As filed with the U.S. Securities and Exchange Commission on
June 28, 2010
Registration
No. 333-166009
UNITED STATES SECURITIES AND
EXCHANGE COMMISSION
Washington, D.C.
20549
Amendment No. 2
to
Form S-1
REGISTRATION
STATEMENT
UNDER
THE SECURITIES ACT OF
1933
Arizona Chemical Ltd.
(Exact name of registrant as
specified in its charter)
| |
|
|
|
|
|
Bermuda
|
|
2821
|
|
98-0650421
|
|
(State or other jurisdiction
of
incorporation or organization)
|
|
(Primary Standard Industrial
Classification Code Number)
|
|
(I.R.S. Employer
Identification No.)
|
4600 Touchton Road East
Building 100
Suite 1500
Jacksonville, Florida 32246
(800) 526-5294
(Address, including zip code,
and telephone number, including area code, of registrant’s
principal executive offices)
Pamela J. Simmons
4600 Touchton Road
East
Building 100
Suite 1500
Jacksonville, Florida
32246
(800) 526-5294
(Name, address, including zip
code, and telephone number, including area code, of agent for
service)
Copies to:
| |
|
|
|
Robert W. Downes
|
|
Kris F. Heinzelman
|
|
Sullivan & Cromwell LLP
|
|
Cravath, Swaine & Moore LLP
|
|
125 Broad Street
|
|
825 Eighth Avenue
|
|
New York, New York 10004
|
|
New York, New York 10019
|
|
(212)
558-4000
|
|
(212) 474-1000
|
Approximate date of commencement of proposed sale to the
public: As soon as practicable after the
effective date hereof.
If any of the securities being registered on this Form are to be
offered on a delayed or continuous basis pursuant to
Rule 415 under the Securities Act of 1933, check the
following
box. o
If this Form is filed to register additional securities for an
offering pursuant to Rule 462(b) under the Securities Act,
check the following box and list the Securities Act registration
statement number of the earlier effective registration statement
for the same
offering. o
If this Form is a post-effective amendment filed pursuant to
Rule 462(c) under the Securities Act, check the following
box and list the Securities Act registration statement number of
the earlier effective registration statement for the same
offering. o
If this Form is a post-effective amendment filed pursuant to
Rule 462(d) under the Securities Act, check the following
box and list the Securities Act registration statement number of
the earlier effective registration statement for the same
offering. o
Indicate by check mark whether the registrant is a large
accelerated filer, an accelerated filer, a non-accelerated
filer, or a smaller reporting company. See the definitions of
“large accelerated filer”, “accelerated
filer” and “smaller reporting company” in Rule
12b-2 of the
Exchange Act. (Check one):
|
|
|
|
| Large accelerated
filer o
|
Accelerated
filer o
|
Non-accelerated
filer þ
|
Smaller reporting
company o
|
(Do not check if a smaller
reporting company)
The Registrant hereby amends this Registration Statement on
such date or dates as may be necessary to delay its effective
date until the Registrant shall file a further amendment which
specifically states that this Registration Statement shall
thereafter become effective in accordance with Section 8(a)
of the Securities Act of 1933 or until the Registration
Statement shall become effective on such date as the Commission,
acting pursuant to said Section 8(a), may determine.
The
information in this preliminary prospectus is not complete and
may be changed. These securities may not be sold until the
registration statement filed with the Securities and Exchange
Commission is effective. This preliminary prospectus is not an
offer to sell nor does it seek an offer to buy these securities
in any jurisdiction where the offer or sale is not permitted.
|
Subject to Completion. Dated
June 28, 2010
Shares
Arizona Chemical Ltd.
Common Shares
This is an initial public offering of common shares of Arizona
Chemical Ltd., or Arizona Chemical. The company is
offering
of the shares to be sold in the offering. The selling
shareholder identified in this prospectus is offering an
additional shares.
The shares to be sold by the company and the selling stockholder
will be sold to the underwriters identified in this prospectus.
Arizona Chemical will not receive any of the proceeds from the
sale of the shares being sold by the selling shareholder.
Prior to this offering, there has been no public market for the
common shares. It is currently estimated that the initial public
offering price per share will be between
$ and
$ . Arizona Chemical intends to
apply to list the common shares on the New York Stock Exchange
under the symbol “ARZ”.
See “Risk Factors” on page 16 to read about
factors you should consider before buying common shares.
Neither the Securities and Exchange Commission nor any other
regulatory body has approved or disapproved of these securities
or passed upon the accuracy or adequacy of this prospectus. Any
representation to the contrary is a criminal offense.
| |
|
|
|
|
|
|
|
|
|
|
|
Per Share
|
|
|
Total
|
|
|
|
|
Initial price to public
|
|
$
|
|
|
|
$
|
|
|
|
Underwriting discount
|
|
$
|
|
|
|
$
|
|
|
|
Proceeds, before expenses, to Arizona Chemical
|
|
$
|
|
|
|
$
|
|
|
|
Proceeds, before expenses, to the selling shareholder
|
|
$
|
|
|
|
$
|
|
|
To the extent that the underwriters sell more
than common
shares, the underwriters have the option to purchase up to an
additional shares
from the selling shareholder at the initial price offered to the
public less the underwriting discount. Arizona Chemical will not
receive any of the proceeds from the common shares sold by the
selling shareholder pursuant to the underwriters’ option to
purchase additional shares.
The underwriters expect to deliver the shares against payment in
New York, New York
on ,
2010.
Goldman, Sachs &
Co.
Prospectus
dated ,
2010
TABLE OF
CONTENTS
Prospectus
Through and
including ,
2010 (the 25th day after the date of this prospectus), all
dealers effecting transactions in these securities, whether or
not participating in this offering, may be required to deliver a
prospectus. This is in addition to a dealer’s obligation to
deliver a prospectus when acting as an underwriter and with
respect to an unsold allotment or subscription.
No dealer, salesperson or other person is authorized to give any
information or to represent anything not contained in this
prospectus. You must not rely on any unauthorized information or
representations. This prospectus is an offer to sell only the
shares offered hereby, but only under circumstances and in
jurisdictions where it is lawful to do so. The information
contained in this prospectus is current only as of its date.
This prospectus will be filed with the Registrar of Companies in
Bermuda in accordance with Bermuda law. In accepting this
prospectus for filing, the Registrar of Companies in Bermuda
does not accept any responsibility for our financial soundness
or the correctness of any of the statements made or opinions
expressed in this prospectus.
i
MARKET AND
INDUSTRY DATA
We use market data, industry forecasts and projections
throughout this prospectus. We have obtained portions of this
information from market research prepared by Arthur D. Little
Benelux S.A./N.V., which we refer to as “ADL”, in a
market study that ADL has prepared for us at our expense in
connection with this offering. In addition, we have obtained
portions of this information from other publicly available
industry publications. The forecasts and projections are based
on industry surveys and the preparers’ experience in the
industry, and there is no assurance that any of the projected
amounts will be achieved. We believe that the surveys and market
research performed by others, including ADL, are reliable. While
we are not aware of any misstatements regarding the industry
data presented herein, estimates involve risks and uncertainties
and are subject to change based on various factors, including
those discussed under the heading “Risk Factors”.
ii
PROSPECTUS
SUMMARY
The following summary highlights selected information
contained elsewhere in this prospectus. It does not contain all
the information that may be important to you in making an
investment decision and should be read together with the more
detailed information contained elsewhere in this prospectus. You
should read this entire prospectus carefully, including the
sections entitled “Risk Factors” and
“Management’s Discussion and Analysis of Financial
Condition and Results of Operations” and our financial
statements and related notes included elsewhere in this
prospectus. In this prospectus, unless the context requires
otherwise, references to “Arizona Chemical”, “the
Company”, “the Issuer”, “we”,
“our” or “us” refer to Arizona Chemical
Ltd., the issuer of the common shares offered hereby and its
consolidated subsidiaries, including Arizona Chem Sweden
Holdings AB, a Swedish company whose financial statements are
included herein, after giving effect to the Reorganization
described below. “Rhône Capital” refers to
Rhône Capital L.L.C. and its affiliated entities, including
Rhône Capital III L.P., the general partner of certain
associated funds with investments in Arizona Chemical, and
“International Paper” refers to International Paper
Company.
Our
Business
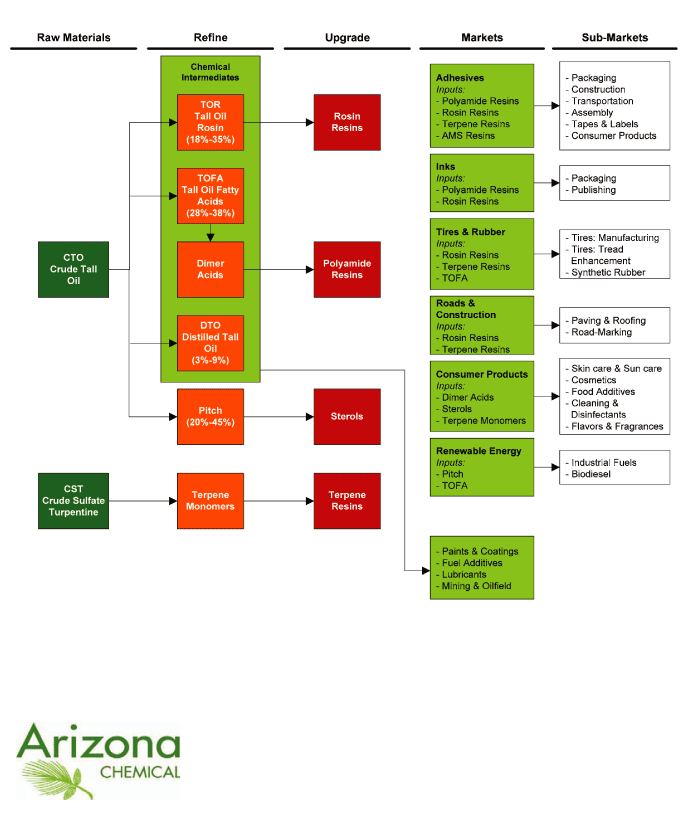
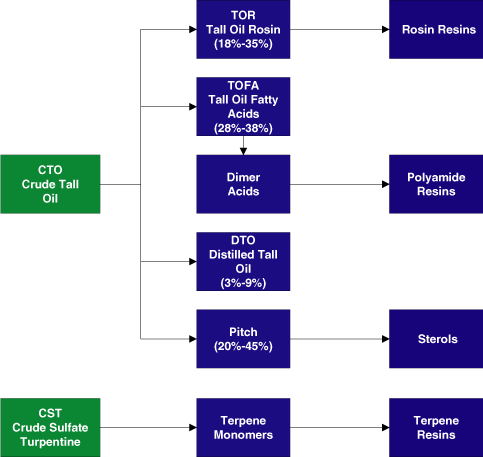
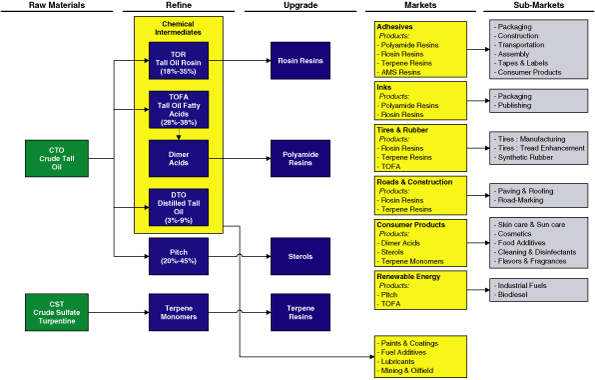
We are the world’s leading supplier of pine-based chemicals
as measured by sales. We refine and further upgrade two primary
feedstocks, crude tall oil, or CTO, and crude sulfate
turpentine, or CST, both of which are wood pulping co-products,
into specialty chemicals. We focus our resources on six target
markets that we believe offer the greatest potential for growth
and in which we offer our highest value-added products. These
markets are (1) adhesives, (2) inks, (3) tires
and rubber, (4) roads and construction, (5) consumer
products and (6) renewable energy. Our leading position in
our target markets is supported by our recognized brands,
including
SYLVATAC®,
SYLVARES®,
SYLVAPRINT®
and
UNI-REZ®,
among others. These products are complemented by a portfolio of
chemical intermediates that includes tall oil rosin, or TOR,
tall oil fatty acid, or TOFA, dimer acid and distilled tall oil,
or DTO, which have contributed steady profit margins and stable
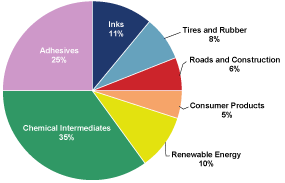
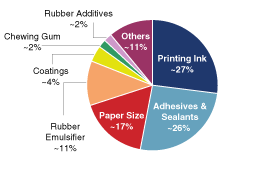
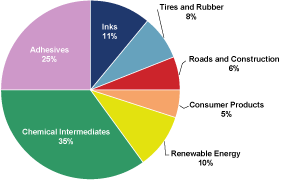
cash flows. The following chart presents the percentage of our
net sales attributable to our six target markets plus our
portfolio of chemical intermediates.
Arizona
Chemical’s 2009 Net Sales $767.5 million
|
|
|
| |
•
|
Adhesives. We are a leading global
supplier of tackifiers to the adhesives industry as measured by
sales and the world’s largest producer of tackifier resins
from renewable resources in terms of volume.
|
| |
| |
•
|
Inks. We are a major supplier of ink
resins to many of the world’s leading printing ink
companies for use in publication and packaging inks.
|
| |
| |
•
|
Tires and Rubber. We are a leading
supplier of resins and additives from renewable resources to the
global tires and rubber industry as measured by sales.
|
1
|
|
|
| |
•
|
Roads and Construction. We are a major
supplier of resins for use in roadmarking, and we believe there
are future opportunities to supply fatty acids and tackifier
resins for bitumen applications in the roofing and paving
sub-markets.
|
| |
| |
•
|
Consumer Products. We sell a diverse
range of specialty materials used in the formulation of consumer
products for the personal care, home care, industrial cleaning
and food ingredients sub-markets.
|
| |
| |
•
|
Renewable Energy. We are the largest
global producer of tall oil pitch, a second generation,
cellulosic biofuel used in municipal heating and industrial
power generation, as measured by volume. Further, our TOFA can
also be used as a raw material in the manufacture of biodiesel.
|
| |
| |
•
|
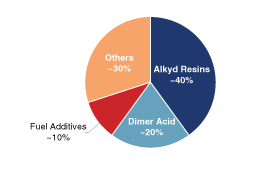
Chemical Intermediates. Our TOR, TOFA,
dimer acid and DTO chemical intermediates are sold into a
diverse range of markets, including, among others, paints and
coatings, lubricants, fuel additives, mining and oilfield, paper
chemicals and polymers.
|
Our products and technical support enhance the value of our
customers’ products by improving their performance,
providing them with essential attributes, lowering costs and
simplifying processes. We have cultivated longstanding
relationships with leading customers in our key markets and have
a history of co-developing many of our products with our
customers to satisfy specific product requirements. Our
innovative products and solutions help our customers replace
non-renewable raw materials with more sustainable alternatives.
In the three months ended March 31, 2010, we generated net
sales of $198.1 million, Adjusted EBITDA of
$27.4 million, net income of $10.0 million and net
cash provided by operating activities of $2.5 million. In
2009, we generated net sales of $767.5 million, Adjusted
EBITDA of $93.9 million, net income of $12.1 million
and net cash provided by operating activities of
$117.3 million, and we used cash in our investing and
financing activities of $44.3 million and
$63.8 million, respectively. During 2009, approximately 42%
of net sales were to customers in the United States and Canada,
49% to customers in Europe, the Middle East and Africa, 5% to
customers in Asia and 4% to customers in Latin America. For a
definition of Adjusted EBITDA, a description of our use of
Adjusted EBITDA as a measure of operating performance and a
reconciliation of Adjusted EBITDA to net income, see
“Summary Selected Historical Consolidated Financial
Information and Other Data”.
We have reinvigorated our business following our acquisition
from International Paper by Rhône Capital in February 2007.
Comparing 2006, the last full year we operated as a division of
International Paper, to 2009, as a stand-alone company, we have
improved our Adjusted EBITDA from $78.3 million to
$93.9 million and improved our Adjusted EBITDA as a
percentage of net sales, which we refer to as our “Adjusted
EBITDA margins”, from 10.2% to 12.2%. During this same
period, our net income fluctuated from $33.1 million in
2006, to net losses in both 2007 and 2008 of $23.6 million and
$26.6 million, respectively, to net income of
$12.1 million in 2009. We have also increased our cash
provided by operating activities from $5.0 million to
$117.3 million, reduced our headcount from 1,399 to
1,075 employees, reduced our manufacturing fixed costs by
$21.7 million (excluding depreciation and the impact of
foreign currency exchange) and reduced working capital
(excluding cash and cash equivalents and the current portion of
long-term debt) from 20.8% to 10.3% of net sales.
We have demonstrated the quality of our business through our
ability to improve our operating results and increase our
margins and cash flow despite the global economic downturn that
began in late 2008. During 2008 and into 2009, we experienced
sales volume declines across the majority of our businesses,
which we believe was exacerbated by inventory destocking.
However, beginning in the second half of 2009, we have seen
improving demand in our end markets and believe the declining
trend in inventory has stabilized. We believe we are well
positioned to continue our recent trajectory as demand patterns
shift toward recovery.
A core component of our strategy is an increased focus on
innovation. We are pursuing multiple opportunities for future
sales growth by developing new higher margin and higher value
products and applications and entering new markets. We expect to
supplement these initiatives with selective
2
acquisitions or investments that enhance our market position
through unique technology or through access to new market
opportunities.
We have a track record of implementing programs that have driven
significant costs out of our business, and we believe there are
additional opportunities to further optimize our manufacturing
footprint, supply chain and overhead, which we continue to
pursue. In addition to these cost reduction programs, we
continue to work to transform our company from a
functional-oriented organization to a process-oriented
organization with an increased focus on our customers. This
effort is supported by our implementation of a new SAP global
enterprise resource planning, or ERP, system, which we completed
in the first half of 2010. We expect this new ERP system to
generate additional value for our shareholders by providing us
with accurate and timely information that will enable us to
target additional margin and cost improvement opportunities.
Key Competitive
Strengths
The following competitive strengths underpin our ability to
create shareholder value by driving sustainable, profitable
top-line growth, improving margins and generating strong cash
flow:
Leading Market
Positions
We are the world’s leading supplier of pine-based chemicals
as measured by sales. According to ADL, we are the leading
global supplier of each of the following products:
|
|
|
| |
•
|
naturally-derived resins used in adhesives, inks and roadmarking
applications;
|
| |
| |
•
|
naturally-derived tackifier resins used in hot melt packaging
and bookbinding adhesives;
|
| |
| |
•
|
naturally-derived tread enhancement resins for passenger car
tires; and
|
| |
| |
•
|
pine-based, non genetically-modified sterols used in food
ingredient and nutrient applications through our joint venture,
Arboris, LLC.
|
Scope and
Scale Provide Global Access
With manufacturing facilities in Finland, France, Germany,
Sweden, the United States and the United Kingdom, we have the
largest manufacturing capacity in the pine-based chemicals
sector. Our supply chain and commercial infrastructure gives us
access to global opportunities as we have the capability to
reliably service an increasingly international client base. We
believe we are the leading global refiner of CTO and the leading
global producer of TOFA and TOR.
Advantageous
Feedstock Position
We are the world’s largest buyer of CTO, and have the
capacity to refine approximately 650,000 metric tonnes of CTO
annually, which, according to ADL, represents approximately 40%
of global CTO refining capacity. We have the ability to process
a wide variety of CTO feedstocks, including lower quality CTO
and black liquor soap, or BLS, a precursor to CTO that we have
the ability in the United States to refine into CTO, providing
us with access to a broad source of raw materials. When we refer
to CTO in this prospectus, we are referring generally to CTO and
BLS, together.
We have long-term supply contracts with International Paper
pursuant to which they agreed to sell to us, and we agreed to
purchase from them, all of the CTO and CST produced at their
existing U.S. paper mills. We also have the option to
purchase all of the CTO and CST produced at International
Paper’s future paper mills worldwide. These contracts
provided us with approximately 20% and 27% of our global CTO and
CST purchases for the three months ended March 31, 2010 and
the year ended December 31, 2009, respectively. CTO and CST
accounted for approximately 68% and 64% of our raw material
purchases for the three months ended March 31, 2010 and the
year ended December 31, 2009, respectively.
3
Stable
Customer Base with Long-standing Relationships and Diverse End
Markets
With approximately 1,000 customers in more than 80 countries, we
have a broad customer base covering many diverse end-user
industries, including adhesives, inks, tires and rubber, roads
and construction, consumer products, renewable energy, fuel
additives, coatings, lubricants and polymer additives. We have
strong, long-standing relationships with our customers, and many
of our products have been developed in cooperation with our
customers, frequently in response to their specific needs.
Commitment to
Innovation
We believe that innovation and new product development are
critical to meeting our customers’ needs and to generating
profitable future growth. We employ over 50 scientific
professionals, many of whom hold Ph.D. degrees. In the five
years ended December 31, 2009, we applied for 61
U.S. patents and 18 Patent Cooperation Treaty patents.
During 2009, we instituted a structured approach to growth with
dedicated cross-functional teams that are directed toward our
six target markets. As part of this approach, we identify and
develop new products based on our understanding of our
customers’ needs and market opportunities. We utilize a
portfolio management system to ensure that we are disciplined in
assessing potential opportunities for new products and to
allocate resources and capital in accordance with our strategic
objectives and priorities.
Our track record of product innovation extends more than
80 years. Examples include:
|
|
|
| |
•
|
adhesive tackifiers designed to enable the use of a higher
amount of recycled content in packaging materials;
|
| |
| |
•
|
high solid adhesive dispersions for labels and tapes that allow
for higher coating speeds and that lower process energy costs;
|
| |
| |
•
|
heat stable rheology, or HSR, ink resins that reduce formulation
complexity for ink manufacturers while improving press
performance;
|
| |
| |
•
|
tire tread resins that promote wet grip, fuel economy and tire
life;
|
| |
| |
•
|
fuel lubricity improvers that ensure low sulfur targets for
diesel fuel can be met; and
|
| |
| |
•
|
emulsions and clear gels for skin and sun care applications.
|
Improving
Profit Margins
Our market leading position, together with the importance of our
portfolio to our customer base, our value pricing and our
ability to shift production and manage costs have enabled us to
generate improving profit margins across diverse macroeconomic
environments. Despite rapidly changing raw material, energy and
freight costs in recent years, as well as the difficult economic
environment that affected the chemicals industry in late 2008
and into 2009, we have been successful at increasing our
Adjusted EBITDA and margins, as illustrated in the chart below.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Combined
|
|
|
|
Three Months
|
|
Three Months
|
|
Year Ended
|
|
Year Ended
|
|
Year Ended
|
|
|
|
Ended
|
|
Ended
|
|
December 31,
|
|
December 31,
|
|
December 31,
|
|
|
|
March 31, 2010
|
|
March 31, 2009
|
|
2009
|
|
2008
|
|
2007
|
|
|
|
|
|
|
|
(dollars in thousands)
|
|
|
|
(Non-GAAP)
|
|
|
|
Net income (loss)
|
|
$
|
9,997
|
|
|
$
|
(2,357
|
)
|
|
$
|
12,098
|
|
|
$
|
(26,599
|
)
|
|
$
|
(23,605
|
)
|
|
Gross margins
|
|
|
20.6
|
%
|
|
|
8.1
|
%
|
|
|
15.7
|
%
|
|
|
13.3
|
%
|
|
|
11.7
|
%
|
|
Adjusted EBITDA
|
|
|
27,378
|
|
|
|
11,043
|
|
|
|
93,859
|
|
|
|
92,723
|
|
|
|
66,898
|
|
|
Adjusted EBITDA
margins
|
|
|
13.8
|
%
|
|
|
6.2
|
%
|
|
|
12.2
|
%
|
|
|
9.3
|
%
|
|
|
7.8
|
%
|
4
For a definition of Adjusted EBITDA, a description of our use of
Adjusted EBITDA as a measure of operating performance and a
reconciliation of Adjusted EBITDA to net income, as well as an
explanation of our use of combined financial information for the
year ended December 31, 2007, see “Summary Selected
Historical Consolidated Financial Information and Other
Data”.
Experienced
and Proven Management Team with Significant Equity
Interest
We have a highly motivated management team with an average of
more than 25 years of experience, combining a core of
pine-based chemical veterans with specialty chemicals experts
from outside the pine chemicals industry. Our management team
has instilled a shareholder-value-based culture throughout our
organization, successfully implemented leading productivity
practices and reinvigorated new product development. Through
management incentive vehicles, or MIVs, formed to facilitate
investment in our company by our senior managers, our management
has a significant investment in AZ Chem Investments Partners LP,
our parent.
Ability to
Satisfy Increasing Demand for Sustainable Raw
Materials
Our products are based on naturally-derived, renewable raw
materials and, as such, we are well placed to benefit from
consumer-driven demand for sustainable alternatives to existing
hydrocarbon-based products. In conjunction with our focus on
innovation, we expect to leverage the sustainable
characteristics of our product range in developing new market
opportunities for our products. For example, we expect that the
sustainable nature of our gellants and other personal care
products will be an increasingly attractive feature in the skin
care and cosmetics markets.
Our Business
Strategy
Building on our competitive strengths, set forth below are the
four key elements to our business strategy.
Organic Growth
Through Market Understanding and Development
Building upon our strength in technological innovation, we focus
our research and development efforts on opportunities that
address the needs of our customers as well as enhance our
profitability and growth profile. We look for opportunities in
new applications, new products and new markets. For example, we
are developing new personal care applications for our polymeric
gellant technology that we believe present improved margin
opportunities. Additionally, our heat stable rheology, or HSR,
ink resin products provide better performance at a lower cost
relative to products historically used in “sheet-fed”
printing applications, and we have begun to market our HSR ink
resin products for use in these applications, which we believe
also offer us improved margin opportunities. Other innovations
include extra-light color rosin esters for adhesive packaging
applications, high solid adhesive dispersions and tire tread
enhancement additives. In relation to new market opportunities,
we are presently developing compatibilizers for modified bitumen
applications, which will enable customers to produce stable,
long-lasting synthetic rubber modified bitumen with higher
sustainable material content. Products we have developed in the
last five years generated approximately 10% of our net sales in
2009.
During 2009, we spent approximately 1% of our annual net sales
on research and development efforts, and we plan to double our
research and development and marketing resources and related
spending within the next three years. In addition to internal
research and development, we expect to expand our product
offerings through select licensing
and/or
purchasing of new formulations, technology and products from
third parties, and we intend to fund select capital expenditure
opportunities where we can expand capacity in high growth, high
margin areas, or reduce costs for an appropriate return on
investment.
5
Geographical
Expansion into BRIC Countries
We expect to continue to serve our existing global customers as
they expand their businesses in emerging countries such as
Brazil, Russia, India and China and to target new customers in
these regions. In 2009, 2.7% of our net sales were generated in
these four countries, and we believe there are significant
opportunities to expand our business in these and other emerging
economies. We plan to manage our expansion into emerging markets
by adding sales and marketing, technical service and other
development related personnel, followed by further
infrastructure expansion as our sales opportunities in these
markets increase.
Active
Management of our Business and Asset Portfolio
We plan to accelerate profitable growth through selected
acquisitions of companies
and/or
assets that focus on our six target markets or on high value
niche applications and that complement our current product
offerings and capabilities. For example, in February 2009, we
acquired Abieta Chemie GmbH, a company located in Gersthofen,
Germany, that specializes in natural resin products. This
acquisition strengthened our position in the tire and rubber
market and secured access for us to new products and technology
that can be leveraged in other strategic markets.
Productivity
Improvement Program
We have a history of implementing programs that have driven
significant costs out of our business. We expect to continue to
expand our successful productivity improvement initiatives by
pursuing operational efficiencies, optimizing available
technologies, maintaining a lean organizational structure,
further reducing fixed costs, capitalizing on our global
procurement organization, rationalizing capacity and efficiently
managing capital spending. We intend to take advantage of the
enhanced flow of information from the implementation of a new
SAP global ERP system, which we completed in the first half of
2010, to further our efforts to improve our margins, drive cost
productivity and generate additional cost savings.
Risk
Factors
Despite our competitive strengths, investing in our common
shares involves substantial risk. In addition, our ability to
execute our business strategy is subject to certain risks. The
risks described under “Risk Factors” may cause us not
to realize the full benefits of our strengths or may cause us to
be unable to successfully execute all or part of our business
strategy. Before you invest in our common shares, you should
carefully consider all the information in this prospectus,
including matters set forth under “Risk Factors”.
Transactions with
Rhône Capital and International Paper
On February 28, 2007, Rhône Capital acquired a group
of operating companies that comprise Arizona Chemical from
International Paper. We refer to this transaction as the
“Acquisition”. Since the Acquisition, the companies
comprising Arizona Chemical have been subsidiaries of AZ Chem
Luxembourg Holdings S.à r.l., which, before giving effect
to the Reorganization described below, is also our direct parent
and a wholly owned subsidiary of AZ Chem Investments Partners LP.
Prior to the completion of this offering, funds associated with
Rhône Capital, which we refer to as the “Rhône
Funds”, held a % limited
partnership interest in AZ Chem Investments Partners LP, while
International Paper held a %
limited partnership interest and AZ Chem MIV I Ltd and AZ Chem
MIV II LP, which are management incentive vehicles, or MIVs,
formed to facilitate investment in our company by our senior
managers, collectively held a %
limited partnership interest.
The Rhône Funds and International Paper are also members of
AZ Chem Investments LLC, the general partner of AZ Chem
Investments Partners LP. The Rhône Funds control AZ Chem
Investments LLC, and therefore control us.
6
See “— Corporate and Other Information and the
Reorganization” for information about the reorganization of
our structure that will occur prior to the completion of this
offering.
Our Credit
Agreements and Our Outstanding Indebtedness
In connection with the Acquisition, we entered into a First Lien
Credit and Guaranty Agreement, which we refer to as our
“First Lien Credit Agreement”, and a Second Lien
Credit and Guaranty Agreement, which we refer to as our
“Second Lien Credit Agreement”. We refer to these
agreements together as our “credit agreements”. As a
result of the indebtedness we incurred under the credit
agreements, we are highly leveraged. As of March 31, 2010,
our total consolidated indebtedness was $331.6 million,
including $327.4 million outstanding under our credit
agreements. As of March 31, 2010, on an as adjusted basis
after giving effect to this offering and the application of the
net proceeds therefrom (assuming we sell the shares for a per
share price equal to $ , the
midpoint of the price range set forth on the cover page of this
prospectus), our total consolidated indebtedness would have been
$ million and we would have
had $60.0 million of borrowing capacity available under the
revolving credit facility under our First Lien Credit Agreement.
See “Description of Our Indebtedness”.
Rhône
Capital
Founded in 1996, Rhône Capital focuses on middle-market
private equity investments in businesses with pan-European or
trans-Atlantic presence or growth prospects. Rhône
Capital’s investment philosophy includes the development of
strong, strategic partnerships with the management of portfolio
companies in which it invests. Rhône Capital currently has
investments in a diversified portfolio of companies.
Corporate and
Other Information and the Reorganization
On February 12, 2010, we were incorporated as a Bermuda
limited company.
Prior to the completion of this offering, AZ Chem Luxembourg
Holdings S.à r.l. will transfer all of the equity interests
in Arizona Chem Sweden Holdings AB, its wholly owned subsidiary,
to us, and we will transfer all of the equity interests in
Arizona Chem Sweden Holdings AB to Arizona Chemical Luxembourg
S.à.r.l, our wholly owned subsidiary. Following these
transfers, AZ Chem Luxembourg Holdings S.à r.l. will
dissolve, and we will be a direct, wholly owned subsidiary of AZ
Chem Investments Partners LP. Additionally, prior to the
completion of this offering, Arizona Chemical Ltd. will effect
a for
1 stock split. We refer to these transactions as the
“Reorganization”.
The historical financial information presented herein is that of
Arizona Chem Sweden Holdings AB and its predecessor prior to the
Acquisition, and following the Reorganization, all of our
business operations will be conducted through Arizona Chem
Sweden Holdings AB and its subsidiaries.
Our principal executive offices are located at 4600 Touchton
Road East, Building 100, Suite 1500, Jacksonville,
Florida 32246 and at Transistorstraat 16, 1322 CE Almere, The
Netherlands and our registered office in Bermuda is located at
Clarendon House, 2 Church Street, Hamilton, HM 11, Bermuda. We
can be reached by telephone at
(800) 526-5294
and +(31) 36 5462 800. Our corporate website address is
www.arizonachemical.com. We do not incorporate
information on, or accessible through, our corporate website
into this prospectus, and you should not consider it a part of
this prospectus.
7
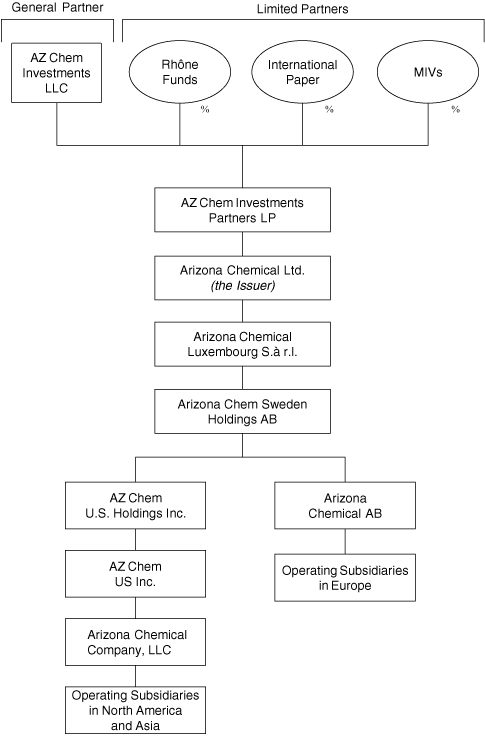
Below is an organizational chart illustrating our corporate
structure after the Reorganization and before giving effect to
this offering.
8
THE
OFFERING
|
|
|
|
Issuer |
|
Arizona Chemical Ltd. |
| |
|
Common shares offered by the issuer |
|
common
shares. |
| |
|
Common shares offered by the selling shareholder |
|
common
shares. |
| |
|
Common shares |
|
Immediately following the consummation of this offering, we will
have million common shares
outstanding. |
| |
|
Use of proceeds |
|
Assuming we sell the shares for a per share price equal to
$ , the midpoint of the price
range set forth on the cover page of this prospectus, will
receive net proceeds from this offering of approximately
$ million after deducting the
estimated underwriting discounts and commissions and estimated
offering expenses payable by us. We intend to use the net
proceeds of this offering to make an approximately
$12 million distribution to our parent entity, AZ Chem
Investments Partners LP, and to pay transaction-related expenses
of $ million. We intend to use the
remaining net proceeds to repay
$ million of indebtedness
under our First Lien Credit Agreement, representing 75% of the
remaining net proceeds, and to repay
$ million of indebtedness
outstanding under our Second Lien Credit Agreement, representing
25% of the remaining net proceeds. We will not receive any
proceeds from the sale of our common shares by the selling
shareholder. |
| |
|
Dividend policy |
|
Other than the distribution we are making to AZ Chem Investments
Partners LP out of the proceeds of this offering, we do not
anticipate paying any cash dividends on our common shares for
the foreseeable future. We intend to retain all available funds
and any future earnings to reduce debt and fund the development
and growth of our business. |
| |
|
NYSE Symbol |
|
“ARZ”. |
| |
|
Risk factors |
|
For a discussion of risks relating to our company, our business
and an investment in our common shares, see “Risk
Factors” and all other information set forth in this
prospectus before investing in our common shares. |
| |
|
|
|
Unless we specifically state otherwise, all information in this
prospectus assumes no exercise by the underwriters of their
option to purchase an
additional shares. |
9
SUMMARY SELECTED
HISTORICAL CONSOLIDATED
FINANCIAL INFORMATION AND OTHER DATA
The following tables set forth summary financial and operating
data on an historical basis for our predecessor and successor
periods (each as defined below). You should read this financial
data in conjunction with “Management’s Discussion and
Analysis of Financial Condition and Results of Operations”,
our historical consolidated financial statements, the notes to
our consolidated financial statements and the other financial
information included elsewhere in this prospectus.
We were acquired on February 28, 2007 by Rhône Capital
in a business combination accounted for under the purchase
method of accounting. The summary selected historical financial
data for the period from January 1, 2007 to
February 28, 2007 is referred to as “predecessor”
and represents the period of time when we were operated as a
division of International Paper. All periods subsequent to the
Acquisition on February 28, 2007, which resulted in a
change of control and a change in accounting basis, are referred
to as “successor”. The summary selected historical
consolidated financial and operating data for the years ended
December 31, 2009 and 2008 and the periods March 1,
2007 to December 31, 2007 and January 1, 2007 to
February 28, 2007 have been derived from the audited
historical consolidated financial statements included elsewhere
in this prospectus. The summary financial data for the three
months ended March 31, 2010 and 2009 and as of
March 31, 2010, have been derived from our unaudited
condensed consolidated financial statements, which are included
elsewhere in this prospectus. The unaudited condensed
consolidated financial statements include, in the opinion of
management, all adjustments, consisting only of normal recurring
adjustments, that management considers necessary for the fair
presentation of the condensed consolidated financial information
set forth in those statements. Results of operations for the
interim periods are not necessarily indicative of the results
that might be expected for any other interim period or for an
entire year.
As a result of the Acquisition, our assets and liabilities were
adjusted to their estimated fair values. In addition, our
consolidated statements of operations data for the successor
period include interest expense resulting from indebtedness
incurred to finance the Acquisition and depreciation and
amortization of fair value adjustments to property, plant and
equipment and intangible assets related to the Acquisition.
Therefore, our successor period financial data generally is not
comparable to our predecessor period financial data.
The combined historical results of operations for the year ended
December 31, 2007 was derived from the audited consolidated
financial statements of our predecessor from January 1,
2007 through February 28, 2007 and of our successor from
March 1, 2007 through December 31, 2007. The combined
historical results of operations are being presented to assist
comparison across the years. The combined information does not
comply with accounting principles generally accepted in the
United States (“GAAP”) and should not be used in
isolation or substitution of the results of operations of either
our predecessor period or our successor period. This data is
being presented for informational purposes only and does not
purport to represent or be indicative of the results that
actually would have been obtained had the Acquisition occurred
on January 1, 2007 or that may be obtained for any future
period.
10
Consolidated
Statements of Operations Data
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Combined
|
|
|
Successor
|
|
|
Predecessor
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(Non-GAAP)
|
|
|
|
|
|
|
|
|
|
|
Three
|
|
|
Three
|
|
|
|
|
|
|
|
|
|
|
|
March 1,
|
|
|
January 1,
|
|
|
|
|
Months
|
|
|
Months
|
|
|
Year
|
|
|
Year
|
|
|
Year
|
|
|
2007
|
|
|
2007
|
|
|
|
|
Ended
|
|
|
Ended
|
|
|
Ended
|
|
|
Ended
|
|
|
Ended
|
|
|
through
|
|
|
through
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
February 28,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
2007
|
|
|
2007
|
|
|
|
|
(dollars in thousands, except per share data)
|
|
|
|
|
Net sales
|
|
$
|
198,051
|
|
|
$
|
177,934
|
|
|
$
|
767,465
|
|
|
$
|
1,001,988
|
|
|
$
|
855,867
|
|
|
$
|
723,797
|
|
|
$
|
132,070
|
|
|
Cost of goods sold
|
|
|
157,333
|
|
|
|
163,445
|
|
|
|
646,986
|
|
|
|
868,536
|
|
|
|
755,415
|
|
|
|
642,341
|
|
|
|
113,074
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gross profit
|
|
|
40,718
|
|
|
|
14,489
|
|
|
|
120,479
|
|
|
|
133,452
|
|
|
|
100,452
|
|
|
|
81,456
|
|
|
|
18,996
|
|
|
Selling, general and administrative
|
|
|
25,673
|
|
|
|
15,464
|
|
|
|
78,200
|
|
|
|
91,936
|
|
|
|
99,583
|
|
|
|
86,684
|
|
|
|
12,899
|
|
|
Unrealized foreign currency exchange (gains) losses(1)
|
|
|
(4,836
|
)
|
|
|
367
|
|
|
|
(9,347
|
)
|
|
|
20,304
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Restructuring and impairment(2)
|
|
|
2,047
|
|
|
|
1,035
|
|
|
|
26,395
|
|
|
|
15,513
|
|
|
|
114
|
|
|
|
114
|
|
|
|
—
|
|
|
Other operating income(3)
|
|
|
—
|
|
|
|
(2,043
|
)
|
|
|
(5,537
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating income (loss)
|
|
|
17,834
|
|
|
|
(334
|
)
|
|
|
30,768
|
|
|
|
5,699
|
|
|
|
755
|
|
|
|
(5,342
|
)
|
|
|
6,097
|
|
|
Interest (expense) income, net
|
|
|
(3,870
|
)
|
|
|
(4,560
|
)
|
|
|
(16,546
|
)
|
|
|
(29,523
|
)
|
|
|
(28,657
|
)
|
|
|
(28,775
|
)
|
|
|
118
|
|
|
Loss on interest rate swaps, net
|
|
|
(709
|
)
|
|
|
(600
|
)
|
|
|
(2,541
|
)
|
|
|
(9,311
|
)
|
|
|
(2,275
|
)
|
|
|
(2,275
|
)
|
|
|
—
|
|
|
Other income(4)
|
|
|
623
|
|
|
|
2,151
|
|
|
|
3,635
|
|
|
|
1,879
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income (loss) before income tax expense (benefit)
|
|
|
13,878
|
|
|
|
(3,343
|
)
|
|
|
15,316
|
|
|
|
(31,256
|
)
|
|
|
(30,177
|
)
|
|
|
(36,392
|
)
|
|
|
6,215
|
|
|
Income tax expense (benefit)
|
|
|
3,885
|
|
|
|
(836
|
)
|
|
|
3,831
|
|
|
|
(4,277
|
)
|
|
|
(6,299
|
)
|
|
|
(8,913
|
)
|
|
|
2,614
|
|
|
Equity in earnings of affiliate(5)
|
|
|
4
|
|
|
|
150
|
|
|
|
613
|
|
|
|
380
|
|
|
|
273
|
|
|
|
189
|
|
|
|
84
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income (loss)
|
|
$
|
9,997
|
|
|
$
|
(2,357
|
)
|
|
$
|
12,098
|
|
|
$
|
(26,599
|
)
|
|
$
|
(23,605
|
)
|
|
$
|
(27,290
|
)
|
|
$
|
3,685
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Earnings per share(6):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic and diluted
|
|
$
|
9,997
|
|
|
$
|
(2,357
|
)
|
|
$
|
12,098
|
|
|
$
|
(26,599
|
)
|
|
|
|
|
|
$
|
(27,290
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average common shares outstanding:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic and diluted
|
|
|
1,000
|
|
|
|
1,000
|
|
|
|
1,000
|
|
|
|
1,000
|
|
|
|
|
|
|
|
1,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Consolidated
Balance Sheet Data
| |
|
|
|
|
|
|
|
|
|
|
|
As of March 31, 2010
|
|
|
|
|
|
|
|
As Adjusted for
|
|
|
|
|
Actual
|
|
|
the Offering(7)
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
43,057
|
|
|
$
|
|
|
|
Total assets
|
|
|
630,275
|
|
|
|
|
|
|
Total debt
|
|
|
331,645
|
|
|
|
|
|
|
Total shareholder’s equity
|
|
|
101,495
|
|
|
|
|
|
11
Other
Data
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
Successor
|
|
Successor
|
|
Successor
|
|
Combined
|
|
Successor
|
|
Predecessor
|
|
|
|
|
|
|
|
|
|
|
|
(Non-GAAP)
|
|
|
|
|
|
|
|
Three
|
|
Three
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Months
|
|
Months
|
|
|
|
|
|
|
|
March 1,
|
|
January 1,
|
|
|
|
Ended
|
|
Ended
|
|
Year Ended
|
|
Year Ended
|
|
Year Ended
|
|
2007 through
|
|
2007 through
|
|
|
|
March 31,
|
|
March 31,
|
|
December 31,
|
|
December 31,
|
|
December 31,
|
|
December 31,
|
|
February 28,
|
|
|
|
2010
|
|
2009
|
|
2009
|
|
2008
|
|
2007
|
|
2007
|
|
2007
|
|
|
|
(dollars in thousands)
|
|
|
|
Gross margin(8)
|
|
|
20.6
|
%
|
|
|
8.1
|
%
|
|
|
15.7
|
%
|
|
|
13.3
|
%
|
|
|
11.7
|
%
|
|
|
11.3
|
%
|
|
|
14.4
|
%
|
|
Depreciation(8)
|
|
$
|
6,599
|
|
|
$
|
6,872
|
|
|
$
|
29,781
|
|
|
$
|
23,311
|
|
|
$
|
23,790
|
|
|
$
|
19,693
|
|
|
$
|
4,097
|
|
|
Amortization
|
|
|
2,444
|
|
|
|
2,565
|
|
|
|
10,834
|
|
|
|
10,899
|
|
|
|
9,445
|
|
|
|
9,120
|
|
|
|
325
|
|
|
Adjusted EBITDA(9)
|
|
|
27,378
|
|
|
|
11,043
|
|
|
|
93,859
|
|
|
|
92,723
|
|
|
|
66,898
|
|
|
|
54,874
|
|
|
|
12,024
|
|
|
Adjusted EBITDA margin
|
|
|
13.8
|
%
|
|
|
6.2
|
%
|
|
|
12.2
|
%
|
|
|
9.3
|
%
|
|
|
7.8
|
%
|
|
|
7.6
|
%
|
|
|
9.1
|
%
|
|
Free cash flow(10)
|
|
$
|
(5,424
|
)
|
|
$
|
1,845
|
|
|
$
|
81,803
|
|
|
$
|
(13,452
|
)
|
|
$
|
8,446
|
|
|
$
|
25,132
|
|
|
$
|
(16,686
|
)
|
|
Net cash provided by (used in) operating activities
|
|
|
2,507
|
|
|
|
11,100
|
|
|
|
117,325
|
|
|
|
20,841
|
|
|
|
32,934
|
|
|
|
45,022
|
|
|
|
(12,088
|
)
|
|
Net cash used in investing activities
|
|
|
(7,931
|
)
|
|
|
(18,027
|
)
|
|
|
(44,294
|
)
|
|
|
(34,293
|
)
|
|
|
(501,929
|
)
|
|
|
(497,331
|
)
|
|
|
(4,598
|
)
|
|
Net cash provided by (used in) financing activities
|
|
|
2,202
|
|
|
|
(1,220
|
)
|
|
|
(63,771
|
)
|
|
|
12,768
|
|
|
|
498,843
|
|
|
|
488,547
|
|
|
|
10,296
|
|
|
|
|
|
(1) |
|
Unrealized foreign currency exchange losses (gains) is primarily
related to translation changes in the balance of the
Euro-denominated debt under our First Lien Credit Agreement.
This is further described in Note 11 of our consolidated
financial statements and Note 8 of our unaudited condensed
consolidated financial statements appearing at the end of this
prospectus and “Management’s Discussion and Analysis
of Financial Condition and Results of Operations”. |
| |
|
|
|
The following table presents unrealized and realized foreign
exchange losses (gains) included in our consolidated statement
of operations. |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
Successor
|
|
Successor
|
|
Successor
|
|
Combined
|
|
Successor
|
|
Predecessor
|
|
|
|
|
|
|
|
|
|
|
|
(Non-GAAP)
|
|
|
|
|
|
|
|
Three
|
|
Three
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Months
|
|
Months
|
|
|
|
|
|
|
|
March 1,
|
|
January 1,
|
|
|
|
Ended
|
|
Ended
|
|
Year Ended
|
|
Year Ended
|
|
Year Ended
|
|
2007 through
|
|
2007 through
|
|
|
|
March 31,
|
|
March 31,
|
|
December 31,
|
|
December 31,
|
|
December 31,
|
|
December 31,
|
|
February 28,
|
|
|
|
2010
|
|
2009
|
|
2009
|
|
2008
|
|
2007
|
|
2007
|
|
2007
|
|
|
|
(dollars in thousands)
|
|
|
|
Unrealized translation related foreign currency exchange (gains)
losses
|
|
$
|
(4,836
|
)
|
|
$
|
367
|
|
|
$
|
(9,347
|
)
|
|
$
|
20,304
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
Realized transaction related foreign currency exchange (gains)
losses — selling, general and administrative
|
|
|
(107
|
)
|
|
|
(1,883
|
)
|
|
|
214
|
|
|
|
234
|
|
|
|
(261
|
)
|
|
|
238
|
|
|
|
(499
|
)
|
|
Realized transaction related foreign currency exchange (gains)
losses — cost of goods sold
|
|
|
(205
|
)
|
|
|
37
|
|
|
|
154
|
|
|
|
65
|
|
|
|
20
|
|
|
|
3
|
|
|
|
17
|
|
|
|
|
|
(2) |
|
Restructuring and impairment includes pre-tax restructuring
charges primarily related to manufacturing facility closures and
employee terminations as part of our cost reduction initiatives.
This is further described in Note 9 of our consolidated
financial statements and Note 7 of our unaudited condensed
consolidated financial statements appearing at the end of this
prospectus and “Management’s Discussion and Analysis
of Financial Condition and Results of Operations”. |
12
|
|
|
|
|
|
The following table presents details of our restructuring and
impairment charges included in our consolidated statement of
operations. |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Combined
|
|
|
Successor
|
|
|
Predecessor
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(Non-GAAP)
|
|
|
|
|
|
|
|
|
|
|
Three
|
|
|
Three
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Months
|
|
|
Months
|
|
|
|
|
|
|
|
|
|
|
|
March 1,
|
|
|
January 1,
|
|
|
|
|
Ended
|
|
|
Ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
2007 through
|
|
|
2007 through
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
February 28,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
2007
|
|
|
2007
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Port St. Joe, Florida
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
5,822
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
Gersthofen, Germany
|
|
|
108
|
|
|
|
—
|
|
|
|
1,251
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Valdosta, Georgia
|
|
|
97
|
|
|
|
—
|
|
|
|
874
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
U.K. facilities
|
|
|
—
|
|
|
|
—
|
|
|
|
339
|
|
|
|
5,369
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Niort, France
|
|
|
648
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,972
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Dover, Ohio
|
|
|
—
|
|
|
|
3
|
|
|
|
—
|
|
|
|
635
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Pensacola, Florida
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
534
|
|
|
|
114
|
|
|
|
114
|
|
|
|
—
|
|
|
Sandarne, Sweden
|
|
|
1,194
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Impairment
|
|
|
—
|
|
|
|
1,032
|
|
|
|
18,109
|
|
|
|
7,003
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
2,047
|
|
|
$
|
1,035
|
|
|
$
|
26,395
|
|
|
$
|
15,513
|
|
|
$
|
114
|
|
|
$
|
114
|
|
|
$
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(3) |
|
Other operating income of $5.5 million recorded in 2009
included $4.9 million of insurance proceeds related to a
fire at our Oulu, Finland manufacturing facility and a gain of
$0.7 million on the sale of capital assets realized as a
result of the closure of our Bedlington, U.K. manufacturing
facility. |
| |
|
(4) |
|
In 2009, other income included a gain on our acquisition of
Abieta of $2.1 million and a gain on settlement with
International Paper of $1.3 million, whereas in 2008, other
income included a gain of $1.9 million on the
extinguishment of a portion of our debt. All of these items were
excluded from the calculation of Adjusted EBITDA. In the three
months ended March 31, 2010, other income primarily related
to insurance proceeds relating to a fire at our Sandarne, Sweden
manufacturing facility. |
| |
|
(5) |
|
Equity in earnings of affiliate relates to our 10% investment in
Arboris, LLC, which was formed in 2002 and is accounted for
under the equity method of accounting as we have the ability to
exercise significant influence. Our share of the earnings from
operations in this investment is recorded in our statement of
operations. This is further described in Note 3 of our
consolidated financial statements appearing at the end of this
prospectus and “Management’s Discussion and Analysis
of Financial Condition and Results of Operations”. |
| |
|
(6) |
|
Prior to the Acquisition, we were an operating division of
International Paper. As a result, the capital structure of our
predecessor did not include common stock or other forms of
equity shares. Accordingly, earnings per share is not available
for periods prior to March 1, 2007. Additionally, the
information presented regarding earnings per share and weighted
average common shares outstanding is that of Arizona Chem Sweden
Holdings AB on an actual basis. The earnings per share of
Arizona Chemical Ltd. on an as adjusted basis after giving
effect to the Reorganization for the three months ended
March 31, 2010 and 2009, the years ended December 31,
2009 and 2008 and the ten month period ended December 31,
2007 would have been $ ,
$ ,
$ ,
$ , and
$ , respectively,
assuming
common shares outstanding. |
| |
|
(7) |
|
Reflects on an adjusted basis the sale
of of
our common shares in this offering at an assumed initial public
offering price of $ per share (the
midpoint of the price range set forth on the cover page of this
prospectus), and the application of the estimated net proceeds
from this offering as described under “Use of
Proceeds”, and assumes this offering had been consummated
on March 31, 2010. |
| |
|
(8) |
|
Included in these amounts in 2009 is accelerated depreciation
associated with the closure of our Port St. Joe facility in the
amount of $1.6 million. Accelerated depreciation had a
negative impact on our gross margins of 1.3% in 2009. |
| |
|
(9) |
|
Adjusted EBITDA represents net income before interest, taxes,
depreciation, and amortization and is adjusted for various items
as defined in our credit agreements. Adjusted EBITDA presented
in the predecessor period was calculated on the same basis as
Adjusted EBITDA |
13
|
|
|
|
|
|
presented in the successor periods pursuant to our credit
agreements. Under the terms of our credit agreements, we use
Adjusted EBITDA to calculate our maximum leverage ratio and our
minimum interest coverage ratio covenants. See
“— Adjusted EBITDA” below. |
| |
|
(10) |
|
Free cash flow represents net cash provided by operating
activities, less purchases of property, plant and equipment,
software spending and proceeds from disposals of property, plant
and equipment. See “ — Free Cash Flow” below. |
Adjusted
EBITDA
Adjusted EBITDA is not a GAAP concept. We present Adjusted
EBITDA because it is used by management to evaluate operating
performance and under our credit agreements, we use it to
calculate our maximum leverage ratio and our minimum interest
coverage ratio covenants. In addition, a reconciliation from net
income to Adjusted EBITDA is required for reporting and covenant
calculation purposes pursuant to our credit agreements. We
consider it an important supplemental measure of our performance
and believe it is frequently used by securities analysts,
investors and other interested parties in the evaluation of
companies in our industry. We also use Adjusted EBITDA for our
executive compensation plan, which bases incentive compensation
payments on our Adjusted EBITDA performance.
Adjusted EBITDA has limitations as an analytical tool, and you
should not consider it in isolation, or as a substitute for
analysis of our results as reported under GAAP. Because of these
limitations, Adjusted EBITDA should not be considered as a
measure of discretionary cash available to us to invest in the
growth of our business. We compensate for these limitations by
relying primarily on our GAAP results and using Adjusted EBITDA
only as supplemental information. A reconciliation of net income
(loss), the most directly comparable GAAP measure, to Adjusted
EBITDA is presented below:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Combined
|
|
|
Successor
|
|
|
Predecessor
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(Non-GAAP)
|
|
|
March 1,
|
|
|
January 1,
|
|
|
|
|
Three months
|
|
|
Three months
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
2007 through
|
|
|
2007 through
|
|
|
|
|
ended March 31,
|
|
|
ended March 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
February 28,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
2007
|
|
|
2007(4)
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Net income (loss)
|
|
$
|
9,997
|
|
|
$
|
(2,357
|
)
|
|
$
|
12,098
|
|
|
$
|
(26,599
|
)
|
|
$
|
(23,605
|
)
|
|
$
|
(27,290
|
)
|
|
$
|
3,685
|
|
|
Interest expense (income), net
|
|
|
3,870
|
|
|
|
4,560
|
|
|
|
16,546
|
|
|
|
29,523
|
|
|
|
28,657
|
|
|
|
28,775
|
|
|
|
(118
|
)
|
|
Income tax expense (benefit)
|
|
|
3,885
|
|
|
|
(836
|
)
|
|
|
3,831
|
|
|
|
(4,277
|
)
|
|
|
(6,299
|
)
|
|
|
(8,913
|
)
|
|
|
2,614
|
|
|
Depreciation and amortization
|
|
|
9,043
|
|
|
|
9,437
|
|
|
|
40,615
|
|
|
|
34,210
|
|
|
|
33,235
|
|
|
|
28,813
|
|
|
|
4,422
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
EBITDA
|
|
|
26,795
|
|
|
|
10,804
|
|
|
|
73,090
|
|
|
|
32,857
|
|
|
|
31,988
|
|
|
|
21,385
|
|
|
|
10,603
|
|
|
Unrealized foreign currency exchange (gains) losses
|
|
|
(4,836
|
)
|
|
|
367
|
|
|
|
(9,347
|
)
|
|
|
20,304
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Restructuring and impairment
|
|
|
2,047
|
|
|
|
1,035
|
|
|
|
26,395
|
|
|
|
15,513
|
|
|
|
114
|
|
|
|
114
|
|
|
|
—
|
|
|
Loss on interest rate swaps, net
|
|
|
709
|
|
|
|
600
|
|
|
|
2,541
|
|
|
|
9,311
|
|
|
|
2,275
|
|
|
|
2,275
|
|
|
|
—
|
|
|
Equity in earnings of affiliate
|
|
|
(4
|
)
|
|
|
(150
|
)
|
|
|
(613
|
)
|
|
|
(380
|
)
|
|
|
(273
|
)
|
|
|
(189
|
)
|
|
|
(84
|
)
|
|
Transaction costs(1)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,316
|
|
|
|
10,271
|
|
|
|
10,271
|
|
|
|
—
|
|
|
Management fees (2)
|
|
|
495
|
|
|
|
351
|
|
|
|
1,538
|
|
|
|
1,990
|
|
|
|
1,295
|
|
|
|
1,295
|
|
|
|
—
|
|
|
Transition costs(3)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
2,984
|
|
|
|
6,667
|
|
|
|
6,575
|
|
|
|
92
|
|
|
Gain on Abieta acquisition
|
|
|
—
|
|
|
|
(2,151
|
)
|
|
|
(2,151
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Gain on settlement with International Paper
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,316
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Selling, general and administrative severance
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
3,121
|
|
|
|
2,235
|
|
|
|
2,235
|
|
|
|
—
|
|
|
Third-party advisor fees
|
|
|
2,135
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,573
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Consulting services
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
3,794
|
|
|
|
7,616
|
|
|
|
7,616
|
|
|
|
—
|
|
|
Gain on debt extinguishment
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,901
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other items
|
|
|
37
|
|
|
|
187
|
|
|
|
3,722
|
|
|
|
2,241
|
|
|
|
4,710
|
|
|
|
3,297
|
|
|
|
1,413
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Adjusted EBITDA
|
|
$
|
27,378
|
|
|
$
|
11,043
|
|
|
$
|
93,859
|
|
|
$
|
92,723
|
|
|
$
|
66,898
|
|
|
$
|
54,874
|
|
|
$
|
12,024
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
14
|
|
|
|
(1) |
|
Transaction costs were related to the Acquisition. These costs
included legal, tax, IT, professional fees and Rhône
Capital transaction fees. |
| |
|
(2) |
|
Management fees are paid to Rhône Group L.L.C., an
affiliate of Rhône Capital, for corporate-level support
activities provided to us. These management fees will no longer
be paid following the completion of the offering. |
| |
|
(3) |
|
Transition costs included fees paid to International Paper in
connection with the Acquisition under the transition services
agreement (governing interim services provided by International
Paper), IT consulting fees, costs for infrastructure build-out
(such as IT and phone systems, treasury function setup,
HR/payroll) and the cost of a carve-out audit, among other items. |
| |
|
(4) |
|
Adjusted EBITDA presented in the predecessor period was
calculated on the same basis as Adjusted EBITDA presented in the
successor periods pursuant to our credit agreements. |
Free Cash
Flow
Free cash flow is not a GAAP concept. We present free cash flow
because our management considers it to be a useful, supplemental
indicator of our liquidity. When measured over time, free cash
flow provides supplemental information to investors concerning
our operating results and our ability to generate cash flows to
satisfy mandatory debt service requirements and make other
non-discretionary expenditures.
Our management believes that consideration of free cash flow
should only be supplemental because free cash flow has
limitations as an analytical financial measure. Our management
compensates for these limitations by relying primarily on our
results under GAAP to evaluate our liquidity and by considering
independently the economic effects of the foregoing items that
are not reflected in free cash flow. As a result of these
limitations, free cash flow should not be considered as a
substitute for other measures of liquidity reported in
accordance with GAAP, including net cash provided by (used in)
operating activities, net cash provided by (used in) investing
activities, net cash provided by (used in) financing activities
or change in cash and cash equivalents. A reconciliation of net
cash provided by (used in) operating activities to free cash
flow, the most directly comparable GAAP measure, is presented
below:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Combined
|
|
|
Successor
|
|
|
Predecessor
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(Non-GAAP)
|
|
|
March 1, 2007
|
|
|
January 1, 2007
|
|
|
|
|
Three months
|
|
|
Three months
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
through
|
|
|
|
|
ended March 31,
|
|
|
ended March 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
February 28,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
2007
|
|
|
2007
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Net cash provided by (used in) operating activities
|
|
$
|
2,507
|
|
|
$
|
11,100
|
|
|
$
|
117,325
|
|
|
$
|
20,841
|
|
|
$
|
32,934
|
|
|
$
|
45,022
|
|
|
$
|
(12,088
|
)
|
|
Less:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Purchases of property, plant and equipment
|
|
|
(4,866
|
)
|
|
|
(6,677
|
)
|
|
|
(22,993
|
)
|
|
|
(34,719
|
)
|
|
|
(22,846
|
)
|
|
|
(18,248
|
)
|
|
|
(4,598
|
)
|
|
Software spending
|
|
|
(3,761
|
)
|
|
|
(3,268
|
)
|
|
|
(13,404
|
)
|
|
|
(142
|
)
|
|
|
(1,642
|
)
|
|
|
(1,642
|
)
|
|
|
—
|
|
|
Proceeds from disposals of property, plant and equipment
|
|
|
—
|
|
|
|
690
|
|
|
|
875
|
|
|
|
212
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other
|
|
|
696
|
|
|
|
—
|
|
|
|
—
|
|
|
|
356
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Free cash flow
|
|
$
|
(5,424
|
)
|
|
$
|
1,845
|
|
|
$
|
81,803
|
|
|
$
|
(13,452
|
)
|
|
$
|
8,446
|
|
|
$
|
25,132
|
|
|
$
|
(16,686
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash (used in) provided by investing activities
|
|
|
(7,931
|
)
|
|
|
(18,027
|
)
|
|
|
(44,294
|
)
|
|
|
(34,293
|
)
|
|
|
(501,929
|
)
|
|
|
(497,331
|
)
|
|
|
(4,598
|
)
|
|
Net cash provided by (used in) financing activities
|
|
|
2,202
|
|
|
|
(1,220
|
)
|
|
|
(63,771
|
)
|
|
|
12,768
|
|
|
|
498,843
|
|
|
|
488,547
|
|
|
|
10,296
|
|
15
RISK
FACTORS
An investment in our common shares involves a high degree of
risk. You should carefully consider the following information,
together with other information in this prospectus, before
buying our common shares. If any of the following risks or
uncertainties occur, our business, financial condition and
results of operations could be materially and adversely
affected, the trading price of our common shares could decline
and you may lose all or a part of your investment in our common
shares.
General Business
Risks
Conditions in
the Global Economy and Capital Markets May Adversely Affect Our
Results of Operations, Financial Condition and Cash
Flows.
Our products are sold in markets that are sensitive to changes
in general economic conditions. Downturns in general economic
conditions can cause fluctuations in demand for our products,
product prices, volumes and margins. A decline in the demand for
our products or a shift to lower margin products due to
deteriorating economic conditions would adversely affect sales
of our products and our profitability and could also result in
impairments of certain of our assets.
Our business and operating results have been and will continue
to be affected by the global recession, including the turbulence
in the credit markets, dislocations in the real estate markets,
fluctuating commodity prices, volatile exchange rates and other
challenges currently affecting the global economy and our
customers. If the current global recession continues for a
significant period of time or further deteriorates, our results
of operations, financial condition and cash flows would be
materially adversely affected.
Demand for
Many of Our Products is Cyclical, and We May Experience
Prolonged Depressed Market Conditions for Our Products, Which
Would Decrease Our Net Sales and Operating
Margins.
Many of our products are ultimately used in end markets which
are cyclical in nature, such as housing, automotive, printed
media, road construction and oil and natural gas drilling. We
primarily sell our products to other manufacturers who use our
products as intermediates and performance modifiers. The housing
market directly affects demand for our products through the
demand for oil-based paints, as well as flooring and
construction adhesives. The automotive market affects demand for
tires, metal working fluids, automotive adhesives in new
automobile construction and after-market parts such as air
filters. The printed media market, such as advertising and
magazine publication, is subject to consumer and discretionary
spending trends, while the road construction market is dependent
on government spending, which affects sales of our products used
in road safety striping. Certain of our products are used in oil
and natural gas drilling applications, the demand for which is
dependent on the overall energy markets. Any adverse development
that negatively affects any or all of these industries could
cause a material decrease in our net sales and operating margins.
Our Business
is Subject to Seasonality That May Affect Our Quarterly
Operating Results and Impact the Market Price of Our Common
Shares.
Seasonal changes and weather conditions typically affect the
inks and roadmarking end-use markets. For example, the demand
for our ink products is typically highest in the third quarter
of the year due to increased demand for holiday catalog
printing. Demand for roadmarking products is typically higher
during the warm weather months and can be affected by adverse
weather conditions during those periods. Because seasonal
weather patterns are difficult to predict, we cannot accurately
estimate fluctuations in our quarterly sales in the roadmarking
market in any given year. If reduced demand in the inks and
roadmarking markets cause our operating results to fall below
the periodic expectations of financial analysts or investors,
the market price of our common shares may decline.
16
We Generally
do not have Long-Term Contracts with Our Customers, and the Loss
of Customers Could Adversely Affect Our Sales and
Profitability.
Our five largest customers accounted for approximately 20% of
our net sales in 2009. With some exceptions, our business is
based primarily upon individual sales orders with our customers.
As such, our customers could cease buying our products from us
at any time, for any reason, with little or no recourse. If a
major customer or multiple smaller customers elected not to
purchase products from us, our business prospects, financial
condition and results of operations would be materially
adversely affected.
We have
Recorded a Significant Amount of Identifiable Intangible Assets,
But We May Never Realize the Full Value of Those
Assets.
Intangible assets are recorded at fair value on the date of
acquisition and, in accordance with GAAP, will be reviewed at
least annually for impairment. Impairment may result from, among
other things, deterioration in our performance, adverse market
conditions, adverse changes in applicable laws or regulations,
including changes that restrict the activities of or affect the
products and services sold by our business, and a variety of
other factors. The amount of any impairment must be expensed
immediately as a charge to our results of operations. Depending
on future circumstances, it is possible that we may never
realize the full value of our intangible assets. Any future
determination of impairment of our intangible assets would have
an adverse effect on our financial condition and results of
operations.
International
Paper and Other Third Parties Provide Operating and Other
Services to Us Under Agreements that are Important to Our
Business. The Failure of Any of These Parties to Perform Their
Obligations, or the Termination of These Agreements, Would
Adversely Affect Our Operations.
The operation of certain of our facilities is dependent on
services provided by third parties. For example, we have
contracts with International Paper pursuant to which
International Paper provides our Savannah, Georgia facility with
waste water treatment and certain other services. Additionally,
Bay County, Florida provides our Panama City facility with waste
water treatment services. Bay County is currently considering
selling the waste water treatment facility used to treat our
waste water to a private company, and we cannot assure you that
we will be able to have our waste water treated by this facility
in the future. In the event that International Paper, Bay County
or another third party fails to provide services to our
facilities, we may be required to obtain these services from
other third parties at an increased cost or to expend capital to
provide these services ourselves. The expenses associated with
obtaining or providing these services, as well as any
interruption in our operations as a result of the failure of the
third party to provide a particular service, may be significant
and may adversely affect our results of operations.
Additionally, although we own our Savannah, Georgia facilities,
our BLS acidulation facility is located on real property leased
from International Paper pursuant to a long-term lease
agreement, and this property is surrounded by the International
Paper mill. In the event we were to have a dispute with
International Paper regarding the terms of our lease agreement
or we were otherwise unable to fully access or utilize this
portion of our Savannah facility, we could incur higher costs
associated with freight to divert BLS to other locations within
our acidulation network. Long-term outages would require the
expenditure of capital for additional acidulation equipment.
Acquisitions
and Joint Ventures that We Pursue May Present Unforeseen
Integration Obstacles and Costs, Increase Our Leverage and
Negatively Impact Our Performance.
We intend to selectively pursue acquisitions of, and joint
ventures with, related businesses as one element of our growth
strategy. There can be no assurance that we will decide to
pursue acquisitions or joint ventures or whether we will be
successful in completing any such transaction. If
17
such transactions are consummated, the risks described below,
and the risks associated with our business generally, may be
increased. These transactions may require us to assume or incur
additional debt financing, resulting in additional leverage and
a complex debt structure. We may pursue acquisitions of a
significantly larger scale than in the past.
The expense incurred in consummating acquisitions or our failure
to integrate acquired businesses successfully into our existing
businesses could negatively impact our results of operations.
Furthermore, we may not be able to realize any anticipated
benefits from acquisitions or joint ventures. The process of
integrating acquired operations into our existing operations may
result in unforeseen operating difficulties and may require
significant financial and management resources that would
otherwise be available for the ongoing development or expansion
of existing operations. Additional risks associated with our
acquisition and joint venture strategy include:
|
|
|
| |
•
|
potential disruption of our ongoing business and distraction of
management;
|
| |
| |
•
|
unexpected loss of key employees or customers of the acquired
company;
|
| |
| |
•
|
conforming the acquired company’s standards, processes,
procedures and controls with our operations;
|
| |
| |
•
|
coordinating new product and process development;
|
| |
| |
•
|
hiring additional management and other critical personnel;
|
| |
| |
•
|
increasing the scope, geographic diversity and complexity of our
operations;
|
| |
| |
•
|
encountering unforeseen obstacles or costs in the integration of
acquired businesses;
|
| |
| |
•
|
the presence of one or more material liabilities of an acquired
company that are unknown to us at the time of the
acquisition; and
|
| |
| |
•
|
unfavorable reception to the acquisition by our customers.
|
Our Business
Could be Materially and Adversely Affected if We Encounter
Problems in the Operation of a New SAP Financial System that
has Replaced Our Prior Systems.
We are highly dependent on our information systems
infrastructure in order to process orders, track inventory, ship
products in a timely manner, prepare invoices to our customers,
generate financial reports and otherwise carry on our business
in the ordinary course. We recently implemented a new global
enterprise resource planning, or ERP, system that we have
licensed from SAP AG. The new ERP system is designed to
integrate all of our data and processes, including supply chain,
inventory and accounting, into a unified system. We expect the
implementation to be completed in phases during the first half
of 2010. The transition to a new ERP system involves numerous
risks, including difficulties in integrating the system with our
current operations, potential delay in the processing of
customer orders for shipment of products, diversion of
management’s attention away from normal daily business
operations, increased demand on our operations support
personnel, initial dependence on unfamiliar systems while
training personnel in its use, increased operating expenses
resulting from training, conversion and transition support
activities and difficulties in accurately reporting our
financial results on a timely basis. If we experience
significant problems with the implementation of this system, the
resulting disruption could adversely affect our business, sales,
results of operations and financial condition.
Risks Related to
Raw Materials
We are Highly
Dependent On Our Key Raw Materials and Lack of Access to
Adequate Supplies of Our Key Raw Materials Would Adversely
Affect Our Results of Operations.
The availability of raw materials is essential to our business.
Our manufacturing process relies on three key raw materials:
crude tall oil, or CTO, crude sulfate turpentine, or CST and, to
a lesser extent, D-limonene. CTO and CST accounted for
approximately 68% and 64% of our raw material purchases for the
three months ended March 31, 2010 and the year ended
December 31, 2009,
18
respectively. We have entered into long-term supply agreements
with International Paper pursuant to which we agreed to buy, and
International Paper agreed to sell, all of International
Paper’s CTO and CST output in the United States. We also
have the option to acquire CTO and CST produced at future
International Paper paper mills worldwide. We satisfied 20% and
27% of our global CTO and CST purchases pursuant to these
agreements for the three months ended March 31, 2010 and
the year ended December 31, 2009, respectively. We also
maintain agreements with others to supply our raw material needs.
CTO and CST are co-products of the kraft pulping process. As a
result, the supply of CTO and CST is based on the demand for the
paper products produced by our suppliers, not on the demand for
CTO or CST. Accordingly, the global supply of CTO and CST is
inherently constrained by the volume of kraft pulp processing
and neither International Paper nor our other suppliers are
required to supply us with a minimum, or floor, amount of CTO or
CST. In addition, substantially all of the global CTO and CST
supply is consumed by the producers of CTO and CST or sold
pursuant to long-term supply agreements, and CTO and CST are not
generally available on the open market. Furthermore, weather
conditions in the Southeastern United States, northern Europe
and other regions where pine trees are grown have in the past,
and may in the future, affect the availability and quality of
pine trees used in the kraft pulping process and, therefore, the
available supply of CTO and CST. For example, in 2005, a wind
storm caused a significant amount of damage to the forests in
Sweden. The trees that were felled during the storm were
processed by the pulp mills over an extended period of time,
which reduced the quantity of CTO and CST yielded from the kraft
pulping process in 2005 and 2006. Accordingly, there can be no
assurance that CTO and CST will be available in quantities
necessary for us to operate our business.
If any of our suppliers fails to meet its obligations under our
supply agreements or is otherwise unable to provide us with an
adequate supply of our raw materials and we are unable to obtain
raw materials from other suppliers, we would be unable to
produce the quantity of products that we have historically
produced and our results of operations would be adversely
affected.
Additionally, we have in the past had disagreements with
International Paper, our largest supplier, regarding the terms
of our CTO and CST supply agreements. Although we believe we
have resolved these disputes to the satisfaction of all parties,
there can be no assurance that we will not have disputes with
International Paper or another supplier of our raw materials in
the future.
The European
Union’s Directive on the Promotion of the Use of Energy
from Renewable Resources and Similar Legislation in the United
States May Incentivize the Use of CTO and CST as Alternative
Fuels.
In December 2008, the European Parliament and the Council of the
European Union passed a directive on the promotion of the use of
energy from renewable sources, which established a 20% European
Community-wide target for energy consumed from renewable sources
relative to the Community’s gross final consumption of
energy, as well as a 10% target for energy consumed from
renewable sources in the transport sector. In order to reach
these targets, the directive established mandatory targets for
each Member State and requires each Member State to adopt a
national renewable energy action plan by June 2010 setting forth
measures to achieve its mandatory targets.
The directive established sustainability criteria for
alternative fuels that must be satisfied in order for the
consumption of a fuel to count toward a Member State’s
national targets. Although we are engaged in discussions with
the European Parliament, the European Commission and various
Member States regarding the appropriate status and treatment of
CTO and CST under the directive and national renewable energy
action plans, CTO and CST currently satisfy the sustainability
criteria.
If a Member State’s national renewable energy action plan
incentivizes the consumption of CTO or CST (or their precursor
chemicals) as alternative fuels, demand for CTO and CST may
increase. We are aware of at least one company, SunPine AB,
located in Pitéa, Sweden, that is in the process of
completing a new manufacturing facility for the production of
biodiesel from CTO at least in part in reliance on possible
incentives under the directive. This biodiesel manufacturing
project, or other
19
similar future projects elsewhere, will increase the demand for
the raw materials that our business currently depends on.
In addition to these developments in the European Union, various
pieces of legislation regarding the use of alternative fuels
have been introduced in the United States Congress. While this
legislation is in its early stages, final legislation may
incentivize the use of CTO and CST as alternative fuels in the
United States.
Because the supply of CTO and CST is inherently constrained by
the volume of paper pulp processing, any diversion of CTO and
CST (or their precursor chemicals) for use as alternative fuels
would reduce the available supply of CTO and CST. If this
diversion were substantial, it could cause prices for CTO and
CST to increase materially, place us at a competitive
disadvantage relative to our competitors who rely on different
primary raw materials, adversely affect our results of
operations and threaten our ability to operate our business.
Fluctuations
in Costs of Our Raw Materials Could Adversely Affect Our Results
of Operations.
Our results of operations are directly affected by the cost of
our raw materials, particularly CTO and CST. Raw materials
accounted for approximately 60% of our costs of goods sold in
2009, and, accordingly, our gross profit and margins could be
adversely affected by changes in the cost of these raw materials
if we are unable to pass the increases on to our customers.
CTO and CST, in addition to their ability to be distilled into
higher value chemicals, can be burned as alternative fuels. Some
paper mills have historically burned CTO to meet their energy
needs rather than sell the CTO they produce to third parties.
Additionally, because of its inherent fuel properties, we
compete for CTO with energy companies and biodiesel
manufacturers, as well as other pine chemical companies. As
discussed above, certain countries have proposed incentivizing
the use of CTO and CST as alternative fuels, which would cause
prices for CTO and CST to increase and place us at a competitive
disadvantage relative to companies that utilize CTO and CST as
an energy source. This could also result in a decrease in
available supply of CTO and CST.
Furthermore, the cost of CTO is driven by energy prices
generally, in addition to supply/demand balance and product
quality, which means that the cost of CTO can fluctuate in
accordance with capacity additions or reductions or significant
facility operating reductions within the CTO market as well as
the broader energy industry.
We cannot assure you that we will be able to pass raw material
price increases through to our customers, and any price increase
that we cannot pass on to our customers will adversely affect
our results of operations.
Increases in
Energy Costs Could have an Adverse Effect on Our Results of
Operations.
Energy purchases constituted approximately 9% and 8% of our cost
of goods sold for the three months ended March 31, 2010 and
the year ended December 31, 2009, respectively. Increases
in energy costs, unless passed on to our customers, would
adversely affect our results of operations. In addition, rising
energy costs may increase our raw material costs. If energy
prices increase significantly, our business or results of
operations may be adversely affected. In addition, rising energy
costs negatively impact our customers and the demand for our
products. These risks will be heightened if our customers or
production facilities are in locations experiencing severe
energy shortages.
Risks Related to
Our Indebtedness
Our
Substantial Indebtedness Could Adversely Affect Our Ability to
Raise Additional Capital to Fund Our Operations, Limit Our
Ability to React to Changes in the Economy or Our Industry and
Prevent Us From Fulfilling Our Obligations Under Our Existing or
Future Indebtedness.
We have substantial indebtedness. As of March 31, 2010, our
total consolidated indebtedness was $331.6 million,
including $327.4 million outstanding under our credit
agreements. As of March 31,
20
2010, on an as adjusted basis after giving effect to this
offering and the use of the proceeds therefrom (assuming that we
sell the shares for a per share price of
$ , the midpoint of the price range
set forth on the cover page of this prospectus), we would have
had $ million of outstanding
indebtedness, including
$ million under our First
Lien Credit Agreement and
$ million outstanding under
our Second Lien Credit Agreement. In fiscal 2010, on an as
adjusted basis, after giving effect to this offering and the use
of the proceeds therefrom, our cash debt service is expected to
be approximately $ million
(including $ million of short
term debt maturities) based on interest rates as
of ,
2010, of which $ million
represents principal and
$ million represents interest.
Our substantial indebtedness could have important consequences
for you, including the following:
|
|
|
| |
•
|
it may limit our ability to obtain additional financing to fund
working capital, capital expenditures, product development,
dividend payments, debt service requirements, strategic
initiatives or other purposes;
|
| |
| |
•
|
it may limit our flexibility in planning for, or reacting to,
changes in our operations or market conditions, and because we
are more highly leveraged than some of our competitors, we may
be at a competitive disadvantage;
|
| |
| |
•
|
it may make us more vulnerable to further downturns in our
industry or the economy;
|
| |
| |
•
|
a substantial portion of our cash flow from operations will be
dedicated to the repayment of principal and interest on our
indebtedness and will not be available for other purposes;
|
| |
| |
•
|
it may limit our ability to acquire other businesses or assets;
and
|
| |
| |
•
|
it may limit our ability to refinance our current indebtedness.
|
Our ability to make scheduled payments on our debt obligations
and to repay our debt obligations as they come due depends on
our financial condition and operating performance as well as
general financial, business and market conditions and other
factors that are beyond our control. There can be no assurances
that our business will generate sufficient cash flow from
operations or that we will be able to borrow additional funds to
satisfy our debt service obligations and other liquidity needs.
The Terms of
Our Existing Indebtedness May Restrict Our Current and Future
Operations.
The terms of our existing indebtedness contain, and any future
indebtedness we may incur would likely contain, restrictive
covenants that impose significant operating and financial
restrictions on our ability to, among other things:
|
|
|
| |
•
|
declare dividends, make distributions or redeem or repurchase
common shares;
|
| |
| |
•
|
prepay, redeem or repurchase other debt;
|
| |
| |
•
|
incur liens or grant negative pledges;
|
| |
| |
•
|
make loans and investments and enter into acquisitions and joint
ventures;
|
| |
| |
•
|
incur additional indebtedness;
|
| |
| |
•
|
amend or otherwise alter or waive any material rights under any
organizational document or any permitted debt agreement;
|
| |
| |
•
|
make capital expenditures;
|
| |
| |
•
|
engage in mergers, acquisitions and asset sales;
|
| |
| |
•
|
engage in sale and lease-back transactions;
|
| |
| |
•
|
conduct transactions with affiliates;
|
21
|
|
|
| |
•
|
alter the nature of our business; and
|
| |
| |
•
|
change our fiscal quarter or fiscal year.
|
In addition, our credit agreements require us to achieve certain
financial results and maintain compliance with specified
financial ratios, which become more restrictive over time. A
failure to comply with these covenants could result in an event
of default under our credit agreements. In the event of any
default under our credit agreements, the lenders thereunder may:
|
|
|
| |
•
|
discontinue lending under the revolving credit facility under
our First Lien Credit Agreement;
|
| |
| |
•
|
elect to declare all borrowings outstanding, together with
accrued and unpaid interest and fees, to be due and payable;
|
| |
| |
•
|
foreclose on our collateral; and
|
| |
| |
•
|
force us to file for bankruptcy protection.
|
See “Description of Our Indebtedness”.
Our
Variable-Rate Indebtedness Subjects Us to Interest Rate Risk,
Which Could Cause Our Annual Debt Service Obligations to
Increase Significantly.
Certain of our indebtedness, including indebtedness under our
credit agreements, are or will be at variable rates of interest
and expose us to interest rate risk. See “Description of
Our Indebtedness”. If interest rates increase, our debt
service obligations on the variable rate indebtedness would
increase even though the amount borrowed remained the same.
Assuming the amount of variable rate indebtedness remains the
same, after giving effect to the offering and the use of the
proceeds therefrom, an increase of 1% in the interest rates
payable on our variable rate indebtedness would increase our
2010 estimated debt service requirements by approximately
$ million.
Despite Our
Substantial Indebtedness, We May Still be Able to Incur
Significantly More Debt, Which Could Intensify the Risks
Relating to Our Indebtedness Described Above.
The terms of the instruments governing our indebtedness
restrict, but do not prohibit, our ability to incur substantial
additional indebtedness in the future. As of March 31,
2010, on an as adjusted basis, after giving effect to this
offering and the use of proceeds therefrom, we would have had
$60.0 million available for additional borrowing under the
revolving credit facility under our First Lien Credit Agreement,
and the covenants under our credit agreements would allow us to
borrow a significant amount of additional indebtedness. The more
leveraged we become, the more we, and in turn our security
holders, become exposed to the risks described above under
“— Our Substantial Indebtedness Could Adversely
Affect Our Ability to Raise Additional Capital to Fund Our
Operations, Limit Our Ability to React to Changes in the Economy
or Our Industry and Prevent Us From Fulfilling Our Obligations
Under Our Existing or Future Indebtedness”.
Risks Related to
Environmental and Safety Matters
Compliance
with Extensive Environmental, and/or Health and Safety
Regulations Could Be Costly, Subject Us to Liability for Fines
or Damages, and/or Require Us to Modify Our Operations or
Products.
Our operations are subject to extensive environmental, health
and safety, or EHS, laws and regulations at national,
international and local levels in each jurisdiction where we
operate. Such laws and regulations include those governing the
discharge of pollutants into the air and water, the use,
management and disposal of hazardous materials and wastes, the
investigation and cleanup of contamination, exposure to
chemicals and other hazards, the control of resin dust and
occupational health and safety. We have incurred, and will
continue to incur, operating costs and capital expenditures to
comply with these laws and regulations. In 2009, we incurred
capital expenditures of
22
$1.3 million for EHS improvements, and we expect to incur
capital expenditures of $2.7 million for EHS improvements
in 2010. Future developments, including the enactment of new or
more stringent laws or regulations could require us to make
additional unplanned environmental expenditures.
Violations of these laws and regulations or the permits required
for our operations could result in restrictions on our operating
activities or the imposition of substantial fines, penalties,
civil or criminal proceedings, third-party property damage
claims (including natural resource damages claims), personal
injury claims, reputational harm, cleanup costs or obligations
or other losses or costs.
Compliance with EHS laws generally increases the cost of
production, including registration and approval requirements,
the costs of transportation and storage of raw materials and
finished products, as well as the costs of the storage and
disposal of wastes, and such costs could have a material adverse
effect on our results of operations. Furthermore, environmental
laws are subject to change and have tended to become stricter
over time. In particular, regulatory efforts to address climate
change and greenhouse gas emissions could affect our cost of
production, the availability of raw feedstock or product sales.
Such changes in environmental laws or their interpretation, or
the enactment of new environmental laws, could result in
materially increased capital expenditures and compliance costs.
Regulation of
Exposure to Certain Process Chemicals Could Require Material
Expenditures or Changes in Our Operations.
Certain regulations applicable to our operations, including the
Occupational Safety and Health Act and the Toxic Substances
Control Act in the United States and the Registration,
Evaluation and Authorization of Chemicals, or REACH, directive
in Europe, prescribe limits restricting exposure to a number of
chemicals used in our operations, including formaldehyde and
nonylphenol, a raw material used in our Niort, France facility
in the manufacture of phenolic ink resins. Future studies on the
health effects of chemicals used in our operations, including
nonylphenol and bisphenol A, which is used in our rosin-based
ink resins, may result in additional regulation or new
requirements in the United States, Europe and elsewhere which
further restrict or prohibit the use of, and exposure to, these
chemicals. Additional regulation of or requirements for these or
other chemicals could require us to change our operations, and
these changes could affect the quality or types of products we
manufacture and materially increase our costs.
We May be
Subject to Cleanup Obligations, Lawsuits and Other Claims
Arising Out of Environmental Contamination or Personal Injuries
Associated with Chemical Manufacturing.
Many of our facilities have environmental contamination. The
investigation and remediation of contamination at our
facilities, including the discovery of contamination arising
from historical industrial operations at our current and former
properties, or at other properties impacted by our operations or
to which our wastes were sent for disposal, may expose us to
cleanup obligations and other damages which could be costly and
may adversely affect our financial condition and results of
operations. Under various environmental laws in the United
States and other countries where we operate, a current or prior
owner or operator of a facility may be liable for the entire
cost of remediation of contamination at the facility or at
locations where wastes were sent for disposal, in many cases
without regard to whether the owner or operator knew of, or
caused, the contamination.
For example, we are aware of soil and groundwater contamination
at our Sandarne, Sweden facility, as well as costs that we will
need to incur in connection with the closure of a landfill at a
former Stora Enso site in Sandarne. Though we do not presently
expect the costs associated with these matters to be material,
it is possible that future unexpected costs associated with
these or any other investigations and remediations could
adversely affect our financial condition and results of
operations.
We face the risk that individuals could seek damages for
personal injury due to exposure to chemicals at our facilities
or elsewhere, or to chemicals or wastes otherwise owned,
controlled or
23
disposed of by us. We may be subject to claims with respect to
workplace exposure, workers’ compensation, claims by
neighbors or others alleging personal injury or property damage
in connection with our operations or waste disposal. The costs
to resolve such claims could be material.
Chemical
Manufacturing is Inherently Hazardous, Which Could Result in
Accidents that Disrupt Our Operations or Expose Us to
Significant Losses or Liabilities.
The hazards associated with chemical manufacturing and the
related storage and transportation of raw materials, products
and wastes are inherent in our operations and the operations of
other occupants with whom we share manufacturing sites. These
hazards could lead to an interruption or suspension of
operations and have an adverse effect on the productivity and
profitability of a particular manufacturing facility or on the
Company as a whole. These potential risks include, but are not
limited to:
|
|
|
| |
•
|
pipeline and storage tank leaks and ruptures;
|
| |
| |
•
|
explosions and fires;
|
| |
| |
•
|
inclement weather and natural disasters, including hurricanes in
the Southeastern United States and other extreme weather
conditions relating to global climate change;
|
| |
| |
•
|
terrorist attack;
|
| |
| |
•
|
mechanical failures; and
|
| |
| |
•
|
chemical spills and other accidental discharges or releases of
toxic or hazardous substances or gases.
|
These hazards may result in personal injury and loss of life,
damage to property and contamination of the environment, which
may result in a suspension of operations and the imposition of
civil or criminal penalties or fines, expenses for remediation
and damage claims brought by governmental entities or third
parties. The loss or shutdown of operations over an extended
period of time at any of our major operating facilities could
have a material adverse effect on our financial condition and
results of operations. For example, in April 2008, a fire
occurred in the refinery portion of our Oulu, Finland facility
due to the failure of a hot oil pump, and in September 2009 a
fire occurred at our Sandarne, Sweden facility as the result of
welding work being performed by an outside contractor. While the
fire at our Sandarne facility had a limited impact on our
production or ability to service our customers, the Oulu fire
required us to shut down that facility for approximately one
month, resulted in property damage of €1.2 million and
caused us to incur a business interruption expense
€4.1 million, including a €2.6 million loss
of profits resulting from lost sales.
In order to mitigate these potential losses and liabilities, we
maintain property, business interruption and casualty insurance
of the types and in the amounts that we believe are customary
for the industry, and the majority of the damage and loss
resulting from the fire at our Oulu facility was covered by our
insurance. However, we are not fully insured against all
potential hazards incidental to our business, and we may not
recover the full amount of a loss or liability under the terms
of our insurance policies.
Legal and
Regulatory Risks
Regulatory and
Statutory Changes Applicable to Us or Our Customers Could
Adversely Affect Our Financial Condition and Results of
Operations.
We and many of the applications for the products in the end-use
markets in which we sell our products are regulated by various
national and local rules, laws and regulations. Changes in any
of these areas could result in additional compliance costs,
seizures, confiscations, recall or monetary fines, any of which
could prevent or inhibit the development, distribution and sale
of our products. For
24
example, changes in environmental regulations restricting the
use of sterols could cause a decline in sales to producers of
that product.
We are Subject
to Customs, International Trade, Export Control, Antitrust,
Zoning and Occupancy and Labor and Employment Laws That Could
Require Us to Modify Our Current Business Practices and Incur
Increased Costs.
We are subject to numerous regulations, including customs and
international trade laws, export control, antitrust laws and
zoning and occupancy laws that regulate manufacturers generally
and/or
govern the importation, promotion and sale of our products, the
operation of factories and warehouse facilities and our
relationship with our customers, suppliers and competitors. If
these regulations were to change or were violated by our
management, employees, suppliers, buying agents or trading
companies, the costs of certain goods could increase, or we
could experience delays in shipments of our goods, be subject to
fines or penalties, or suffer reputational harm, which could
reduce demand for our products and hurt our business and
negatively impact results of operations.
Similarly, these laws affect the availability and pricing of our
raw materials and competing products. For example, we benefit
from certain trade protections, including anti-dumping
protection. If we were to lose these protections, our results of
operations could be adversely affected. Changes in these laws or
regulations may increase the cost of our raw materials or lower
the cost of a competing product, which could hurt our business
and negatively impact results of operations.
Legal requirements are frequently changed and subject to
interpretation, and we are unable to predict the ultimate cost
of compliance with these requirements or their effects on our
operations. We may be required to make significant expenditures
or modify our business practices to comply with existing or
future laws and regulations, which may increase our costs and
materially limit our ability to operate our business.
Due to the
Nature of Our Business and Products, We May Be Liable for
Damages Arising Out of Product Liability Claims.
The sale of our products involves the risk of product liability
claims. For example, some of the chemicals or substances that
are used in our businesses, including alkyl phenols such as
bisphenol A and nonylphenol, and flammable solvents such as
toluene, xylene and alcohols, as well as rosin, formaldehyde and
resin dust, have been identified as having potentially harmful
health effects.
In addition, we may be liable for damages based on product
liability claims brought against our customers in our end-use
markets. Many of our products provide critical performance
attributes to our customers’ products that are ultimately
sold to consumers who could potentially bring product liability
suits in which we could be named as a defendant. If a person
were to bring a product liability suit against one of our
customers, this customer may attempt to seek contribution from
us. A person may also bring a product liability claim directly
against us. A successful product liability claim or series of
claims against us in excess of our insurance coverage limits,
for which we are not otherwise indemnified, could have a
material adverse effect on our financial condition or results of
operations. While we endeavor to protect ourselves from such
claims and exposures in our contractual negotiations, there can
be no assurance that our efforts in this regard will ultimately
protect us from any such claims.
Risks Related to
Worldwide Operations
Our
Substantial Worldwide Operations Subject Us to Risks not Faced
by Competitors that are Based in or that Operate Solely in the
United States, Including Legislative, Judicial, Economic,
Political, Regulatory, Labor and Tax Conditions.
We are a Bermuda company with a significant portion of our
manufacturing operations located outside the United States. In
2009, our net sales outside the United States represented
approximately
25
59% of our total net sales and included sales to more than 80
countries. In addition, we have production facilities located in
Sandarne, Sweden, Oulu, Finland, Chester-le-Street, United
Kingdom, Niort, France and Gersthofen, Germany. Accordingly, our
business is subject to the differing legal, political, social
and regulatory requirements and economic conditions specific to
the countries or regions in which we operate, which could
materially adversely affect our financial performance. Risks
inherent in world-wide operations include, but are not limited
to, the following:
|
|
|
| |
•
|
difficulty in enforcing agreements through the differing legal
systems of the countries in which we operate;
|
| |
| |
•
|
customers in the various countries in which we operate may have
long payment cycles;
|
| |
| |
•
|
countries in which we operate may impose withholding taxes or
otherwise tax our income from our operations, impose tariffs or
adopt other restrictions on trade and investment outside,
including currency exchange controls;
|
| |
| |
•
|
restrictions on our ability to repatriate earnings from
countries in which we operate;
|
| |
| |
•
|
difficulty in enforcing intellectual property rights and our
ability to protect our intellectual property;
|
| |
| |
•
|
high rates of inflation;
|
| |
| |
•
|
fluctuations in exchange rates, including currency devaluation,
may affect product demand and may adversely affect our
profitability;
|
| |
| |
•
|
increased costs of transportation or shipping;
|
| |
| |
•
|
risk of nationalization of private enterprises;
|
| |
| |
•
|
changes in general economic and political conditions in the
countries in which we operate;
|
| |
| |
•
|
changes in laws or regulatory requirements, including those
governing environmental protection, export duties and quotas;
|
| |
| |
•
|
difficulty with staffing and managing widespread
operations; and
|
| |
| |
•
|
required compliance with laws and regulations of numerous
jurisdictions.
|
A portion of our outstanding debt under our First Lien Credit
Agreement is denominated in Euros, and a portion of this debt is
attributable to Arizona Chemical AB, whose functional currency
is the Swedish Kronor. Accordingly, fluctuations in the exchange
rate of the Swedish Kronor to the Euro expose us to both
realized and unrealized foreign currency losses. For example, in
2008, we recognized an unrealized loss of $20.3 million due
to a stronger Euro against the Swedish Kronor, and in 2009, we
recognized $9.3 million and $0.4 million of unrealized
gain and realized loss, respectively. For the three months ended
March 31, 2010, we recognized $4.8 million and $0.3 million of
unrealized gain and realized gain, respectively.
Our overall success as a global business depends, in part, upon
our ability to succeed in differing legal, regulatory, economic,
social, tax and political conditions. We may not continue to
succeed in developing and implementing policies and strategies
that will be effective in each location where we do business.
Our growth strategy includes expansion into new and existing
international markets, and we expect that our operations outside
the United States will account for a larger portion of our
revenue and profitability in the future. As a result, each of
the foregoing risks is likely to take on increased significance.
Currency
Translation Risk and Currency Transaction Risk May Negatively
Affect Our Net Sales, Cost of Sales and Operating Margins and
Could Result in Exchange Losses.
We conduct our business and incur costs in the local currency of
most countries in which we operate. In the three months ended
March 31, 2010 and the year ended December 31, 2009,
our net
26
sales recorded in currencies other than U.S. dollars
represented approximately 51% of our total net sales.
Accordingly, our results of operations are recorded in the
relevant local currency and then translated to
U.S. dollars, our functional currency, at the applicable
currency exchange rate for inclusion in our financial
statements. Changes in exchange rates between those foreign
currencies and the U.S. dollar will affect our net sales,
cost of sales and operating margins and could result in exchange
losses. In addition to currency translation risks, we incur
currency transaction risk whenever one of our operating
subsidiaries enters into either a purchase or a sales
transaction using a different currency from the currency in
which it records revenues. While we do not currently hedge our
exchange rate exposures, we may do so in the future. However,
any hedging transactions we enter into may not be effective or
could result in foreign exchange hedging loss. The impact of
future exchange rate fluctuations on our results of operations
cannot be accurately predicted.
Given the volatility of exchange rates, we may not be able to
effectively manage our currency transaction
and/or
translation risks, and any volatility in currency exchange rates
may have an adverse effect on our financial condition, cash
flows and profitability. We operate our business in countries
that historically have been and may continue to be susceptible
to recessions or currency devaluation, including Mexico and
Turkey. In addition, as we expand our business in emerging
markets, particularly China and Russia, the uncertain regulatory
environment relating to currency policy in these countries could
have a negative impact on our operations there.
Risks Related to
Competition
We Face
Competition From Other Chemical Companies, Which Could Force Us
to Lower Our Prices, Thereby Adversely Affecting Our Business,
Operating Margins, Financial Condition, Cash Flows and
Profitability.
The markets in which we operate are highly competitive, and this
competition could harm our business, operating margins,
financial condition, cash flows and profitability. Our
competitors include major international producers as well as
smaller regional competitors. Current and anticipated future
consolidation among our competitors and customers may cause us
to lose market share as well as put downward pressure on
pricing. Furthermore, there is a trend in the chemical industry
toward relocation of manufacturing facilities to lower-cost
regions such as Asia. Such relocation may permit some of our
competitors to lower their costs and improve their competitive
position. Some of our competitors are larger, have greater
financial resources and have less debt than we do. As a result,
those competitors may be better able to withstand a change in
conditions within our industry and throughout the economy as a
whole. If we do not compete successfully, our business,
operating margins, financial condition, cash flows and
profitability could be adversely affected.
Competition
From Producers of Substitute Products Could Lead to Declines in
Our Net Sales Attributable to These Products.
We face competition from a number of products that are potential
substitutes for our products. In particular, hydrocarbon and
gum-based resins compete with tall oil-based resins in the
adhesives and inks markets, and animal and vegetable-based fatty
acids compete with TOFA. We have in certain cases been subject
to pricing pressure from Chinese manufacturers of gum rosins,
and our hydrocarbon competitors have introduced
metallocene-based products that compete directly with many of
our adhesive tackifiers. Considerable growth in these
substitutes for our products could adversely affect our market
share, net sales and results of operations.
We face substantial risk that certain events, such as new
product development by our competitors, changing customer needs,
production advances for competing products, price changes in raw
materials, our failure to secure patents or the expiration of
patents, could result in declining demand for our products as
our customers switch to substitute products or undertake
manufacturing of such products on their own. If we are unable to
develop and produce or market our products to effectively
compete against our competitors, our results of operations may
materially suffer.
27
Failure to
Develop New Products will Make Us Less
Competitive.
Our results of operations depend to a significant extent on our
ability to expand our product offerings and to continue to
develop our production processes to be a competitive producer.
We may not be able to continue to develop new products,
re-engineer our existing products successfully or bring our
products to market in a timely manner. While we believe that the
products, pricing and services we offer customers are
competitive, we may not be able to continue to attract and
retain customers.
In addition, our customers may introduce new generations of
their own products or require new technological and increased
performance specifications that would require us to develop
customized products. Innovation or other changes in our
customers’ product performance requirements may also
adversely affect the demand for our products. Our future growth
will depend on our ability to gauge the direction of the
commercial and technological progress in all key end-use
markets, and upon our ability to successfully develop,
manufacture and market products in such changing end-use
markets. If we fail to keep pace with evolving technological
innovations or fail to modify our products in response to our
customers’ needs, then our business, financial condition
and results of operations could be adversely affected as a
result of reduced sales of our products.
Pricing
Pressures by Our Customers and Increased Competition May
Adversely Affect Our Results of Operations.
We have in the past experienced pricing pressure from customers
in certain of the markets in which we compete, particularly
chemical intermediates and inks. While we are directing our
business away from producing low-margin products such as those
sold as chemical intermediates and those sold into the inks
market, we currently derive a significant portion of our
revenues from sales of these chemicals and will continue to do
so for the foreseeable future. Accordingly, pricing pressure on
these products may reduce our profitability and adversely affect
our results of operations.
Risks Related to
Intellectual Property
Our Business
Relies on Intellectual Property and Other Proprietary
Information and Our Failure to Protect Our Rights Could Harm Our
Competitive Advantages with Respect to the Manufacturing of Some
of Our Products.
Our success depends to a significant degree upon our ability to
protect and preserve our intellectual property and other
proprietary information of our business. However, we may be
unable to prevent third parties from using our intellectual
property and other proprietary information without our
authorization or from independently developing intellectual
property and other proprietary information that is similar to
ours, particularly in countries where the laws do not protect
our proprietary rights to the same degree as in the United
States. Others’ use of our intellectual property and other
proprietary information could reduce or eliminate any
competitive advantage we have developed, cause us to lose sales
or otherwise harm our business. If it becomes necessary for us
to litigate to protect these rights, any proceedings could be
burdensome and costly, and we may not prevail.
As a result of developing products and markets with selected
customers, we have six patent families that are jointly-held
with certain of our customers. These patents are in the areas of
tackifiers for adhesives and specialty polyamides for fragrance
formulations. At the time of such product and/or market
development, we entered into written agreements with these
customers that limit the rights of each party with respect to
jointly developed inventions and inventions made with the
confidential information of the other party. These written
agreements were made in exchange for exclusive licenses or
supply agreements, for limited times. The risks of such
joint-development agreements include the risk that our partner
will breach its obligations under the agreement. For example our
partner might breach the agreement and begin making products
that we develop or enter into similar development agreements
with our competitors. In the event that our customer exploits a
jointly-held patent or breaches its obligations under a
joint-development agreement, our business could be
28
materially adversely affected. Another risk is that title to
intellectual property that is managed by an agreement may become
clouded. For example, we or our partner may fail to document a
change in patent ownership that occurs when certain conditions
of an agreement are met. Unclear title or undocumented changes
in title make it more difficult to license or sell such a patent.
Any of our patents or patent applications may not provide us
with any competitive advantage and may be challenged by third
parties. Our competitors also may attempt to design around our
patents or copy or otherwise obtain and use our intellectual
property and other proprietary information. Moreover, our
competitors may already hold or have applied for patents in the
United States or abroad that, if enforced or issued, could
possibly prevail over our patent rights or otherwise limit our
ability to manufacture or sell one or more of our products in
the United States or abroad. From time to time, we oppose the
issuance of patent applications in the United States and other
jurisdictions that we consider overbroad or otherwise invalid to
avoid the risk of being sued for patent infringement. With
respect to our pending patent applications, we may not be
successful in securing patents for these claims. Our failure to
secure these patents may limit our ability to protect inventions
that these applications were intended to cover. The expiration
of a patent can result in increased competition with consequent
erosion of profit margins.
In addition, it is our policy to enter into confidentiality
agreements when appropriate with our employees and third parties
to protect our unpatented proprietary manufacturing expertise,
continuing technological innovation and other trade secrets and
know-how,
but our confidentiality agreements could be breached or may not
provide meaningful protection for our trade secrets or
proprietary manufacturing expertise. Adequate remedies may not
be available in the event of an unauthorized use or disclosure
of our trade secrets, manufacturing expertise and
know-how.
Violations by others of our confidentiality agreements and the
loss of employees who have specialized knowledge and expertise
could harm our competitive position and cause our sales and
operating results to decline as a result of increased
competition. In addition, others may obtain knowledge of our
trade secrets and
know-how
through independent development or other access by legal means.
Failure to obtain or maintain unpatented
know-how and
trade secret protection could adversely affect our competitive
position.
We have registered and applied for certain service marks and
trademarks, and will continue to evaluate the registration of
additional service marks and trademarks, as appropriate. The
applicable governmental authorities may not approve our pending
applications. A failure to obtain trademark registrations in the
countries in which we operate could limit our ability to obtain
and retain our trademarks and impede our marketing efforts in
those jurisdictions. Moreover, third parties may seek to oppose
our applications or otherwise challenge the resulting
registrations. In the event that our trademarks are successfully
challenged, we could be forced to rebrand our products, which
could result in loss of brand recognition and could require us
to devote resources to advertising and marketing new brands.
The failure of our patents, trademarks or confidentiality
agreements to protect our intellectual property and other
proprietary information, including our processes, apparatuses,
technology, trade secrets, trade names and proprietary
manufacturing expertise, methods and compounds, could have a
material adverse effect on our competitive advantages over other
producers.
Our Products
May Infringe the Intellectual Property Rights of Others, Which
May Cause Us to Incur Unexpected Costs or Prevent Us from
Selling Our Products.
Many of our competitors have a substantial amount of
intellectual property that we must continually monitor to avoid
infringement. Although it is our policy and intention not to
infringe valid patents, we cannot guarantee that our processes
and products do not and will not infringe issued patents
(whether present or future) or other intellectual property
rights belonging to others, including, without limitation,
situations in which our products, processes or technologies may
be covered by patent applications filed by other parties. From
time to time, we oppose patent applications that we consider
overbroad or otherwise invalid in order to maintain the
necessary freedom to operate fully in our various business lines
without
29
the risk of being sued for patent infringement. If, however,
patents are subsequently issued on any such applications by
other parties, or if patents belonging to others already exist
that cover our products, processes or technologies, we could,
possibly, be liable for infringement or have to take other
remedial or curative actions to continue our manufacturing and
sales activities with respect to one or more products. We may
also be subject to legal proceedings and claims in the ordinary
course of our business, including claims of alleged infringement
of the patents, trademarks and other intellectual property
rights of third parties by us or our licensees in connection
with their use of our products. Intellectual property litigation
is expensive and time-consuming, regardless of the merits of any
claim, and could divert our management’s attention from
operating our business. If we were to discover that our
processes, technologies or products infringe the valid
intellectual property rights of others, we might need to obtain
licenses from these parties or substantially re-engineer our
products in order to avoid infringement. We may not be able to
obtain the necessary licenses on acceptable terms, or at all, or
be able to re-engineer our products successfully. Moreover, if
we are sued for infringement and lose, we could be required to
pay substantial damages
and/or be
enjoined from using or selling the infringing products or
technology. Any of the foregoing could cause us to incur
significant costs and prevent us from selling our products.
Risks Related to
Employment Matters
Our Future
Success Depends on Our Ability to Retain Our Key
Employees.
We are dependent on the services of our management team to
remain competitive in our industry. This team includes Cornelis
Verhaar, our President and Chief Executive Officer, Frederic
Jung, our Vice President and Chief Financial Officer, Gary Reed,
our Vice President and General Manager — North
America, Juhani Tuovinen, our Vice President and General
Manager — Europe, David Cowfer, our Vice President,
Human Resources and Corporate Communications, and Dick
Stuyfzand, our Vice President and General Counsel.
The loss of any member of our senior management team would have
an adverse effect on us, and although we maintain employment
agreements with several members of our senior management, we
cannot assure you that we will be able to retain our management
team or that members of our management team will not compete
against us in the future.
We May Be
Required to Expend Greater Time and Expenses Than Anticipated or
Than Other Companies Expend in Dealing With Our Employees, Some
of Whom are Unionized, Represented by Workers’ Councils or
are Employed Subject to Local Laws That are Less Favorable to
Employers Than the Laws of the United States.
As of March 31, 2010, approximately 54% of our
671 employees in the United States were unionized and are
covered by collective bargaining agreements. In Europe, nearly
all of our employees are represented by local workers councils
and/or unions. In addition, some of our employees are employed
in countries in which employment laws provide greater bargaining
or other rights to employees than the laws of the
United States. Such favorable employment rights require us
to expend greater time and expense in making changes to
employees’ terms of employment or making staff reductions.
For example, the workers’ councils that represent most of
our employees in Europe must approve any changes in conditions
of employment, including salaries and benefits. A significant
dispute with our employees may divert management’s
attention and otherwise hinder our ability to conduct our
business and achieve planned cost savings.
In addition, changes in United States federal, state and local
minimum wage laws and other laws, including those in foreign
countries, relating to employee benefits could cause us to incur
additional wage and benefits costs, which could negatively
impact our profitability.
30
We Operate
Under Collective Bargaining Agreements with Several Unions, and
We May Experience Labor Unrest and Work Stoppages.
We operate under collective bargaining arrangements with several
unions in our U.S. and
non-U.S. facilities.
Although we believe relations with our unions are generally
satisfactory, we cannot be certain that they will remain
satisfactory. Accordingly, we cannot assure you that we will not
encounter strikes or other types of conflicts with labor unions
or our personnel or that such labor disputes will not have an
adverse effect on us.
Significant
Changes in Pension Fund Investment Performance or Assumptions
Relating to Pension Costs May Increase the Valuation of Pension
Obligations, Alter the Funded Status of Pension Plans and
Increase Our Pension Costs.
Our funding policy for pension plans is to accumulate plan
assets that, over the long run, will approximate the present
value of projected benefit obligations. Our pension cost is
materially affected by the discount rate used to measure pension
obligations, the level of plan assets available to fund those
obligations at the measurement date and the expected long-term
rate of return on plan assets. Significant changes in investment
performance or a change in the portfolio mix of invested assets
can result in corresponding increases and decreases in the
valuation of plan assets, particularly equity securities, or in
a change of the expected rate of return on plan assets. A change
in the discount rate would result in a significant increase or
decrease in the valuation of pension obligations, affecting the
reported funded status of our pension plans as well as the net
periodic pension cost in the following financial years.
Similarly, changes in the expected return on plan assets
assumption can result in significant changes in the net periodic
pension cost of the following financial years.
Our Pension
Plans are Currently Underfunded and We May have to Make
Significant Cash Payments to the Plans, Reducing the Cash
Available for Our Business.
We sponsor various pension plans worldwide that are underfunded
and require significant cash payments. For example, in 2009, we
contributed $4.5 million to our pension plans and, in 2008,
we contributed $2.7 million to our pension plans. We are
expected to contribute at least $4.1 million to our pension
plans in 2010. We may also opt to make additional voluntary
contributions to various pension plans worldwide in 2010.
Additionally, if the performance of the assets in our pension
plans does not meet our expectations, or if other actuarial
assumptions are modified, our contributions for those years
could be even higher than we expect. For example, the combined
asset value of funded pension plans worldwide was
$47.6 million as of December 31, 2009. We expect to
earn a 5.9% investment return on our pension assets. In the
event actual investment returns are 1% lower than expected for
one year, we expect our long-term cash requirements to increase
by $0.5 million. If our cash flow from operations is
insufficient to fund our worldwide pension liability, we may be
forced to reduce or delay capital expenditures, seek additional
capital or seek to restructure or refinance our indebtedness.
As of March 31, 2010, our worldwide pension plans were
underfunded by $14.2 million (based on the actuarial
assumptions consistent with GAAP). Our U.S. pension plan is
subject to the Employee Retirement Income Security Act of 1974,
or ERISA, and our benefit plans in other countries are also
subject to laws governing the funding of retirement benefits.
The applicable authorities in the countries in which we operate
(including, in the United States, the Pension Benefit Guaranty
Corporation under ERISA) have the authority to terminate an
underfunded pension plan under certain circumstances, which may
require us to incur a liability equal to the entire amount of
the underfunding for that plan.
31
Risks Related to
Financial Reporting
Our Management
and Independent Registered Public Accounting Firm in the Past
Determined that There have Been Material Weaknesses and
Significant Deficiencies in Our Internal Controls Over Financial
Reporting. If We Fail to Maintain an Effective System of
Internal Controls Over Financial Reporting, We May not be Able
to Accurately Report Our Financial Results.
During their audit of our financial statements as of and for the
ten months ended December 31, 2007, our independent
registered public accounting firm identified certain reportable
conditions that constituted a material weakness in the internal
controls over financial reporting process. These conditions
included a failure by our corporate finance and accounting
function to thoroughly review our consolidated financial
statements, a lack of active monitoring and oversight by our
corporate finance and accounting function of our European
accounting function and the inability of our corporate finance
and accounting function to complete the timely closing of the
financial accounting books and records and prepare our 2007
financial statements. We remedied these conditions in 2008.
In addition, during their audit of our financial statements as
of and for the year ended December 31, 2008 and the ten
months ended December 31, 2007, our independent registered
public accounting firm identified certain conditions that
constituted significant deficiencies in our internal controls
over financial reporting, including a significant deficiency
relating to our statement of cash flows in 2008 and significant
deficiencies regarding our assessment of fraud risk, journal
entries, accounting policy and internal control documentation,
plant accounting, income taxes and reconciliation review in 2007.
Our audit committee and management team agreed with the matters
identified as the material weakness and significant
deficiencies. Failure to maintain an effective system of
internal controls over financial reporting could have a material
adverse effect on our business, financial condition, results of
operations and our ability to accurately report our financial
results.
If We Are
Unable to Implement the Requirements of Section 404 of the
Sarbanes-Oxley
Act in a Timely Manner, or if We Conclude Our Internal Controls
are not Effective in Other Areas, We May Be Subject to Sanctions
or Investigation by Regulatory Authorities and Incur Additional
Compliance Costs and the Financial Markets May React
Negatively.
We are currently performing the system and process evaluation of
our internal controls over financial reporting in order to allow
management to report on, and our independent auditors to attest
to, our internal controls over financial reporting, as required
by Section 404 of the Sarbanes-Oxley Act of 2002, which we
refer to as Section 404. The requirements of
Section 404 will initially apply to us in connection with
the filing of our second annual report after becoming subject to
the reporting requirements of the Securities and Exchange Act of
1934, as amended (the “Exchange Act”). In connection
with our preliminary evaluation, we have identified areas of
internal controls that may need improvement, such as internal
controls related to the segregation of duties, system access and
user security profiles, operating policies and procedures. We
plan to begin the testing necessary to permit the management
certification and auditor attestation required to comply with
Section 404 in 2011. As we complete the evaluation and
testing required by Section 404, we may identify conditions
that may be categorized as significant deficiencies or material
weaknesses in the future.
As of March 31, 2010, we have incurred $0.1 million in
professional fees in connection with our initial implementation
of the systems and internal control documentation required
pursuant to Section 404.
If we are not able to implement the requirements of
Section 404 in a timely manner, management will not be able
to certify as to the effectiveness of our internal control over
financial reporting and we may be subject to sanctions or
investigation by regulatory authorities, such as the Securities
and Exchange Commission (the “SEC”). As a result,
there could be a negative reaction in the financial markets due
to a loss of confidence in the reliability of our financial
statements. In addition, we may
32
be required to incur additional costs in improving our internal
control system
and/or the
hiring of additional personnel. Any such action could adversely
affect our results of operations.
Risks Related to
Our Common Shares and this Offering
We are
Controlled by Rhône Capital, Whose Interests in Our
Business May Be Different From Yours.
After the completion of this offering, Rhône Capital will
exercise control over more than 50% of our outstanding common
shares. As a result, Rhône Capital will, subject to
applicable law, be able to control the composition of our Board
of Directors and therefore will be able to control actions to be
taken by us and our Board of Directors, including amendments to
our memorandum of association and certain provisions of our
bye-laws, the approval of significant corporate transactions
such as the amalgamation, discontinuance, voluntary liquidation
or sale of our company or our assets, as well as the issuance of
additional shares, alteration of our share capital, the
implementation of share repurchase programs, and the issuance of
dividends.
Additionally, Rhône Capital and its affiliates are in the
business of making investments in companies and may from time to
time acquire and hold interests in businesses that compete
directly or indirectly with us. They may also pursue acquisition
opportunities that may be complementary to our business, and, as
a result, those acquisition opportunities may not be available
to us.
As a
“Controlled Company”, We Will be Exempt From Certain
NYSE Governance Requirements.
Because Rhône Capital and its affiliates will control more
than 50% of our voting power after giving effect to this
offering, we will be considered a “controlled company”
for the purposes of the NYSE listing requirements. As such, we
will be exempt from the NYSE corporate governance requirements
that our board of directors, our compensation committee and our
nominating and corporate governance committee meet the standard
of independence established by those corporate governance
requirements. As a result, our Board of Directors and those
committees will have more directors that would not meet the NYSE
independence standards than they would if those standards were
to apply. The NYSE independence standards are intended to ensure
that directors who meet the independence standard are free of
any conflicting interest that could influence their actions as
directors.
There is no
Existing Public Market for Our Common Shares, and We Do Not Know
if One Will Develop, Which Could Impede Your Ability to Sell
Your Shares and Depress the Market Price of Our Common
Shares.
Prior to this offering, there has been no public market for our
common shares. We cannot predict the extent to which investor
interest will lead to the development of an active and liquid
trading market in our common shares. The failure of an active
trading market to develop could affect your ability to sell your
shares and depress the market price of your shares. The initial
public offering price for our common shares will be determined
by negotiations among us, the selling shareholder and the
representatives of the underwriters. The initial public offering
price of our common shares will be based on numerous factors and
may not be indicative of the market price of the common shares
that will prevail in the trading market. The market price of
your shares may fall below the initial public offering price.
The Market
Price of Our Common Shares May be Volatile, Which Could Cause
the Value of Your Investment to Decline.
The market price of our common shares could fluctuate
significantly, in which case you may not be able to resell your
shares at or above the initial public offering price. Some
companies that have had volatile market prices for their
securities have had securities class action suits filed against
them. If a suit were to be filed against us, regardless of the
outcome, it could result in substantial costs and a diversion of
management’s attention and resources. This could have a
material adverse effect on our business, results of operations
and financial condition.
33
The market price of our common shares may fluctuate in response
to a number of events, including:
|
|
|
| |
•
|
our quarterly operating results;
|
| |
| |
•
|
future announcements concerning our business;
|
| |
| |
•
|
changes in financial estimates and recommendations by securities
analysts;
|
| |
| |
•
|
the number of shares to be publicly traded after this offering;
|
| |
| |
•
|
changes in the availability or cost of raw materials;
|
| |
| |
•
|
changes in energy costs;
|
| |
| |
•
|
actions of competitors;
|
| |
| |
•
|
our involvement in acquisitions, strategic alliances or joint
ventures;
|
| |
| |
•
|
market and industry perception of our success, or lack thereof,
in pursuing our growth strategy;
|
| |
| |
•
|
our ability to develop and market new products on a timely basis;
|
| |
| |
•
|
commencement of, or our involvement in litigation;
|
| |
| |
•
|
dilutive issuances of our shares or the shares of our
subsidiaries;
|
| |
| |
•
|
changes in our board or management;
|
| |
| |
•
|
adoption of new or different accounting standards;
|
| |
| |
•
|
changes in government regulations, including environmental
regulations;
|
| |
| |
•
|
difficulties in managing international operations and the burden
of complying with existing and future domestic and foreign laws;
|
| |
| |
•
|
arrival and departure of key personnel;
|
| |
| |
•
|
general market, economic and political conditions; and
|
| |
| |
•
|
natural disasters, terrorist attacks and acts of war.
|
In addition, in recent years the stock market has experienced
significant price and volume fluctuations. These fluctuations
are often unrelated to the operating performance of particular
companies. The price of our common shares could fluctuate based
upon factors that have little or nothing to do with our company
or its ongoing business operations, and these fluctuations could
materially reduce the market price of our common shares.
Future Sales
or the Possibility or Perception of Future Sales of a
Substantial Amount of Our Common Shares Could Adversely Affect
the Market Price of Our Common Shares.
The market price of our common shares could decline as a result
of sales of a large number of our common shares in the public
market after this offering or the perception that these sales
could occur. The sales, or the possibility that these sales may
occur, also might make it more difficult for us to sell equity
securities in the future at a time and at a price that we deem
appropriate.
Upon consummation of this offering, there will
be common
shares outstanding. All common shares sold in this offering will
be freely transferable without restriction or further
registration under the Securities Act of 1933, as amended (the
“Securities Act”). Of the remaining common shares
outstanding,
will be restricted securities within the meaning of
Rule 144 under the Securities Act, but will be eligible for
resale subject to applicable volume, manner of sale, holding
period and other limitations of Rule 144. We and certain of
our shareholders have agreed to a
“lock-up”,
pursuant to which neither we nor they will sell any shares
without the prior consent of Goldman, Sachs & Co. for
180 days after the date of this prospectus, subject to
certain exceptions and extension under certain circumstances.
Following the expiration of the applicable
lock-up
period, all these common
34
shares will be eligible for future sale, subject to the
applicable volume, manner of sale, holding period and other
limitations of Rule 144.
Any common shares reserved for future issuance under our 2010
Long-Term Incentive Plan could also become eligible for sale in
the public market to the extent permitted by the provisions of
the various vesting agreements and Rule 144 under the
Securities Act.
In addition, the Rhône Funds, International Paper and the
MIVs will have certain registration rights with respect to the
common shares that they will retain following this offering. See
“Shares Eligible for Future Sale” for a
discussion of the common shares that may be sold into the public
market in the future, and “Certain Relationships and
Related Party Transactions — Shareholders
Agreement” for a discussion of registration rights.
We may issue our common shares or other securities, including
preference shares, from time to time as consideration for future
acquisitions and investments. If any such acquisition or
investment is significant, the number of our common shares, or
the number or aggregate principal amount, as the case may be, of
other securities that we may issue may in turn be substantial.
We may also grant registration rights covering those common
shares or other securities in connection with any such
acquisitions and investments.
We do not
Expect to Pay Any Dividends for the Foreseeable
Future.
Other than the distribution we are making to
AZ Chem Investments Partners LP out of the
proceeds of this offering, we do not anticipate paying any
dividends to our shareholders for the foreseeable future. In
addition, because we are a holding company, our ability to pay
dividends depends on our receipt of cash dividends and
distributions from our subsidiaries. The terms of our credit
agreements substantially restrict our ability and the ability of
our subsidiaries to pay dividends. Accordingly, investors must
be prepared to rely on sales of their common shares after price
appreciation to earn an investment return, which may never
occur. Investors seeking cash dividends should not purchase our
common shares. Any determination to pay dividends in the future
will be made at the discretion of our board of directors and
will depend upon our results of operations, financial
conditions, contractual restrictions, restrictions imposed by
applicable law or the SEC and other factors our board deems
relevant.
Arizona
Chemical Ltd. is a Holding Company that does not Conduct Any
Business Operations of its Own, and it Depends on the
Performance of its Subsidiaries and Their Ability to Pay
Dividends and Make Distributions to it.
Arizona Chemical Ltd. is a holding company that does not conduct
any business operations of its own. Arizona Chemical Ltd.’s
principal asset is the equity securities of Arizona Chemical
Luxembourg S.à.r.l. which will, upon completion of this
offering, through its subsidiary Arizona Chem Sweden Holdings
AB, own operating subsidiaries. As a result, the issuer is
dependent upon cash dividends and distributions or other
transfers from its subsidiaries to make dividend payments on its
common shares and to meet its other obligations. The ability of
the issuer’s subsidiaries to pay dividends and make other
payments to the issuer will depend on their operating results
and may be restricted by, among other things, applicable
corporate, tax and other laws and regulations, agreements
entered into by the subsidiaries, such as our credit agreements
and any agreement governing any future indebtedness the issuer
or its subsidiaries may incur.
In addition, because Arizona Chemical Ltd. is a holding company,
your claims as shareholders will be structurally subordinated to
all existing and future liabilities and obligations (whether or
not for borrowed money) of its subsidiaries, including
obligations under existing and future indebtedness. Accordingly,
in the event of the issuer’s dissolution, bankruptcy,
liquidation or reorganization, the issuer’s assets and the
assets of its subsidiaries will be available for distribution to
our shareholders only after all of the issuer’s liabilities
and the liabilities of its subsidiaries have been paid in full.
The
35
issuer would not be able to make distributions to its
shareholders until after the payment in full of the claims of
the creditors of its subsidiaries.
If Securities
or Industry Analysts do not Publish Research or Publish
Inaccurate or Unfavorable Research About Our Business, Our Share
Price and Trading Volume Could Decline.
The trading market for our common shares will depend in part on
the research and reports that securities or industry analysts
publish about us or our business. We do not currently have and
may never obtain research coverage by securities and industry
analysts. If no securities or industry analysts commence
coverage of our company, the trading price for our common shares
would be negatively impacted. If we obtain securities or
industry analyst coverage and if one or more of the analysts who
covers us downgrades our common shares or publishes inaccurate
or unfavorable research about our business, our share price
would likely decline. If one or more of these analysts ceases
coverage of us or fails to publish reports on us regularly,
demand for our common shares could decrease, which could cause
our share price and trading volume to decline.
Through its
Affiliates, One of the Underwriters for this Offering is Also a
Creditor of Our Outstanding Indebtedness, and Therefore has
Interests in this Offering Beyond Customary Underwriting
Discounts and Commissions.
Certain affiliates of Goldman, Sachs & Co. are lenders
under our credit agreements, and we expect to use portions of
the net proceeds of this offering to repay indebtedness
outstanding under our credit agreements. As a result, such
affiliates of Goldman, Sachs & Co. may receive
portions of the proceeds of this offering and have interests
beyond customary underwriting discounts and commissions.
For more information on the use of the proceeds of this
offering, see “Use of Proceeds” and
“Underwriting”.
You will
Experience Immediate and Substantial Dilution as a Result of
this Offering and May Experience Additional Dilution in the
Future.
The initial public offering price of the common shares is
substantially higher than the net tangible book value per
outstanding common share. As a result, you would experience
immediate and substantial dilution of
$ per common share, based on an
assumed offering price of $ per
share, the midpoint of the price range set forth on the cover
page of this prospectus. For additional information, see
“Dilution”.
Risks Related to
Holding Shares in a Bermuda Company
We are
Incorporated in Bermuda, and a Significant Portion of Our Assets
are Located Outside the United States. As a Result, it may not
be Possible for Shareholders to Enforce Civil Liability
Provisions of the United States Federal or State Securities
Laws.
We are incorporated under the laws of Bermuda, and a significant
portion of our assets are located outside the United States. It
may not be possible to enforce court judgments obtained in the
United States against us in Bermuda, or in countries other than
the United States where we have assets, based on the civil
liability provisions of the federal or state securities laws of
the United States. In addition, there is some doubt as to
whether the courts of Bermuda and other countries would
recognize or enforce judgments of United States courts obtained
against us or our directors or officers based on the civil
liabilities provisions of the federal or state securities laws
of the United States or would hear actions against us or those
persons based on those laws. We have been advised by our legal
advisors in Bermuda that the United States and Bermuda do not
currently have a treaty providing for the reciprocal recognition
and enforcement of judgments in civil and commercial matters.
Therefore, a final judgment for the payment of money rendered by
any federal or state court in the United States based on civil
liability, whether or not based solely on United States federal
or state
36
securities laws, would not automatically be enforceable in
Bermuda. Similarly, those judgments may not be enforceable in
countries other than the United States where we have assets.
We May Issue
Preference Shares and Our Bye-Laws and Bermuda Law May
Discourage Takeovers, Which Could Affect the Rights of Holders
of Our Common Shares.
Following this offering, our board of directors will have the
authority to issue up to preference
shares without any further vote or action by the shareholders,
in accordance with the provisions of our bye-laws. Since the
preference shares could be issued with liquidation, dividend and
other rights superior to those of the common shares, the rights
of the holders of common shares will be subject to, and may be
adversely affected by, the rights of the holders of any such
preference shares. The issuance of preference shares could have
the effect of making it more difficult for a third party to
acquire a majority of our outstanding voting shares. Further,
the provisions of our bye-laws, including a staggered board of
directors and the provision related to the ability of
shareholders to remove directors only for cause, and of Bermuda
law could have the effect of delaying or preventing a change in
control of us. See “Description of Share Capital”.
Bermuda Law
Differs from the Laws in Effect in the United States and May
Afford Less Protection to Shareholders.
Our shareholders may have more difficulty protecting their
interests than would shareholders of a corporation incorporated
in a jurisdiction of the United States. As a Bermuda company, we
are governed by the Companies Act, 1981 (as amended), or the
“Companies Act”. The Companies Act differs in some
material respects from laws generally applicable to United
States corporations and shareholders, including the provisions
relating to interested directors, mergers, amalgamations and
acquisitions, takeovers, shareholder lawsuits and
indemnification of directors. See “Description of Share
Capital — Delaware Law”, “Certain
Relationships and Related Party Transactions —
Director and Officer Indemnification” and “Description
of Share Capital — Indemnification of Directors and
Officers”.
Under Bermuda law, the duties of directors and officers of a
company are generally owed to the company only. Shareholders of
Bermuda companies generally do not have rights to take action
against directors or officers of the company, and may only do so
in limited circumstances.
In addition, our bye-laws contain a broad waiver by our
shareholders of any claim or right of action, both individually
and on its behalf, against any of its officers or directors. The
waiver applies to any action taken by an officer or director, or
the failure of an officer or director to take any action, in the
performance of his or her duties, except with respect to any
matter involving any fraud or dishonesty on the part of the
officer or director. This waiver limits the right of
shareholders to assert claims against our officers and directors
unless the act or failure to act involves fraud or dishonesty.
We May Become
Subject to Bermuda Taxes After March 28, 2016, Which May
have a Material Adverse Effect on Our Financial Condition and
Operating Results and Your Investment Could be Adversely
Affected.
The Bermuda Minister of Finance, under the Exempted Undertakings
Tax Protection Act 1966 of Bermuda, has given us an assurance
that if any legislation is enacted in Bermuda that would impose
tax computed on profits or income, or computed on any capital
asset, gain or appreciation, or any tax in the nature of estate
duty or inheritance tax, then the imposition of any such tax
will not be applicable to us or any of our operations, shares,
debentures or other obligations, except insofar as such tax
applies to persons ordinarily resident in Bermuda or to any
taxes payable by us in respect of real property leased by us in
Bermuda. See “Material Bermuda and U.S. Federal Income
Tax Considerations”. This assurance by the Bermuda Minister
of Finance expires on March 28, 2016. Given the limited
duration of the Minister of Finance’s assurance, we cannot
be certain that we will not be subject to any Bermuda tax after
March 28, 2016. If the Bermuda Government imposed
significant taxes on our business, our earnings could decline
significantly.
37
If you Acquire
More Than 10% of Our Outstanding Shares, CFC Rules May Apply to
you.
Rhône Capital has informed us that it intends to take the
position that Arizona Chemical Ltd. and certain of its foreign
subsidiaries are controlled foreign corporations
(“CFCs”) for United States federal income tax
purposes. Each “United States shareholder” of a CFC
that directly or indirectly owns shares in the CFC on the last
day of the CFC’s taxable year must generally include in its
gross income for United States federal income tax purposes its
pro rata share of the CFC’s “subpart F
income”, even if the subpart F income is not distributed.
For these purposes, any U.S. person who owns, directly,
indirectly through foreign persons, or constructively (under
applicable constructive ownership rules of the Code) 10% or more
of the total combined voting power of all classes of shares of a
foreign corporation will be considered to be a “United
States shareholder” of the corporation.
From Time to
Time, Certain Taxing Authorities and International
Organizations, Including the U.S. Government and the
Organization for Economic Cooperation and Development, or the
OECD, have Considered Taking Steps that Would Result in an
Increase in Our Taxes, and They May Take Such Steps in the
Future.
A number of taxing authorities have from time to time considered
imposing measures that would increase the amount of taxes we
would be required to pay. Such authorities could implement
changes in the applicable tax laws and regulations in order to
subject Bermuda companies like us to additional taxes. In
addition, the OECD has published reports and launched a global
dialogue among member and non-member countries as to whether to
impose measures to counteract the effects of tax havens and
preferential tax regimes in countries around the world. While
the OECD regards Bermuda as a jurisdiction that has
substantially complied with internationally agreed tax
standards, and as such has listed Bermuda on its “white
list”, we are not able to predict whether any changes will
be made to this classification or the consequences of any such
change.
We are not able to predict what developments with respect to
applicable tax laws may occur or, if they do occur, what impact
they may have on us. U.S. and international taxing
authorities could subject us to additional taxation at any time,
which would reduce our income and could adversely affect our
profitability and the market value of our shares.
38
CAUTIONARY
NOTE REGARDING FORWARD-LOOKING STATEMENTS
Some of the statements made under the headings
“Summary”, “Business”,
“Management’s Discussion and Analysis of Financial
Condition and Results of Operations”, our audited
consolidated financial statements and related notes thereto and
elsewhere in this prospectus contain forward-looking statements
that reflect our plans, beliefs, expectations and current views
with respect to, among other things, future events and financial
performance. Forward-looking statements are often characterized
by the use of words such as “believes”,
“estimates”, “expects”,
“projects”, “may”, “intends”,
“plans” or “anticipates”, or by discussions
of strategy, plans or intentions. Such forward-looking
statements involve known and unknown risks, uncertainties and
other important factors that could cause the actual results,
performance or our achievements, or industry results, to differ
materially from historical results, any future results, or
performance or achievements expressed or implied by such
forward-looking statements. Such risks and uncertainties
include, but are not limited to:
|
|
|
| |
•
|
conditions in the global economy and capital markets;
|
| |
| |
•
|
our dependence on International Paper and other suppliers to
perform their obligations to us;
|
| |
| |
•
|
failure of our suppliers to perform their obligations under
long-term supply agreements, or our inability to replace or
renew these agreements when they expire, could increase our cost
for these materials and interrupt production;
|
| |
| |
•
|
limited availability or increases in prices of raw materials
used in our business;
|
| |
| |
•
|
our substantial level of indebtedness and the operating and
financial restrictions imposed by our debt instruments and
related indentures;
|
| |
| |
•
|
competitive pressures in the specialty chemicals industry;
|
| |
| |
•
|
our ability to continue technological innovation and successful
commercial introduction of new products;
|
| |
| |
•
|
our ability to protect intellectual property and other
proprietary information;
|
| |
| |
•
|
losses due to liabilities arising out of intellectual property
infringement and product liability claims;
|
| |
| |
•
|
losses due to lawsuits arising out of environmental damage or
personal injuries associated with chemical manufacturing;
|
| |
| |
•
|
our ability to comply with extensive environmental, health and
safety laws, including regulation of exposure to chemicals used
in our operations, including formaldehyde, nonylphenol and
bisphenol A, that could require material expenditures or changes
in our operations;
|
| |
| |
•
|
the risk of accidents that could disrupt our operations or
expose us to significant losses or liabilities;
|
| |
| |
•
|
governmental regulations and trade restrictions;
|
| |
| |
•
|
exposure to interest rate and currency fluctuations;
|
| |
| |
•
|
acts of war or terrorism in the United States or worldwide,
political or financial instability in the countries where our
goods are manufactured and sold; and
|
| |
| |
•
|
other risks and uncertainties described in this prospectus.
|
39
These statements are based upon current plans, estimates and
projections, and therefore you should not place undue reliance
on them. Forward-looking statements speak only as of the date
they are made and we undertake no obligation to update publicly
any of them in light of new information or future events.
You should carefully consider the “Risk Factors” and
subsequent public statements, or reports filed with or furnished
to the SEC, before making any investment decision with respect
to our securities. If any of these trends, risks or
uncertainties actually occurs or continues, our business,
financial condition or operating results could be materially
adversely affected, the trading prices of our securities could
decline and you could lose all or part of your investment. All
forward-looking statements attributable to us or persons acting
on our behalf are expressly qualified in their entirety by this
cautionary statement.
40
USE OF
PROCEEDS
We estimate that the net proceeds to us from our sale
of
common shares in this offering will be
$ million, after deducting
underwriting discounts and commissions and estimated expenses
payable by us in connection with this offering. This assumes an
initial public offering price of $
per share, which is the midpoint of the price range set forth on
the cover page of this prospectus. We intend to use the net
proceeds of this offering to make an approximately
$12 million distribution to AZ Chem Investments
Partners LP, our parent entity, which it will use to redeem
certain preferred equity interests the Rhône Funds received
as consideration for their contribution of $9.5 million of
our second lien indebtedness to us in the second half of 2008
(the approximately $12 million payment representing the
equivalent of principal and accrued interest on such
contribution) and to pay transaction-related expenses of
$ million. We will also use
the net proceeds to pay Rhône Group L.L.C. a fee of
€5.0 million in connection with the amendment of a
management agreement under which Rhône Group L.L.C.
provides us with certain services. See “Related Party
Transactions — Purchase and Contribution of
Indebtedness” and “Related Party
Transactions — Management Agreement”. We intend
to use the remaining net proceeds to repay
$ million of borrowings under
our First Lien Credit Agreement, representing 75% of the
remaining net proceeds, and to repay
$ million of borrowings under
our Second Lien Credit Agreement, representing 25% of the
remaining net proceeds.
A $1.00 increase (decrease) in the assumed initial public
offering price of $ per share
would increase (decrease) the net proceeds to us from this
offering by $ million,
assuming the number of shares offered by us, as set forth on the
cover of this prospectus, remains the same and after deducting
underwriting discounts and commissions and estimated expenses
payable by us.
As of March 31, 2010, our total indebtedness outstanding
was $331.6 million, including $327.4 million of
borrowings outstanding under our credit agreements. The
principal amounts of our term loans under our First Lien Credit
Agreement amortize in quarterly installments of
1/4
of 1% of the outstanding principal amount and the remaining
outstanding principal amount matures on February 28, 2013.
The term loans under our Second Lien Credit Agreement mature on
February 28, 2014. Borrowings under our credit agreements
bear interest at variable rates. As of March 31, 2010, the
weighted average interest rate under our First Lien Credit
Agreement was 2.52% and the weighted average interest rate under
our Second Lien Credit Agreement was 5.75%, in each case before
giving effect to our interest rate swaps. See “Description
of Our Indebtedness”.
We will not receive any proceeds from the sale of shares by the
selling shareholder.
41
DIVIDEND
POLICY
Other than the distribution we are making to AZ Chem Investments
Partners LP out of the proceeds of this offering, we do not
anticipate paying any cash dividends on our common shares for
the foreseeable future. We intend to retain all available funds
and any future earnings to reduce debt and fund the development
and growth of our business.
Any future determination to pay dividends will be at the
discretion of our board of directors and will take into account:
|
|
|
| |
•
|
restrictions in our credit agreements;
|
| |
| |
•
|
general economic and business conditions;
|
| |
| |
•
|
our financial condition and results of operations;
|
| |
| |
•
|
our capital requirements and the capital requirements of our
subsidiaries;
|
| |
| |
•
|
the ability of our operating subsidiaries to pay dividends and
make distributions to us; and
|
| |
| |
•
|
such other factors as our board of directors may deem relevant.
|
42
CAPITALIZATION
The following table sets forth (1) the capitalization of
Arizona Chem Sweden Holdings AB as of March 31, 2010 on an
actual basis and (2) the capitalization of Arizona Chemical
Ltd. as of March 31, 2010 on an as adjusted basis after
giving effect to the Reorganization and to reflect the receipt
and use by us of the estimated net proceeds from the sale of
common shares in this offering as described under the heading
“Use of Proceeds”.
This table should be read in conjunction with “Use of
Proceeds”, “Selected Historical Consolidated Financial
Data”, “Management’s Discussion and Analysis of
Financial Condition and Results of Operations” and our
consolidated financial statements and the related notes thereto
included elsewhere in this prospectus.
| |
|
|
|
|
|
|
|
|
|
|
|
As of March 31, 2010
|
|
|
|
|
Actual
|
|
|
As Adjusted(1)
|
|
|
|
|
(dollars in millions, except share data)
|
|
|
|
|
Debt:
|
|
|
|
|
|
|
|
|
|
Senior credit agreements:
|
|
|
|
|
|
|
|
|
|
First Lien Term Loan
|
|
$
|
211.9
|
|
|
$
|
|
|
|
Second Lien Term Loan
|
|
|
115.5
|
|
|
|
|
|
|
Revolving credit facility
|
|
|
—
|
|
|
|
—
|
|
|
Capital lease obligation
|
|
|
3.7
|
|
|
|
3.7
|
|
|
Loans related to Abieta
|
|
|
0.5
|
|
|
|
0.5
|
|
|
|
|
|
|
|
|
|
|
|
|
Total debt
|
|
|
331.6
|
|
|
|
|
|
|
Shareholder’s equity:
|
|
|
|
|
|
|
|
|
|
Common shares — Arizona Chem Sweden Holdings AB,
$14.25 par value per share; 1,000 shares issued and
outstanding, actual
|
|
|
0.0
|
|
|
|
—
|
|
|
Paid-in capital — Arizona Chem Sweden Holdings AB
|
|
|
135.7
|
|
|
|
—
|
|
|
Common shares — Arizona Chemical Ltd., $0.01 par value
per share; no shares authorized or issued and outstanding,
actual
( shares
authorized; shares
issued and outstanding, as adjusted)
|
|
|
—
|
|
|
|
|
|
|
Paid-in capital — Arizona Chemical Ltd.
|
|
|
—
|
|
|
|
|
|
|
Accumulated deficit
|
|
|
(31.8
|
)
|
|
|
|
|
|
Accumulated other comprehensive income
|
|
|
(2.4
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total shareholder’s equity
|
|
|
101.5
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total capitalization
|
|
$
|
433.1
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1) |
|
A $1.00 increase or decrease in the assumed initial public
offering price per share would result in an approximately
$ million increase or
decrease, as applicable, in as adjusted total debt, as adjusted
additional paid-in capital, as adjusted total shareholder’s
equity and as adjusted total capitalization, assuming the number
of shares offered by us, as set forth on the front cover of this
prospectus, remains the same and after deducting the estimated
underwriting discounts and commissions and estimated offering
expenses payable by us. |
43
DILUTION
If you invest in our common shares, your interest will be
diluted to the extent of the difference between the initial
public offering price per share and the as adjusted net tangible
book value per common share after this offering. Our historical
net tangible book value as of March 31, 2010 was
$(7.8) million, or approximately
$ per share
(assuming common shares
outstanding after the Reorganization). Net tangible book value
per share represents the amount of shareholder’s equity
less the net book value of intangible assets, divided by the
number of common shares outstanding at that date.
Net tangible book value dilution per share to new investors
represents the difference between the amount per share paid by
purchasers of common shares in this offering and the as adjusted
net tangible book value per common share immediately after
completion of this offering. After giving effect to (1) the
Reorganization and (2) our sale of common shares in this
offering at an assumed initial public offering price of
$ per share (the midpoint of the
price range set forth on the cover page of this prospectus) and
after deducting the estimated underwriting discounts and
commissions and offering expenses, our as adjusted net tangible
book value as of March 31, 2010 would have been
$ per share. This amount
represents an immediate increase in net tangible book value of
$ per share to existing
shareholders and an immediate dilution in net tangible book
value of $ per share to purchasers
of common shares in this offering, as illustrated in the
following table.
| |
|
|
|
|
|
|
|
|
|
Assumed initial public offering price per share
|
|
|
|
|
|
$
|
|
|
|
Historical net tangible book value per share as of
|
|
|
|
|
|
$
|
|
|
|
Increase per share attributable to new investors
|
|
|
|
|
|
|
|
|
|
As adjusted net tangible book value per share after giving
effect to this offering
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Dilution per share to new investors
|
|
|
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
A $1.00 increase or decrease in the assumed initial public
offering price per share would increase or decrease, as
applicable, the net proceeds to us from this offering by
approximately $ million,
dilution per share to new investors by approximately
$ and net tangible book value
attributable to this offering by
$ per share, after deducting the
estimated underwriting discounts and commissions and estimated
offering expenses payable by us.
The following table summarizes, as of March 31, 2010, on
the as adjusted basis described above, the differences between
existing shareholders and new investors with respect to the
number of common shares purchased from us, the total
consideration paid and the average price per common share paid
by existing shareholders, after giving effect to the issuance of
our common shares in this offering at an assumed initial public
offering price of $ per share (the
midpoint of the range on the cover page of this prospectus),
before deducting the estimated underwriting discounts and
commissions and offering expenses.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
Average
|
|
|
|
|
Shares Purchased
|
|
|
Consideration
|
|
|
Price
|
|
|
|
|
Number
|
|
|
Percent
|
|
|
Amount
|
|
|
Percent
|
|
|
per Share
|
|
|
|
|
Existing shareholders
|
|
|
|
|
|
|
|
%
|
|
$
|
|
|
|
|
|
%
|
|
$
|
|
|
|
New investors
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
|
|
|
|
100
|
%
|
|
$
|
|
|
|
$
|
100
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
44
If the underwriters exercise their option to purchase additional
shares in full, the number of common shares held by existing
shareholders will decrease
to ,
or percent,
of the total number of our common shares outstanding after this
offering, and the number of common shares held by new investors
will increase
to ,
or
percent, of the total number of our common shares outstanding
after this offering.
A $1.00 increase or decrease in the assumed initial public
offering price per share would increase or decrease, as
applicable, total consideration paid by new investors to us in
this offering by $ million
and the average price per share paid by new investors by $1.00,
assuming the number of shares of common stock offered by us, set
forth on the cover of this prospectus, remains the same.
45
SELECTED
CONSOLIDATED FINANCIAL DATA
The following tables present selected historical consolidated
statements of operations, balance sheet and other data for the
periods presented and should only be read in conjunction with
our audited consolidated financial statements and the related
notes thereto, and “Management’s Discussion and
Analysis of Financial Condition and Results of Operations”,
which are included elsewhere in this prospectus.
We were acquired on February 28, 2007 by Rhône Capital
in a business combination accounted for under the purchase
method of accounting. The historical selected consolidated
financial data as of December 31, 2009, 2008 and 2007, the
years ended December 31, 2009 and 2008, and for the period
March 1, 2007 to December 31, 2007 have been derived
from our successor audited historical consolidated financial
statements included elsewhere in this prospectus. The selected
historical consolidated financial and operating data for the
period January 1, 2007 to February 28, 2007, which is
included elsewhere in this prospectus, and the selected
historical consolidated financial and operating data as of and
for the years ended December 31, 2006 and 2005, which is
not included in this prospectus, have been derived from the
audited historical consolidated financial statements of our
predecessor period. The selected consolidated financial data for
the three months ended March 31, 2010 and 2009 and as of March
31, 2010, have been derived from our unaudited condensed
consolidated financial statements, which are included elsewhere
in this prospectus. The unaudited condensed consolidated
financial statements include, in the opinion of management, all
adjustments, consisting only of normal recurring adjustments,
that management considers necessary for the fair presentation of
the unaudited condensed consolidated financial information set
forth in those statements. Results of operations for the interim
periods are not necessarily indicative of the results that might
be expected for any other interim period or for an entire year.
As a result of the Acquisition, our assets and liabilities were
adjusted to their estimated fair values. In addition, our
consolidated statements of operations data for the successor
period include interest expense resulting from indebtedness
incurred to finance the Acquisition and depreciation and
amortization of fair value adjustments to property, plant and
equipment and intangible assets related to the Acquisition.
Therefore, our successor period financial data generally is not
comparable to our predecessor period financial data.
Consolidated
Statements of Operations Data
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Predecessor
|
|
|
Predecessor
|
|
|
Predecessor
|
|
|
|
|
Three
|
|
|
Three
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
months
|
|
|
months
|
|
|
|
|
|
|
|
|
March 1, 2007
|
|
|
January 1,
|
|
|
|
|
|
|
|
|
|
|
ended
|
|
|
ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
2007 through
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
February 28,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
2007
|
|
|
2006
|
|
|
2005
|
|
|
|
|
(dollars in thousands, except per share data)
|
|
|
|
|
Net sales
|
|
$
|
198,051
|
|
|
$
|
177,934
|
|
|
$
|
767,465
|
|
|
$
|
1,001,988
|
|
|
$
|
723,797
|
|
|
$
|
132,070
|
|
|
$
|
767,882
|
|
|
$
|
692,446
|
|
|
Cost of products sold
|
|
|
157,333
|
|
|
|
163,445
|
|
|
|
646,986
|
|
|
|
868,536
|
|
|
|
642,341
|
|
|
|
113,074
|
|
|
|
645,125
|
|
|
|
600,902
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gross profit
|
|
|
40,718
|
|
|
|
14,489
|
|
|
|
120,479
|
|
|
|
133,452
|
|
|
|
81,456
|
|
|
|
18,996
|
|
|
|
122,757
|
|
|
|
91,544
|
|
|
Selling, general and administrative
|
|
|
25,673
|
|
|
|
15,464
|
|
|
|
78,200
|
|
|
|
91,936
|
|
|
|
86,684
|
|
|
|
12,899
|
|
|
|
74,240
|
|
|
|
72,838
|
|
|
Unrealized foreign currency exchange (gains) losses(1)
|
|
|
(4,836
|
)
|
|
|
367
|
|
|
|
(9,347
|
)
|
|
|
20,304
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Restructuring and impairment(2)
|
|
|
2,047
|
|
|
|
1,035
|
|
|
|
26,395
|
|
|
|
15,513
|
|
|
|
114
|
|
|
|
—
|
|
|
|
1,851
|
|
|
|
5,558
|
|
|
Other operating income(3)
|
|
|
—
|
|
|
|
(2,043
|
)
|
|
|
(5,537
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating income (loss)
|
|
|
17,834
|
|
|
|
(334
|
)
|
|
|
30,768
|
|
|
|
5,699
|
|
|
|
(5,342
|
)
|
|
|
6,097
|
|
|
|
46,666
|
|
|
|
13,148
|
|
|
Interest (expense) income, net
|
|
|
(3,870
|
)
|
|
|
(4,560
|
)
|
|
|
(16,546
|
)
|
|
|
(29,523
|
)
|
|
|
(28,775
|
)
|
|
|
118
|
|
|
|
461
|
|
|
|
298
|
|
|
Loss on interest rate swaps, net
|
|
|
(709
|
)
|
|
|
(600
|
)
|
|
|
(2,541
|
)
|
|
|
(9,311
|
)
|
|
|
(2,275
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other income(4)
|
|
|
623
|
|
|
|
2,151
|
|
|
|
3,635
|
|
|
|
1,879
|
|
|
|
—
|
|
|
|
—
|
|
|
|
2,081
|
|
|
|
(177
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income (loss) before income tax expense (benefit)
|
|
|
13,878
|
|
|
|
(3,343
|
)
|
|
|
15,316
|
|
|
|
(31,256
|
)
|
|
|
(36,392
|
)
|
|
|
6,215
|
|
|
|
49,208
|
|
|
|
13,269
|
|
|
Income tax expense (benefit)
|
|
|
3,885
|
|
|
|
(836
|
)
|
|
|
3,831
|
|
|
|
(4,277
|
)
|
|
|
(8,913
|
)
|
|
|
2,614
|
|
|
|
18,161
|
|
|
|
9,090
|
|
|
Equity in earnings of affiliate(5)
|
|
|
4
|
|
|
|
150
|
|
|
|
613
|
|
|
|
380
|
|
|
|
189
|
|
|
|
84
|
|
|
|
2,033
|
|
|
|
(187
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
46
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Predecessor
|
|
|
Predecessor
|
|
|
Predecessor
|
|
|
|
|
Three
|
|
|
Three
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
months
|
|
|
months
|
|
|
|
|
|
|
|
|
March 1, 2007
|
|
|
January 1,
|
|
|
|
|
|
|
|
|
|
|
ended
|
|
|
ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
2007 through
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
February 28,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
2007
|
|
|
2006
|
|
|
2005
|
|
|
|
|
(dollars in thousands, except per share data)
|
|
|
|
|
Net income (loss)
|
|
$
|
9,997
|
|
|
$
|
(2,357
|
)
|
|
$
|
12,098
|
|
|
$
|
(26,599
|
)
|
|
$
|
(27,290
|
)
|
|
$
|
3,685
|
|
|
$
|
33,080
|
|
|
$
|
3,992
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Earnings per share(6):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic and diluted
|
|
$
|
9,997
|
|
|
$
|
(2,357
|
)
|
|
$
|
12,098
|
|
|
$
|
(26,599
|
)
|
|
$
|
(27,290
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average common shares outstanding:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic and diluted
|
|
|
1,000
|
|
|
|
1,000
|
|
|
|
1,000
|
|
|
|
1,000
|
|
|
|
1,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Consolidated Cash
Flow Data
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
Successor
|
|
Successor
|
|
Successor
|
|
Successor
|
|
Predecessor
|
|
Predecessor
|
|
Predecessor
|
|
|
|
Three
|
|
Three
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Months
|
|
Months
|
|
Year
|
|
Year
|
|
March 1
|
|
January 1
|
|
Year
|
|
Year
|
|
|
|
Ended
|
|
Ended
|
|
Ended
|
|
Ended
|
|
through
|
|
through
|
|
Ended
|
|
Ended
|
|
|
|
March 31,
|
|
March 31,
|
|
December 31,
|
|
December 31,
|
|
December 31,
|
|
February 28,
|
|
December 31,
|
|
December 31,
|
|
|
|
2010
|
|
2009
|
|
2009
|
|
2008
|
|
2007
|
|
2007
|
|
2006
|
|
2005
|
|
|
|
(dollars in thousands)
|
|
|
|
Net cash provided by (used in) operating
activities
|
|
$
|
2,507
|
|
|
$
|
11,100
|
|
|
$
|
117,325
|
|
|
$
|
20,841
|
|
|
$
|
45,022
|
|
|
$
|
(12,088
|
)
|
|
$
|
5,048
|
|
|
$
|
44,115
|
|
|
Net cash used in investing activities
|
|
|
(7,931
|
)
|
|
|
(18,027
|
)
|
|
|
(44,294
|
)
|
|
|
(34,293
|
)
|
|
|
(497,331
|
)
|
|
|
(4,598
|
)
|
|
|
(38,131
|
)
|
|
|
(29,228
|
)
|
|
Net cash provided by
(used in) financing activities
|
|
|
2,202
|
|
|
|
(1,220
|
)
|
|
|
(63,771
|
)
|
|
|
12,768
|
|
|
|
488,547
|
|
|
|
10,296
|
|
|
|
28,375
|
|
|
|
(10,875
|
)
|
Consolidated
Balance Sheet Data
| |
|
|
|
|
|
|
|
|
|
|
|
As of March 31, 2010
|
|
|
|
|
|
As Adjusted for
|
|
|
|
Actual
|
|
the Offering(7)
|
|
|
|
(dollars in thousands)
|
|
|
|
Cash and cash equivalents
|
|
$
|
43,057
|
|
|
$
|
|
|
|
Total assets
|
|
|
630,275
|
|
|
|
|
|
|
Total debt
|
|
|
331,645
|
|
|
|
|
|
|
Total shareholder’s equity
|
|
|
101,495
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
Successor
|
|
Successor
|
|
Successor
|
|
Predecessor
|
|
Predecessor
|
|
|
|
March 31,
|
|
December 31,
|
|
December 31,
|
|
December 31,
|
|
December 31,
|
|
February 28,
|
|
|
|
2010
|
|
2009
|
|
2008
|
|
2007
|
|
2006
|
|
2005
|
|
|
|
(dollars in thousands)
|
|
|
|
Total assets
|
|
$
|
630,275
|
|
|
$
|
618,009
|
|
|
$
|
691,084
|
|
|
$
|
708,322
|
|
|
$
|
553,390
|
|
|
$
|
449,672
|
|
|
Total debt
|
|
|
331,645
|
|
|
|
338,853
|
|
|
|
387,665
|
|
|
|
388,885
|
|
|
|
4,231
|
|
|
|
4,010
|
|
|
Total shareholder’s equity(8)
|
|
|
101,495
|
|
|
|
97,607
|
|
|
|
95,509
|
|
|
|
104,045
|
|
|
|
382,854
|
|
|
|
297,089
|
|
47
Other
Data
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
Successor
|
|
Successor
|
|
Successor
|
|
Successor
|
|
Predecessor
|
|
Predecessor
|
|
Predecessor
|
|
|
|
Three
|
|
Three
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Months
|
|
Months
|
|
Year
|
|
Year
|
|
March 1
|
|
January 1
|
|
Year
|
|
Year
|
|
|
|
Ended
|
|
Ended
|
|
Ended
|
|
Ended
|
|
through
|
|
through
|
|
Ended
|
|
Ended
|
|
|
|
March 31,
|
|
March 31,
|
|
December 31,
|
|
December 31,
|
|
December 31,
|
|
February 28,
|
|
December 31,
|
|
December 31,
|
|
|
|
2010
|
|
2009
|
|
2009
|
|
2008
|
|
2007
|
|
2007
|
|
2006
|
|
2005
|
|
|
|
(dollars in thousands)
|
|
|
|
Gross margin(9)
|
|
|
20.6
|
%
|
|
|
8.1
|
%
|
|
|
15.7
|
%
|
|
|
13.3
|
%
|
|
|
11.3
|
%
|
|
|
14.4
|
%
|
|
|
16.0
|
%
|
|
|
13.2
|
%
|
|
Depreciation(9)
|
|
$
|
6,599
|
|
|
$
|
6,872
|
|
|
$
|
29,781
|
|
|
$
|
23,311
|
|
|
$
|
19,693
|
|
|
$
|
4,097
|
|
|
$
|
22,414
|
|
|
$
|
29,178
|
|
|
Amortization
|
|
|
2,444
|
|
|
|
2,565
|
|
|
|
10,834
|
|
|
|
10,899
|
|
|
|
9,120
|
|
|
|
325
|
|
|
|
1,353
|
|
|
|
156
|
|
|
Adjusted EBITDA(10)
|
|
|
27,378
|
|
|
|
11,043
|
|
|
|
93,859
|
|
|
|
92,723
|
|
|
|
54,874
|
|
|
|
12,024
|
|
|
|
78,347
|
|
|
|
62,419
|
|
|
Free cash flow(11)
|
|
|
(5,424
|
)
|
|
|
1,845
|
|
|
|
81,803
|
|
|
|
(13,452
|
)
|
|
|
25,132
|
|
|
|
(16,686
|
)
|
|
|
(33,083
|
)
|
|
|
14,887
|
|
|
|
|
|
(1) |
|
Unrealized foreign currency exchange losses (gains) is primarily
related to translation changes in the balance of the
Euro-denominated debt under our First Lien Credit Agreement.
This is further described in Note 11 of our consolidated
financial statements and Note 8 of our unaudited condensed
consolidated financial statements appearing at the end of this
prospectus and “Management’s Discussion and Analysis
of Financial Condition and Results of Operations”. |
The following table presents unrealized and realized foreign
exchange losses (gains) included in our consolidated statement
of operations.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
Successor
|
|
Successor
|
|
Successor
|
|
Successor
|
|
Predecessor
|
|
Predecessor
|
|
Predecessor
|
|
|
|
Three
|
|
Three
|
|
|
|
|
|
|
|
January 1,
|
|
|
|
|
|
|
|
months
|
|
months
|
|
|
|
|
|
March 1, 2007
|
|
2007
|
|
|
|
|
|
|
|
ended
|
|
ended
|
|
Year Ended
|
|
Year Ended
|
|
through
|
|
through
|
|
Year Ended
|
|
Year Ended
|
|
|
|
March 31,
|
|
March 31,
|
|
December 31,
|
|
December 31,
|
|
December 31,
|
|
February 28,
|
|
December 31,
|
|
December 31,
|
|
|
|
2010
|
|
2009
|
|
2009
|
|
2008
|
|
2007
|
|
2007
|
|
2006
|
|
2005
|
|
|
|
(dollars in thousands)
|
|
|
|
Unrealized translation related foreign currency exchange (gains)
losses
|
|
$
|
(4,836
|
)
|
|
$
|
367
|
|
|
$
|
(9,347
|
)
|
|
$
|
20,304
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
Realized transaction related foreign currency exchange (gains)
losses — selling, general and administrative
|
|
|
(107
|
)
|
|
|
(1,883
|
)
|
|
|
214
|
|
|
|
234
|
|
|
|
238
|
|
|
|
(499
|
)
|
|
|
1,151
|
|
|
|
(192
|
)
|
|
Realized transaction related foreign currency exchange (gains)
losses — cost of goods sold
|
|
|
(205
|
)
|
|
|
37
|
|
|
|
154
|
|
|
|
65
|
|
|
|
3
|
|
|
|
17
|
|
|
|
24
|
|
|
|
2
|
|
|
|
|
|
(2) |
|
Restructuring and impairment includes pre-tax restructuring
charges primarily related to manufacturing facility closures and
employee terminations as part of our cost reduction initiatives.
This is further described in Note 9 of our consolidated
financial statements and Note 7 of our unaudited condensed
consolidated financial statements appearing at the end of this
prospectus and “Management’s Discussion and Analysis
of Financial Condition and Results of Operations”. |
48
|
|
|
|
|
|
The following table presents details of our restructuring and
impairment charges included in our consolidated statement of
operations. |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Predecessor
|
|
|
Predecessor
|
|
|
Predecessor
|
|
|
|
|
Three
|
|
|
Three
|
|
|
|
|
|
|
|
|
March 1,
|
|
|
January 1,
|
|
|
|
|
|
|
|
|
|
|
months
|
|
|
months
|
|
|
|
|
|
|
|
|
2007
|
|
|
2007
|
|
|
|
|
|
|
|
|
|
|
ended
|
|
|
ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
through
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
February 28,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
2007
|
|
|
2006
|
|
|
2005
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Port St. Joe, FL
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
5,822
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
Gersthofen, Germany
|
|
|
108
|
|
|
|
—
|
|
|
|
1,251
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Valdosta, GA
|
|
|
97
|
|
|
|
—
|
|
|
|
874
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
U.K. facilities
|
|
|
—
|
|
|
|
—
|
|
|
|
339
|
|
|
|
5,369
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Niort, France
|
|
|
648
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,972
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Dover, OH
|
|
|
—
|
|
|
|
3
|
|
|
|
—
|
|
|
|
635
|
|
|
|
114
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Pensacola, FL
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
534
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Greaker, Norway
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
4,200
|
|
|
Picayune, MS
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
2,100
|
|
|
Sandarne, Sweden
|
|
|
1,194
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,851
|
|
|
|
(742
|
)
|
|
Impairments
|
|
|
—
|
|
|
|
1,032
|
|
|
|
18,109
|
|
|
|
7,003
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
2,047
|
|
|
$
|
1,035
|
|
|
$
|
26,395
|
|
|
$
|
15,513
|
|
|
$
|
114
|
|
|
$
|
—
|
|
|
$
|
1,851
|
|
|
$
|
5,558
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(3) |
|
Other operating income of $5.5 million recorded in 2009 included
$4.9 million of insurance proceeds related to a fire at our
Oulu, Finland manufacturing facility and a gain of $0.7 million
on the sale of capital assets realized as a result of the
closure of our Bedlington, U.K. manufacturing facility. |
| |
|
(4) |
|
Other income includes a gain on our acquisition of Abieta Chemie
GmbH, which we refer to as “Abieta”, of
$2.1 million and a gain on settlement with International
Paper of $1.3 million in 2009. In 2008 and 2006, other
income included a gain of $1.9 million on the
extinguishment of a portion of our debt and a legal settlement
of $2.1 million, respectively. In the three months ended
March 31, 2010, other income primarily related to insurance
proceeds relating to a fire at our Sandarne, Sweden
manufacturing facility. |
| |
|
(5) |
|
Equity in earnings of affiliate relates to our 10% investment in
Arboris, LLC, which was formed in 2002 and is accounted for
under the equity method of accounting, as we have the ability to
exercise significant influence. Our share of the earnings from
operations in this investment is recorded in our statement of
operations. This is further described in Note 3 of our
consolidated financial statements appearing at the end of this
prospectus and “Management’s Discussion and Analysis
of Financial Condition and Results of Operations”. |
| |
|
(6) |
|
Prior to the Acquisition, we were an operating division of
International Paper. As a result, the capital structure of our
predecessor did not include common stock or other forms of
equity shares. Accordingly, income (loss) attributable to common
shareholders per commons share is not available for periods
prior to March 1, 2007. Additionally, the information
presented regarding earnings per share and weighted average
common shares outstanding is that of Arizona Chem Sweden
Holdings AB on an actual basis. The earnings per share of
Arizona Chemical Ltd. on an as adjusted basis after giving
effect to the Reorganization for the three months ended
March 31, 2010 and 2009, the years ended December 31,
2009 and 2008 and the ten month period ended December 31,
2007 would have been $ ,
$ ,
$ ,
$ , and
$ , respectively,
assuming
common shares outstanding. |
| |
|
(7) |
|
Reflects on an adjusted basis the sale
of of
our common shares in this offering at an assumed initial public
offering price of $ per share (the
mid-point of the price range set forth on the cover page of this
prospectus), and the application of the estimated net proceeds
from this offering as described under “Use of
Proceeds”, and assumes this offering had been consummated
on March 31, 2010. |
49
|
|
|
|
(8) |
|
In the predecessor period, total shareholder’s equity
represents our divisional control account as we were a division
of International Paper. |
| |
|
(9) |
|
Included in these amounts in 2009 is accelerated depreciation
associated with of the closure of our Port St. Joe facility
in the amount of $1.6 million. Accelerated depreciation had
a negative impact on our gross margins of 1.3% in 2009. |
| |
|
(10) |
|
Adjusted EBITDA represents net income before interest, taxes,
depreciation, and amortization and then it is adjusted for
various items as defined in our credit agreements. Adjusted
EBITDA presented in the predecessor period was calculated on the
same basis as Adjusted EBITDA presented in the successor periods
pursuant to our credit agreements. Under the terms of our credit
agreements, we use Adjusted EBITDA to calculate our maximum
leverage ratio and our minimum interest coverage ratio
covenants. See “— Adjusted EBITDA” below. |
| |
|
(11) |
|
Free cash flow represents net cash provided by operating
activities, less purchases of property, plant and equipment,
software spending, proceeds from disposal of property, plant and
equipment, and other items. See “— Free Cash
Flow” below. |
Adjusted
EBITDA
Adjusted EBITDA is not a GAAP concept. We present Adjusted
EBITDA because it is used by management to evaluate operating
performance and because, under our credit agreements, we use it
to calculate our maximum leverage ratio and our minimum interest
coverage ratio covenants. In addition, a reconciliation from net
income to Adjusted EBITDA is required for reporting and covenant
calculation purposes pursuant to our credit agreements. We
consider it an important supplemental measure of our performance
and believe it is frequently used by securities analysts,
investors and other interested parties in the evaluation of
companies in our industry. We also use Adjusted EBITDA for our
executive compensation plan, which bases incentive compensation
payments on our Adjusted EBITDA performance.
Adjusted EBITDA has limitations as an analytical tool, and you
should not consider it in isolation, or as a substitute for
analysis of our results as reported under GAAP. Because of these
limitations, Adjusted EBITDA should not be considered as a
measure of discretionary cash available to us to invest in the
growth of our business. We compensate for these limitations by
relying primarily on our GAAP results and using Adjusted EBITDA
only as supplemental information. A reconciliation of net income
(loss) to Adjusted EBITDA is presented below:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Predecessor
|
|
|
Predecessor
|
|
|
Predecessor
|
|
|
|
|
Three
|
|
|
Three
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
months
|
|
|
months
|
|
|
|
|
|
|
|
|
March 1, 2007
|
|
|
January 1,
|
|
|
|
|
|
|
|
|
|
|
ended
|
|
|
ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
2007 through
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
February 28,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
2007(4)
|
|
|
2006(4)
|
|
|
2005(4)
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Net income (loss)
|
|
$
|
9,997
|
|
|
$
|
(2,357
|
)
|
|
$
|
12,098
|
|
|
$
|
(26,599
|
)
|
|
$
|
(27,290
|
)
|
|
$
|
3,685
|
|
|
$
|
33,080
|
|
|
$
|
3,992
|
|
|
Interest expense (income), net
|
|
|
3,870
|
|
|
|
4,560
|
|
|
|
16,546
|
|
|
|
29,523
|
|
|
|
28,775
|
|
|
|
(118
|
)
|
|
|
(461
|
)
|
|
|
(298
|
)
|
|
Income tax expense (benefit)
|
|
|
3,885
|
|
|
|
(836
|
)
|
|
|
3,831
|
|
|
|
(4,277
|
)
|
|
|
(8,913
|
)
|
|
|
2,614
|
|
|
|
18,161
|
|
|
|
9,090
|
|
|
Depreciation and amortization
|
|
|
9,043
|
|
|
|
9,437
|
|
|
|
40,615
|
|
|
|
34,210
|
|
|
|
28,813
|
|
|
|
4,422
|
|
|
|
23,767
|
|
|
|
29,335
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
EBITDA
|
|
|
26,795
|
|
|
|
10,804
|
|
|
|
73,090
|
|
|
|
32,857
|
|
|
|
21,385
|
|
|
|
10,603
|
|
|
|
74,547
|
|
|
|
42,119
|
|
50
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Predecessor
|
|
|
Predecessor
|
|
|
Predecessor
|
|
|
|
|
Three
|
|
|
Three
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
months
|
|
|
months
|
|
|
|
|
|
|
|
|
March 1, 2007
|
|
|
January 1,
|
|
|
|
|
|
|
|
|
|
|
ended
|
|
|
ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
2007 through
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
February 28,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
2007(4)
|
|
|
2006(4)
|
|
|
2005(4)
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Unrealized foreign currency exchange (gains) losses
|
|
|
(4,836
|
)
|
|
|
367
|
|
|
|
(9,347
|
)
|
|
|
20,304
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Restructuring and impairment
|
|
|
2,047
|
|
|
|
1,035
|
|
|
|
26,395
|
|
|
|
15,513
|
|
|
|
114
|
|
|
|
—
|
|
|
|
1,851
|
|
|
|
5,558
|
|
|
Loss on interest rate swaps, net
|
|
|
709
|
|
|
|
600
|
|
|
|
2,541
|
|
|
|
9,311
|
|
|
|
2,275
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Equity in earnings of affiliate
|
|
|
(4
|
)
|
|
|
(150
|
)
|
|
|
(613
|
)
|
|
|
(380
|
)
|
|
|
(189
|
)
|
|
|
(84
|
)
|
|
|
(2,033
|
)
|
|
|
187
|
|
|
Transaction costs(1)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,316
|
|
|
|
10,271
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Management fees(2)
|
|
|
495
|
|
|
|
351
|
|
|
|
1,538
|
|
|
|
1,990
|
|
|
|
1,295
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Transition costs(3)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
2,984
|
|
|
|
6,575
|
|
|
|
92
|
|
|
|
—
|
|
|
|
—
|
|
|
Gain on Abieta acquisition
|
|
|
—
|
|
|
|
(2,151
|
)
|
|
|
(2,151
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Gain on settlement with International Paper
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,316
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Selling, general and administrative severance
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
3,121
|
|
|
|
2,235
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Third-party advisor fees
|
|
|
2,135
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,573
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Consulting services
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
3,794
|
|
|
|
7,616
|
|
|
|
—
|
|
|
|
2,600
|
|
|
|
—
|
|
|
Gain on debt extinguishment
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,901
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Divestiture incentive program
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
5,200
|
|
|
|
—
|
|
|
Environmental remediation
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
2,100
|
|
|
|
—
|
|
|
U.K. land sale
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(4,900
|
)
|
|
|
—
|
|
|
Arboris loan forgiveness
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(2,000
|
)
|
|
|
—
|
|
|
Workers’ compensation
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
5,100
|
|
|
Hurricane impact
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
3,400
|
|
|
Oulu, Finland strike
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
3,100
|
|
|
Other items
|
|
|
37
|
|
|
|
187
|
|
|
|
3,722
|
|
|
|
2,241
|
|
|
|
3,297
|
|
|
|
1,413
|
|
|
|
982
|
|
|
|
2,955
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Adjusted EBITDA
|
|
$
|
27,378
|
|
|
$
|
11,043
|
|
|
$
|
93,859
|
|
|
$
|
92,723
|
|
|
$
|
54,874
|
|
|
$
|
12,024
|
|
|
$
|
78,347
|
|
|
$
|
62,419
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1) |
|
Transaction costs were related to the Acquisition. These costs
included legal, tax, IT, professional fees, and Rhône
Capital transaction fees. |
| |
|
(2) |
|
Management fees are paid to Rhône Group L.L.C., an
affiliate of Rhône Capital for corporate-level support
activities provided to us. We currently expect to terminate the
management agreement with Rhône Group L.L.C. in connection
with this offering. |
| |
|
(3) |
|
Transition costs included fees paid to International Paper under
the transition services agreement (governing interim services
provided by International Paper), IT consulting fees, costs for
infrastructure build-out (such as IT and phone systems, treasury
function setup, HR/payroll) and the cost of a carve-out audit,
among other items. |
| |
|
(4) |
|
Adjusted EBITDA presented in the predecessor period was
calculated on the same basis as Adjusted EBITDA presented in the
successor periods pursuant to our credit agreements. |
Free Cash
Flow
Free cash flow is not a GAAP concept. We present free cash flow
because our management considers it to be a useful, supplemental
indicator of our liquidity. When measured over time, free cash
flow provides supplemental information to investors concerning
our operating results and our ability to generate cash flows to
satisfy mandatory debt service requirements and make other
non-discretionary expenditures.
Our management believes that consideration of free cash flow
should only be supplemental because free cash flow has
limitations as an analytical financial measure. Our management
compensates for these limitations by relying primarily on our
results under GAAP to evaluate our liquidity and by considering
independently the economic effects of the foregoing items that
are not reflected in free cash flow. As a result of these
limitations, free cash flow should not be considered as a
substitute for other measures of liquidity reported in
accordance with GAAP, including net cash provided by (used
51
in) operating activities, net cash provided by (used in)
investing activities, net cash provided by (used in) financing
activities or change in cash and cash equivalents. A
reconciliation of net cash provided by operating activities to
free cash flow is presented below:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Predecessor
|
|
|
Predecessor
|
|
|
Predecessor
|
|
|
|
|
Three Months
|
|
|
Three Months
|
|
|
|
|
|
|
|
|
March 1,
|
|
|
January 1,
|
|
|
|
|
|
|
|
|
|
|
Ended
|
|
|
Ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
2007 through
|
|
|
2007 through
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
February 28,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
2007
|
|
|
2006
|
|
|
2005
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Net cash provided by (used in) operating activities
|
|
$
|
2,507
|
|
|
$
|
11,100
|
|
|
$
|
117,325
|
|
|
$
|
20,841
|
|
|
$
|
45,022
|
|
|
$
|
(12,088
|
)
|
|
$
|
5,048
|
|
|
$
|
44,115
|
|
|
Less:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Purchase of property, plant and equipment
|
|
|
(4,866
|
)
|
|
|
(6,677
|
)
|
|
|
(22,993
|
)
|
|
|
(34,719
|
)
|
|
|
(18,248
|
)
|
|
|
(4,598
|
)
|
|
|
(38,131
|
)
|
|
|
(29,228
|
)
|
|
Software spending
|
|
|
(3,761
|
)
|
|
|
(3,268
|
)
|
|
|
(13,404
|
)
|
|
|
(142
|
)
|
|
|
(1,642
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Proceeds from disposals of property, plant and equipment
|
|
|
—
|
|
|
|
690
|
|
|
|
875
|
|
|
|
212
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other
|
|
|
696
|
|
|
|
—
|
|
|
|
—
|
|
|
|
356
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Free cash flow
|
|
$
|
(5,424
|
)
|
|
$
|
1,845
|
|
|
$
|
81,803
|
|
|
$
|
(13,452
|
)
|
|
$
|
25,132
|
|
|
$
|
(16,686
|
)
|
|
$
|
(33,083
|
)
|
|
$
|
14,887
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash used in investing activities
|
|
|
(7,931
|
)
|
|
|
(18,027
|
)
|
|
|
(44,294
|
)
|
|
|
(34,293
|
)
|
|
|
(497,331
|
)
|
|
|
(4,598
|
)
|
|
|
(38,131
|
)
|
|
|
(29,228
|
)
|
|
Net cash provided by (used in) financing activities
|
|
|
2,202
|
|
|
|
(1,220
|
)
|
|
|
(63,771
|
)
|
|
|
12,768
|
|
|
|
488,547
|
|
|
|
10,296
|
|
|
|
28,375
|
|
|
|
(10,875
|
)
|
52
MANAGEMENT’S
DISCUSSION AND ANALYSIS OF
FINANCIAL CONDITION AND RESULTS OF OPERATIONS
You should read the following discussion and analysis of our
financial condition and results of operations together with our
consolidated financial statements and related notes included
elsewhere in this prospectus. Some of the information contained
in this discussion and analysis or set forth elsewhere in this
prospectus, including information with respect to plans and
strategy for our business and related financing, includes
forward-looking statements that involve risks and uncertainties.
You should review “Risk Factors” and “Cautionary
Note Regarding Forward-Looking Statements” for a discussion
of important factors that could cause actual results to differ
materially from the results described in or implied by the
forward-looking statements contained in the following discussion
and analysis.
The financial and operating data of our predecessor period
from January 1, 2007 to February 28, 2007 and our
successor period from March 1, 2007 to December 31,
2007, have been combined for comparison purposes to present
twelve months of data for 2007. We believe the combined results
of operations, including segment information, for the twelve
months ended December 31, 2007 provide management and
investors with a more meaningful perspective on our financial
and operational performance than if we did not combine the
results of operations of our predecessor and successor periods
in this manner. The combined results of operations are non-GAAP
financial measures, do not include any pro forma assumptions or
adjustments and should not be used in isolation or substitution
of our predecessor and our successor results. See
“— Results of Operations”.
Our
Company
We are the world’s leading supplier of pine-based chemicals
as measured by sales. We refine and further upgrade two primary
feedstocks, crude tall oil, or CTO, and crude sulfate
turpentine, or CST, both of which are wood pulping co-products,
into specialty chemicals. We focus our resources on six target
markets that we believe offer the greatest potential for growth
and in which we offer our highest value-added products. These
markets are: (1) adhesives, (2) inks, (3) tires and rubber, (4)
roads and construction, (5) consumer products and (6) renewable
energy. Our leading position in our target markets is supported
by our recognized brands, including
SYLVATAC®,
SYLVARES®,
SYLVAPRINT®
and
UNI-REZ®,
among others. These products are complemented by a portfolio of
chemical intermediates that includes tall oil rosin, or TOR,
tall oil fatty acid, or TOFA, dimer acid and distilled tall oil,
or DTO, which have contributed steady profit margins and stable
cash flows. These products are sold into a diverse range of
markets, including paints and coatings, lubricants, fuel
additives, mining and oilfield, paper chemicals and polymers,
among others.
While our business is based predominantly on the refining and
upgrading of CTO and CST, we have the capacity to use both
hydrocarbon-based raw materials and gum rosins where appropriate
and, accordingly, are able to offer tailored solutions for our
customers.
Our products and technical support enhance the value of our
customers’ products by improving their performance,
providing them with essential attributes, lowering costs and
simplifying processes. We have cultivated longstanding
relationships with leading customers in our key markets and have
a history of co-developing many of our products with our
customers to satisfy specific product requirements. Our
innovative products and solutions help our customers replace
non-renewable raw materials with more sustainable alternatives.
We serve approximately 1,000 customers in more than 80 countries
through our worldwide network of ten strategically located
manufacturing facilities, two laboratories and five
representative offices.
Our
History
We were founded in 1930 as a joint venture between International
Paper and American Cyanamid to mine salt cake in Camp Verde,
Arizona. By 1949, our core business had changed from mining salt
cake to the processing of CTO and CST. In 1985, we became the
chemical unit of
53
International Paper after they acquired complete ownership from
American Cyanamid. Between 1984 and 2007, we grew from strategic
acquisitions that were made by International Paper resulting in
the integration of various chemical units into our business. On
February 28, 2007, Rhône Capital acquired our business
from International Paper in the Acquisition. See
“Business — Transactions with Rhône Capital
and International Paper” for further information regarding
the Acquisition.
Our
Transformation since the Acquisition
Since the Acquisition, we have reinvigorated our business. We
have engaged a highly motivated and experienced management team
to lead our company and instill a shareholder-value-based
culture focused on sustainable, profitable top-line growth,
improving margins and strong cash flow generation. We have
achieved continued margin growth, in spite of the recently
difficult economic climate, by focusing on value pricing, a
better understanding of our customers, strict management of our
cost structure and obtaining operational efficiencies.
Cost Saving Initiatives. As part of our
business strategy, we initiated a comprehensive cost reduction
program that drives us to continuously reduce our fixed costs,
improve our processes and the way we run our business and better
serve our customers. A key part of our program has been the
optimization of our manufacturing footprint, and we expect our
cost reduction program to achieve additional efficiencies in our
operations in the future.
The following table presents the aggregate positive impact of
our cost saving initiatives for each of the past three years.
These savings include savings from variable costs, fixed
manufacturing costs and selling, general and administration
costs. These savings are measured by initiative and represent
the amount of savings achieved in each year compared to the cost
structure of the prior year.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
Successor
|
|
Combined
|
|
|
|
Year Ended
|
|
Year Ended
|
|
Year Ended
|
|
|
|
December 31,
|
|
December 31,
|
|
December 31,
|
|
|
|
2009
|
|
2008
|
|
2007
|
|
|
|
|
|
(dollars in millions)
|
|
|
|
|
|
|
|
|
|
(Non-GAAP)
|
|
|
|
Savings Achieved
|
|
$
|
28.9
|
|
|
$
|
14.0
|
|
|
$
|
7.6
|
|
These results were achieved through a series of successful
initiatives. Our most notable projects include:
|
|
|
| |
•
|
In the third quarter of 2009, we relocated a portion of our
rosin upgrading capacity from Valdosta, Georgia to our Savannah,
Georgia manufacturing facility, which resulted in manufacturing
and operating efficiencies
year-over-year.
We expect annual costs savings of approximately
$4.2 million resulting from this initiative and have
achieved cost savings of approximately $1.9 million in
2009. During 2008 and 2009, we spent approximately
$19.3 million in capital expenditures in Savannah to
facilitate this relocation. In 2009, we incurred
$0.9 million for one-time termination benefits related to
terminated employees.
|
| |
| |
•
|
In the second quarter of 2009, we closed our Port St. Joe,
Florida manufacturing facility and transferred production to our
larger manufacturing facilities in Panama City, Florida and
Savannah, Georgia to better optimize our existing capacity. As a
result, we recognized restructuring costs of $5.8 million,
impairment charges of $16.7 million and accelerated
depreciation of $1.6 million in 2009. We expect annual
costs savings of approximately $13.7 million resulting from
these initiatives and achieved cost savings of approximately
$7.3 million in 2009. Additional capital expenditures will
be required at both of these manufacturing facilities to
increase their respective capacity, if and as required to meet
market demands. We incurred $1.8 million for one-time
termination benefits related to terminated employees. The
announcement also was a triggering event for impairment testing
purposes. The test resulted in a $16.7 million charge
related to the impairment of long-lived assets. We
|
54
|
|
|
| |
|
also recognized $4.0 million of other qualified costs
related to the decommissioning and demolition of our Port
St. Joe manufacturing facility.
|
|
|
|
| |
•
|
During 2009, we optimized our use of ocean tankers for bulk
shipments of products from the United States to Europe. By
establishing direct routes and identifying a new carrier, we
were able to eliminate costly transloading activities,
generating savings of $2.5 million during 2009.
|
| |
| |
•
|
In the second quarter of 2008, we relocated our polyamide
production from our Bedlington, U.K. manufacturing facility to
our Dover, Ohio manufacturing facility which resulted in the
restructuring of our dimer business, the closure of our
Bedlington, U.K. manufacturing facility, a reduction in
operations at our Chester-le-Street, U.K. manufacturing facility
and the restructuring of certain product lines at our Dover,
Ohio manufacturing facility. As a result, we recognized
restructuring costs of $8.4 million in 2008. We have
achieved cost savings of approximately $6.8 million,
including $4.6 million in 2009 and $2.2 million in
2008 as a result of these actions.
|
| |
| |
•
|
In the first quarter of 2008, we relocated a portion of our
terpene resin production at our Pensacola, Florida manufacturing
facility to our Panama City, Florida manufacturing facility. As
a result, we recognized restructuring costs of $1.0 million
in 2008. We have achieved cost savings of approximately
$0.9 million in 2008 and nominal incremental cost savings
in 2009 as a result of this relocation.
|
| |
| |
•
|
During 2008, we reduced our head count at our manufacturing
facility in Niort, France. As a result, we recognized
restructuring costs of $3.5 million in 2008. We have
achieved cost savings of $0.5 million in 2009 and
$1.8 million in 2008, as a result of these actions.
|
| |
| |
•
|
During 2007 and 2008, following the Acquisition, we reduced our
selling, general and administrative costs through targeted
reductions in staff and management headcount. As a result, we
recognized severance costs of $3.1 and $2.2 million in 2008
and 2007, respectively. These initiatives generated savings of
approximately $11.2 million, including $5.5 million in
2008 and $5.7 million in 2007.
|
We estimate incremental cost savings during 2010 of
approximately $14.0 million related to the 2009 cost
savings program. We expect to continue to expand our successful
productivity improvement initiatives by pursuing operational
efficiencies, optimizing available technologies, maintaining a
lean organizational structure, reducing fixed costs, profiting
from our global procurement organization, rationalizing capacity
and efficiently managing capital spending. We intend to take
advantage of the enhanced flow of information from the
implementation of a new SAP global enterprise resource planning,
or ERP, system, which we completed in the first half of 2010, to
further our efforts to improve our margins, drive cost
productivity and generate additional cost savings.
Investments. As of March 31, 2010,
we have also invested $17.2 million in a new SAP global ERP
system. As part of this project, we believe we have implemented
best practices and processes for our industry. We will derive
the benefits of this significant investment through improved
service to our customers, improved productivity and insight,
better management of our working capital, reduced costs through
increased flexibility and improved financial management and
corporate governance. We have successfully implemented the new
ERP system in both Europe and North America. The majority of the
costs associated with the ERP system have been incurred and
capitalized in 2009 and we expect to capitalize less than
$2 million associated with the implementation in 2010.
Acquisitions. On February 12,
2009, we acquired Abieta Chemie GmbH, which we refer to as
“Abieta”, for $8.8 million net of cash acquired
and assumed debt of $6.0 million. Abieta is a company based
in Gersthofen, Germany that upgrades gum and tall oil rosin, the
acquisition of which has strengthened our position in the tires
and rubber market.
Other Initiatives. On January 1,
2010, we changed the way we transact business with our European
customers and suppliers by realigning to a single business
entity. In the past, each of our European manufacturing
facilities operated as stand alone companies, transacting
business directly with our customers and suppliers. The new
operating structure will provide a single face to our
55
European customers and suppliers, streamlining our processes and
making it easier for them to do business with us. In addition to
better customer service, this initiative is expected to reduce
administrative costs and further reduce our working capital
requirements.
Our
Markets
We supply a number of products to customers in the adhesives,
inks, tires and rubber, roads and construction, consumer
products and renewable energy markets. We also supply a range of
chemical intermediates, including tall oil rosin, or TOR, tall
oil fatty acid, or TOFA, dimer acid and distilled tall oil, or
DTO. These products are described below and in more detail under
“Business — Our Markets”.
Adhesives
We are a leading global supplier of tackifiers to the adhesives
industry as measured by sales, and the world’s largest
producer of tackifier resins from renewable resources in terms
of volume. The products that we supply to the adhesives industry
include the following:
|
|
|
| |
•
|
Rosin-based tackifiers, which are used to improve adhesion to
difficult to bond surfaces such as highly recycled packaging
materials;
|
| |
| |
•
|
Terpene-based tackifiers, which allow our customers to develop
and manufacture adhesives that bond to a wide variety of plastic
substrates;
|
| |
| |
•
|
Hot melt polyamides, which are finished adhesives that bond to a
wide variety of substrates and offer outstanding resistance to
chemicals and oils; and
|
| |
| |
•
|
Alpha-methyl-styrene (AMS)-based tackifiers, which are
hydrocarbon-based tackifiers that are almost water-white and
adhere well to difficult to bond materials.
|
Inks
We are a major supplier of ink resins for use in publishing and
packaging to many of the world’s leading printing inks
companies. These products include:
|
|
|
| |
•
|
Phenolic rosin esters, which are used in lithographic and
publication gravure inks and confer beneficial attributes such
as enhanced adhesion, high gloss, improved drying speed,
viscosity and color intensity;
|
| |
| |
•
|
Solution metal resinates, which are used in inks for publication
gravure, which is used for high quality, large print run
applications such as catalogs and magazines;
|
| |
| |
•
|
Polyamides, which are resins used in inks for flexible packaging
such as bread bags, shrink sleeve labels, high end lamination
and snack food packaging; and
|
| |
| |
•
|
Solvents, which are used primarily in specialty applications
such as vegetable oil-based “sheet-fed” printing to
improve the solvency power of the vegetable oils.
|
Tires and Rubber
We are a leading supplier of resins and additives from renewable
resources to the global tires and rubber industry as measured by
sales. Our products include:
|
|
|
| |
•
|
Polyterpenes, which are used as tire tread enhancement additives
to improve the combination of wet grip, fuel economy and tire
life;
|
| |
| |
•
|
Disproportionated and non-disproportionated rosin soaps, which
are used as emulsifiers in rubber polymerization; and
|
| |
| |
•
|
AMS resins, which are hydrocarbon-based resins used as tire
tread composition additives to improve the combination of wet
grip, fuel economy and tire life.
|
56
Roads and Construction
We are a major supplier of resins for use in roadmarking in
markets in the United States, Europe and the Middle East, and we
believe there are future opportunities to supply fatty acids and
tackifier resins for bitumen applications in the roofing and
paving sub-markets. Our products include:
|
|
|
| |
•
|
Rosin esters and insoluble maleics, which are used in
thermoplastic pavement markings; and
|
| |
| |
•
|
Hot melt polyamides, which are utilized in pre-formed pavement
graphics.
|
Consumer Products
We sell a diverse range of raw materials and ingredients for use
in the formulation of consumer products used in the personal
care home care, industrial cleaning and food ingredients
sub-markets.
Our products include:
|
|
|
| |
•
|
Alpha pinene and beta pinene, which are used as building blocks
in the manufacture of fragrances, camphor, perfumes, terpineol,
terpene resin derivatives and insecticides and are used in the
institutional and industrial markets as cleaners, solvents and
disinfectants;
|
| |
| |
•
|
Sterols, which are pharmaceutical and food additives used to
reduce cholesterol absorption, through our joint venture,
Arboris, LLC;
|
| |
| |
•
|
Specialty polymeric gellants, which are used to impart
structure, rheology, film forming and wear resistance to a
variety of products formulated for the personal care and
consumer products markets; and
|
| |
| |
•
|
Immobilized functional oils, which allow organic liquids to be
solidified into temperature resistant, robust, three dimensional
objects and are currently sold in the automotive air freshener
market in the United States and Europe.
|
Renewable Energy
We supply tall oil pitch, a second generation, cellulosic
biofuel that is used in municipal heating and industrial power
generation, to the European biofuel market. Additionally, in
2009, we began selling TOFA, a co-product of the CTO refining
process, to a third party for conversion into second generation,
cellulosic biodiesel for use as a transportation fuel.
Chemical Intermediates
We produce a portfolio of pine-based chemical intermediates that
are sold into markets as diverse as paints and coatings,
lubricants, fuel additives, mining and oilfield, paper chemicals
and polymer additives. These products include:
|
|
|
| |
•
|
Tall oil rosin, or TOR, which is used in all major rosin
applications for the manufacture of resins for adhesives, inks
and roadmarking, emulsifiers for rubber size for paper and
chewing gum;
|
| |
| |
•
|
Tall oil fatty acid, or TOFA, which, among its other uses, is
used in alkyd paints, primarily for decorative coatings, helping
to bring durability and gloss and is a key component in
additives used to improve the lubricity of low-sulfur diesel
fuel;
|
| |
| |
•
|
Distilled tall oil, or DTO, which is used as an emulsifier for
metalworking fluids;
|
| |
| |
•
|
Dimer acids, which are used for the production of polyamides for
epoxy coatings, flexographic inks, and high performance adhesive
applications; and
|
| |
| |
•
|
Curing agents, which are used to cure (harden) epoxy resins,
primarily those used in marine and protective coatings.
|
57
Factors Affecting
Our Industry and Business
Challenging Macroeconomic Environment
The results of our operations depend on the demand for our
naturally-derived resins and other pine-based chemical products.
Demand for our manufactured products has been significantly
affected in late 2008 and 2009 by the global decline in general
economic conditions. During the fourth quarter of 2008 and into
the first half of 2009, we experienced the impact of the
weakening global economy, which was a factor in the decline in
our sales volumes of approximately 23% for the first half of
2009 compared to the first half of 2008. While the effects of
the global economic slowdown on our business began to subside in
the second half of 2009, demand remained at reduced levels. This
was a significant factor in the 11% decline in our volumes
during the second half of 2009 compared to the second half of
2008. As the global economy stabilizes and we actively seek
future opportunities for sales growth, we expect growth in our
sales volumes during 2010 compared to 2009.
Pricing
Throughout 2009, decreasing global demand led to downward
pricing pressure within several of our end markets. We
continually assess our pricing and, starting in July 2009, we
announced the implementation of a series of price increases in
Europe and North America which were broad-based across our
adhesives and tires and rubber markets, as well as price
increases in our chemical intermediates and inks markets within
Europe in response to increased demand. In some cases, we
decided to forgo lower priced business in order to strengthen
our market position in higher priced specialized end markets,
such as alpha pinene, which is used in aroma chemical
applications. This is in line with our overall strategy to shift
our focus away from lower margin products to higher priced
specialized end markets and applications.
Raw Materials
Raw materials represented approximately 66% and 59% of our cost
of goods sold for the three months ended March 31, 2010 and
2009, respectively, and 60%, 62% and 61% of our cost of goods
sold for the years ended December 31, 2009, 2008 and 2007,
respectively. Our most significant raw materials are CTO and CST
which represented approximately 68% and 64% of our raw material
purchases in the three months ended March 31, 2010 and the
year ended December 31, 2009, respectively. The prices we pay
for CTO tend to be correlated with oil prices. However, the
sales prices for competitive products derived from alternative
chemistries, such as vegetable oils, biofuels and hydrocarbon
resins, tend to also correlate with oil prices. Historically, we
have been successful in passing on raw material cost increases
to our customers.
We obtained approximately 20% and 27% of our global CTO and CST
purchases for the three months ended March 31, 2010 and the
year ended December 31, 2009, respectively, through our
long-term supply agreements with International Paper. These
agreements are terminable by either party on February 28,
2027 and each anniversary thereafter, subject to a five year
notice provision. We believe that through these contractual
arrangements and through our long-standing relationships with
other suppliers, we can presently source adequate supplies of
CTO and CST at competitive, market-based prices in volumes
sufficient for our commercial needs.
For information regarding risks associated with our supply of
CTO and CST, see “Risk Factors — Risks Related to
Raw Materials”.
Additionally, CTO, CST and pitch, a by-product of the CTO
refining process, possess inherent fuel properties and can be
burned by mills for use in their operations. We maintain
agreements with pulp and paper manufacturers in Europe that
enable us to exchange our pitch by-products for their CTO. These
agreements enable us to both maximize our CTO supply and sell
our pitch by-products, while enabling our suppliers to ensure
that they have adequate supplies of renewable fuels for their
mills.
58
Energy Costs
Our energy purchases represented approximately 9% of our cost of
goods sold for the three months ended March 31, 2010 and
2009 and 8%, 9% and 9% of our cost of goods sold for the years
ended December 31, 2009, 2008 and 2007, respectively.
Natural gas is the primary fuel source utilized in our
operations overall, accounting for nearly 45% of our total
energy costs and approximately 65% in North America during 2009.
The supply of natural gas is critical to our production, and
supply disruptions and major price increases could adversely
impact our operating results. We manage this risk, in part, by
purchasing forward contracts for future delivery of natural gas
that mature on a rolling basis over a twelve month period. Our
European operations primarily rely on electricity and internally
generated biofuels, primarily pitch, which accounted for 71% of
our European energy costs in 2009.
Currency Fluctuations
We operate a geographically diverse business. In 2009,
approximately 42% of net sales were to customers in the United
States and Canada, 49% to customers in Europe, the Middle East
and Africa, 5% to customers in Asia, and 4% to customers in
Latin America. Although we sell and manufacture our products in
many countries, our sales and production costs are mainly
denominated in U.S. dollars, Euros, Swedish Kronor and
British Pound Sterling. Our financial results are subject to
gains and losses on transactions denominated in currencies other
than the functional currency of the relevant operations. Any
gains and losses are included in operating income. These
transactional gains and losses were not material in the three
months ended March 31, 2010 or the years ended
December 31, 2009, 2008 and 2007. See “Summary
Selected Historical Consolidated Financial and Other
Data — Other Data”. With respect to currency
translation risk, our financial condition and results of
operations are measured and recorded in the relevant domestic
currency and then translated into U.S. dollars, our
functional currency, for inclusion in our consolidated financial
statements. Approximately half of our net sales and costs are
denominated in U.S. dollars. We also have net sales and
costs denominated in Euros, Swedish Kronor and British Pound
Sterling. We currently do not enter into foreign currency option
or futures contracts to reduce our exposure to fluctuations in
exchange rates.
We are also exposed to foreign currency translation gains and
losses, including the revaluation of the Euro-denominated debt
under our First Lien Credit Agreement, which is recorded by one
of our subsidiaries, Arizona Chemical AB, whose functional
currency is the Swedish Kronor. In the three months ended
March 31, 2010 and the year ended December 31, 2009,
we recognized an unrealized gain of $4.8 million and
$9.3 million, respectively, primarily associated with the
revaluation of our Euro-denominated debt due to the
strengthening of the Swedish Kronor against the Euro. In
contrast, in 2008 and 2007, we recognized unrealized losses of
$20.3 million and $1.9 million, respectively, in our
statements of operation primarily due to a stronger Euro against
the Swedish Kronor. Historically, we have not undertaken hedging
strategies to minimize the effect of currency fluctuations as we
receive proceeds from certain European segment sales in Euros,
which provides sufficient Euro denominated cash flows to service
our Euro-denominated debt.
Seasonality
Historically, our business has been subject to seasonal
fluctuations of raw material inventory, due to the seasonal
trends in availability of CTO and CST. CTO and CST are
co-products of the kraft pulping process. Yields of CTO and CST
are higher during the first half of the year due to the natural
growth and associated chemical yield cycles of trees in addition
to higher yields from kraft pulping during the cooler months.
CTO and CST receipts rise during the first and second quarters,
generally peaking during the early summer. In addition our
business has seasonal fluctuations associated with customer
demand. In preparation for stronger demand in our markets during
the second and third quarters of the year, we build inventory of
finished goods which generally peaks in the second quarter. Many
factors drive the increase in demand in the second and third
quarters, including the holiday printing season (affecting our
customers in the inks market) and the seasonality of the
roadmarking
59
and construction industries, which see increased activity in
warmer months. As a result, demand and consequently sales have
been historically lower in our first and fourth quarters.
The following table presents the percentage of our net sales by
quarter averaged over the three year period ended
December 31, 2009.
| |
|
|
|
|
|
|
|
Net Sales
|
|
|
|
% Total Average
|
|
|
|
Sales
|
|
|
|
First Quarter
|
|
|
23.6
|
%
|
|
Second Quarter
|
|
|
26.4
|
%
|
|
Third Quarter
|
|
|
26.0
|
%
|
|
Fourth Quarter
|
|
|
24.0
|
%
|
Government Policies
Newly promulgated restrictions on the emission of greenhouse
gases in the European Union and pending legislative and
regulatory initiatives in the United States and elsewhere could
affect the market for our products, our costs of production, the
cost or availability of feedstocks and otherwise affect our
operations. For example, demand for renewable sources of energy
is increasing in line with government-mandated targets, often
with associated incentives, to reduce greenhouse gas emissions
and dependency on fossil fuels which, consequently, could
increase demand and the cost to acquire our CTO and CST
feedstocks. At the same time, applicable laws and regulations
require our facilities to obtain and comply with a wide variety
of environmental permits and authorizations for different
aspects of their operations. Generally, these environmental laws
and regulations are becoming increasingly stringent and the cost
of compliance with these various requirements can be expected to
increase over time. Many of the policies being enacted by
governments are being designed to discourage the consumption of
hydrocarbon products. As many of our products are substitutes
for hydrocarbon-based products, we see potential opportunities
to benefit from such changes.
Results of
Operations
We operate in two segments, North America and Europe, each of
which sells a similar portfolio of products. These segments are
our primary areas of measurement and decision-making. The North
America segment is comprised of all operations within the
American and Asian continents. The European segment is comprised
of all operations in the rest of the world. Net sales in each
segment consist of sales of our products to customers in the
following markets: adhesives, inks, roads and construction,
tires and rubber, consumer products, renewable energy and
chemical intermediates. Our chief executive officer evaluates
our performance, in part, based upon each segment’s net
sales and net income.
The following table presents certain information relating to our
operating results for the three months ended March 31, 2010
and 2009, the years ended December 31, 2009 and
December 31, 2008 and combined historical results of
operations for the year ended December 31, 2007. The 2007
combined historical results were derived from the audited
consolidated financial statements of Arizona Chemical Division a
division of International Paper (predecessor) for the period
January 1, 2007 through February 28, 2007 and Arizona
Chem Sweden Holdings AB (successor) for the period from
March 1, 2007 through December 31, 2007. Prior to the
Acquisition, we operated as a division of International Paper.
Accordingly, all operating, financing and investing activities
were funded through inter-divisional accounts at International
Paper and its other operating divisions and subsidiaries. The
financial statements prior to February 28, 2007 were
prepared from separate records maintained by International Paper
and may not be indicative of the conditions that would have
existed or the results of operations if we would have been
operated as an unaffiliated company. During that period, certain
expenses represent allocations of corporate office items
applicable to International Paper as a whole.
60
We believe that the allocations from International Paper
represents all of our costs of doing business incurred on our
behalf.
The historical operating results are being presented to assist
comparison across the years. The successor periods for the three
months ended March 31, 2010 and 2009, the years ended
December 31, 2009 and December 31, 2008 and the period
from March 1, 2007 through December 31, 2007 in the
operating results include the effect of fair value adjustments
and transaction costs resulting from the Acquisition. Due to the
change in the basis of accounting resulting from the
Acquisition, Arizona Chemical Division predecessor’s
consolidated financial statements and Arizona Chem Sweden
Holdings AB successor’s consolidated financial statements
are not completely comparable. We believe that the scope and
nature of our operating activities were unchanged by the
Acquisition and the effects of purchase accounting and increased
debt arising from the Acquisition are accurately quantified and
disclosed throughout our discussion and analysis below.
The combined information for 2007 is a non-GAAP financial
measure and should not be used in isolation or substitution of
the results of operations of either Arizona Chemical Division
predecessor or Arizona Chem Sweden Holdings AB successor. This
data is being presented for informational purposes only and does
not purport to represent or be indicative of the results that
actually would have been obtained had the Acquisition occurred
on January 1, 2007 or that may be obtained for any future
period.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Combined
|
|
|
Successor
|
|
|
Predecessor
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(Non-GAAP)
|
|
|
|
|
|
|
|
|
|
|
Three Months
|
|
|
Three Months
|
|
|
|
|
|
|
|
|
|
|
|
March 1, 2007
|
|
|
January 1, 2007
|
|
|
|
|
Ended
|
|
|
Ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
through
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
February 28,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
2007
|
|
|
2007
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Net sales
|
|
$
|
198,051
|
|
|
$
|
177,934
|
|
|
$
|
767,465
|
|
|
$
|
1,001,988
|
|
|
$
|
855,867
|
|
|
$
|
723,797
|
|
|
$
|
132,070
|
|
|
Cost of goods sold
|
|
|
157,333
|
|
|
|
163,445
|
|
|
|
646,986
|
|
|
|
868,536
|
|
|
|
755,415
|
|
|
|
642,341
|
|
|
|
113,074
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gross profit
|
|
|
40,718
|
|
|
|
14,489
|
|
|
|
120,479
|
|
|
|
133,452
|
|
|
|
100,452
|
|
|
|
81,456
|
|
|
|
18,996
|
|
|
Gross margin
|
|
|
20.6
|
%
|
|
|
8.1
|
%
|
|
|
15.7
|
%
|
|
|
13.3
|
%
|
|
|
11.7
|
%
|
|
|
11.3
|
%
|
|
|
14.4
|
%
|
|
Selling, general and administrative
|
|
|
25,673
|
|
|
|
15,464
|
|
|
|
78,200
|
|
|
|
91,936
|
|
|
|
99,583
|
|
|
|
86,684
|
|
|
|
12,899
|
|
|
Unrealized foreign currency exchange
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(gains) losses
|
|
|
(4,836
|
)
|
|
|
367
|
|
|
|
(9,347
|
)
|
|
|
20,304
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Restructuring and impairment
|
|
|
2,047
|
|
|
|
1,035
|
|
|
|
26,395
|
|
|
|
15,513
|
|
|
|
114
|
|
|
|
114
|
|
|
|
—
|
|
|
Other operating income
|
|
|
—
|
|
|
|
(2,043
|
)
|
|
|
(5,537
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating income (loss)
|
|
|
17,834
|
|
|
|
(334
|
)
|
|
|
30,768
|
|
|
|
5,699
|
|
|
|
755
|
|
|
|
(5,342
|
)
|
|
|
6,097
|
|
|
Interest (expense) income, net
|
|
|
(3,870
|
)
|
|
|
(4,560
|
)
|
|
|
(16,546
|
)
|
|
|
(29,523
|
)
|
|
|
(28,657
|
)
|
|
|
(28,775
|
)
|
|
|
118
|
|
|
Loss on interest rate swaps, net
|
|
|
(709
|
)
|
|
|
(600
|
)
|
|
|
(2,541
|
)
|
|
|
(9,311
|
)
|
|
|
(2,275
|
)
|
|
|
(2,275
|
)
|
|
|
|
|
|
Other income
|
|
|
623
|
|
|
|
2,151
|
|
|
|
3,635
|
|
|
|
1,879
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income (loss) before income tax expense (benefit)
|
|
|
13,878
|
|
|
|
(3,343
|
)
|
|
|
15,316
|
|
|
|
(31,256
|
)
|
|
|
(30,177
|
)
|
|
|
(36,392
|
)
|
|
|
6,215
|
|
|
Income tax expense (benefit)
|
|
|
3,885
|
|
|
|
(836
|
)
|
|
|
3,831
|
|
|
|
(4,277
|
)
|
|
|
(6,299
|
)
|
|
|
(8,913
|
)
|
|
|
2,614
|
|
|
Equity in earnings of affiliate
|
|
|
4
|
|
|
|
150
|
|
|
|
613
|
|
|
|
380
|
|
|
|
273
|
|
|
|
189
|
|
|
|
84
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income (loss)
|
|
$
|
9,997
|
|
|
$
|
(2,357
|
)
|
|
$
|
12,098
|
|
|
$
|
(26,599
|
)
|
|
$
|
(23,605
|
)
|
|
$
|
(27,290
|
)
|
|
$
|
3,685
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
61
Three Months
Ended March 31, 2010 Compared to Three Months Ended
March 31, 2009
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
|
|
|
Successor
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months
|
|
|
|
|
|
Three Months
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Ended
|
|
|
|
|
|
Ended
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 31,
|
|
|
% of
|
|
|
March 31,
|
|
|
% of
|
|
|
|
|
|
%
|
|
|
|
|
2010
|
|
|
sales
|
|
|
2009
|
|
|
sales
|
|
|
Change
|
|
|
Change
|
|
|
|
|
Net sales
|
|
$
|
198,051
|
|
|
|
100.0
|
%
|
|
$
|
177,934
|
|
|
|
100.0
|
%
|
|
$
|
20,117
|
|
|
|
11.3
|
%
|
|
Cost of goods sold
|
|
|
157,333
|
|
|
|
79.4
|
|
|
|
163,445
|
|
|
|
91.9
|
|
|
|
(6,112
|
)
|
|
|
(3.7
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gross profit
|
|
|
40,718
|
|
|
|
20.6
|
|
|
|
14,489
|
|
|
|
8.1
|
|
|
|
26,229
|
|
|
|
181.0
|
|
|
Selling, general and administrative
|
|
|
25,673
|
|
|
|
13.0
|
|
|
|
15,464
|
|
|
|
8.7
|
|
|
|
10,209
|
|
|
|
66.0
|
|
|
Unrealized foreign currency exchange (gains) losses
|
|
|
(4,836
|
)
|
|
|
(2.4
|
)
|
|
|
367
|
|
|
|
0.2
|
|
|
|
(5,203
|
)
|
|
|
(1,417.7
|
)
|
|
Restructuring and impairment
|
|
|
2,047
|
|
|
|
1.0
|
|
|
|
1,035
|
|
|
|
0.6
|
|
|
|
1,012
|
|
|
|
97.8
|
|
|
Other operating income
|
|
|
—
|
|
|
|
—
|
|
|
|
(2,043
|
)
|
|
|
(1.1
|
)
|
|
|
2,043
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating income (loss)
|
|
|
17,834
|
|
|
|
9.0
|
|
|
|
(334
|
)
|
|
|
(0.2
|
)
|
|
|
18,168
|
|
|
|
5,439.5
|
|
|
Interest expense, net
|
|
|
(3,870
|
)
|
|
|
(2.0
|
)
|
|
|
(4,560
|
)
|
|
|
(2.6
|
)
|
|
|
690
|
|
|
|
15.1
|
|
|
Loss on interest rate swaps, net
|
|
|
(709
|
)
|
|
|
(0.4
|
)
|
|
|
(600
|
)
|
|
|
(0.3
|
)
|
|
|
(109
|
)
|
|
|
(18.2
|
)
|
|
Other income
|
|
|
623
|
|
|
|
0.3
|
|
|
|
2,151
|
|
|
|
1.2
|
|
|
|
(1,528
|
)
|
|
|
(71.0
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income (loss) before income tax expense (benefit)
|
|
|
13,878
|
|
|
|
7.0
|
|
|
|
(3,343
|
)
|
|
|
(1.9
|
)
|
|
|
17,221
|
|
|
|
515.1
|
|
|
Income tax expense (benefit)
|
|
|
3,885
|
|
|
|
2.0
|
|
|
|
(836
|
)
|
|
|
(0.5
|
)
|
|
|
4,721
|
|
|
|
564.7
|
|
|
Equity in earnings of affiliates
|
|
|
4
|
|
|
|
0.0
|
|
|
|
150
|
|
|
|
0.1
|
|
|
|
(146
|
)
|
|
|
(97.3
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income (loss)
|
|
$
|
9,997
|
|
|
|
5.0
|
%
|
|
$
|
(2,357
|
)
|
|
|
(1.3
|
)%
|
|
$
|
12,354
|
|
|
|
524.1
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net Sales. Net sales for the three
months ended March 31, 2010 were $198.1 million, an
increase of $20.1 million, or 11.3%, as compared to
$177.9 million for the three months ended March 31,
2009. This increase was due to an increase in volume of
$6.0 million, which was driven by an increase in demand
across all of our end-markets for our products, with the
exception of Inks. Pricing increases contributed
$6.6 million to the increase in net sales, associated with
price increases in our European segment across all markets. The
effects of foreign currency fluctuations resulted in a favorable
impact on net sales of $7.5 million in the three months
ended March 31, 2010, which was primarily due to a
strengthening of the Swedish Kronor and the Euro against the
U.S. dollar. The components of the quarter-to-quarter
change in net sales are as follows:
| |
|
|
|
|
|
Price increase
|
|
|
3.7
|
%
|
|
Volume increase
|
|
|
3.4
|
%
|
|
Favorable impact of foreign currency
|
|
|
4.2
|
%
|
|
|
|
|
|
|
|
Total net sales increase
|
|
|
11.3
|
%
|
Net sales in each of our reportable segments consist of sales of
our products to customers in the following markets: adhesives,
inks, roads and construction, tires and rubber, consumer
products and renewable energy and sales of our chemical
intermediates. The following table presents net sales
62
mix by market for our North American and European segments for
the periods ended March 31, 2010 and March 31, 2009.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
North America
|
|
|
Europe
|
|
|
|
|
Three Months
|
|
|
Three Months
|
|
|
|
|
|
Three Months
|
|
|
Three Months
|
|
|
|
|
|
|
|
Ended
|
|
|
Ended
|
|
|
|
|
|
Ended
|
|
|
Ended
|
|
|
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
|
|
|
Market
|
|
2010
|
|
|
2009
|
|
|
Change
|
|
|
2010
|
|
|
2009
|
|
|
Change
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Adhesives
|
|
$
|
30,305
|
|
|
$
|
26,348
|
|
|
|
15.0
|
%
|
|
$
|
26,132
|
|
|
$
|
20,959
|
|
|
|
24.7
|
%
|
|
Inks
|
|
|
10,599
|
|
|
|
13,638
|
|
|
|
(22.3
|
)%
|
|
|
6,716
|
|
|
|
9,162
|
|
|
|
(26.7
|
)%
|
|
Tires and Rubber
|
|
|
5,531
|
|
|
|
3,190
|
|
|
|
73.4
|
%
|
|
|
10,182
|
|
|
|
8,801
|
|
|
|
15.7
|
%
|
|
Roads and Construction
|
|
|
6,488
|
|
|
|
5,442
|
|
|
|
19.2
|
%
|
|
|
4,284
|
|
|
|
2,857
|
|
|
|
49.9
|
%
|
|
Consumer Products
|
|
|
3,587
|
|
|
|
3,618
|
|
|
|
(0.9
|
)%
|
|
|
4,087
|
|
|
|
1,940
|
|
|
|
110.7
|
%
|
|
Renewable Energy
|
|
|
583
|
|
|
|
632
|
|
|
|
(7.8
|
)%
|
|
|
21,123
|
|
|
|
17,006
|
|
|
|
24.2
|
%
|
|
Chemical Intermediates
|
|
|
39,240
|
|
|
|
32,687
|
|
|
|
20.0
|
%
|
|
|
29,194
|
|
|
|
31,654
|
|
|
|
(7.8
|
)%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total net sales
|
|
$
|
96,333
|
|
|
$
|
85,555
|
|
|
|
12.6
|
%
|
|
$
|
101,718
|
|
|
$
|
92,379
|
|
|
|
10.1
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
North America. Net sales for the three months
ended March 31, 2010 were $96.3 million, an increase
of $10.8 million, or 12.6%, as compared to
$85.5 million for the three months ended March 31,
2009. Volume increases positively impacted net sales by
$11.7 million, offset by unfavorable pricing pressure
resulting in a decrease of $0.9 million.
| |
|
|
|
|
|
Price decline
|
|
|
(1.1
|
)%
|
|
Volume increase
|
|
|
13.7
|
|
|
|
|
|
|
|
|
Total net sales increase
|
|
|
12.6
|
%
|
Several factors contributed to the change in net sales in North
America, including:
|
|
|
| |
•
|
Chemical intermediates net sales increased by $6.6 million
driven by increased demand for dimer acids in Asian markets and
higher volumes in TOFA sales.
|
| |
| |
•
|
Adhesives net sales increased $4.0 million primarily due to
increased market demand for rosin esters.
|
| |
| |
•
|
Inks net sales decreased by $3.0 million driven by loss of
share at a major North American customer and demand weakness in
publication printing.
|
| |
| |
•
|
Tires and rubber net sales increased $2.3 million due
primarily to an increase in orders from a major customer’s
North American operations.
|
Europe. Net sales for the three months ended
March 31, 2010 were $101.7 million, an increase of
$9.3 million, or 10.1%, as compared to $92.4 million
for the three months ended March 31, 2009. The increase in
net sales was driven by price increases of $7.5 million and
a $7.5 million favorable impact of foreign exchange. The
increase in pricing was offset by a decrease in volumes of
$5.7 million due to lower demand for publication printing
inks and decreased sales of chemical intermediates due to lower
levels of TOFA supply during the three months ended
March 31, 2010.
| |
|
|
|
|
|
Price increase
|
|
|
8.1
|
%
|
|
Volume decline
|
|
|
(6.1
|
)
|
|
Favorable impact of foreign currency
|
|
|
8.1
|
|
|
|
|
|
|
|
|
Total net sales increase
|
|
|
10.1
|
%
|
Several factors contributed to the change in net sales in
Europe, including:
|
|
|
| |
•
|
Adhesives net sales increased $5.2 million due to increased
market demand for rosin esters resulting from a decline in
availability of gum rosin resins. As a result, we were able to
increase both volume and price of our rosin esters.
|
63
|
|
|
| |
•
|
Renewable energy net sales increased $4.1 million primarily
due to higher prices of our pitch by-product.
|
| |
| |
•
|
Price increases across all other markets were partially offset
by lower demand for publication printing inks and lower levels
of chemical intermediates supply during the three months ended
March 31, 2010.
|
Cost of Goods Sold. Cost of goods sold
for the three months ended March 31, 2010 was
$157.3 million, a decrease of $6.1 million, or 3.7%,
as compared to $163.4 million for the three months ended
March 31, 2009. Gross margins rose to 20.6% in the three
months ended March 31, 2010 from 8.1% in the three months
ended March 31, 2009.
The decrease of $6.1 million was due primarily to the
following:
|
|
|
| |
•
|
An increase in sales volumes of $2.1 million in the three months
ended March 31, 2010 compared to the three months ended March
31, 2009.
|
| |
| |
•
|
Lower raw material costs of $14.5 million which were a result of
lower CTO and BLS prices primarily due to energy based pricing
on supply contracts which reset every quarter as well as lower
pentaerythritol, sulfuric and TETA prices associated with lower
commodity prices.
|
| |
| |
•
|
Depreciation and amortization expense included within cost of
goods sold for the three months ended March 31, 2010 was $6.6
million, a decrease of $0.5 million compared to $7.1 million in
the three months ended March 31, 2009 due primarily to
manufacturing facility closures in 2009.
|
| |
| |
•
|
The effect of foreign currency fluctuations resulted in an
unfavorable impact on cost of goods sold of $6.9 million for the
three months ended March 31, 2010 primarily due to the
strengthening of the Swedish Kronor and the Euro against the
U.S. dollar.
|
Selling, General and Administrative
Expenses. Selling, general and administrative
expenses for the three months ended March 31, 2010 were
$25.7 million, an increase of $10.2 million, or 66.0%,
as compared to $15.5 million for the three months ended
March 31, 2009. The increase between the periods was due in
part to an increase of $3.2 million of personnel costs
resulting from an investment in marketing and R&D, a
reduction in personnel costs capitalized as part of our
implementation of SAP, and a temporary increase in personnel in
Europe relating to the realignment and transition of our
European operations to a single business entity. In addition to
the increased personnel costs, an additional $2.3 million
of third party professional fees related to the implementation
of SAP and the preparation of this registration statement for
filing with the Securities and Exchange Commission in connection
with our proposed initial offering, a decrease of
$1.8 million in realized transaction related foreign
currency exchange gains, and an unfavorable impact of
$0.6 million due to the strengthening of the Euro against
the U.S. dollar contributed to the increase in selling,
general and administrative expenses for the three months ended
March 31, 2010 as compared to the three months ended March
31, 2009.
Unrealized Foreign Currency Exchange (Gains)
Losses. Unrealized foreign currency exchange
(gains) losses for the three months ended March 31, 2010
was a gain of $4.8 million, an increase of
$5.2 million as compared to a $0.4 million loss for
the three months ended March 31, 2009. The swing from a net
loss to a net gain was primarily associated with the impact of
exchange rate movements of the Swedish Kronor versus the Euro on
our Euro-denominated debt under the First Lien Credit Agreement.
The average exchange rate for the three months ended
March 31, 2010 and the three months ended March 31,
2009 was 9.60 SEK/EUR and 12.18 SEK/EUR, respectively.
Restructuring and
Impairment. Restructuring and impairment
charges for the three months ended March 31, 2010 were
$2.0 million, an increase of $1.0 million, as compared
to $1.0 million for the three months ended March 31,
2009. Restructuring charges for the three months ended
March 31, 2010 were primarily related to employee severance
expenses of $1.2 million for our Sandarne, Sweden
manufacturing facility and $0.7 million for our Niort,
France manufacturing facility as a result of the realignment of
our business in Europe to a single business entity as of
January 1, 2010. Restructuring and impairment charges for
the three months ended March 31, 2009 were primarily
64
related to the impairment of certain fixed assets at our Dover,
Ohio facility. These restructuring and impairment activities
have all been recorded as expenses in the consolidated
statements of operations. This is further described in
Note 7 of our unaudited condensed consolidated financial
statements appearing at the end of this prospectus.
Other Operating Income. No other
operating income was earned during the three months ended
March 31, 2010. Other operating income for the three months
ended March 31, 2009 of $2.0 million was comprised of
$1.4 million business interruption insurance proceeds
related to a fire at our Oulu, Finland manufacturing facility
and $0.7 million related to the gain on the sale of land at
our Bedlington, U.K. manufacturing facility.
Interest Expense, Net. Interest
expense, net for the three months ended March 31, 2010 was
$3.9 million, a decrease of $0.7 million, or 15.1%, as
compared to $4.6 million for the three months ended
March 31, 2009. The decrease was primarily due to lower
average outstanding debt due to principal payments made in 2009.
Loss on Interest Rate Swaps and Interest Rate Caps,
Net. Our loss on interest rate swaps and
interest rate caps was $0.7 million for the three months
ended March 31, 2010, an increase of $0.1 million as
compared to $0.6 million for the three months ended
March 31, 2009. The increase in the loss was primarily
attributable to a $0.7 million loss recognized in the three
months ended March 31, 2010 on our interest rate caps due
to a decline in interest rates partially offset by the
expiration of two interest rate swap contracts with notional
values of $165 million and €40 million at fixed
rates of 4.825% and 3.998%, respectively.
Other Income. Other income was
$0.6 million for the three months ended March 31,
2010, a decrease of $1.5 million as compared to
$2.1 million for the three months ended March 31,
2009. In the three months ended March 31, 2010, we received
$0.7 million related to insurance proceeds to cover the
loss of certain fixed assets due to a fire which took place in
our Sandarne, Sweden manufacturing facility. The
$2.1 million for the three months ended March 31, 2009
relates to the gain we recognized on our acquisition of Abieta
Chemie GmbH.
Income Tax Expense (Benefit). Income
tax expense for the three months ended March 31, 2010 was
$3.9 million, compared to an income tax benefit of
$0.8 million for the three months ended March 31, 2009
driven by higher income. The effective tax rate was 28.0% for
the three months ended March 31, 2010, as compared to 25.0%
for the three months ended March 31, 2009. The change in
the effective tax rate is attributable to a change in the mix of
country pre-tax profits and losses.
Adjusted EBITDA. Adjusted EBITDA for
the three months ended March 31, 2010 was
$27.4 million, an increase of $16.4 million as
compared to $11.0 million for the three months ended
March 31, 2009. This increase was primarily due to the
following:
|
|
|
| |
•
|
Increase in selling prices positively impacted Adjusted EBITDA
by $6.6 million.
|
| |
| |
•
|
Sales volumes positively impacted Adjusted EBITDA by
$3.9 million.
|
| |
| |
•
|
Lower raw material prices positively impacted Adjusted EBITDA by
$14.5 million in the three months ended March 31, 2010
as compared to the three months ended March 31, 2009,
primarily due to an environment of lower commodity prices that
reduced the cost of CTO and our other raw materials, and our
ability to manage our primary raw material costs.
|
| |
| |
•
|
An increase in selling, general and administrative costs
negatively impacted Adjusted EBITDA by $7.3 million after
taking into account adjustments to Adjusted EBITDA.
|
| |
| |
•
|
Other items totaling $1.2 million also negatively impacted
Adjusted EBITDA, which primarily related to the difference of
$1.4 million in insurance proceeds related to a fire at our
Oulu, Finland manufacturing facility and a gain of
$0.7 million on the sale of capital assets realized as a
result of the closure of our Bedlington, U.K. manufacturing
facility in the three months
|
65
|
|
|
| |
|
ended March 31, 2009 and $0.7 million of insurance
proceeds relating to a fire at our Sandarne, Sweden
manufacturing facility recorded in the three months ended
March 31, 2010.
|
|
|
|
| |
•
|
The aggregate effect of foreign currency fluctuations did not
have a significant impact on Adjusted EBITDA.
|
Year Ended
December 31, 2009 Compared to Year Ended December 31,
2008
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
|
|
|
Successor
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Year Ended
|
|
|
|
|
|
Year Ended
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31,
|
|
|
% of
|
|
|
December 31,
|
|
|
% of
|
|
|
|
|
|
%
|
|
|
|
|
2009
|
|
|
sales
|
|
|
2008
|
|
|
sales
|
|
|
Change
|
|
|
Change
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Net sales
|
|
$
|
767,465
|
|
|
|
100.0
|
%
|
|
$
|
1,001,988
|
|
|
|
100.0
|
%
|
|
$
|
(234,523
|
)
|
|
|
(23.4
|
)%
|
|
Cost of goods sold
|
|
|
646,986
|
|
|
|
84.3
|
|
|
|
868,536
|
|
|
|
86.7
|
|
|
|
(221,550
|
)
|
|
|
(25.5
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gross profit
|
|
|
120,479
|
|
|
|
15.7
|
|
|
|
133,452
|
|
|
|
13.3
|
|
|
|
(12,973
|
)
|
|
|
(9.7
|
)
|
|
Selling, general and administrative
|
|
|
78,200
|
|
|
|
10.2
|
|
|
|
91,936
|
|
|
|
9.2
|
|
|
|
(13,736
|
)
|
|
|
(14.9
|
)
|
|
Unrealized foreign currency exchange (gains) losses
|
|
|
(9,347
|
)
|
|
|
(1.2
|
)
|
|
|
20,304
|
|
|
|
2.0
|
|
|
|
(29,651
|
)
|
|
|
(146.0
|
)
|
|
Restructuring and impairment
|
|
|
26,395
|
|
|
|
3.4
|
|
|
|
15,513
|
|
|
|
1.5
|
|
|
|
10,882
|
|
|
|
70.1
|
|
|
Other operating income
|
|
|
(5,537
|
)
|
|
|
(0.7
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
(5,537
|
)
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating gain (loss)
|
|
|
30,768
|
|
|
|
4.0
|
|
|
|
5,699
|
|
|
|
0.6
|
|
|
|
25,069
|
|
|
|
439.9
|
|
|
Interest expense, net
|
|
|
(16,546
|
)
|
|
|
(2.2
|
)
|
|
|
(29,523
|
)
|
|
|
(2.9
|
)
|
|
|
12,977
|
|
|
|
44.0
|
|
|
Loss on interest rate swaps, net
|
|
|
(2,541
|
)
|
|
|
(0.3
|
)
|
|
|
(9,311
|
)
|
|
|
(0.9
|
)
|
|
|
6,770
|
|
|
|
72.7
|
|
|
Other income
|
|
|
3,635
|
|
|
|
0.5
|
|
|
|
1,879
|
|
|
|
0.2
|
|
|
|
1,756
|
|
|
|
93.5
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income (loss) before income tax expense (benefit)
|
|
|
15,316
|
|
|
|
2.0
|
|
|
|
(31,256
|
)
|
|
|
(3.1
|
)
|
|
|
46,572
|
|
|
|
149.0
|
|
|
Income tax expense (benefit)
|
|
|
3,831
|
|
|
|
0.5
|
|
|
|
(4,277
|
)
|
|
|
(0.4
|
)
|
|
|
8,108
|
|
|
|
189.6
|
|
|
Equity in earnings of affiliate
|
|
|
613
|
|
|
|
0.1
|
|
|
|
380
|
|
|
|
0.0
|
|
|
|
233
|
|
|
|
61.3
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income (loss)
|
|
$
|
12,098
|
|
|
|
1.6
|
%
|
|
|
(26,599
|
)
|
|
|
(2.7
|
)%
|
|
$
|
38,697
|
|
|
|
145.5
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net Sales. Net sales for 2009 were
$767.5 million, a decrease of $234.5 million or 23.4%
as compared to $1,002.0 million for 2008. This decrease was
due to a decline in volume of $198.8 million, which was
driven by a decrease in demand as a result of the recent global
economic downturn, which negatively impacted sales in most of
our markets, except for consumer products. Despite the decline
in volume, we were able to keep pricing relatively flat
year-over-year
in an environment of declining raw material prices, as we
managed our sales mix. Average pricing remained relatively flat
from 2009 to 2008. The effects of foreign currency fluctuations
resulted in an unfavorable impact on net sales of
$51.8 million in 2009, which was primarily due to the
strengthening of the U.S. dollar against the Swedish
Kronor. These decreases were partially offset by our acquisition
of Abieta in 2009, which increased net sales within our tires
and rubber market by $20.4 million.
The components of the
year-over-year
change in net sales are as follows:
| |
|
|
|
|
|
Price decline
|
|
|
(0.4
|
)%
|
|
Volume decline
|
|
|
(19.8
|
)
|
|
Acquisition of Abieta
|
|
|
2.0
|
|
|
Unfavorable impact of foreign currency
|
|
|
(5.2
|
)
|
|
|
|
|
|
|
|
Total net sales decrease
|
|
|
(23.4
|
)%
|
|
|
|
|
|
|
66
Net sales in each of our reportable segments consist of sales of
our products to customers in the following markets: adhesives,
inks, roads and construction, tires and rubber, consumer
products and renewable energy and sales of our chemical
intermediates. The following table presents net sales mix by
market for our North American and European segments for the
periods ended December 31, 2009 and December 31, 2008.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
North America
|
|
|
Europe
|
|
|
Market
|
|
2009
|
|
|
2008
|
|
|
Change
|
|
|
2009
|
|
|
2008
|
|
|
Change
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Adhesives
|
|
$
|
104,469
|
|
|
$
|
129,515
|
|
|
|
(19.3
|
)%
|
|
$
|
92,229
|
|
|
$
|
109,103
|
|
|
|
(15.5
|
)%
|
|
Inks
|
|
|
54,102
|
|
|
|
69,251
|
|
|
|
(21.9
|
)
|
|
|
31,941
|
|
|
|
51,777
|
|
|
|
(38.3
|
)
|
|
Tires and Rubber
|
|
|
15,724
|
|
|
|
16,049
|
|
|
|
(2.0
|
)
|
|
|
43,937
|
|
|
|
23,093
|
|
|
|
90.3
|
|
|
Roads and Construction
|
|
|
28,762
|
|
|
|
34,990
|
|
|
|
(17.8
|
)
|
|
|
14,911
|
|
|
|
18,847
|
|
|
|
(20.9
|
)
|
|
Consumer Products
|
|
|
26,836
|
|
|
|
18,651
|
|
|
|
43.9
|
|
|
|
13,316
|
|
|
|
12,426
|
|
|
|
7.2
|
|
|
Renewable Energy
|
|
|
2,815
|
|
|
|
8,577
|
|
|
|
(67.2
|
)
|
|
|
71,456
|
|
|
|
115,580
|
|
|
|
(38.2
|
)
|
|
Chemical Intermediates
|
|
|
146,394
|
|
|
|
215,212
|
|
|
|
(32.0
|
)
|
|
|
120,573
|
|
|
|
178,917
|
|
|
|
(32.6
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total net sales
|
|
$
|
379,102
|
|
|
$
|
492,245
|
|
|
|
(23.0
|
)%
|
|
$
|
388,363
|
|
|
$
|
509,743
|
|
|
|
(23.8
|
)%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
North America. Net sales of products sold by
our North American segment for 2009 were $379.1 million, a
decrease of $113.1 million, or 23.0%, as compared to
$492.2 million for 2008. The downturn in the global economy
resulted in decreased volume across many of the markets within
our North American segment which negatively impacted net sales
by $111.1 million while price declines negatively impacted
net sales by $2.1 million.
| |
|
|
|
|
|
Price decline
|
|
|
(0.4
|
)%
|
|
Volume decline
|
|
|
(22.6
|
)%
|
|
Impact of foreign currency exchange
|
|
|
0.0
|
%
|
|
|
|
|
|
|
|
Total net sales decrease
|
|
|
(23.0
|
)%
|
|
|
|
|
|
|
Several factors contributed to the change in net sales in North
America, including:
|
|
|
| |
•
|
Chemical intermediates net sales decreased by $68.8 million
from 2008 driven by deteriorating global economic conditions
and, in particular, lower demand in housing, automotive,
coatings and oilfield applications.
|
| |
| |
•
|
Adhesives net sales decreased $25.0 million primarily due
to lower demand in hot melt packaging, while inks net sales
declined $15.1 million driven by demand weakness in
publication printing.
|
| |
| |
•
|
Despite lower demand, we strategically focused on reducing the
portion of our sales mix attributable to lower margin products.
Consumer products net sales increased by $8.2 million
primarily due to shifting our sales mix from certain lower
priced adhesive products to higher priced consumer products.
|
| |
| |
•
|
Roads and construction net sales decreased by $6.2 million,
primarily due to a decrease in sales of our rosin resin products
as a result of aggressive competitor pricing, which we chose not
to match, as we shifted our sales mix to higher priced markets.
|
| |
| |
•
|
Renewable energy net sales decreased by $5.8 million,
primarily due to a decrease in throughput of CTO and a
corresponding reduction in the supply of our pitch by-product.
|
Europe. Net sales of products sold by our
European segment for 2009 were $388.4 million, a decrease
of $121.4 million or 23.8% as compared to
$509.7 million in 2008. The downturn in the global economy
resulted in lower volumes across many of the markets within our
European segment. The decrease in net sales was driven by volume
decreases of $87.7 million, partially offset by the
addition of the Abieta acquisition which positively impacted net
sales by $20.4 million. Notwithstanding lower demand,
67
we strategically focused on reducing the portion of our sales
mix attributable to lower margin products and as a result, we
were able to maintain or increase pricing in some of our
markets. Our pricing was also negatively impacted by lower
global vegetable oil commodity prices and lower oil prices,
which impacted pricing of substitutes for several of our
products, including TOFA and pitch. Price declines negatively
impacted net sales by $2.4 million while foreign currency
fluctuations negatively impacted net sales by
$51.8 million, primarily associated with the weakening of
the Swedish Kronor versus the U.S. dollar.
| |
|
|
|
|
|
Price decline
|
|
|
(0.4
|
)%
|
|
Volume decline
|
|
|
(17.2
|
)
|
|
Acquisition of Abieta
|
|
|
4.0
|
|
|
Unfavorable impact of foreign currency
|
|
|
(10.2
|
)
|
|
|
|
|
|
|
|
Total net sales decrease
|
|
|
(23.8
|
)%
|
|
|
|
|
|
|
Several factors contributed to the change in net sales in
Europe, including:
|
|
|
| |
•
|
Chemical intermediates net sales decreased by $58.3 million
due to lower demand in coatings, metal workings and automotive.
In addition, prices were negatively impacted by a pricing
decline in competing global vegetable oil used in coatings,
consistent with the declining pricing environment for oil and
biofuels.
|
| |
| |
•
|
Renewable energy net sales decreased $44.1 million as
volume declined primarily due to a decrease in availability of
our pitch by-product for sale as a result of lower CTO
throughput, and a reduction in oil prices which negatively
impacted pricing.
|
| |
| |
•
|
Tires and rubber net sales increased by $20.8 million
primarily due to our acquisition of Abieta.
|
| |
| |
•
|
Inks and adhesives net sales declined by $19.8 million and
$16.9 million, respectively, driven by lower demand in
printing ink and hot melt packaging applications, respectively.
|
| |
| |
•
|
Roads and construction net sales decreased by $3.9 million,
primarily due to a decline in net sales of our roadmarking
products. The roadmarking industry was less impacted by the
global economic downturn than some of our other markets as a
result of increased government spending, however, our
year-over-year sales were lower due to shortened customer order
lead times during the peak summer season accompanied by
inventory shortages.
|
| |
| |
•
|
We strategically focused on reducing the portion of our sales
mix attributable to lower margin products and as a result, our
consumer products net sales increased by $0.9 million
primarily due to shifting our sales mix to higher priced
consumer products.
|
Cost of Goods Sold. Cost of goods sold
for 2009 was $647.0 million, a decrease of
$221.6 million or 25.5% as compared to $868.5 million
for 2008. Gross margins rose to 15.7% in 2009 from 13.3% in 2008.
The decrease of $221.6 million was due primarily to the
following:
|
|
|
| |
•
|
Declining sales volumes of $113.4 million.
|
| |
| |
•
|
Declining raw material prices of $40.6 million due
primarily to lower CTO and pentaerythritol prices associated
with lower oil and commodity prices.
|
| |
| |
•
|
Favorable energy prices of $11.1 million as compared to
2008 primarily a result of lower natural gas prices in North
America of $10.4 million.
|
| |
| |
•
|
Restructuring initiatives at certain of our manufacturing
facilities in the United States and Europe yielded cost savings
of $17.6 million in 2009, partially offset by an increase
in fixed costs of $5.0 million associated with the addition
of the Gersthofen, Germany manufacturing facility from our
Abieta acquisition. A focus on cost control and lower production
rates as a result of the economic environment further reduced
manufacturing fixed costs by $6.4 million.
|
68
|
|
|
| |
•
|
Depreciation and amortization included in cost of goods sold for
2009 was $31.3 million, an increase of $6.7 million as
compared to $24.6 million for 2008. Depreciation and
amortization in 2009 includes $1.8 million of depreciation
for the newly acquired Gersthofen, Germany manufacturing
facility.
|
| |
| |
•
|
The effect of foreign currency fluctuations resulted in a
favorable impact on cost of goods sold of $44.1 million in
2009 primarily due to the strengthening of the U.S. dollar
against the Swedish Kronor.
|
Selling, General and Administrative
Expenses. Selling, general and administrative
expenses for 2009 were $78.2 million, a decrease of
$13.7 million, or 14.9%, as compared to $91.9 million
for 2008. The decrease was primarily due to lower transaction
and transition costs of $14.8 million, primarily related to
the Acquisition, as well as a $4.6 million reduction in
salary expense, as salaries associated with our ERP
implementation were capitalized, favorable foreign exchange
impact of $2.9 million, primarily due to the weakening of
the Swedish Kronor to the U.S. dollar. These decreases were
partially offset by increased training and other ERP
implementation costs of $2.3 million, increased incentive
compensation benefits and personnel costs of $3.5 million,
which were primarily due to performance above plan and other
administrative cost increases of $2.8 million. Selling,
general and administrative expenses as a percentage of net sales
increased 1.0% to 10.2% in 2009, compared to 9.2% in 2008.
Unrealized Foreign Currency Exchange (Gains)
Losses. Unrealized foreign currency exchange
losses (gains) for 2009 was a gain of $9.3 million, an
increase of $29.7 million as compared to a
$20.3 million loss for 2008. The swing from a net loss to a
gain was primarily associated with the impact of exchange rate
movements of the Swedish Kronor versus the Euro on our
Euro-denominated debt under the First Lien Credit Agreement. The
average exchange rate for 2009 and 2008 was 10.60 SEK/EUR and
9.58 SEK/EUR, respectively.
Restructuring and
Impairment. Restructuring and impairment
charges for 2009 were $26.4 million, an increase of
$10.9 million, or 70.1%, as compared to $15.5 million
for 2008. Restructuring charges for 2009 were primarily related
to the closure of our Port St. Joe, Florida manufacturing
facility. During 2008, we closed our Bedlington, U.K.
manufacturing facility, reduced capacity at our
Chester-le-Street, U.K. manufacturing facility, restructured
certain product lines at our Dover, Ohio manufacturing facility
and restructured our operations at our Niort, France
manufacturing facility. These restructuring activities have all
been recorded as expenses in the consolidated statements of
operations. We have paid for substantially all of these
restructuring activities as of December 31, 2009 and expect
to incur the remaining cash payments of $2.0 million in
2010. This is further described in Note 9 of our
consolidated financial statements appearing at the end of this
prospectus.
Other Operating Income. Other operating
income was $5.5 million in 2009, an increase of
$5.5 million compared to 2008. Other operating income is
primarily comprised of insurance proceeds of $4.9 million
to reimburse us for lost sales and related costs incurred due to
a fire at our Oulu, Finland manufacturing facility in 2008 and a
$0.7 million gain on sale of land related to the closure of our
Bedlington, U.K. manufacturing facility.
Interest Expense, Net. Interest
expense, net for 2009 was $16.5 million, a decrease of
$13.0 million, or 44.0%, as compared to $29.5 million
for 2008. The decrease was primarily due to lower interest rates
in 2009 compared to 2008 on our floating rate debt, combined
with lower average outstanding debt due to revolver reductions
and principal payments in 2009. We expect our interest expense
to decline further in 2010 due to the expiration of two swap
contracts with notional values of $165 million and
€40 million at fixed rates of 4.825% and 3.998%,
respectively.
Loss on Interest Rate Swaps, Net. Our
loss on interest rate swaps was $2.5 million in 2009, a
decrease of $6.8 million, or 72.7%, as compared to
$9.3 million for 2008. The losses on the swap contracts
resulted principally from declines in LIBOR.
Other Income. Other income was
$3.6 million in 2009, an increase of $1.8 million as
compared to $1.9 million for 2008. In 2009, we recorded a
gain of approximately $2.1 million on our
69
acquisition of Abieta and a gain of $1.3 million on a
settlement with International Paper. In 2008, other income
included a gain of $1.9 million on the extinguishment of a
portion of our debt. During 2008, Rhône Capital purchased
$27.2 million face value of our outstanding second lien
debt. In accordance with our credit agreements, Rhône
Capital contributed 35% of the debt, or $9.5 million face
value, to AZ Chem Investments Partner LP, which debt was then
contributed to us and extinguished, resulting in a gain of
$1.9 million that was recognized as other income in 2008.
Income Tax Expense (Benefit). Income
tax expense for 2009 was $3.8 million, an increase of
$8.1 million compared to the income tax benefit of
$4.3 million for 2008 driven by higher income. The
effective tax rate was 25.0% for 2009, as compared to 13.7% for
2008. Our effective tax rate for 2009 was less than our
statutory Swedish income tax rate of 26.3% primarily due to
valuation allowances by our international subsidiaries for
certain tax benefits that were considered not likely.
Adjusted EBITDA. Adjusted EBITDA for
2009 was $93.9 million, an increase of $1.1 million,
or 1.2%, as compared to $92.7 million for 2008. This
increase was due to the following:
|
|
|
| |
•
|
Declines in volume and lower selling prices in 2009 as compared
to 2008 negatively impacted Adjusted EBITDA by
$64.9 million and $4.4 million respectively.
|
| |
| |
•
|
Raw material prices declined $40.6 million in 2009 as
compared to 2008, primarily due to an environment of lower oil
prices and commodity prices that reduced the cost of CTO and our
other raw materials, and our ability to manage our primary raw
material costs.
|
| |
| |
•
|
Energy prices were $11.1 million lower in 2009 as compared
to 2008 primarily due to declining natural gas prices.
|
| |
| |
•
|
Lower manufacturing fixed costs of $24.0 million were
incurred during 2009 as compared to 2008 due to the partial year
savings resulting from the closure of our Port St. Joe, Florida
manufacturing facility in August 2009 and other cost saving
initiatives within both of our reportable segments, partially
offset by $5.0 million of increased costs associated with
the addition of our Gersthofen, Germany facility from the Abieta
acquisition.
|
| |
| |
•
|
Unfavorable foreign currency exchange of $4.8 million and
higher selling, general and administration costs of
$2.3 million after taking into account adjustments to
Adjusted EBITDA.
|
| |
| |
•
|
Favorable impact from the receipt of $4.9 million of
insurance proceeds to reimburse us for lost sales and costs
incurred during 2008 as a result of a fire at our Oulu, Finland
manufacturing facility and other favorable items of
$1.9 million.
|
During 2009, unusual items in Adjusted EBITDA included the
insurance proceeds from the fire at our Oulu, Finland facility
of $4.9 million, the capitalization of salaries, net of
additional expenses, for the implementation of SAP of
$2.3 million and the gain on sale of land of
$0.7 million related to the closure of our
Bedlington, U.K. manufacturing facility.
Despite a 23.4% decrease in net sales, we increased our Adjusted
EBITDA
year-over-year
in an economic environment of decreasing demand and volumes. We
were able to increase our Adjusted EBITDA margin by proactively
managing pricing in an environment that saw significant price
pressures while decreasing our cost for raw materials and
energy. We managed our cost structure through spending controls
and restructuring activities that reduced production capacity
and related costs in order to meet lower volume needs. Moreover,
we were able to selectively shift products sold from lower
margin to higher margin markets, which we expect to continue to
do as we increasingly focus on our target markets.
Year Ended
December 31, 2008 Compared to the Year Ended
December 31, 2007
The following table presents combined historical results of
operations for the year ended December 31, 2007, and was
derived from the audited consolidated financial statements of
our
70
predecessor period January 1, 2007 through
February 28, 2007 and our successor period from
March 1, 2007 through December 31, 2007.
The historical operating results are being presented to assist
comparison across the years. The successor period for 2007 and
2008 in the operating results includes the effect of fair value
adjustments resulting from the Acquisition. Due to this change
in the basis of accounting, our predecessor consolidated
financial statements and our successor consolidated financial
statements are not necessarily comparable.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Combined
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(Non-GAAP)
|
|
|
|
|
|
|
|
|
|
|
Year Ended
|
|
|
|
|
|
Year Ended
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31,
|
|
|
% of
|
|
|
December 31,
|
|
|
% of
|
|
|
|
|
|
|
|
|
|
|
2008
|
|
|
Sales
|
|
|
2007
|
|
|
Sales
|
|
|
Change
|
|
|
% Change
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Net sales
|
|
$
|
1,001,988
|
|
|
|
100.0
|
%
|
|
$
|
855,867
|
|
|
|
100.0
|
%
|
|
$
|
146,121
|
|
|
|
17.1
|
%
|
|
Cost of goods sold
|
|
|
868,536
|
|
|
|
86.7
|
|
|
|
755,415
|
|
|
|
88.3
|
|
|
|
113,121
|
|
|
|
15.0
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gross profit
|
|
|
133,452
|
|
|
|
13.3
|
|
|
|
100,452
|
|
|
|
11.7
|
|
|
|
33,000
|
|
|
|
32.9
|
|
|
Selling, general and administrative
|
|
|
91,936
|
|
|
|
9.2
|
|
|
|
99,583
|
|
|
|
11.6
|
|
|
|
(7,647
|
)
|
|
|
(7.7
|
)
|
|
Unrealized foreign currency transaction and exchange
losses — net
|
|
|
20,304
|
|
|
|
2.0
|
|
|
|
—
|
|
|
|
—
|
|
|
|
20,304
|
|
|
|
—
|
|
|
Restructuring and impairment
|
|
|
15,513
|
|
|
|
1.5
|
|
|
|
114
|
|
|
|
0.0
|
|
|
|
15,399
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating gain (loss)
|
|
|
5,699
|
|
|
|
0.6
|
|
|
|
755
|
|
|
|
0.1
|
|
|
|
4,944
|
|
|
|
654.8
|
|
|
Interest expense, net
|
|
|
(29,523
|
)
|
|
|
(2.9
|
)
|
|
|
(28,657
|
)
|
|
|
(3.3
|
)
|
|
|
(866
|
)
|
|
|
(3.0
|
)
|
|
Loss on interest rate swaps, net
|
|
|
(9,311
|
)
|
|
|
(0.9
|
)
|
|
|
(2,275
|
)
|
|
|
(0.3
|
)
|
|
|
(7,036
|
)
|
|
|
(309.3
|
)
|
|
Other income
|
|
|
1,879
|
|
|
|
0.2
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,879
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss before income tax benefit
|
|
|
(31,256
|
)
|
|
|
(3.1
|
)
|
|
|
(30,177
|
)
|
|
|
(3.5
|
)
|
|
|
(1,079
|
)
|
|
|
(3.6
|
)
|
|
Income tax benefit
|
|
|
(4,277
|
)
|
|
|
(0.4
|
)
|
|
|
(6,299
|
)
|
|
|
(0.7
|
)
|
|
|
2,022
|
|
|
|
32.1
|
|
|
Equity earnings of affiliate
|
|
|
380
|
|
|
|
0.0
|
|
|
|
273
|
|
|
|
0.0
|
|
|
|
107
|
|
|
|
39.2
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss
|
|
$
|
(26,599
|
)
|
|
|
(2.7
|
)%
|
|
$
|
(23,605
|
)
|
|
|
(2.8
|
)%
|
|
$
|
(2,994
|
)
|
|
|
(12.7
|
)%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net Sales. Net sales for 2008 were
$1,002.0 million, an increase of $146.1 million, or
17.1%, as compared to $855.9 million for 2007. The increase
in net sales was driven by pricing increases of
$86.6 million and volume increases of $38.8 million.
We were able to increase prices and capitalize on increased
global demand within many of our markets as we began to focus
more on value-added benefits of our products to our customers.
The effects of foreign currency fluctuations provided a
favorable impact to net sales of $20.7 million in 2008 as
compared to 2007. The favorable impact was primarily due to the
strengthening of the Euro and the Swedish Kronor against the
U.S. dollar.
The components of the
year-over-year
change in net sales are as follows:
| |
|
|
|
|
|
Price increase
|
|
|
10.1
|
%
|
|
Volume increase
|
|
|
4.6
|
|
|
Favorable impact of foreign currency
|
|
|
2.4
|
|
|
|
|
|
|
|
|
Total net sales increase
|
|
|
17.1
|
%
|
|
|
|
|
|
|
71
The following table presents consolidated net sales mix by
market for the periods ended December 31, 2008 and
December 31, 2007 for our two reportable segments, North
America and Europe.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
North America
|
|
|
Europe
|
|
|
|
|
|
|
|
Combined
|
|
|
|
|
|
|
|
|
Combined
|
|
|
|
|
|
|
|
|
|
|
Non-GAAP
|
|
|
|
|
|
|
|
|
Non-GAAP
|
|
|
|
|
|
Market
|
|
2008
|
|
|
2007
|
|
|
Change
|
|
|
2008
|
|
|
2007
|
|
|
Change
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Adhesives
|
|
$
|
129,515
|
|
|
$
|
130,412
|
|
|
|
(0.7
|
)%
|
|
$
|
109,103
|
|
|
$
|
102,681
|
|
|
|
6.3
|
%
|
|
Inks
|
|
|
69,251
|
|
|
|
64,734
|
|
|
|
7.0
|
|
|
|
51,777
|
|
|
|
64,341
|
|
|
|
(19.5
|
)
|
|
Tires and Rubber
|
|
|
16,049
|
|
|
|
13,534
|
|
|
|
18.6
|
|
|
|
23,093
|
|
|
|
23,935
|
|
|
|
(3.5
|
)
|
|
Roads and Construction
|
|
|
34,990
|
|
|
|
20,029
|
|
|
|
74.7
|
|
|
|
18,847
|
|
|
|
12,905
|
|
|
|
46.0
|
|
|
Consumer Products
|
|
|
18,651
|
|
|
|
18,879
|
|
|
|
(1.2
|
)
|
|
|
12,426
|
|
|
|
12,080
|
|
|
|
2.9
|
|
|
Renewable Energy
|
|
|
8,577
|
|
|
|
8,408
|
|
|
|
2.0
|
|
|
|
115,580
|
|
|
|
62,511
|
|
|
|
84.9
|
|
|
Chemical Intermediates
|
|
|
215,212
|
|
|
|
176,730
|
|
|
|
21.8
|
|
|
|
178,917
|
|
|
|
144,688
|
|
|
|
23.7
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total net sales
|
|
$
|
492,245
|
|
|
$
|
432,726
|
|
|
|
13.8
|
%
|
|
$
|
509,743
|
|
|
$
|
423,141
|
|
|
|
20.5
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
North America. Net sales generated by our
North American segment for 2008 were $492.2 million, an
increase of $59.5 million, or 13.8%, as compared to
$432.7 million for 2007. The increase in net sales was
driven by pricing increases of $33.6 million primarily due
to our management of sales mix and our ability to pass through
raw material price increases to our customers, and an increase
in volume of $26.0 million.
| |
|
|
|
|
|
Price increase
|
|
|
7.8
|
%
|
|
Volume increase
|
|
|
6.0
|
|
|
Impact of foreign currency exchange
|
|
|
0.0
|
|
|
|
|
|
|
|
|
Total net sales increase
|
|
|
13.8
|
%
|
|
|
|
|
|
|
Several factors contributed to the growth in net sales in North
America, including:
|
|
|
| |
•
|
Chemical intermediates net sales increased by $38.5 million
driven by increased volumes and prices due to increased demand
in the PVC stabilizers, oilfield, coatings and metalworking
applications.
|
| |
| |
•
|
Roads and construction net sales increased by
$15.0 million, primarily due to increased sales of
roadmarking resins.
|
| |
| |
•
|
Inks net sales increased by $4.5 million, primarily due to
volume increases in publication printing ink applications,
without a reduction in our pricing.
|
| |
| |
•
|
Tires and rubber net sales increased $2.5 million due to an
introduction of new resins into the tire market. In addition,
net sales of upgraded rosins increased due to increased demand
for rubber emulsification applications.
|
Europe. Net sales for 2008 of
$509.7 million increased by $86.6 million, or 20.5%,
as compared to $423.1 million for 2007. The increase in net
sales was primarily driven by pricing increases of
$53.1 million across most of our markets, a favorable
foreign exchange impact of $20.7 million and a favorable
volume increase of $12.8 million. Within our European
segment, the rise in oil prices and increased demand for
competing products favorably impacted sales prices. The impact
was higher in our European segment compared to North America as
the majority of our global pitch is sold into the Scandinavian
energy market.
72
| |
|
|
|
|
|
Price increase
|
|
|
12.5
|
%
|
|
Volume increase
|
|
|
3.1
|
|
|
Favorable impact of foreign currency
|
|
|
4.9
|
|
|
|
|
|
|
|
|
Total net sales increase
|
|
|
20.5
|
%
|
|
|
|
|
|
|
Several factors contributed to the growth in net sales in
Europe, including:
|
|
|
| |
•
|
Renewable energy net sales increased by $53.1 million due
to an increase in volume, price and a favorable impact of
foreign exchange. The volume increase in net sales is
attributable to a reduction of inventory levels during 2008,
from 2007 highs, in advance of increased prices in the first
quarter of 2008.
|
| |
| |
•
|
Chemical intermediates net sales increased by
$34.2 million, primarily due to an increase in TOFA prices
driven by the increase in demand and pricing for competing
vegetable oils in the renewable energy and coatings market. TOFA
volumes declined as a result of a fire that caused a shutdown in
2008 of our Oulu, Finland manufacturing facility. Curing agent
and dimer acid prices increased due to higher demand in coatings
applications, while DTO prices increased due to stronger demand
in the automotive industry combined with improved strategic
product positioning.
|
| |
| |
•
|
Inks net sales decreased by $12.6 million, primarily due to
decreases in sales volumes of phenolics for printing inks due to
competitive pricing, which we choose not to meet in order to
focus on higher margin products in other markets. In 2008 we
also exited the solid resinate product line.
|
| |
| |
•
|
Adhesives net sales increased by $6.4 million, primarily
due to increased sales for hot melt packaging. We reduced our
prices to defend our market share against foreign competition
and imports, due to an unusually strong Euro compared to other
foreign currencies.
|
| |
| |
•
|
Roads and construction net sales increased by $5.9 million,
primarily due to increased volumes for roadmarking, which was
partially offset by a decline in prices to increase volumes and
to respond to opportunistic imports from foreign competitors
associated with a stronger Euro.
|
Cost of Goods Sold. Cost of goods sold
for 2008 was $868.5 million, an increase of
$113.1 million, or 15.0%, as compared to
$755.4 million for 2007. Gross margins rose to 13.3% in
2008 from 11.7% in 2007.
The increase of $113.1 million was due primarily to the
following:
|
|
|
| |
•
|
Increased sales volume of $33.2 million.
|
| |
| |
•
|
Higher prices for raw materials accounted for $38.8 million
of the increase. Raw material price increases for both CTO and
our secondary raw materials including pentaerythritol and
sulfuric acid were driven by the impact of higher oil and
natural gas prices, increased demand and capacity constraints.
|
| |
| |
•
|
Energy prices increased by $13.3 million from 2007 to 2008,
due to increasing natural gas prices and fuel oil prices.
|
| |
| |
•
|
Depreciation and amortization expense included within cost of
goods sold for 2008 was $24.6 million, a decrease of
$0.6 million, or 2.4%, as compared to $25.2 million in
2007.
|
| |
| |
•
|
Manufacturing fixed costs increased $14.4 million in 2008
as compared to 2007 due to increased tank car and storage costs
to support increased volumes, as well as higher maintenance and
operating costs, partially offset by $6.2 million of
savings primarily from restructuring initiatives in Europe.
|
73
|
|
|
| |
•
|
During 2008 we incurred $2.3 million of increased raw
material, delivery and other operating expenses associated with
a fire at our Oulu, Finland manufacturing facility.
|
| |
| |
•
|
The effect of foreign currency fluctuations provided an
unfavorable impact to cost of goods sold of $17.9 million
in 2008 as compared to 2007. This decrease was primarily due to
the strengthening of the Euro and Swedish Kronor against the
U.S. dollar.
|
We were successful in passing on raw material and energy price
increases to product prices, which is consistent with the
pricing increases referred to above in net sales.
Selling, General and Administrative
Expenses. Selling, general and administrative
expenses for 2008 were $91.9 million, a decrease of
$7.6 million, or 7.7%, as compared to $99.6 million
for 2007. The decrease was primarily due to lower transaction
and transition costs of $11.2 million compared to 2007
related primarily to the Acquisition. Partially offsetting those
declines were higher incentive compensation expense, consulting
expenses, bad debt reserve and other administrative expenses.
Foreign currency exchange resulted in an unfavorable impact of
$2.1 million primarily due to the strengthening of the
Swedish Kronor to the U.S. dollar. Selling, general and
administrative expenses as a percentage of net sales declined
2.4% to 9.2% in 2008, compared to 11.6% in 2007.
Unrealized Foreign Currency Exchange Losses
(Gains). Costs associated with unrealized
foreign currency exchange losses (gains) for 2008 were
$20.3 million. The increase in the unrealized loss was
primarily due to the translation of our Euro-denominated debt
into Swedish Kronor, as the Swedish Kronor weakened against the
Euro during the period. There were no significant unrealized
transaction gains or losses in 2007.
Restructuring and
Impairment. Restructuring and impairment
charges for 2008 were $15.5 million, compared to
$0.1 million in 2007. The restructuring charges were
primarily composed of personnel costs related to three
restructuring activities that occurred in 2008; including the
cost reduction initiatives at the Niort, France manufacturing
facility, the shutdown of the terpene resin production at our
Pensacola, Florida manufacturing facility, and the restructuring
of our dimer business and relocation of our polyamide production
from our Bedlington, U.K. manufacturing facility to Dover, Ohio.
These items are further described in Note 9 of our
consolidated financial statements appearing at the end of this
prospectus.
Interest Expense, Net. Interest
expense, net for 2008 was $29.5 million, an increase of
$0.9 million, or 3.0%, as compared to $28.7 million
for 2007. The year over year change is a result of several
factors, including an increase in debt issuance expenses, two
additional months of expense in 2008 since the debt under our
credit agreements was incurred at the end of February 2007 in
connection with the Acquisition, and a higher revolver balance
in 2008 due to working capital increases associated with higher
raw materials cost and higher demand for our products. These
amounts were offset by a reduction in the outstanding balance of
the term loans under our First Lien and Second Lien Credit
Agreements by approximately $30.1 million from
December 31, 2007, due to excess cash flow payments of
$14.0 million, Rhône Capital’s acquisition of
$27.2 million and subsequent extinguishment of
$9.5 million of our second lien debt and foreign exchange
impact.
Loss on Interest Rate Swaps, Net. Loss
on interest rate swaps, net for 2008 was $9.3 million, an
increase of $7.0 million, as compared to a loss of
$2.3 million for 2007. We had three interest rate swap
agreements that were put in place to hedge the interest rate
risk associated with the $375 million credit facilities
bearing variable interest rates. The increase is primarily due
to a $5.2 million non-cash
mark-to-market
loss in 2008 on these interest rate swaps due to declines in
LIBOR.
Other Income. Other income for 2008 was
$1.9 million as compared to $0 for 2007. During 2008,
Rhône Capital purchased $27.2 million face value of
our outstanding second lien debt. In accordance with our amended
credit agreement, 35% of the debt, or $9.5 million face
value was contributed to us and extinguished, resulting in a
gain of $1.9 million that was recognized as other income in
2008.
74
Income Tax Expense (Benefit). Income
tax benefit for 2008 was $4.3 million, a decrease of
$2.0 million, or 32.1%, as compared to $6.3 million
for 2007. The income tax benefit was primarily related to a
pre-tax loss of $36.4 million incurred from the period of
March 1, 2007 through December 31, 2007. The effective
tax rate was 13.7% for 2008, as compared to 20.9% for 2007 for
the net tax benefit for the full 12-month period. Our effective
tax rate for 2008 was less than our statutory Swedish income tax
rate of 28.0% primarily due to valuation allowances by our
international subsidiaries that could not recognize a tax
benefit for certain tax benefits that were considered unlikely.
Adjusted EBITDA. Adjusted EBITDA for
2008 was $92.7 million, an increase of $25.8 million,
or 38.6%, compared to $66.9 million for 2007. This increase
was due to the following:
|
|
|
| |
•
|
Increases in selling prices from 2007 to 2008 increased Adjusted
EBITDA by $86.6 million. Sales volumes positively impacted
Adjusted EBITDA by $5.6 million.
|
| |
| |
•
|
Higher raw material prices negatively impacted Adjusted EBITDA
by $38.8 million as compared to 2007 due to higher CTO and
secondary raw material prices.
|
| |
| |
•
|
Energy prices increased by $13.3 million from 2007 to 2008
as a result of increasing natural gas and fuel oil prices.
|
| |
| |
•
|
Manufacturing fixed costs increased $8.2 million compared
to 2007. The increase was primarily due to higher costs for
materials used in the manufacturing process, additional tank car
and storage spending, and maintenance costs of
$14.4 million, partially offset by $6.2 million of
costs savings from restructuring initiatives.
|
| |
| |
•
|
Increased costs associated with a fire at our Oulu, Finland
manufacturing facility of $2.3 million and higher selling,
general and administrative costs of $5.9 million after
taking into account adjustments to Adjusted EBITDA.
|
| |
| |
•
|
Favorable foreign exchange impact of $0.6 million.
|
The fire at our Oulu, Finland facility was the most significant
unusual item during 2008, negatively impacting Adjusted EBITDA
by $6.3 million including $2.3 million of additional
expenses and $4.0 million of lost sales margin.
Adjusted EBITDA margin for 2008 was 9.3% as compared to 7.8% for
2007. The increase of 1.5% from 2007 to 2008 was primarily due
to an increase in sales prices, which was partially offset by
higher raw materials and energy prices, and other costs.
Non-GAAP Financial
Measures
Adjusted
EBITDA and EBITDA
Adjusted EBITDA represents net income before interest, taxes,
depreciation, and amortization and then it is adjusted for
various items as defined in our credit agreements. Under the
terms of our credit agreements, we use Adjusted EBITDA to
calculate our maximum leverage ratio and our minimum interest
coverage ratio covenants. The maximum leverage ratio, which is
calculated as of the last day of each fiscal quarter, is the
ratio of (i) consolidated total debt to
(ii) consolidated Adjusted EBITDA for the twelve month
period then ended. The minimum interest coverage ratio, which is
calculated as of the last day of each fiscal quarter, is the
ratio of (i) consolidated Adjusted EBITDA for the twelve
month period then ended to (ii) consolidated interest
expense for the same twelve month period. For more information
about our covenants and our compliance with these covenants
under our credit agreements, see “Source of
Liquidity”, “Description of Our
Indebtedness — First Lien Credit Agreement —
Certain Covenants and Events of Default” and
“Description of Our Indebtedness — Second Lien
Credit Agreement”.
We present Adjusted EBITDA because it is used for reporting and
covenant calculation purposes pursuant to our credit agreements,
and by management to evaluate operating performance, and we
consider it an important supplemental measure of our
performance. We also believe it is frequently
75
used by securities analysts, investors and other interested
parties in the evaluation of companies in our industry. In
addition, our compensation plan bases incentive compensation
payments in part on our Adjusted EBITDA performance.
EBITDA represents net income before interest, taxes,
depreciation and amortization. We present EBITDA because it is
used by management to evaluate operating performance, and we
consider it an important supplemental measure of our
performance. We also believe it is used by other interested
parties in the evaluation of companies in our industry.
Adjusted EBITDA and EBITDA have limitations as analytical tools,
and you should not consider these in isolation, or as a
substitute for analysis of our results as reported under GAAP.
Some of these limitations are:
|
|
|
| |
•
|
Adjusted EBITDA and EBITDA do not reflect our cash expenditures,
or future requirements for capital expenditures or contractual
commitments;
|
| |
| |
•
|
Adjusted EBITDA and EBITDA do not reflect changes in, or cash
requirements for, our working capital needs;
|
| |
| |
•
|
Adjusted EBITDA and EBITDA do not reflect the significant
interest expense, or the cash requirements necessary to service
interest or principal payments, on our debts;
|
| |
| |
•
|
Although depreciation and amortization are non-cash charges, the
assets being depreciated and amortized will often have to be
replaced in the future, and Adjusted EBITDA and EBITDA do not
reflect any cash requirements for such replacements; and
|
| |
| |
•
|
Other companies in our industry may calculate Adjusted EBITDA
and EBITDA differently than we do, limiting its usefulness as a
comparative measure.
|
Because of these and other limitations, Adjusted EBITDA and
EBITDA should not be considered as a measure of discretionary
cash available to us to invest in the growth of our business. We
compensate for these limitations by relying primarily on our
GAAP results and using Adjusted EBITDA and EBITDA only as
supplemental information. See the Consolidated Statements of
Cash Flows included in our financial statements included
elsewhere in this prospectus.
76
A reconciliation of net income (loss), the most directly
comparable GAAP measure, to Adjusted EBITDA is presented below:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Combined
|
|
|
Successor
|
|
|
Predecessor
|
|
|
|
|
Three Months
|
|
|
Three Months
|
|
|
|
|
|
|
|
|
(Non-GAAP)
|
|
|
March 1, 2007
|
|
|
January 1, 2007
|
|
|
|
|
Ended
|
|
|
Ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
through
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
February 28,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
2007
|
|
|
2007(4)
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Net income (loss)
|
|
$
|
9,997
|
|
|
$
|
(2,357
|
)
|
|
$
|
12,098
|
|
|
$
|
(26,599
|
)
|
|
$
|
(23,605
|
)
|
|
$
|
(27,290
|
)
|
|
$
|
3,685
|
|
|
Interest (income) expense, net
|
|
|
3,870
|
|
|
|
4,560
|
|
|
|
16,546
|
|
|
|
29,523
|
|
|
|
28,657
|
|
|
|
28,775
|
|
|
|
(118
|
)
|
|
Income tax expense (benefit)
|
|
|
3,885
|
|
|
|
(836
|
)
|
|
|
3,831
|
|
|
|
(4,277
|
)
|
|
|
(6,299
|
)
|
|
|
(8,913
|
)
|
|
|
2,614
|
|
|
Depreciation and amortization
|
|
|
9,043
|
|
|
|
9,437
|
|
|
|
40,615
|
|
|
|
34,210
|
|
|
|
33,235
|
|
|
|
28,813
|
|
|
|
4,422
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
EBITDA
|
|
|
26,795
|
|
|
|
10,804
|
|
|
|
73,090
|
|
|
|
32,857
|
|
|
|
31,988
|
|
|
|
21,385
|
|
|
|
10,603
|
|
|
Unrealized foreign currency exchange (gains) losses
|
|
|
(4,836
|
)
|
|
|
367
|
|
|
|
(9,347
|
)
|
|
|
20,304
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Restructuring and impairment
|
|
|
2,047
|
|
|
|
1,035
|
|
|
|
26,395
|
|
|
|
15,513
|
|
|
|
114
|
|
|
|
114
|
|
|
|
—
|
|
|
Loss on interest rate swaps
|
|
|
709
|
|
|
|
600
|
|
|
|
2,541
|
|
|
|
9,311
|
|
|
|
2,275
|
|
|
|
2,275
|
|
|
|
—
|
|
|
Equity in earnings of affiliate
|
|
|
(4
|
)
|
|
|
(150
|
)
|
|
|
(613
|
)
|
|
|
(380
|
)
|
|
|
(273
|
)
|
|
|
(189
|
)
|
|
|
(84
|
)
|
|
Transaction costs(1)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,316
|
|
|
|
10,271
|
|
|
|
10,271
|
|
|
|
—
|
|
|
Management fees(2)
|
|
|
495
|
|
|
|
351
|
|
|
|
1,538
|
|
|
|
1,990
|
|
|
|
1,295
|
|
|
|
1,295
|
|
|
|
—
|
|
|
Transition costs(3)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
2,984
|
|
|
|
6,667
|
|
|
|
6,575
|
|
|
|
92
|
|
|
Gain on Abieta acquisition
|
|
|
—
|
|
|
|
(2,151
|
)
|
|
|
(2,151
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Gain on settlement with International Paper
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,316
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Selling, general and administrative severance
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
3,121
|
|
|
|
2,235
|
|
|
|
2,235
|
|
|
|
—
|
|
|
Third-party advisor fees
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,573
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Consulting services
|
|
|
2,135
|
|
|
|
—
|
|
|
|
—
|
|
|
|
3,794
|
|
|
|
7,616
|
|
|
|
7,616
|
|
|
|
—
|
|
|
Gain on debt extinguishment
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,901
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other items
|
|
|
37
|
|
|
|
187
|
|
|
|
3,722
|
|
|
|
2,241
|
|
|
|
4,710
|
|
|
|
3,297
|
|
|
|
1,413
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Adjusted EBITDA
|
|
$
|
27,378
|
|
|
$
|
11,043
|
|
|
$
|
93,859
|
|
|
$
|
92,723
|
|
|
$
|
66,898
|
|
|
$
|
54,874
|
|
|
$
|
12,024
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1) |
|
Transaction costs were related to the Acquisition. These costs
included legal, tax, IT, professional fees, and Rhône
Capital transaction fees. |
| |
|
(2) |
|
Management fees are paid to Rhône Group L.L.C., an
affiliate of Rhône Capital for certain monitoring and other
management services and assistance, including reimbursement of
its out-of-pocket expenses. |
| |
|
(3) |
|
Transition costs included fees paid to International Paper in
connection with the Acquisition under the transition services
agreement, IT consulting fees, costs for infrastructure
build-out and the cost of a carve-out audit, among other items. |
| |
|
(4) |
|
Adjusted EBITDA presented in the predecessor period was
calculated on the same basis as Adjusted EBITDA presented in the
successor periods pursuant to our credit agreements. |
Free Cash
Flow
Free cash flow is defined by us as net cash provided by (used
in) operating activities, less purchases of property, plant and
equipment, software spending, proceeds from disposals of
property, plant and equipment, and other items, all as disclosed
in our consolidated statements of cash flows or the consolidated
statements of operations.
77
We have included data with respect to free cash flow because our
management considers free cash flow to be a useful, supplemental
indicator of our liquidity. When measured over time, free cash
flow provides supplemental information to investors concerning
our operating results and our ability to generate cash flows to
satisfy mandatory debt service requirements and make other
non-discretionary expenditures.
Our management believes that consideration of free cash flow
should be supplemental, however, free cash flow has limitations
as an analytical financial measure, including:
|
|
|
| |
•
|
Free cash flow is subject to variability on a quarterly basis as
a result of the timing of payments made or received related to
accounts receivable, accounts payable and other current
operating assets and liabilities.
|
| |
| |
•
|
Free cash flow may be calculated in a different manner by other
companies in our industry, which limits its usefulness as a
comparative measure.
|
Our management compensates for these limitations by relying
primarily on our results under GAAP to evaluate our operating
results and by considering independently the economic effects of
the foregoing items that are not reflected in free cash flow. As
a result of these limitations, free cash flow should not be
considered a substitute for other measures of liquidity reported
in accordance with GAAP, including net cash provided by (used
in) operating activities, net cash provided by (used in)
investing activities, net cash provided by (used in) financing
activities or change in cash and cash equivalents.
The following table sets forth a reconciliation of net cash
provided by (used in) operating activities to free cash flow,
the most directly comparable GAAP measure:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Combined
|
|
|
Successor
|
|
|
Predecessor
|
|
|
|
|
Three Months
|
|
|
Three Months
|
|
|
|
|
|
|
|
|
(Non-GAAP)
|
|
|
March 1,
|
|
|
January 1,
|
|
|
|
|
Ended
|
|
|
Ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
2007 through
|
|
|
2007 through
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
February 28,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
2007
|
|
|
2007
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Net cash provided by (used in) by operating activities
|
|
$
|
2,507
|
|
|
$
|
11,100
|
|
|
$
|
117,325
|
|
|
$
|
20,841
|
|
|
$
|
32,934
|
|
|
$
|
45,022
|
|
|
$
|
(12,088
|
)
|
|
Less:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Purchases of property, plant and equipment
|
|
|
(4,866
|
)
|
|
|
(6,677
|
)
|
|
|
(22,993
|
)
|
|
|
(34,719
|
)
|
|
|
(22,846
|
)
|
|
|
(18,248
|
)
|
|
|
(4,598
|
)
|
|
Software spending
|
|
|
(3,761
|
)
|
|
|
(3,268
|
)
|
|
|
(13,404
|
)
|
|
|
(142
|
)
|
|
|
(1,642
|
)
|
|
|
(1,642
|
)
|
|
|
—
|
|
|
Proceeds from disposals of property, plant and equipment
|
|
|
—
|
|
|
|
690
|
|
|
|
875
|
|
|
|
212
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other
|
|
|
696
|
|
|
|
—
|
|
|
|
—
|
|
|
|
356
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Free cash flow
|
|
$
|
(5,424
|
)
|
|
$
|
1,845
|
|
|
$
|
81,803
|
|
|
$
|
(13,452
|
)
|
|
$
|
8,446
|
|
|
$
|
25,132
|
|
|
$
|
(16,686
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash used in investing activities
|
|
|
(7,931
|
)
|
|
|
(18,027
|
)
|
|
|
(44,294
|
)
|
|
|
(34,293
|
)
|
|
|
(501,929
|
)
|
|
|
(497,331
|
)
|
|
|
(4,598
|
)
|
|
Net cash provided by (used in) financing activities
|
|
|
2,202
|
|
|
|
(1,220
|
)
|
|
|
(63,771
|
)
|
|
|
12,768
|
|
|
|
498,843
|
|
|
|
488,547
|
|
|
|
10,296
|
|
Free cash flow for the three months ended March 31, 2010
was a deficit of $5.4 million, a decrease of
$7.3 million as compared to $1.8 million for the three
months ended March 31, 2009. Operating cash flow decreased
$8.6 million due to an increase in working capital at the
end of our first quarter relating to the timing of sales earned
during the quarter, partially offset by improved profitability.
Free cash flow for 2009 was $81.8 million, an increase of
$95.3 million as compared to a deficit of
$13.5 million for 2008. Operating cash flow increased
$96.5 million due to improved profitability and reductions
in working capital of $48.5 million. Capital spending
declined $11.7 million as we deferred certain
non-critical
maintenance projects to 2010 to preserve cash, while software
spending related to the global implementation of our ERP system
increased $13.3 million.
78
During 2009, we instituted a working capital management program
focused on improving our working capital as we managed the
impact on our operations of the global economic slowdown. This
program resulted in an 18% decrease in accounts receivable and a
34% decrease in inventory, which ultimately resulted in a
decrease in working capital of $48.5 million. We instituted
long-term working capital improvements to reduce our investment
in working capital measured as a percentage of sales. We are
targeting for working capital as a percent of sales to continue
to decline supported by ongoing cost saving initiatives and the
global implementation of our ERP system.
At March 31, 2010, we experienced an increase in accounts
receivable of $26.3 million, or 26.4%, compared to
December 31, 2009. This is a direct result of the timing of
sales earned during the first quarter of 2010 versus the last
quarter of 2009. In particular, we experienced an increase of
$16.2 million, or 28.2%, in sales in March 2010 compared to
December 2009. Due to higher sales in March 2010, we ended the
month with a higher accounts receivable balance. We are not
aware of any unusual collection issues associated with this
increase in accounts receivable, which is supported by us
maintaining the same percentage of current receivables and
slightly decreasing our days sales outstanding from
December 31, 2009. Accounts payable increased by
$15.2 million, or 18.4%, compared to December 31,
2009, primarily due to increased inventory purchases driven by
the increase in sales. However, as a result of a strong focus on
working capital management, our inventory declined by
$2.7 million, or 3.0%, compared to December 31, 2009.
Free cash flow for 2008 was a deficit of $13.5 million, a
decrease of $21.9 million as compared to free cash flows of
$8.4 million for the same period in the prior year. The
decrease was primarily due to a decrease in operating cash flows
of $12.1 million due to an increase in working capital of
$7.2 million. Capital spending increased $11.9 million
primarily due to capital expenditures at our Savannah, Georgia
facility associated with our relocation of capacity from our
Valdosta, Georgia facility.
Liquidity and
Capital Resources
Summary
At March 31, 2010, we had $43.1 million of cash and
cash equivalents. We ended 2009 with $47.0 million of cash and
cash equivalents, compared to $34.0 million at the end of 2008.
The increase in cash and cash equivalents was primarily due to
lowering our working capital by $48.6 million and
increasing net income by $56.2 million with depreciation,
amortization and impairment excluded. This increase in cash
enabled us to invest $36.4 million in property, plant and
equipment and our new ERP system. We also repaid a portion of
our long-term debt and repaid the amounts outstanding on the
revolving credit facility under out First Lien Credit Agreement
net of proceeds of $58.2 million, which will reduce future
interest expense.
Our debt outstanding at March 31, 2010 under our credit
agreements was $327.4 million. As a result of the global
economic slowdown and market conditions, the cost and
availability of new credit has been and may continue to be
adversely affected by illiquid credit markets and wider credit
spreads. Concern about the stability of the markets generally
and the strength of counterparties specifically has led many
lenders and institutional investors to reduce and, in some
cases, cease to provide funding to borrowers. Continued
difficulties in the markets may adversely affect our liquidity
and financial condition, the liquidity and financial condition
of our customers, our ability to replace maturing liabilities in
a timely fashion, and otherwise constrain access to the capital
markets to meet our liquidity needs, thus resulting in adverse
effects on our financial condition and results of operations.
This credit contraction has not impacted our access to
borrowings.
Working capital, the excess of current assets over current
liabilities excluding cash and cash equivalents and the current
portion of long-term debt, was $92.8 million at
March 31, 2010, an increase of
79
$13.8 million from $79.0 million at December 31,
2009. This increase in working capital was primarily related to
the timing of sales earned during the three months ended
March 31, 2010 compared to the three months ended
December 31, 2009, when sales increased 28.2% in March 2010
compared to December 2009, and we increased our purchases of
inventory. These factors contributed to higher accounts
receivable and accounts payable balances during this period.
Working capital was $79.0 million at the end of 2009, down from
$127.6 million at the end of 2008. Working capital as a
percentage of sales was 10.3%, 12.7% and 14.1% for 2009, 2008
and 2007, respectively.
Our principal sources of liquidity are cash flow from operations
and borrowings under our credit agreements. Going forward, there
can be no assurance that our business will generate sufficient
cash flow from operations or that future borrowings will be
available to fund liquidity needs in an amount sufficient to
enable us to service our indebtedness or other obligations.
Our principal uses of cash are debt service payments as
described below, capital expenditures, and working capital
requirements.
Cash
Flow
The following table summarizes our cash provided by (used in)
from operating, investing and financing activities for the years
ended December 31, 2009, 2008 and 2007:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Predecessor
|
|
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Successor
|
|
|
Combined
|
|
|
March 1,
|
|
|
January 1,
|
|
|
|
|
Three Months
|
|
|
Three Months
|
|
|
|
|
|
|
|
|
(Non-GAAP)
|
|
|
2007
|
|
|
2007
|
|
|
|
|
Ended
|
|
|
Ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
through
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
February 28,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
2007
|
|
|
2007
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Operating
|
|
$
|
2,507
|
|
|
$
|
11,100
|
|
|
$
|
117,325
|
|
|
$
|
20,841
|
|
|
$
|
32,934
|
|
|
$
|
45,022
|
|
|
$
|
(12,088
|
)
|
|
Investing
|
|
|
(7,931
|
)
|
|
|
(18,027
|
)
|
|
|
(44,294
|
)
|
|
|
(34,293
|
)
|
|
|
(501,929
|
)
|
|
|
(497,331
|
)
|
|
|
(4,598
|
)
|
|
Financing
|
|
|
2,202
|
|
|
|
(1,220
|
)
|
|
|
(63,771
|
)
|
|
|
12,768
|
|
|
|
498,843
|
|
|
|
488,547
|
|
|
|
10,296
|
|
|
Effect of foreign exchange
|
|
|
(744
|
)
|
|
|
(4,278
|
)
|
|
|
3,715
|
|
|
|
(3,354
|
)
|
|
|
1,783
|
|
|
|
1,848
|
|
|
|
(65
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Increase (decrease) in cash and cash equivalents
|
|
$
|
(3,966
|
)
|
|
$
|
(12,425
|
)
|
|
$
|
12,975
|
|
|
$
|
(4,038
|
)
|
|
$
|
31,631
|
|
|
$
|
38,086
|
|
|
$
|
(6,455
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating
Activities
Operating cash flows in the three months ended March 31,
2010 were $2.5 million, a decrease of $8.6 million as
compared to $11.2 million in the three months ended
March 31, 2009. This change in operating cash flows was
primarily driven by the following factors:
|
|
|
| |
•
|
Net income increased $12.4 million for reasons previously
described.
|
| |
| |
•
|
Non-cash items affecting net income increased $1.7 million
compared to the three months ended March 31, 2009.
|
| |
| |
•
|
Changes in working capital reduced cash provided by operating
activities by $15.7 million in the three months ended
March 31, 2010, which is $22.7 million lower than the
three months ended March 31, 2009. This reduction of
operating cash flows from working capital was primarily driven
by the following:
|
|
|
|
| |
•
|
Accounts receivable increased $29.4 million, primarily
related to the timing of sales which occurred near the quarter
end.
|
| |
| |
•
|
The increase in accounts receivable was partially offset by an
increase in accounts payable of $20.3 million as a result
of an increase in purchases to support the increase in sales.
|
Operating cash flows in 2009 were $117.3 million, an
increase of $96.5 million as compared to $20.8 million
in 2008. This change in operating cash flows was primarily
driven by the following factors:
|
|
|
| |
•
|
Net income increased $38.7 million for reasons previously
described.
|
| |
| |
•
|
Non-cash items affecting net income declined $16.3 million
compared to 2008.
|
80
|
|
|
| |
•
|
During 2009, we instituted a working capital management program
focused on improving our working capital as we managed the
impact on our operations of the global economic slowdown.
|
| |
| |
•
|
Working capital contributed $58.3 million to operating cash
flows in 2009, $74.0 million higher than 2008. This
contribution of operating cash flows from working capital was
primarily driven by the following:
|
|
|
|
| |
•
|
Accounts receivable declined $25.9 million, of which
$10.5 million was due to a five day decline in days sales
outstanding and the remainder due to lower sales.
|
| |
| |
•
|
Inventory declined $57.4 million of which
$31.2 million was associated with reductions in inventory
on hand as the result of our on-going working capital
improvement initiatives and in response to lower demand. In
addition, $26.1 million of the decline was associated with
lower raw material prices.
|
| |
| |
•
|
The declines in accounts receivable and inventories were
partially offset by a decrease in accounts payable of
$16.8 million as a result of a decline in purchases and
lower purchase prices.
|
Operating cash flows for 2008 were $20.8 million, a
decrease of $12.1 million as compared to $32.9 million
for 2007. This change in operating cash flows was primarily
driven by the following factors:
|
|
|
| |
•
|
Net income declined $3.0 million for reasons previously
described.
|
| |
| |
•
|
Non-cash items affecting net income increased $45.1 million
compared to 2007.
|
| |
| |
•
|
Working capital consumed $15.7 million of operating cash in
2008, a decrease of $54.2 million compared to 2007. The
working capital consumption of operating cash flows related to
the following:
|
|
|
|
| |
•
|
Accounts receivable increased $9.6 million associated with
higher sales and a six day increase in days sales
outstanding.
|
| |
| |
•
|
Inventory increased $6.2 million due primarily to volume,
while accounts payable declined $3.9 million.
|
Investing
Activities
Net cash used in investing activities totaled $8.6 million
for the three months ended March 31, 2010, compared to
$18.0 million for the three months ended March 31,
2009. The decrease of $9.4 million primarily relates to the
acquisition of Abieta in the three months ended March 31,
2009, which impacted cash used in investing activities by
$8.8 million net of cash acquired and assumed debt of
$6.0 million, which excludes factored receivables of
approximately $1.9 million.
Net cash used in investing activities totaled $44.3 million
for 2009 compared to $34.3 million for 2008. The increase
of $10.0 million was driven by lower capital maintenance
spending, mostly offset by spending on the implementation costs
of our ERP system in the amount of $13.4 million. In
addition, during 2009 we completed the acquisition of Abieta for
$8.8 million net of cash acquired and assumed debt of
$6.0 million, which excludes factored receivables of
approximately $1.9 million.
Net cash used in investing activities totaled $34.3 million
for 2008 compared to $501.9 million for 2007, which included
$477.4 million cost of the Acquisition. Capital spending
for 2008 was $34.7 million or $11.9 million higher
than 2007 primarily due to $8.7 million of spending on
expanded capacity at our Savannah, Georgia manufacturing
facility, which was associated with the realignment of our
Valdosta, Georgia manufacturing facility.
Financing
Activities
Net cash provided by financing activities was $2.2 million
in the three months ended March 31, 2010, compared to
$1.2 million used in the three months ended March 31,
2009. This increase is primarily the
81
result of a cash payment of $3.0 million following the
assignment and assumption of the management MIV loans. As
described under “Certain Relationships and Related Party
Transactions — Management Loans”, we provided
loans to certain members of our management to assist them in
financing investments in the MIVs. The MIVs, Rhône Capital
and International Paper all own a percentage of AZ Chem
Investments Partners LP, our parent entity. On February 19,
2010, we entered into an Assignment and Assumption Agreement
with AZ Chem Investments Partners LP pursuant to which we
assigned, and AZ Chem Investments Partners LP assumed, all
outstanding loans to members of our management team and board of
directors in exchange of a cash payment of $3.0 million.
After the assignment and assumption, we have no outstanding
loans related to the MIVs and there will be no future loans by
us related to the MIVs.
Net cash used by financing activities was $63.8 million in
2009 as compared to $12.8 million in 2008. This increase is
primarily the result of the use of cash to reduce our long-term
debt balance during 2009, including a $20.1 million
reduction in the term loan under our First Lien Credit Agreement
and a $29.3 million reduction in the revolving credit
facility under our First Lien Credit Agreement. During 2009 we
also assumed debt of $6.0 million in our acquisition of
Abieta, which we were able to reduce to $0.7 million by
December 31, 2009.
Net cash provided by financing activities in 2008 was
$12.8 million compared to $498.8 million in 2007 which
included $130.0 million issuance of common stock,
$375.0 million of proceeds from issuance of long-term debt
gross of $14.3 million of debt issuance costs associated
with the Acquisition by Rhône Capital. During 2008, we drew
on the revolving credit facility under our First Lien Credit
Agreement to fund working capital needs, in particular to
increase our cash reserves in response to the global credit
crisis in the fourth quarter. During 2008, we permanently
reduced our outstanding debt balance under our First Lien Credit
Agreement by $16.5 million, including a payment of 50% of
the excess cash flow from the prior year as required by our
First Lien Credit Agreement, compared to $1.9 million in
2007 of scheduled debt payments.
Source of
Liquidity
We anticipate that operating cash flow, together with available
borrowings under the revolving credit facility under our First
Lien Credit Agreement, will be sufficient to meet our working
capital requirements, fund strategic growth initiatives and
capital expenditures, and service our debt obligations for at
least the next 12 months. We have available to us
$60 million under the revolving credit facility under our
First Lien Credit Agreement, on which we had no borrowings at
March 31, 2010. Under the terms of our credit agreements we
are subject to certain financial covenants, including
maintenance of a minimum interest coverage ratio, a maximum
leverage ratio and a maximum consolidated capital expenditure
amount. We use consolidated Adjusted EBITDA to calculate these
ratios. We are required to maintain a fiscal quarter end
interest coverage ratio and a fiscal quarter end leverage ratio
as illustrated in the table below. The maximum consolidated
capital expenditure amount, as defined in the First Lien Credit
Agreement, permitted for 2010 is $32 million and
$35 million in 2011 and thereafter; our credit agreements
allow a 50% carryover of any unspent amounts at year-end to be
applied to the following year’s maximum consolidated
capital expenditure. As of March 31, 2010, our interest
coverage ratio was 4.51 and our leverage ratio was 2.78, which
were in compliance with the applicable financial ratios and the
other covenants contained in our credit agreements. The ratio
limits for future periods are as follows:
| |
|
|
|
|
|
|
|
|
|
|
|
Interest
|
|
|
|
|
|
Coverage
|
|
Leverage
|
|
Term
|
|
Ratio(1)
|
|
Ratio(2)
|
|
|
|
March 31, 2010 — March 30, 2011
|
|
|
2.25
|
|
|
|
4.50
|
|
|
March 31, 2011 and thereafter
|
|
|
2.50
|
|
|
|
4.00
|
|
|
|
|
|
(1) |
|
Our interest coverage ratio must exceed the amounts illustrated
above for the periods shown. |
| |
|
(2) |
|
Our leverage ratio must not exceed the amounts illustrated above
for the periods shown. |
82
The maintenance of these financial ratios is based on our level
of profitability relative to our level of indebtedness. If the
global economic environment worsens or other factors arise which
negatively impact our profitability, we may not be able to
satisfy our covenants. If we are unable to satisfy these
covenants in the future, we would need to seek an amendment or
waiver of these financial covenants. The respective lenders
under our credit agreements may not consent to any amendment or
waiver requests that we may make in the future, and, if they do
consent, they may not do so on terms which are favorable to us.
In the event that we were unable to obtain any such waiver or
amendment and we were not able to refinance or repay our debt
instruments, our inability to meet the financial covenants or
other provisions of our credit agreements would constitute an
event of default, which would permit the lenders to accelerate
the amounts due under our credit agreements.
From time to time, we evaluate options with respect to our
overall debt structure, including the possibility of refinancing
our existing credit agreements at least one year in advance of
their respective maturities. The principal amounts of our term
loans under our First Lien Credit Agreement amortize in
quarterly installments of
1/4
of 1% of the outstanding principal amount and the remaining
outstanding principal amount matures on February 28, 2013.
The term loans under our Second Lien Credit Agreement mature on
February 28, 2014. We may also selectively pursue
acquisitions of, and joint ventures with, related businesses as
one element of our growth strategy. There can be no assurance
that we will decide to pursue acquisitions or joint ventures or
whether we will be successful in completing any such
transaction. These transactions may require us to assume or
incur additional debt financing, resulting in additional
leverage. We plan on financing capital expenditures through cash
flows from operations.
For more information about our outstanding indebtedness and our
available borrowings under our credit agreements see
“Description of Our Indebtedness”.
Capital
Expenditures
Our capital expenditure program is a key component of our
long-term strategy. This program includes, among other things,
maintenance, performance improvement and cost reduction
initiatives and environmental, health and safety spending, but
excludes our investment in our new ERP system.
We have historically split investment between maintenance and
expansion-type capital expenditure activities. Maintenance
includes planned and unplanned maintenance and environmental,
health and safety spending and expansion includes cost
reduction, performance improvement and capacity enhancement
spending. In accordance with our accounting policy, expenditures
for major repairs and improvements are capitalized because they
extend the useful lives of the assets, whereas normal repairs
and maintenance are expensed as incurred.
The following table presents maintenance and expansion
expenditures incurred.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Successor
|
|
|
Combined
|
|
|
Successor
|
|
|
Predecessor
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 1,
|
|
|
January 1,
|
|
|
|
|
|
|
|
|
|
|
(Non-GAAP)
|
|
|
2007
|
|
|
2007
|
|
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
through
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
February 28,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
2007
|
|
|
2007
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
Major maintenance
|
|
$
|
10,671
|
|
|
$
|
21,469
|
|
|
$
|
21,333
|
|
|
$
|
17,065
|
|
|
$
|
4,268
|
|
|
Expansion
|
|
|
12,322
|
|
|
|
13,250
|
|
|
|
1,513
|
|
|
|
1,183
|
|
|
|
330
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total capital expenditures
|
|
$
|
22,993
|
|
|
$
|
34,719
|
|
|
$
|
22,846
|
|
|
$
|
18,248
|
|
|
$
|
4,598
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
During 2009, we closely managed our capital spending in response
to the weak economic environment. Despite this environment, we
continued to invest in the capacity expansion at our Savannah,
Georgia manufacturing facility and spent $10.5 million and
$8.7 million in 2009 and 2008, respectively. This capacity
expansion was part of our realignment initiative, and the
partial closure of our Valdosta, Georgia manufacturing facility.
We expect our capitalized maintenance expenditures to
83
increase in 2010 from reduced levels in 2009, to approximately
$20 million, then decline thereafter to a more normalized
annualized trend line of approximately $15 million,
consistent with fewer manufacturing facility sites than we have
had in recent years. We estimate our 2010 expansion capital to
be approximately $2 million. During the three months ended
March 31, 2010, we incurred $3.9 million and
$0.5 million of major maintenance and expansion
expenditures, respectively.
Contractual
Obligations and Commercial Commitments
Our principal outstanding contractual obligations relate to the
debt under our credit agreements and related interest expense,
and operating leases on certain facilities. The following table
sets forth our long-term contractual cash obligations as of
December 31, 2009:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Years ending December 31,
|
|
|
|
|
Total(1)
|
|
|
2010
|
|
|
2011
|
|
|
2012
|
|
|
2013
|
|
|
2014
|
|
|
Thereafter
|
|
|
|
|
(dollars in thousands)
|
|
|
|
|
First Lien Credit Agreement
|
|
$
|
218,543
|
|
|
$
|
3,360
|
|
|
$
|
2,460
|
|
|
$
|
2,460
|
|
|
$
|
210,263
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
Second Lien Credit Agreement
|
|
|
115,498
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
115,498
|
|
|
|
—
|
|
|
Other loans(2)
|
|
|
717
|
|
|
|
575
|
|
|
|
142
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Interest expense(3)
|
|
|
58,875
|
|
|
|
15,356
|
|
|
|
16,419
|
|
|
|
15,095
|
|
|
|
9,950
|
|
|
|
2,055
|
|
|
|
—
|
|
|
Payments on interest rate swaps(4)
|
|
|
2,840
|
|
|
|
2,983
|
|
|
|
(73
|
)
|
|
|
(70
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Operating leases(5)
|
|
|
33,562
|
|
|
|
9,842
|
|
|
|
8,708
|
|
|
|
4,046
|
|
|
|
3,743
|
|
|
|
3,691
|
|
|
|
3,532
|
|
|
Capital lease(6)
|
|
|
4,095
|
|
|
|
299
|
|
|
|
324
|
|
|
|
341
|
|
|
|
360
|
|
|
|
379
|
|
|
|
2,392
|
|
|
Post retirement benefits(7)
|
|
|
4,117
|
|
|
|
4,117
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Natural gas forward contracts(8)
|
|
|
4,378
|
|
|
|
4,378
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total contractual obligations
|
|
$
|
442,625
|
|
|
$
|
40,910
|
|
|
$
|
27,980
|
|
|
$
|
21,872
|
|
|
$
|
224,316
|
|
|
$
|
121,623
|
|
|
$
|
5,924
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1) |
|
The $60 million revolving credit facility under our First
Lien Credit Agreement was not included in the table above as
there was no outstanding balance at December 31, 2009. |
| |
|
(2) |
|
Other loans primarily relate to the fixed rate debt assumed in
our acquisition of Abieta in 2009. |
| |
|
(3) |
|
The LIBOR and Euribor rates assumed for future interest payments
on our floating rate debt resulted in average interest rates
including the spread between 4.70% and 4.20%, respectively, for
all future periods presented. Amounts presented are
representative of estimated cash payments and do not include
amortization of debt issuance costs or other accruals. |
| |
|
(4) |
|
Payments on interest rate swaps are based on mark-to-market
calculations at year-end, based on the expected rates over the
term of the swap agreements. |
| |
|
(5) |
|
Operating leases include various operating leases and other rent
payments. |
| |
|
(6) |
|
Capital leases primarily relate to our office building lease for
our Almere, The Netherlands location that includes executive,
administrative and research and development functions. |
| |
|
(7) |
|
Liabilities for post retirement benefits relate to our foreign
and domestic defined benefit and retirement pension plans for
the following plan year. As of December 31, 2009, our
recorded liability for the U.S. and Non-U.S. pension, early
retirement and jubilee plans was $15.5 million. The table
above includes contributions we expect to make to the plans in
2010. Contributions for years beyond 2010 were excluded from the
table above since we cannot reasonably make estimates of the
timing of future payments. |
| |
|
(8) |
|
Amounts due in 2010 are based on fixed volume and average fixed
price of natural gas forward contracts outstanding at
December 31, 2009. |
84
Major Maintenance and Expansion. At
December 31, 2009, we had open purchase orders totaling
$2.6 million for major maintenance and expansion
expenditures. We intend to fulfill these commitments with cash
generated from our operations. The commitments primarily relate
to the upgrade of fire safety equipment for the Panama City,
Florida manufacturing facility and the replacement of process
equipment at the Dover, Ohio and Savannah, Georgia manufacturing
facilities.
Uncertainty in Income Taxes. As of
December 31, 2009, net unrecognized tax benefits totalled
$0.4 million, which are excluded from the table above since
we cannot make reasonably reliable estimates of the timing of
future payments.
Raw Materials. We have long-term supply
contracts with International Paper pursuant to which they agreed
to sell to us, and we agreed to purchase from them, all of the
CTO and CST produced at their existing U.S. paper mills. We
also have the option to purchase all of the CTO and CST produced
at International Paper’s future paper mills worldwide.
International Paper is not required to produce any minimum
quantities of CTO or CST, and the amount of CTO and CST that we
are required to purchase under these agreements varies depending
upon the amount of CTO and CST actually produced by
International Paper, which is dependent on demand for its
products. However, subject to the agreement of International
Paper, we may purchase less than 100% of the output of each
mill, provided that in the event that we purchase 75% or less of
the output from a particular mill for four consecutive quarters,
we will lose the right to purchase the CTO or CST produced by
that mill. The contracts provide for the purchase price for CTO
to fluctuate, as it is determined by reference to energy prices,
while the purchase price for CST is fixed, subject to adjustment
every three years based on changes in market price. We purchased
approximately $49.8 million, $59.0 million and
$52.0 million of CTO and CST from International Paper for
the years ended December 31, 2009 and 2008 and the ten
months ended December 31, 2007, respectively.
We receive monthly wood processing data from International
Paper’s mills and compute the expected quantity of CTO and
CST that International Paper will provide. We issue purchase
orders and record inventory based upon the weight of CTO and CST
measured by the paper mills. Actual quantities shipped
approximate quantities received and weighed by our manufacturing
facilities.
These contracts provided us with approximately 20% and 27% of
our global CTO and CST purchases for the three months ended
March 31, 2010 and the year ended December 31, 2009,
respectively. CTO and CST accounted for approximately 68% and
64% of our raw material purchases for the three months ended
March 31, 2010 and the year ended December 31, 2009,
respectively. These contracts are terminable by either party on
February 28, 2027 and each anniversary thereafter, subject
to a five year notice requirement.
To ensure adequate supplies of CTO and CST, we also maintain
long-standing relationships with other major suppliers in the
United States and Europe that enable us to manage our CTO and
CST needs.
We believe we are well positioned to acquire the CTO and CST
required for our operations.
Pension Plans. We sponsor various
pension plans worldwide that are underfunded and require
significant cash payments. For example, in 2009, we contributed
$4.5 million to our pension plans and, in 2008, we
contributed $2.7 million to our pension plans. We are
expected to contribute at least $4.1 million to our pension
plans in 2010. We may also opt to make additional voluntary
contributions to various pension plans worldwide in 2010.
Additionally, if the performance of the assets in our pension
plans does not meet our expectations, or if other actuarial
assumptions are modified, our contributions for those years
could be even higher than we expect. For example, the combined
asset value of funded pension plans worldwide was
$47.6 million as of December 31, 2009. We expect to
earn a 5.9% investment return on our pension assets. In the
event actual investment returns are 1% lower than expected for
one year, we expect our long-term cash requirements to increase
by $0.5 million. As of December 31, 2009, our
worldwide pension plans were underfunded by $14.8 million
(based on the actuarial assumptions consistent with GAAP).
85
Other
Contingencies
We had no material operating expenditures for environmental
fines, penalties, or government imposed remedial or corrective
actions during the years ended December 31, 2009, 2008 or
2007.
We are party to various lawsuits, claims and contingent
liabilities arising from the conduct of our business; however,
these matters are not expected to have a material adverse effect
on our consolidated results of operations, cash flows or
financial position.
Off-Balance Sheet
Arrangements
We use natural gas in our production processes and, as a result,
we are exposed to commodity price risk. To offset the impact of
price changes, we enter into forward purchase contracts that
require our contract counterparties to deliver natural gas to us
at the contract maturity dates. As a result of the requirements
of counterparties to physically deliver the gas to us at the
settlement date, these contracts do not qualify as derivatives
for financial reporting purposes.
These contracts help us to partially fix the price of natural
gas that we use in production which offsets adverse changes in
natural gas price movements. The remaining terms of our
contracts range from one month to approximately twelve months.
We are exposed to the risk that our forward purchase contract
counterparties will not deliver the contractual amounts of
natural gas due under these arrangements. We monitor this credit
risk, in part, by tracking the credit standings of contract
counterparties and we reduce the risk of counterparty credit
risk by using multiple contract parties.
We make monthly accruals based on estimated volumes of gas
consumed during the month and a blended average rate that takes
into account the contract price and current market prices for
volumes that are not fixed as the gas is consumed. Total
purchases of natural gas, including amounts purchased under
fixed-price forward contracts, for the three months ended
March 31, 2010 and the years ended December 31, 2009
and 2008 were $5.1 million, $23.1 million and
$32.3 million, respectively, and our expenses were
$5.7 million, $24.0 million and $36.5 million,
respectively.
Natural gas supplies are volatile and, historically, the amount
of exploration, drilling and production changes whenever
significant price changes occur. As a result of the recent
global economic decline, worldwide natural gas usage has
declined. Moreover, during 2009 additional supplies have been
discovered and better production techniques, such as shale
extraction, principally occurring in the United States, have
improved yields and led to further supply increases. As a
result, natural gas prices during 2009 declined approximately
22%.
The availability of natural gas is critical to our production
capacity and any supply disruptions would adversely impact our
ability to produce our products and meet our customers’
needs. In the event of a supply disruption or severe price
increase of natural gas, we may substitute liquid petroleum, or
internally produced renewable energy to satisfy a portion of our
needs.
Quantitative and
Qualitative Disclosures about Market Risk
Interest Rate
Risk
We are exposed to certain market risks as part of our ongoing
business operations. Primary exposures include changes in
interest rates, as borrowings under our credit agreements will
bear interest at floating rates based on LIBOR, Euribor or the
base rate, plus an applicable borrowing margin. For
floating-rate debt, interest rate changes generally impact our
earnings and cash flows, assuming other factors are held
constant.
At March 31, 2010, we had $0.5 million principal
amount of fixed-rate debt and $327.4 million of
floating-rate debt outstanding. An interest rate increase of one
percentage point would cause an increase to cash interest
expense relating to our floating rate debt under our credit
agreements of approximately $3.3 million per year.
86
We use interest rate swaps to manage the risks associated with
variable-rate instruments. As of December 31, 2008, the
Company had three interest rate swap agreements outstanding. One
of these agreements matured in February 2009. The remaining two
agreements matured on February 28, 2010. The Company
entered into a new interest rate swap agreement that became
effective on February 26, 2010 and will mature on
February 28, 2012, which represented approximately 22% of
our floating rate term loan borrowings as of March 31,
2010. In addition, we have an interest rate cap and an interest
rate swap that became effective February 26, 2010 with an
aggregate notional amount of $254.5 million. Details of our
interest rate swaps and caps are as follows:
|
|
|
| |
•
|
An interest rate swap with a total notional amount of
$165.0 million that fixes the interest rate for a
corresponding amount of term loan borrowings under our credit
agreements at 4.825%, which expired on February 26, 2010.
|
| |
| |
•
|
An interest rate swap with a total notional amount of
€40.0 million that fixes the interest rate for a
corresponding amount of term loan borrowings under our First
Lien Credit Agreement at 3.998%, which expired on
February 26, 2010.
|
| |
| |
•
|
An interest rate cap with a total notional amount of
$175.0 million that became effective February 26, 2010
and that caps the interest rate for a corresponding amount of
term loan borrowings under our credit agreements at 3.0%,
expiring on February 28, 2012.
|
| |
| |
•
|
An interest rate swap with a total notional amount of
€53.0 million that became effective February 26,
2010 that fixes the interest rate for a corresponding amount of
term loan borrowings under our First Lien Credit Agreement at
2.125%, expiring on February 28, 2012.
|
Since these contracts have not been designated by our management
as accounting hedges for financial reporting purposes, we report
the swap contracts as derivatives in our balance sheet at their
fair values and any changes in fair value are reported in our
earnings. We use dealer quotations that are based on active
markets to measure fair values.
Foreign
Currency Risk
We conduct operations in many countries around the world. Our
results of operations are subject to both currency transaction
risk and currency translation risk. We incur currency
transaction risk whenever we enter into either a purchase or
sale transaction using a currency other than the reporting
currency of the transacting entity. With respect to currency
translation risk, our financial condition and results of
operations are measured and recorded in the relevant domestic
currency and then translated into U.S. dollars, our
reporting currency, for inclusion in our consolidated financial
statements. In recent years, exchange rates between these
currencies and U.S. dollars have fluctuated significantly
and may do so in the future. Approximately half of our net sales
and costs are denominated in U.S. dollars. We also have net
sales and costs denominated in Euros, Swedish Kronor and British
Pound Sterling.
Our
Euro-denominated
debt under our First Lien Credit Agreement is not hedged for
accounting purposes. Changes in the foreign exchange rate would
impact the Swedish Kronor carrying value of this unhedged
long-term debt, as well as our interest expense and earnings per
share on a full-year bases, as follows:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
EUR change in carrying
|
|
EUR change in Annual
|
Change in
|
|
value of Long-term Debt
|
|
Interest Expense
|
|
EUR vs SEK
|
|
EUR
|
|
USD(1)
|
|
EUR
|
|
USD(1)
|
|
|
|
€
|
0.5
|
|
|
€
|
3.3
|
|
|
$
|
4.6
|
|
|
€
|
0.1
|
|
|
$
|
0.2
|
|
|
|
1.0
|
|
|
|
6.9
|
|
|
|
9.6
|
|
|
|
0.3
|
|
|
|
0.4
|
|
|
|
1.5
|
|
|
|
10.9
|
|
|
|
15.2
|
|
|
|
0.4
|
|
|
|
0.6
|
|
|
|
2.0
|
|
|
|
15.4
|
|
|
|
21.5
|
|
|
|
0.6
|
|
|
|
0.9
|
|
|
|
|
| |
(1)
|
The rate used to calculate the U.S. dollar impact of the changes
was $1.4/€, which is the forecasted exchange rate used in
our 2010 budget.
|
87
Credit
Risk
Our customers are diversified by industry and geography with
approximately 1,000 customers in approximately 80 countries
worldwide and our receivables are not concentrated with any
customer. The recent global economic downturn may affect our
overall credit risk. Where exposed to credit risk, we analyze
the counterparties’ financial condition prior to entering
into an agreement or establishing credit limits and we monitor
the appropriateness of those limits on an ongoing basis. We also
obtain cash, letters of credit or other acceptable forms of
security from customers to provide credit support, where
appropriate, based on our financial analysis of the customer and
the contractual terms and conditions applicable to each
transaction.
Commodity
Price Risk
Our results of operations are directly affected by the cost of
our raw materials, particularly CTO and CST. Raw materials
accounted for approximately 60% of our costs of goods sold in
2009, and, accordingly, our gross profit and margins could be
adversely affected by changes in the cost of these raw materials
if we are unable to pass the increases on to our customers.
Energy purchases constituted approximately 9% of our cost of
goods sold for the three months ended March 31, 2010 and
2009 and 8% of our cost of goods sold for the year ended
December 31, 2009. Increases in energy costs, unless passed
on to our customers, would adversely affect our results of
operations. In addition, rising energy costs may increase our
raw material costs. If energy prices increase significantly, our
business or results of operations may be adversely affected. In
addition, rising energy costs negatively impact our customers
and the demand for our products. These risks will be heightened
if our customers or production facilities are in locations
experiencing severe energy shortages.
As discussed above under “Off-Balance Sheet
Arrangements”, we use forward purchase contracts to reduce
commodity price risk on natural gas purchases. At the end of
2009, an unfavorable 10% change in commodity futures prices
would have resulted in an unrealized net loss of
$0.4 million.
Critical
Accounting Policies
The application of accounting policies and estimates is an
important process that continues to evolve as our operations
change and accounting guidance is issued. We base our estimates
and judgments on historical experience and on other assumptions
that we believe are reasonable at the time of application.
Actual results may differ from these estimates and judgments.
The estimates and judgments may change over time and as more
information becomes available. If actual results differ from
these estimates and judgments, adjustments are made in
subsequent periods to take into consideration the new
information. On an ongoing basis, we evaluate our estimates.
We believe that the following critical accounting policies
affect our more significant judgments and estimates used in the
preparation of our financial statements.
Impairment of
Long-Lived Assets
We evaluate long-lived assets, such as property, plant and
equipment and intangible assets subject to amortization, for
impairment upon the occurrence of events or changes in
circumstances that indicate that the carrying value of the
assets may not be recoverable. Factors considered important that
could result in an impairment review include, but are not
limited to, significant underperformance relative to historical
or planned operating results, significant changes in the manner
of use of the assets, or significant changes in our business
strategies. An impairment loss is recognized when the estimated
undiscounted cash flows expected to result from the use of the
asset, plus net proceeds expected from disposition of the asset
(if any) are less than the carrying value of the asset. When an
impairment loss is recognized, the carrying amount of the asset
is reduced to its estimated fair value based on quoted market
prices or other valuation techniques. Key assumptions in
88
determining the future cash flows include the useful life,
technology, competitive pressures and raw material pricing.
Trade names, which are intangible assets determined to have
indefinite lives and goodwill related to our equity interest in
Arboris, LLC, are reviewed for impairment annually or more
frequently, if events or changes in circumstances indicate that
the intangible asset might be impaired. Fair values for trade
names and goodwill are determined based on discounted cash flows.
The determination of both undiscounted and discounted cash flows
requires us to make significant estimates and consider the
expected course of action at the date of determination.
Subsequent changes in estimated undiscounted and discounted cash
flows arising from changes in anticipated actions could impact
the determination of whether an impairment exists, the amount of
the impairment charge recorded and whether the effects could
materially impact our consolidated financial statements.
Income
Taxes
We conduct operations in Finland, France, Germany, Hong Kong,
Luxembourg, Mexico, The Netherlands, Russia, Singapore, Sweden,
the United Kingdom and the United States. As a result, income
tax amounts are reflected in these consolidated financial
statements for each of those jurisdictions.
We account for income taxes using the asset and liability
method. Under this method, we recognize deferred tax assets and
liabilities attributable to differences between the carrying
amounts of existing assets and liabilities and their respective
tax bases. We measure deferred tax assets and liabilities on a
legal entity basis using enacted tax rates expected to apply to
taxable income in the years in which those temporary differences
are expected to be recovered or settled. We recognize the effect
on deferred tax assets and liabilities of a change in tax rates
in income in the period that includes the enactment date.
Net operating losses and credit carryforwards are recorded in
the event such benefits are expected to be realized. Deferred
taxes result from differences between the financial and tax
bases of our assets and liabilities and are adjusted for changes
in tax rates and tax laws when changes are enacted. Valuation
allowances are recorded to reduce deferred tax assets when it is
more likely than not that a tax benefit will not be realized.
Deferred taxes are not provided for temporary differences
representing earnings of the non-Swedish entities that are
intended to be permanently reinvested.
In assessing the realizability of deferred tax assets,
management considers whether it is more likely than not that
some or all of the deferred tax assets will not be realized. The
ultimate realization of deferred tax assets is dependent upon
the generation of future taxable income during the periods in
which temporary differences become deductible as well as the
reversal of certain deferred tax liabilities. Management also
considers projected future taxable income, tax planning
strategies, and other factors in making this assessment and
establishing an appropriate valuation allowance. Based upon
these factors, management believes it is more likely than not
that we will realize the benefits of these deductible
differences, net of the existing valuation allowances. The
amount of the deferred tax assets considered realizable,
however, could be reduced if expected taxable income or other
factors change in future years.
As of December 31, 2009 and 2008, we had worldwide net
operating loss carryforwards of $160.5 million and
$149.7 million, respectively, of which $41.4 million
and $36.8 million expire in varying amounts between 2017
and 2029, while the remaining $119.1 million and
$112.9 million have indefinite lives based on the laws of
the jurisdictions in which they were generated. We have provided
valuation allowances against certain Net Operating Loss (NOL)
carryforwards due to the uncertainty of their realization.
89
We provide liabilities for uncertain tax positions for federal,
state, local and international exposures relating to periods
subject to audit. The development of liabilities for uncertain
tax positions for these exposures requires judgments about tax
issues, potential outcomes and timing and is a subjective
critical estimate. We assess our tax positions and record tax
benefits based upon management’s evaluation of the facts,
circumstances and information available at the reporting dates.
For those tax positions where it is more-likely-than-not that a
tax benefit will be sustained, we have recorded the largest
amount of tax benefit with a greater than 50% likelihood of
being realized upon settlement with a tax authority that has
full knowledge of all relevant information. For those tax
positions where it is not more likely than not that a tax
benefit will be sustained, no tax benefit has been recognized in
the financial statements.
Pension, Early
Retirement and Long-term Service Awards
We sponsor noncontributory defined benefit pension plans. We
also sponsor an Early Retirement plan and Long-term Service
Award plan. The actuarial determination of the projected benefit
obligations and related benefit expense requires that certain
assumptions be made regarding such variables as expected return
on plan assets, discount rates, rates of future compensation
increases, estimated future employee turnover rates and
retirement dates, distribution election rates, mortality rates,
retiree utilization rates for health care services and health
care cost trend rates. The selection of assumptions requires
considerable judgment concerning future events and has a
significant impact on the amount of the obligations recorded in
the consolidated balance sheets and on the amount of expense
included in the consolidated statements of operations.
Capital market declines experienced during the last half of 2008
have adversely impacted the market value of investment assets
used to fund our defined benefit pension plans. Future changes
in plan asset returns, assumed discount rates and various other
factors related to our pension and post-retirement plans will
impact future pension expense and liabilities.
In selecting the discount rates, we used as our general
benchmark the single discount rate equivalent to discounting the
expected cash flows from each plan using the yields at each
duration whenever possible or an index with duration adjustment
to match the plan’s liability duration where full yield
curve data is not readily available. For the U.S., the discount
rate was derived based on external pension discount yield
curves. For the
non-U.S. plans
the discount rate was derived based on the corporate bond
indices and government bonds adjusted for the Company’s
risk premium and the plan liability duration.
Stock-Based
Awards
As part of the Acquisition, AZ Chem Investments LLC, the general
partner of AZ Chem Investments Partners LP, our parent, formed
AZ Chem MIV I Ltd, which we refer to as MIV I, and AZ Chem
MIV II LP, which we refer to as MIV II. We refer to MIV I
and MIV II together as the “MIVs”. The MIVs were
created as vehicles to enable certain members of our management
and board of directors to participate in the ownership of AZ
Chem Investments Partners LP through equity purchases and grants
of awards under our management incentive plan. Since inception,
AZ Chem Investments LLC granted partnership equity interests in
the MIVs to certain participants. These grants vest over five
years, and the resulting compensation expense is recorded by us
based on the fair value of the Company at the time of each
grant. The compensation cost of grant awards made under our
management incentive plan is charged to selling, general and
administrative expense over the applicable periods.
Recent Accounting
Pronouncements
In June 2009, the FASB issued authoritative guidance to
eliminate the exception to consolidate a qualifying
special-purpose entity, change the approach to determining the
primary beneficiary of a variable interest entity and require
companies to more frequently re-assess whether they must
consolidate variable interest entities. Under the new guidance,
the primary beneficiary of a variable
90
interest entity is identified qualitatively as the enterprise
that has both (a) the power to direct the activities of a
variable interest entity that most significantly impact the
entity’s economic performance and (b) the obligation
to absorb losses of the entity that could potentially be
significant to the variable interest entity or the right to
receive benefits from the entity that could potentially be
significant to the variable interest entity. This standard is
effective for financial statements, for interim or annual
reporting periods, beginning after January 1, 2010. We do
not expect this guidance to have a material impact on our
consolidated financial statements.
In May 2009, the FASB issued new guidance related to the
disclosure of subsequent events. This standard is intended to
establish general standards of accounting for and disclosure of
events that occur after the balance sheet date but before
financial statements are issued or are available to be issued.
Specifically, this standard sets forth the period after the
balance sheet date during which management of a reporting entity
should evaluate events or transactions that may occur for
potential recognition or disclosure in the financial statements,
the circumstances under which an entity should recognize events
or transactions occurring after the balance sheet date in its
financial statements, and the disclosures that an entity should
make about events or transactions that occurred after the
balance sheet date. This guidance is effective for fiscal years
and interim periods ended after June 15, 2009. The
additional disclosures have been included in the notes to our
consolidated financial statements.
In April 2009, the FASB issued authoritative guidance to require
that assets acquired and liabilities assumed in a business
combination that arise from contingencies be recognized at fair
value if fair value can be reasonably determined. If the fair
value of such assets or liabilities cannot be reasonably
determined, then they would generally be recognized in
accordance with certain other pre-existing accounting standards.
This guidance also amends the subsequent accounting for assets
and liabilities arising from contingencies in a business
combination and certain other disclosure requirements. This
guidance was effective for assets or liabilities arising from
contingencies in business combinations for which the acquisition
date was on or after the beginning of our annual reporting
period beginning January 1, 2009. The adoption of this
standard did not have a material impact on our consolidated
financial statements.
In November 2008, the FASB issued authoritative guidance
regarding the accounting for defensive intangible assets.
Defensive intangible assets are assets acquired in a business
combination that the acquirer (a) does not intend to use or
(b) intends to use in a way other than the assets’
highest and best use as determined by an evaluation of market
participant assumptions. While defensive intangible assets are
not being actively used, they are likely contributing to an
increase in the value of other assets owned by the acquiring
entity. This guidance will require defensive intangible assets
to be accounted for as separate units of accounting at the time
of acquisition and the useful life of such assets would be based
on the period over which the assets will directly or indirectly
affect the entity’s cash flows. This guidance would be
effective for intangible assets acquired in a business
combination for which the acquisition date was on or after the
beginning of our annual reporting period beginning
January 1, 2009. The adoption of this standard did not have
a material impact on our consolidated financial statements.
In November 2008, the FASB issued authoritative guidance to
address questions about equity-method accounting. The primary
issues include how the initial carrying value of an equity
method investment should be determined, how to account for any
subsequent purchases and sales of additional ownership interests
and whether the investor must separately assess its underlying
share of the investee’s indefinite-lived intangible assets
for impairment. This guidance was effective for the beginning of
our annual reporting period beginning January 1, 2009. The
adoption of this standard did not have a material impact on our
consolidated financial statements.
91
INDUSTRY
Pine Chemicals
Industry Overview
The pine chemicals industry refines and upgrades raw materials
that are naturally derived from pine trees into specialty
chemicals used in an extensive range of applications and
products across diverse industrial and consumer markets. Pine
chemicals are derived primarily by either refining co-products
of the kraft wood pulping process used in paper manufacturing
(representing approximately 60% of overall industry supply by
volume) or by manually tapping individual pine trees
(representing approximately 40% of overall industry supply by
volume). According to ADL, the global supply for pine chemicals
from all feedstocks in 2009 was approximately 2.5 million
metric tonnes, with sales of approximately $3 billion.
Pine trees are used as the primary raw material for kraft pulp
because of the long fiber nature of the wood, which provides the
strength and durability required for the paper and packaging
applications in which kraft pulp is used. Due to the high
quality supply of pine trees that are found in abundance in the
Southeastern United States, Scandinavia and Russia, pulp and
paper manufacturers have typically centered their kraft pulp
manufacturing operations in these regions. Kraft pulp production
in both the United States and Scandinavia is mature with growth
at rates at or below GDP, but we expect the availability of CTO
and CST to grow with increased kraft pulp manufacturing in
Russia and, to some extent, South America.
The kraft pulping process yields as co-products two pine
chemical feedstocks: crude tall oil, or CTO, and crude sulfate
turpentine, or CST. The term “tall oil” is derived
from the Swedish word “tallolja”, which means
“pine oil”.
Products obtained by manually tapping individual pine trees are
referred to as “gum” products and include gum rosin
and gum turpentine. While the characteristics of CTO, CST and
gum-based products vary, gum rosin is generally interchangeable
for tall oil rosin, a product derived from CTO, as are gum
turpentine fractions for CST fractions.
The process of manually tapping individual pine trees is labor
intensive, and as such, the majority of global production of gum
rosin and gum turpentine originates from emerging economies such
as China and, to a lesser extent, Indonesia and Brazil. As per
capita income in these economies increases over time, we believe
that prices for gum rosin and gum turpentine may increase and
global supply may decrease as tapping becomes less economically
viable and as internal demand for such products displaces export
supply. This has already occurred in other former, major
gum-producing countries, such as the United States, Portugal and
Spain.
CTO is refined into its primary constituent fractions: tall oil
rosin, or TOR, tall oil fatty acid, or TOFA, distilled tall oil,
or DTO, and tall oil pitch, or pitch. In some cases, these
products are sold directly to third parties, but in many cases
they are chemically upgraded via reaction with other materials
into higher priced, value added specialty chemicals. The degree
to which the products are processed depends on the capability of
the manufacturer and market conditions.
According to ADL, the main refiners of CTO are Arizona Chemical,
with annual capacity of 650,000 metric tonnes at locations in
the United States and Europe, MeadWestvaco Corporation, with
annual capacity of 250,000 metric tonnes at locations in the
United States, Georgia-Pacific LLC with annual capacity of
120,000 metric tonnes at locations in the United States,
Forchem Oy, with annual capacity of 175,000 metric tonnes
at one location in Europe and Les Dérivés
Résiniques et Terpéniques with annual capacity of
70,000 metric tonnes in Europe.
CST can be distilled directly into terpene monomers. The main
products resulting from the refining of CST are alpha pinene,
beta pinene and delta 3 carene, which are used as feedstocks in
the adhesives, flavor and fragrance, polymer processing,
solvents and cleaners and chewing gum resin markets.
92
According to ADL, global supply of CST amounted to approximately
140,000 metric tonnes in 2009, more than 90% of which was
refined and approximately 5% of which was burned in pulp mills.
The main refiners of CST are Arizona Chemical, Lyondell-Basell
Millennium Division, International Flavors and Fragrances, Inc.
and Les Dérivés Résiniques et Terpéniques,
with refining capacities of approximately 39,000 metric
tonnes, 40,000 metric tonnes, 25,000 metric tonnes and
20,000 metric tonnes, respectively.
The chart below illustrates the materials produced from the
refining and upgrading of CTO and CST, and indicates the
percentage of each material that results from the refining of
CTO. These percentages can vary depending on a number of
factors, including the species of the pine trees from which the
CTO is derived, the time of year the pine trees were harvested,
the geographic region in which the pine trees grew and,
particularly with respect to DTO, the degree to which the CTO is
refined.
| |
|
|
|
|
|
Raw Materials
|
|
Refine
|
|
Upgrade
|
|
|
TOR and Rosin
Resins
Rosin, such as TOR, is a chemical whose properties allow for a
stronger bond between different substances. This property is
commonly known as tack, and rosin is a tackifier. Rosin is
primarily upgraded chemically into other products, including
(i) rosin ester through a process called
“esterification”, which is the reaction of the base
rosin with an alcohol, (ii) disproportionated rosin through
a process called “disproportionation”, which is a
reaction whereby the base rosin is split into two different
substances, and (iii) hydrogenated rosins through a process
called “hydrogenation”, which is
93
the reaction of the base resin with hydrogen. Rosin derivatives,
which are also known as “resins”, are key ingredients
in the composition of adhesives, inks, tires and roadmarkings.
Resins impart attributes such as bonding strength, viscosity and
drying speed for adhesive products. Resins are also a critical
ingredient in inks, where they are used to enhance or control
adhesion, drying speed, color development and gloss. In the
roadmarking coatings market, rosin-based resins are used as
binders in thermoplastic roadmarkings. Rosin derivatives are
also used extensively as emulsifiers to assist in the mixing of
monomer components in the manufacture of synthetic rubber.
According to ADL, total global rosin production in 2009 was
approximately 1.3 million metric tonnes, approximately 70%
of which was derived from gum rosin and 30% of which was derived
from refining CTO.
The leading producers of TOR derivatives are Arizona Chemical,
with approximately 200,000 metric tonnes of annual capacity,
MeadWestvaco Corporation, with approximately 50,000 metric
tonnes of annual capacity, Georgia-Pacific LLC, with
approximately 30,000 metric tonnes of annual capacity in North
America and Les Dérivés Résiniques et
Terpéniques, with approximately 10,000 metric tonnes of
annual capacity in Europe.
2009 Global Rosin
Consumption by Application
TOFA and Dimer
Acids
According to ADL, TOFA represented approximately 10% of the
global fatty acid market in 2009, with total production of
approximately 500,000 metric tonnes. Fatty acids represent
approximately 3.5% of the global oils and fats market, with
estimated total production of 170 million metric tonnes in
2009. Major applications for TOFA include alkyd resins for
solvent-based paints, lubricity additives for low-sulfur diesel
fuel, heat stabilizers for PVC and flotation agents for
phosphate mining. Another important use is as a starting point
for the manufacture of dimer acid, which in turn forms the basis
for the manufacture of polyamides for use in adhesives, inks and
coating resins. According to ADL, alkyd resins consumed
approximately 40% of the global supply of TOFA in 2009, while
dimers consumed approximately 20% and fuel additives consumed
approximately 10%, with the remaining approximately 30% spread
across many smaller applications, including lubricants.
TOFA competes with unsaturated vegetable oils and fatty acids,
such as those derived from soy, linseed or sunflower. TOFA is
less dependant on seasonal weather conditions than other
agricultural substitutes, resulting in more consistent quality
and less supply volatility. In addition, TOFA is more suitable
than vegetable fatty acids in some applications, leading to
improved performance of the customer’s products, which
results in a pricing premium for TOFA compared to vegetable
fatty acids in some of the markets in which it is used. TOFA is
a non-food derived fatty acid and is expected to benefit from
the resistance to diverting food crops such as soybeans for
industrial uses in light of recent pressure on food prices and
supplies.
94
According to ADL, the main producers of TOFA are Arizona
Chemical, with approximately 200,000 metric tonnes of annual
capacity at locations in the United States and Europe,
MeadWestvaco Corporation, with approximately 70,000 metric
tonnes of annual capacity at locations in North America, Forchem
Oy, with approximately 60,000 metric tonnes of annual capacity
at locations in Europe and Georgia-Pacific LLC, with
approximately 45,000 metric tonnes of annual capacity at
locations in North America.
2009 Global TOFA
Consumption by Application
DTO
DTO is a specialty acid stream from the CTO distillation process
that contains a mixture of TOFA and a variable percentage
(typically
5-30%) of
rosins. CTO refiners are able to produce variable quantities of
DTO based on market demand, which ADL estimated to be 60,000 to
80,000 metric tonnes in 2009. DTO is generally sold without
upgrade and is used as the basis for a variety of applications
from metalworking fluids to specialty alkyds, rubber processing
and oil field drilling muds. DTO is a sustainable alternative to
hydrocarbon esters and is preferred in certain applications such
as metalworking due to its superior performance.
According to ADL, the main producers of DTO are Arizona
Chemical, with approximately 50% of global output at locations
in the United States and Europe, MeadWestvaco Corporation, with
approximately 20% of global output at locations in North America
and Forchem Oy, with approximately 20% of global output at
locations in Europe.
Pitch and
Sterols
Pitch is a by-product of CTO refining and represents
approximately 40% of CTO refining volume, with a global supply
of approximately 650,000 metric tonnes. Pitch is used to produce
fuel blends for industrial and heating applications.
Additionally, sterols can be extracted from pitch for use as a
raw material in high margin pharmaceutical and food additives
used to reduce cholesterol absorption. Sterols typically
constitute approximately 5% of total pitch by volume. Pine
products have a higher concentration of sterols than vegetable
oils and, unlike oil derived from soybeans, pine sterols are not
considered a genetically-modified product. Demand for sterols
produced from pine benefits from consumer resistance to
genetically-modified food products, particularly in Europe.
According to ADL, the three companies that produce pitch-based
sterols are Arboris, LLC, a joint venture in which Arizona
Chemical owns a 10% interest, with approximately 5,000 metric
tonnes of annual capacity, Cognis GmbH, with approximately 2,000
metric tonnes of annual capacity, and Les Dérivés
Résiniques et Terpéniques, with approximately 400
metric tonnes of annual capacity.
95
Markets for
Pine-based Chemicals
Globally, pine chemicals are sold into a wide range of different
markets. We are focused primarily on six markets within which we
believe we have a competitive advantage and opportunities for
growth with higher margin products: (1) adhesives,
(2) inks, (3) roads and construction, (4) tires
and rubber, (5) consumer products and (6) renewable
energy.
Adhesives
Market Overview
Adhesives are generally defined as materials that bond two
surfaces together. The required degree of bonding strength is
dependent on the application. For instance, adhesives used in
packaging or tapes and labels generally require a low bond
strength while adhesives used in industrial applications such as
bookbinding, furniture and woodworking, and appliance
manufacturing require higher bonding strength. Certain adhesive
applications such as those used in building construction and
aerospace require a very high level of bonding strength and are
generally referred to as structural adhesives.
The chemical composition of an adhesive is dictated by the need
for particular performance characteristics, such as bonding
strength, and by processing requirements, such as whether the
adhesive can be applied at high temperature or needs to be
workable under ambient conditions. Accordingly, there are
different classes of adhesives. For example, “hot
melts” are applied at high temperature and form their bonds
upon cooling. “Solvent-based adhesives” are dissolved
in a solvent and the adhesive bond forms when the solvent
evaporates. “Water-based adhesives” are finely
dispersed in water, which is evaporated to form the bond.
Finally, there are many adhesive types that form their bonds by
a chemical reaction taking place during hardening or
“curing”, which are referred to as “reactive
adhesives”.
| |
|
|
2009 Global Tackifier Market by
Chemistry
|
|
2009 Global Tackifier Market by
Application
|
|
(Approximately $1.6 billion)
|
|
(Approximately $1.6 billion)
|
|
|
|
|
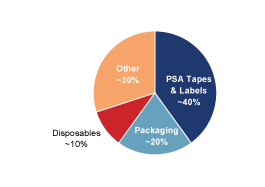
|
Pine chemicals are principally used as tackifiers in certain
classes of industrial adhesives to confer the essential
“grip” to the bonded surfaces. Their use predominates
in hot melt adhesives, although they are also used in other
classes such as water-based and solvent-based. Tackifiers are
generally not required in reactive adhesives for structural
applications because the chemical reactions create sufficiently
high bond strength. Where tackifiers are used, they generally
comprise between 25% and 50% of an adhesive by volume (on a
solid basis), according to ADL. The remaining adhesive is
composed of a polymer, which typically represents between 40% to
60% of the adhesive by volume and various additives such as
waxes to control open time (the time it takes the adhesive to
cool and set after application) and anti-oxidants to protect
them from degradation by oxygen in the air during heating cycles.
96
Demand for hot melt adhesives has grown at rates above the
industry average because they have low emission levels, are
effective on a wide variety of surfaces, are easy to apply and
therefore offer faster line speeds than other adhesive types.
According to ADL, the global demand for industrial adhesives in
2009 was approximately 13 million metric tonnes with gross
revenues of approximately $45 billion. The global market
for tackifiers was estimated to be 1.4 million metric
tonnes, or approximately $1.6 billion, in 2009,
approximately 65% of which was sourced from hydrocarbons, and
approximately 35% of which was sourced from pine-derived
chemicals. Of the tackifiers derived from renewable sources, ADL
estimates that 85% are from upgrades of rosin (tall oil or
gum-based), and the remaining 15% are from CST-based and
D-limonene-based terpenes.
According to ADL, the volume of the industrial adhesives market
grew on average
3-4% per
year for the period from 2006 to 2008, which was driven by their
increased use in developing economies. Tackifier demand grew in
line with the broader industrial adhesives market. In addition
to growing demand from developing economies, the use of
industrial adhesives is increasing due to the following factors:
|
|
|
| |
•
|
replacement of mechanical fasteners with adhesives in order to
increase production rates, accommodate smaller and thinner
devices, reduce weight, and improve strength;
|
| |
| |
•
|
increasing demand for pre-packaged, ready-made foods which
require packaging that can withstand freezer and cooking
temperatures and seals tightly to preserve freshness. Adhesive
technology bonds the different packaging surfaces such as foil
and plastic; and
|
| |
| |
•
|
increasing environmental awareness and regulatory initiatives
relating to sustainable materials are increasing demand for
recycled content in packaging, reduced volatile organic content,
and reduced use of hydrocarbon-derived materials. We believe
pine chemical-derived products are competitive alternatives.
|
Inks Market
Overview
Inks used in large printing presses for the production of
newspapers, magazines, catalogs, brochures and packaging
materials are referred to as printing inks. Printing inks are
composed of a pigment, or color, which is mixed with a resin in
a carrier solvent which can be mineral oil, toluene, alcohol or
water, depending on the printing technology. The resin acts as
the binder, which is a critical component of the ink that holds
the pigment to the printed surface and influences ink attributes
such as gloss, drying speed, viscosity and color intensity. The
solvent is used to adjust the ink viscosity for easy application
and evaporates after the ink is applied. As such, the solvent
also helps control drying speed.
Depending on the printing process and ink properties, ink
manufacturers can select from a number of classes of binder.
According to ADL, pine chemical binders constitute the largest
class of binder, with an approximately 50% share of the inks
market, followed by hydrocarbons, with approximately 20%, alkyds
with approximately 15% and acrylics with approximately 10%.
Various other types of binders make up the remainder. Gum-rosin
resin has historically dominated the rosin-based printing inks
market, as it has been more consistent in performance and easier
to work with than available alternatives. However, pine chemical
companies have pioneered the use of TOR resin as a substitute
for gum products by developing these resins to be as reliable
and reproducible as gum-based resins. Additionally, TOR is
readily available, often in convenient molten form, whereas gum
rosin generally has to be sourced in drums from Asia or Latin
America. As such, ink resin producers in the United States and
Europe have driven the adoption of TOR-based resin, and,
according to ADL, its share of the rosin-based ink resin market
is estimated to be approximately 25% today.
The printing inks market is divided into two principal areas of
application: publication, including inks printed on newspapers,
magazines and catalogs, which represents approximately 30% of
printing inks sales, and packaging, which represents
approximately 40% of printing inks sales, according to ADL. In
publication, gravure and lithographic processes are the primary
means of printing. Gravure is
97
a high quality, cylindrical roller-based printing process used
for high volume “upscale” magazines and catalogs.
Lithography is a rigid plate-based printing process used for
medium to high volume printing such as newsprint, books and
directories. Printing inks based on naturally-derived resins
predominate in these processes, although hydrocarbon-based
resins are also used. In the packaging-market, where the
flexographic process dominates,
naturally-derived
resins, especially fatty acid-derived polyamides, compete with
hydrocarbon-based resins, specifically polyurethanes.
Flexography is a flexible plate-based printing process used for
the printing of flexible packaging, including wrappers, films
and cartons. According to ADL, the global demand for printing
inks in 2009 was approximately 4 million metric tonnes,
with gross revenues of approximately $16.0 billion. The
global demand for ink resin binders was approximately
1.1 million metric tonnes with gross revenues of
approximately $1.5 billion.
According to ADL, the volume of the global printing inks market
is expected to lag GDP growth and grow at the rate of
approximately 1% annually through at least 2013. Demand for
publication inks is expected to decline in developed regions due
to the continued rise of electronic media. The volume of the
global packaging inks-market is expected to grow at 3% per year
from 2010 through 2013 to 2015, driven by demand for packaging
in developed countries, while growing in line with GDP in
emerging markets.
| |
|
|
2009 Global Ink Sales by
Region
|
|
2009 Global Ink Sales by
Application
|
|
($16 billion)
|
|
($16 billion)
|
|
|
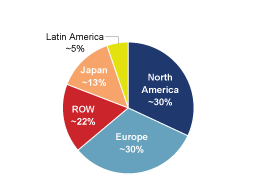
|
|
|
Roads and
Construction Market Overview
The roads and construction market can be divided into three
sub-markets:
roadmarking, paving and roofing.
Roadmarking
The roadmarking sub-market consists of paints and coatings which
are applied to roadways, parking lots and airstrips.
Thermoplastic roadmarkings, where a resin binder is used in
combination with pigments and other materials, such as glass
beads, are one of the most common types of coatings. The glass
beads reflect vehicle lights to make the roadmarkings more
visible, and the binder holds the mixture together and
distributes and suspends the pigments and glass beads in the
thermoplastic. In addition to its binding properties, the binder
gives the final product hardness and durability. Binders used in
thermoplastic roadmarkings can be naturally-derived resins such
as TOR resin and gum rosin resin, or hydrocarbon-based resins,
though rosin-based resins offer superior performance to
hydrocarbon-based resins due to their broad compatibility with
other elements.
Additionally, the use of naturally-derived resins in
roadmarkings has increased in recent years as a result of
increasing demand for sustainable raw materials. Governments
across Europe and parts of the United States have increasingly
required the use of more sustainable materials in roadmarkings,
driving demand for naturally-derived resins.
98
According to ADL, gross sales of roadmarkings in 2008 were
approximately $1.5 billion. In 2008, thermoplastic
roadmarking, with approximately $600 million in sales,
constituted 40% of the market. The primary end consumers are
local and national government road authorities, with growth
driven by new road construction.
| |
|
|
Breakdown of Different
Roadmarking Coating
|
|
Competing Technologies in
Thermoplastic
|
|
Types ($1.5 billion)
|
|
Systems
|
|
|
|
|
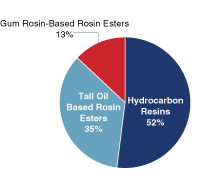
|
Paving
Bitumen, a black, sticky, tar-like substance obtained as the
bottom fraction in the distillation of crude oil, is commonly
used as the binder in paving and roofing applications.
Approximately 10% of the bitumen in the United States and Europe
is modified through the addition of synthetic rubber to impart
favorable attributes such as improved durability and temperature
resistance. As modified bitumen is composed of multiple
substances, including most commonly synthetic rubber based on
styrene-butadiene-styrene (SBS) or styrene-butadiene (SB)
rubber, it is often necessary to add a compatibilizer to
stabilize the mixture so that it can be consistently applied.
Without a compatibilizer, the bitumen and synthetic rubber can
separate during storage and transport. Pine chemical resins,
which are effective in combining with a wide variety of
materials, can be used as the compatibilizer in modified
bitumen. The use of modified bitumen is expected to grow in
excess of 5% globally through 2015 as modified bitumen becomes
increasingly popular relative to standard bitumen and as the
sub-market requires roads and paving to last for increasingly
longer periods of time.
As a result of sustained higher global oil prices, it has become
economically viable for oil companies to employ higher-cost
methods of crude oil distillation that improve their yields. As
a result of the employment of these methods, the quality of the
bitumen that remains after the crude oil is distilled has
declined, and the demand for additives required to stabilize the
bitumen mix, including pine chemical resins, has increased.
Another recent development in modified bitumen has been the use
of recycled tires, referred to as crumb rubber due to its
granular consistency, as a substitute for SBS and SB rubber in
modified bitumen. Governments, particularly those in the United
States, Southern Europe and China actively encourage the use of
crumb rubber in modified bitumen because it is cost effective
and the recycling of tires is considered environmentally
friendly. However, crumb rubber is more difficult to process
than new synthetic rubber and requires higher temperatures and
stronger compatibilizers to prevent the mix from separating.
Roofing
In the flat roof segment of the roofing sub-market, most roofs
are waterproofed with a bitumen roofing membrane. Bitumen is
commonly used as an adhesive to adhere the membrane to the roof
surface. There is a growing trend toward membranes sold with a
self-adhesive incorporated into the product, so-called
“Peel & Stick” products, which, according to
ADL, represent approximately 15% of
99
the volume in the roof membrane market. These
pressure-sensitive
adhesives are bitumen based and require the incorporation of a
tackifier to promote adhesion to the roof. As there is a high
degree of variation in the roofs for which these membranes are
used, these products require a versatile tackifier. Pine-based
products are particularly effective in their versatility
compared to hydrocarbon tackifiers and, as a result, are
becoming the chemistry of choice for this growing range of
products.
According to ADL, the global demand for flat-roof membranes in
2009 was approximately 1.2 billion square meters, with
gross revenues of $4.0 billion, driven by an increasingly
energy conscious society seeking to insulate homes and other
buildings to conserve energy and reduce heating costs. The
volume of the market for these membranes is expected to grow in
line with GDP in North America and Europe and at approximately
5-10% per year in Asia from 2010 through 2013 to 2015.
| |
|
|
Global Demand for Bitumen by
Region
|
|
2009 World Bitumen Demand by
Market
|
|
(Approximately $45 billion)
|
|
(120 million metric tonnes)
|
|
|
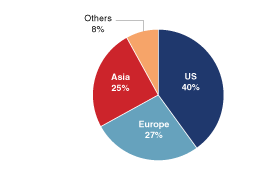
|
|
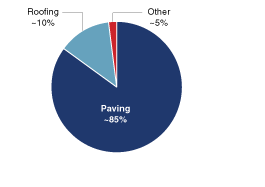
|
Tires and
Rubber Market Overview
Tires
In the passenger car tire market, traditional, lower-cost,
carbon black-filled tires are increasingly being replaced by
high performance, silica-filled tires. Silica-filled tires have
lower rolling resistance than traditional carbon black-filled
tires and therefore offer better fuel economy. However, there is
an inherent trade-off in tire construction between grip, rolling
resistance (which dictates fuel economy) and durability. As a
result, there is significant demand for enhancement additives
that can increase traction in silica-filled tires without
reducing durability or increasing rolling resistance. Various
hydrocarbon-based resins such as alpha-methyl-styrene, or AMS,
and natural-based resins such as terpene resins, can be added to
modify the tread performance in order to optimize wet grip
performance, fuel economy and tire life. The market penetration
of silica-filled tires depends on geography. While silica-filled
tires are more fuel efficient than carbon black-filled tires,
they are also less durable. Consumers in Europe have shown
greater demand for silica-filled tires than those in North
America and Asia, reflecting traditionally higher fuel prices in
Europe that encourage greater fuel efficiency. As a result, the
market share of silica-filled tires in Europe is much higher at
65%, versus 30% in North America and 20% in Asia, according to
ADL.
100
ADL estimates that the global tire market in 2008 was
approximately $130 billion. The volume of the global tire
market is expected to grow at 3% per year through 2015, driven
by growth in developing countries. Demand for high performance,
silica-filled tires is expected to increase 10% per year through
2013, driving a similar increase in the demand for enhancement
additives. In 2009, the market for tread enhancement additives
was approximately $80 million.
| |
|
|
2008 Tire Market Value by
Region
|
|
2008 Tire Market Value by
Vehicle & Tire Type
|
|
($130 billion)
|
|
($130 billion)
|
|
|
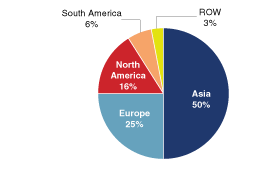
|
|
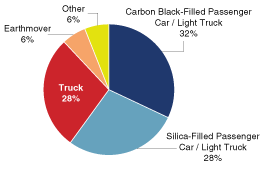
|
Rubber
Rubber is used in tires, hoses, belts, gloves, and
anti-vibration mounts in the automotive industry among other
applications. Various types of synthetic rubber can be made by
combining a number of different monomer building blocks. Rosin
soaps are used as emulsifiers, which are a type of additive used
in the polymerization step (the combining of multiple monomers)
to assist in the mixing and blending of these monomers. At
present, no viable alternatives to rosin-based emulsifiers exist
for the manufacture of synthetic rubber.
ADL estimates that the global synthetic rubber market had a
value of $17 billion in 2009, while the global market for
synthetic rubber additives had an estimated value of
$2 billion.
Naturally-derived
resins used in synthetic rubber production represented a small
percentage of the market for synthetic rubber additives. The
volume of the global market for synthetic rubber emulsifiers is
expected to grow by 2-2.5% per year through 2015, driven
primarily by tires and automotive demand.
| |
|
|
2009 Synthetic Rubber Sales by
Region
|
|
2009 Synthetic Rubber Sales by
End-Use
|
|
($17 billion)
|
|
($17 billion)
|
|
|
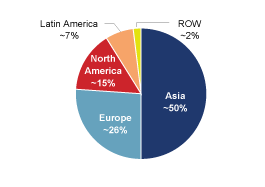
|
|
|
Consumer
Products Market Overview
A key driver today in the personal care industry is the
increasing preference by consumers for sustainable and
naturally-derived ingredients. Pine chemicals primarily function
in this market as
101
gellants and thickening agents in cosmetics and personal care
applications in order to modify certain physical characteristics
of water-based formulations. Common applications include:
|
|
|
| |
•
|
rheology modifiers, which alter the flow and feel of the product;
|
| |
| |
•
|
binders, which modify liquids into cakes, sticks and other
wax-like solids;
|
| |
| |
•
|
gelling agents, which modify liquids into hard and soft
gels; and
|
| |
| |
•
|
protective agents, which stabilize oil-water mixtures and
improve wear resistance.
|
Personal
Care
According to ADL, growth rates in the personal care market vary
between applications, with skin care and sun care growing at
more than 5% per year in volume, while
make-up,
hair care and antiperspirants/deodorants are expected to grow at
approximately 3% per year or less in volume. The volume of the
market for all personal care applications is estimated to be
growing at approximately 5% annually on a global basis.
Naturally-derived chemicals currently represent a small
proportion of the market relative to hydrocarbon-based
materials. However, as a result of shifting consumer
preferences,
naturally-derived
chemicals are expected to increase their market share.
Flavors and
Fragrances
Alpha- and beta-pinene and delta 3-carene, chemicals derived
from CST, are used as raw materials in the manufacture of
certain aroma chemicals used in the flavors and fragrances
market. Pinenes are the bases for a number of terpene-derived
fragrances, and certain fragrances specifically require pinenes
or similar derivatives as the aroma base. Alpha- and beta-pinene
compete with analogous derivatives from gum turpentine in the
flavors and fragrances market. According to ADL, terpene-derived
aroma chemicals accounted for approximately 35% of the
$2.6 billion aroma chemicals market in 2009, and the volume
of this market is expected to grow at approximately 2.5% per
year.
Renewable
Energy Market Overview
Pitch, a by-product of the CTO refining process, is a suitable
alternative to heavy fuel oil used in industrial manufacturing
plants, industrial heating and electrical generation.
Additionally, TOFA, a co-product of the CTO refining process,
like many other
naturally-derived
oils and fatty acids, can be used as a raw material in the
manufacture of bio-diesel.
In Europe, the European Union’s Directive on the Promotion
of the Use of Energy from Renewable Resources established a 20%
European Community-wide target for energy consumed from
renewable sources by 2020, and a 10% target for energy consumed
from renewable sources in the transport sector. In the United
States, the 2005 Energy Policy Act established renewable fuel
standards for transportation, including incentives for
biodiesel, and the standards established by the
U.S. Environmental Protection Agency under the National
Renewable Fuel Standard Program were recently amended, as
required by the Energy Independence and Security Act of 2007, to
establish new specific annual volume standards for cellulosic
biofuel, biomass-based diesel and advanced biofuel and to
establish requirements for higher levels of total renewable fuel
that must be included in transportation fuel.
The demand for both pitch and TOFA is expected to increase as a
result of these and similar legislative initiatives.
102
BUSINESS
General
We are the world’s leading supplier of pine-based chemicals
as measured by sales. We refine and further upgrade two primary
feedstocks, crude tall oil, or CTO, and crude sulfate
turpentine, or CST, both of which are wood pulping co-products,
into specialty chemicals. We focus our resources on six target
markets that we believe offer the greatest potential for growth
and in which we offer our highest value-added products. These
markets are (1) adhesives, (2) inks, (3) tires
and rubber, (4) roads and construction, (5) consumer
products and (6) renewable energy. Our leading position in
our target markets is supported by our recognized brands,
including
SYLVATAC®,
SYLVARES®,
SYLVAPRINT®
and
UNI-REZ®,
among others. These products are complemented by a portfolio of
chemical intermediates that includes tall oil rosin, or TOR,
tall oil fatty acid, or TOFA, dimer acid and distilled tall oil,
or DTO, which have contributed steady profit margins and stable
cash flows. These products are sold into a diverse range of
markets, including paints and coatings, lubricants, fuel
additives, mining and oilfield, paper chemicals and polymers,
among others.
The chart below illustrates the materials produced through the
refining and upgrading of CTO and CST and the markets and
sub-markets into which those materials are sold.
While our business is based predominantly on the refining and
upgrading of CTO and CST, as shown above, we have the capacity
to use both hydrocarbon-based raw materials and gum rosins where
appropriate and, accordingly, are able to offer tailored
solutions for our customers.
Our products and technical support enhance the value of our
customers’ products by improving their performance,
providing them with essential attributes, lowering costs and
simplifying processes. We have cultivated longstanding
relationships with leading customers in our key markets and have
a history of co-developing many of our products with our
customers to satisfy specific product requirements. Our
innovative products and solutions help our customers replace
non-renewable raw materials
103
with more sustainable alternatives. We serve approximately 1,000
customers in more than 80 countries through our worldwide
network of ten strategically located manufacturing facilities,
two laboratories and five representative offices.
Transactions with
Rhône Capital and International Paper
On February 28, 2007, Rhône Capital acquired a group
of operating companies that comprise Arizona Chemical from
International Paper. We refer to this transaction as the
“Acquisition”. Since the Acquisition, the companies
comprising Arizona Chemical have been subsidiaries of AZ Chem
Luxembourg Holdings S.à r.l., which, before giving effect
to the Reorganization described below, is also our direct parent
and a wholly owned subsidiary of AZ Chem Investments Partners LP.
Prior to the completion of this offering, the Rhône Funds
held a % limited partnership
interest in AZ Chem Investments Partners LP, while International
Paper held a % limited partnership
interest and AZ Chem MIV I Ltd and AZ Chem MIV II LP, which are
management incentive vehicles, or MIVs, formed to facilitate
investment in our company by our senior managers, collectively
held a % limited partnership
interest.
The Rhône Funds and International Paper are also members of
AZ Chem Investments LLC, the general partner of AZ Chem
Investments Partners LP. The Rhône Funds control AZ Chem
Investments LLC and therefore control us.
Corporate
Structure and Reorganization
On February 12, 2010, we were organized as a Bermuda
limited company.
Prior to the completion of this offering, AZ Chem Luxembourg
Holdings S.à r.l. will transfer all of the equity interests
in Arizona Chem Sweden Holdings AB, its wholly owned subsidiary,
to us, and we will transfer all of the equity interests in
Arizona Chem Sweden Holdings AB to Arizona Chemical Luxembourg
S.à.r.l., our wholly owned subsidiary. Following these
transfers, AZ Chem Luxembourg Holdings S.à r.l. will
dissolve, and we will be a direct, wholly owned subsidiary of AZ
Chem Investments Partners LP. Additionally, prior to the
completion of this offering, Arizona Chemical Ltd. will effect
a for
1 stock split. We refer to these transactions as the
“Reorganization”.
The historical financial information presented herein is that of
Arizona Chem Sweden Holdings AB and its predecessor prior to the
Acquisition, and following the Reorganization, all of our
business operations will be conducted through Arizona Chem
Sweden Holdings AB and its subsidiaries.
Our principal executive offices are located at 4600 Touchton
Road East, Building 100, Suite 1500, Jacksonville,
Florida 32246 and at Transistorstraat 16, 1322 CE Almere, The
Netherlands and our registered office in Bermuda is located at
Clarendon House, 2 Church Street, Hamilton, HM 11, Bermuda. We
can be reached by telephone at
(800) 526-5294
and +(31) 36 5462 800. Our corporate website address is
www.arizonachemical.com. We do not incorporate
information that is on, or is accessible through, our corporate
website into this prospectus, and you should not consider it a
part of this prospectus.
104
Below is an organizational chart illustrating our corporate
structure after completion of the Reorganization and before
giving effect to this offering.
105
Key Competitive
Strengths
The following competitive strengths underpin our ability to
create shareholder value by driving sustainable, profitable
top-line growth, improving margins and generating strong cash
flow:
Leading Market
Positions
We are the world’s leading supplier of pine-based chemicals
as measured by sales. According to ADL, we are the leading
global supplier of each of the following products:
|
|
|
| |
•
|
naturally-derived
resins used in adhesives, inks and roadmarking applications;
|
| |
| |
•
|
naturally-derived
tackifier resins used in hot melt packaging and bookbinding
adhesives;
|
| |
| |
•
|
naturally-derived
tread enhancement resins for passenger car tires; and
|
| |
| |
•
|
pine-based, non genetically-modified sterols used in food
ingredient and nutrient applications through our joint venture,
Arboris, LLC.
|
Scope and
Scale Provide Global Access
With manufacturing facilities in Finland, France, Germany,
Sweden, the United States and the United Kingdom, we have the
largest manufacturing capacity in the pine-based chemicals
sector. Our supply chain and commercial infrastructure gives us
access to global opportunities as we have the capability to
reliably service an increasingly international client base. We
believe we are the leading global refiner of CTO and the leading
global producer of TOFA and TOR.
Advantageous
Feedstock Position
We are the world’s largest buyer of CTO, and have the
capacity to refine approximately 650,000 metric tonnes of CTO
annually, which, according to ADL, represents approximately 40%
of global CTO refining capacity. We have the ability to process
a wide variety of CTO feedstocks, including lower quality CTO
and BLS, a precursor to CTO that we have the ability in the
United States to refine into CTO, providing us with access to a
broad source of raw materials. When we refer to CTO in this
prospectus, we are referring generally to CTO and BLS, together.
In addition, most of our manufacturing facilities are located in
close proximity to the facilities of our raw material suppliers,
allowing us to procure our raw materials at a low delivered
cost. Furthermore, we work directly with our suppliers at their
production facilities to enhance their CTO and CST yields
through technological improvements, which, in addition to
maximizing our raw material supplies, improves the efficiency of
our suppliers’ operations and enables us to foster strong,
long-lasting relationships with them.
We have long-term supply contracts with International Paper
pursuant to which they agreed to sell to us, and we agreed to
purchase from them, all of the CTO and CST produced at their
existing U.S. paper mills. We also have the option to
purchase all of the CTO and CST produced at International
Paper’s future paper mills worldwide. These contracts
provided us with approximately 20% and 27% of our global CTO and
CST purchases for the three months ended March 31, 2010 and
the year ended December 31, 2009, respectively. CTO and CST
accounted for approximately 68% and 64% of our raw material
purchases for the three months ended March 31, 2010 and the
year ended December 31, 2009, respectively. We also
maintain long-standing relationships with other major suppliers
in the United States and Europe.
Stable
Customer Base with Long-Standing Relationships and Diverse End
Markets
With approximately 1,000 customers in more than 80 countries, we
have a broad customer base covering many diverse end-user
industries, including adhesives, inks, tires and rubber, roads
and construction, consumer products, renewable energy, fuel
additives, coatings, lubricants and polymer additives. Our five
largest customers for each segment accounted for approximately
28%, 26% and 29% of our net sales for the North American segment
and approximately 23%, 25% and 29% of our
106
net sales for the European segment for the years ended
December 31, 2009, 2008 and 2007, respectively. No single
customer accounted for more than 10% of our net sales for the
years ended December 31, 2009, 2008 and 2007. We have strong,
long-standing relationships with our customers, and many of our
products have been developed in cooperation with our customers,
frequently in response to their specific needs. We believe our
customers also value our reliability and global franchise which,
in combination with our technical expertise, have allowed our
products to become critical components of our customers’
formulations and have garnered the customer loyalty that
underpins our long-standing relationships.
Commitment to
Innovation
We believe that innovation and new product development are
critical to meeting our customers’ needs and to generating
profitable future growth. We employ over 50 scientific
professionals, many of whom hold Ph.D. degrees. In the five
years ended December 31, 2009, we applied for 61
U.S. patents and 18 Patent Cooperation Treaty patents.
During 2009, we instituted a structured approach to growth with
dedicated cross-functional teams that are directed toward our
six target markets. As part of this approach, we identify and
develop new products based on our understanding of our
customers’ needs and market opportunities. We utilize a
portfolio management system to ensure that we are disciplined in
assessing potential opportunities for new products and to
allocate resources and capital in accordance with our strategic
objectives and priorities.
Our track record of product innovation extends more than
80 years. Examples include:
|
|
|
| |
•
|
adhesive tackifiers designed to enable the use of a higher
amount of recycled content in packaging materials;
|
| |
| |
•
|
high solid adhesive dispersions for labels and tapes that allow
for higher coating speeds and that lower process energy costs;
|
| |
| |
•
|
heat stable rheology, or HSR, ink resins that reduce formulation
complexity for ink manufacturers while improving press
performance;
|
| |
| |
•
|
tire tread resins that promote wet grip, fuel economy and tire
life;
|
| |
| |
•
|
fuel lubricity improvers that ensure low sulfur targets for
diesel fuel can be met; and
|
| |
| |
•
|
emulsions and clear gels for skin and sun care applications.
|
Improving
Profit Margins
Our market leading position, together with the importance of our
portfolio to our customer base, our value pricing and our
ability to shift production and manage costs have enabled us to
generate improving profit margins across diverse macroeconomic
environments. Despite rapidly changing raw material, energy and
freight costs in recent years, as well as the difficult economic
environment that affected the chemicals industry in late 2008
and into 2009, we have been successful at increasing our
Adjusted EBITDA and margins, as illustrated in the chart below.
107
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months
|
|
Three Months
|
|
|
|
|
|
Combined
|
|
|
|
Ended
|
|
Ended
|
|
Year Ended
|
|
Year Ended
|
|
Year Ended
|
|
|
|
March 31,
|
|
March 31,
|
|
December 31,
|
|
December 31,
|
|
December 31,
|
|
|
|
2010
|
|
2009
|
|
2009
|
|
2008
|
|
2007
|
|
|
|
|
|
|
|
|
|
|
|
(Non-GAAP)
|
|
|
|
(dollars in thousands)
|
|
|
|
Net income (loss)
|
|
$
|
9,997
|
|
|
$
|
(2,357
|
)
|
|
$
|
12,098
|
|
|
$
|
(26,559
|
)
|
|
$
|
(23,605
|
)
|
|
Gross Margins
|
|
|
20.6
|
%
|
|
|
8.1
|
%
|
|
|
15.7
|
%
|
|
|
13.3
|
%
|
|
|
11.7
|
%
|
|
Adjusted EBITDA
|
|
$
|
27,378
|
|
|
$
|
11,043
|
|
|
$
|
93,859
|
|
|
$
|
92,723
|
|
|
$
|
66,898
|
|
|
Adjusted EBITDA margins
|
|
|
13.8
|
%
|
|
|
6.2
|
%
|
|
|
12.2
|
%
|
|
|
9.3
|
%
|
|
|
7.8
|
%
|
For a definition of Adjusted EBITDA, a description of our use of
Adjusted EBITDA as a measure of operating performance and a
reconciliation of Adjusted EBITDA to net income, as well as an
explanation of our use of combined financial information for the
year ended December 31, 2007, see “Summary Selected
Historical Consolidated Financial Information and Other
Data”.
Experienced
and Proven Management Team with Significant Equity
Interest
We have a highly motivated management team with an average of
more than 25 years of experience, combining a core of
pine-based chemical veterans with specialty chemicals experts
from outside the pine chemicals industry. Our management team
has instilled a shareholder-value-based culture throughout our
organization, successfully implemented leading productivity
practices and reinvigorated new product development. Through the
MIVs, our management has a significant investment in AZ Chem
Investments Partners LP, our parent.
Ability to
Satisfy Increasing Demand for Sustainable Raw
Materials
Our products are based on naturally-derived, renewable raw
materials and, as such, we are well placed to benefit from
consumer-driven demand for sustainable alternatives to existing
hydrocarbon-based products. In conjunction with our focus on
innovation, we expect to leverage the sustainable
characteristics of our product range in developing new market
opportunities for our products. For example, we expect that the
sustainable nature of our gellants and other personal care
products will be an increasingly attractive feature in the skin
care and cosmetics markets.
Our Business
Strategy
Building on our competitive strengths, set forth below are the
four key elements to our business strategy.
Organic Growth
Through Market Understanding and Development
Building upon our strength in technological innovation, we focus
our research and development efforts on opportunities that
address the needs of our customers as well as enhance our
profitability and growth profile. We look for opportunities in
new applications, new products and new markets. For example, we
are developing new personal care applications for our polymeric
gellant technology that we believe present improved margin
opportunities. Additionally, our HSR ink resin products provide
better performance at a lower cost relative to products
historically used in
“sheet-fed”
printing applications, and we have begun to market our HSR ink
resin products for use in these applications, which we believe
also offer us improved margin opportunities. Other innovations
include extra-light color rosin esters for adhesive packaging
applications, high solid adhesive dispersions and tire tread
enhancement additives. In relation to new market opportunities,
we are presently developing compatibilizers for modified bitumen
applications, which will enable customers to produce stable,
long-
108
lasting synthetic rubber modified bitumen with higher
sustainable material content. Products we have developed in the
last five years generated approximately 10% of our net sales in
2009.
During 2009, we spent approximately 1% of our annual net sales
on research and development efforts, and we plan to double our
research and development and marketing resources and related
spending within the next three years. In addition to internal
research and development, we expect to expand our product
offerings through select licensing
and/or
purchasing of new formulations, technology and products from
third parties, and we intend to fund select capital expenditure
opportunities where we can expand capacity in high growth, high
margin areas, or reduce costs, for an appropriate return on
investment.
Geographical
Expansion into BRIC Countries
We expect to continue to serve our existing global customers as
they expand their businesses in emerging countries, such as
Brazil, Russia, India and China and to target new customers in
these regions. In 2009, 2.7% of our net sales were generated in
these four countries, and we believe there are significant
opportunities to expand our business in these and other emerging
economies. We plan to manage our expansion into emerging markets
by adding sales and marketing, technical service and other
development related personnel, followed by further
infrastructure expansion as our sales opportunities in these
markets increase.
Active
Management of our Business and Asset Portfolio
We plan to accelerate profitable growth through selected
acquisitions of companies
and/or
assets that focus on our six target markets or on high value
niche applications and that complement our current product
offerings and capabilities. For example, in February 2009, we
acquired Abieta Chemie GmbH, which we refer to as
“Abieta”, a company located in Gersthofen, Germany
that specializes in natural resin products. This acquisition
strengthened our position in the tire and rubber market and
secured access for us to new products and technology that can be
leveraged in other strategic markets.
Productivity
Improvement Program
We have a history of implementing programs that have driven
significant costs out of our business. We expect to continue to
expand our successful productivity improvement initiatives by
pursuing operational efficiencies, optimizing available
technologies, maintaining a lean organizational structure,
further reducing fixed costs, capitalizing on our global
procurement organization, rationalizing capacity and efficiently
managing capital spending. We intend to take advantage of the
enhanced flow of information from the implementation of a new
SAP global enterprise resource planning system, which we
completed in the first half of 2010, to further our efforts to
improve our margins, drive cost productivity and generate
additional cost savings.
109
Our
Markets
The following chart presents the percentage of our net sales
attributable to our six target markets and our portfolio of
chemical intermediaries.
Arizona
Chemical’s 2009 Net Sales $767.5 million
Adhesives
We are a leading global supplier of tackifiers to the adhesives
industry as measured by sales and the world’s largest
producer of tackifier resins from renewable resources in terms
of volume. According to ADL, our share of the approximately
$1.6 billion global tackifiers market in 2009 was
approximately 12%. Approximately 59% of our adhesives products
are sold into the packaging
sub-market,
while another 16% are sold for use in wood and furniture and
assembly adhesives. Our products are also used in bookbinding,
hygiene, non-woven,
pressure-sensitive
and transportation adhesives.
We currently offer a broad range of products to service our
target adhesives
sub-markets,
including rosin-based tackifiers for packaging and
pressure-sensitive
adhesive applications, terpene-based tackifiers for bookbinding,
hygiene and
pressure-sensitive
adhesive applications, AMS tackifiers for bookbinding and
pressure-sensitive
adhesive applications and hot melt polyamides for flexible
packaging.
Despite the relative maturity of the adhesives market in
developed regions, we see new opportunities for growth in
particular
sub-markets.
In
pressure-sensitive
adhesives used for labels and tapes, hydrocarbon tackifiers are
the leading technology today due to their compatibility with the
polymers used in these systems. However, hydrocarbon refiners,
particularly in the United States, have increasingly shifted
their production away from liquid feedstocks such as naphtha
toward gas feedstocks to improve margins. This feedstock shift
has reduced the availability of hydrocarbon tackifier products,
as resin formers required for the production of hydrocarbon
tackifiers are generally derived from the liquid feedstocks.
This shift is driving customers to seek more sustainable
solutions such as our naturally-derived
SYLVATAC®,
SYLVALITE®
and
SYLVARES®
tackifiers. We are also targeting growth in water-based
applications with our
AQUATAC®
rosin ester dispersions. Within our
AQUATAC®
product range, we have developed thicker rosin ester
dispersions, meaning they have lower water content, which allows
our customers to run their coating machines more economically as
less energy is required to extract excess water from the final
adhesive.
In the hygiene
sub-market,
adhesives are used for non-woven applications such as diapers.
At present, high-priced hydrocarbon tackifiers that are odorless
and clear are the prevailing technology. We presently compete in
this market with our low color and low odor styrenated
terpene-based products, which we sell under the
SYLVARES®
and
ZONATAC®
trade names. However, with burgeoning demand for diapers in
lower income markets, diaper producers are re-engineering their
products to reduce cost. We have developed a proprietary rosin
ester based tackifier and related process
110
technology which we believe address odor and color concerns
while also meeting our customers’ cost targets.
Our tackifiers are primarily used in hot melt adhesives, which
are heavily used in the packaging sub-market. Building on the
strength of our current business, we are focused on identifying
high value applications in the packaging market that capitalize
on our expertise in developing new products that facilitate
bonding between surfaces that do not adhere easily. Further, to
address requirements in the flexible packaging market, we are in
the process of developing tackifiers for use in flexible
packaging that can withstand freezer and cooking temperatures
and that seal tightly to create an oxygen barrier. Our expertise
in adhesives and inks is a competitive advantage in developing
flexible packaging solutions.
Across all adhesive
sub-markets,
our products allow our customers to significantly improve the
sustainability of their adhesive systems and reduce the impact
of their products on the environment, while maintaining the
performance the industry demands. In the construction market, we
believe that the trend toward “green” buildings,
together with a legislative push for the reduction of volatile
organic content, or VOC, and improved air quality, is likely to
create further opportunities for our tackifiers. Our tackifiers
are particularly effective in hot melt adhesives, which are a
low VOC alternative to high VOC solvent-based adhesives.
As our customers expand into developing regions, they are
seeking local support from suppliers with whom they have had a
long and successful relationship. We are strategically deploying
resources to these regions using either internal resources,
co-supplier relationships, partnerships, joint ventures or
acquisitions.
Approximately 25%, 24% and 27% of our net sales for the years
ended December 31, 2009, 2008 and 2007, respectively, were
from products sold into the adhesives market. These products
include:
Rosin-Based Tackifiers. Our
SYLVATAC®
and
SYLVALITE®
rosin ester tackifiers are particularly effective in hot melt
adhesives and for bonding surfaces that are difficult to bond,
such as highly-recycled packaging materials. We also offer a
range of rosin ester dispersions under the
AQUATAC®
trade name, for use in water-based adhesive applications such as
paper labels.
Terpene-Based Tackifiers. Our range of
terpene phenol, styrenated terpene and polyterpene resins, which
we market under the
SYLVARES®
and
ZONATAC®
trade names, offer broad polymer compatibility, which allows our
customers to develop and manufacture adhesives that bond to a
wide variety of plastic surfaces. Our products are also used
because they offer certain “in-service” performance
requirements such as low odor, clear color, heat and chemical
resistance, and can be applied at low temperatures.
AMS (Pure Monomer)
Tackifiers. Supplementing our range of
tackifiers based on renewable resources are tackifiers based on
alpha-methyl-styrene, or AMS, a hydrocarbon. Our tackifiers
based on this monomer, which we also market under the
SYLVARES®
trade name, are almost water-white and adhere well to difficult
to bond materials. There are only two other significant
producers of AMS tackifiers and our range of AMS phenolics,
which are used in high-end applications, is unique.
Hot Melt Polyamides. Our
UNI-REZ®
thermoplastic polyamide adhesives are distinct within our range
of offerings in that they are finished, end-use adhesives,
rather than tackifiers. They bond to a wide variety of
substrates and offer outstanding resistance to chemicals and
oils.
Inks
We are a major supplier of ink resins for use in publication and
packaging to many of the world’s leading printing ink
companies. According to ADL, we had approximately 6% of the
approximately $1.5 billion global ink resin binders market
in 2009.
111
The use of electronic media in preference to paper is rising,
which is putting negative pressure on the publication
sub-market,
while the use of packaging continues to grow due to rising
incomes and globalization. In that context, our strategy is to
maintain our traditional lithographic and gravure ink resin
business in the publication sub-market under our
SYLVAPRINT®
trade name, while growing our presence in the flexible packaging
sub-market,
where we currently produce polyamide ink resins under the
UNI-REZ®
trade name.
We are working to develop new technologies and new pine-based
chemistries to address the changing requirements for flexible
packaging materials, including higher regulatory requirements
associated with food contact legislation and the desire to move
to more renewable and recyclable materials. We believe that we
can bring together our portfolio of sustainable chemistries, our
knowledge of both inks and adhesives, and our technical
understanding to develop new products for this market. We have a
dedicated team charged with defining and developing
opportunities in flexible packaging.
In the publication
sub-market,
our focus is on leveraging our proprietary SYLVAPRINT
HSR®
technology, which provides enhanced ink performance in
lithographic printing. This product improves the ability of ink
to adhere to paper, which reduces the need for gellants and
therefore lowers costs for our customers. We are working to
promote acceptance of this new technology both with customers,
and with their customers.
Approximately 11%, 12% and 15% of our net sales for the years
ended December 31, 2009, 2008 and 2007, respectively, were
from products sold into the inks market. These products include:
Phenolic Rosin Esters. Our range of
SYLVAPRINT®
phenolic rosin esters are used in lithographic (offset) and
publication gravure inks and confer beneficial attributes such
as enhanced adhesion, high gloss, improved drying speed,
viscosity and color intensity to our customers’ ink
products. We also offer these resins pre-mixed in mineral oil to
save our customers time and reduce their energy usage.
Solution Metal
Resinates. SYLVAPRINT®
solution metal resinates are used in the high volume commercial
printing market. These resins are used in inks for publication
gravure, which is used for high quality, large print run
applications such as catalogs and magazines.
Polyamides. UNI-REZ®
ink polyamides are resins used in inks for flexible packaging
such as bread bags, shrink sleeve labels, high end lamination
and snack food packaging.
Ink
Solvents. SYLVAPRINT®
ink solvents are esters of fatty acids and are used primarily in
specialty applications such as vegetable oil-based
“sheet-fed” printing to improve the solvency power of
the vegetable oils, such as those derived from soy or linseed.
Tires and
Rubber
We are a leading supplier of tread enhancement resins and
additives from renewable resources to the global tires and
rubber industry as measured by sales, and, according to ADL, we
had approximately 34% of the approximately $80 million
global tire tread enhancement additive market in 2009.
We have grown our business in tire tread enhancement additives
over the last five years under our
SYLVARES®
trade name. Tire tread enhancement additives seek to improve
tire life, fuel economy and grip. Our current sales are based on
our first generation tread enhancement technology, which is
produced using both renewable resources (polyterpenes based on
D-limonene) and hydrocarbons (AMS pure monomer resins). We have
also developed second generation resin technology for which we
own the intellectual property rights and have filed a patent
application. Through this technology, we intend to capture an
increased share of the growing market and expand our global
customer base.
112
We are also developing rosin-based tackifiers to improve the
manufacturing efficiency of tires during the assembly stage. We
have filed a patent application for this product and expect to
achieve our first sales in the first half of 2010.
In the rubber market, our acquisition of Abieta Chemie GmbH in
February 2009 has expanded our geographical presence and made us
the leading supplier of rosin soap-based emulsifiers, which we
sell under the
ABIETA®
and
SYLVAROS®
trade names. These products are used as process control agents
for the polymerization of unsaturated hydrocarbons in the
manufacture of synthetic rubber, which are used in the
manufacture of tires, as well as industrial rubber applications
such as conveyor belts and the manufacture of polymers used in
the automotive industry for vehicle interiors.
Approximately 8%, 4% and 4% of our net sales for the years ended
December 31, 2009, 2008 and 2007, respectively, were from
products sold into the tires and rubber market. These products
include:
Polyterpenes. SYLVARES®
polyterpenes are used as tire tread enhancement additives to
improve the combination of wet grip, fuel economy and tire life.
They are based primarily on D-limonene, a co-product of citrus
fruit farming.
Disproportionated and Non-Disproportionated Rosins
Soaps. Our
SYLVAROS®
rosin soaps products, which we have substantially expanded
following our acquisition of Abieta, are used as emulsifiers in
rubber polymerization. We use both tall oil and gum rosin in the
manufacture of these products.
AMS
Resins. SYLVARES®
AMS resins are tire tread enhancement additives which improve
wet grip, fuel economy and tire life. They are based on AMS, a
hydrocarbon.
Roads and
Construction
We are a major supplier of thermoplastic roadmarking resins in
the United States, Europe and the Middle East and, according to
ADL, we had an estimated 6% share of the approximately
$600 million global market for thermoplastic roadmarking
resins in 2009. We believe there are future opportunities to
supply fatty acids and tackifier resins for bitumen applications
in roofing and paving.
Our current presence is predominantly in the roadmarking
sub-market
where we produce rosin-based binders for thermoplastic coatings
under the
SYLVATAC®
and
SYLVACOAT®
trade names. However, we are also exploring growth opportunities
in the paving and roofing
sub-markets
where consistent with our business strategy.
In roadmarking, we have successfully grown our business as the
use of thermoplastic coatings has increased. Thermoplastic
coatings offer extended service life compared to alternatives
such as paint, and are increasingly used in the growing
“anti-skid” surface market for speed abatement.
Further they offer improved retro-reflectivity which promotes
road safety. Rosin-based binders have taken a larger share of
the growth in thermoplastic coatings, as their availability and
price have been more stable than hydrocarbon binders.
Our chemistries have a high affinity to rubber and a strong
similarity to bitumen. We are seeking to leverage our
understanding of this technology to develop new products for the
paving and roofing
sub-markets.
For instance, in paving, we are developing bitumen
compatibilizers. Demand for compatibilizers is growing due to
the declining quality of bitumen feedstocks and the need to
augment performance through the addition of polymers such as
synthetic rubber.
Another growth initiative is in crumb rubber modified paving. We
have identified a strong need to stabilize crumb rubber blends
for a longer period of time to enable even more widespread
usage. We are currently developing a compatibilizer system which
will allow our customers to create a paving mix at central
terminals for transportation, rather than mixing products at the
paving location, and have identified customers for co-product
development during 2010.
113
We are also developing new products for the bitumen roofing
sub-market. As “Peel and Stick” technology continues
to penetrate the market, there is an opportunity to develop
adhesives which have an improved ability to bond with humid
substrates and which can be applied at lower temperatures. Our
objective is to develop an effective adhesive system together
with selected customers that will utilize our tackifier
technology.
The use of renewable materials such as pine chemicals is also a
significant driver for further growth. We are planning to
increase our marketing efforts in order to promote the
environmentally sustainable nature of our products to national
highway agencies, municipalities, roadmarking manufacturers and
contractors.
Approximately 6%, 5% and 4% of our net sales for the years ended
December 31, 2009, 2008 and 2007, respectively, were from
products sold into the roads and construction market. These
products include:
Rosin Esters and Insoluble Maleics. We
are the leading supplier of rosin derivatives used in
thermoplastic pavement markings, which we sell under the
SYLVATAC®
and
SYLVACOAT®
trade names. Our products improve reflectivity and durability of
the coating through their strong adhesion to glass beads, while
imparting excellent resistance to oil and gasoline.
Hot Melt Polyamides. Hot melt
polyamides are utilized in pre-formed pavement graphics. Hot
melt polyamides are higher value products, which offer ease of
application and durability to formulators.
Consumer
Products
In the consumer products market we sell a diverse range of raw
materials and ingredients for the formulation of consumer
products used in the personal care, home care, industrial
cleaning and food ingredients sub-markets.
Approximately 5%, 3% and 4% of our net sales for the years ended
December 31, 2009, 2008 and 2007, respectively, were from
products sold into the consumer products market. These products
include:
Alpha Pinene and Beta Pinene. Sold
under the
SYLVAPINE®
trade name, our pinene products are used as building blocks in
the manufacture of fragrances, camphor, perfumes, terpineol,
terpene resin derivatives and insecticides. They are also used
in the institutional and industrial market as cleaners, solvents
and disinfectants.
Sterols. Sterols are high margin
pharmaceutical and food additives used to reduce cholesterol
absorption and are produced through the upgrading of pitch.
Unlike soy-based sterols with which they compete, pine-based
sterols are not considered a genetically modified product. We
have a 10% interest in Arboris, LLC, a joint venture that
manufactures sterols from pitch that we supply to it. We have
the right to acquire an additional 30% interest in Arboris.
Specialty Polymeric Gellants. Specialty
polymeric gellants are bio-renewable, polyamide polymers used to
impart structure, rheology, film forming and wear resistance to
a variety of products formulated for the personal care and
consumer products markets. They are sold under the
SYLVACLEAR®,
SYLVAGEL®
and
UNICLEAR®
trade names. Personal care applications include the enhancement
of gloss and wear for mascara, gel bases for lipsticks,
waterproofing agents and film formers in lotions and SPF
boosters for sunscreens. In home care applications they are used
to solidify fragrances for lavatory rim blocks and small space
air fresheners, gel mineral oil in clear candles and to impart a
gel rheology in retail degreasers.
Immobilized Functional Oils
(IFO). Immobilized functional oil, or IFO,
technology allows a wide range of organic liquids to be
solidified into temperature resistant, robust, three dimensional
objects. This new technology is currently being sold in the
automotive air freshener market in the United States and Europe.
It is under customer evaluation for use in applications as
diverse as
114
pesticide delivery through collars and tags for household and
farm animals, laundry care fragrance retention and as a delivery
system for the active ingredients in sun and skin care products.
Renewable
Energy
As a result of governmental mandated targets for the reduction
of greenhouse gas emissions and dependency on fossil fuels, and
the incentives legislatures have implemented or which we expect
legislatures will implement to reach these targets, we expect
that demand for renewable sources of energy will continue to
increase in line with these governmental mandated targets. The
principal legislative initiatives that we expect to impact
demand for renewable sources of energy are the European
Union’s Directive on the Promotion and Use of Energy from
Renewable Resources in Europe and the 2005 Energy Policy Act in
the United States.
For a discussion of risks legislation regarding renewable energy
poses to our business, see “Risk Factors — Risks
Related to Raw Materials”.
Approximately 10%, 12% and 8% of our net sales for the years
ended December 31, 2009, 2008 and 2007, respectively, were
from products sold into the renewable energy market. These
products include:
Pitch. Our pitch by-product is a second
generation, cellulosic biofuel that is used in municipal heating
and industrial power generation. We principally sell pitch into
the European biofuel market, where incentives for the use of
renewable sources of energy are more developed than in the
United States. As described below under
“— Sources and Availability of Raw
Materials”, we also maintain agreements with our raw
material suppliers in Europe that enable us to exchange our
pitch by-product for CTO.
TOFA. In 2009, we began selling TOFA, a
co-product of the CTO refining process, to a third party for
conversion into second generation, cellulosic biodiesel for use
as a transportation fuel.
Chemical
Intermediates
We produce a portfolio of pine-based chemical intermediates that
are sold into markets as diverse as paints and coatings,
lubricants, fuel additives, mining and oilfield, paper chemicals
and polymer additives. Under the trade names of
SYLFAT®,
SYLVATAL®,
UNIDYME®
and
CENTURY®,
our bio-refinery products are utilized in these markets as key
components in many finished goods.
Approximately 35%, 39% and 38% of our net sales for the years
ended December 31, 2009, 2008 and 2007, respectively, were
from the sale of chemical intermediates. These products include:
Tall Oil Rosins
Our
SYLVAROS®
tall oil rosins are used in all major rosin applications for the
manufacture of resins for adhesives, inks and roadmarking,
emulsifiers for rubber, size for paper and chewing gum.
Tall Oil Fatty Acid (TOFA)
According to ADL, our
SYLFAT®
tall oil fatty acids represent approximately 34% of the
approximately 500,000 metric tonne global supply of TOFA.
|
|
|
| |
•
|
TOFA is a key component in additives to improve the lubricity of
low-sulfur diesel fuel, preventing engine fuel pump wear.
|
| |
| |
•
|
TOFA enables producers of phosphate and phosphoric acid to run
their plants more profitably through use as a flotation reagent
in apatite mining.
|
| |
| |
•
|
TOFA is a component in performance additives to aid the heat
resistance of PVC.
|
| |
| |
•
|
TOFA is used in alkyd paints, primarily for decorative coatings,
helping to bring durability and gloss.
|
115
Distilled Tall Oil (DTO)
According to ADL, our
SYLVATAL®
distilled tall oils account for approximately 24% of the
approximately 70,000 metric tonne global market for DTO. DTO is
primarily used as an emulsifier for metalworking fluids, where
in many cases it replaces less environmentally friendly
hydrocarbon-based chemicals.
Dimer Acids
Our
UNIDYME®
dimer acids are used for the production of polyamide resins for
epoxy coatings, flexographic inks, and high performance adhesive
applications. In addition, dimer acids are building blocks in
the production of corrosion inhibitors and emulsifiers for the
production and recovery of petroleum and natural gas. According
to ADL, we had approximately 19% of the approximately 170,000
metric tonne market for dimer acids in 2009, considering both
captive and merchant consumption. Major competitors in this area
include producers of dimer acid from other feedstocks such as
rapeseed and cottonseed oil, and other producers of TOFA-based
dimers. We are the only producer of dimer acids that is both
backward-integrated to CTO and forward-integrated to polyamides.
Curing Agents
Our
UNI-REZ®
polyamide curing agents are used to cure (harden) epoxy resins,
primarily those used in marine and protective coatings. They are
based on TOFA-derived dimer.
Sources and
Availability of Raw Materials
We use three primary raw materials in our operations: CTO, CST
and D-limonene, which together represented 68% of our total raw
material purchases for 2009. When we refer to CTO in this
prospectus, we are generally referring to CTO and BLS, a
precursor to CTO that we have the ability in the United States
to upgrade into CTO, together. Other raw materials used in our
production processes include a variety of organic and inorganic
chemical intermediates, including pentaerythritol, ethylene
amines, phenol, alpha-methyl-styrene and gum rosin.
CTO and
CST
CTO and CST are co-products of the kraft pulping process used
for softwood paper manufacturing. CTO and CST represented 68%
and 64% of our total raw material purchases for the three months
ended March 31, 2010 and the year ended December 31,
2009, respectively. The majority of the world’s CTO and CST
supply is located in North America and Europe, though we expect
that additional supply will develop in regions such as Russia
and South America. Substantially all of the global CTO and CST
supply is consumed by the producers of CTO and CST or sold
pursuant to long-term supply agreements, and CTO and CST are not
generally available for purchase on the open market.
In connection with the Acquisition, we entered into long-term
CTO and CST supply contracts pursuant to which International
Paper agreed to sell to us, and we agreed to purchase from them,
100% of the CTO and CST produced at their U.S. paper mills.
We also have the option to acquire CTO and CST produced at
future International Paper paper mills worldwide. The purchase
price for CTO is determined by reference to the price of natural
gas and fuel oil, while the purchase price for CST is fixed,
subject to adjustment every three years based on changes in
market price. These contracts provided us with approximately 20%
and 27% of our global CTO and CST purchases for the three months
ended March 31, 2010 and the year ended December 31,
2009, respectively. These contracts are terminable by either
party on February 28, 2027 and each anniversary thereafter,
subject to a five-year notice requirement.
We also satisfy our CTO and CST requirements through short-term
contracts with other major pulp and paper manufacturers in the
United States, Europe and Russia. Additionally, CTO, CST and
pitch, a by-product of the CTO refining process, possess
inherent fuel properties and can be burned
116
by mills for use in their operations. We maintain agreements
with pulp and paper manufacturers in Europe that enable us to
exchange our pitch by-products for their CTO. These agreements
enable us to both maximize our CTO supply and sell our pitch
by-products,
while enabling our suppliers to ensure that they have adequate
supplies of renewable fuel for their mills.
CTO prices in Europe are directly correlated with oil prices on
a three month lag, with contract prices based on the average oil
price for the previous quarter. During the 2006 to 2008 period,
CTO prices in Europe steadily increased along with oil prices.
Brent Crude prices averaged $65 per barrel in 2006, $72 in
2007 and $98 in 2008 after having peaked at $135 in July 2008
and ending the year at $44. The significant decline in oil
prices during the fourth quarter of 2008 reduced CTO prices in
the first quarter of 2009, and CTO prices continued to decline
through June 2009. The average Brent Crude price for 2009 was
$62 per barrel. European CTO prices are also impacted by the
Euro to U.S. dollar exchange rate relevant to its impact on oil
price, as well as carbon dioxide emission trading fees and
supply and demand. During the third and fourth quarters of 2009,
average Brent crude oil prices increased 16% and 9%,
respectively, from the prior quarter. Average CTO prices in
Europe during the first quarter of 2010 were 7% higher than
during the fourth quarter of 2009.
CTO prices in North America are driven by a mixed basket of
energy prices including oil, natural gas and coal, as well as
supply and demand. Energy price changes impact North American
CTO prices on a longer lag than in Europe, averaging between
three and six months depending on the supplier mix. During 2006,
North America CTO prices increased consistently with higher
energy costs, and remained relatively stable through 2008,
despite higher energy prices, as a result of management of
supply mix. During the first half of 2009, our prices declined
along with energy prices and we experienced lower demand.
However, during the second half of 2009, natural gas prices
increased 31% as compared to the first half of 2009, and average
CTO prices in North America during the first quarter of 2010
were 8% higher than during the fourth quarter of 2009.
We believe we are currently well positioned to acquire the CTO
and CST required for our operations.
For information regarding risks associated with our supply of
CTO and CST, see “Risk Factors — Risks
Related to Raw Materials”.
D-limonene
D-limonene is a by-product of the citrus industry and
represented 4% of our total raw material purchases in 2009. We
satisfy the majority of our D-limonene requirements through
purchases from several producers in Brazil, and have the ability
to supplement this supply with D-limonene from Florida.
The supply of D-limonene may be affected by seasonal changes and
weather, such as hurricanes or other disruptive weather
patterns. If the supply of D-limonene is reduced as a result of
such a disruption, the price of D-limonene will generally
increase as a result of the reduced supply.
Competition
We compete with a number of other pine chemical manufacturers,
including refiners of CTO and CST, such as Meadwestvaco
Corporation and Georgia-Pacific LLC in North America and Forchem
Oy and Les Dérivés Résiniques et Terpéniques
in Europe and companies that process gum rosin, including Hexion
Specialty Chemicals, Inc. Depending on the market into which we
are selling, we also compete with companies that refine and
upgrade hydrocarbons and vegetable oil. Our hydrocarbon-based
competitors include ExxonMobil and Eastman, and our vegetable
oil-based competitors include Cognis GmbH, Croda International
Plc, Oleon N.V. and Procter & Gamble, Chemicals
Division.
117
In the adhesives and inks markets, our TOR-based resin products
compete against gum-rosin and hydrocarbon-based resins. In
particular, several of our hydrocarbon-based competitors produce
metallocene polymers that directly compete with our tackifier
resins for use in hot melt packaging adhesives.
In the tires and rubber market, where we market our
SYLVARES®
tire tread enhancement additives that optimize wet grip
performance, fuel economy and tire life, several of our
competitors offer hydrocarbon and terpene-based alternatives.
In each of our target markets, we compete on the basis of a
range of factors, including product quality, speed of service
from order to delivery, breadth of product availability,
technical support and price. We also believe our customers base
their decisions on a supplier’s ability to design and
produce customized products (an area which we will increasingly
focus on as we execute our business strategy to develop higher
margin, higher value products targeted at specific customer
needs) and the environmentally sustainable nature of pine
chemicals compared to hydrocarbon-based alternatives.
Seasonality
Historically, our business has been subject to seasonal
fluctuations of raw material inventory, due to the seasonal
trends in availability of CTO and CST. Yields of CTO and CST are
higher during the first half of the year due to the natural
growth and associated chemical yield cycles of trees in addition
to higher yields from kraft pulping during the cooler months.
CTO and CST receipts rise during the first and second quarters,
generally peaking during the early summer. In addition our
business has seasonal fluctuations associated with customer
demand. In preparation for stronger demand in our markets during
the second and third quarters of the year, we build inventory of
finished goods which generally peaks in the second quarter. Many
factors drive the increase in demand in the second and third
quarters, including the holiday printing season (affecting our
customers in the inks market) and the seasonality of the
roadmarking and construction industries, which see increased
activity in warmer months. As a result, demand and consequently
sales have been historically lower in our first and fourth
quarters.
Marketing,
Research and Development and Sales
Our marketing, research and development, and sales functions are
organized to optimize our ability to develop new products and
new applications based on a thorough and informed understanding
of our markets and the chemistries used in our business. We have
established cross-functional, market focused teams, the core of
which are members of our research and development and marketing
functions, supplemented by additional members from other
functions such as sales, manufacturing and finance. The team
members interview our customers and prospective customers to
understand their needs and, by targeting our development of new
products on these requirements, we create mutual value. By
maintaining an ongoing dialogue with our clients throughout the
product development cycle, our cross-functional teams enable us
to focus our development efforts on higher value products and
applications.
We currently have six cross-functional teams, one working in
each of our six target markets: adhesives, inks (for flexible
packaging), tires and rubber, roads and construction, consumer
products and renewable energy.
Marketing
Our marketing team is charged with maintaining our leadership
position within existing markets and profitably expanding our
presence in new markets. Our cross-functional teams are
responsible for identifying unmet customer needs in our target
markets, and our marketing personnel work directly with our
customers and our research and development personnel to develop
products and applications to meet the needs identified.
118
We have recently hired a director of strategic planning and
growth to support our cross-functional teams and to drive these
efforts.
Research and
Development
We primarily conduct our research and development activities in
laboratories at our facilities located in Savannah, Georgia, and
Almere, The Netherlands.
Through the open and ongoing dialogue our cross-functional teams
maintain with our customers, we are able to ensure that our
resources are optimally utilized. We use a portfolio management
system and the
Stagegate®
process to ensure that priority is given to the highest return
projects, that we maintain an optimal mix of long and short term
projects and that we appropriately balance the risk profiles of
these projects. Our research and development personnel develop
new products in response to customer needs and provide technical
service to our customers, which enhances customer relations.
Additionally, we employ process engineers who work with our
research and development personnel to safely and efficiently
transition products from the development phase to full-scale
manufacturing, and who assist in the identification and
development of new process technology and approaches. We
maintain a pilot plant at our Savannah, Georgia facility to
provide production scaling and to evaluate manufacturing
capability during the transition from research to production.
We employ over 50 scientific professionals, many of whom hold
Ph.D. degrees, who are dedicated to our research and development
function. Over the past five years we have applied for 61
U.S. and 18 Patent Cooperation Treaty patents. Products
that we have developed in the last five years generated
approximately 10% of our net sales in 2009.
We spent $3.5 million, $4.0 million and
$4.4 million for research and development for the years
ended December 31, 2009, 2008 and 2007, respectively. We
plan to double our research and development and marketing
resources and related spending within the next three years to
enable us to execute our growth strategy through the development
of new products and applications.
Sales
Our sales network consists of a direct sales force covering 13
countries and a network of distributors and agents in over 50
countries. Our sales personnel are primarily responsible for
maintaining our relationship with our customer base and
coordinating contact between our customers and our research and
development and marketing functions. We use third-party
distributors where distributors have existing platforms and the
use of distributors is cost effective.
Our direct sales force, which was responsible for 93% of net
sales in 2009, is highly developed in North America and Europe.
Numbering 20 globally, our sales account managers have a strong
practical knowledge of the markets they serve with an average of
more than 10 years of experience in the relevant industry.
The technical knowledge of our sales force and their close
alignment with our research and development and marketing
functions allow us to provide product and service options to
specifically meet customers’ individual requirements.
We maintain representative offices in Miami, Florida,
San Juan del Rio, Mexico, Singapore, Shanghai,
People’s Republic of China and St. Petersburg, Russia.
We maintain an extensive network of distributors to serve our
customers in Latin America and the Asia Pacific region. We are
in the process of consolidating our distributor network to
create a more effective and efficient channel to manage certain
of our existing clients and products and to enable our direct
sales personnel to focus on developing the high growth
businesses we are targeting.
In 2009, approximately 42% of our net sales were to customers in
the United States and Canada, 49% to customers in Europe, the
Middle East and Africa, 5% to customers in Asia and 4% to
customers in Latin America. See Note 19 of our consolidated
financial statements for information regarding our North America
and Europe segments and for geographic reporting for the years
ended
119
December 31, 2009 and 2008 and for the ten-month and
two-month periods ended December 31, 2007 and
February 28, 2007, respectively.
Patents,
Trademarks, Copyrights and Other Intellectual Property
Rights
We rely on a variety of U.S. and non-U.S., registered and
unregistered, intellectual property rights in the conduct of our
business, including patents, trademarks and trade secrets. As of
March 31, 2010, we held 354 granted patents (consisting of
96 U.S. granted patents and
258 non-U.S.
granted patents) and had 128 patent applications pending
worldwide. Patents are generally in effect for a period of
20 years after the filing date, and therefore, assuming
most of these applications will be granted, a significant
portion of our patent portfolio is expected to remain in effect
for a long period. The issued patents and the patent
applications have been issued and applied for, respectively, in
the United States and other countries. In the five years ended
December 31, 2009, we filed 61 patent applications in the
United States and 18 Patent Cooperation Treaty patent
applications. We do not expect that the expiration of any single
patent or specific group of patents would have a material impact
on our business. Our material trademarks are expected to remain
in effect unless we decide to abandon any of them, subject to
possible third-party claims challenging our rights. Similarly,
our trade secrets are expected to preserve their status as such
for as long as they are the subject of reasonable efforts, on
our part, to maintain their secrecy.
As a result of developing products and markets with selected
customers, we have six patent families that are jointly-held
with certain of our customers. These patents are in the areas of
tackifiers for adhesives and specialty polyamides for fragrance
formulations. At the time of such product and/or market
development, we entered into written agreements with these
customers that limit the rights of each party with respect to
jointly developed inventions and inventions made with the
confidential information of the other party. These written
agreements were made in exchange for exclusive licenses or
supply agreements, for limited times.
As a general matter, our trade names are protected by trademark
laws. We maintain a number of trademarks, including
“AQUATAC®”,
“CENTURY®”,
“SYLVABLEND®”,
“SYLVACLEAR®”,
“SYLVACOTE®”,
“SYLFAT®”,
“SYLVAPINE®”,
“SYLVAPRINT®”,
“SYLVARES®”,
“SYLVAROS®”,
“SYLVATAL®”,
“UNIDYME®”,
“UNIFLEX®”,
“UNI-REZ®”
and
“ZONATAC®”,
which are registered in the United States and in other countries.
We have accumulated a substantial amount of technical and
business expertise. Our expertise includes product development,
design and formulation, information relating to the applications
in which our products are used, process and manufacturing
technology, including the process and design information used in
the operation, maintenance and debottlenecking of our
manufacturing facilities, and the technical service that we
provide to our customers. We hold extensive discussions with
customers and potential customers to define their market needs
and product application opportunities. Where necessary, we have
implemented trade secret protection for our technical knowledge
through non-analysis, secrecy and related agreements.
Properties
Our principal executive offices are located at 4600 Touchton
Road East, Building 100, Suite 1500, Jacksonville,
Florida 32246 and at Transistorstraat 16, 1322 CE Almere, The
Netherlands.
We believe that our properties and equipment are generally in
good operating condition and are adequate for our present needs.
Production capacity at our sites can vary depending on
feedstock, product mix and operating conditions.
Substantially all of our properties are pledged as collateral to
secure our obligations under our credit agreements.
120
The following table sets forth our principal facilities. Because
our manufacturing properties include a significant amount of
plant equipment that is not located in buildings, we provide the
acreage of these properties in addition to the square footage of
the buildings located on the property. The size of executive and
administrative offices is provided in square feet.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Properties Servicing our North American Segment
|
|
|
|
Owned or
|
|
|
|
|
Square
|
|
|
|
|
|
|
Location
|
|
Leased
|
|
Acreage
|
|
|
Footage
|
|
|
Function
|
|
Principal Products
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Jacksonville, Florida United States
|
|
Leased
|
|
|
N/A
|
|
|
|
27,592
|
|
|
Executive and Administrative
|
|
—
|
|
Panama City, Florida United States
|
|
Owned
|
|
|
37
|
|
|
|
104,854
|
|
|
Manufacturing
|
|
Rosin Resins
Dispersions
Rosin Soap
Terpene Resins
|
|
Pensacola, Florida United States
|
|
Owned
|
|
|
21
|
|
|
|
64,109
|
|
|
Manufacturing
|
|
Terpene Phenolics
|
|
Valdosta, Georgia United States
|
|
Owned
|
|
|
35
|
|
|
|
81,089
|
|
|
Manufacturing
|
|
Rosin Resins
Resins Solutions
|
|
Savannah, Georgia United States
|
|
Owned
|
|
|
56
|
|
|
|
115,900
|
|
|
R&D and Manufacturing
|
|
Rosin Resins
Solution Resinates
|
|
Dover, Ohio,
United States
|
|
Owned
|
|
|
153
|
|
|
|
160,249
|
|
|
Manufacturing
|
|
Fatty Acid Dimers Polyamides
Fatty Acid Esters
Fatty Acid Upgrades
Castor Oil Upgrades
|
Properties Servicing Our European Segment
|
|
|
|
Owned or
|
|
|
|
|
Square
|
|
|
|
|
|
|
Location
|
|
Leased
|
|
Acreage
|
|
|
Footage
|
|
|
Function
|
|
Principal Products
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Almere, The Netherlands
|
|
Leased
|
|
|
N/A
|
|
|
|
42,590
|
|
|
Executive and Administrative and R&D
|
|
—
|
|
Oulu, Finland
|
|
Owned
|
|
|
24
|
|
|
|
173,456
|
|
|
Manufacturing
|
|
Rosin Resins
Rosin Soap
Dispersions
|
|
Chester-le-Street, United Kingdom
|
|
Owned
|
|
|
8
|
|
|
|
57,458
|
|
|
Manufacturing
|
|
Dimer
Specialty Fatty Acid
|
|
Niort, France
|
|
Owned
|
|
|
23
|
|
|
|
186,388
|
|
|
Manufacturing
|
|
Upgrading Adhesives Upgrading Inks
|
|
Sandarne, Sweden
|
|
Owned
|
|
|
66
|
|
|
|
293,788
|
|
|
Manufacturing
|
|
Rosin Esters
Dispersions
|
|
Gersthofen, Germany
|
|
Owned
|
|
|
4
|
|
|
|
39,116
|
|
|
Manufacturing
|
|
Disproportionated Rosin
|
Leases
We lease our Jacksonville, Florida property pursuant to a lease
agreement with FDG Deerwood North LLC. We recently amended this
lease to extend its term to March 1, 2016, and we have the
option to renew the lease for two additional terms of five years
each.
We lease our offices in Almere, Netherlands pursuant to two
lease agreements. The first lease agreement, with N.V.
Hypothecair Belang Gaasperdam I, expires on November 1,
2014. At the end of the lease period, we have a right to
purchase the leased property. The second lease is with Lobevis
Vastgoed B.V. The first period of the lease ends April 30,
2012. We have the right to terminate the
121
lease upon expiration of the first period if we provide one
year’s notice and pay a penalty. If we do not terminate the
lease, the second period of the lease will begin immediately and
end on October 31, 2014. We or the landlord may terminate
the lease upon expiration of the second period, provided that
the terminating party provides one year’s notice. If
neither party provides notice of termination, the lease will
renew for five year periods following October 31, 2014.
Although we own our BLS acidulation facility located in
Savannah, Georgia, this facility is located on land that we
lease from International Paper. This lease expires on
February 28, 2057.
Although we own our Oulu, Finland facilities, these facilities
are located on land that we lease from Enso Oy. This lease
expires on August 31, 2046, and we have the option to
extend the term until August 31, 2095.
Employees
As of March 31, 2010, we had 1,070 employees
worldwide, including 671 employees in the United States and
392 employees in Europe. Approximately 50% of our employees
are management, professional and administrative employees and
50% are hourly employees who principally work in our
manufacturing plants.
Approximately 54% of our employees in the United States are
represented by unions, and all bargaining unit jobs are limited
to those of our hourly-paid manufacturing employees. In Europe,
nearly all of our employees are represented by local works
councils or national labor unions, with the exception of senior
management at each location.
In the United States, we have entered into several new
collective bargaining agreements with our unions, including a
contract at our Dover, Ohio facility that is terminable on
February 11, 2015 and each anniversary thereafter, a
contract at our Panama City, Florida facility that is terminable
on May 13, 2014 and each anniversary thereafter, a contract
at our Pensacola, Florida facility that is terminable on
July 18, 2013 and each anniversary thereafter and a
contract at our Valdosta, Georgia facility that is terminable on
February 13, 2013 and each anniversary thereafter. We are
currently in negotiations with the United Steelworkers Union
with respect to our Savannah, Georgia facility.
We believe that our relationship with our employees is
satisfactory.
Information
Systems
We have implemented an SAP ECC 6.0 ERP system as an upgrade to
our current SAP 4.6 system in the United States and as a
replacement of our prior system in Europe. The implementation of
SAP ECC 6.0 supports our business strategy by establishing
standardized processes and procedures across our operations.
In connection with upgrading our ERP software, we are also
upgrading our technical infrastructure to support an anticipated
increase in transaction processing. We currently maintain two
data centers, one in Jacksonville, Florida and a second in
Almere, The Netherlands. The data stored in these centers is
redundant, meaning each data center stores the same information,
which reduces the risk of a network outage and protects us from
a disaster recovery perspective. Additionally, we are improving
our network to handle anticipated increases in traffic, and are
implementing a new data backup solution to improve data backup
and recovery services.
Environmental,
Health and Safety Regulation
We are subject to extensive environmental, health and safety, or
EHS, laws and regulations in the United States, the European
Union and elsewhere our operations are located. Many of these
laws and regulations impose requirements relating to air
emissions, wastewater discharges, the use, handling and disposal
of hazardous materials and wastes, exposure to chemicals and
other hazards, occupational health and safety, including dust
control, and the investigation and
clean-up of
122
contamination. Our facilities are required to obtain and comply
with a wide variety of environmental permits and authorizations
for different aspects of our operations. Environmental laws can
impose liability for violations or in the event of harm to
people, natural resources or property, and provide for
substantial fines, injunctions and potential civil and criminal
sanctions for violations. Our products, and the raw materials we
handle, are also subject to stringent industrial hygiene
regulations and assessments. The nature of our operations
exposes us to risks of liability pursuant to these laws and
regulations as a result of the production, handling, use,
storage, transportation and sale of materials that can cause
contamination or personal injury when handled improperly or
released into the environment.
Generally these environmental laws and regulations are becoming
increasingly stringent and the cost of compliance with EHS
requirements can be expected to increase over time. In
particular, the European Union’s Registration, Evaluation
and Authorization of Chemicals, or REACH, directive which
requires the registration and evaluation of potential
environmental and health impacts of chemicals manufactured in or
imported to the European Union, became effective in the European
Union in 2007. REACH will impose additional costs on the
chemical industry over the next 10 to 15 years and may
affect customer demand for certain of our products or our
ability to continue to manufacture and sell particular products
in the European Union. We estimate our total cost of compliance
with REACH through the end of 2018 will be approximately
$7.8 million. In addition, restrictions on the emission of
greenhouse gas emissions in the European Union, which will
become increasingly strict in future years, and pending
legislative and regulatory initiatives in the United States and
elsewhere could also increase our cost of production, affect the
cost or availability of feedstock or otherwise adversely affect
our operations. Our operations in Europe are subject to binding
caps on carbon dioxide emissions imposed by Member States of the
European Union. Such measures could result in increased costs
for us to operate and maintain our facilities. In the United
States, various bills and regulatory initiatives have been
introduced that would regulate emissions of carbon dioxide and
other greenhouse gases from industrial facilities. Although we
believe that greenhouse gases may be regulated in the near
future, we cannot yet predict the effect of any such regulation
on our operations.
We believe that we are in material compliance with all current
environmental laws and regulations. We currently estimate that
any expenses incurred in maintaining compliance with these
requirements will not materially affect our results of
operations or cause us to materially exceed our level of
anticipated capital expenditures. However, regulatory
requirements or permit conditions may change, and we cannot
predict the costs of additional measures that may be required to
maintain compliance as a result of future changes or expenses.
Environmental laws and regulations in various jurisdictions also
can impose obligations to clean up contamination from current or
historic operations. Under some circumstances, the current owner
or operator of a site can be held responsible for the entire
cost of the remediation of past contamination regardless of
fault and regardless of whether the activity was legal at the
time that it occurred. We can also be held liable for the
remediation of contamination at offsite locations, including
waste disposal sites that have been affected by past operations.
Evaluating and estimating the potential liability related to
site remediation projects involves significant uncertainties,
and many of our facilities have been affected by contamination
from historic operations.
Environmental contamination in excess of regulatory standards is
known to exist at certain of our facilities, including our
current and former facilities located in Dover, Ohio, Panama
City, Pensacola and Port St. Joe, Florida, Savannah and
Valdosta, Georgia, Sandarne, Sweden, Oulu, Finland, Niort,
France and Gersthofen, Germany. We are also aware that we will
be required to incur costs in connection with the closing of a
landfill at a site we acquired from Stora Enso in Sandarne,
Sweden, though we do not currently expect the costs to be
incurred in connection with this landfill or environmental
contamination at any of our other sites to be material. Although
resolution of environmental liabilities will require future cash
outlays, it is not expected that such outlays will materially
impact our liquidity position, although there can be no
assurance that such impacts could not occur. In
123
accordance with GAAP, as of March 31, 2010, we have accrued
a $4.0 million reserve for environmental liabilities.
While we recognize that we may, in the future, be held liable
with respect for remediation activities beyond those identified
to date, at present we are not aware of any circumstances that
are reasonably expected to give rise to remediation claims that
would have a material adverse effect on our results of
operations or cause us to exceed our projected level of required
remedial expenditures.
Legal
Proceedings
On February 26, 2010, Arboris, LLC filed a lawsuit against
Arizona Chemical Company and Arizona Arboris, Inc. in the United
States District Court, Middle District of Florida regarding
claims for relief under U.S. antitrust statutes as well as
a series of claims related to a tall oil pitch supply agreement
and a ground lease we entered into with them. In its complaint,
which it amended on April 28, 2010, Arboris, LLC seeks
injunctive and declaratory relief, damages plus interest, costs
and attorneys’ fees and punitive damages under U.S.
antitrust statutes. The amount of damages sought by Arboris, LLC
is unspecified. We believe we have strong defenses to these
claims based in part upon the advice of counsel, and we do not
believe these claims will have a material adverse affect on our
business or results of operations.
For more information regarding our relationship with Arboris,
LLC, see Note 14 of our consolidated financial statements
included elsewhere in this prospectus.
Additionally, while we are involved from time to time in
litigation and governmental actions arising in the ordinary
course of business, we are not aware of any actions which we
believe would individually or in the aggregate materially
adversely affect our business, consolidated results of
operations, financial position or cash flows.
124
MANAGEMENT
Directors and
Executive Officers
Set forth below are the names and ages of individuals who
currently serve as our directors and executive officers. Each
person named below has held the position indicated since our
initial organizational meetings held on February 16
and 17, 2010.
| |
|
|
|
|
|
|
|
Name
|
|
Age
|
|
Position
|
|
|
|
Leonard Berlik
|
|
|
62
|
|
|
Director
|
|
John R. Bolton
|
|
|
61
|
|
|
Director
|
|
Petter Johnsson
|
|
|
36
|
|
|
Director
|
|
Dr. Jochen Krautter
|
|
|
67
|
|
|
Director
|
|
Gerald Marterer
|
|
|
64
|
|
|
Director
|
|
Sebastien Mazella di Bosco
|
|
|
31
|
|
|
Director
|
|
Eytan Tigay
|
|
|
42
|
|
|
Director and Chairman of the Board of Directors
|
|
Cornelis Verhaar
|
|
|
56
|
|
|
Director, President and Chief Executive Officer
|
|
Frederic Jung
|
|
|
46
|
|
|
Vice President and Chief Financial Officer
|
|
Gary Reed
|
|
|
50
|
|
|
Vice President and General Manager — North America
|
|
Juhani Tuovinen
|
|
|
54
|
|
|
Vice President and General Manager — Europe
|
|
Dick Stuyfzand
|
|
|
49
|
|
|
Vice President and General Counsel
|
|
David Cowfer
|
|
|
50
|
|
|
Vice President, Human Resources and Corporate Communications
|
|
Glenda Haynes
|
|
|
50
|
|
|
Vice President, Internal Audit
|
|
Gary Garland
|
|
|
55
|
|
|
Vice President, Tax
|
|
Kellie Hardee
|
|
|
41
|
|
|
Treasurer
|
|
Astrid van der Valk
|
|
|
54
|
|
|
Corporate Controller
|
Leonard Berlik has been a member of the Board of
Managers of AZ Chem Investments LLC, the general partner of AZ
Chem Investments Partners LP, our parent, since May 16,
2007. He was previously an executive vice president of Imperial
Chemical Industries PLC and the Chief Executive Officer of
Uniquema BV from March 1, 2000 until June 1, 2006. He
also served as Executive Vice President, Adhesives for National
Starch & Chemical from April 1, 1996 until
February 1, 2000. Throughout Mr. Berlik’s
distinguished career in the chemical industry, he has focused on
the adhesives markets, working in The Netherlands, the United
States, Singapore, England and Germany. Mr. Berlik also
serves on the Board of Directors for Nizo Food Research BV,
Celerant Consulting, Oxea, SARL and Kolb, AG. He has a BS degree
from the University of Tennessee and an MBA from Xavier
University. Mr. Berlik’s extensive senior management
experience in the chemical industry, along with experience
gained from his numerous board memberships, led us to the
conclusion that he should serve as a member of our Board of
Directors.
John R. Bolton has been a member of the Board of
Managers of AZ Chem Investments LLC since January 20, 2010.
Mr. Bolton is currently a senior fellow at the American
Enterprise Institute. He served as the United States Permanent
Representative to the United Nations from August 1, 2005
until December 9, 2006. He also served as Under Secretary
of State for Arms Control and International Security from June
2001 until May 2005 during the administration of President
George W. Bush. Mr. Bolton is an attorney and has been
“of counsel” to the law firm of Kirkland &
Ellis since June 16, 2008. He currently serves on the Board
of Directors of Diamond Offshore Drilling, EMS Technologies and
TRACON Pharmaceuticals. Mr. Bolton has had a distinguished
career in public service and is a graduate of Yale College and
Yale Law School. Mr. Bolton’s legal experience and
board experience, together with his public service, led us to
the conclusion that he should serve as a member of our Board of
Directors.
125
Petter Johnsson has been a member of the Board of
Managers of AZ Chem Investments LLC since November 21,
2009. Mr. Johnsson began working for Rhône Group in
2002 and became a Managing Director in 2008. Previously,
Mr. Johnsson worked for Morgan Stanley Capital Partners
where he evaluated businesses and executed investments in a wide
range of industries. Since October 22, 2009,
Mr. Johnsson has served on the Board of Directors of Venice
Holdings s.r.l. He received a Master of Science in Economics and
Business from the Stockholm School of Economics and an MBA with
high distinction (Baker Scholar) from Harvard Business School.
Mr. Johnsson’s broad experience in the financial sector led
us to the conclusion that he should serve as a member of our
Board of Directors.
Dr. Jochen Krautter has been a member of the
Board of Managers of AZ Chem Investments LLC since July 26,
2007. He has 34 years of experience in the global chemical
industry with the Henkel Group, where he served as Executive
Vice President on the Management Board of Henkel KGaA in
Dusseldorf from 1992 to 2007. Now retired, Dr. Krautter was
a personal liable partner with Henkel KGaA from May 2000 until
his retirement on December 31, 2007 and remains obligated
under this condition until December 31, 2012. Prior to his
tenure as Executive Vice President of Henkel KGaA,
Dr. Krautter held numerous senior leadership positions with
the Henkel Group, including Chief Financial Officer of the
Henkel Group, the Managing Director of Henkel Belgium S.A. and
Henkel Nederland B.V. and Corporate Senior Vice President
Logistics of Henkel KGaA. He also held several sales and
marketing roles within Henkel’s Detergents division.
Dr. Krautter previously served on the boards of Clorox
Company, Loctite, Inc. and Ecolab Inc, although all these board
terms have now expired. Dr. Krautter graduated magna cum
laude with a Master’s degree from the Technical
University of Karlsruhe and summa cum laude with a PhD
from the University of Mannheim. Dr. Krautter’s extensive
senior management experience in the global chemical industry led
us to the conclusion that he should serve as a member of our
Board of Directors.
Gerald Marterer has been a member of the Board of
Managers of AZ Chem Investments LLC since March 7, 2007.
Mr. Marterer served as President and Chief Executive
Officer of Arizona Chemical from March 1, 2007 until his
retirement on August 31, 2008. He previously served for
more than 35 years in various senior management roles with
International Paper, including President of International Paper
Asia, Vice President of Quality and Vice President of the
Industrial Papers Division. He joined Arizona Chemical, which
was at the time a wholly owned subsidiary of International
Paper, in 2002 as the Vice President. He later led the
divestiture effort that culminated in Rhône Capital’s
ownership of Arizona Chemical in March 2007. Mr. Marterer
received his undergraduate degree in Economics from Clarion
University and an MBA from Gannon University. Mr.
Marterer’s experience leading Arizona Chemical, as well as
his extensive experience in senior management roles at
International Paper, led us to the conclusion that he should
serve as a member of our Board of Directors.
Sebastien Mazella di Bosco has been a member of
the Board of Managers of AZ Chem Investments LLC since
June 5, 2008. He joined Rhône Group in 2005.
Mr. Mazella di Bosco previously worked in the investment
banking department of Lazard Frères in the Paris and New
York offices from September 1, 2002 until July 30,
2005, advising clients on mergers and acquisitions with a
primary focus on the consumer, food and retail sectors. He has
been a member of the Board of Directors for Greek Directories
Holdings and Directory Services S.A. since May 2008.
Mr. Mazella di Bosco is a graduate of HEC Paris and holds a
BA in Philosophy from La Sorbonne. Mr. Mazella di
Bosco’s extensive experience in the financial sector and
his experience gained from his board memberships led us to the
conclusion that he should serve as a member of our Board of
Directors.
Eytan Tigay has been a member and chairman of the
Board of Managers of AZ Chem Investments LLC since March 7,
2007. Mr. Tigay joined Rhône Group as a Managing
Director in 2007. Prior to 2007, he was a Managing Director of
Lazard LLC and worked in several of its private equity fund
management businesses. Mr. Tigay also headed Lazard’s
strategic planning efforts and played a leadership role in its
initial public offering. He joined Lazard in 1989.
Mr. Tigay received a BA, magna cum laude, in
Economics from the University of Pennsylvania. Mr. Tigay’s
extensive senior
126
management experience in the financial sector led us to the
conclusion that he should serve as a member of our Board of
Directors.
Cornelis Verhaar has been a member of the Board of
Managers of AZ Chem Investments LLC since he was appointed
President and CEO upon joining Arizona Chemical on
September 1, 2008. His experience includes nearly
30 years of executive leadership in the global chemical
industry, most recently as Executive Vice President of Hexion
Specialty Chemicals, where he was responsible for the
company’s epoxy and coating resins business from 2006
through August 2008. Prior to joining Hexion, he held senior
management positions at companies including Noveon, Inc.,
Johnson Polymer, DeVoe Coatings, and ESHA Group.
Mr. Verhaar received his Masters in Economics from the
University of Amsterdam. Mr. Verhaar’s experience in
the global chemical industry, his technical expertise and his
senior management experience led us to the conclusion that he
should serve as a member of our Board of Directors.
Frederic Jung has been Vice President and CFO of
Arizona Chemical since December 1, 2008. From April 1,
2005 to November 30, 2008, he served as Corporate
Controller of Nalco Company, a $4 billion global producer
of specialty chemicals and services for water and waste
treatment and industrial processes. He also held the position of
CFO for Nalco’s EMEA operations from May 1, 2002 until
March 1, 2005. Prior to joining Nalco in January 2000,
Mr. Jung held several positions with Bombardier Aerospace,
Waste Management, Inc. and SAE Americas, Inc. Mr. Jung is a
graduate of Ecole Superieure Libre de Sciences Commerciales
Appliquees, a business school in France, and holds an MBA from
the University of Connecticut.
Gary Reed has been Vice President and General
Manager — North America of Arizona Chemical since
April 1, 2007. Mr. Reed began his career with Union
Camp in 1982 at the Savannah, GA, Pulp and Paper mill where he
held a number of operational management roles. In 1998, he moved
to Union Camp Chemicals, which was acquired by Arizona Chemical
in 1999. He has held several positions at Arizona Chemical,
including Site Manager — United Kingdom from
August 1, 1998 until June 2001, Inks & Coatings
Business Manager from June 1, 2002 until September 1,
2004 and Director of Marketing — U.S. from
October 1, 2004 until March 31, 2007. Mr. Reed
became a member of the Board of Management of Arboris, LLC on
March 1, 2010. Mr. Reed holds a degree in Chemical
Engineering from the University of Florida.
Juhani Tuovinen has been Vice President and
General Manager — Europe of Arizona Chemical since
March 1, 2007. Mr. Tuovinen began his career at United
Paper Mills in 1980, working in research and development and
commercial management roles in Finland and Germany.
Mr. Tuovinen joined Forchem Oy, a joint venture between
StoraEnso and United Paper Mills in 1994, which was acquired by
Arizona Chemical several years later. He has held a number of
key sales and marketing positions with Arizona Chemical,
including European Business Manager for Paper Chemicals from
March 1, 1997 until April 30, 1999 and European
Business Manager for Adhesives from May 1, 1999 to
June 30, 2005. He was also Managing Director for Europe and
the Business Manager — Adhesives from February 1,
2002 until February 28, 2007. Mr. Tuovinen graduated
with a Masters degree from the Helsinki University of Technology
with a concentration in Chemical Engineering/Polymer Chemistry.
Dick Stuyfzand has been Vice President and General
Counsel of Arizona Chemical since January 19, 2009. Prior
to joining Arizona Chemical, he was Legal Director of Schweppes
International from May 27, 2002 until December 31,
2008. He has over 20 years of international experience in
various industries with private and public companies, including
a major law firm and Royal Ahold in The Netherlands and Cadbury
Schweppes in the U.K. Mr. Stuyfzand earned his law degree
from the University of Utrecht in The Netherlands.
David Cowfer has been Vice President, Human
Resources and Corporate Communications of Arizona Chemical since
March 1, 2007. Prior to joining Arizona Chemical as
Director of Human Resources on June 1, 1999,
Mr. Cowfer had been Business Human Resources Manager for
the Fine Papers Division of International Paper since
June 1, 1996. He has over 28 years of human resources
127
leadership experience with private and public companies,
including International Paper, Eaton Corporation and
Westinghouse Electric Corporation. Mr. Cowfer is a graduate
of the Pennsylvania State University with a degree in Journalism.
Glenda Haynes has been Vice President, Internal
Audit of Arizona Chemical since November 1, 2009. She
joined Arizona Chemical in 1988. Ms. Haynes served as Vice
President of Treasury from March 1, 2007 until
October 31, 2009. She has held numerous senior management
positions in Finance at Arizona Chemical and has led several key
enterprise-wide projects during her tenure, including the March
2007 divestiture and integration with Rhône Capital. She is
a graduate of Texas A&M University with a degree in
Management and is a Certified Public Accountant.
Gary Garland has been Vice President, Tax of
Arizona Chemical since March 25, 2008. Mr. Garland was
Vice President, Taxes for Formica Corporation from
November 19, 2003 until March 24, 2008 and was
Director for Tax Technologies, Inc. He also served as Partner,
International Tax Services at Deloitte & Touche LLP,
and Assistant Tax Director at AT&T. Mr. Garland holds
a Master’s degree in Taxation from the University of
Cincinnati and a Bachelor’s degree in Accounting from
Wright State University and is a Certified Public Accountant.
Kellie Hardee has been Treasurer of Arizona
Chemical since November 1, 2009. She has provided treasury,
finance and accounting services for Arizona Chemical since
January 29, 2007. Prior to joining Arizona Chemical, she
was Vice President, Finance and Treasurer for Winn-Dixie from
June 12, 2000 until December 31, 2006. Ms. Hardee
has previously worked for Arthur Andersen as an Audit Manager,
Armor Holdings as a Controller and PSS Word Medical as a Senior
Accountant. Ms. Hardee holds both a Bachelor’s and a
Master’s degrees in Accounting from Florida State
University and is a Certified Public Accountant.
Astrid van der Valk has been Corporate Controller
of Arizona Chemical since December 1, 2009. Prior to
joining Arizona Chemical, she served as Senior Vice President
and Sector Controller for Philips Healthcare from August 1,
2005 until April 1, 2009. She has over 30 years of
international finance experience in various industries with
Royal Phillips Electronics and AT&T including Chief
Financial Officer for
AT&T-NSI’s
Public Switching Systems business in Europe from 1990 until
1994. She holds a Bachelor’s degree in Business Economics
from HEAO in The Hague and a Master’s degree in Business
Economics from Erasmus University in Rotterdam, The Netherlands.
The Board of
Directors
Our current board of directors has eight members, including
three members, Messrs. Krautter, Berlik and Bolton, who qualify
as “independent” under NYSE and SEC rules.
Our board of directors is divided into three classes, each of
whose members serve for a staggered three-year term. Upon the
expiration of the term of a class of directors, directors in the
class will be up for election for three-year terms at the annual
meeting of stockholders to be held in the year in which the term
expires.
Under our bye-laws, board members are elected by the
shareholders.
Controlled
Company
After the completion of this offering, Rhône Capital will
continue to control a majority of our outstanding common shares.
As a result, we will be a “controlled company” within
the meaning of the NYSE corporate governance standards. Under
the NYSE rules, a company of which more than 50% of the voting
power is held by an individual, group or another company is a
“controlled company” and may elect not to comply with
certain NYSE corporate governance standards, including:
|
|
|
| |
•
|
the requirement that a majority of a board of directors consist
of independent directors;
|
128
|
|
|
| |
•
|
the requirement that we have a nominating and corporate
governance committee that is composed entirely of independent
directors with a written charter addressing the committee’s
purpose and responsibilities;
|
| |
| |
•
|
the requirement that we have a compensation committee that is
composed entirely of independent directors with a written
charter addressing the committee’s purpose and
responsibilities; and
|
| |
| |
•
|
the requirement for an annual performance evaluation of the
nominating and corporate governance and compensation committees.
|
Following this offering, we intend to utilize these exemptions.
As a result, we will not have a majority of independent
directors, our Corporate Governance and Nominating Committee and
our Compensation Committee will not consist entirely of
independent directors and such committees will not be subject to
annual performance evaluations. Accordingly, you will not have
the same protections afforded to shareholders of companies that
are subject to all of the NYSE corporate governance requirements.
Committees of the
Board of Directors
We currently have four standing committees: the Audit Committee,
the Compensation Committee, the Corporate Governance and
Nominating Committee and the Executive Committee.
Audit Committee. Our Audit Committee
consists of Messrs. Krautter, Berlik and Bolton, with
Mr. Krautter as chairperson. SEC and NYSE rules require us
to have one independent audit committee member upon the listing
of our common shares on the NYSE, a majority of independent
directors within 90 days of the date of such listing and
all independent audit committee members within one year of the
date of such listing. Our board of directors has affirmatively
determined that each member of our Audit Committee meets the
definition of “independent director” for purposes of
serving on an audit committee under applicable SEC and NYSE
rules. In addition, Mr. Krautter qualifies as our
“audit committee financial expert”.
The Audit Committee will be responsible for, among other things:
|
|
|
| |
•
|
selecting, hiring and compensating our independent registered
public accounting firm, and pre-approving the audit and
non-audit services to be performed by our independent registered
public accounting firm;
|
| |
| |
•
|
reviewing the independent public accounting firm’s
qualifications, independence and performance;
|
| |
| |
•
|
reviewing the performance of the internal audit services
function;
|
| |
| |
•
|
discussing the scope and results of the audit with the
independent registered public accounting firm and reviewing with
management and the independent registered public accounting firm
our interim and year-end operating results;
|
| |
| |
•
|
reviewing the adequacy and effectiveness of our internal control
policies and procedures;
|
| |
| |
•
|
preparing the Audit Committee report and any other disclosures
required by the SEC to be included in our annual proxy statement;
|
| |
| |
•
|
monitoring the integrity of our financial statements and our
compliance with legal and regulatory requirements;
|
| |
| |
•
|
setting policies regarding the hiring of current and former
employees of the independent registered public accounting firm;
|
| |
| |
•
|
discussing types of information to be disclosed in earnings
press releases and provided to analysts and rating agencies;
|
129
|
|
|
| |
•
|
establishing procedures for receipt, retention and treatment of
complaints received by the Company regarding accounting or
internal controls and the submission of anonymous employee
concerns regarding accounting;
|
| |
| |
•
|
reviewing and discussing any reports concerning material
violations submitted to it by our attorneys or outside counsel
pursuant to the SEC attorney professional responsibility rules;
|
| |
| |
•
|
discussing with our general counsel legal matters having an
impact on financial statements; and
|
| |
| |
•
|
reviewing the policy with respect to related party transactions
and approving or rejecting proposed related party transactions.
|
Our board of directors will adopt a written charter for our
Audit Committee, which will be available on our corporate
website at www.arizonachemical.com upon completion of
this offering.
Compensation Committee. Our
Compensation Committee consists of Messrs. Berlik,
Johnsson, Tigay and Verhaar, with Mr. Tigay as chairperson.
The Compensation Committee will be responsible for, among other
things:
|
|
|
| |
•
|
reviewing and approving corporate goals and objectives relevant
to compensation of our chief executive officer and making
recommendations to the board with respect to the compensation of
other members of management;
|
| |
| |
•
|
recommending, when appropriate, changes to our compensation
philosophy and principles;
|
| |
| |
•
|
evaluating overall compensation and benefits programs;
|
| |
| |
•
|
overseeing regulatory compliance with respect to compensation
matters;
|
| |
| |
•
|
recommending to our board any changes in our incentive
compensation and equity-based plans that are subject to board
approval and overseeing the activities of individuals and
committees responsible for administering these plans;
|
| |
| |
•
|
reviewing and discussing with management, prior to the filing of
the proxy statement or annual report, the disclosures prepared
regarding the operations of the committee and our compensation
policies, including the CD&A and compensation tables (in
addition to preparing a report on executive compensation for the
proxy statement); and
|
| |
| |
•
|
overseeing our management development and succession planning
programs and making recommendations to the board with respect to
any aspects of such programs that are subject to board approval.
|
Our board of directors will adopt a written charter for our
Compensation Committee, which will be available on our corporate
website at www.arizonachemical.com upon completion of
this offering.
Corporate Governance and Nominating
Committee. Our Corporate Governance and
Nominating Committee consists of Messrs. Bolton, Marterer
and Verhaar with Mr. Verhaar as chairperson.
The Corporate Governance and Nominating Committee will be
responsible for, among other things:
|
|
|
| |
•
|
assisting our board of directors in identifying prospective
director nominees, and recommending nominees for each annual
meeting of shareholders to the board of directors;
|
| |
| |
•
|
reviewing developments in corporate governance practices and
developing and recommending governance principles applicable to
our board of directors;
|
| |
| |
•
|
reviewing our code of business conduct and ethics, recommending
any appropriate changes to the code to the board and reviewing
requests for waivers from the code;
|
130
|
|
|
| |
•
|
overseeing the evaluation of our board of directors and
management; and
|
| |
| |
•
|
recommending members for each board committee of our board of
directors.
|
Our board of directors will adopt a written charter for our
Corporate Governance and Nominating Committee, which will be
available on our corporate website at
www.arizonachemical.com upon completion of this offering.
Executive Committee. Our Executive
Committee consists of Messrs. Johnsson, Tigay and Verhaar,
with Mr. Verhaar as chairperson. The purpose of the
committee will be to act, between meetings of the board, with
the authority of the board on matters set forth in the
committee’s charter. Our board of directors will adopt a
written charter for our Executive Committee, which will be
available on our corporate website at www.arizonachemical.com
upon completion of this offering.
Compensation Committee Interlocks and Insider
Participation. During 2009, the functions of
our Compensation Committee were performed by Messrs. Johnsson,
Tigay and Verhaar. Mr. Verhaar also served as our President
and Chief Executive Officer. No executive officer serves as a
member of the board of directors or compensation committee of
any entity that has one or more executives serving as a member
of our board of directors.
Corporate
Governance
Prior to the completion of this offering our board of directors
will adopt policies and procedures to comply with the
Sarbanes-Oxley Act of 2002 and the rules adopted by the SEC and
the NYSE, including a code of conduct and ethics applicable to
our officers, directors and employees. Upon completion of this
offering, our code of conduct and ethics will be available on
our website at www.arizonachemical.com.
131
EXECUTIVE
COMPENSATION
Compensation
Discussion and Analysis
The compensation program for our executive officers is designed
to attract, retain and provide base compensation and meaningful
performance-based incentives for our management. This section
includes information and analysis related to the current and
future compensation programs for our named executive officers:
Cornelis Verhaar, President and Chief Executive Officer;
Frederic Jung, Vice President and Chief Financial Officer;
Juhani Tuovinen, Vice President and General
Manager — Europe; Gary Reed, Vice President and
General Manager — North America; and Dick Stuyfzand,
Vice President and General Counsel.
Compensation
Philosophy
We sustain a “pay for performance” culture for all our
salaried employees around the world. We reward strong
performance through awards that are directly linked to the
achievement of clear and measurable business objectives. We also
believe in encouraging ownership and entrepreneurialism among
our senior management team members.
Our annual performance management and employee development
system, which we refer to as “Waypoint”, is designed
to maintain a consistent link between our goals and individual
objectives, as well as sustain our
pay-for-performance
culture. Each year, employees at all levels, including our named
executive officers, develop formal objectives and later
participate in a mid-year review with their immediate manager
and receive an annual performance evaluation that takes into
account their view of their own performance, their
manager’s assessment and the assessment of other managers
who work closely with them.
We believe strongly in market-based total compensation. Until
the end of 2009, we had not engaged the services of a
compensation consultant. However, in order to ensure that our
compensation programs remain competitive, at the end of 2009, we
engaged Hewitt Associates, Inc. to conduct an executive
compensation survey. This data will be reviewed by the
Compensation Committee later this year and any decisions on
adjustments will be made at that time. Beginning in 2010 and on
a bi-annual basis thereafter, we may engage compensation
consultants to survey global companies in our industry and to
provide updated and appropriate compensation comparisons for our
leadership positions. Our analysis of compensation will consider
base salaries, incentive compensation and long-term incentives.
Our goal is to maintain competitive base salaries for our
executive team (including our named executive officers), but
allow for our performance based incentive systems —
both annual and long-term — to be the true drivers of
executive compensation and a cornerstone of our business model.
The various elements of compensation for named executive
officers and other senior managers are interlinked and decisions
made with respect to the objectives of each form of compensation
change year to year, depending upon the market conditions and
our business goals and other key initiatives. However, we do not
have a specified policy allocating between cash and non-cash
compensation or different forms of non-cash compensation. Our
total compensation program for senior managers are the incentive
plans, notably the annual Management Incentive Plan, which we
refer to as the “MIP”, and, historically, the
longer-term MIV I and MIV II plans (described below). For the
named executive officers, the basic elements of
compensation — base salary, retirement, health and
welfare benefits and other perquisites — are generally
aligned with industry norms and, in most cases, our named
executive officers participate in the same plans as our other
salaried employees. However, for named executive officers,
variable incentive compensation represents a larger portion of
their total compensation than it does for our other salaried
employees. As described below, our named executive officers also
participate in the MIVs. Decisions and the rewards associated
with MIP and MIV I and MIV II plans are strategic and directly
aligned with annual and long-term strategic goals,
132
including the creation of value for shareholders. Any changes in
the named executive officers’ salaries, bonus targets and
benefits must be reviewed and approved by the Compensation
Committee.
Role of the
Compensation Committee
The Compensation Committee reviews officers’ salaries and
annual bonus plan targets and assesses recommendations from the
CEO and the Vice President, Human Resources and Corporate
Communications related to compensation matters and the
individual performance of each officer. It determines
appropriate actions to maintain competitive pay practices for
the leadership group and makes decisions regarding rewards for
the accomplishment of performance goals based on shareholder
return, business growth, value creation and ethical leadership.
The Compensation Committee makes recommendations on any changes
to the board for approval.
Components of
Direct Compensation
Base
Salary
Employment contracts for the named executive officers are
established through negotiations between the officer and Arizona
Chemical. Decisions regarding compensation provided for by these
agreements are made based on biennial survey data and the
officer’s personal credentials and experience. The
Compensation Committee reviews the base salaries of all members
of our Global Leadership Team on an annual basis and determines
if an increase is warranted based on individual performance,
survey comparisons and the recommendations of the CEO. The CEO
prepares and conducts all annual performance evaluations for
members of our Global Leadership Team and the Compensation
Committee conducts the annual performance evaluation for our CEO.
In March 2009, all discretionary base salary increases for
managerial, professional and administrative employees, including
the named executive officers, were suspended due to the ongoing
global recession. The 2009 base salary for each named executive
officer is included in the Summary Compensation Table included
in this prospectus. These salaries are the same as the salaries
that were in effect for 2008. The salary freeze was suspended in
January 2010.
Annual Bonus:
Management Incentive Plan
Pursuant to their employment contracts, our named executive
officers are eligible to receive annual performance bonuses with
a target payout equal to 75% of base salary for
Mr. Verhaar, 60% of base salary for Mr. Jung, 60% of
estimated market midpoint of the salary range for their position
level for Messrs. Reed and Tuovinen and 39% of the estimated
market midpoint of the salary range for the position level for
Mr. Stuyfzand. The estimated market midpoint of the salary range
is generally determined based on the Company’s general
knowledge of market salary ranges and is not determined based on
specific benchmarking.
Our financial performance, as measured by the achievement of
Adjusted EBITDA and working capital goals, represents 80% of the
target annual bonus pool and individual performance metrics
represent 20%. Our MIP is designed to incentivize and reward
business achievement specific to annual targets, with
significant upside for exceeding goals and delivering greater
value to shareholders. Under the MIP, if threshold performance
is not achieved, no MIP payments will be paid for the year. The
MIP provides upside potential for financial performance that
exceeds target and downside for achievement below target but
exceeding threshold performance criteria. An upside multiple of
4 percentage points is added for each percentage point of
achievement above target for the Adjusted EBITDA, and a downside
multiple of 2.5 percentage points is applied for each
percentage point below target. For example, an achievement of
101% of target Adjusted EBITDA results in 104% of the MIP
payment, and an attainment of 99% of target Adjusted EBITDA
results in a pay out of 97.5% of the MIP payment. Similarly, an
upside multiple of 4 percentage points is added for each
percentage point of achievement above target for the estimate of
cash generated from working capital reductions, and a downside
multiple of 2.5 percentage points is applied for each percentage
point below target.
133
Based on working capital as of December 31, 2008 and
planned sales for the fiscal year 2009 and the fourth quarter of
2009, we calculated that a one day change in “Working
Capital Days of Sales”, which we use as a metric for cash
generated from working capital reductions, would have a
$2.2 million estimated impact on cash. We determine this
metric based on achievement of a trade working capital target
measured in “days of sales” calculated at year-end
based on a 3 month average of sales. These factors apply
only to the financial metrics and not to the individual
performance component.
MIP payments are made on the final workday in February each
year, and a participant must be actively employed by Arizona
Chemical on that day to be eligible for payment of a bonus under
the MIP.
Annual bonuses are currently calculated for the named executive
officers in the following manner: the year-end Adjusted EBITDA
achievement with respect to the annual budget determines 60% of
plan payout, 20% is determined based on the Working Capital Days
of Sales target and 20% is determined based on personal
performance with respect to each individual’s annual
performance objectives as communicated at the beginning of each
bonus year. Each named executive officer has performance-based
goals as described in the table below. The personal performance
goals are qualitative in nature, and are based, among other
things, on each executive’s contribution to the
Company’s achievement of its financial performance goals
described above. The Compensation Committee determines whether,
and to what extent, personal performance goals have been
exceeded, met or not met.
As described above, the target pool level for the Management
Incentive Plan is generally calculated by taking the sum of each
MIP eligible participant’s estimated market midpoint of the
salary range for their position level (or in the case of Messrs.
Verhaar and Jung, their base salary) multiplied by their target
payout percentage. The 2009 MIP award pool was, in the
aggregate, 171.1% of the target pool size that would have been
realized for meeting budget targets, though awards varied for
each participant depending upon personal performance. The award
was rounded to 170% for all participants.
Messrs. Verhaar, Jung, Tuovinen, Reed and Stuyfzand each
earned awards of 170% of their personal targets in 2009.
The following table reflects the calculation of 2009 MIP awards
and reflects the determinations made with respect to personal
performance objectives:
2009 Management
Incentive Plan — Final Award Calculation
Adjusted
EBITDA (dollars in millions):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
2009 MIP Pool
|
|
|
|
|
|
|
|
|
|
|
$ 4.9
|
|
|
Adjusted EBITDA Payout %
|
|
|
|
|
|
|
|
|
|
|
60
|
%
|
|
Adjusted EBITDA Portion of MIP Pool (at Target)
|
|
|
|
|
|
|
|
|
|
|
$ 2.9
|
|
|
2009 EBITDA Target
|
|
|
|
|
|
|
|
|
|
|
$80.0
|
|
|
Payment Threshold
|
|
|
|
|
|
|
|
|
|
|
$75.0
|
|
|
Above Target Award Point Multiplier
|
|
|
|
|
|
|
|
|
|
|
4.0
|
x
|
|
Below Target Award Point Multiplier
|
|
|
|
|
|
|
|
|
|
|
2.5
|
x
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Payout % of
|
|
Adjusted EBITDA
|
|
Payout % of
|
|
|
|
|
|
% of
|
|
Adjusted EBITDA
|
|
Portion of
|
|
Total
|
|
|
|
Adjusted EBITDA
|
|
Target
|
|
Portion
|
|
Payout
|
|
MIP Pool
|
|
|
|
Target
|
|
|
80.0
|
|
|
|
100
|
%
|
|
|
100.0
|
%
|
|
|
2.9
|
|
|
|
60.0
|
%
|
|
Achieved
|
|
|
93.7
|
|
|
|
117
|
%
|
|
|
168.7
|
%
|
|
|
4.9
|
|
|
|
101.2
|
%
|
134
Working
Capital (dollars in millions):
| |
|
|
|
|
|
|
|
|
|
Working Capital Days of Sales Payout %
|
|
|
|
|
|
|
20
|
%
|
|
Working Capital Portion of MIP Pool (at Target)
|
|
|
|
|
|
$
|
1.0
|
|
|
2009 Working Capital Days of Sales Target
|
|
|
|
|
|
|
59
|
|
|
Payment Threshold
|
|
|
|
|
|
|
62
|
|
|
Above Target Award Point Multiplier
|
|
|
|
|
|
|
4.0
|
x
|
|
Below Target Award Point Multiplier
|
|
|
|
|
|
|
2.5
|
x
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Working
|
|
Cash Generated
|
|
% of
|
|
Payout % of
|
|
Working Capital
|
|
Payout % of
|
|
|
|
Capital
|
|
in 2009 from
|
|
Cash
|
|
Working Capital
|
|
Portion of
|
|
Total
|
|
|
|
Days of Sales
|
|
Working Capital
|
|
Target
|
|
Portion
|
|
Payout
|
|
MIP Pool
|
|
|
|
Target
|
|
|
59
|
|
|
$
|
30.0
|
|
|
|
100.0
|
%
|
|
|
100
|
%
|
|
$
|
1.0
|
|
|
|
20.0
|
%
|
|
Achieved
|
|
|
54
|
|
|
$
|
41.2
|
|
|
|
137.4
|
%
|
|
|
250
|
%
|
|
$
|
2.4
|
|
|
|
50.0
|
%
|
Personal
Objective (dollars in millions):
| |
|
|
|
|
|
Personal Objective Payout %
|
|
|
20
|
%
|
|
Personal Objective Portion of MIP Pool (at Target)
|
|
$
|
1.0
|
|
| |
|
|
|
|
|
|
|
Goal
|
|
Result
|
|
|
|
Cornelis
Verhaar
|
|
Financial
Leadership in meeting corporate financial targets; implement
cost improvement process
|
|
Exceeded
|
|
|
|
Customer Focus
Provide leadership in achieving growth of sales at key customers
|
|
Achieved
|
|
|
|
Organization
Coordinate establishment of global business processes and
structure; develop culture focused on speed of execution and
openness
|
|
Achieved
|
|
|
|
Strategic Initiatives
Prepare strategic plan; establish growth programs and goals;
review IT infrastructure and capabilities
|
|
Achieved
|
|
|
|
Cost Reduction
Identify and implement initiatives to reduce costs
|
|
Exceeded
|
|
Frederic
Jung
|
|
Cash Management
Leadership in meeting corporate Free Cash Flow and Net Working
Capital Days targets
|
|
Exceeded
|
|
|
|
Savings Initiative
Implement process to achieve 2009 Cost Savings target
|
|
Exceeded
|
|
|
|
Project APEX:ERP (SAP financial system implementation)
On time, on budget
|
|
Achieved
|
|
|
|
Financial Reporting
Ensure accuracy/ consistency; redesign/implement cost structure
and allocations; redesign transfer pricing; improve forecasting
of P&L, cash flow
|
|
Achieved
|
|
|
|
Finance Organization
Evaluate finance team, redesign finance organization, implement
foundations of global shared service center, continuous benchmark
|
|
Achieved
|
|
|
|
Risk Management
Develop/implement strategies for interest rate, foreign exchange
and energy fluctuation protections
|
|
Achieved
|
|
|
|
IT Strategy
Deliver strategic plan of IT strategy and capability including
benchmarking for long term; complete 2009 improvements
|
|
Not Achieved
|
135
| |
|
|
|
|
|
|
|
Goal
|
|
Result
|
|
|
Juhani
Tuovinen
|
|
Financial Performance
Lead European organization to achieve 2009 targets
|
|
Achieved
|
|
|
|
Leadership
Provide leadership to achieve corporate 2009 financial targets
|
|
Exceeded
|
|
|
|
Operation Excellence
Lead European organization to achieve 2009 operational targets
|
|
Achieved
|
|
|
|
People
Lead European organization to secure skills and competencies to
achieve business targets
|
|
Achieved
|
|
|
|
Financial Results — longer term
Lead European organization to achieve targeted long-term results
|
|
Achieved
|
|
|
|
Customer Focus
Lead European organization to achieve targeted service level and
growth results
|
|
Not Achieved
|
Gary
Reed
|
|
Financial Performance
Assist Arizona Chemical to meet North American 2009 fiscal
targets, 2009 working capital targets and global 2009 financial
targets
|
|
Exceeded
|
|
|
|
Operational Excellence
Achieve step change in safety performance while maintaining
environmental performance
|
|
Not Achieved
|
|
|
|
People
Develop organizational capability via people development and
performance management; renegotiate expiring labor agreements
|
|
Achieved
|
|
|
|
Customer Focus
Improve customer satisfaction in North America and achieve
growth objectives
|
|
Achieved
|
Dick
Stuyfzand
|
|
Reinforce Legal and IP functions
Build internal working relationships and across the business
|
|
Achieved
|
|
|
|
Board
Set up and facilitate support function, organize agendas and
meetings of board of directors of AZ Chem Investments LLC
|
|
Achieved
|
|
|
|
Organization (Legal and IP)
Manage and evaluate legal and IP teams and prepare
recommendations re: structure, costs and budget; evaluate and
build relationship with outside counsel network
|
|
Achieved
|
|
|
|
Governance and Compliance
Develop and start implementing governance and internal controls
strategy and framework for initial public offering
|
|
Exceeded
|
Based on the outcome of each of their annual performance
assessments, the Compensation Committee determined that each of
our named executive officers either exceeded or met commitments
under their personal performance goals.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Payout % of
|
|
Personal
|
|
|
|
|
|
|
|
Personal
|
|
Performance Goals
|
|
Payout % of
|
|
|
|
% of
|
|
Performance Goals
|
|
Portion of
|
|
Total
|
|
|
|
Target
|
|
Portion
|
|
Payout
|
|
MIP Pool
|
|
|
|
Target
|
|
|
100
|
%
|
|
|
100.0
|
%
|
|
$
|
1.0
|
|
|
|
20.0
|
%
|
|
Achieved
|
|
|
100
|
%
|
|
|
100.0
|
%
|
|
$
|
1.0
|
|
|
|
20.0
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Calculated Payout
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
171.1
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
of target
award
|
|
136
AZ Chem MIV I
Ltd. and AZ Chem MIV II LP
In June 2007, AZ Chem MIV I Ltd. (“MIV I”)
and AZ Chem MIV II Ltd. (“MIV II”) were
formed as discussed in more detail below under “Other
Compensation Matters — MIV I and
MIV II.” Prior to this offering, the MIVs have been
an integral part of our long-term ownership and compensation
program, designed to foster ownership and entrepreneurial
behavior among senior managers (including our named executive
officers) and to enhance our value for all shareholders by
aligning the interests of management with our company’s
equity owners. Following the consummation of this offering,
there will be no further investments in or grants of equity
awards by the MIVs. All subsequent grants of performance based
equity awards will be made under our 2010 Long-Term Incentive
Plan, which is described below under “Executive
Compensation — Other Compensation Matters —
MIV I and MIV II.”
Grants of Common Profits Interests in MIV I and MIV
II. Executives who are shareholders of MIV I or
limited partners in MIV II, including each of our named
executive officers, have been eligible to receive common profits
interests grants in the respective MIV in which they have an
ownership interest. Eligibility to receive MIV grants is based
on executives being in good standing at the time the MIV grants
are made, which includes executives having “exceeded”
or “met” commitments under their personal performance
goals. The 2009 personal goals of our named executive
officers are described above under “2009 Management
Incentive Plan — Final Award Calculation”. For
2009, each of our named executive officers was determined to
have achieved an overall rating of “met” or
“exceeded” commitments and, therefore, was eligible to
receive an MIV award in 2009. See “— Outstanding
Equity Awards at 2009 Fiscal Year-End” below for more
information concerning the common profits interest grants that
have been made to our named executive officers.
An initial grant of common profits interests was made to the
executives who invested in connection with the formation of MIV
I and MIV II in June 2007. Subsequent grants of common profits
interests were made to those executives in 2008 and 2009. Each
of Messrs. Verhaar, Jung and Stuyfzand received grants of
common profits interests in connection with the commencement of
their employment with us. Additionally, Mr. Verhaar
participated in the grants of common profits interests made
subsequent to his employment in 2008 and 2009, and
Mr. Stuyfzand participated in the grants of common profits
interests made subsequent to his employment in 2009. All common
profits interests vest over a five year period from the date of
grant, with accelerated vesting in certain circumstances.
As of March 31, 2010, the common profits interests
represented approximately 3.9% of the outstanding common equity
interests in AZ Chem Investments Partners LP. On May 26, 2010,
the Compensation Committee made grants of common profits
interests, or CPIs, to our executives who are shareholders or
limited partners in the MIVs, including each of our named
executive officers. These grants represent an additional
approximately 2.1% of the outstanding common interests in AZ
Chem Investments Partners LP and will be the final grants of
common profits interests to be made by the MIVs. In connection
with these grants, the Compensation Committee determined that
each named executive officer “exceeded” or
“met” their respective performance goals for 2009.
Mr. Jung was granted 31,967 CPIs, Mr. Reed was granted 20,000
CPIs, Mr. Stuyfzand was granted 5,000 CPIs, Mr. Tuovinen
was granted 20,000 CPIs and Mr. Verhaar was granted 90,354 CPIs.
These grants were the final grants of common profits interests
to be made by the MIVs. The grants represent awards for service
in 2009 as well as additional grants (subject to certain
clawback provisions) of the remaining common profits interests
that would have been awarded in 2011 and 2012. Unlike prior
grants of common profits interests in the MIVs, the common
profits interests granted on May 26, 2010 have established value
as of the date of grant of $13.36 and recipients who are US
taxpayers may elect either to pay any tax liability associated
with the grant at the time of grant based on the $13.36 per CPI
value or to pay the tax liability based on the value of the CPIs
upon vesting. The CPIs vest over the 5-year period following the
date of grant.
All subsequent grants of performance based equity awards will be
made under our 2010 Long-Term Incentive Plan, which is described
below. When making future equity grants under the 2010 Long-Term
Incentive Plan to executives who have received common profits
interests awards discussed
137
above for 2011 and 2012, the Compensation Committee will
consider the award of other equity based awards.
Fringe
Benefits/Perquisites
Several of the named executive officers receive housing support
pursuant to their employment contracts. Mr. Tuovinen, a
Finnish national, works and lives with his family in The
Netherlands and receives housing support of approximately
$42,000 per year. Mr. Jung, a U.S. expatriate who
currently resides with his family in The Netherlands, receives
approximately $46,700 per year in housing support pursuant to
his employment agreement. In addition, we also provide certain
of our named executive officers (Messrs. Verhaar, Jung,
Tuovinen and Stuyfzand) with automobile allowances, which are
set forth in the All Other Compensation table set forth below.
In 2009, Mr. Jung also received relocation assistance for
his move to The Netherlands, which is also set forth in the All
Other Compensation table below as well as a goods and services
differential allowance of $7,604.75 per month.
Components of
Post-Employment Compensation
Employment
Agreements and Severance Benefits
Messrs. Verhaar, Stuyfzand and Tuovinen have employment
agreements that are customary for senior management employees
based in The Netherlands. Mr. Verhaar’s agreement
includes a non-compete provision for a period of two years
following termination of employment. He is entitled to severance
of two years of continued base salary. Messrs. Stuyfzand
and Tuovinen have no specific severance provision in their
contracts, although both are subject to notice periods upon
resignation and continue to be paid during such notice periods.
There are no non-compete provisions for these two individuals,
but they are subject to confidentiality agreements.
Mr. Jung is currently a U.S. expatriate residing in
The Netherlands (as of August 1, 2009). His employment is
subject to a letter agreement setting forth the terms of his
international assignment, which includes various provisions for
housing, tax equalization, annual home leave and a termination
and severance provision that provides repatriation and
relocation to the U.S. Mr. Jung also entered into a
termination agreement with us upon his original date of hire of
December 1, 2008. This agreement requires a notice period
if he resigns and the payment of one year of base salary, plus
prorated bonus compensation if we terminate his employment for
reasons other than cause. He and Mr. Reed are currently
subject to the provisions of our U.S. Severance Program,
which requires endorsement of a severance agreement in exchange
for a lump sum severance payment and additional benefits,
provided the separation occurs for reasons other than cause. In
addition, if the Company is sold prior to an initial public
offering, then Messrs. Jung, Reed and Stuyfzand would have
an additional severance benefit equal to 12 months of base
salary plus target bonus if their employment is involuntarily
terminated (although this additional severance entitlement will
terminate upon the consummation of this offering).
U.S. Defined
Contribution Savings Plan
Upon the Acquisition, a defined contribution Employee Savings
Plan was created for all United States-based employees, the
Arizona Chemical Savings Plan. The savings plan is a
tax-qualified 401(k) plan, and we match employees’
contributions on the following basis: 70 cents on the dollar for
the first four percent of the employee’s contribution and
50 cents on the dollar for up to an additional four percent.
United
States-based
members of the senior management team, such as Messrs. Jung
and Reed, participate in the savings plan and are subject to the
same provisions as other employees. The savings plan is designed
to maintain competitive retirement benefits and assist employees
in building their retirement fund.
We do not maintain any non-qualified savings plan or any other
deferred compensation plan for the named executive officers or
any other senior managers. We also do not maintain any
tax-qualified or non-qualified defined benefit pension plan.
138
U.S. Profit
Sharing Award
As a means to further enhance retirement savings for United
States-based employees, we maintain a discretionary annual
profit sharing award under the Employee Savings Plan. This award
comes in the form of a discretionary gift contribution made
directly to each
United States-based
salaried employee’s savings plan account based on our
performance. Awards were made during the first quarter of 2008
and 2009 and in both cases, were equivalent to four percent of
annual base pay for each person.
The award is intended to be variable in nature depending upon
the performance of the business. All employees receive the same
percent award.
United States-based
members of the senior leadership team, such as Messrs. Jung
and Reed, are eligible for this award.
The award is designed to foster our defined contribution
approach to retirement planning and savings and maintain
competitive benefits so we may attract and retain employees in
the United States. Decisions regarding the timing and
payment amount are made by the Compensation Committee in
February.
Pension
Schemes for Europe-Based Executives
Messrs. Verhaar, Tuovinen and Stuyfzand are participants in
a defined contribution, collective pension scheme in The
Netherlands with Zwitserleven. The scheme also includes coverage
for long-term disability. The employee is required to contribute
eight percent of the fixed gross annual salary that exceeds
€53,050 per year as the employee’s contribution to the
scheme. Bonus compensation is not included in the pensionable
salary.
Life
Insurance, Long-Term Disability Insurance
In addition to the long-term disability coverage available under
the pension schemes for our Europe-based leaders,
Messrs. Jung and Reed also are eligible for basic life
insurance coverage for our United States-based employees of two
times their annual base salary as well as Accidental Death and
Dismemberment insurance of two times their annual base salary,
travel accident insurance of the lesser of five times their
annual base salary subject to a minimum of $100,000 and a
maximum of $1,500,000 and long-term disability coverage of 60%
to
662/3%
of monthly salary; depending on whether the employee qualifies
for family or primary Social Security benefits due to disability.
Summary
Compensation Table
The following table sets forth certain information concerning
annual compensation for our named executive officers during the
year ending December 31, 2009.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Non-Equity
|
|
Change in
|
|
|
|
|
|
|
|
|
|
|
|
Share
|
|
Incentive Plan
|
|
Pension
|
|
All Other
|
|
|
|
|
|
|
|
Salary
|
|
Awards
|
|
Compensation
|
|
Value
|
|
Compensation
|
|
Total
|
|
Name and Principal Position
|
|
Year
|
|
($)
|
|
($)(4)
|
|
($)(5)
|
|
($)
|
|
($)(6)
|
|
($)
|
|
|
|
Cornelis Verhaar
|
|
|
2009
|
|
|
|
574,012
|
|
|
|
8,245
|
|
|
|
731,865
|
|
|
|
128,880
|
|
|
|
38,011
|
|
|
|
1,481,013
|
|
|
President and Chief
Executive Officer
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Frederic Jung(1)
|
|
|
2009
|
|
|
|
285,000
|
|
|
|
41,518
|
|
|
|
290,700
|
|
|
|
—
|
|
|
|
195,074
|
|
|
|
812,292
|
|
|
Vice President &
Chief Financial Officer
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Juhani Tuovinen(2)
|
|
|
2009
|
|
|
|
315,707
|
|
|
|
7,500
|
|
|
|
305,261
|
|
|
|
26,841
|
|
|
|
70,808
|
|
|
|
726,117
|
|
|
Vice President &
General Manager — Europe
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gary Reed
|
|
|
2009
|
|
|
|
250,000
|
|
|
|
7,000
|
|
|
|
245,140
|
|
|
|
—
|
|
|
|
21,760
|
|
|
|
523,900
|
|
|
Vice President &
General Manager —
North America
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Dick Stuyfzand(3)
|
|
|
2009
|
|
|
|
222,430
|
|
|
|
10,602
|
|
|
|
159,541
|
|
|
|
34,381
|
|
|
|
26,322
|
|
|
|
453,276
|
|
|
Vice President &
General Counsel
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
139
|
|
|
|
(1) |
|
Mr. Jung joined us as Chief Financial Officer on
December 1, 2008. He was transferred from the United States
to The Netherlands on August 1, 2009. All compensation he
received in Euros has been converted to U.S. dollars at an
exchange rate of 1.43503 (exchange rate as of December 31,
2009). |
| |
|
(2) |
|
Mr. Tuovinen’s compensation was paid in Euros, but for
the purposes of this table his compensation has been converted
to U.S. dollars at an exchange rate of 1.43503 (exchange rate as
of December 31, 2009). |
| |
|
(3) |
|
Mr. Stuyfzand joined us on January 19, 2009. His
compensation was paid in Euros, but for the purposes of this
table his compensation has been converted to U.S. dollars at an
exchange rate of 1.43503 (exchange rate as of December 31,
2009). |
| |
|
(4) |
|
Amounts in the “Share Awards” column above reflect the
grant date fair value (as determined in accordance with FASB ASC
Topic 718 — Stock Compensation), based on the
assumptions set forth in Note 17 of our consolidated
financial statements appearing at the end of this prospectus. |
| |
|
(5) |
|
Amounts disclosed in the “Non-Equity Incentive Plan
Compensation” column represent amounts earned under our MIP. |
| |
|
(6) |
|
See following table titled “All Other Compensation”
for details regarding amounts disclosed in the “All Other
Compensation” column for fiscal year 2009. |
All Other
Compensation Table
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Contributions
|
|
|
|
|
|
|
|
|
|
|
|
|
|
to Defined
|
|
|
|
|
|
|
|
|
|
|
|
Auto
|
|
Contribution
|
|
Housing
|
|
Relocation
|
|
|
|
|
|
|
|
Allowance
|
|
Savings Plan
|
|
Assistance
|
|
Assistance
|
|
Total
|
|
Name and Principal Position
|
|
Year
|
|
($)(1)
|
|
($)(2)
|
|
($)(3)
|
|
($)(4)
|
|
($)
|
|
|
|
Cornelis Verhaar
|
|
|
2009
|
|
|
|
38,011
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
38,011
|
|
|
President and Chief Executive Officer
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Frederic Jung
|
|
|
2009
|
|
|
|
30,820
|
|
|
|
6,840
|
|
|
|
18,648
|
|
|
|
138,766
|
|
|
|
195,074
|
|
|
Vice President &
Chief Financial Officer
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Juhani Tuovinen
|
|
|
2009
|
|
|
|
24,780
|
|
|
|
—
|
|
|
|
46,028
|
|
|
|
—
|
|
|
|
70,808
|
|
|
Vice President & General Manager — Europe
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gary Reed
|
|
|
2009
|
|
|
|
—
|
|
|
|
21,760
|
|
|
|
—
|
|
|
|
—
|
|
|
|
21,760
|
|
|
Vice President & General Manager —
North America
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Dick Stuyfzand
|
|
|
2009
|
|
|
|
26,322
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
26,322
|
|
|
Vice President & General Counsel
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1) |
|
Represents the incremental cost to us relating to
Messrs. Verhaar, Tuovinen, Jung and Stuyfzand’s
personal use of our provided and owned automobiles which
incremental cost is equal to the sum of the lease payments and
insurance premium payments made during the applicable fiscal
year for each automobile provided to Messrs. Verhaar,
Tuovinen, Jung and Stuyfzand, respectively. |
| |
|
(2) |
|
Represents Company contributions made on behalf of the indicated
executive officer to the Arizona Chemical Company Employee
Savings Plan, a tax-qualified retirement plan for
U.S.-based
employees of the Company, including Messrs. Jung and Reed. |
| |
|
(3) |
|
Represents payments to cover eligible housing expenses incurred
by Mr. Tuovinen and Mr. Jung in connection with their
personal residences in The Netherlands. |
| |
|
(4) |
|
Represents eligible expenses incurred by Mr. Jung in
connection with his relocation from Chicago, Illinois to Almere,
The Netherlands. |
140
Grants of Plan
Based Awards
The following table sets forth information regarding awards made
to our named executive officers under the MIV I and MIV II plans
during the year ending December 31, 2009.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
All Other Share-Based
|
|
|
|
|
|
Grant
|
|
Awards: Number of
|
|
Grant Date Fair Value of
|
|
Name and Principal Position
|
|
Date
|
|
Units (#)(1)
|
|
Share-Based Awards ($)(5)
|
|
|
|
Cornelis Verhaar
|
|
8/31/09
|
|
|
8,245
|
(2)
|
|
$
|
8,245
|
|
|
President and Chief Executive Officer
|
|
|
|
|
|
|
|
|
|
|
|
Frederic Jung
|
|
8/31/09
|
|
|
41,518
|
(3)
|
|
$
|
41,518
|
|
|
Vice President &
Chief Financial Officer
|
|
|
|
|
|
|
|
|
|
|
|
Juhani Tuovinen
|
|
8/31/09
|
|
|
7,500
|
(2)
|
|
$
|
7,500
|
|
|
Vice President & General Manager — Europe
|
|
|
|
|
|
|
|
|
|
|
|
Gary Reed
|
|
8/31/09
|
|
|
7,000
|
(2)
|
|
$
|
7,000
|
|
|
Vice President & General Manager — North America
|
|
|
|
|
|
|
|
|
|
|
|
Dick Stuyfzand
|
|
8/31/09
|
|
|
10,602
|
(4)
|
|
$
|
10,602
|
|
|
Vice President & General Counsel
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1) |
|
Represents awards of common profits interests. |
| |
|
(2) |
|
Annual performance based grant. |
| |
|
(3) |
|
Initial grant for joining the plan (40,825) plus partial year
performance grant (693). |
| |
|
(4) |
|
Initial grant only. |
| |
|
(5) |
|
As described above under “Grants of Common Profits
Interests in MIV I and MIV II”, the 2009 grants
were in the form of common profits interests, which entitle the
recipient to future profit allowances but generally have $0
value upon grant. |
These amounts reflect the grant date fair value (as determined
in accordance with FASB ASC Topic 718 — Stock
Compensation), based on the assumptions set forth in
Note 17 of our consolidated financial statements included
elsewhere in this prospectus.
141
Outstanding
Equity Awards at 2009 Fiscal Year-End
The following table sets forth information regarding outstanding
awards made to our named executive officers under the MIV I and
MIV II plans, as of December 31, 2009.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Number of Units
|
|
Market Value of Units
|
|
|
|
Grant
|
|
That Have Not
|
|
That Have Not
|
|
Name and Principal
Position
|
|
Date
|
|
Vested (#)(1)
|
|
Vested ($)(2)
|
|
|
|
Cornelis Verhaar
|
|
|
9/1/08
|
|
|
|
98,666
|
|
|
$
|
0
|
|
|
President and Chief Executive Officer
|
|
|
8/31/09
|
|
|
|
8,245
|
|
|
|
|
|
|
Frederic Jung
|
|
|
8/31/09
|
|
|
|
41,518
|
|
|
$
|
0
|
|
|
Vice President &
Chief Financial Officer
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Juhani Tuovinen
|
|
|
2/28/08
|
|
|
|
38,205
|
|
|
$
|
0
|
|
|
Vice President & General
|
|
|
9/1/08
|
|
|
|
9,551
|
|
|
$
|
0
|
|
|
Manager — Europe
|
|
|
8/31/09
|
|
|
|
7,500
|
|
|
$
|
0
|
|
|
Gary Reed
|
|
|
2/28/08
|
|
|
|
38,205
|
|
|
$
|
0
|
|
|
Vice President & General
|
|
|
9/1/08
|
|
|
|
9,551
|
|
|
$
|
0
|
|
|
Manager — North America
|
|
|
8/31/09
|
|
|
|
7,000
|
|
|
$
|
0
|
|
|
Dick Stuyfzand
|
|
|
8/31/09
|
|
|
|
10,602
|
|
|
$
|
0
|
|
|
Vice President & General Counsel
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1) |
|
Represents common profits interests. |
| |
|
(2) |
|
As described above under “Grants of Common Profits
Interests in MIV I and MIV II”, the 2009 grants
were in the form of common profits interests, which entitle the
recipient to future profit allowances but generally have $0
value upon grant. Although the grants are vested, there is no
liquidity unless there is a distribution. |
2009 Pension
Benefits Table
The following table sets forth certain information concerning
pension benefits, as of December 31, 2009, for our named
executive officers who participated in pension schemes
maintained for our European-based executives (as described
above).
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Payments
|
|
|
|
|
|
Number of
|
|
Present Value of
|
|
During
|
|
|
|
|
|
Years of
|
|
Accumulated
|
|
the Last
|
|
|
|
|
|
Credited
|
|
Benefit
|
|
Fiscal Year
|
|
Name and Principal Position
|
|
Plan Name
|
|
Service (#)
|
|
($)(1)
|
|
($)
|
|
|
|
Cornelis Verhaar
|
|
ZwitserLeven Contract GN 2595
|
|
1 year and 5 months
|
|
$
|
159,972
|
|
|
—
|
|
President and Chief
Executive Officer
|
|
|
|
|
|
|
|
|
|
|
|
Juhani Tuovinen
|
|
ZwitserLeven Contract GN 2595
|
|
3 years and 6 months
|
|
$
|
224,413
|
|
|
—
|
|
Vice President & General
Manager — Europe
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Finnish Managing Directors of AZC OY in Finland and EU
|
|
3 years and 1 month
|
|
$
|
41,776
|
|
|
—
|
|
Dick Stuyfzand
|
|
ZwitserLeven Contract GN 2595
|
|
1 Year
|
|
$
|
34,381
|
|
|
—
|
|
Vice President & General
Counsel
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1) |
|
Pension benefits under the ZwitserLeven Contract GN 2595 and the
Finnish Managing Directors of AZC OY in Finland and EU accrue in
Euros, but for purposes of this table the present value of
accumulated benefits under those pension plans have been
converted to U.S. dollars based on the exchange rate in effect
on December 31, 2009 (1.43503 U.S. dollars per Euro). |
142
Potential
Payments Upon Termination or Change in Control
The following tables set forth the estimated value of payments
and benefits that our named executive officers would be entitled
to receive assuming certain terminations of employment
and/or a
change in control of Arizona Chemical, in each case, occurring
on December 31, 2009. The amounts set forth below are in
addition to the amounts they would be entitled to receive
pursuant to the pension plan benefits for our European-based
executives (Messrs. Verhaar, Tuovinen and Stuyfzand) and
any other generally available employee benefit plans (such as
disability, life insurance and accidental death).
Cornelis
Verhaar
Under the terms of Cornelis Verhaar’s August 18, 2008
employment agreement with Arizona Chemical B.V., in the event
Mr. Verhaar’s employment is terminated without
“cause”, he will be entitled to receive severance
benefits in the amount of two years’ of continued base
salary payments. Under his employment agreement,
“cause” is defined as failure to perform duties, gross
misconduct, conviction of a felony, breach of company policy or
fraud or misappropriation of company funds or property.
Mr. Verhaar’s entitlement to receive severance
benefits is conditioned on his continued compliance with certain
terms of his employment agreement, including a confidentiality
provision, which is effective both during and post-employment,
and non-solicitation and non-competition provisions, which apply
for two years following his termination. These provisions apply
regardless of the reason for Mr. Verhaar’s termination.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash-Out Value of
|
|
|
|
|
|
|
|
|
|
Equity Based Awards
|
|
|
|
|
|
|
|
Severance
|
|
that Vest Upon
|
|
Value of Benefits
|
|
|
|
Triggering Event
|
|
Payment
|
|
Triggering Event
|
|
Continuation
|
|
Total
|
|
|
|
By Company for cause or resignation by Executive
|
|
|
—
|
|
|
$
|
448,013
|
(3)
|
|
|
—
|
|
|
$
|
448,013
|
|
|
By Company without cause, pursuant to Company’s election
not to extend the employment term
|
|
$
|
1,198,006
|
(1)
|
|
$
|
500,000
|
(2)
|
|
$
|
40,000
|
(7)
|
|
$
|
1,738,006
|
|
|
Permanent Disability
|
|
$
|
1,018,305
|
(5)
|
|
$
|
500,000
|
(2)
|
|
$
|
95,429
|
(6)
|
|
$
|
1,613,734
|
|
|
Death
|
|
$
|
99,833
|
(4)
|
|
$
|
500,000
|
(2)
|
|
$
|
66,801
|
(6)
|
|
$
|
666,634
|
|
|
|
|
|
(1) |
|
Represents the continuation of base salary for 24 months. |
| |
|
(2) |
|
Represents total value of personal investment in equity plan,
which would be restored without penalty. |
| |
|
(3) |
|
Represents total value of personal investment in equity plan,
which may be restored without penalty at the discretion of the
general partner, but most likely would be restored minus loan
interest. |
| |
|
(4) |
|
Represents two months of salary, paid to spouse or beneficiary. |
| |
|
(5) |
|
Fully-insured disability benefit equivalent to 100% of
one-year’s salary and 70% of a second year’s salary. |
| |
|
(6) |
|
Represents the annual value of benefit continuation in the event
of permanent disability (for two years for the executive
and his spouse) and death (for two years for the
executive’s spouse). |
| |
|
(7) |
|
Represents 24 months of continued family medical and dental
coverage. |
Frederic
Jung
Under the terms of Frederic Jung’s November 5, 2008
employment letter with Arizona Chemical LLC, in the event
Mr. Jung’s employment is terminated by Arizona
Chemical without “cause” or Mr. Jung terminates
his employment for “good reason”, he will be entitled
to receive severance benefits in lump sum equal to one year of
base salary and pro-rata bonus under the MIP for the year
143
of termination. In addition, Mr. Jung and his family will
remain covered under the company’s health and dental
benefit plans for one year after termination and if he is on
foreign assignment at the time of his termination, Arizona
Chemical will relocate Mr. Jung and his family back to the
US. Under his employment agreement, (i) “cause”
is defined as a felony involving moral turpitude; willfully
engaging in fraud or dishonesty relating to his employment;
willful misconduct or gross negligence resulting in material
harm to the company; willful violation of company policies
resulting in material harm to the company; or willful failure to
substantially perform his duties; and (ii) “good
reason” means any adverse change in any material respect to
his title or reporting relationship; any reduction of target MIP
bonus or the annual base salary or the failure to be included on
similar terms and conditions in any employee executive benefit
plans, as peer-level executives of the company; or the
company’s failure to pay his annual base salary, employee
benefits or perquisites in accordance with the company’s
regular pay practices.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash-Out Value of
|
|
|
|
|
|
|
|
|
|
Equity Based Awards
|
|
|
|
|
|
|
|
Severance
|
|
that Vest Upon
|
|
Value of Benefits
|
|
|
|
Triggering Event
|
|
Payment
|
|
Triggering Event
|
|
Continuation
|
|
Total
|
|
|
|
By Company for cause or resignation by Executive
|
|
|
—
|
|
|
$
|
232,644
|
(4)
|
|
|
—
|
|
|
$
|
232,644
|
|
|
By Company without cause, pursuant to Company’s election
not to extend the employment term, or resignation by Executive
for good reason
|
|
$
|
456,000
|
(1)
|
|
$
|
250,000
|
(3)
|
|
$
|
13,114
|
(2)
|
|
$
|
719,114
|
|
|
Disability
|
|
|
—
|
|
|
$
|
250,000
|
(6)
|
|
$
|
170,976
|
(7)
|
|
$
|
420,976
|
|
|
Death
|
|
$
|
23,750
|
(8)
|
|
$
|
250,000
|
(6)
|
|
$
|
570,000
|
(5)
|
|
$
|
843,750
|
|
|
|
|
|
(1) |
|
Represents 12 months of salary and annual bonus at target
(full year). |
| |
|
(2) |
|
Represents 12 months of continued family medical and dental
coverage. |
| |
|
(3) |
|
Represents total value of personal investment in equity plan,
which would be restored without penalty. |
| |
|
(4) |
|
Represents total value of personal investment in equity plan,
which may be restored without penalty at the discretion of the
general partner, but most likely would be restored minus loan
interest. |
| |
|
(5) |
|
Represents life insurance lump sum payment of two times annual
salary, plus one month of salary. |
| |
|
(6) |
|
Represents payment for death and disability provision of equity
plan agreement. |
| |
|
(7) |
|
Annual disability benefit under the Company’s Long-Term
Disability Plan (U.S. only), a fully-insured benefit. |
| |
|
(8) |
|
Death benefit of one-month’s salary is paid to the spouse
or beneficiary. |
Juhani
Tuovinen
Under the terms of Mr. Tuovinen’s June 30, 2006
employment letter with Arizona Chemical B.V., in the event
Mr. Tuovinen’s employment is involuntarily terminated,
he will be entitled to receive severance benefits consistent
with the severance formula set from time to time by the Dutch
district courts (the Kantonrechtersformule). The Dutch court
formula provides for calculation of a severance payment equal to
the number of months of base salary equal to the employee’s
years of service calculated based on the sum of: (i) 0.5
months of salary for each year of service up to age 35,
(ii) 1 month of salary for each year of service
between ages 35 and 45, (iii) 1.5 months of salary for
144
each year of service between ages 45 and 55, and (iv) 2 months
of salary for each year of service after age 55.
Mr. Tuovinen’s entitlement to receive severance
benefits is conditioned on his continued compliance with certain
terms of his employment agreement, including a confidentiality
provision, which is effective both during and post-employment.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash-Out Value of
|
|
|
|
|
|
|
|
|
|
Equity Based Awards
|
|
|
|
|
|
|
|
Severance
|
|
that Vest Upon
|
|
Value of Benefits
|
|
|
|
Triggering Event
|
|
Payment
|
|
Triggering Event
|
|
Continuation
|
|
Total
|
|
|
|
By Company for cause or resignation by executive without good
reason
|
|
|
—
|
|
|
$
|
205,681(3
|
)
|
|
|
—
|
|
|
$
|
205,681
|
|
|
By Company without cause, pursuant to Company’s election
not to extend the employment term, or resignation by Executive
for good reason
|
|
$
|
1,291,308
|
(1)
|
|
$
|
260,000(2
|
)
|
|
|
—
|
|
|
$
|
1,551,308
|
|
|
Disability
|
|
$
|
506,402
|
(6)
|
|
$
|
260,000(4
|
)
|
|
$
|
71,397
|
(7)
|
|
$
|
837,799
|
|
|
Death
|
|
$
|
54,942
|
(5)
|
|
$
|
260,000(4
|
)
|
|
$
|
49,978
|
(7)
|
|
$
|
364,920
|
|
|
|
|
|
(1) |
|
Represents a severance payment based on the Dutch court formula
described above. |
| |
|
(2) |
|
Represents total value of personal investment in equity plan,
which would be restored without penalty. |
| |
|
(3) |
|
Represents total value of personal investment in equity plan,
which may be restored at the discretion of the general partner,
but most likely would be restored minus loan interest. |
| |
|
(4) |
|
Represents payment for death and disability provision of equity
plan agreement. |
| |
|
(5) |
|
Represents two months of salary, paid to spouse or beneficiary. |
| |
|
(6) |
|
Fully-insured disability benefit equivalent to 100% of
one-year’s salary and 70% of a second year’s salary. |
| |
|
(7) |
|
Represents the annual value of benefit continuation in the event
of permanent disability (for two years for the executive and his
spouse) and death (for two years for the executive’s
spouse). |
Gary
Reed
Mr. Reed does not have an employment agreement with Arizona
Chemical. In the event Mr. Reed’s employment is
involuntarily terminated by the company, he will be eligible to
receive severance benefits in accordance with the Arizona
Chemical Company, LLC US Salaried Employee Severance Plan, which
provides severance benefits contingent on an eligible
employee’s agreement to a release and waiver of claims in
favor of Arizona Chemical. Mr. Reed would be entitled to
receive a
145
lump sum severance payment equal to two weeks of pay for each
year or partial year of service and an annual bonus payment at
target.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash-Out Value of
|
|
|
|
|
|
|
|
|
|
Equity Based Awards
|
|
|
|
|
|
|
|
Severance
|
|
that Vest Upon
|
|
Value of Benefits
|
|
|
|
Triggering Event
|
|
Payment
|
|
Triggering Event
|
|
Continuation
|
|
Total
|
|
|
|
By Company for cause or resignation by Executive
|
|
|
—
|
|
|
$
|
158,216
|
(4)
|
|
|
—
|
|
|
$
|
158,216
|
|
|
By Company without cause, pursuant to Company’s election
not to extend the employment term
|
|
$
|
414,430
|
(1)
|
|
$
|
200,000
|
(3)
|
|
$
|
6,557
|
(2)
|
|
$
|
620,987
|
|
|
Disability
|
|
|
N/A
|
|
|
$
|
200,000
|
(6)
|
|
$
|
150,020
|
(7)
|
|
$
|
350,020
|
|
|
Death
|
|
$
|
20,833
|
(8)
|
|
$
|
200,000
|
(6)
|
|
$
|
520,833
|
(5)
|
|
$
|
741,666
|
|
|
|
|
|
(1) |
|
Represents service-related severance of two weeks’ pay for
each year or partial year of service and annual bonus at full
year target. |
| |
|
(2) |
|
Represents six months of continued family medical and dental
coverage. |
| |
|
(3) |
|
Represents total value of personal investment in equity plan,
which would be restored without penalty. |
| |
|
(4) |
|
Represents total value of personal investment in equity plan,
which may be restored without penalty at the discretion of the
general partner, but most likely would be restored minus loan
interest. |
| |
|
(5) |
|
Represents life insurance lump sum payment of two times annual
salary, plus one month of salary. |
| |
|
(6) |
|
Represents payment for death and disability provision of equity
plan agreement. |
| |
|
(7) |
|
Annual disability benefit under our Long-Term Disability Plan
(U.S. only), a fully-insured benefit. |
| |
|
(8) |
|
Death benefit of one-month salary is paid to spouse or
beneficiary. |
Dick
Stuyfzand
Mr. Stuyfzand entered into an employment agreement with
Arizona Chemical B.V. dated November 27, 2008. In the event
Mr. Stuyfzand’s employment is involuntarily
terminated, he will be entitled to receive severance benefits
consistent with the Dutch court formula described above.
Mr. Stuyfzand’s entitlement to receive severance
benefits is conditioned on his continued compliance with certain
terms of his employment agreement, including a confidentiality
provision, which is effective both during and post-employment.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash-Out Value of
|
|
|
|
|
|
|
|
|
|
Equity Based Awards
|
|
|
|
|
|
|
|
Severance
|
|
that Vest Upon
|
|
Value of Benefits
|
|
|
|
Triggering Event
|
|
Payment
|
|
Triggering Event
|
|
Continuation
|
|
Total
|
|
|
|
By Company for cause or resignation by Executive
|
|
|
—
|
|
|
$
|
96,102
|
(3)
|
|
|
—
|
|
|
$
|
96,102
|
|
|
By Company without cause or pursuant to Company’s election
not to extend the employment term
|
|
$
|
161,319
|
(1)
|
|
$
|
100,000
|
(2)
|
|
|
—
|
|
|
$
|
261,319
|
|
|
Disability
|
|
$
|
394,867
|
(6)
|
|
$
|
100,000
|
(4)
|
|
$
|
59,092
|
(7)
|
|
$
|
553,959
|
|
|
Death
|
|
$
|
38,712
|
(5)
|
|
$
|
100,000
|
(4)
|
|
$
|
41,356
|
(7)
|
|
$
|
180,068
|
|
|
|
|
|
(1) |
|
Represents a severance payment based on the Dutch court formula
described above. |
| |
|
(2) |
|
Represents total value of personal investment in equity plan,
which would be restored without penalty. |
146
|
|
|
|
(3) |
|
Represents total value of personal investment in equity plan,
which may be restored without penalty at the discretion of the
general partner, but most likely would be restored minus loan
interest. |
| |
|
(4) |
|
Represents payment for death and disability provision of equity
plan agreement. |
| |
|
(5) |
|
Represents two months of salary, paid to spouse or beneficiary. |
| |
|
(6) |
|
Fully-insured disability benefit equivalent to 100% of one
year’s salary and 70% of a second year’s salary. |
| |
|
(7) |
|
Represents the annual value of benefit continuation in the event
of permanent disability (for two years for the executive and his
spouse) and death (for two years for the executive’s
spouse). |
Director
Compensation
2009 Director
Compensation Table
The following table sets forth certain information concerning
annual compensation for our directors during the year ended
December 31, 2009.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Change in
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Pension Value
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
and
|
|
|
|
|
|
|
|
Fees
|
|
|
|
|
|
|
|
Nonqualified
|
|
|
|
|
|
|
|
Earned or
|
|
|
|
|
|
Non-equity
|
|
Deferred
|
|
|
|
|
|
|
|
Paid in
|
|
Stock
|
|
Option
|
|
Incentive Plan
|
|
Compensation
|
|
All Other
|
|
|
|
|
|
Cash
|
|
Awards
|
|
Awards
|
|
Compensation
|
|
Earnings
|
|
Compensation
|
|
Total
|
|
Name
|
|
($)
|
|
($)
|
|
($)
|
|
($)
|
|
($)
|
|
($)
|
|
($)
|
|
|
|
Leonard Berlik(1)
|
|
|
50,000
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
50,000
|
|
|
John R. Bolton(2)
|
|
|
—
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
—
|
|
|
Petter Johnsson(3)
|
|
|
—
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
—
|
|
|
Dr. Jochen Krautter
|
|
|
50,000
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
50,000
|
|
|
Gerald Marterer
|
|
|
50,000
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
50,000
|
|
|
Sebastien Mazella di Bosco(3)
|
|
|
—
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
—
|
|
|
Andrew Oliver(3)(4)
|
|
|
—
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
—
|
|
|
Eytan Tigay(3)
|
|
|
—
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
—
|
|
|
Cornelis Verhaar(3)
|
|
|
—
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
—
|
|
|
|
|
|
(1) |
|
In connection with his services as a member of the board of
managers of AZ Chem Investments LLC during the one year period
ended February 28, 2009, Mr. Berlik received “phantom
interests” in AZ Chem Investments Partners LP reflecting
the economic equivalent of a $50,000 investment in AZ Chem
Investments Partners LP. In connection with his services as a
member of the board of managers of AZ Chem Investments LLC
during the one year period ended February 28, 2010,
Mr. Berlik received $50,000 cash compensation. |
| |
|
(2) |
|
Mr. Bolton became a director in 2010. |
| |
|
(3) |
|
Directors who are employees or affiliates of Arizona Chemical
are not entitled to receive any compensation for serving on the
board of directors. |
| |
|
(4) |
|
Andrew Oliver ceased to be a director effective as of
May 21, 2010. |
Beginning in 2010, directors, other than directors who are
employees or affiliates of Arizona Chemical, receive an annual
retainer of $70,000 for serving on our board and the committees
of the board. The annual retainer for certain of our directors
will be paid 50% in cash and 50% in restricted common shares.
147
Other
Compensation Matters
MIV I and MIV
II
In June 2007, AZ Chem Investments LLC, which is the general
partner of AZ Chem Investments Partners LP, our parent, formed
AZ Chem MIV I Ltd., which we refer to as “MIV I”, and
AZ Chem MIV II LP, which we refer to as “MIV II” and,
together with MIV I, the “MIVs”. The MIVs were
formed to permit certain of our executives (including each of
our named executive officers) and members of our board of
directors to invest in our company on substantially the same
terms as the Rhône Funds and International Paper, and to
establish a vehicle to enable our executives to share in the
profits of our company based on future performance. As described
below, our executives purchased equity interests in the MIVs
and, as described above under “Executive
Compensation — Compensation Discussion and
Analysis — Components of Direct
Compensation — AZ Chem MIV I Ltd. and AZ Chem
MIV II LP”, each executive is eligible for additional
equity grants in the form of “common profits
interests”. Of the amount invested by each executive, 50%
was borrowed in the form of a loan. Each loan is secured by
interests in the respective MIV and remains outstanding and is
payable to AZ Chem Investments Partners LP upon the earlier of
(1) the date of a distribution from a sale or initial
public offering of AZ Chem Investments Partners LP or all of its
subsidiaries (including our company) or (2) the date that
the executive ceases to be a shareholder or limited partner in
the respective MIV.
Investments in Connection with Formation of MIV I and MIV
II. In connection with the formation of
MIV I, four of our senior executives, including
Mr. Tuovinen, invested $1.35 million to purchase
common and preferred shares of MIV I. Mr. Tuovinen invested
$520,000. Of the $1.35 million invested, 50% of that amount
was borrowed in the form of loans on the terms described above.
In connection with the formation of MIV II, 12 of our senior
executives, including Mr. Reed, invested $3.2 million
to purchase common and preferred interests in MIV II.
Mr. Reed invested $400,000. Of the $3.2 million
invested, 50% of that amount was borrowed in the form of loans,
on the terms described above. In addition, each shareholder in
MIV I and each limited partner in MIV II is eligible for grants
of common profits interests, which are described above under
“Executive Compensation — Compensation Discussion
and Analysis — Components of Direct
Compensation — AZ Chem MIV I Ltd. and AZ Chem
MIV II LP”.
Subsequent Investments by Officers in MIV I and MIV
II. In connection with their employment with our
company, each of Messrs. Verhaar and Stuyfzand purchased
common and preferred shares of MIV I ($1.0 million for
Mr. Verhaar and $200,000 for Mr. Stuyfzand). In
connection with his employment with our company, Mr. Jung
purchased $500,000 of common and preferred interests in MIV II.
Similar to the officers who invested in connection with the
formation of MIV I and MIV II, 50% of each investment was
borrowed in the form of a loan, on the terms described above. In
addition, each of these executives is eligible for grants of
common profits interests in the respective MIV, which are
described more fully below.
Participation in MIV II by Certain Members of our
Board. Mr. Marterer, who is a member of our
board of directors and who was our President and Chief Executive
Officer until his retirement in August 2008, invested
$1.0 million to purchase common and preferred interests in
MIV II in connection with its formation. Mr. Marterer
borrowed 50% of the funds used for his investment in the form of
a loan, on the terms described above and that loan also remains
outstanding. Mr. Marterer was granted common profits
interests in MIV II while he was an executive, on the same terms
as other executives. Messrs. Berlik and Krautter, members
of our board of directors, have invested $550,000 to purchase
common and preferred interests in MIV II and have not borrowed
any part of the funds used for their investments.
Messrs. Berlik and Krautter do not participate in grants of
common profits interests in MIV II.
Equity Participation of MIV I and MIV
II. Following this offering, MIV I and MIV II
will continue to be limited partners in AZ Chem Investments
Partners LP, which will be our largest shareholder. As limited
partners in AZ Chem Investments Partners LP, MIV I and MIV II
are entitled to participate in distributions, including
distributions following a secondary sale of our common shares by
AZ Chem Investments Partners LP, made by AZ Chem Investments
Partners LP, in accordance with the priority
148
of distributions summarized below. All distributions will be
made at the sole discretion of AZ Chem Investments LLC, as the
general partner of AZ Chem Investments Partners LP.
|
|
|
| |
•
|
First, a special preferred distribution to the Rhône Funds
in respect of their special preferred interests. The Rhône
Funds received these special preferred interests in 2008 in
respect of an
in-kind
capital contribution to AZ Chem Investments Partners LP of
$9.5 million of loans issued under our Second Lien Credit
Agreement, which were purchased by the Rhône Funds in the
secondary market. These loans were contributed by AZ Chem
Investments Partners LP to us, and were then extinguished by us.
|
| |
| |
•
|
Second, to the limited partners, including the Rhône Funds,
International Paper, MIV I and MIV II, in respect of their
preferred interests in AZ Chem Investments Partners LP, which
includes a preferred return that accumulates in respect of the
preferred interests. The initial investment by each limited
partner in MIV I and MIV II was allocated between a
preferred interest, which accounted for 90% of the investment
and accumulates a 10% annual preferred return, and a common
interest, which accounted for 10% of the investment and receives
distributions as described below. AZ Chem Investments Partners
LP intends to distribute the net proceeds from its sale of our
common shares in connection with this offering in respect of a
portion of the preferred interests.
|
| |
| |
•
|
Third, to the limited partners, including the Rhône Funds,
International Paper, MIV I and MIV II, in respect of the capital
represented by their common interests.
|
| |
| |
•
|
Fourth, to MIV I and MIV II in respect of their common profits
interests. The amount distributed in respect of the common
profits interests will depend on the amount of common profits
interests outstanding at that time, which will be based upon a
number of factors, including, among others, the amount of common
profits interests vested at such time and any subsequent
repurchases thereof. In addition, if each limited partner in AZ
Chem Investments Partners LP will have achieved an internal rate
of return on capital contributed to AZ Chem Investments Partners
LP of 30% or more, in certain circumstances, these distributions
on common profits interests are made at 150% of the amount
otherwise distributable. After taking into account the further
grant of common profits interests made in the first half of
2010, the common profits interests represent an approximately
5.8% common percentage in AZ Chem Investments Partners LP and
entitle the holders of those interests to up to 8.7% of any
distributions made in accordance with this priority level (if
the 30% internal rate of return on capital contributed to AZ
Chem Investments Partners LP has been achieved).
|
| |
| |
•
|
Fifth, to the limited partners, including the Rhône Funds,
International Paper, MIV I and MIV II, in respect of their
common percentages, which is calculated based upon the sum of
the common interests and the common profits interests, each as
described above, including the receipt of 150% of the amount
otherwise distributable on common profits interests in certain
circumstances.
|
As a result, subject to the discretion of AZ Chem Investments
LLC, as the general partner of AZ Chem Investments Partners LP,
which also has voting and disposition discretion over our common
shares owned by AZ Chem Investments Partners LP, the net cash
proceeds from sales of our common shares by AZ Chem Investments
Partners LP will be distributed to its partners as described
above. In addition, AZ Chem Investments LLC could choose to make
in-kind distributions of our common shares in accordance with
the priorities set forth above. All distributions received by an
MIV are in turn distributed to the shareholders or partners of
the MIV, including our named executive officers, and applied, to
the extent required by the loan documents, to pay AZ Chem
Investments Partners LP the outstanding principal balances on
the loans made to those officers for the purpose of purchasing
their preferred and common interests in the applicable MIV.
Based on expected capital account balances as of March 31,
2010, approximately $190 million would have to be
distributed in accordance with the first three distribution
priorities described above before any distributions would be
149
available for distribution to the common profits interests in
the fourth distribution priority. Distributions to our
executives will first be used to pay back the loans described
above.
Other Provisions Relating to the MIVs. Each of
the MIVs has the right to repurchase the interests of an
executive at any time the executive ceases to be a director,
officer, employee or consultant of our company. The price for
that redemption is equal to the fair market value of the
executive’s interest in the MIV, unless the service is
severed for cause, in which event the redemption price is equal
to the lesser of fair market value or capital invested, as
determined by AZ Chem Investments LLC, as the general partner of
AZ Chem Investments Partners LP, in its sole discretion. Any
outstanding loan, described above, would be due and payable in
connection with any redemption.
2010 Long-Term
Incentive Plan
Prior to the completion of this offering, we intend to adopt a
new long-term incentive plan, which we refer to as our 2010
Plan. The principal purpose of our 2010 Plan is to attract,
retain and motivate selected employees and non-employee
directors through the granting of share-based awards and
cash-based awards. The 2010 Plan will seek to optimize our
profitability and growth through incentives consistent with our
goals and that align the personal interests of the participants
with our shareholders.
The principal features of the 2010 Plan are summarized below.
This summary is qualified in its entirety by reference to the
text of the 2010 Plan, which will be filed as an exhibit to an
amendment to the registration statement of which this prospectus
forms a part.
Share Reserve. Under the 2010
Plan,
of our common shares will be initially reserved for issuance
pursuant to a variety of share-based compensation awards,
including share options, share appreciation rights, or SARs,
restricted share awards, restricted share unit awards,
performance units awards, performance shares awards, cash-based
performance awards, and other share-based awards.
The following counting provisions will be in effect for the
share reserve under the 2010 Plan:
|
|
|
| |
•
|
shares covered by an award will be counted as used only to the
extent they are actually delivered on exercise or settlement of
an award;
|
| |
| |
•
|
to the extent that an award is terminated by expiration,
forfeiture, cancellation or otherwise without the issuance of
the shares subject to the award or settled in cash in lieu of
shares or exchanged pursuant to the administrator’s
permission, prior to the issuance of shares, then the shares
covered by that award will be available for future grants under
the 2010 Plan;
|
| |
| |
•
|
to the extent shares are tendered or withheld to satisfy any
exercise price or tax withholding obligation with respect to an
award under the 2010 Plan, such tendered or withheld shares will
be available for future grants under the 2010 Plan;
|
| |
| |
•
|
to the extent any SAR is exercised and settled in shares, the
difference between the total shares exercised and the net shares
delivered will again be available for future grants under the
2010 plan; and
|
| |
| |
•
|
the payment of dividend equivalents in cash in conjunction with
any outstanding awards will not be counted against the shares
available for issuance under the 2010 Plan.
|
Individual Award Limits. Under the 2010 Plan,
awards granted in a calendar year to any one participant shall
be limited as follows:
|
|
|
| |
•
|
Share options/share appreciation rights: No more
than
common shares covered by share options and share appreciation
rights,
|
| |
| |
•
|
Restricted share/restricted share units: No more
than restricted
shares or restricted share units,
|
150
|
|
|
| |
•
|
Performance shares/units: No more
than shares
(if payable in shares) or the value of
$ if payable in cash,
|
| |
| |
•
|
Cash-based awards: No more than
$ , and
|
| |
| |
•
|
Other share-based awards: No more
than shares.
|
Administration. Our Compensation Committee
will be responsible for administering the 2010 Plan. The
Compensation Committee may employ attorneys, consultants,
accountants, agents and other individuals, any of whom may be
our employee, and the Compensation Committee and our officers
and board of directors will be entitled to rely upon the advice,
opinions or valuations of those individuals. The Compensation
Committee may delegate to one or more of its members or to one
or more of our officers or to one or more agents or advisors
such administrative duties or powers it may deem advisable.
However, the Compensation Committee will not be able to
delegate, to any officer, the responsibility for granting awards
to any of our employees who are considered insiders as defined
by the 2010 Plan. Moreover, only the board of directors or the
Compensation Committee can determine and approve awards made to
nonemployee directors.
Subject to the terms and conditions of the 2010 Plan, the
administrator generally will have the authority to select the
persons to whom awards will be made, to determine the number of
shares to be subject to awards and the terms and conditions of
awards and to make all other determinations and to take all
other actions necessary or advisable for the administration of
the 2010 Plan. The administrator will also be authorized to
adopt, amend or rescind rules relating to administration of the
2010 Plan.
Eligibility. Options, SARs, restricted shares
and all other share-based and cash-based awards under the 2010
Plan may be granted to individuals who are our officers or key
employees, as determined by the Compensation Committee, or are
officers or key employees of certain of our subsidiaries. Such
awards also may be granted to our nonemployee directors.
However, only officers and key employees may be granted
incentive share options, or ISOs, which are described below.
Awards. The 2010 Plan provides that the
administrator may grant or issue share options, SARs, restricted
share awards, restricted share unit awards, performance unit
awards, performance share awards, cash-based awards and other
share based awards, or any combination thereof. Each award will
be set forth in a separate agreement with the person receiving
the award and will indicate the type, terms and conditions of
the award.
|
|
|
| |
•
|
Nonqualified Share Options, or NQSOs, will provide for
the right to purchase our common shares at a specified price
which may not be less than fair market value on the date of
grant and usually will become exercisable (at the discretion of
the administrator) in one or more installments after the grant
date, subject to the participant’s continued employment or
service with us
and/or
subject to the satisfaction of corporate performance targets and
individual performance targets established by the administrator.
NQSOs may be granted for any term specified by the
administrator, but may not exceed ten years.
|
| |
| |
•
|
Incentive Share Options will be designed in a manner
intended to comply with the provisions of Section 422 of
the Internal Revenue Code and will be subject to specified
restrictions contained in the Internal Revenue Code. Among those
restrictions, ISOs must have an exercise price of not less than
the fair market value of a common share on the date of grant,
may only be granted to employees, must not be exercisable after
a period of ten years measured from the date of grant and will
retain their ISO status so long as they are exercised within
90 days after termination of employment (or within one year
after a disability termination). In the case of an ISO granted
to an individual who owns (or is deemed to own) at least 10% of
the total combined voting power of all classes of our common
shares, the 2010 Plan provides that the exercise price must be
at least 110% of the fair market value of a common share on the
date of grant and the ISO must not be exercisable after a period
of five years
|
151
|
|
|
| |
|
measured from the date of grant. Up
to of our common
shares under the 2010 Plan may be granted under ISOs.
|
|
|
|
| |
•
|
Share Appreciation Rights may be granted in connection
with share options or other awards, or separately. SARs granted
in connection with share options or other awards typically will
provide for payments to the holder based upon increases in the
price of our common shares over a set exercise price. The
exercise price of any SAR granted under the 2010 Plan must be at
least 100% of the fair market value of a common share on the
date of grant. The term of a SAR shall not exceed 10 years.
Except as required by Section 162(m) of the Internal
Revenue Code with respect to a SAR intended to qualify as
performance-based compensation as described in
Section 162(m) of the Internal Revenue Code, there are no
restrictions specified in the 2010 Plan on the exercise of SARs
or the amount of gain realizable therefrom, although
restrictions may be imposed by the administrator in the SAR
agreements.
|
| |
| |
•
|
Restricted Shares may be granted to any eligible
individual and made subject to such restrictions as may be
determined by the administrator. Restricted shares, typically,
may be forfeited for no consideration or repurchased by us at
the original purchase price (if any) if the conditions or
restrictions on vesting are not met. In general, restricted
shares may not be sold, or otherwise transferred, until
restrictions are removed or expire. Recipients of restricted
shares, unlike recipients of options, will have voting rights
and may receive dividends paid prior to the time when the
restrictions lapse.
|
| |
| |
•
|
Restricted Share Units may be awarded to any eligible
individual, typically without payment of consideration, but
subject to vesting conditions based on continued employment or
service or on performance criteria established by the
administrator. Like restricted shares, restricted share units
may not be sold, or otherwise transferred or hypothecated, until
vesting conditions are removed or expire. Unlike restricted
shares, shares underlying restricted share units will not be
issued until the restricted share units have vested, and
recipients of restricted share units will not have any voting
rights prior to the time when vesting conditions are satisfied.
|
| |
| |
•
|
Performance Awards may be granted by the administrator in
the form of performance units or performance shares. Generally,
these awards will be based upon specific performance targets and
may be paid in cash or in common shares or in a combination of
both. After the applicable performance period has ended, the
holder of performance units or performance shares shall be
entitled to receive a payout on the value and number of
performance units or performance shares earned by the
participant over the performance period, to be determined as a
function of the extent to which the corresponding performance
goals have been achieved.
|
| |
| |
•
|
Other Awards may be granted by the Compensation
Committee, either in the form of equity-based, equity-related,
or cash-based. If the Compensation Committee exercises its
discretion to establish performance goals, the number
and/or value
of cash-based awards or other share-based awards that will be
paid out to the recipient will depend on the extent to which the
performance goals are met.
|
| |
| |
•
|
Dividend Equivalents may be granted by the Compensation
Committee. Such dividend equivalents will be converted to cash
or additional shares by such formula and at such time and
subject to such limitations as may be determined by the
Compensation Committee. The Compensation Committee may not grant
dividend equivalents based on the dividends declared on shares
that are subject to an options, ISO, or SAR award; and
furthermore, no dividend or dividend equivalents will be paid
out with respect to any unvested performance awards.
|
Change in Control. In the event of involuntary
termination of employment within two years following a change in
control, outstanding options, SARs, restricted stock and
restricted stock units under the 2010 Plan will generally be
subject to accelerated vesting such that 100% of such award will
become vested and exercisable or payable, as applicable, unless
otherwise provided in the applicable award agreement. The
treatment of all other awards shall be determined by the terms
and
152
conditions set forth in the applicable award agreement, or in
the sole discretion of the Compensation Committee. Under the
2010 Plan, a change in control is generally defined as:
|
|
|
| |
•
|
the acquisition by any person of beneficial ownership of 30% or
more of either our then outstanding shares or the combined
voting power of our then outstanding voting securities;
|
| |
| |
•
|
individuals who constitute the continuing directors cease to
constitute at least a majority of the board;
|
| |
| |
•
|
consummation of a reorganization, amalgamation, merger,
consolidation, or business combination involving us, directly or
indirectly, other than a merger, consolidation, reorganization
or business combination which results in our outstanding voting
securities immediately before the transaction continuing to
represent a majority of the voting power of the acquiring
company’s outstanding voting securities and after which no
person or group beneficially owns 20% or more of the outstanding
voting securities of the surviving entity immediately after the
transaction;
|
| |
| |
•
|
the sale, exchange, or transfer of all or substantially all of
our assets; or
|
| |
| |
•
|
shareholder approval of our liquidation or dissolution.
|
Adjustments of Awards. In the event of any
corporate event or transaction, such as an amalgamation, merger,
consolidation, reorganization, recapitalization, right offering,
separation, partial or complete liquidation, share dividend,
share split, reverse share split, split up, spin-off, or other
distribution of our shares or property to a shareholder (other
than an extraordinary dividend), combination of shares, exchange
of shares, dividend in-kind, or other like change in capital
structure or distribution or any similar corporate event or
transaction affecting the number of our outstanding common
shares or the common share price that would require adjustments
to the 2010 Plan or any awards under the 2010 Plan in order to
prevent the dilution or enlargement of the potential benefits
intended to be made available thereunder, the administrator will
make appropriate, proportionate adjustments to:
|
|
|
| |
•
|
the aggregate number and type of shares subject to the 2010 Plan;
|
| |
| |
•
|
the terms and conditions of outstanding awards (including,
without limitation, any applicable performance targets or
criteria with respect to such awards and the number of shares
underlying outstanding awards); and
|
| |
| |
•
|
the grant or exercise price per share of any outstanding awards
under the 2010 Plan.
|
Amendment and Termination. Our board of
directors or the Compensation Committee (with board approval)
may terminate, amend or modify the 2010 Plan at any time and
from time to time. However, we must generally obtain shareholder
approval:
|
|
|
| |
•
|
to increase the number of shares available under the 2010 Plan
(other than in connection with certain corporate events, as
described above);
|
| |
| |
•
|
to grant options with an exercise price that is below 100% of
the fair market value of our common shares on the grant date;
|
| |
| |
•
|
to extend the exercise period for an option beyond ten years
from the date of grant; or
|
| |
| |
•
|
to the extent required by applicable law, rule or regulation
(including any applicable stock exchange rule).
|
Expiration Date. The 2010 Plan will expire on,
and no option or other award may be granted pursuant to the 2010
Plan after the
10th
anniversary of this offering. Any award that is outstanding on
the expiration date of the 2010 Plan will remain in force
according to the terms of the 2010 Plan and the applicable award
agreement.
153
PRINCIPAL AND
SELLING SHAREHOLDER
Beneficial ownership is determined in accordance with the rules
and regulations of the SEC. These rules generally provide that a
person is the beneficial owner of securities if such person has
or shares the power to vote or direct the voting thereof to
dispose or direct the disposition thereof or has the right to
acquire such powers within 60 days. Percentage of
beneficial ownership is based on (i) common shares
outstanding immediately prior to the completion of this offering
and (ii) common shares to be outstanding after the
completion of this offering, assuming no exercise of the
underwriters’ option to purchase additional shares,
or shares,
assuming full exercise of the option to purchase additional
shares. Except as disclosed in the footnotes to this table and
subject to applicable community property laws, we believe that
each shareholder identified in the table possesses sole voting
and investment power over all common shares shown as
beneficially owned by the shareholder.
The following tables set forth information regarding the
beneficial ownership of our common shares (i) immediately
prior to the completion of this offering and (ii) as
adjusted to give effect to this offering by each person or group
who is known by us to own beneficially more than 5% of our
outstanding common shares:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Shares Beneficially
|
|
Shares Beneficially
|
|
|
|
Shares Beneficially
|
|
|
|
Owned After this
|
|
Owned After this
|
|
|
|
Owned Before this
|
|
Shares
|
|
Offering Without
|
|
Offering with Exercise
|
Name and Address of
|
|
Offering
|
|
Offered
|
|
Exercise of Option
|
|
of Option
|
|
Beneficial Owner
|
|
Number
|
|
Percent
|
|
Hereby
|
|
Number
|
|
Percent
|
|
Number
|
|
Percent
|
|
|
|
AZ Chem Investments Partners
LP(1)(2)(3)(4)
|
|
|
|
|
|
|
100
|
%
|
|
|
|
|
|
|
|
|
|
|
|
%
|
|
|
|
|
|
|
|
%
|
|
c/o Rhône
Capital III, L.P.
630 5th Avenue,
New York, New York 10111
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1) |
|
AZ Chem Investments Partners LP is controlled by AZ Chem
Investments LLC, its general partner, which is managed by a
board the members of which are designated by Rhône
Partners III L.P., Rhône Coinvestment
Partners III L.P. and Rhône Offshore Partners III
L.P (together, the “Rhône Funds”). The Rhône
Funds are managed by Rhône Capital III L.P., which is
the general partner of each Rhône Fund. Rhône
Capital III L.P. is managed by Rhône Holdings III
LLC, its general partner, which is managed by Rhône
Capital, its sole member. Each of AZ Chem Investments LLC, the
Rhône Funds, Rhône Capital III L.P., Rhône
Holdings III LLC and Rhône Capital disclaim beneficial
ownership of the shares held by AZ Chem Investments Partners LP. |
| |
|
(2) |
|
The Rhône Funds, International Paper and AZ Chem MIV I Ltd.
and AZ Chem MIV II LP are limited partners in AZ Chem
Investments Partners LP. Prior to this offering, the Rhône
Funds held a % limited partnership
interest in AZ Chem Investments Partners LP, while International
Paper held a % limited partnership
interest and members of our management, through their interests
in AZ Chem MIV I Ltd. and AZ Chem MIV II LP, held
a % limited partnership interest. |
| |
|
(3) |
|
For information regarding the Acquisition, pursuant to which
Rhône Capital acquired Arizona Chemical from International
Paper, see “Business — Transactions with
Rhône Capital and International Paper”. For
information regarding transactions with Rhône Capital
subsequent to the Acquisition, see “Certain Relationships
and Related Party Transactions — Relationship with
Rhône Capital” and “Certain Relationships and
Related Party Transactions — Shareholders
Agreement”. |
| |
|
(4) |
|
AZ Chem Investments Partners LP is an affiliate of Rhône
Group Advisors LLC, a broker-dealer, and certifies that it
bought the securities in the ordinary course of business, and at
the time of the |
154
|
|
|
|
|
|
purchase of the securities to be resold it had no agreements or
understandings, directly or indirectly, with any person to
distribute the securities. |
The following table sets forth information regarding the
beneficial ownership of our common shares (i) immediately
prior to the completion of this offering and (ii) as
adjusted to give effect to this offering by:
|
|
|
| |
•
|
each of our named executive officers;
|
| |
| |
•
|
each of our directors and each director nominee; and
|
| |
| |
•
|
all of our executive officers, directors and director nominees
as a group.
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Shares Beneficially
|
|
Shares Beneficially
|
|
|
|
Shares Beneficially
|
|
|
|
Owned After this
|
|
Owned After this
|
|
|
|
Owned Before this
|
|
Shares
|
|
Offering Without
|
|
Offering with Exercise
|
Name and Address of
|
|
Offering
|
|
Offered
|
|
Exercise of Option
|
|
of Option
|
|
Beneficial Owner
|
|
Number
|
|
Percent
|
|
Hereby
|
|
Number
|
|
Percent
|
|
Number
|
|
Percent
|
|
|
|
Cornelis Verhaar(1)
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
%
|
|
Frederic Jung(1)
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
%
|
|
Juhani Tuovinen(1)
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
%
|
|
Gary Reed(1)
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
%
|
|
Dick Stuyfzand(1)
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
%
|
|
Leonard Berlik(1)(2)
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
%
|
|
John R. Bolton
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
%
|
|
Petter Johnsson
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
%
|
|
Jochen Krautter(1)
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
%
|
|
Gerald Marterer(1)
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
%
|
|
Sebastien Mazella di Bosco
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
%
|
|
Eytan Tigay
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
%
|
|
All executive officers and directors as a group:
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
%
|
|
|
—
|
|
|
|
—
|
%
|
|
|
|
|
(1) |
|
For a discussion of interests certain members of our management
and board of directors have in AZ Chem MIV I Ltd. and AZ Chem
MIV II LP, see “Executive Compensation —
Compensation Discussion and Analysis — Components of
Direct Compensation — AZ Chem MIV I Ltd. and AZ Chem
MIV II LP” and “Executive Compensation —
Other Compensation Matters — MIV I and
MIV II”. |
| |
|
(2) |
|
In connection with his services as a member of the board of
managers of AZ Chem Investments LLC for the one year periods
ended February 28, 2008 and February 28, 2009,
Mr. Berlik received “phantom interests” in AZ
Chem Investments Partners LP reflecting the economic equivalent
of a $100,000 investment in AZ Chem Investments Partners LP. |
155
CERTAIN
RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
Relationship with
Rhône Capital
As described under “Business — Transactions with
Rhône Capital and International Paper”, Rhône
Capital acquired us from International Paper on
February 28, 2007.
Prior to this offering, the Rhône Funds held
a % limited partnership interest in
AZ Chem Investments Partners LP, our parent, and control AZ Chem
Investments LLC, the general partner and manager of AZ Chem
Investments Partners LP. As a result, the Rhône Funds
control AZ Chem Investments Partners LP and therefore control us.
Management
Agreement
In connection with the Acquisition, we entered into a Management
Agreement with Rhône Group L.L.C., an affiliate of
Rhône Capital, pursuant to which Rhône Group L.L.C.,
in exchange for providing certain monitoring and other
management services and assistance, is entitled to receive a
management fee of €1,000,000 per year, payable
semi-annually on each January 1 and July 1 and
reimbursement of
out-of-pocket
expenses. Fees and expenses totaled $0.5 million,
$1.5 million, $2.0 million and $1.3 million for
the three-month period ended March 31, 2010, the years
ended December 31, 2009 and 2008 and the ten-month period
ended December 31, 2007, respectively. We currently expect
that the Management Agreement will be amended in connection with
this offering for a fee of €5.0 million payable by us
to Rhône Group L.L.C., which amendment will eliminate the
yearly management fee.
Settlement
Agreement
On November 17, 2009, we entered into a Settlement
Agreement and Mutual Release, which we refer to as the
“Settlement Agreement”, with AZ Chem Luxembourg
Holdings S.à r.l., our parent before giving effect to the
Reorganization and an affiliate of Rhône Capital, and
International Paper pursuant to which we and AZ Chem Luxembourg
Holdings S.à r.l. agreed to settle a number of claims
against International Paper in exchange for a payment to AZ Chem
Luxembourg Holdings S.à r.l. in the amount of
$1.7 million. In connection with our entry into the
Settlement Agreement, we amended and restated our CTO and CST
supply agreements with International Paper to clarify certain
provisions of the agreements and to modify the mechanism by
which the prices of BLS and CTO are determined.
Under the terms of the Settlement Agreement, in the event that
we make an
out-of-pocket
payment in respect of (i) the closure of a landfill
acquired from Stora Enso that is less than $773,000, or
(ii) the soil and groundwater contamination at our facility
in Sandarne, Sweden that is less than $250,000, we are required
to pay International Paper 50% of the difference between that
amount and our actual
out-of-pocket
costs for that claim.
Purchase and
Contribution of Our Indebtedness
In a series of transactions during the second half of 2008, the
Rhône Funds acquired approximately $27.2 million of
the debt outstanding under our Second Lien Credit Agreement. The
Rhône Funds contributed $9.5 million of this debt to
AZ Chem Investments Partners LP and then AZ Chem Investments
Partners LP contributed this debt to us in accordance with the
terms of our credit agreements, which debt was then
extinguished. In consideration for this contribution, the
Rhône Funds received special preferred equity interests in
AZ Chem Investments Partners LP. We intend to use a portion of
the proceeds of this offering to make a distribution to AZ Chem
Investments Partners LP, which it will use to redeem these
special preferred interests. See “Use of Proceeds”.
We pay the Rhône Funds interest on the second lien debt
they hold in accordance with the terms of our Second Lien Credit
Agreement. Since the Rhône Funds acquired this debt in
2008, the cumulative amount of interest paid to the Rhône
Funds was $1.7 million as of March 31, 2010.
156
Management
Loans
As described under “Executive Compensation”, two
management incentive vehicles, or MIVs, were established in
connection with the Acquisition to enable our management to
invest indirectly in our equity. Beginning in July 2007, we
provided loans to certain members of our management to assist
them in financing a portion of these investments. Each of these
loans bears interest at a rate of 7.88%.
On September 11, 2008, we loaned Cornelis Verhaar, our
President and Chief Executive Officer, $500,000 in connection
with his participation in the MIVs. On February 19, 2010,
this loan was assigned to AZ Chem Investments Partners LP for a
payment to us of $557,752, representing a repayment of principal
of $500,000 and a payment of accrued and unpaid interest of
$57,752.
On February 11, 2009, we loaned Frederic Jung, our Vice
President and Chief Financial Officer, $250,000 in connection
with his participation in the MIVs. On February 19, 2010,
this loan was assigned to AZ Chem Investments Partners LP for a
payment to us of $270,149, representing a repayment of principal
of $250,000 and a payment of accrued and unpaid interest of
$20,149.
On July 2, 2007, we loaned Juhani Tuovinen, our Vice
President and General Manager — Europe, $260,000 in
connection with his participation in the MIVs. On
February 19, 2010, this loan was assigned to AZ Chem
Investments Partners LP for a payment to us of $317,602,
representing a repayment of principal of $260,000 and a payment
of accrued and unpaid interest of $57,602.
On July 2, 2007, we loaned David Cowfer, our Vice
President, Human Relations and Corporate Communications,
$150,000 in connection with his participation in the MIVs. On
February 19, 2010, this loan was assigned to AZ Chem
Investments Partners LP for a payment to us of $183,232,
representing a repayment of principal of $150,000 and a payment
of accrued and unpaid interest of $33,232.
On July 2, 2007, we loaned Gary Reed, our Vice
President and General Manager — North America,
$200,000 in connection with his participation in the MIVs. On
February 19, 2010, this loan was assigned to AZ Chem
Investments Partners LP for a payment to us of $244,309,
representing a repayment of principal of $200,000 and a payment
of accrued and unpaid interest of $44,309.
On June 20, 2007, we loaned Gerald Marterer, a member of
our board of directors, $500,000 in connection with his
participation in the MIVs. On February 19, 2010, this loan
was assigned to AZ Chem Investments Partners LP for a payment to
us of $612,298, representing a repayment of principal of
$500,000 and a payment of accrued and unpaid interest of
$112,298.
Shareholders
Agreement
Pursuant to the terms of the limited partnership agreement of AZ
Chem Investments Partners LP, each of the Rhône Funds,
International Paper and each of the MIVs, which we refer to
below as the “Partners”, has the right to require that
AZ Chem Investments Partners LP distribute to such Partner its
pro rata portion of our shares held by AZ Chem
Investments Partners LP beginning 180 days after the
completion of this offering.
Prior to the completion of this offering, we will enter into a
shareholders agreement with the Partners that will provide,
among other matters, for piggyback registration rights following
a distribution of our common shares to the Partners at any time
that the Partners are not entitled to sell shares in accordance
with Rule 144 under the Securities Act. If at any time we
intend to file a registration statement under the Securities Act
(with certain exceptions) covering a primary or secondary
offering of any of our common shares, we will advise each party
to the shareholders agreement of its right to have its common
shares included in such registration. Upon receipt of a timely
request, we will include in the registration statement all of
our common shares that a party requests for inclusion in the
registration statement, and we will use commercially reasonable
efforts to effect the registration of those shares. Any sale of
our common shares in a piggyback registration will be subject to
customary cutback rights, giving priority to primary sales of
shares by us. However, if we determine not to proceed with a
proposed registration, then we may, at our election, give
written notice of that decision to the Partners and be relieved
of our registration obligation. The shareholders agreement will
impose additional restrictions on the ability of the MIVs to
exercise
157
piggyback registration rights. We will pay all registration
expenses in connection with a piggyback registration, including
all registration, filing and qualification fees, all printing
expenses and all listing fees, but excluding underwriting
discounts and commissions of selling shareholders and fees and
disbursements of counsel for selling shareholders.
Director and
Officer Indemnification
Section 98 of the Companies Act provides generally that a
Bermuda company may indemnify its directors, officers and
auditors against any liability which by virtue of any rule of
law would otherwise be imposed on them in respect of any
negligence, default, breach of duty or breach of trust, except
in cases where such liability arises from fraud or dishonesty of
which such director, officer or auditor may be guilty in
relation to the company. Section 98 further provides that a
Bermuda company may indemnify its directors, officers and
auditors against any liability incurred by them in defending any
proceedings, whether civil or criminal, in which judgment is
awarded in their favor or in which they are acquitted or granted
relief by the Supreme Court of Bermuda pursuant to
section 281 of the Companies Act.
We have adopted provisions in our bye-laws that provide that we
shall indemnify our officers and directors in respect of their
actions and omissions, except in respect of their fraud or
dishonesty. Our bye-laws provide that the shareholders waive all
claims or rights of action that they might have, individually or
in right of the Company, against any of the Company’s
directors or officers for any act or failure to act in the
performance of such director’s or officer’s duties,
except in respect of any fraud or dishonesty of such director or
officer. Section 98A of the Companies Act permits us to
purchase and maintain insurance for the benefit of any officer
or director in respect of any loss or liability attaching to him
in respect of any negligence, default, breach of duty or breach
of trust, whether or not we may otherwise indemnify such officer
or director.
We are in the process of obtaining directors’ and
officers’ insurance for some of our directors, officers,
affiliates, partners or employees for liabilities relating to
the performance of their duties. Such indemnification shall not
extend to any matter which would render it void pursuant to the
Companies Act.
Policy Concerning
Related Party Transactions
In connection with this offering, we will adopt a formal written
policy concerning related party transactions. Under the policy,
our employees, officers and directors will be discouraged from
entering into any transaction that may cause a conflict of
interest for us. In addition, they must report any potential
conflict of interest, including related party transactions, to
their supervisors or our law department. Pursuant to its
charter, our Audit Committee will be required to then evaluate
each related person transaction for the purpose of recommending
to the disinterested members of our board of directors that the
transactions are fair, reasonable and within our policy, and
should be ratified and approved by the board. In evaluating such
proposed transactions, the Audit Committee will be required to
consider the relevant facts and circumstances available and
deemed relevant to the Audit Committee, including: the benefits
of the transaction to our company; the terms of the transaction
and whether they are arm’s-length and in the ordinary
course of our company’s business; the direct or indirect
nature of the related person’s interest in the transaction;
the size and expected term of the transaction; and other facts
and circumstances that bear on the materiality of the related
person transaction under applicable law and listing standards.
Our Audit Committee will recommend approval of only those
transactions that, in light of known circumstances, are in, or
are not inconsistent with, our best interests, as our Audit
Committee determines in the good faith exercise of its
discretion.
158
DESCRIPTION OF
OUR INDEBTEDNESS
On February 28, 2007, in connection with the Acquisition,
(1) Arizona Chem Sweden Holdings AB and certain of its
European and U.S. subsidiaries entered into a First Lien
Credit and Guaranty Agreement with certain lenders and with
Goldman Sachs Credit Partners L.P. and Bank of America, N.A., as
agents (the “First Lien Credit Agreement”), and
(2) certain of our U.S. subsidiaries entered into a
Second Lien Credit and Guaranty Agreement with certain lenders
and with Goldman Sachs Credit Partners L.P. and CapitalSource
Finance LLC, as agents (the “Second Lien Credit
Agreement”). The First Lien Credit Agreement was amended as
of July 1, 2008, July 24, 2008 and November 14,
2008. The Second Lien Credit Agreement was amended as of
July 24, 2008. On May 28, 2010, we entered into
amendments to both our First Lien Credit Agreement and our
Second Lien Credit Agreement, which amendments become effective
upon completion of this offering.
First Lien Credit
Agreement
The First Lien Credit Agreement consists of (1) a five-year
$60.0 million revolving credit facility and
(2) six-year term loans in the original amount of (i)
€75.9 million to Arizona Chemical AB, our European
subsidiary borrower (the “European term loans”) and
(ii) $150 million to AZ Chem US Inc., our
U.S. subsidiary borrower (the “U.S. term
loans”). The revolving credit facility includes borrowing
capacity available for letters of credit and for swing line
loans (that is, borrowings on
same-day
notice). In addition, we may request the establishment of an
incremental term loan facility in an aggregate amount not to
exceed $75.0 million, subject to receipt of commitments
from the first lien term loan lenders or from other eligible
lending institutions acceptable to the administrative agent.
Interest Rate
and Fees
The borrowings under the First Lien Credit Agreement bear
interest at a rate equal to an applicable margin plus, as
specified in the First Lien Credit Agreement, either (1) a
base rate determined by reference to the highest of (i) the
prime rate (being the rate of interest quoted in The Wall Street
Journal, Money Rates Section as the prime rate) and
(ii) the federal funds rate plus
1/2
of 1%, or (2) a LIBOR rate determined by reference to the
costs of funds for deposits in the currency of such borrowing
for the interest period relevant to such borrowing adjusted for
certain additional costs.
The applicable margin for borrowings under the revolving credit
facility is 2.25% with respect to LIBOR borrowings, provided
that such margin will be reduced to (1) 2.00% where the
leverage ratio (as described below under
“— Certain Covenants and Events of Default”)
is less than 4.5x and equal to or greater than 4.0x,
(2) 1.75% where the leverage ratio is less than 4.0x and
equal to or greater than 3.5x, and (3) 1.50% where the
leverage ratio is less than 3.5x.
The applicable margin for borrowings under the revolving credit
facility that are base rate loans as well as for swing line
loans is equal to the applicable margin for revolving LIBOR
borrowings set forth in the preceding paragraph minus 1.00% per
annum.
The applicable margin for U.S. term loans that are LIBOR
borrowings is 2.00% per annum, and the applicable margin for
U.S. term loans that are base rate borrowings is 1.00% per
annum. The applicable margin for European term loans is 2.25%
per annum.
In addition to paying interest on outstanding principal under
the First Lien Credit Agreement, we are also required to pay the
lenders a commitment fee on unused commitments under the
revolving credit facility (taking into account letter of credit
usage) at a rate equal to either 0.50% per annum or, if our
leverage ratio is less than 4.0x, 0.375% per annum. We are also
required to pay letter of credit fees on unused letter of credit
drawings at the same rates as apply to unused commitments under
the revolving facility. During the existence of any payment
default under the First Lien Credit Agreement, the margin on all
obligations under the First Lien Credit Agreement shall increase
by 2% per annum.
159
Amortization
and Availability
The First Lien Credit Agreement requires us to continue making
quarterly amortization payments of the term loans in quarterly
amounts equal to
1/4
of 1% of the total outstanding principal amount for each quarter
through and including the quarter ending December 31, 2012.
The remaining principal balance of the term loans is payable at
the end of the sixth year in a lump-sum equal to the outstanding
principal at that time. The aggregate amount of the term loans
outstanding under the First Lien Credit Agreement as of
March 31, 2010 was $211.9 million.
No amounts were outstanding on the revolving credit facility at
March 31, 2010.
Prepayments
The First Lien Credit Agreement requires us, subject in each
case to certain exceptions, to prepay outstanding term loans in
an amount equal to:
|
|
|
| |
•
|
100% of the net cash proceeds of all asset sales and
dispositions by Arizona Chem Sweden Holdings AB and its
subsidiaries, subject to certain exceptions;
|
| |
| |
•
|
100% of the net cash proceeds from any payment received by
Arizona Chem Sweden Holdings AB and its subsidiaries in respect
of any casualty insurance claim or condemnation proceeding,
subject to certain exceptions;
|
| |
| |
•
|
100% of the net cash proceeds of issuances of certain debt
obligations by Arizona Chem Sweden Holdings AB and its
subsidiaries;
|
| |
| |
•
|
50% of the net cash proceeds from capital contributions to and
equity issuances by Arizona Chem Sweden Holdings AB or any of
its subsidiaries, including a step-down to 25% of net cash
proceeds where our leverage ratio is equal to or less than 3.5x
for the relevant period, provided that with respect to cash
proceeds relating to this offering, we are required to prepay
outstanding term loans in an amount equal to 75% of the net cash
proceeds; and
|
| |
| |
•
|
50% of Arizona Chem Sweden Holdings AB and its
subsidiaries’ annual excess cash flow (as defined in the
First Lien Credit Agreement) minus certain voluntary prepayments
during such period, including a step-down to 25% of excess cash
flow with respect to any period to the extent our leverage ratio
is equal to or less than 3.5x for such period.
|
Voluntary prepayments and commitment reductions are permitted in
whole or in part, without premium or penalty, subject to minimum
prepayment or reduction requirements.
Guaranty and
Security
All of our obligations under the First Lien Credit Agreement are
unconditionally guaranteed by substantially all of our existing
and subsequently acquired or organized subsidiaries. The
obligations under the First Lien Credit Agreement (including the
guarantees) are secured by substantially all of our present and
future assets and all present and future assets of each
guarantor, including but not limited to (1) a
first-priority pledge of all of our common shares and all of the
outstanding common shares owned by us or any guarantor in any
domestic subsidiary, (2) a first-priority pledge of 66% of
the outstanding capital stock owned by us or any guarantor in
any first-tier foreign subsidiary, and (3) perfected
first-priority security interests in all of our other present
and future assets and the other present and future assets of
each guarantor, subject to certain limited exceptions.
160
Certain
Covenants and Events of Default
The First Lien Credit Agreement contains customary
representations and warranties, subject to limitations and
exceptions, and customary covenants restricting our ability to,
among other things and subject to various exceptions:
|
|
|
| |
•
|
declare dividends, make distributions or redeem or repurchase
capital stock;
|
| |
| |
•
|
prepay, redeem or repurchase other debt;
|
| |
| |
•
|
incur liens or grant negative pledges;
|
| |
| |
•
|
make loans and investments and enter into acquisitions and joint
ventures;
|
| |
| |
•
|
incur additional indebtedness;
|
| |
| |
•
|
amend or otherwise alter or waive any material rights under any
organizational document, certain transaction documents relating
to the Acquisition or any permitted debt agreements;
|
| |
| |
•
|
make capital expenditures;
|
| |
| |
•
|
engage in mergers, acquisitions and asset sales;
|
| |
| |
•
|
engage in sale and lease-back transactions;
|
| |
| |
•
|
conduct transactions with affiliates;
|
| |
| |
•
|
alter the nature of our businesses; or
|
| |
| |
•
|
change our fiscal quarter or our fiscal year.
|
The First Lien Credit Agreement also contains certain financial
covenants based on our Adjusted EBITDA. A leverage ratio
covenant requires that we maintain a consolidated total debt to
Adjusted EBITDA ratio that does not exceed (a) 5.5x on the
last day of the fiscal quarter ending on December 31, 2009,
(b) 4.5x on the last day of each fiscal quarter ending on
March 31, June 30, September 30 and December 31,
2010, and (c) 4.0x on the last day of each subsequent
fiscal quarter beginning with the fiscal quarter ending
March 31, 2011. An interest coverage ratio covenant
requires that we maintain an Adjusted EBITDA to consolidated
interest expense ratio (in each case for the four preceding
fiscal quarter period) of greater than (x) 2.0x on the last
day of the fiscal quarter ending December 31, 2009,
(y) 2.25x on the last day of each fiscal quarter ending on
March 31, June 30, September 30 and December 31,
2010, and (z) 2.5x on the last day of each subsequent
fiscal quarter beginning with the fiscal quarter ending
March 31, 2011.
The First Lien Credit Agreement also contains certain customary
affirmative covenants. As of December 31, 2009, we were in
compliance in all material respects with all covenants and
provisions in the First Lien Credit Agreement.
Events of default under the First Lien Credit Agreement include,
but are not limited to:
|
|
|
| |
•
|
our failure to pay principal, interest, fees, reimbursement in
respect of any drawing under a letter of credit, or other
amounts under the First Lien Credit Agreement when due (taking
into account any applicable grace period);
|
| |
| |
•
|
any representation or warranty proving to have been materially
incorrect when made or deemed made;
|
| |
| |
•
|
with respect to certain covenants, covenant defaults (taking
into account any applicable grace period);
|
| |
| |
•
|
bankruptcy events;
|
| |
| |
•
|
a cross-default or, in certain circumstances, cross-acceleration
to certain other debt;
|
| |
| |
•
|
unsatisfied final judgments over certain specified thresholds;
|
161
|
|
|
| |
•
|
a change of control;
|
| |
| |
•
|
certain ERISA defaults; and
|
| |
| |
•
|
the invalidity or impairment of any loan document or any
security interest in relation to the First Lien Credit Agreement.
|
In addition, the First Lien Credit Agreement includes customary
provisions regarding breakage costs incurred in connection with
prepayments, changes in capital adequacy and capital
requirements or their interpretation, illegality, unavailability
and payments free and clear of withholding.
Borrowings under the First Lien Credit Agreement are subject to
the accuracy of representations and warranties (including the
absence of any material adverse effect on
and/or
material adverse development with respect to our business,
operations, properties, assets or condition) and the absence of
any defaults or events of default (including as a result of the
prospective additional borrowing).
Second Lien
Credit Agreement
The provisions of the Second Lien Credit Agreement, which
consists solely of a seven-year term loan in the original amount
of $125 million to AZ Chem US Inc., our
U.S. subsidiary borrower (the “second lien term
loans”), are substantially identical to the corresponding
provisions of the First Lien Credit Agreement described above
with the following principal exceptions:
|
|
|
| |
•
|
the agreement does not establish a revolving credit facility,
nor does it include
sub-facilities
for swing line loans and letters of credit or require payment of
any related fees;
|
| |
| |
•
|
no scheduled amortization payments are required;
|
| |
| |
•
|
the incremental term loan facility is for an amount of up to
only $25 million in the aggregate;
|
| |
| |
•
|
mandatory prepayments are required only at such time as all
amounts outstanding under the First Lien Credit Agreement have
been paid and all commitments thereunder have been terminated,
or otherwise with the consent of the first lien lenders,
provided that we are required to prepay the indebtedness
outstanding under our Second Lien Credit Agreement in an amount
equal to 25% of the net proceeds received from this offering;
|
| |
| |
•
|
the agreement does not require maintenance of a specified
interest coverage ratio although it does require maintenance of
a specified leverage ratio fixed at a level 0.25% higher than
the leverage ratio specified in the First Lien Credit
Agreement; and
|
| |
| |
•
|
the borrowings are guaranteed by, and secured by the assets of,
certain of our U.S. subsidiaries but not by Holdings, the
European borrower, or any of our other European subsidiaries.
|
Borrowings under the Second Lien Credit Agreement bear interest
at a rate equal to either (a) a base rate plus a margin of
4.50% per annum or (b) a LIBOR rate plus a margin of 5.50%
per annum, with each such rate being determined as described
above under “— First Lien Credit and Guaranty
Agreement — Interest Rate and Fees”.
As of March 31, 2010, the aggregate amount of term loans
outstanding under the Second Lien Credit Agreement was
$115.5 million, and we were in compliance in all material
respects with all covenants and provisions in the Second Lien
Credit Agreement.
162
DESCRIPTION OF
SHARE CAPITAL
The following summary is a description of the material terms of
our share capital. We have filed our certificate of
incorporation and memorandum of association and bye-laws as
exhibits to the registration statement of which this prospectus
forms a part. See “Where You Can Find More
Information”. The following summary also highlights
material differences between Bermuda and Delaware corporate laws.
General
We are an exempted company incorporated under the laws of
Bermuda. We are registered with the Registrar of Companies in
Bermuda under registration number 43973. We were incorporated on
February 12, 2010 under the name Arizona Chemical Ltd. Our
registered office is located at Clarendon House, 2 Church
Street, Hamilton, HM11, Bermuda.
Our authorized share capital consists
of common shares of par value
$0.01 per share and preference
shares of par value $0.01 per share.
Pursuant to our bye-laws, subject to the requirements of the
NYSE and to any resolution of the shareholders to the contrary,
our board of directors is authorized to issue any of our
authorized but unissued shares. There are no limitations on the
right of non-Bermudians or non-residents of Bermuda to hold or
vote our shares.
Prior to the completion of this offering, 100% of our common
shares will be held by AZ Chem Luxemburg Holdings S.à.r.l.,
a wholly-owned subsidiary of AZ Chem Investments Partners LP,
our beneficial owner. For information regarding our beneficial
ownership, see “Principal and Selling Shareholder”.
Common
Shares
Immediately following the completion of this offering, there
will be common shares issued and
outstanding. No preference shares will be issued or outstanding
at that time. All of our issued and outstanding common shares
prior to completion of this offering are and will be fully paid,
and all of our shares to be issued in this offering will be
issued fully paid. Holders of common shares have no pre-emptive,
redemption, conversion or sinking fund rights.
In the event of our liquidation, dissolution or winding up, the
holders of common shares are entitled to share equally and
ratably in our assets, if any, remaining after the payment of
all of our debts and liabilities, subject to any liquidation
preference on any issued and outstanding preference shares.
Voting
Holders of common shares are entitled to one vote per share on
all matters submitted to a vote of holders of common shares.
Unless a different majority is required by law or by our
bye-laws, resolutions to be approved by holders of common shares
require approval by a simple majority of votes cast at a meeting
at which a quorum is present. Holders of common shares will vote
together as a single class on all matters presented to the
shareholders for their vote or approval, including the election
of directors.
Any individual who is a shareholder of the Company and who is
present at a meeting may vote in person, as may any corporate
shareholder that is represented by a duly authorized
representative at a meeting of shareholders. Our bye-laws also
permit attendance at general meetings by proxy, provided the
instrument appointing the proxy is in the form specified in the
bye-laws or such other form as the board may determine.
The Companies Act also provides that shareholders may take
action by written consent. Subject to the following, anything
(except for the removal of an auditor before the expiration of
the term of his office or director before the expiration of the
term of his office) which may be done by resolution of the
Company in general meeting or by resolution of a meeting of any
class of the shareholders may,
163
without a meeting and without any previous notice being
required, be done by resolution in writing signed by, or in the
case of a shareholder that is a corporation whether or not a
company within the meaning of the Companies Act, on behalf of,
the shareholders who, at the date that the proposed resolution
in writing is circulated, would be entitled to attend a meeting
and vote on the resolution.
A resolution in writing may be signed by, or in the case of a
shareholder that is a corporation whether or not a company
within the meaning of the Companies Act, on behalf of, all the
shareholders, or all the shareholders of the relevant class
thereof, in as many counterparts as may be necessary. A
resolution in writing is effective once signed by all of the
shareholders on the date that the last shareholder signs the
resolution.
Dividends
We have not declared or paid any cash dividends on our common
shares since our inception. Other than the distribution we are
making to AZ Chem Investments Partners LP out of the
proceeds of this offering, we do not anticipate paying any cash
dividends on our issued capital for the foreseeable future. We
intend to retain all available funds and any future earnings to
reduce debt and fund the development and growth of our business.
Any future determination to pay dividends will be at the
discretion of our board of directors and will take into account:
|
|
|
| |
•
|
restrictions in our credit agreements;
|
| |
| |
•
|
general economic and business conditions;
|
| |
| |
•
|
our financial condition and results of operations;
|
| |
| |
•
|
our capital requirements and the capital requirements of our
subsidiaries;
|
| |
| |
•
|
the ability of our operating subsidiaries to pay dividends and
make distributions to us; and
|
| |
| |
•
|
such other factors as our board of directors may deem relevant.
|
We are a holding company and have no direct operations. As a
result, we will depend upon distributions from our subsidiaries
to pay any dividends.
Additionally, we are subject to Bermuda legal constraints that
may affect our ability to pay dividends on our common shares and
make other payments. Under the Companies Act, we may declare or
pay a dividend out of distributable reserves only if we have
reasonable grounds for believing that we are, or would after the
payment be, able to pay our liabilities as they become due and
if the realizable value of our assets would thereby not be less
than the aggregate of our liabilities, issued share capital and
share premium accounts. Issued share capital is the aggregate
par value of the company’s issued shares, and the share
premium account is the aggregate amount paid for issued shares
over and above their par value. Share premium accounts may be
reduced in certain limited circumstances. Under our bye-laws,
each common share is entitled to dividends if, as and when
dividends are declared by our board of directors, subject to any
preferred dividend right of the holders of any preference shares.
Preference
Shares
Pursuant to Bermuda law and our bye-laws, our board of directors
by resolution may establish one or more series of preference
shares having such number of shares, designations, dividend
rates, relative voting rights, conversion or exchange rights,
redemption rights, liquidation rights and other relative
participation, optional or other special rights, qualifications,
limitations or restrictions as may be fixed by the board of
directors without any further shareholder approval. Such rights,
preferences, powers and limitations as may be established could
also have the effect of discouraging an attempt to obtain
control of Arizona Chemical Ltd. We currently
have authorized preference
shares. We have no present plans to issue any preference shares.
See “Risk Factors — Risks Related to Holdings
164
Shares in a Bermuda Company — We May Issue Preference
Shares and Our Bye-Laws and Bermuda Law May Discourage
Takeovers, Which Could Affect the Rights of Holders of Our
Common Shares”.
Registration
Rights
The Rhône Funds, International Paper and the MIVs will have
certain share rights with respect to the common shares that they
will retain following this offering. See
“Shares Eligible for Future Sale” for a
discussion of the common shares that may be sold into the public
market in the future, and “Certain Relationships and
Related Party Transactions — Shareholders
Agreement” for a discussion of share rights.
Transfer Agent
and Registrar
The register of members will be maintained at the registered
office of the Company in Bermuda in accordance with Bermuda law,
and a branch register will be maintained in the United States by
Computershare Trust Company, N.A., who will serve as branch
registrar and transfer agent.
Lock-Up
Agreements
All current shareholders are subject to
lock-up
agreements under which they have agreed not to transfer or
dispose of, directly or indirectly, any common shares or any
securities convertible into or exercisable or exchangeable for
common shares of the Company, for a period of 180 days
after the date of this prospectus, which is subject to extension
in some circumstances. These agreements are discussed further in
“Shares Eligible for Future Sale — Sales of
Restricted Shares”.
Bermuda
Law
We are an exempted company organized under the laws of Bermuda.
The rights of our shareholders, including those persons who will
become shareholders in connection with this offering, are
governed by Bermuda law, our memorandum of association and our
bye-laws. The laws of Bermuda differ in some material respects
from laws generally applicable to U.S. corporations and
their stockholders. The following is a summary of material
provisions of Bermuda law and our organizational documents not
discussed above.
Variation of
Rights
If at any time we have more than one class of shares, the rights
attaching to any class, unless otherwise provided for by the
terms of issue of the relevant class, may be varied either:
(i) with the consent in writing of the holders of 75% of
the issued shares of that class; or (ii) with the sanction
of a resolution passed by a majority of the votes cast at a
general meeting of the relevant class of shareholders at which a
quorum consisting of at least two persons holding or
representing one-third of the issued shares of the relevant
class is present. Our bye-laws specify that the creation or
issue of shares ranking equally with existing shares will not,
unless expressly provided by the terms of issue of existing
shares, vary the rights attached to existing shares. In
addition, the creation or issue of preference shares ranking
prior to common shares will not be deemed to vary the rights
attached to common shares or, subject to the terms of any other
series of preference shares, to vary the rights attached to any
other series of preference shares.
Rights in
Liquidation
Under Bermuda law, in the event of a liquidation or
winding-up
of a company, after satisfaction in full of all claims and
creditors and subject to the preferential rights accorded to any
series of preference shares and subject to any specific
provisions of the company’s bye-laws, the proceeds of the
liquidation or
winding-up
are distributed pro rata among the holders of common
shares.
165
Meetings of
Shareholders
Under Bermuda law, a company is required to convene at least one
general meeting of shareholders each calendar year. Bermuda law
provides that a special general meeting of shareholders may be
called by the board of directors of a company and must be called
upon the request of shareholders holding not less than 10% of
the paid-up
capital of the company carrying the right to vote at general
meetings. Bermuda law also requires that shareholders be given
at least five days’ advance notice of a general meeting,
but the accidental omission to give notice to any person does
not invalidate the proceedings at a meeting. Our bye-laws
provide that our board of directors may convene an annual
general meeting or a special general meeting. Under our
bye-laws, we must give each shareholder at least
30 days’ notice of the annual general meeting and at
least 10 days’ notice of any special general meeting.
This notice requirement is subject to the ability to hold such
meetings on shorter notice if such notice is agreed: (i) in
the case of an annual general meeting by all of the shareholders
entitled to attend and vote at such meeting; or (ii) in the
case of a special general meeting by a majority in number of the
shareholders entitled to attend and vote at the meeting holding
not less than 95% in nominal value of the shares entitled to
vote at such meeting.
Under Bermuda law, the number of shareholders constituting a
quorum at any general meeting of shareholders is determined by
the bye-laws of a company. Our bye-laws provide that the
presence in person or by proxy of two or more shareholders
entitled to attend and vote and holding shares representing more
than 50% of the combined voting power constitutes a quorum.
Advance Notice
of Shareholder Proposals
Under Bermuda law, shareholders who wish to propose resolutions
for consideration at a meeting of shareholders must give at
least six weeks of advance notice of their proposals. Our
bye-laws provide that notice of shareholder proposals must be
given in writing to our secretary during a specific period prior
to the meeting at which the action is to be taken. Generally, to
be timely, notice must be received at our principal executive
offices (1) in the case of an annual general meeting, not
less than 90 days nor more than 120 days prior to the
first anniversary date of the annual general meeting for the
preceding year and (2) in the case of a special meeting,
not more than ten days following the day on which notice of the
special meeting was mailed or the date that the special meeting
is publicly announced (but in no event later than the day before
the meeting), whichever occurs first.
Access to
Books and Records and Dissemination of Information
Members of the general public have a right to inspect the public
documents of a company available at the office of the Registrar
of Companies in Bermuda. These documents include the
company’s certificate of incorporation, its memorandum of
association, including its objects and powers, and certain
alterations to the memorandum of association. The shareholders
have the additional right to inspect the bye-laws of the
company, minutes of general meetings and the company’s
audited financial statements, which must be presented to the
annual general meeting. The register of members of a company is
also open to inspection by shareholders and by members of the
general public without charge. The register of members is
required to be open for inspection for not less than two hours
in any business day (subject to the ability of a company to
close the register of members for not more than thirty days a
year). A company is required to maintain its share register in
Bermuda but may, subject to the provisions of the Companies Act,
establish a branch register outside of Bermuda. We maintain a
register of members at the registered office of the Company in
Hamilton, Bermuda. A company is required to keep at its
registered office a register of directors and officers that is
open for inspection for not less than two hours in any business
day by members of the public without charge. Bermuda law does
not, however, provide a general right for shareholders to
inspect or obtain copies of any other corporate records.
Classified
Board of Directors; Election and Removal of
Directors
Our board of directors is divided into three classes of
directors serving staggered three year terms. As a result,
approximately one-third of our board of directors will be
elected each year. The
166
number of directors that comprise our board of directors will be
determined by our board of directors. The board may change the
number of directors from time to time, subject to a minimum of
three and a maximum of 15 directors. Our board currently
has eight members. Directors may only be removed for cause, by
vote of shares representing a majority of the combined voting
power of all holders of common shares voting together as a
single class, provided notice of the shareholders meeting
convened to remove the director is given to the director. The
notice must contain a statement of the intention to remove the
director and a summary of the facts justifying the removal and
must be served on the director not less than fourteen days
before the meeting. The director is entitled to attend the
meeting and be heard on the motion for his removal. The
existence of a classified board of directors may deter a
shareholder from removing incumbent directors and simultaneously
gaining control of the board of directors by filling vacancies
with its own nominees.
Board
Actions
The bye-laws of the Company provide that its business is to be
managed and conducted by its board of directors. At common law,
members of a board of directors owe a fiduciary duty to the
company to act in good faith in their dealings with or on behalf
of the company and exercise their powers and fulfill the duties
of their office honestly. This duty includes the following
elements: (i) a duty to act in good faith in the best
interests of the company; (ii) a duty not to make a
personal profit from opportunities that arise from the office of
a director; (iii) a duty to avoid conflicts of interest;
and (iv) a duty to exercise powers for the purpose for
which such powers were intended.
The Companies Act also imposes a duty on directors and officers
of a Bermuda company to: (i) act honestly and in good faith
with a view to the best interests of the company; and
(ii) exercise the care, diligence and skill that a
reasonably prudent person would exercise in comparable
circumstances.
Bermuda law requires that our directors be individuals, but
there is no requirement in our bye-laws or Bermuda law that
directors hold any of our shares. There is also no requirement
in our bye-laws or Bermuda law that our directors must retire at
a certain age.
The remuneration of our directors is determined by the board.
Our directors may also be paid all travel, hotel and other
expenses properly incurred by them in connection with our
business or their duties as directors.
Provided a director discloses a direct or indirect interest in
any contract or arrangement with us as required by Bermuda law,
such director is entitled to vote in respect of any such
contract or arrangement in which he or she is interested unless
he or she is disqualified from voting by the chairman of the
relevant board meeting. A director (including the spouse or
children of the director or any company of which such director,
spouse or children own or control more than 20% of the capital
or loan debt) can not borrow from us (except loans made to
directors who are bona fide employees or former employees
pursuant to an employees’ share scheme), unless
shareholders holding 90% of the total voting rights have
consented to the loan.
Transfer of
Shares
Our board of directors may in its absolute discretion and
without assigning any reason refuse to register the transfer of
a share that is not fully paid. Our board of directors may also
refuse to recognize an instrument of transfer of a share unless
it is accompanied by the relevant share certificate and such
other evidence of the transferor’s right to make the
transfer as our board of directors shall reasonably require.
Subject to these restrictions, a holder of common shares may
transfer the title to all or any of his common shares by
completing a form of transfer in the form set out in our
bye-laws (or as near thereto as circumstances admit) or in such
other common form as the board may accept. The instrument of
transfer must be signed by the transferor and transferee,
although in the case of a fully paid share our board of
directors may accept the instrument signed only by the
transferor.
167
Indemnification
of Directors and Officers
Section 98 of the Companies Act provides generally that a
Bermuda company may indemnify its directors, officers and
auditors against any liability which by virtue of any rule of
law would otherwise be imposed on them in respect of any
negligence, default, breach of duty or breach of trust, except
in cases where such liability arises from fraud or dishonesty of
which such director, officer or auditor may be guilty in
relation to the company. Section 98 further provides that a
Bermuda company may indemnify its directors, officers and
auditors against any liability incurred by them in defending any
proceedings, whether civil or criminal, in which judgment is
awarded in their favor or in which they are acquitted or granted
relief by the Supreme Court of Bermuda pursuant to
section 281 of the Companies Act.
We have adopted provisions in our bye-laws that provide that we
shall indemnify our officers and directors in respect of their
actions and omissions, except in respect of their fraud or
dishonesty. Our bye-laws provide that the shareholders waive all
claims or rights of action that they might have, individually or
in right of the Company, against any of the Company’s
directors or officers for any act or failure to act in the
performance of such director’s or officer’s duties,
except in respect of any fraud or dishonesty of such director or
officer. Section 98A of the Companies Act permits us to
purchase and maintain insurance for the benefit of any officer
or director in respect of any loss or liability attaching to him
in respect of any negligence, default, breach of duty or breach
of trust, whether or not we may otherwise indemnify such officer
or director. We are in the process of purchasing a
directors’ and officers’ liability policy for such a
purpose.
Amendment of
Memorandum of Association and Bye-Laws
Bermuda law provides that the memorandum of association of a
company may be amended by a resolution passed at a general
meeting of shareholders of which due notice has been given. Our
bye-laws provide that no bye-law shall be rescinded, altered or
amended, and no new bye-law shall be made, unless it shall have
been approved by a resolution of our board of directors and by a
resolution of our shareholders.
Under Bermuda law, the holders of an aggregate of not less than
20% in par value of a company’s issued share capital or any
class thereof have the right to apply to the Supreme Court of
Bermuda for an annulment of any amendment of the memorandum of
association adopted by shareholders at any general meeting,
other than an amendment which alters or reduces a company’s
share capital as provided in the Companies Act. Where such an
application is made, the amendment becomes effective only to the
extent that it is confirmed by the Bermuda court. An application
for an annulment of an amendment of the memorandum of
association must be made within 21 days after the date on
which the resolution altering the company’s memorandum of
association is passed and may be made on behalf of the persons
entitled to make the application by one or more of their number
as they may appoint in writing for the purpose. No application
may be made by shareholders voting in favor of the amendment.
Amalgamations
and Appraisal Rights
A Bermuda exempted company may acquire the business of another
Bermuda exempted company or a company incorporated outside
Bermuda when the business of the target company is within the
acquiring company’s objects as set forth in its memorandum
of association.
Under Bermuda law, in the event of an amalgamation of a Bermuda
company with another company, a shareholder of the Bermuda
company who did not vote in favor of the amalgamation and who is
not satisfied that fair value has been offered for his or her
shares in the Bermuda company may within one month of notice of
the shareholders meeting, apply to the Supreme Court of Bermuda
to appraise the fair value of his or her shares. Under Bermuda
law and our bye-laws, the amalgamation of Arizona Chemical Ltd.
with another company or corporation (other than certain
affiliated companies) requires an amalgamation agreement to
first be approved and then recommended by our board of directors
and by resolution of our shareholders. Our bye-laws provide that
a simple majority of the shareholders voting at such meeting is
required to approve the amalgamation agreement.
168
Shareholder
Suits
Class actions and derivative actions are generally not available
to shareholders under Bermuda law. The Bermuda courts, however,
would ordinarily be expected to permit a shareholder to commence
an action in the name of a company to remedy a wrong done to the
company where the act complained of is alleged to be beyond the
corporate power of the company or is illegal or would result in
violation of the company’s memorandum of association or
bye-laws. Furthermore, consideration would be given by a Bermuda
court to acts that are alleged to constitute a fraud against the
minority shareholders or, for instance, where an act requires
the approval of a greater percentage of the company’s
shareholders than that which actually approved it.
When the affairs of a company are being conducted in a manner
which is oppressive or prejudicial to the interests of some part
of the shareholders, one or more shareholders may apply to the
Supreme Court of Bermuda which may make such order as it sees
fit, including an order regulating the conduct of the
company’s affairs in the future or ordering the purchase of
the shares of any shareholder, by other shareholders or by the
company.
Our bye-laws contain a provision by virtue of which our
shareholders waive any claim or right of action that they have,
both individually and on our behalf, against any director or
officer in relation to any action or failure to take action by
such director or officer, except in respect of any fraud or
dishonesty of such director or officer.
Discontinuance
Under Bermuda law, an exempted company may be discontinued and
be continued in a jurisdiction outside Bermuda as if it had been
incorporated under the laws of that other jurisdiction. Our
bye-laws provide that our board of directors may exercise all
our power to discontinue to another jurisdiction without the
need of any shareholder approval.
Takeovers/Compulsory
Acquisition of Shares Held by Minority
Holders
An acquiring party is generally able to acquire compulsorily the
common shares of minority holders in the following ways:
|
|
|
| |
•
|
By a procedure under the Companies Act 1981 known as a
“scheme of arrangement”. A scheme of arrangement could
be effected by obtaining the agreement of the Company and of
holders of common shares, representing in the aggregate a
majority in number and at least 75% in value of the common
shareholders present and voting at a court ordered meeting held
to consider the scheme or arrangement. The scheme of arrangement
must then be sanctioned by the Bermuda Supreme Court. If a
scheme of arrangement receives all necessary agreements and
sanctions, upon the filing of the court order with the Registrar
of Companies in Bermuda, all holders of common shares could be
compelled to sell their shares under the terms of the scheme or
arrangement.
|
| |
| |
•
|
If the acquiring party is a company it may compulsorily acquire
all the shares of the target company, by acquiring pursuant to a
tender offer 90% of the shares or class of shares not already
owned by, or by a nominee for, the acquiring party (the
offeror), or any of its subsidiaries. If an offeror has, within
four months after the making of an offer for all the shares or
class of shares not owned by, or by a nominee for, the offeror,
or any of its subsidiaries, obtained the approval of the holders
of 90% or more of all the shares to which the offer relates, the
offeror may, at any time within two months beginning with the
date on which the approval was obtained, require by notice any
nontendering shareholder to transfer its shares on the same
terms as the original offer. In those circumstances,
nontendering shareholders will be compelled to sell their shares
unless the Supreme Court of Bermuda (on application made within
a one-month period from the date of the offeror’s notice of
its intention to acquire such shares) orders otherwise.
|
| |
| |
•
|
Where one or more parties holds not less than 95% of the shares
or a class of shares of a company, such holder(s) may, pursuant
to a notice given to the remaining shareholders or class of
|
169
|
|
|
| |
|
shareholders, acquire the shares of such remaining shareholders
or class of shareholders. When this notice is given, the
acquiring party is entitled and bound to acquire the shares of
the remaining shareholders on the terms set out in the notice,
unless a remaining shareholder, within one month of receiving
such notice, applies to the Supreme Court of Bermuda for an
appraisal of the value of their shares. This provision only
applies where the acquiring party offers the same terms to all
holders of shares whose shares are being acquired.
|
Delaware
Law
The terms of share capital of corporations incorporated in the
United States, including Delaware, differ from corporations
incorporated in Bermuda. The following discussion highlights
material differences of the rights of a shareholder of a
Delaware corporation compared with the rights of our
shareholders under Bermuda law, as outlined above.
Under Delaware law, a corporation may indemnify its director or
officer (other than in action by or in the right of the
corporation) against expenses (including attorneys’ fees),
judgments, fines and amounts paid in settlement actually and
reasonably incurred in defense of an action, suit or proceeding
by reason of such position if such director or officer
(i) acted in good faith and in a manner he reasonably
believed to be in, or not opposed to, the best interests of the
corporation and (ii) with respect to any criminal action or
proceeding, had no reasonable cause to believe his or her
conduct was unlawful.
Delaware law provides that a majority of the shares entitled to
vote, present in person or represented by proxy, constitutes a
quorum at a meeting of shareholders. In matters other than the
election of directors, with the exception of special voting
requirements related to extraordinary transactions, the
affirmative vote of a majority of shares present in person or
represented by proxy at the meeting and entitled to vote is
required for shareholder action, and the affirmative vote of a
plurality of shares is required for the election of directors.
With certain exceptions, a merger, consolidation or sale of all
or substantially all the assets of a corporation must be
approved by the board of directors and a majority of the
outstanding shares entitled to vote thereon. Under Delaware law,
a shareholder of a corporation participating in certain major
corporate transactions may, under certain circumstances, be
entitled to appraisal rights pursuant to which such shareholder
may receive cash in the amount of the fair value of the shares
held by such shareholder (as determined by a court) in lieu of
the consideration such shareholder would otherwise receive in
the transaction.
Under Delaware law, subject to any restrictions contained in the
company’s certificate of incorporation, a company may pay
dividends out of surplus or, if there is no surplus, out of net
profits for the fiscal year in which the dividend is declared
and for the preceding fiscal year. Delaware law also provides
that dividends may not be paid out of net profits if, after the
payment of the dividend, capital is less than the capital
represented by the outstanding shares of all classes having a
preference upon the distribution of assets.
Delaware law permits corporations to have a classified board of
directors. Delaware law permits the board of directors or any
person who is authorized under a corporation’s certificate
of incorporation or by-laws to call a special meeting of
shareholders. Under Delaware law, the business and affairs of a
corporation are managed by or under the direction of its board
of directors. In exercising their powers, directors are charged
with a fiduciary duty of care to protect the interests of the
corporation and a fiduciary duty of loyalty to act in the best
interests of its shareholders.
Delaware law permits any shareholder to inspect or obtain copies
of a corporation’s shareholder list and its other books and
records for any purpose reasonably related to such person’s
interest as a shareholder.
Class actions and derivative actions generally are available to
shareholders under Delaware law for, among other things, breach
of fiduciary duty, corporate waste and actions not taken in
accordance with applicable law, and the court generally has
discretion in such actions to permit the winning party to
recover attorneys’ fees.
170
SHARES ELIGIBLE
FOR FUTURE SALE
Prior to this offering, there has been no market for our common
shares, and we cannot assure you that a liquid trading market
for our common shares will develop or be sustained after this
offering. Future sales of substantial amounts of common shares,
including shares issued upon exercise of options and warrants,
in the public market after this offering, or the anticipation of
those sales, could adversely affect market prices prevailing
from time to time and could impair our ability to raise capital
through sales of our equity securities. Immediately prior to the
closing of the offering, we will have one holder of common
shares, AZ Chem Investments Partners LP.
Sales of
Restricted Shares
Upon the closing of this
offering, we will have outstanding an aggregate of approximately
common
shares. Of these
shares, common
shares to be sold in this offering,
or common
shares if the underwriters exercise their option to purchase
additional shares in full, will be freely tradable without
restriction or further registration under the Securities Act,
unless the shares are held by any of our affiliates, as that
term is defined in Rule 144 of the Securities Act. All
remaining shares were issued and sold by us in private
transactions and are eligible for public sale only if registered
under the Securities Act or sold in accordance with
Rule 144, which is discussed below.
AZ Chem Investments Partners LP, the holder of all of our
currently outstanding shares, is subject to a
lock-up
agreement under which it has agreed not to transfer or dispose
of, directly or indirectly, any common shares or any securities
convertible into or exercisable or exchangeable for common
shares, for a period of 180 days after the date of this
prospectus, which is subject to extension in some circumstances,
as discussed below.
As a result of the
lock-up
agreement described below and the provisions of Rule 144
under the Securities Act, we expect the
remaining of
our common shares to become eligible for future sale in the
public market pursuant to Rule 144 at varying times after
six months from the date of this prospectus.
Rule 144
In general, under Rule 144, beginning 90 days after
the date of this prospectus, a person who is not our affiliate,
has not been our affiliate for the previous three months and who
has beneficially owned our common shares for at least six months
may sell all such shares. An affiliate or a person who has been
our affiliate within the previous 90 days, and who has
beneficially owned our common shares for at least six months,
may sell within any three-month period a number of shares that
does not exceed the greater of:
|
|
|
| |
•
|
one percent of the number of common shares then outstanding,
which will equal
approximately shares
immediately after this offering; and
|
| |
| |
•
|
the average weekly trading volume of the common shares on the
New York Stock Exchange during the four calendar weeks preceding
the filing of a notice on Form 144 with respect to the sale.
|
All such sales under Rule 144 are subject to the
availability of current public information about us. Sales under
Rule 144 by affiliates or persons who have been affiliates
within the previous 90 days are also subject to manner of
sale provisions and notice requirements. Upon completion of the
180-day
lock-up
period, subject to any extension of the
lock-up
period under circumstances described below,
approximately shares
of our outstanding restricted securities will be eligible for
sale under Rule 144.
Lock-Up
Agreements
We and the selling shareholder have agreed, subject to certain
exceptions, with the underwriters not to dispose of or hedge any
of the common shares or securities convertible into or
exchangeable for common shares during the period from the date
of this prospectus continuing through the date 180 days
after the date of this prospectus, subject to certain
exceptions. Goldman, Sachs & Co. may, in its sole
discretion release any of these shares from these restrictions
at any time without notice. See “Underwriting”.
171
MATERIAL BERMUDA
AND UNITED STATES FEDERAL INCOME TAX CONSIDERATIONS
Bermuda Tax
Considerations
At the present time, there is no Bermuda income or profits tax,
withholding tax, capital gains tax, capital transfer tax, estate
duty or inheritance tax payable by us or by our shareholders in
respect of our common shares. We have obtained an assurance from
the Minister of Finance of Bermuda under the Exempted
Undertakings Tax Protection Act 1966 that, in the event that any
legislation is enacted in Bermuda imposing any tax computed on
profits or income, or computed on any capital asset, gain or
appreciation or any tax in the nature of estate duty or
inheritance tax, such tax shall not, until March 28, 2016,
be applicable to us or to any of our operations or to our
shares, debentures or other obligations except insofar as such
tax applies to persons ordinarily resident in Bermuda or is
payable by us in respect of real property owned or leased by us
in Bermuda.
United States
Federal Income Tax Considerations
Certain Issues
Relating to Our U.S. Federal Income Tax Treatment
This section describes the material United States federal income
tax consequences of owning and disposing of the common shares.
This discussion does not purport to be a comprehensive
description of all tax considerations that may be relevant to
you as owner of the shares. In particular, it applies to you
only if you acquire your shares in this offering and you hold
your shares as capital assets for tax purposes. This discussion
addresses only United States federal income tax consequences and
does not address the tax treatment of the ownership and
disposition of the shares under applicable state or local laws
of any jurisdiction. This section does not apply to you
(i) with respect to shares which you already own at the
time of the offering; in particular, this section does not
discuss the tax consequences of the restructuring which was
completed immediately prior to this offering (as described under
“Business — Corporate Structure and
Reorganization” above; the “Restructuring”) to an
existing holder of shares at the time of the Restructuring, or
(ii) if you are a member of a special class of holders
subject to special rules, including:
|
|
|
| |
•
|
a dealer in securities;
|
| |
| |
•
|
a trader in securities that elects to use a
mark-to-market
method of accounting for securities holdings;
|
| |
| |
•
|
a tax-exempt organization;
|
| |
| |
•
|
a financial institution;
|
| |
| |
•
|
a life insurance company;
|
| |
| |
•
|
a person liable for alternative minimum tax;
|
| |
| |
•
|
a person that actually or constructively owns 10% or more of our
voting shares;
|
| |
| |
•
|
a person that holds shares as part of a straddle or a hedging or
conversion transaction;
|
| |
| |
•
|
a U.S. expatriate; or
|
| |
| |
•
|
a U.S. Holder (as defined below) whose functional currency
is not the U.S. dollar.
|
This section is based on the Internal Revenue Code of 1986 (the
“Code”), as amended, its legislative history, existing
and proposed regulations, published rulings and court decisions,
all as currently in effect. These laws are subject to change,
possibly on a retroactive basis. There is currently no
comprehensive income tax treaty between the United States and
Bermuda.
If a partnership holds the shares, the United States federal
income tax treatment of a partner will generally depend on the
status of the partner and the tax treatment of the partnership.
A partner in a partnership holding the shares should consult its
tax advisor with regard to the United States federal income tax
treatment of an investment in the shares.
172
You are a U.S. Holder if you are a beneficial owner of
shares and you are:
|
|
|
| |
•
|
a citizen or resident of the United States;
|
| |
| |
•
|
a domestic corporation;
|
| |
| |
•
|
an estate whose income is subject to United States federal
income tax regardless of its source; or
|
| |
| |
•
|
a trust if it (i) is subject to the primary supervision of a
court within the United States and one or more United States
persons have the authority to control all substantial decisions
of the trust or (ii) has a valid election in effect under
applicable United States Treasury regulations to be treated as a
United States person.
|
A
“non-U.S. Holder”
is a beneficial owner of shares that is not a United States
Holder.
You should consult your own tax advisor regarding the United
States federal, state and local and the Bermuda and other tax
consequences of owning and disposing of shares in your
particular circumstances.
Taxation of
Distributions
U.S. Holders. Subject to the passive
foreign investment company (or “PFIC”) rules discussed
below, the gross amount of any distribution we pay out of our
current or accumulated earnings and profits (as determined for
United States federal income tax purposes) is subject to United
States federal income taxation as ordinary income. If you are a
noncorporate U.S. Holder, dividends paid to you in taxable
years beginning before January 1, 2011 that constitute
qualified dividend income will be taxable to you at a maximum
tax rate of 15% provided that you hold the shares for more than
60 days during the
121-day
period beginning 60 days before the ex-dividend date and
meet other holding period requirements. Dividends we pay with
respect to the shares generally will be qualified dividend
income, provided that, in the year that you receive the
dividend, the shares are readily tradable on an established
securities market in the United States. Our common shares will
be listed on the NYSE, which is an established securities market
in the United States. Absent new legislation extending the
current rates, beginning on January 1, 2011 dividends will
be subject to ordinary income rates.
The dividend is taxable to you when you receive it, actually or
constructively. The dividend will not be eligible for the
dividends-received deduction generally allowed to United States
corporations in respect of dividends received from other United
States corporations. Distributions in excess of current and
accumulated earnings and profits, as determined for United
States federal income tax purposes, will be treated as a
non-taxable return of capital to the extent of your basis in the
shares and thereafter as capital gain.
We expect that following the Offering we will be 50% or more
owned, by vote or value, by United States persons and that
at least 10% of our earnings and profits will be attributable to
sources within the United States. Accordingly, we expect that
for foreign tax credit purposes, a portion of our dividends will
be treated as derived from sources within the United States.
With respect to any dividend paid for any taxable year, the
United States source ratio of our dividends will be calculated
as follows: the numerator of such ratio will be the portion of
our earnings and profits from sources within the United States
for such taxable year, and the denominator will be the total
amount of our earnings and profits for such taxable year.
However, we do not expect to provide U.S. Holders with such
ratio. The remaining portion of the dividends will be income
from sources outside the United States and will, depending on
your circumstances, be either “passive” or
“general” income for purposes of computing the foreign
tax credit allowable to you. In general, your ability to use
foreign tax credits may be limited and is dependent on your
particular circumstances. U.S. Holders should consult their own
tax advisors with respect to these matters.
Controlled Foreign Corporation Rules. A
foreign corporation will be treated as a “controlled
foreign corporation” (“CFC”) for
United States federal income tax purposes if, on any day
during the
173
taxable year of such foreign corporation, more than 50% of the
equity interests in such corporation, measured by reference to
the combined voting power or value of the equity of the
corporation, is owned directly or by application of the
attribution and constructive ownership rules of
Sections 958(a) and 958(b) of the Code by United States
Shareholders. For this purpose, a “United States
Shareholder” is any United States person that possesses
directly, or by application of the attribution and constructive
ownership rules of Sections 958(a) and 958(b) of the Code,
10% or more of the combined voting power of all classes of
equity in such corporation. If a foreign corporation is a CFC
for an uninterrupted period of 30 days or more during any
taxable year, each United States Shareholder of the corporation
who owns, directly or indirectly, shares in the corporation on
the last day of the taxable year in which it is a CFC will be
required to include in its gross income for United States
federal income tax purposes its pro rata share of the
CFC’s “subpart F income”, even if the subpart F
income is not distributed. Subpart F income generally includes
passive income but also includes certain related party sales and
manufacturing income.
Rhône Capital has informed us that it intends to take the
position that Arizona Chemical Ltd. and certain of its foreign
subsidiaries are CFCs for United States federal income tax
purposes. Accordingly, United States persons who might,
directly, indirectly or constructively, acquire 10% or more of
the common shares of the Issuer or any of its subsidiaries, and
therefore might be a United States Shareholder, should consider
the possible application of the CFC rules, and consult a tax
advisor with respect to such matter.
Non-U.S.
Holders. Dividends paid to you in respect of
shares will not be subject to United States federal income tax
unless the dividends are “effectively connected” with
your conduct of a trade or business within the United States,
and the dividends are attributable to a permanent establishment
that you maintain in the United States if that is required by an
applicable income tax treaty as a condition for subjecting you
to United States taxation on a net income basis. In such cases
you generally will be taxed in the same manner as a
U.S. Holder. If you are a corporate
non-U.S. holder,
“effectively connected” dividends may, under certain
circumstances, be subject to an additional “branch profits
tax” at a 30% rate or at a lower rate if you are eligible
for the benefits of an income tax treaty that provides for a
lower rate.
Taxation of
Capital Gains
U.S. Holders. Subject to the PFIC rules
discussed below, if you sell or otherwise dispose of your
shares, you will recognize capital gain or loss equal to the
difference between the amount that you realize and your tax
basis in your shares. Capital gain of a noncorporate
U.S. Holder is generally taxed at preferential rates where
the holder has a holding period greater than one year. The gain
or loss will generally be income or loss from sources within the
United States for foreign tax credit limitation purposes.
Non-U.S.
Holders. You will not be subject to United
States federal income tax on gain recognized on the sale or
other disposition of your shares unless:
|
|
|
| |
•
|
the gain is “effectively connected” with your conduct
of a trade or business in the United States, and the gain is
attributable to a permanent establishment that you maintain in
the United States if that is required by an applicable income
tax treaty as a condition for subjecting you to United States
taxation on a net income basis; or
|
| |
| |
•
|
you are an individual, you are present in the United States for
183 or more days in the taxable year of the sale and certain
other conditions exist.
|
If you are a corporate
non-U.S.
Holder, “effectively connected” gains that you
recognize may also, under certain circumstances, be subject to
an additional “branch profits tax” at a 30% rate or at
a lower rate if you are eligible for the benefits of an income
tax treaty that provides for a lower rate.
PFIC
Rules
In general, with respect to U.S. Holders, a foreign corporation
will be a PFIC if for any taxable year in which such
corporation’s shares are held by U.S. Holders, at least 75%
of the gross income for the taxable year is passive income or at
least 50% of the value of such corporation’s assets,
determined on the basis of a quarterly average, is attributable
to assets that produce or are held for the production of passive
income.
174
We believe that your shares should not be treated as stock of a
PFIC for United States federal income tax purposes, but this
conclusion is a factual determination that is made annually and
thus may be subject to change. If we were to be treated as a
PFIC, unless a U.S. Holder elects to be taxed annually on a
mark-to-market
basis with respect to the shares, gain realized on the sale or
other disposition of your shares would in general not be treated
as capital gain. Instead, if you are a U.S. Holder, you
would be treated as if you had realized such gain and certain
“excess distributions” ratably over your holding
period for the shares and would be taxed at the highest tax rate
in effect for each such year to which the gain or distribution
was allocated, together with an interest charge in respect of
the tax attributable to each such year. With certain exceptions,
your shares will be treated as stock in a PFIC if we were a PFIC
at any time during your holding period in your shares. Dividends
that you receive from us will not be eligible for the special
tax rates applicable to qualified dividend income if we are
treated as a PFIC with respect to you either in the taxable year
of the distribution or the preceding taxable year, but instead
will be taxable at rates applicable to ordinary income.
Medicare
Tax
For taxable years beginning after December 31, 2012, a
United States person that is an individual or estate, or a
trust that does not fall into a special class of trusts that is
exempt from such tax, is subject to a 3.8% tax on the lesser of
(1) the United States person’s “net
investment income” for the relevant taxable year and
(2) the excess of the United States person’s
modified gross income for the taxable year over a certain
threshold (which in the case of individuals will be between
$125,000 and $250,000, depending on the individual’s
circumstances). A holder’s net investment income will
generally include its gross dividend income and its net gains
from the disposition of shares, unless such dividends or net
gains are derived in the ordinary course of the conduct of a
trade or business (other than a trade or business that consists
of certain passive or trading activities). If you are a United
States person that is an individual, estate or trust, you
are urged to consult your tax advisors regarding the
applicability of the Medicare tax to your income and gains in
respect of your investment in the shares.
Information
with Respect to Foreign Financial Assets
Under recently enacted legislation, individuals that own
“specified foreign financial assets” with an aggregate
value in excess of $50,000 in taxable years beginning after
March 18, 2010 will generally be required to file an
information report with respect to such assets with their tax
returns. “Specified foreign financial assets” include
any financial accounts maintained by foreign financial
institutions, as well as any of the following, but only if they
are not held in accounts maintained by financial institutions:
(i) stocks and securities issued by non-U.S. persons,
(ii) financial instruments and contracts held for
investment that have non-U.S. issuers or counterparties and
(iii) interests in foreign entities. U.S. holders that are
individuals are urged to consult their tax advisors regarding
the application of this legislation to their ownership of the
common shares.
Backup
Withholding and Information Reporting
If you are a noncorporate U.S. Holder, information
reporting requirements, on Internal Revenue Service
Form 1099, generally will apply to:
|
|
|
| |
•
|
dividend payments or other taxable distributions made to you
within the United States; and
|
| |
| |
•
|
the payment of proceeds to you from the sale of shares effected
at a United States office of a broker.
|
Additionally, backup withholding (currently at a rate of 28%)
may apply to such payments if you are a noncorporate
U.S. Holder that:
|
|
|
| |
•
|
fails to provide an accurate taxpayer identification number;
|
| |
| |
•
|
is notified by the Internal Revenue Service that you have failed
to report all interest and dividends required to be shown on
your federal income tax returns; or
|
| |
| |
•
|
in certain circumstances, fails to comply with applicable
certification requirements.
|
175
Pursuant to recently enacted legislation, certain payments made
to corporate U.S. Holders after December 31, 2011 may also
be subject to information reporting and backup withholding.
If you are a
non-U.S. Holder,
you are generally exempt from backup withholding and information
reporting requirements with respect to:
|
|
|
| |
•
|
dividend payments made to you outside the United States by us or
another
non-United
States payor and
|
| |
| |
•
|
other dividend payments and the payment of the proceeds from the
sale of shares effected at a United States office of a broker,
as long as the income associated with such payments is otherwise
exempt from United States federal income tax; and:
|
|
|
|
| |
•
|
the payor or broker does not have actual knowledge or reason to
know that you are a United States person and you have furnished
the payor or broker:
|
|
|
|
| |
•
|
an Internal Revenue Service
Form W-8BEN
or an acceptable substitute form upon which you certify, under
penalties of perjury, that you are a
non-United
States person; or
|
| |
| |
•
|
other documentation upon which it may rely to treat the payments
as made to a
non-United
States person in accordance with U.S. Treasury
regulations; or
|
|
|
|
| |
•
|
you otherwise establish an exemption.
|
Payment of the proceeds from the sale of shares effected at a
foreign office of a broker generally will not be subject to
information reporting or backup withholding. However, a sale of
shares that is effected at a foreign office of a broker will be
subject to information reporting and backup withholding if:
|
|
|
| |
•
|
the proceeds are transferred to an account maintained by you in
the United States;
|
| |
| |
•
|
the payment of proceeds or the confirmation of the sale is
mailed to you at a United States address; or
|
| |
| |
•
|
the sale has some other specified connection with the United
States as provided in U.S. Treasury regulations;
|
unless the broker does not have actual knowledge or reason to
know that you are a United States person and the documentation
requirements described above are met or you otherwise establish
an exemption.
In addition, a sale of shares effected at a foreign office of a
broker will be subject to information reporting if the broker is:
|
|
|
| |
•
|
a United States person;
|
| |
| |
•
|
a controlled foreign corporation for United States tax purposes;
|
| |
| |
•
|
a foreign person 50% or more of whose gross income is
effectively connected with the conduct of a United States trade
or business for a specified three-year period; or
|
| |
| |
•
|
a foreign partnership, if at any time during its tax year:
|
|
|
|
| |
•
|
one or more of its partners are “U.S. persons”,
as defined in U.S. Treasury regulations, who in the
aggregate hold more than 50% of the income or capital interest
in the partnership; or
|
| |
| |
•
|
such foreign partnership is engaged in the conduct of a United
States trade or business;
|
unless the broker does not have actual knowledge or reason to
know that you are a United States person and the documentation
requirements described above are met or you otherwise establish
an exemption. Backup withholding will apply if the sale is
subject to information reporting and the broker has actual
knowledge that you are a United States person.
You generally may obtain a refund of any amounts withheld under
the backup withholding rules that exceed your income tax
liability by filing a refund claim with the United States
Internal Revenue Service.
176
UNDERWRITING
The Company, the selling shareholder and the underwriters named
below have entered into an underwriting agreement with respect
to the shares being offered. Subject to certain conditions, each
underwriter has severally agreed to purchase the number of
shares indicated in the following table. Goldman,
Sachs & Co.
and
are the representatives of the underwriters.
| |
|
|
|
|
|
|
|
Number of
|
|
|
Underwriters
|
|
Shares
|
|
|
|
|
Goldman, Sachs & Co.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
|
|
The underwriters are committed to take and pay for all of the
shares being offered, if any are taken, other than the shares
covered by the option described below unless and until this
option is exercised.
If the underwriters sell more shares than the total number set
forth in the table above, the underwriters have an option to buy
up to an
additional shares
from AZ Chem Investments Partners LP. They may exercise that
option for 30 days. If any shares are purchased pursuant to
this option, the underwriters will severally purchase shares in
approximately the same proportion as set forth in the table
above.
The following tables show the per share and total underwriting
discounts and commissions to be paid to the underwriters by the
Company and the selling shareholder. Such amounts are shown
assuming both no exercise and full exercise of the
underwriters’ option to
purchase
additional shares.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
Full
|
|
Paid by the Company
|
|
No Exercise
|
|
Exercise
|
|
|
|
Per Share
|
|
$
|
|
|
|
$
|
|
|
|
Total
|
|
$
|
|
|
|
$
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
Full
|
|
Paid by the Selling
Shareholder
|
|
No Exercise
|
|
Exercise
|
|
|
|
Per Share
|
|
$
|
|
|
|
$
|
|
|
|
Total
|
|
$
|
|
|
|
$
|
|
|
Shares sold by the underwriters to the public will initially be
offered at the initial public offering price set forth on the
cover of this prospectus. Any shares sold by the underwriters to
securities dealers may be sold at a discount of up to
$ per share from the initial
public offering price. If all the shares are not sold at the
initial public offering price, the representatives may change
the offering price and the other selling terms. The offering of
the shares by the underwriters is subject to receipt and
acceptance and subject to the underwriters’ right to reject
any order in whole or in part.
177
The Company and the selling shareholder have agreed with the
underwriters, subject to certain exceptions, not to dispose of
or hedge any of their common shares or securities convertible
into or exchangeable for common shares during the period from
the date of this prospectus continuing through the date
180 days after the date of this prospectus, except with the
prior written consent of Goldman, Sachs & Co. This
agreement does not apply to any existing employee benefit plans.
See “Shares Eligible for Future Sale” for a
discussion of certain transfer restrictions.
The 180-day
restricted period described in the preceding paragraph will be
automatically extended if: (1) during the last 17 days
of the
180-day
restricted period the Company issues an earnings release or
announces material news or a material event; or (2) prior
to the expiration of the
180-day
restricted period, the Company announces that it will release
earnings results during the
15-day
period following the last day of the
180-day
period, in which case the restrictions described in the
preceding paragraph will continue to apply until the expiration
of the
18-day
period beginning on the issuance of the earnings release or the
announcement of the material news or material event.
Prior to the offering, there has been no public market for the
shares. The initial public offering price has been negotiated
among the Company and the representatives. Among the factors to
be considered in determining the initial public offering price
of the shares, in addition to prevailing market conditions, will
be the Company’s historical performance, estimates of the
business potential and earnings prospects of the Company, an
assessment of the Company’s management and the
consideration of the above factors in relation to market
valuation of companies in related businesses.
An application has been made to list the common shares on the
New York Stock Exchange under the symbol “ARZ”. In
order to meet one of the requirements for listing the common
shares on the NYSE, the underwriters have undertaken to sell
lots of 100 or more shares to a minimum of 2,000 beneficial
holders.
In connection with the offering, the underwriters may purchase
and sell common shares in the open market. These transactions
may include short sales, stabilizing transactions and purchases
to cover positions created by short sales. Shorts sales involve
the sale by the underwriters of a greater number of shares than
they are required to purchase in the offering.
“Covered” short sales are sales made in an amount not
greater than the underwriters’ option to purchase
additional shares from the selling shareholder in the offering.
The underwriters may close out any covered short position by
either exercising their option to purchase additional shares or
purchasing shares in the open market. In determining the source
of shares to close out the covered short position, the
underwriters will consider, among other things, the price of
shares available for purchase in the open market as compared to
the price at which they may purchase additional shares pursuant
to the option granted to them. “Naked” short sales are
any sales in excess of such option. The underwriters must close
out any naked short position by purchasing shares in the open
market. A naked short position is more likely to be created if
the underwriters are concerned that there may be downward
pressure on the price of the common shares in the open market
after pricing that could adversely affect investors who purchase
in the offering. Stabilizing transactions consist of various
bids for or purchases of common shares made by the underwriters
in the open market prior to the completion of the offering.
The underwriters may also impose a penalty bid. This occurs when
a particular underwriter repays to the underwriters a portion of
the underwriting discount received by it because the
representatives have repurchased shares sold by or for the
account of such underwriter in stabilizing or short covering
transactions.
Purchases to cover a short position and stabilizing
transactions, as well as other purchases by the underwriters for
their own accounts, may have the effect of preventing or
retarding a decline in the market price of the Company’s
shares, and together with the imposition of the penalty bid, may
stabilize, maintain or otherwise affect the market price of the
common shares. As a result, the price of the common shares may
be higher than the price that otherwise might exist in the open
market. If these activities are commenced, they may be
discontinued at any time. These transactions may be effected on
NYSE, in the
over-the-counter
market or otherwise.
178
In relation to each Member State of the European Economic Area
which has implemented the Prospectus Directive (each, a Relevant
Member State), each underwriter has represented and agreed that
with effect from and including the date on which the Prospectus
Directive is implemented in that Relevant Member State (the
Relevant Implementation Date) it has not made and will not make
an offer of shares to the public in that Relevant Member State
prior to the publication of a prospectus in relation to the
shares which has been approved by the competent authority in
that Relevant Member State or, where appropriate, approved in
another Relevant Member State and notified to the competent
authority in that Relevant Member State, all in accordance with
the Prospectus Directive, except that it may, with effect from
and including the Relevant Implementation Date, make an offer of
shares to the public in that Relevant Member State at any time:
|
|
|
| |
•
|
to legal entities which are authorized or regulated to operate
in the financial markets or, if not so authorized or regulated,
whose corporate purpose is solely to invest in securities;
|
| |
| |
•
|
to any legal entity which has two or more of (1) an average
of at least 250 employees during the last financial year,
(2) a total balance sheet of more than €43,000,000 and
(3) an annual net turnover of more than €50,000,000,
as shown in its last annual or consolidated accounts;
|
| |
| |
•
|
to fewer than 100 natural or legal persons (other than qualified
investors as defined in the Prospectus Directive) subject to
obtaining the prior consent of the representatives for any such
offer; or
|
| |
| |
•
|
in any other circumstances which do not require the publication
by the Issuer of a prospectus pursuant to Article 3 of the
Prospectus Directive.
|
For the purposes of this provision, the expression an
“offer of shares to the public” in relation to any
shares in any Relevant Member State means the communication in
any form and by any means of sufficient information on the terms
of the offer and the shares to be offered so as to enable an
investor to decide to purchase or subscribe the shares, as the
same may be varied in that Relevant Member State by any measure
implementing the Prospectus Directive in that Relevant Member
State and the expression Prospectus Directive means Directive
2003/71/EC and includes any relevant implementing measure in
each Relevant Member State.
Each underwriter has represented and agreed that:
|
|
|
| |
•
|
it has only communicated or caused to be communicated and will
only communicate or cause to be communicated an invitation or
inducement to engage in investment activity (within the meaning
of Section 21 of the FSMA) received by it in connection
with the issue or sale of the shares in circumstances in which
Section 21(1) of the FSMA does not apply to the
issuer; and
|
| |
| |
•
|
it has complied and will comply with all applicable provisions
of the FSMA with respect to anything done by it in relation to
the shares in, from or otherwise involving the United Kingdom.
|
The shares may not be offered or sold by means of any document
other than (i) in circumstances which do not constitute an
offer to the public within the meaning of the Companies
Ordinance (Cap.32, Laws of Hong Kong), or (ii) to
“professional investors” within the meaning of the
Securities and Futures Ordinance (Cap.571, Laws of Hong Kong)
and any rules made thereunder or (iii) in other
circumstances which do not result in the document being a
“prospectus” within the meaning of the Companies
Ordinance (Cap.32, Laws of Hong Kong), and no advertisement,
invitation or document relating to the shares may be issued or
may be in the possession of any person for the purpose of issue
(in each case whether in Hong Kong or elsewhere), which is
directed at, or the contents of which are likely to be accessed
or read by, the public in Hong Kong (except if permitted to do
so under the laws of Hong Kong) other than with respect to
shares which are or are intended to be disposed of only to
persons outside Hong Kong or only to “professional
investors” within the meaning of the Securities and Futures
Ordinance (Cap.571, Laws of Hong Kong) and any rules made
thereunder.
This prospectus has not been registered as a prospectus with the
Monetary Authority of Singapore. Accordingly, this prospectus
and any other document or material in connection with the offer
or sale, or invitation for subscription or purchase, of the
shares may not be circulated or
179
distributed, nor may the shares be offered or sold, or be made
the subject of an invitation for subscription or purchase,
whether directly or indirectly, to persons in Singapore other
than (i) to an institutional investor under
Section 274 of the Securities and Futures Act,
Chapter 289 of Singapore (the “SFA”),
(ii) to a relevant person, or any person pursuant to
Section 275(1A), and in accordance with the conditions,
specified in Section 275 of the SFA or (iii) otherwise
pursuant to, and in accordance with the conditions of, any other
applicable provision of the SFA.
Where the shares are subscribed or purchased under
Section 275 by a relevant person which is: (a) a
corporation (which is not an accredited investor) the sole
business of which is to hold investments and the entire share
capital of which is owned by one or more individuals, each of
whom is an accredited investor or (b) a trust (where the
trustee is not an accredited investor) whose sole purpose is to
hold investments and each beneficiary is an accredited investor,
shares, debentures and units of shares and debentures of that
corporation or the beneficiaries’ rights and interest in
that trust shall not be transferable for 6 months after
that corporation or that trust has acquired the shares under
Section 275 except: (1) to an institutional investor
under Section 274 of the SFA or to a relevant person, or
any person pursuant to Section 275(1A), and in accordance
with the conditions, specified in Section 275 of the SFA,
(2) where no consideration is given for the transfer or
(3) by operation of law.
The securities have not been and will not be registered under
the Financial Instruments and Exchange Law of Japan (the
Financial Instruments and Exchange Law) and each underwriter has
agreed that it will not offer or sell any securities, directly
or indirectly, in Japan or to, or for the benefit of, any
resident of Japan (which term as used herein means any person
resident in Japan, including any corporation or other entity
organized under the laws of Japan), or to others for re-offering
or resale, directly or indirectly, in Japan or to a resident of
Japan, except pursuant to an exemption from the registration
requirements of, and otherwise in compliance with, the Financial
Instruments and Exchange Law and any other applicable laws,
regulations and ministerial guidelines of Japan.
Certain of the underwriters and their respective affiliates
have, from time to time, performed, and may in the future
perform, various financial advisory and investment banking
services for the Company, for which they received or will
receive customary fees and expenses. An affiliate of Goldman,
Sachs & Co. is a lender under both our First Lien
Credit Agreement and our Second Lien Credit Agreement, the
administrative agent and collateral agent under our First Lien
Credit Agreement and served as lead arranger, bookrunner and
syndication agent under both our First Lien Credit Agreement and
our Second Lien Credit Agreement. We intend to use our net
proceeds from the shares that we sell in this offering to repay
$ million of borrowings under
our First Lien Credit Agreement. As a result, affiliates of
Goldman, Sachs & Co. may receive a portion of the
proceeds of this offering through such repayment. The
underwriters do not expect sales to discretionary accounts to
exceed five percent of the total number of shares offered.
The underwriters and their respective affiliates are full
service financial institutions engaged in various activities,
which may include securities trading, commercial and investment
banking, financial advisory, investment management, principal
investment, hedging, financing and brokerage activities. Certain
of the underwriters and their respective affiliates have, from
time to time, performed, and may in the future perform, various
financial advisory and investment banking services for us, for
which they received or will receive customary fees and expenses.
In the ordinary course of their various business activities, the
underwriters and their respective affiliates may make or hold a
broad array of investments and actively trade debt and equity
securities (or related derivative securities) and financial
instruments (including bank loans) for their own account and for
the accounts of their customers and may at any time hold long
and short positions in such securities and instruments. Such
investment and securities activities may involve our securities
and instruments.
The Company estimates that its share of the total expenses of
the offering, excluding underwriting discounts and commissions,
will be approximately $ .
The Company and the selling shareholder have agreed to indemnify
the several underwriters against certain liabilities, including
liabilities under the Securities Act.
180
VALIDITY OF
COMMON SHARES
The validity of the common shares offered hereby will be passed
upon for us by Conyers Dill & Pearman Limited, Hamilton,
Bermuda. Certain legal matters relating to this offering will be
passed upon for us by Sullivan & Cromwell LLP, New
York, New York. The underwriters have been represented by
Cravath, Swaine & Moore LLP, New York, New York.
EXPERTS
The consolidated financial statements and the related financial
statement schedules of Arizona Chem Sweden Holdings AB and
subsidiaries included in this prospectus as of December 31,
2009 and 2008, and for the years then ended and the ten month
period ended December 31, 2007 have been audited by
Deloitte & Touche LLP, an independent registered
public accounting firm, as stated in their report appearing
herein. Such financial statements and financial statement
schedules are included in reliance upon the report of such firm
given upon their authority as experts in accounting and auditing.
The consolidated financial statements and the related financial
statement schedule of Arizona Chemical Division of International
Paper Company included in this prospectus for the two months
ended February 28, 2007 have been audited by
Deloitte & Touche LLP, an independent registered
public accounting firm, as stated in their report appearing
herein. Such financial statements and financial statement
schedule are included in reliance upon the report of such firm
given upon their authority as experts in accounting and auditing.
All the information set forth in “Industry” and
certain of the information set forth in “Business” has
been included in reliance upon Arthur D. Little Benelux
S.A./N.V.’s authority as an expert on such matters.
WHERE YOU CAN
FIND MORE INFORMATION
We have filed with the Securities and Exchange Commission a
registration statement on
Form S-1,
including exhibits, schedules and amendments filed with this
registration statement, under the Securities Act, with respect
to our common shares to be sold in the offering. This prospectus
does not contain all of the information set forth in the
registration statement and exhibits and schedules to the
registration statement. For further information with respect to
our company and our common shares to be sold in the offering,
reference is made to the registration statement, including the
exhibits and schedules to the registration statement. You may
read and copy this information, including the exhibits and
schedules to the registration statement, without charge at the
public reference room of the SEC, 100 F Street, N.E.,
Room 1580, Washington, DC 20549. You may obtain information
on the operation of the public reference room by calling the SEC
at
1-800-SEC-0330.
Copies of all or a portion of the registration statement can be
obtained from the public reference room of the SEC upon payment
of prescribed fees. Our filings with the SEC, including our
registration statement, are also available to you on the
Securities and Exchange Commission’s website
www.sec.gov.
Upon completion of this offering, we will be subject to the
information reporting requirement of the Exchange Act, and we
intend to file reports, proxy statements and other information
with the SEC.
181
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
| |
|
|
|
|
|
|
|
Page
|
|
|
|
Audited Consolidated Financial Statements
|
|
|
|
|
|
|
|
|
F-2
|
|
|
|
|
|
F-4
|
|
|
|
|
|
F-5
|
|
|
|
|
|
F-6
|
|
|
|
|
|
F-7
|
|
|
|
|
|
F-8
|
|
|
|
|
|
|
|
|
Unaudited Condensed Consolidated Financial Statements
|
|
|
|
|
|
|
|
|
F-45
|
|
|
|
|
|
F-46
|
|
|
|
|
|
F-47
|
|
|
|
|
|
F-48
|
|
F-1
REPORT OF
INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Shareholder of
Arizona Chem Sweden Holdings AB
Jacksonville, Florida
We have audited the accompanying consolidated balance sheets of
Arizona Chem Sweden Holdings AB and subsidiaries (the
“Company”) as of December 31, 2009 and 2008, and
the related consolidated statements of operations,
shareholder’s equity and comprehensive income (loss), and
cash flows for the years then ended and the ten month period
ended December 31, 2007. Our audits also included the
related financial statement schedules listed in the Index at
Item 16. These financial statements and financial statement
schedules are the responsibility of the Company’s
management. Our responsibility is to express an opinion on the
financial statements and financial statement schedules based on
our audits.
We conducted our audits in accordance with the standards of the
Public Company Accounting Oversight Board (United States). Those
standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are
free of material misstatement. The Company is not required to
have, nor were we engaged to perform, an audit of its internal
control over financial reporting. Our audits included
consideration of internal control over financial reporting as a
basis for designing audit procedures that are appropriate in the
circumstances, but not for the purpose of expressing an opinion
on the effectiveness of the Company’s internal control over
financial reporting. Accordingly, we express no such opinion. An
audit also includes examining, on a test basis, evidence
supporting the amounts and disclosures in the financial
statements, assessing the accounting principles used and
significant estimates made by management, as well as evaluating
the overall financial statement presentation. We believe that
our audits provide a reasonable basis for our opinion.
In our opinion, such consolidated financial statements present
fairly, in all material respects, the financial position of
Arizona Chem Sweden Holdings AB and subsidiaries as of
December 31, 2009 and 2008, and the results of their
operations and their cash flows for each of the two years in the
period ended December 31, 2009 and the ten month period
ended December 31, 2007, in conformity with accounting
principles generally accepted in the United States of America.
Also, in our opinion, such financial statement schedules, when
considered in relation to the basic consolidated financial
statements taken as a whole, present fairly in all material
respects the information set forth therein.
Certified Public Accountants
Jacksonville, Florida
March 9, 2010 (May 28, 2010 as to Note 20)
F-2
REPORT OF
INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors
Arizona Chemical Company
Jacksonville, Florida
We have audited the accompanying consolidated statements of
operations, shareholder’s equity and comprehensive income
(loss), and cash flows of the Arizona Chemical Division of
International Paper Company (the “Division”) for the
two months ended February 28, 2007. Our audit also included
the financial statement schedule listed in the Index at
Item 16. These financial statements and financial statement
schedule are the responsibility of the Division’s
management. Our responsibility is to express an opinion on the
financial statements and financial statement schedule based on
our audit.
We conducted our audit in accordance with the standards of the
Public Company Accounting Oversight Board (United States). Those
standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are
free of material misstatement. The Company is not required to
have, nor were we engaged to perform, an audit of its internal
control over financial reporting. Our audits included
consideration of internal control over financial reporting as a
basis for designing audit procedures that are appropriate in the
circumstances, but not for the purpose of expressing an opinion
on the effectiveness of the Company’s internal control over
financial reporting. Accordingly, we express no such opinion. An
audit also includes examining, on a test basis, evidence
supporting the amounts and disclosures in the financial
statements, assessing the accounting principles used and
significant estimates made by management, as well as evaluating
the overall financial statement presentation. We believe that
our audit provides a reasonable basis for our opinion.
In our opinion, such consolidated financial statements present
fairly, in all material respects, the results of operations of
the Division and its cash flows for the two months ended
February 28, 2007, in conformity with accounting principles
generally accepted in the United States of America. Also, in our
opinion, such financial statement schedule, when considered in
relation to the basic consolidated financial statements taken as
a whole, presents fairly in all material respects the
information set forth therein.
As discussed in Note 2 to the financial statements, the
accompanying financial statements have been prepared from the
separate records maintained for the Division by International
Paper Company and may not necessarily be indicative of the
conditions that would have existed or the results of operations
if the Division had been operated as an unaffiliated company.
Portions of certain expenses represent allocations made from
corporate office items applicable to International Paper Company
as a whole.
As discussed in Note 1 to the financial statements, on
February, 28, 2007, Rhône Capital III completed the
purchase of the Division.
Certified Public Accountants
Jacksonville, Florida
August 10, 2007 (March 9, 2010 as to the financial
statements schedule)
F-3
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
(In
thousands, except share and per share amounts)
| |
|
|
|
|
|
|
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Assets
|
|
|
|
|
|
|
|
|
|
Current assets:
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
47,023
|
|
|
$
|
34,048
|
|
|
Accounts receivable, less allowance for doubtful accounts of
$1,000 and $1,279, respectively
|
|
|
99,702
|
|
|
|
121,714
|
|
|
Inventory
|
|
|
89,124
|
|
|
|
135,075
|
|
|
Deferred income taxes
|
|
|
3,038
|
|
|
|
5,769
|
|
|
Prepaid expenses and other current assets
|
|
|
12,863
|
|
|
|
11,830
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current assets
|
|
|
251,750
|
|
|
|
308,436
|
|
|
Property, plant and equipment, net
|
|
|
224,945
|
|
|
|
223,234
|
|
|
Intangible assets, net
|
|
|
113,540
|
|
|
|
135,179
|
|
|
Investment in affiliate
|
|
|
11,583
|
|
|
|
11,192
|
|
|
Other assets
|
|
|
16,191
|
|
|
|
13,043
|
|
|
|
|
|
|
|
|
|
|
|
|
Total assets
|
|
$
|
618,009
|
|
|
$
|
691,084
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities and shareholder’s equity
|
|
|
|
|
|
|
|
|
|
Current liabilities:
|
|
|
|
|
|
|
|
|
|
Accounts payable
|
|
$
|
82,826
|
|
|
$
|
106,706
|
|
|
Accrued liabilities
|
|
|
42,888
|
|
|
|
40,132
|
|
|
Current portion of long-term debt
|
|
|
3,935
|
|
|
|
2,786
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current liabilities
|
|
|
129,649
|
|
|
|
149,624
|
|
|
Deferred income taxes
|
|
|
39,645
|
|
|
|
42,001
|
|
|
Long-term debt
|
|
|
330,823
|
|
|
|
380,619
|
|
|
Capital lease obligations
|
|
|
3,796
|
|
|
|
3,983
|
|
|
Other liabilities
|
|
|
16,489
|
|
|
|
19,348
|
|
|
|
|
|
|
|
|
|
|
|
|
Total liabilities
|
|
|
520,402
|
|
|
|
595,575
|
|
|
Commitments and contingencies (Note 15)
|
|
|
|
|
|
|
|
|
|
Shareholder’s equity:
|
|
|
|
|
|
|
|
|
|
Common shares, $14.25 par value — issued and
outstanding 1,000 shares
|
|
|
14
|
|
|
|
14
|
|
|
Paid-in capital
|
|
|
135,661
|
|
|
|
137,338
|
|
|
Accumulated deficit
|
|
|
(41,791
|
)
|
|
|
(53,889
|
)
|
|
Accumulated other comprehensive income
|
|
|
3,723
|
|
|
|
12,046
|
|
|
|
|
|
|
|
|
|
|
|
|
Total shareholder’s equity
|
|
|
97,607
|
|
|
|
95,509
|
|
|
|
|
|
|
|
|
|
|
|
|
Total liabilities and shareholder’s equity
|
|
$
|
618,009
|
|
|
$
|
691,084
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements
F-4
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
(In thousands,
except share and per share amounts)
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
|
Predecessor
|
|
|
|
|
|
|
|
|
|
|
March 1
|
|
|
|
January 1
|
|
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
|
through
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
February 28,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
2007
|
|
|
Net sales
|
|
$
|
767,465
|
|
|
$
|
1,001,988
|
|
|
$
|
723,797
|
|
|
|
$
|
132,070
|
|
|
Cost of goods sold
|
|
|
646,986
|
|
|
|
868,536
|
|
|
|
642,341
|
|
|
|
|
113,074
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gross profit
|
|
|
120,479
|
|
|
|
133,452
|
|
|
|
81,456
|
|
|
|
|
18,996
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating expenses (income):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Selling, general and administrative
|
|
|
78,200
|
|
|
|
91,936
|
|
|
|
86,684
|
|
|
|
|
12,899
|
|
|
Unrealized foreign currency exchange (gains) losses
|
|
|
(9,347
|
)
|
|
|
20,304
|
|
|
|
—
|
|
|
|
|
—
|
|
|
Restructuring and impairment
|
|
|
26,395
|
|
|
|
15,513
|
|
|
|
114
|
|
|
|
|
—
|
|
|
Other operating income
|
|
|
(5,537
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total operating expenses (income)
|
|
|
89,711
|
|
|
|
127,753
|
|
|
|
86,798
|
|
|
|
|
12,899
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating income (loss)
|
|
|
30,768
|
|
|
|
5,699
|
|
|
|
(5,342
|
)
|
|
|
|
6,097
|
|
|
Interest (expense) income, net
|
|
|
(16,546
|
)
|
|
|
(29,523
|
)
|
|
|
(28,775
|
)
|
|
|
|
118
|
|
|
Loss on interest rate swaps, net
|
|
|
(2,541
|
)
|
|
|
(9,311
|
)
|
|
|
(2,275
|
)
|
|
|
|
—
|
|
|
Other income
|
|
|
3,635
|
|
|
|
1,879
|
|
|
|
—
|
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income (loss) before income tax expense (benefit) and equity in
earnings of affiliates, net of taxes
|
|
|
15,316
|
|
|
|
(31,256
|
)
|
|
|
(36,392
|
)
|
|
|
|
6,215
|
|
|
Income tax expense (benefit)
|
|
|
3,831
|
|
|
|
(4,277
|
)
|
|
|
(8,913
|
)
|
|
|
|
2,614
|
|
|
Equity in earnings of affiliates net of taxes of $356, $218,
$114, and $51, respectively
|
|
|
613
|
|
|
|
380
|
|
|
|
189
|
|
|
|
|
84
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income (loss)
|
|
$
|
12,098
|
|
|
$
|
(26,599
|
)
|
|
$
|
(27,290
|
)
|
|
|
$
|
3,685
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Earnings per share:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic and diluted
|
|
$
|
12,098
|
|
|
$
|
(26,599
|
)
|
|
$
|
(27,290
|
)
|
|
|
|
|
|
|
Weighted average common shares outstanding
|
|
|
1,000
|
|
|
|
1,000
|
|
|
|
1,000
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements
F-5
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
COMPREHENSIVE
INCOME (LOSS)
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Paid-in
|
|
|
Accumulated
|
|
|
Comprehensive
|
|
|
|
|
|
|
|
Shares
|
|
|
Amount
|
|
|
Capital
|
|
|
Deficit
|
|
|
Income
|
|
|
Total
|
|
|
|
|
(In thousands, except per share amounts)
|
|
|
|
|
PREDECESSOR
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance of Divisional Control Account at January 1,
2007
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
362,854
|
|
|
Net income
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
3,685
|
|
|
Foreign currency translation adjustment
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1,364
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Comprehensive income (net of benefits distributed through group
tax sharing agreement with International Paper)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2,321
|
|
|
Funding provided to International Paper
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(2,032
|
)
|
|
Change in cumulative translation adjustment due to International
Paper
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
41
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance of Divisional Control Account at February 28,
2007
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
363,184
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
SUCCESSOR
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at March 1, 2007
|
|
|
1,000
|
|
|
$
|
14
|
|
|
$
|
129,986
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
130,000
|
|
|
Net loss
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(27,290
|
)
|
|
|
—
|
|
|
|
(27,290
|
)
|
|
Foreign currency translation adjustment
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(297
|
)
|
|
|
(297
|
)
|
|
Net gain from pension plans, net of tax
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,632
|
|
|
|
1,632
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Comprehensive loss
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(25,955
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2007
|
|
|
1,000
|
|
|
|
14
|
|
|
|
129,986
|
|
|
|
(27,290
|
)
|
|
|
1,335
|
|
|
|
104,045
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Contribution of debt by Rhône Capital
|
|
|
—
|
|
|
|
—
|
|
|
|
7,352
|
|
|
|
—
|
|
|
|
—
|
|
|
|
7,352
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(26,599
|
)
|
|
|
—
|
|
|
|
(26,599
|
)
|
|
Foreign currency translation adjustment
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
12,755
|
|
|
|
12,755
|
|
|
Net loss from pension plans, net of tax
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(2,044
|
)
|
|
|
(2,044
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Comprehensive loss
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(15,888
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2008
|
|
|
1,000
|
|
|
|
14
|
|
|
|
137,338
|
|
|
|
(53,889
|
)
|
|
|
12,046
|
|
|
|
95,509
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Distribution to parent
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,677
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,677
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
12,098
|
|
|
|
—
|
|
|
|
12,098
|
|
|
Foreign currency translation adjustment
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(5,902
|
)
|
|
|
(5,902
|
)
|
|
Net loss from pension plans, net of tax
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(2,421
|
)
|
|
|
(2,421
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Comprehensive income
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
3,775
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2009
|
|
|
1,000
|
|
|
$
|
14
|
|
|
$
|
135,661
|
|
|
$
|
(41,791
|
)
|
|
$
|
3,723
|
|
|
$
|
97,607
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements
F-6
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
|
Predecessor
|
|
|
|
|
|
|
|
|
|
|
March 1
|
|
|
|
January 1
|
|
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
|
through
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
February 28,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
2007
|
|
|
|
|
(In thousands)
|
|
|
Operating activities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income (loss)
|
|
$
|
12,098
|
|
|
$
|
(26,599
|
)
|
|
$
|
(27,290
|
)
|
|
|
$
|
3,685
|
|
|
Adjustments to reconcile net income (loss) to net cash provided
by (used in) operating activities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Depreciation and amortization
|
|
|
40,615
|
|
|
|
34,210
|
|
|
|
28,813
|
|
|
|
|
4,422
|
|
|
Change in fair value of interest rate swaps
|
|
|
(5,715
|
)
|
|
|
5,183
|
|
|
|
3,417
|
|
|
|
|
—
|
|
|
(Gain) loss on unrealized foreign exchange
|
|
|
(9,347
|
)
|
|
|
20,304
|
|
|
|
—
|
|
|
|
|
—
|
|
|
Impairment loss
|
|
|
18,109
|
|
|
|
7,003
|
|
|
|
—
|
|
|
|
|
—
|
|
|
Gain on extinguishment of debt
|
|
|
—
|
|
|
|
(2,150
|
)
|
|
|
—
|
|
|
|
|
—
|
|
|
Loss (gain) on disposal of property, plant and equipment
|
|
|
63
|
|
|
|
(75
|
)
|
|
|
22
|
|
|
|
|
—
|
|
|
Gain on acquisition
|
|
|
(2,142
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
|
—
|
|
|
Amortization of debt issuance costs
|
|
|
2,520
|
|
|
|
2,896
|
|
|
|
2,237
|
|
|
|
|
—
|
|
|
Deferred income tax expense (benefit)
|
|
|
2,920
|
|
|
|
(4,191
|
)
|
|
|
(10,710
|
)
|
|
|
|
(119
|
)
|
|
Provision for bad debts
|
|
|
144
|
|
|
|
567
|
|
|
|
240
|
|
|
|
|
—
|
|
|
Equity in undistributed earnings of investment in affiliate
|
|
|
(395
|
)
|
|
|
(598
|
)
|
|
|
(303
|
)
|
|
|
|
—
|
|
|
Other, net
|
|
|
115
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
(853
|
)
|
|
Voluntary non-US pension plan contribution
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
(9,152
|
)
|
|
Change in assets and liabilities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts receivable
|
|
|
25,858
|
|
|
|
(9,608
|
)
|
|
|
10,383
|
|
|
|
|
14,115
|
|
|
Inventory
|
|
|
57,414
|
|
|
|
(6,235
|
)
|
|
|
12,353
|
|
|
|
|
(10,974
|
)
|
|
Prepaid expenses and other current assets
|
|
|
1,078
|
|
|
|
(1,861
|
)
|
|
|
3,227
|
|
|
|
|
779
|
|
|
Other assets
|
|
|
(5,426
|
)
|
|
|
2,787
|
|
|
|
4,539
|
|
|
|
|
(390
|
)
|
|
Accounts payable
|
|
|
(16,793
|
)
|
|
|
(3,917
|
)
|
|
|
20,573
|
|
|
|
|
(14,117
|
)
|
|
Accrued liabilities
|
|
|
(1,623
|
)
|
|
|
7,951
|
|
|
|
875
|
|
|
|
|
—
|
|
|
Other liabilities
|
|
|
(2,168
|
)
|
|
|
(4,826
|
)
|
|
|
(3,354
|
)
|
|
|
|
516
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash provided by (used in) operating activities
|
|
|
117,325
|
|
|
|
20,841
|
|
|
|
45,022
|
|
|
|
|
(12,088
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Investing activities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Acquisition of business — net of cash acquired of
$1,101 and $9,259, in 2009 and the ten months ended
December 31, 2007
|
|
|
(8,772
|
)
|
|
|
—
|
|
|
|
(477,441
|
)
|
|
|
|
—
|
|
|
Proceeds from disposals of property, plant and equipment
|
|
|
875
|
|
|
|
212
|
|
|
|
—
|
|
|
|
|
—
|
|
|
Dividends received from equity investment
|
|
|
—
|
|
|
|
356
|
|
|
|
—
|
|
|
|
|
—
|
|
|
Additions to property, plant and equipment
|
|
|
(22,993
|
)
|
|
|
(34,719
|
)
|
|
|
(18,248
|
)
|
|
|
|
(4,598
|
)
|
|
Capitalized software costs
|
|
|
(13,404
|
)
|
|
|
(142
|
)
|
|
|
(1,642
|
)
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash used in investing activities
|
|
|
(44,294
|
)
|
|
|
(34,293
|
)
|
|
|
(497,331
|
)
|
|
|
|
(4,598
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Financing activities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Issuance of common stock
|
|
|
—
|
|
|
|
—
|
|
|
|
130,000
|
|
|
|
|
—
|
|
|
Proceeds from long-term and short-term obligations
|
|
|
17,731
|
|
|
|
69,811
|
|
|
|
384,399
|
|
|
|
|
—
|
|
|
Repayments of long-term and short-term obligations
|
|
|
(75,971
|
)
|
|
|
(56,767
|
)
|
|
|
(11,347
|
)
|
|
|
|
—
|
|
|
Debt issuance costs
|
|
|
(490
|
)
|
|
|
—
|
|
|
|
(14,300
|
)
|
|
|
|
—
|
|
|
Repayment of capital lease obligation
|
|
|
(276
|
)
|
|
|
(276
|
)
|
|
|
(205
|
)
|
|
|
|
(78
|
)
|
|
Settlement of MIV loans
|
|
|
(3,088
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
|
—
|
|
|
Distribution to parent
|
|
|
(1,677
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
|
—
|
|
|
Collections from notes from International Paper
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
12,406
|
|
|
Funding provided to International Paper
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
(2,032
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash (used in) provided by financing activities
|
|
|
(63,771
|
)
|
|
|
12,768
|
|
|
|
488,547
|
|
|
|
|
10,296
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Effect of foreign exchange rate changes on cash and cash
equivalents
|
|
|
3,715
|
|
|
|
(3,354
|
)
|
|
|
1,848
|
|
|
|
|
(65
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Increase (decrease) in cash and cash equivalents
|
|
|
12,975
|
|
|
|
(4,038
|
)
|
|
|
38,086
|
|
|
|
|
(6,455
|
)
|
|
Cash and cash equivalents at beginning of period
|
|
|
34,048
|
|
|
|
38,086
|
|
|
|
—
|
|
|
|
|
17,506
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents at end of period
|
|
$
|
47,023
|
|
|
$
|
34,048
|
|
|
$
|
38,086
|
|
|
|
$
|
11,051
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Supplemental cash flows information:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash paid during the period for:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest (net of amounts capitalized)
|
|
$
|
22,188
|
|
|
$
|
29,126
|
|
|
$
|
25,094
|
|
|
|
$
|
693
|
|
|
Income taxes
|
|
$
|
2,181
|
|
|
$
|
2,355
|
|
|
$
|
2,480
|
|
|
|
$
|
195
|
|
|
Noncash investing and financing activities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Contribution by Rhône capital/debt retirement
|
|
$
|
—
|
|
|
$
|
7,352
|
|
|
$
|
—
|
|
|
|
$
|
—
|
|
|
Purchases of property, plant and equipment in accounts payable
|
|
$
|
5,683
|
|
|
$
|
407
|
|
|
$
|
905
|
|
|
|
$
|
—
|
|
See Notes to Consolidated Financial Statements
F-7
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2009 AND 2008,
THE TEN MONTH PERIOD
ENDED DECEMBER 31, 2007, AND THE TWO MONTH PERIOD ENDED
FEBRUARY 28, 2007
In these notes to the Consolidated Financial Statements, unless
the context requires otherwise, references to “Arizona
Chemical”, the “Company”, “we”,
“our”, or “us” refer to Arizona Chem Sweden
Holdings AB, a Swedish company, and its consolidated
subsidiaries whose financial statements are included herein.
“Rhône Capital” refers to Rhône Capital
L.L.C. and its affiliated entities, including Rhône
Capital III L.P., the general partner of certain associated
funds with investments in Arizona Chemical, and
“International Paper” refers to International Paper
Company. The accompanying consolidated financial statements are
prepared in accordance with accounting principles generally
accepted in the United States of America (“U.S. GAAP”).
We are the world’s leading supplier of pine-based chemicals
as measured by sales. We refine and further upgrade two primary
feedstocks, crude tall oil, or CTO, and crude sulfate
turpentine, or CST, both of which are wood pulping co-products,
into specialty chemicals.
Change of Ownership — On
February 28, 2007, Rhône Capital acquired us from
International Paper pursuant to a Stock Purchase Agreement (the
“Stock Purchase Agreement”) for approximately
$486.7 million plus direct acquisition costs, creating
Arizona Chem Sweden Holdings AB and subsidiaries (the
“Acquisition”). The Acquisition was funded with cash
of $130 million and substantially all of the proceeds from
the issuance of debt totaling $375 million. The Acquisition
resulted in the revaluation of assets and liabilities
establishing a new basis of accounting. The Acquisition was
recorded by allocating the purchase price, including liabilities
assumed, to the assets acquired, based on their estimated fair
values at the acquisition date. The fair values determined at
the acquisition date were greater than the purchase price,
therefore the excess fair value was allocated on a pro rata
basis to all of the acquired long-lived assets.
The valuation of assets and liabilities has been determined and
the purchase price was allocated as follows (in millions of
U.S. dollars):
| |
|
|
|
|
|
Accounts receivable
|
|
$
|
127.1
|
|
|
Inventory
|
|
|
149.6
|
|
|
Other current assets
|
|
|
6.5
|
|
|
Plant, property and equipment
|
|
|
219.9
|
|
|
Intangible assets(1)
|
|
|
153.7
|
|
|
Investment in affiliate
|
|
|
11.3
|
|
|
Other assets
|
|
|
3.3
|
|
|
|
|
|
|
|
|
Total assets acquired
|
|
|
671.4
|
|
|
Accounts payable
|
|
|
87.3
|
|
|
Accrued liabilities
|
|
|
33.3
|
|
|
Deferred income taxes
|
|
|
58.2
|
|
|
Long-term debt
|
|
|
9.4
|
|
|
Other liabilities
|
|
|
5.7
|
|
|
|
|
|
|
|
|
Total liabilities
|
|
|
193.9
|
|
|
Less cash acquired
|
|
|
(9.2
|
)
|
|
|
|
|
|
|
|
Total liabilities assumed
|
|
|
184.7
|
|
|
|
|
|
|
|
|
Net assets acquired
|
|
$
|
486.7
|
|
|
|
|
|
|
|
|
|
|
|
(1) |
|
See Note 8 Intangible Assets |
F-8
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
During the year ended December 31, 2008, certain estimates
used in the purchase price allocation were updated and as a
result, intangible assets and related deferred tax liabilities
were reduced by $3.1 million.
During the year ended December 31, 2009, certain
adjustments to the purchase price were finalized based on the
result of the Settlement and Mutual Release Agreement entered
into on November 17, 2009 between the Company and AZ Chem
Luxembourg Holdings S.à r.l. (“Lux Holdco”) with
International Paper, as discussed in Note 14, “Related
Party Transactions”. As a result, we recorded a gain on
this settlement of $1.3 million and recorded a distribution
to our parent of $1.7 million. This also resulted in us
reducing our intangible assets by $18.4 million, accounts
payable by $11.4 million, deferred income taxes by
$6.7 million, and increasing other liabilities by
$0.1 million.
The Consolidated Financial Statements include the accounts of
the Company and its subsidiaries. All intercompany transactions
have been eliminated. When the Company does not have a
controlling interest in an entity, but exerts significant
influence over the entity, the Company applies the equity method
of accounting.
Predecessor Period — The accompanying
financial statements prior to the Acquisition include the
financial statements of Arizona Chemical Division (the
“Division”), which operated as a division of
International Paper for the two month period ended
February 28, 2007 (the “Predecessor”).
Certain services were provided to the Division by International
Paper, including corporate management, legal, accounting and
tax, treasury, payroll and benefits administration, incentive
compensation administration, risk management, information
technology, and centralized transaction processing. Expenses for
such corporate services totaled $2.2 million for the two
months ended February 28, 2007. Additionally, International
Paper European service centers charged the European entities of
the Division for similar corporate services, which totaled
$0.7 million for the two months ended February 28,
2007. These expenses are included in selling, general and
administrative expenses in the accompanying Predecessor
financial statements. These costs allocated from International
Paper to the Division were based on various allocation methods
including direct consumption, percent of capital employed, and
number of employees. We believe that these allocations include
the expenses incurred on our behalf. These Predecessor financial
statements may not necessarily be indicative of the conditions
that would have existed or the results of operations if the
Division had been operated as an unaffiliated company.
Successor Period — The accompanying
Consolidated Financial Statements include the accounts of the
Company subsequent to the Acquisition.
The Consolidated Financial Statements of the Successor as of and
for the years ended December 31, 2009 and 2008 and for the
period from March 1, 2007 through December 31, 2007
reflect the new basis of accounting resulting from the
Acquisition.
|
|
|
3.
|
Summary of
Significant Accounting Policies
|
Use of Estimates — The preparation of
these Consolidated Financial Statements, in accordance with
U.S. GAAP, requires management to make estimates and
assumptions that affect the amounts reported in the Consolidated
Financial Statements and Notes to Consolidated Financial
Statements. Although these estimates are based on
management’s best available knowledge at the time, actual
results could differ from these estimates. Significant items
subject to such estimates and assumptions include the useful
lives of property, plant and equipment; allowances for doubtful
accounts; the valuation of derivatives, deferred tax asset
valuation allowance and share-based compensation; and
F-9
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
liabilities for pension plans, environmental liabilities, income
tax uncertainties and other contingencies. The current economic
environment has increased the degree of uncertainty inherent in
those estimates and assumptions.
Revenue Recognition — The Company
recognizes revenue when the earnings process is complete. The
Company’s revenues are from the sale of a wide range of
products to a diversified base of customers around the world.
Revenue from sales of products, including amounts billed to
customers for shipping and handling costs, is recognized when
ownership and all risks of loss have been transferred to the
buyer, the price is fixed and determinable, and collectability
is reasonably assured. Revenue in North America is usually
recorded at the time of shipment while revenue from our European
operations is usually recorded when the product is delivered to
the customer’s site, as that is when title and risk of loss
transfers. Accruals are made for sales returns and other
allowances based on the Company’s experience. The Company
accounts for cash sales incentives as a reduction in net sales.
Shipping and Handling Charges and
Expenses — Shipping and handling charges
billed to customers are included in net sales. Shipping and
handling charges included in net sales for the years ended
December 31, 2009 and 2008, the ten month period ended
December 31, 2007 and the two month period ended
February 28, 2007 were $6.8 million,
$10.6 million, $9.8 million and $1.8 million,
respectively. Shipping and handling expenses, such as freight to
customer destinations, are included in cost of goods sold in the
accompanying consolidated statements of operations.
Cost of Goods Sold — Cost of goods sold
includes the cost of inventory (materials and conversion costs)
sold to customers. It also includes shipping and handling costs,
certain warehousing costs, inbound freight charges, receiving
costs, packaging costs, quality assurance costs, internal
transfer costs, other costs of our distribution network, safety,
health and environmental administration, and certain
depreciation and amortization expenses.
Selling, General and Administrative
Expenses — Selling expenses include the cost
of our sales force and marketing staff and their related
expenses. General and administrative expenses primarily
represent the cost of support functions, including information
technology, finance, human resources and legal, as well as
expenses for support facilities, certain depreciation and
amortization, research and development, executive management,
and management incentive plans.
Cash and Cash Equivalents — We invest
our excess cash in investment instruments whose value is not
subject to market fluctuations, such as bank deposits or
certificates of deposit. We consider all investments having an
original maturity of three months or less to be cash
equivalents. We recorded interest income of $0.4 million,
$0.9 million, $1.2 million, and $0.4 million for
the years ended December 31, 2009 and 2008, the ten month
period ended December 31, 2007 and the two month period
ended February 28, 2007.
Fair Value of Financial Instruments —
The Company applies fair value accounting for all financial
assets and liabilities and non-financial assets and liabilities
that are recognized or disclosed at fair value in the financial
statements on a recurring basis. The Company defines fair value
as the price that would be received from selling an asset or
paid to transfer a liability in an orderly transaction between
market participants at the measurement date. When determining
the fair value measurements for assets and liabilities, which
are required to be recorded at fair value, the Company considers
the principal or most advantageous market in which the Company
would transact and the market-based risk measurements or
assumptions that market participants would use in pricing the
asset or liability, such as inherent risk, transfer restrictions
and credit risk.
The carrying amount of cash and cash equivalents, accounts
receivables, accounts payable and certain accrued expenses
approximates fair value due to the short term nature of these
instruments. The fair value of the Company’s floating
interest rate debt is measured using quoted market prices.
F-10
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
The fair value of the interest rate swap agreements are
determined by obtaining dealer quotes for instruments with
similar characteristics. Considerable judgment is required in
developing these estimates.
We record derivative financial instruments at fair value and
include these instruments in the balance sheet as assets or
liabilities. The accounting for changes in the fair value of a
derivative instrument depends on the intended use of the
derivative and the resulting designation. We have not designated
any derivative financial instruments as hedges and therefore
recognize changes in fair value in the statements of operations.
We use natural gas in our production processes and, as a result,
we are exposed to commodity price risk. To partially offset the
impact of price changes, we enter into forward purchase
contracts that require our contract counterparties to deliver
natural gas to us at a fixed price on the contract maturity
dates. As a result of the requirements of counterparties to
physically deliver the gas to us at the settlement date, these
contracts do not qualify as derivatives for financial reporting
purposes.
The term of our contracts range from one month to approximately
twelve months. We make monthly accruals based on estimated
volumes of gas consumed during the month and a blended average
rate that takes into account the contract price and current
market prices for volumes that are not fixed as the gas is
consumed.
Concentrations of Credit Risk — Credit
risk represents the loss that would be recognized if
counterparties failed to perform as contracted. Financial
instruments with credit risk, which consist primarily of trade
accounts receivable, expose the Company to concentrations of
credit risk. We continually monitor the creditworthiness of
customers to whom credit terms are granted in the normal course
of business.
We establish the allowance for doubtful accounts based upon
factors surrounding the credit risks of specific customers,
historical trends, and other information. The allowance for
doubtful accounts was approximately $1.0 million and
$1.3 million at December 31, 2009 and 2008,
respectively. Bad debt expense, net of recoveries, was
$0.1 million, $0.6 million, $0.2 million and $1
thousand for the years ended December 31, 2009 and 2008,
the ten month period ended December 31, 2007 and the two
month period ended February 28, 2007, respectively.
Inventory — Inventory values include all
costs directly associated with manufacturing products (i.e.,
materials, labor, and manufacturing overhead) and are presented
at the lower of cost or market. Cost of raw materials, work in
progress, and finished goods for the Company are determined
using the first in, first out (FIFO) method. Replacement parts
and other supplies are classified as other assets in the
accompanying consolidated balance sheets.
We have the ability to process a wide variety of CTO feedstocks.
We have long term supply contracts with International Paper
pursuant to which they agreed to sell to us, and we agreed to
purchase from them, all of the CTO and CST produced at their
existing U.S. paper mills at current market prices. We also have
the option to purchase all of the CTO and CST produced at
International Paper’s future paper mills worldwide. These
contracts provided us with approximately 27% of our global CTO
requirements and 41% of our global CST requirements in 2009, and
CTO and CST accounted for approximately 68% and 3% of our annual
raw material costs, respectively, in 2009. We also maintain
long-standing relationships with other major suppliers in the
United States and Europe.
We receive monthly wood processing data from the paper mills and
compute the expected quantity of CTO and CST. Based on this
information we schedule transport and freight of the CTO and CST
in primarily Arizona Chemical leased railcars and to a lesser
degree tank wagons. We issue
F-11
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
purchase orders and record inventory based upon the weight of
CTO and CST which is triggered upon shipment from the paper
mills.
Actual quantities shipped approximate quantities received and
weighed by our manufacturing facilities and therefore we do not
experience material valuation issues during transit. The price
of CTO supplied to us by International Paper is determined by
reference to the price of energy, while the purchase price for
CST supplied to us by International Paper is fixed, subject to
adjustment every three years based on changes in market price.
Inventory Purchase and Sale
Arrangements — We enter into agreements with
third parties to purchase CTO, and to sell back to the third
parties the Company’s by-product, pitch. These swap
transactions provide additional long term supplies of our raw
material CTO and revenue from the sale of pitch fuel. These
agreements provide for the sales and purchases to be consummated
at market rates. We account for these inventory purchases and
sale arrangements on a gross basis. We recognized revenue from
the agreements of approximately $36.3 million,
$60.4 million, $32.8 million and $7.0 million for
the years ended December 31, 2009 and 2008, the ten month
period ended December 31, 2007 and the two month period
ended February 28, 2007, respectively.
Property, Plant and Equipment — Plant,
property and equipment are stated at cost, net of accumulated
depreciation. Interest is capitalized on projects meeting
certain criteria and is included in the cost of the assets.
Capitalized interest is depreciated over the same useful lives
as the related assets. Expenditures for major repairs and
improvements are capitalized, whereas normal repairs and
maintenance are expensed as incurred. Depreciation and
amortization are computed using the straight line method over
the assets’ estimated useful lives as follows:
| |
|
|
|
|
|
Years
|
|
|
|
Buildings
|
|
20–40
|
|
Machinery and equipment
|
|
1–15
|
|
Software
|
|
3–6
|
|
Leasehold improvements
|
|
Over the shorter of the term of the lease or the useful life of
the improvements
|
Our policy is to capitalize interest cost incurred on debt
during the construction of major projects that meet any one of
the following criteria: the primary assets being constructed
have a useful life of 40 years or more; or the estimated
construction or development period exceeds two years; or the
estimated construction or development period exceeds six months
and the budgeted cost exceeds $1 million. Interest
capitalized amounted to $0.9 million in 2009. No interest
was capitalized for the years ended December 31, 2008, the
ten month period ended December 31, 2007 and the
two month period ended February 28, 2007.
Asset Retirement Obligation — The
Company has asset retirement obligations associated with the
demolition and decommissioning of manufacturing facility assets
with indeterminate settlement dates. The fair value of these
obligations cannot be reasonably estimated and accordingly a
liability is not recognized. When a date or range of settlement
dates can reasonably be estimated for the retirement of the
assets, the Company will estimate the cost of performing the
retirement activities and record a liability for the fair value
of that cost using established present value techniques.
F-12
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
Intangible Assets — Intangible assets
are stated at cost, net of accumulated amortization.
Amortization is computed using the straight-line method over the
assets’ estimated useful lives as follows:
| |
|
|
|
|
|
Years
|
|
|
|
Trade names
|
|
Indefinite
|
|
Customer relationships
|
|
6–15
|
|
Supply agreements
|
|
7–20
|
|
Core/developed technology
|
|
10–15
|
|
Favorable leaseholds
|
|
39
|
|
Internally developed software
|
|
3
|
Impairment of Long-Lived Assets — We
evaluate long-lived assets, such as property, plant and
equipment and intangible assets subject to amortization, for
impairment upon the occurrence of events or changes in
circumstances that indicate that the carrying value of the
assets may not be recoverable. Factors considered important that
could result in an impairment review include, but are not
limited to, significant underperformance relative to historical
or planned operating results, significant changes in the manner
of use of the assets, or significant changes in our business
strategies. An impairment loss is recognized when the estimated
undiscounted cash flows expected to result from the use of the
asset, plus net proceeds expected from disposition of the asset
(if any) are less than the carrying value of the asset. When an
impairment loss is recognized, the carrying amount of the asset
is reduced to its estimated fair value based on quoted market
prices or other valuation techniques.
Trade names which are intangible assets determined to have
indefinite lives and goodwill related to our equity method
interest in Arboris, LLC are reviewed for impairment
annually or more frequently, if events or changes in
circumstances indicate that the intangible asset might be
impaired. Fair values for trade names and goodwill are
determined based on discounted cash flows.
Income Taxes — We account for income
taxes using the asset and liability method. Under this method,
we recognize deferred tax assets and liabilities attributable to
differences between the carrying amounts of existing assets and
liabilities and their respective tax bases. We measure deferred
tax assets and liabilities on a legal entity basis using enacted
tax rates expected to apply to taxable income in the years in
which those temporary differences are expected to be recovered
or settled. We recognize the effect on deferred tax assets and
liabilities of a change in tax rates in income in the period
that includes the enactment date.
Deferred taxes are not provided for temporary differences
representing earnings of the non-Swedish entities that are
intended to be permanently reinvested.
In assessing the realizability of deferred tax assets,
management considers whether it is more likely than not that
some or all of the deferred tax assets will not be realized. The
ultimate realization of deferred tax assets is dependent upon
the generation of future taxable income during the periods in
which temporary differences become deductible as well as the
reversal of certain deferred tax liabilities. Management also
considers projected future taxable income, tax planning
strategies, and other factors in making this assessment and
establishing an appropriate valuation allowance. Based upon
these factors, management believes it is more likely than not
that the Company will realize the benefits of these deductible
differences, net of the existing valuation allowances. The
amount of the deferred tax assets considered realizable,
however, could be reduced if expected taxable income or other
factors change in future years.
F-13
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
The Company provides liabilities for uncertain tax positions for
Federal, state, local and international exposures relating to
periods subject to audit. The development of liabilities for
uncertain tax positions for these exposures requires judgments
about tax issues, potential outcomes and timing and is a
subjective critical estimate. The Company assesses its tax
positions and records tax benefits based upon management’s
evaluation of the facts, circumstances and information available
at the reporting dates. For those tax positions where it is
more-likely-than-not that a tax benefit will be sustained, the
Company has recorded the largest amount of tax benefit with a
greater than 50% likelihood of being realized upon settlement
with a tax authority that has full knowledge of all relevant
information. For those tax positions where it is not
more-likely-than-not that a tax benefit will be sustained, no
tax benefit has been recognized in the financial statements.
Environmental Costs and Obligations — We
accrue costs associated with environmental obligations, such as
remediation or closure costs, when such costs are probable and
reasonably estimable. We adjust such accruals as further
information develops or circumstances change. We discount costs
of future expenditures for environmental obligations to their
present value when the expected cash flows are reliably
determinable. Legal costs incurred in connection with loss
contingencies are expensed as incurred. Indemnification of
certain environmental remediation costs of $3.3 million and
$2.6 million in 2009 and 2008, respectively, from third
parties, which are probable of realization, are separately
recorded as assets, and are not offset against the related
environmental liability as of December 31, 2009 and 2008.
Translation of Financial Statements and Foreign Currency
Transactions — Foreign currency denominated
assets and liabilities are translated into U.S. dollars
using the exchange rates in effect at the consolidated balance
sheet date. Statements of operations and cash flows are
translated using the average exchange rates throughout the
period. The effect of exchange rate fluctuations on translation
of assets and liabilities is included as a component of
shareholder’s equity in accumulated other comprehensive
income. Gains and losses resulting from foreign currency
transactions are included in earnings in the period in which
they occur. During 2009 we recorded an unrealized translation
gain of $9.3 million, and during 2008 we recorded a loss of
$20.3 million. There were no significant unrealized
transaction gains or losses in 2007. These gains and losses
primarily related to our Euro denominated debt under our First
Lien Credit Agreement, which is recorded within our Swedish
subsidiary that has a Kronor functional currency. As the related
transactions did not result in cash receipts or cash payments
during the period and are included in the determination of net
loss, the amounts are presented as an adjustment to reconcile
net income (loss) to net cash provided by operating activities
in the statement of cash flows.
Equity Investment — We hold a 10%
investment in Arboris, LLC, which was formed in 2002 and is
accounted for under the equity method of accounting, as we have
the ability to exercise significant influence through the
one-third representation on the board. The venture is located on
our Savannah production facility and is engaged in the
production and sale of sterols for pharmaceutical and functional
foods applications. The Company’s investment is stated at
cost, less accumulated amortization of identified intangibles,
plus the Company’s share of post March 1, 2007
earnings, less dividends received. The Company’s book basis
exceeds its pro rata share of Arboris, LLC equity by
approximately $6.3 million and $6.6 million at
December 31, 2009 and 2008, respectively. The excess book
basis is a result of intangibles of $3.6 million and
goodwill of $3.6 million recorded as part of the
acquisition.
Pension, Early Retirement and Long-term Service
Awards — We sponsor noncontributory defined
benefit pension plans. We also sponsor an Early Retirement plan
and a Long-term Service Award plan. The actuarial determination
of the projected benefit obligations and related benefit expense
requires that certain assumptions be made regarding such
variables as expected return on plan assets, discount
F-14
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
rates, rates of future compensation increases, estimated future
employee turnover rates and retirement dates, distribution
election rates, mortality rates, retiree utilization rates for
health care services and health care cost trend rates. The
discount rate assumption is based on current investment yields
on high quality fixed income investments. The salary growth
assumptions include long-term actual experience and expectations
for future growth. The differences between actual experience and
the assumptions are accumulated and amortized over the estimated
future working life of the plan participants. See Note 13
“Retirement Plans” for further details.
As of December 31, 2009, approximately 54% of our
691 employees in the United States were unionized and are
covered by collective bargaining agreements. In Europe nearly
all of our employees are represented by local workers’
councils and/or unions.
Research and Development — Research and
development costs are expensed as incurred. Research and
development costs amounted to $3.5 million,
$4.0 million, $3.5 million and $0.9 million for
the years ended December 31, 2009 and 2008, the ten month
period ended December 31, 2007 and the two month period
ended February 28, 2007, respectively.
Stock-based Awards — As part of the
Acquisition, AZ Chem Investments LLC, the general partner of
AZ Chem Investments Partners LP, our indirect parent,
formed AZ Chem MIV I Ltd, which we refer to as
“MIV I”, and AZ Chem MIV II LP, which we
refer to as “MIV II”. We refer to MIV I and
MIV II together as the “MIVs”. The MIVs were
created as vehicles to enable certain members of our management
and board of directors to participate in the ownership of AZ
Chem Investments Partners LP through equity purchases and grants
of awards under our management incentive plan. AZ Chem
Investments Partners LP controls the Company through Lux Holdco,
which holds all of the issued and outstanding shares of the
Company. Since inception, AZ Chem Investments LLC granted
partnership equity interests in the MIVs to certain
participants. These grants vest over five years, and the
resulting compensation expense is recorded by us based on the
fair value of the Company at the time of each grant. The
compensation cost of grant awards made under our management
incentive plan is charged to selling, general and administrative
expense over the applicable vesting periods.
|
|
|
4.
|
Recent Accounting
Pronouncements
|
In June 2009, the FASB issued authoritative guidance to
eliminate the exception to consolidate a qualifying
special-purpose entity, change the approach to determining the
primary beneficiary of a variable interest entity and require
companies to more frequently reassess whether they must
consolidate variable interest entities. Under the new guidance,
the primary beneficiary of a variable interest entity is
identified qualitatively as the enterprise that has both
(a) the power to direct the activities of a variable
interest entity that most significantly impact the entity’s
economic performance, and (b) the obligation to absorb
losses of the entity that could potentially be significant to
the variable interest entity or the right to receive benefits
from the entity that could potentially be significant to the
variable interest entity. This standard is effective for
financial statements for interim or annual reporting periods
beginning after January 1, 2010. We do not expect this
guidance to have a material impact on our consolidated financial
statements.
In May 2009, the FASB issued new guidance related to the
disclosure of subsequent events. This standard is intended to
establish general standards of accounting for and disclosure of
events that occur after the balance sheet date but before
financial statements are issued or are available to be issued.
Specifically, this standard sets forth the period after the
balance sheet date during which management of a reporting entity
should evaluate events or transactions that may occur for
potential recognition or disclosure in the financial statements,
the circumstances under which an entity should recognize events
or transactions occurring after the balance sheet date in its
financial statements, and the disclosures that an entity should
make about events or transactions that occurred after the
F-15
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
balance sheet date. This guidance is effective for fiscal years
and interim periods ended after June 15, 2009. The
additional disclosures have been included in Note 20
“Subsequent Events”.
In April 2009, the FASB issued authoritative guidance to require
that assets acquired and liabilities assumed in a business
combination that arise from contingencies be recognized at fair
value if fair value can be reasonably determined. If the fair
value of such assets or liabilities cannot be reasonably
determined, then they would generally be recognized in
accordance with certain other pre-existing accounting standards.
This guidance also amends the subsequent accounting for assets
and liabilities arising from contingencies in a business
combination and certain other disclosure requirements. This
guidance was effective for assets or liabilities arising from
contingencies in business combinations for which the acquisition
date was on or after the beginning of our annual reporting
period beginning January 1, 2009. The adoption of this
standard did not have a material impact on our consolidated
financial statements.
In November 2008, the FASB issued authoritative guidance
regarding the accounting for defensive intangible assets.
Defensive intangible assets are assets acquired in a business
combination that the acquirer (a) does not intend to use or
(b) intends to use in a way other than the assets’
highest and best use as determined by an evaluation of market
participant assumptions. While defensive intangible assets are
not being actively used, they are likely contributing to an
increase in the value of other assets owned by the acquiring
entity. This guidance will require defensive intangible assets
to be accounted for as separate units of accounting at the time
of acquisition and the useful life of such assets would be based
on the period over which the assets will directly or indirectly
affect the entity’s cash flows. This guidance was effective
for intangible assets acquired in a business combination for
which the acquisition date was on or after the beginning of our
annual reporting period beginning January 1, 2009. The
adoption of this standard did not have a material impact on our
consolidated financial statements.
In November 2008, the FASB issued authoritative guidance to
address questions about equity-method accounting. The primary
issues include how the initial carrying value of an equity
method investment should be determined, how to account for any
subsequent purchases and sales of additional ownership interests
and whether the investor must separately assess its underlying
share of the investee’s indefinite-lived intangible assets
for impairment. This guidance was effective for the beginning of
our annual reporting period beginning January 1, 2009. The
adoption of this standard did not have a material impact on our
consolidated financial statements.
|
|
|
5.
|
Fair Value of
Financial Instruments
|
The estimated fair values of the Company’s financial
instruments as of December 31, 2009 and 2008 were as
follows (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
2008
|
|
|
|
Carrying
|
|
Fair
|
|
Carrying
|
|
Fair
|
|
Asset (Liability)
|
|
Amount
|
|
Value
|
|
Amount
|
|
Value
|
|
|
|
Cash and cash equivalents
|
|
$
|
47,023
|
|
|
$
|
47,023
|
|
|
$
|
34,048
|
|
|
$
|
34,048
|
|
|
Long-term debt
|
|
$
|
(334,758
|
)
|
|
$
|
(319,868
|
)
|
|
$
|
(383,405
|
)
|
|
$
|
(383,405
|
)
|
|
Interest rate swaps
|
|
$
|
(2,879
|
)
|
|
$
|
(2,879
|
)
|
|
$
|
(8,588
|
)
|
|
$
|
(8,588
|
)
|
|
Interest rate cap
|
|
$
|
1,028
|
|
|
$
|
1,028
|
|
|
|
—
|
|
|
|
—
|
|
The Company enters into interest rate swap agreements to reduce
its cash flow exposure to market risk from changes in interest
rates. As discussed in Note 11, the Company has a
$218.5 million six year first lien term loan and a
$115.5 million seven year second lien term loan outstanding
with variable interest rates based on the London InterBank
Offered Rate (LIBOR) for the U.S. dollar portion of the
loan and Euribor for the Euro portion of the loan.
F-16
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
In March 2007, the Company entered into four interest rate swap
agreements. One of these interest rate swap agreements matured
in February 2008, and another matured in February 2009. One of
the remaining agreements has a notional value of
$165 million as of December 31, 2009 and matures on
February 28, 2010. The other remaining agreement has a
notional value of €40 million and matures on
February 28, 2010. As of December 31, 2008 the total
notional value of the three interest rate swap agreements
outstanding were $195 million, €40 million and
€9 million, respectively. These agreements were put in
place to mitigate the interest rate risk associated with the
$375 million credit facilities bearing variable interest
rates. The notional amount does not represent a measure of
exposure to the Company. On the U.S. dollar agreement, the
Company will pay the counterparty interest at a fixed rate of
4.825% on the declining notional balance and the counterparty
will pay the Company interest at a variable rate equal to three
month LIBOR. On the Euro agreements, the Company will pay the
counterparty interest at a fixed rate of 3.998% for the
€40 million agreement, and the counterparty will pay
the Company interest at a variable rate equal to three month
Euribor.
During the second quarter of 2009, the Company purchased a
3-month USD
LIBOR interest rate cap for $1.1 million. The cap is
effective starting February 26, 2010 and matures
February 28, 2012. The strike price of the cap is 3% on a
notional amount of $175 million. If the
3-month
USD LIBOR exceeds 3% during the term of the cap, we will
receive a cash payment for the difference between the current
rate and 3% applied to the notional amount of $175 million.
As of December 31, 2009, the fair value of the cap was
$1.0 million.
The Company also entered into an interest rate swap starting
February 26, 2010 and maturing February 28, 2012. The
swap has a notional balance of €53 million and a
3-month
Euribor rate fixed at 2.125%. As of December 31, 2009, the
fair value of the swap was a liability of $(0.6) million.
The following summarizes the balance sheet location of our
derivative instruments (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Asset Derivatives
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Balance Sheet
|
|
|
Fair
|
|
|
Balance Sheet
|
|
|
Fair
|
|
|
|
|
Location
|
|
|
Value
|
|
|
Location
|
|
|
Value
|
|
|
|
|
Interest rate cap
|
|
|
Other assets
|
|
|
$
|
1,028
|
|
|
|
—
|
|
|
$
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
|
|
|
$
|
1,028
|
|
|
|
|
|
|
$
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Liability Derivatives
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Balance Sheet
|
|
|
Fair
|
|
|
Balance Sheet
|
|
|
Fair
|
|
|
|
|
Location
|
|
|
Value
|
|
|
Location
|
|
|
Value
|
|
|
|
|
Interest rate swaps
|
|
|
Accrued liabilities
|
|
|
$
|
2,303
|
|
|
|
Accrued liabilities
|
|
|
$
|
7,417
|
|
|
Interest rate swaps
|
|
|
Other liabilities
|
|
|
|
576
|
|
|
|
Other liabilities
|
|
|
|
1,171
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
|
|
|
$
|
2,879
|
|
|
|
|
|
|
$
|
8,588
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The Company did not possess any derivative instruments for the
two month period ended February 28, 2007. The following
summarizes the consolidated statements of operations location of
F-17
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
our derivative instruments for the years ended December 31,
2009 and 2008 and the ten month period ended December 31,
2007 (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Amount of Gain (Loss) Recognized in Income
|
|
Derivatives Not
|
|
Location of Gain
|
|
Year ended
|
|
|
Year ended
|
|
|
March 1 through
|
|
Designated as Hedging
|
|
(Loss) Recognized in
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
Instruments
|
|
Income
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
|
Interest rate swaps
|
|
Loss on interest rate swaps, net
|
|
$
|
5,738
|
|
|
$
|
(5,242
|
)
|
|
$
|
(3,438
|
)
|
|
Interest rate cap
|
|
Loss on interest rate swaps, net
|
|
|
(22
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
|
$
|
5,716
|
|
|
$
|
(5,242
|
)
|
|
$
|
(3,438
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net settlements of $(8.3) million, $(4.1) million and
$1.1 million for the years ended December 31, 2009 and
2008 and the ten month period ended December 31, 2007,
respectively on interest rate swap agreements are also included
in the loss on interest rate swaps.
The methods and assumptions used to estimate the fair value of
each class of financial instruments are set forth below:
Cash and Cash Equivalents — The carrying
amounts approximate fair value because of the relatively short
time between the origination of the instrument and its expected
realization. Foreign currency denominated cash and cash
equivalents are translated into U.S. dollars using exchange
rates in effect at the consolidated balance sheet date.
Long-Term Debt — The fair value of
long-term debt is estimated based on borrowing rates currently
available to the Company for loans with similar terms and
maturities and discounted back to the present value. The Company
obtains fair value measurements on its long-term debt
obligations form third party providers. Significant factors
evaluated include changes in margin on its various loans and its
ability to make future debt payments.
Derivative Financial Instruments — The
fair value of interest rate swaps (used for purposes other than
trading) represents the amount the Company would receive or pay
to terminate swap agreements at the reporting date, taking into
consideration the difference between the contract rate of
interest and rates currently quoted for agreements of similar
terms and maturities. The fair value measurement is estimated
based on a discounted cash flow model using a dealer quoted
interest rate. The fair value measurement of the derivative
instruments in a liability position also considers the current
credit-worthiness of the Company and its counterparties. The
Company is exposed to credit related losses in the event of
nonperformance by counterparties to these financial instruments.
However, counterparties to these agreements are major financial
institutions and the risk of loss due to nonperformance is
considered by management to be minimal.
To assess the inputs used to develop those measurements, a
hierarchy for ranking the quality and reliability of the
information used to determine fair value has been established
that applies to all assets and liabilities that are being
measured and reported on a fair value basis and requires their
classification and disclosure in one of the following three
categories:
Level 1 — Quoted market prices in
active markets for identical assets or liabilities.
Level 2 — Observable market based
inputs or unobservable inputs that are corroborated by market
data.
Level 3 — Unobservable inputs that
are not corroborated by market data.
F-18
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
The following summarizes the valuation of our financial
instruments reported at fair value by the above pricing levels
as of the valuation date listed (in thousands of
U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Derivative
|
|
|
|
|
|
|
|
|
|
Financial
|
|
Fair Value Measurements Using
|
|
|
|
Instruments
|
|
Level 1
|
|
Level 2
|
|
Level 3
|
|
|
|
As of December 31, 2009:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest rate swap
|
|
$
|
(2,879
|
)
|
|
$
|
—
|
|
|
$
|
(2,879
|
)
|
|
$
|
—
|
|
|
Interest rate cap
|
|
$
|
1,028
|
|
|
$
|
—
|
|
|
$
|
1,028
|
|
|
$
|
—
|
|
|
As of December 31, 2008:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest rate swap
|
|
$
|
(8,588
|
)
|
|
$
|
—
|
|
|
$
|
(8,588
|
)
|
|
$
|
—
|
|
Inventory by major category as of December 31, 2009 and
2008, include the following (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Raw materials
|
|
$
|
35,352
|
|
|
$
|
49,980
|
|
|
Finished goods
|
|
|
53,772
|
|
|
|
85,095
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
89,124
|
|
|
$
|
135,075
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
7.
|
Property, Plant
and Equipment, Net
|
Property, plant and equipment, net as of December 31, 2009
and 2008, consist of the following (in thousands of
U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Land
|
|
$
|
14,392
|
|
|
$
|
14,695
|
|
|
Buildings
|
|
|
42,188
|
|
|
|
34,546
|
|
|
Machinery and equipment
|
|
|
203,769
|
|
|
|
191,381
|
|
|
Construction in progress
|
|
|
7,693
|
|
|
|
17,058
|
|
|
Software
|
|
|
24,877
|
|
|
|
9,089
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
292,919
|
|
|
|
266,769
|
|
|
Less accumulated depreciation
|
|
|
(67,974
|
)
|
|
|
(43,535
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Property, plant and equipment, net
|
|
$
|
224,945
|
|
|
$
|
223,234
|
|
|
|
|
|
|
|
|
|
|
|
Property, plant and equipment includes a building occupied under
a capital lease for our Almere offices for $5.0 million and
$5.3 million, net of $1.9 million and
$1.4 million of accumulated amortization, at
December 31, 2009 and 2008, respectively. Amortization
expense related to the capital lease is included in depreciation
expense. The payments remaining under the lease are
$0.3 million for 2010, 2011, and 2012, $0.4 million
for 2013 and 2014, and $2.4 million thereafter. In 2014,
the Company has a purchase option to buy the building for
approximately $2.5 million.
The Company recognized depreciation expense of
$29.8 million, $23.3 million, $19.7 million and
$4.1 million for the years ended December 31, 2009 and
2008, the ten month period ended December 31, 2007 and the
two month period ended February 28, 2007, respectively,
which is reflected in cost of goods sold and selling, general
and administrative expenses in the accompanying consolidated
statements of operations.
F-19
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
|
|
|
8.
|
Intangible
Assets, Net
|
Intangible assets as of December 31, 2009 and 2008, consist
of the following (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Gross
|
|
|
|
|
|
Net
|
|
|
Gross
|
|
|
|
|
|
Net
|
|
|
|
|
Carrying
|
|
|
Accumulated
|
|
|
Carrying
|
|
|
Carrying
|
|
|
Accumulated
|
|
|
Carrying
|
|
|
|
|
Amount
|
|
|
Amortization
|
|
|
Amount
|
|
|
Amount
|
|
|
Amortization
|
|
|
Amount
|
|
|
|
|
Trade names
|
|
$
|
24,910
|
|
|
$
|
—
|
|
|
$
|
24,910
|
|
|
$
|
27,418
|
|
|
$
|
—
|
|
|
$
|
27,418
|
|
|
Customer relationships
|
|
|
44,614
|
|
|
|
(5,602
|
)
|
|
|
39,012
|
|
|
|
43,762
|
|
|
|
(3,366
|
)
|
|
|
40,396
|
|
|
Supply agreements
|
|
|
41,379
|
|
|
|
(12,796
|
)
|
|
|
28,583
|
|
|
|
51,864
|
|
|
|
(8,415
|
)
|
|
|
43,449
|
|
|
Core/developed technology
|
|
|
25,388
|
|
|
|
(5,127
|
)
|
|
|
20,261
|
|
|
|
26,547
|
|
|
|
(3,394
|
)
|
|
|
23,153
|
|
|
Favorable leaseholds
|
|
|
825
|
|
|
|
(59
|
)
|
|
|
766
|
|
|
|
737
|
|
|
|
(37
|
)
|
|
|
700
|
|
|
Internally developed software
|
|
|
170
|
|
|
|
(162
|
)
|
|
|
8
|
|
|
|
171
|
|
|
|
(108
|
)
|
|
|
63
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total intangible assets
|
|
$
|
137,286
|
|
|
$
|
(23,746
|
)
|
|
$
|
113,540
|
|
|
$
|
150,499
|
|
|
$
|
(15,320
|
)
|
|
$
|
135,179
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
We recognized amortization expense for the intangible assets
included above, intangible assets associated with our investment
in Arboris, LLC, and software included in property, plant and
equipment of $10.8 million, $10.9 million,
$9.1 million and $0.3 million for the years ended
December 31, 2009 and 2008, the ten month period ended
December 31, 2007 and the two month period ended
February 28, 2007, respectively.
Amortization is computed using the straight-line method over the
assets’ estimated useful lives. As of December 31,
2009 and 2008, all of the Company’s intangible assets that
had a carrying value were being amortized except for trade
names, which have been determined to have indefinite lives. The
Company annually reviews these assets to determine whether
events and circumstances continue to support the indefinite
useful life designation.
We perform an annual impairment test on October 31. During
our annual impairment test no impairment indicators were noted.
We also reviewed the amortization estimates, methods and the
amortization periods as required by U.S. GAAP for our intangible
assets and as a result of this review, the current economic
environment and the underlying cash flows we reduced the useful
life of our customer relationships from 25 years to
15 years effective November 1, 2009. The change in
estimate was accounted for prospectively.
The estimated aggregate amortization from intangible assets for
each of the next five years is as follows (in thousands of
U.S. dollars):
| |
|
|
|
|
|
|
|
Amortization
|
|
Years Ending December
31
|
|
Expense
|
|
|
|
2010
|
|
$
|
8,466
|
|
|
2011
|
|
$
|
8,457
|
|
|
2012
|
|
$
|
8,457
|
|
|
2013
|
|
$
|
8,457
|
|
|
2014
|
|
$
|
6,672
|
|
F-20
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
|
|
|
9.
|
Restructuring and
Impairment
|
Since the Acquisition, we initiated a comprehensive cost
reduction program that drives us to continuously reduce our
fixed costs, improve our processes and the way we run our
business, and better serve our customers. A key part of our
program has been the optimization of our manufacturing
footprint. This has resulted in the Company recording the
following restructuring and impairment charges in the
consolidated statement of operations.
In the fourth quarter of 2009, we streamlined and moved certain
administrative functions from our Abieta subsidiary to the
Netherlands, eliminating eight positions. We incurred
$1.2 million for
one-time
termination benefits related to these employees.
In the third quarter of 2009, we realigned our rosin capacity
from Valdosta, Georgia to our Savannah, Georgia manufacturing
facility, eliminating 28 positions. We incurred
$0.9 million for
one-time
termination benefits related to these employees.
In the second quarter of 2009, we announced that we would
permanently cease operations and close the Port St. Joe, Florida
(PSJ) manufacturing facility in a cost reduction initiative.
Production was transferred to our larger manufacturing
facilities: Panama City, Florida and Savannah, Georgia. The
announcement led to the termination of 77 employees at the
PSJ manufacturing facility. Of the 77 employees,
approximately 10 were required to work past the minimum
retention period to receive their termination benefits. We
incurred $1.8 million for
one-time
termination benefits related to these employees. The
announcement was also a triggering event for impairment testing
purposes. The test resulted in a $16.7 million charge
related to the impairment of
long-lived
assets. We also recognized $4.0 million of other qualified
costs related to the decommissioning and demolition of the PSJ
manufacturing facility.
On August 26, 2008, we announced that we would restructure
certain product lines at the Dover, Ohio manufacturing facility,
eliminating 24 positions. The affected employees were notified
and provided with a description of their termination benefits.
In 2009 we also recognized $1.4 million in impairment
charges related to the
write-off of
certain fixed assets which were considered to be idle and had no
future use.
On July 15, 2008, we approved a plan to restructure our
dimer business and relocate its polyamide production. The
restructuring affected the Company’s two U.K. manufacturing
facilities by closing Bedlington and reducing operations at
Chester le Street, eliminating 74 positions. The affected
employees were notified and provided with a description of their
termination benefits. The termination benefits are not
contingent on the individuals working past the minimum retention
period. In 2009 we recognized $0.4 million of other
qualified costs related to pension consulting services incurred
in connection with pension schemes for our terminated U.K.
employees.
In January 2008, we restructured a product line at the
Pensacola, Florida manufacturing facility, eliminating 15
positions. The termination benefits are not contingent on the
individuals working past the minimum retention period.
On December 15, 2007, we announced that during 2008 we
would terminate 23 employees at the Niort, France
manufacturing facility in a cost-reduction initiative.
Management developed a detailed termination plan for the
impacted employees prior to December 31, 2007. The affected
employees were notified and provided with a description of their
termination benefits. The termination benefits are not
contingent on the individuals working past the minimum retention
period subject to required approval by the appropriate works
council. Approval was not received as of December 31, 2007,
and as a result, a termination benefit was not recognized for
the period ended December 31, 2007. In March 2008,
appropriate approval was received.
F-21
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
As of December 31, 2009, we have accrued for substantially
all costs associated with the above restructuring activities.
However, due to our comprehensive cost reduction program,
including the optimization of our manufacturing footprint, we
expect to incur additional restructuring and impairment costs of
approximately $2.3 million during 2010 as we implement our
single business entity structure in Europe.
The following table summarizes the activity and remaining
liabilities associated with the restructuring charges as of and
for the years ended December 31, 2009 and 2008, and the ten
month period ended December 31, 2007 (in thousands of
U.S. dollars). There was no restructuring activity in the
two month period ended February 28, 2007. The following
liabilities are included in accrued liabilities.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Termination
|
|
|
|
|
|
|
|
|
|
|
Benefits to Employees
|
|
|
Other Qualified Costs
|
|
|
|
|
|
|
|
North America
|
|
|
Europe
|
|
|
North America
|
|
|
Europe
|
|
|
Total
|
|
|
|
|
Balance — February 28, 2007
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
Costs accrued
|
|
|
103
|
|
|
|
—
|
|
|
|
11
|
|
|
|
—
|
|
|
|
114
|
|
|
Payments
|
|
|
(103
|
)
|
|
|
—
|
|
|
|
(11
|
)
|
|
|
—
|
|
|
|
(114
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance — December 31, 2007
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Costs accrued
|
|
|
992
|
|
|
|
5,001
|
|
|
|
177
|
|
|
|
2,340
|
|
|
|
8,510
|
|
|
Payments
|
|
|
(954
|
)
|
|
|
(4,120
|
)
|
|
|
(177
|
)
|
|
|
(1,949
|
)
|
|
|
(7,200
|
)
|
|
Currency exchange
|
|
|
—
|
|
|
|
(676
|
)
|
|
|
—
|
|
|
|
(201
|
)
|
|
|
(877
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance — December 31, 2008
|
|
|
38
|
|
|
|
205
|
|
|
|
—
|
|
|
|
190
|
|
|
|
433
|
|
|
Costs accrued
|
|
|
2,650
|
|
|
|
1,250
|
|
|
|
4,048
|
|
|
|
339
|
|
|
|
8,287
|
|
|
Payments
|
|
|
(2,326
|
)
|
|
|
(611
|
)
|
|
|
(3,250
|
)
|
|
|
(538
|
)
|
|
|
(6,725
|
)
|
|
Currency exchange
|
|
|
—
|
|
|
|
(12
|
)
|
|
|
—
|
|
|
|
27
|
|
|
|
15
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance — December 31, 2009
|
|
$
|
362
|
|
|
$
|
832
|
|
|
$
|
798
|
|
|
$
|
18
|
|
|
$
|
2,010
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
In addition, the Company recorded impairment of certain land,
buildings and machinery associated with such production for the
years ended December 31, 2009 and 2008. The Company did not
have restructuring related impairment in the ten month period
ended December 31, 2007 or the two month period ended
February 28, 2007. The amount of impairment, consists of
the following (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Land
|
|
$
|
1,211
|
|
|
$
|
—
|
|
|
Machinery and equipment
|
|
|
15,568
|
|
|
|
3,431
|
|
|
Buildings
|
|
|
576
|
|
|
|
2,252
|
|
|
Construction in progress
|
|
|
754
|
|
|
|
—
|
|
|
CPI project
|
|
|
—
|
|
|
|
1,098
|
|
|
Software
|
|
|
—
|
|
|
|
222
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
18,109
|
|
|
$
|
7,003
|
|
|
|
|
|
|
|
|
|
|
|
F-22
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
Accrued liabilities as of December 31, 2009 and 2008,
consist of the following (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Payroll and related accruals
|
|
$
|
21,029
|
|
|
$
|
21,049
|
|
|
Accrued taxes — other than income taxes
|
|
|
3,173
|
|
|
|
2,592
|
|
|
Accrued rebates
|
|
|
979
|
|
|
|
2,081
|
|
|
Accrued environmental remediation
|
|
|
4,062
|
|
|
|
3,120
|
|
|
Short-term swap liability
|
|
|
2,303
|
|
|
|
1,171
|
|
|
Other
|
|
|
11,342
|
|
|
|
10,119
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
42,888
|
|
|
$
|
40,132
|
|
|
|
|
|
|
|
|
|
|
|
Debt and credit agreements at December 31, 2009 and 2008,
consist of the following (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
First lien term loan
|
|
$
|
218,543
|
|
|
$
|
238,603
|
|
|
Second lien term loan
|
|
|
115,498
|
|
|
|
115,498
|
|
|
Revolving credit facility
|
|
|
—
|
|
|
|
29,304
|
|
|
Bank loan
|
|
|
717
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
334,758
|
|
|
|
383,405
|
|
|
Less current portion
|
|
|
(3,935
|
)
|
|
|
(2,786
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Long-term debt
|
|
$
|
330,823
|
|
|
$
|
380,619
|
|
|
|
|
|
|
|
|
|
|
|
The first lien term loan is divided into two loans, one
denominated in U.S. dollars and one denominated in Euros.
The outstanding principal balance as of December 31, 2009
was $126.5 million and $92.1 million, respectively.
Interest is payable monthly or quarterly at LIBOR/Euribor or
prime rate, plus a 2.00% margin for U.S. dollar and 2.25% for
Euro denominated debt. The interest rate was 2.24% and 2.82%,
respectively, at December 31, 2009 and 2.46% and 5.21%,
respectively, at December 31, 2008. A principal payment is
payable quarterly in the amount of
1/4
of 1% of the outstanding term loan balance. Our first lien
credit agreement requires us to make principal payments equal to
50% of our excess cash flow as defined in our credit agreement;
provided that in any year in which we have excess cash flow but
our leverage ratio is less than 3.50:1.00, we are only required
to make a payment equal to 25% of our excess cash flow. For the
year ended December 31, 2009, the amount payable based on
the excess cash flow calculation was $21.0 million. $20.1
million of this was paid in December 2009 and $0.9 million
is included in the current portion of long term debt. There was
no required principal payment related to excess cash flow for
2008 and $14.0 million was paid based on the excess cash
flow calculation for the ten month period ended
December 31, 2007. The first lien term loan is due
February 28, 2013 and is collateralized by a first-priority
lien on substantially all assets and equity interests of the
Company as defined in the First Lien Credit and Guaranty
Agreement dated February 28, 2007.
The second lien term loan has interest payments due based on the
interest period selected (1, 2 or 3 months) at LIBOR or prime
rate, plus a 5.50% margin and 4.50% margin, respectively. The
interest rate was 5.74% and 5.98% at December 31, 2009 and
2008, respectively. The second lien term loan is due
February 28, 2014 and is collateralized by a
second-priority lien on substantially all assets and equity
interests of the Company’s U.S. subsidiaries as
defined in the Second Lien Credit
F-23
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
and Guaranty Agreement dated February 28, 2007.
U.S. assets totaled approximately $328.7 million as of
December 31, 2009.
The Company has a revolving credit facility with availability of
$60 million that can be borrowed in U.S. dollars or
Euros. The Company also has the ability to issue letters of
credit based on availability under the revolving credit
facility. Interest is payable monthly or quarterly at
LIBOR/Euribor or prime rate, plus an applicable margin based on
the Company’s leverage ratio. There was no outstanding
balance at December 31, 2009. Additionally, there is a
standby letter of credit fee, unused line fee and letter of
credit fronting fee. The letter of credit fee and unused line
fee (2.00% and 0.50%, respectively, at December 31, 2009;
and 1.75% and 0.375%, respectively, at December 31,
2008) are based on the Company’s leverage ratio. The
letter of credit fronting fee is 0.125%. The outstanding balance
of the revolving credit facility is due February 28, 2012.
The commitment fee related to the revolving credit facility was
$0.2 million, $0.2 million and $0.2 million for
the years ended December 31, 2009 and 2008 and the ten
month period ended December 31, 2007.
As part of the Company’s acquisition of Abieta Chemie GmbH,
which we refer to as “Abieta”, (see Note 16), the
Company assumed an outstanding bank loan of
€4.5 million ($6.0 million) at a fixed interest
rate of 4.80%. As of December 31, 2009, $0.7 million
was outstanding after repayments made during the year.
Prime loans are due quarterly on calendar quarters. LIBOR loans
are due based on the interest period selected (1, 2 or
3 months). As of December 31, 2009, interest was due
and paid bi-monthly for the U.S. dollar-denominated term
loans and the Euro-denominated term loan.
Deferred loan costs, net reported in the consolidated balance
sheets at December 31, 2009 and 2008, were
$7.7 million and $9.7 million, respectively. Deferred
loan costs are amortized over the applicable life of the
corresponding loan using a method that approximates the
effective interest method. For the years ended December 31,
2009 and 2008 and the ten month period ended December 31,
2007, $2.5 million, $2.1 million and
$2.3 million, respectively, were amortized and are included
in interest expense in the consolidated statements of operations.
The aggregate maturities of debt subsequent to December 31,
2009, are as follows (in thousands of U.S. dollars):
| |
|
|
|
|
|
Years Ending December
31,
|
|
|
|
|
|
|
2010
|
|
$
|
3,935
|
|
|
2011
|
|
|
2,602
|
|
|
2012
|
|
|
2,460
|
|
|
2013
|
|
|
210,263
|
|
|
2014
|
|
|
115,498
|
|
|
Thereafter
|
|
|
—
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
334,758
|
|
|
|
|
|
|
|
The first and second lien term loans and the revolving credit
facility contain certain financial covenants. The financial
covenants require that the Company maintain (i) a certain
interest coverage ratio, (ii) a certain leverage ratio, and
(iii) a maximum limit for capital expenditures in any year.
In addition, certain covenants substantially restrict the
Company’s ability to incur additional indebtedness, create
liens, make certain investments, sell assets, or pay dividends.
The Company’s obligations under these agreements may be
accelerated on certain events of default. As of
December 31, 2009 and 2008, the Company was in compliance
with the covenants.
The first and second lien credit agreements were amended on
July 24, 2008, to allow Rhône Capital to purchase up
to 30% of the aggregate principal amount outstanding on the
second lien term
F-24
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
loan. The amendments require that promptly upon such purchase by
Rhône Capital, at least 35% of the principal amount
purchased must be contributed to the Company. In exchange for
that contribution, the Company would issue additional equity
interests.
In September and November 2008, Rhône Capital acquired
$26.2 million and $1.0 million, respectively, face
value of the Company’s outstanding second lien debt for
$20.1 million and $0.7 million, respectively. In
accordance with the first amendment to the second lien credit
agreement, 35% of the debt was contributed to the Company in
exchange for an increase in Rhône Capital’s equity
interest in the Company. The contributed debt of
$9.2 million and $0.4 million, respectively, was
extinguished, resulting in a gain, net of expenses, on
extinguishment of $1.9 million which is reported in other
operating income in the consolidated statements of operations
and a contribution to paid-in-capital of $7.4 million.
Rhône Capital remains the holder of $17.6 million of
the Company’s outstanding second lien debt. Interest paid
to Rhône Capital was $1.1 million and
$0.4 million for the years ended December 31, 2009 and
2008.
The Company’s income tax expense (benefit) consists of the
following (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
|
Predecessor
|
|
|
|
|
|
|
|
|
|
|
March 1
|
|
|
|
January 1
|
|
|
|
|
Year ended
|
|
|
Year ended
|
|
|
through
|
|
|
|
through
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
February 28,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
2007
|
|
|
U.S. federal
|
|
$
|
89
|
|
|
$
|
(56
|
)
|
|
$
|
—
|
|
|
|
$
|
1,405
|
|
|
U.S. state
|
|
|
158
|
|
|
|
49
|
|
|
|
207
|
|
|
|
|
194
|
|
|
International
|
|
|
664
|
|
|
|
(78
|
)
|
|
|
1,590
|
|
|
|
|
953
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current
|
|
|
911
|
|
|
|
(85
|
)
|
|
|
1,797
|
|
|
|
|
2,552
|
|
|
U.S. federal
|
|
|
200
|
|
|
|
2,925
|
|
|
|
(9,714
|
)
|
|
|
|
258
|
|
|
U.S. state
|
|
|
(52
|
)
|
|
|
207
|
|
|
|
(708
|
)
|
|
|
|
35
|
|
|
International
|
|
|
2,772
|
|
|
|
(7,324
|
)
|
|
|
(288
|
)
|
|
|
|
(231
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deferred
|
|
|
2,920
|
|
|
|
(4,192
|
)
|
|
|
(10,710
|
)
|
|
|
|
62
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income tax expense (benefit)
|
|
$
|
3,831
|
|
|
$
|
(4,277
|
)
|
|
$
|
(8,913
|
)
|
|
|
$
|
2,614
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income tax expense (benefit) was based on tax rates in effect in
the countries and locations in which the Company conducts its
operations and related taxable income is earned. During 2007,
the impact of changes in international tax laws on the reported
tax provision was immaterial. In 2008, Sweden enacted a
reduction in the corporate income tax rate from 28.0% to 26.3%
effective January 1, 2009. The Company realized a tax
benefit of $0.4 million in 2008.
F-25
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
Income (loss) before income taxes was generated in the following
jurisdictions (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
|
Predecessor
|
|
|
|
|
|
|
|
|
|
|
March 1
|
|
|
|
January 1
|
|
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
|
through
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
February 28,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
2007
|
|
|
United States
|
|
$
|
1,299
|
|
|
$
|
8,753
|
|
|
$
|
(33,120
|
)
|
|
|
$
|
4,935
|
|
|
International
|
|
|
14,017
|
|
|
|
(40,009
|
)
|
|
|
(3,272
|
)
|
|
|
|
1,280
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total income (loss) before income tax expense (benefit)
|
|
$
|
15,316
|
|
|
$
|
(31,256
|
)
|
|
$
|
(36,392
|
)
|
|
|
$
|
6,215
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
A reconciliation of the income tax expense (benefit) using the
statutory Swedish income tax rate compared to the Company’s
effective tax rate for the years ended December 31, 2009
and 2008, the ten month period ended December 31, 2007, and
the two month period ended February 28, 2007 is as follows:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Predecessor
|
|
|
|
|
|
|
|
March 1
|
|
|
January 1
|
|
|
|
Year Ended
|
|
Year Ended
|
|
through
|
|
|
through
|
|
|
|
December 31,
|
|
December 31,
|
|
December 31,
|
|
|
February 28,
|
|
|
|
2009
|
|
2008
|
|
2007
|
|
|
2007
|
|
Statutory income tax rate
|
|
|
26.3
|
%
|
|
|
28.0
|
%
|
|
|
28.0
|
%
|
|
|
|
35.0
|
%
|
|
Tax on income of foreign subsidiaries and rate differential
|
|
|
(2.3
|
)
|
|
|
(2.1
|
)
|
|
|
10.8
|
|
|
|
|
4.4
|
|
|
Nondeductible transaction costs
|
|
|
—
|
|
|
|
—
|
|
|
|
(5.8
|
)
|
|
|
|
—
|
|
|
Permanent differences
|
|
|
(15.5
|
)
|
|
|
4.6
|
|
|
|
—
|
|
|
|
|
—
|
|
|
Tax credits
|
|
|
—
|
|
|
|
0.5
|
|
|
|
0.7
|
|
|
|
|
—
|
|
|
Change in valuation allowance
|
|
|
16.2
|
|
|
|
(15.8
|
)
|
|
|
(7.0
|
)
|
|
|
|
—
|
|
|
Reduction in Swedish tax rate
|
|
|
—
|
|
|
|
1.4
|
|
|
|
—
|
|
|
|
|
—
|
|
|
Increase in U.S. state tax rate
|
|
|
—
|
|
|
|
(1.2
|
)
|
|
|
—
|
|
|
|
|
2.4
|
|
|
Other — net
|
|
|
0.3
|
|
|
|
(1.7
|
)
|
|
|
(2.2
|
)
|
|
|
|
0.3
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Effective tax rate
|
|
|
25.0
|
%
|
|
|
13.7
|
%
|
|
|
24.5
|
%
|
|
|
|
42.1
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
F-26
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
The tax effects of significant temporary differences
representing deferred tax assets and liabilities at
December 31, 2009 and 2008, were as follows (in thousands
of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Deferred tax assets:
|
|
|
|
|
|
|
|
|
|
Accrued liabilities
|
|
$
|
734
|
|
|
$
|
1,052
|
|
|
Property, plant and equipment
|
|
|
4,136
|
|
|
|
5,319
|
|
|
Inventory
|
|
|
2,180
|
|
|
|
2,715
|
|
|
Tax credit and net operating loss carryforwards
|
|
|
51,313
|
|
|
|
46,686
|
|
|
Pension
|
|
|
2,549
|
|
|
|
2,314
|
|
|
Derivative instruments
|
|
|
957
|
|
|
|
2,761
|
|
|
Other
|
|
|
2,037
|
|
|
|
2,449
|
|
|
|
|
|
|
|
|
|
|
|
|
Gross deferred tax assets
|
|
|
63,906
|
|
|
|
63,296
|
|
|
Less valuation allowance
|
|
|
(45,305
|
)
|
|
|
(41,167
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net deferred tax assets
|
|
|
18,601
|
|
|
|
22,129
|
|
|
Deferred tax liabilities:
|
|
|
|
|
|
|
|
|
|
Intangible assets
|
|
|
(34,997
|
)
|
|
|
(42,346
|
)
|
|
Property, plant and equipment
|
|
|
(19,672
|
)
|
|
|
(15,434
|
)
|
|
Other
|
|
|
(539
|
)
|
|
|
(580
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Gross deferred tax liabilities
|
|
|
(55,208
|
)
|
|
|
(58,360
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net deferred tax liabilities
|
|
$
|
(36,607
|
)
|
|
$
|
(36,231
|
)
|
|
|
|
|
|
|
|
|
|
|
The Company has valuation allowances against its deferred tax
assets of $45.3 million and $41.2 million at
December 31, 2009 and 2008, respectively. A
$4.1 million increase in the valuation allowance during
2009 was due to net operating losses and other deferred tax
assets generated in 2009 by international subsidiaries.
As of December 31, 2009 and 2008, the Company had worldwide
net operating loss carryforwards of $160.5 million and
$149.7 million, respectively, of which $41.4 million
and $36.8 million expire in varying amounts between 2017
and 2029, while the remaining $119.1 million and
$112.9 million have indefinite lives based on the laws of
the jurisdictions in which they were generated. The Company has
provided valuation allowances against certain Net Operating Loss
(NOL) carryforwards due to the uncertainty of their realization.
The Company files income tax returns in many countries,
principally in the United States, Finland, United Kingdom,
France, Luxembourg, Netherlands, Germany, and Sweden. Generally,
tax years 2003 through 2009 remain open and subject to
examination by the relevant tax authorities.
F-27
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
A reconciliation of the beginning and ending amount of
unrecognized tax benefits for 2009, 2008 and 2007 is as follows
(in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Predecessor
|
|
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
March 1 through
|
|
|
January 1 through
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
February 28,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
2007
|
|
|
Unrecognized Tax Benefit — Beginning of Year
|
|
$
|
530
|
|
|
$
|
329
|
|
|
$
|
224
|
|
|
$
|
224
|
|
|
Gross increases — tax positions in prior period
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Gross decreases — tax positions in prior period
|
|
|
(13
|
)
|
|
|
(48
|
)
|
|
|
—
|
|
|
|
—
|
|
|
Gross increases — tax positions in current period
|
|
|
171
|
|
|
|
249
|
|
|
|
105
|
|
|
|
—
|
|
|
Settlement
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Lapse in Statute of Limitations
|
|
|
(264
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Unrecognized Tax Benefit — End of Year
|
|
$
|
424
|
|
|
$
|
530
|
|
|
$
|
329
|
|
|
$
|
224
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The Company recognizes interest accrued related to unrecognized
tax benefits in interest expense and penalties as a component of
income tax expense. During the years ended December 31,
2009 and 2008, the Company recognized approximately
$0.1 million and $0.2 million in interest and
penalties and no amounts during the 10 month period ended
December 31, 2007. The Company had approximately
$0.4 million and $0.3 million for the payment of
interest and penalties accrued at December 31, 2009 and
2008, respectively.
(a) U.S. Defined Benefit
Plan — The Company sponsors a noncontributory
defined benefit pension plan in the U.S.
All U.S. employees hired prior to July 2004 and retirees of the
Company participate in International Paper’s defined
benefit pension plans. International Paper remains responsible
for all benefits related to years of service prior to
December 31, 2007. The Company implemented its own defined
benefit pension plan for eligible domestic employees on
March 1, 2007. For the two month period ended
February 28, 2007, International Paper allocated periodic
pension costs to the Company of $0.5 million.
Net periodic pension expense for the Company’s
U.S. defined benefit pension plan for the years ended
December 31, 2009 and 2008 and the ten month period ended
December 31, 2007 was as follows (in thousands of
U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 1
|
|
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
|
Service cost
|
|
$
|
790
|
|
|
$
|
817
|
|
|
$
|
706
|
|
|
Interest cost
|
|
|
226
|
|
|
|
142
|
|
|
|
58
|
|
|
Expected return on plan assets
|
|
|
(9
|
)
|
|
|
(12
|
)
|
|
|
—
|
|
|
Actuarial loss recognized
|
|
|
—
|
|
|
|
4
|
|
|
|
—
|
|
|
Amortization of prior service cost
|
|
|
108
|
|
|
|
112
|
|
|
|
92
|
|
|
Amortization of net loss
|
|
|
17
|
|
|
|
—
|
|
|
|
—
|
|
|
Curtailment expense
|
|
|
206
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net periodic benefit cost
|
|
$
|
1,338
|
|
|
$
|
1,063
|
|
|
$
|
856
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The U.S. defined benefit pension plan obligations and
assets are measured annually at December 31.
F-28
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
Pension related
amounts in accumulated other comprehensive income (AOCI) as of
December 31, 2009 and 2008 was comprised of the following
(in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Prior service cost
|
|
$
|
(830
|
)
|
|
$
|
(1,028
|
)
|
|
Net loss
|
|
|
(169
|
)
|
|
|
(354
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Total accumulated other comprehensive income at end of year
|
|
|
(999
|
)
|
|
|
(1,382
|
)
|
|
Less: Income taxes
|
|
|
367
|
|
|
|
477
|
|
|
|
|
|
|
|
|
|
|
|
|
Net accumulated other comprehensive income at end of year
|
|
$
|
(632
|
)
|
|
$
|
(905
|
)
|
|
|
|
|
|
|
|
|
|
|
Other changes in
plan assets and benefit obligations recognized in other
comprehensive income for the years ended December 31, 2009
and 2008 and the ten months ended December 31, 2007 were as
follows (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
|
Prior service cost arising during the year
|
|
$
|
(42
|
)
|
|
$
|
(518
|
)
|
|
$
|
(1,213
|
)
|
|
Net gain arising during the year
|
|
|
168
|
|
|
|
4
|
|
|
|
160
|
|
|
Prior service cost (credit) recognized during the year
|
|
|
132
|
|
|
|
(19
|
)
|
|
|
—
|
|
|
Amortization of prior service cost
|
|
|
108
|
|
|
|
112
|
|
|
|
92
|
|
|
Amortization of net loss
|
|
|
17
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total recognized in other comprehensive income
|
|
|
383
|
|
|
|
(421
|
)
|
|
|
(961
|
)
|
|
Less: Income taxes
|
|
|
(140
|
)
|
|
|
155
|
|
|
|
353
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net amount recognized in other comprehensive income
|
|
$
|
243
|
|
|
$
|
(266
|
)
|
|
$
|
(608
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The estimated amount to be amortized from accumulated other
comprehensive income into net periodic benefit cost in 2010, are
as follows (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
2010
|
|
|
|
|
Amortization of:
|
|
|
|
|
|
Net loss
|
|
$
|
—
|
|
|
Prior service cost
|
|
$
|
106
|
|
As of December 31, 2009 and 2008, the U.S. defined
benefit plan funded status was as follows (in thousands of
U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Change in projected benefit obligation:
|
|
|
|
|
|
|
|
|
|
Projected benefit obligation at beginning of year
|
|
$
|
3,293
|
|
|
$
|
1,817
|
|
|
Service cost
|
|
|
790
|
|
|
|
817
|
|
|
Interest cost
|
|
|
226
|
|
|
|
142
|
|
|
Amendments
|
|
|
42
|
|
|
|
19
|
|
|
Actuarial (gain) loss
|
|
|
(178
|
)
|
|
|
510
|
|
|
Benefits paid
|
|
|
(77
|
)
|
|
|
(12
|
)
|
|
Special termination benefits
|
|
|
74
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
Projected benefit obligation at end of year
|
|
|
4,170
|
|
|
|
3,293
|
|
F-29
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
| |
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Change in plan assets:
|
|
|
|
|
|
|
|
|
|
Fair value of plan assets at beginning of year
|
|
|
584
|
|
|
|
10
|
|
|
Actual return on plan assets
|
|
|
(1
|
)
|
|
|
3
|
|
|
Employer contributions
|
|
|
1,898
|
|
|
|
583
|
|
|
Benefits paid
|
|
|
(77
|
)
|
|
|
(12
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Fair value of plan assets at end of year
|
|
|
2,404
|
|
|
|
584
|
|
|
|
|
|
|
|
|
|
|
|
|
Funded status
|
|
$
|
(1,766
|
)
|
|
$
|
(2,709
|
)
|
|
|
|
|
|
|
|
|
|
|
Accumulated benefit obligation was $4.2 million and
$3.3 million as of December 31, 2009 and 2008,
respectively.
Amounts at December 31, 2009 and 2008, recognized in the
balance sheet consist of (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Noncurrent liabilities
|
|
$
|
(1,766
|
)
|
|
$
|
(2,709
|
)
|
|
|
|
|
|
|
|
|
|
|
The Company expects to contribute $1.0 million to the
pension plan in 2010.
Assumptions — Assumptions used to determine
benefit obligations at December 31, 2009 and 2008,
respectively, are as follows:
| |
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
2008
|
|
|
|
Discount rate
|
|
|
5.95
|
%
|
|
|
6.14
|
%
|
|
Rate of compensation increase
|
|
|
N/A
|
|
|
|
N/A
|
|
Assumptions used to determine net periodic pension cost for the
years ended December 31, 2009 and 2008 and the ten month
period ended December 31, 2007 are as follows:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 1
|
|
|
|
Year Ended
|
|
Year Ended
|
|
through
|
|
|
|
December 31,
|
|
December 31,
|
|
December 31,
|
|
|
|
2009
|
|
2008
|
|
2007
|
|
|
|
Discount rate
|
|
|
5.95
|
%
|
|
|
6.25
|
%
|
|
|
5.75
|
%
|
|
Expected long-term rate of return on plan assets
|
|
|
7.00
|
%
|
|
|
7.00
|
%
|
|
|
N/A
|
|
|
Rate of compensation increase
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
The expected long-term rate of return on plan assets is based on
both the actual asset allocation as well as the investment
policy target allocation of the asset portfolio between various
asset classes and the expected rate of return of each asset
class over various periods of time.
Plan Assets — The Company’s defined
benefit pension plan asset contributions for December 31,
2009 and 2008, was $1.9 million and $0.6 million,
respectively.
In years 2009, 2008 and 2007, the Company was in the process of
developing an investment strategy for the U.S. defined
benefit plan. During this period the Company invested in highly
liquid indexed money market funds with the goal of capital
preservation. As of December 31, 2009 and
F-30
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
2008, 100% of the assets for the Company’s
U.S. defined benefit pension plans were in highly liquid
indexed money market funds.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Fair Value Measurements at December 31, 2009
|
|
|
|
|
|
Quoted Prices in
|
|
Significant
|
|
Significant
|
|
|
|
|
|
Active Market for
|
|
Observable
|
|
Unobservable
|
|
|
|
|
|
Identical Assets
|
|
Inputs
|
|
Inputs
|
|
|
|
Total
|
|
(Level 1)
|
|
(Level 2)
|
|
(Level 3)
|
|
|
|
Federated Investors Prime Obligation Fund
|
|
$
|
2,404
|
|
|
$
|
2,404
|
|
|
|
—
|
|
|
|
—
|
|
The Plan has invested all of its funds in the Federated
Investors Prime Obligations Fund. This money market fund is
rated triple A by all three of the credit rating agencies. The
Plan’s investment is valued at the closing price of the
fund, which approximates fair value.
Estimated future benefit payments at December 31, 2009, for
the Company’s defined benefit pension plan are as follows
(in thousands of U.S. dollars):
| |
|
|
|
|
|
Years Ending December
31,
|
|
|
|
|
|
2010
|
|
$
|
97
|
|
|
2011
|
|
$
|
126
|
|
|
2012
|
|
$
|
171
|
|
|
2013
|
|
$
|
219
|
|
|
2014
|
|
$
|
269
|
|
|
2015-2019
|
|
$
|
2,132
|
|
(b) Non U.S. Defined Benefit Plans
— The Company sponsors defined benefit pension
and retirement plans in certain foreign subsidiaries. Generally,
the Company’s non U.S. defined benefit pension plans are
funded using the projected benefit as a target, in countries
where funding of benefit plans is required.
Net periodic pension expense for the Company’s non
U.S. plans for the years ended December 31, 2009 and
2008, the ten month period ended December 31, 2007, and the
two month period ended February 28, 2007 was as follows (in
thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
|
Predecessor
|
|
|
|
|
|
|
|
|
|
|
March 1
|
|
|
|
January 1
|
|
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
|
through
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
February 28,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
2007
|
|
|
Service cost
|
|
$
|
1,095
|
|
|
$
|
1,745
|
|
|
$
|
1,707
|
|
|
|
$
|
347
|
|
|
Interest cost
|
|
|
2,917
|
|
|
|
2,510
|
|
|
|
2,513
|
|
|
|
|
501
|
|
|
Expected return on plan assets
|
|
|
(2,416
|
)
|
|
|
(2,594
|
)
|
|
|
(2,511
|
)
|
|
|
|
—
|
|
|
Amortization of net (gain) loss
|
|
|
(165
|
)
|
|
|
(97
|
)
|
|
|
17
|
|
|
|
|
—
|
|
|
Settlement
|
|
|
(248
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net periodic benefit cost
|
|
$
|
1,183
|
|
|
$
|
1,564
|
|
|
$
|
1,726
|
|
|
|
$
|
848
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The non U.S. pension plans obligations and assets are
measured annually at December 31.
F-31
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
Pension related amounts recognized in AOCI as of
December 31, 2009 and 2008 was comprised of the following
(in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Prior service cost
|
|
$
|
(76
|
)
|
|
$
|
—
|
|
|
Net (loss) gain
|
|
|
(1,505
|
)
|
|
|
734
|
|
|
|
|
|
|
|
|
|
|
|
|
Total accumulated other comprehensive income at end of year
|
|
|
(1,581
|
)
|
|
|
734
|
|
|
Income tax expense (benefit)
|
|
|
(611
|
)
|
|
|
(816
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net Accumulated other comprehensive income at end of year
|
|
$
|
(2,192
|
)
|
|
$
|
(82
|
)
|
|
|
|
|
|
|
|
|
|
|
Other changes in
plan assets and benefit obligations recognized in other
comprehensive income for the years ended December 31, 2009
and 2008 and the ten month period ended December 31, 2007
were as follows (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
|
Prior service cost arising during the year
|
|
$
|
(74
|
)
|
|
$
|
—
|
|
|
$
|
—
|
|
|
Net gain (loss) arising during the year
|
|
|
260
|
|
|
|
(2,098
|
)
|
|
|
1,513
|
|
|
Prior service cost recognized during the year
|
|
|
—
|
|
|
|
(103
|
)
|
|
|
—
|
|
|
Amortization of net (gain) loss
|
|
|
(2,581
|
)
|
|
|
—
|
|
|
|
16
|
|
|
Exchange rate loss recognized during the year
|
|
|
245
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total recognized in other comprehensive income
|
|
|
(2,150
|
)
|
|
|
(2,201
|
)
|
|
|
1,529
|
|
|
Less: Income taxes
|
|
|
(514
|
)
|
|
|
423
|
|
|
|
711
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net amount recognized in other comprehensive income
|
|
$
|
(2,664
|
)
|
|
$
|
(1,778
|
)
|
|
$
|
2,240
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The estimated amount to be amortized from accumulated other
comprehensive income into net periodic benefit cost in fiscal
year 2010 is as follows (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
2010
|
|
|
|
Amortization of:
|
|
|
|
|
|
Net gain
|
|
$
|
(108
|
)
|
|
Prior service cost
|
|
$
|
5
|
|
F-32
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
As of December 31, 2009 and 2008, the plan funded status
for the Non U.S. defined benefit and retirement plans was as
follows (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
Non U.S. Plan
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Change in projected benefit obligation:
|
|
|
|
|
|
|
|
|
|
Projected benefit obligation at beginning of year
|
|
$
|
43,640
|
|
|
$
|
59,367
|
|
|
Service cost
|
|
|
1,095
|
|
|
|
1,745
|
|
|
Interest cost
|
|
|
2,917
|
|
|
|
2,510
|
|
|
Employee contributions
|
|
|
291
|
|
|
|
458
|
|
|
Plan amendments
|
|
|
74
|
|
|
|
—
|
|
|
Actuarial loss (gain)
|
|
|
5,633
|
|
|
|
(5,901
|
)
|
|
Settlement
|
|
|
(248
|
)
|
|
|
—
|
|
|
Acquisition
|
|
|
2,950
|
|
|
|
—
|
|
|
Benefits paid
|
|
|
(1,722
|
)
|
|
|
(1,673
|
)
|
|
Actual expenses
|
|
|
(75
|
)
|
|
|
—
|
|
|
Foreign currency exchange rate changes
|
|
|
3,654
|
|
|
|
(12,866
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Projected benefit obligation at end of year
|
|
|
58,209
|
|
|
|
43,640
|
|
|
Change in plan assets:
|
|
|
|
|
|
|
|
|
|
Fair value of plan assets at beginning of year
|
|
|
35,304
|
|
|
|
51,109
|
|
|
Actual return on plan assets
|
|
|
5,893
|
|
|
|
(5,026
|
)
|
|
Employer contributions
|
|
|
2,564
|
|
|
|
2,099
|
|
|
Plan participants’ contributions
|
|
|
291
|
|
|
|
458
|
|
|
Benefits paid
|
|
|
(1,722
|
)
|
|
|
(1,673
|
)
|
|
Actual expenses
|
|
|
(75
|
)
|
|
|
—
|
|
|
Foreign currency exchange rate changes
|
|
|
2,935
|
|
|
|
(11,663
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Fair value of plan assets at end of year
|
|
|
45,190
|
|
|
|
35,304
|
|
|
|
|
|
|
|
|
|
|
|
|
Funded status
|
|
$
|
(13,019
|
)
|
|
$
|
(8,336
|
)
|
|
|
|
|
|
|
|
|
|
|
Accumulated benefit obligation was $54.7 million and
$38.1 million as of December 31, 2009 and 2008,
respectively.
Amounts at December 31, 2009 and 2008, recognized in the
balance sheet consist of (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Noncurrent assets
|
|
$
|
700
|
|
|
$
|
102
|
|
|
Current liabilities
|
|
|
(333
|
)
|
|
|
(168
|
)
|
|
Noncurrent liabilities
|
|
|
(13,386
|
)
|
|
|
(8,270
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Total asset/(liability)
|
|
$
|
(13,019
|
)
|
|
$
|
(8,336
|
)
|
|
|
|
|
|
|
|
|
|
|
The Company expects to contribute $2.9 million to the
pension plans in 2010.
Assumptions — The assumptions used to determine
benefit obligations at December 31, 2009 and 2008, are as
follows:
| |
|
|
|
|
|
|
|
2009
|
|
2008
|
|
|
|
Discount rates
|
|
4.50% - 5.75%
|
|
5.75% - 6.50%
|
|
Rate of compensation increase
|
|
2.00% - 4.00%
|
|
2.00% - 4.00%
|
F-33
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
Assumptions used to determine net periodic pension cost for the
years ended December 31, 2009 and 2008, the ten month
period ended December 31, 2007, and the two month period
ended February 28, 2007 are as follows:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Predecessor
|
|
|
|
|
|
|
|
March 1
|
|
|
January 1
|
|
|
|
Year Ended
|
|
Year Ended
|
|
through
|
|
|
through
|
|
|
|
December 31,
|
|
December 31,
|
|
December 31,
|
|
|
February 28,
|
|
|
|
2009
|
|
2008
|
|
2007
|
|
|
2007
|
|
Discount rate
|
|
5.75% – 6.50%
|
|
5.25% – 5.80%
|
|
4.25% – 5.80%
|
|
|
|
4.74%
|
|
|
Expected long-term rate of return on plan assets
|
|
3.50% – 7.00%
|
|
3.50% – 7.00%
|
|
1.75% – 6.8%
|
|
|
|
6.53%
|
|
|
Rate of compensation increase
|
|
2.00% – 4.00%
|
|
2.00% – 4.35%
|
|
2.00% – 4.35%
|
|
|
|
3.25%
|
|
The expected long-term rate of return on plan assets is based on
both the actual asset allocation as well as the investment
policy target allocation of the asset portfolio between various
asset classes and the expected rate of return of each asset
class over various periods of time.
Plan
Assets
The Company’s
non-U.S. plans
primarily have assets in the U.K. and the Netherlands. The
target allocations for plan assets in the U.K. are
50 percent equity securities, 30 percent corporate
bonds, 20 percent U.K. fixed interest or index linked
Gilts. Equity securities primarily include investments in
large-cap and mid-cap companies primarily located in the U.K.
Fixed income securities include corporate bonds of companies and
bonds issued by the U.K. government. Assets in the Netherlands
primarily relate to insurance contracts.
Assets and liabilities for the Company’s foreign defined
benefit and retirement plans are predominantly denominated in
pounds sterling.
As of December 31, 2009 and 2008, the weighted average
asset allocations for the Company’s foreign defined benefit
pension plans at December 31, 2009, by asset category are
as follows:
| |
|
|
|
|
|
|
|
|
|
|
|
2009
|
|
2008
|
|
|
|
Asset category:
|
|
|
|
|
|
|
|
|
|
Cash or cash equivalents
|
|
|
0
|
%
|
|
|
0
|
%
|
|
Equities
|
|
|
38
|
%
|
|
|
42
|
%
|
|
Fixed income
|
|
|
40
|
%
|
|
|
40
|
%
|
|
Insurance contracts
|
|
|
21
|
%
|
|
|
18
|
%
|
|
Other
|
|
|
1
|
%
|
|
|
0
|
%
|
Estimated future benefit payments at December 31, 2009, for
the Company’s foreign defined benefit and retirement
pension plans are as follows (in thousands of U.S. dollars):
| |
|
|
|
|
|
Years Ending December
31
|
|
|
|
|
|
2010
|
|
$
|
1,607
|
|
|
2011
|
|
$
|
1,652
|
|
|
2012
|
|
$
|
1,715
|
|
|
2013
|
|
$
|
1,803
|
|
|
2014
|
|
$
|
1,868
|
|
|
2015-2019
|
|
$
|
11,345
|
|
F-34
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Fair Value Measurements at December 31, 2009
|
|
|
|
|
|
|
|
Quoted Prices in
|
|
|
Significant
|
|
|
Significant
|
|
|
|
|
|
|
|
Active Market for
|
|
|
Observable
|
|
|
Unobservable
|
|
|
|
|
|
|
|
Identical Assets
|
|
|
Inputs
|
|
|
Inputs
|
|
|
Asset Category
|
|
Total
|
|
|
(Level 1)
|
|
|
(Level 2)
|
|
|
(Level 3)
|
|
|
|
|
Cash or cash equivalents
|
|
$
|
17,473
|
|
|
$
|
17,473
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
Equities(i)
|
|
|
17,762
|
|
|
|
17,762
|
|
|
|
—
|
|
|
|
—
|
|
|
Fixed income
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Insurance contracts(ii)
|
|
|
9,441
|
|
|
|
—
|
|
|
|
—
|
|
|
|
9,441
|
|
|
Other
|
|
|
514
|
|
|
|
514
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
45,190
|
|
|
$
|
35,749
|
|
|
$
|
—
|
|
|
$
|
9,441
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(i) |
|
This category comprises low-cost equity index funds that are
valued based on market prices. |
|
|
|
|
(ii) |
|
This category represents insurance contracts that are valued by
reference to the value of bonds with similar maturities. Our
third party pension administrator uses these loan amounts and
the agreed future interest profit amount and discounts them
using government issued yield curves to determine fair value. |
| |
|
|
|
|
|
Rollforward of level 3 plan assets:
|
|
|
|
|
|
Balance at beginning of year
|
|
$
|
6,457
|
|
|
Benefits paid
|
|
|
(212
|
)
|
|
Realized and unrealized gain recognized in other comprehensive
income
|
|
|
1,217
|
|
|
Purchases
|
|
|
1,771
|
|
|
Actual expenses
|
|
|
(75
|
)
|
|
Exchange rates
|
|
|
283
|
|
|
|
|
|
|
|
|
Balance at end of year
|
|
$
|
9,441
|
|
|
|
|
|
|
|
|
|
|
(c)
|
401(k) Savings
Plan (U.S.)
|
Upon the Acquisition, a defined contribution Employee Savings
Plan was created for all US-based employees, the Arizona
Chemical Savings Plan. The savings plan is a tax-qualified
401(k) plan, and employees’ contributions are matched by
the Company on the following basis: 70 cents on the dollar for
the first four percent of the employee’s contribution and
50 cents on the dollar for up to an additional four percent.
As a means to further enhance retirement savings for
U.S.-based
employees in light of the absence of a defined benefit pension
plan, the Company maintains an annual profit sharing award plan.
This award comes in the form of a discretionary gift
contribution made by the Company directly to each
U.S.-based
salaried employee’s savings plan account based on the
Company’s performance. The award is intended to be variable
in nature and it could be less or more than four percent
depending upon the performance of the business. All employees
receive the same percentage award. The Company’s
contributions under these plans amounted to $2.6 million,
$2.5 million, $2.1 million and $0.2 million
approximately for the years ended December 31, 2009 and
2008, the ten month period ended December 31, 2007, and the
two month period ended February 28, 2007, respectively.
|
|
|
(d)
|
Long-term
Service Award Plan
|
The Company maintains a Long-Term Service Awards program, a
deferred compensation plan, for qualified employees in our
German operations. Under the German plan, qualified employees
F-35
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
receive a service gratuity (“Jubilee”) payment once
they have reached a certain number of years of service. Active
employees are granted the following jubilee benefits at the time
of the jubilee:
| |
|
|
|
|
|
|
|
Service
|
|
Extra Holiday
|
|
Benefit
|
|
|
|
25 years
|
|
|
6 days
|
|
|
1/12 of yearly salary
|
|
40 years
|
|
|
6 days
|
|
|
2/12 of yearly salary
|
|
50 years
|
|
|
11 days
|
|
|
3/12 of yearly salary
|
The obligation at December 31, 2009 was $0.1 million.
The rates assumed in the actuarial calculations for the Jubilee
plan at December 31, 2009 are as follows:
| |
|
|
|
|
|
Interest rate
|
|
|
5.25
|
%
|
|
Salary increase
|
|
|
3.00
|
%
|
|
Fluctuation rate
|
|
|
2.00
|
%
|
The following table shows the expected future benefits to be
paid assuming 100% plan participation (in thousands of
U.S. dollars).
| |
|
|
|
|
|
2010
|
|
$
|
16
|
|
|
2011
|
|
$
|
23
|
|
|
2012
|
|
$
|
32
|
|
|
2013
|
|
$
|
22
|
|
|
2014
|
|
$
|
—
|
|
|
2015 – 2019
|
|
$
|
13
|
|
|
|
|
(e)
|
Early
Retirement and Post-employment Programs
|
In Germany, Altersteilzeit (“ATZ”) is an early
retirement program established by law, and is designed to create
an incentive for employees, within a certain age group, to
transition from (full or part-time) employment into retirement
before their legal retirement age. The German government
provides a subsidy to employers taking advantage of this
legislation for bonuses paid to the employee and the additional
contributions paid into the German government pension scheme
under an ATZ arrangement for a maximum of six years. To receive
this subsidy, an employer must meet certain criteria established
by the German government. The Company accrues for ATZ based on
current and future contracts.
The rates assumed by the Company in the actuarial calculations
for the ATZ at December 31, 2009 are as follows:
| |
|
|
|
|
|
Interest rate
|
|
|
5.25
|
%
|
|
Salary increase
|
|
|
3.00
|
%
|
|
Fluctuation rate
|
|
|
2.00
|
%
|
The obligation at December 31, 2009 was $0.3 million.
The following table shows the expected benefits to be paid based
on expected plan participation, net of governmental subsidies
(in thousands of U.S. dollars).
| |
|
|
|
|
|
2010
|
|
$
|
182
|
|
|
2011
|
|
$
|
215
|
|
|
2012
|
|
$
|
122
|
|
|
2013
|
|
$
|
—
|
|
|
2014
|
|
$
|
—
|
|
|
2015 – 2019
|
|
$
|
—
|
|
F-36
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
|
|
|
14.
|
Related Party
Transactions
|
Management
Agreement
In connection with the Acquisition, the Company entered into a
Management Agreement with Rhône Group LLC, an affiliate of
Rhône Capital. Under this agreement, Rhône Group LLC,
in exchange for providing certain management services, has
received a management fee of €1,000,000 plus expenses per
year, payable semi-annually on each January 1 and July 1.
Fees and expenses totaled $1.5 million, $2.0 million,
and $1.3 million for the years ended December 31, 2009
and 2008 and the ten month period ended December 31, 2007,
respectively.
Purchase and
Contribution of Indebtedness
In a series of transactions during the third and fourth quarters
of 2008, Rhône Capital, through Rhône
Partners III L.P., Rhône Coinvestment
Partners III L.P. and Rhône Offshore
Partners III L.P. (collectively referred to as
“Rhône Funds”) acquired approximately
$27.2 million of the debt outstanding under the
Company’s Second Lien Credit Agreement. The Rhône
Funds contributed $9.5 million of this debt to the Company
in accordance with the terms of our credit agreements.
The Company pays the Rhône Funds interest on the second
lien debt they hold in accordance with the terms of our Second
Lien Credit Agreement. The total amount of interest paid to the
Rhône Funds on this debt was $1.1 million and
$0.4 million for the years ended December 31, 2009 and
2008.
Settlement
Agreement
On November 17, 2009, the Company entered into a Settlement
Agreement and Mutual Release (“Settlement Agreement”),
with our parent, Lux Holdco and International Paper. Under the
terms of the Settlement Agreement, the Company and Lux Holdco
agreed to settle a number of claims against International Paper
in exchange for the forgiveness of $11.4 million working
capital adjustments payable to International Paper and a payment
of $1.7 million to Lux Holdco by International Paper. The
majority of this settlement was recorded as a purchase price
adjustment as discussed in Note 1, and the Company recorded
a gain on settlement of $1.3 million. The gain on
settlement is reported in Other Income.
Under the terms of the Settlement Agreement, in the event that
(i) the Company’s
out-of-pocket
costs relating to the closure of a landfill acquired from Stora
Enso are less than $0.8 million, or (ii) the
Company’s
out-of-pocket
costs relating to soil and groundwater contamination at our
facility in Sandarne, Sweden are less than $0.3 million,
the Company is required to pay International Paper 50% of the
difference between that amount and actual
out-of-pocket
costs for that claim.
Management
Loans
The MIVs enable management to invest indirectly in the Company.
Beginning in July 2007, the Company provided loans to certain
members of our management to assist them in financing these
investments. Each of these loans bears interest at a rate of
7.88% and the principal outstanding at December 31, 2009
was $3.1 million. The Company had a corresponding payable
to AZ Chem Investments Partners LP for the same amount,
which was paid during 2009. See Note 17 “Share-Based
Compensation” and Note 20 “Subsequent
Events” for further details.
F-37
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
Arboris,
LLC
We hold a 10% investment in Arboris, LLC. We supply Arboris, LLC
with pitch which is used as a raw material in the sterol
manufacturing process. Revenue related to the sale of pitch is
recognized when the sterol finished product is sold to external
customers. We recognized revenue of $6.4 million,
$6.8 million, $3.9 million and $0.9 million for
the years ended December 31, 2009 and 2008, the ten month
period ended December 31, 2007 and the two month period
ended February 28, 2007, respectively, related to the sale
of pitch.
We also perform plant and administrative services for Arboris,
LLC. We billed Arboris, LLC $4.3 million,
$4.9 million, $9.2 million and $2.2 million for
the years ended December 31, 2009 and 2008, the ten month
period ended December 31, 2007 and the two month period
ended February 28, 2007, respectively, related to these
services. Prior to 2008, these services included other material
and logistic cost reimbursements. These amounts were recorded as
a reduction of the cost of performing such services.
|
|
|
15.
|
Commitments and
Contingencies
|
Operating Leases — The Company leases
administrative, manufacturing and warehousing facilities, and
machinery and other equipment under non-cancelable leases that
expire at various dates through 2015. Rental expense under
operating leases totaled $12.3 million, $16.2 million,
$11.5 million and $2.1 million for the years ended
December 31, 2009 and 2008, the ten month period ended
December 31, 2007 and the two month period ended
February 28, 2007, respectively.
As of December 31, 2009, the future minimum rental payments
due under noncancelable operating leases that have initial or
remaining lease terms in excess of one year are as follows (in
thousands of U.S. dollars):
| |
|
|
|
|
|
Years Ending December
31,
|
|
|
|
|
|
|
2010
|
|
$
|
9,842
|
|
|
2011
|
|
|
8,708
|
|
|
2012
|
|
|
4,046
|
|
|
2013
|
|
|
3,743
|
|
|
2014
|
|
|
3,691
|
|
|
Thereafter
|
|
|
3,532
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
33,562
|
|
|
|
|
|
|
|
Purchase Obligations — The Company has
entered into unconditional purchase agreements with terms of
more than one year that entail fixed payments for periods
lasting up to 2019. Amounts to be paid under these unconditional
purchase agreements total $3.1 million in 2010,
$2.4 million in 2011, $2.4 million in 2012,
$0.3 million in 2013, $0.3 million in 2014, and
$1.2 million in subsequent years.
Litigation — The Company is a party to
various lawsuits, claims, and contingent liabilities arising
from the conduct of its business; however, in the opinion of
management, based on the advice of counsel, they are not
expected to have a material adverse effect on the consolidated
results of operations, cash flows, or financial position of the
Company.
Forward Contracts — We typically enter
into natural gas purchase contracts that extend no more than
twelve months into the future for between 50% - 70% of our
estimated natural gas consumption in our North American
manufacturing facilities. As of December 31, 2009 we had
open purchase commitments of $4.4 million.
F-38
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
|
|
|
16.
|
Acquisition of
Businesses
|
On February 12, 2009, we acquired 100% of the outstanding
equity of Abieta for approximately $9.9 million in cash.
Abieta is a rosin upgrading business focused on emulsifiers for
polymerization and supplies major rubber and plastics
manufacturers in Western Europe.
Abieta has one manufacturing location in Germany. The Company
recognized the assets acquired and liabilities assumed in Abieta
at the acquisition date. Fair values and the results of
operations of Abieta are included in the consolidated financial
statements from the date of acquisition. A valuation of the
acquired assets and liabilities was completed in the third
quarter of 2009, and the purchase price was allocated as follows
(in millions of U.S. dollars):
| |
|
|
|
|
|
|
|
Accounts receivable
|
|
$
|
0.5
|
|
|
Inventory
|
|
|
7.9
|
|
|
Property, plant and equipment
|
|
|
10.4
|
|
|
Intangible assets
|
|
|
3.9
|
|
|
Other assets
|
|
|
0.8
|
|
|
|
|
|
|
|
|
Total assets acquired
|
|
|
23.5
|
|
|
Accounts payable
|
|
|
0.6
|
|
|
Pensions
|
|
|
2.3
|
|
|
Bank loan
|
|
|
6.0
|
|
|
Deferred taxes
|
|
|
0.1
|
|
|
Other liabilities
|
|
|
3.6
|
|
|
|
|
|
|
|
|
Total liabilities
|
|
|
12.6
|
|
|
Less cash acquired
|
|
|
(1.1
|
)
|
|
|
|
|
|
|
|
Total liabilities assumed
|
|
|
11.5
|
|
|
Less gain on Acquisition
|
|
|
2.1
|
|
|
|
|
|
|
|
|
Net assets acquired
|
|
$
|
9.9
|
|
|
|
|
|
|
|
The acquired intangible assets of the Company consist of the
following (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
Remaining
|
|
|
|
|
|
|
Economic Life
|
|
Intangible Assets
Valued
|
|
Fair Value
|
|
|
(Years)
|
|
|
|
Trade name
|
|
$
|
395
|
|
|
Indefinite
|
|
Developed technology
|
|
|
1,052
|
|
|
10
|
|
Customer relationships
|
|
|
2,499
|
|
|
6
|
|
|
|
|
|
|
|
|
|
Total identifiable intangible assets
|
|
$
|
3,946
|
|
|
|
|
|
|
|
|
|
|
|
Pro forma results of operations as if the business had been
acquired as of the beginning of the year of acquisition and as
of the prior-year period have not been presented, as the impact
on the Company’s consolidated financial results would not
have been material.
|
|
|
17.
|
Share-Based
Compensation
|
Share-based compensation cost is measured at the grant date
based on the fair value of the award and is recognized as
expense over the requisite service period.
F-39
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
In June 2007, the MIVs were formed to permit certain of our
executives and members of our Board of Directors to invest
indirectly in our Company, on substantially the same terms as
the Rhône Funds and International Paper, and to establish a
vehicle to enable our executives to share in the profits of our
Company based on future performance. As described below, our
executives purchased equity interests in the MIVs and each
executive is eligible for additional equity grants in the form
of common profits interests. Of the amount invested by each
executive, 50% was borrowed in the form of a loan. Each loan is
secured by the underlying equity interest in the respective MIV
and remains outstanding and is payable to AZ Chem Investments
Partners LP upon the earlier of (1) the date of a
distribution from a sale or initial public offering of AZ Chem
Investments Partners LP or all of its subsidiaries (including
the Company) or (2) the date that the executive ceases to
be a shareholder or limited partner in the respective MIV.
During the years ended December 31, 2009 and 2008 and the
ten month period ended December 31, 2007, AZ Chem
Investments LLC granted common profits interests to certain
participants. The grants vest over three or five years and the
resulting compensation expense is recorded by the Company and
included in selling, general and administrative expense.
These grants do not impact the common stock of the Company. As
of December 31, 2009, the common profits interests
represented approximately 3.9% of the outstanding common equity
interests in AZ Chem Investments Partners LP. Since the
Acquisition, all 1,000 shares of the Company have been
owned by AZ Chem Investments Partners LP through Lux Holdco, our
parent. The Rhône Funds, International Paper and members of
the Company’s management through their interest in MIV I
and MIV II are the sole limited partners of AZ Chem Investments
Partners LP.
We record non-cash compensation expense for the common profits
interests awards based on their grant date fair value over their
vesting period using the straight-line method. We recorded
share-based employee compensation expense of approximately
$0.1 million, $0.1 million and $0.1 million for
the years ended December 31, 2009 and 2008 and the ten
month period ended December 31, 2007, respectively. At
December 31, 2009, there was approximately
$0.3 million of unrecognized compensation cost related to
non-vested common profits interests awards expected to be
recognized over a weighted-average period of 2.70 years.
The Company has 1,000 authorized voting shares outstanding with
a 100 SEK par value that has been historically translated to
$14.25.
On November 17, 2009, the Company entered into a Settlement
Agreement with Lux Holdco and International Paper. Under the
terms of the Settlement Agreement, the Company and Lux Holdco
agreed to settle a number of claims against International Paper
for a payment to Lux Holdco. As part of the settlement Lux
Holdco received a $1.7 million cash settlement from
International Paper which resulted in a decrease to the
Company’s contributed surplus.
In a series of transactions during the fourth quarter of 2008,
Rhône Funds purchased $27.2 million face value of our
outstanding debt under our Second Lien Credit Agreement, of
which 35% of the debt was contributed to us and extinguished.
Rhône Funds’ basis in the debt of $7.4 million
was recorded as a contribution of capital and the
$1.9 million difference between the contribution and the
$9.2 million carrying value of the debt retired was
recorded as other income.
Reportable operating segments include components of an
enterprise about which separate financial information is
available that is evaluated regularly by the chief operating
decision maker,
F-40
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
which has been determined to be our chief executive officer, in
deciding how to allocate resources and in assessing performance.
We operate in two segments, North America and Europe, each of
which sells a similar portfolio of products. These segments are
our primary areas of measurement and decision-making. The North
America segment is comprised of all operations within the
American and Asian continents. The European segment is comprised
of all operations in the rest of the world. Net sales in each
segment consist of sales of our products to customers in the
following markets: adhesives, inks, roads and construction,
tires and rubber, consumer products, renewable energy, and
chemical intermediates. Our chief executive officer evaluates
our performance, in part, based upon each segment’s net
sales and net income.
The accounting policies for our reportable segments are the same
as those described in our summary of significant accounting
policies. The assets and liabilities of the Company are reported
internally in the same manner as the consolidated financial
statements. Information by operating segment as of and for the
years ended December 31, 2009 and 2008 are as follows (in
thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
|
Year Ended
|
|
Year Ended
|
|
|
|
December 31, 2009
|
|
December 31, 2008
|
|
|
|
North America
|
|
Europe
|
|
Eliminations
|
|
Total
|
|
North America
|
|
Europe
|
|
Eliminations
|
|
Total
|
|
|
|
Sales to external customers
|
|
$
|
379,102
|
|
|
$
|
388,363
|
|
|
$
|
—
|
|
|
$
|
767,465
|
|
|
$
|
492,245
|
|
|
$
|
509,743
|
|
|
$
|
—
|
|
|
$
|
1,001,988
|
|
|
Intersegment sales
|
|
|
62,622
|
|
|
|
4,222
|
|
|
|
(66,844
|
)
|
|
|
—
|
|
|
|
90,379
|
|
|
|
2,506
|
|
|
|
(92,885
|
)
|
|
|
—
|
|
|
Depreciation and amortization
|
|
|
22,430
|
|
|
|
18,184
|
|
|
|
—
|
|
|
|
40,614
|
|
|
|
22,362
|
|
|
|
11,848
|
|
|
|
—
|
|
|
|
34,210
|
|
|
Impairment loss
|
|
|
18,109
|
|
|
|
—
|
|
|
|
—
|
|
|
|
18,109
|
|
|
|
—
|
|
|
|
7,003
|
|
|
|
—
|
|
|
|
7,003
|
|
|
Interest expense, net
|
|
|
(11,438
|
)
|
|
|
(5,108
|
)
|
|
|
—
|
|
|
|
(16,546
|
)
|
|
|
(20,861
|
)
|
|
|
(8,662
|
)
|
|
|
—
|
|
|
|
(29,523
|
)
|
|
Income tax expense (benefit)
|
|
|
307
|
|
|
|
3,387
|
|
|
|
137
|
|
|
|
3,831
|
|
|
|
3,808
|
|
|
|
(7,439
|
)
|
|
|
(646
|
)
|
|
|
(4,277
|
)
|
|
Equity in earnings of affiliates, net of taxes
|
|
|
613
|
|
|
|
—
|
|
|
|
—
|
|
|
|
613
|
|
|
|
380
|
|
|
|
—
|
|
|
|
—
|
|
|
|
380
|
|
|
Net income (loss)
|
|
|
1,170
|
|
|
|
10,643
|
|
|
|
285
|
|
|
|
12,098
|
|
|
|
7,017
|
|
|
|
(32,892
|
)
|
|
|
(724
|
)
|
|
|
(26,599
|
)
|
|
Investment in affiliate
|
|
|
11,512
|
|
|
|
71
|
|
|
|
—
|
|
|
|
11,583
|
|
|
|
11,127
|
|
|
|
65
|
|
|
|
—
|
|
|
|
11,192
|
|
|
Total assets
|
|
|
314,062
|
|
|
|
303,947
|
|
|
|
—
|
|
|
|
618,009
|
|
|
|
371,063
|
|
|
|
320,021
|
|
|
|
—
|
|
|
|
691,084
|
|
|
Additions to property, plant and equipment
|
|
|
16,242
|
|
|
|
6,695
|
|
|
|
—
|
|
|
|
22,937
|
|
|
|
21,833
|
|
|
|
12,886
|
|
|
|
—
|
|
|
|
34,719
|
|
|
Software expeditures
|
|
|
9,362
|
|
|
|
4,042
|
|
|
|
—
|
|
|
|
13,404
|
|
|
|
136
|
|
|
|
6
|
|
|
|
—
|
|
|
|
142
|
|
F-41
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
Information by operating segment for the ten month period
ended December 31, 2007 and the two month period ended
February 28, 2007 are as follows (in thousands of U.S.
dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
Predecessor
|
|
|
|
|
|
March 1 through
|
|
|
January 1 through
|
|
|
|
|
|
December 31, 2007
|
|
|
February 28, 2007
|
|
|
|
|
|
North America
|
|
Europe
|
|
Eliminations
|
|
Total
|
|
|
North America
|
|
Europe
|
|
Eliminations
|
|
Total
|
|
|
|
Sales to external customers
|
|
$
|
366,292
|
|
|
$
|
357,505
|
|
|
$
|
—
|
|
|
$
|
723,797
|
|
|
|
$
|
66,434
|
|
|
$
|
65,636
|
|
|
$
|
—
|
|
|
$
|
132,070
|
|
|
|
|
|
|
Intersegment sales
|
|
|
38,878
|
|
|
|
2,653
|
|
|
|
(41,531
|
)
|
|
|
—
|
|
|
|
|
8,914
|
|
|
|
596
|
|
|
|
(9,510
|
)
|
|
|
—
|
|
|
|
|
|
|
Depreciation and amortization
|
|
|
18,813
|
|
|
|
10,000
|
|
|
|
—
|
|
|
|
28,813
|
|
|
|
|
2,669
|
|
|
|
1,753
|
|
|
|
—
|
|
|
|
4,422
|
|
|
|
|
|
|
Impairment loss
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
Interest
(expense)
income, net
|
|
|
(22,808
|
)
|
|
|
(5,967
|
)
|
|
|
—
|
|
|
|
(28,775
|
)
|
|
|
|
1
|
|
|
|
(119
|
)
|
|
|
—
|
|
|
|
(118
|
)
|
|
|
|
|
|
Income tax expense (benefit)
|
|
|
(10,135
|
)
|
|
|
1,222
|
|
|
|
—
|
|
|
|
(8,913
|
)
|
|
|
|
1,893
|
|
|
|
721
|
|
|
|
—
|
|
|
|
2,614
|
|
|
|
|
|
|
Equity in earnings of affiliates, net of taxes
|
|
|
189
|
|
|
|
—
|
|
|
|
—
|
|
|
|
189
|
|
|
|
|
84
|
|
|
|
—
|
|
|
|
—
|
|
|
|
84
|
|
|
|
|
|
|
Net income (loss)
|
|
|
(22,061
|
)
|
|
|
(4,788
|
)
|
|
|
(441
|
)
|
|
|
(27,290
|
)
|
|
|
|
(5,805
|
)
|
|
|
9,490
|
|
|
|
—
|
|
|
|
3,685
|
|
|
|
|
|
|
Additions to property, plant and equipment
|
|
|
13,147
|
|
|
|
5,101
|
|
|
|
—
|
|
|
|
18,248
|
|
|
|
|
3,585
|
|
|
|
1,013
|
|
|
|
—
|
|
|
|
4,598
|
|
|
|
|
|
|
Software expenditures
|
|
|
1,627
|
|
|
|
15
|
|
|
|
—
|
|
|
|
1,642
|
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
Our long-lived assets consist primarily of property, plant,
equipment and intangible assets, and are attributed to the
geographic location in which they are located. Long-lived assets
by geographic area as of December 31, 2009 and 2008 are as
follows (in thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
December 31,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
U.S.
|
|
$
|
195,378
|
|
|
$
|
228,710
|
|
|
Germany
|
|
|
14,044
|
|
|
|
—
|
|
|
Sweden
|
|
|
62,029
|
|
|
|
61,030
|
|
|
Great Britain
|
|
|
3,317
|
|
|
|
2,365
|
|
|
Finland
|
|
|
43,034
|
|
|
|
42,698
|
|
|
France
|
|
|
12,681
|
|
|
|
16,667
|
|
|
Netherlands
|
|
|
8,002
|
|
|
|
6,943
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
338,485
|
|
|
$
|
358,413
|
|
|
|
|
|
|
|
|
|
|
|
F-42
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
For geographic reporting, revenues are attributed to the
geographic location in which the customers’ facilities are
located. Net sales by geographic region were as follows (in
thousands of U.S. dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
|
Predecessor
|
|
|
|
|
|
|
|
|
|
|
March 1
|
|
|
|
January 1
|
|
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
|
through
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
February 28,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
2007
|
|
|
Net Sales
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
U.S.
|
|
$
|
312,249
|
|
|
$
|
397,158
|
|
|
$
|
301,535
|
|
|
|
$
|
54,869
|
|
|
Germany
|
|
|
81,443
|
|
|
|
99,009
|
|
|
|
83,534
|
|
|
|
|
15,956
|
|
|
Sweden
|
|
|
83,833
|
|
|
|
116,102
|
|
|
|
57,132
|
|
|
|
|
11,278
|
|
|
Great Britain
|
|
|
45,160
|
|
|
|
57,728
|
|
|
|
42,925
|
|
|
|
|
6,960
|
|
|
France
|
|
|
27,465
|
|
|
|
34,318
|
|
|
|
28,437
|
|
|
|
|
6,723
|
|
|
Italy
|
|
|
30,948
|
|
|
|
37,271
|
|
|
|
26,862
|
|
|
|
|
4,684
|
|
|
Mexico
|
|
|
17,662
|
|
|
|
22,238
|
|
|
|
14,411
|
|
|
|
|
2,745
|
|
|
Finland
|
|
|
13,219
|
|
|
|
26,025
|
|
|
|
12,542
|
|
|
|
|
1,921
|
|
|
Other
|
|
|
155,486
|
|
|
|
212,139
|
|
|
|
156,419
|
|
|
|
|
26,934
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
767,465
|
|
|
$
|
1,001,988
|
|
|
$
|
723,797
|
|
|
|
$
|
132,070
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
In the two month period ended February 28, 2007, we had
sales activity of 10% with Henkel AG & Co. KGaA, a
European manufacturer. The Company did not have sales greater
than 10% of total net sales with any customer in the years ended
December 31, 2009 and 2008 and the ten month period ended
December 31, 2007.
Net sales by market were as follows (in thousands of U.S.
dollars):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
|
Predecessor
|
|
|
|
|
|
|
|
|
|
|
March 1
|
|
|
|
January 1
|
|
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
|
through
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
February 28,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
2007
|
|
|
Net sales
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Adhesives
|
|
$
|
196,698
|
|
|
$
|
238,618
|
|
|
$
|
191,089
|
|
|
|
$
|
42,004
|
|
|
Inks
|
|
|
86,043
|
|
|
|
121,028
|
|
|
|
110,616
|
|
|
|
|
18,459
|
|
|
Tires and rubber
|
|
|
59,661
|
|
|
|
39,142
|
|
|
|
32,075
|
|
|
|
|
5,394
|
|
|
Roads and construction
|
|
|
43,673
|
|
|
|
53,837
|
|
|
|
28,836
|
|
|
|
|
4,098
|
|
|
Consumer products
|
|
|
40,152
|
|
|
|
31,077
|
|
|
|
26,752
|
|
|
|
|
4,207
|
|
|
Renewable energy
|
|
|
74,271
|
|
|
|
124,157
|
|
|
|
58,257
|
|
|
|
|
12,662
|
|
|
Chemical intermediates
|
|
|
266,967
|
|
|
|
394,129
|
|
|
|
276,172
|
|
|
|
|
45,246
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total net sales
|
|
$
|
767,465
|
|
|
$
|
1,001,988
|
|
|
$
|
723,797
|
|
|
|
$
|
132,070
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The Company evaluated events and transactions that occurred
during the period from December 31, 2009, the date of the
balance sheet, through May 28, 2010, the date the
consolidated
F-43
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
financial statements were available to be issued. Based on that
evaluation, the following subsequent events were identified.
On February 19, 2010, we entered into an Assignment and
Assumption agreement with AZ Chem Investments Partners LP
pursuant to which we assigned, and AZ Chem Investments
Partners LP assumed, loans to certain members of our management
team and board of directors. These loans were assigned in
exchange for total payment to us of $3.5 million,
representing payment in full of $3.0 million of outstanding
principal amount plus accrued and unpaid interest in the amount
of $0.5 million.
On February 26, 2010, Arboris, LLC filed a lawsuit against
us in the United States District Court, Middle District of
Florida regarding claims for relief under U.S. antitrust
statutes as well as a series of claims related to a tall oil
pitch supply agreement and a ground lease we entered into with
them. We believe we have strong defenses to these claims based
in part upon the advice of counsel, and we do not believe these
claims will have a material adverse affect on our business or
results of operations.
On May 26, 2010, the Compensation Committee made grants of
common profits interests to our executives who are shareholders
or limited partners in the MIVs, including each of our named
executive officers. These grants represent an additional
approximately 2.1% of the outstanding common profits interests
in AZ Chem Investments Partners LP and will be the final grants
of common profits interests to be made by the MIVs.
On May 28, 2010, we entered into amendments to both our
First Lien Credit Agreement and our Second Lien Credit
Agreement. The amendments become effective upon completion of an
initial public offering. These amendments primarily relate to
the use of proceeds from the offering and our ability to pay
dividends as a public company.
******
F-44
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
(Unaudited)
(Dollars in thousands, except share and per share amounts)
| |
|
|
|
|
|
|
|
|
|
|
|
March 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
|
|
Assets
|
|
|
|
|
|
|
|
|
|
Current assets:
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
43,057
|
|
|
$
|
47,023
|
|
|
Accounts receivable, less allowance for doubtful accounts of
$384 and $1,000, respectively
|
|
|
126,011
|
|
|
|
99,702
|
|
|
Inventory
|
|
|
86,411
|
|
|
|
89,124
|
|
|
Deferred income taxes
|
|
|
1,798
|
|
|
|
3,038
|
|
|
Prepaid expenses and other current assets
|
|
|
16,224
|
|
|
|
12,863
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current assets
|
|
|
273,501
|
|
|
|
251,750
|
|
|
Property, plant and equipment, net
|
|
|
220,337
|
|
|
|
224,945
|
|
|
Intangible assets, net
|
|
|
109,270
|
|
|
|
113,540
|
|
|
Investment in affiliate
|
|
|
11,497
|
|
|
|
11,583
|
|
|
Other assets
|
|
|
15,670
|
|
|
|
16,191
|
|
|
|
|
|
|
|
|
|
|
|
|
Total assets
|
|
$
|
630,275
|
|
|
$
|
618,009
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities and shareholder’s equity
|
|
|
|
|
|
|
|
|
|
Current liabilities:
|
|
|
|
|
|
|
|
|
|
Accounts payable
|
|
$
|
98,029
|
|
|
$
|
82,826
|
|
|
Accrued liabilities
|
|
|
39,581
|
|
|
|
42,888
|
|
|
Current portion of long-term debt
|
|
|
3,622
|
|
|
|
3,935
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current liabilities
|
|
|
141,232
|
|
|
|
129,649
|
|
|
Deferred income taxes
|
|
|
43,250
|
|
|
|
39,645
|
|
|
Long-term debt
|
|
|
324,308
|
|
|
|
330,823
|
|
|
Capital lease obligations
|
|
|
3,431
|
|
|
|
3,796
|
|
|
Other liabilities
|
|
|
16,559
|
|
|
|
16,489
|
|
|
|
|
|
|
|
|
|
|
|
|
Total liabilities
|
|
|
528,780
|
|
|
|
520,402
|
|
|
Commitments and contingencies (Note 10)
|
|
|
|
|
|
|
|
|
|
Shareholder’s equity:
|
|
|
|
|
|
|
|
|
|
Common shares, $14.25 par value — issued and
outstanding 1,000 shares
|
|
|
14
|
|
|
|
14
|
|
|
Paid-in capital
|
|
|
135,661
|
|
|
|
135,661
|
|
|
Accumulated deficit
|
|
|
(31,794
|
)
|
|
|
(41,791
|
)
|
|
Accumulated other comprehensive income
|
|
|
(2,386
|
)
|
|
|
3,723
|
|
|
|
|
|
|
|
|
|
|
|
|
Total shareholder’s equity
|
|
|
101,495
|
|
|
|
97,607
|
|
|
|
|
|
|
|
|
|
|
|
|
Total liabilities and shareholder’s equity
|
|
$
|
630,275
|
|
|
$
|
618,009
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Condensed Consolidated Financial Statements
F-45
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
(Unaudited)
(Dollars in thousands, except share and per share amounts)
| |
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
|
|
Net sales
|
|
$
|
198,051
|
|
|
$
|
177,934
|
|
|
Cost of goods sold
|
|
|
157,333
|
|
|
|
163,445
|
|
|
|
|
|
|
|
|
|
|
|
|
Gross profit
|
|
|
40,718
|
|
|
|
14,489
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating expenses (income):
|
|
|
|
|
|
|
|
|
|
Selling, general and administrative
|
|
|
25,673
|
|
|
|
15,464
|
|
|
Unrealized foreign currency exchange (gains) losses
|
|
|
(4,836
|
)
|
|
|
367
|
|
|
Restructuring and impairment
|
|
|
2,047
|
|
|
|
1,035
|
|
|
Other operating income
|
|
|
—
|
|
|
|
(2,043
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Total operating expenses (income)
|
|
|
22,884
|
|
|
|
14,823
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating income (loss)
|
|
|
17,834
|
|
|
|
(334
|
)
|
|
Interest expense, net
|
|
|
(3,870
|
)
|
|
|
(4,560
|
)
|
|
Loss on interest rate swaps, net
|
|
|
(709
|
)
|
|
|
(600
|
)
|
|
Other income
|
|
|
623
|
|
|
|
2,151
|
|
|
|
|
|
|
|
|
|
|
|
|
Income (loss) before income tax expense (benefit) and equity in
earnings of affiliates, net of taxes
|
|
|
13,878
|
|
|
|
(3,343
|
)
|
|
Income tax expense (benefit)
|
|
|
3,885
|
|
|
|
(836
|
)
|
|
Equity in earnings of affiliates net of taxes of $2 and $87,
respectively
|
|
|
4
|
|
|
|
150
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income (loss)
|
|
$
|
9,997
|
|
|
$
|
(2,357
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Earnings per share:
|
|
|
|
|
|
|
|
|
|
Basic and diluted
|
|
$
|
9,997
|
|
|
$
|
(2,357
|
)
|
|
Weighted average common shares outstanding
|
|
|
1,000
|
|
|
|
1,000
|
|
See Notes to Condensed Consolidated Financial Statements
F-46
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
(Unaudited)
(Dollars in thousands)
| |
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
|
|
Operating activities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income (loss)
|
|
$
|
9,997
|
|
|
$
|
(2,357
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Adjustments to reconcile net income (loss) to net cash provided
by operating activities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Depreciation and amortization
|
|
|
9,043
|
|
|
|
9,437
|
|
|
|
|
|
|
|
|
|
|
|
|
Change in fair value of interest rate swaps
|
|
|
(787
|
)
|
|
|
(928
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Loss (gain) on unrealized foreign exchange
|
|
|
(4,836
|
)
|
|
|
367
|
|
|
|
|
|
|
|
|
|
|
|
|
Impairment loss
|
|
|
—
|
|
|
|
1,032
|
|
|
|
|
|
|
|
|
|
|
|
|
(Gain) loss on disposal of property, plant and equipment
|
|
|
13
|
|
|
|
(671
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Proceeds from insurance recovery on facility fire
|
|
|
(696
|
)
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
Gain on acquisition
|
|
|
—
|
|
|
|
(2,151
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Amortization of debt issuance costs
|
|
|
651
|
|
|
|
630
|
|
|
|
|
|
|
|
|
|
|
|
|
Deferred income tax expense (benefit)
|
|
|
5,406
|
|
|
|
(1,286
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Provision for bad debts
|
|
|
(619
|
)
|
|
|
178
|
|
|
|
|
|
|
|
|
|
|
|
|
Equity in undistributed earnings of investment in affiliate
|
|
|
(4
|
)
|
|
|
(150
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Change in assets and liabilities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts receivable
|
|
|
(29,427
|
)
|
|
|
16,701
|
|
|
|
|
|
|
|
|
|
|
|
|
Inventory
|
|
|
855
|
|
|
|
9,027
|
|
|
|
|
|
|
|
|
|
|
|
|
Prepaid expenses and other current assets
|
|
|
(3,609
|
)
|
|
|
(3,199
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Other assets
|
|
|
(3,546
|
)
|
|
|
191
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable
|
|
|
20,256
|
|
|
|
(7,716
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Accrued liabilities
|
|
|
(69
|
)
|
|
|
(6,942
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Other liabilities
|
|
|
(121
|
)
|
|
|
(1,063
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net cash provided by operating activities
|
|
|
2,507
|
|
|
|
11,100
|
|
|
|
|
|
|
|
|
|
|
|
|
Investing activities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Acquisition of business — net of cash acquired of
$1,101 in 2009
|
|
|
—
|
|
|
|
(8,772
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Proceeds from disposals of property, plant and equipment
|
|
|
—
|
|
|
|
690
|
|
|
|
|
|
|
|
|
|
|
|
|
Proceeds from insurance recovery on facility fire
|
|
|
696
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
Additions to property, plant and equipment
|
|
|
(4,866
|
)
|
|
|
(6,677
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Capitalized software costs
|
|
|
(3,761
|
)
|
|
|
(3,268
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net cash used in investing activities
|
|
|
(7,931
|
)
|
|
|
(18,027
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Financing activities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Proceeds from long-term and short-term obligations
|
|
|
—
|
|
|
|
16,011
|
|
|
|
|
|
|
|
|
|
|
|
|
Repayments of long-term and short-term obligations
|
|
|
(686
|
)
|
|
|
(17,004
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Debt issuance costs
|
|
|
—
|
|
|
|
(163
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Repayment of capital lease obligation
|
|
|
(75
|
)
|
|
|
(64
|
)
|
|
Settlement of MIV loans
|
|
|
2,963
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash provided by (used in) financing activities
|
|
|
2,202
|
|
|
|
(1,220
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Effect of foreign exchange rate changes on cash and cash
equivalents
|
|
|
(744
|
)
|
|
|
(4,278
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Decrease in cash and cash equivalents
|
|
|
(3,966
|
)
|
|
|
(12,425
|
)
|
|
Cash and cash equivalents at beginning of period
|
|
|
47,023
|
|
|
|
34,048
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents at end of period
|
|
$
|
43,057
|
|
|
$
|
21,623
|
|
|
|
|
|
|
|
|
|
|
|
|
Supplemental cash flows information:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash paid during the period for:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest (net of amounts capitalized)
|
|
$
|
4,319
|
|
|
$
|
5,497
|
|
|
|
|
|
|
|
|
|
|
|
|
Income taxes
|
|
$
|
239
|
|
|
$
|
337
|
|
|
|
|
|
|
|
|
|
|
|
|
Noncash investing and financing activities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Purchases of property, plant and equipment in accounts payable
|
|
$
|
1,294
|
|
|
$
|
2,464
|
|
See Notes to Condensed Consolidated Financial Statements
F-47
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
(Unaudited)
(In thousands of U.S. dollars unless otherwise stated)
|
|
|
1.
|
Organization and
Basis of Presentation
|
In these notes to the unaudited condensed consolidated financial
statements, unless the context requires otherwise, references to
“Arizona Chemical”, the “Company”,
“we”, “our” or “us” refer to
Arizona Chem Sweden Holdings AB, a Swedish company, and its
consolidated subsidiaries whose financial statements are
included herein. “Rhône Capital” refers to
Rhône Capital L.L.C. and its affiliated entities, including
Rhône Capital III L.P., the general partner of certain
associated funds with investments in Arizona Chemical, and
“International Paper” refers to International Paper
Company. The accompanying unaudited condensed consolidated
financial statements are prepared in accordance with accounting
principles generally accepted in the United States of America
(“U.S. GAAP”). The condensed consolidated
financial statements are unaudited; however, in the opinion of
management, they contain all the adjustments (consisting of
those of a normal recurring nature) considered necessary to
present fairly the financial position, results of operations and
cash flows for the periods presented in conformity with
U.S. GAAP applicable to interim periods. Certain
information and footnote disclosures normally included in the
annual financial statements prepared in accordance with
U.S. GAAP have been condensed or omitted pursuant to such
SEC rules and regulations. The condensed consolidated financial
statements should be read in conjunction with the audited
consolidated financial statements of Arizona Chemical for the
year ended December 31, 2009.
We are the world’s leading supplier of pine-based chemicals
as measured by sales. We refine and further upgrade two primary
feedstocks, crude tall oil, or CTO, and crude sulfate
turpentine, or CST, both of which are wood pulping co-products,
into specialty chemicals.
The condensed consolidated financial statements include the
accounts of the Company and its subsidiaries. All intercompany
transactions have been eliminated. When the Company does not
have a controlling interest in an entity, but exerts significant
influence over the entity, the Company applies the equity method
of accounting.
|
|
|
2.
|
Recent Accounting
Pronouncements
|
In January 2010, the Financial Accounting Standards Board (FASB)
issued authoritative guidance that will require entities to make
new disclosures about recurring or nonrecurring fair-value
measurements of assets and liabilities, including (1) the
amounts of significant transfers between Level 1 and
Level 2 fair-value measurements and the reasons for the
transfers, (2) the reasons for any transfers in or out of
Level 3, and (3) information on purchases, sales,
issuances and settlements on a gross basis in the reconciliation
of recurring Level 3 fair value measurements. The FASB also
clarified existing fair-value measurement disclosure guidance
about the level of disaggregation of assets and liabilities, and
information about the valuation techniques and inputs used in
estimating Level 2 and Level 3 fair-value
measurements. Except for certain detailed Level 3
disclosures, which are effective for fiscal years beginning
after December 15, 2010 and interim periods within those
years, the new guidance became effective for the Company’s
March 31, 2010 interim financial statements. The adoption
of this disclosure-only guidance is included in
Note 3 — Fair Value of Financial Instruments and
did not have an impact on the Company’s condensed
consolidated financial statements.
In June 2009, the FASB issued authoritative guidance to
eliminate the exception to consolidate a qualifying
special-purpose entity, change the approach to determining the
primary beneficiary of a variable interest entity and require
companies to more frequently reassess whether they must
consolidate variable interest entities. Under the new guidance,
the primary beneficiary of a variable interest entity is
identified qualitatively as the enterprise that has both
(a) the power to direct the
F-48
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONDENSED CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
(Unaudited)
activities of a variable interest entity that most significantly
impact the entity’s economic performance, and (b) the
obligation to absorb losses of the entity that could potentially
be significant to the variable interest entity or the right to
receive benefits from the entity that could potentially be
significant to the variable interest entity. This standard was
effective for financial statements for interim or annual
reporting periods beginning on or after January 1, 2010.
This guidance did not have a material impact on our condensed
consolidated financial statements.
In May 2009, the FASB issued new guidance related to the
disclosure of subsequent events. This standard is intended to
establish general standards of accounting for and disclosure of
events that occur after the balance sheet date but before
financial statements are issued or are available to be issued.
Specifically, this standard sets forth the period after the
balance sheet date during which management of a reporting entity
should evaluate events or transactions that may occur for
potential recognition or disclosure in the financial statements,
the circumstances under which an entity should recognize events
or transactions occurring after the balance sheet date in its
financial statements, and the disclosures that an entity should
make about events or transactions that occurred after the
balance sheet date. This guidance is effective for fiscal years
and interim periods ended after June 15, 2009. The
additional disclosures have been included in Note 12
“Subsequent Events.”
In April 2009, the FASB issued authoritative guidance that
principally requires publicly traded companies to provide
disclosures about fair value of financial instruments in interim
financial information. The adoption of this disclosure-only
guidance for the Company’s March 31, 2010 interim
consolidated financial statements is included in
Note 3 — Fair Value of Financial Instruments and
did not have an impact on the Company’s condensed
consolidated financial statements.
|
|
|
3.
|
Fair Value of
Financial Instruments
|
The estimated fair values of the Company’s financial
instruments were as follows:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 31, 2010
|
|
|
December 31, 2009
|
|
|
|
|
Carrying
|
|
|
Fair
|
|
|
Carrying
|
|
|
Fair
|
|
|
Asset (Liability)
|
|
Amount
|
|
|
Value
|
|
|
Amount
|
|
|
Value
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
43,057
|
|
|
$
|
43,057
|
|
|
$
|
47,023
|
|
|
$
|
47,023
|
|
|
Long-term debt
|
|
$
|
(327,930
|
)
|
|
$
|
(318,619
|
)
|
|
$
|
(334,758
|
)
|
|
$
|
(319,868
|
)
|
|
Interest rate swaps
|
|
$
|
(1,370
|
)
|
|
$
|
(1,370
|
)
|
|
$
|
(2,879
|
)
|
|
$
|
(2,879
|
)
|
|
Interest rate cap
|
|
$
|
322
|
|
|
$
|
322
|
|
|
$
|
1,028
|
|
|
$
|
1,028
|
|
The Company enters into interest rate swap agreements to
mitigate the interest rate risk associated with the
$375 million credit facilities bearing variable interest
rates. See Note 8 “Debt” for further information
on the credit facilities including the outstanding principal
balance as of the balance sheet dates.
As of December 31, 2008, the Company had three interest
rate swap agreements outstanding. One of these agreements
matured in February 2009. The remaining two agreements matured
on February 28, 2010. The Company entered into a new
interest rate swap agreement that became effective on
February 26, 2010 and will mature on February 28,
2012. The swap has a notional value of €53 million and
a three-month Euribor rate fixed at 2.125%.
During the second quarter of 2009, the Company purchased a
three-month U.S. dollar LIBOR interest rate cap for
$1.1 million. The cap became effective on February 26,
2010 and will mature on February 28, 2012. The strike price
of the cap is 3% on a notional amount of $175 million. If
the three-month U.S. dollar LIBOR rate exceeds 3% during
the term of the cap, we will receive a cash
F-49
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONDENSED CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
(Unaudited)
payment for the difference between the current rate and 3%
applied to the notional amount of $175 million.
The following summarizes the Condensed Consolidated Balance
Sheet location of our derivative instruments:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Asset Derivatives
|
|
|
|
|
March 31, 2010
|
|
|
December 31, 2009
|
|
|
|
|
Balance Sheet
|
|
|
Fair
|
|
|
Balance Sheet
|
|
|
Fair
|
|
|
|
|
Location
|
|
|
Value
|
|
|
Location
|
|
|
Value
|
|
|
|
|
Interest rate cap
|
|
|
Other assets
|
|
|
$
|
322
|
|
|
|
Other assets
|
|
|
$
|
1,028
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
|
|
|
$
|
322
|
|
|
|
|
|
|
$
|
1,028
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Liability Derivatives
|
|
|
|
|
March 31, 2010
|
|
|
December 31, 2009
|
|
|
|
|
Balance Sheet
|
|
|
Fair
|
|
|
Balance Sheet
|
|
|
Fair
|
|
|
|
|
Location
|
|
|
Value
|
|
|
Location
|
|
|
Value
|
|
|
|
|
Interest rate swaps
|
|
|
Accrued liabilities
|
|
|
$
|
—
|
|
|
|
Accrued liabilities
|
|
|
$
|
2,303
|
|
|
Interest rate swaps
|
|
|
Other liabilities
|
|
|
|
1,370
|
|
|
|
Other liabilities
|
|
|
|
576
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
|
|
|
$
|
1,370
|
|
|
|
|
|
|
$
|
2,879
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The following summarizes the Condensed Consolidated Statements
of Operations location of our derivative instruments:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Amount of Gain (Loss) Recognized in Income
|
|
Derivatives Not
|
|
Location of Gain
|
|
|
Three Months Ended
|
|
|
Three Months Ended
|
|
Designated as Hedging
|
|
(Loss) Recognized in
|
|
|
March 31,
|
|
|
March 31,
|
|
|
Instruments
|
|
Income
|
|
|
2010
|
|
|
2009
|
|
|
|
|
Interest rate swaps
|
|
|
Loss on interest rate swaps, net
|
|
|
$
|
1,428
|
|
|
$
|
881
|
|
|
Interest rate cap
|
|
|
Loss on interest rate swaps, net
|
|
|
|
(706
|
)
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
|
|
|
$
|
722
|
|
|
$
|
881
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net settlements of $(1.5) million and $(1.5) million
for the three months ended March 31, 2010 and 2009,
respectively, on interest rate swap agreements are also included
in the loss on interest rate swaps, net.
The methods and assumptions used to estimate the fair value of
each class of financial instruments are set forth below:
Cash and Cash Equivalents — The carrying
amounts approximate fair value because of the relatively short
time between the origination of the instrument and its expected
realization. Foreign currency denominated cash and cash
equivalents are translated into U.S. dollars using exchange
rates in effect at the balance sheet date.
Long-Term Debt — The fair value of
long-term debt is estimated based on borrowing rates currently
available to the Company for loans with similar terms and
maturities and discounted back to the present value. The Company
obtains fair value measurements on its long-term debt
obligations from third party providers. Significant factors
evaluated include changes in margin on its various loans and its
ability to make future debt payments.
Derivative Financial Instruments — The
fair value of interest rate swaps and interest rate caps (used
for purposes other than trading) represents the amount the
Company would receive
F-50
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONDENSED CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
(Unaudited)
or pay to terminate the agreements at the reporting date, taking
into consideration the difference between the contract rate of
interest and rates currently quoted for agreements of similar
terms and maturities. The fair value measurement is estimated
based on a discounted cash flow model using a dealer quoted
interest rate. The fair value measurement of the derivative
instruments in a liability position also considers the current
credit-worthiness of the Company and its counterparties. The
Company is exposed to credit related losses in the event of
nonperformance by counterparties to these financial instruments.
However, counterparties to these agreements are major financial
institutions, and the risk of loss due to nonperformance is
considered by management to be minimal.
To assess the inputs used to develop those measurements, a
hierarchy for ranking the quality and reliability of the
information used to determine fair value has been established
that applies to all assets and liabilities that are being
measured and reported on a fair value basis and requires their
classification and disclosure in one of the following three
categories:
Level 1 — Quoted market prices in
active markets for identical assets or liabilities.
Level 2 — Observable market based
inputs or unobservable inputs that are corroborated by market
data.
Level 3 — Unobservable inputs that
are not corroborated by market data.
The following summarizes the valuation of our financial
instruments reported at fair value by the above pricing levels
as of the valuation date listed:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Derivative
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Financial
|
|
|
Fair Value Measurements Using
|
|
|
|
|
Instruments
|
|
|
Level 1
|
|
|
Level 2
|
|
|
Level 3
|
|
|
|
|
As of March 31, 2010:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest rate swap
|
|
$
|
(1,370
|
)
|
|
$
|
—
|
|
|
$
|
(1,370
|
)
|
|
$
|
—
|
|
|
Interest rate cap
|
|
$
|
322
|
|
|
$
|
—
|
|
|
$
|
322
|
|
|
$
|
—
|
|
|
As of December 31, 2009:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest rate swaps
|
|
$
|
(2,879
|
)
|
|
$
|
—
|
|
|
$
|
(2,879
|
)
|
|
$
|
—
|
|
|
Interest rate cap
|
|
$
|
1,028
|
|
|
$
|
—
|
|
|
$
|
1,028
|
|
|
$
|
—
|
|
Inventory by major category includes the following:
| |
|
|
|
|
|
|
|
|
|
|
|
March 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
|
|
Raw materials
|
|
$
|
32,316
|
|
|
$
|
35,352
|
|
|
Finished goods
|
|
|
54,095
|
|
|
|
53,772
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
86,411
|
|
|
$
|
89,124
|
|
|
|
|
|
|
|
|
|
|
|
F-51
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONDENSED CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
(Unaudited)
5. Property,
Plant and Equipment, Net
Property, plant and equipment, net consists of the following:
| |
|
|
|
|
|
|
|
|
|
|
|
March 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
|
|
Land
|
|
$
|
15,954
|
|
|
$
|
14,392
|
|
|
Buildings
|
|
|
38,845
|
|
|
|
42,188
|
|
|
Machinery and equipment
|
|
|
201,781
|
|
|
|
203,769
|
|
|
Construction in progress
|
|
|
10,264
|
|
|
|
7,693
|
|
|
Software
|
|
|
25,493
|
|
|
|
24,877
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
292,337
|
|
|
|
292,919
|
|
|
Less accumulated depreciation
|
|
|
(72,000
|
)
|
|
|
(67,974
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Property, plant and equipment, net
|
|
$
|
220,337
|
|
|
$
|
224,945
|
|
|
|
|
|
|
|
|
|
|
|
The Company recognized depreciation expense of $6.6 million
and $6.9 million for the three months ended March 31,
2010 and 2009, respectively, which is reflected in cost of goods
sold and selling, general and administrative expenses in the
accompanying unaudited Condensed Consolidated Statements of
Operations.
|
|
|
6.
|
Intangible
Assets, Net
|
Intangible assets, net consist of the following:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 31, 2010
|
|
|
December 31, 2009
|
|
|
|
|
Gross
|
|
|
|
|
|
Net
|
|
|
Gross
|
|
|
|
|
|
Net
|
|
|
|
|
Carrying
|
|
|
Accumulated
|
|
|
Carrying
|
|
|
Carrying
|
|
|
Accumulated
|
|
|
Carrying
|
|
|
|
|
Amount
|
|
|
Amortization
|
|
|
Amount
|
|
|
Amount
|
|
|
Amortization
|
|
|
Amount
|
|
|
|
|
Trade names
|
|
$
|
24,381
|
|
|
$
|
—
|
|
|
$
|
24,381
|
|
|
$
|
24,910
|
|
|
$
|
—
|
|
|
$
|
24,910
|
|
|
Customer relationships
|
|
|
43,344
|
|
|
|
(6,289
|
)
|
|
|
37,055
|
|
|
|
44,614
|
|
|
|
(5,602
|
)
|
|
|
39,012
|
|
|
Supply agreements
|
|
|
41,378
|
|
|
|
(13,640
|
)
|
|
|
27,738
|
|
|
|
41,379
|
|
|
|
(12,796
|
)
|
|
|
28,583
|
|
|
Core/developed technology
|
|
|
24,830
|
|
|
|
(5,445
|
)
|
|
|
19,385
|
|
|
|
25,388
|
|
|
|
(5,127
|
)
|
|
|
20,261
|
|
|
Favorable leaseholds
|
|
|
772
|
|
|
|
(61
|
)
|
|
|
711
|
|
|
|
825
|
|
|
|
(59
|
)
|
|
|
766
|
|
|
Internally developed software
|
|
|
167
|
|
|
|
(167
|
)
|
|
|
—
|
|
|
|
170
|
|
|
|
(162
|
)
|
|
|
8
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total intangible assets
|
|
$
|
134,872
|
|
|
$
|
(25,602
|
)
|
|
$
|
109,270
|
|
|
$
|
137,286
|
|
|
$
|
(23,746
|
)
|
|
$
|
113,540
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
We recognized amortization expense for the intangible assets
included above, intangible assets associated with our investment
in Arboris, LLC, and software included in property, plant and
equipment of $2.4 million and $2.6 million for the
three months ended March 31, 2010 and 2009, respectively.
F-52
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONDENSED CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
(Unaudited)
The estimated aggregate amortization from intangible assets for
each of the next five years is as follows (in thousands of
U.S. dollars):
| |
|
|
|
|
|
|
|
Amortization
|
|
Years Ending December
31
|
|
Expense
|
|
|
|
2010
|
|
$
|
8,466
|
|
|
2011
|
|
$
|
8,457
|
|
|
2012
|
|
$
|
8,457
|
|
|
2013
|
|
$
|
8,457
|
|
|
2014
|
|
$
|
6,672
|
|
|
|
|
7.
|
Restructuring and
Impairment
|
In 2007, we initiated a comprehensive cost reduction program
that drives us to continuously reduce our fixed costs, improve
our processes and the way we run our business, and better serve
our customers. A key part of our program has been the
optimization of our manufacturing footprint. This resulted in
the recognition of restructuring and impairment charges in the
Condensed Consolidated Statement of Operations.
The following table summarizes the activity and remaining
liabilities included in accrued liabilities that are associated
with the restructuring charges:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Termination
|
|
|
|
|
|
|
|
|
|
|
Benefits to Employees
|
|
|
Other Qualified Costs
|
|
|
|
|
|
|
|
North America
|
|
|
Europe
|
|
|
North America
|
|
|
Europe
|
|
|
Total
|
|
|
|
|
Balance — December 31, 2009
|
|
$
|
362
|
|
|
$
|
832
|
|
|
$
|
798
|
|
|
$
|
18
|
|
|
$
|
2,010
|
|
|
Costs accrued
|
|
|
97
|
|
|
|
1,950
|
|
|
|
—
|
|
|
|
—
|
|
|
|
2,047
|
|
|
Payments
|
|
|
(97
|
)
|
|
|
(151
|
)
|
|
|
(501
|
)
|
|
|
—
|
|
|
|
(749
|
)
|
|
Currency exchange
|
|
|
—
|
|
|
|
(74
|
)
|
|
|
—
|
|
|
|
(1
|
)
|
|
|
(75
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance — March 31, 2010
|
|
$
|
362
|
|
|
$
|
2,557
|
|
|
$
|
297
|
|
|
$
|
17
|
|
|
$
|
3,233
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Restructuring costs expensed for the three month period ended
March 31, 2010 were $0.1 million and $2.0 million
in North America and Europe, respectively. These costs primarily
relate to employee severance expenses of $1.2 million for
our Sandarne, Sweden manufacturing facility and
$0.7 million for our Niort, France manufacturing facility
as a result of the realignment of our business in Europe. In the
past, each of our European manufacturing facilities operated as
stand alone companies, transacting business directly with our
customers and suppliers. The new operating structure will
provide a single face to our European customers and suppliers,
streamlining our processes, realigning our production
capabilities to market demands and making it easier for them to
do business with us. We expect to incur approximately
$0.4 million additional restructuring and impairment costs
in subsequent quarters as this realignment is planned in a
phased approach.
Restructuring costs of $3 thousand were incurred for the
three month period ended March 31, 2009 related to our
Dover, Ohio manufacturing facility.
No impairment costs were incurred during the three month period
ended March 31, 2010. Impairment costs incurred for the
three month period ended March 31, 2009 was
$1.0 million, related to the impairment of machinery and
equipment.
F-53
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONDENSED CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
(Unaudited)
Debt and credit agreements consist of the following:
| |
|
|
|
|
|
|
|
|
|
|
|
March 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
|
|
First lien term loan
|
|
$
|
211,895
|
|
|
$
|
218,543
|
|
|
Second lien term loan
|
|
|
115,498
|
|
|
|
115,498
|
|
|
Revolving credit facility
|
|
|
—
|
|
|
|
—
|
|
|
Bank loan
|
|
|
537
|
|
|
|
717
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
327,930
|
|
|
|
334,758
|
|
|
Less current portion
|
|
|
(3,622
|
)
|
|
|
(3,935
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Long-term debt
|
|
$
|
324,308
|
|
|
$
|
330,823
|
|
|
|
|
|
|
|
|
|
|
|
The first lien term loan is divided into two loans, one
denominated in U.S. dollars and one denominated in Euros.
The outstanding principal balance as of March 31, 2010 and
December 31, 2009 was $212.0 million and
$218.5 million, respectively. Interest is payable monthly
or quarterly at LIBOR/Euribor or prime rate, plus a 2.00% margin
for U.S. dollar and 2.25% for Euro-denominated debt. The
interest rates on the U.S. dollar and Euro-denominated debt
was 2.25% and 2.91%, respectively, at March 31, 2010 and
2.24% and 2.82%, respectively, at December 31, 2009. The
first lien term loan is due February 28, 2013.
The second lien term loan has interest payments due based on the
interest period selected (one, two or three months) at LIBOR or
prime rate, plus a 5.50% margin and 4.50% margin, respectively.
The interest rate was 5.75% and 5.74% at March 31, 2010 and
December 31, 2009, respectively. The second lien term loan
is due February 28, 2014 and is collateralized by a
second-priority lien on substantially all assets and equity
interests of the Company’s U.S. subsidiaries as
defined in the Second Lien Credit and Guaranty Agreement dated
February 28, 2007. U.S. assets totaled approximately
$331.6 million and $328.7 million as of March 31,
2010 and December 31, 2009, respectively.
The Company has a revolving credit facility with availability of
$60.0 million that can be borrowed in U.S. dollars or
Euros. The Company also has the ability to issue letters of
credit based on availability under the revolving credit
facility. Interest is payable monthly or quarterly at
LIBOR/Euribor or prime rate, plus an applicable margin based on
the Company’s leverage ratio. There was no outstanding
balance at either March 31, 2010 or December 31, 2009.
The outstanding balance of the revolving credit facility is due
February 28, 2012. The commitment fee related to the
revolving credit facility was $0.1 million and
$30 thousand for the three months ended March 31, 2010
and 2009, respectively.
As part of our February 12, 2009 acquisition of Abieta
Chemie GmbH, a company located in Gersthofen, Germany that
specializes in natural resin products, we assumed an outstanding
bank loan of €4.5 million ($6.0 million) at a
fixed interest rate of 4.80%. As of March 31, 2010 and
December 31, 2009, $0.5 million and $0.7 million,
respectively, was outstanding after repayments made to date.
The first and second lien term loans and the revolving credit
facility contain certain financial covenants. The financial
covenants require that the Company maintain (i) a certain
interest coverage ratio, (ii) a certain leverage ratio and
(iii) a maximum limit for capital expenditures in any year.
In addition, certain covenants substantially restrict the
Company’s ability to incur additional indebtedness, create
liens, make certain investments, sell assets or pay dividends.
The Company’s obligations under
F-54
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONDENSED CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
(Unaudited)
these agreements may be accelerated on certain events of
default. As of March 31, 2010 and December 31, 2009,
the Company was in compliance with the covenants.
Rhône Capital holds $17.6 million of the
Company’s outstanding second lien debt as of March 31,
2010. Interest paid to Rhône Capital was $0.2 million
and $0.3 million for the three months ended March 31,
2010 and 2009, respectively.
(a) U.S. Defined Benefit Plan
— The Company sponsors a noncontributory
defined benefit pension plan in the U.S.
Net periodic pension expense for the Company’s
U.S. defined benefit pension plan was as follows:
| |
|
|
|
|
|
|
|
|
|
|
|
Three Months
|
|
|
Three Months
|
|
|
|
|
Ended
|
|
|
Ended
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
|
|
Service cost
|
|
$
|
204
|
|
|
$
|
212
|
|
|
Interest cost
|
|
|
63
|
|
|
|
22
|
|
|
Expected return on plan assets
|
|
|
(53
|
)
|
|
|
—
|
|
|
Amortization of prior service cost
|
|
|
27
|
|
|
|
28
|
|
|
|
|
|
|
|
|
|
|
|
|
Net periodic benefit cost
|
|
$
|
241
|
|
|
$
|
262
|
|
|
|
|
|
|
|
|
|
|
|
The U.S. defined benefit pension plan obligations and
assets are measured annually at December 31.
The Company contributed $0.2 million and $0.1 million
in the three months ended March 31, 2010 and 2009,
respectively, and expects to contribute $0.8 million to the
defined benefit pension plan in the remainder of 2010.
(b) Non U.S. Defined Benefit Plans
— The Company sponsors defined benefit pension
and retirement plans in certain foreign subsidiaries. Generally,
the Company’s non-U.S. defined benefit pension plans
are funded using the projected benefit as a target, in countries
where funding of benefit plans is required.
Net periodic pension expense for the Company’s
non-U.S. plans was as follows:
| |
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended
|
|
|
Three Months Ended
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
|
|
Service cost
|
|
$
|
323
|
|
|
$
|
274
|
|
|
Interest cost
|
|
|
768
|
|
|
|
616
|
|
|
Expected return on plan assets
|
|
|
(648
|
)
|
|
|
(556
|
)
|
|
Amortization of prior service cost
|
|
|
1
|
|
|
|
—
|
|
|
Amortization of net (gain) loss
|
|
|
(26
|
)
|
|
|
(37
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net periodic benefit cost
|
|
$
|
418
|
|
|
$
|
297
|
|
|
|
|
|
|
|
|
|
|
|
The non U.S. pension plans obligations and assets are
measured annually at December 31.
F-55
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONDENSED CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
(Unaudited)
The Company contributed $0.5 million and $0.3 million
in three months ended March 31, 2010 and 2009,
respectively, and expects to contribute $0.3 million to the
pension plans in the remainder of 2010.
|
|
|
(c)
|
401(k) Savings
Plan (U.S.)
|
The Company has a defined contribution Employee Savings Plan for
all U.S.-based employees, the Arizona Chemical Savings Plan. The
savings plan is a tax-qualified 401(k) plan, and employees’
contributions are matched by the Company on the following basis:
70 cents on the dollar for the first four percent of the
employee’s contribution and 50 cents on the dollar for up
to an additional four percent.
As a means to further enhance retirement savings for
U.S.-based
employees in light of the absence of a defined benefit pension
plan, the Company maintains an annual profit sharing award plan.
This award comes in the form of a discretionary gift
contribution made by the Company directly to each
U.S.-based
salaried employee’s savings plan account based on the
Company’s performance. The award is intended to be variable
in nature and it could be less or more than four percent
depending upon the performance of the business. All employees
receive the same percentage award.
The Company’s contributions under these plans amounted to
approximately $0.6 million and $0.5 million for the
three months ended March 31, 2010 and 2009, respectively.
The Company expects to contribute $1.1 million in the
remainder of 2010.
|
|
|
(d)
|
Long-term
Service Award Plan
|
The Company maintains a Long-Term Service Awards program, a
deferred compensation plan, for qualified employees in our
German operations. Under the German plan, qualified employees
receive a service gratuity (“Jubilee”) payment once
they have reached a certain number of years of service.
The Company’s contributions under this plan amounted to
approximately $1 thousand for the three months ended
March 31, 2010. The Company did not make contributions
under this plan in the three months ended March 31, 2009. The
Company expects to contribute $14 thousand in the remainder of
2010.
|
|
|
(e)
|
Early
Retirement and Post-employment Programs
|
In Germany, Altersteilzeit (“ATZ”) is an early
retirement program established by law, and is designed to create
an incentive for employees, within a certain age group, to
transition from (full or part-time) employment into retirement
before their legal retirement age.
The Company’s contributions under this plan amounted to
approximately $39 thousand for the three months ended
March 31, 2010. The Company did not make contributions
under this plan in the three months ended March 31, 2009. The
Company expects to contribute $156 thousand in the remainder of
2010.
|
|
|
(f)
|
Collective
Bargaining Agreements
|
As of March 31, 2010, approximately 54% of our
671 employees in the United States were unionized and are
covered by collective bargaining agreements. In Europe, nearly
all of our employees are represented by local workers’
councils
and/or
unions.
F-56
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONDENSED CONSOLIDATED FINANCIAL
STATEMENTS — (Continued)
(Unaudited)
|
|
|
10.
|
Commitments and
Contingencies
|
Operating Leases — The Company leases
administrative, manufacturing and warehousing facilities, and
machinery and other equipment under non-cancelable leases that
expire at various dates through 2015.
Purchase Obligations — The Company has
entered into unconditional purchase agreements with terms of
more than one year that entail fixed payments for periods
lasting up to 2019.
Litigation — The Company is a party to
various lawsuits, claims, and contingent liabilities arising
from the conduct of its business; however, in the opinion of
management, based on the advice of counsel, they are not
expected to have a material adverse effect on the consolidated
financial statements of the Company.
On February 26, 2010, Arboris, LLC filed a lawsuit against
us in the United States District Court, Middle District of
Florida regarding claims for relief under U.S. antitrust
statutes as well as a series of claims related to a tall oil
pitch supply agreement and a ground lease we entered into with
them. We believe we have strong defenses to these claims based
in part upon the advice of counsel, and we do not believe these
claims will have a material adverse affect on our business or
consolidated financial statements.
Forward Contracts — We typically enter
into natural gas purchase contracts that extend no more than
twelve months into the future for between 50% - 70% of our
estimated natural gas consumption in our North American
manufacturing facilities.
Environmental Costs and Obligations — We
accrue costs associated with environmental obligations, such as
remediation or closure costs, when such costs are probable and
reasonably estimable. We adjust such accruals as further
information develops or circumstances change. We discount costs
of future expenditures for environmental obligations to their
present value when the expected cash flows are reliably
determinable. Legal costs incurred in connection with loss
contingencies are expensed as incurred. Indemnification of
certain environmental remediation costs of $3.2 million and
$3.3 million as of March 31, 2010 and
December 31, 2009, respectively, from third parties, which
are probable of realization, are separately recorded as assets,
and are not offset against the related environmental liability
as of March 31, 2010 and December 31, 2009. As of
March 31, 2010 and December 31, 2009, accruals related
to environmental obligations totaled $4.0 million and
$4.1 million, respectively.
Reportable operating segments include components of an
enterprise about which separate financial information is
available that is evaluated regularly by the chief operating
decision maker, which has been determined to be our chief
executive officer, in deciding how to allocate resources and in
assessing performance.
We operate in two segments, North America and Europe, each of
which sells a similar portfolio of products. These segments are
our primary areas of measurement and decision making. The North
America segment is comprised of all operations within the
American and Asian continents. The European segment is comprised
of all operations in the rest of the world. Net sales in each
segment consist of sales of our products to customers in the
following markets: adhesives, inks, roads and construction,
tires and rubber, consumer products, renewable energy, and
chemical intermediates. Our chief executive officer evaluates
our performance, in part, based upon each segment’s net
sales and net income.
F-57
ARIZONA CHEM
SWEDEN HOLDINGS AB AND SUBSIDIARIES
NOTES TO
CONDENSED CONSOLIDATED FINANCIAL
STATEMENTS — (Concluded)
(Unaudited)
The assets and liabilities of the Company are reported
internally in the same manner as the condensed consolidated
financial statements. Information by operating segment is as
follows:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended
|
|
Three Months Ended
|
|
|
|
March 31, 2010
|
|
March 31, 2009
|
|
|
|
North America
|
|
Europe
|
|
Eliminations
|
|
Total
|
|
North America
|
|
Europe
|
|
Eliminations
|
|
Total
|
|
|
|
Sales to external customers
|
|
$
|
96,333
|
|
|
$
|
101,718
|
|
|
$
|
—
|
|
|
$
|
198,051
|
|
|
$
|
85,555
|
|
|
$
|
92,379
|
|
|
$
|
—
|
|
|
$
|
177,934
|
|
|
Intersegment sales
|
|
|
18,227
|
|
|
|
65,124
|
|
|
|
(83,351
|
)
|
|
|
—
|
|
|
|
14,215
|
|
|
|
1,338
|
|
|
|
(15,553
|
)
|
|
|
—
|
|
|
Depreciation and amortization
|
|
|
4,795
|
|
|
|
4,248
|
|
|
|
—
|
|
|
|
9,043
|
|
|
|
5,459
|
|
|
|
3,978
|
|
|
|
—
|
|
|
|
9,437
|
|
|
Impairment loss
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,032
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,032
|
|
|
Interest expense, net
|
|
|
(2,780
|
)
|
|
|
(1,090
|
)
|
|
|
—
|
|
|
|
(3,870
|
)
|
|
|
(2,834
|
)
|
|
|
(1,726
|
)
|
|
|
—
|
|
|
|
(4,560
|
)
|
|
Income tax expense (benefit)
|
|
|
4,554
|
|
|
|
(669
|
)
|
|
|
—
|
|
|
|
3,885
|
|
|
|
(1,156
|
)
|
|
|
203
|
|
|
|
117
|
|
|
|
(836
|
)
|
|
Equity in earnings of affiliates, net of taxes
|
|
|
4
|
|
|
|
—
|
|
|
|
—
|
|
|
|
4
|
|
|
|
150
|
|
|
|
—
|
|
|
|
—
|
|
|
|
150
|
|
|
Net income (loss)
|
|
|
9,451
|
|
|
|
546
|
|
|
|
—
|
|
|
|
9,997
|
|
|
|
(3,212
|
)
|
|
|
618
|
|
|
|
237
|
|
|
|
(2,357
|
)
|
|
Additions to property, plant and equipment
|
|
|
(1,885
|
)
|
|
|
(2,981
|
)
|
|
|
—
|
|
|
|
(4,866
|
)
|
|
|
(4,863
|
)
|
|
|
(1,814
|
)
|
|
|
—
|
|
|
|
(6,677
|
)
|
|
Software expenditures
|
|
|
(2,088
|
)
|
|
|
(1,673
|
)
|
|
|
—
|
|
|
|
(3,761
|
)
|
|
|
(2,685
|
)
|
|
|
(583
|
)
|
|
|
—
|
|
|
|
(3,268
|
)
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 31, 2010
|
|
December 31, 2009
|
|
|
|
North America
|
|
Europe
|
|
Eliminations
|
|
Total
|
|
North America
|
|
Europe
|
|
Eliminations
|
|
Total
|
|
|
|
Investment in affiliate
|
|
$
|
11,427
|
|
|
$
|
70
|
|
|
$
|
—
|
|
|
$
|
11,497
|
|
|
$
|
11,512
|
|
|
$
|
71
|
|
|
$
|
—
|
|
|
$
|
11,583
|
|
|
Total assets
|
|
|
331,644
|
|
|
|
298,631
|
|
|
|
—
|
|
|
|
630,275
|
|
|
|
314,062
|
|
|
|
303,947
|
|
|
|
—
|
|
|
|
618,009
|
|
The Company evaluated events and transactions that occurred
during the period from March 31, 2010, the date of the
balance sheet, through May 28, 2010, the date the unaudited
condensed consolidated financial statements were available to be
issued. Based on that evaluation, the following subsequent
events were identified:
On May 28, 2010, the Compensation Committee made grants of
common profits interests to our executives who are shareholders
or limited partners in the MIVs, including each of our named
executive officers. These grants represent an additional
approximately 2.1% of the outstanding common profits interests
in AZ Chem Investments Partners LP and will be the final grants
of common profits interests to be made by the MIVs.
On May 27, 2010, we entered into amendments to both our
First Lien Credit Agreement and our Second Lien Credit
Agreement. The amendments become effective upon completion of an
initial public offering. These amendments primarily relate to
the use of proceeds from the offering and our ability to pay
dividends as a public company.
******
F-58
PART II
INFORMATION NOT
REQUIRED IN PROSPECTUS
|
|
|
Item 13.
|
Other Expenses
of Issuance and Distribution
|
The following table sets forth the fees and expenses, other than
underwriting discounts and commissions, payable in connection
with the registration of the common shares hereunder. All
amounts are estimates except the Securities and Exchange
Commission registration fee, the Financial Industry Regulatory
Authority, Inc. filing fee and the New York Stock Exchange
listing fee.
| |
|
|
|
|
|
Securities and Exchange Commission registration fee
|
|
$
|
8,912.50
|
|
|
Financial Industry Regulatory Authority, Inc. filing fee
|
|
$
|
13,000.00
|
|
|
New York Stock Exchange listing fee
|
|
|
|
|
|
Printing and engraving expenses
|
|
|
|
|
|
Legal fees and expenses
|
|
|
|
|
|
Accounting fees and expenses
|
|
|
|
|
|
Transfer agent and registrar fees and expenses
|
|
|
|
|
|
Blue Sky fees and expenses
|
|
|
|
|
|
Miscellaneous
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Item 14.
|
Indemnification
of Directors and Officers
|
Section 98 of the Companies Act provides generally that a
Bermuda company may indemnify its directors, officers and
auditors against any liability which by virtue of any rule of
law would otherwise be imposed on them in respect of any
negligence, default, breach of duty or breach of trust, except
in cases where such liability arises from fraud or dishonesty of
which such director, officer or auditor may be guilty in
relation to the company. Section 98 further provides that a
Bermuda company may indemnify its directors, officers and
auditors against any liability incurred by them in defending any
proceedings, whether civil or criminal, in which judgment is
awarded in their favor or in which they are acquitted or granted
relief by the Supreme Court of Bermuda pursuant to
section 281 of the Companies Act.
We have adopted provisions in our bye-laws that provide that we
shall indemnify our officers and directors in respect of their
actions and omissions, except in respect of their fraud or
dishonesty. Our bye-laws provide that the shareholders waive all
claims or rights of action that they might have, individually or
in right of the company, against any of the company’s
directors or officers for any act or failure to act in the
performance of such director’s or officer’s duties,
except in respect of any fraud or dishonesty of such director or
officer. Section 98A of the Companies Act permits us to
purchase and maintain insurance for the benefit of any officer
or director in respect of any loss or liability attaching to him
in respect of any negligence, default, breach of duty or breach
of trust, whether or not we may otherwise indemnify such officer
or director.
We are in the process of obtaining directors’ and
officers’ insurance for some of our directors, officers,
affiliates, partners or employees for liabilities relating to
the performance of their duties. Such indemnification shall not
extend to any matter which would render it void pursuant to the
Companies Act.
At present, there is no pending material litigation or
proceeding involving any of our officers or directors where
indemnification will be required or permitted. We are not aware
of any threatened material litigation or proceeding which may
result in a claim for indemnification of an officer or director.
II-1
|
|
|
Item 15.
|
Recent Sales
of Unregistered Securities
|
On February 12, 2010, in connection with its incorporation,
the registrant issued 100 of its common shares to Arizona
Chemical Luxembourg Holdings S.à r.l. for $1.00 in cash.
These shares were offered and sold in reliance on
Section 4(2) of the Securities Act as the offering and sale
of the common shares did not involve a public offering.
|
|
|
Item 16.
|
Exhibits and
Financial Statement Schedules
|
(a) Exhibits
See the Exhibit Index immediately following the signature
page hereto, which is incorporated by reference as if fully set
forth herein.
(b) Financial Statement Schedules
ARIZONA CHEM
SWEDEN HOLDINGS AB
SCHEDULE I
CONDENSED FINANCIAL INFORMATION (Parent Company Only)
BALANCE SHEETS
(dollars in thousands)
| |
|
|
|
|
|
|
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
|
|
Assets
|
|
|
|
|
|
|
|
|
|
Current assets:
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
13
|
|
|
$
|
13
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current assets
|
|
|
13
|
|
|
|
13
|
|
|
Investment in subsidiaries
|
|
|
97,615
|
|
|
|
95,515
|
|
|
|
|
|
|
|
|
|
|
|
|
Total assets
|
|
$
|
97,628
|
|
|
$
|
95,528
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities and shareholder’s equity
|
|
|
|
|
|
|
|
|
|
Current liabilities:
|
|
|
|
|
|
|
|
|
|
Intercompany payables
|
|
$
|
21
|
|
|
$
|
17
|
|
|
Income tax payable
|
|
|
—
|
|
|
|
2
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current liabilities
|
|
|
21
|
|
|
|
19
|
|
|
Shareholder’s equity:
|
|
|
|
|
|
|
|
|
|
Common shares, $14.25 par value — issued and
outstanding 1,000 shares
|
|
|
14
|
|
|
|
14
|
|
|
Paid-in Capital
|
|
|
135,661
|
|
|
|
137,338
|
|
|
Accumulated Deficit
|
|
|
(41,791
|
)
|
|
|
(53,889
|
)
|
|
Accumulated other comprehensive income
|
|
|
3,723
|
|
|
|
12,046
|
|
|
|
|
|
|
|
|
|
|
|
|
Total shareholder’s equity
|
|
|
97,607
|
|
|
|
95,509
|
|
|
|
|
|
|
|
|
|
|
|
|
Total liabilities and shareholder’s equity
|
|
$
|
97,628
|
|
|
$
|
95,528
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Condensed Financial Statements
II-2
ARIZONA CHEM
SWEDEN HOLDINGS AB
SCHEDULE I
CONDENSED FINANCIAL INFORMATION (Parent Company Only)
STATEMENTS OF OPERATIONS
(dollars in thousands)
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
|
|
|
|
|
|
|
|
March 1
|
|
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
|
Net sales
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
Cost of goods sold
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gross profit
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Operating expenses (income):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Selling, general and administrative
|
|
|
2
|
|
|
|
7
|
|
|
|
(9
|
)
|
|
Equity in net (income) loss of subsidiaries
|
|
|
(12,099
|
)
|
|
|
26,574
|
|
|
|
27,297
|
|
|
Unrealized foreign exchange (gain) loss
|
|
|
(1
|
)
|
|
|
19
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total operating expenses (income)
|
|
|
(12,098
|
)
|
|
|
26,600
|
|
|
|
27,288
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income (loss) before income tax expense (benefit)
|
|
|
12,098
|
|
|
|
(26,600
|
)
|
|
|
(27,288
|
)
|
|
Income tax (benefit) expense
|
|
|
—
|
|
|
|
(1
|
)
|
|
|
2
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income (loss)
|
|
$
|
12,098
|
|
|
$
|
(26,599
|
)
|
|
$
|
(27,290
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Condensed Financial Statements
II-3
ARIZONA CHEM
SWEDEN HOLDINGS AB
SCHEDULE I
CONDENSED FINANCIAL INFORMATION (Parent Company Only)
STATEMENTS OF SHAREHOLDER’S EQUITY AND COMPREHENSIVE INCOME
(LOSS)
(dollars in thousands)
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Retained
|
|
|
Accumulated
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Earnings
|
|
|
Other
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Paid-in
|
|
|
(Accumulated
|
|
|
Comprehensive
|
|
|
|
|
|
|
|
Shares
|
|
|
Amount
|
|
|
Capital
|
|
|
Deficit)
|
|
|
Income
|
|
|
Total
|
|
|
|
|
Balance at March 1, 2007
|
|
|
1,000
|
|
|
$
|
14
|
|
|
$
|
129,986
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
130,000
|
|
|
Net loss
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(27,290
|
)
|
|
|
—
|
|
|
|
(27,290
|
)
|
|
Foreign currency translation adjustment
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(297
|
)
|
|
|
(297
|
)
|
|
Net gain from pension plans, net of tax
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,632
|
|
|
|
1,632
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Comprehensive loss
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(25,955
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2007
|
|
|
1,000
|
|
|
|
14
|
|
|
|
129,986
|
|
|
|
(27,290
|
)
|
|
|
1,335
|
|
|
|
104,045
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Contribution of debt by Rhône Capital
|
|
|
—
|
|
|
|
—
|
|
|
|
7,352
|
|
|
|
—
|
|
|
|
—
|
|
|
|
7,352
|
|
|
Net loss
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(26,599
|
)
|
|
|
—
|
|
|
|
(26,599
|
)
|
|
Foreign currency translation adjustment
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
12,755
|
|
|
|
12,755
|
|
|
Net loss from pension plans, net of tax
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(2,044
|
)
|
|
|
(2,044
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Comprehensive loss
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(15,888
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2008
|
|
|
1,000
|
|
|
|
14
|
|
|
|
137,338
|
|
|
|
(53,889
|
)
|
|
|
12,046
|
|
|
|
95,509
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Distribution to Parent
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,677
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,677
|
)
|
|
Net income
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
12,098
|
|
|
|
—
|
|
|
|
12,098
|
|
|
Foreign currency translation adjustment
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(5,902
|
)
|
|
|
(5,902
|
)
|
|
Net loss from pension plans, net of tax
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(2,421
|
)
|
|
|
(2,421
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Comprehensive income
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
3,775
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2009
|
|
|
1,000
|
|
|
$
|
14
|
|
|
$
|
135,661
|
|
|
$
|
(41,791
|
)
|
|
$
|
3,723
|
|
|
$
|
97,607
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Condensed Financial Statements
II-4
ARIZONA CHEM
SWEDEN HOLDINGS AB
SCHEDULE I
CONDENSED FINANCIAL INFORMATION (Parent Company Only)
STATEMENTS OF CASH FLOWS
(dollars in thousands)
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor
|
|
|
|
|
|
|
|
|
|
|
March 1
|
|
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
through
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
|
Operating activities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income (loss)
|
|
$
|
12,098
|
|
|
$
|
(26,599
|
)
|
|
$
|
(27,290
|
)
|
|
Adjustments to reconcile net income (loss) to net cash provided
by (used in) operating activities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Equity in undistributed earnings of subsidiaries
|
|
|
(12,099
|
)
|
|
|
26,574
|
|
|
|
27,297
|
|
|
Change in assets and liabilities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Intercompany payable
|
|
|
4
|
|
|
|
(474
|
)
|
|
|
491
|
|
|
Income tax payable
|
|
|
(2
|
)
|
|
|
—
|
|
|
|
2
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash provided by (used in) operating activities
|
|
|
1
|
|
|
|
(499
|
)
|
|
|
500
|
|
|
Investing activities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash provided by (used in) investing activities
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Financing activities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash provided by (used in) financing activities
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Effect of foreign exchange rate changes on cash and cash
equivalents
|
|
|
(1
|
)
|
|
|
(5
|
)
|
|
|
17
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Increase (decrease) in cash and cash equivalents
|
|
|
—
|
|
|
|
(504
|
)
|
|
|
517
|
|
|
Cash and cash equivalents at beginning of period
|
|
|
13
|
|
|
|
517
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents at end of period
|
|
$
|
13
|
|
|
$
|
13
|
|
|
$
|
517
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Supplemental cash flows information:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash paid during the period for:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income taxes
|
|
$
|
2
|
|
|
$
|
2
|
|
|
$
|
—
|
|
|
Noncash investing and financing activities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Contribution by Rhône Capital/debt retirement
|
|
$
|
—
|
|
|
$
|
7,352
|
|
|
$
|
—
|
|
See Notes to Condensed Financial Statements
II-5
ARIZONA CHEM
SWEDEN HOLDINGS AB
SCHEDULE I
NOTES TO CONDENSED FINANCIAL INFORMATION (Parent Company
Only)
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2009 AND 2008,
AND THE TEN MONTH PERIOD ENDED DECEMBER 31, 2007
Arizona Chem Sweden Holdings AB is a company organized under the
laws of Sweden and a wholly-owned subsidiary of AZ Chem
Luxembourg Holdings S.à.r.l. AZ Chem Luxembourg Holdings
S.à.r.l. is a direct, wholly-owned subsidiary of AZ Chem
Investments Partners LP, the majority of the interests of which
are owned by funds associated with Rhône Capital L.L.C. The
financial statements are prepared in accordance with accounting
principles generally accepted in the United States of America
(“U.S. GAAP”).
Arizona Chem Sweden Holdings AB has two wholly-owned
subsidiaries, AZ Chem US Holdings Inc. and Arizona Chemical AB,
which are the parent companies for our North American and
European operations, respectively.
Pursuant to the rules and regulations of the SEC, the condensed
financial statements of the parent company do not include all of
the information and notes normally included with financial
statements prepared in accordance with accounting principles
generally accepted in the United States of America. In addition,
for purposes of this schedule, the investments in wholly-owned
subsidiaries are accounted for using the equity method of
accounting which is not in accordance with U.S. GAAP. It
is, therefore suggested that these condensed financial
statements be read in conjunction with the consolidated
financial statements and notes thereto.
On February 28, 2007, Rhône Capital III, L.P.
(“Rhône Capital”) acquired us from International
Paper Company (“International Paper”), pursuant to a
Stock Purchase Agreement (the “Stock Purchase
Agreement”) for approximately $486.7 million plus
direct acquisition costs, creating Arizona Chem Sweden Holdings
AB and subsidiaries (the “Acquisition”). The
Acquisition was funded with cash of $130 million and
substantially all of the proceeds from the issuance of debt
totaling $375 million.
In connection with the Acquisition, our subsidiaries entered
into a First Lien Credit and Guaranty Agreement, which we refer
to as our “First Lien Credit Agreement”, and a Second
Lien Credit and Guaranty Agreement, which we refer to as our
“Second Lien Credit Agreement”, respectively. We refer
to these agreements together as our “credit
agreements”. We are the guarantors with respect to these
credit agreements. These credit agreements have certain
restrictions that limit our subsidiaries from paying dividends
or making any other distributions to us and restrict our ability
to pay dividends or any other distributions. As a result of
these transactions, on a consolidated basis we are highly
leveraged. As of December 31, 2009, our total consolidated
indebtedness was $338.9 million.
As part of the credit agreements all credit parties, which
include us and our subsidiaries, have certain restrictions that
limit paying dividends or making any other distributions,
repaying or prepaying any indebtedness owed, making loans or
advances, or transferring, leasing or licensing any of its
property or assets.
II-6
ARIZONA CHEM
SWEDEN HOLDINGS AB
SCHEDULE II
VALUATION AND
QUALIFYING ACCOUNTS AND RESERVES
(dollars in
thousands)
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Charged to
|
|
|
|
|
|
|
|
|
|
Balance,
|
|
(Recovery of)
|
|
|
|
|
|
|
|
|
|
Beginning of
|
|
and Costs
|
|
Charged to
|
|
|
|
Balance, End of
|
|
|
|
Year
|
|
Expenses
|
|
Other Accounts
|
|
Deductions
|
|
Year
|
|
|
|
Deferred Tax Asset
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Valuation Allowances
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Predecessor:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2 months ended February 28, 2007
|
|
$
|
27,682
|
|
|
$
|
816
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
28,498
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10 months ended December 31, 2007
|
|
|
41,364
|
|
|
|
2,570
|
|
|
|
—
|
|
|
|
—
|
|
|
|
43,934
|
|
|
Year ended December 31, 2008
|
|
|
43,934
|
|
|
|
4,918
|
|
|
|
(7,685
|
)
|
|
|
—
|
|
|
|
41,167
|
|
|
Year ended December 31, 2009
|
|
|
41,167
|
|
|
|
2,476
|
|
|
|
1,662
|
|
|
|
—
|
|
|
|
45,305
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Environmental Costs and Obligations
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Environmental Liabilities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Predecessor:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2 months ended February 28, 2007
|
|
$
|
416
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
(7
|
)
|
|
$
|
409
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Successor:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10 months ended December 31, 2007
|
|
|
409
|
|
|
|
2,985
|
|
|
|
—
|
|
|
|
—
|
|
|
|
3,394
|
|
|
Year ended December 31, 2008
|
|
|
3,394
|
|
|
|
441
|
|
|
|
—
|
|
|
|
(642
|
)
|
|
|
3,193
|
|
|
Year ended December 31, 2009
|
|
|
3,193
|
|
|
|
1,980
|
|
|
|
—
|
|
|
|
(1,036
|
)
|
|
|
4,137
|
|
(a) The undersigned registrant hereby undertakes to provide
to the underwriter at the closing specified in the underwriting
agreements, certificates in such denominations and registered in
such names as required by the underwriter to permit prompt
delivery to each purchaser.
(b) Insofar as indemnification for liabilities arising
under the Securities Act may be permitted to directors, officers
and controlling persons of the registrant pursuant to the
foregoing provisions, or otherwise, the registrant has been
advised that in the opinion of the Securities and Exchange
Commission such indemnification is against public policy as
expressed in the Securities Act and is, therefore,
unenforceable. In the event that a claim for indemnification
against such liabilities (other than the payment by the
registrant of expenses incurred or paid by a director, officer
or controlling person of the registrant in the successful
defense of any action, suit or proceeding) is asserted by such
director, officer or controlling person in connection with the
securities being registered, the registrant will, unless in the
opinion of its counsel the matter has been settled by
controlling precedent,
II-7
submit to a court of appropriate jurisdiction the question
whether such indemnification by it is against public policy as
expressed in the Securities Act and will be governed by the
final adjudication of such issue.
(c) The undersigned registrant hereby undertakes that:
i. For purposes of determining any liability under the
Securities Act, the information omitted from the form of
prospectus filed as part of this registration statement in
reliance upon Rule 430A and contained in a form of
prospectus filed by the registrant pursuant to Rule 424(b)(1) or
(4) or 497(h) under the Securities Act shall be deemed to
be part of this registration statement as of the time it was
declared effective.
ii. For the purpose of determining any liability under the
Securities Act, each post-effective amendment that contains a
form of prospectus shall be deemed to be a new registration
statement relating to the securities offered therein, and the
offering of such securities at that time shall be deemed to be
the initial bona fide offering thereof.
II-8
SIGNATURES
Pursuant to the requirements of the Securities Act of 1933, the
registrant has duly caused this Amendment No. 2 to the
Registration Statement to be signed on its behalf by the
undersigned, thereunto duly authorized, in the City of Almere,
the Netherlands, on the 28th day of June, 2010.
Arizona Chemical Ltd.
Cornelis Verhaar
President and Chief Executive Officer
Frederic Jung
Vice President and Chief Financial Officer
Pursuant to the requirements of the Securities Act of 1933, this
Amendment No. 2 to the Registration Statement has been
signed by the following persons in the capacities indicated on
June 28, 2010.
| |
|
|
|
|
|
Signature
|
|
Title
|
|
|
|
*
Cornelis
Verhaar
|
|
Director, President and
Chief Executive Officer
(Principal Executive Officer)
|
|
*
Frederic
Jung
|
|
Vice President and Chief Financial Officer (Principal
Financial Officer)
|
|
*
Astrid
van der Valk
|
|
Corporate Controller
(Controller)
|
|
*
Eytan
Tigay
|
|
Director, Chairman
|
|
*
Leonard
Berlik
|
|
Director
|
|
*
John
R. Bolton
|
|
Director
|
|
*
Jochen
Krautter
|
|
Director
|
|
*
Petter
Johnsson
|
|
Director
|
|
*
Gerald
Marterer
|
|
Director
|
II-9
| |
|
|
|
|
|
Signature
|
|
Title
|
|
|
|
*
Sebastien
Mazella di Bosco
|
|
Director
|
|
*By:
|
|
/s/ Cornelis
Verhaar
Cornelis
Verhaar
Attorney-in-Fact
|
|
|
Pursuant to the requirements of Section 6(a) of the
Securities Act of 1933, the Authorized Representative has duly
caused the registration statement to be signed on its behalf by
the undersigned, solely in her capacity as the duly authorized
representative of Arizona Chemical Ltd. in the United States, in
the City of Jacksonville, State of Florida, on June 28,
2010.
|
|
|
| |
By:
|
/s/ Pamela
J. Simmons
|
Name: Pamela J. Simmons
Title: Counsel
II-10
EXHIBIT INDEX
| |
|
|
|
|
|
Exhibit No.
|
|
Description
|
|
|
|
|
1
|
.1
|
|
Form of Underwriting Agreement.*
|
|
|
3
|
.1
|
|
Certificate of Incorporation and Memorandum of Association.**
|
|
|
3
|
.2
|
|
Form of Bye-laws.*
|
|
|
4
|
.1
|
|
Form of Shareholders Agreement.*
|
|
|
4
|
.2
|
|
Form of Certificate for Common Shares.*
|
|
|
5
|
.1
|
|
Opinion of Conyers Dill & Pearman Limited, Bermuda.*
|
|
|
8
|
.1
|
|
Tax Opinion of Conyers Dill & Pearman Limited, Bermuda.*
|
|
|
8
|
.2
|
|
Tax Opinion of Sullivan & Cromwell LLP.*
|
|
|
10
|
.1
|
|
Second Amended and Restated CTO/BLS Supply Agreement dated as of
November 17, 2009, by and between International Paper
Company and Arizona Chemical Company, LLC.**
|
|
|
10
|
.2
|
|
Second Amended and Restated CST Supply Agreement dated as of
November 17, 2009, by and between International Paper
Company and Arizona Chemical Company, LLC.**
|
|
|
10
|
.3
|
|
First Lien Credit and Guaranty Agreement dated as of
February 28, 2007, among AZ Chem US Inc., as U.S. Borrower,
Proserpina 1073 AB (under change of name to Arizona Chem Sweden
AB), as European Borrower, Proserpina 1072 AB (under change of
name to Arizona Chem Sweden Holdings AB) and certain
subsidiaries of Arizona Chem Sweden Holdings AB, as Guarantors,
various lenders, Goldman Sachs Credit Partners L.P., as Lead
Arranger, Bookrunner, Syndication Agent, Administrative Agent
and Collateral Agent, and Bank of America, N.A., as
Documentation Agent.**
|
|
|
10
|
.4
|
|
First Amendment to First Lien Credit and Guaranty Agreement
dated as of July 1, 2008, by and among AZ Chem US Inc.,
Arizona Chemical AB, Goldman Sachs Credit Partners L.P., as
Administrative Agent, and for purposes of Section IV
thereof, the guarantors listed on the signature pages thereto.**
|
|
|
10
|
.5
|
|
Second Amendment to First Lien Credit and Guaranty Agreement
dated as of July 24, 2008, by and among AZ Chem US Inc.,
Arizona Chemical AB, Goldman Sachs Credit Partners L.P., as
Administrative Agent, and for purposes of Section IV
thereof, the guarantors listed on the signature pages thereto.**
|
|
|
10
|
.6
|
|
Third Amendment to First Lien Credit and Guaranty Agreement
dated as of November 14, 2008 by and among AZ Chem US Inc.,
Arizona Chemical AB, Goldman Sachs Credit Partners L.P., as
Administrative Agent, and for purposes of Section IV
thereof, the guarantors listed on the signature pages thereto.**
|
|
|
10
|
.7
|
|
First Lien Pledge and Security Agreement dated as of
February 28, 2007 between each of the Grantors party
thereto and Goldman Sachs Credit Partners L.P., as Collateral
Agent.**
|
|
|
10
|
.8
|
|
Second Lien Credit and Guaranty Agreement dated as of
February 28, 2007, among AZ Chem US Inc., as Borrower, AZ
Chem US Holdings Inc. and certain subsidiaries of AZ Chem US
Holdings Inc., as Guarantors, various lenders, Goldman Sachs
Credit Partners L.P., as Lead Arranger, Bookrunner and
Syndication Agent, and CapitalSource Finance LLC, as
Administrative Agent and Collateral Agent.**
|
|
|
10
|
.9
|
|
First Amendment to Second Lien Credit and Guaranty Agreement
dated as of July 24, 2008 by and among AZ Chem US Inc.,
CapitalSource Finance LLC, as Administrative Agent, Goldman
Sachs Credit Partners L.P., as Syndication Agent, for purposes
of Section IV thereof, and the guarantors listed on the
signature pages thereto.**
|
|
|
10
|
.10
|
|
Second Lien Pledge and Security Agreement dated as of
February 28, 2007 between each of the Grantors party
thereto and CapitalSource Finance LLC, as Collateral Agent.**
|
|
|
10
|
.11
|
|
Lease Agreement dated as of February 28, 2007, between
International Paper Company and Arizona Chemical Company.**
|
|
|
10
|
.12
|
|
Employment Agreement dated August 18, 2008, between Arizona
Chemical B.V. and Cornelis Verhaar.**
|
| |
|
|
|
|
|
Exhibit No.
|
|
Description
|
|
|
|
|
10
|
.13
|
|
Letter of Understanding for Frederic Jung —
International Assignment in Almere, The Netherlands dated
August 28, 2009 between Arizona Chemical LLC and Frederic
Jung.**
|
|
|
10
|
.14
|
|
Employment Agreement dated November 27, 2008 between
Arizona Chemical B.V. and Dick Stuyfzand.**
|
|
|
10
|
.15
|
|
Employment Agreement dated June 30, 2006 between Arizona
Chemical B.V. and Juhani Tuovinen.**
|
|
|
10
|
.16
|
|
2010 Long-Term Incentive Plan.*
|
|
|
10
|
.17
|
|
Amended and Restated Arizona Chemical Company, LLC U.S. Salaried
Employee Severance Plan and Summary Plan Description dated
February 10, 2010.**
|
|
|
10
|
.18
|
|
Fourth Amendment to First Lien Credit and Guaranty Agreement
dated as of May 28, 2010 by and among AZ Chem US Inc.,
Arizona Chemical AB, Goldman Sachs Credit Partners L.P., as
Administrative Agent, and, for purposes of Section V
thereof, the guarantors listed on the signature pages thereto.**
|
|
|
10
|
.19
|
|
Second Amendment to Second Lien Credit and Guaranty Agreement
dated as of May 28, 2010 by and among AZ Chem US Inc.,
CapitalSource Finance LLC, as Administrative Agent, Goldman
Sachs Credit Partners L.P., as Syndication Agent, and, for
purposes of Section V thereof, the guarantors listed on the
signature pages thereto.**
|
|
|
10
|
.20
|
|
Amended and Restated Arizona Chemical Company, LLC U.S. Salaried
Employee Severance Plan and Summary Plan Description dated
May 1, 2010.
|
|
|
21
|
.1
|
|
Subsidiaries of Arizona Chemical Ltd.**
|
|
|
23
|
.1
|
|
Consent of Conyers Dill & Pearman Limited, Bermuda.*
|
|
|
23
|
.2
|
|
Consent of Deloitte & Touche LLP.
|
|
|
23
|
.3
|
|
Consent of Deloitte & Touche LLP.
|
|
|
23
|
.4
|
|
Consent of Arthur D. Little Benelux S.A./N.V.**
|
|
|
24
|
.1
|
|
Power of Attorney**
|
|
|
|
|
* |
|
To be filed by amendment. |
| |
|
** |
|
Previously filed. |