Attached files
Table of Contents
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
| [X] | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended March 31, 2011
Or
| [ ] | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission file number 1-9601
K-V PHARMACEUTICAL COMPANY
(Exact Name of Registrant as Specified in Its Charter)
| Delaware | 43-0618919 | |
| (State or Other Jurisdiction of Incorporation or Organization) |
(I.R.S. Employer Identification No.) |
One Corporate Woods Drive, Bridgeton, MO 63044
(Address of Principal Executive Offices) (ZIP code)
Registrant’s telephone number, including area code: (314) 645-6600
Securities Registered Pursuant to Section 12(b) of the Act:
| Class A Common Stock, par value $.01 per share | New York Stock Exchange | |
| Class B Common Stock, par value $.01 per share | New York Stock Exchange |
Securities Registered Pursuant to Section 12(g) of the Act:
7% Cumulative Convertible Preferred, par value $.01 per share
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ¨ No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ¨ No x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ¨ No x
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulations S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes ¨ No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§229.405) is not contained herein, and will not be contained herein, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act. (Check one):
| Large accelerated filer | ¨ | Accelerated filer | x | |||
| Non-accelerated filer | ¨ (Do not check if a smaller reporting company) | Smaller reporting company | ¨ |
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ¨ No x
The aggregate market value of the shares of Class A and Class B Common Stock held by non-affiliates of the registrant as of September 30, 2010 the last business day of the registrant’s most recently completed second fiscal quarter, was $84.8 million and $25.0 million respectively. As of April 29, 2011, the registrant had outstanding 48.6 million and 11.2 million shares of Class A Common Stock and Class B Common Stock, respectively.
DOCUMENTS INCORPORATED BY REFERENCE—
Parts of the registrant’s definitive proxy statement for the 2011 Annual Meeting of Shareholders are incorporated by reference in Part III of this Annual Report.
Table of Contents
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K (the “Report”) contains various forward-looking statements within the meaning of the United States Private Securities Litigation Reform Act of 1995 (the “PSLRA”) and which may be based on or include assumptions concerning our operations, future results and prospects. Such statements may be identified by the use of words like “plan,” “expect,” “aim,” “believe,” “project,” “anticipate,” “commit,” “intend,” “estimate,” “will,” “should,” “could,” “potential” and other expressions that indicate future events and trends.
All statements that address expectations or projections about the future, including, without limitation, statements about product development, product launches, regulatory approvals, governmental and regulatory actions and proceedings, market position, acquisitions, sale of assets, revenues, expenditures, resumption of manufacturing and distribution of products and the impact of the recall and suspension of shipments on revenues, and other financial results, are forward-looking statements.
All forward-looking statements are based on current expectations and are subject to risk and uncertainties. In connection with the PSLRA’s “safe harbor” provisions, we provide the following cautionary statements identifying important economic, competitive, political, regulatory and technological factors, among others, that could cause actual results or events to differ materially from those set forth or implied by the forward-looking statements and related assumptions. Such factors include (but are not limited to) the following:
| (1) | our ability to continue as a going concern; |
| (2) | the impact of competitive, commercial payor, governmental (including Medicaid program), physician, patient, public or political responses and reactions, and responses and reactions by medical professional associations and advocacy groups, to the Company’s sales, marketing, product pricing, product access and strategic efforts with respect to Makena™, and its other products, including introduction or potential introduction of generic or competing products, or competition from unapproved therapies or compounded drugs, against products sold by the Company and its subsidiaries, including Makena™, and including competitive or responsive pricing changes; |
| (3) | the possibility of not obtaining the U.S. Food and Drug Administration (the “FDA”) approvals or delay in obtaining FDA approvals; |
| (4) | new product development and launch, including the possibility that any product launch may be delayed or unsuccessful, including with respect to Makena™; |
| (5) | acceptance of and demand for the Company’s new pharmaceutical products, including Makena™, and for our current products upon their return to the marketplace, as well as the number of preterm births for which Makena™ may be prescribed and its safety profile and side effects profile and acceptance of the degree of patient access to, and pricing for, Makena™; |
| (6) | the possibility that any period of exclusivity may not be realized, including with respect to Makena™, a designated Orphan Drug; |
| (7) | the satisfaction or waiver of the terms and conditions for the continued ownership of the full U.S. and worldwide rights to Makena™ set forth in the previously disclosed Makena™ acquisition agreement, as amended; |
| (8) | the consent decree between the Company and the FDA and the Company’s suspension of the production and shipment of all of the products that it manufactures (other than the Potassium Chloride Extended Release Capsule products that are the subject of the FDA letter received September 8, 2010 allowing the return of those products to the marketplace) and the related nationwide recall affecting all of the other products that it manufactures, as well as the related material adverse effect on its revenue, assets and liquidity and capital resources, as more fully described in Part I, Item 1—“Business—(b) Significant Developments—Discontinuation of Manufacturing and Distribution; Product Recalls; and the FDA Consent Decree” in this Report; |
2
Table of Contents
| (9) | the two agreements between the Company and the Office of Inspector General of the U.S. Department of Health and Human Services (“HHS OIG”) pertaining to the exclusion of our former chief executive officer from participation in federal healthcare programs and pertaining to the dissolution of our ETHEX subsidiary, in order to resolve the risk of potential exclusion of our Company, as more fully described in Part I, Item 1—“Business—(b) Significant Developments—Agreements with HHS OIG” in this Report; |
| (10) | the plea agreement between the Company and the U.S. Department of Justice and the Company’s obligations therewith, as well as the related material adverse effect, if any, on its revenue, assets and liquidity and capital resources, as more fully described in Note 1—“Description of Business—Plea Agreement with the U.S. Department of Justice” of the Notes to our consolidated financial statements for the fiscal year ended March 31, 2011, including the notes thereto (the “Consolidated Financial Statements”), in Part II, Item 8 of this Report; |
| (11) | changes in the current and future business environment, including interest rates and capital and consumer spending; |
| (12) | the availability of raw materials and/or products, including Makena™ and Evamist®, manufactured for the Company under contract manufacturing agreements with third parties; |
| (13) | the regulatory environment, including legislative, government or regulatory agency and judicial actions and changes in applicable laws or regulations, including the risk of obtaining necessary state licenses in a timely manner; |
| (14) | fluctuations in revenues; |
| (15) | the difficulty of predicting the pattern of inventory movements by the Company’s customers; |
| (16) | risks that the Company may not ultimately prevail in litigation, including product liability lawsuits and challenges to its intellectual property rights by actual or potential competitors or to its ability to market generic products due to brand company patents and challenges to other companies’ introduction or potential introduction of generic or competing products by third parties against products sold by the Company or its subsidiaries including without limitation the litigation and claims referred to in Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report, and that any adverse judgments or settlements of such litigation, including product liability lawsuits, may be material to the Company; |
| (17) | the possibility that our current estimates of the financial effect of certain announced product recalls could prove to be incorrect; |
| (18) | whether any product recalls or product introductions result in litigation, agency action or material damages; |
| (19) | the possibility of our loss of failure to supply claims by certain of the Company’s customers, including CVS Pharmacy, Inc. and Caremark CVS Corporation, that, despite the formal discontinuation action by the Company of its products, the Company should compensate such customers for any additional costs they allegedly incurred for procuring products the Company did not supply; |
| (20) | the series of putative class action lawsuits alleging violations of the federal securities laws by the Company and certain individuals, as more fully described in Note 15—“Commitments and Contingencies—Litigation and Governmental Inquiries” of the Notes to Consolidated Financial Statements included in this Report; |
| (21) | the possibility that insurance proceeds are insufficient to cover potential losses that may arise from litigation, including with respect to product liability or securities litigation; |
| (22) | the informal inquiries initiated by the Securities and Exchange Commission (the “SEC”) and any related or additional government investigation or enforcement proceedings as more fully described in Note 15—“Commitments and Contingencies—Litigation and Governmental Inquiries” of the Notes to Consolidated Financial Statements included in this Report; |
3
Table of Contents
| (23) | the possibility that the pending investigation by HHS OIG into potential false claims under the Title 42 of the U.S. Code as more fully described in Note 15—“Commitments and Contingencies—Litigation and Governmental Inquiries” of the Notes to Consolidated Financial Statements included in this Report could result in significant civil fines or penalties, including exclusion from participation in federal healthcare programs such as Medicare and Medicaid; |
| (24) | delays in returning, or failure to return, certain or many of the Company’s approved products to market, including loss of market share as a result of the suspension of shipments, and related costs; |
| (25) | the ability to sell or license certain assets, and the purchase prices, milestones, terms and conditions of such transactions; |
| (26) | the possibility that default on one type or class of the Company’s indebtedness could result in cross default under, and the acceleration of, its other indebtedness; |
| (27) | the risks that present or future changes in the Board of Directors or management may lead to an acceleration of the Company’s bonds or to adverse actions by government agencies or our auditors; |
| (28) | the risk that even though the price and 30-day average price of the Company’s Class A Common Stock and Class B Common Stock currently satisfy the quantitative listing standards of the New York Stock Exchange, including with respect to minimum share price and public float, the Company can provide no assurance that they will remain at such levels thereafter; |
| (29) | compliance with debt covenants; and |
| (30) | the risks detailed from time-to-time in the Company’s filings with the SEC. This discussion is not exhaustive, but is designed to highlight important factors that may impact our forward-looking statements. |
Because the factors referred to above, as well as the statements included under the captions Part I, Item 1A—“Risk Factors” and Part II, Item 7—“Management’s Discussion and Analysis of Financial Condition and Results of Operations” and elsewhere in this Report, could cause actual results or outcomes to differ materially from those expressed in any forward-looking statements made by us or on our behalf, you should not place undue reliance on any forward-looking statements. All forward-looking statements attributable to us are expressly qualified in their entirety by the cautionary statements in this “Cautionary Note Regarding Forward-Looking Statements” and the risk factors that are included under the caption Part I, Item 1A—“Risk Factors” in this Report, as supplemented by our subsequent SEC filings. Further, any forward-looking statement speaks only as of the date on which it is made and we are under no obligation to update any of the forward-looking statements after the date of this Report. New factors emerge from time to time, and it is not possible for us to predict which factors will arise, when they will arise and/or their effects. In addition, we cannot assess the impact of each factor on our future business or financial condition or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements.
4
Table of Contents
PART I
| Item 1. | Business |
(a) General Overview
Unless the context otherwise indicates, when we use the words “we,” “our,” “our Company” or “KV” we are referring to K-V Pharmaceutical Company and its wholly-owned subsidiaries, including Ther-Rx Corporation (“Ther-Rx”), Nesher Pharmaceuticals, Inc. (“Nesher”), ETHEX Corporation (“ETHEX”) and Particle Dynamics, Inc. (“PDI”). Unless otherwise noted, when we refer to a specific fiscal year, we are referring to our fiscal year that ended March 31 of that year (for example, fiscal year 2011 refers to the fiscal year ended March 31, 2011).
We were incorporated under the laws of Delaware in 1971 as a successor to a business originally founded in 1942. We are a fully integrated specialty pharmaceutical company that develops, manufactures, acquires and markets technologically-distinguished branded and generic/non-branded prescription pharmaceutical products. Our strategic goal is to become a specialty branded pharmaceutical marketing company primarily focused in women’s healthcare. We have a broad range of dosage form manufacturing capabilities, including tablets, capsules, creams and liquids. We conduct our branded pharmaceutical operations through Ther-Rx and, previously, we conducted our generic/non-branded pharmaceutical operations through ETHEX, which focused principally on technologically-distinguished generic products prior to the cessation of its operations on March 2, 2010 and its dissolution on December 15, 2010. Through PDI, divested in June 2010, we developed, manufactured and marketed technologically advanced, value-added raw material products for the pharmaceutical industry and other markets. In May 2010, we formed a wholly-owned subsidiary, Nesher, to operate as the sales and marketing company for our generic products. The Company expects to complete a divestiture of Nesher and the Company’s generics business and assets by the end of calendar year 2011.
Our original strategy was to engage in the development of proprietary drug delivery systems and formulation technologies which enhance the effectiveness of new therapeutic agents and existing pharmaceutical products. Today we utilize one of those technologies, SITE RELEASE® in two products expected to return to our branded portfolio prior to the end of calendar year 2011. Going forward, our business strategy will be primarily defined by the potential in-licensing and acquisition of pharmaceutical products rather than the historical strategy of the internal development of pharmaceutical products.
During fiscal year 2009, the Company announced six separate voluntary recalls of certain tablet form generic products as a precaution due to the potential existence of oversized tablets. In December 2008, the FDA began an inspection of the Company’s facilities. The Company suspended shipments of all approved tablet-form products in December 2008 and of all other drug products in January 2009. Also, in January 2009, the Company initiated a nationwide voluntary recall affecting most of its products. On March 2, 2009, the Company entered into a consent decree with the FDA regarding its drug manufacturing and distribution. The consent decree was entered by the U.S. District Court, Eastern District of Missouri, Eastern Division on March 6, 2009. As part of the consent decree, the Company agreed not to directly or indirectly do or cause the manufacture, processing, packing, labeling, holding, introduction or delivery for introduction into interstate commerce at or from any of its facilities of any drug, until the Company has satisfied certain requirements designed to demonstrate compliance with the FDA’s current good manufacturing practice (“cGMP”) regulations. The consent decree provides for a series of measures that, when satisfied, will permit the Company to resume the manufacture and distribution of approved drug products. The Company has also agreed not to distribute its products that are not FDA approved, including its prenatal vitamins and hematinic products, unless it obtains FDA approval for such products through the FDA’s New Drug Application (“NDA”) or Abbreviated New Drug Application (“ANDA”) processes. These actions and the requirements under the consent decree have had, and are expected to continue to have, a material adverse effect on the Company’s liquidity position and its results of operations. We resumed shipment of extended-release potassium chloride capsule, Micro-K® 10mEq and Micro-K® 8mEq, in September 2010, resumed shipments of the generic version of Potassium Chloride Extended Release Capsule in December 2010 and we began shipping Makena™ in March 2011. We are continuing to prepare other products for FDA inspection although we do not expect to resume shipping other products until later in fiscal year 2012, at the
5
Table of Contents
earliest. Micro-K and Micro-K 10 Extencaps (potassium chloride, USP) Extended-Release Capsules are indicated for the treatment and prevention of hypokalemia (potassium depletion). Potassium Chloride Extended-Release Capsules, USP, are indicated for the treatment and prevention of hypokalemia (potassium depletion). Nesher Pharmaceuticals’ Potassium Chloride Extended-Release Capsules is the authorized generic product to Micro-K and Micro-K 10 Extencaps. The labeled ingredients for Potassium Chloride Extended-Release Capsules are the same as Micro-K and Micro-K 10 Extencaps. Makena™ (hydroxyprogesterone caproate injection) is the first FDA-approved drug indicated to reduce the risk of preterm birth in women with a singleton pregnancy who have a history of singleton spontaneous preterm birth. Makena™ is not intended for use in women with multiple gestations or other risk factors for preterm birth.
In July 2010, our Board of Directors directed management to explore strategic alternatives with respect to Nesher and the assets and operations of our generic products business, which could include a sale of Nesher. We have retained Jefferies & Co. to advise us with this strategy. In the fourth quarter of fiscal year 2011, management committed to a plan to divest the generics business. In the meantime, we will continue to prepare products for FDA inspection and continue to anticipate the reintroduction of approved products into the market.
(b) Significant Developments
Changes in Management and Directors
At the Annual Meeting of Stockholders for fiscal year 2009 held on June 10, 2010 (the “Annual Meeting”), the stockholders elected Gregory Bentley, Mark A. Dow, Terry B. Hatfield, David S. Hermelin, Marc S. Hermelin, Joseph D. Lehrer and John Sampson to serve as directors with terms expiring at the Annual Meeting of Stockholders for fiscal year 2010. Former members of the Board Jean M. Bellin, Kevin S. Carlie, Jonathon E. Killmer and Norman D. Schellenger were not re-elected.
On June 14, 2010, Stephen A. Stamp resigned, effective immediately, from his position as Chief Financial Officer of our Company. Thomas S. McHugh was appointed Chief Financial Officer and Treasurer effective July 15, 2010. Prior to this appointment, Mr. McHugh served as Chief Accounting Officer and Vice President of Finance—Corporate Controller.
On June 15, 2010, each of Mr. Hatfield and Mr. Sampson resigned as members of the Board, effective as of the earlier of July 7, 2010 or the date a replacement was appointed. Mr. Hatfield served as the Chairman of the Board and Mr. Sampson served on the Audit Committee. Each of Mr. Hatfield and Mr. Sampson indicated that he was resigning because of serious concerns regarding the ability of the newly-constituted Board and senior management to provide the required independent oversight of the business during the current critical period in its history.
On June 17, 2010, the Board appointed Ana I. Stancic as a director to fill the vacancy created by the resignation of Mr. Hatfield. As noted above, Mr. Hatfield’s resignation became effective upon the appointment of Ms. Stancic.
On July 7, 2010, the Board appointed David Sidransky, M.D. as a director to fill the vacancy created by the resignation of Mr. Sampson. As noted above, Mr. Sampson’s resignation became effective upon the appointment of Dr. Sidransky.
On July 29, 2010, the Board increased the total number of Board members to eight (but returning automatically to seven members upon any current director leaving the Board) and appointed Robert E. Baldini as a director to fill the newly-created position.
At a Board meeting held subsequent to the Annual Meeting on June 10, 2010, the Board terminated the employment of David A. Van Vliet, who then served as Interim President and Interim Chief Executive Officer, effective at the end of the 30-day notice period provided for in his employment agreement, during which period he was placed on administrative leave.
6
Table of Contents
Also at that meeting, the Board appointed Gregory J. Divis, Jr. as the Interim President and Interim Chief Executive Officer of our Company. Mr. Divis was subsequently appointed as our permanent President and Chief Executive Officer on November 17, 2010. The other terms of Mr. Divis’ employment were not changed by this appointment.
On November 10, 2010, Marc S. Hermelin voluntarily resigned as a member of the Board. We had been advised that HHS OIG notified Mr. M. Hermelin that he would be excluded from participating in federal healthcare programs effective November 18, 2010. In an effort to avoid adverse consequences to our Company, including a potential discretionary exclusion of our Company from participation in federal healthcare programs, and to enable our Company to secure our expanded financial agreement, as more fully described in Note 13— “Long-Term Debt” of the Notes to Consolidated Financial Statements included in this Report, with U.S. Healthcare, the Company, HHS OIG, Mr. M. Hermelin and his wife (solely with respect to her obligations there under, including as joint owner with Mr. M. Hermelin of certain shares of Company stock) entered into the Settlement Agreement under which Mr. M. Hermelin also resigned as trustee of all family trusts that hold KV stock, agreed to divest his personal ownership interests in our Company’s Class A Common and Class B Common Stock (approximately 1.8 million shares, including shares held jointly with his wife) over an agreed upon period of time in accordance with a divestiture plan and schedule approved by HHS OIG, and agreed to refrain from voting stock under his personal control. In order to implement such agreement, Mr. M. Hermelin and his wife granted to an independent third party immediate irrevocable proxies and powers of attorney to divest their personal stock interests in the Company if Mr. M. Hermelin does not timely do so. The Settlement Agreement also required Mr. M. Hermelin to agree, for the duration of his exclusion, not to seek to influence or be involved with, in any manner, the governance, management, or operations of our Company.
As long as the parties comply with the Settlement Agreement, HHS OIG has agreed not to exercise its discretionary authority to exclude our Company from participation in federal health care programs, thereby allowing our Company and our subsidiaries (with the single exception of ETHEX, which was dissolved pursuant to the Divestiture Agreement with HHS OIG) to continue to conduct business through all federal and state healthcare programs.
As a result of Mr. M. Hermelin’s resignation and the two agreements with HHS OIG, we believe we have resolved our remaining issues with respect to HHS OIG and are positioned to continue to participate in Federal healthcare programs now and in the future.
On June 1, 2011, Patrick J. Christmas joined the Company as the Company’s new Vice President and General Counsel. Mr. Christmas will become the Company’s chief legal officer succeeding Gregory S. Bentley, who is the Company’s Senior Vice President of Law and a member of its Board of Directors. Mr. Bentley has served in that capacity since June 2010 under an arrangement which contemplated that he would return to his private legal practice after the Company filled the General Counsel position. Mr. Bentley will continue to serve as a director of the Company and to counsel the Company after he resumes private practice.
Discontinuation of Manufacturing and Distribution; Product Recalls; and the FDA Consent Decree
In May 2008, we received two reports of an oversized morphine sulfate extended-release tablet in commercial distribution. We conducted an investigation into the possible causes of any such oversized tablets and the likelihood that additional lots of morphine sulfate extended-release tablets or other products might contain oversized tablets. We instituted changes in our manufacturing processes to address the identified causes and intended to prevent any oversized tablets from entering commercial distribution. In addition, in June 2008, ETHEX initiated voluntary recalls of morphine sulfate 30-mg and 60-mg extended-release tablets. In July 2008, a voluntary recall was initiated in Canada by the seller of specific lots of morphine sulfate 60-mg, 30-mg and 15-mg extended-release tablets that we manufactured. On October 15, 2008, ETHEX commenced a voluntary recall of three specific lots of dextroamphetamine sulfate 5-mg tablets as a precaution due to the possible presence of oversized tablets. On November 7, 2008, ETHEX announced a voluntary recall to the consumer level
7
Table of Contents
of multiple lots of five generic products of varying strengths as a precaution due to the potential presence of oversized tablets. These products included: propafenone HCl tablets, isosorbide mononitrate extended-release tablets, morphine sulfate extended-release tablets, morphine sulfate immediate release tablets, and dextroamphetamine sulfate tablets. On November 10, 2008, ETHEX initiated a voluntary recall to the retail level as a precaution due to the possible presence of oversized tablets. This recall affected multiple lots of 18 generic/non-branded products.
On December 15, 2008, the FDA began an inspection of our facilities.
On December 19, 2008, we voluntarily suspended all shipments of our FDA approved drug products in tablet form and commenced a voluntary nationwide single production lot recall of one of our pain management drugs. The 14 products of varying strengths affected by the suspension included metoprolol succinate extended-release tablets (Toprol®-XL), oxycodone HCl tablets and potassium chloride 20 mEq extended-release tablets.
Effective January 22, 2009, we voluntarily suspended the manufacturing and shipment of the remainder of our products, except for three products we distributed but did not manufacture and which did not generate a significant amount of revenue.
On January 28, 2009, we initiated a nationwide voluntary recall of products manufactured or packaged at KV facilities, affecting most of our products. The recall was subsequently expanded on February 3, 2009. This recall affected multiple lots of over 150 branded and generic/non-branded products.
On February 2, 2009, the FDA issued inspectional observations set forth on a “Form 483 Report.”
On March 2, 2009, we entered into a consent decree with the FDA regarding our drug manufacturing and distribution. The consent decree was entered by the U.S. District Court, Eastern District of Missouri, Eastern Division on March 6, 2009 and continues for a period of six years following satisfaction of certain obligations contained in the consent decree after which we may petition the Court for relief from the consent decree. As part of the consent decree, we have agreed not to directly or indirectly do or cause the manufacture, processing, packing, labeling, holding, introduction or delivery for introduction into interstate commerce at or from any of our facilities of any drug, until we have satisfied certain requirements designed to demonstrate compliance with the FDA’s cGMP regulations. The consent decree provides for a series of measures that, when satisfied, will permit us to resume the manufacture and distribution of approved drug products. We have also agreed not to distribute our products that are not FDA approved, including our prenatal vitamins and hematinic products, unless we obtain FDA approval for such products through the FDA’s ANDA or NDA processes.
On March 16, 2009, in response to the consent decree requirements, we initiated the disposal of our existing affected inventory of products, which was completed within the required timeframe.
As part of such measures set forth in the consent decree, we were required to provide, and have provided, to the FDA a work plan (the “Work Plan”) for approval, which sets forth the steps we have taken and will subsequently take to address previously identified deficiencies in our compliance with cGMP regulations.
On July 27, 2009, a representative of the Compliance Branch of the FDA’s Kansas City District, acting in conjunction with the Office of Compliance of the FDA’s Center for Drug Evaluation and Research (“CDER”), notified us by electronic mail that the FDA had completed its review of the Work Plan. Subject to the addition of certain changes, to which we agreed and incorporated into the Work Plan, the FDA accepted the Work Plan. While acceptance of the Work Plan was pending, we, with the knowledge of the FDA, had already begun implementing certain measures set forth in the Work Plan. The final Work Plan, with all requested changes, was submitted to the FDA on August 13, 2009 and accepted by the FDA.
The consent decree further provides that, before resuming manufacturing, we were required to retain and have an independent cGMP expert undertake a review of our facilities and operations and certify compliance
8
Table of Contents
with cGMP regulations. Following that certification, the FDA would make a determination as to whether we are in compliance. On January 13, 2010, our independent cGMP expert, Lachman Consultants (“Lachman”), notified the FDA that Lachman had performed a comprehensive inspection and that our facilities and controls are in compliance with cGMP and the consent decree, but advised us to enact further enhancements to certain aspects of our cGMP systems. In accordance with the advice from Lachman, we continued to enhance our cGMP systems, and Lachman subsequently reinspected our cGMP systems and on April 26, 2010 certified our compliance with all cGMP systems requirements. The next step in the process for resumption of product shipment was for Lachman to certify individual products manufactured under the newly certified cGMP systems. We completed the manufacture of validation batches of the first product, which were successfully completed through Lachman review on July 27, 2010.
Lachman certified the manufacture of the product on July 27, 2010, and the FDA subsequently conducted its own inspection during the week of August 16, 2010 of our facilities, systems and processes as outlined in the consent decree and found no adverse findings. The Company received notification from the FDA on September 8, 2010 of approval for our discontinued generics operations to ship into the marketplace the first product approved under the consent decree, i.e., the Potassium Chloride Extended Release Capsule. We resumed shipment of extended-release potassium chloride capsule, Micro-K® 10mEq and Micro-K® 8mEq, in September 2010. We resumed shipments of the generic version of Potassium Chloride Extended Release Capsule in December 2010 and we began shipping Makena™ in March 2011.
Even after a successful FDA inspection, we anticipate that additional data will need to be generated and submitted to the FDA with respect to certain of our other approved products before we can return them to the market, which may involve performing additional work with regard to product and formulation development. Similarly, the FDA has also informed us that, with respect to certain of our products that are subject to ANDAs or supplemented ANDAs we had submitted before entering into the consent decree, we will need to develop and/or submit additional data before those applications can be considered for approval.
The steps taken by us in connection with the nationwide recall and suspension of shipment of all products manufactured by us and the requirements under the consent decree have had, and are expected to continue to have, a material adverse effect on our results of operations. We do not expect to generate any significant revenues from products that we manufacture until we can resume shipping certain or many of our approved products. In the meantime, we must meet ongoing operating costs related to our employees, facilities and FDA compliance, as well as costs related to the steps we are currently taking to prepare for introducing or reintroducing our products to the market. If we are not able to obtain the FDA’s clearance to resume manufacturing and distribution of more of our approved products in a timely manner and at a reasonable cost, our financial position, results of operations, cash flows and liquidity will continue to be materially adversely affected.
Plea Agreement with the U.S. Department of Justice
As described in Note 1—“Description of Business—Plea Agreement with the U.S. Department of Justice” of the Notes to Consolidated Financial Statements included in this Report, we, at the direction of a special committee of the Board of Directors that was in place prior to June 10, 2010, responded to requests for information from the Office of the United States Attorney for the Eastern District of Missouri and FDA representatives working with that office. In connection therewith, on February 25, 2010, the Board, at the recommendation of the special committee, approved entering into a plea agreement with the Department of Justice.
The plea agreement was executed by the parties and was entered by the U.S. District Court, Eastern District of Missouri, Eastern Division on March 2, 2010. Pursuant to the terms of the plea agreement, ETHEX pleaded guilty to two felony counts, each stemming from the failure to make and submit a field alert report to the FDA in September 2008 regarding the discovery of certain undistributed tablets that failed to meet product specifications. Sentencing pursuant to the plea agreement also took place on March 2, 2010.
9
Table of Contents
Pursuant to the plea agreement, ETHEX agreed to pay a criminal fine in the amount of $23.4 million in four installments. The first installment, in the amount of $2.3 million, was due and paid within 10 days of sentencing. Under the original payment schedule, the second and third installments, each in the amount of $5.9 million, were due on December 15, 2010 and July 11, 2011, respectively. The fourth and final installment, in the amount of $9.4 million, was due on July 11, 2012. On November 15, 2010, upon the motion of the Department of Justice, the court vacated the previous fine installment schedule and imposed a new fine installment schedule using the standard federal judgment rate of 0.22%, payable as follows (the Company made the December 15, 2010 payment as scheduled):
| Payment Amount including interest ($ in millions) |
Payment Due Date | |
| $ 1.0 |
December 15, 2010 | |
| 1.0 |
June 15, 2011 | |
| 1.0 |
December 15, 2011 | |
| 2.0 |
June 15, 2012 | |
| 4.0 |
December 15, 2012 | |
| 5.0 |
June 15, 2013 | |
| 7.1 |
December 15, 2013 |
ETHEX also agreed to pay, within 10 days of sentencing, restitution to the Medicare and the Medicaid programs in the amounts of $1.8 million and $0.6 million, respectively. In addition to the fine and restitution, ETHEX agreed not to contest an administrative forfeiture in the amount of $1.8 million, which was payable and paid within 45 days after sentencing and which satisfied any and all forfeiture obligations ETHEX may have as a result of the guilty plea. In total, ETHEX agreed to pay fines, restitution and forfeiture in the aggregate amount of $27.6 million.
In exchange for the voluntary guilty plea, the Department of Justice agreed that no further federal prosecution will be brought in the Eastern District of Missouri against ETHEX, KV and Ther-Rx regarding allegations of the misbranding and adulteration of any oversized tablets of drugs manufactured by us, and the failure to file required reports regarding these drugs and patients’ use of these drugs with the FDA, during the period commencing on January 1, 2008 through December 31, 2008.
Agreements with HHS OIG
In connection with the guilty plea described above by ETHEX, ETHEX was expected to be excluded from participation in federal healthcare programs, including Medicare and Medicaid. In addition, as a result of the guilty plea by ETHEX, HHS OIG had discretionary authority to also exclude KV from participation in federal healthcare programs. However, we are in receipt of correspondence from HHS OIG that, absent any transfer of assets or operations that would trigger successor liability, HHS OIG has no present intent to exercise its discretionary authority to exclude the Company as a result of the guilty plea by ETHEX.
In connection with the anticipated exclusion of ETHEX from participation in federal healthcare programs, we ceased the operations of ETHEX on March 2, 2010. However, we have retained the ability to manufacture, market and distribute (once the requirements under the consent decree have been met) all generic products and are in possession of all intellectual property related to generic products, including all NDAs and ANDAs pertaining to our brand and generic drug products. We currently do not anticipate that the voluntary guilty plea by ETHEX will have a material adverse effect on our efforts to comply with the requirements pursuant to the consent decree and to resume production and shipments of our approved products.
On November 15, 2010, we entered into the Divestiture Agreement with HHS OIG under which we agreed to sell the assets and operations of ETHEX to unrelated third parties prior to April 28, 2011 and to file articles of dissolution with respect to ETHEX under Missouri law by that date. We filed articles of dissolution for ETHEX on December 15, 2010. Following the filing, ETHEX may not engage in any new business other than winding up
10
Table of Contents
its operations and will engage in a process provided under Missouri law to identify and resolve its liabilities over at least a two-year period. Under the terms of the Divestiture Agreement, HHS OIG agreed not to exclude ETHEX from federal healthcare programs until April 28, 2011 and, upon completion of the sale of the ETHEX assets and of the filing of the articles of dissolution of ETHEX, the agreement will terminate. Civil monetary penalties and exclusion of ETHEX could have occurred if we had failed to meet our April 28, 2011 deadline. The sales of ETHEX’s assets (other than certain fixtures as to which HHS OIG has consented to non-divestiture) were completed prior to the April 28, 2011 deadline and ETHEX no longer has any ongoing material assets or operations other than those required to conclude the winding up process under Missouri law. We have also received a letter from HHS OIG advising us further that assuming that we have complied with all agreements deemed necessary by HHS OIG, HHS OIG would not exclude ETHEX thereafter. On April 1, 2011, we requested confirmation from HHS OIG that the steps and actions described in our reports to HHS OIG constituted full compliance with the duties KV and ETHEX were to complete by April 28, 2011 under the Divestiture Agreement, such that they are not at risk for stipulated penalties or exclusion after that date under the Divestiture Agreement. On April 8, 2011, we received a letter from HHS OIG stating that, at this time, based upon the information provided to HHS OIG in our monthly submissions, HHS OIG has no concerns regarding the actions taken by KV and ETHEX pursuant to the Divestiture Agreement. On May 20, 2011, we received a letter from HHS OIG stating that based on its review of the information provided in our monthly reports, it appeared that the Company and ETHEX had completed our obligations under the Divestiture Agreement.
The Company is also a signatory to the Settlement Agreement between HHS OIG and Mr. M. Hermelin with respect to certain obligations therein, including adopting certain internal policies and notices, reporting violations, reimbursing certain fees of a third party acting under the agreement, and taking certain actions in support of the agreement, including providing certain assistance in the sale of the shares being divested thereunder.
Hologic Agreement
On January 16, 2008, the Company entered into the Original Makena™ Agreement with Hologic. On January 8, 2010, the Company and Hologic entered into Amendment No. 1 to the Original Makena™ Agreement, which, among other things, included a $70 million cash payment for the exclusive rights to Makena™, which was recorded as purchased in-process research and development expense on the statement of operations for the fiscal year ended March 31, 2010. On February 4, 2011, the Company entered into Amendment No. 2 to the Original Makena™ Agreement. The amendments set forth in Amendment No. 2 reduced the payment to be made on the fifth business day following the day on which Hologic gave the Company notice that the FDA has approved Makena™ (the “Transfer Date”) to $12.5 million and revised the schedule for making the remaining payments of $107.5 million. Under these revised payment provisions, after the $12.5 million payment on the Transfer Date and a subsequent $12.5 million payment 12 months after the date the FDA approved Makena™ (the “Approval Date”), the Company has the right to elect between the two alternate payment schedules for the remaining payments, with royalties of 5% of the net sales of Makena™ payable for certain periods and under different circumstances, depending on when the Company elects to make the remaining payments. The Company may make any of the payments on or before their due dates, and the date on which the Company makes the final payment contemplated by the selected payment schedule will be the final payment date, after which royalties, if any, will cease to accrue.
Under the revised payment provisions set forth in Amendment No. 2, after the $12.5 million payment on the Transfer Date and a subsequent $12.5 million payment twelve months after the Approval Date, the Company has the right to elect between the following two alternate payment schedules for the remaining payments:
Payment Schedule 1:
| • | A $45 million payment 18 months after the Approval Date, plus a royalty equal to 5% of net sales of Makena™ made during the period from 12 months after the Approval Date to the date the $45 million payment is made; |
11
Table of Contents
| • | A $20 million payment 21 months after the Approval Date; |
| • | A $20 million payment 24 months after the Approval Date; and |
| • | A $10 million payment 27 months after the Approval Date. |
The royalties will continue to be calculated subsequent to the $45 million milestone payment but do not have to be paid as long as the Company makes subsequent milestone payments when due.
Payment Schedule 2:
| • | A $7.3 million payment 18 months after the Approval Date, plus a royalty equal to 5% of net sales of Makena™ made during the period from 12 months after the Approval Date to 18 months after the Approval Date; |
| • | A $7.3 million payment for each of the succeeding twelve months; |
| • | A royalty payable 24 months following the Approval Date equal to 5% of net sales of Makena™ made during the period from 18 months after the Approval Date to 24 months after the Approval Date; and |
| • | A royalty payable 30 months following the Approval Date equal to 5% of net sales of Makena™ made during the period from 24 months after the Approval Date to 30 months after the Approval Date. |
Notwithstanding anything to the contrary in Amendment No. 2, however, the Company may make any of the foregoing payments on or before their due dates, and the date on which the Company makes the final payment contemplated by the selected payment schedule will be the final payment date, after which no royalties will accrue.
Moreover, if the Company elects Payment Schedule 1 and thereafter elects to pay the $45 million payment earlier than the 18-month deadline, the royalties beginning after 12 months will cease to accrue on the date of the early payment. Additionally, the subsequent payments will be paid in three month intervals following the $45 million payment date.
Lastly, if the Company elects Payment Schedule 1 and thereafter does not make any of the milestone payments when due, Amendment No. 2 provides that no payment default will be deemed to occur, provided the Company timely pays the required royalties accruing in the quarter during which the milestone payment has become due but is not paid.
Under the Indenture governing the $225 million aggregate principal amount of 12% Senior Secured Notes due 2015 (the “2015 Notes”), described further in this Item 1 under “Financing; Private Placement of Class A Common Stock; Private Placement of Notes—Private Placement of 2015 Notes,” the Company agreed to make the $45 million payment under Payment Schedule 1 12 months after the Approval Date and agreed to certain other restrictions on its ability to amend the payment schedules.
Makena™
On February 3, 2011, the Company was informed by Hologic that the FDA granted approval for Makena™. The Company shipped approximately 6,300 vials to its customers, specialty pharmacies and specialty distributors in March 2011. On February 14, 2011, the Company announced the initial list price at $7,500 per vial, or $1,500 per injection and on April 1, 2011 the list price was reduced to $3,450 per vial, or $690 per injection including for March 2011 shipments. Additionally, the Company announced on April 1, 2011 that it would expand its patient financial assistance program so that the majority of women who are clinically eligible for Makena™ would have a co-pay of no more than twenty dollars per injection.
12
Table of Contents
FDA Inspections of KV
In February 2011, the FDA conducted an inspection with respect to the Company’s Clindesse® product (Clindesse® (clindamycin phosphate) Vaginal Cream, 2%, is a single-dose anti-infective product indicated for the treatment of bacterial vaginosis in non-pregnant women.) and issued a Form 483 Report with certain observations. On February 28, 2011, the Company filed its responses with the FDA with respect to such observations. We have advised the FDA of a required equipment change and we are moving forward with the appropriate validation protocol and would expect the FDA to conduct a follow-up inspection. As a result, the Company expects to relaunch Clindesse® and Gynazole-1® in the second half of fiscal 2012.
In March 2011, the FDA conducted an inspection with respect to adverse drug experience reporting. The inspection was completed without any observations being issued by the FDA.
Workforce Reduction and Cost Conservation Actions
On March 30, 2010, we committed to a plan to reduce our employee workforce from 317 to 237 employees. On March 31, 2010, we implemented the plan. On February 25, 2011, the Company further reduced its workforce by 11 and laid off an additional 14 employees. On March 31, 2011, the size of our workforce was approximately 200 employees. The reduction in our workforce is a part of our efforts to conserve our cash and financial resources while we continue working with the FDA to return approved products to market.
On September 13, 2010, we implemented a mandatory salary deferral program for most exempt personnel, ranging from 15% to 25% of base salary, in order to conserve our cash and financial resources. In March 2011, the salaries were reinstated and the salary reduction program terminated.
Financing; Private Placement of Class A Common Stock; Private Placement of Notes
U.S. Healthcare Loan
On September 13, 2010, the Company entered into a loan agreement with U.S. Healthcare I, L.L.C. and U.S. Healthcare II, L.L.C., affiliates of Centerbridge Partners L.P. (collectively, “U.S. Healthcare”) for a $20 million loan secured by assets of the Company. The loan agreement included a period of exclusivity through September 28, 2010 to negotiate an expanded, longer-term financial arrangement among the Company and U.S. Healthcare.
On November 17, 2010, the Company entered into an agreement with U.S. Healthcare for a senior secured debt financing package of up to $120 million consisting of (1) a fully funded $60.0 million term loan (the “Bridge Loan”) that retired the $20 million loan previously provided by U.S. Healthcare on September 13, 2010, and that was provided for general corporate and working capital purposes and (2) a commitment to provide a multi-draw term loan up to an aggregate principal amount of $120 million (the “Multi-Draw Term Loan”) with such additional draws dependent on the achievement by the Company of various conditions as outlined in the related agreement. The Company expensed approximately $1.9 million of unamortized deferred financing costs related to the retirement of the $20 million as required by accounting for debt extinguishments in the quarter ended December 31, 2010.
Under the terms of the Bridge Loan agreement, the Company paid interest at an annual rate of 16.5% (5% of which was payable in kind) with a maturity date in March 2013. The Company furnished as collateral substantially all assets of the Company to secure the loan. The Bridge Loan was guaranteed by certain of the Company’s domestic subsidiaries and the guarantors furnished as collateral substantially all of their assets to secure the guarantee obligations. In addition, the Company issued stock warrants to U.S. Healthcare granting them rights to purchase up to 12.588 million shares of the Company’s Class A Common Stock (the “Initial Warrants”). The Initial Warrants have an exercise price of $1.62 per share, subject to possible anti-dilutive adjustment. These Initial Warrants were valued at $24 million using a Black-Scholes option pricing model utilizing the following assumptions: risk free rate of 1.5%; expected volatility of 99.0%; expected dividend of $0;
13
Table of Contents
and expected life of five years. In recording the Bridge Loan transaction, the Company allocated the proportionate share of the fair value of the Initial Warrants, to the September loan. As a result of the proceeds from the Bridge Loan extinguishing the September loan, the fair value of the Initial Warrants of $8.0 allocated to the September loan was expensed as required by accounting for debt extinguishments in the quarter ended December 31, 2010.
The company then allocated, at their relative fair value, the proportionate share of the fair value of the Initial Warrants of $11.4 as a discount to the Bridge Loan. The discount was being amortized using the effective interest method to interest expense based upon the maturity date of the Bridge Loan. In March 2011, the Company retired the Bridge Loan and expensed the remaining discount in loss on extinguishment of debt.
The $120 million Multi-Draw Term Loan consisted of three tranches that would have been available to the Company following the achievement of certain conditions. The first tranche of $80 million would have been available upon the approval of Makena™ and would have been used to repay the Bridge Loan of $60 million, make a milestone payment to Hologic, and provide funds for general corporate and working capital purposes. The second tranche of $20 million would have been available to the Company upon achieving at least one of certain performance thresholds including either, (1) certain metrics associated with Evamist® (Evamist (estradiol transdermal spray) is a unique transdermal estrogen therapy indicated for the treatment of moderate-to-severe vasomotor symptoms (hot flashes) due to menopause) or (2) receiving FDA approval for the manufacture and distribution of Clindesse® and Gynazole-1®(Gynazole-1 (butoconazole nitrate) Vaginal Cream, 2%, is a single-dose antifungal product indicated for the local treatment of vulvovaginal candidiasis (yeast infections) in non-pregnant women). The proceeds of the second tranche would have been used for general corporate and working capital purposes. The third tranche of $20 million would have been available to the Company upon evidencing its ability, to the satisfaction of U.S. Healthcare, to meet certain liquidity thresholds necessary to satisfy future obligations, including a future milestone payment to Hologic that is due to be paid one year following FDA approval of Makena™. The proceeds from the third tranche would have been used for general corporate and working capital purposes.
The Company and U.S. Healthcare amended the financing arrangements on January 6, 2011 and again on March 2, 2011. Pursuant to the amendments, the Company and U.S. Healthcare amended the Bridge Loan terms and covenants to reflect the Company’s then current projections and timing of certain anticipated future events, including the planned disposition of certain assets. The amendments extended the $60 million payment that was due on March 20, 2011 to three payments of $20 million each with the first payment due (and paid on February 17, 2011) upon closing and funding the private placement of Class A Common Stock, $20 million due in April 2011 and $20 million due in August 2011. In addition, all past covenant issues were waived. As a result of the amendments, the Company would not have been required to sell its generics business by March 20, 2011, but would have been required to cause such sale by August 31, 2011. In addition, the applicable premium (a make-whole payment of interest with respect to payments on the loans prior to maturity) was amended to provide that if the Bridge Loan was repaid in full as a result of a refinancing transaction provided other than by U.S. Healthcare, as occurred on March 17, 2011 with the issuance of the 2015 Notes, a premium was payable to U.S. Healthcare equal to $12.5 million, of which $7.2 million has already been paid in connection with the private placement of Class A Common Stock and the rest was paid in March 2011 and all was recorded as a loss on extinguishment of debt. In addition, on March 17, 2011, an amount of $7.5 million was placed in escrow and will be released to the Company or to U.S. Healthcare on August 31, 2011 or September 30, 2011, as the case may be, depending on the status of the Company’s registration process with the SEC by such dates of the shares underlying the warrants and the Company’s stock price meeting certain specified levels as of the applicable date. In connection with the amendments and certain waivers granted by U.S. Healthcare, the Company issued additional warrants to U.S. Healthcare to purchase up to 7.451 million shares of the Company’s Class A Common Stock, at an exercise price of $1.62 per share, and amended and restated the Initial Warrants (we refer to the additional warrants and the Initial Warrants collectively as the “Warrants”).
The Warrants expire November 17, 2015, but may be extended by up to two additional years if the holders become subject to certain percentage ownership limitations that prevent their exercise in full at the time of their
14
Table of Contents
stated expiration. The Company may require that the holders exercise the Warrants before their expiration if the average of the closing prices of the Class A Common Stock for at least 30 consecutive trading days exceeds $15.00, the closing prices of the Class A Common Stock have exceeded $15.00 for 10 consecutive trading days, the shares issuable upon exercise may be resold under an effective registration statement or the resale is exempt from registration and the shares are listed on the NYSE or the National Association of Securities Dealers Automated Quotation. The Warrants also contain certain anti-dilution provisions included at the request of U. S. Healthcare, pursuant to which the number of shares subject to the Warrants may be increased and the exercise price may be decreased. These anti-dilution provisions are triggered upon certain sales of securities by the Company and certain other events. The Warrants do not contain any preemptive rights. The Warrants also contain certain restrictions on the ability to exercise the Warrants in the event that such exercise would result in the holder of the Warrants owning greater than 4.99% of the shares of the Company’s outstanding Class A Common Stock after giving effect to the exercise. The Warrants are exercisable solely on a cashless exercise basis under which in lieu of paying the exercise price in cash, the holders will be deemed to have surrendered a number of shares of Class A Common Stock with market value equal to the exercise price and will be entitled to receive a net amount of shares of Class A Common Stock after reduction for the shares deemed surrendered. In connection with the issuance of the Warrants, the Company agreed to register up to 20,038,410 shares of our Class A Common Stock issuable upon the exercise of the Warrants.
The Multi-Draw Term Loan, as amended, provided for a total commitment of $118 million. If entered into, the Multi-Draw Term Loan, as amended, would have refinanced the Bridge Loan in full and would have provided $70 million of additional financing consisting of (1) a $30 million tranche B-2 term loan and (2) a $40 million tranche B-3 term loan. The withdrawal schedule under the Multi-Draw Term Loan was revised to allow for release of funds from controlled accounts on the closing date sufficient to repay the Bridge Loan and future draws against the Multi-Draw Term Loan, subject to achievement of certain Makena™ related milestones, of $15 million in March 2011, $15 million in May 2011 and $10 million in each of July, August, September and October 2011. The commitment letter for the Multi-Draw Term Loan would have expired on March 31, 2011.
On February 17, 2011, the Company repaid a portion of the Bridge Loan with proceeds from a private placement of Class A Common Stock, described below, and on March 17, 2011, the Company repaid in full the remaining obligations under the Bridge Loan (including the payment of related premiums) with a portion of the proceeds of the offering of the 2015 Notes, described below (and terminated the related future loan commitments).
In accounting for the Warrants as required under ASC Topic 815, and as a result of the repayment of the Bridge Loan with the issuance of the 2015 Notes on March 17, 2011 and $7.5 million placed in escrow, as described above, the Warrants were recorded as a liability of $175.5 million. The Company then recorded, at fair value, an unrealized gain of $70.7 million for the period ended March 31, 2011 related to the Warrants resulting in a net liability of $112.3 million. Fair value of the Warrants were recorded utilizing a Black-Scholes option pricing model with the following assumptions: risk free rate of 1.8% and 2.1% at March 17, 2011 and March 31, 2011, respectively; expected volatility of 99.0% for both periods; expected dividend yield of $0 for both periods; and an approximate expected life of five years for both periods.
Private Placement of Class A Common Stock
On February 14, 2011, the Company announced that it entered into a definitive agreement with a group of institutional investors to raise approximately $32.3 million of gross proceeds from a private placement of 9.95 million shares of its Class A Common Stock at $3.25 per share. The transaction closed on February 17, 2011. The Company used $20 million of the proceeds from the financing to repay certain outstanding amounts and other outstanding obligations under its credit agreement with U.S. Healthcare. The remaining amount is being used for the launch of Makena™, payment of expenses associated with the private placement and general corporate purposes.
15
Table of Contents
The Company will be required to pay certain cash amounts as liquidated damages at a rate of 1.5% per month of the aggregate purchase price of the shares that are unregistered if it does not meet certain obligations under the agreement with respect to the registration of shares.
Private Placement of the 2015 Notes
On March 17, 2011, the Company completed the offering and sale of the 2015 Notes.
The 2015 Notes bear interest at an annual rate of 12% per year, payable semiannually in arrears on March 15 and September 15 of each year, commencing September 15, 2011. The 2015 Notes will mature March 15, 2015. At any time prior to March 15, 2013, the Company may redeem up to 35% of the aggregate principal amount of the 2015 Notes at a redemption price of 112% of the principal amount of the 2015 Notes, plus accrued and unpaid interest to the redemption date, with the net cash proceeds of one or more equity offerings. At any time prior to March 15, 2013, the Company may redeem all or part of the 2015 Notes at a redemption price equal to (1) the sum of the present value, discounted to the redemption date, of (i) a cash payment to be made on March 15, 2013 of 109% of the principal amount of the 2015 Notes, and (ii) each interest payment that is scheduled to be made on or after the redemption date and on or before March 15, 2013, plus (2) accrued and unpaid interest to the redemption date. At any time after March 15, 2013 and before March 15, 2014, the Company may redeem all or any portion of the 2015 Notes at a redemption price of 109% of the principal amount of the 2015 Notes, plus accrued and unpaid interest to the redemption date. At any time after March 15, 2014, the Company may redeem all or any portion of the 2015 Notes at a redemption price of 100% of the principal amount of the 2015 Notes, plus accrued and unpaid interest to the redemption date. The 2015 Notes are secured by the assets of the Company and certain assets of its subsidiaries.
After an original issue discount of 3%, the Company received proceeds of $218.3 million which were used to fund a first-year interest reserve totaling $27.0 million, repay all existing obligations to U.S. Healthcare totaling approximately $61.1 million and pay fees and expenses associated with the offering of the 2015 Notes of approximately $10.0 million. In connection with these payments, the Company also terminated all future loan commitments with U.S. Healthcare. The remaining proceeds, totaling approximately $120 million will be used for general corporate purposes, including the launch of Makena™.
The 2015 Notes were offered only to accredited investors pursuant to Regulation D under the Securities Act of 1933, as amended.
Ability to Continue as a Going Concern
There is substantial doubt about our ability to continue as a going concern. Our Consolidated Financial Statements included in this Report are prepared using accounting principles generally accepted in the United States applicable to a going concern, which contemplates the realization of assets and liquidation of liabilities in the normal course of business. The historical consolidated financial statements included in this Report do not include any adjustments that might be necessary if we are unable to continue as a going concern. The report of our independent registered public accountants BDO USA, LLP, included in this Report, includes an explanatory paragraph related to our ability to continue as a going concern.
The assessment of our ability to continue as a going concern was made by management considering, among other factors: (1) the timing and number of approved products that will be reintroduced to the market and the related costs; (2) the suspension of shipment of all products manufactured by us and the requirements under the consent decree with the FDA; (3) the possibility that we may need to obtain additional capital despite the proceeds from the offering of the 2015 Notes in March 2011 and the equity we were able to issue in February 2011 (see Note 1—“Description of Business—Significant Developments” of the Notes to the Consolidated Financial Statements included in this Report); (4) the potential outcome with respect to the governmental
16
Table of Contents
inquiries, litigation or other matters described in Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report; (5) our ability to comply with debt covenants; and (6) our ability to obtain future revenues from the sales of Makena™ sufficient to meet our future needs and expectations. Our assessment was further affected by our fiscal year 2011 net loss of $174.0 million, our 2010 net loss of $283.6 million, and the outstanding balance of cash and cash equivalents of $137.6 million as of March 31, 2011. For periods subsequent to March 31, 2011, we expect losses to continue because we are unable to generate any significant revenues from more of our own manufactured products until we are able to begin selling more of our approved products with FDA’s approval and with respect to products manufactured for us by third parties until after we are able to generate significant sales of Makena™ which was approved by the FDA in February 2011. We received notification from the FDA on September 8, 2010 of approval for our discontinued generics operations to ship into the marketplace the first product approved under the consent decree, i.e., the Potassium Chloride Extended Release Capsule. We resumed shipment of extended-release potassium chloride capsule, Micro-K® 10mEq and Micro-K® 8mEq, in September 2010, resumed shipments of the generic version of Potassium Chloride Extended Release Capsule in December 2010 and we began shipping Makena™ in March 2011. We have continued to ship Evamist®, which is manufactured for the Company by a third party, during the period covered by the consent decree. We are continuing to prepare other products for FDA inspection and do not expect to resume shipping other products until prior to the end of calendar year 2011, at the earliest. In addition, we must meet ongoing operating costs as well as costs related to the steps we are currently taking to introduce Makena™, and to prepare for introducing and reintroducing other approved products to the market. If we are not able to obtain the FDA’s clearance to resume manufacturing and distribution of more of our approved products in a timely manner and at a reasonable cost, or if revenues from the sale of approved products introduced or reintroduced into the market place prove to be insufficient, our financial position, results of operations, cash flows and liquidity will continue to be materially adversely affected. These conditions raise substantial doubt about our ability to continue as a going concern.
Based on current financial projections, we believe the continuation of our Company as a going concern is primarily dependent on our ability to address, among other factors: (1) sales of Makena™, which was approved by the FDA in February 2011 notwithstanding recent actions by the FDA to permit continued sale of compounded alternatives and by CMS to permit compounded alternatives to be reimbursed under the Medicaid program; (2) the timing, number and revenue generation of approved products that will be introduced or reintroduced to the market and the related costs; (3) the suspension of shipment of all products manufactured by us and the requirements under the consent decree with the FDA (other than the Potassium Chloride Extended Release Capsule, including Micro-K® 10mEq and Micro-K® 8mEq, products that are the subject of the FDA notification letter discussed above); (4) the possibility that we will need to obtain additional capital; (5) the potential outcome with respect to the governmental inquiries, litigation or other matters described in Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report; and (6) compliance with our debt covenants. While we address these matters, we must continue to meet expected near-term obligations, including normal course operating cash requirements and costs associated with introducing or reintroducing approved products to the market (such as costs related to our employees, facilities and FDA compliance), remaining payments associated with the acquisition and retention of the rights to Makena™ (see Note 5—“Acquisition” of the Notes to Consolidated Financial Statements included in this Report), the financial obligations pursuant to the plea agreement, costs associated with our legal counsel and consultant fees, as well as the significant costs associated with the steps taken by us in connection with the consent decree and the litigation and governmental inquiries. If we are not able to obtain the FDA’s clearance to resume manufacturing and distribution of certain or many of our approved products in a timely manner and at a reasonable cost and/or if we are unable to successfully commercialize Makena™, and/or if we experience adverse outcomes with respect to any of the governmental inquiries or litigation described in Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report, our financial position, results of operations, cash flows and liquidity will continue to be materially adversely affected. See “Risk Factors” included in Item I, Part 1A of this Report regarding additional risks we face with respect to these matters.
17
Table of Contents
In the near term, we are focused on performing the following: (1) addressing the FDA’s concerns regarding patient access to MakenaTM and the Center for Medicare and Medicaid Services’ (“CMS”) policy permitting Medicaid program reimbursement of compounded products; (2) the continued commercial launch of Makena™; (3) meeting the requirements of the consent decree, which will allow our approved products to be reintroduced to the market (other than the Potassium Chloride Extended Release Capsule products, including Micro-K® 10mEq and Micro-K® 8mEq, that are the subject of the FDA notification letter previously discussed); (4) the divestiture of Nesher and the Company’s generics business; and (5) pursuing various means to minimize operating costs and increase cash. Since December 31, 2010, the Company has generated non-recurring cash proceeds to support its on-going operating and compliance requirements from a $32.3 million private placement of Class A Common Stock in February 2011 and the 2015 Notes in March 2011 (which were used, in part, to repay all existing obligations under the agreement with U.S. Healthcare) (see Note 13—“Long-Term Debt” of the Notes to Consolidated Financial Statements included in this Report for a description of the 2015 Notes and U.S. Healthcare loan). While the cash proceeds received to date were sufficient to meet near-term cash requirements, we are pursuing ongoing efforts to increase cash, including, the continued implementation of cost savings, the divestiture of Nesher and the Company’s generics business and other assets and the return of certain additional approved products to market in a timely manner. We cannot provide assurance that we will be able to realize additional cost reductions from reducing our operations, that some or many of our approved products can be returned to the market in a timely manner (other than the Potassium Chloride Extended Release Capsule, including Micro-K® 10mEq and Micro-K® 8mEq products that are the subject of the FDA notification letter previously discussed above), that our higher profit approved products will return to the market in the near term or at all or that we can obtain additional cash through asset sales or the sale of equity or the successful commercial launch of Makena™. If we are unsuccessful in our efforts to introduce or return our products to market at adequate levels, or to sell assets or raise additional equity, we may be required to further reduce our operations, including further reductions of our employee base, or we may be required to cease certain or all of our operations in order to offset the lack of available funding.
We continue to evaluate the sale of certain of our assets, including Nesher and our generics business, which is included in discontinued operations and assets held for sale. To date, we are in advanced discussions for the sale of our generics business, however, we cannot assure that we will complete any transaction on favorable terms or at all. The Company is continuing to work with its advisors and several interested parties to complete a transaction. However, due to general economic conditions, we will likely be exposed to risks related to the overall macro-economic environment, including a lower rate of return than we have historically experienced on our invested assets and being limited in our ability to sell assets. In addition, we cannot provide any assurance that we ultimately will be successful in finding suitable purchasers for the sale of such assets. Even if we are able to find purchasers, we may not be able to obtain attractive terms and conditions for such sales, including attractive pricing. In addition, divestitures of businesses involve a number of risks, including the diversion of management and employee attention, significant costs and expenses, the loss of customer relationships, a decrease in revenues and earnings associated with the divested business, and the disruption of operations in the affected business. In addition, divestitures potentially involve significant post-closing separation activities, which could involve the expenditure of significant financial and employee resources. Inability to consummate identified asset sales or manage the post-separation transition arrangements could adversely affect our business, financial condition, results of operations and cash flows.
(c) Industry Segments
We have historically operated in three industry segments consisting of branded products, specialty generics and specialty raw materials. Prior to the consent decree, we derived revenues primarily from directly marketing our own technologically distinguished brand-name and generic/non-branded products and products marketed under joint development agreements with other companies. Revenues may also be received in the form of licensing revenues and/or royalty payments based upon a percentage of the licensee’s sales of the product when marketing rights to products using our drug delivery technologies are licensed. In March 2009, because of liquidity concerns and expected near-term cash requirements, the Board approved the divestiture of PDI, our
18
Table of Contents
specialty raw materials segment. As a result of the decision to sell PDI, we identified the assets and liabilities of PDI as held for sale at March 31, 2010 and have segregated PDI’s operating results and presented them separately as a discontinued operation for all periods presented. On June 2, 2010, we completed the sale of certain assets associated with the business of PDI.
In connection with the previously anticipated exclusion of ETHEX from participation in federal healthcare programs, we ceased operations of ETHEX on March 2, 2010. In May 2010, we formed a wholly-owned subsidiary, Nesher, to operate as the sales and marketing company for our generic products, and in July 2010, our Board of Directors directed management to explore strategic alternatives with respect to Nesher and the assets and operations of our generic products business. In the fourth quarter of fiscal year 2011, management committed to a plan to divest the generics business. We have retained Jefferies & Co to advise us with this strategy, which could include a sale of Nesher to a third-party. As a result, we identified the assets and liabilities of our generics business at March 31, 2011 and 2010 and have segregated the generics business operating results and presented them separately as discontinued operations for all periods presented. In the meantime, we will continue to prepare products for FDA inspection and continue to anticipate the reintroduction of approved products into the market.
See Note 4—“Restructuring and Impairment Charges,” and Note 23—“Divestitures,” of the Notes to Consolidated Financial Statements included in this Report for further discussion on PDI and our generics business.
(d) Narrative Description of Business
We are a fully integrated specialty pharmaceutical company that develops, manufactures, acquires and markets technologically-distinguished branded and generic/non-branded prescription pharmaceutical products. Our strategic goal is to become a specialty branded pharmaceutical marketing company primarily focused in women’s healthcare. We have a broad range of dosage form manufacturing capabilities, including tablets, capsules, creams and liquids. We conduct our branded pharmaceutical operations through Ther-Rx, and, previously, we conducted our generic/non-branded pharmaceutical operations through ETHEX, which focused principally on technologically-distinguished generic products prior to the cessation of its operations on March 2, 2010 and its dissolution on December 15, 2010. Through PDI, divested in June 2010, we developed, manufactured and marketed technologically advanced, value-added raw material products for the pharmaceutical industry and other markets. In May 2010, we formed a wholly-owned subsidiary, Nesher , to operate as the sales and marketing company for our generic products. The Company expects to complete a divestiture of Nesher and the Company’s generics business and assets by the end of calendar year 2011.
Our original strategy was to engage in the development of proprietary drug delivery systems and formulation technologies which enhance the effectiveness of new therapeutic agents and existing pharmaceutical products. Today we utilize one of those technologies, SITE RELEASE®, in two products expected to return to our branded portfolio in the second half of fiscal year 2012. Going forward, our business strategy will be primarily defined by the potential in-licensing and acquisition of pharmaceutical products rather than the historical strategy of the internal development of pharmaceutical products.
During fiscal year 2009, the Company announced six separate voluntary recalls of certain tablet form generic products as a precaution due to the potential existence of oversized tablets. In December 2008, the FDA began an inspection of the Company’s facilities. The Company suspended shipments of all approved tablet-form products in December 2008 and of all other drug products in January 2009. Also, in January 2009, the Company initiated a nationwide voluntary recall affecting most of its products. On March 2, 2009, the Company entered into a consent decree with the FDA regarding its drug manufacturing and distribution. The consent decree was entered by the U.S. District Court, Eastern District of Missouri, Eastern Division on March 6, 2009. As part of the consent decree, the Company agreed not to directly or indirectly do or cause the manufacture, processing, packing, labeling, holding, introduction or delivery for introduction into interstate commerce at or from any of its
19
Table of Contents
facilities of any drug, until the Company has satisfied certain requirements designed to demonstrate compliance with the FDA’s cGMP regulations. The consent decree provides for a series of measures that, when satisfied, will permit the Company to resume the manufacture and distribution of approved drug products. The Company has also agreed not to distribute its products that are not FDA approved, including its prenatal vitamins and hematinic products, unless it obtains FDA approval for such products through the FDA’s NDA and ANDA processes. These actions and the requirements under the consent decree have had, and are expected to continue to have, a material adverse effect on the Company’s liquidity position and its results of operations. In September 2010, the FDA approved the reopening of the Company’s manufacturing with respect to its first product, the Potassium Chloride Extended Release Capsule, which commenced sales in that month. Additional products are in the process of being brought back to market although the Company does not expect to resume shipping other products until later in fiscal year 2012, at the earliest.
We entered into a plea agreement with the Office of the United States Attorney for the Eastern District of Missouri and the Office of Consumer Litigation of the United States Department of Justice (referred to herein collectively as the “Department of Justice”) on March 2, 2010, pursuant to which ETHEX pleaded guilty to two felony counts, each stemming from the failure to make and submit a field alert report to the FDA in September 2008 regarding the discovery of certain undistributed tablets that failed to meet product specifications. In connection with the guilty plea by ETHEX, ETHEX was expected to be excluded from participation in federal healthcare programs, and in connection with the previously anticipated exclusion of ETHEX from participation in federal healthcare programs, we ceased operations of ETHEX on March 2, 2010 and dissolved it on December 15, 2010.
On November 15, 2010, we entered into a Divestiture Agreement with the Office of the Inspector General of the U.S. Department of Health and Human Services (“HHS OIG”) under which we agreed to sell the assets and operations of ETHEX to unrelated third parties by April 28, 2011 and to file articles of dissolution with respect to ETHEX under Missouri law by that date. We filed articles of dissolution for ETHEX on December 15, 2010. Following such filing, under the Divestiture Agreement, ETHEX may not engage in any new business other than for winding up its operations and will engage in a process provided under Missouri law to identify and resolve its liabilities over at least a two-year period. Under the terms of the agreement, HHS OIG agreed not to exclude ETHEX from federal healthcare programs until April 28, 2011 and, upon completion of the sale of the ETHEX assets and of the filing of the articles of dissolution of ETHEX, the agreement will terminate. Civil monetary penalties and exclusion of ETHEX could have occurred if we had failed to meet our April 28, 2011 deadline. The sales of ETHEX’s assets (other than certain fixtures as to which HHS OIG has consented to non-divestiture) were completed prior to the April 28, 2011 deadline and ETHEX no longer has any material ongoing assets or operations other than those required to conclude the winding up process under Missouri law. We have also received a letter from HHS OIG advising us further that assuming that we have complied with all agreements deemed necessary by HHS OIG, HHS OIG would not exclude ETHEX thereafter. On April 1, 2011, we requested confirmation from HHS OIG that the steps and actions described in our reports to HHS OIG constituted full compliance with the duties KV and ETHEX were to complete by April 28, 2011 under the Divestiture Agreement, such that they are not at risk for stipulated penalties or exclusion after that date. On April 8, 2011, we received a letter from HHS OIG stating that, at this time, based upon the information provided to HHS OIG in our monthly submissions, HHS OIG has no concerns regarding the actions taken by KV and ETHEX pursuant to the Divestiture Agreement. On May 20, 2011, we received a letter from HHS OIG stating that based on its review of the information provided in our monthly reports, it appeared that the Company and ETHEX had completed our obligations under the Divestiture Agreement.
In light of the developments described above, the report of our independent registered public accountants included a statement regarding our ability to continue as a going concern. See the Consolidated Financial Statements included in this Report.
20
Table of Contents
Our Business Today
Because of the steps taken by us with respect to the nationwide recall and suspension of shipment of all products manufactured by us (other than extended-release potassium chloride capsule, Micro-K® 10mEq and Micro-K® 8mEq, Potassium Chloride Extended Release Capsule) and Makena™ and Evamist® (which are manufactured by third parties), the requirements under the consent decree, certain consequences resulting from the entry into the plea agreement and the Divestiture Agreement and the ongoing private litigation and governmental inquiries, we have realigned our operations and business to concentrate our efforts on maintaining and attempting to increase our limited cash and financial resources and expeditiously reintroducing certain approved products to the market. To that end, we have focused our recent efforts as follows:
| • | We have made the strategic decision to tie our future to the development and commercialization of branded specialty pharmaceuticals focused on women’s health. In the near term, the Company is focused on the continuing commercial launch of Makena™ (hydroxyprogesterone caproate injection), which was approved by the FDA in February 2011 and began shipping in March 2011. |
| • | In May 2010, we formed Nesher to operate as the sales and marketing company for our generic products. As we continue to move forward to the approval of additional generic products for re-launch, in July 2010, our Board of Directors directed management to explore strategic alternatives with respect to Nesher and the assets and operations of our generic products business and in the fourth quarter of fiscal year 2011, management committed to a plan to divest the generics business. In the meantime, we will continue to prepare products for FDA inspection and reintroduction of approved products into the market. |
| • | The Company received notification from the FDA on September 8, 2010 of approval for our discontinued generics operations to ship into the marketplace the first product approved under the consent decree, i.e., the Potassium Chloride Extended Release Capsule. The Company resumed shipment of extended-release potassium chloride capsule, Micro-K® 10mEq and Micro-K® 8mEq, in September 2010 and resumed shipments of its generic version, Potassium Chloride Extended Release Capsule, in December 2010. The Company is continuing to prepare other products for FDA inspection. We are currently working with Lachman, an independent cGMP expert retained by us pursuant to the consent decree, to meet the requirements set forth in the consent decree. We do not expect to generate any significant revenues from products that we manufacture until we can resume shipping more of our approved products. We do not expect to resume shipping other products that we manufacture until prior to the end of calendar year 2011, at the earliest. We do not expect to generate any significant revenues from products that we have manufactured by third parties until and unless we begin to generate significant revenues from sales of Makena™ along with our current revenues from the sales of Evamist®. |
| • | In order to address liquidity concerns, the Company secured additional capital including the following: |
| • | On February 17, 2011, the Company closed on a private placement with the selling stockholders of 9.95 million shares of its Class A Common Stock at $3.25 per share to raise approximately $32.3 million of gross proceeds. The Company used $20 million of the proceeds from the financing to repay certain outstanding amounts and other outstanding obligations under its existing credit agreement with U.S. Healthcare. The remaining funds will be used for continuing the commercial launch of Makena™, payment of expenses associated with the private placement and general corporate purposes. |
| • | On March 17, 2011, the Company completed a private placement with a group of institutional investors of the 2015 Notes. After an original issue discount of 3%, the Company received proceeds of $218.3 million which were used to fund a first-year interest reserve totaling $27.0 million, repay all existing obligations to U.S. Healthcare and pay fees and expenses associated with the offering of the 2015 Notes of approximately $10.0 million. The remaining proceeds, totaling approximately $120 million, will be used for general corporate purposes, including continuing the commercial launch of Makena™. |
21
Table of Contents
While these cash proceeds are expected to be sufficient to meet near-term cash requirements, we are pursuing ongoing efforts to increase cash, including, but not limited to the continued implementation of cost savings, the divestiture of Nesher and the Company’s generics business and other assets and the return of certain of our approved products to market (in addition to the Potassium Chloride Extended Release Capsule, including Micro-K® 10mEq and Micro-K® 8mEq products that are the subject of the FDA notification letter previously discussed).
| • | Our restructuring efforts also have included an updated criteria-based review of the pipeline of products we had under development. Product candidates in our development portfolio were evaluated based on factors with respect to each product that included, among other things, market potential, a revised return on investment profile, probability of clinical success, time and cost of development, synergies with our core competencies, and competitive landscape. Based on this evaluation and due to the restructuring effort undertaken to adjust our infrastructure and scale of operations (including a reduction in headcount of approximately 80 employees on March 31, 2010), we realigned our research and development efforts to focus on projects that we believe have the highest potential return on investment in areas focused on women’s healthcare for our branded business. |
| • | We continue to review and modify our quality monitoring and testing programs and procedures in our effort to comply with the terms of the consent decree and cGMP regulations. To that end, we have implemented a series of procedures, which we believe will enhance quality standards in our products. Our goal is, and will continue to be, to enhance the quality, purity, safety and efficacy of each of our products. We believe that adherence to high quality standards can also promote a more efficient utilization of our personnel, materials and production capacity. |
Despite our efforts, however, there can be no assurance that these or other initiatives intended to enable us to reintroduce additional approved products to the market or sell selected assets or businesses will be successful within the time frames currently projected by management or at all.
Ther-Rx—History of Our Branded Products Segment
We established Ther-Rx in 1999 to market brand name prescription pharmaceutical products that incorporated our proprietary technologies. Since its inception, Ther-Rx has generally focused on therapeutic areas within women’s healthcare. By targeting specialized physicians such as Obstetrician/Gynecologists, Ther-Rx has been able to leverage the cost efficiencies of a focused specialty sales force while developing stronger relationships with key prescribers. This strategy coupled with innovative products resulted in the growth of our business to a peak in net revenues of $212.3 million in fiscal year 2008. Due to a nationwide recall and suspension of shipment of all products manufactured by us in fiscal year 2009, as well as entering into a consent decree with the FDA, Ther-Rx’s net revenues declined to $113.2 million in fiscal year 2009, and to $27.3 million in fiscal year 2011.
As more of our approved and Company-manufactured products return to market, we plan to continue focusing our sales and marketing efforts in women’s healthcare. Ther-Rx maintains a corporate sales and marketing management team dedicated to planning and managing Ther-Rx’s sales and marketing efforts. Ther-Rx currently markets Evamist®, a unique transdermal estrogen therapy delivering a low dose of estradiol in a once-daily spray indicated for the treatment of moderate-to-severe vasomotor symptoms due to menopause. Because Evamist® is manufactured by a third-party, we have been able to continue marketing and selling Evamist® pursuant to the terms of the consent decree. Net revenues of Evamist® have grown from $2.6 million in fiscal year 2009 to $8.6 million in fiscal year 2010 and to $13.0 million in fiscal 2011, despite a significant reduction in sales force and marketing- related expenses associated with the recall and suspension of shipments of Ther-Rx’s other products.
In September 2010, we received notification from the FDA of approval under the consent decree to resume shipment into the marketplace of the first of our approved products. Initial shipments of Micro-K® 10mEq and
22
Table of Contents
Micro-K® 8mEq began in September 2010. The FDA is expected to conduct additional inspections with respect to other approved products before deciding whether we may resume manufacturing and marketing such products. Among the products that will be reviewed for re-introduction to the market are Clindesse® and Gynazole-1®, Ther-Rx’s vaginal anti-infective products for bacterial vaginosis and vaginal yeast infections, respectively. If approved for return to market, these products have the potential to contribute significantly to the rebuilding of our branded business.
In February 2011, the FDA approved MakenaTM, a drug that reduces the risk of preterm birth for women with a singleton pregnancy who have a history of singleton spontaneous preterm birth. We believe that the approval and introduction of MakenaTM provides important benefits to women who are eligible for MakenaTM, including:
| • | Manufactured using methods that ensure purity of ingredients, potency of product, and consistency from dose to dose |
| • | Produced in a facility that complies with the FDA’s Good Manufacturing Practices (GMP) requirements and has required periodic FDA inspections |
| • | Dispensed with complete and consistent prescribing information that has been reviewed and approved by the FDA, including patient education materials |
| • | Post-marketing surveillance is conducted to document, analyze, and report adverse events to the FDA |
| • | Standardized distribution across the country |
| • | Financial assistance programs available for both insured and uninsured patients, with no household income cap |
Additionally, Ther-Rx established the MakenaTM Care Connection, a support program for patients and healthcare providers that provides administrative, financial assistance and treatment support for MakenaTM. Administrative and treatment support includes insurance benefit investigation, educational information and a compliance reminder program. Because specialty injectable products like MakenaTM are not typically carried by retail pharmacies, the process for facilitating prescriptions for MakenaTM is managed by this dedicated customer support center.
Ther-Rx has also established a Patient Assistance Program for MakenaTM that provides co-pay assistance (for insured patients), and financial assistance (for uninsured patients). Under the program, patients with a household income of up to $120,000 will pay $20 or less per injection for MakenaTM. This encompasses 85% of the U.S. based on 2009 U.S. census data. Clinically-eligible patients who are uninsured and whose financial need is greatest will receive MakenaTM at no cost. There are no upper-level income caps to qualify for the patient assistance program. Ther-Rx began shipping MakenaTM in March 2011.
Since the initial shipment of vials of MakenaTM in March 2011, Ther-Rx has tracked and accumulated the following key performance metrics (all information is cumulative through May 31, 2011):
| • | Approximately 6,500 vials have been shipped to Ther-Rx customers, including approximately 200 vials that shipped subsequent to March 31, 2011. |
| • | Approximately 1,200 vials have been shipped to doctors and patients. Approximately 15% of these vials were provided at no cost to patients who have demonstrated financial need. Of the remaining 85%, approximately two thirds of the vials were shipped to patients covered by commercial insurance plans and one third were shipped to patients covered by a government insurance plan. |
| • | Approximately 1,700 patient referrals have been made to the MakenaTM Care Connection, of which approximately: |
| • | 900 patients have initiated treatment or are in the enrollment phase or are pending insurance approval and treatment initiation. |
23
Table of Contents
| • | The remaining balance of these referrals (approximately 800) did not lead to filled Makena prescriptions for a variety of reasons including; not meeting the labeled indication, some of which were because they were outside the treatment initiation window and others who did not have the proper obstetric history or were pregnant with multiples; cancellation prior to completion of the insurance benefits verification process either by prescribers or patients for unspecified reasons; or not receiving positive insurance coverage, most of which were referrals for Medicaid patients. |
| • | The data that is available to us indicates that patient co-pays are averaging approximately, $12 per injection including more than 36% of insured patients that had a $0 co-pay. Further, of the patients that have had a co-pay assigned by their insurance plan, 90% of those patients have had an average co-pay of about $9 per injection. This data does not include patients who have benefitted from our financial assistance programs. |
| • | Over 100 different payors (both commercial and Medicaid) have approved at least one Makena prescription. |
Ther-Rx is continuing to work with commercial insurance programs and state Medicaid agencies to increase coverage of and access to MakenaTM. These on-going discussions include negotiations regarding rebates and patient coverage levels, none of which have been finalized as of May 31, 2011.
Ther-Rx is also working towards the re-launch of its anti-infective cream products, Clindesse® and Gynazole-1®. The Company stopped manufacturing these products in the fourth quarter of fiscal year 2009 when it entered into the consent decree. In February 2011, the FDA conducted an inspection with respect to the Company’s Clindesse® product and issued a Form 483 Report with certain observations. On February 28, 2011, the Company filed its responses with the FDA with respect to such observations. We have advised the FDA of a required equipment change and we are moving forward with the appropriate validation protocol and would expect the FDA to conduct a follow-up inspection. As a result, the Company expects to relaunch Clindesse® and Gynazole-1® prior to the end of calendar year 2011.
ETHEX and Nesher—History of Our Discontinued Specialty Generic/Non-Branded Segment
We established ETHEX in 1990 to utilize our portfolio of drug delivery systems to develop and market hard-to-copy generic/non-branded pharmaceuticals. Due to the nationwide recall and suspension of shipment of all products manufactured by us, net revenues for ETHEX declined from $199.1 million in fiscal year 2009 to $143.2 million in fiscal year 2010 and to $8.7 million in fiscal year 2011. Due to the requirements under the consent decree with which we must comply before resuming manufacturing and shipping of our approved generics products, we currently do not generate significant revenues from our specialty generics segment.
In connection with the settlement of three patent infringement cases brought against us by Purdue Pharma on June 9, 2009, we and Purdue entered into a settlement agreement, patent license agreement and a distribution and supply agreement that provided us certain limited rights to sell generic versions of OxyContin® extended-release tablets in the United States. See Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report for more information regarding the litigation with Purdue and descriptions of the settlement agreement, the patent license agreement and the distribution and supply agreement. Substantially all of the $143.2 million of net revenues generated by ETHEX in fiscal year 2010 resulted from the sale of all of the generic OxyContin® allotted pursuant to the Distribution Agreement with Purdue.
In connection with the previously anticipated exclusion of ETHEX from participation in federal healthcare programs, we ceased the operations of ETHEX on March 2, 2010. However, we have retained the ability to manufacture (once the requirements under the consent decree have been met), market and distribute all generic products and are in possession of all intellectual property related to generic products, including all NDAs and ANDAs. We currently do not anticipate that the voluntary guilty plea by ETHEX will have a material adverse effect on our efforts to comply with the requirements pursuant to the consent decree and to resume production and shipments of our approved products.
24
Table of Contents
In May 2010 we formed a wholly-owned subsidiary, Nesher to operate as the sales and marketing company for our generic products. In July 2010, our Board of Directors directed management to explore strategic alternatives with respect to Nesher and the assets and operations of our generic products business and in the quarter ended March 31, 2011 management committed to a plan to divest the Company’s generics business. As a result, the Company reports its generics business as discontinued operations beginning in the quarter ended March 31, 2011. In the meantime, we will continue to prepare products for FDA inspection and continue to anticipate the reintroduction of approved products into the market.
On November 15, 2010, we entered into the Divestiture Agreement with HHS OIG under which we agreed to sell the assets and operations of ETHEX to unrelated third parties prior to April 28, 2011 and to file articles of dissolution with respect to ETHEX under Missouri law by that date. We filed articles of dissolution for ETHEX on December 15, 2010. Following the filing, ETHEX may not engage in any new business other than winding up its operations and will engage in a process provided under Missouri law to identify and resolve its liabilities over at least a two-year period. Under the terms of the Divestiture Agreement, HHS OIG agreed not to exclude ETHEX from federal healthcare programs until April 28, 2011 and, upon completion of the sale of the ETHEX assets and of the filing of the articles of dissolution of ETHEX, the agreement will terminate. Civil monetary penalties and exclusion of ETHEX could have occurred if we had failed to meet our April 28, 2011 deadline. The sales of ETHEX’s assets (other than certain fixtures as to which HHS OIG has consented to non-divestiture) were completed prior to the April 28, 2011 deadline and ETHEX no longer has any ongoing material assets or operations other than those required to conclude the winding up process under Missouri law. We have also received a letter from HHS OIG advising us further that assuming that we have complied with all agreements deemed necessary by HHS OIG, HHS OIG would not exclude ETHEX thereafter. On April 1, 2011, we requested confirmation from HHS OIG that the steps and actions described in our reports to HHS OIG constituted full compliance with the duties KV and ETHEX were to complete by April 28, 2011 under the Divestiture Agreement, such that they are not at risk for stipulated penalties or exclusion after that date under the Divestiture Agreement. On April 8, 2011, we received a letter from HHS OIG stating that, at this time, based upon the information provided to HHS OIG in our monthly submissions, HHS OIG has no concerns regarding the actions taken by KV and ETHEX pursuant to the Divestiture Agreement. On May 20, 2011, we received a letter from HHS OIG stating that based on its review of the information provided in our monthly reports, it appeared that the Company and ETHEX had completed our obligations under the Divestiture Agreement.
PDI—Our Discontinued Value-Added Raw Material Business
PDI develops and markets specialty value-added raw materials, including drugs, directly compressible and microencapsulated products, and other products used in the pharmaceutical industry and other markets. Its products include value-added active drug molecules, vitamins, minerals and other raw material ingredients that provide benefits such as improved taste, altered or controlled release profiles, enhanced product stability and efficiency and other manufacturing process advantages. PDI has also been a significant supplier of value-added raw materials for the development and manufacture of both existing and new products at Ther-Rx and ETHEX. A leased facility used by PDI was damaged by an accidental fire which occurred on June 1, 2009. The incident did not affect any of our finished product manufacturing, packaging, or distribution facilities. PDI’s primary business utilizes contract manufacturing, which was unaffected by the fire. As a result of the decision to sell PDI in March 2009, we identified the assets and liabilities of PDI as held for sale at March 31, 2010 and 2009 and have segregated PDI’s operating results and presented them separately as a discontinued operation for all periods presented in our financial statements.
On June 2, 2010, we completed the sale of certain assets associated with the business of PDI. See Note 23—“Divestitures” of the Notes to Consolidated Financial Statements included in this Report.
Our Business in the Future
Although subject to significant uncertainties as discussed in this Report, our goal is to again be a successful participant in the pharmaceutical industry and we are making substantial efforts to meet the requirements under the consent decree with the aim of expeditiously reintroducing certain approved products to the market.
25
Table of Contents
Assuming that we are successful with our efforts to meet the consent decree requirements and return certain of our approved products to the market, our long-term goals and strategies are planned to incorporate the following key elements:
| • | We plan to focus on projects specific to women’s healthcare that we believe have the highest potential return on investment (which we currently believe to be primarily Makena™), to streamline product lines, to implement Company-wide efficiencies and to make disciplined spending decisions. |
| • | We expect that our future product pipeline will consist of product opportunities resulting from both acquisitions and specifically targeted internal development efforts. We plan to optimize our product development portfolio by building on our core competencies in the area of women’s health for our branded business. We plan to continue to seek branded product acquisition opportunities that are complementary to our current and planned portfolio of women’s health products. |
| • | We expect that our planned portfolio of branded products will reflect our focus on promoting the health and well-being of women. We believe we can address this market in a cost effective way by targeting specialty physician groups with a streamlined specialty sales force. |
| • | We plan to divest our specialty generic/non-branded business. |
Despite our efforts, however, there can be no assurance that these initiatives will be successful. See “Risk Factors” in Part I, Item 1A of this Report.
Sales and Marketing
We have marketed products directly to wholesalers, distributors, retail pharmacy chains, mail order pharmacies and group purchasing organizations. We also have marketed our products indirectly to independent pharmacies, managed care organizations, hospitals, nursing homes, pharmacy benefit management companies and government entities. These customers, referred to as “indirect customers,” purchase our products primarily through our wholesale or distributor customers. We service these customers with an external sales force and internal marketing team.
In the past, we have been able to increase sales of our branded pharmaceutical products through physician sales calls and promotional efforts, including sampling, advertising and direct mail. By offering multiple products to the same group of specialty physicians, we have been able to optimize the effectiveness of our sales force. We plan to continue focusing our specialty-sized sales force on marketing our existing and future core of women’s healthcare products. Because our products are sold to highly targeted specialty physician groups that tend to be relatively concentrated, we believe we can address this market and future opportunities in a cost effective way with a specialty-sized sales force.
Ther-Rx also has a corporate sales and marketing management team dedicated to planning and managing Ther-Rx’s sales and marketing efforts. As part of an ongoing realignment of our cost structure relative to our inability to manufacture and ship products manufactured by us and the requirements under the consent decree, we have reduced our workforce to be in line with the anticipated level of ongoing business. Accordingly, Ther-Rx reduced its branded sales force from approximately 330 specialty sales representatives at December 31, 2008 to approximately 76 specialty sales representatives at March 31, 2010. Subsequent to the approval of MakenaTM, we substantially increased the size of our sales force to support the commercial launch of Makena™ and at March 31, 2011 have a total of approximately 150 sales representatives.
Although we have previously sold our products internationally, we have not had material operations or sales in foreign countries and our sales are not subject to significant geographic concentration outside of the United States.
26
Table of Contents
Research and Development
Our research and development activities have historically included the development of new drug delivery technologies, the formulation of brand name proprietary products and the development of generic versions of previously approved brand name pharmaceutical products. In fiscal years 2011 and 2010, total research and development expenses were $22.1 million and $29.1 million, respectively, excluding acquired in-process research and development.
Our ability to internally develop new products was limited in fiscal year 2010 and 2011 as we concentrated on meeting the requirements of the consent decree. Until the successful FDA inspection in September 2010, we did not have approved cGMP systems to use to manufacture drugs for clinical trials in our own facilities. The consent decree has always allowed us to manufacture drugs for non-clinical laboratory studies or other research and testing that does not involve exposure of human research subjects. Our internal R&D resources in fiscal years 2010 and 2011 were concentrated on assisting the return of previously approved products to the market following the FDA-approved process.
Our restructuring efforts included a criteria-based review of the pipeline of products we had under development at the time of the consent decree. Product candidates in our development portfolio were evaluated based on market potential, probability of clinical success, time and cost of development, and the competitive effect of our inability to progress the internal programs significantly in fiscal years 2009 through 2011. Cost containment efforts also required a reduction in the number of research and development personnel, from approximately 200 to approximately 23 further reducing our ability to progress multiple programs. We realigned our research and development efforts to focus on fewer projects that we believe have the highest potential return on investment in areas such as women’s healthcare for our branded business and complex extended-release products for our generic business.
The focus of our new drug development in fiscal year 2010 and 2011 was working with Hologic to enroll patients in the Makena™ confirmatory post-marketing studies, also known as Phase IV studies, to complete the resubmission to the FDA of this drug for the prevention of preterm birth in women with a history of spontaneous preterm birth. This Complete Response was submitted to the FDA on July 13, 2010 and as noted above, the NDA was approved on February 3, 2011. In addition there are still generic ANDAs under review at the FDA from our Company. Future efforts will focus on niche specialty products that complement the existing women’s health portfolio, through either acquisition or internal development.
Patents and Other Proprietary Rights
When appropriate and available, we actively seek protection for our products and proprietary information by means of U.S. and foreign patents, trademarks, trade secrets, copyrights and contractual arrangements. Patent protection in the pharmaceutical field involves complex legal and factual issues. Moreover, broad patent protection for new formulations or new methods of use of existing chemical compounds is sometimes difficult to obtain, primarily because the active ingredient and many of the formulation techniques have been known for some time. Consequently, some patents claiming new formulations or new methods of use for old drugs may not provide adequate protection against competition. Nevertheless, we intend to continue to seek patent protection when appropriate and available and otherwise to rely on regulatory-related exclusivity and trade secrets to protect certain of our products, technologies and other scientific information. There can be no assurance that any steps taken to protect such proprietary information will be effective.
Our policy is to file patent applications in appropriate situations to protect and preserve, for our own use, technology, inventions and improvements that we consider important to the development of our business. As business conditions change, however, we maintain our ability to quickly adapt by focusing resources on those patent applications that we deem to provide the greatest value or potential value. We currently hold domestic and foreign issued patents, the last of which expires in fiscal year 2027, relating to our controlled-release, site-specific, quick dissolve, and vitamin absorption technologies. We own or hold licenses to 34 U.S. patents and have 17 U.S. patent applications pending. In addition, we have approximately 35 foreign patents and numerous
27
Table of Contents
foreign patents applications pending primarily in Canada, Europe, Australia, Japan, South America, Mexico and South Korea. We depend on our patents and other proprietary rights, although we cannot be certain of their confidentiality, validity and protection (see also “Risk Factors” in Part I, Item 1A).
We currently own more than 300 U.S. and foreign trademark applications and registrations, including trademark protection for certain names of our proprietary controlled-release, taste masking, site-specific and quick dissolve technologies. We intend to continue to seek trademark protection covering new technology and product names as they are developed.
To protect our trademark, domain name, and related rights, we generally rely on trademark and unfair competition laws, which are subject to change. Some, but not all, of our trademarks are registered in the jurisdictions where they are used. Some of our other trademarks are the subject of pending applications in the jurisdictions where they are used or intended to be used and others are not.
Manufacturing and Facilities
All of our facilities at March 31, 2011, aggregating approximately 1.1 million square feet, are located in the St. Louis, Missouri metropolitan area. We own facilities with approximately 1.0 million square feet, with the balance under various leases at pre-determined annual rates under agreements expiring from fiscal year 2012 through fiscal year 2017, subject in most cases to renewal at our option. See Part I Item 2—“Properties” in this Report for more information. Substantially all our real estate interests are pledged to secure our indebtedness.
We are required to maintain all facilities in compliance with the FDA’s cGMP requirements. In addition to compliance with cGMP, each pharmaceutical manufacturer’s facilities must be registered with the FDA. We are required to be registered with the Drug Enforcement Agency (the “DEA”), and similar state and local regulatory authorities because we handle controlled substances. We are also subject to the Environmental Protection Agency (the “EPA”), and similar state and local regulatory authorities if we generate toxic or dangerous wastes. In addition, we must comply with other applicable DEA and EPA requirements. Noncompliance with applicable legal and regulatory requirements can have a broad range of consequences, including warning letters, fines, seizure of products, product recalls, total or partial suspension of production and distribution, refusal to approve NDAs, ANDAs or other applications or revocation of approvals previously granted, withdrawal of product from marketing, injunction, withdrawal of licenses or registrations necessary to conduct business, disqualification from supply contracts with the government, and criminal prosecution. Additionally, under certain circumstances, the FDA also has the authority to revoke drug approvals previously granted.
As previously noted, on March 2, 2009, we entered into a consent decree with the FDA regarding our drug manufacturing and distribution. The consent decree was entered by the U.S. District Court, Eastern District of Missouri, Eastern Division on March 6, 2009 and continues for a period of six years following satisfaction of certain obligations contained in the consent decree after which the Company may petition the Court for relief from the consent decree. As part of the consent decree, we have agreed not to directly or indirectly do or cause the manufacture, processing, packing, labeling, holding, introduction or delivery for introduction into interstate commerce at or from any of our facilities of any drug, until we have satisfied certain requirements designed to demonstrate compliance with the FDA’s cGMP regulations. The consent decree provides for a series of measures that, when satisfied, will permit us to resume the manufacture and distribution of approved drug products marketed by our generic and branded subsidiaries. The consent decree further provides that, before resuming manufacturing, we will retain and have an independent cGMP expert undertake a review of our facilities and certify compliance with the FDA’s cGMP regulations. Following that certification, the FDA was to make a determination as to whether our facilities are in compliance. On January 13, 2010, our independent cGMP expert, Lachman, notified the FDA that Lachman had performed a comprehensive inspection and that our facilities and controls are in compliance with cGMP and the consent decree, but advised us to enact further enhancements to certain aspects of our cGMP systems. In accordance with the advice from Lachman, we continued to enhance our cGMP systems, and Lachman subsequently reinspected our cGMP systems and on April 26, 2010 certified our compliance with all cGMP systems requirements.
28
Table of Contents
The next step in the process for resumption of product shipment was for Lachman to certify individual products manufactured under the newly certified cGMP systems. We completed the manufacture of validation batches of the first product, the Potassium Chloride Extended Release Capsule, which were successfully completed through Lachman review on July 27, 2010.
Lachman certified the manufacture of the product on July 27, 2010, and during the week of August 16, 2010 the FDA subsequently conducted its own inspection of our facilities, systems and processes as outlined in the consent decree and found no adverse findings. The Company received notification from the FDA on September 8, 2010 of approval for our discontinued generics operations under the consent decree to ship into the marketplace the first of our approved products, i.e., the Potassium Chloride Extended Release Capsule. We resumed shipment of extended-release potassium chloride capsule, Micro-K® 10mEq and Micro-K® 8mEq, in September 2010 and we began shipping Makena™ in March 2011. We have continued sales of Evamist®, manufactured for us by a third party manufacturer, during the duration of the consent decree. We are continuing to prepare other products for FDA inspection although we do not expect to resume shipping other products until later in fiscal year 2012, at the earliest.
Competition
The primary means of competition in our industry are innovation and development, timely FDA approval, manufacturing capabilities, product quality, marketing, customer service, drug delivery systems, reputation and price. To compete effectively on the basis of price and remain profitable, a generic drug manufacturer must manufacture its products in a cost-effective manner. Our competitors include other specialty pharmaceutical companies, other generic and branded companies, and brand companies that, prior to patent expiration or as relevant patents expire, license their products to generic manufacturers for sale as what are commonly referred to as “authorized generics” and, to the extent permitted by the FDA and other federal and state agencies and boards, companies selling unapproved therapies or compounded drugs that compete against products sold by the Company and its subsidiaries, including Makena™. Further, regulatory approvals typically are not required for a brand manufacturer to sell its pharmaceutical products directly or through a third-party as authorized generics, nor do such manufacturers face any other significant barriers to entry into such market. It is possible, however, that even if we are able to return some or all of our approved products to the market, certain of our existing customers will purchase smaller quantities or no quantities of our products. Such a potential loss of market share would likely result in limiting the prices we are able to charge for our approved products, which will negatively impact our gross margin. Moreover, entering into the consent decree and our withdrawal and recall of most of our products during fiscal year 2009 may have damaged our reputation in the market, which could result in additional competitive disadvantages.
Competition in the development and marketing of pharmaceutical products is intense and characterized by extensive research efforts and rapid technological progress. Many companies, including those with financial and marketing resources and development capabilities substantially greater than our own, are engaged in developing, marketing and selling products that compete with those that we previously offered. Our branded pharmaceutical products have been subject to competition from alternative therapies during the period of patent protection and thereafter also from generic equivalents. In addition, our generic/non-branded pharmaceutical products have been subject to competition from pharmaceutical companies engaged in the development of alternatives to the generic/non-branded products we previously offered or have undertaken to develop. Our competitors may develop generic products before we do or may have pricing advantages over our products. In our specialty pharmaceutical businesses, we compete primarily on the basis of product efficacy and safety, breadth of product line, differentiated features of our products and price. We believe that, once we have satisfied the requirements of the consent decree, our patents, proprietary trade secrets, technological expertise, product development and manufacturing capabilities will enable us to develop products to compete effectively in the marketplace.
In addition, we have competed for product acquisitions with other pharmaceutical companies. We have also competed with drug delivery companies engaged in the development of alternative drug delivery systems. We are
29
Table of Contents
aware of a number of companies currently seeking to develop new non-invasive drug delivery systems, including oral delivery and transmucosal systems. Based on total assets, annual revenues and market capitalization, our branded and generic businesses are considerably smaller than many of our competitors and other national competitors in these product areas. Many of our competitors have been in business for a longer period of time, have a greater number of products on the market and have greater financial, marketing, managerial and other resources than we do, including greater research and development capabilities and experience. Accordingly, our competitors may succeed in developing competing technologies, obtaining FDA approval for products or gaining market acceptance more readily than we do. If we directly compete with them for the same markets and/or products, their financial strength could prevent us from capturing a meaningful share of those markets.
Government Regulation
General Overview
All pharmaceutical manufacturers are subject to extensive regulation by the federal government, principally the FDA, and, to a lesser extent, by state, local and foreign governments. The Federal Food, Drug and Cosmetic Act (“FDCA”) and other federal and state statutes and regulations govern or influence, among other things, the development, testing, manufacture, safety, labeling, storage, recordkeeping, approval, advertising, promotion, sale, import, export and distribution of pharmaceutical products. Pharmaceutical manufacturers are also subject to certain record-keeping and reporting requirements, establishment registration and product listing, and FDA inspections.
Manufacturers of controlled substances must also comply with the Controlled Substances Act of 1970 (“CSA”) and regulations promulgated by the DEA, as well as similar state and local regulatory requirements for manufacturing, distributing, testing, importing, exporting and handling controlled substances. Manufacturers that generate toxic or dangerous wastes also must comply with applicable environmental laws and their implementing regulations.
Noncompliance with applicable legal and regulatory requirements can have a broad range of consequences, including warning letters, fines, seizure of products, product recalls, total or partial suspension of production and distribution, refusal to approve NDAs, ANDAs or other applications or revocation of approvals previously granted, withdrawal of product from marketing, injunction, withdrawal of licenses or registrations necessary to conduct business, disqualification from supply contracts with the government, and criminal prosecution.
Product development and approval within the FDA regulatory framework take a number of years, involve the expenditure of substantial resources, and are uncertain. Many drug products ultimately do not reach the market because they are not found to be safe or effective or cannot meet the FDA’s other regulatory requirements. After a product is approved, the FDA may revoke or suspend the product approval if compliance with post-market regulatory standards is not maintained or if problems occur after the product reaches the marketplace. In addition, the FDA may require post-marketing studies to monitor the effect of approved products, and may limit further marketing of the product based on the results of these post-market studies or evidence of safety concerns. Further, the current regulatory framework may change and additional regulatory or approval requirements may arise at any stage of our product development that may affect approval, delay the submission or review of an application or require additional expenditures by us. We may not be able to obtain necessary regulatory clearances or approvals on a timely basis, if at all, for any of our products under development. Delays in receipt or failure to receive such clearances or approvals, the loss of previously received clearances or approvals, or failure to comply with existing or future regulatory requirements could have a material adverse effect on our business.
While the Company and its products are subject to rigorous regulation from the FDA, the FDA may elect to exercise enforcement discretion as to whether to enforce its laws and regulations against unapproved therapies and compounded drugs that may compete against the Company’s products, including Makena™. In March 2011,
30
Table of Contents
the FDA has communicated to the Company and also separately issued a press release that, in order to ensure continued access for patients needing 17-alpha hydroxyprogesterone caproate, the FDA intended to refrain at this time from taking enforcement action with respect to compounding pharmacies producing compounded 17-alpha hydroxyprogesterone caproate in response to individual prescriptions for individual patients. The impact of FDA’s statement on the effectiveness of the Company’s orphan drug marketing exclusivity is at present still unclear but a failure by the FDA to take enforcement action against compounding pharmacies may result in substantial sales of compounded alternatives to Makena™ and effective loss of some or all of such marketing exclusivity for the affected period of time. In April 2011, the Company’s representatives met with the FDA staff to discuss access to Makena™ and to provide information to the FDA relevant to its public statement.
In addition, in March 2011, CMS issued an informational bulletin to state Medicaid programs that they can choose to pay for the extemporaneously compounded hydroxyprogesterone caproate as an active pharmaceutical ingredient and this can be covered under the “medical supplies, equipment and appliances suitable for use in the home” portion of home health. Because CMS does not require states to list all of the items they cover under this section in the Medicaid state plan, states can cover hydroxyprogesterone caproate under their current state plans and do not need to submit a state plan amendment to provide for such coverage. The Company believes that this has the potential of excluding Makena™ from being provided under the various state Medicaid programs. The Company estimates that state Medicaid programs cover approximately 40% to 45% of the total number of pregnancies in the United States.
New Product Development and Approval
All applications for FDA approval must contain information relating to product formulation, raw material suppliers, stability, product testing, manufacturing processes, manufacturing facilities, packaging, labeling, quality control, and evidence of safety and effectiveness for intended uses. For a generic drug product, instead of safety and effectiveness data, an application must demonstrate that the proposed product is the same as the branded drug in several key characteristics. There are three types of applications used for obtaining FDA approval of new non-biological drug products:
| • | New Drug Application. An NDA, sometimes referred to as a “full NDA,” generally is submitted when approval is sought to market a drug with active ingredients that have not been previously approved by the FDA. Full NDAs typically are submitted for newly developed branded products and, in certain instances, an applicant submits an NDA or NDA supplement for a change to one of its previously approved products, such as a new dosage form, a new delivery system or a new indication. |
| • | 505(b)(2) New Drug Application. Another form of an NDA is the “505(b)(2) NDA,” which typically is used to seek FDA approval of products that share characteristics (often, the active ingredient(s)) with a previously approved product of another company, but contain modifications to, or differences from, the approved product that preclude submission of an abbreviated new drug application. A 505(b)(2) NDA is in order where at least some of the information required for approval does not come from studies conducted by or for the applicant or for which the applicant has obtained a right of reference. Usually, this means the application relies on the FDA’s previous approval of a similar product or reference listed drug, or published data in scientific literature that are not the applicant’s. |
| • | Abbreviated New Drug Application). An ANDA is filed when approval is sought to market a generic equivalent of a drug product previously approved under an NDA or 505(b)(2) NDA and listed in the FDA publication, “Approved Drug Products with Therapeutic Equivalence Evaluations,” also known as the “Orange Book.” Rather than directly demonstrating the product’s safety and effectiveness, as is required of an NDA, an ANDA must show that the proposed generic product is the same as the previously approved product in terms of active ingredient(s), strength, dosage form, route of administration and bioavailability. In addition, with certain exceptions, the generic product must have the same labeling as the product to which it refers. |
31
Table of Contents
Branded Product Development. The process by which a drug product, other than a generic product, is approved for marketing in the United States can take from three to more than ten years, and generally involves the following:
| • | laboratory and pre-clinical tests; |
| • | submission of an Investigational New Drug (“IND”) application, which must become effective before clinical studies may begin; |
| • | adequate and well-controlled human clinical studies to establish the safety and efficacy of the proposed product for its intended use; |
| • | submission of a full NDA or 505(b)(2) NDA containing, to the extent required, the results of the pre-clinical tests and clinical studies establishing the safety and efficacy of the proposed product for its intended use, as well as extensive data addressing matters such as manufacturing and quality assurance; |
| • | scale-up to commercial manufacturing; |
| • | satisfactory completion of an FDA pre-approval inspection of the manufacturing facilities; and |
| • | FDA approval of the application. |
To the extent that a 505(b)(2) NDA applicant can rely on the referenced application, it may not be required to conduct some of these steps.
Pre-clinical tests include laboratory evaluation of the product, its chemistry, formulation and stability, as well as toxicology and pharmacology animal studies to help define the pharmacological profile of the drug and assess the potential safety and efficacy of the product. Pre-clinical safety tests must be conducted by laboratories that comply with FDA regulations regarding Good Laboratory Practices and the U.S. Department of Agriculture’s Animal Welfare Act and its implementing regulations. Failure to conform to these legal and regulatory standards can lead to invalidation of the tests and the need to conduct them again. The results of these studies, which are submitted to the FDA as part of the IND and are reviewed by the FDA before the commencement of human clinical trials, must demonstrate that the product delivers sufficient quantities of the drug to the bloodstream or intended site of action to produce the desired therapeutic results. These studies must also provide the appropriate supportive safety information necessary for the FDA to determine whether the clinical studies proposed to be conducted under the IND can safely proceed. However, positive results of pre-clinical tests do not necessarily indicate positive results in human clinical trials.
An IND becomes effective 30 days after receipt by the FDA unless the FDA, during that 30 day period, raises concerns or questions about the conduct of the proposed trials as outlined in the IND. If there are concerns or questions, the IND sponsor and the FDA must resolve them before clinical trials may begin. The FDA may authorize trials only on specified terms and may suspend clinical trials at any time on various grounds, including a finding that patients are being exposed to unacceptable health risks. If the FDA places a study on clinical hold, the sponsor must resolve all of the FDA’s concerns before the study may proceed. The IND application process may be extremely costly and can substantially delay development of products. Similar restrictive requirements also apply in other countries.
Clinical trials involve the administration of the investigational product to humans under the supervision of qualified principal investigators. Clinical trials must be conducted in accordance with Good Clinical Practices under protocols submitted to the FDA as part of an IND. In addition, each clinical trial is approved and conducted under the auspices of an independent institutional review board (“IRB”) and with each subject’s informed consent. The IRB at each institution at which a clinical trial is being performed may suspend a clinical trial at any time for a variety of reasons, including a belief that the test subjects are being exposed to an unacceptable health risk. The sponsor may also suspend or terminate a clinical trial at any time.
32
Table of Contents
Human clinical studies are typically conducted in three sequential phases, which may overlap:
| • | Phase I: The drug is initially introduced into a relatively small number of healthy human subjects or patients and is tested for safety, dosage tolerance, mechanism of action, absorption, metabolism, distribution and excretion. |
| • | Phase II: Studies are performed with a limited patient population to identify possible adverse effects and safety risks, to assess the efficacy of the product for specific targeted diseases or conditions, and to determine dosage tolerance and optimal dosage. |
| • | Phase III: When Phase II evaluations demonstrate that a dosage range of the product is effective and has an acceptable safety profile, Phase III trials are undertaken to evaluate further dosage and clinical efficacy and to test further for safety in an expanded patient population at geographically dispersed clinical study sites. |
Success in early-stage clinical trials does not necessarily assure success in later-stage clinical trials. Data obtained from clinical activities are not always conclusive and may be subject to alternative interpretations that could delay, limit or even prevent regulatory approval. New regulations now require the posting of certain details about active clinical trials on government or independent websites (e.g., http://clinicaltrials.gov), and subsequently a limited posting of the results of those trials. This helps prospective patients find out about trials they may wish to enroll in, but also provides some competitive intelligence to other companies working in the field. Failure to post the trial or its results in a timely manner can result in civil penalties and the rejection of the drug application.
The results of the product development, including pre-clinical studies, clinical studies, and product formulation and manufacturing information, are then submitted to the FDA as part of the NDA. The FDA also may conclude that as part of the NDA or the 505(b)(2) NDA, the sponsor must develop a risk evaluation and mitigation strategy (“REMS”) to ensure that the benefits of the drug outweigh the risks. A REMS may have different components, including a package insert directed to patients, a plan for communication with healthcare providers, restrictions on a drug’s distribution, or a medication guide to provide better information to consumers about the drug’s risks and benefits.
The FDA reviews each submitted application before accepting it for filing, and may refuse to file the application if it does not appear to meet the minimal standards for filing. If the FDA refuses to file an application and requests additional information, the application must be resubmitted with the requested information. Once the submission is accepted for filing, the FDA begins an in-depth review of the application to determine, among other things, whether a product is safe and effective for its intended use. As part of this review, the FDA may refer the application to an appropriate advisory committee, typically a panel of clinicians, for review, evaluation and a recommendation.
Acceptance for filing of an application does not assure FDA approval for marketing. The FDA has substantial discretion in the approval process and may disagree with an applicant’s interpretation of the submitted data, which could delay, limit, or prevent regulatory approval. If it concludes that the application does not satisfy the regulatory criteria for approval, the FDA typically issues a Complete Response letter communicating the agency’s decision not to approve the application and outlining the deficiencies in the submission. The Complete Response letter may request additional information, including additional pre-clinical testing or clinical trials. Even if such information and data are submitted, the FDA may ultimately decide that the NDA or 505(b)(2) NDA does not satisfy the criteria for approval.
If the FDA approves the application, the agency may require Phase IV studies as a condition to approval. These studies may involve continued testing of a product and development of data, including clinical data, about the product’s effects in various populations and any side effects associated with long-term use. After approval, the FDA also may require post-marketing studies or clinical trials if new safety information develops. Failure to conduct these studies in a timely manner may result in substantial civil fines.
33
Table of Contents
Generic/Non-Branded Product Development. FDA approval of an ANDA is required before marketing in the United States a generic equivalent of a previously approved drug product, which is known as the “reference listed drug.” Generally, an ANDA must demonstrate that the proposed generic product has the same active ingredient(s) and in the same strength as the reference listed drug, and is in the same dosage form and delivered by the same route of administration. The ANDA also must show that the two products are bioequivalent, which means that, within defined parameters, their active ingredients are absorbed at the same rate and to the same extent. For drugs that are systemically absorbed (as opposed to locally acting), bioequivalence is usually demonstrated by means of bioavailability studies of the active ingredient in the bloodstream, conducted in a relatively small number of human subjects. Further, with certain exceptions, a proposed generic product must have the same labeling as the reference listed drug. Generally, a generic product that meets these criteria and is approved is considered therapeutically equivalent to the reference listed drug, and is given an “A” rating in the FDA’s Orange Book, which communicates FDA’s expectation that the two products can be expected to have the same safety and efficacy profile. This, in turn, often is the basis for substitution of the generic product for the reference listed drug, under state law. There are circumstances under which an ANDA may be submitted for a generic product that differs from the reference listed drug in certain characteristics, such as strength or dosage form. Generic products approved with such differences do not qualify for therapeutic equivalence and generally are not substitutable under state law.
Because it typically does not require pre-clinical and clinical studies (relying instead on the studies establishing the safety and efficacy of the reference listed drug), the ANDA development process is generally less time consuming and complex than the NDA development process. Nonetheless, it does require submission of significant technical information about product formulation and manufacturing, as well as compliance with cGMP. In addition, submission or approval of an ANDA can be subject to delays due to patent rights or regulatory marketing exclusivities associated with the reference listed product. Upon approval, the first-filed ANDA that challenges the patents associated with a reference listed drug may be eligible for a 180-day period of exclusivity during which no other ANDA with the same reference listed drug may be approved. (See “—The Drug Price Competition and Patent Term Restoration Act of 1984” below for a discussion of the patent and exclusivity provisions affecting ANDAs.)
The development process for generic products, although generally much shorter than that of products approved under a NDA or a 505(b)(2) NDA, can still be a matter of years. Because there is no legislation-mandated timeline for review of an ANDA by FDA as exists for an NDA, these applications generally take longer than an original NDA to receive an FDA approval letter. In addition, certain changes to a product approved under an ANDA require submission of an ANDA supplement, and the time it takes to obtain, analyze and present the necessary information or data and have the FDA review the supplement can be significant.
Current Good Manufacturing Practices. One requirement for FDA approval of NDAs, 505(b)(2) NDAs and ANDAs is that our manufacturing facilities, procedures, and operations conform to FDA requirements, generally referred to as cGMP. The requirements for FDA approval encompass all aspects of the production process, including validation and recordkeeping, and involve changing and evolving standards.
The Drug Price Competition and Patent Term Restoration Act of 1984. The Drug Price Competition and Patent Term Restoration Act of 1984, known as the Hatch-Waxman Act, established an abbreviated process for obtaining FDA approval for generic versions of approved branded drug products. In addition to establishing a shorter, less expensive pathway for approval of generic drugs, the Hatch-Waxman Act provides incentives for the development of new branded products and innovations to approved products by means of marketing exclusivities and extension of patent rights.
Five-Year and Three-Year Marketing Exclusivity. The Hatch-Waxman Act provides two distinct marketing exclusivity provisions that may either preclude the submission, or delay the approval, of an ANDA or 505(b)(2) NDA referencing the drug. A five-year marketing exclusivity period is provided to the first applicant to gain approval of an NDA or 505(b)(2) NDA for a product that contains an active ingredient not found in any other
34
Table of Contents
approved product (i.e., a “new chemical entity” or “NCE”). The FDA is prohibited from accepting for filing any ANDA or 505(b)(2) NDA for a product that relies on the approved NCE as the reference listed drug for five years from the date of approval of the NCE, or four years in the case of an ANDA or 505(b)(2) NDA that challenges a patent associated with the reference listed drug. (See discussion below regarding patent challenges.) The five-year exclusivity protects the entire new chemical entity franchise, including all products containing the active ingredient for any use and in any strength or dosage form, from an ANDA or 505(b)(2) NDA, but does not preclude filing, review or approval of a full NDA for a product with the same active ingredient.
The Hatch-Waxman Act also provides three years of marketing exclusivity for applications for products that are not new chemical entities, if the application contains the results of new clinical investigations (other than bioavailability studies) that were essential to approval of the application. Examples of such applications include applications for new indications, dosage forms (including new drug delivery systems), strengths, or conditions of use for an already approved product. This three-year marketing exclusivity period protects against the approval of ANDAs and 505(b)(2) NDAs for the innovation that required clinical data; it does not prohibit the FDA from accepting or approving ANDAs or 505(b)(2) applications for other products containing the same active ingredient. The five- and three-year marketing exclusivity periods apply equally to patented and non-patented drug products.
Orphan Drug Exclusivity. The Orphan Drug Act was enacted by Congress to provide financial incentives for the development of drugs for rare conditions (affecting less than 200,000 individuals per year) in the United States. The orphan designation is granted for a combination of a drug entity and an indication and therefore it can be granted for an existing drug with a new (orphan) indication. Applications are made to the Office of Orphan Products Development at the FDA and a decision or request for more information is rendered in 60 days. New Drug Applications designated as orphan drugs are exempt from user fees, obtain additional clinical protocol assistance, are eligible for tax credits up to 50% of R&D costs, and are granted a seven-year period of exclusivity upon approval. The FDA cannot approve the same drug for the same condition (ANDA or NDA) during this period of exclusivity, except in certain circumstances where a new product demonstrates superiority to the original treatment.
However, in March 2011, the FDA issued a public statement that, in order to ensure continued access for patients needing hydroxyprogesterone caproate, the FDA intended to refrain at this time from taking enforcement action with respect to compounding pharmacies producing compounded 17-alpha hydroxyprogesterone caproate in response to individual prescriptions for individual patients. In addition, in March 2011, CMS issued an informational bulletin to state Medicaid programs that they can choose to pay for the extemporaneously compounded hydroxyprogesterone caproate as an active pharmaceutical ingredient and this can be covered under the “medical supplies, equipment and appliances suitable for use in the home” portion of home health. Because CMS does not require states to list all of the items they cover under this section in the Medicaid state plan, states can cover hydroxyprogesterone caproate under their current state plans and do not need to submit a state plan amendment to provide for such coverage. The Company believes that this has the potential of excluding Makena™ from being provided under the various state Medicaid programs. The Company estimates that state Medicaid programs cover approximately 40% to 45% of the total number of pregnancies in the United States. The impact of the FDA’s and CMS’ statements on the effectiveness of the Company’s orphan drug marketing exclusivity is at present still unclear but a failure by the FDA to take enforcement action against compounding pharmacies, and the permitting of Medicaid reimbursement by CMS for compounded hydroxyprogesterone caproate, may result in substantial sales of compounded alternatives to Makena™ and effective loss of some or all of the Company’s orphan drug marketing exclusivity for the affected period of time.
Paragraph IV Certifications. Under the Hatch-Waxman Act, NDA applicants and NDA holders must provide certain information about patents related to the branded drug for listing in the Orange Book. When an ANDA or 505(b)(2) application is submitted, it must contain one of several possible certifications regarding each of the patents listed in the Orange Book for the branded product that is the reference listed drug. A certification that a listed patent is invalid or will not be infringed by the sale of the proposed product is called a Paragraph IV
35
Table of Contents
certification. Within 30 days of the filing of such an ANDA or 505(b)(2) NDA, the applicant must notify the NDA holder and patent owner that the application has been submitted, and provide the factual and legal basis for the applicant’s opinion that the patent is invalid or not infringed.
Once a patent-challenging ANDA or 505(b)(2) NDA has been filed, the NDA holder or patent holder may challenge the Paragraph IV certification by filing suit for patent infringement. If this is done within 45 days of receiving notice of the Paragraph IV certification, a one-time 30-month stay of FDA’s ability to approve the ANDA or 505(b)(2) application is triggered. The FDA may approve the proposed product before the expiration of the 30-month stay only if a court finds the patent invalid or not infringed, or if the court shortens the period because the parties have failed to cooperate in expediting the litigation.
Although restrictions under the consent decree currently preclude us from testing any new generic drugs under development that are made in our facilities, prior to entering into the consent decree, we had submitted ANDAs with the FDA seeking permission to market generic versions of several branded products, including varying strengths of the following:
| • | Metadate CD® (methylphenidate hydrochloride) Extended-Release Capsules |
| • | Ritalin LA® (methylphenidate hydrochloride) Extended-Release Capsules |
| • | Focalin XR® (dexmethylphenidate hydrochloride) Extended-Release Capsules |
| • | Keppra XR® (levetiracetam) Extended-Release Tablets |
With respect to the first three ANDA submissions noted above, we have sought favorable court rulings in patent infringement actions filed against us by the sponsors of the branded products. On March 1, 2010, we entered into a Settlement Agreement to settle the patent infringement actions with respect to Ritalin LA® and Focalin XR®. See Note 15—“Commitments and Contingencies—Litigation and Governmental Inquiries” of the Notes to Consolidated Financial Statements included in this Report for more information regarding the settlement. On April 2, 2010, we entered into a Settlement and License Agreement to settle the patent infringement action with respect to Metadate CD®. See Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report for more information regarding the settlement.
Paragraph IV opportunities are no longer available to the pending ANDAs due to not obtaining approval or tentative approval of the ANDAs as a result of the consent decree.
180-Day Exclusivity. Under certain circumstances, the Hatch-Waxman Act rewards the first applicant that submits an ANDA containing a Paragraph IV certification with 180 days of marketing exclusivity, during which no later-filed Paragraph IV ANDA for a generic version of the same product can enter the market. The exclusivity does not block approval of products submitted in a 505(b)(2) NDA. The 180-day exclusivity is applied on a “product-by-product” basis, which means that the first applicant (or applicants, if submitting on the same day) to submit an ANDA with a Paragraph IV certification to any listed patent is eligible to receive the single period of exclusivity. Any later applicant that is the first to submit an ANDA containing a Paragraph IV certification to a different patent does not receive exclusivity. The 180-day exclusivity period is triggered only on the first commercial marketing of the first applicant’s (or first applicants’) generic product, but eligibility for exclusivity will be forfeited in certain circumstances, including if the applicant fails to come to market in a timely manner, withdraws the ANDA or all of its Paragraph IV certifications, enters into an anti-competitive agreement with the NDA holder or patent owners, or if all of the patents that earned the applicant the exclusivity expire. On forfeiture, any later-submitted ANDA may be approved once review of the application is complete.
Patent Term Restoration. Under the Hatch-Waxman Act, a portion of the patent term lost during product development and FDA review of an NDA or 505(b)(2) NDA is restored if approval of the application is the first permitted commercial marketing of a drug containing the active ingredient. The patent term restoration period is
36
Table of Contents
generally one-half the time between the effective date of the IND and the date of submission of the NDA, plus the time between the date of submission of the NDA and the date of FDA approval of the product. The maximum period of restoration is five years, and the patent cannot be extended to more than 14 years from the date of FDA approval of the product. Only one patent claiming each approved product is eligible for restoration and the patent holder must apply for restoration within 60 days of approval. The U.S. Patent and Trademark Office (“USPTO”), in consultation with the FDA, reviews and approves the application for patent term restoration.
Drug Products Marketed Without FDA Approval. Drugs manufactured according to the Over-the-Counter (“OTC”) monographs in the Code of Federal Regulations can be lawfully marketed in the US without prior approval. Under the FDCA, FDA regulations, and statements of FDA policy, certain drug products are permitted to be marketed in the U.S. without prior approval. Within the past few years, the FDA has revised its enforcement policies, significantly limiting the circumstances under which these unapproved products may be marketed. If the FDA determines that a company is distributing an unapproved product that requires approval, the FDA may take enforcement action in a variety of ways, including product seizures and seeking a judicial injunction against distribution. Moreover, the FDA has said that, if it takes action against a company for an unrelated violation, it may expand its enforcement to other products manufactured or distributed by the company. Previously, in addition to drugs that have been subject to FDA review and approval, we manufactured and distributed a number of drug products without FDA approval. As part of the consent decree, we have agreed not to manufacture or distribute any drugs that require FDA approval unless we have obtained such FDA approval; when we resume production under the terms of the consent decree, any drug product requiring approval that we manufacture and commercially distribute will be the subject of an approved NDA, 505(b)(2) NDA, or ANDA.
Post-Approval/Marketing Obligations. Products manufactured or distributed pursuant to FDA approvals are subject to continuing regulation by the FDA, which include company obligations to manufacture products in accordance with cGMP, maintain and provide to the FDA updated safety and efficacy information, report adverse experiences with the product, keep certain records and periodic reports, obtain FDA approval of certain manufacturing or labeling changes, and comply with FDA promotion and advertising requirements and restrictions. Failure to meet these obligations can result in various adverse consequences, both voluntary and FDA-imposed, including product recalls, withdrawal of approval, restrictions on marketing, and the imposition of civil fines and criminal penalties against the NDA holder. In addition, later discovery of previously unknown safety or efficacy issues may result in restrictions on the product, manufacturer or NDA holder.
New government requirements may be established that could delay or prevent regulatory approval of our products under development, or affect the conditions under which approved products are marketed. For instance, the Food and Drug Administration Amendments Act of 2007 provides the FDA with expanded authority over drug products after approval. This legislation enhanced the FDA’s authority with respect to post-marketing safety surveillance, including, among other things, the authority to require additional post-marketing studies or clinical trials, labeling changes as a result of safety findings, registration of clinical trials, and making clinical trial results publicly available.
Among other things, drug manufacturers and their subcontractors must register their establishments with the FDA and certain state agencies and list their products with the FDA. The FDA periodically inspects manufacturing facilities in the United States and abroad to assure compliance with the applicable cGMP regulations and other requirements. Facilities also are subject to inspections by other federal, foreign, state or local agencies. In complying with the cGMP regulations, manufacturers must continue to expend time, money and effort in recordkeeping and quality control to assure that the product meets applicable specifications and other post-marketing requirements. At the conclusion of an FDA inspection, the FDA investigator may issue a list of inspectional observations, documented on a Form 483 Report. Typically, a company has an opportunity to respond to the Form 483 Report observations and work with the FDA to address the agency’s concerns. Failure to comply with FDA’s cGMP regulations or other requirements can lead to civil, criminal, or administrative sanctions or remedies, including the suspension of manufacturing operations. On February 2, 2009, at the conclusion of an FDA inspection, we were issued a Form 483 that identified a number of instances of alleged
37
Table of Contents
cGMP noncompliance. As discussed elsewhere, we voluntarily suspended manufacturing and distribution of our products, and on March 2, 2009, we entered into a consent decree with the FDA regarding our drug manufacturing and distribution. See Part I, Item 1—“Business—(b) Significant Developments—Discontinuation of Manufacturing and Distribution; Product Recalls; and the FDA Consent Decree” in this Report for more information. Post-approval marketing and promotion of drugs is an area of particular FDA scrutiny and enforcement. Among the issues addressed by FDA regulations are print and broadcast advertising for prescription drugs, promotional activities involving the Internet, dissemination of materials regarding off-label uses of approved products, and sponsorship of scientific and educational activities. Failure to comply with FDA requirements can have negative consequences, including adverse publicity, enforcement letters from the FDA, mandated corrective advertising or communications with doctors, and civil or criminal penalties. Although physicians may prescribe legally available drugs for uses that are not described in the drug’s labeling (known as “off-label use”), manufacturers may not promote such off-label uses. Manufacturers may, however, engage with medical professionals in scientific or medical dialogue that involves information related to off-label uses, so long as it is done in a non-promotional manner.
Prescription Drug Marketing Act. The Prescription Drug Marketing Act (“PDMA”), which amended the FDCA, imposes detailed requirements on the distribution of prescription drug samples and requires state licensing of wholesale distributors of prescription drugs under federal guidelines that include minimum standards for storage, handling and record keeping. Many states also require registration of out-of-state drug manufacturers and distributors who sell products in their states, and may also impose additional requirements or restrictions on out-of-state firms. These requirements vary widely from state-to-state and are subject to change with little or no direct notice to potentially affected firms. Failure to comply with the PDMA or applicable state requirements can lead to sanctions, including substantial civil and criminal penalties and potential restrictions on sales or other activities within particular states.
International Approvals. For international markets, a pharmaceutical company is subject to regulatory requirements, inspections and product approvals substantially identical to those in the United States. In connection with any future marketing, distribution and license agreements that we may enter into, our licensees or we may accept or assume responsibility for such foreign regulatory approvals. The time and cost required to obtain these international market approvals may be different from those required for FDA approval.
U.S. Drug Enforcement Administration. The DEA regulates certain drug products containing controlled substances, such as opioids, and List I chemicals, such as pseudoephedrine, pursuant to the CSA. The CSA and DEA regulations impose specific requirements on manufacturers and other entities that handle these substances including with respect to registration, recordkeeping, reporting, storage, security and distribution. Recordkeeping requirements include accounting for the amount of product received, manufactured, stored and distributed. Companies handling either controlled substances or List I chemicals are also required to maintain adequate security and to report suspicious orders, thefts and significant losses. The DEA periodically inspects facilities for compliance with the CSA and its regulations. Failure to comply with current and future regulations of the DEA could lead to a variety of sanctions, including revocation or denial of renewal of DEA registrations, injunctions, or civil or criminal penalties. In June 2010, we entered into a Memorandum of Agreement (“MoA”) with the DEA agreeing to certain terms and restrictions related to controlled substances in lieu of the DEA initiating Order to Show Cause proceedings to revoke all of our DEA registrations and to deny any pending applications for registration. The MoA was entered into as a result of a routine DEA inspection in September 2008 during which the DEA concluded that we had systematically failed to provide and maintain effective controls and procedures to guard against the theft and diversion of pseudoephedrine and thereby placed the public health and safety at risk. On October 26, 2010 we completed the disposal of this raw material as witnessed by a DEA investigator, which marked the completion of the condition of our MoA with the DEA.
We utilize controlled substances in certain of our current products and products in development and therefore must meet the requirements of the CSA and the related regulations administered by the DEA. These regulations relate to the manufacture, shipment, storage, sale and use of controlled substances. The DEA limits
38
Table of Contents
the availability of the active ingredients used in certain of our current products and products in development and, as a result, our procurement quota of these active ingredients may not be sufficient to meet commercial demand or complete clinical trials. We must annually apply to the DEA for procurement quota in order to obtain these substances. Any delay or refusal by the DEA in establishing our procurement quota for controlled substances could delay or stop our clinical trials or product launches, or could cause trade inventory disruptions for those products that have already been launched, which could have a material adverse effect on our business, financial condition, results of operations or cash flows.
We are subject to the requirements of the CSA and DEA regulations in the handling of any controlled substances in Schedules II-V or any of the List I chemicals identified in the CSA. Specifically, we are subject to regulation in the current manufacture and distribution of products containing pseudoephedrine, a List I chemical. As a result of a series of amendments to the CSA, the DEA has imposed increased restrictions on the manufacture and distribution of pseudoephedrine products. For example, the Comprehensive Methamphetamine Control Act of 1996 was enacted to authorize the DEA to monitor transactions involving chemicals that may be used illegally in the production of methamphetamine. The Comprehensive Methamphetamine Control Act of 1996 establishes certain registration and recordkeeping requirements for manufacturers of OTC cold, allergy, asthma and diet medicines that contain ephedrine, pseudoephedrine or phenylpropanolamine (“PPA”).
More recently, the Reauthorization Act of 2005 was signed into law on March 9, 2006. The Reauthorization Act of 2005 included the Combat Methamphetamine Epidemic Act. This law further amended the CSA and provided additional requirements for the sale of pseudoephedrine products. Among the various provisions, this national legislation places certain restrictions on the purchase and sale of all products that contain ephedrine, pseudoephedrine or PPA (List I chemical products). The CSA also imposed import and production quotas for List I chemicals, including pseudoephedrine.
The CSA, as amended, also imposed daily restrictions on the amount of List I chemical products a retailer may sell to a consumer (3.6 grams per day) and limitations on the amount of List I chemical products a consumer may purchase (9.0 grams) over a 30-day period. Further, effective September 30, 2006, the CSA requires that (a) retail sellers maintain a logbook that tracks the sales of List I chemical products to individuals, and (b) purchasers provide valid identification in order to purchase List I chemical products. Many states have also enacted legislation regulating the manufacture and distribution of pseudoephedrine products. We are subject to these state requirements as well.
Medicaid and Other Drug Reimbursement Programs. Government reimbursement programs include Medicare, Medicaid, the 340B drug pricing program, Tricare and State Pharmacy Assistance Programs established according to statute, government regulations and policy. The CMS administers the Medicaid drug rebate agreements between the federal government and pharmaceutical manufacturers. Federal law requires that a pharmaceutical manufacturer, as a condition of having its products receive federal reimbursement under Medicaid and Medicare Part B, must pay rebates to state Medicaid programs for all units of its pharmaceuticals that are dispensed to Medicaid beneficiaries and paid for by a state Medicaid program under a fee-for-service arrangement. (In addition, some states require supplemental rebates as a condition of including the manufacturer’s drug on the state Medicaid program’s Preferred Drug List.) Pursuant to recently enacted health reform legislation, rebates will be due on both Medicaid fee-for-service utilization as well as utilization of Medicaid managed care organizations; however, the timing of the implementation of this change remains unclear because it requires revisions to agreements between state Medicaid programs and such organizations.
This federal requirement is effectuated through the Medicaid rebate agreement, which is between each manufacturer and the Secretary of Health and Human Services. The agreement provides that the drug manufacturer will remit a rebate to each state Medicaid agency, on a quarterly basis. Pursuant to recently enacted health reform legislation, effective for rebate periods after December 31, 2009, the rebates are as follows: for non-innovator products, in general generic drugs marketed under ANDAs, the rebate amount is 13% of the Average Manufacturer Price (“AMP”) for the quarter; for innovator products, in general brand-name products
39
Table of Contents
marketed under NDAs, the rebate amount is the greater of 23.1% of the AMP for the quarter or the difference between such AMP and the best price for that same quarter. The 23.1% is lowered to 17.1% for certain clotting factor and pediatric drug products. An additional rebate for innovator products is payable in the amount by which, if any, the product’s AMP has increased at a rate faster than inflation since the product’s launch. Recently enacted health reform legislation changes the additional rebate formula for certain products that qualify as new formulations of existing drugs, effective for drugs paid for by a state as of the first quarter of calendar year 2010, so that the additional rebate for such products can be increased and based on the additional rebate for the original drug. It also caps the total rebate amount for innovator drugs at 100% of the AMP.
We have a Medicaid rebate agreement in effect with the federal government. Federal and/or state governments have and are expected to continue to enact measures aimed at reducing the cost of drugs to such governmental payors as well as the public, including the enactment in December 2003 of Medicare legislation that expanded the scope of Medicare coverage to include outpatient drugs (Part D), starting in January 2006, the Deficit Reduction Act of 2005 (“DRA”), as well as recently enacted health reform legislation. We cannot predict the nature of such measures or their impact on our profitability. Various states have in recent years also adopted Medicaid supplemental drug rebate programs that are intended to provide the individual states with additional manufacturer rebates on Medicaid utilization over and above those required under a manufacturer’s federal Medicaid agreement. States also have created drug coverage and corresponding manufacturer rebate programs for non-Medicaid populations, known as state pharmaceutical assistance programs. These rebate programs are generally designed to mimic the federal drug rebate program in terms of how the manufacturer rebates are calculated. Although there are a number of Medicaid supplemental and state pharmacy assistance rebate programs in which we participate, they are insignificant in the aggregate compared to our quarterly Medicaid drug rebate obligations.
The DRA amended the Medicaid statute in a number of ways, including to revise the methodology for the calculation of federal upper limits, a type of cap on the amounts a state Medicaid program can reimburse pharmacies for multiple source drugs (drugs where there are at least two equivalent versions on the market, for example, a branded and a generic version, or two or more generic versions) dispensed to Medicaid patients, and AMP, as well as to require the public availability of AMP data.
In July 2007, CMS issued a final rule regarding the calculation of AMP as well as the other statutory amendments made by the DRA. This rule, as required by the DRA amendments, requires CMS to use AMP to calculate federal upper limits. Prior to the enactment of this legal requirement, CMS typically used Average Wholesaler Price (“AWP”) or Wholesaler Acquisition Cost (“WAC”) in the calculation of federal upper limits. The rule also rejected requests to postpone the public availability of AMP data. In mid-December 2007, a preliminary injunction was granted, resulting in postponement of the actual implementation of these aspects of the DRA and the rule such that AMP currently cannot be used to calculate federal upper limits and also cannot be disclosed to the public. The relevant court case is still pending and the injunction remains in place, resulting in a continual postponement of the implementation of these requirements. We do not know how the new methodology for calculating federal upper limits, if implemented, will affect our pharmacy customers or to what extent these customers will seek to pass on any decrease in Medicaid reimbursements to us. We cannot predict how the sharing of manufacturer-specific data may impact competition in the marketplace. Recently enacted health reform legislation also will affect these issues. Effective fourth quarter of calendar year 2010, the federal upper limit will be calculated using a weighted average AMP, based only on those multiple source drugs that are nationally available for purchase by retail community pharmacies and where there are three or more equivalent versions of the product on the market. The definition of AMP also will change, which may affect the Medicaid rebate amount, as described above. In addition, this legislation will change the publicly available AMP data to include only weighted average monthly AMPs as well as average retail survey prices determined by the Medicaid program.
A manufacturer must also participate in the 340B drug pricing program in order for federal funds to be available to pay for the manufacturer’s drugs under Medicaid and Medicare Part B. Under this program, the
40
Table of Contents
participating manufacturer agrees to charge statutorily-defined covered entities no more than the 340B discounted price for the manufacturer’s covered outpatient drugs. The formula for determining the discounted purchase price is defined by statute and is based on the AMP and rebate amount for a particular product as calculated under the Medicaid drug rebate program, discussed above. To the extent that recently enacted health reform legislation, as discussed above, causes the statutory and regulatory definitions of AMP and the Medicaid rebate amount to change, these changes also will impact the discounted purchase prices that a manufacturer is obligated to provide under this program. This same health care reform legislation also expands the 340B drug pricing program to include new entity types and obligates manufacturers to sell to covered entities if they sell to any other purchaser, effective for drugs purchased on or after January 1, 2010.
The recently enacted health care reform legislation currently is the subject of further proposed amendments in Congress, which if passed, could further impact these programs and us.
There has been enhanced political attention, governmental scrutiny and litigation at the federal and state levels of the prices paid or reimbursed for pharmaceutical products under Medicaid and other government programs. See “Risk Factors—Investigations of the calculation of average wholesale prices may adversely affect our business” and Note 15—“Commitments and Contingencies—Litigation and Governmental Inquiries” of the Notes to Consolidated Financial Statements included in this Report. In March 2011, CMS issued an informational bulletin to state Medicaid programs that they can choose to pay for the extemporaneously compounded hydroxyprogesterone caproate as an active pharmaceutical ingredient and this can be covered under the “medical supplies, equipment and appliances suitable for use in the home” portion of home health. Because CMS does not require states to list all of the items they cover under this section in the Medicaid state plan, states can cover hydroxyprogesterone caproate under their current state plans and do not need to submit a state plan amendment to provide for such coverage.
Federal Contracting/Pricing Requirements. We also make our products available for purchase by authorized users of the Federal Supply Schedule (“FSS”) of the General Services Administration pursuant to our FSS contract with the Department of Veterans Affairs. Under the Veterans Health Care Act of 1992 (the “VHCA”), we are required to offer deeply discounted FSS contract pricing to four federal agencies—the Department of Veterans Affairs, the Department of Defense (“DoD”), the Coast Guard and the Public Health Service (including the Indian Health Service)—for federal funding to be made available to pay for our products under the Medicaid program and Medicare Part B, and for our products to be eligible to be purchased by those four federal agencies and certain federal grantees. FSS pricing to those four federal agencies must be equal to or less than the federal ceiling price (“FCP”), which is, at a minimum, 24% off the Non-Federal Average Manufacturer Price, or “Non-FAMP,” for the prior fiscal year. In addition, if we are found to have knowingly submitted false information to the government, the VHCA provides for civil monetary penalties not to exceed $100,000 per false item of information in addition to other penalties available to the government.
Tricare Retail Program. On March 17, 2009, the DoD Tricare Management Activity (“TMA”) issued a Final Rule pursuant to Section 703 of the National Defense Authorization Act of 2008 (“NDAA”) to establish a program under which it will seek Federal Ceiling Price-based rebates from drug manufacturers on Tricare retail utilization. Under the Final Rule, which became effective May 26, 2009, DoD asserts an entitlement to rebates on Tricare Retail Pharmacy utilization from January 28, 2008 forward, unless TMA grants a waiver or compromise of amounts due from quarters that have passed prior to execution of a voluntary agreement with DoD. Ther-Rx submitted a request for waiver for the entirety of its rebate liability based on utilization occurring on or after January 28, 2008, and before July 15, 2009, the effective date of its Tricare voluntary rebate agreement with DoD. The total amount for which a waiver was requested is approximately $500,000. ETHEX did not request a waiver, instead opting to pay all rebate liability since the date of enactment of the NDAA. ETHEX also entered into a Tricare voluntary rebate agreement with DoD, which became effective July 13, 2009.
Pursuant to the terms of the Final Rule, during the pendency of Ther-Rx’s waiver request, we are not required to pay rebates for amounts subject to the request and DoD considers our Company in compliance with
41
Table of Contents
the requirements of the Final Rule with respect to the requirement to pay such amounts. In addition, in the event that DoD does not grant our request in full, we have reserved the right to challenge DoD’s assertion that it is owed rebates based on Tricare retail utilization from the date of enactment of the NDAA and prior to the execution of a voluntary agreement.
The Final Rule provides that, absent an agreement from the manufacturer to provide Tricare retail rebates, DoD will designate the manufacturer’s product(s) as Tier 3 (non-formulary) and require that beneficiaries obtain prior authorization in order for the product(s) to be dispensed at a Tricare retail pharmacy. Moreover, DoD has asserted in the Final Rule the right to apply offsets and/or proceeds under the Debt Collection Act, in the event that a company does not pay rebates. DoD required rebate agreements to be submitted by manufacturers on or before June 1, 2009. We submitted proposed voluntary pricing for both Ther-Rx and ETHEX in a timely manner.
Per the Final Rule, rebates are computed by subtracting the applicable FCP from the corresponding Annual Non-FAMP. We believe the estimated liability for Tricare retail utilization from January 28, 2008 through July 14, 2009 is approximately $500,000. It is possible that, pursuant to the waiver request submitted by us, in accordance with the provisions of the Final Rule, DoD will agree to accept a lesser sum or waive payment of the entire amount for these pre-agreement periods.
Anti-Kickback and False Claims Statutes. In addition to the FDA restrictions on marketing of pharmaceutical products, several other types of state and federal laws have been applied to restrict certain marketing practices in the pharmaceutical and medical device industries in recent years. These laws include anti-kickback statutes and false claims statutes.
The federal health care program anti-kickback statute prohibits, among other things, knowingly and willfully offering, paying, soliciting, or receiving remuneration to induce or in return for purchasing, leasing, ordering, or arranging for the purchase, lease, or order of any health care item or service reimbursable under Medicare, Medicaid, or other federally financed healthcare programs. This statute has been interpreted to apply to arrangements between pharmaceutical manufacturers on the one hand and prescribers, purchasers, and formulary managers on the other. Although there are a number of statutory exemptions and regulatory safe harbors protecting certain common activities from prosecution, the exemptions and safe harbors are drawn narrowly, and practices that involve remuneration to individuals or entities in a position to prescribe, purchase, or recommend our products may be subject to scrutiny if they do not qualify for an exemption or safe harbor.
Federal false claims laws prohibit any person from knowingly presenting, or causing to be presented, a false claim for payment to the federal government, or knowingly making, or causing to be made, a false statement to get a false claim paid or to reduce an amount owed to the federal government. Recently, several pharmaceutical and other health care companies have been prosecuted under these laws for allegedly providing free product to customers with the expectation that the customers would bill federal programs for the product or for allegedly misreporting the pricing data on which Medicaid rebates are calculated so as to reduce the amount of rebates owed to state Medicaid programs. Other companies have been prosecuted for causing false claims to be submitted because of the company’s marketing of the product for unapproved, and thus non-reimbursable, uses. We understand that our dissolved subsidiary ETHEX has been named as a defendant in a qui tam litigation filed under seal based on such claims with respect to certain unapproved drug products sold by ETHEX and we have been responding to subpoenas and inquiries by HHS OIG and the U.S. Department of Justice’s U.S. Attorney’s Office in Boston in connection with such claims (see Note 13—“Long-Term Debt” of the Notes to Consolidated Financial Statements included in this Report). The majority of states also have statutes or regulations similar to the federal anti-kickback law and false claims laws, which apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply regardless of the payor. Sanctions under these federal and state laws may include civil monetary penalties, exclusion of a manufacturer’s products from reimbursement under government programs, debarment from contracting with the government, criminal fines, and imprisonment.
42
Table of Contents
Environment
We do not expect that compliance with federal, state or local provisions regulating the discharge of materials into the environment or otherwise relating to the protection of the environment will have a material effect on our capital expenditures, earnings or competitive position.
Employees
We have realigned and restructured our sales force and our production workforce. As a result of such measures, we reduced our employee headcount from approximately 317 on February 28, 2010 to approximately 237 on March 31, 2010 and increased to approximately 271 on March 31, 2011, which includes 97 sales representatives through a contract sales organization.
Available Information
We make available, free of charge through our Internet website (http://www.kvpharmaceutical.com), our Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K and amendments to these reports filed or furnished pursuant to Section 13(a) or 15(d) of the Securities Exchange Act of 1934, as amended, as soon as reasonably practicable after we electronically file these reports with, or furnish them to, the SEC. Also, copies of our Corporate Governance Guidelines, Audit Committee Charter, Compensation Committee Charter, Nominating and Corporate Governance Committee Charter, Code of Ethics for Senior Executives and Standards of Business Ethics for all directors and employees are available on our website, and available in print to any shareholder who requests them. The information posted on our website is not incorporated into this Report .
In addition, the SEC maintains an Internet website (http://www.sec.gov) that contains reports, proxy and information statements, and other information regarding registrants that file electronically with the SEC.
| Item 1A. | RISK FACTORS |
Investing in our Class A and B Common Stock and debt securities involves substantial risk. We operate in a rapidly changing environment that involves a number of risks, some of which are beyond our control. The following risk factors could have a material adverse effect on our business, financial position, results of operations, cash flows or viability. These risk factors may not include all of the important risks that could affect our business or our industry, that could cause our future financial results to differ materially from historic or expected results, or that could cause the market price of our common stock to fluctuate or decline. Because of these and other factors, past financial performance should not be considered an indication of future performance.
There is substantial doubt about our ability to continue as a going concern.
There is substantial doubt about our ability to continue as a going concern. Our Consolidated Financial Statements included in this Report are prepared using accounting principles generally accepted in the United States applicable to a going concern, which contemplates the realization of assets and liquidation of liabilities in the normal course of business. The historical consolidated financial statements included in this Report do not include any adjustments that might be necessary if we are unable to continue as a going concern. The report of our independent registered public accountants BDO USA, LLP, included in this Report, includes an explanatory paragraph related to our ability to continue as a going concern.
The assessment of our ability to continue as a going concern was made by management considering, among other factors: (1) the timing and number of approved products that will be reintroduced to the market and the related costs; (2) the suspension of shipment of all products manufactured by us and the requirements under the consent decree with the FDA; (3) the possibility that we may need to obtain additional capital despite the proceeds from the offering of the 2015 Notes in March 2011 and the equity we were able to issue in February 2011 (see Note 1—“Description of Business—Significant Developments” of the Notes to Consolidated Financial
43
Table of Contents
Statements included in this Report); (4) the potential outcome with respect to the governmental inquiries, litigation or other matters described in Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report; (5) our ability to comply with debt covenants and (6) our ability to obtain future revenues from the sales of Makena™ sufficient to meet our future needs and expectations. Our assessment was further affected by our fiscal year 2011 net loss of $174.0 million, our 2010 net loss of $283.6 million, and the outstanding balance of cash and cash equivalents of $137.6 million as of March 31, 2011. For periods subsequent to March 31, 2011, we expect losses to continue because we are unable to generate any significant revenues from more of our own manufactured products until we are able to begin selling more of our approved products with FDA’s approval and with respect to products manufactured for us by third parties until after we are able to generate significant sales of Makena™ which was approved by the FDA in February 2011. We received notification from the FDA on September 8, 2010 of approval for our discontinued generics operations to ship into the marketplace the first product approved under the consent decree, i.e., the Potassium Chloride Extended Release Capsule. We resumed shipment of extended-release potassium chloride capsule, Micro-K® 10mEq and Micro-K® 8mEq, in September 2010, resumed shipments of the generic version of Potassium Chloride Extended Release Capsule in December 2010 and we began shipping Makena™ in March 2011. We have continued to ship Evamist®, which is manufactured for the Company by a third party, during the period covered by the consent decree. We are continuing to prepare other products for FDA inspection and do not expect to resume shipping other products until the second half of fiscal year 2012, at the earliest. In addition, we must meet ongoing operating costs as well as costs related to the steps we are currently taking to introduce Makena™, and to prepare for introducing and reintroducing other approved products to the market. If we are not able to obtain the FDA’s clearance to resume manufacturing and distribution of more of our approved products in a timely manner and at a reasonable cost, or if revenues from the sale of approved products introduced or reintroduced into the market place prove to be insufficient, our financial position, results of operations, cash flows and liquidity will continue to be materially adversely affected. These conditions raise substantial doubt about our ability to continue as a going concern.
Based on current financial projections, we believe the continuation of our Company as a going concern is primarily dependent on our ability to address, among other factors: (1) sales of Makena™, (which was approved by the FDA) in February 2011 notwithstanding recent actions by the FDA to permit continued sale of compounded alternatives and by CMS to permit compounded alternatives to be reimbursed under the Medicaid program; (2) the timing, number and revenue generation of approved products that will be introduced or reintroduced to the market and the related costs; (3) the suspension of shipment of all products manufactured by us and the requirements under the consent decree with the FDA (other than the Potassium Chloride Extended Release Capsule, including Micro-K® 10mEq and Micro-K® 8mEq, products that are the subject of the FDA notification letter discussed above); (4) the possibility that we will need to obtain additional capital; (5) the potential outcome with respect to the governmental inquiries, litigation or other matters described in Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report; and (6) compliance with our debt covenants. While we address these matters, we must continue to meet expected near-term obligations, including normal course operating cash requirements and costs associated with introducing or reintroducing approved products to the market (such as costs related to our employees, facilities and FDA compliance), remaining payments associated with the acquisition and retention of the rights to Makena™ (see Note 5—“Acquisition” of the Notes to Consolidated Financial Statements included in this Report), the financial obligations pursuant to the plea agreement, costs associated with our legal counsel and consultant fees, as well as the significant costs associated with the steps taken by us in connection with the consent decree and the litigation and governmental inquiries. If we are not able to obtain the FDA’s clearance to resume manufacturing and distribution of certain or many of our approved products in a timely manner and at a reasonable cost and/or if we are unable to successfully commercialize Makena™, and/or if we experience adverse outcomes with respect to any of the governmental inquiries or litigation described in Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report, our financial position, results of operations, cash flows and liquidity will continue to be materially adversely affected. See “Risk Factors” included in Item I, Part 1A of this Report regarding additional risks we face with respect to these matters.
44
Table of Contents
In the near term, we are focused on performing the following: (1) addressing the FDA’s concerns regarding patient access to MakenaTM and CMS policy permitting Medicaid program reimbursement of compounded products; (2) the continued commercial launch of Makena™; (3) meeting the requirements of the consent decree, which will allow our approved products to be reintroduced to the market (other than the Potassium Chloride Extended Release Capsule products, including Micro-K® 10mEq and Micro-K® 8mEq, that are the subject of the FDA notification letter previously discussed); (4) the divestiture of Nesher and the Company’s generics business; and (5) pursuing various means to minimize operating costs and increase cash. Since December 31, 2010, the Company has generated non-recurring cash proceeds to support its on-going operating and compliance requirements from a $32.3 million private placement of Class A Common Stock in February 2011 and the 2015 Notes in March 2011 (which were used, in part, to repay all existing obligations under the agreement with U.S. Healthcare) (see Note 13—“Long-Term Debt” of the Notes to Consolidated Financial Statements included in this Report for a description of the 2015 Notes and U.S. Healthcare loan). While the cash proceeds received to date were sufficient to meet near-term cash requirements, we are pursuing ongoing efforts to increase cash, including, the continued implementation of cost savings, the divestiture of Nesher and the Company’s generics business and other assets and the return of certain additional approved products to market in a timely manner. We cannot provide assurance that we will be able to realize additional cost reductions from reducing our operations, that some or many of our approved products can be returned to the market in a timely manner (other than the Potassium Chloride Extended Release Capsule, including Micro-K® 10mEq and Micro-K® 8mEq products that are the subject of the FDA notification letter previously discussed above), that our higher profit approved products will return to the market in the near term or at all or that we can obtain additional cash through asset sales or the sale of equity or the successful commercial launch of Makena™. If we are unsuccessful in our efforts to introduce or return our products to market at adequate levels, or to sell assets or raise additional equity, we may be required to further reduce our operations, including further reductions of our employee base, or we may be required to cease certain or all of our operations in order to offset the lack of available funding.
We continue to evaluate the sale of certain of our assets, including Nesher and our generics business, which is included in discontinued operations and assets held for sale. To date, we are in advanced discussions for the sale of our generics business; however, we cannot assure that we will complete any transaction on favorable terms or at all. The Company is continuing to work with its advisors and interested parties to complete a transaction. However, due to general economic conditions, we will likely be exposed to risks related to the overall macro-economic environment, including a lower rate of return than we have historically experienced on our invested assets and being limited in our ability to sell assets. In addition, we cannot provide any assurance that we ultimately will be successful in finding suitable purchasers for the sale of such assets. Even if we are able to find purchasers, we may not be able to obtain attractive terms and conditions for such sales, including attractive pricing. In addition, divestitures of businesses involve a number of risks, including the diversion of management and employee attention, significant costs and expenses, the loss of customer relationships, a decrease in revenues and earnings associated with the divested business, and the disruption of operations in the affected business. In addition, divestitures potentially involve significant post-closing separation activities, which could involve the expenditure of significant financial and employee resources. Inability to consummate identified asset sales or manage the post-separation transition arrangements could adversely affect our business, financial condition, results of operations and cash flows.
Our future business success in the next several years, as well as the continuing operation of our Company, depends critically upon our successful market launch of Makena™ and our ability to achieve revenues from the sale of Makena™ consistent with our business expectations. A failure to achieve these objectives and sufficient market success in selling Makena™ will materially adversely affect the success and viability of our Company and would likely result in a default under our debt obligations.
On January 16, 2008, the Company entered into an Asset Purchase Agreement (the “Original Makena™ Agreement”) with Cytyc Prenatal Products Corp. and Hologic, Inc. (Cytyc Prenatal Products Corp. and Hologic, Inc. are referred to collectively as “Hologic”), as further described in Note 5—“Acquisitions” of the Notes to Consolidated Financial Statements included in this Report. Under our agreement with Hologic, we completed the
45
Table of Contents
acquisition of Makena™ upon making a $12.5 million additional payment to Hologic on February 10, 2011 and are currently undertaking the commercial launch of Makena™. Under our agreement with Hologic, we must make subsequent additional milestone payments and our payment obligations are secured by a lien on our rights to Makena™ granted to Hologic. We have certain revenue expectations with respect to both the sale of Makena™ as well as the sales of our approved products that are allowed to return to the market by FDA following successful inspections under the consent decree. If we cannot timely and successfully commercially launch Makena™, and achieve those revenue expectations with respect to Makena™, this would result in material adverse impact on our results of operations and liquidity, and ability to continue as a going concern.
Moreover, if we fail to pay to Hologic any of the remaining payments when they mature under our agreement, as amended, with Hologic, our rights to the Makena™ assets will transfer back to Hologic.
As discussed in Note 5—“Acquisition” of the Notes to Consolidated Financial Statements included in this Report, we modified the Original Makena™ Agreement pursuant to an amendment entered into in January 2010 (“Amendment No. 1”). Pursuant to Amendment No. 1, we made a $70 million cash payment to Hologic upon execution of Amendment No. 1. We entered into a second amendment to the Original Makena™ Agreement on February 4, 2011 (“Amendment No. 2”). Under the Original Makena™ Agreement, as amended (the “Amended Makena™ Agreement”), after the $12.5 million payment we made to Hologic on February 10, 2011, we are required to pay a series of additional future scheduled cash payments in the aggregate amount of $107.5 million upon successful completion of agreed upon milestones. We also may become obligated to pay additional amounts as royalties if we elect to defer certain milestone payments. (The date on which we make the final cash payment is referred to as the “Final Payment Date.”) If, prior to the Final Payment Date, we fail to timely pay a scheduled payment, we are obligated to transfer back to Hologic ownership of the Purchased Assets (as defined in the Amended Makena™ Agreement), including certain improvements made thereto by us, as well as other after-acquired assets and rights used by us in connection with the Makena™ business (the “Retransfer”). If the Retransfer were to occur, we would not be reimbursed for the payments we have made up to that point to Hologic under the Amended Makena™ Agreement. Our failure to pay any of the remaining payments when required under the Amended Makena™ Agreement and the resulting Retransfer would have a material adverse effect on our business, financial condition, results of operations and cash flows.
In connection with its approval under subpart H regulations, the FDA granted an orphan drug designation for Makena™. As part of this designation, the Company was granted a seven-year marketing exclusivity period. The sub-part H regulations allow certain drugs for serious conditions to be submitted for FDA marketing approval under the basis of one controlled clinical trial instead of the usual case of two clinical trials. Typically there is an additional post-marketing commitment to perform a second confirmatory clinical trial. If this trial does not replicate the positive results of the original trial, the FDA can take various actions such as requesting another clinical trial or withdrawing the conditional approval. We cannot be certain of the results of the confirmatory clinical trial (expected in 2016) and what action the FDA may take if the results were not as expected based on the first clinical trial.
The success of the Company’s commercialization of Makena™ is dependent upon a number of factors, including: (1) the Company’s ability to maintain certain net pricing levels and unit sales for Makena™; (2) successfully obtaining agreements for coverage and reimbursement rates on behalf of patients and medical practitioners prescribing Makena™ with third-party payors, including government authorities, private health insurers and other organizations, such as health maintenance organizations (“HMOs”), insurance companies, and Medicaid programs and administrators, and (3) the extent to which pharmaceutical compounders continue to produce non-FDA approved purported substitute product. The Company has been criticized regarding the list pricing of Makena™ in numerous news articles and internet postings. In addition, the Company has received, and may continue to receive, letters criticizing the Company’s list pricing of Makena™ from numerous medical practitioners and advocacy groups, including the March of Dimes, American College of Obstetricians and Gynecologists, American Academy of Pediatrics and the Society for Maternal Fetal Medicine. Several of these advocacy groups have also issued their own press releases regarding their criticism of the pricing of Makena™
46
Table of Contents
and endorsing the statements made by the FDA regarding compounded product (discussed below). In addition, the Company is aware that certain doctors have chosen to continue prescribing the non-FDA approved purported substitute product made by pharmaceutical compounders in lieu of even considering prescribing Makena™.
Further, the Company has received letters from United States Senators and members of the United States Congress asking the Company to reduce its indicated pricing of Makena™ and requesting information with respect to Makena™, its pricing and the Company’s cost relating to Makena™. One of the Senators also sent a letter to CMS asking for CMS’ views on the ramification of the pricing of Makena™ on the Medicaid system and, together with another Senator, has sent a letter to the Federal Trade Commission asking the agency to initiate an investigation of our pricing of Makena™. Staff members of the U.S. Senate Finance Committee have also advised the Company that federal legislation targeted at the Company’s sale of Makena™ may be introduced unless the Company reduces its price. Communications with members of Congress and their staffs indicate that hearings in Congress on the Company’s pricing of Makena™ may occur. The FDA has communicated to the Company and also separately issued a press release that, in order to ensure continued access for patients needing hydroxyprogesterone caproate that the FDA intended to refrain at this time from taking enforcement action with respect to compounding pharmacies producing compounded hydroxyprogesterone caproate in response to individual prescriptions for individual patients. The impact of FDA’s statement on the effectiveness of the Company’s orphan drug marketing exclusivity is at present still unclear but a failure by the FDA to take enforcement action against compounding pharmacies may result in substantial sales of compounded alternatives to Makena™ and effective loss of some or all of such marketing exclusivity for the affected period of time. The Company’s representatives met with the FDA staff to discuss access to Makena™ and to provide information to the FDA relevant to its public statement.
In addition, CMS issued an informational bulletin to state Medicaid programs that they can choose to pay for the extemporaneously compounded hydroxyprogesterone caproate as an active pharmaceutical ingredient (“API”) and this can be covered under the “medical supplies, equipment and appliances suitable for use in the home” portion of home health. Because CMS does not require states to list all of the items they cover under this section in the Medicaid state plan, states can cover hydroxyprogesterone caproate under their current state plans and do not need to submit a state plan amendment to provide for such coverage. The Company believes that this has the potential of excluding Makena™ from being provided under the various state Medicaid programs. The Company estimates that state Medicaid programs cover approximately 40% to 45% of the total number of pregnancies in the United States.
The Company is responding to these criticisms and events in a number of respects, including the announced reduction in the published list price of Makena™ from $1,500 per injection to $690 per injection on April 1, 2011 (prior to expected further discounting of such list price by the mandatory 23.1% Medicaid rebate and other supplemental rebates and discounts currently under negotiation with public and private payors), and the expansion of an already announced patient assistance program for patients who are not covered by health insurance or could otherwise not afford Makena™ or their respective co-pays. Further, the Company is working directly with health insurers, pharmacy benefit managers, Medicaid management companies, and others regarding the net cost of Makena™ coverage and reimbursement programs and other means by which Makena™ would be available to patients. The Company can give no assurance as to whether these responses and negotiations will be successful at obtaining an economically sufficient price or unit sales for Makena™.
The commercial success and viability of the Company is largely dependent upon these efforts and appropriately responding to the media, physician, institutional, advocacy group and governmental concerns and actions regarding the pricing of Makena™. The Company has substantial debt and liabilities that come due over the next several years and the pricing and revenues that the Company must achieve from the sale of Makena™, together with our sales of other products, must be substantial enough to allow us to meet these obligations, refinance or retire such debt and liabilities when due, and generate sufficient profits to ensure the Company’s viability as a pharmaceutical company prior to the end of the orphan drug exclusivity period for Makena™.
47
Table of Contents
We have decided to sell certain assets and to explore strategic alternatives with respect to other assets, including Nesher and our generics business and assets. Such sales could pose risks and may materially adversely affect our business. Our failure to liquidate or sell assets on terms favorable to us, or at all, could have a material adverse effect on our financial condition and cash flows.
We have completed the sale of substantially all of the assets of PDI, and the sale of certain other assets. We currently plan to sell certain of our other assets, and are working to divest Nesher, our generic products business. To date, we have received several initial offers for Nesher. The Company is continuing to work with its advisers and interested parties to complete a transaction. However, due to the general economic slowdown, we will likely be exposed to risks related to the overall macro-economic environment, including a lower rate of return than we have historically experienced on our invested assets and being limited in our ability to sell assets or to identify and carry out advantageous strategic alternatives.
As noted above, we plan to market for sale certain assets and will seek to identify other assets for potential sale and to continue to explore strategic alternatives for our generics products business. However, we cannot provide any assurance that we will ultimately be successful in finding suitable purchasers for the sale of such assets. Even if we are able to find purchasers, we may not be able to obtain attractive terms and conditions for such sales, including attractive pricing. In addition, divestitures of businesses may also involve a number of risks, including the diversion of management and employee attention, significant costs and expenses, the loss of customer relationships, a decrease in revenues and earnings associated with the divested business, and the disruption of operations in the affected business. In addition, divestitures potentially involve significant post-closing separation activities, which could involve the expenditure of significant financial and employee resources.
Our inability to consummate identified sales, manage the post-separation transition arrangements, or identify and carry out advantageous strategic alternatives could adversely affect our business, financial condition, results of operations and cash flows.
If we are unable to address the issues identified in the consent decree and resume manufacturing and distribution of more of our approved products in a timely and cost effective manner, our business, financial position, results of operations and cash flows will continue to be materially adversely affected.
On March 2, 2009, we entered into a consent decree with the FDA regarding our drug manufacturing and distribution, which is described in more detail in “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Discontinuation of Manufacturing and Distribution; Product Recalls; and the FDA Consent Decree” in Part II, Item 7 of this Report. Our actions and the requirements under the consent decree have had, and are expected to continue to have, a material adverse effect on our liquidity position and operating results. Although we have recently resumed manufacturing and shipping of our Potassium Chloride Extended Release Capsule products, we do not expect to generate any significant revenues from products that we manufacture until we resume shipping certain or many of our approved products after successful FDA inspections relevant to those products. In the meantime, we must meet ongoing operating costs related to our employees, facilities and FDA compliance, as well as costs related to the steps we currently are taking to prepare for reintroducing additional products to the market.
The consent decree required us, before resuming manufacturing, to retain an independent cGMP expert to undertake a review of our facilities, policies, procedures and practices and to certify compliance with the FDA’s cGMP regulations. On January 13, 2010, our independent cGMP expert, Lachman, notified the FDA that it had performed a comprehensive inspection and that our facilities and controls are in compliance with cGMP and the consent decree but advised us to enact further enhancements to certain aspects of our cGMP systems. In accordance with the advice from Lachman, we continued to enhance our cGMP systems, and Lachman subsequently reinspected our cGMP systems and on April 26, 2010 certified our compliance with all cGMP systems requirements. On July 27, 2010, we received a certification from Lachman regarding the first group of
48
Table of Contents
our approved products for which we sought FDA approval to manufacture and return to the market. In August 2010, the FDA re-inspected our facilities without issuing any adverse findings, and in September 2010, the FDA determined that our facilities are in compliance, allowing us to resume shipment of our Potassium Chloride Extended Release Capsule products. We are currently carrying out the preparatory work on the next group of products for which Lachman certification and FDA approval is sought.
We currently do not expect to resume shipments of additional products until sometime in the second half of fiscal year 2012. In addition, we expect that any resumption of shipments of additional products will be limited initially to selected products and will be expanded incrementally thereafter. Despite our efforts, there can be no assurance that our initiatives with respect to the additional products that are intended to comply with the requirements under the consent decree and enable us to reintroduce certain of our other approved products to the market will be successful within the time frames currently projected by management or at all. If we are not able to obtain the FDA’s permission to resume manufacturing and distribution of our other products in a timely manner at a reasonable cost, our business, financial position, results of operations and cash flows will continue to be materially adversely affected, which would have a material adverse effect on our ability to continue as a going concern.
In addition, one of our top priorities is to maintain and attempt to increase our cash and financial resources. As a result, if we determine that our current goal of meeting the consent decree requirements and returning our other approved products to market is likely to be significantly delayed, we may decide to further reduce our operations, including further reductions of our employee base, and to significantly curtail some or all of our efforts to meet the consent decree requirements and return our approved products to market. Such decision would be made on an analysis of the costs and benefits of bringing particular additional approved products back to the marketplace as well as based on our ability to manage our near-term cash obligations, to obtain additional capital through asset sales and/or external financing and to expeditiously meet the consent decree requirements and return our approved products to market. If such decision were to be made, we currently anticipate that we would focus our efforts on developing product candidates in our development portfolio that we believe have the highest potential return on investment, which we currently believe to be primarily Makena™. We also expect to evaluate other alternatives available to us in order to increase our cash balance.
Even if we are able to address the issues identified in the consent decree and resume manufacturing and distribution of some or all of our other approved products in a timely and cost-effective manner, our business, financial position, results of operations and cash flows could continue to be materially adversely affected.
As discussed above, we have been unable to manufacture or ship any of our products for an extended period of time, other than, recently, our Potassium Chloride Extended Release Capsule products. We operate in a highly competitive industry and it is possible that, even if we are able to return some or all of our other approved products to the market, certain of our existing customers will purchase smaller quantities or no quantities of our products. Such a potential loss of market share would likely result in limiting the prices we are able to charge for our approved products, which will negatively impact our gross margin.
In addition, our financial position is expected to be adversely affected by our inability to manufacture and distribute our unapproved products until such time as there is an approved ANDA or NDA for each such product, which will only occur if we decide to pursue, and are able to fund, the studies required for such approvals over an extended period of time. It is possible that we may not be able to return all or any of our unapproved products to market. Moreover, entering into the consent decree, as well as the criticism regarding the list pricing of Makena™, may have damaged our reputation in the market, which could result in a competitive disadvantage. Furthermore, general economic conditions, industry cycles and financial, business and other factors affecting our operations, many of which are beyond our control, may affect our future performance. Therefore, even after we are able to resume manufacturing and shipment of our other approved products, our business might not continue to generate cash flow at or above historic levels, which could have a material adverse effect on our financial position, results of operations and cash flows.
49
Table of Contents
We face continuing risks in connection with the plea agreement with the Department of Justice related to allegations of failure to make and submit field alert reports to the FDA in September 2008.
We, at the direction of a special committee of the Board of Directors that was in place prior to June 10, 2010, responded to requests for information from the Office of the United States Attorney for the Eastern District of Missouri and FDA representatives working with that office. As more fully described in Note 1—“Description of Business—Plea Agreement with the U.S. Department of Justice” of the Notes to Consolidated Financial Statements included in this Report, our subsidiary ETHEX entered into a plea agreement with the U.S. Department of Justice that relates to allegations of failure to make and submit field alert reports to the FDA in September 2008 regarding the discovery of certain undistributed tablets that failed to meet product specifications. In exchange for the voluntary guilty plea by ETHEX, the Department of Justice agreed that no further federal prosecution will be brought in the Eastern District of Missouri against ETHEX, KV or Ther-Rx regarding allegations of the misbranding and adulteration of any oversized tablets of drugs manufactured by us, and the failure to file required reports regarding these drugs and patients’ use of these drugs with the FDA, during the period commencing on January 1, 2008 through December 31, 2008. However, the plea agreement does not bind any governmental office or agency other than the United States Attorney for the Eastern District of Missouri and the Office of Consumer Litigation and we cannot rule out regulatory or other actions by governmental entities not party to the plea agreement. In addition, the plea agreement does not limit the rights and authority of the United States of America to take any civil, tax or administrative action against us, and any recommendation in the plea agreement as to the amount of loss or restitution is not binding upon the United States in any civil or administrative action by the government against us.
We may also become subject to claims by private parties with respect to the alleged conduct that is the subject of the plea agreement.
Pursuant to the plea agreement, ETHEX is subject to a criminal fine in the aggregate amount of $23.4 million, payable in four installments of which we currently have $20.1 million remaining to be paid. On November 15, 2010, upon the motion by the Department of Justice, the court vacated the previous fine installment schedule and imposed a new fine installment schedule, which did not change the total fine, using the standard federal judgment rate of 0.22% per annum, payable as follows:
| Payment Amount including interest ($ in millions) |
Payment Due Date | |
| $ 1.0 |
December 15, 2010 | |
| 1.0 |
June 15, 2011 | |
| 1.0 |
December 15, 2011 | |
| 2.0 |
June 15, 2012 | |
| 4.0 |
December 15, 2012 | |
| 5.0 |
June 15, 2013 | |
| 7.1 |
December 15, 2013 |
The Company made the December 15, 2010 payment as scheduled. If we fail to make any of the remaining installment payments, the U.S. Attorney’s Office, in its sole discretion, may void the plea agreement, keep any payments already made under the plea agreement and prosecute us using, among other evidence, the admissions made in the plea agreement.
An adverse resolution of the private and government litigation and governmental inquiries could have a material adverse effect on our business, financial condition, results of operations and cash flows.
We are fully cooperating in certain governmental matters, including SEC informal inquiries, pending litigations with the States of Utah and Louisiana with respect to AWP with respect to ETHEX products in past years, and the pending investigation by HHS OIG and the U.S. Attorney’s Office in Boston into potential false
50
Table of Contents
claims under Title 42 of the U.S. Code with respect to a qui tam action (see Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report). We have also received civil investigative demands from the States of Florida and Texas that appear to pertain to the pricing of certain ETHEX products in past years and to Makena™, respectively. We are not aware whether, or the extent to which, any pending governmental inquiries and/or related private litigation might result in the payment of fines, penalties or judgments or the imposition of operating restrictions on our business; however, if we are required to pay fines, penalties or judgments, the amount could be material.
Furthermore, any governmental enforcement action could require us to operate under significant restrictions, place substantial burdens on management, hinder our ability to attract and retain qualified employees and/or cause us to incur significant costs or damages.
In connection with the guilty plea by ETHEX pursuant to the plea agreement, ETHEX was expected to be excluded from participation in federal healthcare programs, including Medicare and Medicaid. As a result, HHS OIG had discretionary authority to seek to similarly exclude our Company from participation in federal healthcare programs. In addition, in light of the exclusion of Marc S. Hermelin on November 18, 2010, HHS OIG had discretionary authority to seek to similarly exclude our Company from participation in federal healthcare programs. However, on November 10, 2010, Mr. M. Hermelin voluntarily resigned as a member of the Board of Directors. We had been advised that HHS OIG notified Mr. M. Hermelin that he would be excluded from participating in federal healthcare programs effective November 18, 2010. In an effort to avoid adverse consequences to our Company, including the foregoing potential discretionary exclusion of our Company, and to enable our Company to secure our expanded financial agreement, HHS OIG, Mr. M. Hermelin and his wife (solely with respect to shares owned jointly between them and certain other obligations therein) entered into a settlement agreement (the “Settlement Agreement”) under which Mr. M. Hermelin also resigned as trustee of all family trusts that hold KV stock, agreed to divest his personal ownership interests in our Company’s Class A Common and Class B Common stock (approximately 1.8 million shares), including certain shares owned jointly with this wife over an agreed upon period of time in accordance with a divestiture plan and schedule approved by HHS OIG, and agreed to refrain from voting stock under his personal control. In order to implement such agreement, Mr. M. Hermelin and his wife granted to an independent third party immediate irrevocable proxies and powers of attorney to divest his (and their jointly owned) personal stock interests in our Company if Mr. M. Hermelin does not timely do so. The Settlement Agreement also required Mr. M. Hermelin to agree, for the duration of his exclusion, not to seek to influence or be involved with, in any manner, the governance, management, or operations of our Company. As long as the parties comply with the Settlement Agreement, HHS OIG has agreed not to exercise its discretionary authority to exclude our Company from participation in federal health care programs, thereby allowing our Company and our subsidiaries (with the single exception of ETHEX, which was filed for dissolution under state law effective December 15, 2010, pursuant to the Divestiture Agreement with HHS OIG) to continue to conduct business through all federal and state healthcare programs. The Company is also a signatory to the Settlement Agreement between HHS OIG and Mr. M. Hermelin with respect to certain obligations therein, including adopting certain internal policies and notices, reporting violations, reimbursing certain fees of a third party acting under the agreement, and taking certain actions in support of the agreement, including providing certain assistance in the sale of the shares being divested thereunder.
On November 15, 2010, we entered into the Divestiture Agreement with HHS OIG under which we agreed to sell the assets and operations of ETHEX to unrelated third parties prior to April 28, 2011 and to file articles of dissolution with respect to ETHEX under Missouri law by that date. We filed articles of dissolution for ETHEX on December 15, 2010. Following the filing, ETHEX may not engage in any new business other than winding up its operations and will engage in a process provided under Missouri law to identify and resolve its liabilities over at least a two-year period. Under the terms of the Divestiture Agreement, HHS OIG agreed not to exclude ETHEX from federal healthcare programs until April 28, 2011 and, upon completion of the sale of the ETHEX assets and of the filing of the articles of dissolution of ETHEX, the agreement will terminate. Civil monetary penalties and exclusion of ETHEX could have occurred if we had failed to meet our April 28, 2011 deadline. The
51
Table of Contents
sales of ETHEX’s assets (other than certain fixtures as to which HHS OIG has consented to non-divestiture) were completed prior to the April 28, 2011 deadline and ETHEX no longer has any ongoing material assets or operations other than those required to conclude the winding up process under Missouri law. We have also received a letter from HHS OIG advising us further that assuming that we have complied with all agreements deemed necessary by HHS OIG, HHS OIG would not exclude ETHEX thereafter. On April 1, 2011, we requested confirmation from HHS OIG that the steps and actions described in our reports to HHS OIG constituted full compliance with the duties KV and ETHEX were to complete by April 28, 2011 under the Divestiture Agreement, such that they are not at risk for stipulated penalties or exclusion after that date under the Divestiture Agreement. On April 8, 2011, we received a letter from HHS OIG stating that, at this time, based upon the information provided to HHS OIG in our monthly submissions, HHS OIG has no concerns regarding the actions taken by KV and ETHEX pursuant to the Divestiture Agreement. On May 20, 2011, we received a letter from HHS OIG stating that based on its review of the information provided in our monthly reports, it appeared that the Company and ETHEX had completed our obligations under the Divestiture Agreement.
As a result of the foregoing actions and agreements entered into by Mr. M. Hermelin, the two agreements with HHS OIG and the referenced correspondence with HHS OIG, we believe we have resolved our remaining issues with respect to HHS OIG and are positioned to continue to participate in federal healthcare programs now and in the future. However, a failure by the parties to comply with the Settlement Agreement or the Divestiture Agreement could lead to future exclusion of our Company under certain circumstances and any such exclusion would materially harm our Company and its future business and viability.
In addition, we are subject to a number of private litigation matters as more fully described in Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report. If we do not prevail in one or more pending lawsuits, we may be required to pay a significant amount of monetary damages.
Our ongoing private litigation and governmental inquiries also could impair our ability to raise additional capital.
Continuing negative publicity from the consent decree, the plea agreement, our list pricing of Makena™ and ongoing litigation and governmental inquiries may have a material adverse effect on our business, financial condition, results of operations and cash flows.
As a result of the consent decree, the plea agreement, our list pricing of Makena™, ongoing litigation and governmental inquiries and related matters, we have been the subject of negative publicity. This negative publicity may harm our relationships with current and future investors, government regulators, employees, customers and vendors. For example, negative publicity may adversely affect our reputation, which could harm our ability to obtain new customers, maintain existing business relationships with other parties and maintain a viable business in the future. Also, it is possible that the negative publicity and its effect on our work environment could cause our employees to terminate their employment or, if they remain employed by us, result in reduced morale that could have a material adverse effect on our business. In addition, negative publicity has and may continue to adversely affect our stock price and, therefore, employees and prospective employees may also consider our stability and the value of any equity incentives when making decisions regarding employment opportunities. As a result, our business, financial condition, results of operations and cash flows could be materially adversely affected.
The consent decree, the plea agreement, the Settlement Agreement, the Divestiture Agreement, our list pricing of Makena™ and the ongoing governmental and private litigation and governmental inquiries have resulted in significant fees, costs and expenses, diverted management time and resources, and could have a material adverse effect on our business, financial condition and cash flows.
We have incurred significant costs associated with, or consequential to, the steps taken by us in connection with the nationwide recall and suspension of shipment of all products manufactured by us, the requirements
52
Table of Contents
under the consent decree, the plea agreement, the Settlement Agreement, the Divestiture Agreement, our list pricing of Makena™ and the ongoing governmental and private litigation and governmental inquiries, including legal fees (including advancement of legal fees to individuals pursuant to our indemnification obligations), accounting fees, consulting fees and similar expenses. Although we believe that a portion of these expenditures may be recoverable from insurance policies that we have purchased, the insurance does not extend to all of these expenditures, the insurance limits may be insufficient even with respect to expenditures that would otherwise be covered, and the insurance carriers have reserved their rights to contest coverage under the insurance policies on multiple grounds. In addition, our Board, management and employees have expended a substantial amount of time in connection with these matters, diverting resources and attention that would otherwise have been directed toward our operations and implementation of our business strategy.
We expect to continue to spend additional time and incur significant additional professional fees, expenses and costs, including administrative expenses similar to those discussed above, as well as costs associated with the remedial activities adopted by the Audit Committee or the Board.
Pursuant to the plea agreement, we are subject to fines, restitution and forfeiture in the remaining aggregate amount of $20.1 million still to be paid. In addition, we are not aware whether, or the extent to which, any pending governmental inquiries and/or related private litigation might result in the payment of fines, penalties or judgments or the imposition of operating restrictions on our business; however, if we are required to pay fines, penalties or judgments, the amount could be material. In addition, if we do not prevail in one or more pending lawsuits, we may be required to pay a significant amount of monetary damages, which could have a material adverse effect on our financial position, results of operations and cash flows.
We have only recently become current in the filing of our periodic reports with the SEC.
Our efforts to become and remain current with our SEC filings have required and will continue to require substantial management time and attention as well as additional accounting and legal expense. Investors in our securities will not have information regarding the current state of our business and financial condition with which to make decisions regarding investment in our securities. When this information does become available to investors, it may result in an adverse effect on the trading price of our common stock. We will not be able to conduct any registered offerings unless we remain current in our SEC filings and we will not be eligible to use a “short form” registration statement on Form S-3 for a period of 12 months after the time we became current in our SEC filings. If we are not able to timely file and make effective registration statements prior to the dates required under various of our material agreements with our equity and debt holders, we may accrue substantial penalties. Until we are again eligible to use Form S-3, we would be required to use a registration statement on Form S-1 to register securities with the SEC or issue such securities in a private placement, which could increase the cost of raising capital. If we do not remain current with our SEC filings, our securities may be delisted from the NYSE and we may not maintain compliance with certain of our debt covenants.
We are involved in various legal proceedings and may experience unfavorable outcomes of such proceedings.
We are involved in various legal proceedings, including, but not limited to, patent infringement, product liability, breach of contract, stockholder class action lawsuits, qui tam and governmental false claims and pricing lawsuits, and proceedings relating to Medicaid reimbursements that involve claims for, or the possibility of, fines and penalties involving substantial amounts of money or other relief (see the Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report ). Any adverse determination with respect to any legal proceedings or inquiries could have a material adverse effect on our business, financial position and results of operations.
The Board in December 2008 appointed a special committee in response to the initiation of a series of putative class action stockholder lawsuits alleging violations of the federal securities laws by our Company and certain individuals, the initiation of lawsuits alleging violations under the Employee Retirement Income Security
53
Table of Contents
Act (“ERISA”), as well as the receipt by our Company of an informal inquiry from the SEC and certain requests for information from the Office of the United States Attorney for the Eastern District of Missouri and FDA representatives working with that office.
With respect to the securities and ERISA claims and related governmental inquiries, we maintain directors’ and officers’ liability insurance that we believe should cover a portion of the defense and potential liability costs associated with these matters. Nonetheless, the insurance does not extend to all of these expenditures, and the insurance limits may be insufficient even with respect to expenditures that would otherwise be covered. Moreover, the insurance carriers have reserved their rights to contest coverage under the insurance policies on multiple grounds. The expenses associated with these matters have been substantial, and we expect that they will continue to be so. Furthermore, defense of the litigations and cooperation with ongoing governmental inquiries is expected to divert management attention from normal course business operations. An adverse outcome with respect to these matters could have a material adverse effect on our business, financial position and results of operations.
In addition, as previously disclosed, on December 5, 2008, the Board terminated the employment agreement of Marc S. Hermelin, the Chief Executive Officer of the Company at that time, “for cause” (as that term is defined in such employment agreement). Additionally, the Board removed Mr. M. Hermelin as Chairman of the Board and as the Chief Executive Officer, effective December 5, 2008. In accordance with the termination provisions of his employment agreement, the Company determined that Mr. M. Hermelin would not be entitled to any severance benefits. In addition, as a result of Mr. M. Hermelin’s termination “for cause,” the Company determined it was no longer obligated for the retirement benefits specified in the employment agreement. However, Mr. M. Hermelin informed the Company that he believed he effectively retired from his employment with the Company prior to the termination of his employment agreement on December 5, 2008 by the Board. Although no litigation is pending regarding this matter, to the extent Mr. M. Hermelin elects to bring litigation and in the event we ultimately are unsuccessful in such litigation, we may be required to pay substantial amounts to Mr. M. Hermelin, which could have a material adverse effect on our financial condition, liquidity position and capital resources.
On March 22, 2011, Mr. M. Hermelin made a demand on the Company for indemnification with respect to his payment of $1.9 million imposed by the United States District Court as a fine and forfeiture of pecuniary gain as part of the sentence resulting from his guilty plea entered by the Court on March 10, 2011. Mr. M. Hermelin pled guilty to two federal misdemeanor counts as a responsible corporate officer of the Company at the time when a misbranding of two morphine sulfate tablets occurred which contained more of the active ingredient than stated on the label. In addition, the Company has advanced or will advance, under the terms of the Indemnification Agreement, legal expense amounting to approximately $5.0 million to a variety of law firms who represented Mr. M. Hermelin for legal matters including the FDA and SEC investigations, the Department of Justice inquiry, the Audit Committee investigation, HHS OIG exclusion and various class action lawsuits. Under the Company’s standard Indemnification Agreement entered into with all directors, including Mr. M. Hermelin when he served as Chairman of the Board and Chief Executive Officer of the Company, as a condition for the advancement of expenses, each director is required to sign an undertaking to reimburse the Company for the advanced expenses in the event it is found that the director is not entitled to indemnification. Mr. M. Hermelin’s demand for reimbursement of the $1.9 million fine and forfeiture, and whether the advanced legal fees to represent him for various legal matters should be indemnified, is under review by a special committee appointed by the Board of Directors of the Company.
We may be subject to substantial damages for product liability claims.
The design, development, manufacturing and sale of our products involve an inherent business risk of exposure to product liability claims by consumers and other third parties in the event that the use of our products is alleged to have resulted in adverse effects. In particular, the administration of drugs to humans may cause, or
54
Table of Contents
may appear to have caused, adverse side effects (including death) or potentially dangerous drug interactions that we may not learn about or understand fully until the drug has been administered to patients for some time.
As described in Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report, we are involved in various product liability claims, including both lawsuits and pre-litigation claims. Product liability claims are complex in nature and, regardless of their merits or their ultimate outcomes, are costly, divert management’s attention, may adversely affect our reputation, may reduce demand for our products and may result in significant damages. We may also become subject to claims by private parties with respect to the alleged conduct that is the subject of the plea agreement.
We currently have product liability insurance to protect against and manage the risks involved with our products, but we cannot provide assurances that the level or breadth of any insurance coverage will be sufficient to cover fully all potential claims. In addition, we are subject to the risk that our insurers will seek to deny coverage for claims that we believe should be covered. Our insurers have, in certain cases, reserved their rights with respect to certain claims or cases that have been brought against us. Furthermore, adequate insurance coverage might not be available in the future at acceptable costs, if at all. Significant judgments against us for product liability, and the high costs of defense against such lawsuits, not covered or not fully covered by insurance could have a material adverse effect on our business, financial condition, results of operations and cash flows. In addition, even if a product liability claim is not successful, the adverse publicity and time and expense of defending such a claim may interfere with our business.
Our industry is highly regulated and our products are subject to ongoing regulatory review, and even if we address the issues identified in the consent decree and resume manufacturing and distribution of more of our approved products, we likely will continue to be subject to heightened scrutiny with regard to our operations.
Our Company, our drug products, the manufacturing facilities for our drug products, the distribution of our drug products, and our promotion and marketing materials are subject to strict and continual review and periodic inspection by the FDA and other regulatory agencies for compliance with pre-approval and post-approval regulatory requirements, including cGMP regulations, adverse event reporting, advertising and product promotion regulations, and other requirements.
As a result of our consent decree and the plea agreement, we anticipate that we will be scrutinized more closely than other companies by the FDA and other regulatory agencies, even if we address the issues identified in the consent decree and resume manufacturing and distribution of additional products. Failure to comply with manufacturing and other post-approval state or federal laws, regulations of the FDA and other regulatory agencies can, among other things, result in warning letters, fines, increased compliance expense, denial or withdrawal of regulatory approvals, additional product recalls or seizures, forced discontinuance of or changes to important promotion and marketing campaigns, operating restrictions and criminal prosecution. The cost of compliance with pre- and post-approval regulation may have a negative effect on our operating results and financial condition.
In addition, the requirements or restrictions imposed on us or our products may change, either as a result of administratively adopted policies or regulations or as a result of the enactment of new laws and new government oversight. At present, the activities of pharmaceutical companies are subject to heightened scrutiny by federal and state regulators and legislators, and FDA enforcement is expected to increase. Any new statutory or regulatory provisions or policy changes could result in delays or increased costs during the period of product development, clinical trials, and regulatory review and approval, as well as increased costs to assure compliance with any new post-approval regulatory requirements.
55
Table of Contents
If we are unable to commercialize products under development or that we acquire, our future operating results may suffer.
Certain products we develop or acquire will require significant additional development and investment prior to their commercialization. Our research and development activities, pre-clinical studies and clinical trials (where required), manufacturing activities and the anticipated marketing of our product candidates are subject to extensive regulation by a wide range of governmental authorities in the United States, including the FDA. To satisfy FDA regulatory approval standards for the commercial sale of our product candidates, we must, among other requirements, demonstrate in adequate and well-controlled clinical trials that our product candidates are safe and effective.
Even if we believe that data from our pre-clinical and clinical studies demonstrates safety and efficacy, our analysis of such data is subject to confirmation and interpretation by the FDA, which may have different views on the design, scope or results of our clinical trials, which could delay, limit or prevent regulatory approval. The FDA wields substantial discretion in deciding whether a drug meets the approval criteria, and even if approved, such approval may be conditioned on, among other things, restricted promotion, restricted distribution, a risk evaluation mitigation strategy, or post-marketing studies. Such restrictions may negatively affect our ability to market the drug among competitor products, as well as adversely affect our business.
We expect that many of these products will not be commercially available for several years, if at all. We cannot assure you that such products or future products will be successfully developed, prove to be safe and effective in clinical trials (if required), meet applicable regulatory standards, or be capable of being manufactured in commercial quantities at reasonable cost or at all. If we are unable to commercialize products under development or that we acquire, our future operating results may suffer.
Even if we are able to address the issues identified in the consent decree and resume our drug development plans, some of our product candidates may have to undergo rigorous and expensive clinical trials, the results of which are uncertain and could substantially delay or prevent us from bringing drugs to market.
Before we receive regulatory approvals for the commercial sale of any of our drug candidates, our drug candidates are subject to extensive pre-clinical testing and clinical trials to demonstrate their safety and efficacy in humans. Conducting pre-clinical testing and clinical trials is a lengthy, time-consuming, expensive, and uncertain process that often takes many years. Furthermore, we cannot be sure that pre-clinical testing or clinical trials of any drug candidates will demonstrate the safety and efficacy of our drug candidates at all or to the extent necessary to obtain FDA approval. A number of companies in the pharmaceutical industry have suffered significant setbacks in advanced clinical trials, even after obtaining promising results in earlier pre-clinical studies and clinical trials.
We cannot assure you that we or our collaborators will successfully complete the planned clinical trials. Our collaborators or we may experience numerous unforeseen events during, or as a result of, the clinical trial process that could delay or prevent us from receiving regulatory approval or commercializing our product candidates, including the following events:
| • | our clinical trials may produce negative or inconclusive results, and we may decide, or the FDA may require us, to conduct additional clinical and/or pre-clinical studies or to abandon development programs; |
| • | trial results may not meet the level of statistical significance required by the FDA; |
| • | we, independent institutional review boards or the FDA, may suspend or terminate clinical trials if the participating patients are being exposed to unacceptable health risks; and |
| • | the effects of our product candidates on patients may not be the desired effects or may include undesirable side effects or other characteristics that may delay or preclude regulatory approval or limit their commercial use. |
56
Table of Contents
Even if we are able to address the issues identified in the consent decree and resume our drug development plans, our pre-clinical or clinical trials for some of our products under development may be unsuccessful or delayed.
Significant delays in clinical trials could materially affect our product development costs and delay regulatory approval of our product candidates. We do not know whether clinical trials will begin on time, will need to be redesigned or will be completed on schedule, if at all. There are a number of factors that may cause delays or suspension in clinical trials:
| • | delays or failures in obtaining regulatory authorization to commence a trial; |
| • | inability to manufacture sufficient quantities of acceptable materials for use in clinical trials; |
| • | the delay or failure in reaching agreement on contract terms with prospective study sites; |
| • | delays in recruiting patients to participate in a clinical trial; |
| • | the delay or failure in obtaining independent institutional review board review and approval of the clinical trial protocol; |
| • | the failure of third-party clinical trial managers, including clinical research organizations, to perform their oversight of the trials or meet expected deadlines; |
| • | the failure of our clinical investigational sites and related facilities and records to be in compliance with the FDA’s Good Clinical Practices, including the failure to pass FDA inspections of clinical trials; |
| • | unforeseen safety issues; |
| • | inability to secure clinical trial insurance; |
| • | lack of demonstrated efficacy in the clinical trials; |
| • | our inability to reach agreement with the FDA on a trial design that we are able to execute; |
| • | difficulty in adequately following up with patients after treatment; or |
| • | changes in laws, regulation, or regulatory policy. |
If clinical trials for our drug candidates are unsuccessful, delayed or cancelled, we will be unable to meet our anticipated development and commercialization timelines, which could harm our business and cause our stock price to decline.
Suspension of product shipments has exposed us to failure to supply claims from our customers and could expose us to additional claims in the future.
In addition to the loss of revenue, the suspension of product shipments exposes us to possible claims for certain additional costs. Pursuant to arrangements between us and certain of our customers, such customers, despite the formal discontinuation action by us of our products, may assert, and certain customers, including CVS Pharmacy, Inc., have asserted, that we should compensate such customers for any additional costs they incurred for procuring products we did not supply. The amount of such compensation is affected by the price of any replacement product and the terms of the relevant customer agreement. Following our suspension of shipments, the price of certain products increased significantly, thereby potentially increasing the amount of any such compensation. While we have recorded an estimated liability for failure to supply claims as of March 31, 2011 based on notices we received from our customers, the actual amount of liability from current and additional claims we may face, if asserted and determined to be meritorious, could be much higher and could have a material adverse effect on our liquidity position and operating results.
57
Table of Contents
Product recall costs had, and could continue to have, a material adverse effect on our business, financial position, results of operations and cash flows.
We have incurred significant costs and suffered economic losses as a result of the ongoing recall of our products and voluntary disposal of inventory in connection with the recall of our products (see Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report ). Also, as a result of the recall, production suspension of our products and the disposal of our existing inventory of products required under the consent decree, management established inventory reserves to cover estimated inventory losses for all work-in-process and finished goods related to drug products we manufactured, as well as raw materials for those drug products that had no potential use in products to be manufactured in the future. Management was required to make judgments about the future benefit of our raw materials. Actual reserve requirements could differ significantly from management’s estimates, which could have a significant unfavorable impact on our future gross margins.
Investigations of the calculation of average wholesale prices may adversely affect our business.
Many government and third-party payors, including Medicare, Medicaid, HMOs and managed care organizations (“MCOs”) reimburse doctors and others for the purchase of certain prescription drugs based on a drug’s AWP. In the past several years, state and federal government agencies have conducted ongoing investigations of manufacturers’ reporting practices with respect to AWP, in which they have asserted that reporting of inflated AWPs have led to excessive payments for prescription drugs.
The regulations regarding reporting and payment obligations with respect to Medicare and/or Medicaid reimbursement and rebates and other governmental programs are complex. We and other pharmaceutical companies are defendants in a number of suits filed by state attorneys general and have been notified of an investigation by the United States Department of Justice with respect to Medicaid reimbursement and rebates. While we cannot predict the outcome of the investigation, possible remedies that the U.S. government could seek include treble damages, civil monetary penalties, debarment from contracting with the government and exclusion from the Medicare and Medicaid programs. In connection with such an investigation, the U.S. government may also seek a Corporate Integrity Agreement (administered by HHS OIG) with us which could include ongoing compliance and reporting obligations. Because our processes for these calculations and the judgments involved in making these calculations involve, and will continue to involve, subjective decisions and complex methodologies, these calculations are subject to the risk of errors. In addition, they are subject to review and challenge by the applicable governmental agencies, and it is possible that such reviews could result in material changes. Further, effective October 1, 2007, the CMS adopted new rules for AMP based on the provisions of the Deficit Reduction Act of 2005 (the “DRA”). While the matter remains subject to litigation and proposed legislation, one potential significant change as a result of the DRA is that AMP would need to be disclosed to the public. AMP was historically kept confidential by the government and participants in the Medicaid program. Disclosing AMP to competitors, customers, and the public at large could negatively affect our leverage in commercial price negotiations.
In addition, as also disclosed herein, a number of state and federal government agencies are conducting investigations of manufacturers’ reporting practices with respect to AWPs in which they have suggested that reporting of inflated AWP has led to excessive payments for prescription drugs. We and numerous other pharmaceutical companies have been named as defendants in various actions relating to pharmaceutical pricing issues and whether allegedly improper actions by pharmaceutical manufacturers led to excessive payments by Medicare and/or Medicaid.
Any governmental agencies that have commenced, or may commence, an investigation of our Company could impose, based on a claim of violation of fraud and false claims laws or otherwise, civil and/or criminal sanctions, including fines, penalties, debarment from contracting with the government and possible exclusion from federal health care programs including Medicare and/or Medicaid. Some of the applicable laws may impose
58
Table of Contents
liability even in the absence of specific intent to defraud. Furthermore, should there be ambiguity with regard to how to properly calculate and report payments—and even in the absence of any such ambiguity—a governmental authority may take a position contrary to a position we have taken, and may impose civil and/or criminal sanctions. Any such penalties or sanctions could have a material adverse effect on our business, financial position and results of operations.
We have material weaknesses in our internal control over financial reporting and cannot assure you that additional material weaknesses will not be identified in the future. If we fail to maintain an effective system of internal controls or discover material weaknesses in our internal control over financial reporting, we may not be able to report our financial results accurately or timely or detect fraud, which could have a material adverse effect on our business.
Section 404 of the Sarbanes-Oxley Act of 2002 requires us to evaluate the effectiveness of our internal control over financial reporting as of the end of each year, and to include a management report assessing the effectiveness of our internal control over financial reporting in each Report. Section 404 also requires our independent registered public accounting firm to attest to, and report on, the effectiveness of our internal control over financial reporting.
Based upon our evaluation of internal control over financial reporting as of March 31, 2011, we determined that certain material weaknesses as defined by Rule 12b-2 under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), existed at our Company. Specifically, we determined that we had one material weakness in our internal control environment or entity-level controls and three material weaknesses surrounding financial statement presentation and review procedures and application of accounting principles.
Management is in the process of implementing steps to remediate these material weaknesses. However, we cannot provide assurances that such remediation will be effective. See Part II Item 9A-“Controls and Procedures” included in the Report for additional information regarding our internal control over financial reporting.
Our internal control over financial reporting may not prevent all errors and all fraud. A control system, no matter how well designed and operated, can provide only reasonable, not absolute, assurance that the control system’s objectives will be met. Further, the design of a control system must reflect the fact that there are resource constraints, and the benefits of controls must be considered relative to their costs. Controls can be circumvented by the individual acts of some persons, by collusion of two or more people, or by management override of the controls. Over time, controls may become inadequate because changes in conditions or deterioration in the degree of compliance with policies or procedures may occur. Because of the inherent limitations in a cost-effective control system, misstatements due to error or fraud may occur and not be detected.
Also, additional material weaknesses in our internal control over financial reporting may be identified in the future. Any failure to maintain or implement required new or improved controls, or any difficulties we encounter in their implementation, could result in material weaknesses, cause us to fail to timely meet our periodic reporting obligations, or result in material misstatements in our financial statements. Any such failure could also adversely affect the results of periodic management evaluations and annual auditor attestation reports regarding the effectiveness of our internal control over financial reporting required under Section 404 of the Sarbanes-Oxley Act of 2002 and the rules promulgated thereunder. In addition, solely as a result of the material weaknesses, we have determined that our disclosure controls and procedures were not effective as of March 31, 2011. If our internal control over financial reporting or disclosure controls and procedures are not effective, there may be errors in our financial statements that could require a restatement or our filings may not be timely and investors may lose confidence in our reported financial information, which could lead to a decline in our stock price.
59
Table of Contents
A failure to remain in compliance with one or more of the requirements of an outstanding mortgage loan and a related waiver could have a material adverse effect on our business, financial condition and cash flows.
In March 2006, as previously disclosed, we entered into a $43.0 million mortgage loan arrangement, of which approximately $33.0 million remains outstanding as of March 31, 2011. Also, as previously disclosed, we obtained a waiver with respect to certain requirements of the mortgage loan documentation. Failure by us to comply with the terms of the mortgage or the waiver from the lender could result in, among other things, our outstanding obligations with respect to the mortgage loan accelerating and immediately becoming due and payable and resulting in cross-defaults under our convertible notes described below and other debt obligations, which would materially adversely affect our business, financial condition and cash flows.
Pursuant to the indentures governing our convertible notes and our 2015 Notes, our default in the payment of other indebtedness or any final non-appealable judgments could result in such notes becoming immediately due and payable, which could have a material adverse effect on our business, financial condition and cash flows.
In May 2003, we issued $200.0 million principal amount of 2.5% Contingent Convertible Subordinated Notes which mature in 2033 (the “2033 Notes”). We are current in all our financial payment obligations under the indenture governing the 2033 Notes. However, a failure by us or any of our subsidiaries to pay any indebtedness or any final non-appealable judgments in excess of $0.75 million constitutes an event of default under the indenture. An event of default would permit the trustee under the indenture or the holders of at least 25% of the 2033 Notes to declare all amounts owing to be immediately due and payable and exercise other remedies, which would materially adversely affect our business, financial condition and cash flows, as well as our ability to continue as a going concern. In addition, if an event of default under the indenture was to be triggered and the 2033 Notes repaid, we could recognize cancellation of indebtedness income. Such cancellation of indebtedness income would result in a tax liability to the extent not reduced by our tax attributes. Additionally, a default in payment of other indebtedness in the amount of $5 million or more, including an event of default with respect to the 2033 Notes, would trigger a default under the indenture governing the 2015 Notes, which would materially adversely affect our business, financial condition and cash flows, as well as our ability to continue as a going concern.
The indenture governing the 2015 Notes contains operating and financial restrictions which may hinder our ability to pursue business opportunities and operate our business.
The indenture governing the 2015 Notes contains significant operating and financial restrictions that may prevent us from pursuing certain business opportunities and restrict our ability to operate our business. These restrictions will limit or prohibit, among other things, our ability to: (1) incur additional indebtedness or issue certain preferred shares; (2) pay dividends, redeem subordinated debt or make other restricted payments on capital stock; (3) designate our subsidiaries as Unrestricted Subsidiaries (as defined in the indenture); (4) change our line of business; (5) transfer or sell assets, including the capital stock of our subsidiaries; (6) make certain investments or acquisitions; (7) grant liens on our assets; (8) incur dividend or other payment restrictions affecting certain subsidiaries; and (9) merge, consolidate or transfer substantially all of our assets. See Note 13—“Long-Term Debt” of the Notes to Consolidated Financial Statements included in this Report for a further description of the 2015 Notes.
Increased indebtedness may impact our financial condition and results of operations.
At March 31, 2011, we had an aggregate of $458.0 million of outstanding debt, excluding collateralized borrowings, consisting primarily of $200.0 million principal amount of the 2033 Notes, the remaining principal amount of $33.0 million mortgage loan, and $225.0 million principal amount of 2015 Notes. On March 17, 2011, the Company issued the 2015 Notes. The Company used a portion of the proceeds obtained from the issuance of
60
Table of Contents
the 2015 Notes to repay in full its existing obligations under the Bridge Loan with U.S. Healthcare of approximately $61 million (which amount included an applicable make-whole premium). Our level of indebtedness may have several important effects on our future operations, including:
| • | we will be required to use a portion of our cash flow from operations or otherwise for the payment of any principal or interest due on our outstanding indebtedness; |
| • | our outstanding indebtedness and leverage will increase the impact of negative changes in general economic and industry conditions, as well as competitive pressures and increases in interest rates; and |
| • | the level of our outstanding debt and the impact it has on our ability to meet debt covenants may affect our ability to obtain additional financing for working capital, capital expenditures, acquisitions or general corporate purposes. |
Even if and after we are able to resume manufacturing and shipment of our products, general economic conditions, industry cycles and financial, business and other factors affecting our operations, many of which are beyond our control, may affect our future performance. As a result, our business might not continue to generate cash flow at or above historic levels. If we cannot generate sufficient cash flow from operations in the future to service our debt, we may, among other things:
| • | seek additional financing in the debt or equity markets; |
| • | refinance or restructure all or a portion of our indebtedness; |
| • | sell selected assets; |
| • | reduce or delay planned capital expenditures; or |
| • | reduce or delay planned research and development expenditures. |
These measures might not be sufficient to enable us to service our debt. In addition, any financing, refinancing or sale of assets might not be available on economically favorable terms or at all.
Holders of the 2033 Notes may require us to offer to repurchase their 2033 Notes for cash upon the occurrence of a change in control or on May 16, 2013, 2018, 2023 and 2028. The source of funds for any repurchase of the 2033 Notes required as a result of any such events will be our available cash or cash generated from operating activities or other sources, including borrowings, sales of assets, sales of equity or funds provided by a new controlling entity. The use of available cash to fund the repurchase of the 2033 Notes may impair our ability to obtain additional financing in the future. Any such repayment is a restricted payment under the 2015 Notes and is not permitted unless the 2015 Notes are refinanced with other indebtedness or repaid.
Our cost-reducing measures could yield unintended consequences, which could have a material adverse effect on our business, financial condition, results of operations and cash flows.
As previously disclosed, we implemented significant cost savings measures to mitigate the financial impact of our actions to recall all of the products we manufactured and to suspend manufacturing and shipment of our products pending compliance with the terms of the consent decree. These measures included a substantial reduction of our workforce, which commenced on February 5, 2009, and an ongoing realignment of our cost structure. We realigned and restructured the sales force for our Ther-Rx subsidiary, and our production workforce. As a result, we reduced our employee headcount from approximately 852 on December 31, 2008 to approximately 317 on March 31, 2010. On March 31, 2010, we implemented a plan to further reduce our employee workforce from 317 to 237. In February 2011, we announced that we would increase our workforce by approximately 95 individuals to support the launch and marketing of Makena™.
The cost-reducing measures taken by us could yield unintended consequences, such as distraction of our management and employees, the inability to retain and attract new employees, business disruption, a negative impact on morale among remaining employees, attrition beyond our planned reduction in workforce and reduced
61
Table of Contents
employee productivity, any of which could have a material adverse effect on our business, financial condition, results of operations and cash flows. In addition, our reductions in personnel may subject us to risks of litigation, which could result in substantial cost. We cannot guarantee that the cost reduction measures, or other measures we may take in the future, will result in the expected cost savings, or that any cost savings will be unaccompanied by these or other unintended consequences.
Our future growth will also depend upon our ability to develop, acquire, fund and successfully launch new products in addition to Makena™.
In the near term, we are focused on meeting the requirements of the consent decree, which will allow more of our approved products to be reintroduced to the market and continuing the commercial launch of Makena™. We also need to continue to develop and commercialize new brand name products and generic products utilizing our proprietary drug delivery systems to grow our business in the future. To do this, we will need to identify, develop and commercialize technologically enhanced branded products and drugs that are off-patent or where we can challenge the patents and that can be produced and sold by us as generic products using our drug delivery technologies. If we are unable to identify, develop and commercialize new products, we may need to obtain licenses to additional rights to branded or generic products, assuming they would be available for licensing, which could decrease our profitability. We may not be successful in pursuing this strategy.
Prior to entering into the consent decree we had filed ANDAs with the FDA seeking permission to market generic versions of several branded products, including varying strengths of the following:
| • | Metadate CD® (methylphenidate hydrochloride) Extended-Release Capsules |
| • | Ritalin LA® (methylphenidate hydrochloride) Extended-Release Capsules |
| • | Focalin XR® (dexmethylphenidate hydrochloride) Extended-Release Capsules |
| • | Keppra XR® (levetiracetam) Extended-Release Tablets |
With respect to the first three ANDA submissions noted above, we have sought favorable court rulings in patent infringement actions filed against us by the sponsors of the branded products. See Note 15— “Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report for more information regarding patent certifications and litigation. However, in such litigations generally, we cannot be certain of the outcome, and a decision that a relevant patent is valid and infringed likely could significantly delay our ability to market any of the drugs at issue in such lawsuits. In addition, the litigation process itself can impose significant delays and expenses. On March 1, 2010, we entered into a settlement agreement to settle the patent infringement actions with respect to Ritalin LA® and Focalin XR® and on April 2, 2010, we entered into a settlement agreement to settle the patent infringement action with respect to Metadate CD®. See Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report for information regarding the settlement. Due to the consent decree, an approval or a tentative approval was not obtained in the required time frame for any of the Company’s Paragraph IV ANDA filings. Therefore, the 180 days of exclusivity under the Drug Price Competition and Patent Term Restoration Act of 1984, known as the Hatch-Waxman Act, was lost.
We depend on our patents and other proprietary rights and cannot be certain of their confidentiality and protection.
Our success depends, in large part, on our ability to protect our current and future technologies and products, to defend our intellectual property rights and to avoid infringing on the proprietary rights of others. Although the pharmaceutical industry is crowded, we have been issued a substantial number of patents in the U.S. and in many foreign countries, which cover certain of our technologies, and have filed, and expect to continue to file, patent applications seeking to protect newly developed technologies and products. The patent position of pharmaceutical companies can be highly uncertain and frequently involves complex legal and factual
62
Table of Contents
questions. As a result, the breadth of claims allowed in patents relating to pharmaceutical applications or their validity and enforceability cannot accurately be predicted. Patents are examined for patentability at patent offices against bodies of prior art which by their nature may be incomplete and imperfectly categorized. Therefore, even presuming that the patent examiner has been able to identify and cite the best prior art available to him during the examination process, any patent issued to us could later be found by a court or a patent office during post-issuance proceedings to be invalid in view of newly-discovered prior art or already considered prior art or other legal reasons. Furthermore, there are categories of “secret” prior art unavailable to any examiner, such as the prior inventive activities of others, which could form the basis for invalidating any patent. In addition, there are other reasons why a patent may be found to be invalid, such as an offer for sale or public use of the patented invention in the U.S. more than one year before the filing date of the patent application. Moreover, a patent may be deemed unenforceable if, for example, the inventor or the inventor’s agents failed to disclose prior art to the USPTO that they knew was material to patentability.
The coverage claimed in a patent application can be significantly altered during the examination process either in the U.S. or abroad. Consequently, our pending or future patent applications may not result in the issuance of patents or may result in issued patents having claims significantly different from that of the patent application as originally filed. Patents issued to us may be subjected to further proceedings limiting their scope and may not provide significant proprietary protection or competitive advantage. Our patents also may be challenged, circumvented, invalidated or deemed unenforceable. Patent applications in the U.S. filed prior to November 29, 2000 are currently maintained in secrecy until and unless patents issue, and patent applications in certain other countries generally are not published until more than 18 months after they are first filed (which generally is the case in the U.S. for applications filed on or after November 29, 2000). In addition, publication of discoveries in scientific or patent literature often lags behind actual discoveries. As a result, we cannot be certain that we or our licensors will be entitled to any rights in purported inventions claimed in pending or future patent applications or that we or our licensors were the first to file patent applications on such inventions. Furthermore, patents already issued to us or our pending applications may become subject to dispute, and any dispute could be resolved against us. For example, we may become involved in re-examination, reissue or interference proceedings in the USPTO, or opposition proceedings in a foreign country. The result of these proceedings can be the invalidation or substantial narrowing of our patent claims. We also could be subject to court proceedings that could find our patents invalid or unenforceable or could substantially narrow the scope of our patent claims. Even where we ultimately prevail before the USPTO or in litigation, the expense of these proceedings can be considerable. In addition, statutory differences in patentable subject matter may limit the protection we can obtain on some of our inventions outside of the U.S. For example, methods of treating humans are not patentable in many countries outside of the U.S.
These and other issues may prevent us from obtaining patent protection outside of the U.S. Furthermore, once patented in foreign countries, the inventions may be subjected to mandatory working requirements and/or subject to compulsory licensing regulations.
We also rely on trade secrets, unpatented proprietary know-how and continuing technological innovation that we seek to protect, in part by confidentiality agreements with licensees, suppliers, employees and consultants. These agreements may be breached by the other parties to these agreements. We may not have adequate remedies for any breach. Disputes may arise concerning the ownership of intellectual property or the applicability or enforceability of our confidentiality agreements and there can be no assurance that any such disputes would be resolved in our favor.
In addition, our trade secrets and proprietary technology may become known or be independently developed by our competitors, or patents may not be issued with respect to products or methods arising from our research, and we may not be able to maintain the confidentiality of information relating to those products or methods. Furthermore, certain unpatented technology may be subject to intervening rights.
63
Table of Contents
We depend on our trademarks and related rights.
To protect our trademarks and associated goodwill, domain name, and related rights, we generally rely on federal and state trademark and unfair competition laws, which are subject to change. Some, but not all, of our trademarks are registered in the jurisdictions where they are used. Some of our other trademarks are the subject of pending applications in the jurisdictions where they are used or intended to be used, and others are not.
It is possible that third parties may own or could acquire rights in trademarks or domain names in the U.S. or abroad that are confusingly similar to or otherwise compete unfairly with our marks and domain names, or that our use of trademarks or domain names may infringe or otherwise violate the intellectual property rights of third parties. The use of similar marks or domain names by third parties could decrease the value of our trademarks or domain names and hurt our business, for which there may be no adequate remedy.
Third parties may claim that we infringe on their proprietary rights, or seek to circumvent ours.
We have been sued in the past for, and may in the future be required to defend against charges of infringement of patents, trademarks or other proprietary rights of third parties. Such defenses could require us to incur substantial expense and to divert significant effort of our technical and management personnel, and could result in our loss of rights to develop or make certain products or require us to pay monetary damages or royalties to license proprietary rights from third parties. More generally, the outcome of intellectual property litigation and disputes is uncertain and presents a risk to our business.
If an intellectual property dispute is settled through licensing or similar arrangements, costs associated with such arrangements may be substantial and could include ongoing royalties. Furthermore, we cannot be certain that the necessary licenses would be available to us on acceptable terms, if at all. Accordingly, an adverse determination in a judicial or administrative proceeding or failure to obtain necessary licenses could prevent us from manufacturing, using, selling and/or importing into the U.S. certain of our products, and therefore could have a material adverse effect on our business or results of operations. Litigation also may be necessary to enforce our patents against others or to protect our know-how or trade secrets. That litigation could result in substantial expense or put our proprietary rights at risk of loss, and we cannot assure you that any litigation will be resolved in our favor. As noted above, certain patent infringement lawsuits are currently pending against us, any of which could have a material adverse effect on our future business, financial condition, results of operations or cash flows.
Our dependence on key executives and qualified scientific, technical and managerial personnel could affect the development and management of our business.
We are highly dependent upon our ability to attract and retain qualified scientific, technical and managerial personnel. Our recent reductions in our employee base have increased this dependence. There is intense competition for qualified personnel in the pharmaceutical and biotechnology industries, and we cannot be sure that we will be able to continue to attract and retain qualified personnel necessary for the development and management of our business. Although we do not believe the loss of one individual would materially harm our business, our business might be harmed by the loss of services of multiple existing personnel, as well as the failure to recruit additional key scientific, technical and managerial personnel in a timely manner. Much of the know-how we have developed resides in our scientific and technical personnel and is not readily transferable to other personnel. While we have employment agreements with certain of our key executives, we do not ordinarily enter into employment agreements (other than agreements related to confidentiality and proprietary rights) with our other scientific, technical and managerial employees.
We may be adversely affected by the continuing consolidation of our distribution network and the concentration of our customer base.
Our principal customers are wholesale drug distributors, major retail drug store chains, independent pharmacies and mail order firms. These customers comprise a significant part of the distribution network for
64
Table of Contents
pharmaceutical products in the U.S. This distribution network is continuing to undergo significant consolidation marked by mergers and acquisitions among wholesale distributors and the growth of large retail drug store chains. As a result, a small number of large wholesale distributors control a significant share of the market, and the number of independent drug stores and small drug store chains has decreased. We expect that consolidation of drug wholesalers and retailers will increase pricing and other competitive pressures on drug manufacturers. For the three and twelve months ended March 31, 2011, our three largest customers, which are specialty pharmacies and distributors, accounted for 29.7%, 21.5% and 14.5% and 17.3%, 16.6% and 11.2% of our gross revenues, respectively. The loss of any of these customers could materially and adversely affect our business, financial condition, results of operations or cash flows.
We depend on licenses from others, and any loss of these licenses could harm our business, market share and profitability.
We have acquired the rights to manufacture, use and/or market certain products through license agreements. We also expect to continue to obtain licenses for other products and technologies in the future. Our license agreements generally require us to develop the markets for the licensed products. If we do not develop these markets, the licensors may be entitled to terminate these license agreements.
We cannot be certain that we will fulfill all of our obligations under any particular license agreement for any variety of reasons, including lack of sufficient liquidity to fund our obligations, insufficient resources to adequately develop and market a product, lack of market development despite our efforts and lack of product acceptance. Our failure to fulfill our obligations could result in the loss of our rights under a license agreement.
Certain products we have the right to license are at certain stages of clinical tests and FDA approval. Failure of any licensed product to receive regulatory approval could result in the loss of our rights under its license agreement.
We expend a significant amount of resources on research and development efforts that may not lead to successful product introductions.
We conduct research and development primarily to enable us to manufacture and market FDA-approved pharmaceuticals in accordance with FDA regulations. Typically, research costs related to the development of innovative compounds and the filing of NDAs are significantly greater than those expenses associated with ANDA filings. Because of the inherent risk associated with research and development efforts in our industry, particularly with respect to new drugs, our research and development expenditures may not result in the successful introduction of FDA-approved new pharmaceutical products. Also, after we submit an application for approval, the FDA may request that we conduct additional studies and as a result, we may be unable to reasonably determine the total research and development costs to develop a particular product. Finally, we cannot be certain that any investment made in developing products will be recovered, even if we are successful in commercialization. In addition, as set forth in the risk factor entitled “Our future growth will largely depend upon our ability to fund and develop new products” above, although the consent decree does not specifically address these ANDA submissions, we anticipate that final approval of any ANDAs will not occur before the FDA has determined that we are compliant with cGMP. In September 2010, the FDA determined that our facilities were in compliance, allowing us to resume shipment of our Potassium Chloride Extended Release Capsule products, which commenced sales in that month. Additional products are in the process of being brought back to market although the Company does not expect to resume shipping other products until later in fiscal year 2012 at the earliest.
We may not be able to obtain necessary regulatory clearances or approvals on a timely basis, if at all, for any of our products under development, and delays in receipt or failure to receive such clearances or approvals, the loss of previously received clearances or approvals, or failure to comply with existing or future regulatory requirements could have a material adverse effect on our business. To the extent that we expend significant
65
Table of Contents
resources on research and development efforts and are not able, ultimately, to introduce successful new products as a result of those efforts, our business, financial condition, results of operations or cash flows may be materially adversely affected.
Any significant interruption in the supply of raw materials or finished product could have a material adverse effect on our business.
We typically purchase the active pharmaceutical ingredient (i.e., the chemical compounds that produce the desired therapeutic effect in our products) and other materials and supplies that we use in our manufacturing operations, as well as certain finished products (including Evamist® and Makena™), from many different domestic and foreign suppliers.
We also maintain safety stocks in our raw materials inventory, and in certain cases where we have listed only one supplier in our applications with the FDA, have received FDA approval to use alternative suppliers should the need arise. However, there is no guarantee that we will always have timely and sufficient access to a critical raw material or finished product, or access to such materials or products on commercially reasonable terms. A prolonged interruption in the supply of a single-sourced raw material, including the active ingredient, or finished product could cause our business, financial condition, results of operations or cash flows to be materially adversely affected. In addition, our manufacturing capabilities could be impacted by quality deficiencies in the products which our suppliers provide, which could have a material adverse effect on our business.
Where we purchase finished products for sale, it is possible for the ability or willingness of our suppliers to supply us to be disrupted, delayed or terminated, including as a result of regulatory actions by the FDA or other government agencies, including shipping halts, product seizures and recalls affecting such suppliers, or as a result of labor stoppages, facility damage or casualties, or other sources of interruption. Such interruptions could have a material adverse effect on our business.
We utilize controlled substances in certain of our current products and products in development and therefore must meet the requirements of the Controlled Substances Act of 1970 (the “CSA”) and the related regulations administered by the DEA. These regulations relate to the manufacture, shipment, storage, sale and use of controlled substances. The DEA limits the availability of the active ingredients used in certain of our current products and products in development and, as a result, our procurement quota of these active ingredients may not be sufficient to meet commercial demand or complete clinical trials. We must annually apply to the DEA for procurement quota in order to obtain these substances. Any delay or refusal by the DEA in establishing our procurement quota for controlled substances could delay or stop our clinical trials or product launches, or could cause trade inventory disruptions for those products that have already been launched, which could have a material adverse effect on our business, financial condition, results of operations or cash flows.
Our revenues, gross profit and operating results may fluctuate from period to period, depending upon our product sales mix, our product pricing, and our costs to manufacture or purchase products.
Assuming we are able to comply with the requirements under the consent decree and resume the manufacture and distribution of more of our approved products, our future results of operations, financial condition and cash flows will depend to a significant extent upon our product sales mix (the proportion of total sales among products). Factors that may cause our sales mix to vary include:
| • | the number and timing of new product introductions; |
| • | marketing exclusivity on products, if any, which may be obtained; |
| • | the level of competition in the marketplace with respect to certain products; |
| • | the availability of raw materials and finished products from our suppliers; |
| • | the buying patterns of our three largest wholesaler customers; |
66
Table of Contents
| • | the scope and outcome of governmental regulatory action that may involve us; |
| • | periodic dependence on a relatively small number of products for a significant portion of net revenue or income; and |
| • | legal actions brought by our competitors. |
The profitability of our product sales is also dependent upon the prices we are able to charge for our products, the costs to purchase products from third parties, and our ability to manufacture our products in a cost-effective manner. If our revenues and gross profit decline or do not grow as anticipated, we may not be able to correspondingly reduce our operating expenses.
Rising insurance costs could negatively impact profitability.
The cost of insurance, including workers’ compensation, product liability and general liability insurance, has risen significantly in the past few years and may continue to increase. In response, we may increase deductibles and/or decrease certain coverages to mitigate these costs. These increases, and our increased risk due to increased deductibles and reduced coverages, could have a negative impact on our business, financial condition, results of operations or cash flows.
We may continue to incur charges for impairment of intangible and other long-lived assets.
When we acquire the rights to manufacture and sell a product, we record the aggregate purchase price, along with the value of the product-related liabilities we assume, as intangible assets. We use the assistance of valuation experts to help us allocate the purchase price to the fair value of the various intangible assets we have acquired. Then, we must estimate the economic useful life of each of these intangible assets in order to amortize their cost as an expense in our consolidated statements of operations over the estimated economic useful life of the related asset. The factors that affect the actual economic useful life of a pharmaceutical product are inherently uncertain, and include patent protection, physician loyalty and prescribing patterns, competition by products prescribed for similar indications, future introductions of competing products not yet FDA-approved and the impact of promotional efforts, among many others. We consider all of these factors in initially estimating the economic useful lives of our products, and we also continuously monitor these factors for indications of decline in carrying value.
In assessing the recoverability of our intangible and other long-lived assets, we must make assumptions regarding estimated undiscounted future cash flows and other factors. If the estimated undiscounted future cash flows do not exceed the carrying value of the intangible or other long-lived assets being evaluated, we must determine the fair value of the intangible or other long-lived assets. If the fair value of these assets is less than its carrying value, an impairment loss will be recognized in an amount equal to the difference. If these estimates or their related assumptions change in the future, we may be required to record impairment charges for these assets. We review intangible assets for impairment at least annually and all long-lived and intangible assets whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. If we determine that an intangible or long-lived asset is impaired, a non-cash impairment charge would be recognized.
Because circumstances can change, the value of intangible and long-lived assets we record may not be realized by us. If we determine that impairment has occurred, we would be required to write-off the impaired portion of the unamortized assets, which could have a material adverse effect on our results of operations in the period in which the write-off occurs. In addition, in the event of a sale of any of our assets, we might not recover our recorded value of the associated assets.
In connection with the steps taken by us with respect to the nationwide recall and suspension of shipment of all products manufactured by us, the requirements under the consent decree, the ongoing private litigation and governmental inquiries, and certain other events in the fourth quarter of fiscal year 2011 and 2010, we completed an evaluation of each of our intangible assets, and as a result of these evaluations, recognized certain impairment charges.
67
Table of Contents
There are inherent uncertainties involved in the estimates, judgments and assumptions used in the preparation of our financial statements, and any changes in those estimates, judgments and assumptions could have a material adverse effect on our financial condition and results of operations.
The consolidated financial statements that we file with the SEC are prepared in accordance with U.S. generally accepted accounting principles (“U.S. GAAP”). The preparation of financial statements in accordance with U.S. GAAP involves making estimates and judgments that affect the reported amounts of assets, liabilities, revenues and expenses, and the related disclosure of contingent assets and liabilities. A summary of our significant accounting practices is included in Note 2—“Summary of Significant Accounting Policies” of the Notes to Consolidated Financial Statements included in this Report. The most significant estimates we are required to make under U.S. GAAP include, but are not limited to, those related to revenue recognition and reductions to gross revenues, inventory valuation, intangible asset valuations, property and equipment valuations, stock-based compensation, income taxes and loss contingencies related to legal proceedings. We periodically evaluate estimates used in the preparation of the consolidated financial statements for reasonableness, including estimates provided by third parties. Appropriate adjustments to the estimates will be made prospectively, as necessary, based on such periodic evaluations. We base our estimates on, among other things, currently available information, market conditions, historical experience and various assumptions, which together form the basis of making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Although we believe that our assumptions are reasonable under the circumstances, estimates would differ if different assumptions were utilized and these estimates may prove in the future to have been inaccurate.
Enactment of the Patient Protection and Affordable Care Act (the “Affordable Care Act”), legislative proposals, reimbursement policies of third parties, cost-containment measures and health care reform could affect the marketing, pricing and demand for our products.
The enactment of the Affordable Care Act on March 23, 2010, as well as various additional legislative proposals, including proposals relating to prescription drug benefits, could materially impact the pricing and sale of our products. Further, reimbursement policies of third parties may affect the marketing of our products. Our ability to market our products will depend in part on reimbursement levels for the cost of the products and related treatment established by health care providers, including government authorities, private health insurers and other organizations, such as HMOs and MCOs. Insurance companies, HMOs, MCOs, Medicaid and Medicare administrators and others regularly challenge the pricing of pharmaceutical products and review their reimbursement practices. In addition, the following factors could significantly influence the purchase of pharmaceutical products, which could result in lower prices and a reduced demand for our products:
| • | the trend toward managed health care in the U.S.; |
| • | the growth of organizations such as HMOs and MCOs; |
| • | legislative proposals to reform health care and government insurance programs; and |
| • | price controls and non-reimbursement of new and highly priced medicines for which the economic therapeutic rationales are not established. |
The Affordable Care Act is a comprehensive and very complex and far-reaching statute. The cost-containment measures and health care reforms in the Affordable Care Act and in other legislative proposals could affect our ability to sell our products in many possible ways. The Obama administration’s fiscal year 2010 budget included proposals to reduce Medicare and Medicaid spending and reduce drug spending. We are unable to predict the ultimate impact of the Affordable Care Act, or the content or timing of any future healthcare reform legislation and its impact, on us. Those reforms may have a material adverse effect on our financial condition and results of operations.
The reimbursement status of a newly approved pharmaceutical product may be uncertain. Reimbursement policies and decisions, either generally affecting all pharmaceutical companies or specifically affecting us, may not include some of our products or government agencies or third parties may assert that certain of our products
68
Table of Contents
are not eligible for Medicaid, Medicare or other reimbursement and were not so eligible in the past, possibly resulting in demands for damages or refunds. Even if reimbursement policies of third parties grant reimbursement status for a product, we cannot be sure that these reimbursement policies will remain in effect. Limits on reimbursement could reduce the demand for our products. The unavailability or inadequacy of third-party reimbursement for our products could reduce or possibly eliminate demand for our products. We are unable to predict whether governmental authorities will enact additional legislation or regulation which will affect third-party coverage and reimbursement that reduces demand for our products.
Our ability to market generic pharmaceutical products successfully depends, in part, on the acceptance of the products by independent third parties, including pharmacies, government formularies and other retailers, as well as patients. In the past, we have manufactured, and assuming we comply with the requirements under the consent decree we anticipate that in the future we will again manufacture, a number of prescription drugs that are used by patients with severe health conditions. Although the brand-name products generally have been marketed safely for many years prior to our introduction of a generic/non-branded alternative, there is a possibility that one of these products could produce a side effect that could result in an adverse effect on our ability to achieve acceptance by managed care providers, pharmacies and other retailers, customers and patients. If these independent third parties do not accept our products, it could have a material adverse effect on our business, financial condition, results of operations or cash flows.
Extensive industry regulation has had, and will continue to have, a significant impact on our industry and our business, especially our product development, manufacturing and distribution capabilities.
All pharmaceutical companies, including us, are subject to extensive, complex, costly and evolving regulation by the federal government, principally the FDA and, to a lesser extent, the DEA and state government agencies. The Federal Food, Drug, and Cosmetic Act (the “FDCA”), the CSA and other federal statutes and regulations govern or influence the testing, manufacturing, packing, labeling, storing, record keeping, safety, approval, advertising, promotion, sale and distribution of our products. Failure to comply with applicable FDA or other regulatory requirements may result in criminal prosecution, civil penalties, injunctions or holds, recall or seizure of products and total or partial suspension of production, as well as other regulatory actions against our products and us.
In addition to compliance with cGMP requirements, drug manufacturers must register each manufacturing facility with the FDA. Manufacturers and distributors of prescription drug products are also required to be registered in the states where they are located and in certain states that require registration by out-of-state manufacturers and distributors. Manufacturers also must be registered with the DEA and similar applicable state and local regulatory authorities if they handle controlled substances, and with the EPA and similar state and local regulatory authorities if they generate toxic or dangerous wastes, and must also comply with other applicable DEA and EPA requirements.
From time to time, governmental agencies have conducted investigations of pharmaceutical companies relating to the distribution and sale of drug products to government purchasers or subject to government or third-party reimbursement. However, standards sought to be applied in the course of governmental investigations can be complex and may not be consistent with standards previously applied to our industry generally or previously understood by us to be applicable to our activities.
The process for obtaining governmental approval to manufacture and market pharmaceutical products is rigorous, time-consuming and costly, and we cannot predict the extent to which we may be affected by legislative and regulatory developments. We are dependent on receiving FDA and other governmental or third-party approvals prior to manufacturing, marketing and shipping many of our products. Consequently, we cannot predict whether we will obtain FDA or other necessary approvals or whether the rate, timing and cost of such approvals will adversely affect our product introduction plans or results of operations.
69
Table of Contents
We are subject to various federal and state laws pertaining to health care fraud and abuse, including anti-kickback and false claims laws.
Several types of state and federal laws, including anti-kickback and false claims statutes, have been applied to restrict certain marketing practices in the pharmaceutical industry in recent years. See Part I, Item 1—“Business—(d) Narrative Description of Business—Government Regulation—New Product Development and Approval—Anti-Kickback and False Claims Statutes” for more information.
We endeavor to comply with the applicable fraud and abuse laws and to operate within related statutory exemptions and regulatory safe harbors protecting certain common activities from prosecution. However, the exemptions and safe harbors are drawn narrowly, and practices that involve remuneration to individuals or entities in a position to prescribe, purchase, or recommend our products may be subject to scrutiny if they do not qualify for an exemption or safe harbor.
Violations of fraud and abuse laws may be punishable by civil and/or criminal sanctions, including substantial fines and civil monetary penalties, debarment from contracting with the government, as well as the possibility of exclusion from federal and state health care programs, including Medicaid, Medicare and Veterans Administration health programs. Furthermore, the laws applicable to us are broad in scope and are subject to evolving interpretations and permit governmental authorities to exercise significant discretion. Any determination by a governmental authority that we are not in compliance with applicable laws and regulations could have a material adverse effect on our reputation, business operations and financial results.
Our indemnification obligations and limitations of our director and officer liability insurance may have a material adverse effect on our business, financial condition, results of operations and cash flows.
Under Delaware law, our Amended Certificate of Incorporation and By-Laws and certain indemnification agreements to which we are a party, we have an obligation to indemnify, or we have otherwise agreed to indemnify, certain of our current and former directors, officers and associates with respect to current and future inquiries, investigations and litigation (see Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report). In connection with some of these pending matters, we are required to, or we have otherwise agreed to, advance, and have advanced, significant legal fees and related expenses to several of our current and former directors, officers and associates and expect to continue to do so while these matters are pending. Certain of these obligations may not be “covered matters” under our directors’ and officers’ liability insurance, or there may be insufficient coverage available. Further, in the event the directors, officers and associates are ultimately determined to not be entitled to indemnification, we may not be able to recover the amounts we previously advanced to them.
In addition, we have incurred significant expenses in connection with the pending inquiries, investigations and litigation. We maintain directors’ and officers’ liability insurance for non-indemnifiable securities claims and have met the retention limits under these policies with respect to these pending matters. We cannot provide any assurances that pending claims, or claims yet to arise, will not exceed the limits of our insurance policies, that such claims are covered by the terms of our insurance policies or that our insurance carrier will be able to cover our claims. Due to these insurance coverage limitations, we may incur significant unreimbursed costs to satisfy our indemnification and other obligations, which may have a material adverse effect on our financial condition, results of operations and cash flows.
On March 22, 2011, Mr. M. Hermelin made a demand on the Company for indemnification with respect to his payment of $1.9 million imposed by the United States District Court as a fine and forfeiture of pecuniary gain as part of the sentence resulting from his guilty plea entered by the Court on March 10, 2011. The Company has paid or will pay as an advancement, approximately $5.0 million to Mr. M. Hermelin under the terms of his Indemnification Agreement for legal fees covering various matters including the FDA and SEC investigations, the Department of Justice inquiry, the Audit Committee investigation, HHS OIG exclusion and various class
70
Table of Contents
action lawsuits, under the terms of the Indemnification Agreement between Mr. M. Hermelin and the Company that was entered into when he served as Chairman of the Board and Chief Executive Officer of the Company. Under the Company’s standard Indemnification Agreement entered into with each director, as a condition for the advancement of expenses, each director is required to sign an undertaking to reimburse the Company for the advanced expenses in the event that it is found that the director is not entitled to indemnification. Mr. M. Hermelin’s demand for reimbursement of the $1.9 million fine and forfeiture is under review by a special committee appointed by the Board of Directors of the Company.
Current global economic conditions may adversely affect our industry, business, financial position and results of operations.
The global economy is currently undergoing a period of unprecedented volatility, and the future economic environment may continue to be less favorable than that of recent years. This has led, and could further lead, to reduced consumer spending in the foreseeable future, and this may include spending on health care. While generic drugs present an attractive alternative to higher-priced branded products, our sales could be negatively impacted if patients forego obtaining healthcare. In addition, reduced consumer spending may drive us and our competitors to decrease prices. These conditions may adversely affect our industry, business, financial position and results of operations.
Risks Relating to our Common Stock
The market price of our securities has been and may continue to be volatile.
The market prices of securities of companies engaged in pharmaceutical development and marketing activities historically have been highly volatile and the market price of our common stock has significantly declined. In addition, any or all of the following may have a significant impact on the market price of our common stock, among other factors: our ability to continue as a going concern; developments with respect to Makena™ such as its market launch and future revenues and federal and state governmental actions with respect to Makena™, its sales, the coverage and reimbursement of Makena™, and sales of compounded alternatives and other alternative therapies; developments with respect to our compliance with our debt obligations; developments regarding our compliance with the consent decree and returning certain or many of our products to market, including loss of market share as a result of the suspension of shipments, and related costs; developments regarding the relevant parties’ compliance with the plea agreement, the Divestiture Agreement or the Settlement Agreement; the sale by Mr. M. Hermelin or the Hermelin family trusts of their ownership interests in the Company; developments regarding our ability to raise additional capital; our recent financing arrangements; developments regarding private and government litigation and governmental inquiries; our reporting of prices used by government agencies or third parties in setting reimbursement rates; the introduction by other companies of generic or competing products; the eligibility of our products for Medicaid, Medicare or other reimbursement; announcements by us or our competitors of technological innovations or new commercial products; delays in the development or approval of products; regulatory withdrawals of our products from the market; developments or disputes concerning patent or other proprietary rights; publicity regarding actual or potential medical results relating to products marketed by us or products under development; regulatory developments in both the U.S. and foreign countries; publicity regarding actual or potential acquisitions; public concern as to the safety of our drug technologies or products; financial condition and results which are different from securities analysts’ forecasts; economic and other external factors; and period-to-period fluctuations in our financial results.
Future sales of common stock could adversely affect the market price of our Class A or Class B Common Stock.
As of March 31, 2011, an aggregate of 3,126,960 shares of our Class A Common Stock and 20,000 shares of our Class B Common Stock were issuable upon exercise of outstanding stock options under our stock option plans, and an additional 2,858,173 shares of our Class A Common Stock and 1,316,550 shares of Class B Common Stock were reserved for the issuance of additional options and shares under these plans (the Class A
71
Table of Contents
Common Stock and the Class B Common Stock are referred to collectively in this Report as the “common stock”). In addition, as of March 31, 2011, 20,038,410 shares of Class A Common Stock were reserved for issuance upon exercise of the Warrants, 8,691,880 shares of Class A Common Stock were reserved for issuance upon conversion of $200.0 million principal amount of 2033 Notes, and 337,500 shares of our Class A Common Stock were reserved for issuance upon conversion of our outstanding 7% cumulative convertible preferred stock (“7% Convertible Preferred Stock”). In the Settlement Agreement, Mr. M. Hermelin agreed to dispose of approximately 1,800,000 shares which, if sold on the open market, could have an adverse effect on the trading price of our shares. In addition, we have agreed to register the resale of 9,950,000 shares of Class A Common Stock issued to certain institutional investors in February 2011.
Future sales of our common stock and instruments convertible or exchangeable into our common stock and transactions involving equity derivatives relating to our common stock, or the perception that such sales or transactions could occur, could adversely affect the market price of our common stock.
Our By-Laws require the unanimous approval by the members of the Board of certain acts or resolutions of the Board, which could limit our ability to issue equity securities or raise capital.
Section 13 of Article III of our By-Laws provides that the following acts or resolutions of the Board or any committee of the Board require approval by a unanimous affirmative vote or unanimous written consent of the members of the Board then in office (other than any directors who affirmatively recuse themselves prior to the vote):
| • | the approval of any agreement or contract, or the issuance of any security, which confers stockholder voting rights; |
| • | the increase in the number of the members of the Board, in accordance with Section 1 of Article III of the By-Laws, to a number which is in excess of eight; and |
| • | the approval of any contract, agreement or other document or instrument which contains any provision (1) which imposes a penalty, acceleration of debt, purchase obligation or other adverse effect upon the corporation resulting from the election or appointment of any individual to the Board or the removal of any member of the Board or (2) which restricts, limits or dilutes the right of the stockholders to elect or appoint any individual to the Board or to remove any member of the Board. |
As a result, our ability to issue equity securities or to enter into agreements that include certain provisions related to a change of control may be limited, which could adversely affect our ability to raise capital and to meet our obligations as they become due.
Our Board of Directors has the authority to issue preferred stock which could affect the rights of holders of our common stock.
Our Amended Certificate of Incorporation authorizes the issuance of “blank check” preferred stock with such designations, rights and preferences as may be determined from time to time by the Board of Directors. Accordingly, the Board of Directors is empowered, without stockholder approval, to issue preferred stock with dividend, liquidation, conversion, voting or other rights which could adversely affect the voting power or other rights of the holders of common stock. Holders of common stock will have no preemptive rights to subscribe for a pro rata portion of any preferred stock which may be issued. In the event of issuance, the preferred stock could be utilized, under certain circumstances, as a method of discouraging, delaying or preventing a change in control. The possible impact on takeover could adversely affect the price of the common stock. Although we have no present intention to issue any shares of preferred stock, we may do so in the future.
Our Amended Certificate of Incorporation and Delaware law may have anti-takeover effects.
Our Amended Certificate of Incorporation authorizes the issuance of common stock in two classes, Class A Common Stock and Class B Common Stock. Each share of Class A Common Stock entitles the holder to one-twentieth of one vote on all matters to be voted upon by stockholders, while each share of Class B Common
72
Table of Contents
Stock entitles the holder to one full vote on each matter considered by the stockholders. In addition, our Board, with a unanimous vote, has the authority to issue additional shares of preferred stock and to determine the price, rights, preferences, privileges and restrictions of those shares without any further vote or action by the stockholders. The rights of the holders of common stock will be subject to, and may be adversely affected by, the rights of the holders of any preferred stock that may be issued in the future. The existence of two classes of common stock with different voting rights and the ability of our Board to issue additional shares of preferred stock could make it more difficult for a third party to acquire a majority of our voting stock. Other provisions of our Amended Certificate of Incorporation and By-Laws also may have the effect of discouraging, delaying or preventing a merger, tender offer or proxy contest, which could have an adverse effect on the market price of our Class A Common Stock.
In addition, certain provisions of Delaware law applicable to our Company could also delay or make more difficult a merger, tender offer or proxy contest involving our Company, including Section 203 of the Delaware General Corporation Law, which prohibits a Delaware corporation from engaging in any business combination with any “interested stockholder” (as defined in the statute) for a period of three years unless certain conditions are met. In addition, our senior management is entitled to certain payments upon a change in control and certain of the stock options we have granted provide for the acceleration of vesting in the event of a change in control of our company.
If we do not meet the New York Stock Exchange continued listing requirements, our common stock may be delisted.
In order to maintain our listing on the NYSE, we must continue to meet the NYSE minimum share price listing rule, the minimum market capitalization rule and other continued listing criteria. If our common stock were delisted, it could (1) reduce the liquidity and market price of our common stock; (2) negatively impact our ability to raise equity financing and access the public capital markets; and (3) materially adversely impact our results of operations and financial condition. At certain points during calendar year 2010, the price and 30-day average price of our Class A Common Stock and Class B Common Stock failed to satisfy the quantitative listing standards of the NYSE. Even though the price and 30-day average price of our Class A Common Stock and Class B Common Stock have again begun satisfying the quantitative listing standards of the NYSE, including with respect to minimum share price and public float, we can provide no assurance that they will remain at such levels.
We do not currently intend to pay dividends on our common stock.
Since 1980, we have not declared or paid any cash dividends on our common stock and we do not plan to do so in the foreseeable future. No dividends may be paid on Class A Common Stock or Class B Common Stock unless all dividends on the Cumulative Convertible Preferred Stock have been declared and paid. The Indentures for our 2015 Notes prohibit us from paying dividends in most cases. Holders of one class of common stock are entitled to receive dividends, other than dividends payable in our capital stock, only if dividends in the same type of property are simultaneously declared with respect to the other class of common stock. When, and if, we declare and distribute dividends on the Class B Common Stock, other than dividends payable in our capital stock, the Class A Common Stock must receive a dividend in the amount of 120% of the dividend distributed on the Class B Common Stock. Therefore, you are not likely to receive any dividends on your common stock for the foreseeable future.
| Item 1B. | Unresolved Staff Comments |
None.
73
Table of Contents
| Item 2. | Properties |
Our corporate headquarters is located at One Corporate Woods Drive, Bridgeton, Missouri. We currently lease a building from a partnership affiliated with Marc S. Hermelin, our former Chairman and former Board member and former Chief Executive Officer and David S. Hermelin, a director. This lease expires on December 31, 2011.
In addition, we lease or own the facilities shown in the following table. All of these facilities are located in the St. Louis, Missouri metropolitan area.
| SQUARE | LEASE | RENEWAL | ||||||||
| FOOTAGE |
USAGE |
EXPIRES | OPTIONS | |||||||
| Leased Facilities |
||||||||||
| 35,000 KV Offices |
12/31/11 | 3 Years | ||||||||
| 23,000 |
KV Office/R&D/Mfg. | 12/31/11 | 5 Years(1) | |||||||
| 41,316 |
KV Warehouse | 11/30/11 | None | |||||||
| 33,860 |
KV Offices | 05/01/13 | 5 Years | |||||||
| 133,176 |
||||||||||
| Owned Facilities |
||||||||||
| 124,000 |
KV Office/Mfg.(3) | |||||||||
| 121,731 |
KV Office/Whse./Lab(2) | |||||||||
| 90,336 |
KV Mfg.(3) | |||||||||
| 90,000 |
KV Lab(3) | |||||||||
| 315,000 |
KV Mfg./Whse./Corporate/Nesher/Ther-Rx Office(2) | |||||||||
| 260,000 |
Nesher/Ther-Rx(2) | |||||||||
| 1,001,067 |
||||||||||
| (1) | Two five-year options that the Company does not intend to exercise. |
| (2) | In March 2006, we entered into a $43.0 million mortgage loan arrangement with one of our primary lenders secured, in part, by these properties. This loan bears interest at a rate of 5.91% (and a default rate of 10.905%) and matures on April 1, 2021. |
| (3) | Assets are classified as available for sale at March 31, 2011. |
Properties used in our operations are considered suitable for the purposes for which they are used and are believed to be adequate to meet our needs for the reasonably foreseeable future. However, we will consider leasing or purchasing additional facilities from time to time, when attractive facilities become available, to accommodate the consolidation of certain operations and to meet future operational plans.
| Item 3. | Legal Proceedings |
The information set forth under Note 15—“Commitments and Contingencies—Litigation and Governmental Inquiries” of the Notes to Consolidated Financial Statements included in this Report is incorporated in this Item 3 by reference.
74
Table of Contents
| Item 4. | (Removed and Reserved) |
| Item 4A. | Executive Officers of the Registrant |
The following is a list of our current executive officers, their ages, their positions with our Company and their principal occupations for at least the past five years.
| NAME |
AGE |
POSITION HELD AND PAST EXPERIENCE | ||
| Gregory J. Divis, Jr. | 44 | President and Chief Executive Officer since November 2010; Interim President and Interim Chief Executive Officer from June 2010 to present; President, Ther-Rx Corporation from July 2007 to present; Vice President, Business Development and Life Cycle Management, Sanofi-Aventis U.S. from February 2006 to July 2007; Vice President Sales, Respiratory East, Sanofi-Aventis U.S. from June 2004 to February 2006; Executive Director, Sales and Marketing National Accounts, Reliant Pharmaceuticals from December 2003 to June 2004; Vice President and Country Manager United Kingdom and Ireland, Schering-Plough from May 2002 to December 2003; Vice President, Field Operations Oncology-Biotech Division, Schering-Plough from October 2000 to April 2002. | ||
| Thomas S. McHugh | 46 | Chief Financial Officer and Treasurer since July 2010; Chief Accounting Officer of our Company since February 2010; Interim Chief Financial Officer, Interim Treasurer, Vice President of Finance and Corporate Controller of our Company from September 2009 to April 2010; Vice President of Finance and Corporate Controller of our Company from January 2009 to July 2010; Managing Director and Global Controller, BearingPoint, Inc. from 2005 to 2008; Chief Financial Officer, Huttig Building Products, Inc., from 2000 to 2005. | ||
| Gregory S. Bentley | 61 | Senior Vice President - Law and Secretary of our Company since June 2010 and August 2010, respectively; Member, Board of Directors of our Company since June 2010; in private practice as an attorney from January 2009 to June 2010; Senior Vice President and General Counsel of our Company from April 2006 to January 2009; Executive Vice President, General Counsel, Secretary and Corporate Compliance Officer, aaiPharma, Inc. from 1999 to April 2006. | ||
| Mark T. Hartman | 52 | President Generics Division of our Company since October 2010; President, North America, Wockhardt USA, LLC from 2009 to 2010; President, North America Generics, Dr. Reddy’s Laboratories, Inc. from 2002 to 2008. | ||
| Patrick J. Christmas | 40 | Vice President and General Counsel of our Company since June 1, 2011; General Counsel and Secretary for the Wellstat Companies from 2007 to 2011; General Counsel of BioVeris Corporation from 2005 to 2007. | ||
The executive officers serve at the discretion of the Board of Directors.
75
Table of Contents
PART II
| Item 5. | Market for the Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities |
| (a) | Principal Market |
Our Class A Common Stock and Class B Common Stock are traded on the New York Stock Exchange under the symbols KV.A and KV.B, respectively.
| (b) | Approximate Number of Holders of Common Stock |
As of April 29, 2011, there were 676 holders of record of Class A Common Stock and 271 holders of record of Class B Common Stock (not separately counting shareholders whose shares are held in “nominee” or “street” names).
| (c) | Stock Price and Dividend Information |
The high and low sales prices of our Class A and Class B Common Stock during each quarter of fiscal years 2011, 2010 and 2009, as reported on the New York Stock Exchange, were as follows:
| CLASS A COMMON STOCK | ||||||||||||||||
| FISCAL YEAR 2011 | FISCAL YEAR 2010 | |||||||||||||||
| QUARTER |
HIGH | LOW | HIGH | LOW | ||||||||||||
| First |
2.03 | 0.81 | 3.68 | 1.11 | ||||||||||||
| Second |
3.26 | 0.61 | 3.42 | 1.37 | ||||||||||||
| Third |
2.95 | 1.93 | 5.36 | 2.66 | ||||||||||||
| Fourth |
13.55 | 1.13 | 4.01 | 1.62 | ||||||||||||
| CLASS B COMMON STOCK | ||||||||||||||||
| FISCAL YEAR 2011 | FISCAL YEAR 2010 | |||||||||||||||
| QUARTER |
HIGH | LOW | HIGH | LOW | ||||||||||||
| First |
2.40 | 1.00 | 8.43 | 1.39 | ||||||||||||
| Second |
3.38 | 0.70 | 4.75 | 2.27 | ||||||||||||
| Third |
3.15 | 2.24 | 6.00 | 3.63 | ||||||||||||
| Fourth |
13.52 | 1.41 | 4.10 | 1.98 | ||||||||||||
The high and low sales prices of our Class A Common Stock on April 29, 2011 were $4.16 and $3.93, respectively and of our Class B Common Stock on April 29, 2011 were $4.14 and $3.95, respectively, as reported on the NYSE. Since 1980, we have not declared or paid any cash dividends on our common stock and we do not plan to do so in the foreseeable future. No dividends may be paid on Class A Common Stock or Class B Common Stock unless all dividends on the 7% Convertible Preferred Stock have been declared and paid. Holders of one class of common stock are entitled to receive dividends, other than dividends payable in our capital stock, only if dividends in the same type of property are simultaneously declared with respect to the other class of common stock. When, and if, we declare and distribute dividends on the Class B Common Stock, other than dividends payable in our capital stock, the Class A Common Stock must receive a dividend in the amount of 120% of the dividend distributed on the Class B Common Stock. Also, the terms of the indenture governing the 2015 Notes restrict our ability to pay most dividends. The Board declared accrued dividends of $0.1 million and paid dividends of $0 in fiscal year 2011 and declared and paid dividends of $0.1 in fiscal year 2010 on 40,000 shares of outstanding 7% Convertible Preferred Stock. There were no undeclared and unaccrued cumulative preferred dividends at March 31, 2011.
76
Table of Contents
The Board reviews our dividend policy periodically. Any payment of dividends in the future will depend upon our earnings, capital requirements, financial condition and other factors considered relevant by our Board of Directors. See also Note 18—“Stock-Based Compensation” of the Notes to Consolidated Financial Statements included in this Report for information relating to our equity compensation plans.
| Issuer Purchases of Equity Securities | ||||||||||||||||
| Period |
Total number of shares purchased(a) |
Weighted average price paid per share |
Total number of shares purchased as part of publicly announced plans or programs |
Maximum number of shares (or units) that may yet be purchased under the plans or programs |
||||||||||||
| 1/1/11-1/31/11 |
— | — | — | — | ||||||||||||
| 2/1/11-2/28/11 |
— | — | — | — | ||||||||||||
| 3/1/11-3/31/11 |
— | — | — | — | ||||||||||||
| Total |
— | $ | — | — | — | |||||||||||
| (a) | Shares could be purchased from employees upon their departure from the Company pursuant to the terms of our 1991 and 2001 Stock Option Plans. |
Equity Compensation Plan Information
The following information regarding our compensation plans is furnished as of March 31, 2011, the end of our most recently completed fiscal year.
| Equity Compensation Plan Information Regarding Class A Common Stock |
||||||||||||
| Number of securities to be issued upon exercise of outstanding options, warrants and rights (a) |
Weighted-average exercise price of outstanding options, warrants and rights (b) |
Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) (c) |
||||||||||
| Plan Category |
||||||||||||
| Equity compensation plans approved by security holders(1) |
3,126,960 | $ | 5.92 | 2,858,173 | ||||||||
| Equity compensation plans not approved by security holders |
— | — | — | |||||||||
| Total |
3,126,980 | $ | 5.92 | 2,858,173 | ||||||||
| (1) | Consists of our 1991 and 2001 Incentive Stock Option Plans. See Note 18—“Stock-Based Compensation” of the Notes to Consolidated Financial Statements included in this Report. |
| Equity Compensation Plan Information Regarding Class B Common Stock |
||||||||||||
| Number of securities to be issued upon exercise of outstanding options, warrants and rights (a) |
Weighted-average exercise price of outstanding options, warrants and rights (b) |
Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) (c) |
||||||||||
| Plan Category |
||||||||||||
| Equity compensation plans approved by security holders(1) |
20,000 | $ | 24.77 | 1,316,550 | ||||||||
| Equity compensation plans not approved by security holders |
— | — | — | |||||||||
| Total |
20,000 | $ | 24.77 | 1,316,550 | ||||||||
| (1) | Consists of our 1991 and 2001 Incentive Stock Option Plans. See Note 18—“Stock-Based Compensation” of the Notes to Consolidated Financial Statements included in this Report. |
77
Table of Contents
Stock Price Performance Graph
Set forth below is a line-graph presentation comparing cumulative stockholders returns for the last five fiscal years on an indexed basis with the NYSE Composite Index and the S&P Pharmaceuticals Index, a nationally recognized industry standard index. The graph assumes the investment of $100 in our Class A Common Stock and $100 in our Class B Common Stock, the NYSE Composite Index, and the S&P Pharmaceuticals Index on March 31, 2006, and reinvestment of all dividends. Our stock’s performance may not continue into the future with the same or similar trends depicted in the graph below.
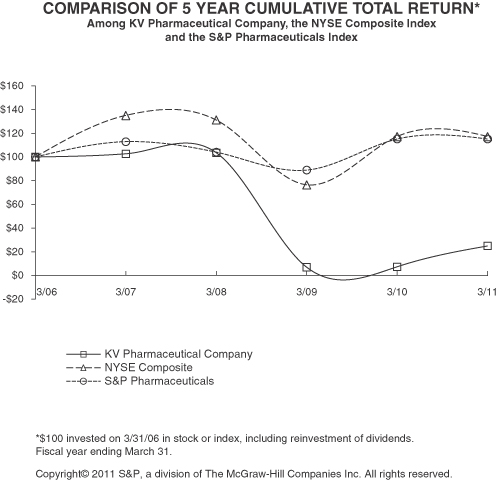
| Years Ended March 31, | ||||||||||||||||||||
| 2007 | 2008 | 2009 | 2010 | 2011 | ||||||||||||||||
| KV Pharmaceutical Company Class A |
102.53 | 103.48 | 6.84 | 7.30 | 24.83 | |||||||||||||||
| KV Pharmaceutical Company Class B |
102.41 | 104.23 | 10.00 | 8.76 | 24.65 | |||||||||||||||
| NYSE Composite |
135.00 | 131.15 | 76.42 | 117.20 | 117.20 | |||||||||||||||
| S&P Pharmaceuticals |
112.78 | 103.93 | 88.85 | 115.03 | 115.03 | |||||||||||||||
78
Table of Contents
| Item 6. | Selected Financial Data |
The following tables set forth selected historical consolidated financial data for our Company. The information in the following tables should be read in conjunction with our Consolidated Financial Statements and the Notes thereto in Part II, Item 8—“Financial Statements and Supplementary Data” and Part II, Item 7—“Management’s Discussion and Analysis of Financial Condition and Results of Operations” included in this Report to fully understand factors that may affect the comparability of the information presented below.
In March 2009, our Board approved management’s decision to market for sale PDI, our specialty materials segment. As a result of this decision, we have segregated PDI’s operating results and presented them separately as a discontinued operation for all periods presented. PDI was sold in June 2010.
In July 2010, our Board of Directors directed management to explore strategic alternatives with respect to Nesher and the assets and operations of our generic products business. In the fourth quarter of fiscal year 2011, management committed to a plan to divest the generics business. As a result, we identified the assets and liabilities of our generics business and have segregated the generics business operating results and presented them separately as discontinued operations for all periods presented.
BALANCE SHEET DATA
| March 31, | ||||||||||||||||||||
| 2011 | 2010 | 2009 | 2008 | 2007 | ||||||||||||||||
| (in millions) | ||||||||||||||||||||
| Cash, cash equivalents and marketable securities |
$ | 137.6 | $ | 60.7 | $ | 75.7 | $ | 129.0 | $ | 242.6 | ||||||||||
| Other current assets(a) |
148.9 | 31.0 | 154.5 | 269.6 | 207.0 | |||||||||||||||
| Property and equipment, net |
67.6 | 78.4 | 93.8 | 102.6 | 102.5 | |||||||||||||||
| Total assets |
564.7 | 358.6 | 659.2 | 890.4 | 720.3 | |||||||||||||||
| Current liabilities |
367.8 | 172.8 | 227.1 | 308.5 | 78.0 | |||||||||||||||
| Long-term debt excluding current maturity(b) |
418.3 | 233.2 | 200.7 | 68.7 | 239.5 | |||||||||||||||
| Shareholders’ equity (deficit) |
(374.0 | ) | (139.2 | ) | 139.5 | 453.6 | 360.4 | |||||||||||||
| (a) | We classified ARS of $65.9 million, $66.0 million and $81.5 million as of March 31, 2010, 2009 and 2008, respectively, as non-current assets to reflect the current lack of liquidity in these investments. ARS of $57.2 million is classified as current assets at March 31, 2011 because our agreement with Citigroup Global Markets Inc. expires in January 2012. At March 31, 2007, these investments were classified as marketable securities (see Note 6—“Investment Securities” of the Notes to Consolidated Financial Statements included in this Report). |
| (b) | We were not in compliance with one or more of the requirements of the mortgage loan documentation as of March 31, 2010 and 2009. However, since we received a letter in August 2010 approving certain waivers of covenants under our mortgage loan agreement, the mortgage debt obligation that remained outstanding under the mortgage arrangement was classified as a long-term liability as of March 31, 2010 (see Part II, Item 7—“Management’s Discussion and Analysis of Financial Condition and Results of Operations—Liquidity and Capital Resources” in this Report for additional discussion). The Company is in compliance with the waivers at March 31, 2011. However, based upon current financial projections, the Company does not anticipate meeting the March 31, 2012 minimum net worth requirement. Accordingly, the loan is classified as a current liability at March 31, 2011. Also, holders of our 2033 Notes had the right to require us to repurchase all or a portion of our 2033 Notes on May 16, 2008 and, accordingly, we classified the 2033 Notes as a current liability as of March 31, 2008. Since no holders required us to repurchase all or a portion of our 2033 Notes on this date and because the next occasion holders may require us to repurchase all or a portion of our 2033 Notes is May 16, 2013, the 2033 Notes were classified as a long-term liability as of March 31, 2011, 2010 and 2009 (see Note 13—“Long-Term Debt” of the Notes to Consolidated Financial Statements included in this Report). |
79
Table of Contents
STATEMENT OF OPERATIONS DATA
| Years ended March 31, | ||||||||||||||||||||
| 2011 | 2010 | 2009 | 2008 | 2007 | ||||||||||||||||
| (in millions, except per share data) | ||||||||||||||||||||
| Net revenues(a) |
$ | 27.3 | $ | 9.1 | $ | 113.2 | $ | 207.8 | $ | 182.4 | ||||||||||
| Cost of sales |
1.1 | 1.6 | 15.1 | 24.4 | 23.8 | |||||||||||||||
| Operating expenses(b)(f)(h) |
125.7 | 314.8 | 388.9 | 258.2 | 183.5 | |||||||||||||||
| Loss from continuing operations(c)(d)(e)(g)(i) |
(156.2 | ) | (285.6 | ) | (267.8 | ) | (45.9 | ) | (14.5 | ) | ||||||||||
| Income (loss) from discontinued operations(j) |
(30.6 | ) | (7.2 | ) | (45.8 | ) | 132.3 | 71.1 | ||||||||||||
| Gain on sale of discontinued operations(k) |
12.8 | 9.2 | — | — | — | |||||||||||||||
| Net income (loss)(c)(d)(g) |
(174.0 | ) | (283.6 | ) | (313.6 | ) | 86.4 | 58.6 | ||||||||||||
| Earnings (loss) from continuing operations per share: |
||||||||||||||||||||
| Diluted—Class A common |
$ | (3.05 | ) | $ | (5.73 | ) | $ | (5.39 | ) | $ | (0.70 | ) | $ | (0.18 | ) | |||||
| Diluted—Class B common |
$ | (3.05 | ) | $ | (5.73 | ) | $ | (5.39 | ) | (0.60 | ) | (0.16 | ) | |||||||
| Shares used in per share calculation: |
||||||||||||||||||||
| Diluted—Class A common |
51.3 | 49.9 | 49.7 | 59.1 | 59.0 | |||||||||||||||
| Diluted—Class B common |
12.2 | 12.1 | 12.1 | 12.3 | 12.5 | |||||||||||||||
| Preferred Stock dividends |
$ | 0.1 | $ | 0.1 | $ | 0.1 | $ | 0.1 | $ | 0.1 | ||||||||||
| (a) | Net revenues in the fourth quarter of fiscal year 2011 included approximately $13.7 million resulting from sales of MakenaTM. The FDA approved MakenaTM in February 2011 and the Company began shipping vials in March 2011(see Part II, Item 7—“Management’s Discussion and Analysis of Financial Condition and Results of Operations” included in this Report). |
| (b) | Operating expenses in fiscal year 2010 included charges of $85.8 million related to the impairment of intangible assets and property and equipment of our continuing operations (see Note 4—“Restructuring and Impairment Charges” of the Notes to Consolidated Financial Statements included in this Report) and $70.0 million of purchased in-process research and development expense for the cash payment made upon execution of the Makena Amendment (see Note 5—“Acquisition” of the Notes to Consolidated Financial Statements included in this Report). |
| (c) | The loss from continuing operations and net loss in year ended March 31, 2011 included a loss on extinguishment of debt of $106.2 million in connection with extinguishing the Bridge Loan in November 2010 and March 2011 (see Part II Item 7—“Management’s Discussion and Analysis of Financial Condition and Results of Operations” included in this Report). |
| (d) | The loss from continuing operations and net loss in fiscal year 2010 included the impact of an $88.2 million valuation allowance that was charged to income tax expense during the fiscal year ended March 31, 2010 for deferred tax assets that the Company determined would not be realized as tax deductions in the future. |
| (e) | Loss from continuing operations in fiscal 2011 includes an unrealized gain of $70.7 million for warrants. |
| (f) | Operating expenses in fiscal year 2009 included charges of $36.6 million related to impairment of intangible assets, $9.8 million of severance benefits, $0.8 million of expense for administrative costs associated with product recalls, $50.0 million of litigation expense related to actual and probable legal settlements, and $2.0 million of purchased in-process research and development expenses related to a milestone payment made in accordance with the Makena™ acquisition. |
| (g) | The loss from continuing operations and the net loss in fiscal year 2009 included the impact of an $82.4 million valuation allowance that was charged to income tax expense during the year ended March 31, 2009 for deferred tax assets that we determined would not be realized as tax deductions in the future. |
| (h) | Operating expenses in fiscal year 2008 included purchased in-process research and development expenses of $10.0 million and $7.5 million recorded in connection with the Evamist® and Makena™ acquisitions, respectively. |
80
Table of Contents
| (i) | Net income in fiscal year 2007 included $2.0 million, net of tax, related to the cumulative effect of a change in accounting principle for the adoption of the FASB’s authoritative guidance on share-based payments. |
| (j) | As a result of the decision by the Company to sell PDI, the Company has identified the assets and liabilities of PDI as held for sale in the Company’s consolidated balance sheets at March 31, 2010 and 2009 and has segregated PDI’s operating results and presented them separately as a discontinued operation for all periods presented. The Company has committed to a plan to divest the Company’s generics business. As a result the Company decision to divest the generic’s business, the Company has segregated generic’s business operating results and presented them separately as a discontinued operation for all periods presented. Additionally, in fiscal year 2010, included in discontinued operations is a gain on sale of $14.0 million for the sale of the Company’s Paragraph IV ANDA with the FDA for a generic equivalent version of GlaxoSmithKline’s Duac® gel to Perrigo Company and of $0.5 million for the sale of certain intellectual property and other assets associated with the ANDA for the generic version of Lotensin® 5-mg, 10-mg, 20-mg, and 40-mg Tablets to Huahai US, Inc. (see Part II, Item 7—“Management’s Discussion and Analysis of Financial Condition and Results of Operations” included in this Report). |
| (k) | During fiscal year 2011, the Company sold PDI on June 2, 2010 and recorded a gain on sale of $5.9 million, net of tax and sold the Sucralfate ANDA for approximately $11.0 million, generating a gain of $6.9 million, net of tax. |
81
Table of Contents
| Item 7. | Management’s Discussion and Analysis of Financial Condition and Results of Operations |
Except for the historical information contained herein, the following discussion contains forward-looking statements that are subject to known and unknown risks, uncertainties, and other factors that may cause our actual results to differ materially from those expressed or implied by such forward-looking statements. These risks, uncertainties and other factors are discussed throughout this Report and specifically under the captions “Cautionary Note Regarding Forward-Looking Statements” and “Risk Factors.” In addition, the following discussion and analysis of the financial condition and results of operations should be read in conjunction with “Consolidated Financial Data” and our Consolidated Financial Statements and the notes thereto appearing elsewhere in this Report. Also, for purposes of the following discussion, please note that, in March 2009, the Board approved management’s decision to market for sale PDI, our specialty materials segment. As a result of the decision to sell PDI, we identified the assets and liabilities of PDI as held for sale at March 31, 2010 and March 31, 2009 and we have segregated PDI’s operating results and presented them separately as a discontinued operation for all periods presented. The Company sold PDI on June 2, 2010 and recognized a gain of $5.9 million, net of tax. Additionally, during the fourth quarter of fiscal year ended March 31, 2011, management committed to a plan to sell our generics business. As a result, we have identified the assets and liabilities of the generics business and identified them as held for sale at March 31, 2011 and 2010 and we have segregated the generic’s business operating results and presented them as discontinued operations for all periods presented.
Background
We are a fully integrated specialty pharmaceutical company that develops, manufactures, acquires and markets technologically-distinguished branded and generic/non-branded prescription pharmaceutical products. Our strategic goal is to become a specialty branded pharmaceutical marketing company primarily focused in women’s healthcare. We have a broad range of dosage form manufacturing capabilities, including tablets, capsules, creams, liquids and ointments. We conduct our branded pharmaceutical operations through Ther-Rx and, previously, we conducted our generic/non-branded pharmaceutical operations through ETHEX, which focused principally on technologically-distinguished generic products. Through PDI, divested in June 2010, we developed, manufactured and marketed technologically advanced, value-added raw material products for the pharmaceutical industry and other markets. In May 2010, we formed a wholly-owned subsidiary, Nesher, to operate as the sales and marketing company for our generic products. The Company expects to complete a divestiture of Nesher and the Company’s generics business by the end of calendar year 2011.
Our original strategy was to engage in the development of proprietary drug delivery systems and formulation technologies which enhance the effectiveness of new therapeutic agents and existing pharmaceutical products. Today we utilize one of those technologies, SITE RELEASE® in two products expected to return to our branded portfolio in the second half of fiscal year 2012. Going forward, our business strategy will be primarily defined by the potential in-licensing and acquisition of pharmaceutical products rather than the historical strategy of the internal development of pharmaceutical products.
See Part I, Item 1—“Business—(b) Significant Developments” in this Report for information on recent business activity related to our Company, including changes in management and directors, discontinuation of manufacturing and distribution , product recalls, the consent decree with the FDA, the plea agreement with the Department of Justice, the agreements with the Office of Inspector General of the U.S. Department of Health and Human Services, the terms of acquisition of Makena™, the FDA’s approval of Makena™, the FDA inspections of our Company, our workforce reductions and cost conservation actions, recent financing, private placement and debt issuances, and our ability to continue as a going concern.
Ability to Continue as a Going Concern
There is substantial doubt about our ability to continue as a going concern. Our Consolidated Financial Statements included in this Report are prepared using accounting principles generally accepted in the United
82
Table of Contents
States applicable to a going concern, which contemplates the realization of assets and liquidation of liabilities in the normal course of business. The historical consolidated financial statements included in this Report do not include any adjustments that might be necessary if we are unable to continue as a going concern. The report of our independent registered public accountants, BDO USA, LLP, included in this Report, includes an explanatory paragraph related to our ability to continue as a going concern.
The assessment of our ability to continue as a going concern was made by management considering, among other factors: (1) the timing and number of approved products that will be reintroduced to the market and the related costs; (2) the suspension of shipment of all products manufactured by us and the requirements under the consent decree with the FDA; (3) the possibility that we may need to obtain additional capital despite the proceeds from the offering of the 2015 Notes in March 2011 and the equity we were able to issue in February 2011 (see Note 1—“Description of Business—Significant Developments” of the Notes to Consolidated Financial Statements included in this Report); (4) the potential outcome with respect to the governmental inquiries, litigation or other matters described in Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report; (5) our ability to comply with debt covenants; and (6) our ability to obtain future revenues from the sales of Makena™ sufficient to meet our future needs and expectations. Our assessment was further affected by our fiscal year 2011 net loss of $174.0 million, our 2010 net loss of $283.6 million, and the outstanding balance of cash and cash equivalents of $137.6 million as of March 31, 2011. For periods subsequent to March 31, 2011, we expect losses to continue because we are unable to generate any significant revenues from more of our own manufactured products until we are able to begin selling more of our approved products with FDA’s approval and with respect to products manufactured for us by third parties until after we are able to generate significant sales of Makena™ which was approved by the FDA in February 2011. We received notification from the FDA on September 8, 2010 of approval for our discontinued generics operations to ship into the marketplace the first product approved under the consent decree, i.e., the Potassium Chloride Extended Release Capsule. We resumed shipment of extended-release potassium chloride capsule, Micro-K® 10mEq and Micro-K® 8mEq, in September 2010, resumed shipments of the generic version of Potassium Chloride Extended Release Capsule in December 2010 and we began shipping Makena™ in March 2011. We have continued to ship Evamist®, which is manufactured for the Company by a third party, during the period covered by the consent decree. We are continuing to prepare other products for FDA inspection and do not expect to resume shipping other products until the second half of fiscal year 2012, at the earliest. In addition, we must meet ongoing operating costs as well as costs related to the steps we are currently taking to introduce Makena™, and to prepare for introducing and reintroducing other approved products to the market. If we are not able to obtain the FDA’s clearance to resume manufacturing and distribution of more of our approved products in a timely manner and at a reasonable cost, or if revenues from the sale of approved products introduced or reintroduced into the market place prove to be insufficient, our financial position, results of operations, cash flows and liquidity will continue to be materially adversely affected. These conditions raise substantial doubt about our ability to continue as a going concern.
Based on current financial projections, we believe the continuation of our Company as a going concern is primarily dependent on our ability to address, among other factors: (1) sales of Makena™, which was approved by the FDA in February 2011 notwithstanding recent actions by the FDA to permit continued sale of compounded alternatives and by CMS to permit compounded alternatives to be reimbursed under the Medicaid program; (2) the timing, number and revenue generation of approved products that will be introduced or reintroduced to the market and the related costs; (3) the suspension of shipment of all products manufactured by us and the requirements under the consent decree with the FDA (other than the Potassium Chloride Extended Release Capsule, including Micro-K® 10mEq and Micro-K® 8mEq, products that are the subject of the FDA notification letter discussed above); (4) the possibility that we will need to obtain additional capital; (5) the potential outcome with respect to the governmental inquiries, litigation or other matters described in Note 15— “Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report; and (6) compliance with our debt covenants. While we address these matters, we must continue to meet expected near-term obligations, including normal course operating cash requirements and costs associated with introducing or reintroducing approved products to the market (such as costs related to our employees, facilities
83
Table of Contents
and FDA compliance), remaining payments associated with the acquisition and retention of the rights to Makena™ (see Note 5—“Acquisition” of the Notes to Consolidated Financial Statements included in this Report), the financial obligations pursuant to the plea agreement, costs associated with our legal counsel and consultant fees, as well as the significant costs associated with the steps taken by us in connection with the consent decree and the litigation and governmental inquiries. If we are not able to obtain the FDA’s clearance to resume manufacturing and distribution of certain or many of our approved products in a timely manner and at a reasonable cost and/or if we are unable to successfully commercialize Makena™, and/or if we experience adverse outcomes with respect to any of the governmental inquiries or litigation described in Note 15— “Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report, our financial position, results of operations, cash flows and liquidity will continue to be materially adversely affected. See “Risk Factors” included in Item I, Part 1A of this Report regarding additional risks we face with respect to these matters.
In the near term, we are focused on performing the following: (1) addressing the FDA’s concerns regarding patient access to MakenaTM and CMS policy permitting Medicaid program reimbursement of compounded products; (2) the continued commercial launch of Makena™; (3) meeting the requirements of the consent decree, which will allow our approved products to be reintroduced to the market (other than the Potassium Chloride Extended Release Capsule products, including Micro-K® 10mEq and Micro-K® 8mEq, that are the subject of the FDA notification letter previously discussed); (4) the divestiture of Nesher and the Company’s generics business; and (5) pursuing various means to minimize operating costs and increase cash. Since December 31, 2010, the Company has generated non-recurring cash proceeds to support its on-going operating and compliance requirements from a $32.3 million private placement of Class A Common Stock in February 2011 and the 2015 Notes in March 2011 (which were used, in part, to repay all existing obligations under the agreement with U.S. Healthcare) (see Note 13—“Long-Term Debt” of the Notes to Consolidated Financial Statements included in this Report for a description of the 2015 Notes and U.S. Healthcare loan). While the cash proceeds received to date were sufficient to meet near-term cash requirements, we are pursuing ongoing efforts to increase cash, including, the continued implementation of cost savings, the divestiture of Nesher and the Company’s generics business and other assets and the return of certain additional approved products to market in a timely manner. We cannot provide assurance that we will be able to realize additional cost reductions from reducing our operations, that some or many of our approved products can be returned to the market in a timely manner (other than the Potassium Chloride Extended Release Capsule, including Micro-K® 10mEq and Micro-K® 8mEq products that are the subject of the FDA notification letter previously discussed above), that our higher profit approved products will return to the market in the near term or at all or that we can obtain additional cash through asset sales or the sale of equity or the successful commercial launch of Makena™. If we are unsuccessful in our efforts to introduce or return our products to market at adequate levels, or to sell assets or raise additional equity, we may be required to further reduce our operations, including further reductions of our employee base, or we may be required to cease certain or all of our operations in order to offset the lack of available funding.
We continue to evaluate the sale of certain of our assets, including Nesher and our generics business, which is included in discontinued operations and assets. To date, we are in advanced discussions for the sale of our generics business; however, we cannot assure that we will complete any transaction on favorable terms or at all. The Company is continuing to work with its advisors and several interested parties to complete a transaction. However, due to general economic conditions, we will likely be exposed to risks related to the overall macro-economic environment, including a lower rate of return than we have historically experienced on our invested assets and being limited in our ability to sell assets. In addition, we cannot provide any assurance that we ultimately will be successful in finding suitable purchasers for the sale of such assets. Even if we are able to find purchasers, we may not be able to obtain attractive terms and conditions for such sales, including attractive pricing. In addition, divestitures of businesses involve a number of risks, including the diversion of management and employee attention, significant costs and expenses, the loss of customer relationships, a decrease in revenues and earnings associated with the divested business, and the disruption of operations in the affected business. In addition, divestitures potentially involve significant post-closing separation activities, which could involve the expenditure of significant financial and employee resources. Inability to consummate identified asset sales or manage the post-separation transition arrangements could adversely affect our business, financial condition, results of operations and cash flows.
84
Table of Contents
Results of Operations
FISCAL YEAR 2011 COMPARED TO FISCAL YEAR 2010
Net Revenues
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2011 | 2010 | $ | % | ||||||||||||
| Total net revenues |
$ | 27.3 | $ | 9.1 | $ | 18.2 | 200.0 | % | ||||||||
Net revenues in fiscal year 2011 increased $18.2 million, or 200.2%, as compared to fiscal year 2010. The increase in net revenues was a result of $13.7 million of net revenues recorded in fiscal year 2011 related to the initial launch of MakenaTM. Also contributing to the increase was a $4.4 million increase in net revenues for Evamist®, which represents a 50% increase over fiscal year 2010.
Gross Profit
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2011 | 2010 | $ | % | ||||||||||||
| Total gross profit |
$ | 26.2 | $ | 7.5 | $ | 18.7 | 249.3 | % | ||||||||
| as % of total net revenues |
96.0 | % | 82.4 | % | ||||||||||||
The overall increase in gross profit of $18.7 million resulted primarily from $13.7 million related to the initial launch of MakenaTM, and an increase of $4.3 million or a 61.9% increase related to Evamist®.
Research and Development
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2011 | 2010 | $ | % | ||||||||||||
| Research and development |
$ | 22.1 | $ | 29.1 | $ | (7.0 | ) | (24.1 | )% | |||||||
| as % of net revenues |
81.0 | % | 319.8 | % | ||||||||||||
Research and development expenses consisted mainly of personnel-related costs, laboratory tests for proposed branded products, clinical studies to determine the safety and efficacy of proposed branded products, and material used in research and development activities. The decrease in research and development expense of $7.0 million for fiscal year 2011 was primarily due to a reduction in the work force and lower costs associated with the testing of drugs under development.
Selling and Administrative
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2011 | 2010 | $ | % | ||||||||||||
| Selling and administrative |
$ | 96.0 | $ | 111.7 | $ | (15.7 | ) | (14.1 | )% | |||||||
| as % of net revenues |
351.6 | % | 1,227.4 | % | ||||||||||||
The reduction in selling and administrative expenses was primarily due to lower personnel costs and lower depreciation and amortization expenses due to impairment charges recorded in fiscal year 2010 on intangible assets that lowered the cost of the related assets.
85
Table of Contents
Restructuring and Impairment Charges
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2011 | 2010 | $ | % | ||||||||||||
| Restructuring |
$ | 0.1 | $ | 3.2 | $ | (3.1 | ) | (96.9 | )% | |||||||
| as a % of net revenues |
0.4 | % | 35.2 | % | ||||||||||||
| Intangible Assets Impairment |
— | 82.3 | (82.3 | ) | -100.0 | % | ||||||||||
| as a % of net revenues |
0.0 | % | 904.4 | % | ||||||||||||
| Manufacturing, Distribution and Packaging Assets Impairment |
— | 3.5 | (3.5 | ) | -100.0 | % | ||||||||||
| as a % of net revenues |
0.0 | % | 38.5 | % | ||||||||||||
| Total Restructuring and Impairment Charges |
$ | 0.1 | $ | 89.0 | $ | (88.9 | ) | -99.9 | % | |||||||
| as a % of net revenues |
0.4 | % | 978.0 | % | ||||||||||||
Restructuring
On March 31, 2010, as part of an ongoing realignment of our cost structure we made further reductions of our workforce and employee headcount was reduced by approximately 80 for continuing operations. On February 25, 2011, the Company further reduced its workforce by 11 and laid off an additional 14 employees. We recorded expense of $0.1 million in fiscal year 2011 for severance benefits related to this headcount reduction. During fiscal 2010 we incurred $3.2 million in termination costs for reductions in our workforce.
Impairment
We assess the impairment of our long-lived assets whenever events or changes in circumstances indicate that the carrying value may not be recoverable. The factors that the Company considers in its assessment include the following: (1) significant underperformance of the assets relative to expected historical or projected future operating results; (2) significant changes in the manner of the Company’s use of the acquired assets or the strategy for its overall business; (3) significant negative industry or economic trends; and (4) significant adverse changes as a result of legal proceedings or governmental or regulatory actions. Refer to Note 4—“Restructuring and Impairment” of the Notes to Consolidated Financial Statements in this Report for further information regarding the assessment of long-lived assets for impairment.
During the fourth quarter of fiscal year 2011, we decided to divest the generics business and as a result of this decision, we evaluated our long-lived assets for impairment.
Certain significant events occurred in the three months ended March 31, 2010 that indicated that the carrying value of certain assets as of March 31, 2010 may not be recoverable. These events included: (a) the expectation of when we would be able to resume manufacturing and shipment and begin generating cash flow from the sale of certain of our approved products, (b) the entry into the plea agreement with the U.S. Department of Justice (see Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements in this Report) and (c) the reduction of our workforce that occurred on March 31, 2010 (see Note 4—“Restructuring and Impairment Charges” of the Notes to Consolidated Financial Statements in this Report).
Based on the events described above, we determined that a triggering event occurred in the fourth quarter of fiscal year 2011 and 2010 giving rise to the need to assess the recoverability of our long-lived assets. Based on the assessment of the recoverability of our long-lived assets, it was determined that future undiscounted cash flows were not sufficient to support the carrying value of certain of our long-lived assets, and this resulted in material non-cash charges for impairment of property and equipment and intangible assets in the quarters ended March 31, 2011 and 2010. Cash flow projections require a significant level of judgment and estimation in order to determine a number of interdependent variables and assumptions such as probability, timing, pricing and
86
Table of Contents
various cost factors. Cash flow projections are highly sensitive to changes in these variables and assumptions. We determined the following Manufacturing, Distribution & Packaging asset group (“MD&P”) assets were impaired:
| • | MD&P, an asset group consisting primarily of property and equipment used in manufacturing operations and a generic product portfolio was evaluated for impairment. Based upon the Company’s analysis, it was determined that MD&P assets were impaired as of March 31, 2011 and the Company recognized an impairment charge of $10.0 million and recorded the impairment charge in discontinued operations as the impaired assets in the 2011 analysis related to our generics business. See additional discussion below concerning our loss from discontinued operations. |
| • | Based upon the Company’s analysis, an impairment charge of $3.5 million was recorded during fiscal year March 31, 2010 to reduce the carrying value of our MD&P assets for continuing operations to their fair value. The Company recorded an impairment charge of $43.1 million for MD&P assets during the fiscal year March 31, 2010, related to our discontinued operations to reduce the carrying value of our MD&P assets for discontinued operations to their fair value. See additional discussion below concerning our loss from discontinued operations. |
Based upon the Company’s analysis, it was determined the following intangible assets were impaired as of March 31, 2010 due primarily to:
| • | The intangible assets related to Evamist®, had approximately $116.0 million of unamortized costs as of March 31, 2010 and remaining useful lives of 8 to 12 years related to product rights, trademark rights and rights under the sublicense agreement. To test impairment of these assets, the Company performed a cash flow analysis, which incorporated a number of significant assumptions and estimates. The sum of the projected undiscounted cash flows, which extend over the useful life assigned to the longest lived asset, did not exceed the carrying amount of the intangible assets as of March 31, 2010 and it was concluded that the assets related to Evamist®, were impaired as of that date. The Company recorded $79.0 million during fiscal year 2010 as an impairment charge to reduce the carrying value of the intangible assets related to Evamist® to their aggregate estimated fair value. |
Purchased In-Process Research and Development
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2011 | 2010 | $ | % | ||||||||||||
| Purchased in-process research and development |
$ | — | $ | 70.0 | $ | (70.0 | ) | (100.0 | )% | |||||||
| as % of net revenues |
0.0 | % | 769.2 | % | ||||||||||||
The decrease in purchased in-process research and development costs were pursuant to amendment number one to the agreement with Hologic (see Note 5—“Acquisition” of the Notes to Consolidated Financial Statements in this Report), which required a $70.0 million cash payment upon execution of the MakenaTM Amendment in January 2010. Because the product was not FDA approved at the time this payment was made and the project would have no alternative use if not approved, we recorded this payment as a purchased in-process research and development expense.
Litigation and Governmental Inquiries, net
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2011 | 2010 | $ | % | ||||||||||||
| Litigation and governmental inquiries, net |
$ | 7.4 | $ | 15.0 | $ | (7.6 | ) | (50.7 | )% | |||||||
| as % of net revenues |
27.1 | % | 164.8 | % | ||||||||||||
87
Table of Contents
The decrease in litigation and governmental inquiries expense of $7.6 million for fiscal year 2011 was primarily due to significant costs recognized in fiscal year 2010 which did not repeat in fiscal year 2011. The decrease was primarily related to the settlement with Strides that was recorded in fiscal year 2010 (see Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements in this Report).
Extinguishment of Debt
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2011 | 2010 | $ | % | ||||||||||||
| Loss on extinguishment of debt |
$ | 106.2 | $ | — | $ | 106.2 | NA | % | ||||||||
In November 2010, the Company entered into a senior secured debt financing arrangement with U.S. Healthcare which retired an existing $20.0 million loan. At the time the $20.0 million loan was retired, the Company wrote-off a proportionate share of a discount related to fair value of warrants of $8.0 million that were allocated to this loan. We also wrote-off approximately $1.9 million of deferred financing costs related to the $20.0 million loan. The Company and U.S. Healthcare amended the financing arrangements on January 6, 2011 and again on March 2, 2011. On March 17, 2011 with the issuance of the 2015 Notes, the loan with U.S. Healthcare was repaid. As a result of the repayment of the loans with U.S. Healthcare we recorded the following in loss on extinguishment of debt:
| • | A premium payment of $12.5 million; |
| • | Unamortized deferred financing costs of $3.9 million; and |
| • | $79.9 million in warrants expense. |
Change in warrant liability
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2011 | 2010 | $ | % | ||||||||||||
| Change in warrant liability |
$ | 70.7 | $ | — | $ | 70.7 | NA | % | ||||||||
The change in warrant liability is a result of the mark to market adjustment of the warrant liability from March 18, 2011 to March 31, 2011.
Interest Expense, net and other
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2011 | 2010 | $ | % | ||||||||||||
| Interest expense, net |
$ | 13.4 | $ | 2.2 | $ | 11.2 | 509.1 | % | ||||||||
The increase in interest expense for fiscal year 2011 resulted primarily from the increase in borrowing by the Company during fiscal 2011 and accretion of the present value of the liability to Hologic that was recorded as interest expense related to the Company’s investment in MakenaTM.
Income Tax Expense (Benefit)
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2011 | 2010 | $ | % | ||||||||||||
| Income tax expense (benefit) |
$ | 7.8 | $ | (23.9 | ) | $ | 31.7 | (132.6 | )% | |||||||
| Effective tax rate |
(4.7 | )% | 13.8 | % | ||||||||||||
88
Table of Contents
The provision for income taxes for fiscal year 2011 was primarily due to the timing of certain deferred tax liabilities which are not scheduled to reverse within the applicable carry forward periods. The benefit for fiscal year 2010 was due to the availability to carry back operating losses to prior periods partially offset by the timing of certain deferred tax liabilities which are not scheduled to reverse within the applicable carry forward periods for the deferred tax assets. Both periods were impacted by the recording of a valuation allowance for deferred tax assets that we determined were not more likely than not to be realized as future tax deductions.
Loss from Discontinued Operations
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2011 | 2010 | $ | % | ||||||||||||
| Loss from discontinued operations |
$ | (30.6 | ) | $ | (7.2 | ) | $ | (23.4 | ) | 325.0 | % | |||||
During the fourth quarter of fiscal year 2011, we committed to a plan to divest the generics business. During the fourth quarter of fiscal year 2009, our Board authorized management to sell PDI, our specialty materials segment (see Note 23—“Divestitures” of the Notes to Consolidated Financial Statements in this Report for more information regarding the divestiture of our generics business and the sale of PDI). Therefore, we have segregated the generic’s business and PDI’s operating results and presented them separately as discontinued operation for all periods presented. (see Note 23—“Divestitures” of the Notes to Consolidated Financial Statements included in this Report.). The reason for the losses was primarily due to the fact that the Company continues to not be able to produce product under the terms of the consent decree, except for Potassium Chloride Extended Release Capsule for which the Company received approval in September 2010. (see Note 15— “Commitments and Contingencies” of the Notes to Consolidated Financial Statements in this Report concerning the consent decree).
As a result of management’s decision to divest the generics business and an evaluation of offers received for the generics business it was determined that the assets related to MD&P were impaired. The Company recorded an impairment charge of $10.0 million during fiscal year 2011 as part of discontinued operations to reduce the carrying value of MD&P assets, product rights, and other assets to estimated fair value. (See Note 23—“Divestitures” of Notes to Consolidated Financial Statements in this Report).
The Company recorded revenues of approximately $143.0 million generated from the sale of all of the limited quantity of generic OxyContin® pursuant to the Distribution Agreement with Purdue, partially offset by approximately $20.0 million recognized as costs of sales for royalty fees owed to Purdue and costs of product paid to Purdue pursuant to the Distribution Agreement.
The Company recorded an impairment charge of $43.1 million for MD&P assets during the fiscal year March 31, 2010, related to our discontinued operations to reduce the carrying value of our MD&P assets for discontinued operations to their fair value.
Gains on sale in Discontinued Operations
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2011 | 2010 | $ | % | ||||||||||||
| Gains on sale in discontinued operations |
$ | 12.8 | $ | 9.2 | $ | 3.6 | 39.1 | % | ||||||||
During fiscal year 2011, the Company recognized a pre-tax gain of $11.0 million on the sale of certain intellectual property ($6.9 million, net of tax) and other assets related to our ANDA, submitted with the FDA for the approval to engage in the commercial manufacture and sale of 1gm/10mL sucralfate suspension. All activities related to the intellectual property of 1gm/10mL sucralfate suspension were expensed as incurred resulting in a gain equal to the cash proceeds received. Additionally, the Company sold PDI on June 2, 2010 and recognized a
89
Table of Contents
gain of $5.9 million net of tax. During fiscal year 2010, the Company recognized a pre-tax gain of $14.0 million for the sale of the generic equivalent version of GlaxoSmithKline’s Duac® gel to Perrigo Company ($8.7 million, net of tax).
The above activity was recorded in discontinued operations as a result of our decision to divest the generics business and PDI.
FISCAL YEAR 2010 COMPARED TO FISCAL YEAR 2009
Net Revenues
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2010 | 2009 | $ | % | ||||||||||||
| Total net revenues |
$ | 9.1 | $ | 113.2 | $ | (104.1 | ) | (92.0 | )% | |||||||
The decrease in branded product sales of $104.1 million for fiscal year 2010 was primarily due to ceasing all of our manufacturing operations, as described in Item 1—“Business—(b) Significant Recent Developments”, during the fourth quarter of fiscal year 2009. As a result, branded product net revenues in fiscal year 2010 were derived primarily from Evamist® which is manufactured by a third-party. Net revenues from Evamist® during fiscal year 2010 were approximately $8.6 million, while Evamist®, net revenues were nominal during fiscal year 2009. During fiscal year 2009, net revenues of three of our other branded products were $34.0 million, $22.9 million and $50.0 million, respectively.
The decrease in branded product net revenues for fiscal year 2010 was partially offset by $3.5 million received in the three months ended June 30, 2009 as license revenue related to the transfer of certain existing product registrations, manufacturing technology and intellectual property rights.
Gross Profit
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2010 | 2009 | $ | % | ||||||||||||
| Total gross profit |
$ | 7.5 | $ | 98.1 | $ | (90.6 | ) | (92.4 | )% | |||||||
| as % of total net revenues |
82.4 | % | 86.7 | % | ||||||||||||
The decrease in gross profit for fiscal year 2010 was due to the impact of ceasing all of our manufacturing operations, as described in Item 1—“Business—(b) Significant Recent Developments”, during the fourth quarter of fiscal year 2009. This limited our net revenues to sales of products manufactured by third parties and revenue generated from license agreements. Additionally, because we ceased all manufacturing activities, all costs associated with our manufacturing operations were immediately expensed as incurred.
Research and Development
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2010 | 2009 | $ | % | ||||||||||||
| Research and development |
$ | 29.1 | $ | 69.8 | $ | (40.7 | ) | (58.3 | )% | |||||||
| as % of net revenues |
319.8 | % | 61.7 | % | ||||||||||||
Research and development expenses consisted mainly of personnel-related costs, laboratory tests for proposed branded products, clinical studies to determine the safety and efficacy of proposed branded products, and material used in research and development activities. The decrease in research and development expense of
90
Table of Contents
$40.7 million for fiscal year 2010 was primarily due to lower personnel costs associated with the reduction in force that occurred in the fourth quarter of fiscal year 2009, lower costs associated with the testing of drugs under development due to a reduction in bioequivalency studies and lower material costs as a result of decreased research and development activities during fiscal year 2010 as compared to fiscal year 2009.
Selling and Administrative
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2010 | 2009 | $ | % | ||||||||||||
| Selling and administrative |
$ | 111.7 | $ | 220.7 | $ | (109.0 | ) | (49.4 | )% | |||||||
| as % of net revenues |
1,227.5 | % | 195.0 | % | ||||||||||||
The decrease in selling and administrative expense resulted primarily from the net impact of the following:
| • | $46.9 million decrease in marketing promotion expenses primarily related to suspending shipment of all approved tablet-form products in December 2008 and all other drug products in January 2009; |
| • | $57.1 million decrease due primarily to reductions in management, sales and other personnel related to the significant reductions in our workforce which occurred beginning in the fourth quarter of fiscal year 2009; |
| • | $3.7 million decrease in facility expenses due to the ongoing realignment of our cost structure that was necessitated by our product recalls and the requirements under the consent decree; |
| • | $8.3 million decrease in professional fees as a result of the decrease in fees related to the internal investigation conducted by the Audit Committee, which was completed in June 2009; |
| • | $8.2 million decrease in expenses for consulting, legal and other professional services for fiscal year 2010 due to a decrease in litigation activity; |
| • | $3.0 million decrease in product development costs as a result of our discontinuation of manufacturing and distribution and the ongoing realignment of our cost structure; |
| • | $10.8 million reversal in compensation expense recorded during fiscal year 2009 and that did not recur in fiscal year 2010. The reversal in fiscal year 2009 was for a previously recorded accrual associated with the former Chief Executive Officer’s retirement benefits as a result of the termination “for cause” of the former Chief Executive Officer’s employment agreement by the Board. Mr. M. Hermelin has informed us, however, that he believed he effectively retired from his employment with the Company prior to the termination of his employment agreement on December 5, 2008 by the Board; |
| • | $6.1 million decrease in compensation expense that was recorded during fiscal year 2009 and that did not recur in fiscal year 2010. In fiscal year 2009 we reduced a previously recorded accrual for payroll related withholding taxes on disqualified incentive stock options that resulted from a settlement with the IRS for calendar years 2004 to 2007; and |
| • | $7.6 million increase in FDA review expenses in fiscal year 2010 associated with engaging external resources to assist with reviews. |
91
Table of Contents
Due to the write-off of the intangible assets described below, amortization expense decreased by $2.0 million in fiscal year 2010 versus fiscal year 2009.
Restructuring and Impairment Charges
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2010 | 2009 | $ | % | ||||||||||||
| Restructuring |
$ | 3.2 | $ | 9.8 | $ | (6.6 | ) | (67.3 | )% | |||||||
| as a % of net revenues |
35.2 | % | 8.7 | % | ||||||||||||
| Intangible Assets Impairment |
82.3 | 36.6 | 45.7 | 124.9 | % | |||||||||||
| as a % of net revenues |
904.4 | % | 32.3 | % | ||||||||||||
| Manufacturing, Distribution and Packaging Assets Impairment |
3.5 | — | 3.5 | NA | % | |||||||||||
| as a % of net revenues |
38.5 | % | 0.0 | % | ||||||||||||
| Total Restructuring and Impairment Charges |
$ | 89.0 | $ | 46.4 | $ | 42.6 | 91.8 | % | ||||||||
| as a % of net revenues |
978.0 | % | 41.0 | % | ||||||||||||
Restructuring
On March 31, 2010, as part of an ongoing realignment of our cost structure we made further substantial reductions of our workforce and employee headcount was reduced for continuing operations. We recorded expense of $3.2 million in fiscal year 2010 for severance benefits related primarily to these terminations. During the quarter ended March 31, 2009, we reduced our headcount by approximately 564 employees through a combination of terminations and layoffs which resulted in severance benefits of approximately $9.8 million.
Impairment
We assess the impairment of long-lived and intangible assets whenever events or changes in circumstances indicate that the carrying value may not be recoverable. The factors that we consider in our assessment include the following: (1) significant underperformance of the assets relative to expected historical or projected future operating results; (2) significant changes in the manner of our use of the acquired assets or the strategy for its overall business; (3) significant negative industry or economic trends; and (4) significant adverse changes as a result of legal proceedings or governmental or regulatory actions. Certain significant events occurred in the three months ended March 31, 2010 that indicated that the carrying value of certain assets as of March 31, 2010 may not be recoverable. These events included: (a) the expectation of when we would be able to resume manufacturing and shipment and begin generating cash flow from the sale of certain of our approved products, (b) the entry into the plea agreement with the U.S. Department of Justice (see Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements in this Report) and (c) the substantial reduction of our workforce that occurred on March 31, 2010 (see Note 4—“Restructuring and Impairment Charges” of the Notes to Consolidated Financial Statements this Report). Furthermore, we decided to divest the generics business in the fourth quarter of fiscal 2011 and restated prior years to record our generics business as discontinued operations. See additional discussion below concerning our loss on discontinued operations.
Based on the events described above, we determined that a triggering event occurred in the fourth quarter of fiscal year 2010 giving rise to the need to assess the recoverability of our long-lived assets. Based on the assessment of the recoverability of our long-lived assets, it was determined that future undiscounted cash flows were not sufficient to support the carrying value of certain of our long-lived assets, and this resulted in material non-cash charges for impairment of property and equipment and intangible assets in the quarter ended March 31, 2010. Cash flow projections require a significant level of judgment and estimation in order to determine a number of interdependent variables and assumptions such as probability, timing, pricing and various cost factors. Cash flow projections are highly sensitive to changes in these variables and assumptions.
92
Table of Contents
Based upon management’s analysis, it was determined the following assets were impaired:
| • | Intangible assets related to Evamist®, had approximately $116.0 million of unamortized costs as of March 31, 2010 and remaining useful lives of 9 to 13 years related to product rights, trademark rights and rights under the sublicense agreement. To test impairment of these assets, we performed a cash flow analysis, which incorporates a number of significant assumptions and estimates. The sum of the projected undiscounted cash flows, which extend over the useful life assigned to the longest lived asset, did not exceed the carrying amount of the intangible assets as of March 31, 2010 and we concluded that the assets related to Evamist®, were impaired as of that date. We recorded $79.0 million during fiscal year 2010 as an impairment charge to reduce the carrying value of the intangible assets related to Evamist®, to their estimated fair value. |
| • | Intangible assets related to Micro-K® and patents and trademarks, had approximately $16.2 million and $3.3 million of unamortized costs as of March 31, 2010, respectively. We performed a cash flow analysis that extended over the average remaining useful life of these assets and determined that the assets related to patents and trademarks were impaired. We recorded an impairment charge of $3.3 million during fiscal year 2010 to reduce the carrying value of patents and trademarks to their estimated fair value. |
| • | Manufacturing, Distribution & Packaging asset group (“MD&P”), an asset group consisting primarily of property and equipment used in our manufacturing operations and a generic product portfolio, had approximately $112.4 million of net book value as of March 31, 2010. We performed a cash flow analysis that extended over the average remaining useful life of the assets and determined that the assets related to MD&P were impaired. We recorded an impairment charge of $3.5 million during fiscal year 2010 to reduce the carrying value of manufacturing equipment, and other assets to estimated fair value for our continuing operations and $43.1 million was recorded in discontinued operations. See further discussion below concerning discontinued operations. |
Additionally, because of the events that occurred in the beginning in the third quarter of 2009, which included four voluntary recalls involving 25 generic products and the suspension of all shipments of our FDA-approved tablet-form products, we completed an evaluation of each of our intangible assets. Based upon management’s analysis, it was determined that the following intangible assets were impaired for our continuing operations:
| • | Pursuant to the terms of the consent decree, we agreed not to distribute our unapproved products, including our prenatal vitamins and hematinic products, unless we obtain approval through the FDA’s ANDA or NDA processes. Since such products are unable to generate any revenues and are not approved by the FDA, we recorded $34.1 million during fiscal year 2009 as an impairment charge to write-off the remaining net book value of the intangible assets related to our branded prenatal vitamins and hematinic products. |
| • | During fiscal year 2009, we recognized an impairment charge of $2.5 million for the intangible assets related to a product we had under development to treat endometriosis. It was determined at the end of our Phase II study that this product was not effective because its efficacy was not better than that of a placebo. As a result, we decided to abandon development of the product in fiscal year 2009 and wrote-off the remaining net book value of the related intangible assets. |
| • | With respect to the intangible assets related to Evamist™, we had approximately $125.5 million of unamortized costs and remaining useful lives of 9 to 13 years as of March 31, 2009. To test impairment of these assets, we performed a cash flow analysis, which incorporates a number of significant assumptions and estimates that are subject to change as actual results become known. The sum of the projected undiscounted cash flows, which extend over the useful life assigned to the assets, slightly exceeds the carrying amount of the intangible assets as of March 31, 2009 and we concluded that the asset is not impaired as of that date. However, events and circumstances, primarily as of the fourth |
93
Table of Contents
| quarter of fiscal year 2010, required management to revise its earlier assumptions and estimates and such revisions resulted in the fiscal year 2010 impairments described above. |
Purchased In-Process Research and Development
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2010 | 2009 | $ | % | ||||||||||||
| Purchased in-process research and development |
$ | 70.0 | $ | 2.0 | $ | 68.0 | 3,400.0 | % | ||||||||
| as % of net revenues |
769.2 | % | 01.8 | % | ||||||||||||
In January 2008, we entered into a definitive asset purchase agreement to acquire the U.S. and worldwide rights to Makena™ (hydroxyprogesterone caproate) (see Note 5—“Acquisition” of the Notes to Consolidated Financial Statements in this Report). Under the terms of the asset purchase agreement, we made a $2.0 million cash payment in May 2008 upon achievement of a milestone. Because the product was not FDA approved at the time when this payment was made, we recorded this payment as an in-process research and development charge.
Pursuant to the Amended MakenaTM Agreement (see Note 5—“Acquisition” of the Notes to Consolidated Financial Statements in this Report), a $70.0 million cash payment was due upon execution of the MakenaTM Amendment. We made that payment in the fourth quarter of fiscal year 2010. Because the product was not FDA approved at the time when this payment was made and the project would have no alternative use if not approved, we recorded this payment as an in-process research and development charge.
Litigation and Governmental Inquiries, net
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2010 | 2009 | $ | % | ||||||||||||
| Litigation and governmental inquiries, net |
$ | 15.0 | $ | 50.0 | $ | (35.0 | ) | (70.0 | )% | |||||||
| as % of net revenues |
164.8 | % | 44.2 | % | ||||||||||||
The decrease in litigation and governmental inquiries expense of $35.0 million for fiscal year 2010 versus fiscal year 2009 was primarily due to significant costs recognized in fiscal year 2009 associated with:
| • | the plea agreement with the U.S. Department of Justice recorded during the three months ended September 30, 2008 (see Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements in this Report); |
| • | various product liability actions (based on our product liability insurance coverage and the number of product liability actions that were pending, that had been settled and that we expect to occur) related to the voluntary product recalls initiated by us in calendar years 2008 and early 2009 and alleged damages as a result of the ingestion of purportedly oversized tablets allegedly distributed in 2007 and 2008 (see Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements in this Report); and |
| • | the settlement agreement with Axcan ScandiPharm, Inc. (“Axcan”) that was recorded during fiscal year 2009 (see Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements in this Report). |
Additionally, we recorded an adjustment of $1.9 million in fiscal year 2010 to reduce our expected direct liability (as discussed above) for product liability actions related to the voluntary product recalls initiated by us in calendar year 2008 and early 2009 and alleged damages as a result of the ingestion of purportedly oversized tablets allegedly manufactured and distributed by us.
94
Table of Contents
The decreases noted above were partially offset by a settlement agreement with Strides that was reached on December 13, 2010. At March 31, 2010, the Company had a carrying value of approximately $14.4 million related to an investment in preferred shares of Strides, including accrued dividends. Under the terms of the agreement, both parties agreed to settle all claims and disputes, Strides agreed to pay the Company $7.3 million, and the Company agreed to redeem all previously issued preferred shares. The difference between the carrying value of the preferred shares and accrued dividends at March 31, 2010 and the payment made by Strides was recorded as litigation expense and the carrying value of the preferred shares and accrued dividends was reduced to $7.3 million.
Interest Expense
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2010 | 2009 | $ | % | ||||||||||||
| Interest expense, net |
$ | 2.2 | $ | 15.6 | $ | (13.4 | ) | (85.9 | )% | |||||||
Interest and Other Income (Expense)
The decrease in interest expense, net of $13.4 million for fiscal year 2010 versus fiscal year 2009 consisted of the following:
| • | Decreases in expense of $10.9 million related to other-than-temporary impairment recognized into earnings during fiscal year 2009 on our ARS, offset in part by a $1.3 million gain recognized for a settlement agreement with one of our brokers for a $10.0 million ARS; |
| • | Increases in income of $3.5 million due to the recognition of foreign currency transaction gains on an investment denominated in the Indian Rupee as compared to transactions losses on the investment recognized in fiscal year 2009; and |
| • | Declines in interest income of $3.7 million due to the impact of lower short-term market interest rates, which reduced our weighted average interest rate on interest-earning assets for fiscal year 2010 as compared to fiscal year 2009. |
Income Tax Benefit
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2010 | 2009 | $ | % | ||||||||||||
| Income tax benefit |
$ | (23.9 | ) | $ | (38.6 | ) | $ | 14.7 | (38.1 | )% | ||||||
| Effective tax rate |
7.7 | % | 12.6 | % | ||||||||||||
The benefit for income taxes for fiscal years 2010 and 2009 was primarily due to the availability to carry back operating losses to prior periods. The benefit for 2009 included the carry back of operating losses to the two prior years partially offset by a valuation allowance. The benefit for 2010 included the additional carry back period for a period of five years allowed as a result of a change in tax law. The benefit for both years was partially offset by the timing of certain deferred tax liabilities which are not scheduled to reverse within the applicable carry forward periods for the deferred tax assets.
Income (loss) from Discontinued Operations
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2010 | 2009 | $ | % | ||||||||||||
| Income (loss) from discontinued operations |
$ | (7.2 | ) | $ | (45.8 | ) | $ | 38.6 | (84.3 | )% | ||||||
95
Table of Contents
During the fourth quarter of fiscal year 2011 we committed to a plan to divest the generics business. During the fourth quarter of fiscal year 2009, our Board authorized management to sell PDI, our specialty materials segment (see Note 23—“Divestitures” of the Notes to Consolidated Financial Statements in this Report for more information regarding the sale of PDI). Therefore, we have segregated our generics business and PDI’s operating results and presented them separately as a discontinued operation for all periods presented. (See Note 23— “Divestitures” of Notes to Consolidated Financial Statements included in this Report.) The decrease in loss from discontinued operations for fiscal year 2010 was due to decreases in PDI’s total operating expenses, mostly due to an insurance gain, partially offset by decreases in gross profit. PDI was sold in June 2010. In addition, during the three months ended December 31, 2009, the Company received from Purdue and sold to its customers all of the generic OxyContin® allotted under the Distribution Agreement and recognized net revenue of approximately $143.0 million in the consolidated statement of operations. The Company recognized the revenue when it had determined that persuasive evidence of an arrangement existed, the customers’ payment ability had been reasonably assured, title and risk of ownership had been transferred to the customers, and the Company’s price to its customers was determined. Additionally, the Company recorded approximately $20.0 million as cost of sales in the three months ended December 31, 2009, which included royalty fees and the cost of the generic OxyContin® supplied by Purdue. Accordingly, the Company recognized gross profit of $123.0 million in the three months ended December 31, 2009 as a result of the Distribution Agreement entered into with Purdue. This income was offset by an impairment charge of $43.1 million for MD&P assets recorded during the fiscal year March 31, 2010, related to our discontinued operations to reduce the carrying value of our MD&P assets for discontinued operations to their fair value.
Gain on sale in Discontinued Operations
| Years Ended March 31, | Change | |||||||||||||||
| ($ in millions): |
2010 | 2009 | $ | % | ||||||||||||
| Gain on sale in discontinued operations |
$ | 9.2 | $ | — | $ | 9.2 | NA | % | ||||||||
During fiscal year 2010, we completed the sale to Perrigo Company of our Paragraph IV ANDA for a generic equivalent version of GlaxoSmithKline’s Duac® gel. Under the terms of the transaction, we received $14.0 million from Perrigo Company at closing ($8.7 million, net of tax) and will receive an additional $2.0 million as a milestone payment upon the completion of a successful technical transfer. Also during fiscal year 2010, we recognized a $0.5 million gain from the sale of certain intellectual property and other assets associated with our ANDA for the generic version of Lotensin® 5-mg, 10-mg, 20-mg, and 40-mg Tablets to Huahai US, Inc.
The above gains on sale were recorded in discontinued operations as a result of our decision during the fourth quarter of fiscal year 2011 to divest the generics business.
Liquidity and Capital Resources
Cash and cash equivalents and working capital (deficiency) were $137.6 million and $(81.3 million), respectively, at March 31, 2011, compared to $60.7 million and ($81.1 million), respectively, at March 31, 2010. Working capital is defined as total current assets minus total current liabilities. The working capital deficiency decreased primarily due to an increase in cash and cash equivalents of $76.9 million, primarily resulting from the March 2011 private placement of Class A Common Stock and 2015 Notes, the reclassification to current assets of our ARS investments and increase in restricted cash of $34.5 million, and an increase in receivables of $32.4 million primarily related to initial sales of MakenaTM . These were offset by an increase in accrued liabilities of $190.2 million primarily related to warrants in connection with the loan from U.S. Healthcare and a liability recorded for future milestone payments due to Hologic for our purchase of the rights to MakenaTM which were offset by a decrease in accounts payable of $13.3 million which was related to timing of payments to our suppliers.
96
Table of Contents
For the fiscal year ended March 31, 2011, net cash used in operating activities of $159.5 million resulted primarily from our net loss offset by noncash items of $54.8 million primarily related to depreciation, gain on sale of assets, loss on debt extinguishments, gain on warrants and impairment of assets. Also contributing to the cash used in operating activities was an increase in receivables which was due to our initial sales of MakenaTM that occurred during the fourth quarter of fiscal year 2011 and decrease in accounts payable and accrued liabilities due to timing of payments.
For the fiscal year ended March 31, 2011, net cash flow provided by investing activities of $0.2 million included the $22.0 million related to the sale of PDI, net of fees and the amount held in escrow, $11.0 million cash proceeds pursuant to the sale of Sucralfate and $7.3 million proceeds from an investment in Strides. Additionally, the Company received approximately $3.5 million in insurance proceeds related to a fire that occurred in 2009 at PDI. This was offset by restricted cash related to a loan with our lenders of $7.5 million, one year of interest on deposit of $27.0 million related to our 2015 Notes, and a $12.5 payment to Hologic for MakenaTM product rights.
For the fiscal year ended March 31, 2011, net cash provided by financing activities of $236.2 million resulted primarily from proceeds of approximately $80.0 million received from U.S. Healthcare and $218.3 million, net of loan discount, from the 2015 Notes and private equity offering of $29.7 million. These increases were primarily offset by the payment and retirement of loans due to the U.S. Healthcare.
At March 31, 2011, our investment securities included $61.5 million in original principal amount of auction rate securities (“ARS”). Consistent with our investment policy guidelines, the ARS held by us are AAA-rated securities with long-term nominal maturities secured by student loans which are guaranteed by the U.S. Government. Liquidity for the ARS is typically provided by an auction process which allows holders to sell their notes and resets the applicable interest rate at pre-determined intervals, typically between seven to 35 days. However, with the liquidity issues experienced in global credit and capital markets, the ARS experienced failed auctions beginning in February 2008 and throughout fiscal years 2009 through 2011. An auction failure means that the parties wishing to sell their securities could not be matched with an adequate volume of buyers. The securities for which auctions have failed continue to accrue interest at the contractual rate and continue to be auctioned every seven, 14, 28 or 35 days, as the case may be, until the auction succeeds, the issuer calls the securities, or they mature. (See Note 6—“Investment Securities” of the Notes to Consolidated Financial Statements included in this Report for more information regarding the settlement agreement and the proceeds received in connection therewith.)
Our debt balance, including current maturities, was $510.4 million at March 31, 2011 compared to $297.1 million at March 31, 2010. The balances include a $52.4 million and $61.2 million collateralized obligation related to our ARS at March 31, 2011 and March 31, 2010, respectively.
In March 2006, we entered into a $43.0 million mortgage loan arrangement with LaSalle National Bank Association, in part to refinance $9.9 million of existing mortgages. The $32.8 million of net proceeds we received from the mortgage loan was used for working capital and general corporate purposes. The mortgage loan, which is secured by four of our buildings, bears interest at a rate of 5.91% (and a default rate of 10.905%) and matures on April 1, 2021. We were not in compliance with one or more of the requirements of the mortgage loan arrangement as of March 31, 2010. However, on August 5, 2010, we received a letter (“Waiver Letter”) approving certain waivers of covenants under the mortgage loan and certain other loan documents entered into in connection with the execution of the mortgage loan (collectively, the “Loan Documents”). In the Waiver Letter, the lenders consented to the following under the Loan Documents:
| • | Waiver of the requirement that we deliver audited balance sheets, statements of income and expenses and cash flows; |
| • | Waiver of the requirement that we certify financial statements delivered under the Loan Documents; |
97
Table of Contents
| • | Waiver of the requirement that we deliver to the lenders Form 10-Ks within 75 days of the close of the fiscal year, Form 10-Qs within 45 days of the close of each of the first three fiscal quarters of the fiscal year, and copies of all IRS tax returns and filings; and |
| • | Waiver, until March 31, 2012, of the requirement that we maintain a net worth, as calculated in accordance with the terms of the Loan Documents, of at least $250 million on a consolidated basis. |
With respect to the waiver of the requirement to deliver Form 10-Ks and Form 10-Qs, we agreed to bring our filings current effective with the submission of our Form 10-Q for the quarter ended December 31, 2010 and become timely on a go-forward basis with the filing of our Form 10-K for fiscal year 2011. Effective with the filing of this Form 10-K, the Company is in compliance with the SEC filing requirements contained in the waiver. However, based on current financial projections, the Company does not anticipate meeting the March 31, 2012 minimum net worth requirement. Accordingly, the mortgage is classified as a current liability as of March 31, 2011. If the Company is ultimately unable to meet the minimum net worth requirement by March 31, 2012, it will need to seek an additional waiver from the mortgage lender.
In May 2003, we issued $200.0 million principal amount of the 2033 Notes that are convertible, under certain circumstances, into shares of our Class A Common Stock at an initial conversion price of $23.01 per share. The 2033 Notes bear interest at a rate of 2.50% and mature on May 16, 2033. We are also obligated to pay contingent interest at a rate equal to 0.5% per annum during any six-month period commencing May 16, 2006, if the average trading price of the 2033 Notes per $1,000 principal amount for the five-trading day period ending on the third trading day immediately preceding the first day of the applicable six-month period equals $1,200 or more. We may redeem some or all of the 2033 Notes at any time on or after May 21, 2006, at a redemption price, payable in cash, of 100% of the principal amount of the 2033 Notes, plus accrued and unpaid interest (including contingent interest, if any) to the date of redemption. Holders may require us to repurchase all or a portion of their 2033 Notes on May 16, 2013, 2018, 2023 and 2028, or upon a change in control, as defined in the indenture governing the 2033 Notes, at 100% of the principal amount of the 2033 Notes, plus accrued and unpaid interest (including contingent interest, if any) to the date of repurchase, payable in cash.
In December 2005, we entered into a financing arrangement with St. Louis County, Missouri related to expansion of our operations in St. Louis County. Up to $135.5 million of industrial revenue bonds could have been issued to us by St. Louis County relative to capital improvements made through December 31, 2009. This agreement provides that 50% of the real and personal property taxes on up to $135.5 million of capital improvements will be abated for a period of ten years subsequent to the property being placed in service. Industrial revenue bonds totaling $129.9 million were outstanding at December 31, 2010 and March 31, 2011, respectively. The industrial revenue bonds are issued by St. Louis County to us upon our payment of qualifying costs of capital improvements, which are then leased by us for a period ending December 1, 2019, unless earlier terminated. We have the option at any time to discontinue the arrangement and regain full title to the abated property. The industrial revenue bonds bear interest at 4.25% per annum and are payable as to principal and interest concurrently with payments due under the terms of the lease. We have classified the leased assets as property and equipment and have established a capital lease obligation equal to the outstanding principal balance of the industrial revenue bonds. Lease payments may be made by tendering an equivalent portion of the industrial revenue bonds. As the capital lease payments to St. Louis County may be satisfied by tendering industrial revenue bonds (which is our intention), the capital lease obligation, industrial revenue bonds and related interest expense and interest income, respectively, have been offset for presentation purposes in the consolidated financial statements.
In September 2010 we entered into an agreement with U.S. Healthcare for a loan of $20 million which was subsequently retired in November 2010 when we entered into a new agreement with U.S. Healthcare for a senior secured debt financing package for up to $120 million which was subsequently amended in January 2011 and again in March 2011. In March 2011, the Company repaid in full all the remaining obligations with U.S. Healthcare and terminated the future loan commitments. (See Note 13—“Long-Term Debt” for a description of
98
Table of Contents
the financing with U.S. Healthcare and Note 21—“Equity Transactions” of the Notes to Consolidated Financial Statements in this Report for a description of our $32.3 million private placement of Class A Common Stock and private placement of $225 million aggregate principal amount of the 2015 Notes a portion of the proceeds of which were used to repay the loan obligations with U.S. Healthcare.)
On February 14, 2011, the Company announced that it entered into a definitive agreement with a group of institutional investors to raise approximately $32.3 of gross proceeds from a private placement of 9.95 million shares of its Class A Common Stock at $3.25 per share. The transaction closed on February 17, 2011. The Company used $20.0 of the proceeds from the financing to repay certain outstanding amounts and other outstanding obligations under its credit agreement with U.S. Healthcare. The remaining amount will be used for the launch of Makena™, payment of expenses associated with the transaction and general corporate purposes.
On March 17, 2011, the Company completed the offering and sale of the 2015 Notes. The 2015 Notes bear interest at an annual rate of 12% per year, payable semiannually in arrears on March 15 and September 15 of each year, commencing September 15, 2011. The 2015 Notes will mature March 15, 2015. After an original issue discount of 3%, the Company received proceeds of $218.3 million which were used to fund a first-year interest reserve totaling $27.0 million (reflected as restricted cash on the balance sheet), repay all existing obligations to U.S. Healthcare totaling approximately $61.1 million and pay fees and expenses associated with the offering of the 2015 Notes of approximately $10.0 million. In connection with these payments, the Company also terminated all future loan commitments with U.S. Healthcare. The remaining proceeds, totaling approximately $120.0 million will be used for general corporate purposes, including the launch of Makena™. The 2015 Notes were offered only to accredited investors pursuant to Regulation D under the Securities Act of 1933, as amended (See Note 13—“Long-Term Debt” for a description of the 2015 Notes of the Notes to Consolidated Financial Statements in this Report).
The following table summarizes our contractual obligations (in millions):
| Total | Less Than 1 Year |
2-3 Years |
4-5 Years |
More Than 5 Years |
||||||||||||||||
| Obligations at March 31, 2011 |
||||||||||||||||||||
| Long-term debt obligations(1) |
$ | 458.0 | $ | 33.0 | $ | 200.0 | $ | 225.0 | $ | — | ||||||||||
| Scheduled interest obligations(2) |
231.7 | 33.9 | 67.3 | 39.7 | 90.8 | |||||||||||||||
| Operating lease obligations |
3.5 | 1.3 | 1.3 | 0.7 | 0.2 | |||||||||||||||
| Hologic obligation(5) |
107.5 | 57.5 | 50.0 | — | — | |||||||||||||||
| DOJ obligations |
19.4 | 1.0 | 3.0 | 8.7 | 6.7 | |||||||||||||||
| Total contractual cash obligations(3)(4) |
$ | 820.1 | $ | 126.7 | $ | 321.6 | $ | 274.1 | $ | 97.7 | ||||||||||
| (1) | The next date holders of the 2033 Notes may require us to repurchase all or a portion of their Notes is May 16, 2013. On March 17, 2011, the Company completed the sale of the 2015 Notes. The 2015 Notes bear interest at an annual rate of 12% per year, payable semiannually in arrears on March 15 and September 15 of each year, commencing September 15, 2011. The 2015 Notes will mature March 15, 2015. |
| (2) | Scheduled interest payments represent the estimated interest payments on the building mortgages, the 2033 Notes and the 2015 Notes. |
| (3) | Excluded from the contractual obligations table is the liability for unrecognized tax benefits totaling $1.2 million. This liability for unrecognized tax benefits has been excluded because we cannot make a reliable estimate of the period in which the unrecognized tax benefits will be realized. |
| (4) | The terms of the Evamist® asset purchase agreement provide for two future payments upon achievement of certain net sales milestones. If Evamist®achieves $100.0 million of net sales in a fiscal year, a one-time payment of $10.0 million will be made, and if net sales levels reach $200.0 million in a fiscal year, a one-time payment of up to $20.0 million will be made. |
99
Table of Contents
| (5) | On February 3, 2011, the Company was informed by Hologic that the FDA granted approval for Makena™ and started shipping product in March 2011. Under the revised payment provisions set forth in Amendment No. 2, after the $12.5 million payment on the Transfer Date and a subsequent $12.5 million payment twelve months after the Approval Date, the Company has the right to elect between the following two alternate payment schedules for the remaining payments: |
Payment Schedule 1:
| • | A $45.0 million payment 18 months after the Approval Date, plus a royalty equal to 5% of net sales of Makena™ made during the period from 12 months after the Approval Date to the date the $45.0 million payment is made; |
| • | A $20.0 million payment 21 months after the Approval Date; |
| • | A $20.0 million payment 24 months after the Approval Date; and |
| • | A $10.0 million payment 27 months after the Approval Date. |
The royalties will continue to be calculated subsequent to the $45.0 million milestone payment but don’t have to be paid as long as the Company makes subsequent milestone payments when due.
Payment Schedule 2:
| • | A $7.3 million payment 18 months after the Approval Date, plus a royalty equal to 5% of net sales of Makena™ made during the period from 12 months after the Approval Date to 18 months after the Approval Date; |
| • | A $7.3 million payment for each of the succeeding twelve months; |
| • | A royalty payable 24 months following the Approval Date equal to 5% of net sales of Makena™ made during the period from 18 months after the Approval Date to 24 months after the Approval Date; and |
| • | A royalty payable 30 months following the Approval Date equal to 5% of net sales of Makena™ made during the period from 24 months after the Approval Date to 30 months after the Approval Date. |
Notwithstanding anything to the contrary in Amendment No. 2, however, the Company may make any of the foregoing payments on or before their due dates, and the date on which the Company makes the final payment contemplated by the selected payment schedule will be the final payment date, after which no royalties will accrue
Moreover, if the Company elects Payment Schedule 1 and thereafter elects to pay the $45.0 million payment earlier than the 18-month deadline, the royalties beginning after 12 months will cease to accrue on the date of the early payment. Additionally, the subsequent payments will be paid in three months intervals following the $45.0 million payment date.
Lastly, if the Company elects Payment Schedule 1 and thereafter does not make any of the milestone payments when due, Amendment No. 2 provides that no payment default will be deemed to occur, provided the Company timely pays the required royalties accruing in the quarter during which the milestone payment has become due but is not paid.
Pursuant to the Indenture governing the $225 million aggregate principal amount of the 2015 Notes, the Company agreed to make the $45.0 million payment under Payment Schedule 1 in February 2012 and agreed to certain other restrictions on its ability to amend the payment schedules.
100
Table of Contents
Ability to Continue as a Going Concern
There is substantial doubt about our ability to continue as a going concern. Our Consolidated Financial Statements included in this Report are prepared using accounting principles generally accepted in the United States applicable to a going concern, which contemplates the realization of assets and liquidation of liabilities in the normal course of business. The historical consolidated financial statements included in this Report do not include any adjustments that might be necessary if we are unable to continue as a going concern. The report of our independent registered public accountants BDO USA, LLP, included in this Report, includes an explanatory paragraph related to our ability to continue as a going concern.
The assessment of our ability to continue as a going concern was made by management considering, among other factors: (1) the timing and number of approved products that will be reintroduced to the market and the related costs; (2) the suspension of shipment of all products manufactured by us and the requirements under the consent decree with the FDA; (3) the possibility that we may need to obtain additional capital despite the proceeds from the offering of the 2015 Notes in March 2011 and the equity we were able to issue in February 2011 (see Note 1—“Description of Business—Significant Developments” of the Notes to the Consolidated Financial Statements included in this Report); (4) the potential outcome with respect to the governmental inquiries, litigation or other matters described in Note 15—“Commitments and Contingencies” of the Notes to the Consolidated Financial Statements included in this Report; (5) our ability to comply with debt covenants and (6) our ability to obtain future revenues from the sales of Makena™ sufficient to meet our future needs and expectations. Our assessment was further affected by our fiscal year 2011 net loss of $174.0 million, our 2010 net loss of $283.6 million, and the outstanding balance of cash and cash equivalents of $137.6 million as of March 31, 2011. For periods subsequent to March 31, 2011, we expect losses to continue because we are unable to generate any significant revenues from more of our own manufactured products until we are able to begin selling more of our approved products with FDA’S approval and with respect to products manufactured for us by third parties until after we are able to generate significant sales of Makena™ which was approved by the FDA in February 2011. We received notification from the FDA on September 8, 2010 of approval for our discontinued generics operations to ship into the marketplace the first product approved under the consent decree, i.e., the Potassium Chloride Extended Release Capsule. We resumed shipment of extended-release potassium chloride capsule, Micro-K® 10mEq and Micro-K® 8mEq, in September 2010, resumed shipments of the generic version of Potassium Chloride Extended Release Capsule in December 2010 and we began shipping Makena™ in March 2011. We have continued to ship Evamist®, which is manufactured for the Company by a third party, during the period covered by the consent decree. We are continuing to prepare other products for FDA inspection and do not expect to resume shipping other products until prior to the end of calendar year 2011, at the earliest. In addition, we must meet ongoing operating costs as well as costs related to the steps we are currently taking to introduce Makena™, and to prepare for introducing and reintroducing other approved products to the market. If we are not able to obtain the FDA’s clearance to resume manufacturing and distribution of more of our approved products in a timely manner and at a reasonable cost, or if revenues from the sale of approved products introduced or reintroduced into the market place prove to be insufficient, our financial position, results of operations, cash flows and liquidity will continue to be materially adversely affected. These conditions raise substantial doubt about our ability to continue as a going concern.
Based on current financial projections, we believe the continuation of our Company as a going concern is primarily dependent on our ability to address, among other factors: (1) sales of Makena™, which was approved by the FDA in February 2011 notwithstanding recent actions by the FDA to permit continued sale of compounded alternatives and by CMS to permit compounded alternatives to be reimbursed under the Medicaid program; (2) the timing, number and revenue generation of approved products that will be introduced or reintroduced to the market and the related costs; (3) the suspension of shipment of all products manufactured by us and the requirements under the consent decree with the FDA (other than the Potassium Chloride Extended Release Capsule, including Micro-K® 10mEq and Micro-K® 8mEq, products that are the subject of the FDA notification letter discussed above); (4) the possibility that we will need to obtain additional capital; (5) the potential outcome with respect to the governmental inquiries, litigation or other matters described in Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report;
101
Table of Contents
and (6) compliance with our debt covenants. While we address these matters, we must continue to meet expected near-term obligations, including normal course operating cash requirements and costs associated with introducing or reintroducing approved products to the market (such as costs related to our employees, facilities and FDA compliance), remaining payments associated with the acquisition and retention of the rights to Makena™ (see Note 5—“Acquisition” of the Notes to Consolidated Financial Statements included in this Report), the financial obligations pursuant to the plea agreement, costs associated with our legal counsel and consultant fees, as well as the significant costs associated with the steps taken by us in connection with the consent decree and the litigation and governmental inquiries. If we are not able to obtain the FDA’s clearance to resume manufacturing and distribution of certain or many of our approved products in a timely manner and at a reasonable cost and/or if we are unable to successfully commercialize Makena™, and/or if we experience adverse outcomes with respect to any of the governmental inquiries or litigation described in Note 15—“Commitments and Contingencies” of the Notes to Consolidated Financial Statements included in this Report, our financial position, results of operations, cash flows and liquidity will continue to be materially adversely affected. See “Risk Factors” included in Item I, Part 1A of this Report regarding additional risks we face with respect to these matters.
In the near term, we are focused on performing the following: (1) addressing the FDA’s concerns regarding patient access to MakenaTM and CMS policy permitting Medicaid program reimbursement of compounded products; (2) the continued commercial launch of Makena™; (3) meeting the requirements of the consent decree, which will allow our approved products to be reintroduced to the market (other than the Potassium Chloride Extended Release Capsule products, including Micro-K® 10mEq and Micro-K® 8mEq, that are the subject of the FDA notification letter previously discussed); (4) the divestiture of Nesher and the Company’s generics business; and (5) pursuing various means to minimize operating costs and increase cash. Since December 31, 2010, the Company has generated non-recurring cash proceeds to support its on-going operating and compliance requirements from a $32.3 million private placement of Class A Common Stock in February 2011 and the 2015 Notes in March 2011 (which were used, in part, to repay all existing obligations under the agreement with U.S. Healthcare) (see Note 13—“Long-Term Debt” of the Notes to Consolidated Financial Statements included in this Report for a description of the 2015 Notes and U.S. Healthcare loan). While the cash proceeds received to date were sufficient to meet near-term cash requirements, we are pursuing ongoing efforts to increase cash, including, the continued implementation of cost savings, the divestiture of Nesher and the Company’s generics business and other assets and the return of certain additional approved products to market in a timely manner. We cannot provide assurance that we will be able to realize additional cost reductions from reducing our operations, that some or many of our approved products can be returned to the market in a timely manner (other than the Potassium Chloride Extended Release Capsule, including Micro-K® 10mEq and Micro-K® 8mEq products that are the subject of the FDA notification letter previously discussed above), that our higher profit approved products will return to the market in the near term or at all or that we can obtain additional cash through asset sales or the sale of equity or the successful commercial launch of Makena™. If we are unsuccessful in our efforts to introduce or return our products to market at adequate levels, or to sell assets or raise additional equity, we may be required to further reduce our operations, including further reductions of our employee base, or we may be required to cease certain or all of our operations in order to offset the lack of available funding.
We continue to evaluate the sale of certain of our assets, including Nesher and our generics business, which is included in discontinued operations and assets. To date, we are in advanced discussions for the sale of our generics business, however, we cannot assure that we will complete any transaction on favorable terms or at all. The Company is continuing to work with its advisors and several interested parties to complete a transaction. However, due to general economic conditions, we will likely be exposed to risks related to the overall macro-economic environment, including a lower rate of return than we have historically experienced on our invested assets and being limited in our ability to sell assets. In addition, we cannot provide any assurance that we ultimately will be successful in finding suitable purchasers for the sale of such assets. Even if we are able to find purchasers, we may not be able to obtain attractive terms and conditions for such sales, including attractive pricing. In addition, divestitures of businesses involve a number of risks, including the diversion of management and employee attention, significant costs and expenses, the loss of customer relationships, a decrease in revenues
102
Table of Contents
and earnings associated with the divested business, and the disruption of operations in the affected business. In addition, divestitures potentially involve significant post-closing separation activities, which could involve the expenditure of significant financial and employee resources. Inability to consummate identified asset sales or manage the post-separation transition arrangements could adversely affect our business, financial condition, results of operations and cash flows.
Current and Anticipated Liquidity Position
At March 31, 2011, we had approximately $138 million in cash and cash equivalents. During the quarter ended March 31, 2011 we had total cash inflows of approximately $272 million resulting primarily from the proceeds of a $32 million private placement of our Class A common stock and the private placement of $225 million aggregate principal amount of 2015 Notes. The remainder of the cash inflows was primarily attributable to cash collections from our customers, mostly related to sales of MakenaTM that occurred in March 2011. Our cash expenditures during the quarter ended March 31, 2011 were approximately $166 million which included approximately $27 million for on-going operating expenses, $2 million for debt service payments, $11 million in fees and expenses associated with the private placement of Class A Common Stock and the 2015 Notes, and $5 million for costs related to our FDA consultants and other compliance related costs. Also, in February 2011 we made a $12.5 million milestone payment to Hologic, following FDA approval of MakenaTM. Finally, we used approximately $73.4 million during the quarter ended March 31, 2011 to repay in full our outstanding obligations to U.S. Healthcare, $27.0 million to establish a first year interest reserve on the 2015 Notes and $7.5 million to establish an escrow balance that may be payable to U.S. Healthcare if certain conditions related to the registration of the warrants that were issued to them and the trading price of our Class A common stock are not met.
We currently project that our cash balance at June 30, 2011 will be in the range of $100 million to $110 million. We currently estimate that during the quarter ending June 30, 2011, we will generate cash of $25 million to $30 million from the collection of customer receivables and the monetization of certain assets. Barring any actions we may take to reduce our operating expenses, we currently estimate that during the quarter ending June 30, 2011 our cash expenditures for operating costs will be approximately $30 million to $35 million for on-going operating expenses, approximately $3 million to $4 million for debt service payments, approximately $1 million to $2 million related to legal payments and approximately $5 million to $6 million for costs related to our FDA compliance and other compliance-related costs. Of the costs described above for on-going operating expenses, legal payments and FDA compliance and other compliance-related costs, we estimate that approximately 25% relates to our generics business and the remainder to our branded business and corporate-related costs. In addition, we expect that our cash expenditures in the quarter ending June 30, 2011 will include customer allowances of approximately $24 million to $27 million, which result primarily from compensating our customers for the 54% decrease in the list price of MakenaTM after the product was first shipped to them in March 2011.
We expect that our cash operating outlays, excluding milestone payments to Hologic (described above), scheduled payments to the Department of Justice, debt repayments, and customer allowances, will continue in the ranges noted above until we are able to divest the generics business. Our future cash inflows will be generated primarily from collection of customer receivables. The majority of our cash inflow from customer collections for periods beyond March 31, 2011 is expected to be derived from sales of Makena™ and from on-going sales of Evamist® and sales of both the branded and generic versions of the Potassium Chloride Extended Release Capsule. We also expect to return Clindesse® and Gynazole-1® to the market during the second half of fiscal year 2012. However, we are currently unable to estimate the amount or timing of collections from sales of our products for periods beyond June 30, 2011.
At March 31, 2011, we had $503.7 million of outstanding debt, net of loan discounts, consisting of $218.3 million of 2015 Notes, $200.0 million principal amount of convertible notes, $52.4 million of collateralized borrowing related to ARS and $33.0 million remaining principal balance of a mortgage loan.
103
Table of Contents
On February 14, 2011, the Company announced that it entered into a definitive agreement with a group of institutional investors to raise approximately $32.3 million of gross proceeds from a private placement of 9.95 million shares of its Class A Common Stock at $3.25 per share. The transaction closed on February 17, 2011. The Company used $20.0 million of the proceeds from the financing to repay certain outstanding amounts and other outstanding obligations under its credit agreement with U.S. Healthcare. The remaining amount will be used for the launch of Makena™ and payment of expenses associated with the private placement and general corporate purposes.
On March 17, 2011, we completed the 2015 Notes Offering that generated approximately $218 million of net proceeds (see Note 13—“Long-term Debt” of the Notes to Consolidated Financial Statements included in this Report). A portion of the proceeds from the 2015 Notes were used to repay existing obligations to U.S. Healthcare of approximately $61 million (which amount includes an applicable make-whole premium), establish a one year interest reserve for the 2015 Notes totaling $27 million, and pay fees and expenses associated with the 2015 Notes of approximately $10 million. In connection with these payments, the Company also terminated all future loan commitments with U.S. Healthcare. Net cash provided to the Company from the 2015 Notes Offering, after payment of the items noted above, was approximately $120 million. The 2015 Notes were offered only to accredited investors pursuant to Regulation D under the Securities Act of 1933, as amended.
On February 4, 2011, the Company entered into Amendment No. 2 to the Original Makena™ Agreement (See Note 5—“Acquisition” of the Notes to Consolidated Financial Statements in this Report).
The Amendment set forth in Amendment No. 2 reduced the payment to be made on the Transfer Date to $12.5 million and revised the schedule for making the remaining $107.5 million.
Under the revised payment provisions set forth in Amendment No. 2, after the $12.5 million payment on the Transfer Date and a subsequent $12.5 million payment twelve months after the Approval Date, the Company has the right to elect between the following two alternate payment schedules for the remaining payments:
Payment Schedule 1:
| • | A $45.0 million payment 18 months after the Approval Date, plus a royalty equal to 5% of net sales of Makena™ made during the period from 12 months after the Approval Date to the date the $45.0 million payment is made; |
| • | A $20.0 million payment 21 months after the Approval Date; |
| • | A $20.0 million payment 24 months after the Approval Date; and |
| • | A $10.0 million payment 27 months after the Approval Date. |
The royalties will continue to be calculated subsequent to the $45.0 million milestone payment but don’t have to be paid as long as the Company makes subsequent milestone payments when due.
Payment Schedule 2:
| • | A $7.3 million payment 18 months after the Approval Date, plus a royalty equal to 5% of net sales of Makena™ made during the period from 12 months after the Approval Date to 18 months after the Approval Date; |
| • | A $7.3 million payment for each of the succeeding twelve months; |
| • | A royalty payable 24 months following the Approval Date equal to 5% of net sales of Makena™ made during the period from 18 months after the Approval Date to 24 months after the Approval Date; and |
| • | A royalty payable 30 months following the Approval Date equal to 5% of net sales of Makena™ made during the period from 24 months after the Approval Date to 30 months after the Approval Date. |
104
Table of Contents
Notwithstanding anything to the contrary in Amendment No. 2, however, the Company may make any of the foregoing payments on or before their due dates, and the date on which the Company makes the final payment contemplated by the selected payment schedule will be the final payment date, after which no royalties will accrue
Moreover, if the Company elects Payment Schedule 1 and thereafter elects to pay the $45.0 million payment earlier than the 18-month deadline, the royalties beginning after 12 months will cease to accrue on the date of the early payment. Additionally, the subsequent payments will be paid in three months intervals following the $45.0 million payment date.
Lastly, if the Company elects Payment Schedule 1 and thereafter does not make any of the milestone payments when due, Amendment No. 2 provides that no payment default will be deemed to occur, provided the Company timely pays the required royalties accruing in the quarter during which the milestone payment has become due but is not paid.
We are continuously reviewing our projected cash expenditures and are evaluating measures to reduce expenditures on an ongoing basis. In addition, a top priority is to maintain and attempt to increase our limited cash and financial resources. As a result, if we determine that our current goal of meeting the consent decree requirements and returning our approved products to market is likely to be significantly delayed, we may decide, in addition to selling certain of our assets, to further reduce our operations, to significantly curtail some or all of our efforts to meet the consent decree requirements and return our approved products to market and/or to outsource to a third-party some or all of our manufacturing operations when and if we return our approved products to market. Such decision would be made based on our ability to manage our near-term cash obligations, to obtain additional capital through asset sales and/or external financing and to expeditiously meet the consent decree requirements and return our approved products to market. If such decision were to be made, we currently anticipate that we would focus our management efforts on developing product candidates in our development portfolio that we believe have the highest potential return on investment, which we currently believe to be primarily Makena™. We also expect to evaluate other alternatives available to us in order to increase our cash balance.
Off-Balance Sheet Arrangements
We do not maintain any off-balance sheet financing arrangements, other than a financing arrangement with St. Louis County, Missouri related to the expansion of our operations in St. Louis County (see Note 14—“Taxable Industrial Revenue Bonds” of the Notes to Consolidated Financial Statements included in this Report).
Inflation
Inflation may apply upward pressure on the cost of goods and services used by us in the future. However, we believe that the net effect of inflation on our operations during the past three years has been minimal. In addition, changes in the mix of products sold and the effects of competition have made a comparison of changes in selling prices less meaningful relative to changes in the overall rate of inflation over the past three fiscal years.
Critical Accounting Estimates
Our Consolidated Financial Statements are presented on the basis of U.S. generally accepted accounting principles. Certain of our accounting policies are particularly important to the presentation of our financial condition and results of operations and require the application of significant judgment by our management. As a result, amounts determined under these policies are subject to an inherent degree of uncertainty. In applying these policies, we make estimates and judgments that affect the reported amounts of assets, liabilities, revenues and expenses and related disclosures. We base our estimates and judgments on historical experience, the terms of existing contracts, observance of trends in the industry, information that is obtained from customers and outside
105
Table of Contents
sources, and on various other assumptions that we believe to be reasonable and appropriate under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Although we believe that our estimates and assumptions are reasonable, actual results may differ significantly from our estimates. Changes in estimates and assumptions based upon actual results may have a material impact on our results of operations and/or financial condition. Our critical accounting estimates are described below.
Revenue Recognition and Provisions for Estimated Reductions to Gross Revenues
Revenue is generally realized or realizable and earned when persuasive evidence of an arrangement exists, the customer’s payment ability has been reasonably assured, title and risk of ownership have been transferred to the customer, and the seller’s price to the buyer is fixed or reasonably determinable. When these conditions have not been met, revenue is deferred and reflected as a liability on the consolidated balance sheet until any uncertainties or contingencies have been resolved. We also enter into long-term agreements under which we assign marketing rights for products we have developed to pharmaceutical markets. Royalties under these arrangements are earned based on the sale of products.
Concurrent with the recognition or deferral of revenue, we record estimated provisions for product returns, sales rebates, payment discounts, chargebacks, and other sales allowances. These provisions are established based upon consideration of a variety of factors, including but not limited to, historical relationship to revenues, historical payment and return experience, estimated and actual customer inventory levels, customer rebate arrangements, and current contract sales terms with wholesale and indirect customers. We record a liability for product returns related to recalls and for failure to supply claims when their occurrence becomes estimable and probable.
From time to time, we provide incentives to our wholesale customers, such as trade show allowances or stocking allowances that they in turn use to accelerate distribution to their end customers. We believe that such incentives are normal and customary in the industry. Sales allowances are accrued and revenue is recognized as sales are made pursuant to the terms of the allowances offered to the customer. Due to the nature of these allowances, we believe we are able to accurately calculate the required provisions for the allowances based on the specific terms in each agreement. Additionally, customers will normally purchase additional product ahead of regular demand to take advantage of the temporarily lower cost resulting from the sales allowances. This practice has been customary in the industry and we believe would be part of a customer’s ordinary course of business inventory level. We reserve the right, with our major wholesale customers, to limit the amount of these forward buys. In addition, we understand that certain of our wholesale customers may try to anticipate the timing of price increases and have made, and may continue to make, business decisions to buy additional product in anticipation of future price increases. This practice has been customary in the industry and we believe would be part of a customer’s ordinary course of business inventory level.
We evaluate inventory levels at our wholesale customers through an internal analysis that considers, among other things, wholesaler purchases, wholesaler contract sales, available end consumer prescription information and inventory data received from our three largest wholesale customers. We believe that our evaluation of wholesaler inventory levels allows us to make reasonable estimates of our reserve balances. Further, our products are typically sold with adequate shelf life to permit sufficient time for our wholesaler customers to sell our products in their inventory through to the end consumer.
106
Table of Contents
The following table reflects the fiscal year 2011 activity for each accounts receivable reserve (in thousands):
| Beginning Balance |
Current Provision Related to Sales Made in the Current Period |
Current Provision Related to Sales Made in the Prior Periods |
Actual Returns or Credits in the Current Period |
Ending Balance |
||||||||||||||||
| Year Ended March 31, 2011 |
||||||||||||||||||||
| Accounts Receivable Reserves: |
||||||||||||||||||||
| Chargebacks |
$ | — | $ | 3.3 | $ | — | $ | (0.4 | ) | $ | 2.9 | |||||||||
| Cash Discounts and Other Allowances |
0.1 | 1.3 | — | (0.4 | ) | 1.0 | ||||||||||||||
| Liabilities: |
||||||||||||||||||||
| Sales Rebates |
1.0 | 2.2 | — | (2.1 | ) | 1.1 | ||||||||||||||
| Sales Returns |
7.3 | 2.1 | — | (4.6 | ) | 4.8 | ||||||||||||||
| Medicaid Rebates |
3.6 | 3.3 | — | — | 6.9 | |||||||||||||||
| Product Recall Returns |
2.9 | 0.2 | — | — | 3.1 | |||||||||||||||
| Failure to Supply Claims |
12.3 | — | — | (2.3 | ) | 10.0 | ||||||||||||||
| Price protection |
— | 26.0 | — | (0.2 | ) | 25.8 | ||||||||||||||
| Other |
0.3 | 0.7 | — | (0.3 | ) | 0.7 | ||||||||||||||
| Total |
$ | 27.5 | 39.1 | $ | — | (10.3 | ) | 56.3 | ||||||||||||
The estimation process used to determine our reserve provisions has been applied on a consistent basis and no material adjustments have been necessary to increase or decrease our reserves as a result of a significant change in underlying estimates. We use a variety of methods to assess the adequacy of our reserves to ensure our financial statements are fairly stated. These include reviews of customer inventory data, customer contract programs and product pricing trends to analyze and validate the reserves. The increase in the accounts receivable and accrued liability reserves of $28.8 million was primarily the result of increased price protection reserve associated with the price reduction of Makena™. The list price was decreased from $1,500 per injection to $690 per injection.
The following table reflects the fiscal year 2010 activity for each accounts receivable reserve:
| Beginning Balance |
Current Provision Related to Sales Made in the Current Period |
Current Provision Related to Sales Made in the Prior Periods |
Actual Returns or Credits in the Current Period |
Ending Balance |
||||||||||||||||
| Year Ended March 31, 2010 |
||||||||||||||||||||
| Accounts Receivable Reserves: |
||||||||||||||||||||
| Chargebacks |
$ | — | $ | 0.1 | $ | — | $ | (0.1 | ) | $ | — | |||||||||
| Cash Discounts and Other Allowances |
0.2 | 0.2 | — | (0.3 | ) | 0.1 | ||||||||||||||
| Liabilities: |
||||||||||||||||||||
| Sales Rebates |
4.6 | 1.2 | — | (4.8 | ) | 1.0 | ||||||||||||||
| Sales Returns |
12.0 | 3.7 | — | (8.4 | ) | 7.3 | ||||||||||||||
| Medicaid Rebates |
6.3 | 0.7 | — | (3.4 | ) | 3.6 | ||||||||||||||
| Medicare and Medicaid restitution |
2.3 | — | — | (2.3 | ) | — | ||||||||||||||
| Product Recall Returns |
40.7 | — | — | (37.8 | ) | 2.9 | ||||||||||||||
| Failure to Supply Claims |
17.1 | — | — | (4.8 | ) | 12.3 | ||||||||||||||
| Other |
2.6 | 0.3 | — | (2.6 | ) | 0.3 | ||||||||||||||
| Total |
$ | 85.8 | $ | 6.2 | $ | — | $ | (64.5 | ) | $ | 27.5 | |||||||||
107
Table of Contents
We use a variety of methods to assess the adequacy of our reserves to ensure our financial statements are fairly stated. These include reviews of customer inventory data, customer contract programs and product pricing trends to analyze and validate the reserves.
The decrease in the accounts receivable and accrued liability reserves of $58.3 million was primarily the result of applying credits owed to our customers for returns and failure to supply costs associated with the product recalls in fiscal year 2009 against outstanding amounts such customers owed to us and of making payments associated with return and failure to supply costs to customers that did not have outstanding amounts owed to us.
The current provision related to sales rebates made in prior periods reflects the impact of a billing audit performed by one of our customers. As a result, we increased our chargeback reserve for the amount of the adjustment shown in the fiscal year ended March 31, 2010.
These reserves and their respective provisions are discussed in further detail below.
Chargebacks—We market and sell products directly to wholesalers, distributors, warehousing pharmacy chains, mail order pharmacies and other direct purchasing groups. We also market products indirectly to independent pharmacies, non-warehousing chains, managed care organizations, and group purchasing organizations, collectively referred to as “indirect customers.” We enter into agreements with some indirect customers to establish contract pricing for certain products. These indirect customers then independently select a wholesaler from which to purchase the products at these contracted prices. Alternatively, we may pre-authorize wholesalers to offer specified contract pricing to other indirect customers. Under either arrangement, we provide credit to the wholesaler for any difference between the contracted price with the indirect customer and the wholesaler’s invoice price. This credit is called a chargeback.
At March 31, 2011, we evaluated the adequacy of the reserve for chargebacks by comparing it against subsequent credits issued.
Shelf-Stock Adjustments—These adjustments, which are included in the chargeback reserves, represent credits issued to our wholesale customers that result from a decrease in our WAC. Decreases in our invoice prices are discretionary decisions we make to reflect market conditions. These credits are customary in the industry and are intended to reduce a wholesale customer’s inventory cost to better reflect current market prices. Generally, we provide credits to customers at the time the price reduction occurs based on the inventory that is owned by them on the effective date of the price reduction. Since a reduction in WAC reduces the chargeback per unit, or the difference between WAC and the contract price, shelf-stock adjustments are typically included as part of the reserve for chargebacks because the price reduction credits act essentially as accelerated chargebacks. Although we have contractually agreed to provide price adjustment credits to our major wholesale customers at the time they occur, the impact of any such price reductions not included in the reserve for chargebacks is immaterial to the amount of revenue recognized in any given period. Sales Returns—Consistent with industry practice, we maintain a returns policy that allows our direct and indirect customers to return product six months prior to expiration and within one year after expiration. This policy is applicable to both our branded and specialty generics business segments. Upon recognition of revenue from product sales to customers, we provide for an estimate of product to be returned. This estimate is determined by applying a historical relationship of customer returns to gross sales. We evaluate the reserve for sales returns by calculating historical return rates using data from the last 12 months on a product specific basis and by class of trade (wholesale versus retail chain). The calculated percentages are applied against estimates of inventory in the distribution channel on a product specific basis. To determine the inventory levels in the wholesale distribution channel, we utilize actual inventory information from our major wholesale customers and estimate the inventory positions of the remaining wholesalers based on historical buying patterns. For inventory held by our non-wholesale customers, we use the last two months of sales to the direct buying chains and the indirect buying retailers as an estimate.
108
Table of Contents
Medicaid Rebates—Federal law requires that a pharmaceutical manufacturer, as a condition of having its products receive federal reimbursement under Medicaid and Medicare Part B, must pay rebates to state Medicaid programs for units of its pharmaceuticals that are dispensed to Medicaid beneficiaries and paid for by a state Medicaid program. Medicaid rebates apply to both our branded and specialty generic/non-branded segments. Individual states invoice us for Medicaid rebates on a quarterly basis using statutorily determined rates: for non-innovator products, in general generic drugs marketed under ANDAs, the rebate amount, effective the first quarter of calendar year 2010, is 13% of the AMP for the quarter. Prior to the first quarter of calendar year 2010, the percentage was 11%. For innovator products, in general brand-name products marketed under NDAs, the rebate amount, effective the first quarter of calendar year 2010, is the greater of 23.1% of the AMP for the quarter (15.1% prior to the first quarter of calendar year 2010) or the difference between such AMP and the best price for that same quarter. This 23.1% is reduced to 17.1% in the case of certain clotting factors and drugs approved exclusively for pediatric indications. An additional rebate for innovator products is payable in the amount by which, if any, the product’s AMP has increased at a rate faster than inflation. For certain new formulations of existing products, the new formulation’s additional rebate may be based on the additional rebate of the original formulation of the drug. The total rebate amount for each particular product is its Unit Rebate Amount, or “URA.” The amount owed is based on the number of units paid for by each state Medicaid program in a quarter extended by the URA. Historically, these units were limited to those paid for by each state Medicaid program under fee-for-service arrangements, but effective March 23, 2010 the utilization was expanded to include that paid for under Medicaid managed care capitated arrangements. The reserve for Medicaid rebates is based on expected payments, which are affected by patient usage and estimated inventory in the distribution channel. We estimate patient usage by calculating a payment rate as a percentage of related gross sales, which is then applied to current period sales.
Reserve for Failure to Supply Claims—We have historically entered into product purchase arrangements with certain customers that include a provision that requires us to reimburse these customers for price differences on product orders that we are unable to fulfill. We are able to estimate provisions for supply failures based on the specific terms in each arrangement. We incurred failure to supply claims in fiscal year 2009 due primarily to us not manufacturing or shipping any of our drug products during the third and fourth quarters of fiscal year 2009. There were no increases to the reserve for failure to supply claims recorded in fiscal year 2010.
Liabilities for Product Returns Related to Recalls—Beginning in May 2008 through November 2008, we announced six separate voluntary recalls of certain tablet form generic products as a precaution due to the potential existence of oversized tablets. Liabilities for product returns related to recalls are based on estimated and actual customer inventory levels at the time of the recall and actual contract pricing.
Inventory Valuation
Inventories consist of finished goods held for distribution, raw materials and work in process. Our inventories are stated at the lower of cost or market, with cost determined on the first-in, first-out basis. In evaluating whether inventory should be stated at the lower of cost or market, we consider such factors as the amount of inventory on hand and in the distribution channel, estimated time required to sell existing inventory, remaining shelf life and current and expected market conditions, including levels of competition. We establish reserves, when necessary, for slow-moving, excess and obsolete inventories based upon our historical experience and management’s assessment of current product demand.
Inventories also include costs related to certain products that are pending regulatory approval. From time to time, we capitalize inventory costs associated with products prior to regulatory approval based on management’s judgment of probable future regulatory approval, commercial success and realizable value. Such judgment incorporates our knowledge and best estimate of where the product is in the regulatory review process, our required investment in the product, market conditions, competing products and our economic expectations for the product post-approval relative to the risk of manufacturing the product prior to approval. If final regulatory
109
Table of Contents
approval for such products is denied or delayed, we revise our estimates and judgments about the recoverability of the capitalized costs and, where required, provide reserves for such inventory in the period those estimates and judgments change.
During fiscal year 2009, we announced six separate voluntary recalls of certain tablet-form generic products as a precaution due to the potential existence of oversized tablets. Beginning in December 2008, we determined that we were not able to establish the recoverability of production related inventory costs because of uncertainties associated with the risk of additional product recalls. As a result, production and overhead costs were recognized directly into cost of sales rather than capitalized into inventory.
Intangible and Other Long-Lived Assets
Our intangible assets principally consist of product rights, license agreements and trademarks resulting from product acquisitions and legal fees and similar costs relating to the development of patents and trademarks. Intangible assets that are acquired are stated at cost, less accumulated amortization, and are amortized on a straight-line basis over their estimated useful lives, which range from nine to 20 years. We determine amortization periods for intangible assets that are acquired based on our assessment of various factors impacting estimated useful lives and cash flows of the acquired products. Such factors include the product’s position in its life cycle, the existence or absence of like products in the market, various other competitive and regulatory issues, and contractual terms. Significant changes to any of these factors may result in a reduction in the intangible asset’s useful life and an acceleration of related amortization expense.
We assess the impairment of intangible and other long-lived assets whenever events or changes in circumstances indicate that the carrying value may not be recoverable. Factors we consider important which could trigger an impairment review include the following: (1) significant underperformance relative to expected historical or projected future operating results; (2) significant changes in the manner of our use of the acquired assets or the strategy for our overall business; and (3) significant negative industry or economic trends.
When we determine that the carrying value of an intangible or other long-lived asset may not be recoverable based upon the existence of one or more of the above indicators of impairment, we first perform an assessment of the asset’s recoverability. Recoverability is determined by comparing the carrying amount of an asset against an estimate of the undiscounted future cash flows expected to result from its use and eventual disposition. If the sum of the expected future undiscounted cash flows is less than the carrying amount of the asset, an impairment loss is recognized based on the excess of the carrying amount over the estimated fair value of the asset.
During the assessment as of March 31, 2011, management did identify events that were indicative of impairment. The Company recorded an impairment charge of $10.0 million in discontinued operations. (See Note 4—“Restructuring and Impairment” for further information regarding the assessment of long-lived assets for impairment in the Notes to Consolidated Financial Statements included in this Report)
Stock-Based Compensation
We account for stock-based compensation expense in accordance with the applicable FASB authoritative guidance, which requires the measurement and recognition of compensation expense, based on estimated fair values, for all share-based compensation awards made to employees and directors over the vesting period of the awards. Determining the fair value of share-based awards at the grant date requires judgment to identify the appropriate valuation model and estimate the assumptions, including the expected term of the stock options and expected stock-price volatility, to be used in the calculation. Judgment is also required in estimating the percentage of share-based awards that are expected to be forfeited. We estimate the fair value of stock options granted using the Black-Scholes option-pricing model with assumptions based primarily on historical data. If actual results differ significantly from these estimates, stock-based compensation expense and our results of operations could be materially impacted.
110
Table of Contents
Income Taxes
Our deferred tax assets and liabilities are determined based on temporary differences between the financial reporting and tax basis of assets and liabilities and tax credit carry forwards. Deferred tax assets and liabilities are measured using the currently enacted tax rates that apply to taxable income in effect for the years in which those tax assets are expected to be realized or settled. We record a valuation allowance to reduce deferred tax assets to the amount that is believed more likely than not to be realized. If all or part of the net deferred tax assets are determined not to be realizable in the future, an adjustment to the valuation allowance would be charged to earnings in the period such determination is made. Similarly, if we subsequently realize deferred tax assets that were previously determined to be unrealizable, the respective valuation allowance would be reversed, resulting in a positive adjustment to earnings in the period such determination is made.
We compute our annual tax rate based on the statutory tax rates and tax planning opportunities available to us in the various jurisdictions in which we earn income. Significant judgment is required in determining our annual tax rate and in evaluating tax positions. We establish reserves for uncertain tax positions if the positions are more likely than not to be sustained upon audit. We adjust these reserves in light of changing facts and circumstances, such as the settlement of a tax audit. Our annual tax rate includes the impact of reserve provisions and changes to reserves.
Management regularly evaluates our tax positions taken on tax returns that we file using information about recent court decisions and legislative activities. Many factors are considered in making these evaluations, including past history, recent interpretations of tax law, and the specific facts and circumstances of each matter. Because tax regulations are subject to interpretation and tax litigation is inherently uncertain, these evaluations can involve a series of complex judgments about future events and can rely heavily on estimates and assumptions. The recorded tax liabilities are based on estimates and assumptions that have been deemed reasonable by management. However, if our estimates are not representative of actual outcomes, recorded tax liabilities could be materially impacted.
Contingencies
We are involved in various legal proceedings, some of which involve claims for substantial amounts. An estimate is made to accrue for a loss contingency relating to any of these legal proceedings if we determine it is probable that a liability was incurred as of the date of the financial statements and the amount of loss can be reasonably estimated. Because of the subjective nature inherent in assessing the future outcome of litigation and because of the potential that an adverse outcome in legal proceedings could have a material impact on our financial condition or results of operations, such estimates are considered to be critical accounting estimates. We have reviewed and determined that at March 31, 2011, there were certain legal proceedings in which we are involved that met the conditions described above. Accordingly, we have accrued a loss contingency relating to such legal proceedings. See Note 12—“Accrued Liabilities” of the Notes to Consolidated Financial Statements included in this Report.
Recently Issued Accounting Standards
Accounting Standards Adopted
The Company adopted FASB Accounting Standards Codification (“ASC”) 2010-27-Other Expenses (Topic 720): Fees Paid to the Federal Government by Pharmaceutical Manufacturers (A consensus of the FASB Emerging Issues Task Force). The update addresses questions concerning how pharmaceutical manufactures should recognize and classify in their income statements fees mandated by the Patient Protection and Affordable Care Act as amended by the Health Care and Education Reconciliation Act.
Based on the Company’s 2011 product base and overall sales as a percentage of the industry, management concluded that the fee for 2011 would not be material.
111
Table of Contents
Accounting Standards Not Yet Adopted
In October 2009, the FASB issued new accounting guidance for recognizing revenue for a multiple-deliverable revenue arrangement. The new guidance amends the existing guidance for separately accounting for individual deliverables in a revenue arrangement with multiple deliverables, and removes the criterion that an entity must use objective and reliable evidence of fair value to separately account for the deliverables. The new guidance also establishes a hierarchy for determining the value of each deliverable and establishes the relative selling price method for allocating consideration when vendor specific objective evidence or third-party evidence of value does not exist. We must adopt the new guidance prospectively for new revenue arrangements entered into or materially modified beginning in the first quarter of fiscal year 2012. Earlier adoption is permitted. We are currently evaluating the impact that the new guidance will have on our Consolidated Financial Statements and the timing of our adoption.
In January 2010, the FASB issued Accounting Standards Update 2010-06, “Improving Disclosures about Fair Value Measurements” (“Update 2010-06”). Update 2010-06 provides amendments to current standards to require new disclosures for transfers of assets and liabilities between Levels 1 and 2 and for activity in Level 3 fair value measurements. Furthermore, the update provides amendments to clarify that a reporting entity should provide fair value measurement disclosures for each class of assets and liabilities and should provide disclosures about the valuation techniques and inputs used to measure fair value for both recurring and nonrecurring fair value measurements for those measurements that fall in either Level 2 or Level 3. Update 2010-06 becomes effective for interim and annual reporting periods beginning after December 15, 2010. The Company is currently evaluating this new statement.
In April 2010, the FASB issued ASU No. 2010-17, “Milestone Method of Revenue Recognition”, which amends FASB ASC Topic 605. ASU No. 2010-17 provides guidance on defining a milestone and determining when it may be appropriate to apply the milestone method of revenue recognition for research or development transactions. Research or development arrangements frequently include payment provisions whereby a portion or all of the consideration is contingent upon milestone events such as successful completion of phases in a study or achieving a specific result from the research or development efforts. The amendments in this ASU provide guidance on the criteria that should be met for determining whether the milestone method of revenue recognition is appropriate. ASU 2010-17 is effective for fiscal years and interim periods within those years beginning on or after June 15, 2010, with early adoption permitted. We are currently evaluating the impact that the new guidance will have on our Consolidated Financial Statements and the timing of our adoption.
| Item 7A. | Quantitative and Qualitative Disclosures About Market Risk |
Our exposure to market risk stems from fluctuating interest rates associated with our investment securities and our variable rate indebtedness that is subject to interest rate changes.
On February 25, 2009, we initiated legal action against Citigroup Global Markets Inc. (“CGMI”), through which we acquired the auction rate securities (“ARS”) we held at that time, in the District Court for the Eastern District of Missouri. On January 21, 2010, we entered into a Purchase and Release Agreement with CGMI pursuant to which CGMI agreed to purchase the ARS for an aggregate purchase price of approximately $61.7 million. We also received a two-year option (which expires on January 21, 2012) to reacquire the ARS (in whole or on a class-by-class basis) for the prices at which they were sold, as well as the right to receive further payments in the event any ARS are redeemed prior to the expiration of the option. We agreed to release CGMI from any liability or claim arising from our investment in ARS sold by CGMI, and we agreed with CGMI to take all necessary steps to dismiss with prejudice all claims asserted by us against CGMI regarding the ARS, including the legal action referred to above.
At March 31, 2011, our investment securities included $61.5 million in original principal amount of ARS (see Note 6—“Investment Securities” of the Notes to Consolidated Financial Statements included in this Report for more information regarding the settlement agreement and the proceeds received in connection therewith).
112
Table of Contents
Consistent with our investment policy guidelines, the ARS held by us are AAA-rated securities with long-term nominal maturities secured by student loans which are guaranteed by the U.S. Government. Liquidity for the ARS is typically provided by an auction process which allows holders to sell their notes and resets the applicable interest rate at pre-determined intervals, typically between seven to 35 days. However, with the liquidity issues experienced in global credit and capital markets, the ARS experienced failed auctions beginning in February 2008 and throughout fiscal years 2009, 2010 and 2011. An auction failure means that the parties wishing to sell their securities could not be matched with an adequate volume of buyers. The securities for which auctions have failed continue to accrue interest at the contractual rate and continue to be auctioned every 7, 14, 28 or 35 days, as the case may be, until the auction succeeds, the issuer calls the securities, or they mature.
The annual favorable impact on our net income as a result of a 100 basis point (where 100 basis points equals 1%) increase in short-term interest rates would be approximately $0.4 million based on our average cash and cash equivalents balances during the fiscal year ended March 31, 2011, compared to an increase of $1.1 million during the fiscal year ended March 31, 2010.
In May 2003, we issued $200.0 million principal amount of 2033 Notes. The interest rate on the 2033 Notes is fixed at 2.50% and therefore not subject to interest rate changes. Beginning May 16, 2006, we became obligated to pay contingent interest at a rate equal to 0.5% per annum during any six-month period, if the average trading price of the 2033 Notes per $1,000 principal amount for the five-trading day period ending on the third trading day immediately preceding the first day of the applicable six-month period equals $1,200 or more. In November 2007, the average trading price of the 2033 Notes reached the threshold for the applicable five-day trading period which resulted in the payment of contingent interest and beginning November 16, 2007 the 2033 Notes began to bear interest at a rate of 3.00% per annum. In May 2008, the average trading price of the 2033 Notes fell below the contingent interest threshold for the five-day trading period and beginning May 16, 2008 the 2033 Notes began to pay interest at a rate of 2.50% per annum. We may redeem some or all of the 2033 Notes at any time on or after May 21, 2006, at a redemption price, payable in cash, of 100% of the principal amount of the 2033 Notes, plus accrued and unpaid interest (including contingent interest, if any) to the date of redemption. Holders may require us to repurchase all or a portion of their 2033 Notes on May 16, 2013, 2018, 2023 and 2028, or upon a change in control, as defined in the indenture governing the 2033 Notes, at 100% of the principal amount of the 2033 Notes, plus accrued and unpaid interest (including contingent interest, if any) to the date of repurchase, payable in cash. Holders had the right to require us to repurchase all or a portion of their 2033 Notes on May 16, 2008 and, accordingly, we classified the 2033 Notes as a current liability as of March 31, 2008. Additionally, if an acceleration of the mortgage debt obligation were to occur as described above, and we are unable to pay it in full, an event of default could also be deemed to have occurred on the 2033 Notes. If an event of default is deemed to have occurred on the 2033 Notes, the principal amount plus any accrued and unpaid interest on the 2033 Notes could also become immediately due and payable. Since no holders required us to repurchase all or a portion of their 2033 Notes on May 16, 2008 and because the next date holders may require us to repurchase all or a portion of their 2033 Notes is May 16, 2013, the 2033 Notes were classified as a long-term liability as of March 31, 2009. The 2033 Notes are subordinate to all of our existing and future senior obligations.
In March 2006, we entered into a $43.0 million mortgage loan secured by three of our buildings that matures in April 2021. The interest rate on this loan is fixed at 5.91% per annum (and a default rate of 10.905% per annum) and not subject to market interest rate changes.
On March 17, 2011, the Company completed the offering and sale of the 2015 Notes. The 2015 Notes bear interest at an annual rate of 12% per year, payable semiannually in arrears on March 15 and September 15 of each year, commencing September 15, 2011. The 2015 Notes will mature March 15, 2015. After an original issue discount of 3%, the Company received proceeds of $218.3 million which were used to fund a first-year interest reserve totaling $27.0 million (reflected as restricted cash on the balance sheet), repay all existing obligations to U.S. Healthcare totaling approximately $61.1 million and pay fees and expenses associated with the offering of the 2015 Notes of approximately $10.0 million. In connection with these payments, the Company also terminated
113
Table of Contents
all future loan commitments with U.S. Healthcare. The remaining proceeds, totaling approximately $120.0 million will be used for general corporate purposes, including the launch of Makena™. The 2015 Notes were offered only to accredited investors pursuant to Regulation D under the Securities Act of 1933, as amended (See Note 13—“Long-Term Debt” for a description of the 2015 Notes of the Notes to Consolidated Financial Statements in this Report).
114
Table of Contents
| Item 8. | Financial Statements and Supplementary Data |
Report of Independent Registered Public Accounting Firm
Board of Directors and Shareholders
K-V Pharmaceutical Company
Bridgeton, Missouri
We have audited the accompanying consolidated balance sheets of K-V Pharmaceutical Company and Subsidiaries as of March 31, 2011 and 2010 and the related consolidated statements of operations, comprehensive loss, shareholders’ equity (deficit), and cash flows for the years then ended. In connection with our audits of the financial statements, we have also audited the information in financial statement Schedule II listed in the accompanying index for the years ended March 31, 2011 and 2010. These consolidated financial statements and financial statement schedule are the responsibility of the Company’s management. Our responsibility is to express an opinion on these consolidated financial statements and financial statement schedule based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements and schedule. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of K-V Pharmaceutical Company and Subsidiaries at March 31, 2011 and 2010 and the results of their operations and their cash flows for the years then ended, in conformity with accounting principles generally accepted in the United States of America.
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 3 to the consolidated financial statements, the Company has suspended the shipment of all but one of the products manufactured by the Company and must comply with a consent decree with the Food and Drug Administration before approved products can be reintroduced to the market. Significant negative impacts on operating results and cash flows from these actions include: recurring losses from operations, a shareholders’ deficit, and negative working capital; the potential inability of the Company to raise additional capital or debt financing; suspension of manufacturing; significant uncertainties related to litigation and governmental inquiries; and potential debt covenant violations. These factors raise substantial doubt about the Company’s ability to continue as a going concern. Management’s plans in regard to these matters are also discussed in Note 3. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Also, in our opinion, the 2011 and 2010 information in the financial statement schedule, when considered in relation to the basic consolidated financial statements taken as a whole, presents fairly, in all material respects, the information set forth therein.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), K-V Pharmaceutical Company’s internal control over financial reporting as of March 31, 2011, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) and our report dated June 10, 2011 expressed an adverse opinion on the effectiveness of the Company’s internal control over financial reporting.
/s/ BDO USA, LLP
Chicago, Illinois
June 10, 2011
115
Table of Contents
Report of Independent Registered Public Accounting Firm
The Board of Directors and Shareholders
K-V Pharmaceutical Company:
We have audited the accompanying consolidated statements of operations, comprehensive loss, shareholders’ equity (deficit), and cash flows of K-V Pharmaceutical Company and subsidiaries (the Company) for the year ended March 31, 2009. In connection with our audit of the consolidated financial statements, we also have audited financial statement Schedule II for the year ended March 31, 2009. These consolidated financial statements and financial statement schedule are the responsibility of the Company’s management. Our responsibility is to express an opinion on these consolidated financial statements and financial statement schedule based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audit provides a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the Company’s results of operations and cash flows for the year ended March 31, 2009, in conformity with U.S. generally accepted accounting principles. Also, in our opinion, the related financial statement schedule for the year ended March 31, 2009, when considered in relation to the basic consolidated financial statements taken as a whole, presents fairly, in all material respects, the information set forth therein.
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 3 to the consolidated financial statements in Item 8 of the March 31, 2009 Form 10-K, the Company has suspended the shipment of all products manufactured by the Company and must comply with a consent decree with the FDA before approved products can be reintroduced to the market. Significant negative impacts on operating results and cash flows from these actions including the potential inability of the Company to raise capital; suspension of manufacturing; significant uncertainties related to litigation and governmental inquiries; and debt covenant violations raise substantial doubt about the Company’s ability to continue as a going concern. Management’s plans in regard to these matters are also discussed in Note 3 to the consolidated financial statements in Item 8 of the March 31, 2009 Form 10-K. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
/s/ KPMG LLP
St. Louis, Missouri
March 25, 2010, except as to note 23, which is as of June 10, 2011
116
Table of Contents
K-V PHARMACEUTICAL COMPANY AND SUBSIDIARIES
(In millions, except share and per share information)
| March 31, | ||||||||
| 2011 | 2010 | |||||||
| ASSETS | ||||||||
| Current Assets: |
||||||||
| Cash and cash equivalents |
$ | 137.6 | $ | 60.7 | ||||
| Investment securities |
57.2 | — | ||||||
| Restricted cash |
34.5 | — | ||||||
| Receivables, net |
33.4 | 1.0 | ||||||
| Inventories, net |
1.0 | 1.2 | ||||||
| Other current assets |
13.8 | 17.0 | ||||||
| Current assets held for sale |
9.0 | 11.8 | ||||||
| Total Current Assets |
286.5 | 91.7 | ||||||
| Property and equipment, less accumulated depreciation |
67.6 | 78.4 | ||||||
| Investment securities |
— | 65.9 | ||||||
| Intangible assets, net |
150.9 | 37.5 | ||||||
| Other assets |
15.1 | 12.4 | ||||||
| Non-current assets held for sale |
44.6 | 72.7 | ||||||
| Total Assets |
$ | 564.7 | $ | 358.6 | ||||
| LIABILITIES | ||||||||
| Current Liabilities: |
||||||||
| Accounts payable |
$ | 25.7 | $ | 39.0 | ||||
| Accrued liabilities |
141.9 | 64.0 | ||||||
| Warrant liability |
112.3 | — | ||||||
| Current maturities of long-term debt |
85.4 | 63.9 | ||||||
| Current liabilities associated with assets held for sale |
2.5 | 5.9 | ||||||
| Total Current Liabilities |
367.8 | 172.8 | ||||||
| Long-term debt, less current maturities |
418.3 | 233.2 | ||||||
| Other long-term liabilities |
95.2 | 47.7 | ||||||
| Deferred tax liability |
57.4 | 44.1 | ||||||
| Total Liabilities |
938.7 | 497.8 | ||||||
| Commitments and Contingencies |
||||||||
| SHAREHOLDERS’ DEFICIT | ||||||||
| 7% cumulative convertible Preferred Stock, $.01 par value; $25.00 stated and liquidation value; 840,000 shares authorized; issued and outstanding— 40,000 shares at both March 31, 2011 and 2010 (convertible into Class A shares on a 8.4375-to-one basis) |
— | — | ||||||
| Class A and Class B Common Stock, $.01 par value; 150,000,000 and 75,000,000 shares authorized, respectively; |
0.5 | 0.4 | ||||||
| Class A—issued 52,013,609; outstanding 48,604,559 at March 31, 2011 and issued 41,157,609; outstanding 37,736,660 at March 31, 2010, respectively |
||||||||
| Class B—issued 11,300,857; outstanding 11,206,285 at March 31, 2011 and issued 12,206,857; outstanding 12,112,285 at March 31, 2010 (convertible into Class A shares on a one-for-one basis) |
0.1 | 0.1 | ||||||
| Additional paid-in capital |
109.1 | 170.0 | ||||||
| Accumulated deficit |
(428.0 | ) | (253.9 | ) | ||||
| Accumulated other comprehensive income |
1.7 | 1.6 | ||||||
| Less: Treasury stock, 3,409,050 shares of Class A and 94,572 shares of Class B Common Stock at March 31, 2011, and 3,404,366 shares of Class A and 94,572 shares of Class B Common Stock at March 31, 2010, at cost |
(57.4 | ) | (57.4 | ) | ||||
| Total Shareholders’ Deficit |
(374.0 | ) | (139.2 | ) | ||||
| Total Liabilities and Shareholders’ Deficit |
$ | 564.7 | $ | 358.6 | ||||
See Accompanying Notes to Consolidated Financial Statements.
117
Table of Contents
K-V PHARMACEUTICAL COMPANY AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF OPERATIONS
(In millions, except per share data)
| Years ended March 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Net revenues |
$ | 27.3 | $ | 9.1 | $ | 113.2 | ||||||
| Cost of sales |
1.1 | 1.6 | 15.1 | |||||||||
| Gross profit |
26.2 | 7.5 | 98.1 | |||||||||
| Operating expenses: |
||||||||||||
| Research and development |
22.1 | 29.1 | 69.8 | |||||||||
| Selling and administrative |
96.0 | 111.7 | 220.7 | |||||||||
| Restructuring and impairment charges |
0.1 | 89.0 | 46.4 | |||||||||
| Purchased in-process research and development |
— | 70.0 | 2.0 | |||||||||
| Litigation and governmental inquiries, net |
7.4 | 15.0 | 50.0 | |||||||||
| Gains on sale of assets |
0.1 | — | — | |||||||||
| Total operating expenses |
125.7 | 314.8 | 388.9 | |||||||||
| Operating loss |
(99.5 | ) | (307.3 | ) | (290.8 | ) | ||||||
| Other expense (income): |
||||||||||||
| Loss on extinguishment of debt |
106.2 | — | — | |||||||||
| Change in warrant liability |
(70.7 | ) | — | — | ||||||||
| Interest expense |
15.4 | 8.0 | 9.3 | |||||||||
| Interest and other (income) expense |
(2.0 | ) | (5.8 | ) | 6.3 | |||||||
| Total other expense (income), net |
48.9 | 2.2 | 15.6 | |||||||||
| Loss from continuing operations before income taxes |
(148.4 | ) | (309.5 | ) | (306.4 | ) | ||||||
| Income tax provision (benefit) |
7.8 | (23.9 | ) | (38.6 | ) | |||||||
| Loss from continuing operations |
(156.2 | ) | (285.6 | ) | (267.8 | ) | ||||||
| Net loss from discontinued operations |
||||||||||||
| (net taxes of $(7.4), $(4.1) and $(7.0)) |
(30.6 | ) | (7.2 | ) | (45.8 | ) | ||||||
| Gain on sale of discontinued operations |
||||||||||||
| (net taxes of $7.4, $5.3 and $-) |
12.8 | 9.2 | — | |||||||||
| Net loss |
$ | (174.0 | ) | $ | (283.6 | ) | $ | (313.6 | ) | |||
| Years ended March 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Earnings (loss) per share from continuing operations: |
||||||||||||
| Basic and diluted—Class A and B common |
$ | (3.05 | ) | $ | (5.73 | ) | $ | (5.39 | ) | |||
| Earnings (loss) per share from discontinued operations: |
||||||||||||
| Basic and diluted—Class A and B common |
$ | (0.60 | ) | $ | (0.14 | ) | $ | (0.92 | ) | |||
| Earnings per share from gain on sale of discontinued operations: |
||||||||||||
| Basic and diluted—Class A and B common |
$ | 0.25 | $ | 0.18 | $ | — | ||||||
| Earnings (loss) per share: |
||||||||||||
| Basic and diluted—Class A and B common |
$ | (3.40 | ) | $ | (5.69 | ) | $ | (6.31 | ) | |||
| Shares used in per share calculation: |
||||||||||||
| Basic—Class A common |
39.1 | 37.8 | 37.6 | |||||||||
| Basic and diluted—Class B common |
12.2 | 12.1 | 12.1 | |||||||||
| Diluted—Class A common |
51.3 | 49.9 | 49.7 | |||||||||
See Accompanying Notes to Consolidated Financial Statements.
118
Table of Contents
K-V PHARMACEUTICAL COMPANY AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF COMPREHENSIVE LOSS
(In millions)
| Years Ended March 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Net loss |
$ | (174.0 | ) | $ | (283.6 | ) | $ | (313.6 | ) | |||
| Unrealized gain (loss) on available for sale securities: |
||||||||||||
| Unrealized holding gain (loss) during the period |
(0.1 | ) | 0.8 | (5.9 | ) | |||||||
| Reclassification of losses included in net loss |
— | — | 10.4 | |||||||||
| Tax impact related to unrealized holding gain (loss) and reclassification of losses |
— | (0.3 | ) | (1.6 | ) | |||||||
| Foreign currency translation adjustment |
0.2 | 0.3 | (0.6 | ) | ||||||||
| Total other comprehensive income |
0.1 | 0.8 | 2.3 | |||||||||
| Total comprehensive loss |
$ | (173.9 | ) | $ | (282.8 | ) | $ | (311.3 | ) | |||
See Accompanying Notes to Consolidated Financial Statements.
119
Table of Contents
K-V PHARMACEUTICAL COMPANY AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF SHAREHOLDERS’ EQUITY (DEFICIT)
(In millions)
| Years Ended March 31, 2011, 2010 and 2009 | ||||||||||||||||||||||||||||||||
| Preferred Stock |
Class A Common Stock |
Class B Common Stock |
Additional Paid-In Capital |
Retained Earnings (Accumulated Deficit) |
Accumulated Other Comprehensive Income (Loss) |
Treasury Stock |
Total Shareholders’ Equity (Deficit) |
|||||||||||||||||||||||||
| Balance at March 31, 2008 |
$ | — | $ | 0.4 | $ | 0.1 | $ | 159.4 | $ | 351.3 | $ | (1.5 | ) | $ | (56.2 | ) | $ | 453.5 | ||||||||||||||
| Net loss |
— | — | — | — | (313.6 | ) | — | — | (313.6 | ) | ||||||||||||||||||||||
| Dividends paid on preferred stock |
— | — | — | — | (0.1 | ) | — | — | (0.1 | ) | ||||||||||||||||||||||
| Conversion of less than 0.1 Class B Shares to Class A shares |
— | — | — | — | — | — | — | — | ||||||||||||||||||||||||
| Stock-based compensation |
— | — | — | 3.5 | — | — | — | 3.5 | ||||||||||||||||||||||||
| Purchase of common stock for treasury |
— | — | — | — | — | — | (0.9 | ) | (0.9 | ) | ||||||||||||||||||||||
| Stock options exercised- 0.3 shares of Class A and 0.1 shares of Class B |
— | — | — | 2.5 | — | — | — | 2.5 | ||||||||||||||||||||||||
| Cumulative effect of change in accounting principle |
— | — | — | — | (7.8 | ) | — | — | (7.8 | ) | ||||||||||||||||||||||
| Other comprehensive income |
— | — | — | — | — | 2.3 | — | 2.3 | ||||||||||||||||||||||||
| Balance at March 31, 2009 |
— | 0.4 | 0.1 | 165.4 | 29.8 | 0.8 | (57.1 | ) | 139.4 | |||||||||||||||||||||||
| Net loss |
— | — | — | — | (283.6 | ) | — | — | (283.6 | ) | ||||||||||||||||||||||
| Dividends paid on preferred stock |
— | — | — | — | (0.1 | ) | — | — | (0.1 | ) | ||||||||||||||||||||||
| Conversion of less than 0.1 Class B Shares to Class A shares |
— | — | — | — | — | — | — | — | ||||||||||||||||||||||||
| Stock-based compensation |
— | — | — | 3.8 | — | — | — | 3.8 | ||||||||||||||||||||||||
| Purchase of common stock for treasury |
— | — | — | — | — | — | (0.3 | ) | (0.3 | ) | ||||||||||||||||||||||
| Stock options exercised- 0.1 shares of Class A and less than 0.1 shares of Class B |
— | — | — | 0.8 | — | — | — | 0.8 | ||||||||||||||||||||||||
| Other comprehensive income |
— | — | — | — | — | 0.8 | — | 0.8 | ||||||||||||||||||||||||
| Balance at March 31, 2010 |
— | 0.4 | 0.1 | 170.0 | (253.9 | ) | 1.6 | (57.4 | ) | (139.2 | ) | |||||||||||||||||||||
| Net loss |
— | — | — | — | (174.0 | ) | — | — | (174.0 | ) | ||||||||||||||||||||||
| Dividends paid on preferred stock |
— | — | — | — | (0.1 | ) | — | — | (0.1 | ) | ||||||||||||||||||||||
| Conversion of 0.1 Class B Shares to Class A shares |
— | 0.1 | — | 0.1 | — | — | — | 0.2 | ||||||||||||||||||||||||
| Stock-based compensation |
— | — | — | 3.0 | — | — | — | 3.0 | ||||||||||||||||||||||||
| Purchase of common stock for treasury |
— | — | — | — | — | — | — | — | ||||||||||||||||||||||||
| Stock options exercised- less than 0.1 shares of Class A and less than 0.1 shares of Class B |
— | — | — | 0.2 | — | — | — | 0.2 | ||||||||||||||||||||||||
| Issuance of warrants |
— | — | — | 81.6 | — | — | — | 81.6 | ||||||||||||||||||||||||
| Reclassification of warrants as liabilities |
— | — | — | (175.5 | ) | — | — | — | (175.5 | ) | ||||||||||||||||||||||
| Private equity offering |
— | — | — | 29.7 | — | — | — | 29.7 | ||||||||||||||||||||||||
| Other comprehensive income |
— | — | — | — | — | 0.1 | — | 0.1 | ||||||||||||||||||||||||
| Balance at March 31, 2011 |
$ | — | $ | 0.5 | $ | 0.1 | $ | 109.1 | $ | (428.0 | ) | $ | 1.7 | $ | (57.4 | ) | $ | (374.0 | ) | |||||||||||||
See Accompanying Notes to Consolidated Financial Statements.
120
Table of Contents
K-V PHARMACEUTICAL COMPANY AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CASH FLOWS
(In millions)
| Years ended March 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Operating Activities: |
||||||||||||
| Net loss |
$ | (174.0 | ) | $ | (283.6 | ) | $ | (313.6 | ) | |||
| Adjustments to reconcile net loss to net cash used in operating activities: |
||||||||||||
| Purchased in-process research and development |
— | 70.0 | 2.0 | |||||||||
| Depreciation and amortization |
20.0 | 33.5 | 35.9 | |||||||||
| Loss on property and equipment |
— | 1.8 | 3.0 | |||||||||
| Gain from sale of assets, net |
(20.2 | ) | (14.5 | ) | — | |||||||
| Change in warrant liability |
(70.7 | ) | — | — | ||||||||
| Loss on extinguishment of debt |
106.2 | — | — | |||||||||
| Impairment of assets |
11.6 | 128.5 | 36.6 | |||||||||
| Loss on investment securities |
— | — | 9.4 | |||||||||
| Involuntary conversion gain in discontinued operations |
(3.5 | ) | (5.6 | ) | — | |||||||
| Deferred income tax provision |
8.4 | 41.9 | 24.3 | |||||||||
| Deferred compensation |
. | — | (8.6 | ) | ||||||||
| Stock-based compensation |
3.0 | 3.8 | 3.5 | |||||||||
| Changes in operating assets and liabilities: |
— | — | ||||||||||
| Receivables, net |
(33.9 | ) | 19.2 | 109.5 | ||||||||
| Inventories, net |
(2.0 | ) | 16.8 | 68.4 | ||||||||
| Income taxes |
3.3 | 40.6 | (77.5 | ) | ||||||||
| Accounts payable and accrued liabilities |
(12.1 | ) | (64.2 | ) | 83.7 | |||||||
| Other assets and liabilities, net |
4.4 | 11.7 | 21.2 | |||||||||
| Net cash used in operating activities |
(159.5 | ) | (0.1 | ) | (2.2 | ) | ||||||
| Investing Activities: |
||||||||||||
| Purchase of property and equipment |
(0.3 | ) | (10.2 | ) | (23.6 | ) | ||||||
| Proceeds from sale of property and equipment |
0.7 | — | — | |||||||||
| Insurance proceeds |
3.5 | 5.6 | — | |||||||||
| Proceeds from sale of business/assets held for sale, net of fees |
42.0 | — | — | |||||||||
| Purchase of marketable securities |
— | — | (0.6 | ) | ||||||||
| Sale of marketable securities |
1.3 | 1.1 | 52.1 | |||||||||
| Purchased in-process research and development |
— | (70.0 | ) | (2.0 | ) | |||||||
| Intangible asset acquisition |
(12.5 | ) | — | — | ||||||||
| Increase in restricted cash |
(34.5 | ) | — | — | ||||||||
| Cash paid for acquired assets |
— | — | (3.0 | ) | ||||||||
| Net cash provided by (used in) investing activities |
0.2 | (73.5 | ) | 22.9 | ||||||||
| Financing Activities: |
||||||||||||
| Principal payment on long-term debt |
(83.7 | ) | (2.7 | ) | (2.6 | ) | ||||||
| Proceeds from issuance of debt and warrants |
299.1 | — | — | |||||||||
| Proceeds from collateralized obligation |
— | 61.8 | — | |||||||||
| Redemption of collateralized obligation |
(8.8 | ) | (0.5 | ) | — | |||||||
| Proceeds from private equity offering |
29.7 | — | — | |||||||||
| Repayment of borrowing on line of credit |
— | — | (30.0 | ) | ||||||||
| Dividends paid on preferred stock |
(0.1 | ) | (0.1 | ) | (0.1 | ) | ||||||
| Purchase of common stock for treasury |
— | (0.3 | ) | (0.9 | ) | |||||||
| Cash deposits received for stock options |
— | — | 0.7 | |||||||||
| Net cash provided by (used in) financing activities |
236.2 | 58.2 | (32.9 | ) | ||||||||
| Effect of foreign exchange rate changes on cash |
— | 0.4 | (0.6 | ) | ||||||||
| Increase (decrease) in cash and cash equivalents |
76.9 | (15.0 | ) | (12.8 | ) | |||||||
| Cash and cash equivalents: |
||||||||||||
| Beginning of year |
60.7 | 75.7 | 88.5 | |||||||||
| End of period |
$ | 137.6 | $ | 60.7 | $ | 75.7 | ||||||
| Supplemental cash flow disclosures: |
||||||||||||
| Cash paid for interest |
$ | 22.2 | $ | 7.3 | $ | 9.1 | ||||||
| Cash paid for income taxes |
$ | 0.1 | $ | 0.3 | $ | 8.3 | ||||||
| Non-cash investing and financing activities: |
||||||||||||
| Stock options exercised (at expiration of two-year forfeiture period) |
$ | 0.2 | $ | 0.8 | $ | 2.5 | ||||||
| Warrants reclassified to liabilities from equity |
$ | 175.5 | $ | — | $ | — | ||||||
| Makena product rights |
$ | 107.5 | $ | — | $ | — | ||||||
See Accompanying Notes to Consolidated Financial Statements.
121
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(Dollars in millions, except per share data)
| 1. | Description of Business |
General Overview
K-V Pharmaceutical Company was incorporated under the laws of Delaware in 1971 as a successor to a business originally founded in 1942. K-V Pharmaceutical Company and its wholly-owned subsidiaries, including Ther-Rx Corporation (“Ther-Rx”), Nesher Pharmaceuticals, Inc. (“Nesher”), ETHEX Corporation (“ETHEX”) and Particle Dynamics, Inc. (“PDI”), are referred to in the following Notes to Consolidated Financial Statements as “KV” or the “Company.” We are a fully integrated specialty pharmaceutical company that develops, manufactures, acquires and markets technologically-distinguished branded and generic/non-branded prescription pharmaceutical products. Our strategic goal is to become a specialty branded pharmaceutical marketing company primarily focused in women’s healthcare. We have a broad range of dosage form manufacturing capabilities, including tablets, capsules, creams and liquids. We conduct our branded pharmaceutical operations through Ther-Rx and, previously, we conducted our generic/non-branded pharmaceutical operations through ETHEX, which focused principally on technologically-distinguished generic products prior to the cessation of its operations on March 2, 2010 and its dissolution on December 15, 2010. Through PDI, divested in June 2010, we developed, manufactured and marketed technologically advanced, value-added raw material products for the pharmaceutical industry and other markets. In May 2010, we formed a wholly-owned subsidiary, Nesher, to operate as the sales and marketing company for our generic products. The Company expects to complete a divestiture of Nesher and the Company’s generics business and assets by the end of calendar year 2011.
Our original strategy was to engage in the development of proprietary drug delivery systems and formulation technologies which enhance the effectiveness of new therapeutic agents and existing pharmaceutical products. Today, we utilize one of those technologies, SITE RELEASE® in two products expected to return to our branded portfolio prior to the end of calendar year 2011. Going forward, our business strategy will be primarily defined by the potential in-licensing and acquisition of pharmaceutical products rather than the historical strategy of the internal development of pharmaceutical products.
During fiscal year 2009, the Company announced six separate voluntary recalls of certain tablet form generic products as a precaution due to the potential existence of oversized tablets. In December 2008, the Food and Drug Administration (the “FDA”) began an inspection of the Company’s facilities. The Company suspended shipments of all approved tablet-form products in December 2008 and of all other drug products in January 2009. Also, in January 2009, the Company initiated a nationwide voluntary recall affecting most of its products. On March 2, 2009, the Company entered into a consent decree with the FDA regarding its drug manufacturing and distribution. The consent decree was entered by the U.S. District Court, Eastern District of Missouri, and Eastern Division on March 6, 2009. As part of the consent decree, the Company agreed not to directly or indirectly do or cause the manufacture, processing, packing, labeling, holding, introduction or delivery for introduction into interstate commerce at or from any of its facilities of any drug, until the Company has satisfied certain requirements designed to demonstrate compliance with the FDA’s current good manufacturing practice (“cGMP”) regulations. The consent decree provides for a series of measures that, when satisfied, will permit the Company to resume the manufacture and distribution of approved drug products. The Company has also agreed not to distribute its products that are not FDA approved, including its prenatal vitamins and hematinic products, unless it obtains FDA approval for such products through the FDA’s New Drug Application (“NDA”) or Abbreviated New Drug Application (“ANDA”) processes. These actions and the requirements under the consent decree have had, and are expected to continue to have, a material adverse effect on the Company’s liquidity position and its results of operations. We resumed shipment of extended-release potassium chloride capsule, Micro-K® 10mEq and Micro-K® 8mEq, in September 2010, resumed shipments of the generic version Potassium Chloride Extended Release Capsule in December 2010 and we began shipping Makena™, a drug manufactured by a third party, in March 2011. We are continuing to prepare other products for FDA inspection although we do not expect to resume shipping other products until later in fiscal year 2012, at the earliest.
122
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
In July 2010, our Board of Directors directed management to explore strategic alternatives with respect to Nesher and the assets and operations of our generic products business and in the fourth quarter of fiscal year 2011, management committed to a plan to divest the generics business. We have retained Jefferies & Co., Inc., to advise us with the divestiture of Nesher and the Company’s generics business. In the meantime, we will continue to prepare products for FDA inspection and continue to anticipate the reintroduction of approved products into the market.
Both PDI and our generics business have been presented herein as discontinued operations. See Note 23—“Divestitures” for further details.
Significant Developments
Changes in Management and Directors
At the Annual Meeting of Stockholders for fiscal year 2009 held on June 10, 2010 (the “Annual Meeting”), the stockholders elected Gregory Bentley, Mark A. Dow, Terry B. Hatfield, David S. Hermelin, Marc S. Hermelin, Joseph D. Lehrer and John Sampson to serve as directors with terms expiring at the Annual Meeting of Stockholders for fiscal year 2010. Former members of the Board Jean M. Bellin, Kevin S. Carlie, Jonathon E. Killmer and Norman D. Schellenger were not re-elected.
On June 14, 2010, Stephen A. Stamp resigned, effective immediately, from his position as Chief Financial Officer of our Company. Thomas S. McHugh was appointed Chief Financial Officer and Treasurer effective July 15, 2010. Prior to this appointment, Mr. McHugh served as Chief Accounting Officer and Vice President of Finance—Corporate Controller.
On June 15, 2010, each of Mr. Hatfield and Mr. Sampson resigned as members of the Board, effective as of the earlier of July 7, 2010 or the date a replacement was appointed. Mr. Hatfield served as the Chairman of the Board and Mr. Sampson served on the Audit Committee. Each of Mr. Hatfield and Mr. Sampson indicated that he was resigning because of serious concerns regarding the ability of the newly-constituted Board and senior management to provide the required independent oversight of the business during the current critical period in its history.
On June 17, 2010, the Board appointed Ana I. Stancic as a director to fill the vacancy created by the resignation of Mr. Hatfield. As noted above, Mr. Hatfield’s resignation became effective upon the appointment of Ms. Stancic.
On July 7, 2010, the Board appointed David Sidransky, M.D. as a director to fill the vacancy created by the resignation of Mr. Sampson. As noted above, Mr. Sampson’s resignation became effective upon the appointment of Dr. Sidransky.
On July 29, 2010, the Board increased the total number of Board members to eight (but returning automatically to seven members upon any current director leaving the Board) and appointed Robert E. Baldini as a director to fill the newly-created position.
At a Board meeting held subsequent to the Annual Meeting on June 10, 2010, the Board terminated the employment of David A. Van Vliet, who then served as Interim President and Interim Chief Executive Officer, effective at the end of the 30-day notice period provided for in his employment agreement, during which period he was placed on administrative leave.
123
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Also at that meeting, the Board appointed Gregory J. Divis, Jr. as the Interim President and Interim Chief Executive Officer of our Company. Mr. Divis was subsequently appointed as our permanent President and Chief Executive Officer on November 17, 2010. The other terms of Mr. Divis’ employment were not changed by this appointment.
On November 10, 2010, Marc S. Hermelin voluntarily resigned as a member of the Board. We had been advised that Office of Inspector General of the U.S. Department of Health and Human Services (“HHS OIG”) notified Mr. M. Hermelin that he would be excluded from participating in federal healthcare programs effective November 18, 2010. In an effort to avoid adverse consequences to our Company, including a potential discretionary exclusion of our Company from participation in federal healthcare programs, and to enable our Company to secure our expanded financial agreement, as more fully described in Note 13—“Long-Term Debt”,with the Company, HHS OIG, Mr. M. Hermelin and his wife (solely with respect to her obligations thereunder, including as joint owner with Mr. M. Hermelin of certain shares of Company stock) entered into the Settlement Agreement under which Mr. M. Hermelin also resigned as trustee of all family trusts that hold KV stock, agreed to divest his personal ownership interests in our Company’s Class A Common and Class B Common Stock (approximately 1.8 million shares, including shares held jointly with his wife) over an agreed upon period of time in accordance with a divestiture plan and schedule approved by HHS OIG, and agreed to refrain from voting stock under his personal control. In order to implement such agreement, Mr. M. Hermelin and his wife granted to an independent third party immediate irrevocable proxies and powers of attorney to divest their personal stock interests in the Company if Mr. M. Hermelin does not timely do so. The Settlement Agreement also required Mr. M. Hermelin to agree, for the duration of his exclusion, not to seek to influence or be involved with, in any manner, the governance, management, or operations of our Company.
As long as the parties comply with the Settlement Agreement, HHS OIG has agreed not to exercise its discretionary authority to exclude our Company from participation in federal health care programs, thereby allowing our Company and our subsidiaries (with the single exception of ETHEX, which is being dissolved pursuant to the Divestiture Agreement with HHS OIG) to continue to conduct business through all federal and state healthcare programs.
As a result of Mr. M. Hermelin’s resignation and the two agreements with HHS OIG, we believe we have resolved our remaining issues with respect to HHS OIG and are positioned to continue to participate in Federal healthcare programs now and in the future.
On June 1, 2011, Patrick Christmas joined the Company as Vice President and General Counsel, Mr. Christmas will become the Company’s chief legal officer succeeding Gregory S. Bentley, who is the Company’s Senior Vice President of Law and a member of its Board of Directors. Mr. Bentley has served in that capacity since June 2010 under an arrangement which contemplated that he would return to his private legal practice after the Company filled the General Counsel position. Mr. Bentley will continue to serve as a director of the Company and to counsel the Company after he resumes private practice.
Discontinuation of Manufacturing and Distribution; Product Recalls; and the FDA Consent Decree
In May 2008, we received two reports of an oversized morphine sulfate extended-release tablet in commercial distribution. We conducted an investigation into the possible causes of any such oversized tablets and the likelihood that additional lots of morphine sulfate extended-release tablets or other products might contain oversized tablets. We instituted changes in our manufacturing processes to address the identified causes and intended to prevent any oversized tablets from entering commercial distribution. In addition, in June 2008, ETHEX initiated voluntary recalls of morphine sulfate 30-mg and 60-mg extended-release tablets. In July 2008, a
124
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
voluntary recall was initiated in Canada by the seller of specific lots of morphine sulfate 60-mg, 30-mg and 15-mg extended-release tablets that we manufactured. On October 15, 2008, ETHEX commenced a voluntary recall of three specific lots of dextroamphetamine sulfate 5-mg tablets as a precaution due to the possible presence of oversized tablets. On November 7, 2008, ETHEX announced a voluntary recall to the consumer level of multiple lots of five generic products of varying strengths as a precaution due to the potential presence of oversized tablets. These products included: propafenone HCl tablets, isosorbide mononitrate extended-release tablets, morphine sulfate extended-release tablets, morphine sulfate immediate release tablets, and dextroamphetamine sulfate tablets. On November 10, 2008, ETHEX initiated a voluntary recall to the retail level as a precaution due to the possible presence of oversized tablets. This recall affected multiple lots of 18 generic/non-branded products.
On December 15, 2008, the FDA began an inspection of our facilities.
On December 19, 2008, we voluntarily suspended all shipments of our FDA approved drug products in tablet form and commenced a voluntary nationwide single production lot recall of one of our pain management drugs. The 14 products of varying strengths affected by the suspension included metoprolol succinate extended-release tablets (metoprolol), oxycodone HCl tablets and potassium chloride 20 mEq extended-release tablets.
Effective January 22, 2009, we voluntarily suspended the manufacturing and shipment of the remainder of our products, except for three products we distribute but do not manufacture and which do not generate a significant amount of revenue.
On January 28, 2009, we initiated a nationwide voluntary recall of products manufactured or packaged at KV facilities, affecting most of our products. The recall was subsequently expanded on February 3, 2009. This recall affected multiple lots of over 150 branded and generic/non-branded products.
On February 2, 2009, the FDA issued inspectional observations set forth on a “Form 483 Report.”
On March 2, 2009, we entered into a consent decree with the FDA regarding our drug manufacturing and distribution. The consent decree was entered by the U.S. District Court, Eastern District of Missouri, Eastern Division on March 6, 2009 and continues for a period of six years following satisfaction of certain obligations contained in the consent decree after which we may petition the Court for relief from the consent decree. As part of the consent decree, we have agreed not to directly or indirectly do or cause the manufacture, processing, packing, labeling, holding, introduction or delivery for introduction into interstate commerce at or from any of our facilities of any drug, until we have satisfied certain requirements designed to demonstrate compliance with the FDA’s cGMP regulations. The consent decree provides for a series of measures that, when satisfied, will permit us to resume the manufacture and distribution of approved drug products. We have also agreed not to distribute our products that are not FDA approved, including our prenatal vitamins and hematinic products, unless we obtain FDA approval for such products through the FDA’s ANDA or NDA processes.
On March 16, 2009, in response to the consent decree requirements, we initiated the disposal of our existing affected inventory of products, which was completed within the required timeframe.
As part of such measures set forth in the consent decree, we were required to provide, and have provided, to the FDA a work plan (the “Work Plan”) for approval, which sets forth the steps we have taken and will subsequently take to address previously identified deficiencies in our compliance with cGMP regulations.
On July 27, 2009, a representative of the Compliance Branch of the FDA’s Kansas City District, acting in conjunction with the Office of Compliance of the FDA’s Center for Drug Evaluation and Research (“CDER”),
125
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
notified us by electronic mail that the FDA had completed its review of the Work Plan. Subject to the addition of certain changes, to which we agreed and incorporated into the Work Plan, the FDA accepted the Work Plan. While acceptance of the Work Plan was pending, we, with the knowledge of the FDA, had already begun implementing certain measures set forth in the Work Plan. The final Work Plan, with all requested changes, was submitted to the FDA on August 13, 2009 and accepted by the FDA.
The consent decree further provides that, before resuming manufacturing, we were required to retain and have an independent cGMP expert undertake a review of our facilities and operations and certify compliance with cGMP regulations. Following that certification, the FDA would make a determination as to whether we are in compliance. On January 13, 2010, our independent cGMP expert, Lachman Consultants (“Lachman”),, notified the FDA that Lachman had performed a comprehensive inspection and that our facilities and controls are in compliance with cGMP and the consent decree, but advised us to enact further enhancements to certain aspects of our cGMP systems. In accordance with the advice from Lachman, we continued to enhance our cGMP systems, and Lachman subsequently reinspected our cGMP systems and on April 26, 2010 certified our compliance with all cGMP systems requirements.
The next step in the process for resumption of product shipment was for Lachman to certify individual products manufactured under the newly certified cGMP systems. We completed the manufacture of validation batches of the first product, which were successfully completed through Lachman review on July 27, 2010.
Lachman certified the manufacture of the product on July 27, 2010, and the FDA subsequently conducted its own inspection during the week of August 16, 2010 of our facilities, systems and processes as outlined in the consent decree and found no adverse findings. The Company received notification from the FDA on September 8, 2010 of approval for our discontinued generics operations to ship into the marketplace the first product approved under the consent decree, i.e., the Potassium Chloride Extended Release Capsule. We resumed shipment of extended-release potassium chloride capsule, Micro-K® 10mEq and Micro-K® 8mEq, in September 2010. We resumed shipments of the generic version Potassium Chloride Extended Release Capsule in December 2010 and we began shipping Makena™, a drug manufactured for us by a third party, in March 2011.
Even after a successful FDA inspection, we anticipate that additional data will need to be generated and submitted to the FDA with respect to certain of our other approved products before we can return them to the market, which may involve performing additional work with regard to product and formulation development. Similarly, the FDA has also informed us that, with respect to certain of our products that are subject to ANDAs or supplemented ANDAs we had submitted before entering into the consent decree, we will need to develop and/or submit additional data before those applications can be considered for approval.
The steps taken by us in connection with the nationwide recall and suspension of shipment of all products manufactured by us and the requirements under the consent decree have had, and are expected to continue to have, a material adverse effect on our results of operations. We do not expect to generate any significant revenues from products that we manufacture until we can resume shipping certain or many of our approved products. In the meantime, we must meet ongoing operating costs related to our employees, facilities and FDA compliance, as well as costs related to the steps we are currently taking to prepare for introducing or reintroducing our products to the market. If we are not able to obtain the FDA’s clearance to resume manufacturing and distribution of more of our approved products in a timely manner and at a reasonable cost, our financial position, results of operations, cash flows and liquidity will continue to be materially adversely affected.
Plea Agreement with the U.S. Department of Justice
At the direction of a special committee of the Board of Directors that was in place prior to June 10, 2010, we responded to requests for information from the Office of the United States Attorney for the Eastern District of
126
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Missouri and FDA representatives working with that office. In connection therewith, on February 25, 2010, the Board, at the recommendation of the special committee, approved entering into a plea agreement with the Department of Justice.
The plea agreement was executed by the parties and was entered by the U.S. District Court, Eastern District of Missouri, Eastern Division on March 2, 2010. Pursuant to the terms of the plea agreement, ETHEX pleaded guilty to two felony counts, each stemming from the failure to make and submit a field alert report to the FDA in September 2008 regarding the discovery of certain undistributed tablets that failed to meet product specifications. Sentencing pursuant to the plea agreement also took place on March 2, 2010.
Pursuant to the plea agreement, ETHEX agreed to pay a criminal fine in the amount of $23.4 in four installments. The first installment, in the amount of $2.3, was due and paid within 10 days of sentencing. Under the original payment schedule, the second and third installments, each in the amount of $5.9, were due on December 15, 2010 and July 11, 2011, respectively. The fourth and final installment, in the amount of $9.4, was due on July 11, 2012. On November 15, 2010, upon the motion of the Department of Justice, the court vacated the previous fine installment schedule and imposed a new fine installment schedule using the standard federal judgment rate of 0.22%, payable as follows (the Company paid the December 15, 2010 payment):
| Payment Amount including interest |
Payment Due Date | |
| $ 1.0 |
December 15, 2010 | |
| 1.0 |
June 15, 2011 | |
| 1.0 |
December 15, 2011 | |
| 2.0 |
June 15, 2012 | |
| 4.0 |
December 15, 2012 | |
| 5.0 |
June 15, 2013 | |
| 7.1 |
December 15, 2013 |
ETHEX also agreed to pay, within 10 days of sentencing, restitution to the Medicare and the Medicaid programs in the amounts of $1.8, and $0.6, respectively. In addition to the fine and restitution, ETHEX agreed not to contest an administrative forfeiture in the amount of $1.8, which was payable and paid within 45 days after sentencing and which satisfied any and all forfeiture obligations ETHEX may have as a result of the guilty plea. In total, ETHEX agreed to pay fines, restitution and forfeiture in the aggregate amount of $27.6.
In exchange for the voluntary guilty plea, the Department of Justice agreed that no further federal prosecution will be brought in the Eastern District of Missouri against ETHEX, KV and Ther-Rx regarding allegations of the misbranding and adulteration of any oversized tablets of drugs manufactured by us, and the failure to file required reports regarding these drugs and patients’ use of these drugs with the FDA, during the period commencing on January 1, 2008 through December 31, 2008.
Agreements with HHS OIG
In connection with the guilty plea described above by ETHEX, ETHEX was expected to be excluded from participation in federal healthcare programs, including Medicare and Medicaid. In addition, as a result of the guilty plea by ETHEX, HHS OIG had discretionary authority to also exclude KV from participation in federal healthcare programs. However, we are in receipt of correspondence from HHS OIG that, absent any transfer of assets or operations that would trigger successor liability, HHS OIG has no present intent to exercise its discretionary authority to exclude the Company as a result of the guilty plea by ETHEX.
127
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
In connection with the anticipated exclusion of ETHEX from participation in federal healthcare programs, we ceased the operations of ETHEX on March 2, 2010. However, we have retained the ability to manufacture, market and distribute (once the requirements under the consent decree have been met) all generic products and are in possession of all intellectual property related to generic products, including all NDAs and ANDAs pertaining to our brand and generic drug products. We currently do not anticipate that the voluntary guilty plea by ETHEX will have a material adverse effect on our efforts to comply with the requirements pursuant to the consent decree and to resume production and shipments of our approved products.
On November 15, 2010, we entered into the Divestiture Agreement with HHS OIG under which we agreed to sell the assets and operations of ETHEX to unrelated third parties prior to April 28, 2011 and to file articles of dissolution with respect to ETHEX under Missouri law by that date. We filed articles of dissolution for ETHEX on December 15, 2010. Following the filing, ETHEX may not engage in any new business other than winding up its operations and will engage in a process provided under Missouri law to identify and resolve its liabilities over at least a two-year period. Under the terms of the Divestiture Agreement, HHS OIG agreed not to exclude ETHEX from federal healthcare programs until April 28, 2011 and, upon completion of the sale of the ETHEX assets and of the filing of the articles of dissolution of ETHEX, the agreement will terminate. Civil monetary penalties and exclusion of ETHEX could have occurred if we had failed to meet our April 28, 2011 deadline. The sales of ETHEX’s assets (other than certain fixtures as to which HHS OIG has consented to non-divestiture) were completed prior to the April 28, 2011 deadline and ETHEX no longer has any ongoing assets or operations other than those required to conclude the winding up process under Missouri law. We have also received a letter from HHS OIG advising us further that assuming that we have complied with all agreements deemed necessary by HHS OIG, HHS OIG would not exclude ETHEX thereafter. On April 1, 2011, we requested confirmation from HHS OIG that the steps and actions described in our reports to HHS OIG constituted full compliance with the duties KV and ETHEX were to complete by April 28, 2011 under the Divestiture Agreement, such that they are not at risk for stipulated penalties or exclusion after that date under the Divestiture Agreement. On April 8, 2011, we received a letter from HHS OIG stating that, at this time, based upon the information provided to HHS OIG in our monthly submissions, HHS OIG has no concerns regarding the actions taken by KV and ETHEX pursuant to the Divestiture Agreement. On May 20, 2011, we received a letter from HHS OIG stating that based on its review of the information provided in our monthly reports, it appeared that the Company and ETHEX had completed our obligations under the Divestiture Agreement.
Hologic Agreement
On January 16, 2008, the Company entered into the Original Makena™ Agreement with Cytyc Prenatal Products Corp. and Hologic, Inc. (Cytyc Prenatal Products Corp. and Hologic, Inc. are referred to collectively as “Hologic”) to acquire the U.S. and worldwide royalty free, exclusive rights to Makena™ (hydroxyprogesterone caproate) and certain related assets upon approval of the pending New Drug Application for Makena™. On January 8, 2010, the Company and Hologic entered into Amendment No. 1 to the Original Makena™ Agreement, which, among other things, included a $70.0 cash payment for the exclusive rights to Makena™, which was recorded as purchased in-process research and development expense on the statement of operations for the fiscal year ended March 31, 2010. On February 4, 2011, the Company entered into Amendment No. 2 to the Original Makena™ Agreement. The amendments set forth in Amendment No. 2 reduced the payment to be made on the fifth business day following the day on which Hologic gave the Company notice that the FDA has approved Makena™ (the “Transfer Date”) to $12.5 and revised the schedule for making the remaining payments of $107.5. Under these revised payment provisions, after the $12.5 payment on the Transfer Date and a subsequent $12.5 payment 12 months after the date the FDA approved Makena™ (the “Approval Date”), the Company has the right to elect between the two alternate payment schedules for the remaining payments, with royalties of 5% of the net sales of Makena™ payable for certain periods and under different circumstances, depending on when the
128
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Company elects to make the remaining payments. The Company may make any of the payments on or before their due dates, and the date on which the Company makes the final payment contemplated by the selected payment schedule will be the final payment date, after which royalties, if any, will cease to accrue.
Under the revised payment provisions set forth in Amendment No. 2, after the $12.5 payment on the Transfer Date and a subsequent $12.5 payment twelve months after the Approval Date, the Company has the right to elect between the following two alternate payment schedules for the remaining payments:
Payment Schedule 1:
| • | A $45.0 payment 18 months after the Approval Date, plus a royalty equal to 5% of net sales of Makena™ made during the period from 12 months after the Approval Date to the date the $45.0 payment is made; |
| • | A $20.0 payment 21 months after the Approval Date; |
| • | A $20.0 payment 24 months after the Approval Date; and |
| • | A $10.0 payment 27 months after the Approval Date. |
The royalties will continue to be calculated subsequent to the $45.0 milestone payment but do not have to be paid as long as the Company makes subsequent milestone payments when due.
Payment Schedule 2:
| • | A $7.3 payment 18 months after the Approval Date, plus a royalty equal to 5% of net sales of Makena™ made during the period from 12 months after the Approval Date to 18 months after the Approval Date; |
| • | A $7.3 payment for each of the succeeding twelve months; |
| • | A royalty payable 24 months following the Approval Date equal to 5% of net sales of Makena™ made during the period from 18 months after the Approval Date to 24 months after the Approval Date; and |
| • | A royalty payable 30 months following the Approval Date equal to 5% of net sales of Makena™ made during the period from 24 months after the Approval Date to 30 months after the Approval Date. |
Notwithstanding anything to the contrary in Amendment No. 2, however, the Company may make any of the foregoing payments on or before their due dates, and the date on which the Company makes the final payment contemplated by the selected payment schedule will be the final payment date, after which no royalties will accrue.
Moreover, if the Company elects Payment Schedule 1 and thereafter elects to pay the $45.0 payment earlier than the 18-month deadline, the royalties beginning after 12 months will cease to accrue on the date of the early payment. Additionally, the subsequent payments will be paid in three month intervals following the $45.0 payment date.
Lastly, if the Company elects Payment Schedule 1 and thereafter does not make any of the milestone payments when due, Amendment No. 2 provides that no payment default will be deemed to occur, provided the Company timely pays the required royalties accruing in the quarter during which the milestone payment has become due but is not paid.
129
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Under the Indenture governing the $225 aggregate principal amount of 12% Senior Secured Notes due 2015 (the “2015 Notes”), described further below the Company agreed to make the $45 payment under Payment Schedule 1 12 months after the Approval Date and agreed to certain other restrictions on its ability to amend the payment schedules.
Makena™
On February 3, 2011, the Company was informed by Hologic that the FDA granted approval for Makena™. The Company shipped approximately 6,200 vials to its customers, specialty pharmacies and specialty distributors in March 2011. On February 14, 2011, the Company announced the initial list price at $7,500 per vial, or $1,500 per injection and on April 1, 2011 the list price was reduced to $3,450 per vial, or $690 per injection, including for all March 2011 shipments. Additionally, the Company announced on April 1, 2011 that it would expand its patient financial assistance program so that the majority of women who are clinically eligible for Makena™ would have a co-pay of no more than twenty dollars per injection.
FDA Inspections of KV
In February 2011, the FDA conducted an inspection with respect to the Company’s Clindesse® product and issued a Form 483 Report with certain observations. On February 28, 2011, the Company filed its responses with the FDA with respect to such observations.
In March 2011, the FDA conducted an inspection with respect to adverse events. The inspection was completed without any observations being issued by the FDA.
Workforce Reduction and Cost Conservation Actions
On March 30, 2010, we committed to a plan to reduce our employee workforce from 317 to 237 employees. On March 31, 2010, we implemented the plan. On February 25, 2011, the Company further reduced its workforce by 11 and laid off an additional 14 employees. On March 31, 2011, the size of our workforce was approximately 300 employees, including 97 sales representatives that work for us through a contract sales organization. The reduction in our workforce is a part of our efforts to conserve our cash and financial resources while we continue working with the FDA to return approved products to market.
On September 13, 2010, we implemented a mandatory salary deferral program for most of our exempt personnel, ranging from 15% to 25% of base salary, in order to conserve our cash and financial resources. In March 2011, the salaries of exempt personnel were reinstated.
Financing; Private Placement of Class A Common Stock; Private Placement of Notes
U.S. Healthcare Loan
On September 13, 2010, the Company entered into a loan agreement with U.S. Healthcare I, L.L.C. and U.S. Healthcare II, L.L.C., affiliates of Centerbridge Partners L.P. (collectively, “U.S. Healthcare”) for a $20.0 loan secured by assets of the Company. The loan agreement included a period of exclusivity through September 28, 2010 to negotiate an expanded, longer-term financial arrangement among the Company and U.S. Healthcare.
On November 17, 2010, the Company entered into an agreement with U.S. Healthcare for a senior secured debt financing package of up to $120.0 consisting of (1) a fully funded $60.0 term loan (the “Bridge Loan”) that retired the $20 loan previously provided by U.S. Healthcare on September 13, 2010, and that was provided for
130
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
general corporate and working capital purposes and (2) a commitment to provide a multi-draw term loan up to an aggregate principal amount of $120 (the “Multi-Draw Term Loan”) with such additional draws dependent on the achievement by the Company of various conditions as outlined in the related agreement. The Company expensed approximately $1.9 of unamortized deferred financing costs related to the retirement of the $20 as required by accounting for debt extinguishments in the quarter ended December 31, 2010.
Under the terms of the Bridge Loan agreement, the Company paid interest at an annual rate of 16.5% (5% of which was payable in kind) with a maturity date in March 2013. The Company furnished as collateral substantially all assets of the Company to secure the loan. The Bridge Loan was guaranteed by certain of the Company’s domestic subsidiaries and the guarantors furnished as collateral substantially all of their assets to secure the guarantee obligations. In addition, the Company issued stock warrants to U.S. Healthcare granting them rights to purchase up to 12.588 million shares of the Company’s Class A Common Stock (the “Initial Warrants”). The Initial Warrants have an exercise price of $1.62 per share, subject to possible standard anti-dilutive adjustment. These Initial Warrants were valued at $24.0 using a Black-Scholes option pricing model utilizing the following assumptions: risk free rate of 1.5%; expected volatility of 99.0%; expected dividend of $0; and expected life of five years.
In recording the Bridge Loan transaction, the Company allocated the proportionate share of the fair value of the Initial Warrants, to the September loan. As a result of the proceeds from the Bridge Loan extinguishing the September loan, the fair value of the Initial Warrants of $8.0 allocated to the September loan was expensed as required by accounting for debt extinguishments in the quarter ended December 31, 2010.
The company then allocated, at their relative fair value, the proportionate share of the fair value of the Initial Warrants of $11.4 as a discount to the Bridge Loan. The discount was being amortized using the effective interest method to interest expense based upon the maturity date of the Bridge Loan. In March 2011, the Company retired the Bridge Loan and expensed the remaining discount in loss on extinguishment of debt.
The $120.0 Multi-Draw Term Loan consisted of three tranches that would have been available to the Company following the achievement of certain conditions. The first tranche of $80.0 would have been available upon the approval of Makena™ and would have been used to repay the Bridge Loan of $60.0, make a milestone payment to Hologic, and provide funds for general corporate and working capital purposes. The second tranche of $20.0 would have been available to the Company upon achieving at least one of certain performance thresholds including either, (1) certain metrics associated with Evamist®, or (2) receiving FDA approval for the manufacture and distribution of Clindesse® and Gynazole-1®. The proceeds of the second tranche would have been used for general corporate and working capital purposes. The third tranche of $20.0 would have been available to the Company upon evidencing its ability, to the satisfaction of U.S. Healthcare, to meet certain liquidity thresholds necessary to satisfy future obligations, including a future milestone payment to Hologic that is due to be paid one year following FDA approval of Makena™. The proceeds from the third tranche would have been used for general corporate and working capital purposes.
The Company and U.S. Healthcare amended the financing arrangements on January 6, 2011 and again on March 2, 2011. Pursuant to the amendments, the Company and U.S. Healthcare amended the Bridge Loan terms and covenants to reflect the Company’s then current projections and timing of certain anticipated future events, including the planned disposition of certain assets. The amendments extended the $60.0 payment that was due on March 20, 2011 to three payments of $20.0 each with the first payment due (and paid on February 18, 2011) upon closing and funding the private placement of Class A Common Stock, $20.0 due in April 2011 and $20.0 due in August 2011. In addition, all past covenant issues were waived. As a result of the amendments, the Company would not have been required to sell its generics business by March 20, 2011, but would have been
131
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
required to cause such sale by August 31, 2011. In addition, the applicable premium (a make-whole payment of interest with respect to payments on the loans prior to maturity) was amended to provide that if the Bridge Loan was repaid in full as a result of a refinancing transaction provided other than by U.S. Healthcare, as occurred on March 17, 2011 with the issuance of the 2015 Notes, a premium was paid to U.S. Healthcare equal to $12.5, of which $7.2 was paid in connection with the private placement of Class A Common Stock and the rest was paid on March 17, 2011 and recorded as a loss on extinguishment of debt. In addition, on March 17, 2011, an amount of $7.5 was placed in escrow and will be released to the Company or to U.S. Healthcare on August 31, 2011 or September 30, 2011, as the case may be, depending on the status of the Company’s registration process with the SEC by such dates of the shares underlying the warrants and the Company’s stock price meeting certain specified levels as of the applicable date. In connection with the amendments and certain waivers granted by U.S. Healthcare, the Company issued additional warrants to U.S. Healthcare to purchase up to 7.5 million shares of the Company’s Class A Common Stock, at an exercise price of $1.62 per share, and amended and restated the Initial Warrants (We refer to the additional warrants and the Initial Warrants collectively as the “Warrants”). See Note 24—“Warrant Liability” for more information on the Warrants.
The Multi-Draw Term Loan, as amended, provided for a total commitment of $118.0. If entered into, the Multi-Draw Term Loan, as amended, would have refinanced the Bridge Loan in full and would have provided $70.0 of additional financing consisting of (1) a $30.0 tranche B-2 term loan and (2) a $40.0 tranche B-3 term loan. The withdrawal schedule under the Multi-Draw Term Loan was revised to allow for release of funds from controlled accounts on the closing date sufficient to repay the Bridge Loan and future draws against the Multi-Draw Term Loan, subject to achievement of certain Makena™ related milestones, of $15.0 in March 2011, $15.0 in May 2011 and $10.0 in each of July, August, September and October 2011. The commitment letter for the Multi-Draw Term Loan would have expired on March 31, 2011.
On February 17, 2011, the Company repaid a portion of the Bridge Loan with proceeds from a private placement of Class A Common Stock, described below, and on March 17, 2011, the Company repaid in full the remaining obligations under the Bridge Loan (including the payment of related premiums) with a portion of the proceeds of the offering of the 2015 Notes, described below (and terminated the related future loan commitments).
In addition to the $1.9 and the $8.0 debt extinguishment charges described above, the March 2011 debt extinguishment resulted in the write-off of related unamortized deferred financing costs of $3.9, the $7.5 escrow-payment, the $12.5 premium payment and the expense associated with the 7.5 million warrants of $72.2, for an aggregate loss on debt extinguishment of $106.2.
Private Placement of Class A Common Stock
On February 14, 2011, the Company announced that it entered into a definitive agreement with a group of institutional investors to raise approximately $32.3 of gross proceeds from a private placement of 9.95 shares of its Class A Common Stock at $3.25 per share. The transaction closed on February 17, 2011. The Company used $20.0 of the proceeds from the financing to repay certain outstanding amounts and other outstanding obligations under its credit agreement with U.S. Healthcare. The remaining amount is being used for the launch of Makena™, payment of expenses associated with the private placement and general corporate purposes.
The Company will be required to pay certain cash amounts as liquidated damages at a rate of 1.5% per month of the aggregate purchase price of the shares that are not registered per month if it does not meet certain obligations under the agreement with respect to the registration of shares. No such damages are recorded as obligations as of March 31, 2011 as the Company expects to timely register the shares.
132
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Private Placement of the 2015 Notes
On March 17, 2011, the Company completed the offering and sale of the 2015 Notes. The 2015 Notes bear interest at an annual rate of 12% per year, payable semiannually in arrears on March 15 and September 15 of each year, commencing September 15, 2011. The 2015 Notes will mature March 15, 2015. At any time prior to March 15, 2013, the Company may redeem up to 35% of the aggregate principal amount of the 2015 Notes at a redemption price of 112% of the principal amount of the 2015 Notes, plus accrued and unpaid interest to the redemption date, with the net cash proceeds of one or more equity offerings. At any time prior to March 15, 2013, the Company may redeem all or part of the 2015 Notes at a redemption price equal to (1) the sum of the present value, discounted to the redemption date, of (i) a cash payment to be made on March 15, 2013 of 109% of the principal amount of the 2015 Notes, and (ii) each interest payment that is scheduled to be made on or after the redemption date and on or before March 15, 2013, plus (2) accrued and unpaid interest to the redemption date. At any time after March 15, 2013 and before March 15, 2014, the Company may redeem all or any portion of the 2015 Notes at a redemption price of 109% of the principal amount of the 2015 Notes, plus accrued and unpaid interest to the redemption date. At any time after March 15, 2014, the Company may redeem all or any portion of the 2015 Notes at a redemption price of 100% of the principal amount of the 2015 Notes, plus accrued and unpaid interest to the redemption date. The 2015 Notes are secured by the assets of the Company and certain assets of its subsidiaries.
After an original issue discount of 3%, the Company received proceeds of $218.3 which were used to fund a first-year interest reserve totaling $27.0 (reflected as restricted cash on the balance sheet), repay all existing obligations to U.S. Healthcare totaling approximately $61.1 and pay fees and expenses associated with the offering of the 2015 Notes of approximately $10.0. In connection with these payments, the Company also terminated all future loan commitments with U.S. Healthcare. The remaining proceeds, totaling approximately $120.0 will be used for general corporate purposes, including the launch of Makena™.
The 2015 Notes were offered only to accredited investors pursuant to Regulation D under the Securities Act of 1933, as amended.
New Subsidiary
In May 2010, we formed a wholly-owned subsidiary, Nesher, to operate as the sales and marketing company for our generic products. In July 2010, our Board of Directors directed management to explore strategic alternatives with respect to Nesher and the assets and operations of our generic products business, which could include a sale of Nesher. We have retained Jefferies & Co., Inc. to advise us with this strategy. In the meantime, we will continue to prepare products for FDA inspection and continue to anticipate the reintroduction of approved products into the market.
Refer also to Note 23—“Divestitures” for discussion of recent events and developments related to Nesher and our generics business.
| 2. | Summary of Significant Accounting Policies |
Basis of Presentation
The Company’s consolidated financial statements are prepared in accordance with U.S. generally accepted accounting principles (“GAAP”). The consolidated financial statements include the accounts of the Company and its wholly-owned subsidiaries. All material inter-company accounts and transactions have been eliminated in
133
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
consolidation. Certain reclassifications, none of which affected net income (loss) or accumulated deficit, have been made to prior year amounts to conform to the current year presentation.
Reclassification
Certain reclassifications of prior year amounts have been made to conform to the current year presentation.
Discontinued Operations
During the fourth quarter of the fiscal year 2009, the Board of Directors authorized management to sell PDI. During the fourth quarter of fiscal year 2011 the Company committed to a plan to divest its generics business. We sold PDI on June 2, 2010. The Company identified the assets and liabilities of PDI and its generics business as held for sale in the Company’s consolidated balance sheet at March 31, 2011 and 2010 and has segregated PDI’s and the generics business’s operating results separately for all periods presented. See Note 24—“Divestitures” for information regarding the sale of PDI and generics business.
Use of Estimates
The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities, the disclosure of contingent liabilities at the date of the financial statements, and the reported amounts of revenues and expenses during the reporting period. Actual results in subsequent periods may differ from the estimates and assumptions used in the preparation of the accompanying consolidated financial statements.
The most significant estimates made by management include revenue recognition and reductions to gross revenues, inventory valuation, intangible and other long-lived assets valuation, stock-based compensation, income taxes, and loss contingencies related to legal proceedings. Management periodically evaluates estimates used in the preparation of the consolidated financial statements and makes changes on a prospective basis when adjustments are necessary.
Cash and Cash Equivalents
Cash and cash equivalents consist of interest-bearing deposits that can be redeemed on demand and investments that have original maturities of three months or less.
Restricted Cash
Restricted cash consists of (1) $27.0 to fund a first-year interest reserve for the 2015 Notes; and (2) $7.5 placed in escrow and to be released to the Company or U.S. Healthcare on August 31, 2011 or September 30, 2011, as the case may be, depending on the status of the Company’s registration process with the SEC by such dates and the Company’s stock price meeting certain specified levels as of the applicable date.
Investment Securities
The Company’s investment securities consist of auction rate securities (“ARS”) that are accounted for as available-for-sale securities. Available-for-sale securities are carried at fair value with temporary unrealized gains and losses, net of tax, recorded within other comprehensive income (loss) as a separate component of shareholders’ equity. The Company has no trading or held-to-maturity securities. The cost of investment securities sold is determined by the specific identification method. Dividend and interest income are accrued as earned. The Company reviews its investments quarterly for declines in fair value that are other-than-temporary. Investments that have declined in market value that are determined to be other-than-temporary are charged to other income (expense), net, by writing that investment down to fair value.
134
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
ARS generally have long-term stated maturities of 20 to 30 years. However, these securities have certain economic characteristics of short-term investments due to a rate-setting mechanism and the ability to liquidate them through a Dutch auction process that occurs at pre-determined intervals of up to 35 days. The Company classified these securities as non-current investment securities at March 31, 2010 to reflect the current lack of liquidity in these investments. The Company classified the securities as short-term at March 31, 2011 as the settlement agreement the Company entered into expires on January 21, 2012. See Note 6—“Investment Securities” concerning liquidity and also for information regarding a settlement agreement the Company entered into regarding its holdings of ARS and the proceeds received in connection therewith.
Inventories
Inventories consist of finished goods held for distribution, raw materials and work in process. Inventories are stated at the lower of cost or market, with the cost determined on the first-in, first-out (FIFO) basis. Reserves for obsolete, excess or slow-moving inventory are established by management based on evaluation of inventory levels, forecasted demand and market conditions.
Inventories also include costs related to certain products that are pending regulatory approval. From time to time, the Company capitalizes inventory costs associated with products prior to regulatory approval based on management’s judgment of probable future regulatory approval, commercial success and realizable value. Such judgment incorporates the Company’s knowledge and best estimate of where the product is in the regulatory review process, the Company’s required investment in the product, market conditions, competing products and the Company’s economic expectations for the product post-approval relative to the risk of manufacturing the product prior to approval. If final regulatory approval for such products is denied or delayed, the Company revises its estimates and judgments about the recoverability of the capitalized costs and, where required, provides reserves for such inventory in the period those estimates and judgments change.
During fiscal year 2009, the Company announced six separate voluntary recalls of certain tablet-form generic products as a precaution due to the potential existence of oversized tablets. Beginning in December 2008, the Company determined that it was not able to establish the recoverability of production related inventory costs because of uncertainties associated with the risk of additional product recalls. The Company ceased all manufacturing activities during the fourth quarter of fiscal year 2009, and its net revenues since then have been limited to sales of products manufactured by third parties except for Potassium Chloride Extended Release Capsule which we began shipping in September 2010. As a result, most costs associated with the Company’s manufacturing operations were recognized directly into cost of sales rather than capitalized into inventory during fiscal years ended March 31, 2011 and 2010.
Property and Equipment
Property and equipment are stated at cost, less accumulated depreciation. Major renewals and improvements are capitalized, while routine maintenance and repairs are expensed as incurred. At the time properties are retired from service, the cost and accumulated depreciation are removed from the respective accounts and any related gains or losses are reflected in earnings. The Company capitalizes interest on qualified construction projects.
Depreciation expense is computed over the estimated useful lives of the related assets using the straight-line method. The estimated useful lives are principally 10 years for land improvements, 10 to 40 years for buildings and improvements, 3 to 15 years for machinery and equipment, and 3 to 10 years for office furniture and equipment. Leasehold improvements are amortized on a straight-line basis over the shorter of the respective lease terms or the estimated useful life of the assets.
135
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
The Company assesses property and equipment for impairment whenever events or changes in circumstances indicate that the carrying value may not be recoverable. Certain significant events occurred in the three months ended March 31, 2011 that indicated that the carrying value of certain assets as of March 31, 2011 were not recoverable. Also, the Company recorded impairment charges in fiscal year 2010. Refer to the “Impairment of Long-Lived Assets” section below, to Note 4—“Restructuring and Impairment Charges” and to Note 9—“Property and Equipment” for further information regarding the assessment of property and equipment for impairment of assets.
Intangible Assets
Intangible assets consist of product rights, license agreements and trademarks resulting from product acquisitions and legal fees and similar costs relating to the development of patents and trademarks. Intangible assets that are acquired are stated at cost, less accumulated amortization, and are amortized on a straight-line basis over estimated useful lives ranging from 7 to 20 years. Costs associated with the development of patents and trademarks are amortized on a straight-line basis over estimated useful lives ranging from 5 to 17 years.
The Company evaluates its intangible assets for impairment at least annually or whenever events or changes in circumstances indicate that an intangible asset’s carrying amount may not be recoverable. Recoverability is determined by comparing the carrying amount of an intangible asset to an estimate of the undiscounted future cash flows expected to result from its use and eventual disposition. If the sum of the expected future undiscounted cash flows is less than the carrying amount of the intangible asset, an impairment loss is recognized based on the excess of the carrying amount over the estimated fair value of the intangible asset. Certain significant events occurred in 2010 and 2009 that indicated that the carrying value of certain intangible assets were not recoverable. Refer to the “Impairment of Long-Lived Assets” section below, to Note 4—“Restructuring and Impairment” and to Note 10—“Intangible Assets” for further information regarding the assessment of intangible assets for impairment.
Impairment of Long-Lived Assets
The Company assesses the impairment of its assets whenever events or changes in circumstances indicate that the carrying value may not be recoverable. The factors that the Company considers in its assessment include the following: (1) significant underperformance of the assets relative to expected historical or projected future operating results; (2) significant changes in the manner of the Company’s use of the acquired assets or the strategy for its overall business; (3) significant negative industry or economic trends; and (4) significant adverse changes as a result of legal proceedings or governmental or regulatory actions (see Note 4—“Restructuring and Impairment” for further information regarding the assessment of long-lived assets for impairment).
Non-marketable Equity Investments
Non-marketable equity investments for which the Company does not have the ability to exercise significant influence over operating and financial policies (generally less than 20% ownership) are accounted for using the cost method. Such investments are included in “Other assets” in the accompanying consolidated balance sheets and relate primarily to the Company’s investment at March 31, 2010 in the preferred stock of Strides Arcolab Limited (“Strides”).
On December 13, 2010, the Company and Strides entered into a Settlement Agreement and Release to settle all disputes and claims against each other related to a previously existing License and Supply Agreement between the two companies. Under the terms of the agreement, Strides paid the Company $7.3 and the Company
136
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
agreed to redeem the preferred shares of Strides that it owns. Accordingly, the carrying value of Strides was reduced to $7.3 as of March 31, 2010. The Company received $7.3 during the fiscal year ended March 31, 2011. See Note 15—“Commitments and Contingencies.”
Revenue Recognition
Revenue is generally realized or realizable and earned when persuasive evidence of an arrangement exists, the customer’s payment ability has been reasonably assured, title and risk of ownership have been transferred to the customer, and the seller’s price to the buyer is fixed or reasonably determinable. The Company also enters into long-term agreements under which it assigns marketing rights for products it has developed to pharmaceutical markets. Royalties under these arrangements are earned based on the sale of products.
Beginning in December 2008, the Company determined it was not able to reasonably estimate future product returns at the time its drug products were shipped because of uncertainties associated with the risk of additional product recalls. As a result, revenue on shipments of its drug products was deferred until such uncertainties were resolved, which occurred on the earlier of the consumption by the end user or when these products were no longer subject to recall.
Product shipments that are recorded as deferred revenue are reflected as a current liability on the consolidated balance sheet. For shipments where revenue recognition is deferred, the Company records revenue when the uncertainties leading to deferral are resolved. Since the Company suspended all shipments of its approved drug products in tablet form in December 2008 and suspended the manufacturing and shipment of the remainder of its products in January 2009, the product recall became probable and estimable in the quarter ended March 31, 2009. Thus, the Company determined that the uncertainties leading to deferral were resolved, and it did not have any deferred revenue recorded on its consolidated balance sheet at March 31, 2011 or 2010.
Concurrent with the recognition or deferral of revenue, the Company records estimated provisions for product returns, sales rebates, payment discounts, chargebacks, and other sales allowances. When the occurrence of product recalls becomes probable, the Company also records estimated liabilities for product returns related to recalls. The Company records failure to supply claims when they are probable and reasonably estimable. Provisions are established based upon consideration of a variety of factors, including but not limited to, historical relationship to revenues, historical payment and return experience, estimated and actual customer inventory levels, customer rebate and failure to supply arrangements, current contract sales terms with wholesale and indirect customers, and subsequent payment activity. The following briefly describes each provision and how such provisions are estimated.
| • | Cash Discounts—Payment discounts are reductions to invoiced amounts offered to customers for payment within a specified period and are estimated utilizing historical customer payment experience. |
| • | Sales Rebates—Sales rebates are offered to certain customers to promote customer loyalty and encourage greater product sales. These rebate programs provide that, upon the attainment of pre-established volumes or the attainment of revenue milestones for a specified period, the customer receives credit against purchases. Other promotional programs are incentive programs periodically offered to customers. Due to the nature of these programs, the Company is able to estimate provisions for rebates and other promotional programs based on the specific terms in each agreement. |
| • | Sales Returns—Consistent with common industry practices, the Company has agreed to terms with its customers to allow them to return product that is within a certain period of the expiration date, although the Company is not always obligated to take product back from its customers. Upon recognition of revenue from product sales to customers, the Company provides for an estimate of product to be returned. This estimate is determined by applying a historical relationship of customer returns to amounts invoiced. |
137
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
| • | Chargebacks—The Company markets and sells products directly to wholesalers, distributors, warehousing pharmacy chains, mail order pharmacies and other direct purchasing groups. The Company also markets products indirectly to independent pharmacies, non-warehousing chains, managed care organizations, and group purchasing organizations, collectively referred to as “indirect customers.” The Company enters into agreements with some indirect customers to establish contract pricing for certain products. These indirect customers then independently select a wholesaler from which to purchase the products at these contracted prices. Alternatively, the Company may pre-authorize wholesalers to offer specified contract pricing to other indirect customers. Under either arrangement, the Company provides credit to the wholesaler for any difference between the contracted price with the indirect customer and the wholesaler’s invoice price. This credit is called a chargeback. Provisions for estimated chargebacks are calculated primarily using historical chargeback experience, actual contract pricing, estimated and actual wholesaler inventory levels, and subsequent payment activity. |
| • | Price Protection—Generally, the Company provides credits to wholesale customers for decreases that are made to selling prices for the value of inventory that is owned by these customers at the date of the price reduction. These credits are customary in the industry and are intended to reduce a wholesale customer’s inventory cost to better reflect current market prices. Since a reduction in the wholesaler’s invoice price reduces the chargeback per unit, price reduction credits are typically included as part of the reserve for chargebacks because they act essentially as accelerated chargebacks. Although the Company contractually agreed to provide price adjustment credits to its major wholesale customers at the time they occur, the impact of any such price reductions not included in the reserve for chargebacks is immaterial to the amount of revenue recognized in any given period. |
| • | Medicaid Rebates—Federal law requires that a pharmaceutical manufacturer, as a condition of having its products receive federal reimbursement under Medicaid and Medicare Part B, must pay rebates to state Medicaid programs for all units of its pharmaceuticals that are dispensed to Medicaid beneficiaries and paid for by a state Medicaid program. The provision for Medicaid rebates is based upon historical experience of claims submitted by the various states. The Company also monitors Medicaid legislative changes to determine what impact such legislation may have on the provision for Medicaid rebates. |
| • | Product Recall Returns—Liabilities for product returns related to recalls are based on estimated and actual customer inventory levels at the time of the recall and actual contract pricing. |
| • | Failure to Supply— The Company has entered into purchase agreements with certain customers that include a provision whereby the Company is required to reimburse these customers for price differences on product orders that the Company is unable to fulfill. The Company estimates this liability based on the terms of the agreements. |
Actual product returns, chargebacks and other sales allowances incurred are dependent upon future events and may be different from the Company’s estimates. The Company continually monitors the factors that influence sales allowance estimates and makes adjustments to these provisions when management believes that actual product returns, chargebacks and other sales allowances may differ from established allowances.
138
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
The provisions discussed above are presented in the consolidated financial statements as reductions to gross revenues or deferred revenue and a decrease to accounts receivable or an increase to accrued liabilities. Provisions totaled $39.1 and $6.2 for the fiscal years ended March 31, 2011 and 2010, respectively. The reserve balances related to the provisions are included in “Receivables, net” or “Accrued liabilities” in the accompanying consolidated balance sheets. A summary of fiscal year 2011 and 2010 changes for each reserve or liability follows:
| Beginning Balance |
Additions | Reductions | Ending Balance |
|||||||||||||
| Year Ended March 31, 2011 |
||||||||||||||||
| Accounts Receivable Reserves: |
||||||||||||||||
| Chargebacks |
$ | — | $ | 3.3 | $ | (0.4 | ) | $ | 2.9 | |||||||
| Cash Discounts and Other Allowances |
0.1 | 1.3 | (0.4 | ) | 1.0 | |||||||||||
| Liabilities: |
||||||||||||||||
| Sales Rebates |
1.0 | 2.2 | (2.1 | ) | 1.1 | |||||||||||
| Sales Returns |
7.3 | 2.1 | (4.6 | ) | 4.8 | |||||||||||
| Medicaid Rebates |
3.6 | 3.3 | — | 6.9 | ||||||||||||
| Product Recall Returns |
2.9 | 0.2 | — | 3.1 | ||||||||||||
| Failure to Supply Claims |
12.3 | — | (2.3 | ) | 10.0 | |||||||||||
| Price Protection |
— | 26.0 | (0.2 | ) | 25.8 | |||||||||||
| Other |
0.3 | 0.7 | (0.3 | ) | 0.7 | |||||||||||
| Total |
$ | 27.5 | $ | 39.1 | $ | (10.3 | ) | $ | 56.3 | |||||||
| Beginning Balance |
Additions | Reductions | Ending Balance |
|||||||||||||
| Year Ended March 31, 2010 |
||||||||||||||||
| Accounts Receivable Reserves: |
||||||||||||||||
| Chargebacks |
$ | — | $ | 0.1 | $ | (0.1 | ) | $ | — | |||||||
| Cash Discounts and Other Allowances |
0.2 | 0.2 | (0.3 | ) | 0.1 | |||||||||||
| Liabilities: |
||||||||||||||||
| Sales Rebates |
4.6 | 1.2 | (4.8 | ) | 1.0 | |||||||||||
| Sales Returns |
12.0 | 3.7 | (8.4 | ) | 7.3 | |||||||||||
| Medicaid Rebates |
6.3 | 0.7 | (3.4 | ) | 3.6 | |||||||||||
| Medicare and Medicaid restitution |
2.3 | — | (2.3 | ) | — | |||||||||||
| Product Recall Returns |
40.7 | — | (37.8 | ) | 2.9 | |||||||||||
| Failure to Supply Claims |
17.1 | — | (4.8 | ) | 12.3 | |||||||||||
| Other |
2.6 | 0.3 | (2.6 | ) | 0.3 | |||||||||||
| Total |
$ | 85.8 | $ | 6.2 | $ | (64.5 | ) | $ | 27.5 | |||||||
The liabilities for sales rebates, sales returns, Medicaid rebates, Medicare and Medicaid restitution, product recall returns, failure to supply claims, price protection and chargeback audit adjustments are classified as accrued liabilities in the consolidated balance sheets as of March 31, 2011 and 2010. The expense associated with these accruals is classified in the consolidated statement of operations for the year ended March 31, 2011 as a reduction of gross revenue.
Concentration of Credit Risk
The Company extends credit on an uncollateralized basis primarily to wholesale drug distributors and retail pharmacy chains throughout the U.S. As a result, the Company is required to estimate the level of receivables
139
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
which ultimately will not be paid. The Company calculates this estimate based on prior experience supplemented by a customer specific review when it is deemed necessary. On a periodic basis, the Company performs evaluations of the financial condition of all customers to further limit its credit risk exposure. Actual losses from uncollectible accounts have historically been insignificant.
The Company’s three largest customers accounted for approximately 38.5%, 29.9% and 14.4% and 56.5%, 42.8% and 0.7% of gross receivables at March 31, 2011 and 2010, respectively.
For the fiscal year ended March 31, 2011, the Company’s three largest customers accounted for 17.3%, 16.6% and 11.2% of gross revenues, respectively. For the fiscal years ended March 31, 2010 and 2009, the Company’s three largest customers accounted for 37.3%, 35.8% and 11.1% and 44.5%, 33.2% and 9.2% of gross revenues, respectively.
The Company maintains cash balances at certain financial institutions that are greater than the federally insurable limit.
Shipping and Handling Costs
The Company classifies shipping and handling costs in cost of sales. The Company does not derive revenue from shipping.
Research and Development
Research and development costs, including licensing fees for early stage development products, are expensed in the period incurred.
The Company has licensed the exclusive rights to co-develop and market various products with other drug delivery companies. These collaborative agreements usually require the Company to pay up-front fees and ongoing milestone payments. These costs are expensed as research and development expense. Payments made to third parties subsequent to regulatory approval are capitalized with that cost generally amortized over the shorter of the life of the product or the term of the licensing agreement.
The Company accrues estimated costs associated with clinical studies performed by contract research organizations based on the total of costs incurred through the balance sheet date. The Company monitors the progress of the trials and their related activities to the extent possible, and adjusts the accruals accordingly. These accrued costs are recorded as a component of research and development expense.
Advertising
Costs associated with advertising are expensed in the period in which the advertising is used and these costs are included in selling and administrative expense. Advertising expenses totaled $4.5, $6.0 and $31.4 for the years ended March 31, 2011, 2010 and 2009, respectively. Advertising expense includes the cost of product samples given to physicians.
Litigation
The Company is subject to litigation in the ordinary course of business and to certain other contingencies (see Note 15—“Commitments and Contingencies”). Legal fees and other expenses related to litigation and
140
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
contingencies are recorded as incurred. The Company, in consultation with its legal counsel, also assesses the need to record a liability for litigation and contingencies on a case-by-case basis. Accruals are recorded when the Company determines that a loss related to a matter is both probable and reasonably estimable.
Deferred Financing Costs
Deferred financing costs of $9.7 and $5.9 were incurred in connection with the issuance of 2015 Notes and convertible debt, respectively. The 2015 Notes costs are being expensed on a straight line basis into interest expense over the term of the 2015 Note. The costs incurred with the convertible debt were being amortized into interest expense on a straight-line basis over the five-year period that ended on May 16, 2008, the first date the debt could be put by the holders to the Company. Unamortized deferred financing costs associated with the other debt that was retired during the fiscal year ended March 31, 2011 are included in loss on debt extinguishment upon retirement.
Earnings Per Share
The Company has two classes of common stock: Class A Common Stock and Class B Common Stock that is convertible into Class A Common Stock. With respect to dividend rights, holders of Class A Common Stock are entitled to receive cash dividends per share equal to 120% of the dividends per share paid on the Class B Common Stock. For purposes of calculating basic earnings (loss) per share, undistributed earnings (loss) are allocated to each class of common stock based on the contractual participation rights of each class of security.
The Company presents diluted earnings (loss) per share for Class B Common Stock for all periods using the two-class method which does not assume the conversion of Class B Common Stock into Class A Common Stock. The Company presents diluted earnings (loss) per share for Class A Common Stock using the if-converted method which assumes the conversion of Class B Common Stock into Class A Common Stock, if dilutive.
Basic earnings (loss) per share is computed using the weighted average number of common shares outstanding during the period, except that it does not include unvested common shares subject to repurchase. Diluted earnings (loss) per share is computed using the weighted average number of common shares and, if dilutive, potential common shares outstanding during the period. Potential common shares consist of the incremental common shares issuable upon the exercise of stock options, unvested common shares subject to repurchase, convertible preferred stock and the convertible notes. The dilutive effects of outstanding stock options and unvested common shares subject to repurchase are reflected in diluted earnings (loss) per share by application of the treasury stock method. Shares related to convertible preferred stock and convertible notes are reflected on an if-converted basis. The computation of diluted earnings (loss) per share for Class A Common Stock assumes the conversion of the Class B Common Stock, while the diluted earnings (loss) per share for Class B Common Stock does not assume the conversion of those shares.
Income Taxes
Income taxes are accounted for under the asset and liability method where deferred tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement carrying amount of existing assets and liabilities and the respective tax basis and for tax credit carry forwards. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that includes the enactment date. A valuation allowance is established when it is more likely than not that some portion or all of the deferred tax assets will not be realized. The Company evaluates the realizability of its deferred tax assets by assessing its
141
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
valuation allowance and by adjusting the amount of such allowance, if necessary. The factors used to assess the likelihood of realization include historical cumulative losses, the forecast of future taxable income and available tax planning strategies that could be implemented to realize the net deferred tax assets. Failure to achieve forecasted taxable income in applicable tax jurisdictions could affect the ultimate realization of deferred tax assets and could result in an increase in the Company’s effective tax rate on future earnings.
The Company accounts for uncertain tax positions in accordance with the authoritative guidance issued by the FASB. The authoritative guidance addresses the determination of whether tax benefits claimed or expected to be claimed on a tax return should be recorded in the financial statements. The Company may recognize the tax benefit from an uncertain tax position only if it is more likely than not that the tax position will be sustained on examination by the taxing authorities, based on the technical merits of the position. The tax benefits recognized in the financial statements from such a position should be measured based on the largest benefit that has a greater than 50% likelihood of being realized upon ultimate settlement. The Company recognizes interest and penalties, if any, related to unrecognized tax benefits in income tax expense.
Stock-Based Compensation
The Company accounts for stock-based compensation in accordance with the authoritative guidance issued by the FASB for share-based payments, which requires the Company to measure and record compensation expense, based on estimated fair values, for all share-based compensation awards made to employees and directors over the vesting period of the awards.
The Company estimates the fair value of stock options granted using the Black-Scholes option pricing model, which requires the use of subjective and complex assumptions. These assumptions include estimating the expected term that options granted are expected to be outstanding, the expected volatility of the Company’s stock price for a period commensurate with the expected term of the related options, and the risk-free rate with a maturity closest to the expected term of the Company’s stock options.
Segment Reporting
The Company had two reportable segments—branded products and specialty generic/non-branded products. The Company has committed to a plan to divest the generics business and reported its generics business as discontinued operations. The Company’s Chief Operating Decision Maker and Board of Directors review profit and loss information on the remaining business on a consolidated basis to assess performance, make overall operating decisions and make resource allocations. The Company’s activities are closely interrelated and share services. Based on these factors, the Company has concluded that it currently operates in one segment.
Comprehensive Income (Loss)
Comprehensive income (loss) includes all changes in equity during a period except those that resulted from investments by or distributions to the Company’s shareholders. Other comprehensive income (loss) refers to revenues, expenses, gains and losses that, under GAAP, are included in comprehensive income (loss), but excluded from net income (loss) as these amounts are recorded directly as an adjustment to shareholders’ equity (deficit). For the Company, other comprehensive income (loss) is comprised of the net changes in unrealized gains and losses on available-for-sale securities and the foreign currency translation adjustments recorded as result of translating Nesher Solutions Ltd. balances from Israeli shekels to U.S. dollars. As of March 31, 2011, the accumulated other comprehensive income (loss) includes a $1.7 unrealized gain on the auction rate securities. As of March 31, 2010, the accumulated other comprehensive income (loss) balance includes a $1.6 unrealized gain on the auction rate securities.
142
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Fair Value of Financial Instruments
The Company’s financial instruments consist primarily of cash and cash equivalents, receivables, investments, trade accounts payable, the convertible debt, embedded derivatives related to the issuance of the convertible debt and a mortgage loan arrangement. The carrying amounts of cash and cash equivalents, receivables and trade accounts payable are representative of their respective fair values due to their relatively short maturities.
Based on a valuation analysis, the Company’s investments in ARS of $61.5 and $71.6 had a fair value of $57.2 and $65.9 at March 31, 2011 and 2010, respectively (see Note 7—“Fair Value Measures”).
The fair and carrying value of the Company’s investment in the preferred stock of Strides at March 31, 2010 was $7.3.
Based on quoted market rates, the Company’s convertible debt had a fair value of $150.0 and $101.0 at March 31, 2011 and 2010, respectively. The carrying amount of the mortgage loan and 2015 Notes arrangement approximates its fair value because its terms are similar to those which can be obtained for similar financial instruments in the current marketplace.
Derivative Financial Instruments
The Company’s derivative financial instruments consist of embedded derivatives related to the convertible debt. These embedded derivatives include certain conversion features and a contingent interest feature. Although the conversion features represent embedded derivative financial instruments, based on the de minimis value of these features at the time of issuance and at March 31, 2011, no value has been assigned to these embedded derivatives. The contingent interest feature provides unique tax treatment under the Internal Revenue Service’s contingent debt regulations. In essence, interest accrues, for tax purposes, on the basis of the instrument’s comparable yield (the yield at which the issuer would issue a fixed rate instrument with similar terms).
Foreign Currency
For the Company’s sole foreign operating entity, Nesher Solutions Ltd. the local currency is the functional currency. The Company translates asset and liability balances at exchange rates in effect at the end of the period and income and expense transactions at the average exchange rates in effect during the period. Resulting translation adjustments are reported as a separate component of accumulated other comprehensive income (loss) included in shareholders’ equity (deficit).
Gains and losses from foreign currency transactions are included in the consolidated statements of operations as part of interest and other expense (income). The amount of foreign currency gains (losses) included in the Company’s consolidated statement of operations was $0, $1.2 and $(2.6) for the fiscal years ended March 31, 2011, 2010 and 2009, respectively.
Authoritative accounting guidance issued by the FASB requires that the effect of exchange rate changes on cash held in foreign currencies be reported as a separate item in the reconciliation of beginning and ending cash and cash equivalents. All other foreign currency cash flows are reported in the applicable line of the consolidated statement of cash flows using an approximation of the exchange rate in effect at the time of the cash flows.
Accounting Standards Adopted
The Company adopted FASB Accounting Standards Codification (“ASC”) 2010-27-Other Expenses (Topic 720): Fees Paid to the Federal Government by Pharmaceutical Manufacturers (A consensus of the FASB
143
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Emerging Issues Task Force). The update addresses questions concerning how pharmaceutical manufactures should recognize and classify in their income statements fees mandated by the Patient Protection and Affordable Care Act as amended by the Health Care and Education Reconciliation Act.
Based on the Company’s 2011 product base and overall sales as a percentage of the industry, management concluded that the fee for 2011 would not be material.
Accounting Standards Not Yet Adopted
In October 2009, the FASB issued new accounting guidance for recognizing revenue for a multiple-deliverable revenue arrangement. The new guidance amends the existing guidance for separately accounting for individual deliverables in a revenue arrangement with multiple deliverables, and removes the criterion that an entity must use objective and reliable evidence of fair value to separately account for the deliverables. The new guidance also establishes a hierarchy for determining the value of each deliverable and establishes the relative selling price method for allocating consideration when vendor specific objective evidence or third-party evidence of value does not exist. We must adopt the new guidance prospectively for new revenue arrangements entered into or materially modified beginning in the first quarter of fiscal year 2012. Earlier adoption is permitted. We are currently evaluating the impact that the new guidance will have on our Consolidated Financial Statements and the timing of our adoption.
In January 2010, the FASB issued Accounting Standards Update 2010-06, “Improving Disclosures about Fair Value Measurements” (“Update 2010-06”). Update 2010-06 provides amendments to current standards to require new disclosures for transfers of assets and liabilities between Levels 1 and 2 and for activity in Level 3 fair value measurements. Furthermore, the update provides amendments to clarify that a reporting entity should provide fair value measurement disclosures for each class of assets and liabilities and should provide disclosures about the valuation techniques and inputs used to measure fair value for both recurring and nonrecurring fair value measurements for those measurements that fall in either Level 2 or Level 3. Update 2010-06 becomes effective for interim and annual reporting periods beginning after December 15, 2010. The Company is currently evaluating this new statement.
In April 2010, the FASB issued ASU No. 2010-17, “Milestone Method of Revenue Recognition”, which amends FASB ASC Topic 605. ASU No. 2010-17 provides guidance on defining a milestone and determining when it may be appropriate to apply the milestone method of revenue recognition for research or development transactions. Research or development arrangements frequently include payment provisions whereby a portion or all of the consideration is contingent upon milestone events such as successful completion of phases in a study or achieving a specific result from the research or development efforts. The amendments in this ASU provide guidance on the criteria that should be met for determining whether the milestone method of revenue recognition is appropriate. ASU 2010-17 is effective for fiscal years and interim periods within those years beginning on or after June 15, 2010, with early adoption permitted. We are currently evaluating the impact that the new guidance will have on our Consolidated Financial Statements and the timing of our adoption.
| 3. | Going Concern and Liquidity Considerations |
There is substantial doubt about the Company’s ability to continue as a going concern. The Company’s consolidated financial statements are prepared using GAAP applicable to a going concern, which contemplates the realization of assets and liquidation of liabilities in the normal course of business. The accompanying historical consolidated financial statements do not include any adjustments that might be necessary if the Company is unable to continue as a going concern.
144
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
The assessment of our ability to continue as a going concern was made by management considering, among other factors: (1) the timing and number of approved products that will be reintroduced to the market and the related costs; (2) the suspension of shipment of all products manufactured by us and the requirements under the consent decree with the FDA; (3) the possibility that we may need to obtain additional capital despite the proceeds from the offering of the 2015 Notes in March 2011 and the equity we were able to issue in February 2011 (see Note 1—“Description of Business—Significant Developments” ); (4) the potential outcome with respect to the governmental inquiries, litigation or other matters described in Note 15—“Commitments and Contingencies”; (5) our ability to comply with debt covenants; and (6) our ability to obtain future revenues from the sales of Makena™ sufficient to meet our future needs and expectations. Our assessment was further affected by our fiscal year 2011 net loss of $174.0, our 2010 net loss of $283.6, and the outstanding balance of cash and cash equivalents of $137.6 million as of March 31, 2011. For periods subsequent to March 31, 2011, we expect losses to continue because we are unable to generate any significant revenues from more of our own manufactured products until we are able to begin selling more of our approved products with FDA’s approval and with respect to products manufactured for us by third parties until after we are able to generate significant sales of Makena™ which was approved by the FDA in February 2011. We received notification from the FDA on September 8, 2010 of approval for our discontinued generics operations to ship into the marketplace the first product approved under the consent decree, i.e., the Potassium Chloride Extended Release Capsule. We resumed shipment of extended-release potassium chloride capsule, Micro-K® 10mEq and Micro-K® 8mEq, in September 2010, resumed shipments of the generic version of Potassium Chloride Extended Release Capsule in December 2010 and we began shipping Makena™ in March 2011. We have continued to ship Evamist®, which is manufactured for the Company by a third party, during the period covered by the consent decree. We are continuing to prepare other products for FDA inspection and do not expect to resume shipping other products until the second half of fiscal year 2012, at the earliest. In addition, we must meet ongoing operating costs as well as costs related to the steps we are currently taking to introduce Makena™, and to prepare for introducing and reintroducing other approved products to the market. If we are not able to obtain the FDA’s clearance to resume manufacturing and distribution of more of our approved products in a timely manner and at a reasonable cost, or if revenues from the sale of approved products introduced or reintroduced into the market place prove to be insufficient, our financial position, results of operations, cash flows and liquidity will continue to be materially adversely affected. These conditions raise substantial doubt about our ability to continue as a going concern.
Based on current financial projections, we believe the continuation of our Company as a going concern is primarily dependent on our ability to address, among other factors: (1) sales of Makena™, which was approved by the FDA in February 2011 notwithstanding recent actions by the FDA to permit continued sale of compounded alternatives and by CMS to permit compounded alternatives to be reimbursed under the Medicaid program; (2) the timing, number and revenue generation of approved products that will be introduced or reintroduced to the market and the related costs; (3) the suspension of shipment of all products manufactured by us and the requirements under the consent decree with the FDA (other than the Potassium Chloride Extended Release Capsule, including Micro-K® 10mEq and Micro-K® 8mEq, products that are the subject of the FDA notification letter discussed above); (4) the possibility that we will need to obtain additional capital; (5) the potential outcome with respect to the governmental inquiries, litigation or other matters described in Note 15—“Commitments and Contingencies” ; and (6) compliance with our debt covenants. While we address these matters, we must continue to meet expected near-term obligations, including normal course operating cash requirements and costs associated with introducing or reintroducing approved products to the market (such as costs related to our employees, facilities and FDA compliance), remaining payments associated with the acquisition and retention of the rights to Makena™ (see Note 5—“Acquisition” ), the financial obligations pursuant to the plea agreement, costs associated with our legal counsel and consultant fees, as well as the significant costs associated with the steps taken by us in connection with the consent decree and the litigation and governmental inquiries. If we are not able to obtain the FDA’s clearance to resume manufacturing and
145
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
distribution of certain or many of our approved products in a timely manner and at a reasonable cost and/or if we are unable to successfully commercialize Makena™, and/or if we experience adverse outcomes with respect to any of the governmental inquiries or litigation described in Note 15—“Commitments and Contingencies” , our financial position, results of operations, cash flows and liquidity will continue to be materially adversely affected.
In the near term, we are focused on performing the following: (1) addressing the FDA’s concerns regarding patient access to MakenaTM and CMS policy permitting Medicaid program reimbursement of compounded products; (2) the continued commercial launch of Makena™; (3) meeting the requirements of the consent decree, which will allow our approved products to be reintroduced to the market (other than the Potassium Chloride Extended Release Capsule products, including Micro-K® 10mEq and Micro-K® 8mEq, that are the subject of the FDA notification letter previously discussed); (4) the divestiture of Nesher and the Company’s generics business; and (5) pursuing various means to minimize operating costs and increase cash. Since December 31, 2010, the Company has generated non-recurring cash proceeds to support its on-going operating and compliance requirements from a $32.3 million private placement of Class A Common Stock in February 2011 and the 2015 Notes in March 2011 (which were used, in part, to repay all existing obligations under the agreement with U.S. Healthcare) (see Note 13—“Long-Term Debt” for a description of the 2015 Notes and U.S. Healthcare loan). While the cash proceeds received to date were sufficient to meet near-term cash requirements, we are pursuing ongoing efforts to increase cash, including, the continued implementation of cost savings, the divestiture of Nesher and the Company’s generics business and other assets and the return of certain additional approved products to market in a timely manner. We cannot provide assurance that we will be able to realize additional cost reductions from reducing our operations, that some or many of our approved products can be returned to the market in a timely manner (other than the Potassium Chloride Extended Release Capsule, including Micro-K® 10mEq and Micro-K® 8mEq products that are the subject of the FDA notification letter previously discussed above), that our higher profit approved products will return to the market in the near term or at all or that we can obtain additional cash through asset sales or the sale of equity or the successful commercial launch of Makena™. If we are unsuccessful in our efforts to introduce or return our products to market at adequate levels, or to sell assets or raise additional equity, we may be required to further reduce our operations, including further reductions of our employee base, or we may be required to cease certain or all of our operations in order to offset the lack of available funding.
We continue to evaluate the sale of certain of our assets, including Nesher and our generics business, which is included in discontinued operations, and assets held for sale. To date, we are in advanced discussions for the sale of our generics business, however, we cannot assure that we will complete any transaction on favorable terms or at all. The Company is continuing to work with its advisors and several interested parties to complete a transaction. However, due to general economic conditions, we will likely be exposed to risks related to the overall macro-economic environment, including a lower rate of return than we have historically experienced on our invested assets and being limited in our ability to sell assets. In addition, we cannot provide any assurance that we ultimately will be successful in finding suitable purchasers for the sale of such assets. Even if we are able to find purchasers, we may not be able to obtain attractive terms and conditions for such sales, including attractive pricing. In addition, divestitures of businesses involve a number of risks, including the diversion of management and employee attention, significant costs and expenses, the loss of customer relationships, a decrease in revenues and earnings associated with the divested business, and the disruption of operations in the affected business. In addition, divestitures potentially involve significant post-closing separation activities, which could involve the expenditure of significant financial and employee resources. Inability to consummate identified asset sales or manage the post-separation transition arrangements could adversely affect our business, financial condition, results of operations and cash flows.
146
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
| 4. | Restructuring and Impairment Charges |
| 2011 | 2010 | 2009 | ||||||||||
| Restructuring |
$ | 0.1 | $ | 3.2 | $ | 9.8 | ||||||
| Intangible Assets Impairment |
— | 82.3 | 36.6 | |||||||||
| Manufacturing, Distribution and Packaging Assets Impairment |
— | 3.5 | — | |||||||||
| Total Restructuring and Impairment Charges |
$ | 0.1 | $ | 89.0 | $ | 46.4 | ||||||
Restructuring
During fiscal year 2011, as part of an ongoing realignment of its cost structure, the Company made further reductions of its workforce and employee headcount. On March 31, 2010, headcount was reduced by approximately 80. During the quarter ended March 31, 2009, the Company reduced its headcount by approximately 564 employees through a combination of terminations and layoffs. The Company recorded restructuring expense of $0.1, $3.2 and $9.8 in fiscal years 2011, 2010 and 2009, respectively, for severance benefits related primarily to the headcount reductions described above.
The activity in accrued severance is summarized as follows:
| 2011 | 2010 | 2009 | ||||||||||
| Balance at beginning of year |
$ | 2.5 | $ | 7.0 | $ | — | ||||||
| Provision for severance benefits(a) |
1.8 | 3.2 | 8.8 | |||||||||
| Amounts charged to accrual |
(3.7 | ) | (7.7 | ) | (1.8 | ) | ||||||
| Balance at end of year |
$ | 0.6 | $ | 2.5 | $ | 7.0 | ||||||
| (a) | Includes amounts that were not classified as restructuring charges. |
Impairment
During the fourth quarter of fiscal year 2011, we made a decision to divest the generics business and as a result of this decision, we evaluated our long-lived assets for impairment.
Certain significant events occurred in the three months ended March 31, 2010 that indicated that the carrying value of certain assets as of March 31, 2010 may not be recoverable. These events included: (a) the expectation of when we would be able to resume manufacturing and shipment and begin generating cash flow from the sale of certain of our approved products, (b) the entry into the plea agreement with the U.S. Department of Justice (see Note 15—“Commitments and Contingencies”) and (c) the reduction of our workforce that occurred on March 31, 2010.
Based on the events described above, we determined that a triggering event occurred in the fourth quarter of fiscal years 2011 and 2010 giving rise to the need to assess the recoverability of our long-lived assets. Based on the assessment of the recoverability of our long-lived assets, it was determined that future undiscounted cash flows were not sufficient to support the carrying value of certain of our long-lived assets, and this resulted in material non-cash charges for impairment of property and equipment and intangible assets in the quarters ended March 31, 2011 and 2010. Cash flow projections require a significant level of judgment and estimation in order to determine a number of interdependent variables and assumptions such as probability, timing, pricing and various cost factors. Cash flow projections are highly sensitive to changes in these variables and assumptions.
147
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Based upon the Company’s analysis, it was determined the following assets, which are included in discontinued operations, were impaired as of March 31, 2011 and the Company recognized an impairment charge in loss from discontinued operations of $10.0:
| • | With respect to the Manufacturing, Distribution & Packaging asset group (“MD&P”), an asset group consisting primarily of property and equipment used in manufacturing operations and a generic product portfolio, the Company had approximately $39.8 of net book value as of March 31, 2011 prior to the impairment charge. As a result of management’s decision to divest the generics business and an evaluation of offers received for the generics business it was determined that the assets related to MD&P were impaired. The Company recorded an impairment charge of $10.0 during fiscal year 2011 as part of discontinued operations to reduce the carrying value of manufacturing equipment, product rights, and other assets to estimated fair value. (See Note 23—“Divestitures”). |
Based upon the Company’s analysis, it was determined the following assets were impaired as of March 31, 2010:
| • | With respect to the intangible assets related to Evamist®, the Company had approximately $116.0 of unamortized costs as of March 31, 2010 and remaining useful lives of 8 to 12 years related to product rights, trademark rights and rights under the sublicense agreement. To test impairment of these assets, the Company performed a cash flow analysis, which incorporates a number of significant assumptions and estimates. The sum of the projected undiscounted cash flows, which extend over the useful life assigned to the longest lived asset, did not exceed the carrying amount of the intangible assets as of March 31, 2010 and it was concluded that the assets related to Evamist®, were impaired as of that date. The Company recorded $79.0 during fiscal year 2010 as an impairment charge to reduce the carrying value of the intangible assets related to Evamist® to their aggregate estimated fair value. |
| • | With respect to the intangible assets related to Micro-K® and patents and trademarks, the Company had approximately $16.2 and $3.3 of unamortized costs as of March 31, 2010, respectively. The Company performed a cash flow analysis that extended over the average remaining useful life of the assets and determined that the assets related to patents and trademarks were impaired. The Company recorded an impairment charge of $3.3 during fiscal year 2010 to reduce the carrying value of patents and trademarks to estimated fair value. |
| • | With respect to the Manufacturing, Distribution & Packaging asset group (“MD&P”), an asset group consisting primarily of property and equipment used in manufacturing operations and a generic product portfolio, the Company had $112.4 of net book value as of March 31, 2010. We performed a cash flow analysis that extended over the average remaining useful life of the assets and determined that the assets related to MD&P were impaired. The Company recorded an impairment charge of $46.6 during fiscal year 2010 of which $43.1 was part of discontinued operations, to reduce the carrying value of manufacturing equipment, product rights, and other assets to estimated fair value. (See Note 23— “Divestitures”). |
During the fiscal year ended March 31, 2009, the Company recognized impairment charges of $36.6 for the following:
| • | Pursuant to the terms of the consent decree with the FDA, the Company agreed not to distribute its unapproved products, including its branded prenatal vitamins and hematinic products, until they are approved through the FDA’s Abbreviated New Drug Application (“ANDA”) or New Drug Application (“NDA”) processes. Since such products are unable to generate any revenues and are not approved by the FDA, the Company recorded $34.1 during the fiscal year ended March 31, 2010 as an impairment charge to write-off the remaining net book value of the intangible assets related to its branded prenatal vitamins and hematinic products. |
148
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
| • | In January 2009, the Company decided to no longer pursue approval of an acquired product to treat endometriosis. As a result, the Company recorded $2.5 during the fiscal year ended March 31, 2009 to write-off the remaining net book value of intangible assets related to this product. |
With respect to the intangible assets related to Evamist®, at March 31, 2009, the Company had approximately $125.5 of unamortized costs and remaining useful lives of 9 to 13 years. To test impairment of these assets in fiscal year ended March 31, 2009, the Company performed a cash flow analysis, which incorporated a number of significant assumptions and estimates that were subject to change as actual results became known. The sum of the projected undiscounted cash flows as of March 31, 2009, which extended over the useful life assigned to the assets, slightly exceeded the carrying amount of the intangible assets as of March 31, 2009 and the Company concluded that the asset was not impaired as of that date. However, events and circumstances, primarily during the fourth quarter of fiscal year 2010, required management to revise its earlier assumptions and estimates and such revisions resulted in the fiscal year 2010 impairments described above.
With respect to the asset groups at March 31, 2009, these asset groups were reviewed for recoverability by comparing the asset’s carrying amounts to undiscounted expected future cash flows. Carrying values were determined to be fully recoverable, and no impairment loss was recognized. The recoverability analysis reflected the terms of the Consent Decree entered into with the FDA and the then expectation of when the Company would return to market certain or many of its approved products.
| 5. | Acquisition |
On January 16, 2008, the Company entered into the Original Makena™ Agreement with Cytyc Prenatal Products Corp. and Hologic, Inc. (Cytyc Prenatal Products Corp. and Hologic, Inc. are referred to collectively as “Hologic”) to acquire the U.S. and worldwide royalty free, exclusive rights to Makena™ (hydroxyprogesterone caproate) and certain related assets upon approval of the pending New Drug Application for Makena™. On January 8, 2010, the Company and Hologic entered into Amendment No. 1 to the Original Makena™ Agreement, which, among other things, included a $70.0 cash payment for the exclusive rights to Makena™, which was recorded as purchased in-process research and development expense on the statement of operations for the fiscal year ended March 31, 2010. On February 4, 2011, the Company entered into Amendment No. 2 to the Original Makena™ Agreement. The amendments set forth in Amendment No. 2 reduced the payment to be made on the fifth business day following the day on which Hologic gave the Company notice that the FDA has approved Makena™ (the “Transfer Date”) to $12.5 and revised the schedule for making the remaining payments of $107.5. Under these revised payment provisions, after the $12.5 payment on the Transfer Date and a subsequent $12.5 payment 12 months after the date the FDA approved Makena™ (the “Approval Date”), the Company has the right to elect between the two alternate payment schedules for the remaining payments, with royalties of 5% of the net sales of Makena™ payable for certain periods and under different circumstances, depending on when the Company elects to make the remaining payments. The Company may make any of the payments on or before their due dates, and the date on which the Company makes the final payment contemplated by the selected payment schedule will be the final payment date, after which royalties, if any, will cease to accrue.
Under the revised payment provisions set forth in Amendment No. 2, after the $12.5 payment on the Transfer Date and a subsequent $12.5 payment twelve months after the Approval Date, the Company has the right to elect between the following two alternate payment schedules for the remaining payments:
Payment Schedule 1:
| • | A $45.0 payment 18 months after the Approval Date, plus a royalty equal to 5% of net sales of Makena™ made during the period from 12 months after the Approval Date to the date the $45.0 payment is made; |
| • | A $20.0 payment 21 months after the Approval Date; |
149
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
| • | A $20.0 payment 24 months after the Approval Date; and |
| • | A $10.0 payment 27 months after the Approval Date. |
The royalties will continue to be calculated subsequent to the $45.0 milestone payment but do not have to be paid as long as the Company makes subsequent milestone payments when due.
Payment Schedule 2:
| • | A $7.3 payment 18 months after the Approval Date, plus a royalty equal to 5% of net sales of Makena™ made during the period from 12 months after the Approval Date to 18 months after the Approval Date; |
| • | A $7.3 payment for each of the succeeding twelve months; |
| • | A royalty payable 24 months following the Approval Date equal to 5% of net sales of Makena™ made during the period from 18 months after the Approval Date to 24 months after the Approval Date; and |
| • | A royalty payable 30 months following the Approval Date equal to 5% of net sales of Makena™ made during the period from 24 months after the Approval Date to 30 months after the Approval Date. |
Notwithstanding anything to the contrary in Amendment No. 2, however, the Company may make any of the foregoing payments on or before their due dates, and the date on which the Company makes the final payment contemplated by the selected payment schedule will be the final payment date, after which no royalties will accrue.
Moreover, if the Company elects Payment Schedule 1 and thereafter elects to pay the $45.0 payment earlier than the 18-month deadline, the royalties beginning after 12 months will cease to accrue on the date of the early payment. Additionally, the subsequent payments will be paid in three month intervals following the $45.0 payment date.
Lastly, if the Company elects Payment Schedule 1 and thereafter does not make any of the milestone payments when due, Amendment No. 2 provides that no payment default will be deemed to occur, provided the Company timely pays the required royalties accruing in the quarter during which the milestone payment has become due but is not paid.
Under the Indenture governing the $225 aggregate principal amount of 12% Senior Secured Notes due 2015 (the “2015 Notes”), described further below the Company agreed to make the $45 payment under Payment Schedule 1 12 months after the Approval Date and agreed to certain other restrictions on its ability to amend the payment schedules.
| 6. | Investment Securities |
The carrying amount of available-for-sale securities and their approximate fair values at March 31, 2011 and 2010 were as follows:
| March 31, 2011 | ||||||||||||||||
| Cost | Gross Unrealized Gains |
Gross Unrealized Losses |
Fair Value |
|||||||||||||
| Auction rate securities |
$ | 54.3 | 2.9 | — | $ | 57.2 | ||||||||||
| March 31, 2010 | ||||||||||||||||
| Cost | Gross Unrealized Gains |
Gross Unrealized Losses |
Fair Value |
|||||||||||||
| Non-current auction rate securities |
$ | 62.9 | 3.0 | — | $ | 65.9 | ||||||||||
150
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
At March 31, 2008, the Company had short-term marketable securities that were recorded at fair value based on quoted market prices using the specific identification method and consisted of mutual funds comprised of U.S. government investments. During the fiscal year ended March 31, 2009, a realized loss of $0.1 was recognized upon sale of these securities.
At March 31, 2011 and 2010, the Company had $61.5 and $71.6, respectively, of original principal invested in ARS. These securities all have a maturity in excess of 10 years. The Company’s investments in ARS primarily represent interests in collateralized debt obligations supported by pools of student loans, the principal of which is guaranteed by the U.S. Government. ARS backed by student loans are viewed as having low default risk and therefore very low risk of credit downgrade. The ARS held by the Company are AAA-rated securities with long-term nominal maturities for which the interest rates are reset through a Dutch auction process that occurs at pre-determined intervals of up to 35 days. The auctions historically have provided a liquid market for these securities.
On February 25, 2009, the Company initiated legal action against Citigroup Global Markets Inc. (“CGMI”), through which it acquired the ARS the Company held at that time, in the District Court for the Eastern District of Missouri. On January 21, 2010, the Company and CGMI entered into a Purchase and Release Agreement pursuant to which CGMI agreed to purchase the Company’s remaining ARS for an aggregate purchase price of approximately $61.7. The Company also received a two-year option (which expires on January 21, 2012) to reacquire the ARS (in whole or on a class-by-class basis) for the prices at which they were sold, as well as the right to receive further payments in the event any ARS are redeemed prior to the expiration of the option.
With the liquidity issues experienced in global credit and capital markets, the ARS held by the Company at March 31, 2011 and 2010 experienced multiple failed auctions beginning in February 2008 as the amount of securities submitted for sale exceeded the amount of purchase orders. Given the failed auctions, the Company’s ARS are illiquid until a successful auction for them occurs. Accordingly, $65.9 of ARS at March 31, 2010 were classified as non-current assets and are included in the line item “Investment securities” in the accompanying Consolidated Balance Sheets. As a result of the agreement with CGMI expiring within one year of the balance sheet date at March 31, 2011, the Company classified $57.2 as a current asset and are included in the line item “Investment securities” in the accompanying Consolidated Balance Sheets at March 31, 2011.
In accordance with authoritative guidance ASC 860, Transfers and Servicing, the Company accounted for the ARS transfer to CGMI, including the two-year option to reacquire the ARS, as a secured borrowing with pledge of collateral. The transfer of the ARS to CGMI does not meet all of the conditions set forth in ASC 860 in order to be accounted for as a sale. As a secured borrowing with pledge of collateral, the Company was required to record a short-term liability (“collateralized borrowing”) as of March 31, 2011 for the ARS sale proceeds, representing a borrowing of cash from CGMI (see Note 13—“Long-Term Debt”). The ARS have been transferred to CGMI and serve as a pledge of collateral under this borrowing. The Company shall continue to carry the ARS as an asset in the accompanying Consolidated Balance Sheets, and it will continue to adjust to the ARS’ fair value on a quarterly basis (see Note 7—“Fair Value Measures”). In the event any ARS are redeemed prior to the expiration of the option, and upon such expiration, the Company will account for the redemptions as a sale pursuant to ASC 860.
The Company faces significant liquidity concerns as discussed in Note 3—“Going Concern and Liquidity Considerations.” As a result, the Company determined in fiscal year 2009 that it could no longer support its previous assertion that it had the ability to hold impaired securities until their forecasted recovery. Accordingly, the Company concluded that the ARS became other-than-temporarily impaired during December 2008 and recorded a $9.1 loss into earnings during the three months ended December 31, 2008. This adjustment reduced the carrying value of the ARS to $63.7 at December 31, 2008. The estimated fair value of the Company’s ARS
151
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
holdings at March 31, 2010 was $65.9. The Company recorded discount accretion of $0.2 on the carrying value of ARS and recorded the $2.9 difference between the fair value and carrying value of the ARS at March 31, 2011 in accumulated other comprehensive income as an unrealized gain of $1.7, net of tax.
The Company recorded discount accretion of $0.3 as carrying value of ARS and recorded the $3.0 difference between the fair value and carrying value of the ARS at March 31, 2010 in accumulated other comprehensive income as an unrealized gain of $1.6, net of tax.
Since the transfer of the ARS to CGMI on January 21, 2010, $10.1 of ARS were redeemed in the year ended March 31, 2011. The Company received from CGMI cash proceeds in the amount of $1.3, representing the difference between the principal amount of the securities redeemed and the price in which they were sold to CGMI. The Company also recorded a loss in the Consolidated Statement of Operations for the year ended March 31, 2011 in the amount of $0.6, representing the difference between the principal amount of the securities redeemed and their carrying value prior to redemption.
Since the transfer of the ARS to CGMI on January 21, 2010, $0.5 of ARS were redeemed in the quarter ended March 31, 2010. The Company received from CGMI cash proceeds in the amount of $0.1, representing the difference between the principal amount of the securities redeemed and the price in which they were sold to CGMI. The Company also recorded a gain in the Consolidated Statement of Operations for the year ended March 31, 2010 in the amount of $0.1, representing the difference between the principal amount of the securities redeemed and their carrying value prior to redemption.
The ARS are valued based on a discounted cash flow model that considers, among other factors, the time to work out the market disruption in the traditional trading mechanism, the stream of cash flows (coupons) earned until maturity, the prevailing risk free yield curve, credit spreads applicable to a portfolio of student loans with various tenures and ratings and an illiquidity premium. These factors were used in a Monte Carlo simulation based methodology to derive the estimated fair value of the ARS.
| 7. | Fair Value Measures |
In September 2006, the FASB issued authoritative guidance for fair value measurements. The Company implemented the authoritative guidance, effective April 1, 2008, which relates to disclosures for financial assets, financial liabilities, and any other assets and liabilities that are recognized or disclosed at fair value in the consolidated financial statements on a recurring basis. The guidance defines fair value as the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. In order to increase consistency and comparability in fair value measurements, the authoritative guidance established a fair value hierarchy that ranks the quality and reliability of the information used to measure fair value. Financial assets and liabilities carried at fair value are classified and disclosed in one of the following three categories:
| • | Level 1—Primarily consists of financial assets and liabilities whose values are based on unadjusted quoted prices for identical assets or liabilities in an active market that the Company has the ability to access. |
| • | Level 2—Includes financial instruments measured using significant other observable inputs that are valued by reference to similar assets or liabilities, such as: quoted prices for similar assets or liabilities in active markets; quoted prices for identical or similar assets or liabilities in markets that are not active; inputs other than quoted prices that are observable for the asset or liability; and inputs that are derived principally from or corroborated by observable market data by correlation or other means. |
152
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
| • | Level 3—Comprised of financial assets and liabilities whose values are based on prices or valuation techniques that require inputs that are both unobservable and significant to the overall fair value measurement. These inputs reflect management’s own assumptions about the assumptions a market participant would use in pricing the asset or liability. |
The following tables present the Company’s fair value hierarchy as of March 31, 2011 and 2010 for those financial assets measured at fair value on a recurring basis:
| Fair Value Measurements at March 31, 2011 | ||||||||||||||||
| Total | Level 1 | Level 2 | Level 3 | |||||||||||||
| Current ARS |
$ | 57.2 | $ | — | $ | — | $ | 57.2 | ||||||||
| Warrant Liability |
(104.8 | ) | — | — | (104.8 | ) | ||||||||||
| Fair Value Measurements at March 31, 2010 | ||||||||||||||||
| Total | Level 1 | Level 2 | Level 3 | |||||||||||||
| Non-current ARS |
$ | 65.9 | $ | — | $ | — | $ | 65.9 | ||||||||
The Company’s investments in ARS primarily represent interests in collateralized debt obligations supported by pools of student loans, the principal of which is guaranteed by the U.S. Government. The ARS held by the Company are AAA-rated securities with long-term nominal maturities for which the interest rates are reset through a Dutch auction process that occurs at pre-determined intervals of up to 35 days. Due to the lack of observable market quotes and an illiquid market for the Company’s ARS portfolio that existed as of March 31, 2011 and 2010, the Company utilized a valuation model that relied exclusively on Level 3 inputs, including those that are based on expected cash flow streams and collateral values (see Note 6—“Investment Securities”).
The Company’s warrant liability represents warrants issued to U.S. Healthcare to purchase an aggregate of up to 20.1 million shares of Class A Common Stock at an exercise price of $1.62 per share. Certain March 17, 2011 amendments to the arrangement with U.S. Healthcare resulted in the warrants no longer qualifying as equity instruments (See Note 24—“Warrant Liability”).
The contingent interest feature of the $200.0 principal amount of Contingent Convertible Subordinated Notes (see Note13—“Long-Term Debt”) meets the criteria of and qualifies as an embedded derivative. Although this feature represents an embedded derivative financial instrument, based on its de minimis value at the time of issuance and at March 31, 2011, no value has been assigned to this embedded derivative.
The following tables present the changes in fair value for financial assets measured at fair value on a recurring basis using significant unobservable inputs (Level 3):
ARS (Level 3)
| 2011 | 2010 | |||||||
| Balance at beginning of year |
$ | 65.9 | $ | 66.0 | ||||
| Realized losses resulting from ARS being deemed other-than-temporarily impaired |
— | — | ||||||
| Unrealized (losses) gains included in other comprehensive income |
(0.1 | ) | 0.7 | |||||
| Accretion of investment impairment |
0.2 | 0.3 | ||||||
| Sales of ARS |
(8.8 | ) | (1.1 | ) | ||||
| Balance at end of year |
$ | 57.2 | $ | 65.9 | ||||
153
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Warrant Liability (Level 3)
| 2011 | ||||
| Balance at beginning of year |
$ | — | ||
| Initial valuation |
(175.5 | ) | ||
| Unrealized gain included in other income |
70.7 | |||
| Balance at end of year |
$ | (104.8 | ) | |
| 8. | Inventories |
Inventories, net of reserves, as of March 31 consisted of:
| 2011 | 2010 | |||||||
| Raw materials |
$ | — | $ | 0.8 | ||||
| Finished goods |
1.0 | 0.4 | ||||||
| $ | 1.0 | $ | 1.2 | |||||
Reserves for obsolete inventory are deducted from the related inventory balances. The activity in the Company’s reserve for obsolete inventory is summarized as follows:
| 2011 | 2010 | 2009 | ||||||||||
| Balance at beginning of year |
$ | 3.3 | $ | 13.5 | $ | — | ||||||
| Provisions for obsolete inventory |
— | 0.4 | 17.2 | |||||||||
| Amounts charged against reserves |
(0.8 | ) | (10.6 | ) | (3.7 | ) | ||||||
| Balance at end of year. |
$ | 2.5 | $ | 3.3 | $ | 13.5 | ||||||
The Company ceased all manufacturing activities during the fourth quarter of fiscal year 2009 and its revenues are limited to sales of products manufactured by third parties, except for, starting in 2010, Potassium Chloride Extended Release Capsule by its discontinued generics business. Additionally, most costs associated with the Company’s manufacturing operations are recognized directly into cost of sales rather than capitalized into inventory except for the activity related to Potassium Chloride Extended Release Capsule.
| 9. | Property and Equipment |
Property and equipment as of March 31 consisted of:
| 2011 | 2010 | |||||||
| Buildings and building improvements |
$ | 70.7 | $ | 58.4 | ||||
| Machinery and equipment |
8.8 | 13.3 | ||||||
| Office furniture and equipment |
35.6 | 31.1 | ||||||
| Leasehold improvements |
— | 20.9 | ||||||
| Land and improvements |
4.8 | 4.8 | ||||||
| Construction-in-process |
— | 0.9 | ||||||
| Property and equipment held for sale |
5.3 | 3.3 | ||||||
| 125.2 | 132.7 | |||||||
| Less accumulated depreciation |
(57.6 | ) | (54.3 | ) | ||||
| Net property and equipment |
$ | 67.6 | $ | 78.4 | ||||
154
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Capital additions to property and equipment were $0.3, $10.2 and $23.6 (includes PDI and Nesher capital additions) for the fiscal years ended March 31, 2011, 2010 and 2009, respectively. Depreciation of property and equipment was $6.2, $8.5 and $9.5 for the fiscal years ended March 31, 2011, 2010 and 2009, respectively.
The Company recorded a reserve in the amount of $1.9 during fiscal year ended March 31, 2009 primarily relating to the impairment of property and equipment held for sale. The related loss provision is recorded as selling and administrative expense in the consolidated statement of operations.
| 10. | Intangible Assets |
Intangible assets as of March 31 consisted of:
| 2011 | 2010 | |||||||||||||||||||||||
| Gross Carrying Amount (a) |
Accumulated Amortization |
Net Carrying Amount |
Gross Carrying Amount (a) |
Accumulated Amortization |
Net Carrying Amount |
|||||||||||||||||||
| Product rights acquired: |
||||||||||||||||||||||||
| Makena™ |
$ | 119.6 | $ | (2.8 | ) | $ | 116.8 | $ | — | $ | — | $ | — | |||||||||||
| Evamist™ |
21.2 | (9.0 | ) | 12.2 | 21.2 | (7.9 | ) | 13.3 | ||||||||||||||||
| Trademarks acquired: |
||||||||||||||||||||||||
| Evamist™ |
5.1 | (2.5 | ) | 2.6 | 5.1 | (2.3 | ) | 2.8 | ||||||||||||||||
| License agreements: |
||||||||||||||||||||||||
| Evamist™ |
35.6 | (16.5 | ) | 19.1 | 35.6 | (14.7 | ) | 20.9 | ||||||||||||||||
| Covenants not to compete: |
||||||||||||||||||||||||
| Evamist™ |
0.6 | (0.6 | ) | — | 0.6 | (0.6 | ) | — | ||||||||||||||||
| Trademarks and patents |
2.1 | (2.1 | ) | — | 1.3 | (1.3 | ) | — | ||||||||||||||||
| Other |
0.4 | (0.2 | ) | 0.2 | 0.7 | (0.2 | ) | 0.5 | ||||||||||||||||
| Total intangible assets |
$ | 184.6 | $ | (33.7 | ) | $ | 150.9 | $ | 64.5 | $ | (27.0 | ) | $ | 37.5 | ||||||||||
| (a) | Gross Carrying Amount is shown net of impairment charges. |
As of March 31, 2011, the Company’s product rights acquired, trademark rights acquired, license agreements, covenants not to compete, trademarks and patents, and other intangible assets have original weighted average useful lives of approximately 8 years, 15 years, 15 years, 9 years, 13 years, and 5 years, respectively. Amortization of intangible assets was $6.1, $10.3 and $12.4 for the fiscal years ended March 31, 2011, 2010 and 2009, respectively.
The Company evaluates its intangible assets for impairment whenever events or changes in circumstances indicate an intangible asset’s carrying amount may not be recoverable. As more fully described in Note 4—“Restructuring and Impairment Charges”, as a result of certain triggering events and related impairment assessments that occurred in the quarter ended March 31, 2010, the Company recorded intangible asset impairment charges of $82.3 in fiscal year 2010 related to our continuing operations. There were no impairment charges recorded in fiscal year 2011 for intangible assets of continuing operations.
Assuming no other additions, disposals or adjustments are made to the carrying values and/or useful lives of the intangible assets, annual amortization expense on product rights, trademarks acquired and other intangible assets is estimated to be approximately $20.4 in each of the five succeeding fiscal years.
The $119.6 Makena™ product rights represents the $12.5 payment made on the Transfer Date plus the present value of the remaining payments due to Hologic as described in Note 5—“Acquisition” and is being
155
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
amortized over its estimated patent protected life of seven years. As there are no performance conditions related to the remaining installment payments, the Company recorded the remaining obligations in current liabilities ($57.3) and non-current liabilities ($50.0) based upon their expected due dates.
| 11. | Other Assets |
Other assets as of March 31 consisted of:
| 2011 | 2010 | |||||||
| Life insurance, cash surrender value |
$ | 4.5 | $ | 4.2 | ||||
| Preferred stock investments and accrued dividends |
0.4 | 7.7 | ||||||
| Deferred financing costs, net |
9.7 | 0.2 | ||||||
| Deposits and other |
0.5 | 0.3 | ||||||
| Other assets |
$ | 15.1 | $ | 12.4 | ||||
See Note 2—“Summary of Significant Accounting Policies” and Note 15—“Commitments and Contingencies” for description of preferred stock investments and accrued dividends.
| 12. | Accrued Liabilities |
Accrued liabilities as of March 31 consisted of:
| 2011 | 2010 | |||||||
| Salaries, wages, incentives and benefits |
$ | 10.3 | $ | 6.8 | ||||
| Accrued severance |
0.6 | 6.2 | ||||||
| Accrued interest payable |
3.3 | 2.2 | ||||||
| Professional fees |
4.0 | 4.8 | ||||||
| Failure to supply claims |
10.0 | 12.3 | ||||||
| Product recalls |
3.1 | 4.6 | ||||||
| Price protection. |
25.8 | — | ||||||
| Accrued litigation and governmental inquiries |
11.6 | 12.5 | ||||||
| Makena obligation, current portion |
57.3 | — | ||||||
| Medicaid rebates |
6.9 | 3.6 | ||||||
| Sales returns |
4.8 | 7.3 | ||||||
| Sales rebates |
1.1 | 1.0 | ||||||
| Other |
3.1 | 2.7 | ||||||
| $ | 141.9 | $ | 64.0 | |||||
The liability for product recalls includes the impact of products expected to be returned, the estimated unpaid cost of processing fees and other administrative expenses associated with the recalls. The estimated liability for product returns related to recalls was based on estimated and actual customer inventory levels and actual contract pricing.
Accrued litigation and governmental inquiries consists of settlement obligations as well as loss contingencies recognized by the Company because settlement was determined to be probable and the related payouts were reasonably estimable. For the fiscal year ended March 31, 2011, the Company recorded litigation expense in the amount of $7.4, $6.3 of which was recorded for the HHS OIG matter (see Note 15—“Commitments and Contingencies”). For the fiscal year ended March 31, 2010, the Company recorded litigation expense in the amount
156
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
of $15.0 for certain matters. For the fiscal year ended March 31, 2009, the Company recorded litigation and governmental inquiries expense in the amount of $14.4 for product liability actions related to the voluntary product recalls initiated by the Company in calendar year 2008 and early 2009 and alleged damages as a result of the ingestion of purportedly oversized tablets allegedly distributed by the Company (see Note 15—“Commitments and Contingencies”). Of the remaining product liability at March 31, 2011, $7.3 was recorded in accrued liabilities and $2.9 was recorded in other long-term liabilities.
Pursuant to the plea agreement with the U.S. Department of Justice, the Company agreed to pay criminal fines, restitution to the Medicare and Medicaid programs and an administrative forfeiture in the aggregate amount of $27.6 (see Note 15—“Commitments and Contingencies”). In the fiscal year ended March 31, 2009, the Company recorded litigation and governmental inquiries expense of $23.6 for the present value of the administrative forfeiture and five expected installment payments related to the criminal fines and reduced net revenues by $2.3 for the restitution payments owed to the Medicare and Medicaid programs. In the fiscal years ended March 31, 2011, 2010 and 2009, the Company recorded $0.3, $0.4 and $0.3, respectively, of interest accretion expense representing the difference between the present value and the undiscounted amount of the fines and penalties, which is recognized ratably over the period during which payments are due and payable pursuant to the plea agreement. At March 31, 2011, $2.0 of the aggregate plea agreement amount was recorded in accrued liabilities and $17.4 was recorded in other long-term liabilities. In addition, long term liabilities include $1.5 related to long-term tax liabilities, $6.1 for life insurance for the former CEO, and $37.3 for other legal matters. At March 31, 2010, $2.8 of the aggregate plea agreement amount was recorded in accrued liabilities and $19.1 was recorded in other long-term liabilities. In addition, at March 31, 2010, long-term liabilities include $7.8 related to long-term tax liabilities, $5.8 for former CEO life insurance, and $13.3 for other legal matters.
The Makena™ obligation, current portion represents the current portion of the remaining milestone payments owed to Hologic related to the Makena™ product rights as described further in Note 5—“Acquisition”.
On April 1, 2011, the Company reduced the list price of Makena™. As a result, the Company recorded a price protection reserve of $25.8 based upon the change in list price.
| 13. | Long-Term Debt |
Long-term debt as of March 31 consisted of:
| 2011 | 2010 | |||||||
| Convertible notes |
$ | 200.0 | $ | 200.0 | ||||
| Senior notes (less unamortized discount of $6.7 million ) |
218.3 | — | ||||||
| Building mortgages |
33.0 | 35.3 | ||||||
| Collateralized borrowing |
52.4 | 61.2 | ||||||
| Software financing arrangement |
— | 0.6 | ||||||
| 503.7 | 297.1 | |||||||
| Less current portion |
(85.4 | ) | (63.9 | ) | ||||
| $ | 418.3 | $ | 233.2 | |||||
Convertible notes
In May 2003, the Company issued $200.0 principal amount of 2.5% Contingent Convertible Subordinated Notes (the “Notes”) that are convertible, under certain circumstances, into shares of Class A Common Stock at an initial conversion price of $23.01 per share. The Notes, which mature on May 16, 2033, bear interest that is payable on May 16 and November 16 of each year at a rate of 2.50% per annum. The Company also is obligated
157
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
to pay contingent interest at a rate equal to 0.5% per annum during any six-month period from May 16 to November 15 and from November 16 to May 15, with the initial six-month period commencing May 16, 2006, if the average trading price of the Notes per $1.0 principal amount for the five trading day period ending on the third trading day immediately preceding the first day of the applicable six-month period equals $1.2 or more. In November 2007, the average trading price of the Notes reached the threshold for the five-day trading period that resulted in the payment of contingent interest and for the period from November 16, 2007 to May 15, 2008 the Notes paid interest at a rate of 3.00% per annum. In May 2008, the average trading price of the Notes fell below the contingent interest threshold for the five-day trading period and beginning May 16, 2008 the Notes began to pay interest at a rate of 2.50% per annum, which is the current rate as of March 31, 2011.
The Company may redeem some or all of the Notes at any time, at a redemption price, payable in cash, of 100% of the principal amount of the Notes, plus accrued and unpaid interest, including contingent interest, if any. Holders may require the Company to repurchase all or a portion of their Notes on May 16, 2013, 2018, 2023 and 2028 or upon a change in control, as defined in the indenture governing the Notes, at a purchase price, payable in cash, of 100% of the principal amount of the Notes, plus accrued and unpaid interest, including contingent interest, if any. Since the next occasion holders may require the Company to repurchase all or a portion of their Notes is May 16, 2013, the Notes were classified as a long-term liability as of March 31, 2011 and 2010. The Notes are subordinate to all of the Company’s existing and future senior obligations.
The Notes are convertible, at the holders’ option, into shares of the Company’s Class A Common Stock prior to the maturity date under the following circumstances:
| • | during any future quarter, if the closing sale price of the Company’s Class A Common Stock over a specified number of trading days during the previous quarter is more than 120% of the conversion price of the Notes on the last trading day of the previous quarter. The Notes are initially convertible at a conversion price of $23.01 per share, which is equal to a conversion rate of approximately 43.4594 shares per $1.0 principal amount of Notes; |
| • | if the Company has called the Notes for redemption; |
| • | during the five trading day period immediately following any nine consecutive trading day period in which the trading price of the Notes per $1.0 principal amount for each day of such period was less than 95% of the product of the closing sale price of our Class A Common Stock on that day multiplied by the number of shares of our Class A Common Stock issuable upon conversion of $1.0 principal amount of the Notes; or |
| • | upon the occurrence of specified corporate transactions. |
The Company has reserved 8.7 million shares of Class A Common Stock for issuance in the event the Notes are converted.
The Notes, which are unsecured, do not contain any restrictions on the payment of dividends, the incurrence of additional indebtedness or the repurchase of the Company’s securities, and do not contain any financial covenants. However, a failure by the Company or any of its subsidiaries to pay any indebtedness or any final non-appealable judgments in excess of $0.8 constitutes an event of default under the indenture. An event of default would permit the trustee under the indenture or the holders of at least 25% of the Notes to declare all amounts owing to be immediately due and payable and exercise other remedies.
Senior notes
On March 17, 2011, the Company completed a private placement with a group of institutional investors of $225.0 aggregate principal amount of 12% Senior Secured Notes due 2015.
158
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
The 2015 Notes bear interest at an annual rate of 12% per year, payable semiannually in arrears on March 15 and September 15 of each year, commencing September 15, 2011. The 2015 Notes will mature March 15, 2015. At any time prior to March 15, 2013, the Company may redeem up to 35% of the aggregate principal amount of the 2015 Notes at a redemption price of 112% of the principal amount of the 2015 Notes, plus accrued and unpaid interest to the redemption date, with the net cash proceeds of one or more equity offerings. At any time prior to March 15, 2013, the Company may redeem all or part of the 2015 Notes at a redemption price equal to (1) the sum of the present value, discounted to the redemption date, of (i) a cash payment to be made on March 15, 2013 of 109% of the principal amount of the 2015 Notes, and (ii) each interest payment that is scheduled to be made on or after the redemption date and on or before March 15, 2013, plus (2) accrued and unpaid interest to the redemption date. At any time after March 15, 2013 and before March 15, 2014, the Company may redeem all or any portion of the 2015 Notes at a redemption price of 109% of the principal amount of the 2015 Notes, plus accrued and unpaid interest to the redemption date. At any time after March 15, 2014, the Company may redeem all or any portion of the 2015 Notes at a redemption price of 100% of the principal amount of the 2015 Notes, plus accrued and unpaid interest to the redemption date. The 2015 Notes are secured by certain assets of the Company and certain assets of its subsidiaries.
After an original issue discount of 3%, the Company received proceeds of $218.3 which were used to fund a first year interest reserve totaling $27.0, repay all existing obligations to the U.S. Healthcare totaling approximately $61.1 and pay fees and expenses associated with the Notes Offering of $9.7. In connection with these payments, the Company also terminated all future loan commitments with the U.S. Healthcare. The remaining proceeds, totaling approximately $120.0 will be used for general corporate purposes, including the launch of Makena™.
The 2015 Notes were offered only to accredited investors pursuant to Regulation D under the Securities Act of 1933, as amended.
Building mortgages
In March 2006, the Company entered into a $43.0 mortgage loan arrangement (the “Mortgage Loan”) with one of its primary lenders, in part, to refinance $9.9 of existing mortgages. The $32.8 of net proceeds the Company received from the mortgage loan was used for working capital and general corporate purposes. The Mortgage Loan, which is secured by four of the Company’s buildings, bears interest at a rate of 5.91% and matures on April 1, 2021. The Company is current in all its financial payment obligations under the Mortgage Loan arrangement. On August 5, 2010, the Company received a letter (the “Waiver Letter”) approving certain waivers of covenants under the Mortgage Loan, and certain other loan documents entered into in connection with the execution of the Mortgage Loan (collectively, the “Loan Documents”). Accordingly, the Company recorded the mortgage as a long-term liability at March 31, 2010 since the Waiver Letter was received prior to filing the Form 10-K for the fiscal year ended March 31, 2010. In the Waiver Letter, the lenders consented to the following under the Loan Documents:
| • | Waiver of the requirement that we deliver audited balance sheets, statements of income and expenses and cash flows; |
| • | Waiver of the requirement that we certify financials delivered under the Loan Documents; |
| • | Waiver of the requirement that we deliver to the lenders Form 10-Ks within 75 days of the close of the fiscal year, Form 10-Qs within 45 days of the close of each of the first three fiscal quarters of the fiscal year, and copies of all IRS tax returns and filings; and |
| • | Waiver, until March 31, 2012, of the requirement that we maintain a net worth, as calculated in accordance with the terms of the Loan Documents, of at least $250.0on a consolidated basis. |
159
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
With respect to the waiver of the requirement to deliver Form 10-Ks and Form 10-Qs, we agreed to bring our SEC filings current effective with the submission of our Form 10-Q for the quarter ended December 31, 2010, which the Company did on March 31, 2011 and become timely on a go- forward basis effective with the filing of this Form 10-K for the fiscal year ending March 31, 2011. This waiver applied to our then existing late filings at the time the Waiver Letter was received.
In addition to the waivers our subsidiaries ETHEX and PDI were removed as guarantors under the Loan Documents and to Nesher Pharmaceuticals Inc. was added as a new guarantor under the Loan Documents. Effective with the filing of this Form 10-K, the Company is in compliance with the SEC filing requirements contained in the waiver. However, based on current financial projections, the Company does not anticipate meeting the March 31, 2012 minimum net worth requirement. Accordingly, the mortgage is classified as a current liability as of March 31, 2011. If the Company is ultimately unable to meet the minimum net worth requirement by March 31, 2012, it will need to seek an additional waiver from the mortgage lender.
Collateralized borrowing
On February 25, 2009, the Company initiated legal action against Citigroup Global Markets Inc. (“CGMI”), through which it acquired the ARS the Company held at that time, in the District Court for the Eastern District of Missouri. On January 21, 2010, the Company and CGMI entered into a Purchase and Release Agreement pursuant to which CGMI agreed to purchase the Company’s remaining ARS for an aggregate purchase price of approximately $61.7. The Company also received a two-year option (which expires on January 21, 2012) to reacquire the ARS (in whole or on a class-by-class basis) for the prices at which they were sold, as well as the right to receive further payments in the event any ARS are redeemed prior to the expiration of the option.
In accordance with authoritative guidance ASC 860, Transfers and Servicing, the Company accounted for the ARS transfer to CGMI, including the two-year option to reacquire the ARS, as a secured borrowing with pledge of collateral. The transfer of the ARS to CGMI does not meet all of the conditions set forth in ASC 860 in order to be accounted for as a sale. As a secured borrowing with pledge of collateral, the Company was required to record a short-term liability (“collateralized borrowing”) as of March 31, 2011 and 2010 for the ARS sale proceeds, representing a borrowing of cash from CGMI. The ARS have been transferred to CGMI and serve as a pledge of collateral under this borrowing. The Company shall continue to carry the ARS as an asset in the accompanying Consolidated Balance Sheets, and it will continue to adjust to the ARS’ fair value on a quarterly basis (see Note 7—“Fair Value Measures”). In the event any ARS are redeemed prior to the expiration of the option, the Company will account for the redemptions as a sale pursuant to ASC 860. Through March 31, 2011, $10.1 par value of ARS ($8.8 at CGMI purchase cost) were redeemed.
Software financing arrangement
The Company renegotiated the contract with the seller during fiscal year 2011 by extending the agreement for three years and now pays only for products and services as they are incurred instead of upfront which was previously financed with a financial institution at March 31, 2010.
160
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Other
The aggregate scheduled maturities of long-term debt, (as adjusted by the August 2010 Waiver Letter) as of March 31, 2011 were as follows:
| Due in one year. |
$ | 85.4 | ||
| Due in two years |
— | |||
| Due in three years. |
200.0 | |||
| Due in four years |
225.0 | |||
| Due in five years |
— | |||
| Thereafter |
— | |||
| $ | 510.4 | |||
| 14. | Taxable Industrial Revenue Bonds |
In December 2005, the Company entered into a financing arrangement with St. Louis County, Missouri related to expansion of its operations in St. Louis County. Up to $135.5 of industrial revenue bonds could have been issued to the Company by St. Louis County relative to capital improvements made through December 31, 2009. This agreement provides that 50% of the real and personal property taxes on up to $135.5 of capital improvements will be abated for a period of ten years subsequent to the property being placed in service. Industrial revenue bonds totaling $129.9 were outstanding at March 31, 2011. The industrial revenue bonds are issued by St. Louis County to the Company upon its payment of qualifying costs of capital improvements, which are then leased by the Company through December 1, 2019, unless earlier terminated. The Company has the option at any time to discontinue the arrangement and regain full title to the abated property. The industrial revenue bonds bear interest at 4.25% per annum and are payable as to principal and interest concurrently with payments due under the terms of the lease. The Company has classified the leased assets as property and equipment and has established a capital lease obligation equal to the outstanding principal balance of the industrial revenue bonds. Lease payments may be made by tendering an equivalent portion of the industrial revenue bonds. As the capital lease payments to St. Louis County may be satisfied by tendering industrial revenue bonds (which is the Company’s intention), the capital lease obligation, industrial revenue bonds and related interest expense and interest income, respectively, have been offset for presentation purposes in the consolidated financial statements.
| 15. | Commitments and Contingencies |
Leases
The Company leases manufacturing, office and warehouse facilities, equipment and automobiles under operating leases expiring through fiscal year 2023. Total rent expense for the fiscal years ended March 31, 2011, 2010 and 2009 were $2.2, $2.9 and $5.0, respectively.
Facility leases with free rent periods or rent escalation clauses are expensed on a straight-line basis over the life of the lease commencing at lease inception.
161
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Future minimum lease commitments under non-cancelable operating leases are as follows:
| 2012 |
$ | 1.3 | ||
| 2013 |
0.9 | |||
| 2014 |
0.4 | |||
| 2015 |
0.3 | |||
| 2016 |
0.3 | |||
| Thereafter. |
0.2 | |||
| $ | 3.4 | |||
Contingencies
The Company is currently subject to legal proceedings and claims that have arisen in the ordinary course of business. While the Company is not presently able to determine the potential liability, if any, related to all such matters, the Company believes the matters it currently faces, individually or in the aggregate, could have a material adverse effect on its financial condition or operations or liquidity.
The Company has licensed the exclusive rights to co-develop and market various generic equivalent products with other drug delivery companies. These collaboration agreements require the Company to make up-front and ongoing payments as development milestones are attained. On January 8, 2010, the Company and Hologic entered into an amendment to the original Makena™ asset purchase agreement. On February 4, 2011, the Company entered into an Amendment No. 2 to the Original Agreement. See Note 5—“Acquisition” for more information about the amended agreement.
On December 5, 2008, the Board terminated the employment agreement of Marc S. Hermelin, the Chief Executive Officer of the Company at that time, “for cause” (as that term is defined in such employment agreement). Additionally, the Board removed Mr. M. Hermelin as Chairman of the Board and as the Chief Executive Officer, effective December 5, 2008. In accordance with the termination provisions of his employment agreement, the Company determined that Mr. M. Hermelin would not be entitled to any severance benefits. In addition, as a result of Mr. M. Hermelin’s termination “for cause,” the Company determined it was no longer obligated for the retirement benefits specified in the employment agreement. However, Mr. M. Hermelin informed the Company that he believed he effectively retired from his employment with the Company prior to the termination of his employment agreement on December 5, 2008 by the Board. If it is determined that Mr. M. Hermelin did effectively retire prior to December 5, 2008, the actuarially determined present value (as calculated in December 2008) of the retirement benefits due to him would total $36.9. On November 10, 2010, Mr. M. Hermelin voluntarily resigned as a member of the Board. On March 22, 2011, Mr. M. Hermelin made a demand on the Company for indemnification with respect to his payment of $1.9 imposed by the United States District Court as a fine and forfeiture of pecuniary gain as part of the sentence resulting from his guilty plea entered by the Court on March 10, 2011. Mr. M. Hermelin pled guilty to two federal misdemeanor counts as a responsible corporate officer of the Company at the time when a misbranding of two morphine sulfate tablets occurred which contained more of the active ingredient than stated on the label. In addition, the Company has advanced or will advance, under the terms of the Indemnification Agreement, legal expense amounting to approximately $5.0 to a variety of law firms who represented Mr. M. Hermelin for legal matters including the FDA and SEC investigations, the Department of Justice inquiry, the Audit Committee investigation, HHS OIG exclusion and various class action lawsuits. Under the Company’s standard Indemnification Agreement entered into with all directors, including Mr. M. Hermelin when he served as Chairman of the Board and Chief Executive Officer of the Company, as a condition for the advancement of expenses, each director is required to sign an undertaking to reimburse the Company for the advanced expenses in the event it is found that the director is not entitled to indemnification.
162
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Mr. M. Hermelin’s demand for reimbursement of the $1.9 fine and forfeiture, and whether the advance of legal fees to represent him for various legal matters should be indemnified, is under review by a special committee appointed by the Board of Directors of the Company.
Litigation and Governmental Inquiries
Resolution of one or more of the matters described below could have a material adverse effect on the Company’s results of operations, financial condition or liquidity. The Company intends to vigorously defend its interests in the matters described below while cooperating in governmental inquiries.
Accrued litigation consists of settlement obligations as well as loss contingencies recognized by the Company because settlement was determined to be probable and the related payouts were reasonably estimable. While the outcome of the current claims cannot be predicted with certainty, the possible outcome of claims is reviewed at least quarterly and an adjustment to the Company’s accrual is recorded as deemed appropriate based upon these reviews. Based upon current information available, the resolution of legal matters individually or in aggregate could have a material adverse effect on the Company’s results of operations, financial condition or liquidity. The Company is unable to estimate the possible loss or range of losses at March 31, 2011.
Due to the FDA consent decree, an approval or a tentative approval was not obtained in the required time frame for any of the Company’s Paragraph IV ANDA filings. Therefore, the 180 days Hatch-Waxman exclusivity was lost.
The Company and ETHEX were named as defendants in a case brought by CIMA LABS, Inc. and Schwarz Pharma, Inc. and styled CIMA LABS, Inc. et al. v. KV Pharmaceutical Company et al., filed in U.S. District Court for the District of Minnesota. CIMA alleged that the Company and ETHEX infringed on a CIMA patent in connection with the manufacture and sale of Hyoscyamine Sulfate Orally Dissolvable Tablets, 0.125 mg. The Court entered a stay pending the outcome of the U.S. Patent and Trademark Office’s (“USPTO”) reexamination of a patent at issue in the suit. On August 17, 2009, the Court entered an order “administratively” terminating this action in Minnesota, but any party has the right to seek leave to reinstitute the case. On September 30, 2009, on appeal of the Examiner’s rejection of the claims, the Board of Patent Appeals and Interferences affirmed the Examiner’s rejections. After the Board’s denial of CIMA’s appeal, CIMA requested a rehearing with the Board. On March 28, 2011, the USPTO ruled the appealed claims are unpatentable. CIMA can reopen prosecution or request a hearing within two months.
The Company and/or ETHEX have been named as defendants in certain multi-defendant cases alleging that the defendants reported improper or fraudulent pharmaceutical pricing information, i.e., Average Wholesale Price, or AWP, and/or Wholesale Acquisition Cost, or WAC, information, which allegedly caused the governmental plaintiffs to incur excessive costs for pharmaceutical products under the Medicaid program. Cases of this type have been filed against the Company and/or ETHEX and other pharmaceutical manufacturer defendants by the States of Massachusetts, Alabama, Mississippi, Louisiana, Utah and Iowa, by New York City, and by approximately 45 counties in New York State. The State of Mississippi effectively voluntarily dismissed the Company and ETHEX without prejudice on October 5, 2006 by virtue of the State’s filing an Amended Complaint on such date that does not name either the Company or ETHEX as a defendant. On August 13, 2007, ETHEX settled the Massachusetts lawsuit and received a general release of liability with no admission of liability. On October 7, 2008, ETHEX settled the Alabama lawsuit for $2.0 and received a general release of liability with no admission of liability. On November 25, 2009, ETHEX settled the New York City and New York county cases (other than the Erie, Oswego and Schenectady County cases) for $3.0 and received a general release of liability. On February 23, 2010, ETHEX settled the Iowa lawsuit for $0.5 and received a general release of liability. On August 25, 2010, ETHEX settled the Erie, Oswego and Schenectady Counties lawsuit for $0.1 and received a general release of liability. On October 21, 2010, the Company received a subpoena from the
163
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Florida Office of Attorney General requesting information related to ETHEX’s pricing and marketing activities. The Company is currently complying with the State’s request for documents and pricing information. In November 2010, the Company and ETHEX were served with a complaint with respect to an AWP case filed by the State of Louisiana. In January 2011, the Company filed Defendants’ Exceptions of Nonconformity and Vagueness of the Petition, Improper Cumulation and Joinder, No Right of Action, Prescription and Preemption and No Cause of Action with respect to the Louisiana lawsuit.
The Company received a subpoena from the HHS OIG in 2007, seeking documents with respect to two of ETHEX’s nitroglycerin products. Both are unapproved products, that is, they have not received FDA approval. (In certain circumstances, FDA approval may not be required for drugs to be sold in the marketplace.) The subpoena stated that it is in connection with an investigation into potential false claims under Title 42 of the U.S. Code, and appears to pertain to whether these products were eligible for reimbursement under federal health care programs. On or about July 2, 2008, the Company received a supplementary subpoena in this matter, seeking additional documents and information. In a letter dated August 4, 2008, that subpoena was withdrawn and a separate supplementary subpoena was substituted. In October 2009, HHS OIG identified five additional products as being subject to its investigation: Hydro-tussin (carbinoxamine); Guaifenex (extended release); Hyoscyamine sulfate (extended-release); Hycoclear (hydrocodone); and Histinex (hydrocodone). The Company has provided additional documents requested in the subpoena, as supplemented. Discussions with the U.S. Department of Justice and the United States Attorney’s Office for the District of Massachusetts indicate that this matter is a False Claims Act qui tam action that is currently still under seal and that the government is reviewing similar claims relating to other drugs manufactured by ETHEX, as well as drugs manufactured by other companies. The Company has not been provided a copy of the qui tam complaint. On or about March 26, 2009, the Company consented to an extension of the time during which the government may elect to intervene in the qui tam lawsuit. The Company has been in discussions with the HHS OIG and Department of Justice regarding possible settlement of these claims.
On December 12, 2008, by letter, the Company was notified by the staff of the SEC that it had commenced an informal inquiry to determine whether there have been violations of certain provisions of the federal securities laws. On November 23, 2010, by email, the Company was notified by the staff of the SEC that it had commenced an informal inquiry pertaining to potential insider trading and requested information pertaining to an employee. The Company is cooperating with the government and, among other things, has provided copies of requested documents and information. On February 22, 2011, the staff of the SEC sent the Company a letter advising it that it had closed this inquiry pertaining to potential insider trading as to the Company and did not intend to recommend any enforcement action pertaining to the Company.
The Company, at the direction of a special committee of the Board of Directors that was in place prior to June 10, 2010, responded to requests for information from the Office of the United States Attorney for the Eastern District of Missouri and FDA representatives working with that office. In connection therewith, on February 25, 2010, the Board, at the recommendation of the special committee, approved entering into a plea agreement subject to court approval with the Office of the United States Attorney for the Eastern District of Missouri and the Office of Consumer Litigation of the United States Department of Justice (referred to herein collectively as the “Department of Justice”).
The plea agreement was executed by the parties and was entered by the U.S. District Court, Eastern District of Missouri, Eastern Division on March 2, 2010. Pursuant to the terms of the plea agreement, ETHEX pleaded guilty to two felony counts, each stemming from the failure to make and submit a field alert report to the FDA in September 2008 regarding the discovery of certain undistributed tablets that failed to meet product specifications. Sentencing pursuant to the plea agreement also took place on March 2, 2010.
164
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Pursuant to the plea agreement, ETHEX agreed to pay a criminal fine in the amount of $23.4 in four installments. The first installment, in the amount of $2.3, was due within 10 days of sentencing. The second and third installments, each in the amount of $5.9, were originally due on December 15, 2010 and July 11, 2011, respectively. The fourth and final installment, in the amount of $9.4, was originally due on July 11, 2012. ETHEX also agreed to pay, within 10 days of sentencing, restitution to the Medicare and the Medicaid programs in the amounts of $1.8 and $0.6, respectively. In addition to the fine and restitution, ETHEX agreed not to contest an administrative forfeiture in the amount of $1.8, which was payable 45 days after sentencing and satisfies any and all forfeiture obligations ETHEX may have as a result of the guilty plea. In total, ETHEX agreed to pay fines, restitution and forfeiture in the aggregate amount of $27.6.
On November 15, 2010, upon the motion of the Department of Justice, the court vacated the previous fine installment schedule and imposed a new fine installment schedule using the standard federal judgment rate of 0.22% per annum, payable as follows:
| Payment Amounts including interest |
Payment Due Date | |
| $ 1.0 |
December 15, 2010 | |
| 1.0 |
June 15, 2011 | |
| 1.0 |
December 15, 2011 | |
| 2.0 |
June 15, 2012 | |
| 4.0 |
December 15, 2012 | |
| 5.0 |
June 15, 2013 | |
| 7.1 |
December 15, 2013 |
The Company made its first installment payment due on December 15, 2010.
In exchange for the voluntary guilty plea, the Department of Justice agreed that no further federal prosecution will be brought in the Eastern District of Missouri against ETHEX, the Company or the Company’s wholly-owned subsidiary, Ther-Rx, regarding allegations of the misbranding and adulteration of any oversized tablets of drugs manufactured by the Company, and the failure to file required reports regarding these drugs and patients’ use of these drugs with the FDA, during the period commencing on January 1, 2008 through December 31, 2008.
In connection with the guilty plea by ETHEX, ETHEX was expected to be excluded from participation in federal healthcare programs, including Medicare and Medicaid. In addition, as a result of the guilty plea by ETHEX, HHS OIG had discretionary authority to also exclude the Company from participation in federal healthcare programs. However, the Company is in receipt of correspondence from HHS OIG stating that, absent any transfer of assets or operations that would trigger successor liability, HHS OIG has no present intent to exercise its discretionary authority to exclude the Company as a result of the guilty plea by ETHEX.
In connection with the previously anticipated exclusion of ETHEX from participation in federal healthcare programs, the Company ceased operations of ETHEX on March 2, 2010. However, the Company has retained the ability to manufacture, market and distribute (once the requirements under the consent decree have been met) all generic products and is in possession of all intellectual property related to generic products, including all NDAs and ANDAs.
On November 15, 2010, the Company entered into the Divestiture Agreement with HHS OIG under which the Company agreed to sell the assets and operations of ETHEX to unrelated third parties prior to April 28, 2011 and to file articles of dissolution with respect to ETHEX under Missouri law by such date. Following such filing,
165
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
ETHEX may not engage in any new business other than for winding up its operations and will engage in a process provided under Missouri law to identify and resolve its liabilities over at least a two year period. Under the terms of the agreement, HHS OIG agreed not to exclude ETHEX from federal healthcare programs until April 28, 2011 and, upon completion of the sale of the ETHEX assets and of the filing of the articles of dissolution of ETHEX, the agreement will terminate. Civil monetary penalties and exclusion of ETHEX could have occurred if the Company had failed to meet its April 28, 2011 deadline. The Company has also received a letter from HHS OIG advising it further that assuming that it has complied with all agreements deemed necessary by HHS OIG, ETHEX has filed its articles of dissolution, and ETHEX no longer has any ongoing assets or operations other than those required to conclude the winding up process under Missouri law, HHS OIG would not exclude ETHEX thereafter. The Company has notified all parties of its intent to dissolve ETHEX and notifications were sent out on January 28, 2011. ETHEX has sold its assets in accordance with the Divestiture Agreement on May 20, 2011, we received a letter from HHS OIG stating that based upon its review of the information provided in our monthly reports, it appeared that the Company and ETHEX had completed our obligation under the Divestiture Agreement.
The Company currently does not anticipate that the voluntary guilty plea by ETHEX will have a material adverse effect on the Company’s efforts to comply with the requirements pursuant to the consent decree and to resume production and shipments of its approved products.
On November 10, 2010, Marc S. Hermelin voluntarily resigned as a member of the Board. The Company had been advised that HHS OIG notified Mr. Hermelin that he would be excluded from participating in federal healthcare programs effective November 18, 2010. In an effort to avoid adverse consequences to the Company, including a potential discretionary exclusion of the Company, and to enable it to secure its expanded financial agreement with U.S. Healthcare, the Company, HHS OIG, Mr. Hermelin and his wife (solely with respect to her obligations thereunder, including as joint owner with Mr. Hermelin of certain shares of Company stock) entered into the Settlement Agreement under which Mr. Hermelin also resigned as trustee of all family trusts that hold KV stock, agreed to divest his personal ownership interests in the Company’s Class A Common and Class B Common Stock (approximately 1.8 million shares, including shares held jointly with his wife) over an agreed upon period of time in accordance with a divestiture plan and schedule approved by HHS OIG, and agreed to refrain from voting stock under his personal control. In order to implement such agreement, Mr. Hermelin and his wife granted to an independent third party immediate irrevocable proxies and powers of attorney to divest their personal stock interests in the Company if Mr. Hermelin does not timely do so. The Settlement Agreement also required Mr. Hermelin to agree, for the duration of his exclusion, not to seek to influence or be involved with, in any manner, the governance, management, or operations of the Company. On March 14, 2011, Mr. Hermelin pleaded guilty to two federal misdemeanor counts pertaining to misbranding of two oversized morphine sulfate tablets, as a responsible corporate officer of the Company at the time that such tablets were introduced into interstate commerce. On March 22, 2011, Mr. M. Hermelin made a demand on the Company for indemnification with respect to his payment of $1.9 imposed by the United States District Court as a fine and forfeiture of pecuniary gain as part of the sentence resulting from his guilty plea entered by the Court on March 10, 2011. Mr. M. Hermelin pled guilty to two federal misdemeanor counts as a responsible corporate officer of the Company at the time when a misbranding of two morphine sulfate tablets occurred which contained more of the active ingredient than stated on the label. In addition, the Company has advanced or will advance, under the terms of the indemnification Agreement, legal expense amounting to approximately $5.0 to a variety of law firms who represented Mr. M. Hermelin for legal matters including the FDA and SEC investigations, the Department of Justice inquiry, the Audit Committee investigation, HHS OIG exclusion and various class action lawsuits. Under the Company’s standard Indemnification Agreement entered into with all directors, including Mr. M. Hermelin when he served as Chairman of the Board and Chief Executive Officer of the Company, as a condition for the advancement of expenses, each director is required to sign an undertaking to reimburse the Company for the advanced expenses in the event it is found that the director is not entitled to indemnification. Mr. M.
166
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Hermelin’s demand for reimbursement of the $1.9 fine and forfeiture, and whether the advanced legal fees to represent him for various legal matters should be indemnified, is under review by a special committee appointed by the Board of Directors of the Company.
As long as the parties comply with the Settlement Agreement, HHS OIG has agreed not to exercise its discretionary authority to exclude the Company from participation in federal health care programs, thereby allowing the Company and its subsidiaries (with the single exception of ETHEX, which has been dissolved pursuant to the Divestiture Agreement with HHS OIG) to continue to conduct business through all federal and state healthcare programs.
As a result of Mr. Hermelin’s resignation and the two agreements with HHS OIG, the Company believes that it has resolved its remaining issues with respect to HHS OIG and is positioned to continue to participate in Federal health care programs now and in the future.
The Company has received a subpoena from the State of California Department of Justice seeking documents with respect to ETHEX’s NitroQuick product. In an email dated August 12, 2009, the California Department of Justice advised that after reading CMS Release 151, it might resolve the subpoena that was issued. The Company provided limited information requested by the California Department of Justice on October 7, 2009, and on November 10, 2009 the California Department of Justice informed the Company that the California Department of Justice is contemplating what additional information, if any, it will request.
On February 27, 2009, by letter, the Company was notified by the U.S. Department of Labor that it was conducting an investigation of the Company’s Fifth Restated Profit Sharing Plan and Trust, to determine whether such plan is conforming with the provisions of Title I of the Employee Retirement Income Security Act (“ERISA”) or any regulations or orders there under. The Company cooperated with the Department of Labor in its investigation and on August 27, 2009, the Department of Labor notified the Company it had completed a limited review and no further review was contemplated at that time. On July 7, 2010, by letter, the U.S. Department of Labor notified the Company it was again conducting a review of the Company’s Fifth Restated Profit Sharing Plan and Trust. The Company provided the requested documents and has heard nothing further.
On August 12, 2010, by letter, the Company was notified by the U.S. Department of Labor that it was conducting a wage and hour investigation to assess compliance with the Fair Labor Standards Act. The Company is cooperating with the Department of Labor and has made available the requested documents. The Department of Labor had initially advised that it believed that the Company was liable for $160,000. In February 2011, a tentative settlement has been reached with the Department of Labor for approximately $30,000 with a release covering the two year statute of limitations period pertaining to FLSA claims. All payments allegedly due to affected former employees have been made by the Company.
On February 3, 2009, plaintiff Harold Crocker filed a putative class-action complaint against the Company in the United States District Court for the Eastern District of Missouri, Crocker v. KV Pharmaceutical Co., et al., No. 4-09-cv-198-CEJ. The Crocker case was followed shortly thereafter by two similar cases, also in the Eastern District of Missouri (Bodnar v. KV Pharmaceutical Co., et al., No. 4:09-cv-00222-HEA, on February 9, 2009, and Knoll v. KV Pharmaceutical Co., et al., No. 4:09-cv-00297-JCH, on February 24, 2009). The two later cases were consolidated into Crocker so that only a single action existed thereafter, and the plaintiffs filed a Consolidated Amended Complaint on June 26, 2009 (“Complaint”).
The Complaint purported to state claims against the Company and certain current and former employees for alleged breach of fiduciary duties to participants in the Company’s 401(k) plan. Defendants, including the Company and certain of its directors and officers, moved to dismiss the amended complaint on August 25, 2009, and briefing of those motions was completed on October 19, 2009. The court granted the motion to dismiss the
167
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Company and all individual defendants on March 24, 2010. A motion to alter or amend the judgment and second amended consolidated complaint was filed on April 21, 2010. The Company, on May 17, 2010, filed a Memorandum in Opposition to plaintiff’s motion to alter or amend the judgment and for leave to amend the consolidated complaint. On October 20, 2010, the Court denied plaintiffs’ motion to alter or amend the judgment and for leave to amend the complaint. Plaintiffs requested mediation and the Company agreed to this request. On February 15, 2011, during such mediation, this litigation was settled by an agreement in principle of the parties for an amount equal to $3.0, payable in full from the Company’s insurance coverage.
On December 2, 2008, plaintiff Joseph Mas filed a complaint against the Company, in the United States District Court for the Eastern District of Missouri, Mas v. KV Pharma. Co., et al., Case No. 08-CV-1859. On January 9, 2009, plaintiff Herman Unvericht filed a complaint against the Company also in the Eastern District of Missouri, Unvericht v. KV Pharma. Co., et al., Case No. 09-CV-0061. On January 21, 2009, plaintiff Norfolk County Retirement System filed a complaint against the Company, again in the Eastern District of Missouri, Norfolk County Retirement System v. KV Pharma. Co., et al., Case No. 09-CV-00138. The operative complaints in these three cases purport to state claims arising under Sections 10(b) and 20(a) of the Securities Exchange Act of 1934 on behalf of a putative class of stock purchasers. On April 15, 2009, the Honorable Carol E. Jackson consolidated the Unvericht and Norfolk County cases into the Mas case already before her. The amended complaint for the consolidated action, styled Public Pension Fund Group v. KV Pharma. Co., et al., Case No. 4:08-CV-1859 (CEJ), was filed on May 22, 2009. Defendants, including the Company and certain of its directors and officers, moved to dismiss the amended complaint on July 27, 2009, and briefing was completed on the motions to dismiss on September 3, 2009. The court granted the motion to dismiss the Company and all individual defendants in February 2010. On March 18, 2010, the plaintiffs filed a motion for relief from the order of dismissal and to amend their complaint, and also filed a notice of appeal. The Company filed its opposition to plaintiffs’ motion for relief from judgment and to amend the complaint on April 8, 2010. Briefing was completed on April 29, 2010. On October 20, 2010, the Court denied plaintiffs’ motion for relief from the order of dismissal and to amend pleadings. On November 1, 2010, plaintiffs’ filed a notice of appeal.
On October 2, 2009, the U.S. Equal Employment Opportunity Commission sent the Company a Notice of Charge of Discrimination regarding a charge, dated September 23, 2009, of employment discrimination based on religion (in connection with the termination of his employment with the Company) filed against the Company by David S. Hermelin, a current director and former Vice President, Corporate Strategy and Operations Analysis of the Company. On January 29, 2010, the Company filed its response to the Notice of Charge of Discrimination, which stated the Company’s position that Mr. D. Hermelin’s termination had nothing to do with religious discrimination and that his claim should be dismissed.
The Company and/or ETHEX are named defendants in at least 39 pending product liability or other lawsuits that relate to the voluntary product recalls initiated by the Company in late 2008 and early 2009. The plaintiffs in these lawsuits allege damages as a result of the ingestion of purportedly oversized tablets allegedly distributed in 2007 and 2008. The lawsuits are pending in federal and state courts in various jurisdictions. The 39 pending lawsuits include 9 that have settled but have not yet been dismissed. In the 39 pending lawsuits, two plaintiffs allege economic harm, 27 plaintiffs allege that a death occurred, and the plaintiffs in the remaining lawsuits allege non-fatal physical injuries. Plaintiffs’ allegations of liability are based on various theories of recovery, including, but not limited to strict liability, negligence, various breaches of warranty, misbranding, fraud and other common law and/or statutory claims. Plaintiffs seek substantial compensatory and punitive damages. Two of the lawsuits are putative class actions seeking economic damages with respect to recalled products, one of the lawsuits is on behalf of 29 claimants, and the remaining lawsuits are individual lawsuits or have two plaintiffs. One of these putative class actions, styled LeFaivre v. KV Pharmaceutical Company et al., seeks economic damages with respect to recalled metoprolol succinate product. During January 2011, the decision of the U.S. District Court dismissing the case in favor of the Company was reversed on appeal. The Company
168
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
requested reconsideration by the appellate court, which was denied in March 2011, and the Company has filed a motion for appellate review en banc, which was denied by the court on May 12, 2011. The case has been returned to the district court for further proceedings. The Company believes that these lawsuits are without merit and is vigorously defending against them, except where, in its judgment, settlement is appropriate. In addition to the 39 pending lawsuits, there are at least 31 pending pre-litigation claims (at least 6 of which involve a death) that may or may not eventually become lawsuits. The Company has also resolved a significant number of related product liability lawsuits and pre-litigation claims. In addition to self insurance, the Company possesses third party product liability insurance, which the Company believes is applicable to the pending lawsuits and claims.
The Company and ETHEX are named as defendants in a complaint filed by CVS Pharmacy, Inc. (“CVS”) in the United States District Court for the District of Rhode Island on or about February 26, 2010 and styled CVS Pharmacy, Inc. v. K-V Pharmaceutical Company and Ethex Corporation (No. CA-10-095) (“CVS Complaint”). The CVS Complaint alleges three claims: breach of contract, breach of implied covenant of good faith and fair dealing, and, in the alternative, promissory estoppels. CVS’ claims are premised on the allegation that the Company and/or ETHEX failed to perform their alleged promises to either supply CVS with its requirements for certain generic drugs or reimburse CVS for any higher price it must pay to obtain the generic drugs. CVS seeks damages of no less than $100.0, plus interest and costs. The Company was served with the CVS Complaint on March 8, 2010. An Answer was filed on April 14, 2010. On June 2, 2010, the Company filed a Motion to dismiss this action based on failure to join an indispensible party and lack of standing. On July 21, 2010, CVS filed objections to the Company’s Motion to Stay Discovery and Motion to Dismiss. On July 28, 2010, the Judge denied the Company’s Motion to Stay Discovery pending the Motion to Dismiss without issuing a decision. On January 28, 2011, the federal magistrate recommended that the Company’s Motion to Dismiss the Complaint be granted. The plaintiff is filing a notice of objection to the magistrate’s recommendation. In March 2011, CVS and its parent CVS Caremark Corporation filed a similar complaint, seeking damages similar to those sought in the federal case and adding another breach of contract claim, in state court in Superior Court of Providence County, Rhode Island, against the Company, ETHEX and Nesher. The parties have entered into a stipulation to stay the time for the Company to answer the state court complaint until June 10, 2011.
Robertson v. Ther-Rx Corporation, U.S. District Court for the Middle District of Alabama, Civil Case No. 2:09-cv-01010-MHT-TFM, filed October 30, 2009, by a Ther-Rx sales representative asserting non-exempt status and the right to overtime pay under the Fair Labor Standards Act for a class of Ther-Rx sales representatives and under the Family and Medical Leave Act of 1993 (with respect to plaintiff’s pregnancy) and Title VII of the Civil Rights Act of 1964 (also with respect to termination allegedly due to her pregnancy and to her complaints about being terminated allegedly as a result of her pregnancy). An additional seven Ther-Rx sales representatives have joined as plaintiffs. Class certification arguments are pending before the court. On December 22, 2010, a settlement in principle was reached between the parties for $0.3 and on May 12, 2011, the court approved the settlement agreements.
The Company entered into a License and Supply Agreement (“Agreement”) with Strides Arcolab and Strides, Inc. (collectively “Strides”) as well as a Share Purchase Agreement with Strides Arcolab on May 5, 2005. Strides purported to terminate the Agreement on March 11, 2009 due to the Company’s alleged failure to provide adequate assurances on its ability to perform under the Agreement to which the Company denied that the Agreement was terminated. On October 20, 2009, the Company filed a Statement of Claim and Requests for Arbitration with the International Chamber of Commerce alleging that Strides had anticipatorily repudiated the Agreement. On January 26, 2010, Strides filed its Answer and Counterclaims generally denying the allegations and on March 11, 2010, the Company filed its Answer generally denying Strides’ counterclaims. On December 13, 2010, the parties settled the arbitration by an agreed termination of the agreements between the parties, Strides’ retaining all rights to the product development work done under the agreements, the Company’s returning Strides’ stock certificates, and Strides’ paying the Company $7.3.
169
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
On October 13, 2009, the Company filed a Complaint in the United States District Court for the Eastern District of Missouri, Eastern Division, against J. Uriach & CIA S.A. (“Uriach”) seeking damages for breach of contract and misappropriation of the Company’s trade secrets and that Uriach be enjoined from further use of the Company’s confidential information and trade secrets. On September 28, 2010, the Court issued a Memorandum and Order granting defendant’s Motion to Dismiss for lack of personal jurisdiction of defendant, J. Uriach & CIA, S.A. The Company has appealed the decision.
On August 24, 2010, Westmark Healthcare Distributors, Inc. filed an action in the Third Judicial District Court In and For Salt Lake County, State of Utah, against Ther-Rx demanding payment of $0.1 for recalled, returned pharmaceutical products.
On March 17, 2011, the Company was served with a complaint by the trustee in bankruptcy for Qualia Clinical Services, Inc. asserting a breach of contract claim for approximately $0.3 for certain clinical work done by such Company.
On April 13, 2011, the Company received notice of a Civil Investigation Demand from the State of Texas Attorney General’s Office, Antitrust Division, regarding the possibility of a reduction in competition in the market for pharmaceutical treatments for premature labor, including 17P and Makena™. The Company is cooperating with this civil investigation demand’s request for information and documents.
From time to time, the Company is involved in various other legal proceedings in the ordinary course of its business. While it is not feasible to predict the ultimate outcome of such other proceedings, the Company believes the ultimate outcome of such other proceedings will not have a material adverse effect on its results of operations, financial condition or liquidity.
There are uncertainties and risks associated with all litigation and there can be no assurance the Company will prevail in any particular litigation. During the year ended March 31, 2011, 2010 and 2009, the Company recorded expense of $7.4, $15.0 and $50.0, respectively, for litigation and governmental inquiries. At March 31, 2011 and 2010, the Company had accrued $48.9 and $46.5, respectively, for estimated costs for litigation and governmental inquiries.
| 16. | Employment Agreements |
The Company has employment agreements with certain officers and key employees which extend for one to five years. These agreements provide for base levels of compensation and, in certain instances, also provide for incentive bonuses and separation benefits. Also, the agreement with the former Chief Executive Officer, Marc Hermelin, (“former CEO”) contained provisions for partial salary continuation under certain conditions, contingent upon non-compete restrictions and providing consulting services to the Company as specified in the agreement. In addition, the former CEO’s employment agreement provided for retirement compensation to be paid in the form of a single annuity equal to 30% of the former CEO’s final average compensation payable each year beginning at retirement and continuing for the longer of ten years or the life of the former CEO. In accordance with this agreement, the Company recognized retirement expense up to the time the former CEO was terminated (discussed below) of $1.8 for the fiscal year ended March 31, 2009.
On December 5, 2008, the Board terminated the employment agreement of Marc S. Hermelin, the Chief Executive Officer of the Company at that time, “for cause” (as that term is defined in such employment agreement). Additionally, the Board removed Mr. M. Hermelin as Chairman of the Board and as the Chief Executive Officer, effective December 5, 2008. In accordance with the termination provisions of his employment agreement, the Company determined that Mr. M. Hermelin would not be entitled to any severance benefits. In
170
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
addition, as a result of Mr. M. Hermelin’s termination “for cause,” the Company determined it was no longer obligated for the retirement benefits specified in the employment agreement and, accordingly, reversed the previously recorded accrual which reduced selling and administrative expense by $10.3 for the fiscal year ended March 31, 2009 (see Note 15—“Commitments and Contingencies”). The former CEO has informed the Company, however, that he believes he effectively retired from his employment with the Company prior to the termination of his employment agreement on December 5, 2008 by the Board.
| 17. | Income Taxes |
The income tax provisions (benefit) for the fiscal years ended March 31, 2011, 2010 and 2009, are based on estimated federal and state taxable income (loss) using the applicable statutory rates. The current and deferred federal and state income tax provision / (benefit) for continuing operations for the fiscal years ended March 31, 2011, 2010 and 2009 were as follows:
| 2011 | 2010 | 2009 | ||||||||||
| Provision/(Benefit) |
||||||||||||
| Current: |
||||||||||||
| Federal |
$ | (57.4 | ) | $ | (46.5 | ) | $ | (101.5 | ) | |||
| State |
(4.6 | ) | (4.0 | ) | (6.6 | ) | ||||||
| (62.0 | ) | (50.5 | ) | (108.1 | ) | |||||||
| Deferred: |
||||||||||||
| Federal |
(0.5 | ) | (57.3 | ) | (12.0 | ) | ||||||
| State |
— | (4.4 | ) | (0.9 | ) | |||||||
| (0.5 | ) | (61.7 | ) | (12.9 | ) | |||||||
| (62.5 | ) | (112.2 | ) | (121.0 | ) | |||||||
| Increase in valuation allowance |
70.3 | 88.3 | 82.4 | |||||||||
| Tax provision/(benefit) |
$ | 7.8 | $ | (23.9 | ) | $ | (38.6 | ) | ||||
The reasons for the differences between the provision / (benefit) for income taxes for continuing operations and the expected federal income taxes at the U.S. statutory rate are as follows:
| 2011 | 2010 | 2009 | ||||||||||
| Expected income tax expense (benefit) |
$ | (52.1 | ) | $ | (108.3 | ) | $ | (119.2 | ) | |||
| State income taxes, net of federal income tax benefit |
(2.5 | ) | (5.5 | ) | (5.1 | ) | ||||||
| Business credits |
— | (0.1 | ) | (3.8 | ) | |||||||
| Domestic manufacturer deduction adjustment for loss carry back |
— | 0.5 | 3.7 | |||||||||
| Adjustment to unrecognized tax benefits reserve |
(5.9 | ) | 1.4 | (6.7 | ) | |||||||
| Non deductible expenses related to plea agreement |
— | — | 8.4 | |||||||||
| Non deductible share based payment expense |
1.0 | 1.2 | 0.7 | |||||||||
| Non taxable gain on warrants |
(25.9 | ) | — | — | ||||||||
| Non deductible debt extinguishment charges |
16.8 | — | — | |||||||||
| Adjustment to operating loss deferred tax asset |
5.6 | — | — | |||||||||
| Other |
0.5 | (1.4 | ) | 1.0 | ||||||||
| (62.5 | ) | (112.2 | ) | (121.0 | ) | |||||||
| Change in valuation allowance |
70.3 | 88.3 | 82.4 | |||||||||
| Provision for income tax expense (benefit) |
$ | 7.8 | $ | (23.9 | ) | $ | (38.6 | ) | ||||
171
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
As of March 31, 2011 and 2010, the tax effect of temporary differences between the tax basis of assets and liabilities and their financial reporting amounts were as follows:
| 2011 | 2010 | |||||||||||||||
| Current | Non- Current |
Current | Non- Current |
|||||||||||||
| Gross Deferred Tax Assets: |
||||||||||||||||
| Revenue related accrued liabilities |
$ | 18.2 | $ | — | $ | 8.8 | $ | — | ||||||||
| Accrued compensation |
3.1 | — | 1.2 | — | ||||||||||||
| Intangible assets |
— | 58.6 | — | 61.0 | ||||||||||||
| Stock-based compensation |
0.5 | — | 1.3 | — | ||||||||||||
| Payroll tax liability |
0.2 | — | 0.4 | — | ||||||||||||
| Litigation and governmental inquiries liability |
3.5 | 7.3 | 3.6 | 5.5 | ||||||||||||
| Interest expense |
2.7 | — | — | — | ||||||||||||
| Auction rate securities |
— | 2.6 | — | 3.2 | ||||||||||||
| Impairment on property and equipment |
— | — | — | 0.6 | ||||||||||||
| Operating losses |
— | 136.3 | — | 80.8 | ||||||||||||
| Credit carry forwards |
— | 9.7 | — | 9.6 | ||||||||||||
| Other |
1.8 | 0.1 | 1.8 | 2.7 | ||||||||||||
| $ | 30.0 | $ | 214.6 | $ | 17.1 | $ | 163.4 | |||||||||
| Gross Deferred Tax Liabilities: |
||||||||||||||||
| Property and equipment |
$ | — | $ | (0.1 | ) | $ | — | $ | (2.9 | ) | ||||||
| Intangible assets |
$ | — | $ | (2.0 | ) | $ | — | $ | (1.6 | ) | ||||||
| Convertible notes interest |
— | (57.9 | ) | — | (48.0 | ) | ||||||||||
| Inventories |
— | — | — | — | ||||||||||||
| Prepaid insurance |
(1.1 | ) | — | (0.5 | ) | — | ||||||||||
| Other |
— | (0.5 | ) | — | — | |||||||||||
| (1.1 | ) | (60.5 | ) | (0.5 | ) | (52.5 | ) | |||||||||
| Valuation allowance |
(29.4 | ) | (211.5 | ) | (15.6 | ) | (155.0 | ) | ||||||||
| Net deferred tax asset (liability) |
$ | (0.5 | ) | $ | (57.4 | ) | $ | 1.0 | $ | (44.1 | ) | |||||
In assessing the realizability of deferred tax assets, management considers whether it is more likely than not that some portion or all of the deferred tax assets will not be realized. The ultimate realization of deferred tax assets is dependent upon the generation of future taxable income during the periods in which those temporary differences become deductible. Management considers all significant available positive and negative evidence, including the existence of losses in recent years, the timing of deferred tax liability reversals, projected future taxable income, taxable income in carry back years, and tax planning strategies to assess the need for a valuation allowance. Based upon the level of current taxable loss, projections for future taxable income over the periods in which the temporary differences are deductible, the taxable income in available carry back years and tax planning strategies, management believes it is not more likely than not the Company will realize the benefits of these deductible differences. The operating loss for the year ended March 31, 2009 and 2010 resulted in operating loss carry forwards. The operating loss for the year ended March 31, 2011 will also be carried forward. The Company established an additional valuation allowance of $70.3 for the current year. This was charged to income tax expense for year ended March 31, 2011.
The Company has Federal loss carry forwards of approximately $372.0 and state loss carry forwards of approximately $495.0 at March 31, 2011. The Company also has tax credit carry forwards for alternative
172
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
minimum tax, research credit, and foreign tax credit of $9.7 as of March 31, 2011. The loss carry forwards begin to expire in the year 2030. The alternative minimum tax credits have no expiration date. The research credit and foreign tax credit begin to expire in the year 2026 and 2017, respectively.
At March 31, 2011, the Company had $1.2 of gross unrecognized tax benefit reserves, all of which represents the amount of unrecognized tax benefits that, if recognized, would favorably affect the effective income tax rate in future periods.
A reconciliation of the unrecognized tax benefits at the beginning and end of the year for fiscal years 2011, 2010 and 2009 is as follows:
| Balance of unrecognized tax benefits at March 31, 2008 |
$ | 11.7 | ||
| Increase (decrease) in unrecognized tax benefits resulting from tax positions taken during current period |
0.4 | |||
| Increase (decrease) in unrecognized tax benefits resulting from tax positions taken in prior periods |
— | |||
| Reduction to unrecognized tax benefits as a result of settlement with taxing authorities |
(5.4 | ) | ||
| Reduction to unrecognized tax benefits as a result of the lapse of the applicable statute of limitations |
(1.5 | ) | ||
| Balance of unrecognized tax benefits at March 31, 2009 |
5.2 | |||
| Increase (decrease) in unrecognized tax benefits resulting from tax positions taken during current period |
— | |||
| Increase (decrease) in unrecognized tax benefits resulting from tax positions taken in prior periods |
2.4 | |||
| Reduction to unrecognized tax benefits as a result of settlement with taxing authorities |
(0.1 | ) | ||
| Reduction to unrecognized tax benefits as a result of the lapse of the applicable statute of limitations |
(0.6 | ) | ||
| Balance of unrecognized tax benefits at March 31, 2010 |
6.9 | |||
| Increase (decrease) in unrecognized tax benefits resulting from tax positions taken during current period |
— | |||
| Increase (decrease) in unrecognized tax benefits resulting from tax positions taken in prior periods |
— | |||
| Reduction to unrecognized tax benefits as a result of settlement with taxing authorities |
(4.9 | ) | ||
| Reduction to unrecognized tax benefits as a result of the lapse of the applicable statute of limitations |
(0.8 | ) | ||
| Balance of unrecognized tax benefits at March 31, 2011 |
$ | 1.2 | ||
The Company recognizes interest and penalties associated with uncertain tax positions as a component of income tax expense. At April 1, 2010, the Company had accrued $0.9 for interest and penalties. During fiscal year ended March 31, 2011, the Company accrued an additional $0.2 of interest and penalties and released $0.8 of interest and penalties as a result of settlements and the expiration of the statute of limitations. As of March 31, 2011, the accrual for interest and penalties was $0.3.
It is anticipated the Company will recognize approximately $1.0 of unrecognized tax benefits within the next 12 months as a result of the expected expiration of the relevant statute of limitations.
The Company is subject to taxation in the U.S. and various states and is subject to examination by those authorities. The Company’s federal statute of limitations has expired for fiscal years prior to 2006 and the relevant state statutes vary.
173
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Management regularly evaluates the Company’s tax positions taken on previously filed tax returns using information about recent court decisions and legislative activities. Many factors are considered in making these evaluations, including past history, recent interpretations of tax law, and the specific facts and circumstances of each matter. Because tax law and regulations are subject to interpretation and tax litigation is inherently uncertain, these evaluations can involve a series of complex judgments about future events and can rely heavily on estimates and assumptions. The recorded tax liabilities are based on estimates and assumptions that have been deemed reasonable by management. However, if the Company’s estimates are not representative of actual outcomes, recorded tax liabilities could be materially impacted.
On November 6, 2009, President Obama signed into law H.R. 3548, the Worker, Homeownership, and Business Assistance Act of 2009. This new law provides an optional longer net operating loss carry back period and allows most taxpayors the ability to elect a carry back period of three, four or five years (the net operating loss carry back period was previously limited to two years). This election can only be made for one year for net operating losses incurred for a tax year ending after December 31, 2007 and beginning before January 1, 2010. The Company has elected to apply this extended carry back period to its tax year ended March 31, 2009. The Company elected a carry back period of five years. The Company filed an Application for Tentative Refund with the Internal Revenue Service for this additional carry back period and subsequently received a refund in the amount of $23.8 in February 2010.
| 18. | Stock-Based Compensation |
In August 2002, the Company’s shareholders approved the Company’s 2001 Incentive Stock Option Plan (the “2001 Plan”), which allowed for the issuance of up to 3 million shares of Class A Common Stock and 1.5 million shares of Class B Common Stock. In September 2008, shareholders approved an amendment to the 2001 Plan to increase by 3 million the number of shares of Class A Common Stock available for issuance under the 2001 Plan. Under the Company’s stock option plan, options to acquire shares of common stock have been made available for grant to all employees. Each option granted has an exercise price of not less than 100% of the market value of the common stock on the date of grant. The contractual life of each option is generally ten years and the options generally vest at the rate of 10% per year from the date of grant.
The Company estimates the fair value of stock options granted using the Black-Scholes option pricing model (the “Option Model”). The Option Model requires the use of subjective and complex assumptions, including the option’s expected term and the estimated future price volatility of the underlying stock, which determine the fair value of the share-based awards. The Company’s estimate of expected term was determined based on the average period of time that options granted are expected to be outstanding considering current vesting schedules and the historical exercise patterns of existing option plans and the two-year forfeiture period (described below). The expected volatility assumption used in the Option Model is based on historical volatility over a period commensurate with the expected term of the related options. The risk-free interest rate used in the Option Model is based on the yield of U.S. Treasuries with a maturity closest to the expected term of the Company’s stock options.
The Company’s stock option agreements for options granted prior to January 2009 include a post-exercise service condition which provides that exercised options are to be held by the Company for a two-year period during which time the shares cannot be sold by the employee. If the employee’s employment was terminated voluntarily or involuntarily (other than by retirement, death or disability) during the two-year period, these stock option agreements provide the Company with the option of repurchasing the shares at the lower of the exercise price or the fair market value of the stock on the date of termination. This repurchase option was considered a forfeiture provision and the two-year period was included in determining the requisite service period over which stock-based compensation was recognized. The requisite service period initially was equal to the expected term (as discussed above) and was revised when an option exercise occurred.
174
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
If stock options with the two-year post-exercise service condition expire unexercised or an employee terminates employment after options become exercisable, no compensation expense associated with the exercisable, but unexercised, options was reversed. In those instances where an employee terminated employment before options become exercisable or the Company repurchased, compensation expense for these options was reversed as a forfeiture.
When an employee exercises stock options with the two-year post exercise service condition, the exercise proceeds received by the Company are recorded as a deposit and classified as a current liability for the two-year forfeiture period. The shares issuable upon exercise of these options are accounted for as issued when the two-year forfeiture period lapses. Until the two-year forfeiture requirement is met, the underlying shares are not considered outstanding and are not included in calculating basic earnings per share.
Commencing in January 2009, the Company granted stock options that vest over three years. Additionally in April 2009, the Company granted stock options that vest over twenty months. Options granted during fiscal 2011 vest over five years. These options were granted without a two-year post-exercise service condition. Similar to previous grants, these options have an exercise price of not less than 100% of the market value of the common stock on the date of grant and the contractual life of each option is ten years. The Company’s estimate of the expected term for these options was calculated using the Company’s historical exercise patterns of previously issued options, adjusted to exclude the impact of the two-year forfeiture period.
The Company recognized stock-based compensation of $3.0 and related tax benefits of $0.1, prior to the consideration of a valuation allowance, for the fiscal year ended March 31, 2011 and stock-based compensation of $3.8 and $3.5, respectively, and related tax benefits of $0.2 and $0.6, respectively, for the fiscal years ended March 31, 2010 and 2009, prior to the consideration of a valuation allowance. Stock-based compensation for fiscal year ended March 31, 2009 was reduced by $1.4 for unvested stock-based compensation expense previously recognized for options granted to employees terminated in the February 2009 workforce reduction.
Cash received as deposits for option exercises was $-, $- and $0.7 for fiscal years ended March 31, 2011, 2010 and 2009, respectively related to options granted prior to 2009. All employees were subject to a blackout period during fiscal 2010 and 2011 and could not exercise any options. There was no actual tax benefit realized for tax deductions from option exercises for the fiscal years ended March 31, 2011, 2010 and 2009, due to the valuation allowance on the Company’s deferred tax assets.
The following weighted average assumptions were used to value stock options granted during the fiscal years ended March 31, 2011, 2010 and 2009:
| Years ended March 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Dividend yield |
None | None | None | |||||||||
| Expected volatility |
106 | % | 99 | % | 73 | % | ||||||
| Risk-free interest rate |
1.95 | % | 2.50 | % | 1.96 | % | ||||||
| Expected term |
6.4 years | 5.9 years | 6.6 years | |||||||||
| Weighted average fair value per share at grant date |
$ | 1.49 | $ | 2.33 | $ | 2.13 | ||||||
175
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
A summary of the changes in the Company’s stock option plan for the fiscal years ended March 31, 2011, 2010 and 2009 consisted of the following:
| Shares (in millions) |
Weighted Average | Aggregate Intrinsic Value |
||||||||||||||
| Exercise Price |
Remaining Expected Term |
|||||||||||||||
| Balance, March 31, 2008 |
3.9 | 18.84 | ||||||||||||||
| Options granted |
2.3 | 6.20 | ||||||||||||||
| Options exercised |
(0.4 | ) | 7.19 | $ | — | |||||||||||
| Options canceled |
(1.6 | ) | 18.90 | |||||||||||||
| Balance, March 31, 2009 |
4.2 | 12.49 | ||||||||||||||
| Options granted |
1.3 | 2.94 | ||||||||||||||
| Options exercised |
(0.1 | ) | 9.75 | — | ||||||||||||
| Options canceled |
(1.1 | ) | 13.00 | |||||||||||||
| Balance, March 31, 2010 |
4.3 | 9.59 | ||||||||||||||
| Options granted |
1.6 | 1.79 | ||||||||||||||
| Options exercised |
— | 12.50 | — | |||||||||||||
| Options canceled |
(2.7 | ) | 8.95 | |||||||||||||
| Balance, March 31, 2011 |
3.2 | $ | 6.05 | 8.1 | $ | — | ||||||||||
| Expected to vest at March 31, 2011 |
2.3 | $ | 6.05 | 8.1 | $ | — | ||||||||||
| Options exercisable at March 31, 2011 (excluding shares in the two-year forfeiture period) |
1.1 | $ | 7.98 | 7.1 | $ | — | ||||||||||
As of March 31, 2011, the Company had $5.4 of total unrecognized compensation expense related to stock option grants, which will be recognized over the remaining weighted average period of 2.2 years.
| 19. | Employee Benefits |
Profit Sharing Plan
The Company has a qualified trustee profit sharing plan (the “Plan”) covering substantially all non-union employees. At the discretion of senior management, it was determined that the Company would not make a contribution to the Plan for the fiscal years ended March 31, 2011, 2010 and 2009. The Plan includes features as described under Section 401(k) of the Internal Revenue Code.
The Company’s contributions to the 401(k) investment funds are 50% of the first 7% of the salary contributed by each participant. Contributions of $0.3, $0.6 and $1.8 were made to the 401(k) investment funds for the fiscal years ended March 31, 2011, 2010 and 2009, respectively.
Effective October 8, 2010, the Company temporarily suspended the match of participants’ contributions to the 401(k) investment funds.
Health and Medical Insurance Plan
The Company contributes to health and medical insurance programs for its employees. The Company self-insures the first $0.15 of each employee’s covered medical claims. In fiscal year 2005, the Company established a Voluntary Employees’ Beneficiary Association (“VEBA”) for its non-union employees to fund payments made
176
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
by the Company for covered medical claims. Total health and medical insurance expense for the two plans was $13.4 for the fiscal year ended March 31, 2008. Total health and medical insurance expense for the non-union program was $4.2 and $9.6 for the fiscal years ended March 31, 2011 and 2010, respectively.
| 20. | Related Party Transactions |
The Company currently leases certain real property from a partnership affiliated with the former Chairman and CEO and a current director of the Company. Lease payments made for this property for the fiscal years ended March 31, 2011, 2010 and 2009 totaled $0.3 in each period. This lease expires on December 31, 2011 and lease payments in the fiscal year ending March 31, 2012 are expected to be $0.2. The Company does not intend to extend this lease beyond its expiration.
| 21. | Equity Transactions |
As of March 31, 2011 and 2010, the Company had 40,000 shares of 7% Cumulative Convertible Preferred Stock (par value $.01 per share) outstanding at a stated value of $25 per share. The preferred stock is non-voting with dividends payable quarterly. The preferred stock is redeemable by the Company at its stated value. Each share of preferred stock is convertible into Class A Common Stock at a conversion price of $2.96 per share. The preferred stock has a liquidation preference of $25 per share plus all accrued but unpaid dividends prior to any liquidation distributions to holders of Class A or Class B Common Stock. No dividends may be paid on Class A or Class B Common Stock unless all dividends on the Cumulative Convertible Preferred Stock have been declared and paid. There were no undeclared and accrued cumulative preferred dividends at March 31, 2011 and 2010.
The Company has reserved 750,000 shares of Class A Common Stock for issuance under KV’s 2002 Consultants Plan. These shares may be issued from time to time in consideration for consulting and other services provided to the Company by independent consultants. Since inception of this plan, the Company has issued 47,732 Class A shares as payment for certain milestones under product development agreements. Related compensation was recorded equal to the price of the issued shares.
Holders of Class A Common Stock are entitled to receive dividends per share equal to 120% of the dividends per share paid on the Class B Common Stock and have one-twentieth vote per share as Class B Common Stock in the election of directors and on certain other matters.
Private Placement of Class A Common Stock
On February 14, 2011, the Company announced that it entered into a definitive agreement with a group of institutional investors to raise approximately $32.3 of gross proceeds from a private placement of 9.95 million shares of its Class A Common Stock at $3.25 per share. The transaction closed on February 17, 2011. The Company used $20.0 of the proceeds from the financing to repay certain outstanding amounts and other outstanding obligations under its credit agreement with U.S. Healthcare. The remaining amount will be used for the launch of Makena™, payment of expenses associated with the transaction and general corporate purposes.
The Company will be required to pay certain cash amounts as liquidated damages at a rate of 1.5% of the aggregate purchase price of the shares that are unregistered per month if it does not meet certain obligations under the agreement with respect to the registration of the shares. No such amounts have been accrued as the Company expects to meet these obligations.
177
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
| 22. | Earnings (Loss) Per Share |
The following table sets forth the computation of basic loss per share:
| 2011 | 2010 | 2009 | ||||||||||||||||||||||
| Class A | Class B | Class A | Class B | Class A | Class B | |||||||||||||||||||
| Basic earnings (loss) per share: |
||||||||||||||||||||||||
| Numerator: |
||||||||||||||||||||||||
| Allocation of undistributed earnings (loss) from continuing operations |
$ | (119.0 | ) | $ | (37.2 | ) | $ | (216.4 | ) | $ | (69.2 | ) | $ | (202.7 | ) | $ | (65.1 | ) | ||||||
| Allocation of undistributed earnings from discontinued operations |
(23.3 | ) | (7.3 | ) | (5.5 | ) | (1.7 | ) | (34.7 | ) | (11.1 | ) | ||||||||||||
| Allocation of undistributed gain on sale of discontinued operations |
9.8 | 3.0 | 7.0 | 2.2 | ||||||||||||||||||||
| Allocation of undistributed earnings (loss) |
$ | (132.5 | ) | $ | (41.5 | ) | $ | (214.9 | ) | $ | (68.7 | ) | $ | (237.4 | ) | $ | (76.2 | ) | ||||||
| Denominator (in millions): |
||||||||||||||||||||||||
| Weighted average shares outstanding |
39.1 | 12.2 | 37.8 | 12.1 | 37.8 | 12.1 | ||||||||||||||||||
| Less—weighted average unvested common shares subject to repurchase |
— | — | — | — | (0.2 | ) | (0.1 | ) | ||||||||||||||||
| Number of shares used in per share computations |
39.1 | 12.2 | 37.8 | 12.1 | 37.6 | 12.0 | ||||||||||||||||||
| Basic earnings (loss) per share from continuing operations |
$ | (3.05 | ) | $ | (3.05 | ) | $ | (5.73 | ) | $ | (5.73 | ) | $ | (5.39 | ) | $ | (5.39 | ) | ||||||
| Basic earnings (loss) per share from discontinued operations |
(0.60 | ) | (0.60 | ) | (0.14 | ) | (0.14 | ) | (0.92 | ) | (0.92 | ) | ||||||||||||
| Basic earnings per share from gain on sale of discontinued operations |
0.25 | 0.25 | 0.18 | 0.18 | — | — | ||||||||||||||||||
| Basic earnings (loss) per share |
$ | (3.40 | ) | $ | (3.40 | ) | $ | (5.69 | ) | $ | (5.69 | ) | $ | (6.31 | ) | $ | (6.31 | ) | ||||||
| Diluted earnings (loss) per share: |
||||||||||||||||||||||||
| Numerator: |
||||||||||||||||||||||||
| Allocation of undistributed earnings (loss) from continuing operations |
$ | (119.0 | ) | $ | (37.2 | ) | $ | (216.4 | ) | $ | (69.2 | ) | $ | (202.7 | ) | $ | (65.1 | ) | ||||||
| Reallocation of undistributed earnings (loss) from continuing operations as a result of conversion of Class B to Class A shares |
(37.2 | ) | — | (69.2 | ) | — | (65.1 | ) | — | |||||||||||||||
| Allocation of undistributed earnings (loss) from continuing operations for diluted computation |
(156.2 | ) | (37.2 | ) | (285.6 | ) | (69.2 | ) | (267.8 | ) | (65.1 | ) | ||||||||||||
| Allocation of undistributed earnings from discontinued operations |
(23.3 | ) | (7.3 | ) | (5.5 | ) | (1.7 | ) | (34.7 | ) | (11.1 | ) | ||||||||||||
| Reallocation of undistributed earnings from discontinued operations as a result of conversion of Class B to Class A shares |
(7.3 | ) | — | (1.7 | ) | — | (11.1 | ) | — | |||||||||||||||
| Allocation of undistributed earnings from discontinued operations for diluted computation |
(30.6 | ) | (7.3 | ) | (7.2 | ) | (1.7 | ) | (45.8 | ) | (11.1 | ) | ||||||||||||
| Allocation of undistributed gain on sale of discontinued operations |
9.8 | 3.0 | 7.0 | 2.2 | — | — | ||||||||||||||||||
| Reallocation of undistributed gain on sale of discontinued operations as a result of conversion of Class B to Class A shares |
3.0 | — | 2.2 | — | — | — | ||||||||||||||||||
| Allocation of undistributed gain on sale of discontinued operations for diluted computation |
12.8 | 3.0 | 9.2 | 2.2 | — | — | ||||||||||||||||||
| Allocation of undistributed earnings (loss) |
$ | (186.8 | ) | $ | (44.5 | ) | $ | (292.8 | ) | $ | (70.9 | ) | $ | (313.6 | ) | $ | (76.2 | ) | ||||||
| Denominator (in millions): |
||||||||||||||||||||||||
| Number of shares used in basic computation |
39.1 | 12.2 | 37.8 | 12.1 | 37.6 | 12.1 | ||||||||||||||||||
| Weighted average effect of dilutive securities: |
||||||||||||||||||||||||
| Conversion of Class B to Class A shares |
12.2 | — | 12.1 | — | 12.1 | — | ||||||||||||||||||
| Number of shares used in per share computations from continuing operations |
51.3 | 12.2 | 49.9 | 12.1 | 49.7 | 12.1 | ||||||||||||||||||
| Number of shares used in per share computations from discontinuing operations |
51.3 | 12.2 | 49.9 | 12.1 | 49.7 | 12.1 | ||||||||||||||||||
| Diluted earnings (loss) per share from continuing operations |
$ | (3.05 | ) | $ | (3.05 | ) | $ | (5.73 | ) | $ | (5.73 | ) | $ | (5.39 | ) | $ | (5.39 | ) | ||||||
| Diluted earnings (loss) per share from discontinued operations |
(0.60 | ) | (0.60 | ) | (0.14 | ) | (0.14 | ) | (0.92 | ) | (0.92 | ) | ||||||||||||
| Diluted earnings per share from gain on sale of discontinued operations |
0.25 | 0.25 | 0.18 | 0.18 | — | — | ||||||||||||||||||
| Diluted earnings (loss) per share(1) |
$ | (3.40 | ) | $ | (3.40 | ) | $ | (5.69 | ) | $ | (5.69 | ) | $ | (6.31 | ) | $ | (6.31 | ) | ||||||
178
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
| (1) | For the fiscal year ended March 31, 2011, the conversion of 1,267 employee stock options, 338 preferred shares, and 8,692 shares of Class A Common Stock, issuable upon conversion of our convertible notes, were excluded from the computation of diluted earnings per share because their effect would have been anti-dilutive. For the fiscal year ended March 31, 2010, the conversion of 1,304 employee stock options, 338 preferred shares, and 8,692 of Class A Common Stock, issuable upon conversion of our convertible notes, were excluded from the computation of diluted loss per share because their effect would have been anti-dilutive. For the fiscal year ended March 31, 2009, the conversion of 245 employee stock options, 338 preferred shares, and 8,692 shares of Class A Common Stock, issuable upon conversion of our convertible notes, were excluded from the computation of diluted earnings per share because their effect would have been anti-dilutive. |
| 23. | Divestitures |
Sale of Sucralfate ANDA
On May 7, 2010, the Company received $11.0 in cash proceeds, and a right to receive an additional payment of $2.0 based on the occurrence of certain events, from the sale of certain intellectual property and other assets related to the Company’s ANDA, submitted with the FDA for the approval to engage in the commercial manufacture and sale of 1gm/10mL sucralfate suspension. All prior activities related to the intellectual property were expensed as incurred and the Company has no future involvement with the product resulting in a recognized gain equal to the cash proceeds received which is included in discontinued operations. The $2.0 will be recorded as a gain when, and if, the events stipulated in the agreement occur and payment is earned.
Sale of PDI
In March 2009, because of liquidity concerns and the Company’s expected near-term cash requirements, the Company’s Board approved management’s recommendation to market PDI for sale. PDI, formerly a wholly-owned subsidiary of the Company, develops and markets specialty value-added raw materials, including drugs, directly compressible and micro encapsulated products, and other products used in the pharmaceutical industry and other markets. As a result of the decision to sell PDI, the Company identified the assets and liabilities at PDI as held for sale at March 31, 2010. The activity of PDI is recorded in discontinued operations for all periods presented.
On June 2, 2010 (the “Closing Date”), pursuant to the Asset Purchase Agreement (the “PDI Agreement”) by and among the Company, PDI, DrugTech Corporation (“DrugTech”) and Particle Dynamics International, LLC (the “Purchaser”), the Company, PDI and DrugTech sold to the Purchaser certain assets associated with the business of PDI (as described below, the “Divested PDI Assets”).
The Divested PDI Assets, as more fully described in the PDI Agreement, consist of all of the right, title and interest in, to and under (1) the assets, rights, interests and other properties, real, personal and mixed, tangible and intangible, and goodwill owned by PDI and used by PDI on the Closing Date in its business, which consists of developing and marketing specialty value-added raw materials, including drugs, directly compressible and micro-encapsulated products and other products used in the pharmaceutical industry and other markets (including but not limited to the products specifically identified in the PDI Agreement) for the pharmaceutical, nutritional, food and personal-care industries using proprietary technologies, (2) the intellectual property owned by DrugTech related to certain PDI product lines, including U.S. and foreign patents and trademarks, and (3) certain leases with respect to facilities used by PDI that were leased by the Company. The Purchaser also agreed to hire approximately 24 employees of the Company that were employed in the operation of the PDI business.
179
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
In consideration for the Divested PDI Assets, the Purchaser (1) paid to the Company on the Closing Date $24.6 in cash, subject to certain operating working capital adjustments, and (2) assumed certain liabilities, including certain contracts. The Company incurred fees of $0.6 in connection with the transaction. The Purchaser deposited $2.0 of the purchase price in an escrow arrangement for post-closing indemnification purposes. Any uncontested amounts that remain in the escrow account in December 2011 will be paid to the Company. The operating working capital adjustments, assumed liabilities and escrow arrangement are more fully described in the PDI Agreement. In addition, the Purchaser also agreed to pay to the Company four contingent earn-out payments in total aggregate amount up to, but not to exceed, $5.5.
The four earn-out payments are determined as follows:
| • | For every dollar of EBITDA (as such term is defined in the PDI Agreement) earned by the Purchaser or its affiliates during the first year following the Closing Date with respect to sales of PDI products in excess of $7.4, the Company will receive three dollars, up to a maximum aggregate amount of $1.8 (the “First Earn-Out”). |
| • | For every dollar of EBITDA earned by the Purchaser or its affiliates during the second year following the Closing Date with respect to sales of PDI products in excess of $8.4, the Company will receive $3.00, up to a maximum aggregate amount of $1.8 (the “Second Earn-Out”). In addition, to the extent that the First Earn-Out is not fully earned during the first year following the Closing Date, for every dollar of EBITDA earned by the Purchaser or its affiliates during the second year following the Closing Date with respect to sales of PDI products in excess of $7.4, the Company will receive one and half dollars, up to a maximum aggregate amount of $1.3. However, the sum of the total aggregate earn-out payments payable after the first and the second year following the Closing Date may not exceed $3.7. |
| • | For every dollar of EBITDA earned by the Purchaser or its affiliates during the third year following the Closing Date with respect to sales of PDI products in excess of $8.9, the Company will receive three dollars, up to a maximum aggregate amount of $1.8 (the “Third Earn-Out”). In addition, to the extent that the Second Earn-Out is not fully earned during the second year following the Closing Date, for every dollar of EBITDA earned by the Purchaser or its affiliates during the third year following the Closing Date with respect to sales of PDI products in excess of $8.4, the Company will receive one and half dollars, up to a maximum aggregate amount of $1.3. |
| • | To the extent that the Third Earn-Out is not fully earned during the third year following the Closing Date, for every dollar of EBITDA earned by the Purchaser or its affiliates during the fourth year following the Closing Date with respect to sales of PDI products in excess of $8.9, the Company will receive one and half dollars up to a maximum aggregate amount of $1.3. |
The above-described earn-out payments are fully subordinated to outstanding indebtedness of the Purchaser pursuant to certain subordination arrangements entered into on the Closing Date by the Company. In connection with the sale of the Divested PDI Assets, the Company and the Purchaser also entered into a transition services agreement on the Closing Date, pursuant to which the Company agrees to provide certain transition assistance to the Purchaser for up to a one-year period.
The Company recorded a gain on sale of $5.9, net of tax, in connection with the PDI transaction in the quarter ended June 30, 2010 and a deferred gain of $2.0 related to the amounts held in escrow. Any awards that remain in escrow in December 2011 will be paid to the Company and will be recognized as a gain.
180
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
The tables below reflect the operating results of PDI for the fiscal years ended March 31, 2011, 2010 and 2009 and its net assets as of March 31, 2010.
| Year Ended March 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Net revenues |
$ | 2.7 | $ | 16.8 | $ | 20.7 | ||||||
| Cost of sales |
2.5 | 12.7 | 12.0 | |||||||||
| Gross profit |
0.2 | 4.1 | 8.7 | |||||||||
| Operating expenses: |
||||||||||||
| Selling and administrative |
(3.3 | ) | (4.5 | ) | 0.8 | |||||||
| Total operating expenses |
(3.3 | ) | (4.5 | ) | 0.8 | |||||||
| Operating income |
3.5 | 8.6 | 7.9 | |||||||||
| Income tax |
1.3 | 3.1 | 2.9 | |||||||||
| Net income |
$ | 2.2 | $ | 5.5 | $ | 5.0 | ||||||
| Gain on sale of assets (net taxes of $3.4, $- and $-) |
$ | 5.9 | $ | — | $ | — | ||||||
| March 31, 2010 |
||||
| Receivables, net |
$ | 3.6 | ||
| Inventories, net |
3.7 | |||
| Total current assets held for sale |
7.3 | |||
| Property and equipment, less accumulated depreciation |
6.7 | |||
| Intangible assets and goodwill, net |
0.6 | |||
| Total assets held for sale |
$ | 14.6 | ||
| Accounts payable and accrued liabilities |
1.1 | |||
| Total liabilities associated with assets held for sale |
$ | 1.1 | ||
On June 1, 2009, a leased facility used by PDI was damaged by an accidental fire. The incident did not affect any of the Company’s finished product manufacturing, packaging or distribution facilities. The Company received insurance proceeds of $5.6 during the fiscal year ended March 31, 2010, which were used to repair and restore the damaged facility. The insurance proceeds have been reflected as a gain within selling and administrative expenses in the periods in which payment was received, while expenditures have been reflected as operating expenses or capitalized property and equipment in the period incurred. In the second quarter of fiscal 2011, the Company received additional insurance proceeds and recorded additional gains of $3.5 which is classified as a gain within selling and administrative expenses.
Generics Business
The Company has committed to a plan to divest the Company’s generic’s business. As a result the Company decision to divest the generic’s business, the Company recorded the generic’s business in the Consolidated Statement of Operations as discontinued operations.
In connection with litigation described in greater detail in Note 15—“Commitments and Contingencies,” on June 9, 2009, the Company entered into a Distribution and Supply Agreement (the “Distribution Agreement”) with Purdue Pharma L.P., The P.F. Laboratories, Inc. and Purdue Pharmaceuticals L.P. (collectively “Purdue”).
181
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
On September 1, 2009, the Company elected for the Distribution Agreement to become effective as of September 1, 2009. Pursuant to the terms of the Distribution Agreement, Purdue provided the Company with a limited quantity of certain generic versions of OxyContin®, as specified in the Distribution Agreement, to be distributed by the Company during a limited period, and the Company paid Purdue a royalty fee and the cost of manufacturing the product supplied by Purdue.
During the three months ended December 31, 2009, the Company received from Purdue and sold to its customers all of the generic OxyContin® allotted under the Distribution Agreement and recognized net revenue of approximately $143.0 in the consolidated statement of operations. The Company recognized the revenue when it had determined that persuasive evidence of an arrangement existed, the customers’ payment ability had been reasonably assured, title and risk of ownership had been transferred to the customers, and the Company’s price to its customers was determined. Additionally, the Company recorded approximately $20.0 as cost of sales in the three months ended December 31, 2009, which included royalty fees and the cost of the generic OxyContin® supplied by Purdue. Accordingly, the Company recognized gross profit of $123.0 in the three months ended December 31, 2009 as a result of the Distribution Agreement entered into with Purdue.
We are presently in negotiations with a third party to sell our generics business.
The tables below reflect the operating results of our generics business for the fiscal years ended March 31, 2011, 2010 and 2009 and its net assets as of March 31, 2011 and 2010.
| Year Ended March 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Net revenues |
$ | 8.7 | $ | 143.2 | $ | 199.1 | ||||||
| Cost of sales |
31.4 | 98.3 | 217.3 | |||||||||
| Gross profit |
(22.7 | ) | 44.9 | (18.2 | ) | |||||||
| Operating expenses: |
||||||||||||
| Selling and administrative |
8.8 | 18.5 | 41.9 | |||||||||
| Restructuring and impairment charges |
10.0 | 46.2 | 0.6 | |||||||||
| Total operating expenses |
18.8 | 64.7 | 42.5 | |||||||||
| Operating income |
(41.5 | ) | (19.8 | ) | (60.7 | ) | ||||||
| Income tax |
(8.7 | ) | (7.2 | ) | (9.9 | ) | ||||||
| Net loss |
$ | (32.8 | ) | $ | (12.6 | ) | $ | (50.8 | ) | |||
| Gain on sale of assets (net taxes of $4.0, $5.3 and $-) |
$ | 6.9 | $ | 9.2 | $ | — | ||||||
| March 31, 2011 |
March 31, 2010 |
|||||||
| Receivables, net |
$ | 2.9 | $ | 0.2 | ||||
| Inventories, net |
6.1 | 4.3 | ||||||
| Total current assets held for sale |
9.0 | 4.5 | ||||||
| Property and equipment, less accumulated depreciation |
30.2 | 44.5 | ||||||
| Intangible assets |
14.4 | 16.2 | ||||||
| Other assets |
— | 4.7 | ||||||
| Total assets held for sale |
$ | 53.6 | $ | 69.9 | ||||
| Accounts payable and accrued liabilities |
2.5 | 4.8 | ||||||
| Total liabilities associated with assets held for sale |
$ | 2.5 | $ | 4.8 | ||||
182
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
| 24. | Warrant Liability |
As described in Note 1—“Description of Business”, the Company issued Warrants to U.S. Healthcare to purchase an aggregate of up to 20.1 million shares of Class A Common Stock at an exercise price of $1.62 per share and expiring in November 2015. While the recorded value of these aggregate Warrants of $81.6 was originally reflected as equity upon issuance, certain March 17, 2011 amendments to the arrangements with U.S. Healthcare resulted in the Warrants no longer qualifying as equity instruments. The primary provision that created this result was the depositing of $7.5 by the Company into an escrow account that would be released to the Company or to U.S. Healthcare depending on the timing of the registration and the stock price of the shares underlying the Warrants. With this contingent feature, all of the Warrants are required under ASC Topic 815 to be accounted for as liabilities until such contingency is settled. Accordingly, the Company reclassified the March 17, 2011 fair value of the Warrants from equity to liabilities and, as required by relevant accounting rules, re-valued the Warrants as of March 31, 2011. The $70.7 decrease in the value of the Warrants from March 18, 2011 to March 31, 2011 is reflected as a change in warrant liabilities in our consolidated statement of operations which resulted primarily from a decline in the Company’s stock price.
In regards to the contingent release of our escrow account to U.S. Healthcare, the Company determined that the fair value of that contingency, based on our assessed probability of meeting the requirements to have that escrow released back to us and the timing of any potential release to U.S. Healthcare was $7.5. This amount was established as a liability as of March 17, 2011 and is included with the fair value of the Warrants as the contingency is now a component of the Warrants. Because the original value of all of the Warrants have been expensed by March 31, 2011 (some immediately upon issuance and some as amortized debt discount or write off of unamortized discount upon the early retirement of the related U.S., Healthcare loans, all as described in Note 1), the Company also immediately expensed the fair value of this new contingent feature associated with those Warrants.
The following table includes the significant assumptions used in the Black-Scholes option pricing model to value the Warrants, all of which remain outstanding as of March 31, 2011, at the indicated dates:
| November 2010 Issuance |
March 2011 Issuance |
March 17, 2011 Reclassification |
March 31, 2011 Revaluation |
|||||||||||||
| Number of Warrants |
12,587,511 | 7,450,899 | 20,038,410 | 20,038,410 | ||||||||||||
| Aggregate fair value(a) |
$ | 24.0 | $ | 62.2 | $ | 175.5 | $ | 104.8 | ||||||||
| Risk free discount rate |
1.50 | % | 1.82 | % | 1.82 | % | 2.15 | % | ||||||||
| Volatility rate |
99.00 | % | 99.00 | % | 99.00 | % | 99.00 | % | ||||||||
| Dividend rate |
0.00 | % | 0.00 | % | 0.00 | % | 0.00 | % | ||||||||
| Contractual life (years) |
5.0 | 4.9 | 4.7 | 4.6 | ||||||||||||
| (a) | Prior to the value of the contingency feature described above |
The Warrants expire November 17, 2015, but may be extended by up to two additional years if the holders become subject to certain percentage ownership limitations that prevent their exercise in full at the time of their stated expiration. The Company may require that the holders exercise the Warrants before their expiration if the average of the closing prices of the Class A Common Stock for at least 30 consecutive trading days exceeds $15.00, the closing prices of the Class A Common Stock have exceeded $15.00 for 10 consecutive trading days, the shares issuable upon exercise may be resold under an effective registration statement or the resale is exempt from registration and the shares are listed on the NYSE or the National Association of Securities Dealers Automated Quotation. The Warrants also contain certain anti-dilution provisions included at the request of U. S. Healthcare, pursuant to which the number of shares subject to the Warrants may be increased and the exercise price may be decreased. These anti-dilution provisions are triggered upon certain sales of securities by the
183
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
Company and certain other events. The Warrants do not contain any preemptive rights. The Warrants also contain certain restrictions on the ability to exercise the Warrants in the event that such exercise would result in the holder of the Warrants owning greater than 4.99% of the shares of the Company’s outstanding Class A Common Stock after giving effect to the exercise. The Warrants are exercisable solely on a cashless exercise basis under which in lieu of paying the exercise price in cash, the holders will be deemed to have surrendered a number of shares of Class A Common Stock with market value equal to the exercise price and will be entitled to receive a net amount of shares of Class A Common Stock after reduction for the shares deemed surrendered. In connection with the issuance of the Warrants, the Company agreed to register up to 20,038,410 shares of our Class A Common Stock issuable upon the exercise of the Warrants.
| 25. | Quarterly Financial Results (unaudited) |
The following tables set forth selected unaudited consolidated quarterly financial information for the fiscal years ended March 31, 2011 and 2010.
| 1st Quarter |
2nd Quarter |
3rd Quarter |
4th Quarter |
Full Year |
||||||||||||||||
| Year Ended March 31, 2011 |
||||||||||||||||||||
| Net revenues(a) |
$ | 4.0 | $ | 3.0 | $ | 3.5 | $ | 16.8 | $ | 27.3 | ||||||||||
| Gross profit (loss) |
3.2 | 2.6 | 3.2 | 17.2 | 26.2 | |||||||||||||||
| Loss from continuing operations(c) |
(38.5 | ) | (28.7 | ) | (38.1 | ) | (50.9 | ) | (156.2 | ) | ||||||||||
| Income from discontinued operations(c) |
(9.0 | ) | (5.8 | ) | (8.5 | ) | (7.3 | ) | (30.6 | ) | ||||||||||
| Gain on sale of discontinued operations(c) |
12.8 | — | — | — | 12.8 | |||||||||||||||
| Net income (loss)(a)(b)(c) |
(34.6 | ) | (34.5 | ) | (46.7 | ) | (58.2 | ) | (174.0 | ) | ||||||||||
| Earnings (loss) per share from continuing operations: |
||||||||||||||||||||
| Basic and diluted—Class A and B common |
(0.77 | ) | (0.58 | ) | (0.76 | ) | (0.92 | ) | (3.05 | ) | ||||||||||
| Earnings per share from discontinued operations: |
||||||||||||||||||||
| Basic and diluted—Class A and B common |
(0.18 | ) | (0.11 | ) | (0.18 | ) | (0.13 | ) | (0.60 | ) | ||||||||||
| Earnings per share from gain on sale of discontinued operations: |
||||||||||||||||||||
| Basic and diluted—Class A and B common |
0.25 | — | — | — | 0.25 | |||||||||||||||
| Earnings (loss) per share: |
||||||||||||||||||||
| Basic and diluted—Class A and B common |
(0.70 | ) | (0.69 | ) | (0.94 | ) | (1.05 | ) | (3.40 | ) | ||||||||||
| (a) | Net revenues in the fourth quarter of fiscal year 2011 included approximately $13.7 of net revenue resulting from sale of MakenaTM. |
| (b) | The loss from continuing operations and net loss in year ended March 31, 2011 included a loss on extinguishment of debt of $106.2 in connection with extinguishing the Bridge Loan in November 2010 and March 2011. |
| (c) | As a result of the decision by the Company to sell PDI, the Company has identified the assets and liabilities of PDI as held for sale in the Company’s consolidated balance sheets at March 31, 2011, 2010 and 2009 and has segregated PDI’s operating results and presented them separately as a discontinued operation for all periods presented. The Company sold PDI on June 2, 2010 and recognized a gain of $5.9, net of tax. The Company has committed to a plan to divest the Company’s generic’s business. As a result the Company decision to divest the generic’s business, the Company has segregated generic’s business operating results and presented them separately as a discontinued operation for all periods presented. |
184
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(Dollars in millions, except per share data)
| 1st Quarter |
2nd Quarter |
3rd Quarter |
4th Quarter |
Full Year |
||||||||||||||||
| Year Ended March 31, 2010 |
||||||||||||||||||||
| Net revenues |
$ | 5.7 | $ | 2.6 | $ | 3.1 | $ | (2.3 | ) | $ | 9.1 | |||||||||
| Gross profit (loss) |
4.9 | 2.3 | 2.9 | (2.6 | ) | 7.5 | ||||||||||||||
| Income (loss) from continuing operations(a)(c)(d) |
(38.5 | ) | (39.2 | ) | 21.6 | (229.5 | ) | (285.6 | ) | |||||||||||
| Income from discontinued operations(b) |
(16.5 | ) | (14.8 | ) | 77.8 | (53.7 | ) | (7.2 | ) | |||||||||||
| Gain on sale of discontinued operations(e) |
— | — | 9.2 | — | 9.2 | |||||||||||||||
| Net income (loss)(a)(b)(c)(d)(e) |
(55.0 | ) | (54.0 | ) | 108.6 | (283.2 | ) | (283.6 | ) | |||||||||||
| Earnings (loss) per share from continuing operations: |
||||||||||||||||||||
| Basic—Class A common |
(0.77 | ) | (0.79 | ) | 0.45 | (4.59 | ) | (5.73 | ) | |||||||||||
| Basic—Class B common |
(0.77 | ) | (0.79 | ) | 0.37 | (4.59 | ) | (5.73 | ) | |||||||||||
| Diluted—Class A common |
(0.77 | ) | (0.79 | ) | 0.37 | (4.59 | ) | (5.73 | ) | |||||||||||
| Diluted—Class B common. |
(0.77 | ) | (0.79 | ) | 0.32 | (4.59 | ) | (5.73 | ) | |||||||||||
| Earnings per share from discontinued operations: |
||||||||||||||||||||
| Basic—Class A common |
(0.33 | ) | (0.30 | ) | 1.63 | (1.07 | ) | (0.14 | ) | |||||||||||
| Basic—Class B common |
(0.33 | ) | (0.30 | ) | 1.36 | (1.07 | ) | (0.14 | ) | |||||||||||
| Diluted—Class A common |
(0.33 | ) | (0.30 | ) | 1.27 | (1.07 | ) | (0.14 | ) | |||||||||||
| Diluted—Class B common |
(0.33 | ) | (0.30 | ) | 1.09 | (1.07 | ) | (0.14 | ) | |||||||||||
| Earnings per share from gain on sale of discontinued operations: |
||||||||||||||||||||
| Basic—Class A common |
— | — | 0.19 | — | 0.18 | |||||||||||||||
| Basic—Class B common |
— | — | 0.16 | — | 0.18 | |||||||||||||||
| Diluted—Class A common |
— | — | 0.15 | — | 0.18 | |||||||||||||||
| Diluted—Class B common |
— | — | 0.12 | — | 0.18 | |||||||||||||||
| Earnings (loss) per share: |
||||||||||||||||||||
| Basic—Class A common |
(1.10 | ) | (1.09 | ) | 2.27 | (5.66 | ) | (5.69 | ) | |||||||||||
| Basic—Class B common |
(1.10 | ) | (1.09 | ) | 1.89 | (5.66 | ) | (5.69 | ) | |||||||||||
| Diluted—Class A common |
(1.10 | ) | (1.09 | ) | 1.79 | (5.66 | ) | (5.69 | ) | |||||||||||
| Diluted—Class B common |
(1.10 | ) | (1.09 | ) | 1.53 | (5.66 | ) | (5.69 | ) | |||||||||||
| (a) | Operating expenses in the fourth quarter of fiscal year 2010 included $70.0 of purchased in-process research and development expense for the cash payment (the “Amendment Payment”) made upon execution of the Makena Amendment (see Note 5—“Acquisition”). |
| (b) | Operating expenses in the third quarter of fiscal year 2010 included $14.5 gain on sale for the sale of the Company’s Paragraph IV ANDA with the FDA for a generic equivalent version of GlaxoSmithKline’s Duac® gel to Perrigo Company and the sale of certain intellectual property and other assets associated with the ANDA for the generic version of Lotensin® 5-mg, 10-mg, 20-mg, and 40-mg Tablets to Huahai US, Inc. |
| (c) | Operating expenses in the fourth quarter of fiscal year 2010 included charges of $85.8 related to impairment of intangible assets and property and equipment (see Note 4—“Restructuring and Impairment Charges” and Note 10—“Intangible Assets”). |
| (d) | Income (loss) from continuing operations and net income (loss) for fiscal year 2010 included the impact of a $88.2 valuation allowance that was charged to income tax expense during the fiscal year ended March 31, 2010 for deferred tax assets that the Company determined would not be realized as tax deductions in the future. |
| (e) | During fiscal year 2010, we completed the sale to Perrigo Company of our Paragraph IV ANDA for a generic equivalent version of GlaxoSmithKline’s Duac® gel. Under the terms of the transaction, we received $14.0 from Perrigo Company at closing ($8.7, net of tax) and will receive an additional $2.0 as a milestone payment upon the completion of a successful technical transfer. Also during fiscal year 2010, we recognized a $0.5 gain from the sale of certain intellectual property and other assets associated with our ANDA for generic version of Lotensin® 5-mg, 10-mg, 20-mg, and 40-mg Tablets to Huahai US, Inc. |
185
Table of Contents
Report of Independent Registered Public Accounting Firm
Board of Directors and Shareholders
K-V Pharmaceutical Company
Bridgeton, Missouri
We have audited K-V Pharmaceutical Company’s internal control over financial reporting as of March 31, 2011, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (the COSO criteria). K-V Pharmaceutical Company’s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting, included in the accompanying Item 9A Management’s Report on Internal Control Over Financial Reporting. Our responsibility is to express an opinion on the company’s internal control over financial reporting based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audit also included performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
A material weakness is a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of the company’s annual or interim financial statements will not be prevented or detected on a timely basis. Material weaknesses have been identified and described in management’s assessment in the areas of entity-level controls (control awareness, personnel, and monitoring and remediation of deficiencies) and financial statement preparation, review procedures, and application of accounting principles (manual journal entries, account reconciliations and documentation, spreadsheets, customer and supplier agreements, application of generally accepted accounting principles, and Medicaid rebates). These material weaknesses were considered in determining the nature, timing, and extent of audit tests applied in our audit of the 2011 financial statements, and this report does not affect our report dated June 10, 2011, on those financial statements.
In our opinion, K-V Pharmaceutical Company did not maintain, in all material respects, effective internal control over financial reporting as of March 31, 2011, based on the COSO criteria. We do not express an opinion
186
Table of Contents
or any other form of assurance on management’s statements referring to any corrective actions taken by the Company after the date of management’s assessment.
We have also audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), the consolidated balance sheets of K-V Pharmaceutical Company and Subsidiaries as of March 31, 2011 and 2010 and the related consolidated statements of operations, comprehensive loss, shareholders’ equity (deficit), and cash flows for the years then ended and our report dated June 10, 2011, expressed an unqualified opinion thereon. Our report on the consolidated financial statements contains an explanatory paragraph regarding the Company’s ability to continue as a going concern.
/s/ BDO USA, LLP
Chicago, Illinois
June 10, 2011
187
Table of Contents
| Item 9. | Changes in and Disagreements With Accountants on Accounting and Financial Disclosure |
None.
| Item 9A. | Controls and Procedures |
| (a) | Management’s Report on Internal Control Over Financial Reporting |
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as defined in Rule 13a-15(f) promulgated under the Securities Exchange Act of 1934, as amended (the “Exchange Act”). We conducted an evaluation under the supervision and with the participation of our management, including the Chief Executive Officer (the “CEO”) and Chief Financial Officer (the “CFO”), of the effectiveness our internal control over financial reporting as of March 31, 2011. In conducting this evaluation, our management used the criteria established in Internal Control—Integrated Framework, issued by the Committee of Sponsoring Organizations of the Treadway Commission (“COSO”).
Internal control over financial reporting is a process designed by, or under the supervision of, the company’s principal executive and principal financial officers, or persons performing similar functions, and effected by the company’s board of directors, management, and other personnel, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with GAAP and includes those policies and procedures that:
(1) Pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company;
(2) Provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with GAAP, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and
(3) Provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the company’s assets that could have a material effect on the financial statements.
Because of inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions or that the degree of compliance with the policies or procedures may deteriorate.
Rule 12b-2 promulgated under the Exchange Act defines a material weakness as a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of the company’s annual or interim financial statements will not be prevented or detected on a timely basis.
In connection with the evaluation, the Company identified the material weaknesses in our internal control over financial reporting described below. Based on the results of the evaluation, our CEO and CFO have concluded that our internal control over financial reporting was not effective as of March 31, 2011. The following summary describes the material weaknesses that were identified:
| 1. | Material weakness in entity-level controls. We did not maintain an effective control environment or entity-level controls with respect to the risk assessment, information and communications and monitoring components of internal control. We did not: |
| a. | design adequate controls to identify and address risks critical to financial reporting, including monitoring controls and controls to ensure remediation of identified deficiencies. |
188
Table of Contents
| 2. | Material weakness surrounding financial statement preparation and review procedures and application of accounting principles. Our policies and procedures did not adequately address the financial reporting risks associated with the preparation and review of our financial statements. We did not: |
| a. | design controls over access, changes to and review of our spreadsheets used in the preparation of financial statements; |
| b. | design controls necessary to ensure that information for new and modified agreements was identified and communicated to those responsible for evaluating the accounting implications; and |
| c. | develop policies and procedures necessary to adequately address the financial reporting risks associated with the application of certain accounting principles and standards. |
Such deficiencies resulted in a reasonable possibility that a material misstatement of our annual or interim consolidated financial statements would not be prevented or detected on a timely basis.
| (b) | Changes in Internal Control over Financial Reporting |
Other than as described below under (d) Remediation Activities; some of which represent continual efforts which culminated to remediate several previously identified material weaknesses, there were no changes in our internal control over financial reporting (as such term is defined in Rules 13a-15(f) and 15d-15(f) promulgated under the Exchange Act) during the quarter ended March 31, 2011 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
| (c) | Evaluation of Disclosure Controls and Procedures |
Under the supervision and with the participation of our management, including our Chief Executive Officer and our Chief Financial Officer, we conducted an evaluation of the effectiveness of our disclosure controls and procedures, as such terms are defined in Rules 13a-15(e) and 15d-15(e) promulgated under the Exchange Act, as of March 31, 2011, the end of the period covered by this Annual Report on Form 10-K.
Disclosure controls and procedures are controls and procedures designed to ensure that information required to be disclosed in our reports filed under the Exchange Act, such as this Annual Report on Form 10-K, is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms. Disclosure controls are also designed to ensure that such information is accumulated and communicated to our management, including the Chief Executive Officer and Chief Financial Officer, as appropriate to allow timely decisions regarding required disclosure.
As a result of the material weaknesses in our internal control over financial reporting described above, our management, including our Chief Executive Officer and our Chief Financial Officer, concluded that our disclosure controls and procedures were not effective as of March 31, 2011.
| (d) | Remediation Activities |
Beginning in the fourth quarter of fiscal year 2009 and continuing through fiscal year 2011, we began designing and implementing controls, in order to remediate the material weaknesses described above in “—(a) Management’s Report on Internal Control Over Financial Reporting.” We expect that our remediation efforts, including design, implementation and testing will continue throughout fiscal year 2012. Our efforts to date and the remaining amount of time we believe is needed to remediate our material weaknesses has been impacted by, among other things, significant reductions in our workforce and changes in personnel since the fourth quarter of fiscal year 2009, management’s focus on multiple priorities including obtaining financing, returning our products to market and becoming current with our Securities and Exchange Commission (“SEC”)
189
Table of Contents
filings. While unremediated, these material weaknesses have the potential to result in our failure to prevent or detect material misstatements in our annual or interim consolidated financial statements. We will continue our remediation efforts described below and we plan to provide an update on the status of our remediation activities with future reports to be issued on Form 10-Q and Form 10-K.
As previously disclosed, in August 2008, the Audit Committee, with the assistance of legal counsel, including FDA regulatory counsel with respect to FDA matters, and other advisers, conducted an internal investigation with respect to a range of specific allegations involving, among other items, FDA regulatory and other compliance matters and management misconduct. The investigation was substantially completed in December 2008 and the investigation of all remaining matters was completed in June 2009.
The investigation focused on, among other areas, FDA and other healthcare regulatory and compliance matters, financial analysis and reporting, employment and labor issues, and corporate governance and oversight. As a result of its findings from the investigation, the Audit Committee, with the assistance of its legal counsel, including FDA regulatory counsel with respect to FDA matters, and other advisors, prepared and approved a remedial framework, which was previously disclosed in the Form 10-K for fiscal year 2009. Some of the measures included in the remedial framework are intended to remediate certain material weaknesses and are listed below.
Since the quarter ended March 31, 2009, the following actions have been taken and management believes that implementation is substantially complete with respect to the following actions to remediate the material weaknesses listed above:
| 1. | Expanded the membership of our disclosure committee to include executives with responsibilities over our operating divisions and regulatory affairs; and reviewing, revising and updating existing corporate governance policies and procedures. |
| 2. | Added quarterly executive meeting sessions between our Audit Committee and senior personnel in our legal, compliance, quality, finance and internal audit departments. |
| 3. | Reorganized and relocated our legal department adjacent to the Chief Executive Officer’s office to facilitate greater access to the legal department and more extensive involvement of the legal department in corporate governance and compliance matters. |
| 4. | Adopted measures to strengthen and enhance compliance with FDA regulations and related regulatory compliance, including: |
| • | retained outside consultants and counsel for FDA regulatory matters and, with their assistance, reviewing and revising our policies, procedures and practices to enhance compliance with the FDA’s current good manufacturing practice requirements; |
| • | enhanced compliance with FDA drug application, approval and post-approval requirements; |
| • | evaluated compliance with applicable foreign laws and regulations; and |
| • | implemented internal reporting policies pursuant to which our chief compliance officer will report periodically to the non-management members of the Board. |
| 5. | Defined and documented roles and responsibilities within the financial statement closing process including required reviews and approval of account reconciliations, journal entries and methodologies used to analyze account balances. |
| 6. | Implemented month-end closing schedules and closing checklists to ensure timely and documented completion of the financial statements. |
| 7. | Identified and implemented steps to improve communication, coordination and oversight with respect to the application of critical accounting policies and the determination of estimates. |
190
Table of Contents
| 8. | Identified and implemented steps to improve information flow between the Finance department and other functional areas within our Company to ensure that information that could affect the financial statements is considered. |
| 9. | Defined specific roles and responsibilities within the Finance department to improve accounting research and implementation of accounting policies. |
| 10. | Implemented processes and procedures to (1) identify and assess whether certain entities are appropriately exempt from the Medicaid best price calculation and (2) evaluate Public Health Service (“PHS”) pricing requests. |
| 11. | Hired a Corporate Controller, Director of Financial Reporting and a Director of Accounting with expertise in controls over financial reporting, financial statement closing procedures and GAAP. |
| 12. | Established a monthly business review process to ensure an in-depth senior management review of business segment results on a timely basis. |
| 13. | Implemented adherence to and deadline compliance with pre-established month-end, quarter-end and year-end closing schedules and closing checklists to ensure timely and documented completion of the financial statements. |
Management believes it is making progress and is continuing to proactively implement the following measures and actions in order to remediate the material weaknesses listed above:
| 1. | Develop and document comprehensive accounting policies and procedures, including documentation of the methods for applying accounting policies through detailed process maps and procedural narratives. |
| 2. | Identify and implement specific steps to improve information flow between the Finance department and other functional areas to ensure that information that could affect the financial statements, including the effects of all material agreements, is identified, communicated and addressed on a timely basis. |
| 3. | Develop and implement a policy and procedure to control the access, modification and review processes for spreadsheets that are used in the preparation of our financial statements and other disclosures. |
| 4. | Conduct further training and education of the Finance department personnel on critical accounting policies and procedures, including account reconciliations and financial statement closing procedures, to develop and maintain an appropriate level of skills for proper identification and application of accounting principles. |
| 5. | Conduct training and education for personnel outside the Finance department on critical accounting policies and procedures to improve the level of control awareness at our Company and to ensure an appropriate level of understanding of the proper application of accounting principles that are critical to our financial reporting. |
| 6. | Establish periodic meetings between the contracting functions and the Finance department to improve communication regarding the evaluation and reporting of PHS pricing requests and related matters. |
Our registered public accounting firm, BDO USA, LLP, has issued an attestation report on the Company’s internal control over financial reporting that is included with this Report.
| (e) | Management’s Conclusions on the Remediation Plan |
Management believes the remediation measures described above will strengthen our internal control over financial reporting and remediate the remaining material weaknesses we have identified. However, we have not yet implemented all of these measures, including the testing of operating effectiveness of some of those measures
191
Table of Contents
that have been implemented to date. We are committed to continuing to improve our internal control processes and will continue to diligently review our financial controls and procedures. However, any control system, regardless of how well designed, operated and evaluated, can provide only reasonable, not absolute, assurance that the control objective will be met. As we continue to evaluate and work to improve our internal control over financial reporting, we may take additional measures to address control deficiencies and/or determine not to complete certain of the remediation measures described above
| Item 9B. | Other Information |
None.
192
Table of Contents
PART III
| ITEM 10. | DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCES |
The information contained under the caption “INFORMATION CONCERNING NOMINEES AND DIRECTORS CONTINUING IN OFFICE” in the Company’s definitive proxy statement to be filed pursuant to Regulation 14(A) for its 2011 Annual Meeting of Shareholders, which involves the election of directors, is incorporated herein by this reference.
| ITEM 11. | EXECUTIVE COMPENSATION |
The information contained under the captions “EXECUTIVE COMPENSATION” and “INFORMATION AS TO STOCK OPTIONS” in the Company’s definitive proxy statement to be filed pursuant to Regulation 14(A) for its 2011 Annual Meeting of Shareholders is incorporated herein by this reference.
| ITEM 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS |
The information contained under the captions “SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS” and “SECURITY OWNERSHIP OF MANAGEMENT” in the Company’s definitive proxy statement to be filed pursuant to Regulation 14(A) for its 2011 Annual Meeting of Shareholders is incorporated herein by this reference.
| ITEM 13. | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS AND DIRECTOR INDEPENDENCE |
The information contained under the caption “COMPENSATION COMMITTEE INTERLOCKS AND INSIDER PARTICIPATION; TRANSACTIONS WITH DIRECTORS AND EXECUTIVE OFFICERS” in the Company’s definitive proxy statement to be filed pursuant to Regulation 14(A) for its 2011Annual Meeting of Shareholders is incorporated herein by this reference.
| ITEM 14. | PRINCIPAL ACCOUNTING FEES AND SERVICES |
The information contained under the caption “FEES BILLED BY INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM” in the Company’s definitive proxy statement to be filed pursuant to Regulation 14(A) for its 2011 Annual Meeting of Shareholders is incorporated herein by this reference.
193
Table of Contents
PART IV
| Item 15. | Exhibits, Financial Statement Schedules |
The following documents are filed as a part of this Report:
| Page | ||||||
| (a) 1. |
Financial Statements: |
|||||
| The following consolidated financial statements of the Company are included in Item 8—“Financial Statements and Supplementary Data” of this Report: |
||||||
| Report of Independent Registered Public Accounting Firm (BDO USA, LLP) |
115 | |||||
| Report of Independent Registered Public Accounting Firm (KPMG LLP) |
116 | |||||
| 117 | ||||||
| Consolidated Statements of Operations for the Years Ended March 31, 2011, 2010 and 2009 |
118 | |||||
| 119 | ||||||
| 120 | ||||||
| Consolidated Statements of Cash Flows for the Years Ended March 31, 2011, 2010 and 2009 |
121 | |||||
| 122 | ||||||
| 2. |
Financial Statement Schedule: |
|||||
| 196 | ||||||
| 3. |
Exhibits. See the Exhibit Index on pages 159 through 162 of this Report. Management contracts and compensatory plans are designated on the Exhibit Index. |
|||||
194
Table of Contents
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| K-V PHARMACEUTICAL COMPANY | ||
| By: | /s/ Gregory J. Divis | |
| Gregory J. Divis President and Chief Executive Officer (Principal Executive Officer) | ||
| By: | /s/ Thomas S. McHugh | |
| Thomas S. McHugh Chief Financial Officer (Principal Financial Officer) | ||
Date: June 10, 2011
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in their capacities as members of the Board of Directors of the Company on the date indicated:
Signature as of June 10, 2011
| By: | /s/ Robert E. Baldini | |
| Robert E. Baldini | ||
| By: | /s/ Gregory Bentley | |
| Gregory Bentley | ||
| By: | /s/ Mark A. Dow | |
| Mark A. Dow | ||
| By: | /s/ David S. Hermelin | |
| David S. Hermelin | ||
| By: | /s/ Joseph D. Lehrer | |
| Joseph D. Lehrer | ||
| By: | /s/ David Sidransky, M.D | |
| David Sidransky, M.D. | ||
| By: | /s/ Ana I. Stancic | |
| Ana I. Stancic | ||
195
Table of Contents
VALUATION AND QUALIFYING ACCOUNTS
Financial statements of K-V Pharmaceutical Company (separately) are omitted because KV is primarily an operating company and its subsidiaries included in the financial statements are wholly-owned and are not materially indebted to any person other than through the ordinary course of business.
| Balance at beginning of year |
Additions charged to costs and expenses |
Amounts charged to reserves |
Balance at end of year |
|||||||||||||
| Year Ended March 31, 2009: |
||||||||||||||||
| Allowance for doubtful accounts |
$ | 0.9 | $ | (0.6 | ) | $ | 0.3 | $ | — | |||||||
| Reserves and liabilities for sales allowances |
24.5 | 97.2 | 35.9 | 85.8 | ||||||||||||
| Inventory obsolescence |
— | 17.2 | 3.7 | 13.5 | ||||||||||||
| $ | 25.4 | $ | 113.8 | $ | 39.9 | $ | 99.3 | |||||||||
| Year Ended March 31, 2010: |
||||||||||||||||
| Allowance for doubtful accounts |
$ | — | $ | — | $ | — | $ | — | ||||||||
| Reserves and liabilities for sales allowances |
85.8 | (1) | 6.2 | 64.5 | 27.5 | |||||||||||
| Inventory obsolescence |
13.5 | 0.4 | 10.6 | 3.3 | ||||||||||||
| $ | 99.3 | $ | 6.6 | $ | 75.1 | $ | 30.8 | |||||||||
| Year Ended March 31, 2011: |
||||||||||||||||
| Allowance for doubtful accounts |
$ | — | $ | — | $ | — | $ | — | ||||||||
| Reserves and liabilities for sales allowances |
27.5 | 39.1 | 10.3 | 56.3 | ||||||||||||
| Inventory obsolescence |
3.3 | — | 0.8 | 2.5 | ||||||||||||
| $ | 30.8 | $ | 39.1 | $ | 11.1 | $ | 58.8 | |||||||||
196
Table of Contents
EXHIBIT INDEX
| Exhibit No. |
Description | |
| 3.1 | Certificate of Incorporation of the Company, as amended through September 5, 2008, filed as Exhibit 3.1 to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2009, is incorporated herein by reference. | |
| 3.2 | By-Laws of the Company, as amended through December 29, 2009, which was filed as Exhibit 3.2 to the Company’s Current Report on Form 8-K filed January 4, 2010, are incorporated herein by reference. | |
| 4.1 | Certificate of Designation of Rights and Preferences of 7% Cumulative Convertible preferred stock of the Company, effective June 9, 1987, and related Certificate of Correction, dated June 17, 1987, which was filed as Exhibit 4(f) to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 1987, are incorporated herein by reference. | |
| 4.2 | Indenture dated as of May 16, 2003, by and between the Company and Deutsche Bank Trust Company Americas, which was filed as Exhibit 4.1 to the Company’s Current Report on Form 8-K filed May 21, 2003, is incorporated herein by reference. | |
| 4.3 | Registration Rights Agreement dated as of May 16, 2003, by and between the Company and Deutsche Bank Securities, Inc., as representative of the several Purchasers, which was filed as Exhibit 4.2 to the Company’s Current Report on Form 8-K filed May 21, 2003, is incorporated herein by reference. | |
| 4.4 | Promissory Note, dated March 23, 2006 between MECW, LLC and LaSalle National Bank Association, which was filed as Exhibit 99 to the Company’s Current Report on Form 8-K filed March 29, 2006, is incorporated herein by reference. | |
| 4.5 | Credit Agreement, dated as of June 9, 2006, among the Company and its subsidiaries, LaSalle Bank National Association, Citibank, F.S.B. and the other lenders thereto, which was filed as Exhibit 4(g) to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2006, is incorporated herein by reference. | |
| 4.6 | Credit and Guaranty Agreement, dated November 17, 2010, among the Company, Certain Subsidiaries of K-V Pharmaceutical Company, Various Lenders, and U.S. Healthcare I, L.L.C., filed as Exhibit 4.6 to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2010, is incorporated herein by reference. | |
| 4.7 | Pledge and Security Agreement, dated November 17, 2010, among the Company, Each of the Grantors, and U.S. Healthcare I, L.L.C., which was filed as Exhibit 4.7 to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2010, is incorporated herein by reference. | |
| 4.8 | Waiver to Credit Agreement, dated as of February 9, 2011, among the Company, U.S. Healthcare I, L.L.C. and U.S. Healthcare II, L.L.C., which was filed as Exhibit 10.1 to the Company’s Quarterly Report on Form 10-Q for the quarter ended June 30, 2010, is incorporated herein by reference. | |
| 4.9 | Amendment No. 2 to Credit Agreement, dated as of March 2, 2011, among the Company, U.S. Healthcare I, L.L.C. and U.S. Healthcare II, L.L.C., which was filed as Exhibit 10.6 to the Company’s Quarterly Report on Form 10-Q for the quarter ended June 30, 2010, is incorporated herein by reference. | |
| 4.10 | Promissory Note (Tranche B-1 Term Loan Note N-1), dated November 17, 2010, among the Company and U.S. Healthcare I, L.L.C., which was filed as Exhibit 4.8 to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2010, is incorporated herein by reference. | |
197
Table of Contents
| Exhibit No. |
Description | |
| 4.11 | Promissory Note (Tranche B-1 Term Loan Note N-2), dated November 17, 2010, among the Company and U.S. Healthcare II, L.L.C., which was filed as Exhibit 4.9 to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2010, is incorporated herein by reference. | |
| 4.12 | Stock Warrant Purchase Agreement, dated as of February 10, 2011, by and among the Company, U.S. Healthcare I, L.L.C. and U.S. Healthcare II, L.L.C., which was filed as Exhibit 10.2 to the Company’s Quarterly Report on Form 10-Q for the quarter ended June 30, 2010, is incorporated herein by reference. | |
| 4.13 | Restated Stock Purchase Warrant Certificate No. W-1, dated November 17, 2010, issued by the Company to U.S. Healthcare I, L.L.C. and U.S. Healthcare II, L.L.C., which was filed as Exhibit 4.10 to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2010, is incorporated herein by reference. | |
| 4.14 | Restated Stock Purchase Warrant Certificate No. W-2, dated November 30, 2010, issued by the Company to U.S. Healthcare I, L.L.C. and U.S. Healthcare II, L.L.C., which was filed as Exhibit 4.11 to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2010, is incorporated herein by reference. | |
| 4.15 | Stock Purchase Warrant Certificate No. W-3 dated March 2, 2011, issued by the Company to U.S. Healthcare I, L.L.C. and U.S. Healthcare II, L.L.C., which was filed as Exhibit 10.5 to the Company’s Quarterly Report on Form 10-Q for the quarter ended June 30, 2010, is incorporated herein by reference. | |
| 4.16 | Registration Rights Agreement, dated November 17, 2010, among the Company, U.S. Healthcare I, L.L.C. and U.S. Healthcare II, L.L.C., which was filed as Exhibit 4.12 to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2010, is incorporated herein by reference. | |
| 4.17 | Second Amended and Restated Registration Rights Agreement, dated as of March 2, 2011, by and among the Company, U.S. Healthcare I, L.L.C. and U.S. Healthcare II, L.L.C., which was filed as Exhibit 10.7 to the Company’s Quarterly Report on Form 10-Q for the quarter ended June 30, 2010, is incorporated herein by reference. | |
| 4.18 | Securities Purchase Agreement, dated as of February 14, 2011, by and among the Company and the buyers, which was filed as Exhibit 10.1 to the Company’s Current Report on Form 8-K filed February 16, 2011, is incorporated herein by reference. | |
| 4.19 | Registration Rights Agreement, dated February 14, 2011, by and among the Company and the Investors, which was filed as Exhibit 10.2 to the Company’s Current Report on Form 8-K filed February 16, 2011, is incorporated herein by reference. | |
| 4.20 | Indenture, dated March 17, 2011, between the Company and Wilmington Trust FSB, as trustee, which was filed as Exhibit 4.1 to the Company’s Current Report on Form 8-K filed March 23, 2011, is incorporated herein by reference. | |
| 4.21 | Form of 12% Senior Secured Note due 2015, which was filed as Exhibit 4.2 to the Company’s Current Report on Form 8-K filed March 23, 2011, is incorporated herein by reference. | |
| 4.22 | Form of Purchase Agreement for the 12% Senior Secured Notes due 2015, which was filed as Exhibit 10.1 to the Company’s Current Report on Form 8-K filed March 23, 2011, is incorporated herein by reference. | |
| 4.23 | Pledge and Security Agreement, dated as of March 17, 2011, by and among the Company, the Grantors and Wilmington Trust FSB, as collateral agent, which was filed as Exhibit 10.2 to the Company’s Current Report on Form 8-K filed March 23, 2011, is incorporated herein by reference. | |
198
Table of Contents
| Exhibit No. |
Description | |
| 10.1 | Lease of the Company’s facility at 2503 South Hanley Road, St. Louis, Missouri, and amendment thereto, between the Company as Lessee and Marc S. Hermelin as Lessor, which was filed as Exhibit 10(n) to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 1983, is incorporated herein by reference. | |
| 10.2 | Amendment to the Lease for the facility located at 2503 South Hanley Road, St. Louis, Missouri, between the Company as Lessee and Marc S. Hermelin as Lessor, which was filed as Exhibit 10(p) to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 1992, is incorporated herein by reference. | |
| 10.3 | Amendment to Lease dated February 17, 1997, for the facility located at 2503 South Hanley Road, St. Louis, Missouri, between the Company as Lessee and Marc S. Hermelin as Lessor, which was filed as Exhibit 10(aa) to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 1997, is incorporated herein by reference. | |
| 10.4* | KV Pharmaceutical Company Fifth Restated Profit Sharing Plan and Trust Agreement dated May 31, 2010, filed as Exhibit 10.4 to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2010, is incorporated herein by reference. | |
| 10.5* | Fourth Amendment to and Restatement, dated as of January 2, 1997, of the KV Pharmaceutical Company 1991 Incentive Stock Option Plan, which was filed as Exhibit 10(y) to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 1997, is incorporated herein by reference. | |
| 10.6* | K-V Pharmaceutical Company 2001 Incentive Stock Option Plan, as amended, which was filed as Appendix B to the Company’s Definitive Proxy Statement on Schedule 14A filed July 29, 2008, is incorporated herein by reference. | |
| 10.7* | Form of 2001 Incentive Stock Option Plan Award Agreement for Employees, which was filed as Exhibit 10.2 to the Company’s Current Report on Form 8-K filed November 22, 2005, is incorporated herein by reference. | |
| 10.8* | Form of 2001 Incentive Stock Option Plan Award Agreement for Directors, which was filed as Exhibit 10.3 to the Company’s Current Report on Form 8-K filed November 22, 2005, is incorporated herein by reference. | |
| 10.9* | Form of Incentive Stock Option Agreement for grants to employees pursuant to the K-V Pharmaceutical Company 2001 Incentive Stock Option Plan, which was filed as Exhibit 10.1 to the Company’s Current Report on Form 8-K filed February 6, 2009, is incorporated herein by reference. | |
| 10.10* | Form of Nonqualified Stock Option Agreement for grants to employees pursuant to the K-V Pharmaceutical Company 2001 Incentive Stock Option Plan, which was filed as Exhibit 10.2 to the Company’s Current Report on Form 8-K filed February 6, 2009, is incorporated herein by reference. | |
| 10.11* | Form of Nonqualified Stock Option Agreement for grants to directors pursuant to the K-V Pharmaceutical Company 2001 Incentive Stock Option Plan, which was filed as Exhibit 10.3 to the Company’s Current Report on Form 8-K filed February 6, 2009, is incorporated herein by reference. | |
| 10.12* | Form of Incentive Stock Option Agreement for grants to employees pursuant to the K-V Pharmaceutical Company 2001 Incentive Stock Option Plan, which was filed as Exhibit 10.13 to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2009, is incorporated herein by reference. | |
199
Table of Contents
| Exhibit No. |
Description | |
| 10.13* | Form of Nonqualified Stock Option Agreement for grants to employees pursuant to the K-V Pharmaceutical Company 2001 Incentive Stock Option Plan, which was filed as Exhibit 10.14 to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2009, is incorporated herein by reference. | |
| 10.14* | Form of Nonqualified Stock Option Agreement for grants to directors pursuant to the K-V Pharmaceutical Company 2001 Incentive Stock Option Plan, which was filed as Exhibit 10.15 to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2009, is incorporated herein by reference. | |
| 10.15* | Agreement between the Company and Marc S. Hermelin, dated December 16, 1996, with supplemental letter attached, which was filed as Exhibit 10(z) to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 1997, is incorporated herein by reference. | |
| 10.16* | Amendment, dated as of October 30, 1998, to Employment Agreement between the Company and Marc S. Hermelin, which was filed as Exhibit 10(ee) to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 1999, is incorporated herein by reference. | |
| 10.17* | Amendment, dated December 2, 1999, to Employment Agreement between the Company and Marc S. Hermelin, which was filed as Exhibit 10(ii) to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2000, is incorporated herein by reference. | |
| 10.18* | Amendment, dated November 5, 2004, to Employment Agreement between the Company and Marc S. Hermelin, which was filed as Exhibit 10(a) to the Company’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2004, is incorporated herein by reference. | |
| 10.19* | Stock Option Agreement dated as of July 26, 2002, granting a stock option to Marc S. Hermelin, which was filed as Exhibit 10(rr) to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2003, is incorporated herein by reference. | |
| 10.20* | Stock Option Agreement dated as of May 30, 2003, granting a stock option to Marc S. Hermelin, which was filed as Exhibit 10(yy) to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2004, is incorporated herein by reference. | |
| 10.21* | Separation Agreement and General Release between the Company and Ronald J. Kanterman, dated as of September 2, 2009, which was filed as Exhibit 10.30 to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2009, is incorporated herein by reference. | |
| 10.22* | Consulting and Confidentiality Agreement between the Company and Ronald J. Kanterman, dated as of September 2, 2009, which was filed as Exhibit 10.31 to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2009, is incorporated herein by reference. | |
| 10.23* | Employment Agreement between the Company and David A. Van Vliet, Interim President and Interim Chief Executive Officer, dated November 23, 2009, which was filed as Exhibit 10.1 to the Company’s Current Report on Form 8-K filed November 25, 2009, is incorporated herein by reference. | |
| 10.24* | Employment Agreement between the Company and Gregory J. Divis, Jr., President of Ther-Rx Corporation, dated November 19, 2009, which was filed as Exhibit 10.2 to the Company’s Current Report on Form 8-K filed November 25, 2009, is incorporated herein by reference. | |
| 10.25* | Employment Agreement between the Company and Thomas S. McHugh, Chief Financial Officer, Treasurer, Chief Accounting Officer, dated July 15, 2010, which was filed as Exhibit 10.31 to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2010, is incorporated herein by reference. | |
| 10.26** | Asset Purchase Agreement by and between the Company and VIVUS, Inc., dated as of March 30, 2007, which was filed as Exhibit 10.1 to the Company’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2007, is incorporated herein by reference. | |
200
Table of Contents
| Exhibit No. |
Description | |
| 10.27 | Asset Purchase Agreement by and among the Company, Cytyc Prenatal Products Corp. and Hologic, Inc., dated as of January 16, 2008, which was filed as Exhibit 10(ii) to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2008, is incorporated herein by reference. | |
| 10.28 | Amendment, dated January 8, 2010, to the Asset Purchase Agreement by and among the Company, Cytyc Prenatal Products Corp. and Hologic, Inc., dated as of January 16, 2008, which was filed as Exhibit 10.48 to the Company’s Annual Report on Form 10-K for the fiscal year ended March 31, 2009, is incorporated herein by reference. | |
| 10.29 | Form of Indemnification Agreement for directors, which was filed as Exhibit 10.1 to the Company’s Current Report on Form 8-K filed October 29, 2008, is incorporated herein by reference. | |
| 10.30* | Form of Indemnification Agreement for Interim Chief Executive Officer, which was filed as Exhibit 10.1 to the Company’s Current Report on Form 8-K filed December 31, 2009, is incorporated herein by reference. | |
| 10.31* | Form of Indemnification Agreement for Certain Executive Officers, which was filed as Exhibit 10.2 to the Company’s Current Report on Form 8-K filed December 31, 2009, is incorporated herein by reference. | |
| 10.32* | Form of Retention Agreement, which was filed as Exhibit 10.1 to the Company’s Current Report on Form 8-K filed May 5, 2009, is incorporated herein by reference. | |
| 10.33 | Consent Decree of Permanent Injunction as filed in the United States District Court, Eastern District of Missouri, Eastern Division on March 2, 2009, which was filed as Exhibit 99.2 to the Company’s Current Report on Form 8-K filed March 3, 2009, is incorporated herein by reference. | |
| 10.34 | Plea Agreement, Guidelines Recommendations and Stipulations as filed in the United States District Court, Eastern District of Missouri, Eastern Division on March 2, 2010, which was filed as Exhibit 99.1 to the Company’s Current Report on Form 8-K filed March 3, 2010, is incorporated herein by reference. | |
| 10.35 | Settlement Agreement dated as of June 9, 2009, among Purdue Pharma L.P., The P.F. Laboratories, Inc., Purdue Pharmaceuticals L.P. and the Company, which was filed as Exhibit 10.1 to the Company’s Current Report on Form 8-K filed June 10, 2009, is incorporated herein by reference. | |
| 10.36** | Patent License Agreement, dated as of June 9, 2009, among Purdue Pharma L.P., The P.F. Laboratories, Inc., Purdue Pharmaceuticals L.P. and the Company, which was filed as Exhibit 10.2 to the Company’s Current Report on Form 8-K filed June 10, 2009, is incorporated herein by reference. | |
| 10.37** | Distribution and Supply Agreement, dated as of June 9, 2009, between Purdue Pharma L.P. and the Company, which was filed as Exhibit 10.3 to the Company’s Current Report on Form 8-K filed June 10, 2009, is incorporated herein by reference. | |
| 10.38 | Asset Purchase Agreement dated as of June 2, 2010 by and among Particle Dynamics International, LLC, Particle Dynamics, Inc., Drug Tech Corporation and KV Pharmaceutical Company, which was filed as Exhibit 2.1 to the Company’s Current Report on Form 8-K filed June 8, 2010, is incorporated herein by reference. | |
| 10.39 | Amendment No. 2, dated February 3, 2011, to the Asset Purchase Agreement by and among the Company, Cytyc Prenatal Products Corp. and Hologic, Inc., dated as of January 16, 2008, which was filed as Exhibit 10.1 to the Company’s Current Report filed February 8, 2011, is incorporated herein by reference. | |
201
Table of Contents
| Exhibit No. |
Description | |
| 21 | List of Subsidiaries, filed herewith. | |
| 23.1 | Consent of BDO USA, LLP, filed herewith. | |
| 23.2 | Consent of KPMG LLP, filed herewith. | |
| 31.1 | Certification of Chief Executive Officer pursuant to Rule 13a-14(a) or Rule 15d-14a(a), as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002, filed herewith. | |
| 31.2 | Certification of Chief Financial Officer pursuant to Rule 13a-14(a) or Rule 15d-14a(a), as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002, filed herewith. | |
| 32.1 | Certification pursuant to 18 U.S. C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, filed herewith. | |
| 32.2 | Certification pursuant to 18 U.S. C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, filed herewith. | |
| * | Management contract or compensation plan. |
| ** | Confidential portions of this exhibit have been redacted and filed separately with the SEC pursuant to a confidential treatment request in accordance with Rule 24b-2 of the Securities Exchange Act of 1934, as amended. |
202
