Attached files
| file | filename |
|---|---|
| EX-32.1 - EX-32.1 - AMERICAN MEDICAL SYSTEMS HOLDINGS INC | c56509exv32w1.htm |
| EX-21.1 - EX-21.1 - AMERICAN MEDICAL SYSTEMS HOLDINGS INC | c56509exv21w1.htm |
| EX-31.1 - EX-31.1 - AMERICAN MEDICAL SYSTEMS HOLDINGS INC | c56509exv31w1.htm |
| EX-23.1 - EX-23.1 - AMERICAN MEDICAL SYSTEMS HOLDINGS INC | c56509exv23w1.htm |
| EX-31.2 - EX-31.2 - AMERICAN MEDICAL SYSTEMS HOLDINGS INC | c56509exv31w2.htm |
| EX-10.24 - EX-10.24 - AMERICAN MEDICAL SYSTEMS HOLDINGS INC | c56509exv10w24.htm |
| EX-10.26 - EX-10.26 - AMERICAN MEDICAL SYSTEMS HOLDINGS INC | c56509exv10w26.htm |
Table of Contents
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
Form 10-K
| Annual Report Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 |
| For the fiscal year ended: | Commission file number: | |
| January 2, 2010 | 000 – 30733 |
AMERICAN MEDICAL SYSTEMS HOLDINGS, INC.
(Exact Name of Registrant as Specified in Its Charter)
| Delaware | 41-1978822 | |
| (State of Incorporation) | (IRS Employer Identification No.) |
10700 Bren Road West
Minnetonka, Minnesota 55343
(Address of Principal Executive Offices, Including Zip Code)
Minnetonka, Minnesota 55343
(Address of Principal Executive Offices, Including Zip Code)
Registrant’s Telephone Number, Including Area Code:
952-930-6000
952-930-6000
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class: | Name of each exchange on which registered: | |
| Common stock, par value $.01 per share | The Nasdaq Stock Market LLC | |
| (Nasdaq Global Select Market) |
Securities registered pursuant to Section 12(g) of the Act:
None
None
Indicate by check mark if the registrant is well-known seasoned issuer, as defined in Rule 405
of the Securities Act. Yes þ No o
Indicate by check mark if the registrant is not required to file reports pursuant to Section
13 or Section 15(d) of the Act. Yes o No þ
Indicate by check mark whether the registrant (1) has filed all reports required to be filed
by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or
for such shorter period that the Registrant was required to file such reports), and (2) has been
subject to such filing requirements for the past 90 days. Yes þ No o
Indicate by check mark whether the registrant has submitted electronically and posted on its
corporate Web site, if any, every Interactive Data File required to be submitted and posted
pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months
(or for such shorter period that the registrant was required to submit and post such files). Yes
o No o
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation
S-K is not contained herein, and will not be contained, to the best of registrant’s knowledge, in
definitive proxy or information statements incorporated by reference in Part III of this Form 10-K
or any amendment to this Form 10-K. o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated
filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large
accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the
Exchange Act. (Check one):
| Large accelerated filer þ | Accelerated filer o | Non-accelerated filer o | Smaller reporting company o | |||
| (Do not check if a smaller reporting company) |
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of
the Act). Yes o No þ
As of July 2, 2009, the last business day of the registrant’s most recently completed second
fiscal quarter, the aggregate market value of the common stock of the registrant (based upon the
closing price of the common stock as of that date as reported by The Nasdaq Stock Market LLC and
excluding outstanding shares beneficially owned by directors, executive officers, and affiliates)
was approximately $821,310,455.
As
of February 19, 2010, 75,120,750 shares of Common Stock of the registrant were
outstanding.
Part III of this Annual Report on Form 10-K incorporates by reference information (to the
extent specific sections are referred to in this Annual Report) from the registrant’s Proxy
Statement for its 2010 Annual Meeting of Stockholders to be held April 29, 2010 (the “2010 Proxy
Statement”).
AMERICAN MEDICAL SYSTEMS HOLDINGS, INC.
FORM 10-K
For the Fiscal Year Ended January 2, 2010
TABLE OF CONTENTS
| Description | Page | |||||||
| 1 | ||||||||
| 10 | ||||||||
| 18 | ||||||||
| 18 | ||||||||
| 19 | ||||||||
| 19 | ||||||||
| 20 | ||||||||
| 22 | ||||||||
| 24 | ||||||||
| 41 | ||||||||
| 42 | ||||||||
| 42 | ||||||||
| 42 | ||||||||
| 43 | ||||||||
| 44 | ||||||||
| 44 | ||||||||
| 44 | ||||||||
| 44 | ||||||||
| 44 | ||||||||
| 45 | ||||||||
| F-1 | ||||||||
| E-1 | ||||||||
| EX-10.24 | ||||||||
| EX-10.26 | ||||||||
| EX-21.1 | ||||||||
| EX-23.1 | ||||||||
| EX-31.1 | ||||||||
| EX-31.2 | ||||||||
| EX-32.1 | ||||||||
Table of Contents
FORWARD-LOOKING INFORMATION
This Annual Report on Form 10-K contains forward-looking statements. Any statements not of
historical fact may be considered forward-looking statements. These statements by their nature
involve substantial risks and uncertainties, and actual results may differ materially from those
expressed in such forward-looking statements as a result of many factors, including, but not
limited to, those discussed under the “Forward-Looking Statements” section of Part II, Item 7,
“Management’s Discussion and Analysis of Financial Condition and Results of Operations.”
As used in this report, references to “American Medical Systems,” the “company,” “we,” “our” or
“us,” unless the context otherwise requires, refer to American Medical Systems Holdings, Inc. and
its subsidiaries.
We own or have rights to various trademarks, trade names or service marks, including the following:
AMS 700 ®,
AMS 800 ®, AMS 700 LGX
®, AMS 800
®, AMS 700
® MS™, InVance
® AdVance
®, InhibiZone
®,
Acticon®, UroLume
®, InhibiZone
®, Tactile Pump
®, Momentary Squeeze Pump™, Spectra™, GreenLight
HPS®, GreenLight PV
®, StoneLight
®, SureFlex, TherMatrx
®, MiniArc
®, Monarc
®, Acticon
®, Apogee
®,
Perigee®, InteXen
®, and Elevate
®. The trademarks Viagra
®, Levitra
®, Cialis
®, and Her Option®
referred to in this annual report on Form 10-K are the registered trademarks of others.
PART I
| Item 1. | Business |
Overview
Our company was formed in 1972 and we completed our 37th year of operations in 2009,
with a continued focus on technological innovation, market expansion and financial strength.
We are a world leader in developing and delivering innovative medical technology solutions to
physicians treating men’s and women’s pelvic health conditions, thereby recognized as a technology
leader in the markets we serve. We have built a business that delivers growth, fueled by a robust
pipeline of innovative products for significant, under-penetrated markets. We have a diverse
product portfolio, which treats men’s incontinence, erectile dysfunction, and benign prostatic
hyperplasia (BPH), and treats women’s incontinence and pelvic floor prolapse. We estimate there are
as many as 1.6 billion incidences of these conditions in the global markets we serve, with many
people suffering from multiple conditions. Treatment options for these conditions vary
considerably depending on the severity of the condition. Approximately 350 million of these men
and women have conditions sufficiently severe so as to profoundly diminish their quality of life
and significantly impact their relationships. Our addressable market is contained within this
group of patients. Our product development and acquisition strategies have focused on expanding
our product offering for surgical solutions, including less-invasive solutions for surgeons and
their patients. Our primary physician customers include urologists, gynecologists, urogynecologists
and colorectal surgeons.
Our net sales grew from $501.6 million in 2008 to $519.3 million in 2009. In 2009, men’s health
contributed $234.6 million, or 45 percent of total net sales, BPH therapy contributed $114.5
million, or 22 percent of total net sales, and women’s health contributed $170.2 million, or 33
percent of total net sales.
Our men’s health products for treating incontinence and erectile restoration, and our women’s
health products for treating pelvic floor repair, were key drivers in revenue growth in 2009. In
particular, the AMS 800 ® (our artificial urinary sphincter product) and the AMS
700 ® (our erectile restoration product), led our sales growth in men’s health, and the
successful launch in mid-2009 of the new Elevate® anterior product for pelvic floor
repair led our women’s health sales growth in the second half of the year. Our 2009 sales saw a
modest decline over 2008 in our BPH therapy products, which is our full line of products to deliver
minimally invasive procedures for the treatment of obstructive BPH and urinary stones. However, we
achieved accelerating sales growth in our BPH therapy product line each sequential quarter
throughout 2009, and 2009 sales of fibers used by the GreenLight HPS ® (High
Performance System) laser system exceeded 2008 fiber sales. Our women’s health products for
treating incontinence had modest growth in 2009 due to the continued solid growth of MiniArc
®.
1
Table of Contents
In 2009, we continued to focus on initiatives which reduce working capital, manage expenses and
drive operating leverage throughout our business. We generated cash from operating activities of
$122.8 million in 2009, compared to $115.8 million in 2008. We also reduced our debt by $130.8
million in 2009, compared to $119.7 million in 2008. We earned net income of $84.8 million in
2009, compared to $30.8 million in 2008.
Based on our areas of competitive strength, our strategy is to expand the reach of our products
and address unmet needs in both established and new pelvic health markets. We determined our female sterilization
assets and technology (Ovion technology) and our Her Option® global endometrial cryoablation product
line did not fit our long-term strategy. During the third quarter of 2009, we sold our Ovion technology for $23.6
million (see Notes to Consolidated Financial Statements — No. 5, Goodwill and Intangible Assets). In
February of 2010, we sold the Her Option® product line for $20.5 million (see Notes
to Consolidated Financial Statements — No. 16, Subsequent Events). The sale of these
non-strategic assets will allow us to concentrate our efforts and resources on improving and
expanding the global reach of our products to restore quality of life to men and women through our
innovative, life-changing solutions.
We maintain a website at www.AmericanMedicalSystems.com. We are not including the information
contained on our website as a part of, nor incorporating it by reference into, this Annual Report
on Form 10-K. We make available free of charge on our website our Annual Reports on Form 10-K,
Quarterly Reports on Form 10-Q, Current Reports on Form 8-K, and amendments to these reports, as
soon as reasonably practicable after we electronically file such material with, or furnish such
material to, the Securities and Exchange Commission.
Markets and Products
In recent years, the number of people seeking treatment for various pelvic health disorders has
grown with the publicity for new treatments and drug therapies, but the portion of afflicted
patients seeking treatment remains relatively low. When patients seek treatment, they generally
begin with options that will be as minimally invasive as possible, such as pharmaceutical
therapies. Also, when patients initially seek treatment, their first physician contact is usually
with a general practitioner and not with a surgical specialist. If less invasive options have
proven unsuccessful, patients and their physicians may then consider surgery as a solution for
their pelvic health disorder.
Sales of our products benefit from some of the same factors which drive sales in many other medical
device companies: an aging population with a desire to maintain a high quality of life, the
expanding availability of safe and effective treatments, minimally invasive solutions, expanded
options for in-office treatments, and increasing patient and physician awareness of these
treatments.
The diseases we treat can profoundly affect the quality of one’s life and the burden of these
diseases increases with age. The incidence of incontinence, erectile dysfunction and benign
prostatic hyperplasia in men increases with age and with the incidence of prostate cancer surgery.
Female incontinence and pelvic organ prolapse are linked to pregnancy and childbirth among younger
women, but also occur independently as women age.
As a result, we believe that as the “baby boomer” generation continues to age, the growth in the
prospective patient pool for our products will accelerate. We also believe that this demographic
group and those that follow will be less willing to accept the natural deterioration of body
functions. We believe their desire to maintain a consistent quality of life will amplify their
increased demand for our products and therapies. As a result, our strategy of providing an
expanding portfolio of treatment options is an important business driver. In the last several
years, we have successfully introduced new products and therapies to meet our target physician and
patient needs. Our product development and acquisition strategies have focused on expanding our
product offering with products and procedures that improve outcomes, reduce operating time and
trauma, economically benefit the overall healthcare system, and thereby increase the value of our
products to physicians, patients, and payers.
Increasing patient awareness of these new treatments is critical to our continued success. We
believe that advertising by pharmaceutical companies and increased private internet access to
healthcare information has greatly increased patients’ awareness of treatment options for their
medical conditions. For example, erectile dysfunction has become a more widely recognized disease
largely due to the pharmaceutical industry’s extensive advertising campaign for Viagra®,
Levitra® and Cialis®. Going forward, we expect continued advertisements to
drive awareness of other pelvic health disorders. As individuals seek medical treatment, we expect
many of them will learn about and choose a treatment using one of our products. We facilitate that
decision by working closely with physicians who are skilled in procedures using our products and
therapies, and by co-sponsoring meetings and community health talks where patients can learn more
about the benefits of these procedures. In 2009, thousands of men and women attended community
health talks on the conditions we treat. While the principal focus of our education efforts
continues to be with physicians, we continue to expand our patient awareness initiatives, primarily
through conducting co-sponsored education and marketing activities with physician practices, and
will continue to focus on patient initiatives in the future. Building physician awareness
continues to be an important element of our education and marketing strategy. Physician training on
the anatomy, physiology and surgical procedures surrounding pelvic health has become one of our
core competencies. We trained approximately 9,600 physicians on our products and therapies in
2009. We believe our extensive experience in the pelvic health arena has resulted in a very strong
franchise with urologists and we are working to build a similarly strong franchise with
urogynecologists,
2
Table of Contents
surgical gynecologists and colorectal surgeons. The gynecology specialty is critical to our growth
because most women who suffer from incontinence, pelvic organ prolapse and other pelvic disorders
are likely to be referred to a gynecologist after first seeking help from their primary care
physician. Longer term, we believe that colorectal surgeons will also be important to our success.
The expansion of our product offering, increasing physician and patient awareness and our focus on
expanding our international operations, has greatly increased our business opportunities. We have
developed and successfully introduced a number of new products and product improvements, most
recently the MiniArc® Single-Incision Sling for female incontinence, the AMS
700 LGX® and Spectra™ (one-piece concealable penile prosthesis) for erectile
restoration, and the Elevate® posterior and anterior transvaginal pelvic floor repair
system. We placed continuous focus on expanding our marketing of the GreenLight
HPS® lasers and fibers for the treatment of BPH and the
StoneLight® laser and fibers for the treatment of urinary stones. We initiated
clinical studies for a variety of products in our incontinence, BPH therapy and pelvic floor repair
businesses during 2009. We remain committed to spending approximately ten percent of our sales
over the long term on research and development in order to develop new products and product
improvements, generate robust clinical data, and continue to be recognized as the world leader in
pelvic health innovation.
Men’s Health
Over 50 million men worldwide suffer from urinary incontinence, the involuntary release of urine
from the body. In men, this can be caused by nerve and sphincter damage from prostate cancer
surgery. Male incontinence may be managed with a catheter and leg bag to collect the urine, or
with pads and diapers to absorb the leaks. These measures are far from ideal, as they come with
recurring replacement product costs, the potential for infection, and embarrassing leaks and odor,
not to mention a significantly diminished quality of life or need for managed care.
Since 1972, when we introduced the predecessor to today’s AMS 800® Artificial Urinary
Sphincter, we have been the primary medical device company supplying surgical solutions for male
incontinence. This fully implanted system includes an inflatable urethral cuff to restrict flow
through the urethra, and a control pump which allows the patient to discreetly open the cuff when
he wishes to urinate. Since 2000, we have also been selling the InVance® sling system,
a less-invasive procedure for men with moderate incontinence. Our newer solution, the
AdVance® sling system for the treatment of mild to moderate stress urinary incontinence,
has been a key driver of our success since its release in 2007. The AMS 800® with
InhibiZone® has successfully reduced surgical infections, similar to the results seen in
erectile restoration applications. We also offer the UroLume® endoprosthesis stent as a
less invasive procedure for men within this group who may not be good surgical candidates, as well
as for men suffering from bulbar urethral strictures. Our Acticon® Neosphincter is used
to treat severe fecal incontinence, the loss of bowel control, in men for whom less invasive
treatments have failed.
Erectile dysfunction is the inability to achieve or maintain an erection sufficient for sexual
intercourse. When this condition is not improved by drugs, it is most often caused by vascular
disease, complications from diabetes, or prostate surgery which can damage both nerves and arteries
necessary for erectile function. This disease can also be caused by spinal cord injury, and may
have a psychogenic component. We estimate that erectile dysfunction may affect over 400 million
men and their partners around the world. The primary treatment for erectile dysfunction is the
class of drugs referred to as PDE-5 inhibitors. Less than 70 percent of patients using these drugs
have a positive response. The failed patient may try a vacuum device or a topical or injected drug
before considering a penile implant such as those we offer. If the patient elects to have implant
surgery, the surgeon implants a penile prosthesis, which is a consistent, reliable solution.
We lead the penile implant market with a series of semi-rigid malleable prostheses and a complete
range of more naturally functioning inflatable prostheses, including the AMS 700®
MS™. We have continued to improve our market leading implants over the years, and our
AMS 700® inflatable prostheses includes a Parylene coating on certain internal surfaces
of the prosthesis to increase durability, the InhibiZone® antibiotic coating for which
we received U.S. Food and Drug Administration (FDA) approval in July 2009 for our product claim
that InhibiZone® reduces the rate of revision surgery due to surgical infections, and
the Tactile Pump® and Momentary Squeeze Pump™, designed to improve ease of use for
patients. Physician preference for these new products contributed to the growth in erectile
restoration sales in the last three years.
3
Table of Contents
BPH Therapy
Our products can be used to relieve restrictions on the normal flow of urine from the bladder
caused by bladder obstructions, generally the result of BPH or bulbar urethral strictures.
Symptoms of BPH include increased urination frequency, sudden urges to urinate, and weak urine
flow. More than 70 percent of men over age 60 have some symptoms of BPH. Prior to the development
of less invasive therapies, the conventional treatment for those experiencing a physical
obstruction of the prostatic urethra was a surgical removal of the prostatic tissue performed under
general anesthesia in the operating room known as a transurethral resection of the prostate (TURP).
We offer men an alternative to a TURP, that is the GreenLight™ photovaporization of the
prostate. This laser therapy is designed to reduce the comorbidities associated with a TURP. The
GreenLight PV® laser system has paved the way for creating a new standard of care in the
treatment of BPH. This new standard of BPH care is further advanced by the GreenLight
HPS® which provides shorter treatment times with similar long-term results as the
earlier GreenLight PV® system, thereby enhancing user comfort. The GreenLight
HPS® offers a more intense laser beam balancing vaporization of tissue with coagulation
to prevent blood loss and providing enhanced surgical control compared to other laser systems, and
numerous system improvements over the GreenLight PV® system, for true plug-and-play
functionality. We also offer the StoneLight® laser and SureFlex™ fiber
optics for the treatment of urinary stones. StoneLight® is a lightweight and portable
15-watt holmium laser that offers the right amount of power to effectively fragment most urinary
stones. The SureFlex™ fiber optic line is engineered to deliver more energy safely and
effectively, even under maximum scope deflection, for high performance holmium laser lithotripsy.
For those men not yet to the point of urethral obstruction, but for whom symptomatic relief is
desired, therapeutic options include pharmaceuticals as well as less-invasive tissue ablation
techniques that can be performed in a physician’s office, including microwave therapy or
radiofrequency energy delivered to the prostate. The market for an office-based therapy for BPH
has remained relatively flat, at approximately 90,000 men treated annually, partially due to the
continued adoption of laser delivered BPH treatments. It is within this office-based market
segment that our TherMatrx® offering is positioned.
Women’s Health
Over 500 million women in our global markets suffer from urinary or fecal incontinence. These
diseases can lead to debilitating medical and social problems, ranging from embarrassment to
anxiety and depression. There are three types of urinary incontinence: stress, urge, and mixed
incontinence (a combination of stress and urge). While stress incontinence is generally caused by
a weakening of the pelvic floor and resultant hypermobility of the urethra, urge incontinence is
more complex and currently not as well understood. Pads and diapers are often used to contain and
absorb leaks, and may be acceptable for controlling mild incontinence. Drug therapy and electrical
nerve stimulation are currently used to treat urge incontinence. Incontinence may be treated
through exercises to strengthen pelvic floor muscles, or through the injection of collagen or some
other bulking agent into the wall of the urethra or bladder neck to narrow the passage. Surgical
solutions are generally recommended only if these other therapies are not effective. Our current
products in the market treat stress incontinence, which generally results from a weakening of the
tissue surrounding the bladder and urethra which can be a result of pregnancy, childbirth and
aging.
We offer a broad range of systems to restore female continence led by Monarc® and
MiniArc®. Monarc®, launched in 2003, has established itself as a standard
treatment for stress incontinence. It incorporates unique helical needles to place a
self-fixating, sub-fascial hammock through the obturator foramin. Our MiniArc®
Single-Incision Sling incontinence solution was released in 2007 and requires just one incision to
surgically place a small sling under the urethra, which minimizes tissue disruption and potential
for blood loss, thereby allowing the procedure to be done with less anesthesia on an outpatient
basis.
The Acticon® neosphincter, an extension of our urinary control technology, is used to
treat severe fecal incontinence primarily as the result of complications from childbirth, including
the episiotomy.
Pregnancy, labor, and childbirth may also cause pelvic organ prolapse and other pelvic floor
disorders. Prolapse and other pelvic floor defects may be treated with a variety of open,
laparoscopic, and transvaginal surgeries. Over 450,000 procedures are performed annually around the
world to repair some form of pelvic organ prolapse in women. These procedures have historically
been performed through the use of suture and graft materials designed
4
Table of Contents
for other surgical applications. We offer less invasive solutions for pelvic floor repair, with the
Apogee® and Perigee® systems. The Apogee® system is designed to
repair vaginal vault prolapse, a condition often resulting from the removal of the supporting
mechanisms for the apex of the vagina as the result of hysterectomy. The Perigee® system
targets repair of cystocele, or the herniation of the bladder through the anterior wall of the
vagina. We also offer InteXen®, a biologic graft as an alternative to our synthetic mesh
solutions. In 2008, we introduced the Elevate® transvaginal pelvic floor repair system,
with no external incisions. Using an anatomically designed needle and self-fixating tips,
Elevate® allows for safe, simple and precise mesh placement through a single vaginal
incision. The posterior system was launched in 2008 and the anterior system was launched in 2009.
Selling and Marketing
We sell our products in the United States, Canada, Australia, Brazil, and many western European
countries through direct field representatives. At the end of 2009, we had 500 employees in our
global sales and marketing force. We also ended 2009 with 68 independent distributors who represent
our products in countries where we do not have direct field representatives and accounted for
approximately 6.2 percent of our worldwide sales. In specific laser therapy markets in the United
States, we sell through a number of mobile providers. No single customer or group of customers
accounts for more than ten percent of our total sales. Local market conditions, including the
regulatory and competitive situation, determine the type of products we sell in each market.
Our marketing organization is responsible for understanding patient and physician needs, guiding
new product development, and increasing the awareness, understanding, and preference for our
products among physicians and patients.
In pricing our products we consider our costs of developing, manufacturing, and distributing the
products—including the cost of regulatory compliance and physician training—and the value they
bring to patients and the health care system. Similarly, we typically structure price increases to
coincide with the introduction of improved features, benefits and clinical-proved sources, which
add more value to our products.
Manufacturing and Supply
We use approximately 130,000 square feet of our facilities in Minnesota, California and Arizona for
manufacturing, warehousing, and distribution of our products. In 2010, we plan to consolidate our
laser therapy operations by moving the Arizona activities to our California facility, which we
anticipate will result in improved efficiencies by eliminating unnecessary redundancies. We
utilize warehouses to support local distribution in countries outside the U.S. where we have direct
sales representation. We maintain a single-shift manufacturing operation and employ lean
manufacturing approaches for the reduction of waste in manufacturing processes and alignment of
production with customer demand. Some of our products utilize raw materials or components that are
either single or sole-sourced.
We maintain a comprehensive quality assurance and quality control program, which includes
documentation of all material specifications, operating procedures, equipment maintenance, and
quality control test methods. Our documentation systems comply with appropriate FDA and
International Organization for Standards (ISO) requirements.
Research and Development
We are committed to developing new products and improving our current products to provide
physicians and patients with better clinical outcomes through less invasive and more efficiently
delivered therapies. Most of our research and development activities are conducted in our Minnesota
and California facilities, although we also work with physicians, research hospitals, and
universities around the world. Many of the ideas for new and improved products come from a global
network of leading physicians, who work with us in evaluating new concepts and in conducting
clinical trials to gain regulatory approvals. We conduct applied research in areas that we think
will likely lead to product commercialization activities. This research is often done at a
technology platform level such that the science can be utilized to develop a number of different
products. The development process for any new product can range from several months to several
years, primarily depending on the regulatory pathway required for approval.
5
Table of Contents
Our product development engineers work closely with their marketing partners to identify important
needs in the urology, gynecology, urogynecology and colorectal markets. The team then analyzes
the opportunities to optimize the value of the product development portfolio. During 2009, our
product development teams continued to improve our current product lines and develop new products
to increase our market share and also expand the markets we serve. In addition, we believe our
clinical data will continue to drive market expansion for our therapies and demonstrates our
technology leadership position. The following two items are notable examples of new product
development and the importance of clinical data. In mid-2009, we had our successful launch of the
new Elevate® anterior product for treating pelvic floor repair that led our women’s health sales
growth in the second half of the year, and in July 2009, the FDA approved our InhibiZone
® anti-infection claim indicating we have the only inflatable penile prosthesis with
clinical evidence showing a significant reduction in the rate of revision surgery due to
infection.
Our spending on research and development activities, including clinical and regulatory work totaled
$52.8 million, $46.2 million and $43.3 million in 2009, 2008 and 2007, respectively. These research
and development dollars represented 10.2 percent, 9.2 percent and 9.3 percent of sales for each
year respectively. We plan to target research and development spending at approximately 10 percent
of sales for the foreseeable future.
Competition
Competition in the medical device industry is intense and characterized by extensive research
efforts and rapid technological progress. The primary competitive factors include clinical
outcomes, distribution capabilities, and price relative to (1) competitive technologies and (2)
reimbursements to physicians and hospitals for their services. With certain of our products, our
competitors may have greater resources with which to develop and market products, broader
distribution resources, and scale economies which we do not have. Our competitive advantage is
driven by our focus on the pelvic health market and our ability to develop new products and
innovative procedures, obtain regulatory clearance, ensure regulatory compliance, protect our
intellectual property, protect the proprietary technology of our products and manufacturing
processes and maintain and develop preference for our products among physicians and patients. All
of these abilities require recruiting, retaining, and developing skilled and dedicated employees,
and maintaining and developing excellent relationships with physicians and suppliers.
Intellectual Property
We rely on intellectual property including patents, trade secrets, technical innovations, and
various licensing agreements to protect and build our competitive position. We own 268 issued U.S.
patents, with approximately half of such patents being issued or acquired in the last four years,
and numerous international patents covering various aspects of our technology. We also have U.S.
and international patent applications pending. We review competitive products and patents to
actively enforce our rights and to avoid infringing the legitimate rights of others.
We file patent applications to protect technology, inventions, and improvements that we consider
important, but we cannot ensure our applications will be granted, or that, if granted, the patents
will provide broad protection for our products, or that our competitors will not challenge or
circumvent these patent rights. Costs to defend our patents or to protect our activities from the
patent claims of others could be substantial, even if we are successful in defending the claims. We
do not believe that any of our products infringe any valid claims of patents or other proprietary
rights held by others.
Government Regulation
Numerous governmental authorities, principally the FDA and comparable foreign regulatory agencies,
regulate the development, testing, manufacturing, labeling, marketing, and distribution of our
products. In Europe and certain other countries, we comply with the European Union Directives for
Medical Devices and certify our compliance with the CE Mark. In other countries outside the United
States, we ensure appropriate registration and authorization. In the U.S., our products fall into
FDA Classes I, II, and III depending on the indications for use and the risk the products pose to
the patient. Class I includes devices with the least risk and Class III includes those with the
greatest risk.
The class to which our products are assigned determines the type of pre-marketing application
required for FDA clearance. If the product is classified as Class I or II, and if it is not exempt,
a 510(k) will be required to obtain
6
Table of Contents
marketing clearance. It generally takes several months from the date of most 510(k) submissions to
obtain clearance, and it may take longer, particularly if a clinical trial is required. Class III
devices generally require a pre-market approval application (PMA) supported by investigational
clinical studies. The PMA process can be expensive, uncertain, require detailed and comprehensive
data, and generally takes significantly longer than the 510(k) process.
If human clinical trials of a device are required, either for a 510(k) submission or a PMA, the
sponsor of the trial, usually the manufacturer or the distributor of the device, must file an
investigational device exemption (IDE) application prior to commencing human clinical trials. The
FDA may not approve the IDE and, even if it is approved, the FDA may not accept that the data
derived from the studies supports the safety and efficacy of the device or warrants the
continuation of clinical trials.
Our penile implant, artificial urinary and bowel sphincters, UroLume® endoprosthesis
stent, and TherMatrx® products have been approved through the Product Development
Protocol (PDP) or PMA process. Our other products were approved through the 510(k) pre-market
notification process. We have conducted clinical trials to support our PDP and PMA regulatory
approvals.
The FDA and international regulatory authorities also periodically inspect our operations to assure
themselves of our compliance with applicable quality system regulations. We must comply with a host
of regulatory requirements that apply to medical devices and drug device combination products
marketed worldwide. If we fail to comply with these regulatory requirements, our business,
financial condition, and results of operations could be significantly harmed.
Third-Party Reimbursement
Most of our products are purchased by hospitals which are reimbursed for their services by
third-party payers including Medicare, Medicaid, comparable foreign agencies, private health care
insurance, and managed care plans. The reimbursement environment facing our customers varies
widely, as do our customers’ systems for dealing with such variation.
Many third-party payers (including Medicare, Medicaid, and other large, influential payers) at
times seek to reduce their costs by denying coverage for certain procedures, including new
procedures for which efficacy has not yet been well established, or are reimbursing at rates which
do not cover the full cost of procedures. These activities may be particularly detrimental to us
because we are developing new products for new procedures. These new products and procedures may
not find market acceptance because of delays in third-party payer acceptance of the medical value
of the new procedures.
The level of third party reimbursement has fluctuated from time to time in the past, may fluctuate
in the future, and is subject to review or withdrawal at any time. The level of reimbursement may
influence whether customers purchase our products. Reimbursement rates vary depending on whether
the procedure is performed in a hospital, ambulatory surgery center or physician office. While our
sales history of devices in the U.S. does not reflect an obvious correlation between sales levels
and changes in Centers for Medicare & Medicaid Services (CMS) reimbursement rates, office-based
business may be more directly impacted by reimbursement rate fluctuations than our hospital-based
business has been historically.
Employees
As of January 2, 2010, we employed 1,221 people in the following areas: 334 in manufacturing; 366
in U.S. sales, marketing and distribution; 146 in administration; 107 in regulatory, clinical and
quality assurance; 90 in research and development; and 178 internationally. We do not have any
organized labor unions. We believe we have an excellent relationship with our employees.
Accounting Periods
We have a 52-or 53-week fiscal year ending on the Saturday nearest December 31. Accordingly,
fiscal years 2009, 2008 and 2007 ended on January 2, 2010, January 3, 2009 and December 29, 2007,
respectively, and are identified in this report as 2009, 2008 and 2007. Fiscal year 2008 had 53
weeks and fiscal years 2009 and 2007 consisted of 52 weeks.
7
Table of Contents
Financial Information about Geographic Areas
Approximately 28.0 percent, 29.1 percent, and 28.0 percent of our consolidated revenues in 2009,
2008, and 2007, respectively, were from sales to customers outside of the United States. See Notes
to Consolidated Financial Statements — No. 12, Industry Segment Information and Foreign Operations
for more information.
Executive Officers of American Medical Systems Holdings, Inc.
The persons listed below are our current executive officers. Our executive officers are elected
annually. There is no family relationship among any of the directors or executive officers, and
no executive officer has been involved during the past ten years in any legal proceedings described
in applicable Securities and Exchange Commission Regulations.
| Name | Age | Title | ||||
Anthony P. Bihl, III
|
53 | President and Chief Executive Officer | ||||
Mark A. Heggestad
|
51 | Executive Vice President and Chief Financial Officer | ||||
Whitney D. Erickson
|
43 | Vice President, General Manager Men’s Health | ||||
Maximillian D. Fiore
|
55 | Senior Vice President and Chief Technology Officer | ||||
Jeanne M. Forneris
|
56 | Senior Vice President and General Counsel | ||||
Francois X. Georgelin
|
44 | Vice President, General Manager Europe/Middle East/Africa Region | ||||
Joe W. Martin
|
57 | Senior Vice President, General Manager BPH Therapy | ||||
John F. Nealon
|
47 | Senior Vice President, General Manager Women’s Health | ||||
Thomas K. Rasmussen
|
56 | Vice President Minnetonka Operations & Worldwide Logistics | ||||
Randall R. Ross
|
47 | Senior Vice President, Human Resources | ||||
Michael E. Ryan
|
50 | Vice President, General Manager Asia Pacific/Latin America Region | ||||
Anthony “Tony” P. Bihl, III was named Chief Executive Officer in April 2008. Mr. Bihl has over 25
years experience in financial, operational, and global management and medical instrument
manufacturing. He served as Chief Executive Officer of Siemens Medical Solutions Diagnostics
Division from January to November 2007, after its acquisition of the Diagnostics Division of Bayer
HealthCare LLC, an operating unit of Bayer AG, a chemical and pharmaceutical company, where he
served in various positions since 2000. From September 2004 to December 2006, he served as the
President of the Diagnostics Division of Bayer Healthcare, LLC. In May 2002, he became the Senior
Vice President of Business Planning and Administration (BPA), where he was responsible for the
oversight, direction and management of the various functions within BPA, including accounting,
supply chain, strategic planning, business development, information management and technology, and
the Viterion TeleHealthcare joint venture with Matsushita. From January 2000 to May 2002, he
served as the Vice President of Finance & Controlling for Bayer Diagnostics’ Laboratory Testing
segment. Prior to his time with Bayer, Mr. Bihl held various managerial positions of increasing
responsibility within finance and operations in the medical diagnostics, diagnostics imaging and
biotechnology businesses.
Mark A. Heggestad has served as our Executive Vice President and Chief Financial Officer since
December 2006. Mr. Heggestad has over 20 years of experience in financial leadership roles in the
medical device industry. From 1987 to 2006, he served in various management positions at
Medtronic, Inc., a global leader in medical technologies, including Vice President of Finance and
IT for the Cardiac Surgery Business, Vice President of
Corporate Audit & Compliance Assurance and Vice President of Corporate Finance, Assistant
Controller. Prior to 1987, Mr. Heggestad was an audit manager for KPMG, LLP.
8
Table of Contents
Whitney D. Erickson has served as our Vice President and General Manager of Men’s Health since
January 2007. Ms. Erickson has over 20 years of global experience including roles in process
technology, operations leadership, marketing, business development and general management. She was
previously with Honeywell International, where she spent 11 years, most recently as a Vice
President for Business Development from 2004 to 2007, involved in integration of various Honeywell
acquisitions. Prior to Honeywell, Ms. Erickson was with the former James River Corporation, as well
as General Electric. She has worked in a variety of industries including polymers, pharmaceutical
packaging and chemical intermediates as well as security hardware and power transformation.
Maximillian D. Fiore has served as our Senior Vice President and Chief Technology Officer since
August 2009. Mr. Fiore has over 25 years experience in engineering, R&D, quality, and
manufacturing. Most recently, he was Senior Vice President of Research, Development and Operations
from August 2005 to January 2009 at OmniSonics Medical Technologies in Wilmington, Massachusetts.
OmniSonics is a venture backed medical device company focused on developing breakthrough products
for the treatment of vascular occlusive disease. Prior to OmniSonics has was Vice President of R&D
from May 2003 to August 2005 for Smith & Nephew Endoscopy Division in Andover, MA. From 1994 until
2003 he was Vice President of Engineering at Aradigm Corporation in California. Prior to that, Mr.
Fiore was the Director of Engineering at Johnson & Johnson — Lifescan from 1990 to 1994. Prior to
that, Mr. Fiore held managerial positions at Abbott Laboratories from 1982 until 1990.
Jeanne Forneris, J.D. joined our company as our Senior Vice President and General Counsel in
February 2010. Ms. Forneris served as Vice President of Strategic Investments for the Cardiac
Rhythm Disease Management Division of Medtronic, Inc. from January 2007 to February 2010, and as
Vice President and Senior Counsel for the Cardiac Rhythm Disease Management Division of Medtronic
from July 1999 to January 2007. Prior to her time at Medtronic, Ms. Forneris served as Vice
President and General Counsel of the M. A. Mortenson Company and also worked in private practice.
Francois X. Georgelin has served as Vice President and General Manager of the Europe/Middle
East/Africa (EMEA) Region since January 2009. Mr. Georgelin has 15 years of leadership experience
in international sales and marketing. From 2006 to January 2009 he served as Vice President of
Emerging Markets, EMEA, for Gambro, a European-based health care company, with responsibility for
both direct sales as well as sales through more than 50 distributors. Prior to Gambro, Mr.
Georgelin held sales and marketing leadership positions throughout Europe at Medtronic (where he
served as the Director, Cardiac Surgery Europe West from 2003 to 2006), Johnson & Johnson (Ethicon
and Gynecare), and Air Liquide.
Joe W. Martin has served as Senior Vice President and General Manager BPH Therapy since April 2009.
Mr. Martin brings over 25 years of leadership experience in medical products, including capital
equipment and surgical products. Most recently, Mr. Martin served as the CEO from 2007 to 2009 of
Milestone Scientific, a leading provider of medical and dental solutions. Prior to Milestone
Scientific, Mr. Martin spent 13 years at Bayer Healthcare in a variety of leadership positions
including president of the diabetes care division from 2000 to 2005. Before joining Bayer
Healthcare, he held leadership positions of increasing responsibility in sales and marketing,
including international roles, during his 12 years in the diagnostics division of Abbott
Laboratories.
John F. Nealon has served as Senior Vice President and General Manager, Women’s Health since April
2008. Prior to his appointment as General Manager, Mr. Nealon served as Senior Vice President of
Business Development from April 2005 to July 2008. From January 2002 to April 2005, he served as
our Vice-President of Global Marketing. Mr. Nealon has over 15 years of experience in the medical
device industry. From 1996 to 2001, he served on the management team at Survivalink, a start-up
medical device company which developed and marketed automated external defibrillators (AEDs). His
final three years at Survivalink, he served as Vice-President, Marketing and International Sales.
In 1996, he served as Director of Product Marketing for Summit Medical. From 1989 to 1996, he
served in a variety of global product marketing roles at GE Medical Systems in the x-ray, surgery
and cardiology businesses.
Thomas K. Rasmussen has served as Vice President of the Minnetonka Operations and Worldwide
Logistics since August 2007. Mr. Rasmussen has over 32 years of experience in engineering,
quality, R&D, manufacturing and supply chain. From 1996 to 2007, Mr. Rasmussen worked for Smiths
Medical as Director of Operations — Infusion
9
Table of Contents
Systems, then Vice President Medical Delivery Operations, and finally as Vice President of the U.S.
Supply Chain. Prior to Smiths Medical he worked for the 3M Company primarily within it various
Health Care divisions in many different business functions and locations.
Randall R. Ross has served as Senior Vice President of Human Resources since June 2009. Mr. Ross
has over 20 years of experience in the field of Human Resource Management. Most recently, he was
Vice President of Human Resources from 2000 to 2009 for Best Buy, a global retailer of technology
and entertainment products and services, where he led the HR function for the company’s 4,000-plus
corporate staff and its subsidiaries. Prior to Best Buy, he had Human Resource leadership
assignments at Target Corporation and Dow Chemical Company. He also served in a number of roles at
the University of St. Thomas (Minnesota), including adjunct faculty member in the graduate program
in Human Resource Development, staff psychologist and instructor for the University’s Center for
Business Excellence. Mr. Ross is a licensed psychologist in the state of Minnesota.
Michael E. Ryan has served as our Vice President and General Manager, Asia Pacific/Latin America
Region, since January 2009. Mr. Ryan has more than 22 years of Sales and Marketing experience
within the U.S. and International AMS sales organization. Previously, Mr. Ryan served as our Vice
President, Sales and Marketing, Intercontinental from May 2007 to January 2009, and he was Area
Sales Director, responsible for the Eastern United States, from November 2005 to May 2007. He has
opened and expanded AMS direct affiliate offices around the globe and developed the extensive
international distributor sales team in Europe, Latin America and Asia Pacific.
| Item 1A. | Risk Factors |
The following risk factors should be considered carefully in connection with any evaluation of our
business, financial condition, results of operations, prospects and an investment in our common
stock. Additionally, the following risk factors could cause our actual results to materially differ
from those reflected in any forward-looking statements.
Our revenues and operating results may be negatively affected and we may not achieve future growth
projections if we fail to compete successfully against our competitors or fail to develop our
presence in new markets and technologies.
Our competitors include several large medical device manufacturers, including Johnson & Johnson,
Medtronic, Inc., C.R. Bard, Inc., Boston Scientific Corporation, Coloplast and Hologic, Inc. These
and other of our competitors may have greater resources, more widely accepted products, better
distribution channels, less invasive therapies, greater technical capabilities and stronger name
recognition than we do. This is particularly the case when we enter new markets or develop
technologies for new therapies. We expect our competitors will continue to improve their products
and develop new competing products, including less invasive or non-invasive products,
pharmaceuticals and cell or gene therapies. These new technologies and products may beat our
products to the market, be more effective than our products, render our products obsolete by
substantially reducing the prevalence of the conditions our products and therapies treat, or
provide the same benefits as our existing products at the same or lower price. We may be unable to
compete effectively with our competitors, or achieve our internally established growth targets, if
we cannot maintain our market share and introduce new alternative products, techniques, therapies
and technologies in the markets we serve.
Worldwide economic conditions may adversely affect our business, operating results and financial
condition.
We believe the worldwide economic crisis has resulted and may continue to result in some reduction
in the procedures using our products. Although a majority of our products are subject to
reimbursement from third party government and non-government entities, some procedures that use our
products can be deferred by patients. In light of the current economic conditions, patients may not
have employer-provided healthcare, be as willing to take time off from work or spend their money on
deductibles and co-payments often required in connection with the procedures that use our products.
Beyond patient demand, hospitals and clinics may be less likely to purchase capital equipment given
the current economic conditions and credit environment. In addition, economic conditions could
affect the financial strength of our vendors and their ability to fulfill their commitments to us,
and could also affect the financial strength of our customers and our ability to collect accounts
receivable. While we believe current economic conditions may have contributed to a softening in
our recent revenue growth rates, the specific impact is difficult to measure. Furthermore, we
cannot predict how these economic conditions will impact our future sales,
cost of goods sold, or bad debt expense.
10
Table of Contents
Disruptions in the global financial markets could impact the ability of our counterparties and
others to perform their obligations to us and our ability to obtain future financing.
Our credit risk consists of cash and cash equivalents, short-term investments, trade receivables,
derivative instruments, lending commitments and insurance relationships in the ordinary course of
business. We place cash, cash equivalents, short-term investments and derivative instruments with
high quality financial institutions, which we monitor regularly and take action where possible to
mitigate risk. We do not hold investments in auction rate securities, mortgage backed securities,
collateralized debt obligations, individual corporate bonds, special investment vehicles or any
other investments which are generally considered higher risk investments. The carrying value of
accounts receivable approximates fair value due to the relatively short payment terms on these
instruments, or current payment patterns of our customers. To date, all lending commitments remain
available to us, and we have not incurred any charges specific to the increased credit risks.
Insurance programs are with carriers that remain highly rated and we have no significant pending
claims. However, disruptions in the capital and credit markets could cause our counterparties and
others to breach their obligations or commitments to us under our contracts with them. While all of
our debt maturities are long-term, any debt amendment or requirement for financing in the future
could have a significant negative impact on our financing costs.
Our sales may be adversely affected if physicians do not recommend, endorse or accept our products.
We rely upon physicians to recommend, endorse and accept our products. Many of the products we
acquired or are developing are based on new treatment methods. Acceptance of our products is
dependent on educating the medical community as to the distinctive characteristics, perceived
benefits, clinical efficacy, and cost-effectiveness of our products compared to competitive
products, and on training physicians in the proper application of our products. We believe our
products address major market opportunities, but if we are unsuccessful in educating physicians
about the benefits of our products, or our products are identified in regulatory agency public
health communications, our sales and earnings could be adversely affected. In addition, most of
our products are used by physicians who are required to maintain certain levels of medical
malpractice insurance to maintain their hospital privileges. As the cost of this insurance
increases, certain physicians who have used our products to treat their patients may stop
performing surgeries or providing therapies. Unless the patients who would have been treated by
these physicians are referred to other physicians who would use our products, sales of our products
could decline.
Our growth will be slowed if new products are delayed or are not accepted.
As part of our growth strategy, we intend to introduce a number of new products and product
improvements. Product introductions depend upon a variety of factors, including timely receipt of
appropriate regulatory approvals. If we do not introduce these new products and product
improvements on schedule, for any reason, or if they are not well accepted by the market or
approved, in a timely manner or at all, by applicable regulatory authorities, our business may be
adversely affected.
Our sales could decline if our procedures are not accepted by patients.
We predominantly sell implants and therapies for surgical procedures or treatments. If patients do
not accept our products and therapies, our sales may decline. Patient acceptance of our products
and therapies depends on a number of factors, including the failure of non-invasive therapies, the
degree of invasiveness involved in the procedures using our products, the rate and severity of
complications, and other adverse side effects from the procedures using our products. Patients are
more likely to first consider non-invasive alternatives to treat their urological and related
disorders. Broader patient acceptance of alternative therapies or the introduction of new oral
medications or other less-invasive therapies could adversely affect our business.
Our products face the risk of technological obsolescence, which, if realized, could have a material
adverse effect on our business.
The medical device industry is characterized by rapid and significant technological change. We
depend on our medical device technology and products to generate revenue. Therefore, we face the
risk that third parties will succeed in developing or marketing technologies and products that are
more effective than ours or that would render
11
Table of Contents
our technology and products obsolete or noncompetitive. Additionally, new, less invasive
procedures and medications could be developed that replace or reduce the importance of current
procedures that use our products or may cause our customers to delay or defer purchasing our
products. Accordingly, our success depends in part upon our ability to respond quickly to medical
and technological changes through the development and introduction of new products. The relative
speed with which we can develop products, complete clinical testing and regulatory clearance or
approval processes, train physicians in the use of our products, gain reimbursement acceptance, and
supply commercial quantities of the products to the market are expected to be important competitive
factors. Any delays could result in a loss of market acceptance and market share. Product
development involves a high degree of risk, and we cannot provide assurance that our new product
development efforts will result in any commercially successful products.
If our strategies to improve the performance of our laser therapy business are not effective in
sustaining long-term profitability and sales growth, our business, operating results and financial
condition could suffer.
In 2009, we implemented a number of strategies to improve the profitability and sales growth of our
BPH therapy business. We completed an internal reorganization and hired a general manager for BPH
therapy, to focus dedicated resources to this part of our business. We also reorganized our sales
force, conducted sales training, implemented cost reductions, and improved the reliability of our
laser therapy products in order to increase revenues, reduce costs, and improve profitability. If
these strategies are not effective in sustaining long-term profitability and sales growth, our
business may be negatively impacted.
Changes in third party reimbursement for our products and therapies may influence our customers’
purchasing activity.
Our physician and hospital customers depend on third party government and non-government entities
around the world to reimburse them for services provided to patients. The level of such third party
reimbursement has fluctuated from time to time in the past, may fluctuate in the future, and is
subject to review or withdrawal at any time. The level of reimbursement may influence whether
customers purchase our products. Reimbursement rates vary depending on whether the procedure is
performed in a hospital, ambulatory surgery center or physician office. While our sales history of
devices in the U.S. does not reflect an obvious correlation between sales levels and changes in the
Center for Medicare and Medicaid Services, or CMS, reimbursement rates, office-based business may
be more directly impacted by reimbursement rate fluctuations than our hospital-based business has
been historically. For example, CMS is revising the methodology for calculating the physician
practice expense component of the physician fee schedule, which accounts for, among other things,
the cost of devices when a procedure is performed in a physician office or clinic. A significant
change in practice expense payment levels may play a role in physician choices. Furthermore, a
significant portion of our international sales are in Europe, where health care regulations and
reimbursement for medical devices vary significantly from country to country. This changing
environment could adversely affect our ability to sell our products in some European countries.
Additionally, as we continue to expand into new global markets, we face the potential for lengthy
reimbursement approval timeframes, process delays or a lack of transparency in certain
reimbursement approval requirements. In summary, any unfavorable change in reimbursement could
have a negative impact on our business.
The impact of healthcare reform in the U.S. on us is uncertain and may adversely affect our
business.
Recently, President Obama and members of the U.S. Congress have proposed significant reforms to the
U.S. healthcare system. Although the various proposals have received support in Congressional
committees, there still is no consensus on key issues such as whether there will be an employer
mandate for healthcare insurance or a public healthcare insurance program and how to pay for
increased costs. To the extent that the number of uninsured or underinsured patients is reduced,
demand for our products could increase. However, efforts to pay for healthcare reform through a
proposed new tax on medical device companies and efforts to contain healthcare costs, directly
through pricing or reimbursement controls, or indirectly by government-sponsored healthcare
insurance, could have a material adverse effect on our sales and results of operations.
Accordingly, the impact healthcare reform could have on our business, and on the medical device
industry as a whole, remains uncertain.
Conversion of our 2036 Notes and 2041 Notes into common stock could result in dilution to our
shareholders.
12
Table of Contents
As of January 2, 2010, we have $62.0 million in principal amount of convertible senior subordinated
notes due 2036 (2036 Notes) and 250.0 million in principal amount of convertible senior
subordinated notes due 2041 (2041 Notes). Our 2036 Notes and 2041 Notes (Convertible Notes) are
convertible, at the option of the holder, into shares of our common stock at an initial conversion
price of $19.406 per share, subject to adjustment. Upon conversion, in lieu of shares of our
common stock, for each $1,000 principal amount of Convertible Notes a holder will receive an amount
in cash equal to the lesser of (i) $1,000 or (ii) the conversion value (determined in the manner
set forth in the indenture under which the Convertible Notes were issued) of the number of shares
of our common stock as determined based on the conversion rate. If the conversion value exceeds
$1,000, we will also deliver, in addition to cash, a number of shares of our common stock equal to
the sum of the daily share amounts, as defined in the indenture. The number of shares of common
stock issuable upon conversion of the Convertible Notes increases as the market price of our common
stock increases, as described in the “Liquidity and Capital Resources” section of Part II, Item 7.
Management’s Discussion and Analysis of Financial Condition and Results of Operations. All of the
above conversion rights are subject to certain limitations imposed by our Credit Facility.
Failure to satisfy our Convertible Notes and Credit Facility obligations and maintain compliance
with the loan agreements and debt covenants could have a material adverse effect on our business.
As of January 2, 2010, we have $125.3 million outstanding under our Credit Facility as described in
Part II, Item 8, Notes to Consolidated Financial Statements — No. 7, Debt. In addition to cash
generated from operations, our Credit Facility represents our primary source of liquidity. The
Credit Facility contains various restrictive covenants, compliance with which is essential to
continued credit availability. Among the most significant of these restrictive covenants are
financial covenants which require us to maintain predetermined ratio levels related to leverage,
interest coverage, fixed charges, and a limit on capital expenditures. The covenants and
restrictions contained in the Credit Facility could limit our ability to fund our business, make
capital expenditures, and make acquisitions or other investments in the future. Failure to comply
with any of these financial and other affirmative and negative covenants, or failure to make
required payments on our Credit Facility and Convertible Notes, would constitute an event of
default under the terms of the Credit Facility and under the terms of our Convertible Notes due to
cross default provisions, entitling a majority of the bank lenders to, among other things,
terminate future credit availability under the loan agreements, and/or increase the interest rate
on outstanding debt, and/or accelerate the maturity of outstanding obligations under the loan
agreements.
Our Convertible Notes and our Credit Facility may adversely affect our business in a number of
ways, including the following:
| • | requiring us to use a substantial portion of our cash to pay principal and interest on our debt instead of utilizing those funds for other purposes such as working capital, capital expenditures, and acquisitions; |
| • | limiting our ability to obtain any necessary additional financing in the future for working capital, capital expenditures, debt service requirements, or other purposes; |
| • | decreasing our debt ratings and increasing our cost of borrowed funds; |
| • | making us more vulnerable to a downturn in our business or the economy generally; and |
| • | subjecting us to the risk of being forced to refinance at higher interest rates when these amounts are due. |
Our borrowing costs are sensitive to fluctuations in interest rates, and changes in interest rates
may affect our profitability.
Because our Credit Facility carries a floating interest rate tied to LIBOR, we are subject to
market risk exposure related to changes in interest rates. We use interest rate swaps to manage
our interest rate risk on a portion of our floating rate debt. Under these arrangements, we
effectively convert an amount of the principal balance under our Credit Facility, equal to a
notional amount agreed upon in the financial instrument, from a floating rate to a fixed rate in a
swap (or a range of floating rates should we utilize a collar or a limit on the floating rate in a
cap), for an agreed-upon period of time. There is no certainty that we will continue to enter into
financial instruments to hedge our risks in the future. Also, when we enter into a financial
arrangement for a portion of our risk, there is no certainty that it would be effective or the rate
will be at market for the entire term of the instrument. At the expiration or termination of any
such financial instrument, we are again exposed to the market risk of increases in interest rates
for our floating rate debt. We are also subject to the conditions of the debt market. Our capital
13
Table of Contents
structure includes term loans and convertible note debt which are traded in public and private
transactions with fluctuating prices. Any future amendment to our credit facility could result in
higher costs due to the trading price or market conditions at that time.
We may not be able to supply products that incorporate materials or components which are single or
sole-sourced.
Some of our products utilize raw materials or components that are either single- or sole-sourced.
These sources of supply could encounter manufacturing difficulties or may unilaterally decide to
stop supplying us because of product liability concerns or other factors. We currently rely on
single source suppliers for the silicone and fabric used in our male prostheses and for the porcine
dermis and mesh used in many of our female products. Furthermore, we use single sources for the
TherMatrx® consoles and disposables. A key component of the InteXen® and
IntePro® antibiotic technology is also procured from a single source. We rely on single
and sole source suppliers for certain components in our GreenLight HPS® system. We do
not have written agreements with certain suppliers requiring them to supply us with these raw
materials or components, and we cannot be certain that we would be able to timely or
cost-effectively replace any of these sources upon any disruption. The loss of any of these
suppliers could have a material adverse effect on our financial results in the near term, as we
would be required to qualify alternate designs or sources.
The start-up, transfer, termination or interruption of any of these relationships or products, or
the failure of our suppliers to supply product to us on a timely basis or in sufficient quantities,
would likely cause us to be unable to meet customer orders for our products and harm our reputation
with customers and our business. If we obtain a new supplier for a component, we may need to
obtain FDA approval of a PMA supplement to reflect changes in product manufacturing and the FDA may
require additional testing of any component from new suppliers prior to our use of these
components. Further, if FDA approval of a PMA supplement is required, any delays in delivery of
our product to customers would be extended and our costs associated with the change in product
manufacturing may increase.
Loss of our principal manufacturing and distribution facilities would adversely affect our
financial position.
We are currently operating with one manufacturing shift at each of our three principal locations.
In 2010, we plan to consolidate our laser therapy operations by moving the Arizona activities to
our California facility, which would result in two principal locations (Minnesota and California).
We distribute our products from one location for a given product line. Although we believe we have
adequate physical capacity to serve our business operations for the foreseeable future, and we
carry property insurance on our facilities, we do not have a back up facility, and the loss or
impairment of either of our Minnesota or California facilities would have a material adverse effect
on our sales, earnings, and financial condition.
Inadequate data submissions or clinical study results which do not support a product approval may
delay or preclude a product’s commercialization.
Regulatory authorities around the world dictate different levels of manufacturing and design
information and/or clinical data for various products and therapies in order to ensure their safety
and efficacy. In the event the data submitted is deemed inadequate or the clinical study results do
not support approval by any one or more of these regulatory authorities, a product may either not
be fit for commercialization or may require a redesign to satisfy the regulatory authorities and/or
clinical study outcomes. In addition, though a product’s clinical results may meet the regulatory
requirements for product approval and commercialization, market acceptance and adoption of the
product may not meet our expectations.
Our sale of products could be reduced if we are unable to comply with regulatory requirements or
obtain the regulatory approvals necessary to market our products in the United States and foreign
jurisdictions.
If we fail to receive regulatory approval for future products, or for modifications to the design,
labeling or indications of existing products, we will be unable to market and sell these products.
In the United States, we must obtain approval from the FDA before we can begin commercializing most
of our products. The FDA approval processes are typically lengthy and expensive, and approval is
never certain. Products distributed outside of the United States are also subject to foreign
government regulations which vary from country to country. The time required to obtain approval
from a foreign country may be longer or shorter than that required for FDA approval. In addition,
we are required to comply with medical device reporting regulations, which require us to report to
FDA or
14
Table of Contents
similar governmental bodies in other countries when our products cause or contribute to a death or
serious injury or malfunction in a way that would be reasonably likely to contribute to death or
serious injury if the malfunction were to recur. FDA disagreement with the approach taken to
comply with regulatory requirements or our failure to obtain the necessary product approvals could
result in government authorities:
| • | imposing fines and penalties on us; |
| • | preventing us from manufacturing or distributing our products; |
| • | bringing civil or criminal charges against us; |
| • | delaying the introduction or denying marketing approval of our new products; |
| • | recalling, withdrawing, or seizing our products; and |
| • | requiring additional regulatory filings and/or approvals. |
In the event we fail to comply with manufacturing regulations, we could be prevented from selling
our products.
In order to commercially manufacture our products, we must comply with the FDA’s and other
authorities’ manufacturing regulations which govern design controls, quality systems, labeling
requirements and documentation policies and procedures. The FDA and foreign authorities
periodically inspect our manufacturing facilities for compliance with these requirements. Our
failure to comply with these manufacturing regulations may prevent or delay us from marketing or
distributing our products, or cause the FDA to take other enforcement actions against us which
could have a negative impact on our business.
We may experience an interruption in sales of a product and incur significant costs and negative
publicity if that product is recalled or withdrawn.
In the event that any of our products present a health hazard to the patient or physician, fail to
meet product performance criteria or specifications, including labeling, or fail to comply with
applicable laws and regulations including those administered by the FDA and foreign jurisdictions,
we could voluntarily recall or withdraw the products. The FDA and similar international regulatory
bodies have the authority to require us to recall or withdraw our products in the event of material
deficiencies or defects in design or manufacturing. A government mandated or voluntary recall or
withdrawal by us could occur as a result of unanticipated safety risks, manufacturing errors or
design defects, including defects in labeling. In addition, significant negative publicity could
result in an increased number of product liability claims, whether or not these claims are
supported by applicable law. We have initiated product recalls in the past and there is a
possibility that we may recall or withdraw products in the future and that future recalls or
withdrawals could result in significant costs to us and in significant negative publicity which
could harm our ability to market our products in the future.
Our business may suffer if our new products are not cleared to market in the United States or any
other market.
We sell some of our products only in international markets because they have not been approved for
marketing in the United States. We may be unable to sell future products in Europe, the United
States or any other market for a number of reasons. These reasons include, among others, that the
potential products could be:
| • | ineffective or cause harmful side effects during preclinical testing or clinical trials; |
| • | difficult to manufacture on a large scale; or |
| • | uneconomical for the healthcare reimbursement system. |
Should financial results severely decline, we might have to record a significant goodwill
impairment charge.
We are required to evaluate goodwill each year for impairment. If we determine the fair value is
less than the carrying value, an impairment loss will be recorded in our statement of operations.
The determination of fair value is a highly subjective exercise and can produce significantly
different results based on the assumptions used and methodologies employed. It is likely that if
our financial results were to decline substantially and if macroeconomic conditions eroded
substantially, we would have to record a non-cash goodwill impairment loss in our statement of
operations.
15
Table of Contents
We may be unable to adequately protect our intellectual property rights or obtain necessary
intellectual property rights from third parties which could adversely affect our business,
including losing market share to our competitors and the inability to operate our business
profitably.
Our success depends in part on our ability to obtain and defend patent and other intellectual
property rights that are important to the commercialization of our products and therapies. We rely
on patents, trade secrets, copyrights, know-how, trademarks, license agreements and contractual
provisions to establish our intellectual property rights and protect our products. These legal
means, however, afford only limited protection and may not adequately protect our rights. In
addition, we cannot be assured that pending patent applications will be issued. The U.S. Patent
and Trademark Office, or PTO, may deny or significantly narrow claims made under patent
applications and the issued patents, if any, may not provide us with sufficient commercial
protection. We could incur substantial costs in proceedings before the PTO. These proceedings
could result in adverse decisions as to the priority of our inventions. We cannot be sure that
patents we hold or may hold in the future will not be successfully challenged, invalidated or
circumvented in the future. Others, including our competitors, may independently develop similar
or competing technology or design around any of our patents and may have or may in the future seek
to apply for and obtain patents that may prevent, limit or interfere with our ability to make,
issue, use and sell our products and product candidates. We have not secured patent protection in
certain foreign countries in which our products are sold. The laws of some of the countries in
which our products are or may be sold may not protect our products and intellectual property to the
same extent as U.S. laws, or at all. We may be unable to protect our rights in trade secrets and
unpatented proprietary technology in these countries.
We seek to protect our trade secrets and unpatented proprietary technology, in part, with
confidentiality agreements with our employees and consultants. We cannot ensure, however, that:
| • | these agreements will not be breached; |
| • | we will have adequate remedies for any breach; or |
| • | our trade secrets will not otherwise become known to or independently developed by our competitors. |
Any disclosure of confidential information to third parties or into the public domain could allow
our competitors to use such information in competition against us. In addition, we may be subject
to damages resulting from claims that we or our employees have wrongfully used or disclosed trade
secrets or other proprietary information of their former employers.
We could incur significant costs and/or be required to stop the sale of the related product as a
result of litigation or other proceedings relating to patent and other intellectual property
rights.
Our success and competitive position depends in part on our ability to effectively prosecute claims
against others that we believe are infringing our intellectual property rights and to defend
against such claims made against us. The medical device industry is highly litigious with respect
to patents and other intellectual property rights. Companies in the medical device industry have
used intellectual property litigation to seek to gain a competitive advantage. In the future, we
may become a party to lawsuits involving patents or other intellectual property. A legal
proceeding, regardless of the outcome, would draw upon our financial resources and divert the time
and efforts of our management. If we lose one of these proceedings, a court, or a similar foreign
governing body, could require us to pay significant damages to third parties, require us to seek
licenses from third parties and pay ongoing royalties, or require us to redesign our products. If
we were unable to develop alternative technologies or acquire a license upon reasonable terms we
may be prevented from manufacturing, using or selling our products. In addition to being costly,
protracted litigation to defend or enforce our intellectual property rights could result in our
customers or potential customers deferring or limiting their purchase or use of the affected
products until the litigation is resolved.
We could incur significant costs or other negative impacts if significant product liability claims
are made against us.
The manufacture and sale of medical devices exposes us to risk of product liability claims. In the
past, and at present, we have a number of product liability claims relating to our products. In the
future, we may be subject to additional product liability claims, some of which may damage our
reputation, divert the time, attention and resources of our management, require us to pay
substantial damage awards as a result of any successful claim, or otherwise have a negative impact
on our business. As our product and therapy portfolio broadens into the treatment
16
Table of Contents
of additional medical indications, our historical product liability experience may not be a
reflection of our longer term future exposure. As a result of our exposure to product liability
claims, we currently carry product liability insurance with policy limits per occurrence and in the
aggregate that we believe to be adequate. We cannot provide assurance, however, whether this
insurance is sufficient, or if not, whether we will be able to obtain sufficient insurance to cover
the risks associated with our business or whether such insurance will be available at premiums that
are commercially reasonable. If a product liability claim or series of claims is brought against
us for uninsured liabilities or for amounts in excess of our insurance coverage, our business could
suffer.
We are required to comply with broad, pervasive and continually changing federal and state “fraud
and abuse” laws, and, if we are unable to fully comply with such laws, we could face substantial
penalties and our products could be excluded from government healthcare programs.
We are subject to various federal and state laws pertaining to healthcare fraud and abuse. These
laws, which directly or indirectly affect our ability to operate our business, include, but are not
limited to, the following:
| • | the federal Anti-Kickback Statute, which prohibits persons from knowingly and willfully soliciting, offering, receiving or providing remuneration, directly or indirectly, in cash or in kind, to induce either the referral of an individual, or the purchase, lease or order (or the arranging for or recommending of the purchase, lease or order) of a good or service, for which payment may be made under federal healthcare programs, such as the Medicare and Medicaid programs, and corresponding state laws; |
| • | the federal False Claims Act, which imposes civil and criminal liability on individuals and entities who submit, or cause to be submitted, false or fraudulent claims for payment to the government; and |
| • | the federal False Statements Statute, which prohibits knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false statement in connection with the delivery of or payment for healthcare benefits, items or services. |
In the recent past, federal and state enforcement authorities, as well as private “whistleblowers”
operating under the “qui tam” provisions of the federal False Claims Act, have sought to enforce
these laws against manufacturers of medical devices, alleging, among other things, that certain
financial relationships with physicians are not bona fide consulting or other legitimate
agreements, but are instead intended to induce those physicians to use and recommend company
products. In some instances, these actions have resulted in substantial fines, penalties, and
governmental supervision of those companies’ operations. Because our business necessitates
frequent contact with physicians and other healthcare professionals, including financial
relationships such as consulting agreements, training programs, and cooperative marketing
arrangements, we have implemented a broad-based corporate compliance program, and voluntarily
follow the AdvaMed Code of Ethics on Interactions with Health Care Professionals, in order to
maintain compliance and inform our employees regarding the foregoing laws and regulations.
However, if our past or present operations are found to be in violation of any of the laws
described above or other similar governmental regulations to which we or our customers are subject,
we or our officers may be subject to the applicable penalty associated with the violation,
including civil and criminal penalties, damages, fines, imprisonment, exclusion from the Medicare
and Medicaid programs and the curtailment or restructuring of our operations. Similarly, if the
physicians or other providers or entities with which we do business are found to be non-compliant
with applicable laws, they may be subject to sanctions, which could also have a negative impact on
us. Any action against us for violation of these laws, even if we successfully defend against it,
could cause us to incur significant legal expenses, divert our management’s attention from the
operation of our business and damage our reputation especially when considering the high public
scrutiny in this area. If enforcement action were to occur, our reputation and our business and
financial condition may be harmed, even if we were to prevail or settle the action.
Our international operations expose us to various risks, including risks related to fluctuations in
foreign currency exchange rates.
We derive a significant portion of our net sales from operations in international markets. During
fiscal 2009, 2008 and 2007, 28.0 percent, 29.1 percent and 28.0 percent, respectively, of our sales
were to customers outside the United States. Some of these sales were to governmental entities and
other organizations with extended payment terms. A number of factors, including differing economic
conditions, changes in political climate, differing tax structures, changes in diplomatic and trade
relationships, and political or economic instability in the countries where we do business, could
affect payment terms and our ability to collect foreign receivables. We have little influence over
these factors and changes could have a material adverse impact on our business. In addition,
foreign sales are
17
Table of Contents
influenced by fluctuations in currency exchange rates, primarily the Euro, Canadian dollar,
Australian dollar, and Great Britain pound. Increases in the value of the foreign currencies
relative to the U.S. dollar would positively impact our earnings and decreases in the value of the
foreign currencies relative to the U.S. dollar would negatively impact our earnings.
We use derivative instruments, such as foreign exchange forward contracts, to hedge a portion of
estimated currency exposures. The use of derivatives would only offset the portion hedged for
adverse effects of an unfavorable change in foreign currency exchange rates and would also offset a
portion of favorable movements in rates.
The risks of selling and shipping our products and of purchasing components and products
internationally may adversely impact our revenues, results of operations and financial condition.
The sale and shipping of our products and services across international borders subject us to
extensive U.S. and foreign governmental trade regulations, such as various anti-bribery laws,
including the U.S. Foreign Corrupt Practices Act, export control laws, customs and import laws, and
anti-boycott laws. Any failure to comply with applicable laws and regulations could impact us in a
variety of ways that include, but are not limited to, significant criminal, civil and
administrative penalties, including imprisonment of individuals, fines and penalties, denial of
export privileges, seizure of shipments, restrictions on certain business activities, and exclusion
or debarment from government contracting. Also, the failure to comply with applicable legal and
regulatory obligations could result in the disruption of our shipping and sales activities.
In addition, some countries in which we sell our products are, to some degree, subject to
political, economic and/or social instability. Our international sales operations expose us and our
representatives, agents and distributors to risks inherent in operating in foreign jurisdictions.
These risks include:
| • | the imposition of additional U.S. and foreign governmental controls or regulations; |
| • | the imposition of costly and lengthy new export licensing requirements; |
| • | the imposition of U.S. and/or international sanctions against a country, company, person or entity with whom the company does business that would restrict or prohibit continued business with the sanctioned country, company, person or entity; |
| • | economic instability; |
| • | changes in duties and tariffs, license obligations and other non-tariff barriers to trade; |
| • | the imposition of new trade restrictions; |
| • | the imposition of restrictions on the activities of foreign agents, representatives and distributors; |
| • | scrutiny of foreign tax authorities which could result in significant fines, penalties and additional taxes being imposed on us; |
| • | pricing pressure that we may experience internationally; |
| • | laws and business practices favoring local companies; |
| • | difficulties in enforcing or defending intellectual property rights; and |
| • | exposure to different legal and political standards due to our conducting business in several foreign countries. |
We cannot provide assurance that one or more of these factors will not harm our business. Any
material decrease in our international sales would adversely impact our revenues, results of
operations and financial condition.
| Item 1B. | Unresolved Staff Comments |
None.
| Item 2. | Properties |
Our corporate headquarters, main warehouse, research & development and manufacturing operations are
located in Minnetonka, Minnesota, consisting of 230,000 square feet. This includes 50,000 square
feet of office space in our Minnetonka facility that was added during 2007 to accommodate our
current and future expected growth. We also
18
Table of Contents
lease a manufacturing facility with approximately 20,000 square feet in Phoenix, Arizona, and three
facilities with approximately 80,000 square feet of manufacturing, research & development and
warehouse space in San Jose, California. We believe we have sufficient manufacturing space and
capacity to meet production requirements for our products for 2010.
We lease office space for our international management, sales, and administration personnel in
Australia, Brazil, Canada, France, Germany, the Netherlands, Spain and the United Kingdom.
| Item 3. | Legal Proceedings |
We have been and are currently subject to various legal proceedings that arise in the ordinary
course of business, including product liability claims and patent related issues. In May 2009, we
received a subpoena from the Office of Inspector General (OIG) of the United States Department of
Health and Human Services requesting certain documents related to specific business interactions
with one customer since January 1, 2004 pertaining to our laser therapy products used to treat BPH.
We have provided the documents requested by OIG and will continue to respond promptly to any
additional requests for information. At this time we cannot predict the timing or outcome of the
review.
| Item 4. | Submission of Matters to a Vote of Security Holders |
Not Applicable
19
Table of Contents
PART II
Item 5. Market for American Medical Systems’ Common Equity, Related Stockholder Matters and Issuer
Purchases of Equity Securities
Market Information
Our common stock is currently traded on the Nasdaq Global Select Market under the symbol AMMD. The
following table sets forth, for the periods indicated, the high and low closing sales prices per
share of our common stock as reported on the Nasdaq Global Select Market. These prices do not
include adjustments for retail mark-ups, mark-downs, or commissions.
| 2009 | 2008 | |||||||||||||||
| High | Low | High | Low | |||||||||||||
First quarter |
$ | 11.92 | $ | 8.90 | $ | 14.98 | $ | 13.20 | ||||||||
Second quarter |
$ | 16.40 | $ | 11.06 | $ | 15.87 | $ | 13.99 | ||||||||
Third quarter |
$ | 17.10 | $ | 14.23 | $ | 18.20 | $ | 13.95 | ||||||||
Fourth quarter |
$ | 19.49 | $ | 15.27 | $ | 17.93 | $ | 8.25 | ||||||||
Holders
On
February 19, 2010, there were approximately 104 stockholders of
record and approximately 17,390 beneficial stockholders.
Dividends
We have never declared or paid cash dividends. We intend to retain all future earnings for the
operation and expansion of our business. We do not anticipate declaring or paying cash dividends on
our common stock in the foreseeable future. In addition, our current Credit Facility places certain
restrictions on paying cash dividends.
Recent Sales of Unregistered Equity Securities
During the fourth quarter ended January 2, 2010, we did not issue or sell any shares of our common
stock or other equity securities of ours without registration under the Securities Act of 1933, as
amended.
Issuer Purchases of Equity Securities
We did not purchase any shares of our common stock or our other equity securities registered
pursuant to Section 12 of the Securities Exchange Act of 1934, as amended, during the fourth
quarter ended January 2, 2010.
20
Table of Contents
Stock Performance Graph
The following graph compares the annual cumulative total stockholder return on our common stock
from December 31, 2004 until December 31, 2009, with the annual cumulative total return over the
same period of the Nasdaq Market Value Index and the Medical Appliances & Equipment Index.
Morningstar prepared the data points.
The comparison assumes the investment of $100 in each of our common stock, the Nasdaq Market Value
Index and the Comparable Company Index on December 31, 2004, and the reinvestment of all dividends.
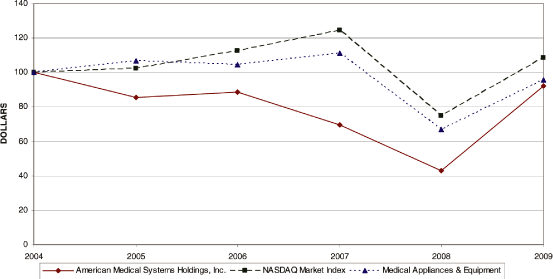
| 12/31/2004 | 12/30/2005 | 12/29/2006 | 12/28/2007 | 1/2/2009 | 12/31/2009 | |||||||||||||||||||
AMS Common Stock |
$ | 100.00 | $ | 85.29 | $ | 88.59 | $ | 69.46 | $ | 42.76 | $ | 92.27 | ||||||||||||
NASDAQ Market Index |
$ | 100.00 | $ | 102.20 | $ | 112.68 | $ | 124.57 | $ | 74.71 | $ | 108.56 | ||||||||||||
Medical Appliances
& Equipment |
$ | 100.00 | $ | 106.64 | $ | 104.77 | $ | 111.07 | $ | 67.07 | $ | 95.83 | ||||||||||||
21
Table of Contents
| Item 6. | Selected Financial Data |
The following tables present five years of data (in thousands) from our statement of operations and
balance sheet.
| Statement of Operations Data | 2009 | 2008 | 2007 | 2006 | 2005 | |||||||||||||||
Net sales |
$ | 519,270 | $ | 501,641 | $ | 463,928 | $ | 358,318 | $ | 262,591 | ||||||||||
Cost of sales |
92,211 | 111,097 | 105,592 | 68,872 | 46,111 | |||||||||||||||
Gross profit |
427,059 | 390,544 | 358,336 | 289,446 | 216,480 | |||||||||||||||
Operating expenses |
||||||||||||||||||||
Marketing and selling |
173,854 | 175,670 | 169,495 | 123,204 | 92,001 | |||||||||||||||
Research and development |
52,765 | 46,247 | 43,315 | 33,877 | 20,966 | |||||||||||||||
In-process research and development (1) |
— | 7,500 | 7,500 | 94,035 | 9,220 | |||||||||||||||
General and administrative |
45,196 | 39,281 | 43,070 | 34,417 | 21,713 | |||||||||||||||
Integration costs (2) |
— | — | 1,103 | 1,712 | — | |||||||||||||||
Litigation settlement (3) |
— | — | 14,303 | — | — | |||||||||||||||
Amortization of intangibles (4) |
13,161 | 34,465 | 18,264 | 12,393 | 7,884 | |||||||||||||||
Total operating expenses |
284,976 | 303,163 | 297,050 | 299,638 | 151,784 | |||||||||||||||
Operating income (expense) |
142,083 | 87,381 | 61,286 | (10,192 | ) | 64,696 | ||||||||||||||
Other (expense) income |
||||||||||||||||||||
Royalty and other |
1,632 | 2,279 | 8,099 | 1,984 | 500 | |||||||||||||||
Interest income |
175 | 747 | 1,153 | 2,754 | 1,246 | |||||||||||||||
Interest expense (5) |
(19,636 | ) | (27,398 | ) | (37,760 | ) | (18,395 | ) | (217 | ) | ||||||||||
Amortization of financing costs (6) |
(15,790 | ) | (18,482 | ) | (16,145 | ) | (14,434 | ) | — | |||||||||||
Gain on extinguishment of debt (7) |
10,125 | 5,631 | — | — | — | |||||||||||||||
Gain on sale of non-strategic assets (8) |
17,446 | — | — | — | — | |||||||||||||||
| — — — - | ||||||||||||||||||||
Total other (expense) income |
(6,048 | ) | (37,223 | ) | (44,653 | ) | (28,091 | ) | 1,529 | |||||||||||
Income (loss) from continuing operations
before income taxes |
136,035 | 50,158 | 16,633 | (38,283 | ) | 66,225 | ||||||||||||||
Provision for income taxes (9) |
51,197 | 19,323 | 11,888 | 9,469 | 26,950 | |||||||||||||||
Net income (loss) from continuing operations |
84,838 | 30,835 | 4,745 | (47,752 | ) | 39,275 | ||||||||||||||
Loss from discontinued operations, net of tax
benefit of $0.4 million and $2.7 million for 2007
and 2006, respectively (10) |
— | — | (691 | ) | (5,435 | ) | — | |||||||||||||
Net income (loss) |
$ | 84,838 | $ | 30,835 | $ | 4,054 | $ | (53,187 | ) | $ | 39,275 | |||||||||
Net income (loss) per share |
||||||||||||||||||||
Basic net income (loss) from continuing
operations |
$ | 1.14 | $ | 0.42 | $ | 0.07 | $ | (0.68 | ) | $ | 0.57 | |||||||||
Discontinued operations, net of tax |
— | — | (0.01 | ) | (0.08 | ) | — | |||||||||||||
Basic net income (loss) |
$ | 1.14 | $ | 0.42 | $ | 0.06 | $ | (0.76 | ) | $ | 0.57 | |||||||||
Diluted net income (loss) from
continuing operations |
$ | 1.14 | $ | 0.42 | $ | 0.06 | $ | (0.68 | ) | $ | 0.55 | |||||||||
Discontinued operations, net of tax |
— | — | (0.01 | ) | (0.08 | ) | — | |||||||||||||
Diluted net income (loss) |
$ | 1.14 | $ | 0.42 | $ | 0.06 | $ | (0.76 | ) | $ | 0.55 | |||||||||
Balance Sheet Data |
2009 | 2008 | 2007 | 2006 | 2005 | |||||||||||||||
Cash, cash equivalents, and short-term investments |
$ | 50,538 | $ | 42,965 | $ | 35,181 | $ | 29,541 | $ | 46,390 | ||||||||||
Working capital |
133,076 | 130,999 | 143,298 | 135,635 | 69,533 | |||||||||||||||
Total assets |
1,047,151 | 1,044,497 | 1,115,868 | 1,126,620 | 359,326 | |||||||||||||||
Long-term debt |
346,229 | 484,582 | 577,096 | 611,539 | — | |||||||||||||||
Stockholders’ equity |
545,359 | 427,482 | 383,371 | 345,189 | 302,879 | |||||||||||||||
22
Table of Contents
| (1) | In 2008 and 2007, we recognized $7.5 million each year for in-process research and development charges related to the payments for achieving certain milestones related to our BioControl acquisition. In 2006, we recognized $25.6 million, $2.1 million, $62.1 million and $4.3 million, respectively, for in-process research and development charges related to the acquisitions of BioControl, Solarant, Laserscope and Ovion. In 2005, we recognized in-process research and development charges of $9.2 million related to the acquisition of Ovion. | |
| (2) | In 2007 and 2006, we recorded $1.1 million and $1.7 million, respectively, of integration costs associated with the 2006 Laserscope acquisition, primarily related to travel, legal, consulting and retention bonuses. | |
| (3) | During 2007, we recorded a charge of $14.3 million for litigation settlements, primarily for the arbitration award to the former shareholders of CryoGen, Inc. (CryoGen) concerning an earnout payment related to our 2002 acquisition of CryoGen. See Notes to Consolidated Financial Statements — No. 3, Litigation Settlements for more information. | |
| (4) | In 2008, we recorded additional amortization expense of $17.1 million for the acceleration of amortization to adjust the carrying value of certain intangible assets to their current fair values. For more information regarding this acceleration of of amortization, see Notes to Consolidated Financial Statements — No. 5, Goodwill and Intangible Assets. | |
| (5) | During 2009, interest expense included interest incurred on our 2036 Notes and 2041 Notes. During 2008, 2007 and 2006, interest expense included interest incurred on our 2036 Notes. In 2009, average borrowings under the 2036 Notes and 2041 Notes were $245.6 million and $71.4 million, respectively. Average borrowings under the 2036 Notes were $371.1 million during 2008, and $373.8 million during 2007 and 2006. Interest expense also included interest incurred on our senior secured credit facility entered into on July 20, 2006. Our average borrowings under this facility were approximately $191.8 million, $281.7 million and $332.3 million during 2009, 2008 and 2007, respectively, and $366.0 million from inception through December 30, 2006. We also incurred interest expense related to short-term borrowing activity during 2006. For a more complete description of these items, see Notes to Consolidated Financial Statements — No. 7, Debt. | |
| (6) | Amortization of financing costs relates to the deferred financing costs and debt discount for our convertible notes and our senior secured credit facility. Charges during 2006 also include a $7.0 million commitment fee for a bridge loan of up to $180 million in preparation for the acquisition of Laserscope. We did not use this financing for the Laserscope acquisition. See Notes to Consolidated Financial Statements — No. 7, Debt for more information. | |
| (7) | In 2009, we recognized a $10.1 million gain on extinguishment of debt related to our extinguishment of $27.3 million of 2036 Notes in the first quarter and our exchange of $250.0 million of 2036 Notes for 2041 Notes in the third quarter. During the fourth quarter of 2008, we recognized a $5.6 million gain on extinguishment of $34.5 million of convertible notes. See Notes to Consolidated Financial Statements — No. 7, Debt for more information. | |
| (8) | During the third quarter of 2009, we recognized a $17.4 million gain related to the sale of our Ovion technology. See Notes to Consolidated Financial Statements — No. 5, Goodwill and Intangible Assets for more information. | |
| (9) | In 2007, we experienced adverse tax effects from the $14.3 million of litigation settlement charges primarily resulting from the resolution of the CryoGen arbitration (see Notes to Consolidated Financial Statements — No. 3, Litigation Settlements). Partially offsetting this unfavorable impact was the favorable settlement of a tax audit for $0.9 million, which allowed us to release a reserve for uncertain tax benefits. The in-process research and development charges described above for 2005 and 2006 have no related tax benefit, except for BioControl. In 2006, we received a $2.4 million tax refund associated with the favorable agreement reached with the IRS involving the review of our 2001 and 2002 federal income tax returns. | |
| (10) | In conjunction with our acquisition of Laserscope in the third quarter of 2006, we committed to a plan to divest Laserscope’s aesthetics business. On January 16, 2007, we sold the aesthetics business to Iridex Corporation. The financial results of the aesthetics business have been reported as discontinued operations beginning from the date of acquisition of July 20, 2006 through the date of sale of January 16, 2007. The income tax benefit from the loss from discontinued operations was $0.4 million and $2.7 million in 2007 and 2006, respectively. For a more complete description of the discontinued operations and the related impact on our financial results, refer to Notes to Consolidated Financial Statements — No. 2, Discontinued Operations and Sale of Aesthetics Business. |
23
Table of Contents
| Item 7. | Management’s Discussion and Analysis of Financial Condition and Results of Operations |
Introductory Overview
We are a world leader in developing and delivering innovative medical technology solutions to
physicians treating men’s and women’s pelvic health conditions, thereby recognized as a technology
leader in the markets we serve. We have built a business that delivers growth, fueled by a robust
pipeline of innovative products for significant, under-penetrated markets. We have a diverse
product portfolio, which treats men’s incontinence, erectile dysfunction, and benign prostatic
hyperplasia (BPH), and treats women’s incontinence and pelvic floor prolapse. We estimate there are
as many as 1.6 billion incidences of these conditions in the global markets we serve, with many
people suffering from multiple conditions. Treatment options for these conditions vary considerably
depending on the severity of the condition. Approximately 350 million of these men and women have
conditions sufficiently severe so as to profoundly diminish their quality of life and significantly
impact their relationships. Our addressable market is contained within this group of patients. Our
product development and acquisition strategies have focused on expanding our product offering for
surgical solutions, including less-invasive solutions for surgeons and their patients. Our primary
physician customers include urologists, gynecologists, urogynecologists and colorectal surgeons.
We completed our 37th year of operations in 2009, with a continued focus on
technological innovation, market expansion and financial strength.
Our net sales grew from $501.6 million in 2008 to $519.3 million in 2009. In 2009, men’s health
contributed $234.6 million, or 45 percent of total net sales, BPH therapy contributed $114.5
million, or 22 percent of total net sales, and women’s health contributed $170.2 million, or 33
percent of total net sales.
Our men’s health products for treating incontinence and erectile restoration, and our women’s
health products for treating pelvic floor repair, were key drivers in revenue growth in 2009. In
particular, the AMS 800 ® (our artificial urinary sphincter product) and the AMS
700 ® (our erectile restoration product), led our sales growth in men’s health, and the
successful launch in mid-2009 of the new Elevate® anterior product for pelvic floor
repair led our women’s health sales growth in the second half of the year. Our 2009 sales saw a
modest decline over 2008 in our BPH therapy products, which is our full line of products to deliver
minimally invasive procedures for the treatment of obstructive BPH and urinary stones. However, we
achieved accelerating sales growth in our BPH therapy product line each sequential quarter
throughout 2009, and 2009 sales of fibers used by the GreenLight HPS ® (High Performance
System) laser system exceeded 2008 fiber sales. Our women’s health products for treating
incontinence had modest growth in 2009, driven primarily by solid sales growth of MiniArc
®.
In 2009, we continued to focus on initiatives which reduce working capital, manage expenses and
drive operating leverage throughout our business. We generated cash from operating activities of
$122.8 million in 2009, compared to $115.8 million in 2008. We also reduced our debt by $130.8
million in 2009, compared to $119.7 million in 2008. We earned net income of $84.8 million in 2009,
compared to $30.8 million in 2008.
In January of 2007, shortly after the acquisition of Laserscope, we sold the aesthetics portion of
that business. All of the information in this report, unless specifically stated otherwise,
excludes the Laserscope aesthetics business, which we reported as discontinued operations during
the six month period, July 2006 to January 2007, during which we held this business.
24
Table of Contents
Results of Operations
Sales trends
The following table compares net sales of our product lines and geographies between 2009 and 2008,
and between 2008 and 2007.
| (in thousands) | 2009 | 2008 | $ Increase | % Increase | 2008 | 2007 | $ Increase | % Increase | ||||||||||||||||||||||||
Net Sales |
||||||||||||||||||||||||||||||||
Product Line |
||||||||||||||||||||||||||||||||
Men’s health |
$ | 234,594 | $ | 219,211 | $ | 15,383 | 7.0 | % | $ | 219,211 | $ | 188,519 | $ | 30,692 | 16.3 | % | ||||||||||||||||
BPH therapy |
114,468 | 116,346 | (1,878 | ) | -1.6 | % | 116,346 | 125,497 | (9,151 | ) | -7.3 | % | ||||||||||||||||||||
Women’s health |
170,208 | 166,084 | 4,124 | 2.5 | % | 166,084 | 149,912 | 16,172 | 10.8 | % | ||||||||||||||||||||||
Total |
$ | 519,270 | $ | 501,641 | $ | 17,629 | 3.5 | % | $ | 501,641 | $ | 463,928 | $ | 37,713 | 8.1 | % | ||||||||||||||||
Geography |
||||||||||||||||||||||||||||||||
United States |
$ | 373,898 | $ | 355,678 | $ | 18,220 | 5.1 | % | $ | 355,678 | $ | 334,258 | $ | 21,420 | 6.4 | % | ||||||||||||||||
International |
145,372 | 145,963 | (591 | ) | -0.4 | % | 145,963 | 129,670 | 16,293 | 12.6 | % | |||||||||||||||||||||
Total |
$ | 519,270 | $ | 501,641 | $ | 17,629 | 3.5 | % | $ | 501,641 | $ | 463,928 | $ | 37,713 | 8.1 | % | ||||||||||||||||
Percent of net sales |
||||||||||||||||||||||||||||||||
Product Line |
||||||||||||||||||||||||||||||||
Men’s health |
45.2 | % | 43.7 | % | 43.7 | % | 40.6 | % | ||||||||||||||||||||||||
BPH therapy |
22.0 | % | 23.2 | % | 23.2 | % | 27.1 | % | ||||||||||||||||||||||||
Women’s health |
32.8 | % | 33.1 | % | 33.1 | % | 32.3 | % | ||||||||||||||||||||||||
Total |
100.0 | % | 100.0 | % | 100.0 | % | 100.0 | % | ||||||||||||||||||||||||
Geography |
||||||||||||||||||||||||||||||||
United States |
72.0 | % | 70.9 | % | 70.9 | % | 72.0 | % | ||||||||||||||||||||||||
International |
28.0 | % | 29.1 | % | 29.1 | % | 28.0 | % | ||||||||||||||||||||||||
Total |
100.0 | % | 100.0 | % | 100.0 | % | 100.0 | % | ||||||||||||||||||||||||
Net Sales. In 2009, net sales increased by 3.5 percent or $17.6 million over 2008. The
strengthening of the U.S. dollar in 2009 compared to 2008 reduced revenue growth approximately 1.7
percentage points or $8.2 million. Sales growth in 2009 was led by growth from the
Elevate® anterior and posterior transvaginal pelvic floor repair systems (we launched
Elevate® anterior in 2009 and Elevate® posterior in 2008), the
MiniArc® Single-Incision Sling for treating female incontinence, GreenLight™
laser fibers for treating BPH and our erectile restoration product line. This growth has
been partially offset by declines in our BPH therapy capital equipment sales and our uterine health
product line sales. We believe the worldwide economic downturn may have contributed to a softening
in our recent revenue growth rates; however, the specific impact is difficult to measure. Although
a majority of our products are subject to reimbursement from third party government and
non-government entities, some procedures that use our products can be deferred by patients. In
light of worldwide economic conditions, patients may not have employer-provided healthcare, be as
willing to take time off from work or spend their money on deductibles and co-payments often
required in connection with the procedures that use our products.
In 2008, net sales increased by 8.1 percent or $37.7 million over 2007 due to the success of
product launches, particularly the AdVance® male sling for treating mild male
incontinence, the MiniArc® Single-Incision Sling, and the InhibiZone®-coated
AMS 800® Artificial Urinary Sphincter, along with launches of products outside the
United
States, specifically the AMS 700® MS™ for erectile restoration. In addition,
the weakening of the U.S. dollar in 2008 compared to 2007 increased revenue growth approximately
1.0 percentage point or $4.9 million.
Men’s health products. Net sales from men’s health products grew 7.0 percent or $15.4 million over
2008. This includes the negative impact of foreign currency fluctuations of approximately 1.9
percentage points or $4.2 million.
In 2009, we experienced balanced growth from both the male continence and erectile restoration
product lines. The increase in male continence was mainly due to higher volume, while the increase
in the erectile restoration product line is the result of several factors, including: U.S. Food and
Drug Administration (FDA) approval in July 2009 of our InhibiZone® anti-infection claim
indicating we have the only inflatable penile prosthesis with clinical evidence
25
Table of Contents
showing a
significant reduction in the rate of revision surgery due to infection; our patient education and
outreach programs in the U.S.; and the result of shifts in product mix.
The largest portion of the increase in 2008 over 2007 is in the male continence product line,
driven by the continued success of the AdVance® male sling and the AMS 800®
with InhibiZone®. We also saw growth in sales of our erectile restoration products.
BPH therapy products. Net sales from BPH therapy products declined 1.6 percent or $1.9 million
compared to 2008. This includes the negative impact of foreign currency fluctuations of
approximately 1.4 percentage points or $1.6 million.
We are continuing to see the benefits of initiatives started in early 2009 related to our BPH
product line: we created a separate business unit and hired a general manager to lead it, increased
our U.S. sales force by approximately 50%, and implemented comprehensive sales training, which
resulted in double digit sales growth in U.S. GreenLight™ fiber sales in 2009 compared
to 2008. The increase in fiber sales was offset by a decline in laser console sales, as this area
of our business is more directly impacted by economic pressures on hospital capital purchases. We
also experienced a decline in sales of our TherMatrx® product line, which is used for
treatment of non-obstructive BPH.
The decline in 2008 compared to 2007 was driven by lower sales of our laser therapy products, where
we did not achieve anticipated utilization rates resulting from a slowing in the surgical markets
likely due to the increased use of drugs to treat BPH as well as increased competition. Our
TherMatrx® product line sales also declined due to lower than expected adoption rates
for this office-based procedure.
Women’s health products. Net sales from women’s health products grew 2.5 percent or $4.1 million
over 2008. This includes the negative impact of foreign currency fluctuations of approximately 1.4
percentage points or $2.4 million.
In 2009, we experienced strong growth from the Elevate® posterior and anterior
transvaginal pelvic floor repair systems, which drove growth in the pelvic floor repair product
line. The female continence product line experienced a slight increase compared to 2008, as the
MiniArc® sling contributed strong growth over 2008. The Her Option® product
line for the treatment of menorrhagia, or excessive uterine bleeding, experienced a decline in
revenues and units in 2009. We divested the Her Option® product line on February 16,
2010. See Notes to Consolidated Financial Statements — No. 16, Subsequent Events.
The growth in 2008 was largely due to the female continence product line, driven by the
MiniArc® sling that was introduced in 2007 as it contributed strong growth in dollars
and units. The Her Option® product line experienced a decline in revenues and units
compared to the prior year. Revenue growth in this area was impacted by lower than expected
adoption rates for this office-based procedure.
International sales and foreign exchange effects. Our international net sales decreased $0.6
million, or 0.4 percent in 2009 compared to 2008, largely as a result of the negative impact of
approximately $8.2 million in foreign currency exchange rate changes, with the strengthening of the
U.S. dollar. The negative foreign currency impact was offset by sales growth of $7.6 million,
which was led by our male continence product line. Because a portion of the expenses associated
with international sales are foreign currency denominated costs, changes in currency rates do not
affect net income and cash flows from operations by the same dollar amount as they affect net
sales.
26
Table of Contents
Our international net sales grew $16.3 million, or 12.6 percent in 2008 from 2007. Of this
growth, $4.9 million, or 3.7 percentage points, was due to favorable currency exchange rates in the
markets in which we conduct business in a foreign currency.
| Customer location | 2009 | 2008 | $ Increase | % Increase | 2008 | 2007 | $ Increase | % Increase | ||||||||||||||||||||||||
Within U.S. |
$ | 373,898 | $ | 355,678 | $ | 18,220 | 5.1 | % | $ | 355,678 | $ | 334,258 | $ | 21,420 | 6.4 | % | ||||||||||||||||
International |
||||||||||||||||||||||||||||||||
Before currency impact |
153,578 | 145,963 | 7,615 | 5.2 | % | 141,109 | 129,670 | 11,439 | 8.8 | % | ||||||||||||||||||||||
Subtotal |
527,476 | 501,641 | 25,835 | 5.2 | % | 496,787 | 463,928 | 32,859 | 7.1 | % | ||||||||||||||||||||||
Currency impact |
(8,206 | ) | — | (8,206 | ) | — | 4,854 | — | 4,854 | — | ||||||||||||||||||||||
Total |
$ | 519,270 | $ | 501,641 | $ | 17,629 | 3.5 | % | $ | 501,641 | $ | 463,928 | $ | 37,713 | 8.1 | % | ||||||||||||||||
Operating Expenses
The following table compares the dollar and percentage change in the Consolidated Statement of
Operations between 2009 and 2008, and between 2008 and 2007.
| $ Increase | % | $ Increase | % | |||||||||||||||||||||||||||||
| (in thousands) | 2009 | 2008 | (Decrease) | Change | 2008 | 2007 | (Decrease) | Change | ||||||||||||||||||||||||
Net sales |
$ | 519,270 | $ | 501,641 | $ | 17,629 | 3.5 | % | $ | 501,641 | $ | 463,928 | $ | 37,713 | 8.1 | % | ||||||||||||||||
Cost of sales |
92,211 | 111,097 | (18,886 | ) | -17.0 | % | 111,097 | 105,592 | 5,505 | 5.2 | % | |||||||||||||||||||||
Gross profit |
427,059 | 390,544 | 36,515 | 9.3 | % | 390,544 | 358,336 | 32,208 | 9.0 | % | ||||||||||||||||||||||
Operating expenses |
||||||||||||||||||||||||||||||||
Marketing and selling |
173,854 | 175,670 | (1,816 | ) | -1.0 | % | 175,670 | 169,495 | 6,175 | 3.6 | % | |||||||||||||||||||||
Research and development |
52,765 | 46,247 | 6,518 | 14.1 | % | 46,247 | 43,315 | 2,932 | 6.8 | % | ||||||||||||||||||||||
In-process research & development |
— | 7,500 | (7,500 | ) | n/a | 7,500 | 7,500 | — | 0.0 | % | ||||||||||||||||||||||
General and administrative |
45,196 | 39,281 | 5,915 | 15.1 | % | 39,281 | 43,070 | (3,789 | ) | -8.8 | % | |||||||||||||||||||||
Integration costs |
— | — | — | n/a | — | 1,103 | (1,103 | ) | n/a | |||||||||||||||||||||||
Litigation settlement |
— | — | — | n/a | — | 14,303 | (14,303 | ) | n/a | |||||||||||||||||||||||
Amortization of intangibles |
13,161 | 34,465 | (21,304 | ) | -61.8 | % | 34,465 | 18,264 | 16,201 | 88.7 | % | |||||||||||||||||||||
Total operating expenses |
284,976 | 303,163 | (18,187 | ) | -6.0 | % | 303,163 | 297,050 | 6,113 | 2.1 | % | |||||||||||||||||||||
Operating income |
142,083 | 87,381 | 54,702 | 62.6 | % | 87,381 | 61,286 | 26,095 | 42.6 | % | ||||||||||||||||||||||
Royalty income |
3,073 | 4,474 | (1,401 | ) | -31.3 | % | 4,474 | 5,028 | (554 | ) | -11.0 | % | ||||||||||||||||||||
Interest income |
175 | 747 | (572 | ) | -76.6 | % | 747 | 1,153 | (406 | ) | -35.2 | % | ||||||||||||||||||||
Interest expense |
(19,636 | ) | (27,398 | ) | (7,762 | ) | -28.3 | % | (27,398 | ) | (37,760 | ) | (10,362 | ) | -27.4 | % | ||||||||||||||||
Amortization of financing costs |
(15,790 | ) | (18,482 | ) | (2,692 | ) | -14.6 | % | (18,482 | ) | (16,145 | ) | 2,337 | 14.5 | % | |||||||||||||||||
Gain on extinguishment of debt |
10,125 | 5,631 | 4,494 | 79.8 | % | 5,631 | — | 5,631 | n/a | |||||||||||||||||||||||
Gain on sale of non-strategic assets |
17,446 | — | 17,446 | n/a | — | — | — | n/a | ||||||||||||||||||||||||
Other (expense) income |
(1,441 | ) | (2,195 | ) | 754 | -34.4 | % | (2,195 | ) | 3,071 | (5,266 | ) | -171.5 | % | ||||||||||||||||||
Income from continuing
operations before income taxes |
136,035 | 50,158 | 85,877 | 171.2 | % | 50,158 | 16,633 | 33,525 | 201.6 | % | ||||||||||||||||||||||
Provision for income taxes |
51,197 | 19,323 | 31,874 | 165.0 | % | 19,323 | 11,888 | 7,435 | 62.5 | % | ||||||||||||||||||||||
Net income from continuing
operations |
84,838 | 30,835 | 54,003 | 175.1 | % | 30,835 | 4,745 | 26,090 | 549.8 | % | ||||||||||||||||||||||
Loss from discontinued operations,
net of tax benefit of $0.4 million
for 2007 |
— | — | — | n/a | — | (691 | ) | 691 | n/a | |||||||||||||||||||||||
Net income |
$ | 84,838 | $ | 30,835 | $ | 54,003 | 175.1 | % | $ | 30,835 | $ | 4,054 | $ | 26,781 | n/a | |||||||||||||||||
27
Table of Contents
The following table shows the Consolidated Statement of Operations as a percentage of net
sales for 2009, 2008 and 2007.
| 2009 | 2008 | 2007 | ||||||||||
Net sales |
100.0 | % | 100.0 | % | 100.0 | % | ||||||
Cost of sales |
17.8 | % | 22.1 | % | 22.8 | % | ||||||
Gross profit |
82.2 | % | 77.9 | % | 77.2 | % | ||||||
Operating expenses |
||||||||||||
Marketing and selling |
33.5 | % | 35.0 | % | 36.5 | % | ||||||
Research and development |
10.2 | % | 9.2 | % | 9.3 | % | ||||||
In-process research and development |
0.0 | % | 1.5 | % | 1.6 | % | ||||||
General and administrative |
8.7 | % | 7.8 | % | 9.3 | % | ||||||
Integration costs |
0.0 | % | 0.0 | % | 0.2 | % | ||||||
Litigation settlement |
0.0 | % | 0.0 | % | 3.1 | % | ||||||
Amortization of intangibles |
2.5 | % | 6.9 | % | 3.9 | % | ||||||
Total operating expenses |
54.9 | % | 60.4 | % | 64.0 | % | ||||||
Operating income |
27.4 | % | 17.4 | % | 13.2 | % | ||||||
Royalty income |
0.6 | % | 0.9 | % | 1.1 | % | ||||||
Interest income |
0.0 | % | 0.1 | % | 0.2 | % | ||||||
Interest expense |
-3.8 | % | -5.5 | % | -8.1 | % | ||||||
Amortization of financing costs |
-3.0 | % | -3.7 | % | -3.5 | % | ||||||
Gain on extinguishment of debt |
1.9 | % | 1.1 | % | 0.0 | % | ||||||
Other (expense) income |
-0.3 | % | -0.4 | % | 0.7 | % | ||||||
Income from continuing operations before income taxes |
26.2 | % | 10.0 | % | 3.6 | % | ||||||
Provision for income taxes |
9.9 | % | 3.9 | % | 2.6 | % | ||||||
Net income from continuing operations |
16.3 | % | 6.1 | % | 1.0 | % | ||||||
Net income |
16.3 | % | 6.1 | % | 0.9 | % | ||||||
Cost of sales. Gross margin improved to 82.2 percent in 2009 compared to 77.9 percent in 2008
as a result of multiple items. We achieved improved margins in our BPH laser therapy products
driven largely by increased reliability, which resulted in lower warranty and service costs. We
also realized higher margins across all business lines through cost containment and changes in the
mix of products sold, particularly with lower capital sales and increased fiber sales in BPH
therapy than in the previous year. Future gross profit will continue to depend upon product mix,
production levels, labor costs, raw material costs and our ability to manage overhead costs.
Gross margin improved 0.7 percentage points from 2007 to 2008. Margins improved in 2008 as a
result of changes in the mix of products sold, improved inventory planning, lower warranty and
service costs on our BPH laser therapy products and higher average selling prices. These
improvements were partially offset by a decline in margins on our BPH laser therapy products from
about 54 percent in 2007 to 50 percent in 2008, primarily as a result of lower production volume
with the conclusion of the aesthetics product supply agreement. Historically, margins have
generally benefited from stable overhead costs, which account for more than a quarter of our cost
of sales.
Marketing and selling. Marketing and selling expenses as a percentage of sales decreased by 1.5
percentage points to 33.5 percent in 2009 from 2008. The decrease is primarily the result of
decreased distribution expenses due to lower fuel costs. We will continue to make investments in
existing and new geographies in order to drive revenue growth, with an emphasis on driving
geographic expansion.
Marketing and selling expenses as a percentage of sales decreased by 1.5 percentage points in 2008
from 2007. The decrease relates to leveraging of investments made in 2007 primarily for the
transition from independent distributors
28
Table of Contents
to our direct sales force for international laser therapy
sales. As these investments were better leveraged in 2008, the result is lower expense as a
percentage of revenue.
Research and development. Research and development includes costs to develop and improve current
and possible future products plus the costs for regulatory and clinical activities for these
products. Research and development expenses as a percentage of revenue were 10.2 percent in 2009,
9.2 percent in 2008, and 9.3 percent in 2007. These ratios are in line with our long-term goal for
spending in research and development of approximately ten percent of sales.
In-process research and development. The 2008 in-process research and development (IPR&D) expense
represents a $7.5 million milestone payment related to our acquisition of BioControl Medical, Ltd.,
(BioControl) for the in-process development of an implantable electrical stimulation device to
treat urge incontinence and interstitial cystitis (IC). There was a similar milestone payment of
$7.5 million in 2007, also related to BioControl. The following paragraphs describe the status of
previously acquired IPR&D projects that remain in progress at January 2, 2010.
During 2006, we recognized IPR&D charges of $94.0 million, of which $25.6 million related to the
acquisition of BioControl. As noted above, we recognized additional IPR&D charges for BioControl of
$7.5 million during 2007 and $7.5 million during 2008. Since the technology purchased had not yet
reached technological feasibility and lacked an alternative future use, the full purchase price of
$40.6 million was charged to in-process research and development. The development efforts were
less than 50 percent complete at the time of the acquisition. During 2008, based on findings from
earlier feasibility studies, we incorporated the results of these studies into product
enhancements. In 2009, based on results of the clinical trial, we decided not to pursue further
development of the product as currently designed. We will continue product development on the
modified product design over the next two years and then determine our timeline for a new clinical
trial.
Also during 2006, we recognized in-process research and development charges of $62.1 million
related to our acquisition of Laserscope, primarily associated with in-process fiber development
which had not yet reached technological feasibility and lacked an alternative future use. This
included the development of fibers to treat bladder tumors, strictures and renal cancer, as well as
other laser indications. Development for these therapies was estimated to be less than 50 percent
complete at the time of acquisition. In 2008, we launched the extended application fiber for
strictures and bladder tumors. We expect to develop further enhancements for BPH therapy
treatments in addition to developing new laser therapy treatments. We are still in the development
stages for the remaining therapies and expect products to be developed from this in-process
development to reach marketability over the next several years.
General and administrative. General and administrative costs increased $5.9 million in 2009 mainly
due to an increase in compensation related expenses, including stock-based compensation, as well as
cost control measures implemented during 2008, which reduced expenses.
General and administrative costs decreased $3.8 million in 2008 compared to 2007 due to lower
stock-based compensation expense, lower legal costs compared to the prior year, and cost control
measures implemented during 2008.
Integration costs. Integration costs for 2007 includes costs incurred to integrate the acquired
Laserscope operations into overall AMS operations, primarily for legal, consulting and retention
bonuses.
Litigation settlement. During 2007, we recorded a charge of $14.3 million for litigation
settlements, primarily related to the arbitration award to the former shareholders of CryoGen, Inc.
(CryoGen) concerning an earnout payment related to our 2002 acquisition of CryoGen. (See Notes to
Consolidated Financial Statements — No. 3, Litigation Settlements.)
Amortization of intangibles. Amortization of intangibles includes amortization expense on our
definite-lived intangible assets, consisting of patents, licenses and developed technology. The
decrease in intangible amortization expense in 2009 is driven by a $17.1 million impairment charge
in 2008 for the acceleration of amortization to adjust the carrying value of certain intangible
assets related to the TherMatrx® product line and GreenLight PV® technology
to their current fair values, which also resulted in lower on-going amortization expense in 2009.
(See Notes to Consolidated Financial Statements — No. 5, Goodwill and Intangible Assets.)
29
Table of Contents
The increase in intangible amortization expense in 2008 compared to 2007 is driven by a $17.1
million impairment charge for the acceleration of amortization to adjust the carrying value of
certain intangible assets related to the TherMatrx® product line and GreenLight
PV® technology to their current fair values. (See Notes to Consolidated Financial
Statements — No. 5, Goodwill and Intangible Assets.)
Royalty income. Our royalty income is from licensing our intellectual property. We do not
directly influence sales of the products on which these royalties are based and cannot give any
assurance as to future income levels. Royalty income in 2009 decreased $1.4 million compared to
2008 mainly due to a one-time royalty payment we received in 2008 related to a portion of our
urinary incontinence technology acquired in 2006.
We had a one-time item in royalty income in each year in 2008 and 2007. The $4.5 million of
royalty income in 2008 includes a one-time payment related to a portion of our urinary incontinence
technology acquired in 2006, and the $5.0 million in royalty income in 2007 includes a one-time
paid up license of our microwave therapy technology.
Interest income. Interest income of $0.2 million decreased in 2009 compared to 2008, mainly
because of low interest rates earned on our money market funds throughout 2009.
Interest income decreased from $1.2 million in 2007 to $0.7 million in 2008 primarily due to lower
interest rates in 2008 compared to 2007.
Interest expense. Interest expense decreased by $7.8 million in 2009 compared to 2008 mainly due
to the impact of debt reductions made over the past year. Interest expense includes interest
incurred on our convertible senior subordinated notes due 2036 (2036 Notes), which carry a fixed
interest rate of 3.25 percent, the interest incurred on our convertible senior subordinated notes
due 2041 (2041 Notes), which carry a fixed interest rate of 4.00 percent, and the interest incurred
on our Credit Facility, which generally carries a floating interest rate of LIBOR plus 2.25
percent. We have entered into interest rate swaps, which were designated as cash flow hedging
instruments and which have remaining terms of one to six months as of January 2, 2010. The
notional amount of the hedges represents a significant portion of our floating rate debt. The
notional amount of the swap contracts amortizes over their terms, and the amount of floating rate
debt hedged in the future will depend on prepayments and additional contracts. Including the
impact of interest rate swaps, our weighted average interest rate on the credit facility was 4.6
percent and 4.8 percent during 2009 and 2008, respectively. Average borrowings during 2009 on the
Credit Facility were $191.8 million, compared to average borrowings during 2008 of $281.7 million.
Average borrowings on our 2036 Notes were $245.6 million and $371.1 million for 2009 and 2008,
respectively. Average borrowings on our 2041 Notes, which we issued in September of 2009 in
exchange for 2036 Notes (see Notes to Consolidated Financial Statements — 7, Debt), were $71.4
million for 2009.
Interest expense decreased by $10.4 million in 2008 compared to 2007 due to debt reductions in 2008
and a decrease in our effective interest rate from 7.6 percent in 2007 to 4.8 percent in 2008,
which was mainly due to a lower LIBOR rate in 2008. (See Notes to Consolidated Financial
Statements — No. 7, Debt.)
Amortization of financing costs. Amortization of financing costs in 2009 and 2008 was $15.8
million and $18.5 million, respectively, and was comprised of the incremental non-cash interest
cost of our 2036 Notes and 2041 Notes and amortization of the costs associated with the issuance of
the Credit Facility, the 2036 Notes and the 2041 Notes. The $2.7 million decrease in 2009 is due
to the impact of debt prepayments made over the past year, as we recognize a pro rata portion of
the related debt discount and debt issuance costs when we retire debt.
Amortization of financing costs increased $2.3 million in 2008 compared to 2007 due to higher
amortization of the non-cash interest cost of our 2036 Notes due to the use of the effective
interest method in determining the amortization.
Gain on extinguishment of debt. In the first quarter of 2009, we repurchased 2036 Notes with a
principal amount of $27.3 million and we recorded a pre-tax gain on extinguishment of $4.6 million.
In addition, on September 21, 2009, we exchanged $250.0 million in principal of the 2036 Notes for
$250.0 million in principal of 2041 Notes. We accounted for this transaction as an extinguishment
of debt, and we recorded a pre-tax gain on extinguishment of $5.6 million in the third quarter of
2009. In 2008, we repurchased 2036 Notes with a principal amount of $34.5 million, and we recorded
a pre-tax gain on extinguishment of $5.6 million.
30
Table of Contents
Gain on sale of non-strategic assets. During the third quarter of 2009, we sold our female
sterilization assets and technology (Ovion technology) for $23.6 million. The consideration, less
the carrying value of the intangible asset and related disposal costs, resulted in a pre-tax gain
of $17.4 million. The transaction included termination of a royalty agreement, and as a result,
royalty income will be reduced by approximately $0.5 million per quarter going forward. In
addition, as a result of this asset sale agreement, and separate agreements completed with third
parties, we eliminated all existing and potential obligations and liabilities under previous
agreements associated with the Ovion technology.
Other (expense) income. Other (expense) income was a loss of $1.4 million in 2009 compared to a
loss of $2.2 million in 2008. The primary cause of the change in other (expense) income relates to
fluctuations in foreign currencies against the U.S. dollar on foreign denominated inter-company
receivables and payables and the impact of our foreign currency hedge transactions.
Other (expense) income was a loss of $2.2 million in 2008 compared to a gain of $3.1 million in
2007. The primary cause of the change in other (expense) income relates to losses due to the
impact of fluctuations in foreign currencies, mainly the Euro, against the U.S. dollar on foreign
denominated inter-company receivables and payables. Partially offsetting this fluctuation were net
gains from final payments and adjustments related to our disposal of the Laserscope aesthetics
business, and income from license and milestone fees from an agreement to license one of our
technologies. Also, fiscal year 2008 included an impairment charge related to our investment in
Iridex stock which we determined was other-than-temporarily impaired.
Provision for income taxes. Our effective income tax expense rate was 37.6% for 2009 and was
slightly higher than the U.S. statutory tax rate applied to pretax income, primarily as a result of
state taxes which were, in part, offset by manufacturing tax incentives and the federal research
and development tax credit.
Our effective tax rate for 2008 of 38.5 percent on income from continuing operations was higher
than the U.S. statutory tax rate applied to pretax income, primarily as a result of state taxes,
which were offset by manufacturing tax incentives, the federal research and development tax credit,
and a tax benefit due to the settlement of an income tax audit.
Liquidity and Capital Resources
Cash, cash equivalents, and short-term investments were $50.5 million as of January 2, 2010,
compared to $43.0 million as of January 3, 2009. Short-term investments consist mostly of highly
liquid money market funds that have not experienced any negative impact on liquidity or a decline
in principal value.
Cash flows from operating activities
Net cash provided by operating activities increased to $122.8 million in 2009 from $115.8 million
in 2008, which is an increase of $7.0 million, or 6.1%. One of the primary reasons for the $7.0
million increase in cash provided by operating activities is the cash payment of $15.0 million in
2008 for settlement of litigation of the Cryogen arbitration award (see Notes to Consolidated
Financial Statements — No. 3, Litigation Settlements), which reduced cash provided by operating
activities last year. This was partially offset by other changes in operating assets and
liabilities that provided less cash in 2009 compared to 2008. We used a large portion of the cash
provided by operating activities to pay down our debt.
Net cash provided by operating activities increased from $47.8 million in 2007 to $115.8 million in
2008, which is an increase of approximately $68 million. The two main drivers in the increase in
cash from operating activities are changes in inventory and increased net income. Inventory
balances decreased $20.9 million during 2008, resulting from our improved demand planning to
optimize our inventory levels. This compares to an increase in inventory
during 2007 of $30.5 million due mainly to a larger mix of products as several new products and
product configurations were introduced in 2007, and also due to a decline in inventory efficiency.
The change in inventory generated incremental cash of $51.4 million, on a year over year basis.
Incremental cash of $26.8 million was generated from higher net income, adjusted for
reclassifications and non-cash items, in 2008 compared to 2007.
In addition, due to improvements in our days sales outstanding, accounts receivable decreased in
2008 and generated cash of $11.0 million, while accounts receivable increased in 2007 and was a use
of cash of $9.8 million. This
31
Table of Contents
change in accounts receivable on a year over year basis resulted in
incremental cash of $20.8 million. Offsetting the above increases in cash flows from operating
activities was our payment of $15.0 million for settlements of litigation, primarily due to the
CryoGen arbitration award, and the change in other assets related primarily to the divestiture of
the aesthetics business, which results in a year over year use of cash of $21.1 million.
Cash flows from investing activities
Cash provided by investment activities was $18.6 million in 2009, compared to cash used of $42.2
million in 2008, which equates to an increase in cash provided of $60.7 million. The increase in
cash provided by investing activities is mainly the result of two significant items. First, the
sale of our Ovion technology in the third quarter of 2009 provided net cash of $19.0 million.
Second, we had an increase in cash provided by short-term investment activities of $42.4 million
(during 2009 we made sales of short-term investments of $30.7 million offset by purchases of
short-term investments of $18.8 million, for net cash provided of $11.9 million, compared to net
cash used of $30.5 million in 2008).
Cash used in investing activities was $42.2 million and $1.3 million in 2008 and 2007,
respectively. The increase in cash used of approximately $40.9 million is mainly due to the
increase in short-term investments of $30.2 million in 2008. Also, during 2008 and 2007, we
received $4.7 million and $22.1 million, respectively, as a result of the divestiture of the
aesthetics business, and we made a $7.5 million milestone payment during each year related to our
acquisition of BioControl.
Cash flows from financing activities
Cash used for financing activities was $121.6 million in 2009, versus $98.2 million in 2008. Cash
used for prepayment of the Credit facility increased $18.3 million, from 85.2 million in 2008 to
$103.5 million in 2009. In addition, we repurchased 2036 Notes with a principal amount of $27.3
million for a cash payment of $21.1 million in 2009 for a total debt reduction of $130.8 million in
2009. We also paid $7.7 million for debt issuance costs related to our convertible note exchange
in September 2009 (see Notes to Consolidated Financial Statements — No. 7, Debt). Cash received
from the issuance of common stock was $10.2 million and $8.9 million in 2009 and 2008,
respectively, the majority of which came from our employees exercising stock options.
Cash used in financing activities was $98.2 million and $39.0 million in 2008 and 2007,
respectively. In 2008 and 2007, we made payments on our long-term debt of $85.2 million and $50.1
million, respectively. In December 2008, we repurchased 2036 Notes with a principal amount of
$34.5 million for a cash payment of $23.4 million. We received cash from the issuance of common
stock for $8.9 million and $10.8 million in 2008 and 2007, respectively, the majority of which came
from our employees exercising stock options.
2036 Notes. We issued our 2036 Notes with a stated maturity of July 1, 2036 pursuant to an
Indenture dated as of June 27, 2006 as supplemented by the first supplemental indenture dated
September 6, 2006 (the 2036 Notes Indenture) between us, certain of our significant domestic
subsidiaries, as guarantors of the 2036 Notes, and U.S. Bank National Association, as trustee for
the benefit of the holders of the 2036 Notes, which specifies the terms of the 2036 Notes. The
2036 Notes bear interest at the rate of 3.25 percent per year, payable semiannually. The 2036
Notes are our direct, unsecured, senior subordinated obligations, rank junior to our Credit
Facility and will rank junior in right of payment to all of our future senior secured debt as
provided in the 2036 Notes Indenture. The 2036 Notes have the same rank as our 2041 Notes.
In addition to regular interest on the 2036 Notes, we will also pay contingent interest beginning
July 1, 2011 at 0.25% of the average trading price of the 2036 Notes, if the average trading price
for the five consecutive trading days immediately before the last trading day preceding the
relevant six-month period equals or exceeds 120 percent of the principal amount of the 2036 Notes.
The 2036 Notes are convertible under certain circumstances for cash and
shares of our common stock, if any, at a conversion rate of 51.5318 shares of our common stock per
$1,000 principal amount of 2036 Notes (which is equal to an initial conversion price of
approximately $19.406 per share), subject to adjustment. Upon conversion, we would be required to
satisfy up to 100 percent of the principal amount of the 2036 Notes solely in cash, with any
amounts above the principal amount to be satisfied in shares of our common stock.
32
Table of Contents
If a holder elects to convert its 2036 Note in connection with a designated event or change that
occurs prior to July 1, 2013, we will pay, to the extent described in the 2036 Notes Indenture, a
make whole premium by increasing the conversion rate applicable to such 2036 Notes. All of the
above conversion rights will be subject to certain limitations imposed by our Credit Facility.
We may also redeem the 2036 Notes on or after July 6, 2011 at specified redemption prices as
provided in the 2036 Notes Indenture plus accrued and unpaid interest and contingent interest.
Holders of the 2036 Notes may require us to purchase all or a portion of their 2036 Notes for cash
on July 1, 2013, July 1, 2016, July 1, 2021, July 1, 2026, and July 1, 2031 or in the event of a
designated event or change, at a purchase price equal to 100 percent of the principal amount of the
2036 Notes to be repurchased plus accrued and unpaid interest and contingent interest.
2041 Notes. We issued our 2041 Notes with a stated maturity of September 15, 2041 pursuant to an
Indenture dated as of September 21, 2009 (the 2041 Notes Indenture) between us, certain of our
significant domestic subsidiaries, as guarantors of the 2041 Notes, and U.S. Bank National
Association, as trustee for the benefit of the holders of the 2041 Notes, which specifies the terms
of the 2041 Notes. The 2041 Notes bear interest at the rate of 4.00 percent per year, payable
semiannually. The 2041 Notes are our direct, unsecured, senior subordinated obligations, rank
junior to our Credit Facility and will rank junior in right of payment to all of our future senior
debt as provided in the 2041 Notes Indenture. The 2041 Notes have the same rank as our 2036 Notes.
In addition to regular interest on the 2041 Notes, we will also pay contingent interest beginning
September 15, 2016 at 0.75% of the average trading price of the 2041 Notes, if the average trading
price for the five consecutive trading days immediately before the first day of such semiannual
period equals or exceeds 130 percent of the principal amount of the 2041 Notes. The 2041 Notes are
convertible under certain circumstances for cash and shares of our common stock, if any, at a
conversion rate of 51.5318 shares of our common stock per $1,000 principal amount of 2041 Notes
(which is equal to an initial conversion price of approximately $19.406 per share), subject to
adjustment. Upon conversion, we would be required to satisfy up to 100 percent of the principal
amount of the 2041 Notes solely in cash, with any amounts above the principal amount to be
satisfied in shares of our common stock.
If a holder elects to convert its 2041 Note in connection with a designated event or change, we
will pay, to the extent described in the 2041 Notes Indenture, a make whole premium by increasing
the conversion rate applicable to such 2041 Notes. All of the above conversion rights will be
subject to certain limitations imposed by our Credit Facility.
We may also redeem the 2041 Notes on or after September 15, 2016 at specified redemption prices as
provided in the 2041 Notes Indenture plus accrued and unpaid interest and contingent interest.
Holders of the 2041 Notes may require us to purchase all or a portion of their 2041 Notes for cash
on September 15, 2016 or in the event of a designated event or change, at a purchase price equal to
100 percent of the principal amount of the 2041 Notes to be repurchased plus accrued and unpaid
interest and contingent interest.
2036 Notes and 2041 Notes — Potential Dilution. Prior to conversion, our 2036 Notes and 2041
Notes represent potentially dilutive common share equivalents that must be considered in our
calculation of diluted earnings per share (EPS). When there is a net loss, common share
equivalents are excluded from the computation because they have an anti-dilutive effect. In
addition, when the conversion price of our 2036 Notes and 2041 Notes is greater than the average
market price of our stock during any period, the effect would be anti-dilutive and we would exclude
the 2036 Notes and 2041 Notes from the EPS computation. However, when the average market price of
our stock during any period is greater than the conversion price of the 2036 Notes and 2041 Notes,
the impact is dilutive and the 2036 Notes and 2041 Notes will affect the number of common share
equivalents used in the diluted EPS calculation. The degree to which the 2036 Notes and 2041 Notes
are dilutive increases as the market price of our stock increases.
The following table illustrates the number of common share equivalents that would potentially be
included in weighted average common shares for the calculation of diluted EPS, assuming various
market prices of our stock:
33
Table of Contents
| If the average | The number of common share equivalents potentially included in the computation of | |||||||||||||||||
| market price | of diluted EPS would be (1): | Percent Dilution (2) | ||||||||||||||||
| of our stock is: | 2036 Notes | 2041 Notes | Total | 2036 Notes | 2041 Notes | Total | ||||||||||||
$19.00 |
— (anti-dilutive) | — (anti-dilutive) | — (anti-dilutive) | 0.0 | % | 0.0 | % | 0.0 | % | |||||||||
$20.00 |
0.1 million | 0.4 million | 0.5 million | 0.1 | % | 0.5 | % | 0.6 | % | |||||||||
$22.50 |
0.4 million | 1.8 million | 2.2 million | 0.6 | % | 2.3 | % | 2.9 | % | |||||||||
$25.00 |
0.7 million | 2.9 million | 3.6 million | 0.9 | % | 3.7 | % | 4.6 | % | |||||||||
$27.50 |
0.9 million | 3.8 million | 4.7 million | 1.2 | % | 4.8 | % | 6.0 | % | |||||||||
$30.00 |
1.1 million | 4.5 million | 5.6 million | 1.5 | % | 5.7 | % | 7.2 | % | |||||||||
| (1) | Common share equivalents are calculated using the treasury stock method. The formula to calculate the potentially dilutive shares related to our Convertible Notes is as follows: |
| ( | Principal Amount |
x | Market price | — | Principal | ) | = | Potentially dilutive shares | |||||
| $19.406 conversion price | of stock | Amount | included in EPS | ||||||||||
| Market price of stock | |||||||||||||
| (2) | The percent dilution is based on 74,715,845 outstanding shares as of January 2, 2010. |
For the twelve months ended January 2, 2010 and January 3, 2009, our Convertible Notes were
excluded from the diluted net income per share calculation because the conversion price was greater
than the average market price of our stock.
Credit Facility. On July 20, 2006, our wholly-owned subsidiary, American Medical Systems, Inc.
(AMS), entered into a senior secured Credit Facility. AMS and each majority-owned domestic
subsidiary of AMS are parties to the Credit Facility as guarantors of all of the obligations of AMS
arising under the Credit Facility. The obligations of AMS and each of the guarantors arising under
the Credit Facility are secured by a first priority security interest on substantially all of their
respective assets, including a mortgage on the AMS facility in Minnetonka, Minnesota.
The six-year senior secured Credit Facility consists of (i) term loan debt and (ii) a revolving
credit facility of up to $65.0 million which is available to fund ongoing working capital needs,
including future capital expenditures and permitted acquisitions. During January 2008, we borrowed
$12.0 million under the revolving credit facility to fund the payment of certain litigation
settlements (refer to Notes to Consolidated Financial Statements – No. 3, Litigation Settlements).
We repaid the outstanding balance with operating cash in February 2008.
Our Credit Facility contains affirmative and negative covenants and other limitations (subject
to various carve-outs and baskets) regarding us, AMS, and in some cases, the subsidiaries of AMS.
The covenants limit: (a) investments, capital expenditures, dividend payments, the disposition of
material assets other than in the ordinary course of business, and mergers and acquisitions under
certain conditions, (b) transactions with affiliates, unless such transactions are completed in the
ordinary course of business and upon fair and reasonable terms, (c) liens and indebtedness, and (d)
substantial changes in the nature of our business. Our Credit Facility contains customary
financial covenants for secured credit facilities, consisting of maximum total and senior debt
leverage ratios and minimum interest coverage and fixed charge coverage ratios. These financial
covenants adjust from time to time during the term of the Credit Facility. The covenants and
restrictions contained in the Credit Facility could limit our ability to fund our business, make
capital expenditures, and make acquisitions or other investments in the future.
On August 12, 2009, we received a Consent and Second Amendment to our Credit Facility, which
allowed us to exchange a portion of our existing convertible senior subordinated notes for new
convertible senior subordinated notes (see Notes to Consolidated Financial Statements – No. 7,
Debt). On October 29, 2007, we entered into a First Amendment of our Credit Facility to modify
certain financial covenant ratios as defined in the Credit Facility (the Amendment). Pursuant to
the terms of the Amendment, certain of the financial tests and covenants provided in Section 6.8 of
the Credit Facility were amended and restated, including the interest coverage ratio, the total
leverage ratio, the fixed charge coverage ratio, and the maximum consolidated capital expenditures.
As of January 2, 2010, we were in compliance with all financial covenants as defined in our Credit
Facility which are summarized as follows:
34
Table of Contents
| Financial Covenant | Required Covenant | Actual Result | ||||
Total Leverage Ratio (1) |
3.50:1.00 (maximum) | 2.48 | ||||
Senior Leverage Ratio (2) |
2.00:1.00 (maximum) | 0.71 | ||||
Interest Coverage Ratio (3) |
4.00:1.00 (minimum) | 8.97 | ||||
Fixed Charge Coverage Ratio (4) |
1.50:1.00 (minimum) | 2.35 | ||||
Maximum Capital Expenditures (5) |
$17.5 million | $5.9 million | ||||
| (1) | Total outstanding debt to Consolidated Adjusted EBITDA for the trailing four quarters. | |
| (2) | Total outstanding senior secured debt to Consolidated Adjusted EBITDA for the trailing four quarters. | |
| (3) | Ratio of Consolidated Adjusted EBITDA for the trailing four quarters to cash interest expense for such period. | |
| (4) | Ratio of Consolidated Adjusted EBITDA for the trailing four quarters to fixed charges (cash interest expense, scheduled principal payments on debt, capital expenditures, income taxes paid, earn-out and milestone payments) for such period. | |
| (5) | Limit of capital expenditures for the full year. |
The ratios are based on EBITDA, on a rolling four quarters, calculated with certain adjustments
(Consolidated Adjusted EBITDA). Consolidated Adjusted EBITDA is a non-GAAP financial measure that
is defined in our Credit Facility as earnings before interest, income taxes, depreciation,
amortization, and other non-cash items reducing net income including IPR&D and stock compensation
charges, less other non-cash items increasing net income. Consolidated Adjusted EBITDA should not
be considered an alternative measure of our net income, operating performance, cash flow or
liquidity. It is provided as additional information relative to compliance with our debt
covenants.
Any failure to comply with any of these financial and other affirmative and negative covenants
would constitute an event of default under the Credit Facility, entitling a majority of the bank
lenders to, among other things, terminate future credit availability under the Credit Facility,
increase the interest rate on outstanding debt, and accelerate the maturity of outstanding
obligations under the Credit Facility.
Our borrowing arrangements are further described in Notes to Consolidated Financial Statements –
No. 7, Debt.
Contractual Obligations
The following table sets forth the future commitments for our long-term debt and operating leases.
| Less than | More than | |||||||||||||||||||
| (in millions) | Total | 1 year | 1-3 years | 3-5 years | 5 years | |||||||||||||||
Long-term debt obligations (1) |
$ | 437.3 | $ | 1.3 | $ | 124.0 | $ | — | $ | 312.0 | ||||||||||
Operating lease commitments |
8.6 | 3.4 | 4.6 | 0.6 | — | |||||||||||||||
Total |
$ | 445.9 | $ | 4.7 | $ | 128.6 | $ | 0.6 | $ | 312.0 | ||||||||||
| (1) | Our 2036 Notes have a maturity of July 1, 2036 and our 2041 Notes have a maturity of September 15, 2041 and thus the principal of the Convertible Notes has been included in the “more than 5 years” column in the table above. Holders of our 2036 Notes may require us to purchase all or a portion of their 2036 Notes for cash on July 1, 2013, July 1, 2016, July 1, 2021, July 1, 2026, and July 1, 2031. Holders of the 2041 Notes may require us to purchase all or a portion of their 2041 Notes for cash on September 15, 2016. |
In addition to the amounts shown in the table above, $18.2 million of unrecognized tax benefits
have been recorded as liabilities, and we are uncertain as to if or when such amounts may be
settled.
On April 26, 2006, we acquired certain issued patents and other assets from BioControl, an Israeli
company focused on developing medical devices for the application of electrical stimulation
technology. We acquired an exclusive license for the use of the patents and technologies in
urology, gynecology and other pelvic health applications. The purchase price was comprised of an
initial payment of $25.0 million, milestone payments for relevant
35
Table of Contents
accomplishments through and
including FDA approval of the product of up to $25.0 million, and royalties over the first ten
years of the related license agreement. In the fourth quarter of 2007, we made a milestone payment
of $7.5 million. In August 2008, we and BioControl amended the asset purchase and license
agreements. Under these amendments, we agreed that the conditions for achieving the first
milestone had been satisfied, and in the third quarter of 2008 we paid an additional $7.5 million
for this milestone. In addition, BioControl agreed to eliminate our obligations to use
commercially reasonable efforts to complete the remaining third milestone, and they released and
waived all claims relating to such obligations. We remain liable to make the third milestone
payment of $10.0 million if and when the payment conditions are satisfied, and we agreed to make
certain other payments in the event that we transfer the BioControl technology to another party
prior to achieving the third milestone. The royalty period was also extended for an additional
three years.
We believe that funds generated from operations, together with our balances in cash and cash
equivalents, as well as short-term investments and our revolving Credit Facility, will be
sufficient to finance current operations, planned capital expenditures, servicing of existing debt
and any contingent payments that become due related to the acquisition described above.
Critical Accounting Policies and Estimates
We base our estimates on historical experience and on various other assumptions that we believe to
be reasonable under the circumstances. Management’s discussion and analysis of financial condition
and results of operations is based upon the consolidated financial statements, which have been
prepared in accordance with accounting principles generally accepted in the United States. The
preparation of these financial statements requires us to make estimates and judgments that affect
(1) the reported amounts of assets, liabilities, revenues, and expenses and (2) the related
disclosure of contingent assets and liabilities. Estimates are used for items including but not
limited to, those related to accounts receivable and sales return obligations, inventories, fair
values of acquired assets and liabilities under the purchase method of accounting, impairment
testing of long-lived assets, warranty, legal contingencies, valuation of share-based payments and
income taxes. The critical accounting policies and estimates that are most important in fully
understanding and evaluating the financial condition and results of operations are discussed below.
Revenue Recognition Policy
We sell our products primarily through a direct sales force. A portion of our revenue is generated
from consigned inventory or from inventory with field representatives. For these products, revenue
is recognized at the time the product has been used or implanted. For all other transactions, we
recognize revenue when title to the goods and risk of loss transfer to customers, providing there
are no remaining performance obligations required from us or any matters requiring customer
acceptance. In cases where we utilize distributors or ship product directly to the end user,
we recognize revenue upon shipment provided all revenue recognition criteria have been met. We
record estimated sales returns, discounts and rebates as a reduction of net sales in the sale
period when revenue is recognized.
Occasionally, sales of capital equipment have post-sale obligations, such as installation and
extended service contracts, which are fulfilled after product shipment. For each multiple element
arrangement, we determine if each element is a separate unit of accounting by ensuring that (1) the
element has stand alone value to the customer, (2) there is objective evidence of the fair value
for the element, and (3) if the arrangement includes a general right of return relative to the
delivered item, delivery of the undelivered items is considered probable and in our control. To
determine the fair value for each element in an arrangement, we rely primarily upon vendor specific
objective evidence (VSOE) of fair value using the price charged when we sell that element
separately, or in the case of hardware that we do not sell separately, we rely upon vendor
objective evidence of fair value in the form of competitor pricing of the same or interchangeable
products. We defer revenue attributable to the post-shipment obligations and recognize such
revenue when the obligation is fulfilled.
We provide incentives to customers, including volume based rebates. Customers are not required to
provide documentation that would allow us to reasonably estimate the fair value of the benefit
received and we do not receive an identifiable benefit in exchange for the consideration.
Accordingly, the incentives are recorded as a reduction of revenue.
36
Table of Contents
All of our customers have rights of return for the occasional ordering or shipping error. We
maintain an allowance for these returns and reduce reported revenue for expected returns from
shipments during each reporting period. This allowance is based on historical and current trends in
product returns. At January 2, 2010 this allowance was $2.1 million, and it was $2.6 million at
January 3, 2009.
Allowance for Doubtful Accounts
We estimate the allowance for doubtful accounts by analyzing those accounts receivable that have
reached their due date and by applying rates based upon historical write-off trends and specific
account reserves. Accounts are written off sooner in the event of bankruptcy or other circumstances
that make further collection unlikely. When it is deemed probable that a customer account is
uncollectible, that balance is written off against the existing allowance. Different estimates
could have material variances in the amount and timing of our reported results for any period. In
addition, actual results could be different from current estimates, possibly resulting in increased
future charges to earnings.
The allowance for doubtful accounts was $3.4 million and $3.5 million at January 2, 2010 and
January 3, 2009, respectively, which represented 3.2 percent and 3.7 percent of gross accounts
receivable, respectively. The decrease in allowance compared to the prior year is due to a
reduction in the specific reserves related to certain international accounts.
Derivative Instruments Policy
For information regarding our use of derivatives see Item 7A, Quantitative and Qualitative
Disclosures about Market Risk, and also see Notes to Consolidated Financial Statements – No. 11,
Derivative Instruments and Hedging Activities.
Inventories
Inventories are stated at the lower of cost or market determined on the first-in-first-out method.
Each quarter, we evaluate our inventories for obsolescence and excess quantities. This evaluation
includes analyses of inventory levels, historical loss trends, expected product lives, product at
risk of expiration, sales levels by product, and projections of future sales demand. We reserve
inventories we consider obsolete. In addition, we record an allowance for inventory quantities in
excess of forecasted demand. Inventory allowances were $5.0 million and $4.2 million at the end of
2009 and 2008, respectively. If future demand or market conditions are less favorable than current
estimates, additional inventory adjustments would be required and would adversely affect income in
the period the adjustment is made.
Purchase Accounting and Valuation of IPR&D
When we acquire another company, the purchase price is allocated, as applicable, between in process
research and development (IPR&D), other identifiable intangible assets, tangible assets, and
goodwill. IPR&D is defined as the value assigned to those projects for which the related products
have not received regulatory approval and have no alternative future use. The amount of the
purchase price allocated to IPR&D and other intangible assets is determined by estimating the
future cash flows of each project or technology and discounting the net cash flows back to their
present values. The discount rate used is determined at the time of acquisition in accordance with
accepted valuation methods. For IPR&D, these methodologies include consideration of the risk of the
project not achieving commercial feasibility.
The forecast data employed in the analysis of our various IPR&D charges was based upon internal
product level forecast and external market information. The forecast data and assumptions are
inherently uncertain and unpredictable. However, based upon the information available at this
time, we believe the forecast data and assumptions used are reasonable. These assumptions may be
incomplete or inaccurate, and no assurance can be given that unanticipated events and circumstances
will not occur. Unless otherwise noted, forecast and assumptions have not changed materially from
the date the appraisals were completed.
At the time of acquisition, we expect all acquired IPR&D will reach technological feasibility, but
there can be no assurance that the commercial viability of these projects will actually be
achieved. The nature of the efforts to
37
Table of Contents
develop the acquired technologies into commercially viable
products consists principally of planning, designing and conducting clinical trials necessary to
obtain regulatory approvals. The risks associated with achieving commercialization include, but
are not limited to, delay or failure to obtain regulatory approvals to conduct clinical trials,
failure of clinical trials, delay or failure to obtain required market clearances, and patent
litigation. If commercial viability were not achieved, we would not realize the original estimated
financial benefits expected for these projects. We fund all costs to complete IPR&D projects with
internally generated cash flows.
Goodwill
Goodwill is the excess of the purchase price over the fair value of net assets, including IPR&D, of
acquired businesses. Goodwill is tested for impairment annually during the fourth quarter, or
whenever a change in circumstances or the occurrence of events suggest the remaining value may not
be recoverable. The goodwill impairment tests are considered critical due to the amount of goodwill
recorded on our consolidated balance sheets and the judgment required in determining fair value
amounts. Goodwill was $690.9 million as of January 2, 2010 and $690.1 million as of January 3,
2009.
For our annual goodwill impairment assessment, we currently use the market approach to determine
the fair value of our single reporting unit. The approach calculates fair value as our market
capitalization, plus a control premium, less cash and short-term investments, plus debt, to reach
the enterprise fair value. Our market capitalization is calculated by multiplying our common
shares outstanding by the average market price of our common stock for the 15 business days prior
to, and 15 business days after, our assessment date. We used a control premium of 30% for our
impairment assessment, as we believe this is within a reasonable range of historical control
premiums paid for acquisitions in the medical device industry. We selected this method as being
the most meaningful in preparing our goodwill assessment because it bases fair value on the quoted
market price of our stock in an active market, while also factoring in that a market participant
would be willing to pay a premium over our market capitalization to obtain full control of the
company. The result of our most recent impairment assessment completed in the fourth quarter of
2009 was a fair value of $1.9 billion, compared to the carrying value of our reporting unit of
$515.7 million. See Notes to Consolidated Financial Statements – No. 5, Goodwill and Intangible
Assets, for further information.
Other Intangible Assets
Other intangible assets consist primarily of purchased technology, patents, and trademarks and are
generally amortized using the straight-line method over their estimated useful lives. We review our
intangible assets for impairment annually or as changes in circumstance or the occurrence of events
suggest the remaining value may not be recoverable. Intangible assets with indefinite lives are not
amortized, but are tested for impairment annually during the fourth quarter or whenever there is an
impairment indicator. Impairment, if any, is recognized through acceleration of amortization and
recorded as amortization of intangibles. For further discussion of definite-lived intangibles for
which amortization was accelerated during 2008, refer to Notes to Consolidated Financial Statements
– No. 5, Goodwill and Intangible Assets. Other intangible assets, net of accumulated amortization,
were $101.6 million as of January 2, 2010 and $109.7 million as of January 3, 2009.
Warranty Accrual / Allowance
We warrant all of our products to be free from manufacturing defects. In addition, if a product
fails, we may provide replacements at no cost or at a substantial discount from list price. We
maintain a warranty allowance to cover the cost of replacements for our erectile restoration,
incontinence, BPH, and urinary stones products. When we sell products, we record an expense for the
expected costs of future warranty-related claims, and increase the warranty allowance by an
equivalent amount. We reduce the warranty allowance by the cost of the replacement device when an
actual claim is awarded. Thus, the balance of the warranty allowance is an estimate of the future
cost of honoring our warranty obligation. Factors influencing this estimate include historical
claim rates, changes in product performance, frequency of use by the patient, the patient’s
performance expectations, and changes in the terms of our product replacement policy. Product
reliability is a function of raw material properties, manufacturing processes, and surgical
technique.
38
Table of Contents
At January 2, 2010, our accrued warranty allowance was $2.3 million compared to $3.3 million at
January 3, 2009. If we experience changes in any of the factors that influence this estimate, we
will make adjustments to this accrued warranty allowance.
Product Liability Accrual
Each quarter, we estimate the uninsured portion of legal representation and settlement costs of
product liability claims and lawsuits. This evaluation consists of reviewing historical claims
costs as well as assessing future trends in medical device liability cases. Social and political
factors, as well as surgeon and medical facility responsibility, make litigation costs hard to
predict. Accruals for future litigation costs were $1.0 million at January 2, 2010, versus $0.8
million at January 3, 2009. The accrual amount reflects the estimate related to identified claims
and lawsuits. If, in the future, we determine that this accrual is inadequate, the adjustment
would reduce reported income in the period we recorded the adjustment.
Valuation of Share-Based Payments
Our share-based compensation plans include stock options and restricted stock. We also sponsor an
employee stock purchase plan. We account for these plans using a fair value method. Stock options
and grants are valued using the Black-Scholes closed-form model for estimating the fair value of
employee stock options and similar instruments. This model is based on several key inputs. Risk
free interest rates are based on the applicable federal Treasury bill rate. Stock price volatility
is determined based on historical rates over the comparable option expected life. Expected option
lives are determined based on employee groups with similar exercise patterns, as determined by the
historical activity. Expense is reduced each period for expected forfeitures, the rate of which
was determined based on historical rates. We use the straight-line method of expense attribution
that results in a straight-line amortization of the compensation expense over the vesting period
for all options.
We recognize compensation expense for the fair value of restricted stock grants issued based on the
closing stock price on the date of grant. Compensation expense recognized on shares issued under
our Employee Stock Purchase Plan is based on the value of an option to purchase shares of our stock
at a 15 percent discount to the stock price.
Total stock-based compensation expense recognized during the fiscal years ended January 2, 2010,
January 3, 2009 and December 29, 2007 was $9.0 million, $8.9 million and $12.4 million,
respectively. See Notes to Consolidated Financial Statements – No. 8, Stock-Based Compensation for
further information regarding our stock-based compensation programs.
Income Taxes
In the preparation of the consolidated financial statements, income taxes in each of the
jurisdictions in which we operate are determined. This process involves estimating and judgment
for current tax liabilities, assessing deferred tax assets and liabilities, valuation allowances
and tax reserves. The tax rules require that certain items have tax treatment that is different
from the consolidated financial statements. The different tax treatment may be permanent
or temporary which is reflected in our effective tax rate and related tax accounts in the
consolidated financial statements.
Our deferred tax assets include such items as timing differences on certain accruals, reserves, and
deferred revenue. Other deferred tax assets exist for net operating losses on various federal and
state tax returns, research and development and foreign tax credits. Our deferred tax liabilities
include such items as amortization of trademarks and other intangibles, and contingent interest on
the Convertible Notes.
We review deferred tax assets and determine the need for a valuation allowance on a quarterly
basis. The valuation allowance assessment considers historical taxable income, estimates of future
taxable income, and the impact of tax planning strategies. If a determination is made that we
would not realize all or part of the deferred tax assets, an adjustment to the deferred tax asset
valuation allowance and a charge to income in the period of the determination would be made. As of
January 2, 2010, no valuation allowance is required.
We recognize the tax benefit from an uncertain tax position only if it is more likely than not that
the tax position will be sustained on examination by the taxing authorities, based on the technical
merits of the position. The tax benefits
39
Table of Contents
recognized in the financial statements from such a
position are measured based on the largest benefit that has a greater than fifty percent likelihood
of being realized upon ultimate settlement.
We assess our reserves for uncertain tax benefits on a quarterly basis. We believe that all of our
tax positions are fully supportable. However, we establish a reserve for uncertain tax benefits
for actual tax benefits claimed or planned to be claimed on tax return filings in excess of what is
allowed to be recognized for financial statement purposes.
Recent Accounting Pronouncements
See Notes to Consolidated Financial Statements – No.1, Business Description and Significant
Accounting Policies.
Forward-Looking Statements
This Annual Report on Form 10-K contains not only historical information, but also forward-looking
statements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the
Securities Exchange Act of 1934 and are subject to the safe harbor created by those sections. In
addition, we or others on our behalf may make forward-looking statements from time to time in oral
presentations, including telephone conferences and/or web casts open to the public, in press
releases or reports, on our Internet web site or otherwise. All statements other than statements
of historical facts included in this report or expressed by us orally from time to time that
address activities, events or developments that we expect, believe or anticipate will or may occur
in the future are forward-looking statements including, in particular, the statements about our
plans, objectives, strategies, the outcome of contingencies such as legal proceedings, and
prospects regarding, among other things, our financial condition, results of operations and
business. We have identified some of these forward-looking statements in this report with words
like “believe,” “may,” “could,” “would,” “might,” “project,” “will,” “should,” “expect,” “intend,”
“plan,” “predict,” “anticipate,” “estimate,” or “continue” or the negative of these words or other
words and terms of similar meaning. These forward-looking statements may be contained in the notes
to our consolidated financial statements and elsewhere in this report, including under the heading
“Management’s Discussion and Analysis of Financial Condition and Results of Operations.”
Forward-looking statements are based on management’s beliefs, certain assumptions and current
expectations and factors that affect all businesses operating in a global market as well as matters
specific to us. These uncertainties and factors are difficult to predict and many of them are
beyond our control.
The following are some of the uncertainties and factors known to us that could cause our actual
results to differ materially from what we have anticipated in our forward-looking statements:
successfully competing against competitors; physician acceptance, endorsement, and use of our
products; potential product recalls or technological obsolescence; successfully managing increased
debt leverage and related credit facility financial covenants; the impact of worldwide economic
conditions on our operations; the disruption in global financial markets potential impact on the
ability of our counterparties to perform their obligations and our ability to obtain future
financing; factors impacting the stock market and share price and its impact on the dilution of
convertible securities; ability of
our manufacturing facilities to meet customer demand; reliance on single or sole-sourced suppliers;
loss or impairment of a principal manufacturing facility; clinical and regulatory matters; timing
and success of new product introductions; patient acceptance of our products and therapies; changes
in and adoption of reimbursement rates; healthcare reform legislation in the U.S.; adequate
protection of our intellectual property rights; product liability claims; and currency and other
economic risks inherent in selling our products internationally.
For more information regarding these and other uncertainties and factors that could cause our
actual results to differ materially from what we have anticipated in our forward-looking statements
or otherwise could materially adversely affect our business, financial condition or operating
results, refer to this Annual Report on Form 10-K under Part I, Item 1A, “Risk Factors.”
All forward-looking statements included in this report are expressly qualified in their entirety by
the foregoing cautionary statements. We wish to caution readers not to place undue reliance on any
forward-looking statement that speaks only as of the date made and to recognize that
forward-looking statements are predictions of future results, which may not occur as anticipated.
Actual results could differ materially from those anticipated in the forward-looking statements and
from historical results, due to the uncertainties and factors described above, as well
40
Table of Contents
as others
that we may consider immaterial or do not anticipate at this time. The risks and uncertainties
described above are not exclusive and further information concerning us and our business, including
factors that potentially could materially affect our financial results or condition, may emerge
from time to time. We assume no obligation to update, amend or clarify forward-looking statements
to reflect actual results or changes in factors or assumptions affecting such forward-looking
statements. We advise you, however, to consult any further disclosures we make on related subjects
in our annual reports on Form 10-K, quarterly reports on Form 10-Q and current reports on Form 8-K
that we file with or furnish to the Securities and Exchange Commission.
Item 7A. Quantitative and Qualitative Disclosures about Market Risk
We use derivatives to mitigate our exposure to volatility in interest and foreign currency exchange
rates. We hedge only exposures in the ordinary course of business.
Interest Rates
We have interest rate risk as a result of the floating LIBOR index that is used to determine the
interest rates on our Credit Facility. Accordingly, we have entered into various fixed interest
rate swap contracts. As of January 2, 2010, the notional amount of the outstanding swap contracts,
which mature over the next one to six months, represented a majority of our floating rate debt.
Based on a sensitivity analysis, as of January 2, 2010, an instantaneous and sustained
100-basis-point increase in interest rates affecting our floating rate debt obligations, and
assuming that we take no counteractive measures, would result in a decrease in income before income
taxes of approximately $1.0 million over the next 12 months. The estimated impact to income takes
into account the mitigating effect of the interest rate swap agreements. The fair market value of
the outstanding swap contracts is a derivative liability of $0.3 million at January 2, 2010. The
notional amount of the contracts amortizes over their terms, and the amount of floating rate debt
hedged in the future will depend on prepayments and additional contracts.
Currency
Our operations outside of the United States are maintained in their local currency. All assets and
liabilities of our international subsidiaries are translated to U.S. dollars at period-end exchange
rates. Translation adjustments arising from the use of differing exchange rates are included in
accumulated other comprehensive income in stockholders’ equity. Gains and losses on foreign
currency transactions and short term inter-company receivables from foreign subsidiaries are
included in other (expense) income.
During fiscal 2009 and 2008, revenues from sales to customers outside the United States were 28.0
percent and 29.1 percent of total consolidated revenues, respectively. International accounts
receivable, inventory, cash and short-term investments, and accounts payable were 41.3 percent, 8.3
percent, 28.2 percent, and 25.7 percent of total consolidated accounts for each of these items as
of January 2, 2010. The reported results of our foreign operations will be influenced by their
translation into U.S. dollars by currency movements against the U.S. dollar. The result of a
uniform 10 percent strengthening in the value of the U.S. dollar in 2009 relative to each of the
currencies in which
our revenues and expenses are denominated would have resulted in a decrease in net income of
approximately $3.4 million during 2009.
During 2009, we entered into various foreign exchange forward contracts to manage a portion of our
exposure to foreign exchange rate fluctuations on our forecasted sales to and receivables from
certain subsidiaries, denominated in Euros, British pounds, Canadian dollars and Australian
dollars. At January 2, 2010, our net investment in foreign subsidiaries translated into dollars
using the period end exchange rate was $34.6 million and the potential loss in fair value resulting
from a hypothetical 10 percent strengthening in the value of the U.S. dollar currency exchange rate
amounts to $3.5 million. Actual amounts may differ.
Credit Risk
Credit risk on financial instruments arises from the potential for counterparties to default on
their obligations to us. Recent economic events, including failures of financial service companies
and the related liquidity crisis, have considerably disrupted the capital and credit markets. Our
credit risk consists of trade receivables, cash and cash equivalents, short-term investments,
derivative instruments, lending commitments and insurance relationships in the ordinary course of
business.
41
Table of Contents
The carrying value of accounts receivable approximates fair value due to the relatively short
periods to maturity on these instruments. Accounts receivable are primarily due from hospitals and
clinics located mainly in the United States and Western Europe. Although we do not require
collateral from our customers, concentrations of credit risk in the United States are mitigated by
a large number of geographically dispersed customers. We do not presently anticipate losses in
excess of allowances provided associated with trade receivables, although collection could be
impacted by the underlying economies of the countries.
We place cash, cash equivalents, short-term investments and derivative instruments with high
quality financial institutions, which we monitor regularly and take action where possible to
mitigate risk. We do not hold investments in auction rate securities, mortgage backed securities,
collateralized debt obligations, individual corporate bonds, special investment vehicles or any
other investments which have been directly impacted by the recent worldwide financial crisis. We
have not incurred any charges specific to the increased volatility in credit markets and credit
risk. Insurance programs are with carriers that remain highly rated and we have no significant
pending claims. We do not expect our current or future credit risk exposures to have a significant
impact on our operations. However, there can be no assurance that our business will not experience
any adverse impact from credit risk in the future.
To date, all previous lending commitments remain available to us. The administrative agent on our
Credit Facility was in Chapter 11 bankruptcy protection from November 1, 2009 until December 10,
2009. The administrative agent on our Credit Facility also provides $25 million of our $65 million
revolving credit facility. During the bankruptcy reorganization, there was no interruption in the
administrative agent’s service level, and our entire $65 million revolver commitment remains
available.
Inflation
We do not believe that inflation has had a material effect on our results of operations in recent
years and periods. There can be no assurance, however, that our business will not be adversely
affected by inflation in the future.
Item 8. Financial Statements and Supplementary Data
Our Consolidated Financial Statements and the reports of our independent registered public
accounting firm are included in this Annual Report on Form 10-K beginning on page F-1. The index to
this report and the financial statements are included in Part IV, Item 15.
Item 9. Changes In and Disagreements with Accountants on Accounting and Financial Disclosure
None
Item 9A. Controls and Procedures
Disclosure Controls and Procedures
Under the supervision and with the participation of our management, including our Chief Executive
Officer (CEO) and Chief Financial Officer (CFO), we evaluated the effectiveness of the design and
operation of our disclosure controls and procedures (as defined in Rule 13a-15(e) under the
Exchange Act of 1934). Based on that evaluation, our CEO and CFO concluded that our disclosure
controls and procedures were effective as of January 2, 2010.
Changes in Internal Control over Financial Reporting
There were no changes in our internal control over financial reporting identified in connection
with the above-referenced evaluation by management of the effectiveness of our internal control
over financial reporting that occurred during our fourth quarter ended January 2, 2010.
Management’s Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining effective internal control over
financial reporting. Our internal control over financial reporting is designed to provide
reasonable assurance to our management and our Board of Directors regarding the preparation and
fair presentation of published financial statements.
42
Table of Contents
Because of its inherent limitations, internal control over financial reporting may not prevent or
detect misstatements. Therefore, even those systems determined to be effective can provide only
reasonable assurance with respect to financial statement preparation and presentation.
Management assessed the effectiveness of our internal control over financial reporting as of
January 2, 2010. In making this assessment, we used the criteria set forth by the Committee of
Sponsoring Organizations of the Treadway Commission (COSO) in Internal Control – Integrated
Framework. Based on our assessment, we believe that, as of January 2, 2010, our internal control
over financial reporting is effective based on those criteria.
Our internal control over financial reporting as of January 2, 2010, has been audited by Ernst &
Young LLP, the independent registered public accounting firm who also audited our consolidated
financial statements, as stated in their report which appears on page F-2 of this Form 10-K.
Item 9B. Other Information
None
43
Table of Contents
PART III
Item 10. Directors and Executive Officers of the Registrant
Directors of the Registrant
The information in the “Election of Directors — Information About the Nominees and Other Directors”
section of our 2010 Proxy Statement is incorporated in this Annual Report on Form 10-K by
reference.
Executive Officers of the Registrant
Information about our executive officers is included in this Annual Report on Form 10-K under Part
I, Item 1, “Executive Officers of American Medical Systems Holdings, Inc.” and incorporated herein
by reference.
Compliance with Section 16(a) of the Exchange Act
The information in the “Section 16(a) Beneficial Ownership Reporting Compliance” section of our
2010 Proxy Statement is incorporated in this Annual Report on Form 10-K by reference.
Audit Committee Financial Expert
The information under the heading “Audit Committee” in the “Election of Directors — Board and Board
Committees” section of our 2010 Proxy Statement is incorporated in this Annual Report on Form 10-K
by reference.
Identification of the Audit Committee
The information under the heading “Audit Committee” in the “Election of Directors — Board and Board
Committees” section of our 2010 Proxy Statement is incorporated in this Annual Report on Form 10-K
by reference.
Code of Ethics
Our Code of Ethics for Senior Financial Management applies to our chief executive officer, chief
financial officer, controller, and other employees performing similar functions that have been
identified by the chief executive officer, and meets the requirements of the Securities and
Exchange Commission. We have posted our Code of Ethics for Senior Financial Management on our
website at www.AmericanMedicalSystems.com. We intend to disclose any amendments to and any waivers
from a provision of our Code of Ethics for Senior Financial Management on our website within five
business days following such amendment or waiver. The information contained in or connected to our
website is not incorporated by reference into this Form 10-K and should not be considered part of
this or any report that we file with or furnish to the Securities and Exchange Commission.
Item 11. Executive Compensation
The information in the “Compensation Discussion and Analysis,” the “Executive Compensation” and the
“Director Compensation” sections of our 2010 Proxy Statement is incorporated in this Annual Report
on Form 10-K by reference.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder
Matters
The information in the “Executive Compensation—Securities Authorized for Issuance Under Equity
Compensation Plans” and “Principal Stockholders and Management Beneficial Ownership” sections of
our 2010 Proxy Statement is incorporated in this Annual Report on Form 10-K by reference.
Item 13. Certain Relationships and Related Transactions, and Director Independence
The information in the “Related Person Relationships and Transactions,” the “Election of Directors
– Information about the Nominees and Other Directors” and the “Election of Directors – Board and
Board Committees” sections of our 2010 Proxy Statement is incorporated in this Annual Report on
Form 10-K by reference.
Item 14. Principal Accountant Fees and Services
The information in the “Audit and Non-Audit Fees” section of our 2010 Proxy Statement is
incorporated in this Annual Report on Form 10-K by reference.
44
Table of Contents
PART IV
Item 15. Exhibits and Financial Statement Schedule
(a) Financial Statements
Our following Consolidated Financial Statements and Reports of Independent Registered Public
Accounting Firm thereon are included herein (page numbers refer to pages in this Annual Report on
Form 10-K).
| F-1 | ||||
| F-3 | ||||
| F-4 | ||||
| F-5 | ||||
| F-6 | ||||
| F-7 |
(b) Financial Statement Schedule
Our schedule of valuation and qualifying accounts (in thousands) should be read in conjunction with
the consolidated financial statements (page numbers refer to pages in the Annual Report on Form
10-K). All other schedules are omitted because they are not applicable or the required information
is shown in the financial statements or notes thereto.
(c) Exhibits
The exhibits to this Annual Report on Form 10-K are listed in the Exhibit Index on pages E-1 to E-7
to this report. A copy of any of the exhibits listed in the Exhibit Index will be sent at a
reasonable cost to any stockholder upon receipt from any such person of a written request for any
such exhibit. Requests should be sent to the attention of Corporate Secretary, American Medical
Systems Holdings, Inc., 10700 Bren Road West, Minnetonka, Minnesota 55343.
The following is a list of each management contract or compensatory plan or arrangement required to
be filed as an exhibit (or incorporated by reference) to this Annual Report on Form 10-K:
| 1. | Employment Agreement, dated December 18, 2006, between Mark A. Heggestad and American Medical Systems, Inc. | ||
| 2. | First Amendment to Employment Agreement, dated March 6, 2008, between Mark A. Heggestad and American Medical Systems, Inc. | ||
| 3. | Employment Agreement, dated as of April 22, 2008, between American Medical Systems, Inc. and Anthony P. Bihl, III. | ||
| 4. | Employment Agreement, dated October 20, 2008, between Francois Georgelin and American Medical Systems Europe B.V. | ||
| 5. | Employment Agreement, effective as of April 1, 2009, between Joe W. Martin and American Medical Systems, Inc. | ||
| 6. | Employment Agreement, effective as of August 3, 2009, between Maximillian Fiore and American Medical Systems, Inc. | ||
| 7. | Separation Agreement, dated June 9, 2009, by and between Jan Dick and American Medical Systems, Inc. |
45
Table of Contents
| 8. | 2000 Equity Incentive Plan, as amended. | ||
| 9. | Form of Incentive Stock Option Agreement under the 2000 Equity Incentive Plan, as amended. | ||
| 10. | Form of Non-Qualified Stock Option Agreement under the 2000 Equity Incentive Plan, as amended. | ||
| 11. | Employee Stock Purchase Plan as amended. | ||
| 12. | 2005 Stock Incentive Plan (As Amended and Restated). | ||
| 13. | Form of Stock Option Certificate for Directors under the 2005 Stock Incentive Plan. | ||
| 14. | Form of Stock Option Certificate for Executive Officers under the 2005 Stock Incentive Plan. | ||
| 15. | Form of Notice of Amendment to Stock Option Certificate/Agreement for Executive Officers of American Medical Systems Holdings, Inc. | ||
| 16. | Form of Stock Option Certificate for Executive Officers under the 2005 Stock Incentive Plan (Version Modified in 2010). | ||
| 17. | Form of Restricted Stock Award for Executive Officers under the 2005 Stock Incentive Plan. | ||
| 18. | Form of Indemnification Agreement with Executive Officers and Directors. | ||
| 19. | Form of Change in Control Severance Agreement. | ||
| 20. | Form of First Amendment to Change in Control Severance Agreement. | ||
| 21. | Form of Change in Control Severance Agreement (Version Modified in 2009). | ||
| 22. | Change in Control Severance Agreement, dated as of April 22, 2008, between American Medical Systems Holdings, Inc. and Anthony P. Bihl, III. | ||
| 23. | The American Medical Systems, Inc. Executive Severance Pay Plan | ||
| 24. | Summary of Director Compensation. | ||
| 25. | Summary of Named Executive Officer Compensation (2009). | ||
| 26. | Summary of Named Executive Officer Compensation (2010). | ||
| 27. | 2010 Executive Variable Incentive Plan. |
46
Table of Contents
FINANCIAL STATEMENTS AND NOTES THERETO
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM ON CONSOLIDATED
FINANCIAL STATEMENTS
FINANCIAL STATEMENTS
The Board of Directors and Stockholders
American Medical Systems Holdings, Inc.
American Medical Systems Holdings, Inc.
We have audited the accompanying consolidated balance sheets of American Medical Systems Holdings,
Inc. as of January 2, 2010 and January 3, 2009, and the related consolidated statements of
operations, changes in stockholders’ equity, and cash flows for each of the three years in the
period ended January 2, 2010. Our audits also included the financial statement schedule listed in
Item 15(b). These financial statements and schedule are the responsibility of the Company’s
management. Our responsibility is to express an opinion on these financial statements and schedule
based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight
Board (United States). Those standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are free of material misstatement. An
audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the
financial statements. An audit also includes assessing the accounting principles used and
significant estimates made by management, as well as evaluating the overall financial statement
presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material
respects, the consolidated financial position of American Medical Systems Holdings, Inc. at January
2, 2010 and January 3, 2009, and the consolidated results of its operations and its cash flows for
each of the three years in the period ended January 2, 2010, in conformity with U.S. generally
accepted accounting principles. Also, in our opinion, the related financial statement schedule,
when considered in relation to the basic financial statements taken as a whole, presents fairly, in
all material respects, the information set forth therein.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight
Board (United States), American Medical Systems Holdings, Inc.’s internal control over financial
reporting as of January 2, 2010, based on criteria established in Internal Control-Integrated
Framework, issued by the Committee of Sponsoring Organizations of the Treadway Commission and our
report dated March 1, 2010, expressed an unqualified opinion thereon.
/s/ Ernst & Young LLP
Minneapolis, Minnesota
March 1, 2010
March 1, 2010
F-1
Table of Contents
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM ON INTERNAL
CONTROL OVER FINANCIAL REPORTING
CONTROL OVER FINANCIAL REPORTING
The Board of Directors and Stockholders
American Medical Systems Holdings, Inc.
American Medical Systems Holdings, Inc.
We have
audited American Medical Systems Holdings, Inc.’s internal control over financial reporting
as of January 2, 2010, based on criteria established in Internal Control-Integrated Framework
issued by the Committee of Sponsoring Organizations of the Treadway Commission (the COSO criteria).
American Medical Systems Holdings, Inc.’s management is responsible for maintaining effective
internal control over financial reporting and for its assessment of the effectiveness of internal
control over financial reporting included in the accompanying Report of Management on Internal
Control Over Financial Reporting. Our responsibility is to express an opinion on the company’s
internal control over financial reporting based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight
Board (United States). Those standards require that we plan and perform the audit to obtain
reasonable assurance about whether effective internal control over financial reporting was
maintained in all material respects. Our audit included obtaining an understanding of internal
control over financial reporting, assessing the risk that a material weakness exists, testing and
evaluating the design and operating effectiveness of internal control based on the assessed risk,
and performing such other procedures as we considered necessary in the circumstances. We believe
that our audit provides a reasonable basis for our opinion.
A company’s internal control over financial reporting is a process designed to provide reasonable
assurance regarding the reliability of financial reporting and the preparation of financial
statements for external purposes in accordance with generally accepted accounting principles. A
company’s internal control over financial reporting includes those policies and procedures that (1)
pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the
transactions and dispositions of the assets of the company; (2) provide reasonable assurance that
transactions are recorded as necessary to permit preparation of financial statements in accordance
with generally accepted accounting principles, and that receipts and expenditures of the company
are being made only in accordance with authorizations of management and directors of the company;
and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized
acquisition, use, or disposition of the company’s assets that could have a material effect on the
financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or
detect misstatements. Also, projections of any evaluation of effectiveness to future periods are
subject to the risk that controls may become inadequate because of changes in conditions, or that
the degree of compliance with the policies or procedures may deteriorate.
In our opinion, American Medical Systems Holdings, Inc. maintained, in all material respects,
effective internal control over financial reporting as of January 2, 2010, based on the COSO
criteria.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight
Board (United States), the consolidated balance sheets of American
Medical Systems Holdings, Inc. as
of January 2, 2010, and January 3, 2009, and the related consolidated statements of operations,
changes in stockholders’ equity, and cash flows for each of the three years in the period ended
January 2, 2010 of American Medical Systems Holdings, Inc., and our report dated March 1, 2010, expressed an unqualified opinion thereon.
/s/ Ernst & Young LLP
Minneapolis, Minnesota
March 1, 2010
March 1, 2010
F-2
Table of Contents
American Medical Systems Holdings, Inc.
Consolidated Statements of Operations
(In thousands, except per share data)
| 2009 | 2008 | 2007 | ||||||||||
Net sales |
$ | 519,270 | $ | 501,641 | $ | 463,928 | ||||||
Cost of sales |
92,211 | 111,097 | 105,592 | |||||||||
Gross profit |
427,059 | 390,544 | 358,336 | |||||||||
Operating expenses |
||||||||||||
Marketing and selling |
173,854 | 175,670 | 169,495 | |||||||||
Research and development |
52,765 | 46,247 | 43,315 | |||||||||
In-process research and development |
— | 7,500 | 7,500 | |||||||||
General and administrative |
45,196 | 39,281 | 43,070 | |||||||||
Integration costs |
— | — | 1,103 | |||||||||
Litigation settlement |
— | — | 14,303 | |||||||||
Amortization of intangibles |
13,161 | 34,465 | 18,264 | |||||||||
Total operating expenses |
284,976 | 303,163 | 297,050 | |||||||||
Operating income |
142,083 | 87,381 | 61,286 | |||||||||
Other (expense) income |
||||||||||||
Royalty income |
3,073 | 4,474 | 5,028 | |||||||||
Interest income |
175 | 747 | 1,153 | |||||||||
Interest expense |
(19,636 | ) | (27,398 | ) | (37,760 | ) | ||||||
Amortization of financing costs |
(15,790 | ) | (18,482 | ) | (16,145 | ) | ||||||
Gain on extinguishment of debt |
10,125 | 5,631 | — | |||||||||
Gain on sale of non-strategic assets |
17,446 | — | — | |||||||||
Other (expense) income |
(1,441 | ) | (2,195 | ) | 3,071 | |||||||
Total other (expense) |
(6,048 | ) | (37,223 | ) | (44,653 | ) | ||||||
Income from continuing operations before income taxes |
136,035 | 50,158 | 16,633 | |||||||||
Provision for income taxes |
51,197 | 19,323 | 11,888 | |||||||||
Net income from continuing operations |
84,838 | 30,835 | 4,745 | |||||||||
Loss from discontinued operations, net of tax benefit |
— | — | (691 | ) | ||||||||
Net income |
$ | 84,838 | $ | 30,835 | $ | 4,054 | ||||||
Net income per share |
||||||||||||
Basic net income from continuing operations |
$ | 1.14 | $ | 0.42 | $ | 0.07 | ||||||
Discontinued operations, net of tax |
— | — | (0.01 | ) | ||||||||
Basic net income |
$ | 1.14 | $ | 0.42 | $ | 0.06 | ||||||
Diluted net income from continuing operations |
$ | 1.14 | $ | 0.42 | $ | 0.06 | ||||||
Discontinued operations, net of tax |
— | — | (0.01 | ) | ||||||||
Diluted net income |
$ | 1.14 | $ | 0.42 | $ | 0.06 | ||||||
Weighted average common shares used in calculation |
||||||||||||
Basic |
74,097 | 72,942 | 72,061 | |||||||||
Diluted |
74,675 | 73,899 | 73,593 | |||||||||
| The accompanying notes are an integral part of the consolidated financial statements. | ||||||||||||
F-3
Table of Contents
American Medical Systems Holdings, Inc.
Consolidated Balance Sheets
(In thousands, except share and per share data)
| January 2, 2010 | January 3, 2009 | |||||||
Assets |
||||||||
Current assets |
||||||||
Cash and cash equivalents |
$ | 30,670 | $ | 11,642 | ||||
Short term investments |
19,868 | 31,323 | ||||||
Accounts receivable, net |
102,590 | 93,078 | ||||||
Inventories, net |
30,276 | 38,500 | ||||||
Deferred income taxes |
14,870 | 12,908 | ||||||
Other current assets |
6,067 | 6,858 | ||||||
Total current assets |
204,341 | 194,309 | ||||||
Property, plant and equipment, net |
44,120 | 48,280 | ||||||
Goodwill |
690,899 | 690,097 | ||||||
Developed and core technology, net |
51,631 | 62,315 | ||||||
Other intangibles, net |
49,937 | 47,349 | ||||||
Other long-term assets, net |
6,223 | 2,147 | ||||||
Total assets |
$ | 1,047,151 | $ | 1,044,497 | ||||
Liabilities and Stockholders’ Equity |
||||||||
Current liabilities |
||||||||
Accounts payable |
$ | 9,114 | $ | 7,830 | ||||
Income taxes payable |
4,495 | 7,782 | ||||||
Accrued compensation expenses |
29,603 | 22,876 | ||||||
Accrued warranty expense |
2,293 | 3,287 | ||||||
Other accrued expenses |
25,760 | 21,535 | ||||||
Total current liabilities |
71,265 | 63,310 | ||||||
Long-term debt |
346,229 | 484,582 | ||||||
Deferred income taxes |
62,347 | 50,044 | ||||||
Long-term income taxes payable |
18,206 | 15,327 | ||||||
Long-term employee benefit obligations |
3,745 | 3,752 | ||||||
Total liabilities |
501,792 | 617,015 | ||||||
Stockholders’ equity |
||||||||
Common stock, par value $.01 per share; authorized 200,000,000 shares;
issued and outstanding: 74,715,839 shares at January 2, 2010 and
73,668,415 shares at January 3, 2009 |
747 | 737 | ||||||
Additional paid-in capital |
399,468 | 369,594 | ||||||
Accumulated other comprehensive income |
6,381 | 3,226 | ||||||
Retained earnings |
138,763 | 53,925 | ||||||
Total stockholders’ equity |
545,359 | 427,482 | ||||||
Total liabilities and stockholders’ equity |
$ | 1,047,151 | $ | 1,044,497 | ||||
The accompanying notes are an integral part of the consolidated financial statements.
F-4
Table of Contents
American Medical Systems Holdings, Inc.
Consolidated Statements of Changes in Stockholders’ Equity
(In thousands)
| Accumulated | ||||||||||||||||||||||||
| Common Stock | Additional | Other | ||||||||||||||||||||||
| Par | Paid-In | Retained | Comprehensive | |||||||||||||||||||||
| Shares | Value | Capital | Earnings | Income | Total | |||||||||||||||||||
Balances at December 30, 2006 |
71,059 | $ | 711 | $ | 321,024 | $ | 19,299 | $ | 4,155 | $ | 345,189 | |||||||||||||
Comprehensive income |
||||||||||||||||||||||||
Net income |
— | — | — | 4,054 | — | 4,054 | ||||||||||||||||||
Foreign currency translation adjustment, net of tax
of $0.3 million |
— | — | — | — | 3,251 | 3,251 | ||||||||||||||||||
Unrealized loss on available-for-sale securities, net of
tax benefit of $0.3 million |
— | — | — | — | (433 | ) | (433 | ) | ||||||||||||||||
Net change for post-retirement plan, net of tax |
— | — | — | — | (63 | ) | (63 | ) | ||||||||||||||||
Total comprehensive income |
6,809 | |||||||||||||||||||||||
Issuance of common stock: |
||||||||||||||||||||||||
Stock options exercised |
604 | 6 | 8,324 | — | — | 8,330 | ||||||||||||||||||
Employee stock purchase plan |
164 | 2 | 2,498 | — | — | 2,500 | ||||||||||||||||||
Restricted stock awards |
60 | — | — | — | — | — | ||||||||||||||||||
InnovaQuartz settlement |
372 | 4 | 7,371 | — | — | 7,375 | ||||||||||||||||||
Stock-based compensation |
— | — | 12,700 | — | — | 12,700 | ||||||||||||||||||
Income tax benefit from stock option plans |
— | — | 731 | — | — | 731 | ||||||||||||||||||
Adjustment to initially apply FASB Interpretation No. 48 |
— | — | — | (263 | ) | — | (263 | ) | ||||||||||||||||
Balances at December 29, 2007 |
72,259 | 723 | 352,648 | 23,090 | 6,910 | 383,371 | ||||||||||||||||||
Comprehensive income |
||||||||||||||||||||||||
Net income |
— | — | — | 30,835 | — | 30,835 | ||||||||||||||||||
Foreign currency translation adjustment, net of tax
of $0.3 million |
— | — | — | — | (1,768 | ) | (1,768 | ) | ||||||||||||||||
Change in derivatives, net of tax of $1.2 million |
— | — | — | — | (1,982 | ) | (1,982 | ) | ||||||||||||||||
Recognition of previously unrealized losses on
available-for-sale securities, net of tax of $0.3 million |
— | — | — | — | 433 | 433 | ||||||||||||||||||
Unrealized loss on available-for-sale securities,
net of tax of $0.1 million |
— | — | — | — | (102 | ) | (102 | ) | ||||||||||||||||
Net change for post-retirement plan,
net of tax of $0.1 million |
— | — | — | — | (265 | ) | (265 | ) | ||||||||||||||||
Total comprehensive income |
— | — | — | — | — | 27,151 | ||||||||||||||||||
Issuance of common stock: |
— | — | — | — | — | |||||||||||||||||||
Stock options exercised |
1,017 | 10 | 5,441 | — | — | 5,451 | ||||||||||||||||||
Employee stock purchase plan |
306 | 3 | 3,420 | — | — | 3,423 | ||||||||||||||||||
Restricted stock awards |
86 | 1 | — | — | — | 1 | ||||||||||||||||||
Stock-based compensation |
— | — | 8,818 | — | — | 8,818 | ||||||||||||||||||
Income tax benefit from stock option plans |
844 | 844 | ||||||||||||||||||||||
Repurchase of convertible notes, net of adjustments |
— | — | (1,577 | ) | — | — | (1,577 | ) | ||||||||||||||||
Balances at January 3, 2009 |
73,668 | $ | 737 | $ | 369,594 | $ | 53,925 | $ | 3,226 | $ | 427,482 | |||||||||||||
Comprehensive income |
||||||||||||||||||||||||
Net income |
— | — | — | 84,838 | — | 84,838 | ||||||||||||||||||
Foreign currency translation adjustment, net of tax
of $0.2 million |
— | — | — | — | 2,010 | 2,010 | ||||||||||||||||||
Change in derivatives, net of tax of $0.4 million |
— | — | — | — | 693 | 693 | ||||||||||||||||||
Unrealized gain on available-for-sale securities,
net of tax of $0.2 million |
— | — | — | — | 296 | 296 | ||||||||||||||||||
Net change for post-retirement plan,
net of tax of $0.1 million |
— | — | — | — | 156 | 156 | ||||||||||||||||||
Total comprehensive income |
— | — | — | — | — | 87,993 | ||||||||||||||||||
Issuance of common stock: |
— | — | — | — | — | |||||||||||||||||||
Stock options exercised |
716 | 7 | 7,381 | — | — | 7,388 | ||||||||||||||||||
Employee stock purchase plan |
271 | 2 | 2,800 | — | — | 2,802 | ||||||||||||||||||
Restricted stock awards |
61 | 1 | — | — | — | 1 | ||||||||||||||||||
Stock-based compensation |
— | — | 8,988 | — | — | 8,988 | ||||||||||||||||||
Income tax benefit from stock option plans |
296 | 296 | ||||||||||||||||||||||
Issuance of convertible notes, net of tax of $28.8 million |
47,585 | 47,585 | ||||||||||||||||||||||
Repurchase of convertible notes, net of adjustments |
— | — | (37,176 | ) | — | — | (37,176 | ) | ||||||||||||||||
Balances at January 2, 2010 |
74,716 | $ | 747 | $ | 399,468 | $ | 138,763 | $ | 6,381 | $ | 545,359 | |||||||||||||
The accompanying notes are an integral part of the consolidated financial statements.
F-5
Table of Contents
American Medical Systems Holdings, Inc.
Consolidated Statements of Cash Flows
(In thousands)
| 2009 | 2008 | 2007 | ||||||||||
Cash flows from operating activities |
||||||||||||
Net income |
$ | 84,838 | $ | 30,835 | $ | 4,054 | ||||||
Loss from discontinued operations, net of tax benefit |
— | — | (691 | ) | ||||||||
Income from continuing operations |
84,838 | 30,835 | 4,745 | |||||||||
Adjustments to reconcile net income from continuing operations
to net cash provided by operating activities: |
||||||||||||
Depreciation |
10,025 | 10,089 | 8,587 | |||||||||
Gain on extinguishment of debt |
(10,125 | ) | (5,631 | ) | — | |||||||
Gain on sale of non-strategic assets |
(17,446 | ) | — | — | ||||||||
Amortization of intangibles |
13,161 | 34,465 | 18,264 | |||||||||
Amortization of deferred financing costs |
15,790 | 18,482 | 16,145 | |||||||||
In-process research and development charges |
— | 7,500 | 7,500 | |||||||||
Non-cash impairment of available-for-sale securities |
— | 843 | — | |||||||||
Net payments for settlement of derivative contracts |
556 | 1,385 | — | |||||||||
Excess tax benefit from exercise of stock options |
(566 | ) | (1,498 | ) | (215 | ) | ||||||
Tax benefit on exercised stock option arrangements |
1,613 | 844 | 731 | |||||||||
Change in net deferred income taxes |
(74 | ) | (4,035 | ) | 7,951 | |||||||
Stock-based compensation |
8,988 | 8,942 | 12,398 | |||||||||
Changes in operating assets and liabilities, net of acquired amounts: |
||||||||||||
Accounts receivable |
(8,100 | ) | 10,978 | (9,828 | ) | |||||||
Inventories |
9,228 | 20,947 | (30,516 | ) | ||||||||
Accounts payable and accrued expenses |
13,395 | (13,842 | ) | (3,868 | ) | |||||||
Other assets |
1,513 | (4,547 | ) | 15,857 | ||||||||
Net cash provided by operating activities |
122,796 | 115,757 | 47,751 | |||||||||
Cash flows from investing activities |
||||||||||||
Purchase of property, plant and equipment |
(5,865 | ) | (6,101 | ) | (14,173 | ) | ||||||
Purchase of business, net of cash acquired |
— | — | (781 | ) | ||||||||
Disposal of business |
— | 4,691 | 22,116 | |||||||||
Sale of non-strategic assets, net |
18,982 | — | — | |||||||||
Purchase of investments in technology |
— | (7,500 | ) | (7,500 | ) | |||||||
Purchase of other intangibles |
(5,927 | ) | (1,352 | ) | (382 | ) | ||||||
Purchase of short-term investments |
(18,820 | ) | (70,505 | ) | (30,187 | ) | ||||||
Sale of short-term investments |
30,755 | 39,999 | 29,570 | |||||||||
Net payments for settlement of derivative contracts |
(556 | ) | (1,385 | ) | — | |||||||
Net cash provided by (used in) investing activities |
18,569 | (42,153 | ) | (1,337 | ) | |||||||
Cash flows from financing activities |
||||||||||||
Issuance of common stock |
10,190 | 8,874 | 10,830 | |||||||||
Excess tax benefit from exercise of stock options |
566 | 1,498 | 215 | |||||||||
Proceeds from short-term borrowings |
— | 12,000 | — | |||||||||
Repayments of short-term borrowings |
— | (12,000 | ) | — | ||||||||
Debt issuance costs |
(7,697 | ) | — | — | ||||||||
Payments on senior secured credit facility |
(103,510 | ) | (85,202 | ) | (50,069 | ) | ||||||
Repurchase of convertible senior subordinated notes |
(21,125 | ) | (23,373 | ) | — | |||||||
Net cash used in financing activities |
(121,576 | ) | (98,203 | ) | (39,024 | ) | ||||||
Cash provided by (used in) continuing operations |
19,789 | (24,599 | ) | 7,390 | ||||||||
Cash used in discontinued operations |
||||||||||||
Operating activities |
— | — | (691 | ) | ||||||||
Cash used in discontinued operations |
— | — | (691 | ) | ||||||||
Effect of currency exchange rates on cash |
(761 | ) | 2,197 | (1,706 | ) | |||||||
Net increase (decrease) in cash and cash equivalents |
19,028 | (22,402 | ) | 4,993 | ||||||||
Cash and cash equivalents at beginning of period |
11,642 | 34,044 | 29,051 | |||||||||
Cash and cash equivalents at end of period |
$ | 30,670 | $ | 11,642 | $ | 34,044 | ||||||
Supplemental disclosure |
||||||||||||
Cash paid for interest |
$ | 15,733 | $ | 37,646 | $ | 39,075 | ||||||
Cash paid (refunded) for taxes |
47,556 | 13,710 | (11,662 | ) | ||||||||
Stock issued to settle contingent liabilities under InnovaQuartz purchase agreement |
— | — | 7,375 | |||||||||
The accompanying notes are an integral part of the consolidated financial statements.
F-6
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
1. Business Description and Significant Accounting Policies
Business Description
American Medical Systems Holdings, Inc. manufactures and markets a broad line of proprietary
surgical products to urologists, gynecologists and urogynecologists for erectile restoration,
benign prostatic hyperplasia (BPH), male urethral stricture, urinary and fecal incontinence, and
pelvic floor repair.
As further discussed in Note 2, Discontinued Operations and Sale of Aesthetics Business, in 2007,
consistent with the plans announced with the Laserscope acquisition, we sold the Laserscope
aesthetics business. The results of operations for this business for the period prior to the sale,
which occurred January 16, 2007, are presented in the discontinued operations section of the
statements of operations for the year ended December 29, 2007. Unless otherwise noted, disclosures
of revenues and expenses in the Notes to Consolidated Financial Statements refer to continuing
operations only.
Principles of Consolidation
The consolidated financial statements include the accounts of American Medical Systems Holdings,
Inc. and its subsidiaries after elimination of inter-company transactions and accounts.
Accounting Periods
We have a 52-or 53-week fiscal year ending on the Saturday nearest December 31. Accordingly,
fiscal years 2009, 2008 and 2007 ended on January 2, 2010, January 3, 2009 and December 29, 2007,
respectively, and are identified herein as 2009, 2008 and 2007. Fiscal year 2008 had 53 weeks and
fiscal years 2009 and 2007 consisted of 52 weeks.
Cash and Cash Equivalents
For financial reporting purposes, we consider all highly liquid investments purchased with an
original maturity of three months or less to be cash equivalents. Our cash and cash equivalent
balances are primarily maintained in our operating accounts.
Short-Term Investments
We classify investments as available-for-sale securities and record them at fair value. Our
short-term investments consist of money market funds, mutual fund shares, short-term bonds and
publicly traded equity securities. Unrealized gains or losses, net of related income taxes, are
recorded in accumulated other comprehensive income in stockholders’ equity. Realized gains
(losses) from the sale of investments are recorded into income
in the period of sale. The following table summarizes the components of the balance of our short-term investments
(in thousands):
| January 2, 2010 | January 3, 2009 | |||||||||||||||
| Fair | Unrealized | Fair | Unrealized | |||||||||||||
| Description of Securities | Value | Gains | Value | Losses | ||||||||||||
Money market funds |
$ | 18,504 | $ | — | $ | 30,846 | $ | — | ||||||||
Mutual fund
shares and short-term bonds with a maturity of less than one year |
709 | — | 285 | — | ||||||||||||
Publicly traded equity securities |
655 | 299 | 192 | 164 | ||||||||||||
Total |
$ | 19,868 | $ | 299 | $ | 31,323 | $ | 164 | ||||||||
The publicly traded equity securities are comprised solely of the stock of Iridex Corporation.
For more information regarding the Iridex stock, refer to Note 2, Discontinued Operations and Sale
of Aesthetics Business.
F-7
Table of Contents
Concentration of Risks
Credit risk on financial instruments arises from the potential for counterparties to default on
their obligations to us. Recent economic events, including failures of financial service companies
and the related liquidity crisis, have considerably disrupted the capital and credit markets. Our
credit risk consists of trade receivables, cash and cash equivalents, short-term investments,
derivative instruments, lending commitments and insurance relationships in the ordinary course of
business.
The carrying value of accounts receivable approximates fair value due to the relatively short
periods to maturity on these instruments. Accounts receivable are primarily due from hospitals and
clinics located mainly in the United States and Western Europe. Although we do not require
collateral from our customers, concentrations of credit risk in the United States are mitigated by
a large number of geographically dispersed customers. We do not presently anticipate losses in
excess of allowances provided associated with trade receivables, although collection could be
impacted by the underlying economies of the countries.
We place cash, cash equivalents, short-term investments and derivative instruments with high
quality financial institutions, which we monitor regularly and take action where possible to
mitigate risk. We do not hold investments in auction rate securities, mortgage backed securities,
collateralized debt obligations, individual corporate bonds, special investment vehicles or any
other investments which have been directly impacted by the recent financial crisis. We have not
incurred any charges specific to the increased volatility in credit markets and credit risk.
Insurance programs are with carriers that remain highly rated and we have no significant pending
claims. We do not expect our current or future credit risk exposures to have a significant impact
on our operations. However, there can be no assurance that our business will not experience any
adverse impact from credit risk in the future.
To date, all previous lending commitments remain available to us. The administrative agent on our
Credit Facility was in Chapter 11 bankruptcy protection from November 1, 2009 until December 10,
2009. The administrative agent on our Credit Facility also provides $25 million of our $65 million
revolving credit facility. During the bankruptcy reorganization, there was no interruption in the
administrative agent’s service level, and our entire $65 million revolver commitment remains
available.
Allowance for Doubtful Accounts
We estimate the allowance for doubtful accounts by analyzing those accounts receivable that have
reached their due date and by applying rates based upon historical write-off trends and specific
account reserves. Accounts are written off sooner in the event of bankruptcy or other circumstances
that make further collection unlikely. When it is deemed probable that a customer account is
uncollectible, that balance is written off against the existing allowance. Bad debt expense was
$1.6 million, $1.8 million and $1.0 million in 2009, 2008 and 2007, respectively. The allowance for
doubtful accounts was $3.4 million and $3.5 million at January 2, 2010 and January 3, 2009,
respectively.
Inventories
Inventories are stated at the lower of cost or market value, determined on the first-in-first-out
method. On a quarterly basis, we evaluate inventories for obsolescence and excess quantities. The
evaluation includes analyses of inventory levels, historical loss trends, expected product lives,
product at risk of expiration, sales levels by product, and projections of future sales demand. We
reserve for inventory we consider obsolete. In addition, we record an allowance for inventory
quantities in excess of forecasted demand.
F-8
Table of Contents
Property, Plant and Equipment
Property, plant and equipment, and major system software are carried at cost less accumulated
depreciation. Depreciation is recorded using straight-line or accelerated methods over the
following estimated useful asset lives:
| Asset class | Useful lives | |||
Building |
15-30 years | |||
Machinery and equipment |
3-12 years | |||
Furniture, fixtures, and other |
3-12 years | |||
Software |
3-5 years | |||
Maintenance, repairs, and minor improvements are charged to expense as incurred. Significant
improvements are capitalized. To the extent that we experience changes in the usage of equipment or
invest in enhancements to equipment, the estimated useful lives of equipment may change in a future
period.
In-Process Research and Development
When we acquire another company, the purchase price is allocated, as applicable, between in-process
research and development (IPR&D), other identifiable intangible assets, tangible assets, and
goodwill. IPR&D is defined as the value assigned to those projects for which the related products
have not received regulatory approval and have no alternative future use. The amount of the
purchase price allocated to IPR&D and other intangible assets is determined by estimating the
future cash flows of each project or technology and discounting the net cash flows back to their
present values. The discount rate used is determined at the time of acquisition in accordance with
accepted valuation methods. The discount rate used in the valuation of IPR&D for the 2006
acquisition of Laserscope was estimated to be 16 percent to reflect the risk characteristics and
uncertainty related to the development and commercialization assumptions. Costs related to
manufacturing, distribution and marketing of the products are included in the projections. Also
included are the expected research and development and clinical and regulation expenses projected
to be incurred to bring the product to market. For IPR&D, these methodologies include
consideration of the risk of the project not achieving commercial feasibility.
The forecast data employed in the analysis of our various IPR&D charges was based upon internal
product level forecast and external market information. The forecast data and assumptions are
inherently uncertain and unpredictable. However, based upon the information available at this
time, we believe the forecast data and assumptions used are reasonable. These assumptions may be
incomplete or inaccurate, and no assurance can be given that unanticipated events and circumstances
will not occur. Unless otherwise noted, forecast and assumptions have not changed materially from
the date the appraisals were completed.
At the time of acquisition, we expect all acquired IPR&D will reach technological feasibility, but
there can be no assurance that the commercial viability of these projects will be achieved. The
nature of the efforts to develop the acquired technologies into commercially viable products
consists principally of planning, designing and conducting clinical trials necessary to obtain
regulatory approvals. The risks associated with achieving commercialization include, but are not
limited to, delay or failure to obtain regulatory approvals to conduct clinical trials, failure of
clinical trials, delay or failure to obtain required market clearances, and patent litigation. If
commercial viability were not achieved, we would not realize the original estimated financial
benefits expected for these projects. We fund all costs to complete IPR&D projects with internally
generated cash flows.
We recognized $7.5 million of expense for IPR&D in 2008 for a milestone payment related to our
acquisition of BioControl Medical, Ltd. (BioControl) for the in-process development of an
implantable electrical stimulation device to treat urge incontinence and interstitial cystitis.
There was a similar milestone payment of $7.5 million in 2007 related to BioControl. We remain
liable to make a third milestone payment of $10 million to BioControl if and when the payment
conditions are satisfied. In addition, we agreed to make certain other payments in the event we
transfer the BioControl technology to another party prior to achieving the third milestone. These
additional contingent payments will be allocated to in-process research and development if made, as this was
the only asset acquired in the BioControl acquisition.
F-9
Table of Contents
Goodwill
Goodwill is the excess of the purchase price over the fair value of the other net assets, including
IPR&D, of acquired businesses. Goodwill is not amortized, but is assigned to reporting units and
tested for impairment annually during the fourth quarter, or whenever there is an impairment
indicator. We operate as one reporting unit engaged in developing, manufacturing, and marketing
medical devices. We assess goodwill impairment indicators quarterly, or more frequently, if a
change in circumstances or the occurrence of events suggests the remaining value may not be
recoverable.
The first step of the impairment test for goodwill compares the fair value of a reporting unit with
its carrying amount, including goodwill and other indefinite lived intangible assets. If the fair
value is less than the carrying amount, the second step determines
the amount of impairment, if any, by
comparing the implied fair value of the goodwill with the carrying amount of that goodwill. An
impairment charge is recognized only when the calculated fair value of a reporting unit, including
goodwill and indefinite lived intangible assets, is less than its carrying amount. Refer to Note
5, Goodwill and Intangible Assets, for discussion of our results of impairment testing.
Other Intangible Assets
Other intangible assets consist primarily of purchased technology, patents, and trademarks and are
generally amortized using the straight-line method over their estimated useful lives. We review our
intangible assets for impairment annually or as changes in circumstance or the occurrence of events
suggest the remaining value may not be recoverable. Intangible assets with indefinite lives are not
amortized, but are tested for impairment annually during the fourth quarter or whenever there is an
impairment indicator. Impairment, if any, is recognized through acceleration of amortization and
recorded as amortization of intangibles. Refer to Note 5, Goodwill and Intangible Assets, for
discussion of our results of impairment testing.
Long-Lived Assets
We record impairment losses on long-lived assets used in operations when events and circumstances
indicate the assets may be impaired and the undiscounted cash flows estimated to be generated by
those assets are less than the carrying amount of those assets. Periodically, if an indicator of
impairment exists, we measure any potential impairment utilizing discounted cash flows as an
estimate of fair value. Refer to Note 5, Goodwill and Intangible Assets, for discussion of our
results of impairment testing.
Revenue Recognition
We sell our products primarily through a direct sales force. A portion of our revenue is generated
from consigned inventory or from inventory with field representatives. For these products, revenue
is recognized at the time the product has been used or implanted. For all other transactions, we
recognize revenue when title to the goods and risk of loss transfer to our customers providing
there are no remaining performance obligations required from us or any matters requiring customer
acceptance. In cases where we utilize distributors or ship product directly to the end user, we
recognize revenue upon shipment provided all revenue recognition criteria have been met. We record
estimated sales returns, discounts and rebates as a reduction of net sales in the period
revenue is recognized.
Occasionally, sales of capital equipment have post-sale obligations, such as installation and
extended service contracts, which are fulfilled after product shipment. For each multiple element
arrangement, we determine if each element is a separate unit of accounting by ensuring that (1) the
element has stand alone value to the customer, (2) there is objective evidence of the fair value
for the element, and (3) if the arrangement includes a general right of return relative to the
delivered item, delivery of the undelivered items is considered probable and in our control. To
determine the fair value for each element in an arrangement, we rely primarily upon vendor specific
objective evidence (VSOE) of fair value using the price charged when we sell that element
separately, or in the case of hardware that we do not sell separately, we rely upon vendor
objective evidence of fair value in the form of competitor pricing of the same or interchangeable products. We defer revenue attributable to the
post-shipment obligations and recognize such revenue when the obligation is fulfilled.
We provide incentives to customers, including volume based rebates. Customers are not required to
provide documentation that would allow us to reasonably estimate the fair value of the benefit
received and we do not
receive an identifiable benefit in exchange for the consideration.
Accordingly, the incentives are recorded as a reduction of revenue.
F-10
Table of Contents
All of our customers have rights of return for the occasional ordering or shipping error. We
maintain an allowance for these returns and reduce reported revenue for expected returns from
shipments during each reporting period. This allowance is based on historical and current trends in
product returns. This allowance was $2.1 million and $2.6 million at January 2, 2010 and January 3,
2009, respectively.
Royalty Income
Royalties from licensees are based on third-party sales of licensed products and are recorded as
other income in accordance with contract terms when third-party results are reliable, measurable,
and collectibility is reasonably assured. Royalty estimates are made in advance of amounts
collected using historical and forecasted trends.
Research and Development
Research and development costs are expensed as incurred and include costs of all research
activities as well as other engineering and technical effort, including clinical and regulatory
activities, required to develop a new product or make significant improvement to an existing
product or manufacturing process.
Advertising and Promotional Costs
Advertising and promotional costs are charged to operations in the year incurred. Advertising and
promotion costs charged to operations during 2009, 2008 and 2007 were $5.9 million, $5.2 million
and $6.3 million, respectively.
Product Warranty Costs
We provide a warranty allowance to cover the cost of replacements for our erectile restoration, BPH
therapy, urinary stones, and incontinence products. The warranty allowance is an estimate of the
future cost of honoring our warranty obligations. Warranty costs are included as part of the cost
of goods sold.
We warrant all of our products to be free from manufacturing defects. In addition, if a product
fails, we may provide replacements at no cost or a substantial discount from list price. When we
sell products, we record an expense for the expected costs of future warranty-related claims, and
increase the warranty allowance by an equivalent amount. We reduce the warranty allowance by the
cost of the replacement device when an actual claim is awarded.
Product Liability
We estimate the uninsured portion of legal representation and settlement costs of product liability
claims and lawsuits quarterly. This evaluation consists of reviewing historical claims costs as
well as assessing future trends in medical device liability cases. Social and political factors, as
well as surgeon and medical facility responsibility, make litigation costs hard to predict. If, in
the future, we determine that our accrual is inadequate, the adjustment would reduce reported
income in the period we recorded the adjustment.
Software Development Costs
We capitalize certain costs incurred in connection with developing or obtaining software for
internal use. The net book value of capitalized software costs was $6.9 million as of January 2,
2010 and $6.5 million as of January 3, 2009. Depreciation expense on capitalized software cost was
$2.4 million, $2.4 million, and $2.2 million for 2009, 2008 and 2007, respectively.
Capitalized Interest
We capitalize interest cost as part of the historical cost of construction of certain facilities
and development of certain software up to the date the asset is ready for its intended use. We had
no capitalized interest in 2008 and 2009. Capitalized interest was $0.4 million in 2007.
F-11
Table of Contents
Income Taxes
We account for income taxes using the liability method. With this method, deferred tax assets and
liabilities are recorded based on the differences between the tax basis of assets and liabilities
and their carrying amounts for financial reporting purposes using enacted tax rates in effect in
the years in which the differences are expected to reverse.
We have significant amounts of deferred tax assets that are reviewed for recoverability and then
valued accordingly. We evaluate the realizable value of the deferred tax assets on a quarterly and
yearly basis, as well as assess the need for valuation allowances by considering historical levels
of income, estimates of future taxable income, and the impact of tax planning strategies. We record
a valuation allowance to reduce deferred tax assets when we believe all or part of our deferred tax
assets will not be realized.
We recognize tax positions in our financial statements when it is more likely than not the position
will be sustained upon examination based on the technical merits of the position.
Foreign Currency Translation
The financial statements for operations outside the United States are maintained in their local
currency. All assets and liabilities of our international subsidiaries are translated to United
States dollars at year-end exchange rates, while elements of the statement of operations are
translated at average exchange rates in effect during the year. Translation adjustments arising
from the use of differing exchange rates are included in accumulated other comprehensive income in
stockholders’ equity with the exception of inter-company balances not considered permanently
invested which are included in other income (loss). The balance of cumulative translation
adjustments included in accumulated other comprehensive income was $7.5 million and $5.5 million at
January 2, 2010 and January 3, 2009, respectively. Gains and losses on foreign currency
transactions are also included in other income (loss).
Derivatives and Hedging Activities
All derivatives are recorded on the consolidated balance sheet at their fair value. Changes in the
fair value of derivatives are recorded each period in earnings or other comprehensive income (loss)
(OCI) depending on the type of hedging instrument and the effectiveness of those hedges. See Note
11, Derivative Instruments and Hedging Activities for a description of our derivative instruments
and hedging activities during 2009 and 2008. We had no derivative instruments or hedging
activities in 2007.
Use of Estimates
The preparation of financial statements in conformity with accounting principles generally accepted
in the United States (U.S. GAAP) requires management to make estimates and assumptions that affect
the reported amounts of assets and liabilities, the disclosure of contingent assets and
liabilities, and the reported amounts of revenues and expenses during the reporting period. Actual
results could differ from these estimates. The most significant areas which require management’s
estimates relate to the allowances for doubtful accounts receivable, sales return reserve, excess
and obsolete inventories, impairment testing of long-lived assets, product warranty, product
liability claims, valuation of share-based payments and income taxes. We are subject to risks and
uncertainties, such as changes in the health care environment, competition, and legislation that
may cause actual results to differ from estimated results.
Stock-Based Compensation
We measure stock-based compensation expense based on the fair value of the award on the date of
grant. We recognize compensation expense for the fair value of restricted stock grants issued
based on the closing stock price
on the date of grant. Compensation expense recognized on shares issued under our Employee Stock
Purchase Plan is based on the value of an option to purchase shares of our stock at a 15 percent
discount to the stock price.
F-12
Table of Contents
Net Income per Share
We present both basic and diluted net income per share amounts. Basic net income per share is
calculated by dividing net income by the weighted average number of common shares outstanding
during the year. Diluted net income per share is based upon the weighted average number of common
shares and dilutive common share equivalents outstanding during the year. Common share equivalents
include stock options and restricted stock awards under our employee stock plans and potential
issuances of stock under the assumed conversion of our Convertible Notes utilizing the treasury
stock method. For further information regarding our Convertible Notes, refer to Note 7, Debt.
Common share equivalents are excluded from the computation in periods in which they have an
anti-dilutive effect. Stock options and restricted stock awards for which the exercise price
exceeds the average market price over the period have an anti-dilutive effect on net income per
share and, accordingly, are excluded from the calculation. When there is a net loss, other
potentially dilutive securities are not included in the calculation of net loss per share since
their inclusion would be anti-dilutive. In addition, common share equivalents related to our
Convertible Notes are anti-dilutive when the market price of our stock is below the conversion
price of our Convertible Notes and, therefore, are excluded from the calculation.
The following table presents information necessary to calculate basic and diluted net income (loss)
per common share and common share equivalents for the years ended 2009, 2008 and 2007:
| (in thousands, except per share data) | 2009 | 2008 | 2007 | |||||||||
Net income from continuing operations |
$ | 84,838 | $ | 30,835 | $ | 4,745 | ||||||
Weighted-average shares outstanding for basic net income per share |
74,097 | 72,942 | 72,061 | |||||||||
Dilutive effect of stock options and restricted shares |
578 | 957 | 1,532 | |||||||||
Adjusted weighted-average shares outstanding for |
||||||||||||
diluted net income per share |
74,675 | 73,899 | 73,593 | |||||||||
Net income per share |
||||||||||||
Basic net earnings from continuing operations |
$ | 1.14 | $ | 0.42 | $ | 0.07 | ||||||
Diluted net earnings from continuing operations |
$ | 1.14 | $ | 0.42 | $ | 0.06 | ||||||
Employee stock options and restricted stock awards of 5,076,960, 5,521,429 and 4,003,247 for
fiscal 2009, 2008 and 2007, respectively, were excluded from the diluted net income per share
calculation because their effect would be anti-dilutive. In addition, our Convertible Notes were
excluded from the diluted net income per share calculation in 2007 through 2009 because the
conversion price was greater than the average market price of our stock during the periods.
Comprehensive Income
Comprehensive income is the sum of net income as reported and other comprehensive income. Other
comprehensive income (loss) resulted from foreign currency translation adjustments, gains (losses)
on derivative instruments qualifying as hedges, post-retirement plan liability adjustments, and
gains (losses) on available-for-sale investments. For more information on derivative instruments,
see Note 11, Derivative Instruments and Hedging Activities.
F-13
Table of Contents
The components of comprehensive income for 2009, 2008 and 2007, were as follows:
| (in thousands) | 2009 | 2008 | 2007 | |||||||||
Net income |
$ | 84,838 | $ | 30,835 | $ | 4,054 | ||||||
Foreign currency translation gain (loss),
net of taxes of ($245), ($206) and ($264),
respectively |
2,010 | (1,768 | ) | 3,251 | ||||||||
Fair value adjustment on derivatives designated as cash flow hedges,
net of taxes of $1,850 and $1,200, respectively |
(3,058 | ) | (1,982 | ) | — | |||||||
Reclassification adjustments on cash flow hedges settled and included
in net income, net of tax of ($2,270) |
3,751 | — | — | |||||||||
Recognition of previously unrealized losses on
available-for-sale securities, net of tax of ($263) |
— | 433 | — | |||||||||
Unrealized gain (loss) on available-for-sale securities,
net of taxes of ($179), $62 and $263, respectively |
296 | (102 | ) | (433 | ) | |||||||
Prior service cost for post-retirement plan,
net of taxes of ($23), $14 and $15, respectively |
39 | (24 | ) | (24 | ) | |||||||
Net gain (loss) for post-retirement plan,
net of taxes of ($71), $110 and $20, respectively |
117 | (241 | ) | (39 | ) | |||||||
Comprehensive income |
$ | 87,993 | $ | 27,151 | $ | 6,809 | ||||||
The after-tax components of accumulated other comprehensive income as of January 2, 2010,
January 3, 2009 and December 29, 2007, were as follows:
| Net Unrealized | ||||||||||||||||||||
| (Loss) on | ||||||||||||||||||||
| Derivative | Net Unrealized | |||||||||||||||||||
| Instruments | Post-retirement | Foreign Currency | (Loss) Gain on | Total Accumulated | ||||||||||||||||
| Qualifying as | Plan Liability | Translation | Available-for-sale | Other Comprehensive | ||||||||||||||||
| (in thousands) | Hedges | Adjustment | Adjustment | Investments | Income | |||||||||||||||
Balance at December 29, 2007 |
$ | — | $ | 85 | $ | 7,258 | $ | (433 | ) | $ | 6,910 | |||||||||
Balance at January 3, 2009 |
$ | (1,982 | ) | $ | (180 | ) | $ | 5,490 | $ | (102 | ) | $ | 3,226 | |||||||
Balance at January 2, 2010 |
$ | (1,289 | ) | $ | (24 | ) | $ | 7,500 | $ | 194 | $ | 6,381 | ||||||||
Recent Accounting Pronouncements
In June 2009, the Financial Accounting Standards Board (FASB) issued Statement of Financial
Accounting Standards No. 168, The FASB Accounting Standards Codification and the Hierarchy of
Generally Accepted Accounting Principles—a replacement of FASB Statement No. 162 (the
Codification). The Codification reorganized existing U.S. accounting and reporting standards into
a single source of authoritative accounting principles arranged by topic. The Codification
supersedes all existing U.S. accounting standards; all other accounting literature not included in
the Codification, except for rules and interpretive releases of the Securities and Exchange
Commission
(SEC), which are also sources of authoritative U.S. GAAP for SEC registrants, is considered
non-authoritative. The Codification was effective on a prospective basis for interim and annual
reporting periods ending after September 15, 2009. We adopted the Codification in our interim
period ended October 3, 2009. The adoption of the Codification changed our references to U.S. GAAP,
but it had no impact on our consolidated financial position or results of operations.
In December 2007, the FASB issued revised guidance for the accounting for business combinations.
The revised guidance, which is now a part of ASC 805, Business Combinations, established
requirements for how an acquirer recognizes and measures in its financial statements the
identifiable assets acquired, the liabilities assumed, any noncontrolling interests in the acquiree
and the goodwill acquired. Some of the key requirements relate to the accounting treatment for
certain specific acquisition related items including: (1) accounting for acquired IPR&D as an
indefinite-lived intangible asset until approved or discontinued rather than as an immediate
expense; (2)
F-14
Table of Contents
expensing acquisition costs rather than adding them to the cost of an acquisition; (3)
expensing restructuring costs in connection with an acquisition rather than adding them to the cost
of an acquisition; (4) including the fair value of contingent consideration at the date of an
acquisition in the cost of an acquisition; and (5) recording the fair value of contingent
liabilities that are more likely than not to occur at the date of an acquisition. The revised
guidance also includes new disclosure requirements. Generally, the effect of the accounting for
acquisitions will depend on future acquisitions, as we will apply its provisions to business
combinations occurring subsequent to January 4, 2009. In addition, the accounting for the
resolution of any tax uncertainties remaining related to previous acquisitions will be subject to
the provisions of the amended guidance in ASC 805, Business Combinations.
In April 2009, the FASB issued new guidance related to assets acquired and liabilities assumed in a
business combination that arise from contingencies. The new guidance, which is now a part of ASC
805, Business Combinations, applies to all assets acquired and liabilities assumed in a business
combination that arise from contingencies and requires the acquirer to recognize such assets and
liabilities if the acquisition date fair value can be determined during the measurement period. If
the acquisition date fair value cannot be determined during the measurement period, the asset or
liability should be recognized at the acquisition date if the following criteria are met: (1)
information available before the end of the measurement period indicates it is probable that an
asset existed or a liability had been incurred at the acquisition date, and (2) the amount of the
asset or liability can be reasonably estimated. The new guidance is effective for business
combinations for which the acquisition date is on or after the beginning of an entity’s fiscal year
that begins on or after December 15, 2008, and we will apply the new guidance to business
combinations occurring after January 4, 2009.
In March 2008, the FASB issued new guidance on the disclosure of derivative instruments and hedging
activities. The new guidance, which is now a part of ASC 815, Derivatives and Hedging Activities,
requires enhanced disclosures about an entity’s derivative and hedging activities in order to
improve the transparency of financial reporting. The provisions of the new guidance were effective
for fiscal years and interim periods beginning after November 15, 2008. We adopted the new
guidance in the first quarter of 2009. See Note 12, Derivative Instruments and Hedging Activities,
for additional information, including required disclosures. The adoption did not have a material
impact on our consolidated financial position or results of operations, as it is a disclosure only
standard.
Effective January 4, 2009, we adopted new FASB guidance on accounting for convertible debt. ASC
470, Debt, changes the balance sheet classification of a component of our convertible notes between
equity and debt, and results in additional non-cash economic interest cost which has been reflected
in the Consolidated Statement of Operations for all periods presented.
In April 2009, the FASB issued new guidance related to the disclosure of the fair value of
financial instruments. The new guidance, which is now a part of ASC 825, Financial Instruments,
requires fair value disclosures in both interim as well as annual financial statements in order to
provide more timely information about the effects of current market conditions on financial
instruments. The new guidance is effective for interim and annual periods ending after June 15,
2009. We adopted the new guidance for our interim period ending July 4, 2009. See Note 10, Fair
Value Measurements, for additional information and required disclosures. The adoption did not have
a material impact on our consolidated financial position or results of operations, as it is a
disclosure only standard.
In May 2009, the FASB issued new guidance for accounting for subsequent events. The new guidance,
which is now a part of ASC 855, Subsequent Events, establishes the accounting for, and disclosure
of, events that occur after the balance sheet date but before financial statements are issued or
are available to be issued.
In October 2009, the FASB issued Accounting Standards Update (ASU) 2009-13, Revenue Recognition
(Topic 605): Multiple-Deliverable Revenue Arrangements—a consensus of the FASB Emerging Issues Task
Force (ASU 2009-13). ASU 2009-13 will separate multiple-deliverable arrangements into more
circumstances than under existing U.S. GAAP and will establish a selling price hierarchy for
determining the selling price of a deliverable. In addition, it will replace the term “fair value”
in the revenue allocation guidance with “selling price” to clarify the allocation of revenue is
based on entity-specific assumptions rather than assumptions of a market place participant,
F-15
Table of Contents
eliminate the use of the residual method for allocation, and expand on-going disclosure
requirements. ASU 2009-13 is effective for fiscal years beginning on or after June 15, 2010 and
can be applied prospectively or retrospectively. We plan to adopt this updated accounting guidance
for multiple-deliverable revenue arrangements on a prospective basis for our fiscal year beginning
on January 2, 2011, and the adoption is not expected to have a material impact on our consolidated
financial position or results of operations.
2. Discontinued Operations and Sale of Aesthetics Business
On January 16, 2007, we sold Laserscope’s aesthetics business to Iridex Corporation (Iridex) for a
sale price consisting of $26.0 million of cash consideration and 213,435 shares of Iridex
unregistered common stock (subject to certain post-closing adjustments), and up to an additional
$9.0 million as determined by the book value of certain inventory following termination of a
manufacturing transition period of approximately six to nine months. In August 2007, we agreed
with Iridex on the amount and payment plan for the final post-closing purchase price adjustment and
an amount for the fair value of inventory to be purchased at the termination of the manufacturing
transition period. The agreement included an increase to the purchase price of $1.1 million and an
additional $4.1 million for the purchase of inventory. Pursuant to the payment plan, Iridex paid
these amounts in scheduled payments through the third quarter of 2008. During 2008, Iridex paid
approximately $2.6 million of receivables for which we had previously established reserves, which
we recorded in other income.
Included in short-term investments at January 2, 2010 and January 3, 2009 is $0.7 million and
$0.2 million, respectively, for the value of unregistered Iridex common stock received as partial
consideration for the sale of the Laserscope aesthetics business. The common stock is accounted for
as an available-for-sale security with changes in value recorded through other comprehensive
income, unless any decline in value below the current book value is considered
other-than-temporary, in which case resulting adjustments are recorded in earnings. In the first
quarter of 2008, we determined that our investment was other-than-temporarily impaired and we
recorded an impairment charge of $0.8 million. At January 2, 2010, the unrealized gain on the
stock is $0.3 million.
The financial results of the aesthetics business for the period prior to the sale, which occurred
January 16, 2007, are presented in the discontinued operations section of the statements of
operations. There was no discontinued operations activity in 2008 and 2009. The following table
represents the results of discontinued operations for the year ended December 29, 2007:
| (in thousands) | 2007 | |||
Net sales |
$ | 515 | ||
Loss from discontinued operations before income taxes |
(1,075 | ) | ||
Income tax benefit |
384 | |||
Loss from discontinued operations, net of taxes |
$ | (691 | ) | |
In conjunction with the sale of the aesthetics business, we entered into a supply agreement
with Iridex whereby we agreed to manufacture and supply to Iridex certain aesthetics devices during
a transition period. The agreement expired on October 16, 2007. Iridex reimbursed us for our cost
to produce the products. Certain of the final purchase orders under the supply agreement were
delivered after expiration of the agreement. Delivery was completed in the third quarter of 2008.
In addition, we also entered into an agreement with Iridex to provide administrative services at no
charge during a transition period of 60 days. We presented the results of operations of the
aesthetics business as discontinued operations because we believe that the cash flows under these
agreements were not significant and we had no significant continuing involvement in the operations
of the aesthetics business.
3. Litigation Settlements
We made cash payments of $15.0 million in 2008 for litigation settlements, primarily due to the
arbitration award to the former shareholders of CryoGen, Inc. On March 15, 2006, we received a
demand for arbitration by Robert A. Knarr, as shareholder representative, on behalf of the former
shareholders of CryoGen, Inc. On December 30, 2002, we acquired CryoGen, Inc. pursuant to the
Agreement and Plan of Merger, dated as of December 13, 2002, as
F-16
Table of Contents
amended, among our wholly-owned subsidiary, American Medical Systems, Inc., CryoGen, Inc. and
Robert A. Knarr, as shareholders’ representative. The arbitration demand alleged that we breached
the merger agreement by, among other things, failing to use commercially reasonable efforts to
promote, market and sell the Her Option System and by acting in bad faith and thereby negatively
impacting the former CryoGen shareholders’ right to an earnout payment under the merger agreement.
On December 18, 2007, the arbitration panel issued its decision in the arbitration proceeding, and
awarded the CryoGen shareholders an earnout payment. We recorded a charge to earnings for the
arbitration award in December 2007, and we paid the arbitration award in January 2008.
4. Balance Sheet Information
The following provides additional information (in thousands) concerning selected balance sheet
accounts:
| 2009 | 2008 | |||||||
Accounts receivable |
||||||||
Trade accounts receivable |
$ | 103,084 | $ | 95,297 | ||||
Other receivables |
2,945 | 1,317 | ||||||
Allowance for doubtful accounts |
(3,439 | ) | (3,536 | ) | ||||
Net accounts receivable |
$ | 102,590 | $ | 93,078 | ||||
Inventories |
||||||||
Raw materials |
$ | 10,117 | $ | 11,611 | ||||
Work in process |
3,399 | 4,841 | ||||||
Finished goods |
21,791 | 26,283 | ||||||
Obsolescence allowance |
(5,031 | ) | (4,235 | ) | ||||
Net inventories |
$ | 30,276 | $ | 38,500 | ||||
Property, plant and equipment |
||||||||
Land and building |
$ | 41,436 | $ | 41,002 | ||||
Machinery and equipment |
12,108 | 11,868 | ||||||
Software |
20,863 | 18,112 | ||||||
Furniture, fixtures, and other |
19,108 | 18,598 | ||||||
Accumulated depreciation |
(49,395 | ) | (41,300 | ) | ||||
Net property, plant and equipment |
$ | 44,120 | $ | 48,280 | ||||
Accrued compensation expenses |
||||||||
Accrued payroll |
$ | 5,090 | $ | 4,429 | ||||
Accrued bonuses and commissions |
14,031 | 10,594 | ||||||
Accrued vacation |
5,426 | 4,069 | ||||||
Accrued benefits and other compensation |
5,056 | 3,784 | ||||||
Total accrued compensation expenses |
$ | 29,603 | $ | 22,876 | ||||
Other accrued expenses |
||||||||
Accrued interest |
$ | 3,922 | $ | 213 | ||||
Deferred revenue |
2,685 | 2,183 | ||||||
Accrued other |
19,153 | 19,139 | ||||||
Total other accrued expenses |
$ | 25,760 | $ | 21,535 | ||||
Long-term employee benefit obligations |
||||||||
Accumulated postretirement benefit obligation |
$ | 3,631 | $ | 3,623 | ||||
Other long-term benefit obligations |
114 | 129 | ||||||
Total long-term employee benefit obligations |
$ | 3,745 | $ | 3,752 | ||||
F-17
Table of Contents
5. Goodwill and Intangible Assets
The changes in carrying amount of goodwill for 2009 and 2008 are as follows:
| (in thousands) | 2009 | 2008 | ||||||
| | | | ||||||||
Goodwill, beginning of the period |
$ | 690,097 | $ | 690,478 | ||||
Adjustments to goodwill from acquisitions |
— | 759 | ||||||
Effect of currency translation |
802 | (1,140 | ) | |||||
Goodwill, end of the period |
$ | 690,899 | $ | 690,097 | ||||
The additional goodwill during 2008 relates primarily to tax adjustments related to
preacquisition tax contingencies and tax basis differences.
Amortized trademarks relate to trademarks acquired as part of our Influence acquisition in 1999.
We are amortizing these trademarks over a remaining useful life of one year. The unamortized
trademarks relate to the GreenLight™ and StoneLight® trademarks acquired as part of our
acquisition of Laserscope in 2006 (the Laserscope Trademarks). The Laserscope Trademarks are
classified as indefinite-lived intangible assets and are not amortized because there is no
foreseeable limit on the period of time the Laserscope Trademarks are expected to contribute to our
cash flows. Definite-lived intangibles are being amortized over periods ranging from one to nine
years.
The following table provides additional information concerning intangible assets:
| January 2, 2010 | January 3, 2009 | |||||||||||||||||||||||||||
| Weighted avg | Gross | Gross | ||||||||||||||||||||||||||
| remaining life | carrying | Accumulated | Net book | carrying | Accumulated | Net book | ||||||||||||||||||||||
| (in thousands) | (years) | amount | amortization | value | amount | amortization | value | |||||||||||||||||||||
Developed and core technology |
5.6 | $ | 137,553 | $ | (85,922 | ) | $ | 51,631 | $ | 137,553 | $ | (75,238 | ) | $ | 62,315 | |||||||||||||
Other intangibles |
||||||||||||||||||||||||||||
Amortized |
||||||||||||||||||||||||||||
Patents |
5.9 | 11,510 | (9,693 | ) | 1,817 | 11,510 | (9,317 | ) | 2,193 | |||||||||||||||||||
Licenses |
7.6 | 15,913 | (9,034 | ) | 6,879 | 9,312 | (7,659 | ) | 1,653 | |||||||||||||||||||
Royalty agreement |
0.0 | — | — | — | 2,970 | (1,148 | ) | 1,822 | ||||||||||||||||||||
Trademarks |
1.0 | 2,208 | (1,767 | ) | 441 | 2,208 | (1,327 | ) | 881 | |||||||||||||||||||
Total
amortized other intangible assets |
7.0 | 29,631 | (20,494 | ) | 9,137 | 26,000 | (19,451 | ) | 6,549 | |||||||||||||||||||
Unamortized |
||||||||||||||||||||||||||||
Trademarks |
n/a | 40,800 | — | 40,800 | 40,800 | — | 40,800 | |||||||||||||||||||||
Total other intangibles |
70,431 | (20,494 | ) | 49,937 | 66,800 | (19,451 | ) | 47,349 | ||||||||||||||||||||
Total intangible assets |
$ | 207,984 | $ | (106,416 | ) | $ | 101,568 | $ | 204,353 | $ | (94,689 | ) | $ | 109,664 | ||||||||||||||
During the second quarter of 2009, we purchased a license for the exclusive rights to certain
patents through the year 2018 for $9 million, of which $5.9 million has been paid as of January 2,
2010, and the remaining $3.1 million is structured to be paid out within approximately two years,
in quarterly intervals. All payments related to the patents are included in licenses and will be
amortized over approximately eight years, which is the expected remaining
useful life of the patents.
During the third quarter of 2009, we sold our female sterilization assets and technology (Ovion
technology) to Conceptus, Inc. for $23.6 million. The consideration, less the carrying value of the
intangible asset and related disposal costs resulted in a pre-tax gain of $17.4 million, which is
included in “gain on sale of non-strategic assets” in the Consolidated Statements of Operations.
We review our intangible assets for impairment annually or as changes in circumstance or the
occurrence of events suggest the remaining value may not be recoverable. We did not recognize any
impairment of intangible assets in 2009.
F-18
Table of Contents
In the fourth quarter of 2008, upon performing our annual assessment of intangible assets, we
determined that the projected future cash flows related to certain acquired developed technology
intangible assets (for our TherMatrx® and GreenLight PV
® technology) had declined from our previous
expectations. The projected declines in cash flows are primarily due to changes in the BPH market
resulting from faster-than-expected customer conversion to the GreenLight HPS
® technology, which is
the next generation of technology compared to GreenLight PV
® technology. As a result of our
assessment, we concluded that the carrying value of the acquired developed technology intangible
assets exceeded the undiscounted expected cash flows. Therefore, during the fourth quarter of
2008, we performed a discounted cash flow (DCF) analysis, using the excess earnings method, to
adjust the asset carrying values to their estimated fair values.
In performing our DCF analysis in 2008, we made assumptions about the amount and timing of future
expected cash flows, terminal value growth rates, appropriate discount rates, contributory asset
charges and tax amortization benefits. The amount and timing of future cash flows within our DCF
analysis is based on our most recent budget, long range strategic plans and other estimates. The
terminal growth rate is used to calculate the value of cash flows beyond the last projected period
in our DCF analysis and reflects our best estimates for stable, perpetual growth of cash flows. We
use estimates of market participant weighted average cost of capital (WACC) as a basis for
determining the discount rate to apply to our expected future cash flows. The discount rate used
in our 2008 impairment analysis was 12.5 percent. In addition, an adjustment to the indicated
values was made to reflect the hypothetical tax benefits associated with amortizing an asset for
income tax purposes.
In 2008, based on the DCF analysis we recorded a $17.1 million impairment to adjust the asset
carrying values to their estimated fair value. The impairment was recognized through acceleration
of amortization and reported in the “Amortization of intangibles” line item in the Consolidated
Statements of Operations.
The following discloses actual and expected aggregate amortization expense for currently-owned
intangible assets (in thousands) for 2007 through 2014:
| Year | Actual | Expected | ||||||
2007 |
$ | 18,264 | $ | — | ||||
2008 |
34,465 | — | ||||||
2009 |
13,161 | — | ||||||
2010 |
— | 12,306 | ||||||
2011 |
— | 11,580 | ||||||
2012 |
— | 9,401 | ||||||
2013 |
— | 8,871 | ||||||
2014 |
— | 6,754 | ||||||
6. Warranties
Many of our products are sold with warranty coverage for periods ranging from one year up to the
patient’s lifetime. The warranty allowance is our estimate of the expected future cost of honoring
current warranty obligations. Factors
influencing this estimate include historical claim rates, surgical infection rates, changes in
product performance, the frequency of use by the patient, the patient’s performance expectations,
and changes in the terms of our policies. Changes in the warranty balance for 2009 and 2008 are
disclosed in the table below.
| (in thousands) | 2009 | 2008 | ||||||
Balance, beginning of period |
$ | 3,287 | $ | 3,001 | ||||
Provisions for warranty |
1,517 | 5,577 | ||||||
Claims processed |
(2,511 | ) | (5,291 | ) | ||||
Balance, end of period |
$ | 2,293 | $ | 3,287 | ||||
7. Debt
Senior Secured Credit Facility
On July 20, 2006, in conjunction with the Laserscope acquisition, our wholly-owned subsidiary,
American Medical Systems, Inc. (AMS), entered into a credit and guarantee agreement (the Credit
Facility) with CIT Healthcare LLC,
F-19
Table of Contents
as agent, and certain lenders from time to time party thereto
(the Lenders). AMS and each majority-owned domestic subsidiary of AMS are parties to the Credit
Facility as guarantors of all of the obligations of AMS arising under the Credit Facility. Each of
the subsidiary guarantors is 100 percent owned by us and the guarantees are joint and several. The
obligations of AMS and each of the guarantors arising under the Credit Facility are secured by a
first priority security interest granted to the agent on substantially all of their respective
assets, including a mortgage on the AMS facility in Minnetonka, Minnesota.
The six-year senior secured Credit Facility consists of (i) term loan debt and (ii) a revolving
credit facility of up to $65.0 million which is available to fund ongoing working capital needs,
including future capital expenditures and permitted acquisitions. During January 2008, we borrowed
$12.0 million under the revolving credit facility to fund the payment of certain litigation
settlements (refer to Note 3, Litigation Settlements). We repaid the outstanding balance with
operating cash in February 2008. As of January 2, 2010 and January 3, 2009, there were $125.3
million and $228.8 million, respectively, of term loans outstanding under the Credit Facility.
At our option, term loans under the Credit Facility (other than swing line loans) bear interest at
a variable rate based on LIBOR or an alternative variable rate based on the greater of the prime
rate or the federal funds effective rate plus 0.5 of 1.0 percent (Federal Funds Rate) plus an
applicable margin. The applicable margin for term loans based on LIBOR is 2.25 percent per annum,
while the applicable margin for term loans based on the prime rate or the Federal Funds Rate is
1.25 percent per annum. As of January 2, 2010, all debt under the Credit Facility had a variable
interest rate based on the LIBOR index. The applicable margin for loans under the revolving credit
facility is determined by reference to our total leverage ratio, as defined in the Credit Facility.
In addition to initial Credit Facility fees and reimbursement of agent expenses, we are obligated
to pay commitment fees on the revolving credit facility.
The term loans amortize 1.0 percent of the current principal balance quarterly from December 2006
through September 2011 and the remaining 95 percent will amortize December 2011 through July 2012.
In addition, mandatory prepayments are due under the Credit Facility equal to (i) 50 percent of
Excess Cash Flow (defined generally as net income, plus depreciation and amortization and other
non-cash charges including IPR&D, plus decreases or minus increases in working capital, minus
capital expenditures (to the extent not financed) and amortization payments with respect to the
term loan, and any other indebtedness permitted under the loan documents), (ii) 100 percent of the
net proceeds of any asset sale (subject to a limited reinvestment option and a $2.5 million
exception), (iii) 100 percent of the net proceeds of any debt (including convertible securities) or
preferred stock issuance, and (iv) 50 percent of the net proceeds of any other equity issuance.
Amounts due under the Credit Facility may also be voluntarily prepaid without premium or penalty.
Amortization and other prepayments of $103.5 million and $85.2 million were made during the year
January 2, 2010 and January 3, 2009, respectively.
The Credit Facility contains affirmative and negative covenants and other limitations (subject to
various carve-outs and baskets). The covenants limit: (a) the making of investments, the amount of
capital expenditures, the payment of dividends and other payments with respect to capital, the
disposition of material assets other than in the ordinary course of business, and mergers and
acquisitions under certain conditions, (b) transactions with affiliates unless such transactions
are completed in the ordinary course of business and upon fair and reasonable terms, (c) the
incurrence of liens and indebtedness, and (d) substantial changes in the nature of the companies’
business. The Credit Facility also contains financial covenants which require us to maintain
predetermined ratio levels related to leverage, interest coverage, fixed charges, and a limit on
capital expenditures. In addition, the Credit Facility contains customary events of default,
including payment and covenant defaults and material inaccuracy of representations. The Credit
Facility further permits the taking of customary remedial action upon the occurrence and
continuation of an event of default, including the acceleration of obligations then outstanding
under the Credit Facility.
Fees of $10.5 million are classified as debt discount and are being accreted to amortization of
financing costs using the effective interest method over a six year period. Additional debt
issuance costs of approximately $2.4 million are recorded as other long term assets and are being
amortized over six years using the straight-line method. Upon payment of the prepayments described
above, a pro rata portion of the related fees and debt issuance costs of $1.8 million and $2.0
million was immediately charged to amortization of financing costs in the years ending January 2,
2010 and January 3, 2009, respectively.
F-20
Table of Contents
The scheduled amortization payments under the Credit Facility are adjusted after each prepayment.
As of January 2, 2010, the amortization payments for the next five years are as follows (in
thousands):
Fiscal 2010 |
$ | 1,295 | ||
Fiscal 2011 |
31,732 | |||
Fiscal 2012 |
92,280 | |||
Fiscal 2013 |
— | |||
Fiscal 2014 |
— |
Amendment of Credit Facility
On August 12, 2009, we received a Consent and Second Amendment to our Credit Facility, which
allowed us to exchange a portion of our existing convertible senior subordinated notes for new
convertible senior subordinated notes as discussed below. On October 29, 2007, we entered into a
First Amendment of our Credit Facility to modify certain financial covenant ratios as defined in
the Credit Facility (the Amendment). Pursuant to the terms of the Amendment, certain of the
financial tests and covenants provided in Section 6.8 of the Credit Facility were amended and
restated, including the interest coverage ratio, the total leverage ratio, the fixed charge
coverage ratio, and the prior year maximum consolidated capital expenditures.
Convertible Senior Subordinated Notes Due 2036
On June 27, 2006, we issued convertible senior subordinated notes with a stated maturity of July 1,
2036 (the 2036 Notes). The 2036 Notes bear a fixed interest rate of 3.25 percent per year, payable
semiannually. The 2036 Notes are our direct, unsecured, senior subordinated obligations, rank
junior to the senior secured Credit Facility and will rank junior in right of payment to all of our
future senior secured debt as provided in the indenture for the 2036 Notes. The 2036 Notes have
the same rank as our convertible notes that are due in 2041, which are discussed below.
In March 2009, we repurchased 2036 Notes with a principal amount of $27.3 million in exchange for a
cash payment of $21.1 million. In connection with this transaction, we recorded a pre-tax gain on
extinguishment of debt of $4.6 million in the year ended January 2, 2010. In December 2008, we
repurchased 2036 Notes with a principal
amount of $34.5 million in exchange for a cash payment of $23.4 million. In connection with this
transaction, we recorded a pre-tax gain on extinguishment of debt of $5.6 million in the year ended
January 3, 2009.
On September 21, 2009, we exchanged $250.0 million in principal of the 2036 Notes for $250.0
million in principal of new convertible senior subordinated notes with a stated maturity of
September 15, 2041 (the 2041 Notes). We accounted for this transaction as an
extinguishment of debt, and we recorded a pre-tax gain on extinguishment of $5.6 million in the
year ended January 2, 2010. Further information on the 2041 Notes is provided following this
section.
We separately account for the liability and equity components of our 2036 Notes in a manner that
reflects our nonconvertible borrowing rate. The equity component of our 2036 Notes was $45.4
million and $105.0 million as of January 2, 2010 and January 3, 2009, respectively, and is recorded
in additional paid-in capital. As of January 2, 2010, the principal amount of the liability
component, its unamortized discount, and its net carrying amount were $62.0 million, $11.5 million
and $50.5 million, respectively. The unamortized discount will be amortized over a remaining
period of 3.5 years and the amortization expense is included in “amortization of financing costs”
on the Consolidated Statements of Operations. As of January 3, 2009, the principal amount of the
liability component, its unamortized discount, and its net carrying amount were $339.3 million,
$77.2 million and $262.1 million, respectively. The effective interest rate on the liability
component was 9.5% for each of the years ended January 2, 2010 and January 3, 2009. During the
year ended January 2, 2010, we recognized $8.0 million of interest expense representing the
contractual interest coupon on our 2036 Notes, and $10.4 million of amortization expense related to
the discount on the liability component. During the year ended January 3, 2009, we recognized
$12.2 million of interest expense representing the contractual interest coupon on our 2036 Notes,
and $14.7 million of amortization expense related to the discount on the liability component.
In addition to regular interest on the 2036 Notes, we will also pay contingent interest beginning
July 1, 2011 at 0.25% of the average trading price of the 2036 Notes, if the average trading
price for the five consecutive trading
F-21
Table of Contents
days immediately before the last trading day preceding the
relevant six-month period equals or exceeds 120 percent of the principal amount of the 2036 Notes.
Our 2036 Notes are convertible under the following circumstances for cash and shares of our common
stock, if any, at a conversion rate of 51.5318 shares of our common stock per $1,000 principal
amount of 2036 Notes (which is equal to an initial conversion price of approximately $19.406 per
share), subject to adjustment: (1) when, during any fiscal quarter, the last reported sale price of
our common stock is greater than 130% of the conversion price for at least 20 trading days in the
30 trading-day period ending on the last trading day of the preceding fiscal quarter; (2) during
the five trading days immediately after any five consecutive trading-day period in which the
trading price of a 2036 Note for each day of that period was less than 98% of the product of the
closing price of our common stock and the applicable conversion rate; (3) if specified
distributions to holders of our common stock occur; (4) if we call the 2036 Notes for redemption;
(5) if an event or change occurs that results in conversion according to the Indenture; or (6)
during the 60 days prior to, but excluding, any scheduled repurchase date or maturity date. Upon
conversion, we would be required to satisfy up to 100 percent of the principal amount of the 2036
Notes solely in cash, with any amounts above the principal amount to be satisfied in shares of our
common stock. If a holder elects to convert its 2036 Notes in connection with a designated event
or change that occurs prior to July 1, 2013, we will pay, to the extent described in the Indenture,
a make whole premium by increasing the conversion rate applicable to such 2036 Notes. Conversion of
our 2036 Notes into common stock could result in dilution to our shareholders. From time to time,
our 2036 Notes hold a fair value below their conversion rate. Any redemption due to the trading
price discount, described in (2) above, would be subject to the restrictions imposed by the Credit
Facility and would occur at the lower of market or conversion value, which would likely be
substantially below the par value of the debt. All of the above conversion rights will be subject
to certain limitations imposed by our Credit Facility.
We have the right to redeem for cash all or a portion of the 2036 Notes on or after July 6, 2011 at
specified redemption prices as provided in the Indenture plus accrued and unpaid interest and
contingent interest. Holders of the 2036 Notes may require us to purchase all or a portion of their
2036 Notes for cash on July 1, 2013; July 1, 2016; July 1, 2021; July 1, 2026; and July 1, 2031 or
in the event of a designated event or change, at a purchase price equal to 100 percent of the
principal amount of the 2036 Notes to be repurchased plus accrued and unpaid interest and
contingent interest.
Convertible Senior Subordinated Notes Due 2041
On September 21, 2009, we exchanged $250.0 million in principal amount of our 2036 Notes for newly
issued 2041 Notes. The 2041 Notes bear a fixed interest rate of 4.0 percent per year, payable
semiannually. The 2041 Notes are our direct, unsecured, senior subordinated obligations, rank
junior to the senior secured Credit Facility and will rank junior in right of payment to all of our
future senior debt as provided in the indenture for the 2041 Notes. The 2041 Notes have the same
rank as our 2036 Notes.
Similar to our 2036 Notes, we separately account for the liability and equity components of our
2036 Notes in a manner that reflects our nonconvertible borrowing rate. The excess of the principal
amount of the liability component over its carrying amount is treated as debt discount and
amortized using the interest method. In addition, debt issuance costs of approximately $7.7
million were allocated to the liability and equity components of the 2041 Notes. Approximately
$5.3 million of the debt issuance costs were allocated to the liability component, recorded in
other long-term assets, and are being amortized using the straight line method over seven years
(representing the time period until the first put date under the 2041 Notes). Approximately $2.4
million of the debt issuance costs were allocated to the equity component and are treated as equity
issuance costs and are not amortized.
The equity component of our 2041 Notes was $76.4 million as of January 2, 2010, and is recorded in
additional paid-in capital. As of January 2, 2010, the principal amount of the liability
component, its unamortized discount, and its net carrying amount were $250.0 million, $76.7 million
and $173.3 million, respectively. The unamortized discount will be amortized over a remaining
period of 6.7 years and the amortization expense is included in “amortization of financing costs”
on the Consolidated Statements of Operations. The effective interest rate on the liability
component was 10.2% for the year ended January 2, 2010. During the year ended January 2, 2010, we
recognized $2.9 million of interest expense representing the contractual interest coupon on our
2041 Notes, and $2.2 million of amortization expense related to the discount on the liability
component.
F-22
Table of Contents
In addition to regular interest on the 2041 Notes, we will also pay contingent interest beginning
September 15, 2016 at 0.75% of the average trading price of the 2041 Notes, if the average trading
price for the five trading days immediately before the first trading day preceding the relevant
six-month period equals or exceeds 130 percent of the principal amount of the 2041 Notes.
Our 2041 Notes are convertible under the following circumstances for cash and shares of our common
stock, if any, at a conversion rate of 51.5318 shares of our common stock per $1,000 principal
amount of 2041 Notes (which is equal to an initial conversion price of approximately $19.406 per
share), subject to adjustment: (1) when, during any fiscal quarter commencing after January 2, 2010
(and only during such fiscal quarter), the last reported sale price of our common stock for at
least 20 trading days (whether or not consecutive) during the period of 30 consecutive trading days
ending on the last trading day of the preceding calendar quarter is greater than or equal to 130%
of the applicable conversion price on the applicable trading day; (2) during the five business day
period after any five consecutive trading day period in which the trading price per $1,000
principal amount of 2041 Notes for each day of that period was less than 98% of the product of the
last reported sale price of our common stock and the applicable conversion rate; (3) if we call the
2041 Notes for redemption; (4) if specified distributions to holders of our common stock occur; (5)
if an event or change occurs that results in conversion according to the Indenture; or (6) during
the 60 days prior to, but excluding, any scheduled repurchase date or maturity date. Upon
conversion, we would be required to satisfy up to 100 percent of the principal amount of the 2041
Notes solely in cash, with any amounts above the principal amount to be satisfied in shares of our
common stock. If a holder elects to convert its 2041 Notes in connection with a designated event
or change, we will pay, to the extent described in the Indenture, a make whole premium by
increasing the conversion rate applicable to such 2041 Notes. Conversion of our 2041 Notes into
common stock could result in dilution to our shareholders. Similar to our 2036 Notes, from time to
time, our 2041 Notes may hold a fair value below their conversion rate. Any redemption due to the
trading price discount, described in (2) above, would be subject to the restrictions imposed by the
Credit Facility and would occur at the lower of market or conversion value, which would likely be
substantially below the par value of the debt. All of the above conversion rights will be subject
to certain limitations imposed by our Credit Facility.
We have the right to redeem for cash all or a portion of the 2041 Notes on or after September 15,
2016 at specified redemption prices as provided in the Indenture plus accrued and unpaid interest
and contingent interest. Holders of the 2041 Notes may require us to purchase all or a portion of
their 2041 Notes for cash on September 15, 2016 or in the event of a designated event or change, at
a purchase price equal to 100 percent of the principal amount of the 2041 Notes to be repurchased
plus accrued and unpaid interest and contingent interest.
Supplemental Guarantor Information
The 2036 Notes and the 2041 Notes are fully and unconditionally guaranteed on an unsecured senior
subordinated basis by four of our significant domestic subsidiaries: American Medical Systems,
Inc., AMS Sales Corporation, AMS Research Corporation and Laserscope (the Guarantor Subsidiaries).
Each of the subsidiary guarantors is 100 percent owned by us. The guarantees are joint and
several, and are subordinated in right of payment to the guaranteed obligations of our significant
domestic subsidiaries under our senior Credit Facility.
The following supplemental condensed consolidating financial information presents the statements of
operations for each of the years ended January 2, 2010, January 3, 2009, and December 29, 2007, the
balance sheets as of January 2, 2010 and January 3, 2009, and the statements of cash flows for each
of the years ended January 2, 2010, January 3, 2009, and December 29, 2007, for the Guarantor
Subsidiaries as a group, and separately for our non-Guarantor Subsidiaries as a group. In the
condensed consolidating financial statements, we and the Guarantor Subsidiaries account for
investment in wholly-owned subsidiaries using the equity method.
F-23
Table of Contents
American Medical Systems Holdings, Inc.
Notes to Consolidated Financial Statements — (Continued)
Condensed Consolidating Statement of Operations
(In thousands)
Notes to Consolidated Financial Statements — (Continued)
Condensed Consolidating Statement of Operations
(In thousands)
| Year Ended Janaury 2, 2010 | ||||||||||||||||||||
| American | ||||||||||||||||||||
| Medical | Non- | |||||||||||||||||||
| Systems | Guarantor | Guarantor | Consolidated | |||||||||||||||||
| Holdings, Inc. | Subsidiaries | Subsidiaries | Eliminations | Total | ||||||||||||||||
Net sales |
$ | — | $ | 473,590 | $ | 113,242 | $ | (67,562 | ) | $ | 519,270 | |||||||||
Cost of sales |
— | 91,205 | 68,237 | (67,231 | ) | 92,211 | ||||||||||||||
Gross profit |
— | 382,385 | 45,005 | (331 | ) | 427,059 | ||||||||||||||
Operating expenses |
||||||||||||||||||||
Marketing and selling |
— | 137,014 | 36,840 | — | 173,854 | |||||||||||||||
Research and development |
— | 52,811 | (46 | ) | — | 52,765 | ||||||||||||||
General and administrative |
— | 45,196 | — | — | 45,196 | |||||||||||||||
Amortization of intangibles |
— | 13,161 | — | — | 13,161 | |||||||||||||||
Total operating expenses |
— | 248,182 | 36,794 | — | 284,976 | |||||||||||||||
Operating income |
— | 134,203 | 8,211 | (331 | ) | 142,083 | ||||||||||||||
Other (expense) income |
||||||||||||||||||||
Royalty income |
— | 3,073 | — | — | 3,073 | |||||||||||||||
Interest income |
— | 281 | 57 | (163 | ) | 175 | ||||||||||||||
Interest expense |
(10,857 | ) | (8,753 | ) | (198 | ) | 172 | (19,636 | ) | |||||||||||
Amortization of financing costs |
(12,873 | ) | (2,917 | ) | — | — | (15,790 | ) | ||||||||||||
Gain on extinguishment of debt |
10,125 | — | — | — | 10,125 | |||||||||||||||
Gain on sale of non-strategic
assets |
— | 17,446 | — | — | 17,446 | |||||||||||||||
Other income (expense) |
— | (1,709 | ) | 269 | (1 | ) | (1,441 | ) | ||||||||||||
Total other (expense) income |
(13,605 | ) | 7,421 | 128 | 8 | (6,048 | ) | |||||||||||||
(Loss) income before income taxes |
(13,605 | ) | 141,624 | 8,339 | (323 | ) | 136,035 | |||||||||||||
Provision for income taxes |
(5,130 | ) | 53,236 | 3,214 | (123 | ) | 51,197 | |||||||||||||
Equity in earnings of subsidiary |
93,513 | 5,125 | — | (98,638 | ) | — | ||||||||||||||
Net income |
$ | 85,038 | $ | 93,513 | $ | 5,125 | $ | (98,838 | ) | $ | 84,838 | |||||||||
F-24
Table of Contents
American Medical Systems Holdings, Inc.
Notes to Consolidated Financial Statements — (Continued)
Condensed Consolidating Statement of Operations
(In thousands)
Notes to Consolidated Financial Statements — (Continued)
Condensed Consolidating Statement of Operations
(In thousands)
| Year Ended Janaury 3, 2009 | ||||||||||||||||||||
| American | ||||||||||||||||||||
| Medical | Non- | |||||||||||||||||||
| Systems | Guarantor | Guarantor | Consolidated | |||||||||||||||||
| Holdings, Inc. | Subsidiaries | Subsidiaries | Eliminations | Total | ||||||||||||||||
Net sales |
$ | — | $ | 451,054 | $ | 113,080 | $ | (62,493 | ) | $ | 501,641 | |||||||||
Cost of sales |
— | 109,873 | 64,637 | (63,413 | ) | 111,097 | ||||||||||||||
Gross profit |
— | 341,181 | 48,443 | 920 | 390,544 | |||||||||||||||
Operating expenses |
||||||||||||||||||||
Marketing and selling |
— | 136,340 | 39,330 | — | 175,670 | |||||||||||||||
Research and development |
— | 46,247 | — | — | 46,247 | |||||||||||||||
In-process research and
development |
— | 7,500 | — | — | 7,500 | |||||||||||||||
General and administrative |
— | 39,280 | 1 | — | 39,281 | |||||||||||||||
Amortization of intangibles |
— | 34,465 | — | — | 34,465 | |||||||||||||||
Total operating expenses |
— | 263,832 | 39,331 | — | 303,163 | |||||||||||||||
Operating income |
— | 77,349 | 9,112 | 920 | 87,381 | |||||||||||||||
Other (expense) income |
||||||||||||||||||||
Royalty income |
— | 2,974 | 1,500 | — | 4,474 | |||||||||||||||
Interest income |
— | 1,509 | 104 | (866 | ) | 747 | ||||||||||||||
Interest expense |
(12,234 | ) | (15,140 | ) | (890 | ) | 866 | (27,398 | ) | |||||||||||
Amortization of financing costs |
(14,804 | ) | (3,678 | ) | — | — | (18,482 | ) | ||||||||||||
Gain on extinguishment of debt |
5,631 | — | — | — | 5,631 | |||||||||||||||
Other income (expense) |
— | (860 | ) | (1,310 | ) | (25 | ) | (2,195 | ) | |||||||||||
Total other (expense) income |
(21,407 | ) | (15,195 | ) | (596 | ) | (25 | ) | (37,223 | ) | ||||||||||
(Loss) income before income taxes |
(21,407 | ) | 62,154 | 8,516 | 895 | 50,158 | ||||||||||||||
Provision for income taxes |
(8,069 | ) | 23,970 | 3,084 | 338 | 19,323 | ||||||||||||||
Equity in earnings of subsidiary |
43,616 | 5,432 | — | (49,048 | ) | — | ||||||||||||||
Net income |
$ | 30,278 | $ | 43,616 | $ | 5,432 | $ | (48,491 | ) | $ | 30,835 | |||||||||
F-25
Table of Contents
American Medical Systems Holdings, Inc.
Notes to Consolidated Financial Statements — (Continued)
Condensed Consolidating Statement of Operations
(In thousands)
Notes to Consolidated Financial Statements — (Continued)
Condensed Consolidating Statement of Operations
(In thousands)
| Year Ended December 29, 2007 | ||||||||||||||||||||
| American | ||||||||||||||||||||
| Medical | Non- | |||||||||||||||||||
| Systems | Guarantor | Guarantor | Consolidated | |||||||||||||||||
| Holdings, Inc. | Subsidiaries | Subsidiaries | Eliminations | Total | ||||||||||||||||
Net sales |
$ | — | $ | 423,879 | $ | 97,555 | $ | (57,506 | ) | $ | 463,928 | |||||||||
Cost of sales |
— | 103,964 | 57,871 | (56,243 | ) | 105,592 | ||||||||||||||
Gross profit |
— | 319,915 | 39,684 | (1,263 | ) | 358,336 | ||||||||||||||
Operating expenses |
||||||||||||||||||||
Marketing and selling |
— | 135,960 | 33,535 | — | 169,495 | |||||||||||||||
Research and development |
— | 43,066 | 249 | — | 43,315 | |||||||||||||||
In-process research and
development |
— | 7,500 | — | — | 7,500 | |||||||||||||||
General and administrative |
— | 42,997 | 73 | — | 43,070 | |||||||||||||||
Integration costs |
— | 1,103 | — | — | 1,103 | |||||||||||||||
Litigation settlement |
— | 14,303 | — | — | 14,303 | |||||||||||||||
Amortization of intangibles |
— | 14,783 | 3,481 | — | 18,264 | |||||||||||||||
Total operating expenses |
— | 259,712 | 37,338 | — | 297,050 | |||||||||||||||
Operating income |
— | 60,203 | 2,346 | (1,263 | ) | 61,286 | ||||||||||||||
Other (expense) income |
||||||||||||||||||||
Royalty income |
— | 5,028 | — | — | 5,028 | |||||||||||||||
Interest income |
— | 1,401 | 57 | (305 | ) | 1,153 | ||||||||||||||
Interest expense |
(11,921 | ) | (25,480 | ) | (659 | ) | 300 | (37,760 | ) | |||||||||||
Amortization of financing costs |
(13,397 | ) | (2,748 | ) | — | — | (16,145 | ) | ||||||||||||
Other income (expense) |
— | 3,233 | (137 | ) | (25 | ) | 3,071 | |||||||||||||
Total other (expense) income |
(25,318 | ) | (18,566 | ) | (739 | ) | (30 | ) | (44,653 | ) | ||||||||||
(Loss)
income from continuing operations before income taxes |
(25,318 | ) | 41,637 | 1,607 | (1,293 | ) | 16,633 | |||||||||||||
Provision for income taxes |
(8,756 | ) | 20,524 | 610 | (490 | ) | 11,888 | |||||||||||||
Net (loss) income from continuing operations |
(16,562 | ) | 21,113 | 997 | (803 | ) | 4,745 | |||||||||||||
Loss from discontinued operations, net of tax |
— | (691 | ) | — | — | (691 | ) | |||||||||||||
Equity in earnings of subsidiary |
21,419 | 997 | — | (22,416 | ) | — | ||||||||||||||
Net income |
$ | 4,857 | $ | 21,419 | $ | 997 | $ | (23,219 | ) | $ | 4,054 | |||||||||
F-26
Table of Contents
American Medical Systems Holdings, Inc.
Notes to Consolidated Financial Statements — (Continued)
Condensed Consolidating Balance Sheet
Notes to Consolidated Financial Statements — (Continued)
Condensed Consolidating Balance Sheet
(In thousands)
| As of January 2, 2010 | ||||||||||||||||||||
| American | ||||||||||||||||||||
| Medical | Non- | |||||||||||||||||||
| Systems | Guarantor | Guarantor | Consolidated | |||||||||||||||||
| Holdings, Inc. | Subsidiaries | Subsidiaries | Eliminations | Total | ||||||||||||||||
Assets |
||||||||||||||||||||
Current assets |
||||||||||||||||||||
Cash and cash equivalents |
$ | — | $ | 16,973 | $ | 13,697 | $ | — | $ | 30,670 | ||||||||||
Short-term investments |
4,834 | 14,489 | 545 | — | 19,868 | |||||||||||||||
Accounts receivable, net |
614,392 | 58,359 | 29,772 | (599,933 | ) | 102,590 | ||||||||||||||
Inventories, net |
— | 27,750 | 6,853 | (4,327 | ) | 30,276 | ||||||||||||||
Deferred income taxes |
— | 13,466 | 1,404 | — | 14,870 | |||||||||||||||
Other current assets |
— | 4,947 | 1,120 | — | 6,067 | |||||||||||||||
Total current assets |
619,226 | 135,984 | 53,391 | (604,260 | ) | 204,341 | ||||||||||||||
Property, plant and equipment, net |
— | 42,661 | 1,459 | — | 44,120 | |||||||||||||||
Goodwill |
— | 628,193 | 86,727 | (24,021 | ) | 690,899 | ||||||||||||||
Developed and core technology, net |
— | 51,631 | — | — | 51,631 | |||||||||||||||
Other intangibles, net |
— | 49,937 | — | — | 49,937 | |||||||||||||||
Investment in subsidiaries |
190,818 | 45,579 | — | (236,397 | ) | — | ||||||||||||||
Other long-term assets, net |
5,133 | 839 | 251 | — | 6,223 | |||||||||||||||
Total assets |
$ | 815,177 | $ | 954,824 | $ | 141,828 | $ | (864,678 | ) | $ | 1,047,151 | |||||||||
Liabilities and Stockholders’ Equity |
||||||||||||||||||||
Current liabilities |
||||||||||||||||||||
Accounts payable |
$ | (5,058 | ) | $ | 558,217 | $ | 72,015 | $ | (616,060 | ) | $ | 9,114 | ||||||||
Accrued compensation expenses |
— | 24,350 | 5,253 | — | 29,603 | |||||||||||||||
Accrued warranty expense |
— | 2,293 | — | — | 2,293 | |||||||||||||||
Income taxes payable |
(13,272 | ) | 16,371 | 1,396 | — | 4,495 | ||||||||||||||
Other accrued expenses |
3,885 | 17,826 | 4,049 | — | 25,760 | |||||||||||||||
Total current liabilities |
(14,445 | ) | 619,057 | 82,713 | (616,060 | ) | 71,265 | |||||||||||||
Non-current liabilities |
||||||||||||||||||||
Long-term debt |
223,876 | 122,353 | — | — | 346,229 | |||||||||||||||
Intercompany loans payable |
— | — | 12,221 | (12,221 | ) | — | ||||||||||||||
Deferred income taxes |
60,387 | 645 | 1,315 | — | 62,347 | |||||||||||||||
Long-term income taxes payable |
— | 18,206 | — | — | 18,206 | |||||||||||||||
Long-term employee benefit
obligations |
— | 3,745 | — | — | 3,745 | |||||||||||||||
Total non-current liabilities |
284,263 | 144,949 | 13,536 | (12,221 | ) | 430,527 | ||||||||||||||
Total liabilities |
269,818 | 764,006 | 96,249 | (628,281 | ) | 501,792 | ||||||||||||||
Stockholders’ equity |
||||||||||||||||||||
Common stock |
747 | — | 9 | (9 | ) | 747 | ||||||||||||||
Additional paid-in capital |
399,468 | 3,424 | 57,540 | (60,964 | ) | 399,468 | ||||||||||||||
Accumulated other
comprehensive income |
6,381 | (202 | ) | 7,137 | (6,935 | ) | 6,381 | |||||||||||||
Retained earnings (deficit) |
138,763 | 187,596 | (19,107 | ) | (168,489 | ) | 138,763 | |||||||||||||
Total stockholders’ equity |
545,359 | 190,818 | 45,579 | (236,397 | ) | 545,359 | ||||||||||||||
Total liabilities and stockholders’ equity |
$ | 815,177 | $ | 954,824 | $ | 141,828 | $ | (864,678 | ) | $ | 1,047,151 | |||||||||
F-27
Table of Contents
American Medical Systems Holdings, Inc.
Notes to Consolidated Financial Statements — (Continued)
Condensed Consolidating Balance Sheet
Notes to Consolidated Financial Statements — (Continued)
Condensed Consolidating Balance Sheet
(In thousands)
| As of January 3, 2009 | ||||||||||||||||||||
| American | ||||||||||||||||||||
| Medical | Non- | |||||||||||||||||||
| Systems | Guarantor | Guarantor | Consolidated | |||||||||||||||||
| Holdings, Inc. | Subsidiaries | Subsidiaries | Eliminations | Total | ||||||||||||||||
Assets |
||||||||||||||||||||
Current assets |
||||||||||||||||||||
Cash and cash equivalents |
$ | — | $ | 3,143 | $ | 8,499 | $ | — | $ | 11,642 | ||||||||||
Short-term investments |
15,756 | 15,433 | 134 | — | 31,323 | |||||||||||||||
Accounts receivable, net |
618,891 | 68,158 | 24,817 | (618,788 | ) | 93,078 | ||||||||||||||
Inventories, net |
— | 36,521 | 5,226 | (3,247 | ) | 38,500 | ||||||||||||||
Deferred income taxes |
— | 12,412 | 496 | — | 12,908 | |||||||||||||||
Other current assets |
— | 5,832 | 1,026 | — | 6,858 | |||||||||||||||
Total current assets |
634,647 | 141,499 | 40,198 | (622,035 | ) | 194,309 | ||||||||||||||
Property, plant and equipment, net |
— | 46,751 | 1,529 | — | 48,280 | |||||||||||||||
Goodwill |
— | 628,193 | 85,925 | (24,021 | ) | 690,097 | ||||||||||||||
Developed and core technology, net |
— | 62,315 | — | — | 62,315 | |||||||||||||||
Other intangibles, net |
— | 47,349 | — | — | 47,349 | |||||||||||||||
Investment in subsidiaries |
110,868 | 32,824 | — | (143,692 | ) | — | ||||||||||||||
Other long-term assets, net |
578 | 1,418 | 151 | — | 2,147 | |||||||||||||||
Total assets |
$ | 746,093 | $ | 960,349 | $ | 127,803 | $ | (789,748 | ) | $ | 1,044,497 | |||||||||
Liabilities and Stockholders’ Equity |
||||||||||||||||||||
Current liabilities |
||||||||||||||||||||
Accounts payable |
$ | 11,461 | $ | 556,362 | $ | 70,944 | $ | (630,937 | ) | $ | 7,830 | |||||||||
Accrued compensation expenses |
— | 19,252 | 3,624 | — | 22,876 | |||||||||||||||
Accrued warranty expense |
— | 3,300 | (13 | ) | — | 3,287 | ||||||||||||||
Income taxes payable |
(980 | ) | 6,657 | 2,105 | — | 7,782 | ||||||||||||||
Other accrued expenses |
91 | 18,504 | 2,940 | — | 21,535 | |||||||||||||||
Total current liabilities |
10,572 | 604,075 | 79,600 | (630,937 | ) | 63,310 | ||||||||||||||
Non-current liabilities |
||||||||||||||||||||
Long-term debt |
262,094 | 222,488 | — | — | 484,582 | |||||||||||||||
Intercompany loans payable |
— | — | 15,119 | (15,119 | ) | — | ||||||||||||||
Deferred income taxes |
45,945 | 3,839 | 260 | — | 50,044 | |||||||||||||||
Long-term income taxes payable |
— | 15,327 | — | — | 15,327 | |||||||||||||||
Long-term employee benefit
obligations |
— | 3,752 | — | — | 3,752 | |||||||||||||||
Total non-current liabilities |
308,039 | 245,406 | 15,379 | (15,119 | ) | 553,705 | ||||||||||||||
Total liabilities |
318,611 | 849,481 | 94,979 | (646,056 | ) | 617,015 | ||||||||||||||
Stockholders’ equity |
||||||||||||||||||||
Common stock |
737 | — | 9 | (9 | ) | 737 | ||||||||||||||
Additional paid-in capital |
369,594 | 3,424 | 67,368 | (70,792 | ) | 369,594 | ||||||||||||||
Accumulated other
comprehensive income |
3,226 | (1,334 | ) | 4,367 | (3,033 | ) | 3,226 | |||||||||||||
Retained earnings (deficit) |
53,925 | 108,778 | (38,920 | ) | (69,858 | ) | 53,925 | |||||||||||||
Total stockholders’ equity |
427,482 | 110,868 | 32,824 | (143,692 | ) | 427,482 | ||||||||||||||
Total liabilities and stockholders’ equity |
$ | 746,093 | $ | 960,349 | $ | 127,803 | $ | (789,748 | ) | $ | 1,044,497 | |||||||||
F-28
Table of Contents
American Medical Systems Holdings, Inc.
Notes to Consolidated Financial Statements — (Continued)
Condensed Consolidating Statement of Cash Flows
(In thousands)
Notes to Consolidated Financial Statements — (Continued)
Condensed Consolidating Statement of Cash Flows
(In thousands)
| Year Ended January 2, 2010 | ||||||||||||||||||||||
| American | ||||||||||||||||||||||
| Medical | Non- | |||||||||||||||||||||
| Systems | Guarantor | Guarantor | Consolidated | |||||||||||||||||||
| Holdings, Inc. | Subsidiaries | Subsidiaries | Eliminations | Total | ||||||||||||||||||
Cash flows from operating activities |
||||||||||||||||||||||
Net cash provided by operating activities |
$ | 18,066 | $ | 98,005 | $ | 6,725 | $ | — | $ | 122,796 | ||||||||||||
Cash flows from investing activities |
||||||||||||||||||||||
Purchase of property, plant and equipment |
— | (5,493 | ) | (372 | ) | — | (5,865 | ) | ||||||||||||||
Sale of non-strategic assets, net |
— | 18,982 | — | — | 18,982 | |||||||||||||||||
Purchase of other intangibles |
— | (5,927 | ) | — | — | (5,927 | ) | |||||||||||||||
Purchase of short term investments |
— | (18,158 | ) | (662 | ) | — | (18,820 | ) | ||||||||||||||
Sale of short-term investments |
— | 30,500 | 255 | — | 30,755 | |||||||||||||||||
Net payments from settlement of derivative contracts |
— | (556 | ) | — | — | (556 | ) | |||||||||||||||
Net cash provided by (used in) investing activities |
— | 19,348 | (779 | ) | — | 18,569 | ||||||||||||||||
Cash flows from financing activities |
||||||||||||||||||||||
Intercompany notes |
— | (14 | ) | 14 | — | — | ||||||||||||||||
Issuance of common stock |
10,190 | — | — | — | 10,190 | |||||||||||||||||
Excess tax benefit from exercise of stock options |
566 | — | — | — | 566 | |||||||||||||||||
Debt issuance costs |
(7,697 | ) | — | — | — | (7,697 | ) | |||||||||||||||
Payments on senior secured credit facility |
— | (103,510 | ) | — | — | (103,510 | ) | |||||||||||||||
Repurchase of convertible senior subordinated
notes |
(21,125 | ) | — | — | — | (21,125 | ) | |||||||||||||||
Net cash used in financing activities |
(18,066 | ) | (103,524 | ) | 14 | — | (121,576 | ) | ||||||||||||||
Effect of exchange rates on cash |
— | — | (761 | ) | — | (761 | ) | |||||||||||||||
Net increase in cash and cash equivalents |
— | 13,829 | 5,199 | — | 19,028 | |||||||||||||||||
Cash and cash equivalents at beginning of period |
— | 3,143 | 8,499 | — | 11,642 | |||||||||||||||||
Cash and cash equivalents at end of period |
$ | — | $ | 16,972 | $ | 13,698 | $ | — | $ | 30,670 | ||||||||||||
F-29
Table of Contents
American Medical Systems Holdings, Inc.
Notes to Consolidated Financial Statements — (Continued)
Condensed Consolidating Statement of Cash Flows
(In thousands)
Notes to Consolidated Financial Statements — (Continued)
Condensed Consolidating Statement of Cash Flows
(In thousands)
| ` | ||||||||||||||||||||
| Year Ended January 3, 2009 | ||||||||||||||||||||
| American | ||||||||||||||||||||
| Medical | Non- | |||||||||||||||||||
| Systems | Guarantor | Guarantor | Consolidated | |||||||||||||||||
| Holdings, Inc. | Subsidiaries | Subsidiaries | Eliminations | Total | ||||||||||||||||
Cash flows from operating activities |
||||||||||||||||||||
Net cash provided by (used in) operating
activities |
$ | 12,969 | $ | 107,771 | $ | (4,983 | ) | $ | — | $ | 115,757 | |||||||||
Cash flows from investing activities |
||||||||||||||||||||
Purchase of property, plant and equipment |
— | (5,468 | ) | (633 | ) | — | (6,101 | ) | ||||||||||||
Disposal of business |
— | 4,691 | — | — | 4,691 | |||||||||||||||
Purchase of investments in technology |
— | (7,500 | ) | — | — | (7,500 | ) | |||||||||||||
Purchase of other intangibles |
— | (1,352 | ) | — | — | (1,352 | ) | |||||||||||||
Purchase of short-term investments |
— | (70,440 | ) | (65 | ) | — | (70,505 | ) | ||||||||||||
Sale of short-term investments |
— | 39,731 | 268 | — | 39,999 | |||||||||||||||
Net payments from settlement of derivative
contracts |
— | (1,385 | ) | — | — | (1,385 | ) | |||||||||||||
Net cash used in investing activities |
— | (41,723 | ) | (430 | ) | — | (42,153 | ) | ||||||||||||
Cash flows from financing activities |
||||||||||||||||||||
Intercompany notes |
— | 626 | (626 | ) | — | — | ||||||||||||||
Issuance of common stock |
8,874 | — | — | — | 8,874 | |||||||||||||||
Excess tax benefit from exercise of stock
options |
1,498 | — | — | — | 1,498 | |||||||||||||||
Proceeds from short-term borrowings |
— | 12,000 | — | — | 12,000 | |||||||||||||||
Repayments of short-term borrowings |
— | (12,000 | ) | — | — | (12,000 | ) | |||||||||||||
Payments on senior secured credit facility |
— | (85,202 | ) | — | — | (85,202 | ) | |||||||||||||
Repurchase of convertible senior subordinated
notes |
(23,373 | ) | — | — | — | (23,373 | ) | |||||||||||||
Net cash used in financing activities |
(13,001 | ) | (84,576 | ) | (626 | ) | — | (98,203 | ) | |||||||||||
Effect of exchange rates on cash |
— | — | 2,197 | — | 2,197 | |||||||||||||||
Net decrease in cash and cash equivalents |
(32 | ) | (18,528 | ) | (3,842 | ) | — | (22,402 | ) | |||||||||||
Cash and cash equivalents at beginning of period |
32 | 21,671 | 12,341 | — | 34,044 | |||||||||||||||
Cash and cash equivalents at end of period |
$ | — | $ | 3,143 | $ | 8,499 | $ | — | $ | 11,642 | ||||||||||
F-30
Table of Contents
American Medical Systems Holdings, Inc.
Notes to Consolidated Financial Statements — (Continued)
Condensed Consolidating Statement of Cash Flows
(In thousands)
Notes to Consolidated Financial Statements — (Continued)
Condensed Consolidating Statement of Cash Flows
(In thousands)
| Year Ended December 29, 2007 | ||||||||||||||||||||
| American | ||||||||||||||||||||
| Medical | Non- | |||||||||||||||||||
| Systems | Guarantor | Guarantor | Consolidated | |||||||||||||||||
| Holdings, Inc. | Subsidiaries | Subsidiaries | Eliminations | Total | ||||||||||||||||
Cash flows from operating activities |
||||||||||||||||||||
Net cash (used in) provided by operating activities |
$ | (12,151 | ) | $ | 55,039 | $ | 4,863 | $ | — | $ | 47,751 | |||||||||
Cash flows from investing activities |
||||||||||||||||||||
Purchase of property, plant and equipment |
— | (12,983 | ) | (1,190 | ) | — | (14,173 | ) | ||||||||||||
Purchase of business, net of cash acquired |
— | — | (781 | ) | — | (781 | ) | |||||||||||||
Disposal of business |
— | 22,116 | — | 22,116 | ||||||||||||||||
Purchase of investments in technology |
— | (7,500 | ) | — | — | (7,500 | ) | |||||||||||||
Purchase of other intangibles |
— | (382 | ) | — | — | (382 | ) | |||||||||||||
Purchase of short-term investments |
— | (30,079 | ) | (108 | ) | — | (30,187 | ) | ||||||||||||
Sale of short-term investments |
— | 29,560 | 10 | — | 29,570 | |||||||||||||||
Net cash provided by (used in) investing activities |
— | 732 | (2,069 | ) | — | (1,337 | ) | |||||||||||||
Cash flows from financing activities |
||||||||||||||||||||
Intercompany notes |
— | 2,089 | (2,089 | ) | — | — | ||||||||||||||
Dividend from parent |
— | (8,334 | ) | 8,334 | — | |||||||||||||||
Issuance of common stock |
10,830 | — | — | — | 10,830 | |||||||||||||||
Excess tax benefit from exercise of stock options |
215 | — | — | — | 215 | |||||||||||||||
Payments on long-term debt |
— | (50,069 | ) | — | — | (50,069 | ) | |||||||||||||
Net cash provided by (used in) financing activities |
11,045 | (56,314 | ) | 6,245 | — | (39,024 | ) | |||||||||||||
Cash used in discontinued operations |
||||||||||||||||||||
Operating activities |
— | (691 | ) | — | — | (691 | ) | |||||||||||||
Net cash used in discontinued operations |
— | (691 | ) | — | — | (691 | ) | |||||||||||||
Effect of exchange rates on cash |
— | — | (1,706 | ) | — | (1,706 | ) | |||||||||||||
Net (decrease) increase in cash and cash equivalents |
(1,106 | ) | (1,234 | ) | 7,333 | — | 4,993 | |||||||||||||
Cash and cash equivalents at beginning of period |
1,138 | 22,905 | 5,008 | — | 29,051 | |||||||||||||||
Cash and cash equivalents at end of period |
$ | 32 | $ | 21,671 | $ | 12,341 | $ | — | $ | 34,044 | ||||||||||
F-31
Table of Contents
8. Stock-Based Compensation
At January 2, 2010 we have one active stock-based employee compensation plan under which new awards
may be granted.
The following table presents a summary of the share-based compensation expense recognized for these
plans:
| Year Ended | Year Ended | Year Ended | ||||||||||
| (in thousands) | January 2, 2010 | January 3, 2009 | December 29, 2007 | |||||||||
Stock-option awards |
$ | 7,074 | $ | 7,721 | $ | 9,574 | ||||||
Restricted stock awards |
1,073 | 602 | 2,228 | |||||||||
Employee stock purchase plan |
841 | 619 | 596 | |||||||||
Total share-based compensation expense |
$ | 8,988 | $ | 8,942 | $ | 12,398 | ||||||
The following table presents the statement of operations classification of pre-tax stock-based
compensation expense, for stock options, restricted stock awards and the employee stock purchase
plan, recognized for the years ended January 2, 2010, January 3, 2009 and December 29, 2007:
| Year Ended | Year Ended | Year Ended | ||||||||||
| (in thousands) | January 2, 2010 | January 3, 2009 | December 29, 2007 | |||||||||
Cost of sales |
$ | 976 | $ | 2,069 | $ | 1,584 | ||||||
Marketing and selling |
1,981 | 3,454 | 4,187 | |||||||||
Research and development |
1,208 | 2,254 | 2,766 | |||||||||
General and administrative |
4,823 | 1,165 | 3,861 | |||||||||
Total share-based compensation expense |
$ | 8,988 | $ | 8,942 | $ | 12,398 | ||||||
The total income tax benefit recognized in our statement of operations for share-based
compensation arrangements was $3.0 million, $2.6 million and $3.6 million for the years ended
January 2, 2010, January 3, 2009 and December 29,2007, respectively.
The cash flows resulting from tax deductions in excess of the compensation cost recognized for
those options (excess tax benefits) are classified as financing cash flows. Excess tax benefits of
$0.6 million, $1.5 million and $0.2 million were classified as financing cash inflows for the
twelve-month periods ended January 2, 2010, January 3, 2009
and December 29, 2007, respectively.
Our 2005 Stock Incentive Plan (2005 Plan), which replaced our 2000 Equity Incentive Plan (2000
Plan), was amended in May 2009 to increase the number of shares permitted for the grant of equity
incentive awards to our employees, consultants and directors from
6,600,000 to 11,600,000 shares of common stock, plus the number of shares under our 2000 Plan as of
May 5, 2005 subject to outstanding option adjustments, for total grants available of 27,140,383.
We have granted options and restricted stock awards to purchase shares for an aggregate of
20,469,076 shares (net of cancellations) under both plans and 6,671,307 shares remain available for
future grants under our 2005 Plan.
Options granted under the plans generally become exercisable for twenty-five percent of the shares
on the first anniversary date of the grant and 6.25 percent at the end of each quarter thereafter.
Options are granted with an exercise price equal to the fair market value of the common stock on
the date of the grant.
Options granted under our 2000 Plan generally have a stated expiration, if not exercised or earlier
terminated, ten years after the date of grant. Options granted under our 2005 Plan generally have a
stated expiration, if not exercised or earlier terminated, seven years after the date of grant.
F-32
Table of Contents
Activity under our 2000 Plan and 2005 Plan for the twelve months ended January 2, 2010, January 3,
2009 and December 29, 2007 was as follows:
| Weighted average | Aggregate | |||||||||||
| Options | exercise price | Intrinsic | ||||||||||
| outstanding | per share | Value | ||||||||||
| (in thousands) | ||||||||||||
Balance at December 30, 2006 |
7,255,574 | $ | 14.25 | |||||||||
Granted |
1,401,600 | $ | 18.50 | |||||||||
Exercised |
(603,698 | ) | $ | 13.80 | ||||||||
Cancelled or expired |
(465,584 | ) | $ | 18.44 | ||||||||
Balance at December 29, 2007 |
7,587,892 | $ | 14.82 | |||||||||
Granted |
1,453,040 | $ | 14.41 | |||||||||
Exercised |
(1,016,891 | ) | $ | 5.36 | ||||||||
Cancelled or expired |
(1,003,700 | ) | $ | 19.06 | ||||||||
Balance at January 3, 2009 |
7,020,341 | $ | 15.50 | |||||||||
Granted |
1,713,874 | $ | 12.11 | |||||||||
Exercised |
(715,864 | ) | $ | 10.32 | ||||||||
Cancelled or expired |
(856,806 | ) | $ | 16.53 | ||||||||
Balance at January 2, 2010 |
7,161,545 | $ | 15.08 | $ | 31,676 | |||||||
Options exercisable at January 2, 2010 |
4,390,413 | $ | 15.94 | $ | 16,098 | |||||||
Exercise prices and weighted average remaining contractual life for options outstanding as of
January 2, 2010, excluding estimated forfeitures, are summarized as follows:
| Options Outstanding | Options Exercisable | |||||||||||||||||||
| Weighted | Weighted | Weighted | ||||||||||||||||||
| average | average | average | ||||||||||||||||||
| Number | remaining | exercise | Number | exercise | ||||||||||||||||
| Range of exercise prices | of shares | contractual life | price | of shares | price | |||||||||||||||
$0.83 — $11.17 |
1,895,785 | 4.1 years | $ | 9.56 | 984,310 | $ | 8.62 | |||||||||||||
$11.35 — $14.92 |
1,799,439 | 5.2 years | 14.04 | 669,420 | 14.27 | |||||||||||||||
$14.94 — $18.75 |
1,857,364 | 4.1 years | 17.28 | 1,293,230 | 17.60 | |||||||||||||||
$18.87 — $21.68 |
1,608,957 | 3.4 years | 20.22 | 1,443,453 | 20.24 | |||||||||||||||
Total |
7,161,545 | 4.2 years | $ | 15.08 | 4,390,413 | $ | 15.94 | |||||||||||||
The total intrinsic value of options exercised during the twelve months ended January 2, 2010
was $4.5 million. The total intrinsic value at January 2, 2010 is based on our closing stock price
on the last trading day of the year for in-the-money options. The weighted-average remaining
contractual term of options exercisable at January 2, 2010 was 3.3 years.
The fair value of each option award is estimated on the date of grant using the Black-Scholes
valuation model, incorporating key assumptions on volatility and expected option lives based on our
analysis of historical indicators. Forfeitures are estimated based on historical indicators. We
adopted the straight-line method of expense attribution that results in a straight-line
amortization of the compensation expense over the vesting period for all options.
F-33
Table of Contents
The following table provides the weighted average fair value of options granted to employees and
the related assumptions used in the Black-Scholes model:
| 2009 | 2008 | 2007 | ||||||||||
Fair value of options granted |
$ | 4.18 | $ | 5.00 | $ | 6.50 | ||||||
Risk free interest rate |
1.94 | % | 2.88 | % | 4.44 | % | ||||||
Expected dividend rate |
0.00 | % | 0.00 | % | 0.00 | % | ||||||
Stock price volatility |
36.61 | % | 34.81 | % | 32.88 | % | ||||||
Expected life of option |
5 years | 5 years | 5 years | |||||||||
Expected life: We analyze historical employee exercise and termination data to estimate the
expected life assumption. We believe that historical data currently represents the best estimate
of the expected life of a new employee option. For determining the fair value of options, we use
different expected lives for the general employee population in the United States, employees in
international offices and for officers and directors. We examined the historical pattern of option
exercises to determine if there was a discernable pattern as to how different classes of employees
exercised their options. Our analysis showed that officers and directors hold their stock options
for a longer period of time before exercising compared to the rest of the employee population and
that United States employees hold their stock options for a longer period of time before exercising
as compared to international employees.
Expected volatility: We estimate the volatility of our common stock by using the historical
volatility over the expected life of the applicable option. We made the decision to use historical
volatility due to the limited availability of actively traded options for our common stock from
which to derive implied volatility.
Risk-free rate of return: The rate is based on the U.S. Treasury zero-coupon yield curve on the
grant date for a term similar to the expected life of the options.
Dividend yield: We have not paid dividends in the past and do not anticipate paying any cash
dividends in the foreseeable future, therefore a dividend yield of zero is assumed.
As of January 2, 2010, we had $13.6 million of total unrecognized compensation cost, net of
estimated forfeitures, related to unvested share-based compensation arrangements granted under our
2005 Plan. We expect that cost to be recognized over a weighted average period of 2.6 years.
During the year ended January 2, 2010, stock options were exercised to acquire 715,864 shares.
Cash received upon exercise was $7.4 million. The tax benefit realized upon exercise was $1.6
million.
Restricted Stock
Restricted stock awards are granted to employees under the 2005 Stock Incentive Plan upon hire or
based on performance criteria established by management. Restricted stock awards are independent
of stock option awards and are subject to forfeiture if employment terminates prior to the release
of the restrictions. We grant restricted stock which generally vests over a four year period.
During the vesting period, ownership of the shares cannot be transferred. Restricted stock is
considered issued and outstanding at the grant date and has the same dividend and voting rights as
other common stock. We recognize compensation expense for the fair value of the restricted stock
grants issued based on the closing stock price on the date of grant. The plan does not designate
the specific number of shares available for restricted stock grants, as these are issued from the
full pool of shares available under the 2005 Stock Incentive Plan. The option pool is reduced by
two shares for each restricted share granted.
The following table summarizes restricted stock activity during the twelve months ended January 2,
2010, January 3, 2009 and December 29, 2007:
F-34
Table of Contents
| Unvested Shares | Weighted average | |||||||
| outstanding | grant date fair value | |||||||
Balance at December 30, 2006 |
120,526 | $ | 18.49 | |||||
Granted |
197,322 | 18.39 | ||||||
Vested |
(60,125 | ) | 18.72 | |||||
Cancelled |
(41,003 | ) | 18.67 | |||||
Balance at December 29, 2007 |
216,720 | 18.30 | ||||||
Granted |
94,800 | 14.70 | ||||||
Vested |
(86,601 | ) | 18.50 | |||||
Cancelled |
(32,351 | ) | 17.67 | |||||
Balance at January 3, 2009 |
192,568 | 16.54 | ||||||
Granted |
127,150 | 15.16 | ||||||
Vested |
(60,579 | ) | 16.99 | |||||
Cancelled |
(25,614 | ) | 15.44 | |||||
Balance at January 2, 2010 |
233,525 | $ | 15.79 | |||||
Employee Stock Purchase Plan
We have an Employee Stock Purchase Plan (ESPP) which allows employees to elect, in advance of each
calendar quarter, to contribute up to 10 percent of their compensation, subject to certain
limitations, to purchase shares of common stock at the lower of 85 percent of the fair market value
on the first or last day of each quarter. Compensation expense recognized on shares issued under
our ESPP is based on the value of an option to purchase shares of our stock at a 15 percent
discount to the stock price. Shares purchased under the employee stock purchase plan were 270,980
during the year. The plan was amended in May 2008 to increase the number of shares reserved under
the plan from 1,000,000 to 2,000,000 common shares and to delete the term and termination date of
the plan. Shares issued under the plan through January 2, 2010 total 1,348,698, with a balance
available to be issued of 651,302.
9. Commitments and Contingent Liabilities
Product Liability
We are self-insured for product liability claims below $1.0 million for each occurrence and $3.0
million in the aggregate, and maintain product liability insurance above these limitations.
We are involved in a number of claims and lawsuits considered normal in our business, including
product liability matters. While it is not possible to predict the outcome of legal actions, we
believe that any liability resulting from the pending claims and suits that would potentially
exceed existing accruals would not have a material, adverse effect on our financial position or on
our results of operations or cash flows for any period.
F-35
Table of Contents
Operating Leases
Future minimum operating lease obligations for automobiles, office space, and other facilities were
as follows at January 2, 2010:
| (in thousands) | ||||
2010 |
$ | 3,438 | ||
2011 |
2,634 | |||
2012 |
1,923 | |||
2013 |
625 | |||
2014 |
14 | |||
2015 and beyond |
— | |||
Total |
$ | 8,634 | ||
Rent expense was $3.5 million, $3.3 million and $2.7 million in 2009, 2008 and 2007,
respectively. The automobiles, which are typically leased for three years, are used by sales
personnel. The office obligations include the Laserscope facilities in California and Arizona, and
sales offices outside the U.S.
10. Fair Value Measurements
Generally accepted accounting principles define and establish a framework for measuring fair value
and providing disclosure about fair value measurements. Furthermore, U.S. GAAP specifies a
hierarchy of valuation techniques based upon whether the inputs to those valuation techniques
reflect assumptions other market participants would use based upon market data obtained from
independent sources (observable inputs) or reflect our own assumptions of market participant
valuation (unobservable inputs). We have categorized our financial assets and liabilities, based
on the priority of the inputs to the valuation technique, into a three-level fair value hierarchy.
If the inputs used to measure the financial instruments fall within different levels of the
hierarchy, the categorization is based on the lowest level input that is significant to the fair
value measurement of the instrument.
Assets and Liabilities Measured at Fair Value on a Recurring Basis
The following table summarizes our financial assets measured at fair value on a recurring basis as
of January 2, 2010 (in thousands):
| Fair Value Measurements as of January 2, 2010 Using | ||||||||||||
| Quoted Prices in | Significant Other | Significant | ||||||||||
| Active Markets for | Observable | Unobservable | ||||||||||
| Description | Identical Assets (Level 1) | Inputs (Level 2) | Inputs (Level 3) | |||||||||
Assets |
||||||||||||
Money market funds |
$ | 18,504 | $ | — | $ | — | ||||||
Available-for-sale securities |
655 | — | — | |||||||||
Other short-term investments |
163 | 546 | — | |||||||||
Derivatives |
— | 10 | — | |||||||||
Total |
$ | 19,322 | $ | 556 | $ | — | ||||||
Liabilities |
||||||||||||
Derivatives |
$ | — | $ | 1,949 | $ | — | ||||||
Money market funds: Our money market funds are highly liquid investments with a maturity of
three months or less. These assets are classified within Level 1 of the fair value hierarchy
because the money market funds are valued using quoted market prices in active markets.
Available-for-sale securities: As of January 2, 2010, our available-for-sale securities included
unregistered common stock of Iridex Corporation. These are valued using quoted market prices
multiplied by the number of shares owned.
Other short-term investments: Other short-term investments consist of mutual fund shares and
short-term bonds. Investments for which quoted market prices are available are categorized as
Level 1 in the fair value hierarchy. For
F-36
Table of Contents
the remaining investments, which have maturities of three months or less, the carrying amount
is a reasonable estimate of fair value and these have been classified as Level 2.
Derivatives: The total fair value of various interest rate swap contracts as of January 2, 2010 is
a liability of $0.3 million, reported in other accrued expenses. The fair value of various foreign
exchange forward contracts as of January 2, 2010 includes liabilities of $1.7 million, reported in other accrued expenses. We measure
our derivatives at fair value on a recurring basis using significant observable inputs, which is
Level 2 as defined in the fair value hierarchy. Refer to Note 11, Derivative Instruments and
Hedging Activities, for more information regarding our derivatives.
Assets and Liabilities Measured at Fair Value on a Nonrecurring Basis
Fair value measurements of nonfinancial assets and liabilities are primarily used in the impairment
analysis of goodwill and other intangible assets. We review goodwill and other intangible assets
for impairment annually, during the fourth quarter of each fiscal year, or as circumstances
indicate the possibility of impairment. During the year ended January 2, 2010, we had no
significant measurements of assets or liabilities at fair value on a nonrecurring basis subsequent
to their initial recognition.
Fair Value of Debt
The fair value of the Convertible Notes (see Note 7, Debt) was estimated using quoted market
prices. The fair value of the Credit Facility was estimated using a discounted cash flow analysis
based on our current estimated incremental borrowing rate for a similar borrowing arrangement.
The following table summarizes the principal outstanding and estimated fair values of our long-term
debt, including current maturities (in thousands):
| January 2, 2010 | January 3, 2009 | |||||||||||||||
| Carrying Value | Fair Value | Carrying Value | Fair Value | |||||||||||||
2036 Notes |
$ | 61,985 | $ | 70,270 | $ | 339,250 | $ | 229,944 | ||||||||
2041 Notes |
250,000 | 304,983 | — | — | ||||||||||||
Credit Facility |
125,307 | 123,230 | 228,817 | 193,003 | ||||||||||||
| $ | 437,292 | $ | 498,483 | $ | 568,067 | $ | 422,947 | |||||||||
11. Derivative Instruments and Hedging Activities
We are exposed to certain risks relating to our ongoing business operations. We use derivatives to
mitigate a portion of our exposure to volatility in interest and foreign currency exchange rates.
Interest rate swaps are used to manage interest rate risk associated with our floating rate debt.
Foreign exchange forward contracts are used to manage the currency risk associated with forecasted
sales to and receivables from certain subsidiaries, denominated in their local currencies. We
hedge only exposures in the ordinary course of business.
We account for our derivative instruments at fair value provided we meet certain documentary and
analytical requirements to qualify for hedge accounting treatment. Hedge accounting creates the
potential for a Consolidated Statement of Operations match between the changes in fair values of
derivatives and the changes in cost of the associated underlying transactions, in this case
interest expense and translation gain or loss. Derivatives held by us are designated as hedges of
specific exposures at inception, with an expectation that changes in the fair value will
essentially offset the change in the underlying exposure. Discontinuance of hedge accounting is
required whenever it is subsequently determined that an underlying transaction is not going to
occur, with any gains or losses recognized in the Consolidated Statement of Operations at such
time, with any subsequent changes in fair value recognized currently in earnings. Fair values of
derivatives are determined based on quoted prices for similar contracts.
The interest rate swap contracts outstanding at January 2, 2010 are designated as cash flow hedges
of the floating rate interest payments for a portion of our borrowings under the Credit Facility.
The portion of borrowings subject to these swap contracts declines over time, ranging from $70.0
million to $30.0 million. These contracts have remaining terms between one and six months. In
addition, we have foreign currency exchange forward contract
derivatives outstanding at January 2, 2010 which are designated as cash flow hedges of currency
fluctuations for a portion of our forecasted sales to certain subsidiaries, denominated in Euros,
British pounds, Canadian dollars and Australian dollars. These contracts have remaining terms
between one and eleven months. The notional amount of
F-37
Table of Contents
the foreign exchange forward contracts
designated as cash flow hedges was $48.4 million and $33.5 million at January 2, 2010 and January
3, 2009, respectively. We have also entered into foreign exchange forward contracts to manage a
portion of our exposure to foreign exchange rate fluctuations on certain inter-company receivables
denominated in Euros, British pounds, Canadian dollars and Australian dollars. These contracts are
not designated as an accounting hedge, and the notional amount of these contracts at January 2,
2010 and January 3, 2009 was $10.9 million and $13.5 million, respectively. The associated
underlying transactions are expected to occur within the next month.
The effective portion of the change in fair value of the
interest rate swaps and foreign currency
exchange contracts is reported in accumulated other comprehensive income, a component of
stockholders’ equity, and is being recognized as an adjustment to interest expense or other income
(expense), respectively, over the same period the related expenses are recognized in earnings.
Ineffectiveness would occur when changes in the market value of the hedged transactions are not
completely offset by changes in the market value of the derivatives. Gains and losses on
derivatives representing hedge ineffectiveness or hedge components excluded from the assessment of
effectiveness are recognized currently in earnings when incurred. No ineffectiveness was
recognized during 2009 or 2008. Amounts due from counterparties (unrealized hedge gains) or owed
to counterparties (unrealized hedge losses) are included in accounts receivable, net or other
accrued expenses, respectively. Cash receipts or payments related to our derivatives are generally
classified in the Consolidated Statements of Cash Flow as cash flows from operating activities,
consistent with the related items being hedged, unless the derivative is not designated as a hedge
or if hedge accounting is discontinued, in which case the receipts or payments are classified as
cash flows from investing activities.
During the third quarter of 2008, due to market changes in the
yield curve we replaced our existing
fixed interest rate swap contracts, which were based on three month LIBOR, with new swap contracts
based on one month LIBOR, to hedge the floating rate interest payments for a portion of our
borrowings under the Credit Facility. The net gain on the terminated swaps of $0.4 million was
immediately recognized in other income (expense), since the original forecasted transactions based
on the previous index were no longer probable of occurring.
Information on the location and amounts of derivative fair values in the Consolidated Balance
Sheets is presented in the table below.
| Fair Values of Derivative instruments | ||||||||||||||||||||||||||||||||
| Asset Derivatives | Liability Derivatives | |||||||||||||||||||||||||||||||
| January 2, 2010 | January 3, 2009 | January 2, 2010 | January 3, 2009 | |||||||||||||||||||||||||||||
| Balance | Balance | Balance | Balance | |||||||||||||||||||||||||||||
| Sheet | Fair | Sheet | Fair | Sheet | Fair | Sheet | Fair | |||||||||||||||||||||||||
| (in thousands) | Location | Value | Location | Value | Location | Value | Location | Value | ||||||||||||||||||||||||
Derivatives designated as hedging instruments |
||||||||||||||||||||||||||||||||
Interest rate swap contracts |
Other current assets | $ | — | Other current assets | $ | — | Other accrued expenses | $ | 299 | Other accrued expenses | $ | 2,663 | ||||||||||||||||||||
Foreign exchange forward
contracts |
Other current assets | — | Other current assets | 659 | Other accrued expenses | 1,650 | Other accrued expenses | 1,729 | ||||||||||||||||||||||||
Total derivatives designated as
hedging instruments |
$ | — | $ | 659 | $ | 1,949 | $ | 4,392 | ||||||||||||||||||||||||
|
Derivatives not designated as hedging instruments |
||||||||||||||||||||||||||||||||
Foreign exchange forward
contracts |
Other current assets | $ | 10 | Other current assets | $ | 223 | Other accrued expenses | $ | — | Other accrued expenses | $ | — | ||||||||||||||||||||
Total derivatives not designated as
hedging instruments |
$ | 10 | $ | 223 | $ | — | $ | — | ||||||||||||||||||||||||
Total derivatives |
$ | 10 | $ | 882 | $ | 1,949 | $ | 4,392 | ||||||||||||||||||||||||
At January 2, 2010, approximately $0.3 million of the existing loss on the interest rate swap
contracts designated as a cash flow hedge, and approximately $1.7 million of the existing loss on
the foreign exchange forward contracts designated as a cash flow hedge, both of which are included
in accumulated other comprehensive income, are
F-38
Table of Contents
expected to be reclassified into earnings within the
next twelve months.
We are exposed to credit losses in the event of non-performance by counterparties on these
financial instruments, and although no assurances can be given, we do not expect any of the
counterparties to fail to meet its obligations. The credit exposure related to these financial
instruments is represented by the fair value of contracts with a positive fair value at the
reporting date. To manage credit risks, we enter into derivative instruments with high quality
financial institutions, which we monitor regularly and take action where possible to mitigate risk.
Information on the location and amounts of derivative gains and losses recorded in other
comprehensive income (OCI) and recorded in the Consolidated Statements of Operations is presented
in the table below.
The Effect of Derivative Instruments on the Consolidated Statement of Operations
for the Years Ended January 2, 2010 and January 3, 2009
(in thousands)
for the Years Ended January 2, 2010 and January 3, 2009
(in thousands)
| Location of Gain | ||||||||||||||||||||
| Derivatives in | Amount of Gain (Loss) | (Loss) Reclassified | Amount of Gain (Loss) Reclassified | |||||||||||||||||
| Cash Flow | Recognized in OCI on Derivatives | from Accumulated | from Accumulated OCI into Income | |||||||||||||||||
| Hedging | (Effective Portion) | OCI into Income | (Effective Portion) | |||||||||||||||||
| Relationships | January 2, 2010 | January 3, 2009 | (Effective Portion) | January 2, 2010 | January 3, 2009 | |||||||||||||||
Interest rate swap
contracts |
$ | 2,372 | $ | (2,662 | ) | Interest expense | $ | (3,341 | ) | $ | (163 | ) | ||||||||
Interest rate swap
contracts |
— | — | Other (expense) income | — | 381 | (a) | ||||||||||||||
Foreign exchange
contracts |
(1,259 | ) | (519 | ) | Other (expense) income | (2,680 | ) | 832 | ||||||||||||
Total |
$ | 1,113 | $ | (3,181 | ) | $ | (6,021 | ) | $ | 1,050 | ||||||||||
| (a) | This net gain on terminated interest rate swap contracts was immediately recognized in other (expense) income, as the original forecasted | |
| transactions were no longer probable of occurring. |
| Derivatives not | Location of Gain (Loss) | Amount of Gain (Loss) Recognized | ||||||||||
| Designated as | Recognize in Income | in Income on Derivatives | ||||||||||
| Hedging Instruments | on Derivatives | January 2, 2010 | January 3, 2009 | |||||||||
Foreign exchange contracts |
Other (expense) income | $ | (410 | ) | $ | (1,162 | ) | |||||
Total |
$ | (410 | ) | $ | (1,162 | ) | ||||||
12. Industry Segment Information and Foreign Operations
Since our inception, we have operated in the single industry segment of developing, manufacturing,
selling and marketing medical devices. In the first quarter of 2007, consistent with the plans
announced with the Laserscope acquisition, we sold the Laserscope aesthetics business (see Note 2,
Discontinued Operations and Sale of Aesthetics
Business). We have presented the operations of this business as discontinued operations from the
date of acquisition of July 20, 2006 through the date of disposal of January 16, 2007. As such,
the following data excludes the results of the aesthetics business for all periods presented.
We distribute products through our direct sales force and independent sales representatives in the
United States, Canada, Australia, Brazil and Western Europe. Additionally, we distribute products
through foreign independent distributors, primarily in Europe, Asia, and South America, who then
sell the products to medical institutions. No customer or distributor accounted for ten percent or
more of net sales during 2009, 2008 or 2007. Foreign subsidiary sales are predominantly to
customers in Western Europe, Canada, Australia and Brazil and our foreign subsidiary assets are
located in the same countries. At the end of 2009 and 2008, consolidated accounts receivable
included $42.4 million and $36.1 million due from customers located outside of the United States.
The following table presents net sales and long-lived assets (excluding deferred taxes) by
geographical territory. No individual foreign country’s net sales or long-lived assets are
material.
F-39
Table of Contents
| (in thousands) | 2009 | 2008 | 2007 | |||||||||
United States |
||||||||||||
Net sales |
$ | 373,898 | $ | 355,678 | $ | 334,258 | ||||||
Long-lived assets |
824,886 | 833,095 | 872,318 | |||||||||
International |
||||||||||||
Net sales |
145,372 | 145,963 | 129,670 | |||||||||
Long-lived assets |
17,924 | 17,092 | 18,165 | |||||||||
13. Postretirement Benefits
We have an unfunded postretirement plan in the United States, which provides medical, dental, and
life insurance benefits at reduced rates to certain retirees and their eligible dependents.
Employees hired before 2000 are eligible if they meet age and service requirements and qualify for
retirement benefits. We provide funds to the plans as benefits are paid. We recognize the status of
our postretirement plan as a net asset or liability, with an offsetting adjustment to accumulated
other comprehensive income in shareholders’ equity.
The cost of our postretirement benefit plan (in thousands) was as follows:
| 2009 | 2008 | 2007 | ||||||||||
Service cost |
$ | 174 | $ | 183 | $ | 155 | ||||||
Interest cost |
208 | 208 | 175 | |||||||||
Amortization of net prior service cost |
62 | (38 | ) | (38 | ) | |||||||
Net benefit costs |
$ | 444 | $ | 353 | $ | 292 | ||||||
The following tables present reconciliations of the benefit obligation of the plan and the
plan assets of the plan (in thousands):
| 2009 | 2008 | |||||||
Change in benefit obligation |
||||||||
Benefit obligation at beginning of year |
$ | 3,886 | $ | 3,272 | ||||
Service cost |
174 | 183 | ||||||
Interest cost |
208 | 208 | ||||||
Actuarial (gain) loss |
(187 | ) | 386 | |||||
Benefit payments |
(217 | ) | (163 | ) | ||||
Benefit obligation at end of year |
$ | 3,864 | $ | 3,886 | ||||
Change in plan assets |
||||||||
Fair value of assets at beginning of year |
$ | — | $ | — | ||||
Actual return on plan assets |
— | — | ||||||
Employer contributions |
217 | 163 | ||||||
Benefit payments |
(217 | ) | (163 | ) | ||||
Fair value of plan assets at end of year |
$ | — | $ | — | ||||
Amounts recognized in the statement of financial position consist of:
| 2009 | 2008 | |||||||
Current |
$ | (233 | ) | $ | (263 | ) | ||
Long term |
(3,631 | ) | (3,623 | ) | ||||
Net amount of accrued benefit cost |
$ | (3,864 | ) | $ | (3,886 | ) | ||
Amounts recognized in accumulated other comprehensive income consist of:
| 2009 | 2008 | |||||||
Net actuarial loss (gain) |
$ | 7 | $ | 194 | ||||
Net prior service cost |
32 | 94 | ||||||
Total accumulated other comprehensive income |
$ | 39 | $ | 288 | ||||
The net prior service cost in accumulated other comprehensive income consists of two
components: one component for a negative plan amendment in 2000 (2000 component), and a second
component for a positive plan amendment
F-40
Table of Contents
in 2006 (2006 component). These two components are being amortized differently, based on expected
future service periods at the time of the amendment. The 2000 component was fully amortized in
2009, and in 2010 we estimate that the amortization of the 2006 component from accumulated other
comprehensive income will result in a charge to net periodic benefit cost for $0.0 million.
The benefits expected to be paid in each of the next five fiscal years and the aggregate for the
five fiscal years thereafter are projected as follows (in thousands):
Projected Medical Benefit Payments
2010 |
$ | 240 | ||
2011 |
251 | |||
2012 |
261 | |||
2013 |
295 | |||
2014 |
311 | |||
2015-2019 |
1,772 |
The assumptions used in estimating the annual cost related to these plans include:
| 2009 | 2008 | |||||||
Discount rate |
5.75 | % | 5.75 | % | ||||
Rate of future compensation increase |
4.00 | % | 4.00 | % | ||||
An average increase of 8.5 percent in the cost of covered health care benefits was assumed for
2009 and is projected to gradually decrease to 5.0 percent by 2016 and remain at that level
thereafter. Because of the subsidy caps, the assumed health care cost trend rates have a slight
effect on the amounts reported for our postretirement plan. A one-percentage-point change in the
assumed health care cost trend rates would have the following effects (in thousands):
| 1-Percentage- | 1-Percentage- | |||||||
| Point Increase | Point Decrease | |||||||
Effect on total of service and interest cost |
$ | 4 | $ | (4 | ) | |||
Effect on post-retirement benefit obligation |
44 | (41 | ) | |||||
14. Savings and Investment Plan
The AMS Savings and Investment Plan (the Plan) allows employees in the United States to contribute
a portion of their compensation to the Plan. We match a portion of these contributions.
Additionally, we make profit sharing contributions to the Plan based upon a percent of operating
profit as established annually by senior management and the Compensation Committee of the Board of
Directors. All of our United States employees who satisfy certain service requirements are eligible
to participate in the Plan. Matching contributions of $3.7 million, $3.3 million and $3.0 million
were made in 2009, 2008 and 2007, respectively. Profit sharing contributions were $5.0 million,
$3.7 million and $2.3 million in 2009, 2008 and 2007, respectively.
15. Income Taxes
Components of our income from continuing operations before income taxes are as follows (in
thousands):
| Pretax income | 2009 | 2008 | 2007 | |||||||||
U.S. |
$ | 127,504 | $ | 43,140 | $ | 11,323 | ||||||
Foreign |
8,531 | 7,018 | 5,310 | |||||||||
Total |
$ | 136,035 | $ | 50,158 | $ | 16,633 | ||||||
F-41
Table of Contents
Components of income tax expense for continuing operations are as follows (in thousands):
| Income tax expense | 2009 | 2008 | 2007 | |||||||||
Current |
||||||||||||
Federal |
$ | 40,762 | $ | 17,190 | $ | 433 | ||||||
State |
7,173 | 4,166 | 1,796 | |||||||||
Foreign |
3,336 | 2,002 | 1,708 | |||||||||
Deferred |
||||||||||||
Federal |
931 | (3,761 | ) | 6,384 | ||||||||
State |
(973 | ) | (791 | ) | 1,270 | |||||||
Foreign |
(32 | ) | 517 | 297 | ||||||||
Total |
$ | 51,197 | $ | 19,323 | $ | 11,888 | ||||||
A reconciliation of income tax expense for continuing operations computed at the United States
statutory rate to our provision for income taxes is as follows (in thousands):
| Income tax reconciliation | 2009 | 2008 | 2007 | |||||||||
Statutory rate |
$ | 47,612 | $ | 17,556 | $ | 5,821 | ||||||
State taxes |
4,030 | 2,194 | 1,994 | |||||||||
Manufacturing tax incentives |
(2,092 | ) | (851 | ) | — | |||||||
Meals and entertainment |
565 | 541 | 618 | |||||||||
Foreign rate differential and other |
759 | 80 | (64 | ) | ||||||||
Research and development credits |
(1,214 | ) | (917 | ) | (1,085 | ) | ||||||
Stock-based compensation |
327 | 738 | 1,098 | |||||||||
Audit settlements and refund claim |
— | (712 | ) | (852 | ) | |||||||
Litigation settlement |
— | — | 4,256 | |||||||||
Other |
1,210 | 694 | 102 | |||||||||
Total |
$ | 51,197 | $ | 19,323 | $ | 11,888 | ||||||
During 2009 and 2008, we recognized U.S. tax benefits related to a tax deduction for domestic
manufacturing activities, which resulted in a reduction in income tax expense of $2.1 million and
$0.9 million, respectively. During 2007, due to the utilization of acquired net operating loss
carryforwards, we did not realize a domestic manufacturing deduction benefit.
During 2008, we recognized $0.7 million in previously unrecognized tax benefits due to the
settlement of an income tax audit. During 2007, we recognized $0.9 million in previously
unrecognized tax benefits due to the settlement of an income tax audit.
During 2007, we recorded litigation settlement charges of $14.3 million, primarily due to the
arbitration award to the former shareholders of CryoGen, Inc. (see Note 3, Litigation Settlements),
with no related tax benefit.
F-42
Table of Contents
Deferred income taxes reflect the net tax effects of temporary differences between the carrying
amounts of assets and liabilities for financial reporting purposes and the amounts used for income
tax purposes. Significant components of deferred income tax assets and liabilities at the end of
2009 and 2008 are as follows (in thousands):
| 2009 | 2008 | |||||||
Deferred tax assets: |
||||||||
Federal net operating loss carryforwards |
$ | 8,938 | $ | 9,618 | ||||
Reserves and allowances |
11,315 | 9,730 | ||||||
Workforce, patents and license |
2,124 | 2,975 | ||||||
Compensation accruals |
4,165 | 3,888 | ||||||
Accrued state tax and interest |
2,207 | 1,593 | ||||||
Stock-based compensation |
6,619 | 5,464 | ||||||
State net operating loss carryforwards and
other credits |
3,362 | 4,387 | ||||||
Convertible debt issuance and transaction costs |
3,144 | 1,678 | ||||||
Other |
2,705 | 1,995 | ||||||
Valuation allowance |
— | (504 | ) | |||||
Total deferred tax assets |
44,579 | 40,824 | ||||||
Deferred tax
liabilities: |
||||||||
Goodwill |
11,076 | 9,685 | ||||||
Prepaid insurance and other |
1,979 | 1,286 | ||||||
Developed technology |
706 | 2,969 | ||||||
Trademarks and royalty agreements |
15,545 | 16,397 | ||||||
Contingent interest on debt |
62,750 | 47,623 | ||||||
Total deferred tax liabilities |
92,056 | 77,960 | ||||||
Net deferred tax liability |
$ | (47,477 | ) | $ | (37,136 | ) | ||
On January 2, 2010, we have tax effected foreign tax loss carryforwards of approximately $0.4
million with no expiration. Realization of future tax benefits related to these net deferred
assets is dependent on many factors, including the ability to generate taxable income in the
related jurisdictions. We believe that future foreign income will be sufficient to realize these
loss carryforwards and accordingly have not provided a valuation allowance against these assets.
We have U.S. federal tax loss carryforwards of approximately $25.5 million which are realizable
under IRC Section 382. They expire between 2018 and 2026. Management believes that future taxable
income will be sufficient to realize these tax loss carryforwards and has established a deferred
tax asset of $8.9 million.
As of January 2, 2010, undistributed earnings of international subsidiaries of approximately $17.6
million were considered to have been reinvested indefinitely and, accordingly, we have not provided
U.S. taxes on such earnings.
F-43
Table of Contents
Changes in
our unrecognized tax benefits (UTBs) are as follows:
| Unrecognized | ||||||||||||||||||||
| Gross | Income Tax | |||||||||||||||||||
| Federal, | Accrued | Gross UTBs, | Deferred | Benefits, Net of | ||||||||||||||||
| State and | Interest and | including | Federal and | Deferred Federal | ||||||||||||||||
| Foreign | Penalties on | interest and | State Income | and State | ||||||||||||||||
| (in thousands) | UTBs | UTBs | penalties | Tax Benefits | Benefits | |||||||||||||||
Balance at December 29, 2007 |
$ | 12,857 | $ | 557 | $ | 13,414 | $ | (1,766 | ) | $ | 11,648 | |||||||||
Additions for tax positions related to a prior period |
2,664 | 145 | 2,809 | 24 | 2,833 | |||||||||||||||
Additions for tax positions related to the current period |
944 | — | 944 | (97 | ) | 847 | ||||||||||||||
Reductions related to the closing of Statutes of Limitations |
— | — | — | — | — | |||||||||||||||
Reductions related to settlements with tax authorities |
(1,514 | ) | (326 | ) | (1,840 | ) | 246 | (1,594 | ) | |||||||||||
Balance at January 3, 2009 |
14,951 | 376 | 15,327 | (1,593 | ) | 13,734 | ||||||||||||||
Additions for tax positions related to a prior period |
1,500 | 604 | 2,104 | (547 | ) | 1,557 | ||||||||||||||
Additions for tax positions related to the current period |
993 | — | 993 | (144 | ) | 849 | ||||||||||||||
Reductions related to the closing of Statutes of Limitations |
(182 | ) | (36 | ) | (218 | ) | 77 | (141 | ) | |||||||||||
Reductions related to settlements with tax authorities |
— | — | — | — | — | |||||||||||||||
Balances at January 2, 2010 |
17,262 | 944 | 18,206 | (2,207 | ) | 15,999 | ||||||||||||||
Less: |
||||||||||||||||||||
UTBs attributable to timing items included above |
982 | — | 982 | — | 982 | |||||||||||||||
Total UTBs that, if recognized, would impact the
effective income tax rate as of January 2, 2010 |
$ | 16,280 | $ | 944 | $ | 17,224 | $ | (2,207 | ) | $ | 15,017 | |||||||||
We have classified all of our liability for unrecognized tax benefits as of January 2, 2010 as
a non-current liability, as no payments are anticipated within one year. We recognize accrued
interest and penalties related to unrecognized tax benefits in our tax provision in the
Consolidated Statements of Operations, which is consistent with the recognition of these items in
prior reporting periods. The balance of accrued interest and penalties at the reporting periods is
presented in the table above.
We have no federal income tax audits under way at the current time. Our federal returns are
subject to examination for 2006 and subsequent years.
State and foreign income tax returns are generally subject to examination for a period of three to
five years after filing of the respective return. The state impact of any federal changes remains
subject to examination by various states for a period of up to one year after formal notification
to the states.
We do not expect any significant change in the amount of unrecognized tax benefits during the next
12 months.
16. Subsequent Events
On February 16, 2010, we sold our Her Option® global endometrial cryoablation product line for
treating excessive uterine bleeding to CooperSurgical, Inc., a subsidiary of The Cooper Companies,
Inc., for $20.5 million. The final sale price is subject to adjustment based on working capital
balances at the time of sale.
17. Quarterly Financial Data (unaudited; in thousands, except per share data)
The following table presents quarterly financial data for 2009 and 2008. In our opinion, this
quarterly information has been prepared on the same basis as the consolidated financial statements
and includes all adjustments (consisting only of normal recurring adjustments) necessary for a fair
presentation of the unaudited quarterly results.
F-44
Table of Contents
| 2009 | 2008 | |||||||||||||||||||||||||||||||
| First | Second | Third | Fourth | First | Second | Third | Fourth | |||||||||||||||||||||||||
| 13 weeks | 13 weeks | 13 weeks | 13 weeks | 13 weeks | 13 weeks | 13 weeks | 14 weeks | |||||||||||||||||||||||||
Net sales |
$ | 123,638 | $ | 126,388 | $ | 123,231 | $ | 146,013 | $ | 120,362 | $ | 129,797 | $ | 117,468 | $ | 134,014 | ||||||||||||||||
Gross profit |
100,296 | 104,780 | 101,947 | 120,036 | 91,372 | 100,612 | 92,605 | 105,955 | ||||||||||||||||||||||||
Operating income |
30,093 | 33,700 | 33,207 | 45,083 | 20,489 | 28,093 | 17,557 | 21,242 | ||||||||||||||||||||||||
Net income |
17,080 | 16,865 | 28,621 | 22,272 | 6,087 | 11,818 | 3,505 | 9,425 | ||||||||||||||||||||||||
Net income per share: |
||||||||||||||||||||||||||||||||
Basic net income |
$ | 0.23 | $ | 0.23 | $ | 0.39 | $ | 0.30 | $ | 0.08 | $ | 0.16 | $ | 0.05 | $ | 0.13 | ||||||||||||||||
Diluted net income |
$ | 0.23 | $ | 0.23 | $ | 0.38 | $ | 0.30 | $ | 0.08 | $ | 0.16 | $ | 0.05 | $ | 0.13 | ||||||||||||||||
In the first quarter of 2009, we recorded a gain of $4.6 million related to the extinguishment
of a portion of our 2036 Notes (see Note 7, Debt). Royalty income during the second quarter of
2009 decreased $1.6 million due to a one-time royalty payment of $1.5 million in 2008 related to a
portion of our urinary incontinence technology acquired in 2006. In the third quarter of 2009, we
recorded $17.4 million gain on the sale of our Ovion technology to Conceptus. In addition, during
the third quarter of 2009 we exchanged $250.0 million of principal of 2036 Notes for $250.0 million
of principal of 2041 Notes. We accounted for this transaction as an extinguishment of debt, and we
recorded a pre-tax gain on extinguishment of $5.6 million (see Note 7, Debt).
During the second quarter of 2008, we recorded a charge of $1.1 million for the final negotiated
settlement of distributor terminations related to the Laserscope acquisition. Royalty income
during the quarter included a one-time royalty payment of $1.5 million related to a portion of our
urinary incontinence technology acquired in 2006. In addition, we recorded a $1.2 million gain
resulting from Iridex’s payment of receivables related to our disposal of the Laserscope aesthetics
business, for which we had previously established reserves. In the third quarter of 2008, we
recorded $7.5 million of one-time in-process research and development (IPR&D) charges related to a
milestone payment to BioControl Medical, Ltd. (BioControl), from whom we acquired certain issued
patents and other assets during 2006. Also during the quarter, other income (expense) decreased
approximately $1.7 million compared to the third quarter of 2007, primarily due to the impact of
fluctuations in foreign currencies, mainly the Euro, against the U.S. dollar on foreign denominated
inter-company receivables and payables. Partially offsetting this fluctuation were net gains from
final payments and adjustments related to our disposal of the Laserscope aesthetics business, gains
on the settlement of certain derivative contracts, and income from license and milestone fees from
an agreement to license one of our technologies. During the fourth quarter of 2008, we recorded an
acceleration of amortization of $17.1 million related to certain developed technology intangible
assets for which the carrying value of the assets exceeded the present value of our forecasted cash
flows related to the technologies (see Note 5, Goodwill and Intangible Assets). Also during the
fourth quarter, we recorded a gain of $5.6 million related to the extinguishment of a portion of
our 2036 Notes (see Note 7, Debt).
Quarterly and annual earnings per share are calculated independently based on the weighted average
number of shares outstanding during the period.
Sales and operating results have varied and are expected to continue to vary significantly from
quarter to quarter as a result of seasonal patterns, with the first and third quarters of each year
typically having lower sales and the fourth quarter of each year typically having the highest
sales.
F-45
Table of Contents
Financial
Statement Schedule — Schedule II — Valuation and Qualifying Accounts.
This schedule of valuation and qualifying accounts (in thousands) should be read in conjunction
with the consolidated financial statements. These amounts exclude the aesthetics business, which
was classified as part of liabilities of discontinued operations prior to its sale on January 16,
2007. All other schedules are omitted because they are not applicable or the required information
is shown in the financial statements or notes thereto.
| Balance at | Additions charged to: | Balance at | ||||||||||||||||||
| Beginning of | Costs and | Other | End of | |||||||||||||||||
| Period | Expenses | Accounts | Deductions | Period | ||||||||||||||||
Valuation Accounts: |
||||||||||||||||||||
Year ended December 29, 2007 |
||||||||||||||||||||
Deducted from asset accounts |
||||||||||||||||||||
Allowance for doubtful accounts |
$ | 3,115 | $ | 950 | $ | — | $ | 967 | (1) | $ | 3,098 | |||||||||
Allowance for obsolete inventories |
$ | 2,504 | $ | 2,487 | $ | — | $ | 2,103 | (2) | $ | 2,888 | |||||||||
Allowance for sales returns |
$ | 1,816 | $ | 13,814 | (4) | $ | — | $ | 13,319 | (3),(4) | $ | 2,311 | ||||||||
Year ended January 3, 2009 |
||||||||||||||||||||
Deducted from asset accounts |
||||||||||||||||||||
Allowance for doubtful accounts |
$ | 3,098 | $ | 1,832 | $ | — | $ | 1,394 | (1) | $ | 3,536 | |||||||||
Allowance for obsolete inventories |
$ | 2,888 | $ | 5,737 | $ | — | $ | 4,390 | (2) | $ | 4,235 | |||||||||
Allowance for sales returns |
$ | 2,311 | $ | 13,664 | (4) | $ | — | $ | 13,398 (3), | (4) | $ | 2,577 | ||||||||
Year ended January 2, 2010 |
||||||||||||||||||||
Deducted from asset accounts |
||||||||||||||||||||
Allowance for doubtful accounts |
$ | 3,536 | $ | 1,648 | $ | — | $ | 1,745 | (1) | $ | 3,439 | |||||||||
Allowance for obsolete inventories |
$ | 4,235 | $ | 4,281 | $ | — | $ | 3,485 | (2) | $ | 5,031 | |||||||||
Allowance for sales returns |
$ | 2,577 | $ | 10,996 | $ | — | $ | 11,434 (3) | $ | 2,139 | ||||||||||
Qualifying Accounts: |
||||||||||||||||||||
Year ended December 29, 2007 |
||||||||||||||||||||
Product liability allowance |
$ | 548 | $ | 797 | $ | — | $ | 450 | (5) | $ | 895 | |||||||||
Accrued warranty expense |
$ | 2,715 | $ | 5,844 | (7) | $ | — | $ | 5,558 (6), | (7) | $ | 3,001 | ||||||||
Year ended January 3, 2009 |
||||||||||||||||||||
Product liability allowance |
$ | 895 | $ | 201 | $ | — | $ | 316 | (5) | $ | 780 | |||||||||
Accrued warranty expense |
$ | 3,001 | $ | 5,577 | (7) | $ | — | $ | 5,291 (6), | (7) | $ | 3,287 | ||||||||
Year ended January 2, 2010 |
||||||||||||||||||||
Product liability allowance |
$ | 780 | $ | 837 | $ | — | $ | 627 | (5) | $ | 990 | |||||||||
Accrued warranty expense |
$ | 3,287 | $ | 1,517 | $ | — | $ | 2,511 | (6) | $ | 2,293 | |||||||||
Notes:
| (1) | Uncollectable accounts written off, net of recoveries | |
| (2) | Obsolete and excess inventory disposals | |
| (3) | Returned product | |
| (4) | Includes activity under capital equipment upgrade and even exchange programs, which has increased with the growth in our capital equipment business. | |
| (5) | Product liability claims | |
| (6) | Product warranty claims | |
| (7) | Includes reserves and claims for product rework issues related to the Greenlight HPS® console. |
F-46
Table of Contents
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the
registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto
duly authorized.
| Dated: March 1, 2010 |
AMERICAN MEDICAL SYSTEMS HOLDINGS, INC. |
|||
| By | /s/ Anthony P. Bihl, III | |||
| Anthony P. Bihl, III | ||||
| President and Chief Executive Officer | ||||
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed
below on March 1, 2010 by the following persons on behalf of the registrant and in the capacities
indicated.
| Signature | Title | |
/s/
Anthony P. Bihl, III
|
President and Chief Executive Officer | |
| (Principal Executive Officer) | ||
/s/ Mark A. Heggestad
|
Executive Vice President and Chief Financial Officer | |
| (Principal Financial and Accounting Officer) | ||
/s/ Richard B. Emmitt
|
Director | |
/s/ Albert Jay Graf
|
Director | |
/s/ Jane E. Kiernan
|
Director | |
/s/ Robert McLellan, M.D.
|
Director | |
/s/ Christopher H. Porter,
Ph.D.
|
Director | |
/s/ D. Verne Sharma
|
Director | |
/s/ Thomas E. Timbie
|
Director | |
F-47
Table of Contents
AMERICAN MEDICAL SYSTEMS HOLDINGS, INC.
EXHIBIT INDEX TO ANNUAL REPORT
ON FORM 10-K
For the Year Ended January 2, 2010
| Item No. | Item | Filing Method | ||
2.1
|
Agreement and Plan of Merger, dated as of June 3, 2005, by and among American Medical Systems, Inc., Oak Merger Corp., Ovion Inc., Jeffrey P. Callister, and W. Stephen Tremulis, as Principal Stockholders, and Jeffrey P. Callister, as Stockholders’ Representative. | Incorporated by reference to Exhibit 10.1 of the Company’s Form 8-K filed on June 6, 2005 (File No. 000-30733). | ||
2.2
|
Asset Purchase Agreement, dated April 26, 2006, between American Medical Systems, Inc. and BioControl Medical, Ltd. | Incorporated by reference to Exhibit 10.1 of the Company’s Form 8-K filed on April 27, 2006 (File No. 000-30733). | ||
2.3
|
First Amendment to Asset Purchase Agreement, dated August 8, 2008, by and between American Medical Systems, Inc. and Bio Control Medical (B.C.M.), Ltd. | Incorporated by reference to Exhibit 10.1 of the Company’s Form 8-K filed on August 19, 2008 (File No. 000-30733). | ||
2.4
|
Agreement and Plan of Merger, dated as of May 8, 2006, by and among American Medical Systems, Inc., Xenon Merger Corp., a wholly owned subsidiary of American Medical Systems, Inc., Solarant Medical, Inc., and Warburg Pincus Equity Partners, L.P., as stockholders’ representative. | Incorporated by reference to Exhibit 10.1 of the Company’s Form 8-K filed on May 9, 2006 (File No. 000-30733). | ||
2.5
|
Asset Purchase Agreement, dated November 30, 2006, by and among American Medical Systems, Inc., Laserscope, and Iridex Corporation. | Incorporated by reference to Exhibit 10.1 of the Company’s Form 8-K filed on December 6, 2006 (File No. 000-30733). | ||
2.6
|
Asset Purchase Agreement, dated as of September 30, 2009, between American Medical Systems, Inc. and Conceptus, Inc. | Incorporated by reference to Exhibit 2.1 of the Company’s Form 10-Q for the Fiscal Quarter Ended October 3, 2009 (File No. 000-30733). | ||
2.7
|
Asset Purchase Agreement, dated as of February 16, 2010, between American Medical Systems, Inc. and CooperSurgical, Inc. | Incorporated by reference to Exhibit 2.1 of the Company’s Form 8-K filed on February 22, 2010 (File No. 000-30733). | ||
3.1
|
Second Amended and Restated Certificate of Incorporation of the Company. | Incorporated by reference to Exhibit 3.1 of the Company’s Form S-3 filed on June 19, 2006 (File No. 333-135135). | ||
3.2
|
Bylaws, as amended, of the Company. | Incorporated by reference to Exhibit 3.2 of the Company’s Form 10-K for the Fiscal Year Ended January 3, 2004 (File No. 000-30733). |
E-1
Table of Contents
| Item No. | Item | Filing Method | ||
4.1
|
Certificate of Incorporation of the Company. | See Exhibit 3.1 above. | ||
4.2
|
Bylaws, as amended, of the Company. | See Exhibit 3.2 above. | ||
4.3
|
Form of Indenture for Senior Debt Securities. | Incorporated by reference to Exhibit 4.2 of the Company’s Form S-3 filed on August 12, 2009 (File No. 333-161290). | ||
4.4
|
Form of Senior Debt Security. | Incorporated by reference to Exhibit 4.3 of the Company’s Form S-3 filed on August 12, 2009 (File No. 333-161290). | ||
4.5
|
Form of Indenture for Subordinated Debt Securities. | Incorporated by reference to Exhibit 4.4 of the Company’s Form S-3 filed on August 12, 2009 (File No. 333-161290). | ||
4.6
|
Form of Subordinated Debt Security. | Incorporated by reference to Exhibit 4.5 of the Company’s Form S-3 filed on August 12, 2009 (File No. 333-161290). | ||
4.7
|
Indenture, dated as of June 27, 2006, between American Medical Systems Holdings, Inc., the Notes Guarantors (as defined therein), and U.S. Bank National Association, as trustee. | Incorporated by reference to Exhibit 4.1 of the Company’s Form 8-K filed on June 28, 2006 (File No. 000-30733). | ||
4.8
|
Form of 3 1/4% Convertible Senior Subordinated Note. | Incorporated by reference to Exhibit 4.2 of the Company’s Form 8-K filed on June 28, 2006 (File No. 000-30733). | ||
4.9
|
First Supplemental Indenture, dated as of September 6, 2006, by and between Laserscope and U.S. Bank National Association, as trustee. | Incorporated by reference to Exhibit 4.1 of the Company’s Form 8-K filed on September 8, 2006 (File No. 000-30733). | ||
4.10
|
Guarantee, dated as of September 6, 2006, made by Laserscope in favor of U.S. Bank National Association, as trustee. | Incorporated by reference to Exhibit 4.2 of the Company’s Form 8-K filed on September 8, 2006 (File No. 000-30733). | ||
4.11
|
Indenture, dated as of September 21, 2009, between American Medical Systems Holdings, Inc., the Subsidiary Guarantors (as defined therein), and U.S. Bank National Association, as trustee. | Incorporated by reference to Exhibit 4.1 of the Company’s Form 8-K filed on September 22, 2009 (File No. 000-30733). | ||
4.12
|
Form of 4.00% Convertible Senior Subordinated Note due 2041. | Incorporated by reference to Exhibit 4.2 of the Company’s Form 8-K filed on September 22, 2009 (File No. 000-30733). | ||
4.13
|
Form of Subsidiary Guarantee entered into by each of the Subsidiary Guarantors. | Incorporated by reference to Exhibit 4.3 of the Company’s Form 8-K filed on September 22, 2009 (File No. 000-30733). |
E-2
Table of Contents
| Item No. | Item | Filing Method | ||
10.1
|
Employment Agreement, dated December 18, 2006, between Mark A. Heggestad and American Medical Systems, Inc. | Incorporated by reference to Exhibit 10.4 of the Company’s Annual Report on Form 10-K for the Fiscal Year Ended December 30, 2006 (File No. 000-30733). | ||
10.2
|
First Amendment to Employment Agreement, effective as of March 6, 2008, Mark A. Heggestad and American Medical Systems, Inc. | Incorporated by reference to Exhibit 10.7 of the Company’s Annual Report on Form 10-K for the Fiscal Year Ended January 3, 2009 (File No. 000-30733). | ||
10.3
|
Employment Agreement, dated as of April 22, 2008, between American Medical Systems, Inc. and Anthony P. Bihl, III. | Incorporated by reference to Exhibit 10.1 of the Company’s Form 8-K filed April 29, 2008 (File No. 000-30733). | ||
10.4
|
Employment Agreement, dated October 20, 2008, between Francois Georgelin and American Medical Systems Europe B.V. | Incorporated by reference to Exhibit 10.2 of the Company’s Form 10-Q for the Fiscal Quarter Ended July 4, 2009 (File No. 000-30733). | ||
10.5
|
Employment Agreement, effective as of April 1, 2009, between Joe W. Martin and American Medical Systems, Inc. | Incorporated by reference to Exhibit 10.3 of the Company’s Form 10-Q for the Fiscal Quarter Ended July 4, 2009 (File No. 000-30733). | ||
10.6
|
Employment Agreement, effective as of August 3, 2009, between Maximillian Fiore and American Medical Systems, Inc. | Incorporated by reference to Exhibit 10.2 of the Company’s Form 10-Q for the Fiscal Quarter Ended October 3, 2009 (File No. 000-30733). | ||
10.7
|
Separation Agreement, dated June 9, 2009, by and between Jan Dick and American Medical Systems, Inc. | Incorporated by reference to Exhibit 10.3 of the Company’s Form 10-Q for the Fiscal Quarter Ended October 3, 2009 (File No. 000-30733). | ||
10.8
|
2000 Equity Incentive Plan, as amended. | Incorporated by reference to Exhibit 10.1 of the Company’s Form 10-Q for the Fiscal Quarter Ended June 28, 2003 (File No. 000-30733). | ||
10.9
|
Form of Incentive Stock Option Agreement under the 2000 Equity Incentive Plan, as amended. | Incorporated by reference to Exhibit 10.10 of the Company’s Registration Statement on Form S-1 (File No. 333-37488). | ||
10.10
|
Form of Non-Qualified Stock Option Agreement under the 2000 Equity Incentive Plan, as amended. | Incorporated by reference to Exhibit 10.11 of the Company’s Registration Statement on Form S-1 (File No. 333-37488). | ||
10.11
|
Employee Stock Purchase Plan, as amended. | Incorporated by reference to Exhibit 10.2 of the Company’s Form 10-Q for the Fiscal Quarter Ended October 1, 2005 (File No. 000-30733). |
E-3
Table of Contents
| Item No. | Item | Filing Method | ||
10.12
|
2005 Stock Incentive Plan (As Amended and Restated). | Incorporated by reference to Exhibit 10.1 of the Company’s Form 10-Q for the Fiscal Quarter Ended April 4, 2009 (File No. 000-30733). | ||
10.13
|
Form of Stock Option Certificate for Directors under the 2005 Stock Incentive Plan (As Amended and Restated) | Incorporated by reference to Exhibit 10.20 of the Company’s Annual Report on Form 10-K for the Fiscal Year Ended December 31, 2005 (File No. 000-30733). | ||
10.14
|
Form of Stock Option Certificate for Executive Officers under the 2005 Stock Incentive Plan (As Amended and Restated) | Incorporated by reference to Exhibit 10.21 of the Company’s Annual Report on Form 10-K for the Fiscal Year Ended December 31, 2005 (File No. 000-30733). | ||
10.15
|
Form of Notice of Amendment to Stock Option Certificate/Agreement for Executive Officers of American Medical Systems Holdings, Inc. | Incorporated by reference to Exhibit 10.6 of the Company’s Form 10-Q for the Fiscal Quarter Ended July 2, 2006 (File No. 000-30733). | ||
10.16
|
Form of Stock Option Certificate for Executive Officers under the 2005 Stock Incentive Plan (Version Modified in 2010). | Incorporated by reference to Exhibit 10.1 of the Company’s Form 8-K filed on February 19, 2010 (File No. 000-30733). | ||
10.17
|
Form of Restricted Stock Award for Executive Officers under the 2005 Stock Incentive Plan. | Incorporated by reference to Exhibit 10.2 of the Company’s Form 8-K filed on February 19, 2010 (File No. 000-30733). | ||
10.18
|
Form of Indemnification Agreement with Executive Officers and Directors. | Incorporated by reference to Exhibit 10.22 of the Company’s Annual Report on Form 10-K for the Fiscal Year Ended December 31, 2005 (File No. 000-30733). | ||
10.19
|
Form of Change in Control Severance Agreement. | Incorporated by reference to Exhibit 10.3 of the Company’s Quarterly Report on Form 10-Q for the Fiscal Quarter Ended March 31, 2007 (File No. 000-30733). | ||
10.20
|
Form of First Amendment to Change in Control Severance Agreement. | Incorporated by reference to Exhibit 10.24 of the Company’s Annual Report on Form 10-K for the Fiscal Year Ended January 3, 2009 (File No. 000-30733). | ||
10.21
|
Form of Change in Control Severance Agreement (Version Modified in 2009). | Incorporated by reference to Exhibit 10.1 of the Company’s Form 10-Q for the Fiscal Quarter Ended July 4, 2009 (File No. 000-30733). |
E-4
Table of Contents
| Item No. | Item | Filing Method | ||
10.22
|
Change in Control Severance Agreement, dated as of April 22, 2008, between American Medical Systems Holdings, Inc. and Anthony P. Bihl, III. | Incorporated by reference to Exhibit 10.2 of the Company’s Form 8-K filed April 29, 2008 (File No. 000-30733). | ||
10.23
|
The American Medical Systems, Inc. Executive Severance Pay Plan. | Incorporated by reference to Exhibit 10.4 of the Company’s Form 8-K filed on February 19, 2010 (File No. 000-30733). | ||
10.24
|
Summary of Director Compensation. | Filed with this Annual Report on Form 10-K. | ||
10.25
|
Summary of Named Executive Officer Compensation (2009). | Incorporated by reference to Exhibit 10.28 of the Company’s Annual Report on Form 10-K for the Fiscal Year Ended January 3, 2009 (File No. 000-30733). | ||
10.26
|
Summary of Named Executive Officer Compensation (2010). | Filed with this Annual Report on Form 10-K. | ||
10.27
|
2010 Executive Variable Incentive Plan. | Incorporated by reference to Exhibit 10.3 of the Company’s Form 8-K filed on February 19, 2010 (File No. 000-30733). | ||
10.28
|
Amended and Restated License Agreement, dated January 1, 2008, between American Medical Systems, Inc. and BioControl Medical, Ltd. | Incorporated by reference to Exhibit 10.30 of the Company’s Annual Report on Form 10-K for the Fiscal Year Ended January 3, 2009 (File No. 000-30733). | ||
10.29
|
First Amendment to Amended and Restated License Agreement dated August 8, 2008, by and between American Medical Systems, Inc. and Bio Control Medical (B.C.M), Ltd. | Incorporated by reference to Exhibit 10.2 of the Company’s Form 8-K filed August 19, 2008 (File No. 000-30733). | ||
10.30
|
Credit and Guaranty Agreement, dated as of July 20, 2006, by and among American Medical Systems, Inc., as borrower, American Medical Systems Holdings, Inc. and certain of its subsidiaries, as guarantors, CIT Capital Securities LLC, as co-lead arranger and sole bookrunner, KeyBank National Association, as co-lead arranger and syndication agent, CIT Healthcare LLC, as administrative agent and collateral agent, General Electric Capital Corporation, as documentation agent, and various lenders. | Incorporated by reference to Exhibit 10.1 of the Company’s Form 8-K filed on July 26, 2006 (File No. 000-30733). | ||
10.31
|
First Amendment to Credit and Guaranty Agreement, dated as of October 29, 2007, by and among American Medical Systems, Inc., each of the other credit parties which is a signatory thereto and CIT Healthcare LLC, as administrative agent. | Incorporated by reference to Exhibit 10.1 of the Company’s Form 8-K filed on October 29, 2007 (File No. 000-30733). |
E-5
Table of Contents
| Item No. | Item | Filing Method | ||
10.32
|
Pledge and Security Agreement, dated as of July 20, 2006, between each of the grantors party thereto and CIT Healthcare LLC, as administrative agent and collateral agent. | Incorporated by reference to Exhibit 10.2 of the Company’s Form 8-K filed on July 26, 2006 (File No. 000-30733). | ||
10.33
|
Mortgage, Security Agreement, Assignment of Rents and Leases and Fixture Financing Statement, dated as of July 20, 2006, executed by American Medical Systems, Inc. to and for the benefit of CIT Healthcare LLC, as administrative agent and collateral agent. | Incorporated by reference to Exhibit 10.3 of the Company’s Form 8-K filed on July 26, 2006 (File No. 000-30733). | ||
10.34
|
Net Lease Agreement, dated as of June 20, 2000, by and between Laserscope and Realtec Properties. | Incorporated by reference to Exhibit 10.6 of Laserscope’s Annual Report on Form 10-K filed on March 28, 2001 (File No. 000-18053). | ||
10.35
|
Net Lease Agreement, dated as of October 18, 2000, by and between Laserscope and Realtec Properties. | Incorporated by reference to Exhibit 10.6A of Laserscope’s Annual Report on Form 10-K filed on March 28, 2001 (File No. 000-18053). | ||
10.36
|
Settlement Agreement, dated as of August 14, 2007, by and among Iridex Corporation, American Medical Systems, Inc. and Laserscope. | Incorporated by reference to Exhibit 10.1 of the Company’s Form 8-K filed on August 20, 2007 (File No. 000-30733). | ||
10.37
|
Consent and Second Amendment to Credit and Guaranty Agreement by and among American Medical Systems, Inc., each of the other Credit Parties which is a signatory thereto and CIT Healthcare LLC. | Incorporated by reference to Exhibit 99.1 of the Company’s Form 8-K filed on August 14, 2009 (File No. 000-30733). | ||
21.1
|
Subsidiaries of American Medical Systems Holdings, Inc. | Filed with this Annual Report on Form 10-K. | ||
23.1
|
Consent of Ernst & Young LLP. | Filed with this Annual Report on Form 10-K. | ||
31.1
|
Certification by Chief Executive Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | Filed with this Annual Report on Form 10-K. | ||
31.2
|
Certification by Chief Financial Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | Filed with this Annual Report on Form 10-K. | ||
32.1
|
Certification pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. | Filed with this Annual Report on Form 10-K. |
E-6
