponding product revenues.
Our research and clinical efforts may not result in drugs that the FDA, EMA or foreign regulatory authorities consider safe for humans and effective for indicated uses, which would have a material adverse effect on our business, financial condition and results of operations. After clinical trials are completed for any drug candidate, if at all, the FDA, EMA and foreign regulatory authorities have substantial discretion in the drug approval process of the drug candidate in their respective jurisdictions and may require us to conduct additional clinical testing or perform post-marketing studies which would cause us to incur additional costs. Incurring such costs may have a material adverse effect on our business, financial condition and results of operations.
We have only limited experience in regulatory affairs, and some of our drug candidates may be based on new technologies. These factors may affect our ability or the time we require to obtain necessary regulatory approvals.
We have only limited experience in filing and prosecuting the applications necessary to gain regulatory approvals for medical devices and drug candidates. Moreover, some of the drug candidates that are likely to result from our development programs may be based on new technologies that have not been extensively tested in humans. The regulatory requirements governing these types of drug candidates may be less well defined or more rigorous than for conventional products. As a result, we may experience a longer regulatory process in connection with obtaining regulatory approvals of any products that we develop.
| 51 |
If Shire or any of our other competitors are able to obtain orphan drug exclusivity for any products that are competitive with our products, we may be precluded from selling or obtaining approval of our competing products by the applicable regulatory authorities for a significant period of time.
In the United States, orphan drug status may be designated for a drug that has the potential to treat a “rare disease or condition,” which generally is a disease or condition that affects fewer than 200,000 individuals within the United States. Orphan drug designation does not convey an advantage in, or shorten the duration of, the review and approval process. If a drug candidate which has orphan drug designation subsequently receives the first approval for the indication in a jurisdiction for which it has such designation, the drug candidate is entitled to orphan exclusivity in that jurisdiction, meaning that the applicable regulatory authority may not approve any other application to market the same drug for the same indication, except in very limited circumstances, for seven years. There can be no assurance that a drug candidate that receives orphan drug designation will receive orphan drug marketing exclusivity. More than one drug can have orphan designation for the same indication.
Foreign regulations regarding orphan drugs are similar to those in the United States but there are several conceptual differences. For example, the exclusivity period in the European Union is generally 10 years. The EMA/European Commission has granted orphan drug designation and exclusivity to VPRIV in the European Union. This designation may preclude the marketing authorization of taliglucerase alfa in the European Union for a 10-year period commencing upon the August 2010 marketing authorization of VPRIV in the European Union. Any marketing protection granted to VPRIV by the EMA/European Commission in connection with the orphan drug designation may have a material adverse effect on our business, financial condition and results of operations.
From time to time, we may apply to the FDA or any comparable foreign regulatory authority for orphan drug designation for any one or more of our drug candidates. For example, in September 2009, the FDA’s Office of Orphan Product Development granted taliglucerase alfa orphan drug status. In January 2010, the COMP, after reviewing all relevant clinical data, recommended that the European Commission grant orphan drug designation to taliglucerase alfa for the treatment of Gaucher disease. However, none of our other drug candidates have been designated as an orphan drug and there is no guarantee that the FDA will grant such designation in the future. In addition, neither orphan drug designation nor orphan drug exclusivity prevents competitors from developing or marketing different drugs for that indication. Even if we obtain orphan drug exclusivity for one or more indications for one of our drug candidates, we may not be able to maintain the exclusivity. For example, if a competitive product that is the same drug or biologic as one of our drug candidates is shown to be clinically superior to the drug candidate, any orphan drug exclusivity granted to the drug candidate will not block the approval of the competitive product. The grant of orphan drug designation to VPRIV may preclude our ability to receive the same designation for taliglucerase alfa. Failure to obtain or maintain orphan drug exclusivity for taliglucerase alfa or any of our other drug candidates may have a material adverse effect on our business, results of operation and financial condition. If VPRIV or any other drug receives orphan drug exclusivity in any jurisdiction for Gaucher disease or any other indication for a product that competes with one of the indications for one of our drug candidates, we may be prevented from marketing the applicable drug candidate in the jurisdiction during the applicable exclusivity period which will have a material adverse effect on our business, results of operations and financial condition.
Risks Related to Intellectual Property Matters
If we fail to adequately protect or enforce our intellectual property rights or secure rights to third party patents, the value of our intellectual property rights would diminish and our business, competitive position and results of operations would suffer.
As of December 31, 2011, we had 81 pending patent applications, one joint pending patent application with a third party and license rights to seven pending patent applications. However, the filing of a patent application does not mean that we will be issued a patent, or that any patent eventually issued will be as broad as requested in the patent application or sufficient to protect our technology. Any modification required to a current patent application may delay the approval of such patent application which would have a material adverse effect on our business, results of operations and financial condition. In addition, there are a number of factors that could cause our patents, if granted, to become invalid or unenforceable or that could cause our patent applications to not be granted, including known or unknown prior art, deficiencies in the patent application or the lack of originality of the technology. Our competitive position and future revenues will depend in part on our ability and the ability of our licensors and collaborators to obtain and maintain patent protection for our products, methods, processes and other technologies, to preserve our trade secrets, to prevent third parties from infringing on our proprietary rights and to operate without infringing the proprietary rights of third parties. We have filed U.S. and international patent applications for process patents, as well as composition of matter patents, for taliglucerase alfa. However, we cannot predict:
| · | the degree and range of protection any patents will afford us against competitors and those who infringe upon our patents, including whether third parties will find ways to invalidate or otherwise circumvent our licensed patents; |
| · | if and when patents will issue; |
| 52 |
| · | whether or not others will obtain patents claiming aspects similar to those covered by our licensed patents and patent applications; or |
| · | whether we will need to initiate litigation or administrative proceedings, which may be costly, and whether we win or lose. |
As of December 31, 2011, we hold, or have license rights to, 38 patents (including one joint patent). If patent rights covering our products or technologies are not sufficiently broad, they may not provide us with sufficient proprietary protection or competitive advantages against competitors with similar products and technologies. Furthermore, if the U.S. Patent and Trademark Office or foreign patent offices issue patents to us or our licensors, others may challenge the patents or circumvent the patents, or the patent office or the courts may invalidate the patents. Thus, any patents we own or license from or to third parties may not provide any protection against our competitors and those who infringe upon our patents.
Furthermore, the life of our patents is limited. The patents we hold relating to our ProCellEx protein expression system will expire in 2017. If patents issue from other currently pending patent applications, those patents will expire between 2024 and 2028.
We rely on confidentiality agreements that could be breached and may be difficult to enforce which could have a material adverse effect on our business and competitive position.
Our policy is to enter agreements relating to the non-disclosure of confidential information with third parties, including our contractors, consultants, advisors and research collaborators, as well as agreements that purport to require the disclosure and assignment to us of the rights to the ideas, developments, discoveries and inventions of our employees and consultants while we employ them. However, these agreements can be difficult and costly to enforce. Moreover, to the extent that our contractors, consultants, advisors and research collaborators apply or independently develop intellectual property in connection with any of our projects, disputes may arise as to the proprietary rights to the intellectual property. If a dispute arises, a court may determine that the right belongs to a third party, and enforcement of our rights can be costly and unpredictable. In addition, we rely on trade secrets and proprietary know-how that we seek to protect in part by confidentiality agreements with our employees, contractors, consultants, advisors or others. Despite the protective measures we employ, we still face the risk that:
| · | these agreements may be breached; |
| · | these agreements may not provide adequate remedies for the applicable type of breach; or |
| · | our trade secrets or proprietary know-how will otherwise become known. |
Any breach of our confidentiality agreements or our failure to effectively enforce such agreements would have a material adverse effect on our business and competitive position.
If we infringe the rights of third parties we could be prevented from selling products, forced to pay damages and required to defend against litigation which could result in substantial costs and may have a material adverse effect on our business, results of operations and financial condition.
We have not received to date any claims of infringement by any third parties. However, as our drug candidates progress into clinical trials and commercialization, if at all, our public profile and that of our drug candidates may be raised and generate such claims. Defending against such claims, and occurrence of a judgment adverse to us, could result in unanticipated costs and may have a material adverse effect on our business and competitive position. If our products, methods, processes and other technologies infringe the proprietary rights of other parties, we may incur substantial costs and we may have to:
| · | obtain licenses, which may not be available on commercially reasonable terms, if at all; |
| · | redesign our products or processes to avoid infringement; |
| · | stop using the subject matter claimed in the patents held by others, which could cause us to lose the use of one or more of our drug candidates; |
| · | defend litigation or administrative proceedings that may be costly whether we win or lose, and which could result in a substantial diversion of management resources; or |
| · | pay damages. |
Any costs incurred in connection with such events or the inability to sell our products may have a material adverse effect on our business, results of operations and financial condition.
| 53 |
If we cannot meet requirements under our license agreements, we could lose the rights to our products, which could have a material adverse effect on our business.
We depend on licensing agreements with third parties to maintain the intellectual property rights to certain of our product candidates. Presently, we have licensed rights from the Yeda which allow us to use their technology and discoveries for the development, production and sale of enzymatically active mutations of GCD and derivatives thereof for the treatment of Gaucher disease. In addition, pursuant to our agreement with the Yissum Research and Development Company, or Yissum, the technology transfer arm of the Hebrew University of Jerusalem, Israel, and the Boyce Thompson Institute for Plant Research, at Cornell University, we have received an exclusive worldwide right and license to certain technology, including patents and additional patent applications relating to acetylcholinesterase (AChE), for all therapeutic and prophylactic indications as well as an exclusive license not limited to such indications with respect to certain of these patents and patent applications. Under the agreement with Yissum, we intend to develop a proprietary plant cell-based acetylcholinesterase (AChE) and its molecular variants for the use in several therapeutic and prophylactic indications, including a biodefense program. In addition, we have licensed certain rights relating to our production of taliglucerase alfa from Virginia Tech. Our license agreements require us to make payments and satisfy performance obligations in order to maintain our rights under these agreements. All of these agreements last either throughout the life of the patents that are the subject of the agreements, or with respect to other licensed technology, for a number of years after the first commercial sale of the relevant product.
In addition, we are responsible for the cost of filing and prosecuting certain patent applications and maintaining certain issued patents licensed to us. If we do not meet our obligations under our license agreements in a timely manner, we could lose the rights to our proprietary technology which could have a material adverse effect on our business, results of operations and financial condition.
Risks Relating to Our Operations in Israel
Potential political, economic and military instability in the State of Israel, where the majority of our senior management and our research and development facilities are located, may adversely affect our results of operations.
Our executive office and operations are located in the State of Israel. Accordingly, political, economic and military conditions in Israel directly affect our business. Since the State of Israel was established in 1948, a number of armed conflicts have occurred between Israel and its Arab neighbors. Any hostilities involving Israel or the interruption or curtailment of trade between Israel and its present trading partners, or a significant downturn in the economic or financial condition of Israel, could affect adversely our operations. Since October 2000 there have been increasing occurrences of terrorist violence. Ongoing and revived hostilities or other Israeli political or economic factors could harm our operations and product development and cause our revenues to decrease. Furthermore, several countries, principally those in the Middle East, still restrict business with Israel and Israeli companies. These restrictive laws and policies may limit seriously our ability to sell our products in these countries.
Although Israel has entered into various agreements with Egypt, Jordan and the Palestinian Authority, there have been times since October 2000 when Israel has experienced an increase in unrest and terrorist activity. The establishment in 2006 of a government in the Palestinian Authority by representatives of the Hamas militant group has created additional unrest and uncertainty in the region. In mid-2006, there was a war between Israel and the Hezbollah in Lebanon, resulting in thousands of rockets being fired from Lebanon up to 50 miles into Israel. Our current facilities are located in northern Israel, are in range of rockets that were fired from Lebanon into Israel during the war and suffered minimal damages during one of the rocket attacks. Our insurance policies do not cover us for these types of damages or for any resulting disruption in our operations. The Israeli government, as a matter of law, provides coverage for the reinstatement value of direct damages that are caused by terrorist attacks or acts of war; however, the government may cease providing such coverage or the coverage might not be enough to cover potential damages. If our facilities are damaged as a result of hostile action, our operations may be materially adversely affected.
In addition to the foregoing, since the end of 2010, numerous acts of protest and civil unrest have taken place in several countries in the Middle East and North Africa, many of which involved significant violence. The civil unrest in Egypt, which borders Israel, resulted in the resignation of its president Hosni Mubarak, and to significant changes to the country's government. In Syria, also bordering Israel, large civilian protests are continuing to take place. The ultimate effect of these developments on the political and security situation in the Middle East and on Israel's position within the region is not clear at this time.
| 54 |
Our operations may be disrupted by the obligations of our personnel to perform military service which could have a material adverse effect on our business.
Many of our male employees in Israel, including members of senior management, are obligated to perform up to one month (in some cases more) of annual military reserve duty until they reach the age of 45 and, in the event of a military conflict, could be called to active duty. Our operations could be disrupted by the absence of a significant number of our employees related to military service or the absence for extended periods of military service of one or more of our key employees. A disruption could have a material adverse effect on our business.
Because a certain portion of our expenses is incurred in New Israeli Shekels, or NIS, our results of operations may be seriously harmed by currency fluctuations and inflation.
We report our financial statements in U.S. dollars, our functional currency. Although most of our expenses are incurred in U.S. dollars, we pay a portion of our expenses in New Israeli Shekels, or NIS and, as a result, we are exposed to risk to the extent that the inflation rate in Israel exceeds the rate of devaluation of the NIS in relation to the U.S. dollar or if the timing of these devaluations lags behind inflation in Israel. In that event, the U.S. dollar cost of our operations in Israel will increase and our U.S. dollar-measured results of operations will be adversely affected. To the extent that the value of the NIS increases against the dollar, our expenses on a dollar cost basis increase. Our operations also could be adversely affected if we are unable to guard against currency fluctuations in the future. To date, we have not engaged in hedging transactions. In the future, we may enter into currency hedging transactions to decrease the risk of financial exposure from fluctuations in the exchange rate of the U.S. dollar against the NIS. These measures, however, may not adequately protect us from material adverse effects.
The tax benefits available to us require that we meet several conditions and may be terminated or reduced in the future, which would increase our taxes.
We are able to take advantage of tax exemptions and reductions resulting from the “Approved Enterprise” status of our facilities in Israel. To remain eligible for these tax benefits, we must continue to meet certain conditions, including making specified investments in property and equipment, and financing at least 30% of such investments with share capital. If we fail to meet these conditions in the future, the tax benefits would be canceled and we may be required to refund any tax benefits we already have enjoyed. These tax benefits are subject to investment policy by the Investment Center and may not be continued in the future at their current levels or at any level. In recent years the Israeli government has reduced the benefits available and has indicated that it may further reduce or eliminate some of these benefits in the future. The termination or reduction of these tax benefits or our inability to qualify for additional “Approved Enterprise” approvals may increase our tax expenses in the future, which would reduce our expected profits and adversely affect our business and results of operations. Additionally, if we increase our activities outside of Israel, for example, by future acquisitions, such increased activities generally may not be eligible for inclusion in Israeli tax benefit programs.
The Israeli government grants we have received for certain research and development expenditures restrict our ability to manufacture products and transfer technologies outside of Israel and require us to satisfy specified conditions. If we fail to satisfy these conditions, we may be required to refund grants previously received together with interest and penalties which could have a material adverse effect on our business and results of operations.
Our research and development efforts have been financed, in part, through grants that we have received from the OCS. We, therefore, must comply with the requirements of the Israeli Law for the Encouragement of Industrial Research and Development, 1984, and related regulations, or the Research Law.
Under the Research Law we are prohibited from manufacturing products developed using these grants outside of the State of Israel without special approvals, although the Research Law does enable companies to seek prior approval for conducting manufacturing activities outside of Israel without being subject to increased royalties. We may not receive the required approvals for any proposed transfer of manufacturing activities. Even if we do receive approval to manufacture products developed with government grants outside of Israel, we may be required to pay an increased total amount of royalties (possibly up to 300% of the grant amounts plus interest), depending on the manufacturing volume that is performed outside of Israel, as well as at a possibly increased royalty rate. This restriction may impair our ability to outsource manufacturing or engage in similar arrangements for those products or technologies.
| 55 |
Additionally, under the Research Law, we are prohibited from transferring the OCS-financed technologies and related intellectual property rights outside of the State of Israel, except under limited circumstances and only with the approval of the OCS' Research Committee. We may not receive the required approvals for any proposed transfer and, if received, we may be required to pay the OCS a portion of the consideration that we receive upon any sale of such technology by a non-Israeli entity. The scope of the support received, the royalties that we have already paid to the OCS, the amount of time that has elapsed between the date on which the know-how was transferred and the date on which the OCS grants were received and the sale price and the form of transaction will be taken into account in order to calculate the amount of the payment to the OCS. Approval of the transfer of technology to residents of the State of Israel is required, and may be granted in specific circumstances only if the recipient abides by the provisions of applicable laws, including the restrictions on the transfer of know-how and the obligation to pay royalties. No assurance can be made that approval to any such transfer, if requested, will be granted.
These restrictions may impair our ability to sell our technology assets or to outsource manufacturing outside of Israel. The restrictions will continue to apply for a certain period of time even after we have repaid the full amount of royalties payable for the grants. For the years ended December 31, 2010 and 2011, we recorded grants totaling $3.8 million and $3.3 million from the OCS, respectively. The grants represent 10% and 9%, respectively, of our gross research and development expenditures for the years ended December 31, 2010 and 2011. If we fail to satisfy the conditions of the Research Law, we may be required to refund certain grants previously received together with interest and penalties, and may become subject to criminal charges, any of which could have a material adverse effect on our business, results of operations and financial condition.
Investors may have difficulties enforcing a U.S. judgment, including judgments based upon the civil liability provisions of the U.S. federal securities laws against us, our executive officers and most of our directors or asserting U.S. securities laws claims in Israel.
Most of our directors and officers are not residents of the United States and most of their assets and our assets are located outside the United States. Service of process upon our non-U.S. resident directors and officers and enforcement of judgments obtained in the United States against us, some of our directors and executive officers may be difficult to obtain within the United States. We have been informed by our legal counsel in Israel that investors may find it difficult to assert claims under U.S. securities laws in original actions instituted in Israel or obtain a judgment based on the civil liability provisions of U.S. federal securities laws against us, our officers and our directors. Israeli courts may refuse to hear a claim based on a violation of U.S. securities laws against us or our officers and directors because Israel is not the most appropriate forum to bring such a claim. In addition, even if an Israeli court agrees to hear a claim, it may determine that Israeli law and not U.S. law is applicable to the claim. If U.S. law is found to be applicable, the content of applicable U.S. law must be proved as a fact which can be a time-consuming and costly process. Certain matters of procedure will also be governed by Israeli law. There is little binding case law in Israel addressing the matters described above.
Israeli courts might not enforce judgments rendered outside Israel which may make it difficult to collect on judgments rendered against us. Subject to certain time limitations, an Israeli court may declare a foreign civil judgment enforceable only if it finds that:
| · | the judgment was rendered by a court which was, according to the laws of the state of the court, competent to render the judgment; |
| · | the judgment may no longer be appealed; |
| · | the obligation imposed by the judgment is enforceable according to the rules relating to the enforceability of judgments in Israel and the substance of the judgment is not contrary to public policy; and |
| · | the judgment is executory in the state in which it was given. |
Even if these conditions are satisfied, an Israeli court will not enforce a foreign judgment if it was given in a state whose laws do not provide for the enforcement of judgments of Israeli courts (subject to exceptional cases) or if its enforcement is likely to prejudice the sovereignty or security of the State of Israel. An Israeli court also will not declare a foreign judgment enforceable if:
| · | the judgment was obtained by fraud; |
| · | there is a finding of lack of due process; |
| · | the judgment was rendered by a court not competent to render it according to the laws of private international law in Israel; |
| · | the judgment is at variance with another judgment that was given in the same matter between the same parties and that is still valid; or |
| · | at the time the action was brought in the foreign court, a suit in the same matter and between the same parties was pending before a court or tribunal in Israel. |
| 56 |
Risks Related to Investing in Our Common Stock
The market price of our common stock may fluctuate significantly.
The market price of our common stock may fluctuate significantly in response to numerous factors, some of which are beyond our control, such as:
| · | the results of our ongoing studies regarding our lead product candidate taliglucerase alfa, or communications from the FDA or other regulatory authorities regarding our NDA or other marketing authorization applications, including the approval of any such filings by the FDA or other applicable regulatory authorities, or any delay or failure by the FDA or other applicable regulatory authorities to approve any such filings; |
| · | developments concerning intellectual property rights and regulatory approvals; |
| · | the announcement of new products or product enhancements by us or our competitors; |
| · | variations in our and our competitors’ results of operations; |
| · | changes in earnings estimates or recommendations by securities analysts, if our common stock is covered by analysts; |
| · | developments in the biotechnology industry; and |
| · | general market conditions and other factors, including factors unrelated to our operating performance. |
Further, stock markets in general, and the market for biotechnology companies in particular, have recently experienced price and volume fluctuations. Continued market fluctuations could result in extreme volatility in the price of our common stock, which could cause a decline in the value of our common stock. Price volatility of our common stock may be worse if the trading volume of our common stock is low. We have not paid, and do not expect to pay, any cash dividends on our common stock as any earnings generated from future operations will be used to finance our operations. As a result, investors will not realize any income from an investment in our common stock until and unless their shares are sold at a profit.
Our common stock is listed for trade on more than one stock exchange, and this may result in price variations.
Our common stock is listed for trade on both the NYSE Amex and the TASE. Dual-listing may result in price variations between the exchanges due to a number of factors. First, our common stock is traded in U.S. dollars on the NYSE Amex and in NIS on the TASE. In addition, the exchanges are open for trade at different times of the day and on different days. For example, the TASE opens generally during Israeli business hours, Sunday through Thursday while the NYSE Amex opens generally during U.S. business hours, Monday through Thursday. The two exchanges also have differing vacation schedules. Differences in the trading schedules, as well as volatility in the exchange rate of the two currencies, among other factors, may result different trading prices for our common stock on the two exchanges. Other external influences may have different effects on the trading price of our common stock on the two exchanges.
Future sales of our common stock could reduce our stock price.
The market price of our common stock could drop significantly if our existing shareholders sell a large number of shares of our common stock or are perceived by the market as intending to sell them. All of the shares sold in our public offering in October 2007 were freely tradable without restriction or further registration under the federal securities laws, unless purchased by our “affiliates” as that term is defined in Rule 144 under the Securities Act. In addition, all of the outstanding shares of our common stock are freely tradable without restriction or further registration under the federal securities laws, unless owned by our affiliates. At December 31, 2011, there were options issued and outstanding to purchase 7,379,722 shares of our common stock with a weighted average exercise price of $3.87 per share. Also at December 31, 2011, there were 47,920 shares of common stock remaining available for future for issuance in connection with future grants of incentives under our 2006 Stock Incentive Plan. In addition, four of our executive officers have entered into trading plans established under Rule 10b5-1 under the Securities Act that allow for sales of approximately 1.3 million shares upon receipt of FDA approval of taliglucerase alfa, if at all.
Directors, executive officers, principal shareholders and affiliated entities own a significant percentage of our capital stock, and they may make decisions that an investor may not consider to be in the best interests of our shareholders.
Our directors, executive officers, principal shareholders and affiliated entities beneficially own, in the aggregate, approximately 44% of our outstanding common stock. As a result, if some or all of them acted together, they would have the ability to exert substantial influence over the election of our Board of Directors and the outcome of issues requiring approval by our shareholders. This concentration of ownership may have the effect of delaying or preventing a change in control of our company that may be favored by other shareholders. This could prevent the consummation of transactions favorable to other shareholders, such as a transaction in which shareholders might otherwise receive a premium for their shares over current market prices.
| 57 |
Failure to maintain effective internal controls in accordance with Section 404 of the Sarbanes-Oxley Act could have a material adverse effect on our business and operating results. In addition, current and potential shareholders could lose confidence in our financial reporting, which could have a material adverse effect on the price of our common stock.
Effective internal controls are necessary for us to provide reliable financial reports and effectively prevent fraud. If we cannot provide reliable financial reports or prevent fraud, our results of operation could be harmed.
Section 404 of the Sarbanes-Oxley Act of 2002 requires annual management assessments of the effectiveness of our internal controls over financial reporting and a report by our independent registered public accounting firm addressing these assessments. We continuously monitor our existing internal controls over financial reporting systems to confirm that they are compliant with Section 404, and we may identify deficiencies that we may not be able to remediate in time to meet the deadlines imposed by the Sarbanes-Oxley Act. This process may divert internal resources and will take a significant amount of time and effort to complete.
If, at any time, it is determined that we are not in compliance with Section 404, we may be required to implement new internal control procedures and reevaluate our financial reporting. We may experience higher than anticipated operating expenses as well as increased independent auditor fees during the implementation of these changes and thereafter. Further, we may need to hire additional qualified personnel. If we fail to maintain the adequacy of our internal controls, as such standards are modified, supplemented or amended from time to time, we may not be able to conclude on an ongoing basis that we have effective internal controls over financial reporting in accordance with Section 404 of the Sarbanes-Oxley Act, which could result in our being unable to obtain an unqualified report on internal controls from our independent auditors. Failure to maintain an effective internal control environment could also cause investors to lose confidence in our reported financial information, which could have a material adverse effect on the price of our common stock.
Compliance with changing regulation of corporate governance and public disclosure may result in additional expenses, divert management’s attention from operating our business which could have a material adverse effect on our business.
There have been other changing laws, regulations and standards relating to corporate governance and public disclosure in addition to the Sarbanes-Oxley Act, as well as new regulations promulgated by the Commission and rules promulgated by the national securities exchanges, including the NYSE Amex and the NASDAQ. These new or changed laws, regulations and standards are subject to varying interpretations in many cases due to their lack of specificity, and as a result, their application in practice may evolve over time as new guidance is provided by regulatory and governing bodies, which could result in continuing uncertainty regarding compliance matters and higher costs necessitated by ongoing revisions to disclosure and governance practices. As a result, our efforts to comply with evolving laws, regulations and standards are likely to continue to result in increased general and administrative expenses and a diversion of management time and attention from revenue-generating activities to compliance activities. Our board members, Chief Executive Officer and Chief Financial Officer could face an increased risk of personal liability in connection with the performance of their duties. As a result, we may have difficulty attracting and retaining qualified board members and executive officers, which could have a material adverse effect on our business. If our efforts to comply with new or changed laws, regulations and standards differ from the activities intended by regulatory or governing bodies, we may incur additional expenses to comply with standards set by regulatory authorities or governing bodies which would have a material adverse effect on our business, financial condition and results of operations.
The issuance of preferred stock or additional shares of common stock could adversely affect the rights of the holders of shares of our common stock.
Our Board of Directors is authorized to issue up to 100,000,000 shares of preferred stock without any further action on the part of our shareholders. Our Board of Directors has the authority to fix and determine the voting rights, rights of redemption and other rights and preferences of preferred stock. Currently, we have no shares of preferred stock outstanding.
| 58 |
Our Board of Directors may, at any time, authorize the issuance of a series of preferred stock that would grant to holders the preferred right to our assets upon liquidation, the right to receive dividend payments before dividends are distributed to the holders of common stock and the right to the redemption of the shares, together with a premium, before the redemption of our common stock, which may have a material adverse effect on the rights of the holders of our common stock. In addition, our Board of Directors, without further shareholder approval, may, at any time, issue large blocks of preferred stock. In addition, the ability of our Board of Directors to issue shares of preferred stock without any further action on the part of our shareholders may impede a takeover of our company and may prevent a transaction that is favorable to our shareholders.
Under the rules of the TASE, other than incentives under our 2006 Stock Option Plan, we were prohibited from issuing any securities of any class or series different than the common stock that is listed on the TASE for the 12-month period immediately succeeding our initial listing, which occurred on September 6, 2010. As of the date hereof, the rules of the TASE allow us to issue securities with preferential rights with respect to dividends but such other securities may not include voting rights. The foregoing does not limit our liability to issue and grant options and warrants for the purchase of shares of our common stock.
Item 1B. Unresolved Staff Comments
None.
Item 2. Properties
Our manufacturing facility and executive offices are located in Carmiel, Israel. The facilities currently contain approximately 16,000 sq/ft of manufacturing space and additional 48,000 sq/ft of laboratory, warehouse and office space and are leased at a rate of approximately $75,000 per month. In addition, we are entitled to use an additional 13,000 sq/ft in the same facility, which we intend to utilize in connection with an anticipated expansion of our manufacturing facilities. Our facilities are equipped with the requisite laboratory services required to conduct our business, and we believe that the existing facilities are adequate to meet our needs for the foreseeable future. We have leased the facility through 2017, subject to three options exercisable by us to extend the term for a five-year period, for an aggregate of 15 additional years. Upon the exercise of each option to extend the term of the lease, if any, the then current base rent shall be increased by 10%. We also lease an office in Ramat Gan, Israel, for approximately $2,200 per month.
Item 3. Legal Proceedings
We are not involved in any material legal proceedings.
Item 4. Mine Safety Disclosures
Not applicable.
| 59 |
PART II
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Our common stock began trading on the NYSE Amex on March 12, 2007 under the symbol “PLX.” Our common stock is also listed on the TASE under the symbol "PLX." The following table sets forth the quarterly high and low closing prices for our common stock on the NYSE Amex.
| Price Range | ||||||||
| High | Low | |||||||
| Fourth Quarter 2011 | $ | 6.57 | $ | 4.42 | ||||
| Third Quarter 2011 | $ | 6.99 | $ | 4.36 | ||||
| Second Quarter 2011 | $ | 7.24 | $ | 6.09 | ||||
| First Quarter 2011 | $ | 10.46 | $ | 5.83 | ||||
| Fourth Quarter 2010 | $ | 10.00 | $ | 8.40 | ||||
| Third Quarter 2010 | $ | 9.00 | $ | 5.99 | ||||
| Second Quarter 2010 | $ | 7.00 | $ | 5.84 | ||||
| First Quarter 2010 | $ | 7.70 | $ | 6.56 | ||||
These quotations reflect prices between dealers and do not include retained mark-ups, mark-downs and commissions and may not necessarily represent actual transactions.
There were approximately 46 holders of record of our common stock at February 1, 2012. A substantially greater number of holders of our common stock are “street name” or beneficial holders, whose shares are held of record by banks, brokers and other financial institutions. To date, we have not declared or paid any cash dividends on our common stock. We do not anticipate paying any dividends on our common stock in the foreseeable future.
STOCK PERFORMANCE GRAPH
The following graph compares the cumulative total shareholder return data for our common stock from December 31, 2006 through December 31, 2011 to the cumulative return over such time period of (i) The AMEX Composite Index and (ii) The AMEX Biotechnology Index. The graph assumes an investment of $100 on December 31, 2006 in each of our common stock, and the stocks comprising the AMEX Composite Index and the stocks comprising the AMEX Biotechnology Index, including dividend reinvestment, if any.
The stock price performance shown on the graph below represents historical price performance and is not necessarily indicative of any future stock price performance. The historical performance of our common stock prior to March 11, 2007, represents the performance of our common stock prior to its listing for trade on the NYSE AMEX. Notwithstanding anything to the contrary set forth in any of our previous filings under the Securities Act or the Exchange Act, which might incorporate future filings made by us under those statutes, this Stock Performance Graph will not be incorporated by reference into any of those prior filings, nor will such report or graph be incorporated by reference into any future filings made by us under those Acts.
| 60 |
| 61 |
Item 6. Selected Financial Data
The selected consolidated financial data below should be read in conjunction with “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and our consolidated financial statements and the related notes included elsewhere in this Annual Report on Form 10-K. The selected consolidated statements of operations data for the years ended December 31, 2011, 2010 and 2009 and the selected consolidated balance sheet data as of December 31, 2011 and 2010, are derived from the audited consolidated financial statements included elsewhere in this Annual Report. The statement of operations data for the years ended December 31, 2008 and 2007 and the balance sheet data as of December 31, 2009, 2008 and 2007 are derived from audited financial statements not included in this Annual Report. The historical results presented below are not necessarily indicative of future results.
Year Ended December 31, | ||||||||||||||||||||
| 2007 | 2008 | 2009 | 2010 | 2011 | ||||||||||||||||
| (in thousands, except share and per share amounts) | ||||||||||||||||||||
| Consolidated Statement of Operations Data: | ||||||||||||||||||||
| Revenues | - | - | $ | 388 | $ | 6,642 | $ | 8,386 | ||||||||||||
| Company’s share in collaboration agreement | - | - | - | 4,602 | (5,418 | ) | ||||||||||||||
| Cost of revenues | - | - | 3,575 | 4,383 | 1,525 | |||||||||||||||
| Gross profit (loss) | - | - | (3,187 | ) | 6,861 | 1,443 | ||||||||||||||
| Research and development expenses, net | $ | 13,570 | $ | 17,401 | 21,638 | 29,951 | 31,043 | |||||||||||||
| General and administrative expenses | 20,594 | 6,770 | 7,144 | 6,876 | 6,931 | |||||||||||||||
| Financial income, net | 2,080 | 1,757 | 529 | 968 | 2 | |||||||||||||||
| Net loss | $ | 32,084 | $ | 22,414 | $ | 31,440 | $ | 28,998 | $ | 36,529 | ||||||||||
| Net loss per share of common stock, basic and diluted | $ | 0.48 | $ | 0.30 | $ | 0.41 | $ | 0.36 | $ | 0.43 | ||||||||||
| Weighted average number of shares of common stock used in computing net loss per share of common stock | 67,187,329 | 75,890,633 | 76,942,840 | 80,960,300 | 84,645,364 | |||||||||||||||
| Consolidated Balance Sheet Data: | ||||||||||||||||||||
| Cash and cash equivalents | $ | 61,813 | $ | 42,596 | $ | 81,266 | $ | 35,900 | $ | 27,001 | ||||||||||
| Other assets | 6,324 | 8,215 | 17,405 | 28,829 | 24,804 | |||||||||||||||
| Total assets | 68,137 | 50,811 | 98,671 | 64,729 | 51,805 | |||||||||||||||
| Current liabilities | 3,762 | 5,527 | 21,530 | 18,903 | 18,693 | |||||||||||||||
| Total liabilities | 4,452 | 6,464 | 82,788 | 76,052 | 77,882 | |||||||||||||||
| Shareholders’ equity (net of capital deficiency) | 63,685 | 44,347 | 15,883 | (11,323 | ) | (26,077 | ) | |||||||||||||
| * | Represents less than $1. |
| 62 |
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
You should read the following discussion and analysis of our financial condition and results of operations together with our consolidated financial statements and the related notes included elsewhere in this Annual Report on Form 10-K. Some of the information contained in this discussion and analysis, particularly with respect to our plans and strategy for our business and related financing, includes forward-looking statements that involve risks and uncertainties. You should read “Risk Factors” in Item 1A of this Annual Report on Form 10-K for a discussion of important factors that could cause actual results to differ materially from the results described in or implied by the forward-looking statements contained in the following discussion and analysis.
Overview
We are a biopharmaceutical company focused on the development and commercialization of recombinant therapeutic proteins based on our proprietary ProCellEx® protein expression system. Using our ProCellEx system, we are developing a pipeline of proprietary and biosimilar or “generic” versions of recombinant therapeutic proteins based on our plant cell-based expression technology that target large, established pharmaceutical markets and that rely upon known biological mechanisms of action. Our initial commercial focus has been on complex therapeutic proteins, including proteins for the treatment of genetic disorders, such as Gaucher disease and Fabry disease. We believe our ProCellEx protein expression system will enable us to develop proprietary recombinant proteins that are therapeutically equivalent or superior to existing recombinant proteins currently marketed for the same indications. Because we are primarily targeting biologically equivalent versions of highly active, well-tolerated and commercially successful therapeutic proteins, we believe our development process is associated with relatively less risk compared to other biopharmaceutical development processes for completely novel therapeutic proteins.
Our lead product development candidate is taliglucerase alfa for the treatment of Gaucher disease, which we are developing using our ProCellEx protein expression system. The FDA has granted taliglucerase alfa a Prescription Drug User Fee Act (PDUFA) date of May 1, 2012. We first filed an NDA for taliglucerase alfa with the FDA on December 9, 2009, and on February 25, 2011, the FDA issued a CRL indicating that the FDA’s review was complete but questions remain that preclude the approval of the NDA in its then current form. The main questions raised by the FDA regarding the NDA related to the clinical and CMC sections. In the clinical section of the CRL, the FDA requested additional data from our ongoing switchover trial and our long-term extension trial. At the time the NDA was submitted, full data from these trials was not available. In the CMC section of the CRL, the FDA requested information regarding testing specifications and assay validation. The FDA did not request additional clinical studies in the CRL. We met with the FDA in May 2011 to clarify the FDA’s requests, and in July 2011 we submitted our reply to the CRL. In August 2011, the FDA notified us that it had accepted for review the resubmission of the taliglucerase alfa NDA, had deemed the resubmission a class 2, or 6-month, response and established February 1, 2012 as the new PDUFA date. In December 2011, the FDA notified us that it had decided to extend the PDUFA date to May 1, 2012. In its notification, the FDA stated that its decision related to certain clinical information regarding taliglucerase alfa we had submitted in November 2011 in response to an FDA request. The requested information related mainly to the presentation of select data provided in the NDA. As this information was requested and provided within 90 days of the February 1, 2012 PDUFA goal date, the FDA had the option to extend the PDUFA goal date to provide adequate time for the FDA to complete its review. A three-month extension cycle is the standard period granted. No additional data was requested by the FDA in the notification, nor were we notified of any specific deficiency in the taliglucerase alfa NDA.
In the fourth quarter of 2010, Pfizer, our commercialization partner, filed, with our cooperation, an MAA for taliglucerase alfa with the EMA. As part of its ongoing review of the MAA, the EMA delivered a list of outstanding points to be addressed by the applicant. Among the topics currently in discussion, is the orphan drug designation and exclusivity granted by the EMA/European Commission to VPRIV, which could prevent the marketing authorization of taliglucerase alfa in the European Union for a 10-year market exclusivity period commencing as of the August 2010 marketing authorization of VPRIV in the European Union. As part of the MAA procedure, Pfizer, with our cooperation, is challenging VPRIV's orphan market protection with respect to taliglucerase alfa pursuant to the EU orphan drug regulation. The EU orphan drug regulation provides for the possibility of such a challenge, and for an exception to this exclusivity to be granted, based on a number of factors, including contribution to patient care, clinical, supply, capacity and others.
Following the submission of the taliglucerase alfa MAA to the EMA, a good manufacturing practices, or GMP, audit of our manufacturing facility was performed and it was found compliant with EMA GMP in December 2011.
| 63 |
In January 2010, the EMA's Committee for Orphan Medicinal Products, or COMP, after reviewing all relevant clinical data, recommended that the European Commission grant orphan drug designation to taliglucerase alfa for the treatment of Gaucher disease and in March 2010, the European Commission granted orphan designation to taliglucerase alfa.
In addition, marketing applications have been submitted for taliglucerase alfa with each of the Israeli MOH, ANVISA and the Australian TGA.
In February 2010, the Israeli MOH completed a successful GMP audit of our manufacturing facilities in Carmiel, Israel. The audit was performed as part of the Israeli MOH’s evaluation of our manufacturing process for taliglucerase alfa. On February 20, 2011, we received a letter from the FDA notifying us that the FDA had completed its review of the Establishment Inspection Report in connection with the FDA’s inspection of our facility, and that the FDA had classified our facility as acceptable. In June 2011, ANVISA completed a successful GMP audit of the facility and determined that the facility is acceptable and in December 2011, the IMB completed a successful GMP audit of our facility and issued a Certificate of GMP Compliance of a Manufacturer for the facility. The IMB certificate is accepted by all health authorities in the European Union under the European Union's centralized marketing authorization procedure, and by authorities of several other countries that recognize EU certification.
In addition to the completed phase III clinical trial we completed in September 2009, we initiated a double-blind, follow-on extension study as part of the trial during the second quarter of 2008. Eligible patients who successfully completed nine months of treatment in our phase III clinical trial were offered the opportunity to participate in the extension study and continue to receive taliglucerase alfa at the same dose they received in the phase III clinical trial for an additional 15 months in a blinded manner. In February 2012, we announced data from 26 adult patients enrolled in the trial. According to the data, after 24 months, patients continued to demonstrate a significant improvement in all parameters with a similar safety profile as seen in the phase III clinical trial. Furthermore, those patients who were followed specifically for their bone parameters using Quantitative Chemical Shift Imaging (QCSI) MRI continued to show bone marrow improvement over time. We have also initiated a home care treatment program for patients enrolled in the extension study.
In the second quarter of 2011 we successfully completed a nine-month, worldwide, multi-center, open-label, switch-over clinical study evaluating the safety and efficacy of switching Gaucher patients from with Cerezyme to taliglucerase alfa. The results of the switchover trial demonstrate that over the nine-month treatment period, patients remained stable with regard to spleen volume, liver volume, platelet count and hemoglobin concentration, the efficacy endpoints of the switchover trial, after switching from Cerezyme to taliglucerase alfa. The safety analysis of the switchover trial demonstrated that taliglucerase alfa was well tolerated, and no drug related serious adverse events were reported. Patients in the extension trial and the switchover trial are still being treated with taliglucerase alfa.
In December 2009, we filed a proposed pediatric investigation plan to the Pediatric Committee of the EMA, which was approved during the first quarter of 2010. We have since initiated the pediatric study, completed enrollment of all the naïve pediatric patients required according to the study protocol and are in the final stages of pediatric patient enrollment.
On November 30, 2009, Protalix Ltd. and Pfizer entered into the Pfizer Agreement pursuant to which Pfizer was granted an exclusive, worldwide license to develop and commercialize taliglucerase alfa. Under the terms and conditions of the Pfizer Agreement, Protalix Ltd. retained the right to commercialize taliglucerase alfa in Israel. In connection with the execution of the Pfizer Agreement, Pfizer made an upfront payment to Protalix Ltd. of $60.0 million in connection with the execution of the agreement and subsequently paid Protalix Ltd. an additional $5.0 million upon its filing of a proposed pediatric investigation plan to the Pediatric Committee of the EMA. Protalix Ltd. is also eligible to receive potential milestone payments totaling $50.0 million for the successful achievement of other regulatory milestones. According to the agreement, Pfizer and Protalix Ltd. are required to share future revenues and expenses for the development and commercialization of taliglucerase alfa on a 60% and 40% basis, respectively, and have agreed to a specific allocation of the responsibilities for the continued development efforts for taliglucerase alfa.
Although taliglucerase alfa has not yet been approved for commercial sale, patients are being treated with taliglucerase alfa, both in the framework of our clinical trials and related studies and in compassionate use programs, special access agreements, named patient provisions and other programs designed to make sure that treatments are available to Gaucher patients in light of recent shortages of approved treatments. In July 2009, following a request by the FDA, we submitted a treatment protocol to the FDA in order to address an expected shortage of the current enzyme replacement therapy approved for Gaucher disease. The treatment protocol was approved by the FDA in August 2009, and we are continuing to treat patients in the United States under this protocol. On July 13, 2010, we announced that the French regulatory authority had granted an ATU for taliglucerase alfa for the treatment of Gaucher disease. An ATU is the regulatory mechanism used by the French Health Products and Safety Agency to make non-approved drugs available to patients in France when a genuine public health need exists. This ATU allows patients with Gaucher disease in France to receive treatment with taliglucerase alfa before marketing authorization for the product is granted in the European Union. Payment for taliglucerase alfa has been secured through government allocations to hospitals. In addition to the United States and France, taliglucerase alfa is currently being provided to Gaucher disease patients under special access agreements or Named Patient provisions in Brazil and in the rest of the world. Hundreds of patients, in the aggregate, have been treated with taliglucerase alfa.
| 64 |
On August 10, 2010, Pfizer entered into a $30 million short-term supply agreement with the Ministry of Health of Brazil pursuant to which Protalix and Pfizer have provided taliglucerase alfa to the Ministry of Health of Brazil for the treatment of patients with Gaucher disease. During the first quarter of 2011, we and Pfizer supplied the remaining products deliverable under the short-term supply agreement. Revenue generated from the Ministry of Health of Brazil was recorded by Pfizer, and we recorded our share of the revenue in accordance with the terms and conditions of the Pfizer Agreement. During the second and fourth quarters of 2011, the Ministry of Health of Brazil requested that Pfizer consider the replacement of certain vials that might expire during 2012. As a result, Pfizer recorded an allowance for sales return. If and when such vials are replaced, revenues will be recorded upon the supply of such replaced vials, and if it is determined that there is no longer a need for the replacement of vials, the allowance will be reversed and the revenues will be recognized accordingly. In addition, we and the Ministry of Health of Brazil are in discussions relating to a possible long-term supply agreement that contemplates, among other matters, providing certain components of our manufacturing technology to the Ministry of Health of Brazil for implementation by it in Brazil. We are currently unable to assess whether these discussions will result in an agreement and we can make no assurance that we will be able to enter into such an agreement on favorable terms, if at all. In any event, we do not expect to enter into a long-term supply agreement with the Ministry of Health of Brazil until we receive marketing approval of taliglucerase alfa from the FDA or ANVISA, if at all.
Although Gaucher disease is a relatively rare disease, it represents a large commercial market due to the severity of the symptoms and the chronic nature of the disease. According to public reports by Sanofi, consolidated sales of Cerezyme were €441.0 million (or $579.6 million), a growth of 11.1% compared to the same period in 2010. According to Genzyme, it has suffered interruptions and other limitations in its Cerezyme production and release since September 2009 and, as a result, Cerezyme supplies have been limited. Shire reported annual worldwide sales of VPRIV of approximately $256 million in 2011, a growth of 79% compared to the same period in 2010 (VPRIV was approved for sale in the United States in February 2010 and in the European Union in August 2010).
In addition to taliglucerase alfa, we are developing an innovative product pipeline using our ProCellEx protein expression system. Our product pipeline currently includes, among other candidates: (1) PRX-102, a therapeutic protein candidate for the treatment of Fabry disease, a rare, genetic lysosomal disorder in humans; (2) PRX-105, a plant cell expressed pegylated recombinant acetylcholinesterase product candidate for biodefense and other indications; (3) PRX-106, or pr-antiTNF, a plant cell expressed recombinant fusion protein made from the soluble form of the human TNF receptor (TNFR) and an antibody portion, which is being developed as a treatment of certain immune diseases such as rheumatoid arthritis, juvenile idiopathic arthritis, ankylosing, spondylitis, psoriatic arthritis and plaque psoriasis; (4) an orally administrated glucocerebrosidase enzyme for treating Gaucher disease patients utilizing the oral delivery of the recombinant enzyme produced within carrot cells; and (5) two additional undisclosed therapeutic proteins, both of which are being evaluated in animal studies. We participated in a pre IND meeting with respect to one of the undisclosed product candidates in the first quarter of 2012 and a pre IND meeting for the second candidate is planned for later this year. In March 2010, we initiated a preliminary phase I clinical trial of PRX-105 which we completed in June 2010. We are currently preparing for further efficacy trials of this product candidate in larger animals. In our preclinical studies we utilized an analogue to nerve gas. However, we anticipate that we will use live nerve gas rather than an analogue in the proposed additional efficacy trials in animals. In December 2010, we held a pre- IND meeting with the FDA with respect to PRX-102. We completed preclinical trials in small and large animals with respect to PRX-102, and we expect to submit an IND to the FDA during the second quarter of 2012 in connection with an anticipated phase I/II clinical study of PRX-102 in Fabry patients, and to initiate the trial once the IND is approved, if at all. In December 2011 we held a pre IND meeting with respect to PRX 106 and we expect to submit an IND around year end. We are also conducting pre clinical trials of our orally-administrated glucocerebrosidase enzyme for the treatment of Gaucher disease in small and large animals.
Except for the rights to commercialize taliglucerase alfa worldwide (other than Israel) which we licensed to Pfizer, we hold the worldwide commercialization rights to our proprietary development candidates, and we intend to establish an internal, commercial infrastructure and targeted sales force to market taliglucerase alfa in Israel and our other products, if approved, in North America, the European Union and in other significant markets, including Israel. In addition we are continuously evaluating potential strategic marketing partnerships.
| 65 |
In September 2009, we announced preliminary preclinical data regarding pr-antiTNF. Our pr-antiTNF product candidate has an amino acid sequence that is similar to Enbrel® which is one of the treatments for patients of those diseases. We believe that we may be able to reduce the development risks and time to market for our product candidates as our product candidates are based on well-understood proteins with known biological mechanisms of action. Based on our pre IND meeting with the FDA held in December 2011, we believe we may submit an IND around year end and, if approved, commence a phase I clinical trial shortly thereafter with no need for an external partner.
Our business is conducted by our wholly-owned subsidiary, Protalix Ltd., which we acquired through a reverse merger transaction effective December 31, 2006. The merger transaction was treated as a recapitalization for accounting purposes and, as such, the results of operations discussed below are those of Protalix Ltd. Prior to the merger transaction, we had not conducted any operations for several years. Protalix Ltd. was originally incorporated in Israel in December 1993. Since its inception in December 1993, Protalix Ltd. has generated significant losses in connection with its research and development, including the clinical development of taliglucerase alfa. Since we do not generate significant revenue from any of our product candidates, we expect to continue to generate losses in connection with the continued clinical development of taliglucerase alfa and the research and development activities relating to our technology and other drug candidates. We except that our expenditures in connection with the research and development activities will continue to represent a significant portion of our expenses for the next several years, and will require further financial resources if the activities are successful. As a result, we believe that our operating losses may continue to be substantial over the next several years. However, if taliglucerase alfa is launched successfully, we anticipate that we will generate revenues to offset any such losses. We may need to obtain additional funds for the commercialization of our lead product, taliglucerase alfa, and to further develop the research and clinical development of our other programs.
Critical Accounting Policies
Our significant accounting policies are more fully described in Note 1 to our consolidated financial statements appearing at the end of this Annual Report on Form 10-K. We believe that the accounting policies below are critical for one to fully understand and evaluate our financial condition and results of operations.
The discussion and analysis of our financial condition and results of operations is based on our financial statements, which we prepared in accordance with U.S. generally accepted accounting principles. The preparation of these financial statements requires us to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements, as well as the reported revenues and expenses during the reporting periods. On an ongoing basis, we evaluate such estimates and judgments, including those described in greater detail below. We base our estimates on historical experience and on various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions.
Functional Currency
The currency of the primary economic environment in which our operations are conducted is the U.S. dollar. As we have no significant source of revenues, we considered the currency of the primary economic environment to be the currency in which we expend cash. Most of our expenses and capital expenditures are incurred in dollars, and a significant source of our financing has been provided in U.S. dollars.
Revenues
Currently, our sole source of revenues comes from sales of taliglucerase alfa pursuant to our license agreement with Pfizer. We recognize revenue when the earnings process is complete, which is when revenue is realized or realizable and earned, there is persuasive evidence a revenue arrangement exists, delivery of goods or services has occurred, the sales price is fixed or determinable and collectability is reasonably assured.
We recognize revenue and milestone payments received pursuant to the Pfizer Agreement in accordance with guidance regarding revenue recognition and accounting for revenue arrangements with multiple deliverables. Pursuant to this guidance, we determine whether our arrangement with Pfizer involves multiple revenue-generating deliverables that should be accounted for as a combined unit of accounting or separate units of accounting for revenue recognition purposes. If we determine that there are multiple units of accounting, the consideration from the arrangement is allocated among the separate units based on a relative fair value allocation. If the arrangement represents a single unit of accounting, the revenue is recognized over the performance obligation period. As the arrangement with Pfizer requires our continued involvement with respect to the proposed commercialization of taliglucerase alfa, the non-refundable, up-front license payments we received from Pfizer are deferred and recognized over the related performance period. We estimated the performance period of 14 years based on the date that the last relevant patent relating to taliglucerase alfa expires.
| 66 |
Under the terms and conditions of the Pfizer Agreement, we are entitled to 40% of the net profits or loss from sales of taliglucerase alfa by Pfizer and reimbursement of our certain related expenses we incur in connection with Pfizer's sales. We retained all rights to taliglucerase alfa in Israel. We recognize our share of net profit or loss under the Pfizer Agreement based on reports we receive from Pfizer summarizing the results of the collaborative activities under the agreement for the applicable period. Under the terms of the Pfizer Agreement, for its subsidiaries operating outside the United States, financial information is included based on the fiscal year ending November 30, while financial information for the U.S. entity is included based on the fiscal year ending December 31.
We recognize revenues received from the sale of a product to Pfizer upon delivery to Pfizer. The revenues represent our cost with respect to the product sold.
Research and Development Expense
We expect our research and development expense to remain our primary expense in the near future as we continue to develop our product candidates. Research and development expense consists of:
| · | internal costs associated with research and development activities; | |
| · | payments made to third party contract research organizations, investigative sites and consultants; | |
| · | manufacturing development costs; | |
| · | personnel-related expenses, including salaries, benefits, travel, and related costs for the personnel involved in research and development; | |
| · | activities relating to the advancement of product candidates through preclinical studies and clinical trials; and | |
| · | facilities and other allocated expenses, which include direct and allocated expenses for rent and maintenance of facilities, as well as laboratory and other supplies. |
The following table identifies our current major research and development projects:
| Project | Status | Expected Near Term Milestones | ||
| taliglucerase alfa for the treatment of Gaucher disease |
NDA and MAA (EMA, ANVISA and the Israeli MOH) filed
|
PDUFA date of May 1, 2012 from the FDA; respond to EMA query | ||
| PRX 102 – alpha-GAL-A | Preclinical | File IND and initiate phase I/II clinical trials | ||
Acetylcholinesterase |
Phase I clinical trial |
Additional phase I clinical trials
| ||
| pr-antiTNF | Research | Complete pre clinical development; file IND and initiate phase I clinical trials |
All of our projects, other than taliglucerase alfa and our acetylcholinesterase product, are in the preclinical or research phase with relatively immaterial costs. Most of our research and development costs were incurred in connection with our phase III clinical trial of taliglucerase alfa. Our internal resources, employees and infrastructure are not tied to any individual research project and are typically deployed across all of our projects. We currently do not record and maintain research and development costs per project.
The costs and expenses of our projects are partially funded by grants we have received from the OCS. Each grant is deducted from the related research and development expenses as the costs are incurred. For additional information regarding the grant process, see “Business—Israeli Government Programs—Encouragement of Industrial Research and Development Law, 1984” in Item 1 of this Annual Report. There can be no assurance that we will continue to receive grants from the OCS in amounts sufficient for our operations, if at all.
| 67 |
At this time, due to the inherently unpredictable nature of preclinical and clinical development processes and given the early stage of our preclinical product development programs, we are unable to estimate with any certainty the costs we will incur in the continued development of the product candidates in our pipeline for potential commercialization. Clinical development timelines, the probability of success and development costs can differ materially from expectations. While we are currently focused on advancing each of our product development programs, our future research and development expenses will depend on the clinical success of each product candidate, as well as ongoing assessments of each product candidate’s commercial potential. In addition, we cannot forecast with any degree of certainty which product candidates may be subject to future collaborations, when such arrangements will be secured, if at all, and to what degree such arrangements would affect our development plans and capital requirements. See “Risk Factors—All of our product candidates other than taliglucerase alfa and our acetylcholinesterase product are in pre-clinical or research stages. If we are unable to develop and commercialize our product candidates, our business will be adversely affected” and “—We may not obtain the necessary U.S., EMA or other worldwide regulatory approvals to commercialize our drug candidates in a timely manner, if at all, which would have a material adverse effect on our business, financial condition and results of operations.”
We expect our research and development expenses to continue to be our primary expense in the future as we continue the advancement of our clinical trials and preclinical product development programs for our product candidates other than taliglucerase alfa. The lengthy process of completing clinical trials and seeking regulatory approval for our product candidates requires expenditure of substantial resources. Any failure or delay in completing clinical trials, or in obtaining regulatory approvals, could cause a delay in generating product revenue and cause our research and development expense to increase and, in turn, have a material adverse effect on our operations. We filed a revised NDA for taliglucerase alfa with the FDA in the third quarter of 2011 and currently have a PDUFA date of May 1, 2012. In addition, during 2010, we submitted a marketing application regarding taliglucerase alfa to the Israeli MOH and similar applications have been submitted to each of the EMA, ANVISA and the Australian TGA. Due to the factors set forth above, we are not able to estimate with any certainty when we would recognize any net cash inflows from our projects. See “Risk Factors—Clinical trials are very expensive, time-consuming and difficult to design and implement and may result in unforeseen costs which may have a material adverse effect on our business and results of operations.”
Share-Based Compensation
The discussion below regarding share-based compensation relates to share-based compensation paid by Protalix Ltd., our wholly-owned subsidiary.
In accordance with the guidance, we record the benefit of any grant to a non-employee and remeasure the benefit in any future vesting period for the unvested portion of the grants, as applicable. In addition, we use the straight-line accounting method for recording the benefit of the entire grant, unlike the graded method we use to record grants made to employees.
We measure share-based compensation cost for all share-based awards at the fair value on the grant date and recognition of share-based compensation over the service period for awards that we expect will vest. The fair value of stock options is determined based on the number of shares granted and the price of our ordinary shares, and calculated based on the Black-Scholes valuation model. We recognize such value as expense over the service period, net of estimated forfeitures, using the accelerated method.
For purposes of determining the fair value of the options to purchase common stock granted to employees and non-employees during the fiscal year ended December 31, 2011, including shares held by non employees that vested during such period, our management used the fair value of our common stock which was the closing sale price of our common stock on the NYSE Amex on the date of calculation.
The guidance requires companies to estimate the expected term of the option rather than simply using the contractual term of an option. Because of lack of data on past option exercises by employees, the expected term of the options could not be based on historic exercise patterns. Accordingly, we adopted the simplified method, according to which companies may calculate the expected term as the average between the vesting date and the expiration date, assuming the option was granted as a “plain vanilla” option.
In performing the valuation, we assumed an expected 0% dividend yield in the previous years and in the next years. We do not have a dividend policy and given the lack of profitability, dividends are not expected in the foreseeable future, if at all. The guidance stipulates a number of factors that should be considered when estimating the expected volatility, including the implied volatility of traded options, historical volatility and the period that the shares of the company are being publicly traded. As we did not have any traded shares or options for periods before December 31, 2006, the expected volatility figures used in this valuation have been calculated utilizing a blended approach by using the historical volatility of traded shares of similar companies for periods prior to December 31, 2006 and our own traded shares for periods subsequent to December 31, 2006. In addition, we examined the standard deviation of shares of similar biotechnology companies that engage in research and development, generally with no significant revenues. We found that the volatility of our shares and the shares of comparable companies was in the range of 40%-75% over periods of three to six years. The volatility used for each grant differed based on its expected term. For the term of each grant of our options, the historical volatility was calculated based upon the overall trading history of the common stock of comparable companies.
| 68 |
The risk-free interest rate described above has been based on the implied yield of U.S. federal reserve zero–coupon government bonds. The remaining term of the bonds used for each valuation was equal to the expected term of the grant. This methodology has been applied to all grants valued by us. The guidance requires the use of a risk–free interest rate based on the implied yield currently available on zero–coupon government issues of the country in whose currency the exercise price is expressed, with a remaining term equal to the expected life of the option being valued. This requirement has been applied for all grants valued as part of this report.
Year Ended December 31, 2011 Compared to the Year Ended December 31, 2010
Revenues
We recorded revenue of $8.4 million for the year ended December 31, 2011, an increase of $1.8 million or 27%, compared to revenue of $6.6 million for the year ended December 31, 2010. The increase resulted primarily from increases in the supply of taliglucerase alfa vials to Pfizer during the year ended December 31, 2011. Revenues also include the amortization of the $65 million upfront and milestone payments received from Pfizer. The payments were recorded as deferred revenue and the amounts will be amortized over the performance period, estimated at approximately 14 years, at a rate of approximately $1.1 million per quarter.
Our share in the Collaboration Agreement
We recorded $5.4 million of loss as our share in the collaboration under the Pfizer Agreement during the year ended December 31, 2011, a decrease of $10.0 million, or 217%, compared to the $4.6 million of income recorded for the year ended December 31, 2010. Our share in the collaboration's loss for the year ended December 31, 2011 resulted primarily from our 40% share of the allowance for sales return of taliglucerase alfa recorded by Pfizer in connection with the short term supply agreement between Pfizer and the Ministry of Health of Brazil. During 2011, the Ministry of Health of Brazil requested that Pfizer consider the replacement of certain vials that might expire during 2012. As a result, Pfizer recorded an allowance for sales return. The allowance was partially offset by our share in the income generated through sale of taliglucerase alfa via the ATU program in France. Under the terms and conditions of the Pfizer Agreement, we record income or loss equal to 40% of the profit or loss realized from sales of taliglucerase alfa and related expenses incurred based on reports we receive from Pfizer summarizing the results of the collaborative activities under the Pfizer Agreement for the applicable period. Under the terms and conditions of the Pfizer Agreement, financial information of Pfizer’s subsidiaries that operate outside the United States is included based on the fiscal year ending November 30, while financial information for the U.S. entity is included based on the fiscal year ending December 31.
Research and Development Expenses
Research and development expenses were $37.8 million for the year ended December 31, 2011, an increase of $127,000 from $37.7 million for the year ended December 31, 2010. The research and development expenses for the year ended December 31, 2011 were partially offset by grants received from the OCS of approximately $3.3 million, and $3.4 million of reimbursements for certain expenses in accordance with the terms and conditions of the Pfizer Agreement.
We expect research and development expenses to continue to be our primary expense as we enter into a more advanced stage of preclinical and clinical trials for certain of our product candidates.
General and Administrative Expenses
General and administrative expenses were $6.9 million for the year ended December 31, 2011, an increase $55,000 from $6.9 million for the year ended December 31, 2010. We expect that our general and administrative expenses will increase as we become a commercial company.
Financial Expenses and Income
Financial income was $2,000 for the year ended December 31, 2011, a decrease of $966,000, or 99%, from $968,000 for the year ended December 31, 2010. The decrease resulted primarily from the devaluation of the New Israeli Shekel against the U.S. dollar and lower interest rates available for short term deposits during the year ended December 31, 2011.
| 69 |
Year Ended December 31, 2010 Compared to the Year Ended December 31, 2009
Revenues
We recorded revenue of $6.6 million for the year ended December 31, 2010 compared to revenue of $388,000 for the year ended December 31, 2009. The increase resulted primarily from the amortization of the $65.0 million of upfront and milestone payments received from Pfizer over the entire fiscal year. Such payments were amortized over one month for the year ended December 31, 2009. The payments were recorded as deferred revenue and the amounts will be amortized over the performance period, estimated at approximately 14 years, at a rate of approximately $1.1 million per quarter. The amortization during the year ended December 31, 2010 resulted in the recognition of revenue of $4.6 million, or an increase of $4.2 million, from $388,000 recognized during the year ended December 31, 2009. In addition, we recorded revenues in the amount of approximately $2.0 million in connection with products delivered to Pfizer under our license agreement. No products were sold during 2009.
Our share in the Collaboration Agreement
We recorded $4.6 million of income as our share in the collaboration under the Pfizer Agreement during the year ended December 31, 2010. This income resulted primarily from our share of the approximately $21.8 million of revenues generated by Pfizer’s sale of taliglucerase alfa to the Ministry of Health of Brazil under the short term supply agreement during Pfizer's 2010 fiscal year. No share in the collaboration was recorded for the year ended December 31, 2009. Under the terms and conditions of the Pfizer Agreement, we recorded revenue equal to 40% of the profits realized from sales of taliglucerase alfa, and related expenses incurred based on reports we received from Pfizer summarizing the results of the collaborative activities under the agreement for the applicable period. Under the terms of the Pfizer Agreement, for its subsidiaries operating outside the United States, financial information is included based on the fiscal year ending November 30, while financial information for the U.S. entity is included based on the fiscal year ending December 31.
Research and Development Expenses
Research and development expenses were $37.7 million for the year ended December 31, 2010, an increase of $10.3 million, or 38%, from $27.4 million for the year ended December 31, 2009. The increase resulted primarily from an increase of $2.7 million in development expenses related to salaries for personnel involved in research and development, and $4.1 million in related subcontractors and consultants expenses, mainly in connection with our expanded access program for taliglucerase alfa. The increase in research and development expenses was partially offset by the recognition of grants from the OCS and Pfizer’s reimbursement of us, collectively equal to $7.7 million. The offsets to our research and development expenses for the year ended December 31, 2010 represent an increase of approximately $2.0 million, or 35%, compared to the recognition of grants from the OCS equal to $5.7 million that offset our research and development expenses for the year ended December 31, 2009.
General and Administrative Expenses
General and administrative expenses were $6.9 million for the year ended December 31, 2010, a decrease of $268,000, or approximately 4%, from $7.1 million for the year ended December 31, 2009. The decrease resulted primarily from a $389,000 decrease in salaries expense during 2010. We expect that our general and administrative expenses will increase as we add additional personnel and continue to comply with the reporting and other obligations applicable to public companies in the United States.
Financial Expenses and Income
Financial income was $968,000 for the year ended December 31, 2010, an increase of $439,000, or approximately 83%, compared to $529,000 for the year ended December 31, 2009. The increase resulted primarily from increases in the exchange rate that resulted in income of approximately $546,000.
| 70 |
Liquidity and Capital Resources
Sources of Liquidity
As a result of our significant research and development expenditures and the lack of any approved products to generate significant product sales revenue, we have not been profitable and have generated operating losses since our inception. To date, we have funded our operations primarily with proceeds equal to $31.3 million from the sale of convertible preferred and ordinary shares of Protalix Ltd., and an additional $14.1 million in connection with the exercise of warrants issued in connection with the sale of such shares, through December 31, 2008. In addition, on October 25, 2007, we generated gross proceeds of $50 million in connection with an underwritten public offering of our common stock and on each of March 23, 2011 and February 22, 2012, we generated gross proceeds of $22.0 million and $27.2 million, respectively, in connection with underwritten public offerings of our common stock. We believe that the funds currently available to us as are sufficient to satisfy our capital needs for at least 12 months.
The following table summarizes our past funding sources:
| Security | Year | Number of Shares | Amount(1) | |||||||||
| Ordinary Shares | 1996-2000 | 18,801,527 | (2) | $ | 1,100,000 | |||||||
| Series A Convertible Preferred Shares | 2001 | 11,635,090 | $ | 2,000,000 | ||||||||
| Series B Convertible Preferred Shares(3) | 2004-2005 | 7,175,621 | $ | 4,500,000 | ||||||||
| Series C Convertible Preferred Shares(4) | 2005 | 5,513,422 | $ | 7,700,000 | ||||||||
| Ordinary Shares(5) | 2006 | 10,637,880 | $ | 16,000,000 | ||||||||
| Common Stock | 2007 | 10,000,000 | $ | 50,000,000 | ||||||||
| Common Stock | 2011 | 4,000,000 | $ | 22,000,000 | ||||||||
| Common Stock | 2012 | 5,175,000 | $ | 27,200,000 | ||||||||
| (1) | Gross proceeds; does not include proceeds from warrant exercises. |
| (2) | Includes the issuance of ordinary shares to founders. |
| (3) | During 2005, 1,035,569 Series B Preferred Shares were converted on a 1:1 basis into Series C Preferred Shares for no additional consideration. Also, in connection with such funding, warrants to purchase 181,228 Series B Preferred Shares were issued for no additional consideration with an aggregate exercise price of $103,000. As of the closing date of the merger, 168,034 of such warrants were exercised for net proceeds equal to approximately $96,000 and 13,194 of such warrants were forfeited. |
| (4) | In connection with such funding, warrants to purchase an additional 8,862,803 Series C Preferred Shares were granted to the investors for no additional consideration with a total exercise price equal to $9.0 million. As of the closing date of the merger, 5,296,279 of such warrants were exercised for net proceeds equal to $8.7 million, 3,384,502 were assumed by our company and 182,022 expired. |
| (5) | In connection with such funding, warrants to purchase 3,875,416 ordinary shares were issued for no additional consideration with an aggregate exercise price equal to $5.3 million. These warrants were exercised in full on January 31, 2007. |
In addition to the foregoing, Pfizer paid Protalix Ltd. $60.0 million as an upfront payment in connection with the execution of the Pfizer Agreement and subsequently paid to Protalix Ltd. an additional $5.0 million upon Protalix Ltd.'s meeting a certain milestone. Protalix Ltd. is also eligible to receive potential milestone payments of up to $50.0 million for the successful achievement of other regulatory-related milestones. Protalix Ltd. is also entitled to payments equal to 40% of the net profits earned by Pfizer on its sales of taliglucerase alfa. In calculating net profits there are certain agreed upon limits on the amounts that may be deducted from gross sales for certain expenses and costs of goods sold.
Cash Flows
Net cash used in operations was $23.6 million for the year ended December 31, 2011. The net loss for 2011 of $36.5 million was further increased by a decrease in deferred revenues of approximately $3.0 million. The net loss was partially offset by non-cash charges for share-based compensation of $886,000, depreciation of $3.6 million and a decrease in accounts receivable of approximately $3.9 million and an increase in accounts payable of $6.1 million. Net cash used in investing activities for 2011 was $5.9 million and consisted primarily of purchases of property and equipment. Net cash provided from financing activities during 2011 was approximately $20.9 million primarily due to our underwritten public offering of common stock in the first quarter of 2011.
| 71 |
Net cash used in operations was $38.5 million for the year ended December 31, 2010. The net loss for 2010 of $29.0 million was further increased by an increase in accounts receivable of approximately $6.9 million and a decrease of $4.6 million in deferred revenues. The net loss was partially offset by non-cash charges for share-based compensation of $1.3 million, and depreciation of $3.1 million. Net cash used in investing activities for 2010 was $8.0 million and consisted primarily of purchases of property and equipment. Net cash provided from financing activities during 2010 was approximately $501,000 due to option exercises.
Future Funding Requirements
We expect that our operating losses may continue to be substantial over the next several years. However, we anticipate that we will generate revenues to offset any such losses upon the successful launch of taliglucerase alfa, if at all. We expect to incur significant research and development expenses, including expenses related to the hiring of personnel and the advancement of our additional pipeline of product candidate into the various clinical trials. We expect that general and administrative expenses will increase as we expand our finance and administrative staff, add infrastructure, and incur additional costs related to our preparation for the commercial phase for our lead product candidate, taliglucerase alfa. In addition, we are working on the expansion of our manufacturing facility that would meet approximately half of the market for our lead product candidate, which would increase our capital expenditures significantly and is estimated to cost approximately $25.0 million in total.
We believe that our existing cash and cash equivalents and the net proceeds from our public underwritten offering of February 2012 will be sufficient to enable us to fund our operating expenses and capital expenditure requirements at least for the next 12 months. We have based this estimate on assumptions that are subject to change and may prove to be wrong, and we may be required to use our available capital resources sooner than we currently expect. Because of the numerous risks and uncertainties associated with the development and commercialization of our product candidates, we are unable to estimate the amounts of increased capital outlays and operating expenditures associated with our current and anticipated clinical trials.
Our future capital requirements will depend on many factors, including the progress and results of our clinical trials, costs of commercialization activities, including product marketing, sales and distribution and whether these efforts will be performed internally or through some form of collaboration with third parties, the duration and cost of discovery and preclinical development, and laboratory testing and clinical trials for our product candidates, the timing and outcome of regulatory review of our product candidates, the costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing patent claims and other intellectual property rights and the number and development requirements of other product candidates that we pursue.
We may need to finance our future cash needs through public or private equity offerings, debt financings, or additional corporate collaboration and licensing arrangements. We currently do not have any commitments for future external funding, other than the potential regulatory-related milestone payments from Pfizer. We may need to raise additional funds more quickly if one or more of our assumptions prove to be incorrect or if we choose to expand our product development efforts more rapidly than we presently anticipate. We may also decide to raise additional funds even before we need them if the conditions for raising capital are favorable. Any sale of additional equity or debt securities will likely result in dilution to our shareholders. The incurrence of indebtedness would result in increased fixed obligations and could also result in covenants that would restrict our operations. Additional equity or debt financing, grants or corporate collaboration and licensing arrangements may not be available on acceptable terms, if at all. If adequate funds are not available, we may be required to delay, reduce the scope of or eliminate our research and development programs, reduce our planned commercialization efforts or obtain funds through arrangements with collaborators or others that may require us to relinquish rights to certain product candidates that we might otherwise seek to develop or commercialize independently.
Effects of Inflation and Currency Fluctuations
Inflation generally affects us by increasing our cost of labor and clinical trial costs. We do not believe that inflation has had a material effect on our results of operations during the years ended December 31, 2009, 2010 or 2011.
Currency fluctuations could affect us by increased or decreased costs mainly for goods and services acquired outside of Israel. We do not believe currency fluctuations have had a material effect on our results of operations during the years ended December 31, 2009, 2010 or 2011.
| 72 |
Off-Balance Sheet Arrangements
We have no off-balance sheet arrangements as of December 31, 2010 and 2011.
Recently Issued Accounting Pronouncements
None.
Contractual Obligations
The following table summarizes our significant contractual obligations at December 31, 2011 (in thousands):
| Total | Less than 1 year | 1-3 years | 3-5 years | More than 5 years | ||||||||||||||||
| Operating lease obligations | $ | 5,046 | $ | 1,415 | $ | 2,110 | $ | 1,521 | - | |||||||||||
| Purchase obligations (1) | $ | 2,758 | $ | 2,758 | - | - | - | |||||||||||||
| Certain clinical contract and building contractor obligations | $ | 821 | $ | 657 | $ | 164 | - | - | ||||||||||||
| Liability for employee rights upon retirement | $ | 1,700 | - | - | - | $ | 1,700 | |||||||||||||
| Total | $ | 10,325 | $ | 4,830 | $ | 2,274 | $ | 1,521 | $ | 1,700 | ||||||||||
(1) Represents open purchase orders issued to certain suppliers and other vendors mainly in connection with our research and development activities that were outstanding as of December 31, 2011.
The foregoing table does not include (i) annual license fees, which are currently immaterial, (ii) payments we may be required to make to certain of our licensors in the time periods set forth above upon the achievement of agreed-upon milestones and (iii) royalty payments payable by us to certain of our licensors in connection with the commercial sale of our product candidates, if any. If all of the contingencies with respect to milestone payments under our research and development agreements are met, the aggregate milestone payments payable would be approximately $950,000 and would be payable, if at all, as our projects progress over the course of a number of years. The royalty payments payable in connection with sales of each of our product candidates, if at all, shall not exceed low, single-digit percentages of net sales of the product.
Selected Quarterly Financial Data (unaudited)
| Three Months Ended | ||||||||||||||||||||||||||||||||
| 2010 | 2011 | |||||||||||||||||||||||||||||||
| (U.S. dollars in thousands) | ||||||||||||||||||||||||||||||||
| March 31 | June 30 | Sept. 30 | Dec. 31 | March 31 | June 30 | Sept. 30 | Dec. 31 | |||||||||||||||||||||||||
| Revenues | $ | 1,141 | $ | 1,141 | $ | 3,184 | $ | 1,176 | $ | 4,128 | $ | 764 | $ | 1,132 | $ | 2,362 | ||||||||||||||||
| Net loss | $ | 7,955 | $ | 10,041 | $ | 2,474 | $ | 8,528 | $ | 5,052 | $ | 13,689 | $ | 8,875 | $ | 8,913 | ||||||||||||||||
| Net loss per share of common stock, basic and diluted | $ | 0.10 | $ | 0.12 | $ | 0.03 | $ | 0.11 | $ | 0.06 | $ | 0.16 | $ | 0.10 | $ | 0.10 | ||||||||||||||||
Item 7A. Quantitative and Qualitative Disclosures About Market Risk
Currency Exchange Risk
The currency of the primary economic environment in which our operations are conducted is the dollar. We are currently have no significant source of revenues; therefore we consider the currency of the primary economic environment to be the currency in which we expend cash. Approximately 50% of our expenses and capital expenditures are incurred in dollars, and a significant source of our financing has been provided in U.S. dollars. Since the dollar is the functional currency, monetary items maintained in currencies other than the dollar are remeasured using the rate of exchange in effect at the balance sheet dates and non-monetary items are remeasured at historical exchange rates. Revenue and expense items are remeasured at the average rate of exchange in effect during the period in which they occur. Foreign currency translation gains or losses are recognized in the statement of operations.
Approximately 35% of our costs, including salaries, expenses and office expenses, are incurred in NIS. Inflation in Israel may have the effect of increasing the U.S. dollar cost of our operations in Israel. If the U.S. dollar declines in value in relation to the NIS, it will become more expensive for us to fund our operations in Israel. A revaluation of 1% of the NIS will affect our income before tax by less than 1%. The exchange rate of the U.S. dollar to the NIS, based on exchange rates published by the Bank of Israel, was as follows:
| 73 |
| Year Ended December 31, | ||||||||||||
| 2009 | 2010 | 2011 | ||||||||||
| Average rate for period | 3.933 | 3.733 | 3.578 | |||||||||
| Rate at year-end | 3.775 | 3.549 | 3.821 | |||||||||
To date, we have not engaged in hedging transactions. In the future, we may enter into currency hedging transactions to decrease the risk of financial exposure from fluctuations in the exchange rate of the U.S. dollar against the NIS. These measures, however, may not adequately protect us from material adverse effects due to the impact of inflation in Israel.
Interest Rate Risk
Our exposure to market risk is confined to our cash and cash equivalents. We consider all short term, highly liquid investments, which include short-term deposits with original maturities of three months or less from the date of purchase, that are not restricted as to withdrawal or use and are readily convertible to known amounts of cash, to be cash equivalents. The primary objective of our investment activities is to preserve principal while maximizing the interest income we receive from our investments, without increasing risk. We invest any cash balances primarily in bank deposits and investment grade interest-bearing instruments. We are exposed to market risks resulting from changes in interest rates. We do not use derivative financial instruments to limit exposure to interest rate risk. Our interest gains may decline in the future as a result of changes in the financial markets.
Item 8. Financial Statements and Supplementary Data
See the Index to Consolidated Financial Statements on Page F-1 attached hereto.
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
None.
Item 9A. Controls and Procedures
Evaluation of Disclosure Controls and Procedures
We conducted an evaluation of the effectiveness of the design and operation of our disclosure controls and procedures as of the end of the period covered by this Form 10-K. The controls evaluation was conducted under the supervision and with the participation of management, including our Chief Executive Officer and Chief Financial Officer. Disclosure controls and procedures are controls and procedures designed to reasonably assure that information required to be disclosed in our reports filed under the Exchange Act, such as this Form 10-K, is recorded, processed, summarized and reported within the time periods specified in the Commission’s rules and forms. Disclosure controls and procedures are also designed to reasonably assure that such information is accumulated and communicated to our management, including the Chief Executive Officer and Chief Financial Officer, as appropriate to allow timely decisions regarding required disclosure.
The evaluation of our disclosure controls and procedures included a review of the controls’ objectives and design, our implementation of the controls and their effect on the information generated for use in this Form 10-K. In the course of the controls evaluation, we reviewed identified data errors, control problems or acts of fraud, and sought to confirm that appropriate corrective actions, including process improvements, were being undertaken. This type of evaluation will be performed on a quarterly basis so that the conclusions of management, including the Chief Executive Officer and Chief Financial Officer, concerning the effectiveness of the disclosure controls and procedures can be reported in our periodic reports on Form 10-Q and Form 10-K. The overall goals of these various evaluation activities are to monitor our disclosure controls and procedures, and to modify them as necessary. Our intent is to maintain the disclosure controls and procedures as dynamic systems that change as conditions warrant.
Based on the controls evaluation, our Chief Executive Officer and Chief Financial Officer have concluded that, as of the end of the period covered by this Form 10-K, our disclosure controls and procedures were effective to provide reasonable assurance that information required to be disclosed in our Exchange Act reports is recorded, processed, summarized and reported within the time periods specified by the Commission, and that material information related to our company and our consolidated subsidiary is made known to management, including the Chief Executive Officer and Chief Financial Officer, particularly during the period when our periodic reports are being prepared.
| 74 |
Management Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting to provide reasonable assurance regarding the reliability of our financial reporting and the preparation of financial statements for external purposes in accordance with U.S. generally accepted accounting principles. Internal control over financial reporting includes those policies and procedures that: (i) pertain to the maintenance of records that in reasonable detail accurately and fairly reflect the transactions and dispositions of our assets; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with U.S. generally accepted accounting principles, and that receipts and expenditures of our company are being made only in accordance with authorizations of management and our directors; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of our assets that could have a material effect on our financial statements.
Management assessed our internal control over financial reporting as of December 31, 2011, the end of our fiscal year. Management based its assessment on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission. Management’s assessment included evaluation of elements such as the design and operating effectiveness of key financial reporting controls, process documentation, accounting policies and our overall control environment.
Based on our assessment, management has concluded that our internal control over financial reporting was effective as of the end of the fiscal year to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external reporting purposes in accordance with U.S. generally accepted accounting principles. We reviewed the results of management’s assessment with the Audit Committee of our Board of Directors.
The effectiveness of our internal control over financial reporting as of December 31, 2011 has been audited by Kesselman & Kesselman, an independent registered public accounting firm, as stated in their report included herein.
Inherent Limitations on Effectiveness of Controls
Our management, including our Chief Executive Officer and Chief Financial Officer, does not expect that our disclosure controls and procedures or our internal control over financial reporting will prevent or detect all error and all fraud. A control system, no matter how well designed and operated, can provide only reasonable, not absolute, assurance that the control system’s objectives will be met. The design of a control system must reflect the fact that there are resource constraints, and the benefits of controls must be considered relative to their costs. Further, because of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that misstatements due to error or fraud will not occur or that all control issues and instances of fraud, if any, within a company have been detected. These inherent limitations include the realities that judgments in decision-making can be faulty and that breakdowns can occur because of simple error or mistake. Controls can also be circumvented by the individual acts of some persons, by collusion of two or more people or by management override of the controls. The design of any system of controls is based in part on certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions. Projections of any evaluation of controls effectiveness to future periods are subject to risks. Over time, controls may become inadequate because of changes in conditions or deterioration in the degree of compliance with policies or procedures.
There were no changes in our internal control over financial reporting (as defined in Rules 13a-15f and 15d-15f under the Exchange Act) that occurred during the quarter ended December 31, 2011 that have materially affected, or that are reasonably likely to materially affect, our internal control over financial reporting.
Item 9B. Other Information
None.
| 75 |
PART III
Item 10. Directors, Executive Officers and Corporate Governance
Our directors and executive officers, their ages and positions as of February 1, 2012 are as follows:
| Name | Age | Position | ||
| Directors | ||||
| Zeev Bronfeld | 60 | Interim Chairman of the Board | ||
| David Aviezer, Ph.D., MBA | 47 | Director, President and Chief Executive Officer | ||
| Yoseph Shaaltiel, Ph.D. | 58 | Director and Executive VP, Research and Development | ||
| Alfred Akirov (1)(2)(3) | 71 | Director | ||
| Amos Bar Shalev (1)(2)(3) | 59 | Director | ||
| Yodfat Harel (1)(2)(3) | 39 | Director | ||
| Roger D. Kornberg, Ph.D. | 64 | Director | ||
| Eyal Sheratzky | 43 | Director | ||
| Executive Officers | ||||
| Einat Brill Almon, Ph.D. | 52 | Senior Vice President, Product Development | ||
| Yossi Maimon, CPA | 41 | Vice President, Chief Financial Officer, Treasurer and Secretary | ||
| Sandra L. Lauterbach | 43 | Vice President, Sales and Commercial Affairs | ||
| Tzvi Palash | 55 | Chief Operating Officer |
(1) Member of Nominating Committee
(2) Member of Audit Committee
(3) Member of Compensation Committee
Zeev Bronfeld. Mr. Bronfeld has served as a director of Protalix Ltd. since 1996 and as our director since December 31, 2006. Mr. Bronfeld brings to us vast experience in management and value building of biotechnology companies. Mr. Bronfeld is an experienced businessman who is involved in a number of biotechnology companies. He is a co-founder of Biocell Ltd. (TASE:BCEL), an Israeli publicly traded holding company specializing in biotechnology companies and has served as its Chief Executive Officer since 1986. Mr. Bronfeld currently serves as a director of Biocell Ltd., D. Medical Industries Ltd. (NASDAQ:DMED, TASE:DMDC), Biomedix Incubator Ltd. (TASE:BMDX), D.N.A. Biomedical Solutions Ltd. (TASE:DNA), Gefen Biomed Investments Ltd. (TASE:GEFEN) and Nasvax Ltd. (TASE:NSVX), all of which are public companies traded on the TASE (except D. Medical which is dual-listed on the TASE and the Nasdaq Stock Market LLC). Mr. Bronfeld is also a director of each of the following privately-held companies: Ecocycle Israel Ltd., Contipi Ltd., Spring Health Solutions Ltd., G-Sense Ltd., L.N. Innovative Technologies, A.T.I Ashkelon Industries Information Technologies Ltd., The Trendlines Group, MOFET B'Yehuda – Industrial Research & Development in Judea Ltd., Incubator for Management of Technological Entrepreneurship Misgav Ltd., A.Y.M.B. Holdings and Investments Ltd., Spring-Set Health Solutions Ltd., TransBiodiesel Ltd., Nanutra Ltd., Entera Bio Ltd., Incubator for Management of Technological Entrepreneurship Misgave Ltd., M.B.R.T. Development and Investments Ltd., Macrocure Ltd., Nanothera Ltd., Spearhead Investment Ltd. and Stimatix G.I. Ltd. Mr. Bronfeld received a B.A. in Economics from the Hebrew University in 1975. We believe Mr. Bronfeld's qualifications to serve on our Board of Directors include his years of experience in the management of private and public Israeli companies, including life science companies.
David Aviezer, Ph.D., MBA. Dr. Aviezer has served as Chief Executive Officer of Protalix Ltd. since 2002 and its director since 2005 and as our director since December 31, 2006. On December 31, 2006, he became our President and Chief Executive Officer. Dr. Aviezer has over 15 years of experience in biotechnology management, advancing products from early-stage research up to their regulatory approval and commercialization. Prior to joining Protalix Ltd., from 1996 to 2002, he served as General Manager of ProChon Biotech Ltd., an Israeli company focused on orthopedic disorders. Previously, Dr. Aviezer was a visiting scientist at the Medical Research Division of American Cyanamid, a subsidiary of Wyeth which was subsequently acquired by Pfizer (NYSE:PFE), in New York. Since 1996, Dr. Aviezer has served as an Adjunct Lecturer at Bar Ilan University. Dr. Aviezer is the recipient of the Clore Foundation Award and the J.F. Kennedy Scientific Award. He holds a Ph.D. in Molecular Biology and Biochemistry from the Weizmann Institute of Science, an MBA from the Bar Ilan University Business School, an M.Sc. in Biology from the Weizmann Institute of Science and a B.Sc. in Biology from Bar Ilan University. We believe Dr. Aviezer's qualifications to serve on our Board of Directors include his position as our President and Chief Executive Officer as well as his previous experience in the management of biotechnology companies.
| 76 |
Yoseph Shaaltiel, Ph.D. Dr. Shaaltiel founded Protalix Ltd. in 1993 and has served as a member of our Board of Directors and as our Vice President, Research and Development since December 31, 2006. Prior to establishing Protalix Ltd., from 1988 to 1993, Dr. Shaaltiel was a Research Associate at the MIGAL Technological Center. He also served as Deputy Head of the Biology Department of the Biological and Chemical Center of the Israeli Defense Forces and as a Biochemist at Makor Chemicals Ltd. Dr. Shaaltiel was a Postdoctoral Fellow at the University of California at Berkeley and at Rutgers University in New Jersey. He has co-authored over 40 articles and abstracts on plant biochemistry and holds seven patents. Dr. Shaaltiel received his Ph.D. in Plant Biochemistry from the Weizmann Institute of Science, an M.Sc. in Biochemistry from the Hebrew University and a B.Sc. in Biology from the Ben Gurion University. We believe Dr. Shaaltiel's qualifications to serve on our Board of Directors include his role in founding our company and his continued role in the management of our company.
Alfred Akirov. Mr. Akirov has served as our director since January 2008. Mr. Akirov is the founder, chairman of the Board of Directors and chief executive officer of Alrov (Israel) Ltd. (TASE: ALRO), or Alrov Israel, an Israeli publicly-traded company listed on the TASE. Mr. Akirov founded Alrov Israel in 1978 and it is currently one of Israel's largest real-estate companies. Alrov Israel holds shares of our company directly and indirectly through its subsidiary, Techno-Rov Holdings (1993) Ltd., of which Alrov Israel owns 80% of the outstanding capital stock. Mr. Akirov serves in different capacities, including chairman, chief executive officer and director, for a number of private companies in Alrov Israel's subsidiaries and the Techno-Rov portfolios. We believe Mr. Akirov's qualifications to serve on our Board of Directors include his years of experience in the management of Israeli businesses.
Amos Bar Shalev. Mr. Bar Shalev has served as our director since July 2008. Mr. Bar Shalev served as a director of Protalix Ltd. from 2005 through January 31, 2008, and as our director from December 31, 2006 through January 31, 2008. Mr. Bar Shalev was not nominated for reelection at our annual meeting of shareholders on January 31, 2008. On July 14, 2008, our Board of Directors appointed Mr. Bar Shalev to serve on the board, at which time he was reappointed to the board of directors of Protalix Ltd. as well. Mr. Bar Shalev brings to us extensive experience in managing technology companies. Currently, Mr. Bar Shalev serves as a director of Technorov Holdings (1993) Ltd. and manages its portfolio. In addition, he has served on the board of directors of Aposense Ltd. (TASE: APOS), an Israeli publicly-traded company listed on the TASE, since 2011, and Highcon Systems Ltd., a privately-held Israeli company, since 2010. From 1997 through 2004, he was a Managing Director of TDA Capital Partners, a management company of the TGF (Templeton Tadiran) Fund. From 2004 through 2007, he was the President of Win Buyer Ltd. He has served on the board of directors of many other companies, including, among others, NESS Ltd. (acquired by BioNess Inc.), Idanit (acquired by Scitex Corporation Ltd.), Objet Geometrix, Verisity, Scitex Vision (acquired by Hewlett Packard), Golden Wings Investment Company Ltd., the venture capital fund of the Israeli Air Force Veterans Business Club, Win Buyer Ltd. and Sun Light Ltd. He received his B.Sc. in Electrical Engineering from the Technion, Israel in 1978 and M.B.A. from the Tel Aviv University in 1981. He holds the highest award from the Israeli Air Force for technological achievements. We believe Mr. Bar Shalev's qualifications to serve on our Board of Directors include his years of experience in the management of Israeli businesses.
Yodfat Harel. Ms. Harel has served as our director since June 2007. Since 2006, Ms. Harel has been a Managing Director of Tamares Capital Ltd., a private investment group with interests in real estate, technology, manufacturing, leisure and media. At Tamares Capital, Ms. Harel serves as the Business Development Director and the head of the Israel office. Prior to joining Tamares Capital, from 2004 to 2006, she was the Head of the Medical Desk of Orbotech, Ltd. (NASDAQ:ORBK), a company providing high-tech inspection and imaging solutions for bare printed circuit board (PCB), flat panel display (FPD) and PCB assembly manufacturing worldwide. Prior to that, from 1994 to 2003, she was a Managing Director of Harel-Hertz Investment House Ltd., a business investment company with offices in Tel Aviv, Israel and Tokyo, Japan. In 2002, Harel-Hertz Investment House became the Israeli representative office for ITX Corporation, a publicly-traded company in Japan. Ms. Harel currently serves on the board of directors of Tamares Capital, Tamares Hotels, Tamares Real Estate, Storewiz and Halman-Aldubi Provident Funds, Ltd. Ms. Harel holds a B.A. in Communication and Political Science from Bar Ilan University and an executive M.B.A. from Bradford University, Great Britain. She has also completed programs in Directors' Studies and Advanced Advertising and Marketing at the Israel Management Center. We believe Ms. Harel's qualifications to serve on our Board of Directors include her experience in the management of Israeli and other businesses.
Roger D. Kornberg, Ph.D. Professor Kornberg has served as our director since February 2008. He has served as a director of Teva Pharmaceuticals (NASDAQ:TEVA, TASE:TEVA) since 2007. Professor Kornberg is a member of the U.S. National Academy of Sciences and the Winzer Professor of Medicine in the Department of Structural Biology at Stanford University, Stanford, California. He has been a member of the faculty of Stanford University since 1972. Prior to that, he was a professor at Harvard Medical School. Professor Kornberg is a renowned biochemist and in 2006 he was awarded the Nobel Prize in Chemistry in recognition for his studies of the molecular basis of eukaryotic transcription, the process by which DNA is copied to RNA. Professor Kornberg is also the recipient of several awards, including the 2001 Welch Prize, the highest award granted in the field of chemistry in the United States, and the 2002 Leopold Mayer Prize, the highest award granted in the field of biomedical sciences from the French Academy of Sciences. He received his B.S. in Chemistry from Harvard University in 1967 and his Ph.D. in Chemistry from Stanford University in 1972. He holds honorary degrees from universities in Europe and Israel, including the Hebrew University in Jerusalem, where he currently is a visiting professor. We believe Professor Kornberg's qualifications to serve on our Board of Directors include his expertise in chemistry and medicine and his experience in the academic arena.
| 77 |
Eyal Sheratzky. Mr. Sheratzky has served as a director of Protalix Ltd. since 2005 and as our director since December 31, 2006. Mr. Sheratzky has served as a director of Ituran Location & Control (NASDAQ:ITRN), a publicly-traded company listed on the Nasdaq, since 1995 and as a Co-Chief Executive Officer since 2003. Prior to such date, he served as an alternate Chief Executive Officer of Ituran from 2002 through 2003 and as Vice President of Business Development from 1999 through 2002. Mr. Sheratzky serves as a director of Moked Ituran Ltd. and certain of Ituran's other subsidiaries. Mr. Sheratzky also serves as a director of D. Medical Industries Ltd. (NASDAQ:DMED, TASE:DMDC), as well as of Nilimedix Ltd., its subsidiary. From 1994 to 1999 he served as the Chief Executive Officer of Moked Services, Information and Investments Ltd. and as legal advisor to several of Ituran's affiliated companies. Mr. Sheratzky holds LL.B and LL.M degrees from Tel Aviv University School of Law and an Executive M.B.A. degree from Kellogg School of Management at Northwestern University. We believe Mr. Sheratzky's qualifications to serve on our Board of Directors include his years of experience in the management of Israeli public and private businesses.
Einat Brill Almon, Ph.D. Dr. Almon joined Protalix Ltd. in December 2004, originally as a Senior Director and later as a Vice President and then Senior Vice President, Product Development, and became our Senior Vice President, Product Development on December 31, 2006. Dr. Almon has many years of experience in the management of life science projects and companies, including biotechnology and agrobiotech, with direct experience in clinical, device and scientific software development, as well as a strong background and work experience in Intellectual Property. Prior to joining Protalix Ltd., from 2001 to 2004, she served as Director of R&D and IP of Biogenics Ltd., a company that developed an autologous platform for tissue-based protein drug delivery. Biogenics, based in Israel, is a wholly-owned subsidiary of Medgenics Inc. Dr. Almon has trained as a biotechnology patent agent at leading IP firms in Israel. Dr. Almon holds a Ph.D. and an M.Sc. in molecular biology of cancer research from the Weizmann Institute of Science, a B.Sc. from the Hebrew University and has carried out Post-Doctoral research at the Hebrew University in the area of plant molecular biology.
Yossi Maimon, CPA. Mr. Maimon joined Protalix Ltd. on October 15, 2006 as its Chief Financial Officer and became our Vice President and Chief Financial Officer on December 31, 2006. Prior to joining Protalix, from 2002 to 2006, he served as the Chief Financial Officer of Colbar LifeScience Ltd., a biomaterial company focusing on aesthetics, where he led all of the corporate finance activities, fund raisings and legal aspects of Colbar including the sale of Colbar to Johnson and Johnson. Mr. Maimon has a B.A. in accounting from the City University of New York and an MBA from Tel Aviv University, and he is a Certified Public Accountant in the United States (New York State) and Israel.
Sandra L. Lauterbach. Ms. Lauterbach has served as our Vice President, Sales and Commercial Affairs since December 18, 2009. Prior to joining our company, Ms. Lauterbach was the Vice President of Marketing, Endocrinology of EMD Serono, Inc., from July 2008 through July 2009. Prior to that, from August 2003 through July 2008, she served in a number of positions at Genzyme Corporation, the last position being the Senior Director, Global Marketing of Fabrazyme. Ms. Lauterbach holds a B.Sc. in Molecular Biology from the University of Wisconsin--Madison and an MBA from the University of South Florida.
Tzvi Palash. Mr. Palash has served as Protalix Ltd.’s Chief Operating Officer since September 6, 2010. Prior to joining Protalix Ltd., from 2006 through 2010, Mr. Palash served as a General Manager of ColBar LifeScience Ltd., a biotechnology company specializing in reconstructive medicine and tissue engineering that was acquired by a division of Johnson & Johnson in 2006. In that position, Mr. Palash served as a member of the Global Aesthetic Management Team at the Consumer Group of Johnson & Johnson. Prior to that, from 2001 through 2006, Mr. Palash served as the Vice President, Operations of ColBar LifeScience, and he has served in different positions at Teva. Mr. Palash holds an M.Sc. in Biochemistry from the Hebrew University and a B.Sc. in Biology from the Tel Aviv University.
Section 16(a) Beneficial Ownership Reporting Compliance
Section 16(a) of the Exchange Act requires our directors, executive officers and holders of more than 10% of our common stock to file with the Commission reports regarding their ownership and changes in ownership of our equity securities. We believe that all Section 16 filings requirements were met by our officers and directors during 2011. In making this statement, we have relied solely upon examination of the copies of Forms 3, 4 and 5, Schedule 13s and written representations of our former and current directors, officers and 10% shareholders.
| 78 |
Audit Committee
We require that all Audit Committee members possess the required level of financial literacy and at least one member of the Audit Committee meet the current standard of requisite financial management expertise as required by the NYSE Amex and applicable rules and regulations of the SEC. Messrs. Bar Shalev and Akirov, and Ms. Harel have been appointed by the Board of Directors to serve on the Audit Committee until their respective successors have been duly elected.
Our Audit Committee operates under a formal charter that governs its duties and conduct.
All members of the Audit Committee are independent from our executive officers and management.
Our independent registered public accounting firm reports directly to the Audit Committee.
Our Audit Committee meets with management and representatives of our registered public accounting firm prior to the filing of officers’ certifications with the Commission to receive information concerning, among other things, effectiveness of the design or operation of our internal controls over financial reporting, as required by Section 404 of the Sarbanes-Oxley Act of 2002.
Our Audit Committee has adopted a Policy for Reporting Questionable Accounting and Auditing Practices and Policy Prohibiting Retaliation against Reporting employees to enable confidential and anonymous reporting of improper activities to the Audit Committee.
Messrs. Bar Shalev and Akirov qualify as “audit committee financial experts” under the applicable rules of the SEC. In making the determination as to these individuals’ status as audit committee financial experts, our Board of Directors determined they have accounting and related financial management expertise within the meaning of the aforementioned rules, as well as the listing standards of the NYSE Amex.
Code of Business Conduct and Ethics
We have adopted a Code of Business Conduct and Ethics that includes provisions ranging from restrictions on gifts to conflicts of interest. All of our employees and directors are bound by this Code of Business Conduct and Ethics. Violations of our Code of Business Conduct and Ethics may be reported to the Audit Committee.
The Code of Business Conduct and Ethics includes provisions applicable to all of our employees, including senior financial officers and members of our Board of Directors and is posted on our website (www.protalix.com). We intend to post amendments to or waivers from any such Code of Business Conduct and Ethics.
| Item 11. | Executive Compensation |
Compensation Discussion and Analysis
The primary goals of the Compensation Committee of our Board of Directors with respect to executive compensation are to attract and retain the most talented and dedicated executives possible, to tie annual and long-term cash and stock incentives to achievement of specified performance objectives, and to align executives’ incentives with shareholder value creation. To achieve these goals, the Compensation Committee intends to implement and maintain compensation plans that tie a portion of executives’ overall compensation to key strategic goals such as developments in our clinical path, the establishment of key strategic collaborations, the build-up of our pipeline and the strengthening of our financial position. The Compensation Committee evaluates individual executive performance with a goal of setting compensation at levels the committee believes are comparable with executives in other companies of similar size and stage of development operating in the biotechnology industry while taking into account our relative performance and our own strategic goals.
Elements of Compensation
Executive compensation consists of following elements:
Base Salary. Base salaries for our executives are established based on the scope of their responsibilities taking into account competitive market compensation paid by other companies for similar positions. Generally, we believe that executive base salaries should be targeted near the median of the range of salaries for executives in similar positions with similar responsibilities at comparable companies. The Compensation Committee did not make any compensation decisions during 2011. The companies reviewed by the Compensation Committee in making its compensation decisions in February 2010 include, among other companies, the following:
| 79 |
| · | Keryx Biopharmaceuticals, Inc. |
| · | Savient Pharmaceuticals, Inc. |
| · | Biomarin Pharmaceutical Inc. |
| · | Amicus Therapeutics, Inc. |
| · | Mannkind Corporation |
| · | Nektar Therapeutics |
| · | Theravance, Inc. |
The Compensation Committee intends to continue reviewing and revising the peer group periodically to ensure that it continues to reflect companies similar to us in size and development stage. The Compensation Committee also reviews an executive compensation report and analysis of publicly-traded biotechnology companies prepared by a third party for additional data and other information regarding executive compensation for comparative purposes.
Base salaries are usually reviewed annually, and adjusted from time to time to realign salaries with market levels after taking into account individual responsibilities, performance and experience. The Compensation Committee has decided to delay awarding any increases in the salaries of our executive officers, or awarding any cash bonuses, until after the anticipated FDA approval of our lead product candidate. The base salaries of our named executive officers are set forth in “Employment Arrangements.”
Annual Bonus. The Compensation Committee has the authority to award discretionary annual bonuses to our executive officers. For 2010, the Compensation Committee established a formal bonus plan for certain milestones, as described below. The discretionary annual bonus awards were intended to compensate officers for achieving financial, clinical, regulatory and operational goals and for achieving individual annual performance objectives. For any given year, the compensation objectives vary, but relate generally to strategic factors such as developments in our clinical path, the execution of a license agreement for the commercialization of product candidates, the establishment of key strategic collaborations, the build-up of our pipeline and financial factors such as capital raising. Bonuses are awarded generally based on corporate performance, with adjustments made within a range for individual performance, at the discretion of the Compensation Committee. The Compensation Committee determines, on a discretionary basis, the size of the entire bonus pool and the amount of the actual award to each named executive officer.
The Compensation Committee selects, in its discretion, the executive officers of our company or our subsidiary who are eligible to receive bonuses for any given year. Any bonus granted by the Compensation Committee will generally be paid in the first quarter of the year, unless such bonus was, by its terms, made payable upon the achievement of a specific milestone. The Compensation Committee has not fixed a minimum or maximum award for any executive officer’s annual discretionary bonus, unless specified in the officer’s employment agreement.
Each of our executive officers is eligible for a discretionary annual bonus under his or her employment agreement. The Compensation Committee determined the discretionary annual bonus to be paid to our executive officers for performance in 2009 and 2010. The Compensation Committee has not fixed a minimum or a maximum amount for any officer’s annual discretionary bonus, nor is any executive officer entitled to a minimum or maximum bonus amount under his or her employment agreement.
On February 25, 2010, our Board of Directors, acting upon the resolution of a majority of our independent directors, decided to pay bonuses to our executive officers and other employees in two tranches. The aggregate amount of all of the bonuses awarded or reserved for award by the Board of Directors pursuant to the resolution was approximately $2.6 million. The first tranche of bonus payments awarded in February 2010 to our named executive officers and other employees was for approximately $1.1 million in the aggregate. These bonuses were made on a discretionary basis to acknowledge and compensate our executive officers for their contributions towards the completion of our phase III clinical trial of our lead product candidate, taliglucerase alfa, the upgrade of our manufacturing facility during the years 2008 and 2009, and specifically with the execution of the license and supply agreement with Pfizer relating to taliglucerase alfa. The decision to grant the awards constituting the first tranche of bonuses in 2010 was not based on any predetermined goal set for any named executive officer. However, in making this compensation decision, the Compensation Committee took into account the Board of Directors’ decision to refrain from awarding bonuses to our executive officers and others in 2009 due to the general market conditions and our cash balance at that time. Of the approximately $1.1 million made available for the first tranche of the bonuses, our Board of Directors awarded Dr. Aviezer $500,000; Dr. Shaaltiel $160,000; Dr. Brill Almon $160,000; and Mr. Maimon $160,000. These bonus payments were made in March 2010.
The remaining $1.5 million approved by our Board of Directors in February 2010 was reserved for future payment to our named executive officers and other employees. The second tranche of bonus payments will generally become payable upon the achievement of the milestones described in the following tables, if at all:
| 80 |
First shipment of taliglucerase alfa
| Named Executive Officer | Anticipated Bonus Amount | |||
| David Aviezer, Ph.D., MBA | — | |||
| Yoseph Shaaltiel, Ph.D. | $ | 100,000 | ||
| Einat Brill Almon, Ph.D. | $ | 20,000 | ||
| Yossi Maimon | $ | 20,000 | ||
| Total | $ | 140,000 | ||
Approval of taliglucerase alfa by the FDA
| Named Executive Officer | Anticipated Bonus Amount | |||
| David Aviezer, Ph.D., MBA | $ | 400,000 | ||
| Yoseph Shaaltiel, Ph.D. | $ | 140,000 | ||
| Einat Brill Almon, Ph.D. | $ | 140,000 | ||
| Yossi Maimon | $ | 140,000 | ||
| Total | $ | 820,000 | ||
The first shipment of taliglucerase alfa was made in 2010 and bonuses were paid accordingly. Other than the achievement of the corporate milestones set forth above, the anticipated amounts allocated to each executive officer were not based upon any predetermined goals with respect to any individual named executive officer. Further, no individual goals have been communicated to any individual named executive officer with respect to the eventual payment of the bonuses. The remaining approximately $500,000 was allocated to other employees, subject to the same criteria.
Options. Our 2006 Stock Option Plan authorizes us to grant options to purchase shares of common stock to our employees, directors and consultants. Our Compensation Committee is the administrator of the stock option plan. Stock option grants are generally made at the commencement of employment and following a significant change in job responsibilities or to meet other special retention or performance objectives. The Compensation Committee reviews and approves stock option awards to executive officers based upon a review of competitive compensation data, its assessment of individual performance, a review of each executive’s existing long-term incentives, and retention considerations. The exercise price of stock options granted under the 2006 Stock Incentive Plan must be equal to at least 100% of the fair market value of our common stock on the date of grant; however, in certain circumstances, grants may be made at a lower price to Israeli grantees who are residents of the State of Israel. We have not awarded stock options to any of our executive officers since February 2010, except in connection with the hiring of our Chief Operating Officer and our Vice President, Human Resources.
Severance and Change in Control Benefits. Pursuant to the employment agreements entered into with each of our executive officers based in Israel, the executive officer is entitled to be insured by Protalix Ltd. under a Manager’s Policy in lieu of severance. The intention of such Manager’s Policies is to provide the Israel-based officers with severance protection of one month’s salary for each year of employment. In addition, stock option agreements with each of our named executive officers, as amended, provide that all of the outstanding options of each Named Executive Officer are subject to accelerated vesting immediately upon a change in control of our company.
Other Compensation. Consistent with our compensation philosophy, we intend to continue to maintain our current benefits for our executive officers; however, the Compensation Committee in its discretion may revise, amend, or add to the officer’s executive benefits if it deems it advisable. As an additional benefit to all of our Israel-based Named Executive Officers and for most of our employees, we generally contribute to certain funds amounts equaling a total of approximately 15% of their gross salaries for certain pension and other savings plans for the benefit of the Named Executive Officers. In addition, in accordance with customary practice in Israel, our Israel-based executives’ agreements require us to contribute towards their vocational studies, and to provide annual recreational allowances, a company car and a company phone. We believe these benefits are currently equivalent with median competitive levels for comparable companies.
Executive Compensation. We refer to the “Summary Compensation Table” set forth in Section 11 of the Annual Report on Form 10-K for information regarding the compensation earned during the fiscal year ended December 31, 2011 by: our President and Chief Executive Officer, our Executive Vice President, Research and Development, our Senior Vice President, Product Development, our Vice President and Chief Financial Officer and our Chief Operation Officer, who we refer to collectively as the “Named Executive Officers.”
| 81 |
The Compensation Committee is currently in the process of reviewing all the compensation elements of the named executive officers and of other members of the management team, and updating the peer companies. The Compensation Committee expects to adopt new benefits plans prior to the anticipated FDA approval of taliglucerase alfa, if at all, which plans will likely be subject to the FDA's approval of taliglucerase alfa on or about the upcoming May 1, 2012 action date. Any new bonus plan is expected to be based on our achievement of one or more major milestones.
Compensation Committee Report
The above report of the Compensation Committee does not constitute soliciting material and shall not be deemed filed or incorporated by reference into any other filing by us under the Securities Act of 1933 or the Securities Exchange Act of 1934.
The Compensation Committee has reviewed and discussed the Compensation Discussion and Analysis set forth below with our management. Based on this review and discussion, the Compensation Committee has recommended to our Board of Directors that the Compensation Discussion and Analysis be included in our Annual Report on Form 10–K and our annual proxy statement on Schedule 14A.
Respectfully submitted on February 27, 2012, by the members of the Compensation Committee of the Board of Directors.
Yodfat Harel
Alfred Akirov
Amos Bar Shalev
Summary Compensation Table
The following table sets forth a summary for the fiscal years ended December 31, 2011, 2010 and 2009, respectively, of the cash and non-cash compensation awarded, paid or accrued by us or Protalix Ltd. to each of our President and Chief Executive Officer, our Executive Vice President, Research and Development, our Senior Vice President, Product Development, our Vice President and Chief Financial Officer and our Vice President and Chief Operating Officer, who we refer to collectively as the “Named Executive Officers.” There were no restricted stock awards, long-term incentive plan payouts or other compensation paid during fiscal years 2011, 2010 and 2009 by us or Protalix Ltd. to the Named Executive Officers, except as set forth below. All of the Named Executive Officers are employees of our subsidiary, Protalix Ltd. All currency amounts are expressed in U.S. dollars.
| Name and Principal Position | Year | Salary($) | Bonus ($) | Option
Award(s) ($) | All
Other Compensation ($)(1) | Total ($) | ||||||||||||||||||
| David Aviezer, Ph.D., MBA | 2011 | 531,608 | - | 53,578 | 113,971 | 699,157 | ||||||||||||||||||
| President and | 2010 | 508,868 | - | 145,537 | 107,972 | 762,377 | ||||||||||||||||||
| Chief Executive Officer | 2009 | 427,970 | 500,000 | 529,951 | 62,167 | 1,520,088 | ||||||||||||||||||
| Yoseph Shaaltiel, Ph.D. | 2011 | 305,280 | - | 27,830 | 80,063 | 413,173 | ||||||||||||||||||
| Executive Vice President, | 2010 | 292,095 | 100,000 | 70,286 | 76,491 | 538,872 | ||||||||||||||||||
| Research and Development | 2009 | 196,271 | 160,000 | 225,336 | 37,981 | 619,588 | ||||||||||||||||||
| Einat Brill Almon, Ph.D. | 2011 | 263,357 | - | 32,557 | 71,074 | 366,988 | ||||||||||||||||||
| Senior Vice President, | 2010 | 251,940 | 20,000 | 97,007 | 66,591 | 435,538 | ||||||||||||||||||
| Product Development | 2009 | 172,210 | 160,000 | 283,388 | 36,927 | 652,525 | ||||||||||||||||||
| Yossi Maimon, CPA | 2011 | 286,220 | - | 18,304 | 69,290 | 373,814 | ||||||||||||||||||
| Vice President, | 2010 | 268,154 | 20,000 | 63,175 | 68,156 | 419,485 | ||||||||||||||||||
| Chief Financial Officer | 2009 | 186,478 | 160,000 | 253,030 | 41,051 | 640,559 | ||||||||||||||||||
| Tzvi Palash (2) | 2011 | 233,452 | - | 339,011 | 76,490 | 648,953 | ||||||||||||||||||
| Chief Operating Officer | 2010 | 71,956 | 207,075 | 24,094 | 303,125 | |||||||||||||||||||
| (1) | Includes employer contributions to pension and/or insurance plans and other miscellaneous payments. |
| (2) | Mr. Palash joined our company in September 2010. |
No awards were granted to any Named Executive Officer during 2011.
| 82 |
Outstanding Equity Awards at Fiscal Year-End
The following table sets forth information with respect to the Named Executive Officers concerning equity awards as of December 31, 2011.
| Option Awards | |||||||||||||||
| Name |
Number of Securities Underlying Unexercised Options Exercisable (#) | Number
of Securities Underlying Unexercised Options Unexercisable (#) |
Option Exercise Price ($) | Option
Expiration Date | |||||||||||
| David Aviezer | 326,267 | - | 0.120 | 12/8/2013 | |||||||||||
| 977,297 | - | 0.972 | 9/10/2016 | ||||||||||||
| 533,332 | 66,668 | 5.00 | 2/7/2018 | ||||||||||||
| 100,000 | - | 2.65 | 2/25/2019 | ||||||||||||
| 250,000 | 6.90 | 2/25/2020 | |||||||||||||
| Yoseph Shaaltiel | 122,162 | (1) | - | 0.001 | 6/30/2016 | ||||||||||
| 234,424 | 29,304 | 5.00 | 2/7/2018 | ||||||||||||
| 50,000 | - | 2.65 | 2/25/2019 | ||||||||||||
| 145,000 | 6.90 | 2/25/2020 | |||||||||||||
| Einat Brill Almon | 116,848 | - | 0.972 | 8/13/2016 | |||||||||||
| 276,688 | 34,584 | 5.00 | 2/7/2018 | ||||||||||||
| 50,000 | - | 2.65 | 2/25/2019 | ||||||||||||
| 130,000 | 6.90 | 2/25/2020 | |||||||||||||
| Yossi Maimon | 75,964 | - | 0.972 | 9/19/2016 | |||||||||||
| 155,556 | 19,444 | 5.00 | 2/7/2018 | ||||||||||||
| 50,000 | - | 2.65 | 2/25/2019 | ||||||||||||
| 130,000 | 6.90 | 2/25/2020 | |||||||||||||
| Tzvi Palash | 50,000 | 110,000 | 7.55 | 8/29/2020 | |||||||||||
(1) Pursuant to a divorce settlement, Dr. Shaaltiel is required to transfer 50% of these options to his former spouse.
There were no option exercises by, or vesting of stock awards of, any Named Executive Officer during the year ended December 31, 2011.
Potential Payments upon Termination or Change-in-Control
Each of our Named Executive Officers is entitled to be insured by Protalix Ltd. under a Manager’s Policy in lieu of severance upon termination. The intention of such Manager’s Policies is to provide the Israel-based officers with severance protection of one month’s salary for each year of employment. We do not provide any change in control benefits to our Named Executive Officers except that their stock option agreements, as amended, provide that all of the outstanding options of each Named Executive Officer are subject to accelerated vesting immediately upon a change in control of our company, as defined in our 2006 Stock Incentive Plan. Since as of December 31, 2011 all options that have not been fully vested on such date, had exercise price higher than the closing price on that date, if we had experienced a change of control on December 31, 2011, the value of the acceleration of the stock options held by each of Dr. Aviezer, Dr. Shaaltiel, Dr. Brill Almon, Mr. Maimon and Mr. Palash would be zero.
| 83 |
Employment Arrangements
David Aviezer, Ph.D., MBA. Dr. Aviezer originally served as Protalix Ltd.’s Chief Executive Officer on a consultancy basis pursuant to a Consulting Services Agreement between Protalix Ltd. and Agenda Biotechnology Ltd., a company wholly-owned by Dr. Aviezer. On September 11, 2006, Protalix Ltd. entered into an employment agreement with Dr. Aviezer pursuant to which he agreed to be employed as Protalix Ltd.’s President and Chief Executive Officer, which agreement supersedes the Consultancy Services Agreement. Dr. Aviezer currently serves as our President and Chief Executive Officer. Dr. Aviezer’s current monthly base salary is NIS 148,000 (approximately $38,730) and he is entitled to an annual bonus at the Board’s discretion. The monthly salary is subject to cost of living adjustments from time to time. Dr. Aviezer is eligible to receive a substantial bonus in the event of certain public offerings or acquisition transactions, which bonus shall be at the discretion of the Board, and certain specified bonuses in the event Protalix achieves certain specified milestones. In connection with the employment agreement, in addition to other options already held by Dr. Aviezer granted to Dr. Aviezer options to purchase 16,000 ordinary shares of Protalix Ltd. at an exercise price equal to $59.40 per share, which we assumed as options to purchase 977,297 shares of our common stock at $0.97 per share. Such options vest quarterly retroactively from June 1, 2006, over a four-year period. In addition, in 2008 we granted to Dr. Aviezer an option to purchase 600,000 shares of our common stock at an exercise price equal to $5.00 per share. The option vests variably over a five-year period that commenced on January 1, 2008. In 2009, we granted Dr. Aviezer an option to purchase 100,000 shares of our common stock at an exercise price equal to $2.65 per share. As of December 31, 2009, all of those options had fully vested. In 2010, we granted Dr. Aviezer an option to purchase 250,000 shares of our common stock at an exercise price equal to $6.90 per share, which option vests quarterly over a three-year period commencing upon FDA approval of taliglucerase alfa, if at all. Dr. Aviezer’s employment agreement is terminable by either party on 90 days’ written notice for any reason and we may terminate the agreement for cause without notice. Dr. Aviezer is entitled to be insured by Protalix Ltd. under a Manager’s Policy in lieu of severance, company contributions towards vocational studies, annual recreational allowances, a company car and a company phone. Dr. Aviezer is entitled to 26 working days of vacation. All stock options that have not vested as of the date of termination shall be deemed to have been forfeited.
Yoseph Shaaltiel, Ph.D. Dr. Shaaltiel founded Protalix Ltd. in 1993 and currently serves as our Executive Vice President, Research and Development. Dr. Shaaltiel entered into an employment agreement with Protalix Ltd. on September 1, 2001. Pursuant to the employment agreement, his current monthly base salary is NIS 85,000 (approximately $22,250) per month. The employment agreement is terminable by Protalix Ltd. on 90 days’ written notice for any reason and we may terminate the agreement for cause without notice. In 2008 we granted to Dr. Shaaltiel an option to purchase 263,728 shares of our common stock at an exercise price equal to $5.00 per share. The option vests variably over a five-year period that commenced on January 1, 2008. In 2009, we granted Dr. Shaaltiel an option to purchase 50,000 shares of our common stock at an exercise price equal to $2.65 per share. As of December 31, 2009, all of those options had fully vested. In 2010, we granted Dr. Shaaltiel an option to purchase 145,000 shares of our common stock at an exercise price equal to $6.90 per share, which option vests quarterly over a three-year period commencing upon FDA approval of taliglucerase alfa, if at all. Dr. Shaaltiel is entitled to be insured by Protalix Ltd. under a Manager’s Policy in lieu of severance, company contributions towards vocational studies, annual recreational allowances, a company car and a company phone. Dr. Shaaltiel is entitled to 33 working days of vacation.
Einat Brill Almon, Ph.D. Dr. Brill Almon joined Protalix Ltd. on December 19, 2004 as its Vice President, Product Development, pursuant to an employment agreement effective on December 19, 2004 by and between Protalix Ltd. and Dr. Brill Almon, and currently serves as our Senior Vice President, Product Development. Pursuant to the employment agreement, her current monthly base salary is NIS 73,500 per month (approximately $19,240). She is also entitled to certain specified bonuses in the event that Protalix achieves certain specified clinical development milestones within specified timelines. In connection with the employment agreement, Protalix agreed to grant to Dr. Brill Almon options to purchase 7,919 ordinary shares of Protalix Ltd. at exercise prices equal to $24.36 and $59.40 per share, which we assumed as options to purchase 483,701 shares of our common stock at $0.40 and $0.97 per share. The options vest over four years. In addition, in 2008 we granted to Dr. Brill Almon an option to purchase 311,272 shares of our common stock at an exercise price equal to $5.00 per share. The option vests variably over a five-year period that commenced on January 1, 2008. In 2009, we granted to Dr. Brill Almon an option to purchase 50,000 shares of our common stock at an exercise price equal to $2.65 per share. As of December 31, 2009, all of those options had fully vested. In 2010, we granted Dr. Almon an option to purchase 130,000 shares of our common stock at an exercise price equal to $6.90 per share, which option vests quarterly over a three-year period commencing upon FDA approval of taliglucerase alfa, if at all. The employment agreement is terminable by either party on 60 days’ written notice for any reason and we may terminate the agreement for cause without notice. Dr. Brill Almon is entitled to be insured by Protalix Ltd. under a Manager’s Policy in lieu of severance, company contributions towards vocational studies, annual recreational allowances, a company car and a company phone at up to NIS 1,000 per month. Dr. Brill Almon is entitled to 28 working days of vacation. All stock options that have not vested as of the date of termination shall be deemed to have been forfeited.
Yossi Maimon, CPA. Mr. Maimon joined Protalix Ltd. as its Chief Financial Officer pursuant to an employment agreement effective as of October 15, 2006 by and between Protalix Ltd. and Mr. Maimon and currently serves as our Chief Financial Officer. Pursuant to the employment agreement, his current monthly base salary is NIS 73,500 (approximately $19,240) and Mr. Maimon is entitled to an annual discretionary bonus and additional discretionary bonuses in the event Protalix achieves significant financial milestones, subject to the Board’s sole discretion. The monthly salary is subject to cost of living adjustments from time to time. In connection with the employment agreement, Protalix agreed to grant to Mr. Maimon options to purchase 10,150 ordinary shares of Protalix Ltd. at an exercise price equal to $59.40 per share, which we assumed as options to purchase 619,972 shares of our common stock at $0.97 per share. The first 25% of such options shall vest on the first anniversary of the grant date and the remainder shall vest quarterly in 12 equal increments. In addition, in 2008 we granted to Mr. Maimon an option to purchase 175,000 shares of our common stock at an exercise price equal to $5.00 per share. The option vests variably over a five-year period that commenced on January 1, 2008. In 2009, we granted to Mr. Maimon an option to purchase 50,000 shares of our common stock at an exercise price equal to $2.65 per share. As of December 31, 2009, all of those options had fully vested. In 2010, we granted Mr. Maimon an option to purchase 130,000 shares of our common stock at an exercise price equal to $6.90 per share, which option vests quarterly over a three-year period commencing upon FDA approval of taliglucerase alfa, if at all. The employment agreement is terminable by either party on 60 days’ written notice for any reason and we may terminate the agreement for cause without notice. Mr. Maimon is entitled to be insured by Protalix Ltd. under a Manager’s Policy in lieu of severance, company contributions towards vocational studies, annual recreational allowances, a company car and a company phone. Mr. Maimon is entitled to 28 working days of vacation. All stock options that have not vested as of the date of termination shall be deemed to have been forfeited.
| 84 |
Tzvi Palash. Mr. Palash joined Protalix Ltd. as its Chief Operating Officer pursuant to an employment agreement effective September 6, 2010 and currently serves as our Chief Operating Officer. Pursuant to the employment agreement, Mr. Palash’s current monthly base salary is NIS 69,000 (approximately $18,060) and Mr. Palash is entitled to an annual discretionary bonus for performance subject to the sole discretion of our compensation committee. The monthly salary is subject to cost of living adjustments from time to time as may be required by law. In connection with the employment agreement, we granted to Mr. Palash options to purchase 160,000 shares of our common stock with an exercise price equal to $7.55 per share. The first 25% of such options vested on the first anniversary of the grant date and the remainder vest quarterly in 12 equal increments. The employment agreement is terminable by either party on 60 days’ written notice for any reason and we may terminate the agreement for cause without notice. Mr. Palash is entitled to be insured by Protalix Ltd. under a Manager’s Policy in lieu of severance, company contributions towards vocational studies, annual recreational allowances, a company car, a company phone, a company laptop and lodging accommodations in the Carmiel area. Mr. Palash is entitled to 24 working days of vacation.
2006 Stock Incentive Plan
Our Board of Directors and a majority of our shareholders approved our 2006 Stock Incentive Plan on December 14, 2006 and cancelled our 1998 stock option plan (no options were outstanding under the 1998 plan at that time). We have reserved 9,741,655 shares of our common stock for issuance, in the aggregate, under the 2006 Stock Incentive Plan, subject to adjustment for a stock split or any future stock dividend or other similar change in our common stock or our capital structure. As of December 31, 2011, options to acquire 47,920 shares of common stock remain available for grant under our 2006 Stock Incentive Plan.
Our 2006 Stock Incentive Plan provides for the grant of stock options, restricted stock, restricted stock units, stock appreciation rights and dividend equivalent rights, collectively referred to as “awards.” Stock options granted under the 2006 Stock Incentive Plan may be either incentive stock options under the provisions of Section 422 of the Internal Revenue Code, or non-qualified stock options. Incentive stock options may be granted only to employees. Awards other than incentive stock options may be granted to employees, directors and consultants.
The 2006 Stock Incentive Plan is also designed to comply with the provisions of the Israeli Income Tax Ordinance New Version, 1961 (including as amended pursuant to Amendment 132 thereto) (the “tax ordinance”) and is intended to enable us to grant awards to grantees who are Israeli residents as follows: (i) awards to employees pursuant to Section 102 of the tax ordinance; and (ii) awards to non-employees pursuant to Section 3(I) of the tax ordinance. For this purpose, “employee” refers only to employees, office holders and directors of our company or a related entity excluding those who are considered “Controlling Shareholders” pursuant to, or otherwise excluded by, the tax ordinance. In accordance with the terms and conditions imposed by the Tax Ordinance, grantees who receive awards under the 2006 Stock Incentive Plan may be afforded certain tax benefits in Israel as described below.
Our Board of Directors or the Compensation Committee, referred to as the “plan administrator,” will administer our 2006 Stock Incentive Plan, including selecting the grantees, determining the number of shares to be subject to each award, determining the exercise or purchase price of each award, and determining the vesting and exercise periods of each award.
The exercise price of stock options granted under the 2006 Stock Incentive Plan must be equal to at least 100% of the fair market value of our common stock on the date of grant; however, in certain circumstances, grants may be made at a lower price to Israeli grantees who are residents of the State of Israel. If, however, incentive stock options are granted to an employee who owns stock possessing more than 10% of the voting power of all classes of our stock or the stock of any parent or subsidiary of our company, the exercise price of any incentive stock option granted must equal at least 110% of the fair market value on the grant date and the maximum term of these incentive stock options must not exceed five years. The maximum term of all other awards must not exceed 10 years (or five years in the case of an incentive stock option granted to any participant who owns stock representing more than 10% of the voting power of all classes of our stock or the stock of any parent or subsidiary of our company). The plan administrator will determine the exercise or purchase price (if any) of all other awards granted under the 2006 Stock Incentive Plan.
| 85 |
Under the 2006 Stock Incentive Plan, incentive stock options and options to Israeli grantees may not be sold, pledged, assigned, hypothecated, transferred or disposed of in any manner other than by will or by the laws of descent or distribution and may be exercised during the lifetime of the participant only by the participant. Other awards shall be transferable by will or by the laws of descent or distribution and to the extent and in the manner authorized by the plan administrator by gift or pursuant to a domestic relations order to members of the participant’s immediate family. The 2006 Stock Incentive Plan permits the designation of beneficiaries by holders of awards, including incentive stock options.
If the service of a participant in the 2006 Stock Incentive Plan is terminated for any reason other than cause, the participant may exercise awards that were vested as of the termination date for a period ending upon the earlier of 12 months from the date of termination (or such shorter or longer period set forth in the award agreement) or the expiration date of the awards unless otherwise determined by the plan administrator. If the service of a participant in the 2006 Stock Incentive Plan is terminated for cause, the participant may exercise awards that were vested as of the termination date for a period ending upon the earlier of 14 days from the date of termination (or such shorter or longer period set forth in the award agreement) or the expiration date of the awards unless otherwise determined by the plan administrator.
In the event of a corporate transaction, all awards will terminate unless assumed by the successor corporation. Unless otherwise provided in a participant’s award agreement, in the event of a corporate transaction and with respect to the portion of each award that is assumed or replaced, then such portion will automatically become fully vested and exercisable immediately upon termination of a participant’s service if the participant is terminated by the successor company or us without cause within 12 months after the corporate transaction. With respect to the portion of each award that is not assumed or replaced, such portion will automatically become fully vested and exercisable immediately prior to the effective date of the corporate transaction so long as the participant’s service has not been terminated prior to such date.
In the event of a change in control, except as otherwise provided in a participant’s award agreement, following a change in control (other than a change in control that also is a corporate transaction) and upon the termination of a participant’s service without cause within 12 months after a change in control, each award of such participant that is outstanding at such time will automatically become fully vested and exercisable immediately upon the participant’s termination. In addition, the stock options issued to each of our Named Executive Officers are subject to accelerated vesting immediately upon a change in control of our company, as defined in our 2006 Stock Incentive Plan.
Under our 2006 Stock Incentive Plan, a corporate transaction is generally defined as:
· a merger or consolidation in which we are not the surviving entity, except for the principal purpose of changing our company’s state of incorporation;
· the sale, transfer or other disposition of all or substantially all of our assets;
· the complete liquidation or dissolution of our company;
· any reverse merger in which we are the surviving entity but our shares of common stock outstanding immediately prior to such merger are converted or exchanged by virtue of the merger into other property, whether in the form of securities, cash or otherwise, or in which securities possessing more than forty percent (40%) of the total combined voting power of our outstanding securities are transferred to a person or persons different from those who held such securities immediately prior to such merger; or
· acquisition in a single or series of related transactions by any person or related group of persons of beneficial ownership of securities possessing more than fifty percent (50%) of the total combined voting power of our outstanding securities but excluding any such transaction or series of related transactions that the plan administrator determines not to be a corporate transaction (provided however that the plan administrator shall have no discretion in connection with a corporate transaction for the purchase of all or substantially all of our shares unless the principal purpose of such transaction is changing our company’s state of incorporation).
Under our 2006 Stock Incentive Plan, a change of control is defined as:
· the direct or indirect acquisition by any person or related group of persons of beneficial ownership of securities possessing more than fifty percent (50%) of the total combined voting power of our outstanding securities pursuant to a tender or exchange offer made directly to our shareholders and which a majority of the members of our board (who have generally been on our board for at least 12 months) who are not affiliates or associates of the offeror do not recommend shareholders accept the offer; or
| 86 |
· a change in the composition of our board over a period of 12 months or less, such that a majority of our board members ceases, by reason of one or more contested elections for board membership, to be comprised of individuals who were previously directors of our company.
Unless terminated sooner, the 2006 Stock Incentive Plan will automatically terminate in 2016. Our Board of Directors has the authority to amend, suspend or terminate our 2006 Stock Incentive Plan. No amendment, suspension or termination of the 2006 Stock Incentive Plan shall adversely affect any rights under awards already granted to a participant. To the extent necessary to comply with applicable provisions of federal securities laws, state corporate and securities laws, the Internal Revenue Code, the rules of any applicable stock exchange or national market system, and the rules of any non-U.S. jurisdiction applicable to awards granted to residents therein (including the Tax Ordinance), we shall obtain shareholder approval of any such amendment to the 2006 Stock Incentive Plan in such a manner and to such a degree as required.
Impact of Israeli Tax Law
The awards granted to employees pursuant to Section 102 of the Tax Ordinance under the 2006 Stock Incentive Plan may be designated by us as approved options under the capital gains alternative, or as approved options under the ordinary income tax alternative.
To qualify for these benefits, certain requirements must be met, including registration of the options in the name of a trustee. Each option, and any shares of common stock acquired upon the exercise of the option, must be held by the trustee for a period commencing on the date of grant and deposit into trust with the trustee and ending 24 months thereafter.
Under the terms of the capital gains alternative, we may not deduct expenses pertaining to the options for tax purposes.
Under the 2006 Stock Incentive Plan, we may also grant to employees options pursuant to Section 102(c) of the Tax Ordinance that are not required to be held in trust by a trustee. This alternative, while facilitating immediate exercise of vested options and sale of the underlying shares, will subject the optionee to the marginal income tax rate of up to 50% as well as payments to the National Insurance Institute and health tax on the date of the sale of the shares or options. Under the 2006 Stock Incentive Plan, we may also grant to non-employees options pursuant to Section 3(I) of the Tax Ordinance. Under that section, the income tax on the benefit arising to the optionee upon the exercise of options and the issuance of common stock is generally due at the time of exercise of the options.
These options shall be further subject to the terms of the tax ruling that has been obtained by Protalix Ltd. from the Israeli tax authorities in connection with the merger. Under the tax ruling, the options issued by us in connection with the assumption of Section 102 options previously issued by Protalix Ltd. under the capital gains alternative shall be issued to a trustee, shall be designated under the capital gains alternative and the issuance date of the original options shall be deemed the issuance date for the assumed options for the calculation of the respective holding period.
| 87 |
Compensation of Directors
The following table sets forth information with respect to compensation of our non-employee directors during fiscal year 2011. The fees to our current directors were paid by Protalix Ltd.
| Name | Fees Earned or Paid in Cash ($) | Total ($) | ||||||
| Current Directors | ||||||||
| Zeev Bronfeld | 33,000 | 33,000 | ||||||
| Alfred Akirov | 33,000 | 33,000 | ||||||
| Amos Bar Shalev | 50,000 | 50,000 | ||||||
| Yodfat Harel | 33,000 | 33,000 | ||||||
| Roger D. Kornberg | 33,000 | 33,000 | ||||||
| Eyal Sheratzky | 33,000 | 33,000 | ||||||
Our Board of Directors will review director compensation annually and adjust it according to then current market conditions and corporate governance guidelines.
Compensation Committee Interlocks and Insider Participation
Our Compensation Committee currently consists of Messrs. Akirov and Bar Shalev and Ms. Harel, who were appointed to the Committee during 2009. No member of our Compensation Committee or any executive officer of our company or of Protalix Ltd. has a relationship that would constitute an interlocking relationship with executive officers or directors of another entity. No Compensation Committee member is or was an officer or employee of ours or of Protalix Ltd. Further, none of our executive officers serves on the board of directors or compensation committee of any entity that has one or more executive officers serving as a member of our Board of Directors or Compensation Committee.
| 88 |
| Item 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters |
Security Ownership of Certain Beneficial Owners and Management
The following table sets forth information, as of February 1, 2012, regarding beneficial ownership of our common stock:
| · | each person who is known by us to own beneficially more than 5% of our common stock; |
| · | each director; |
| · | each of our Chief Executive Officer, our Executive Vice President, Research and Development, our Senior Vice President, Product Development, our Chief Financial Officer, our Vice President, Sales and Commercial Affairs and our Chief Operating Officer; and |
| · | all of our directors and executive officers collectively. |
Unless otherwise noted, we believe that all persons named in the table have sole voting and investment power with respect to all shares of our common stock beneficially owned by each of them. For purposes of these tables, a person is deemed to be the beneficial owner of securities that can be acquired by such person within 60 days from February 1, 2012 upon exercise of options, warrants and convertible securities. Each beneficial owner’s percentage ownership is determined by assuming that options, warrants and convertible securities that are held by such person (but not those held by any other person) and that are exercisable within such 60 days from such date have been exercised. The information set forth below is based upon information obtained from the beneficial owners, upon information in our possession regarding their respective holdings and upon information filed by the holders with the SEC. The percentages of beneficial ownership are based on 85,631,957 shares of our common stock outstanding as of February 1, 2012.
The address for all directors and officers is c/o Protalix BioTherapeutics, Inc., 2 Snunit Street, Science Park, POB 455, Carmiel, Israel, 20100.
| Name and Address of Beneficial Owner | Amount and Nature of Beneficial Ownership | Percentage of Class | ||||||
| Board of Directors and Executive Officers | ||||||||
| David Aviezer, Ph.D., MBA (1) | 1,953,653 | 2.3 | % | |||||
| Yoseph Shaaltiel, Ph.D. (2) | 1,177,663 | 1.4 | ||||||
| Alfred Akirov (3) | 5,286,046 | 6.2 | ||||||
| Amos Bar Shalev (4) | 1,680 | * | ||||||
| Zeev Bronfeld (5) | 14,466,319 | 16.9 | ||||||
| Yodfat Harel | — | — | ||||||
| Roger D. Kornberg, Ph.D. (6) | 50,000 | * | ||||||
| Eyal Sheratzky | — | — | ||||||
| Einat Brill Almon, Ph.D. (7) | 452,182 | * | ||||||
| Yossi Maimon (8) | 286,381 | * | ||||||
| Tzvi Palash (9) | 60,000 | * | ||||||
| Sandra E. Lauterbach (10) | 80,000 | * | ||||||
| All executive officers and directors as a group (12 persons) (11) | 23,812,157 | 27.8 | ||||||
| 5% Holders | ||||||||
| Biocell Ltd. (12) | 14,466,319 | 16.9 | ||||||
| Federated Investors Inc. (13) | 8,646,942 | 10.1 | ||||||
| Al-Rov (Israel) Ltd. (14) | 5,286,046 | 6.2 | ||||||
| Baillie Gifford & Co. (15) | 4,852,882 | 5.7 | ||||||
* less than 1%.
| (1) | Consists of 1,953,653 shares of our common stock issuable upon exercise of outstanding options within 60 days of February 1, 2012. Does not include 300,001 shares of common stock issuable upon exercise of outstanding options that are not exercisable within 60 days of February 1, 2012. |
| 89 |
| (2) | Consists of 763,754 shares of our common stock held by Dr. Shaaltiel and 413,912 shares of our common stock issuable upon exercise of outstanding options within 60 days of February 1, 2012. Does not include 166,978 shares of common stock issuable upon exercise of outstanding options that are not exercisable within 60 days of February 1, 2012. |
| (3) | Consists of 3,600,000 shares of our common stock held by Al-Rov (Israel) Ltd., or Al-Rov Israel, and 1,686,046 shares of our common stock held by Techno-Rov Holdings (1993) Ltd., or Techno-Rov Holdings. Al-Rov Israel owns 100% of the outstanding shares of Al-Rov Technologies Holdings LTD, the holder of 80% of Techno-Rov Holdings. Mr. Akirov is the Chairman of Al-Rov Israel and the Chief Executive Officer of Techno-Rov Holdings and has the power to control their respective investment decisions. Mr. Akirov disclaims beneficial ownership of these shares except to the extent of his pecuniary interest therein. |
| (4) | Mr. Bar Shalev is a director of Techno-Rov Holdings. |
| (5) | Consists of 14,466,319 shares of our common stock held by Biocell Ltd. Mr. Bronfeld is a director and Chief Executive Officer of Biocell. Mr. Bronfeld disclaims beneficial ownership of these shares except to the extent of his pecuniary interest therein. |
| (6) | Consists of 50,000 shares of our common stock issuable upon exercise of outstanding options within 60 days of February 1, 2012. |
| (7) | Consists of 452,182 shares of our common stock issuable upon exercise of outstanding options within 60 days of February 1, 2012. Does not include 155,938 shares of common stock issuable upon exercise of outstanding options that are not exercisable within 60 days of February 1, 2012. |
| (8) | Consists of 286,381 shares of our common stock issuable upon exercise of outstanding options within 60 days of February 1, 2012. Does not include 144,583 shares of common stock issuable upon exercise of outstanding options that are not exercisable within 60 days of February 1, 2012. |
| (9) | Consists of 60,000 shares of common stock issuable upon exercise of outstanding options that are not exercisable within 60 days of February 1, 2012. Does not include 100,000 shares of common stock issuable upon exercise of outstanding options that are not exercisable within 60 days of February 1, 2012. |
| (10) | Consists of 80,000 shares of common stock issuable upon exercise of outstanding options that are not exercisable within 60 days of February 1, 2012. Does not include 80,000 shares of common stock issuable upon exercise of outstanding options that are not exercisable within 60 days of February 1, 2012. |
| (11) | Consists of 20,516,119 shares of our common stock and 3,296,038 shares of our common stock issuable upon exercise of outstanding options within 60 days of February 1, 2012. Does not include 947,500 shares of common stock issuable upon exercise of outstanding options that are not exercisable within 60 days of February 1, 2012. |
| (12) | The address is Moshe Aviv Tower, 7 Jabotinsky Street, Ramat Gan, Israel. Biocell Ltd.’s investment and voting decisions are made collectively by its board of directors. |
| (13) | Based solely on a Schedule 13G/A filed by Federated Investors Inc. on February 10, 2012. |
| (14) | The address is Alrov Tower, 46 Rothschild Blvd., Tel Aviv, Israel 66883. Consists of 3,600,000 shares of our common stock held by Al-Rov Israel directly and 1,686,046 shares of our common stock held by Techno-Rov Holdings. Al-Rov Israel owns 100% of the outstanding shares of Al-Rov Technologies Holdings LTD, the holder of 80% of Techno-Rov Holdings. Mr. Akirov is the Chairman of Al-Rov Israel and the Chief Executive Officer of Techno-Rov Holdings and has the power to control their respective investment decisions. |
| (15) | Based solely on a Schedule 13G/A filed by Baillie Gifford & Co. on January 17, 2012. According to Baillie Gifford & Co., it has sole dispositive power over 4,852,882 shares of common stock and sole voting power over 2,305,339 shares of common stock. |
| 90 |
Equity Compensation Plan Information
The following table provides information as of December 31, 2011 with respect to the shares of our common stock that may be issued under our existing equity compensation plan.
| A | B | C | ||||||||||
| Number of Securities Remaining | ||||||||||||
| Number of Securities | Available for Future Issuance | |||||||||||
| to be Issued | Weighted Average | Under Equity Compensation Plans | ||||||||||
| Upon Exercise of | Exercise Price of | (Excluding Securities Reflected in | ||||||||||
| Plan Category | Outstanding Options | Outstanding Options | Column A) | |||||||||
| Equity Compensation Plans Approved by Shareholders | 6,747,856 | $ | 3.27 | 47,920 | ||||||||
| Equity Compensation Plans Not Approved by Shareholders | 631,866 | $ | 10.24 | - | ||||||||
| Total | 7,379,722 | $ | 3.87 | 47,920 | ||||||||
| Item 13. | Certain Relationships and Related Transactions, and Director Independence |
On March 17, 2005, Protalix Ltd. entered into a Management Services Agreement with Pontifax Management Company, Ltd. in connection with the purchase of Protalix’s Series B Preferred Shares by the Pontifax Funds. Pursuant to the Management Services Agreement, Mr. Hurvitz served as a member of our Board of Directors and later as the Chairman of our Board of Directors until his resignation in March 2010. In consideration for Mr. Hurvitz’s services, Protalix was required to pay Pontifax Management Company a fee equal to $3,000 per month plus required taxes on such payment. In addition, in connection with the execution of the Management Services Agreement and the later appointment of Mr. Hurvitz as Chairman of our Board of Directors, Protalix issued to Pontifax, in the aggregate, a number of options that, upon our December 2006 merger transaction, were converted into options to purchase 3,384,502 shares of our common stock. Under the terms of the assumed Management Services Agreement, we are obligated only to use our best efforts to nominate Mr. Hurvitz for election to our Board of Directors, which remains subject to the review and approval of the Nominating Committee of the Board of Directors and the entire Board of Directors, as applicable. During the year 2010 through the date of Mr. Hurvitz’s resignation, the fee payable under this agreement was increased to $33,000 per annum, which is the same fee payable to the other non-executive directors. No further payments were due to Pontifax Management Company after Mr. Hurvitz’s resignation from our Board of Directors.
On September 14, 2006, Protalix Ltd. entered into a collaboration and licensing agreement with Teva for the development and manufacture of two proteins using ProCellEx, our proprietary protein expression system. Mr. Hurvitz, our former Chairman, was the chairman of Teva’s board of directors when we entered into the agreement. Phillip Frost, M.D., a former director and a major shareholder of our company, is the chairman of Teva’s board of directors and Professor Kornberg, a member of our Board of Directors also serves as a member of Teva’s board of directors. The agreement provides that we will collaborate with Teva on the research and development of two proteins using ProCellEx. We and Teva identified two proteins for research and development activities under the agreement, but in 2009 both of the projects were terminated for commercial reasons. Other elements of our collaboration with Teva are currently ongoing.
All related party transactions are reviewed and approved by the Audit Committee, as required by the Audit Committee Charter.
Corporate Governance and Independent Directors
In compliance with the listing requirements of the NYSE Amex, we have a comprehensive plan of corporate governance for the purpose of defining responsibilities, setting high standards of professional and personal conduct and assuring compliance with such responsibilities and standards. We currently regularly monitor developments in the area of corporate governance to ensure we are in compliance with the standards and regulations required by the NYSE Amex. A summary of our corporate governance measures follows.
| 91 |
Independent Directors
We believe a majority of the members of our Board of Directors are independent from management. When making determinations from time to time regarding independence, the Board of Directors will reference the listing standards adopted by the NYSE Amex as well as the independence standards set forth in the Sarbanes-Oxley Act of 2002 and the rules and regulations promulgated by the SEC under that Act. In particular, our Audit Committee periodically evaluates and reports to the Board of Directors on the independence of each member of the Board. We anticipate our audit committee will analyze whether a director is independent by evaluating, among other factors, the following:
| · | Whether the member of the Board of Directors has any material relationship with us, either directly, or as a partner, shareholder or officer of an organization that has a relationship with us; |
| · | Whether the member of the Board of Directors is a current employee of our company or any of our subsidiaries, or was an employee of our company or any of our subsidiaries within three years preceding the date of determination; |
| · | Whether the member of the Board of Directors is, or in the three years preceding the date of determination has been, affiliated with or employed by (i) a present internal or external auditor of our company or any affiliate of such auditor or (ii) any former internal or external auditor of our company or any affiliate of such auditor, which performed services for us within three years preceding the date of determination; |
| · | Whether the member of the Board of Directors is, or in the three years preceding the date of determination has been, part of an interlocking directorate, in which any of our executive officers serve on the Compensation Committee of another company that concurrently employs the member as an executive officer; |
| · | Whether the member of the Board of Directors receives any compensation from us, other than fees or compensation for service as a member of the Board of Directors and any committee of the Board of Directors and reimbursement for reasonable expenses incurred in connection with such service and for reasonable educational expenses associated with Board of Directors or committee membership matters; |
| · | Whether an immediate family member of the member of the Board of Directors is a current executive officer of our company or was an executive officer of our company within three years preceding the date of determination; |
| · | Whether an immediate family member of the member of the Board of Directors is, or in the three years preceding the date of determination has been, affiliated with or employed in a professional capacity by (i) a present internal or external auditor of ours or any of our affiliates or (ii) any former internal or external auditor of our company or any affiliate of ours which performed services for us within three years preceding the date of determination; and |
| · | Whether an immediate family member of the member of the Board of Directors is, or in the three years preceding the date of determination has been, part of an interlocking directorate, in which any of our executive officers serve on the Compensation Committee of another company that concurrently employs the immediate family member of the member of the Board of Directors as an executive officer. |
The above list is not exhaustive and we anticipate that the Audit Committee will consider all other factors which could assist it in its determination that a director will have no material relationship with us that could compromise that director’s independence.
Under these standards, our Board of Directors has determined that Messrs. Akirov and Bar Shalev and Ms. Harel are considered “independent” pursuant to the rules of the NYSE Amex and Section 10A(m)(3) of the Exchange Act. In addition, our Board of Directors has determined that at least two of these directors are able to read and understand fundamental financial statements and have substantial business experience that results in their financial sophistication, qualifying them for membership on any audit committee we form. Our Board of Directors has also determined that Messrs. Akirov, Bar Shalev, Bronfeld and Sheratzky, Ms. Harel and Dr. Kornberg are “independent” pursuant to the rules of the NYSE Amex.
The position of chairman of the board is not held by our chief executive officer at this time. The Board of Directors does not have a policy mandating the separation of these functions. We believe it is in our best interest that Mr. Bronfeld serve as the interim chairman of the board. This decision was based on Mr. Bronfeld’s experience in the healthcare industry in Israel and globally and his years of experience serving on the board of directors of many public and private companies. Our non-management directors hold formal meetings, separate from management, at least twice per year. We have no formal policy regarding attendance by our directors at annual shareholders meetings, although we encourage such attendance and anticipate most of our directors will attend these meetings. Messrs. Bronfeld and Bar Shalev, Dr. Aviezer, Dr. Shaaltiel and Ms. Harel, attended our 2011 annual meeting of shareholders.
| 92 |
The Board’s Role in Risk Oversight
Our Board of Directors oversees an enterprise-wide approach to risk management, designed to support the achievement of business objectives, including organizational and strategic objectives, to improve long-term organizational performance and enhance shareholder value. The involvement of our Board of Directors in setting our business strategy is a key part of its assessment of management’s plans for risk management and its determination of what constitutes an appropriate level of risk for the company. The participation of our Board of Directors in our risk oversight process includes receiving regular reports from members of senior management on areas of material risk to our company, including operational, financial, legal and regulatory, and strategic and reputational risks. While the full board has the ultimate oversight responsibility for the risk management process, various committees of the board also have responsibility for risk management. For example, financial risks, including internal controls, are overseen by the audit committee and risks that may be implicated by our executive compensation programs are overseen by the compensation committee. Upon identification of a risk, the assigned board committee or our full Board of Directors discuss or review risk management and risk mitigation strategies. Additional review or reporting on enterprise risks is conducted as needed or as requested by our Board of Directors or a committee thereof.
| Item 14. | Principal Accountant Fees and Services |
The following table sets forth fees billed to us by our independent registered public accounting firm during the fiscal years ended December 31, 2011 and 2010 for: (i) services rendered for the audit of our annual financial statements and the review of our quarterly financial statements; (ii) services by our independent registered public accounting firm that are reasonably related to the performance of the audit or review of our financial statements and that are not reported as Audit Fees; (iii) services rendered in connection with tax compliance, tax advice and tax planning; and (iv) all other fees for services rendered.
| Year ended December 31, | ||||||||
| 2011 | 2010 | |||||||
| Audit Fees | $ | 252,000 | $ | 289,740 | ||||
| Audit Related Fees | $ | 10,000 | $ | 28,353 | ||||
| Tax Fees | $ | 44,448 | $ | 63,008 | ||||
| All Other Fees | $ | 36,000 | - | |||||
Policy on Audit Committee Pre-Approval of Audit and Permissible Non-Audit Services of Independent Auditors
Prior to entering into the engagement letter with our independent registered accountants, our Audit Committee approved the 2011 audit fees. For fiscal year 2012, our Audit Committee has approved fees for certain permissible non-audit services to be rendered by our independent registered accounting firm.
| 93 |
PART IV
| Item 15. | Exhibits and Financial Statement Schedules |
The following documents are filed as part of this Annual Report on Form 10-K:
1. Financial Statements. The following Consolidated Financial Statements of Protalix BioTherapeutics, Inc. are included in Item 8 of this Annual Report on Form 10-K:
| Page | ||
| Report of Independent Registered Public Accounting Firm | F-2 | |
| Consolidated Balance Sheets as of December 31, 2010 and 2011 | F-3 | |
| Consolidated Statements of Operations for the years ended December 31, 2009, 2010 and 2011 | F-4 | |
| Consolidated Statements of Changes in Shareholders’ Equity (Capital Deficiency) for the years ended December 31, 2009, 2010 and 2011 | F-5 | |
| Consolidated Statements of Cash Flows for the years ended December 31, 2009, 2010 and 2011 | F-6 | |
| Notes to Consolidated Financial Statements | F-8 |
2. Financial Statement Schedule. Financial statement schedules have been omitted since they are either not required, are not applicable or the required information is shown in the consolidated financial statements or related notes.
3. Exhibits.
| Incorporated by Reference | ||||||||||||
| Exhibit | File | Filed | ||||||||||
| Number | Exhibit Description | Form | Number | Exhibit | Date | Herewith | ||||||
| 3.1 | Amended and Restated Articles of Incorporation of the Company | S-4 | 333-48677 | 3.4 | March 26, 1998 | |||||||
| 3.2 | Article of Amendment to Articles of Incorporation dated June 9, 2006 | 8-A | 001-33357 | 3.2 | March 9, 2007 | |||||||
| 3.3 | Article of Amendment to Articles of Incorporation dated December 13, 2006 | 8-A | 001-33357 | 3.3 | March 9, 2007 | |||||||
| 3.4 | Article of Amendment to Articles of Incorporation dated December 26, 2006 | 8-A | 001-33357 | 3.4 | March 9, 2007 | |||||||
| 3.5 | Article of Amendment to Articles of Incorporation dated February 26, 2007 | 8-A | 001-33357 | 3.5 | March 9, 2007 | |||||||
| 3.6 | Amended and Restated Bylaws of the Company | 10-Q | 001-33357 | 3.6 | August 8, 2008 | |||||||
| 10.1 | 2006 Stock Incentive Plan | 10-K | 001-33357 | 10.1 | July 13, 2007 | |||||||
| 10.2 | Employment Agreement between Protalix Ltd. and Yoseph Shaaltiel, dated as of September 1, 2004 | 8-K | 001-33357 | 10.3 | January 8, 2007 | |||||||
| 94 |
| 10.3 | Employment Agreement between Protalix Ltd. and Einat Almon, dated as of December 19, 2004 | 8-K | 001-33357 | 10.3 | January 8, 2007 | |||||||
| 10.4 | Employment Agreement between Protalix Ltd. and David Aviezer, dated as of September 11, 2006 | 8-K | 001-33357 | 10.4 | January 8, 2007 | |||||||
| 10.5 | Employment Agreement between Protalix Ltd. and Yossi Maimon, dated as of October 15, 2006 | 8-K | 001-33357 | 10.5 | January 8, 2007 | |||||||
| 10.6† | License Agreement entered into as of April 12, 2005, by and between Icon Genetics AG and Protalix Ltd. | 8-K/A | 001-33357 | 10.6 | September 20, 2007 | |||||||
| 10.7† | Research and License Agreement between Yeda Research and Development Company Limited and Protalix Ltd. dated as of March 15, 2006 | 8-K/A | 001-33357 | 10.7 | September 20, 2007 | |||||||
| 10.8† | Agreement between Teva Pharmaceutical Industries Ltd. and Protalix Ltd., dated September 14, 2006 | 8-K/A | 001-33357 | 10.8 | September 20, 2007 | |||||||
| 10.9 | Lease Agreement between Protalix Ltd. and Angel Science Park (99) Ltd., dated as of October 28, 2003 as amended on April 18, 2005 | 8-K | 001-33357 | 10.9 | January 8, 2007 | |||||||
| 10.10 | Merger Agreement and Plan of Reorganization made and entered into as of August 21, 2006, by and among the Company, Protalix Acquisition Co., Ltd. and Protalix Ltd. | 8-K | 001-33357 | 10.10 | January 8, 2007 | |||||||
| 10.11 | Stock Option Award Agreement grant by and between the Company and Steven Rubin, dated as of December 31, 2006 | 10-K | 001-33357 | 10.13 | March 30, 2007 | |||||||
| 10.12 | First Amendment to the December 31, 2006 Stock Option Award Agreement by and between the Company and Steven Rubin, effective as of February 28, 2007 | 10-K | 001-33357 | 10.16 | March 30, 2007 | |||||||
| 10.13 | Scientific Advisory Board Agreement dated August 5, 2007 by and between the Company and Aaron Ciechanover, M.D. | 8-K | 001-33357 | 10.1 | August 6, 2007 | |||||||
| 10.14† | Research and License Agreement made on August 8, 2007, by and between Yissum Research Development Company of Jerusalem, the Boyce Thompson Institute and Protalix Ltd. | 10-Q | 001-33357 | 10.1 | November 14, 2007 |
| 95 |
| 10.15 | Unprotected Lease Agreement | 10-K | 001-33357 | 10.21 | March 17, 2008 | |||||||
| 10.16† | Exclusive License and Supply Agreement dated as of November 30, 2009 between Protalix Ltd. and Pfizer Inc. | 10-K | 001-33357 | 10.16 | February 26, 2010 | |||||||
| 10.17 | Employment Agreement by and between the Company and Sandra Lauterbach dated as of December 17, 2009 | 8-K | 001-33357 | 10.1 | December 27, 2009 | |||||||
| 10.18 | Employment Agreement by and between Protalix Ltd., and Tzvi Palash dated as of August 29, 2010 | 8-K | 001-33357 | 10.1 | September 7, 2010 | |||||||
| 10.19† | License Agreement between Protalix BioTherapeutics Ltd. and Virginia Tech Intellectual Properties, Inc. | 10-Q | 001-33357 | 10.2 | November 8, 2010 | |||||||
| 21.1 | Subsidiaries | 10-K | 001-33357 | 21.1 | February 26, 2010 | |||||||
| 23.1 | Consent of Kesselman & Kesselman, Certified Public Accountant (Isr.), A member of PricewaterhouseCoopers International Limited, independent registered public accounting firm for the Registrant | X | ||||||||||
| 31.1 | Certification of Chief Executive Officer pursuant to Rule 13a-14(a) as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 | X | ||||||||||
| 31.2 | Certification of Chief Financial Officer pursuant to Rule 13a-14(a) as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 | X | ||||||||||
| 32.1 | 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, Certification of Chief Executive Officer | X | ||||||||||
| 32.2 | 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, Certification of Chief Financial Officer | X | ||||||||||
| 101.INS | XBRL INSTANCE FILE | X |
| 96 |
| 101.SCH | XBRL SHEMA FILE | X | ||||||||||
| 101.CAL | XBRL CALCULATION FILE | X | ||||||||||
| 101.DEF | XBRL DEFINITION FILE | X | ||||||||||
| 101.LAB | XBRL LABEL FILE | X | ||||||||||
| 101.PRE | XBRL PRESENTATION FILE | X |
† Portions of this exhibit were omitted and have been filed separately with the Secretary of the Securities and Exchange Commission pursuant to the Registrant’s application requesting confidential treatment under Rule 24b-2 of the Exchange Act.
| 97 |
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized, as of February 27, 2012.
| PROTALIX BIOTHERAPEUTICS, INC. | |
| By: | /s/ David Aviezer |
| David Aviezer, Ph.D. | |
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints David Aviezer, Ph.D. and Yossi Maimon, and each of them, as his true and lawful attorneys-in-fact and agents, with full power of substitution and re-substitution, for him and in his name, place and stead, in any and all capacities, to sign any and all amendments to this Annual Report on Form 10-K, and to file the same, with all exhibits thereto, and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, and each of them, full power and authority to do and perform each and every act and thing requisite and necessary to be done in connection therewith, as fully to all intents and purposes as he might or could do in person, hereby ratifying and confirming that said attorneys-in-fact and agents, or any of them, or their or his substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the Registrant and in the capacities and on the dates indicated.
| Signature | Title | Date | ||
| /s/ David Aviezer | President, Chief Executive Officer | February 27, 2012 | ||
| David Aviezer, Ph.D. | (Principal Executive Officer) and Director | |||
| /s/ Yossi Maimon | Chief Financial Officer, Treasurer and | February 27, 2012 | ||
| Yossi Maimon | Secretary (Principal Financial | |||
| and Accounting Officer) | ||||
| /s/ Yoseph Shaaltiel | Executive VP, Research and Development | February 27, 2012 | ||
| Yoseph Shaaltiel, Ph.D. | and Director | |||
| /s/ Alfred Akirov | Director | February 27, 2012 | ||
| Alfred Akirov | ||||
| /s/ Amos Bar Shalev | Director | February 27, 2012 | ||
| Amos Bar Shalev | ||||
| /s/ Zeev Bronfeld | Director | February 27, 2012 | ||
| Zeev Bronfeld | ||||
| /s/ Yodfat Harel | Director | February 27, 2012 | ||
| Yodfat Harel | ||||
| /s/ Eyal Sheratzky | Director | February 27, 2012 | ||
| Eyal Sheratzky |
| 98 |
PROTALIX BIOTHERAPEUTICS, INC.
CONSOLIDATED FINANCIAL STATEMENTS
TABLE OF CONTENTS
| Page | ||
| REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM | F-2 | |
| CONSOLIDATED FINANCIAL STATEMENTS | ||
| Consolidated Balance Sheets as of December 31, 2010 and 2011 | F-3 | |
| Consolidated Statements of Operations for the years ended December 31, 2009, 2010 and 2011 | F-4 | |
| Consolidated Statements of Changes in Shareholders’ Equity (Capital Deficiency) for the years ended December 31, 2009, 2010 and 2011 | F-5 | |
| Consolidated Statements of Cash Flows for the years ended December 31, 2009, 2010 and 2011 | F-6 | |
| Notes to Consolidated Financial Statements | F-8 |
The dollar amounts are stated in U.S. dollars ($)
| F-1 |

REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the shareholders of
PROTALIX BIOTHERAPEUTICS, INC.
In our opinion, the accompanying consolidated balance sheets and the related statements of operations, changes in shareholders’ equity (capital deficiency) and cash flows present fairly, in all material respects, the financial position of Protalix BioTherapeutics, Inc. and its subsidiaries at December 31, 2010 and 2011, and the results of their operations and their cash flows for each of the three years in the period ended December 31, 2011, in conformity with accounting principles generally accepted in the United States of America. Also, in our opinion, the Company maintained, in all material respects, effective internal control over financial reporting as of December 31, 2011, based on criteria established in Internal Control - Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). The Company’s management is responsible for these financial statements, for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting, included in the accompanying “Management Report on Internal Control over Financial Reporting” appearing under Item 9A. Our responsibility is to express opinions on these financial statements and on the Company’s internal control over financial reporting based on our integrated audits. We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the financial statements are free of material misstatement and whether effective internal control over financial reporting was maintained in all material respects. Our audits of the financial statements included examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, and evaluating the overall financial statement presentation. Our audit of internal control over financial reporting included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audits also included performing such other procedures as we considered necessary in the circumstances. We believe that our audits provide a reasonable basis for our opinions.
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that: (i) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
| Tel-Aviv, Israel | /s/ Kesselman & Kesselman |
| February 27, 2012 | Kesselman & Kesselman |
| Certified Public Accountant (Isr.) | |
| A member firm of PricewaterhouseCoopers | |
| International Limited |

Kesselman & Kesselman, Trade Tower, 25 Hamered Street, Tel-Aviv 68125, Israel, P.O Box 452 Tel-Aviv 61003 Telephone: +972 -3- 7954555, Fax:+972 -3- 7954556, www.pwc.co.il
| F-2 |
PROTALIX BIOTHERAPEUTICS, INC.
CONSOLIDATED BALANCE SHEETS
(U.S. dollars in thousands, except shares and per share amounts)
| December 31, | ||||||||
| 2010 | 2011 | |||||||
| ASSETS | ||||||||
| CURRENT ASSETS: | ||||||||
| Cash and cash equivalents | $ | 35,900 | $ | 27,001 | ||||
| Accounts receivable: | ||||||||
| Trade | 7,013 | 1,374 | ||||||
| Other | 2,231 | 3,837 | ||||||
| Inventories | 1,189 | 279 | ||||||
| Total current assets | 46,333 | 32,491 | ||||||
| FUNDS IN RESPECT OF EMPLOYEE RIGHTS UPON RETIREMENT | 942 | 1,043 | ||||||
| PROPERTY AND EQUIPMENT, NET | 17,454 | 18,271 | ||||||
| Total assets | $ | 64,729 | $ | 51,805 | ||||
| LIABILITIES NET OF CAPITAL DEFICIENCY | ||||||||
| CURRENT LIABILITIES: | ||||||||
| Accounts payable and accruals: | ||||||||
| Trade | $ | 6,272 | $ | 5,032 | ||||
| Other | 8,068 | 7,540 | ||||||
| Deferred revenues | 4,563 | 6,121 | ||||||
| Total current liabilities | 18,903 | 18,693 | ||||||
| LONG TERM LIABILITIES: | ||||||||
| Deferred revenues | 55,486 | 50,923 | ||||||
| Long term liability | 6,566 | |||||||
| Liability for employee rights upon retirement | 1,663 | 1,700 | ||||||
| Total long term liabilities | 57,149 | 59,189 | ||||||
| Total liabilities | 76,052 | 77,882 | ||||||
| COMMITMENTS | ||||||||
| CAPITAL DEFICIENCY: | ||||||||
| Common Stock, $0.001 par value: | ||||||||
| Authorized - as of December 31, 2010 and 2011, | ||||||||
| 150,000,000 shares; issued and outstanding - as of December 31, 2010 and 2011, 81,248,472 and 85,630,157 shares, respectively | 81 | 86 | ||||||
| Additional paid-in capital | 124,044 | 145,814 | ||||||
| Accumulated deficit | (135,448 | ) | (171,977 | ) | ||||
| Total capital deficiency | (11,323 | ) | (26,077 | ) | ||||
| Total liabilities net of capital deficiency | $ | 64,729 | $ | 51,805 | ||||
The accompanying notes are an integral part of the consolidated financial statements.
| F-3 |
PROTALIX BIOTHERAPEUTICS, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS
(U.S. dollars in thousands, except shares and per share amounts)
| Year ended December 31, | ||||||||||||
| 2009 | 2010 | 2011 | ||||||||||
| REVENUES | $ | 388 | $ | 6,642 | $ | 8,386 | ||||||
| COMPANY’S SHARE IN COLLABORATION AGREEMENT | 4,602 | (5,418 | ) | |||||||||
| COST OF REVENUES | (3,575 | ) | (4,383 | ) | (1,525 | ) | ||||||
| GROSS PROFIT (LOSS) | (3,187 | ) | 6,861 | 1,443 | ||||||||
| RESEARCH AND DEVELOPMENT EXPENSES | (27,390 | ) | (37,691 | ) | (37,818 | ) | ||||||
| Less – grants and reimbursements | 5,752 | 7,740 | 6,775 | |||||||||
| RESEARCH AND DEVELOPMENT EXPENSES, NET | (21,638 | ) | (29,951 | ) | (31,043 | ) | ||||||
| GENERAL AND ADMINISTRATIVE EXPENSES | (7,144 | ) | (6,876 | ) | (6,931 | ) | ||||||
| OPERATING LOSS | (31,969 | ) | (29,966 | ) | (36,531 | ) | ||||||
| FINANCIAL INCOME – NET | 529 | 968 | 2 | |||||||||
| NET LOSS FOR THE YEAR | $ | (31,440 | ) | $ | (28,998 | ) | $ | (36,529 | ) | |||
| Net loss per share of common stock – basic and diluted: | $ | 0.41 | $ | 0.36 | $ | 0.43 | ||||||
| Weighted average number of shares of common stock used in computing loss per share of common stock, basic and diluted: | 76,942,840 | 80,960,300 | 84,645,364 | |||||||||
The accompanying notes are an integral part of the consolidated financial statements.
| F-4 |
PROTALIX BIOTHERAPEUTICS, INC.
CONSOLIDATED STATEMENT OF CHANGES IN SHAREHOLDERS’ EQUITY
(CAPITAL DEFICIENCY)
(U.S. dollars in thousands, except share data)
| Additional | ||||||||||||||||||||
| Common | Common | paid–in | Accumulated | |||||||||||||||||
| Stock | Stock | Capital | deficit | Total | ||||||||||||||||
| Number of shares | Amount | |||||||||||||||||||
| Balance at January 1, 2009 | 75,938,059 | 76 | 119,281 | (75,010 | ) | 44,347 | ||||||||||||||
| Changes during 2009: | ||||||||||||||||||||
| Share-based compensation | 2,683 | - | 2,683 | |||||||||||||||||
| Exercise of options granted to employees and non-employees (includes net exercise) | 4,903,178 | 5 | 288 | - | 293 | |||||||||||||||
| Net Loss | (31,440 | ) | (31,440 | ) | ||||||||||||||||
| Balance at December 31, 2009 | 80,841,237 | 81 | 122,252 | (106,450 | ) | 15,883 | ||||||||||||||
| Changes during 2010: | ||||||||||||||||||||
| Share-based compensation | 1,282 | 1,282 | ||||||||||||||||||
| Exercise of options granted to employees and non-employees (includes net exercise) | 407,235 | * | 510 | 510 | ||||||||||||||||
| Net Loss | (28,998 | ) | (28,998 | ) | ||||||||||||||||
| Balance at December 31, 2010 | 81,248,472 | 81 | 124,044 | (135,448 | ) | (11,323 | ) | |||||||||||||
| Changes during 2011: | ||||||||||||||||||||
| Common stock issued for cash (net of issuance costs of $1,410) (see note 7c) | 4,000,000 | 4 | 20,586 | 20,590 | ||||||||||||||||
| Share-based compensation | 886 | 886 | ||||||||||||||||||
| Exercise of options granted to employees and non-employees (includes net exercise) | 381,685 | 1 | 298 | 299 | ||||||||||||||||
| Net Loss | (36,529 | ) | (36,529 | ) | ||||||||||||||||
| Balance at December 31, 2011 | 85,630,157 | 86 | 145,814 | (171,977 | ) | (26,077 | ) | |||||||||||||
* Represents an amount less than $1.
The accompanying notes are an integral part of the consolidated financial statements.
| F-5 |
PROTALIX BIOTHERAPEUTICS, INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(U.S. dollars in thousands)
| Year ended December 31, | ||||||||||||
| 2009 | 2010 | 2011 | ||||||||||
| CASH FLOWS FROM OPERATING ACTIVITIES: | ||||||||||||
| Net Loss | $ | (31,440 | ) | $ | (28,998 | ) | $ | (36,529 | ) | |||
| Adjustments required to reconcile net loss to net cash provided by (used in) operating activities: | ||||||||||||
| Share based compensation | 2,683 | 1,282 | 886 | |||||||||
| Depreciation and write down of fixed assets | 1,990 | 3,133 | 3,631 | |||||||||
| Financial expenses (income), net (mainly exchange differences) | (166 | ) | (591 | ) | 350 | |||||||
| Changes in accrued liability for employee rights upon retirement | 265 | 377 | 155 | |||||||||
| Gain on amounts funded in respect of employee rights upon retirement | (81 | ) | (26 | ) | (14 | ) | ||||||
| Loss on sale of fixed assets | 29 | 8 | ||||||||||
| Changes in operating assets and liabilities: | ||||||||||||
| Increase (decrease) in deferred revenues (including non-current portion) | 64,612 | (4,563 | ) | (3,005 | ) | |||||||
| Decrease (increase) in accounts receivable | (1,224 | ) | (6,860 | ) | 3,891 | |||||||
| Decrease (Increase) in inventories | (1,189 | ) | 910 | |||||||||
| Increase (decrease) in accounts payable and accruals (including long term ) | 7,784 | (1,029 | ) | 6,147 | ||||||||
| Net cash provided by (used in) operating activities | $ | 44,452 | $ | (38,464 | ) | $ | (23,570 | ) | ||||
| CASH FLOWS FROM INVESTING ACTIVITIES: | ||||||||||||
| Purchase of property and equipment | $ | (6,195 | ) | $ | (7,855 | ) | $ | (5,705 | ) | |||
| Proceeds from sale of property and equipment | 73 | 2 | ||||||||||
| Amounts funded in respect of employee rights upon retirement, net | (52 | ) | (137 | ) | (165 | ) | ||||||
| Net cash used in investing activities | $ | (6,174 | ) | $ | (7,992 | ) | $ | (5,868 | ) | |||
| CASH FLOWS FROM FINANCING ACTIVITIES: | ||||||||||||
| Issuance of shares, net of issuance cost | 20,650 | |||||||||||
| Exercise of options | $ | 293 | $ | 501 | $ | 277 | ||||||
| Net cash provided by financing activities | $ | 293 | $ | 501 | $ | 20,927 | ||||||
| EFFECT OF EXCHANGE RATE CHANGES ON CASH | 99 | 589 | (388 | ) | ||||||||
| NET INCREASE (DECREASE) IN CASH AND CASH EQUIVALENTS | 38,670 | (45,366 | ) | (8,899 | ) | |||||||
| BALANCE OF CASH AND CASH EQUIVALENTS AT BEGINNING OF YEAR | 42,596 | 81,266 | 35,900 | |||||||||
| BALANCE OF CASH AND CASH EQUIVALENTS AT END OF YEAR | $ | 81,266 | $ | 35,900 | $ | 27,001 | ||||||
The accompanying notes are an integral part of the consolidated financial statements.
| F-6 |
PROTALIX BIOTHERAPEUTICS, INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(U.S. dollars in thousands)
(CONTINUED)
| Year ended December 31, | ||||||||||||
| 2009 | 2010 | 2011 | ||||||||||
| SUPPLEMENTARY INFORMATION ON INVESTING AND FINANCING ACTIVITIES NOT INVOLVING CASH FLOWS: | ||||||||||||
| Purchase of property and equipment | $ | 4,525 | $ | 2,720 | $ | 1,473 | ||||||
| Issuance cost not yet paid and accruals – other | $ | 60 | ||||||||||
| Exercise of options granted to employees | $ | 9 | $ | 31 | ||||||||
The accompanying notes are an integral part of the consolidated financial statements.
| F-7 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 1 - SIGNIFICANT ACCOUNTING POLICIES
| a. | General |
| 1. | Operation |
Protalix BioTherapeutics, Inc. (collectively with its subsidiaries, “The Company”), and its wholly-owned subsidiary, Protalix Ltd. (the “Israeli Subsidiary” or “Protalix Ltd.”), are biopharmaceutical companies focused on the development and commercialization of recombinant therapeutic proteins based on the Company’s proprietary ProCellEx® protein expression system (“ProCellEx”). In September 2009, Protalix Ltd. formed another wholly-owned subsidiary under the laws of the Netherlands in connection with the European Medicines Agency ("EMA") application process in Europe. The Company’s two subsidiaries are referred to collectively herein as the “Subsidiaries.” The Company’s lead product development candidate is taliglucerase alfa for the treatment of Gaucher disease which the Company is developing using ProCellEx. In addition to taliglucerase alfa, the Company is developing certain other products using ProCellEx.
In September 2009, the Company successfully completed its phase III pivotal trial of taliglucerase alfa. In July 2010, the U.S. Food and Drug Administration (“FDA”) notified the Company that it had accepted for review the Company’s new drug application ("NDA") for taliglucerase alfa for the treatment of Gaucher disease and that it granted to taliglucerase alfa a Prescription Drug User Fee Act (PDUFA) action date of February 25, 2011. On February 25, 2011, the FDA issued a Complete Response Letter ("CRL") indicating that the FDA’s review of the taliglucerase alfa NDA was complete but questions remain that precluded the approval of the NDA in its then current form. In August 2011, the Company submitted a revised NDA in response to the CRL. During the same month, the FDA notified the Company that it had accepted the revised NDA for review, had deemed the resubmission a class 2, or 6-month, response and established February 1, 2012 as the new PDUFA date. In December 2011, the FDA notified the Company that it had decided to extend the PDUFA date to May 1, 2012.
In the fourth quarter of 2010, Pfizer Inc. ("Pfizer"), the Company's commercialization partner, filed, with the Company's cooperation, a Marketing Authorization Application ("MAA") for taliglucerase alfa with the EMA. As part of its ongoing review of the MAA, the EMA delivered a list of outstanding points to be addressed by the applicant. Among the topics currently in discussion, is the orphan drug designation and exclusivity granted by the EMA/European Commission to VPRIV, Shire plc's ("Shire") Gaucher disease treatment, which could prevent the marketing authorization of taliglucerase alfa in the European Union for a 10-year market exclusivity period commencing as of the August 2010 marketing authorization of VPRIV in the European Union. As part of the MAA procedure, Pfizer, with the Company's cooperation, is challenging VPRIV's orphan market protection with respect to taliglucerase alfa pursuant to the EU orphan drug regulation. The EU orphan drug regulation provides for the possibility of such a challenge, and for an exception to this exclusivity to be granted, based on a number of factors, including contribution to patient care, clinical, supply, capacity and others.
Following the submission of the taliglucerase alfa MAA to the EMA, a good manufacturing practices, or GMP, audit of the Company's manufacturing facility was performed and it was found compliant with EMA GMP in December 2011.
In January 2010, the EMA's Committee for Orphan Medicinal Products, after reviewing all relevant clinical data, recommended that the European Commission grant orphan drug designation to taliglucerase alfa for the treatment of Gaucher disease and in March 2010, the European Commission granted orphan designation to taliglucerase alfa. In September 2009, the FDA’s Office of Orphan Product Development granted taliglucerase alfa Orphan Drug Status.
| F-8 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 1 - SIGNIFICANT ACCOUNTING POLICIES (continued):
On November 30, 2009, Protalix Ltd. and Pfizer entered into an Exclusive License and Supply Agreement (the “Pfizer Agreement”) pursuant to which Protalix Ltd. granted Pfizer an exclusive, worldwide license to develop and commercialize taliglucerase alfa, except in Israel. Under the terms and conditions of the Pfizer Agreement, Protalix Ltd. retained the right to commercialize taliglucerase alfa in Israel. See Note 2.
Although taliglucerase alfa has not yet been approved for commercial sale, patients are being treated with taliglucerase alfa, both in the framework of the Company's clinical trials and related studies and in compassionate use programs, special access agreements, named patient provisions and other programs designed to ensure that treatments are available to Gaucher patients in light of recent shortages of approved treatments. In July 2009, following a request by the FDA, the Company submitted a treatment protocol to the FDA in order to address an expected shortage of the current enzyme replacement therapy approved for Gaucher disease. The treatment protocol was approved by the FDA in August 2009, and the Company is continuing to treat patients in the United States under this protocol. On July 13, 2010, the Company announced that the French regulatory authority had granted an Autorisation Temporaire d'Utilisation (ATU), or Temporary Authorization for Use, for taliglucerase alfa for the treatment of Gaucher disease. An ATU is the regulatory mechanism used by the French Health Products and Safety Agency to make non-approved drugs available to patients in France when a genuine public health need exists. This ATU allows Gaucher disease patients in France to receive treatment with taliglucerase alfa before marketing authorization for the product is granted in the European Union. Payment for taliglucerase alfa has been secured through government allocations to hospitals. In addition to the United States and France, taliglucerase alfa is currently being provided to Gaucher disease patients under special access agreements or Named Patient provisions in Brazil and in other countries.
On August 10, 2010, Pfizer entered into a short-term supply agreement with the Ministry of Health of Brazil pursuant to which the Company and Pfizer have provided taliglucerase alfa to the Ministry of Health of Brazil for the treatment of Gaucher disease patients. During the remainder of 2010 and the first quarter of 2011, the Company and Pfizer completed the supply of products deliverable under the short-term supply agreement. During 2011, Pfizer recorded an allowance for sales returns in connection with the supply agreement because the Brazilian Ministry of Health requested that Pfizer consider the replacement of certain vials that might expire during 2012. Revenue, net of allowance for sales returns, generated from the Brazilian Ministry of Health was recorded by Pfizer, and the Company recorded its share of such revenues in accordance with the terms and conditions of the Pfizer Agreement.
| 2. | Liquidity and Financial Resources |
Successful completion of the Company’s development programs and its transition to normal operations is dependent upon obtaining necessary regulatory approvals from the FDA prior to selling its products within the United States, and foreign regulatory approvals must be obtained to sell its products internationally. Each FDA and EMA approval triggers a $25,000 milestone payment from Pfizer, pursuant to the Pfizer Agreement. There can be no assurance that the Company will receive regulatory approval of any of its product candidates, and a substantial amount of time may pass before the Company achieves a level of revenues adequate to support its operations, if at all. The Company also expects to incur substantial expenditures in connection with the regulatory approval process for each of its product candidates during their respective developmental periods. Obtaining marketing approval will be directly dependent on the Company’s ability to implement the necessary regulatory steps required to obtain marketing approval in the United States and in other countries. The Company cannot predict the outcome of these activities.
| F-9 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 1 - SIGNIFICANT ACCOUNTING POLICIES (continued):
Based on its current cash resources and commitments, and cash received in a public underwritten offering on February 2012 (see note 11b), the Company believes it will be able to maintain its current planned development activities and the corresponding level of expenditures for at least the next 12 months, although no assurance can be given that it will not need additional funds prior to such time. If there are unexpected increases in general and administrative expenses or research and development expenses, the Company may need to seek additional financing during the next 12 months.
| b. | Basis of presentation |
The Company’s financial statements have been prepared in accordance with generally accepted accounting principles in the United States (“U.S. GAAP”).
| c. | Use of estimates in the preparation of financial statements |
The preparation of financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities, the disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results may differ from those estimates.
| d. | Functional currency |
The dollar is the currency of the primary economic environment in which the operations of the Company and its Subsidiaries are conducted. The Company’s revenues are derived in dollars. Most of the Company’s expenses and capital expenditures are incurred in dollars, and the major source of the Company’s financing has been provided in dollars.
Transactions and balances originally denominated in dollars are presented at their original amounts. Balances in non-dollar currencies are translated into dollars using historical and current exchange rates for non-monetary and monetary balances, respectively. For non-dollar transactions and other items (stated below) reflected in the statements of operations, the following exchange rates are used: (i) for transactions – exchange rates at the transaction dates or average rates; and (ii) for other items (derived from non-monetary balance sheet items such as depreciation and amortization, etc.) – historical exchange rates. Currency transaction gains and losses are carried to financial income or expenses, as appropriate.
| e. | Cash equivalents |
The Company considers all short-term, highly liquid investments, which include short-term bank deposits with original maturities of three months or less from the date of purchase, that are not restricted as to withdrawal or use and are readily convertible to known amounts of cash, to be cash equivalents.
| f. | Inventories |
Inventories are valued at the lower of cost or market. Cost of raw and packaging materials and purchased products is determined using the “moving average” basis.
Manufacturing costs for product candidates are expensed as research and development expenses. The Company considers regulatory approval of product candidates to be uncertain, and product manufactured prior to regulatory approval may not be sold unless regulatory approval is obtained. As such, the manufacturing costs for product candidates incurred prior to regulatory approval are not capitalized as inventory. Inventories for which there are orders or are probable to be sold even without the regulatory approval, are capitalized.
| F-10 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 1 - SIGNIFICANT ACCOUNTING POLICIES (continued):
Cost of finished products that are capitalized is determined as follows: the value of the raw and packaging materials component is determined primarily on a using the “moving average” basis; the value of the labor and overhead component is determined on an average basis over the production period.
| g. | Property and equipment |
| 1. | Property and equipment are stated at cost, net of accumulated depreciation and amortization. |
| 2. | The Company’s assets are depreciated by the straight-line method on the basis of their estimated useful lives as follows: |
| Years | ||
| Laboratory equipment | 5 | |
| Furniture | 10-15 | |
| Computer equipment | 3 |
Leasehold improvements are amortized by the straight-line method over the expected lease term, which is shorter than the estimated useful life of the improvements.
| h. | Impairment in value of long-lived assets: |
The Company tests long-lived assets for impairment if an indication of impairment exists. If the sum of expected future cash flows of definite life assets (undiscounted and without interest charges) is less than the carrying amount of such assets, the Company recognizes an impairment loss, and writes down the assets to their estimated fair values, calculated based on expected future discounted cash flows.
| i. | Income taxes |
| 1. | Deferred income taxes |
Deferred taxes are determined utilizing the assets and liabilities method based on the estimated future tax effects of the differences between the financial accounting and tax bases of assets and liabilities under the applicable tax laws. Deferred tax balances are computed using the tax rates expected to be in effect when those differences reverse. A valuation allowance in respect of deferred tax assets is provided if, based upon the weight of available evidence, it is more likely than not that some or all of the deferred tax assets will not be realized. The Company has provided a full valuation allowance with respect to its deferred tax assets.
The guidance prohibits the recognition of deferred tax liabilities or assets that arise from differences between the financial reporting and tax bases of assets and liabilities that are measured from the local currency into dollars using historical exchange rates, and that result from changes in exchange rates or indexing for tax purposes. Consequently, the above mentioned differences with respect to Protalix Ltd. were not reflected in the computation of deferred tax assets and liabilities.
| 2. | Uncertainty in income taxes |
Tax benefits recognized in the financial statements are those that the Company’s management deems at least more likely than not to be sustained, based on technical merits. The amount of benefits recorded for these tax benefits is measured as the largest benefit the Company’s management deems more likely than not to be sustained.
| F-11 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 1 - SIGNIFICANT ACCOUNTING POLICIES (continued):
| j. | Revenue Recognition |
The Company recognizes revenue when the earnings process is complete, which is when revenue is realized or realizable and earned, there is persuasive evidence a revenue arrangement exists, delivery of goods or services has occurred, the sales price is fixed or determinable and collectability is reasonably assured.
| 1. | Revenues from the license and supply agreement with Pfizer |
The Company recognizes revenue and milestone payments received pursuant to the Pfizer Agreement in accordance with guidance regarding revenue recognition and accounting for revenue arrangements with multiple deliverables. As the arrangement with Pfizer requires the Company’s continued involvement with respect to the proposed commercialization of taliglucerase alfa, the non-refundable, up-front license payment the Company received from Pfizer was deferred and recognized over the related performance period. The Company estimated the performance period of 14 years (commencing upon the date of the Company's receipt of the up-front license payment payable by Pfizer under the Pfizer Agreement) based on the date the last relevant patent expires. See Note 2. The Company adjusts the performance periods, if appropriate, based on the applicable facts and circumstances.
| 2. | Revenues from selling products to Pfizer |
The Company recognizes revenues from products sold to Pfizer upon delivery, when the sales price is fixed or determinable and collectability is reasonably assured.
| 3. | Company’s share in the collaboration agreement |
Under the terms and conditions of the Pfizer Agreement, the Company is entitled to 40% of the profits or loss from sales of taliglucerase alfa, and related expenses incurred, except with respect to sales in Israel, where the Company retained exclusive marketing rights. Since Pfizer bears most of the risks and rewards relating to the agreement, the Company’s share in the profits and loss in the agreement is recognized on a net basis. The Company recognizes its share of net profit or loss from the Pfizer Agreement based on reports it receives from Pfizer summarizing the results of the collaborative activities under the agreement for the applicable period. Under the terms of the Pfizer Agreement, for its subsidiaries operating outside the United States, financial information is included based on the fiscal year ending November 30, while financial information for the U.S. entity is included based on the fiscal year ending December 31.
| k. | Research and development costs |
Research and development costs are expensed as incurred and consist primarily of personnel, subcontractors and consultants (mainly in connection with clinical trials), facilities, equipment and supplies for research and development activities. Grants received by the Israeli Subsidiary from the Office of the Chief Scientist of Israel’s Ministry of Industry, Trade and Labor (the “OCS”) are recognized when the grant becomes receivable, provided there is reasonable assurance that the Company or the Subsidiary will comply with the conditions attached to the grant and there is reasonable assurance the grant will be received. The grant is deducted from the research and development expenses as the applicable costs are incurred.
Reimbursements received from Pfizer are recognized when the reimbursements become receivable, provided there is reasonable assurance that the Company will comply with the conditions attached to the reimbursements and there is reasonable assurance the reimbursements will be received. The reimbursements are deducted from the research and development expenses as the applicable costs are incurred.
| F-12 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 1 - SIGNIFICANT ACCOUNTING POLICIES (continued):
In connection with purchases of assets, amounts assigned to intangible assets to be used in a particular research and development project that have no alternative future use are charged to research and development costs at the purchase date.
Nonrefundable advance payments for goods or services that will be used or rendered for future research and development activities are deferred and amortized over the period that the goods are consumed or the related services are performed.
| l. | Comprehensive loss |
The Company has no other comprehensive loss components other than net loss for the reported periods.
| m. | Concentration of credit risks and trade receivable |
Financial instruments that potentially subject the Company to concentration of credit risk consist principally of bank deposits. The Company deposits these instruments with highly rated financial institutions, mainly in Israeli banks, and, as a matter of policy, limits the amounts of credit exposure to any one financial institution. The Company has not experienced any credit losses in these accounts and does not believe it is exposed to any significant credit risk on these instruments.
The Company's trade receivable represents amounts to be received from Pfizer, as the Company currently receives all of its revenues from Pfizer. The Company does not require Pfizer to post collateral with respect to the receivables. The Company performs periodic credit evaluations of Pfizer’s financial condition and believes there is no significant risk with respect to Pfizer’s payment of the receivables.
| n. | Share-based compensation |
The Company accounts for employee’s share-based payment awards classified as equity awards using the grant-date fair value method. The fair value of share-based payment transactions is recognized as an expense over the requisite service period, net of estimated forfeitures. The Company estimates forfeitures based on historical experience and anticipated future conditions.
The Company elected to recognize compensation cost for an award with only service conditions that has a graded vesting schedule using the accelerated method based on the multiple-option award approach.
When stock options are granted as consideration for services provided by consultants and other non-employees, the grant is accounted for based on the fair value of the consideration received or the fair value of the stock options issued, whichever is more reliably measurable. The fair value of the options granted is measured on a final basis at the end of the related service period and is recognized over the related service period using the straight-line method.
| o. | Net Loss per share |
Basic and diluted loss per share (“LPS”) are computed by dividing net loss by the weighted average number of shares of the Company's Common Stock, par value $0.001 per share (the "Common Stock") outstanding for each period.
Diluted LPS does not include options to purchase 10,660,447, 7,761,168 and 7,469,088 shares of Common Stock for the fiscal years ended December 31, 2009, 2010 and 2011, respectively, because the effect would be anti-dilutive.
| F-13 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 2 - LICENSE AND SUPPLY AGREEMENT
On November 30, 2009, Protalix Ltd. and Pfizer entered into the Pfizer Agreement pursuant to which Pfizer was granted an exclusive, worldwide license to develop and commercialize taliglucerase alfa, except in Israel. Under the terms and conditions of the Pfizer Agreement, Protalix Ltd. retained the right to commercialize taliglucerase alfa in Israel. Under the Pfizer Agreement, Pfizer made an upfront payment to Protalix Ltd. of $60,000 in connection with the execution of the agreement and shortly thereafter paid Protalix Ltd. an additional $5,000 upon the Company’s filing of a proposed pediatric investigation plan to the Pediatric Committee of the EMA. Protalix Ltd. is also eligible to receive additional potential milestone payments totaling up to $50,000 for the successful achievement of other regulatory milestones. Protalix Ltd. is entitled to 40% of the profits earned on Pfizer’s sales of taliglucerase alfa. Such profit will be calculated while, in addition to other adjustments, taking into account Protalix Ltd.’s cost of goods sold and Pfizer’s commercial expenses, with certain expenses capped or borne solely by one party ("Collaboration Operation"). Of the losses incurred by the Collaboration Operation through December 31, 2011, 40% will be deducted from the Company's share in the profits from future years. This deduction will limited to a certain percentage of any quarterly profit. As of December 31, 2011, the Company accrued a liability in respect of these losses equal to $7,888, out of which $6,566 is classified as a long term liability, and the remainder is included in accounts payable and accruals-other.
The Company has determined that the initial, non-refundable upfront license fee payment of $60,000 together with the first $5,000 payment will be recognized on a straight line basis as revenue over the estimated relationship period. The Company has estimated that the relationship period for its arrangement with Pfizer will be approximately 14 years (commencing upon the Company’s receipt of the up-front license payment) based on the Company’s last material patent relating to taliglucerase alfa to expire.
The Company’s deliverables under this collaboration include an exclusive license to taliglucerase alfa as an enzyme replacement therapy for the treatment of Gaucher disease, certain research and development services as required under the Pfizer Agreement for taliglucerase alfa, manufacturing of taliglucerase alfa and optional participation in a joint steering committee.
According to the terms and conditions of the Pfizer Agreement, the Company retained manufacturing rights and sells its products to Pfizer. In addition, Pfizer is required to reimburse the Company for certain costs it incurs in connection with certain development expenses for taliglucerase alfa. In connection with the payments received under the Pfizer Agreement, Protalix Ltd. is obligated to pay certain royalties. See Note 6a.
NOTE 3 - PROPERTY AND EQUIPMENT
| a. | Composition of property and equipment grouped by major classifications is as follows: |
| December 31, | ||||||||
| 2010 | 2011 | |||||||
| Laboratory equipment | $ | 11,478 | $ | 13,079 | ||||
| Furniture and computer equipment | 1,306 | 1,522 | ||||||
| Leasehold improvements | 11,221 | 13,705 | ||||||
| Equipment under construction | 1,038 | 1,113 | ||||||
| $ | 25,043 | $ | 29,419 | |||||
| Less – accumulated depreciation and amortization | (7,589 | ) | (11,148 | ) | ||||
| $ | 17,454 | $ | 18,271 | |||||
| b. | Depreciation and amortization in respect of property and equipment totaled $1,990, $3,133 and $3,631 for the years ended December 31, 2009, 2010 and 2011, respectively, out of which the Company recorded a total write down of $0, $43 and $10, respectively. |
| F-14 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 4 - INVENTORIES
| a. | Inventory at December 31, 2010 and 2011 consisted of the following: |
| December 31, | ||||||||
| 2010 | 2011 | |||||||
| Raw materials | $ | 553 | $ | 279 | ||||
| Finished goods | 636 | - | ||||||
| Total inventory | $ | 1,189 | $ | 279 | ||||
| b. | During the year ended December 31, 2010 and 2011, the Company recorded a $2,692 and $363, respectively, write-down of inventory under cost of revenues. See Note 9d. |
NOTE 5 - LIABILITY FOR EMPLOYEE RIGHTS UPON RETIREMENT
The Israeli Subsidiary is required to make a severance payment upon dismissal of an employee, or upon termination of employment in certain circumstances. The severance pay liability to the employees (based upon length of service and the latest monthly salary - one month’s salary for each year employed) is recorded on the Company’s balance sheets under “Liability for employee rights upon retirement.” The liability is recorded as if it were payable at each balance sheet date on an undiscounted basis.
The liability is funded in part from the purchase of insurance policies or by the establishment of pension funds with dedicated deposits in the funds. The amounts used to fund these liabilities are included in the Company’s balance sheets under “Funds in respect of employee rights upon retirement.” These policies are the Company’s assets. However, under labor agreements and subject to certain limitations, any policy may be transferred to the ownership of the individual employee for whose benefit the funds were deposited. In the years ended December 31, 2009, 2010 and 2011, the Company deposited $79, $174 and $181, respectively, with insurance companies in connection with its severance payment obligations.
In accordance with the current employment agreements with certain employees, the Company makes regular deposits with certain insurance companies for accounts controlled by each applicable employee in order to secure the employee’s rights upon retirement. The Company is fully relieved from any severance pay liability with respect to each such employee after it makes the payments on behalf of the employee. The liability accrued in respect of these employees and the amounts funded, as of the respective agreement dates, are not reflected in the Company’s balance sheets, as the amounts funded are not under the control and management of the Company and the pension or severance pay risks have been irrevocably transferred to the applicable insurance companies (the “Contribution Plans”).
The amounts of severance pay expenses were $488, $1,118 and $816 for the years ended December 31, 2009, 2010 and 2011, respectively, of which $335, $531 and $641 in the years ended December 31, 2009, 2010 and 2011, respectively, were in respect of a Contribution Plan. Gain on amounts funded in respect of employee rights upon retirement totaled $81, $26 and $14 for the years ended December 31, 2009, 2010 and 2011, respectively.
The Company expects to contribute approximately $779 in the year ending December 31, 2012 to insurance companies in connection with its severance liabilities for its operations for that year, $607 of which will be contributed to one or more Contribution Plans.
During the five-year period following December 31, 2011, the Company expects to pay future benefits to one employee upon the employee's normal retirement age. The Company anticipates that the benefits payable will be immaterial.
| F-15 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 6 - COMMITMENTS
| a. | Royalty commitments |
| 1. | The Company is obligated to pay royalties to the OCS on proceeds from the sale of products developed from research and development activities that were funded, partially, by grants from the OCS. At the time the grants were received, successful development of the related projects was not assured. |
In the case of failure of a project that was partly financed as described above, the Company is not obligated to pay any such royalties or repay funding received from the OCS.
Under the terms of the funding arrangements with the OCS, royalties of 3% to 6% are payable on the sale of products developed from projects funded by the OCS, which payments shall not exceed, in the aggregate, 100% of the amount of the grant received (dollar linked), plus, commencing upon January 1, 2001, interest at annual rate based on LIBOR. In addition, if the Company receives approval to manufacture products developed with government grants outside the State of Israel, it will be required to pay an increased total amount of royalties (possibly up to 300% of the grant amounts plus interest), depending on the manufacturing volume that is performed outside the State of Israel, and, possibly, an increased royalty rate.
Royalty expenses to the OCS are included in the statement of operations as a component of the cost of revenues and were approximately $1,950, $769 and $158 during the years ended December 31, 2009, 2010 and 2011, respectively.
At December 31, 2011, the maximum royalty amount payable by the Company under these funding arrangements is approximately $19,820 (without interest, assuming 100% of the funds are payable).
| 2. | The Company is a party to certain research and license agreements. Under the agreements, the Company is obligated to pay royalties at varying rates from its future revenues. The aggregate royalties payable under all of the agreements is equal to a percentages of net sales of licensed products in the teens. Royalty expenses under the agreements are included in the statement of operations as a component of the cost of revenues and were approximately $1,625, $115 and $19 during the years ended December 31, 2009, 2010 and 2011, respectively. |
Under each agreement, the Company is also obligated to pay milestone, licensing and other payments to the counterparties of the agreement. The payments under the agreements are for varying amounts and are subject to varying conditions. If all of the contingencies with respect to milestone payments under the research and license agreements are met, the aggregate milestone payments payable would be approximately $950 and would be payable, if at all, as the Company’s projects progress over the course of a number of years. In addition, milestone payments of $70 and $100 were paid in respect of the said agreements during the years ended December 31,2009 and 2010, respectively.
None of the agreements has a fixed termination date. Subject to earlier termination for other reasons, each agreement terminates after a certain number of years following the first commercial sale of any licensed product under the agreement or after a certain number of years without the initiation of commercial sales of any product under the agreement.
| b. | Subcontracting Agreements |
The Company has entered into sub-contracting agreements with several clinical providers and consultants in Israel, the United States and certain other countries in connection with its primary product development process and with expenditure of the company’s manufacturing facilities. As of December 31, 2011, total commitments under said agreements were approximately $821.
| F-16 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 6 – COMMITMENTS (continued):
| c. | Lease Agreements |
The Company is a party to a number of lease agreements for its facilities, the latest of which expires in 2017. The Company has the option to extend certain of such agreements on three occasions for additional five-year periods, for a total of 15 additional years. Under the leases, the aggregate monthly rental payments are approximately $75. As of December 31, 2011, the Company provided bank guarantees of approximately $208, in the aggregate, to secure the fulfillment of its obligations under the lease agreements. The future minimum lease payments required in each of the next five years under the operating leases for such premises are approximately as follows: 2012 - $901, 2013 - $890, 2014 - $883, 2015 – $883 and 2016 – $638. Lease expenses totaled $780, $891 and $994 for the years ended December 31, 2009, 2010 and 2011, respectively.
| d. | Vehicle Lease and Maintenance Agreements |
In July 2004, the Company entered into several three-year lease and maintenance agreements for vehicles which are regularly amended as new vehicles are leased. The current monthly lease fees aggregate approximately $46. The minimum expected lease payments for the years ending December 31, 2012, 2013 and 2014 are $514, $267 and $70, respectively.
| e. | Teva Agreement |
On September 14, 2006, the Company entered into an agreement (the “Teva Agreement”) with Teva Pharmaceutical Industries Ltd. (“Teva”) under which the Company agreed to collaborate on the research and development of two proteins to be identified by Teva and the Company, using ProCellEx. The Teva Agreement also identifies additional matters for collaboration between Teva and the Company. Subsequently, two proteins were identified to be researched and developed under the agreement but in 2009, both of the projects were terminated for commercial reasons. Other elements of the Company’s collaboration with Teva are currently ongoing.
| f. | Yissum Agreement |
On August 8, 2007, the Company signed an agreement with the Yissum Research and Development Company, the technology transfer arm of the Hebrew University of Jerusalem, Israel, and the Boyce Thompson Institute for Plant Research, at Cornell University, Ithaca, New York, to develop a proprietary plant cell-based acetylcholinesterase (AChE) and its molecular variants for the use in several therapeutic and prophylactic indications, including a biodefense program and organophosphate-based pesticide treatment. Pursuant to the agreement, the Company has received an exclusive worldwide right and license to certain technology, including patents and additional patent applications relating to AChE (the “Licensed Technology”), for all therapeutic and prophylactic indications. In consideration for the license, the Company is required to make certain milestone payments equal to up to $700, in the aggregate, upon its achievement of clinical milestones and royalties from sales derived from any drugs developed by the Company with the Licensed Technology. The agreement does not terminate until either party to the agreement elect to terminate the agreement, subject to certain terms and conditions set forth therein.
| F-17 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 7 - SHARE CAPITAL
| a. | Rights of the Company’s Common Stock |
The Company’s common stock is listed on the NYSE Amex and, since September 6, 2010, on the Tel Aviv Stock Exchange. Each share of Common Stock is entitled to one vote. The holders of shares of Common Stock are also entitled to receive dividends whenever funds are legally available, when and if declared by the Board of Directors. Since its inception, the Company has not declared any dividends.
| b. | Stock based compensation |
On December 14, 2006, the Board of Directors adopted the Protalix BioTherapeutics, Inc. 2006 Stock Incentive Plan (the “Plan”). The grant of options to Israeli employees under the Plan is subject to the terms stipulated by Sections 102 and 102A of the Israeli Income Tax Ordinance. Each option grant is subject to the track chosen by the Company, either Section 102 or Section 102A of the Israeli Income Tax Ordinance, and pursuant to the terms thereof, the Company is not allowed to claim, as an expense for tax purposes, the amounts credited to employees as a benefit, including amounts recorded as salary benefits in the Company’s accounts, in respect of options granted to employees under the Plan, with the exception of the work-income benefit component, if any, determined on the grant date. For Israeli non-employees, the share option plan is subject to Section 3(i) of the Israeli Income Tax Ordinance.
As of December 31, 2011, 47,920 shares of Common Stock remain available for grant under the Plan.
For purposes of determining the fair value of the options granted to employees and non-employees, the Company’s management uses the fair value of the Common Stock.
From January 1, 2009 through December 31, 2011, the Company granted options to certain employees and non-employees as follows:
| 1. | Options granted to employees: |
| a) | Below is a table summarizing all of the option grants to employees for each of the three years in the period ended December 31, 2011: |
| Year of Grant | No. of options granted | Exercise price range | Vesting period | Fair value at grant | Expiration Period | |||||||||||||||
| 2009* | 504,000 | $ | 2.65 | Upon achievement of certain milestones* | $ | 1,068 | 10 years | |||||||||||||
| 2009 | 120,400 | $ | 2.65 | 4 years | $ | 212 | 10 years | |||||||||||||
| 2010 | 1,016,000 | $ | 6.90 | 3 years commencing upon achievement of a certain milestone | $ | 5,673 | 10 years | |||||||||||||
| 2010 | 428,000 | $ | 6.32-$9.66 | 4 years | $ | 2,147 | 10 years | |||||||||||||
| 2,068,400 | ||||||||||||||||||||
* The milestone was achieved as of December 31, 2009 and the options vested in full.
| F-18 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 7– SHARE CAPITAL (continued):
Set forth below are grants made by the Company to employees (including related parties) during the three-year period ended December 31, 2011 (such grants appear in the table above):
| 1. | In February, 2009, the Company’s Board of Directors approved the grant of options to purchase 624,400 shares of Common Stock to the Company’s Chief Executive Officer and certain officers and employees of the Company with an exercise price equal to $2.65 per share. The options vest as follows: |
(i) 504,000 of the options vest immediately upon the achievement of certain clinical and operational performance milestones, which milestones must be achieved within one year of the date of grant or the options will be forfeited. The options expire within a period of 10 years from the date of grant. The vesting conditions of these options were satisfied prior to December 31, 2009. Accordingly, these options were fully vested at December 31, 2009 and the Company recognized all of the expenses for these options during 2009.
(ii) 120,400 of the options vest as follows: 25% within one year from the date of grant, with the remainder vesting in 12 equal quarterly tranches over 36 months. The options expire within a period of 10 years from the date of grant. The Company’s management assumed the simplified method to reflect the expected life regarding these options. The Company used the simplified method in 2009 as the Company did not have sufficient historical exercise data to provide a reasonable basis upon which to estimate expected term due to the limited period of time its equity shares have been publicly traded.
| 2. | In February 2010, the Company’s Board of Directors approved the grant of options to purchase 1,016,000 shares of Common Stock, in the aggregate, to the Company’s Chief Executive Officer and certain officers and employees of the Company with an exercise price equal to $6.90 per share. The options vest quarterly over a three-year period commencing upon the FDA’s approval of taliglucerase alfa, if at all. The Company treated the awards as performance-based awards and, given that the performance condition is outside the Company's control, concluded that the performance condition was not probable until the performance condition actually occurs. As a result, no share-based compensation expense was recognized as of December 31, 2011. The Company will start recording these expenses following the FDA’s marketing approval of taliglucerase alfa, if at all. |
| 3. | In February 2010, the Company’s Board of Directors approved the grant of options to purchase 160,000 shares of Common Stock with an exercise price equal to $6.81 per share to a new executive officer of the Company. The options expire on the tenth anniversary of the date of grant, unless terminated earlier. The first 25% of the options vest on the first anniversary of the date of grant and the remaining 75% vest in 12 equal tranches on a quarterly basis for three years thereafter. |
| 4. | In September 2010, the Company’s Board of Directors approved the grant of options to purchase 160,000 shares of Common Stock to a new executive officer of the Company with an exercise price equal to $7.55 per share and options to purchase 40,000 shares of Common Stock to a new employee of the Company with an exercise price equal to $6.32 per share. The options vest over a four-year period, with the first 25% vesting on the first anniversary of the applicable date of the grant and the remaining 75% vesting in equal tranches on a quarterly basis for a three-year period thereafter. The options expire on the tenth anniversary of the date of grant, unless terminated earlier. |
| F-19 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 7– SHARE CAPITAL (continued):
| 5. | In November 2010, the Company’s Board of Directors approved the grant of options to purchase 68,000 shares of Common Stock to a new officer of the Company with an exercise price equal to $9.66 per share. The options vest over a four-year period, with the first 25% vesting on the first anniversary of the applicable date of the grant and the remaining 75% vesting in equal tranches on a quarterly basis for a three-year period thereafter. The options expire on the tenth anniversary of the date of grant, unless terminated earlier. |
| 6. | In September 2010, the Company’s Board of Directors modified the terms of the options previously granted to an executive in 2001, by extending the life of the options until 2016. At the date of modification, all of the options were fully vested. The Company concluded that there was no incremental increase in the value of the awards and therefore no accounting charges need to be recorded in connection with the modifications. |
| b) | The fair value of options granted during the years ended December 31, 2009 and 2010 were $1,280 and $7,820 respectively. The fair value of each option granted is estimated on the date of grant using the Black-Scholes option-pricing model, with the following weighted average assumptions: |
| 2009 | 2010 | |||||||
| Dividend yield | 0% | 0% | ||||||
| Expected volatility | 75% | 75% | ||||||
| Risk-free interest rate | 2.74% | 3.23% | ||||||
| Expected life – in years | 9.2 | 8.8 | ||||||
The expected volatility is based on the historical volatility of the Common Stock and those of comparable companies. The risk-free interest rate assumption is based on observed interest rates appropriate for the expected term of the stock options granted in dollar terms. The Company’s management uses the contractual term or its expectations, based on historical incidence of option exercises, as applicable (during 2009 - using the simplified method), of each option as its expected life. The pre-vesting forfeiture rate of approximately 6.3% is estimated based on pre-vesting forfeiture experience.
The total unrecognized compensation cost of employee stock options at December 31, 2011 is $6,218 (net of forfeiture rate) out of which $5,639 is expected to be recognized over a three-year period commencing upon the FDA’s approval of taliglucerase alfa, if at all. The remaining compensation cost of $579 is expected to be recognized over a weighted average period of 0.9 years.
The total cash received from employees as a result of employee stock option exercises for the years ended December 31, 2009, 2010 and 2011 was $293, $501 and $277, respectively. The Company did not realize any tax benefit in connection with these exercises.
During 2011, the Company issued 181,685 shares of Common Stock in connection with the exercise of 182,093 options by certain employees of the Company. The Company received cash proceeds equal to $268 in connection with such exercises. Of such options, 2,132 options were exercised on a “net-exercise” basis.
| F-20 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 7– SHARE CAPITAL (continued):
| 2. | Options granted to consultants, directors, and other service providers: |
As of December 31, 2011, the Company has recognized and recorded all compensation costs related to outstanding options for consultants, directors and other services providers.
No cash was received from consultants as a result of consultant stock option exercises for the years ended December 31, 2009, 2010, and $0.2 was received in 2011. The Company did not realize any tax benefits in connection with these exercises.
During 2009, the Company issued 3,853,441 shares of Common Stock in connection with the exercise of 3,866,093 options by certain consultants, directors, and other service providers of the Company. The Company did not receive cash proceeds in connection with such exercises as all of such options were exercised on a “net-exercise” basis.
During 2011, the Company issued 200,000 shares of Common Stock in connection with the exercise of 200,000 options by certain consultant of the Company. The Company received cash proceeds equal to $0.2 in connection with such exercise.
| 3. | A summary of share option plans, and related information, under all of the Company’s equity incentive plans for the years ended December 31, 2009, 2010 and 2011 are as follows: |
| a. | Options granted to employees: |
| Year ended December 31, | ||||||||||||||||||||||||
| 2009 | 2010 | 2011 | ||||||||||||||||||||||
| Weighted | Weighted | Weighted | ||||||||||||||||||||||
| Number | average | Number | average | Number | average | |||||||||||||||||||
| of | exercise | of | exercise | of | exercise | |||||||||||||||||||
| options | price | options | price | options | price | |||||||||||||||||||
| Outstanding at beginning of year | 5,890,641 | $ | 2.118 | 5,366,729 | $ | 2.476 | 6,367,979 | $ | 3.576 | |||||||||||||||
| Changes during the year: | ||||||||||||||||||||||||
| Granted | 624,400 | 2.650 | 1,444,000 | 7.076 | ||||||||||||||||||||
| Forfeited and Expired | 1,400 | 2.650 | 13,441 | 3.551 | 44,856 | 6.207 | ||||||||||||||||||
| Exercised (*) | 1,146,912 | 0.733 | 429,309 | 1.602 | 182,093 | 1.652 | ||||||||||||||||||
| Outstanding at end of year | 5,366,729 | $ | 2.476 | 6,367,979 | $ | 3.576 | 6,141,030 | $ | 3.563 | |||||||||||||||
| Exercisable at end of year | 3,680,382 | $ | 1.785 | 4,267,850 | $ | 2.183 | 4,647,834 | $ | 2.581 | |||||||||||||||
(*) The total intrinsic value of options exercised during the years ended December 31, 2009, 2010 and 2011, was $7,258, $3,050 and $1,252, respectively.
| F-21 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 7– SHARE CAPITAL (continued):
| b. | Options granted to consultants, directors, and other service providers: |
| Year ended December 31, | ||||||||||||||||||||||||
| 2009 | 2010 | 2011 | ||||||||||||||||||||||
| Weighted | Weighted | Weighted | ||||||||||||||||||||||
| Number | average | Number | average | Number | average | |||||||||||||||||||
| of | exercise | of | exercise | of | exercise | |||||||||||||||||||
| options | price | options | price | options | price | |||||||||||||||||||
| Outstanding at beginning of year | 5,304,785 | $ | 1.285 | 1,438,692 | $ | 4.697 | 1,438,692 | $ | 4.697 | |||||||||||||||
| Changes during the year-Exercised (*) | 3,866,093 | 0.016 | 200,000 | 0.001 | ||||||||||||||||||||
| Outstanding at end of year | 1,438,692 | $ | 4.697 | 1,438,692 | $ | 4.697 | 1,238,692 | $ | 5.385 | |||||||||||||||
| Exercisable at end of year | 1,407,234 | $ | 4.674 | 1,421,734 | $ | 4.653 | 1,235,567 | $ | 5.385 | |||||||||||||||
| (*) | The total intrinsic value of options exercised during the years ended December 31, 2009, 2010 and 2011, was $41,281, $0 and $1,886, respectively. |
| c. | The following tables summarize information concerning outstanding and exercisable options as of December 31, 2011: |
| December 31, 2011 | ||||||||||||||||
| Options outstanding | Options exercisable | |||||||||||||||
| Number of | Weighted | Number of | Weighted | |||||||||||||
| options | average | options | average | |||||||||||||
| outstanding | remaining | exercisable | remaining | |||||||||||||
| Exercise | at end of | contractual | at end of | contractual | ||||||||||||
| prices | year | life | year | life | ||||||||||||
| $ | 0.001 | 719,207 | 3.54 | 719,207 | 3.54 | |||||||||||
| $ | 0.120 | 987,356 | 2.30 | 987,356 | 2.30 | |||||||||||
| $ | 0.399 | 26,673 | 3.35 | 26,673 | 3.35 | |||||||||||
| $ | 0.972 | 1,325,988 | 4.47 | 1,325,988 | 4.47 | |||||||||||
| $ | 2.350 | 160,000 | 6.82 | 120,000 | 6.82 | |||||||||||
| $ | 2.650 | 604,956 | 7.15 | 573,550 | 7.15 | |||||||||||
| $ | 3.020 | 50,000 | 6.10 | 46,875 | 6.10 | |||||||||||
| $ | 5.000 | 1,720,000 | 6.10 | 1,559,210 | 6.10 | |||||||||||
| $ | 6.810 | 160,000 | 8.10 | 70,000 | 8.10 | |||||||||||
| $ | 6.900 | 1,010,000 | 8.15 | |||||||||||||
| $ | 7.550 | 160,000 | 8.66 | 50,000 | 8.66 | |||||||||||
| $ | 9.660 | 68,000 | 8.84 | 17,000 | 8.84 | |||||||||||
| $ | 16.700 | 387,542 | 5.00 | 387,542 | 5.00 | |||||||||||
| 7,379,722 | 5,883,401 | |||||||||||||||
The aggregate intrinsic value of the total outstanding and of total vested and exercisable options as of December 31, 2011 is $15,551 and $15,371, respectively.
| F-22 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 7– SHARE CAPITAL (continued):
| d. | The following table illustrates the effect of share-based compensation on the statement of operations: |
| Year ended December 31, | ||||||||||||
| 2009 | 2010 | 2011 | ||||||||||
| Research and development expenses | $ | 1,489 | $ | 630 | $ | 422 | ||||||
| General and administrative expenses | 1,194 | 652 | 464 | |||||||||
| $ | 2,683 | $ | 1,282 | $ | 886 | |||||||
| c. | Public Offering |
On March 23, 2011, the Company issued and sold 4,000,000 shares of Common Stock in an underwritten public offering at a price to the public of $5.50 per share. The net proceeds to the Company were approximately $20,590 (net of underwriting commissions and issuance costs of $1,410).
In February 2012, the Company issued and sold 5,175,000 shares of Common Stock. See Note 11b.
NOTE 8 - TAXES ON INCOME
| a. | The Company |
Protalix BioTherapeutics, Inc. is taxed according to U.S. tax laws. The Company's income is, or will be, taxed in the United States at the rate of up to 39%.
| b. | Protalix Ltd. |
The Israeli Subsidiary is taxed according to Israeli tax laws:
| 1. | Measurement of results for tax purposes |
Since 2008, the Company has measured the results of the Israeli Subsidiary for tax purposes in nominal terms in NIS. Pursuant to the Israel Income Tax Law (Adjustments for Inflation), 1985 (the “Adjustments Law”), the Subsidiary’s results for tax purposes have been measured through 2007 on a real basis, based on changes in the Israel consumer price index.
| 2. | Tax rates |
The income of the Israeli Subsidiary, other than income from “Approved Enterprises,” is taxed in Israel at the regular rate. See 3 below. According to the provisions of the Law for Amending the Israel Income Tax Ordinance, 2005, corporate tax rates will be gradually lowered, resulting in a corporate tax rate for 2009 and thereafter: 2009 – 26% and for 2010 and thereafter – 25%.
On July 14, 2009, the Israel Economic Efficiency Law (Legislation Amendments for Applying the Economic Plan for 2009 and 2010), 2009, became effective, stipulating, among other things, an additional gradual decrease in tax rates in 2011 and thereafter, as follows: 2011 – 24%, 2012 – 23%, 2013 – 22%, 2014 – 21%, 2015 – 20% and 2016 and thereafter – 18%.
On December 5, 2011, the "Tax Burden Distribution Law" (hereinafter - the 2011 amendment) was officially published, discontinuing a previously approved gradual decrease in corporate tax provided in the 2009 amendment, and setting the corporate tax rate in Israel for 2012 and thereafter to 25%.
Capital gain is subject to capital gain tax according to corporate tax rate in the year of selling the assets.
| F-23 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 8 – TAXES ON INCOME (continued):
| 3. | The Law for the Encouragement of Capital Investments, 1959 (the “Encouragement of Capital Investments Law”) |
Under the Encouragement of Capital Investments Law, including Amendment No. 60 to the Encouragement of Capital Investments Law as published in April 2005, by virtue of the “Approved Enterprise” or “Benefited Enterprise” status the Israeli Subsidiary is entitled to various tax benefits as follows:
| a. | Reduced tax rates |
Income derived from the Approved Enterprise during a 10-year period commencing upon the year in which the enterprise first realizes taxable income is tax exempt, provided that the maximum period to which it is restricted by the Encouragement of Capital Investments Law has not elapsed.
The Israeli Subsidiary has an “Approved Enterprise” plan since 2004 and “Benefited Enterprise” plan since 2009. The period of benefits in respect of the main enterprise of the Company has not yet commenced. The period during which the Company is entitled to benefits in connection with the Benefited Enterprise expires in 2021.
If the Israeli Subsidiary subsequently pays a dividend out of income derived from the “Approved Enterprise” or “Benefited Enterprise” during the tax exemption period, it will be subject to a tax on the amount distributed, including any company tax on these amounts, at the rate which would have been applicable had such income not been exempted.
In addition to the corporate taxes in Israel, the Company might be subject to a withholding tax on the U.S. revenue source portion of the payments made to the Company for its share of Pfizer’s net profits under the Pfizer Agreement. The withholding tax rate is currently 15%.
| b. | Accelerated depreciation |
The Israeli Subsidiary is entitled to claim accelerated depreciation, as provided by Israeli law, in the first five years of operation of each asset, in respect of buildings, machinery and equipment used by the Approved Enterprise and the Benefited Enterprise.
| c. | Conditions for entitlement to the benefits |
The Israeli Subsidiary’s entitlement to the benefits described above is subject to its fulfilling the conditions stipulated by the law, rules and regulations published thereunder, and the instruments of approval for the specific investment in an approved enterprise. If there is any failure by the Israeli Subsidiary to comply with these conditions, the benefits may be cancelled and the Subsidiary may be required to refund the amount of the benefits, in whole or in part, with interest. The Company received a final implementation approval with respect to its “Approved Enterprise” from the Investment Center.
| d. | Amendment of the Law for the Encouragement of Capital Investments, 1959 |
The Encouragement of Capital Investments Law was amended as part of the Economic Policy Law for the years 2011-2012, which was passed by the Israeli Knesset on December 29, 2010 (the “Capital Investments Law Amendment”).
| F-24 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 8 – TAXES ON INCOME (continued):
The Capital Investments Law Amendment sets alternative benefit tracks to those currently in effect under the provisions of the Encouragement of Capital Investments Law.
The benefits granted to the Benefited Enterprises will be unlimited in time, unlike the benefits granted to special Benefited enterprises, which will be limited for a 10-year period. The benefits shall be granted to companies that will qualify under criteria set in the law; for the most part, those criteria are similar to the criteria that were set in the Encouragement of Capital Investments Law prior to its amendment.
Under the transitional provisions of the Encouragement of Capital Investments Law, the Company is entitled to take advantage of the tax benefits available under the Encouragement of Capital Investments Law prior to its amendment until the end of the benefits period, as defined in the Encouragement of Capital Investments Law. The Company will be allowed to set the “year of election” no later than tax year 2012, provided that the minimum qualifying investment was made not later than the end of 2010. On each year during the benefits period, the Company will be able to elect that the Capital Investments Amendment apply to the Company, thereby making the tax rates described above available to the Company. An election to have the Capital Investments Amendment apply is irrecoverable.
| 4. | The Law for the Encouragement of Industry (Taxation), 1969: |
The Israeli Subsidiary is an “industrial company,” as defined under the Law for the Encouragement of Industry (Taxation), 1969 (the “Law for the Encouragement of Industry”). As such, the Israeli Subsidiary is entitled to claim depreciation at increased rates for equipment used in industrial activity, as stipulated by regulations published under Law for the Encouragement of Industry, and has done so.
Under the provisions of the Income Tax Regulations “Accelerated Depreciation in respect of Equipment acquired during the Defined Period” (Temporary Orders), industrial companies whose operations are mostly “eligible operations” are entitled to claim accelerated depreciation at a rate of 50% on machinery and equipment acquired from June 1, 2008 to May 31, 2009.
| c. | Tax losses carried forward to future years |
As of December 31, 2011, the Company had aggregate net operating loss (NOL) carry-forwards equal to approximately $63,040 that are available to reduce future taxable income as follows:
| 1. | The Company |
The NOL carry-forward of the Company equal to approximately $11,740 may be restricted under Section 382 of the Internal Revenue Code (“IRC”). IRC Section 382 applies whenever a corporation with NOL experiences an ownership change. As a result of IRC Section 382, the taxable income for any post change year that may be offset by a pre-change NOL may not exceed the general IRC Section 382 limitation, which is the fair market value of the pre-change entity multiplied by the IRC long-term tax exempt rate.
| 2. | Protalix Ltd. |
At December 31, 2011, the Israeli Subsidiary had approximately $51,300 of NOL carry-forwards that are available to reduce future taxable income with no limited period of use.
| F-25 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 8 – TAXES ON INCOME (continued):
| d. | Deferred income taxes: |
The components of the Company’s net deferred tax assets at December 31, 2010 and 2011 were as follows:
| December 31, | ||||||||
| 2010 | 2011 | |||||||
| In respect of: | ||||||||
| Research and Development expenses | $ | 4,119 | ||||||
| Property and equipment | $ | (143 | ) | (149 | ) | |||
| Holiday and recreation pay | 296 | 305 | ||||||
| Severance pay obligation | 130 | 164 | ||||||
| Deferred revenues | 3,691 | 4,746 | ||||||
| Net operating loss carry forwards | 3,599 | 4,579 | ||||||
| Valuation allowance | (7,573 | ) | (13,764 | ) | ||||
| - | - | |||||||
Deferred taxes are computed using the tax rates expected to be in effect when those differences reverse. The Company used tax rates of 39%, 25% and 0%.
| e. | Reconciliation of the theoretical tax expense to actual tax expense |
The main reconciling items between the statutory tax rate of the Company and the effective rate is the provision for full valuation allowance in respect of tax benefits from carry forward tax losses due to the uncertainty of the realization of such tax benefits (see above).
| f. | Tax assessments |
In accordance with the Income Tax Ordinance, as of December 31, 2011, all of Protalix Ltd.’s tax assessments through tax year 2007 are considered final.
A summary of open tax years by major jurisdiction is presented below:
| Jurisdiction: | Years: | |
| Israel | 2008-2011 | |
| United States (*) | 2002-2011 | |
| Netherlands | 2009-2011 |
(*) Includes federal, state and local (or similar provincial jurisdictions) tax positions.
| F-26 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 9 - SUPPLEMENTARY FINANCIAL STATEMENT INFORMATION:
Balance sheets:
| December 31, | ||||||||
| 2010 | 2011 | |||||||
| a. Accounts receivable - other: | ||||||||
| Deferred costs* | $ | 1,558 | ||||||
| Institutions | $ | 634 | 375 | |||||
| State of Israel (see Note 6a ) | 878 | 1,296 | ||||||
| Restricted deposit | 226 | 208 | ||||||
| Prepaid expenses | 354 | 235 | ||||||
| Sundry | 139 | 165 | ||||||
| $ | 2,231 | $ | 3,837 | |||||
| * Manufacturing costs of inventory, paid by Pfizer, but not delivered. | ||||||||
| b. Accounts payable and accruals – other: | ||||||||
| Payroll and related expenses | $ | 1,199 | $ | 1,108 | ||||
| Provision for vacation and recreation pay | 1,235 | 1,221 | ||||||
| Accrued expenses | 2,030 | 2,327 | ||||||
| Royalties payable | 884 | 89 | ||||||
| Liability in connection with collaboration operation- current portion | 1,322 | |||||||
| Property and equipment supplier | 2,720 | 1,473 | ||||||
| $ | 8,068 | $ | 7,540 | |||||
| F-27 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 9 – SUPPLEMENTARY FINANCIAL STATEMENT INFORMATION (continued):
Statement of operations:
| Year ended December 31, | ||||||||||||
| 2009 | 2010 | 2011 | ||||||||||
| c. Revenues: | ||||||||||||
| Deferred revenues from the license and supply agreement with Pfizer | $ | 388 | $ | 4,563 | $ | 4,563 | ||||||
| Revenues from selling products to Pfizer | 2,079 | 3,823 | ||||||||||
| $ | 388 | $ | 6,642 | $ | 8,386 | |||||||
| d. Cost of Revenues: | ||||||||||||
| Cost of products sold | $ | 807 | $ | 985 | ||||||||
| Write down of inventories | 2,692 | 363 | ||||||||||
| Royalties expenses | $ | 3,575 | 884 | 177 | ||||||||
| $ | 3,575 | $ | 4,383 | $ | 1,525 | |||||||
| e. Research and development expenses - net: | ||||||||||||
| Payroll and related expenses | $ | 10,479 | $ | 13,176 | $ | 14,042 | ||||||
| Subcontractors and consultants | 7,469 | 11,550 | 11,314 | |||||||||
| Materials and consumables | 3,852 | 4,947 | 3,425 | |||||||||
| Rent, insurance and maintenance | 2,238 | 3,457 | 4,113 | |||||||||
| Patent registration and licensing | 387 | 571 | 449 | |||||||||
| Depreciation and write down | 1,799 | 2,456 | 3,057 | |||||||||
| Other | 1,166 | 1,534 | 1,418 | |||||||||
| 27,390 | 37,691 | 37,818 | ||||||||||
| Less - grants and reimbursements: | ||||||||||||
| Development costs reimbursements from Pfizer | 3,922 | 3,430 | ||||||||||
| Grants (see Note 6a) | 5,752 | 3,818 | 3,345 | |||||||||
| $ | 21,638 | $ | 29,951 | $ | 31,043 | |||||||
| f. General and administrative expense: | ||||||||||||
| Payroll and related expenses | $ | 2,804 | $ | 2,415 | $ | 2,481 | ||||||
| Management and consulting fees | 681 | 221 | 215 | |||||||||
| Rent, insurance and maintenance | 347 | 463 | 498 | |||||||||
| Professional fees | 2,274 | 2,274 | 2,326 | |||||||||
| Travel | 251 | 308 | 229 | |||||||||
| Depreciation | 191 | 309 | 363 | |||||||||
| Other | 596 | 886 | 819 | |||||||||
| $ | 7,144 | $ | 6,876 | $ | 6,931 | |||||||
| F-28 |
PROTALIX BIOTHERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(U.S. dollars in thousands)
NOTE 10 - RELATED PARTY TRANSACTIONS
| Year ended December 31, | ||||||||||||
| 2009 | 2010 | 2011 | ||||||||||
| a. Management and consulting fees to the Chairman of the Board | $ | 33 | $ | 6 | - | |||||||
| b. Compensation to the non-executive directors (except the Chairman of the Board in 2009, and includes the interim Chairman of the Board in 2010 and 2011) | $ | 209 | $ | 215 | $ | 215 | ||||||
NOTE 11 - SUBSEQUENT EVENTS
| a. | During January and February 2012, the Company issued a total of 10,239 shares of Common Stock in connection with the exercise of options to purchase 10,239 shares of Common Stock by certain employees of the Company. The Company received aggregate cash proceeds equal to approximately $5 in connection with the exercise of such options. |
| b. | On February 22, 2012, the Company issued and sold 5,175,000 shares of Common Stock in an underwritten public offering at a price to the public of $5.25 per share. The net proceeds to the Company were approximately $25,389 (net of underwriting commissions and issuance costs of $1,567). The offering included the exercise by the underwriters of an over-allotment option to purchase 675,000 shares of Common Stock. |
| F-29 |
