Attached files
| file | filename |
|---|---|
| EX-23.1 - EXHIBIT 23.1 - DIONEX CORP /DE | c05412exv23w1.htm |
| EX-32.1 - EXHIBIT 32.1 - DIONEX CORP /DE | c05412exv32w1.htm |
| EX-32.2 - EXHIBIT 32.2 - DIONEX CORP /DE | c05412exv32w2.htm |
| EX-21.1 - EXHIBIT 21.1 - DIONEX CORP /DE | c05412exv21w1.htm |
| EX-31.2 - EXHIBIT 31.2 - DIONEX CORP /DE | c05412exv31w2.htm |
| EX-31.1 - EXHIBIT 31.1 - DIONEX CORP /DE | c05412exv31w1.htm |
| EX-10.12 - EXHIBIT 10.12 - DIONEX CORP /DE | c05412exv10w12.htm |
Table of Contents
UNITED STATES SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 10-K
ANNUAL REPORT
PURSUANT TO SECTION 13 OR 15(d) OF THE
SECURITIES EXCHANGE ACT OF 1934
PURSUANT TO SECTION 13 OR 15(d) OF THE
SECURITIES EXCHANGE ACT OF 1934
| þ | ANNUAL REPORT PURSUANT TO SECTION 13 or 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the Fiscal Year Ended June 30, 2010
OR
| o | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
Commission File Number 0-11250
DIONEX CORPORATION
(Exact name of
registrant as specified in its charter)
| Delaware (State or other jurisdiction of Incorporation or organization) |
94-2647429 (I.R.S. Employer Identification No.) |
|
| 1228 Titan Way, | ||
| Sunnyvale, California (Address of principal executive offices) |
94085 (Zip Code) |
Registrant’s telephone number, including area code
(408) 737-0700
(408) 737-0700
Securities registered pursuant to Section 12(b) of the Act:
| Title of Each Class | Name of Each Exchange on Which Registered | |
| Common Stock, par value $.001 per share | The Nasdaq Stock Market LLC |
Securities registered pursuant to Section 12(g) of the Act:
None
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of
the Securities Act. YES þ NO o
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or
Section 15(d) of the Act. YES o NO þ
Indicate by check market whether the registrant (1) has filed all reports required to be filed by
Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for
such shorter period that the registrant was required to file such reports), and (2) has been
subject to such filing requirements for the past 90 days. YES þ NO o
Indicate by check mark whether the registrant has submitted electronically and posted on its
corporate Web site, if any, every Interactive Data File required to be submitted and posted
pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period
that the registrant was required to submit and post such files). Yes o No o
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is
not contained herein and will not be contained, to the best of registrant’s knowledge, in
definitive proxy or information statements incorporated by reference in Part III of this Form 10-K
or any amendment to this Form 10-K. þ
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a
non-accelerated filer or a smaller reporting company. See definition of “large accelerated filer”,
“accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer þ | Accelerated filer o | Non-Accelerated filer o | Smaller reporting company o | |||
| (do not check if a smaller reporting company) |
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the
Exchange Act). Yes o No þ
The aggregate market value of the registrant’s Common Stock held by non-affiliates on December 31,
2009 (based upon the closing price of such stock as of such date) was
$1,285,409,824.
As of
August 27, 2010, 17,456,907 shares of the registrant’s Common Stock were outstanding.
Portions of the registrant’s definitive Proxy Statement for the Annual Meeting of Stockholders to
be held on October 26, 2010 are incorporated by reference in Part III of this Annual Report.
TABLE OF CONTENTS
| PART I | ||||||||
| 3 | ||||||||
| 9 | ||||||||
| 13 | ||||||||
| 13 | ||||||||
| 13 | ||||||||
| PART II | ||||||||
| 13 | ||||||||
| 16 | ||||||||
| 17 | ||||||||
| 24 | ||||||||
| 26 | ||||||||
| 50 | ||||||||
| 50 | ||||||||
| 50 | ||||||||
| PART III | ||||||||
| 52 | ||||||||
| 52 | ||||||||
| 52 | ||||||||
| 52 | ||||||||
| 52 | ||||||||
| PART IV | ||||||||
| 52 | ||||||||
| 53 | ||||||||
| 57 | ||||||||
| Exhibit 10.12 | ||||||||
| Exhibit 21.1 | ||||||||
| Exhibit 23.1 | ||||||||
| Exhibit 31.1 | ||||||||
| Exhibit 31.2 | ||||||||
| Exhibit 32.1 | ||||||||
| Exhibit 32.2 | ||||||||
2
Table of Contents
CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS
Certain statements set forth or incorporated by reference in this Form 10-K, as well as in the
Dionex Annual Report to Stockholders for the year ended June 30, 2010, constitute “forward-looking
statements” within the meaning of Section 27A of the Securities Act of 1933, as amended,
Section 21E of the Securities and Exchange Act of 1934, as amended (the “Exchange Act”), and the
Private Securities Litigation Reform Act of 1995, and are made under the safe harbor provisions
thereof. Such statements are subject to certain risks, uncertainties and other factors that may
cause actual results, performance or achievements, or industry conditions, to be materially
different from any future results, performance or achievements, or industry results, expressed or
implied by such forward-looking statements. Such risks and uncertainties include: general economic
conditions, foreign currency fluctuations, the risks associated with international sales and
operations, risks associated with the transition to upgraded accounting information and technology
infrastructure systems, costs associated with potential enhancements to internal controls over
financial reporting, changes in our provision for income taxes or adverse outcomes resulting from
examination of our income tax returns, natural disasters, terrorist attacks or acts of war, credit
risks associated with our customers, fluctuation in worldwide demand for analytical
instrumentation, fluctuations in quarterly operating results, risks associated with maintaining a
significant portion of our cash overseas, competition from other products, existing product
obsolescence, new product development, including market receptiveness, the ability to manufacture
products on an efficient and timely basis and at a reasonable cost and in sufficient volume, the
ability to attract and retain talented employees and other risks as set forth under “Item 1A. “Risk
Factors” and elsewhere in this Form 10-K. Readers are cautioned not to place undue reliance on
these forward-looking statements that reflect management’s analysis only as of the date hereof.
Dionex undertakes no obligation to update these forward-looking statements.
PART I
| Item 1. | BUSINESS |
GENERAL
Dionex Corporation (“Dionex,” “we,” “us,” and “our”) was founded in 1975 to help scientists
automate and apply chemistry solutions for improved laboratory productivity. We design,
manufacture, market, and service a range of liquid chromatography systems, sample preparation
devices, and related products used to separate and quantify the individual components of complex
chemical mixtures relevant to several major industrial, research, and laboratory markets. Our
systems are complementary products that can be integrated together and used along with other
analytical instruments. Dionex Corporation was incorporated as a California corporation in 1980 and
reincorporated in Delaware in 1986.
Our current portfolio of liquid chromatography (LC) systems is focused in two product areas: ion
chromatography (IC) and high performance liquid chromatography (HPLC). In addition, we offer a mass
spectrometer detector that can be coupled with either IC or HPLC systems. For sample preparation,
we provide accelerated solvent extraction (ASE®) systems and AutoTrace®
instruments. In addition, we also develop and manufacture columns, consumables, suppressors,
detectors, automation, and software analysis systems for use in or with liquid chromatography
systems. All these products can be used to analyze chemical substances in the environment and in a
broad range of industrial and scientific applications.
BUSINESS UNITS
Dionex has two business units, Chemical Analysis Business Unit (CABU) and Life Sciences Business
Unit (LSBU), which have similar economic characteristics; product processes; products and services;
types and classes of customers; methods of distribution and regulatory environments. These units,
for which discrete financial information is available, are regularly reviewed and evaluated by the
chief operating decision maker, which we have determined to be our Chief Executive Officer. Because
of these similarities, the two units have been aggregated into one reporting segment for financial
statement purposes. Financial information for our one reportable segment is provided in Note 15 in
the Notes to the Consolidated Financial Statements.
Information concerning revenues and long-lived assets attributable to each of the Company’s
products, services and geographic areas is set forth in Note 15 in the Notes to the Consolidated
Financial Statements, which is included elsewhere in this Annual Report on Form 10-K.
PRODUCTS
Ion Chromatography
Ion chromatography is a form of chromatography that separates ionic (charged) or highly polar
molecules (e.g. sugars and carbohydrates), usually found in water-based solutions, and typically
detects them based on their electrical conductivity. The sale of our IC systems and related
columns, suppressors, detectors, automation, and other products accounted for over 60% of our net
sales in each of fiscal 2010, 2009, and 2008.
3
Table of Contents
Our IC products are used in a wide range of analytical applications, including environmental
monitoring, quality control of pharmaceuticals, corrosion monitoring, and evaluation of raw
materials, quality control of industrial processes and products, research and development, and
regulation of the chemical composition of food, beverage, and cosmetic products. Major customers
include environmental testing laboratories, life science and food companies, chemical/petrochemical
firms, power generation facilities, electronics manufacturers, government agencies, and academic
institutions.
Dionex has been an innovator in ion chromatography for over 30 years. This year, we extended our
history of innovation with the introduction of the first commercially available capillary IC
system, the ICS-5000 Capillary RFICtm System. The heart of this innovation is the IC
Cubetm which houses all of the capillary chemistry in a unique “plug and play” design.
Capillary IC has a powerful value proposition: greatly reduced sample size requirements, increased
sensitivity and reduced running costs.
In addition to the ICS-5000 Capillary RFICtm System, we offer a full range of IC
products to address the analytical needs of our customers. These other system consists of the
following:
| • | Our ICS-2100 RFIC system, introduced in fiscal 2009, provides RFIC eluent generation and regeneration, along with sample-preparation capabilities. The ICS-2100 is an integrated and preconfigured RFIC system designed to perform with all types of electrolytically generated eluents for isocratic and gradient IC separations. The system is controlled from an LCD touch pad front panel or using our Chromeleon Chromatography Data System software (“Chromeleon”). The ICS-2100 occupies minimal space and delivers all the advantages of an RFIC system, including ease-of use, consistency and superior performance compared to systems using manually prepared eluents. |
| • | Our ICS-1600 system, introduced in fiscal 2009, is a fully integrated and preconfigured system with RFIC-ER (Eluent Regeneration) capabilities designed to perform IC separations using suppressed conductivity detection. The system is controlled from an LCD touch pad front panel or using Chromeleon software. |
| • | Our ICS-1100, introduced in fiscal 2009, is an integrated and preconfigured system with RFIC-ER capabilities that performs IC separations using conductivity detection. |
| • | Our ICS-900 integrated IC system, introduced in fiscal 2008, is designed for routine ion analysis and provides easy operation and stable performance. It occupies minimal bench space, making it an attractive solution for small laboratories. |
High Performance Liquid Chromatography
HPLC is a form of chromatography that separates a wide range of small and large molecules, such as
pharmaceuticals, metabolites, and biologics. HPLC typically quantifies the components by measuring
the amount of light that the molecules absorb or emit when exposed to a light source. Sales of our
HPLC products and related columns, suppressors, detectors, automation, and other products accounted
for approximately 30% of our net sales in each of fiscal 2010, 2009, and 2008.
In fiscal 2010, we revamped all of our HPLC systems to Ultra High Pressure LC (UHPLC+) systems.
UHPLC features high pressures, high flow rates and small particles in columns to accelerate
separations. Our UHPLC+ portfolio consists of four major systems:
| • | UltiMate® 3000 Basic Automated LC system is optimized for reliability and ease-of-use with routine LC applications, and also offers UHPLC application compatibility up to 620 bar. The ACC-3000 Autosampler Column Compartment is at the heart of the system. Its unique instrument design combines a rugged sample injection principle with a powerful column oven. |
| • | UltiMate® 3000 Standard Analytical systems offer full support of all HPLC applications and provide UHPLC application compatibility up to 620 bar, allowing customers to move to UHPLC whenever necessary. |
| • | UltiMate® 3000 Rapid Separation LC (“RSLC”) system provides higher performance and flexibility in UHPLC applications up to 1,000 bar. Precision-engineered instrumentation, advanced data processing, and highly optimized chemistries enable the most challenging chromatographic applications. This system offers industry leading versatility covering the maximum range of UHPLC applications, while keeping fully compatible to conventional LC. |
| • | UltiMate® 3000 RSLCnano system was developed for maximum performance in the field of proteomics and other complex life science applications. The robust, continuous direct-flow delivery is designed for interruption-free analysis providing the highest resolution of sample components. This feature, in addition to the widest flow-pressure footprint enables the application of UHPLC to the nano scale, allowing scientists to tune for the highest resolution or the fastest analysis time, even for trypsin-digested peptide patterns of utmost complexity. |
4
Table of Contents
Our acquisition from ESA Biosciences, Inc. of its Life Sciences Tools business in fiscal 2010
increased our portfolio of detection strategies for HPLC. The Corona® ultra™
Charged Aerosol Detector for UHPLC systems is a universal detector, allowing determination of
analytes independent of charge or chromophore. The Coulochem® and CoulArray®
electrochemical detectors are targeted for neuroscience, clinical, and metabolomics research.
Together with our new fluorescence detector, the FLD-3000, these detection solutions have greatly
improved our ability to serve our pharmaceutical, biopharmaceutical, and life sciences customers.
This acquisition further expands our presence in life sciences and opens some new markets in
clinical research and neurosciences.
Other Dionex product offerings
Sample Preparation - In fiscal 2008, we introduced the Accelerated Solvent Extraction (“ASE”) 150
and ASE 350 systems which extract components of interest from solid and semisolid samples using
common solvents at elevated temperatures and pressures. The ASE 150 and 350 systems offer several
advantages over other solvent-based extraction techniques including lower solvent consumption,
reduced extraction time, higher throughput, automation and ease of use. Both of these systems
contain pH-hardened flow pathways featuring Dionium™ components allowing the extraction
of samples that have been pretreated with strong acids or bases. The ASE 350 also features
built-in solvent mixing capability that allows sampling from three different bottles to allow the
customer to perform extractions using a wide variety of solvents. Along with these ASE systems, we
added ASE Prep CR NA+ and ASE Prep CR H+ adsorbents which neutralize strong
acids or bases so samples can be extracted with organic or aqueous solvents to remove analytes of
interest as part of the ASE process.
In fiscal 2009, we acquired our AutoTrace® line of products from Caliper Life Sciences.
The AutoTrace SPE workstation automates the extraction of large volume aqueous samples prior to
analysis. In fiscal 2010, we introduced the new AutoTrace 280 SPE instrument which automates SPE
processes: conditioning, loading, rinsing and elution. AutoTrace 280 can be used for a wide
variety of SPE testing methods including those approved by the Environmental Protection Agency
(EPA) for testing of ground water, surface water, waste water and drinking water and other organic
pollutants such as endocrine disrupters and personal care product ingredients in aqueous samples.
AutoTrace 280 complements our ASE, IC and HPLC product lines, giving us the opportunity to expand
our sample preparation market.
Mass Spectrometry - Mass spectrometry (MS) is used to identify the molecular weight of compounds
within a sample substance and renders structural molecular information. Through an agreement with
Thermo Fisher Scientific, we offer the MSQ Plus™ mass spectrometer which can be coupled
with certain IC and HPLC products. LC/MS and IC/MS systems using the MSQ Plus are used worldwide,
particularly in the pharmaceutical market, but also for environmental testing, drug, food and
beverage quality control, and many other applications.
In fiscal 2006, we introduced the Dionex Chromatography Mass Spectrometry Link
(DCMSLink™) software which provides an interface for controlling a wide range of our
chromatography instruments from third-party mass spectrometry software (MS software) such as
Analyst® from Applied Biosystems/MDS Sciex (now owned by Danaher Corporation),
Xcalibur® from Thermo Fisher Scientific Inc., and HyStar™ from Bruker
Corporation. In fiscal 2009, we introduced version 2.7 of DCMSLink which delivered many
new features, including a unique graphical control interface for Dionex instruments, providing
real-time modification of instrument parameters. The new version also supports digital acquisition
of 3-D data from the UltiMate 3000 photodiode array detector, for assessing peak purities, spectral
matching and peak tracking. DCMSLink enables us to expand to additional markets for our
chromatography systems to customers who desire the quality and features of our chromatography with
higher-end mass spectrometers.
Process Instrumentation - In fiscal 2009, we introduced the Integral™ Process Analyzer
for continuous on-line monitoring necessary in a variety of industrial applications. The Integral
systems combine the Dionex ICS and UltiMate systems with advanced sample handling and preparation
modules and industrial enclosures to automate a wide variety of in-process analyses. Major
applications for Integral are in the power generation industry for the continuous monitoring of
corrosive contaminants in boiler water, the semiconductor industry for continuous monitoring of
contaminants in high purity water, and the pharmaceutical and chemical industries for continuous
monitoring of biological and chemical synthesis processes. Integral uses our Chromeleon PA software
for automation, data acquisition, reporting, and security. The software allows the user to view
analyzer status, handle alarms, and interface with the computing and control systems in place at
the enterprise where Integral is installed.
Chromatography Data Management Software Systems (CDS) - We offer a family of chromatography
data management software products that automate sample handling, system operation, and data
analysis for chromatography systems. Our industry-leading CDS package called Chromeleon can control
IC and HPLC systems, either manufactured by Dionex or other vendors. Our advanced CDS provides
analysis and automation support for applications such as discovery processes, quality control, and
complex research activities such as proteomics and biomarker discovery. Chromeleon software is
offered in our Chromeleon 6.8 version to customers desiring to control several IC or HPLC systems
at once. Chromeleon 6.8 can control over 250 LC and gas chromatography (GC) instruments and modules
produced by more than 30 vendors. In fiscal 2009, we introduced Chromeleon® 7
Chromatography Data System, a next-generation chromatography data system that provides the fastest,
easiest path from samples to results, boosting productivity and improving user experience.
Chromeleon 7 includes capabilities to speed up data evaluation by providing an even more intuitive
way to access all information for an experiment and includes new methods to identify peaks and save
time in analysis.
5
Table of Contents
Consumables - We offer a wide variety of consumables for our IC, HPLC and sample
preparation customers. Our primary consumable items are columns, suppressors, and RFIC eluent
generation cartridges. These consumables are replaced at regular intervals depending on the volume
of use and sample composition.
Columns - A chromatography column consists of a hollow cylinder packed with a unique material
allowing for separation of the various chemical components contained in a sample. We develop and
manufacture separation materials such as ion-exchange resins, silica-based bonded phases, and
monolithic phases using proprietary processes that are designed and tested for specific
applications in the HPLC and IC markets. We offer polymer-based ion-exchange and silica-based
reversed-phase columns supporting capillary, analytical, preparative, and semi-preparative scale
applications. Our most recent introductions for column technologies focus on Fast IC columns
designed for faster analysis of standard anions and sugars as well as UHPLC columns for faster HPLC
separations used for a broad range of customer applications. We also offer a number of columns in
monolith formats for increased resolution, speed and capacity for IC and HPLC application including
applications for protein separations. In addition, we offer a variety of micro, capillary and nano
LC columns in varying formats packed with a variety of chromatographic materials.
Eluent Suppressors - We manufacture the SRS 300 family of Self-Regenerating Suppressors to enhance
detection and sensitivity in ion chromatography. Our offering includes an array of suppressors that
include the SRS 300, the Atlas® Electrolytic Suppressor, and the
MicroMembrane™ Suppressor (MMS III).
RFIC
Eluent Generation Cartridges - We manufacture eluent generation cartridges used with RFIC
systems for the automatic production of high-purity eluents. We offer cartridges for generation of
hydroxide, methanesulfonic acid, and carbonate/bicarbonate eluents designed to cover the range of
ions and polar compounds that our customers typically measure.
IonSwifttm line - Introduced in 2010, these are new columns for IC based on our patented
monolith technology and feature fast mass transfer and improved resolution even at elevated flow
rates with low backpressure. This faster analysis and high resolution are key benefits of this new
monolith column line. This chemistry can be coupled with the new ICS-5000, providing customers the
experience of Fast IC in capillary, microbore or standard bore format.
Fast IonPac line - We also introduced in 2010 new resin based fast IC columns for analytical and
capillary flow rates based on our traditional packed-particle IonPac line. These anion and cation
columns allow increased flow rates, therefore reducing typical run times to less than five minutes.
Other services - We provide services and service products through our customer and field service
organization. These services include maintenance contracts, spare part sales, customer training and
sales of other products and valued-added services (refer to the “Technical Support, Installation
and Service” section below).
CUSTOMERS, MARKETING, AND SALES
Our products are used extensively in the environmental, life science, and industrial markets that
typically utilize chromatography and extraction technologies. The environmental market is
characterized by water analysis, safety and security applications, and pollution testing, with
chemists from private and governmental laboratories being our primary customers in this field. The
life science markets we serve include the pharmaceutical and biopharmaceutical segment,
biosciences, and medical sciences, with customers from industrial, academic, and government
accounts. Industrial markets include the electronics and power industries, with a demand for
analyzing the high-purity water quality in their production facilities. We also serve many of the
world’s largest industrial companies within the chemical industry, which produce specialty
chemicals/petrochemicals, and consumer products, and increasingly within the food and beverage
market, which test for product quality assurance, purity, and contaminants.
One of our key marketing strategies is to target all market segments mentioned above to increase
demand for our chromatography solutions. This is accomplished by approaching all existing and
potential customers through direct marketing and sales activities including direct sales calls,
mailings, advertising, electronic marketing, seminars, and workshops. In addition, we build
visibility and branding for our global presence through scientific conferences and exhibitions.
Continuous growth in all these markets results from identifying new customers in existing sales
regions, extending geographic penetration, and increasing demand for our products and technical
support capabilities. In addition, we grow through the constant introduction of new technologies
and products which stimulates our current customers to adopt our more advanced offerings.
6
Table of Contents
The second component of our marketing strategy is to explore and develop new applications through
collaborations with existing and potential customers, and to leverage these new methods in other
market areas. A prerequisite to establishing this process is the availability of highly skilled
technical and support staff to work with customers to define and develop these novel applications.
In order to meet and exceed customer expectations in our commercial markets, our goal is to
optimize and diversify our technology expertise in the LC market, including sample preparation,
separation, analysis, testing, and data management.
Geographically, we market and distribute our products and services through our sales force in
Austria, Australia, Belgium, Brazil, Canada, China, Denmark, France, Germany, India, Ireland,
Italy, Japan, Korea, the Netherlands, New Zealand, Singapore, Sweden, Switzerland, Taiwan, the
United Kingdom, and the United States (U.S.). In each of these countries other than Canada, we
maintain one or more local sales offices in order to support and service our customers in the
regions. In other international locations where we do not have a direct sales force, we have
developed a network of distributors and sales agents. In fiscal 2010, our net sales by geographic
region were approximately 27% in North America, 38% in Europe and 35% in Asia/Pacific and other. In
fiscal 2009, our net sales by geographic region were approximately 26% in North America, 41% in
Europe and 33% in Asia/Pacific and other. In fiscal 2008, our net sales by geographic region were
approximately 28% in North America, 43% in Europe and 29% in Asia/Pacific and other.
We manufacture our products based upon our forecast of customer demand and maintain adequate
inventories of completed modules or finished goods in advance of receipt of firm orders. System or
instrument orders are generally placed by the customer on an as-needed basis, and instruments are
usually shipped within four to six weeks after receipt of an order. We do not maintain a
substantial backlog, and backlog as of any particular date may not be indicative of our actual
sales in any succeeding period. The level of backlog as of June 30, 2010 was $68 million and as of
June 30, 2009 was $55 million.
FINANCIAL INFORMATION ABOUT GEOGRAPHIC AREAS
For financial information by geographic area, refer to Note 15 of the Notes to Consolidated
Financial Statements in Item 8 of this Form 10-K.
COMPETITION
Competition in our industry is based upon the performance capabilities of the analytical
instruments, technical support and after-market services, the manufacturer’s reputation as a
technological leader and selling prices. We believe that performance capabilities are the most
important of these criteria. Customers measure system performance based on sensitivity (the ability
to discern minute quantities of a particular sample component), selectivity (the ability to
distinguish between similar components), speed and throughput of analysis, and the range of
chemical and biological samples the system can effectively analyze. We believe that we enjoy a
favorable reputation in terms of performance capabilities.
Companies competing with us in LC portion of the analytical instruments market include Agilent
Technologies, Inc., Shimadzu Corporation, Thermo Fisher Scientific Inc., Waters Corporation and
Metrohm Ltd.
We believe we have a substantial market share in the IC market, which is a segment of the broader
LC market. Our IC systems generally compete with a number of analytical techniques used in
identifying and quantifying ionic and polar compounds. The primary source of competition are
conventional manual and automated wet chemistry procedures and certain modified liquid
chromatography systems that are used with or without an ion suppression device. Companies competing
with us in the IC market include Metrohm AG, Shimadzu Corporation, Waters Corporation and other
smaller companies.
We believe we have a smaller but growing market share in the combined ultra-high pressure,
analytical and capillary/nano-LC HPLC portions of the LC market. Our UltiMate 3000 UHPLC+ systems
compete directly with other manufacturers in ultra high pressure, standard and capillary-/nano-
HPLC applications. We believe that the UltiMate UHPLC+ systems have certain benefits over competing
systems, including advanced pump and dual pump technology, thermostatted temperature control, and
high performance auto sampling capabilities. The UltiMate 3000 offers benefits over competing
systems, including the ability to analyze minute contents of sample at very low flow rates or at
higher pressure over a wider range of flow rates. In addition, we are unique in that our entire
HPLC product line is compatible with ultra high pressure methods giving the customer flexibility in
transferring methods from standard to UHPLC methods. We also believe that our Chromeleon software
package is considered the most advanced chromatography data management system in the market which
provides us a competitive advantage over other vendor’s software offerings. Our competitors in the
HPLC market include such vendors as Agilent Technologies, Inc., Shimadzu Corporation, Thermo Fisher
Scientific, Waters Corporation and various smaller competitors.
7
Table of Contents
Our Accelerated Solvent Extraction systems compete directly with standard Soxhlet, sonication,
supercritical fluid extraction and microwave extraction techniques provided by other companies. We
believe that our ASE systems have certain benefits compared to competing techniques, including
faster extraction time, reduced solvent usage, built-in automation and ease of use.
PATENTS AND LICENSES
We have a patent portfolio covering certain technologies employed in our products. These
technologies include but are not limited to those used in our instruments, suppressors, eluent
generators, columns, sample preparation devices, pumps, autosamplers, and detectors. The portfolio
includes both apparatus and method of use patents. Our patents are presently issued in the U.S. and
number of foreign countries including those in North America, Europe and Asia. As a matter of
company policy, we vigorously protect our intellectual property rights and seek patent coverage on
developments that we regard as strategic, material and patentable. Our patents, including those
licensed from others, expire between 2010 and 2029. We believe that, while our patent portfolio has
value, no single patent or patent application is in itself essential and that the invalidity or
expiration of any single patent would not have a material adverse effect on our business.
We regard our Chromeleon software as proprietary and we rely on a combination of patents,
copyrights, trademarks, trade secret laws and other proprietary rights, laws, license agreements
and other restrictions on disclosure, copying and transferring title to protect our rights to our
software products. We have one issued patent and a limited number of patent applications covering
our software, and existing copyright laws afford only limited protection. In addition, the laws of
some foreign countries do not protect our proprietary rights to the same extent as do the laws of
the U.S.
INTERNATIONAL OPERATIONS
Financial information about foreign and domestic operations and export sales is provided in Note 15
of the Notes to Consolidated Financial Statements found elsewhere in this report.
We have subsidiaries in Austria, Australia, Brazil, Canada, China, Denmark, France, Germany, India,
Ireland, Italy, Japan, Korea, the Netherlands, New Zealand, Singapore, Sweden, Switzerland, Taiwan,
the United Kingdom and the U.S. Our foreign sales are affected by fluctuations in currency exchange
rates and by regulation adopted by foreign governments. Such fluctuations have materially affected,
both positively and negatively, our results of operation in past periods and will likely materially
affect our results of operations in the future. Export sales are subject to certain controls and
restrictions, but we have not experienced any material difficulties related to these limitations.
MANUFACTURING AND SUPPLIERS
We produce most of our consumables, including IC and HPLC columns and IC suppressors, and assemble
our IC systems and modules in our California manufacturing facilities. We assemble the systems and
modules for our UltiMate 3000 systems in our manufacturing facility in Germany. We assemble our
charged aerosol (CAD) and electrochemical HPLC detectors in our Massachusetts manufacturing
location. We have developed proprietary processes for the manufacture of polystyrene-based resins
and for packing columns with these resins. We believe that our resins, columns and suppressor
manufacturing know-how are critical to the performance and reliability of our chromatography
systems. We require each employee and consultant to sign a nondisclosure agreement to protect our
proprietary processes. However, there can be no assurances that these agreements will provide
meaningful protection or adequate remedies for our proprietary processes in the event of
unauthorized use or disclosure.
We have emphasized a modular design for the principal subsystems of our pumping and flow systems,
sample injection systems, chromatography modules, detectors, and control and data analysis systems.
We believe that this modular approach has enabled us to meet the wide range of system
configurations required by our customers. Manufacturing has transitioned into flow-line production
for our major systems while maintaining subassembly cell production for our integrated modules.
These practices have enhanced our ability to effectively manage our inventory levels.
We manufacture many subassemblies used in our products. Components, including formed-plastic and
sheet-metal packaging materials, machine-metal parts, integrated circuits, microprocessors,
microcomputers and certain detector and data analysis modules, are purchased from other
manufacturers. Most of the raw materials, components and supplies purchased by us are available
from a number of different suppliers, although a number of items are purchased from limited or
single source of supply.
8
Table of Contents
TECHNICAL SUPPORT, INSTALLATION AND SERVICE
Users of our chromatography systems may require technical support before and after a system sale.
Services provided before the sale are recorded in operating expenses as incurred. Chromatography
systems sold by us generally include a one-year warranty. These costs are accrued for at the time
of the system sale. Installation and certain basic user training are provided to the customer, with
revenues for these services recognized at the time the services are provided. Maintenance contracts
may be purchased by customers to cover equipment no longer under warranty. Maintenance work not
performed under warranty or maintenance contracts is performed on a time and materials basis. We
offer training courses and periodically send our customers information on applications development.
We install and service our products through our own field service organizations in Austria,
Australia, Brazil, Belgium, Canada, China, Denmark, France, Germany, India, Italy, Ireland, Japan,
Korea, the Netherlands, New Zealand, Singapore, Sweden, Switzerland, Taiwan, the United Kingdom and
the U.S. Installation and service in other foreign countries are typically provided by our
distributors or agents.
RESEARCH AND PRODUCT DEVELOPMENT
Our research and product development efforts are focused on increasing the performance of our
chromatography and other products and expanding the number of chemical and biological compounds
that can be analyzed efficiently with our products. Research and product development expenditures
were $31.6 million, $29.0 million, and $28.9 million in fiscal 2010, 2009, and 2008, respectively.
We pursue active development programs in the areas of system hardware, applications, CDS,
suppressors, and resin and column technologies. There can be no assurances that our product
development efforts will be successful or that the products developed will be accepted by the
marketplace.
EMPLOYEES
We had approximately 1,550 employees as of June 30, 2010 and 1,400 employees as of June 30, 2009.
AVAILABLE INFORMATION
We maintain a website at www.dionex.com; however, information found on our website is not
incorporated by reference into this report. We make available free of charge on or through our
website our proxy statement, annual report on Form 10-K, quarterly reports on Form 10-Q, and
amendments to those reports, filed or furnished pursuant to Section 13(a) or 15(d) of the Exchange
Act as soon as reasonably practicable after we electronically file such material with, or furnish
it to, the Securities and Exchange Commission (the “SEC”). Our Board of Directors has adopted
charters for the Audit, Compensation, and Nominating and Corporate Governance Committees of our
Board of Directors and a Code of Business Ethics and Values applicable to all of our officers and
employees. These charters and our Code of Business Ethics and Values are available on our website
at http://investor.dionex.com/governance-PDFs.cfm and a printed copy of this information is
available without charge by sending a written request to: Investor Relations, Dionex Corporation,
1228 Titan Way, Sunnyvale, California 94085. The public may read and copy any materials we file
with the SEC at the SEC’s Public Reference Room at 100 F Street, N.E., Room 1580, Washington, DC
20549, and may obtain information on the operation of the Public Reference Room by calling the SEC
at 1-800-SEC-0330. The SEC maintains an Internet site that contains reports, proxy and information
statements, and other information regarding issuers that file electronically with the SEC at
http://www.sec.gov.
| Item 1A. | RISK FACTORS. |
You should consider carefully the following risk factors as well as other information in this
report before investing in any of our securities. If any of the following risks actually occur, our
business operating results and financial condition could be adversely affected. This could cause
the market price for our common stock to decline, and you may lose all or part of your investment.
These risk factors include any material changes to, and supersede, the risk factors previously
disclosed in our most recent annual report on Form 10-K and subsequently filed quarterly reports on
Form 10-Q.
A downturn in economic conditions could affect our operating results.
Our business, financial condition and results of operations have been affected by the weaker global
economic conditions of the last several years. These conditions resulted in reduced sales of our
products in the last two quarters of fiscal 2009 and the first two quarters of fiscal 2010. In a
continued economic recession or under other adverse economic conditions, our customers may be less
likely to purchase our products and vendors may be more likely to fail to meet contractual terms. A
further downturn in economic conditions or a slow recovery from the current recession may make it
more difficult for us to maintain and continue our revenue growth and profitability performance
resulting in a material adverse effect on our business.
9
Table of Contents
Foreign currency fluctuations related to international operations may adversely affect our
operating results.
We derived over 70% of our net sales from outside the U.S. in fiscal 2010 and expect to continue to
derive the majority of net sales from outside the U.S. for the foreseeable future. Most of our
sales outside the U.S. are denominated in the local currency of our customers. As a result, the
U.S. dollar value of our net sales varies with currency rate fluctuations. Significant changes in
the value of the U.S. dollar relative to certain foreign currencies could have a material adverse
effect on our results of operations. In recent periods, our results of operations have been
positively affected from the depreciation of the U.S. dollar against the Euro, the Japanese yen and
several other foreign currencies, but there can be no assurance that this positive impact will
continue. In the past, our results of operations have also been negatively impacted by the
appreciation of the U.S. dollar against other currencies.
Economic, political and other risks associated with international sales and operations could
adversely affect our results of operations.
Because we sell our products worldwide and have significant operations outside of the U.S., our
business is subject to risks associated with doing business internationally. We anticipate that
revenue from international operations will continue to represent a majority of our total net sales.
In addition, we expect that the proportion of our employees, contract manufacturers, suppliers, job
functions and manufacturing facilities located outside the U.S. will increase. Accordingly, our
future results could be harmed by a variety of factors, including:
| • | interruption to transportation flows for delivery of parts to us and finished goods to our customers; |
| • | changes in a specific country’s or region’s economic, political or other conditions; |
| • | trade protection measures and import or export licensing requirements; |
| • | difficulty in staffing and managing widespread operations; |
| • | differing labor regulations; |
| • | differing protection of intellectual property; |
| • | unexpected changes in regulatory requirements; and |
| • | geopolitical turmoil, including terrorism and war. |
If we fail to effectively transition to our new and/or upgraded accounting information and
technology infrastructure systems, it may have an adverse impact on our business and results of
operations.
We may experience difficulties in transitioning to new or upgraded accounting information and
technology infrastructure systems, including loss of data and decreases in productivity as
personnel become familiar with new, upgraded or modified systems. Our accounting information and
technology infrastructure systems will require modification and refinement as we grow and as our
business and customers’ needs change, which could prolong the difficulties we experience with
systems transitions, and we may not always employ the most effective information systems. If we
experience difficulties in implementing new or upgraded accounting information and technology
infrastructure systems or experience significant system failures, or if we are unable to
successfully modify our accounting information and technology infrastructure systems and respond to
changes in our customers’ needs in a timely manner, it may have an adverse impact on our business
and results of operations.
We continually evaluate our system of internal controls over financial reporting and may make
enhancements where appropriate, which may require significant resources.
Effective internal controls are necessary for us to provide reliable financial reports and
effectively prevent fraud. We continually evaluate and, where appropriate, enhance our policies,
procedures and internal controls. If we fail to maintain the adequacy of our internal controls, as
such standards are modified, supplemented or amended from time to time, we could be subject to
regulatory scrutiny and investors could lose confidence in the accuracy and completeness of our
financial reports. In addition, failure to maintain adequate internal controls could result in
financial statements that do not accurately reflect our financial condition. Implementing
changes when necessary may take a significant amount of time, money, and management resources and
may require specific compliance training of our directors, officers and other personnel.
10
Table of Contents
Changes in our provision for income taxes or adverse outcomes resulting from examination of our
income tax returns could adversely affect our results.
Our provision for income taxes is subject to volatility and could be adversely affected by
earnings being lower than anticipated in countries that have lower tax rates and higher than
anticipated in countries that have higher tax rates; by changes in the valuation of our deferred
tax assets and liabilities; by expiration of or lapses in the R&D tax credit laws; by transfer
pricing adjustments; by tax effects of nondeductible compensation; by changes in accounting
principles; or by changes in tax laws and regulations including possible U.S. or foreign changes to
the taxation of earnings of our foreign subsidiaries, and the deductibility of expenses
attributable to foreign income, or the foreign tax credit rules. Significant judgment is required
to determine the recognition and measurement attribute prescribed in Financial Accounting Standards
Board (FASB) Accounting Standards Codification 740, Income Taxes (“ASC 740”) (formerly referenced
as FASB Interpretation No. 48, “Accounting for Uncertainty in Income Taxes —an interpretation of
FASB Statement No. 109”). ASC 740 applies to all income tax positions, including the potential
recovery of previously paid taxes, which if settled unfavorably could adversely impact our
provision for income taxes or additional paid-in capital. In addition, we are subject to the
continuous examination of our income tax returns by the Internal Revenue Service and other tax
authorities. We regularly assess the likelihood of adverse outcomes resulting from these
examinations to determine the adequacy of our provision for income taxes. There can be no assurance
that the outcomes from these continuous examinations will not have an adverse effect on our
operating results and financial condition.
Natural disasters, terrorist attacks or acts of war may cause damage or disruption to us and our
employees, facilities, information systems, security systems, vendors and customers, which could
significantly impact our net sales, costs and expenses, and financial condition.
We have significant manufacturing and distribution facilities, particularly in the western U.S.
(i.e. California) and in Germany. In particular, California has experienced a number of
earthquakes, wildfires, flooding, landslides and other natural disasters in recent years.
Occurrences of these types of events could damage or destroy our facilities which may result in
interruptions to our business and losses that exceed our insurance coverage. Terrorist attacks
have contributed to economic instability in the U.S. (such as those that occurred on September 11,
2001), and further acts of terrorism, bioterrorism, violence or war could affect the markets in
which we operate, our business operations, our expectations and other forward-looking statements
contained or incorporated in this document. Any of these events could have an adverse effect on
our operating results and financial condition.
Credit risks associated with our customers may adversely affect our financial position or result of
operations.
Because trade credit is extended to many of our customers, the current global economic condition
may adversely affect our ability to collect on accounts receivable that are owed to us. In general,
our customers are evaluated for their credit worthiness as part of our operating policy, and
letters of credit are utilized to mitigate credit risks when possible. We believe we have adopted
the appropriate operating policies to address the customer credit risk under a stable economic
environment. Nevertheless, given the current global economic situation we could experience delays
in collection on accounts receivable that are owed to us. As a result, this could adversely affect
our financial position or result of operations.
Fluctuations in worldwide demand for analytical instrumentation could affect our operating results.
The demand for analytical instrumentation products can fluctuate depending upon capital expenditure
cycles. Most companies consider our instrumentation products capital equipment and some customers
may be unable to secure the necessary capital expenditure approvals due to general economic or
customer specific conditions. Significant fluctuations in demand could harm our results of
operations.
Fluctuations in our quarterly operating results may cause our stock price to decline.
A high proportion of our costs are fixed due in part to our significant sales and marketing,
research and product development and manufacturing costs. Declines in revenue caused by
fluctuations in currency rates, worldwide demand for analytical instrumentation or other factors
could disproportionately affect our quarterly operating results, which may in turn cause our stock
price to decline.
A significant portion of our cash is maintained overseas.
Most of our short-term debt is in the U.S. While there is a substantial cash requirement in the
U.S. to fund operations and capital expenditures, service debt obligations, finance potential
acquisitions and continue authorized stock repurchases, a significant portion of our cash is
maintained and generated from foreign operations. Our financial condition and results of operations
could be adversely impacted if we are unable to maintain a sufficient level of cash flow in the
U.S. to address these requirements through cash from U.S. operations, efficient and timely
repatriation of cash from overseas and other sources obtained at an acceptable cost.
11
Table of Contents
Our results of operations and financial condition will suffer if we do not introduce new
products that are attractive to our customers on a timely basis.
Our products are highly technical in nature. As a result, many of our products must be developed
months or even years in advance of the potential need by a customer. If we fail to introduce new
products and enhancements as demand arises or in advance of the competition, our products are
likely to become obsolete over time, which would harm operating results. Also, if the market is not
receptive to our newly-developed products, our results of operations would be adversely impacted
and we may be unable to recover the costs of research and product development and marketing
associated with such products.
The analytical instrumentation market is highly competitive, and our inability to compete
effectively in this market would adversely affect our results of operations and financial
condition.
The analytical instrumentation market is highly competitive and we compete with many companies on a
local and international level that are significantly larger than we are and have greater resources,
including larger sales forces and technical staff. Competitors may introduce more effective and
less costly products and, in doing so, may make it difficult for us to acquire and retain
customers. If this occurs, our market share may decline and operating results could suffer.
We may experience difficulties with obtaining components from sole- or limited-source suppliers, or
manufacturing delays, either of which could adversely affect our results of operations.
Most raw materials, components and supplies that we purchase are available from many suppliers.
However, certain items are purchased from sole or limited-source suppliers and a disruption of
these sources could adversely affect our ability to ship products as needed. A prolonged inability
to obtain certain materials or components would likely reduce product inventory, hinder sales and
harm our reputation with customers. Worldwide demand for certain components may cause the cost of
such components to rise or limit the availability of these components, which could have an adverse
effect on our results of operations.
We manufacture products in our facilities in Germany, the Netherlands and the U.S. Any prolonged
disruption to the operations at these facilities, whether due to labor unrest, supplier issues,
damage to the physical plants or equipment or other reasons, could also adversely affect our
results of operations.
Our executive officers and other key employees are critical to our business, they may not remain
with us in the future and finding talented replacements may be difficult.
Our operations require managerial and technical expertise. Each of our executive officers and key
employees is employed “at will” and may leave our employment at any time. In addition, we operate
in a variety of locations around the world where the demand for qualified personnel may be
extremely high and is likely to remain so for the foreseeable future. As a result, competition for
personnel can be intense and the turnover rate for qualified personnel may be high. The loss of any
of our executive officers or key employees could cause us to incur increased operating expenses and
divert senior management resources in searching for replacements. An inability to hire, train and
retain sufficient numbers of qualified employees would seriously affect our ability to conduct our
business.
We may be unable to protect our intellectual property rights and may face intellectual property
infringement claims.
Our success will depend, in part, on our ability to obtain patents, maintain trade secret
protection and operate without infringing the proprietary rights of third parties. We cannot be
certain that:
| • | any of our pending patent applications or any future patent applications will result in issued patents; |
| • | the scope of our patent protection will exclude competitors or provide competitive advantages to us; |
| • | any of our patents will be held valid if subsequently challenged; or |
| • | others will not claim rights in or ownership of the patents and other proprietary rights held by us. |
12
Table of Contents
Furthermore, we cannot be certain that others have not or will not develop similar products,
duplicate any of our products or design around any patents issued, or that may be issued, in the
future to us or to our licensors. Whether or not patents are issued to us or to our licensors,
others may hold or receive patents which contain claims having a scope that covers products
developed by us. We could incur substantial costs in defending any patent infringement suits,
whether or not such suits have merit, or in asserting any patent rights, including those granted by
third parties. In addition, we may be required to obtain licenses to patents or proprietary rights
from third parties. There can be no assurance that such licenses will be available on acceptance
terms, if at all.
Our issued U.S. patents, and corresponding foreign patents, expire at various dates ranging from
2010 to 2029. When each of our patents expires, competitors may develop and sell products based on
the same or similar technologies as those covered by the expired patent. We have invested in
significant new patent applications, and we cannot be certain that any of these applications will
result in an issued patent to enhance our intellectual property rights.
| Item 1B. | UNRESOLVED STAFF COMMENTS |
Not applicable.
| Item 2. | PROPERTIES |
We own nine buildings in Sunnyvale, California, providing approximately 252,000 square feet of
space utilized for administration, marketing, sales, service, research and product development and
manufacturing. We also own an approximate 20,000 square foot building utilized for sales, service
and administration in Idstein, Germany, with approximately 77,000 square foot building for
marketing, research and development, manufacturing and administration in Germering, Germany and an
approximate 32,000 square foot building in Osaka, Japan for sales, service and administration. As
part of the acquisition of ESA Life Sciences Tools business in fiscal 2010, we acquired an
approximately 58,000 square foot building utilized for research and development and manufacturing
in Chelmsford, Massachusetts.
We lease sales and service offices in Austria, Australia, Brazil, China, Denmark, France, Germany,
India, Ireland, Italy, Japan, Korea, the Netherlands, Singapore, Sweden, Switzerland, Taiwan, the
United Kingdom and the U.S. In addition, we lease marketing and research and product development
offices in Salt Lake City, Utah. We also lease marketing and research and product development
offices in Amsterdam, the Netherlands. Our facilities are well maintained, adequate to conduct our
current business.
| Item 3. | LEGAL PROCEEDINGS |
We are a party to various legal proceedings arising in the ordinary course of our business, but are
not currently a party to any legal proceeding that management believes will have a material adverse
effect on our financial position or results of operations.
PART II
| Item 5. | MARKET FOR OUR COMMON STOCK, RELATED STOCKHOLDER MATTERS AND ISSUER REPURCHASES OF EQUITY SECURITIES |
MARKET PRICE OF COMMON STOCK
Our common stock is traded in the over-the-counter market through the Nasdaq Global Select Market
under the symbol DNEX. The following table sets forth, for the periods indicated, the high and low
sales price as reported by the Nasdaq Global Select Market.
| Fiscal 2010 | Fiscal 2009 | |||||||||||||||
| Quarter | High | Low | High | Low | ||||||||||||
First |
$ | 65.91 | $ | 56.09 | $ | 74.83 | $ | 61.39 | ||||||||
Second |
$ | 73.88 | $ | 62.97 | $ | 64.59 | $ | 43.01 | ||||||||
Third |
$ | 76.13 | $ | 65.71 | $ | 52.62 | $ | 40.03 | ||||||||
Fourth |
$ | 85.97 | $ | 74.46 | $ | 63.59 | $ | 47.83 | ||||||||
As of
August 27, 2010, there were 666 holders of record of our common stock as shown on the records
of our transfer agent.
DIVIDENDS
As of August 27, 2010, we have paid no cash dividends on our common stock and we do not anticipate
doing so in the foreseeable future.
13
Table of Contents
ISSUER PURCHASES OF EQUITY SECURITIES
During the fourth quarter of fiscal 2010, we repurchased shares of our common stock under a
systematic program to manage the dilution created by shares issued under employee stock plans and
for other purposes. This program authorizes repurchases in the open market or in private
transactions. We started a series of repurchase programs in fiscal 1989, with the Board of
Directors most recently authorizing in October 2008 future repurchases of 1,000,000 shares of
common stock as well as authorizing the repurchase of additional shares of common stock equal to
the number of common shares issued pursuant to our employee stock plans.
The following table indicates common shares repurchased and additional shares added to the program
during the quarter ended June 30, 2010:
| Total | ||||||||||||||||||||
| Number of | Maximum | |||||||||||||||||||
| Shares | Number of | |||||||||||||||||||
| Purchased | Shares that | |||||||||||||||||||
| Total | Avg. | as Part of | Additional | May Yet be | ||||||||||||||||
| Number of | Price | Publicly | Shares | Purchased | ||||||||||||||||
| Shares | Paid per | Announced | Authorized for | Under the | ||||||||||||||||
| Period | Purchased | Share | Program(1) | Purchase(1) | Program(2) | |||||||||||||||
April 1 - 30, 2010 |
— | 8,290,151 | 3,229 | 1,000,740 | ||||||||||||||||
May 1 - 31, 2010 |
218,216 | $ | 80.26 | 8,508,367 | 106,362 | 888,886 | ||||||||||||||
June 1 - 30, 2010 |
81,302 | $ | 76.01 | 8,589,669 | 10,260 | 817,844 | ||||||||||||||
Total |
299,518 | $ | 79.11 | 8,589,669 | 119,851 | 817,844 | ||||||||||||||
| (1) | The number of shares represents the number of shares issued pursuant to employee stock plans that are authorized for purchase. | |
| (2) | The number of shares includes a total of 1,000,000 shares of common stock approved for repurchase in October 2008 plus that number of shares of common stock equal to the number of shares issued pursuant to employee stock plans subsequent to October 2008 minus the number of shares purchased since October 2008. |
14
Table of Contents
PERFORMANCE GRAPH
This section is not “soliciting material”, is not deemed “filed” with the SEC and is not to be
incorporated by reference in any filing of the Company under the Securities Act of 1933, as
amended, or the Securities Exchange Act of 1934, as amended, whether made before or after the date
hereof and irrespective of any general incorporation language in any such filing.
The following graph demonstrates a comparison of cumulative total returns for our Common Stock, the
SIC Code Index and the Standard & Poor’s 500 Stock Index, assuming $100 invested as of June 30,
2005:
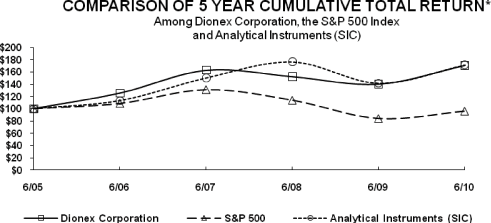
Assumes
$100 invested June 30 2005 — Assumes dividend reinvested
Fiscal year ended June 30, 2010
Fiscal year ended June 30, 2010
COMPARISON OF CUMULATIVE TOTAL RETURN OF ONE OR MORE
COMPANIES, PEER GROUPS, INDUSTRY INDEXES AND/OR BROAD MARKETS
COMPANIES, PEER GROUPS, INDUSTRY INDEXES AND/OR BROAD MARKETS
| Fiscal Year Ending | ||||||||||||||||||||||||
| COMPANY/INDEX/MARKET | 6/30/2005 | 6/30/2006 | 6/30/2007 | 6/30/2008 | 6/30/2009 | 6/30/2010 | ||||||||||||||||||
Dionex Corporation |
100.00 | 125.37 | 162.82 | 152.22 | 139.98 | 170.78 | ||||||||||||||||||
Analytical Instruments (SIC) |
100.00 | 113.14 | 150.44 | 176.81 | 141.62 | 171.54 | ||||||||||||||||||
S&P Composite |
100.00 | 108.63 | 131.00 | 113.81 | 83.98 | 96.09 | ||||||||||||||||||
15
Table of Contents
| Item 6. | SELECTED CONSOLIDATED FINANCIAL DATA |
Statement of operations information:
| Year Ended June 30, | ||||||||||||||||||||
| 2010 | 2009 (1) | 2008 (1) | 2007 (1) | 2006 (1) | ||||||||||||||||
| (In thousands, except per share amounts) | ||||||||||||||||||||
Net sales |
$ | 419,607 | $ | 385,048 | $ | 377,538 | $ | 327,284 | $ | 291,300 | ||||||||||
Cost of sales |
139,637 | 128,651 | 126,756 | 109,015 | 99,857 | |||||||||||||||
Gross profit |
279,970 | 256,397 | 250,782 | 218,269 | 191,443 | |||||||||||||||
Operating expenses: |
||||||||||||||||||||
Selling, general and administrative |
160,062 | 143,171 | 142,545 | 123,525 | 113,241 | |||||||||||||||
Research and product development |
31,586 | 28,979 | 28,943 | 24,737 | 22,392 | |||||||||||||||
Total operating expenses |
191,648 | 172,150 | 171,488 | 148,262 | 135,633 | |||||||||||||||
Operating income |
88,322 | 84,247 | 79,294 | 70,007 | 55,810 | |||||||||||||||
Interest income |
342 | 1,300 | 2,212 | 1,435 | 1,874 | |||||||||||||||
Interest expense |
(175 | ) | (493 | ) | (878 | ) | (335 | ) | (184 | ) | ||||||||||
Other income (expense), net |
592 | (1,146 | ) | (1,433 | ) | 696 | 1,200 | |||||||||||||
Income before taxes on income |
89,081 | 83,908 | 79,195 | 71,803 | 58,700 | |||||||||||||||
Taxes on income |
28,659 | 27,818 | 25,598 | 25,968 | 22,820 | |||||||||||||||
Net income |
60,422 | 56,090 | 53,597 | 45,835 | 35,880 | |||||||||||||||
Less: Net income attributable to noncontrolling interests |
1,366 | 635 | 797 | 513 | 187 | |||||||||||||||
Net income attributable to Dionex Corporation |
$ | 59,056 | $ | 55,455 | $ | 52,800 | $ | 45,322 | $ | 35,693 | ||||||||||
Basic earnings per share attributable to Dionex Corporation |
$ | 3.35 | $ | 3.10 | $ | 2.85 | $ | 2.37 | $ | 1.78 | ||||||||||
Diluted earnings per share attributable to Dionex Corporation |
$ | 3.28 | $ | 3.04 | $ | 2.77 | $ | 2.31 | $ | 1.74 | ||||||||||
Shares used in computing earnings per share amounts: |
||||||||||||||||||||
Basic |
17,643 | 17,903 | 18,506 | 19,136 | 20,013 | |||||||||||||||
Diluted |
17,983 | 18,228 | 19,072 | 19,615 | 20,527 | |||||||||||||||
| (1) | Fiscal years 2006 to 2009 were retrospectively adjusted for presentation purposes in connection with a certain new accounting standard adopted related to noncontrolling interest in subsidiaries. |
Balance sheet information:
| At June 30, | ||||||||||||||||||||
| 2010 | 2009 | 2008 | 2007 | 2006 | ||||||||||||||||
| (In thousands) | ||||||||||||||||||||
Working capital |
$ | 140,129 | $ | 128,223 | $ | 106,269 | $ | 93,780 | $ | 97,769 | ||||||||||
Total assets |
361,988 | 329,984 | 330,430 | 272,188 | 250,402 | |||||||||||||||
Long-term debt |
— | — | — | — | — | |||||||||||||||
Dionex stockholders’ equity |
248,783 | 222,414 | 196,749 | 185,708 | 185,382 | |||||||||||||||
16
Table of Contents
| ITEM 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
Cautionary Statement Regarding Forward-Looking Statements
Except for historical information contained herein, the discussion below and in the notes to our
financial statements contained in this Report on Form 10-K contains forward-looking statements
within the meaning of Section 27A of the Securities Act of 1933, as amended, Section 21E of the
Exchange Act and the Private Securities Litigation Reform Act of 1995, and are made under the safe
harbor provisions thereof. Such statements are subject to certain risks, uncertainties and other
factors that may cause actual results, performance or achievements, or industry results, to be
materially different from any future results, performance or achievements, or industry results,
expressed or implied by such forward-looking statements. Such risks and uncertainties include,
among other things: general economic conditions, foreign currency fluctuations, the risks
associated with international sales and operations, fluctuations in worldwide demand for analytical
instrumentation, fluctuations in quarterly operating results, competition from other products,
existing product obsolescence, new product development, including market receptiveness, the ability
to manufacture products on an efficient and timely basis and at a reasonable cost and in sufficient
volume, the ability to attract and retain talented employees and other risks as described in more
detail under the heading “Risk Factors.” Readers are cautioned not to place undue reliance on these
forward-looking statements that reflect management’s analysis only as of the date hereof. We
undertake no obligation to update these forward-looking statements.
Overview
Dionex Corporation designs, manufactures, markets and services analytical instrumentation and
related accessories and chemicals. Our products are used to analyze chemical substances in the
environment and in a broad range of industrial and scientific applications. Our systems are used in
environmental analysis and by the pharmaceutical, life sciences, chemical/petrochemical, power
generation, food and electronics industries in a variety of applications.
During fiscal 2010, Dionex achieved growth in certain of its end-user markets, including the life
sciences, environmental, chemical/petrochemical, food and beverage and electronics markets. We saw
weaker demand in our power market, which resulted in decreased sales. Our chemical/petrochemical,
food, and beverage, electronics and power markets are the most sensitive of our end-markets to
economic conditions. These markets showed weaker demand in the first half of the fiscal 2010 but
rebounded in the last half of fiscal 2010. We were pleased to report sales growth in all three of
our major geographic regions. We saw a strong rebound in sales in North America in the second half
of fiscal 2010 as all of our end markets, except electronics, showed stronger demand resulting in
growth for the full fiscal year. In our European regions, our life sciences customers increased
spending during fiscal 2010, along with our chemical/petrochemical customers but these increases
were offset by lower sales in our environmental and food and beverage markets. Our net sales in
Asia Pacific region increased by 16% in fiscal 2010 driven by growth in Japan, China, India, Taiwan
and Singapore. Despite the weaker economic conditions in the first six months of the fiscal year,
we also achieved our key initiatives for the year. In product development, we introduced the
largest number of new instruments and consumables in any year in the history of the company. We
believe that these new products and related applications position us well for future growth.
Looking ahead to next year, we anticipate that foreign currency fluctuations will have a negative
impact on our reported sales and earnings per share for the first quarter and the full fiscal year
of approximately 2 percentage points. Looking at our regions, we anticipate that we will see good
growth in North America in fiscal 2011 as the economy continues to recover. We anticipate that
sales in Europe will grow but not as strongly as North America in fiscal 2011. Finally, sales in
Asia/Pacific will likely grow but more in line with European rates, as we had one large sale in
Japan in fiscal 2010 that will not repeat in fiscal 2011. We expect that our gross margin will
remain in the 66% to 67% range in fiscal 2011 and that our operating expenses will increase less
than sales.
17
Table of Contents
Results of Operations
The following table summarizes our consolidated statements of income for the last three fiscal
years with each line item shown as a percentage of net sales.
| Year Ended June 30 | ||||||||||||
| 2010 | 2009 | 2008 | ||||||||||
Net sales |
100.0 | % | 100.0 | % | 100.0 | % | ||||||
Cost of sales |
33.3 | 33.4 | 33.6 | |||||||||
Gross profit |
66.7 | 66.6 | 66.4 | |||||||||
Operating expenses: |
||||||||||||
Selling, general and administrative |
38.2 | 37.2 | 37.8 | |||||||||
Research and product development |
7.5 | 7.5 | 7.7 | |||||||||
Total operating expenses |
45.7 | 44.7 | 45.5 | |||||||||
Operating income |
21.0 | 21.9 | 20.9 | |||||||||
Interest income, net |
0.1 | 0.2 | 0.4 | |||||||||
Other income (expense), net |
0.1 | (0.3 | ) | (0.4 | ) | |||||||
Income before taxes |
21.2 | 21.8 | 20.9 | |||||||||
Taxes on income |
6.8 | 7.2 | 6.8 | |||||||||
Net income |
14.4 | 14.6 | 14.1 | |||||||||
Less: Net income attributable to noncontrolling interests |
0.3 | 0.2 | 0.2 | |||||||||
Net income attributable to Dionex |
14.1 | % | 14.4 | % | 13.9 | % | ||||||
Unless otherwise noted below, our fiscal years ending June 30, 2010, June, 30, 2009 and June 30,
2008 are referred to as “2010,” “2009,” and “2008,” respectively.
Net Sales
Net sales increased $34.6 million, or 9% in 2010 compared to 2009 due to higher demand for our
products in the North America and Asia Pacific regions, partially driven by our acquisition of the
ESA Life Sciences Tools business and introduction of the Ultimate 3000 RSLCnano system and ICS-5000
RFIC System towards the end of 2010. Dionex is subject to the effects of foreign currency
fluctuations, which have an impact on net sales and gross profits. Foreign currency fluctuations
increased net sales by 3% in 2010, decreased net sales by 3% in 2009 and increased net sales by 5%
in 2008. Additionally, changes in sales or demand in any year due to price changes were
insignificant.
Including the impact of foreign currency fluctuations, percentage changes in net sales were as
follows:
| From 2009 | From 2008 | |||||||
| to | to | |||||||
| 2010 | 2009 | |||||||
Total: |
9 | % | 2 | % | ||||
By geographic region: |
||||||||
North America |
12 | % | -6 | % | ||||
Europe |
1 | % | -3 | % | ||||
Asia/Pacific |
16 | % | 17 | % | ||||
18
Table of Contents
Excluding the impact of foreign currency fluctuations, percentage changes in net sales were as
follows:
| From 2009 | From 2008 | |||||||
| to | to | |||||||
| 2010 | 2009 | |||||||
Total: |
6 | % | 5 | % | ||||
By geographic region: |
||||||||
North America |
12 | % | -5 | % | ||||
Europe |
-2 | % | 2 | % | ||||
Asia/Pacific |
12 | % | 18 | % | ||||
Net sales outside North America accounted for 73%, 74%, and 72% of net sales in 2010, 2009, and
2008, respectively. Direct sales accounted for 94%, 95%, and 93% of net sales in 2010, 2009, and
2008, respectively. International distributors and representatives in Europe, Asia and other
international markets accounted for the remaining balance of net sales. There were no significant
price changes to our products during the three-year period ended June 30, 2010.
Net sales from a regional perspective
North America - Net sales in this region increased 12% in 2010 compared to 2009 primarily due to
strong demand for our products in the life science, chemical/petrochemical, food and beverage and
environmental markets, partially driven by the acquisition of the ESA Life Sciences Tools business
that was completed in September 2009. Net sales decreased by 6% in fiscal 2009 compared to 2008
primarily due to the unfavorable economic conditions affecting the demand for our products in all
our end markets, except for our food and beverage customers.
Europe - Net sales in this region increased 1% in reported dollars in 2010 compared to 2009
(excluding the impact of foreign currency, net sales decreased 2% in 2010 compared to 2009)
reflecting weaker demand in our environmental and power markets, offset by increased demand for our
products in the life science and chemical/petrochemical markets offset by weaker demand in our
environmental and power markets. Net sales decreased 3% in reported dollars in 2009 compared to
2008 (excluding the impact of foreign currency, net sales increased 2% in 2009 compared to fiscal
2008) reflecting growth in our HPLC products offset by an unfavorable foreign currency fluctuation
as result of the strengthening U.S. dollar against the Euro in 2009.
Asia Pacific - Net sales in this region increased 16% in reported dollars in 2010 compared to 2009
(excluding the impact of foreign currency, net sales in Asia Pacific increased 12% in 2010 compared
to 2009) driven by higher demand for our products in the life sciences, environmental, food and
beverage, and electronic markets, primarily from Japan (which includes a significant one-time order
in 2010), China, India, Taiwan and Singapore. We continue to make investments in our sales and
service organization in certain emerging markets in Asia, such as China and India. Net sales
increased 17% in reported dollars in 2009 compared to 2008 (excluding the impact of foreign
currency, net sales in Asia Pacific increased 18% in 2009 compared to 2008) partially due to
continued sales force expansion in emerging markets driving higher demand for our products in the
life sciences, environmental and food and beverage markets in countries such as China, India and
Taiwan.
Net sales from a product line perspective
IC - Net sales of our IC products increased 7% in 2010 compared to 2009 driven by higher demand in
the environmental and industrial markets for our RFIC instrumentation, consumables and services in
North America and Asia/Pacific regions. Net sales increased 2% in 2009 compared to 2008 driven
primarily by higher demand in the environmental and food and beverage markets for our RFIC
instrumentation, consumables and services in the Asia/Pacific region.
HPLC - Net sales of HPLC products increased 16% in fiscal 2010 compared to 2009 primarily due to net
sales generated from our ESA products acquired in September 2009, combined with significant new
products introduced during the year, including our new RSLCnano systems. Net sales increased
slightly by 1% in 2009 compared to 2008 driven primarily by increased demands in Asia/Pacific,
partially offset by weaker demand in North America.
Gross Profit
Gross profit increased by approximately 9% in 2010 compared to 2009 and increased by approximately
2% in 2009 compared to 2008. Gross profit as a percentage of sales was 66.7%, 66.6% and 66.4% in
2010, 2009, and 2008, respectively. The increase in gross profit between the years was proportional
to the increase in net sales in those same years. Gross profit as a percentage of net sales in 2010
compared to 2009 remained relatively consistent, while gross profit as a percentage of sales in
2009 compared to 2008 increased slightly due to a higher percentage of sales occurring
internationally where there are higher gross profits in certain countries, partially offset by a
strengthening U.S. dollar against certain local currencies resulting in unfavorable foreign
currency fluctuations.
19
Table of Contents
Selling, General and Administrative
Selling, general and administrative (SG&A) expenses as a percentage of net sales were approximately
38%, 37%, and 38% in 2010, 2009, and 2008, respectively. SG&A increased 12% to $160.1 million in
2010 compared to $143.2 million in 2009 due to headcount increases, including staff added from the
ESA acquisition, and increased selling and other expenses which are attributed to the higher net
sales. In addition, our Oracle IT implementation efforts in Europe added operating expenses during
the year. SG&A expenses increased less than 1% to $143.2 million in 2009 compared to $142.6 million
in fiscal 2008 which included $3.6 million of expenses from our continued expansion efforts in
Asia/Pacific region offset by a decrease of $5.0 million due to foreign currency fluctuations. A
significant portion of the increased expenses in Asia/Pacific region was attributable to increased
selling costs.
Research and Product Development
Research and product development (R&D) expenses as a percentage of net sales were approximately 8%
in each year for 2010, 2009, and 2008. While R&D expenses increased by 9% in 2010 compared to 2009
due to increased headcount from the ESA acquisition, R&D as a percentage of net sales in 2009
compared to 2008 was in line with the revenue contributed by the acquisition of the ESA Life
Sciences Tools business.
Interest Income
Interest income was $0.3 million, $1.3 million, and $2.2 million in 2010, 2009, and 2008,
respectively. The declining trend in interest income from 2008 to 2010 was primarily due to lower
interest rates in the U.S. and Europe.
Interest Expense
Interest expense was $0.2 million, $0.5 million, and $0.9 million in 2010, 2009, and 2008,
respectively. The declining trend in interest expense from 2008 to 2010 was primarily due to lower
interest rates on short-term borrowings.
Other Income (Expense), Net
Other income (expense), net was $0.6 million, ($1.1 million), and ($1.4 million) in 2010, 2009, and
2008, respectively. The changes were primarily due to changes in foreign currency exchanges gains
or losses in those years.
Taxes on Income
Our effective tax rate was 32.0%, 33.4%, and 32.6% for fiscal 2010, 2009, and 2008, respectively.
Our effective tax rate is affected by the mix of taxable income among the various tax jurisdictions
in which we do business. The decrease in the effective tax rate in 2010 compared to 2009 was
primarily due to favorable resolutions of certain tax audits where we had previously accounted for
uncertain tax positions. The increase in the effective tax rate in 2009 compared to 2008 was
primarily due to higher state taxes.
Net income Attributable to Noncontrolling Interests
Net income attributing to noncontrolling interest was $1.4 million, $0.6 million and $0.8 million
in 2010, 2009, and 2008, respectively. These amounts result from net income in our majority-owned
subsidiaries.
Earnings per Share
Diluted earnings per share were $3.28, $3.04, and $2.77 in 2010, 2009, and 2008, respectively. The
increase in diluted earnings per share in 2010 compared to 2009 and 2009 compared to 2008 was
primarily due to increased sales during those years.
Liquidity and Capital Resources
Cash, cash equivalents and short-term investments combined were approximately $70.3 million as of
each of June 30, 2010 and 2009. Our working capital was $140.1 million and $128.2 million as of
June 30, 2010, and 2009, respectively.
Cash generated by operating activities was $67.8 million, $64.1 million, and $70.7 million in
2010, 2009, and 2008, respectively. The increase in operating cash flow in 2010 compared to 2009
was due primarily to improved operating results resulting from higher revenues, higher deferred
revenue and an increase in accrued liabilities and income taxes payable, partially offset by higher
prepaid expense and higher accounts receivable from an increase in day sales outstanding. The
decrease in operating cash flows in 2009 compared to 2008 was due to higher inventory levels due to
a new subsidiary, higher prepaid income taxes on intercompany dividends partially offset by higher
operating results, lower accruals for variable compensation and lower accounts receivable due to
faster payments from customers.
20
Table of Contents
Cash used for investing activities was $34.0 million, $17.9 million, and $15.3 million in
2010, 2009, and 2008, respectively. The increase of cash used in 2010 compared to 2009 was
primarily due to our purchase of the ESA Life Science Tools business for approximately $21.1
million in fiscal 2010 as well as our ongoing global Oracle system upgrade and its associated
worldwide rollout project cost and other related capital expenditures. The increase of cash used in
2009 compared to 2008 was primarily due to our purchase of the AutoTrace product line from Caliper
Life Sciences, Inc. for approximately $5 million in fiscal 2009 as well as an ongoing Oracle system
upgrade and its associated worldwide rollout project cost and other related capital expenditures.
Cash used for financing activities was $29.3 million, $48.3 million and $37.9 million in 2010,
2009, and 2008, respectively. Financing activities for all three years consisted primarily of
common stock repurchases, partially offset by proceeds from issuances of shares pursuant to our
equity incentive plans, repayment of short-term obligations, and the tax benefits related to stock
option plans in 2010. In 2010, we repurchased 775,644 shares of our common stock on the open market
for $54.9 million, compared with 791,517 shares repurchased for $44.2 million in 2009 and
928,131 shares repurchased for $70.3 million in 2008.
Our available lines of credit totaled $25.7 million and $28.8 million at June 30, 2010 and 2009,
respectively. The decrease in available lines of credit was due to increased borrowings outstanding
during fiscal 2010. We believe our cash flow from operations, our existing cash and cash
equivalents and our bank lines of credit will be adequate to meet our cash requirements for at
least the next 12 months. The line of credit matures on December 31, 2010 and it is our intention
to renew the line of credit. The impact of inflation on our financial position and results of
operations was not significant during any of the periods presented.
CONTRACTUAL OBLIGATIONS
The following summarizes our contractual obligations as of June 30, 2010, and the effect such
obligations are expected to have on our liquidity and cash flows in future periods:
| Payments Due by Period | ||||||||||||||||||||
| Less Than | 1-3 | 4-5 | After 5 | |||||||||||||||||
| (in thousands) | Total | 1 Year | Year | Years | Years | |||||||||||||||
Short-Term Borrowings |
$ | 3,149 | $ | 3,149 | $ | — | $ | — | $ | — | ||||||||||
Purchase Obligations |
527 | — | 527 | — | — | |||||||||||||||
Operating Lease Obligations |
12,336 | 5,507 | 4,367 | 1,102 | 1,360 | |||||||||||||||
Total |
$ | 16,012 | $ | 8,656 | $ | 4,894 | $ | 1,102 | $ | 1,360 | ||||||||||
As of June 30, 2010, we had approximately $8.0 million recorded pertaining to unrecognized tax
benefits. We are unable to make a reasonable estimate as to when these amounts will be paid or
released due to the uncertainties related to these tax matters, therefore these amounts are
excluded from the table above.
ACCOUNTING PRONOUNCEMENTS
Refer to Note 1 in the Notes to Consolidated Financial Statements contained elsewhere in this
Annual Report on Form 10-K, which is incorporated herein by reference
CRITICAL ACCOUNTING POLICIES AND ESTIMATES
Summary
The preparation of consolidated financial statements requires us to make estimates and assumptions
that affect the reported amounts of assets and liabilities and disclosure of contingent assets and
liabilities at the date of the financial statements and the reported amounts of net sales and
expenses during the reporting period. We evaluate our estimates, including those related to product
returns and allowances, inventory valuation, goodwill and other intangible assets, income taxes,
warranty and installation provisions, and contingencies, on an on-going basis.
We base our estimates on historical experience and on various other assumptions that are believed
to be reasonable under the circumstances, the results of which form the basis for making judgments
about the carrying values of assets and liabilities that are not readily apparent from other
sources. Actual results may differ from these estimates. We believe the following critical
accounting policies affect our more significant judgments and estimates used in the preparation of
our consolidated financial statements.
21
Table of Contents
Revenue Recognition Policy
Net sales consist primarily of sales of products, software licenses, and services (including
installation, training, other consulting services, and extended maintenance contracts on our
products). Dionex recognizes revenue when persuasive evidence of an arrangement exists, the product
has been delivered or service has been performed, the sales price is fixed or determinable, and
collection is reasonably assured. Delivery of the product is generally considered to have occurred
when shipped. Shipping charges billed to customers are included in net sales, and the related costs
are included in cost of sales.
In all cases, the portion of revenue allocated to any undelivered elements is deferred until those
items are delivered to the customer or the services are performed. Delivery of the product is
generally considered to have occurred when shipped. Undelivered elements in our sales arrangements,
which are not considered to be essential to the functionality of a product, generally include
maintenance, installation services, and/or training that are delivered after the related products
have been delivered. Product installation consists of system set-up, calibration and basic
functionality training and generally requires one to three days depending on the product. The fair
value of these product installation services is calculated by applying standard service billing
rates to the estimate of the number of hours to install a specific product based on historical
experience. These estimated hours for product installation have historically been accurate and
consistent from product to product. The fair value of training services are based on the price
charged when the element is sold separately or, if not sold separately, when the price is
established by authorized management. However, to the extent these estimates were to reflect
unfavorable variability, our ability to maintain objective reliable evidence of fair value for such
element could be impacted, which in turn could delay the recognition of the revenue currently
allocated to the delivered elements. For sales of extended maintenance contracts with a separate
contract price, Dionex defers revenue equal to the separately stated price and recognize revenue on
a straight-line basis over the contract period.
Sales from products are typically not subject to rights of return and, historically, actual sales
returns have not been significant. Dionex sells products through its direct sales force and through
distributors and resellers. Sales through distributors and resellers are recognized as revenue upon
sale to the distributor or reseller as these sales are considered to be final and no right of
return or price protection exists. Customer acceptance is generally limited to performance under
our published product specifications. When additional customer acceptance conditions apply, all
revenue related to the sale is deferred until acceptance is obtained.
In accordance with the software revenue guidance, Dionex recognizes revenue from perpetual software
licenses at the inception of the license term assuming all revenue recognition criteria have been
met. Dionex uses the residual method to allocate revenue to software licenses at the
inception of the license term when vendor specific objective evidence (“VSOE”) of fair value for
the undelivered elements exists, such as software installations, and all other revenue recognition
criteria have been satisfied. If Dionex cannot objectively determine the fair value of any
undelivered elements included in these multiple-element arrangements, revenue is deferred until all
elements are delivered, or until fair value can be objectively determined for the remaining
undelivered elements.
Product Warranty
The sales of Dionex products generally include a limited one-year warranty on product quality. The
estimated cost of product warranty claims is accrued at the time the sale is recognized, based on
historical experience. While Dionex believes that historical experience provides a reliable basis
for estimating such warranty cost, unforeseen quality issues or component failure rates could
result in future costs in excess of such estimates, or alternatively, improved quality and
reliability in our products could result in actual expenses that are below those currently
estimated.
Loss Provisions on Accounts Receivable and Inventory
Dionex maintains allowances for doubtful accounts for estimated losses resulting from the inability
of customers to make required payments. If the financial condition of any of our customers were to
deteriorate, resulting in an impairment of their ability to make payments, additional allowances
may be required. Dionex assesses collectability based on a number of factors including, but not
limited to, past transaction history with the customer, the credit-worthiness of the customer,
independent credit reports, industry trends and the macro-economic environment. Sales returns and
allowances are estimates of future product returns related to current period revenue. Material
differences may result in the amount and timing of our revenue for any period.
Inventories, consisting of raw materials, work-in-process and finished goods are stated at the
lower of cost (first-in, first-out) or market. Dionex plans production based on orders received and
forecasted demand and assesses the valuation of its inventory on a quarterly basis. Products may be
unsalable due to market and economic conditions, technology changes, new product introductions and
changes in strategic direction and require estimates that may include uncertain elements. Actual
demand may differ from forecasted demand and such differences may have a material effect on
recorded inventory values. Based on management’s estimate, adjustments to reduce the cost of
inventory to its net realizable value, if required, are made for estimated excess or obsolete
inventory.
22
Table of Contents
Long-Lived Assets, Intangible Assets with Finite Lives and Goodwill
Dionex assesses for the impairment of long-lived assets, intangible assets and goodwill whenever
events or changes in circumstances indicate that the carrying value may not be recoverable. In
addition, we assess goodwill and intangible assets with indefinitely lives for impairment at least
annually. Factors Dionex consider important which could trigger an impairment review include but
are not limited to the following:
| • | Significant underperformance relative to historical or projected future operating results; |
| • | Significant negative industry or economic trends; and |
| • | Significant changes or developments in strategic technology. |
When Dionex determines that the carrying value of long-lived assets and intangible assets with
finite lives may not be recoverable based upon the existence of one or more of the above or other
indicators, we measure any impairment based on a projected discounted cash flow method using a
discount rate determined by our management to be commensurate with the risk inherent in our current
business model. Goodwill is tested for impairment by comparing the fair values of related reporting
units to their carrying values. Dionex performs an impairment review for goodwill at least annually
in April of each year.
Intangible assets with indefinite lives are not amortized but rather are tested for impairment
annually, or more frequently if events or changes in circumstances indicate that the asset might be
impaired. The impairment test, which is performed in April of each year, involves comparing the
fair value of the intangible asset with its carrying amount. If the carrying amount exceeds the
fair value of the intangible asset, an impairment loss is recognized in an amount equal to that
excess. No impairment charges for indefinite life intangible assets have been recorded in any
period presented.
Taxes on Income
As part of the process of preparing the consolidated financial statements, Dionex estimates income
taxes in each of the jurisdictions in which we operate. The determination of our tax provision is
subject to judgments and estimates due to the complexity of the tax laws that we are subject to in
several tax jurisdictions. This process involves our estimate of our actual current tax exposure
together with assessing temporary differences resulting from differing treatment of items, such as
depreciation, amortization and inventory reserves for tax and accounting purposes. These
differences result in deferred tax assets and liabilities, which are included with our consolidated
balance sheets.
Dionex accounts for income taxes in accordance with the accounting standards for income taxes which
require that deferred tax assets and liabilities be recognized for the effect of temporary
differences between the carrying amounts of assets and liabilities for financial reporting purposes
and the amounts used for income tax purposes. These accounting standards also require that deferred
tax assets be reduced by a valuation allowance if it is more likely than not that some or all of
the deferred tax asset will not be realized.
In first quarter of fiscal 2008, Dionex adopted a new accounting standards which clarifies the
accounting for uncertainty in income taxes recognized in an enterprise’s financial statements. This
interpretation prescribes a recognition threshold and measurement attribute for the financial
statement recognition and measurement of a tax position taken or expected to be taken in a tax
return. As a result of the interpretation, we recognize the tax liability for uncertain income tax
positions on the income tax return based on the two-step process prescribed in the interpretation.
The first step is to determine whether it is more likely than not that each income tax position
would be sustained upon audit. The second step is to estimate and measure the tax benefit as the
amount that has a greater than 50% likelihood of being realized upon ultimate settlement with the
tax authority. Estimating these amounts requires us to determine the probability of various
possible outcomes. Dionex evaluates these uncertain tax positions on a quarterly basis. This
evaluation is based on the consideration of several factors, including changes in facts or
circumstances, changes in applicable tax law, settlement of issues under audit, and new exposures.
If Dionex later determine that the exposure is lower or that the liability is not sufficient to
cover our revised expectations, Dionex will adjust the liability and effect a related change in tax
provision during the period in which Dionex makes such determination.
23
Table of Contents
Stock-Based Compensation
Dionex accounts for stock-based compensation costs in accordance with the accounting standards for
stock-based compensation, which requires that all share-based payments to employees be recognized
in the statements of operations based on their fair values. The fair value of each option on the
date of grant is estimated using the Black-Scholes option-pricing model using the single option
approach for options granted after June 30, 2005. Dionex recognizes the expense ratably on a
straight-line basis over the requisite service period. The stock-based compensation expense
recognized in the consolidated statements of operations is based on awards that ultimately are
expected to vest; therefore, the amount of expense has been reduced for estimated forfeitures. This
accounting standard requires forfeitures to be estimated at the time of grant and revised, if
necessary, in subsequent periods if actual forfeitures differ from those estimates. Forfeitures
were estimated based on historical experience. If actual results differ significantly from these
estimates, stock-based compensation expense and Dionex’s results of operations could be materially
impacted. In addition, if Dionex employs different assumptions in the application of this
accounting standard, the compensation expense that Dionex records in the future periods may differ
significantly from what Dionex has recorded in the current period.
| Item 7A. | QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK |
Dionex is exposed to financial market risks from fluctuations in foreign currency exchange rates,
interest rates and stock prices of marketable securities. With the exception of the stock price
volatility of our marketable equity securities, we manage our exposure to these and other risks
through our regular operating and financing activities and, when appropriate, through our hedging
activities. Our policy is not to use hedges or other derivative financial instruments for
speculative purposes. We deal with a diversified group of major financial institutions to limit the
risk of nonperformance by any one institution on any financial instrument. Separate from our
financial hedging activities, material changes in foreign exchange rates, interest rates and, to a
lesser extent, commodity prices could cause significant changes in the costs to manufacture and
deliver our products and in customers’ buying practices. We have not substantially changed our risk
management practices during fiscal 2010 and we do not currently anticipate significant changes in
financial market risk exposures in the near future that would require us to change our current risk
management practices.
Foreign Currency Exchange
Revenues generated from international operations are generally denominated in foreign currencies.
We entered into forward foreign exchange contracts to hedge against fluctuations of intercompany
account balances. Market value gains and losses on these hedge contracts are substantially offset
by fluctuations in the underlying balances being hedged, and the net financial impact is not
expected to be material in future periods. As of June 30, 2010, we had forward exchange contracts to
sell foreign currencies totaling approximately $23.7 million, including approximately $17.7 million
in Euros, $4.3 million in Japanese yen, $0.8 million in Australian dollars and $0.9 million in
Canadian dollars. As of June 30, 2009, we had forward exchange contracts to sell foreign currencies
totaling approximately $15.2 million, including approximately $9.8 million in Euros, $3.9 million
in Japanese yen, $0.9 million in Australian dollars and $0.6 million in Canadian dollars. At June
30, 2010 and 2009, the aggregate unrealized gains or losses on the forward exchange contracts were
not material.
Dionex uses a $10 million cross-currency swap arrangement for Japanese yen designated as a net
investment hedge to hedge the exchange rate exposure of our net investment in its Japanese
subsidiary. During fiscal 2010, the arrangement was extended to March 2012. Prior to January 1,
2008, this derivative instrument did not qualify for net investment hedge accounting and was deemed
to be an ineffective hedge instrument because, at the inception of the hedge transaction, there was
no formal documentation of the hedging relationship and our risk management objective and strategy
for undertaking the hedge. Therefore, Dionex marked to market the decrease in value of
approximately $1.0 million for the six months ended December 31, 2007 and this amount was recorded
in other expense, net. Starting on January 1, 2008, Dionex determined that the cross-currency swap
qualified as a net investment hedge in which the effective portion of the gain or loss on the
derivative instruments is reported in accumulated other comprehensive income as part of the foreign
currency translation adjustment. Dionex evaluates the need to redesignate the hedging relationship
on a quarterly basis. In fiscal 2010 and 2009, we marked to market $1.2 million and $1.1 million,
respectively, in unrealized losses associated with the hedge, which is reported in accumulated
other comprehensive income as part of the foreign currency translation adjustment.
A sensitivity analysis assuming a hypothetical 10% movement in foreign currency exchange rates
applied to our hedging contracts and underlying balances being hedged as of June 30, 2010 and 2009
indicated that these market movements would not have a material effect on our business, operating
results or financial condition.
Foreign currency rate fluctuations can impact the U.S. dollar translation of our foreign operations
in our consolidated financial statements. Currency fluctuations increased sales by 3% in fiscal
2010 and decreased sales by 3% in fiscal 2009 and increased sales by 5% in fiscal 2008.
24
Table of Contents
Interest and Investment Income
Our interest and investment income is subject to changes in the general level of U.S. interest
rates. Changes in U.S. interest rates affect the interest earned on our cash equivalents and
short-term investments. A sensitivity analysis assuming a hypothetical 10% movement in interest
rates applied to our investment balances as of June 30, 2010 and 2009 indicated that such market
movement would not have a material effect on our business, operating results or financial
condition. Actual gains or losses in the future may differ materially from this analysis, depending
on our actual balances and changes in the timing and amount of interest rate movements.
Debt and Interest Expense
As of June 30, 2010, Dionex had short-term notes payable of $3.1 million. A sensitivity analysis
assuming a hypothetical 10% movement in interest rates applied to our outstanding debt balance as of
June 30, 2010, indicated that such market movement would not have a material effect on our
business, operating results or financial condition. Actual gains or losses in the future may differ
materially from this analysis, depending on changes in the timing and amount of interest rate
movements and the level of borrowings maintained by us.
25
Table of Contents
| Item 8. | FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA |
| Page | ||||
FINANCIAL STATEMENTS |
||||
| 27 | ||||
| 28 | ||||
| 29 | ||||
| 30 | ||||
| 31 | ||||
| 32 | ||||
26
Table of Contents
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders of Dionex Corporation:
We have audited the accompanying consolidated balance sheets of Dionex Corporation and subsidiaries
(the “Company”) as of June 30, 2010 and 2009, and the related consolidated statements of income,
stockholders’ equity and comprehensive income, and cash flows for each of the three years in the
period ended June 30, 2010. These financial statements are the responsibility of the Company’s
management. Our responsibility is to express an opinion on these financial statements based on our
audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight
Board (United States). Those standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are free of material misstatement. An
audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the
financial statements. An audit also includes assessing the accounting principles used and
significant estimates made by management, as well as evaluating the overall financial statement
presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, such consolidated financial statements present fairly, in all material respects,
the financial position of Dionex Corporation and subsidiaries at June 30, 2010 and 2009, and the
results of their operations and their cash flows for each of the three years in the period ended
June 30, 2010, in conformity with accounting principles generally accepted in the United States of
America.
We have also audited, in accordance with the standards of the Public Company Accounting Oversight
Board (United States), the Company’s internal control over financial reporting as of June 30, 2010,
based on the criteria established in Internal Control—Integrated Framework issued by the Committee
of Sponsoring Organizations of the Treadway Commission and our report dated August 27, 2010
expressed an unqualified opinion on the Company’s internal control over financial reporting.
/s/ Deloitte & Touche LLP
San Jose, California
August 27, 2010
San Jose, California
August 27, 2010
27
Table of Contents
DIONEX CORPORATION
CONSOLIDATED BALANCE SHEETS
(In thousands, except share and per share amounts)
AS OF JUNE 30
AS OF JUNE 30
| 2010 | 2009 | |||||||
ASSETS |
||||||||
Current assets: |
||||||||
Cash and cash equivalents |
$ | 69,830 | $ | 69,684 | ||||
Short-term investments |
448 | 641 | ||||||
Accounts receivable (net of allowance for doubtful accounts of $543 and $560 in 2010 and
2009, respectively) |
86,780 | 70,535 | ||||||
Inventories |
37,458 | 31,274 | ||||||
Deferred taxes |
14,036 | 12,171 | ||||||
Prepaid expenses and other current assets |
18,991 | 21,917 | ||||||
Total current assets |
227,543 | 206,222 | ||||||
Property, plant and equipment, net |
76,062 | 71,927 | ||||||
Goodwill |
35,013 | 29,354 | ||||||
Intangible assets, net |
13,859 | 8,506 | ||||||
Other assets |
9,511 | 13,975 | ||||||
Total assets |
$ | 361,988 | $ | 329,984 | ||||
LIABILITIES AND STOCKHOLDERS’ EQUITY |
||||||||
Current liabilities: |
||||||||
Borrowings under line of credit |
$ | 3,149 | $ | 64 | ||||
Accounts payable |
17,303 | 16,545 | ||||||
Accrued liabilities |
33,980 | 31,222 | ||||||
Deferred revenues |
25,203 | 22,559 | ||||||
Income taxes payable |
5,247 | 4,581 | ||||||
Accrued product warranty |
2,532 | 3,028 | ||||||
Total current liabilities |
87,414 | 77,999 | ||||||
Deferred and other income taxes payable |
16,427 | 24,348 | ||||||
Other long-term liabilities |
7,272 | 3,707 | ||||||
Commitments and contingencies (Note 14) |
||||||||
Stockholders’ equity: |
||||||||
Preferred stock (par value $.001 per share; 1,000,000 shares authorized; none outstanding) |
— | — | ||||||
Common stock
(par value $.001 per share; 80,000,000 shares authorized; 17,437,276 and
17,759,690 shares issued and outstanding in 2010 and 2009, respectively) and capital in
excess of par value |
207,855 | 186,649 | ||||||
Retained earnings |
34,195 | 21,459 | ||||||
Accumulated other comprehensive income |
6,733 | 14,306 | ||||||
Total Dionex Corporation stockholders’ equity |
248,783 | 222,414 | ||||||
Noncontrolling interests |
2,092 | 1,516 | ||||||
Total stockholders’ equity |
250,875 | 223,930 | ||||||
Total liabilities and stockholders’ equity |
$ | 361,988 | $ | 329,984 | ||||
See notes to consolidated financial statements.
28
Table of Contents
DIONEX CORPORATION
CONSOLIDATED STATEMENTS OF INCOME
(In thousands, except per share amounts)
YEAR ENDED JUNE 30
YEAR ENDED JUNE 30
| 2010 | 2009 | 2008 | ||||||||||
Net sales |
$ | 419,607 | $ | 385,048 | $ | 377,538 | ||||||
Cost of sales |
139,637 | 128,651 | 126,756 | |||||||||
Gross profit |
279,970 | 256,397 | 250,782 | |||||||||
Operating expenses: |
||||||||||||
Selling, general and administrative |
160,062 | 143,171 | 142,545 | |||||||||
Research and product development |
31,586 | 28,979 | 28,943 | |||||||||
Total operating expenses |
191,648 | 172,150 | 171,488 | |||||||||
Operating income |
88,322 | 84,247 | 79,294 | |||||||||
Interest income |
342 | 1,300 | 2,212 | |||||||||
Interest expense |
(175 | ) | (493 | ) | (878 | ) | ||||||
Other income (expense), net |
592 | (1,146 | ) | (1,433 | ) | |||||||
Income before taxes |
89,081 | 83,908 | 79,195 | |||||||||
Taxes on income |
28,659 | 27,818 | 25,598 | |||||||||
Net income |
60,422 | 56,090 | 53,597 | |||||||||
Less: Net income attributable to noncontrolling interests |
1,366 | 635 | 797 | |||||||||
Net income attributable to Dionex Corporation |
$ | 59,056 | $ | 55,455 | $ | 52,800 | ||||||
Basic earnings per share attributable to Dionex Corporation |
$ | 3.35 | $ | 3.10 | $ | 2.85 | ||||||
Diluted earnings per share attributable to Dionex Corporation |
$ | 3.28 | $ | 3.04 | $ | 2.77 | ||||||
Shares used in computing earnings per share: |
||||||||||||
Basic |
17,643 | 17,903 | 18,506 | |||||||||
Diluted |
17,983 | 18,228 | 19,072 | |||||||||
See notes to consolidated financial statements.
29
Table of Contents
DIONEX CORPORATION
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY AND COMPREHENSIVE INCOME
(In thousands, except for share amounts)
| Accumulated | ||||||||||||||||||||||||||||
| Other | ||||||||||||||||||||||||||||
| Common Stock | Retained | Comprehensive | Noncontrolling | Comprehensive | ||||||||||||||||||||||||
| Shares | Amount | Earnings | Income | Interest | Total | Income | ||||||||||||||||||||||
Balance, June 30, 2007 |
18,845,802 | $ | 161,409 | $ | 13,223 | $ | 11,076 | $ | 722 | $ | 186,430 | |||||||||||||||||
Distributions to noncontrolling interest |
(179 | ) | (179 | ) | ||||||||||||||||||||||||
Net income |
52,800 | 797 | 53,597 | $ | 53,597 | |||||||||||||||||||||||
Cumulative translation adjustments (net of tax of $6,308) |
13,042 | 13,042 | 13,042 | |||||||||||||||||||||||||
Unrealized gain on securities |
4 | 4 | 4 | |||||||||||||||||||||||||
Comprehensive income, net of tax |
66,643 | |||||||||||||||||||||||||||
Comprehensive income attributable to noncontrolling interests |
(797 | ) | ||||||||||||||||||||||||||
Comprehensive income attributable to Dionex Corporation |
$ | 65,846 | ||||||||||||||||||||||||||
Common stock issued under employee stock-based compensation
plans |
213,042 | 9,143 | 9,143 | |||||||||||||||||||||||||
Repurchase of common stock |
(928,131 | ) | (8,278 | ) | (62,019 | ) | (70,297 | ) | ||||||||||||||||||||
Stock-based compensation expense |
5,939 | 5,939 | ||||||||||||||||||||||||||
Tax benefit from employee stock transactions |
1,832 | 1,832 | ||||||||||||||||||||||||||
Adoption of uncertain tax positions accounting standards |
(1,422 | ) | (1,422 | ) | ||||||||||||||||||||||||
Balance, June 30, 2008 |
18,130,713 | $ | 170,045 | $ | 2,582 | $ | 24,122 | $ | 1,340 | $ | 198,089 | |||||||||||||||||
Distributions to noncontrolling interest |
(459 | ) | (459 | ) | ||||||||||||||||||||||||
Net income |
55,455 | 635 | 56,090 | $ | 56,090 | |||||||||||||||||||||||
Cumulative translation adjustments (net of tax of $3,805) |
(9,815 | ) | (9,815 | ) | (9,815 | ) | ||||||||||||||||||||||
Unrealized loss on securities |
(1 | ) | (1 | ) | (1 | ) | ||||||||||||||||||||||
Comprehensive income, net of tax |
46,274 | |||||||||||||||||||||||||||
Comprehensive income attributable to noncontrolling interests |
(635 | ) | ||||||||||||||||||||||||||
Comprehensive income attributable to Dionex Corporation |
$ | 45,639 | ||||||||||||||||||||||||||
Common stock issued under employee stock-based compensation
plans |
420,494 | 15,864 | 15,864 | |||||||||||||||||||||||||
Repurchase of common stock |
(791,517 | ) | (7,654 | ) | (36,578 | ) | (44,232 | ) | ||||||||||||||||||||
Stock-based compensation expense |
6,594 | 6,594 | ||||||||||||||||||||||||||
Tax benefit from employee stock transactions |
1,800 | 1,800 | ||||||||||||||||||||||||||
Balance, June 30, 2009 |
17,759,690 | $ | 186,649 | $ | 21,459 | $ | 14,306 | $ | 1,516 | $ | 223,930 | |||||||||||||||||
Distributions to noncontrolling interest |
(790 | ) | (790 | ) | ||||||||||||||||||||||||
Net income |
59,056 | 1,366 | 60,422 | $ | 60,422 | |||||||||||||||||||||||
Cumulative translation adjustments (net of tax of $4,020) |
(7,574 | ) | (7,574 | ) | (7,574 | ) | ||||||||||||||||||||||
Unrealized gain on securities |
1 | 1 | 1 | |||||||||||||||||||||||||
Comprehensive income |
52,849 | |||||||||||||||||||||||||||
Comprehensive income attributable to noncontrolling interests |
(1,366 | ) | ||||||||||||||||||||||||||
Comprehensive income attributable to Dionex Corporation |
$ | 51,483 | ||||||||||||||||||||||||||
Common stock issued under employee stock-based compensation
plans |
453,230 | 20,373 | 20,373 | |||||||||||||||||||||||||
Repurchase of common stock |
(775,644 | ) | (8,575 | ) | (46,320 | ) | (54,895 | ) | ||||||||||||||||||||
Stock-based compensation expense |
6,783 | 6,783 | ||||||||||||||||||||||||||
Tax benefit from employee stock transactions |
2,625 | 2,625 | ||||||||||||||||||||||||||
Balance, June 30, 2010 |
17,437,276 | $ | 207,855 | $ | 34,195 | $ | 6,733 | $ | 2,092 | $ | 250,875 | |||||||||||||||||
See notes to consolidated financial statements.
30
Table of Contents
DIONEX CORPORATION
CONSOLIDATED STATEMENTS OF CASH FLOWS
(In thousands)
YEAR ENDED JUNE 30
YEAR ENDED JUNE 30
| 2010 | 2009 | 2008 | ||||||||||
Cash flows from operating activities: |
||||||||||||
Net income |
$ | 60,422 | $ | 56,090 | $ | 53,597 | ||||||
Adjustments to reconcile net income to net cash provided by operating activities: |
||||||||||||
Depreciation and amortization |
12,092 | 9,491 | 8,767 | |||||||||
Stock-based compensation |
6,783 | 6,594 | 5,939 | |||||||||
Allowance for bad debts |
69 | 135 | (232 | ) | ||||||||
Loss on disposal of fixed assets |
344 | 363 | 543 | |||||||||
Tax benefit related to stock transactions |
(2,625 | ) | (1,800 | ) | (1,832 | ) | ||||||
Deferred income taxes |
930 | 1,184 | (944 | ) | ||||||||
Changes in assets and liabilities: |
||||||||||||
Accounts receivable |
(19,835 | ) | (38 | ) | (1,321 | ) | ||||||
Inventories |
(5,681 | ) | (2,343 | ) | 1,688 | |||||||
Prepaid expenses and other assets |
(58 | ) | (7,322 | ) | (725 | ) | ||||||
Accounts payable |
1,821 | 432 | 1,506 | |||||||||
Deferred revenue |
4,215 | 2,079 | 2,398 | |||||||||
Accrued liabilities |
6,486 | (1,298 | ) | 3,912 | ||||||||
Income taxes payable |
3,214 | 733 | (2,895 | ) | ||||||||
Accrued product warranty |
(406 | ) | (228 | ) | 290 | |||||||
Net cash provided by operating activities |
67,771 | 64,072 | 70,691 | |||||||||
Cash flows from investing activities: |
||||||||||||
Proceeds from sale of marketable securities |
225 | — | 124 | |||||||||
Purchase of short term investments |
— | (568 | ) | (77 | ) | |||||||
Purchase of property, plant and equipment |
(13,019 | ) | (11,379 | ) | (13,847 | ) | ||||||
Purchase of intangible assets |
(112 | ) | — | (2,071 | ) | |||||||
Acquisition, net of cash acquired |
(21,147 | ) | (5,976 | ) | 543 | |||||||
Proceeds from property, plant and equipments disposed |
42 | — | — | |||||||||
Net cash used for investing activities |
(34,011 | ) | (17,923 | ) | (15,328 | ) | ||||||
Cash flows from financing activities: |
||||||||||||
Net
borrowings (payments) under line-of-credit |
3,087 | (21,746 | ) | 21,506 | ||||||||
Proceeds from issuance of common stock |
20,373 | 15,864 | 9,143 | |||||||||
Repayments of capital lease obligations |
— | — | (58 | ) | ||||||||
Tax benefit related to stock transactions |
2,625 | 1,800 | 1,832 | |||||||||
Repurchase of common stock |
(54,895 | ) | (44,232 | ) | (70,297 | ) | ||||||
Dividends paid |
(453 | ) | — | — | ||||||||
Net cash used for financing activities |
(29,263 | ) | (48,314 | ) | (37,874 | ) | ||||||
Effect of exchange rate changes on cash |
(4,351 | ) | (3,775 | ) | 3,197 | |||||||
Net increase (decrease) in cash and equivalents |
146 | (5,940 | ) | 20,686 | ||||||||
Cash and
cash equivalents, beginning of year |
69,684 | 75,624 | 54,938 | |||||||||
Cash and cash equivalents, end of year |
$ | 69,830 | $ | 69,684 | $ | 75,624 | ||||||
Supplemental disclosures of cash flow information: |
||||||||||||
Income taxes paid |
$ | 20,685 | $ | 28,865 | $ | 31,162 | ||||||
Interest expense paid |
$ | 151 | $ | 451 | $ | 790 | ||||||
Supplemental schedule of non-cash investing and financing activities: |
||||||||||||
Accrued purchases of property, plant and equipment |
$ | 477 | $ | 919 | $ | 1,563 | ||||||
Accrued purchase consideration |
$ | — | $ | 657 | $ | — | ||||||
Elimination of equity interest associated with step-acquisition of business |
$ | — | $ | 760 | $ | — | ||||||
Effect of adoption of uncertain tax positions accounting standards: |
||||||||||||
Cumulative effect on retained earnings |
$ | — | $ | — | $ | 1,422 | ||||||
Increase in deferred and other income taxes payable |
$ | — | $ | — | $ | 16,033 | ||||||
Increase in other assets |
$ | — | $ | — | $ | 11,409 | ||||||
Decrease in income taxes payable |
$ | — | $ | — | $ | 3,202 | ||||||
Capital lease obligation for equipment purchases |
$ | — | $ | — | $ | 487 | ||||||
See notes to consolidated financial statements.
31
Table of Contents
DIONEX CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Note 1: Summary of Significant Accounting Policies
Organization and Basis of Presentation
Dionex Corporation designs, manufactures and markets chromatography systems for chemical analysis.
Our products are used in environmental analysis and by the pharmaceutical, life sciences,
chemical/petrochemical, power generation, food and electronics industries in a variety of
applications. The consolidated financial statements include the accounts of Dionex Corporation and
all of our wholly and majority owned subsidiaries (referred to in this report as “Dionex,” “we,”
“us” and “our”).
All significant intercompany accounts and transactions have been eliminated in consolidation. The
preparation of financial statements in conformity with accounting principles generally accepted in
the United States of America requires management to make estimates and assumptions that affect the
reported amounts of assets and liabilities, disclosure of contingent assets and liabilities at the
date of the financial statements, and the reported amounts of net sales and expenses during the
reported periods. Actual results could differ materially from those estimates and assumptions.
Cash, Cash Equivalents and Short-Term Investments
Cash and cash equivalents are carried at market value. Dionex considers all highly liquid
investments with maturities of three months or less at the date of purchase to be cash equivalents.
Dionex has classified short-term investments, which consist primarily of debt and equity
securities as “held-to-maturity” or “available-for-sale.” Securities classified as held-to-maturity
are reported at amortized cost. Available-for-sale securities are reported at fair market value,
with the aggregate unrealized holding gains and losses (net of tax effect) reported as a component
of accumulated other comprehensive income in stockholders’ equity. Dionex does not have any
held-to-maturity securities as of June 30, 2010 and 2009.
Concentration of Credit Risk
Dionex places cash, cash equivalents and short-term investments with and in the custody of
financial institutions that management believes are of high credit quality, which at times, are in
excess of amounts insured by the Federal Deposit Insurance Corporation (FDIC). Management monitors
the ongoing creditworthiness of these institutions. To date, Dionex has not experienced significant
credit losses on these financial instruments.
Dionex sells its products primarily to large organizations in diversified industries worldwide.
Credit risk is further mitigated by our credit evaluation process and the reasonably short
collection terms. Dionex does not require collateral or other security to support accounts
receivable and Dionex maintains allowances for potential credit losses.
Dionex is subject to certain risks and uncertainties and believes that changes in any of the
following areas could have a material adverse effect on our future financial position or results of
operations. Such factors include, among others: the continuation or spread of economic
uncertainties; risks related to international operations, including foreign currency fluctuations;
the importance of meeting customer demand for new products; competition in the analytical
instrumentation market; our ability to maintain inventories; the importance of attracting and
retaining key personnel; our ability to protect our proprietary information and acceptance of new
products.
Inventories
Inventories, consisting of raw materials, work-in-process and finished goods are stated at the
lower of cost (first-in, first-out) or market. Dionex plans production based on orders received and
forecasted demand and assesses the valuation of its inventory on a quarterly basis. Products may be
unsalable due to market and economic conditions, technology changes, new product introductions and
changes in strategic direction and require estimates that may include uncertain elements. Actual
demand may differ from forecasted demand and such differences may have a material effect on
recorded inventory values. Based on management’s estimate, adjustments to reduce the cost of
inventory to its net realizable value, if required, are made for estimated excess or obsolete
inventory.
32
Table of Contents
Property, Plant and Equipment
Property, plant and equipment are stated at cost less accumulated depreciation. Dionex capitalizes
additions and improvements and expense maintenance and repairs as incurred. Depreciation is
computed using the straight-line method based on estimated useful lives, ranging from 3 to
30 years. Leasehold improvements are amortized over the lesser of the useful life or the remaining
term of the lease.
Capitalized Software
Dionex capitalizes certain internal and external costs incurred to acquire or create internal use
software. Capitalized software is included in property, plant, and equipment and is depreciated
using the straight-line method over the estimated useful life of the software, generally 3 to 5
years.
Goodwill and Intangible Assets
Dionex tests goodwill for impairment on an annual basis in April or more frequently if we believe
indicators of impairment exist. The performance of the test involves a two-step process. The first
step requires comparing the fair value of the reporting unit to its net book value, including
goodwill. A potential impairment exists if the fair value of the reporting unit is lower than its
net book value. The second step of the process is only performed if a potential impairment exists,
and it involves comparing the aggregate fair value of the reporting unit’s net assets other than
goodwill to the fair value of the reporting unit as a whole. Goodwill is considered impaired, and
an impairment charge is recorded, if the excess of the fair value of the reporting unit over the
fair value of the net assets is less than the carrying value of goodwill. Dionex found no
impairment as a result of our fiscal 2010, 2009, and 2008 annual impairment reviews, as the fair
value of our operating segments were in excess of its carrying value.
Intangible assets with finite lives include purchased technology, patents, trademarks and other
intellectual properties. Intangible assets with finite lives are recorded at their fair market
values as of the date of acquisition and amortized over their estimated useful lives of up to ten
years.
Valuation of Long-lived Assets
The carrying value of our long-lived assets is reviewed for impairment whenever events or changes
in circumstances indicated that an asset may not be recoverable.
Dionex looks at current and future
profitability, as well as current and future undiscounted cash flows, as primary indicators of
recoverability. If impairment is determined to exist, any related impairment loss is calculated
based on the amount by which the carrying value of the asset exceeds the fair value of the asset
with fair value determined on a discounted cash flow basis.
Intangible assets with indefinite lives are not amortized but rather are tested for impairment
annually, or more frequently if events or changes in circumstances indicate that the asset might be
impaired. The impairment test, which is performed in April of each year, involves comparing the
fair value of the intangible asset with its carrying amount. If the carrying amount exceeds the
fair value of the intangible asset, an impairment loss is recognized in an amount equal to that
excess. No impairment charges for indefinite life intangible assets have been recorded in any
period presented.
Revenue Recognition
Net sales consist primarily of sales of products, software licenses, and services (including
installation, training, other consulting services, and extended maintenance contracts on our
products). Dionex recognizes revenue when persuasive evidence of an arrangement exists, the product
has been delivered or service has been performed, the sales price is fixed or determinable, and
collection is reasonably assured. Delivery of the product is generally considered to have occurred
when shipped. Shipping charges billed to customers are included in net sales, and the related costs
are included in cost of sales.
In all cases, the portion of revenue allocated to any undelivered elements is deferred until those
items are delivered to the customer or the services are performed. Delivery of the product is
generally considered to have occurred when shipped. Undelivered elements in our sales arrangements,
which are not considered to be essential to the functionality of a product, generally include
maintenance, installation services, and/or training that are delivered after the related products
have been delivered. Product installation consists of system set-up, calibration and basic
functionality training and generally requires one to three days depending on the product. The fair
value of these product installation services is calculated by applying standard service billing
rates to the estimate of the number of hours to install a specific product based on historical
experience. These estimated hours for product installation have historically been accurate and
consistent from product to product. The fair value of training services are based on the price
charged when the element is sold separately or, if not sold separately, when the price is
established by authorized management. However, to the extent these estimates were to reflect
unfavorable variability, our ability to maintain objective reliable evidence of fair value for such
element could be impacted, which in turn could delay the recognition of the revenue currently
allocated to the delivered elements. For sales of extended maintenance contracts with a separate
contract price, Dionex defers revenue equal to the separately stated price and recognize revenue on
a straight-line basis over the contract period.
33
Table of Contents
Sales from products are typically not subject to rights of return and, historically, actual
sales returns have not been significant. Dionex sells products through its direct sales force and
through distributors and resellers. Sales through distributors and resellers are recognized as
revenue upon sale to the distributor or reseller as these sales are considered to be final and no
right of return or price protection exists. Customer acceptance is generally limited to performance
under our published product specifications. When additional customer acceptance conditions apply,
all revenue related to the sale is deferred until acceptance is obtained.
In accordance with the software revenue guidance, Dionex recognizes revenue from perpetual software
licenses at the inception of the license term assuming all revenue recognition criteria have been
met. Dionex uses the residual method to allocate revenue to software licenses at the
inception of the license term when vendor specific objective evidence (“VSOE”) of fair value for
the undelivered elements exists, such as software installations, and all other revenue recognition
criteria have been satisfied. If Dionex cannot objectively determine the fair value of any
undelivered elements included in these multiple-element arrangements, revenue is deferred until all
elements are delivered, or until fair value can be objectively determined for the remaining
undelivered elements.
Product Warranty
The sales of Dionex products generally include a limited one-year warranty on product quality. The
estimated cost of product warranty claims is accrued at the time the sale is recognized, based on
historical experience. While Dionex believes that historical experience provides a reliable basis
for estimating such warranty cost, unforeseen quality issues or component failure rates could
result in future costs in excess of such estimates, or alternatively, improved quality and
reliability in our products could result in actual expenses that are below those currently
estimated.
Advertising
All advertising costs are expensed as incurred and are included in selling, general and
administrative in the consolidated statements of income. Advertising expenses were $5.6 million,
$4.4 million, and $5.2 million for fiscal 2010, 2009, and 2008, respectively.
Stock-based Compensation Plans
All share-based payments to employees are recognized as expense in the statements of income based
on their fair values. The fair value of each option on the date of grant is estimated using the
Black-Scholes option-pricing model using the single option approach for options granted after
June 30, 2005. Dionex recognizes the expense ratably on a straight-line basis over the requisite
service period. The stock-based compensation expense recognized in the consolidated statements of
operations is based on awards that ultimately are expected to vest; therefore, the amount of
expense has been reduced for estimated forfeitures. This accounting standard requires forfeitures
to be estimated at the time of grant and revised, if necessary, in subsequent periods if actual
forfeitures differ from those estimates. Forfeitures were estimated based on historical experience.
If actual results differ significantly from these estimates, stock-based compensation expense and
Dionex’s results of operations could be materially impacted. In addition, if Dionex employs
different assumptions in the application of this accounting standard, the compensation expense that
Dionex records in the future periods may differ significantly from what Dionex has recorded in the
current period.
Common Stock Repurchases
Dionex has an ongoing authorization, amended in October 2008, from Dionex’s Board of Directors to
repurchase shares of our common stock in the open market or in private transactions. For each share
repurchased, Dionex reduces the common stock account by the average value per share reflected in
the account prior to the repurchase with the excess allocated to retained earnings. Dionex
currently retires all shares upon repurchase.
In fiscal 2010, Dionex repurchased 775,644 shares of our common stock on the open market for $54.9
million (an average of $70.77 per share), compared with 791,517 shares repurchased for $44.2
million (an average of $55.88 per share) in 2009 and 928,131 shares repurchased for $70.3 million
(an average of $75.74 per share) in fiscal 2008.
Translation of Foreign Currency
Foreign subsidiaries designate the local currency as their functional currency. Translation
adjustments for their assets and liabilities into U.S. dollars at the balance sheet dates and the
results of their operations using average rates for the year are included as a component of
accumulated other comprehensive income.
34
Table of Contents
Derivative Securities
Starting in fiscal 2010, Dionex adopted certain recently issued accounting standards related to
derivative instruments and hedging. The adoption requires additional disclosures about the
objectives and strategies for using derivative instruments, the accounting for the derivative
instruments and related hedged items and the effect of derivative instruments and related hedged
items on the consolidated financial statements, as well as the level of derivative activity Dionex
engages in. The adoption had no impact on Dionex’s consolidated financial statements. All
derivatives, whether designated in hedging relationships or not, are required to be recorded on the
consolidated balance sheets at fair value as either assets or liabilities. If the derivative is
designated as a fair-value hedge, the changes in the fair value of the derivative and of the hedged
item attributable to the hedged risk are recognized in earnings. If the derivative is designated as
a cash flow hedge or net investment hedge, the effective portions of changes in the fair value of
the derivative are recorded in other comprehensive income and are recognized in earnings when the
hedged item affects earnings; ineffective portions of changes in fair value are recognized in
earnings.
Dionex operates on a global basis and is exposed to foreign currency exchange rate fluctuations in
the normal course of its business. As part of its risk management strategy, Dionex uses derivative
instruments to manage exposures to foreign currency. The objective is to offset gains and losses
resulting from these exposures with losses and gains on the derivative contracts used to hedge
them, thereby reducing volatility of earnings or protecting fair value of assets and liabilities.
Dionex does not have any leveraged derivatives nor use derivative contracts for speculative
purposes. Currently, Dionex designates its derivatives as hedges of the foreign currency exposure
of a net investment in a foreign operation (‘‘net investment hedges’’). Dionex considers the impact
of its counterparties’ credit risk on the fair value of the contracts as well as the ability of
each party to execute under the contracts. Dionex also assesses and documents, both at the hedges’
inception and on a quarterly basis, whether the derivatives that are used in hedging transactions
are highly effective in offsetting changes in fair values or cash flows associated with the hedged
items. For derivatives not designated as hedging instruments, Dionex categorizes those economic
hedges as other derivatives.
Net Investment Hedge
Dionex uses a $10 million cross-currency swap arrangement for Japanese yen designated as a net
investment hedge to hedge the exchange rate exposure of our net investment in our Japanese
subsidiary. During fiscal 2010, the arrangement was extended to March 2012. Prior to January 1,
2008, this derivative instrument did not qualify for net investment hedge accounting and was deemed
to be an ineffective hedge instrument because, at the inception of the hedge transaction, there was
no formal documentation of the hedging relationship and our risk management objective and strategy
for undertaking the hedge. Therefore, Dionex marked to market the decrease in value of
approximately $1.0 million for the six months ended December 31, 2007 and this amount was recorded
in other expense, net. Starting on January 1, 2008, Dionex determined that the cross-currency swap
qualified as a net investment hedge in which the effective portion of the gain or loss on the
derivative instruments is reported in accumulated other comprehensive income as part of the foreign
currency translation adjustment. Dionex evaluates the need to redesignate the hedging relationship
on a quarterly basis.
Other Derivatives
Other derivatives not designated as hedging instruments consist primarily of foreign exchange
forward contracts with high quality financial institutions to manage our exposure to the impact of
fluctuations in foreign currency exchange rates on our intercompany receivables balances. Principal
hedged currencies include the Euro, Japanese yen, Australian dollar and Canadian dollar. The
periods of these forward contracts is approximately 30 days and have varying notional amounts that
are intended to be consistent with changes in the underlying exposures and require Dionex to
exchange foreign currencies for U.S. dollars at maturity.
Total gross notional amounts for outstanding derivatives were as follows as of June 30:
| (in thousands) | 2010 | 2009 | ||||||
Currency forwards: |
||||||||
Euro |
$ | 17,659 | $ | 9,785 | ||||
Japanese yen |
4,282 | 3,938 | ||||||
Australian dollar |
791 | 857 | ||||||
Canadian dollar |
853 | 635 | ||||||
Currency swaps |
10,000 | 10,000 | ||||||
Total |
$ | 33,585 | $ | 25,215 | ||||
35
Table of Contents
The following table shows derivative instruments measured at gross fair value and balance sheet
location on the consolidated balance sheet as of June 30, 2010:
| Accrued | Other Long- | |||||||
| (In thousands) | Liabilities | Term Liabilities | ||||||
Derivatives designated as hedging instruments |
||||||||
Currency swaps |
$ | — | $ | 3,390 | ||||
Derivatives not designated as hedging instruments |
||||||||
Currency forwards |
$ | 290 | $ | — | ||||
The following table shows the effect of derivative instruments designated as hedging instruments
and not designated as hedging instruments in the consolidated statements of operations in fiscal
years ended June 30:
| (In thousands) | 2010 | 2009 | ||||||
Derivatives designated as hedging instruments |
||||||||
Net investment hedges |
||||||||
Unrealized gain/ (loss) recognized in other comprehensive income on derivatives |
$ | (1,165 | ) | $ | (1,126 | ) | ||
Realized gain/ (loss) reclassified from other comprehensive income into income |
$ | — | $ | — | ||||
Derivatives not designated as hedging instruments |
||||||||
Currency forwards |
||||||||
Gain included other income/(expense), net |
$ | 878 | $ | 396 | ||||
Comprehensive Income
Comprehensive income consists of two components, net income and other comprehensive income. Other
comprehensive income refers to revenue, expenses, gains and losses that under Generally Accepted
Accounting Principles (“GAAP”) are recorded as an element of stockholders’ equity but are excluded
from net income. Dionex’s other comprehensive income consists of foreign currency translation
adjustments from those subsidiaries not using the U.S. dollar as their functional currency and
unrealized gains and losses on available-for-sale securities, and net deferred gains and losses on
certain derivative instruments accounted for as net investment hedges.
Recently Adopted Accounting Pronouncements
In June 2009, a new accounting standard was issued that establishes the hierarchy of GAAP that are
to be used as the source of authoritative accounting principles recognized by the Financial
Accounting Standards Board (“FASB”) for non-governmental entities in preparation of financial
statements in conformity with GAAP in the United States. This standard was effective for interim
and annual periods ending after September 15, 2009. The adoption of this standard did not have a
material effect on our consolidated financial statements.
In February 2010, the FASB issued a new accounting standard to remove the requirement of disclosing
the date through which subsequent events have been evaluated in order to remedy potential conflicts
with other requirements. However, in accordance with this new accounting standard and existing SEC
rules, subsequent events are still required to be evaluated through the date the financial
statements are issued.
In April 2009, the FASB issued three accounting standards that were intended to provide additional
application guidance and enhance disclosures about fair value measurements and impairments of
securities. The provisions included in these standards were intended to (i) clarify the objective
and method of fair value measurement even when there has been a significant decrease in market
activity for the asset being measured, (ii) establish a new model for measuring
other-than-temporary impairments for debt securities, including establishing criteria for when to
recognize a write-down through earnings versus other comprehensive income and (iii) require fair
value disclosures in interim periods for all financial instruments. Dionex adopted these accounting
standards in the first quarter of fiscal 2010 and it did not have a material impact on our
consolidated financial position and results of operations but did expand our associated
disclosures.
36
Table of Contents
In December 2007, the FASB issued a new accounting standard related to noncontrolling interests.
The new standard establishes accounting and reporting standards for ownership interests in
subsidiaries held by parties other than the parent, the amount of consolidated net income
attributable to the parent and to the noncontrolling interest, changes in a parent’s ownership
interest, and the valuation of retained noncontrolling equity investments when a subsidiary is
deconsolidated. The new standard also establishes disclosure requirements that clearly identify and
distinguish between the interests of the parent and the interests of the noncontrolling owners.
Dionex adopted this new accounting standard in the first quarter of fiscal 2010, which impacted
certain captions previously used on the Consolidated Statements of Income, largely identifying net
income including noncontrolling interests and net income attributable to Dionex. Certain captions
on the Consolidated Balance Sheets and Consolidated Statements of Cash flows have also changed.
In
December 2007, the FASB issued a new accounting standard related to noncontrolling
interests in subsidiaries to establish accounting and reporting standards for the noncontrolling
interest in a subsidiary and for the deconsolidation of a subsidiary. It clarifies that a
noncontrolling interest in a subsidiary, which is sometimes referred to as a minority interest, is
a third-party ownership interest in the consolidated entity that should be reported as a component
of equity in the consolidated financial statements. Among other requirements, the new guidance
requires the consolidated statement of income to be reported at amounts that include the amounts
attributable to both the parent and the noncontrolling interest. The new guidance also requires
disclosure on the face of the consolidated statement of income of the amounts of consolidated net
income attributable to the parent and to the noncontrolling interest. This accounting standard is
effective for financial statements issued for fiscal years, and interim periods within those fiscal
years, beginning on or after December 15, 2008. Effective July 1, 2009, we adopted the accounting
standard related to noncontrolling interests in subsidiaries. In addition, the presentation and
disclosure requirements of the ASU have been applied retrospectively to our consolidated balance
sheet as of June 30, 2009, and our consolidated statements of income, stockholders’ equity and
comprehensive income, and cash flows for the years ended June 30, 2009 and 2008.
In December 2007, the FASB issued a new accounting standard related to business combinations. The
new standard expands the definition of a business and a business combination; requires recognition
of assets acquired, liabilities assumed, and contingent consideration at their fair value on the
acquisition date with subsequent changes recognized in earnings; requires acquisition-related
expenses and restructuring costs to be recognized separately from the business combination and
expensed as incurred; requires in-process research and development to be capitalized at fair value
as an indefinite lived intangible asset; and requires that changes in accounting for deferred tax
asset valuation allowances and acquired income tax uncertainties after the measurement period be
recognized as a component of provision for taxes. The new standard also establishes disclosure
requirements to enable the evaluation of the nature and financial effects of the business
combination. In April 2009, the FASB issued a new standard that clarified the accounting for
pre-acquisition contingencies. Dionex adopted these new business combination standards in the first
quarter of fiscal 2010.
In April 2008, the FASB issued a new accounting standard that amends the factors that should be
considered in developing renewal or extension assumptions used for purposes of determining the
useful life of a recognized intangible asset. This standard was intended to improve the consistency
between the useful life of a recognized intangible asset and the period of expected cash flows used
to measure the fair value of the asset and other U.S. GAAP. Dionex adopted this accounting
standard in the first quarter of fiscal 2010 and it did not have a material impact on our
consolidated financial statements.
In March 2008, the FASB issued a new accounting standard related to disclosures about derivative
instruments and hedging activities. This standard enhances financial disclosure by requiring that
objectives for using derivative instruments be described in terms of underlying risk and accounting
designation in the form of tabular presentation, requiring transparency with respect to the
entity’s liquidity from using derivatives, and cross-referencing an entity’s derivative information
within its financial footnotes. Dionex adopted this standard in the first quarter of fiscal 2010
and it did not have a material impact on our consolidated financial position and results of
operations but did expand our disclosures about derivative instruments and hedging activities.
In October 2009, the FASB issued two new accounting standards for revenue recognition with multiple
deliverable revenue arrangement to provide for (i) updated guidance on whether multiple
deliverables exist, how the deliverables in an arrangement should be separated, and the
consideration allocated; (ii) require a vendor to allocate revenue in an arrangement as determined
by vendor specific objective evidence of selling price (VSOE), if it exists, otherwise third-party
evidence of selling price. If neither exists for a deliverable, the a vendor shall use its best
estimate of the selling price for that deliverable; (iii) require a vendor to allocate revenue
using the relative selling price method; and (iv) require expanded qualitative and quantitative
disclosures. In October 2009, the FASB also issued a new accounting standard for certain revenue
arrangements that include software elements. This standard requires that tangible products that
contain software components and non-software components that function together to deliver the
tangible products essential functionality to be outside of the scope of the software revenue
guidance. Both accounting standards are effective for revenue arrangements entered into or
materially modified in fiscal years beginning on or after June 15, 2010, with early adoption
permitted. These new standards are required to be adopted in the first quarter of fiscal 2011.
Dionex does not expect these new standards to have a material impact our consolidated financial
statements.
37
Table of Contents
In January 2010, the FASB issued a new accounting standard to amend the disclosure
requirements related to recurring and nonrecurring fair value measurements. The guidance requires
new disclosures on the transfers of assets and liabilities between Level 1 and Level 2 of the fair
value measurement hierarchy, including the reasons and the timing of the transfers. Additionally,
the guidance requires a roll forward of activities on purchases, sales, issuance, and settlements
of the assets and liabilities measured using significant unobservable inputs (Level 3 fair value
measurements). The guidance became effective for us with the reporting period beginning January 1,
2010, except for the disclosure on the roll forward activities for Level 3 fair value measurements,
which will become effective in the first quarter of fiscal 2011. Other than requiring additional
disclosures, adoption of this new standard is not expected to have a material impact on our
financial statements.
Note 2: Earnings Per Share
Dionex calculates basic earnings per share using net income and the weighted average number of
common shares outstanding during the reporting period. The diluted earnings per share calculation
includes any dilutive effect of the number of common shares that would be issued assuming
conversion of all potentially dilutive securities outstanding under the treasury stock method.
The reconciliation of the numerators and denominators of the basic and diluted earnings per share
calculations was as follows:
| Year Ended June 30 | ||||||||||||
| (in thousands, except per share amounts) | 2010 | 2009 | 2008 | |||||||||
Numerator: |
||||||||||||
Net income attributable to Dionex |
$ | 59,056 | $ | 55,455 | $ | 52,800 | ||||||
Denominator: |
||||||||||||
Shares used to compute net income per common share — basic |
17,643 | 17,903 | 18,506 | |||||||||
Effect of dilutive stock awards |
340 | 325 | 566 | |||||||||
Shares used to compute net income per common share — diluted |
17,983 | 18,228 | 19,072 | |||||||||
Basic earnings per share attributable to Dionex |
$ | 3.35 | $ | 3.10 | $ | 2.85 | ||||||
Diluted earnings per share attributable to Dionex |
$ | 3.28 | $ | 3.04 | $ | 2.77 | ||||||
Antidilutive common equivalent shares related to stock
options excluded from the calculation of diluted shares |
416 | 627 | 349 | |||||||||
Note 3: Fair Value Measurements
In accordance with the accounting standards for fair value measurements and disclosures, assets and
liabilities are measured at fair value on a recurring basis as of June 30, 2010 and 2009. Dionex
applies the following fair value hierarchy, which prioritizes the inputs used to measure fair value
into three levels and bases the categorization within the hierarchy upon the lowest level of input
that is available and significant to the fair value measurement:
| • | Level 1 input utilize observable data such as quoted prices in active markets for identical assets or liabilities that the reporting entity has the ability to access at the measurement date. |
| • | Level 2 inputs utilize data points other than quoted prices in active markets that are observable for the asset or liability, either directly or indirectly. |
| • | Level 3 inputs utilize unobservable data points for the asset of liability in which there is little or no market data, which require the reporting entity to develop its own assumptions. |
The following table summarizes our assets and liabilities measured at fair value on a recurring
basis:
| Fair Value Measurements as of June 30, 2010 | ||||||||||||||||
| (in thousands) | Total | Level 1 | Level 2 | Level 3 | ||||||||||||
Assets: |
||||||||||||||||
Money market (1) |
$ | 73 | $ | 73 | $ | — | $ | — | ||||||||
Marketable securities (2) |
7 | 7 | — | — | ||||||||||||
Equity indexed derivatives (2) |
441 | — | 441 | — | ||||||||||||
Total |
$ | 521 | $ | 80 | $ | 441 | $ | — | ||||||||
Liabilities: |
||||||||||||||||
Foreign currency contracts (3) |
$ | 3,680 | $ | — | $ | 3,680 | $ | — | ||||||||
38
Table of Contents
| Fair Value Measurements as of June 30, 2009 | ||||||||||||||||
| Total | Level 1 | Level 2 | Level 3 | |||||||||||||
Assets: |
||||||||||||||||
Money market (1) |
$ | 2,998 | $ | 2,998 | $ | — | $ | — | ||||||||
Marketable securities (2) |
8 | 8 | — | — | ||||||||||||
Equity indexed derivatives (2) |
633 | — | 633 | — | ||||||||||||
Total |
$ | 3,639 | $ | 3,006 | $ | 633 | $ | — | ||||||||
Liabilities: |
||||||||||||||||
Foreign currency contracts (3) |
$ | 2,225 | $ | — | $ | 2,225 | $ | — | ||||||||
| (1) | Included in “cash and cash equivalents” in the consolidated balance sheet as of June 30, 2010 and 2009 | |
| (2) | Included in “short-term investments” in the consolidated balance sheet as of June 30, 2010 and 2009 | |
| (3) | Included in “other long-term liabilities” and/or “other accrued liabilities” in the consolidated balance sheets as of June 30, 2010 and 2009 |
Money market funds and available-for-sale investments are generally valued using quoted market
prices and therefore are classified within level 1 of the fair value hierarchy. Derivative
financial instruments are classified within level 2, as there is not an active market for each
contract, but the inputs used to calculate the value of the instruments are tied to active markets.
Investments designated as available-for-sale and certain derivative instruments are reported at
fair value, with unrealized gains and losses, net of tax, included in stockholders’ equity.
Realized gains and losses from the sale of these instruments (if any) are recorded in earnings.
Note 4: Short-Term Investments
Short-term investments are recorded at their fair value. The difference between the fair value and
amortized cost of short-term investments classified as
available-for-sale (“AFS”) securities is recorded
in other comprehensive income, net of deferred taxes. Dionex does not hold any auction-rate
securities. As of June 30, 2010 and 2009, short-term investments included an equity indexed
derivative totaling $441,000 and $633,000, respectively.
The aggregate market value, cost basis, and gross unrealized gains and losses of short-term
investments classified as AFS securities were as follows:
| Gross Unrealized | ||||||||||||
| (in thousands) | Cost | Gain / (Loss) | Fair Value | |||||||||
June 30, 2010, Marketable securities |
$ | 8 | $ | (1 | ) | $ | 7 | |||||
June 30, 2009, Marketable securities |
$ | 8 | $ | — | $ | 8 | ||||||
Investments with maturities greater than three months, but less than one year are classified as
short-term investments.
39
Table of Contents
Note 5: Balance Sheet Details
Balance sheet details were as follows as of June 30:
| (in thousands) | 2010 | 2009 | ||||||
Inventories: |
||||||||
Finished goods |
$ | 23,788 | $ | 19,070 | ||||
Work in process |
1,781 | 1,119 | ||||||
Raw materials and subassemblies |
11,889 | 11,085 | ||||||
Total inventories |
$ | 37,458 | $ | 31,274 | ||||
| (in thousands) | 2010 | 2009 | ||||||
Property, plant and equipment, net: |
||||||||
Land |
$ | 25,098 | $ | 24,533 | ||||
Buildings and improvements |
48,396 | 47,060 | ||||||
Machinery, equipment and tooling |
50,411 | 40,960 | ||||||
Furniture and fixtures |
12,194 | 10,884 | ||||||
Construction-in-progress |
13 | 2,408 | ||||||
Total property, plant and equipment, gross |
136,112 | 125,845 | ||||||
Less: accumulated depreciation |
(60,050 | ) | (53,918 | ) | ||||
Total property, plant and equipment, net |
$ | 76,062 | $ | 71,927 | ||||
Depreciation expense was $9.8 million, $8.5 million and $7.6 million in fiscal 2010, 2009, and
2008, respectively.
| (in thousands) | 2010 | 2009 | ||||||
Accrued liabilities: |
||||||||
Accrued payroll and related expenses |
$ | 22,851 | $ | 18,267 | ||||
Other accrued liabilities |
11,129 | 12,955 | ||||||
Total accrued liabilities |
$ | 33,980 | $ | 31,222 | ||||
Note 6: Acquisitions
Dionex adopted the newly issued accounting standard for business combinations in the first quarter
of fiscal 2010. This standard requires an acquiring company to measure all assets acquired and
liabilities assumed, including contingent considerations and all contractual contingencies, at fair
value as of the acquisition date. In addition, an acquiring company is required to capitalize IPR&D
and either amortize it over the life of the product or write it off if the project is abandoned or
impaired. This accounting standard is applicable to acquisitions completed by Dionex in the first
quarter of fiscal 2010. Previous standards generally required post-acquisition adjustments related
to business combination deferred tax asset valuation allowances and liabilities for uncertain tax
positions to be recorded as an increase or decrease to goodwill. This new accounting standard does
not permit this accounting and generally requires any such changes to be recorded in current period
income tax expense. Thus, all changes to valuation allowances and liabilities for uncertain tax
positions established in acquisition accounting, whether the business combination was accounted for
under previous standards or under the newly issued accounting standard, will be recognized in
current period income tax expense, if such changes are made after the initial purchase accounting
allocation period (not to exceed one year).
In September 2009, Dionex acquired certain assets and liabilities from the Life Sciences Tools and
Laboratory Services business divisions of ESA Biosciences, Inc. This acquisition qualified as a
business combination under the accounting standards and was integrated into our Life Sciences
Business Unit (“LSBU”) operating segment increasing our portfolio of High Performance Liquid
Chromatography (“HPLC”) solutions and expanding our presence in the life sciences market. Purchase
price paid was $21.1 million in cash, plus the assumption of certain liabilities of $486,000.
Acquisition related costs charged to selling, general and administrative were approximately
$350,000. Dionex recorded $7.0 million to goodwill (deductible for tax purposes) and $7.3 million
to identifiable intangible assets. In fiscal 2010, ESA products resulted in net sales of
approximately $12.8 million and the impact on our earnings for the fiscal year was not material to
our consolidated financial statement.
In November 2008, Dionex acquired certain assets and liabilities from Caliper Life Sciences,
Inc.’s sample preparation line of products (collectively known as “AutoTrace”) to complement our
analytical solutions in both Chemical Analysis Business Unit (“CABU”) and LSBU operating segments.
This acquisition qualified as a business combination and included the AutoTrace developed
technology, trade name, inventories, non-competition covenant, customer list, and its related
existing servicing obligations as of the purchase date. Purchase price paid was approximately $5.0
million in cash, plus the assumption of certain liabilities of $65,000 and acquisition related
costs of $23,000. Dionex recorded $2.4 million to goodwill (deductible for tax purposes) and $2.7
million to identifiable intangible assets.
40
Table of Contents
In July 2008, Dionex acquired a 100% ownership interest in a Swedish company, in which Dionex
previously held a 30% equity interest with a carrying amount of $760,000 at June 30, 2008, using a
combination of cash and earn-out payment arrangements. The total purchase consideration for the
incremental 70% ownership interest was approximately $1.5 million, excluding contingent earn-out
payments based on a percentage of net income in 2009 through 2011. This acquisition allowed Dionex
to take control of this Swedish distributor for the purpose of further expanding the business and
operations. Approximately $952,000 of the purchase price consideration was paid during fiscal 2009,
with the remaining $527,000 payable on July 1, 2011 and included within other long-term liabilities
as of June 30, 2010. As a result of this acquisition, the $760,000 equity interest previously included
within other assets was eliminated, and Dionex recorded $1.5 million to goodwill (deductible for
tax purposes) and $830,000 to identifiable intangible assets.
Results of operations for the acquisitions have been included in our consolidated financial
statements subsequent to the date of acquisition, and have not been presented on a pro forma basis
as the revenue and income from operations are not material to our consolidated results. Pro forma
results of operations in accordance with authoritative guidance for prior periods have not been
presented because the effect of the acquisition was not material to our prior period consolidated
financial results.
Note 7: Goodwill and Intangible Assets
Changes in the carrying amount of goodwill were as follows for the fiscal years ended June 30:
| (in thousands) | 2010 | 2009 | ||||||
Balance, beginning of the fiscal year |
$ | 29,354 | $ | 26,670 | ||||
Additions due to business combinations |
7,032 | 3,974 | ||||||
Foreign currency translation impact |
(1,373 | ) | (1,290 | ) | ||||
Balance, end of the fiscal year |
$ | 35,013 | $ | 29,354 | ||||
After completing our annual impairment reviews in April of fiscal 2010, 2009, and 2008, we
concluded that goodwill was not impaired in any year.
Intangible assets (net of accumulated amortization) were as follows as of June 30:
| 2010 | 2009 | |||||||||||||||||||||||
| Carrying | Accumulated | Carrying | Accumulated | |||||||||||||||||||||
| (in thousands) | Amount | Amortization | Net | Amount | Amortization | Net | ||||||||||||||||||
Patents and trademarks |
$ | 8,788 | $ | (2,580 | ) | $ | 6,208 | $ | 7,088 | $ | (1,978 | ) | $ | 5,110 | ||||||||||
Developed technology |
14,893 | (10,456 | ) | 4,437 | 11,504 | (10,327 | ) | 1,177 | ||||||||||||||||
Customer relationships |
5,119 | (1,905 | ) | 3,214 | 3,391 | (1,172 | ) | 2,219 | ||||||||||||||||
Total |
$ | 28,800 | $ | (14,941 | ) | $ | 13,859 | $ | 21,983 | $ | (13,477 | ) | $ | 8,506 | ||||||||||
As a result of the acquisition from ESA Bioscience Inc. in fiscal 2010, Dionex recorded trade name
of $1.7 million, which is not subject to amortization and is included in Patent and trademarks;
developed technology of $4.0 million with a weighted average life of 7 years; and other intangible
assets of $1.6 million with a weighted average life of 6.5 years. As a result of the AutoTrace
acquisition in fiscal 2009, Dionex recorded the trade name of approximately $1.1 million, which is
not subject to amortization and is included in Patent and trademarks; developed technology of $1.3
million with a weighted average life of 7 years; and other intangible assets of $230,000 with a
weighted average life of 6.1 years.
Amortization expense related to all finite intangible assets was $2.1 million, $1.2 million and
$1.1 million for fiscal 2010, 2009, and 2008, respectively. Estimated future amortization
expense related to finite lived intangible assets as of June 30, 2010 is as follows:
| Remaining | ||||
| Amortization | ||||
| (in thousands) | Expense | |||
Year Ending June 30, |
||||
2011 |
$ | 2,746 | ||
2012 |
2,431 | |||
2013 |
1,787 | |||
2014 |
1,323 | |||
2015 and thereafter |
2,634 | |||
Total |
$ | 10,921 | ||
41
Table of Contents
Note 8: Financing Arrangements
Dionex has unsecured lines of credit with various domestic and foreign banks, which have been used
primarily to minimize our exposure to foreign currency fluctuations and to fund share repurchases.
Our lines of credit totaled $25.7 million and $28.8 million
as of June 30, 2010 and 2009,
respectively. Borrowings in each country bear interest at local reference rates, which ranged from
1.9% to 9.8% at June 30, 2010. There was $3.1 million and $64,000 outstanding under these lines as of
June 30, 2010 and 2009, respectively. Such line of credit agreement impose certain financial
restrictions relating to cash dividends, working capital and tangible net worth and expires
December 31, 2011. Dionex was in compliance with these covenants as of June 30, 2010 and 2009.
One of our foreign subsidiaries discounts trade notes receivable with banks. Total notes receivable
discounted was approximately
$8.2 million and $12.2 million in fiscal 2010 and 2009, respectively. The uncollected balances of
notes receivable due to the discounting banks as of June 30, 2010 and 2009 were approximately $2.3
million and $3.1 million, respectively. Dionex has a contingent liability to repurchase these
notes under certain conditions. Dionex has determined that the carrying amount of the contingent
liability under this guarantee was insignificant as of June 30, 2010 and 2009 based on its past
experience of discounting trade notes receivable.
Note 9: Warranty
Dionex accrues estimated product warranty costs at the time of sale, which are included in cost of
sales in the consolidated statements of operations. While Dionex engages in extensive product
quality programs and processes, including actively monitoring and evaluating the quality of its
component supplies, our warranty obligation is affected by product failure rates, material usage
and service delivery costs incurred in correcting a product failure. The amount of the accrued
warranty liability is based on historical information, such as past experience, product failure
rates, number of units repaired and estimated costs of material and labor. The liability is
reviewed for reasonableness at least quarterly.
The
changes in the warranty provision were as follows for the fiscal
years ended June 30:
| (in thousands) | 2010 | 2009 | 2008 | |||||||||
Balance, beginning of the fiscal year |
$ | 3,028 | $ | 3,444 | $ | 2,875 | ||||||
Additions |
3,655 | 4,907 | 5,954 | |||||||||
Charged (credits) to other accounts (1) |
(137 | ) | (249 | ) | 296 | |||||||
Settlements |
(4,014 | ) | (5,074 | ) | (5,681 | ) | ||||||
Balance, end of the fiscal year |
$ | 2,532 | $ | 3,028 | $ | 3,444 | ||||||
| (1) | Effects of exchange rate changes. |
Note 10: Stock-Based Compensation
Dionex’s Board of Directors authorized the 2004 Equity Incentive Plan (the “2004 Plan”). The 2004
Plan was last Amended and Restated in fiscal 2008. Shares reserved for future issuance under the
2004 Plan may be used for grants of stock options (“options”), restricted stock units (“RSU”), and
other types of awards. Participation in the 2004 Plan is limited to consultants and employees,
including officers and directors. Options granted under the 2004 Plan are either incentive or
nonqualified stock options and generally become exercisable in increments over a period of four
years from the date of grant and expire generally ten years from the grant date. RSU generally vest
over a four-year period for directors and non-officer employees and a five-year period for
officers. The delivery of the shares for non-officer employees occurs on the vesting date of the
RSU. The delivery of shares for directors and officers occurs on the earlier of: (i) five years
from grant date of the RSU, or (ii) termination of service.
The Board of Directors authorizes the granting of options, RSU and other type of awards, and
determines the employees and consultants to whom options, RSU, or other stock awards are to be
granted, the number of shares, term, vesting schedule and other terms and conditions of the
options, RSU or other stock awards. The exercise prices of the options shall not be less than the
fair market value of common stock on the date of grant. RSUs granted under the 2004 Plan generally
do not require a cash payment from a participant to whom an award is made. The fair value of RSUs
granted during the three year ended June 30, 2010, was determined based on the number of RSUs
granted and the quoted price of Dionex’s common stock on the date of grant.
Additionally, Dionex sponsors the Employee Stock Purchase Plan (the “ESPP”) in which eligible
employees may contribute up to 10% of their base compensation to purchase shares of common stock at
a price equal to 85% of the lower of the market value of the stock at the beginning or end of each
six-month offer period, subject to certain annual limitations. Dionex recognizes expense based on a
15% discount of the fair market value for those purchases.
42
Table of Contents
The consolidated statements of operations for fiscal 2010, 2009, and 2008 included the following
stock-based compensation expense related to options, RSUs, and ESPP:
| (in thousands) | 2010 | 2009 | 2008 | |||||||||
Cost of sales |
$ | 823 | $ | 794 | $ | 602 | ||||||
Selling, general and administrative expenses |
4,509 | 4,097 | 3,847 | |||||||||
Research and product development expenses |
1,451 | 1,703 | 1,490 | |||||||||
Tax benefit |
(2,171 | ) | (2,065 | ) | (1,919 | ) | ||||||
Total |
$ | 4,612 | $ | 4,529 | $ | 4,020 | ||||||
Restricted Stock Units
The following table summarizes RSU activity under the 2004 Plan for the fiscal years ended June 30:
| 2010 | 2009 | 2008 | ||||||||||||||||||||||
| Wtd. Avg. | Wtd. Avg. | Wtd. Avg. | ||||||||||||||||||||||
| Grant-Date | Grant-Date | Grant-Date | ||||||||||||||||||||||
| Shares | Fair Value | Shares(1) | Fair Value | Shares | Fair Value | |||||||||||||||||||
RSUs outstanding, beginning of fiscal year |
50,000 | $ | 62.56 | 5,000 | $ | 87.03 | — | $ | — | |||||||||||||||
Granted |
48,876 | 66.04 | 45,000 | 59.84 | 5,000 | 87.03 | ||||||||||||||||||
Released |
(7,600 | ) | 60.84 | — | — | — | — | |||||||||||||||||
Canceled |
(3,500 | ) | 73.45 | — | — | — | — | |||||||||||||||||
RSUs outstanding, end of fiscal year |
87,776 | $ | 64.21 | 50,000 | $ | 62.56 | 5,000 | $ | 87.03 | |||||||||||||||
| (1) | Total RSU granted previously reported excluded 25,000 RSU granted to our Chief Executive Officer in fiscal 2009. Such amounts have been added to the table above. |
The aggregate fair value of Dionex common stock on the date that the RSUs released was $0.6 million
in fiscal 2010 (no RSUs were released in fiscal 2009 and 2008). As of
June 30, 2010, there was $4.0
million in unrecognized compensation costs related to RSU granted under the 2004 Plan. These costs
are expected to be recognized over a weighted average period of 3.6 years.
Stock Options
The following table summarizes option activity under the 2004 Plan for fiscal years ended June 30:
| 2010 | 2009 | 2008 | ||||||||||||||||||||||
| Wtd. Avg. | Wtd. Avg. | Wtd. Avg. | ||||||||||||||||||||||
| Exercise | Exercise | Exercise | ||||||||||||||||||||||
| Shares | Price | Shares | Price | Shares | Price | |||||||||||||||||||
Options outstanding, beginning of fiscal year |
1,699,640 | $ | 53.02 | 1,792,708 | $ | 47.20 | 1,694,471 | $ | 41.91 | |||||||||||||||
Granted |
236,765 | 66.36 | 304,250 | 67.10 | 347,750 | 72.81 | ||||||||||||||||||
Exercised |
(406,753 | ) | 44.53 | (376,577 | ) | 36.35 | (180,073 | ) | 40.41 | |||||||||||||||
Canceled |
(55,025 | ) | 65.26 | (20,741 | ) | 59.35 | (69,440 | ) | 63.77 | |||||||||||||||
Options outstanding, end of fiscal year |
1,474,627 | $ | 57.05 | 1,699,640 | $ | 53.02 | 1,792,708 | $ | 47.20 | |||||||||||||||
Weighted average fair value of options granted during the year |
$ | 21.90 | $ | 20.77 | $ | 23.88 | ||||||||||||||||||
The
aggregate intrinsic value of outstanding stock options as of June 30, 2010 was $25.8 million.
The total intrinsic value of options exercised were $11.5 million, $7.3 million, and $6.9 million in
fiscal 2010, 2009, and 2008 respectively. As of June 30, 2010,
there was $9.2 million of total
unrecognized compensation cost related to stock options. These costs are expected to be recognized
over a weighted average period of 2.6 years.
The following table summarizes options outstanding that have vested or expected to vest and
exercisable as of June 30, 2010:
| Wtd. Avg. | Wtd. Avg. | Aggregate | ||||||||||||||
| Number of | Exercise | Remaining | Intrinsic Value | |||||||||||||
| Shares | Price | Contractual Term | Price | |||||||||||||
| (in years) | (in millions) | |||||||||||||||
Vested or expected to vest |
1,447,939 | $ | 56.89 | 6.29 | $ | 25.5 | ||||||||||
Exercisable |
950,192 | $ | 51.78 | 5.12 | $ | 21.6 | ||||||||||
Exercise and pre-vesting forfeiture assumptions based on analysis of historical data.
43
Table of Contents
Options outstanding and exercisable was as follows as of June 30, 2010:
| Options Outstanding | Options Exercisable | ||||||||||||||||||||||||
| Weighted Avg. | Weighted | Weighted Avg. | Weighted | ||||||||||||||||||||||
| Remaining | Avg. | Remaining | Avg. | ||||||||||||||||||||||
| Range of | Number | Contractual | Exercise | Number | Contractual | Exercise | |||||||||||||||||||
| Exercise Prices | Outstanding | Life (Yrs) | Price | Exercisable | Life (Yrs) | Price | |||||||||||||||||||
| $ | 23.98 – 39.47 | 248,503 | 2.22 | $ | 32.84 | 248,503 | $ | 32.84 | |||||||||||||||||
| 46.47 – 49.93 | 246,594 | 4.81 | 47.90 | 242,844 | 47.92 | ||||||||||||||||||||
| 53.38 – 54.86 | 303,686 | 6.92 | 53.62 | 211,022 | 53.55 | ||||||||||||||||||||
| 54.87 – 70.01 | 237,991 | 9.25 | 65.99 | 7,591 | 70.10 | ||||||||||||||||||||
| 70.32 – 72.56 | 247,933 | 7.08 | 72.42 | 166,983 | 72.41 | ||||||||||||||||||||
| 74.27 – 87.03 | 189,920 | 8.19 | 74.82 | 73,249 | 74.71 | ||||||||||||||||||||
| $ | 23.98 – 87.03 | 1,474,627 | 6.34 | $ | 57.05 | 950.192 | 5.12 | $ | 51.78 | ||||||||||||||||
As of June 30, 2010, 658,638 shares were available for future grants under the 2004 Plan.
The following assumptions were used to determine the fair value of the stock options using the
Black-Scholes-Merton options pricing model for the fiscal years ended June 30:
| 2010 | 2009 | 2008 | ||||||||||
Volatility |
33 – 34 | % | 29 – 35 | % | 28 – 29 | % | ||||||
Risk-free interest rate |
2.4 – 2.6 | % | 1.9 – 3.3 | % | 2.9 – 4.6 | % | ||||||
Expected life (in years) |
4.6 | 4.7 | 4.7 | |||||||||
Expected dividend |
$ | 0.00 | $ | 0.00 | $ | 0.00 | ||||||
Employee Stock Purchase Plan
The following table summarizes ESPP share activity under the 2004 Plan for the fiscal years ended
June 30:
| 2010 | 2009 | 2008 | ||||||||||
Number of shares issued during the year |
45,677 | 43,917 | 32,969 | |||||||||
Weighted average purchase price per share |
$ | 49.43 | $ | 49.57 | $ | 56.59 | ||||||
Weighted average fair value per award |
$ | 15.50 | $ | 8.75 | $ | 12.48 | ||||||
As of June 30, 2010, 545,656 shares were reserved for future issuances under the ESPP.
The following assumptions were used to determine the fair value
of ESPP shares for the fiscal years ended June 30:
| 2010 | 2009 | 2008 | ||||||||||
Volatility |
20 – 36 | % | 24 – 56 | % | 21 – 31 | % | ||||||
Risk-free interest rate |
0.2 – 0.3 | % | 0.5 – 2.0 | % | 2.1 – 4.8 | % | ||||||
Expected life (in years) |
0.50 | 0.50 | 0.50 | |||||||||
Expected dividend |
$ | 0.00 | $ | 0.00 | $ | 0.00 | ||||||
Determining Fair Value for Options and ESPP
| • | Valuation and amortization method – Dionex estimates the fair value of stock options granted using the Black-Scholes-Merton option – pricing formula and a single option award approach. This fair value is then amortized on a straight-line basis over the requisite service periods of the awards, which is generally the vesting period. |
| • | Expected Term – The expected term represents the period that our stock-based awards are expected to be outstanding and was determined based on historical experience of similar awards, giving consideration to the contractual terms of the stock-based awards, vesting schedules and expectations of future employee behavior as influenced by changes to the terms of our stock-based awards. |
| • | Expected Volatility – Our computation of expected volatility for fiscal 2010, 2009, and 2008 is based on a combination of historical and market-based implied volatility. |
| • | Risk-Free Interest Rate – The risk-free interest rate used in the Black-Scholes-Merton valuation method is based on the implied yield currently available on U.S. Treasury zero-coupon issues with an equivalent remaining term. |
| • | Expected Dividend – The expected dividend assumption is based on our current expectations about our anticipated dividend policy. |
44
Table of Contents
Note 11: Employee 401(k) Plan
Dionex has a 401(k) tax deferred savings plan covering most U.S. employees. Participants may
contribute up to 10% of their compensation and Dionex will make matching contributions limited to
5% of each participant’s compensation. Matching contributions vest in 25% increments each year.
Matching contributions made were $1.9 million, $1.8 million, and $1.6 million in fiscal 2010,
2009, and 2008, respectively.
Note 12: Comprehensive Income
Comprehensive income consists of two components, net income and other comprehensive income. Other
comprehensive income refers to revenue, expenses, gains and losses that under GAAP are recorded as
an element of stockholders’ equity but are excluded from net income. Dionex’s other comprehensive
income consists of foreign currency translation adjustments from those subsidiaries not using the
U.S. dollar as their functional currency and unrealized gains and losses on available-for-sale
securities, and net deferred gains and losses on certain derivative instruments accounted for as
net investment hedges.
The components of accumulated other comprehensive income (net of taxes), were as follows as of June
30:
| (in thousands) | 2010 | 2009 | 2008 | |||||||||
Cumulative foreign currency translation, net of tax |
$ | 6,733 | $ | 14,307 | $ | 24,118 | ||||||
Accumulated unrealized gain (loss) on available-for-sale securities, net of tax |
— | (1 | ) | 4 | ||||||||
Total accumulated other comprehensive income |
$ | 6,733 | $ | 14,306 | $ | 24,122 | ||||||
The components of other comprehensive income and related tax effects were as follows for the fiscal
years ended June 30:
| (in thousands) | 2010 | 2009 | 2008 | |||||||||
Foreign currency translation, net of tax |
$ | (7,574 | ) | $ | (9,815 | ) | $ | 13,042 | ||||
Changes in unrealized holding gain (loss) on AFS securities, net |
1 | (1 | ) | 4 | ||||||||
Total other comprehensive income (loss) |
$ | (7,573 | ) | $ | (9,816 | ) | $ | 13,046 | ||||
Foreign currency translation includes unrealized losses on fair value of net investment hedges of
$1.2 million, $1.1 million, and $0.6 million for the years ended
June 30, 2010, 2009, and 2008,
respectively. The aggregated tax effect related to the changes in fair value of net investment
hedges and foreign currency translation were $4.0 million,
$3.8 million, and $6.3 million for 2010,
2009, and 2008, respectively. The tax effect related to the changes in unrealized gain/loss on
available-for-sale securities were not significant for periods presented.
Note 13: Taxes on Income
The provision for taxes on income was as follows for the fiscal years ended June 30:
| (in thousands) | 2010 | 2009 | 2008 | |||||||||
Current: |
||||||||||||
Federal |
$ | 13,338 | $ | 15,420 | $ | 17,433 | ||||||
State |
1,137 | 1,755 | (1,481 | ) | ||||||||
Foreign |
12,367 | 9,128 | 10,197 | |||||||||
Total current |
26,842 | 26,303 | 26,149 | |||||||||
Deferred: |
||||||||||||
Federal |
925 | 2,734 | 105 | |||||||||
State |
(800 | ) | (560 | ) | 122 | |||||||
Foreign |
805 | (990 | ) | (1,171 | ) | |||||||
Total deferred |
930 | 1,184 | (944 | ) | ||||||||
Total taxes on income attributable to Dionex |
27,772 | 27,487 | 25,205 | |||||||||
Total taxes on income attributable to noncontrolling interest |
887 | 331 | 393 | |||||||||
Total taxes on income |
$ | 28,659 | $ | 27,818 | $ | 25,598 | ||||||
45
Table of Contents
The domestic and foreign components of income were as follows for the fiscal years ended June 30:
| (in thousands) | 2010 | 2009 | 2008 | |||||||||
Domestic |
$ | 59,188 | $ | 58,226 | $ | 57,830 | ||||||
Foreign |
27,640 | 24,716 | 20,175 | |||||||||
Total income attributable to Dionex |
$ | 86,828 | $ | 82,942 | $ | 78,005 | ||||||
Total income attributable to noncontrolling interest |
$ | 2,253 | $ | 966 | $ | 1,190 | ||||||
Total income |
$ | 89,081 | $ | 83,908 | $ | 79,195 | ||||||
Deferred income taxes reflect the net tax effects of temporary differences between the carrying
amounts of assets and liabilities for financial reporting purposes and the amounts used for income
tax purposes. Non-current deferred tax assets are included in “Other Assets” on the consolidated
balance sheets as of June 30, 2010 and 2009.
The components of the current and non-current deferred tax assets and liabilities was as follows
for the fiscal years ended June 30:
| (in thousands) | 2010 | 2009 | ||||||
Current deferred tax assets: |
||||||||
Accounting accruals deductible in different periods for tax purposes |
$ | 5,787 | $ | 6,192 | ||||
Stock-based compensation |
5,596 | 4,787 | ||||||
Other |
2,653 | 1,192 | ||||||
Total current deferred tax assets |
14,036 | 12,171 | ||||||
Non-current deferred tax assets: |
||||||||
Book depreciation in excess of tax |
— | 229 | ||||||
Accumulated translation adjustment |
1,758 | — | ||||||
Others |
35 | — | ||||||
Non-current deferred tax assets (1) |
$ | 1,793 | $ | 229 | ||||
Accelerated depreciation |
(2,768 | ) | (1,676 | ) | ||||
Tax amortization of intangibles in excess of book |
(4,645 | ) | (3,450 | ) | ||||
Accumulated translation adjustment |
— | (3,043 | ) | |||||
Others |
(781 | ) | — | |||||
Total deferred tax liabilities (2) |
(8,194 | ) | (8,169 | ) | ||||
Net deferred tax assets |
$ | 7,635 | $ | 4,231 | ||||
| (1) | Included in “Other assets” in the consolidated balance sheets as of June 30, 2010 and 2009 | |
| (2) | Included in “Deferred and other income taxes payable” in the consolidated balance sheets as of June 30, 2010 and 2009 |
The differences between the U.S. federal statutory income tax rate and Dionex’s effective tax rate
were as follows for the fiscal years ended June 30:
| 2010 | 2009 | 2008 | ||||||||||
U.S. federal statutory income tax rate |
35.0 | % | 35.0 | % | 35.0 | % | ||||||
State income taxes, net of Federal income tax effect |
(0.1 | ) | 0.8 | (1.3 | ) | |||||||
Extraterritorial income exclusion/Manufacturing deduction |
(1.1 | ) | (1.3 | ) | (1.4 | ) | ||||||
Taxes on foreign income |
(0.3 | ) | (2.0 | ) | 1.0 | |||||||
Other |
(1.5 | ) | 0.9 | (0.7 | ) | |||||||
Effective tax rate |
32.0 | % | 33.4 | % | 32.6 | % | ||||||
Income taxes paid were $20.7 million, $28.9 million, and $31.2 million during fiscal 2010, 2009,
and 2008, respectively.
Dionex has not provided for Federal income taxes on approximately $68.3 million of undistributed
earnings of certain foreign subsidiaries, which we intend to permanently reinvest in subsidiary
operations. If these earnings were distributed to us as the parent, foreign tax credits available
under current law would substantially eliminate the resulting Federal income tax liability.
Our total amount of unrecognized tax benefits as of June 30, 2010 was $8.0 million, of which $1.9
million, if recognized, would affect our effective tax rate compared to $14.1 million on June 30,
2009, of which $3.2 million, if recognized, would have affected our effective tax rate. The
liability for income taxes associated with uncertain tax positions is classified in deferred and
other income taxes payable.
46
Table of Contents
A reconciliation of the July 1, 2007 through June 30, 2010 amount of unrecognized tax benefits
is as follows:
| (in thousands) | 2010 | 2009 | 2008 | |||||||||
Balance, beginning of the fiscal year |
$ | 14,081 | $ | 14,232 | $ | 14,356 | ||||||
Increases (decreases) for tax positions of prior years |
(3,186 | ) | 1,601 | — | ||||||||
Increases (decreases) for tax positions of the current year |
272 | 1,699 | 3,104 | |||||||||
Decrease from expiration of statute of limitation |
(3,187 | ) | (3,451 | ) | (3,228 | ) | ||||||
Balance, end of the fiscal year |
$ | 7,980 | $ | 14,081 | $ | 14,232 | ||||||
Dionex records interest and penalties related to unrecognized tax benefits in income tax expense.
As of June 30, 2010, Dionex has approximately $1.7 million accrued for estimated interest related to
uncertain tax positions compared to approximately $2.0 million as of June 30, 2009. During the fiscal
year ended June 30 2010, we accrued a total of $541,000 in interest on these uncertain tax
positions. As of June 30, 2010, we had approximately $69,000 accrued for estimated penalties related
to uncertain tax positions compared to approximately $78,000 on June 30, 2009. During the fiscal
year ended June 30, 2010, we accrued no additional penalties on these uncertain tax positions.
As of June 30,2010, we have various unrecognized tax benefits for which it is reasonably possible that the statute of
limitations will expire in various jurisdictions with the next twelve months, of which $1.1 million will reduce our
effective tax rate.
Dionex is subject to audit by the Internal Revenue Service starting in fiscal years 2009 and onward
and California Franchise Tax Board for the fiscal years 2006 through the fiscal year 2009. As we
have operations in most other US states, other state tax authorities may assess deficiencies
related to prior year activities; however, the years open to assessment vary with each state.
Dionex also files income tax returns for non-US jurisdictions; the most significant of which are
Germany, Japan, the UK and Hong Kong. The years open to adjustment for Germany are for the fiscal
years 2004 through 2009, fiscal years 2004 through 2009 for the UK and Hong Kong and fiscal years
2003 through 2009 for Japan. We had R&D credits carryforward totaling $1.6 million as of June 30,
2010.
A number of years may elapse before an uncertain tax position is audited and ultimately settled. It
is difficult to predict the ultimate outcome or the timing of resolution for uncertain tax
positions. It is reasonably possible that the amount of unrecognized tax benefits could
significantly increase or decrease within the next twelve months. These changes could result from
the settlement of ongoing litigation, the completion of ongoing examinations, the expiration of the
statute of limitations, or other circumstances. At this time, an estimate of the range of the
reasonably possible change cannot be made. Consequently, Dionex cannot conclude on the range of
cash payments that will be made within the next twelve months associated with its uncertain tax
positions.
Note 14: Commitments and Contingencies
In July 2008, Dionex acquired a Swedish company using a combination of cash and post acquisition
earn-out payment arrangements. Under the purchase agreement, earn-out payments of 70% for fiscal
2009 and 30% for fiscal 2010 and 2011 as a percentage of the acquired company’s net income are
payable to the seller at the end of each fiscal year. Each earn-out payment is contingent upon
results of operations in which no payments were made related to fiscal 2010 and 2009.
Certain facilities and equipment are leased under non-cancelable operating leases. Dionex generally
pays taxes, insurance and maintenance costs on leased facilities and equipment. Rental expense for
all operating leases was $7.5 million, $7.6 million and $7.2 million in fiscal 2010, 2009, and
2008, respectively.
Minimum rental commitments under these non-cancelable operating leases as of June 30, 2010, are as
follows:
(In thousands)
Fiscal 2011 |
$ | 5,507 | ||
Fiscal 2012 |
2,897 | |||
Fiscal 2013 |
1,470 | |||
Fiscal 2014 |
800 | |||
Fiscal 2015 |
302 | |||
Thereafter |
1,360 | |||
Total |
$ | 12,336 | ||
Dionex enters into standard indemnification agreements with many of our customers and certain
other business partners in the ordinary course of business. These agreements include provisions for
indemnifying the customer against any claim brought by a third party to the extent any such claim
alleges that our product infringes a patent, copyright or trademark, or violates any other
proprietary rights of that third party. The maximum potential amount of future payments Dionex
could be required to make under these indemnification agreements is not estimable, however, Dionex
has not incurred any costs to defend lawsuits or settle claims related to these indemnification
agreements. No material claims for such indemnifications were outstanding as of June 30, 2010.
Dionex has not recorded any liabilities for these indemnification agreements as of June 30, 2010 or
2009.
47
Table of Contents
Note 15: Business Segment Information
The accounting standard for segment reporting establishes standards for reporting information about
operating segments in annual financial statements and requires selected information for those
segments to be presented in interim financial reports of public business enterprises. It also
establishes standards for related disclosures about products and services, geographic areas and
major customers. Our business activities, for which discrete financial information is available,
are regularly reviewed and evaluated by the chief operating decision maker (our Chief Executive
Officer). As a result of this evaluation, Dionex determined that it has two operating segments:
CABU and LSBU.
CABU sells ion chromatography and sample preparation products, chromatography data systems
software, services and related consumables. LSBU sells HPLC products, chromatography data systems
software, services and related consumables. These two operating segments are aggregated into one
reportable segment for financial statement purposes. Both operating segments have similar economic
characteristics; product processes; products and services; types and classes of customers; methods
of distribution and regulatory environments. Because of these similarities, the two segments have
been aggregated into one reporting segment for financial statement purposes.
Net sales for our products and services were as follows for the fiscal years ended June 30:
| (in thousands) | 2010 | 2009 | 2008 | |||||||||
Products |
$ | 357,553 | $ | 332,965 | $ | 329,485 | ||||||
Installation and training services |
11,973 | 11,287 | 10,433 | |||||||||
Maintenance |
50,081 | 40,796 | 37,620 | |||||||||
Consolidated net sales |
$ | 419,607 | $ | 385,048 | $ | 377,538 | ||||||
Net sales to unaffiliated customers, classified by major locations in which Dionex operates, were
as follows for the fiscal years ended June 30:
| (in thousands) | 2010 | 2009 | 2008 | |||||||||
United States |
125,176 | $ | 110,084 | $ | 96,250 | |||||||
Europe, excluding Germany |
114,609 | 118,327 | 116,482 | |||||||||
Germany |
39,295 | 37,756 | 40,836 | |||||||||
Japan |
47,714 | 38,529 | 34,996 | |||||||||
China |
52,918 | 47,655 | 33,452 | |||||||||
Other International |
39,895 | 32,697 | 55,522 | |||||||||
Consolidated net sales |
$ | 419,607 | $ | 385,048 | $ | 377,538 | ||||||
Net sales above are attributed to geographic area based on customers’ shipment locations. No
individual customer accounted for more than 10% of net sales in fiscal 2010, 2009, and 2008. No
individual customer accounted for more than 10% of consolidated accounts receivable as of June 30,
2010 and 2009.
Property, plant and equipment (net), classified by major locations in which Dionex operates, were
as follows as of June 30:
| (in thousands) | 2010 | 2009 | 2008 | |||||||||
United States |
$ | 44,288 | $ | 37,679 | $ | 34,526 | ||||||
Europe, excluding Germany |
1,110 | 1,614 | 2,007 | |||||||||
Germany |
19,127 | 22,035 | 25,785 | |||||||||
Japan |
10,849 | 10,099 | 9,464 | |||||||||
China |
317 | 239 | 137 | |||||||||
Other International |
371 | 261 | 416 | |||||||||
Consolidated property, plant, and equipment (net) |
$ | 76,062 | $ | 71,927 | $ | 72,335 | ||||||
Amounts
previously reported as of June 30, 2009 and 2008 for long-lived
assets were $112.7 million and $111.1 million, respectively, and included goodwill,
intangibles and other assets of $40.8 million and $38.8 million, respectively.
Such amounts have been removed from the June 30, 2009 and 2008 disclosure above. We believe that our tangible long-lived assets, which consist of our net
property and equipment, are exposed to greater geographic area risks and uncertainties than
intangible assets and long-term cost deferrals, because these tangible assets are difficult to move
and are relatively illiquid.
48
Table of Contents
Note 16: Quarterly Results of Operations (unaudited)
The following is a summary of the unaudited quarterly results of operations for fiscal 2010 and
2009:
| Quarter | ||||||||||||||||
| (In thousands, except per share amounts) | First | Second | Third | Fourth | ||||||||||||
Fiscal 2010: |
||||||||||||||||
Net sales |
$ | 90,664 | $ | 109,164 | $ | 112,782 | $ | 106,997 | ||||||||
Gross profit |
59,623 | 72,189 | 77,487 | 70,671 | ||||||||||||
Net income attributable to Dionex |
10,336 | 16,582 | 17,731 | 14,407 | ||||||||||||
Basic earnings per share |
0.58 | 0.94 | 1.01 | 0.82 | ||||||||||||
Diluted earnings per share |
0.57 | 0.92 | 0.99 | 0.81 | ||||||||||||
Fiscal 2009: |
||||||||||||||||
Net sales |
$ | 93,435 | $ | 103,041 | $ | 94,396 | $ | 94,176 | ||||||||
Gross profit |
62,711 | 69,570 | 64,225 | 59,891 | ||||||||||||
Net income attributable to Dionex |
11,816 | 17,026 | 15,175 | 11,438 | ||||||||||||
Basic earnings per share |
0.65 | 0.95 | 0.85 | 0.64 | ||||||||||||
Diluted earnings per share |
0.64 | 0.93 | 0.84 | 0.63 | ||||||||||||
Subsequent to the issuance of the Company’s condensed consolidated financial statements for the
third quarter ended March 31, 2009, Dionex discovered two immaterial errors that affected our gross
margin and earnings per share for the quarter ended March 31, 2009. The first error related to
in-transit inventory that was improperly recorded on the balance sheet instead of cost of sales.
The second error was related to an intercompany transfer pricing adjustment that was not properly
eliminated in consolidation during the third quarter causing inventory to be overstated and cost of
sales to be understated. As a result of these errors, our gross profit and net income, as reported
for the three months ended March 31, 2009, are overstated by $1.7 million and $1.1 million,
respectively, and our basic and diluted earnings per share are overstated by $0.06 and $0.06,
respectively. Adjustments were recorded during the fourth quarter in fiscal 2009 to correct these
errors. Consequently, our gross profit and net income, as reported, are understated by $1.7 million
and $1.1 million, respectively, and our basic and diluted earnings per share are understated by
$0.06 and $0.06, respectively, for the three months ended June 30, 2009.
49
Table of Contents
| Item 9. | CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE |
Not applicable.
| Item 9A. | CONTROLS AND PROCEDURES |
Management’s Evaluation of Disclosure Controls and Procedures
We carried out an evaluation, under the supervision and with the participation of our Chief
Executive Officer and Chief Financial Officer, of the effectiveness of our “disclosure controls and
procedures” (as defined in rules promulgated under the Exchange Act Rules 13a-15(e) and 15d-15(e)).
Based on that evaluation, the Chief Executive Officer and Chief Financial Officer have concluded
that our disclosure controls and procedures as of June 30, 2010 were effective to ensure that
information required to be disclosed by us in reports that we file or submit under the Exchange Act
is (i) recorded processed, summarized and reported within the time periods specified in the SEC’s
rules and forms and (ii) accumulated and communicated to our management, including our principal
executive and principal financial officers, to allow timely decisions regarding required
disclosures.
Management’s Annual Report on Internal Control over Financial Reporting
Our management, including our Chief Executive Officer and Chief Financial Officer, is responsible
for establishing and maintaining internal control over financial reporting (as defined in Exchange
Act Rules 13a-15(f) and 15d-15(f)) for us. Internal control over financial reporting is a process
designed to provide reasonable assurance regarding the reliability of financial reporting and the
preparation of financial statements for external purposes in accordance with accounting principles
generally accepted in the United States of America. Our management conducted an assessment of the
effectiveness of our internal control over financial reporting as of June 30, 2010 based on
criteria established in the “Internal Control — Integrated Framework” issued by the Committee of
Sponsoring Organizations of the Treadway Commission. Based on our assessment, our management
concluded that, as of June 30, 2010, our internal control over financial reporting was effective.
Our independent registered public accounting firm, Deloitte & Touche LLP, has audited the
effectiveness of our internal control over financial reporting as of June 30, 2010, as stated in
their report that appears below.
Changes in Internal Control over Financial Reporting
No changes in our internal control over financial reporting occurred during the quarter ended June
30, 2010 that have materially affected or are reasonably likely to materially affect our internal
control over financial reporting.
| Item 9B. | OTHER INFORMATION |
Not applicable.
50
Table of Contents
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders of Dionex Corporation:
We have audited the internal control over financial reporting of Dionex Corporation and
subsidiaries (the “Company”) as of June 30, 2010, based on criteria established in Internal Control
— Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway
Commission. The Company’s management is responsible for maintaining effective internal control
over financial reporting and for its assessment of the effectiveness of internal control over
financial reporting, included in the accompanying Management’s Annual Report on Internal Control
over Financial Reporting. Our responsibility is to express an opinion on the Company’s internal
control over financial reporting based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight
Board (United States). Those standards require that we plan and perform the audit to obtain
reasonable assurance about whether effective internal control over financial reporting was
maintained in all material respects. Our audit included obtaining an understanding of internal
control over financial reporting, assessing the risk that a material weakness exists, testing and
evaluating the design and operating effectiveness of internal control based on the assessed risk,
and performing such other procedures as we considered necessary in the circumstances. We believe
that our audit provides a reasonable basis for our opinion.
A company’s internal control over financial reporting is a process designed by, or under the
supervision of, the company’s principal executive and principal financial officers, or persons
performing similar functions, and effected by the company’s board of directors, management, and
other personnel to provide reasonable assurance regarding the reliability of financial reporting
and the preparation of financial statements for external purposes in accordance with generally
accepted accounting principles. A company’s internal control over financial reporting includes
those policies and procedures that (1) pertain to the maintenance of records that, in reasonable
detail, accurately and fairly reflect the transactions and dispositions of the assets of the
company; (2) provide reasonable assurance that transactions are recorded as necessary to permit
preparation of financial statements in accordance with generally accepted accounting principles,
and that receipts and expenditures of the company are being made only in accordance with
authorizations of management and directors of the company; and (3) provide reasonable assurance
regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the
company’s assets that could have a material effect on the financial statements.
Because of the inherent limitations of internal control over financial reporting, including the
possibility of collusion or improper management override of controls, material misstatements due to
error or fraud may not be prevented or detected on a timely basis. Also, projections of any
evaluation of the effectiveness of the internal control over financial reporting to future periods
are subject to the risk that the controls may become inadequate because of changes in conditions,
or that the degree of compliance with the policies or procedures may deteriorate.
In our opinion, the Company maintained, in all material respects, effective internal control over
financial reporting as of June 30, 2010, based on the criteria established in Internal
Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway
Commission.
We have also audited, in accordance with the standards of the Public Company Accounting Oversight
Board (United States), the consolidated financial statements and consolidated financial statement
schedule as of and for the year ended June 30, 2010, of the Company, and our reports dated August
27, 2010 expressed unqualified opinions on those consolidated financial statements and consolidated
financial statement schedule.
/s/ Deloitte & Touche LLP
San Jose, California
August 27, 2010
August 27, 2010
51
Table of Contents
PART III
| Item 10. | DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE |
IDENTIFICATION OF DIRECTORS AND EXECUTIVE OFFICERS
The information required by Item 10 of Form 10-K with respect to identification of directors and
executive officers is incorporated by reference to the information contained in the sections
captioned “Election of Directors”, “Executive Officers of Dionex Corporation” and “Section 16(a)
Beneficial Ownership Reporting Compliance” in our definitive Proxy Statement for the Annual Meeting
of Stockholders to be held October 26, 2010 (the “2010 Proxy Statement”), which will be filed in
accordance with Regulation 14A under the Exchange Act.
CORPORATE GOVERNANCE
The information required by Item 10 of Form 10-K with respect to the Audit Committee is
incorporated by reference to the information contained in “Election of Directors” in the 2010 Proxy
Statement.
CODE OF ETHICS
The information required by Item 10 of Form 10-K with respect to the Code of Ethics is incorporated
by reference to the information contained in “Code of Ethics” in the 2010 Proxy Statement.
| Item 11. | EXECUTIVE COMPENSATION |
The information required by Item 11 of Form 10-K regarding executive compensation is incorporated
by reference to the information contained in the sections captioned “Compensation of Directors and
Executive Officers,” “Compensation Discussion and Analysis,” “Compensation Committee Interlocks and
Insider Participation”, “Compensation Tables”, and “Compensation Committee Report” in the 2010
Proxy Statement.
| Item 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS |
The information required by Item 12 of Form 10-K is incorporated by reference to the information
contained in the section captioned “Security Ownership Certain Beneficial Owners and Management” in
the 2010 Proxy Statement.
| Item 13. | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE |
The information required by Item 13 of Form 10-K is incorporated by reference to the information
contained in the section captioned “Election of Directors” and “Transactions with Related Persons”
in the 2010 Proxy Statement.
| Item 14. | PRINCIPAL ACCOUNTING FEES AND SERVICES |
The information required by Item 14 of Form 10-K is incorporated by reference to the information
contained in the sections captioned “Independent Registered Public Accounting Firm’s Fees,” “Policy
on Audit Committee Pre-Approval” and “Audit Committee Disclosure” in the 2010 Proxy Statement.
PART IV
| Item 15. | EXHIBITS AND FINANCIAL STATEMENT SCHEDULES |
(a) (1) Financial Statements — See Index to Financial Statements at page 26 of this Report.
(2) Financial Statement Schedule — See Index to Financial Statement Schedules at pages 56 of
this Report.
(3) Exhibits — See Exhibit Index at page 59 through 60 of this Report.
52
Table of Contents
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, we have
duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| DIONEX CORPORATION |
||||
| By | /s/ Frank Witney | |||
| Frank Witney | ||||
| President, Chief Executive Officer and Director | ||||
Date: August 27, 2010
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below constitutes and
appoints Frank Witney and Craig A. McCollam, and each or either of them, each with the power of
substitution, his attorney-in-fact, to sign any amendments to this report, with exhibits thereto
and other documents in connection therewith, with the Securities and Exchange Commission, hereby
ratifying and confirming all that each of said attorneys-in-fact, or his substitute or substitutes,
may do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed
below by the following persons on behalf of Dionex Corporation and in the capacities and on the
dates indicated.
| Signature | Title | Date | ||
/s/ Frank Witney
|
President, Chief Executive Officer, and Director (Principal Executive Officer) | August 27, 2010 | ||
/s/ Craig A. McCollam
|
Executive Vice President and Chief Financial Officer (Principal Financial and Accounting Officer) | August 27, 2010 | ||
/s/ A. Blaine Bowman
|
Director | August 27, 2010 | ||
/s/ David L. Anderson
|
Director | August 27, 2010 | ||
/s/ Roderick McGeary
|
Director | August 27, 2010 | ||
/s/ Riccardo Pigliucci
|
Lead Director | August 27, 2010 | ||
/s/ Michael W. Pope
|
Director | August 27, 2010 |
53
Table of Contents
INDEX TO FINANCIAL STATEMENT SCHEDULES
| Page | ||||
FINANCIAL STATEMENT SCHEDULE |
||||
| 55 | ||||
| 56 | ||||
All other schedules are omitted because they are not required, are not applicable or the
information is included in the consolidated financial statements or notes thereto.
54
Table of Contents
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders of Dionex Corporation:
We have audited the consolidated financial statements of Dionex Corporation and subsidiaries (the
“Company”) as of June 30, 2010 and 2009, and for each of the three years in the period ended June
30, 2010, and the Company’s internal control over financial reporting as of June 30, 2010, and have
issued our reports thereon dated August 27, 2010. Such consolidated financial statements and
reports are included elsewhere in this Annual Report on Form 10-K for the year-ended June 30, 2010.
Our audits also included the consolidated financial statement schedule of the Company listed in
Item 15. This consolidated financial statement schedule is the responsibility of the Company’s
management. Our responsibility is to express an opinion based on our audits. In our opinion, such
consolidated financial statement schedule, when considered in relation to the basic consolidated
financial statements taken as a whole, presents fairly, in all material respects, the information
set forth therein.
/s/ Deloitte & Touche LLP
San Jose, California
August 27, 2010
August 27, 2010
55
Table of Contents
SCHEDULE II
DIONEX CORPORATION
VALUATION AND QUALIFYING ACCOUNTS AND RESERVES
YEAR ENDED JUNE 30, 2010, 2009, AND 2008
VALUATION AND QUALIFYING ACCOUNTS AND RESERVES
YEAR ENDED JUNE 30, 2010, 2009, AND 2008
| Charged | ||||||||||||||||||||
| Balance | (Credited) | Balance | ||||||||||||||||||
| Beginning of | to Other | End of | ||||||||||||||||||
| Year | Additions | Accounts(1) | Deductions(2) | Year | ||||||||||||||||
| (In thousands) | ||||||||||||||||||||
YEAR ENDED JUNE 30, 2010: |
$ | 560 | $ | 99 | $ | (44 | ) | $ | (72 | ) | $ | 543 | ||||||||
Allowance for doubtful accounts |
||||||||||||||||||||
YEAR ENDED JUNE 30, 2009: |
$ | 524 | $ | 163 | $ | (102 | ) | $ | (25 | ) | $ | 560 | ||||||||
Allowance for doubtful accounts |
||||||||||||||||||||
YEAR ENDED JUNE 30, 2008: |
$ | 610 | $ | (95 | ) | $ | 120 | $ | (111 | ) | $ | 524 | ||||||||
Allowance for doubtful accounts |
||||||||||||||||||||
| (1) | Effects of exchange rate changes | |
| (2) | Accounts written off, net of recoveries |
56
Table of Contents
EXHIBIT INDEX
| Exhibit | ||||||||
| Number | Description | Reference | ||||||
| 3.1 | Restated Certificate of Incorporation, filed December 12, 1988
|
(1 | ) | |||||
| 3.2 | Certificate of Amendment of Restated Certificate of Incorporation, filed December 1, 1999 (Exhibit 3.2)
|
(8 | ) | |||||
| 3.3 | Amended and Restated Bylaws, August 6, 2008 (Exhibit 99.1)
|
(12 | ) | |||||
| 10.1 | * | Medical Care Reimbursement Plan as amended October 30, 2007 (Exhibit 10.1)
|
(9 | ) | ||||
| 10.2 | Credit Agreement dated December 23, 2009 between Wells Fargo Bank and Dionex Corporation (Exhibit 10.2)
|
(3 | ) | |||||
| 10.3 | * | Management Incentive Bonus Plan dated August 6, 2009 (Exhibit 99.1)
|
(14 | ) | ||||
| 10.4 | * | Dionex Corporation 2004 Equity Incentive Plan, as amended October 2007 (Exhibit 10.1)
|
(10 | ) | ||||
| 10.5 | * | Form of Stock Option Agreement for non-employee directors (Exhibit 10.5)
|
(11 | ) | ||||
| 10.6 | * | Form of Stock Option Agreement for other than non-employee directors (Exhibit 10.6)
|
(11 | ) | ||||
| 10.7 | * | Form of Stock Unit Award Agreement (Exhibit 10.2)
|
(10 | ) | ||||
| 10.8 | * | Form of International Stock Option Agreement (Exhibit 10.8)
|
(9 | ) | ||||
| 10.9 | * | Form of Stock Unit Award Agreement for U.S. employees
|
(2 | ) | ||||
| 10.10 | * | Form of Stock Unit Award Agreement for International employees
|
(2 | ) | ||||
| 10.11 | * | Employee Stock Participation Plan (Exhibit 10.13)
|
(5 | ) | ||||
| 10.12 | * | Form of Performance Stock Unit Grant Notice |
||||||
| 10.13 | * | Change in Control Severance Benefit Plan as amended April 26, 2010
|
(2 | ) | ||||
| 10.14 | * | Summary of Compensation Arrangements with named executive officers
|
(7 | ) | ||||
| 10.15 | * | Form of Indemnification Agreement (Exhibit 10.1)
|
(13 | ) | ||||
| 21.1 | Subsidiaries of Dionex Corporation |
|||||||
| 23.1 | Consent of Independent Registered Public Accounting Firm |
|||||||
| 24.1 | Power of Attorney (reference is made to the signature page of this report on Form 10-K) |
|||||||
| 31.1 | Certification of Chief Executive Officer Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 |
|||||||
| 31.2 | Certification of Chief Financial Officer Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 |
|||||||
| 32.1 | † | Certification of the Chief Executive Officer Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
||||||
| 32.2 | † | Certification of the Chief Financial Officer Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
||||||
| (1) | Incorporated by reference to the indicated exhibit in our Form 10-Q filed September 20, 1989. | |
| (2) | Incorporated by reference to the indicated exhibit in our Form 8-K filed April 28, 2010. |
57
Table of Contents
| (3) | Incorporated by reference to the indicated exhibit in our Form 10-Q filed February 9, 2010. | |
| (4) | Incorporated by reference to the indicated exhibit in our Form 10-Q filed February 9, 2010. | |
| (5) | Incorporated by reference to the indicated exhibit in our Form 10-K filed September 10, 2004. | |
| (6) | Incorporated by reference to the indicated exhibit in our Form 8-K filed December 22, 2004. | |
| (7) | Incorporated by reference to the indicated exhibit in our Form 8-K filed August 9, 2010 and August 16, 2010. | |
| (8) | Incorporated by reference to the indicated exhibit in our Form 10-K filed August 29, 2007. | |
| (9) | Incorporated by reference to the indicated exhibit in our Form 10-Q filed November 9, 2007. | |
| (10) | Incorporated by reference to the indicated exhibit in our Form 8-K filed October 15, 2007. | |
| (11) | Incorporated by reference to the indicated exhibit in our Form 10-Q filed February 8, 2008. | |
| (12) | Incorporated by reference to the indicated exhibit in our Form 8-K filed August 11, 2008. | |
| (13) | Incorporated by reference to the indicated exhibit in our Form 8-K filed November 3, 2008. | |
| (14) | Incorporated by reference to the indicated exhibit in our Form 8-K filed August 10, 2009. | |
| (15) | Incorporated by reference to the indicated exhibit in our Form 10-Q filed February 9, 2010. |
| * | Management contract or compensatory plan or arrangement. | |
| † | This certification accompanies the Form 10-K to which it relates, is not deemed filed with the Securities and Exchange Commission and is not to be incorporated by reference into any filing of the Registrant under the Securities Act of 1933, as amended, or the Securities Exchange Act of 1934, as amended (whether made before or after the date of the Form 10-K), irrespective of any general incorporation language contained in such filing. |
58
