Attached files
| file | filename |
|---|---|
| EX-32 - UV FLU TECHNOLOGIES INC | v208037_ex32.htm |
| EX-31.1 - UV FLU TECHNOLOGIES INC | v208037_ex31-1.htm |
| EX-31.2 - UV FLU TECHNOLOGIES INC | v208037_ex31-2.htm |
UNITED
STATES SECURITIES AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM
10-K
(Mark
One)
|
x
|
ANNUAL
REPORT PURSUANT TO SECTION 13 OR 15(d) OF
THE
SECURITIES EXCHANGE ACT OF 1934
|
For
the fiscal year ended September 30, 2010
Commission
File Number: 333-140322
UV
FLU TECHNOLOGIES, INC.
(Exact
name of registrant as specified in its charter)
|
Nevada
|
98-0496885
|
|||
|
(State
or Other Jurisdiction of
|
(I.R.S.
Employer
|
|||
|
Incorporation
or Organization)
|
Identification
Number)
|
|||
|
1694
Falmouth Road, Suite 125
Centerville,
Massachusetts 02632-2933
|
||||
|
(Address
of principal executive offices) (Zip Code)
|
||||
|
(508)
362-5455
|
||||
|
(Registrant’s
telephone number, including area
code)
|
||||
Securities
registered pursuant to Section 12(b) of the Act:
|
None
|
None
|
|
|
(Title
of each class)
|
(Name
of each exchange on which
registered)
|
Securities
registered pursuant to Section 12(g) of the Act:
Common
Stock, $0.001 par value
(Title of
class)
Indicate
by check mark if the registrant is a well-known seasoned issuer, as defined in
Rule 405 of the Securities Act.
¨
Yes x No
Indicate
by check mark if the registrant is not required to file reports pursuant to
Section 13 or Section 15(d) of the Act. ¨
Yes x No
Indicate
by check mark whether the registrant (1) has filed all reports required to be
filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the
preceding twelve months (or for such shorter period that the registrant was
required to file such reports), and (2) has been subject to such filing
requirements for the past 90 days. x
Yes o
No
Indicate
by check mark whether the registrant has submitted electronically and posted on
its corporate Web site, if any, every Interactive Data File required to be
submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this
chapter) during the preceding 12 months (or for such shorter period that the
registrant was required to submit and post such files). Yes o No
o
Indicate
by check mark if disclosure of delinquent filers pursuant to Item 405 of
Regulation S-K, is not contained herein, and will not be contained, to the best
of the registrant’s knowledge, in definitive proxy or information statements
incorporated by reference in Part III of this Form 10-K or any amendment of this
Form 10-K.
¨
Indicate
by check mark whether the registrant is a large accelerated filer, an
accelerated filer, a non-accelerated filer, or a smaller reporting
company. See the definitions of “large accelerated filer” and “small
reporting company” in Rule 12b-2 of the Exchange Act.
|
Large
accelerated filer
¨
|
Accelerated
filer
¨
|
Non-accelerated
filer*
¨
|
Smaller
reporting company
x
|
|
*(Do
not check if a smaller reporting
company)
|
|||
Indicate
by check mark whether the registrant is a shell company (as defined in Rule
12b-2 of the Exchange Act) ¨
Yes x No.
The
aggregate market value of the common stock held by non-affiliates as of March
31, 2010 (the last trading day of the second quarter) was $13,073,000, based on
the last sale price of common stock sold.
As of
January 10, 2011, the last practicable date, 45,080,000 shares of the
registrant’s Common Stock were outstanding at a par value of
$0.001.
DOCUMENTS
INCORPORATED BY REFERENCE: Exhibits incorporated by reference are referred to
under Part IV.
TABLE
OF CONTENTS
|
Page
|
||
|
PART
I
|
||
|
Item
1. Business
|
4
|
|
|
Item
1A. Risk Factors
|
11
|
|
|
Item
1B. Unresolved Staff Comments
|
17
|
|
|
Item
2. Properties
|
17
|
|
|
Item
3. Legal Proceedings
|
17
|
|
|
Item
4. Reserved
|
17
|
|
|
PART
II
|
||
|
Item
5. Market For Registrant’s Common Equity, Related
Stockholder Matters And Issuer Purchases of Equity
Securities
|
18
|
|
|
Item
6. Selected Financial Data
|
18
|
|
|
Item
7. Management’s Discussion and Analysis of
Financial Condition and Results of Operations
|
18
|
|
|
Item
7A. Quantitative and Qualitative Disclosures About Market
Risk
|
23
|
|
|
Item
8. Financial Statements and Supplementary
Data
|
23
|
|
|
Item
9. Changes in and Disagreements With Accountants on
Accounting and Financial Disclosure
|
24
|
|
|
Item
9A. Controls and Procedures
|
24
|
|
|
Item
9B. Other Information
|
25
|
|
|
PART
III
|
||
|
Item
10. Directors, Executive Officers and Corporate
Governance
|
25
|
|
|
Item
11. Executive Compensation
|
27
|
|
|
Item
12. Security Ownership of Certain Beneficial Owners and
Management and Related Stockholder Matters
|
29
|
|
|
Item
13. Certain Relationships and Related Transactions, and
Director Independence
|
30
|
|
|
Item
14. Principal Accounting Fees and Services
|
31
|
|
|
PART
IV
|
||
|
Item
15. Exhibits, Financial Statement Schedules
|
32
|
|
|
Index
to Financial Statements
|
F-1
to F-12
|
|
|
Signatures
|
||
|
Exhibits
|
2
Statement
Regarding Forward-Looking Statements
The
statements contained in this report on Form 10-K that are not purely historical
are forward-looking statements within the meaning of applicable securities
laws. Forward-looking statements include statements regarding our
“expectations,” “anticipation,” “intentions,” “beliefs,” or “strategies”
regarding the future, whether or not those words are
used. Forward-looking statements also include statements regarding
revenue, margins, expenses, and earnings analysis for fiscal 2010 and
thereafter; anticipated levels of future revenues and earnings from the
operations of UV Flu Technologies, Inc. (the “Company,” “we,” “us,” or “our”);
and projected costs and expenses related to our operations,
liquidity, capital resources, availability of future equity capital on
commercially reasonable terms. All forward-looking statements
included in this report are based on information available to us as of the
filing date of this report, and we assume no obligation to update any such
forward-looking statements. Our actual results could differ
materially from the forward-looking statements. Among the factors
that could cause actual results to differ materially are the factors discussed
in Item 1A. Risk Factors.
3
PART
I
ITEM
1. BUSINESS
Background
UV Flu
Technologies, Inc. (“we”, “us”, “our,” or the “Company”) was organized under the
laws of the State of Nevada on April 4, 2006 under the name “Northwest Chariots,
Inc.” We were engaged in the business of renting and selling
electrically powered human transporters, like electric bicycles, chariots, and
quads. Following our fiscal year ended September 30, 2009, we decided
to change our product mix to air purification products and to focus on the
research, development, manufacturing, and sales of air purification systems and
products.
In
furtherance of our business objectives, on November 12, 2009, we effected a
32-for-1 forward stock split of all our issued and outstanding shares of common
stock, and we merged with our wholly-owned subsidiary, UV Flu Technologies,
Inc., for the purposes of effecting a name change to “UV Flu Technologies,
Inc.”
Effective
November 15, 2009, we acquired AmAirpure Inc.’s air purification technology,
product, inventory, and certain equipment pursuant to an Asset Purchase
Agreement with AmAirpure, Inc. We issued 15,000,000 shares of our
common stock to shareholders of AmAirpure in connection with the asset
acquisition. Additionally, on November 25, 2009, we entered into a
Distribution Agreement with Puravair Distributors LLC (“Puravair”) where we
appointed Puravair as our exclusive master distributor for our Viratech UV-400
product and our other products for the professional, medical, and commercial
markets in the U.S. and Canada. On September 30, 2010, we terminated
our Distribution Agreement with Puravair and began adding new distributors,
which totaled five as of year end.
The
latest production runs of our Viratech UV-400 product incorporate our patented
UV bacteria killing technology, which has been cleared by the FDA for use as a
medical device. In June 2010, we expanded our market reach by
introducing the latest generation of our Viratech UV-400 product into the
residential and hospitality markets.
On
October 28, 2010, we entered into a binding letter of intent with The Red Oak
Trust (“Red Oak”) (the “LOI”) in connection with our proposed acquisition of one
hundred percent (100%) of the issued and outstanding units of RxAir Industries,
LLC, a Nevada limited liability company (“RxAir”), which is wholly owned by Red
Oak (the “Acquisition”). At the closing of the Acquisition, RxAir
will become a wholly-owned subsidiary of the Company. We are
currently renegotiating the terms of the LOI and we intend to enter into a
definitive agreement soon.
We
currently have limited revenues from operations. We have begun
marketing and distributing our products and expect to conduct some targeted
direct selling in the next several months.
Our
Solution
Around
the world, there is a growing awareness of the increasingly poor air quality,
particularly with the recent outbreak of H1N1 swine flu and other respiratory
pathogens. The public has long been aware of the dangers associated
with outdoor air pollution, but never before has there been such growing public
concern about the quality of indoor air as well. Today’s lifestyles,
coupled with modern building construction practices have created significant
challenges in maintaining healthy indoor air. Poor indoor air quality
has been proven scientifically to cause increased asthma and allergy related
symptoms, as well as contribute to the spread of disease. These
factors have created a large and growing need for products which will improve
indoor air quality in the workplace and at home.
We fill
the need for improved air purification systems in the workplace and at home with
proprietary technology utilizing high-energy ultraviolet radiation (UV) inside a
“killing chamber” which destroys airborne bacteria, deactivates allergens, and
mold. The product also reduces odors, and the concentrations of
Volatile Organic Compounds (VOC’S). We plan to develop and market products,
which will improve indoor air quality effectively, efficiently, economically and
do so in an environmentally friendly way.
Our
product safely kills over 99% of airborne bacteria. Extensive
independent laboratory testing confirms that this unique technology kills other
airborne contaminants as well.
4
Market
Overview
The
market for air purification equipment has been gaining momentum as a result of
rising concerns over indoor air quality and increasing health consciousness
among consumers. Products within this group that until recently had
been viewed as a luxury are now found in an increasing number of homes and
commercial workspaces. As a result, the North American air
purification equipment market is expected to expand significantly.
The U.S.
indoor air quality market generated $7.7 billion in 2008, with the equipment
segment accounting for $3.6 billion. Continuing media attention given to the
health effects of toxic mold, the outbreak of infectious diseases such as swine
flu, and the increase in chronic respiratory diseases such as asthma have
resulted in new interest in, and attention to, indoor air
quality. Building owners and operators are expected to purchase
growing quantities of indoor air quality-related equipment in the hope of
reducing or eliminating these contaminants from their buildings.
Employers
have incentive to keep workers healthy. In 2008, 425 million sick
days were taken by employees, costing an estimated $60 billion in lost
productivity. “Presenteeism,” a new term coined for employees working
while unhealthy, is even worse for employers and the healthcare system, costing
approximately $160 billion annually in lost productivity. (Kalorama
Information, “The Market for Wellness Programs”, September 2009).
Increased
media attention and growing public concern over pandemics, antibiotic resistant
superbugs, asthma, allergies, tuberculosis, Sick Building Syndrome, and toxic
mold are spurring governments, employers, and homeowners to take action to
safeguard their health. Demand for products that combat the spread of
airborne illness is rising exponentially, as starkly highlighted by public
reaction to the outbreak of H1N1. The Wall Street Journal reported
the pandemic will create a $7 billion windfall for manufacturers of swine flu
vaccine this year.Between July and September of 2009, shipments of hand
sanitizer rose a remarkable 129%.
This
heightened awareness has spread to the growing $7.7 billion indoor air quality
market, increasing demand for products that improve air quality and remove
airborne pathogens in commercial buildings, hospitals, schools and
homes. Poor indoor air quality is scientifically proven to cause the
spread of infectious disease and increase asthma and allergy
symptoms. The Environmental Protection Agency reports the air inside
structures, where people now spend approximately 90% of their time, poses a
greater threat to health than outdoor air.
The air
cleaner market is very large with multiple levels. It encompasses an
extremely wide range of products, designed to improve the quality of indoor
air. In order to deal with the increasingly complex issue of indoor
air quality, commercial enterprises must often purchase and use multiple
solutions in their effort to provide a safe indoor
environment. Including the products designed to freshen the air,
surface sprays, portable air cleaners, filters for HVAC systems, other
“specialty” solutions for the removal of smoke and airborne microorganisms, the
total market for air cleaning products tops an estimated $6 billion per
year.
As
summarized below, the commercial market (estimated over $5 billion) for air
purification products can be broken into three different target segments: (1)
Medical; (2) Hospitality; and (3) Office. A listing of selected
market opportunities within each segment can be found below. As
awareness of poor indoor air quality and its impact on worker productivity
grows, demand for products, which go beyond masking and filtration will continue
to grow.
Commercial
Market
|
Medical
|
Hospitality
|
Office
|
||
|
Hospitals
|
Hotels
|
Commercial
|
||
|
Nursing
Homes
|
Motels
|
Small
Office
|
||
|
Medical
Offices
|
Inns
|
Home
Office
|
||
|
Dentists
|
Restaurants
|
Day
Care
|
||
|
Clinics
|
|
|
According
to IMR Research, the consumer market is estimated to be in the millions of units
and over $1 billion in sales. This market has been growing at an
annual rate of 17% since 1992. Products utilizing HEPA or HEPA-type
filters dominate the market, representing over two-thirds of unit
sales.
5
About 80%
of the unit volume sells at prices below $200. These products are
typically sold through major mass merchants including home center outlets,
specialty catalogs and on the Internet.
While
representing slightly less than 35% of the unit sales, products retailing over
$200 represent more than 50% of the dollar volume. This includes
high-end HEPA air cleaners, electrostatic devices, and, previously, ozone
generators. In addition to traditional retail outlets handling upper
end products, they can also be found in specialty catalogs and sold on a
direct-to-consumer basis. With the recent disappearance of a company
that held a large market share in this sector of the market, a significant and
growing market exists for premium price air cleaners.
Consumer
Market
Products
utilizing filters (standard and HEPA) dominate the consumer
market. Electrostatic products are starting to appear in a portable
form. There are currently only two major brands in this market
sector, so there is also a growing opportunity and demand for us to develop
consumer targeted indoor air quality products.
Scientific
Overview
We
develop highly innovative germicidal air purification technology that disinfects
indoor air by deactivating allergens and killing airborne pathogens including
bacteria, viruses, and mold. Our flagship product, the ViraTech
UV-400, utilizes high-intensity germicidal ultraviolet radiation (UV-C) inside a
killing chamber that goes beyond filtering to trap and destroy harmful microbes.
Extensive independent testing by EPA and FDA certified laboratories confirms the
proprietary system captures and kills airborne bacteria, including Bacillus
subtilis, Pseudomonas aeruginosa, and Staphylococcus aureus, at rates exceeding
99.2% on a first-pass basis. We recently concluded independent
EPA/FDA certified laboratory tests, showing the rates of inactivation of a MS-2
virus surrogate were almost identical to our results on the inactivation of
bacteria.
We
combine our air purification technology (see diagram below), a sophisticated
electronic ballast system with an electronic control module in a product which
is totally effective yet user-friendly. The technology uses
Ultraviolet C (UVC) in a proprietary, replaceable cartridge which provides 12
months of continuous operation, 24 hours a day, seven days a
week. Inside the cartridge, pathogens and bacteria and, in fact,
anything with a DNA is killed or neutralized. A gross particulate
screen removes the remaining debris from the air stream. This sealed
cartridge can be discarded in the regular trash at the end of its useful
life.
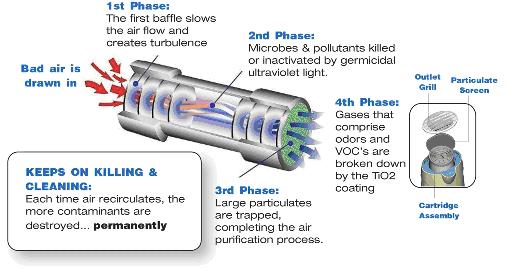
During
the actual purification process, air enters through the base of the
purifier. A quiet fan, mounted at the unit’s lower end, pushes
incoming contaminated air into the bottom of the replaceable
cartridge. Inside, the air flows through the first of two inline
baffle sets. The chamber, created by these baffles, slows the airflow
and provides turbulence necessary for proper elimination of
impurities. Next, the air stream enters the main
chamber. Here, impurities in the air are bombarded with germicidal
UVC, killing the bacteria and neutralizing the impurities. The
interior of the kill chamber is coated with TiO2, which then reduces the
concentrations of odors and Volatile Organic Compounds (VOC’s), by reacting with
the gases that make contact with the interior surface of the chamber, thus
breaking them down to the molecular level. After purification in the
main chamber, the air stream passes through a second set of inline baffles,
which refocuses the flow. It then exits the unit through a gross
particulate screen. This screen removes neutralized pathogens and
other particles left in the air stream. The final air stream contains
only purified air.
6
Electronic
controls insure that our product is completely effective during the life of the
cartridge, allows performance monitoring of all major electrical components,
simplifies operation and endows the units with appropriate safety
features. An indicator light provides information regarding the state
of the cartridge and indicates when a replacement is necessary.
Product
The
ViraTech UV-400
The
ViraTech UV-400 will be the lead product we produce and
distribute. The ViraTech UV-400 portable model is 32 inches tall and
uses a 6 inch diameter cartridge. The second product currently being
designed is approximately 1/3 smaller and will use a 4.5
inch diameter cartridge (see rendering below). For the U.S. market,
all portable models operate on 110/120 volt current and do not require any
special wiring. In each product family, there is a design provision
for 220/240 volt operation, which allows for adaptation to international
standards or use in heavy-duty commercial applications. Each unit is
designed to run continuously and will have a minimum active life of 5
years. Cartridge replacement is signaled when as the 9000 lamp hour
mark approaches a green light on the front of the unit turns red, and when the
useful life is hit, the unit shuts down until the cartridge is
replaced. This ensures that the unit maintains adequate germicidal UV
strength. Recommended replacement frequency assumes 24 hour per day,
seven days a week usage. Because the units run with less resistance
than traditional HEPA air filters, the fan is smaller, which results in a
quieter product. Depending on room size, each unit will recycle air
in a room at the rate of 5 to 10 times per hour.
Large
and Small Portable Units
The
Company has designed a revolutionary smaller unit, specifically for the
residential marketplace. The unit will be approximately 27” high, and
will combine the Company’s patented UV killing chamber technology with state of
the art HEPA filter technology. Thus, the unit will “Kill and
Trap.” The unit will be designed to be sold at a lower price point
and will have independent EPA/FDA certified lab results to support every
claim. The unit includes a molded handle and an aesthetically
pleasing design, which will make it desirable for homes, hotel rooms, and
offices. The Company has already been in contact with noted Allergy
Specialists that are interested to get prototypes. The product will
be sold on infomercials and through the Company’s other distribution
channels. The product will be sold at a similar price point to the
Sharper Image ‘Ion Breeze,” which sold almost 3 million units.

Sales
and Distribution Channels
We will
focus initially on the commercial market, targeting medical, hospitality, and
commercial property customers. We will sell and lease our products
directly or through distributors to medical accounts including hospitals, doctor
and dentist offices, clinics, and nursing homes. Initial sales are
anticipated as the result of interaction with key hospital and nursing home
administrators as well as medical equipment distributors and medical sales
representatives. We plan to leverage longstanding contacts within the
healthcare industry to drive placement.
We are
seeking a master distribution agreement with a large medical equipment supplier
with an active sales force. We aim to form sector-specific
distribution agreements with established suppliers and distributors targeting
the hospitality, food service, and lodging markets. We plan to
strategically target the large consumer market for indoor air quality products
once commercial sales have progressed.
7
Customers
We
currently have units in operation at the Seagate Hotel, Boca Raton, Florida; the
15 Beacon Hotel, Boston, Massachusetts; the Miami Anatomical Research Center;
and several other locations.
Competition
Awareness
of the effects of poor indoor air quality continues to grow and will push
customers to look for more efficacious products. We believe that
companies currently supplying air purification products to commercial accounts
will be looking for additional products to sell and new sources of on-going
revenue. We believe our ViraTech UV-400 product represents a
significant opportunity to distributors for first sales as well as the on-going
replacement cartridge sales volume into a growing installed user
base.
Our
products compete broadly with other current companies offering air purification
products, including companies that offer UV air purification
products. Honeywell was one of the best-known brand names in the
commercial segment of the market. 3M Corporation purchased the
commercial air treatment division of Honeywell and now markets their product
line. In the consumer market segment, we compete with Honeywell,
Duracraft, and Enviracaire brands, all of which compete at the middle and
upper-middle price points with HEPA-based products and are offered by Kaz,
Incorporated. Kaz purchased Honeywell’s consumer products division
and continues to market product under the existing brand names along with a wide
variety of other brands. Recently, Alpine Industries left the
consumer segment of the market, leaving an opening for new and existing
companies to assume Alpine’s market share.
These
products are in direct competition with our products for market
share. We have made comparisons of competitive air masking and
filtration products to determine the extent to which these competitive products
achieve their advertising claims. We believe we are one of the few
companies that publish laboratory reports of our products performance
capabilities. Based upon these comparisons, we do not believe there
is any existing product which can provide safe, effective operation and still
deliver high levels of purification against four forms of airborne contaminants
(molds, spores, viruses, and bacteria). At one end of the product
spectrum, the market includes the replaceable air freshening devices found in
restrooms that are intended to mask lavatory and other odors. While
masking odors, these products are not capable of attacking the source of the
odor. The products also require frequent and costly
replacement.
The
predominant middle market air cleansing product forms are HEPA type air cleaners
and filtration devices built into HVAC systems. Filtration products
have some effectiveness against airborne particulates, but they do little to
combat odors or pollutants like smoke, bacteria and viruses. Many of
these filtering systems are out-of-date. They all require regular
filter replacement and maintenance, which ultimately impacts their level of
effectiveness. Changing the filter requires wearing protective
clothing and a breathing device. Filters connected to central HVAC
systems represent significant sales potential and costs to customers because of
the sizable installed base of such systems.
At the
high end of the spectrum are custom-designed air purification devices, which
include large freestanding germicidal UV or ozone generating
machines. These machines, which represent a very small portion of the
market, are expensive (from $1,000 to $10,000 per unit). Since design
and technology of custom units exposes the environment to the UV radiation or
ozone, to be effective either (1) they require evacuation of a room or house to
run safely at high concentrations of ozone or risk of eye damage to the UV lamp
or (2) their performance against airborne contaminants is relatively
poor.
Research
and Development
Our
research and development activities are focused principally on the development
of new products that serve the air purification market and on significant
upgrades to our existing products. We have completed the design of
our second product, a smaller, lower cost version of the ViraTech
UV-400. We expect to begin shipping the new model into commercial
accounts before the end of September 2011.
8
Manufacturing
Our
manufacturing strategy is to utilize high quality, low cost contract
manufacturers to provide the routine production of our products. We
will outsource the manufacture of the majority of our products.
Quality
Control
Our
quality strategy emphasizes rigorous internal and independent laboratory testing
to maintain the highest levels of quality control for our
products. To insure that the proper style and feature set were
identified, a number of potential design combinations were
developed. These initial designs were presented to consumers in
special triad focus group session and played a major role in selection of the
final design. These groups also helped to identify the message, which
will be used when we begin our public relations and consumer advertising
programs.
In order
to eliminate any potential product liability issues, extensive testing has been
done with existing prototypes. This testing confirms that all
products meet worldwide electrical and safety standards. Separately,
each electrical component to be used will be certified by its manufacturer as
meeting ETL and/or UL standards.
Our
quality system has been created to be harmonized with national and international
standards and is focused to ensure it is appropriate for the specific devices we
manufacture. Our corporate quality policies govern the methods used
in, and the facilities and controls used for, the design, manufacture,
packaging, labeling, storage, installation, and servicing of all finished
devices intended for human use. These requirements are intended to
ensure that finished devices will be safe and effective and otherwise in
compliance with the Federal Food, Drug, and Cosmetic Act and other governmental
agencies.
In order
to validate the efficacy of our technology, we conducted numerous tests at
independent, FDA certified laboratories between 1996 and 2007. We
will continue to conduct similar tests in the future.
Independent
EPA and FDA certified laboratory testing (results below) confirms the system
captures and kills airborne bacteria, including Bacillus subtilis, Pseudomonas
aeruginosa, and Staphylococcus aureus. The Company recently announced
that additional testing was performed at the same certified test facility
showing rates of inactivation for a MS-2 virus surrogate comparable to the
results shown with bacteria.
NorthEast
Laboratories Test Results on the ViraTech UV-400 Unit
|
Microbe
|
Population
Before
|
Population
After
|
Inactivation
%
|
Average
%
|
k
cm2/μW
-s
|
Effective
Dose μW-
s/cm2
|
||||||
|
Bacillus
subtilis
|
420
|
<1
|
99.76
|
99.71
|
0.001686
|
3583
|
||||||
|
300
|
<1
|
99.67
|
3383
|
|||||||||
|
Pseudomonas
|
8370
|
29
|
99.65
|
99.72
|
0.002375
|
2385
|
||||||
|
Aeruginosa
|
140,000
|
300
|
99.79
|
2588
|
||||||||
|
Klebsiella
pneumoniae
|
19,000
|
79
|
99.58
|
99.10
|
0.000548
|
10005
|
||||||
|
20,360
|
280
|
98.62
|
7822
|
|||||||||
|
Staphylococcus
aureus
|
600
|
7
|
98.83
|
99.30
|
0.003475
|
1281
|
||||||
|
16,000
|
36
|
99.78
|
1754
|
|||||||||
|
Effective
Mean UV Dose
|
4100
|
|||||||||||
|
UVGI
Rating Value (URV)
|
15
|
|
Note:
Results for Bacillus represent a minimum.
Source: Northeast Laboratories, Inc.
129 Mill Street Berlin, CT
06037
|
Regulation
Our
products are subject to regulation by numerous government agencies, including
the FDA and comparable foreign agencies. To varying degrees, each of
these agencies requires us to comply with laws and regulations governing the
development, testing, manufacturing, labeling, marketing, distribution,
installation and servicing of our research, investigational, and
commercially-distributed medical devices. These international,
national, state, and local agencies set the legal requirements for ensuring our
products are safe and effective. Virtually every activity associated
with the manufacture and sale of our products are scrutinized on a defined basis
and failure to implement and maintain a quality management system could subject
us to civil and criminal penalties.
9
Our
ViraTech UV-400 product was issued by the FDA as a Class II medical device in
November 2008. FDA clearance to sell our product as a Class II
medical device provides invaluable credibility in the marketplace. By
granting a listing, the FDA indicates it has reviewed all aspects of a product,
including efficacy of the technology, independent test results and product
safety to insure that the product complies with our claims. Few air
purification products are listed by the FDA, and it is extremely important that
we expend the resources necessary to maintain this listing as a Class II medical
device with the FDA. The Company has recently concluded independent
lab tests, which show the rates of inactivation on a MS-2 virus surrogate were
comparable to the results shown with bacteria. Although the Company
makes no claims for the inactivation of viruses, it plans on submitting the
results for further FDA evaluation.
Because
Class II devices have a lower potential safety risk to the patient, user, or
caregiver, a full premarket analysis are not required for a Class II
device. A premarket notification, known as a 510(k) submission, is
required to demonstrate that the device is as safe and effective as a
substantially equivalent medical device that has been legally marketed in the
U.S. prior to May 29, 1976. Once the FDA has notified us that the
product file has been cleared, the medical device may be marketed and
distributed in the U.S. Some products that have minimal risk to the
intended user are deemed by the FDA as exempt from the FDA approval or clearance
process.
Failure
to comply with applicable FDA requirements can result in fines, injunctions,
civil penalties, recall, or seizure of products, total or partial suspension of
production, or loss of distribution rights. It may also include the
refusal of the FDA to grant approval of a PMA or clearance of a
510(k). Actions by the FDA may also include withdrawal of marketing
clearances and possibly criminal prosecution. Such actions, if taken
by the FDA, could have a material adverse effect on our business, financial
condition, and results of operation.
Internationally,
we will be required to comply with a multitude of other regulatory requirements
similar to those of the FDA before we are legally able to market and sell our
products in such international markets.
Environmental
Laws
We do not
manufacture the products that we sell and are therefore not subject to
environmental laws that regulate the manufacture of products. The
plants that manufacture our products may be subject to environmental regulations
and will have to comply with such regulations in order to deliver marketable
products to us. We may be required to comply with national and
international environmental regulations related to shipping, storage and
disposal of our products, and our quality management system will ensure that we
are in compliance with all relevant environmental laws.
Patents
and Proprietary Rights
We own
the rights to U.S. Patent No. 6939397 with 43 claims covering innovative
removable cartridge, housing, UV chamber, UV radiation source, and baffle
technology.
While a
patent has been issued, we realize that (a) we will benefit from patents issued
only if we are able to market our products in sufficient quantities of which
there is no assurance; (b) substitutes for these patented items, if not already
in existence, may be developed; (c) the granting of a patent is not a
determination of the validity of a patent, such validity can be attacked in
litigation or we or owner of the patent may be forced to institute legal
proceedings to enforce validity; and (d) the costs of such litigation, if any,
could be substantial and could adversely affect us.
Backlog
We
currently have no backlog of orders.
Employees
As of
September 30, 2010, we had no full-time employees, although we engage
contractors as needed and each of our officers and directors devotes a portion
of his and her time to the affairs of our Company.
10
Where
you can find more information
We are
required to file annual reports on Form 10-K, quarterly reports on Form 10-Q,
current reports on Form 8-K, and other information with the Securities and
Exchange Commission (the “SEC”). The public can obtain copies of
these materials by visiting the SEC’s Public Reference Room at 100 F Street, NE,
Room 1580, Washington, DC 20549, by calling the SEC at 1-800-732-0330, or by
accessing the SEC’s website at http://www.sec.gov. In addition, as
soon as reasonably practicable after these materials are filed with or furnished
to the SEC, we will make copies available to the public free of charge through
our website, http://www.uvflutech.com. The information on our website
is not incorporated into, and is not part of, this annual report.
ITEM
1A. RISK
FACTORS
With
the exception of historical facts stated herein, the matters discussed in this
report on Form 10-K are “forward looking” statements that involve risks and
uncertainties that could cause actual results to differ materially from
projected results. Such “forward looking” statements include, but are
not necessarily limited to statements regarding anticipated levels of future
revenues and earnings from the operations of the Company, projected costs and
expenses related to our operations, liquidity, capital resources, and
availability of future equity capital on commercially reasonable
terms. Factors that could cause actual results to differ materially
are discussed below. We disclaim any intent or obligation to publicly
update these “forward looking” statements, whether as a result of new
information, future events, or otherwise.
An
investment in our common stock is subject to risks inherent to our
business. The material risks and uncertainties that management
believes affect us are described below. Before making an investment
decision, you should carefully consider the risks and uncertainties described
below together with all of the other information included or incorporated by
reference in this report. The risks and uncertainties described below
are not the only ones facing us. Additional risks and uncertainties
that management is not aware of or focused on or that management currently deems
immaterial may also impair our business operations. This report is
qualified in its entirety by these risk factors.
If any of
the following risks actually occur, our financial condition and results of
operations could be materially and adversely affected. If this were
to happen, the value of our common stock could decline significantly, and you
could lose all or part of your investment.
Our
limited operating history may not serve as an adequate basis to judge our future
prospects and results of operations.
We have a
limited operating history. As such, our historical operating results
may not provide a meaningful basis for evaluating our business, financial
performance and prospects. Accordingly, you should not rely on our
results of operations for any prior periods as an indication of our future
performance. Our operations will be subject to all the risks inherent
in the establishment of a developing enterprise and the uncertainties arising
from the absence of a significant operating history. We are in the
development stage and potential investors should be aware of the difficulties
normally encountered by enterprises in the development stage. If our
business plan is not successful, and we are not able to operate profitably,
investors may lose some or all of their investment in our company.
We
cannot accurately predict future revenues or profitability in the emerging
market for air purifiers.
The
market for ultra violet indoor air purifiers is rapidly evolving. As
is typical for a rapidly evolving industry, demand and market acceptance for
recently introduced products are subject to a high level of
uncertainty. Moreover, since the market for our products is evolving,
it is difficult to predict the future growth rate, if any, and size of this
market.
Because
of our lack of an operating history and the emerging nature of the markets in
which we compete, we are is unable to accurately forecast our revenues or our
profitability. The market for our products and the long-term
acceptance of our products are uncertain, and our ability to attract and retain
qualified personnel with industry expertise, particularly sales and marketing
personnel, is uncertain. To the extent we are unsuccessful in
increasing revenues, we may be required to appropriately adjust spending to
compensate for any unexpected revenue shortfall, or to reduce our operating
expenses, causing us to forego potential revenue generating activities, either
of which could have a material adverse effect on our business, results of
operations and financial condition.
11
We
have incurred losses in prior periods and may incur losses in the
future.
We
incurred net losses of approximately $566,153 for our fiscal year ended
September 30, 2010. As of September 30, 2010, we had an accumulated
deficit of approximately $695,012. We have not achieved profitability
in any period, and we expect to continue to incur net losses for the foreseeable
future. Should we continue to incur net losses in future periods, we
may not be able to increase the number of employees or our investment in capital
equipment, sales and marketing programs and research and development in
accordance with present plans. Continuation of net losses may also
require us to secure additional financing sooner than expected. Such
financing may not be available in sufficient amounts, or on terms acceptable to
us and may dilute existing shareholders.
We
will require additional capital in the future in order to maintain and expand
our operations. Failure to obtain required capital would adversely
affect our business.
Until
such time as we become profitable, we will be required to obtain additional
financing or capital investments in order to maintain and expand our operations
and take advantage of future business opportunities. Obtaining
additional financing will be subject to, among other factors, market conditions,
industry trends, investor sentiment and investor acceptance of our business plan
and management. These factors may make the timing, amount, terms and
conditions of additional financing unattractive or unavailable to
us. There are no assurances that we will be able to raise cash from
equity or debt financing efforts or that, even if raised, such cash would be
sufficient to satisfy our anticipated capital requirements. Further,
there is no assurance concerning the terms on which such capital might be
available. Failure to obtain financing sufficient to meet our
anticipated capital requirements could have a material adverse effect on our
business, operating results and financial condition.
If
our products do not achieve greater market acceptance, or if alternative brands
are developed and gain market traction, our business would be adversely
affected.
Our
success is dependent upon the successful development and marketing of our
products. Our future success depends on increased market acceptance
of our air purifier product lines. The air purification community may
not embrace our product line. Acceptance of our products will depend
on several factors, including cost, product effectiveness, convenience,
strategic partnerships and reliability. We also cannot be sure that
our business model will gain wide acceptance among retailers or the air purifier
community. If the market fails to continue to develop, or develops
more slowly than we expect, our business, results of operations and financial
condition will be adversely affected. Moreover, if new air purifier
brands are developed, our prospective products and current technologies could
become less competitive or obsolete. Any of these factors could have
a material and adverse impact on our growth and profitability.
The
markets in which we operate are very competitive, and many of our competitors
and potential competitors are larger, more established and better capitalized
than we are.
Although
air purification technology is a rapidly emerging technology, the market for
these products is highly competitive and we expect that competition will
continue to intensify. Our products compete broadly with other
current companies offering air purification technology, including companies that
offer UV air purification technology, such as 3M Corporation and
Sears. These products compete directly with the products offered by
us.
Many
competitors have longer operating histories, larger customer bases, and greater
financial, research and development, technical, marketing and sales, and
personnel resources than we have. Given their capital resources, the
larger companies with whom we compete or may compete in the future, are in a
better position to substantially increase their manufacturing capacity, research
and development efforts or to withstand any significant reduction in orders by
customers in our markets. Such larger companies typically have
broader and more diverse product lines and market focus and thus are not as
susceptible to downturns in a particular market. In addition, some of
our competitors have been in operation much longer than we have been and
therefore may have more longstanding and established relationships with current
and potential customers.
Because
we are small and do not have much capital, we must limit our
activities. Our relative lack of capital and resources will adversely
affect our ability to compete with large entities that market air purifier
products. We compete against other air purifier manufacturers and
retailers, some of which sell their products globally, and some of these
providers have considerably greater resources and abilities than we
have. These competitors may have greater marketing and sales
capacity, established sales and distribution networks, significant goodwill and
global name recognition. Furthermore, it may become necessary for us
to reduce our prices in response to competition. A reduction in
prices of our products could adversely affect our revenues and
profitability.
12
In
addition, other entities not currently offering products similar to us may enter
the market. Any delays in the general market acceptance of our
products may harm our competitive position. Any such delay would
allow our competitors additional time to improve their service or product
offerings, and provide time for new competitors to develop. Increased
competition may result in pricing pressures, reduced operating margins and loss
of market share, which could have an adverse effect on our business, operating
results and financial condition.
Inability
of our officers and directors to manage the growth of the business may limit our
success.
We expect
to grow as we execute or business strategy. Rapid growth
would place a significant strain on our management and operational
resources. In addition, we expect the demands on our infrastructure
and technical support resources to grow along with our customer base, and if we
are successful in implementing our marketing strategy, it could experience
difficulties responding to demand for our products and technical support in a
timely manner and in accordance with market expectations. These
demands may require the addition of new management personnel or the development
of additional expertise by existing management personnel. There can
be no assurance that our networks, procedures or controls will be adequate to
support our operations or that management will be able to keep pace with such
growth. Failure to manage growth effectively could have a material
adverse effect on our business, operating results and financial
condition.
As
we expand, management will be faced with new challenges due to increases in
operating expenses and risks related to expansion.
As our
business grows and expands, we will spend substantial financial and other
resources on developing and introducing new products and expanding our sales and
marketing organization, strategic relationships and operating
infrastructure. If our business and revenues grow, we
expect that our cost of revenues, sales and marketing expenses, general and
administrative expenses, operations and customer support expenses will
increase.
If
we fail to integrate our recent acquisitions with our operations, our business
could suffer.
We
recently acquired air purification technology and products from AmAirpure, Inc.,
and in the future we may acquire more air purification technologies, businesses
or assets. The integration of acquired businesses, technologies or
assets requires significant effort and entails risks. We may find it
difficult to integrate operations of acquired businesses as personnel may leave
and licensees, distributors or suppliers may terminate their arrangements or
demand amended terms to these arrangements. Additionally, our
management may have their attention diverted while trying to integrate
businesses or assets that may be acquired. If we are not able to
successfully integrate any businesses or assets that we acquire, we may not
realize the anticipated benefits of these acquisitions.
Our
success depends on our ability to capitalize on our strategic relationships and
partnerships with suppliers, distributors, purchasers and users of our
products.
We will
rely on strategic relationships with third parties to expand our distribution
channels and to undertake joint product development and marketing
efforts. Our ability to increase sales depends on marketing our
products through new and existing strategic relationships. We intend
to partner with established existing suppliers and distributors in order to
reach target markets such as the medical, healthcare, hospitality, food service
and lodging markets. The termination of one or more of our strategic
relationships may have a material adverse effect on our business, operating
results and financial condition.
Our
intellectual property may not protect our products, and/or our products may
infringe on the intellectual property rights of third parties.
We regard
our trademarks, trade secrets and similar intellectual property as critical to
our success and attempt to protect such property with registered and common law
trademarks and copyrights, restrictions on disclosure and other actions to
forestall infringement. Despite precautions implemented by us,
unauthorized third parties may copy certain portions of our products or reverse
engineer or obtain and use information regarded by us as
proprietary. We have secured one patent in the United States, have
filed an application for an additional patent, and may seek additional patents
in the future. We do not know if a patent will issue on the patent
application or whether any future patent applications will be issued with the
scope of the claims sought by us, or whether any patents received by us will be
challenged or invalidated. In addition, many other organizations are
engaged in research and product development efforts that may overlap with our
products. Such organizations may currently have, or may obtain in the
future, legally blocking proprietary rights, including patent rights, in one or
more products or methods under development or consideration by
us. These rights may prevent us from commercializing products, or may
require us to obtain a license from the organizations to use the
technology. We may not be able to obtain any such licenses that may
be required on reasonable financial terms, if at all, and cannot be sure that
the patents underlying any such licenses will be valid or
enforceable. The laws of some foreign countries do not protect
proprietary rights to the same extent as the laws of the United
States. Our means of protecting our proprietary rights in the United
States or abroad may not be adequate and competitors may independently develop
similar technology and products. Third parties may infringe or
misappropriate our copyright, trademarks and similar proprietary
rights. In addition, other parties may assert infringement claims
against us. We cannot be certain that our products do not infringe
issued patents that may relate to our products. We may be subject to
legal proceedings and claims from time to time in the ordinary course of
business, including claims of alleged infringement of the trademarks and other
intellectual property rights of third parties. Intellectual property
litigation is expensive and time consuming and may divert management’s attention
away from running our business which may have a material adverse effect on our
business, operating results and financial condition.
13
The
value of our technology may be vulnerable to the discovery of unknown
technological defects.
Our
products depend on complex technology. Complex technology often
contains defects, particularly when first introduced or when new versions are
released. Although we conduct extensive testing, there is a
possibility that technology defects may not be detected until after the product
has been released. Although we have not experienced any material
technology defects to date, it is possible that despite testing, defects may
occur in the products. The defects may result in damage to our
reputation or increase costs, cause us to lose revenue or delay market
acceptance or divert our development resources, any of which may have a material
adverse effect on our business, operating results and financial
condition.
Government and
private insurance plans may not adequately reimburse patients for our products,
which could result in reductions in sales or selling prices for our
products.
Our
ability to sell our products will depend in some part on the extent to which
reimbursement for the cost of our products will be available from government
health administration authorities, private health insurers and other
organizations. In November 2008, the U.S. Food and Drug
Administration (“FDA”) cleared our ViraTech UV-400 product as a Class II medical
device, and we believe that certain purchasers of our product may generally
qualify for reimbursement of some of the costs of purchasing our product,
subject to the terms and conditions of their insurance plan or Medicare or
Medicaid. Third party payers such as insurance companies are
increasingly challenging the prices charged for medical products and can,
without notice, deny coverage for treatments that may include the use of our
products. Therefore, even if a product is cleared for marketing, we
cannot be assured that reimbursement will be allowed for the product, that the
reimbursement amount will be adequate or, that the reimbursement amount, even if
initially adequate, will not subsequently be reduced. Additionally,
future legislation or regulations concerning the healthcare industry or third
party or governmental coverage and reimbursement, particularly legislation or
regulation limiting consumers’ reimbursement rights, may harm our
business.
As we
develop new products, those products will generally not qualify for
reimbursement, if at all, until they are cleared for marketing and until they
are approved for reimbursement under policies of insurance, Medicare and
Medicaid. We do not file claims and bill governmental programs or
other third party payers directly for reimbursement for our
products. However, we are still subject to laws and regulations
relating to governmental reimbursement programs, particularly Medicaid and
Medicare.
Failure to comply
with anti-kickback and fraud regulations could result in substantial penalties
and changes in our business operations.
The
federal Anti-Kickback Law prohibits persons from knowingly and willfully
soliciting, receiving, offering or providing remuneration, directly or
indirectly, to induce either the referral of an individual, or the furnishing,
recommending or arranging for a good or service, for which payment may be made
under a federal healthcare program such as the Medicare and Medicaid
programs. The U.S. government has interpreted this law broadly to
apply to the marketing and sales activities of manufacturers and distributors
like us. Many states and other governments have adopted laws similar
to the federal Anti-Kickback Law. We are also subject to other
federal and state fraud laws applicable to payment from any third party
payer. These laws prohibit persons from knowingly and willfully
filing false claims or executing a scheme to defraud any healthcare benefit
program, including private third party payers. These laws may apply
to manufacturers and distributors who provide information on coverage, coding,
and reimbursement of their products to persons who do bill third party
payers. Any violation of these laws and regulations could result in
civil and criminal penalties (including fines), increased legal expenses and
exclusions from governmental reimbursement programs, all of which could have a
material adverse effect upon our business, financial conditions and results of
operations.
14
Complying with
Food and Drug Administration, or FDA, and other regulations is an expensive and
time-consuming process, and any failure to comply could have a materially
adverse effect
on our business,
financial condition, or results of operations.
Based on
the intended use of some of our products, our products can be subject
to significant federal government regulation. Those regulations could
restrict the sale and/or marketing of some of our
products. The manufacture, packaging, labeling, advertising,
promotion, distribution, and sale of our anti-microbial products are subject to
regulation by federal and state governmental agencies in the United States and
other countries, including the FDA and the U.S. Federal Trade Commission
(“FTC”). We note that failure to comply with FDA regulations can
result in adverse governmental enforcement action including civil and criminal
action, injunctions, recalls, seizures and fines. Any action of this
type by the FDA may materially adversely affect our ability to market our
products.
Likewise,
failure to comply with FTC rules and standards could result in significant
fines, injunctions, cease and desist orders, advertising limitations, and a
variety of other enforcement sanctions available to the FTC. FTC
would take action if it deemed advertising to be false or
misleading. In particular, representations made about our
products must be backed by ”competent and reliable scientific
evidence” sufficient to support the claims made for the product. FTC
would deem the failure of such an advertisement or labeling to be backed by that
kind of evidence false FTC and the dissemination of it to be deemed an
unfair or deceptive practice. Any enforcement action by the FTC could
materially adversely affect our ability to market our products.
We cannot
predict the nature of any future laws, regulations, interpretations, or
applications, nor can we determine what effect additional governmental
regulations or administrative orders, when and if promulgated, would have on our
business. They could include, however, requirements for the redesign
of our products, the recall or discontinuance of certain products, additional
record keeping and reporting, expanded documentation of the scientific support
or performance of certain products, and/or changes in labels and
advertising. Any of these requirements could have a material adverse
effect on the company.
Product sales, introductions or
modifications may be delayed or canceled as a result of FDA regulations or
similar foreign regulations, which could cause our sales and profits to
decline.
Before we
can market or sell a new medical device in the United States, we must obtain FDA
clearance, which can be a lengthy and time-consuming process and thus very
costly. We will have to receive clearance from the FDA to market our
products in the United States under Section 510(k) of the Federal Food,
Drug, and Cosmetic Act or our products must be found to be exempt from the
Section 510(k) clearance process.
Any new
product introduction or existing product modification could be subjected to a
lengthier, more rigorous FDA examination process. For example, in
certain cases we may need to conduct clinical trials of a new product before
submitting a 510(k) notice. Additionally, we may be required to
obtain premarket approvals for our products. The requirements of
these more rigorous processes could delay product introductions and increase the
costs associated with FDA compliance. Marketing and sale of our
products outside the United States are also subject to regulatory clearances and
approvals, and if we fail to obtain these regulatory approvals, our sales could
suffer.
We cannot
assure you that any new products we develop will receive required regulatory
approvals from U.S. or foreign regulatory agencies.
We are subject to
substantial regulation related to quality standards applicable to our
manufacturing and quality processes. Our failure to comply with these
standards could have an adverse effect on our business, financial condition, or
results of operations.
The FDA
regulates the approval, manufacturing, and sales and marketing of our products
in the U.S. Although we outsource the manufacture of our products and do not
currently manufacture any products currently, our manufacturers may be
required to register with the FDA and may be subject to periodic inspection by
the FDA for compliance with the FDA’s Quality System Regulation (“QSR”)
requirements, which require manufacturers of medical devices to adhere to
certain regulations, including testing, quality control and documentation
procedures. In addition, the federal Medical Device Reporting
regulations require our manufacturers to provide information to the FDA whenever
there is evidence that reasonably suggests that a device may have caused or
contributed to a death or serious injury or, if a malfunction were to occur,
could cause or contribute to a death or serious injury. Compliance
with applicable regulatory requirements is subject to continual review and is
rigorously monitored through periodic inspections by the FDA. Failure
to comply with current governmental regulations and quality assurance guidelines
could lead to temporary manufacturing shutdowns, product recalls or related
field actions, product shortages or delays in product
manufacturing. Efficacy or safety concerns, an increase in trends of
adverse events in the marketplace, and/or manufacturing quality issues with
respect to our products could lead to product recalls or related field actions,
withdrawals, and/or declining sales.
15
Our
profitability and success is subject to risks associated with potential general
economic downturn.
Recently,
the general health of the U.S. economy has been relatively weakened
substantially, a consequence of which has been declining spending by individuals
and companies. To the extent the general economic health of the U.S.
continues to decline, or to the extent individuals or companies fear such a
decline will continue, such individuals and companies may continue to reduce
expenditures such as those for the products offered by us because such products
may be considered dispensable items in a recession. A continued
decline could delay decisions among certain of our customers to purchase our
products or could delay decisions by prospective customers to make initial
evaluations of our products. Such delays may have a material adverse
effect on our business, operating results and financial condition.
A
limited public trading market exists for our common stock, which makes it more
difficult for our stockholders to sell their common stock in the public
markets.
Although
our common stock is quoted on the OTC Bulletin Board, or OTCBB, under the symbol
“UVFT,” there is currently no active public trading market for our common
stock. No assurance can be given that an active market will develop
or that a stockholder will ever be able to liquidate our shares of common stock
without considerable delay, if at all. Many brokerage firms may not
be willing to effect transactions in our securities. Furthermore, our
future stock price may be impacted by factors that are unrelated or
disproportionate to our operating performance. These market
fluctuations, as well as general economic, political and market conditions, such
as recessions, lack of available credit, interest rates or international
currency fluctuations may adversely affect the future market price and
liquidity of our common stock.
Our common stock may be subject to
the penny stock rules which may make it more difficult to sell our common
stock.
Because
our common stock is not listed on any national securities exchange, trading in
our common stock is also subject to the regulations regarding trading in “penny
stocks,” which are those securities trading for less than $5.00 per
share. The following is a list of the general restrictions on the
sale of penny stocks:
|
·
|
Before
the sale of penny stock by a broker-dealer to a new purchaser, the
broker-dealer must determine whether the purchaser is suitable to invest
in penny stocks. To make that determination, a broker-dealer
must obtain, from a prospective investor, information regarding the
purchaser’s financial condition and investment experience and
objectives. Subsequently, the broker-dealer must deliver to the
purchaser a written statement setting forth the basis of the suitability
finding and obtain the purchaser’s signature on such
statement.
|
|
·
|
A
broker-dealer must obtain from the purchaser an agreement to purchase the
securities. This agreement must be obtained for every purchase
until the purchaser becomes an “established customer.” A broker-dealer may
not affect a purchase of a penny stock less than two business days after a
broker-dealer sends such agreement to the
purchaser.
|
|
·
|
The
Securities Exchange Act of 1934, or the Exchange Act, requires that before
effecting any transaction in any penny stock, a broker-dealer must provide
the purchaser with a “risk disclosure document” that contains, among other
things, a description of the penny stock market and how it functions and
the risks associated with such investment. These disclosure
rules are applicable to both purchases and sales by
investors.
|
|
·
|
A
dealer that sells penny stock must send to the purchaser, within ten days
after the end of each calendar month, a written account statement
including prescribed information relating to the
security.
|
These
requirements can severely limit the liquidity of securities in the secondary
market because few brokers or dealers are likely to be willing to undertake
these compliance activities. As a result of our common stock not
being listed on a national securities exchange and the rules and restrictions
regarding penny stock transactions, an investor’s ability to sell to a third
party and our ability to raise additional capital may be limited. We
make no guarantee that our market-makers will continue to make a market in our
common stock, or that any market for our common stock will
continue.
We
cannot guarantee that investors will be paid any dividends.
We have
never declared or paid dividends on our common stock. We intend to
retain earnings, if any, to support the development of our business and
therefore do not anticipate paying cash dividends for the foreseeable
future. Payment of future dividends, if any, will be at the
discretion of our board of directors after taking into account various factors,
including current financial condition, operating results and current and
anticipated cash needs.
16
Nevada
law and our articles of incorporation authorize us to issue shares of stock,
which shares may cause substantial dilution to our existing shareholders and/or
have rights and preferences greater than our common stock.
Pursuant
to our Articles of Incorporation, we have, as of the date of this Report,
75,000,000 shares of common stock authorized. As of the date of this
Report, we have 45,080,000 shares of common stock issued and
outstanding. As a result, our Board of Directors has the ability to
issue a large number of additional shares of common stock without shareholder
approval, which if issued could cause substantial dilution to our then
shareholders.
We are subject to new corporate
governance and internal control reporting requirements, and our costs related to
compliance with, or our failure to comply with existing and future requirements,
could adversely affect our business .
We face
corporate governance requirements under the Sarbanes-Oxley Act of 2002, as well
as new rules and regulations subsequently adopted by the SEC and the Public
Company Accounting Oversight Board. These laws, rules and regulations
continue to evolve and may become increasingly stringent in the
future. We are required to include management’s report on internal
controls as part of our annual report pursuant to Section 404 of the
Sarbanes-Oxley Act. We strive to continuously evaluate and improve
our control structure to help ensure that we comply with Section 404 of the
Sarbanes-Oxley Act. The financial cost of compliance with these laws,
rules and regulations is expected to remain substantial. We cannot
assure you that we will be able to fully comply with these laws, rules and
regulations that address corporate governance, internal control reporting and
similar matters. Failure to comply with these laws, rules and
regulations could materially adversely affect our reputation, financial
condition and the value of our securities.
If
we are unable to successfully recruit qualified and experienced employees and
personnel, we may not be able to execute our business plan.
Our
ability to increase revenues will depend in large part on our ability to
successfully recruit, train and retain sales marketing
personnel. There can be no assurance that we will be able to find,
attract and retain existing employees or that we will be able to find, attract
and retain qualified personnel on acceptable terms. Competition for
additional qualified personnel is intense and we may not be able to hire or
retain personnel with relevant experience. Any delays or difficulties
encountered by us in hiring or retaining qualified personnel may adversely
affect our business, operating results and financial condition.
We
are dependent on our key employees.
Our
success depends to a significant extent upon the continued service of our senior
management and key executives, including John J. Lennon, President, CEO and
Chief Financial Officer. Our success depends on the skills,
experience and performance of senior management and other key personnel, many of
whom have also worked together for only a short period of time. We do
not have long-term employment agreements with any member of senior management or
other key personnel. Our success also depends on our ability to
recruit, train or retain qualified personnel. The loss of the
services of any of the key members of senior management, other key personnel, or
our inability to recruit, train or retain senior management or key personnel may
have a material adverse effect on our business, operating results and financial
condition.
ITEM
1B. UNRESOLVED
STAFF COMMENTS.
None.
ITEM
2. PROPERTIES.
We
currently maintain our registered offices at 1694 Falmouth Road #125,
Centerville, MA 02632-2933.
ITEM
3. LEGAL
PROCEEDINGS
None.
ITEM
4. RESERVED.
Not
applicable.
17
PART
II
|
ITEM
5.
|
MARKET
FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER
PURCHASES OF EQUITY SECURITIES.
|
Market
Information
Since
inception, there has been a limited trading market for our common
stock. Our common stock is listed on the OTC Bulletin Board
(“OTCBB”) exchange under the symbol UVFT.OB. Our common stock has
been listed on the OTCBB since November 12, 2009. Prior to that time,
there was no public market for our common stock. The table below
lists the high and low closing prices per share of our common stock since our
stock was first traded , as quoted on OTCBB.
|
Fiscal
2010
|
High
|
Low
|
||||||
|
First
Quarter
|
$ | .37 | $ | .23 | ||||
|
Second
Quarter
|
$ | .39 | $ | .23 | ||||
|
Third
Quarter
|
$ | .51 | $ | .34 | ||||
|
Fourth
Quarter
|
$ | .41 | $ | .24 | ||||
Trading
in our common stock has been sporadic and the quotations set forth above are not
necessarily indicative of actual market conditions. All prices
reflect inter-dealer prices without retail mark-up, mark-down, or commission and
may not necessarily reflect actual transactions.
Stockholders
As of
January 10, 2010, we had 45,080,000 shares of common stock outstanding held
by 94 shareholders.
Dividends
We have
not paid dividends to date and do not anticipate paying any dividends in the
foreseeable future. Our Board of Directors intends to follow a policy
of retaining earnings, if any, to finance our growth. The declaration
and payment of dividends in the future will be determined by our Board of
Directors in light of conditions then existing, including our earnings,
financial condition, capital requirements and other factors.
Securities
Authorized For Issuance under Equity Compensation Plans
There are
no securities authorized for issuance under any equity compensation plans during
the fiscal year ended September 30, 2010.
Purchases
of Equity Securities by the Issuer and Affiliated Purchases
There were no issuer purchases of our
equity securities during the fiscal year ended September 30, 2010.
ITEM
6. SELECTED
FINANCIAL DATA
As a
smaller reporting company, as defined in Rule 12b-2 of the Exchange Act, we are
not required to provide the information required by this item.
|
ITEM
7.
|
MANAGEMENT’S
DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF
OPERATIONS
|
The
following discussion should be read in conjunction with our consolidated
financial statements and notes thereto included elsewhere in this
Report. Forward looking statements are statements not based on
historical information and which relate to future operations, strategies,
financial results or other developments. Forward-looking statements
are based upon estimates, forecasts, and assumptions that are inherently subject
to significant business, economic, and competitive uncertainties and
contingencies, many of which are beyond our control and many of which, with
respect to future business decisions, are subject to change. These
uncertainties and contingencies can affect actual results and could cause actual
results to differ materially from those expressed in any forward-looking
statements made by us, or on our behalf. We disclaim any obligation
to update forward-looking statements.
18
The
discussion and financial statements contained herein are for our fiscal year
ended September 30, 2010 and September 30, 2009. The following
discussion regarding our financial statements should be read in conjunction with
our financial statements included herewith.
Financial
Condition as of September 30, 2010
We
reported total current assets of $103,312 at September 30, 2010, consisting of
accounts receivable and prepaids. Total current liabilities reported
of $691,639 consisted of $149,775 in accounts payable and accrued liabilities
and $541,864 in notes payable. We had a working capital deficiency of
$588,327 at September 30, 2010.
Net Loss
increased from $20,828 for the year ended September 30, 2009 to $566,153 at
September 30, 2010.
We are
currently a development stage company focused in the air purification industry,
and evaluating opportunities for expansion within that industry through
acquisition or other strategic relationships.
Plan
of Operation
Background
We were
organized under the laws of the State of Nevada on April 4, 2006 under the name
“Northwest Chariots, Inc.” and were engaged in the business of renting and
selling electrically powered human transporters, like electric bicycles,
chariots and quads. Subsequent to our fiscal year ended September 30,
2009, we decided to change our product mix to air purification products and to
focus on the research, development, manufacturing and sales of air purification
systems and products.
In
furtherance of our business objectives, on November 12, 2009, we effected a
32-for-1 forward stock split of all our issued and outstanding shares of common
stock, and we merged with our wholly-owned subsidiary, UV Flu Technologies,
Inc., for the purposes of effecting a name change to “UV Flu Technologies,
Inc.”
Effective
November 15, 2009, we acquired AmAirpure Inc.’s air purification technology,
product, inventory, and certain equipment pursuant to an Asset Purchase
Agreement with AmAirpure, Inc. We issued 15,000,000 shares of our
common stock to shareholders of AmAirpure in connection with the asset
acquisition. Additionally, on November 25, 2009, we entered into a
Distribution Agreement with Puravair Distributors LLC (“Puravair”) where we
appointed Puravair as our exclusive master distributor for our Viratech UV-400
product and our other products for the professional, medical, and commercial
markets in the U.S. and Canada. On September 30, 2010, we terminated
our Distribution Agreement with Puravair and began adding new distributors,
which totaled five as of year end.
The
latest production runs of our Viratech UV-400 product incorporate our patented
UV bacteria killing technology, which has been cleared by the FDA for use as a
medical device. In June 2010, we expanded our market reach by
introducing the latest generation of our Viratech UV-400 product into the
residential and hospitality markets.
On
October 28, 2010, we entered into a binding letter of intent with The Red Oak
Trust (“Red Oak”) (the “LOI”) in connection with our proposed acquisition of one
hundred percent (100%) of the issued and outstanding units of RxAir Industries,
LLC, a Nevada limited liability company (“RxAir”), which is wholly owned by Red
Oak (the “Acquisition”). At the closing of the Acquisition, RxAir
will become a wholly-owned subsidiary of the Company. We are
currently renegotiating the terms of the LOI and we intend to enter into a
definitive agreement soon.
We
currently have minimal revenue from operations. In order to meet our
business objectives, we will need to raise additional funds through equity or
convertible debt financing. There can be no assurance that we will be
successful in raising additional funds and, if unsuccessful, our plans for
expanding operations and business activities may have to be
curtailed. Any attempt to raise funds, through debt or equity
financing, would likely result in dilution to existing
shareholders.
Cash
and Cash Equivalents
As of
September 30, 2010, we had cash of $nil. As such, we anticipate that
we will have to raise additional capital through debt or equity financings to
fund our operations and execute our business plan during the next 6 to 12
months.
19
Critical
Accounting Policies
The
preparation of financial statements in conformity with United States generally
accepted accounting principles requires management of our company to make
estimates and assumptions that affect the reported amounts of assets and
liabilities, the disclosure of contingent assets and liabilities at the date of
the financial statements and the reported amounts of revenues and expenses
during the reporting periods.
Our
management routinely makes judgments and estimates about the effects of matters
that are inherently uncertain. As the number of variables and
assumptions affecting the probable future resolution of the uncertainties
increase, these judgments become even more subjective and
complex. Our significant accounting policies are discussed in Note 3
to our financial statements for the fiscal year ended September 30, 2010
included in the Form 10-K. We have identified the following
accounting policies, described below, as the most important to an understanding
of our current financial condition and results of operations.
This
summary of significant accounting policies is presented to assist in
understanding our financial statements. Our financial statements and
notes are representations of our management who is responsible for their
integrity and objectivity. These accounting policies conform to
generally accepted accounting principles in the United States of America
(“GAAP”) and have been consistently applied in the preparation of the financial
statements. The financial statements are stated in United States of
America dollars.
(a) Organizational and Start-up
Costs. Costs of start-up activities, including organizational
costs, are expensed as incurred in accordance with Accounting Standards
Codification (“ASC”) subtopic 720-15 (formerly Statements of Position (“SOP”)
98-5).
(b) Income Taxes. We
have adopted the ASC subtopic 740-10
(formerly Statement of Financial Accounting Standards (“SFAS”) No. 109 –
“Accounting for Income Taxes”). ASC 740-10 requires the use of the
asset and liability method of accounting of income taxes. Under the
asset and liability method of ASC 740-10, deferred tax assets and liabilities
are recognized for the future tax consequences attributable to temporary
differences between the financial statements carrying amounts of existing assets
and liabilities and their respective tax bases. Deferred tax assets
and liabilities are measured using enacted tax rates expected to apply to
taxable income in the years in which those temporary differences are expected to
be recovered or settled.
We
provide deferred taxes for the estimated future tax effects attributable to
temporary differences and carry forwards when realization is more likely than
not.
(c) Inventories. Inventories
are stated at the lower of cost or market, with cost being determined using the
first-in first-out method. Inventories consist of purchased goods
held for resale.
(d) Property and
Equipment. Property and equipment is stated at cost, and is
depreciated over estimated useful lives using primarily the straight line method
for financial reporting purposes. Useful lives range from 3 to 5
years. We evaluate equipment at least annually for
impairment.
(e) Advertising. Advertising
costs are expensed as in accordance with ASC subtopic 720-35 (formerly SOP
93-7).
(f) Basic and Diluted Loss Per
Share. In accordance with ASC subtopic 260-10 (formerly SFAS
No. 128 – “Earnings Per Share”), the basic loss per common share is computed by
dividing net loss available to common stockholders by the weighted average
number of common shares outstanding. Diluted loss per common share is
computed similar to basic loss per common share except that the denominator is
increased to include the number of additional common shares that would have been
outstanding if the potential common shares had been issued and if the additional
common shares were dilutive. At September 30, 2009 and 2008, we had
no stock equivalents that were anti-dilutive and excluded in the earnings per
share computation.
(g) Estimated Fair Value of Financial
Instruments. The carrying value of our financial instruments,
consisting of cash, and accounts payable approximate their fair value due to the
short-term maturity of such instruments. Unless otherwise noted, it
is management’s opinion that we are not exposed to significant interest,
currency or credit risks arising from these financial statements.
(h) Revenue
Recognition. It is our policy that revenues will be recognized
in accordance with ASC subtopic 605-10 (formerly SEC Staff Accounting Bulletin
(SAB) No. 104, “Revenue Recognition.”). Under ASC 605-10, product
revenues (or service revenues) are recognized when persuasive evidence of an
arrangement exists, delivery has occurred (or service has been performed), the
sales price is fixed and determinable and collectability is reasonably
assured. Revenues are recognized only when the Company has
transferred to the customer the significant risk and rewards of ownership of the
goods, title to the products transfers, the amount is fixed and determinable,
evidence of an agreement exists, there is reasonable assurance of collection of
the sales proceeds, the Company has no future obligations, and the customer
bears the risk of loss. This occurs at the time of shipment (FOB
shipping point) of the products from the Company’s warehouse and an invoice is
prepared.. There are no rights of return and exchange is allowed only
on defective products. Recognition of revenue is not affected as the
right of exchange results in new units being shipped to the customer once
defective units have been received by the company and verified as
defective.
20
(i) Currency. Our
functional currency is the United States Dollar. Realized gain or
loss on foreign currency transactions are reflected in the income
statement.
(j) Use of
Estimates. The preparation of financial statements in
conformity with GAAP requires management to make estimates and assumptions that
affect the reported amounts of assets and liabilities and disclosure of
contingent assets and liabilities at the date of the financial statements and
the reported amount of revenues and expenses during the reporting
period. Actual results could differ from those
estimates.
(k) Cash and Cash
Equivalents. Cash is comprised of cash on hand and demand
deposits. Cash equivalents include short-term highly liquid
investments with original maturities of three months or less that are readily
convertible to known amounts of cash and which are subject to an insignificant
risk of change in value.
(l) Concentrations. Financial
instruments that potentially subject us to concentrations of credit risk consist
principally of cash and cash equivalents. At September 30, 2009, we
had $2,522 U.S. funds in deposit in a business bank account and U.S. equivalent
of $297 in Canadian funds in a business account which are not insured by
agencies of the U.S. Government.
(m) Recent Accounting
Pronouncements. In August 2010, the FASB issued Accounting
Standard Updates No. 2010-21 (ASU No. 2010-21) “Accounting for Technical
Amendments to Various SEC Rules and Schedules” and No. 2010-22 (ASU No. 2010-22)
“Accounting for Various Topics – Technical Corrections to SEC
Paragraphs”. ASU No 2010-21 amends various SEC paragraphs pursuant to
the issuance of Release no. 33-9026: Technical Amendments to Rules, Forms,
Schedules and Codification of Financial Reporting Policies. ASU No.
2010-22 amends various SEC paragraphs based on external comments received and
the issuance of SAB 112, which amends or rescinds portions of certain SAB
topics. Both ASU No. 2010-21 and ASU No. 2010-22 are effective upon
issuance. The amendments in ASU No. 2010-21 and No. 2010-22 will not
have a material impact on the Company’s financial statements.
The FASB
issued ASC subtopic 855-10 (formerly SFAS 165 “Subsequent
Events”), incorporating guidance on subsequent events into
authoritative accounting literature and clarifying the time following the
balance sheet date which management reviewed for events and transactions that
may require disclosure in the financial statements. The Company
adopted this standard effective the second quarter of 2009 The
standard increased our disclosure by requiring disclosure of the date through
which subsequent events have been reviewed. The standard did not
change our procedures for reviewing subsequent events. ASC 855-10 is
included in the “Subsequent Events” accounting guidance.
In April
2009, the FASB issued ASC subtopic 820-10 (formerly Staff Position
No. FAS 157-4,
Determining Fair Value When Volume and Level of Activity for the Asset or
Liability Have Significantly Decreased and Identifying Transactions That Are Not
Orderly”) . ASC 820-10 provides guidance on how to determine
the fair value of assets and liabilities when the volume and level of activity
for the asset/liability has significantly decreased. FSP 157-4
also provides guidance on identifying circumstances that indicate a transaction
is not orderly. In addition, FSP 157-4 requires disclosure in
interim and annual periods of the inputs and valuation techniques used to
measure fair value and a discussion of changes in valuation
techniques. The Company is evaluating the effect of the adoption of
FSP 157-4 and determined that it did not have a material impact on its
results of operations and financial position.
In
July 2006, the FASB issued ASC subtopic 740-10 (formerly Interpretation No.
(“FIN”) 48, “ Accounting
for Uncertainty in Income Taxes “). ASC 740-10 sets forth a
recognition threshold and valuation method to recognize and measure an income
tax position taken, or expected to be taken, in a tax return. The
evaluation is based on a two-step approach. The first step requires
an entity to evaluate whether the tax position would “more likely than not,”
based upon its technical merits, be sustained upon examination by the
appropriate taxing authority. The second step requires the tax
position to be measured at the largest amount of tax benefit that is greater
than 50 percent likely of being realized upon ultimate settlement. In
addition, previously recognized benefits from tax positions that no longer meet
the new criteria would no longer be recognized. The application of
this Interpretation will be considered a change in accounting principle with the
cumulative effect of the change recorded to the opening balance of retained
earnings in the period of adoption. Adoption of this new standard is
not expected to have a material impact on our financial position, results of
operations or cash flows.
21
In April
2008, the FASB issued ASC 815-40 (formerly Emerging Issues Task Force (“EITF”)
07-05, “Determining whether an
Instrument (or Embedded Feature) Is Indexed to an Entity’s Own Stock
“). ASC815-40 applies to any freestanding financial instruments or
embedded features that have the characteristics of a derivative, and to any
freestanding financial instruments that are potentially settled in an entity’s
own common stock. ASC 815-40 is effective for financial statements
issued for fiscal years beginning after December 15, 2008. The
Company is currently evaluating the impact ASC 815-40, but does not expect the
adoption of this pronouncement will have a material impact on its financial
position, results of operations or cash flows.
International
Financial Reporting Standards
In
November 2008, the SEC issued for comment a proposed roadmap regarding potential
use of financial statements prepared in accordance with International Financial
Reporting Standards (“IFRS”) as issued by the International Accounting Standards
Board. Under the proposed roadmap, the Company would be required to prepare
financial statements in accordance with IFRS in fiscal year 2014, including
comparative information also prepared under IFRS for fiscal 2013 and
2012. The Company is currently assessing the potential impact of IFRS
on its financial statements and will continue to follow the proposed roadmap for
future developments.
Results
of Operations
The
following is Management’s discussion and analysis of certain significant factors
which have affected our financial condition and results of operations
during the periods included in the accompanying financial
statements.
Results
of Operations for the Year Ended September 30, 2010 as Compared to the Year
Ended September 30, 2009
Sales
and Rental Revenue
During
the fiscal year ended September 30, 2010, we received gross revenue of
$107,703 as compared to $18,000 for the year ended September 30,
2009. The increase is primarily related to the increase in sales and
leasing of units from our new product line, reflecting higher market penetration
of our product in the commercial market and in particular in the medical and
hospitality industries.
Net
Income (Loss)
During
the fiscal year ended September 30, 2010, our net loss was $566,153 as compared
to $20,828 for the year ended September 30, 2009. The increase in net
loss is due to an increase in our general and administrative expenses of
$566,728 for the year ended September 30, 2010 as compared to $27,730 for the
year ended September 30, 2009 as a result of increased business activity
associated with development of our new product line, as well as an increase in
interest expense of $22,659 as compared to $nil (zero) for the year ended
September 30, 2009.
General
and Administrative Expenses
During
the fiscal year ended September 30, 2010, we incurred total expenses of $566,728
as compared to $27,730 for the year ended September 30, 2009. The
increase is primarily related to expenses for marketing, office and
administration, professional fees, consulting and investor relation costs
increased due to an increase business activity associated with development of
the new product line. The increase in these particular expenses
are as follows:
|
September
30, 2010
|
September
30, 2009
|
|||||||
|
Marketing
|
$ | 89,541 | $ | - | ||||
|
Office
and administration
|
58,756 | 5,860 | ||||||
|
Professional
fees
|
130,965 | 17,948 | ||||||
|
Consulting
|
152,924 | - | ||||||
|
Investor
relations
|
67,042 | - | ||||||
|
Total
|
$ | 499,228 | $ | 23,808 | ||||
Bad debts
increased from $nil (zero) for the year ended September 30, 2009 to $67,500 for
the year ended September 30, 2010 and was due to the write off of the receivable
from Puravair Distributors LLC that was deemed uncollectible under our
Distribution Agreement with Puravair, such Distribution Agreement was terminated
on September 30, 2010.
Depreciation
and amortization expense decreased from $3,922 for the year ended September 30,
2009 to $nil for the year ended September 30, 2010 as the office equipment was
sold during the year resulting in a net gain of $1,116.
Expenses
or other cash flows in this period may not be indicative of future periods as we
are in the early development stage.
22
Liquidity
and Capital Resources
As of
September 30, 2010, we had cash of $nil (zero), and working capital deficiency
of $588,327. During the period ended September 30, 2010, we funded
our operations from receipts of sales revenues and proceeds from loans
payable. We have recognized a total of $143,603 in revenues since our
inception and incurred $541,864 in loans payable. In order to
survive, we are dependant on increasing our sales
volume. Additionally, we plan to continue further financings and
believe that this will provide sufficient working capital to fund our operations
for at least the next 12 months. Changes in our operating plans,
increased expenses, additional acquisitions, or other events may cause us to
seek additional equity or debt financing in the future.
For the
period ended September 30, 2010, we used $548,083 in cash flows to fund
operating activities as compared to $1,950 for the period ended September 30,
2009. The increase is primarily due to an increase in our general and
administrative expenses for the year ended September 30, 2010 as compared to the
year ended September 30, 2009 as a result of increased business activity
associated with development of our new product line For the period
ended September 30, 2010, net cash provided by operating activities reflected
$1,116 gain on sale of assets, $125,627 in changes in current assets and
inventory, and we used $144,813 in accounts payable and accrued
liabilities.
For the
period ended September 30, 2010, cash flows from investing activities was $3,400
due to proceeds from the sale of equipment as compared to $nil (zero) for the
period ended September 30, 2009.
For the
period ended September 30, 2010, cash flows from financing activities was
$541,864 from proceeds of loans payable as compared to $nil (zero) for the
period ended September 30, 2009.
We
anticipate that our cash requirements will be significant in the near term due
to contemplated development, purchasing, marketing and sales of our air
purification technologies and products. Accordingly, we expect to
continue to use loans payable to fund operations for at least the remaining of
our fiscal year ended September 30, 2011, as we look to generating sufficient
revenue to meet our needs.
Off-Balance
Sheet Arrangements
We
presently do not have any off-balance sheet arrangements.
Capital
Expenditures
We did
not make any capital expenditures in the fiscal year ended September 30,
2010.
Contractual
Obligations
The
following table outlines payments due under our significant contractual
obligations over the periods shown, exclusive of interest:
|
Payments Due by
Period
|
||||||||||||||||
|
Contract Obligations
At September 30, 2010
|
Total
|
Less than
1 Year
|
1-3 years
|
3-5 years
|
More than
5 years
|
|||||||||||
|
Total Debt
|
$
|
541,864
|
$ |
541,864
|
$
|
-
|
$
|
-
|
$
|
-
|
||||||
The above
table outlines our obligations as of September 30, 2010 and does not reflect any
changes in our obligations that have occurred after that
date.
ITEM
7A. QUANTITATIVE
AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
As a
smaller reporting company, as defined in Rule 12b-2 of the Exchange Act, we are
not required to provide the information required by this item.
ITEM
8. FINANCIAL
STATEMENTS AND SUPPLEMENTARY DATA
Reference
is made to the financial statements, the report of our independent registered
public accounting firm, and the notes thereto commencing at page F-1 of this
report, which financial statements, report, and notes are incorporated herein by
reference.
23
|
ITEM
9.
|
CHANGES
IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL
DISCLOSURE.
|
On
December 21, 2009, Bateman McDuffie, Inc. (formerly known as Bateman & Co.,
Inc., P.C.) (“Bateman”) was dismissed as our independent registered public
accounting firm. Our Board of Directors approved the decision to
change our independent registered public accounting firm.
During
the last two fiscal years ended September 30, 2010 and 2009, and further through
the date of dismissal of Bateman, there have been no disagreements with Bateman
on any matter of accounting principles or practices, financial statement
disclosure, or auditing scope or procedure, which disagreement if not resolved
to the satisfaction of Bateman, would have caused them to make reference to the
subject matter of the disagreement(s) in connection with their report on our
financial statements for such years; and there were no reportable events, as
listed in Item 304(a)(1)(iv) of Regulation S-K.
On
December 21, 2009, we engaged Weaver & Martin, LLC (“Weaver”) as our new
independent registered public accounting firm to audit our financial
statements. During the two most recent fiscal years and the interim
periods preceding the engagement, we did not consult with Weaver regarding (i)
the application of accounting principles to a specific transaction, either
completed or proposed, or the type of audit opinion that might be rendered on
our financial statements, and no written report or oral advice was provided to
us by Weaver concluding there was an important factor to be considered by us in
reaching a decision as to an accounting, auditing or financial reporting issue;
or (ii) any matter that was either the subject of a disagreement, as that term
is defined in Item 304 (a)(1)(iv) of Regulation S-K or a reportable event, as
that term is described in Item 304 (a)(1)(v) of Regulation S-K.
ITEM
9A. CONTROLS AND
PROCEDURES
We
carried out an evaluation, under the supervision and with the participation of
our management, including our Principal Executive Officer along with our
Principal Financial Officer, of the effectiveness of the design of the our
disclosure controls and procedures (as defined by Exchange Act
Rule 13a-15(e) and 15d-15(e)) as of the end of our fiscal year pursuant to
Exchange Act Rule 13a-15. Based upon that evaluation, our
Principal Executive Officer along with our Principal Financial Officer concluded
that our disclosure controls and procedures are effective at September 30, 2010
in ensuring that information required to be disclosed by us in reports that we
file or submit under the Exchange Act is recorded, processed, summarized, and
reported within the time periods specified in the Securities and Exchange
Commission’s rules and forms and that such information is accumulated and
communicated to our management, including our Principal Executive Officer and
Principal Financial Officer, as appropriate, to allow timely decisions regarding
required disclosure.
Management’s
Report on Internal Control over Financial Reporting
Our
management is responsible for establishing and maintaining adequate internal
control over financial reporting as defined in Rules 13a-15(f) and
15d-15(f) under the Exchange Act. Under the supervision and with the
participation of our management, including our Chief Executive Officer and Chief
Financial Officer, we conducted an evaluation of the effectiveness of our
internal control over financial reporting based on criteria established in the
framework in Internal
Control – Integrated Framework issued by the Committee of Sponsoring
Organizations of the Treadway Commission. Based on this evaluation,
our management concluded that our internal control over financial reporting was
effective as of September 30, 2010.
Because
of its inherent limitations, internal control over financial reporting may not
prevent or detect misstatements. Projections of any evaluation of effectiveness
to future periods are subject to the risk that controls may become inadequate
because of changes in conditions, or that the degree of compliance with the
policies or procedures may deteriorate. All internal control systems,
no matter how well designed, have inherent limitations. Therefore,
even those systems determined to be effective can provide only reasonable
assurance with respect to financial statement preparation and
presentation.
Changes
in Internal Control over Financial Reporting
We have
had very limited operations and there were no changes in our internal controls
over financial reporting that occurred during the three months ended September
30, 2010 that have materially affected, or are reasonably likely to materially
affect, our internal controls over financial reporting.
24
ITEM
9B. OTHER
INFORMATION.
There
were no items requiring reporting on Form 8-K that were not reported on Form 8-K
during the fourth quarter of the year covered by this Form 10-K.
PART
III
ITEM
10. DIRECTORS,
EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE.
Directors
and Executive Officers
The
following table sets forth the names and ages of our current directors and
executive officers, the principal offices and positions held by each
person:
|
Person
|
Age
|
Position
|
||
|
John
J. Lennon
|
55
|
President,
Treasurer, Chief Executive Officer, Chief Financial Officer,
and Director
|
||
|
Roger
Nelson
|
64
|
Secretary
and Director
|
||
|
Dr.
Dennis J. Rosen, M.D., FAAP
|
64
|
Director
|
||
|
Thomas
J. Mahowald
|
55
|
Director
|
Our Board
of Directors believes that its members encompass a range of talent, skill, and
experience sufficient to provide sound and prudent guidance with respect to our
operations and interests. The information below with respect to our
directors includes each director’s experience, qualifications, attributes, and
skills that led our Board of Directions to the conclusion that he should serve
as a director.
John J. Lennon, President, Chief Executive Officer,
Chief Financial Officer, and Director. On
November 12, 2009, our Board of Directors appointed Mr. John J. Lennon as
President, Secretary, Chief Financial Officer and member of the Board of
Directors. From September 21, 2009 to March 18, 2010, Mr. Lennon has
served as President, Chief Executive Officer, Chief Financial Officer and
Secretary of Celestial Delights USA Corp, and as Chief Financial Officer till
June 24, 2010; from May 30, 2008 to July 7, 2009, as Treasurer, VP of Finance
and Director of Brite-Strike Tactical Illumination Products, Inc.; from February
13, 2008 to June 2, 2009, as President, Chief Financial Officer and Secretary of
American Petro-Hunter, Inc., from June 2009, as Chief Financial Officer and
Secretary of American Petro-Hunter, Inc.; from May 2009 to October 30, 2009, as
President, Chief Financial Officer and Secretary of GC China Turbine Corp.; from
March 26, 2009, as President, Chief Financial Officer and Secretary of LED Power
Group, Inc.; and from 2004, as President of Chamberlain Capital
Partners. Chamberlain Capital Partners assists companies in the area
of maximizing shareholder value through increased sales, cost reduction and
refined business strategy. Mr. Lennon has also assisted companies in
obtaining debt financing, private placements or other methods of
funding. He is responsible for corporate reporting, press releases,
and funding related initiatives for American Durahomes, a private corporation,
where he acts as a Director, and from 2005 to December 31, 2008, acted as
Treasurer/Director/VP of Finance for US Starcom, a public entity. On
December 31, 2007, Mr. Lennon was appointed Chief Executive Officer, President,
Chief Financial Officer, Secretary, Treasurer and director of Explortex Energy
Inc., a publicly reporting company, which is a natural resource exploration
company engaged in the participation in drilling of oil and gas in the United
States. Mr. Lennon resigned from his positions at Explortex Energy Inc. on
June 29, 2009. From 1987 to 2004, Mr. Lennon served as Senior Vice
President of Janney Montgomery Scott, Osterville, MA, Smith Barney and
Prudential Bache Securities, managing financial assets for high net worth
individuals. Mr.
Lennon currently serves on the Board of Directors of Celestial Delights USA
Corp., AmAirPure, Inc., American Petro-Hunter, Inc. and LED Power Group, Inc.
Roger Nelson, Secretary and
Director. On January 6, 2010, our Board of Directors appointed
Mr. Nelson as Secretary and a member of the Board of Directors. Mr.
Nelson brings over 36 years of experience in the medical equipment and health
care sector. Since 1996, he was Senior Vice President at Eco-Rx,
Inc., a company specializing in the air purification
industry. Prior to that, he was the principle of Nelson &
Associates, an independent health care sales and consulting
organization. From 1980 to 1991, he held senior positions with
medical equipment manufacturers Retec, USA and Everest &
Jennings. His career in the sector began in 1973 with a lengthy
period at several divisions of Johnson & Johnson. Mr. Nelson was
educated at the University of Connecticut, with a B.S. in Business
Administration received in 1969.
25
Dr. Dennis J. Rosen, M.D.,
FAAP, Director. On
January 6, 2010, our Board of Directors appointed Dr. Rosen as a member of the
Board of Directors. Dr. Rosen has practiced medicine as a
Developmental Pediatrician since 1975. He is currently teaching at
UMASS Medical Center in Worcester and in private practice in Amherst,
Massachusetts. He has been the Director of Developmental Clinics in
multiple community hospitals in Western Massachusetts since his training at the
State University of New York at Buffalo, where he earned his medical degree. Dr.
Rosen completed his residency at Boston City Hospital and post-graduate
developmental pediatric training at Children’s Hospital Medical Center in Boston
in 1976. Dr. Rosen earned his M.D. from the University of New York at
Buffalo in 1971 and his Bachelors of Science Degree from the University of Rhode
Island in 1967. Dr. Rosen is a nationally known speaker and advisory
board member to numerous international pharmaceutical companies.
Thomas J. Mahowald, Director. On
December 31, 2010, our Board of Directors appointed Mr. Thomas J.
Mahowald as a member of the Board of Directors. From 2009 to the
present, Mr. Mahowald has served as the Vice President, Sales & Marketing of
Pike Research Group, a global leader in Cleantech market
research. From 2004 to 2008, Mr. Mahowald served as Principal of
Encompass Technology Partners, a business development firm specializing in
strategic planning and sales strategy, product branding
and positioning, merchandising, and global sales channel development,
including e-commerce. From 1996 to 2004, Mr. Mahowald served as Vice
President of Sales, Major Accounts for International Data Corporation, a global
provider of market intelligence, advisory services, and events for the
information technology, telecommunications, and consumer technology
markets. Prior to 1996, Mr. Mahowald’s served as Vice President,
Sales for Studio Solutions, Inc. from 1991 to 1996; Director of International
Marketing for Precision Visuals, Inc. from 1983 to 1991; and International Sales
Manager for the Satellite Communication Division of Harris Corporation form 1981
to 1983. Mr. Mahowald received his M.B.A in International Management
from the Thunderbird School of Global Management in 1981 and his B.S. in Finance
and Accounting from the University of Colorado at Boulder in 1977.
Significant
Employees
As of
September 30, 2010, we had no full-time employees, although we engage
contractors as necessary and each of our officers and directors devotes a
portion of his and her time to the affairs of our Company.
Certain
Relationships
There are
no arrangements, understandings, or family relationships pursuant to which our
executive officers or directors were selected. There are no related
party transactions between us and our executive officers or
directors.
Nominations
to the Board of Directors
Our
directors take a critical role in guiding our strategic direction and oversee
the management of the Company. Board of Director candidates are
considered based upon various criteria, such as their broad-based business and
professional skills and experiences, a global business and social perspective,
concern for the long-term interests of the stockholders, diversity, and personal
integrity and judgment. Accordingly, we seek to attract and retain
highly qualified directors.
In
carrying out its responsibilities, the Board of Directors will consider
candidates suggested by stockholders. If a stockholder wishes to
formally place a candidate's name in nomination, however, he or she must do so
in accordance with the provisions of the Company's Bylaws.
Committees
of the Board of Directors
Audit
Committee
Our Board
of Directors has not established a
separate audit committee within the meaning of
Section 3(a)(58)(A) of the Securities Exchange Act of
1934, as
amended (the ”Exchange Act”). Instead,
the entire Board of Directors acts as the audit committee within the meaning of
Section 3(a)(58)(B) of the Exchange Act. We are seeking
candidates for outside directors to serve on a separate audit committee when we
establish one. Due to our small size and limited operations and
resources, it has been difficult to recruit outside directors.
26
Audit
Committee Financial Expert
We
currently do not have an audit committee financial expert or an independent
audit committee expert on our Board of Directors.
Other
Committees
Our Board
of Directors has not established separately-designated standing nominating or
compensation committees due to our financial conditions, small size, and limited
operations and resources.
Code
of Ethical Conduct
We have
adopted a Code of Ethical Conduct applicable to all employees including our
principal executive officer and principal financial officer, which is available
on our website at http://www.uvflutech.com.
Compliance
with Section 16(a) of the Securities Exchange Act of 1934
Section
16(a) of the Securities and Exchange Act of 1934, as amended, requires any
person who is a director or executive officer or who beneficially holds more
than ten percent (10%) of any class of our securities which have been registered
with the Securities and Exchange Commission, to file reports of initial
ownership and changes in ownership with the Securities and Exchange
Commission. These persons are also required under the regulations of
the Securities and Exchange Commission to furnish us with copies of all Section
16(a) reports they file.
To our
knowledge, based solely on our review of the copies of the Section 16(a) reports
furnished to us and a review of our shareholders of record for the fiscal year
ended September 30, 2010, we believe that each person who, at any time during
such fiscal year was a director, officer, or beneficial owner of more than 10%
of our common stock, complied with all Section 16(a) filing requirements during
such fiscal year except that Mr. John J. Lennon filed one late report on Form 4
relating to the acquisition of shares of our common stock.
ITEM
11. EXECUTIVE
COMPENSATION
General
Philosophy
Our Board
of Directors is responsible for establishing and administering the Company’s
executive and director compensation.
Executive
Compensation
The Board
of Directors has approved a management fee to Mr. Lennon, our President,
Treasurer, Chief Executive Officer, Chief Financial Officer, and Director, in
the amount of $1,500 per week. This weekly fee will pay Mr. Lennon
for time in performing administrative, management, and business development
functions for us. This fee was determined by our Board of Directors
by considering the amount of time Mr. Lennon will provide to the Company and
also taking into consideration the financial condition of the
Company.
Compensation
Summary
The
summary compensation table below shows certain compensation information for
services rendered in all capacities to us by our principal executive officer and
by each other executive officer whose total annual salary and bonus exceeded
$100,000 during the fiscal periods ended September 30, 2010 and September 30,
2009. Other than as set forth below, no executive officer’s total
annual compensation exceeded $100,000 during our last fiscal
period.
27
Summary
Compensation Table
|
Name and Principal
Position (a)
|
Year
(b)
|
Salary
($)
(c)
|
Bonus
($)
(d)
|
Stock
Awards
($)
(e)(2)
|
Option
Awards
($)
(f)
|
Non
Equity
Incentive
Plan
Compensa
tion
($)
(g)
|
Non-qualified
Deferred
Compensation
Earnings
($)
(h)
|
All Other
Compensa
tion
($)
(i)
|
Total
($)
(j)
|
||||||||||||||||||||||||
|
John
J. Lennon, President, Treasurer, Chief Executive Officer, Chief Financial
Officer, and Director
|
2010
|
$
|
78,000
|
$
|
-0-
|
$
|
-0-
|
$
|
-0-
|
-0-
|
-0-
|
$
|
-0-
|
$
|
78,000
|
||||||||||||||||||
|
2009
|
$
|
-0-
|
$
|
-0-
|
$
|
-0-
|
$
|
-0-
|
-0-
|
-0-
|
$
|
-0-
|
$
|
-0-
|
|||||||||||||||||||
|
Roger
Nelson, Secretary
|
2010
|
$
|
-0-
|
$
|
-0-
|
$
|
-0-
|
$
|
-0-
|
-0-
|
-0-
|
$
|
-0-
|
$
|
-0-
|
||||||||||||||||||
|
2009
|
$
|
-0-
|
$
|
-0-
|
$
|
-0-
|
$
|
-0-
|
-0-
|
-0-
|
$
|
-0-
|
$
|
-0-
|
|||||||||||||||||||
Compensation
of Directors
We have
no standard arrangement to compensate directors for their services in their
capacity as directors. Directors are not paid for meetings
attended. We do reimburse directors for reasonable expenses incurred
during the course of their performance. There has been no
compensation awarded to, earned by, or paid to any of our named directors during
the last fiscal year.
Outstanding
Equity Awards at Fiscal 2010 Year-End
None of
our named executive officers had any outstanding equity awards as of the fiscal
year ended September 30, 2010.
Option
Exercises and Vested Stock in Fiscal 2010
None of
our named executive officers exercised stock options or had any restricted stock
during the fiscal year ended September 30, 2010.
Employment
and Other Agreements
There are
no employment agreements with any officers or directors other than the monetary
payment to them as more fully described elsewhere in this Form
10-K.
We do not
offer any annuity, pension, or retirement benefits to be paid to any of our
officers, directors, or employees in the event of retirement. There
are also no compensatory plans or arrangements with respect to any individual
named above which results or will result from the resignation, retirement, or
any other termination of employment with our company, or from a change in the
control of our Company.
28
Risk
Management Considerations
We
believe that our compensation policies and practices for our employees,
including our executive officers, do not create risks that are reasonably likely
to have a material adverse effect on our Company.
|
ITEM
12.
|
SECURITY
OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED
STOCKHOLDER MATTERS.
|
Security
Ownership Of Certain Beneficial Owners And Management
The
following table sets forth, as of January 10, 2011, the number and
percentage of outstanding shares of our common stock owned by (i) each person
known to us to beneficially own more than 5% of our outstanding common stock,
(ii) each director, (iii) each named executive officer, and (iv) all executive
officers and directors as a group. Share ownership is deemed to
include all shares that may be acquired through the exercise or conversion of
any other security immediately or within the next 60 days. Such
shares that may be so acquired are also deemed outstanding for purposes of
calculating the percentage of ownership for that individual or any group of
which that individual is a member. Unless otherwise indicated, the
stockholders listed possess sole voting and investment power with respect to the
shares shown.
|
Name and Address of Beneficial Owner
(1)
|
Amount and Nature of
Beneficial Ownership (2)
|
Percent of Class
of Common Stock
|
||||||
|
Directors and Executive
Officers
|
||||||||
|
John
J. Lennon
1694
Falmouth Road, Suite 147
Centerville,
Massachusetts 02632-2933
|
6,525,000
|
14.47
|
%
|
|||||
|
Roger
Nelson
103
Lawlor
Tolland
CT 06084
|
300,000
|
*
|
||||||
|
Glenn
Bushee
1145
Franklin Street
Duxbury,
MA 02332
|
375,000
|
*
|
||||||
|
Dr.
Dennis J. Rosen, M.D., FAAP
157
Columbia Dr
Amherst,
MA 01002
|
150,000
|
*
|
||||||
|
All
directors and executive officers as a group (4 persons)
|
7,350,000
|
16.30
|
%
|
|||||
|
5%
Stockholders
|
||||||||
|
Nu
Earth Corp.
495
Grand Boulevard
Suite
206
Miramar
Beach, FL 32550
|
3,750,000
|
8.32
|
%
|
|||||
|
*
|
Less
than 1%.
|
|
(1)
|
Unless
otherwise noted, the security ownership disclosed in this table is of
record and beneficial. Unless provided for otherwise, the
address for each of the beneficial owners named below is the Company’s
business address.
|
|
(2)
|
Under
Rule 13d-3 under the Exchange Act, shares not outstanding but subject to
options, warrants, rights, conversion privileges pursuant to which such
shares may be acquired in the next 60 days are deemed to be outstanding
for the purpose of computing the percentage of outstanding shares owned by
the persons having such rights, but are not deemed outstanding for the
purpose of computing the percentage for such other
persons.
|
29
We do not
know of any other shareholder who has more than 5 percent of the issued
shares.
There are
no voting trusts or similar arrangements known to us whereby voting power is
held by another party not named herein. We know of no trusts,
proxies, power of attorney, pooling arrangements, direct or indirect, or any
other contract arrangement or device with the purpose or effect of divesting
such person or persons of beneficial ownership of our common shares or
preventing the vesting of such beneficial ownership.
Changes
in Control
Other
than as disclosed elsewhere in this Form 10-K, there are no existing
arrangements that may result in a change in control of the Company.
Securities
Authorized for Issuance Under Equity Compensation Plans
We do not
have any equity compensation plans.
ITEM
13. CERTAIN
RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE.
Related
Party Transactions
During
our fiscal year ended September 30, 2010 and the previous fiscal year, there
were no transactions with related parties.
Review,
Approval or Ratification of Transactions with Related Persons
Although
we adopted a Code of Ethical Conduct on December 10, 2009, we still rely on our
board to review related party transactions on an ongoing basis to prevent
conflicts of interest. Our board reviews a transaction in light of
the affiliations of the director, officer or employee and the affiliations of
such person’s immediate family. Transactions are presented to our
board for approval before they are entered into or, if this is not possible, for
ratification after the transaction has occurred. If our board finds
that a conflict of interest exists, then it will determine the appropriate
remedial action, if any. Our board approves or ratifies a transaction
if it determines that the transaction is consistent with the best interests of
the Company. For the above transaction, the board approved and
ratified the transaction, finding it in the best interest of the
Company.
Director
Independence
During fiscal 2010, our Board of
Directors has determined, after considering all the relevant facts and
circumstances, that Mr. Roger Nelson, Mr. Thomas J. Mahowald, and Dr. Dennis J.
Rosen are independent directors because they have no relationship with us that
would interfere with their exercise of independent judgment. Mr.
Lennon is not an independent director of our company because of his past or
current position as an executive officer of our company. There are no
family relationships among any of our directors or officers. We
evaluate independence by the standards for director independence established by
applicable laws, rules, and listing standards including, without limitation, the
standards for independent directors established by The New York Stock Exchange,
Inc., The NASDAQ National Market, and the Securities and Exchange
Commission.
Subject
to some exceptions, these standards generally provide that a director will not
be independent if (a) the director is, or in the past three years has been,
an employee of ours; (b) a member of the director’s immediate family is, or
in the past three years has been, an executive officer of ours; (c) the
director or a member of the director’s immediate family has received more than
$120,000 per year in direct compensation from us other than for service as a
director (or for a family member, as a non-executive employee); (d) the
director or a member of the director’s immediate family is, or in the past three
years has been, employed in a professional capacity by our independent public
accountants, or has worked for such firm in any capacity on our audit;
(e) the director or a member of the director’s immediate family is, or in
the past three years has been, employed as an executive officer of a company
where one of our executive officers serves on the compensation committee; or
(f) the director or a member of the director’s immediate family is an
executive officer of a company that makes payments to, or receives payments
from, us in an amount which, in any twelve-month period during the past three
years, exceeds the greater of $1,000,000 or two percent of that other company’s
consolidated gross revenues.
30
ITEM
14. PRINCIPAL
ACCOUNTING FEES AND SERVICES.
The
following table shows the fees paid or accrued by us for the audit and other
services provided by Weaver & Martin for the fiscal periods
shown.
|
September 30, 2010
|
September
30, 2009
|
|||||||
|
Audit
Fees &
|
$ | $ | ||||||
|
Audit
Related Fees
|
12,500 | 12,000 | ||||||
|
Tax
Fees
|
— | — | ||||||
|
All
Other Fees
|
— | — | ||||||
|
Total
|
$ | 12,500 | $ | 12,000 | ||||
Audit
fees consist of fees billed for professional services rendered for the audit of
our financial statements and review of the interim financial statements included
in quarterly reports and services that are normally provided by the above
auditors in connection with statutory and regulatory fillings or
engagements
In the
absence of a formal audit committee, the full Board of Directors pre-approves
all audit and non-audit services to be performed by the independent registered
public accounting firm in accordance with the rules and regulations promulgated
under the Securities Exchange Act of 1934, as amended. The Board of
Directors pre-approved 100% of the audit, audit-related and tax services
performed by the independent registered public accounting firm for the fiscal
year ended September 30, 2010. The percentage of hours expended on
the principal accountant’s engagement to audit the Company’s financial
statements for the most recent fiscal year that were attributed to work
performed by persons other than the principal accountant’s full-time, permanent
employees was 0%.
31
PART
IV
ITEM
15. EXHIBITS,
FINANCIAL STATEMENT SCHEDULES.
Exhibits
The
following exhibits are included as part of this report by
reference:
|
3.1(a)
|
Articles
of Incorporation, as amended (1)
|
|
3.1(b)
|
Articles
of Merger as filed with the Secretary of State of Nevada on November 12,
2009 (2)
|
|
3.2
|
Bylaws
of the Company (3)
|
|
10.1
|
Asset
Purchase Agreement dated December 16, 2009 (4)
|
|
10.2
|
Letter
of Intent by and between The Red Oak Trust and UV Flu Technologies, Inc.,
dated October 28, 2010 (5)
|
|
14.1
|
Code
of Ethics (6)
|
|
16.1
|
Letter
regarding change of Independent Registered Public Accounting Firm
(7)
|
|
31.1
|
Rule
13(a) — 14(a)/15(d) — 14(a) Certification (Principal Executive
Officer)
|
|
31.2
|
Rule
13(a) — 14(a)/15(d) — 14(a) Certification (Principal Financial
Officer)
|
|
32
|
Certification
Pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of
the Sarbanes-Oxley Act of 2002
|
(1)
Incorporated herein by reference to exhibits previously filed on Registrant’s
Current Report on Form 8-K, filed on November 12, 2009.
(2)
Incorporated herein by reference to the Articles of Merger as filed with the
Secretary of State of Nevada on November 12, 2009.
(3)
Incorporated herein by reference to exhibits previously filed on Registrant’s
Current Report on Form SB-2, filed on January 30, 2007.
(4)
Incorporated herein by reference to exhibits previously filed on Registrant’s
Current Report on Form 8-K, filed on December 18, 2009.
(5)
Incorporated herein by reference to exhibits previously filed on Registrant’s
Current Report on Form 8-K, filed on October 29, 2010.
(6)
Incorporated herein by reference to exhibits previously filed on Registrant’s
Annual Report on Form 10-K, filed on December 29, 2009.
(7)
Incorporated herein by reference to exhibits previously filed on Registrant’s
Current Report on Form 8-K, filed on December 23, 2009.
32
SIGNATURES
Pursuant
to the requirements of Section 13 or 15(d) of the Securities Exchange Act of
1934, the registrant has duly caused this report to be signed on its behalf by
the undersigned, thereunto duly authorized.
| UV FLU TECHNOLOGIES, INC. | |||
|
Date:
January 13, 2011
|
By:
|
/s/ John Lennon | |
|
John
Lennon
Chief
Executive Officer, Chief Financial Officer,
President, and Director
|
|||
Pursuant
to the requirements of the Securities Exchange Act of 1934, this report has been
signed below by the following persons on behalf of the registrant and in the
capacities and on the dates indicated.
|
Date:
January 13, 2011
|
By:
|
/s/ John Lennon | |
|
John
Lennon
Chief
Executive Officer, Chief Financial Officer,
President, and Director
|
|||
|
Date:
January 13, 2011
|
By:
|
/s/ Roger Nelson | |
|
Roger
Nelson
Secretary
and Director
|
|||
|
Date:
January 13, 2011
|
By:
|
/s/ Thomas J. Mahowald | |
|
Thomas
J. Mahowald
Director
|
|||
|
Date:
January 13, 2011
|
By:
|
/s/ Dr. Dennis J. Rosen | |
|
Dr.
Dennis J. Rosen, M.D., FAAP
Director
|
|||
33
UV
FLU TECHNOLOGIES, INC
(A
Development Stage Company)
INDEX TO
FINANCIAL STATEMENTS
with
REPORT OF
INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
SEPTEMBER
30, 2010
|
Page
|
||
|
Report
of Independent Registered Public Accounting Firm Weaver &
Martin
|
F-2
|
|
|
Financial
Statements:
|
||
|
Balance
Sheets at September 30, 2010 and 2009
|
F-3
|
|
|
Statements
of Operations for the years ended September 30, 2010 and 2009 and from
April 4, 2006 through September 30, 2010
|
F-4
|
|
|
Statements
of Stockholders’ Equity from April 4, 2006 through September 30,
2010
|
F-5
|
|
|
Statements
of Cash Flows for the years ended September 30, 2010 and 2009 and from
April 4, 2006 through September 30, 2010
|
F-6
|
|
|
Notes
to Financial Statements
|
|
F-7
|
F-1
REPORT
OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
WEAVER
& MARTIN
To the
Board of Directors and Stockholders
UV Flu
Technologies, Inc.
Centerville,
Massachusetts
REPORT OF
INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
We have
audited the balance sheet of UV Flu Technologies, Inc. (a development stage
company) as of September 30, 2010 and 2009 and the related statements of
operations, stockholders’ equity, and cash flows for the years then ended and
the period of April 4, 2006 (inception) to September 30, 2010. UV Flu
Technologies, Inc.’s management is responsible for these financial
statements. Our responsibility is to express an opinion on these
financial statements based on our audit.
We
conducted our audit in accordance with the standards of the Public Company
Accounting Oversight Board (United States). Those standards require
that we plan and perform the audit to obtain reasonable assurance about whether
the financial statements are free of material misstatement. The
Company is not required to have, nor were we engaged to perform, an audit of its
internal control over financial reporting. Our audit included
consideration of internal control over financial reporting as a basis for
designing audit procedures that are appropriate in the circumstances, but not
for the purpose of expressing an opinion on the effectiveness of the Company’s
internal control over financial reporting. Accordingly, we express no
such opinion. Our audit of the financial statements includes
examining, on a test basis, evidence supporting the amounts and disclosures in
the financial statements, assessing the accounting principles used and
significant estimates made by management, and evaluating the overall financial
statement presentation. We believe that our audit provides a
reasonable basis for our opinion.
In our
opinion, the financial statements referred to above present fairly, in all
material respects, the financial position of UV Flu Technologies, Inc. as of
September 30, 2010 and 2009 and the results of its operations, stockholders’
equity, and cash flows for the years then ended and the period of April 4, 2006
(inception) to September 30, 2010 in conformity with accounting principles
generally accepted in the United States of America.
The
accompanying financial statements have been prepared assuming that the Company
will continue as a going concern. As discussed in Note 2 to the financial
statements, the Company has suffered recurring losses from operations and is
dependent upon the continued sale of its securities or obtaining debt financing
for funds to meet its cash requirements. These factors raise substantial doubt
about the Company’s ability to continue as a going concern. The
financial statements do not include any adjustments that might result from the
outcome of this uncertainty.
/s/
Weaver & Martin, LLC
Weaver
& Martin, LLC
Kansas
City, Missouri
January
13, 2011
F-2
UV
FLU TECHNOLOGIES, INC
(A
Development Stage Company)
BALANCE
SHEETS
|
September
30, 2010
|
September
30, 2009
|
|||||||
|
ASSETS
|
||||||||
|
Current
Assets
|
||||||||
|
Cash
|
$ | - | $ | 2,819 | ||||
|
Accounts
receivable
|
7,404 | - | ||||||
|
Prepaid
|
95,908 | - | ||||||
|
Total
Current Assets
|
103,312 | 2,819 | ||||||
|
Inventory
|
66,815 | - | ||||||
|
Property
and Equipment
|
||||||||
|
Equipment, net of accumulated
depreciation of $nil and $2,587
|
- | 2,284 | ||||||
|
Total
Assets
|
$ | 170,127 | $ | 5,103 | ||||
|
LIABILITIES
|
||||||||
|
Current
Liabilities:
|
||||||||
|
Accounts payable and accrued
expenses
|
$ | 149,775 | $ | 4,962 | ||||
|
Loans payable (Note
5)
|
541,864 | - | ||||||
|
Total Current
Liabilities
|
691,639 | 4,962 | ||||||
|
STOCKHOLDERS’
EQUITY
|
||||||||
|
Capital
Stock (Note 6)
|
||||||||
|
Authorized:
|
||||||||
|
75,000,000 common shares, par
value $0.001 per share
|
||||||||
|
Issued and
outstanding:
|
||||||||
|
45,080,000 common shares at
September 30, 2010 and142,080,000 common shares at September 30,
2009,
|
45,080 | 4,440 | ||||||
|
Additional
paid-in capital
|
128,420 | 124,560 | ||||||
|
Deficit
Accumulated During the Development Stage
|
(695,012 | ) | (128,859 | ) | ||||
|
Total
Stockholders’ Equity
|
(521,512 | ) | 141 | |||||
|
Total
Liabilities and Stockholders’ Equity
|
$ | 170,127 | $ | 5,103 | ||||
The
accompanying notes are an integral part of these statements.
F-3
UV
FLU TECHNOLOGIES, INC
(A
Development Stage Company)
STATEMENTS
OF OPERATIONS
|
Year
Ended
September
30, 2010
|
Year
Ended
September
30, 2009
|
Cumulative
April 4, 2006 (Inception) Through
September
30, 2010
|
||||||||||
|
Sales
and Rental Revenues
|
$ | 107,703 | $ | 18,000 | $ | 143,603 | ||||||
|
Cost
of Sales
|
85,582 | 11,120 | 106,202 | |||||||||
|
Gross
Profit
|
22,121 | 6,880 | 37,401 | |||||||||
|
Expenses
|
||||||||||||
|
Bad debt
|
67,500 | - | 67,500 | |||||||||
|
Depreciation and
amortization
|
- | 3,922 | 6,064 | |||||||||
|
Marketing
|
89,541 | - | 108,500 | |||||||||
|
Office and
administration
|
58,756 | 5,860 | 99,101 | |||||||||
|
Organizational
costs
|
- | - | 1,705 | |||||||||
|
Professional
fees
|
130,965 | 17,948 | 209,113 | |||||||||
|
Consulting
|
152,924 | - | 152,924 | |||||||||
|
Investor
relations
|
67,042 | - | 67,042 | |||||||||
|
Total
Expenses
|
566,728 | 27,730 | 711,949 | |||||||||
|
(Loss)
from Operations
|
(544,607 | ) | (20,850 | ) | (674,548 | ) | ||||||
|
Other
Income (Expense)
|
||||||||||||
|
Gain on sale of
assets
|
1,116 | - | 1,116 | |||||||||
|
Interest expense
|
(22,659 | ) | - | (22,659 | ) | |||||||
|
Gain (Loss) on
foreign
exchange
|
(3 | ) | 22 | 1,079 | ||||||||
|
Net
(Loss)
|
$ | (566,153 | ) | $ | (20,828 | ) | $ | (695,012 | ) | |||
|
Basic
And Diluted Loss Per Share
|
$ | (0.01 | ) | $Nil | ||||||||
|
Weighted
Average Number Of Shares Outstanding
|
45,597,808 | 142,080,000 | ||||||||||
The
accompanying notes are an integral part of these statements.
F-4
UV
FLU TECHNOLOGIES, INC
(A
Development Stage Company)
STATEMENT
OF STOCKHOLDERS’ EQUITY AND OTHER COMPREHENSIVE INCOME
| Deficit | ||||||||||||||||||||||||
| Accumulated | Accumulated | |||||||||||||||||||||||
|
Additional
|
during the | Other | ||||||||||||||||||||||
|
Capital
Stock
|
Paid-in
|
development
|
Comprehensive | |||||||||||||||||||||
|
Shares
|
Amount
|
Capital
|
stage
|
Income
(loss)
|
Total
|
|||||||||||||||||||
|
Inception,
|
||||||||||||||||||||||||
|
April
4, 2006 - Shares issued for cash at $0.01
|
1,500,000 | $ | 1,500 | $ | 13,500 | $ | - | $ | - | $ | 15,000 | |||||||||||||
|
September
25, 2006 - Shares issued for cash at $0.01
|
2,000,000 | 2,000 | 18,000 | - | - | 20,000 | ||||||||||||||||||
|
Net
loss for the period ended September 30, 2006
|
- | - | - | (1,751 | ) | - | (1,751 | ) | ||||||||||||||||
|
Balance,
September 30, 2006
|
3,500,000 | 3,500 | 31,500 | (1,751 | ) | - | 33,249 | |||||||||||||||||
|
September
18, 2007 - Shares issued for cash at $0.10
|
940,000 | 940 | 93,060 | - | - | 94,000 | ||||||||||||||||||
|
Net
loss for the year ended September 30, 2007
|
- | - | - | (36,600 | ) | - | (36,849 | ) | ||||||||||||||||
| - | ||||||||||||||||||||||||
|
Balances,
September 30, 2007
|
4,440,000 | 4,440 | 124,560 | (38,600 | ) | - | 90,400 | |||||||||||||||||
|
Net
loss for the year ended September 30, 2008
|
- | - | - | (68,355 | ) | - | (68,355 | ) | ||||||||||||||||
|
Change
in other comprehensive income - Foreign
|
||||||||||||||||||||||||
|
currency
translation adjustment
|
- | - | - | - | (840 | ) | (840 | ) | ||||||||||||||||
|
Total
comprehensive loss (Memo total)
|
(69,195 | ) | ||||||||||||||||||||||
|
Balances,
September 30, 2008
|
4,440,000 | 4,440 | 124,560 | (106,955 | ) | (840 | ) | 21,205 | ||||||||||||||||
|
Net
Loss for the year ended September 30, 2009
|
- | - | - | (20,828 | ) | - | (20,828 | ) | ||||||||||||||||
|
Change
in other comprehensive income - Foreign
|
||||||||||||||||||||||||
|
currency
translation adjustment
|
- | - | - | - | (236 | ) | (236 | ) | ||||||||||||||||
|
Total
comprehensive loss (Memo total)
|
(21,064 | ) | ||||||||||||||||||||||
|
Balances,
September 30, 2009
|
4,440,000 | 4,440 | 124,560 | (127,783 | ) | (1,076 | ) | 141 | ||||||||||||||||
|
October
12, 2009 - Shares surrendered and cancelled
|
(3,500,000 | ) | (3,500 | ) | 3,500 | - | - | - | ||||||||||||||||
|
November
12, 2009 - Forward split of shares of 32.1
|
29,140,000 | 29,140 | (29,140 | ) | - | |||||||||||||||||||
|
December
16, 2009 - Shares issued for assets
|
15,000,000 | 15,000 | 29,500 | 44,500 | ||||||||||||||||||||
|
Net
Loss or the year ended September 30, 2010
|
(566,153 | ) | (566,153 | ) | ||||||||||||||||||||
|
Consolidate
- Foreign currency translation adjustment
|
(1,076 | ) | 1,076 | - | ||||||||||||||||||||
|
Balances,
September 30, 2010
|
45,080,000 | $ | 45,080 | $ | 128,420 | $ | (695,012 | ) | $ | - | $ | (521,512 | ) | |||||||||||
The
accompanying notes are an integral part of these statements.
F-5
UV
FLU TECHNOLOGIES, INC
(A
Development Stage Company)
STATEMENTS
OF CASH FLOWS
|
Year
ended
September
30, 2010
|
Year
ended
September
30, 2009
|
Cumulative
April 4, 2006 (Inception)
Through
September
30, 2010
|
||||||||||
|
Cash
Flows from Operating Activities:
|
||||||||||||
|
Net
(loss)
|
$ | (566,153 | ) | $ | (20,828 | ) | $ | (695,012 | ) | |||
|
Adjustments
to Reconcile Net Loss to Net Cash Used by Operating
Activities:
|
||||||||||||
|
Depreciation
and amortization
|
- | 3,922 | 6,064 | |||||||||
|
Gain
on sale of assets
|
(1,116 | ) | - | (1,116 | ) | |||||||
|
Changes
in current assets and liabilities
|
||||||||||||
|
Accounts
receivable
|
(7,404 | ) | - | (7,404 | ) | |||||||
|
Prepaid
|
(95,908 | ) | - | (95,908 | ) | |||||||
|
Inventory
|
(22,315 | ) | 11,119 | (22,315 | ) | |||||||
|
Accounts
payable and accrued liabilities
|
144,813 | 3,837 | 149,775 | |||||||||
|
Net
Cash Flows provided by Operating Activities
|
(548,083 | ) | (1,950 | ) | (665,916 | ) | ||||||
|
Cash
Flows from Investing Activities:
|
||||||||||||
|
Purchase
of equipment
|
- | - | (4,871 | ) | ||||||||
|
Sales
proceeds of equipment
|
3,400 | - | 3,400 | |||||||||
|
Increase
in website development costs
|
- | - | (3,477 | ) | ||||||||
|
Net
Cash Flows provided by Investing Activities
|
3,400 | - | (4,948 | ) | ||||||||
|
Cash
Flows From Financing Activity:
|
||||||||||||
|
Proceeds
from loans payable
|
541,864 | - | 541,864 | |||||||||
|
Sale
of common shares
|
- | - | 129,000 | |||||||||
|
Net
Cash Flows provided by Financing Activities
|
541,864 | - | 670,864 | |||||||||
|
Net
Cash Flows
|
(2,819 | ) | (1,950 | ) | - | |||||||
|
Cash,
Beginning Of Period
|
2,819 | 4,769 | - | |||||||||
|
Cash,
End Of Period
|
$ | - | $ | 2,819 | $ | - | ||||||
|
Supplemental
Disclosure Of Cash Flow Information
|
||||||||||||
|
Cash
paid for:
|
||||||||||||
|
Interest
|
$ | - | $ | - | $ | - | ||||||
|
Income
taxes
|
$ | - | $ | - | $ | - | ||||||
|
Inventory
acquired through issuance of shares
|
$ | 44,500 | $ | - | $ | 44,500 | ||||||
|
Shares
issued
|
15,000,000 | - | 15,000,000 | |||||||||
The
accompanying notes are an integral part of these statements.
F-6
UV
FLU TECHNOLOGIES, INC
(A
Development Stage Company)
NOTES
TO FINANCIAL STATEMENTS
|
1.
|
NATURE
AND CONTINUANCE OF OPERATIONS
|
|
|
a)
|
Organization
|
UV FLU
TECHNOLOGIES, INC (referred to herein as “we”, “us”, “our” and similar terms)
was incorporated as NORTHWEST CHARIOTS INCORPORATED in the State of Nevada,
United States of America, on April 04, 2006. On November 12, 2009,
the Company changed its name from “Northwest Chariots Incorporated” to “UV Flu
Technologies, Inc. The Company year-end is September
30th.
|
|
b)
|
Development
Stage Activities
|
The
Company is in the development stage and has only realized minimal revenue from
our planned operations. To generate revenue, our new business plan is
to focus on the research, development, manufacturing and sales of air
purification systems and products. We will focus initially on the
commercial market, targeting medical, hospitality and commercial property
customers in the United States.
Based
upon our business plan, we are a development stage
enterprise. Accordingly, we present our financial statements in
conformity with the accounting principles generally accepted in the United
States of America that apply in establishing operating
enterprises. As a development stage enterprise, we disclose the
deficit accumulated during the development stage and the cumulative statements
of operations and cash flows from our inception to the current balance sheet
date.
|
2.
|
BASIS
OF PRESENTATION – GOING CONCERN
|
Our
accompanying financial statements have been prepared in conformity with GAAP,
which contemplates our continuation as a going concern. However, we
have minimal business operations to date. In addition, at September
30, 2010, we had incurred losses of $695,012, and have working capital deficit
of $588,327. These matters raise substantial doubt about our ability
to continue as going concern. In view of these matters, realization
of certain of the assets in the accompanying balance sheet is dependent upon our
ability to meet our financing requirements, raise additional capital, and the
success of our future operations. There is no assurance that future
capital raising plans will be successful in obtaining sufficient funds to assure
our eventual profitability. While management believes that actions
planned and presently being taken to revise our operating and financial
requirements provide the opportunity for us to continue as a going concern,
there is no assurance the actions will be successful. In addition,
recent events in worldwide capital markets may make it more difficult for us to
raise additional equity or debt capital.
Our
financial statements do not include any adjustments that might result from these
uncertainties.
|
3.
|
SIGNIFICANT
ACCOUNTING POLICIES
|
This
summary of significant accounting policies is presented to assist in
understanding our financial statements. Our financial statements and
notes are representations of our management who is responsible for their
integrity and objectivity. These accounting policies conform to
generally accepted accounting principles in the United States of America
(“GAAP”) and have been consistently applied in the preparation of the financial
statements. The financial statements are stated in United States of
America dollars.
|
|
a)
|
Organizational
and Start-up Costs
|
Costs of
start-up activities, including organizational costs, are expensed as incurred in
accordance with Accounting Standards Codification (“ASC”) subtopic 720-15
(formerly Statements of Position (“SOP”) 98-5).
F-7
UV
FLU TECHNOLOGIES, INC
(A
Development Stage Company)
NOTES
TO FINANCIAL STATEMENTS
|
3.
|
SIGNIFICANT
ACCOUNTING POLICIES (cont’d)
|
|
|
b)
|
Income
Taxes
|
We have
adopted the ASC subtopic 740-10
(formerly Statement of Financial Accounting Standards (“SFAS”) No. 109 –
“Accounting for Income Taxes”). ASC 740-10 requires the use of the
asset and liability method of accounting of income taxes. Under the
asset and liability method of ASC 740-10, deferred tax assets and liabilities
are recognized for the future tax consequences attributable to temporary
differences between the financial statements carrying amounts of existing assets
and liabilities and their respective tax bases. Deferred tax assets
and liabilities are measured using enacted tax rates expected to apply to
taxable income in the years in which those temporary differences are expected to
be recovered or settled.
We
provide deferred taxes for the estimated future tax effects attributable to
temporary differences and carry forwards when realization is more likely than
not. See Note 7.
|
|
c)
|
Inventories
|
Inventories
are stated at the lower of cost or market, with cost being determined using the
first-in first-out method. Inventories consist of purchased goods
held for resale.
|
|
d)
|
Property
and Equipment
|
Property
and equipment is stated at cost, and is depreciated over estimated useful lives
using primarily the straight line method for financial reporting
purposes. Useful lives range from 3 to 5 years. We
evaluate equipment at least annually for impairment. As of September
30, 2010, all of the property and equipment had been sold for a net gain of
$1,116. The Company has no property and equipment as of
September 30, 2010.
|
|
e)
|
Advertising
|
Advertising
costs are expensed as in accordance with ASC subtopic 720-35 (formerly SOP
93-7).
|
|
f)
|
Basic
and Diluted Loss Per Share
|
In
accordance with ASC subtopic 260-10 (formerly SFAS No. 128 – “Earnings Per
Share”), the basic loss per common share is computed by dividing net loss
available to common stockholders by the weighted average number of common shares
outstanding. Diluted loss per common share is computed similar to
basic loss per common share except that the denominator is increased to include
the number of additional common shares that would have been outstanding if the
potential common shares had been issued and if the additional common shares were
dilutive. At September 30, 2010 and 2009, we had no stock equivalents
that were anti-dilutive and excluded in the earnings per share
computation.
|
|
g)
|
Estimated
Fair Value of Financial Instruments
|
The
carrying value of our financial instruments, consisting of cash, and accounts
payable approximate their fair value due to the short-term maturity of such
instruments. Unless otherwise noted, it is management’s opinion that
we are not exposed to significant interest, currency or credit risks arising
from these financial statements. See Note 8.
|
|
h)
|
Revenue
Recognition
|
It is our
policy that revenues will be recognized in accordance with ASC subtopic 605-10
(formerly SEC Staff Accounting Bulletin (SAB) No. 104, "Revenue
Recognition. Revenues are recognized only when the Company has
transferred to the customer the significant risk and rewards of ownership of the
goods, title to the products transfers, the amount is fixed and determinable,
evidence of an agreement exists, there is reasonable assurance of collection of
the sales proceeds, the Company has no future obligations, and the customer
bears the risk of loss. This occurs at the time of shipment (FOB
shipping point) of the products from the Company’s warehouse and an invoice is
prepared.. There are no rights of return and exchange is allowed only on
defective products. Recognition of revenue is not affected as the right of
exchange results in new units being shipped to the customer once defective units
have been received by the company and verified as defective.
F-8
UV
FLU TECHNOLOGIES, INC
(A
Development Stage Company)
NOTES
TO FINANCIAL STATEMENTS
|
3.
|
SIGNIFICANT
ACCOUNTING POLICIES (cont’d)
|
|
|
i)
|
Currency
|
Our
functional currency is the United States Dollar. Realized gain or
loss on foreign currency transactions are reflected in the income
statement.
|
|
j)
|
Use
of Estimates
|
The
preparation of financial statements in conformity with GAAP requires management
to make estimates and assumptions that affect the reported amounts of assets and
liabilities and disclosure of contingent assets and liabilities at the date of
the financial statements and the reported amount of revenues and expenses during
the reporting period. Actual results could differ from those
estimates.
|
|
k)
|
Cash
and Cash Equivalents
|
Cash is
comprised of cash on hand and demand deposits. Cash equivalents include
short-term highly liquid investments with original maturities of three months or
less that are readily convertible to known amounts of cash and which are subject
to an insignificant risk of change in value.
|
l)
|
Concentrations
|
Financial
instruments that potentially subject us to concentrations of credit risk consist
principally of cash and cash equivalents.
|
|
m)
|
Recent
Accounting Pronouncements
|
In August
2010, the FASB issued Accounting Standard Updates No. 2010-21 (ASU No. 2010-21)
“Accounting for Technical Amendments to Various SEC Rules and Schedules” and No.
2010-22 (ASU No. 2010-22) “Accounting for Various Topics – Technical Corrections
to SEC Paragraphs”. ASU No 2010-21 amends various SEC paragraphs
pursuant to the issuance of Release no. 33-9026: Technical Amendments to Rules,
Forms, Schedules and Codification of Financial Reporting
Policies. ASU No. 2010-22 amends various SEC paragraphs based on
external comments received and the issuance of SAB 112, which amends or rescinds
portions of certain SAB topics. Both ASU No. 2010-21 and ASU No.
2010-22 are effective upon issuance. The amendments in ASU No.
2010-21 and No. 2010-22 will not have a material impact on the Company’s
financial statements.
|
|
International
Financial Reporting Standards
|
|
|
|
|
|
In
November 2008, the Securities and Exchange Commission (“SEC”) issued for
comment a proposed roadmap regarding potential use of financial statements
prepared in accordance with International Financial Reporting Standards
(“IFRS”) as issued by the International Accounting Standards Board. Under
the proposed roadmap, the Company would be required to prepare financial
statements in accordance with IFRS in fiscal year 2014, including
comparative information also prepared under IFRS for fiscal 2013 and 2012.
The Company is currently assessing the potential impact of IFRS on its
financial statements and will continue to follow the proposed roadmap for
future developments.
|
|
4.
|
ASSET
ACQUISITION
|
On
December 16, 2009, the Company entered into an Asset Purchase Agreement with
AmAirpure, Inc., a related party, whereby the Company acquires certain of their
assets relating to the design, development, and manufacture of technology and
products including air purification systems. The agreement resulted
in the issuance of 15,000,000 shares of common stock of the Company. The assets
acquired included inventory, valued at sellers cost of $44,500, and a patent
valued at $nil. The shares given were valued at the fair market value
of the assets acquired.
F-9
UV
FLU TECHNOLOGIES, INC
(A
Development Stage Company)
NOTES
TO FINANCIAL STATEMENTS
|
5.
|
LOANS
PAYABLE
|
At
September 30, 2010, the Company had unrelated third party loans payable totaling
$541,864 (September 30, 2009 - $nil). Interest expense relating to
these notes was $22,659 for the year ended September 30, 2010. The
loans bear interest at a rate of 10% per annum, and are due on
demand. Arrears in payment of the principal and any interest shall
bear interest at the rate of 30% per year calculated annually.
|
6.
|
COMMON
STOCK
|
Our
authorized common stock consists of 75,000,000 shares with a par value of $0.001
per share.
On April
04, 2006, we issued 48,000,000 shares of common stock at a price of $0.01 for
cash totalling $15,000.
On
November 25, 2006, we issued 64,000,000 shares of common stock at a price of
$0.01 for cash totalling $20,000.
On
September 18, 2007, we issued 30,080,000 shares of common stock at a price of
$0.10 for cash totalling $94,000.
On October 12, 2009, 112,000,000 shares
of common stock were surrendered and cancelled.
On November 12, 2009, a forward split
32:1 was approved and enacted.
On
December 16, 2009, we issued 15,000,000 shares of common stock for assets
purchased. See Note 4.
As at September 30, 2010 and 2009,
there are no outstanding options or warrants.
|
7.
|
INCOME
TAXES
|
We are
subject to US and Canadian income taxes. To date, we have accumulated
losses of approximately $695,012, and therefore have paid no income
tax. We expect tax rates in both the US and Canada to be
approximately 34%. Substantially all operations prior to September
30, 2009 were in Canada. Substantially all operations during the
period ending September 30, 2010 and going forward will be in the United
States.
Deferred
income taxes arise from temporary timing differences in the recognition of
income and expenses for financial reporting and tax purposes. Our
deferred tax assets consist entirely of the benefit from net operating loss
(“NOL”) carry-forwards. The NOL carry-forwards expire in
2028. Our deferred tax assets are offset by a valuation allowance due
to the uncertainty of the realization of the NOL carry-forwards. NOL
carry-forwards may be further limited by a change our ownership and other
provisions of the tax laws.
The
provision for refundable Federal income tax, using an effective tax rate of
thirty-four percent (34%), consists of the following:
|
Year
Ended
September
30,
2010
|
Year
Ended
September
30,
2009
|
|||||||
|
Refundable
Federal income tax attributable to:
|
||||||||
|
Current
operations
|
$ | (192,000 | ) | $ | (7,000 | ) | ||
|
Change
in deferred tax valuation allowance
|
192,000 | 7,000 | ||||||
|
Net
refundable amount
|
- | - | ||||||
F-10
UV
FLU TECHNOLOGIES, INC
(A
Development Stage Company)
NOTES
TO FINANCIAL STATEMENTS
|
7.
|
INCOME
TAXES (cont’d)
|
The
cumulative tax effect at the expected rate of thirty-four percent (34%) of
significant items comprising our net deferred tax amount is as
follows:
|
September
30,
2010
|
September
30,
2009
|
|||||||
|
Deferred
tax asset attributable to:
|
||||||||
|
Net
operating loss carryover
|
$ | 236,000 | $ | 43,400 | ||||
|
Less:
Valuation allowance
|
(236,000 | ) | (43,400 | ) | ||||
|
Net
deferred tax asset
|
- | - | ||||||
At
September 30, 2010, we had an unused Canadian NOL carryover of approximating
$128,859 that is available to offset future taxable income in Canada; it expires
the beginning in 2026. At September 30, 2010, we has unused NOL carryover of
approximately $566,153 that is available to offset future taxable income in the
U.S., it expires beginning in 2030.
|
8.
|
FAIR
VALUE MEASUREMENTS
|
The
Company adopted ASC Topic 820-10 at the beginning of 2009 to measure the fair
value of certain of its financial assets required to be measured on a recurring
basis. The adoption of ASC Topic 820-10 did not impact the Company’s
financial condition or results of operations. ASC Topic 820-10
establishes a fair value hierarchy that prioritizes the inputs to valuation
techniques used to measure fair value. The hierarchy gives the
highest priority to unadjusted quoted prices in active markets for identical
assets or liabilities (Level 1 measurements) and the lowest priority to
unobservable inputs (Level 3 measurements). ASC Topic 820-10 defines
fair value as the price that would be received to sell an asset or paid to
transfer a liability in an orderly transaction between market participants on
the measurement date. A fair value measurement assumes that the
transaction to sell the asset or transfer the liability occurs in the principal
market for the asset or liability. The three levels of the fair value
hierarchy under ASC Topic 820-10 are described below:
Level
1 – Valuations based on quoted prices in active markets for identical
assets or liabilities that an entity has the ability to access.
Level
2 – Valuations based on quoted prices for similar assets and liabilities in
active markets, quoted prices for identical assets and liabilities in markets
that are not active, or other inputs that are observable or can be corroborated
by observable data for substantially the full term of the assets or
liabilities.
Level
3 – Valuations based on inputs that are supportable by little or no market
activity and that are significant to the fair value of the asset or
liability.
The
following table presents a reconciliation of all assets and liabilities measured
at fair value on a recurring basis as of September 30, 2010:
|
Level
1
|
Level
2
|
Level
3
|
Fair
Value
|
|||||||||||||
|
Cash
|
$ | - | $ | - | $ | - | $ | - | ||||||||
|
Accounts
receivable
|
- | 7,404 | - | 7,404 | ||||||||||||
|
Accounts payable
|
- | 149,775 | - | 149,775 | ||||||||||||
|
Notes Payable
|
- | 541,864 | - | 541,864 | ||||||||||||
F-11
|
8.
|
FAIR
VALUE MEASUREMENTS (cont’d)
|
The
following table presents a reconciliation of all assets and liabilities measured
at fair value on a recurring basis as of September 30, 2009:
|
Level
1
|
Level
2
|
Level
3
|
Fair
Value
|
|||||||||||||
|
Cash
|
$ | 2,819 | $ | - | $ | - | $ | 2,819 | ||||||||
|
Accounts
receivable
|
- | - | - | - | ||||||||||||
|
Accounts payable
|
- | 4,962 | - | 4,962 | ||||||||||||
|
Notes Payable
|
- | - | - | - | ||||||||||||
|
9.
|
COMMITMENTS
|
The
Company entered into a lease agreement on July 27, 2010 for office space for one
year beginning August 15, 2010. The commitments for the fiscal year
ending September 30, 2011 total $9,000. Rent expense for the year
ending September 30, 2010 was $4,200.
|
10.
|
SUBSEQUENT
EVENTS
|
In
preparing these financial statements, the Company has evaluated events and
transactions for potential recognition or disclosure through the date the
financial statements were issued.
On
October 28, 2010, UV Flu Technologies, Inc., entered into a binding letter of
intent with The Red Oak Trust in connection with the Company’s proposed
acquisition of one hundred percent (100%) of the issued and outstanding unit of
RxAir Industries, LLC, a Nevada limited liability company (“RxAir”), which is
wholly owned by Red. The purchase price shall be $125,000 and one
million (1,000,000) shares of common stock of the Company. The
proposed acquisition, will include a consulting agreement and issuance of three
hundred thousand shares of common stock of the Company to three key personnel of
RxAir.
F-12
