Attached files
| file | filename |
|---|---|
| 8-K - FORM 8-K - BIOCANCELL THERAPEUTICS INC. | form8k100915.htm |

BioCancell Therapeutics, Inc.
BioCancell Therapeutics Inc.
Targeted Cancer Therapy
Targeted Cancer Therapy
Uri Danon, CEO
September 2010

2
This presentation contains forward-looking statements within the meaning of the Federal Securities Laws
and the Israeli Securities Laws that involves risks and uncertainties . These forward-looking statements,
relate to, without limitation, statements about our market opportunities, our strategy, our competition, the
further development and potential safety and efficacy of our products, our projected revenue and expense
levels and the adequacy of our available cash resources.
and the Israeli Securities Laws that involves risks and uncertainties . These forward-looking statements,
relate to, without limitation, statements about our market opportunities, our strategy, our competition, the
further development and potential safety and efficacy of our products, our projected revenue and expense
levels and the adequacy of our available cash resources.
Drug discovery and development involve a high degree of risk. Factors that might cause material
differences include, among others, risks relating to: the successful preclinical development of our product
candidates; the completion of clinical trials; the successful completion of the process with the FDA,
foreign regulatory bodies and other governmental regulation, including the FDA`s review of any filings we
make in connection with the treatment protocol; uncertainties related to the ability to attract and retain
partners for our technologies and products under development; and other factors described in our public
filings.
differences include, among others, risks relating to: the successful preclinical development of our product
candidates; the completion of clinical trials; the successful completion of the process with the FDA,
foreign regulatory bodies and other governmental regulation, including the FDA`s review of any filings we
make in connection with the treatment protocol; uncertainties related to the ability to attract and retain
partners for our technologies and products under development; and other factors described in our public
filings.
This presentation does not constitute or form part of, and should not be construed as constituting or
forming part of, any offer or invitation to sell or issue, or any solicitation of any offer to purchase or
subscribe for, any shares in the Company, nor shall any part of this presentation nor the fact of its
distribution form part of or be relied on in connection with any contract or investment decision relating
thereto, nor does it constitute a recommendation regarding the securities of the Company.
forming part of, any offer or invitation to sell or issue, or any solicitation of any offer to purchase or
subscribe for, any shares in the Company, nor shall any part of this presentation nor the fact of its
distribution form part of or be relied on in connection with any contract or investment decision relating
thereto, nor does it constitute a recommendation regarding the securities of the Company.
Although we believe that the expectations reflected in these forward-looking statements are based upon
reasonable assumptions, no assurance can be given that such expectations will be attained or that any
deviations will not be material. No reliance may be placed for any purposes whatsoever on the
information contained in this presentation or on its completeness. No representation or warranty, express
or implied, is given by or on behalf of the Company and/or its subsidiaries or any of their directors, officers
or employees or any other person as to the accuracy or completeness of the information or opinions
contained in this presentation and no liability whatsoever is accepted by the Company and/or its
subsidiaries, or any of their members, directors, officers or employees or any other person for any loss
howsoever arising, directly or indirectly, from any use of such information or opinions or other wise arising
in connection therewith.
reasonable assumptions, no assurance can be given that such expectations will be attained or that any
deviations will not be material. No reliance may be placed for any purposes whatsoever on the
information contained in this presentation or on its completeness. No representation or warranty, express
or implied, is given by or on behalf of the Company and/or its subsidiaries or any of their directors, officers
or employees or any other person as to the accuracy or completeness of the information or opinions
contained in this presentation and no liability whatsoever is accepted by the Company and/or its
subsidiaries, or any of their members, directors, officers or employees or any other person for any loss
howsoever arising, directly or indirectly, from any use of such information or opinions or other wise arising
in connection therewith.
Safe Harbor

3
BioCancell Overview
• Developing innovative Targeted Cancer Therapy for the treatment
of cancer, based on Professor Avraham Hochberg’s research
of cancer, based on Professor Avraham Hochberg’s research
• Lead product candidate BC-819 in phase IIb for bladder cancer,
phase I/IIa for pancreatic and ovarian cancer
phase I/IIa for pancreatic and ovarian cancer
• Strong Intellectual Property: 7 patent families
• Incorporated in Delaware (July 2004)
• Listed on TASE (BICL) in Aug. 2006, SEC filing since June 2009
• 16 employees
• Fundraising: $17.8M
• Major Stockholders: Clal Biotechnology Industries Ltd. (18.6%),
Professor Avraham Hochberg (10.4%), Tikcro (3.9%)
Professor Avraham Hochberg (10.4%), Tikcro (3.9%)

4
Technology Platform
• Targeted Cancer Therapy platform based on H19 gene
• H19 is expressed uniquely within cancer cells, while not
expressed in normal cells
expressed in normal cells
• BC-819 drug candidate uses the H19 gene to produce diphtheria
toxin in cancer cells, destroying the cancer cells without affecting
normal cells
toxin in cancer cells, destroying the cancer cells without affecting
normal cells
• Diagnosis of H19 gene is prerequisite for treatment
• It is a targeted treatment without side effects*
• Platform potential: H19 expressed in more than 40 different cancer
indications, in between 70% to 84% of cancer population
indications, in between 70% to 84% of cancer population
*Detected to date

5
After birth, H19 is expressed only in cancer cells, therefore a
significant marker of cancer cells
significant marker of cancer cells
Mechanism of action of H19 is fully deciphered; the expression
of H19 in cancer cells promote tumor development
of H19 in cancer cells promote tumor development
Direct mechanistic connection exists between H19 and p53, a
central protein involved in cancer cell proliferation
central protein involved in cancer cell proliferation
H19 - Oncofetal gene
(Fetus)
6
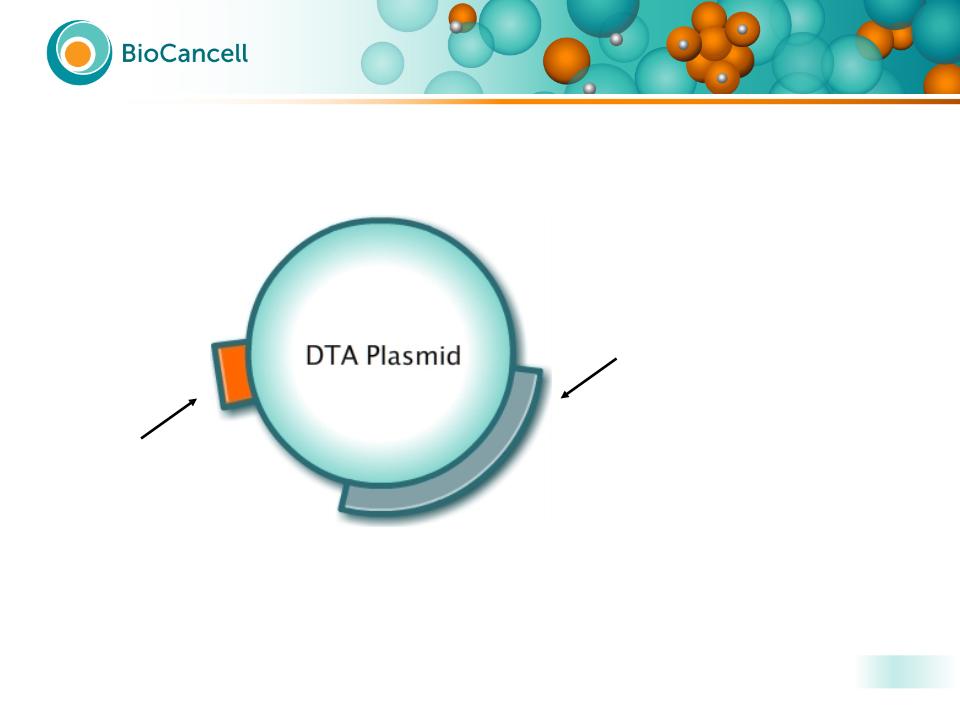
The Drug - BC-819 Plasmid
H19 Promoter
A DNA plasmid containing the H19 gene regulatory
sequences that drive the expression of the Diphtheria
Toxin A gene (DTA-H19)
sequences that drive the expression of the Diphtheria
Toxin A gene (DTA-H19)
Diphtheria Toxin sequence

7
Cancer cell
Normal rapidly-dividing cell
Trigger activated
Trigger not activated
H19’s transcription factors just in cancer
cell nucleus, activate plasmid to produce
diphtheria toxin (DTA)
cell nucleus, activate plasmid to produce
diphtheria toxin (DTA)
No H19 transcription factors for activation
of plasmid to produce diphtheria toxin
(DTA)
of plasmid to produce diphtheria toxin
(DTA)
Cancer cell killed
No change
Mechanism of Action

8
The Advantages of BC-819
1. Excellent safety profile and without side effects* - increases
compliance vs. high patient withdrawal in chemotherapy
compliance vs. high patient withdrawal in chemotherapy
1. Reduces Multi-Drug Resistance (MDR) - a major disadvantage of
chemotherapy
chemotherapy
2. Targeted Cancer Therapy
3. Same drug for multiple cancer types; 40 cancer types expressing
H19 in between 70% to 84% of cancer population
H19 in between 70% to 84% of cancer population
4. Low cost manufacturing vs. other biological products
*Detected to date

9
2010
2012
2013
2011
Ovarian
cancer
cancer
Phase IIb
Phase I/IIa
All indications have FDA
Fast Track potential
Fast Track potential
Bladder
cancer
Phase IIb
FDA Meeting
Phase III
Phase I/IIa (was completed in 2007)
Pancreatic
cancer
cancer
Phase IIb
Phase I/IIa
Phase III
FDA Meeting
FDA Meeting
* Provided clinical success, regulatory approvals, availability of financial resources / strategic collaboration/s
Planned: BC-819+Gemzar
9 patients
11 patients
33 patients,
stage 1 completed

10
Market Size
* Globocan; World Population Prospects and American Cancer Society, estimated 2009 for 7 major
pharmaceutical markets
pharmaceutical markets
** Drug label has FDA’s black box warning
*** Sales for all indications
****H19 expression in cancer patient population: bladder = 84%, ovarian = 75%, pancreatic = 70%
|
|
New
Cases* |
Prevalence
|
Deaths
/Year |
Competitors’ Drugs
- Annual Sales*** |
Adverse events
|
|
Bladder
cancer |
175,000
|
1,500,000
|
37,000
|
BCG** $200M
|
Dysuria, urinary
frequency, hematuria, cystitis, nocturia |
|
Ovarian
cancer |
60,000
|
400,000
|
40,000
|
Doxil** $650M
Taxotere** $3B
Hycamtin** $325M
Gemzar $1.72B
|
Immunosupression
anemia, diarrhea, nausea, hair loss |
|
Pancreatic
cancer |
100,000
|
90,000
|
90,000
|
Gemzar $1.72B
Tarceva $1.66B
|
Immunosupression
anemia, diarrhea, nausea, hair loss |

11
BC-819 - Additional Indications
BC-819 has been successfully tested in animals for the
treatment of lung cancer, liver cancer and kidney cancer
treatment of lung cancer, liver cancer and kidney cancer
Market potential (annual sales) for other drugs in those
indications:
* Source: Data Monitor
• Lung Cancer (NSCLC):
Tarceva (Erlotinib) - $1.66B
• Liver Cancer:
5-Fluorouracil (Adrucil, Efudex, Fluoroplex) - Blockbuster
• Kidney Cancer (RCC):
Sorafenib (Nexavar) - $900M

12
|
2004
|
2005
|
2006
|
2007
|
2008
|
|
$651M
|
$1,571M
|
$2,795M
|
$3,875M
|
$4,914M
|
Herceptin - breast cancer, for “only” 40,000 potential patients
|
2004
|
2005
|
2006
|
2007
|
2008
|
|
$3,188M
|
$3,920M
|
$4,567M
|
$5,206M
|
$5,590M
|
(new approved indications,
contributed to an increase in sales)
contributed to an increase in sales)
Lung (NSCLC) RCC/EMEA HER2- negative
breast cancer
breast cancer
* Source: Data Monitor
Marketed Blockbuster Drugs - Sales
Avastin - originally approved for colorectal cancer

13
ALL
AML
Brain
Breast
CLL
Colorectal
NSCLC
SCLC
Melanoma
NHL
Prostate
Testis
Thyroid
Uterine
Liver
Hodgkin's
Kidney
Bladder
CML
Esophagus
Head&Neck
Myeloma
Ovary
Pancreas
Stomach
0.00
0.10
0.20
0.30
0.40
0.50
0.60
0.70
0.80
0.90
1.00
0
25,000
50,000
75,000
100,000
125,000
150,000
175,000
200,000
225,000
250,000
New Cases - US, 2007
Lethal Cancers: High Unmet Medical Need
BioCancell’s Target Indications
Create High Value

14
|
M&A transaction
|
Sum
|
Most advanced R&D programs in purchase date
|
|
Onyx Pharma acquired
Proteolix (10/2009) |
$851M
|
Carfilzomib, a proteasome inhibitor, in phase IIb
trial for multiple myeloma |
|
Sanofi-Aventis acquired
BiPar Sciences (4/2009) |
$500M
|
Iniparib, a PARP1 inhibitor, in phase II for several
cancer indications |
|
Eisai acquired
Morphotek (3/2007) |
$325M
|
MORAb-003 in phase I/II for ovarian cancer and
MORAb-009 in phase I for pancreatic cancer |
|
Co-development & co-
promotion agreement |
Most advanced R&D
programs in deal date |
Upfront
|
Future
milestones |
Royalties
on sales |
|
OncoGenex (Nasdaq:
OGXI) and Teva (12/2009) |
OGX-011, completed
phase II in prostate and lung cancer |
$60M
|
up to
$370M |
10%-25%
|
|
Clavis Pharma (Oslo:
CLAVIS.OL) and Clovis Oncology (11/2009) |
CP-4126 (11/2009), in
phase II for pancreatic cancer |
$15M
|
up to
$365M |
Tiered
double - digit |
Value creation by M&A / Licensing

15
• Purpose - assess the safety and preliminary efficacy of BC-819
• 18 patients with TCC who failed treatment with BCG
• Successfully concluded in Aug, 2007 (Journal of Urology; Sidi et al.
2008)
2008)
Results:
- No Serious Adverse Events related to BC-819 were detected
- 56% (10/18 patients) did not experience recurrence of tumor
Phase I/IIa in Bladder Cancer

16
Phase IIb in Bladder Cancer
• Purpose - assessing efficacy and safety of BC-819 in 33 patients
• Seven sites in Israel and one in the US (BCG Oncology, Arizona)
• Refractory patients with TCC that failed treatment with BCG or
chemotherapy and whose alternative treatment is bladder
removal
chemotherapy and whose alternative treatment is bladder
removal
• Interim result of first stage of phase IIb (18 patients):
- No Serious Adverse Events related to BC-819 were detected
- 84% responders*
- 56% showed non-recurrence of tumor
- 50% showed complete tumor ablation
- 22% showed a complete response**
* Either non-recurrence, tumor ablation or both
** Both non-recurrence and tumor ablation
The major problem
in refractory bladder
cancer;
cancer;
predicts what to expect
in post-marketing
approval treatment
in post-marketing
approval treatment
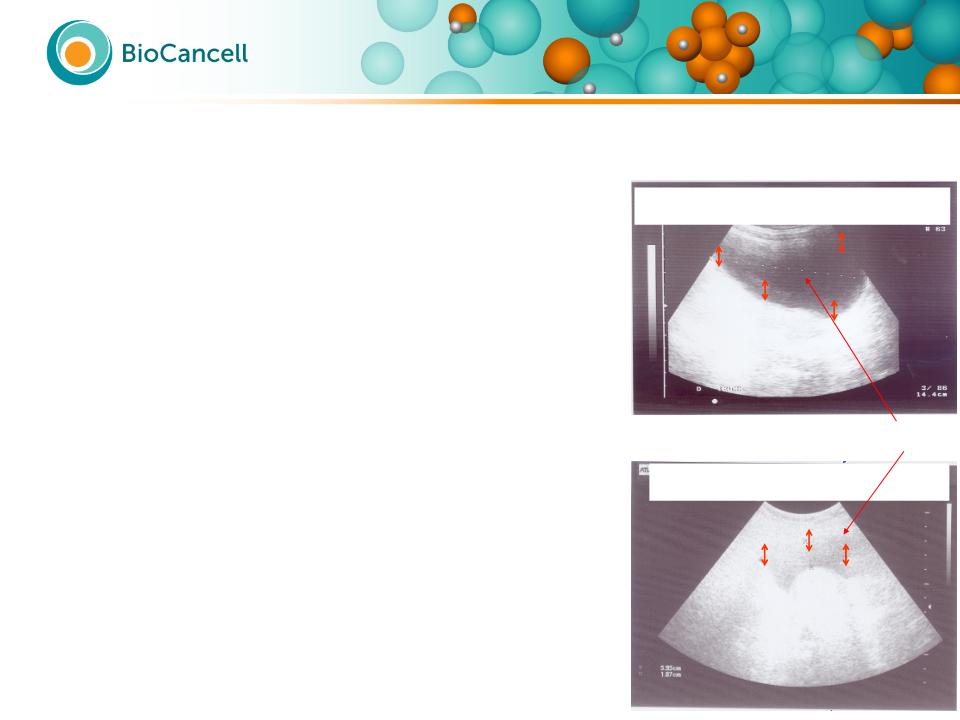
17
31.10.07- before treatments
02.01.08- After treatment #8
(Ascites)
Ovarian Cancer Compassionate Use
Background:
●A 69 year-old female patient, failed all
available treatments, chemotherapy resistance
●IP treated with BC-819 (80mg to 140mg)
After treatment with BC-819:
●Lived 18 months beyond patient’s
original 3 month life-expectancy
●No SAEs were reported
●Tumor growth arrested
●Cancer marker CA-125 dropped by ~50%
●Reduction of number of cancerous cells in
patient’s ascitic fluid
●Ascitic fluid level was significantly reduced
●The patient and her physicians reported
significant clinical improvement

18
Phase I/IIa in Ovarian Cancer
• Purpose - determine safety and the optimal dose for
intraperitoneal delivery of BC-819
intraperitoneal delivery of BC-819
• Phase I/IIa, Dose-Escalation, Safety, Pharmacokinetic and
Preliminary Efficacy Study
Preliminary Efficacy Study
• Conducted in Israel
• 11 patients with ovarian cancer to be treated
• FDA granted ‘Orphan Drug’ status for BC-819 in the US, for
treatment of ovarian cancer
treatment of ovarian cancer

19
Animal Model for Pancreatic Cancer

20
Phase I/IIa in Pancreatic Cancer
• Purpose - determine safety and the optimal dose of BC-819
• Phase I/IIa, Dose-Escalation, Safety, Pharmacokinetic and
Preliminary Efficacy Study
Preliminary Efficacy Study
• Conducted in Israel and in the US at the University of Maryland,
Baltimore
Baltimore
• Total population of 9 patients with unresectable pancreatic
cancer to be treated intratumorally 4 times with BC-819, twice a
week for 2 weeks (8 patients have been treated to date)
cancer to be treated intratumorally 4 times with BC-819, twice a
week for 2 weeks (8 patients have been treated to date)
• Analysis of results is expected shortly
• FDA granted ‘Orphan Drug’ status for BC-819 in the US, for
treatment of pancreatic cancer
treatment of pancreatic cancer

21
The 2nd Generation Drug - BC-821
• Use of both the H19 and IGF2-
P4 genes (double promoter
plasmid) as a treatment
platform for targeted treatment
P4 genes (double promoter
plasmid) as a treatment
platform for targeted treatment
• Status: pre-clinical results in
animals
animals
• The drug covers 100% of the
eligible cancer patients (30%-
50% more than BC-819)
eligible cancer patients (30%-
50% more than BC-819)

22
Patent granted for BC-819, was submitted on Oct. 1997
Extension Strategies:
 Orphan drugs (7 years) - already approved for ovarian and
Orphan drugs (7 years) - already approved for ovarian andpancreatic cancers
 Extension due to drug development process (up to 5 years)
Extension due to drug development process (up to 5 years) Database protection (5-10 years)
Database protection (5-10 years) New US legislation (up to 12 years)
New US legislation (up to 12 years) New patent application for BC-821 was submitted in 2008
Intellectual Property

23
Key Anticipated Events
• Pre clinical results of co-administration of BC-819 and Gemzar
(the intended regime in phase IIb in pancreatic cancer)
(the intended regime in phase IIb in pancreatic cancer)
• Completion of phase I/IIa in pancreatic cancer
• Recruitment of 2nd cohort of patients in phase I/IIa ovarian
cancer
cancer
• 2011:‘Fast-Track’ designation for pancreatic cancer
• 2011: Initiation of phase IIb in pancreatic cancer
• 2011: Completion of phase IIb in bladder cancer
• 2011: Completion of phase I/IIa in ovarian cancer
• Around end of 2011: Interim results of phase IIb in pancreatic
cancer
cancer

24
• Correct target: H19 gene, with fully deciphered mechanism of action,
which has major role in cancer development, expressed in over 40
types of cancer. Recently, mechanistically linked to p53 protein
which has major role in cancer development, expressed in over 40
types of cancer. Recently, mechanistically linked to p53 protein
• Ground-breaking Targeted Cancer Therapy, destroying only cancer
cells, without side effects*
cells, without side effects*
• Interim phase II results show efficacy
• Blockbuster potential
• Experienced management team (with 7 Ph.D.’s)
The Opportunity
*Detected to date

25
Thank You
Please visit us at
www.biocancell.com
www.biocancell.com
