Attached files
| file | filename |
|---|---|
| EX-23.1 - Bohai Pharmaceuticals Group, Inc. | v193404_ex23-1.htm |
As
filed with the Securities and Exchange Commission on August 12, 2010
Registration
No. 333-165149
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM
S-1/A
(Amendment
No. 5)
REGISTRATION
STATEMENT UNDER THE SECURITIES ACT OF 1933
Bohai
Pharmaceuticals Group, Inc.
(Exact
name of registrant as specified in its charter)
|
Nevada
|
2834
|
98-0588402
|
||
|
(State
or other jurisdiction of
incorporation
or organization)
|
(Primary
Standard Industrial
Classification
Code Number)
|
(I.R.S.
Employer
Identification
No.)
|
c/o
Yantai Bohai Pharmaceuticals Group Co. Ltd.
No.
9 Daxin Road, Zhifu District
Yantai,
Shandong Province, China 264000
+86
(535)-685-7928
(Address,
including zip code, and telephone number, including area code, of registrant’s
principal executive offices)
Mr.
Hongwei Qu
President
and Chief Executive Officer
Bohai
Pharmaceuticals Group, Inc.
c/o
Yantai Bohai Pharmaceuticals Group Co. Ltd.
No.
9 Daxin Road, Zhifu District
Yantai,
Shandong Province, China 264000
+86
(535)-685-7928
(Name,
address including zip code, and telephone number, including area code, of agent
for service)
Copies
to:
Barry
I. Grossman, Esq.
Lawrence
A. Rosenbloom, Esq.
Ellenoff
Grossman & Schole LLP
150
East 42nd Street, 11th Floor
New
York, NY 10017
212-370-1300
212-370-7889
(fax)
Registrant’s
telephone number: +86 (535)-685-7928
APPROXIMATE
DATE OF PROPOSED SALE TO PUBLIC: From time to time after this
Registration
Statement becomes effective.
If any of
the securities being registered on this Form are to be offered on a delayed or
continuous basis pursuant to Rule 415 under the Securities Act of 1933, check
the following box: ¨
If this
Form is filed to register additional securities for an offering pursuant to Rule
462(b) under the Securities Act, check the following box and list the Securities
Act registration statement number of the oearlier effective
registration statement for the same offering.
If this
Form is a post-effective amendment filed pursuant to Rule 462(c) under the
Securities Act, check the following box and list the Securities Act registration
statement number of the earlier effective oregistration statement
for the same offering.
If this
Form is a post-effective amendment filed pursuant to Rule 462(d) under the
Securities Act, check the following box and list the Securities Act registration
statement number of the earlier effective oregistration statement
for the same offering.
Indicate
by check mark whether the registrant is a large accelerated filer, an
accelerated filer, a non-accelerated filer, or a smaller reporting company. See
the definitions of “large accelerated filer,” “accelerated filer,” and “smaller
reporting company” in Rule 12b-2 of the Exchange Act. (Check one):
|
Large
accelerated filer
|
¨
|
Accelerated
filer
|
¨
|
|
Non-accelerated
filer
|
¨
|
Smaller
reporting company
|
x
|
|
(Do
not check if a smaller reporting company)
|
CALCULATION
OF REGISTRATION FEE
|
Title of each class of securities
to be registered
|
Amount to be
registered(1)
|
Proposed
maximum
offering
price per
unit
|
Proposed
maximum
aggregate
offering
price
|
Amount of
registration fee
|
|||||||||||
|
common
stock, par value $0.001 per share, offered by certain selling stockholders
|
7,032,529 shares
|
$ | 1.00 | (2) | $ | 7,032,529 | $ | 499.10 | * | ||||||
|
common
stock, par value $0.001 per share, underlying the principal of convertible
notes held by certain selling stockholders (1)
|
6,000,000 shares
|
(3) | $ | 2.00 | $ | 12,000,000 | $ | 855.60 | * | ||||||
|
common
stock, par value $0.001 per share, underlying the interest of convertible
notes held by certain selling stockholders (1)
|
690,000
shares
|
(4) | $ | 2.00 | $ | 1,380,000 | $ | 98.40 | * | ||||||
|
common
stock, par value $0.001 per share, underlying warrants held by certain
selling stockholders (1)
|
6,000,000 shares
|
(5) | $ | 2.40 | $ | 14,400,000 | $ | 1,026.72 | * | ||||||
|
common
stock, par value $0.001 per share, underlying placement agent warrants
held by certain selling stockholders
|
600,000 shares
|
(6) | $ | 2.40 | $ | 1,440,000 | $ | 102.67 | * | ||||||
|
TOTAL
|
20,322,529 shares
|
$ | 36,252,529 | $ | 2,680.89 | ||||||||||
|
*
|
Previously
paid.
|
|
(1)
|
Pursuant
to Rule 416 of the Securities Act of 1933, also registered hereby are such
additional and indeterminable number of shares as may be issuable due to
adjustments for changes resulting from stock dividends, stock splits and
similar changes as well as anti-dilution provisions applicable to the
notes and warrants.
|
|
(2)
|
Estimated
pursuant to Rule 457(f)(2) under the Securities Act of 1933 solely for the
purpose of calculating the amount of the registration fee, based on the
book value of such securities received by the Company in the Share
Exchange.
|
|
(3)
|
The
6,000,000 shares of common stock are being registered for resale by
certain selling stockholders named in this registration statement, which
shares are issuable by the registrant upon the conversion of the principal
amount of the registrant’s 8% Convertible Notes due January 5, 2012.
|
|
(4)
|
The 690,000 shares of common stock are being registered for resale by certain selling stockholders named in this registration statement, which shares are potentially issuable by the registrant upon the conversion of the interest accrued under the registrant’s 8% Convertible Notes due January 5, 2012. |
|
(5)
|
The
6,000,000 shares of common stock are being registered for resale by
certain selling stockholders named in this registration statement, which
shares are issuable by the registrant upon the exercise of the
registrant’s common stock purchase warrants issued on January 5, 2010.
|
|
(6)
|
The
600,000 shares of common stock are being registered for resale by certain
selling stockholders named in this registration statement, which shares
are issuable by the registrant upon the exercise of the placement agent
warrants issued on January 5, 2010.
|
The
registrant hereby amends this registration statement on such date or dates as
may be necessary to delay its effective date until the registrant shall file a
further amendment which specifically states that this registration statement
shall thereafter become effective in accordance with Section 8(a) of the
Securities Act of 1933 or until the registration statement shall become
effective on such date as the Securities and Exchange Commission, acting
pursuant to Section 8(a) may determine.
The
information in this prospectus is not complete and may be changed. We
may not sell these securities until the registration statement filed with the
Securities and Exchange Commission (“SEC”) is effective. This
prospectus is not an offer to sell these securities and is not soliciting an
offer to buy these securities in any state where the offer or sale is not
permitted.
|
Preliminary
Prospectus
|
Subject
to Completion, dated August 12, 2010
|

20,322,529
Shares of
Common
Stock
This
prospectus relates to the sale of up to a total of 20,322,529 shares of common
stock of Bohai Pharmaceuticals Group, Inc., a Nevada corporation, that may be
sold from time to time by the selling stockholders named in this prospectus and
their successors and assigns. The shares of common stock subject to
this prospectus include: (i) 5,482,529 shares of common stock issued in
connection with our January 5, 2010 share exchange transaction and 1,550,000
other shares of restricted common stock; (ii) 6,690,000 shares issuable upon
conversion by certain selling stockholders of the principal and interest
underlying our 8% Convertible Notes due January 5, 2012, which we refer to
herein as the Notes; (iii) 6,000,000 shares issuable upon the exercise by
certain selling stockholders of our common stock purchase warrants issued on
January 5, 2010, which we refer to herein as the Warrants; and (iv) 600,000
shares upon issuable upon the exercise by certain selling stockholders of
placement agents’ warrants issued on January 5, 2010, which we refer to herein
as the Placement Agent Warrants. The securities offered for resale
hereby were issued to the applicable selling stockholders in private placement
or other exempt transactions completed prior to the filing of the registration
statement of which this prospectus is a part.
The
selling stockholders may sell all or a portion of their shares through public or
private transactions at prevailing market prices or at privately negotiated
prices. Information regarding the selling stockholders and the times
and manner in which they may offer and sell the shares under this prospectus is
provided under “Selling Stockholders” and “Plan of Distribution” in this
prospectus. We have agreed to pay all the costs and expenses of this
registration.
We will
not receive any of the proceeds from the sale of shares by the selling
stockholders. We may receive proceeds upon exercise of Warrants or
the Placement Agent Warrants, and any proceeds we receive will be used for
general corporate purposes and for working capital.
Our
common stock is listed for quotation on the Over-the-Counter Bulletin Board, or
OTCBB, under the symbol “BOPH”. There is very limited trading in our
common stock. On July 23, 2010, the most recent day that our stock
traded, the last reported price per share of our common stock was
$2.25. You are urged to obtain current market quotations of our
common stock before purchasing any of the shares being offered for sale pursuant
to this prospectus.
An
investment in our securities is highly speculative, involves a high degree of
risk and should be considered only by persons who can afford the loss of their
entire investment. See “Risk Factors” beginning on page 6 of this
prospectus.
Neither
the SEC nor any state securities commission has approved or disapproved of these
securities or passed upon the accuracy or adequacy of this
prospectus. Any representation to the contrary is a criminal
offense.
The date of this
prospectus is
[ ],
2010.
TABLE
OF CONTENTS
|
Page
|
|
|
Number
|
|
|
About
This Prospectus
|
-i-
|
|
Prospectus
Summary
|
1
|
|
Risk
Factors
|
6
|
|
Cautionary
Note Regarding Forward-Looking Statements
|
30
|
|
Use
of Proceeds
|
31
|
|
Determination
of Offering Price
|
31
|
|
Management’s
Discussion and Analysis of Financial Conditions and Results of Operations
|
32
|
|
Business
|
42
|
|
Management
|
59
|
|
Executive
Compensation
|
62
|
|
Certain
Relationships and Related Transactions
|
63
|
|
Security
Ownership of Certain Beneficial Owners and Management
|
65
|
|
Description
of Securities
|
66
|
|
Selling
Stockholders
|
68
|
|
Plan
of Distribution
|
76
|
|
Market
for Common Equity and Related Stockholder Matters
|
79
|
|
Legal
Matters
|
79
|
|
Experts
|
79
|
|
Changes
In and Disagreements With Accountants on Accounting and Financial
Disclosure
|
80
|
|
Where
You Can Find More Information
|
80
|
|
Index
to Financial Statements
|
F-1
|
ABOUT
THIS PROSPECTUS
This
prospectus is part of a registration statement we filed with the
SEC. You should rely only on the information provided in this
prospectus and incorporated by reference in this prospectus. We have
not authorized anyone to provide you with information different from that
contained in or incorporated by reference into this prospectus. This
prospectus does not constitute an offer to sell or a solicitation of an offer to
buy any securities other than the common stock offered by this
prospectus. This prospectus does not constitute an offer to sell or a
solicitation of an offer to buy any common stock in any circumstances in which
such offer or solicitation is unlawful. The selling stockholders are
offering to sell, and seeking offers to buy, shares of common stock only in
jurisdictions where offers and sales are permitted.
Neither
the delivery of this prospectus nor any sale made in connection with this
prospectus shall, under any circumstances, create any implication that there has
been no change in our affairs since the date of this prospectus or that the
information contained by reference to this prospectus is correct as of any time
after its date. The information in this prospectus is accurate only
as of the date of this prospectus, regardless of the time of delivery of this
prospectus or of any sale of common stock. The rules of the SEC may
require us to update this prospectus in the future.
-i-
PROSPECTUS
SUMMARY
The
following summary highlights selected information contained in this
prospectus. This summary does not contain all the information you
should consider before investing in the securities. Before making an
investment decision, you should read the entire prospectus carefully, including
the information set forth under the headings “Risk Factors” and “Management’s
Discussion and Analysis of Financial Condition and Results of Operations,” and
the financial statements and the notes to the financial statements included in
this prospectus.
As
used throughout this prospectus, the terms “BOPH”, “Company”, “we,” “us,” or
“our” refer to Bohai Pharmaceuticals Group, Inc., a Nevada corporation, together
with: (i) its wholly owned subsidiary, Chance High International Limited, a
British Virgin Islands company (“Chance High”); (ii) its indirect wholly foreign
owned subsidiary, Yantai Shencaojishi Pharmaceuticals Co., Ltd., a PRC company
(“WFOE”), and the WFOE’s variable interest entity, Yantai Bohai Pharmaceuticals
Group Co., Ltd., a PRC company (“Bohai”). In this prospectus, we
sometimes refer to BOPH, Chance High, WFOE and Bohai collectively as the
“Group.”
As
used in this prospectus, “China” or “the PRC” refers to the People’s Republic of
China.
Our
Company
We are
engaged in the production, manufacturing and distribution in China of herbal
pharmaceuticals based on traditional Chinese medicine, which we refer to herein
as Traditional Chinese Medicine or TCM. We are based in the city of
Yantai, Shandong Province, China and our operations are exclusively in
China.
Our
medicines address rheumatoid arthritis, viral infections, gynecological
diseases, cardio vascular issues and respiratory diseases. We
obtained Drug Approval Numbers for 29 varieties of traditional Chinese herbal
medicines in 2004 and currently produce 10 varieties of approved traditional
Chinese herbal medicines in seven delivery systems: tablets, granules, capsules,
syrup, concentrated powder, tincture and medicinal wine. Of these 10
products, 5 are prescription drugs and 5 are over the counter, or OTC,
products. In a significant development, on December 1, 2009, two of
our lead products, Tongbi Capsules and Tablets and Lung Nourishing Cream, became
eligible for reimbursement under China’s National Medical Insurance Program.
Background
and Key Events
We were
incorporated under the laws of the State of Nevada under the name Link Resources
Inc. on January 9, 2008. Our principal office was in Calgary,
Alberta, Canada. Prior to January 5, 2010, we were a public “shell”
company in the exploration stage since our formation and had not yet realized
any revenues. We entered into a Mineral Lease Agreement on April 1,
2008 for two mining claims in Pershing County, Nevada, in an area known as the
Goldbanks East Prospect. We terminated the lease on July 7,
2009.
Share
Exchange with Chance High
Pursuant
to the Share Exchange Agreement entered into on January 5, 2010 (the “Share
Exchange Agreement”), and related share exchange (the “Share Exchange”) by and
among us, Chance High, and the shareholders of Chance High (the “Chance High
Shareholders”), we acquired Chance High and its indirect, controlled subsidiary
Bohai, a Chinese company engaged the production, manufacturing and distribution
in China of herbal medicines, including capsules and other products, based on
Traditional Chinese Medicine. The closing of the Share Exchange (the
“Closing”) took place on January 5, 2010. As of the Closing, pursuant
to the terms of the Share Exchange Agreement, we acquired all of the outstanding
equity securities (the “Chance High Shares”) of Chance High from the Chance High
Shareholders, and the Chance High Shareholders transferred and contributed all
of their Chance High Shares to us. In exchange, we issued to Chance
High Shareholders an aggregate of 13,162,500 newly issued shares of our common
stock. Certain of the Chance High Shareholders are selling
stockholders hereunder.
1
In
addition, pursuant to the terms of the Share Exchange Agreement, Anthony
Zaradic, our former sole officer and director (“Zaradic”), cancelled a total of
1,500,000 shares of common stock owned by him. As a further condition
of the Share Exchange, effective as of January 5, 2010, Zaradic resigned from
all of his positions with our company and Hongwei Qu (“Qu”), the former
principal stockholder and Executive Director of Bohai, was appointed as our
President, Chief Executive Officer, Interim Chief Financial Officer, Treasurer
and Secretary and also, effective January 16, 2010, as our sole
director. In June 2010, Mr. Qu relinquished the positions of Interim
Chief Financial Officer, Treasurer and Secretary and we appointed Gene Hsiao as
our Chief Financial Officer. On July 12, 2010, we appointed three
independent directors to our board of directors.
January
5, 2010 Private Placement and Related Agreements
Securities Purchase
Agreement. On January 5, 2010, we entered into a Securities
Purchase Agreement (the “Securities Purchase Agreement”) with certain accredited
investors, who are selling stockholders hereunder (the “Investors”) and Euro
Pacific Capital, Inc. (“Euro Pacific”), as representative of the Investors,
relating to a private placement by us of 6,000,000 units consisting of Notes and
Warrants, which we refer to herein as the private placement. The
consummation of the private placement resulted in gross proceeds to us of
$12,000,000 and net proceeds of approximately $9,700,000. Each unit
consisted of a $2.00 principal amount, two year convertible Note and a three
year Warrant to purchase one share of our common stock at $2.40 per share,
subject to certain conditions. Euro Pacific acted as the lead
placement agent and Chardan Capital Markets, LLC acted as co-placement agent of
the private placement.
Registration Rights
Agreement. In connection with the private placement, we
entered into Registration Rights Agreement (the “Registration Rights Agreement”)
with the Investors which sets forth the rights of the Investors to have the
shares of common stock underlying the Notes and Warrants issued in the private
placement registered with the Securities and Exchange Commission (“SEC”) for
public resale. The filing of the registration statement of which this
prospectus is a part is intended to satisfy certain of our obligations the
Registration Rights Agreement.
Securities Escrow
Agreement. Also in connection with the private placement, we
entered into a Securities Escrow Agreement (the “Securities Escrow Agreement”)
with Euro Pacific, as representative of the Investors, our principal
stockholder, Glory Period Limited, a British Virgin Islands company that we
refer to herein as Glory Period and which was the majority shareholder of Chance
High prior to the Share Exchange, and Escrow, LLC, as escrow agent (the “Escrow
Agent”). Pursuant to the Securities Escrow Agreement, Glory Period
has pledged and deposited a stock certificate representing 1 million shares of
our common stock (the “Escrow Shares”) into escrow in order to provide security
to the Investors in the event of an occurrence of an event of default under the
Notes. Upon the earlier to occur of the full repayment of all amounts
due to the Investors under the Notes or the conversion of fifty percent of the
principal face value of Notes into shares of common stock, the Investors’ rights
in and to the Escrow Shares shall terminate. Glory Period is
controlled by Qu through certain contractual relationships described elsewhere
in this prospectus.
2
Closing Escrow Agreement.
Pursuant to a Closing Escrow Agreement (the “Closing Escrow Agreement”)
that we entered into in connection with the private placement on December 10,
2009, we placed a total of $240,000 of proceeds from the private placement (the
“Holdback Amount”) with the Escrow Agent. The Holdback Amount
represents an amount sufficient to satisfy the payment to the Investors of one
quarterly interest payment due on the aggregate principal amount of all Notes
issued in the private placement. If, subject to certain conditions
and after applicable notice and cure periods, an event of default is declared by
Euro Pacific with respect to our failure to make a quarterly interest payment to
Investors, the Escrow Agent shall disburse such portion of the Holdback Amount
to the Investors, and we shall be obligated to deposit additional amounts equal
to the Holdback Amount with Escrow Agent. At such time as
seventy-five percent of the aggregate shares of common stock underlying the
Notes have been issued upon conversion of the Notes, all remaining funds of the
Holdback Amount shall promptly be disbursed to us.
Corporate
Name Change
On January 29, 2010, we entered into an
Agreement and Plan of Merger (the “Merger Agreement”) pursuant to which we
merged with a newly formed, wholly owned subsidiary called Bohai Pharmaceuticals
Group, Inc., a Nevada corporation (“Merger Sub” and such merger transaction, the
“Merger”). Upon the consummation of the Merger, the separate
existence of Merger Sub ceased and our stockholders became stockholders of the
surviving company named Bohai Pharmaceuticals Group, Inc. As
permitted by Chapter 92A.180 of Nevada Revised Statutes, the sole purpose of the
Merger was to effect a change of our corporate name.
Change
of Our Independent Registered Accounting Firm
Effective
January 29, 2010, upon the approval of our board of directors, we dismissed John
Kinross-Kennedy as our independent registered public accountant and appointed
Parker Randall CF (H.K.) CPA Limited as our independent registered public
accounting firm
Corporate
Structure and Related Agreements
Our post
Share Exchange organization structure is summarized below:
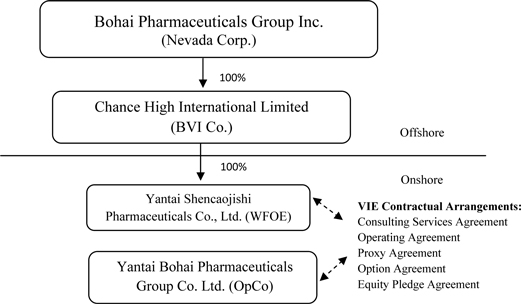
3
Chance
High owns 100% of the issued and outstanding capital stock of the
WFOE. On December 7, 2009, the WFOE entered into a series of variable
interest entity contractual agreements (the “VIE Agreements”) with Bohai and its
three shareholders, which include Qu (our Chairman, President and Chief
Executive Officer, who owns 90% of Bohai’s shares) and two unaffiliated
parties. Pursuant to the VIE Agreements, WFOE does not directly own
the equity of our operating subsidiary, but rather effectively assumed
management of the business activities of Bohai and has the right to appoint all
executives and senior management and the members of the board of directors of
Bohai. The VIE Agreements are comprised of a series of agreements,
including a Consulting Services Agreement, Operating Agreement and Proxy
Agreement, through which WFOE has the right to advise, consult, manage and
operate Bohai for an annual fee in the amount of Bohai’s yearly net profits
after tax. Additionally, Bohai’s shareholders have pledged their
rights, titles and equity interest in Bohai as security for WFOE to collect
consulting and services fees provided to Bohai through an Equity Pledge
Agreement. In order to further reinforce WFOE’s rights to control and
operate Bohai, Bohai’s shareholders granted WFOE an exclusive right and option
to acquire all of their equity interests in Bohai through an Option
Agreement.
In
addition, on December 7, 2009, Mr. Qu entered into a call option agreement (the
“Call Option Agreement”) with Joshua Tan (“Tan”), the sole shareholder of Glory
Period. The Call Option Agreement became effective upon the closing
of the Share Exchange. Under the Call Option Agreement, Tan shall
transfer up to 100% shares of Glory Period within the next 3 years to Qu for
nominal consideration, which would give Qu indirect ownership of a significant
percentage of our common stock. The Call Option Agreement provides
that Tan shall not dispose any of the shares of Glory Period without Qu’s prior
written consent.
Following
the consummation of the Share Exchange, Glory Period holds 55% of the issued and
outstanding shares of our common stock (not taking into consideration the shares
of common stock underlying the Notes and Warrants issued in the private
placement). The shares of our common stock held by Glory Period are
not being offered for resale pursuant to this prospectus.
Executive
Offices
Our
executive office is located at No. 9 Daxin Road, Zhifu District, Yantai,
Shandong Province, P.R. China 264000. Our telephone number is
+86(535)-685-7928.
4
THE
OFFERING
|
Common
stock outstanding before the offering
|
16,500,000
|
|
|
Common
stock offered by selling stockholders
|
Up
to 20,322,529 shares of common stock held by the selling stockholders or
underlying securities held by the selling stockholders.
|
|
|
Common
stock to be outstanding after the offering
|
Up
to 28,850,000 shares, assuming full conversion or exercise of the Notes,
Warrants and Placement Agent Warrants.
|
|
|
OTBCC
Symbol
|
BOPH. No
active market for our common stock presently exists.
|
|
|
Use
of proceeds
|
We
will not receive any proceeds from the sale of the common stock offered
hereby. However, we may receive up to a maximum of $15.84
million of proceeds from the exercise of the warrants held by certain
selling stockholders, which proceeds we would expect to use for general
working capital. No assurances can be given, however, that all
or any portion of such warrants will ever be
exercised.
|
5
RISK
FACTORS
Our
business, operations and financial condition are subject to various significant
risks. Some of these risks are described below and you should take
these risks into account in making a decision to invest in our common
stock. If any of the following risks actually accurs, we may not be
able to conduct our business as currently planned and our financial condition
and operating results could be seriously harmed. In that case, the
market price of our common stock could decline and you could lose all or part of
your investment in our common stock.
Risks
Related to Our Business
Our
limited operating history may not serve as an adequate basis to judge our future
prospects and results of operations.
Our
limited operating history in the traditional Chinese herbal medicines industry
may not provide a meaningful basis for evaluating our business. Bohai
entered into its current line of business in September 2004. Although Bohai’s
revenues have grown rapidly since its inception, we cannot guarantee that we
will maintain profitability or that we will not incur net losses in the
future. We will continue to encounter risks and difficulties that
companies at a similar stage of development frequently experience, including the
potential failure to:
|
|
·
|
obtain
sufficient working capital to support our
expansion;
|
|
|
·
|
maintain
or protect our intellectual
property;
|
|
|
·
|
maintain
our proprietary technology;
|
|
|
·
|
expand
our product offerings and maintain the quality of our
products;
|
|
|
·
|
manage
our expanding operations and continue to fill customers’ orders on
time;
|
|
|
·
|
maintain
adequate control of our expenses allowing us to realize anticipated
revenue growth;
|
|
|
·
|
implement
our product development, marketing, sales and acquisition strategies and
adapt and modify them as
needed;
|
|
|
·
|
integrate
any future acquisitions; and
|
|
|
·
|
anticipate
and adapt to changing conditions in the Chinese herbal medicines industry
resulting from changes in government regulations, mergers and acquisitions
involving our competitors, technological developments and other
significant competitive and market
dynamics.
|
If we are
unable to address any or all of the foregoing risks, our business may be
materially and adversely affected.
6
We
will likely need to raise additional funds in the future to grow our business,
which funds may not be available on acceptable terms or at all, and, without
additional funds, we may not be able to maintain or expand our
business.
We expect
that the net proceeds from our January 2010 private placement, together with
cash generated from our operations, will be sufficient to fund our projected
operations for at least the next 12 months. It is likely however that
in the future we will require substantial funds in order to fund operating
expenses and growth plans to develop manufacturing, marketing and sales
capabilities and to cover public company costs. Without enough funds,
we may not be able to meet these goals. We may seek additional
funding through public or private financing or through collaborative
arrangements with strategic partners.
You
should also be aware that in the future:
|
|
·
|
We
cannot be certain that additional capital will be available on favorable
terms, if at all;
|
|
|
·
|
Any
available additional financing may not be adequate to meet our goals;
and
|
|
|
·
|
Any
equity financing would result in dilution to our
stockholders.
|
If we
cannot raise additional funds when needed, or on acceptable terms, we may not be
able to effectively execute our growth strategy, take advantage of future
opportunities, or respond to competitive pressures or unanticipated
requirements. In addition, we may be required to scale back or
discontinue our production and development program, or obtain funds through
strategic alliances that may require us to relinquish certain
rights.
We
have significant short-term debt obligations, which mature in less than one
year. Our inability to extend the maturities of, or to refinance,
this debt could result in defaults, and in certain instances, foreclosures on
our assets. Moreover, we may be unable to obtain financing to fund
ongoing operations and future growth.
We
currently depend on short-term bank loans and net revenues to meet our
short-term cash requirements. As of March 31, 2010, our total bank
debt outstanding was $4.38 million which carries maturity periods ranging from
six months to one year, while the short-term and revolving nature of these
credit facilities is common in China. The majority of this debt is
guaranteed by third-parties and our CEO, Mr. Qu, and a portion is secured by our
inventories and fixed assets. In China, short-term bank loans
generally mature in one year or less and contain no specific renewal
terms. However, it is customary practice for banks and borrowers to
negotiate roll-overs or renewals of short-term borrowings on an on-going basis
shortly before they mature. Although we have renewed our short-term
borrowings in the past, we cannot assure you that we will be able to renew these
loans in the future as they mature. If we are unable to obtain
renewals of these loans or sufficient alternative funding on reasonable terms
from banks or other parties, we will have to repay these borrowings with the
cash on our balance sheet or cash generated by our future operations, if
any.
Moreover,
we cannot assure you that our business will generate sufficient cash flow from
operations to repay these borrowings. Failure to obtain extensions of
the maturity dates of, or to refinance, these obligations or to obtain
additional equity financing to meet these debt obligations would result in an
event of default with respect to such obligations and could result in the
foreclosure on the collateral. The sale of such collateral at
foreclosure would significantly disrupt our ability to produce products for our
customers in the quantities required by customer orders or deliver products in a
timely fashion, which could significantly lower our revenues and
profitability.
7
In
addition, we may be exposed to changes in interest rates. If interest
rates increase substantially, our results of operations could be adversely
affected.
We
have not yet developed comprehensive independent corporate governance.
As of the
date of this prospectus, we have no audit, compensation, or nominating
committees of our board of directors and have not established formal corporate
governance procedures. A lack of independent controls over our
corporate affairs may result in potential or actual conflicts of interest
between Mr. Qu and our stockholders. We presently have no policy to
resolve such conflicts. The absence of customary standards of
corporate governance may leave our stockholders without protections against
interested director transactions, conflicts of interest, if any, and similar
matters and any potential investors may be reluctant to provide us with funds
necessary to expand our operations.
We
have been heavily dependent on sales of four key products.
Four of
our products, namely Tongbi Capsules, Tongbi Tablets, Lung Nourishing Cream and
Tongshangning Tablets represented approximately 22%, 16%, 26%, and 15%,
respectively, of total sales for the fiscal year ended June 30,
2009. We expect that a significant portion of our future revenue will
continue to be derived from sales of these four products. If one or
more of these products were to become subject to a problem such as loss of
Certificates of Protected Variety of Traditional Chinese Medicine, unexpected
side effects, regulatory proceedings, publicity adversely affecting user
confidence or pressure from competing products, or if a new, more effective
treatment should be introduced, the negative impact on our revenues could be
significant. We held the Certificates of Protected Variety of
Traditional Chinese Medicine (Grade Two) issued by State Food and Drug
Administration of China (“SFDA”) for Tongbi Capsules and Shangtongning Tablets
which gave exclusive or near-exclusive rights to manufacture and distribute
these two medicines. These certificates expired in September 2009 and
we have filed an application for extending the protection period on March 12,
2009 for Tongbi Capsules. We can not assure you that we will obtain
the approvals to renew the Certificate of Protected Variety of Traditional
Chinese Medicine and the loss of such protection will have a material adverse
effect on our revenues. If we are unable to obtain approvals, these
products can be manufactured and sold by other pharmaceutical manufacturers in
China once the relevant protection periods elapse, which would increase our
competition and potentially have an adverse effect on our sales.
We
may not be able to adequately protect our intellectual property, which could
cause us to be less competitive and negatively impact our business.
We regard
our trademarks, trade secrets, patents and similar intellectual property as
critical to our success. We hold the trademark “Xian Ge” registered
with the PRC Trademark Bureau under the State Administration for Industry and
Commerce with a valid term effective through February 23, 2013. We
have received a patent in the PRC for lung nourishing cream with its production
method for the treatment of Lung-qi Deficiency Cough and Chronic
Bronchitis. If we are unable to obtain or maintain registered
intellectual property protections for our proprietary products or methods, these
products or methods could be infringed upon, which could materially adversely
affect our business.
We rely
on trademark, patent and trade secret law, as well as confidentiality agreement
with certain of our employees to protect our proprietary rights. For
senior managers, we include a standard confidentiality clause into the
employment agreement to prevent them from disclosing the formula or processing
procedure to outside parties. No assurance can be given that our intellectual
property will not be challenged, invalidated, infringed or circumvented, or that
our intellectual property rights will provide competitive advantages to
us. Any material impairment of our intellectual property rights could
have a material adverse effect on our business.
8
The
availability of counterfeit versions of our products could adversely affect our
sales volume, revenue and profitability and brand value.
The
availability of sales of counterfeits of our products in China could adversely
impact our sales and potentially damage the value and reputation of our
brands. For example, we recently discovered evidence of a counterfeit
Tongbi Capsule sold in China which we believe infringes on our intellectual
property rights. We have addressed this situation with applicable PRC
authorities and do not believe it will adversely effect our company, but similar
situations may arise in the future which could adversely impact our sales,
profitability and brand value. Additionally, consumers who mistake
counterfeit Tongbi Capsules or counterfeits of our other products for our
products may attribute quality and efficacy deficiencies in the counterfeit
product to our brands and discontinue purchasing our brands, which would have an
adverse effect on our sales and profitability.
We
face competition in the pharmaceutical market in the PRC and such competition
could cause our sales revenue and profits to decline.
According
to SFDA in China, there were approximately 5,071 pharmaceutical manufacturing
companies in the PRC as of the end of June 2004, of which approximately 3,237
manufacturers obtained certificates of Good Manufacturing Practices
Certification (“GMP”). After GMP certification became a mandatory
requirement on July 1, 2004, approximately 1,834 pharmaceutical manufacturers
were forced to cease production. Only the 3,237 pharmaceutical
manufacturers with GMP certifications may continue their manufacturing
operations. As of the end of 2006, there were 4,682 enterprises
manufacturing medicines and formulation in China. The certificates,
permits, and licenses required for pharmaceutical operation in the PRC create a
potentially significant barrier for new competitors seeking entrance into the
market. Despite these obstacles, we face competitors that will
attempt to create, or are already marketing, products in the PRC that are
similar to ours. Many of our current and potential competitors have
significantly longer operating histories and significantly greater managerial,
financial, marketing, technical and other competitive resources, as well as
greater name recognition, than we do. These competitors may be able
to respond more quickly to new or changing opportunities and customer
requirements and may be able to undertake more extensive promotional activities,
offer more attractive terms to customers or adopt more aggressive pricing
policies. We cannot assure you that we will be able to compete
effectively with current or future competitors or that the competitive pressures
we face will not harm our business.
Our
business depends and will depend substantially on the continuing efforts of our
present and future executive officers, and our business may be severely
disrupted if we lose, are unable to obtain or unable to replace their
services.
Our
future prospects depend substantially on the continued services of our
President, Chief Executive Officer and Chairman of the Board, Mr.
Qu. We have no employment agreement with Mr. Qu and do not maintain
key man life insurance on Mr. Qu’s life. We also have other corporate
officers and key employees, and if Mr. Qu or one or more of our future executive
officers or key employees are unable or unwilling to continue in their
positions, we may not be able to replace them readily, if at
all. Therefore, our business may be severely disrupted, and we may
incur additional expenses to recruit and retain new officers. In
addition, if any of our executives joins a competitor or forms a competing
company, we may lose some of our customers.
9
Our
business and growth will suffer if we are unable to hire and retain key
personnel that are in high demand.
Our
future performance depends on our ability to attract and retain highly skilled
chemists, pharmaceutical engineers, technical, marketing and sales personnel,
especially qualified personnel for our operations in China. Qualified
individuals are in high demand in China, and there are insufficient experienced
personnel to fill the demand. Therefore, we may not be able to
attract or retain the personnel we need to succeed. Our business
development would be hindered if we lost the services of some key
personnel.
Our
business is highly dependent on continually developing or acquiring new and
advanced products, technologies, and processes and failure to do so may cause us
to lose our competitiveness in the pharmaceutical industry and may cause our
profits to decline.
To remain
competitive in the pharmaceutical industry, it is important to continually
develop new and advanced products, technologies and processes. There
is no assurance that our competitors’ new products, technologies and processes
will not render our company’s existing products obsolete or
non-competitive. Our company’s competitiveness in the pharmaceutical
market therefore relies upon our ability to enhance our current products,
introduce new products, and develop and implement new technologies and
processes. Our company’s failure to technologically evolve and/or
develop new or enhanced products may cause us to lose our competitiveness in the
pharmaceutical industry and may cause our profits to decline. It is
likely that our efforts to grow our products lines will be focused on
acquisitions of such products from third parties. There are many
risks attendant to the acquisition of assets or companies, including
availability, pricing, competition and, if acquisitions are consummate,
integration. If we are unable to so acquire and integrate new
products, our revenue and profitability may suffer.
Our
research and development may be costly and/or untimely, and there are no
assurances that our research and development will either be successful or
completed within the anticipated timeframe, if ever at all.
We do not
presently rely on research and development activities as our business is focused
on expanding sales of our existing products. However, in the future,
the research and development of new products may play an important role for our
company. Development of new products requires significant research,
development and clinical testing efforts, and we currently have limited
resources to devote to and limited capabilities to conduct the development of
new products. We have only one full-time employee who is engaged in
research and development, so we mainly dependent on a third-party, Yantai
Tianzheng Medicine Research and Development Co., Ltd., to perform the limited
amount of research and development that we undertake. If research and
development activities become more important for us, and if we or third parties
that we retain are unable to perform research and development successfully, our
business and results of operations could be negatively impacted.
As of the
date of this prospectus, we have two products, namely Forsythia Capsule and Fern
Injection, under research and development. The research and
development of new products is costly and time consuming, and there are no
assurances that our research and development of new products will either be
successful or completed within the anticipated time frame, if ever at
all. There are also no assurances that if the product is developed,
that it will lead to actual commercialization and sales.
10
The
commercial performance of our products depends upon the degree of market
acceptance among the medical community and failure to attain market acceptance
among the medical community may have an adverse impact on our operations and
profitability.
The
commercial performance of our products depends upon the degree of market
acceptance among the medical community, such as hospitals and
physicians. Even if our products are approved by SFDA, and even if
our products are authorized to be eligible for reimbursement under Chinese
national medical insurance programs, there is no assurance that physicians will
prescribe or recommend our products to patients. Furthermore, a
product’s prevalence and use at hospitals may be contingent upon our
relationship with the medical community. The acceptance of our
products among the medical community may depend upon several factors, including
but not limited to, the product’s acceptance by physicians and patients as a
safe and effective treatment, cost effectiveness, potential advantages over
alternative treatments, and the prevalence and severity of side
effects. Failure to attain market acceptance among the medical
community may have an adverse impact on our operations and
profitability.
We
may not be able to obtain the regulatory approvals or clearances that are
necessary to commercialize our products.
The PRC
and other countries impose significant statutory and regulatory obligations upon
the manufacture and sale of pharmaceutical products. Each regulatory
authority typically has a lengthy approval process in which it examines
pre-clinical and clinical data and the facilities in which the product is
manufactured. Regulatory submissions must meet complex criteria to
demonstrate the safety and efficacy of the ultimate products. Addressing these
criteria requires considerable data collection, verification and analysis. We
may spend time and money preparing regulatory submissions or applications
without assurances as to whether they will be approved on a timely basis or at
all.
Our
product candidates, some of which are currently in the early stages of
development, will require significant additional development and pre-clinical
and clinical testing prior to their commercialization. These steps and the
process of obtaining required approvals and clearances can be costly and
time-consuming. If our potential products are not successfully developed, cannot
be proven to be safe and effective through clinical trials, or do not receive
applicable regulatory approvals and clearances, or if there are delays in the
process:
|
|
·
|
the
commercialization of our products could be adversely
affected;
|
|
|
·
|
any
competitive advantages of the products could be diminished;
and
|
|
|
·
|
revenues
or collaborative milestones from the products could be reduced or
delayed.
|
Governmental
and regulatory authorities may approve a product candidate for fewer indications
or narrower circumstances than requested or may condition approval on the
performance of post-marketing studies for a product candidate. Even if a product
receives regulatory approval and clearance, it may later exhibit adverse side
effects that limit or prevent its widespread use or that would force us to
withdraw the product from the market.
Any
marketed product and its manufacturer will continue to be subject to strict
regulation after approval. Results of post-marketing programs may limit or
expand the further marketing of products. Unforeseen problems with an approved
product or any violation of regulations could result in restrictions on the
product, including its withdrawal from the market and possible civil
actions.
11
In
manufacturing our products we will be required to comply with applicable good
manufacturing practices regulations, which include requirements relating to
quality control and quality assurance, as well as the maintenance of records and
documentation. If we cannot comply with regulatory requirements, including
applicable good manufacturing practice requirements, we may not be allowed to
develop or market the product candidates. If we or our manufacturers fail to
comply with applicable regulatory requirements at any stage during the
regulatory process, we may be subject to sanctions, including fines, product
recalls or seizures, injunctions, refusal of regulatory agencies to review
pending market approval applications or supplements to approve applications,
total or partial suspension of production, civil penalties, withdrawals of
previously approved marketing applications and criminal
prosecution.
Our
current and future products may have inadvertent and/or harmful side effects
which would expose us to the risks of litigation and a loss of
revenue.
All
medicines have certain side effects. Although all of our medicines
sold on market have passed proper testing and are approved by SFDA, the products
may still inadvertently adverse effects on the health of the consumers. If such
side effect is identified after marketing and sale of the products, the products
may be required to be withdrawn from the market, or have a change in labeling.
If a product liability claim is brought against us, it may, regardless of merit
or eventual outcome, result in damage to our reputation, breach of contracts
with consumers, decreased demand for our products, costly litigation and loss of
revenue.
Natural
disasters, weather conditions and other environmental factors affect our raw
material supply, and a reduction in the quality or quantity of our herb supplies
may have material adverse consequences on our financial results.
Our
business may be adversely affected by weather and environmental factors beyond
our control, such as natural disasters and adverse weather
conditions. The production of our products depends on the
availability of raw materials, a significant portion of which are herbs.
These herbs tend to be very sensitive crops, which can be readily damaged
by harsh weather, by disease, and by pests. If our suppliers’ crops are
destroyed by drought, flood, storm, blight, or the other woes of farming, we
will not be able to meet the demands of our customers, which will have a
material adverse effect on our business and financial condition and results.
Our
certificates, permits, and license are subject to governmental control and
renewal, and the failure to obtain renewal would cause all or part of our
operation to be suspended and have a material adverse effect on our financial
condition.
We are
subject to various PRC laws and regulations pertaining to the pharmaceutical
industry. We have obtained certain certificates, permits, and
licenses required for the operation of a pharmaceutical enterprise and the
manufacturing and distribution of pharmaceutical products in the
PRC. Some of the permits and license have expired or are about to
expire. We hold a Permit for the Production of Medicine (Lu
Zb20050330) issued by Shandong Branch of SFDA on January 1, 2006 which allows us
to engage in the production of tablets, capsules, granules, syrup, concentrated
decoctions, tincture (for oral use) and medical wine. Such permit
expires on December 31, 2010 and is material to our business. We also
hold a GMP Certificate (No. Lu K0587) issued by Shandong Branch of SFDA on June
18, 2009, the scope of inspection of which is tablets, capsules, granules,
syrup, concentrated decoctions, tincture and medical wine. Such
certificate expires on June, 14, 2014. The Permit for the Production
of Medicine and GMP certificates are each valid for a term of five years and
must be renewed before their expiration.
12
We hold a
Drug Approval Number (“DAN”) for each of our products, and the valid terms of
such DANs have expired. We submitted the applications for
re-registration on June 29, 2007 which were accepted by SFDA, although the
approvals have not yet been granted. We have been advised that the
approval processes for these drugs have been started to be reviewed by the
Shandong Branch of SFDA. During the renewal period, we will be
permitted to continue manufacturing these drugs as if the renewals had been
approved. Our license to produce medical wine has a term valid
through December 31, 2010.
During
the application or renewal process for our licenses and permits, we will be
evaluated and re-evaluated by the appropriate governmental authorities and must
comply with the prevailing standards and regulations, which may change from time
to time. In the event that we are not able to obtain or renew
the certificates, permits and licenses, all or part of our operations may be
suspended by the government, which would have a material adverse effect on our
business and financial condition. Furthermore, if escalating
compliance costs associated with governmental standards and regulations restrict
or prohibit any part of our operations, it may adversely affect our results of
operations and profitability.
We held
the Certificates of Protected Variety of Traditional Chinese Medicine (Grade
Two) (the “Certificate of Protection”) issued by SFDA for two of our products,
Tongbi Capsules and Shangtongning Tablets. The protection periods for
both Tongbi Capsules and Shangtongning Tablets expired in September
2009. We have submitted application to extend the protection periods
for Tongbi Capsules to extend such protection period on March 12, 2009 and SFDA
has recently started its review process. We have decided not to
submit extension application of Shangtongning Tablets, because the SFDA will not
approve a Certificate of Protection for Shangtongning Tablets or any other
products that are currently produced by more than three manufacturers in China
according to applicable Chinese SFDA regulations. Our inability to regain the
Certificate of Protection for Tongbi Capsules, which is one of our leading
products, and the loss of the Certificate of Protection for our product
Shangttongning Tablets, may grant other manufactures the right to produce
similar products, which would result in the loss of competitive advantage and
could adversely impact our sales results.
Our
failure to fully comply with PRC labor laws, including laws relating to social
insurance, may expose us to potential liability and increased
costs.
Companies
operating in China must comply with a variety of labor laws, including certain
pension, health insurance, unemployment insurance and other welfare-oriented
payment obligations. Our failure to comply with these laws could have
a material adverse effect on our business. For example, we are
currently paying social insurance for our 105 full-time employees. We also
have 304 sales representatives that we believe we are not required to pay social
insurance for as these sales representatives are not legally employees of ours,
but are rather independent contractors. We have not paid social insurance
for 195 of our full-time employees whose personal identification files cannot be
transferred to us since they are not registered residents in Yantai, Shandong
Province, and as an alternative we have paid these employees compensations
included in their monthly salary with an amount equals to the amount of monthly
social insurance that we are required to pay and the employees could pay the
social insurance by themselves. We believe these employees have been
covered by social insurance and we are not required to make any contributions to
the government in addition to the amount we have paid to these
employees. However, our interpretation of these requirements may be
wrong, and the PRC regulatory authorities may not take the same view as we do on
this subject. If the PRC regulatory authorities take the view that we are
required to pay social insurance for our independent contractors or other
employees, our failure to make previous payments may be in violation of
applicable PRC labor laws and we cannot assure you that PRC governmental
authorities will not impose penalties on us for failure to comply. In
addition, in the event that any current or former employee files a complaint
with the PRC government, we may be subject to making up the social insurance
payment obligations as well as paying administrative fines. The total cost
of these payments and any related fines or penalties could be very significant
and could have a material adverse effect on our working
capital.
13
In
addition, the new PRC Labor Contract Law took effect January 1, 2008 and governs
standard terms and conditions for employment, including termination and lay-off
rights, contract requirements, compensation levels and consultation with labor
unions, among other topics. In addition, the law limits
non-competition agreements with senior management and other employees who have
access to confidential information to two years and imposes restrictions or
geographical limits. This new labor contract law will increase our
labor costs, which could adversely impact our results of
operations.
We
are subject to PRC government price control of drugs which may limit our
profitability and even cause us to stop manufacturing certain
products.
The State
Development and Reform Commission of the PRC (“SDRC”) and the price
administration bureaus of the relevant provinces of the PRC in which the
pharmaceutical products are manufactured are responsible for the retail price
control over our pharmaceutical products. The SDRC sets the price
ceilings for certain pharmaceutical products in the PRC. All of our products
except those under the protection periods are subject to such price controls as
of the date of this Memorandum and we prices our medicines well under
government-mandated caps. There is no assurance that whether our other products
will remain unaffected by the price control. Where our products are
subject to a price ceiling, we will need to adjust the product price to meet the
requirement and to accommodate for the pricing of competitors in the competition
for market shares. The price ceilings set by the SDRC may limit our
profitability, and in some instances, such as where the price ceiling is below
production costs, may cause us to stop manufacturing certain products which may
adversely affect our results of operations.
Because
we may not be able to obtain business insurance in the PRC, we may not be
protected from risks that are customarily covered by insurance in the United
States.
Business
insurance is not readily available in the PRC. To the extent that we
suffer a loss of a type which would normally be covered by insurance in the
United States, such as product liability and general liability insurance, we
would incur significant expenses in both defending any action and in paying any
claims that result from a settlement or judgment. We have not
obtained fire, casualty and theft insurance, and there is no insurance coverage
for our raw materials, goods and merchandise, furniture and buildings in
China. Any losses incurred by us will have to be borne by us without
any assistance, and we may not have sufficient capital to cover material damage
to, or the loss of, our production facility due to fire, severe weather, flood
or other cause, and such damage or loss would have a material adverse effect on
our financial condition, business and prospects.
We
may be subject to product liability claims, for which we have no
insurance.
We may
produce products which inadvertently have an adverse pharmaceutical effect on
the health of individuals. Existing laws and regulations in China do not
require us to maintain third party liability insurance to cover product
liability claims. However, if a product liability claim is brought against
us, it may, regardless of merit or eventual outcome, result in damage to our
reputation, breach of contracts with our customers, decreased demand for our
products, costly litigations, product recalls, loss of revenue, and our
inability to commercialize some products.
14
Our
indemnification obligations could adversely affect our business, financial
condition and results of operations.
Our
governing documents require us to indemnify our current and former directors,
officers, employees and agents against most actions of a civil, criminal,
administrative or investigative nature. Generally, we are required to
advance indemnification expenses prior to any final adjudication of an
individual’s culpability. The expense of indemnifying our current and
former directors, officers and employees and agents in their defense or related
expenses as a result of any actions related to the internal investigation and
financial restatement may be significant and in excess of any insurance coverage
we may have. As such, there is a risk that our indemnification
obligations could divert needed financial resources and may adversely affect our
business, financial condition and results of operations.
Potential
environmental liability could have a material adverse effect on our operations
and financial condition.
As a
manufacturer, we are subject to various Chinese environmental laws and
regulations on air emission, waste water discharge, solid wastes and
noise. We are in the process of applying for Pollution Discharge
Permit, other than that we believe that our operations are in substantial
compliance with current environmental laws and regulations. We can not assure
you that we may not be able to comply with these regulations at all times as the
Chinese environmental legal regime is evolving and becoming more
stringent. Therefore, if the Chinese government imposes more
stringent regulations in future, we may have to incur additional and potentially
substantial costs and expenses in order to comply with new regulations, which
may negatively affect our results of operations. Furthermore, no
assurance can be given that all potential environmental liabilities have been
identified or properly quantified or that any prior owner, operator, or tenant
has not created an environmental condition unknown to us. If we fail
to comply with any of the present or future environmental regulations in any
material aspects, we may suffer from negative publicity and be subject to claims
for damages that may require us to pay substantial fines or have our operations
suspended or even be forced to cease operations.
Risks
Relating to the Our Corporate Structure
Our
corporate structure, in particular the VIE Agreements, are subject to
significant risks, as set forth in the following risk factors.
The
PRC government may determine that the VIE Agreements which we utilize to control
our operating subsidiary are not in compliance with applicable PRC laws, rules
and regulations and that they are therefore unenforceable.
In the
PRC it is widely understood that foreign invested enterprises are forbidden or
restricted to engage in certain businesses or industries which are sensitive to
the economy. As we intend to centralize our management and operation
in the PRC without being restricted to conduct certain business activities which
are important for our current or future business but are restricted or might be
restricted in the future, we believe our VIE Agreements will be essential for
our business operation. In order for WFOE to manage and operate our
business through Bohai in the PRC, the VIE Agreements were entered into under
which almost all the business activities of Bohai are managed and operated by
WFOE and almost all economic benefits and risks arising from the business of
Bohai are transferred to WFOE.
There are
risks involved with the operation of Bohai under the VIE
Agreements. We have been advised by PRC legal counsel that if the PRC
government determines the VIE Agreement used to control the operating company to
be unenforceable as they circumvent the PRC restrictions relating to foreign
investment restrictions, the relevant regulatory authorities would have broad
discretion in dealing with such breach, including:
15
|
|
·
|
imposing
economic penalties;
|
|
|
·
|
discontinuing
or restricting the operations of WFOE or
Bohai;
|
|
|
·
|
imposing
conditions or requirements in respect of the VIE Agreements with which
WFOE may not be able to comply;
|
|
|
·
|
requiring
us to restructure the relevant ownership structure or
operations;
|
|
|
·
|
taking
other regulatory or enforcement actions that could adversely affect our
business; and
|
|
|
·
|
revoking
the business license and/or the licenses or certificates of WFOE, and/or
voiding the VIE Agreements.
|
Any of
these actions could have a material adverse impact on our business, financial
condition and results of operations.
We
depend upon the VIE Agreements in conducting our production, manufacturing and
distribution of traditional Chinese herbal medicines in the PRC, which may not
be as effective as direct ownership.
We
conduct our production, manufacturing and distribution of traditional Chinese
herbal medicines in the PRC and generate the revenues through the VIE
Agreements. The VIE Agreements may not be as effective in providing
us with control over Bohai as direct ownership. The VIE Agreements
are governed by PRC laws and provide for the resolution of disputes through
arbitration proceedings pursuant to PRC laws. Accordingly, the VIE
Agreements will be interpreted in accordance with PRC laws. If Bohai
or its shareholders fail to perform the obligations under the VIE Agreements, we
may have to rely on legal remedies under PRC laws, including seeking specific
performance or injunctive relief, and claiming damages, and there is a risk that
we may be unable to obtain these remedies. The legal environment in
China is not as developed as in other jurisdictions. As a result,
uncertainties in the PRC legal system could limit our ability to enforce the VIE
Agreements.
The
pricing arrangement under the VIE Agreements may be challenged by the PRC tax
authorities.
We could
face adverse tax consequences if the PRC tax authorities determine that the VIE
Agreements were not entered into based on arm’s length
negotiations. If the PRC tax authorities determine that the VIE
Agreements were not entered into on an arm’s length basis, they may adjust the
income and expenses of our company for PRC tax purposes which could result in
higher tax liability.
We
rely on the approval certificates and business license held by Bohai and any
deterioration of the relationship between WFOE and Bohai could materially and
adversely affect the overall business operation of our company.
Pursuant
to the VIE Agreements, our production, manufacturing and distribution of
traditional Chinese herbal medicines in China is undertaken on the basis of the
approvals, certificates and business license as well as other requisite licenses
held by Bohai. There is no assurance that Bohai will be able to renew
its licenses or certificates when their terms expire with substantially similar
terms as the ones they currently hold.
16
Further,
our relationship with Bohai is governed by the VIE Agreements, which are
intended to provide us, through our indirect ownership of WFOE, with effective
control over the business operations of Bohai. However, the VIE
Agreements may not be effective in providing control over the applications for
and maintenance of the licenses required for our business
operations. Bohai could violate the VIE Agreements, go bankrupt,
suffer from difficulties in its business or otherwise become unable to perform
its obligations under the VIE Agreements and, as a result, our operations,
reputation, business and stock price could be severely harmed.
If
WFOE exercises the purchase options over Bohai’s equity pursuant to the VIE
Agreements, the payment of purchase prices could materially and adversely affect
the financial position of our company.
Under the
VIE Agreements, WFOE holds an option to purchase all or a portion of the equity
of Bohai at a price, pro rata in case of not all, based on the capital paid in
by the Bohai shareholders (namely, $2.94 million or RMB 20 million
). In the case that applicable PRC laws and regulations require an
appraisal of the equity interest or provide other restriction on the purchase
price, the purchase price shall be the lowest price permitted under the
applicable PRC laws and regulations. As Bohai is already a contractually
controlled affiliate to our company, WFOE’s purchase of Bohai’s equity would not
bring immediate benefits to our company and the exercise of the option and
payment of the purchase prices could adversely affect the financial position of
our company.
Risks
Associated With Doing Business in China
There
are substantial risks associated with doing business in China, some of which are
addressed in the following risk factors.
The
Chinese government exerts substantial influence over the manner in which we must
conduct our business activities.
We are
dependent on our relationship with the local government in the province in which
we operate our business. The Chinese government has exercised and
continues to exercise substantial control over virtually every sector of the
Chinese economy through regulation and state ownership. Our ability
to operate in China may be harmed by changes in its laws and regulations,
including those relating to taxation, environmental regulations, land use
rights, property and other matters. The central or local governments
of in the PRC jurisdictions may impose new, stricter regulations or
interpretations of existing regulations that would require additional
expenditures and efforts on our part to ensure our compliance with such
regulations or interpretations. Accordingly, government actions in
the future, including any decision not to continue to support recent economic
reforms and to return to a more centrally planned economy or regional or local
variations in the implementation of economic policies, could have a significant
effect on economic conditions in China or particular regions thereof, and could
require us to divest ourselves of any interest we then hold in Chinese
properties.
Future
inflation in China may inhibit our ability to conduct business in China. In
recent years, the Chinese economy has experienced periods of rapid expansion and
high rates of inflation. Rapid economic growth can lead to growth in
the money supply and rising inflation. If prices for our products
rise at a rate that is insufficient to compensate for the rise in the costs of
supplies, it may have an adverse effect on profitability. These
factors have led to the adoption by Chinese government, from time to time, of
various corrective measures designed to restrict the availability of credit or
regulate growth and contain inflation. High inflation may in the
future cause Chinese government to impose controls on credit and/or prices, or
to take other action, which could inhibit economic activity in China, and
thereby harm the market for our products.
17
Our
operations and assets in China are subject to significant political and economic
uncertainties and the company may lose all of its assets and operations if the
Chinese government alters its policies to further restrict foreign participation
in business operating in the PRC.
Changes
in PRC laws and regulations, or their interpretation, or the imposition of
confiscatory taxation, restrictions on currency conversion, imports and sources
of supply, devaluations of currency or the nationalization or other
expropriation of private enterprises could have a material adverse effect on our
business, results of operations and financial condition. Under its
current leadership, the Chinese government has been pursuing economic reform
policies that encourage private economic activities and greater economic
decentralization. There is no assurance, however, that the Chinese
government will continue to pursue these policies, or that it will not
significantly alter these policies from time to time without notice. We may lose all of our
assets and operations if the Chinese government alters its policies to further
restrict foreign participation in business operating in the PRC.
We derive all of our sales in China
and a slowdown or other adverse developments in the PRC economy may materially
and adversely affect our customers, demand for our products and our
business.
All of
our sales are generated in China. We anticipate that sales of our
products in China will continue to represent all of our total sales in the near
future. Although the PRC economy has grown significantly in recent
years, we cannot assure you that such growth will continue. The
industry which we are involved in the PRC is relatively new and growing, but we
do not know how sensitive we are to a slowdown in economic growth or other
adverse changes in the PRC economy which may affect demand for our
products. In addition, the Chinese government also exercises
significant control over Chinese economic growth through the allocation of
resources, controlling payment of foreign currency-denominated obligations,
setting monetary policy and providing preferential treatment to particular
industries or companies. Efforts by the Chinese government to slow
the pace of growth of the Chinese economy could result in reduced demand for our
products. A slowdown in overall economic growth, an economic downturn
or recession or other adverse economic developments in the PRC may materially
reduce the demand for our products and materially and adversely affect our
business.
Currency fluctuations and
restrictions on currency exchange may adversely affect our business, including
limiting our ability to convert Chinese Renminbi into foreign currencies and, if
Chinese Renminbi were to decline in value, reducing our revenue in U.S. dollar
terms.
Our
reporting currency is the U.S. dollar and our operations in China use their
local currency as their functional currencies. Substantially all of
our revenue and expenses are in the Chinese currency, the
Renminbi. We are subject to the effects of exchange rate fluctuations
with respect to any of these currencies. For example, the value of
the Renminbi depends to a large extent on Chinese government policies and
China’s domestic and international economic and political developments, as well
as supply and demand in the local market. Since 1994, the official
exchange rate for the conversion of the Renminbi to the U.S. dollar had
generally been stable and the Renminbi had appreciated slightly against the U.S.
dollar. In July 2005, the Chinese government changed its policy of
pegging the value of the Renminbi to the U.S. dollar. Under this
policy, which was halted in 2008 due to the worldwide financial crisis, the
Renminbi was permitted to fluctuate within a narrow and managed band against a
basket of certain foreign currencies. In June 2010, the Chinese
government announced its intention to again allow the Renminbi to fluctuate
within the 2005 parameters. It is possible that the Chinese
government could adopt an even more flexible currency policy, which could result
in more significant fluctuation of Renminbi against the U.S. dollar, or it could
adopt a more restrictive policy. We can offer no assurance that the
Renminbi will be stable against the U.S. dollar or any other foreign
currency.
18
Our
financial statements are translated into U.S. dollars at the average exchange
rates in each applicable period. To the extent the U.S. dollar
strengthens against foreign currencies, the translation of these foreign
currencies denominated transactions results in reduced revenue, operating
expenses and net income for our international operations. Similarly,
to the extent the U.S. dollar weakens against foreign currencies, the
translation of these foreign currency denominated transactions results in
increased revenue, operating expenses and net income for our international
operations. We are also exposed to foreign exchange rate fluctuations
as we convert the financial statements of our foreign consolidated subsidiaries
into U.S. dollars in consolidation. If there is a change in foreign
currency exchange rates, the conversion of the foreign consolidated
subsidiaries’ financial statements into U.S. dollars will lead to a translation
gain or loss which is recorded as a component of other comprehensive
income. In addition, we have certain assets and liabilities that are
denominated in currencies other than the relevant entity’s functional
currency. Changes in the functional currency value of these assets
and liabilities create fluctuations that will lead to a transaction gain or
loss. We have not entered into agreements or purchased instruments to
hedge our exchange rate risks, although we may do so in the
future. The availability and effectiveness of any hedging transaction
may be limited and we may not be able to hedge our exchange rate
risks.
The
State Administration of Foreign Exchange (“SAFE”) restrictions on currency
exchange may limit our ability to receive and use our sales revenue effectively
and to pay dividends, and the restrictions may cause a delay in payment of
interest on the Notes.
All of
our sales revenue and expenses are denominated in the Chinese currency,
Renminbi. Under PRC law, the Renminbi is currently convertible under
the “current account,” which includes dividends and trade and service-related
foreign exchange transactions, but not under the “capital account,” which
includes foreign direct investment and loans. Currently, our PRC
operating subsidiary, Bohai, may purchase foreign currencies for settlement of
current account transactions, including payments of dividends to us, without the
approval of SAFE, by complying with certain procedural
requirements. However, the relevant PRC government authorities may
limit or eliminate our ability to purchase foreign currencies in the
future. Since a significant amount of our future revenue will be
denominated in Renminbi, any existing and future restrictions on currency
exchange may limit our ability to utilize revenue generated in Renminbi to fund
our business activities outside China that are denominated in foreign
currencies.
All of
our income is derived from the consulting fees we receive from Bohai through the
VIE Agreements. SAFE restrictions may delay the payment of dividends,
since we have to comply with certain procedural requirement and we may
experience difficulties in completing the administrative procedures necessary to
obtain and remit foreign currency for the payment of dividends from the profits
of WFOE, and it thus may delay our payment of interest to the Notes
holders.
Foreign
exchange transactions by PRC operating subsidiaries under the capital account
continue to be subject to significant foreign exchange controls and require the
approval of or need to register with PRC government authorities, including
SAFE. In particular, if Bohai, our PRC operating subsidiary, borrows
foreign currency through loans from us or other foreign lenders, these loans
must be registered with SAFE, and if we finance Bohai by means of additional
capital contributions, these capital contributions must be approved by certain
government authorities, including the Ministry of Commerce, or their respective
local counterparts. These limitations could affect Bohai’s ability to
obtain foreign exchange through debt or equity financing.
19
The PRC
government also may at its discretion restrict access in the future to foreign
currencies for current account transactions. If the foreign exchange
control system prevents us from obtaining foreign currency, we may be unable to
pay the interest and principal on the Notes, pay dividends or meet obligations
that may be incurred in the future that require payment in foreign
currency.
The
PRC State Administration of Foreign Exchange restrictions on the use of offshore
holding companies in mergers and acquisitions in China may create regulatory
uncertainties that could restrict or limit our ability to operate.
In
November 2005, SAFE issued a public notice, known as Circular 75, concerning
foreign exchange registrations that are required in order to use of offshore
holding companies in mergers and acquisitions in China. The public notice
provides that if an offshore company controlled by PRC residents intends to
acquire a PRC company, such acquisition will be subject to registration with the
relevant foreign exchange authorities. The public notice also suggested
that registration with the relevant foreign exchange authorities is required for
any sale or transfer by the PRC residents of shares in an offshore holding
company that owns an onshore company. PRC residents must each submit a
registration form to the local SAFE branch with respect to their ownership
interests in the offshore company, and must also file an amendment to such
registration if the offshore company experiences material events, such as
changes in the share capital, share transfer, mergers and acquisitions, spin-off
transactions or use of assets in China to guarantee offshore
obligations.
Internal
implementing guidelines issued by SAFE, which became public in June 2007 (known
as Notice 106), expanded the reach of Circular 75 by: (i) purporting to cover
the establishment or acquisition of control by PRC residents of offshore
entities which merely acquire “control” over domestic companies or assets, even
in the absence of legal ownership; (ii) adding requirements relating to the
source of the PRC resident’s funds used to establish or acquire the offshore
entity; (iii) covering the use of existing offshore entities for offshore
financings; (iv) purporting to cover situations in which an offshore special
purpose vehicle, or SPV, establishes a new subsidiary in China or acquires an
unrelated company or unrelated assets in China; and (v) making the domestic
affiliate of the SPV responsible for the accuracy of certain documents which
must be filed in connection with any such registration, notably, the business
plan which describes the overseas financing and the use of proceeds.
Amendments to registrations made under Circular 75 are required in connection
with any increase or decrease of capital, transfer of shares, mergers and
acquisitions, equity investment or creation of any security interest in any
assets located in China to guarantee offshore obligations, and Notice 106 makes
the offshore SPV jointly responsible for these filings. In the case of an
SPV which was established, and which acquired a related domestic company or
assets, before the implementation date of Circular 75, as applied by SAFE in
accordance with Notice 106, may result in fines and other penalties under PRC
laws for evasion of applicable foreign exchange restrictions. Any such
failure could also result in the SPV’s affiliates being impeded or prevented
from distributing their profits and the proceeds from any reduction in capital,
share transfer or liquidation to the SPV, or from engaging in other transfers of
funds into or out of China.
The PRC
regulatory authorities may take the view that our acquisition of indirect
ownership and controlling interest in Bohai through VIE arrangements shall be
subject to SAFE approval and registration. Any adverse action taken against us
by PRC regulatory authorities could significantly and negatively impact our
operations and the trading market for our common stock.
20
PRC
regulations and potential registration requirements relating to acquisitions of
PRC companies by foreign entities may create regulatory uncertainties that could
restrict or limit our ability to operate. Similarly, our failure to obtain
the prior approval of PRC authorities for our January 2010 private placement and
the listing and trading of our common stock could have a material adverse effect
on our business, operating results, reputation and trading price of our common
stock.
On August
8, 2006, the PRC Ministry of Commerce (“MOC”), joined by the State-owned Assets
Supervision and Administration Commission of the State Council, the State
Administration of Taxation, the State Administration for Industry and Commerce,
the China Securities Regulatory Commission (“CSRC”) and SAFE, released a
substantially amended version of the Provisions for Foreign Investors to Merge
with or Acquire Domestic Enterprises (the “Revised M&A Regulations”), which
took effect September 8, 2006. These new rules significantly revised
China’s regulatory framework governing onshore-to-offshore restructurings and
foreign acquisitions of domestic enterprises. These new rules signify
greater PRC government attention to cross-border merger, acquisition and other
investment activities, by confirming MOC as a key regulator for issues related
to mergers and acquisitions in China and requiring MOC approval of a broad range
of merger, acquisition and investment transactions. Further, the new rules
establish reporting requirements for acquisition of control by foreigners of
companies in key industries, and reinforce the ability of the Chinese government
to monitor and prohibit foreign control transactions in key
industries.
Among
other things, the revised M&A Regulations include new provisions that
purport to require that an offshore SPV, formed for listing purposes and
controlled directly or indirectly by PRC companies or individuals must obtain
the approval of the CSRC prior to the listing and trading of such SPV’s
securities on an overseas stock exchange. On September 21, 2006, the CSRC
published on its official website procedures specifying documents and materials
required to be submitted to it by SPVs seeking CSRC approval of their overseas
listings. However, the application of this PRC regulation remains unclear
with no consensus currently existing among the leading PRC law firms regarding
the scope and applicability of the CSRC approval requirement.
Our
principal stockholder, Glory Period, is 100% owned by Joshua Tan, a Singapore
citizen. As of the date of this prospectus, Glory Period holds 55% of our
outstanding common stock. Mr. Tan and Mr. Qu (the principal founder of
Bohai and our Chairman, President and Chief Executive Officer) entered into the
Call Option Agreement on December 7, 2009 by which Mr. Qu has an option to
acquire all the issued and outstanding shares of Glory Period within three years
for nominal consideration. Chance High, as the wholly owned subsidiary of
our company, formed WFOE on November 23, 2009 and WFOE obtained effective and
substantial control over Bohai further through executing the VIE Agreements on
December 7, 2009 by and among WFOE, Bohai and the three shareholders of Bohai
(including Mr. Qu). The PRC regulatory authorities may take the view that
entry into the VIE Agreements by WFOE and Bohai resulting in Mr. Qu, a PRC
resident becoming the majority owner and effective controlling party of our
company which acquired 100% indirect ownership of Bohai. The PRC
regulatory authorities may also take the view that the relevant parties should
fully disclose to SAFE or MOC of the overall restructuring arrangements, the
existence of the Share Exchange and related VIE Agreements. If the PRC
regulatory authorities take the view that the Share Exchange and VIE arrangement
constitutes a reverse ,merger or round-trip investment under the M&A
Regulations, we cannot assure you we may be able to obtain the approval required
from the national offices of MOC.
21
If the
PRC regulatory authorities take the view that the Share Exchange and the VIE
Agreements constitutes a reverse merger acquisition or round-trip investment
without the approval of the national offices of MOC, they could invalidate the
Share Exchange and VIE Agreements. Additionally, the PRC regulatory
authorities may take the view that any public offering plan of us will require
the prior approval of CSRC. If we cannot obtain MOC or CSRC approval in
case we are required to do so, our business and financial performance will be
materially adversely affected. We may also face regulatory actions or
other sanctions from the MOC or other PRC regulatory agencies. These
regulatory agencies may impose fines and penalties on our operations in the PRC,
limit our operating privileges in the PRC, delay or restrict the repatriation of
the proceeds from the Private Placement into the PRC, or take other actions that
could have a material adverse effect on our business, financial condition,
results of operations, reputation and prospects, as well as the trading price of
our common stock.
Also, if
later the CSRC requires that we obtain its approval, we may be unable to obtain
a waiver of the CSRC approval requirements, if and when procedures are
established to obtain such a waiver. Any uncertainties and/or negative publicity
regarding this CSRC approval requirement could have a material adverse effect on
the trading price of our common stock. Furthermore, published news reports
in China recently indicated that the CSRC may have curtailed or suspended
overseas listings for Chinese private companies. These news reports have created
further uncertainty regarding the approach that the CSRC and other PRC
regulators may take with respect to transactions that we have engaged in our may
in the future engage in. Any adverse action taken against us by PRC
regulatory authorities could significantly and negatively impact our operations
and the trading market for our common stock.
Because
our assets are located outside of the United States and half of our directors,
including our Chairman, and the majority of our officers reside outside of the
United States, it may be difficult for you to enforce your rights based on the
United States federal securities laws against us and these persons in the United
States or to enforce judgments of United States courts against us or him in the
PRC.
Our
Chairman of the Board and principal executive officer, Mr. Qu, resides outside
of the United States in China. In addition, another of our directors and a
majority of our officers are also located in China. Furthermore, our
operating subsidiary is located in the PRC and all of its assets are located
outside of the United States. China does not have a treaty with United
States providing for the reciprocal recognition and enforcement of judgments of
courts. It may therefore be difficult for investors in the United States
to enforce their legal rights based on the civil liability provisions of the
United States federal securities laws against us in the courts of either the
United States or the PRC and, even if civil judgments are obtained in courts of
the United States, to enforce such judgments in the PRC courts. Further,
it is unclear if extradition treaties now in effect between the United States
and the PRC would permit effective enforcement against us or our officers and
directors of criminal penalties, under the United States federal securities laws
or otherwise.
We
may have limited legal recourse under PRC laws if disputes arise under our
contracts with third parties.
The
Chinese government has enacted some laws and regulations dealing with matters
such as corporate organization and governance, foreign investment, commerce,
taxation and trade. However, their experience in implementing,
interpreting and enforcing these laws and regulations is limited, and our
ability to enforce commercial claims or to resolve commercial disputes is
unpredictable. If our new business ventures are unsuccessful, or other
adverse circumstances arise from these transactions, we face the risk that the
parties to these ventures may seek ways to terminate the transactions, or, may
hinder or prevent us from accessing important information regarding the
financial and business operations of these acquired companies. The
resolution of these matters may be subject to the exercise of considerable
discretion by agencies of the Chinese government, and forces unrelated to the
legal merits of a particular matter or dispute may influence their
determination. Any rights we may have to specific performance, or to seek an
injunction under PRC laws, in either of these cases, are severely limited, and
without a means of recourse by virtue of the Chinese legal system, we may be
unable to prevent these situations from occurring. The occurrence of any
such events could have a material adverse effect on our business, financial
condition and results of operations. Although legislation in China over
the past 30 years has significantly improved the protection afforded to various
forms of foreign investment and contractual arrangements in China, these laws,
regulations and legal requirements are relatively new and their interpretation
and enforcement involve uncertainties, which could limit the legal protection
available to us, and foreign investors, including you. The inability to
enforce or obtain a remedy under any of our future agreements could result in a
significant loss of business, business opportunities or capital and could have a
material adverse impact on our operations.
22
We
must comply with the Foreign Corrupt Practices Act.
We are
required to comply with the United States Foreign Corrupt Practices Act, which
prohibits U.S. companies from engaging in bribery or other prohibited payments
to foreign officials for the purpose of obtaining or retaining business.
Foreign companies, including some of our competitors, are not subject to these
prohibitions. Corruption, extortion, bribery, pay-offs, theft and other
fraudulent practices occur from time-to-time in mainland China. If our
competitors engage in these practices, they may receive preferential treatment
from personnel of some companies, giving our competitors an advantage in
securing business or from government officials who might give them priority in
obtaining new licenses, which would put us at a disadvantage. Although we
inform our personnel that such practices are illegal, we have not established
formal policies or procedures for prohibiting or monitoring this conduct, and we
can not assure you that our employees or other agents will not engage in such
conduct for which we might be held responsible. If our employees or other
agents are found to have engaged in such practices, we could suffer severe
penalties.
If
we make equity compensation grants to persons who are PRC citizens, they may be
required to register with SAFE. We may also face regulatory uncertainties
that could restrict our ability to adopt equity compensation plans for our
directors and employees and other parties under PRC laws.
On April
6, 2007, SAFE issued the “Operating Procedures for Administration of Domestic
Individuals Participating in the Employee Stock Ownership Plan or Stock Option
Plan of An Overseas Listed Company, also know as “Circular 78.” It is
not clear whether Circular 78 covers all forms of equity compensation plans or
only those which provide for the granting of stock options. For any plans which
are so covered and are adopted by a non-PRC listed company, such as our company,
after April 6, 2007, Circular 78 requires all participants who are PRC citizens
to register with and obtain approvals from SAFE prior to their participation in
the plan. In addition, Circular 78 also requires PRC citizens to register
with SAFE and make the necessary applications and filings if they participated
in an overseas listed company’s covered equity compensation plan prior to April
6, 2007. We believe that the registration and approval requirements
contemplated in Circular 78 will be burdensome and time consuming.
In the
future, we may adopt an equity incentive plan and make numerous stock option
grants under the plan to our officers, directors and employees, some of whom are
PRC citizens and may be required to register with SAFE. If it is
determined that any of our equity compensation plans are subject to Circular 78,
failure to comply with such provisions may subject us and participants of our
equity incentive plan who are PRC citizens to fines and legal sanctions and
prevent us from being able to grant equity compensation to our PRC employees. In
that case, our ability to compensate our employees and directors through equity
compensation would be hindered and our business operations may be adversely
affected.
23
Due
to various restrictions under PRC laws on the distribution of dividends by our
PRC operating companies, we may not be able to pay dividends to our
stockholders.
The
Wholly-Foreign Owned Enterprise Law (1986), as amended and the Wholly-Foreign
Owned Enterprise Law Implementing Rules (1990), as amended and the Company Law
of the PRC (2006) contain the principal regulations governing dividend
distributions by wholly foreign owned enterprises. Under these regulations,
wholly foreign owned enterprises, such as WFOE, may pay dividends only out of
their accumulated profits, if any, determined in accordance with PRC accounting
standards and regulations. Additionally, WFOE is required to set aside a certain
amount of their accumulated profits each year, if any, to fund certain reserve
funds. These reserves are not distributable as cash dividends except in
the event of liquidation and cannot be used for working capital
purposes.
Furthermore,
if our consolidated subsidiaries in China incur debt on their own in the future,
the instruments governing the debt may restrict its ability to pay dividends or
make other payments. If we or our consolidated subsidiaries are unable to
receive all of the revenues from our operations due to these contractual or
dividend arrangements, we may be unable to pay dividends on our common
stock.
We
may have difficulty establishing adequate management, governance, legal and
financial controls in the PRC.
The PRC
historically has been deficient in western style management, governance and
financial reporting concepts and practices, as well as in modern banking, and
other control systems. Our current management has little experience with
western style management, governance and financial reporting concepts and
practices, and we may have difficulty in hiring and retaining a sufficient
number of qualified employees to work in the PRC. As a result of these
factors, and especially given that we expect to be a publicly listed company in
U.S. and subject to regulation as such, we may experience difficulty in
establishing management, governance legal and financial controls, collecting
financial data and preparing financial statements, books of account and
corporate records and instituting business practices that meet western
standards. We may have difficulty establishing adequate management,
governance, legal and financial controls in the PRC. Therefore, we may, in
turn, experience difficulties in implementing and maintaining adequate internal
controls as required under Section 404 of the Sarbanes-Oxley Act of 2002 and
other applicable laws, rules and regulations. This may result in
significant deficiencies or material weaknesses in our internal controls which
could impact the reliability of our financial statements and prevent us from
complying with SEC rules and regulations and the requirements of the
Sarbanes-Oxley Act of 2002. Any such deficiencies, weaknesses or lack of
compliance could have a materially adverse effect on our business and the public
announcement of such deficiencies could adversely impact our stock
price.
It
may be difficult to protect and enforce our intellectual property rights under
PRC laws.
Intellectual
property rights in China are still developing, and there are uncertainties
involved in the protection and the enforcement of such rights. We will
need to pay special attention to protecting our intellectual property and trade
secrets. Failure to do so could lead to the loss of a competitive
advantage that could not be compensated by our damages award.
24
If
our land use rights are revoked, we would have no operational
capabilities.
Under
Chinese law land is owned by the state or rural collective economic
organizations. The state issues to tenants the rights to use property. Use
rights can be revoked and the tenants forced to vacate at any time when
redevelopment of the land is in the public interest. The public interest
rationale is interpreted quite broadly and the process of land appropriation may
be less than transparent. Through our operating subsidiary Bohai, we rely
on these land use rights as the cornerstone of our operations for both our
manufacturing facility and our corporate headquarters. The loss of such
rights would have a material adverse effect on our company as we would be
required to relocate our facilities and obtain new land use rights, and there is
a risk that we would not be able to accomplish such a relocation with reasonable
cost or at all.
In
addition, we currently do not maintain a land use right certificate for a piece
of land located in Xingfu Twelve Village of Zhifu District with the area of
11,222 square meters, on which we have built our corporate headquarters.
In the process of the planning of Yantai City, the usage of this land use right
has been changed from “industrial use” to “commercial use” and therefore, the
process for the land use right certificate on five relevant parcels of land
including the land occupied by Bohai is suspended until the completion of the
planning. We can not assure you that we will eventually obtain the land
use right certificate for this land with reasonable cost.
Risks
Related to Our Common Stock
Trading
in our common stock has been extremely limited, so investors may not be able to
sell as many of their shares as they want at prevailing prices.
Our
common stock is listed for quotation on OTCBB under the symbol “BOHP.OB”, but
trading in our common stock has been extremely limited. Trading in our
common stock may not fully commence for a variety of reasons, and even trading
does commence, it is expected to continue to be limited. If limited
trading in the common stock continues, it may be difficult for investors to sell
such shares in the public market at any given time at prevailing prices.
Also, the sale of a large block of common stock, should it occur, could depress
the market price of the common stock to a greater degree than a company that
typically has a higher volume of trading of its securities.
The
registration statement of which this prospectus forms a part may not remain
effective, which could impact the liquidity of our common stock.
Under the
terms of our January 2010 Registration Rights Agreement, we are obligated to
include the shares of common stock underlying the Notes and Warrants in an
effective registration statement, an the registration statement of which this
prospectus forms a part is intended to satisfy these obligations. From
time to time, it will be necessary for us to file post-effective amendments to
the registration statement when subsequent events so require. We intend to
use our best efforts to keep the registration statement current, but may not be
able to do so. If the registration statement is not current in the future,
investors’ ability to sell the shares of common stock underlying the Notes and
Warrants will be limited, which would have a material adverse effect on the
liquidity of our common stock.
There
is currently extremely limited trading in our common stock, and the limited
public trading market that may develop in the future may cause extreme
volatility in our stock price.
Although
our common stock is listed for quotation on the OTCBB, there has been extremely
limited trading in our stock. Even if a market for our common stock does
develop, there is a risk that a meaningful, consistent and liquid trading market
may not develop. Moreover, stocks with limited trading markets have
historically experienced extreme price and volume fluctuations and have
particularly affected the market prices of many smaller companies like us.
Our common stock is thus expected to be subject to significant volatility when
and if trading commences. Sales of substantial amounts of our common
stock, or the perception that such sales might occur, could adversely affect
prevailing market prices of our common stock.
25
An
active and visible trading market for our common stock may not
develop.
We cannot
predict whether an active market for our common stock will develop in the
future. In the absence of an active trading market:
|
|
·
|
Investors
may have difficulty buying and selling or obtaining market
quotations;
|
|
|
·
|
Market
visibility for our common stock may be limited;
and
|
|
|
·
|
A
lack of visibility for our common stock may have a depressive effect on
the market price for our common
stock.
|
The OTCBB
is an unorganized, inter-dealer, over-the-counter market that provides
significantly less liquidity than NASDAQ or the NYSE AMEX. The trading
price of the common stock is expected to be subject to significant fluctuations
in response to variations in quarterly operating results, changes in analysts’
earnings estimates, announcements of innovations by us or our competitors,
general conditions in the industry in which we operate and other factors.
These fluctuations, as well as general economic and market conditions, may have
a material or adverse effect on the market price of our common
stock.
The
market price for our stock may be volatile.
The
market price for our stock may be volatile and subject to wide fluctuations in
response to factors including the following:
|
|
·
|
actual
or anticipated fluctuations in our quarterly operating
results;
|
|
|
·
|
changes
in financial estimates by securities research
analysts;
|
|
|
·
|
conditions
in pharmaceutical markets;
|
|
|
·
|
changes
in the economic performance or market valuations of other pharmaceutical
companies;
|
|
|
·
|
announcements
by us or our competitors of new products, acquisitions, strategic
partnerships, joint ventures or capital
commitments;
|
|
|
·
|
addition
or departure of key personnel;
|
|
|
·
|
fluctuations
of exchange rates between RMB and the U.S.
dollar;
|
|
|
·
|
intellectual
property or other litigation; and
|
|
|
·
|
general
economic or political conditions in
China.
|
In
addition, the securities market has from time to time experienced significant
price and volume fluctuations that are not related to the operating performance
of particular companies. These market fluctuations may also materially and
adversely affect the market price of our stock.
26
The
accounting treatment for our convertible securities is complex and subject to
judgments concerning the valuation of embedded derivative rights within the
applicable securities. Fluctuations in the valuation of these rights could
cause us to take charges to our earnings and make our financial results
unpredictable.
Our Notes
and Warrants issued in January 2010 contain, or may be
deemed to contain from time to time, embedded
derivative rights in accordance with GAAP. These derivative rights, or
similar rights in convertible securities we may issue in the future, need to be,
or may need to be, separately valued as of the end of each accounting period in
accordance with GAAP. Changes in the valuations of these rights, the
valuation methodology or the assumptions on which the valuations are based could
cause us to take charges to our earnings, which would adversely impact our
results of operations. Moreover, the methodologies, assumptions and
related interpretations of accounting or regulatory authorities associated with
these embedded derivatives are complex and in some cases uncertain, which could
cause our accounting for these derivatives, and as a result, our financial
results, to fluctuate. There is a risk that questions could arise from
investors or regulatory authorities concerning the appropriate accounting
treatment of these instruments, which could require us to restate previous
financial statements, which in turn could adversely impact our results of
operations, our reputation and our public stock price.
Shares
eligible for future sale may adversely affect the market price of our common
stock, as the future sale of a substantial amount of outstanding stock in the
public marketplace could reduce the price of our common stock.
Pursuant
to the terms of the Registration Rights Agreement and Securities Purchase
Agreement, we agreed to file the registration statement of which this prospectus
forms a part with the SEC to register the common stock underlying the Notes,
Warrants and Placement Agent Warrants for public resale. All of such
shares may be freely sold and transferred following conversion or exercise of
the Notes and Warrants if such registration statement remains effective.
Additionally, concurrently with the closing of the Private Placement, we engaged
in a Share Exchange, and following the Share Exchange, the former shareholders
of Chance High (other than Glory Period) may be eligible to sell all or some of
their shares of common stock by means of ordinary brokerage transactions in the
open market pursuant to such registration statement or SEC Rule 144, subject to
certain limitations. In general, pursuant to Rule 144, a stockholder (or
stockholders whose shares are aggregated) who has satisfied an one-year holding
period may, under certain circumstances, sell within any three-month period a
number of securities which does not exceed the greater of 1% of the then
outstanding shares of common stock or the average weekly trading volume of the
class during the four calendar weeks prior to such sale. As of the closing
of the Share Exchange, and not accounting for the private placement, 1% of our
issued and outstanding shares of common stock equals approximately162,500
shares. Rule 144 also permits, under certain circumstances, the sale of
securities, without any limitations, by a non-affiliate that has satisfied a
one-year holding period. Any substantial sale of common stock pursuant to
any resale prospectus or Rule 144 may have an adverse effect on the market price
of our common stock by creating an excessive supply.
Our
controlling stockholder may exercise significant influence over us.
Our
controlling stockholder, Glory Period Limited, owns approximately 55% of our
outstanding common stock as of the closing of the Share Exchange. Tan is
the sole shareholder of Glory Period and Qu is the sole director of Glory
Period. Either Tan or Qu has a controlling influence in determining the
outcome of any corporate transaction or other matters submitted to our
stockholders for approval, including mergers, consolidations and the sale of all
or substantially all of our assets, election of directors, and other significant
corporate actions. Tan and Qu may also have the power to prevent or cause
a change in control. In addition, without the consent of Tan and Qu, we
could be prevented from entering into transactions that could be beneficial to
us. As Qu serves as our principal executive officer and Tan has provided
consulting services to Bohai in the past, the interests of Tan and Qu may differ
from the interests of our other stockholders, which could create conflicts of
interest and the potential for approval of actions which may not be in the best
interests of all of our stockholders.
27
Compliance
with complex and changing regulation of corporate governance and public
disclosure, and our management’s inexperience with such regulations, will result
in additional expenses and creates a risk of non-compliance.
Changing
laws, regulations and standards relating to corporate governance and public
legal and accounting disclosure, including the Sarbanes-Oxley Act of 2002 and
related SEC regulations, have created uncertainty for public companies and
significantly increased the costs and risks associated with accessing the public
markets and public reporting. Our management team will need to invest
significant management time and financial resources to comply with both existing
and evolving standards for public companies, which will lead to increased
general and administrative expenses and a diversion of management time and
attention from revenue generating activities to compliance activities. In
addition, our management located in the PRC has little experience with
compliance with U.S. laws (including securities laws). This inexperience
may cause us to fall out of compliance with applicable regulatory requirements,
which could lead to restatements of our financial statements, breaches of
covenants in our investor agreements, regulatory enforcement actions against us
and a negative impact on our stock price and our business
generally.
The
challenges we have with properly complying with applicable disclosure and
accounting regulations were evidenced when we were required to restate our
financial statements for the period ended March 31, 2010 to account for the
embedded derivative liabilities associated with Notes and Warrants. There
is a risk that we will face similar challenges in the future.
If
we fail to maintain an effective system of internal controls, we may not be able
to accurately report our financial results or prevent fraud.
We are
subject to reporting obligations under the U.S. securities laws. The SEC,
as required by Section 404 of the Sarbanes-Oxley Act of 2002, adopted rules
requiring every public company to include a management report on such company’s
internal controls over financial reporting in its annual report, which contains
management’s assessment of the effectiveness of our internal controls over
financial reporting. In addition, we may be required to have an
independent registered public accounting firm attest to and report on
management’s assessment of the effectiveness of our internal controls over
financial reporting. Our management may conclude that our internal
controls over our financial reporting are not effective. Moreover, even if
our management concludes that our internal controls over financial reporting are
effective, our independent registered public accounting firm may still decline
to attest to our management’s assessment or may issue a report that is qualified
if it is not satisfied with our controls or the level at which our controls are
documented, designed, operated or reviewed, or if it interprets the relevant
requirements differently from us. Our reporting obligations as a public
company will place a significant strain on our management, operational and
financial resources and systems for the foreseeable future. Effective
internal controls, particularly those related to revenue recognition, are
necessary for us to produce reliable financial reports and are important to
prevent fraud. As a result, our failure to achieve and maintain effective
internal controls over financial reporting could result in the loss of investor
confidence in the reliability of our financial statements, which in turn could
harm our business and negatively impact the trading price of our stock.
Furthermore, we anticipate that we will incur considerable costs and use
significant management time and other resources in an effort to comply with
Section 404 and other requirements of the Sarbanes-Oxley Act.
28
Our
common stock may be considered a “penny stock,” and thereby be subject to
additional sale and trading regulations that may make it more difficult to
sell.
Our
common stock, which is currently and will be quoted for trading on OTCBB, may be
considered to be a “penny stock” if it does not qualify for one of the
exemptions from the definition of “penny stock” under Section 3a51-1 of the
Exchange Act, as amended. Our common stock may be a “penny stock” if it
meets one or more of the following conditions: (i) the stock trades at a price
less than $5 per share; (ii) it is NOT traded on a “recognized” national
exchange; (iii) it is not quoted on the Nasdaq Capital Market, or even if so,
has a price less than $5 per share; or (iv) is issued by a company that has been
in business less than three years with net tangible assets less than $5
million. The principal result or effect of being designated a “penny
stock” is that securities broker-dealers participating in sales of our common
stock will be subject to the “penny stock” regulations set forth in Rules 15-2
through 15g-9 promulgated under the Exchange Act. For example, Rule 15g-2
requires broker-dealers dealing in penny stocks to provide potential investors
with a document disclosing the risks of penny stocks and to obtain a manually
signed and dated written receipt of the document at least two business days
before effecting any transaction in a penny stock for the investor’s
account. Moreover, Rule 15g-9 requires broker-dealers in penny stocks to
approve the account of any investor for transactions in such stocks before
selling any penny stock to that investor. This procedure requires the
broker-dealer to: (i) obtain from the investor information concerning his or her
financial situation, investment experience and investment objectives; (ii)
reasonably determine, based on that information, that transactions in penny
stocks are suitable for the investor and that the investor has sufficient
knowledge and experience as to be reasonably capable of evaluating the risks of
penny stock transactions; (iii) provide the investor with a written statement
setting forth the basis on which the broker-dealer made the determination in
(ii) above; and (iv) receive a signed and dated copy of such statement from the
investor, confirming that it accurately reflects the investor’s financial
situation, investment experience and investment objectives. Compliance
with these requirements may make it more difficult and time consuming for
holders of our common stock to resell their shares to third parties or to
otherwise dispose of them in the market or otherwise.
We
do not foresee paying cash dividends in the foreseeable future and, as a result,
our investors’ sole source of gain, if any, will depend on capital appreciation,
if any.
We do not
plan to declare or pay any cash dividends on our shares of common stock in the
foreseeable future and currently intend to retain any future earnings for
funding growth. As a result, investors should not rely on an investment in our
securities if they require the investment to produce dividend income. Capital
appreciation, if any, of our shares may be investors’ sole source of gain for
the foreseeable future. Moreover, investors may not be able to resell their
common stock at or above the price they paid for them.
29
CAUTIONARY
NOTE REGARDING FORWARD-LOOKING STATEMENTS
Some of
the statements contained in this prospectus that are not historical facts are
“forward-looking statements” which can be identified by the use of terminology
such as “estimates,” “projects,” “plans,” “believes,” “expects,” “anticipates,”
“intends,” or the negative or other variations, or by discussions of strategy
that involve risks and uncertainties. We urge you to be cautious of the
forward-looking statements, that such statements, which are contained in this
Registration Statement, reflect our current beliefs with respect to future
events and involve known and unknown risks, uncertainties and other factors
affecting our operations, market growth, services and products. No
assurances can be given regarding the achievement of future results, as actual
results may differ materially as a result of the risks we face, and actual
events may differ from the assumptions underlying the statements that have been
made regarding anticipated events. Factors that may cause actual results,
our performance or achievements, or industry results, to differ materially from
those contemplated by such forward-looking statements include without
limitation:
|
|
·
|
our
financial position, business strategy and other plans and objectives for
future operations;
|
|
|
·
|
the
ability of our management team to execute its plans to meet its
goals;
|
|
|
·
|
our
ability to attract and retain qualified management
personnel;
|
|
|
·
|
our
growth strategies;
|
|
|
·
|
anticipated
trends in our business;
|
|
|
·
|
our
ability to consummate or integrate
acquisitions;
|
|
|
·
|
our
liquidity and ability to finance our operations and acquisition and
development activities;
|
|
|
·
|
the
timing, cost and procedure for proposed
acquisitions;
|
|
|
·
|
the
impact of government regulation in China and
elsewhere;
|
|
|
·
|
estimates
regarding future net revenues or
profits;
|
|
|
·
|
planned
capital expenditures (including the amount and nature
thereof);
|
|
|
·
|
estimates,
plans and projections relating to acquired properties or
businesses;
|
|
|
·
|
the
possibility that our acquisitions may involve unexpected
costs;
|
|
|
·
|
the
impact of competition;
|
|
|
·
|
general
economic conditions, whether internationally, nationally or in the
regional and local market areas in which we are doing business, that may
be less favorable than
expected;
|
30
|
|
·
|
other
economic, competitive, governmental, legislative, regulatory, geopolitical
and technological factors that may negatively impact our businesses,
operations and pricing; and
|
|
|
·
|
our
implementation of required financial, accounting and disclosure controls
and procedures and related corporate governance
policies.
|
All
written and oral forward-looking statements made in connection with this
prospectus that are attributable to us or persons acting on our behalf are
expressly qualified in their entirety by these cautionary statements.
Given the uncertainties that surround such statements, you are cautioned not to
place undue reliance on such forward-looking statements.
USE
OF PROCEEDS
This
prospectus relates to shares of our common stock that may be offered and sold
from time to time by the selling stockholders named herein. There will be
no proceeds to us from the sale of shares of common stock in this
offering.
We will
not receive any proceeds from the sale of shares by the selling stockholders
other than the exercise price of any Warrants and Placement Agent Warrants that
are exercised by the applicable selling stockholders, the proceeds of which we
expect to use for our general working capital. If all of these Warrants
and Placement Agent Warrants are exercised for cash, then we will receive gross
proceeds of $15,840,000. No assurances can be given, however, that all or
any portion of such warrants will ever be exercised.
DETERMINATION
OF OFFERING PRICE
The
selling stockholders may sell these shares in the over-the-counter market or
otherwise, at market prices prevailing at the time of sale, at prices related to
the prevailing market price, or at negotiated prices. We will not receive
any proceeds from the sale of shares by the selling stockholders but we may
receive proceeds upon the exercise, if any, of the Warrants and Placement Agent
Warrants.
31
MANAGEMENT’S
DISCUSSION AND ANALYSIS OF FINANCIAL CONDITIONS
AND
RESULTS OF OPERATIONS
The
following discussion and analysis of financial condition and results of
operations relates to the operations and financial condition reported in the
unaudited condensed consolidated financial statements of the Company for the
three months ended March 31, 2010 and 2009, and for the fiscal years ended June
30, 2009 and 2008, and should be read in conjunction with the related audited
and unaudited financial statements and related notes included in this
prospectus. Those statements in the following discussion that are not
historical in nature should be considered to be forward looking statements that
are inherently uncertain. Actual results and the timing of the events may differ
materially from those contained in these forward looking statements due to a
number of factors, including those discussed in the “Cautionary Note on Forward
Looking Statements” set forth elsewhere in this prospectus.
Overview
We were
incorporated under the laws of the State of Nevada on January 9, 2008. Since
January 5, 2010, our business consists of the production, manufacturing and
distribution of herbal pharmaceuticals in the PRC which are based on traditional
Chinese medicine. We are based in the city of Yantai, Shandong Province,
China.
Our
medicines are intended to address rheumatoid arthritis, viral infections,
gynecological diseases, cardio vascular issues and respiratory diseases. We have
obtained Drug Approval Numbers in China for 29 varieties of traditional Chinese
herbal medicines in 2004 and we currently produce 10 varieties of approved
traditional Chinese herbal medicines in seven delivery systems: tablets,
granules, capsules, syrup, concentrated powder, tincture and medicinal wine. Of
these 10 products, 5 are prescription drugs and 5 are over-the-counter products.
Prior to
January 5, 2010, we were a public “shell” company operating under the name “Link
Resources, Inc.” On January 5, 2010, we consummated a share exchange transaction
(the “Share Exchange”) pursuant to which we acquired Chance High, the parent
company of Yantai Bohai Pharmaceuticals Group Co. Ltd., our principal operating
subsidiary, which is a Chinese variable interest entity that we (through a
Chinese wholly-owned foreign enterprise subsidiary) control through certain
contractual arrangements.
Use
of Estimates
In
preparing financial statements in conformity with accounting principles
generally accepted in the United States of America, management makes estimates
and assumptions that affect the reported amounts of assets liabilities and
disclosures of contingent assets and liabilities at the dates of the financial
statements, as well as the reported amounts of revenues and expenses during the
reporting periods. These accounts and estimates include, but are not limited to,
the valuation of accounts receivable, inventories, deferred income taxes,
derivative liabilities, and the estimation of useful lives of property, plant
and equipment. Actual results could differ from those estimates.
Accounts
Receivable
Accounts
receivable consists of amounts due from customers. We extend unsecured credit to
our customers in the ordinary course of business but mitigate the associated
risks by performing credit checks and actively pursuing past due accounts. An
allowance for doubtful accounts is established and determined based on
management’s assessment of known requirements, aging of receivables, payment
history, the customer’s current credit worthiness and the economic
environment.
32
Revenue
recognition
Revenue
represents the invoiced value of goods sold recognized upon the delivery of
goods to customers. Revenue is recognized when all of the following criteria are
met:
|
|
·
|
Persuasive
evidence of an arrangement exists;
|
|
|
·
|
Delivery
has occurred or services have been rendered;
|
|
|
·
|
The
seller’s price to the buyer is fixed or determinable; and
|
|
|
·
|
Collectability
is reasonably assured. Payments have been established.
|
Inventories
Inventories
are valued at the lower of cost or market with cost is determined on the
weighted average method. Finished goods inventories consist of raw materials,
direct labor and overhead associated with the manufacturing process. In
assessing the ultimate realization of inventories, management makes judgments as
to future demand requirements compared to current or committed inventory levels.
Our reserve requirements generally increase or decrease due to management’s
projected demand requirements, market conditions and product life cycle changes.
As of March 31, 2010 and June 30, 2009, we did not make any allowance for
slow-moving or defective inventories.
Property,
plant and equipment
Property,
plant and equipment, other than construction in progress, are stated at cost,
less depreciation and amortization and accumulated impairment loss. Cost
represents the purchase price of the asset and other costs incurred to bring the
asset into its existing use. Maintenance, repairs and betterments, including
replacement of minor items, are charged to expense; major additions to physical
properties are capitalized.
Depreciation
of property, plant and equipment is calculated to written off the cost, less
their estimated residual value, if any, using the straight-line method over
their estimated useful lives. The principal annual rates are as
follows:
|
Leas
ehold land and buildings
|
30
to 40 years
|
|
Motor
vehicles
|
10
years
|
|
Plant
and machinery
|
10
years
|
|
Office
equipment
|
5
years
|
Upon sale
or disposition, the applicable amounts of asset cost and accumulated
depreciation are removed from the accounts and the net amount less proceeds from
disposal is charged or credited to income.
Construction
in progress primarily represents costs incurred to construct the Group’s
corporate campus and machinery under construction. The corporate campus
was completed and at began production in 2009. Assets under construction
are not depreciated until the construction is completed and the assets are ready
for their intended use.
33
Income
Taxes
Income
taxes are accounted for under the asset and liability method. Deferred tax
assets and liabilities are recognized for the future tax consequences
attributable to differences between the financial statement carrying amounts of
existing assets and liabilities and their respective tax bases and operating
loss and tax credit carry-forwards. Deferred tax assets are reduced by a
valuation allowance to the extent management concludes it is more likely than
not that the assets will not be realized. Deferred tax assets and liabilities
are measured using enacted tax rates expected to apply to taxable income in the
years in which those temporary differences are expected to be recovered or
settled. The effect on deferred tax assets and liabilities of a change in tax
rates is recognized in the statements of income and comprehensive income in the
periods that includes the enactment date.
Operating
Results
Three
Months Ended March 31, 2010 Compared to Three Months Ended March 31,
2009
|
Three Months Ended
March 31,
|
||||||||
|
2010
|
2009
|
|||||||
|
(unaudited)
RESTATED
|
(unaudited)
|
|||||||
|
Sales
|
$ | 15,323,878 | $ | 12,476,400 | ||||
|
Less:
Sales Tax
|
(231,870 | ) | (200,622 | ) | ||||
|
Net
sales
|
15,092,008 | 12,275,778 | ||||||
|
Cost
of sales
|
(2,609,515 | ) | (2,019,864 | ) | ||||
|
Gross
profit
|
12,482,493 | 10,255,914 | ||||||
|
Selling,
general and administrative expenses
|
(9,202,873 | ) | (7,670,130 | ) | ||||
|
Interest
expenses
|
(381,700 | ) | (52,921 | ) | ||||
|
Operating
income
|
2,897,920 | 2,532,863 | ||||||
|
Other
income
|
- | 158 | ||||||
|
Finance
and non-operating incomes (expenses)
|
829,773 | (4,618 | ) | |||||
|
Income
before taxes
|
3,727,693 | 2,528,403 | ||||||
|
Income
taxes
|
(585,135 | ) | (423,569 | ) | ||||
|
Net
income
|
$ | 3,142,558 | $ | 2,104,834 | ||||
Sales
Net sales
for the three months ended March 31, 2010 increased by approximately $2,816,230,
or 22.94%, to $15,092,008 as compared to $12,275,778 for the three months ended
March 31, 2009. This increase was primarily due to the significant
increase of our revenue on all the products as a result of the marketing
strategy we implemented during the three months ended March 31, 2010.
Cost
of Sales
Our cost
of sales for the three months ended March 31, 2010 was $ 2,609,515 as compared
to $2,019,864 for the three months ended March 31, 2009, representing an
increase of 29.19%. The increase was mainly attributable to the
increase in cost of raw material by $265,960 as a result of the increase of
sales.
Gross
Profit
We achieved
gross profit of $12,482,493 for the three months ended March 31, 2010, compared
to $10,255,914 for the same quarter of the previous year, representing a 21.71 %
quarter to quarter increase. Our overall gross profit margin as a
percentage of revenue are 82.71% and 83.30% for the three months ended March 31,
2010 and 2009, respectively.
34
Selling,
General and Administrative Expenses
Our
operating expenses, consisting of selling, general and administrative expenses,
increased by approximately $1,532,743, to $9,202,873, for the three months ended
March 31, 2010, from $7,670,130 for the same quarter of the previous year.
This increase was mainly attributable to an increase of advertising expense and
sales conferences this quarter compared to the same quarter last year. We
increased product promotion activities through various media, especially through
television advertising in different provinces within the PRC during the quarter
compared to the same quarter last year.
Interest
Expense
Interest
expense was $381,700 for the three months ended March 31, 2010, compared to
$52,921 for the three months ended March 31, 2009, an increase of $328,779.
The increase was principally due to effective interest charge of $347,793 on
convertible notes issued in connection with a private placement on January 5,
2010.
Finance
and Non-Operating Income (Expenses)
Finance and
non-operating income was $829,773 for the period ended March 31, 2010
compared to non-operating expenses of $4,618 from the same quarter last year, an
increase of income of $834,391. The increase was principally due to
non-cash income of $1,083,350 associated with a change in fair
value of warrants, and amortization of deferred fees of $253,577 in connection
with our private placement on January 5, 2010.
Income
Tax
Our
provisions for income taxes for the three months ended March 31, 2010 and 2009
were $585,135 and $423,569, an increase of $161,566 or 38.14% from period to
period. Certain corporate expenses such as amortization of financing cost,
changes in derivative liabilities, and effective interest charges on convertible
note were permanently excluded from Chinese income tax calculations. If
such non-taxable expenses were excluded, the ratios between provisions for
income taxes and income before tax solely generated from Bohai, our operating
company in China, would be 18.03% and 16.75%, respectively for the three months
ended March 31, 2010 and 2009.
Net
Income
We had a
net income of $3,142,558 for the three months ended March 31, 2010, as compared
to net income of $2,104,834 for the three months ended March 31, 2009,
an increase in net income of $1,037,724. The increase in net income
was primarily attributable to incomes from non-operating incomes related to
non-cash activities on fair value measurement of $1,083,350 for our warrants
offset by effective interest charge of $347,793 and amortization of
deferred fees of $253,577 in connection with our private placement and
share exchange transactions that occurred in the quarter ended March 31,
2010. The increase was also attributed by increase in total gross
profit and offset by increase in advertising expenses this quarter compared
to the same quarter last year.
Nine
Months Ended March 31, 2010 Compared to the Nine Months Ended March 31,
2009
The
following table sets forth our statement of operations for the period
indicated:
35
|
Nine Months Ended
March 31,
|
||||||||
|
2010
|
2009
|
|||||||
|
(unaudited)
RESTATED
|
(unaudited)
|
|||||||
|
Sales
|
$ | 46,072,455 | $ | 35,833,215 | ||||
|
Less:
Sales Tax
|
(729,975 | ) | (580,207 | ) | ||||
|
Net
sales
|
45,342,480 | 35,253,008 | ||||||
|
Cost
of sales
|
(7,475,740 | ) | (5,966,876 | ) | ||||
|
Gross
profit
|
37,866,740 | 29,286,132 | ||||||
|
Selling,
general and administrative expenses
|
(28,208,753 | ) | (21,675,279 | ) | ||||
|
Interest
expenses
|
(538,008 | ) | (168,422 | ) | ||||
|
Operating
income
|
9,119,979 | 7,442,431 | ||||||
|
Non-operating
income
|
18,864 | 158 | ||||||
|
Finance
and non-operating incomes (expenses)
|
807,681 | (4,682 | ) | |||||
|
Income
before taxes
|
9,946,524 | 7,437,907 | ||||||
|
Income
taxes
|
(2,193,931 | ) | (1,224,833 | ) | ||||
|
Net
income
|
$ | 7,752,593 | $ | 6,213,074 | ||||
Sales
Net sales
for the nine months ended March 31, 2010 was $45,342,480, an increase of
approximately $10,089,472, or 28.62%, from $35,253,008 in the nine months ended
March 31, 2009. This increase was primarily due to the significant increase of
our revenue on all the products as a result of the marketing strategy we have
implemented since the calendar year ended December 31, 2009.
Cost
of Sales
Our cost
of sales for the nine months ended March 31, 2010 was $7,475,740 as compared to
$5,966,876 for the nine months ended March 31, 2009, representing an increase of
$1,508,864, or 25.28%. The increase was mainly attributable to the increase of
raw materials cost by $1,033,246 as a result of the increase of
sales.
Gross
Profit
For the
nine months ended March 31, 2010 as compared to the nine months ended March 31,
2009, we generated gross profit of $37,866,740 and $29,286,132, respectively,
reflecting an increase of approximately $8,580,608, or 29.30%. The increase in
our gross profit was mainly due to significantly increase of sales
income.
Selling,
General and Administrative Expenses
We incurred
general and administrative expenses of $28,208,753 for the nine months ended
March 31, 2010, representing an increase of $6,533,474, or 30.14%, compared to
$21,675,279 for the nine months ended March 31, 2009. This increase was mainly
due to the increase of costs of advertising and sales conferences throughout
China.
36
Interest
Expense
Interest
expense was $538,008 for the nine months ended March 31, 2010, compared to
$168,422 for the nine months ended March 31, 2009, an increase of
$369,586. The increase was principally due to effective interest charge of
$347,793 on convertible notes issued in connection with a private placement on
January 5, 2010.
Finance
and Non-Operating Incomes (expenses)
Finance
and non-operating income was $807,681 for the nine months ended March 31,
2010 compared to non-operating expenses of $4,682 for the nine months ended
March 31, 2009, an increase of non-operating incomes of $812,363. The
increase was principally due to non-cash income of $1,083,350 associated
with a change in fair value of warrants and amortization of deferred fees
of $253,577 in connection with our private placement on January 5, 2010.
Income
Tax
For the
nine months ended March 31, 2010 and 2009, income tax expenses were $2,193,931
and $1,224,833 respectively, representing an increase of $969,098, or 79.12%.
Certain corporate expenses such as amortization of financing cost, changes in
fair value of warrants, and effective interest charges on convertible note were
permanently excluded from Chinese income tax calculations. If such non-taxable
expenses were excluded, the income tax expenses would represent 23.04% and
16.46% of the operating income before taxes generated from our major operating
subsidiary, Bohai, for the nine months ended March 31, 2010 and 2009,
respectively. The increase was mainly due to an increase of income before tax
generated from our Bohai subsidiary for the period ended March 31, 2010 compared
to the period ended March, 31, 2009, as well as an under estimate of tax
expenses for the nine months ended on March 31, 2009 compared to the same
periods in 2010.
Net
Income
We had
net income of $7,752,593, or 17.10% of net revenue, for the nine months ended
March 31, 2010, as compared to net income $6,213,074, or 17.62% of net revenue,
for the nine months ended March 31, 2009, representing an increase of
$1,539,519. The increase was attributed by increase in total gross profit offset
by increase in advertising expenses, certain non-cash charges related to
effective interest of $347,793, non-cash incomes of changes in fair value
of warrants of $1,083,350, and amortization of deferred fees of $253,577 in
connection with our private placement and share exchange transactions that
occurred in the quarter ended March 31, 2010.
Liquidity
and Capital Resources
We have
historically funded our operation primarily through paid-in capital, sales of
goods and short term loans from financial institutions in China. Net increase in
cash for the three months ended March 31, 2010 was $7,883,867 compared to net
increase in cash of $1,729,968 for the three months ended March 31, 2009. During
the three months ended March 31, 2010, net cash used from operating activities
was $2,449,103. Cash payments to placement agent and other financing costs were
$1,570,000. Cash borrowed from bank and investors amounted to $4,381,153 and
$12,000,000 respectively and repayment of borrowing from previous period
amounted to $4,393,665, which resulted in a net cash inflow from financing
activities of $10,417,488 and a total net increase in cash of $7,883,867. We may
seek to raise additional capital through sales of common stock, as well as
seeking financing from third parties.
37
Operating
activities
For the
three months ended March 31, 2010, cash used in operating activities totaled
$2,449,103 compared to cash provided by operating activities of $2,503,932 for
the three months ended March 31, 2009. The decrease on net cash provided in
operating activities was primarily attributable to cash used in prepayment for
other payable and accrued liabilities during the three month ended March 31,
2010.
Investing
activities
For the
three months ended March 31, 2010, net cash outflow for investing activities was
approximately $84,518 compared to net cash used by investing activities of
$191,977 in the three months ended March 31, 2009. This decrease on net cash
used by investing activities was primarily attributable to decrease on cash used
for purchase of property plant and equipment during the period.
Financing
activities
Financing
activities provided net cash inflow of $10,417,488 during the three months ended
March 31, 2010. The net cash inflow was primarily as a result of issue of
convertible promissory notes during the period. As of March 31, 2010, cash
payments to placement agent and other financing costs were $1,570,000. The
proceeds from short-term borrowings and convertible promissory notes amounted to
$4,381,153 and $12,000,000 respectively and the repayment of borrowings amounted
to $4,393,665 resulted in a net cash inflow by financing activities of
$10,417,488.
Off-Balance
Sheet Arrangements
We did
not engage in any “off-balance sheet arrangements” (as that term is defined in
Item 303(a)(4)(ii) of Regulation S-K) during the three months ended March 31,
2010. We have not entered into any financial guarantees or other commitments to
guarantee the payment obligations of any third parties. We have not entered into
any derivative contracts that are indexed to our shares and classified as
stockholder’s equity or that are not reflected in our condensed consolidated
financial statements. Furthermore, we do not have any retained or contingent
interest in assets transferred to an unconsolidated entity that serves as
credit, liquidity or market risk support to such entity. We do not have any
variable interest in any unconsolidated entity that provides financing,
liquidity, market risk, or credit support to us or engages in leasing, hedging,
or research and development services with us.
Inflation
In recent
years, the Chinese economy has experienced periods of rapid expansion and high
rates of inflation. During the past ten years, the rate of inflation in China
has been as high as 20.7% and as low as -2.2%. These factors have led to the
adoption by Chinese government, from time to time, of various corrective
measures designed to restrict the availability of credit or regulate growth
and contain inflation. While inflation has been more moderate since 1995, high
inflation may in the future cause Chinese government to impose controls on
credit and/or prices, or to take other action, which could inhibit economic
activity in China, and thereby harm the market for our
products.
38
Fiscal
Year Ended June 30, 2009 Compared to the Fiscal Year Ended June 30,
2008
The
following table sets forth our statement of operations for the period
indicated:
|
Year Ended
June 30, 2009
|
Year Ended
June 30, 2008
|
|||||||
|
Sales
|
$ | 50,170,014 | $ | 38,172,513 | ||||
|
Less:
Sales Tax
|
(821,400 | ) | (629,489 | ) | ||||
|
Net
sales
|
49,348,614 | 37,543,024 | ||||||
|
Cost
of sales
|
(7,975,267 | ) | (5,950,680 | ) | ||||
|
Gross
profit
|
41,373,347 | 31,592,344 | ||||||
|
Selling,
general and administrative expenses
|
(31,347,139 | ) | (22,608,164 | ) | ||||
|
Interest
expenses
|
(184,404 | ) | (234,101 | ) | ||||
|
Operating
income
|
9,841,804 | 8,750,079 | ||||||
|
Non-operating
income
|
49,447 | 8 | ||||||
|
Non-operating
costs
|
(36,366 | ) | (835 | ) | ||||
|
Income
before taxes
|
9,854,885 | 8,749,252 | ||||||
|
Income
taxes
|
(1,906,985 | ) | (2,303,712 | ) | ||||
|
Net
income
|
$ | 7,947,900 | $ | 6,445,540 | ||||
Gross
Profit
For the
fiscal year ended June 30, 2009 as compared to the fiscal year ended June 30,
2008, we generated gross profit of $41,373,347 and $31,592,344 respectively,
reflecting an increase of approximately $9,781,003, or 30.96%. The increase in
our gross profit was mainly due to significantly increase of sales
income.
Selling,
General and Administrative Expenses
We
incurred general and administrative expenses of $31,347,139 for the fiscal year
ended June 30, 2009, representing an increase of $8,738,957, or 38.65%, compared
to 22,608,164 for the fiscal year ended June 30, 2008. This increase was mainly
due to the increase of cost of advertising and percentages of commissions to
sales representative allocated in branch office through out China.
Interest
Expense
Interest
expense was $184,404 for the fiscal year ended June 30, 2009, compared to
$234,101 for the fiscal year ended June 30, 2008. The decrease was largely
due to repayment of bank loans utilized in the year 2007 during the period.
The loans were used for working capital and capital expenditures for the
expansion of production through acquisition of production plants and equipment
to improve the efficiency of production.
39
Income
tax
For the
fiscal year ended June 30, 2009, income tax expense was $1,906,985, a decrease
of $396,727, or 17.22%, from $2,303,712 for the fiscal year ended June 30, 2008.
The decrease was mainly due to the decrease of enterprise income tax in PRC from
33% to 25% since January 1, 2008.
Net
Income
We had
net income of $7,947,900, or 16.11% of net revenue for the fiscal year ended
June 30, 2009 as compared to net income of $6,445,540 or 17.17% of net revenue
for the fiscal year ended June 30, 2008, representing an increase of $1,502,360,
or 23.31%. The increase in our net income was the result of the significant
increase of our revenue generated in the fiscal year of 2009
Liquidity
and Capital Resources
We have
historically funded our operation primarily through paid-in capital, sales of
goods and short term loans from financial institutions. Net cash used in
operating activities for the year ended June 30, 2009 was $1,513,062 compared to
net cash provided in operating activities of $2,359,072 for the year ended June
30, 2008. During the year ended June 30, 2009, cash borrowed from banks amounted
to $5,860,000 and repayment of borrowing from previous years amounted to
$1,896,700, which resulted in a net cash inflow in financing activities of
$3,963,300. Management is planning to raise additional capital through
sales of common stock, as well as seeking financing from third
parties.
Operating
activities
For the
fiscal year ended June 30, 2009, net cash used in operating activities totaled
$1,513,062 compared to net cash provided by operating activities of $2,359,072
for the fiscal year ended June 30, 2008. This decrease was primarily
attributable to a significant increase of cash used in prepayment for
advertising expense and repayment of accounts payable.
Investing
activities
For the
fiscal year ended June 30, 2009, net cash outflow for investing activities was
approximately $788,993 compared to net cash outflow for investing activities of
$80,647 in the fiscal year ended June 30, 2008. This increase was primarily
attributable to significant increase of capital investment in acquisition of
property, plant and equipment during the year. During the fiscal year ended June
30, 2009, we spent $227,690, $59,459, $295,610 and $220,884 on acquisitions of
plant and equipment, purchase of office equipment, purchase of motor
vehicle and construction in progress, respectively.
Financing
activities
Financing
activities provided net cash inflow of $3,963,300 during the fiscal year ended
June 30, 2009. The net cash inflow was primarily as a result of proceeds
of significant borrowing during the year. As of June 30, 2009, the proceeds from
short-term borrowings amounted to $5,860,000 and the repayment of borrowings
amounted to $1,896,700, which resulted in a net cash inflow by financing
activities of $3,963,300.
40
Off-Balance
Sheet Arrangements
We did
not engage in any “off-balance sheet arrangements” (as that term is defined in
Item 303(a)(4)(ii) of Regulation S-K) during the fiscal year ended June 30,
2010. We have not entered into any financial guarantees or other
commitments to guarantee the payment obligations of any third parties. We have
not entered into any derivative contracts that are indexed to our shares and
classified as shareholder’s equity or that are not reflected in our
condensed consolidated financial statements. Furthermore, we do not have any
retained or contingent interest in assets transferred to an unconsolidated
entity that serves as credit, liquidity or market risk support to such entity.
We do not have any variable interest in any unconsolidated entity that provides
financing, liquidity, market risk, or credit support to us or engages in
leasing, hedging, or research and development services with us.
Inflation
In recent
years, the Chinese economy has experienced periods of rapid expansion and high
rates of inflation. During the past ten years, the rate of inflation in
China has been as high as 20.7% and as low as -2.2%. These factors have
led to the adoption by Chinese government, from time to time, of various
corrective measures designed to restrict the availability of credit or regulate
growth and contain inflation. While inflation has been more moderate since
1995, high inflation may in the future cause Chinese government to impose
controls on credit and/or prices, or to take other action, which could inhibit
economic activity in China, and thereby harm the market for our
products.
41
BUSINESS
Our
Business
We are
engaged in the production, manufacturing and distribution of herbal
pharmaceuticals based on traditional Chinese medicine, or TCM, in the PRC.
We are based in the city of Yantai, Shandong Province, China, and our operations
are exclusively in China.
Our
medicines address rheumatoid arthritis, viral infections, gynecological
diseases, cardio vascular issues and respiratory diseases. We obtained
Drug Approval Numbers for 29 varieties of traditional Chinese herbal medicines
in 2004 and it currently produces 10 varieties of approved traditional Chinese
herbal medicines in seven delivery systems: tablets, granules, capsules, syrup,
concentrated powder, tincture and medicinal wine. Of these 10 products, 5
are prescription drugs and 5 are OTC products.
We
believe our rapid growth in recent years has been supported by the continuing
expansion of the market for TCM in the PRC. This market is forecasted to
reach $24 billion by 2010 according to the Information Office of the State
Council of the People’s Republic of China, “Status Quo of Drug
Supervision.”
In a
significant development, on December 1, 2009, two of our lead products, Tongbi
Capsules and Tablets and Lung Nourishing Cream, became eligible for
reimbursement under China’s National Medical Insurance Program. In fiscal
year ended June 30, 2009, these two products accounted for more than 50% of our
revenues.
The
Chinese government has previously awarded us exclusive rights to manufacture
Tongbi Capsules and we share manufacturing rights with one or more manufacturers
for Shangtongning Tablets and Anti-flu Granules. We held the Certificates
of Protected Variety of Traditional Chinese Medicine (Grade Two) (the
“Certificate of Protection”) issued by State Food and Drug Administration of
China (“SFDA”) for Tongbi Capsules and Shangtongning Tablets which give us
exclusive or near-exclusive rights to manufacture and distribute these two
medicines. The protection periods for both Tongbi Capsules and
Shangtongning Tablets expired in September 2009 and we are seeking to extend the
protection periods. We have submitted the application of Tongbi Capsules
to extend such protection period on March 12, 2009 and SFDA has recently started
its review process. We expect such approval may be granted by end of June
this year. We have decided not to submit extension application of
Shangtongning Tablets, because the SFDA will not approve Certificate of
Protection for Shangtongning Tablets or any other products that are currently
produced by more than three manufacturers in China according to applicable
Chinese SFDA regulations.
As of
August 1, 2010, we had approximately 600 employees, including approximately 300
sales representatives, operating from 20 offices throughout China. More
than 50% of our workforce is engaged in sales and distribution activities.
Our
strategy is to leverage the “protected” status and national insurance coverage
for certain of our pharmaceutical products to aggressively increase market
penetration throughout China, the world’s most populous nation. By
utilizing our distribution platform, in addition to utilizing mass media and
other marketing methods to build awareness of our brand, we will seek
significantly grow our revenues and earnings.
42
We are
placing particular marketing focus on our Lung Nourishing Cream over the next
several years. Lung Nourishing Cream is one of our most popular products,
and its main ingredient, Laiyang Pear, to our knowledge, is not available in
other similar pharmaceutical products. We applied for a patent for Lung
Nourishing Cream with its production method for the treatment of Lung-qi
Deficiency Cough and Chronic Bronchitis, which application was approved by the
State Intellectual Property Office of the PRC on June 23, 2010. For these
reasons, we believe Lung Nourishing Cream contains a novel formulation for the
treatment of asthma and other common respiratory ailments with an emphasis on
the improvement of overall lung function and health. We believe this
represents an exceptional market opportunity.
Our
business strategy will seek to capitalize on new government programs established
in early 2009 to extend health insurance coverage to previously uncovered
Chinese citizens. The PRC government’s new health care policies are also
designed to encourage the use of TCM and its approach to wellness and treatment
of disease. As a result, the government continues to expand the number of
TCMs eligible for reimbursement under national medical insurance programs.
This has resulted in medical professionals working in the state-run medical
facilities to increasingly prescribe and recommend TCM products of the type we
manufacture and market. The state-run facilities provide the majority of
medical care in China. Two of our lead products, Lung Nourishing Cream and
Tongbi Capsules and Tablets, are authorized to be eligible for insurance
coverage, with others expected to follow. Currently public health
officials in China are developing general consumer awareness of increasing
problems and concerns with respiratory and lung health caused by pervasive
national air pollution. This nationwide epidemic is an unfortunate
by-product of the robust development of China’s expansive manufacturing and
industrial activities. Increased air pollution is a cause and contributory
factor to a range of acute respiratory illnesses including chronic conditions
such as asthma. As a result, we intend to significantly increase our
advertising for our Lung Nourishing Cream.
As is not
uncommon for Chinese companies listed in the U.S., we control our Chinese
operating subsidiary, Bohai, pursuant to the VIE Agreements, under which we
assume management of the business activities of Bohai and have the right to
appoint all executives and senior management and the members of the board of
directors of Bohai. Under these arrangements, however, we do not,
directly or indirectly, own any shares in Bohai, which are owned by Mr. Qu, our
Chairman, President and Chief Executive Officer and two unaffiliated
parties.
Overview
of Traditional Chinese Medicine
In China,
Traditional Chinese Medicine is not an alternative form of therapy but is used
in the state-run hospitals alongside modern medicine. For its
practitioners and advocates, TCM is a complete medical system that is used to
treat disease in all its forms. TCM is also believed to promote long
term wellness and vigor. Many modern-day drugs have been developed from herbal
sources. These include drugs designed to treat asthma and hay fever
such as ephedrine; hepatitis remedies from fruits and licorice roots and a
number of anticancer agents from trees and shrubs.
For the
Chinese, however, health is more than just the absence of
disease. Chinese herbal medicine is not only intended to cure but to
enhance the capacity for enjoyment, fulfillment and
happiness. Accordingly, there are herbal drugs that are used to
invigorate, nourish blood, calm tension and regulate menstruation.
The roots
of TCM date back thousands of years and include a number of therapeutic
approaches. These include herbal medications, acupuncture, dietary manipulation,
massage and others. Very early works of Chinese medical literature date back as
much as 2,500 years while other classics appeared approximately 2,000 years ago
during the Han Dynasty. Medicine in China continued to develop throughout the
Middle Ages when emperors commissioned the creation of various scholarly works
that compiled and documented hundreds of medicines derived from herbs, animal
sources and minerals. In addition, these works described their therapeutic uses.
In the 1950s, TCM was further modernized and reformed by the PRC
government.
43
The
emphasis on wellness and the avoidance of disease is considered by some to be a
key distinction between TCM and western medical practice which has been seen as
more heavily oriented toward the treatment of disease and less toward
prevention. While TCM has remained a substantial part of medical
treatment in China and throughout East Asia, recent decades have seen increasing
acceptance throughout the United States, Europe and elsewhere. This
growth is, in part, driven by increasingly educated and empowered consumers of
medical care who seek organic, natural and alternative approaches to western
medical treatments and prescription drugs. Medical doctors are also
accelerating the process of acceptance, as doctors trained in the western
tradition in Europe, the United States and elsewhere are integrating TCM and
alternative treatments in their everyday practice. Additionally, a
growing number of physicians specifically trained in TCM, acupuncture and other
modalities are opening offices in communities in the U.S. and around the
world.
We
believe that the sales of TCM in China reflect the central and still growing
role these therapies play in medical care in that nation. According
to Helmut Kaiser Consultancy, in 2005, total sales revenue for Chinese herbal
medicine manufactured in China was $13.6 billion which accounted for 25.8% of
all medicine manufactured in China. This segment had total profit of
$1.76 billion which accounted for 29% of the total profit of the Chinese drug
industry. In 2006, there were approximately 1,400 Chinese herbal
medicine manufacturers with an annual growth rate of 15%, much higher than the
comparable period GDP growth. According to Helmut Kaiser Consultancy,
as a result of the increasing wealth of China and an aging population, it is
estimated that by 2010, China will be the fifth largest market for herbal
medicines in the world exceeding more than $24 billion in sales.
According
to a published report by PricewaterhouseCoopers, in 2009 China had more than
7,000 distributors supplying pharmaceuticals to hospitals and
pharmacies. According to such report, most Chinese seeking medical
care go directly to the hospitals where more than 80% of the medicines used
throughout China are prescribed. Only recently have chain drug stores
begun to appear allowing drugs to be obtained in many areas without a visit to a
hospital.
Our
Products
Overview
We
obtained Drug Approval Numbers for 29 varieties of traditional Chinese herbal
medicines in 2004 and currently produces 10 TCM pharmaceutical products, all
derived from herbal and organic sources. These include both
prescription and non-prescription over-the-counter (OTC)
medicines. The first five-year valid terms of such Drug
Approval Numbers have expired. We submitted the applications for
re-registration on June 29, 2007 which were accepted by SFDA. We have
been advised that the approval processes for these drugs has been recently
commenced by the Shandong Branch of SFDA. During the renewal period,
we are permitted to continue manufacturing these drugs as if the renewals had
been approved.
The
following is a list of approved pharmaceutical products that we are producing
with their intended uses:
Lung Nourishing
Cream. Lung Nourishing Cream is designed to moisten the lung
and relieve coughs and can be used to treat weak lung and chest tightness, poor
chronic cough, shortness of spontaneous breath and chronic
bronchitis.
44
Tongbi
Capsules. This product is designed to promote blood
circulation and relieve swelling and pain, and can be used to treat cold
resistance, liver and kidney deficiency, arthralgia syndrome and rheumatoid
arthritis.
Tongbi
Tablets. Tongbi Tablets are designed to regulate and fortify
the blood promote blood circulation and relieve swelling pain and can be used to
treat alpine resistance, liver and kidney deficiency, including rheumatoid
arthritis.
Shangtongning
Tablets. This product is designed to alleviate pain and can be
used to treat bruises.
Zhuangyuan shenhailong Medicinal
Wine. This liquid product is designed to promote kidney
function and can be used to treat the weakness waist and knee fatigue, insomnia
and forgetfulness.
Danqi Tablet. This
product is designed to improve blood circulation and can be used to treat blood
stasis, cardio-thoracic pain, dizziness and headache, and menstrual
pain.
Fukangning
Tablet. This product is designed to improve blood circulation
and can be used to treat blood stasis, cardio-thoracic pain, dizziness, headache
and menstrual pain.
Bazhen Yimu
Cream. This product is designed for menstruation conditioning
and can be used to treat dizziness, palpitation, fatigue, weakness and other
menstrual symptoms and can also be used to ease menstrual flow.
Huoxue Shujin
Ting. This product is designed to promote blood circulation
and relieve blood congestion, and can be used to treat pain in the waist and
leg, numbness in the feet and hands and arthritis.
Anti-flu
Granules. This product is designed to detoxify the body, and
can be used to treat cold caused by exogenous wind-heat with symptoms such as
fever, headache, stuffy nose, sneezing, pharyngodynia, generalized weakness and
soreness.
Of our 10
products, currently 5 (Tongbi Tablets, Tongbi Capsules, Shangtongning Tablets,
Danqi Tablets and Huoxue Shunjin Ting) are available only through
prescription.
In
addition to the 10 medicines currently in production, we hold the rights to
produce 19 other herb-based pharmaceutical formulations. We
anticipate that we will commence the manufacturing and distribution for these
additional products if and when appropriate business conditions
develop.
Product
Types
Bohai has
five production lines for the manufacturing of medicines in five forms,
including tablets, granules, capsules, syrup, and medicinal wine. In
our fiscal year June 30, 2009, we produced:
|
|
·
|
1.35
billion tablets and capsules;
|
|
|
·
|
30
million bags granules (370 million
granules);
|
|
|
·
|
15
million bottles/units of concentrated
decoctions;
|
|
|
·
|
10
million bottles/units of syrup;
|
45
|
|
·
|
1
million bottles/units of tinctures;
and
|
|
|
·
|
1
million bottles/units medical wine
|
One of
our pharmaceutical products have been granted “protected” status by the PRC
government, a marketplace classification used by the government to regulate both
production and distribution of traditional and herbal
medicines. These “protected” medicines are not patented in the
traditional commercial sense but are essentially proprietary. The
protection refers, in part, to standardizations of formulae which require that
medicines of the same name have the same type and proportion of
ingredients. The “protected” designation grants us exclusive
manufacturing and distribution rights within China over certain protected
products or with up to six manufacturers in other cases.
We have
the exclusive rights to manufacture Tongbi Capsules and we share manufacturing
rights with one or more manufacturers for Shangtongning Tablets and Anti-flu
Granules. The exclusive rights usually have a term of seven years and
can be extended for another seven year period after the initial seven year
period elapses. The protection periods for both Tongbi Capsules and
Shangtongning Tablets expired in September 2009 and we have filed an application
for extending the protection period on March 12, 2009 for Tongbi Capsules. We
have decided not to submit extension application of Shangtongning Tablets,
because the SFDA shall not approve Certificate of Protection for Shangtongning
Tablets or any other products that are currently produced by more than three
manufacturers in China according to applicable Chinese SFDA
regulations. The shared manufacturing rights for Anti-flu Granules
expire on July 9, 2012.
During
2010, we expect to increase marketing and advertising of Tongbi Capsules and
Tablets, which are formulated to treat various forms of
arthritis. Sales of our Tongbi medicines are expected to grow in
2010. In addition to the Tongbi medicines and the Lung Nourishing
Cream, other substantial contributors to our revenues include its Shangtongning
Tablets which are also expected to grow in 2010. Sales of our OTC
product Bazhen Yimu Cream, used to strengthen the immune system, the enhancement
of physical strength and conditioning, are also projected to grow in
2010.
We price
our medicines well under government-mandated caps and at a premium to most
competitors because we use high quality raw materials and rely on strict quality
control and management to produce high quality finished products. We
therefore believe, subject to applicable clinical analysis, that the purity,
potency and effectiveness of our ingredients are superior to similar products in
the Chinese marketplace.
Overview
of the Chinese Market
The
People’s Republic of China is undergoing the world’s most important and powerful
economic transformation. This transformation includes the confluence of
its ancient culture with modern trends in business, technology and
finance. As a result, Chinese operating companies are capitalizing on
unmatched growth opportunities in this evolving and growing
marketplace. Although average income is approximately one-tenth that
of developed western nations, business growth and market reform-driven policies
have given the country’s 1.3 billion citizens more purchasing power than
ever.
46
According
to a report published in Newsweek, total consumer spending in China reached $1.7
trillion in 2007, compared with $12 trillion in the U.S. In its China
Consumer Survey published in January 2010, Credit Suisse found that household
income in China of the bottom 20% of those surveyed rose by 50% since 2004,
while the top 10% had grown 255% to around RMB34,000 per
month. Credit Suisse expects China’s share of global consumption to
increase from 5.2% at US$1.72 trillion in 2009 to 23.1% at US$15.94 trillion in
2020, overtaking the U.S. as the largest consumer market in the world. Further,
research on Chinese consumers by management consulting firm McKinsey classifies
two million households out of a population of 1.3 billion as “wealthy,” based on
fairly modest annual earnings of more than $30,000. An enormous
middle class is rising, however, numbering some 70 million urban households, but
these still earn $5,000-$10,000 a year. China’s National Bureau of
Statistics, based on a random survey of 65,000 urban households in China, found
that the average (annual) disposable income of urban residents in the first half
of 2009 was U.S. $1,300, an increase of 9.8% compared to the same period last
year. When price factors are deducted, this is equivalent to a real
increase of 11.2%. The average consumption expenditure amount of
urban residents in the first half of 2009 was U.S. $876, an increase of 8.9%
compared to the same period last year. When price factors are deducted, this is
equal to a real increase of 10.3%.
TCM
Industry Drivers
We
believe that demographic, governmental and related factors in the China will be
favorable to growth and expansion of our business.
Growing Prosperity of the Chinese
People. The increased spending power of China’s population
continues to be reflected in the increased consumption of health products and
medical services between 2007 and 2010. According to Euromonitor
data, spending by Chinese people on these goods and services will increase from
$100 billion in 2007 to $145 billion in 2010.
Population
and Aging
|
|
·
|
The
total population of China was 1.32 billion at the end of 2007, according
to official government estimates.
|
|
|
·
|
Due
to improved healthcare, the elderly population of China is
growing.
|
|
|
·
|
The
health/medical costs associated with care for elderly in China are
approximately five (5x) times that of younger
people.
|
|
|
·
|
China
had 170 million elderly people in 2007 but will have an expected 230
million elderly by 2015 according to “Consumer Lifestyles in China:
Consumer Trends, China’s Grey Population,” by Euromonitor,
2009.
|
|
|
·
|
The
proportion of the China’s population aged 65 and over will rise from just
10% of the overall population in 1995 to 22% by 2030, according to the
World Bank.
|
|
|
·
|
From
1995 to 2030 it is estimated that the ratio of working-age people to
pensioners will decrease from 9.7:1 to 4.2:1. China’s national
estimates vary slightly from World Bank figures, but still show in
increase in the proportion of the population over 65 years from 7% in 2000
to 9.4% in 2007, according to China Country Profile 2009, The Economist
Intelligence Unit Ltd.
|
Government Policies in Health Care
and TCM. In April of 2009 the PRC government implemented a new
national medical and health plan. Among other features, this new plan
extended national medical insurance coverage to China’s rural areas, where the
bulk of the population resides. This expanded coverage will
eventually encompass virtually all of China’s 1.3 billion citizens, greatly
expanding the market for TCM pharmaceuticals, as well as other health care
products and services. This has led to massive potential for
increased sales growth for Bohai and other providers of TCM pharmaceutical
products.
47
According
to Espicom Business Intelligence, in the next three years, the PRC government’s
health care investment will rise to $125 billion, compared with $96 billion for
2008. Direct health care subsidies of urban and rural residents will
amount to $57 billion. China’s health care investment is expected to
witness a growth of 19.7% and the overall growth rate will reach more than
25%.
Government Support of Traditional
Chinese Medicine. Among its public health initiatives, the
Chinese government officially supports use of TCM to enhance wellness and to
treat chronic and acute diseases. The government has also commenced a program to
evaluate TCM and herbal-based pharmaceuticals for coverage and reimbursement
under national medical insurance. In 2002, TCM was declared a
“national strategic industry” in the government’s “Development Outline of
Traditional Chinese Medicine Modernization (2002 – 2010).”
Decreased
Competition. According to the Information Office of the State
Council of the PRC, prior to 2009, there were approximately 6,000 Chinese
pharmaceutical manufacturers. That number is being significantly
reduced through both marketplace attrition and direct government involvement,
decreasing competition and increasing potential sales opportunities for the
surviving companies. Other companies are expected to fail through
lack of size and innovative and aggressive management. According to a
2009 report published by KMPG, of the approximately 4,500 pharmaceutical
companies in China, the majority are small players with limited local market
reach, and rapid consolidation between medium and large players in the sector is
anticipated since the Chinese government has been encouraging industry
consolidation with an effort to improve the Good Manufacturing Practice (GMP)
standard, enforce GMP certification and to better control the pricing of
drugs..
Growth
Strategy
Our
strategic initiatives for the foreseeable future are designed to aggressively
capitalize on the health and wellness needs of increasingly wealthy and
empowered consumer class in China. In particular, we are seeking to
capitalize on the government policies that extended medical insurance in 2009 to
hundreds of millions of Chinese citizens living in rural areas, representing a
vast untapped market of potential consumers who previously lacked access to
national medical insurance. As part of its reforms to expand and
improve public health and medical care, the PRC government is promoting the use
of herbal-based TCM and expanding insurance coverage to 100% of an increasing
number of medicines.
Our
strategic initiatives include the following:
Grow Hospital
Presence. We have targeted 600 hospitals in 100 locations
throughout China for direct marketing of Bohai products. As part of
this initiative, our sales team will expand its marketing activities to educate
hospital personnel about the our product lines and train hospital employees in
the preventative and curative qualities of these products. The
initial focus will capitalize on the best known and most popular of our
products, such as Tongbi capsules and Shangtongning tablets, using these as
door-openers for our other medicines.
The
average cost to “open” each hospital to our products is $1,500 with the
following provinces targeted:
48
|
Cities
|
Number of Hospitals
|
|
|
Zhejiang
|
100
|
|
|
Jiangsu
|
80
|
|
|
Anhui
|
150
|
|
|
Shandong
|
200
|
|
|
Sichuan
|
20
|
|
|
Hubei
|
100
|
Build Awareness of the Lung
Nourishing Cream. We have allocated a significant portion of
the proceeds from our January 2010 private placement for
brand-building. We will primarily target consumers through television
and print advertising to expand awareness of the uses and effectiveness of our
Lung Nourishing Cream. The advertising will incorporate messaging
targeting smokers and workers with occupational diseases as well as city
dwellers exposed to smog. It is expected that the consumer television
advertising program will initially be focused in the following areas:
|
TV Station Location
|
|
Shandong
|
|
Anhui
|
|
Hubei
|
|
Sichuan
|
|
Chongqing
|
|
Shanxi
|
|
Jiangsu
|
|
Liaoning
|
Expand to Rural
Areas. We expect to execute a comprehensive marketing campaign
targeting 100 rural counties as a result of the national government’s emphasis
on expansion of healthcare and health insurance into the country’s rural
areas. We plan on starting its rural marketing in Shandong, Anhui,
Liaoning and Hubei. Our sales team will market its products to
pharmacies, hospitals, physicians, herbal medicine experts, media outlets and
other opinion leaders in these rural areas. The main purpose is to be
listed in the New Rural Cooperative Medical Directory which is farmer-friendly
and assists these rural dwellers with reimbursement of medical
expenses.
Generally,
the cost of this professional relationship-building with each rural county is
$3,000 with a listing fee in the New Rural Cooperative Medical Directory costing
in excess of $1,000,000. We planned to hire 80 sales people for this
effort and to equip each with a minivan to enable in-person
contacts. Each employee will be responsible for 5
counties. 30 sales people for this effort have been hired as of the
date of this prospectus. All of these sales and marketing initiatives
will involve both OTC and prescribed products.
49
Build Product Line and Product
Awareness. Brand awareness marketing will include the
promotion and introduction to new markets of the current popular Bohai products
such as Tongbi Tablets, Lung Nourishing Cream and Champion Sea Dragon Wine
Medicine. As part of its increase in sales and marketing staff, we
plan to have special trainers and presenters who can conduct promotional events
and seminars to increase awareness of our products.
Seek Acquisitions of Complimentary
Companies or Assets. We believe that there may be TCM
companies or assets in China that would be complimentary with our current
product offerings and which could fit well with our sales and marketing
platform. We may seek to acquire such assets or companies as a means
to grow our revenue and profitability.
Competitive
Advantages
We
believe there are several key factors that will continue to differentiate us
from our competition in the PRC:
|
|
·
|
“Protected” Status of Bohai
Products. One of our 10 products currently enjoys
protected status by the PRC government. We have submitted an
application for extending the protection period for this
product. This status regulates competition, granting us
exclusive or near-exclusive rights to manufacture and sell the protected
products.
|
|
|
·
|
Insurance Coverage for Lead
Bohai Products. Two of our lead products, Lung
Nourishing Cream and Tongbi Capsules and Tablets, are listed in the
Catalogue Eligible for Medicine Reimbursement as of December 1,
2009. This means that these medicines are eligible for
reimbursement under the national health insurance.
|
|
|
·
|
Relatively
low research and development (including acquisition) costs of TCM,
compared with western pharmaceutical companies.
|
|
|
·
|
Highly
trained sales force of approximately 300 people as of August 2010.
|
Raw
Materials and Suppliers
The
principal raw materials used for the production of our distributed products are
honey, laiyang pear paste, Sichuan fritillaria, pangolin, and Chinese
angelica. Raw materials, as well as packaging materials, are sourced
from various independent suppliers in the PRC. We have no long term
agreements with our suppliers, and purchase raw materials on a purchase order
basis. We try to maintain relationships with at least two vendors for
each major raw material in order to ensure a reliable supply of raw materials at
reasonable prices. We believe there is ample supply in the market for
the foreseeable future of the ingredients for our products. Our
principal suppliers include Anhui Dechang Pharmaceutical Tablet Co., Ltd.,
Shandong Yantai Medical Materials Purchasing and Supply Station, Zibo Taibao
Forgery-proof Product Co., Ltd., and Zhejiang Yuhuan Kangning Medicine Packaging
CO., Ltd. In the fiscal year ended June 30, 2009, one supplier
accounted for over 10% of our purchases of raw materials, although currently no
single supplier accounts for over 10% of our purchases of raw
materials. Approximately 30% of the raw material is purchased from
Anhui Dechang Pharmaceutical Tablet Co., Ltd., Shandong Yantai Medical Materials
Purchasing and Supply Station and Shandong Cangli Medicine Co.,
Ltd.
50
Research
and Development
We
currently have limited resources to devote to and limited capabilities to
conduct the development of new products; and as such research and development
activities are not presently material to our business. We have only
one full-time employee who is engaged in research and development, so we mainly
dependent on a third-party, Yantai Tianzheng Medicine Research and Development
Co., Ltd., to perform research and development for us. We currently
have two products, namely Forsythia Capsule and Fern Injection, under research
and development in association with Yantai Tianzheng Medicine Research and
Development Co., Ltd.
We, like
other TCM manufacturers, enjoy relatively low research and development expenses
as most TCM medicines are based on standardized formula. In 2008,
SFDA promulgated a notice of registration of Chinese traditional medicine
providing that TCM composed of classic prescriptions shall be exempted from
pharmacological and toxicological tests and studies. The notice
defined classic prescription and classic TCM formulas as those herbal remedies
recorded in ancient Chinese medicine books from Qing Dynasty or earlier which
are currently widely used. According to such notice, the production
and manufacturing of TCM products are subject to non-clinical safety studies
only and exempted from pharmacological and toxicological tests and
studies. Thus, TCM products are entitled to obtain faster SFDA
approval. As such, we enjoy relatively low research and development
expenses because most of our products are based on classic TCM formulas that are
covered by this notice.
The
research and development process includes toxicological tests, pharmalogical and
qualitative research, preparation for production and other miscellaneous
costs. We intend to introduce one new product by 2014 which is
currently identified as a Shujin Pain Relief soft capsule. The total
cost to develop this product is not expected to exceed $150,000. We
may also seek to acquire new products through acquisitions of other TCM
companies in the future.
Manufacturing
Although
TCM is thousands of years old, we believe that our product manufacturing and
procedures are the most modern and up-to-date available. We employ
rigorous standards for product quality control and safety. The
manufacturing facility owned by us is conducted in the city of Yantai in
Shandong Province in a state-of-the-art 18,000 square-meter facility that meets
or exceeds the latest Good Manufacturing and Quality Management Practice
standards (referred to in China as “GMP”).
GMP
standards are the government’s benchmark for pharmaceutical manufacturers in
China and must be met for the manufacturer to be eligible to market domestically
or enter world markets. On March 31, 2009, we completed a GMP review
which included examination of 225 items including development technology,
production, quality assurance, quality control, material handling and
engineering. As a result of that review, we were been re-certified
for a new five-year period.
Through
stringent application of GMP standards, the PRC government has reduced the
number of marginal medicine manufacturers by one-third, from 6,000 to
4,000. It is expected that TCM and pharmaceutical companies such as
ours that have received full GMP approval by the government will enjoy the
competitive benefits of their strict adherence to quality control, safety,
health and manufacturing standards.
Our
advanced and mechanized facilities utilize controlled, clean-room procedures
with sophisticated water filtration and materials processing
systems. In our annual operations, we process 800 tons of herbal
plants to extract, isolate and purify the compounds used in its medicines and
health supplements. The
manufacturing staff consists of approximately 200 production employees and
approximately 20 quality control inspectors as of August 2010.
51
Marketing,
Sales and Distribution
Bohai’s
products are sold either by prescription through hospitals or Over The Counter
(OTC) through local pharmacies and retail drug store chains. Sales and
distribution are managed and executed by approximately 300 sales representatives
located in 20 offices throughout China as of August 2010. These
employees are trained in all details of each product and are encouraged to
develop strong ties with physicians, hospitals and pharmacies in their local
areas.
Our
distribution and marketing initiatives for the next several years will be
focused on achieving the following goals:
Expand hospital
presence. We intend to further develop business in 600
hospitals in provinces we already serve including Shandong, Zhejiang, Jiangsu,
Anhui, Sichuan and Hubei. We believe that we will generate additional
revenue from the newly developed hospitals in those provinces.
Expand distribution to the rural
market. We believe that the Chinese government’s expansion in
2009 of national medical insurance coverage to the rural population provides us
with new and largely untapped markets. Some of our products, namely
Lung Nourishing Cream, Tongbi Capsules, Tongbi Tablets and Bazhen Yimu Cream
have been listed in government’s New National Medical Insurance Catalogue in
Shandong and Anhui Province as of November 30, 2009, and we expect to gain a
competitive advantage in these newly accessible rural markets.
During
2010, we plan to develop relationships with 100 new county-level hospitals in
rural areas of Shandong, Anhui, Liaoning and Hubei to establish its primary
marketing and distribution network in these rural areas.
Expand prescription medicine sales
organization. A key element of our growth strategy is
increased outreach to physicians. These outreach programs will focus
on the five current pharmaceutical products of ours that are available by
prescription only and will typically take place at state-run hospitals where
virtually all Chinese citizens obtain their medical care. Our
educational training programs will be designed to inform doctors of the range of
our pharmaceutical products including diseases or health/wellness concerns
targeted and proper usage of the medicines. Management will allocate
a portion of the proceeds derived from our January 2010 private placement to
expand existing physician-education programs.
Expand OTC team to drive market
share. Our management intends to accelerate and expand sales
of our OTC medicines through promotion and advertising targeting
consumers. The marketing programs will principally utilize
television, print advertising and news releases.
Competition
China’s
domestic pharmaceutical industry is highly competitive, with hundreds of
companies vying to reach consumers through more than 100,000 pharmacies.
In some categories in which we compete there are many other companies
offering the same competitive products. The market continues to attract
new entrants because the per capita medicine consumption in China is still low,
compared to developed countries, and that shows promise for substantial
growth.
52
Competitive
factors primarily include price and quality. We believe that we are able to
effectively compete in our market segment in China based upon the quality of our
product, given our new GMP certified manufacturing facility and our reputation
in the market place.
Many of
our current and potential competitors have significantly longer operating
histories and significantly greater managerial, financial, marketing, technical
and other competitive resources, as well as greater name recognition, than we
do. These competitors may be able to respond more quickly to new or changing
opportunities and customer requirements and may be able to undertake more
extensive promotional activities, offer more attractive terms to customers or
adopt more aggressive pricing policies. We cannot assure you that we will be
able to compete effectively with current or future competitors or that the
competitive pressures we face will not harm our business.
I
ntellectual Property
We market
our products under the trademark “Xian Ge” which is registered with the PRC
Trademark Bureau under the State Administration for Industry and
Commerce. Currently, another company is licensed to utilize our
registered trademark “Xian Ge”. We have received a patent in the PRC
for our Lung Nourishing Cream with its production method for the treatment of
Lung-qi Deficiency Cough and Chronic Bronchitis.
Government
Regulation
We are
subject to many general regulations governing business entities and their
behavior in China and in any other jurisdiction in which we have operations. In
particular, we are subject to laws and regulations covering food, dietary
supplements and pharmaceutical products. Such regulations typically
deal with licensing, approvals and permits. Any change in product
licensing may make our products more or less available on the
market. Such changes may have a positive or negative impact on the
sale of our products and may directly impact the associated costs in compliance
and our operational and financial viability.
Our only
sales market is presently in China. We are subject to the
Pharmaceutical Administrative Law, which governs the licensing, manufacturing,
marketing and distribution of pharmaceutical products in China and sets
penalties for violations of the law. We are also subject to the Food
Sanitation Law, which provides for the food sanitation standards to be
followed.
Under
SFDA guidelines for licensing of pharmaceutical products, all pharmaceutical
manufacturers must obtain and maintain GMP Certificate. We hold a GMP
Certificate (No. Lu K0587) issued by Shandong Branch of SFDA on June 18,
2009. Because our manufacturing facility has obtained the National
GMP Certificate, we are authorized to produce products in seven modes which
are tablets, capsules, granules, syrup, concentrated decoctions, tincture and
medical wine. Such certificate expires on June, 14,
2014.
We hold a
Permit for the Production of Medicine (Lu Zb20050330) issued by Shandong Branch
of SFDA on January 1, 2006 which allows us to engage in the production of
tablets, capsules, granules, syrup, concentrated decoctions, tincture (for oral
use) and medical wine. Such permit expires on December 31, 2010 and
we are planning to submit renewal application in compliance with the
requirements of the Shandong Branch of SFDA.
The
Permit for the Production of Medicine and GMP certificates are each valid for a
term of five years and must be renewed before their expiration.
53
We
obtained a Drug Approval Number for each of our products in 2004. The first five
year valid terms of such Drug Approval Numbers have expired. We
submitted the applications for re-registration on June 29, 2007 which were
accepted by SFDA. We have been advised that the approval processes
for these drugs have been recently commenced by the Shandong Branch of
SFDA. During the renewal period, we are permitted to continue
manufacturing these drugs as if the renewals had been approved.
The
governmental approval process in China’s newly developed health product is as
follows: a product sample is sent to a clinical testing agent designated by the
Ministry of Health, which conducts extensive clinical testing and examinations
to verify if the product has the specified functions as stated by the company
producing the product. A report will be issued by the clinical
testing agent confirming or negating such functions. It generally takes
approximately six months to one year for the report to be
issued. This report then has to be submitted to a provincial Health
Management Commission for approval. A letter of approval issued by such
commission will then be submitted to the Ministry of Health for the issuance of
a certificate that authorizes the sales and marketing of the product in China.
The whole process generally takes one and a half to two years.
Because
our subsidiary Yantai Shencaojishi Pharmaceuticals Co., Ltd. is a wholly foreign
owned enterprise, we are subject to the law on foreign investment enterprises in
China, and the foreign company provisions of the Company Law of China, which
governs the conduct of our subsidiary and its officers and directors.
Additionally, we are also subject to varying degrees of regulations and permit
system by the Chinese government.
Currently
we have not developed a market in U.S. so we believe we are not subject to any
of regulations by the U.S. Food and Drug Administration.
Environment
Regulation
We
believe we are in compliance with the Environmental Protection Law of the PRC,
as well as applicable local regulations, except that as of the date of this
Memorandum we are in the process of applying for the Pollution Discharge Permit.
Zhifu District Branch of Yantai Environment Protection Bureau issued a
written document on September 10, 2009, and stated that we have never been
subject to sanction due to violation of relevant environmental protection laws
since the incorporation. In addition to compliance with PRC laws and
local regulations, we consistently undertake active efforts to ensure the
environmental sustainability of our operations. Because the manufacturing
of herb and plant-based products does not generally cause significant damage or
pollution to the environment, the cost of complying with applicable
environmental laws is not material. In the event we fail to comply with
applicable laws, we may be subject to penalties.
Properties
Our
corporate headquarters is located at No. 9 Daxin Road, Zhifu District, Yantai,
Shandong Province, China. Under the current PRC laws, land is owned
by the state, and parcels of land in rural areas which is known as collective
land is owned by the rural collective economic organization. “Land
use rights” are granted to an individual or entity after payment of a land use
right fee is made to the applicable state or rural collective economic
organization. Land use rights allow the holder of the right to use the land for
a specified long-term period. We have a land use right, expiring in
2047, for a total of approximately 30,637 square meters of land, on which we
maintain our manufacturing facility. We currently have not obtained a
land use right certificate for a piece of land located in Xingfu Twelve Village
of Zhifu District with the area of 11,222 square meters, on which we maintain
our corporate headquarters. In the process of the planning of Yantai
City, the usage of the aforesaid land use right has been changed from
“industrial use” to “commercial use” and therefore, the approval process for the
land use right certificates on five relevant parcels of land including the land
occupied by us is suspended until the completion of the planning. We
can not assure you that we will eventually obtain the land use right certificate
for this land.
54
Employees
Substantially
all of our employees are located in China. As of August 1, 2010, we
had approximately 600 employees, including approximately 300 sales
representatives, operating from 20 offices throughout China. There
are no collective bargaining contracts covering any of our employees. We believe
our relationship with our employees is satisfactory.
We are
required to contribute a portion of our employees’ total salaries to the Chinese
government’s social insurance funds, including medical insurance, unemployment
insurance and job injuries insurance, and a housing assistance fund, in
accordance with relevant regulations. In the last three years, we
contributed approximately $62,930, $80,839 and $85,024 for the fiscal years
ended June 30, 2006, 2007 and 2008, respectively. We expect the amount of
contribution to the government’s social insurance funds to increase in the
future as we expand its workforce and operations.
January
2010 Share Exchange and Private Placement Transaction
We were
originally incorporated under the laws of the State of Nevada under the name
Link Resources Inc. on January 9, 2008. Our principal office was in
Calgary, Alberta, Canada. Prior to January 5, 2010, we were a public
“shell” company in the exploration stage since our formation and had not yet
realized any revenues. We entered into a Mineral Lease Agreement on
April 1, 2008 for two mining claims in Pershing County, Nevada, in an area known
as the Goldbanks East Prospect. We terminated the lease on July 7,
2009.
Share
Exchange with Chance High
Pursuant
to the Share Exchange Agreement entered into on January 5, 2010, we acquired
Chance High and its indirect, controlled subsidiary Bohai, a Chinese company
engaged the production, manufacturing and distribution in China of herbal
medicines, including capsules and other products, based on Traditional Chinese
Medicine. On January 5, 2010, pursuant to the terms of the Share
Exchange Agreement, we acquired all of the Chance High Shares of Chance High
from the Chance High Shareholders, and the Chance High Shareholders transferred
and contributed all of their Chance High Shares to us. In exchange,
we issued to Chance High Shareholders an aggregate of 13,162,500 newly issued
shares of our common stock. Certain of the Chance High Shareholders
are selling stockholders hereunder.
In
addition, pursuant to the terms of the Share Exchange Agreement, Zaradic, our
former sole officer and director, cancelled a total of 1,500,000 shares of
common stock owned by him. As a further condition of the Share
Exchange, effective as of January 5, 2010, Zaradic resigned from all of his
positions with our company and Qu, the former principal stockholder and
Executive Director of Bohai, was appointed as our President, Chief Executive
Officer, Interim Chief Financial Officer, Treasurer and Secretary and also,
effective January 16, 2010, as our sole director. In June 2010, Mr.
Qu relinquished the positions of Interim Chief Financial Officer, Treasurer and
Secretary and we appointed Gene Hsiao as our Chief Financial
Officer. On July 12, 2010, we appointed three independent directors
to our board of directors.
55
Private
Placement and Related Agreements
Securities Purchase
Agreement. On January 5, 2010, we entered into the Securities
Purchase with the Investors and Euro Pacific, as representative of the
Investors, relating to a private placement by us of 6,000,000 units consisting
of Notes and Warrants. The consummation of the private placement
resulted in gross proceeds to us of $12,000,000 and net proceeds of
approximately $9,700,000. Each unit consisted of a $2.00 principal
amount, two year convertible Note and a three year Warrant to purchase one share
of our common stock at $2.40 per share, subject to certain
conditions. Euro Pacific acted as the lead placement agent and
Chardan Capital Markets, LLC acted as co-placement agent of the private
placement.
Pursuant to the Securities Purchase
Agreement, we have agreed that we shall:
(a) Within
six (6) months of the closing of the private placement, appoint individuals
constituting a majority of “independent” directors (as defined under the Nasdaq
Marketplace rules) to the our board of directors, with one such director being
designated by Euro Pacific, and with at least two of such directors being fluent
in English. As of the date of this prospectus, we have fulfilled this
agreement.
(b) Within
six (6) months of the closing of the private placement, enter into a 24 month
agreement with a new Chief Financial Officer who is reasonably satisfactory to
Euro Pacific and who is proficient in: (i) U.S. generally accepted accounting
principals; (ii) transactions similar to the ones contemplated by Securities
Purchase Agreement; and (iii) U.S. public company listings and the related
filing and compliance requirements. As of the date of this
prospectus, we have fulfilled this agreement .
(c) Within
three (3) months of the closing of the private placement, enter into a 12 month
agreement with an investor and public relations firm that is reasonably
satisfactory to Euro Pacific. As of the date of this prospectus, we
have fulfilled this agreement.
Registration Rights
Agreement. In connection with the private placement, we
entered into the Registration Rights Agreement with the Investors which sets
forth the rights of the Investors to have the shares of common stock underlying
the Notes and Warrants issued in the private placement registered with the SEC
for public resale. The filing of the registration statement of which
this prospectus is a part is intended to satisfy certain of our obligations the
Registration Rights Agreement.
Pursuant
to the Registration Rights Agreement, we agreed to file a registration statement
on Form S-1 (“Registration Statement”) by March 5, 2010 (which agreement was
fulfilled) and use our commercially reasonable best efforts to have the
Registration Statement declared effective by the SEC within 160 days after the
required filing deadline to register 100% of the common stock underlying: (i)
the Notes, (ii) the Warrants and the Private Placement Warrants, and (iii) any
capital stock of the Company issued or issuable, with respect to the registered
shares of common stock as a result of any stock split, stock dividend,
recapitalization, exchange or similar event or otherwise, without regard to any
limitations on exercises of the Warrants.
The
Registration Rights Agreement provides that if we fail to file, obtain and
maintain effectiveness of Registration Statement due to “filing failure” or
“maintenance failure” (each as defined in the Registration Rights Agreement), we
shall pay to Investors, distributed pro rata, equal to one percent (1%) of the
aggregate purchase price paid for the Notes and Warrants (the “Registration
Delay Payments”), provided that in no event shall the aggregate amount of
Registration Delay Payments exceed, in the aggregate, six percent (6%) of such
aggregate purchase price, or $720,000.
56
Securities Escrow
Agreement. Also in connection with the private placement, we
entered into the Securities Escrow Agreement with Euro Pacific, as
representative of the Investors, our principal stockholder, Glory Period, and
the Escrow Agent. Pursuant to the Securities Escrow Agreement, Glory
Period has pledged and deposited a stock certificate representing 1 million
shares of our common stock (the “Escrow Shares”) into escrow in order to provide
security to the Investors in the event of an occurrence of an event of default
under the Notes. Upon the earlier to occur of the full repayment of
all amounts due to the Investors under the Notes or the conversion of fifty
percent of the principal face value of Notes into shares of common stock, the
Investors’ rights in and to the Escrow Shares shall terminate. Glory
Period is controlled by Qu through certain contractual relationships described
elsewhere in this prospectus.
Closing Escrow Agreement.
Pursuant to the Closing Escrow Agreement that we entered into in
connection with the private placement on December 10, 2009, we placed a total of
$240,000 of proceeds from the private placement (the “Holdback Amount”) with the
Escrow Agent. The Holdback Amount represents an amount sufficient to
satisfy the payment to the Investors of one quarterly interest payment due on
the aggregate principal amount of all Notes issued in the private
placement. If, subject to certain conditions and after applicable
notice and cure periods, an event of default is declared by Euro Pacific with
respect to our failure to make a quarterly interest payment to Investors, the
Escrow Agent shall disburse such portion of the Holdback Amount to the
Investors, and we shall be obligated to deposit additional amounts equal to the
Holdback Amount with Escrow Agent. At such time as seventy-five
percent of the aggregate shares of common stock underlying the Notes have been
issued upon conversion of the Notes, all remaining funds of the Holdback Amount
shall promptly be disbursed to us.
Certain Rights of Euro
Pacific. From and after the closing of the private placement,
we have agreed with Euro Pacific that if we decide to engage any placement
agent, underwriter or investment bank on a fee basis in connection with any
private placement of our securities or our affiliates and executive officers (a
“Subsequent Offering”) for a period of twelve (12) months from the date of the
closing of the private placement, we shall give prompt written notice of such an
event to Euro Pacific, and Euro Pacific shall be entitled to a 5 day right of
first refusal, beginning on the day Euro Pacific receives such written notice
from us of such Subsequent Offering, to act as agent or manager for such private
placement. In addition, Euro Pacific shall be entitled 10.0% of the
gross proceeds received by us with respect to any equity or equity-linked
financing transactions consummated within twelve (12) months from the closing of
the private placement with any investor introduced to is by Euro
Pacific.
Contractual
Arrangements with Bohai and their Shareholders
On
January 9, 2008 our company was incorporated under the laws of the State of
Nevada under the name Link Resources Inc. On July 2, 2009, we established our
wholly owned subsidiary, Chance High International Limited., in Hong Kong. Other
than the Company equity interest in Chance High, the Company does not own any
assets or conduct any operations. On November 23, 2009, Chance High established
one wholly owned subsidiary, Yantai Shencaojishi Pharmaceuticals Co., Ltd.
(“WFOE” or “Shencaojishi”) in Yantai, Shandong Province of PRC. Other than
Shencaojishi, Chance High does not own any assets or conduct any operations.
Shencaojishi was formed to operate Yantai Bohai Pharmaceuticals Group, Inc. (the
“Domestic Company” or “Yantai Bohai”) by contract.
57
On
December 7, 2009, the WFOE entered into a series of variable interest entity
contractual agreements (the “VIE Agreements”) with Bohai and its three
shareholders, including Mr. Hongwei Qu, Jianwei Wang and Lu Liang (the “Bohai
Shareholders”). Pursuant to the VIE Agreements, WFOE effectively
assumed management of the business activities of Bohai and has the right to
appoint all executives and senior management and the members of the board of
directors of Bohai. The VIE Agreements are comprised of a series of
agreements, including a Consulting Services Agreement, Operating Agreement and
Proxy Agreement, through which WFOE has the right to advise, consult, manage and
operate Bohai for an annual fee in the amount of Bohai’s yearly net profits
after tax. Additionally, Bohai’s shareholders have pledged their
rights, titles and equity interest in Bohai as security for WFOE to collect
consulting and services fees provided to Bohai through an Equity Pledge
Agreement. In order to further reinforce WFOE’s rights to control and
operate Bohai, Bohai’s shareholders granted WFOE an exclusive right and option
to acquire all of their equity interests in Bohai through an Option
Agreement. Accordingly, we have consolidated Bohai’s historical
financial results in our financial statements as a variable interest entity
pursuant to U.S. GAAP following the date of the agreements and combined such
results prior to the date of the agreements.
We have
been advised by PRC legal counsel AllBright Law Offices, in an opinion dated
December 31, 2009, that: (1) our inner-PRC shareholding structure complies
with PRC laws and regulations; (2) the contractual arrangements between the
WFOE, Chance High, Bohai and Bohai’s shareholders are valid and binding on all
parties to these arrangements and do not violate relevant PRC laws or
regulations; (3) the each of the WFOE and Bohai has the requisite corporate
power to own, lease and operate its properties, to enter into contracts and to
conduct its business and (4) each of the WFOE and Bohai is qualified to do
business in the respective jurisdiction of its establishment.
Equity Interest Pledge Agreement.
The WFOE and Bohai Shareholders have entered into Equity Interest Pledge
Agreements, pursuant to which each shareholder pledges all of his shares of
Bohai to the WFOE in order to guarantee cash-flow payments under the applicable
Consulting Services Agreement. The Equity Pledge Agreement further entitles the
WFOE to collect dividends from Bohai during the term of the pledge.
Consulting Service Agreement.
Bohai and the WFOE has entered into a Consulting Services Agreement,
which provides that the WFOE will be the exclusive provider of technology
services to Bohai and Bohai will pay all of its net income based on the
quarterly financial statements to the WFOE for such services. Any
such payment from the WFOE to the Company would need to comply with applicable
Chinese laws affecting payments from Chinese companies to non-Chinese
companies. See “Risk Factors – Risks Associated With Doing Business
in China.”
Operating Agreement. Pursuant
to the operating agreement among the WFOE, Bohai and each of Bohai Shareholder,
the WFOE provides guidance and instructions on Bohai’s daily operations and
financial affairs. The Bohai Shareholders must designate the candidates
recommended by the WFOE as their representatives on their respective boards of
directors. The WFOE has the right to appoint senior executives of Bohai. In
addition, the WFOE agrees to guarantee Bohai’s performance under any agreements
or arrangements relating to Bohai’s business arrangements with any third party.
Bohai, in return, agrees to pledge its accounts receivable and all of its assets
to the WFOE. Moreover, Bohai agrees that without the prior consent of the WFOE,
Bohai will not engage in any transactions that could materially affect its
assets, liabilities, rights or operations, including, without limitation,
incurrence or assumption of any indebtedness, sale or purchase of any assets or
rights, incurrence of any encumbrance on any of its assets or intellectual
property rights in favor of a third party or transfer of any agreements relating
to its business operation to any third party.
58
MANAGEMENT
The
following table sets forth the name, age, and position of our directors, our
executive officers and key employees as of the date of this
prospectus. Executive officers are elected annually by our board of
directors. Each executive officer or key employee holds his office
until he resigns, is removed by the board of directors, or his successor is
elected and qualified, subject to applicable employment
agreements. We have a classified board of directors under which each
of our directors is designated as a part of one of three separate classes, with
the directors in one class being elected annually by our stockholders at our
annual meeting of stockholders for a term of three years. Each
director holds his office until his successor is elected and qualified or his
earlier resignation or removal. As used below, the term “Bohai” means
Yantai Bohai Pharmaceuticals Group Co., Ltd., a PRC company and our operating
subsidiary.
|
Name
|
Age
|
Position/Director Class
|
||
|
Hongwei
Qu
|
35
|
President,
Chief Executive Officer and Chairman of the Board of Directors (Class 1)
|
||
|
Gene
Hsiao
|
47
|
Chief
Financial Officer
|
||
|
Ning
Tang
|
50
|
Vice
President – Operations
|
||
|
Hongbin
Shan
|
41
|
Vice
President – Sales and Marketing
|
||
|
Chunhong
Jiang
|
45
|
Secretary
and Treasurer
|
||
|
Chengde
Wang
|
62
|
Director
(Class 1)
|
||
|
Louis
A. Bevilacqua, Esq.
|
41
|
Director
(Class 2)
|
||
|
Adam
Wasserman
|
|
46
|
|
Director
(Class 3)
|
Hongwei Qu. Mr. Qu
became our President, Chief Executive Officer, Interim Chief Financial Officer,
Treasurer and Secretary as of January 5, 2010, and, became the sole director and
Chairman of the our board of directors effective as of January 16, 2010 upon
filing of Schedule 14(f) with the SEC on January 6, 2010 in compliance with
Section 14(f) of the Exchange Act. Mr. Qu relinquished the positions
of Interim Chief Financial Officer, Secretary and Treasurer in June
2010. From 2001 to May 2007, Mr. Qu was the founder and principal
officer of Yantai Hangwei Medical Trading Co., a PRC company engaged in the
wholesale of drugs and medical products and retail of medical devices. In May
2007, Mr. Qu took principal responsibilities for the acquisition of
Bohai. From May 2007 until present, Mr. Qu has served as the General
Manger and Executive Director of Boha. . Mr. Qu has significant experience in
the medical and pharmaceutical sectors in China. Mr. Qu graduated
from Shandong Economic University with a bachelor degree.
Gene Hsiao. Mr.
Hsiao was appointed as our Chief Financial Officer in June 2010. Mr.
Hsiao has over 15 years of experience in corporate finance and
management. Prior to his appointment with us, Mr. Hsiao served as
Chief Financial Officer for China Advanced Construction Materials Group Inc.
(Nasdaq:CADC) from 2008 to 2010, where he was responsible for all U.S. affairs
as well as corporate finance functions in China. From 2000 to 2008,
he served as Controller of Milligan and Company, LLC, where he managed the
overall accounting and financial reporting functions as well as the company’s
internal control processes. From 1997 to 1999, he served as Finance
Manager for J&J Snack Foods Corporation (Nasdaq:JJSF), where he was
responsible for financial reporting and SEC schedule
preparation. From 1995 to 1997, he served as Accounting Supervisor of
RCN Corporation (Nasdaq:RCNI) and as the Senior Operation Analyst at ARAMARK
Corporation from 1992 to 1995. Mr. received his B.S. degree from
Drexel University in Philadelphia.
59
Ning Tang was appointed as
Bohai’s Vice President — Operations in November 2007 and our Vice President –
Operations in June 2010. Mr. Tang has over 25 years of experience in
management of pharmaceuticals companies in China. Prior to his
appointment with Bohai, Mr. Tang served as General Manager for Yantai Xiangyu
Environmental Protection Equipment Co., Ltd. from 2004 to 2007, where he was
responsible for all affairs of corporate operations in China. He
served as Vice President of Yantai Rongchang Pharmaceuticals Co., Ltd. from 1998
to 2004, where he managed the departments of operation, administration,
manufacturing and product quality. From 1986 to 1998, he served
as Deputy Director for Yantai TCM Pharmaceuticals Corporation, where he was
responsible for production, product quality, purchase, research and development
and sales. He received his B.S. degree in international trade and
business from Shandong Economic University.
Hongbin Shan was appointed as
Bohai’s General Manager of Sales in May 2010 and as the our Vice President –
Sales and Marketing as of June 2010. Mr. Shan has over 10 years of
experience in sales, marketing and management. Prior to his
appointment with us, from 1994 to 2010, Mr. Shan served as General Manager for
the Qingdao Branch, Shandong Province of Shandong Green Leaf Pharmaceutical Co.,
Ltd. and manager of the Su-Min Region (including Jiangsu, Fujian, Hubei, Jiangxi
and Anhui provinces) where he was responsible for all affairs of marketing and
sales. He also served in the capacity of Assistant Director of the
Oncology Division, responsible for the national market of Shandong Green Leaf’s
tumor line. He received his B.S. degree from Yantai University and
educational certificate from the senior MBA program of Tsinghua
University.
Chunhong Jiang was appointed
as Bohai’s General Manager of Finance, Secretary and Treasurer in May 2007 and
as our Secretary and Treasurer in June 2010. Ms. Jiang has over 20
years of experience in corporate finance, accounting and
management. Prior to her appointment with Bohai, Ms. Jiang served as
Financial Manager for Yantai Furao Trading Group from 2004 to
2007. She served as Financial Manager and department director for
Yantai Garment Company, a subsidiary of China Garment Group from 1994 to 2003,
where she was responsible for overall accounting and financial reporting
functions. She served as statistician, accountant and financial chief for Yantai
Hardware Factory from 1987 to 1993, where she managed the overall statistics,
accounting and financial reporting functions. She graduated from
Shandong Economic University.
Chengde Wang became an
independent director of our company on July 12, 2010. Mr. Wang has
served as the director medical doctor and Ph.D./MD advisor of Beijing Shuntiande
Chinese Medicine Hospital since October 2005, where he is responsible for
managing medical practice and research projects. Prior to joining
Beijing Shuntiande Chinese Medicine Hospital, Mr. Wang worked at Guang Anmen
Hospital under China Academy of Chinese Medical Science and was the professor
and the chief physician in the Beijing University of Chinese
Medicine. Mr. Wang is an expert in Traditional Chinese Medicine and
has been honored by the P.R.C. State Council. He is a member of
National Committee of The Chinese People’s Political Consultative Conference, a
director of Cooperation Center of State Administration of Traditional Chinese
Medicine with Taiwan, Hong Kong and Macao, director and Secretary-General of the
Center of Traditional Chinese Medicine Society and expert of review committee of
National Essential Drugs Association. Mr. Wang graduated from Beijing
University of Chinese Medicine.
Louis A. Bevilacqua, Esq.
became an independent director of our company on July 12, 2010. From
October 2008 to present, Mr. Bevilacqua has been a partner in the Corporate and
Securities Group at the law firm of Pillsbury Winthrop Shaw Pittman LLP and is
resident in the firm’s Washington, DC office. Prior to joining
Pillsbury, Mr. Bevilacqua was a partner in the Business and Finance Group at the
law firm of Thelen LLP during the period from January 2003 through October
2008. Mr. Bevilacqua has broad experience in public offerings and
private placements of securities, Exchange Act compliance, angel and venture
capital financings, other areas of equity and debt financing and mergers,
acquisitions and other business combinations, including “roll up” and “reverse
acquisition” transactions. Mr. Bevilacqua is a leader of Pillsbury’s
China Capital Markets practice and has significant experience representing
China-based middle market public companies. Mr. Bevilacqua obtained
his JD from Fordham University School of Law in 1994, where he became a member
of the Order of the Coif, and he obtained his undergraduate degree from Fordham
University, where he graduated with honors.
60
Adam Wasserman became an
independent director of our company on July 12, 2010. Mr. Wasserman
has served as the chief financial officer of Gold Horse International, Inc.
(OTCBB:GHII) since July 2007, chief financial officer of Emerald Acquisition
Corporation (PINK:PEAR) since June 2010 and as a director of China Direct
Industries, Inc. (NASDAQ:CDII) since January 2010. Since November
1999, Mr. Wasserman has been CEO of CFO Oncall, Inc., a Weston, Florida-based
provider of consulting and accounting services specializing in SEC reporting,
financial reporting, budgeting and planning, mergers and acquisitions, audit
preparation services, accounting department supervision ,and internal
controls. Mr. Wasserman has previously served as the chief financial
officer of Explorations Group Inc. (January 2002 until December 2005), Colmena
Corp. (May 2003 until June 2004), China Wind Systems, Inc. (November 2007 to
December 2008), Genesis Pharmaceuticals Enterprises, Inc. (October 2001 until
October 2007), and other companies, all client companies of CFO Oncall,
Inc. From June 1991 to November 1999, he was Senior Audit Manager at
American Express Tax and Business Services, in Fort Lauderdale, Florida where
his responsibilities included supervising, training and evaluating senior staff
members, work paper review, auditing, maintaining positive client relations,
preparation of tax returns and preparation of financial statements and the
related footnotes. From September 1986 to May 1991, he was employed by Deloitte
& Touche, LLP. During his employment, his significant assignments
included audits of public (SEC reporting) and private companies, tax preparation
and planning, management consulting, systems design, staff instruction, and
recruiting. Mr. Wasserman holds a Bachelor of Science in Accounting
from the State University of New York at Albany. He is a CPA (New
York) and a member of The American Institute of Certified Public Accountants and
is a director, treasurer and executive board member of Gold Coast Venture
Capital Association.
Audit,
Nominating, Compensation Committees and Director Independence
Although
our board of directors is comprised of a majority of “independent” directors (as
defined under the Nasdaq Marketplace rules), our board of directors presently
does not have standing audit, nominating or compensation committees and the
entire board is performing the functions normally associated with an audit,
nominating and compensation committee. We expect that we will seek to
form audit and other board committees in a manner consistent with
exchange-listed companies at such time as we apply for a listing on an exchange.
As part
of obligations under the Securities Purchase Agreement in connection with our
January 2010 private placement, one of our directors is designated by Euro
Pacific Capital, the lead placement agent for such private
placement. Louis A. Bevilacqua is the director designated by Euro
Pacific Capital.
Communication
with our Directors
Stockholders
or other interested parties may communicate with our directors by sending mail
to Mr. Hongwei Qu, c/o Yantai Bohai Pharmaceuticals Group Co. Ltd., No.9 Daxin
Road, Zhifu District, Yantai, Shandong Province, China 264000.
Board
of Directors’ Meetings
During
our fiscal year ending June 30, 2010, we did not hold any meetings of the board
of directors, although our board of directors did act by unanimous written
consent.
61
EXECUTIVE
COMPENSATION
The
following table sets forth all cash compensation paid by Bohai, for the fiscal
years ended June 30, 2009 and 2008. The table below sets forth the positions and
compensations for each officer and director of Bohai during such
periods. All the officers were paid in RMB and the amounts reported
in this table have been converted from Renminbi to U.S. dollars based on the
June 30, 2009 conversion rate of RMB 6.8319 to $1.
COMPENSATION
SUMMARY TABLE
|
Name and
Principal
Position
|
Fiscal Year
|
Salary
($)
|
Bonus
($)
|
Stock
Awards
($)
|
Option
Awards
($)
|
Non-Equity
Incentive Plan
Compensation
($)
|
Change in
Pension Value
and
Nonqualified
Deferred
Compensation
Earnings
($)
|
All Other
Compensation
($)
|
Total
($)
|
|||||||||||||||||||||||||
|
Hongwei
Qu,
|
2008
|
(1) | $ | 14,480.5 | $ | 5,321.79 | — | — | — | — | — | $ | 19,802.25 | |||||||||||||||||||||
|
President
and
|
2009
|
(2) | $ | 14,644.5 | $ | 4,910.78 | $ | 19,555.28 | ||||||||||||||||||||||||||
|
Chief
Executive Officer
|
||||||||||||||||||||||||||||||||||
Option/SAR
Grants in Last Fiscal Year
We have
not granted any stock options to our executive officers or directors from
inception through the date hereof.
Director
Compensation
We do not
pay our directors any fees or other compensation for acting as directors. We
have not paid any fees or other compensation to any of our directors for acting
as directors to date.
Employment
Contracts
We
presently do not have any employment agreements or other compensation
arrangements with Mr. Qu.
62
CERTAIN
RELATIONSHIPS AND RELATED TRANSACTIONS
Reorganization
Related Transactions
Chance
High owns 100% of the issued and outstanding capital stock of WFOE, a wholly
foreign owned enterprise incorporated under the laws of the PRC. On
December 7, 2009, WFOE entered into the VIE Agreements with Bohai, a company
incorporated under the laws of the PRC, and its three shareholders which include
Mr. Qu (our President and Chief Executive Officer, who owns 90% of Bohai’s
shares) and two unaffiliated parties. Pursuant to the VIE Agreements,
WFOE does not directly own the equity of our operating subsidiary, but rather
assumed management of the business activities of Bohai and has the right to
appoint all executives and senior management and the members of the board of
directors of Bohai. The VIE Agreements are comprised of a series of
agreements, including a Consulting Services Agreement, Operating Agreement,
Proxy Agreement, Equity Pledge Agreement, and Option Agreement, through which
WFOE has the right to advise, consult, manage and operate Bohai for an annual
fee in the amount of Bohai’s yearly net profits after
tax. Additionally, Bohai’s shareholders have pledged their rights,
titles and equity interest in Bohai as security for WFOE to collect consulting
and services fees provided to Bohai through an Equity Pledge Agreement. In order
to further reinforce WFOE’s rights to control and operate Bohai, Bohai’s
shareholders have granted WFOE the exclusive right and option to acquire all of
their equity interests in Bohai through an Option Agreement.
Through
WFOE, Chance High operates and controls Bohai through the VIE Agreements. WFOE
used the contractual arrangements to acquire control of Bohai, instead of using
a complete acquisition of Bohai’s assets or equity to make Bohai a wholly-owned
subsidiary of WFOE because: (i) PRC laws governing share exchanges with foreign
entities, which became effective on September 8, 2006, make the consequences of
such acquisitions uncertain and (ii) other than by share exchange transactions,
PRC laws require Bohai to be acquired for cash and WFOE was not able to raise
sufficient funds to pay the full appraised value for Bohai’s assets or shares as
required under PRC laws.
Slow Walk Arrangements
On
December 7, 2009, Mr. Qu, who is a PRC citizen, entered into the Call Option
Agreement with Joshua Tan, a Singapore passport holder and the sole shareholder
of Glory Period. Under the Call Option Agreement, Mr. Qu shall have
right and option to acquire up to 100% shares of Glory Period for nominal
consideration within the next 3 years. The Call Option Agreement also
provides that Mr. Tan shall not dispose any of the shares of Glory Period
without Mr. Qu’s consent.
Guarantee for the loans with banks by
Mr. Qu
Mr. Qu,
our President, Chief Executive Officer and Chairman, is providing a guaranty for
Bohai’s loans with Pudong Development Bank Qingdao Branch in a total amount of
$2.2 million, or RMB 15 million.
Loans
to Mr. Qu
As of
September 30, 2009, Bohai extended a loan of $1,465,000 to Mr.
Qu. The loan was unsecured, interest bearing at 3.93% per annum and
has no fixed term of repayment. The loan was repaid on December 10,
2009.
63
Other
Other
than employment and the foregoing arrangements, none of the following persons
has any direct or indirect material interest in any transaction to which we are
a party since our incorporation or in any proposed transaction to which we are
proposed to be a party: (i) any of Bohai’s directors or officers; (ii) any
person who beneficially owns, directly or indirectly, shares carrying more than
10% of the voting rights attached to our common stock; or any relative or spouse
of any of the foregoing persons, or any relative of such spouse, who has the
same house as such person or who is a director or officer of any parent or
subsidiary of our company.
64
SECURITY
OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The
following table sets forth certain information regarding our common stock
beneficially owned as of the date of this prospectus for: (i) each stockholder
known to be the beneficial owner of 5% or more of our outstanding common stock,
(ii) each executive officer and director, and (iii) all executive officers and
directors as a group, on a pro forma basis, assuming none of the Notes have been
converted into common stock and none of the Warrants and Agent Warrants were
exercised as of such date. The information contained in the following
table is provided for disclosure purposes only as there can be no assurance that
the actual ownership will be as set forth therein based on the assumptions
used.
|
Name of Beneficial Owner
|
Shares of Common
Stock Owned
|
Percent of Class
Beneficially Owned (1)
|
||||||
|
Glory
Period Limited (2)(3)(4)
|
8,942,471 | 54.19 | % | |||||
|
Hongwei
Qu (4)
|
8,942,471 | 54.19 | % | |||||
|
Gene
Hsiao (5)
|
— | — | ||||||
|
All
Executive Officers and Directors as a group
|
8,942,471 | 54.19 | % | |||||
|
(1)
|
Based
on 16,500,000 shares of common stock issued and outstanding as of the date
of this prospectus.
|
|
(2)
|
Joshua
Tan is the sole shareholder of Glory Period, but pursuant to a Call Option
Agreement, he has no right to sell any shares without prior written
consent by Hongwei Qu.
|
|
(3)
|
Hongwei
Qu is the executive director of Glory
Period.
|
|
(4)
|
On
December 7, 2009, Mr. Qu, who is a PRC citizen, entered into the Call
Option Agreement with Mr. Tan, a Singapore passport holder and the sole
shareholder of Glory Period. Under the Call Option Agreement,
Mr. Qu shall have right and option to acquire up to 100% shares of Glory
Period for nominal consideration within the next 3 years. The
Call Option Agreement also provides that Mr. Tan shall not dispose any of
the shares of Glory Period without Mr. Qu’s
consent.
|
|
(5)
|
Mr.
Hsiao is our Chief Financial Officer. Pursuant to his
employment agreement with us, Mr. Hsiao is entitled to be granted up to an
aggregate of 120,000 shares of our common stock, vesting in three annual
installments of 40,000 beginning June 4, 2011, provided he is then
employed by our company.
|
65
DESCRIPTION
OF SECURITIES
Our
common stock was registered under the Securities Exchange Act of 1934, as
amended, pursuant to a Form 8-A (File Number 000-53401) that was filed with the
SEC on September 4, 2008.
Our
authorized capital consists of 150,000,000 shares of common stock and 10,000,000
shares of preferred stock, par value $01 per share. There are no
outstanding shares of preferred stock as of the date of this
prospectus. A majority of our then stockholders approved an amendment
to our Articles of Incorporation to increase our authorized shares of common
stock from 75,000,000 to 150,000,000, which amendment was filed with the
Secretary of State of Nevada on December 17, 2009.
As of the
date of this prospectus, we have 16,500,000 shares of common stock issued and
outstanding.
Description of Common
Stock. Our common stock is entitled to one vote per share on
all matters submitted to a vote of the stockholders, including the election of
directors. Except as otherwise required by law, the holders of common
stock will possess all voting power. Generally, all matters to be
voted on by stockholders must be approved by a majority (or, in the case of
election of directors, by a plurality) of the votes entitled to be cast by all
shares of common stock that are present in person or represented by
proxy. Holders of our common stock representing a majority of the
voting power of our capital stock issued, outstanding and entitled to vote,
represented in person or by proxy, are necessary to constitute a quorum at any
meeting of stockholders. Our Articles of Incorporation, as amended,
do not provide for cumulative voting in the election of
directors. Holders of common stock have no pre-emptive rights, no
conversion rights and there are no redemption provisions applicable to the
common stock.
Holders
of our common stock are entitled to share in all dividends that the board of
directors, in its discretion, declares from legally available funds. In the
event of a liquidation, dissolution or winding up, each outstanding share
entitles its holder to participate pro rata in all assets that remain after
payment of liabilities and after providing for each class of stock, if any,
having preference over the common stock.
Notes Issued in Connection with the
Private Placement. In connection with our January 2010 private
placement, we offered and sold $12,000,000 worth of Notes convertible into
6,000,000 shares of common stock (plus additional shares which may be issued
upon conversion of interest underlying the Notes).
The Notes
are unsecured, payable on January 5, 2012 and carry an interest rate of 8% per
annum payable quarterly in arrears. We have placed in escrow with the
Escrow Agent an amount of the proceeds of the private placement equal to one
quarter worth of interest payments on the Notes to secure prompt interest
payments, or $240,000. Until such time as 75% of the Notes are
converted into shares of common stock, if such escrow is depleted in order to
make interest payments, we will replenish such escrow amount.
At the
option of each holder, the Notes may be converted into common stock at a price
of $2.00 per share, which conversion price is subject to customary weighted
average and stock based anti-dilution protection.
66
The Notes
contains standard events of default, including: (i) failure to file the
Registration Statement with the SEC within the prescribed period; (ii) failure
to have the Registration Statement deemed effective by the SEC within the
prescribed period; (iii) failure to maintain the effectiveness of the
Registration Statement thereafter; (iv) nonpayment of principal or interest; (v)
termination of registration or suspension of reporting obligations under the
Securities Exchange Act of 1934, as amended, suspension from trading on the
OTCBB (or an exchange), or failure to file reports with the SEC on a timely
basis as required by the Securities Exchange Act of 1934, as amended; (vi)
material breach of representations, warranties and other obligations under the
transaction documents associated with the Securities Purchase Agreement; (vii)
enforcement proceedings; (viii) cross default and cross acceleration; (ix)
insolvency, winding up and other market standard analogous events; (x)
moratorium and nationalization; (xi) proceedings against us or our consolidated
subsidiaries with potential loss/damage of U.S.$5 million or more; and (xii)
illegality of Notes under any applicable law.
The Notes
also contain customary affirmative and negative covenants, including negative
covenants which restrict our ability to do the following without the consent of
Euro Pacific, as representative of the Investors: (i) incur, or permit to exist,
any indebtedness for borrowed money in excess of (A) US$10,000,000
during the twelve (12) month period beginning on January 5, 2010, or (B)
US$15,000,000 during period beginning on January 5, 2010 and ending on January
5, 2012 (the maturity date of the Notes), except in the ordinary course of our
business; (ii) lend or advance money, credit or property to or invest in (by
capital contribution, loan, purchase or otherwise) any person or entity in
excess of US$2,000,000 except: (A) investments
in United States Government obligations, certificates of deposit of any banking
institution with combined capital and surplus of at least $200,000,000; (B)
accounts receivable arising out of sales in the ordinary course of business; and
(C) inter-company loans between and among us and our subsidiaries; (iii) pay
dividends or make any other distribution on shares of our capital stock; (iv)
create, assume or permit to exist, any lien on any of our property or assets now
owned or hereafter acquired, subject to existing liens and certain exceptions;
(v) assume guarantees, subject to certain exceptions; (vi) engage in
“sale-leaseback” transactions, subject to certain exceptions; (vii) make capital
expenditures in excess of US$5,000,000 in any fiscal year, subject to certain
exceptions; and (viii) materially alter our business.
Warrants. We
issued Warrants to purchase 6,000,000 shares of common stock in conjunction with
the private placement. Each Warrant entitles the holder to purchase
one share of common stock. The Warrants shall be exercisable in whole
or in part, at an initial exercise price per share of $2.40, which exercise
price is subject to customary weighted average and stock based anti-dilution
protection. The Warrants may be exercised at any time upon the
election of the holder, beginning on the date of issuance and ending of the
third anniversary of the closing of the private placement, or January 5,
2013. The Warrants are not redeemable.
In the
event of our liquidation, dissolution or winding up, the holders of Warrants
will not be entitled to participate in the distribution of our
assets. In addition, holders of Warrants do not have voting,
pre-emptive, subscription or other rights of stockholders in respect of the
Warrants, nor shall such holders be entitled to receive dividends.
Placement Agent
Warrants. In connection with the private placement, on January
5, 2010, we issued to affiliates of Euro Pacific and to Chardan Capital Markets,
LLC (“Chardan”) three-year Placement Agent Warrants to purchase 600,000 shares
of common stock at an exercise price of $2.40 per share. The
Placement Agent Warrants are substantially identical to the Warrants issued to
the Investors in the Private Placement, except that such warrants may not be
exercised until the six (6) month anniversary of the later of: (i) the date of
effectiveness of the registration statement of which this prospectus is a part
or (ii) the date of commencement of sales in connection with this offering
pursuant to separate lock-up agreements. The holders of the Placement
Agent Warrants are selling stockholders hereunder.
Transfer
Agent and Registrar
The
transfer agent for our common stock is Island Stock Transfer. Our
transfer agent’s address is 100 2nd Avenue South, Suite 705 S, St.Petersburg,
FL, 33701.
67
SELLING
STOCKHOLDERS
We are
registering for the resale shares of our common stock held by the selling
stockholders identified below. We are registering the shares to
permit the selling stockholders and their pledges, donees, transferees and other
successors-in-interest that receive their shares from a selling stockholder as a
gift, partnership distribution or other non-sale related transfer after the date
of this prospectus to resell the shares when and as they deem
appropriate.
The
following table presents information as of the date of this prospectus and sets
forth:
|
|
●
|
the
name of the selling stockholders;
|
|
|
●
|
the
number of shares of our common stock that may be offered for resale for
the account of the selling stockholder under this
prospectus;
|
|
|
●
|
the
number and percentage of shares of our common stock that the selling
stockholder beneficially owned prior to the offering for resale of the
shares under this prospectus; and
|
|
|
●
|
the
number and percentage of shares of our common stock to be beneficially
owned by the selling stockholder after the offering of the resale shares
(assuming all of the offered resale shares are sold by the selling
stockholders).
|
The
20,322,529 shares of our common stock registered for public resale pursuant to
this prospectus and listed under the column “Shares of Common Stock Included in
Prospectus” on the table set forth below consist of:
|
|
(i)
|
7,032,529
shares that were: (i) issued on January 5, 2010 in connection with the
Share Exchange or (ii) held by holders of “restricted” shares under
applicable law, in each case as described in the footnotes to the table
below;
|
|
|
(ii)
|
Up
to 12,690,000 shares issuable upon conversion or exercise of Notes
(including principal and, potentially, interest) and Warrants that were
issued in our January 2010 private placement; and
|
|
|
(iii)
|
600,000
shares issuable upon exercise of the Placement Agent
Warrants.
|
Euro
Pacific Capital, Inc. (“Euro Pacific”) and Chardan Capital Markets, LLC
(“Chardan”) were engaged by us to provide placement agent services in connection
with our Janaury 2010 private placement. In exchange for providing
these services, Euro Pacific and Chardan received an aggregate $1,200,000 in
cash commissions and an aggregate of three-year Placement Agent Warrants to
purchase 600,000 shares of common stock at exercise price of $2.40 per share, of
which 31,500 placement agent warrants were issued to Chardan and 568,500
placement agent warrants were issued to affiliates of Euro
Pacific. The Placement Agent Warrants are substantially identical to
the Warrants issued to the Investors in the private placement, except that such
warrants may not be exercised until the six (6) month anniversary of the later
of: (i) the date of effectiveness of the registration statement of which this
prospectus is a part or (ii) the date of commencement of sales in connection
with this offering pursuant to separate lock-up agreements. Euro
Pacific and Chardan received these securities to be resold in the ordinary
course of business and at the time of the issuance of the securities to be
resold, had no agreements or understandings, directly or indirectly, with any
person to distribute the securities.
68
The below
table assumes that all of the currently outstanding Warrants and Notes will be
exercised and converted for common stock and all of the securities will be sold
in this offering. However, any or all of the securities listed below
may be retained by any of the selling stockholders, and therefore, no accurate
forecast can be made as to the number of securities that will be held by the
selling stockholders upon termination of this offering. The selling
stockholders are not making any representation that any shares covered by this
prospectus will be offered for sale. We believe that, based on information
provided to us by each of the selling stockholders, the selling stockholders
listed in the table have sole voting and investment powers with respect to the
securities indicated. As indicated below, certain selling
stockholders are broker-dealers or affiliates of broker-dealers.
|
Name of Selling Stockholders
|
Shares of
Common
Stock
Included In
Prospectus
|
Shares
Beneficially
Owned Prior
To Offering (1)
|
Percentage
of Shares
Before
Offering (1)
|
Shares
Beneficially
Owned After
Offering (2)
|
Percentage of
Shares After
Offering (2)
|
|||||||||||||||
|
IRA
FBO Robert Stephen Adams Pershing LLC As Custodian (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Selwyn
Adelson (3)
|
60,000 | 60,000 | * | 0 | 0 | % | ||||||||||||||
|
Syed
Hasnat Ahmed & Mirian F Ahmed JT TEN (3)
|
110,000 | 110,000 | * | 0 | 0 | % | ||||||||||||||
|
Am-Per
Enterprises Inc. (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
David
Arita Tod Dtd 05/10/2009 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
William
C Arthur By Pass Trust Dated 10/18/1990 UAD 10/18/90 (3)
|
43,200 | 43,200 | * | 0 | 0 | % | ||||||||||||||
|
James
V. Bacon Trust Dtd 09/14/1995 UAD 03/26/09 (3)
|
250,000 | 250,000 | 1.52 | % | 0 | 0 | % | |||||||||||||
|
IRA
FBO Jeffrey P Baker Pershing LLC as Custodian (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Michael
Baldwin (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Balfour
Hollow LLC (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
The
Sarah J. Basler Living Trust UAD 07/02/98 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Richard
E Benamy (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Middlesex
Ortho Surgeons 401k FBO Lawrence Berson (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Jeff
Blackburn (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Ronald
Bovasso & Linda Bovasso JT TEN (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Bradley
Anesthesiology PC Proft Sharing Plan & Tst (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Ten
Brink Trust Dated 10/02/1986 UAD 10/02/86 (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
White
Pine Productions Defined Benefit Pension Plan (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
SEP
FBO James Brown, Pershing LLC as Custodian (3)
|
90,000 | 90,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Pat Browne Pershing LLC as Custodian (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Scott
Burns (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Robert
Carlson & Michelle Carlson JT TEN (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Brad
K Carr & Roxane Carr JT TEN (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
69
|
Lowell
Cerise (3)
|
75,000 | 75,000 | * | 0 | 0 | % | ||||||||||||||
|
SRC
Corporation Defined Benefit Pension Plan (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Donald
T Clemetson (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Roland
Cram Tod Dtd 06/03/2009 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Integrity
Funds LP (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
The
2000 Jorge &Elena Echeverria Family Trust UAD 11/09/00
(3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Hy Echt Pershing LLC As Custodian (3)
|
60,000 | 60,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Ralph Dale Edson Pershing LLC as Custodian (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Jonathan
Edwards & Virginia C Adams JT TEN (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
The
Arthur Eklund & Janet Eklund 1998 Inter Vivos Trust
(3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Steven
Jay Epstein (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Donald Fagen Pershing LLC as Custodian (3)
|
250,000 | 250,000 | 1.52 | % | 0 | 0 | % | |||||||||||||
|
Sep
FBO Vic Ferrer Pershing LLC as Custodian (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Walter
Friesen (3)
|
150,000 | 150,000 | * | 0 | 0 | % | ||||||||||||||
|
The
Alexander Galuz and Yana Galuz Jt Living Tst UAD 08/24/05
(3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Andrew
Garnock (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Richard
Glaser MDDBPP and Trust (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
The
Goldschlager Family Trust UAD 06/24/04 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Richard
Griff & Jackie Griff JT TEN (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Richard
D Helppie Jr Trust UAD 04/02/92 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Howard
J Hickingbotham Jr & Sandra B Hickingbotham JT TEN(3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
The
Robert K Heimann Living Trust UAD 07/24/01 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Hoke
Living Trust UAD 04/19/02 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Ulrich
Honighausen & Amanda Honighausen JT TEN (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Herschel
Hunter Trust UAD 11/30/88 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Bert Huntsinger Pershing LLC as Custodian (3)
|
199,000 | 199,000 | 1.2 | % | 0 | 0 | % | |||||||||||||
|
Ingram
Living Trust Dated 11/02/2005 UAD 11/02/05 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Ajay
Kalra (3)
|
80,000 | 80,000 | * | 0 | 0 | % | ||||||||||||||
|
Karges
Revocable Intervivos Trust UAD 04/29/85 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Jon Murray Karkow Pershing LLC as Custodian (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Patrick
Kirk & Gloria Kirk JTWROS (3)
|
50,000 | 50,000 | * | 0 | 0 | % |
70
|
Darrel
Lee Kloeckner (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Norman
S Kramer & Linda L Kramer JT TEN (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Thomas A Ladner Pershing LLC as Custodian (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Scott
and Lori Langmack Family Trust UAD 06/22/02 (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Sep
FBO Carter Laren Pershing LLC as Custodian (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
David
W Larson & Jennifer L Larson JT TEN (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Scott
R. Lennes IRA LLC (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Gregg Linhoff Pershing LLC as Custodian (3)
|
110,000 | 110,000 | * | 0 | 0 | % | ||||||||||||||
|
Sep
FBO George Madaraz Pershing LLC as Custodian (3)
|
70,000 | 70,000 | * | 0 | 0 | % | ||||||||||||||
|
Sep
FBO Gerald E Manwill Pershing LLC as Custodian (3)
|
75,000 | 75,000 | * | 0 | 0 | % | ||||||||||||||
|
David
Marble (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Northern
Star Growth Trust Dtd 10/20/1998 UAD 10/20/98 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Mitchell
Martin & Deborah Martin JT TEN (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Brent
May PS Plan FBO F Brent May, Francis Brent TTWW; Dr. May
DMD
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Stephen
P Mccarron PSP-Pershing LLC as Custodian (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Joseph McCarthy Pershing LLC as Custodian (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Rod
Mcintyre Trust UA Dated 5/1/01 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Jerry Mcwilliams Pershing LLC as Custodian (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
The
Meister Non-Exempt Marital Trust UAD 11/17/83 (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Carlos
Alfonso Merino Rev Living Trust UAD 12/04/96 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Mark Mitchell Pershing LLC as Custodian (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Mark
R Mitchell M.D., a Medical Corporation (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
MMH
Group, LLC (3)
|
95,800 | 95,800 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Gerald Mona Pershing LLC as Custodian (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Kevin
Moore (3)
|
75,000 | 75,000 | * | 0 | 0 | % | ||||||||||||||
|
Deepak
Munjal (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Kenneth Henry Nass Pershing LLC as Custodian (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Tim Nass Pershing LLC as Custodian (3)
|
62,000 | 62,000 | * | 0 | 0 | % | ||||||||||||||
|
Mary
Neiberg (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Kevin
P O'neill & Suzanne Odell Oneill JT TEN (3)
|
50,000 | 50,000 | * | 0 | 0 | % |
71
|
Joseph
A & Pamela M Panella Living Trust 1 UAD 05/11/04 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Brent
Paulger and Sharissa Paulger JT TEN (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Tina
C Peterson & Hendrikus M Schraven JT TEN (3)
|
200,000 | 200,000 | * | 0 | 0 | % | ||||||||||||||
|
E.A.
Pickering Painting Inc. (3)
|
60,000 | 60,000 | * | 0 | 0 | % | ||||||||||||||
|
POM
Investments LLC (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Bruce
Walker Ravenel III (3)
|
250,000 | 250,000 | 1.52 | % | 0 | 0 | % | |||||||||||||
|
M.
Carl Rice Self Employed Retirement Plan #1 (3)
|
250,000 | 250,000 | 1.52 | % | 0 | 0 | % | |||||||||||||
|
John
Russell Riedmueller & Nicole Cameron Riedmueller Ten Com
(3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
John
A Rupp Trust UAD 03/25/94 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Steven
V Sann Tod Dtd 10/16/2009 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Robert
C Sayson & Alice K Sayson JT TEN (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Peter
Schortmann and Susan Schortmann JT TEN (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Kimberly
S Schwenke (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Christianna
Seidel Separate Property Trust UAD 11/05/99 (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
James
A Sheahan & Melody K Sheahan JT TEN (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Donald Francis Shoff Pershing LLC as Custodian (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
John
D Smead (3)
|
75,000 | 75,000 | * | 0 | 0 | % | ||||||||||||||
|
Spielman
and Elkin Revocable Trust UAD 06/14/19(3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Diane Spolum Pershing LLC as Custodian (3)
|
250,000 | 250,000 | 1.52 | % | 0 | 0 | % | |||||||||||||
|
Spongbob
Ventures II LLC (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Amy
J Stefanik Revocable Trust UAD 02/06/01 (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Lynn Rollins Stull Pershing LLC as Custodian (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Charles Sullivan Pershing LLC as Custodian (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Gerard Surerus Pershing LLC as Custodian (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO James A Tamborello Pershing LLC as Custodian (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Abdolhosayn
Taslimi and Shidan Taslimi JT TEN (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Mehran
M Taslimi (3)
|
500,000 | 500,000 | * | 0 | 0 | % | ||||||||||||||
|
Ruha
Taslimi and Shidan Taslimi JT TEN (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Shidan
Taslimi (3)
|
500,000 | 500,000 | 3.03 | % | 0 | 0 | % | |||||||||||||
|
Susanne
A Taslimi (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Trillion
Growth China LP (3)
|
250,000 | 250,000 | 1.54 | % | 0 | 0 | % | |||||||||||||
|
Robert
Vecchione (3)
|
50,000 | 50,000 | * | 0 | 0 | % |
72
|
IRA
FBO Matthew A Walton Pershing LLC as Custodian (3)
|
110,000 | 110,000 | * | 0 | 0 | % | ||||||||||||||
|
Timothy
M Weaver (3)
|
250,000 | 250,000 | 1.52 | % | 0 | 0 | % | |||||||||||||
|
Gramercy
87 LLC (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Dipaolo
Worthington Family Trust Dtd 1/31/2008 UAD 01/31/08 (3)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Lambert
Wu & Liying Chu JT TEN (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Wymond
Investments, LLC (3)
|
500,000 | 500,000 | 3.03 | % | 0 | 0 | % | |||||||||||||
|
Layne
Yoshida (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
IRA
FBO Paul Harper Zink Pershing LLC as Custodian (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Jayhawk
Private Equity II, L.P. (3)
|
500,000 | 500,000 | 3.03 | % | 0 | 0 | % | |||||||||||||
|
North
Military Ltd. (3)
|
200,000 | 200,000 | 1.2 | % | 0 | 0 | % | |||||||||||||
|
Chadds
Ford Ltd. (3)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Chardan
SPAC Asset Management (3)
|
150,000 | 150,000 | * | 0 | 0 | % | ||||||||||||||
|
Cawston
Enterprises, Ltd. (4)
|
812,500 | 812,500 | 4.92 | % | 0 | 0 | % | |||||||||||||
|
Regeneration
Capital Group, LLC (5)
|
491,875 | 491,875 | 2.98 | % | 0 | 0 | % | |||||||||||||
|
Perry
A Lerner (5)
|
87,500 | 87,500 | * | 0 | 0 | % | ||||||||||||||
|
SAI
LLC (5)
|
87,500 | 87,500 | * | 0 | 0 | % | ||||||||||||||
|
Emanuel
Addington Living Trust (5)
|
62,500 | 62,500 | * | 0 | 0 | % | ||||||||||||||
|
Joseph
Douek (5)
|
87,500 | 87,500 | * | 0 | 0 | % | ||||||||||||||
|
IRA
For The Benefit of Barry Fox (5)
|
87,500 | 87,500 | * | 0 | 0 | % | ||||||||||||||
|
La
Mancha Capital, LLC (5)
|
95,000 | 95,000 | * | 0 | 0 | % | ||||||||||||||
|
Altbachco,
LLC (5)
|
106,375 | 106,375 | * | 0 | 0 | % | ||||||||||||||
|
Richard
Kaufman (5)
|
87,000 | 87,000 | * | 0 | 0 | % | ||||||||||||||
|
Trilogy
Capital Partners, Inc. (5)
|
31,250 | 31,250 | * | 0 | 0 | % | ||||||||||||||
|
JHVM
Investments LLC (5)
|
25,000 | 25,000 | * | 0 | 0 | % | ||||||||||||||
|
Genesis
Capital Advisors, LLC (5)
|
125,000 | 125,000 | * | 0 | 0 | % | ||||||||||||||
|
Michael
Rosenblum (5)
|
31,250 | 31,250 | * | 0 | 0 | % | ||||||||||||||
|
Norman
Kunin (5)
|
15,625 | 15,625 | * | 0 | 0 | % | ||||||||||||||
|
Dene
LLC (5)
|
31,250 | 31,250 | * | 0 | 0 | % | ||||||||||||||
|
Michael
Levitt (5)
|
9,375 | 9,375 | * | 0 | 0 | % | ||||||||||||||
|
Barry
Cervantes (6)
|
12,500 | 12,500 | * | 0 | 0 | % | ||||||||||||||
|
Steven
Straub (6)
|
12,500 | 12,500 | * | 0 | 0 | % | ||||||||||||||
|
SODA
Ventures LLC (6)
|
100,000 | 100,000 | * | 0 | 0 | % | ||||||||||||||
|
Regeneration
Capital Group, LLC (6)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
Gerald
Altbach (7)
|
152,500 | 152,500 | * | 0 | 0 | % |
73
|
Wang
Shulian (8)
|
650,228 | 650,228 | 3.94 | % | 0 | 0 | % | |||||||||||||
|
Liu
Shaocui (8)
|
487,013 | 487,013 | 2.95 | % | 0 | 0 | % | |||||||||||||
|
Jia
Peicai (8)
|
161,899 | 161.899 | * | 0 | 0 | % | ||||||||||||||
|
Zhang
Jingxin (8)
|
161,899 | 161,899 | * | 0 | 0 | % | ||||||||||||||
|
Liang
Jianxin (8)
|
161,899 | 161,899 | * | 0 | 0 | % | ||||||||||||||
|
Yu
Bohai (8)
|
325,114 | 325,114 | 1.97 | % | 0 | 0 | % | |||||||||||||
|
An
Zhongnan (8)
|
161,899 | 161,899 | * | 0 | 0 | % | ||||||||||||||
|
Xu
Xiaosheng (8)
|
161,899 | 161,899 | * | 0 | 0 | % | ||||||||||||||
|
Dong
Qin (8)
|
81,213 | 81,213 | * | 0 | 0 | % | ||||||||||||||
|
Li
Yanzhi (8)
|
406,195 | 406,195 | 2.46 | % | 0 | 0 | % | |||||||||||||
|
Fu
Wei (8)
|
487,013 | 487,013 | 2.95 | % | 0 | 0 | % | |||||||||||||
|
Wang
Zhizhen (8)
|
487,013 | 487,013 | 2.95 | % | 0 | 0 | % | |||||||||||||
|
Portswealth
Holdings Ltd. (8)
|
486,745 | 486,745 | 2.95 | % | 0 | 0 | % | |||||||||||||
|
Thomas
Z. Tan (9)
|
150,000 | 150,000 | * | 0 | 0 | % | ||||||||||||||
|
Peter
D. Schiff (9)
|
209,250 | 209,250 | 1.27 | % | 0 | 0 | % | |||||||||||||
|
William
G. McBean (9)
|
209,250 | 209,250 | 1.27 | % | 0 | 0 | % | |||||||||||||
|
Chardan
Capital Markets, LLC (10)
|
31,500 | 31,500 | * | 0 | 0 | % | ||||||||||||||
|
Lawrence
D and Christine I Isen Family
Trust (11)
|
50,000 | 50,000 | * | 0 | 0 | % | ||||||||||||||
|
TGR
Group, LLC (11)
|
30,000 | 30,000 | * | 0 | 0 | % | ||||||||||||||
|
Robert
Gleckman (11)
|
10,000 | 10,000 | * | 0 | 0 | % | ||||||||||||||
|
David
Bahr (11)
|
20,000 | 20,000 | * | 0 | 0 | % | ||||||||||||||
|
David
S Nagelberg Revocable Trust July 2, 2003 (11)
|
42,500 | 42,500 | * | 0 | 0 | % | ||||||||||||||
|
Core
Energy Enterprises, Inc. (11)
|
42,500 | 42,500 | * | 0 | 0 | % | ||||||||||||||
|
Richard
Cohen (11)
|
8,000 | 8,000 | * | 0 | 0 | % | ||||||||||||||
|
Marc
Levy (11)
|
8,000 | 8,000 | * | 0 | 0 | % |
*
Represents less than 1% of total outstanding
common stock.
(1) Beneficial
ownership is determined in accordance with Rule 13d-3 of the Exchange Act. In
computing the number of shares beneficially owned by a person and the percentage
ownership of that person, securities that are currently convertible or
exercisable into shares of our common stock, or convertible or exercisable into
shares of our common stock within 60 days of the date hereof are deemed
outstanding. Such shares, however, are not deemed outstanding for the purpose of
computing the percentage ownership of any other person. Except as indicated in
the footnotes to the following table, each stockholder named in the table has
sole voting and investment power with respect to the shares set forth opposite
such stockholder’s name. The percentage of beneficial ownership is based on
16,500,000 shares of common stock outstanding as of the date of this
prospectus.
(2) Assumes
that all securities registered will be sold.
74
(3) We
issued an aggregate of 6,000,000 of units for gross proceeds of $12 million on
January 5, 2010. Each unit consisted of an unsecured $2.00 principal
amount Notes payable 24 months from January 5, 2010 with an interest rate of 8%
per annum payable quarterly in arrears, and a three year Warrant to purchase one
share of our common stock. The amount of shares referenced for the
selling stockholder includes: (i) shares of common stock beneficially owned by
the selling stockholder or shares which are issuable upon conversion of the Note
held by the selling stockholder at an exercise price at $2.00 per share, which
shares represent 50% of the registrable securities held by the selling
stockholder; (ii) up to a maximum aggregate of 690,000 shares of common stock
(which are not reflected in the table about due to the contingent nature of the
issuance thereof) representing conversion of interest underlying the Notes (with
each selling stockholder eligible to receive a portion of such shares depending
on when they convert their Notes) and (iii) shares of common stock beneficially
owned by the selling stockholder which are issuable upon exercise of the Warrant
at an exercise price at $2.40 per share, which shares represent 50% of the
registrable securities held by the selling stockholder.
(4) Cawston
Enterprises Ltd. acted as PRC financial advisor to Bohai in connection with the
Share Exchange and concurrent private placement transaction, for which Cawston
received shares of our common stock as compensation.
(5) The
shares held by the selling stockholder are part of an aggregate of 1,637,500
shares of common stock issued to Regeneration Capital Group, LLC and its
affiliates (“Regeneration”) or subsequently privately sold by Regeneration (see
Footnote 12). Regeneration and its affiliates: (i) were investors in
our company prior to the consummation of the January 5, 2010 Share Exchange
and/or (ii) acted as U.S. financial advisor to Bohai in connection with the
Share Exchange and concurrent private placement transaction, for which
Regeneration received shares of our common stock as compensation.
(6) The
shares held by the selling stockholder are part of an aggregate 200,000 shares
of common stock issued upon conversion of short term bridge loans totaling
$400,000 which were obtained from four investors in June 2009.
(7) Selling
stockholder acted as a consultant to us prior to our January 5, 2010 share
exchange and private placement.
(8) Selling
stockholder was a minority shareholder of Chance High Limited and received the
registrable securities as part of the Share Exchange transaction which closed on
January 5, 2010.
(9) Selling
stockholder is an affiliate of Euro Pacific, a FINRA member and SEC registered
broker-dealer that acted as the lead placement agent in our January 5, 2010
private placement. Securities represent shares of common stock
underlying the Placement Agent Warrant issued to the selling stockholder.
(10) Chardan
is a FINRA member and SEC registered broker-dealer that acted as co-placement
agent in our January 5, 2010 private placement. Registrable
securities represents shares of common stock underlying the Placement Agent
Warrant issued to the selling stockholder.
(11) The
shares held by the selling stockholder were privately purchased from members of
Regeneration in April and May 2010 in transactions exempt from registration
under the Securities Act of 1933, as amended.
75
PLAN
OF DISTRIBUTION
The
selling stockholders and any of their respective pledgees, donees, assignees and
other successors-in-interest may, from time to time, sell any or all of their
shares of common stock on any stock exchange, market or trading facility on
which the shares are traded or in private transactions. These sales may be at
fixed or negotiated prices. The selling stockholders may use any one or more of
the following methods when selling shares:
|
|
●
|
ordinary
brokerage transactions and transactions in which the broker-dealer
solicits the purchaser;
|
|
|
●
|
block
trades in which the broker-dealer will attempt to sell the shares as agent
but may position and resell a portion of the block as
principal;
|
|
|
●
|
facilitate
the transaction;
|
|
|
●
|
purchases
by a broker-dealer as principal and resale by the broker-dealer for its
account;
|
|
|
●
|
an
exchange distribution in accordance with the rules of the applicable
exchange;
|
|
|
●
|
privately-negotiated
transactions;
|
|
|
●
|
broker-dealers
may agree with the selling stockholders to sell a specified number of such
shares at a stipulated price per
share;
|
|
|
●
|
through
the writing of options on the
shares;
|
|
|
●
|
a
combination of any such methods of sale;
and
|
|
|
●
|
any
other method permitted pursuant to applicable
law.
|
The
selling stockholders may also sell shares under Rule 144 of the Securities Act
of 1933, as amended (the “Securities Act”), if available, rather than under this
prospectus. The selling stockholders shall have the sole and absolute discretion
not to accept any purchase offer or make any sale of shares if it deems the
purchase price to be unsatisfactory at any particular time.
The
selling stockholders or their respective pledgees, donees, transferees or other
successors in interest, may also sell the shares directly to market makers
acting as principals and/or broker-dealers acting as agents for themselves or
their customers. Such broker-dealers may receive compensation in the form of
discounts, concessions or commissions from the selling stockholders and/or the
purchasers of shares for whom such broker-dealers may act as agents or to whom
they sell as principal or both, which compensation as to a particular
broker-dealer might be in excess of customary commissions. Market makers and
block purchasers purchasing the shares will do so for their own account and at
their own risk. It is possible that a selling stockholder will attempt to sell
shares of common stock in block transactions to market makers or other
purchasers at a price per share which may be below the then existing market
price. We cannot assure that all or any of the shares offered in this prospectus
will be issued to, or sold by, the selling stockholders. The selling
stockholders and any brokers, dealers or agents, upon effecting the sale of any
of the shares offered in this prospectus, may be deemed to be “underwriters” as
that term is defined under the Securities Act, the Exchange Act and the rules
and regulations of such acts. In such event, any commissions received by such
broker-dealers or agents and any profit on the resale of the shares purchased by
them may be deemed to be underwriting commissions or discounts under the
Securities Act.
76
We are
required to pay all fees and expenses incident to the registration of the
shares, including fees and disbursements of counsel to the selling stockholders,
but excluding brokerage commissions or underwriter discounts.
The
selling stockholders, alternatively, may sell all or any part of the shares
offered in this prospectus through an underwriter. The selling
stockholders have not entered into any agreement with a prospective underwriter
and there is no assurance that any such agreement will be entered
into.
The
selling stockholders may pledge their shares to their brokers under the margin
provisions of customer agreements. If a selling stockholder defaults on a margin
loan, the broker may, from time to time, offer and sell the pledged shares. The
selling stockholders and any other persons participating in the sale or
distribution of the shares will be subject to applicable provisions of the
Exchange Act, and the rules and regulations under such act, including, without
limitation, Regulation M. These provisions may restrict certain activities of,
and limit the timing of purchases and sales of any of the shares by, the selling
stockholders or any other such person. In the event that any of the selling
stockholders are deemed an affiliated purchaser or distribution participant
within the meaning of Regulation M, then the selling stockholders will not be
permitted to engage in short sales of common stock. Furthermore, under
Regulation M, persons engaged in a distribution of securities are prohibited
from simultaneously engaging in market making and certain other activities with
respect to such securities for a specified period of time prior to the
commencement of such distributions, subject to specified exceptions or
exemptions. In addition, if a short sale is deemed to be a stabilizing activity,
then the selling stockholders will not be permitted to engage in a short sale of
our common stock. All of these limitations may affect the marketability of the
shares.
If a
selling stockholder notifies us that it has a material arrangement with a
broker-dealer for the resale of the common stock, then we would be required to
amend the registration statement of which this prospectus is a part, and file a
prospectus supplement to describe the agreements between the selling stockholder
and the broker-dealer.
In
compliance with the guidelines of the Financial Industry Regulatory Authority,
Inc., or FINRA, the maximum consideration or discount to be received by any
member of the FINRA may not exceed 8% of the aggregate amount of the securities
offered pursuant to this prospectus.
Selling
stockholders hereunder that are FINRA members, or affiliates of FINRA members,
are precluded from, directly or indirectly, offering, selling, agreeing to offer
or sell, transferring, assigning, pledging, hypothecating or subjecting to
hedging, short sale, derivative, put or call transaction, all or any portion of
those certain placement agents’ warrants of the Company issued to such selling
stockholders on January 5, 2010, or any shares of the company’s common stock
thereunder, for the period beginning on the later of: (i) the date of
effectiveness of the registration statement of which this prospectus forms a
part or (ii) the date of commencement of sales pursuant to this prospectus and
ending on the six (6) month anniversary of such date, except in accordance with
FINRA Rule 5110 (g)(2).
77
Euro
Pacific and Chardan are FINRA members and were engaged by us to provide
placement agent services in connection with our January 2010 private placement.
In exchange for providing these services, Euro Pacific and Chardan received an
aggregate $1,200,000 in cash commissions and three-year Placement Agent Warrants
to purchase an aggregate of 600,000 shares of common stock at an exercise price
of $2.40 per share. 31,500 Placement Agent Warrants were issued to Chardan, and
568,500 Placement Agent Warrants were issued to affiliates of Euro Pacific
consisting of Thomas Z. Tan, Peter D. Schiff and William G. McBean, who received
150,000, 209,250 and 209,250 Placement Agent Warrants, respectively. The
Placement Agent Warrants are substantially identical to the Warrants issued to
the Investors in the private placement, except with respect to the lock-up
period described above. Euro Pacific, its affiliates and Chardan received these
securities to be resold in the ordinary course of business and at the time of
the issuance and as of the date of this prospectus, had no agreements or
understandings, directly or indirectly, with any person to distribute the
securities.
Euro
Pacific, Chardan, and Euro Pacific’s affiliates have no role in this offering,
except that Chardan and Messrs. Tan, Schiff and McBean are selling stockholders,
and Euro Pacific and Chardan may engage in ordinary brokerage transactions for
any selling stockholders or act as broker-dealers in the ordinary course of
business with respect to the shares offered hereby, in all such cases for
commissions not to exceed 4.5% in the aggregate.
78
MARKET
FOR COMMON EQUITY AND RELATED STOCKHOLDER MATTERS
Market
Information
Our
common stock is listed for quotation on the OTCBB under the symbol “BOPH.” From
January 9, 2008 until February 8, 2010, our common stock was listed for
quotation on the OTCBB under the symbol “LINK”.
There is very limited trading in our
common stock. On July 23, 2010, the most recent day that our stock
traded, the last reported price per share of our common stock was
$2.25. The absence of meaningful transactions in the common
stock indicates there is no established trading market for our common
stock. You are urged
to obtain current market quotations of our common stock before purchasing any of
the shares being offered for sale pursuant to this prospectus.
Holders
As of
August 11, 2010, there
were 16,500,000 shares of our common stock outstanding held by approximately 47
stockholders of record. The number of our stockholders of record
excludes any estimate by us of the number of beneficial owners of shares held in
street name, the accuracy of which cannot be guaranteed.
Dividend
Policy
We have
not paid any cash dividends on our common stock to date, and we have no
intention of paying cash dividends in the foreseeable future. Whether we will
declare and pay dividends in the future will be determined by our board of
directors at their discretion, subject to certain limitations imposed under
Delaware corporate law. In addition, our ability to pay dividends may be
affected by the foreign exchange controls in China. The timing, amount and form
of dividends, if any, will depend on, among other things, our results of
operations, financial condition, cash requirements and other factors deemed
relevant by our board of directors.
LEGAL
MATTERS
The
validity of our common stock offered hereby has been passed upon by Ellenoff
Grossman & Schole LLP, New York, New York.
EXPERTS
The
consolidated balance sheet of the Company and its subsidiary for the
fiscal years ended June 30, 2008 and 2009, and the related consolidated
statements of operations, changes in stockholders’ equity and cash flows
appearing in this registration statement have been so included in reliance on
the Report of Parker Randall CF (H.K.) CPA Limited, an independent registered
public accounting firm, appearing elsewhere in this prospectus, given on the
authority of such firm as experts in accounting and auditing.
79
CHANGES
IN AND DISAGREEMENTS WITH ACCOUNTANTS
ON
ACCOUNTING AND FINANCIAL DISCLOSURE
Effective
January 29, 2010, upon the approval of our board of directors, we dismissed John
Kinross-Kennedy as our independent registered public accountant.
During
the prior fiscal years ended May 31, 2009 and 2008, John Kinross-Kennedy’s
reports on the financial statements of our company contained no adverse opinion
or disclaimer of opinion and were not qualified or modified as to uncertainty,
audit scope or accounting principle.
During
the fiscal years ended May 31, 2009 and 2008 and subsequent period through
January 29, 2010, there were no disagreements (as defined in Item 304(a)(1)(iv)
of Regulation S-K) between us and John Kinross-Kennedy on any matter of
accounting principles or practices, financial statement disclosure or auditing
scope or procedure, which disagreements, if not resolved to the satisfaction of
John Kinross-Kennedy, would have caused him to make reference thereto in his
report on financial statements for such years.
During
the fiscal years ended May 31, 2009 and 2008 and subsequent period through
January 29, 2010, there were no reportable events as defined in Regulation S-K
Item 304(a)(1)(v).
On
January 29, 2010, upon the approval of our board of directors, Parker Randall CF
(H.K.) CPA Limited (the “Parker Randall”) was appointed as our independent
registered public accounting firm. During our prior fiscal years
ended May 31, 2009 and 2008 and subsequent period through January 31,2010, we
did not consult with Parker Randall regarding any of the matters or events set
forth in Item 304(a)(2)(i) and Item 304(a)(2)(ii) of Regulation
S-K.
WHERE
YOU CAN FIND MORE INFORMATION
This
prospectus is part of a Registration Statement on Form S-1 we have filed with
the SEC. We have not included in this prospectus all of the information
contained in the Registration Statement and you should refer to our Registration
Statement and its exhibits for further information.
We file
annual, quarterly, and special reports, proxy statements, and other information
with the SEC. You may read and copy any document we file at the SEC’s public
reference room at 100 F Street, N.E., Washington, D.C. 20549. Copies
of these materials may also be obtained from the SEC at prescribed rates by
writing to the Public Reference Section of the SEC, 100 F. Street, N.E.,
Washington, D.C. 20549. You may obtain information about the operation of the
SEC public reference room in Washington, D.C. by calling the SEC at
1-800-SEC-0330. Our filings are also available to the public from commercial
document retrieval services and at the website maintained by the SEC at
http://www.sec.gov.
80
BOHAI
PHARMACEUTICALS GROUP, INC.
AND
ITS SUBSIDIARIES
INDEX
TO FINANCIAL STATEMENTS
|
Page
|
||
|
Interim
Financial Statements (unaudited):
|
||
|
Condensed
Consolidated Balance Sheets as of June 30, 2009 (audited) and March 31,
2010 (restated)
|
F-2
|
|
|
Condensed
Consolidated Statements of Income for three months ended March 31, 2009
and 2010, and for the nine months ended March 31, 2009 and 2010
(restated)
|
F-3
|
|
|
Condensed
Consolidated Statements of Cash Flows for three months ended March 31,
2009 and 2010 (restated), and for the nine months ended March 31, 2009 and
2010 (restated)
|
F-4
|
|
|
Notes
to Condensed and Consolidated Financial Statements
|
F-5
|
|
|
Consolidated
Financial Statements:
|
||
|
Report
of Independent Registered Public Accounting Firm
|
F-27
|
|
|
Consolidated
Balance Sheets as of June 30, 2009 and 2008
|
F-28
|
|
|
Consolidated
Statement of Income for the years ended June 2009 and 2008
|
F-29
|
|
|
Consolidated
Statement of Stockholders’ Equity for the years ended June 2009 and
2008
|
F-30
|
|
|
Consolidated
Statement of Cash Flows as of June 30, 2009 and 2008
|
F-31
|
|
|
Notes
to the Consolidated Financial Statements
|
F-32
|
F-1
BOHAI
PHARMACEUTICALS GROUP, INC.
CONDENSED
CONSOLIDATED BALANCE SHEETS
AS
OF MARCH 31, 2010 AND JUNE 30, 2009
|
As of
|
As of
|
|||||||||
|
March 31,
|
June 30,
|
|||||||||
|
Notes
|
2010
|
2009
|
||||||||
|
(Unaudited)
|
(Audited)
|
|||||||||
|
RESTATED
|
||||||||||
|
ASSETS
|
||||||||||
|
Current
assets
|
||||||||||
|
Cash
and cash equivalents
|
$
|
15,574,909
|
$
|
2,493,510
|
||||||
|
Accounts
receivable
|
11,530,595
|
11,096,866
|
||||||||
|
Other
receivables and prepayments
|
4
|
6,969,569
|
12,993,800
|
|||||||
|
Amount
due from equity holder
|
5
|
-
|
1,465,000
|
|||||||
|
Inventories
|
6
|
970,664
|
307,834
|
|||||||
|
Total
current assets
|
35,045,737
|
28,357,010
|
||||||||
|
Non-current
assets
|
||||||||||
|
Property,
plant and equipment, net
|
8
|
7,946,215
|
8,149,279
|
|||||||
|
Intangible
assets
|
7
|
17,273,007
|
17,298,720
|
|||||||
|
Deferred
fees on convertible notes
|
13
|
1,898,877
|
-
|
|||||||
|
Total
non-current assets
|
27,118,099
|
25,447,999
|
||||||||
|
TOTAL
ASSETS
|
$
|
62,163,836
|
$
|
53,805,009
|
||||||
|
LIABILITES
AND STOCKHOLDERS’ EQUITY
|
||||||||||
|
Current
liabilities
|
||||||||||
|
Short-term
borrowings
|
9
|
$
|
4,381,153
|
$
|
5,860,000
|
|||||
|
Notes
payable
|
10
|
-
|
400,000
|
|||||||
|
Accounts
payable
|
842,350
|
971,208
|
||||||||
|
Other
accrued liabilities
|
11
|
1,941,088
|
12,339,355
|
|||||||
|
Income
taxes payable
|
17
|
1,389,733
|
677,666
|
|||||||
|
Total
current liabilities
|
8,554,324
|
20,248,229
|
||||||||
|
Derivative
liabilities – investor warrants
|
12
|
4,839,673
|
-
|
|||||||
|
Derivative
liabilities – agent warrants
|
12
|
483,967
|
-
|
|||||||
|
Convertible
notes, net of amortization (6,000,000 units with 8% interest)
|
12
|
121,127
|
-
|
|||||||
|
TOTAL
LIABILITIES
|
13,999,091
|
20,248,229
|
||||||||
|
STOCKHOLDERS’
EQUITY
|
||||||||||
|
Common
stock, $0.001 par value, 150,000,000 shares authorized, 16,250,000 shares
issued and outstanding as of March 31, 2010, 3,450,000 shares issued and
outstanding as of June 30, 2009
|
19
|
16,250
|
3,450
|
|||||||
|
Additional
paid-in capital
|
19
|
9,553,666
|
2,974,520
|
|||||||
|
Capital
reserve
|
5,836,000
|
5,836,000
|
||||||||
|
Accumulated
other comprehensive income
|
382,108
|
490,931
|
||||||||
|
Statutory
reserves
|
2,974,137
|
2,201,811
|
||||||||
|
Retained
earnings
|
29,402,584
|
22,050,068
|
||||||||
|
Total
stockholders’ equity
|
48,164,745
|
33,556,780
|
||||||||
|
TOTAL
LIABILITIES AND STOCKOLDERS’ EQUITY
|
$
|
62,163,836
|
$
|
53,805,009
|
||||||
See
accompanying notes to the financial statements
F-2
BOHAI
PHARMACEUTICALS GROUP, INC.
CONDENSED
CONSOLIDATED STATEMENT OF INCOME
FOR
THE THREE MONTHS AND NINE MONTHS ENDED MARCH 31, 2010 AND 2009
|
Three months
|
Three months
|
Nine months
|
Nine months
|
|||||||||||||||
|
ended
|
ended
|
ended
|
ended
|
|||||||||||||||
|
March 31,
|
March 31,
|
March 31,
|
March 31,
|
|||||||||||||||
|
Notes
|
2010
|
2009
|
2010
|
2009
|
||||||||||||||
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
|||||||||||||||
|
RESTATED
|
RESTATED
|
|||||||||||||||||
|
Sales
|
$
|
15,323,878
|
$
|
12,476,400
|
$
|
46,072,455
|
$
|
35,833,215
|
||||||||||
|
Less:
Sales Tax
|
(231,870
|
)
|
(200,622
|
)
|
(729,975
|
)
|
(580,207
|
)
|
||||||||||
|
Net
sales
|
15,092,008
|
12,275,778
|
45,342,480
|
35,253,008
|
||||||||||||||
|
Cost
of sales
|
(2,609,515
|
)
|
(2,019,864
|
)
|
(7,475,740
|
)
|
(5,966,876
|
)
|
||||||||||
|
Gross
profit
|
12,482,493
|
10,255,914
|
37,866,740
|
29,286,132
|
||||||||||||||
|
Selling,
general and administrative expenses
|
14
|
(9,202,873
|
)
|
(7,670,130
|
)
|
(28,208,753
|
)
|
(21,675,279
|
)
|
|||||||||
|
Interest
expenses
|
15
|
(381,700
|
)
|
(52,921
|
)
|
(538,008
|
)
|
(168,422
|
)
|
|||||||||
|
Operating
income
|
2,897,920
|
2,532,863
|
9,119,979
|
7,442,431
|
||||||||||||||
|
Other
income
|
-
|
158
|
18,864
|
158
|
||||||||||||||
|
Finance
and non-operating incomes (expenses)
|
16
|
829,773
|
(4,618
|
)
|
807,681
|
(4,682
|
)
|
|||||||||||
|
Income
before taxes
|
3,727,693
|
2,528,403
|
9,946,524
|
7,437,907
|
||||||||||||||
|
Income
taxes
|
17
|
(585,135
|
)
|
(423,569
|
)
|
(2,193,931
|
)
|
(1,224,833
|
)
|
|||||||||
|
Net
income
|
$
|
3,142,558
|
$
|
2,104,834
|
$
|
7,752,593
|
$
|
6,213,074
|
||||||||||
See
accompanying notes to the financial statements
F-3
BOHAI
PHARMACEUTICALS GROUP CO., LTD.
CONDENSED
CONSOLIDATED STATEMENTS OF CASH FLOWS
FOR
THE THREE MONTHS AND NINE MONTHS ENDED MARCH 31, 2010 AND 2009
|
Three
months
|
Three
months
|
Nine
months
|
Nine
months
|
|||||||||||||
|
ended
|
ended
|
ended
|
ended
|
|||||||||||||
|
March 31,
2010
|
March 31,
2009
|
March 31,
2010
|
March 31,
2009
|
|||||||||||||
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
|||||||||||||
|
RESTATED
|
RESTATED
|
|||||||||||||||
|
Cash
flows from operating activities
|
||||||||||||||||
|
Net
income
|
$ | 3,142,558 | $ | 2,104,834 | $ | 7,752,593 | $ | 6,213,074 | ||||||||
|
Adjustments
to reconcile net income to net cash (used in)/ provided by operating
activities
|
||||||||||||||||
|
Depreciation
|
84,518 | 213,912 | 224,656 | 284,387 | ||||||||||||
|
Loss
on disposals of property, plant and equipment
|
- | - | 10,942 | - | ||||||||||||
|
Amortization
of deferred fees on convertible notes
|
253,577 | - | 253,577 | - | ||||||||||||
|
Interest
expense on convertible notes
|
121,126 | - | 121,126 | - | ||||||||||||
|
Change
in fair value of warrants
|
(1,083,350 | ) | - | (1,083,350 | ) | - | ||||||||||
|
Changes
in assets and liabilities
|
||||||||||||||||
|
Decrease/
(increase) in accounts receivable
|
250,769 | (1,383,561 | ) | (433,728 | ) | (1,119,683 | ) | |||||||||
|
Decrease
in other receivables and prepayments
|
6,504,589 | 546,973 | 4,127,297 | 8,311,379 | ||||||||||||
|
Decrease/
(increase) in amount due from equity holder
|
- | - | 1,465,000 | (108,426 | ) | |||||||||||
|
(Increase)/
decrease in inventories
|
(198,160 | ) | 206,841 | (662,830 | ) | 138,503 | ||||||||||
|
Decrease/
(increase) in accounts payable
|
87,292 | (257,958 | ) | (128,858 | ) | (1,923,974 | ) | |||||||||
|
(Decrease)/
increase in accrued liabilities
|
(11,338,820 | ) | 783,952 | (10,407,917 | ) | (12,430,148 | ) | |||||||||
|
(Decrease)/
increase in income taxes payable
|
(273,202 | ) | 288,939 | 2,905,998 | 1,453,411 | |||||||||||
|
Net
cash (used in)/ provided by operating activities
|
(2,449,103 | ) | 2,503,932 | 4,144,506 | 818,523 | |||||||||||
|
Cash
flows from investing activities
|
||||||||||||||||
|
Purchases
of property, plant and equipment
|
(84,518 | ) | (191,977 | ) | (280,804 | ) | (505,153 | ) | ||||||||
|
Proceeds
from disposals of property, plant and equipment
|
- | - | - | - | ||||||||||||
|
Net
cash used in investing activities
|
(84,518 | ) | (191,977 | ) | (280,804 | ) | (505,153 | ) | ||||||||
|
Cash
flows from financing activities
|
||||||||||||||||
|
Cash
fees on placement agent and other financing costs
|
(1,570,000 | ) | - | (1,570,000 | ) | - | ||||||||||
|
Proceeds
of convertible promissory notes
|
12,000,000 | - | 12,000,000 | - | ||||||||||||
|
Proceeds
of borrowings
|
4,381,153 | 1,314,713 | 4,381,153 | 4,382,377 | ||||||||||||
|
Repayment
of borrowings
|
(4,393,665 | ) | (1,896,700 | ) | (5,860,000 | ) | (3,271,413 | ) | ||||||||
|
Net
cash provided by/ (used in) financing activities
|
10,417,488 | (581,987 | ) | 8,951,153 | 1,110,964 | |||||||||||
|
Net
increase in cash and cash equivalents
|
7,883,867 | 1,729,968 | 12,814,855 | 1,424,334 | ||||||||||||
|
Effect
of foreign currency translation on cash and cash equivalents
|
(201,866 | ) | (26,482 | ) | 266,544 | 227,015 | ||||||||||
|
Cash
and cash equivalents at beginning of period
|
7,892,908 | 776,913 | 2,493,510 | 829,050 | ||||||||||||
|
Cash
and cash equivalents at end of period
|
$ | 15,574,909 | $ | 2,480,399 | $ | 15,574,909 | $ | 2,480,399 | ||||||||
|
Supplemental
cash flow information:
|
||||||||||||||||
|
Interest
paid on convertible notes
|
$ | 226,667 | $ | - | $ | 226,667 | $ | - | ||||||||
|
Placement
agent warrants issued
|
$ | 582,454 | $ | - | $ | 582,454 | $ | - | ||||||||
See
accompanying notes to the financial statements
F-4
BOHAI
PHARMACEUTICALS GROUP, INC.
NOTES
TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
FOR
THE THREE MONTHS AND NINE MONTHS ENDED MARCH 31, 2010
(UNAUDITED)
(Stated
in US Dollars)
|
1.
|
ORGANIZATION
AND PRINCIPAL ACTIVITIES
|
Bohai
Pharmaceuticals Group, Inc. (formerly known as Link Resources, Inc.) was
incorporated under the laws of the State of Nevada on January 9, 2008. Until
January 5, 2010, its principal office was located in Calgary, Alberta, Canada.
The Company was a public “shell” company in the exploration stage since its
formation and had not yet realized any revenues from its planned
operations. The Company entered into a Mineral Lease Agreement on
April 1, 2008 for two mining claims in Pershing County, Nevada, in an area known
as the Goldbanks East Prospect. The Company terminated the lease on
July 7, 2009.
Pursuant
to a Share Exchange Agreement, dated January 5, 2010 (the “Share Exchange
Agreement” and the transactions contemplated thereby, the “Share Exchange”), the
Company acquired Chance High International Limited, a British Virgin Islands
company (“Chance High”) from Chance High’s shareholders (the “Chance High
Shareholders”) and, as a result, acquired Chance High’s indirect, controlled
subsidiary, Yantai Bohai Pharmaceuticals Group Co., Ltd. (“Bohai”), a Chinese
company engaged the production, manufacturing and distribution in the People’s
Republic of China (“China” or the “PRC”) of herbal medicines, including capsules
and other products, based on traditional Chinese medicine. The
closing of the Share Exchange (the “Closing”) took place on January 5, 2010 (the
“Closing Date”).
On the
Closing Date, pursuant to the terms of the Share Exchange Agreement, the Company
acquired all of the outstanding equity securities (the “Chance High Shares”) of
Chance High from the Chance High Shareholders, and the Chance High Shareholders
transferred and contributed all of their Chance High Shares to the
Company. In exchange, the Company issued to Chance High Shareholders
an aggregate of 13,162,500 newly issued shares of common stock, par value $0.001
per share (the “Common Stock”). In addition, pursuant to the terms of
the Share Exchange Agreement, Anthony Zaradic, the former President and Chief
Executive Officer of the Company, cancelled a total of 1,500,000 shares of
Common Stock.
Chance
High owns 100% of the issued and outstanding capital stock of the Yantai
Shencaojishi Pharmaceuticals Co., Ltd. (“WFOE”). On December 7, 2009,
the WFOE entered into a series of variable interest entity contractual
agreements (the “VIE Agreements”) with Bohai and its three shareholders,
including Mr. Hongwei Qu, currently the Company’s Chairman, Chief Executive
Officer and President (“Qu”), pursuant to which WFOE effectively assumed
management of the business activities of Bohai and has the right to appoint all
executives and senior management and the members of the board of directors of
Bohai. The Company, Chance High, WFOE and Bohai are referred to
herein collectively as the “Group”.
The VIE
Agreements are comprised of a series of agreements, including a Consulting
Services Agreement, Operating Agreement and Proxy Agreement, through which WFOE
has the right to advise, consult, manage and operate Bohai for an annual fee in
the amount of Bohai’s yearly net profits after tax. Additionally,
Bohai’s shareholders pledged their rights, titles and equity interest in Bohai
as security for WFOE to collect consulting and services fees provided to Bohai
through an Equity Pledge Agreement. In order to further reinforce
WFOE’s rights to control and operate Bohai, Bohai’s shareholders granted WFOE an
exclusive right and option to acquire all of their equity interests in Bohai
through an Option Agreement.
F-5
|
2.
|
BASIS
OF PRESENTATION
|
The Group
maintains its general ledger and journals with the accrual method accounting for
financial reporting purposes. Accounting policies adopted by the Group conform
to generally accepted accounting principles in the United States of America (“US
GAAP”) and have been consistently applied in the presentation of financial
statements, which are compiled on the accrual basis of accounting.
This
basis of accounting differs in certain material respects from that used for the
preparation of the books of account of the Group, which are prepared in
accordance with the accounting principles and the relevant financial regulations
applicable to enterprises with limited liabilities established in the PRC (“PRC
GAAP”), the accounting standards used in the places of their
domicile. The accompanying financial statements reflect necessary
adjustments not recorded in the books of account of the Group to present them in
conformity with US GAAP.
The
interim financial statements as of and for the three months and nine moths ended
March 31, 2010 reflect all adjustments which, in the opinion of management, are
necessary to present fairly the financial position, results of operations and
cash flows for the period presented in accordance with the accounting principles
generally accepted in the United States of America. All adjustments are of a
normal recurring nature.
The Share
Exchange is being accounted for as a reverse recapitalization presumed to be
effected on January 1, 2010 instead of January 5, 2010. The operating
results, financial position and cash flows of the Group are consolidated since
January 2010. The operating results, financial position and cash
flows for Chance High and its subsidiaries for the three and nine month periods
ended March 31 2009, as at June 30, 2009 and for period July 2009 to March 2010
nine-month period are being presented for comparative figures illustration
purpose only.
|
3.
|
SUMMARY
OF SIGNIFICANT ACCOUNTING POLICIES
|
|
(a)
|
Basis
of Presentation and Consolidation
|
Pursuant
to the Share Exchange Agreement, Link Resources, Inc. accounted for the
transaction as reverse recapitalization according to ASC 805-40-25-1 so that the
Share Exchange was considered a reverse recapitalization with Chance High as the
accounting acquirer and Link Resources, Inc. as the accounting
acquiree. As the transaction between Link Resource Inc. and Chance
High Limited is treated as reverse acquisition, no goodwill was
recorded. The historical financial statements of the Company
therefore include those of Chance High and its subsidiaries (WFOE and Bohai)
prior to the consummation of the Share Exchange and those of Link Resources,
Chance High and its subsidiaries after the consummation of the Share
Exchange. Intercompany transactions and balances are eliminated in
consolidation. Pursuant to an Agreement and Plan of Merger that Link
Resources, Inc. entered into on January 29, 2010, Link Resources Inc. changed
its corporate name to “Bohai Pharmaceuticals Group, Inc.” The sole
purpose of such merger was to effect a change of the Company’s corporate name
.
The
Company has adopted FAS ASC 810-10-15-14 and also FIN 46R, which requires that a
Variable Interest Entity (“VIE”) to be consolidated by a company if that company
is entitled to receive a majority of the VIE’s residual returns and have direct
ability to made decision on all operation activities of the voting right of the
VIE.
F-6
The
Operating Agreement indicated that Bohai, as operating company, which is wholly
foreign owned under control of Chance High that empowers to WFOE the direct
ability to made decision on all the operation activities of the voting right of
Bohai.
Under
Consultant Service Agreement entered between WFOE and Bohai on December 7, 2009,
Bohai agreed to pay all of net income to WOFE quarterly as consultant fee.
Accordingly, WOFE has the right to receive the expected residual returns of
Bohai.
Under
above mentioned contractual arrangement, the Company, Bohai Pharmaceuticals
Group, Inc. qualifies as the primary beneficiaries such a controlling financial
interest of Bohai as operating under FASB ASC230-10-45 and FASB Interpretation
No. 46R “Consolidation of Variable Interest Entities” (“FIN 46R”), an
Interpretation of Accounting Research Bulletin No. 51. The result of
subsidiaries or variable interest entities acquired prior to date of Share
Exchange Agreement on January 5, 2010 entered are included in the condensed
consolidated financial statement.
As of
March 31, 2010, the particulars of the Company’s subsidiaries are as
follows:
|
Name of
Company
|
Place of
incorporation
|
Date of
incorporation
|
Attributable
equity
interest
|
Issued capital
|
|||||
|
Chance
High International Limited
|
British
Virgin Islands
|
July
2, 2009
|
100
|
%
|
USD50,000
|
||||
|
Yantai
Shencaojishi Pharmaceuticals Co., Ltd.
|
People’s
Republic of China
|
November
25, 2009
|
100
|
%
|
USD9,500,000
|
||||
|
Yantai
Bohai Pharmaceuticals Group Co., Ltd.
|
People’s
Republic of China
|
July
8, 2004
|
*
|
USD2,918,000
(RMB20,000,000)
|
|||||
*
The Group has indirect controlling interest of Bohai
under the VIE Agreements entered on December 7, 2009, which are described in
Note 1 above.
Initial
measurement of VIE - The Company initially measures the assets, liabilities, and
non-controlling interests of the VIEs at their carrying amount at the date of
the acquisitions.
Accounting
after initial measurement of VIE - Subsequent accounting for the assets,
liabilities, and non-controlling interest of a consolidated variable interest
entity are accounted for as if the entity were consolidated based on voting
interests and the usual accounting rules for which the VIE operates are applied
as they would to a consolidated subsidiary as follows:
|
|
·
|
Carrying
amounts of the VIE are consolidated into the financial statements of the
Company as the primary beneficiary (referred as “Primary Beneficiary” or
“PB”); and
|
|
|
·
|
Inter-company
transactions and balances, such as revenues and costs, receivables and
payables between or among the Primary Beneficiary and the VIE(s) are
eliminated in their entirety.
|
F-7
|
(b)
|
Economic
and Political Risks
|
The
Group’s operations are conducted solely in the PRC. There are
significant risks associated with doing business in the PRC, among others,
political, economic, legal and foreign currency exchange risks. The
Group’s results may be adversely affected by changes in the political and social
conditions in the PRC, and by changes in governmental policies with respect to
laws and regulations, anti-inflationary measures, currency conversion,
remittances abroad, and rates and methods of taxation, among other
things.
|
(c)
|
Use
of Estimates
|
In
preparing the condensed consolidated financial statements in conformity with US
GAAP, management makes estimates and assumptions that affect the reported
amounts of assets and liabilities and disclosures of contingent assets and
liabilities at the dates of the financial statements, as well as the reported
amounts of revenues and expenses during the reporting periods. These
accounts and estimates include, but are not limited to, the valuation of
accounts receivable, inventories, deferred income taxes, derivative liabilities,
and the estimation on useful lives of plant and machinery. Actual
results could differ from those estimates.
|
(d)
|
Cash
and Cash Equivalents
|
The
Group considers all highly liquid investments purchased with original
maturities of three months or less to be cash equivalents. The Group
maintains bank accounts only in the PRC. The Company does not
maintain any bank accounts in the United States of America. As of
March 31, 2010 and June 30, 2009, there were cash and cash equivalents of
$15,574,909 and $2,493,510 respectively.
|
(e)
|
Accounts
Receivable
|
Accounts
receivable consists of amounts due from customers. The Group extends unsecured
credit to its customers in the ordinary course of business but mitigates the
associated risks by performing credit checks and actively pursuing past due
accounts. An allowance for doubtful accounts is established and
determined based on management’s assessment of known requirements, aging of
receivables, payment history, the customer’s current credit worthiness and the
economic environment.
|
(f)
|
Inventories
|
Inventories
are valued at the lower of cost or market with cost is determined on the
weighted average method. Finished goods inventories consist of raw materials,
direct labor and overhead associated with the manufacturing process. In
assessing the ultimate realization of inventories, the management makes
judgments as to future demand requirements compared to current or committed
inventory levels. The Group’s reserve requirements generally
increase/decrease due to management projected demand requirements, market
conditions and product life cycle changes. As of March 31, 2010 and
June 30, 2009, the Group did not make any allowance for slow-moving or defective
inventories.
|
(g)
|
Fair
value of Financial Instruments
|
The carrying
values of the Group’s financial instruments, including cash and cash
equivalents, accounts receivables, other receivables and prepayments, short-term
borrowings, accounts payables, derivative instruments, and other accrued
liabilities are at their fair values due to the maturity of such
instruments.
F-8
|
(h)
|
Intangible
Assets
|
Intangible
assets consist of “Pharmaceutical Formulas”, acquired with indefinite
useful live are measured initially at cost and not subject to amortization shall
be tested for impairment annually or more frequently if there is indication of
impairment. If the carrying amount exceeds fair value, an impairment loss should
be recognized. Subsequently reversal of a recognized impairment loss is
prohibited.
(i) Property,
plant and equipment
Property,
plant and equipment, other than construction in progress, are stated at cost
less depreciation and amortization and accumulated impairment
loss. Cost represents the purchase price of the asset and other costs
incurred to bring the asset into its existing use. Maintenance,
repairs and betterments, including replacement of minor items, are charged to
expense; major additions to physical properties are capitalized.
Depreciation
of property, plant and equipment is calculated to written off the cost, less
their estimated residual value, if any, using the straight-line method over
their estimated useful lives. The principal annual rates are as
follows:
|
Leasehold
land and buildings
|
30
to 40 years
|
|
Motor
vehicles
|
10
years
|
|
Plant
and machinery
|
10
years
|
|
Office
equipment
|
5
years
|
Upon sale
or disposition, the applicable amounts of asset cost and accumulated
depreciation are removed from the accounts and the net amount less proceeds from
disposal is charged or credited to income.
Construction
in progress primarily represents costs incurred to construct the Group’s
corporate campus and machinery under construction. The corporate
campus was completed and at began production in 2009. Assets under
construction are not depreciated until the construction is completed and the
assets are ready for their intended use.
|
(j)
|
Accounting
for the Impairment of Long-Lived Assets
|
The Group
uses ASC Topic 360, which addresses financial accounting and reporting for the
impairment or disposal of long-lived assets. The
Group periodically evaluates the carrying value of long-lived assets to be
held and used in accordance with ASC Topic 360. ASC Topic 360
requires impairment losses to be recorded on long-lived assets used in
operations when indicators of impairment are present and the undiscounted cash
flows estimated to be generated by those assets are less than the assets’
carrying amounts. In that event, a loss is recognized based on the amount by
which the carrying amount exceeds the fair market value of the long-lived
assets. Loss on long-lived assets to be disposed of is determined in
a similar manner, except that fair market values are reduced for the cost of
disposal. Based on its review, the Group believes that, as of March 31, 2010 and
June 30, 2009, there were no significant impairments of its long-lived assets.
|
(k)
|
Foreign
Currency Translation
|
The Group
maintains its condensed consolidated financial statements in the functional
currency. The functional currency of the Group is the Chinese Renminbi (RMB).
Monetary assets and liabilities denominated in currencies other than the
functional currency are translated into the functional currency at rates of
exchange prevailing at the balance sheet dates. Transactions denominated in
currencies other than the functional currency are translated into the functional
currency at the exchanges rates prevailing at the dates of the transaction.
Exchange gains or losses arising from foreign currency transactions are included
in the determination of net income for the respective periods.
F-9
For
financial reporting purposes, the financial statements of the Group which are
prepared using the functional currency have been translated into United States
dollars. Assets and liabilities are translated at the exchange rates at the
balance sheet dates and revenue and expenses are translated at the average
exchange rates and stockholders’ equity is translated at historical exchange
rates. Any translation adjustments resulting are not included in determining net
income but are included in foreign exchange adjustment to other comprehensive
income, a component of stockholders’ equity.
|
Three months
ended
March 31,
2010
|
Three months
ended
March 31,
2009
|
Nine months
ended
March 31,
2010
|
Nine months
ended
March 31,
2009
|
|||||||||||||
|
Year
end US$: RMB exchange rate
|
6.83610
|
6.84560
|
6.83610
|
6.84560
|
||||||||||||
|
Average
periodic US$: RMB exchange rate
|
6.83603
|
6.84658
|
6.83773
|
6.85094
|
||||||||||||
RMB is
not freely convertible into foreign currency and all foreign exchange
transactions must take place through authorized institutions. No
representation is made that the RMB amounts could have been, or could be,
converted into US$ at the rates used in translation.
|
(l)
|
Revenue
Recognition
|
Revenue
represents the invoiced value of goods sold recognized upon the delivery of
goods to customers. Revenue is recognized when all of the following criteria are
met:
|
|
·
|
Persuasive
evidence of an arrangement exists;
|
|
|
·
|
Delivery
has occurred or services have been rendered;
|
|
|
·
|
The
seller’s price to the buyer is fixed or determinable; and
|
|
|
·
|
Collectability
is reasonably assured. Payments have been established.
|
The
return policy of goods is recognized in actual basis when the goods sold were
being returned by customer then the closing balance of the period/year is
actually deducted by the increment of relevant cost of sales. As the
sales return rate is low and immaterial, no such provision is required to be
made.
|
(m)
|
Cost
of Revenue
|
Regarding
the trading of medicine, the respective cost of revenue consists primarily of
material cost, labor cost, overhead associated with the manufacturing process
and related expenses which are directly attributable to the
trading.
|
(n)
|
Research
and Development Costs
|
Research
and development costs are charged as expense when incurred and included in
operating expenses.
F-10
|
(o)
|
Income
Taxes
|
Income
taxes are accounted for under the asset and liability
method. Deferred tax assets and liabilities are recognized for the
future tax consequences attributable to differences between the financial
statement carrying amounts of existing assets and liabilities and their
respective tax bases and operating loss and tax credit
carry-forwards. Deferred tax assets are reduced by a valuation
allowance to the extent management concludes it is more likely than not that the
assets will not be realized. Deferred tax assets and liabilities are
measured using enacted tax rates expected to apply to taxable income in the
years in which those temporary differences are expected to be recovered or
settled. The effect on deferred tax assets and liabilities of a
change in tax rates is recognized in the statements of income and
comprehensive income in the periods that includes the enactment
date.
|
(p)
|
Comprehensive
Income
|
Comprehensive
income is defined to include all changes in equity except those resulting from
investments by owners and distributions to owners. Among other
disclosures, all items that are required to be recognized under current
accounting standards as components of comprehensive income are required to be
reported in a condensed consolidated financial statement that is presented with
the same prominence as other financial statements. The Group’s current
components of other comprehensive income are the foreign currency translation
adjustment.
|
(q)
|
Commitments
and Contingencies
|
Liabilities
for loss contingencies arising from claims, assessments, litigation, fines and
penalties and other sources are recorded when it is probable that a liability
has been incurred and the amount of the assessment can be reasonably
estimated.
|
(r)
|
Earning
per Shares
|
The Group
reports basic earnings per share in accordance with ASC Topic 260, “Earnings Per
Share”. Basic earnings/(loss) per share is computed by dividing net
income/(loss) by weighted average number of shares of common stock outstanding
during the period. Diluted earnings per share is computed by dividing net income
by the weighted average number of shares of common stock, common stock
equivalents and potentially dilutive securities outstanding during the period.
Common equivalent shares are excluded from the computation in periods for which
they have an anti-dilutive effect. Stock options for which the exercise price
exceeds the average market price over the period are anti-dilutive and,
accordingly, are excluded from the calculation. At March 31, 2010, the Group had
no common stock equivalents that could potentially dilute future earnings per
share
|
(s)
|
Recent
Accounting Pronouncements
|
In March
2008, the FASB issued SFAS No. 161, DISCLOSURES ABOUT DERIVATIVE INSTRUMENTS AND
HEDGING ACTIVITIES (an amendment to SFAS No. 133). This statement is effective
for condensed consolidated financial statements issued for fiscal year and
interim periods beginning after November 15, 2008 and requires enhanced
disclosures with respect to derivative and hedging activities. The Group will
comply with the disclosure requirements of this statement if it utilizes
derivative instruments or engages in hedging activities upon its
effectiveness.
In April
2008, the FASB issued FASB Staff Position No. 142-3, DETERMINATION OF THE USEFUL
LIFE OF INTANGIBLE ASSETS (“FSP No. 142-3”) to improve the consistency between
the useful life of a recognized intangible asset (under SFAS No. 142) and the
period of expected cash flows used to measure the fair value of the intangible
asset (under SFAS No. 141(R)). FSP No. 142-3 amends the factors to be considered
when developing renewal or extension assumptions that are used to estimate an
intangible asset’s useful life under SFAS No. 142. The guidance in the new staff
position is to be applied prospectively to intangible assets acquired after
December 31, 2008. In addition, FSP No.142-3 increases the disclosure
requirements related to renewal or extension assumptions. The Company does not
believe implementation of FSP No. 142-3 have a material impact on its condensed
consolidated financial statements.
F-11
In May
2008, the FASB issued statement No. 162, THE HIERARCHY OF GENERALLY ACCEPTED
ACCOUNTING PRINCIPLES. This statement identifies the sources of accounting
principles and the framework for selecting the principles to be used in the
preparation of financial statements of nongovernmental entities that are
presented in conformity with generally accepted accounting principles (GAAP) in
the United States (the GAAP hierarchy). This statement is effective 60 days
following the SEC’s approval of the Public Company Accounting Oversight Board
amendments to AU Section 411, “the Meaning of Present Fairly in Conformity With
Generally Accepted Accounting Principles”.
In May
2008, the FASB issued FSP Accounting Principles Board (“APB”) 14-1 “Accounting
for Convertible Debt instruments That May Be Settled in Cash upon Conversion
(Including Partial Cash Settlement)” (“FSP APB 14-1”). FSP APB 14-1
requires the issuer of certain convertible debt instruments that may be settled
in cash (or other assets) on conversion to separately account for the liability
(debt) and equity (conversion option) components of the instrument in a manner
that reflects the issuer’s non-convertible debt borrowing rate. FSP
APB 14-1 is effective for fiscal years beginning after December 15, 2008 on
a retroactive basis. As we do not have convertible debt at this time, we
currently believe the adoption of FSP APB 14-1 will have no effect on our
combined results of operations and financial condition.
In May
2008, the FASB issued Statement No. 163, ACCOUNTING FOR FINANCE GUARANTEE
INSURANCE CONTRACTS - AN INTERPRETATION OF FASB STATEMENT NO. 60. The premium
revenue recognition approach for a financial guarantee insurance contract links
premium revenue recognition to the amount of insurance protection and the period
in which it is provided. For purposes of this statement, the amount of insurance
protection provided is assumed to be a function of the insured principal amount
outstanding, since the premium received requires the insurance enterprise to
stand ready to protect holders of an insured financial obligation from loss due
to default over the period of the insured financial obligation. This Statement
is effective for condensed consolidated financial statements issued for fiscal
years beginning after December 15, 2008.
In June
2008, the FASB issued FASB Staff Position Emerging Issues Task Force (EITF) No.
03-6-1, DETERMINING WHETHER INSTRUMENTS GRANTED IN SHARE-BASED PAYMENT
TRANSACTIONS ARE PARTICIPATING SECURITIES (“FSP EITF No. 03-6-1”). Under FSP
EITF No. 03-6-1, unvested share-based payment awards that contain rights to
receive nonforfeitable dividends (whether paid or unpaid) are participating
securities, and should be included in the two-class method of computing EPS. FSP
EITF No. 03-6-1 is effective for fiscal years beginning after December 15, 2008,
and interim periods within those years, and is not expected to have a
significant impact on the Company’s condensed consolidated financial
statements.
In April
2009, the FASB issued FSP 157-4, DETERMINING FAIR VALUE WHEN THE VOLUME AND
LEVEL OF ACTIVITY FOR THE ASSET OR LIABILITY HAVE SIGNIFICANTLY DECREASED AND
IDENTIFYING TRANSACTIONS THAT ARE NOT ORDERLY (“FSP 157-4”). FSP 157-4 provides
additional guidance for estimating fair value in accordance with SFAS 157
when the volume and level of activity for the asset or liability have
significantly decreased. FSP 157-4 also includes guidance on identifying
circumstances that indicate a transaction is not orderly. FSP 157-4 is effective
for interim and annual reporting periods ending after June 15, 2009, with
early adoption permitted for periods ending after March 15, 2009. FSP 157-4 does
not require disclosures for earlier periods presented for comparative purposes
at initial adoption. In periods after initial adoption, FSP 157-4 requires
comparative disclosures only for periods ending after initial adoption. The
adoption of the provisions of FSP 157-4 is not anticipated to materially impact
on the Company’s results of operations or the fair values of its assets and
liabilities.
F-12
In May
2009, the FASB issued SFAS No. 165, SUBSEQUENT EVENTS (“SFAS 165”). SFAS 165
establishes general standards for accounting for and disclosure of events that
occur after the balance sheet date but before financial statements are issued or
available to be issued and was effective for interim and annual periods ending
after June 15, 2009. The adoption of SFAS No. 165 did not have an impact on the
Company’s results of operations or financial condition. The Company evaluated
all subsequent events that occurred from January 1, 2010 through January 30,
2010, inclusive, and does not found any material subsequent events are required
to disclose.
In June
2009, the FASB issued SFAS No. 166 ACCOUNTING FOR TRANSFERS OF FINANCIAL ASSETS
(“SFAS 166). This statement is intended to improve the relevance,
representational faithfulness, and comparability of the information that a
reporting entity provides in its financial reports about a transfer of financial
assets; the effects of a transfer on its financial position, financial
performance, and cash flows; and a transferor’s continuing involvement in
transferred financial assets. This Statement must be applied as of the beginning
of each reporting entity’s first annual reporting period that begins after
November 15, 2009, and is required to be adopted by the Company in the first
quarter of fiscal year 2011. Earlier application is prohibited. This Statement
must be applied to transfers occurring on or after the effective date. The
Company does not expect the adoption of SFAS 166 to have a material impact
on the Company’s financial position, results of operations and cash
flows.
In June
2009, the FASB issued SFAS No.167, “Amendments to FASB Interpretation No.46(R)”,
which is codified as ASC 810. ASC 810 amends FASB Interpretation No.46(R),
“Variable Interest Entities” for determining whether an entity is a variable
interest entity (“VIE”) and requires an enterprise to perform an analysis to
determine whether the enterprise’s variable interest or interests give it a
controlling financial interest in a VIE. Under ASC 810, an enterprise has a
controlling financial interest when it has a) the power to direct the activities
of a VIE that most significantly impact the entity’s economic performance and b)
the obligation to absorb losses of the entity or the right to receive benefits
from the entity that could potentially be significant to the VIE. ASC 810 also
requires an enterprise to assess whether it has an implicit financial
responsibility to ensure that a VIE operates as designed when determining
whether it has power to direct the activities of the VIE that most significantly
impact the entity’s economic performance. ASC 810 also requires ongoing
assessments of whether an enterprise is the primary beneficiary of a VIE,
requires enhanced disclosures and eliminates the scope exclusion for qualifying
special-purpose entities. ASC 810 shall be effective as of the beginning of each
reporting entity’s first annual reporting period that begins after November 15,
2009, for interim periods within that first annual reporting period, and for
interim and annual reporting periods thereafter. Earlier application is
prohibited. ASC 810 is effective for the Company in the first quarter of fiscal
2011. The Company is currently evaluating the effect of ASC 810 on its condensed
consolidated financial statements and results of operation and is currently not
yet in a position to determine such effects.
F-13
In August
2009, the FASB issued Accounting Standards Update (“ASU”) No. 2009-05,
“Measuring Liabilities at Fair Value”, which is codified as ASC 820, “Fair Value
Measurements and Disclosures”. This Update provides amendments to ASC 820-10,
Fair Value Measurements and Disclosures –Overall, for the fair value measurement
of liabilities. This Update provides clarification that in circumstances in
which a quoted price in an active market for the identical liability is not
available, a reporting entity is required to measure fair value using a
valuation technique that uses the quoted price of the identical liability
when traded as an asset, quoted prices for similar liabilities or similar
liabilities when traded as assets, or that is consistent with the principles of
ASC 820. The amendments in this Update also clarify that when estimating the
fair value of a liability, a reporting entity is not required to include a
separate input or adjustment to other inputs relating to the existence of a
restriction that prevents transfer of the liability. The amendments in this
Update also clarify that both a quoted price in an active market for the
identical liability at the measurement date and the quoted price for the
identical liability when traded as an asset in an active market when no
adjustments to the quoted price of the assets are required are Level 1 fair
value measurements. ASC 820 is effective for the first reporting period
(including interim periods) beginning after August 28, 2009. The adoption of
this Update did not have a significant impact to the Company’s condensed
consolidated financial statements.
In
December 2009, the FASB issued ASU No. 2009-17, “Improvements to Financial
Reporting by Enterprises Involved with Variable Interest Entities (“ASU
2009-17”)”. ASU 2009-17 amends the variable-interest entity guidance in FASB ASC
810-10-05-8 to clarify the accounting treatment for legal entities in which
equity investors do not have sufficient equity at risk for the entity to finance
its activities without financial support. ASU 2009-17 shall be effective as of
the beginning of each reporting entity’s first annual reporting period that
begins after November 15, 2009. ASU 2009-17 is effective for the Company in the
first quarter of fiscal 2011. The Company is currently evaluating the effect of
ASU 2009-17 on its condensed consolidated financial statements and results of
operation and is currently not yet in a position to determine such
effects.
None of
the above new pronouncements has current application to the Group, but may be
applicable to the Company’s future financial reporting.
|
4.
|
OTHER
RECEIVABLES AND PREPAYMENTS
|
Other
receivables and prepayments consist of the following:
|
As of
March 31,
2010
|
As of
June 30,
2009
|
|||||||
|
(Unaudited)
|
(Audited)
|
|||||||
|
Prepayment
for advertising and promotion
|
$
|
2,925,645
|
$
|
1,736,025
|
||||
|
Loan
to a third party
|
-
|
1,465,000
|
||||||
|
Other
receivables
|
3,312,433
|
9,792,775
|
||||||
|
Deposit
for closing of private placement
|
731,491
|
-
|
||||||
|
Total
other receivables and prepayments
|
$
|
6,969,569
|
$
|
12,993,800
|
||||
Loan to a
third party is unsecured, interest bearing at 5.31% per annum and repayable on
June 8, 2010.
|
5.
|
AMOUNT
DUE FROM EQUITY HOLDER
|
Amount
due from an equity holder consists of the following:
|
As of
March 31,
2010
|
As of
June 30,
2009
|
|||||||
|
(Unaudited)
|
(Audited)
|
|||||||
|
Amount
due from a equity holder
|
$
|
-
|
$
|
1,465,000
|
||||
F-14
The
amount due from an equity holder is unsecured, interest bearing at 3.93% per
annum and has no fixed term of repayment.
|
6.
|
INVENTORIES
|
Inventories
consist of the following:
|
As of
|
As of
|
|||||||
|
|
March 31,
|
June 30,
|
||||||
|
|
2010
|
2009
|
||||||
|
(Unaudited)
|
(Audited)
|
|||||||
|
Raw
materials
|
$
|
393,475
|
$
|
250,405
|
||||
|
Finished
goods
|
577,189
|
57,429
|
||||||
|
Total
inventories
|
$
|
970,664
|
$
|
307,834
|
||||
|
7.
|
INTANGIBLE
ASSETS
|
Intangible
assets consist of the following:
|
As of
|
As of
|
|||||||
|
|
March 31,
|
June 30,
|
||||||
|
|
2010
|
2009
|
||||||
|
(Unaudited)
|
(Audited)
|
|||||||
|
Pharmaceuticals
formulas, at cost
|
$
|
17,273,007
|
$
|
17,298,720
|
||||
|
8.
|
PROPERTY,
PLANT AND EQUIPMENT, NET
|
The
following is a summary of property, plant and equipment:
|
As of March 31,
2010
|
As of June 30,
2009
|
|||||||
|
(Unaudited)
|
(Audited)
|
|||||||
|
Cost
|
||||||||
|
Leasehold
land and buildings
|
$
|
7,598,806
|
$
|
7,447,211
|
||||
|
Plant
and equipment
|
1,235,146
|
1,156,557
|
||||||
|
Office
equipment
|
77,597
|
74,700
|
||||||
|
Motor
vehicles
|
412,954
|
389,075
|
||||||
|
Total
|
9,324,503
|
9,067,543
|
||||||
|
Accumulated
depreciation
|
||||||||
|
Leasehold
land and buildings
|
553,961
|
414,133
|
||||||
|
Plant
and equipment
|
671,032
|
617,279
|
||||||
|
Office
equipment
|
20,660
|
10,681
|
||||||
|
Motor
vehicles
|
132,635
|
112,768
|
||||||
|
Total
|
1,378,287
|
1,154,861
|
||||||
|
Construction
in progress
|
-
|
236,597
|
||||||
|
Net
|
||||||||
|
Leasehold
land and buildings
|
7,044,845
|
7,033,078
|
||||||
|
Plant
and equipment
|
564,114
|
539,278
|
||||||
|
Office
equipment
|
56,936
|
64,019
|
||||||
|
Motor
vehicles
|
280,320
|
276,307
|
||||||
|
Property,
plant and equipment, net
|
$
|
7,946,215
|
$
|
8,149,279
|
||||
F-15
Depreciation
expenses for the three months and nine months ended March 31, 2010 were $84,518
and $224,656 respectively.
As of
March 31, 2010 and June 30, 2009, Bohai had pledged plant and machinery having a
carrying amount of $552,710 and $562,331 to secure a bank loan to Bohai.
|
9.
|
SHORT-TERM
BORROWINGS
|
Bohai
obtained several short-term loan facilities from financial institution in the
PRC. Short-term borrowings as of March 31, 2010 consist of the
following:
|
Loan from
financial
institution
|
Loan period
|
Annual
Interest rate
|
Secured by
|
Amount
(unaudited)
|
||||||||
|
China
Construction Bank
|
From
Feb 24, 2010 to
Feb
23, 2011
|
5.8410
|
%
|
Personal
guarantee by equity holder
|
$
|
3,510,773
|
||||||
|
Rural
Credit Union
|
From
Sep 28, 2009 to
Sep
26, 2010
|
5.7525
|
%
|
Yantai
Ka Wah Medical Equipment Co. Ltd
|
285,990
|
|||||||
|
Yantai
Laishan Rural Credit Union
|
From
Sep 28, 2009 to
Sep
26, 2010
|
7.5225
|
%
|
Bohai’s
machinery and vehicle
|
584,390
|
|||||||
|
Total
|
$
|
4,381,153
|
||||||||||
Short-term
borrowings as of June 30, 2009 consist of the following:
|
Loan from
financial
institution
|
Loan period
|
Annual
Interest rate
|
Secured by
|
Amount
(Audited)
|
||||||||
|
Shanghai
Pudong Development
Limited
|
From
Dec 12, 2008 to
Dec
11, 2009
|
6.6960
|
%
|
Haiyang
Construction Industry Training Centre and personal guarantee by equity
holders
|
$
|
2,197,500
|
||||||
|
Yantai
City Commercial Bank
|
From
Jan 20, 2009 to
Jan
20, 2010
|
6.9030
|
%
|
Yantai
Hai Pu Can End Making Co. Ltd
|
1,318,500
|
|||||||
|
Yantai
Laishan Rural Credit Union
|
From
Sep 27, 2008 to
Sep
26, 2009
|
9.3600
|
%
|
Yantai
Ka Wah Medical Equipment Co. Ltd
|
293,000
|
|||||||
|
Yantai
Laishan Rural Credit Union
|
From
Sep 27, 2008 to
Sep
26, 2009
|
12.2400
|
%
|
Company’s
machinery and vehicle
|
586,000
|
|||||||
|
China
Construction Bank
|
From
May 12, 2009 to
Nov
11, 2009
|
0.0000
|
%
|
Personal
guarantee by equity holders
|
1,465,000
|
|||||||
|
Total
|
$
|
5,860,000
|
||||||||||
F-16
|
10.
|
NOTES
PAYABLE
|
In June
2009, the Company issued four promissory notes for an aggregate of
$400,000. The funds had been raised previously on the Company’s
behalf by counsel in a private placement and held in trust
accounts. Funds were expended from the trusts for professional fees
and due diligence related to an ongoing search for suitable business
opportunities, anticipating merger. In June 2009, the Company assumed
responsibility for the fees and due diligence costs. On January 5,
2010, the notes converted pursuant to their terms into shares of Common Stock as
a result of the Share Exchange transaction.
|
11.
|
OTHER
ACCRUED LIABILITIES
|
Other accrued
liabilities consist of the following:
|
As of
|
As of
|
|||||||
|
|
March 31,
|
June 30,
|
||||||
|
|
2010
|
2009
|
||||||
|
(Unaudited)
|
(Audited)
|
|||||||
|
Accrued
selling expenses
|
$
|
579,545
|
$
|
1,677,026
|
||||
|
Accrued
staff costs
|
212,198
|
173,130
|
||||||
|
Value
added tax payable
|
716,822
|
709,688
|
||||||
|
Other
accrued expenses
|
206,865
|
77,374
|
||||||
|
Others
payable
|
225,658
|
9,702,137
|
||||||
|
Total
other accrued liabilities
|
$
|
1,941,088
|
$
|
12,339,355
|
||||
|
12.
|
CONVERTIBLE
PROMISSORY NOTES AND WARRANTS
|
On
January 5, 2010, pursuant to a Securities Purchase Agreement (the “Securities
Purchase Agreement”) with 128 accredited investors (the “Investors”), the
Company sold 6,000,000 units for aggregate gross proceeds of $12,000,000, each
unit consisting of an 8% senior convertible promissory note in the principal
amount of $2 and one common stock purchase warrant (each, an “Investor Warrant”
and collectively, the “Investor Warrants”). By agreement with the
Investors, each investor received: (i) a single Note representing the aggregate
number of Notes purchased by them as part of the units (each, a “Note” and
collectively, the “Notes”) and (ii) a single Investor Warrant representing the
aggregate number of Investor Warrants purchased by them as part of the units.
The Notes
bear interest at 8% per annum, payable quarterly in arrears on the last day of
each fiscal quarter of the Company. No principal payments are
required until maturity of the Notes on January 5, 2012. Each Note,
plus all accrued but unpaid interest thereon, is convertible, in whole but not
in part, at any time at the option of the holder, into shares of the Company’s
common stock, par value $0.001 per share, at a conversion price of $2.00 per
share, subject to adjustment as set forth in the Note. The 128 Notes
issued have face amounts that range from $43,200 to $500,000 .
F-17
The
conversion price of the Notes is subject to standard anti-dilution adjustments
for stock splits and similar events. In addition, in the event the
Company issue or sells any additional shares of Common Stock or instruments
convertible or exchangeable for Common Stock at a price per share less than the
conversion price then in effect or without consideration, then the conversion
price upon each such issuance will be adjusted to that price determined by
multiplying the conversion price then in effect by a fraction: (1) the numerator
of which is the sum of (x) the number of shares of Common Stock outstanding
immediately prior to the issuance of such additional shares of Common Stock plus
(y) the number of shares of Common Stock which the aggregate consideration for
the total number of such additional shares of Common Stock so issued would
purchase at a price per share equal to the conversion price then in effect, and
(2) the denominator of which is the number of shares of Common Stock outstanding
immediately after the issuance of such additional shares of Common Stock.
Notwithstanding any provision of the Note to the contrary, no adjustment will
cause the conversion price to be less than $1.00, as adjusted for any stock
dividend, stock split, stock combination, reclassification or similar
transaction.
The Notes
contain certain Events of Default, including non-payment of interest or
principal when due, bankruptcy, failure to maintain a listing of the Common
Stock or to make required filings on a timely basis. No premium is
payable by the Company if an Event of Default occurs. However, upon
an Event of Default, and provided no more than 50% of the aggregate face amount
of the Notes have been converted, the Investors holding Notes have the right to
receive a portion, based on their pro-rata participation in the transaction, of
1,000,000 shares of the Company’s Common Stock that have been placed in escrow
by the Company’s principal stockholder. The shares in escrow will be returned to
the principal stockholder when 50% of the aggregate face amount of the Notes has
been converted or, if later, when the Notes are repaid.
The
Investor Warrants expire on January 5, 2013 and may be exercised by the holder
at any time to purchase one share of Common Stock at an exercise price of $2.40
per share (subject to adjustment as set forth in the Investor
Warrants). The exercise price of the Investor Warrants is subject to
adjustment in the same manner as the conversion price of the Notes described
above, except that the exercise price will not be adjusted to less than $1.20,
as adjusted for any stock dividend, stock split, stock combination,
reclassification or similar transaction. The Investor Warrants may
only be exercised for cash and do not permit the holder to perform a cashless
exercise.
In
connection with the sale of the units, the Company paid its placement agents a
cash fee of $1,200,000. In addition, the placement agents received warrants (the
“Placement Agent Warrants” and, together with the Investor Warrants, the
“Warrants”) to purchase 600,000 shares of Common Stock, which warrants are
substantially identical to the Investor Warrants, except that, pursuant to
separate lock-up agreements executed by the holders of the Placement Agent
Warrants, the Placement Agent Warrants are not exercisable until the six month
anniversary of the later of: (i) the date of effectiveness of the registration
statement registering the resale of the Common Stock underlying the Notes and
Warrants or (ii) the date of commencement of sales in connection with such
registration statement.
In
addition to the placement agent fee, the Company paid $370,000 of legal and
other expenses. As required by the Securities Purchase Agreement,
$500,000 of the proceeds from the sale of the units were placed in escrow to pay
investor relations expenses to be incurred by the Company and $240,000,
equivalent to one quarter’s interest expense on the Notes, was also placed in
escrow. The interest escrow will be released to the Company at such
time as 75% of all shares underlying the Notes have been issued upon conversion
of Notes. After payment of the placement agent fees and other
expenses and the amounts required to be placed in escrow, the Company received
net proceeds of $9,690,000. At March 31, 2010, $731,491 remained in
escrow and is included in Other Receivables and Prepayments (see Note 4).
F-18
The
Company also entered into a Registration Rights Agreement with the
Investors. The Company agreed to file, no later than March 6, 2010, a
registration statement to register the shares underlying the Notes and the
Warrants and to have such registration statement effective no later than August
13, 2010. The required registration statement was filed on March 2,
2010 but has not become effective as of March 31, 2010. If the
registration statement is not effective by the due date or if, after the
effective date, sales of securities included in the registration
statement cannot be made (including, without limitation, because of a failure to
keep the registration statement effective, to disclose such information as is
necessary for sales to be made pursuant to the registration statement, to
register a sufficient number of shares of Common Stock or to maintain the
listing of the Common Stock) then, as liquidated damages (and in complete
satisfaction and to the exclusion of any claims or remedies inuring to any
holder of the securities) the Company is required to pay an amount in cash equal
to 1% of the aggregate purchase price paid by the Investors on each of the
following dates: (i) 20 days following the date of a Filing Failure; (ii) 20
days following the date of an Effectiveness Failure; (iii) 30 days following the
initial day of a Maintenance Failure; (iv) on every thirtieth day thereafter
(pro-rated for periods totaling less than thirty days) until such failure is
cured. The payments to be made by the Company are limited to a maximum of 6% of
the aggregate amount paid by the Investors ($720,000). As of March
31, 2010, the Company does not expect to incur any registration delay payments
and has not accrued any such payments.
On and
effective as of June 30, 2010, the Company entered into an Amendment and
Agreement with the Investors, pursuant to which the Company and the Investors
agreed to make certain amendments to the Notes and the Warrants. Pursuant to the
Amendment, except for the standard anti-dilution adjustments for stock splits
and similar events, the anti-dilution protection provisions in the Notes and the
Warrants were eliminated and a provision specifically precluding net cash
settlement by the Company of the Notes and the Warrants was added. In return,
and subject to certain non-financing exceptions, the Company agreed not to issue
any new equity securities at a price per share below $2.20 until the earlier of
(i) January 5, 2013 or (ii) the date on which, collectively with any prior
conversions or exercises of Notes and Warrants, 75% of the principal face value
of the Notes in the aggregate has been converted into shares of Common Stock and
Warrants representing, in the aggregate, 75% of the aggregate shares of Common
Stock underlying the Warrants have been exercised. This Amendment will not
change the Company’s accounting for the Notes and the Warrants described below.
Valuation
At the
time the Notes and Warrants were issued, there had not been any market activity
for the Common Stock. Accordingly, determining the fair value
of the Common Stock required the Company to make complex and
subjective judgments. The Company estimated the value of its
enterprise as of January 5, 2010 based on a review of the enterprise value
derived from the use of market and income valuation approaches. The
Company also reviewed an asset-based approach to assess whether the result of
such an approach was consistent with the value derived from the market and
income valuation approaches. The market approach was based on the
market price to earnings multiple for companies considered by management to be
comparable to the Company. The income approach was based on applying
discount rates to estimated future net income. The estimated
enterprise value was then allocated to the Company’s existing outstanding Common
Stock, the Notes and the Warrants using the option pricing
method. The option pricing method was based on the two year period to
maturity of the Notes and the three year period to expiration of the Warrants,
risk-free interest rates commensurate with those periods and the expected
volatility used was based on a review of the historical volatility of companies
considered by management to be comparable to the Company.
F-19
Based on
the allocation of the estimated enterprise value, the Company estimated the fair
value of the Common Stock at $2.28 per share, as of January 5,
2010. The Investor Warrants and the Placement Agent Warrants were
valued at $5,824,538 and $582,454, respectively, based on the estimated fair
value of the Common Stock of $2.28, a term equal to the remaining life of the
Warrants, an expected dividend yield of 0%, a risk-free interest rate of 1.57%
based on constant maturity rates published by the U.S. Federal Reserve
applicable to the remaining life of the Warrants and estimated volatility of
65%, based on a review of the historical volatility of companies considered by
management to be comparable to the Company. As noted above, prior to
the June 30, 2010 Amendment described above, the Warrants contained a down-round
anti-dilution protection feature. As of January 5, 2010, the value of
this feature was not considered to be material and no adjustment was made for it
in the estimated fair value of the Warrants.
Accounting
for Convertible Notes
At
January 5, 2010 and March 31, 2010, the conversion options embedded in the Notes
are not derivative instruments as defined in FASB ASC 815-10-15-83 because the
Notes do not permit or require net settlement, there is no market mechanism
outside the contracts that permits net settlement and the shares to be received
on conversion of the Notes are not readily convertible to cash. At the time the
Notes were issued, there had not been any market activity for the Common
Stock. On March 31, 2010, an initial trade of 500 shares of the
Common Stock occurred in the market, the only trading activity during that
period. The Notes can be exercised only in whole but not in part and
through March 31, 2010 and continuing, there has been insufficient trading
volume to permit the shares to be received on conversion of each Note to be
readily sold in the market, thus precluding the shares to be received by the
holder of each Note from being readily convertible to cash.
In future
periods, whether or not the embedded conversion option in each Note is
considered to be a derivative instrument will depend on whether or not the
aggregate number of shares to be received on exercise of each of the 128 Notes,
which Notes can be exercised only in whole but not in part, could be readily
sold in the market without significantly affecting the market price of the
Common Stock, thus permitting the shares received by the holder of each Note to
be readily convertible to cash. At each reporting date, the Company
will re-evaluate each Note, based on the level of activity in the market for the
Common Stock at that time, to determine whether or not the embedded conversion
option in each Note is a derivative instrument. Depending on the
trading volume for the Common Stock that develops in the future and the face
amount of each Note, the embedded conversion option may be considered a
derivative instrument for some Notes but not for others and its status as a
derivative instrument may vary from period to period.
FASB ASC
815-10-15-74 provides that a contract which would otherwise meet the definition
of a derivative instrument but that is both (a) indexed to a company’s own stock
and (b) classified in stockholders’ equity in the statement of financial
position would not be considered a derivative financial
instrument. FASB ASC 815-40-15 and 815-40-25 provide guidance for
determining whether those two criteria are met. Because the Company’s
functional currency is the Renminbi but the Notes are denominated in U.S.
Dollars, FASB ASC 815-40-15-7I provides that the embedded conversion options are
not considered to be indexed only to the Company’s Common
Stock. Furthermore, prior to the June 30, 2010 Amendment described
above, the criteria that the instruments be indexed only to the Common Stock was
also not met because the conversion price of the Notes would be reduced if the
Company issued securities at a lower exercise or conversion
price. Because the requirement that the instruments be indexed only
to the Common Stock is not met, the exemption in FASB ASC 815-10-15-74 will not
be available and the Company will account for the embedded conversion options in
the Notes as derivative instrument liabilities, if and when the shares to be
issued on conversion are considered to be readily convertible to cash..
F-20
Derivative
financial instruments are initially measured at their fair value. For derivative
financial instruments that are accounted for as liabilities, the derivative
instrument is initially recorded at its fair value and is then re-valued at each
reporting date, with changes in the fair value reported as charges or credits to
income. If and when the embedded conversion option in any of the
Notes first qualifies as a derivative instrument, the fair value at that time of
the embedded derivative instrument will be re-classified and
separately recognized and subsequently marked-to-market each reporting period,
as long as the embedded conversion option continues to qualify as a derivative
instrument. If the embedded conversion option ceases to be a
derivative instrument, it will be marked-to-market as of the date of
re-classification but thereafter will no longer be marked-to-market.
Warrants
Because
the Company’s functional currency is the Renminbi but the Warrants are
denominated in U.S. Dollars, the Warrants are not considered to be indexed only
to the Company’s Common Stock. Furthermore, prior to the June 30,
2010 Amendment described above, the criteria that the instruments be indexed
only to the Common Stock was also not met because the exercise price of the
Warrants would be reduced if the Company issued securities at a lower
exercise or conversion price. In accordance with ASC 815-10-S99-4,
the Warrants (including the Placement Agent Warrants) are accounted for at fair
value, with changes in their fair value charged or credited to income each
period.
At
January 5, 2010, the Investor Warrants were valued at $5,824,538, as described
above. At March 31, 2010, the Investor Warrants were re-valued at $4,839,673
using a binomial model, based on the closing market price on that date of $2.10,
a term equal to the remaining life of the Warrants, an expected dividend yield
of 0%, a risk-free interest rate of 1.47% based on constant maturity rates
published by the U.S. Federal Reserve applicable to the remaining life of the
Warrants and estimated volatility of 65%, based on a review of the historical
volatility of companies considered by management to be comparable to the
Company. The effect of the down-round anti-dilution protection was not
considered to be material and no adjustment was made for it in the estimated
fair value of the Investor Warrants.
The
Placement Agent Warrants were initially valued at $582,454, as described
above.. The cost of these instruments, together with the cash fees
paid to the placement agents and the other fees and expenses paid by the
Company, as described above, in the aggregate amount of $2,152,454, have been
deferred and are being amortized on a straight-line basis over the two year
period to maturity of the Notes. At March 31, 2010, the Placement Agent Warrants
were re-valued at $483,967, based on the closing market price on that date of
$2.10, a term equal to the remaining life of the Warrants, an expected dividend
yield of 0%, a risk-free interest rate of 1.47% and estimated volatility of 65%.
The effect of the down-round anti-dilution protection was not considered to be
material and no adjustment was made for it in the estimated fair value of the
Placement Agent Warrants.
The
aggregate change in the value of the Investor and Placement Agent Warrants
between January 5, 2010 and March 31, 2010 of $1,083,350 has been credited to
income.
Convertible
Notes
The
Investor Warrants were initially recorded at their fair value of $5,824,538 and
the remainder of the $12,000,000 gross proceeds received from the Investors of
$6,175,463 was allocated to the Notes. Based on the proceeds
allocated to the Notes, the Notes are convertible into Common Stock at an
effective conversion price of approximately $1.03 per share. Because
the effective conversion price is less than the fair value of the Common Stock
at the time the Notes were issued, the Company recognized a beneficial
conversion feature, which was limited to the amount of proceeds allocated to the
Notes of $6,175,463. The Notes were initially recorded at a carrying
value of zero and are being amortized, together with interest accruing on the
Notes, to their maturity value over the period to maturity, at an effective
interest rate of approximately 540% per annum. Interest expense for
the period ended March 31, 2010 was $347,793. After payment of cash
interest due on March 31, 2010 of $226,667, the amortized cost carrying value of
the Notes at March 31, 2010 was $121,127.
F-21
Escrowed
Shares
As of
January 5, 2010 and at March 31, 2010, the Company’s principal stockholder is
obligated to deliver 1,000,000 shares of Common Stock to the Investors if
certain Events of Default occur (as defined in the Notes). The fair
value of this obligation is not considered to be material as the probability of
such events occurring is currently considered to be
minimal. Accordingly, at January 5, 2010 and March 31, 2010, no
liability for this obligation has been recognized.
Subsequent
Conversions
On April
1, 2010, Notes with an aggregate face amount of $500,000 were converted into
250,000 shares of Common Stock.
|
13.
|
DEFERRED
FEES ON CONVERTIBLE NOTES
|
The Company incurred total placement
fees of $2,152,454 in connection with our private placement of Convertible Notes
(see Note 12) that occurred on January 5, 2010. The placement fees
are being amortized on a straight line basis over the two year expected life of
the Convertible Notes, starting on the date of closing, January 5, 2010.
|
As of
|
As of
|
|||||||
|
|
March 31,
|
June 30,
|
||||||
|
|
2010
|
2009
|
||||||
|
(Unaudited)
|
(Audited)
|
|||||||
|
RESTATED
|
||||||||
|
Deferred
fees, beginning balance on January 5, 2010
|
$
|
2,152,454
|
$
|
-
|
||||
|
Amortization
of deferred fees
|
(253,577
|
) |
-
|
|||||
|
Deferred
fees, ending balance
|
$
|
1,898,877
|
$
|
-
|
||||
|
14.
|
SELLING,
GENERAL AND ADMINISTRATIVE EXPENSES
|
Selling,
general and administrative expenses consist of the following:
|
Three months
|
Three months
|
Nine months
|
Nine months
|
|||||||||||||
|
ended
|
ended
|
ended
|
ended
|
|||||||||||||
|
March 31,
|
March 31,
|
March 31,
|
March 31,
|
|||||||||||||
|
2010
|
2009
|
2010
|
2009
|
|||||||||||||
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
|||||||||||||
|
RESTATED
|
RESTATED
|
|||||||||||||||
|
Accommodation
|
$
|
1,106,984
|
$
|
565,205
|
$
|
2,880,151
|
$
|
2,724,140
|
||||||||
|
Advertising
and promotion
|
2,767,549
|
2,251,404
|
8,732,252
|
5,834,757
|
||||||||||||
|
Commission
|
1,060,462
|
1,503,000
|
3,464,415
|
4,726,934
|
||||||||||||
|
Conference
|
738,898
|
3,106
|
3,070,497
|
10,159
|
||||||||||||
|
Depreciation
|
9,397
|
118
|
27,872
|
353
|
||||||||||||
|
Staff
costs
|
608,263
|
566,413
|
1,571,400
|
1,256,934
|
||||||||||||
|
Travelling
|
717,766
|
1,132,551
|
2,000,775
|
1,933,017
|
||||||||||||
|
Research
and development cost
|
146,640
|
147,972
|
442,046
|
38,492
|
||||||||||||
|
Other
operating expenses
|
2,046,914
|
1,500,361
|
6,019,345
|
5,150,493
|
||||||||||||
|
Total
selling, general and administrative expenses
|
$
|
9,202,873
|
$
|
7,670,130
|
$
|
28,208,753
|
$
|
21,675,279
|
||||||||
F-22
|
15.
|
INTEREST
EXPENSES
|
Interest
expenses consist of the following:
|
Three months
|
Three months
|
Nine months
|
Nine months
|
|||||||||||||
|
ended
|
ended
|
ended
|
ended
|
|||||||||||||
|
March 31,
|
March 31,
|
March 31,
|
March 31,
|
|||||||||||||
|
2010
|
2009
|
2010
|
2009
|
|||||||||||||
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
|||||||||||||
|
Interest
on short-term borrowings wholly repayable within 1 year
|
$
|
33,907
|
$
|
52,921
|
$
|
190,215
|
$
|
168,422
|
||||||||
|
Effective
interest charge on Convertible Notes
|
347,793
|
-
|
347,793
|
-
|
||||||||||||
|
Total
interest expenses
|
$
|
381,700
|
$
|
52,921
|
$
|
538,008
|
$
|
168,422
|
||||||||
|
16.
|
FINANCE
AND NON-OPERATING (INCOMES) EXPENSES
|
Finance
and non-operating expense consists of the following:
|
Three months
|
Three months
|
Nine months
|
Nine months
|
|||||||||||||
|
ended
|
ended
|
ended
|
ended
|
|||||||||||||
|
March 31,
|
March 31,
|
March 31,
|
March 31,
|
|||||||||||||
|
2010
|
2009
|
2010
|
2009
|
|||||||||||||
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
|||||||||||||
|
RESTATED
|
RESTATED
|
|||||||||||||||
|
Amortization
of deferred fee – Convertible Notes
|
$
|
253,577
|
$
|
-
|
$
|
253,577
|
$
|
-
|
||||||||
|
Change
in fair value of warrants (credit)
|
(1,083,350
|
) |
-
|
(1,083,350
|
) |
-
|
||||||||||
|
Non-operating
expense
|
-
|
4,618
|
22,092
|
4,682
|
||||||||||||
|
Total
finance and non-operating (incomes) expenses
|
$
|
(829,773
|
) |
$
|
4,618
|
$
|
(807,681
|
) |
$
|
4,682
|
||||||
F-23
|
17.
|
INCOME
TAXES
|
|
Three months
|
Three months
|
Nine months
|
Nine months
|
|||||||||||||
|
ended
|
ended
|
ended
|
ended
|
|||||||||||||
|
March 31,
|
March 31,
|
March 31,
|
March 31,
|
|||||||||||||
|
2010
|
2009
|
2010
|
2009
|
|||||||||||||
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
|||||||||||||
|
Income
before taxes
|
$
|
3,727,693
|
$
|
2,528,403
|
$
|
9,946,524
|
$
|
7,437,907
|
||||||||
|
Income
tax expenses
|
$
|
585,135
|
$
|
423,569
|
$
|
2,193,931
|
$
|
1,224,833
|
||||||||
PRC
Tax
The PRC’s
legislative body, the National People’s Congress, adopted the unified Enterprise
Income Tax (“EIT”) Law on March 16, 2007. This new tax law replaces the existing
separate income tax laws for domestic enterprises and foreign-invested
enterprises and became effective on January 1, 2008. Under the new tax law, a
unified income tax rate is set at 25% for both domestic enterprises and
foreign-invested enterprises. However, there will be a transition period for
enterprises, whether foreign-invested or domestic, that are currently receiving
preferential tax treatments granted by relevant tax authorities. Enterprises
that are subject to an enterprise income tax rate lower than 25% may continue to
enjoy the lower rate and will transit into the new rate over a five year period
beginning on the effective date of the EIT Law. Enterprises that are currently
entitled to exemptions for a fixed term may continue to enjoy such treatment
until the exemption term expires. Preferential tax treatments may continue to be
granted to industries and projects that qualify for such preferential treatments
under the new law.
United
States Tax
The
Company is subject to income tax in the United States. No provision for income
tax in the United States has been made as the Company had no taxable income for
the three months and nine months ended March 31, 2010. The statutory tax rate is
34%.
A
reconciliation of the effective income tax rate to the US federal rate is as
follows:
|
March 31,
2010
|
June 30,
2009
|
|||||||
|
United
States Tax at statutory rate
|
$
|
3,381,818
|
$
|
2,528,889
|
||||
|
Foreign
tax rate difference
|
(1,187,887
|
) |
(1,384,301
|
)
|
||||
|
Exemption
in taxation
|
(2,193,931
|
)
|
(1,144,588
|
)
|
||||
|
$
|
-
|
$
|
-
|
|||||
Certain
of the Group’s revenue (including sales revenue) are subject to output VAT
generally calculated at 6%, 7%, 13% and 17% of the selling price. Input credit
relating to input VAT paid on purchase can be used to offset the output VAT.
The net
amount of VAT expense for the 9 months ended are shown as follows:
|
March 31,
2010
|
March 31,
2009
|
|||||||
|
Net
value added tax expense
|
$
|
6,627,557
|
$
|
5,274,607
|
||||
F-24
|
18.
|
COMMITMENTS
AND CONTINGENCIES
|
There are
no foreseeable material commitments or contingencies as of March 31, 2010 and
June 30, 2009.
|
19.
|
COMMON
EQUITY
|
Common
stock - $0.001 par value
|
Note
|
Common
Shares
|
|||||
|
Common
shares outstanding - June 30, 2009
|
3,450,000
|
|||||
|
Shares
issued for consulting services
|
a
|
1,137,500
|
||||
| Shares outstanding prior to January 5, 2010 reverse re-capitalization |
4,587,500
|
|||||
|
Shares
cancelled
|
b
|
(1,500,000
|
)
|
|||
|
Issued
to shareholders of Chance High Limited under Share Exchange Agreement date
January 5, 2010 (see Note 2)
|
b
|
13,162,500
|
||||
|
As
of March 31, 2010
|
16,250,000
|
|||||
(a) Prior
to the reverse re-capitalization, on December 17, 2009, the Company issued
687,500 shares to Cawston Enterprises Limited and 450,000 shares to Regeneration
Capital Group, LLC and its affiliates for consulting services fees rendered to
the Company. As described in Note 2, for accounting purposes, Chance
High is deemed to have acquired the Company as of January 5,
2010. The fair value of these shares was accounted for by the Company
prior to its acquisition by Chance High and is not reflected in these
consolidated financial statements.
(b) On
the Closing Date, pursuant to the terms of the Share Exchange Agreement date
January 5, 2010 (see Note 2), the Company acquired all of the outstanding equity
securities (the “Chance High Shares”) of Chance High from the Chance High
Shareholders, and the Chance High Shareholders transferred and contributed all
of their Chance High Shares to the Company. In exchange, the Company
issued to Chance High Shareholders an aggregate of 13,162,500 newly issued
shares of common stock, par value $0.001 per share (the “Common Stock”).
In addition, pursuant to the terms of the Share Exchange Agreement, Anthony
Zaradic, the former President and Chief Executive Officer of the Company,
cancelled a total of 1,500,000 shares of Common Stock.
F-25
As of March 31, 2010, the
Company has 150,000,000 shares of common stock authorized and 16,250,000 shares
of common stock issued and outstanding.
|
20.
|
STATUTORY
RESERVES
|
According
to the laws and regulations in the PRC, Bohai is required to provide for certain
statutory funds, namely, reserve fund by an appropriation from net profit after
taxes but before dividend distribution based on the local statutory financial
statements of the PRC company prepared in accordance with the accounting
principles and relevant financial regulations.
Bohai in
PRC is required to allocate at least 10% of its net profit to the reserve fund
until the balance of such fund has reached 50% of its registered capital.
Appropriation of enterprise expansion fund are determined at the discretion of
it directors.
The
reserve fund can only be used, upon approval by the relevant authority, to
offset accumulated losses or increase capital. The enterprise expansion fund can
only be used to increase capital upon approval by the relevant
authority.
|
21.
|
FINANCIAL
INSTRUMENTS
|
|
(a)
|
Credit
Risk
|
The
potential credit risk of the company is mainly attributable to its debtors and
bank balances. In respect of debtors, the Group has policies in place to
ensure that it will only accept customers from countries which are politically
stable and customers with an appropriate credit history. In addition, all
the bank balances were made with financial institutions with high-credit
quality. Thus, the Group is not considered to be subject to significant
credit risk.
|
(b)
|
Interest
Rate Risk
|
The
Group’s interest rate risk is primarily attributable to its short-term
borrowings, loan to a third party and loan to equity holders. The Group’s
borrowings carry interest at fixed rate. The management has not used any
interest rate swaps to hedge its exposure to interest rate risk.
|
(c)
|
Fair
Value Estimation
|
All of
the carrying amounts of the Group’s financial assets and liabilities of short
term maturities approximate their fair values.
|
22.
|
SUBSEQUENT
EVENTS
|
On and
effective as of June 30, 2010, Bohai Pharmaceuticals Group, Inc. (the "Company")
entered into an Amendment and Agreement (the “Agreement”) with Euro Pacific
Capital, Inc., as investor representative (the “Investor Representative”),
pursuant to which the Company and the Investor Representative agreed to make
certain amendments to the Company's 8% subordinated, unsecured Convertible
Promissory Notes, issued on January 5, 2010. The Amendment will not
result in any changes to the Company’s accounting for the Notes and Warrants.
Effective
July 12, 2010, the board of directors (consisting of Hongwei Qu, the sole
director) of Bohai Pharmaceuticals Group, Inc. (the "Company"), by written
consent to action and pursuant to the Company's Amended and Restated Bylaws
(which provides for a classified board of directors of the Company), appointed
three persons to serve on the board of directors of the Company.
|
23.
|
RESTATEMENT
|
Based on comments received by the
Company from the staff of the U.S. Securities and Exchange Commission, the
Company recently identified certain errors in its previously issued financial
statements for the three months and nine months ended March 31, 2010, which
financial statements were originally issued on May 17, 2010 and were initially
restated by the Company on June 24, 2010. These errors, which have
been corrected, resulting in the restated financial statements appearing herein,
include the following items. The accounting for these items is more
fully described in Note 12.
(i) the
Warrants and the Investor Warrants (each as defined in Note 12) should be valued
at fair market value and classified as a liability.
(ii) the
Notes (as defined in Note 12) should be stated net of a beneficial conversion
feature.
(iii) the
costs associated with the issuance of the Notes and Warrants have been deferred
and are being amortized over the two year period to maturity of the
Notes.
The
Warrants are not afforded equity treatment and, as such, all future changes in
the fair value of these Warrants will be recognized through earnings until such
time as the Warrants are exercised or expire.
The
Notes are accounted for net of a beneficial conversion feature and the
beneficial conversion feature will be amortized as a component of interest
expense over the term of the Notes.
The
tables below reflect the resulting changes to the financial statements (with the
numbers set forth under “As Previously Reported” being the results as disclosed
in the Company’s initially restated financial statements filed on June 24,
2010):
|
Three
Months Ended March 31, 2010
|
Nine
Months Ended March 31, 2010
|
|||||||||||||||
|
As
Previously
Reported
|
As
Restated
|
As
Previously
Reported
|
As
Restated
|
|||||||||||||
|
Statement
of Income Data:
|
||||||||||||||||
|
Net
sales
|
$ | 15,092,008 | $ | 15,092,008 | $ | 45,342,480 | $ | 45,342,480 | ||||||||
|
Cost
of sales
|
2,609,515 | 2,609,515 | 7,475,740 | 7,475,740 | ||||||||||||
|
Gross
profit
|
12,482,493 | 12,482,493 | 37,866,740 | 37,866,740 | ||||||||||||
|
Selling,
general and administrative expenses
|
9,465,589 | 9,202,873 | 28,471,469 | 28,208,753 | ||||||||||||
|
Interest
expense
|
260,574 | 381,700 | 416,882 | 538,008 | ||||||||||||
|
Operating
income
|
2,756,330 | 2,897,920 | 8,978,389 | 9,119,979 | ||||||||||||
|
Other
income
|
- | - | 18,864 | 18,864 | ||||||||||||
|
Finance
and non-operating income (expense)
|
(1,973,702 | ) | 829,773 | (1,995,794 | ) | 807,681 | ||||||||||
|
Income
before taxes
|
782,628 | 3,727,693 | 7,001,459 | 9,946,524 | ||||||||||||
|
Income
taxes
|
585,135 | 585,135 | 2,193,931 | 2,193,931 | ||||||||||||
|
Net
income
|
$ | 197,493 | $ | 3,142,558 | $ | 4,807,528 | $ | 7,752,593 | ||||||||
F-26
|
Balance
Sheet Data:
|
||||||||||||||||
|
March
31, 2010
|
June
30, 2009
|
|||||||||||||||
|
As
Previously
Reported
|
As
Restated
|
As
Previously
Reported
|
As
Restated
|
|||||||||||||
|
Current
assets
|
35,045,737 | 35,045,737 | $ | 28,357,010 | $ | 28,357,010 | ||||||||||
|
Other
non-current assets
|
25,219,222 | 25,219,222 | 25,447,999 | 25,447,999 | ||||||||||||
|
Deferred
fees on convertible notes
|
- | 1,898,877 | - | - | ||||||||||||
|
Total
assets
|
60,264,959 | 62,163,836 | 53,805,009 | 53,805,009 | ||||||||||||
|
Current
liabilities
|
9,240,124 | 8,554,324 | 20,248,229 | 20,248,229 | ||||||||||||
|
Derivative
liabilities – warrants
|
- | 5,323,640 | - | - | ||||||||||||
|
Convertible
notes, net
|
11,863,217 | 121,127 | - | - | ||||||||||||
|
Total
liabilities
|
21,103,341 | 13,999,091 | 20,248.229 | 20,248.229 | ||||||||||||
|
Stockholders’
Equity
|
||||||||||||||||
|
Additional
paid-in capital
|
3,378,203 | 9,553,666 | 2,974,520 | 2,974,520 | ||||||||||||
|
Retained
earnings
|
26,457,519 | 29,402,584 | 22,050,068 | 22,050,068 | ||||||||||||
| Accumulated other comprehensive income | 499,509 | 382,108 | 490,931 | 490,931 | ||||||||||||
|
Other
capital accounts
|
8,826,387 | 8,826,387 | 8,041,261 | 8,041,261 | ||||||||||||
|
Total
stockholders’ equity
|
39,161,618 | 48,164,745 | 33,556,780 | 33,556,780 | ||||||||||||
|
Total
liabilities and stockholders’ equity
|
$ | 60,264,959 | $ | 62,163,836 | $ | 53,805,009 | $ | 53,805,009 | ||||||||
The
table below summarizes the effects of the changes made to the Company’s
financial statements:
Statement of Income Data:
|
|
Three
Months
Ended
March
31, 2010
|
Nine
Months
Ended
March
31, 2010
|
||||||
|
Net
income, as previously reported
|
$ | 197,493 | $ | 4,807,528 | ||||
|
Adjustments:
|
||||||||
|
Deferred
fees
|
1,297,041 | 1,297,041 | ||||||
|
Interest
expense on convertible notes
|
(121,126 | ) | (121,126 | ) | ||||
|
Change
in warrant liabilities
|
1,769,150 | 1,769,150 | ||||||
|
Total
net income adjustment
|
2,945,065 | 2,945,065 | ||||||
|
Net
income, as restated
|
$ | 3,142,558 | $ | 7,752,593 | ||||
F-27
Balance Sheet Data:
|
March 31, 2010
|
June 30, 2009
|
|||||||
|
Total
assets, as previously reported
|
$ | 60,264,959 | $ | 53,805,009 | ||||
|
Deferred
fees on convertible notes, restated
|
1,898,877 | — | ||||||
|
Total
assets, as restated
|
62,163,836 | 53,805,009 | ||||||
|
Total
liabilities, as previously reported
|
21,103,341 | 20,248,229 | ||||||
|
Adjustments:
|
||||||||
|
Convertible
notes
|
(11,742,090 | ) | — | |||||
|
Warrant
liabilities, reclassified
|
685,800 | — | ||||||
|
Warrant
liabilities, restated
|
4,637,840 | — | ||||||
|
Total
liabilities adjustment
|
(7,104,250 | ) | — | |||||
|
Total
liabilities, as restated
|
13,999,091 | 20,248,229 | ||||||
|
Shareholders’
equity, as previously reported
|
39,161,618 | 33,556,780 | ||||||
|
Adjustments:
|
||||||||
|
Accumulated
other comprehensive income, restated
|
(117,401 | ) | — | |||||
|
Additional
paid-in capital - beneficial conversion feature,
restated
|
6,175,463 | — | ||||||
|
Net
income adjustment, restated
|
2,945,065 | — | ||||||
|
Net
Shareholders equity adjustment
|
9,003,127 | — | ||||||
|
Shareholders’
equity, as restated
|
$ | 48,164,745 | $ | 33,556,780 | ||||
F-28
REPORT
OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To: The
Board of Directors and Stockholders of Bohai Pharmaceuticals Group,
Inc.
We have
audited the accompanying consolidated balance sheets of Bohai Pharmaceuticals
Group, Inc. (the “Company”) as of June 30, 2009 and 2008 and the related
statements of income, stockholders' equity and cash flow for each of the two
years in the years ended June 30, 2009 and 2008. These financial statements are
the responsibility of the Company's management. Our responsibility is to express
an opinion on these financial statements based on our audits.
We
conducted our audit in accordance with standards of the Public Company
Accounting Oversight Board (United States). Those standards require that we plan
and perform the audit to obtain reasonable assurance about whether the financial
statements are free of material misstatement. An audit includes examining, on a
test basis, evidence supporting the amounts and disclosures in the financial
statements. An audit also includes assessing the accounting principles used and
significant estimates made by management, as well as evaluating the overall
financial statement presentation. We believe that our audits provide a
reasonable basis for our opinion.
In our
opinion, the consolidated financial statements referred to above present fairly,
in all material respects, the financial position of the Company as of June 30,
2009 and 2008, and the results of its operations and its cash flow for each of
the two years in the years ended June 30, 2009 and 2008, in conformity with
accounting principles generally accepted in the United States of
America.
|
s/ Parker Randall CF (H.K.) CPA
Limited
|
|
Parker
Randall CF (H.K.) CPA Limited
|
|
Certified
Public Accountants
|
|
Hong
Kong
|
|
26
February 2010
|
F-29
BOHAI
PHARMACEUTICALS GROUP, INC.
CONSOLIDATED
BALANCE SHEETS
AS
OF JUNE 30, 2009 AND 2008
(Stated
in US Dollars)
|
As
of
|
As
of
|
||||||||||
|
June
30,
|
June
30,
|
||||||||||
|
Notes
|
2009
|
2008
|
|||||||||
|
ASSETS
|
|||||||||||
|
Current
assets
|
|||||||||||
|
Cash
and cash equivalents
|
$ | 2,494,166 | $ | 829,050 | |||||||
|
Accounts
receivable
|
11,070,129 | 8,918,209 | |||||||||
|
Other
receivables and prepayments
|
4
|
12,993,800 | 10,557,082 | ||||||||
|
Amount
due from equity holder
|
5
|
1,465,000 | - | ||||||||
|
Inventories
|
6
|
307,834 | 1,049,191 | ||||||||
|
Total
current assets
|
28,330,929 | 21,353,532 | |||||||||
|
Non-current
assets
|
|||||||||||
|
Property,
plant and equipment, net
|
8
|
8,149,279 | 7,663,164 | ||||||||
|
Intangible
assets
|
7
|
17,298,720 | 17,227,872 | ||||||||
|
Total
non-current assets
|
25,447,999 | 24,891,036 | |||||||||
| 25,447,999 | 24,891,036 | ||||||||||
|
TOTAL
ASSETS
|
$ | 53,778,928 | $ | 46,244,568 | |||||||
|
LIABILITES
AND STOCKHOLDERS' EQUITY
|
|||||||||||
|
Current
liabilities
|
|||||||||||
|
Short-term
borrowings
|
9
|
$ | 5,860,000 | $ | 1,896,700 | ||||||
|
Accounts
payable
|
971,208 | 3,065,389 | |||||||||
|
Other
accrued liabilities
|
10
|
12,335,672 | 14,695,614 | ||||||||
|
Amount
due to equity holder
|
11
|
- | 108,426 | ||||||||
|
Income
taxes payable
|
677,666 | 598,190 | |||||||||
|
Total
current liabilities
|
19,844,546 | 20,364,319 | |||||||||
|
STOCKHOLDERS'
EQUITY
|
|||||||||||
|
Common
stock, $0.001 par value, 150,000,000 shares authorized,
|
|||||||||||
|
3,450,000
shares issued and outstanding
|
14
|
3,450 | 3,450 | ||||||||
|
Additional
paid-in capital
|
2,968,550 | 2,968,550 | |||||||||
|
Capital
reserve
|
5,836,000 | 5,836,000 | |||||||||
|
Accumulated
other comprehensive income
|
490,931 | 384,698 | |||||||||
|
Statutory
reserves
|
15
|
2,201,811 | 1,464,861 | ||||||||
|
Retained
earnings
|
22,433,640 | 15,222,690 | |||||||||
|
Total
stockholders' equity
|
33,934,382 | 25,880,249 | |||||||||
| 33,934,382 | 25,880,249 | ||||||||||
|
TOTAL
LIABILITIES AND SHAREHOLDERS' EQUITY
|
$ | 53,778,928 | $ | 46,244,568 | |||||||
See
accompanying notes to the financial statements
F-30
BOHAI
PHARMACEUTICALS GROUP, INC.
CONSOLIDATED
STATEMENT OF INCOME
FOR
THE YEARS ENDED JUNE 2009 AND 2008
(STATED
IN US DOLLARS)
|
Notes
|
2009
|
2008
|
|||||||||
|
Sales
|
$ | 50,170,014 | $ | 38,172,513 | |||||||
|
Less:
Sales Tax
|
(821,400 | ) | (629,489 | ) | |||||||
|
Net
sales
|
49,348,614 | 37,543,024 | |||||||||
|
Cost
of sales
|
(7,975,267 | ) | (5,950,680 | ) | |||||||
|
Gross
profit
|
41,373,347 | 31,592,344 | |||||||||
|
Selling,
general and administrative expenses
|
12
|
(31,347,139 | ) | (22,608,164 | ) | ||||||
|
Interest
expenses
|
13
|
(184,404 | ) | (234,101 | ) | ||||||
|
Operating
income
|
9,841,804 | 8,750,079 | |||||||||
|
Non-operating
income
|
49,447 | 8 | |||||||||
|
Non-operating
costs
|
(36,366 | ) | (835 | ) | |||||||
|
Income
before taxes
|
9,854,885 | 8,749,252 | |||||||||
|
Income
taxes
|
13
|
(1,906,985 | ) | (2,303,712 | ) | ||||||
|
Net
income
|
$ | 7,947,900 | $ | 6,445,540 | |||||||
See
accompanying notes to the financial statements
F-31
BOHAI
PHARMACEUTICALS GROUP CO., LTD.
STATEMENT
OF STOCKHOLDERS’ EQUITY
FOR
THE YEARS ENDED JUNE 30, 2009 AND 2008
(Stated
in US Dollars)
|
Common
stock
|
Additional
Paid-in
Capital
|
Capital
reserve
|
Accumulated
other
comprehensive
income
|
Statutory
reserves
|
Retained
earnings
|
Total
|
||||||||||||||||||||||
|
Balance,
July 1, 2007
|
$ | 3,450 | $ | 2,968,550 | $ | 5,836,000 | $ | - | $ | 885,025 | $ | 9,356,986 | $ | 19,050,011 | ||||||||||||||
|
Net
income for the year
|
- | - | - | - | 579,836 | 5,865,704 | 6,445,540 | |||||||||||||||||||||
|
Foreign
currency
|
||||||||||||||||||||||||||||
|
translation
adjustment
|
- | - | - | 384,698 | - | - | 384,698 | |||||||||||||||||||||
|
Balance,
June 30, 2008
|
$ | 3,450 | $ | 2,968,550 | $ | 5,836,000 | $ | 384,698 | $ | 1,464,861 | $ | 15,222,690 | $ | 25,880,249 | ||||||||||||||
|
Net
income for the year
|
- | - | - | - | 736,950 | 7,256,278 | 7,947,900 | |||||||||||||||||||||
|
Foreign
currency
|
||||||||||||||||||||||||||||
|
translation
adjustment
|
- | - | - | 106,233 | - | - | 106,233 | |||||||||||||||||||||
|
Balance,
June 30, 2009
|
$ | 3,450 | $ | 2,968,550 | $ | 5,836,000 | $ | 490,931 | $ | 2,201,811 | $ | 22,478,968 | $ | 33,934,382 | ||||||||||||||
See
accompanying notes to the financial statements
F-32
BOHAI
PHARMACEUTICALS GROUP, INC.
CONSOLIDATED
STATEMENT OF CASH FLOWS
FOR
THE YEAR ENDED JUNE 30, 2009 AND 2008
(Stated
in US Dollars)
|
2009
|
2008
|
|||||||
|
Cash
flows from operating activities
|
||||||||
|
Net
income
|
$ | 7,947,900 | $ | 6,445,540 | ||||
|
Adjustments
to reconcile net income to net cash provided
|
||||||||
|
by
operating activities
|
||||||||
|
Depreciation
|
302,762 | 283,417 | ||||||
|
Loss
on disposals of property, plant and equipment
|
31,630 | - | ||||||
|
Changes
in assets and liabilities
|
||||||||
|
Increase
in accounts receivable
|
(2,151,920 | ) | (1,885,681 | ) | ||||
|
(Increase)/decrease
in other receivables and prepayments
|
(2,436,718 | ) | 1,244,060 | |||||
|
Decrease
in inventories
|
741,357 | 655,446 | ||||||
|
Increase
in amount due from equity holder
|
(1,465,000 | ) | (108,426 | ) | ||||
|
(Decrease)/increase
in accounts payable
|
(2,094,181 | ) | 242,734 | |||||
|
Decrease
in accrued liabilities
|
(2,468,368 | ) | (4,678,508 | ) | ||||
|
Increase
in income taxes payable
|
79,476 | 160,490 | ||||||
|
Net
cash provided by operating activities
|
(1,513,062 | ) | 2,359,072 | |||||
|
Cash
flows from investing activities
|
||||||||
|
Purchases
of property, plant and equipment
|
(803,643 | ) | (80,647 | ) | ||||
|
Proceeds
from disposals of property, plant and equipment
|
14,650 | - | ||||||
|
Net
cash used in investing activities
|
(788,993 | ) | (80,647 | ) | ||||
|
Cash
flows from financing activities
|
||||||||
|
Proceeds
from sales of common stock
|
- | 54,000 | ||||||
|
Proceeds
of borrowings
|
5,860,000 | 1,896,700 | ||||||
|
Repayment
of borrowings
|
(1,896,700 | ) | (4,085,200 | ) | ||||
|
Net
cash provided by/(used in) financing activities
|
3,963,300 | (2,134,500 | ) | |||||
|
Net
increase in cash and cash equivalents
|
1,661,245 | 143,925 | ||||||
|
Effect
of foreign currency translation on cash and cash
equivalents
|
3,871 | 384,698 | ||||||
|
Cash
and cash equivalents at beginning of year
|
829,050 | 300,427 | ||||||
|
Cash
and cash equivalents at end of year
|
$ | 2,494,166 | $ | 829,050 | ||||
See
accompanying notes to the financial statements
F-33
BOHAI
PHARMACEUTICALS GROUP, INC.
FOR
THE YEAR ENDED JUNE 30, 2009 AND 2008
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
(Stated
in US Dollars)
|
1.
|
ORGANIZATION
AND PRINCIPAL ACTIVITIES
|
Bohai
Pharmaceuticals Group, Inc. (formerly known as Link Resources, Inc.) was
incorporated under the laws of the State of Nevada on January 9,
2008. Until January 5, 2010, its principal office was located in
Calgary, Alberta, Canada. The Company was a public “shell” company in
the exploration stage since its formation and had not yet realized any revenues
from its planned operations. The Company entered into a Mineral Lease
Agreement on April 1, 2008 for two mining claims in Pershing County, Nevada, in
an area known as the Goldbanks East Prospect. The Company terminated
the lease on July 7, 2009.
Pursuant
to a Share Exchange Agreement, dated January 5, 2010 (the “Share Exchange
Agreement” and the transactions contemplated thereby, the “Share Exchange”), the
Company acquired Chance High International Limited, a British Virgin Islands
company (“Chance High”) from Chance High’s shareholders (the “Chance High
Shareholders”) and, as a result, acquired Chance High’s indirect, controlled
subsidiary, Yantai Bohai Pharmaceuticals Group Co., Ltd. (“Bohai”), a Chinese
company engaged the production, manufacturing and distribution in the People’s
Republic of China (“China” or “the PRC”) of herbal medicines, including capsules
and other products, based on traditional Chinese medicine. The
closing of the Share Exchange (the “Closing”) took place on January 5, 2010 (the
“Closing Date”).
On the
Closing Date, pursuant to the terms of the Share Exchange Agreement, the Company
acquired all of the outstanding equity securities (the “Chance High Shares”) of
Chance High from the Chance High Shareholders, and the Chance High Shareholders
transferred and contributed all of their Chance High Shares to the
Company. In exchange, the Company issued to Chance High Shareholders
an aggregate of 13,162,500 newly issued shares of common stock, par value $0.001
per share (the “Common Stock”). In addition, pursuant to the terms of
the Share Exchange Agreement, Anthony Zaradic, the former President and Chief
Executive Officer of the Company, cancelled a total of 1,500,000 shares of
Common Stock.
Chance
High owns 100% of the issued and outstanding capital stock of the Yantai
Shencaojishi Pharmaceuticals Co., Ltd. (“WOFE”). On December 7, 2009,
the WOFE entered into a series of variable interest entity contractual
agreements (the “VIE Agreements”) with Bohai and its three shareholders,
including Mr. Hongwei Qu, currently the Company’s Chairman, Chief Executive
Officer and President (“Qu”), pursuant to which WOFE effectively assumed
management of the business activities of Bohai and has the right to appoint all
executives and senior management and the members of the board of directors of
Bohai. Chance High, WOFE and Bohai are referred to herein
collectively as the “Group”.
The VIE
Agreements are comprised of a series of agreements, including a Consulting
Services Agreement, Operating Agreement and Proxy Agreement, through which WOFE
has the right to advise, consult, manage and operate Bohai for an annual fee in
the amount of Bohai’s yearly net profits after tax. Additionally,
Bohai’s shareholders pledged their rights, titles and equity interest in Bohai
as security for WOFE to collect consulting and services fees provided to Bohai
through an Equity Pledge Agreement. In order to further reinforce
WOFE’s rights to control and operate Bohai, Bohai’s shareholders granted WOFE an
exclusive right and option to acquire all of their equity interests in Bohai
through an Option Agreement. As all of the companies in the Group are
under common control, this has been accounted for as a reorganization of
entities and the consolidated financial statements have been prepared as if the
reorganization had occurred retroactively under “as-if” basis which implies the
financial result of the Company prior to the date of Share Exchange Agreement
entered on January 5, 2010 has consolidated with the financial results of both
Chance High, WOFE and Bohai occurred at the beginning of the reporting period.
The Company has consolidated Bohai’s operating results, assets and liabilities
within its financial statements.
F-34
|
2.
|
BASIS
OF PRESENTATION
|
The Group
maintains its general ledger and journals with the accrual method accounting for
financial reporting purposes. Accounting policies adopted by the
Group conform to generally accepted accounting principles in the United States
of America (“US GAAP”) and have been consistently applied in the presentation of
financial statements, which are compiled on the accrual basis of
accounting.
This
basis of accounting differs in certain material respects from that used for the
preparation of the books of account of the Group, which are prepared in
accordance with the accounting principles and the relevant financial regulations
applicable to enterprises with limited liabilities established in the PRC (“PRC
GAAP”), the accounting standards used in the places of their
domicile. The accompanying financial statements reflect necessary
adjustments not recorded in the books of account of the Group to present them in
conformity with US GAAP.
The
consolidated financial statements as of and for the ended June 30, 2009 reflect
all adjustments which, in the opinion of management, are necessary to present
fairly the financial position, results of operations and cash flows for the
period presented in accordance with the accounting principles generally accepted
in the United States of America. All adjustments are of a normal
recurring nature.
|
3.
|
SUMMARY
OF SIGNIFICANT ACCOUNTING POLICIES
|
|
(a)
|
Basis
of Presentation and consolidation
|
The
condensed consolidated financial statements are prepared on the basis with
assumption the Share Exchange was undergone at the beginning of July 1,
2008. The historical consolidated financial statements of the Group
will be those of Bohai Pharmaceuticals Group, Inc. and of the consolidated
entities from the July 1, 2008, the deemed date of Share Exchange, and
subsequent. The consolidated financial statements for the Company for
the years ended June 30, 2009 and 2008, include the financial statements of
Chance High, and its wholly owned subsidiary, WOFE, and Bohai, the Company’s
principal operating subsidiary, which is a Chinese variable interest entity that
WFOE controls through certain contractual arrangements. Intercompany
transactions and balances are eliminated in consolidation.
The
Company has adopted FAS ASC 810-10-15-14 and also FIN 46R, which requires that a
Variable Interest Entity (“VIE”) to be consolidated by a company if that company
is entitled to receive a majority of the VIE’s residual returns and have direct
ability to made decision on all operation activities of the voting right of the
VIE.
The
Operating Agreement indicated that Bohai, as operating company, which is wholly
foreign owned under control of Chance High that empowers to WFOE the direct
ability to made decision on all operation activities of the voting right of
Bohai.
Under
Consultant Service Agreement entered between WFOE and Bohai on December 7, 2009,
Bohai is agreed to pay all of net income to WFOE quarterly as consultant fee.
Accordingly, WOFE has the right to receive the expected residual returns of
Bohai.
F-35
Under
above mentioned contractual arrangement, the Company, Bohai Pharmaceuticals
Group, Inc. qualifies as the primary beneficiaries such a controlling financial
interest of Bohai as operating company under FASB ASC230-10-45 and FASB
Interpretation No. 46R “Consolidation of Variable Interest Entities” (“FIN
46R”), an Interpretation of Accounting Research Bulletin No. 51. The result of
subsidiaries or variable interest entities acquired prior to date of Share
Exchange Agreement on January 5, 2010 entered are included in the consolidated
financial statement.
As of
December 31, 2009, the particulars of the Company’s subsidiaries are as
follows:
|
Name of
Company
|
Place of
incorporation
|
Date of
incorporation
|
Attributable
equity
interest
|
Issued capital
|
||||||||
|
Chance
High International Limited
|
British
Virgin Islands
|
July 02,
2009
|
100 | % |
USD50,000
|
|||||||
|
Yantai
Shencaojishi Pharmaceuticals Co., Ltd.
|
People’s
Republic of China
|
November
25, 2009
|
100 | % |
USD9,500,000
|
|||||||
|
Yantai
Bohai Pharmaceuticals Group Co., Ltd.
|
People’s
Republic of China
|
July
8, 2004
|
* |
USD2,918,000
(RMB20,000,000)
|
||||||||
* The
Group has indirect controlling interest of Bohai under the VIE Agreements
entered on December 7, 2009, which are described in Note 1 above.
|
(b)
|
Economic
and Political Risks
|
The
Group’s operations are conducted solely in the PRC. There are
significant risks associated with doing business in the PRC, among others,
political, economic, legal and foreign currency exchange risks. The
Group’s results may be adversely affected by changes in the political and social
conditions in the PRC, and by changes in governmental policies with respect to
laws and regulations, anti-inflationary measures, currency conversion,
remittances abroad, and rates and methods of taxation, among other
things.
|
(c)
|
Use
of Estimates
|
In
preparing of the consolidated financial statements in conformity with US GAAP,
management makes estimates and assumptions that affect the reported amounts of
assets and liabilities and disclosures of contingent assets and liabilities at
the dates of the financial statements, as well as the reported amounts of
revenues and expenses during the reporting periods. These accounts
and estimates include, but are not limited to, the valuation of accounts
receivable, inventories, deferred income taxes and the estimation on useful
lives of plant and machinery. Actual results could differ from those
estimates.
F-36
|
(d)
|
Cash
and Cash Equivalents
|
The Group
considers all highly liquid investments purchased with original maturities of
three months or less to be cash equivalents. The Group maintains bank
accounts only in the PRC. The Company does not maintain any bank
accounts in the United States of America. As of December 31, 2009 and
June 30, 2009, there were cash and cash equivalents of $7,892,908 and $2,493,510
respectively.
|
(e)
|
Accounts
Receivable
|
Accounts
receivable consists of amounts due from customers. The Group extends unsecured
credit to its customers in the ordinary course of business but mitigates the
associated risks by performing credit checks and actively pursuing past due
accounts. An allowance for doubtful accounts is established and
determined based on management’s assessment of known requirements, aging of
receivables, payment history, the customer’s current credit worthiness and the
economic environment.
|
(f)
|
Inventories
|
Inventories
are valued at the lower of cost or market with cost is determined on the
weighted average method. Finished goods inventories consist of raw materials,
direct labour and overhead associated with the manufacturing process. In
assessing the ultimate realization of inventories, the management makes
judgments as to future demand requirements compared to current or committed
inventory levels. The Group’s reserve requirements generally
increase/decrease due to management projected demand requirements, market
conditions and product life cycle changes. As of December 31, 2009 and June 30,
2009, the Group did not make any allowance for slow-moving or defective
inventories.
|
(g)
|
Fair
value of Financial Instruments
|
The
carrying values of the Group’s financial instruments, including cash and cash
equivalents, accounts receivables, other receivables and prepayments, short-term
borrowings, accounts payables, and other accrued liabilities their fair values
due to the short-term maturity of such instruments.
|
(h)
|
Intangible
Assets
|
Intangible
assets as “Pharmaceutical Formulas”, which acquired and with indefinite useful
live are measured initially at cost and not subject to amortization shall be
tested for impairment annually or more frequently if there is indication of
impairment. If the carrying amount exceeds fair value, an impairment loss should
be recognized. Subsequently reversal of a recognized impairment loss is
prohibited.
|
(i)
|
Property,
plant and equipment
|
Property,
plant and equipment, other than construction in progress, are stated at cost
less depreciation and amortization and accumulated impairment
loss. Cost represents the purchase price of the asset and other costs
incurred to bring the asset into its existing use. Maintenance,
repairs and betterments, including replacement of minor items, are charged to
expense; major additions to physical properties are capitalized.
Depreciation
of property, plant and equipment is calculated to written off the cost, less
their estimated residual value, if any, using the straight-line method over
their estimated useful lives. The principal annual rates are as
follows:-
F-37
|
Leasehold
land and buildings
|
30 to 40 years
|
|
Motor
vehicles
|
10 years
|
|
Plant
and machinery
|
10 years
|
|
Office
equipment
|
5 years
|
Upon sale
or disposition, the applicable amounts of asset cost and accumulated
depreciation are removed from the accounts and the net amount less proceeds from
disposal is charged or credited to income.
Construction
in progress mainly represents expenditures in respect of the Group’s corporate
campus and machinery under construction, which the campus was completed and at
which production began in 2009. Assets under construction are not
depreciated until the construction is completed and the assets are ready for
their intended use.
|
(j)
|
Accounting
for the Impairment of Long-Lived
Assets
|
The Group
adopted Statement of Financial Accounting Standards No. 144, “Accounting for the
Impairment or Disposal of Long-Live Assets” (“SFAS 144”), which addresses
financial accounting and reporting for the impairment or disposal of long-lived
assets. The Group periodically evaluates the carrying value of
long-lived assets to be held and used in accordance with SFAS 144. SFAS 144
requires impairment losses to be recorded on long-lived assets used in
operations when indicators of impairment are present and the undiscounted cash
flows estimated to be generated by those assets are less than the assets’
carrying amounts. In that event, a loss is recognized based on the amount by
which the carrying amount exceeds the fair market value of the long-lived
assets. Loss on long-lived assets to be disposed of is determined in a similar
manner, except that fair market values are reduced for the cost of disposal.
Based on its review, the Group believes that, as of December 31, 2009 and June
30, 2009, there were no significant impairments of its long-lived
assets.
|
(k)
|
Foreign
Currency Translation
|
The Group
maintains its financial statements in the functional currency. The
functional currency of the Group is the Chinese Renminbi
(RMB). Monetary assets and liabilities denominated in currencies
other than the functional currency are translated into the functional currency
at rates of exchange prevailing at the balance sheet
dates. Transactions denominated in currencies other than the
functional currency are translated into the functional currency at the exchanges
rates prevailing at the dates of the transaction. Exchange gains or
losses arising from foreign currency transactions are included in the
determination of net income for the respective periods.
For
financial reporting purposes, the consolidated financial statements of the Group
which are prepared using the functional currency have been translated into
United States dollars. Assets and liabilities are translated at the exchange
rates at the balance sheet dates and revenue and expenses are translated at the
average exchange rates and stockholders’ equity is translated at historical
exchange rates. Any translation adjustments resulting are not included in
determining net income but are included in foreign exchange adjustment to other
comprehensive income, a component of stockholders’ equity.
| 6.30.2009 | 6.30.2008 | |||||||
|
Period
end US$ : RMB exchange rate
|
6.84480 | 6.87180 | ||||||
|
Average
periodic US$ : RMB exchange rate
|
6.84819 | 7.29062 |
F-38
RMB is
not freely convertible into foreign currency and all foreign exchange
transactions must take place through authorized institutions. No
representation is made that the RMB amounts could have been, or could be,
converted into US$ at the rates used in translation.
|
(l)
|
Revenue
Recognition
|
Revenue
represents the invoiced value of goods sold recognized upon the delivery of
goods to customers. Revenue is recognized when all of the following criteria are
met:
|
|
·
|
Persuasive
evidence of an arrangement exists;
|
|
|
·
|
Delivery
has occurred or services have been
rendered;
|
|
|
·
|
The
seller’s price to the buyer is fixed or determinable;
and
|
|
|
·
|
Collectability
is reasonably assured. Payments have been
established.
|
The
Company’s return policy of goods and services is recognized in actual basis when
the goods sold are returned by customers and the closing balance of revenue is
actually deducted by the relevant cost of sales. The
sales return rate is very low and management believes that the return
amount is immaterial and therefore no such provision should be
stated.
|
(m)
|
Cost
of Revenue
|
Regarding
the trading of medicine, the respective cost of revenue consists primarily of
material cost, labour cost, overhead associated with the manufacturing process
and related expenses which are directly attributable to the
trading.
|
(n)
|
Research
and Development Costs
|
Research
and development costs are charged as expense when incurred and included in
operating expenses.
|
(o)
|
Income
Taxes
|
Income
taxes are accounted for under the asset and liability
method. Deferred tax assets and liabilities are recognized for the
future tax consequences attributable to differences between the financial
statement carrying amounts of existing assets and liabilities and their
respective tax bases and operating loss and tax credit
carry-forwards. Deferred tax assets are reduced by a valuation
allowance to the extent management concludes it is more likely than not that the
assets will not be realized. Deferred tax assets and liabilities are
measured using enacted tax rates expected to apply to taxable income in the
years in which those temporary differences are expected to be recovered or
settled. The effect on deferred tax assets and liabilities of a
change in tax rates is recognized in the statements of income and comprehensive
income in the periods that includes the enactment date.
|
(p)
|
Comprehensive
Income
|
Comprehensive
income is defined to include all changes in equity except those resulting from
investments by owners and distributions to owners. Among other
disclosures, all items that are required to be recognized under current
accounting standards as components of comprehensive income are required to be
reported in a financial statement that is presented with the same prominence as
other financial statements. The Group’s current components of other
comprehensive income are the foreign currency translation
adjustment.
F-39
|
(q)
|
Commitments
and Contingencies
|
Liabilities
for loss contingencies arising from claims, assessments, litigation, fines and
penalties and other sources are recorded when it is probable that a liability
has been incurred and the amount of the assessment can be reasonably
estimated.
|
(r)
|
Earning
per Shares
|
The Group
reports basic earnings per share in accordance with SFAS 128, “Earnings Per
Share”. Basic earnings/(loss) per share is computed by dividing net
income/(loss) by weighted average number of shares of common stock outstanding
during the period. Diluted earnings per share is computed by dividing net income
by the weighted average number of shares of common stock, common stock
equivalents and potentially dilutive securities outstanding during the period.
At June 30, 2009, the Group had no common stock equivalents that could
potentially dilute future earnings per share.
|
(s)
|
Recent
Accounting Pronouncements
|
In March
2008, the FASB issued SFAS No. 161, DISCLOSURES ABOUT DERIVATIVE INSTRUMENTS AND
HEDGING ACTIVITIES (an amendment to SFAS No. 133). This statement is effective
for financial statements issued for fiscal year and interim periods beginning
after November 15, 2008 and requires enhanced disclosures with respect to
derivative and hedging activities. The Group will comply with the disclosure
requirements of this statement if it utilizes derivative instruments or engages
in hedging activities upon its effectiveness.
In April
2008, the FASB issued FASB Staff Position No. 142-3, DETERMINATION OF THE USEFUL
LIFE OF INTANGIBLE ASSETS (“FSP No. 142-3”) to improve the consistency between
the useful life of a recognized intangible asset (under SFAS No. 142) and the
period of expected cash flows used to measure the fair value of the intangible
asset (under SFAS No. 141(R)). FSP No. 142-3 amends the factors to be considered
when developing renewal or extension assumptions that are used to estimate an
intangible asset’s useful life under SFAS No. 142. The guidance in the new staff
position is to be applied prospectively to intangible assets acquired after
December 31, 2008. In addition, FSP No.142-3 increases the disclosure
requirements related to renewal or extension assumptions. The Company does not
believe implementation of FSP No. 142-3 have a material impact on its
consolidated financial statements.
In May
2008, the FASB issued statement No. 162, THE HIERARCHY OF GENERALLY ACCEPTED
ACCOUNTING PRINCIPLES. This statement identifies the sources of accounting
principles and the framework for selecting the principles to be used in the
preparation of financial statements of nongovernmental entities that are
presented in conformity with generally accepted accounting principles (GAAP) in
the United States (the GAAP hierarchy). This statement is effective 60 days
following the SEC’s approval of the Public Company Accounting Oversight Board
amendments to AU Section 411, “the Meaning of Present Fairly in Conformity With
Generally Accepted Accounting Principles”.
In May
2008, the FASB issued FSP Accounting Principles Board (“APB”) 14-1 “Accounting
for Convertible Debt instruments That May Be Settled in Cash upon Conversion
(Including Partial Cash Settlement)” (“FSP APB 14-1”). FSP APB 14-1 requires the
issuer of certain convertible debt instruments that may be settled in cash (or
other assets) on conversion to separately account for the liability (debt) and
equity (conversion option) components of the instrument in a manner that
reflects the issuer’s non-convertible debt borrowing rate. FSP APB 14-1 is
effective for fiscal years beginning after December 15, 2008 on a retroactive
basis. As we do not have convertible debt at this time, we currently believe the
adoption of FSP APB 14-1 will have no effect on our combined results of
operations and financial condition.
F-40
In May
2008, the FASB issued Statement No. 163, ACCOUNTING FOR FINANCE GUARANTEE
INSURANCE CONTRACTS - AN INTERPRETATION OF FASB STATEMENT NO. 60. The premium
revenue recognition approach for a financial guarantee insurance contract links
premium revenue recognition to the amount of insurance protection and the period
in which it is provided. For purposes of this statement, the amount of insurance
protection provided is assumed to be a function of the insured principal amount
outstanding, since the premium received requires the insurance enterprise to
stand ready to protect holders of an insured financial obligation from loss due
to default over the period of the insured financial obligation. This Statement
is effective for financial statements issued for fiscal years beginning after
December 15, 2008.
In June
2008, the FASB issued FASB Staff Position Emerging Issues Task Force (EITF) No.
03-6-1, DETERMINING WHETHER INSTRUMENTS GRANTED IN SHARE-BASED PAYMENT
TRANSACTIONS ARE PARTICIPATING SECURITIES (“FSP EITF No. 03-6-1”). Under FSP
EITF No. 03-6-1, unvested share-based payment awards that contain rights to
receive nonforfeitable dividends (whether paid or unpaid) are participating
securities, and should be included in the two-class method of computing EPS. FSP
EITF No. 03-6-1 is effective for fiscal years beginning after December 15, 2008,
and interim periods within those years, and is not expected to have a
significant impact on the Group’s consolidated financial
statements.
In April
2009, the FASB issued FSP 157-4, DETERMINING FAIR VALUE WHEN THE VOLUME AND
LEVEL OF ACTIVITY FOR THE ASSET OR LIABILITY HAVE SIGNIFICANTLY DECREASED AND
IDENTIFYING TRANSACTIONS THAT ARE NOT ORDERLY (“FSP 157-4”). FSP 157-4 provides
additional guidance for estimating fair value in accordance with SFAS 157 when
the volume and level of activity for the asset or liability have significantly
decreased. FSP 157-4 also includes guidance on identifying circumstances that
indicate a transaction is not orderly. FSP 157-4 is effective for interim and
annual reporting periods ending after June 15, 2009, with early adoption
permitted for periods ending after March 15, 2009. FSP 157-4 does not require
disclosures for earlier periods presented for comparative purposes at initial
adoption. In periods after initial adoption, FSP 157-4 requires comparative
disclosures only for periods ending after initial adoption. The adoption of the
provisions of FSP 157-4 is not anticipated to materially impact on the Company’s
results of operations or the fair values of its assets and
liabilities.
In May
2009, the FASB issued SFAS No. 165, SUBSEQUENT EVENTS (“SFAS 165”). SFAS 165
establishes general standards for accounting for and disclosure of events that
occur after the balance sheet date but before financial statements are issued or
available to be issued and was effective for interim and annual periods ending
after June 15, 2009. The adoption of SFAS No. 165 did not have an impact on the
Company’s results of operations or financial condition. The Company evaluated
all subsequent events that occurred from January 1, 2010 through January 30,
2010, inclusive, and does not found any material subsequent events are required
to disclose.
In June
2009, the FASB issued SFAS No. 166 ACCOUNTING FOR TRANSFERS OF FINANCIAL ASSETS
(“SFAS 166). This statement is intended to improve the relevance,
representational faithfulness, and comparability of the information that a
reporting entity provides in its financial reports about a transfer of financial
assets; the effects of a transfer on its financial position, financial
performance, and cash flows; and a transferor’s continuing involvement in
transferred financial assets. This Statement must be applied as of the beginning
of each reporting entity’s first annual reporting period that begins after
November 15, 2009, and is required to be adopted by the Company in the first
quarter of fiscal year 2011. Earlier application is prohibited. This Statement
must be applied to transfers occurring on or after the effective date. The Group
does not expect the adoption of SFAS 166 to have a material impact on the
Group’s financial position, results of operations and cash
flows.
F-41
In June
2009, the FASB issued SFAS No.167, “Amendments to FASB Interpretation No.46(R)”,
which is codified as ASC 810. ASC 810 amends FASB Interpretation No.46(R),
“Variable Interest Entities” for determining whether an entity is a variable
interest entity (“VIE”) and requires an enterprise to perform an analysis to
determine whether the enterprise’s variable interest or interests give it a
controlling financial interest in a VIE. Under ASC 810, an enterprise has a
controlling financial interest when it has a) the power to direct the activities
of a VIE that most significantly impact the entity’s economic performance and b)
the obligation to absorb losses of the entity or the right to receive benefits
from the entity that could potentially be significant to the VIE. ASC 810 also
requires an enterprise to assess whether it has an implicit financial
responsibility to ensure that a VIE operates as designed when determining
whether it has power to direct the activities of the VIE that most significantly
impact the entity’s economic performance. ASC 810 also requires ongoing
assessments of whether an enterprise is the primary beneficiary of a VIE,
requires enhanced disclosures and eliminates the scope exclusion for qualifying
special-purpose entities. ASC 810 shall be effective as of the beginning of each
reporting entity’s first annual reporting period that begins after November 15,
2009, for interim periods within that first annual reporting period, and for
interim and annual reporting periods thereafter. Earlier application is
prohibited. ASC 810 is effective for the Company in the first quarter of fiscal
2011. The Group is currently evaluating the effect of ASC 810 on its
consolidated financial statements and results of operation and is currently not
yet in a position to determine such effects.
In August
2009, the FASB issued Accounting Standards Update (“ASU”) No. 2009-05,
“Measuring Liabilities at Fair Value”, which is codified as ASC 820, “Fair Value
Measurements and Disclosures”. This Update provides amendments to ASC 820-10,
Fair Value Measurements and Disclosures –Overall, for the fair value measurement
of liabilities. This Update provides clarification that in circumstances in
which a quoted price in an active market for the identical liability is not
available, a reporting entity is required to measure fair value using a
valuation technique that uses the quoted price of the identical liability when
traded as an asset, quoted prices for similar liabilities or similar liabilities
when traded as assets, or that is consistent with the principles of ASC 820. The
amendments in this Update also clarify that when estimating the fair value of a
liability, a reporting entity is not required to include a separate input or
adjustment to other inputs relating to the existence of a restriction that
prevents transfer of the liability. The amendments in this Update also clarify
that both a quoted price in an active market for the identical liability at the
measurement date and the quoted price for the identical liability when traded as
an asset in an active market when no adjustments to the quoted price of the
assets are required are Level 1 fair value measurements. ASC 820 is effective
for the first reporting period (including interim periods) beginning after
August 28, 2009. The adoption of this Update did not have a significant impact
to the Company’s financial statements.
In
December 2009, the FASB issued ASU No. 2009-17, “Improvements to Financial
Reporting by Enterprises Involved with Variable Interest Entities (“ASU
2009-17”)”. ASU 2009-17 amends the variable-interest entity guidance in FASB ASC
810-10-05-8 to clarify the accounting treatment for legal entities in which
equity investors do not have sufficient equity at risk for the entity to finance
its activities without financial support. ASU 2009-17 shall be effective as of
the beginning of each reporting entity’s first annual reporting period that
begins after November 15, 2009. ASU 2009-17 is effective for the Company in the
first quarter of fiscal 2011. The Company is currently evaluating the
effect of ASU 2009-17 on its consolidated financial statements and results of
operation and is currently not yet in a position to determine such
effects.
None of
the above new pronouncements has current application to the Group, but may be
applicable to the Group’s future financial reporting.
F-42
|
4.
|
OTHER
RECEIVABLES AND PREPAYMENTS
|
Other
receivables and prepayments consisted of the following :
|
As
of
|
As
of
|
|||||||
|
June
30,
|
June
30,
|
|||||||
|
2009
|
2008
|
|||||||
|
Prepayment
for advertising and promotion
|
$ | 1,736,025 | $ | 45,958 | ||||
|
Loan
to a third party
|
1,465,000 | - | ||||||
|
Loans
interest receivable
|
18,459 | - | ||||||
|
Other
receivables
|
9,774,316 | 10,511,124 | ||||||
| $ | 12,993,800 | $ | 10,557,082 | |||||
Loan to a
third party is unsecured, interest bearing at 4.32% per annum and repayable on
November 11, 2009.
|
5.
|
AMOUNT
DUE FROM EQUITY HOLDER
|
|
As
of
|
As
of
|
|||||||
|
June
30,
|
June
30,
|
|||||||
|
2009
|
2008
|
|||||||
|
Mr.
Qu Hong Wei
|
$ | 1,465,000 | $ | - | ||||
The
amount due from an equity holder is unsecured, interest bearing at 3.93% per
annum and has no fixed term of repayment.
|
6.
|
INVENTORIES
|
Inventories
consisted of the following :
|
As
of
|
As
of
|
|||||||
|
June
30,
|
June
30,
|
|||||||
|
2009
|
2008
|
|||||||
|
Raw
materials
|
$ | 250,405 | $ | 716,303 | ||||
|
Finished
goods
|
57,429 | 332,888 | ||||||
|
Total
inventories
|
$ | 307,834 | $ | 1,049,191 | ||||
F-43
|
7.
|
INTANGIBLE
ASSETS
|
Intangible
assets consisted of the following:
|
As
of
|
As
of
|
|||||||
|
June
30,
|
June
30,
|
|||||||
|
2009
|
2008
|
|||||||
|
Pharmaceuticals
formulas, at cost
|
$ | 17,298,720 | $ | 17,227,872 | ||||
|
8.
|
PROPERTY,
PLANT AND EQUIPMENT, NET
|
Property,
plant and equipment consisted of the following :
|
As
of
|
As
of
|
|||||||
|
June
30,
|
June
30,
|
|||||||
|
2009
|
2008
|
|||||||
|
Cost
|
||||||||
|
Leasehold
land and buildings
|
$ | 7,447,211 | $ | 7,416,710 | ||||
|
Plant
and equipment
|
1,156,557 | 988,491 | ||||||
|
Office
equipment
|
74,700 | 15,179 | ||||||
|
Motor
vehicles
|
389,075 | 277,210 | ||||||
|
Total
|
9,067,543 | 8,697,590 | ||||||
|
Accumulated
depreciation
|
||||||||
|
Leasehold
land and buildings
|
414,133 | 229,132 | ||||||
|
Plant
and equipment
|
617,279 | 571,539 | ||||||
|
Office
equipment
|
10,681 | 5,956 | ||||||
|
Motor
vehicles
|
112,768 | 248,292 | ||||||
|
Total
|
1,154,861 | 1,054,919 | ||||||
|
Net
|
||||||||
|
Leasehold
land and buildings
|
7,033,078 | 7,187,578 | ||||||
|
Plant
and equipment
|
775,875 | 437,445 | ||||||
|
Office
equipment
|
64,019 | 9,223 | ||||||
|
Motor
vehicles
|
276,307 | 28,918 | ||||||
|
Total
|
8,149,279 | 7,663,164 | ||||||
|
Construction
in progress
|
236,597 | 20,493 | ||||||
|
Property,
plant and equipment, net
|
$ | 8,149,279 | $ | 7,663,164 | ||||
F-44
Depreciation
expense for the year ended June 30, 2009 was $302,762 and for the year ended
June 30, 2008, was $283,417 respectively.
As of
June 30, 2009 and June 30, 2008, the Group has pledged plant and machinery
having a carrying amount of $562,331 and $Nil to secure a bank loan to
Bohai.
|
9.
|
SHORT-TERM
BORROWINGS
|
The
Company obtained several short-term loan facilities from financial institution
in the PRC.
Short-term
borrowings as of June 30, 2009 consisted of the following :
|
Loan
from
|
Annual
|
||||||||||
|
financial
institution
|
Loan
period
|
Interest
rate
|
Secured
by
|
Amount
|
|||||||
|
Shanghai
Pudong
|
From
Dec 12, 2008
|
6.6960 | % |
Haiyang
Construction
|
$ | 2,197,500 | |||||
|
Development
Limited
|
to
Dec 11, 2009
|
Industry
Training Centre
|
|||||||||
|
and
personal guarantee
|
|||||||||||
|
by
equity holders
|
|||||||||||
|
Yantai
City
|
From
Jan 20, 2009
|
6.9030 | % |
Yantai
Hai Pu Can End
|
1,318,500 | ||||||
|
Commercial
Bank
|
to
Jan 20, 2010
|
Making
Co. Ltd
|
|||||||||
|
Yantai
Laishan Rural
|
From
Sep 27, 2008
|
9.3600 | % |
Yantai
Ka Wah Medical
|
293,000 | ||||||
|
Credit
Union
|
to
Sep 26, 2009
|
Equipment
Co. Ltd
|
|||||||||
|
Yantai
Laishan Rural
|
From
Sep 27, 2008
|
12.2400 | % |
Company's
machinery and
|
586,000 | ||||||
|
Credit
Union
|
to
Sep 26, 2009
|
vehicle
|
|||||||||
|
China
Construction
|
From
May 12, 2009
|
0.0000 | % |
Personal
guarantee by
|
1,465,000 | ||||||
|
Bank
|
to
Nov 11, 2009
|
equity
holders
|
|
||||||||
| $ | 5,860,000 | ||||||||||
Short-term
borrowings as of June 30, 2008 consisted of the following :
F-45
|
Loan
from
|
Annual
|
||||||||||
|
financial
institution
|
Loan
period
|
Interest
rate
|
Secured
by
|
Amount
|
|||||||
|
Yantai
City
|
From
Jan 31, 2008
|
8.0925 | % |
Yantai
Hai Pu Can End
|
$ | 1,313,100 | |||||
|
Commercial
Bank
|
to
Jan 20, 2009
|
Making
Co. Ltd
|
|||||||||
|
Industrial
and Commercial
|
From
Jan 25, 2008
|
7.4700 | % |
Shan
Dong Shui Hong
|
583,600 | ||||||
|
Bank
of China
|
to
Jan 24, 2009
|
Pharmaceutical
Co. Ltd
|
|
||||||||
| $ | 1,896,700 | ||||||||||
|
10.
|
OTHER
ACCRUED LIABILITIES
|
Other
accrued liabilities consisted of the following :
|
As
of
|
As
of
|
|||||||
|
June
30,
|
June
30,
|
|||||||
|
2009
|
2008
|
|||||||
|
Advanced
from customers
|
$ | - | $ | 735,680 | ||||
|
Accrued
selling expenses
|
1,677,026 | 321,717 | ||||||
|
Accrued
staff costs
|
173,130 | 200,135 | ||||||
|
Value
added tax payable
|
709,688 | 541,152 | ||||||
|
Other
taxes payable
|
77,374 | 58,863 | ||||||
|
Payable
to Pharmaceutical formulas vendors
|
- | 2,724,087 | ||||||
|
Other
accrued expenses
|
9,702,137 | 10,113,980 | ||||||
|
|
||||||||
| $ | 12,339,355 | $ | 14,695,614 | |||||
|
11.
|
AMOUNT
DUE TO EQUITY HOLDER
|
|
As
of
|
As
of
|
|||||||
|
June
30,
|
June
30,
|
|||||||
|
2009
|
2009
|
|||||||
|
Mr.
Qu Hong Wei
|
$ | - | $ | 108,426 | ||||
The
amount due to equity is interest-free, unsecured and repayable on
demand.
F-46
|
12.
|
SELLING,
GENERAL AND ADMINISTRATIVE EXPENSES
|
Selling,
general and administrative expenses consisted of the followings :
|
Year
ended
|
Year
ended
|
|||||||
|
June
30,
|
June
30,
|
|||||||
|
2009
|
2008
|
|||||||
|
Accommodation
|
$ | 5,151,876 | $ | 4,593,639 | ||||
|
Advertising
and promotion
|
10,959,424 | 8,679,512 | ||||||
|
Audit
fee
|
8,937 | 1,928 | ||||||
|
Commission
|
1,419,478 | 135,453 | ||||||
|
Conference
|
5,089,283 | 3,877,918 | ||||||
|
Depreciation
|
20,514 | 9,033 | ||||||
|
Staff
costs
|
1,536,496 | 998,763 | ||||||
|
Travelling
|
2,755,259 | 1,649,162 | ||||||
|
Research
and development cost
|
293,000 | 344 | ||||||
|
Other
operating expenses
|
4,112,872 | 2,662,412 | ||||||
| $ | 31,347,139 | $ | 22,608,164 | |||||
|
13.
|
INTEREST
EXPENSES
|
|
Year
ended
|
Year
ended
|
|||||||
|
June
30,
|
June
30,
|
|||||||
|
2009
|
2008
|
|||||||
|
Interest
on short-term bank borrowings wholly repayable within one
year
|
$ | 184,404 | $ | 234,101 | ||||
F-47
|
14.
|
INCOME
TAXES
|
The
components of the provision for income taxes are:
|
Year
ended
|
Year
ended
|
|||||||
|
June
30,
|
June
30,
|
|||||||
|
2009
|
2008
|
|||||||
|
Current
taxes
|
||||||||
|
United
States
|
$ | - | $ | - | ||||
|
PRC
|
1,906,985 | 2,303,712 | ||||||
| $ | 1,906,985 | $ | 2,303,712 | |||||
United States
Tax
The
Company is subject to income tax in the United States. No provision for income
tax in the United States has been made as the Company had no taxable income for
the years ended June 30, 2009 and 2008. The statutory tax rate is
34%.
A
reconciliation of the effective income tax rate to the US federal rate is as
follows:
|
2009
|
2008
|
|||||||
|
United
States Tax at statutory rate
|
$ | 3,350,661 | $ | 2,974,746 | ||||
|
Foreign
tax rate difference
|
(886,940 | ) | (787,433 | ) | ||||
|
Exemption
in taxation
|
(2,463,721 | ) | (2,187,313 | ) | ||||
| $ | - | $ | - | |||||
As of
June 30, 2009, the Company incurred $45,328 of net operating losses carry
forwards available for federal tax purposes that may be used to offset future
taxable income and will begin to expire in 2028, if unutilized. The Company has
provided for a full valuation allowance against the deferred tax assets of
$15,412 on the expected future tax benefits from the net operating loss carry
forwards as the management believes it is more likely than not that these assets
will not be realized in the future.
PRC Tax
PRC's
legislative body, the National People's Congress, adopted the unified Enterprise
Income Tax ("EIT") Law on March 16, 2007. This new tax law replaces the existing
separate income tax laws for domestic enterprises and foreign-invested
enterprises and became effective on January 1, 2008. Under the new tax law, a
unified income tax rate is set at 25% for both domestic enterprises and
foreign-invested enterprises. However, there will be a transition period for
enterprises, whether foreign-invested or domestic, that are currently receiving
preferential tax treatments granted by relevant tax authorities. Enterprises
that are subject to an enterprise income tax rate lower than 25% may continue to
enjoy the lower rate and will transit into the new rate over a five year period
beginning on the effective date of the EIT Law. Enterprises that are currently
entitled to exemptions for a fixed term may continue to enjoy such treatment
until the exemption term expires. Preferential tax treatments may continue to be
granted to industries and projects that qualify for such preferential treatments
under the new law.
F-48
Certain
of the Group's revenue (including sales revenue) are subject to output VAT
generally calculated at 6%, 7%, 13% and 17% of the selling price. Input credit
relating to input VAT paid on purchase can be used to offset the output
VAT.
The net
amount of VAT expense for the years are shown as follows:
|
2009
|
2008
|
|||||||
|
Net
value added tax expense
|
$ | 7,443,011 | $ | 5,700,705 | ||||
The
deferred tax asset and liability has not been recognized because of no valuation
allowance to be established as of June 30, 2009 and June 30, 2008.
|
15.
|
COMMITMENTS
AND CONTINGENCIES
|
There is
no foreseeable commitments or contingencies for the year ended June 30, 2009 and
June 30, 2008.
|
16.
|
SHARE
CAPITAL
|
As of
June 30, 2009, the Company has authorized 75,000,000 of $0.001 par common stock,
of which 3,450,000 shares were issued and outstanding.
|
17.
|
STATUTORY
RESERVES
|
According
to the laws and regulations in the PRC, Bohai is required to provide
for certain statutory funds, namely, reserve fund by an appropriation from net
profit after taxes but before dividend distribution based on the local statutory
financial statements of the PRC company prepared in accordance with the
accounting principles and relevant financial regulations.
Bohai in
PRC is required to allocate at least 10% of its net profit to the reserve fund
until the balance of such fund has reached 50% of its registered capital.
Appropriation of enterprise expansion fund are determined at the discretion of
it directors.
The
reserve fund can only be used, upon approval by the relevant authority, to
offset accumulated losses or increase capital. The enterprise expansion fund can
only be used to increase capital upon approval by the relevant
authority.
|
18.
|
FINANCIAL
INSTRUMENTS
|
|
(a)
|
Credit
risk
|
The
potential credit risk of the company is mainly attributable to its debtors and
bank balances. In respect of debtors, the Group has policies in place
to ensure that it will only accept customers from countries which are
politically stable and customers with an appropriate credit
history. In addition, all the bank balances were made with financial
institutions with high-credit quality. Thus, the Group is not
considered to be subject to significant credit risk.
F-49
|
(b)
|
Interest
rate risk
|
The
Group's interest rate risk is primarily attributable to its short-term
borrowings, loan to a third party and loan to equity holders. The Group’s
borrowings carry interest at fixed rate. The management has not used any
interest rate swaps to hedge its exposure to interest rate risk.
|
(c)
|
Fair
value estimation
|
All of
the carrying amounts of the Group's financial assets and liabilities of short
term maturities approximate their fair values.
|
19.
|
SUBSEQUENT
EVENTS
|
Share
exchange transaction
On
January 5, 2010, the Company completed a share exchange transaction (the “Share
Exchange”) and concurrent US$12 million private placement (the “Private
Placement”) pursuant to which it acquired the parent company of Yantai Bohai
Pharmaceuticals Group Co., Ltd. (“Bohai”). Based in Yantai, Shandong
Province, China, Bohai is engaged in the production, manufacturing and
distribution of traditional Chinese herbal medicines, including capsules and
other products, in China.
In the
Share Exchange, the shareholders of Chance High International Limited, Bohai’s
indirect parent company organized in the British Virgin Islands (“Chance High”),
exchanged all of their Chance High equity for 13,162,500 newly issued shares of
the Company, representing approximately 81% of the outstanding shares of Company
common stock prior to the Private Placement. As a result of the Share
Exchange, Chance High became a directly held, wholly-owned subsidiary of the
Company. Also as a result of the Share Exchange, a change of control
of the Company for purposes of the Company's promissory notes occurred and such
notes were converted as a result into an aggregate of 200,000 shares of common
stock of the Company.
As part
of the share exchange transaction, the Company's sole director and officer,
Anthony Zaradic, resigned and Hongwei Qu, Bohai’s Executive Director, was
appointed President, Chief Executive Officer and interim Chief Financial Officer
of the Group as well as, effective January 16, 2010, the Group’s sole
director.
In the
Private Placement, the Group issued 6,000,000 units at $2.00 per unit, resulting
in gross proceeds of $12,000,000. Net proceeds to the Company were
approximately $9,700,000, which will be utilized to support the business of
Bohai. Each unit consists of a $2.00 principal amount, two year
convertible note and a three year warrant to purchase one share of Company
common stock at $2.40 per share, subject to certain conditions.
F-50
You
should rely only on the information contained in this document. We
have not authorized anyone to provide you with information that is
different. This document may only be used where it is legal to sell
these securities. The information in this document may only be
accurate on the date of this document.
Additional
risks and uncertainties not presently known or that are currently deemed
immaterial may also impair our business operations. The risks and
uncertainties described in this document and other risks and uncertainties which
we may face in the future will have a greater impact on those who purchase our
common stock. These purchasers will purchase our common stock at the
market price or at a privately negotiated price and will run the risk of losing
their entire investment.

BOHAI
PHARMACEUTICALS GROUP, INC.
20,322,529
Shares of
Common
Stock
PROSPECTUS
,
2010
PART
II
INFORMATION
NOT REQUIRED IN PROSPECTUS
Item
13. Other Expenses of Issuance and Distribution
The
following table sets forth an itemization of all estimated expenses, all of
which we will pay, in connection with the issuance and distribution of the
securities being registered:
|
Nature of Expense
|
Amount
|
|||
|
SEC
registration fee
|
$ | 2,484.092 | ||
|
Legal
fees and expenses
|
55,000 | |||
|
Accounting
fees and expenses
|
15,000 | |||
|
TOTAL
|
$ | 72,484.092 | ||
Item
14. Indemnification of Directors and Officers
Charter
Provisions and Other Agreement of the Company
The
current bylaws of the Company provides that the Board of the Company shall cause
the Company to indemnify a current or former director, officer and Secretary of
the Company, or a current or former director, officer and Company of a
corporation of which the Company is or was a stockholder and the heirs and
personal representative of any such person against all costs, charges and
expenses to settle an action, judgment or proceeding to which they are made a
party by reason of their position of director or officer of the
Company.
The
Company is permitted by the Bylaws to purchase and maintain insurance for any
director, officer, employee or agent of the Company or as a director, officer,
employee or agent of the Company of which the Company is or was a stockholder
and his or her heirs or personal representatives against a liability incurred by
him as a Director, officer, employee or agent.
Nevada
Law
Our company is incorporated under the
laws of the State of Nevada. Section 78.7502 of the Nevada Revised Statutes
provides that a Nevada corporation may indemnify any person who was or is a
party or is threatened to be made a party to any threatened, pending or
completed action, suit or proceeding, whether civil, criminal, administrative or
investigative, except an action by or in the right of the corporation, by reason
of the fact that he is or was a director, officer, employee or agent of the
corporation, or is or was serving at the request of the corporation as a
director, officer, employee or agent of another corporation, partnership, joint
venture, trust or other enterprise, against expenses, including attorneys’ fees,
judgments, fines and amounts paid in settlement actually and reasonably incurred
by him in connection with the action, suit or proceeding if he acted in good
faith and in a manner which he reasonably believed to be in or not opposed to
the best interests of the corporation, and, with respect to any criminal action
or proceeding, had no reasonable cause to believe his conduct was unlawful. The
termination of any action, suit or proceeding by judgment, order, settlement,
conviction or upon a plea of nolo contendere or its
equivalent, does not, of itself, create a presumption that the person did not
act in good faith and in a manner which he reasonably believed to be in or not
opposed to the best interests of the corporation, and that, with respect to any
criminal action or proceeding, he had reasonable cause to believe that his
conduct was unlawful.
Section 78.7502 further provides a
Nevada corporation may indemnify any person who was or is a party or is
threatened to be made a party to any threatened, pending or completed action or
suit by or in the right of the corporation to procure a judgment in its favor by
reason of the fact that he is or was a director, officer, employee or agent of
the corporation, or is or was serving at the request of the corporation as a
director, officer, employee or agent of another corporation, partnership, joint
venture, trust or other enterprise against expenses, including amounts paid in
settlement and attorneys' fees actually and reasonably incurred by him in
connection with the defense or settlement of the action or suit if he acted in
good faith and in a manner which he reasonably believed to be in or not opposed
to the best interests of the corporation. Indemnification may not be made for
any claim, issue or matter as to which such a person has been adjudged by a
court of competent jurisdiction, after exhaustion of all appeals therefrom, to
be liable to the corporation or for amounts paid in settlement to the
corporation, unless and only to the extent that the court in which the action or
suit was brought or other court of competent jurisdiction determines upon
application that in view of all the circumstances of the case, the person is
fairly and reasonably entitled to indemnity for such expenses as the court deems
proper.
Section
78.751 of the Nevada Revised Statutes provides that discretionary
indemnification under Section 78.7502 unless ordered by a court or advanced
pursuant to subsection 2 of section 78.751, may be the corporation only as
authorized in the specific case upon a determination that indemnification of the
director, officer, employee or agent is proper in the circumstances. The
determination must be made by:
|
|
·
|
By
the stockholders;
|
|
|
·
|
By
the board of directors by majority vote of a quorum consisting of
directors - who were not parties to the action, suit or
proceeding;
|
|
|
·
|
If
a majority vote of a quorum consisting of directors who were not parties
to the action, suit or proceeding so orders, by independent legal counsel
in a written opinion; or
|
|
|
·
|
If
a quorum consisting of directors who were not parties to the action, suit
or proceeding cannot be obtained, by independent legal counsel in a
written opinion.
|
The
Articles of Incorporation, the Bylaws or an agreement made by the corporation
may provide that the expenses of officers and directors incurred in defending a
civil or criminal action, suit or proceeding must be paid by the corporation as
they are incurred and in advance of the final disposition of the action, suit or
proceeding, upon receipt of an undertaking by or on behalf of the director or
officer to repay the amount if it is ultimately determined by a court of
competent jurisdiction that he is not entitled to be indemnified by the
corporation. The provisions of this subsection do not affect any rights to
advancement of expenses to which corporate personnel other than directors or
officers may be entitled under any contract or otherwise by law.
The
indemnification and advancement of expenses authorized in or ordered by a court
pursuant to NRS Section 78.751:
|
|
·
|
does
not exclude any other rights to which a person seeking indemnification or
advancement of expenses may be entitled under the articles of
incorporation or any bylaw, agreement, vote of stockholders or
disinterested directors or otherwise, for either an action in his official
capacity or an action in another capacity while holding his office, except
that indemnification, unless ordered by a court pursuant to section
78.7502 or for the advancement of expenses made pursuant to subsection 2
of section 78.751, may not be made to or on behalf of any director or
officer if a final adjudication establishes that his acts or omissions
involved intentional misconduct, fraud or a knowing violation of the law
and was material to the cause of action;
and
|
|
|
·
|
continues
for a person who has ceased to be a director, officer, employee or agent
and inures to the benefit of the heirs, executors and administrators of
such a person.
|
Insofar
as indemnification for liabilities arising under the Securities Act of 1933, as
amended (the “Securities Act”), may be permitted to directors, officers and
controlling persons of our company under Nevada law or otherwise, we have been
advised the opinion of the Securities and Exchange Commission is that such
indemnification is against public policy as expressed in the Securities Act and
is, therefore, unenforceable. In the event a claim for
indemnification against such liabilities (other than payment by us for expenses
incurred or paid by a director, officer or controlling person of our company in
successful defense of any action, suit, or proceeding) is asserted by a
director, officer or controlling person in connection with the securities being
registered, we will, unless in the opinion of its counsel the matter has been
settled by controlling precedent, submit to a court of appropriate jurisdiction,
the question of whether such indemnification by it is against public policy in
the Securities Act and will be governed by the final adjudication of such
issue.
Item
15. Recent Sales of Unregistered Securities
The
following private placements of the Company’s securities were made in reliance
upon the exemption from registration under Section 4(2) of the Securities Act of
1933, as amended and/or Rule 506 of Regulation D promulgated under the
Securities Act.
In
connection with the consummation of the transactions contemplated by our January
5, 2010 Share Exchange and private placement:
(i) Pursuant
to the Share Exchange Agreement, the Company issued 13,162,500 shares of common
stock to Chance High Shareholders in exchange for 100% of the outstanding shares
of Chance High. Such securities were not registered under the
Securities Act. These securities qualified for exemption under
Section 4(2) of the Securities Act since the issuance securities by us did not
involve a public offering. The offering was not a “public offering”
as defined in Section 4(2) of the Securities Act and met the requirements to
qualify for exemption under Section 4(2) of the Securities Act.
(ii) Pursuant
to the Securities Purchase Agreement, the Company issued to the Investors a
total of 6,000,000 Units for $12 million with each Unit consisting of a
unsecured $2.00 principal amount Notes payable 24 months from January 5, 2010
with an interest rate of 8% per annum payable quarterly in arrears, and a three
year Warrant to purchase one share of Common Stock. The Notes and
Warrants were not registered under the Securities Act. The issuance
of these securities was exempt from registration under Regulation D and
Section 4(2) of the Securities Act. The Company made this
determination based on the representations of Investors, which included, in
pertinent part, that such Investors were “accredited investors” within the
meaning of Rule 501 of Regulation D promulgated under the Securities Act and
that such Investors were acquiring the Notes and Warrants for investment
purposes for their own respective accounts and not as nominees or agents, and
not with a view to the resale or distribution thereof, and that the Investors
understood that the Notes and Warrants may not be sold or otherwise disposed of
without registration under the Securities Act or an applicable exemption
therefrom.
(iii) In
connection with the Private Placement, on January 5, 2010, the Company issued to
affiliates of Euro Pacific and to Chardan three-year Placement Agent Warrants to
purchase 600,000 shares of Common Stock at an exercise price of $2.40 per
share. 31,500 placement agent warrants were issued to Chardan and
568,500 placement agent warrants were issued to affiliates of Euro
Pacific. The Agent Warrants are substantially identical to the
Warrants issued to the Investors in the Private Placement, except that such
warrants may not be exercised until the six (6) month anniversary of the later
of: (i) the date of effectiveness of this registration statement or (ii) the
date of commencement of sales in connection with this offering pursuant to
separate lock-up agreements. Such issuance of the Agent Warrants was
not registered under the Securities Act. The issuance of the Agent
Warrants was exempt from registration under Section 4(2) of the Securities
Act.
Outstanding Convertible
Notes. On June 15, 2009, the Company issued four convertible
notes for an aggregate of $400,000, which notes were amended on August 7,
2009. The principal of such notes shall, automatically and without
any action on the part of the holders, convert into one share of Common Stock
for each two dollars converted, upon the occurrence of Change in Control as
defined in the notes. All of such outstanding convertible notes were converted
into Common Stock upon the Share Exchange, resulting in the issuance of 200,000
shares of Common Stock.
Notes Issued in Connection with the
Private Placement. In connection with the Private Placement,
the Company offered and sold $12,000,000 worth of Notes convertible into
6,000,000 shares of Common Stock. The issuance of the Notes was not
registered under the Securities Act as such issuance was exempt from
registration under Section 4(2) of the Securities Act and Regulation D
promulgated thereunder.
The Notes
are unsecured, payable 24 months from the date of Closing with an interest rate
of 8% per annum payable quarterly in arrears. The Company has placed
in escrow with the Escrow Agent an amount of the proceeds of the Private
Placement equal to one quarter worth of interest payments on the Notes to secure
prompt interest payments, or $240,000. Until such time as 75% of the
Notes are converted into shares of Common Stock, if such escrow is depleted in
order to make interest payments, Bohai will replenish such escrow
amount. At the option of the holder, the Notes may be converted into
Common Stock at a price of $2.00 per share, which conversion price is subject to
customary weighted average and stock based anti-dilution
protection.
The Notes
contain standard events of default, including: (i) failure to file the a
registration statement to register the Common Stock underlying the Notes and
Warrants (the “Registration Statement”) with the SEC within the prescribed
period; (ii) failure to have the Registration Statement deemed effective by the
SEC within the prescribed period; (iii) failure to maintain the effectiveness of
the Registration Statement thereafter; (iv) nonpayment of principal or interest;
(v) termination of registration or suspension of reporting obligations under the
Exchange Act, suspension from trading on the OTCBB (or an exchange), or failure
to file reports with the SEC on a timely basis as required by the Exchange Act;
(vi) material breach of representations, warranties and other obligations under
the transaction documents associated with the Securities Purchase Agreement;
(vii) enforcement proceedings; (viii) cross default and cross acceleration; (ix)
insolvency, winding up and other market standard analogous events; (x)
moratorium and nationalization; (xi) proceedings against the Company or its
consolidated subsidiaries with potential loss/damage of U.S.$5 million or more;
and (xii) illegality of Notes under any applicable law.
The Notes
also contain customary affirmative and negative covenants of the Company,
including negative covenants which restrict the Company’s ability to do the
following without the consent of Euro Pacific, as representative of the
Investors: (i) incur, or permit to exist, any indebtedness for
borrowed money in excess of (A) US$10,000,000 during the twelve (12) month
period beginning on January 5, 2010, or (B) US$15,000,000 during period
beginning on January 5, 2010 and ending on January 5, 2012 (the maturity date of
the Notes), except in the ordinary course of the Company’s business; (ii) lend
or advance money, credit or property to or invest in (by capital contribution,
loan, purchase or otherwise) any person or entity in excess of US$2,000,000 except: (A) investments
in United States Government obligations, certificates of deposit of any banking
institution with combined capital and surplus of at least $200,000,000; (B)
accounts receivable arising out of sales in the ordinary course of business; and
(C) inter-company loans between and among the Company and its subsidiaries;
(iii) pay dividends or make any other distribution on shares of the capital
stock of the Company; (iv) create, assume or permit to exist, any lien on any of
the Company’s property or assets now owned or hereafter acquired, subject to
existing liens and certain exceptions; (v) assume guarantees, subject to certain
exceptions; (vi) engage in “sale-leaseback” transactions, subject to certain
exceptions; (vii) make capital expenditures in excess of US$5,000,000 in any
fiscal year, subject to certain exceptions; and (viii) materially alter the
Company’s business.
Warrants. The
Warrants to purchase Common Stock were issued in conjunction with the Private
Placement. The Company offered and sold Warrants to purchase
6,000,000 shares of Common Stock. The issuance of the Warrants was
not registered under the Securities Act as such issuance was exempt from
registration under Section 4(2) of the Securities Act and Regulation D
promulgated thereunder. Each Warrant entitles the holder to purchase
one share of Common Stock. The Warrants shall be exercisable in whole
or in part, at an initial exercise price per share of $2.40, which exercise
price is subject to customary weighted average and stock based anti-dilution
protection. The Warrants may be exercised at any time upon the
election of the holder, beginning on the date of issuance and ending of the
third anniversary of the closing of the Private Placement. The
Warrants shall not be redeemable.
In the
event of the Company’s liquidation, dissolution or winding up, the holders of
Warrants will not be entitled to participate in the distribution of assets of
the Company. In addition, holders of Warrants do not have voting,
pre-emptive, subscription or other rights of stockholders in respect of the
Warrants, nor shall the Holders be entitled to receive dividends.
Item
16. Exhibits
|
Exhibit No.
|
Description
|
|
|
2.1
|
Share
Exchange Agreement, dated January 5, 2010, by and among the Company,
Chance High and Shareholders of Chance High (1)
|
|
|
3.1
|
Articles
of Incorporation of the Company (2)
|
|
|
3.2
|
Bylaws
of the Company (3)
|
|
|
3.3
|
Certificate
of Amendment to Articles of Incorporation (4)
|
|
|
3.4
|
Articles
of Merger and Agreement and Plan of Merger as filed with the Secretary of
State of Nevada on January 29, 2010 (5)
|
|
|
4.1
|
Form
of Note issued to the Investors in the Private Placement, dated January 5,
2010 (1)
|
|
|
4.2
|
Form
of Warrant issued to the Investors in the Private Placement, dated January
5, 2010 (1)
|
|
|
4.3
|
Form
of Placement Agent Warrant issued to affiliates of Euro Pacific Capital,
Inc. and to Chardan Capital Markets, LLC, dated January 5, 2010
(1)
|
|
|
5.1
|
Opinion
of Ellenoff Grossman & Schole LLP*
|
|
|
10.1
|
Securities
Purchase Agreement, dated January 5, 2010, by and among the Company, the
Investors in the Private Placement and Euro Pacific Capital, Inc. as
representative of the Investors (1)
|
|
|
10.2
|
Registration
Rights Agreement, dated January 5, 2010, by and among the Company and the
Investors in the Private Placement (1)
|
|
|
10.3
|
Securities
Escrow Agreement, dated January 5, 2010, by and among the Company, Euro
Pacific Capital, Inc., as representative of the Investors, Glory Period
Limited and Escrow, LLC, as escrow agent (1)
|
|
|
10.4
|
Closing
Escrow Agreement, dated December 10, 2009, by and among the Company, Euro
Pacific Capital, Inc., as representative of the Investors, and Escrow,
LLC, as escrow agent
(1)
|
|
21.1
|
Subsidiaries
of the Registrant *
|
|
|
23.1
|
Consent
of Parker Randall CF (H.K.) CPA Limited +
|
|
|
23.2
|
Consent
of Ellenoff Grossman & Schole LLP (included in Exhibit
5.1)
|
|
|
23.3
|
Consent
of AllBright Law Offices*
|
|
|
99.1
|
Unofficial
English translation of Consulting Services Agreement, dated December 7,
2009, between Bohai and the WFOE (1)
|
|
|
99.2
|
Unofficial
English translation of Operating Agreement, dated December 7, 2009, by and
among Bohai, its shareholders and the WFOE (1)
|
|
|
99.3
|
Unofficial
English translation of Voting Rights Proxy Agreement, dated December 7,
2009, by and among Bohai, its shareholders and the WFOE
(1)
|
|
|
99.4
|
Unofficial
English translation of Equity Pledge Agreement, dated December 7, 2009, by
and among Bohai, its shareholders and the WFOE (1)
|
|
|
99.5
|
Unofficial
English translation of Option Agreement, dated December 7, 2009, by and
among Bohai, its shareholders and the WFOE (1)
|
|
|
99.6
|
Unofficial
English translation of Call Option Agreement dated December 7, 2009
(1)
|
|
|
99.7
|
Opinion
of AllBright Law Offices, dated December 31, 2009, with respect to certain
PRC matters*
|
|
*
|
Previously
filed
|
|
+
|
Filed
herewith
|
|
(1)
|
Incorporated
by reference to the Company’s Current Report of Form 8-K, filed on January
11, 2010.
|
|
(2)
|
Incorporated
by reference to Exhibit 3.1 to the Company’s Registration Statement of
Form S-1 (File Number 333-153102), filed on August 20,
2008.
|
|
(3)
|
Incorporated
by reference to Exhibit 3.2 to the Company’s Registration Statement of
Form S-1 (File Number 333-153102), filed on August 20,
2008.
|
|
(4)
|
Incorporated
by reference to Exhibit 3.1 to the Company’s Current Report on Form 8-K
filed on December 17, 2009.
|
|
(5)
|
Incorporated
by reference to Exhibit 3.1 to the Company’s Current Report on Form 8-K,
filed on February 4, 2010.
|
Item
17. Undertakings
The
undersigned registrant hereby undertakes:
(1) To
file, during any period in which offers or sales are being made, a
post-effective amendment to this registration statement:
(i) To
include any prospectus required by section 10(a)(3) of the Securities
Act;
(ii) To
reflect in the prospectus any facts or events arising after the effective date
of the registration statement (or the most recent post-effective amendment
thereof) which, individually or in the aggregate, represent a fundamental change
in the information set forth in the registration statement. Notwithstanding the
foregoing, any increase or decrease in volume of securities offered (if the
total dollar value of securities offered would not exceed that which was
registered) and any deviation from the low or high end of the estimated maximum
offering range may be reflected in the form of prospectus filed with
the SEC pursuant to Rule 424(b) if, in the aggregate, the changes in volume
and price represent no more than 20% change in the maximum aggregate offering
price set forth in the “Calculation of Registration Fee” table in the effective
registration statement; and
(iii) To
include any material information with respect to the plan of distribution not
previously disclosed in the registration statement or any material change to
such information in the registration statement;
(2) That,
for the purpose of determining any liability under the Securities Act, each such
post-effective amendment shall be deemed to be a new registration statement
relating to the securities offered therein, and the offering of such securities
at that time shall be deemed to be the initial bona fide offering
thereof.
(3) To
remove from registration by means of a post-effective amendment any of the
securities being registered which remain unsold at the termination of the
offering.
(4) That,
for the purpose of determining liability under the Securities Act to any
purchaser:
(i) If
the registrant is relying on Rule 430B:
(A)
Each prospectus filed by the registrant
pursuant to Rule 424(b)(3) shall be deemed to be part of the registration
statement as of the date the filed prospectus was deemed part of and included in
the registration statement; and
(B)
Each prospectus required to be filed pursuant to
Rule 424(b)(2), (b)(5), or (b)(7) as part of a registration statement in
reliance on Rule 430B relating to an offering made pursuant to Rule
415(a)(1)(i), (vii), or (x) for the purpose of providing the information
required by section 10(a) of the Securities Act shall be deemed to be part of
and included in the registration statement as of the earlier of the date such
form of prospectus is first used after effectiveness or the date of the first
contract of sale of securities in the offering described in the prospectus. As
provided in Rule 430B, for liability purposes of the issuer and any person that
is at that date an underwriter, such date shall be deemed to be a new effective
date of the registration statement relating to the securities in the
registration statement to which that prospectus relates, and the offering of
such securities at that time shall be deemed to be the initial bona fide
offering thereof. Provided, however, that no statement made in a registration
statement or prospectus that is part of the registration statement or made in a
document incorporated or deemed incorporated by reference into the registration
statement or prospectus that is part of the registration statement will, as to a
purchaser with a time of contract of sale prior to such effective date,
supersede or modify any statement that was made in the registration statement or
prospectus that was part of the registration statement or made in any such
document immediately prior to such effective date; or
(ii) If
the registrant is subject to Rule 430C, each prospectus filed pursuant to Rule
424(b) as part of a registration statement relating to an offering, other than
registration statements relying on Rule 430B or other than prospectuses filed in
reliance on Rule 430A, shall be deemed to be part of and included in the
registration statement as of the date it is first used after effectiveness.
Provided, however, that no statement made in a registration statement or
prospectus that is part of the registration statement or made in a document
incorporated or deemed incorporated by reference into the registration statement
or prospectus that is part of the registration statement will, as to a purchaser
with a time of contract of sale prior to such first use, supersede or modify any
statement that was made in the registration statement or prospectus that was
part of the registration statement or made in any such document immediately
prior to such date of first use.
(5) That,
for the purpose of determining liability of the registrant under the Securities
Act to any purchaser in the initial distribution of the securities, the
undersigned registrant undertakes that in a primary offering of securities of
the undersigned registrant pursuant to this registration statement, regardless
of the underwriting method used to sell the securities to the purchaser, if the
securities are offered or sold to such purchaser by means of any of the
following communications, the undersigned registrant will be a seller to the
purchaser and will be considered to offer or sell such securities to such
purchaser:
(i) Any
preliminary prospectus or prospectus of the undersigned registrant relating to
the offering required to be filed pursuant to Rule 424;
(ii) Any
free writing prospectus relating to the offering prepared by or on behalf of the
undersigned registrant or used or referred to by the undersigned
registrant;
(iii) The
portion of any other free writing prospectus relating to the offering containing
material information about the undersigned registrant or its securities provided
by or on behalf of the undersigned registrant; and
(iv)
Any other communication that is an offer in the offering made
by the undersigned registrant to the purchaser.
Insofar
as indemnification for liabilities arising under the Securities Act may be
permitted to directors, officers and controlling persons of the registrant
pursuant to the foregoing provisions, or otherwise, the registrant has been
advised that in the opinion of the SEC such indemnification is against public
policy as expressed in the Securities Act and is, therefore, unenforceable. In
the event that a claim for indemnification against such liabilities (other than
the payment by the registrant of expenses incurred or paid by a director,
officer or controlling person of the registrant in the successful defense of any
action, suit or proceeding) is asserted by such director, officer or controlling
person in connection with the securities being registered, the registrant will,
unless in the opinion of its counsel the matter has been settled by controlling
precedent, submit to a court of appropriate jurisdiction the question whether
such indemnification by it is against public policy as expressed in the
Securities Act and will be governed by the final adjudication of such
issue.
SIGNATURES
Pursuant
to the requirements of the Securities Act of 1933, the registrant has duly
caused this Amendment No. 5 to the registration statement to be signed on
its behalf by the undersigned, in the City of Yantai, Shandong Province, China
on August 12, 2010.
|
BOHAI
PHARMACEUTICALS GROUP, INC.
|
|
|
By:
|
/s/
Hongwei Qu
|
|
Name:
Hongwei Qu
|
|
|
Title: President
and Chief Executive Officer
|
|
|
(Principal
Executive Officer)
|
|
|
By:
|
/s/
Gene Hsiao
|
|
Name:
Gene Hsiao
|
|
|
Title: Chief
Financial Officer
|
|
|
(Principal
Financial and Accounting
Officer)
|
|
POWER
OF ATTORNEY
KNOW ALL
PERSON BY THESE PRESENTS that each individual whose signature appears below
constitute and appoints Hongwei Qu his true and lawful attorneys-in-fact and
agents, with full power of substitution and resubstitution, for him and in his
name, place and stead, in any and all capacities (including his capacity as a
director and/or officer), to sign any and all amendments (including
post-effective amendments) to this Registration Statement, and to sign any
registration statement for the same offering covered by the Registration
Statement that is to be effective upon filing pursuant to Rule 462(b)
promulgated under the Securities Act, and all post-effective amendments thereto,
and to file the same, will all exhibits thereto, and all other documents in
connection therewith, with the Securities and Exchange Commission, granting unto
said attorneys-in-fact and agents, and each of them, full power and authority to
do and perform each and every act and thing requisite and necessary to be done
in and about the premises, as fully to all intents and purposes as he or she
might or could do in person, hereby ratifying and confirming all that said
attorneys-in-fact and agents or either of them, or his, her or their substitute
or substitutes, may lawfully do or cause to be done or by virtue hereof.
Pursuant
to the requirements of the Securities Act of 1933, this registration statement
has been signed by the following persons in the capacities and on the dates
indicated.
|
Signature
|
Title
|
Date
|
||
|
/s/ Hongwei Qu
|
Chairman
of the Board, Chief Executive
|
August
12, 2010
|
||
|
Hongwei
Qu
|
Officer
and President
|
|||
|
/s/ Chengde Wang
|
Director
|
August
12, 2010
|
||
|
Chengde
Wang
|
||||
|
/s/ Louis A. Bevilacqua.
|
Director
|
August
12, 2010
|
||
|
Louis
A. Bevilacqua
|
||||
|
/s/ Adam Wasserman
|
Director
|
August
12, 2010
|
||
|
Adam
Wasserman
|
