Attached files
| file | filename |
|---|---|
| EX-23.1 - CardioGenics Holdings Inc. | v209469_ex23-1.htm |
| EX-32.1 - CardioGenics Holdings Inc. | v209469_ex32-1.htm |
| EX-31.1 - CardioGenics Holdings Inc. | v209469_ex31-1.htm |
| EX-31.2 - CardioGenics Holdings Inc. | v209469_ex31-2.htm |
| EX-21.1 - CardioGenics Holdings Inc. | v209469_ex21-1.htm |
| EX-23.2 - CardioGenics Holdings Inc. | v209469_ex23-2.htm |
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
_________________________
FORM
10-K
_________________________
|
|
þ
|
Annual report pursuant to
Section 13 or 15(d) of the Securities Exchange Act of
1934
|
|
|
for
the fiscal year ended October 31,
2010
|
OR
|
|
¨
|
Transition report pursuant to
Section 13 or 15(d) of the Securities Exchange Act of
1934
|
|
|
for
the transition period from __________ to
__________.
|
Commission
file number: 000-28761
CARDIOGENICS
HOLDINGS INC.
(Exact
name of registrant as specified in its charter)
|
Nevada
(State
or other jurisdiction of
incorporation
or organization)
|
88-0380546
(I.R.S.
Employer
Identification
Number)
|
|
6295
Northam Drive, Unit 8, Mississauga, Ontario L4V 1W8
(Address
of principal executive offices) (Zip code)
(905)
673-8501
(Registrant’s
telephone number, including area code)
Securities
registered pursuant to Section 12(b) of the
Act: None
|
|
|
Securities
registered pursuant to Section 12(g) of the Act:
Common
Stock—$0.00001 par value
Series
2 Class B Common Stock—$0.00001 par value
Series
3 Class B Common Stock—$0.00001 par
value
|
|
Indicate
by check mark if the registrant is a well-known seasoned issuer, as defined in
Rule 405 of the Securities Act. Yes ¨ No þ
Indicate
by check mark if the registrant is not required to file reports pursuant to
Section 13 or Section 15(d) of the Act. Yes ¨ No þ.
Indicate
by check mark whether the registrant (1) has filed all reports required to be
filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the
preceding 12 months (or for such shorter period that the registrant was required
to file such reports), and (2) has been subject to such filing requirements for
the past 90 days. Yes þ No ¨.
Indicate
by check mark whether the registrant has submitted electronically andposted on
its corporate Web site, if any, every Interactive Data File required tobe
submitted and posted pursuantto Rule 405 of Regulation S-T (§232.405 of this
chapter) during the preceding 12 months (or for such shorter period that the
registrant was required to submit and post such files). Yes ¨ No ¨.
Indicate
by check mark if disclosure of delinquent filers pursuant to Item 405 of
Regulation S-K is not contained herein, and will not be contained, to the best
of registrant’s knowledge, in definitive proxy or information statements
incorporated by reference in Part III of this Form 10-K or any amendment to this
Form 10-K. þ
Indicate
by check mark whether the registrant is a large accelerated filer, an
accelerated filer, non-accelerated filer or a small. See definition
of “large accelerated filer, accelerated filer and smaller reporting company” in
Rule 12b-2 of the Exchange Act. (Check one):
|
Large
accelerated filer ¨
|
Accelerated
filer ¨
|
Non-accelerated
filer ¨ (Do
not check if smaller reporting company)
|
Smaller
reporting company þ
|
Indicate
by check mark whether the registrant is a shell company (as defined in Rule
12b-2 of the Act). Yes ¨ No þ.
The
aggregate market value of the registrant’s voting and non-voting common stock
held by non-affiliates on January 10, 2011 (based on the closing stock price on
the OTC Bulletin Board) on such date was approximately $33,418,717.
As of
January 10, 2011 the Registrant had the following number of shares of its
capital stock outstanding: 29,320,647 shares of Common Stock, 1 share of Series
1 Preferred Voting Stock, par value $0.0001, representing 14 exchangeable shares
of the Registrant’s subsidiary, CardioGenics ExchangeCo Inc., which are
exchangeable into 24,388,908 shares of the Registrant’s Common Stock, 380,931
shares of Series 2 Class B Common Stock and 21,500 shares of Series 3 Class B
Common Stock.
CARDIOGENICS
HOLDINGS INC.
ANNUAL
REPORT ON FORM 10-K
FOR
THE FISCAL YEAR ENDED OCTOBER 31, 2010
TABLE
OF CONTENTS
|
Page
|
|
|
Part
I
|
1
|
|
Item
1. Business
|
1
|
|
Item
1A. Risk Factors
|
12
|
|
Item
1B. Unresolved Staff Comments
|
19
|
|
Item
2. Properties
|
19
|
|
Item
3. Legal Proceedings
|
19
|
|
Item
4. (Removed and Reserved)
|
20
|
|
Part
II
|
20
|
|
Item
5. Market for Registrant’s Common Equity, Related Stockholder Matters and
Issuer Purchases of Securities
|
20
|
|
Item
6. Selected Financial Data
|
22
|
|
Item
7. Management’s Discussion and Analysis or Plan of
Operation
|
22
|
|
Item7A.
Quantitive and Qualitative Disclosure about Market Risk
|
28
|
|
Item
8. Financial Statements and Supplementary Data
|
28
|
|
Item
9. Changes in and Disagreements with Accountants on Accounting and
Financial Disclosure
|
28
|
|
Item
9A. Controls and Procedures
|
29
|
|
Item
9B. Other Information
|
29
|
|
Part
III
|
30
|
|
Item
10. Directors, Executive Officers and Corporate Governance
|
30
|
|
Item
11. Executive Compensation
|
32
|
|
Item
12. Security Ownership of Certain Beneficial Owners and Related
Stockholder Matters
|
35
|
|
Item
13. Certain Relationships and Related Transactions, and Director
Independence
|
36
|
|
Item
14. Principal Accounting Fees and Services
|
36
|
|
Part
IV
|
38
|
|
Item
15. Exhibits, Financial Statement Schedules
|
38
|
i
Part
I
ITEM
1. BUSINESS
This
Annual Report on Form 10-K contains forward-looking statements within the
meaning of Section 27A of the Securities Act of 1933, as amended (the “Securities Act”), and Section
21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). Such
statements are based upon current expectations that involve risks and
uncertainties. Any statements contained herein that are not statements of
historical fact may be deemed to be forward-looking statements. Words such as
“may,” “will,” “should,” “estimates,” “predicts,” “potential,” “continue,”
“strategy,” “believes,” “anticipates,” “plans,” “expects,” “intends” and similar
expressions are intended to identify forward-looking statements. Our discussions
relating to our liquidity and capital resources, our business strategy, our
competition, and the future of our market segment, our acquisition of
CardioGenics Inc., an Ontario Canada corporation (“CardioGenics”), among
others, contain such statements. Our actual results and the timing of certain
events may differ significantly from the results discussed in the
forward-looking statements.
Our
forward-looking statements in this Annual Report on Form 10-K are based on
management’s current views and assumptions regarding future events and speak
only as of their dates. We undertake no obligation to publicly update
or revise any forward-looking statements, whether as a result of new
information, future events or otherwise, except as required by the federal
securities laws. Unless the context requires otherwise, the terms
“we,” “us” and “our” refer to CardioGenics Holdings Inc., our predecessors and
subsidiaries. Our acquisition of CardioGenics as discussed in this Annual Report
on Form 10-K is sometimes referred to as the “CardioGenics
Acquisition.”
OVERVIEW
Prior to
the CardioGenics Acquisition, our primary business was providing financial and
investment information to the investment community which we have been doing
since 1989. In May 1999, we began offering our services on a
subscription fee basis to the general public for the first time through our
website at jagnotes.com. Through our website and our traditional
fax-based service, we offer timely financial data, reports and
commentary.
Our
online services currently consist of a subscription-based service that offers
two specific products, the JAGNotes (Upgrade/Downgrade) Report and the Rumor
Room, providing timely market reports, including breaking news and potentially
market moving information. We currently derive revenues primarily
from the sale of subscriptions.
In July
2009, we consummated the CardioGenics Acquisition and the main focus of our
business switched from offering our customers fee-based financial information to
the development of products targeting the immunoassay segment of the
point-of-care in vitro diagnostic (“IVD”) testing market. See “—Our
Strategy—Acquisition of CardioGenics.” In order to better reflect the
new focus of our business, we changed our name to CardioGenics Holdings Inc. in
October 2009.
We are a
Nevada corporation. Our address is 6295 Northam Drive, Unit 8, Mississauga,
Ontario, Canada L4V 1W8, and our telephone number is 905-673-8501.
COMPANY
BACKGROUND
JagNotes,
Inc.
We have
been providing financial information to the investment community since 1989. In
May 1999, we began offering our services on a subscription fee basis to the
general public for the first time through our website at jagnotes.com. Through
our website and our traditional fax-based service, we offer timely financial
data, reports and commentary.
Our
online services currently consist of a subscription-based service that offers
two specific products, the JAGNotes (Upgrade/Downgrade) Report and the Rumor
Room, providing timely market reports, including breaking news and potentially
market moving information. We currently derive revenues primarily from the sale
of subscriptions.
From 1989
to 1992, we operated as an unincorporated business entity. In 1992, we
incorporated in the State of New Jersey as New Jag, Inc. On December 14, 1993,
JagNotes, Inc. merged with and into New Jag Inc., and we changed our name to
JagNotes, Inc. We operated as JagNotes, Inc. until March 1999 when we were
acquired by Professional Perceptions, Inc., a Nevada corporation, which
subsequently changed our name to JagNotes.com Inc.
Until
1999, we targeted only a limited audience of financial professionals and did not
engage in organized sales and marketing efforts. In 1999, we decided to change
focus by expanding onto the Internet and targeting retail subscribers with the
hope of expanding our subscriber base and business.
1
We
undertook a corporate reorganization in January 2002 in order to distinguish and
better manage separate areas of business. On January 4, 2002, we formed JAG
Media LLC, a Delaware limited liability company and wholly-owned subsidiary. The
assets and liabilities of our current fax and Internet subscription business
were transferred to JAG Media LLC. In order to better reflect the overall
business in which we expected to engage and the corporate structure we intended
to use to conduct that business, we changed our name from JagNotes.com Inc. to
JAG Media Holdings, Inc. effective April 8, 2002.
On
November 24, 2004, through one of our subsidiaries, Pixaya (UK) Limited (“Pixaya”), we purchased certain
development stage software products and related assets in the United Kingdom
from TComm Limited, a company organized in the United Kingdom. We subsequently
changed the name of our subsidiary, JAG Media LLC, to Pixaya LLC in order to
better reflect its role as owner of Pixaya and primary provider of support for
our Pixaya products in the United States. Due to cash constraints, we ceased
financing development and marketing by Pixaya of our SurvayaCam product, a
mobile surveillance system which streams live video in real time from the point
of use back to a control center and, if desired, to other locations. To date, we
have only made minimal sales of SurvayaCam as part of our prior marketing and
distribution efforts.
In light
of the difficulties we encountered in growing our JAG Notes subscription
business and Pixaya business, we began seeking merger and acquisition
candidates, in related and unrelated lines of businesses, to augment our current
business. On July 31, 2009, we completed the acquisition of CardioGenics, a
developer of products targeting the immunoassay segment of the Point-of-Care IVD
testing market, based in Ontario, Canada. See “—Our Acquisition of
CardioGenics.” On February 11, 2010 we sold our Pixaya LLC subsidiary, and its
related JAG Notes subscription and Pixaya businesses, since we believe it would
be more beneficial for our resources to be devoted solely to the development and
commercialization of our core CardioGenics products.
CardioGenics
Inc.
CardioGenics
was founded in Toronto, Canada in 1997 by Dr. Yahia Gawad to develop technology
and products targeting the immunoassay segment of the IVD testing market. These
include:
|
|
§
|
The
QL Care Analyzer (the “QLCA”), a
state-of-the-art proprietary Point-of-Care (“POC”) immunoassay
analyzer;
|
|
|
§
|
A
series of immunoassay tests to detect cardiac markers (the “Cardiovascular Tests”);
and,
|
|
|
§
|
Paramagnetic
beads developed through its proprietary method, which improves their light
collection (the “Beads”).
|
OUR
INDUSTRY
CardioGenics IVD POC Testing
Markets
IVD
Market
In
vitro diagnostics (IVD) refers to testing that aims for the identification of
disease states outside the body, using samples such as body fluids (blood,
urine) and tissues (biopsies and tissue sections). The IVD is a well established
market, offering essential products (tests, components and machinery) used by
physicians and clinical chemistry personnel to assess disease conditions. The
world market for IVD is estimated at $42 billion in 2007 and is expected to grow
6% annually to $56.3 billion by 20121.
North America, Europe, Japan and Western Europe currently make up 81% of the
total IVD market, and this is expected to decrease to 76% by 2012 as China and
India become more significant players in the IVD market. Sales of IVD products
in emerging economies in Latin America and Eastern Europe are expected to grow
from 4% of the market in 2007 to 5% in 2012. Overall, sales growth of IVD
products in emerging markets will account for 10-20% annual growth in the IVD
market, while the developed world will see annual growth of 3-6%.2
1 This
includes all laboratory, hospital-based products and OTC products, according to
Kalorama Information, The Worldwide Market for In Vitro Diagnostics Tests,
6th
Edition, June 2008
2 Kalorama
Information, The Worldwide Market for In Vitro Diagnostics Tests, 6th
Edition, June 2008, p3
2
The
following table summarizes the market size and projections of the IVD market and
the sub-sectors where our products will compete:
|
Product
|
2007
|
2008
|
2009
|
2010
|
2011
|
|||||||||||||||
|
IVD (billions)
|
42.1 | 44.5 | 47.1 | 49.1 | 52.9 | |||||||||||||||
|
Immunoassay
Testing (billions)
|
4.185 | 4.435 | 4.695 | 4.975 | 5.260 | |||||||||||||||
|
POC
Testing (billions)
|
1.625 | 1.715 | 1.815 | 1.910 | 2.02 | |||||||||||||||
|
Cardiac
Marker Tests (millions)
|
425 | 471.75 | 523.64 | 581.24 | 645.17 | |||||||||||||||
In 2007,
16 top tier IVD companies occupied 78% of the global market ($32
billion). Since 2005, there has been a trend toward consolidation at
all levels of the IVD market. In 2007, three top tier companies, DPC,
Dade Behring and Bayer Diagnostics, merged to become Siemens Medical
Diagnostics.
Immunoassay
Market
The 2007
world market for all immunoassays excluding infectious diseases is estimated at
$4,185 million3, and
by 2012 the market is projected to grow by 6% annually to reach $5,605 million
worldwide. Immunoassays sales for cardiac markers were 785 million in 2007, or
12% of market, and this is expected to increase to 1,050 million (12%) by
20124. The following
Table illustrates the relationships between the top IVD companies and sales of
IVD products.
Revenue History of Leading
Immunoassay Vendors, $ million 2005-20075
|
2007
|
2006
|
2005
|
||||||||||
|
Abbott
Diagnostics
|
2,100 | 1,900 | 1,800 | |||||||||
|
Siemans/Dade
Behring
|
825 | 785 | 750 | |||||||||
|
Siemens/Bayer
|
750 | 714 | 680 | |||||||||
|
Beckman
Coulter
|
596 | 484 | 402 | |||||||||
|
Siemens/DPC
|
595 | 517 | 473 | |||||||||
|
Roche
|
575 | 509 | 450 | |||||||||
|
bioMérieux
|
363 | 362 | 353 | |||||||||
|
Fujirebio
|
299 | 277 | 279 | |||||||||
|
Ortho
|
200 | 190 | 160 | |||||||||
|
TOTAL
|
6,303 | 5,738 | 5,347 | |||||||||
Immunoassay
testing segment of the IVD market is characterized by:
· Expanding
opportunities after completion of the human genome project.
· Demand
for automated and sensitive POC immunoassay analyzers.
3 $6.685
million including infectious diseases
4 Kalorama
Information, The Worldwide Market for In Vitro Diagnostics Tests, 6th
Edition, June 2008, p401
5
Estimated. Kalorama Information, The Worldwide Market for In Vitro Diagnostics
Tests, 6th
Edition, June 2008, p402
3
|
·
|
Search
for an ideal POC platform.
|
|
·
|
Increased
mergers and acquisition among top tier IVD companies to achieve more
complete product lines
|
|
·
|
Greater
cooperation between test developers and top tier IVD
companies.
|
Over the
next 5-10 years, the immunoassay business will see:
|
|
·
|
The
continued automation of routine immunoassays – thyroid, anemia, fertility,
therapeutic drug monitoring and drugs of abuse;
and
|
|
|
·
|
More
new assays and test categories for disease risk evaluation.6
|
Point-Of-Care
(POC) Testing Market
Point Of
Care (POC) testing refers to a laboratory assay that can be performed outside of
a centralized facility, with results available within minutes. POC testing is
divided into personal use tests, such as pregnancy tests, and professional use
tests, that are administered in a physician‘s office or hospital emergency ward.
Our tests will compete in the professional use testing market
sector.
The
market for professional7 POC immunoassays is
estimated at $1,625 million in 2007 and with the 14% projected growth, this
market will reach $2,770 million in 2012. It is anticipated that most of the
growth will come from increased use of cardiac markers and new assays for cancer
markers and diabetes/cardiac disease markers. The market for professional POC
tests for cardiac markers is estimated at $425 million in 2007 (11%) and this is
expected to increase to $850 million (15%) by 2012.7
There is
a wide perception that POC tests are more expensive than lab-based tests and
that patient test results are lost to the historical record. There is also the
perception that once the patient leaves the acute care area, the baseline POC
tests done in that unit are of little value because the POC testing results do
not correlate with lab-based systems.
Two
critical characteristics are necessary for potential POC test products to become
more prevalent; POC testing results must correlate with lab results and the POC
devices must be more consistent and robust in delivering those
results.
The
impact of POC testing on improving patients’ care is clear and has been well
documented. Further, the impact of POC testing on saving healthcare resources
was also demonstrated by numerous agencies and institutions.
Cardiovascular
Disease Testing Market
Cardiac
markers are proteins released from heart muscle when it is damaged as a result
of a heart attack (myocardial infarction), when the blood supply to part of the
heart is interrupted. Physicians use cardiac markers in two ways – to diagnose a
cardiac event in a hospital emergency room or within the hospital or to evaluate
a risk of a cardiovascular event occurring. The routine markers of myocardial
infarction – CK-MB, troponin and myoglobin and recently BNP are used in the
acute care and tests such as cholesterol are used to evaluate risk.
The world
market for cardiac markers is estimated at $740 million in 2007, and with
projected annual growth of 5%, will reach $1,050 million in 2012.
Until
recently, Troponin and CK-MB were the lead cardiac markers. Brain Natriuetic
Pepetide (BNP) was recently introduced to differentiate between a myocardial
infarction and heart failure. A number of companies are focused on developing
new cardiac markers.
6 Kalorama
Information, The Worldwide Market for In Vitro Diagnostics Tests, 6th
Edition, June 2008
7
Administered in a professional setting, i.e. not home
tests.
4
Magnetic
Particles Market
Magnetic
particles, or beads, are widely used as the solid phase for binding tests for
automating and simplifying the methods for isolation and detection of
biomolecules in both research and routine clinical laboratories. Eight of the
top 10 IVD companies employ magnetic particles in their fully automated
analyzers.
An
independent 2006 market research report, prepared for CardioGenics by Adventus
Research Inc. (the “Adventus Report”) and sponsored by the National Research
Council of Canada (NRC), estimated the market for magnetic beads for
immunoassays and molecular diagnostics to be approximately $900 million (between
$833 million and $1.3 billion). The report of market size did not include
magnetic beads produced in-house by some of the IVD test manufacturers or beads
produced for research applications. The Adventus Report was conducted using
several methods, including interviews with leading particle-manufacturers and
the end-users, published industry reports and data from leading IVD
manufacturers.
As stated
in the Adventus Report, according to Dynal, a leading magnetic beads
manufacturer, the largest part of its Molecular Systems’ business is OEM sales
of magnetic beads to IVD companies. Dynal stated that “the IVD market is very
large, and still growing. However, the magnetic bead-based part of this market
is growing at an even higher rate per year”.8 According to Dynal,
immunoassays make up more than USD 4 billion of the IVD market, and magnetic
beads are now the gold standard for immunoassay testing, as opposed to older
technologies such as microtitre plate based tests. Nucleic acid testing makes up
a smaller portion of the IVD market, USD 2 billion, but is fast growing.
Magnetic beads are also the most common solid phase employed in this
market.
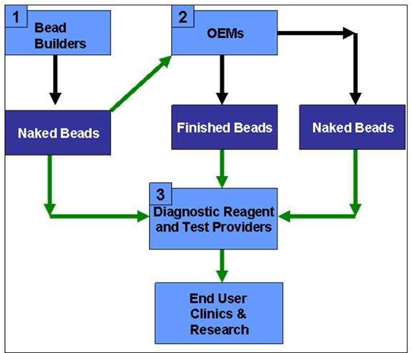
Furthermore,
according to Dynal, as stated in the Adventus Report, end-user business rather
than OEM business (referred to as functionalized and naked beads markets
respectively) goes to research and routine laboratories within Genomics,
Expression Profiling and Proteomics. The market size for Genomics, including DNA
and RNA extraction and purification products was USD 300 million in 2001 while
the market size of Pharmacogenomics was estimated to be USD 2.3 billion in
2001.
8 Adventus
Report
5
As stated
in the Adventus Report, according to Gen-Probe, which is a leading DNA clinical
testing company, other markets that are employing magnetic beads as a solid
phase are growing also. Further, magnetic particles are used for Separation of
Microorganisms in Food and Water Testing and also for HLA testing for organ
transplantation.
Source:
Gen-Probe presentation- May 2006
OUR
PRODUCTS
The CardioGenics
Products
QL
Care Analyzer

The QLCA
represents a shift in the design of POC analyzers. The QLCA is a small,
portable, stand-alone and completely automated point-of-care immunoassay
analyzer. The QLCA has successfully miniaturized lab test technology, and
combined it with a simplified mechanical design and proprietary triggering
mechanism.
The QLCA
uses a proprietary self-metering cartridge to perform immunoassay tests at the
POC. Each cartridge is pre-loaded with our beads, which have been coated with
specific bioluminescent proteins linked to the target marker. A drop of whole
blood added to the Cartridge creates the chemiluminescent reaction needed to
deliver sensitive and accurate test results. Operation of the QLCA does not
require specialized training and testing can be completed in 15
minutes.
POC
immunoassay analyzers are not new; however, none of the commercial analyzers can
replicate the sensitivity and accuracy of a test done in a medical lab. The QLCA
delivers the required laboratory sensitivity and accuracy. The QLCA employs
chemical light generation or “chemiluminescence“ (“CL“), the same technology
used in the medical labs. The QLCA uses a patented automated electronic process
to trigger CL, which enhances light collection, speeds up marker binding and
increases sensitivity.
6
We have
rigorously tested the QLCA protocols and have compared our test results against
medical laboratory test data. Based on these internal test results, we have
consistently met or exceeded the sensitivity standards of medical laboratory
immunoassay equipment.
Cardiovascular
Tests
To
support the use of the QLCA, we have developed four immunoassay tests designed
to identify cardiac markers in the blood at the time of a heart
attack.
|
Test
|
Description | ||
|
Troponin
I (TnI)
|
§
|
TnI
testing is the current routine testing for a heart
attack.
|
|
|
§
|
TnI
is a heart muscle protein, released in the bloodstream shortly after a
heart attack (myocardial infarction or MI).
|
||
|
§
|
Current
laboratory analyzers cannot detect TnI before 4-6 hours after the onset of
symptoms, when TnI concentration in the blood reaches its detection
threshold.
|
||
|
§
|
Our
test will take only 15 minutes to deliver quantitative results, allowing
physicians to obtain much more rapid results and therefore accelerate
patient triage.
|
||
|
Plasminogen
Activator Inhibitor Type-1 (PAI-1)
|
§
|
This
test will help to optimize the performance of a heart drug (“tPA” or tissue
Plasminogen Activator), a clot buster used as the first line of therapy
for MI patients.
|
|
|
§
|
This
proprietary whole blood test will quantify PAI-1 levels within 15
minutes.
|
||
|
§
|
Forty
percent of patients do not respond to tPA, a fact recognized only after
the “golden hour” (the time period in which permanent heart damage can be
prevented) has passed.
|
||
|
Heart
Failure Risk Stratification (HFRS)
|
§
|
We
have discovered a family of related proteins that are released into the
bloodstream during heart failure.
|
|
|
§
|
We
are developing a proprietary test, the Heart Failure Risk Stratification
or HFRS test to stratify the risk of death in patients with heart failure,
thus permitting the initiation of appropriate therapy at an early
stage.
|
||
|
Heart
Failure Genomics Risk (HFGR)
|
§
|
We
are developing a proprietary HFGR test that predicts the response of heart
failure patients to routinely administered drugs.
|
|
|
§
|
The
need to measure the precise response to these drugs in a timely manner
would minimize the trial and error methods now used by doctors to optimize
drugs best suited to each
patient.
|
||
These
tests are designed to be administered in the diagnostic and management process
of patients with heart disease. The full scope of our core technology, as well
as the know-how we have developed respecting aspects of chemical entrapment in
bioassays, are covered under our patent applications.
Upon
receipt of FDA approval, we intend to market the QLCA and the Cardiovascular
Tests through a major IVD distributor. We have initiated preliminary discussions
with several of the Tier 1 IVD companies, and we anticipate that we will
commence negotiations with one or more distribution partners before we receive
FDA approval. In
accordance with industry practice, we intend to enter into a license agreement
with our distribution partner for the manufacture and distribution of our
products.
7
Paramagnetic
Beads
Medical
laboratories widely use paramagnetic particles as a solid surface in
heterogeneous immunoassay tests utilizing the process of phase separation done
by eletromagnetic field. Such tests involve the measurement of light
generated on the surface area of paramagnetic beads coated with bio-organic
material.

Our Beads
represent a significant product advance. Most paramagnetic beads are made of
iron oxide, and all are traditionally black or brown. We have developed a
proprietary process that coats the beads with a layer of silver, making them
white, and more sensitive to light. Our production process is also significantly
less expensive than those used by our competitors. We have internally tested our
Beads against all commercially available beads, and have found our silver-coated
Beads to be five times more sensitive than traditional black or brown magnetic
particles.
On
January 19, 2009 CardioGenics Inc., one of our Canadian subsidiaries, entered
into a Supply, Development & Distribution Agreement with Merck Chimie S.A.S.
(“Merck Chimie”) (the “Merck Agreement”), pursuant to which CardioGenics is
required to furnish Merck Chimie with certain quantities of CardioGenics’
proprietary silver-coated paramagnetic beads (the “CardioGenics Test Samples”),
which Merck Chimie is then required to encapsulate, on a test-basis, using Merck
Chimie’s proprietary encapsulation process. After Merck Chimie selects the best
encapsulation process, Merck Chimie agreed to then establish the manufacturing
parameters for the final encapsulated beads (the “Merck Encapsulated Beads”) and
thereafter scale-up production for commercial distribution of the Merck
Encapsulated Beads. Currently, Merck Chimie is still in the process of refining
its encapsulation of the CardioGenics Test Samples.
Pursuant
to the Merck Agreement, Merck Chimie has the exclusive right, for ten (10)
years, to distribute the Merck Encapsulated Beads on a worldwide basis, with
CardioGenics receiving 30% of the net sales proceeds of the Merck Encapsulated
Beads and Merck receiving 70% of such net sales proceeds. Merck is responsible
for manufacturing and distributing the Merck Encapsulated Beads.
On July
12, 2010 CardioGenics entered into a second agreement with Merck Chimie, which
is a non-exclusive supply agreement pursuant to which CardioGenics is adopting
its proprietary biological-linking technology to magnetic beads supplied by
Merck Chimie. The Merck beads enhanced by CardioGenics’ proprietary
biological-linking technology are designed to increase yields in antibody
manufacturing that can potentially reduce the cost of making certain
antibody-based drugs. The agreement is for a term of four (4) years and
CardioGenics will be paid an agreed upon fee for each gram of beads processed by
CardioGenics and shipped to Merck Chimie.
In
addition to the agreements with Merck Chimie, we have also entered into
Materials Transfer Agreements (“MTA”) with two other major international beads
distributors with respect to our proprietary magnetic beads. Under the first
MTA, CardioGenics will furnish the distributor with its silver-coated magnetic
beads for polymer coating by the distributor. In addition, the distributor will
provide CardioGenics with their magnetized bead prototypes, which CardioGenics
will then silver-coat with its proprietary silver-coating technology. Under the
second MTA, CardioGenics will furnish the distributor with its silver-coated
magnetic beads for polymer coating and subsequent testing and evaluation by the
distributor. Upon completion of the testing process, CardioGenics and the
distributors will evaluate the test results and determine whether to further
pursue commercialization of the resulting products.
Since the
CardioGenics business described above is now our primary business, we decided to
sell our Pixaya LLC subsidiary, and its related JAG Notes subscription and
Pixaya businesses, so we can focus our attention and resources exclusively on
our primary CardioGenics business. The sale of our Pixaya subsidiary was
completed on February 11, 2010.
OUR
STRATEGY
The
success of our business depends on our ability to obtain the requisite financing
and be able to:
|
|
●
|
complete
the development of our QLCA and our cardiovascular
tests;
|
|
|
●
|
obtain
FDA approval of our QLCA and the cardiovascular
tests;
|
|
|
●
|
develop
further tests that can be run on our
QLCA;
|
|
|
●
|
commercialize
our Beads.
|
We will
require additional funds in order to implement our full business strategy.
Accordingly, we will need to raise additional funds through public or private
financing, strategic relationships or other arrangements. We do not anticipate
generating any significant revenue until after our first cardiovascular test has
been approved by the FDA and our Beads are commercialized by Merck Chimie
pursuant to our agreement with them.
8
Since our
strength is product development and innovation, our strategy is focused on
exploiting this strength. In terms of product development and innovation, we
employ our internal resources to develop our products through the various phases
of development. We also rely on external service providers to supplement our
internal talents in product development.
We will
outsource product manufacturing. In terms of the QLCA, both the cartridge
assembly as well as the analyzer assembly will be contracted out to different
OEM providers with the facilities and expertise to deliver quality products. We
will maintain a quality control process to ensure that the products meet the
predetermined specifications.
Product
marketing and distribution will by achieved through partnerships with global
companies with wide reach. As we have done with our magnetic beads, the QLCA
will be marketed by a third party through licensing and distribution
agreements.
We are
also focusing on protecting our intellectual property and know how though
maintaining a patent filing process on a global basis as well as maintaining
confidentiality agreements with our staff, employees and service providers under
contractual agreements.
Although
we believe in these strategies, goals and targets, we cannot guarantee that we
will be successful in implementing them or that, even if implemented, they will
be effective in creating a profitable business. In addition, we are dependent on
having sufficient cash to carry out our strategies
Regulation
CardioGenics
Products
Our QL
Analyzer, Cartridge and Tests are classified as medical devices. Our beads are
reagents of medical testing equipment. Accordingly, they are subject to a number
of regulations in the jurisdictions where our products will be
sold.
United
States
The
testing, production and sale of IVD products are subject to regulation by
numerous state and federal government authorities, principally the
FDA.
Pursuant
to the U.S. Federal Food, Drug
and Cosmetic Act (“FD&C Act”), the FDA regulates the preclinical and
clinical testing, manufacture, labeling, distribution and promotion of medical
devices.
Medical
devices are classified into three categories, Class I, Class II or Class III.
The classification of a device is based on the level of control necessary to
assure the safety and effectiveness of the device. Generally, the complexity of
the submission and the approval times are based on the regulatory class of the
device. Device classification depends on the intended use and also the
indications for use of the device. Classification is also based on the risk the
device poses to the patient and/or the user. Class I devices include devices
with the lowest risk, and Class III devices are those with the greatest risk.
Class I devices are subject to general control, Class II devices are subject to
general controls and special controls, and Class III devices are subject to
general controls and must receive a Premarket Assessment or PMA by the
FDA.
Before
some Class I and most Class II devices can be introduced in the market, either
the manufacturer or distributor of the device is required to follow the
pre-market notification process described in section 510(k) of the FD&C Act.
A 510(k) is a pre-marketing submission made to the FDA to demonstrate that the
device to be marketed is as safe and effective, and is substantially equivalent
to a legally marketed device. Applicants must compare their 510(k) device to one
or more similar devices currently on the US market and support their claims for
substantial equivalency. The FDA requires a rigorous demonstration of
substantial equivalency. It generally takes three to six months from submission
to obtain 510(k) clearance. If any device cleared through 510(k) is modified or
enhanced, or if there is a change of use of the device, a new amended 510(k)
application must be submitted. According to FDA regulations and our management
team’s prior experiences with submissions of similar products, our QLCA and
launch product (TnI) will be classified as a Class II device and will be
subjected to the 510(K) process. Further, a second test product of ours (HFRS)
will also be subjected to the same 510(K) process. As for both tests, predicate
devices are commercially available. For other test products,
depending on the claims and with a prior agreement with the FDA, the submissions
would be either a PMA or 510(K). We have not yet approached the FDA for that
purpose.
9
Canada
Health
Canada sets out the requirements governing the sale, importation and
advertisement of medical devices. These regulations are intended to ensure that
medical devices distributed in Canada are both safe and effective. We are also
required to comply with certain procedures for the disposal of waste products
under the Canadian Code of Practice for the Management of Biological Waste (the
“Code”). We believe we are currently in compliance with all required Code
provisions.
Europe
Our
products will be subject to registration under the EU Medical Device Directives
for in-vitro diagnostic products.
Other
countries
Our
products will be subject to the regulations of any country where they are sold,
and we will make the necessary applications for approval on a country-by-country
basis.
Competition
CardioGenics
Competitors
Numerous
companies provide Point Of Care (POC) products, many with cardiovascular test
products. However, in terms of quantitative POC
products, few companies operate in this space with marketed devices. These
include:
|
·
|
Biosite
Diagnostics Incorporated;
|
|
·
|
Response
Biomedicals Corp.;
|
|
·
|
Roche
POC division; and
|
|
·
|
i-Stat
division of Abbott Diagnostics
|
The first
3 companies employ fluorescence measurements in their platforms, while i-Stat
employs electrochemical testing. We believe that our technology and products in
development will offer superior products to the POC market. None of the above
companies offer chemiluminescence in its platform, a technology that is
well-recognized for its superiority as evidenced by its dominance in the
laboratory testing market. We believe that harnessing chemiluminescence in our
QLCA will fulfill the clinical demands for fast and accurate quantitative
results at patient bedsides.
Research
and Development
Our
efforts are focused on the development of our QLCA and our cardiovascular tests
and the commercialization of our beads. Over the years 2010 and 2009 we incurred
expenses of $692,000 and $1,572,000, respectively, on those
efforts.
Website
Technical Information
Our
CardioGenics website (www.cardiogenics.com) is maintained by us internally and
is hosted by DreamHost, which has hosting facilities located in Brea,
California.
Employees
As of
October 31, 2010, we had ten (10) employees. Of those employees, only Yahia
Gawad, our Chief Executive Officer, has an employment agreement with the
Company.
Acquisition
of CardioGenics
On July
31, 2009 we completed the acquisition of CardioGenics by CardioGenics ExchangeCo
Inc. (“ExchangeCo”), our
Ontario, Canada subsidiary, pursuant to the terms of a Share Purchase Agreement
dated May 22, 2009 among ExchangeCo, JAG Media Holdings, Inc., CardioGenics and
CardioGenics’ principal stockholder, Yahia Gawad (the “Share Purchase Agreement”).
CardioGenics is considered the acquirer in the transaction for accounting and
financial reporting purposes.
10
In
connection with the acquisition, ExchangeCo acquired all of the outstanding
common shares of CardioGenics (the “CardioGenics Common Shares”),
excluding 173,869 CardioGenics Common Shares in the aggregate owned by two (2)
minority stockholders of CardioGenics (the “Dissenting Stockholders”), in
consideration for the issuance of 422,183,610 shares of our common stock to the
CardioGenics stockholders at the closing, as further described below (the “Share Consideration”). In
consideration for the surrender of their CardioGenics Common Shares, the
CardioGenics stockholders had the option to receive at the closing their
pro-rata allocation of the Share Consideration in the form of (a) our common
shares or (b) “Exchangeable Shares“ of ExchangeCo, which are exchangeable into
our common shares in accordance with the terms of a Voting and Exchange Trust
Agreement dated July 6, 2009 among JAG Media, ExchangeCo, and WeirFoulds LLP, as
trustee and the rights and preferences of the Exchangeable Shares. Those
CardioGenics stockholders who elected to receive directly our common shares were
issued, in the aggregate, 145,528,195 common shares at the closing and those
CardioGenics stockholders who elected to receive Exchangeable Shares were issued
16 Exchangeable Shares at the closing, which are exchangeable at any time into
276,655,415 of our common shares, in the aggregate. The Share Consideration
issued at the closing provided the CardioGenics stockholders with direct and
indirect ownership of approximately 85% of our outstanding common stock, on a
fully diluted basis.
Immediately
prior to the closing, all CardioGenics debenture holders converted their
debentures into CardioGenics Common Shares in accordance with the terms of their
respective debentures, as required by the terms of the Share Purchase Agreement.
Accordingly, such former debenture holders became CardioGenics stockholders for
purposes of the acquisition and received their pro-rata allotment of the Share
Consideration in the form of JAG Common Shares and/or Exchangeable Shares at the
closing in consideration for the surrender of the CardioGenics Common Shares
they received upon conversion of their debentures.
Also
prior to the closing, CardioGenics closed on an equity investment round of
financing totaling $2,715,000. These equity investors in CardioGenics became
CardioGenics stockholders for purposes of the acquisition and received their
pro-rata allotment of the Share Consideration in the form of our common
shares.
All of
our common shares received by CardioGenics stockholders in exchange for their
CardioGenics Common Shares may not be registered for resale and, therefore,
shall remain subject to the rights and restrictions of Rule 144. All
Exchangeable Shares received by CardioGenics stockholders in exchange for their
CardioGenics Common Shares (and any of our common shares into which such
Exchangeable Shares may be exchanged) also may not be registered for resale
prior to six (6) months following the closing and, therefore shall remain
subject to the rights and restrictions of Rule 144 prior to any such
registration.
Also at
the closing, all holders of CardioGenics warrants entitling the holders to
purchase CardioGenics Common Shares at various prices exchanged their
CardioGenics warrants for warrants to purchase, in the aggregate, 36,148,896 of
our common shares at exercise prices of $0.047 per share, in accordance with the
terms of the Share Purchase Agreement and the respective warrants. The terms of
these newly issued warrants did not include any registration rights for the
warrant holders. CardioGenics had no options to acquire CardioGenics Common
Shares outstanding as of the closing.
At the
closing, our then current directors resigned as directors of JAG Media and its
subsidiaries after appointing their successors and our then current officers
also resigned as officers and executives of JAG Media and its subsidiaries.
After their resignation and the closing, our former directors entered into
consulting agreements with the Company pursuant to which they are rendering
various services to assist us in connection with certain transition
matters. Each
consulting agreement is for a term of 18 months, with each party receiving
500,000 shares of the Company’s common stock, issued pursuant to our 1999
Long-Term Incentive Plan, as compensation for their services under the
consulting agreements.
Following
the closing, a majority of our stockholders approved, by written consent, an
amendment to our articles of incorporation, which provided for (a) a change in
our corporate name from “JAG Media Holdings, Inc.“ to “CardioGenics Holdings
Inc.” and (b) an increase in the number of our authorized JAG Common Shares from
500,000,000 to 650,000,000.
Financing
Arrangements
Equity
Line of Credit with YA Global
In
connection with the CardioGenics Acquisition, on March 12, 2009 we entered into
a Standby Equity Distribution Agreement with YA Global Master SPV Ltd. (“YA Ltd
”) (the “ SEDA ”) pursuant to which YA Ltd agreed to purchase up to $5,000,000
of our common stock (the “ Commitment Amount ”) over the course of the
thirty-six (36) months following the date the registration statement for the
shares to be issued pursuant to the SEDA is first declared effective (the “
Commitment Period ”). We will have the right, but not the obligation, to sell
common stock to YA Ltd during the Commitment Period. Concurrent with the
execution of the SEDA, we also entered into a Registration Rights Agreement with
YA Ltd pursuant to which we agreed to register the shares of our common stock to
be issued in connection with the SEDA. Such registration was never filed and,
accordingly, the SEDA never became effective. Effective March 19, 2010, the
Company and YA Ltd. terminated these agreements by mutual consent.
11
Increase
in Authorized Shares
In
October 2009 a majority of our stockholders approved, by written consent, an
amendment to our articles of incorporation, which provided for, among other
matters, an increase in the number of our authorized shares of common stock from 500,000,000 to
650,000,000.
Reverse
Stock Split
As
authorized by our Board of Directors, on June 18, 2010, we filed a “Certificate
of Change” with the Nevada Secretary of State’s Office, which effected a 1:10
share consolidation of our outstanding and authorized shares of common stock. As
a result of this share consolidation our authorized shares of common stock was
reduced from 650,000,000 to 65,000,000 and our outstanding shares of common
stock as of such date were consolidated in accordance with the 1:10 share
consolidation ratio.
Facilities
See “Item
2.—Properties.”
Legal
Proceedings
See “Item
3.—Legal Proceedings.”
Where
You Can Find More Information About Us
We are
required to file annual, quarterly and current reports, proxy statements and
other information with the SEC. You can read and copy any of this
information at the SEC’s Public Reference Room at 100 F Street, N.E.,
Washington, D.C. 20549 on official business days during the hours of 10:00 a.m.
to 3:00 p.m. You may obtain information on the operation of the
Public Reference Room by calling the SEC at 1-800-SEC-0330. This
information is also available from the SEC’s website at http://www.sec.gov. We
will also gladly send any filing to you upon your written request to Dr. Yahia
Gawad, our Chief Executive Officer, at 6295 Northam Drive, Unit 8, Mississauga,
Ontario L4V 1W8.
ITEM
1A. RISK FACTORS
Risks Related to Our
CardioGenics Business and Industry
The
global financial crisis has had, and may continue to have, an impact on our
business and financial condition.
The
ongoing global financial crisis may limit our ability to access the capital
markets at a time when we would like, or need, to raise capital, which could
have an impact on our ability to react to changing economic and business
conditions. Accordingly, if the global financial crisis and current economic
downturn continue or worsen, our business, results of operations and financial
condition could be materially and adversely affected.
The
requirements of being a public company may strain our resources and distract our
management
As a
public company, we are subject to the reporting requirements of the Securities
Exchange Act of 1934, as amended and the Sarbanes-Oxley Act of 2002 (the
“Sarbanes-Oxley Act”). These requirements place a strain on our systems and
resources. The Exchange Act requires that we file annual, quarterly
and current reports with respect to our business and financial condition. The
Sarbanes-Oxley Act requires that we maintain effective disclosure controls and
procedures and internal controls for financial reporting. Management
has identified the following material weaknesses in our internal controls over
financial reporting: 1. lack of documented policies and procedures; 2. lack of
resources to account for complex and unusual transactions; and, 3. there is no
effective segregation of duties, which includes monitoring controls, between the
members of management.
We are
also required to document and test our internal control procedures in order to
satisfy the requirements of Section 404 of the Sarbanes-Oxley Act, which
requires annual management assessments of the effectiveness of our internal
controls over financial reporting. We may not be able to remediate these
weaknesses in time to meet the deadlines imposed by the Sarbanes-Oxley Act. If
we fail to achieve and maintain the adequacy of our internal
controls, as such standards are modified, supplemented or amended
from time to time, we may not be able to ensure that we can conclude on an
ongoing basis that we have effective internal controls over financial reporting
in accordance with the Sarbanes-Oxley Act.
12
In order
to maintain and improve the effectiveness of our disclosure controls and
procedures and internal control over financial reporting, significant resources
and management oversight will be required. This may divert management’s
attention from other business concerns, which could have a material adverse
effect on our business, financial condition, results of operations and cash
flows. In addition, we may need to hire additional accounting and financial
staff with appropriate public company experience and technical accounting
knowledge, and we cannot assure you that we will be able to do so in a timely
fashion.
We
have not earned any revenues in our CardioGenics business unit since its
incorporation and only have a limited operating history in its current business,
which raise doubt about our ability to continue as a going concern.
Our
CardioGenics business unit has a limited operating history in its current
business and must be considered in the development stage. It has not generated
any revenues since its inception and we will, in all likelihood, continue to
incur operating expenses without significant revenues until we complete
development of our Cardiovascular Tests and commercialize our QLCA and the
Cardiovascular Tests. The primary source of funds for our CardioGenics business
unit has been the sale of common stock. We cannot assure that we will be able to
generate any significant revenues or income. These circumstances make us
dependent on additional financial support until profitability is achieved. There
is no assurance that we will ever be profitable and we have not yet achieved
profitable operations. These factors raise substantial doubt that we will be
able to continue as a going concern.
We
need to raise additional financing to support the research and development of
our CardioGenics business but we cannot be sure that we will be able to obtain
additional financing on terms favorable to us when needed. If we are unable to
obtain additional financing to meet our needs, our operations may be adversely
affected or terminated.
Our
ability to develop new test products for our QLCA is dependent upon our ability
to raise significant additional financing when needed. If we are unable to
obtain such financing, we will not be able to fully develop and commercialize
our platform and technology. Our future capital requirements will depend upon
many factors, including:
|
•
|
continued
scientific progress in our research and development
programs;
|
|
•
|
costs
and timing of conducting clinical trials and seeking regulatory approvals
and patent prosecutions;
|
|
•
|
competing
technological and market
developments;
|
|
•
|
our
ability to establish additional collaborative relationships;
and
|
|
•
|
the
effect of commercialization activities and facility expansions if and as
required.
|
We have
limited financial resources and to date, no cash flow from the operations of our
CardioGenics business unit and we are dependent for funds on our ability to sell
our common stock, primarily on a private placement basis. There can be no
assurance that we will be able to obtain financing on that basis in light of
factors such as the market demand for our securities, the state of financial
markets generally and other relevant factors. Any sale of our common stock in
the future will result in dilution to existing stockholders. Furthermore, there
is no assurance that we will not incur debt in the future, that we will have
sufficient funds to repay any future indebtedness or that we will not default on
our future debts, jeopardizing our business viability. Finally, we may not be
able to borrow or raise additional capital in the future to meet our needs or to
otherwise provide the capital necessary to continue the development of our
technology, which might result in the loss of some or all of your investment in
our common stock.
We
may acquire other businesses, license rights to technologies or products, form
alliances, or dispose of or spin-off businesses, which could cause us to incur
significant expenses and could negatively affect profitability.
We
may pursue acquisitions, technology licensing arrangements, and strategic
alliances, or dispose of or spin-off some of our businesses, as part of our
business strategy. We may not complete these transactions in a timely manner, on
a cost-effective basis, or at all, and may not realize the expected benefits. If
we are successful in making an acquisition, the products and technologies that
are acquired may not be successful or may require significantly greater
resources and investments than originally anticipated. We may not be able to
integrate acquisitions successfully into our existing business and could incur
or assume significant debt and unknown or contingent liabilities. We could also
experience negative effects on our reported results of operations from
acquisition or disposition-related charges, amortization of expenses related to
intangibles and charges for impairment of long-term assets.
13
The
expiration or loss of patent protection and licenses may affect our future
revenues and operating income.
Much of
our business relies on patent and trademark and other intellectual property
protection. Although most of the challenges to our intellectual property would
likely come from other businesses, governments may also challenge intellectual
property protections. To the extent our intellectual property is successfully
challenged, invalidated, or circumvented or to the extent it does not allow us
to compete effectively, our business will suffer. To the extent that countries
do not enforce our intellectual property rights or to the extent that countries
require compulsory licensing of our intellectual property, our future revenues
and operating income will be reduced. Our principal patents and trademarks are
described in greater detail in the sections captioned, "Patents, Trademarks, and
Licenses."
Competitors'
intellectual property may prevent us from selling our products or have a
material adverse effect on our future profitability and financial
condition.
Competitors
may claim that one or more of our products infringe upon their intellectual
property. Resolving an intellectual property infringement claim can be costly
and time consuming and may require us to enter into license agreements. We
cannot guarantee that we would be able to obtain license agreements on
commercially reasonable terms. A successful claim of patent or other
intellectual property infringement could subject us to significant damages or an
injunction preventing the manufacture, sale or use of our affected products. Any
of these events could have a material adverse effect on our profitability and
financial condition.
We
may not be able to adequately protect our intellectual property
We
believe the patents, trade secrets and other intellectual property we use are
important to our business, and any unauthorized use of such intellectual
property by third parties may adversely affect our business and reputation. We
rely on the intellectual property laws and contractual arrangements with our
employees, business partners and others to protect such intellectual property
rights. Filing, prosecuting, defending and enforcing patents on all of our
technologies and products throughout the world would be prohibitively expensive.
Competitors may, without our authorization, use our intellectual property to
develop their own competing technologies and products in jurisdictions where we
have not obtained patent protection. These technologies and products may not be
covered by any of our patent claims or other intellectual property rights.
Furthermore, the validity, enforceability and scope of protection of
intellectual property in some countries where we may conduct business is
uncertain and still evolving, and these laws may not protect intellectual
property rights to the same extent as the laws of the United
States.
Many
companies have encountered significant problems in protecting and defending
their intellectual property rights in foreign jurisdictions. Many countries,
including certain countries in Europe, have compulsory licensing laws under
which a patent owner may be compelled to grant licenses to third parties (for
example, the patent owner has failed to “work” the invention in that country or
the third party has patented improvements). In addition, many countries limit
the enforceability of patents against government agencies or government
contractors. In these countries, the patent owner may have limited remedies,
which could materially diminish the value of the patent. Moreover, litigation
involving patent or other intellectual property matters in the United States or
in foreign countries may be necessary in the future to enforce our intellectual
property rights, which could result in substantial costs and diversion of our
resources, and have a material adverse effect on our business, financial
condition and results of operations.
We
are subject to numerous governmental regulations and it can be costly to comply
with these regulations and to develop compliant products and
processes.
Our
products are subject to regulation by the U.S. Food and Drug Administration
(“FDA”), and numerous international, federal, and state authorities. The process
of obtaining regulatory approvals to market a medical device can be costly and
time-consuming, and approvals might not be granted for future products, or
additional uses of existing products, on a timely basis, if at all. Delays in
the receipt of, or failure to obtain approvals for, future products, or
additional uses of existing products, could result in delayed realization of
product revenues, reduction in revenues, and in substantial additional costs. In
particular, in the United States our products are regulated under the 1976
Medical Device Amendments to the Food, Drug and Cosmetic Act, which is
administered by the FDA. We believe that the FDA will classify our products as
“Class II” devices, thus requiring us to submit to the FDA a pre-market
notification form or 510(k). The FDA uses the 510(k) to substantiate product
claims that are made by medical device manufacturers prior to marketing. In our
510(k) notification, we must, among other things, establish that the product we
plan to market is “substantially equivalent” to (1) a product that was on
the market prior to the adoption of the 1976 Medical Device Amendment or
(2) a product that the FDA has previously cleared.
14
The FDA
review process of a 510(k) notification can last anywhere from three to six
months, and the FDA must issue a written order finding “substantial equivalence”
before a company can market a medical device. We are currently developing a
group of cardiovascular tests that we will have to clear with the FDA through
the 510(k) notification procedures. These test products are crucial for our
success and if we do not receive 510(k) clearance for a particular product, we
will not be able to market these products in the United States, which will have
a material adverse effect on our revenues, profitability and financial
condition.
In
addition, no assurance can be given that we will remain in compliance with
applicable FDA and other regulatory requirements once clearance or approval has
been obtained for a product. We must incur expense and spend time and effort to
ensure compliance with these complex regulations. Possible regulatory actions
could include warning letters, fines, damages, injunctions, civil penalties,
recalls, seizures of our products and criminal prosecution. These actions could
result in, among other things: substantial modifications to our business
practices and operations; refunds, recalls, or seizures of our products; a total
or partial shutdown of production while we or our suppliers remedy the alleged
violation; the inability to obtain future pre-market clearances or approvals;
and, withdrawals or suspensions of current products from the market. Any of
these events could disrupt our business and have a material adverse effect on
our revenues, profitability and financial condition.
Changes
in third-party payor reimbursement regulations can negatively affect our
business.
By
regulating the maximum amount of reimbursement they will provide for blood
testing services, third-party payors, such as HMOs, pay-per-service insurance
plans, Medicare and Medicaid, can indirectly affect the pricing or the relative
attractiveness of our diagnostic products. For example, the Centers for Medicare
and Medicaid Services set the level of reimbursement of fees for blood testing
services for Medicare beneficiaries. If third-party payors decrease the
reimbursement amounts for blood testing services, it may decrease the amount
that physicians and hospitals are able to charge patients for such services.
Consequently, we would either need to charge less for our products or incur a
reduction in our profit margins. If the government and third-party payors do not
provide for adequate coverage and reimbursement levels to allow health care
providers to use our products, the demand for our products will
decrease.
Laws
and regulations affecting government benefit programs could impose new
obligations on us, require us to change our business practices, and restrict our
operations in the future.
Our
industry is also subject to various federal, state, and international laws and
regulations pertaining to government benefit program reimbursement, price
reporting and regulation, and health care fraud and abuse, including
anti-kickback and false claims laws, the Medicaid Rebate Statute, the Veterans
Health Care Act, and individual state laws relating to pricing and sales and
marketing practices. Violations of these laws may be punishable by criminal
and/or civil sanctions, including, in some instances, substantial fines,
imprisonment, and exclusion from participation in federal and state health care
programs, including Medicare, Medicaid, and Veterans Administration health
programs. These laws and regulations are broad in scope and they are subject to
evolving interpretations, which could require us to incur substantial costs
associated with compliance or to alter one or more of our sales or marketing
practices. In addition, violations of these laws, or allegations of such
violations, could disrupt our business and result in a material adverse effect
on our revenues, profitability, and financial condition.
Our
research and development efforts may not succeed in developing commercially
successful products and technologies, which may cause our revenue and
profitability to decline.
To remain
competitive, we must continue to launch new products and technologies. To
accomplish this, we must commit substantial efforts, funds, and other resources
to research and development. A high rate of failure is inherent in the research
and development of new products and technologies. We must make ongoing
substantial expenditures without any assurance that its efforts will be
commercially successful. Failure can occur at any point in the process,
including after significant funds have been invested.
15
Promising
new product candidates may fail to reach the market or may only have limited
commercial success because of efficacy or safety concerns, failure to achieve
positive clinical outcomes, inability to obtain necessary regulatory approvals,
limited scope of approved uses, excessive costs to manufacture, the failure to
establish or maintain intellectual property rights, or infringement of the
intellectual property rights of others. Even if we successfully develop new
products or enhancements or new generations of our existing products, they may
be quickly rendered obsolete by changing customer preferences, changing industry
standards, or competitors' innovations. Innovations may not be accepted quickly
in the marketplace because of, among other things, entrenched patterns of
clinical practice or uncertainty over third-party reimbursement. We cannot state
with certainty when or whether any of our products under development will be
launched or whether any products will be commercially successful. Failure to
launch successful new products or new uses for existing products may cause our
products to become obsolete, causing our revenues and operating results to
suffer.
New
products and technological advances by our competitors may negatively affect our
results of operations.
Our
products face intense competition from our competitors' products. Competitors'
products may be safer, more effective, more effectively marketed or sold, or
have lower prices or superior performance features than our products. We cannot
predict with certainty the timing or impact of the introduction of competitors'
products.
We
depend on key members of our management and scientific staff and, if we fail to
retain and recruit qualified individuals, our ability to execute our business
strategy and generate sales would be harmed.
We are
highly dependent on the principal members of our management and scientific
staff. The loss of any of these key personnel, including in particular Dr. Yahia
Gawad, our Chief Executive Officer, might impede the achievement of our business
objectives. We may not be able to continue to attract and retain skilled and
experienced scientific, marketing and manufacturing personnel on acceptable
terms in the future because numerous medical products and other high technology
companies compete for the services of these qualified individuals. We currently
do not maintain key man life insurance on any of our employees.
The
manufacture of many of our products is a highly exacting and complex process,
and if we or one of our suppliers encounter problems manufacturing products, our
business could suffer.
The
manufacture of many of our products is a highly exacting and complex process,
due in part to strict regulatory requirements. Problems may arise during
manufacturing for a variety of reasons, including equipment malfunction, failure
to follow specific protocols and procedures, problems with raw materials,
natural disasters, and environmental factors. In addition, we may use single
suppliers for certain products and materials. If problems arise during the
production of a batch of product, that batch of product may have to be
discarded. This could, among other things, lead to increased costs, lost
revenue, damage to customer relations, time and expense spent investigating the
cause and, depending on the cause, similar losses with respect to other batches
or products. If problems are not discovered before the product is released to
the market, recall and product liability costs may also be incurred. To the
extent we or one of our suppliers experience significant manufacturing problems,
this could have a material adverse effect on our revenues and
profitability.
Significant
safety issues could arise for our products, which could have a material adverse
effect on our revenues and financial condition.
All
medical devices receive regulatory approval based on data obtained in controlled
testing environments of limited duration. Following regulatory approval, these
products will be used over longer periods of time with many patients. If new
safety issues arise, we may be required to change the conditions of use for a
product. For example, we may be required to provide additional warnings on a
product's label or narrow its approved use, either of which could reduce the
product's market acceptance. If serious safety issues with one of our products
arise, sales of the product could be halted by us or by regulatory authorities.
Safety issues affecting suppliers' or competitors' products also may reduce the
market acceptance of our products.
In
addition, in the ordinary course of business, we may be the subject of product
liability claims and lawsuits alleging that our products or the products of
other companies that we promote, or may be incorporated in our products, have
resulted or could result in an unsafe condition for or injury to patients.
Product liability claims and lawsuits and safety alerts or product recalls,
regardless of their ultimate outcome, may have a material adverse effect on our
business, reputation and financial condition, as well as on our ability to
attract and retain customers. Product liability losses are
self-insured.
16
The
international nature of our business subjects us to additional business risks
that may cause our revenue and profitability to decline.
Since we
intend to market our products internationally, our business will be subject to
risks associated with doing business internationally. The risks associated with
any such operations outside the United States include:
|
•
|
changes
in foreign medical reimbursement policies and
programs;
|
|
•
|
multiple
foreign regulatory requirements that are subject to change and that could
restrict our ability to manufacture, market, and sell our
products;
|
|
•
|
differing
local product preferences and product
requirements;
|
|
•
|
trade
protection measures and import or export licensing
requirements;
|
|
•
|
difficulty
in establishing, staffing, and managing foreign
operations;
|
|
•
|
differing
labor regulations;
|
|
•
|
potentially
negative consequences from changes in or interpretations of tax
laws;
|
|
•
|
political
and economic instability;
|
|
•
|
inflation,
recession and fluctuations in foreign currency exchange and interest
rates; and,
|
|
•
|
compulsory
licensing or diminished protection of intellectual
property.
|
These
risks may, individually or in the aggregate, have a material adverse effect on
our revenues and profitability.
Other
factors can have a material adverse effect on our future profitability and
financial condition.
Many
other factors can affect our profitability and financial condition,
including:
|
•
|
Changes
in or interpretations of laws and regulations including changes in
accounting standards, taxation requirements and environmental laws in
domestic or foreign jurisdictions.
|
|
•
|
Changes
in the rate of inflation (including the cost of raw materials,
commodities, and supplies), interest rates and the performance of
investments held by us.
|
|
•
|
Changes
in the creditworthiness of counterparties that transact business with or
provide services to our distributors or
us.
|
|
•
|
Changes
in business, economic, and political conditions, including: war, political
instability, terrorist attacks in the U.S. and other parts of the world,
the threat of future terrorist activity in the U.S. and other parts of the
world and related military action; natural disasters; the cost and
availability of insurance due to any of the foregoing events; labor
disputes, strikes, slow-downs, or other forms of labor or union activity;
and, pressure from third-party interest
groups.
|
|
•
|
Changes
in our business units and investments and changes in the relative and
absolute contribution of each to earnings and cash flow resulting from
evolving business strategies, changing product mix, changes in tax rates
both in the U.S. and abroad and opportunities existing now or in the
future.
|
|
•
|
Changes
in the buying patterns of a major distributor, retailer, or wholesale
customer resulting from buyer purchasing decisions, pricing, seasonality,
or other factors, or other problems with licensors, suppliers,
distributors, and business
partners.
|
|
•
|
Difficulties
related to our information technology systems, any of which could
adversely affect business operations, including any significant breakdown,
invasion, destruction, or interruption of these
systems.
|
|
•
|
Changes
in credit markets impacting our ability to obtain financing for our
business operations.
|
|
•
|
Legal
difficulties, any of which could preclude or delay commercialization of
products or adversely affect profitability, including claims asserting
statutory or regulatory violations, adverse litigation decisions, and
issues regarding compliance with any governmental consent
decree.
|
17
Risks Related to Our Capital
Structure
Our shareholders may experience
significant dilution from the exercise of warrants to purchase shares of our
common stock.
As a
result of our acquisition of CardioGenics, former CardioGenics warrant holders
exchanged their warrants to purchase CardioGenics Common Shares for warrants to
purchase our Common Shares. Currently, the warrants held by such former
CardioGenics warrant holders entitle them to purchase up to 3,826,974 of our
Common Shares at prices of $0.47 per share.
Accordingly,
you may experience substantial dilution upon exercise of these warrants. In
addition, you may experience substantial dilution if the price of our Common
Shares increases to a level greater than the exercise price of these
warrants.
Future
Issuance of Our Common Stock Could Dilute Current Stockholder or Adversely
Affect the Market.
Future
issuances of our common stock could be at values substantially below the price
paid by the current holders of our common stock. In addition, common stock could
be issued to fend off unwanted tender offers or hostile takeovers without
further stockholder approval. Sales of substantial amounts of our common stock
in the public market, or even just the prospect of such sales, could depress the
prevailing market price of our common stock and our ability to raise equity
capital in the future.
The market for our common stock is
limited.
Our
common stock is traded on the OTC Bulletin Board. Trading activity in our stock
has fluctuated and at times been limited. We cannot guarantee that a
consistently active trading market for our stock will continue, especially while
we remain on the OTC Bulletin Board.
Because our common stock currently
trades below $5.00 per share and is quoted on the OTCBB, our common stock is
considered by the SEC to be a “penny stock,” which adversely affects our
liquidity.
Our
common stock does not currently qualify for listing on any national securities
exchange, and we do not anticipate that it will qualify for such a listing in
the short-term future. If our common stock continues to be quoted on the OTC
Bulletin Board or is traded on the Pink Sheets, and if the trading price of our
common stock remains less than $5.00 per share, our common stock is considered a
“penny stock,” and trading in our common stock is subject to the requirements of
Rule 15g-9 under the Exchange Act. Under this rule, brokers or dealers who
recommend low-priced securities to persons other than established customers and
accredited investors must satisfy special sales practice requirements. The
broker or dealer must make an individualized written suitability determination
for the purchaser and receive the purchaser’s written consent prior to the
transaction. SEC regulations also require additional disclosure in connection
with any trades involving a penny stock, including the delivery, prior to any
penny stock transaction, of a disclosure schedule explaining the penny stock
market and its associated risks. These requirements could severely limit the
liquidity of such securities in the secondary market because few brokers or
dealers are likely to undertake these compliance activities. In addition to the
applicability of the penny stock rules, another risk associated with trading in
penny stocks may be large price fluctuations.
Our amended charter contains
provisions that may discourage an unaffiliated party to take us
over.
Without
further stockholder action, our Board of Directors could authorize the issuance
of additional shares of our common stock as well as preferred stock with special
voting rights by class or with more than one vote per share, to a “white knight”
in order to deter a potential buyer. This might have the effect of
preventing or discouraging an attempt by a party unable to obtain the approval
of our Board of Directors to take over or otherwise gain control of
us.
Terms of subsequent financings may
adversely impact your investment.
We may
have to raise equity, debt or preferred stock financing in the future. Your
rights and the value of your investment in our Common Shares could be reduced.
For example, if we issue secured debt securities, the holders of the debt would
have a claim against our assets that would be prior to the rights of
stockholders until the debt is paid. Interest on these debt securities would
increase costs and negatively impact operating results.
18
Preferred
stock could be issued in series from time to time with such designations,
rights, preferences, and limitations as needed to raise capital. The terms of
preferred stock could be more advantageous to those investors than to the
holders of our Common Shares.
Our
articles of incorporation do not provide stockholders the pre-emptive right to
buy shares from the company. As a result, you will not have the automatic
ability to avoid dilution in your percentage ownership of the
company.
Control
of our stock is now held by the former CardioGenics shareholders.
The prior
shareholders of CardioGenics, as of the closing of the CardioGenics Acquisition,
owned, directly or indirectly, approximately 85% of our outstanding common
stock. While their percentage would decline if and to the extent new shares of
our common stock are issued, you should expect these persons to exert continuing
influence over all matters requiring shareholder approval, including the
election of directors. You may have little to no practical control over such
matters.
It
is not likely that we will pay dividends on the common stock or any other class
of stock
We intend
to retain any future earnings for the operation and expansion of our business.
We do not anticipate paying cash dividends on our common stock, or any other
class of stock, in the foreseeable future. Stockholders should look solely to
appreciation in the market price of our Common Shares to obtain a return on
investment.
Our stockholders ownership of our
common stock may be in doubt due to possible naked short selling of our common
stock.
We
believe, but cannot confirm, that speculators may have engaged in a practice
commonly known as a “naked short” sale of our common stock, which means that
certain brokers may be permitting their short selling customers to sell shares
of our common stock that their customers do not own and may have failed to
borrow and therefore deliver the shares sold to the purchaser of the shares. We
have from time to time been included by NASDAQ on the Regulation SHO Threshold
Security List, which is indicative of a significant amount of naked shorting in
the stock. Because naked shorting may result in an artificial depression of our
stock price, our stockholders could lose all or part of their investment in our
common stock. As a result of this naked short selling, there may be a
substantial number of purchasers who believe they are our stockholders, but who
in fact would not be stockholders since their brokers may never have received
any shares of our common stock for their account. In addition, investors who
believe they are our stockholders may not have received a stock dividend to
which they are entitled or may have been deprived of the right to vote some or
all of their shares.
ITEM
1B. UNRESOLVED STAFF COMMENTS
Not
Applicable.
ITEM
2. PROPERTIES
Our
executive and administrative headquarters are currently located at 6295 Northam
Drive, Units 7 & 8, Mississauga, Ontario L4V 1W8 Canada. We rent this space
at a cost of CDN$6,432 per month.
The
servers for our websites are housed at separate locations as described
above. See “Item
1.—Business—Website Technical Information.” We believe that
our facilities are adequate for our current needs and that, if our lease is not
renewed on commercially reasonable terms, we will be able to locate suitable new
office space and obtain a suitable replacement for our executive and
administrative headquarters.
ITEM
3. LEGAL PROCEEDINGS
On April
22, 2009, CardioGenics was served with a statement of claim in the Province
of Ontario, Canada, from a prior contractor claiming compensation for wrongful
dismissal and ancillary causes of action including payment of monies in
realization of his investment in CardioGenics, with an aggregate claim of
$514,000. The Company considers all the claims to be without any
merit, has already delivered a statement of defence and intends
to vigorously defend the action. If the matter eventually proceeds to
trial, the Company does not expect to be found liable on any ground or for any
cause of action.
19
On
January 14, 2010, Flow Capital Advisors, Inc. (“Flow Capital”) filed a
lawsuit against JAG Media Holdings Inc. in the Circuit Court of the 17th
Judicial Circuit In and For Broward County Florida (Case No.
10001713). Pursuant to this lawsuit, Flow Capital alleges that JAG
Media Holdings breached a Non-Circumvention Agreement it had entered into with
Flow Capital, dated January 1, 2004. JAG Media Holdings has moved to dismiss the
case because Flow Capital is not registered to transact business in the state of
Florida and is therefore barred from maintaining the suit under applicable law.
The motion is pending although Flow Capital has since registered, and if the
motion is denied JAG Media Holdings expects to file an answer asserting various
defenses and vigorously opposing the suit.
On
January 15, 2010 Flow Capital filed a lawsuit against CardioGenics Inc., and
another defendant in the United States District Court for the Southern District
of Florida, Fort Lauderdale Division (Case No. 10-CV-60066-Martinez-Brown). This
lawsuit alleges that CardioGenics (i) breached a Finder’s Fee Agreement in
connection with the CardioGenics Acquisition; and (ii) breached a
non-circumvention agreement. Flow Capital is claiming that it is entitled to the
finder’s fee equal to eight percent (8%) of the JAG Media Holdings shares
received by CardioGenics, or the equivalent monetary value of the stock.
CardioGenics has moved to dismiss the lawsuit for lack of jurisdiction against
it in Florida, and that motion is pending.
On
October 26, 2010 Karver International Inc. filed a lawsuit in the 11th
Judicial Circuit in and for Miami-Dade County, Florida against CardioGenics
Holdings Inc. and several other defendants including affiliates, officers and
directors of CardioGenics Holdings, Inc. The Plaintiff generally
alleges that the named defendants made certain alleged misrepresentations in
connection with the purchase of shares of CardioGenics Holdings
Inc. On December 20, 2010 CardioGenics Holdings Inc. and other
defendants filed a motion to dismiss on the basis that the court lacks personal
jurisdiction over most defendants, that an enforceable forum selection clause
requires that the action be litigated in Ontario, Canada that the doctrine of
forum non conveniens
requires dismissal in favor of the Ontario forum, and that the complaint suffers
from numerous other technical deficiencies warranting dismissal (e.g., failure
to attach documents to the Complaint, failure to plead fraud with particularity,
etc.). The motion is currently pending. Should the motion
be denied, CardioGenics Holdings, Inc. will continue to pursue vigorous defense
to this action.
ITEM
4. (REMOVED AND RESERVED)
PART
II
ITEM
5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS
AND ISSUER PURCHASES OF SECURITIES
For the
period covered below, our common stock (other than our class B common stock) is
traded on the OTC Bulletin Board under the symbol CGNH. In October 2009, our
symbol was changed from JAGH to CGNH as a result of the CardioGenics
Acquisition. The following table based on Bloomberg L.P. reflects quarterly high
and low bid prices of our common stock from January 31, 2009 through October 31,
2010. Such prices are inter-dealer quotations without retail
mark-ups, mark-downs or commissions, and may not represent actual
transactions.
|
Fiscal Year Ending October 31,
2010
|
||||
|
Quarter
Ended
|
High
$
|
Low
$
|
||
|
October
31, 2010
|
2.27
|
1.47
|
||
|
July
31, 2010
|
2.13
|
1.19
|
||
|
April
30, 2010
|
2.93
|
0.697
|
||
|
January
31, 2010
|
0.93
|
0.52
|
||
|
Fiscal Year Ending October 31,
2009
|
||||
|
Quarter
Ended
|
High
$
|
Low
$
|
||
|
October
31, 2009
|
0.78
|
0.20
|
||
|
July
31, 2009
|
0.38
|
0.37
|
||
|
April
30, 2009
|
0.17
|
0.14
|
||
|
January
31, 2009
|
0.23
|
0.18
|
20
On
January 10, 2011, the closing bid price for our common stock was
$1.84. A public trading market for our Series 2 and Series 3 Class B
common stock has never developed.
As of
January 10, 2011, there were 29,320,647 shares of our common stock outstanding,
380,931 shares of our Series 2 Class B common stock outstanding and 21,500
shares of our Series 3 Class B common stock outstanding and 3,602 stockholders
of record with respect to such shares. There was also outstanding 1
share of Series 1 Preferred Voting Stock, par value $0.0001, representing 14
Exchangeable Shares, which are exchangeable into 24,388,908 shares of our common
stock.
In
addition, there are 1,321 additional stockholders who did not turn in their
shares of prior classes of our common stock in connection with our
recapitalizations in 2002 and 2004. These stockholders, upon
presentation of their shares, are entitled to receive shares of our common stock
in exchange. As of January 10, 2011 17,362 Series 1 Class B common shares,
107,720 Class A common shares and 12,366 original JagNotes.com Inc. common
shares remained unconverted.
Dividend
Policy
We have
never paid any cash dividends on our common stock and anticipate that, for the
foreseeable future, no cash dividends will be paid on our common
stock.
Equity
Compensation Plans Information
See the
information provided under “Item 12.—Security Ownership of Certain Beneficial
Owners and Related Stockholder Matters—Equity Compensation Plan
Information.”
Recent
Sales of Unregistered Securities
On May
25, 2006 we issued 1,250,000 shares of our common stock to YA Global as
repayment for $250,000 of the outstanding principal amount of the Promissory
Note and a $1,900,000 10% secured convertible debenture with a maturity of three
years in consideration of the remaining $1,750,000 of the outstanding principal
amount of the Promissory Note and $150,000 in accrued and unpaid
interest. This issuance was exempt from registration under the
Securities Act, pursuant to Section 3(a)(9) thereof.
In May of
2006, we issued two secured convertible debentures to YA Global in an aggregate
principal amount of $1,250,000, as follows: (i) a $1,250,000 10% secured
convertible debenture and (ii) a $1,000,000 10% secured convertible debenture,
each with a maturity of three years. This issuance was exempt from
registration under the Securities Act, pursuant to Section 4 (2) thereof, as YA
Global is an accredited investor.
Pursuant
to the terms of the secured convertible debentures, YA Global has converted the
entire principal and accrued interest on the debentures into an aggregate of
14,651,265 shares of common stock, as set forth below:
|
Date
|
Principal Amount
Converted
|
Number of Shares
|
||||||
|
June
28, 2006
|
$ | 25,000 | 125,313 | |||||
|
July
21, 2006
|
$ | 50,000 | 279,642 | |||||
|
August
3, 2006
|
$ | 50,000 | 279,642 | |||||
|
August
15, 2006
|
$ | 50,000 | 289,855 | |||||
|
September
14, 2006
|
$ | 50,000 | 556,793 | |||||
|
September
18, 2006
|
$ | 50,000 | 556,793 | |||||
|
November
20, 2006
|
$ | 150,000 | 1,246,883 | |||||
|
November
30, 2006
|
$ | 105,000 | 872,818 | |||||
|
January
5, 2007
|
$ | 100,000 | 629,327 | |||||
|
August
23, 2007
|
$ | 500,000 | 1,250,000 | |||||
|
August
28, 2007
|
$ | 420,000 | 1,050,000 | |||||
|
September
25, 2007
|
$ | 550,000 | 1,375,000 | |||||
|
September
28, 2007
|
$ | 130,000 | 325,000 | |||||
|
November
14, 2007
|
$ | 300,000 | 750,000 | |||||
|
December
17, 2007
|
$ | 1,000,000 | 2,500,000 | |||||
|
January
24, 2008
|
$ | 295,000 | 737,500 | |||||
|
January
31, 2008
|
$ | 860,679 | 2,151,699 | |||||
On May
25, 2006, as partial consideration for YA Global’s purchase of the secured
convertible debentures, we issued five warrants to purchase an aggregate of
12,000,000 shares of our common stock, as follows. For a description
of such warrants, see “Item 1.—Business—Financing
Arrangements—Warrants.” This issuance was exempt from registration
under the Securities Act, pursuant to Section 4(2) thereof, as YA Global is an
accredited investor.
21
Pursuant
to the terms of the warrants, as amended, YA Global has exercised all of the
shares under the warrants as follows:
|
Warrant No.
|
Exercise Date
|
# of Shares Exercised
|
Exercise Price
|
Gross Proceeds
|
||||||||||
|
CCP-1
|
January
31, 2008
|
700,000 | $ | 0.40 | $ | 280,000 | ||||||||
|
May
16, 2008
|
1,300,000 | $ | 0.40 | $ | 520,000 | |||||||||
|
CCP-2
|
April
30, 2009
|
2,000,000 | $ | 0.05 | $ | 100,000 | ||||||||
|
CCP-3
|
March
12, 2009
|
2,000,000 | $ | 0.07 | $ | 140,000 | ||||||||
|
CCP-4
|
September
30, 2008
|
1,750,000 | $ | 0.20 | $ | 350,000 | ||||||||
|
May
29, 2009
|
625,000 | * | $ | 0.20 | $ | 0.00 | ||||||||
|
CCP-5
|
June
1, 2009
|
2,000,000 | $ | 0.40 | $ | 800,000 | ||||||||
|
July
28, 2009
|
750,000 | $ | 0.18 | $ | 135,000 | |||||||||
|
August
4, 2009
|
250,000 | $ | 0.18 | $ | 45,000 | |||||||||
| 11,375,000 | $ | 2,370,000 | ||||||||||||
*This
portion of warrant No CCP-4 was exercised on a “cashless basis“ resulting in a
reduction of 625,000 warrant shares under this warrant
The
Company claims an exemption from the registration requirements of the Securities
Act of 1933, as amended, for the issuance of shares to as provided above
pursuant to Section 4(2) of the Act and/or Rule 506 of Regulation D promulgated
thereunder since, among other things, the transaction does not involve a public
offering, the purchaser is an “accredited investor” and/or qualified
institutional buyers, the purchaser has access to information about the Company
and its purchase, the purchaser will take the securities for investment and not
resale, and the Company is taking appropriate measures to restrict the transfer
of the securities.
Purchases
of Equity Securities
There
were no repurchases made for any class or series of securities in a month within
the fourth quarter of the fiscal year ended October 31, 2010.
ITEM
6. SELECTED FINANCIAL DATA
We are a
smaller reporting company as defined by Rule 12b-2 of the Exchange Act and are
not required to provide information under this item.
ITEM
7. MANAGEMENT’S DISCUSSION AND ANALYSIS OR PLAN OF
OPERATION
This
annual report contains forward-looking statements relating to future events or
our future financial performance. In some cases you can identify
forward-looking statements by terminology such as “may”, “should”, “expects”,
“plans”, “anticipates”, “believes”, “estimates”, “predicts”, “potential”, or
“continue” or the negative of these terms or other comparable
terminology. These statements are only predictions and involve known
and unknown risks, uncertainties and other factors which may cause our or our
industry’s actual results, levels of activity or performance to be materially
different from any future results, levels of activity or performance expressed
or implied by these forward-looking statements.
Although
we believe that the expectations reflected in the forward-looking statements are
reasonable, we cannot guarantee future results, levels of activity or
performance. You should not place reliance on these statements, which
speak only as of the date that they were made. These cautionary
statements should be considered with any written or oral forward-looking
statements that we may issue in the future. Except as required by
applicable law, including the securities laws of the United States, we do not
intend to update any of the forward –looking statements to conform these
statements to actual results, later events or circumstances or to reflect the
occurrence of unanticipated events.
22
In
this annual report unless otherwise specified, all dollar amounts are expressed
in United States dollars and all references to “common shares” refer to the
common shares of our capital stock.
The
management’s discussion and analysis of our financial condition and results of
operations are based upon our financial statements, which have been prepared in
accordance with accounting principles generally accepted in the United States of
America (“GAAP”).
The
financial statements contained herein include the results CardioGenics, Inc. and
its subsidiaries and CardioGenics Holdings, Inc. and its subsidiaries
(“CardioGenics Holdings, Inc.”) (the latter from July 31, 2009, date of
acquisition) which are collectively referred to as the “Company.”
CardioGenics
Holdings, Inc. was until February 11, 2010 a provider of Internet-based equities
research and financial information that offered its subscribers a variety of
stock market research, news and analysis, including "JAG Notes", CardioGenics
Holdings, Inc.'s flagship early morning consolidated research
product.
On July
31, 2009, JAG Media Holdings, Inc. completed a reverse acquisition of privately
held CardioGenics Inc. (“CardioGenics”), an Ontario, Canada Corporation. The
acquisition was effected pursuant to a Share Purchase Agreement dated May 22,
2009 by and among JAG Media Holdings, Inc., CardioGenics Inc. and CardioGenics
ExchangeCo Inc., the Company’s wholly owned subsidiary (“ExchangeCo”). In
accordance with the terms of the Share Purchase Agreement, 99% of the holders of
common shares of CardioGenics Inc. (two (2) minority shareholders of
CardioGenics holding in aggregate 17,387 common shares of CardioGenics Inc. did
not participate) surrendered their CardioGenics Common Shares to ExchangeCo.
ExchangeCo caused JAG Media Holdings, Inc. to issue to the CardioGenics
shareholders 42,218,361 shares of the Company’s common stock, par value $0.00001
per share (the “Share Consideration”). The Share Consideration provides the
former CardioGenics shareholders with direct and/or indirect ownership of
approximately 85% of JAG Media Holdings, Inc.’s outstanding common stock (on a
fully diluted basis) as of July 31, 2009.
On
October 27, 2009 the name of the Company was changed from Jag Media Holdings,
Inc. to CardioGenics Holdings, Inc.
CardioGenics
develops technology and products targeting the immunoassay segment of the In-Vitro Diagnostic testing
market. CardioGenics has developed the QL Care Analyzer, a proprietary Point Of
Care immuno-analyzer, which will run a number of diagnostic tests
under development by CardioGenics, the first of which will be a
series of cardiovascular diagnostic tests. As part of its core proprietary
technology, CardioGenics has also developed a proprietary method for silver
coating paramagnetic microspheres (a fundamental platform component of
immunoassay equipment), which improve instrument sensitivity to light.
CardioGenics’ principal offices are located in Mississauga, Ontario,
Canada.
With the
acquisition of CardioGenics, the Company’s business is now refocused on
developing technologies and products for the point-of-care In Vitro Diagnostics
market.
On
February 11, 2010, the Company entered into an LLC Membership Interest Purchase
Agreement with Rothcove Partners LLS (“Rothcove”) pursuant to which the Company
sold its interest in the internet-based equities research and financial
information business to Rothcove.
On April
23, 2010, the Company’s Board of Directors approved a reverse stock split of its
issued and outstanding common shares. The total authorized shares of
common stock was at the same time reduced to 65,000,000. The Board of
Directors selected a ratio of one-for-ten and the reverse stock split was
effective on June 20, 2010. Trading of the Company’s common stock on
the Over-The-Counter Capital Market on a split adjusted basis began at the open
of trading on June 21, 2010. The reverse stock split affected all
shares of the Company’s common stock, as well as options to purchase the
Company’s common stock and other equity incentive awards and warrants that were
outstanding immediately prior to the effective date of the reverse stock
split. All references to common shares and per-share data for prior
periods have been retroactively restated to reflect the reverse stock split as
if it had occurred at the beginning of the earliest period
presented.
Results
of Operations for the Years Ended October 31, 2010 and October 31,
2009
The
following table sets forth the Company’s results of operations for the years
ended October 31, 2010 and October 31, 2009 and for the year ended October 31,
2009 includes the results of operations of CardioGenics Holdings, Inc. from July
31, 2009 (date of acquisition) to October 31, 2009:
23
|
Year Ended October 31,
|
||||||||
|
2010
|
2009
|
|||||||
|
Operating
Expenses:
|
||||||||
|
Amortization
of property and equipment
|
$ | 20,932 | $ | 26,157 | ||||
|
Amortization
of patent application costs
|
3,023 | 4,181 | ||||||
|
Write-off
of patent application costs
|
105,345 | 23,803 | ||||||
|
General
and administrative
|
1,160,509 | 1,597,010 | ||||||
|
Write-off
of goodwill
|
— | 12,780,214 | ||||||
|
Research
and product development, net of investment tax credits
|
692,036 | 1,572,337 | ||||||
|
Total
operating expenses and operating loss
|
1,981,845 | 16,003,702 | ||||||
|
Other
Expenses (Income):
|
||||||||
|
Interest
expense and bank charges (Net)
|
30,166 | 571,840 | ||||||
|
Loss
on change in value of derivative liability
|
— | 12,421,023 | ||||||
|
Loss
(gain) on foreign exchange transactions
|
104,182 | (184,389 | ) | |||||
|
Total
other expenses (income)
|
134,348 | 12,808,474 | ||||||
|
Loss
from Continuing Operations
|
(2,116,193 | ) | (28,812,176 | ) | ||||
|
Discontinued
Operations
|
||||||||
|
Gain
on sale of subsidiary
|
90,051 | — | ||||||
|
Loss
from discontinued operations
|
(12,355 | ) | (115,407 | ) | ||||
|
Net
Loss
|
$ | (2,038,497 | ) | $ | (28,927,583 | ) | ||
Revenues
Cardiogenics
is a development stage company and as such has no revenues from
operations.
Operating
expenses
General
and administrative expenses
General
and administrative expenses consist primarily of compensation to officers,
occupancy costs, professional fees, listing costs and other office expenses. The
change in general and administrative expenses is attributable primarily to an
increase in professional and consulting fees and a reduction to zero in shares
rewarded to officers in the current period.
Write-off
of goodwill
On July
31, 2009, JAG Media Holdings, Inc. completed a reverse acquisition of privately
held CardioGenics Inc. (“CardioGenics”), an Ontario, Canada Corporation. The
acquisition was effected pursuant to a Share Purchase Agreement dated May 22,
2009 by and among JAG Media Holdings, Inc., CardioGenics Inc. and CardioGenics
ExchangeCo Inc., the Company’s wholly owned subsidiary (“ExchangeCo”). The
purchase price, arrived at based on the average price of JAG Media Holdings,
Inc.’s common shares, was allocated primarily to goodwill of $12,780,214.
Subsequent to completing the purchase management has analyzed the operations of
the acquired company and determined that considerable resources would have to be
allocated to those operations to make them significantly viable. Since the
Company intends to concentrate its activities on developing technologies and
products for the point-of-care In Vitro Diagnostics market. In November 2009,
management decided to sell its interest in the JAG Notes business. Offers were
received in the range of $40,000 to $60,000. Based on these offers, management
determined that the value of the goodwill associated therewith was impaired. A
$12.8 million goodwill impairment charge was recorded in the fourth quarter of
2009.
24
Research
and product development costs, net of investment tax credits
Research
and development expenses consist primarily of salaries and wages paid to
officers and employees engaged in those activities and supplies consumed
therefor. The change in research and development expenses is attributable
primarily to an increase in compensation to an officer in 2010, an increase in
staff engaged in R&D in 2010, the absence of research tax credits in 2010
(2009-$160,000) and a reduction in shares awarded to officers and employees in
the current period.
Other
expenses (income)
Interest
expense and bank charges, net
The
decrease in interest expense of approximately $542,000 arises from the fact that
the debentures and director’s loan which existed and carried interest in 2009
were for the most part paid off before the beginning of fiscal
2010.
Loss
from discontinued operations and gain on sale of subsidiary
On
February 11, 2010, the Company sold its JAG Media division, realizing
a gain of approximately $90,000. The Company has treated the
operating results of that division in these financial statements as loss from
discontinued operations.
Loss
on change in value of derivative liability
The loss
on change in value of the derivative liability for the year ended October 31,
2009 arises out of the conversion of warrants of CardioGenics Inc. to warrants
of the Company on completion of the reverse acquisition on July 31, 2009 and the
issuance of options and warrants to an agent for assisting in the conversion of
debentures to common shares in concert with the acquisition. The Company
determined that, based on the guidance in “Accounting for Derivative Financial
Instruments Indexed to, and Potentially Settled in a Company’s Own Stock”, the
Company was prohibited from concluding that it would have a sufficient number of
authorized and unissued shares to net-share settle any of those warrants and
options or any other warrants or options previously issued or granted to
non-employees because the conversion of our secured convertible debentures and
the related warrants could have resulted in the issuance of an indeterminable
number of common shares as they were convertible at a discount from the market
price. The Company therefore had to record the warrants and options at fair
value of $12,325,833 and $95,190 respectively.
On
September 30, 2009, the Company’s articles of incorporation were amended to
increase the total number of common shares authorized for issuance from
500,000,000 shares to 650,000,000 shares of common stock, par value $0.00001 per
share. As a result, the total number of shares of all classes of capital stock
authorized for issuance by the Company increased from 550,440,000 shares to
700,440,000 shares with a par value of $.00001 per share, of which 50,000,000
shares are authorized for issuance as preferred stock, 500,000,000 shares are
authorized for issuance as common stock, 400,000 shares are authorized for
issuance as Series 2 Class B common stock and 40,000 shares are authorized for
issuance as Series 3 Class B common stock. The increase in authorized capital
allowed management to conclude that it does have a sufficient number of
authorized and unissued shares to net-share settle any of those warrants or any
other warrants or options previously issued or granted to employees or
non-employees. As a result of this increase, the fair value of this derivative
liability was determined to be zero and therefore the re-valued amount of
$13,501,360 was credited to Additional Paid-In Capital.
Loss
(gain) on foreign exchange transactions
The
Company conducts the majority of its transactions in Canadian dollars. The
foreign exchange loss (gain) (2010-$104,182, 2009-($184,389)) results from
currency movements on transactions settled during the year.
Liquidity
and Capital Resources
For the
year ended October 31, 2010 the Company incurred a net loss of approximately
$2,038,000 (2009-$28,900,000) and a cash flow deficiency from operating
activities of approximately $2,163,000 (2009-$735,000). The Company has not yet
established an ongoing source of revenues sufficient to cover our operating
costs and allow us to continue as a going concern. The Company has funded its
activities to date almost exclusively from debt and equity financings. These
matters raise substantial doubt about the Company’s ability to continue as a
going concern and our independent auditors included an explanatory paragraph to
emphasize such doubt in their report on the audit of our financial
statements.
25
The
Company will continue to require substantial funds to continue research and
development, including preclinical studies and clinical trials of our products,
and to commence sales and marketing efforts. The Company’s plans
include financing activities such as private placements of its common stock and
issuances of convertible debt instruments. The Company is also
actively pursuing industry collaboration activities including product licensing
and specific project financing.
The
Company believes that it will be successful in obtaining the necessary financing
to fund its operations, meet revenue projections and manage costs; however,
there are no assurances that such additional funding will be achieved and that
the Company will succeed in its future operations.
Off-Balance
Sheet arrangements
The
Company is not a party to any off balance sheet arrangements.
Seasonality
The
Company does not believe that its business is materially affected by seasonal
trends or inflation. On an ongoing basis, the Company will attempt to minimize
any effect of inflation on its operating results by controlling operating costs
and whenever possible, seeking to insure that subscription rates, and other
revenues when and if they are realized, reflect increases in costs due to
inflation.
Summary
of Critical Accounting Policies and Estimates
The
discussion and analysis of the Company’s financial condition and results of its
operations are based upon its consolidated financial statements, which have been
prepared in accordance with accounting principles generally accepted in the
United States of America for financial statements filed with the SEC.
|
(a)
|
Convertible
Debentures
|
In
accordance with guidance in accounting for convertible securities with
beneficial conversion features or contingently adjustable conversion ratios, the
Company recognized an imbedded beneficial conversion feature present in the
convertible debentures. The Company allocated a portion of the proceeds equal to
the intrinsic value of that feature to additional paid-in
capital. The debt discount attributed to the beneficial conversion
feature is amortized over the convertible debenture's maturity period as
interest expense using the effective yield method.
In
addition, the Company recognized the value attributable to the warrants to
additional paid-in capital and a discount against the convertible debentures.
The Company valued the warrants using the Black-Scholes pricing
model. The debt discount attributed to the value of the warrants
issued is amortized over the convertible debenture’s maturity period as interest
expense using the effective yield method.
|
(b)
|
Research
and Development Costs
|
Expenditures
for research and development are expensed as incurred and include, among other
costs, those related to the production of prototype products, including payroll
costs. Amounts expected to be received from governments under Scientific
Research Tax Credit arrangements are offset against current
expenses. The Company recognizes revenue from restricted grants in
the period in which the Company has incurred the expenditures in compliance with
the specific restrictions.
|
(c)
|
Income
Taxes
|
The
Company utilizes the liability method of accounting for income taxes as set
forth in the authoritative guidance. Under the liability method, deferred taxes
are determined based on the temporary differences between the financial
statement and tax basis of assets and liabilities using tax rates expected to be
in effect during the years in which the basis differences reverse. A valuation
allowance is recorded when it is more likely than not that some of the deferred
tax assets will not be realized. As there is no certainty that the Company will
generate taxable income in the foreseeable future to utilize tax losses
accumulated to date, no provision for ultimate tax reduction has been made in
these financial statements.
26
On
November 1, 2007, the Company adopted the guidance issued for accounting for
uncertainty in income taxes which provides detailed guidance for the financial
statement recognition, measurement and disclosure of uncertain tax positions
recognized in an enterprise’s financial statements. Income tax positions must
meet a more-likely-than-non recognition threshold at the effective date to be
recognized upon the adoption of the guidance and in subsequent periods. The
Company recognizes potential accrued interest and penalties related to
unrecognized tax benefits within operations as income tax expense. Upon
adoption, there were no adjustments required.
|
(d)
|
Stock-Based
Compensation
|
The
Company follows the authoritative guidance for stock-based compensation which
requires that new, modified and unvested share-based payment transactions with
employees, such as grants of stock options and restricted stock, be recognized
in the financial statements based on their fair value at the grant date and
recognized as compensation expense over their vesting periods. The Company has
also considered the related guidance of the Security and Exchange Commission
(“SEC”). The Company estimates the fair value of stock options and shares issued
as compensation to employees and directors as of the date of grant using the
Black-Scholes pricing model and restricted stock based on the per share
value. The Company also follows the guidance for equity instruments
that are issued to other than employees for acquiring, or in conjunction with
selling, goods or services for equity instruments issued to consultants which
provides guidance on transactions in which (1) the fair value of the equity
instruments is more reliably measurable than the fair value of the goods or
services received and (2) the counterparty receives shares of stock, stock
options, or other equity instruments in settlement of the entire transaction or,
if the transaction is part cash and part equity instruments, in settlement of
the portion of the transaction for which the equity instruments constitute the
consideration. Options issued with a nominal exercise price in
exchange for services rendered were measured at the fair value of the underlying
services rendered on the date of grant. The expense was recorded to the
statement of operations with a corresponding increase in share capital with no
additional increase in the number of shares as they were legally not yet
exercised.
|
(e)
|
Foreign
Currency Translation
|
The
Company maintains its accounting records for its Canadian operations in Canadian
dollars. Transactions in United States dollars (“USD”) are translated into
Canadian dollars at rates in effect at the date of the transaction and gains or
losses on such transactions are recorded at the time of settlement in the
statement of operations.
The
Company’s reporting currency is the United States Dollar. Foreign
denominated assets and liabilities of the Company are translated into USD at the
prevailing exchange rates in effect at the end of the reporting period, the
historical rate for shareholders’ equity and a weighted average of exchange rate
in effect during the period for expenses, gains and
losses. Adjustments that arise from translation into the reporting
currency are recorded in the accumulated other comprehensive income (loss)
component of stockholders’ equity (deficit).
|
(f)
|
Goodwill
|
Goodwill
and other intangible assets with indefinite lives are tested for impairment
annually, as required by pronouncement, “Goodwill and Other Intangible
Assets”. First, the fair value of the reporting unit is compared to
its carrying value. If the fair value is less than the carrying value, a second
step is performed. In the second step, an implied goodwill value is determined
by deducting the fair value of all tangible and intangible net assets of the
reporting unit from the fair value of the reporting unit. If the implied fair
value of the goodwill as calculated is less than the carrying amount of the
goodwill, an impairment charge is recorded for the difference.
|
(g)
|
Non-controlling
Interest in Consolidated Financial
Statements
|
The
Company follows the authoritative guidance for accounting and reporting for
minority interests which characterizes non-controlling interests as a component
of equity within the consolidated balance sheets.
27
Recent
Accounting Pronouncements
In
September 2006, “Fair Value Measurements” pronouncement was issued which defines
fair value, establishes a framework for measuring fair value in accordance with
accounting principles generally accepted in the United States, and expands
disclosures about fair value measurements. This pronouncement is effective for
financial statements issued for the Company’s fiscal year beginning November 1,
2008, with earlier application encouraged. Any amounts recognized upon adoption
as cumulative effect adjustments will be recorded to the opening balance of
retained earnings in the year of adoption. On February 12, 2008, the effective
date for non-financial assets and liabilities was delayed to fiscal years
beginning on November 15, 2008; however, the effective date for financial assets
remains intact. The Company has adopted the fair value measurements
pronouncement for current assets and liabilities in these financial statements
which has not had a material effect on its consolidated financial
statements.
In
December 2007, “Business Combinations” pronouncement was issued, which
establishes principles and requirements for how an acquirer recognizes and
measures in its financial statements the identifiable assets acquired, the
liabilities assumed, and any non-controlling interest in an acquiree, including
the recognition and measurement of goodwill acquired in a business
combination. This Pronouncement is effective as of the beginning of
the first fiscal year beginning on or after December 15,
2008. Earlier adoption is prohibited. The pronouncement
did not have a material effect on its consolidated financial
statements.
ITEM
7A. QUANTITATIVE AND QUALITATIVE DISCLOSURE ABOUT MARKET RISK
We are a
smaller reporting company as defined by Rule 12b-2 of the Exchange Act and are
not required to provide information under this item.
ITEM
8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
The
consolidated financial statements and supplementary data required in this item
are set forth beginning on Page F-1 of this Annual Report on Form
10-K.
ITEM
9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL
DISCLOSURE
2010
None.
2009
On July
31, 2009, JAG Media Holdings, Inc. completed a reverse acquisition of privately
held CardioGenics Inc. (“CardioGenics”), an Ontario, Canada Corporation. The
auditors of CardioGenics were BDO Dunwoody LLP. The auditors of JAG Media
Holdings, Inc. were J.H. Cohn LLP.
Given the
long history of J.H. Cohn LLP as auditors of JAG Media Holdings, Inc., the
Company elected to appoint J.H. Cohn LLP as auditors of the successor company,
herein referred to as the Company.
The
reports of BDO Dunwoody LLP on CardioGenics’ financial statements for the fiscal
years ended October 31, 2008 and 2007 did not contain an adverse opinion or a
disclaimer of opinion and were not qualified or modified as to uncertainty,
audit scope, or accounting principles.
During
our fiscal years ended October 31, 2008 and 2007 and the subsequent interim
period through August 31, 2009, the date on which the directors approved the
engagement of J.H. Cohn LLP and BDO Dunwoody LLP ceased being our auditors,
there were no disagreements between us and BDO Dunwoody LLP on any matter of
accounting principles or practices, financial statement disclosure, or auditing
scope or procedure, which disagreements, if not resolved to the satisfaction of
BDO Dunwoody LLP, would have caused BDO Dunwoody LLP to make reference to the
subject matter of the disagreements in connection with its audit reports on our
financial statements. During our past fiscal year ended October 31, 2009 BDO
Dunwoody LLP did not advise us of any of the matters specified in Item
304(a)(1)(v) of Regulation S-K.
28
ITEM
9A. CONTROLS AND PROCEDURES
Evaluation
of Disclosure Controls and Procedures
Our
management, consisting of our Chief Executive Officer and Chief Financial
Officer, has evaluated the effectiveness of our disclosure controls and
procedures (as such term is defined in Rules 13a-15(e) and 15d-15(e) under the
Exchange Act) using the
Committee of Sponsoring Organizations of the Treadway Commission (COSO)
framework, in connection with the preparation of this Annual Report on
Form 10-K, as of October 31, 2010.
Based on
the review described above, our Chief Executive Officer and Chief Financial
Officer determined that our disclosure controls and procedures were not
effective as of the end of the period covered by this report.
Management’s
Report on Internal Control Over Financial Reporting
Management
is responsible for establishing and maintaining adequate internal control over
financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) under the
Exchange Act) to provide reasonable assurance regarding the reliability of our
financial reporting and preparation of financial statement for external purposes
in accordance with U.S. generally accepted accounting principles. A control
system, no matter how well designed and operated, can provide only reasonable
assurance of achieving the desired control objectives. Because of the inherent
limitations in all control systems, internal controls over financial reporting
may not prevent or detect misstatements. The design and operation of a control
system must also reflect that there are resource constraints and management is
necessarily required to apply its judgment in evaluating the cost-benefit
relationship of possible controls.
Our
management concluded that as at October 31, 2010 our internal controls over
financial reporting were not effective. Management has identified the following
material weaknesses in our internal controls over financial
reporting:
|
•
|
lack
of documented policies and
procedures;
|
|
•
|
lack
of resources to account for complex and unusual transactions;
and
|
|
•
|
there
is no effective separation of duties, which includes monitoring controls,
between the members of
management.
|
Management
is currently evaluating what steps can be taken in order to address these
material weaknesses.
This
Annual Report on Form 10-K does not include an attestation report of our
registered public accounting firm regarding internal controls over financial
reporting. Management’s report was not subject to attestation by our registered
public accounting firm pursuant to temporary rules of the SEC that permit us to
provide only a management’s report.
Changes
in Internal Controls Over Financial Reporting
There
were no significant changes (including corrective actions with regard to
significant deficiencies or material weaknesses) in our internal controls over
financial reporting that occurred during the quarter ended October 31, 2010,
that have materially affected, or are reasonably likely to materially affect,
our internal control over financial reporting.
ITEM
9B. OTHER INFORMATION
There are
no items that required disclosure in a Form 8-K during the fourth quarter of the
year covered by this Form 10-K that were not reported by the
Company.
29
PART
III
ITEM
10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
The
following table sets forth the name, age and position of each of the members of
our board of directors, executive officers, and certain significant employees as
of the fiscal year ending October 31, 2010. All directors are elected to hold
office until the next annual meeting of stockholders following election and
until their successors are duly elected and qualified. Executive officers are
appointed by the Board of Directors and serve at the discretion of the
Board.
Board
of Directors and Executive Officers
|
Name
|
Age
|
Position
|
||
|
Yahia
Gawad
|
52
|
Director
& Chief Executive Officer
|
||
|
Alexander
D.G. Reid
|
72
|
Director
|
||
|
J
Neil Tabatznik
|
60
|
Director/Acting
Chairman
|
||
|
Linda
J. Sterling
|
49
|
Director
& Secretary
|
||
|
James
Essex
|
62
|
Chief
Financial
Officer
|
Yahia Gawad, MB,
Ch.B., MD, MSc. (age 52, Director and Chief Executive Officer of CardioGenics
since 1997). Dr. Gawad is a
Physician/Scientist with primary training in Cardiology, Biochemistry and
Immunology. He received his medical education and post-graduate
training at the University of Alexandria and the University of Toronto. Dr.
Gawad's academic and commercial experience and expertise include many years of
designing and managing cardiovascular disease research and product
development.
Dr. Gawad
was a co-founder of a division of Nanogen (NGEN) (formerly Syn X and Skye
Pharmatech) where he held the position of Vice-President, Medical Affairs. Prior
to that, he was Director of Clinical Research and Development at Spectral
Diagnostics Inc. (now Nanogen).
For the
past 16 years, he has been working extensively on cardiac diagnostic test
products. He has prepared, submitted and obtained FDA regulatory
approvals for several cardiac test products currently being marketed (including
Cardiac Status Troponin I®, Myoglobin® and Myoglobin/CK-MB®, registered
trademarks of Spectral Diagnostics Inc.). Through his expertise and
contributions to an international committee, a new cardiac test, Troponin I, is
now in routine clinical use.
In
addition, Dr. Gawad has researched, developed and published several other tests.
Dr. Gawad has received several awards and scholarships and was a member of both
the Clinical Committee of the American Heart Association and the POC division of
the American Association for Clinical Chemistry. He has served as a reviewer for
the editorial board of the American Journal of Cardiology (1999-2003). Dr. Gawad
published extensively and presented his research and clinical findings at
national and international symposia.
Neil Tabatznik
(age 60, Director of CardioGenics since 2005, Acting Chairman of CardioGenics
since 2009). Mr.
Tabatznik is the Chairman, CEO of Arrow Pharmaceuticals Inc. Arrow
Pharmaceuticals is part of a global generic drug company established in 2000,
and has seen rapid growth from $0 to $700 million in 8 years. The Arrow Group
has sales operations in 5 continents and employs more than 1000 people
worldwide. Prior to Arrow Pharmaceuticals, Mr. Tabatznik was the Chairman, CEO
of Genpharm Inc. (1993-2000), which was acquired by MerckKGaA in 1994 and is now
a part of Mylan Inc. the world's third largest generic and specialty
pharmaceutical company. He was a Barrister-at-Law in London and was called to
the Bar of England and Wales in 1978. He has extensive expertise in
pharmaceutical manufacturing and negotiations of agreements with multinational
companies.
Alexander D.G.
Reid (age 72, Director of CardioGenics since 1998). Mr. Reid has been in the
financial community with experience in public and private companies for over 30
years. He has held numerous positions and board memberships in various financial
and non-financial corporations. For many years, Mr. Reid was the author of the
market business column in the Financial Post. Through his writing, various
business models have been analysed and critiqued. He has been
involved with the Company as a shareholder since 1999;
30
Linda J. Sterling
(age 49, Corporate Secretary of CardioGenics since 2003, Director of
CardioGenics since 2009). Ms. Sterling has been in
the legal community in the capacity as a Law Clerk with both Stikeman Elliott
LLP and Davies Ward Phillips & Vineberg LLP since 1999. She developed
expertise with both public and private company legal compliance and has been
responsible for CardioGenics' compliance and maintenance of corporate governance
since 2001. She is currently in the process of being licensed as a Legal
Executive (F.Inst.L.C.O.), with the Institute of Law Clerks of Ontario, of which
she is a member. She has held the position of CEO and director of Sterling
Studios since 1989.
James A. Essex,
CA, MBA (age 62, Chief Financial Officer of CardioGenics since 2001) Mr.
Essex has been with CardioGenics since 1999. He founded J. Hunter &
Associates Inc. in 1990, a private financial consulting firm. Previously, he was
a co-owner, President and COO of Calais Investigations, Inc., a private company
(from 1993 to 1998), a Vice President of Confederation Trust (1989) and a Vice
President of Chemical Bank of Canada (now JP Morgan Chase Bank of Canada) from
1977 through 1987.
Family
Relationships
There are
no family relationships among the directors and executive officers.
Involvement
in Legal Proceedings
We know
of no pending proceedings to which any director, member of senior management, or
affiliate is either a party adverse to us, or our subsidiaries, or has a
material interest adverse to us or our subsidiaries.
None of
our executive officers or directors have (i) been involved in any bankruptcy
proceedings within the last five years, (ii) been convicted in or has pending
any criminal proceedings, (iii) been subject to any order, judgment or decree
enjoining, barring, suspending or otherwise limiting involvement in any type of
business, securities or banking activity or (iv) been found to have
violated any federal, state or provincial securities or commodities law and such finding has not been reversed,
suspended or vacated.
Board
Committees
Our Board
of Directors does not have standing audit, nominating or compensation
committees. Instead, the functions that might be delegated to such committees
are carried out by our entire Board of Directors, to the extent required.
Our Board of Directors anticipates forming one or more of such committees during
our 2011 fiscal year.
Nomination
of Directors
There
have been no material changes to the procedures by which our security holders
may recommend nominees to our Board of Directors.
Section
16(a) Beneficial Ownership Reporting Compliance
Under the
securities laws of the United States, our directors, executive officers and any
person holding more than 10% of our common stock are required to file initial
forms of ownership of our common stock and reports of changes in that ownership
at the SEC. Specific due dates for these forms have been established,
and we are required to disclose in this report any failure to file by these
dates.
Based
solely on our review of the copies of such forms received by it with respect to
fiscal year 2010, or written representations from certain reporting persons, to
the best of our knowledge, all reports were filed on a timely
basis.
Code
of Ethics
We have
adopted a Code of Ethics (our “Code of Ethics”) that applies
to our Chief Executive Officer and Chief Financial Officer. We will
provide to any person without charge, upon request, a copy of our Code of Ethics
by sending such request to the attention: Yahia Gawad, Chief
Executive Officer, CardioGenics Holdings Inc., 6295 Northam Drive, Unit 8,
Mississauga, Ontario L4V 1W8. The Company will promptly disclose any
amendments or waivers to our Code of Ethics on Form 8-K.
31
ITEM
11. EXECUTIVE COMPENSATION
As a
“smaller reporting company,” CardioGenics has elected to follow scaled
disclosure requirements for smaller reporting companies with respect to Part III, Item 11 – Executive
Compensation. Under the scaled disclosure obligations, CardioGenics is
not required to provide CompensationDiscussion and Analysis
and certain other tabular and narrative disclosures relating to executive
compensation. Nor is CardioGenics required to quantify payments due to the named
executives upon termination of employment. Management believes that the scaled
disclosure for the Company’s executive compensation policy and practices is
appropriate because CardioGenics is small for a publicly-traded company, has
only three named executives and has a relatively simple compensation policy and
structure that has not changed in the last fiscal year.
Summary
Compensation Table
The
following table provides information concerning compensation of CardioGenics’
named executives for CardioGenics’ last two completed fiscal years ending
October 31, 2009 and 2010.
|
Name &
Principal
Position
|
Year
|
Salary
$
|
Bonus
$
|
Stock
Awards
$
|
Option
Awards
$
|
Non-Equity
Incentive Plan
Compensation
$
|
Non-Qualified
Deferred
Compensation
Earnings
$
|
All
Other
Compensation
$
|
Total
$
|
||||||||||||||||||||||
|
Dr.
Yahia Gawad,
|
2010
|
143,968 |
(1)
|
143,968 | |||||||||||||||||||||||||||
| Chief Executive Officer |
2009
|
85,426 |
(1)
|
927,235 |
(2)
|
— | — | — | — | 1,012,661 | |||||||||||||||||||||
|
James
A. Essex,
|
2010
|
35,881 |
(1)
|
35,881 | |||||||||||||||||||||||||||
| Chief Financial Officer |
2009
|
— | 162,936 |
(2)
|
— | — | — | — | 162,936 | ||||||||||||||||||||||
|
Linda
J. Sterling,
|
2010
|
45,590 | 45,590 | ||||||||||||||||||||||||||||
| Secretary |
2009
|
— | 294,008 |
(2)
|
— | — | — | — | 294,008 | ||||||||||||||||||||||
|
(1)
|
Cash
compensation is stated in the table in U.S. dollars. To the extent any
cash compensation was paid in Canadian dollars, it has been converted into
U.S. dollars based on the average Canadian/U.S. dollar exchange rate for
the years ended October 31, 2010 and October 31, 2009.
|
|
(2)
|
This
amount represents the dollar amount recognized for financial statement
reporting purposes with respect to the fiscal year ended October 31, 2009
for stock awards granted in May and July 2009, a portion of which was in
respect of fiscal years 2001 through 2008 and was immediately vested. The
fair value is calculated using the price at which CardioGenics was selling
stock through private placements around the date of grant. This amount
reflects our accounting expense for these awards, and does not correspond
to the actual value that will be recognized by the named
executives.
|
Other
Benefit Plans
The
Company has no defined benefit or actuarial pension plans.
Employment
Agreements
We
currently do not have written employment agreements with any of our current
officers or executive personnel, except for Dr. Yahia Gawad who has a 3 year
employment agreement with CardioGenics Holdings Inc. with an annual salary of
$150,000, heath and dental insurance coverage on terms not less favorable than
the health insurance coverage to be offered by the Company to its employees,
performance bonuses in the form of cash and stock options to be proposed to the
Board of Directors on an annual basis, non-compete agreement for 24 months after
effective takeover and 18 months full salary severance pay and benefit for
firing without cause. Further, for each calendar year of the Term he will be
entitled to five (5) weeks paid vacation. Also, he will be eligible for Stock
Option incentives to the executives as approved by the Board of
Directors.
32
With
respect to our former directors and executives, Messrs. Thomas J. Mazzarisi and
Stephen J. Schoepfer, they each received to the date of change in control an
annual base salary of $150,000 pursuant to their amended and restated employment
agreements.
Pursuant
to these employment agreements, Messrs. Mazzarisi and Schoepfer were also
entitled to the same medical and other benefits, including health and life
insurance coverage, as are provided to our other employees. The
agreements also provided that in the event the employment of Messrs. Mazzarisi
and Schoepfer are terminated without cause or such executive resigns for good
reasons as defined in the employment agreements, they shall be entitled to
receive (i) continued medical and life insurance coverage for a period equal to
the greater of one year or the number of years and fractions thereof between the
date of such termination and the end of the term (the “Severance Period”), (ii) a
lump sum cash payment equal to the executive’s highest rate of annual salary in
effect during the term multiplied by the Severance Period, (iii) a lump sum cash
payment equal to the number of accrued and unused vacation days calculated at
the executive’s then current salary rate and (iv) accelerated vesting of all of
the executive’s outstanding stock options. Such cash payments are
required to be made within ten days of termination of employment, and shall not
be subject to offset amounts earned by the executive in respect of any
subsequent employment, nor is the executive required to seek any such subsequent
employment.
The
employment agreements further provided that, immediately prior to a “change in
control” (as defined in our 1999 Long-Term Incentive Plan), Messrs. Mazzarisi
and Schoepfer were to be granted an option to acquire 100,000 shares of our
common stock (subject to equitable adjustments for stock splits, etc.) at an
exercise price equal to the fair market value of the average closing bid price
for shares of our common stock for the 30 days prior to such change in control,
which option shall be fully vested and immediately exercisable in full and
expire on a date which is the earlier of ten years from such change in control
and three years after termination of employment. Generally, under our
1999 Long-Term Incentive Plan a “change in control” shall be deemed to have
occurred (i) if there is an acquisition of 30% or more of our then outstanding
shares of common stock, (ii) Messrs. Mazzarisi and Schoepfer cease for any
reason to constitute at least a majority of the members of our Board of
Directors, or (iii) a merger, shall have occurred. However, a change
in control shall not be deemed to have occurred if consummation of such a
transaction would result in at least 70% of the total voting power represented
by our voting securities outstanding immediately after such transaction being
beneficially owned by at least 75% of the holders of our outstanding voting
securities immediately prior to the transaction, with the voting power of each
such continuing holder relative to other such continuing holders not
substantially altered in the transaction.
As the
CardioGenics Acquisition resulted in a “change in control” as described above,
Messrs. Mazzarisi and Schoepfer were each granted upon their resignation at the
closing an option to acquire 100,000 shares of our common stock at an exercise
price of $3.40 per share, in accordance with the terms of their respective
employment agreements.
Outstanding
Equity Awards at October 31, 2010 Fiscal Year End
|
Name
|
Number of
Securities
underlying
unexercised
options
exercisable
|
Number of
Securities
underlying
unexercised
options
unexercisable
|
Option
exercise or
base price
per share
($/Share)
|
Option
Expiration Date
|
|||||||||
|
Thomas
J. Mazzarisi
|
50,000 | 0 | 0.20 |
August
31, 2011
|
|||||||||
| 100,000 | 0 | 3.40 |
August
1, 2019
|
||||||||||
|
Stephen
J. Schoepfer
|
25,000 | 0 | 0.20 |
August
31, 2011
|
|||||||||
| 100,000 | 0 | 3.40 |
August
1,
2019
|
||||||||||
33
Director
Compensation
Non-Employee
Directors' Compensation
In fiscal
2010 our policy for compensation of non-employee directors was as
follows:
|
|
1.
|
Non-employee
directors do not receive an annual cash base retainer.
|
|
|
2.
|
At
the discretion of the full Board of Directors, nonemployee directors may
receive shares of the Company’s common stock. The number and terms of such
shares is within the discretion of the full Board of
Directors.
|
|
|
3.
|
Directors
who are officers or employees of CardioGenics do not receive separate
consideration for their service on the Board of
Directors.
|
Fiscal
Year 2010 Director Compensation Table
|
Name
|
Stock
Award
As
Director
$
|
Stock
Award
(Other)
$
|
Total(1)
$
|
||||||
|
J.
Neil Tabatznik
|
0 | 0 | |||||||
|
Alexander
D.G. Reid
|
0 | 0 | |||||||
|
(1)
|
As
of October 31, 2010, the aggregate number of shares underlying stock
awards granted to each non-employee director was as follows: Mr. Tabatznik
(561,648) and Mr. Reid (52,393).
|
Indemnification
of Officers and Directors
Our
amended and restated Articles of Incorporation provide that we shall indemnify
our officers, directors, employees and agents to the full extent permitted by
Nevada law. Our Bylaws include provisions to indemnify our officers
and directors [and other persons] against expenses (including judgments, fines
and amounts paid for settlement) incurred in connection with actions or
proceedings brought against them by reason of their serving or having served as
officers, directors or in other capacities. We do not, however,
indemnify them in actions in which it is determined that they have not acted in
good faith or have acted unlawfully or not in our best interest. In
the case of an action brought by or in the right of us, we shall indemnify them
only to the extent of expenses actually and reasonably incurred by them in
connection with the defense or settlement of these actions and we shall not
indemnify them in connection with any matter as to which they have been found to
be liable to us, unless the deciding court determines that, notwithstanding such
liability, that person is fairly entitled to indemnity in light of all the
relevant circumstances.
We do not
currently maintain director’s and officer’s liability insurance but we may do so
in the future.
Insofar
as indemnification for liabilities arising under the Securities Act may be
permitted to our directors and officers pursuant to the foregoing provisions, or
otherwise, we have been advised that in the opinion of the SEC such
indemnification is against public policy as expressed in the Securities Act and
is, therefore, unenforceable.
34
ITEM
12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND RELATED STOCKHOLDER
MATTERS
The
following table sets forth information regarding the beneficial ownership of our
common stock as of January 10, 2011 (except as otherwise indicated) by (i) each
person known by us to be the beneficial owner of more than 5% of our common
stock, (ii) each director and nominee to be a director, (iii) each named
executive officer and (iv) all directors and executive officers as a
group. Except as otherwise indicated below, each of the persons named
in the table has sole voting and investment power with respect to the shares set
forth opposite such person’s name. Unless otherwise indicated the
address of each person listed in this table is c/o CardioGenics Holdings Inc,
6295 Northam Drive, Unit 8, Mississauga, Ontario, Canada L4V 1W8.
|
Name & Address of Beneficial
Owner
|
Number of Shares
Beneficially Owned
|
Percentage of Class**
|
||||||
|
Yahia
Gawad
|
18,144,652
|
33.85
|
%
|
|||||
|
Alexander
D.G. Reid
|
523,196
|
*
|
||||||
|
J.
Neil Tabatznik
|
2,482,534
|
(1)
|
4.63
|
%
|
||||
|
Linda
J. Sterling
|
1,501,617
|
2.80
|
%
|
|||||
|
James
Essex
|
398,183
|
*
|
||||||
|
Thomas
J. Mazzarisi
|
174,350
|
(2)
|
*
|
|||||
|
Stephen
J. Schoepfer
|
153,050
|
(3)
|
*
|
|||||
|
All
executive officers and directors as a group (6 persons)
|
23,377,582
|
43.61
|
%
|
|||||
|
*
|
Less
than one percent (1%)
|
|
**
|
Based
on 53,609,161 shares of common stock issued and
outstanding
|
|
(1)
|
Included
157,178 shares of common stock issuable upon exercise of a
warrant
|
|
(2)
|
Includes
150,000 shares of common stock issuable upon the exercise of stock
options
|
|
(3)
|
Includes
125,000 shares of common stock issuable upon the exercise of stock
options
|
Equity
Compensation Plan Information
The
following table summarizes the shares of our common stock authorized for
issuance under our equity compensation plans as of October 31,
2010.
|
Number of
securities
to be issued upon
exercise of
outstanding options,
warrants and rights
|
Weighted average
exercise price of
outstanding
options, warrants
and rights
|
Number of
securities remaining
available for future
issuance under
equity compensation
plans (excluding
securities reflected
in column (a))
|
||||||||||
|
(a)
|
(b)
|
(c)
|
||||||||||
|
Equity
compensation plans approved by security holders
|
Not
applicable
|
Not
applicable
|
Not
applicable
|
|||||||||
|
Equity
Compensation Plans not approved by security holders
|
275,000 | — | 325,000 |
(1)
|
||||||||
|
TOTAL
|
275,000 | — | 325,000 | |||||||||
|
(1)
|
The
maximum number of shares that may be subject to outstanding awards under
our 1999 Long-Term Incentive Plan is 600,000 shares of Common
Stock. Because this limitation applies only to outstanding
awards under the plan, as the outstanding options included in column (a)
are either exercised, forfeited or expire pursuant to their terms, the
number of shares remaining available for future issuance in column (c)
shall be increased by the number of shares subject to such option so
exercised, forfeited or expired.
|
35
Our 1999
Long-Term Incentive Plan provides our directors, officers, employees and
consultants with the opportunity to participate in our ownership. Our Board of
Directors acts as the committee under the plan which administers the plan,
addressing participation, the awards offered and any applicable conditions of
exercise. In making these determinations, our Board of Directors will generally
consider the participant’s position and record of service to us. The Board of
Directors may issue options, stock appreciation rights, restricted stock,
deferred stock, bonus stock, awards in lieu of cash obligations, dividend
equivalents and other stock based awards, all subject to terms and conditions to
be set by the Board of Directors. The plan also contains standard provisions
dealing with matters such as adjustment of the number of shares subject to
options and covered by the plan in addition to amendment and termination of the
plan.
ITEM
13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR
INDEPENDENCE
As a
smaller reporting company, we are required to follow the scaled disclosure
requirements with respect to this Part III, Item 13 – Certain
Relationships and Related Transactions, and Director Independence. The
disclosures related to review of related person transactions are not applicable
to smaller reporting companies.
Certain
Relationships and Related Transactions
During
the year ended October 31, 2009, the Company utilized advances from a director
totaling approximately $885,000 bearing interest at 10% per annum. On July 31,
2009, the advances from director plus interest of $108,613 were converted to
common shares at a price of $.37 per share for the advances and $.50 for the
interest.
Director
Independence
The Board
of Directors currently consists of four members, two of whom are “independent”
as defined under applicable rules of the SEC and The NASDAQ Stock Market LLC.
The two independent members of the Board of Directors are Neil Tabatznik and
Alexander D. G. Reid. However, since our stock trades on the OTC Bulletin Board,
we are not required to have independent directors.
For a
director to be considered independent, the Board must determine that the
director has no relationship, which, in the opinion of the Board, would
interfere with the exercise of independent judgment in carrying out the
responsibilities of a director.
ITEM
14. PRINCIPAL ACCOUNTING FEES AND SERVICES
J.H. Cohn
LLP has served as our independent auditors since August 31, 2009. The
appointment of J.H. Cohn LLP as our independent public accountants was
unanimously approved by the Board of Directors. J.H. Cohn LLP is the successor
to our former independent auditors, BDO Dunwoody LLP (“BDO”). BDO served as our
independent auditors from December 1, 2007 until August 31, 2009.
The
following table sets forth the aggregate fees paid by CardioGenics for the
fiscal years ended October 31, 2010 and 2009 to our independent
auditors:
|
Fiscal Year Ended
October 2010
|
Fiscal Year Ended
October 2009
|
|||||||
|
Audit
Fees
|
$ | 60,000 |
(1)
|
$ | 60,000 |
(1)
|
||
|
Audit
Related Fees
|
$ | 46,728 |
(2)
|
$ | 36,616 |
(2)
|
||
|
Tax
Fees
|
$ | 0 | $ | 0 | ||||
|
All
Other Fees
|
$ | 0 | $ | 0 | ||||
|
Total
|
$ | 106,728 | $ | 96,616 | ||||
(1)
Represents estimated audit fees for the fiscal year ended October 31,
2010.
(2)
Represents charges of J.H. Cohn LLP, CardioGenics’ auditor in fiscal year ended
October 31, 2010 for review of interim financial statements.
(3) J.H.
Cohn LLP did not provide and did not bill for any tax services.
36
All
Other Fees
There
were no other fees billed by J.H. Cohn LLP in the years ended October 31, 2010
or October 31, 2009.
Pre-Approval
Policies and Procedures
The Board
of Directors is required to pre-approve the rendering by our independent auditor
of audit or permitted non-audit services. The Board of Directors pre-approved
all of the services rendered by J.H. Cohn LLP for the audit of the consolidated
financial statements included in our Annual Reports on Form 10-K and reviews of
consolidated financial statements included in our Quarterly Reports on Form
10-Q.
The
services provided for 2010 were 56% audit services and 44% audit related
fees. The services provided above for 2009 were 63% audit services
and 37% audit related fees.
37
PART
IV
ITEM
15. EXHIBITS, FINANCIAL STATEMENT SCHEDULES
Exhibits
The
following Exhibits are filed as part of this Annual Report on Form 10-K or
incorporated by reference.
|
Exhibit
No.
|
Description
|
|
|
3.1
|
Amended
and Restated Articles of Incorporation of
Registrant. Incorporated by reference to the Registrant’s Form
10-QSB filed with the SEC on June 19, 2006.
|
|
|
3.2
|
Bylaws
of Registrant. Incorporated by reference to the Registrant’s
Form SB-2 filed with the SEC on September 30, 1999.
|
|
|
3.3
|
Certificate
of Designation of Series 1 Preferred Stock of Registrant. Incorporated by
reference to the Registrant’s Form 8-K filed with the SEC on July 24,
2009.
|
|
|
3.4
|
Articles
of Amendment of CardioGenics ExchangeCo Inc. effective July 14 2009 and
Articles of Incorporation of CardioGenics ExchangeCo Inc. Effective May
22, 2009
|
|
|
3.5
|
Certificate
of Amendment to Articles of Incorporation of Registrant. Incorporated by
reference to the Registrant’s Form DEF 14C filed with the SEC on September
9, 2009.
|
|
|
4.1
|
Form
of Common Stock Certificate. Incorporated by reference to the
Registrant’s Form 10-KSB filed with the SEC on November 8,
2005.
|
|
|
4.2
|
Form
of Series 2 Class B Stock Certificate. Incorporated by reference to the
Registrant’s Form 10-KSB filed with the SEC on November 8,
2005.
|
|
|
4.3
|
Securities
Purchase Agreement, effective May 25, 2006, with YA Global. Incorporated
by reference to the Registrant’s Form 8-K filed with the SEC on June 1,
2006.
|
|
|
4.4
|
Letter
Agreement, dated January 31, 2008, relating to the conversion of the
remaining principal balance of the convertible secured
debentures. Incorporated by reference to the Registrant’s Form
8-K filed with the SEC on February 6, 2008.
|
|
|
4.5
|
Warrant
No. CCP-1 for 2,000,000 shares of common stock issued to YA Global,
effective May 25, 2006. Incorporated by reference to the Registrant’s Form
8-K filed with the SEC on June 1, 2006.
|
|
|
4.6
|
Warrant
No. CCP-2 for 2,000,000 shares of common stock issued to YA Global,
effective May 25, 2006. Incorporated by reference to the
Registrant’s Form 8-K filed with the SEC on June 1,
2006.
|
|
|
4.7
|
Warrant
No. CCP-3 for 2,000,000 shares of common stock issued to YA Global,
effective May 25, 2006. Incorporated by reference to the
Registrant’s Form 8-K filed with the SEC on June 1,
2006.
|
|
|
4.8
|
Warrant
No. CCP-4 for 3,000,000 shares of common stock issued to YA Global,
effective May 25, 2006. Incorporated by reference to the
Registrant’s Form 8-K filed with the SEC on June 1,
2006.
|
|
|
4.9
|
Warrant
No. CCP-5 for 3,000,000 shares of common stock issued to YA Global,
effective May 25, 2006. Incorporated by reference to the
Registrant’s Form 8-K filed with the SEC on June 1,
2006.
|
|
|
4.10
|
Letter
Agreement, amending Warrant No. CCP-4. Incorporated by reference to the
Registrant’s Form 8-K filed with the SEC on October 3,
2008.
|
38
|
Exhibit
No.
|
Description
|
|
|
4.11
|
Investor
Registration Rights Agreement, effective May 25, 2006, with YA Global.
Incorporated by reference to the Registrant’s Form 8-K filed with the SEC
on June 1, 2006.
|
|
|
10.1
|
Non-Binding
Letter of Intent, dated October 1, 2008, by and among the Registrant,
BlueCreek, e2 Business and YA Global. Incorporated by reference
to the Registrant’s Form 8-K filed with the SEC on October 3,
2008.
|
|
|
10.2
|
1999
Long-Term Incentive Plan, as amended. Incorporated by reference
to Exhibit [ ] to the Registrant’s Form S-8 filed with the SEC
on May 1, 2002.
|
|
|
10.3
|
Amended
and Restated Employment Agreement, dated August 31, 2001, between Thomas
J. Mazzarisi and Registrant. Incorporated by reference to
Exhibit 10.21 in Amendment No. 1 to the Registrant’s Form SB-2 filed with
the SEC on September 26, 2001.
|
|
|
10.4
|
Amended
and Restated Employment Agreement, dated August 31, 2001, between Stephen
J. Schoepfer and Registrant. Incorporated by reference to
Exhibit 10.20 in Amendment No. 1 to the Registrant’s Form SB-2 filed with
the SEC on September 26, 2001.
|
|
|
10.5
|
Amendment
to Amended and Restated Employment Agreement, dated as of November 3,
2005, between Registrant and Thomas J. Mazzarisi. Incorporated
by reference to the Registrant’s Form 10-KSB filed with the SEC on
November 8, 2005.
|
|
|
10.6
|
Amendment
to Amended and Restated Employment Agreement, dated as of November 3,
2005, between Registrant and Stephen J. Schoepfer. Incorporated
by reference to the Registrant’s Form 10-KSB filed with the SEC on
November 8, 2005.
|
|
|
10.7
|
Amendment
to Amended and Restated Employment Agreement, dated as of November 12,
2007, by and between Registrant and Thomas J.
Mazzarisi. Incorporated by reference to Exhibit 10.6 of
Registrant’s Form 10-K filed with the SEC on November 13,
2008.
|
|
|
10.8
|
Amendment
to Amended and Restated Employment Agreement, dated as of November 12,
2007, by and between Registrant and Stephen J. Schoepfer. Incorporated by
reference to Exhibit 10.7 of the Registrant’s Form 10-K filed with the SEC
on November 13, 2008
|
|
|
10.9
|
Extension
of Amended and Restated Employment Agreement dated as of November 12, 2008
between registrant and Thomas J. Mazzarisi. Incorporated by reference to
Exhibit 10.9 of the Registrant’s Form 10-K filed with the SEC on November
13, 2008.
|
|
|
10.10
|
Extension
of Amended and Restated Employment Agreement dated as of November 12, 2008
between registrant and Stephen J. Schoepfer. Incorporated by reference to
Exhibit 10.10 of the Registrant’s Form 10-K filed with the SEC on November
13, 2008.
|
|
|
10.11
|
Consulting
Agreement, dated November 12, 2007, between the Registrant and Walsh
Organization, Inc.
Incorporated
by reference to Exhibit 10.31 of the Registrant’s Annual Report on Form
10-KSB filed November 13, 2003.
|
|
|
10.12
|
Power
of Attorney and Contingent Fee Contract, dated June 14, 2002, among the
Registrant, Gary Valinoti and the Law Firm of O’Quinn, Laminack &
Pirtle. Incorporated by reference to Exhibit 10.32 of the Registrant’s
Annual Report on Form 10-KSB filed November 13, 2003.
|
|
|
10.13
|
Subscription
Agreement, dated December 10, 2002, between the Registrant and Bay Point
Investment Partners LLC. Incorporated by reference to the Registrant’s
Registration Statement on Form SB-2 filed on January 9,
2003.
|
|
|
10.14
|
Placement
Agent Agreement, dated December 10, 2002, between the Registrant and RMC 1
Capital Markets, Inc. Incorporated by reference to the Registrant’s
Registration Statement on Form SB-2 filed on January 9,
2003.
|
39
|
Exhibit
No.
|
Description
|
|
|
10.15
|
Placement
Agent Agreement, dated as of June 19, 2003, between the Registrant and RMC
1 Capital Markets, Inc., as amended on August 12, 2003. Incorporated by
reference to the Registrant’s Current Report on Form 8-K filed on August
13, 2003.
|
|
|
10.16
|
Subscription
Agreement, dated as of June 19, 2003, between the Registrant and Bay Point
Investment Partners LLC, as amended on August 12, 2003. Incorporated by
reference to the Registrant’s Current Report on Form 8-K filed on August
13, 2003.
|
|
|
10.17
|
Subscription
Agreement, dated as of September 25, 2003, between the Registrant and
Kuekenhof Equity Fund L.P. Incorporated by reference to Exhibit 10.39 of
the Registrant’s Form 10-KSB filed with the SEC on November 13,
2003.
|
|
|
10.18
|
Non-Circumvention/Non-Disclosure
Agreement, dated as of January 1, 2004 between Flow Capital Advisors Inc.
and the Registrant. Incorporated by reference to the Registrant’s Form 8-K
filed with the SEC on July 25, 2007.
|
|
|
10.19
|
Finder’s
Fee Agreement, dated as of January 5, 2004, between the Registrant and
Flow Capital Advisors, Inc. Incorporated by reference to the Registrant’s
Form 8-K filed with the SEC on January 20, 2004.
|
|
|
10.20
|
Finder’s
Fee Agreement, dated as of March 14, 2005, by and between the Registrant
and Flow Capital Advisors, Inc. Incorporated by reference to the
Registrant’s Form 8-K filed with the SEC on July 25,
2007.
|
|
|
10.21
|
Irrevocable
Transfer Agent Instructions, effective May 25, 2006, between the
Registrant and YA Global. Incorporated by reference to the Registrant’s
Form 8-K filed with the SEC on June 1, 2006.
|
|
|
10.22
|
Letter,
dated as of June 17, 2008, from Cryptometrics regarding termination of the
agreement and plan of merger between the Registrant and Cryptometrics.
Incorporated by reference to the Registrant’s Form 8-K filed with the SEC
on June 18, 2008.
|
|
|
10.23
|
Stand-By
Equity Distribution Agreement dated March 12, 2009 between Registrant and
YA Global Master SPV Ltd. Incorporated by reference to the Registrant’s
Form 8-K filed with the SEC on March 13, 2009.
|
|
|
10.24
|
Registration
Rights Agreement dated March 12, 2009 between Registrant and YA Global
Master SPV Ltd. Incorporated by reference to the Registrant’s Form 8-K
filed with the SEC on March 13, 2009.
|
|
|
10.23
|
Share
Purchase Agreement dated May 22, 2009 between Registrant, CardioGenics
ExchnageCo Inc., CardioGenics Inc. And Yahia Gawad, Principal Shareholder
of CardioGenics Inc.
|
|
|
10.24
|
Voting
and Exchange Trust Agreement dated July 6, 2009 among Registrant,
CardioGenics ExchangeCo Inc. and Weirfoulds LLP. Incorporated by reference
to the Registrant’s Form 8-K filed with the SEC on July 6,
2009.
|
|
|
10.25
|
Support
Agreement dated July 6, 2009 between Registrant and CardioGenics
ExchangeCo Inc. Incorporated by reference to the Registrant’s Form 8-K
filed with the SEC on July 6, 2009.
|
|
|
10.26
|
Agreement
dated September 10, 2009 between Registrant and The Investor’s Relations
Group, Inc. by reference to the Registrant’s Form 8-K filed with the SEC
on September 11, 2009.
|
|
|
10.27
|
Agreement
dated September 28, 2009 between Registrant and Gilford
Securities Incorporated by reference to the Registrant’s Form 8-K
filed with the SEC on October 2, 2009.
|
|
|
10.28
|
Retainer
Agreement dated January 20, 2010 between Registrant and Wolf, Axelrod
& Weinberger Associates
LLC
|
40
|
Exhibit
No.
|
Description
|
|
|
10.29
|
Letter
of Agreement dated January 18, 2010 between Registrant and The Investor
Relations Group, Inc.
|
|
|
10.30
|
Employment
agreement dated July 31, 2009 between Registrant and Dr. Yahia
Gawad.
|
|
|
10.31
|
LLC
Membership Interest Purchase Agreement dated February 10, 2010 between
Registrant and Rothcove Partners LLC.
|
|
|
14.1
|
Code
of Ethics. Incorporated by reference to the Registrant’s Form
10-KSB filed with the SEC on November 13, 2003.
|
|
|
21.1
|
Subsidiaries
of Registrant.*
|
|
|
23.1
|
Consent
of J.H. Cohn LLP*
|
|
|
23.2
|
Consent
of BDO Dunwoody LLP*
|
|
|
31.1
|
Section
302 Certification of Chief Executive Officer*
|
|
|
31.2
|
Section
302 Certification of Chief Financial Officer*
|
|
|
32.1
|
Section
906 Certification of Chief Executive Officer and Chief Financial
Officer*
|
*Filed
herewith
41
|
CardioGenics
Holdings Inc.
(A
Development Stage Company)
Table
of Contents
October 31, 2010 and
2009
|
|
Consolidated
Financial Statements
|
|
|
Report
of Independent Registered Public Accounting Firm
|
F-1
|
|
Report
of Independent Registered Public Accounting Firm
|
F-2
|
|
Consolidated
Balance Sheets
|
F-3
|
|
Consolidated
Statements of Operations
|
F-4
|
|
Consolidated
Statements of Stockholders’ Equity (Deficit)
|
F-5-F-12
|
|
Consolidated
Statements of Cash Flows
|
F-13
|
|
Notes
to Consolidated Financial Statements
|
F-14-F-33
|
|
CardioGenics
Holdings Inc.
(A
Development Stage Company)
Report
of Independent Registered Public Accounting Firm
|
To the
Board of Directors and Stockholders
CardioGenics
Holdings, Inc.
We have
audited the accompanying consolidated balance sheet of CardioGenics Holdings
Inc. (a development stage company) and Subsidiaries as of October 31, 2010, and
the related consolidated statements of operations, stockholders' equity
(deficit) and cash flows for each of the two years in the period ended October
31, 2010 and for the period from November 20, 1997 (date of inception) to
October 31, 2010. These consolidated financial statements are the responsibility
of the Company's management. Our responsibility is to express an opinion on
these consolidated financial statements based on our audits. The
financial statements of CardioGenics, Inc. for the period from November 20, 1997
to October 31, 2008 were audited by other auditors whose report dated July 29,
2009 expressed an unqualified opinion on those statements with explanatory
paragraphs relating to the Company’s ability to continue as a going
concern.
We
conducted our audits in accordance with the standards of the Public Company
Accounting Oversight Board (United States). Those standards require that we plan
and perform the audit to obtain reasonable assurance about whether the
consolidated financial statements are free of material misstatement. An audit
includes examining, on a test basis, evidence supporting the amounts and
disclosures in the financial statements. An audit also includes assessing the
accounting principles used and significant estimates made by management, as well
as evaluating the overall financial statement presentation. We believe that our
audits provide a reasonable basis for our opinion.
In our
opinion, the consolidated financial statements referred to above present fairly,
in all material respects, the financial position of CardioGenics Holdings, Inc.
and Subsidiaries as of October 31, 2010, and their results of operations and
cash flows for each of the two years in the period ended October 31, 2010, and
for the period from November 20, 1997 (date of inception) to October 31, 2010,
in conformity with accounting principles generally accepted in the United States
of America.
The
consolidated financial statements referred to above have been prepared assuming
that the Company will continue as a going concern. As further discussed in Note
2 to the consolidated financial statements, the Company's operations have
generated recurring losses and negative cash flows from operating activities.
Such matters raise substantial doubt about the Company's ability to continue as
a going concern. Management's plans concerning these matters are also described
in Note 2. The accompanying consolidated financial statements do not include any
adjustments that might result from the outcome of these
uncertainties.
/s/ J.H.
Cohn LLP
Roseland,
New Jersey
January
31, 2011
F-1
|
CardioGenics
Holdings Inc.
(A
Development Stage Company)
Report
of Independent Registered Public Accounting Firm
|
To the
Directors and Shareholders of
CardioGenics
Holdings Inc. (formerly CardioGenics Inc.)
(A
Development Stage Company)
We have
audited the accompanying consolidated balance sheet of CardioGenics Holdings
Inc. (formerly CardioGenics Inc.) (a development stage company) as at October
31, 2008 and the related consolidated statements of operations, stockholders’
equity (deficit) and cash flows for the year ended October 31,
2008. These consolidated financial statements are the responsibility
of the Company’s management. Our responsibility is to express an
opinion on these consolidated financial statements based on our
audit.
We
conducted our audit in accordance with the standards of the Public Company
Accounting Oversight Board (United States). Those standards require that we plan
and perform the audit to obtain reasonable assurance about whether the
consolidated financial statements are free of material misstatement. The Company
is not required to have, nor were we engaged to perform, an audit of its
internal control over financial reporting. Our audit included consideration of
internal control over financial reporting as a basis for designing audit
procedures that are appropriate in the circumstances, but not for the purpose of
expressing an opinion on the effectiveness of the Company’s internal control
over financial reporting. Accordingly we express no such opinion. An audit also
includes examining, on a test basis, evidence supporting the amounts and
disclosures in the consolidated financial statements and assessing the
accounting principles used and significant estimates made by management, as well
as evaluating the overall financial statement presentation. We believe that our
audit provides a reasonable basis for our opinion.
In our
opinion, the consolidated financial statements referred to above present fairly,
in all material respects, the financial position of CardioGenics Holdings Inc.
(formerly CardioGenics Inc.) (a development stage company) as at
October 31, 2008 and the results of its operations and its cash flows for
the year ended October 31, 2008 in conformity with accounting principles
generally accepted in the United States of America.
The
accompanying consolidated financial statements have been prepared assuming that
the Company will continue as a going concern. As discussed in Note 2 to the
financial statements, the Company has suffered recurring net losses and negative
cash flows from operations. These matters raise substantial doubt about the
Company’s ability to continue as a going concern. Management’s plans regarding
these matters are also described in Note 2. The accompanying consolidated
financial statements do not include any adjustments that might result from the
outcome of this uncertainty.
(Signed)
“BDO Canada
LLP”
Chartered
Accountants, Licensed Public Accountants
Toronto,
Ontario
July 29,
2009
F-2
|
CardioGenics
Holdings Inc.
(A
Development Stage Company)
Consolidated
Balance Sheets
|
|
October 31,
|
October 31,
|
|||||||
|
2010
|
2009
|
|||||||
|
Assets
|
||||||||
|
Current
|
||||||||
|
Cash
and Cash Equivalents
|
$ | 1,844,752 | $ | 2,388,516 | ||||
|
Share
Subscriptions Receivable
|
115,000 | — | ||||||
|
Deposits
and Prepaid Expenses
|
89,774 | 11,996 | ||||||
|
Refundable
Taxes Receivable
|
21,959 | 14,878 | ||||||
|
Government
Grants and Investment Tax Credits Receivable
|
156,482 | 175,554 | ||||||
| 2,227,967 | 2,590,944 | |||||||
|
Property
and Equipment, net
|
87,465 | 54,338 | ||||||
|
Patents,
net
|
170,703 | 241,980 | ||||||
| 258,168 | 296,318 | |||||||
| $ | 2,486,135 | $ | 2,887,262 | |||||
|
Liabilities
and Stockholders' Equity (Deficit)
|
||||||||
|
Current
Liabilities
|
||||||||
|
Accounts
Payable and Accrued Expenses
|
$ | 523,155 | $ | 651,037 | ||||
|
Due
to Director
|
15,149 | 147,102 | ||||||
|
Liabilities
of Discontinued Operations
|
— | 100,000 | ||||||
|
Debentures
Payable
|
— | 25,000 | ||||||
|
Current
Portion of Capital Lease Obligation
|
20,992 | — | ||||||
| 559,296 | 923,139 | |||||||
|
Long
Term Liabilities
|
||||||||
|
Capital
Lease Obligation, net of current portion
|
20,881 | — | ||||||
| 20,881 | — | |||||||
|
Mandatorily
redeemable Class B common stock; par value $.00001 per
share:
|
||||||||
|
400,000
shares designated as series 2; 381,749 shares issued and
outstanding
|
4 | 4 | ||||||
|
40,000
shares designated as series 3; 21,500 shares issued and
outstanding
|
— | — | ||||||
| 4 | 4 | |||||||
|
Commitments
and contingencies
|
||||||||
|
Stockholders'
Equity (Deficit)
|
||||||||
|
Preferred
stock; par value $.0001 per share,
5,000,000
shares authorized, none issued
|
— | — | ||||||
|
Common
stock; par value $.00001 per share;
65,000,000
shares authorized,
28,620,257
and 21,767,100 common shares and
24,388,904
and 27,665,540 exchangeable shares issued and
outstanding
as at October 31, 2010 and 2009, respectively
|
514 | 495 | ||||||
|
Additional
paid-in capital
|
37,441,728 | 35,543,722 | ||||||
|
Deficit
accumulated during development stage
|
(35,006,558 | ) | (32,982,286 | ) | ||||
|
Accumulated
other comprehensive loss
|
(237,508 | ) | (319,815 | ) | ||||
|
Total
CardioGenics Holdings Inc. Stockholders’ Equity
|
2,198,176 | 2,242,116 | ||||||
|
Non-Controlling
Interest
|
(292,222 | ) | (277,997 | ) | ||||
|
Total
Equity
|
1,905,954 | 1,964,119 | ||||||
|
Total
liabilities and stockholders' equity
|
$ | 2,486,135 | $ | 2,887,262 | ||||
See notes
to consolidated financial statements.
F-3
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Consolidated
Statements of Operations
|
|
For
the Years Ended October 31, 2010 and 2009 and
|
|
Cumulative from November 20, 1997 (Date of
Inception) to October 31,
2010
|
|
Cumulative
|
||||||||||||
|
From
|
||||||||||||
|
November 20,
|
||||||||||||
|
1997
|
||||||||||||
|
(Date of
|
||||||||||||
|
For
the Years Ended
|
Inception)
to
|
|||||||||||
|
October 31,
|
October
31,
|
|||||||||||
|
2010
|
2009
|
2010
|
||||||||||
|
Operating
Expenses
|
||||||||||||
|
Depreciation
and amortization of Property and Equipment
|
$ | 20,932 | $ | 26,157 | $ | 181,040 | ||||||
|
Amortization
of Patent Application Costs
|
3,023 | 4,181 | 7,204 | |||||||||
|
Write-off
of Patent Application Costs
|
105,345 | 23,803 | 159,076 | |||||||||
|
General
and Administrative
|
1,160,509 | 1,597,010 | 4,189,730 | |||||||||
|
Write-off
of Goodwill
|
— | 12,780,214 | 12,780,214 | |||||||||
|
Research
and Product Development, Net of Investment Tax Credits
|
692,036 | 1,572,337 | 3,287,751 | |||||||||
|
Total
operating expenses and operating loss
|
1,981,845 | 16,003,702 | 20,605,015 | |||||||||
|
Other
Expenses (Income)
|
||||||||||||
|
Interest
Expense and Bank Charges (Net)
|
30,166 | 571,840 | 2,116,501 | |||||||||
|
Loss
on Change in Value of Derivative Liability
|
— | 12,421,023 | 12,421,023 | |||||||||
|
Loss
(Gain) on Foreign Exchange Transactions
|
104,182 | (184,389 | ) | 118,528 | ||||||||
|
Total
other expenses (income)
|
134,348 | 12,808,474 | 14,656,052 | |||||||||
|
Loss
from Continuing Operations
|
(2,116,193 | ) | (28,812,176 | ) | (35,261,067 | ) | ||||||
|
Discontinued
Operations
|
||||||||||||
|
Gain
on Sale of Subsidiary
|
90,051 | — | 90,051 | |||||||||
|
Loss
from Discontinued Operations
|
(12,355 | ) | (115,407 | ) | (127,762 | ) | ||||||
|
Net
Loss
|
(2,038,497 | ) | (28,927,583 | ) | (35,298,778 | ) | ||||||
|
Net
Loss Attributed to Non-Controlling Interest
|
(14,225 | ) | (211,670 | ) | (292,220 | ) | ||||||
|
Net
Loss Attributed to CardioGenics Holdings Inc.
|
$ | (2,024,272 | ) | $ | (28,715,913 | ) | (35,006,558 | ) | ||||
|
Basic
and Fully Diluted Net Loss per Common Share
|
(0.04 | ) | (0.95 | ) | ||||||||
|
Weighted-average
number of Common Shares
|
49,624,916 | 30,385,058 | ||||||||||
See notes
to consolidated financial statements.
F-4
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Consolidated
Statements of Stockholders' Equity (Deficit)
|
|
Cumulative from November 20, 1997 (Date of
Inception) to October 31,
2010
|
|
Deficit
|
||||||||||||||||||||||||||||
|
Accumulated
|
||||||||||||||||||||||||||||
|
During
|
Accumulated
|
Total
|
||||||||||||||||||||||||||
|
Additional
|
the
|
Other
|
Stockholders'
|
|||||||||||||||||||||||||
|
Common Stock
|
Paid-in
|
Development
|
Comprehensive
|
Noncontrolling
|
Equity
|
|||||||||||||||||||||||
|
Shares
|
Amount
|
Capital
|
Stage
|
Income (Loss)
|
Interest
|
(Deficiency)
|
||||||||||||||||||||||
|
Issuance
of common shares for cash November 1998
|
1,592,732 | $ | 16 | $ | (15 | ) | $ | 1 | ||||||||||||||||||||
|
Issuance
of common shares for cash December 1998, $.00
|
796,366 | 8 | 35,028 | 35,036 | ||||||||||||||||||||||||
|
Issuance
of common shares for cash March 1998, $.00
|
551,611 | 6 | 24,442 | 24,448 | ||||||||||||||||||||||||
|
Issuance
of common shares for cash April 1998, $.00
|
12,986,611 | 130 | 5,573 | 5,703 | ||||||||||||||||||||||||
|
Issuance
of common shares for cash May 1998, $.01
|
210,249 | 2 | 17,297 | 17,299 | ||||||||||||||||||||||||
|
Issuance
of common shares for cash August 1998, $.00
|
2,787,281 | 28 | 200 | 228 | ||||||||||||||||||||||||
|
Issuance
of common shares for cash September 1998, $.01
|
84,100 | 1 | 6,570 | 6,571 | ||||||||||||||||||||||||
|
Issuance
of common shares for cash October 1998, $.01
|
31,949 | — | 2,500 | 2,500 | ||||||||||||||||||||||||
|
Comprehensive
Income (Loss)
|
||||||||||||||||||||||||||||
|
Net
Loss
|
$ | (81,208 | ) | (81,208 | ) | |||||||||||||||||||||||
|
Other
Comprehensive Income (Loss)
|
||||||||||||||||||||||||||||
|
Currency
Translation Adjustment
|
$ | (2,096 | ) | (2,096 | ) | |||||||||||||||||||||||
|
Total
Comprehensive Income (Loss)
|
(81,208 | ) | (2,096 | ) | (83,304 | ) | ||||||||||||||||||||||
|
Balance
at October 31, 1998
|
19,040,899 | $ | 191 | $ | 91,595 | $ | (81,208 | ) | $ | (2,096 | ) | $ | — | $ | 8,482 | |||||||||||||
|
Deficit
|
||||||||||||||||||||||||||||
|
Accumulated
|
||||||||||||||||||||||||||||
|
During
|
Accumulated
|
Total
|
||||||||||||||||||||||||||
|
Additional
|
the
|
Other
|
Stockholders'
|
|||||||||||||||||||||||||
|
Common Stock
|
Paid-in
|
Development
|
Comprehensive
|
Noncontrolling
|
Equity
|
|||||||||||||||||||||||
|
Shares
|
Amount
|
Capital
|
Stage
|
Income (Loss)
|
Interest
|
(Deficiency)
|
||||||||||||||||||||||
|
Balance
November 1, 1998
|
19,040,899 | $ | 191 | $ | 91,595 | $ | (81,208 | ) | $ | (2,096 | ) | $ | — | $ | 8,482 | |||||||||||||
|
Issuance
of common shares for cash November 1998, $.01
|
32,066 | — | 2,500 | 2,500 | ||||||||||||||||||||||||
|
Issuance
of common shares for cash February 1999, $.01
|
159,273 | 2 | 14,287 | 14,289 | ||||||||||||||||||||||||
|
Commission
paid on issuance of common stock for cash February 1999
|
(935 | ) | (935 | ) | ||||||||||||||||||||||||
|
Issuance
of common shares for cash March 1999, $.01
|
278,728 | 3 | 24,707 | 24,710 | ||||||||||||||||||||||||
|
Commission
paid on issuance of common stock for cash March 1999
|
(1,647 | ) | (1,647 | ) | ||||||||||||||||||||||||
|
Issuance
of common shares for cash to minority shareholders April 1999,
$.01
|
— | — | 10,707 | 10,707 | ||||||||||||||||||||||||
|
Commission
paid on issuance of common stock for cash April 1999
|
(627 | ) | (627 | ) | ||||||||||||||||||||||||
|
Issuance
of common shares for cash April 1999, $.01
|
39,818 | — | 3,814 | 3,814 | ||||||||||||||||||||||||
|
Commission
paid on issuance of common stock for cash April 1999
|
(314 | ) | (314 | ) | ||||||||||||||||||||||||
|
Issuance
of common shares for cash July 1999, $.01
|
119,455 | 1 | 10,073 | 10,074 | ||||||||||||||||||||||||
|
Issuance
of common shares for cash August 1999, $.01
|
119,455 | 1 | 10,045 | 10,046 | ||||||||||||||||||||||||
|
Net
loss attributable to noncontrolling interest
|
(604 | ) | (604 | ) | ||||||||||||||||||||||||
|
Comprehensive
Income (Loss)
|
||||||||||||||||||||||||||||
|
Net
Loss
|
(100,141 | ) | (100,141 | ) | ||||||||||||||||||||||||
|
Other
Comprehensive Income (Loss)
|
||||||||||||||||||||||||||||
|
Currency
Translation Adjustment
|
(3,489 | ) | (3,489 | ) | ||||||||||||||||||||||||
|
Total
Comprehensive Income (Loss)
|
(100,141 | ) | (3,489 | ) | (103,630 | ) | ||||||||||||||||||||||
|
Balance
at October 31, 1999
|
19,789,694 | $ | 198 | $ | 164,205 | $ | (181,349 | ) | $ | (5,585 | ) | $ | (604 | ) | $ | (23,135 | ) | |||||||||||
See notes
to consolidated financial statements.
F-5
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Consolidated
Statements of Stockholders' Equity (Deficit)
|
|
Cumulative from November 20, 1997 (Date of
Inception) to October 31,
2010
|
|
Deficit
|
||||||||||||||||||||||||||||
|
Accumulated
|
||||||||||||||||||||||||||||
|
During
|
Accumulated
|
Total
|
||||||||||||||||||||||||||
|
Additional
|
the
|
Other
|
Stockholders'
|
|||||||||||||||||||||||||
|
Common Stock
|
Paid-in
|
Development
|
Comprehensive
|
Noncontrolling
|
Equity
|
|||||||||||||||||||||||
|
Shares
|
Amount
|
Capital
|
Stage
|
Income (Loss)
|
Interest
|
(Deficiency)
|
||||||||||||||||||||||
|
Balance
November 1, 1999
|
19,789,694 | $ | 198 | $ | 164,205 | $ | (181,349 | ) | $ | (5,585 | ) | $ | (604 | ) | $ | (23,135 | ) | |||||||||||
|
Issuance
of common shares for cash November 1999, $.03
|
318,546 | 3 | 99,997 | 100,000 | ||||||||||||||||||||||||
|
Issuance
of common shares for minority shareholders as employee compensation
December 1999, $.03
|
— | — | 3,396 | 3,396 | ||||||||||||||||||||||||
|
Issuance
of common shares for cash March 2000, $.03
|
167,237 | 2 | 43,124 | 43,126 | ||||||||||||||||||||||||
|
Issuance
of common shares for minority shareholders for cash March, 2000,
$.03
|
— | — | 25,330 | 25,330 | ||||||||||||||||||||||||
|
Issuance
of common shares for cash April 2000, $.03
|
23,891 | — | 6,128 | 6,128 | ||||||||||||||||||||||||
|
Loan
Payable plus interest exchanged for shares July 2000, $.03
|
356,772 | 4 | 111,996 | 112,000 | ||||||||||||||||||||||||
|
Issuance
of common shares for minority shareholders as employee compensation
October 2000, $.03
|
— | — | 6,611 | 6,611 | ||||||||||||||||||||||||
|
Issuance
of stock options in exchange for services rendered October
2000
|
— | 11,570 | 11,570 | |||||||||||||||||||||||||
|
Net
loss attributable to noncontrolling interest
|
(1,840 | ) | (1,840 | ) | ||||||||||||||||||||||||
|
Comprehensive
Income (Loss)
|
||||||||||||||||||||||||||||
|
Net
Loss
|
(152,525 | ) | (152,525 | ) | ||||||||||||||||||||||||
|
Other
Comprehensive Income (Loss)
|
||||||||||||||||||||||||||||
|
Currency
Translation Adjustment
|
921 | 921 | ||||||||||||||||||||||||||
|
Total
Comprehensive Income (Loss)
|
(152,525 | ) | 921 | (151,604 | ) | |||||||||||||||||||||||
|
Balance
at October 31, 2000
|
20,656,140 | $ | 207 | $ | 472,357 | $ | (333,874 | ) | $ | (4,664 | ) | $ | (2,444 | ) | $ | 131,582 | ||||||||||||
|
Deficit
|
||||||||||||||||||||||||||||
|
Accumulated
|
||||||||||||||||||||||||||||
|
During
|
Accumulated
|
Total
|
||||||||||||||||||||||||||
|
Additional
|
the
|
Other
|
Stockholders'
|
|||||||||||||||||||||||||
|
Common Stock
|
Paid-in
|
Development
|
Comprehensive
|
Noncontrolling
|
Equity
|
|||||||||||||||||||||||
|
Shares
|
Amount
|
Capital
|
Stage
|
Income (Loss)
|
Interest
|
(Deficiency)
|
||||||||||||||||||||||
|
Balance
November 1, 2000
|
20,656,140 | $ | 207 | $ | 472,357 | $ | (333,874 | ) | $ | (4,664 | ) | $ | (2,444 | ) | $ | 131,582 | ||||||||||||
|
Issuance
of common shares as
|
||||||||||||||||||||||||||||
|
employee
compensation
|
||||||||||||||||||||||||||||
|
October
2001, $.03
|
2,410 | — | 925 | 925 | ||||||||||||||||||||||||
|
Issuance
of common share for minority
|
||||||||||||||||||||||||||||
|
shareholders
as employee compensation
|
||||||||||||||||||||||||||||
|
October
2001, $.03
|
— | — | 6,169 | 6,169 | ||||||||||||||||||||||||
|
Issuance
of stock options in
|
||||||||||||||||||||||||||||
|
exchange
for services rendered
|
||||||||||||||||||||||||||||
|
October
2001
|
— | — | 22,269 | 22,269 | ||||||||||||||||||||||||
|
Net
loss attributable to noncontrolling interest
|
(1,500 | ) | (1,500 | ) | ||||||||||||||||||||||||
|
Comprehensive
Income (Loss)
|
||||||||||||||||||||||||||||
|
Net
Loss
|
(114,761 | ) | (114,761 | ) | ||||||||||||||||||||||||
|
Other
Comprehensive Income (Loss)
|
||||||||||||||||||||||||||||
|
Currency
Translation Adjustment
|
(10,528 | ) | (10,528 | ) | ||||||||||||||||||||||||
|
Total
Comprehensive Income (Loss)
|
(114,761 | ) | (10,528 | ) | (125,289 | ) | ||||||||||||||||||||||
|
Balance
at October 31, 2001
|
20,658,550 | $ | 207 | $ | 501,720 | $ | (448,635 | ) | $ | (15,192 | ) | $ | (3,944 | ) | $ | 34,156 | ||||||||||||
See notes
to consolidated financial statements.
F-6
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Consolidated
Statements of Stockholders' Equity (Deficit)
|
|
Cumulative from November 20, 1997 (Date of
Inception) to October 31,
2010
|
|
Deficit
|
||||||||||||||||||||||||||||
|
Accumulated
|
||||||||||||||||||||||||||||
|
During
|
Accumulated
|
Total
|
||||||||||||||||||||||||||
|
Additional
|
the
|
Other
|
Stockholders'
|
|||||||||||||||||||||||||
|
Common Stock
|
Paid-in
|
Development
|
Comprehensive
|
Noncontrolling
|
Equity
|
|||||||||||||||||||||||
|
Shares
|
Amount
|
Capital
|
Stage
|
Income (Loss)
|
Interest
|
(Deficiency)
|
||||||||||||||||||||||
|
Balance
November 1, 2001
|
20,658,550 | $ | 207 | $ | 501,720 | $ | (448,635 | ) | $ | (15,192 | ) | $ | (3,944 | ) | $ | 34,156 | ||||||||||||
|
Issuance
of common shares for cash June 2002, $.03
|
1,051,211 | 11 | 319,011 | 319,022 | ||||||||||||||||||||||||
|
Issuance
of common shares to minority shareholders for cash July 2002,
$.03
|
— | — | 3,235 | 3,235 | ||||||||||||||||||||||||
|
Issuance
of common shares for cash September 2002, $.03
|
20,957 | — | 6,345 | 6,345 | ||||||||||||||||||||||||
|
Issuance
of common shares for minority shareholders as employee compensation
October 2002, $.03
|
— | — | 9,505 | 9,505 | ||||||||||||||||||||||||
|
Issuance
of stock options in exchange for services rendered October
2002
|
— | — | 70,518 | 70,518 | ||||||||||||||||||||||||
|
Net
loss attributable to noncontrolling interest
|
(2,243 | ) | (2,243 | ) | ||||||||||||||||||||||||
|
Comprehensive
Income (Loss)
|
||||||||||||||||||||||||||||
|
Net
Loss
|
(156,214 | ) | (156,214 | ) | ||||||||||||||||||||||||
|
Other
Comprehensive Income (Loss)
|
||||||||||||||||||||||||||||
|
Currency
Translation Adjustment
|
(11,506 | ) | (11,506 | ) | ||||||||||||||||||||||||
|
Total
Comprehensive Income (Loss)
|
(156,214 | ) | (11,506 | ) | (167,720 | ) | ||||||||||||||||||||||
|
Balance
at October 31, 2002
|
21,730,718 | $ | 218 | $ | 910,334 | $ | (604,849 | ) | $ | (26,698 | ) | $ | (6,187 | ) | $ | 272,818 | ||||||||||||
|
Deficit
|
||||||||||||||||||||||||||||
|
Accumulated
|
||||||||||||||||||||||||||||
|
During
|
Accumulated
|
Total
|
||||||||||||||||||||||||||
|
Additional
|
the
|
Other
|
Stockholders'
|
|||||||||||||||||||||||||
|
Common Stock
|
Paid-in
|
Development
|
Comprehensive
|
Noncontrolling
|
Equity
|
|||||||||||||||||||||||
|
Shares
|
Amount
|
Capital
|
Stage
|
Income (Loss)
|
Interest
|
(Deficiency)
|
||||||||||||||||||||||
|
Balance
November 1, 2002
|
21,730,718 | $ | 218 | $ | 910,334 | $ | (604,849 | ) | $ | (26,698 | ) | $ | (6,187 | ) | $ | 272,818 | ||||||||||||
|
Issuance
of common shares for cash May 2003, $.03
|
28,292 | — | 9,871 | 9,871 | ||||||||||||||||||||||||
|
Issuance
of common shares for minority shareholders for cash May 2003
$.03
|
— | — | 10,967 | 10,967 | ||||||||||||||||||||||||
|
Issuance
of warrants in conjunction with convertible debentures September
2003
|
358,406 | 358,406 | ||||||||||||||||||||||||||
|
Issuance
of common shares as employee compensation October 2003,
$.04
|
56,584 | 1 | 20,421 | 20,422 | ||||||||||||||||||||||||
|
Issuance
of common shares for minority shareholders as employee compensation
October 2003, $.04
|
— | — | 7,564 | 7,564 | ||||||||||||||||||||||||
|
Issuance
of stock options in exchange for services rendered October
2003
|
— | — | 23,580 | 23,580 | ||||||||||||||||||||||||
|
Net
loss attributable to noncontrolling interest
|
(3,825 | ) | (3,825 | ) | ||||||||||||||||||||||||
|
Comprehensive
Income (Loss)
|
||||||||||||||||||||||||||||
|
Net
Loss
|
(228,993 | ) | (228,993 | ) | ||||||||||||||||||||||||
|
Other
Comprehensive Income (Loss)
|
||||||||||||||||||||||||||||
|
Currency
Translation Adjustment
|
42,957 | 42,957 | ||||||||||||||||||||||||||
|
Total
Comprehensive Income (Loss)
|
(228,993 | ) | 42,957 | (186,036 | ) | |||||||||||||||||||||||
|
Balance
at October 31, 2003
|
$ | 21,815,594 | $ | 219 | $ | 1,341,143 | $ | (833,842 | ) | $ | 16,259 | $ | (10,012 | ) | $ | 513,767 | ||||||||||||
See notes
to consolidated financial statements.
F-7
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Consolidated
Statements of Stockholders' Equity (Deficit)
|
|
Cumulative from November 20, 1997 (Date of
Inception) to October 31,
2010
|
|
Deficit
|
||||||||||||||||||||||||||||
|
Accumulated
|
||||||||||||||||||||||||||||
|
During
|
Accumulated
|
Total
|
||||||||||||||||||||||||||
|
Additional
|
the
|
Other
|
Stockholders'
|
|||||||||||||||||||||||||
|
Common Stock
|
Paid-in
|
Development
|
Comprehensive
|
Noncontrolling
|
Equity
|
|||||||||||||||||||||||
|
Shares
|
Amount
|
Capital
|
Stage
|
Income (Loss)
|
Interest
|
(Deficiency)
|
||||||||||||||||||||||
|
Balance
November 1, 2003
|
21,815,594 | $ | 219 | $ | 1,341,143 | $ | (833,842 | ) | $ | 16,259 | $ | (10,012 | ) | $ | 513,767 | |||||||||||||
|
Issuance
of warrants in conjunction with convertible debentures September
2004
|
152,628 | 152,628 | ||||||||||||||||||||||||||
|
Issuance
of common shares as employee compensation October 2004,
$.04
|
123,646 | 1 | 47,316 | 47,317 | ||||||||||||||||||||||||
|
Issuance
of common shares as directors' compensation October 2004,
$.04
|
157,177 | 2 | 60,147 | 60,149 | ||||||||||||||||||||||||
|
Issuance
of stock options in exchange for services rendered October
2004
|
— | — | 27,669 | 27,669 | ||||||||||||||||||||||||
|
Issuance
of options to directors and committee chairmen for services rendered in
October 2004
|
54,582 | 54,582 | ||||||||||||||||||||||||||
|
Net
loss attributable to noncontrolling interest
|
(9,774 | ) | (9,774 | ) | ||||||||||||||||||||||||
|
Comprehensive
Income (Loss)
|
||||||||||||||||||||||||||||
|
Net
Loss
|
(592,706 | ) | (592,706 | ) | ||||||||||||||||||||||||
|
Other
Comprehensive Income (Loss)
|
||||||||||||||||||||||||||||
|
Currency
Translation Adjustment
|
(6,136 | ) | (6,136 | ) | ||||||||||||||||||||||||
|
Total
Comprehensive Income (Loss)
|
(592,706 | ) | (6,136 | ) | (598,842 | ) | ||||||||||||||||||||||
|
Balance
at October 31, 2004
|
22,096,417 | $ | 222 | $ | 1,683,485 | $ | (1,426,548 | ) | $ | 10,123 | $ | (19,786 | ) | $ | 247,496 | |||||||||||||
See notes
to consolidated financial statements.
F-8
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Consolidated
Statements of Stockholders' Equity (Deficit)
|
|
Cumulative from November 20, 1997 (Date of
Inception) to October 31,
2010
|
|
Deficit
|
||||||||||||||||||||||||||||
|
Accumulated
|
||||||||||||||||||||||||||||
|
During
|
Accumulated
|
Total
|
||||||||||||||||||||||||||
|
Additional
|
the
|
Other
|
Stockholders'
|
|||||||||||||||||||||||||
|
Common Stock
|
Paid-in
|
Development
|
Comprehensive
|
Noncontrolling
|
Equity
|
|||||||||||||||||||||||
|
Shares
|
Amount
|
Capital
|
Stage
|
Income (Loss)
|
Interest
|
(Deficiency)
|
||||||||||||||||||||||
|
Balance
November 1, 2004
|
22,096,417 | $ | 222 | $ | 1,683,485 | $ | (1,426,548 | ) | $ | 10,123 | $ | (19,786 | ) | $ | 247,496 | |||||||||||||
|
Issuance
of common shares as employee compensation November 2004,
$.04
|
9,431 | — | 3,760 | 3,760 | ||||||||||||||||||||||||
|
Issuance
of common shares as employee compensation December 2004,
$.04
|
9,431 | — | 3,692 | 3,692 | ||||||||||||||||||||||||
|
Issuance
of common shares as employee compensation January 2005,
$.04
|
9,431 | — | 3,674 | 3,674 | ||||||||||||||||||||||||
|
Issuance
of common shares as employee compensation February 2005,
$.04
|
9,431 | — | 3,629 | 3,629 | ||||||||||||||||||||||||
|
Issuance
of common shares as employee compensation March 2005, $.04
|
9,431 | — | 3,701 | 3,701 | ||||||||||||||||||||||||
|
Issuance
of common shares as employee compensation April 2005, $.04
|
9,431 | — | 3,641 | 3,641 | ||||||||||||||||||||||||
|
Issuance
of common shares as employee compensation May 2005, $.04
|
9,431 | — | 3,584 | 3,584 | ||||||||||||||||||||||||
|
Issuance
of common shares as employee compensation June 2005, $.04
|
9,431 | — | 3,628 | 3,628 | ||||||||||||||||||||||||
|
Issuance
of common shares as employee compensation July 2005, $.04
|
9,431 | — | 3,680 | 3,680 | ||||||||||||||||||||||||
|
Issuance
of common shares as employee compensation August 2005,
$.04
|
9,431 | — | 3,737 | 3,737 | ||||||||||||||||||||||||
|
Issuance
of common shares as employee compensation September 2005,
$.04
|
9,431 | — | 3,821 | 3,821 | ||||||||||||||||||||||||
|
Issuance
of common shares as employee compensation October 2005,
$.04
|
9,431 | — | 3,822 | 3,822 | ||||||||||||||||||||||||
|
Issuance
of stock options in exchange for services rendered October
2005
|
— | — | 33,973 | 33,973 | ||||||||||||||||||||||||
|
Net
loss attributtable to noncontrolling interest
|
(11,195 | ) | (11,195 | ) | ||||||||||||||||||||||||
|
Comprehensive
Income (Loss)
|
||||||||||||||||||||||||||||
|
Net
Loss
|
(682,408 | ) | (682,408 | ) | ||||||||||||||||||||||||
|
Other
Comprehensive Income (Loss)
|
||||||||||||||||||||||||||||
|
Currency
Translation Adjustment
|
(13,288 | ) | (13,288 | ) | ||||||||||||||||||||||||
|
Total
Comprehensive Income (Loss)
|
(682,408 | ) | (13,288 | ) | (695,696 | ) | ||||||||||||||||||||||
|
Balance
at October 31, 2005
|
22,209,589 | $ | 222 | $ | 1,761,827 | $ | (2,108,956 | ) | $ | (3,165 | ) | $ | (30,981 | ) | $ | (381,053 | ) | |||||||||||
|
Deficit
|
||||||||||||||||||||||||||||
|
Accumulated
|
||||||||||||||||||||||||||||
|
During
|
Accumulated
|
Total
|
||||||||||||||||||||||||||
|
Additional
|
the
|
Other
|
Stockholders'
|
|||||||||||||||||||||||||
|
Common Stock
|
Paid-in
|
Development
|
Comprehensive
|
Noncontrolling
|
Equity
|
|||||||||||||||||||||||
|
Shares
|
Amount
|
Capital
|
Stage
|
Income (Loss)
|
Interest
|
(Deficiency)
|
||||||||||||||||||||||
|
Balance
November 1, 2005
|
22,209,589 | $ | 222 | $ | 1,761,827 | $ | (2,108,956 | ) | $ | (3,165 | ) | $ | (30,981 | ) | $ | (381,053 | ) | |||||||||||
|
Issuance
of common shares as employee compensation November 2005,
$.04
|
10,478 | — | 4,232 | 4,232 | ||||||||||||||||||||||||
|
Issuance
of common shares in exchange for services rendered December 2005,
$.04
|
10,478 | — | 4,305 | 4,305 | ||||||||||||||||||||||||
|
Issuance
of common shares in exchange for services rendered January 2006,
$.04
|
10,478 | — | 4,321 | 4,321 | ||||||||||||||||||||||||
|
Issuance
of stock options in exchange for services rendered October
2006
|
— | — | 2,658 | 2,658 | ||||||||||||||||||||||||
|
Net
loss attributable to noncontrolling interest
|
(8,561 | ) | (8,561 | ) | ||||||||||||||||||||||||
|
Comprehensive
Income (Loss)
|
||||||||||||||||||||||||||||
|
Net
Loss
|
(522,532 | ) | (522,532 | ) | ||||||||||||||||||||||||
|
Other
Comprehensive Income (Loss)
|
||||||||||||||||||||||||||||
|
Currency
Translation Adjustment
|
(25,688 | ) | (25,688 | ) | ||||||||||||||||||||||||
|
Total
Comprehensive Income (Loss)
|
(522,532 | ) | (25,688 | ) | (548,220 | ) | ||||||||||||||||||||||
|
Balance
at October 31, 2006
|
22,241,023 | $ | 222 | $ | 1,777,343 | $ | (2,631,488 | ) | $ | (28,853 | ) | $ | (39,542 | ) | $ | (922,318 | ) | |||||||||||
See notes
to consolidated financial statements.
F-9
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Consolidated
Statements of Stockholders' Equity (Deficit)
|
|
Cumulative from November 20, 1997 (Date of
Inception) to October 31,
2010
|
|
Deficit
|
||||||||||||||||||||||||||||
|
Accumulated
|
||||||||||||||||||||||||||||
|
During
|
Accumulated
|
Total
|
||||||||||||||||||||||||||
|
Additional
|
the
|
Other
|
Stockholders'
|
|||||||||||||||||||||||||
|
Common Stock
|
Paid-in
|
Development
|
Comprehensive
|
Noncontrolling
|
Equity
|
|||||||||||||||||||||||
|
Shares
|
Amount
|
Capital
|
Stage
|
Income (Loss)
|
Interest
|
(Deficiency)
|
||||||||||||||||||||||
|
Balance
November 1, 2006
|
22,241,023 | $ | 222 | $ | 1,777,343 | $ | (2,631,488 | ) | $ | (28,853 | ) | $ | (39,542 | ) | $ | (922,318 | ) | |||||||||||
|
Incremental
increase in fair value of warrants in conjunction with re-structuring of
debentures, April 2007
|
44,096 | 44,096 | ||||||||||||||||||||||||||
|
Net
loss attributable to noncontrolling
interest
|
(4,972 | ) | (4,972 | ) | ||||||||||||||||||||||||
|
Comprehensive
Income (Loss)
|
||||||||||||||||||||||||||||
|
Net
Loss
|
(303,477 | ) | (303,477 | ) | ||||||||||||||||||||||||
|
Other
Comprehensive Income (Loss)
|
||||||||||||||||||||||||||||
|
Currency
Translation Adjustment
|
(184,432 | ) | (184,432 | ) | ||||||||||||||||||||||||
|
Total
Comprehensive Income (Loss)
|
(303,477 | ) | (184,432 | ) | (487,909 | ) | ||||||||||||||||||||||
|
Balance
at October 31, 2007
|
22,241,023 | $ | 222 | $ | 1,821,439 | $ | (2,934,965 | ) | $ | (213,285 | ) | $ | (44,514 | ) | $ | (1,371,103 | ) | |||||||||||
|
Deficit
|
||||||||||||||||||||||||||||
|
Accumulated
|
||||||||||||||||||||||||||||
|
During
|
Accumulated
|
Total
|
||||||||||||||||||||||||||
|
Additional
|
the
|
Other
|
Stockholders'
|
|||||||||||||||||||||||||
|
Common Stock
|
Paid-in
|
Development
|
Comprehensive
|
Noncontrolling
|
Equity
|
|||||||||||||||||||||||
|
Shares
|
Amount
|
Capital
|
Stage
|
Income (Loss)
|
Interest
|
(Deficiency)
|
||||||||||||||||||||||
|
Balance
November 1, 2007
|
22,241,023 | $ | 222 | $ | 1,821,439 | $ | (2,934,965 | ) | $ | (213,285 | ) | $ | (44,514 | ) | $ | (1,371,103 | ) | |||||||||||
|
Issuance
of warrants in conjunction with re-structuring of debentures October
2008
|
231,580 | 231,580 | ||||||||||||||||||||||||||
|
Net
loss attributable to noncontrolling interest
|
(21,813 | ) | (21,813 | ) | ||||||||||||||||||||||||
|
Comprehensive
Income (Loss)
|
||||||||||||||||||||||||||||
|
Net
Loss
|
(1,331,408 | ) | (1,331,408 | ) | ||||||||||||||||||||||||
|
Other
Comprehensive Income (Loss)
|
||||||||||||||||||||||||||||
|
Currency
Translation Adjustment
|
422,766 | 422,766 | ||||||||||||||||||||||||||
|
Total
Comprehensive Income (Loss)
|
(1,331,408 | ) | 422,766 | (908,642 | ) | |||||||||||||||||||||||
|
Balance
at October 31, 2008
|
22,241,023 | $ | 222 | $ | 2,053,019 | $ | (4,266,373 | ) | $ | 209,481 | $ | (66,327 | ) | $ | (2,069,978 | ) | ||||||||||||
See notes
to consolidated financial statements.
F-10
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Consolidated
Statements of Stockholders' Equity (Deficit)
|
|
Cumulative from November 20, 1997 (Date of
Inception) to October 31,
2010
|
|
Deficit
|
||||||||||||||||||||||||||||
|
Accumulated
|
||||||||||||||||||||||||||||
|
During
|
Accumulated
|
Total
|
||||||||||||||||||||||||||
|
Additional
|
the
|
Other
|
Stockholders'
|
|||||||||||||||||||||||||
|
Common Stock
|
Paid-in
|
Development
|
Comprehensive
|
Noncontrolling
|
Equity
|
|||||||||||||||||||||||
|
Shares
|
Amount
|
Capital
|
Stage
|
Income (Loss)
|
Interest
|
(Deficiency)
|
||||||||||||||||||||||
|
Balance
November 1, 2008
|
22,241,023 | $ | 222 | $ | 2,053,019 | $ | (4,266,373 | ) | $ | 209,481 | $ | (66,327 | ) | $ | (2,069,978 | ) | ||||||||||||
|
Issuance
of common shares as payment of debenture interest, January 2009, $0.05 per
share
|
495,094 | 5 | 236,238 | 236,243 | ||||||||||||||||||||||||
|
Issuance
of common shares on exercise of options, April 2009
|
570,980 | 6 | 22 | 28 | ||||||||||||||||||||||||
|
Issuance
of common shares as employee compensation for the years 2001 to 2008, May
2009, $0.04 per share
|
3,153,878 | 32 | 1,298,753 | 1,298,785 | ||||||||||||||||||||||||
|
Issuance
of common shares to directors, pursuant to debenture financing of January
2009, May 2009, $0.04 per share
|
928,394 | 9 | 382,521 | 382,530 | ||||||||||||||||||||||||
|
Issuance
of common shares in exchange for services rendered, June 2009, $0.04 per
share
|
5,023 | — | 2,062 | 2,062 | ||||||||||||||||||||||||
|
Issuance
of common shares for cash June 2009, $0.04 per share
|
24,090 | — | 8,602 | 8,602 | ||||||||||||||||||||||||
|
Issuance
of common shares in exchange for services rendered, July 2009, $0.04 per
share
|
47,153 | — | 20,250 | 20,250 | ||||||||||||||||||||||||
|
Issuance
of common shares as payment of director compensation, for the years 2004
through 2009, July 2009, $0.04 per share
|
241,005 | 2 | 103,498 | 103,500 | ||||||||||||||||||||||||
|
Issuance
of common shares as employee compensation pursuant to reverse merger
transaction, July 2009, $0.04 per share
|
1,173,592 | 12 | 503,988 | 504,000 | ||||||||||||||||||||||||
|
Issuance
of common shares to retire debentures, July 2009, $0.03 per
share
|
3,346,028 | 34 | 997,538 | 997,572 | ||||||||||||||||||||||||
|
Issuance
of common shares as payment of debenture interest, January 2009, July
2009, $0.05 per share
|
855,712 | 9 | 418,659 | 418,668 | ||||||||||||||||||||||||
|
Issuance
of common shares to retire director's loan, July 2009 $0.04 per
share
|
2,377,813 | 24 | 884,976 | 885,000 | ||||||||||||||||||||||||
|
Issuance
of common shares as payment of interest on director's loan, July 2009,
$0.04 per share
|
218,556 | 2 | 108,633 | 108,635 | ||||||||||||||||||||||||
|
Issuance
of common shares for cash, July 2009, $0.04 per share
|
6,540,017 | 66 | 2,714,934 | 2,715,000 | ||||||||||||||||||||||||
|
Issuance
of common shares as compensation for consulting contract, July 2009, $0.38
per share
|
100,000 | 1 | 379,999 | 380,000 | ||||||||||||||||||||||||
|
Issuance
of common shares on exercise of warrants by YA Global for cash August
2009
|
25,000 | — | 45,000 | 45,000 | ||||||||||||||||||||||||
|
Beneficial
conversion charge on 3rd debenture
|
— | 335,000 | 335,000 | |||||||||||||||||||||||||
|
Beneficial
conversion charge on director's loan
|
— | 117,109 | 117,109 | |||||||||||||||||||||||||
|
Reclassification
of warrants to derivative liability
|
(786,710 | ) | (786,710 | ) | ||||||||||||||||||||||||
|
Assumption
of options in reverse merger
|
644,806 | 644,806 | ||||||||||||||||||||||||||
|
Reclassification
of derivative liability on increase of authorized shares
|
13,501,360 | 13,501,360 | ||||||||||||||||||||||||||
|
Effect
of Reverse Merger
|
7,089,282 | 71 | 11,573,465 | — | — | 11,573,536 | ||||||||||||||||||||||
|
Net
loss attributable to noncontrolling interest
|
(211,670 | ) | (211,670 | ) | ||||||||||||||||||||||||
|
Comprehensive
Income (Loss)
|
||||||||||||||||||||||||||||
|
Net
Loss
|
(28,715,913 | ) | (28,715,913 | ) | ||||||||||||||||||||||||
|
Other
Comprehensive Income (Loss)
|
||||||||||||||||||||||||||||
|
Currency
Translation Adjustment
|
(529,296 | ) | (529,296 | ) | ||||||||||||||||||||||||
|
Total
Comprehensive Loss
|
(28,715,913 | ) | (529,296 | ) | (29,245,209 | ) | ||||||||||||||||||||||
|
Balance
October 31, 2009
|
49,432,640 | $ | 495 | $ | 35,543,722 | $ | (32,982,286 | ) | $ | (319,815 | ) | $ | (277,997 | ) | $ | 1,964,119 | ||||||||||||
See notes
to consolidated financial statements.
F-11
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Consolidated
Statements of Stockholders' Equity (Deficit)
|
|
Cumulative from November 20, 1997 (Date of
Inception) to October 31,
2010
|
|
Deficit
|
||||||||||||||||||||||||||||
|
Accumulated
|
||||||||||||||||||||||||||||
|
During
|
Accumulated
|
Total
|
||||||||||||||||||||||||||
|
Additional
|
the
|
Other
|
Stockholders'
|
|||||||||||||||||||||||||
|
Common Stock
|
Paid-in
|
Development
|
Comprehensive
|
Noncontrolling
|
Equity
|
|||||||||||||||||||||||
|
Shares
|
Amount
|
Capital
|
Stage
|
Income (Loss)
|
Interest
|
(Deficiency)
|
||||||||||||||||||||||
|
Balance
November 1, 2009
|
49,432,640 | $ | 495 | $ | 35,543,722 | $ | (32,982,286 | ) | $ | (319,815 | ) | $ | (277,997 | ) | $ | 1,964,119 | ||||||||||||
|
Issuance
of common shares in exchange
|
||||||||||||||||||||||||||||
|
for
services rendered January 2010, $1.40
|
35,000 | — | 49,000 | 49,000 | ||||||||||||||||||||||||
|
Common
shares issued on exercise of
|
||||||||||||||||||||||||||||
|
warrants,
February 2010
|
75,000 | 1 | 35,249 | 35,250 | ||||||||||||||||||||||||
|
Common
shares issued for cash, February 2010
|
77,000 | 1 | 76,999 | 77,000 | ||||||||||||||||||||||||
|
Common
shares issued in exchange for
|
||||||||||||||||||||||||||||
|
services
rendered, May 2010
|
78,371 | 1 | 88,199 | 88,200 | ||||||||||||||||||||||||
|
Common
shares issued in exchange for
|
||||||||||||||||||||||||||||
|
services
rendered, June 2010
|
50,000 | — | 18,000 | 18,000 | ||||||||||||||||||||||||
|
Common
shares issued for cash, October 2010
|
3,261,150 | 15 | 1,515,558 | 1,515,575 | ||||||||||||||||||||||||
|
Common
shares issued for subscription receivable, October 2010
|
230,000 | 1 | 114,999 | 115,000 | ||||||||||||||||||||||||
|
Net
loss attributable to noncontrolling interest
|
(14,225 | ) | (14,225 | ) | ||||||||||||||||||||||||
|
Comprehensive
Income (Loss)
|
||||||||||||||||||||||||||||
|
Net
Loss
|
(2,024,272 | ) | (2,024,272 | ) | ||||||||||||||||||||||||
|
Other
Comprehensive Income (Loss)
|
||||||||||||||||||||||||||||
|
Currency
Translation Adjustment
|
82,307 | 82,307 | ||||||||||||||||||||||||||
|
Total
Comprehensive Income (Loss)
|
(2,024,272 | ) | 82,307 | (1,941,965 | ) | |||||||||||||||||||||||
|
Balance
at October 31, 2010
|
53,009,161 | $ | 514 | $ | 37,441,728 | $ | (35,006,558 | ) | $ | (237,508 | ) | $ | (292,222 | ) | $ | 1,905,954 | ||||||||||||
See notes
to consolidated financial statements.
F-12
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Consolidated
Statements of Cash Flows
|
|
Years
Ended October 31, 2010 and 2009 and
|
|
Cumulative from November 20, 1997 (Date of
Inception) to October 31,
2010
|
|
Cumulative from
|
||||||||||||
|
November 20, 1997
|
||||||||||||
|
Years Ended
|
(Date of Inception)
|
|||||||||||
|
October 31
|
To October 31,
|
|||||||||||
|
2010
|
2009
|
2010
|
||||||||||
|
Cash
flows from operations activities
|
||||||||||||
|
Net
Loss for the Period
|
$ | (2,038,497 | ) | $ | (28,927,583 | ) | $ | (35,298,778 | ) | |||
|
Adjustments
to reconcile net loss for the period to net cash used in operating
activities
|
||||||||||||
|
Depreciation
and amortization of Property and Equipment
|
20,932 | 26,157 | 181,040 | |||||||||
|
Amortization
of Patent Application Costs
|
3,023 | 4,181 | 7,204 | |||||||||
|
Write-off
of Patent Application Costs
|
105,345 | 23,803 | 159,076 | |||||||||
|
Write-off
of Goodwill
|
— | 12,780,214 | 12,780,214 | |||||||||
|
Amortization
of Deferred Debt Issuance Costs
|
— | — | 511,035 | |||||||||
|
Loss
on Extinguishment of Debt
|
— | — | 275,676 | |||||||||
|
Loss
on Change in Value of Derivative Liability
|
— | 12,421,023 | 12,421,023 | |||||||||
|
Interest
Accrued and Foreign Exchange Loss on Debt
|
— | 356,608 | 922,539 | |||||||||
|
Unrealized
Foreign Currency Exchange Gains
|
— | (184,389 | ) | 25,092 | ||||||||
|
Beneficial
Conversion Charge included in
|
||||||||||||
|
Interest
Expense
|
— | 452,109 | 452,109 | |||||||||
|
Common
Stock Issued as Employee or Officer/Director Compensation
|
— | 2,288,815 | 2,508,282 | |||||||||
|
Common
Stock Issued for Services Rendered
|
155,200 | 402,312 | 557,512 | |||||||||
|
Stock
Options Issued for Services Rendered
|
— | — | 192,238 | |||||||||
|
Stock
Options Issued to Directors and
|
||||||||||||
|
Committee
Chairman
|
— | — | 54,582 | |||||||||
|
Changes
in Operating Assets and Liabilities, Net
of Acquisition Share
Subscriptions Receivable
|
(115,000 | ) | — | (115,000 | ) | |||||||
|
Deposits
and Prepaid Expenses
|
(77,778 | ) | (2,898 | ) | (88,985 | ) | ||||||
|
Refundable
Taxes Receivable
|
(7,081 | ) | (4,923 | ) | (21,095 | ) | ||||||
|
Government
Grants and Investment Tax Credits Receivable
|
19,072 | 55,522 | (136,420 | ) | ||||||||
|
Accounts
Payable and Accrued Expenses
|
(227,880 | ) | (425,731 | ) | (244,747 | ) | ||||||
|
Advances
|
— | — | 131 | |||||||||
|
Net
cash used in operating activities
|
(2,162,664 | ) | (734,780 | ) | (4,857,268 | ) | ||||||
|
Cash
flows from investing activities
|
||||||||||||
|
Cash
Acquired from Acquisition
|
— | 195,885 | 195,885 | |||||||||
|
Purchase
of Property and Equipment
|
(11,058 | ) | (8,950 | ) | (204,424 | ) | ||||||
|
Patent
Application Costs
|
(37,091 | ) | (15,164 | ) | (296,806 | ) | ||||||
|
Net
cash provided by (used in) investing activities
|
(48,149 | ) | 171,771 | (305,345 | ) | |||||||
|
Cash
flows from financing activities
|
||||||||||||
|
(Repayment)
of Capital Lease Obligation
|
(2,044 | ) | — | (2,044 | ) | |||||||
|
Due
to Director
|
(131,953 | ) | — | 740,479 | ||||||||
|
Issue
of Debentures
|
— | 371,333 | 1,378,305 | |||||||||
|
Issue
of Common Shares on Exercise of Stock options
|
— | 31 | 31 | |||||||||
|
Issue
of Common Shares on Exercise of Warrants
|
35,250 | — | 35,250 | |||||||||
|
Issue
of Common Shares for Cash
|
1,707,575 | 2,768,602 | 5,276,422 | |||||||||
|
Redemption
of 10% Senior Convertible Debentures
|
(25,000 | ) | (369,972 | ) | (394,972 | ) | ||||||
|
Net
cash provided by financing activities
|
1,583,828 | 2,769,994 | 7,033,471 | |||||||||
|
Effect
of foreign exchange on cash and cash equivalents
|
83,221 | (72,341 | ) | (26,096 | ) | |||||||
|
Cash
and Cash Equivalents
|
||||||||||||
|
Increase
(Decrease) in cash and cash equivalents during the period
|
(543,764 | ) | 2,134,644 | 1,844,762 | ||||||||
|
Beginning
of Period
|
2,388,516 | 253,872 | — | |||||||||
|
End
of Period
|
$ | 1,844,752 | $ | 2,388,516 | $ | 1,844,762 | ||||||
See notes
to consolidated financial statements.
F-13
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
|
|
1.
|
Nature
of Business
|
The
accompanying audited consolidated financial statements have been prepared in
accordance with the requirements of Form 10-K and Article 8 of Regulation S-X of
the Securities and Exchange Commission (the “Commission”) and include the
results of CardioGenics, Inc. and JAG Media Holdings, Inc and its subsidiaries
(”JAG Media”) (from July 31, 2009, date of acquisition) which are
collectively referred to as the “Company.”
CardioGenics
Inc. (“CardioGenics”) was incorporated on November 20, 1997 in the Province of
Ontario, Canada, and carries on the business of development and
commercialization of diagnostic test products to the In Vitro Diagnostics
testing market. CardioGenics has several test products that are in various
stages of development.
CardioGenics’
business is that of a development-stage company, with a limited history of
operations and whose revenues, to date, have been primarily comprised of grant
revenue and Scientific Research Tax Credits from government agencies. There can
be no assurance that the Company will be successful in obtaining regulatory
approval for marketing of any of the existing or future products that the
Company will succeed in developing.
On
July 31, 2009, CardioGenics acquired the business of JAG Media Holdings, Inc.
(“JAG Media”)(see Note 4). The business acquired is
that of gathering and compiling financial and investment information from
various financial institutions and other Wall Street professionals. Revenues of
the acquired business of JAG media are generated by releasing such financial
information to subscribers in a consolidated format on a timely basis through
facsimile transmissions and a web site. Further, software focused on streaming
video solutions was acquired through the acquisition of JAG Media by
CardioGenics. Historically, further development of this software has been
limited as a result of JAG Media’s lack of financial resources.
On
February 11, 2010, the Company entered into an LLC Membership Interest Purchase
Agreement with Rothcove Partners LLS (“Rothcove”) pursuant to which the Company
sold its interest in JAG Media to Rothcove (see Note 19).
References
herein to CardioGenics common shares has been retrospectively adjusted to
reflect the exchange ratio of 20.957 established in the Share Purchase
Agreement.
On
October 27, 2009 the name of the Company was changed from Jag Media Holdings,
Inc. to CardioGenics Holdings, Inc.
On April
23, 2010, the Company’s Board of Directors approved a reverse stock split of its
issued and outstanding common shares. The total authorized shares of
common stock was at the same time reduced to 65,000,000. The Board of
Directors selected a ratio of one-for-ten and the reverse stock split was
effective on June 20, 2010. Trading of the Company’s common stock on
the Over-The-Counter Capital Market on a split adjusted basis began at the open
of trading on June 21, 2010. The reverse stock split affected all
shares of the Company’s common stock, as well as options to purchase the
Company’s common stock and other equity incentive awards and warrants that were
outstanding immediately prior to the effective date of the reverse stock
split. All references to common shares and per-share data for prior
periods have been retroactively restated to reflect the reverse stock split as
if it had occurred at the beginning of the earliest period
presented.
F-14
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
|
|
2.
|
Basis
of Presentation
|
The
accompanying consolidated financial statements have been prepared using the
accounting principles generally accepted in the United States of America
applicable to a going concern, which contemplates the realization of assets and
the satisfaction of liabilities and commitments in the normal course of
business.
The
Company has incurred operating losses and has experienced negative cash flows
from operations since inception. The Company has a deficit
accumulated during the development stage to October 31, 2010 of approximately
$35.0 million. The Company has not yet established an ongoing source of revenues
sufficient to cover its operating costs and to allow it to continue as a going
concern. The Company has funded its activities to date almost
exclusively from debt and equity financings. These conditions raise substantial
doubt about the Company’s ability to continue as a going concern.
The
Company will continue to require substantial funds to continue research and
development, including preclinical studies and clinical trials of its products,
and to commence sales and marketing efforts, if the FDA and other regulatory
approvals are obtained. In order to meet its operating cash flow requirements
Management’s plans include financing activities such as private placements of
its common stock and issuances of convertible debt instruments. Management is
also actively pursuing industry collaboration activities including product
licensing and specific project financing.
While the
Company believes it will be successful in obtaining the necessary financing to
fund its operations, meet revenue projections and manage costs, there are no
assurances that such additional funding will be achieved and that it will
succeed in its future operations. The financial statements do not include any
adjustments relating to the recoverability and classification of recorded asset
amounts or amounts of liabilities that might be necessary should the Company be
unable to continue in existence.
|
|
3.
|
Summary
of Significant Accounting Policies
|
|
|
(a)
|
Principles
of Consolidation
|
The
consolidated financial statements include the accounts of the Company and its
100% owned subsidiaries. All significant intercompany transactions and balances
have been eliminated.
|
|
(b)
|
Development
Stage Company
|
The
accompanying financial statements have been prepared in accordance with the
provisions of the guidance for development stage enterprises.
|
|
(c)
|
Cash
and Cash Equivalents
|
The
Company considers all highly liquid investments purchased with an
original maturity of three months or less to be cash equivalents.
|
|
(d)
|
Share
Subscription Receivable
|
Subscriptions
for common shares received are recorded as receivable on the subscription date
with the offsetting credit to capital on the same date.
|
|
(e)
|
Government
Grants and Investment Tax Credits
Receivable
|
The
Company’s accounts include claims for investment tax credits relating to
scientific research activities of the Company prior to the acquisition described
at Note 4. The qualification and recording of this activity for investment tax
credit purposes is established by Canadian Income Tax authorities when the
income tax returns for the period are assessed. The credit has been recognized
in the statement of operations in the year in which the expenses were
incurred.
F-15
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
Subsequent
to the acquisition described at Note 4, the Company no longer qualifies to
receive refunds of Investment Tax Credits (“ITCs”) resulting from scientific
research. Currently ITCs resulting from scientific research are
carried forward to a time when the company becomes tax paying at which time said
ITCs are applicable against taxes payable.
|
|
(f)
|
Property
and Equipment
|
Property
under capital leases and the related obligation for future lease payments are
initially recorded at an amount equal to the lesser of fair value of the
property or equipment and the present value of those lease
payments. Property and equipment is depreciated using methods and
rates as follows:
|
Furniture
and Fixtures
|
20%
declining balance
|
|
|
Lab
Equipment
|
20%
declining balance
|
|
|
Computer
Equipment – Hardware
|
30%
declining balance
|
|
|
Computer
Equipment – Software
|
50%
declining balance
|
|
|
Leasehold
Improvements
|
Straight-line
over the lesser of the life of the asset or the life of the
lease
|
|
|
(g)
|
Patents
|
Capitalized
patent costs represent legal and application costs incurred to establish
patents. Capitalized patent costs are amortized on a straight line method over
the related patent term. As patents are abandoned, the net book value of the
patent is written off.
|
|
(h)
|
Impairment
or Disposal of Long-Lived Assets
|
The
Company assesses the impairment of long-lived assets under the guidance of
standards for the impairment or disposal of long-lived assets whenever events or
changes in circumstances indicate that the carrying value may not be
recoverable. For long-lived assets to be held and used, the Company
recognizes an impairment loss only if its carrying amount is not recoverable and
exceeds its fair value. The carrying amount of the long-lived asset
is not recoverable if it exceeds the sum of the undiscounted cash flows expected
to result from the use and eventual disposal of the asset.
|
|
(i)
|
Convertible
Debentures
|
In
accordance with guidance in accounting for convertible securities with
beneficial conversion features or contingently adjustable conversion ratios, the
Company recognized an imbedded beneficial conversion feature present in the
convertible debentures. The Company allocated a portion of the proceeds equal to
the intrinsic value of that feature to additional paid-in
capital. The debt discount attributed to the beneficial conversion
feature is amortized over the convertible debenture's maturity period as
interest expense using the effective yield method.
In
addition, the Company recognized the value attributable to the warrants to
additional paid-in capital and a discount against the convertible debentures.
The Company valued the warrants using the Black-Scholes pricing
model. The debt discount attributed to the value of the warrants
issued is amortized over the convertible debenture’s maturity period as interest
expense using the effective yield method.
|
|
(j)
|
Research
and Development Costs
|
Expenditures
for research and development are expensed as incurred and include, among other
costs, those related to the production of prototype products, including payroll
costs. Amounts expected to be received from governments under Scientific
Research Tax Credit arrangements are offset against current
expenses. The Company recognizes revenue from restricted grants in
the period in which the Company has incurred the expenditures in compliance with
the specific restrictions.
F-16
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
|
(k)
|
Income
Taxes
|
The
Company utilizes the liability method of accounting for income taxes as set
forth in the authoritative guidance. Under the liability method, deferred taxes
are determined based on the temporary differences between the financial
statement and tax basis of assets and liabilities using tax rates expected to be
in effect during the years in which the basis differences reverse. A valuation
allowance is recorded when it is more likely than not that some of the deferred
tax assets will not be realized. As there is no certainty that the Company will
generate taxable income in the foreseeable future to utilize tax losses
accumulated to date, no provision for ultimate tax reduction has been made in
these financial statements.
On
November 1, 2007, the Company adopted the guidance issued for accounting for
uncertainty in income taxes which provides detailed guidance for the financial
statement recognition, measurement and disclosure of uncertain tax positions
recognized in an enterprise’s financial statements. Income tax positions must
meet a more-likely-than-not recognition threshold at the effective date to be
recognized upon the adoption of the guidance and in subsequent periods. The
Company recognizes potential accrued interest and penalties related to
unrecognized tax benefits within operations as income tax expense. Upon
adoption, there were no adjustments required.
|
(l)
|
Stock-Based
Compensation
|
The
Company follows the authoritative guidance for stock-based compensation which
requires that new, modified and unvested share-based payment transactions with
employees, such as grants of stock options and restricted stock, be recognized
in the financial statements based on their fair value at the grant date and
recognized as compensation expense over their vesting periods. The Company has
also considered the related guidance of the Security and Exchange Commission
(“SEC”). The Company estimates the fair value of stock options and shares issued
as compensation to employees and directors as of the date of grant using the
Black-Scholes pricing model and restricted stock based on the per share
value. The Company also follows the guidance for equity instruments
that are issued to other than employees for acquiring, or in conjunction with
selling, goods or services for equity instruments issued to consultants
which provides guidance on transactions in which (1) the fair value
of the equity instruments is more reliably measurable than the fair value of the
goods or services received and (2) the counterparty receives shares of stock,
stock options, or other equity instruments in settlement of the entire
transaction or, if the transaction is part cash and part equity instruments, in
settlement of the portion of the transaction for which the equity instruments
constitute the consideration. Options issued with a nominal exercise
price in exchange for services rendered were measured at the fair value of the
underlying services rendered on the date of grant. The expense was recorded to
the statement of operations with a corresponding increase in share capital with
no additional increase in the number of shares as they were legally not yet
exercised.
|
(m)
|
Net
Loss Per Common Share
|
Basic
loss per share is computed by dividing loss available to common stockholders by
the weighted average number of common shares outstanding during the period.
Diluted earnings (loss) per share gives effect to all dilutive potential common
shares outstanding during the period. The computation of diluted earnings (loss)
per share does not assume conversion, exercise or contingent exercise of
securities that would have an anti-dilutive effect on earnings (loss) per
share.
|
(n)
|
Comprehensive
Loss
|
Other
comprehensive loss, which includes only foreign currency translation
adjustments, is shown in the Statements of Changes in Stockholders’ Equity
(Deficit).
F-17
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
|
|
(o)
|
Concentration
of Credit Risk
|
The
Company maintains cash balances, at times, with financial institutions in excess
of amounts insured by the Canada Deposit Insurance Corporation and the Federal
Deposit Insurance Corporation. Management monitors the soundness of
these institutions and has not experienced any collection losses with these
financial institutions.
|
|
(p)
|
Use
of Estimates
|
The
preparation of financial statements in conformity with accounting principles
generally accepted in the United States of America requires management to make
estimates and assumptions that affect the reported amounts of assets and
liabilities and disclosure of contingent assets and liabilities at the dates of
the financial statements and the reported amounts of revenues and expenses
during the reporting periods. Actual results could differ from those
estimates. By their nature, these estimates are subject to
uncertainty and the effect on the consolidated financial statements of changes
in such estimates in future periods could be material.
|
|
(q)
|
Foreign
Currency Translation
|
The
Company maintains its accounting records for its Canadian operations in Canadian
dollars. Transactions in United States dollars (“USD”) are translated into
Canadian dollars at rates in effect at the date of the transaction and gains or
losses on such transactions are recorded at the time of settlement in the
statement of operations.
The
Company’s reporting currency is the United States Dollar. Foreign
denominated assets and liabilities of the Company are translated into USD at the
prevailing exchange rates in effect at the end of the reporting period, the
historical rate for stockholders’ equity and a weighted average of exchange rate
in effect during the period for expenses, gains and
losses. Adjustments that arise from translation into the reporting
currency are recorded in the accumulated other comprehensive income (loss)
component of stockholders’ equity (deficit).
|
|
(r)
|
Financial
Instruments
|
The
carrying values of cash and cash equivalents, other current assets, accounts
payable and accrued expenses approximate their fair values due to their
short-term nature. Long-term debt and convertible debentures
approximate their fair value based upon the borrowing rates available for the
nature of the underlying debt.
|
|
(s)
|
Revenue
Recognition
|
Fees for
subscriptions are generally billed in advance on a monthly, quarterly,
semi-annual or annual basis. Revenues from subscriptions are
recognized ratably over the subscription period. Subscription fees
collected that relate to periods subsequent to the date of the consolidated
balance sheets are included in deferred revenues.
|
|
(t)
|
Effects
of Recent Accounting Pronouncements
|
In
September 2006, “Fair Value Measurements” pronouncement was issued which defines
fair value, establishes a framework for measuring fair value in accordance with
accounting principles generally accepted in the United States, and expands
disclosures about fair value measurements. This pronouncement is effective for
financial statements issued for the Company’s fiscal year beginning November 1,
2008, with earlier application encouraged. Any amounts recognized upon adoption
as cumulative effect adjustments will be recorded to the opening balance of
retained earnings in the year of adoption. On February 12, 2008, the effective
date for non-financial assets and liabilities was delayed to fiscal years
beginning after November 15, 2008; however, the effective date for financial
assets remains intact. The Company has adopted the fair value measurements
pronouncement for current assets and liabilities which has not had a material
effect on its consolidated financial statements.
F-18
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
In
December 2007, “Business Combinations” pronouncement was issued, which
establishes principles and requirements for how an acquirer recognizes and
measures in its financial statements the identifiable assets acquired, the
liabilities assumed, and any non-controlling interest in an acquiree, including
the recognition and measurement of goodwill acquired in a business
combination. This Pronouncement is effective as of the beginning of
the first fiscal year beginning on or after December 15,
2008. Earlier adoption is prohibited. This pronouncement
did not have a material effect on its consolidated financial
statements.
|
4.
|
Acquisition
|
On July
31, 2009, the Company completed a reverse acquisition of privately held
CardioGenics Inc. (“CardioGenics”), an Ontario, Canada Corporation. The
acquisition was effected pursuant to a Share Purchase Agreement dated May 22,
2009 by and among the Company, CardioGenics Inc. and CardioGenics ExchangeCo
Inc., the Company’s wholly owned subsidiary (“ExchangeCo”). In accordance with
the terms of the Share Purchase Agreement, 99% of the holders of common shares
of CardioGenics Inc. (two (2) minority shareholders of CardioGenics Inc. holding
in aggregate 17,387
common shares of CardioGenics Inc. did not participate) surrendered their
CardioGenics Common Shares. CardioGenics Inc. caused the Company to issue to
ExchangeCo or CardioGenics’ shareholders 42,218,361 shares of the Company’s
common stock, par value $0.00001 (the “Share Consideration”). The CardioGenics
Inc.’s shareholders had the option to receive their pro-rata allocation of the
Share Consideration in the form of (a) the Company’s common stock (the “JAG
Consideration Shares”) or (b) exchangeable shares of ExchangeCo. Inc., which
shares shall be exchangeable at any time after July 31, 2009 into a number of
shares of the Company’s common stock equal to such shareholders’ pro rata
allocation of the Share Consideration (the “Exchangeable Shares”). The
Exchangeable Shares have the same voting rights, dividend entitlements and other
attributes as JAG Media common stock. Exchangeable Shares will automatically be
exchanged for JAG Media common stock five years from July 31, 2009, and in
certain other events. The Share Consideration provides the former CardioGenics
shareholders with direct and/or indirect ownership of approximately 85% of JAG
Media’s outstanding common stock (on a fully diluted basis) as of July 31,
2009.
On July
31, 2009, 14,552,819 common shares of JAG Media were issued to former
shareholders of CardioGenics and 27,665,541 common shares of JAG Media were
issued to ExchangeCo. These shares are not registered for resale and, therefore,
shall remain subject to the rights and restrictions of Rule 144. All
Exchangeable Shares received by the CardioGenics shareholders in exchange for
their CardioGenics Common Shares (and any JAG Media common stock into which such
Exchangeable Shares may be exchanged) shall not be registered for resale prior
to six (6) months following July 31, 2009 and, therefore, shall remain subject
to the rights and restrictions of Rule 144 prior to any such
registration.
The Share
Consideration provided the CardioGenics Inc.’s shareholders with direct and/or
indirect ownership of approximately 85% of the Company’s outstanding common
stock (on a fully diluted basis) as of July 31, 2009. Based on the five-day
average price of the Company’s common stock of $1.60 per share, the purchase
price approximated was $11,573,536, plus approximately $342,880 of acquisition
costs plus the fair value of options and warrants assumed of
$644,806.
F-19
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
A summary
of the purchase price allocation is as follows:
|
Common
stock issued
|
$ | 11,573,536 | ||
|
Acquisition
costs incurred
|
342,880 | |||
|
Fair
value of options and warrants assumed
|
644,806 | |||
|
Total
purchase price
|
$ | 12,561,222 | ||
The
purchase price has been allocated as follows based on the fair values of the
assets and liabilities acquired:
|
Cash
|
$ | 195,885 | ||
|
Accounts
payable
|
(386,177 | ) | ||
|
Derivative
liability for warrants assumed
|
(28,700 | ) | ||
|
Goodwill
|
12,780,214 | |||
|
Total
|
$ | 12,561,222 |
The
following pro forma consolidated financial information presents the combined
results of operations of JAG Media Holdings, Inc. and CardioGenics, Inc. as if
the acquisition had occurred as of November 1, 2009 and 2008, after giving
effect to certain adjustments, including the issuance of JAG Media Holdings,
Inc. common stock as part of the purchase price. For the purpose of this pro
forma presentation, both JAG Media Holdings, Inc.’s and CardioGenics, Inc.’s
financial information is presented for the years ended October 31, 2010 and
2009, respectively. The pro forma condensed consolidated financial information
does not necessarily reflect the results of operations that would have occurred
had JAG Media Holdings, Inc. and CardioGenics, Inc. been a single entity during
such periods (see 1).
|
Years ended
October 31,
|
||||||||
|
2010
|
2009
|
|||||||
|
Revenues
|
$ | 14,852 | $ | 130,826 | ||||
|
Net
Loss
|
$ | (2,038,497 | ) | $ | (29,859,758 | ) | ||
|
Weighted-average
shares of Common stock outstanding: Basic and diluted
|
49,624,916 | 35,427,442 | ||||||
|
Basic
and diluted net loss per common share
|
$ | (0.04 | ) | $ | (0.84 | ) | ||
F-20
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
|
5.
|
Property
and Equipment
|
The costs
and accumulated depreciation and amortization of property and equipment are
summarized as follows:
|
October 31
|
||||||||
|
2010
|
2009
|
|||||||
|
Furniture
and Fixtures
|
$ | 12,120 | $ | 7,983 | ||||
|
Lab
Equipment
|
149,262 | 99,340 | ||||||
|
Computer
Hardware
|
19,490 | 19,490 | ||||||
|
Computer
Software
|
8,433 | 8,433 | ||||||
|
Leasehold
Improvements
|
91,269 | 91,269 | ||||||
|
Total
Property and Equipment
|
280,574 | 226,515 | ||||||
|
Less
Accumulated Depreciation and Amortization
|
193,109 | 172,177 | ||||||
|
Property
and Equipment, Net
|
$ | 87,465 | $ | 54,338 | ||||
Depreciation
and amortization expense amounted to $20,932 and $26,157 for the years ended
October 31, 2010 and 2009, respectively.
|
6.
|
Patents
|
The costs
and accumulated amortization of patents are summarized as follows:
|
October 31
|
||||||||
|
2010
|
2009
|
|||||||
|
Patents
|
$ | 177,907 | $ | 246,161 | ||||
|
Less:
Accumulated Amortization
|
(7,204 | ) | (4,181 | ) | ||||
|
Patents,
Net
|
$ | 170,703 | $ | 241,980 | ||||
|
Weighted-Average
Life
|
17 Years
|
17 Years
|
||||||
|
|
Amortization
expense amounted to $3,023 and $4,181 for the years ended October 31, 2010
and 2009, respectively. Amortization expense is expected to be
approximately $3,000 per year for the years ended October 31, 2011 through
2015. During the years ended October 31, 2010 and 2009, the
Company wrote off approximately $105,345 and $23,803 of net book value of
patents, respectively, for abandoned
patents.
|
|
7.
|
Goodwill
|
Goodwill
and other intangible assets with indefinite lives are tested for impairment
annually, as required by pronouncement, “Goodwill and Other Intangible
Assets”. First, the fair value of the reporting unit is compared to
its carrying value. If the fair value is less than the carrying value, a second
step is performed. In the second step, an implied goodwill value is determined
by deducting the fair value of all tangible and intangible net assets of the
reporting unit from the fair value of the reporting unit. If the implied fair
value of the goodwill as calculated is less than the carrying amount of the
goodwill, an impairment charge is recorded for the difference.
As
indicated above, the goodwill impairment assessment is a two step
process.
F-21
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
A $12.8
million goodwill impairment charge was recorded in the fourth quarter of
2009. This impairment charge was determined by management soliciting
offers for sale of the JAG Notes division and receiving therefor offers in the
range of $40,000 to $60,000. In November 2009, the company made a
decision to sell the Jag Notes.
On
February 10, 2010, the Company entered into an LLC Membership Interest Purchase
Agreement with Rothcove Partners LLC pursuant to which the Company sold its 100%
membership interest in its Pixaya LLC subsidiary to Rothcove. In
consideration for its acquisition of the Pixaya LLc membership interest,
Rothcove assumed $100,000 in accounts payable of Pixaya LLC and its subsidiary
Pixaya (UK) Limited. The transaction closed on February 11,
2010.
A summary
of the change in the Company’s goodwill for the years ended October 31,
2010 and 2009 is as follows:
|
October
31,
|
||||||||
|
2010
|
2009
|
|||||||
|
Goodwill
related to purchase (see Note 4)
|
$ | 0 | $ | 12,780,214 | ||||
|
Goodwill
impairment charge- JAG
Notes
|
0 | (12,780,214 | ) | |||||
|
Goodwill
|
$ | 0 | $ | 0 | ||||
|
8.
|
Due
to Director
|
The
amount due to a director is due on demand and carries interest at 10% per
annum. On July 31, 2009, $885,000 due to a director was converted to
2,377,813 common shares of JAG Media. A beneficial conversion charge of $117,109
was credited against additional paid-in capital. Accrued interest on the
director’s loan of $108,635 was converted to 218,556 common shares of JAG Media.
at the same time.
On
January 28, 2009, the Company issued to a director and an officer of the Company
a new series of Debentures in the amount of $371,333. The Debentures
were for a term of two years and carried interest at 10% per
annum. At July 31, 2009, the holders of the Debentures exchanged the
Debentures, plus accrued interest, for 1,701,708 common shares of JAG Media
common stock. A beneficial conversion charge of $335,000 was credited against
additional paid-in capital.
|
9.
|
Income
Taxes
|
The
Company adopted the provisions of the guidance for uncertainty in income taxes
on August 1, 2007. The guidance clarifies the accounting for uncertainty in
income taxes recognized in an enterprise’s financial statement, and prescribes a
recognition threshold and measurement process for financial statement
recognition and measurement of a tax position taken or expected to be taken in a
tax return. It also provides guidance on derecognition classification, interest
and penalties accounting in interim periods disclosure and
transition.
Based on
the Company’s evaluation, management has concluded that there are no significant
tax positions requiring recognition in the consolidated financial
statements.
The
Company has incurred losses in Canada since inception, which have generated net
operating loss carryforwards for income tax purposes. The net
operating loss carryforwards arising from Canadian sources as of October 31,
2010 were $8,169,337 (2009 - $6,429,964) which will expire from 2014 through
2029.
F-22
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
All
fiscal years except 2010 and 2009 have been assessed; however, claims relating
to research and development credits are open for review for the fiscal years
ended October 2010, 2009, 2008, 2007, and July 2009.
As of
October 31, 2010 and 2009, the Company had net operating loss carryforwards from
US sources of approximately $40,076,000 and $40,154,000, respectively available
to reduce future Federal taxable income which will expire from 2019 through
2029.
For the
years ended October 31, 2010 and 2009, the Company’s effective tax rate differs
from the statutory rate principally due to the net operating losses for which no
benefit was recorded.
As of
October 31, 2010 and 2009, the Company’s deferred tax assets consisted of the
effects of temporary differences attributable to the following:
|
October 31
|
||||||||
|
2010
|
2009
|
|||||||
|
Property
and Equipment
|
$ | (21,512 | ) | $ | (15,232 | ) | ||
|
Net
operating loss carryforwards
|
17,269,873 | 16,712,575 | ||||||
|
Unrealized
foreign exchange
|
(15,304 | ) | 15,951 | |||||
|
Investment
tax credits
|
(100,000 | ) | (50,903 | ) | ||||
|
Total
Deferred Tax Assets
|
17,133,057 | 16,662,391 | ||||||
|
Valuation
Allowance
|
(17,133,057 | ) | (16,662,391 | ) | ||||
|
Net
Deferred Income Taxes
|
$ | — | $ | — | ||||
A
reconciliation of the Canadian combined statutory rate to the Company’s
effective tax rate for the years ended October 31, 2010 and 2009 is as
follows:
|
October 31
|
||||||||
|
2010
|
2009
|
|||||||
|
Statutory
rate
|
30 | % | 34 | % | ||||
|
Decrease
in income tax rate resulting from:
|
||||||||
|
Changes
in tax rate
|
— | — | ||||||
|
Permanent
differences
|
(12.5 | )% | (33.3 | )% | ||||
|
Change
in valuation allowance
|
(17.5 | )% | (0.7 | )% | ||||
|
Effective
tax rate
|
0.0 | % | 0.0 | % | ||||
F-23
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
|
10.
|
Accounts
Payable and Accrued Expenses
|
|
October 31
|
||||||||
|
2010
|
2009
|
|||||||
|
Accounts
Payable
|
$ | 109,913 | $ | 199,868 | ||||
|
Research
and Development
|
26,288 | 31,864 | ||||||
|
Executive
Compensation
|
— | 110,916 | ||||||
|
Investor
Relations
|
87,500 | — | ||||||
|
Patent
Application Costs
|
20,245 | 40,194 | ||||||
|
Legal
Fees
|
246,915 | 231,062 | ||||||
|
Accounting
Fees
|
32,294 | 137,133 | ||||||
|
Total
|
$ | 523,155 | $ | 751,037 | ||||
|
11.
|
Stock-Based
Compensation
|
The
Company follows the guidance for stock-based compensation. Stock-based employee
compensation related to stock options for each of the years ended October 31,
2010 and 2009 amounted to $-0-.
The
following is a summary of the common stock options granted, forfeited or expired
and exercised under the Plan:
|
Weighted
|
||||||||
|
Average
|
||||||||
|
Exercise
|
||||||||
|
Options
|
Price
|
|||||||
|
Outstanding
– October 31, 2008
|
990,120 | $ | 0.02 | |||||
|
Granted
|
— | — | ||||||
|
Forfeited/expired
|
(419,140 | ) | $ | 0.60 | ||||
|
Exercised
|
(570,980 | ) | — | |||||
|
Assumed
upon JAG Media acquisition
|
275,000 | $ | 2.50 | |||||
|
Granted
|
30,000 | $ | 0.90 | |||||
|
Outstanding
– October 31, 2009
|
305,000 | $ | 2.34 | |||||
|
Granted
|
— | — | ||||||
|
Forfeited/expired
|
— | — | ||||||
|
Exercised
|
— | — | ||||||
|
Outstanding
– October 31, 2010
|
305,000 | $ | 2.34 | |||||
|
Exercisable
|
305,000 | $ | 2.34 | |||||
Options
typically vest immediately at the date of grant. As such, the Company
does not have any unvested options or unrecognized compensation expense at
October 31, 2010 and 2009.
F-24
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
The fair
value of each option granted is estimated on grant date using the Black-Scholes
option pricing model which takes into account as of the grant date the exercise
price and expected life of the option, the current price of the underlying stock
and its expected volatility, expected dividends on the stock and the risk-free
interest rate for the term of the option. The Company granted to a consultant
30,000 stock options during the year ended October 31, 2009 and no stock options
during the year ended October 31, 2010. The following is the average
of the data used to calculate the fair value:
|
Risk
Free Interest Rate
|
1.1 – 3.4 | % | ||
|
Expected
Life (Years)
|
1.83 – 9.75 | % | ||
|
Expected
Volatility
|
50.11 – 62.14 | % | ||
|
Expected
Dividends
|
$ | — |
The
following table summarizes information on stock options outstanding at October
31, 2010:
|
Options Outstanding and Exercisable
|
|||||||||||||||||
|
|
Weighted
|
||||||||||||||||
|
Number
|
Weighted
|
Average
|
|||||||||||||||
|
Outstanding
|
Average
|
Remaining
|
Aggregate
|
||||||||||||||
|
Range of
|
at
|
Exercise
|
Life
|
Intrinsic
|
|||||||||||||
|
Exercise Price
|
October 31, 2010
|
Price
|
(Years)
|
Value
|
|||||||||||||
| $ |
0.20
|
75,000 | $ | 0.20 | .83 | ||||||||||||
| $ |
0.90
|
30,000 | 0.90 | 8.75 | |||||||||||||
| $ |
3.40
|
200,000 | 3.40 | 8.75 | |||||||||||||
| 305,000 | 6.80 | $ | 14,000 | ||||||||||||||
|
For the Year Ended October 31,
|
||||||||
|
2010
|
2009
|
|||||||
|
Weighted
Average Fair Value of Options Granted
|
$ | — | $ | 0.35 | ||||
|
Cash
Received for Exercise of Stock Options
|
$ | — | $ | 28 | ||||
The
intrinsic value is calculated as the difference between the market value as of
October 31, 2010 and the exercise price of the shares. The market
value as of October 31, 2010 was $0.76 as reported by the NASDAQ Stock
Market.
|
12.
|
Capital
Lease Obligations
|
The
Company finances certain equipment acquisitions through a capital lease
agreement that expires in 2012. Future minimum rental payments under
capital leases and related information in years subsequent to October 31, 2010
are presented in the table below:
|
October 31
|
||||||||
|
2010
|
2009
|
|||||||
|
Total
amount payable in equal monthly installments of $2,003
|
$ | 46,029 | $ | — | ||||
|
Less:
Amount representing interest
|
4,156 | — | ||||||
|
Present
value of minimum lease payments
|
41,873 | — | ||||||
|
Less:
Current portion
|
20,992 | — | ||||||
| $ | 20,881 | $ | — | |||||
F-25
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
Payments
under the capital lease are due as follows:
|
For the years ended October
31
|
||||
|
2011
|
$ | 24,036 | ||
|
2012
|
24,036 | |||
|
2013
|
— | |||
|
2014
|
— | |||
|
2015
|
— | |||
| $ | 48,072 | |||
The net
book value of equipment under capital lease at October 31, 2010 is $41,720 and
is included in property and equipment.
|
13.
|
Stockholders’
Equity (Deficit)
|
Equity
Instruments Issued for Services Rendered
During
the years ended October 31, 2000 through 2006 CardioGenics Inc. issued stock
options with a nominal exercise price in exchange for services rendered to
CardioGenics Inc. The fair value of each stock option was measured at the fair
value of the underlying services on the date of grant. The fair value
of each grant was charged to the related expense in the statement of
operations.
The
Company assumed options outstanding at JAG Media entitling the employees to
purchase 75,000 common shares of the Company’s stock at a price of $0.20 per
share to August 31, 2011. The Company issued options to employees entitling the
employees to purchase 200,000 common shares of the Company’s stock at a price of
$3.40 per share to July 31, 2019, based upon change of control provisions in
their employment agreements. All these options were immediately
vested. The fair value of the 275,000 options is included in the
purchase price.
On August
1, 2009, the Company issued options to a consultant entitling the consultant to
purchase 30,000 common shares of the Company’s stock at a price of $0.90 per
share to July 31, 2019. These options were immediately
vested.
F-26
|
CardioGenics
Holdings Inc.
|
|
|
(A
Development Stage Company)
|
|
|
Notes
to Consolidated Financial Statements
|
|
|
October 31, 2010 and 2009
|
|
October
31,
|
||||||||
|
2010
|
2009
|
|||||||
| Warrants | ||||||||
|
Issued
to subscribers to the debenture financing of 2003 and its related
extension entitling the holder to purchase 1 common share of the Company
at an exercise price of $0.47 per common share up to and including July
31, 2012
|
2,046,808 | 2,121,808 | ||||||
|
Issued
to subscribers to the debenture financing of 2004 and its related
extension entitling the holder to purchase 1 common share in the Company
at an exercise price of $0.47 per common share up to and including July
31, 2012
|
1,043,659 | 1,043,659 | ||||||
|
Issued
to agents for the debenture financings of 2003 and 2004 entitling the
holder to purchase 1 common share in the Company at an exercise
price of $0.47 per common share up to and including July 31,
2012
|
208,417 | 208,417 | ||||||
|
Issued
to former employee entitling the holder to purchase 1 common share in the
company at an exercise price of $0.47 per common share up to and including
July 31, 2012
|
136,220 | 136,220 | ||||||
|
Issued
to Consultants July 31, 2009, entitling the holder to purchase 1 common
share of the company at an exercise price of $0.90 per share up to and
including July 31, 2012
|
104,785 | 104,785 | ||||||
|
Issued
to consultant August 1, 2009, entitling the holder to purchase 1 common
share in the company at an exercise price of $0.90 per common share up to
and including July 31, 2017
|
287,085 | 287,085 | ||||||
|
Total
Warrants outstanding
|
3,826,974 | 3,901,974 | ||||||
F-27
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
As of
July 31, 2009, the conversion of the warrants would result in the issuance of
common shares in excess of the number of common shares authorized, the Company
determined that based on the guidance on derivative financial instruments
indexed to, and potentially settled in a Company’s own stock, the Company would
be prohibited from concluding that it would have a sufficient number of
authorized and unissued shares to net-share settle any of those warrants or any
other warrants or options previously issued or granted to non-employees.
Therefore, as of the date of the reverse acquisition, the Company recorded the
related fair value of all warrants issued with prior debentures previously
issued to non-employees as a liability. Subsequent changes in the fair value of
such options and warrants at the end of each reporting period were to be
recorded as charges or credits to the Company’s results of
operations. On September 30, 2009, the Company’s authorized share
capital was amended to increase the number of authorized common shares to
65,000,000. At that time the outstanding options and warrants were
re-valued with the subsequent valuation of $13,501,360 re-classified to
additional paid-In capital.
At July
31, 2009, the Company assumed the remainder of warrants dated May 24, 2006
entitling YA Global to purchase 25,000 shares of the Company’s common stock for
$4.00 per share. These warrants were exercised August 5,
2009.
|
14.
|
Standby
Equity Distribution Agreement
|
On March
12, 2009, the Company and YA Global Master SPV Ltd. (“YA Ltd”) entered into a
Standby Equity Distribution Agreement (The “SEDA”) pursuant to which YA Ltd
agreed to purchase up to $5,000,000 of the Company’s common stock (the
“Commitment Amount”) over the course of the thirty-six (36) months following the
date the registration statement for the shares to be issued pursuant to the SEDA
is first declared effective (the “Commitment Period”). The Company shall have
the right, but not the obligation, to sell common stock to YA Ltd during the
Commitment Period. Each right to sell common stock to YA Ltd is an “Advance”
under the SEDA.
On March
12, 2009, concurrent with the execution of the SEDA, the Company and YA Ltd also
entered into a Registration Rights Agreement (the “Registration Rights
Agreement”) pursuant to which the Company agreed to register the shares of the
Company’s common stock to be issued in connection with the SEDA (the
“Registrable Securities”). The Company may not file the registration statement
for the Registrable Securities (the “Registration Statement”) prior to the tenth
(10th)
trading day following the Commencement Date and the Company shall not have the
ability to make any Advances under the SEDA until the Registration Statement is
declared effective. The Company shall cause the Registration Statement that has
been declared effective to remain effective at all times until all Registrable
Securities under the Registration Statement cease to be Registrable Securities.
Once issued, Registrable Securities cease to be Registrable Securities when (i)
such Registrable Securities have been disposed of pursuant to the Registration
Statement; (ii) such Registrable Securities have been sold under circumstances
under which all of the applicable conditions of Rule 144 ( or any similar
provision there in force) are met; or, (iii) in the opinion of counsel to the
Company such Registrable Securities may permanently be sold without registration
and without any time, volume or manner limitations pursuant to Rule
144.
Effective
March 19, 2010 the Company and YA Ltd. terminated these agreements by mutual
consent.
|
15.
|
Authorized
Share Capital
|
On
September 30, 2009, the Company’s articles of incorporation were amended to
increase the total number of common shares authorized for issuance from
500,000,000 shares to 650,000,000 shares of common stock, par value $0.00001 per
share. On April 23, 2010, the Company’s Board of Directors approved a
reverse stock split of its issued and outstanding common shares. The
total authorized shares was at the same time reduced to
65,000,000. The Board of Directors selected a ratio of one-for-ten
and the reverse split was effective June 20, 2010. As a result, the
total number of shares of all classes of capital stock authorized for issuance
by the Company increased from 700,440,000 shares to 70,044,000 shares with a par
value of $.00001 per share, of which 5,000,000 shares are authorized for
issuance as preferred stock, 65,000,000 shares are authorized for issuance as
common stock, 40,000 shares are authorized for issuance as Series 2 Class B
common stock and 4,000 shares are authorized for issuance as Series 3 Class B
common stock.
F-28
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
The
shares of Series 2 and Series 3 Class B common stock will be non-voting, have
dividend and liquidation rights equal to Class A common stock and be
redeemable. Redemption by the Company shall be mandatory within six
months (or as soon thereafter as permitted by law) following the final
resolution of any successor lawsuit brought by the Company relating to the
subject matter of the Company’s now dismissed lawsuit against certain brokerage
firms (Jag Media Holdings, Inc. v. A.G. Edwards & Sons, et al.) in U.S.
District Court for the Southern District of Texas, which date shall be
determined by the Board of Directors. The redemption price per share
of the Series 2 Class B common stock will be the greater of (i) the par value of
each share or (ii) the amount obtained by dividing (a) 90% of the net proceeds
to the Company of such lawsuit after payment of fees and expenses incurred in
connection with such lawsuit and all taxes on net income accrued or paid with
respect to such amount by (b) the total number of shares of Series 2 Class B
common outstanding. The redemption price per share of the Series 3
Class B common stock will be the greater of (i) par value of each share or (ii)
.0025% of 10% of the net proceeds to the Company of such lawsuit after payment
fees and expenses incurred in connection with such lawsuit and all taxes on net
income accrued or paid with respect to such amount.
Since the
value of the Series 2 and Series 3 Class B common stock is contingent upon the
outcome of a pending or successor litigation, the Company recorded the shares of
Series 2 and Series 3 Class B common stock that were originally issuable during
the year ended July 31, 2003 at their total par value of $4.20. Since
the Company will be required to distribute substantially all the proceeds of the
pending litigation to holders of Series 2 and Series 3 Class B common stock, the
Company had classified the shares as the equivalent of mandatorily redeemable
preferred stock and excluded their carrying value from stockholders’ equity
(deficit) in the accompanying October 31, 2010 consolidated balance sheets
pursuant to the rules and regulations of the Securities and Exchange Commission
and “Accounting for Certain Financial Instruments with Characteristics of Both
Liabilities and Equity.”
|
16.
|
Issuance
of Common Stock
|
During
the year ended October 31, 2010 the Company issued the following common
shares:
|
Year Ended
October 31, 2010
|
||||||||
|
# of shares
|
Amount
|
|||||||
|
Issued
to an employee-shareholder for services rendered
|
21,090 | $ | 23,200 | |||||
|
Issued
to non-employee shareholders for services rendered
|
47,281 | $ | 55,000 | |||||
|
Issuance
to third parties for services rendered
|
95,000 | $ | 77,000 | |||||
|
Issued
on exercise of warrants
|
75,000 | $ | 35,250 | |||||
|
Issued
to unrelated third party for cash
|
3,108,150 | $ | 1,592,575 | |||||
|
Issued
to unrelated third party for subscription receivable
|
230,000 | $ | 115,000 | |||||
The fair
value of shares issued to employee-shareholders and non-employee shareholders
for services rendered were measured at the fair value of the services rendered
on the date rendered.
F-29
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
|
17.
|
Net
Loss per Share
|
The
following table sets forth the computation of weighted-average shares
outstanding for calculating basic and diluted earnings per share
(EPS):
|
Years Ended
October 31,
|
||||||||
|
2010
|
2009
|
|||||||
|
Weighted-average
shares - basic
|
49,624,916 | 30,385,058 | ||||||
|
Effect
of dilutive securities
|
— | — | ||||||
|
Weighted-average
shares - diluted
|
49,624,916 | 30,385,058 | ||||||
Basic
earnings per share “EPS” and diluted EPS for the years ended October 31, 2010
and 2009 have been computed by dividing the net loss available to common
stockholders for each respective period by the weighted average shares
outstanding during that period. All outstanding options, warrants and shares to
be issued upon the exercise of the outstanding options and warrants representing
4,131,974 and 4,206,974 incremental shares respectively have been excluded from
the years ended October 31, 2010 and 2009, respectively, computation of diluted
EPS as they are antidilutive given the net losses generated.
|
18.
|
Commitments
and Contingent Liabilities
|
Leases
The
Company has entered into an operating lease agreement for the use of operating
space.
Aggregate
minimum annual lease commitments of the Company under the non-cancelable
operating lease as of October 31, 2010 are as follows:
|
Year
|
Amount
|
|||
|
2011
|
$ | 36,408 | ||
|
Thereafter
|
— | |||
|
Total
Minimum Lease Payments
|
$ | 36,408 | ||
Lease
expense amounted to $70,633 and $60,883 for the years ended October 31, 2010 and
2009, respectively.
The
preceding data reflects existing leases and does not include replacements upon
their expiration. In the normal course of business, operating leases
are generally renewed or replaced by other leases.
Lawsuits
|
|
a)
|
On
April 22, 2009, the Company was served with a statement of claim from a
former employee claiming compensation for wrongful dismissal and ancillary
causes of action including payment of monies in realization of his
investment in the Company, with an aggregate claim of
$514,000. The Company considers all the claims to be without
any merit, has already delivered a statement of defense and intends to
vigorously defend the action. If the matter eventually proceeds
to trial, the Company does not expect to be found liable on any ground or
for any cause of action.
|
F-30
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
|
|
b)
|
On
January 14, 2010, Flow Capital Advisors Inc. (“Flow Capital”) filed a
lawsuit against JAG Media Holdings Inc. in the Circuit Court of the
17th
Judicial Circuit In and For Broward County Florida (Case No.
10001713). Pursuant to this lawsuit, Flow Capital alleges that
JAG Media Holdings breached a Non-Circumvention Agreement it had entered
into with Flow Capital, dated January 1, 2004. Jag Media
Holdings has moved to dismiss the case because Flow Capital is not
registered to transact business in the State of Florida and is therefore
barred from maintaining the suit under applicable law. The
motion is pending although Flow Capital has since registered, and if the
motion is denied, JAG Media Holdings expects to file an answer asserting
various defenses and vigorously oppose the
suit.
|
On
January 15, 2010, Flow Capital filed a lawsuit against CardioGenics Inc., and
another defendant in the United States District Court for the Southern District
of Florida, Fort Lauderdale Division (Case No.
10-CV-60066-Martinez-Brown). The lawsuit alleges that CardioGenics
(i) breached a Finder’s Fee Agreement in connection with the CardioGenics
Acquisition; and (ii) breached a Non-Circumvention Agreement. Flow
Capital is claiming that it is entitled to the finder’s fee equal to eight
percent (8%) of the JAG Media Holdings shares received by CardioGenics, or the
equivalent monetary value of the stock. CardioGenics has moved to
dismiss the lawsuit for lack of jurisdiction in Florida. That motion
remains pending, although the parties are presently engaged in
discovery.
|
|
c)
|
On
October 26, 2010, Karver International Inc. filed a lawsuit in the 11th
Judicial Circuit in and for Miami-Dade County, Florida against
CardioGenics Holdings Inc. and several other defendants including
affiliates, officers and directors of CardioGenics Holdings,
Inc. The Plaintiff generally alleges that the named defendants
made certain alleged misrepresentations in connection with the purchase of
shares of CardioGenics Holdings Inc. On December 20, 2010,
CardioGenics Holdings Inc. and other defendants filed a motion to dismiss
on the basis that the court lacks personal jurisdiction over most
defendants, that an enforceable forum selection clause requires that the
action be litigated in Ontario, Canada that the doctrine of forum non conveniens
requires dismissal in favor of the Ontario forum, and that the complaint
suffers from numerous other technical deficiencies warranting dismissal
(e.g., failure to attach documents to the Complaint, failure to plead
fraud with particularity, etc.). The motion is currently
pending. Should the motion be denied, CardioGenics Holdings,
Inc. will continue to pursue vigorous defenses to this
action.
|
|
18.
|
Supplemental
Disclosure of Cash Flow Information
|
|
Years
Ended
|
||||||||
|
October 31
|
||||||||
|
2010
|
2009
|
|||||||
|
Cash
paid during the year for:
|
||||||||
|
Interest
|
$ | 35,441 | $ | 4,464 | ||||
|
Income
taxes
|
$ | — | $ | — | ||||
|
Non-cash
financing activity – fixed assets acquired through capital
lease
|
$ | 43,001 | $ | — | ||||
F-31
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
|
19.
|
Assets
and Liabilities from Discontinued
Operations
|
On
February 10, 2010, the Company entered into an LLC Membership Interest Purchase
Agreement with Rothcove Partners LLS (“Rothcove”) pursuant to which the Company
would sell its 100% membership interest in its Pixaya LLC subsidiary to
Rothcove. In consideration for the acquisition of the Pixaya LLC
membership interest, Rothcove assumed $100,000 in accounts payable to Pixaya LLC
and its subsidiary Pixaya (UK) Limited (collectively “JAG
Media”). The transaction closed on February 11, 2010.
As a
result of the sale of JAG Media, certain reclassifications of assets,
liabilities, revenues, costs and expenses have been made to the prior period
consolidated financial statements to reflect the operations of JAG Media as
discontinued operations. Specifically, the Company has reclassified
the results of operations of JAG Media for all periods presented to “Loss from
Discontinued Operations” within the consolidated statements of
operations. In addition, the remaining liabilities of the business
divested in February 2010 have been reclassified to “Liabilities of Discontinued
Operations”. Revenues from discontinued operations were $14,852 and
$21,539 for the years ended October 31, 2010 and 2009,
respectively. Loss from discontinued operations was ($12,355) for the
year ended October 31, 2010.
At
October 31, 2010 and 2009, Liabilities from Discontinued Operations comprise the
following:
|
October 31,
|
||||||||
|
2010
|
2009
|
|||||||
|
Liabilities
|
||||||||
|
Accounts
Payable
|
$ | — | $ | 100,000 | ||||
|
Total
Liabilities from Discontinued Operations
|
$ | — | $ | 100,000 | ||||
|
20.
|
Subsequent
Events
|
On
December 2, 2010, the Company issued 100,000 common shares per an investors’
relations consulting agreement.
On
December 2, 2010, the Company issued 600,000 common shares for cash
consideration of $300,000.
|
21.
|
Segment
Reporting
|
The
Company’s operating segments are regularly reviewed by the chief operating
decision maker for purposes of allocating resources and assessing
performance.
The
Company’s financial reporting is organized into two business segments: In Vitro
Diagnostics and Jag Notes.
The
information presented below includes certain expense allocations between the
corporate office and the operating business segments. The information is
presented after all intercompany and intersegment eliminations and is therefore
not necessarily indicative of the results that would be achieved had the
business segments been stand-alone businesses.
F-32
|
CardioGenics
Holdings Inc.
|
|
(A
Development Stage Company)
|
|
Notes
to Consolidated Financial Statements
|
|
October 31, 2010 and
2009
|
The
results of operations and other data for the two operating segments for the
years ended October 31, 2010 and 2009 are as follows:
|
In Vitro
Diagnostics
|
Jag Notes
|
Consolidated
|
||||||||||
|
2010
|
|
|
||||||||||
|
General
and administrative
|
$ | 1,160,509 | $ | — | $ | 1,160,509 | ||||||
|
Research
and development
|
692,036 | — | 692,036 | |||||||||
|
Depreciation
expense and amortization of intangibles
|
129,300 | — | 129,300 | |||||||||
|
Loss
from continuing operations
|
(1,981,845 | ) | — | (1,981,845 | ) | |||||||
|
Discontinued
Operations:
|
||||||||||||
|
Loss
from discontinued operations
|
— | (12,355 | ) | (12,355 | ) | |||||||
|
Loss
from operations
|
$ | (1,981,845 | ) | $ | (12,355 | ) | $ | (1,969,490 | ) | |||
|
Interest
expense
|
$ | 30,166 | $ | — | $ | 30,166 | ||||||
|
Capital
expenditures — property and equipment
|
11,058 | — | 11,058 | |||||||||
|
Capital
expenditures — patent application costs
|
37,091 | — | 37,091 | |||||||||
|
Total
assets
|
2,486,135 | — | 2,486,135 | |||||||||
|
In Vitro
Diagnostics
|
Jag Notes
|
Consolidated
|
||||||||||
|
2009
|
|
|
||||||||||
|
General
and administrative
|
$ | 1,597,010 | $ | — | $ | 1,597,010 | ||||||
|
Research
and Development
|
1,572,337 | — | 1,572,337 | |||||||||
|
Goodwill
impairment charges
|
— | 12,780,214 | 12,780,214 | |||||||||
|
Depreciation
expense and amortization of intangibles
|
54,141 | — | 54,141 | |||||||||
|
Loss
from continuing operations
|
(3,223,488 | ) | (12,780,214 | ) | (16,003,702 | ) | ||||||
|
Discontinued
Operations:
|
||||||||||||
|
Loss
from discontinued operations
|
— | (115,407 | ) | (115,407 | ) | |||||||
|
Loss
from operations
|
$ | (3,223,488 | ) | $ | (12,895,621 | ) | $ | (16,119,109 | ) | |||
|
Interest
expense
|
$ | 571,840 | $ | — | $ | 571,840 | ||||||
|
Capital
expenditures — property and equipment
|
8,950 | — | 8,950 | |||||||||
|
Capital
expenditures — patent application costs
|
15,164 | — | 15,164 | |||||||||
|
Total
assets
|
2,839,865 | 47,397 | 2,887,262 | |||||||||
F-33
SIGNATURES
Pursuant
to the requirements of Section 13 or 15(d) of the Securities Exchange Act of
1934, the Registrant has duly caused this report to be signed on its behalf by
the undersigned, thereunto duly authorized.
|
CARDIOGENICS
HOLDINGS INC.
|
||
|
By:
|
/s/
Yahia Gawad
|
|
|
|
Yahia
Gawad
|
|
|
|
Chief
Executive Officer
|
|
|
Dated:
January 31, 2011
|
||
Pursuant
to the requirements of the Securities Exchange Act of 1934, this report has been
signed below by the following persons on behalf of the Registrant and in the
capacities and on the date indicated.
|
SIGNATURE
|
TITLE
|
DATE
|
||
|
/s/
Yahia
Gawad
|
Chief
Executive Officer
|
January
31, 2011
|
||
|
Yahia
Gawad
|
||||
|
/s/
James
Essex
|
Chief
Financial Officer
|
January
31, 2011
|
||
|
James
Essex
|
||||
|
|
|
EXHIBIT
INDEX
|
21.1
|
Subsidiaries
of Registrant
|
|
|
23.1
|
Consent
of J.H. Cohn LLP
|
|
|
23.2
|
Consent
of BDO Dunwoody LLP
|
|
|
31.1
|
Section
302 Certification of Chief Executive Officer
|
|
|
31.2
|
Section
302 Certification of Chief Financial Officer
|
|
|
32.1
|
Section
906 Certification of Chief Executive Officer and Chief Financial
Officer
|
